Abstract
The growing demand for sustainable, biocompatible, and multifunctional sensing materials has intensified interest in melanin and its derivatives, including melanin-inspired polymers and composites. Melanin is a naturally occurring biopolymer whose intricate structure and diverse chemical composition give rise to a remarkable combination of optical, electrical, and chemical properties. Key physicochemical characteristics, such as broadband optical absorption, hydration-dependent conductivity, redox activity, and metal ion coordination, are closely linked to melanin’s signal transduction capabilities and underpin its relevance in sensing applications. Recent advances in melanin-based sensing technologies encompass pH, humidity, chemical, biological, and optical platforms, with particular emphasis on hybrid systems incorporating graphene, silicon, or nanomaterials, and printable or wearable device architectures. These developments have enabled enhanced performance and broadened potential application fields. However, persistent challenges, including intrinsic heterogeneity, limited selectivity, relatively low electrical conductivity, and poor long-term operational stability, still limit practical implementation. Emerging molecular engineering and advanced fabrication strategies are being developed to address these limitations. Together, these findings position melanin as a versatile, eco-compatible, and functionally rich material, with a significant potential to underpin the next generation of sustainable sensing technologies.
1. Introduction
Sensors are devices capable of transducing a physical, chemical, or biological stimulus into a measurable signal and serve as fundamental tools across a wide range of fields, including environmental monitoring, health diagnostics, food safety, and wearable electronics [1,2]. As the demand grows for sensors that are more sensitive, selective, biocompatible, flexible, sustainable, and capable of multifunctional detection, the design of materials with novel and advantageous combinations of properties becomes crucial [3,4]. A particularly promising class of bio-inspired materials comprises melanin and its derivatives, including melanin-inspired polymers or composites. Melanin-based sensors have attracted increasing attention in recent years [5,6,7,8] because of melanin’s unique optical, chemical, electronic, and structural properties, as well as its biocompatibility and potential for sustainable sourcing.
This growing interest is also reflected in the temporal evolution of the scientific literature, although a robust statistical or trend-based analysis remains challenging due to the relatively recent emergence and interdisciplinary scope of research specifically devoted to melanin-based sensors. Before the late 2010s, studies explicitly integrating melanin into sensor technologies were limited and largely centered on pigment characterization, melanometric measurements, or analytical quantification rather than on fully developed functional sensing platforms [9,10,11,12,13,14]. In the period that followed, research activity remained relatively modest and niche, with publications primarily reporting early proof-of-concept electrochemical or optical sensing approaches that leveraged the adhesive, redox-active, and metal-chelating properties of polydopamine [15,16,17,18]. From the early 2020s onward, a noticeable expansion of the field can be observed, marked by the emergence of dedicated review articles and a diversification of application areas, including environmental monitoring, food safety, biosensing, and wearable technologies. In more recent years, publication activity has increased substantially and appears to be sustained, driven by advances in melanin-based electrochemical interfaces, optical probes, molecularly imprinted sensors, and hybrid material systems [19,20,21,22].
Overall, melanin provides a multifunctional platform for developing a new generation of sustainable and high-performance sensors [19,23]. Ongoing research on its synthesis, characterization, and integration into diverse device architectures underscores its substantial potential to transform applications ranging from biomedical diagnostics and wearable electronics to environmental monitoring. The originality and novelty of this work arise from the systematized, updated, and practice-oriented presentation of a large number of selected works, with emphasis on recent findings and pivotal studies, regarding the potential applications of melanin-based materials in sensing.
2. Melanins: Types, Structure, and Occurrence
Melanin is a biopolymer present across all biological kingdoms, whose molecular structure remains only partially elucidated [24,25]. It serves different functions, including radiation protection, pigmentary and structural coloration, thermoregulation, and radical scavenging [23,24,26,27].
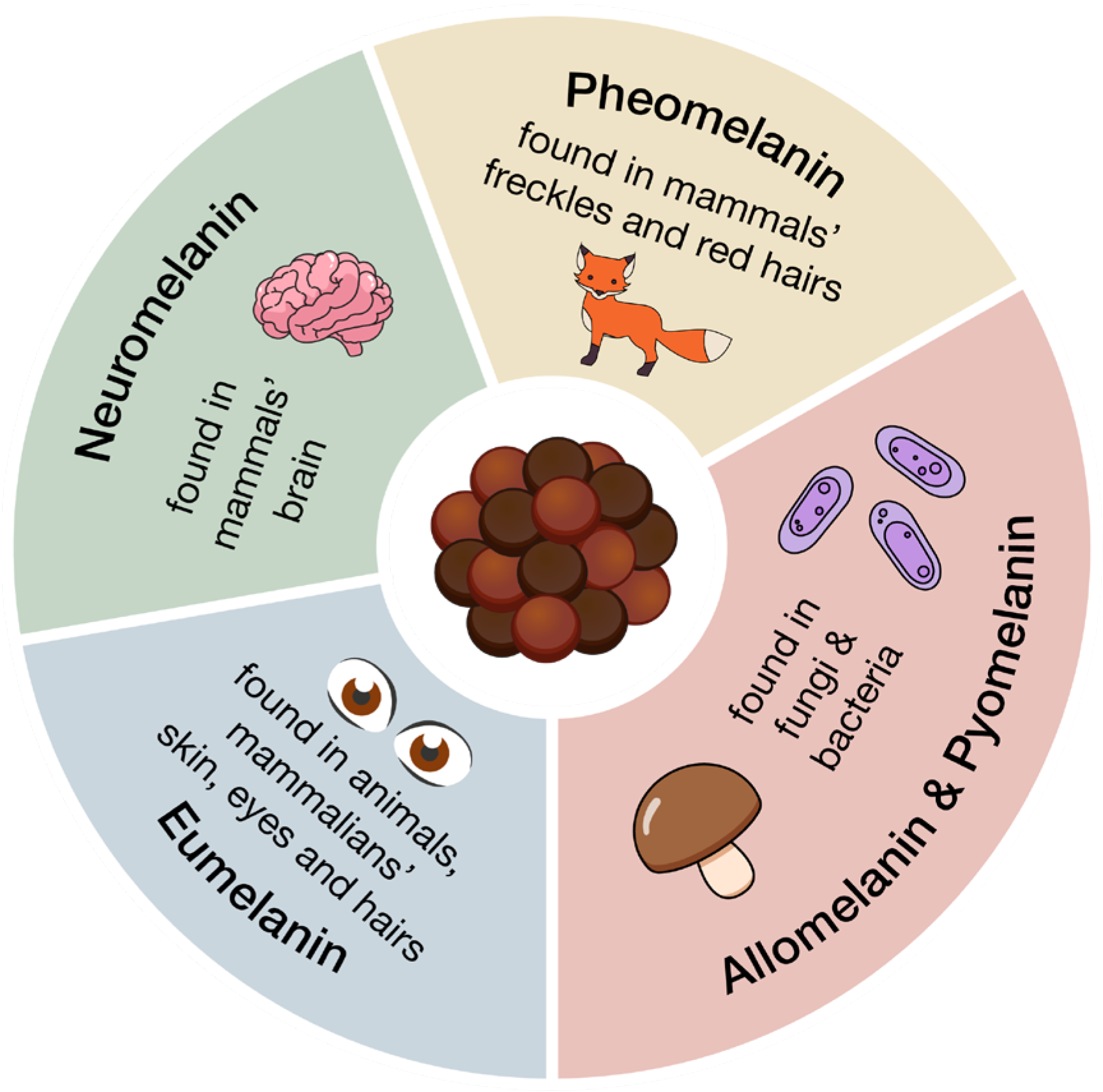
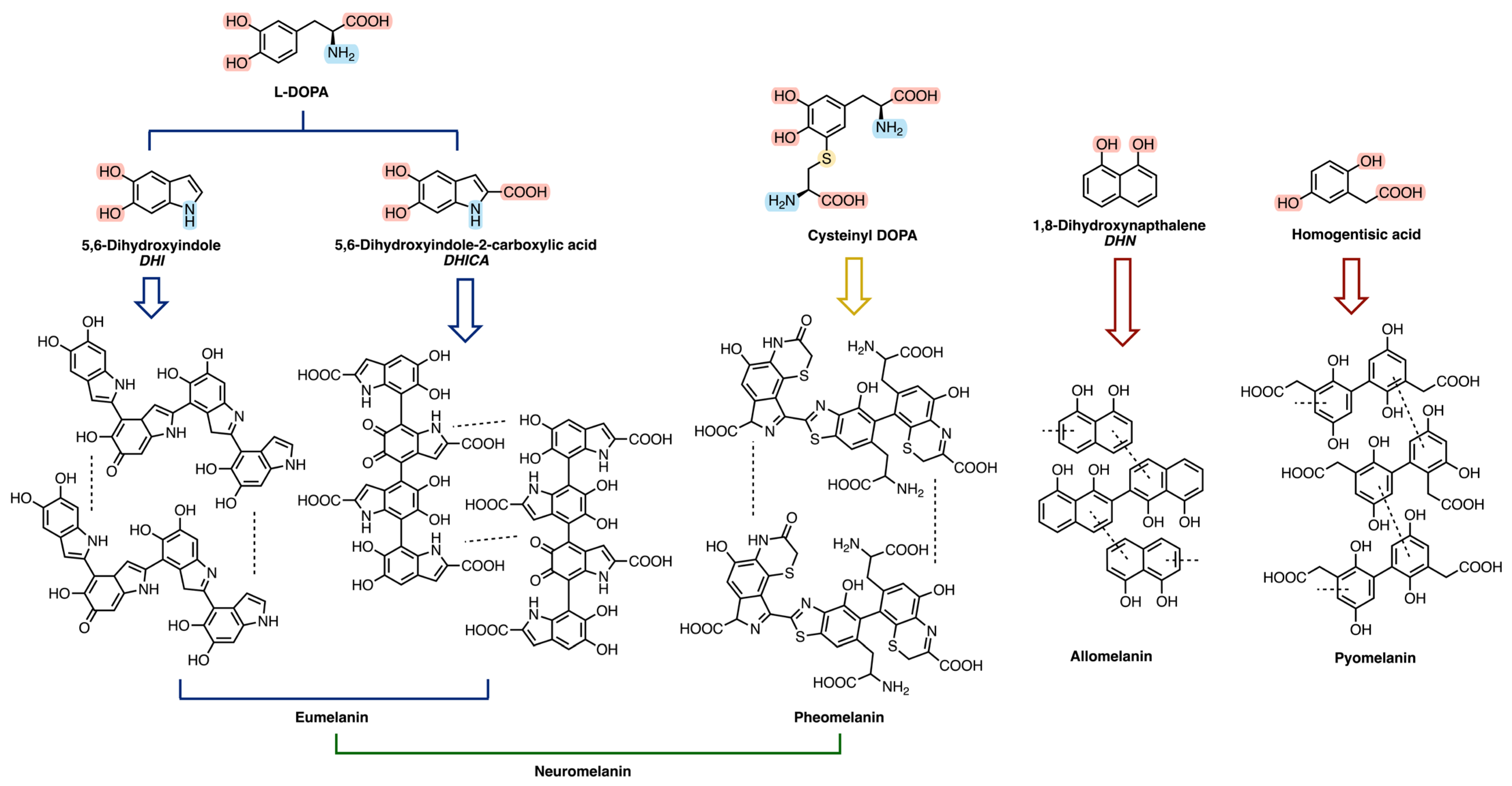
Melanin is a generic term, indicating a family of biologically significant pigments that can be classified, based on their chemical precursor and structure, into five main categories (summarized in Figure 1 and Figure 2 and Table 1): eumelanin, pheomelanin, neuromelanin, allomelanin, and pyomelanin [24,27]. The chemical composition of these biopolymers includes the presence of diverse functional groups, which enable melanins to interact with various organic compounds, both through hydrogen bonding, and π-π stacking, and contribute to their varied coloration and functional properties [23,27,28,29,30].
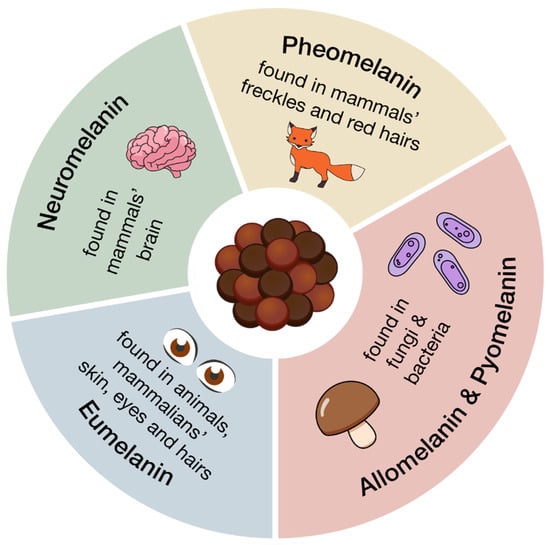
Figure 1.
Principal classes of melanin polymers and their main source.
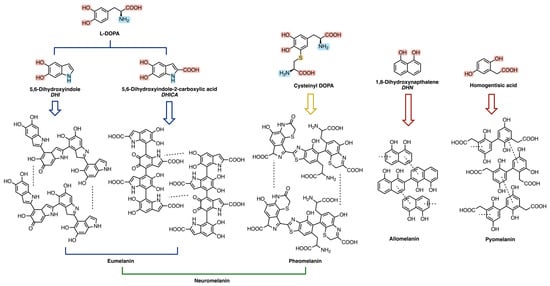
Figure 2.
Monomeric and oligomeric structures of the main classes of melanin polymers.

Table 1.
Main classes of melanin polymers.
Among these functionalities, the most abundant is the hydroxyl group—often occurring in its oxidized (quinonic) form—which characterizes all five classes of melanins, followed in prevalence by the carboxyl group [23,24]. In addition, nitrogen-containing moieties, such as indoles and amines, are typically present, except for allomelanin and pyomelanin [23,24].
Eumelanin, deriving from the oxidative polymerization of L-DOPA (L-3,4-dihydroxyphenylphenylalanine), itself a product of tyrosine metabolic pathway, is brownish-black and it is mostly present in animals, in mammalian skin, eyes, and hair [23,24,31,32]. Pheomelanin, whose key structural unit is cysteinyl-DOPA, is instead characterized by a yellow-reddish-brown color, typical of red hair and freckles [23,24]. It differs from eumelanin due to the presence of sulfur-containing units (i.e., benzothiazines and benzothiazoles) [24,29]; its biosynthesis indeed starts from the polymerization of 5-S-cysteinyl-dopa, itself deriving from dopaquinone in the presence of cysteine [24,28,30]. Neuromelanin contains both pheomelanin and eumelanin, and, as the name suggests, this pigment is found in the brain, but its function is still unclear [23,33,34,35]. Its formation involves the oxidative polymerization of dopamine and cysteine [23,33,34,35].
Allomelanin and pyomelanin are produced by fungi and bacteria. The latter derives from homogentisic acid, while 1,8-dihydroxynaphthalene (DHN) is the main precursor of the former [23,36,37,38,39]. Auricularia auricola is a notable example of a fungus able to synthesize great amounts of melanin, characterized by various biological activities, therefore making it interesting for industrial applications [37].
On a larger scale, melanin monomers assemble themselves into stacked oligomers, mainly through π–π interactions and hydrogen bonding. These subsequently cluster together to form larger particles, usually in the range between 100 and 200 nm, with a spacing between the stacked layers of about 0.34 nm [23]. This hierarchical organization plays a key role in determining melanin’s electronic and optoelectronic behavior [38].
3. Key Physicochemical and Electrical Properties of Melanin Polymers
The ubiquitous distribution of melanin highlights its biological importance [24,30], while its diverse physicochemical properties make it valuable for a wide range of applications, including biomedicine, bioelectronics, cosmeceuticals, and environmental bioremediation [23,28,32].
Its versatility is intrinsically linked to its unique physicochemical features (summarized in Table 2), arising from its complex, highly variable molecular structure, which depends on its origin, type, and specific precursors [23,38].

Table 2.
Summary of physicochemical and electrical properties of melanin polymers.
3.1. Optical Properties
Melanin exhibits a broadband optical absorption extending across the ultraviolet (UV), visible, and near-infrared (NIR) regions [56,57]. This feature is due to its complex and heterogeneous polymeric structure; the polymerization of various indole and indole-quinone moieties, as well as their subsequent supramolecular assembly via hydrogen bonding, π−π interaction, or cation−π interaction, results in complex and diversified spectra, dependent on the structure of the monomeric species involved [38,39,56,57,58].
This property underpins one of melanin’s primary biological roles, photoprotection, by enabling the efficient dissipation of high-energy radiation and preventing subsequent photochemical damage [21,58].
Moreover, it has also motivated a wide array of innovative technological applications. Recent studies demonstrated that melanin–silicon hybrid junctions can serve as effective broadband photodetectors [21], while melanin nanoparticles, integrated with silk protein, could be exploited for the design of photoresponsive, self-healable optoelectronic skins [40], highlighting their potential in bio-integrated applications. Functionalized melanin nanoplates instead demonstrated their efficiency in photoacoustic imaging and photothermal tumor eradication [41]. Recently, several synthetic melanins have also been employed as heterogeneous photocatalysts, mediating the visible-light-driven reductive dehalogenation of a series of α-halogen carbonyl derivatives [42].
3.2. Charge Transport Properties
Melanin is also characterized by unique charge transport properties, exhibiting indeed both electronic and protonic conductivity [43].
Due to this remarkable feature, these biopolymers can be categorized as mixed ionic–electronic conductors (MIECs) and therefore be considered highly valuable materials in sustainable electronics applications, e.g., bioelectronics, neuromorphic computing, soft robotics, or energy storage [43,44,59], in addition to their possible employment as electrochemical sensors.
Recently, conditions determining the prevalence of one conductivity mechanism over the other have been widely investigated [43,44,45,46], and it emerged that the hydration state plays a major role in this sense: when wet or humid, proton conduction is facilitated by the presence of water molecules, whereas in drier conditions, electronic conduction may dominate [43,46].
This high sensitivity to moisture and changes in hydration also makes melanin considerable for humidity sensors and related applications [19].
3.3. Metal Chelation and Redox Activity
Melanins also exhibit significant metal-ion chelating activity due to the presence of the above-mentioned functional groups in their structure [48,60]. This ability to form stable complexes is crucial as transition metal ions, particularly iron and copper, can catalyze the formation of highly reactive free radicals through Fenton-type reactions, thereby exacerbating oxidative stress [61]. By binding them, melanin effectively inactivates their pro-oxidant potential, contributing to its overall antioxidant defense mechanism [60].
Moreover, melanin’s excellent radical scavenging ability—arising from its significant redox activity—is well-documented. Acting as a natural antioxidant, this biopolymer plays a protective role against oxidative stress—protecting cells and tissues from harmful reactive oxygen species (ROS) and free radicals—and environmental damage [62,63,64].
The mechanism of free radical scavenging often involves the donation of hydrogen atoms or electrons from the antioxidant to the free radical, thereby converting the radical into a more stable, non-radical species. Spectroelectrochemical reverse engineering has indeed demonstrated its ability to exchange electrons [64].
Both characteristics have been exploited in different applications. For example, eumelanin electrodes showed their potential in electrochemical energy storage due to their redox behavior [47]. Conversely, melanin’s chelating ability has been exploited in environmental applications, such as the removal of heavy metals from aqueous solutions. Biosynthesized melanin, derived from the marine bacterium Pseudomonas stutzeri, demonstrated efficient adsorption capacities for heavy metals like Mercury, Chromium, Lead, and Copper, showcasing the practical utility of melanin’s chelating properties [48,49].
Melanin redox and metal-ion chelating activity opens the possibility for its application in chemical transduction (e.g., a sensor detecting reactive oxygen species or redox-active analytes).
3.4. Biocompatibility and Biodegradability
The intrinsic biocompatibility (i.e., the ability of a material to interact with biological systems without eliciting adverse reactions) of melanin is an attribute consistently highlighted in numerous studies [50,51,55,65,66], as its biodegradability, defined as the ability of the material to be broken down by natural biological processes, such as the action of microorganisms, over time [23,65,66].
Several oxidative pathways have been reported for the degradation of melanin polymers in physiological environments, among which are enzymatic and microbial mechanisms [24,32,60]. Reactive oxygen species can indeed cleave indole–quinone linkages in eumelanin, leading to their depolymerization into smaller, water-soluble fragments [67,68]. Enzymatic degradation has been described both in vitro and in vivo [69], particularly by peroxidases, including lignin peroxidase [70], as well as laccase-like oxidases [71], which catalyze melanin oxidation in the presence of hydrogen peroxide or molecular oxygen. Microbial degradation has also been reported, especially in fungi and soil bacteria that secrete oxidative enzymes such as laccases and manganese peroxidases, resulting in partial mineralization over periods ranging from days to weeks [72].
Both these features make melanin—alongside other natural biopolymers, as cellulose, alginate, chitosan or lignin [73,74]—well-suited for bio-based wearable electronics: studies have indeed explored the potential use of biodegradable conductive and semiconductive melanin films or scaffolds for nerve tissue engineering [50,51]. Moreover, melanin-based materials remarked their efficiency in specific drug delivery [52,53,54,55] and diagnostic applications involving direct contact with biological tissues and fluids, as well as for medical implants or transient devices where the material’s eventual breakdown and clearance from the body are desirable, reducing long-term safety concerns and the need for removal surgeries, demonstrating that melanin’s biocompatibility and biodegradability represent a crucial advantage related to these materials [23,50,55,66].
4. Examples of Melanin-Based Sensors
Several classes of melanin-based sensors are currently under development and active investigation, reflecting the growing recognition of melanins as versatile functional materials for sensing applications. These sensing systems, summarized in Table 3, exploit the distinctive physicochemical properties of melanins, and explore potential applications across different sensing platforms. In the following sections, we analyze in detail the main categories of melanin-based sensors, with particular emphasis on pH, humidity, and optical sensors, highlighting their operating principles, material design strategies, and representative performance characteristics.

Table 3.
Main classes of melanin-based sensors.
4.1. pH Sensors
As pH sensors are widely employed in fields such as biochemistry and medical diagnostics, their continued development is crucial [15,87,88,89]. In recent years, growing emphasis has been placed on wearable sensors, particularly those integrating pH sensors with biomarker detectors, developed for continuous and non-invasive health monitoring [87,88,89]. In this sense, melanin has emerged as a highly attractive material, as demonstrated by multiple examples, summarized in Table 4, due to its above-mentioned features, such as the ability to modulate its electrical properties in response to pH variations, since some of its functional groups can be protonated or deprotonated depending on pH value. This feature is suitable for integration into bioelectronic devices, including Extended-Gate Field-Effect Transistors (EGFETs) for pH sensing applications [15,90,91].

Table 4.
Melanin-based pH sensors.
In the last decade, pH sensors based on EGFET architectures employing melanin thin films as the active sensing layer have indeed been developed [15]. The melanin films, synthesized from different precursors and deposited on conductive substrates such as indium tin oxide and gold, have been extensively characterized using atomic force microscopy (AFM) and electrochemical impedance spectroscopy. Device testing across a broad pH range (2–12) revealed sensitivities between 31.3 and 48.9 mV/pH, depending on both the melanin precursor and the substrate. The sensing mechanism has been attributed to specific binding sites in melanin, particularly hydroxyl groups and quinone imine moieties. In addition, sensor performance was strongly influenced by deposition conditions, substrate choice, and surface roughness, with increased roughness providing a larger effective sensing area and enhanced sensitivity. The integration of melanin into EGFETs represents a promising avenue for developing sensitive, biocompatible, and versatile pH sensors, particularly for applications requiring non-invasive or continuous monitoring in biological systems. Continued research into melanin’s properties and its integration with advanced device architectures will likely lead to further improvements in sensor performance and expand its application scope.
Beyond EGFET-based configurations, the integration of synthetic eumelanin into robust solid-state platforms using standard microfabrication techniques represents a significant advance toward scalable and miniaturized wearable devices. In particular, the successful use of conventional photolithography to pattern sulfonated-eumelanin thin films demonstrates that the material’s fundamental indole-based chemical structure, film topography, and charge-transport behavior remain unaffected by standard processing steps, as confirmed by a comprehensive characterization, by AFM, Raman spectroscopy, and X-ray photoelectron spectroscopy (XPS), combined with direct and alternating current electrical measurements. Interdigitated electrode architectures fabricated with these films enabled the development of compact multisensory platforms capable of both pH and humidity sensing. Within physiologically relevant conditions (pH 5–8), sulfonated-eumelanin-based pH sensors exhibited sensitivities of approximately 50.1 mV·pH−1 and demonstrated stable operation over time, highlighting their suitability for applications such as wound-healing monitoring [75].
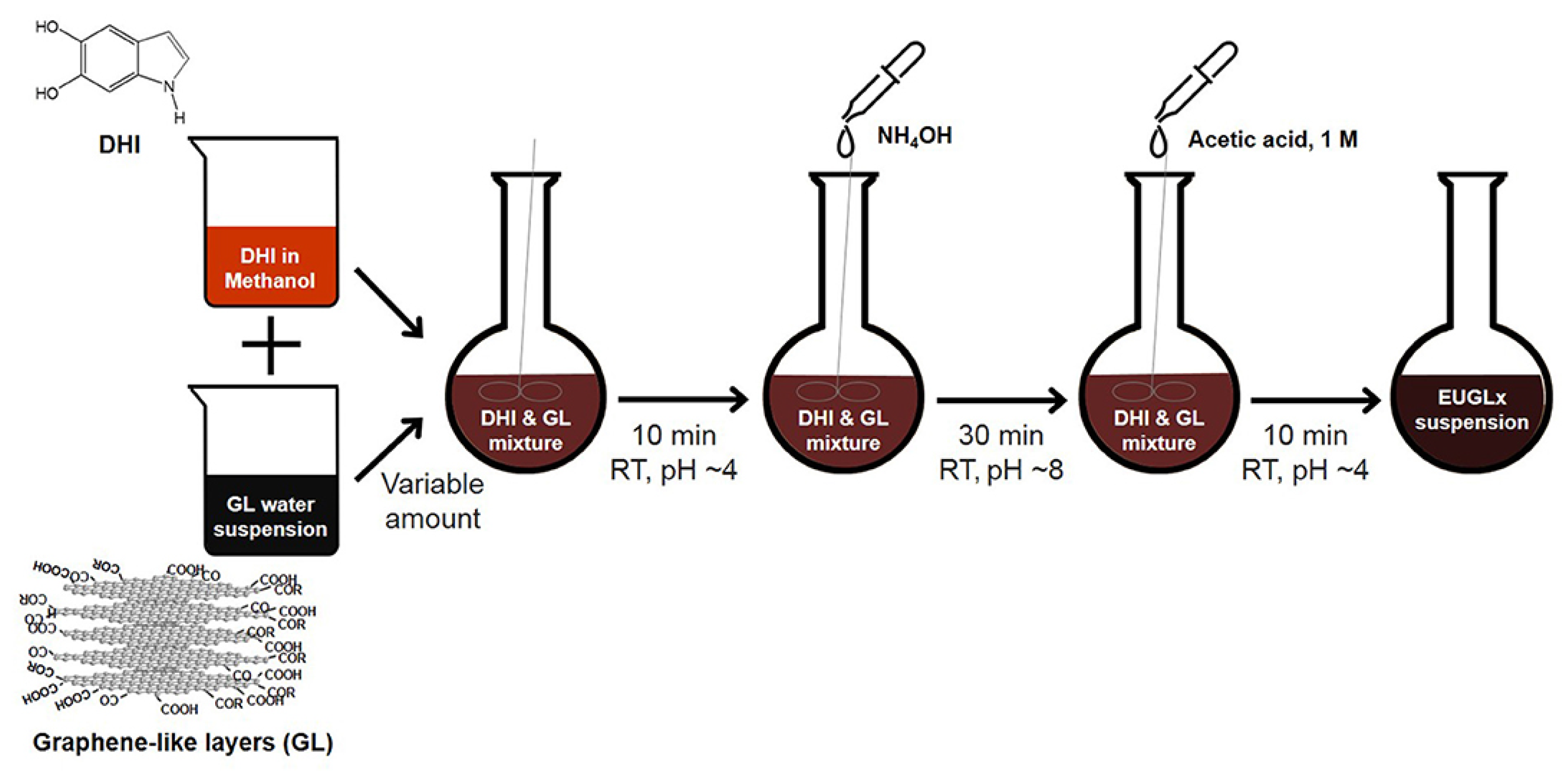
Moreover, a particularly promising direction involves the integration of melanin with graphene-based materials, increasingly applied due to their adjustable properties and ease of functionalization, in flexible and wearable biomedical sensors to monitor physiological signals and biochemical parameters [92,93,94]. These derivatives, including graphene oxide and reduced graphene oxide, offer outstanding electrical conductivity, high carrier mobility, large surface area, and easy functionalization, making them ideal for flexible and wearable biomedical sensors [76,77,78]. Hybrid graphene–melanin systems have been shown to enhance charge transport and sensing performance through strong synergistic interactions. In particular, polymerization of eumelanin precursors in the presence of graphene-like layers (Figure 3) leads to tighter molecular packing and improved electrical conductivity, providing an effective strategy for tuning bioelectronic properties [76].
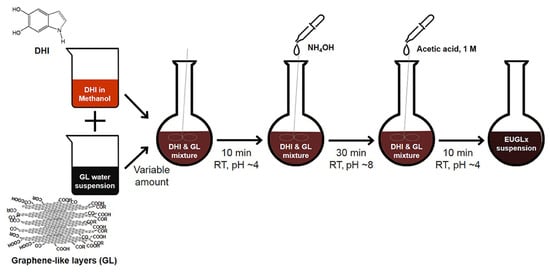
Figure 3.
Schematic diagram of the synthetic procedure adopted for the preparation of the hybrid graphene–melanin sensors. Reproduced with permission [76]. Copyright 2019, Frontiers.
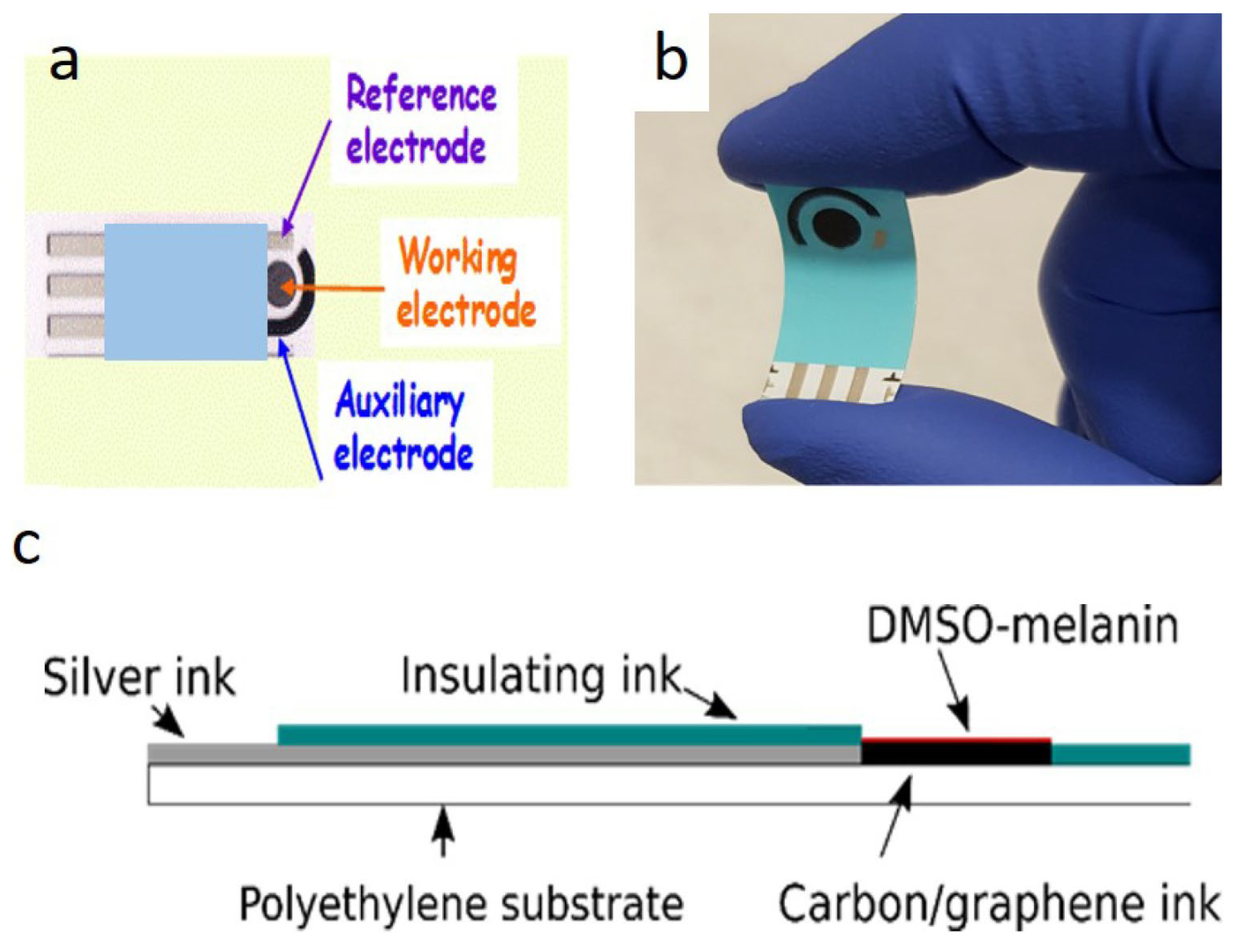
In addition, printable and flexible graphene–melanin pH sensors have been successfully fabricated using scalable techniques such as screen printing. In these devices, a conductive carbon or graphene layer is coated with a pH-responsive DMSO–melanin film (Figure 4) that serves as the active sensing element. Variations in hydrogen ion concentration modulate the electrochemical properties of the melanin layer, which are transduced by the graphene electrodes. These sensors have demonstrated high sensitivity (up to 62 ± 7 mV/pH), excellent reproducibility, and reliable operation within physiologically relevant pH ranges, making them well suited for point-of-care diagnostics, biotechnology, and continuous health monitoring [77,78].
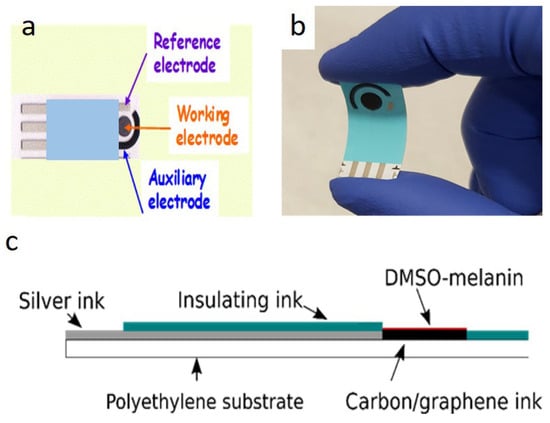
Figure 4.
Printable and flexible graphene-melanin pH sensor. Top view (a), photograph demonstrating the flexibility of the sensor (b), cross section (c). Reproduced with permission [77]. Copyright 2020, IOP Science.
Graphene–melanin pH sensors have also been validated in complex biological environments, including bacterial culture media, where they accurately monitored pH variations during microbial growth. Their robustness and repeatability highlight their potential for applications such as bioreactor monitoring. Moreover, the ability of these sensors to continuously and in real time track pH fluctuations makes them strong candidates for wound-care applications, where pH is a key indicator of infection and healing status [76,77,78].
Furthermore, additive manufacturing techniques such as screen printing, inkjet printing, and 3D printing could enable the precise placement of materials to form components on various flexible substrates, such as plastics, paper, cloth, and even skin, so to broaden the application landscape of melanin-based pH sensing technologies. [95,96,97].
4.2. Humidity Sensors
Melanins hold potential in humidity sensing applications (see Table 5 for examples) due to their hygroscopic nature and their ability to respond to changes in moisture content, which lead to measurable modification in their electrical or optical characteristics [19,29,43,59]. Melanin’s humidity-sensing capabilities are due to its ability to absorb and interact with water molecules [16,19,79]. This interaction can change melanin’s charge-transport properties, leading to measurable variations in electrical resistance, impedance, or capacitance, detectable by an appropriate sensor [43,98].

Table 5.
Melanin-based humidity sensors.
High-performance humidity sensors based on self-assembled dopamine-melanin thin films have been reported by Hong and coworkers [16], in which films are formed at the air–solution interface during dopamine polymerization and subsequently transferred onto a variety of substrates. The film thickness can be precisely controlled at the nanometer scale by tuning the polymerization time, while atomic force microscopy reveals smooth and compact surface morphologies. These sensors display ultrasensitive, linear, and reproducible responses across the full relative humidity range (0–100% RH), with electrical resistance decreasing by several orders of magnitude as humidity increases. Their response and recovery times are on the order of only a few seconds (typically below 3 s and 7 s, respectively) outperforming many conventional inorganic humidity-sensing materials. The pronounced sensitivity to moisture further enables unconventional applications such as fingertip position detection, highlighting the potential of dopamine-melanin thin films for touchless interfaces, artificial skin, and bionanoelectronic sensing devices.
Further advances have been achieved by the same group, through the development of water-soluble dopamine–melanin, obtained by controlling the initial pH of the dopamine solution. This approach enables the fabrication of dense, uniform nanoscale films via solution processing, which is critical for thin-film bioelectronics. Humidity sensors based on these films exhibit exceptional performance, with resistance changes exceeding five orders of magnitude across the full humidity range and ultrafast response and recovery times below 0.5 s, significantly outperforming conventional inorganic devices [79].
Additionally, a recent study has also demonstrated as a synthetic sulfonated eumelanin derivative can be robustly integrated, through photolithography, into solid-state electronic devices. The humidity sensing measurements performed with these readily prepared bio-inspired sensors revealed conductivity changes spanning up to five orders of magnitude between dry and hydrated states, along with rapid response and recovery when exposed to human breath, underscoring the versatility of eumelanin for physiological monitoring applications [75].
In parallel, the integration of melanin into flexible and printable sensor platforms has emerged as a key strategy for advancing sustainable electronics. In particular, inkjet-printed melanin-based humidity sensors can combine high sensing performance with reduced environmental impact. Figure 5 shows a notable example is the development of flexible humidity sensors fabricated by inkjet printing biobased melanin derived from black soldier fly (BSF) waste onto interdigitated silver electrodes. The addition of choline chloride to the melanin ink increases ionic conductivity and enhances sensitivity while reducing hysteresis. These devices exhibit fast detection and recovery times (~0.8 s) and a strong impedance response, with up to a 170-fold change between 30% and 90% RH. Although prolonged exposure to high humidity can reduce sensitivity, stable long-term operation is achieved, and the sensors’ low embodied energy, substrate reusability, and potential biodegradability make them attractive for wearable, agricultural, and healthcare applications [19].

Figure 5.
Inkjet-printed bio-based melanin composite humidity sensor. Schematic (a) and real (b) representation of inkjet-printed melanin onto a silver interdigitated electrode (IDE) on a carrier substrate, (b) photograph of the sensor; (c) Photograph demonstrating the flexibility of the sensor. Reproduced with permission [19]. Copyright 2024, ACS.
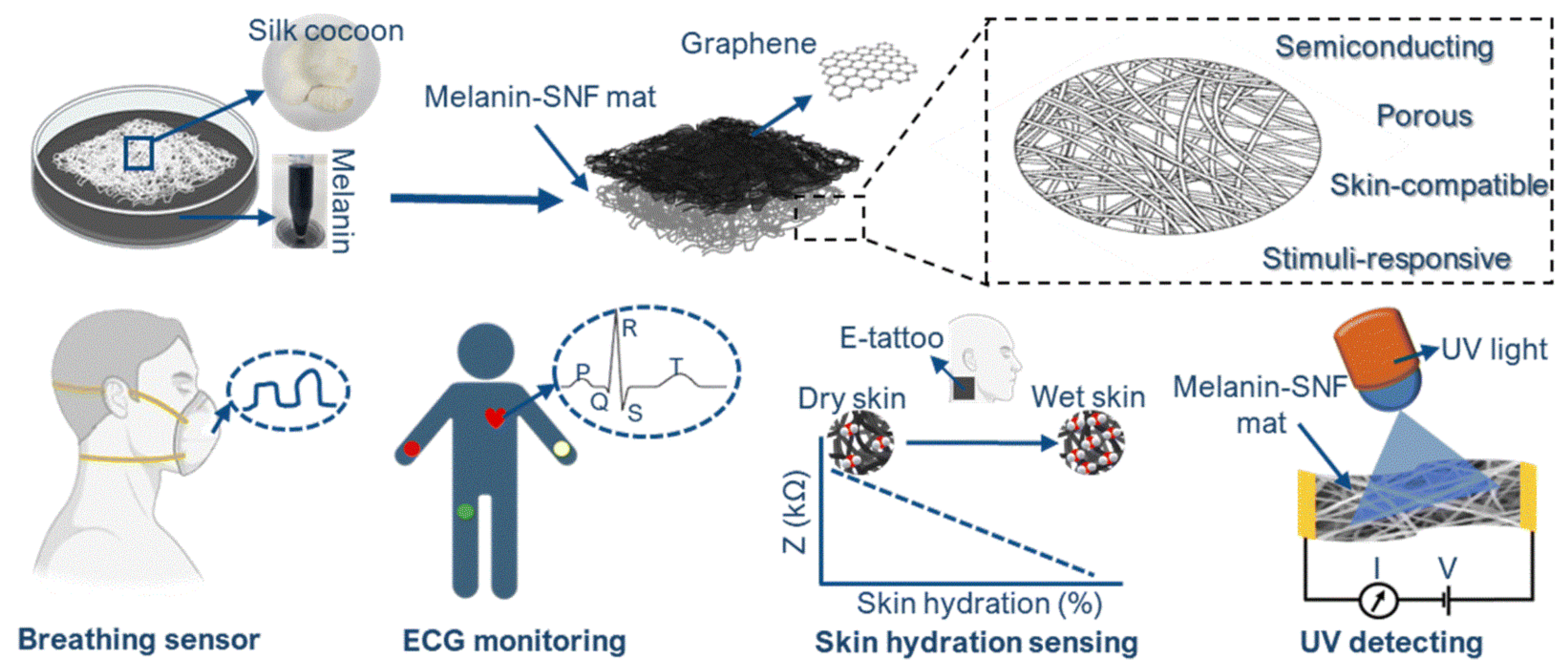
The versatility of melanin-based humidity sensing has recently been extended to multifunctional wearable systems (Figure 6). Ultrathin electronic tattoos composed of melanin-doped silk nanofibers combined with graphene exploit melanin’s humidity-responsive conductivity for skin hydration and breath sensing, while also enabling light and UV detection. These devices conform closely to the skin, maintain breathability and biocompatibility, and allow long-term wear, demonstrating the potential of melanin-based materials for imperceptible, real-time physiological and environmental monitoring [20].
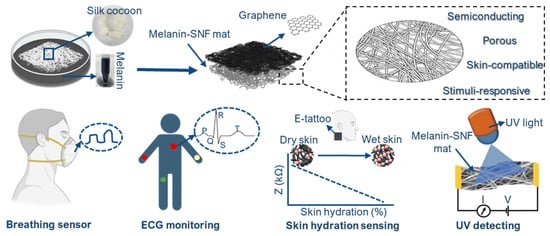
Figure 6.
Schematic illustration showing the fabrication and potential applications of the multifunctional graphene/melanin-doped silk nanofibers electronic tattoo. Reproduced with permission [20]. Copyright 2025, RSC.
Overall, both biosourced and synthetic melanin offers a sustainable, biocompatible, and versatile platform for advanced humidity-sensing applications, enabling the development of high-performance sensors [6,19,98].
4.3. Optical Sensors
Exploiting optical sensors, it is possible to detect changes in light properties when an analyte is present [22,99]. Melanin-based materials, such as melanin films, can offer improvement to this kind of device due to their unique surface chemistry, broadband photodetection, and high electrical conductivity [21,22].
The use of melanin in bio-integrated optoelectronics is indeed another significant development (see Table 6 for significant examples). For example, the incorporation of melanin nanoparticles into silk-protein hydrogel forms a biocompatible, semiconducting layer suitable for bioimage sensors [21,40]. Such devices can interface with biological tissues, offering opportunities in clinical diagnosis and therapy; however, they are particularly sensitive to moisture due to the presence of melanin, and consequently changes in humidity influence the sensors’ performance [21,40].

Table 6.
Melanin-based optical sensors.
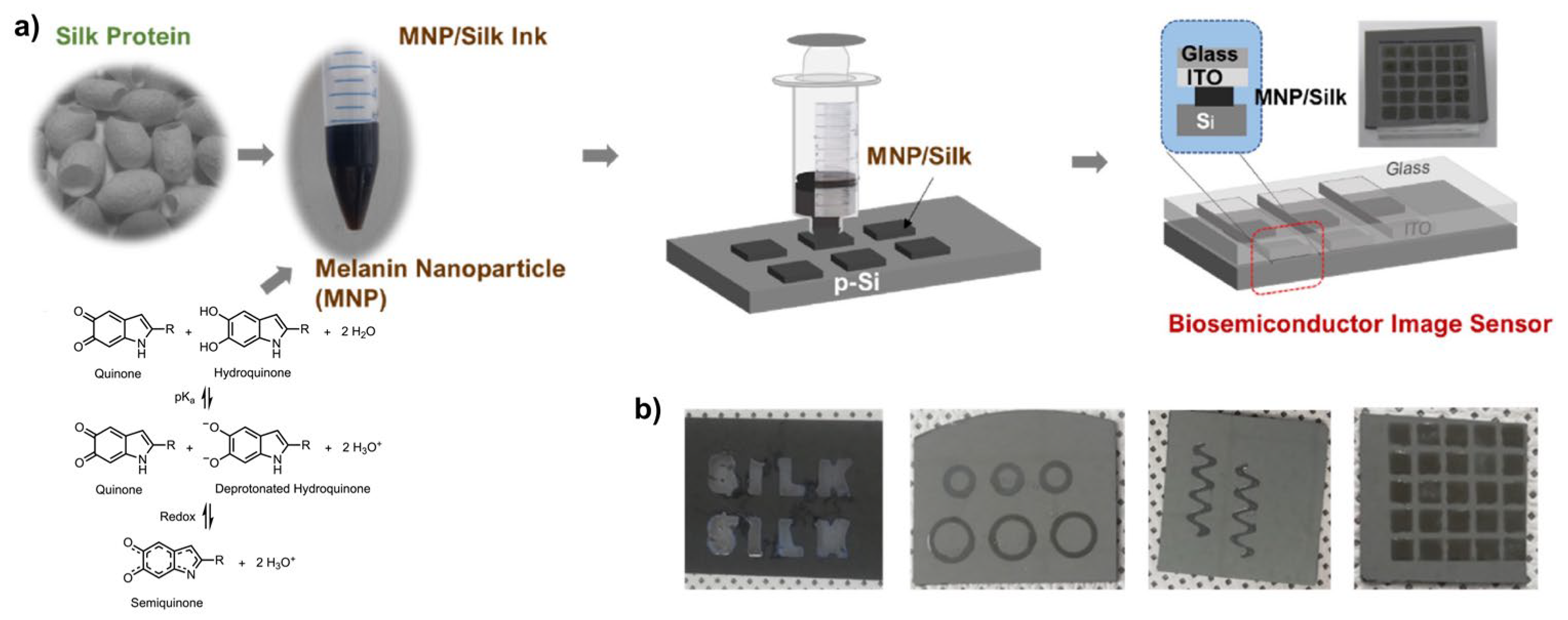
A particularly significant advance (Figure 7) is represented by the incorporation of melanin into bio-integrated optoelectronic systems. For example, melanin nanoparticles embedded in silk-protein hydrogels form biocompatible, semiconducting layers suitable for bioimage sensors and wearable optoelectronics [21]. In these hybrid materials, the hydrated silk matrix enhances ionic conductivity and facilitates efficient charge transport within the melanin network. When interfaced with silicon, these composites form bio–semiconductor junctions capable of broadband photodetection, exhibiting uniform photoresponse from ultraviolet to near-infrared wavelengths. The resulting devices show fast photo-switching behavior driven by effective charge accumulation and transport at the bio-semiconductor/Si interface. Notably, their optoelectronic performance is strongly influenced by ambient humidity, as water uptake in the melanin-containing hydrogel modulates charge transport, rendering these systems inherently sensitive to moisture.
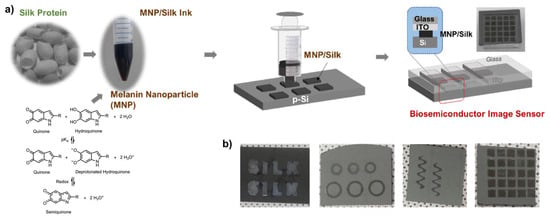
Figure 7.
MNP-silk hydrogel/Si optical sensors. (a) Device fabrication process: 3D-printing of an array on Si followed by its attachment to indium tin oxide (ITO)/glass; (b) Photographs of printed structures of melanin NP-silk hydrogel on silicon. Reproduced with permission [21]. Copyright 2023, Elsevier.
Building on this concept, fully biomaterial-based optoelectronic skins have been developed by the same group using melanin nanoparticles embedded in silk protein hydrogels. These platforms combine transparency, flexibility, and skin compatibility with photoresponsive and p-type semiconducting behavior that is further enhanced under illumination. In addition to photodetection, such optoelectronic skins exhibit self-healing properties and multifunctionality, enabling operation as strain, humidity, and UV light sensors, as well as image pixels that convert light patterns into electrical signals. These features highlight the potential of melanin–silk systems for artificial electronic skin, bio-integrated sensing, and next-generation wearable biomedical devices [40].
Beyond bio-integrated optoelectronics, melanin has also been successfully combined with nanostructured photonic materials to achieve highly selective optical chemosensing. For instance, melanin nanoparticles integrated with nitrogen-doped graphene quantum dots and embedded within molecularly imprinted polymers have been employed as ultrasensitive fluorescence probes for the selective detection of prometryn among triazine herbicides [22]. In this hierarchical nanocomposite, melanin provides bio-inspired affinity, graphene quantum dots act as efficient fluorescence quenchers, and the molecularly imprinted polymer ensures molecular selectivity. The resulting optosensor demonstrates high sensitivity, excellent selectivity, and reliable performance in real samples, offering a cost-effective and environmentally friendly alternative to conventional analytical techniques.
Moreover, melanin-inspired oligomeric derivatives have been theoretically investigated as optical sensing materials for the selective detection of polynitroaromatic compounds. Computational studies indicate strong and reversible interactions between these bioinspired materials and dinitro- and trinitroaromatic analytes, leading to significant modulation of electronic, optical, and vibrational properties. While analyte-dependent absorption shifts are not always consistent, bandgap reductions suggest promising routes for luminescence-based detection, positioning melanin-inspired compounds as sustainable platforms for optical and optoelectronic sensing of toxic nitroaromatic compounds [80].
Melanin has also been integrated with inorganic semiconductors to form hybrid optoelectronic sensors. In particular, melanin/porous silicon heterojunctions have been developed as multifunctional platforms for photodetection and chemical sensing. These diode-like structures exhibit rectifying behavior and have been demonstrated to detect analytes such as glucose and volatile organic compound vapors without the need for additional biological recognition elements. Although their photovoltaic performance remains limited, such heterojunctions are well suited for incorporation as embedded photodetectors within sensor chips, combining optical and chemical sensing capabilities in a biocompatible architecture [81].
Overall, these studies demonstrate that melanin and melanin-inspired materials constitute a versatile and powerful class of optical sensing materials. Their integration into bio-integrated optoelectronics, nanophotonic composites, and hybrid semiconductor devices enables sensitive, selective, and multifunctional optical sensors, with strong potential for applications in environmental monitoring, food safety, healthcare diagnostics, and wearable and implantable bioelectronics.
4.4. Other Melanin-Based Sensors
Melanin nanoparticles are also useful in further sensor-related applications, as summarized in Table 7. Among them, there is photoacoustic imaging, which combines the deep penetration of ultrasonic imaging with the high specificity of optical imaging, while being non-invasive [27], since they can convert light into acoustic waves, which are then detected by a photoacoustic sensor [82]. Recently, the development of optical ultrasound sensors has brought some advantages, including high sensitivity, broad bandwidth, and miniaturization [27]. Owing to their high photothermal conversion efficiency, melanins represent an attractive alternative to noble metal nanoparticles, whose photoacoustic efficiency can be limited by scattering losses and thermal dissipation [27,82,100].

Table 7.
Other melanin-based sensors.
Biomimetic strategies have leveraged melanin’s intrinsic optical functionality at the cellular level. A notable example is the development of whole-cell photoacoustic sensors based on pigment relocalization in melanophore cells. In this system, melanin-filled melanosomes dynamically redistribute within cells in response to ligand binding to G protein–coupled receptors, producing detectable changes in photoacoustic signal amplitude and frequency. This approach represents the first photoacoustic sensor for GPCR-mediated signaling and establishes a generalizable framework for multiplexed molecular sensing using mobile endogenous chromophores [17].
Advances in biosynthetic strategies have enabled scalable production of melanin nanoparticles with tailored optoelectronic properties. Multi-enzyme biosynthesis in Escherichia coli has yielded melanin nanoparticles with enhanced polymerization, stronger near-infrared absorption, and high photothermal conversion efficiency (~66.5%), resulting in improved photoacoustic imaging performance and effective photothermal therapy in breast tumor models. Such biosynthetically regulated systems provide a safe and tunable platform for future cancer diagnostics and therapy [82].
Hybrid melanin–metal nanostructures have also been engineered to further enhance photoacoustic contrast. Ternary core–shell nanoparticles composed of silver cores, DHICA-derived melanin, and a silica matrix demonstrate that synergistic interactions at the metal–polymer interface significantly amplify photoacoustic signals. Precise control over silver content and particle size enables optimization of optical absorption and thermal confinement, while the silica shell stabilizes the structure and prevents aggregation. These biocompatible hybrid nanoparticles show strong potential not only for high-performance photoacoustic imaging but also for combined photothermal therapy and theranostic applications [83].
Moreover, the high affinity of melanin-based materials for metal ions enables the sustainable surface modification of metal and metal-oxide nanoparticles, producing multifunctional biohybrid platforms with magnetic, photothermal, catalytic, antimicrobial, and antioxidant properties [84]. These characteristics are particularly valuable for biosensing, bioimaging, and theranostics, although challenges remain in achieving scalable synthesis, precise control of physicochemical properties, and predictable in vivo behavior.
Melanin–metal hybrids have been successfully applied to nonenzymatic electrochemical sensing. For example, melanin-supported Prussian blue nanoparticles doped with Fe3+, Cu2+, or Ni2+ have been developed as highly sensitive amperometric sensors for hydrogen peroxide. In these systems, melanin serves as an eco-friendly and stable support while enhancing electron transfer and long-term stability. The resulting sensors exhibit low detection limits, wide linear ranges, and successful application in real samples, such as tooth-whitening gels, effectively mimicking enzymatic activity without the associated instability of biological catalysts [18].
Melanin-based materials have also proven highly effective for heavy metal detection using diverse transduction mechanisms. Tea-melanin-coated quartz crystal microbalance sensors represent the first example of piezoelectric platforms capable of real-time, highly sensitive metal ion detection [9]. These systems show exceptional sensitivity to Hg2+ ions, with detection limits down to the ppb range, and selective responses to a wide variety of metal ions. Structural and conformational changes in melanin upon metal binding, confirmed by AFM, circular dichroism, and infrared spectroscopy, underpin the sensing mechanism and highlight the suitability of quartz crystal microbalance as a tool for probing melanin–metal interactions. [9,85].
Complementary electrochemical approaches have employed synthetic melanin nanoparticle–coated electrodes for voltammetric detection of heavy metals such as Pb, Cd, Co, Zn, Ni, and Fe. Melanin films deposited in situ on conductive substrates exhibit stable, reproducible, and highly sensitive responses, with particularly strong selectivity toward ferric ions. These platforms demonstrate excellent linearity and low detection limits, supporting their use in environmental monitoring of water contaminants. [85].
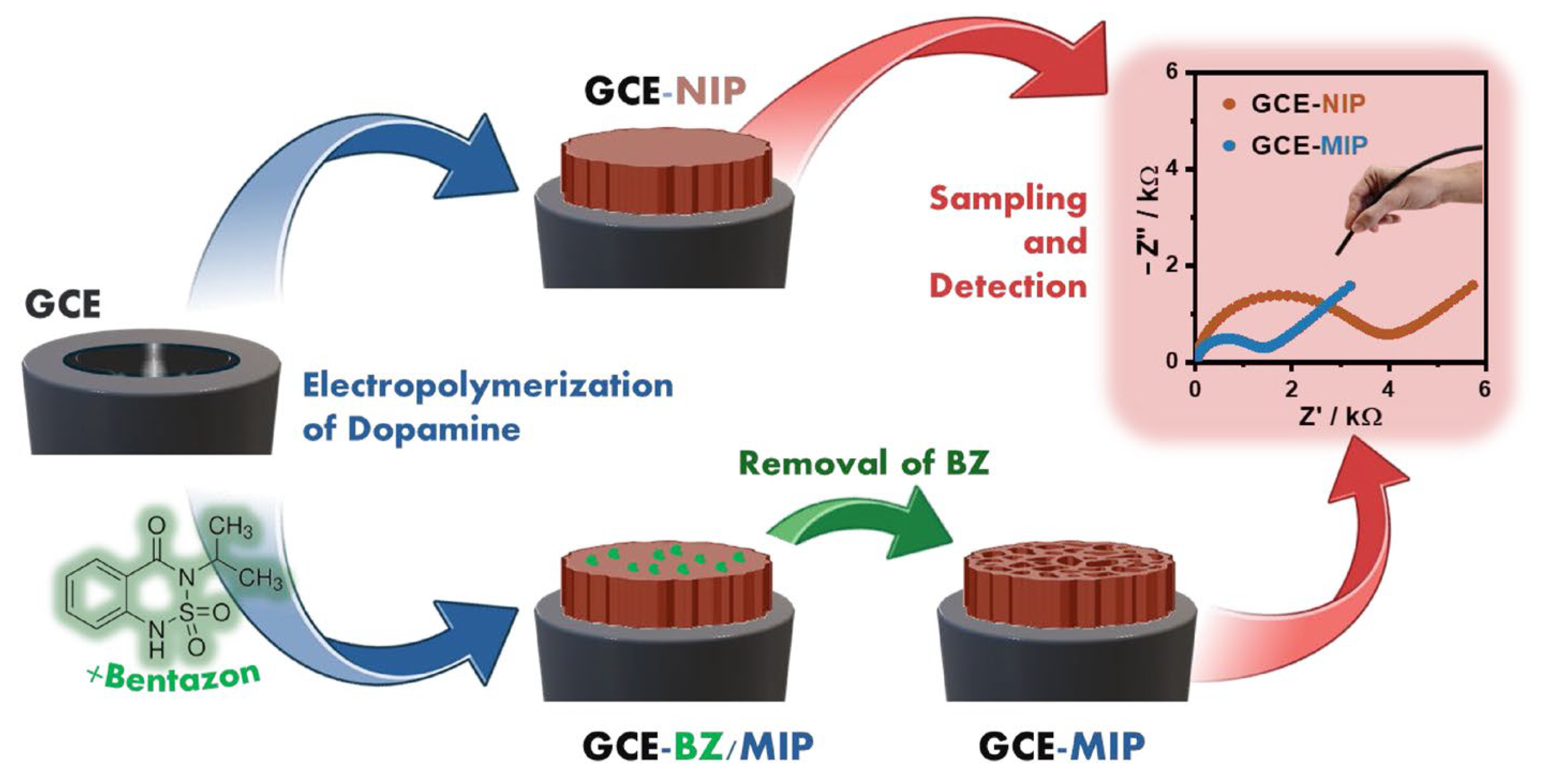
Beyond metals, melanin-based sensors have shown promising performance in the detection of several pharmaceutical products, pesticides, and related contaminants, as thoroughly reported in a recent review by Pane et al. [8]. In this regard, a new electrochemical sensing strategy based on the tunable electropolymerization of dopamine in the presence of bentazon has been developed [86]. Bentazon, an organophosphorus pesticide widely used for insect control, was employed as a template molecule to fabricate a molecularly imprinted polymer on a glassy carbon electrode (Figure 8). During electropolymerization, melanin forms a tailor-made film containing cavities that match the size and stereochemical features of bentazon, resulting in a highly selective recognition interface. Detection is achieved using ferricyanide as a redox probe, with signal changes governed by occupancy of the imprinted cavities. The resulting sensor exhibits remarkable sensitivity, enabling bentazon detection in the femtomolar range, along with excellent selectivity against potential interferents. Importantly, the platform was successfully applied to real water samples without any sample pretreatment, clearly discriminating bentazon-contaminated samples from controls. This approach highlights the broad applicability of dopamine electropolymerization for constructing highly selective and sensitive sensors, which can be readily adapted to detect other pesticides or small-molecule targets by simply changing the template molecule.
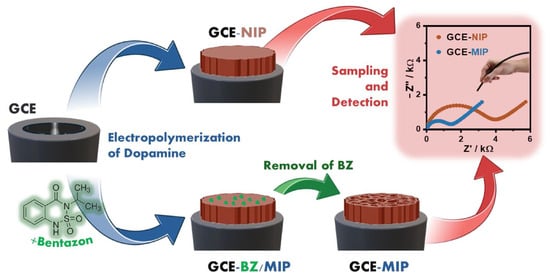
Figure 8.
Process of melanin film formation on the glassy carbon electrode (GCE) in the absence of the target molecule (GCE-NIP) and fabrication of the molecular imprinted polymer (GCE-BZ/MIP) on a GCE (blue arrow), removal of bentazon and opening of selective holes (GCE-MIP) (green arrow) and sampling and detection of bentazon (red arrow). Reproduced with permission [86]. Copyright 2025, Elsevier.
5. Importance and Potential Impacts
Lately, melanin-based sensors are gaining significant attention in scientific research due to the unique physicochemical properties of these biopolymers, among which is an inherent adhesiveness [8]. This, combined with their biocompatibility and versatility, makes them ideal for surface modification and the fabrication of electrochemical sensors. Furthermore, the ease of preparation and modification enhances their appeal in sustainable technological development [19,23,28,29]. Melanin-based sensors can detect environmentally relevant targets, and nanoparticles can be incorporated into environmental sensors to detect toxins, heavy metals, and organic pollutants [22,81]. Due to their ability to undergo reversible redox reactions, melanins can interact with various analytes, transducing chemical signals into measurable electrical responses [47].
In biomedical applications, melanin-based wearable biosensors are revolutionizing healthcare by enabling continuous health monitoring, early disease detection, and personalized medicine [21,40,76,81].
Melanin-based sensors are also exploited in chemical and biological sensing, detecting substances in the body or the environment. For example, glucose sensing is useful for people affected by diabetes, whereas cortisol monitoring provides insight into a person’s stress level [81,101]. Such sensors can facilitate early diagnosis and notify emergency services when critical health data are detected [102].
To create ultrathin interfaces with biological matter, two-dimensional materials can be used, and melanin could be integrated with 2D material-based electronics to enhance the sensor’s performance [76,77,78,86]. In addition, melanin offers environmental sustainability benefits as a biomaterial derived from natural sources [19,24]. Moreover, melanin’s preparation is easy and environmentally friendly, which makes it even more appealing in the biosensors field [19,34,35].
Furthermore, melanin is optimal for optical sensing, due to its photoabsorptive behavior [21,38,40,41]. The interplay between plasmonic fields and optical properties represents another area where melanin’s optical characteristics might be exploited [83,103]. In conclusion, melanin’s properties and its integration into various sensor architectures promise significant advancements in creating more effective, sustainable, and user-friendly sensing solutions for the future.
6. Challenges and Limitations
Despite the several advantages they offer, melanin-based sensors face significant challenges that impede their widespread application, mainly associated with their chemical and structural complexity. A major challenge, indeed, involves reproducibility and standardization, stemming from the inherent structural heterogeneity of both natural and synthetic melanin, in terms of monomer composition, oxidation levels, and impurity content [19,28,31]. This intrinsic variability compromises the reliability and comparability of sensing outcomes, thereby hindering the consistent sensing performance across different production batches [8,28]. Sensitivity, selectivity, and the potential for cross-interference represent further hurdles: multiple mechanisms—not yet fully understood—including surface binding, changes in oxidation state, hydration, and ionic or electronic conduction, all contribute to melanin’s sensing properties [98,104]. Moreover, melanins’ various binding sites for protons, metal ions, and small molecules increase the risk of cross-sensitivity [8,46].
The relative influence of these mechanisms can vary significantly with environmental conditions: parameters such as pH, humidity, and ionic strength can therefore affect the signal for a target analyte [19,21,40]. Processes such as moisture-induced swelling, or slow charge diffusion, potentially coupled with redox chemistry, can contribute to slow sensor kinetics, hysteresis, and signal drift, thereby exerting a critical influence on both sensors’ response and recovery time [21,40,98]. Consequently, achieving long-term stability under harsh environmental conditions remains a significant challenge for these sensors [8,37].
An additional constraint lies in melanin’s relatively low electronic conductivity: although melanin exhibits mixed ionic and electronic conduction, its electrical conductivity remains substantially lower compared to that of conventional inorganic semiconductors or metals [98]. This constraint affects its performance in applications relying on efficient electron transfer—such as advanced biosensors for pathogen detection—where fast response times and low-noise signal processing are essential. While enhancement strategies through composite formation, doping, or morphological engineering can be exploited to improve conductivity, they often introduce additional complexity to the fabrication process [8]. Scalability, cost, and processing represent additional challenges, since obtaining high-purity, consistent-quality melanin, processing it into uniform thin films or nanoscale structures, and integrating them with electrodes demand significant engineering efforts.
Mechanical robustness and integration are also crucial: in the case of wearable, flexible, or implantable devices, the sensing layer must withstand mechanical stress and stable electrical contacts [19,95,96,97]. Fabrication techniques must indeed balance cost-effectiveness, throughput, and performance for widespread adoption. Overcoming these limitations will be vital for melanin-based sensors to realize their full potential across a range of advanced sensing applications.
7. Future Directions
Melanin-based sensors are an emerging area of research, exploiting melanin’s properties for various purposes [19,92,95]. The future directions in this field include enhancing the electrical conductivity and optoelectronics properties of melanin-based materials, improving the fabrication of flexible and wearable electronics, expanding the range of detectable analytes, and improving the integration into sophisticated biosensing platforms for different applications ranging from medical diagnostics to environmental monitoring [7,50,55,66,80,81].
Melanin can be synthesized via oxidative polymerization of dopamine or extracted from plants, animals, bacteria, and fungi [24,25]. Both natural and synthetic melanin exhibit intrinsic biocompatibility, antioxidant activity, free-radical scavenging, and metal ion chelation [48,60,61,62,63,64]. Furthermore, melanin can be incorporated into conductive polymers, forming a material with tunable properties, which makes it useful for sensing in clinical, food quality, and environmental monitoring applications [44].
A fundamental future direction is the development of advanced fabrication techniques. These materials can be obtained through simple, green procedures, making them sustainable and economic [24,34,35]. A screen-printing process has indeed been employed to fabricate an electrochemical pH sensor based on graphene and melanin [77]. Another promising way of sensor preparation could contemplate the integration of melanin-like nanomaterials with laser-induced graphene, aiming to obtain a sensor with tunable surface chemistry, high electrical conductivity, mechanical flexibility, and biocompatibility [105].
Moreover, expanding the range of analytes detectable by melanin-based sensors is also a key focus. Some researchers are trying to obtain sensors for nitroaromatic compounds, which are toxic and dangerous for the environment [80]. Melanin has also been studied to engineer metal and metal-oxide nanoparticles for groundbreaking biomedical applications, including diagnostics and therapeutics [84]. Integration of melanin-based materials into advanced biosensing platforms is also gaining traction. For example, biomimetic melanin-like deposition has been used to enhance the sensitivity of sandwich-type electrochemiluminescence (ECL) immunosensors [106]. Since melanins present a lot of useful properties, optimal in the biosensor field, in the future they can possibly be integrated into microelectromechanical systems (MEMS) [107]. Other properties are still being studied, such as the ability of melanin-like nanoparticles to adsorb light and convert it into heat [108]. Another exciting development is represented by the use of melanin bio-inks on paper-based laser-induced graphemic electrodes for the detection of neurotransmitters [109]. In conclusion, the focus remains on leveraging melanin’s natural advantages while addressing existing challenges to realize its full potential in next-generation sensing technologies.
8. Conclusions
This review has critically examined melanin and melanin-derived materials as emerging platforms for sensing technologies, highlighting both their multifunctional physicochemical nature and the underlying mechanisms that govern their sensing behavior. A key insight is that melanin’s sensing performance is not attributable to a single dominant property, but rather to the complex interplay between hydration-dependent charge transport, redox activity, molecular binding affinity, and supramolecular organization. While this multifunctionality enables applicability across chemical, biological, optical, and environmental sensors, it also introduces intrinsic complexity that must be carefully managed in device design.
Despite a growing number of compelling proof-of-concept demonstrations, the translation of melanin-based sensors from laboratory studies to robust, real-world technologies remains challenging. Structural heterogeneity, batch-to-batch variability, and incomplete control over oxidation state and morphology continue to limit reproducibility and comparability across studies. Moreover, the strong sensitivity of melanin to environmental factors such as humidity, pH, and ionic strength—while advantageous for certain sensing modalities—can lead to signal drift, cross-sensitivity, and long-term instability if not deliberately engineered. These issues underscore the need for standardized synthesis protocols, rigorous materials characterization, and clearer correlations between molecular structure, processing conditions, and device-level performance.
Another critical limitation concerns electrical performance. Although melanin functions as a mixed ionic–electronic conductor, its relatively low electronic conductivity constrains applications that require fast response times, low noise, or high-frequency operation. Strategies based on hybridization with conductive fillers, nanostructuring, or molecular engineering have shown promise, but they often increase fabrication complexity and may compromise sustainability or biocompatibility if not carefully optimized.
Looking forward, progress in this field will depend on a transition from largely empirical demonstrations toward mechanism-driven design principles. Future research should prioritize: (i) the development of chemically defined and reproducible melanin analogues; (ii) advanced in situ and operando techniques to disentangle ionic, electronic, and redox contributions during sensing; (iii) scalable and low-energy fabrication routes compatible with flexible and wearable electronics; and (iv) system-level integration with data processing, multiplexed architectures, and closed-loop sensing platforms. Computational modeling and machine-learning-assisted materials discovery may also play a pivotal role in accelerating rational design and performance optimization.
In conclusion, melanin-based materials occupy a unique position at the intersection of bioinspired chemistry, sustainable materials science, and next-generation sensing. Their inherent ability to couple chemical, optical, and electrical responses within a single biopolymeric platform is particularly compelling for biosensing applications, where biocompatibility, multifunctionality, and environmental adaptability are essential. While substantial challenges remain, addressing them through coordinated interdisciplinary efforts could transform melanin from a versatile laboratory material into a reliable and scalable component of future biomedical, environmental, and industrial sensing systems.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| L-DOPA | L-3,4-dihydroxyphenylphenylalanine |
| DHN | 1,8 dihydroxynaphthalene |
| UV | Ultraviolet |
| NIR | Near-infrared |
| MIECs | Mixed Ionic–Electronic Conductors |
| ROS | Reactive Oxygen Species |
| EGFETs | Extended-Gate Field-Effect Transistors |
| AFM | Atomic Force Microscopy |
| XPS | X-ray photoelectron spectroscopy |
| BSF | black soldier fly |
| IDE | interdigitated electrode |
| GCE | glassy carbon electrode |
| ECL | Electrochemiluminescence |
| MEMS | Microelectromechanical Systems |
References
- Abdulhussain, S.H.; Mahmmod, B.M.; Alwhelat, A.; Shehada, D.; Shihab, Z.I.; Mohammed, H.J.; Abdulameer, T.H.; Alsabah, M.; Fadel, M.H.; Ali, S.K.; et al. A Comprehensive Review of Sensor Technologies in IoT: Technical Aspects, Challenges, and Future Directions. Computers 2025, 14, 342. [Google Scholar] [CrossRef]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Xue, N.; Zhou, W.; Qin, B.; Qiu, B.; Fang, G.; Sun, X. Recent Progress in Flexible Wearable Sensors for Real-Time Health Monitoring: Materials, Devices, and System Integration. Micromachines 2025, 16, 1124. [Google Scholar] [CrossRef]
- Ferreira, R.G.; Silva, A.P.; Nunes-Pereira, J. Current On-Skin Flexible Sensors, Materials, Manufacturing Approaches, and Study Trends for Health Monitoring: A Review. ACS Sens. 2024, 9, 1104. [Google Scholar] [CrossRef] [PubMed]
- Al-Shamery, N.; Biyashev, D.; Blancafort, L.; Camus, A.; Gianneschi, N.C.; De Olivera Graeff, C.F.; Kohler, B.; Li, S.; Lu, K.Q.; Lumb, J.P.; et al. From melanogenesis to melanin technologies. Commun. Chem. 2025, 8, 323. [Google Scholar] [CrossRef]
- Mostert, A.B. Melanin, the What, the Why and the How: An Introductory Review for Materials Scientists Interested in Flexible and Versatile Polymers. Polymers 2021, 13, 1670. [Google Scholar] [CrossRef]
- Eom, T.; Ozlu, B.; Ivanová, L.; Lee, S.; Lee, H.J.; Krajčovič, J.; Shim, B.S. Multifunctional Natural and Synthetic Melanin for Bioelectronic Applications: A Review. Biomacromolecules 2024, 25, 5489. [Google Scholar] [CrossRef]
- Pane, A.; Vicenzi, S.; Mattioli, C.; Mordini, D.; Menichetti, A.; Montalti, M. Melanin-Related Materials in Electrochemical Sensors for Monitoring the Environment and Food. Biosensors 2025, 15, 631. [Google Scholar] [CrossRef]
- Huang, G.S.; Wang, M.-T.; Su, C.-W.; Chen, Y.-S.; Hong, M.-Y. Picogram detection of metal ions by melanin-sensitized piezoelectric sensor. Biosens. Bioelectron. 2007, 23, 319. [Google Scholar] [CrossRef]
- Morresi, L.; Ficcadenti, M.; Pinto, N.; Murri, R.; Cuccilioni, M.; Angeletti, M.; Tombesi, P. Optical and electrical behavior of synthetic melanin thin films spray-coated. Energy Procedia 2010, 2, 177. [Google Scholar] [CrossRef]
- Rubianes, M.D.; Rivas, G.A. Use of a Melanin-Type Polymer to Improve the Selectivity of Glucose Biosensors. Anal. Lett. 2003, 36, 1311. [Google Scholar] [CrossRef]
- González, R.; Sánchez, A.; Chicharro, M.; Rubianes, M.D.; Rivas, G.A. Dopamine and Glucose Sensors Based on Glassy Carbon Electrodes Modified with Melanic Polymers. Electroanalysis 2004, 16, 1244. [Google Scholar] [CrossRef]
- Bothma, J.P.; de Boor, J.; Divakar, U.; Schwenn, P.E.; Meredith, P. Device-Quality Electrically Conducting Melanin Thin Films. Adv. Mater. 2008, 20, 3539. [Google Scholar] [CrossRef]
- Meredith, P. Utilization of Melanin thin films as a potential semiconductor. Adv. Coat. Surf. Technol. 2009, 22, 8. [Google Scholar]
- Piacenti da Silva, M.; Colnaghi Fernandes, J.; Biziak de Figueiredo, N.; Congiu, M.; Mulato, M.; de Oliveira Graeff, C.F. Melanin as an active layer in biosensors. AIP Adv. 2014, 4, 037120. [Google Scholar] [CrossRef]
- Wu, T.; Wee, B.; Hong, J. An Ultrasensitive and Fast Moisture Sensor Based on Self-Assembled Dopamine–Melanin Thin Films. Adv. Mater. Inter. 2015, 2, 1500203. [Google Scholar] [CrossRef]
- Lauri, A.; Soliman, D.; Omar, M.; Stelzl, A.; Ntziachristos, V.; Westmeyer, G.G. Whole-Cell Photoacoustic Sensor Based on Pigment Relocalization. ACS Sens. 2019, 4, 603. [Google Scholar] [CrossRef]
- de Souza, F.S.; Costa, T.G.; Feldhaus, M.J.; Szpoganicz, B.; Spinelli, A. Nonenzymatic Amperometric Sensors for Hydrogen Peroxide Based on Melanin-Capped Fe3+-, Cu2+-, or Ni2+- Modified Prussian Blue Nanoparticles. IEEE Sens. J. 2015, 15, 4749. [Google Scholar] [CrossRef]
- Krebsbach, P.; Rincón-Iglesias, M.; Pietsch, M.; Henel, C.; Lanceros-Mendez, S.; Phua, J.W.; Ambrico, M.; Hernandez-Sosa, G. Inkjet-Printed Bio-Based Melanin Composite Humidity Sensor for Sustainable Electronics. ACS Appl. Mater. Interfaces 2024, 16, 42555. [Google Scholar] [CrossRef]
- Joshi, S.R.; Leea, S.; Kim, S. Semiconducting and environmentally responsive melanin-doped silk nanofibers for multifunctional electronic tattoos. Mater. Horiz. 2025, 12, 4444. [Google Scholar] [CrossRef]
- Gogurla, N.; Wahab, A.; Kim, S. A biomaterial-silicon junction for photodetection. Mater. Today Bio 2023, 20, 100642. [Google Scholar] [CrossRef]
- Chumkaeo, P.; Davis, F.; Bunkoed, O. A melanin-integrated graphene quantum dots embedded molecularly imprinted polymer: A nano-fluorescence sensing probe for trace determination of prometryn. J. Photochem. Photobiol. A 2024, 447, 115247. [Google Scholar] [CrossRef]
- Ozlu, B.; Ahmed, M.B.; Muthoka, R.M.; Wen, Z.; Bea, Y.; Youk, J.H.; Lee, Y.; Yoon, M.H.; Shim, B.S. Naturally derived electrically active materials for eco-friendly electronics. Mater. Today Adv. 2024, 21, 100470. [Google Scholar] [CrossRef]
- D’Ischia, M.; Wakamatsu, K.; Napolitano, A.; Briganti, S.; Garcia-Borron, J.-C.; Kovacs, D.; Meredith, P.; Pezzella, A.; Picardo, M.; Sarna, T.; et al. Melanins and melanogenesis: Methods, standards, protocols. Pigment Cell Melanoma Res. 2013, 26, 616. [Google Scholar] [CrossRef] [PubMed]
- Cao, W.; Zhou, X.; McCallum, N.C.; Hu, Z.; Ni, Q.Z.; Kapoor, U.; Heil, C.M.; Cay, K.S.; Zand, T.; Mantanona, A.J.; et al. Unraveling the Structure and Function of Melanin through Synthesis. J. Am. Chem. Soc. 2021, 143, 2622. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Dhinojwala, A.; Gianneschi, N.; Shawkey, M. Interactions of melanin with electromagnetic radiation: From fundamentals to applications. Chem. Rev. 2024, 124, 7165. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Han, Y.; Dong, J.; Lv, S.; Zhang, R. Melanin/melanin-like nanoparticles: As a naturally active platform for imaging-guided disease therapy. Mater. Today Bio 2024, 23, 100894. [Google Scholar] [CrossRef]
- Pralea, I.-E.; Moldovan, R.-C.; Petrache, A.-M.; Ilieș, M.; Hegheș, S.-C.; Ielciu, I.; Nicoară, R.; Moldovan, M.; Ene, M.; Radu, M.; et al. From Extraction to Advanced Analytical Methods: The Challenges of Melanin Analysis. Int. J. Mol. Sci. 2019, 20, 3943. [Google Scholar] [CrossRef]
- Song, W.; Yang, H.; Liu, S.; Yu, H.; Li, D.; Li, P.; Xing, R. Melanin: Insights into structure, analysis, and biological activities for future development. J. Mater. Chem. B 2023, 11, 7528. [Google Scholar] [CrossRef]
- Heppner, F.; Al-Shamery, N.; Lee, P.S.; Bredow, T. Tuning melanin: Theoretical analysis of functional group impact on electrochemical and optical properties. Mater. Adv. 2024, 5, 5251. [Google Scholar] [CrossRef]
- Choudhury, A.; Ghosh, D. Elucidating the Structure of Melanin and Its Structure–Property Correlation. Acc. Chem. Res. 2025, 58, 1509. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Li, W.; Gu, Z.; Wang, L.; Guo, L.; Ma, S.; Li, C.; Sun, J.; Han, B.; Chang, J. Recent Advances and Progress on Melanin: From Source to Application. Int. J. Mol. Sci. 2023, 24, 4360. [Google Scholar] [CrossRef] [PubMed]
- Wallstein, N.; Capucciati, A.; Pöppl, A.; Schnohr, C.S.; Sturini, M.; Pampel, A.; Jäger, C.; Zecca, L.; Zucca, F.A.; Monzani, E.; et al. Modeling Midbrain and Brainstem Neuromelanins to Characterize Metal Binding and Associated MRI Contrast in Parkinson’s and Alzheimer’s Diseases. Angew. Chem. Int. Ed. 2025, 64, e202509102. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, E.; Capucciati, A.; Prada, I.; Zucca, F.A.; D’Arrigo, G.; Pontiroli, D.; Brindelli, M.G.; Sturini, M.; Bubacco, L.; Monzani, E.; et al. Synthesis, structure characterization, and evaluation in microglia cultures of neuromelanin analogues suitable for modeling Parkinson’s Disease. ACS Chem. Neurosci. 2017, 8, 501. [Google Scholar] [CrossRef]
- Capucciati, A.; Monzani, E.; Sturini, M.; Nicolis, S.; Zucca, F.A.; Bubacco, L.; Bortolus, M.; Zecca, L.; Casella, L. Water-soluble melanin-protein-Fe/Cu conjugates derived from norepinephrine as reliable models for neuromelanin of human brain Locus Coeruleus. Angew. Chem. Int. Ed. 2022, 61, e202204787. [Google Scholar] [CrossRef]
- Sajjad, W.; Din, G.; Rafiq, M.; Iqbal, M.; Khan, S.; Zada, S.; Barkat, A.; Kang, S. Pigment production by cold-adapted bacteria and fungi: Colorful tale of cryosphere with wide range applications. Extremophiles 2020, 24, 447. [Google Scholar] [CrossRef]
- Xue, J.; Wang, Y.; Qi, X.; Zeng, W.; Zhang, Y.; Hong, L. The physicochemical properties and antioxidant and bacteriostatic activities of Auricularia auricula melanin modificated by Arginine. Food Meas. 2024, 18, 7443. [Google Scholar] [CrossRef]
- Shafreen, M.; Mukesh, K.; Saleena, L.M. Microbial cell factories for melanin production: Progress and opportunities for industrial production. Biologia 2024, 79, 1461. [Google Scholar] [CrossRef]
- Watt, A.A.R.; Bothma, J.P.; Meredith, P. The supramolecular structure of melanin. Soft Matter 2009, 5, 3754. [Google Scholar] [CrossRef]
- Wahab, A.; Gogurla, N.; Park, J.Y.; Kim, S. Architecting Silk Protein and Melanin for Photoresponsive and Self-Healable Optoelectronic Skins. Adv. Mater. Technol. 2022, 7, 2101271. [Google Scholar] [CrossRef]
- Qu, B.; Zhang, X.; Han, Y.; Peng, X.; Sun, X.; Zhang, R. IR820 functionalized melanin nanoplates for dual-modal imaging and photothermal tumor eradication. Nanoscale Adv. 2020, 2, 2587–2594. [Google Scholar] [CrossRef]
- Capucciati, A.; Foli, V.; Lioniello, P.; Alfieri, M.L.; Cassera, E.; Merli, D.; Manini, P.; Ravelli, D. Exploring the Reactivity of Melanins as Photocatalysts for Reductive Dehalogenations. Eur. J. Org. Chem. 2024, 27, e202400191. [Google Scholar] [CrossRef]
- Sheliakina, M.; Mostert, A.M.; Meredith, P. Decoupling Ionic and Electronic Currents in Melanin. Adv. Funct. Mater. 2018, 28, 1805514. [Google Scholar] [CrossRef]
- Nozella, N.L.; Morais Lima, J.V.; Furlan de Oliveira, R.; de Oliveira Graeff, C.F. Melanin/PEDOT:PSS blend as organic mixed ionic electronic conductor (OMIEC) for sustainable electronics. Mater. Adv. 2023, 4, 4732–4743. [Google Scholar] [CrossRef]
- Kumar, P.; Di Mauro, E.; Zhang, S.; Pezzella, A.; Soavi, F.; Santato, C.; Cicoira, F. Melanin-based flexible supercapacitors. J. Mater. Chem. C 2016, 4, 9516–9525. [Google Scholar] [CrossRef]
- Paulin, J.V.; Pereira, M.P.; Bregadiolli, B.A.; Cachaneski-Lopes, J.P.; de Oliveira Graeff, C.F.; Batagin-Neto, A.; Bufon, C.C.B. Controlling ions and electrons in aqueous solution: An alternative point of view of the charge-transport behavior of eumelanin-inspired material. J. Mater. Chem. C 2023, 11, 6107–6118. [Google Scholar] [CrossRef]
- Gouda, A.; Soavi, F.; Santato, C. Eumelanin electrodes in buffered aqueous media at different pH values. Electrochim. Acta 2020, 347, 136250. [Google Scholar] [CrossRef]
- Manirethan, V.; Balakrishnan, R.M. Batch and continuous studies on the removal of heavy metals using biosynthesised melanin impregnated activated carbon. Environ. Technol. Innov. 2020, 20, 101085. [Google Scholar] [CrossRef]
- Manirethan, V.; Raval, K.; Rajan, R.; Thaira, H.; Balakrishnan, R.M. Kinetic and thermodynamic studies on the adsorption of heavy metals from aqueous solution by melanin nanopigment obtained from marine source: Pseudomonas stutzeri. J. Environ. Manag. 2018, 213, 315–324. [Google Scholar] [CrossRef]
- Bettinger, C.J.; Bruggeman, J.P.; Misra, A.; Borenstein, J.T.; Langer, R. Biocompatibility of biodegradable semiconducting melanin films for nerve tissue engineering. Biomaterials 2009, 30, 3050–3057. [Google Scholar] [CrossRef]
- Agrawal, L.; Vimal, S.K.; Barzaghi, P.; Shiga, T.; Terenzio, M. Biodegradable and Electrically Conductive Melanin-Poly (3-Hydroxybutyrate) 3D Fibrous Scaffolds for Neural Tissue Engineering Applications. Macromol. Biosci. 2022, 22, 2200315. [Google Scholar] [CrossRef]
- Zeng, Y.; Sun, Q.; Xie, C.; Cao, H.; Liang, Z.; Cao, S.; Tang, B. Melanin Nanoparticle-Enabled Local Drug Delivery Platform for Preventing Postoperative Recurrence of Liver Cancer. ACS Appl. Mater. Interfaces 2025, 17, 40184. [Google Scholar] [CrossRef] [PubMed]
- Karkoszka, M.; Rok, J.; Wrześniok, D. Melanin Biopolymers in Pharmacology and Medicine—Skin Pigmentation Disorders, Implications for Drug Action, Adverse Effects and Therapy. Pharmaceuticals 2024, 17, 521. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Jing, W.; Fu, W.; Yan, Z.; Ma, Y.; Li, X.; Ji, H.; Zhang, R. Melanin theranostic nanoplatform as an efficient drug delivery system for imaging-guided renal fibrosis therapy. Biomater. Adv. 2023, 147, 213333. [Google Scholar] [CrossRef] [PubMed]
- Ballard, M.; Shafiee, A.; Grage, E.; DeMarco, M.; Atala, A.; Ghadiri, E. Inkjet Printing of Synthesized Melanin Nanoparticles as a Biocompatible Matrix for Pharmacologic Agents. Nanomaterials 2020, 10, 1840. [Google Scholar] [CrossRef]
- Zou, Y.; Wang, T.; Lin, X.; Yang, L.; Li, L. Regulation of the Light Absorption and Photothermal Performance of Melanin-Like Polymers. Acc. Chem. Res. 2025, 58, 2815–2829. [Google Scholar] [CrossRef]
- Chen, C.T.; Chuang, C.; Cao, J.; Ball, V.; Ruch, D.; Buehler, M.J. Excitonic effects from geometric order and disorder explain broadband optical absorption in eumelanin. Nat. Commun. 2014, 5, 3859. [Google Scholar] [CrossRef]
- D’Alba, L.; Shawkey, M.D. Melanosomes: Biogenesis, Properties, and Evolution of an Ancient Organelle. Physiol. Rev. 2019, 99, 1–19. [Google Scholar] [CrossRef]
- Nozella, N.L.; Paulin, J.V.; Nogueira, G.L.; Guerra, N.B.; de Oliveira, R.F.; de Oliveira Graeff, C.F. Probing n-Type Conduction in Eumelanin Using Organic Electrochemical Transistors. ACS Appl. Electron. Mater. 2025, 7, 3176–3181. [Google Scholar] [CrossRef]
- Sarna, T.; Swartz, H.M.; Zadlo, A. Interaction of Melanin with Metal Ions Modulates Their Cytotoxic Potential. Appl. Magn. Reason. 2022, 53, 105. [Google Scholar] [CrossRef]
- Cassera, E.; Ferrari, E.; Vignati, D.A.L.; Capucciati, A. The interaction between metals and catecholamines: Oxidative stress, DNA damage, and implications for human health. Brain Res. Bull. 2025, 226, 111366. [Google Scholar] [CrossRef] [PubMed]
- Seagle, B.L.L.; Rezai, K.A.; Gasyna, E.M.; Kobori, Y.; Rezaei, K.A.; Norris, J.R. Time-Resolved Detection of Melanin Free Radicals Quenching Reactive Oxygen Species. J. Am. Chem. Soc. 2005, 127, 11220–11221. [Google Scholar] [CrossRef] [PubMed]
- Rudrappa, M.; Nayaka, S.; Suresh Kumar, R. In Silico Molecular Docking Approach of Melanin Against Melanoma Causing MITF Proteins and Anticancer, Oxidation–Reduction, Photoprotection, and Drug-Binding Affinity Properties of Extracted Melanin from Streptomyces sp. strain MR28. Appl. Biochem. Biotechnol. 2023, 195, 4368–4386. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Kang, M.; Tschirhart, T.; Malo, M.; Dadachova, E.; Cao, G.; Yin, J.J.; Bentley, W.E.; Payne, G.F. Spectroelectrochemical Reverse Engineering Demonstrates That Melanin’s Redox and Radical Scavenging Activities Are Linked. Biomacromolecules 2017, 18, 4084–4098. [Google Scholar] [CrossRef]
- Di Mauro, E.; Rho, D.; Santato, C. Biodegradation of bio-sourced and synthetic organic electronic materials towards green organic electronics. Nat. Commun. 2021, 12, 3167. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, J.; Shi, M.; Zhang, Y. Natural melanin: From biological functions to biofunctionalized nanoparticles in advanced biomedicine. Biomater. Adv. 2025, 176, 214368. [Google Scholar] [CrossRef]
- Ito, S.; Kikuta, M.; Koike, S.; Szewczyk, G.; Sarna, M.; Zadlo, A.; Sarna, T.; Wakamatsu, K. Roles of reactive oxygen species in UVA-induced oxidation of 5,6-dihydroxyindole-2-carboxylic acid-melanin as studied by differential spectrophotometric method. Pigment Cell Melanoma Res. 2016, 29, 340. [Google Scholar] [CrossRef]
- Żądto, A.; Ito, S.; Sarna, M.; Wakamatsu, K.; Mokrzyński, K.; Sarna, T. The role of hydrogen peroxide and singlet oxygen in the photodegradation of melanin. Photochem. Photobiol. Sci. 2020, 19, 654. [Google Scholar] [CrossRef]
- Mammone, T.; Marenus, K.; Muizzuddin, N.; Maes, D. Evidence and utility of melanin degrading enzymes. J. Cosmet. Sci. 2004, 55, 116. [Google Scholar]
- Sadaqat, B.; Khatoon, N.; Malik, A.Y.; Jamal, A.; Farooq, U.; Ali, M.I.; He, H.; Liu, F.J.; Guo, H.; Urynowicz, M.; et al. Enzymatic decolorization of melanin by lignin peroxidase from Phanerochaete chrysosporium. Sci. Rep. 2020, 10, 20240. [Google Scholar] [CrossRef]
- Shin, S.K.; Hyeon, J.E.; Joo, Y.C.; Jeong, D.W.; You, S.K.; Han, S.O. Effective melanin degradation by a synergistic laccase-peroxidase enzyme complex for skin whitening and other practical applications. Int. J. Biol. Macromol. 2019, 129, 181. [Google Scholar] [CrossRef]
- Nadhilah, D.; Andriani, A.; Agustriana, E.; Nuryana, I.; Mubarik, N.R.; Dewi, K.S.; Rahmani, N.; Yanto, D.H.Y.; Ismayati, M.; Perwitasari, U.; et al. Co-catalysis of melanin degradation by laccase-manganese peroxidase complex from Trametes hirsuta OK271075 for application in whitening cosmetics. J. Biocatal. Biotransform. 2024, 42, 273–285. [Google Scholar] [CrossRef]
- Karavasili, D.; Lazaridou, K.; Ntrivala, M.A.; Pitsavas, A.C.; Baziakou, Z.; Papadimitriou, M.; Bikiaris, N.D.; Balla, E.; Terzopoulou, Ζ. Biobased Polymers in Printed Electronics: From Renewable Resources to Functional Devices. Polymers 2026, 18, 301. [Google Scholar] [CrossRef]
- Zhang, Z.; Feng, B.; Yan, J.; Zhao, W.; Sun, J. Advances in bio-based wearable flexible sensors. Green Chem. 2025, 27, 1604. [Google Scholar] [CrossRef]
- Paulin, J.V.; Albano, L.G.S.; Camargo, D.H.S.; Pereira, M.P.; Bregadiolli, B.A.; De Oliveira Graeff, C.F.; Bufon, C.C.B. Eumelanin-based multisensory platform: A case of study for photolithographic patterning. Appl. Mater. Today 2022, 28, 101525. [Google Scholar] [CrossRef]
- Di Capua, R.; Gargiulo, V.; Alfè, M.; De Luca, G.M.; Skála, T.; Mali, G.; Pezzella, A. Eumelanin Graphene-Like Integration: The Impact on Physical Properties and Electrical Conductivity. Front Chem. 2019, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Tehrani, Z.; Whelan, S.P.; Mostert, A.B.; Paulin, J.V.; Ali, M.M.; Daghigh Ahmadi, M.M.; de Oliveira Graeff, C.F.; Guy, O.J.; Gethin, D.T. Printable and flexible graphene pH sensors utilising thin film melanin for physiological applications. 2D Mater. 2020, 7, 024008. [Google Scholar] [CrossRef]
- Whelan, S.P.; Tehrani, Z.; Peacock, M.; Paulin, J.V.; Guy, O.; Gethin, D. Investigation into the Suitability of Screen Printed Graphene-Melanin pH Sensors for Use in Bacterial Culturing Applications. J. Electroanal. Chem. 2022, 904, 115868. [Google Scholar] [CrossRef]
- Wu, T.-F.; Hong, J.-D. Synthesis of water-soluble dopamine–melanin for ultrasensitive and ultrafast humidity sensor. Sens. Actuators B Chem. 2016, 224, 178. [Google Scholar] [CrossRef]
- Cachaneski-Lopes, J.P.; Hawthorne, F.; Woellner, C.F.; Nelson, T.L.; Hiorns, R.C.; de Oliveira Graeff, C.F.; Bégué, D.; Batagin-Neto, A. Melanin-Based Compounds as Low-Cost Sensors for Nitroaromatics: Theoretical Insights on Molecular Interactions and Optoelectronic Responses. ACS Omega 2025, 10, 31908. [Google Scholar] [CrossRef]
- Volynskyi, D.; Dusheiko, M.; Madan, R.; Kutuzov, N.; Obukhova, T. Melanin/porous silicon heterojunctions for solar cells and sensors applications. In Proceedings of the 2020 IEEE 40th International Conference on Electronics and Nanotechnology (ELNANO); IEEE: New York, NY, USA, 2020; Volume 3, pp. 343–346. [Google Scholar]
- Yang, L.; Huang, Y.; Liu, C.; Wang, Y.; Wang, H.; Yan, F.; Su, Z. Metabolic Regulation of Biosynthesis of Melanin Nanoparticles for Enhancing Photothermal Therapy of Breast Tumor. Small 2025, 21, e06711. [Google Scholar] [CrossRef]
- Silvestri, B.; Armanetti, P.; Pota, G.; Vitiello, G.; Pezzella, A.; Menichetti, L.; Giannini, V.; Luciani, G. Enhanced Photoacoustic Response by Synergistic Ag–Melanin Interplay at the Core of Ternary Biocompatible Hybrid Silica-Based Nanoparticles. ACS Appl. Mater. Interfaces 2023, 15, 46756. [Google Scholar] [CrossRef]
- Abdullah, W.; Razak, N.N.A.N.A.; Dheyab, M.A.; Salem, F.; Aziz, A.A.; Alanezi, S.T.; Oladzadabbasabadi, N.; Ghasemlou, M. Melanin-Driven Green Synthesis and Surface Modification of Metal and Metal-Oxide Nanoparticles for Biomedical Applications. Adv. Funct. Mater. 2025, 35, 2503017. [Google Scholar] [CrossRef]
- Hefny, M.; Orabi, R.G.; Kamel, M.M.; Kalil, H.; Bayachou, M.; Mostafa, N.Y. Exploring the Electrochemical Signatures of Heavy Metals on Synthetic Melanin Nanoparticle-Coated Electrodes: Synthesis and Characterization. Appl. Nano 2025, 6, 11. [Google Scholar] [CrossRef]
- Córdoba-Aceituno, M.; Cuesta-Chamorro, C.M.; Sánchez-Obrero, G.; Madueño, R.; Blázquez, M.; Pineda, T.; Del Caño, R. Polydopamine as a molecularly imprinted polymer (MIP) for the detection of Bentazon. Electrochim. Acta 2025, 454, 146679. [Google Scholar] [CrossRef]
- Taheri, M.; Deen, I.A.; Packirisamy, M.; Deen, M.J. Metal Oxide-Based Electrical/electrochemical Sensors for Health Monitoring Systems. TrAC Trends Anal. Chem. 2024, 171, 117509. [Google Scholar] [CrossRef]
- Youssef, K.; Ullah, A.; Rezai, P.; Hasan, A.; Amirfazli, A. Recent Advances in Biosensors for Real Time Monitoring of PH, Temperature, and Oxygen in Chronic Wounds. Mater. Today Bio 2023, 22, 100764. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Fu, D.; Ma, T.; Chen, M.; Wang, F.; Chen, G.; Wang, Z. Sensing Patches for Biomarker Identification in Skin-Derived Biofluids. Biosens. Bioelectron. 2024, 258, 116326. [Google Scholar] [CrossRef]
- Ren, H.; Liang, K.; Li, D.; Chen, Y.; Tang, Y.; Wang, Y.; Li, F.; Liu, G.; Zhu, B. Field-Effect Transistor-Based Biosensor for pH Sensing and Mapping. Adv. Sens. Res. 2023, 2, 2200098. [Google Scholar] [CrossRef]
- Janićijević, Ž.; Nguyen-Le, T.-A.; Baraban, L. Extended-Gate Field-Effect Transistor Chemo- and Biosensors: State of the Art and Perspectives. Next Nanotechnol. 2023, 3, 100025. [Google Scholar] [CrossRef]
- Salvo, P.; Melai, B.; Calisi, N.; Paoletti, C.; Bellagambi, F.; Kirchhain, A.; Trivella, M.G.; Fuoco, R.; Di Francesco, F. Graphene-based devices for measuring pH. Sens. Actuators B Chem. 2018, 256, 976. [Google Scholar] [CrossRef]
- Ahmad, K.; Raza, W.; Oh, T.W. Progress in graphene quantum dots, graphene oxide and reduced graphene oxide based materials for chemoresistive gas sensors. J. Environ. Chem. Eng. 2025, 13, 118157. [Google Scholar] [CrossRef]
- Salem, A.H.M.; Zhang, J.; Lam, A.; Wang, H.; Cheema, U.; Lovat, L.B.; Tiwar, M.K. Robust pH Sensing Using a Graphene Oxide and Covalent Organic Frameworks Composite for Gastro-esophageal Reflux Disease Diagnosis. Adv. Healthc. Mater. 2025, 14, e02106. [Google Scholar] [CrossRef] [PubMed]
- Orzari, L.O.; Kalinke, C.; Silva-Neto, H.A.; Rocha, D.S.; Camargo, J.R.; Coltro, W.K.T.; Janegitz, B.C. Screen-Printing vs Additive Manufacturing Approaches: Recent Aspects and Trends Involving the Fabrication of Electrochemical Sensors. Anal. Chem. 2025, 97, 1482. [Google Scholar] [CrossRef] [PubMed]
- Zub, K.; Hoeppener, S.; Schubert, U.S. Inkjet Printing and 3D Printing Strategies for Biosensing, Analytical, and Diagnostic Applications. Adv. Mater. 2022, 34, 2105015. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, M.; Zhang, L.; Xu, J.; Xiao, X.; Zhang, X. Inkjet-printed flexible sensors: From function materials, manufacture process, and applications perspective. Mater. Today Commun. 2022, 31, 103263. [Google Scholar] [CrossRef]
- Vahidzadeh, E.; Kalra, A.P.; Shankar, K. Melanin-Based Electronics: From Proton Conductors to Photovoltaics and Beyond. Biosens. Bioelectron. 2018, 122, 127. [Google Scholar] [CrossRef]
- Paul, T.; Roy Choudhury, D.; Ghosh, D.; Saha, C. Advancements in Optical Sensors for Explosive Materials Identification: A Comprehensive Review. Results Chem. 2024, 8, 101602. [Google Scholar] [CrossRef]
- Luo, C.; Zhou, R.; Li, Y.; Li, M.; Wen, X.; Li, M.-Y.; Deng, S.; Liu, S.; Gao, H.; Lu, H. High-Efficiency Photoacoustic Transducers Based on Plasmonic EGaIn Liquid Metal Nanoparticles. Nanoscale 2025, 17, 13880. [Google Scholar] [CrossRef]
- Cao, X.; Halder, A.; Tang, Y.; Hou, C.; Wang, H.; Duus, J.Ø.; Chi, Q. Engineering Two-Dimensional Layered Nanomaterials for Wearable Biomedical Sensors and Power Devices. Mater. Chem. Front. 2018, 2, 1944. [Google Scholar] [CrossRef]
- Kireev, D.; Kutagulla, S.; Hong, J.; Wilson, M.N.; Ramezani, M.; Kuzum, D.; Ahn, J.-H.; Akinwande, D. Atomically Thin Bioelectronics. Nat. Rev. Mater. 2024, 9, 906. [Google Scholar] [CrossRef] [PubMed]
- Sadrolhosseini, A.R.; Hamidi, S.M. Recent Applications of Quantum Plasmonic Techniques for Detection of Toxic Materials. Plasmonics 2024, 20, 5573. [Google Scholar] [CrossRef]
- Bradley, M.; Barrett, S.; McKelvey, T.; Carpenter, J.; Dean, D. Mitigating Melanin-Induced Bias in Pulse Oximetry: Optical, Algorithmic, Engineering, Hardware and Modeling Tools. Sens. Bio-Sens. Res. 2025, 50, 100876. [Google Scholar] [CrossRef]
- Dinh Le, T.-S.; Tran, Y.-V.; Gao, Y.; Valerio, V.L.; Ge, Z.; Lim, C.T. Laser-Induced Graphene for Biomedical Applications: Innovations in Health Monitoring and Diagnostics. Nanoscale Horiz. 2025, 10, 2688. [Google Scholar] [CrossRef]
- Ma, H.; Zhao, Y.; Liu, Y.; Zhang, Y.; Wu, D.; Li, H.; Wei, Q. A Compatible Sensitivity Enhancement Strategy for Electrochemiluminescence Immunosensors Based on the Biomimetic Melanin-Like Deposition. Anal. Chem. 2017, 89, 13049. [Google Scholar] [CrossRef]
- Priya, S.; Mohan, S.; Kuppusamy, R.; Suyambulingam, I.; Baby, B.; Ramesh, R.; Han, S.S. Advances in Bio-Microelectromechanical System-Based Sensors for Next-Generation Healthcare Applications. ACS Omega 2025, 10, 34088. [Google Scholar] [CrossRef] [PubMed]
- Menichetti, A.; Vicenzi, S.; Pane, A.; Mordini, D.; Mancin, F.; Montalti, M. Photothermal Release by Melanin-like Nanoparticles: Biomedical Applications. J. Funct. Biomater. 2025, 16, 243. [Google Scholar] [CrossRef]
- Isasmendi, J.; Castrillo Fernández, M.; Weiss, L.; Trouillon, R.; Santato, C. Sepia Melanin Bio-Inks on Paper-Based Laser-Induced Graphenic Electrodes for Neurotransmitter Detection. Meet. Abstr. 2025, 1, 2885. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.