Abstract
In the present study, an amperometric aflatoxin B1 sensor was constructed via modifying a nano-graphite carbon paste microelectrode with a cationic surfactant of cetyltrimethylammonium bromide (CTAB) and a perfluorosulfonic acid resin of Nafion through a simple and controllable electrochemical scanning method. The experiment results show that CTAB–Nafion composite film has a good catalytic effect on the electrochemical response of aflatoxin B1. The electrocatalytic mechanism was investigated with the aid of different analytical techniques, including square wave voltammetry, electrochemical impedance spectroscopy, chronocoulometry, energy-dispersive spectroscopy and scanning electron microscopy. Under the optimal conditions, the linear range of the sensor is from 0.1 nM to 100 nM, and its detection limit and sensitivity are 20 pM (S/N = 3) and (24.9 ± 1.51) μA/nM, respectively. The accurate and rapid detection of aflatoxin B1, which has strong carcinogenicity, is of great significance for food quality monitoring and the protection of human health. Therefore, finally, the sensor was used to detect the concentration of aflatoxin B1 in milk and soy sauce samples, and the favorable recovery results indicated its good application prospects.
1. Introduction
As a group of toxics, mutagenic, and carcinogenic secondary metabolites, aflatoxins constitute a serious threat to human and animal health [1]. Among the four main derivatives in aflatoxins, aflatoxin B1 (AFB1) is a kind of Group 1 carcinogen because of its widest distribution, strongest toxicity, and greatest harm [2,3]. AFB1 is often found in moldy foods including peanuts, walnuts, and the products of soybeans, rice, corn, macaroni, seasonings, milk, edible oil, etc. [4]. Therefore, to realize the rapid and accurate determination of AFB1 in food is of great biomedical significance.
Through the efforts of analytical chemists, various techniques have been developed and applied in the detection of AFB1, such as high-performance liquid chromatography (HPLC) [5,6], surface plasmon resonance (SPR) [7], enzyme-linked immunosorbent assay (ELISA) [8,9], surface enhanced Raman scattering (SERS) [10,11], infrared spectroscopy [12,13], thin layer chromatography (TLC) [14,15], electrochemical sensing, etc. Among the above methods, HPLC, SPR, ELISA and SERS have high sensitivity and precision; however, expensive equipment and complex sample pretreatment processes are usually needed, which limit their application in real-time detection. In comparison, the electrochemical sensing method has attracted more attention due to its unique advantages of simple operation, low cost, short analysis time, low sample and reagent consumption, ease of integration, etc. [16,17]. With the development of materials science, many AFB1 electrochemicals or biosensors with good performance have been constructed [18,19]. In the above reported sensors, the most widely used substrate is glassy carbon electrode, which has excellent physical and chemical stability, good conductivity, low thermal expansion coefficient and good airtightness. However, it is expensive, and its surface is easily contaminated but not easy to renew. Therefore, to reduce the costs and simplify the operations of the AFB1 sensors, looking for other, more suitable substrates is essential.
Carbon paste electrode (CPE) may be a good choice. It is a new type of electrode made from a mixture of conductive carbon powder and binder. CPEs have been widely used in electrochemical research due to their advantages, including simple preparation, low costs, easy surface renewal, low background current, and wide potential window [20,21]. Moreover, nano-graphite (NG) has emerged as a promising material for the development of sensitive electrochemical sensors due to its exceptional electronic, thermal, and mechanical properties [22]. However, the addition of non-conductive adhesives during the preparation process usually leads to their low conductivity and sensitivity. To solve this problem, one effective way is modifying the surface of CPE with functional materials, such as nanomaterials, conductive polymers, surfactants, etc.
Functionalizing the surface of an electrode with surfactants shows obvious advantages, including simple preparation, easy maintenance, and affordability. As a cationic surfactant with distinctive properties, cetyltrimethylammonium bromide (CTAB) has been applied widely in the field of electrochemical sensing [23,24,25,26,27]. Research shows that CTAB can not only improve the sensitivity and selectivity of sensors, but also promotes the charge movement between the analytes and the electrode surface [28]. Electrode pollution is the main factor of limiting the application of electrochemicals and biosensors. An effective way to solve the issue is the incorporation of non-fouling coatings in electrochemical systems. One of the most well-known non-fouling membranes is Nafion™ [29], which was developed by DuPont and possesses excellent ionic conductivity, chemical inertness, thermal stability, and ion selectivity [30]. Thus, it has been applied widely in constructing electrochemicals or biosensors [31].
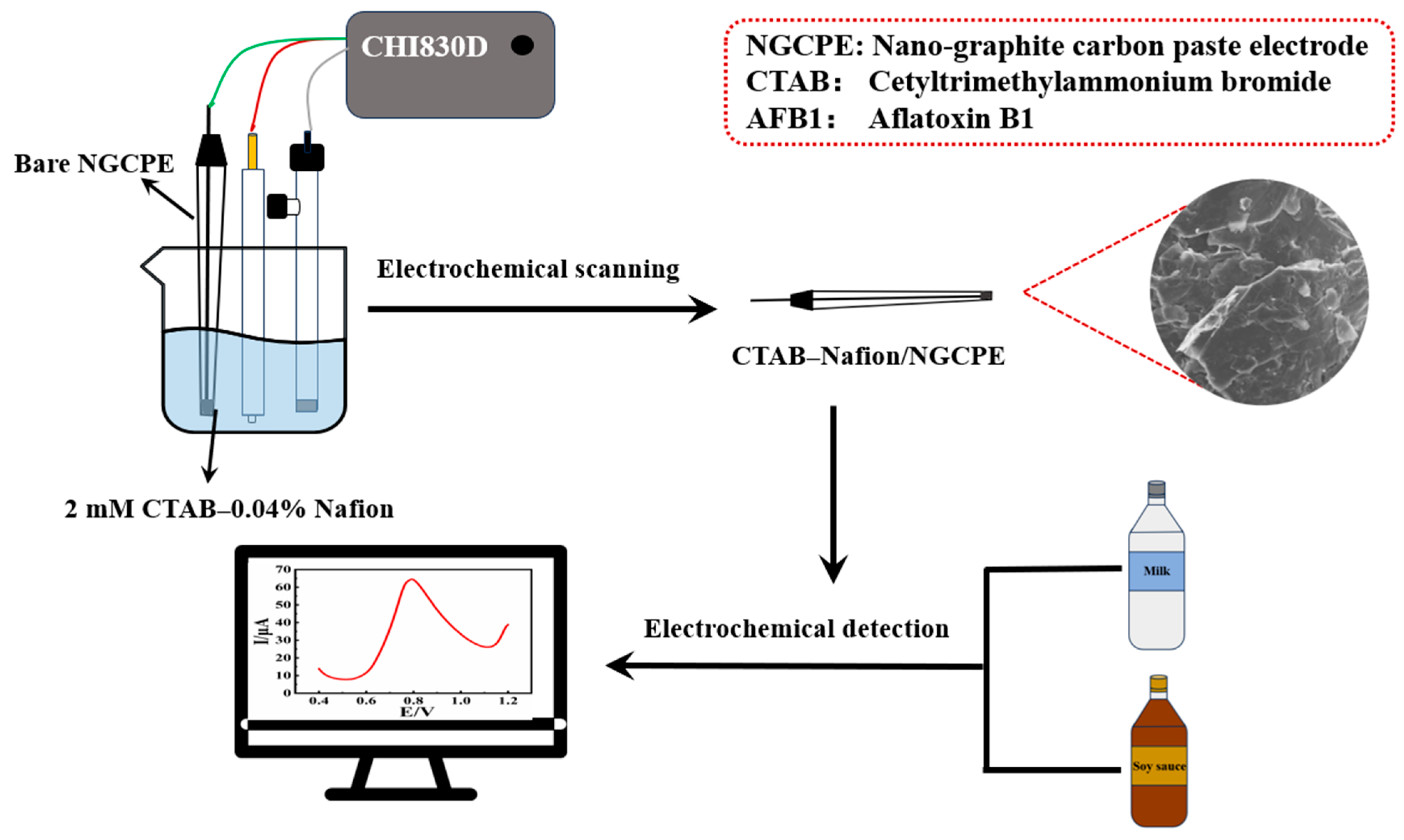
In this work, a nano-graphite carbon paste electrode (NGCPE) was homemade and modified with a CTAB–Nafion composite membrane through a simple electrochemical scanning method. The fabricated CTAB–Nafion/NGCPE shows an obvious catalytic effect on the electrochemical response of aflatoxin B1. The CTAB–Nafion/NGCPE was characterized with the aid of different techniques, such as scanning electron microscopy, energy-dispersive spectroscopy, Raman spectroscopy, electrochemical impedance spectroscopy, etc. The electrochemical experimental results demonstrated that the CTAB–Nafion/NGCPE has good sensitivity and selectivity, a short response time and good reproducibility, and could be applied successfully in the determination of aflatoxin B1 from milk and soy sauce. The schematic diagram of the preparation processes of CTAB–Nafion/NGCPE and its application is shown in Scheme 1.
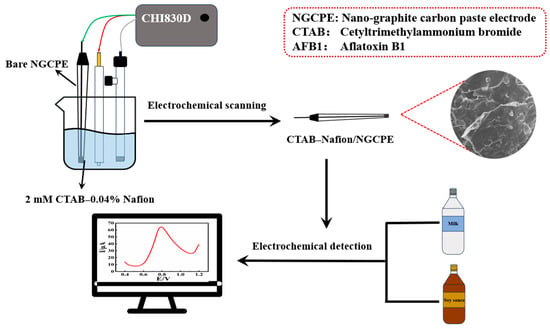
Scheme 1.
The schematic diagram of the preparation and application of CTAB–Nafion/NGCPE.
2. Experimental Section
2.1. Chemical and Reagents
Aflatoxin B1 (purity ≥ 98%), cetyltrimethylammonium bromide (CTAB, C19H42BrN, purity, 99%), and phosphate buffer solution powder were purchased from Shanghai Yuanye Biotechnology Co., Ltd. (Shanghai, China). Nano-graphite (∅ = 40 nm, purity, 99%) was purchased from Shanghai Aladdin Reagent Co., Ltd. (Shanghai, China). Ethanol, glucose (Glu), K3[Fe(CN)6] (purity ≥ 99.5%), K4[Fe(CN)6] (purity ≥ 99.5%), KCl (purity ≥ 99.5%), NaCl (purity ≥ 99.5%), and CaCl2 (purity ≥ 96%) were all purchased from Sinopharm Chemical Reagent Co., Ltd. (Beijing, China). Liquid paraffin was purchased from Jiangsu Qiangsheng Functional Chemical Co., Ltd. (Suzhou, China). Copper wire (∅ = 600 μm), milk and soy sauce were purchased from the local supermarket. All reagents were used without further purification. Ultra-pure water was used to prepare all the solutions.
2.2. Apparatus
An electrochemical workstation purchased from Chenhua Instrument Co., Ltd. (Shanghai, China) with a model number of CHI830D was used to carry out most of the electrochemical measurements. All the electrochemical experiments, including conditional optimization, mechanism study and performance evaluation, were finished through a three-electrode system. In the three-electrode system, the counter electrode is a platinum wire electrode, the working electrode is a bare or modified NGCPE, and the reference electrode is a saturated calomel electrode (SCE). A field emission scanning electron microscope with a model number of Hitachi SU8010 (Tokyo, Japan) was applied in carrying out the morphology characterization and element analysis of the electrode surface. The electrochemical impedance spectroscopy (EIS) was obtained on an electrochemical workstation with a model number of Zahner Zennium-PP211 (Kronach, Germany). The pH value of the solutions was adjusted by using a pHS-3E acidity meter purchased from Shanghai Youke Instrument Co., Ltd. (Shanghai, China). The ultra-pure water was prepared through a PGUV-10-AS pure water instrument purchased from Pinguan Instrument Equipment Co., Ltd. (Wuhan, China).
2.3. Preparation of Different Electrodes
2.3.1. Preparation of Bare NGCPE
The bare NGCPE was prepared by referring to the methods in the literature [32]. The first step is inserting a copper wire with a diameter of 600 μm into a pipette tip with a head diameter of 200 μm and fixing the tail of the pipette tip through a heating and melting method by using an alcohol lamp. Then, 50 mg nano-graphite powder and 6 µL liquid paraffin were manually ground in a small agate mortar until uniform to make the carbon paste. Finally, the bare NGCPE was prepared by packing the nano-graphite paste into the head cavity of the pipette tip, and manually polishing it on weighing paper until a smooth surface was obtained.
To refresh the surface of NGCPEs, the old paste was removed by sonication in ethanol for 3 min, followed by repacking with fresh nano-graphite paste.
2.3.2. Preparation of CTAB–Nafion/NGCPE
CTAB–Nafion/NGCPEs were prepared by immersing bare NGCPEs into a water solution containing 2.0 mM CTAB and 0.04% Nafion, and electrochemical scanning through cyclic voltammetry with a potential range of −1.6 V to +1.6 V, a scanning cycle number of 10 and a scan rate of 100 mV/s. Then, after washing the above electrodes with water and drying them at room temperature, CTAB–Nafion/NGCPEs were obtained.
3. Results and Discussion
3.1. Morphological Characterization of Different Electrodes
Scanning electron microscopy (SEM) was used to carry out the morphology characterization of the surface of bare NGCPE and CTAB–Nafion/NGCPE. The results shown in Figure 1 indicate that the surface of bare NGCPE appears compact (Figure 1A). In comparison, as shown in Figure 1B and the inset image in Figure 1B, the surface of CTAB–Nafion/NGCPE exhibits a rougher texture. The structure is profit for the increase in the electrode surface area and the enrichment quantity of AFB1, which plays an important role in improving the sensitivity of the sensor.

Figure 1.
SEM images of bare NGCPE (A) and CTAB–Nafion/NGCPE (B). The yellow dashed box in (B) presents a magnified view.
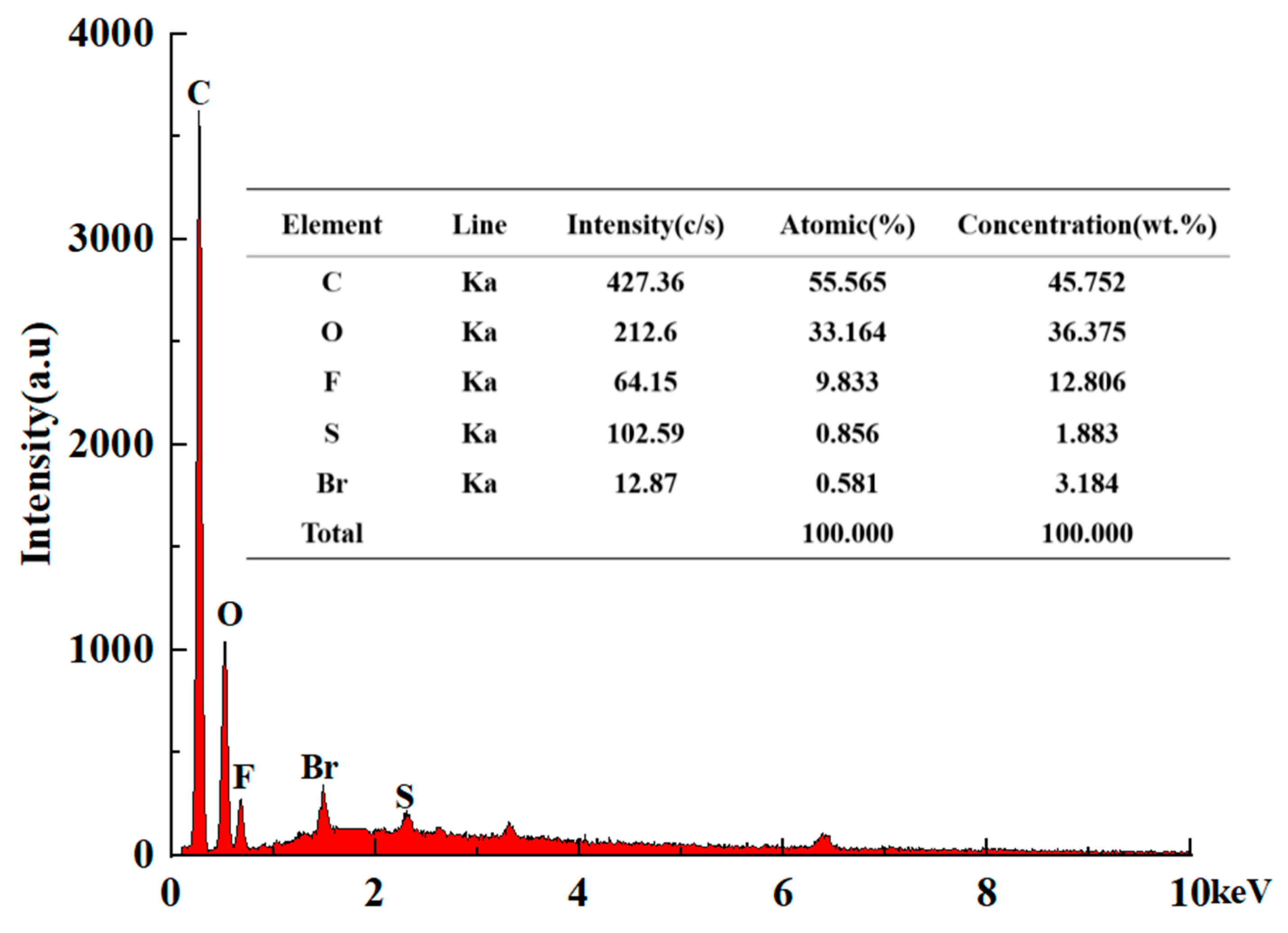
The energy-dispersive spectroscopy (EDS) technique was used to carry out the element analysis of CTAB–Nafion/NGCPE and the results are shown in Figure 2. Obviously, there are C, O, F, S, and Br elements on the surface of CTAB–Nafion/NGCPE. C element is from nano-graphite, paraffin oil, CTAB and Nafion, and its concentration is the highest. The second most abundant element is oxygen, which could be from Nafion and the oxidation of the electrode surface during the electrochemical scanning. F element and S element are from Nafion. Br element is from CTAB. It is a good evidence for the successful modification of CTAB and Nafion on the surface of NGCPE. Moreover, according to the concentration of S element and Br element, the concentration ratio of CTAB and Nafion on the electrode surface is about 0.25.
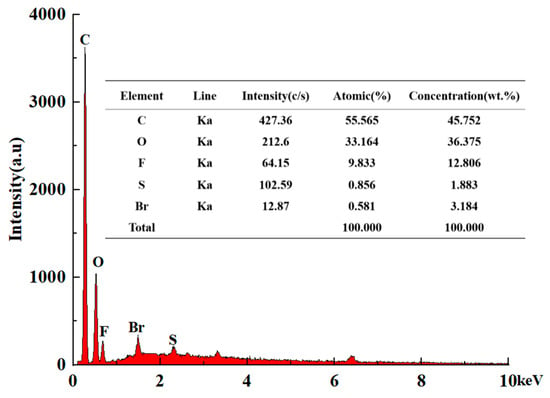
Figure 2.
EDS spectrum of CTAB–Nafion/NGCPE.
3.2. Electrochemical Behaviors of Different Electrodes
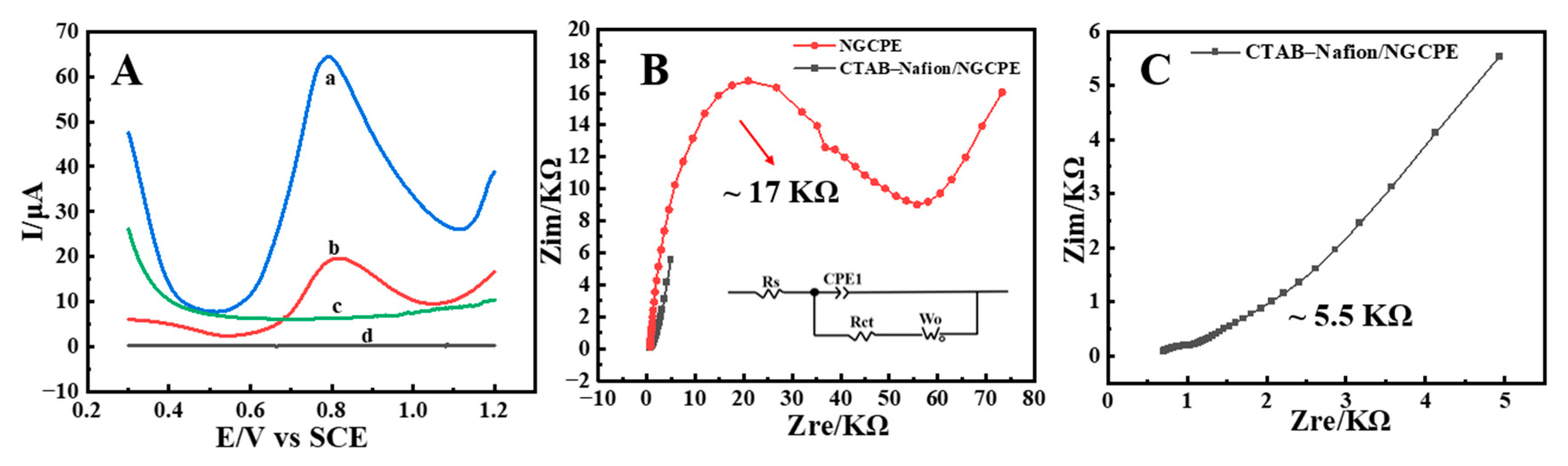
Both cyclic voltammetry (CV) and square wave voltammetry (SWV) are the commonly used techniques in the field of electrochemicals. In comparison, higher sensitivity screening can be obtained by SWV due to the minimal contribution of nonfaradaic currents [33]. Therefore, in the present research, SWV was used to evaluate the electrocatalytic capability of CTAB–Nafion/NGCPEs to AFB1 in 0.1 M PBS (pH = 5). From the results shown in Figure 3A, it could be observed that, in the absence of AFB1, almost no electrochemical response was observed on both bare NGCPE (curve d) and CTAB–Nafion/NGCPE (curve c). In the presence of 5 nM AFB1, a good electrochemical reduction peak with Ep = 0.812 V and Ip = 13.52 μA appeared on bare NGCPE (curve b), and a better response with Ep = 0.792 V and Ip = 45.88 μA was obtained on CTAB–Nafion/NGCPE (curve a). The higher sensitivity of the modified electrode indicated that the CTAB–Nafion composite has effective catalytical ability to stimulate the electrochemical response of AFB1, which may be due to the synergistic effect of CTAB and Nafion. Aflatoxin B1 and Nafion have strong hydrophobicity and are difficult to dissolve in water. In the structure of CTAB, there is a hydrophobic hexadecyl chain and a hydrophilic ammonium ion. The hydrophobic interaction between AFB1 and the hexadecyl chain of CTAB together with Nafion may play essential roles in the enrichment of AFB1 on the sensing interface [34].
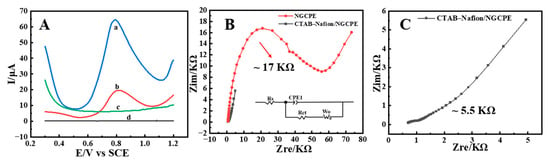
Figure 3.
Square wave voltammograms of bare NGCPE (b and d) and CTAB–Nafion/NGCPE (a and c) in 0.1 M PBS (pH = 5) with (a and b) or without (c and d) 5 nM AFB1 (A). EIS of bare NGCPE and CTAB–Nafion/NGCPE in 5.0 mM [Fe(CN)6] 3−/4− solution containing 0.1 M KCl (B). EIS of CTAB–Nafion/NGCPE (C).
As shown in Figure 3B,C, the effect of CTAB–Nafion on the voltammetric behavior of AFB1 was also investigated through electrochemical impedance spectroscopy (EIS) experiments. In a frequency range of 0.1 Hz to 100 kHz, the oscillation amplitude was adjusted to 5 mV. The work potential was optimized for the peak potential for each electrode versus saturated calomel electrode (SCE) employing [Fe(CN)6]3−/4− with a concentration of 5.0 mM as the probe. The equivalent circuit used for fit is depicted and was compatible with the Nyquist diagram. Here, RS, CPE, RCT and Wo represent solution resistance, a constant phase element that is corresponding to double-layer capacitance, the charge transfer resistance and the diffusion coefficient, respectively. According to the methods reported in the literature [35], the fitted values of Rct for bare NGCPE and CTAB–Nafion/NGCPE were 17 kΩ and 5.5 KΩ, respectively. The lower Rct value of CTAB–Nafion/NGCPE indicates that the charge-transfer process is faster when compared with bare NGCPE, which is in agreement with the voltammetric studies.
3.3. Optimizations
The results of the optimizations are shown in the supplementary material. The optimal conditions for the preparation and application of the sensor are as follows: CTAB–Nafion polymerization with 10 turns (Figure S1A); 50 s for accumulation time (Figure S1B), 0.1 V for accumulation potential (Figure S1C) and 5.0 for the pH value of the electrolyte (Figure S1D).
3.4. Influence of the Acid-Base Environment
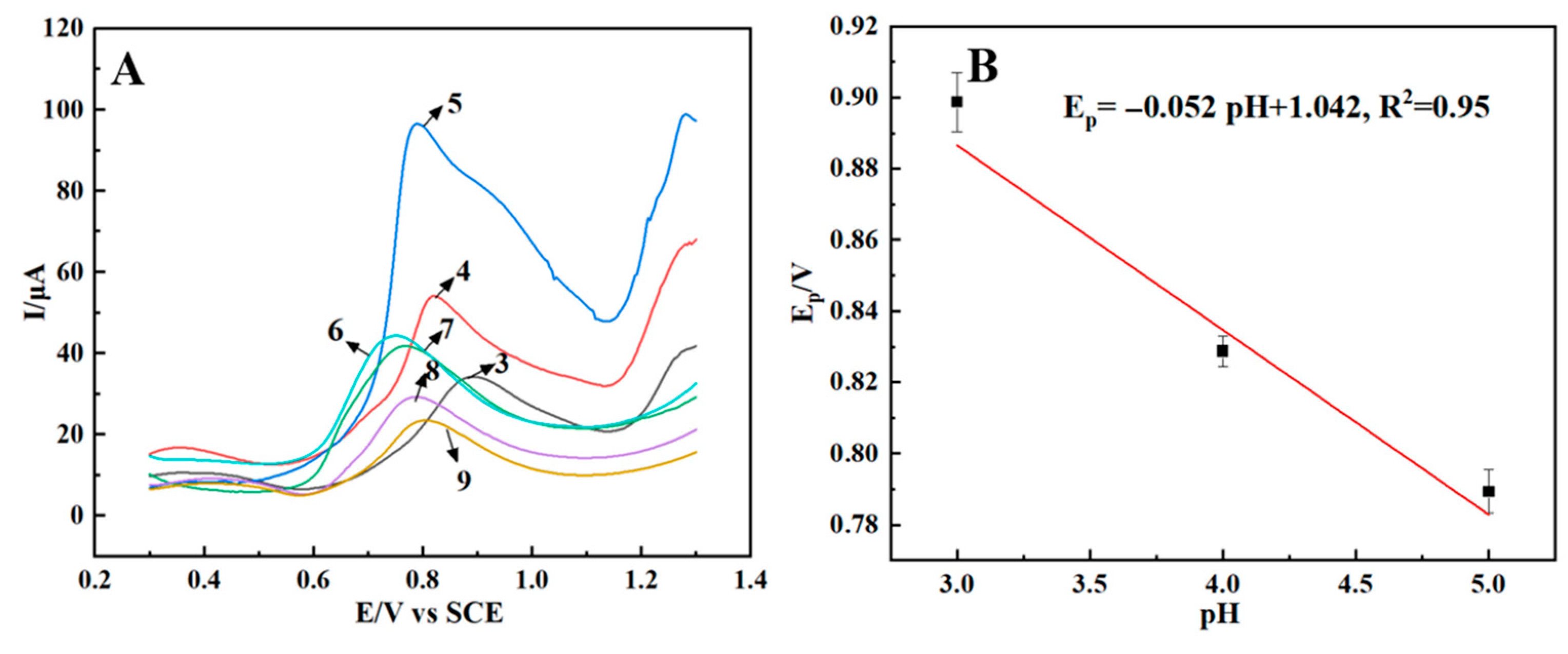
The detection acid-base environment can influence the charge states of both the sensing interface and the AFB1 molecules, thereby altering their electrostatic interactions. The influence of pH was investigated using square wave voltammetry (SWV), with the results presented in Figure 4A. When the pH value of the electrolyte changes from 3.0 to 5.0, the peak potential (Ep) of AFB1 shifted linearly towards more negative values with increasing pH, following the relationship Ep (V) = 1.042–0.052 pH (R2 = 0.95) (Figure 4B), which indicates that, in the electrochemical reaction process of AFB1 on the sensor, there are protons involved and the number of protons equals the number of electron transferred.
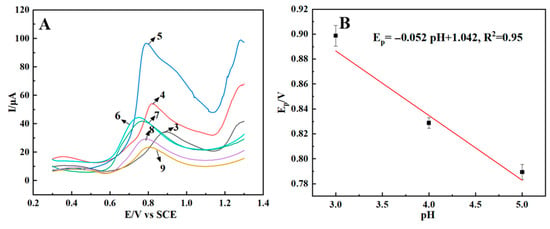
Figure 4.
Square wave voltammograms of 5 nM AFB1 on CTAB–Nafion/NGCPE in 0.1 M PBS with different pH values, and the number besides each line is the pH value of the supporting electrolyte (A). The linear relationship between the reduction peak potential of AFB1 and the pH value of electrolyte (B).
3.5. Influence of the Scan Rates
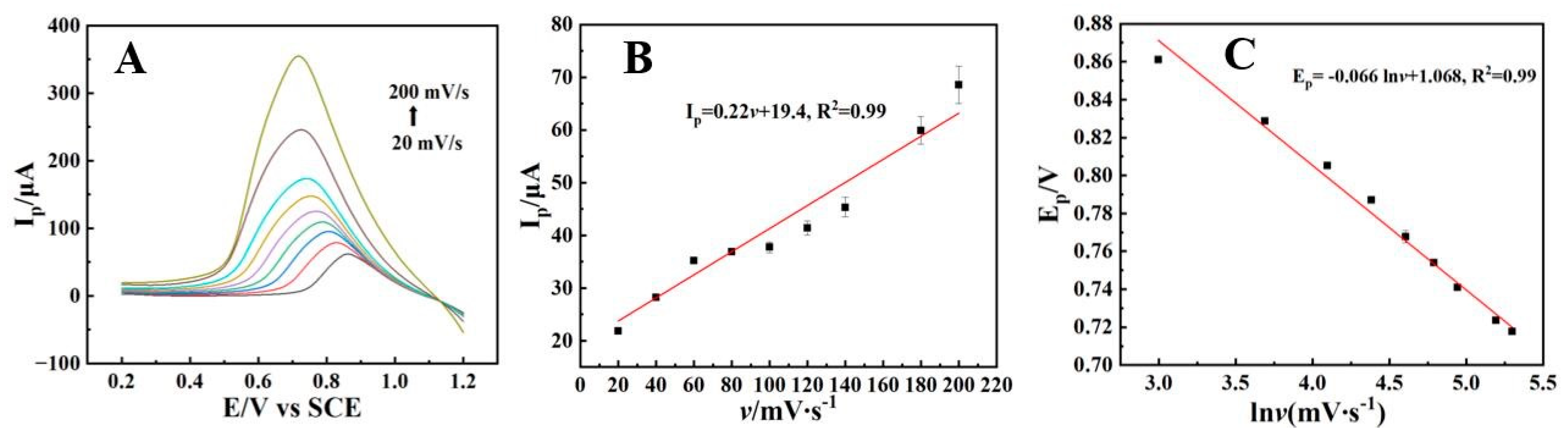
In order to reveal the electrochemical reaction mechanism of AFB1 on the sensor, linear sweep voltammetry (LSV) was used to investigate the influence of scan rates on the electrochemical behavior of AFB1 at CTAB–Nafion/NGCPE (Figure 5A). The data analysis results shown in Figure 5B show that the peak current (Ip) of AFB1 exhibits a good linear relationship with the scan rate (v) in the scan rate range of 20–200 mV/s: Ip (μA) = 0.22 v (mV/s) + 19.4 (R2 = 0.99), indicating that the electrochemical reaction of AFB1 at the CTAB–Nafion/NGCPE is an adsorption-controlled process. According to Laviron’s theory [36], the redox peak potential shows a linear relationship with lnv, expressed as the following equation:
Ep = E0′ + (RT/αnF)ln(RTK0/αnF) − (RT/αnF)ln(v/mV/s)
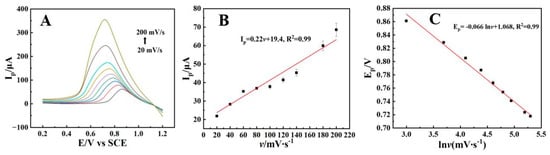
Figure 5.
Linear sweep voltammograms of 5 nM AFB1 on the CTAB–Nafion/NGCPE at different scan rates and from inner to outer the scan rates are 20, 40, 60, 80, 100, 120, 140, 180 and 200 mV/s, respectively (A). The linear dependence of the peak current (Ip) on the scan rate (v) (B). The linear relationship between the reduction peak potential (Ep) and the natural logarithm of the scan rate (lnv) (C).
In Equation (1), E0′ is the formal potential, T is the absolute temperature, n is the number of electrons transferred, F is the Faraday constant, is the electron transferring coefficient, K0 is the standard rate constant and D is the diffusion coefficient.
Based on the data summarized in Figure 5C, the relationship between Ep and lnv is followed the equation: Ep (V) = 0.066 lnv (mV/s) + 1.068 (R2 = 0.99). By combining this result with Equation (1), the number of electrons transferred (n) during the electrochemical reaction of AFB1 at the electrode surface is calculated to be approximately 1. It shows that the electrochemical reduction in AFB1 on the CTAB–Nafion/NGCPE is an adsorption-controlled process involving the transfer of a single electron. Therefore, on the sensor, the electrochemical reaction mechanism of AFB1 is speculated as follows, which is consistent with the literature [37].
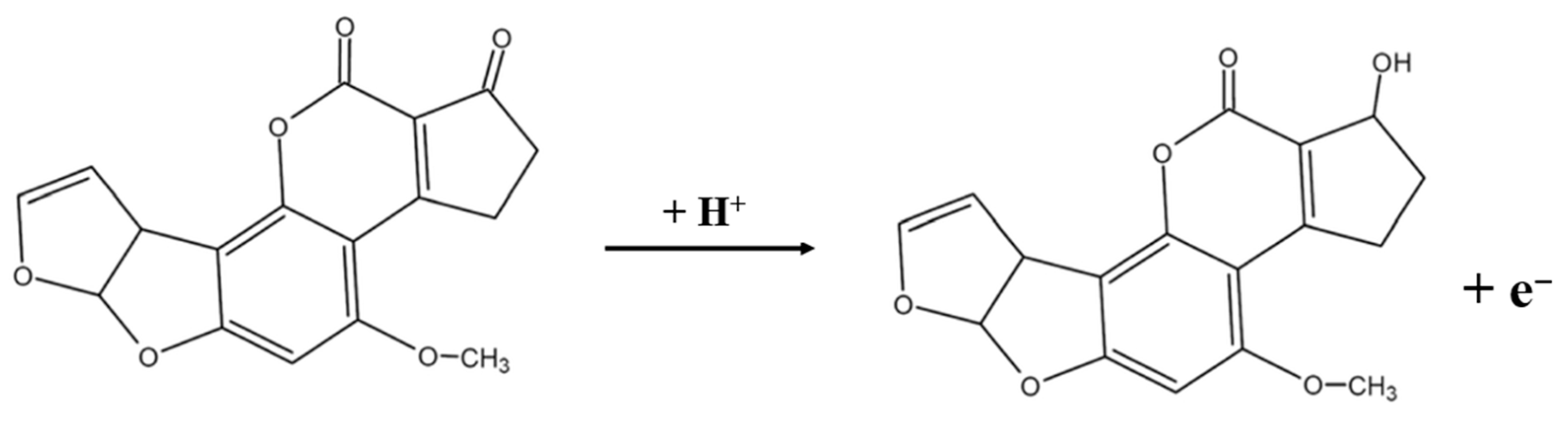
3.6. Adsorption Quantity of AFB1 on Different Electrodes
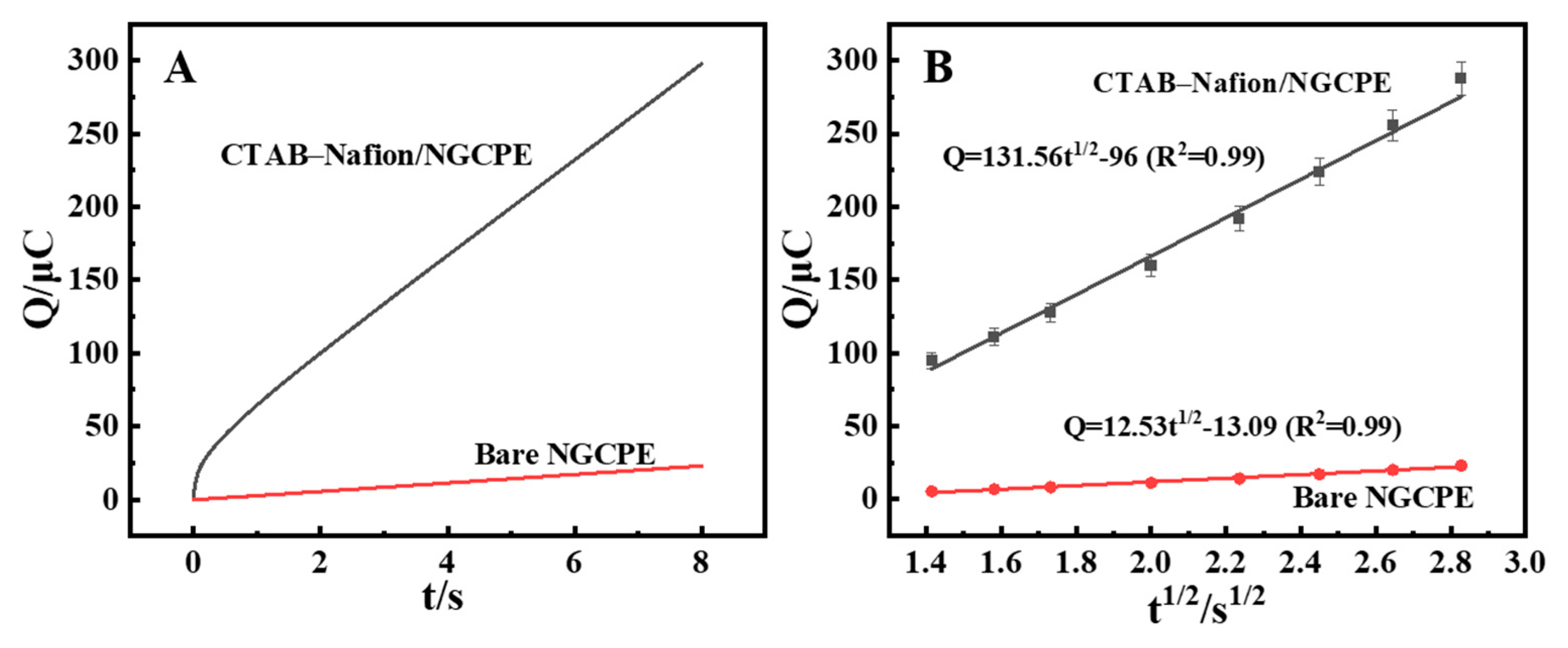
Chronocoulometry (CC) was employed to compare the enrichment quantity of AFB1 on the surface of bare NGCPE and CTAB–Nafion/NGCPE (Figure 6A). Further investigation confirmed that the CTAB–Nafion/NGCPE facilitates greater accumulation of AFB1 compared to the bare NGCPE, as evidenced by the results presented in Figure 6B. The charge (Q) associated with the electrode reaction exhibited a proportional relationship with the square root of time (t1/2). This observation aligns with the Cottrell equation, which describes the relationship between the amount of adsorbed substance (Q) and the square root of the reaction time (t1/2) as follows [38]:
Q = 2ncFAD01/2t1/2π−1/2 + Qdl + nFAτ0
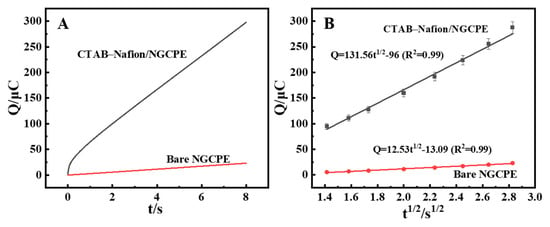
Figure 6.
Chronocoulometric curves of bare NGCPE and CTAB–Nafion/NGCPE in 0.1 M PBS (pH = 5.0) containing 5 nM AFB1 (A). The linear relationship between Q and t1/2 (B).
In Equation (2), F is the Faraday constant, c is the concentration of AFB1, n is the number of electrons transferred, A is the area of electrode surface, D0 is the diffusion coefficient, and Qdl is the electric double layer charge. Combining the results in Figure 6B and Equation (2), the surface adsorption amounts of AFB1 on CTAB–Nafion/NGCPE (1.22 × 10−7 mol/cm2) was calculated to be more than 3-fold that on bare NGCPE (3.66 × 10−8 mol/cm2), which may be due to the larger surface area of the CTAB–Nafion/NGCPE (Figure S2) [39].
3.7. Performances of the AFB1 Sensor
3.7.1. Linear Range, Sensitivity, Detection Limit and Response Time
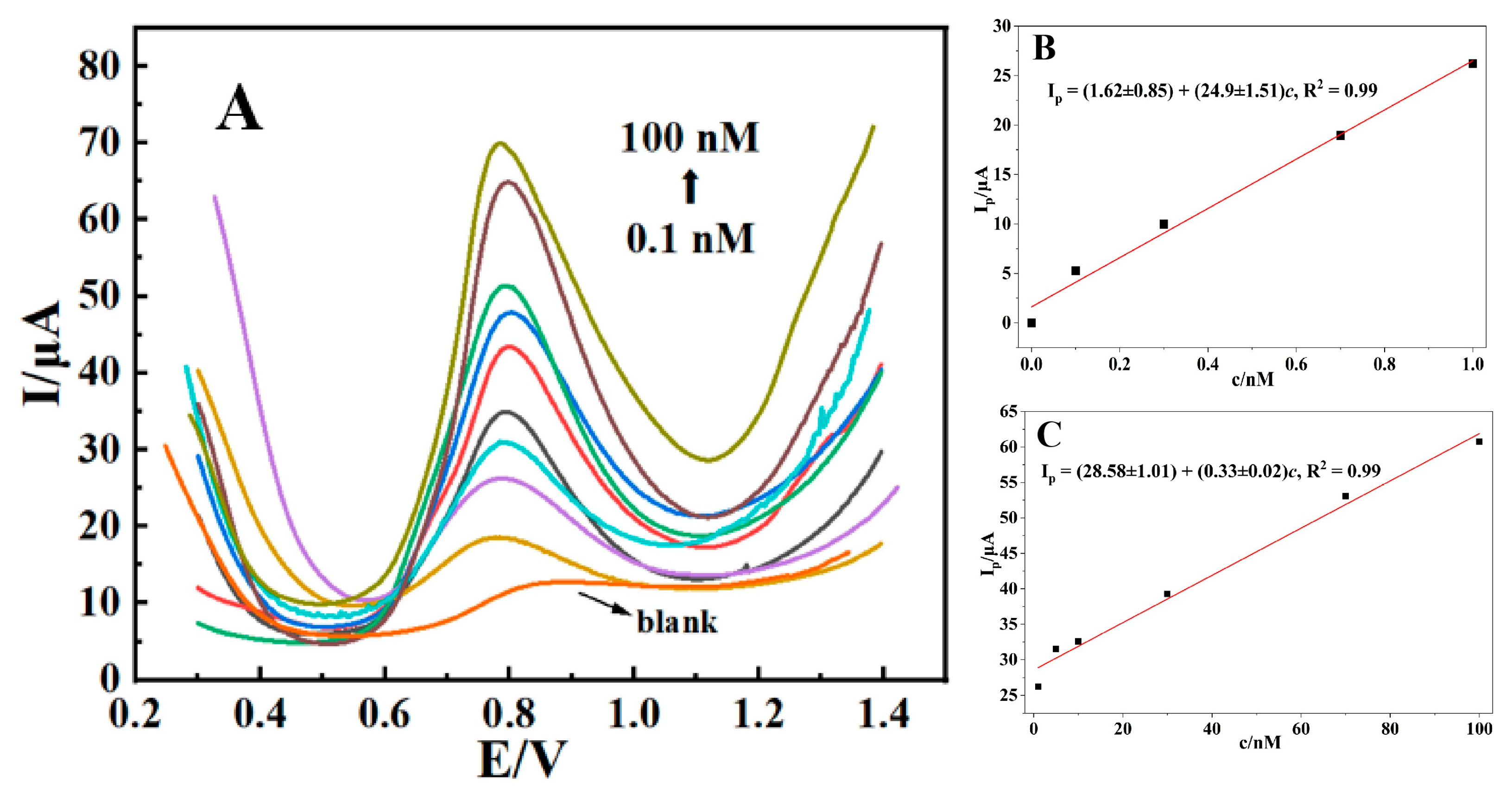
SWV was used to evaluate the linear range and detection limit of the sensor. From Figure 7A, it could be observed that the peak current of AFB1 increased with its concentration. As shown in Figure 7B, from 0.1 nM to 1 nM, the linear equation is Ip = (1.62 ± 0.85) + (24.9 ± 1.51)c, R2 = 0.99, with a detection limit of 20 pM (S/N = 3) and a sensitivity of (24.9 ± 1.51) μA/nM. As shown in Figure 7C, from 1 nM to 100 nM, the linear equation is Ip = (28.58 ± 1.01) + (0.33 ± 0.02)c, R2 = 0.99, with a sensitivity of (0.33 ± 0.02) μA/nM.
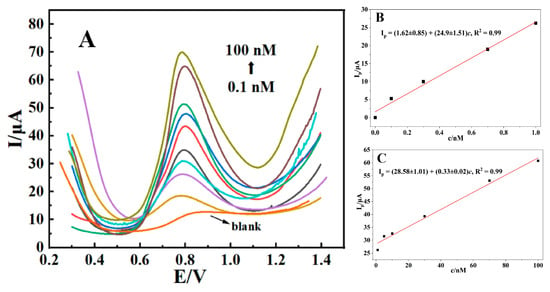
Figure 7.
Square wave voltammograms of CTAB–Nafion/NGCPE in 0.1 M PBS (pH = 5.0) containing different concentrations of AFB1 (A). Linear relationship between the peak current (Ip) and the concentration (c) of AFB1 in the range of 0.1 nM~1 nM (B) and 1 nM~100 nM (C).
3.7.2. Reproducibility, Stability and Selectivity
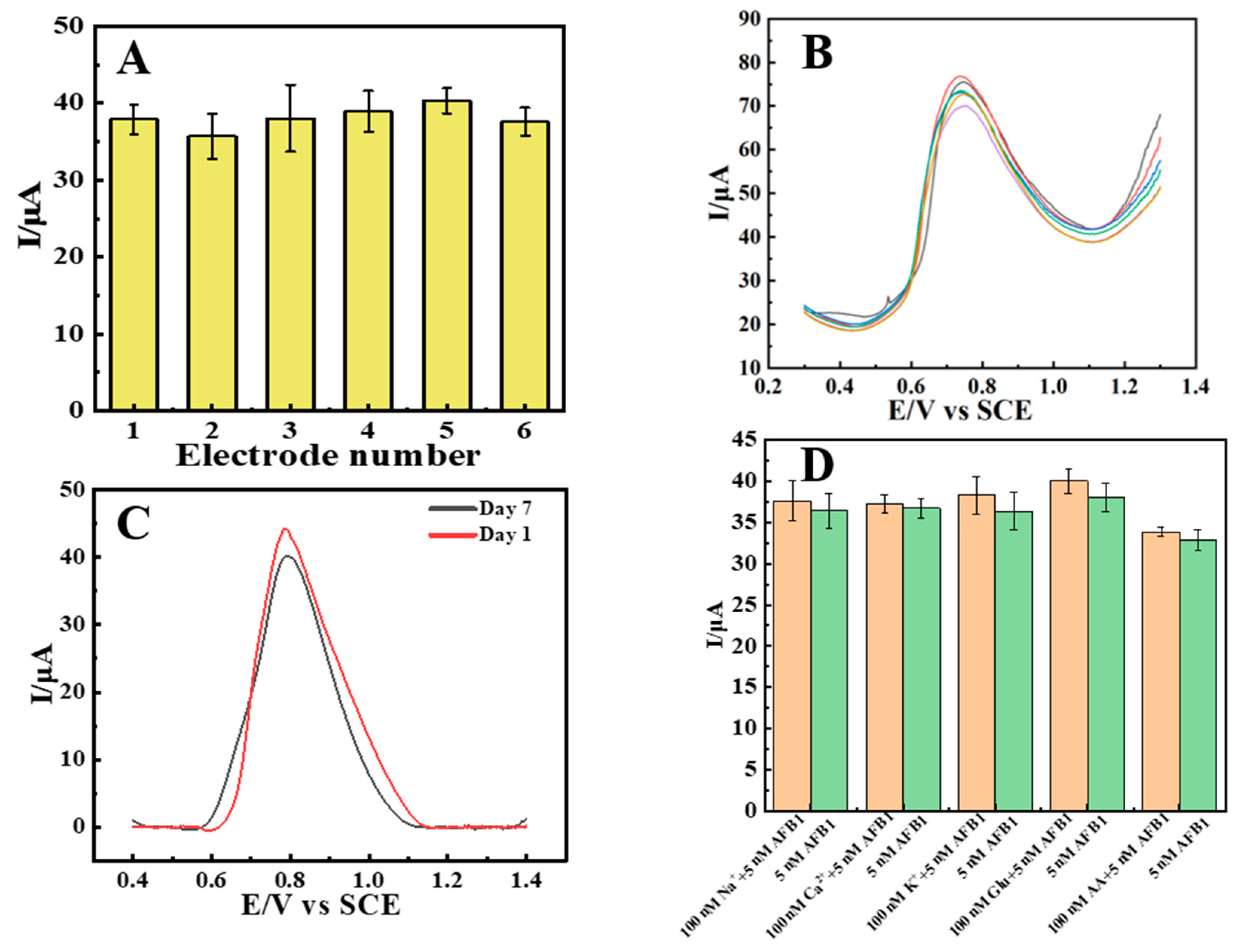
SWV was applied to evaluate the reproducibility, stability and selectivity of the sensor. By using six independently prepared CTAB–Nafion/NGCPEs to carry out the parallel determination of 5 μM AFB1 (Figure 8A), the relative standard deviation (RSD) was calculated to be 6.7%, while the RSD was calculated to be 12.3% for six replicate measurements using a single CTAB–Nafion/NGCPE (Figure 8B). These results are a symbol of the good reproducibility of the sensor. Furthermore, the electrochemical signal retained 91.6% of its initial response after the sensor was stored in air for seven days, demonstrating its good stability (Figure 8C). The selectivity of the sensor was assessed in the presence of potential interferents, including metal ions and glucose. As shown in Figure 8D, the peak current response to 5 μM AFB1 showed no significant change upon the introduction of these interfering substances.
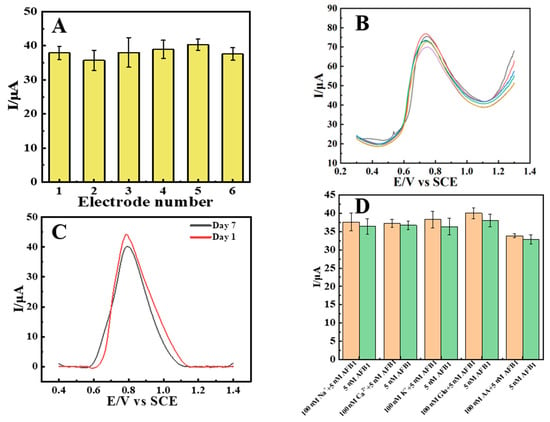
Figure 8.
Electrochemical response of 5 nM AFB1 at six different CTAB–Nafion/NGCPEs (A). Electrochemical response of 10 nM AFB1 at one CTAB–Nafion/NGCPE for six parallel measurements (B). Detecting 5 nM AFB1 with the same CTAB–Nafion/NGCPE initially and one week later (C). Evaluation of the selectivity of the sensor. The orange and green bars show the electrochemical response of 5 nM AFB1 with and without different interferents, respectively (D).
3.8. Real Sample Analysis
To evaluate the sensor’s practicality, spiked recovery tests were conducted to determine the AFB1 concentrations in skim milk and soy sauce samples purchased from a local supermarket. One milliliter of each sample was diluted fivefold with 0.1 M PBS, followed by sequential addition of AFB1 at three concentrations (5 nM, 10 nM, and 20 nM). Detection was then performed using SWV technology. The results are shown in Table 1. The average recovery rate for skim milk samples was 96.3%, while that for soy sauce samples reached 101.1%. These results fully demonstrate the good accuracy of the sensor.

Table 1.
Detection results of AFB1 in real samples.
4. Conclusions
Nano-graphite was used directly without any pretreatment to fabricate the bare NGCPE, and the CTAB–Nafion composite was modified on its surface through simple and controllable cyclic voltammetry. The CTAB–Nafion/NGCPE exhibited good catalytic effects on the electrochemical reaction of AFB1 and can be used as a sensor for AFB1. This study encompasses characterization of the modified NGCPE surface, investigation of the electrochemical reaction mechanism, and comprehensive performance evaluation of the sensor. Under the optimal conditions, the electrochemical response of AFB1 on the sensor increases linearly when its concentration changes from 0.1 nM to 100 nM, with a detection limit of 20 pM and a sensitivity of (24.9 ± 1.51) μA/nM. Comparing with other reported sensors [40,41,42], the developed sensor in this work offers advantages including simple operation, low cost, and good reproducibility, selectivity, and stability. It is expected to provide important references for the development of electrochemical sensors for AFB1 detection.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/chemosensors14040077/s1, Figure S1: Effects of the CTAB–Nafion scanning cycles (A), accumulation time (B) and accumulation potential (C) and pH value of electrolyte (D); Figure S2: CV plots of Ip versus v1/2 for bare NGCPE (A), CTAB–Nafion/NGCPE (C) in 1 M KCl solution containing 5 mM [Fe(CN)6]3−/4− (0.05, 0.075, 0.1, 0.125, 0.15, 0.175, 0.2 V/s), and linear plots of Ip versus v1/2 on bare NGCPE (B) and CTAB–Nafion/NGCPE (D). Table S1. Performance comparison between CTAB–Nafion/NGCPE and other reported sensors. Figure S3. SWV diagrams detected in actual milk (A) and soy sauce (B) samples. Figure S4. CV diagram of CTAB–Nafion/NGCPE in 0.1 M PBS solution containing 5 μM AFB1. Table S2. t-test of the sample analysis results.
Author Contributions
Conceptualization, methodology, writing—review and editing, J.M.; investigation and writing—original draft preparation, H.L.; resources and visualization, J.M. and D.Z.; software, H.L. and S.H.; validation and formal analysis, X.H. and T.X.; supervision and project administration, S.H. and D.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (No. 52472291), the Open Project Funds of the Key Laboratory of Condiment Supervision Technology, State Administration for Market Regulation (No. KLCST-2024-02), the Fundamental Research Funds for the Central Universities, South-Central Minzu University (No. CZZ24004), and the Technical Service Project Funds from Wuhan Jinzhixiu Optoelectronic Technology Co., Ltd. (No. HZY25161).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data that support the findings of this study are included within the article and any supplementary files.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Dinçkaya, E.; Kınık, Ö.; Sezgintürk, M.K.; Altuğ, Ç.; Akkoca, A. Immobilization of anti-aflatoxin B1 antibody by UV polymerization of aniline and aflatoxin B1 detection via electrochemical impedance spectroscopy. Artif. Cells Blood Substit. Biotechnol. 2012, 40, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Yi, S.R.; Wei, X.H.; Li, T.T.; Ai, G.L.; Guo, Y.P.; Guo, L. Rapid detection of aflatoxins B1, B2, G1, G2, and total aflatoxins in citri reticulatae pericarpium and coicis seed based on nitrogen-doped carbon quantum dots. J. Food Compos. Anal. 2025, 148, 108497. [Google Scholar] [CrossRef]
- Khoshbin, Z.; Moslem, B.; Mohammadi, F.; Moeenfard, M.; Abnous, K.; Taghdisi, S.M. A label-free fluorescence aptasensor for ultrasensitive detection of aflatoxin B1: Reliable for food safety and healthcare. Microchem. J. 2025, 218, 115319. [Google Scholar] [CrossRef]
- Rushing, B.R.; Selim, M.I. Aflatoxin B1: A review on metabolism, toxicity, occurrence in food, occupational exposure, and detoxification methods. Food Chem. Toxicol. 2019, 124, 81–100. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, N.; Gao, H.L.; Guo, Z.X.; Lu, A.X.; Guo, X.J.; Lu, J.H.; Luan, Y.X. Determination of Aflatoxin B1 in Lotus Seed by High Performance Liquid Chromatography with Aptamer Affinity Column for Purification and Enrichment. Chin. J. Anal. Chem. 2020, 48, 662–669. [Google Scholar] [CrossRef]
- Campos, W.E.O.; Rosas, L.B.; Neto, A.P.; Mello, R.A.; Vasconcelos, A.A. Extended validation of a senstive and robust method for simultaneous quantification of aflatoxins B1, B2, G1 and G2 in Brazil nuts by HPLC-FLD. J. Food Compos. Anal. 2017, 60, 90–96. [Google Scholar] [CrossRef]
- Sun, L.; Wu, L.; Zhao, Q. Aptamer based surface plasmon resonance sensor for aflatoxin B1. Microchim. Acta 2017, 184, 2605–2610. [Google Scholar] [CrossRef]
- García-Fonseca, S.; Ballesteros-Gómez, A.; Rubio, S. Restricted access supramolecular solvents for sample treatment in enzyme-linked immuno-sorbent assay of mycotoxins in food. Anal. Chim. Acta 2016, 935, 129–135. [Google Scholar] [CrossRef]
- Cao, J.L.; Wang, T.; Wu, K.; Zhou, F.J.; Feng, Y.Z.; Li, J.G.; Deng, A.P. A Highly Sensitive and Group-Specific Enzyme-Linked Immunosorbent Assay (ELISA) for the Detection of AFB1 in Agriculture and Aquiculture Products. Molecules 2024, 29, 2280. [Google Scholar] [CrossRef]
- Yin, L.M.; Cai, J.R.; Ma, L.X.; You, T.Y.; Arslan, M.; Jayan, H.; Zou, X.B.; Gong, Y.Y. Dual function of magnetic nanocomposites-based SERS lateral flow strip for simultaneous detection of aflatoxin B1 and zearalenone. Food Chem. 2024, 446, 138817. [Google Scholar] [CrossRef]
- Hu, R.J.; Zhang, J.D.; Zhang, J.W.; Rong, J.F.; Fu, F.F.; Lin, Z.Y.; Dong, Y.Q. Direct and sensitive SERS detection of aflatoxin B1 based on aggregated silver nanocages. Anal. Chim. Acta 2026, 1383, 344893. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Li, F.; Guang, P.W.; Yang, X.H.; Pan, H.Y.; Huang, F.R. Detection of Aflatoxin B1 in Peanut Oil Using Attenuated Total Reflection Fourier Transform Infrared Spectroscopy Combined with Partial Least Squares Discriminant Analysis and Support Vector Machine Models. J. Food Prot. 2021, 84, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Deng, J.H.; Bai, X.; Monteiro, D.D.N.; Jiang, H. Quantitative analysis of aflatoxin B1 of peanut by optimized support vector machine models based on near-infrared spectral features. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 303, 123208. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.L.; Jia, Q.; Liu, C.Y.; Wang, W.; Duan, L.F.; Yang, G.H.; Han, C.Q.; Li, H.T. Thin layer chromatography combined with surface-enhanced raman spectroscopy for rapid sensing aflatoxins. J. Chromatogr. A 2018, 1579, 115–120. [Google Scholar] [CrossRef]
- Jangampalli Adi, P.; Matcha, B. Analysis of aflatoxin B1 in contaminated feed, media, and serum samples of Cyprinus carpio L. by high-performance liquid chromatography. Food Qual. Saf. 2018, 2, 199–204. [Google Scholar] [CrossRef]
- Wu, M.Q.; Ma, Y.; Huang, Y.R.; Zhang, X.H.; Dong, J.; Sun, D.P. An ultrasensitive electrochemical aptasensor based on zeolitic imidazolate framework-67 loading gold nanoparticles and horseradish peroxidase for detection of aflatoxin B1. Food Chem. 2024, 456, 140039. [Google Scholar] [CrossRef]
- Chokkareddy, R.; Redhi, G.G. Fe3O4 Nanorods-RGO-ionic Liquid Nanocomposite Based Electrochemical Sensor for Aflatoxin B1 in Ground Paprika. Electroanalysis 2022, 34, 501–511. [Google Scholar] [CrossRef]
- Khattiya, A.; Karaket, R.; Mathaweesansurn, A.; Detsri, E. Ultrasensitive electrochemical aptasensors based on trimetallic AuPt-Ru nanoparticles decorated RGO with disposable and low-cost goldleaf electrode for aflatoxin B1 quantification in agricultural products. Microchem. J. 2025, 208, 112631. [Google Scholar] [CrossRef]
- Liu, X.N.; Liu, S.J.; Kong, L.; Zou, M.X.; Na, G.Q.; Ouyang, F.; Yang, Q.Y.; Wang, J.L. Advancements in nanomaterial-based sensors for dual and multi-mode detection of aflatoxin B1. Trends Food Sci. Technol. 2025, 163, 105120. [Google Scholar] [CrossRef]
- El Abbadi, S.; Elgamouz, A.; Kawde, A.N.; Douma, M.; El Moustansiri, H.; Tijani, N. Fe3O4 nanoparticle-modified exfoliated carbon paste electrode for enhanced electrochemical detection of dopamine. Chem. Inorg. Mater. 2025, 7, 100125. [Google Scholar] [CrossRef]
- Mohammadzadeh Jahani, P.; Tajik, S.; Beitollahi, H.; Nejad, F.G.; Dourandish, Z. Determination of Methotrexate Using an Electrochemical Sensor Based on Carbon Paste Electrode Modified with NiO Nanosheets and Ionic Liquid. Chemosensors 2024, 12, 266. [Google Scholar] [CrossRef]
- Zojaji, Z.; Alizadeh, T. Synthesis of Nano-graphite via physical methods for simultaneous and sensitive determination of heavy metal ions. J. Electrochem. Soc. 2026, 173, 047502. [Google Scholar] [CrossRef]
- Abbas, M.M.; Ali, Y.Y.; Ayad, M.F.; Fares, N.V. Surfactant-assisted nanocomposite pencil graphite electrode: A green, practical approach for electrochemical monitoring of Riluzole in different matrices. Microchem. J. 2025, 217, 114983. [Google Scholar] [CrossRef]
- Dongmo, L.M.; Pecheu, C.N.; Jiokeng, S.L.Z.; Dzuibo, S.R.N.; Djuffo, L.A.Y.; Tajeu, K.Y.; Ngantchouko, C.B.N.; François, K.; Tonle, I.K. Amino-montmorillonite crystalline clay as electrode modifier for electrochemical detection of ciprofloxacin in presence of cetyltrimethylammonium bromide. ChemElectroChem 2024, 11, e202400123. [Google Scholar] [CrossRef]
- Zhang, L.N.; Ma, H.N.; Sun, H.L.; Zhang, X.Y.; Sun, R.M.; Zheng, H.R.; Wang, H.; Wang, Y.R.; Guo, J.Y.; Liu, Y.Q. A copper-organic framework/graphene modified electrode for sensitive electrochemical detection of bisphenol A. J. Appl. Electrochem. 2025, 55, 177–188. [Google Scholar] [CrossRef]
- Sahana, H.R.; Nayaka, Y.A.; Pradeepa, E. Selective detection of carbendazim in the presence of 2,4-dichlorophe-noxyacetic acid via electrochemical route using CTAB@MET modified pencil graphite electrode as a sensor. J. Mol. Struct. 2025, 1330, 141476. [Google Scholar] [CrossRef]
- Chen, T.F.; Huang, Y.Z.; Liu, X.J.; Zhu, C.N.; Liu, C.; Zheng, D.Y. Electrochemical detection of catechol with a disposable carbon nanotube paste electrode modified with CTAB. Phys. Scr. 2025, 100, 015024. [Google Scholar] [CrossRef]
- Malode, S.J.; Prabhu, K.K.; Shetti, N.P.; Reddy, K.R. Highly sensitive electrochemical assay for selective detection of Aminotriazole based on TiO2/poly (CTAB) modified sensor. Environ. Technol. Innov. 2021, 21, 101222. [Google Scholar] [CrossRef]
- Khederlou, K.; Lefevre, N.; Ramsey, S.A.; Fu, E. Nafion™ electrode coating for enhanced electrochemical detection of carbamazepine from whole human saliva. J. Appl. Electrochem. 2025, 55, 3021–3032. [Google Scholar] [CrossRef]
- Er, E.; Çelikkan, H.; Erk, N. A novel electrochemical nano-platform based on graphene/platinum nanoparticles/nafion composites for the electrochemical sensing of metoprolol. Sens. Actuators B Chem. 2017, 238, 779–787. [Google Scholar] [CrossRef]
- Ghanbari, M.H.; Norouzi, Z.; Biesalski, M.; Etzold, B.J. Superior performance of N-doped carbon Nanoonions/Nafion based microfluidic electrochemical Cd2+ sensor when compared to Screen-Printed Carbon-Based electrode devices. Microchem. J. 2024, 201, 110506. [Google Scholar] [CrossRef]
- Zheng, D.Y.; Liu, X.J.; Zhu, S.Y.; Cao, H.M.; Chen, Y.G.; Hu, S.S. Sensing nitric oxide with a carbon nanofiber paste electrode modified with a CTAB and nafion composite. Microchim. Acta 2015, 182, 2403–2410. [Google Scholar] [CrossRef]
- Della Pelle, F.; Compagnone, D. Chemical sensing of food phenolics and antioxidant capacity. In Advanced Sensor Technology: Biomedical, Environmental, and Construction; Barhoum, A., Altintas, Z., Eds.; Elsevier: Amsterdam, The Netherlands, 2023. [Google Scholar]
- Chen, T.F.; Zhang, S.S.; Zhu, C.N.; Liu, C.; Liu, X.J.; Hu, S.S.; Zheng, D.Y.; Zhang, J.C. Application of surfactants in the electrochemical sensing and biosensing of biomolecules and drug molecules. Anal. Methods 2024, 16, 3607–3619. [Google Scholar] [CrossRef] [PubMed]
- Maritan, A.; Toigo, F. On skewed arc plots of impedance of electrodes with an irreversible electrode process. Electrochim. Acta 1990, 35, 141–145. [Google Scholar] [CrossRef]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. Interfacial Electrochem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Franco Guzmán, M.; Castañeda Ovando, A.; Mendoza Huizar, L.H.; Flores, B.C.; León, S.H.; Perales, L.E.R.; Romero, G.A.Á. Voltammetric determination of aflatoxin B1 using a bismuth layer-gold nanoparticles modified glassy carbon electrode. J. Appl. Electrochem. 2025, 55, 487–497. [Google Scholar] [CrossRef]
- Anson, F.C. Application of potentiostatic current integration to the study of the adsorption of cobalt(iii)-(ethylenedinitrilo(tetraacetate) on mercury electrodes. Anal. Chem. 1964, 36, 932–934. [Google Scholar] [CrossRef]
- Ngamchuea, K.; Eloul, S.; Tschulik, K.; Compton, R.G. Planar diffusion to macro disc electrodes—what electrode size is required for the Cottrell and Randles-Sevcik equations to apply quantitatively? J. Solid State Electrochem. 2014, 18, 3251–3257. [Google Scholar] [CrossRef]
- Wang, C.; Li, Y.P.; Zhao, Q. A signal-on electrochemical aptasensor for rapid detection of aflatoxin B1 based on competition with complementary DNA. Biosens. Bioelectron. 2019, 144, 111641. [Google Scholar] [CrossRef]
- Wood, M.; Mugo, S.M. A MIP-enabled stainless-steel hypodermic needle sensor for electrochemical detection of aflatoxin B1. Anal. Methods 2022, 14, 2063–2071. [Google Scholar] [CrossRef]
- Gevaerd, A.; Banks, C.E.; Bergamini, M.F.; Marcolino, L.H., Jr. Nanomodified Screen-Printed Electrode for direct determination of Aflatoxin B1 in malted barley samples. Sens. Actuators B Chem. 2020, 307, 127547. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.