Abstract
The accurate monitoring of metal ions is essential for applications that include environmental protection, food safety, and biomedical diagnostics. These areas depend on highly sensitive and selective methods for detecting both toxic and biologically relevant ions. Electrochemical sensors have emerged as promising devices due to their excellent sensitivity, cost-effectiveness, and ease of use. Within these sensor systems, ionophores, either synthetic or natural ligands that exhibit selective ion binding, are fundamental in boosting analytical performance. This review outlines the current progress of ionophore-based electrochemical sensors for metal-ion analysis, emphasizing material selection, architectural strategies, and practical applications. Key classes of ionophores, such as crown ethers, calixarenes, Schiff bases, porphyrins, and oxime derivatives, are discussed with an emphasis on their recognition mechanisms. We also examine strategies for incorporating ionophores into diverse electrochemical sensor configurations and explore recent advances in technologies, such as all-solid-state sensor construction and the development of portable analytical devices. This review bridges the chemistry of ionophores with sensor engineering and serves as a resource for the rational development of advanced platforms for metal-ion sensing.
1. Introduction
The accurate detection of metal ions is critical in diverse sectors, such as environmental monitoring, food safety regulation, and biomedical diagnostics. The demand for analytical techniques with high sensitivity and selectivity has steadily increased in these domains [1,2]. Many metal ion detection methodologies have been established in recent decades. Notable examples are atomic absorption spectroscopy, inductively coupled plasma mass spectrometry, and fluorescence spectroscopy [3,4]. However, these prevailing analytical techniques exhibit considerable shortcomings. They necessitate costly apparatus, elaborate sample pretreatment, and extensive operator expertise. In addition, they frequently show limited applicability for real-time or on-site investigations. Because of these constraints, electrochemical sensors have been identified as promising alternatives for future metal-ion analysis systems. These sensors provide several key benefits, including compactness, reduced cost, expedited analysis, and enhanced portability [5]. Among the various strategies aimed at improving analytical performance, the integration of ionophores into sensor design has attracted interest as a strategy to concurrently optimize selectivity and sensitivity [6,7].
Ionophores, whether synthetic or natural, are ligands with selective affinity for specific metal ions [8,9,10,11]. In electrochemical sensors, ionophores act as molecular recognition elements that impart chemical selectivity by forming reversible complexes with the target ions. Typically, they function as either neutral or ionic carriers and are incorporated in small amounts into ion-selective membranes (ISM) that contain a plasticizer and lipophilic ionic sites (ion exchangers) [12,13]. Ionophores selectively bind and partition the target ion when embedded within a polymeric membrane or immobilized on a functionalized electrode surface. This process alters ionic activity and chemical potential within the membrane phase. This complexation process induces distinct variations in potential, current, or impedance, enabling highly selective and sensitive metal-ion detection [14,15,16]. The structural characteristics of ionophores, including charge, coordination geometry, hydrophobicity, membrane thickness, and immobilization method, serve as key design parameters that determine trade-offs between sensitivity, selectivity, and stability [17,18].
In recent years, various advanced functional materials, such as covalent organic frameworks (COFs), metal–organic frameworks, and porous carbons, have been extensively investigated for electrochemical sensing applications [19,20,21]. However, these materials often require complex processes, such as crystallization or high-temperature synthesis. They rely on further steps to achieve ion selectivity, including post-synthetic modification or pore engineering. In contrast, ionophores possess well-defined coordination sites that can be precisely tuned at the molecular level according to the ionic radius, charge, and electronic affinity of the target metal ion [22]. Moreover, ionophore-based membranes can be easily fabricated using simple solution-based methods (e.g., solution casting and drop-casting) [23]. This approach offers low-cost, scalable fabrication and provides excellent compatibility with soft polymeric matrices. Imine-based COFs are susceptible to hydrolysis under acidic or alkaline conditions [24]. In contrast, many ionophores, like crown ethers, calixarenes, and Schiff-base derivatives, show superior hydrolytic stability, often due to intramolecular hydrogen bonding and hydrophobic shielding [25]. These features identify ionophores as a unique class of molecularly designable components that combine high selectivity, structural versatility, and chemical robustness for metal-ion detection.
This review comprehensively examines the structural characteristics and mechanistic principles of ionophores governing metal-ion selectivity. We used these insights to systematically summarize electrochemical sensor design strategies incorporating ionophores and compare operational mechanisms and structural architectures according to measurement techniques. Recent developments aimed at enhancing ionophore performance are also discussed. Finally, we outline future perspectives to guide innovation in next-generation electrochemical metal ion-sensing technologies.
2. Types of Ionophores
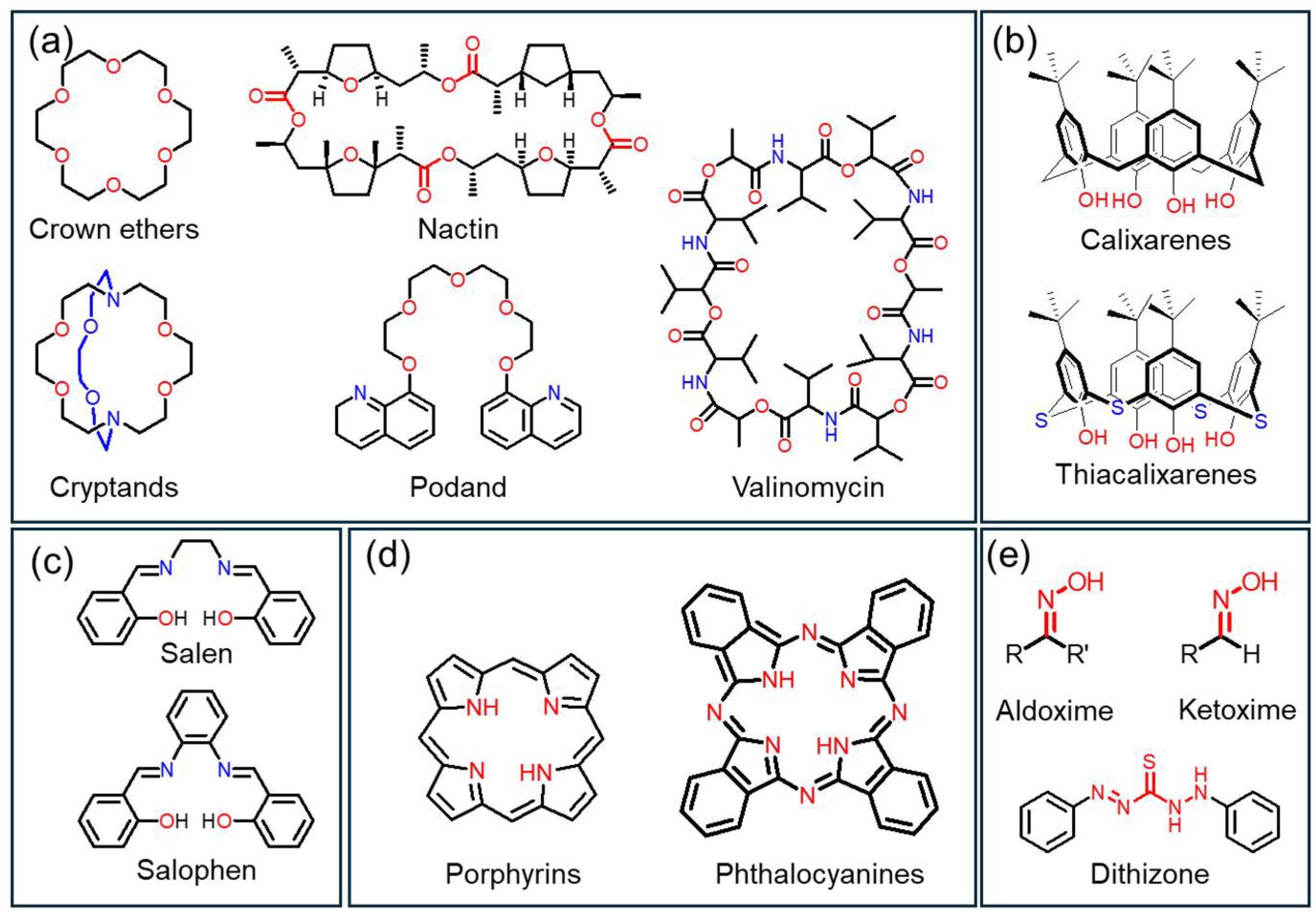
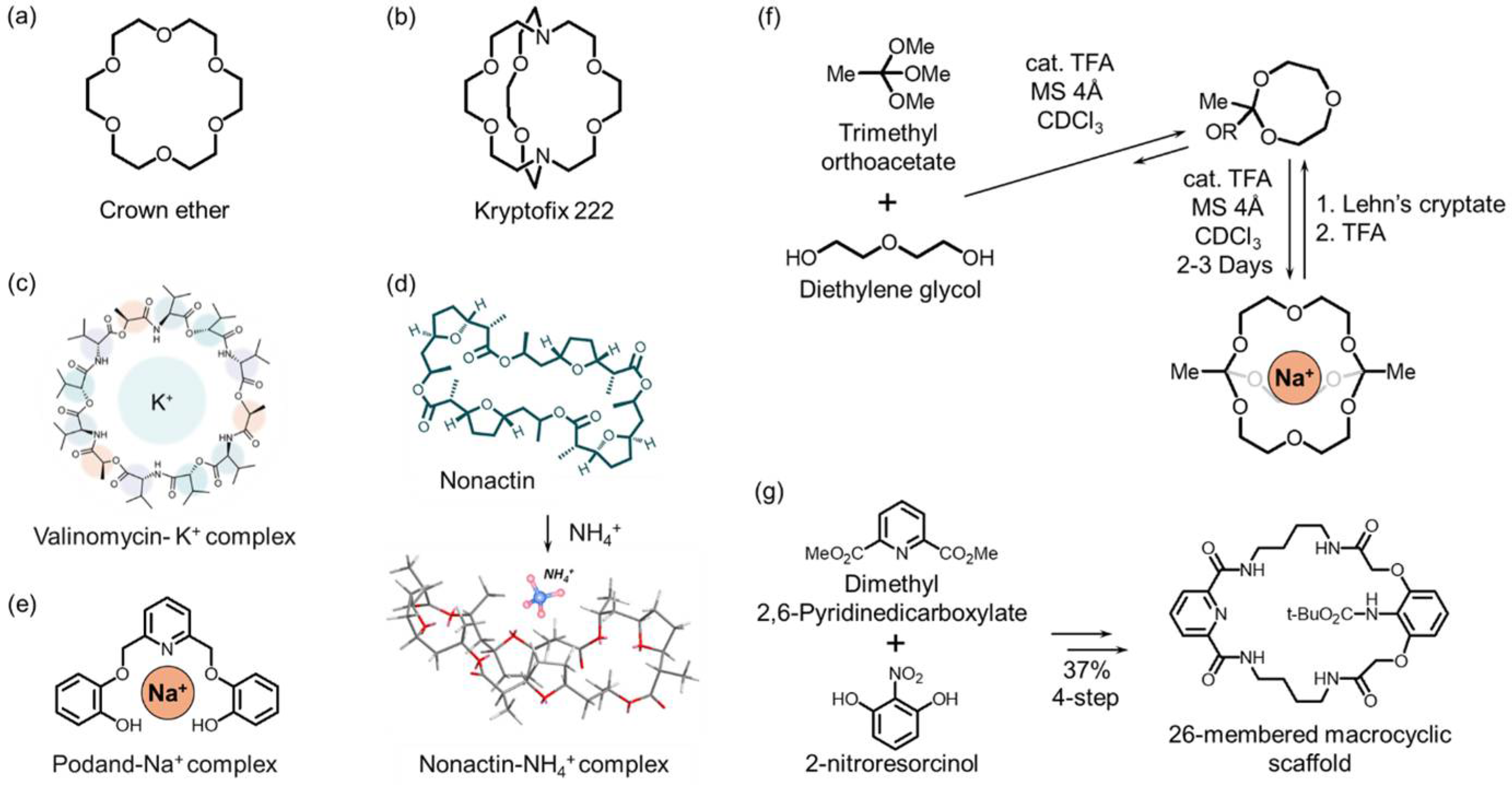
This review categorizes ionophores according to structural families as macrocycles (crowns, cryptands, valinomycin, nactin, and podand), calix/thiacalixarenes, Schiff bases (salen/salophen), porphyrins/phthalocyanines, and oxime/dithizone derivatives (Figure 1). Factors in each scaffold, such as cavity size, donor arrangement, and rigidity/electronic characteristics, determine binding strength and the transport mechanism. The following sections elaborate on these details.
Figure 1.
Classification of ionophores by chemical structure: (a) macrocycles (crown ethers, cryptands, nactin, valinomycin) and podand (b) calix- and thiacalixarenes, (c) Schiff bases (salen/salophen), (d) porphyrins/phthalocyanines, and (e) oxime/dithizone derivatives.
2.1. Macrocyclic Ligands
Macrocyclic ionophores selectively capture metal ions in an internal cavity precisely organized with multidentate donor atoms. Representative macrocyclic ionophore classes include crown ethers, cryptands, and biologically derived macrocyclic compounds, such as valinomycin and nactins. These compounds exhibit the macrocyclic effect, which refers to markedly enhanced binding affinity and selectivity compared to their acyclic analogs [26,27]. This effect arises from preorganized conformations and restricted flexibility, which minimize entropic losses during complexation, resulting in thermodynamically more stable host-guest complexes.
Crown ethers are cyclic polyethers consisting of repeating –(CH2CH2O)– units (Figure 2a). Since their discovery by Pedersen in 1967, crown ethers have been shown to form 1:1 or 2:1 (sandwich type) host-guest complexes with various cations and exhibit excellent chemical and thermal stability and broad applicability in electrochemical sensors [28]. Ion selectivity is not solely governed by cavity size or donor atom type, but by a combination of energetic and structural factors, including solvation and desolvation energies, complex stability, electron density distribution, and conformational rigidity. In particular, selectivity among alkali metal ions can be quantitatively explained not only by size match and charge density compatibility but also by differences in complex formation free energy (ΔGcomplex) [29,30]. Aromatic-ring-containing derivatives, such as benzo-oxa-crowns, exhibit enhanced photophysical properties and have been applied to study complex stability and structure, fluorescent sensing, molecular logic devices, and Cs+ extraction from nuclear waste [31]. Even simple derivatives (e.g., 12-crown-4) have enabled the selective detection of Al3+, Zn2+, Cu2+, Li+, and Pb2+, leading to the development of numerous sensors [32,33].
Cryptands are macrobicyclic hosts in which bridgehead linkers cross to form a three-dimensional cage-like cavity (Figure 2b) [34]. They provide stronger guest encapsulation and finer control of size and shape recognition than crown ethers. This behavior arises from geometrical complementarity and differences in ΔGcomplex and stability constants (β). A classic example, Kryptofix K222, exhibits binding selectivity in the order of Ba2+ > Sr2+ > K+ > Ca2+ > Na+ > Li+, Mg2+, reflecting the combined effects of solvation and ionic size on thermodynamic selectivity. By tailoring the cavity size and introducing side-arm functionalities, cryptands can be optimized for target ions with specific charge densities, solvation properties, and complex stability differences [35]. For instance, macrobicyclic cryptands functionalized with bis(picolinate) arms demonstrate high stability constants (log β ≈ 1.4–4.0) and efficient labeling for Ba2+, Sc3+, La3+, Lu3+, In3+, and Pb2+, making them valuable in the field of radiopharmaceutical chemistry [36].
Valinomycin is a cyclic depsipeptide in which ester and amide linkages alternate to create a cavity that nearly matches the ionic radius of K+ (1.33 Å) (Figure 2c). Consequently, it exhibits a binding affinity for K+ more than 104 times higher than that for Na+ or Li+ and has long served as the benchmark ionophore for K+-selective electrodes [37,38]. Similarly, nactin-type molecules (e.g., nonactin, monactin, and dinactin), derived from Streptomyces species, possess 16-membered cyclic structures that form highly symmetric complexes with NH4+ and K+ through ester carbonyl and ether oxygen donors (Figure 2d) [39,40]. Both valinomycin and nactins exemplify how the preorganization of donor atoms within a rigid macrocyclic framework leads to outstanding thermodynamic stability and selectivity.
Podands are acyclic multidentate ligands lacking a ring structure. They consist of a central atom (typically N or P) connected to flexible donor arms such as –O– or –NH– groups (Figure 2e). Although structurally noncyclic, they are functionally related to macrocyclic ionophores. Podands do not exhibit the macrocyclic effect but serve as valuable acyclic analogs and model systems for understanding ion-ligand interactions. Their structural flexibility allows rapid association–dissociation kinetics and facilitates synthesis, providing advantages in sensor design, such as fast response and reproducibility. During the 1970s and 1980s, Simon’s research group developed numerous podand-based neutral carrier ion-selective electrodes for Na+, Ca2+, and H+ ions, laying the foundation for modern neutral-carrier and optical sensors [41,42].
From a synthetic standpoint, modern chemical methodologies enable the efficient construction of macrocycles and cryptands. Dynamic covalent chemistry, performed through acid-catalyzed or metal-templated self-assembly, allows reversible component exchange that directs the formation of high-yield monometallic cryptates. The resulting cage structures can then be disassembled under acidic conditions to release encapsulated guest ions (Figure 2f) [43]. In addition, hydrogen-bond and halide (e.g., Cl−)-templated assembly strategies can produce high yields (~61%) of 26-membered macrocycles without requiring high-dilution conditions. The subsequent mild, selective late-stage functionalization of endocyclic (lariat) substituents enables the generation of diverse derivatives, including unclosed cryptand structures (Figure 2g) [44].
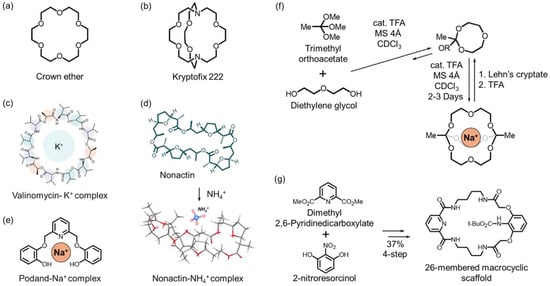
Figure 2.
(a) Representative crown ether structures. (b) Structure of the cryptand Kryptofix 222. (c) Schematic molecular structures for valinomycin-K+ complex. Reprinted with permission from Ref. [38] Copyright 2009 Sage Journals. (d) Schematic molecular structures for nonactin and nonactin-NH4+ complex. Reprinted with permission from Ref. [40] Copyright 2022 American Chemical Society. (e) Schematic molecular structures for podand-Na+ complex. (f) Orthoester-exchange-driven dynamic covalent self-assembly to metal-templated cryptates. Reprinted with permission from Ref. [43]. Copyright 2015 Springer Nature. (g) Hydrogen-bond/halide (Cl−)-templated macrocyclization (precursor to “unclosed cryptand” architectures). Reprinted with permission from Ref. [44]. Copyright 2015 American Chemical Society.
Figure 2.
(a) Representative crown ether structures. (b) Structure of the cryptand Kryptofix 222. (c) Schematic molecular structures for valinomycin-K+ complex. Reprinted with permission from Ref. [38] Copyright 2009 Sage Journals. (d) Schematic molecular structures for nonactin and nonactin-NH4+ complex. Reprinted with permission from Ref. [40] Copyright 2022 American Chemical Society. (e) Schematic molecular structures for podand-Na+ complex. (f) Orthoester-exchange-driven dynamic covalent self-assembly to metal-templated cryptates. Reprinted with permission from Ref. [43]. Copyright 2015 Springer Nature. (g) Hydrogen-bond/halide (Cl−)-templated macrocyclization (precursor to “unclosed cryptand” architectures). Reprinted with permission from Ref. [44]. Copyright 2015 American Chemical Society.
2.2. Calixarenes and Thiacalixarenes
Calixarenes are phenolic macrocycles produced by the base-catalyzed condensation of phenol with formaldehyde. Due to their versatility, including the encapsulation of neutral and ionic guests, extraction, sensing, and catalysis, as well as their high degree of structural tunability, calixarenes are widely used as ionophore scaffolds [45,46]. One of their main advantages is the ability to independently modify the lower rim (phenolic O positions) and the upper rim (para positions), permitting precise control over cavity polarity, rigidity, and donor configuration, attributes that are especially valuable in electrochemical sensing [47,48]. Modification of the upper rim commonly starts with the removal of tert-butyl groups and protection of the lower rim, followed by Ni-catalyzed halogenation or iodination, subsequent reduction, and a Suzuki–Miyaura coupling to introduce donor groups, fluorophores, or redox-active tags. Similar methodologies are utilized for lower-rim functionalization (Figure 3a) [49].
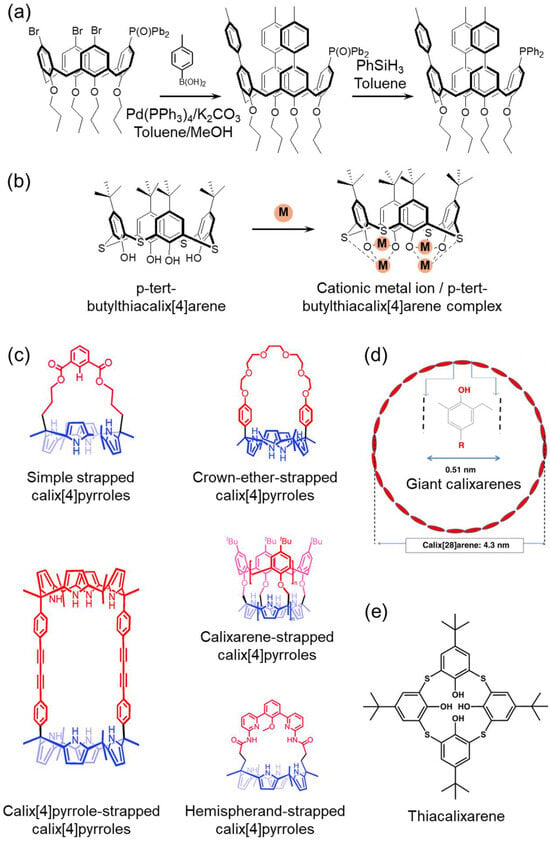
Figure 3.
(a) Functionalization process of calixarene molecules. Reprinted with permission from Ref. [49]. Copyright 2011 Arkivoc. (b) Conformation changes in calixarene molecules. Reprinted with permission from Ref. [50]. Copyright 2019 Taylor & Francis. (c) Different structures of calixarene-based molecules. Reprinted with permission from Ref. [51]. Copyright 2022 Royal Society of Chemistry. (d) Molecular structure of giant calixarenes. Reprinted with permission from Ref. [52]. Copyright 2019 Springer Nature. (e) Structure of thiacalixarene molecules.
Conformations, such as cone, partial-cone, 1,2-alternate, and 1,3-alternate conformations, play a critical role in preorganization and dictate the stereochemical pathway of ion recognition. The selection of base (e.g., NaH, Cs2CO3, t-BuOK) and specific protection/deprotection protocols during synthesis can shift the populations of different conformers and affect the overall yield, enabling the installation of O4/O6 donor arrays or mixed O/S donor systems to optimize binding to the target ion (Figure 3b) [50]. Additionally, structural modifications, such as calix-crown and calix-quinone/pyrrole/pyridine architectures, further broaden the accessible scope to include ion pairs, anions, and cations (Figure 3c) [51].
Synthetic advancements now enable giant calixarene formation, where the phenolic repetition unit count can be tailored from ~17 to ~90. The formation of larger macrocycles is favored by heavier alkali bases (e.g., RbOH and CsOH), increased base/phenol ratios, specific processing conditions, and solvent polarity. These variables allow the precise modulation of cavity geometry to meet the requirements of ion capture or transport applications (Figure 3d) [52].
Thiacalixarenes, in which methylene bridges are substituted with sulfur atoms, introduce lower rotational energy barriers (enhanced flexibility), a slightly expanded cavity size, and an overall softer donor environment, thereby increasing their selectivity for metal ions, such as Pb2+, Hg2+, Ag+, and Cu2+ (Figure 3e) [53]. In particular, the sulfur-bridged thiacalix[4]arene ionophore provides an optimal balance of cavity geometry and donor softness, resulting in markedly enhanced complexation stability toward Cu2+ ions [54].
2.3. Schiff Bases (Salophen and Salen Derivatives)
Schiff bases (imines or azomethines) represent a versatile class of ionophores characterized by a functional C=N bond that serves as the principal motif for metal-ion recognition. This bond is formed through a condensation reaction between a primary amine and an aldehyde (or ketone). The imine nitrogen (N), together with the adjacent phenolic oxygen (O), provides lone-pair electron donors, enabling the formation of stable coordination complexes with a wide range of metal ions (Figure 4a) [55]. Schiff-base ionophores exhibit precise molecular tunability, allowing the donor atom density, molecular rigidity, and electronic properties to be freely adjusted by varying substituents, bridging units, or aromatic spacers. In particular, salen-type (derived from ethylenediamine and salicylaldehyde) and salophen-type (based on o-phenylenediamine) structures feature an N2O2 donor array, forming square-planar, thermodynamically stable metal–ligand complexes with transition-metal ions (Figure 4b) [56]. Such coordination geometries offer both high thermodynamic stability and reversible complexation behavior (chelation reversibility).
The metal-ion selectivity of Schiff-base ionophores arises from the cooperative contribution of multiple factors, including: (1) the ionic radius and charge of the metal ion, (2) electronic interactions between metal d-orbitals and donor-atom lone pairs, (3) hard/soft matching behavior based on the hard-soft acid-base (HSAB) principle, and (4) the geometric configuration and rigidity of the donor-defined cavity [57,58]. These parameters collectively determine the ΔGcomplex and β value, which serve as key thermodynamic indicators for quantitatively evaluating ionophore selectivity toward specific metal ions.
From a photophysical perspective, Schiff-base ionophores undergo distinct changes in their electronic structures and optical properties upon metal coordination. Complexation with metal ions suppresses C=N isomerization and internal bond rotations, thereby enhancing π-conjugation and molecular planarity. Simultaneously, phenolic O–H deprotonation and metal–ligand charge transfer alter the HOMO-LUMO energy gap, leading to measurable variations in absorption intensity and emission wavelengths (Figure 4c) [59,60,61,62]. These structural rigidifications and electronic rearrangements manifest as chelation-enhanced fluorescence or metal-induced quenching, depending on the redox character and coordination strength of the bound metal ion. Chemically, Schiff-base ionophores offer excellent synthetic accessibility and structural versatility. The incorporation of electron-withdrawing groups (e.g., –NO2 and –CF3) increases the complexation stability with soft metal ions such as Cu2+ and Zn2+, while electron-donating groups (e.g., –OH and –OCH3) enhance basicity and strengthen affinity toward hard metal ions such as Fe3+ and Al3+ (Figure 4d). Moreover, the introduction of aromatic backbones or increased structural rigidity further improves complex stability and hydrolytic resistance, preventing decomposition under humid or mildly basic conditions.
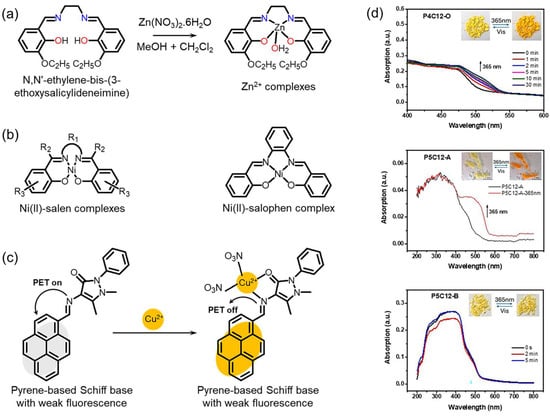
Figure 4.
(a) Molecular design of Shiff base molecule for metal ion sensor. Reprinted with permission from Ref. [55]. Copyright 2019 Elsevier. (b) Metal–ligand geometry using different Shiff bases. Reprinted with permission from Ref. [56]. Copyright 2023 MDPI. (c) The principle of light emission enhancement based on the detection of metal ions in Schiff base. Reprinted with permission from Ref. [61]. Copyright 2018 Elsevier. (d) Implementation of aggregation-induced emission using Schiff Base. Reprinted with permission from Ref. [62]. Copyright 2022 Elsevier.
Figure 4.
(a) Molecular design of Shiff base molecule for metal ion sensor. Reprinted with permission from Ref. [55]. Copyright 2019 Elsevier. (b) Metal–ligand geometry using different Shiff bases. Reprinted with permission from Ref. [56]. Copyright 2023 MDPI. (c) The principle of light emission enhancement based on the detection of metal ions in Schiff base. Reprinted with permission from Ref. [61]. Copyright 2018 Elsevier. (d) Implementation of aggregation-induced emission using Schiff Base. Reprinted with permission from Ref. [62]. Copyright 2022 Elsevier.
2.4. Porphyrins and Phthalocyanines
Porphyrins and phthalocyanines belong to the tetrapyrrolic macrocycle (porphyrinoid) family, each containing an 18π-electron-conjugated system. Their electronic structures and coordination characteristics can be precisely modulated by the choice of the central metal and the peripheral substituents located at the meso- and β- positions [63,64]. This high degree of tunability makes them highly valuable as ionophore scaffolds capable of selective metal-ion recognition and binding.
Free-base porphyrins, which possess four pyrrolic nitrogen atoms, primarily form stable coordination complexes with cations by donating their lone pairs. In contrast, metalloporphyrins, particularly those incorporating Co(II) or Ni(II), often retain a residual positive charge because the positive charge of the central metal is not fully compensated by the porphyrin ligand framework. As a result, these complexes can engage in electrostatic interactions or coordination with nearby anions (e.g., Cl−, NO3−, and SCN−). Such anion binding is considered a characteristic feature of metallated porphyrins rather than the porphyrin ligand itself [65,66,67]. In other words, the ability to bind anions does not arise from the intrinsic binding properties of the porphyrin macrocycle but from the charge imbalance created upon metal coordination.
These structural and electronic modulations are typically achieved through two major design strategies. First, the metallation process, which introduces different central metals such as Co, Cu, or Fe, reshapes the electron distribution of the macrocycle and alters the inherent coordination tendencies of the metal. Second, the installation of electron-withdrawing groups or electron-donating groups at the meso- or β-positions is a key method for fine-tuning ligand properties. These groups fine-tune the hardness or softness of the donor atoms that interact with the metal center. Ultimately, this modulation influences the overall β value of the resulting metal–ligand complex, thereby controlling its stability [68,69]. Additional modifications of anchoring groups, such as –thiol, pyridyl, or sulfonate functionalities, further enhance immobilization behavior and polarity control, thereby influencing coordination equilibria and charge-transfer characteristics. Collectively, these design variables determine essential ionophore performance metrics, including ion-binding selectivity, metal–ligand binding strength, and electron-transfer efficiency (Figure 5a) [70].
Some porphyrinoid ionophores exhibit ion-binding behavior that depends strongly on the oxidation state of the central metal. Such oxidation-state modulation serves as a powerful means to fine-tune binding affinity, charge distribution, and ion-transport equilibria. For example, Co(II)- or Ni(II)-centered metalloporphyrins exhibit selective anion binding due to their residual positive charge, in which both the electronic environment of the metal and the redistribution of electron density across the ligand contribute to selective recognition processes (Figure 5b) [71]. Phthalocyanines, which share structural and electronic similarities with porphyrins, also allow the precise regulation of metal-ion coordination stability and selectivity by adjusting the central metal (e.g., Co and Ni) and peripheral substituents. Their strong d-orbital-π-conjugation interactions enhance the binding strength and stability of metal–ligand complexes, enabling the selective recognition of specific metal ions (Figure 5c,d) [72,73,74,75].
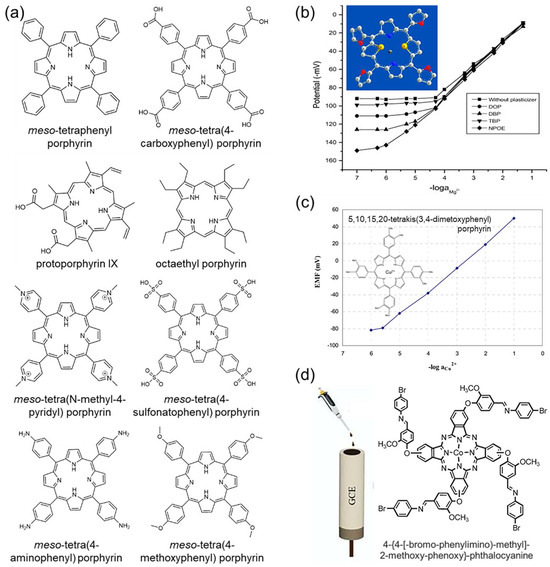
Figure 5.
(a) Synthetic modification of the porphyrin molecule. Reprinted with permission from Ref. [70]. Copyright 2023 Wiley. (b) Porphyrin-based metal ion selection electrode. Reprinted with permission from Ref. [71]. Copyright 2011 Elsevier. (c) Application of free base porphyrin without metal center to ionophore. Reprinted with permission from Ref. [73]. Copyright 2008 MDPI. (d) Phthalocyanine-based ion selection electrode. Reprinted with permission from Ref. [75]. Copyright 2020 Elsevier.
Figure 5.
(a) Synthetic modification of the porphyrin molecule. Reprinted with permission from Ref. [70]. Copyright 2023 Wiley. (b) Porphyrin-based metal ion selection electrode. Reprinted with permission from Ref. [71]. Copyright 2011 Elsevier. (c) Application of free base porphyrin without metal center to ionophore. Reprinted with permission from Ref. [73]. Copyright 2008 MDPI. (d) Phthalocyanine-based ion selection electrode. Reprinted with permission from Ref. [75]. Copyright 2020 Elsevier.
2.5. Oxime and Dithizone Derivatives
Oxime and dithizone derivatives represent major classes of chelating ligands containing N, O (or N, S) donor atoms. These ligands provide high molecular design versatility, as their electronic and steric properties can be precisely tuned through simple synthetic modifications and substituent variations [76,77]. Such structural flexibility is essential for ionophore functionality, enabling selective coordination with specific metal ions. In particular, functional groups such as C=N–OH (oxime) and C=S–NH (dithizone) can easily undergo isomerization and deprotonation, promoting resonance stabilization and maximizing chelation-induced rigidity upon metal binding. These structural features act as key mechanisms that enhance both the binding selectivity and binding sensitivity of the ligand (Figure 6a) [78,79].
Oxime-based ligands exhibit high selective affinity toward divalent cations such as Co2+. In these coordination systems, anionic additives such as sodium tetraphenylborate (NaTPB) stabilize the ion-pairing equilibrium and facilitate the formation of ionophore–metal complexes, thereby significantly improving selective binding performance [80]. Moreover, modified oxime derivatives, such as benzothiazine-dioxime, demonstrate strong coordination selectivity toward Ni2+ (Figure 6b) [81].
Dithizone (1,5-diphenylthiocarbazone), which contains soft S and N donor atoms, exhibits strong binding affinity toward metal ions such as Pb2+, Cu2+, and Ag+. It has also been widely utilized for selective Ni2+ recognition (Figure 6c) [82]. Due to the chemical nature of these ligands, pH variation plays a crucial role in determining their metal-binding behavior. Changes in pH alter the degree of deprotonation of the C=N–OH or C=S–NH groups, which in turn affects both the coordination form of the ligand and its intrinsic stability. Consequently, pH-dependent protonation–deprotonation equilibria are a key factor controlling the stability and selectivity of metal–ligand binding interactions.
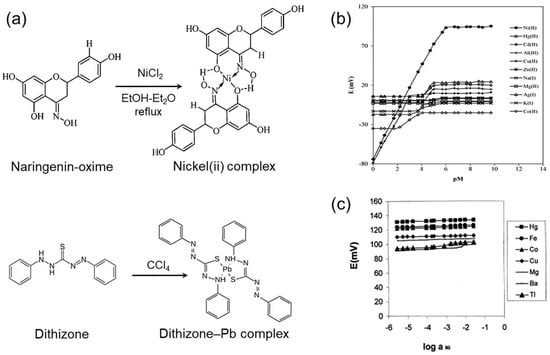
Figure 6.
(a) Transformation of oxime and dithizone. Reprinted with permission from Refs. [78,79]. Copyright 2023 Royal Society of Chemistry & copyright 2016 Springer Nature. (b) Selectivity of oxime-based molecules for nickel ions. Reprinted with permission from Ref. [81]. Copyright 2006 Elsevier. (c) Selectivity of dithizone-based molecules for nickel ions. Reprinted with permission from Ref. [82]. Copyright 2001 Elsevier.
Figure 6.
(a) Transformation of oxime and dithizone. Reprinted with permission from Refs. [78,79]. Copyright 2023 Royal Society of Chemistry & copyright 2016 Springer Nature. (b) Selectivity of oxime-based molecules for nickel ions. Reprinted with permission from Ref. [81]. Copyright 2006 Elsevier. (c) Selectivity of dithizone-based molecules for nickel ions. Reprinted with permission from Ref. [82]. Copyright 2001 Elsevier.
3. Application as an Ion-Selective Electrode Component
3.1. Structure and Working Principle of an Ion-Selective Electrode
An ion-selective electrode (ISE) is an electrochemical transducer that selectively measures the activity of a specific ion and generates an electromotive force (EMF) [83,84]. An ISE operates in conjunction with a reference electrode. A typical polymeric-membrane ISE consists of an ionophore, a plasticizer, and a lipophilic ionic additive (Figure 7a). The membrane functions through extraction and ion-exchange equilibria occurring at the sample/membrane interface, thereby forming a phase-boundary potential (Figure 7b). Consequently, the EMF follows the Nernst equation and varies logarithmically with ion activity [85,86]. Within the membrane, the ionophore forms reversible complexes with the target ion. This process lowers the chemical potential of the ion in the membrane phase and stabilizes the thermodynamic equilibrium at the phase boundary. Therefore, the electrode response should be interpreted not in terms of kinetic barrier lowering but as thermodynamic stabilization resulting from complex formation. Complexation decreases free ion activity, leading to a well-defined equilibrium potential that exhibits Nernstian or near-Nernstian slopes [87,88].
The stability of the ion-ionophore complex is governed by factors such as the cavity size of the ionophore, the spatial arrangement of donor atoms, the ionophore’s charge state (neutral or ionic), and the dielectric constant of the plasticizer, as well as ionic additive concentrations [89]. The leaching resistance and immobilization mode of the ionophore (bulk doping or surface tethering) directly affect long-term stability, potential reproducibility, and the response kinetics of the electrode.
ISE architectures are generally classified as liquid-contact and solid-contact types. A liquid-contact ISE contains an internal filling solution connected to an internal reference electrode; this solution typically consists of 0.01–0.1 M chloride (e.g., KCl or AgCl) of the respective ion [90]. Such systems provide excellent reproducibility and standardized calibration procedures but suffer from practical limitations, such as electrolyte leakage, demanding maintenance, position sensitivity, and restrictions on miniaturization (Figure 7c). ISE selectivity is optimized by tuning the combination of the ionophore and ionic additive and by adjusting the polarity and hydrophobicity of the plasticizer. These design principles are grounded in the classical theoretical framework and standard methodology established by Morf in 1981 [84].
A solid-contact ISE (SC-ISE) eliminates the need for an internal solution by directly coupling the membrane to the electronic conductor through an interfacial charge-transfer layer (Figure 7d). Two main strategies are employed to stabilize the potential at this interface: (1) mixed ionic–electronic conducting layers, which utilize conducting polymers, redox-active systems, or ion-to-electron exchange resins that mediate ion-to-electron transduction and stabilize the interfacial potential; and (2) high-capacitance layers, composed of carbonaceous materials that minimize potential drift through charge accumulation [91]. Recently, hybrid SC-ISE designs integrating both mechanisms have been developed to maximize interfacial capacitance, suppress water-layer formation, and enhance long-term potential stability and reproducibility.
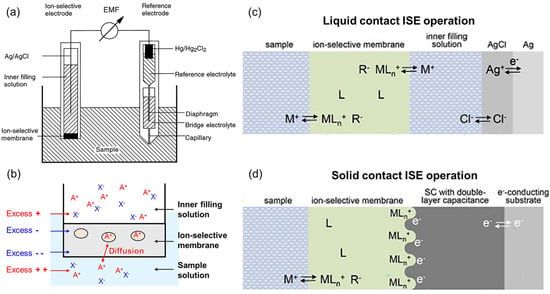
Figure 7.
(a) The overall structure of ISE Reprinted with permission from Ref. [85]. Copyright 1997 American Chemical Society. (b) The overall operation principle of ISE. (c) Liquid contact ISE operation principle (operation mechanism near internal filling liquid). Reprinted with permission from Ref. [90]. Copyright 2016 Elsevier. (d) All-solid contact ISE operation principle (operation mechanism in the middle layer center). Reprinted with permission from Ref. [92]. Copyright 2024 MDPI.
Figure 7.
(a) The overall structure of ISE Reprinted with permission from Ref. [85]. Copyright 1997 American Chemical Society. (b) The overall operation principle of ISE. (c) Liquid contact ISE operation principle (operation mechanism near internal filling liquid). Reprinted with permission from Ref. [90]. Copyright 2016 Elsevier. (d) All-solid contact ISE operation principle (operation mechanism in the middle layer center). Reprinted with permission from Ref. [92]. Copyright 2024 MDPI.
3.2. Principles of Selectivity Design of ISE Using Ionophore
Ionophore-based ISEs achieve selectivity through two key processes at the molecular level. The first involves the chemical equilibrium formed within the membrane phase, and the second concerns the ion-exchange and charge-transfer mechanisms that occur at the sample/membrane interface. The primary design objective is to maximize the membrane-phase stability constant (βmem) of the ionophore-ion complex formed within a plasticized polymer matrix containing lipophilic ionic sites, while simultaneously minimizing the partitioning of interfering ions [92,93]. Selectivity originates from the thermodynamics of complexation. The quantitative relationship between competing ions can be expressed by the selectivity coefficient (). Therefore, the selectivity of an ionophore for a target ion relative to a reference ion is determined directly by the difference in ΔGcomplex. Recent studies involving calixarenes and calix[4]pyrroles elucidated correlations between βmem and interionic selectivity, demonstrating that the ionophore selectivity observed in ISEs can be accurately predicted from thermodynamic data [94,95,96]. The critical factors to consider in the design of ISEs include HSAB compatibility, the geometry and size of the cavity, the spatial arrangement of donor atoms (N/O/S), and the charge state of the ionophore (neutral or ionic). These parameters, together with the dielectric constant and hydrophobicity of the plasticizer and the type and concentration of the lipophilic ionic sites, can be finely optimized to achieve significant improvement in practical selectivity [2].
3.2.1. Primary Coordination–Chemistry Design
The coordination chemistry of an ionophore forms the fundamental core of ion selectivity in ISEs. The ionophore’s donor atom type (N/O/S), coordination number, geometry, and cavity rigidity collectively determine the βmem, which dictates the thermodynamic preference of a metal ion for complexation within the membrane phase. The resulting stability of the ion-ligand complex directly governs both the equilibrium potential and the Nernstian slope behavior of ISEs [97].
Beyond donor-atom identity, the coordination denticity and geometry of the ligand play crucial roles in defining selectivity. Bidentate and tridentate ligands often form small chelate rings that enhance kinetic stability but may leave open coordination sites vulnerable to interference. In contrast, tetradentate and hexadentate macrocyclic ligands, such as crown ethers, calixarenes, and Schiff-base complexes, preorganize donor atoms in a rigid spatial arrangement, minimizing conformational entropy and maximizing thermodynamic selectivity [98,99]. The coordination geometry, whether square-planar, tetrahedral, or octahedral, further dictates the spatial compatibility between the metal ion’s electronic configuration and the ligand donor set, following crystal field stabilization and HSAB-matching principles [100,101].
Aromatic-ring extension in the ligand framework provides enhanced π-conjugation and charge delocalization, which stabilizes metal–ligand interaction via π-backbonding or induced dipole effects. For instance, extending a salicylaldehyde-derived Schiff base to a naphthyl- or anthracenyl-extended analog increases rigidity and field strength, improving Cu2+ and Ni2+ complexation [102]. Likewise, the incorporation of fused aromatic systems in calixarene or porphyrinoid scaffolds has been shown to improve selectivity through enhanced cavity rigidity and charge transfer capacity [103].
The presence and position of specific functional groups also critically modulate donor strength and selectivity. Electron-donating substituents (–OH, –OCH3, and –NH2) increase electron density at donor atoms, promoting stronger binding to hard cations such as Fe3+ and Al3+. Conversely, electron-withdrawing substituents (–NO2, –CF3, and –CN) lower basicity and favor coordination with softer cations like Ag+, Pb2+, and Hg2+ [104]. The arrangement and symmetry of donor sites, whether linear, cyclic, tripodal, or macrocyclic, control the preorganization and steric accessibility of the binding cavity, which directly affects βmem and overall metal-ion recognition efficiency [105].
Researchers have systematically tuned these coordination factors through donor-atom combinations (e.g., N2O2, On, or N/S mixed donors), macrocycle size optimization, and side-arm (lariat) substitutions to achieve superior selectivity and operational stability. Such structural tuning optimizes the geometric complementarity between the ionophore and target ion and minimizes non-specific binding and the partitioning of interfering ions, resulting in improved ISE performance reproducibility and selectivity [106].
3.2.2. Secondary Interactions and Ligand Preorganization
When primary coordination alone does not confer sufficient selectivity, secondary interactions, such as lariat (side-arm) donor effects, hydrogen bonding, π-cation/anion interactions, and hydrophobic pocket effects, play essential roles in reinforcing the ion-ligand binding environment. Ligand preorganization minimizes the entropic penalty associated with complexation, facilitating more stable thermodynamic binding. Furthermore, adjusting the pKa of donor sites (e.g., imine nitrogen or phenolic oxygen) and introducing electron-withdrawing groups or electron-donating groups promotes deprotonation and enhances resonance stabilization during metal-ion coordination. These effects have been well established in the design of crown ethers, salen/salophen (N2O2), and porphyrinoid ionophores, and directly contribute to improved membrane-phase selectivity and reproducibility [70].
3.2.3. Membrane-Phase Engineering
The selectivity and detection limit of a given ionophore are largely governed by the membrane composition. The type and concentration of lipophilic ionic sites maintain electroneutrality and control the transport mechanism between neutral-carrier and ionic-carrier modes. Additionally, the dielectric constant, viscosity, and hydrophobicity of the plasticizer determine ion mobility, extraction efficiency, and potential stability within the membrane, thereby influencing the Nernstian response slope, linear dynamic range, and limit of detection (LOD). By applying thermodynamic models of membrane equilibria, the ionophore: ionic additive: plasticizer ratio can be optimized to simultaneously balance selectivity and reproducibility [11,85].
3.2.4. Stability and Reproducibility
Ionophore leaching and potential drift must be minimized to ensure long-term reliability. Increasing the ionophore’s lipophilicity (e.g., through long alkyl chain substitution) or covalent immobilization within a polymer backbone effectively suppresses diffusion into the aqueous phase. In SC-ISEs, interfacial potential stability is achieved through two major strategies: (1) introducing a mixed ionic–electronic conducting layer (e.g., conducting polymers, redox mediators, or ion-to-electron exchange resins) to mediate charge transfer, and (2) incorporating a high-capacitance layer (e.g., carbon-based nanomaterials) to buffer parasitic charge accumulation. Recently, hybrid SC-ISE architectures combining these two mechanisms have been developed to maximize interfacial capacitance, suppress water-layer formation, and significantly enhance long-term potential stability and reproducibility. For instance, TEMPO-functionalized carbon nanotubes provide both high-charge storage capacity and redox buffering, effectively stabilizing EMF signals during extended measurement periods [106,107].
3.2.5. Thermodynamics of Complexation
The thermodynamics of ion-ionophore complexation provide a quantitative foundation for understanding ISE selectivity. The equilibrium can be expressed as Equation (1):
where M denotes the metal ion and L represents the ionophore. The β value and its corresponding Gibbs free energy change (ΔG = −RT ln β) describe the driving force for complex formation. More negative ΔG values indicate more stable complexes, resulting in lower ion activity within the membrane and a thermodynamically defined electrode potential. The difference in stability constants between competing ions quantitatively determines the selectivity coefficient, Equation (2):
Hence, ion selectivity does not arise from kinetic barriers but from thermodynamic differences in complexation affinity. The β values are typically obtained through solvent extraction, spectrophotometric titration, or potentiometric analysis using model membrane systems. Notably, direct correlations between ΔGcomplex and () in calix[4]arene and calix[4]pyrrole ionophores have been experimentally validated, demonstrating that selectivity patterns observed in ISEs can be quantitatively predicted from fundamental thermodynamic parameters [108,109,110]. These thermodynamic insights provide a rigorous scientific link between molecular recognition, membrane equilibrium, and macroscopic electrode responses.
4. Application in Electrochemical Sensor Systems
Ionophore-based ion-selective electrodes have been widely incorporated into various electrochemical-sensing formats to achieve selective metal-ion determination in complex sample matrices. This chapter highlights how ISE membranes designed with custom ionophores serve as recognition-transduction interfaces in potentiometric, potentiostatic (amperometric/voltametric), impedimetric, photoelectrochemical, and Field-Effect Transistor (FET)-supported platforms.
4.1. Potentiometric Sensors
A potentiometric ISE measures the open-circuit potential relative to a reference electrode, generating an EMF that varies logarithmically with the activity of the target ion (Figure 8a) [108]. The interfacial potential formed at the sample/membrane boundary arises from ion-exchange equilibria and is governed by βmem of the ion-ionophore complex within the membrane, which serves as a key determinant of the selectivity coefficient. In polymer-based membranes, the ionophore is accompanied by a lipophilic ionic site (e.g., NaTPB) to maintain electroneutrality, thereby regulating selective ion partitioning and transport. Accordingly, the compositional ratio among the ionophore, NaTPB, and plasticizer defines whether the transport proceeds via a neutral-carrier or ionic-carrier mechanism, producing either Nernstian or near-Nernstian responses [108,109]. However, ionophore selectivity cannot be interpreted solely as a single-ligand effect. NaTPB, in particular, can repel anions and favor uptake of cations by providing negatively charged TPB− sites, thus influencing the apparent selectivity of the electrode [110]. Therefore, the observed selectivity Hg(II)-ISE should be interpreted as the combined result of the ionophore-NaTPB system, emphasizing the importance of quantitatively accounting for the ion-exchanger contribution [111].
The potential stability and reproducibility of the electrode can be improved using an SC configuration [112,113]. A Ca2+-selective SC-ISE employing nitrogen-doped mesoporous carbon as a conductive interlayer suppresses water-layer formation through its high double-layer capacitance and hydrophobicity, exhibiting a stable Nernstian slope of 26.3 ± 3.1 mV·dec−1 over a 10 µM–0.1 M range and a detection limit of 3.2 µM (Figure 8b) [114]. In addition, the incorporation of TEMPO-functionalized multiwalled carbon nanotubes forms a high-capacitance redox buffer layer at the electrode surface, effectively minimizing potential drift [106,107]. Although such configurations achieve an E0 reproducibility within ±1 mV, this corresponds to a quasi-calibration-free response rather than truly calibration-free behavior, since a deviation of 1 mV translates into roughly 4% activity error for monovalent ions and 8% for divalent ions (Figure 8c).
From the perspective of ionophore selection, crown ether, calixarene, and salen-type ligands can be rationally designed by matching cavity size and donor-site arrangement to the HSAB character and ionic radius of the target ion. For example, O-donor arrays in 18-crown-6 or calix[4]arene derivatives are well suited for Ca2+, whereas S-donor thia-crown ligands are advantageous for Pb2+. Such HSAB-guided combinations and differences in βmem values provide a quantitative basis for predicting selectivity coefficients [115].
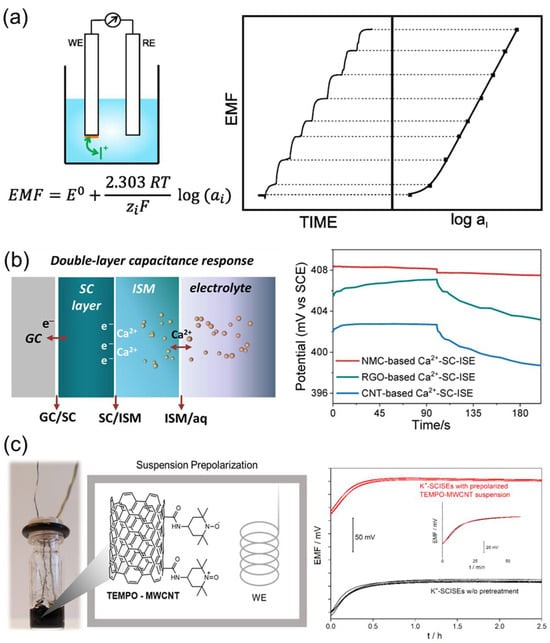
Figure 8.
(a) Principle of operation of potentiometric ISE sensor. Reprinted with permission from Ref. [108]. Copyright 2019 MDPI. (b) Example of calcium-selective SC-ISEs. Reprinted with permission from Ref. [114]. Copyright 2022 MDPI. (c) Example of calibration-free ISE sensor. Reprinted with permission from Ref. [107]. Copyright 2022 American Chemical Society.
Figure 8.
(a) Principle of operation of potentiometric ISE sensor. Reprinted with permission from Ref. [108]. Copyright 2019 MDPI. (b) Example of calcium-selective SC-ISEs. Reprinted with permission from Ref. [114]. Copyright 2022 MDPI. (c) Example of calibration-free ISE sensor. Reprinted with permission from Ref. [107]. Copyright 2022 American Chemical Society.
4.2. Potentiostatic Sensors
Current-type sensors measure the interfacial charge-transfer current generated when a fixed potential or potential waveform is applied. The diffusion-limited current can be expressed as Equation (3):
which depends on the concentration gradient near the interface, the diffusion-layer thickness, and the effective electrode area [116]. Even for metal ions that do not undergo direct electron transfer, measurable current responses can be obtained through ion-transfer electrochemistry at the interface between two immiscible electrolyte solutions (ITIES) (Figure 9a) [117].
Ionophores enhance selectivity and sensitivity through two main mechanisms. (1) Within ITIES or organic-phase membranes, the ionophore reversibly complexes with the target ion, thereby lowering the free-energy (ΔGtrans) for ion transfer across the interface. As a result, the ion-transfer peak current observed during potential sweeps becomes proportional to the analyte concentration. (2) Selective adsorption and preconcentration of the target ion are achieved when the ionophore is immobilized on the electrode surface via thiol-Au self-assembly, polymer entrapment, or nanoparticle supports. Subsequent differential pulse voltammetry or square-wave voltammetry measurements then detect changes in faradaic current or peak potential, which directly reflect the ion concentration [117,118,119]. For instance, a thiolated calix[4]arene ligand self-assembled onto a gold-nanoparticle/screen-printed carbon electrode enables the simultaneous differential pulse voltammetry detection of Pb2+ and Cu2+, exhibiting high reproducibility and recovery in real river-water samples (Figure 9b) [120]. Additionally, the Amemiya group developed a gel-based ITIES pen-type electrochemical device that provides both mechanical robustness and portability, enabling the quantitative analysis of mixed samples through ionophore-assisted ion transfer (Figure 9c) [121].
The selection of ionophores for potentiostatic sensors is guided by ΔGtrans, complex stability constant, and the match of polarity/hydrophobicity to the membrane environment. For example, crown ether derivatives are effective for hydrophilic metal cations such as K+ and Na+, whereas calixpyrrole-type ligands are better suited for less hydrophilic anions or soft cations [122]. Considering such structure-function correlations, Bakker and Amemiya proposed a predictive framework for estimating the selectivity coefficients of ITIES-based current-type ISEs directly from differences in β values [123].
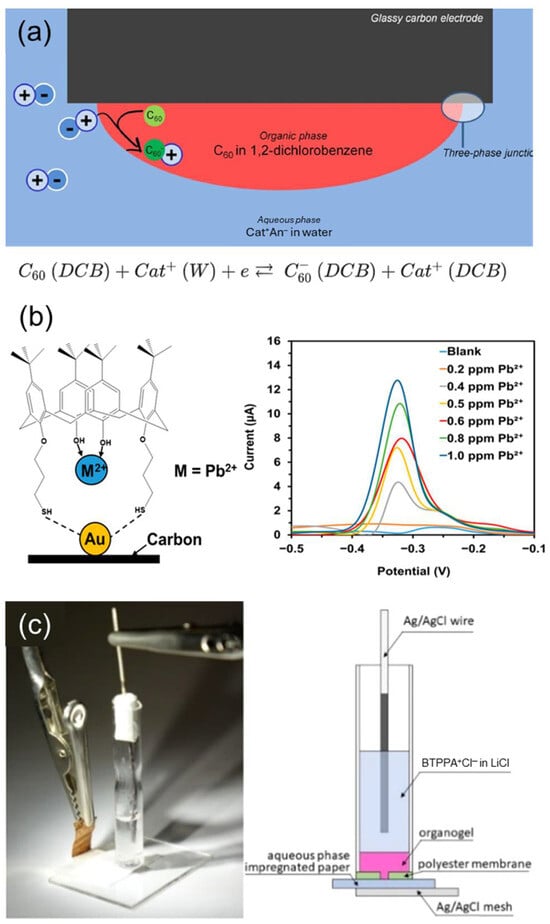
Figure 9.
(a) Principle of operation of potentiostatic ISE sensor. Reprinted with permission from Ref. [117]. Copyright 2019 Elsevier. (b) thiolated calix[4]arene introduced potentiostatic ISE for detecting Pb2+ in river-water samples. Reprinted with permission from Ref. [120]. Copyright 2021 MDPI. (c) Portable electrochemical pen ITIES voltammetry sensor. Reprinted with permission from Ref. [121]. Copyright 2020 American Chemical Society.
Figure 9.
(a) Principle of operation of potentiostatic ISE sensor. Reprinted with permission from Ref. [117]. Copyright 2019 Elsevier. (b) thiolated calix[4]arene introduced potentiostatic ISE for detecting Pb2+ in river-water samples. Reprinted with permission from Ref. [120]. Copyright 2021 MDPI. (c) Portable electrochemical pen ITIES voltammetry sensor. Reprinted with permission from Ref. [121]. Copyright 2020 American Chemical Society.
4.3. Impedimetric Sensors
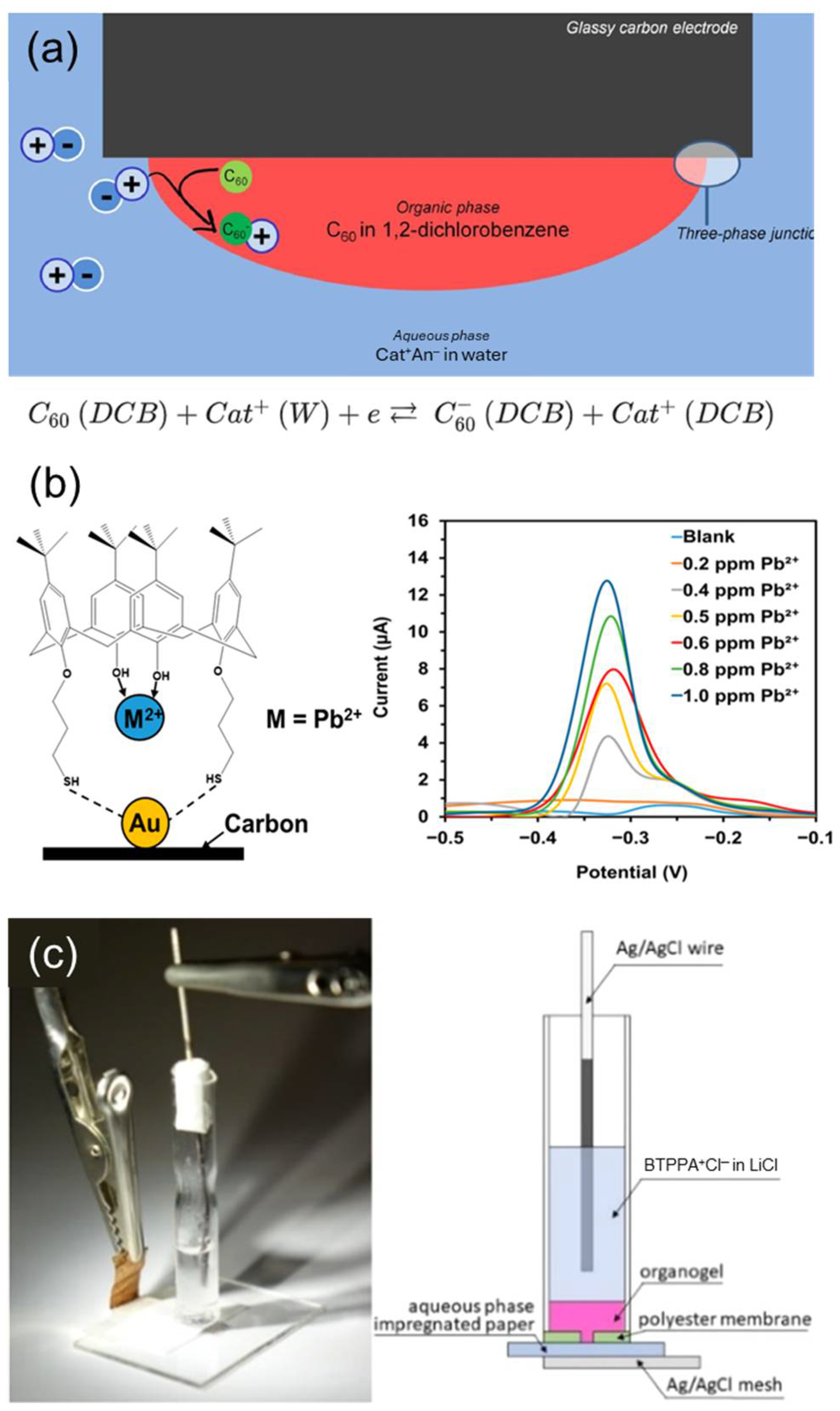
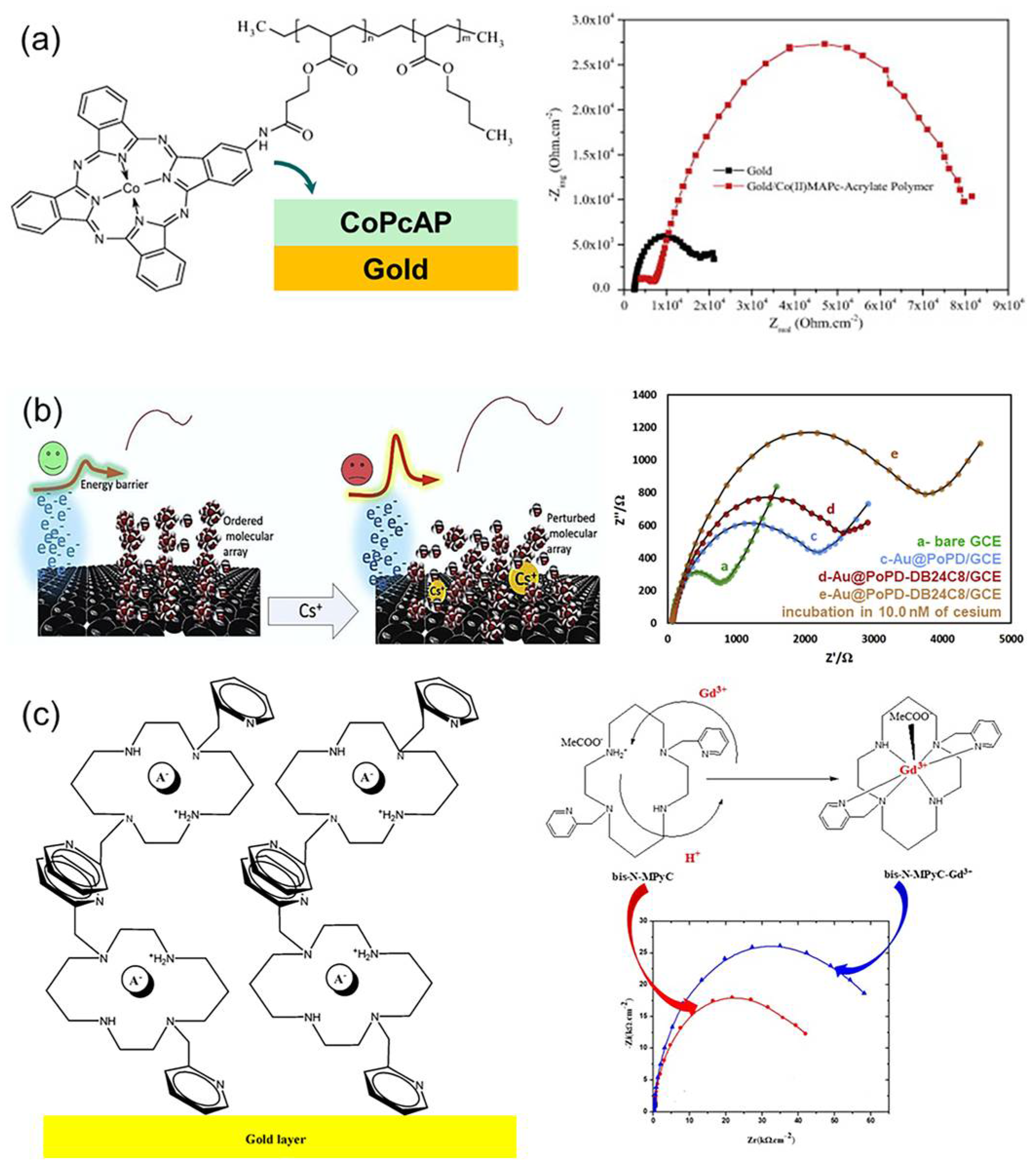
Impedimetric sensors evaluate interfacial charge-transfer and diffusion processes by applying a small-amplitude AC potential and analyzing the frequency-dependent impedance Z(ω) [124]. The resulting spectra are typically interpreted using a modified Randles equivalent circuit, which consists of a solution resistance (Rs), a double-layer capacitance (Cdl), a charge-transfer resistance (Rct), and a Warburg diffusion element (Figure 10a) [125]. However, in practical measurements, only the high-frequency semicircle corresponding to the bulk response is often resolved, while the low-frequency semicircle associated with the interfacial processes is not clearly separated. Therefore, interpreting interfacial charge transfer based solely on Rct values can be misleading; the corresponding Cdl values must also be reported to ensure accurate analysis. In particular, since Rct inherently varies with ion concentration, the ionophore merely modulates the Rct value at a given concentration, rather than altering its fundamental dependence [126].
When an ionophore is incorporated, complexation of the target ion at the interface modifies the local Cdl and the effective dielectric constant of the membrane, thereby tuning the overall impedance response. For example, a DB24C8 (dibenzo-24-crown-8) functionalized poly(o-phenylenediamine) electrode induces measurable changes in both ΔRct and ΔCdl upon binding of Cs+, achieving a detection limit of 0.3 μM as confirmed by electrochemical impedance spectroscopy (EIS) and scanning force microscopy analyses (Figure 10b) [127]. In another case, a 2-methylpyridine-functionalized cyclam ligand immobilized on a gold electrode exhibited an RC-series circuit-type response upon complexation with Gd3+, yielding a detection limit of 35 pM even in synthetic urine samples (Figure 10c) [128]. Overall, impedimetric sensors enable ultrasensitive metal-ion detection by precisely controlling interfacial electron-transfer kinetics and dielectric properties through the rational design of ionophore-membrane-electrode combinations.
Figure 10.
(a) Principle of operation of Impedimetric ISE sensor. Reprinted with permission from Ref. [125]. Copyright 2015 Elsevier. (b) DB24C8-based screen-printed carbon electrode for cesium ion detection. Reprinted with permission from Ref. [127]. Copyright 2020 Elsevier. (c) 2-methylpyridine-substituted cyclam-based gold electrode for Gadolinium ion detection. Reprinted with permission from Ref. [128]. Copyright 2021 MDPI.
Figure 10.
(a) Principle of operation of Impedimetric ISE sensor. Reprinted with permission from Ref. [125]. Copyright 2015 Elsevier. (b) DB24C8-based screen-printed carbon electrode for cesium ion detection. Reprinted with permission from Ref. [127]. Copyright 2020 Elsevier. (c) 2-methylpyridine-substituted cyclam-based gold electrode for Gadolinium ion detection. Reprinted with permission from Ref. [128]. Copyright 2021 MDPI.
4.4. Photoelectrochemical Sensors
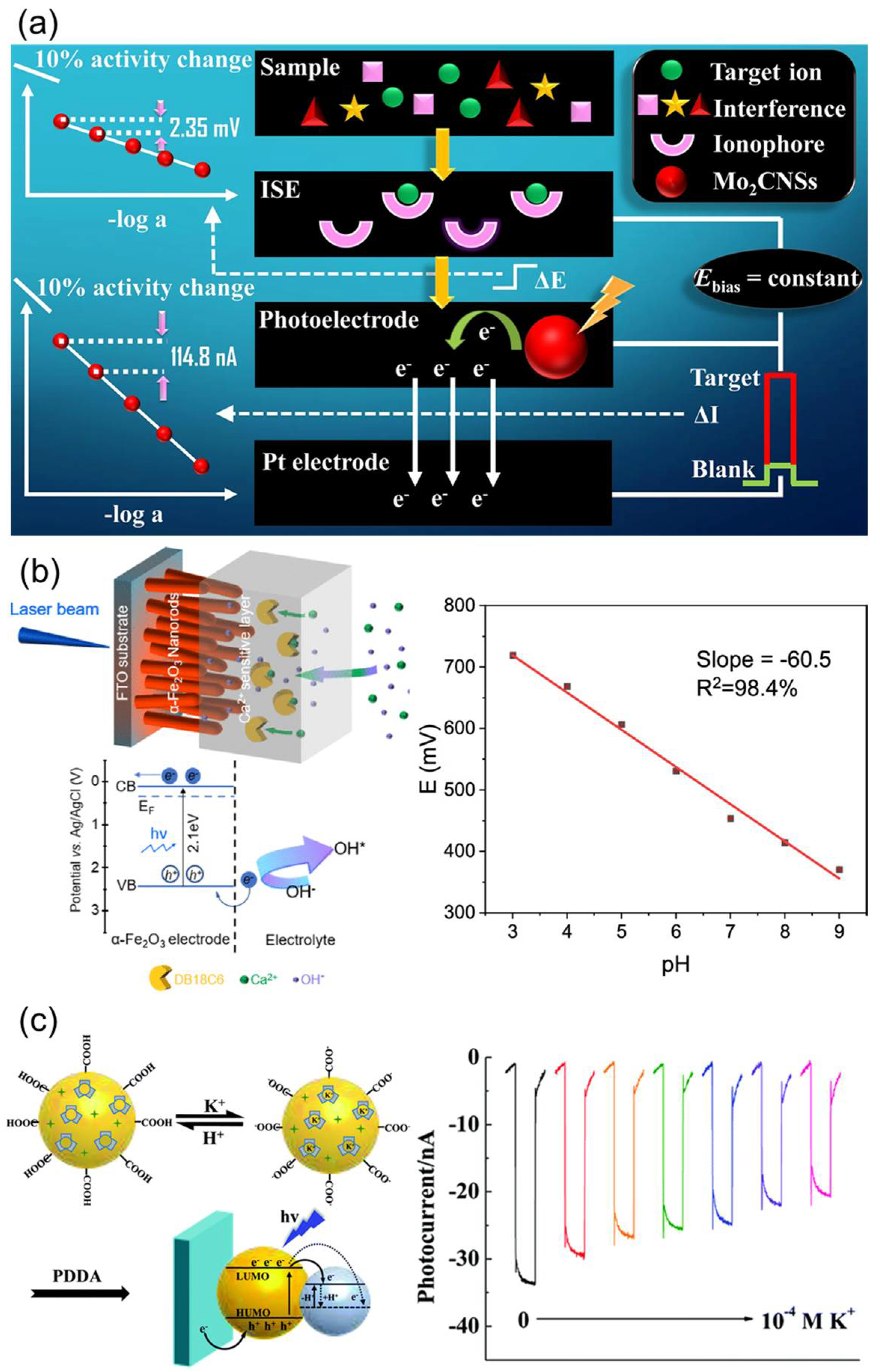
Photoelectrochemical (PEC) sensors detect changes in ionic activity by converting the motion of photogenerated electron-hole pairs into measurable current or potential signals. Unlike conventional potential-based ISEs, PEC sensors exploit variations in charge redistribution and band bending that occur at the interface between a semiconductor photoelectrode and an ISM (Figure 11a) [129]. For instance, when a Ca2+-selective polyvinyl chloride (PVC)-based ISM is coated onto semiconductor photoelectrodes such as α-Fe2O3 (hematite) or BiVO4, the binding of metal ions to ionophores induces the reorganization of the interfacial electric double layer. This modifies the band structure and shifts the flat-band potential, thereby altering the charge recombination rate and resulting in a photocurrent response that depends on Ca2+ activity (Figure 11b) [130]. Such systems typically achieve detection limits in the order of 10−6 M and exhibit excellent selectivity. Similarly, polymer-dot architectures embedded with a K+-selective ionophore (valinomycin) show photocurrent suppression due to the deprotonation of –COOH groups that accompany K+ complexation. This modulates hole density and the dielectric environment within the film, producing a signal that correlates with K+ concentrations (Figure 11c) [131]. These polymer-dot–ionophore composite membranes exhibit fast response times and high selectivity, making them promising candidates for non-contact or wearable ion-sensing applications [132].
Within PEC–ISE systems, the ionophore operates not only as a chemical recognition element but also as a mediator that modulates interfacial electric fields and assists photo-induced charge transfer. Optimizing membrane parameters—such as plasticizer polarity, ionophore concentration, and membrane thickness—helps stabilize the interfacial photo-potential and improves coupling with the underlying photoelectrode. Recent studies have introduced hybrid PEC sensors that employ nanostructured photoelectrodes, including Au/ZnS/ZnO heterostructures. In these platforms, synergistic band alignment among ZnO nanorods, ZnS microspheres, and Au nanoparticles enhances photocurrent generation and accelerates charge-transfer kinetics under illumination [133]. As a result, these systems enable sensitive and selective Cu2+ detection, with a linear range of 0.1–10 µM, a detection limit down to 0.01 µM, fast photoresponse, and strong operational stability. Due to their low power consumption, optical tunability, and compatibility with aqueous media, PEC–ISE hybrid architectures are emerging as promising candidates for next-generation environmental and biomedical ion-monitoring technologies.
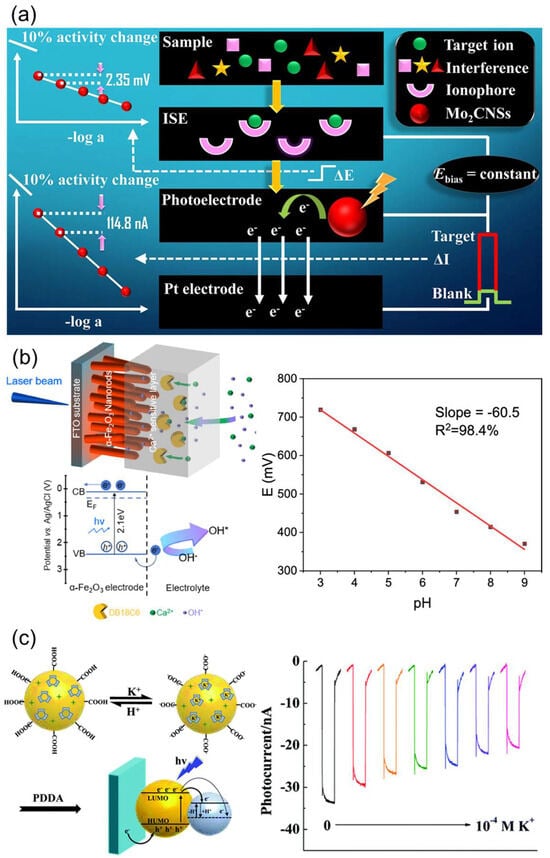
Figure 11.
(a) Principle of operation of photoelectrochemical ISE sensor. Reprinted with permission from Ref. [129]. Copyright 2022 Springer Nature. (b) Ultrathin ionophore-containing PVC-based ion-selective membrane for Ca2+ detection. Reprinted with permission from Ref. [130]. Copyright 2022 American Chemical Society. (c) Layer-by-layer films of K+-selective polymer-dot ISE systems. Reprinted with permission from Ref. [131]. Copyright 2021 Royal Society of Chemistry.
Figure 11.
(a) Principle of operation of photoelectrochemical ISE sensor. Reprinted with permission from Ref. [129]. Copyright 2022 Springer Nature. (b) Ultrathin ionophore-containing PVC-based ion-selective membrane for Ca2+ detection. Reprinted with permission from Ref. [130]. Copyright 2022 American Chemical Society. (c) Layer-by-layer films of K+-selective polymer-dot ISE systems. Reprinted with permission from Ref. [131]. Copyright 2021 Royal Society of Chemistry.
4.5. FET-Based Sensors
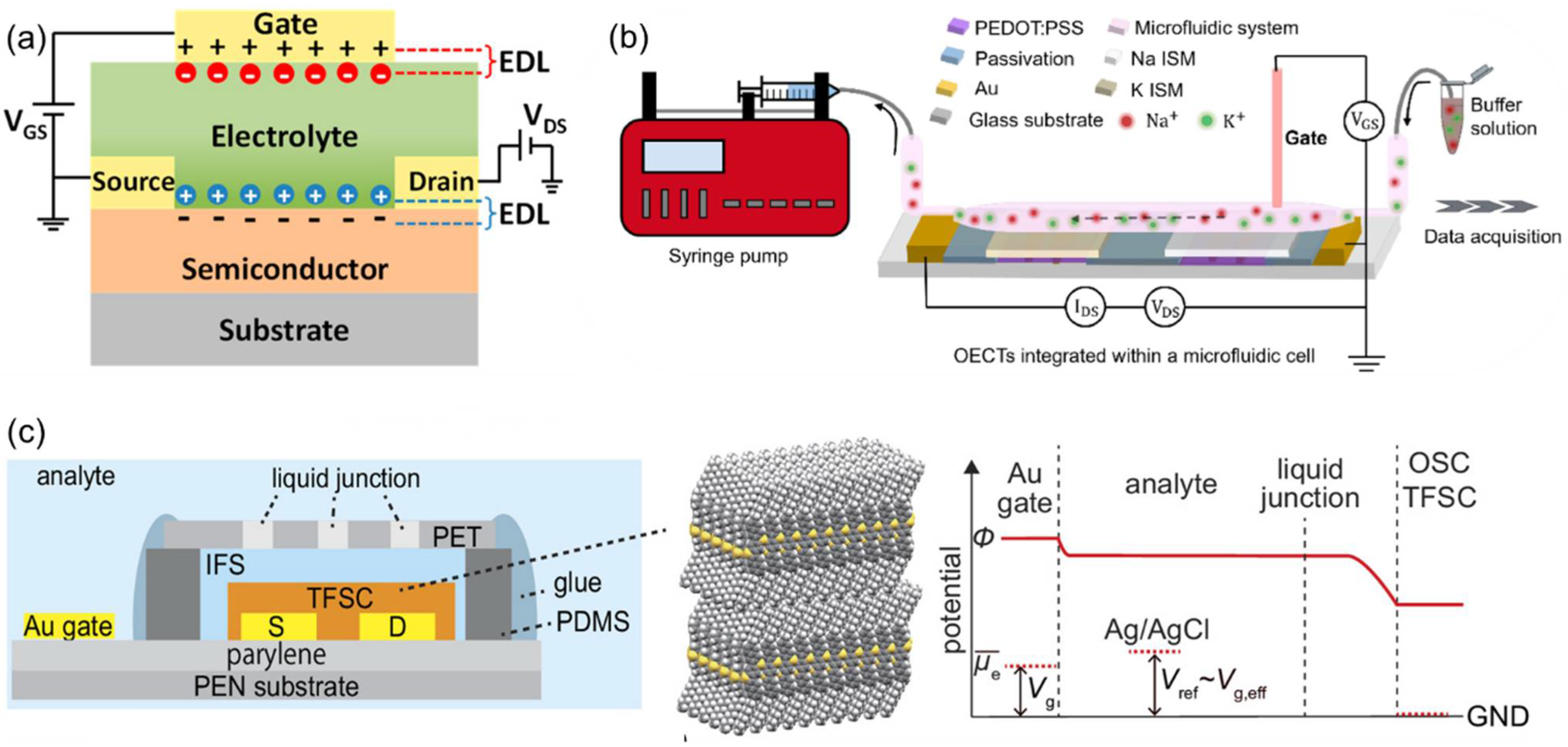
P. Bergveld and R.P. Buck laid the groundwork for field-effect potentiometric sensors, combining ion-selective membranes with semiconductor transducers for direct electrochemical-to-electronic conversion. Their work demonstrated that surface-potential modulation at the electrolyte–gate interface enables quantitative ion detection in FET-based sensors [134,135]. Field-effect transistor-based ion sensors quantify ionic activity by converting potential variations at the electrolyte-gate interface into changes in channel conductance or threshold voltage [136]. In this configuration, the electric double layer functions as the gate dielectric, amplifying the signal through capacitive coupling (Figure 12a) [137]. Representative device architectures include ion-sensitive FETs (ISFETs), extended-gate FETs (EGFETs), organic electrochemical transistors (OECTs), and electrolyte double-layer transistors (EDLTs). Within these devices, an ISM formed on the gate electrode acts as the sensing layer, where the ionophore reversibly complexes with the target ion. This interaction modifies the internal membrane potential (ψ0) and the electric double layer charge distribution, adjusting the dielectric constant and the ion-electron equilibrium of the system. Consequently, these changes appear as shifts in the ISD-VG transfer curves of the transistor.
Ionophore selectivity is determined by HSAB compatibility, cavity size, donor-atom configuration, and ionophore charge state (neutral or ionic). Furthermore, optimization of the membrane thickness, plasticizer dielectric constant, hydrophobicity, and ionic-additive concentration enables the precise control of in-membrane diffusion equilibria and interfacial potential stability. For instance, PEDOT:PSS-based OECT arrays coated with ISM containing valinomycin (K+) or ETH129 (Na+) ionophores, when integrated with a microfluidic cell, allow real-time, multiplexed ion detection (Figure 12b) [136]. By finely controlling ISM thickness and solvent compatibility, these systems minimize device-to-device response variations and ensure linear signal calibration. In another example, an organic single-crystal EDLT equipped with a valinomycin-based K+-selective ISM was shown to operate differentially with a quasi-reference transistor, achieving potential drift below 0.5 mV·h−1 and effectively replacing traditional Ag/AgCl reference electrodes (Figure 12c) [138].
Ionophore selection must consider not only its binding affinity but also the electrochemical stability of the gate interface, band alignment between the membrane and transistor channel, and ion-to-electron transfer kinetics. For example, crown ethers, which exhibit rapid association–dissociation kinetics, are ideal for ISFET/EGFET-type sensors, whereas salen and calixarene ligands, characterized by high binding constants and low leaching rates, are advantageous for precision OECT/EDLT-type sensors [139,140]. Recently, redox-active ionophores and polymer-tethered ionophores have been developed to simultaneously enhance membrane leaching resistance and electrical capacitance. Overall, FET-based ion sensors achieve faster response times, lower LODs, and higher integration capability than potential-based ISEs, and when combined with microfluidic or wearable technologies, they represent a promising platform for next-generation real-time ion-monitoring systems.
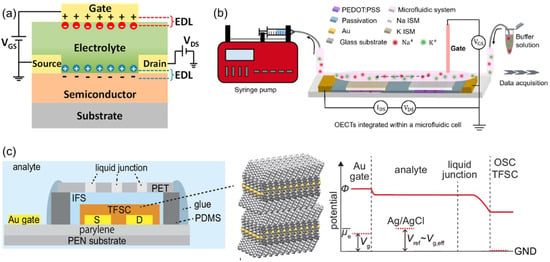
Figure 12.
(a) Principle of operation of electrolyte-double-layer transistor-based sensor. Reprinted with permission from Ref. [137]. Copyright 2019 MDPI. (b) OECT/ISM microarray for simultaneous Na+/K+ sensing. Reprinted with permission from Ref. [136]. Copyright 2024 Elsevier. (c) EDLT with a quasi-reference transistor (qRE) for K+ sensing without bulky Ag/AgCl. Reprinted with permission from Ref. [138]. Copyright 2024 PNAS.
Figure 12.
(a) Principle of operation of electrolyte-double-layer transistor-based sensor. Reprinted with permission from Ref. [137]. Copyright 2019 MDPI. (b) OECT/ISM microarray for simultaneous Na+/K+ sensing. Reprinted with permission from Ref. [136]. Copyright 2024 Elsevier. (c) EDLT with a quasi-reference transistor (qRE) for K+ sensing without bulky Ag/AgCl. Reprinted with permission from Ref. [138]. Copyright 2024 PNAS.
Using the design principles, material selection strategies, and measurement criteria discussed above, Table 1 provides a comparative summary of the performance achieved by ionophore-based electrochemical sensors for various metal-ion analytes. The table highlights the structural characteristics of ionophores, including crown ethers, calixarenes, salen/salophene-type chelates, and molecularly imprinted polymers, and illustrates their correlations with specific electrode compositions and measurement modes. It further demonstrates how the arrangement of donor atoms and coordination geometry influences both selectivity and detection limits. In addition, the effects of anionic additives such as NaTPB and potassium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate, as well as plasticizer polarity, on the selectivity coefficients are emphasized, confirming that overall selectivity is not solely due to the ionophore itself but from synergistic interactions within the entire membrane composition. From a signal-transduction perspective, potentiometric sensors offer high long-term stability with Nernstian responses, while voltammetric and impedimetric sensors provide broader dynamic ranges and tunable response kinetics. Meanwhile, FET-based ion sensors, which integrate high-capacitance channels with ionophore-containing membranes, achieve quasi-calibration-free operation with potential variations maintained within ±1 mV. Table 1 compiles representative data from recent studies, showing how variations in ionophore structure, membrane formulation, and signal-transduction mechanisms correlate with key analytical parameters such as selectivity, detection limit, and response stability. This comparison provides a factual basis for selecting suitable ionophore-membrane combinations tailored to specific target ions and sensing platforms.

Table 1.
Comparison of Operation and Performance of Ionopore-based Electrochemical Sensor.
Table 1.
Comparison of Operation and Performance of Ionopore-based Electrochemical Sensor.
| Ionophore Type | ISE Composition | Target Ion | Operating Method | LOD | Slop/Linear Range | Ref. |
|---|---|---|---|---|---|---|
| Bis-thiophenalpropanediamine | Ionophore/DBP/NaTPB/PVC | Copper (Cu2+) | Potentiometric | 2.0 × 10−8 M | 29.3 ± 0.7 mV/decade 1.0 × 10−1 M–6.0 × 10−8 M | [141] |
| Pentathia-15-crown-5 | Ionophore/DBP/NaTPB/PVC | Mercury (Hg2+) | Potentiometric | 2.51 × 10−5 M | 32.1 mV/decade 2.51 × 10−5 M–1.0 × 10−1 M | [111] |
| 1,3-Alternate thiacalix[4]crown | Ionophore/o-NPOE/NaTPB/PVC | Mercury (Hg2+) | Potentiometric | 7.76 × 10−7 M | 30.39 mV/decade 1.0 × 10−1 M–5.0 × 10−7 M | [142] |
| tert-Butylcalix[4]arene-tetrakis(N,N-dimethylthioacetamide) | Ionophore/NaTPB/PVC/o-NPOE | Lead (Pb2+) | Potentiometric | 4 × 10−10 M | 29.0 ± 0.2 mV/decade 10−10 M–10−5 M | [143] |
| Arsenite ion imprinted polymer | Ionophore/BEHS 1/PVC/TOAB 2 | Arsenite (AsO2−) | Potentiometric | 1 × 10−12 M | −18 mV/decade 1 × 10−11 M–1 × 10−2 M | [144] |
| Glyoxal bis(2-hydroxianil) | Ionophore/DBP/PVC | Chromium (Cr3+) | Potentiometric | 6.3 × 10−7 M | 19.8 ± 0.5 mV/decade 3.0 × 10−6–1.0 × 10−2 M | [145] |
| 1-nitroso-2-naphtol | Ionophore/nitrobenzene/NaTPB | Neodymium (Nd3+) | Potentiometric | 6.5 × 10−7 M | 19.7 ± 0.4 mV/decade 1.0 × 10−6–1.0 × 10−2 M | [146] |
| S-methyl N-(methylcarbamoyloxy) thioacetimidate | Ionophore/chloronaphthalene/NaTPB/PVC | Iron (Fe3+) | Potentiometric | 5.0 × 10−6 M | 20 mV/decade 6.3 × 10−6–1.0 × 10−1 M | [147] |
| Calix[4]arene derivatives containing four imine groups | Ionophore/DOS/PVC | Silver (Ag+) | Potentiometric | 1.0 × 10−6 M | 57.90 mV/decade 1.0 × 10−1–1.0 × 10−5 M | [148] |
| Polyaniline-zirconium(IV)iodate | Ionophore/PVC | Lead (Pb2+) | Potentiometric | 3.31 × 10−9 M | 29.28 mV/decade 1.0 × 10−12–1.0 × 10−1 M | [149] |
| Tetrathia-12-crown-4 | Ionophore/PVC/NPOE/KTCPB 3 | Cadmium (Cd2+) | Potentiometric | 1.0 × 10−7 M | 29.0 ± 1.0 mV decade−1 4 × 10−7–1 × 10−1 M | [150] |
| Bis[(benzo-15-crown-5)-4-ylmethyl] | Ionophore/PVC/DOS | Potassium (K+) | Impedimetric | - | 2.7 mM–18.7 mM | [151] |
| Bis-N-(dimethylpyridine)-armed cyclam | Ionophore | Gadolinium (Gd3+) | Impedimetric | 35 pM | 7.3 (ΔRf/Rf0)/decade 1 × 10−10–1 × 10−5 M | [128] |
| 2-[2,2-bis[[2-(dicyclohexylamino)-2-oxoethoxy]methyl]butoxy]-N,N-dicyclohexylacetamide | Ionophore/NaTPB/PVC | Lithium (Li+) | Voltammetric | 10−3 M | 59.9 mV/log(aLi) 1 × 10−4 5 M–1 × 10−3 M. | [152] |
| Valinomycin | Ionophore/NaTPB/4-hydroxy-TEMPO 4/PVC/DOS | Potassium (K+) | Voltammetric | 7.9 × 10−6 M | 54.8 mV 0.5 mM–1 mM | [153] |
| N,Nʹ-(disulfanediylbis(2,1-phenylene))bis(1-(1H-indol-3-yl)meth animine) | Ionophore/graphite powder/paraffin oil | Copper (Cu2+) | Voltammetric | 9.0 × 10−7 M | 1.99–23.9 μM | [154] |
| 2-(Octadecyloxymethyl)pyridine | Ionophore/NaTPB/PVC/DOS | Silver (Ag+) | Voltammetric | <500 nM | 5–100 nM | [155] |
| 4-tert-butylcalix[4]arene-tetrakis(N,N-dimethylthioacetamide) | Ionophore/NHAP 5/Nafion | Lead (Pb2+) | Voltammetric | 1.0 nM | 5.0 nM–0.8 μM | [156] |
| Valinomycin | Ionophore/NaTPB/PVC/o-NPOE | Potassium (K+) | Amperometric | 1.0 × 10−6 M | (30 ± 2) nA·mL(−17/12)·h3/4 1.0 × 10−6–1.0 × 10−4 M | [157] |
| N, N, N′, N′-tetracyclohexyl-3-oxapentanediamide | Ionophore/NaTPB/PVC/o-NPOE | Calcium (Ca2+) | Amperometric | 10−7 M | 22.2 ± 1.4 mM 1.0 × 10−6–1.0 × 10−2 M | [158] |
1 Bis(2-ethyl hexyl) sebacate; 2 Tetra octyl ammonium bromide; 3 potassium tetrakis(4-chlorophenyl) borate; 4 4-hydroxy-2,2,6,6- tetramethylpiperidine 1-oxyl; 5 hydroxyapatite.
5. Recent Research Trends
Recent advances in ionophore-based electrochemical sensors have rapidly evolved in four major directions: hybrid device integration, low-cost and portable sensor development, multi-analyte detection, and wearable applications. These developments are driven by the need for higher selectivity and sensitivity, as well as the demand for improved long-term stability, reproducibility, and durability in aqueous environments.
Recently, hybrid-sensing platforms that combine traditional ISEs with optical or digital signal-transduction modules have attracted significant attention. For example, Phoonsawat et al. developed a smartphone-based K+/Cl− dual-analyte sensor integrating an activity-to-concentration conversion algorithm and an empirical ionic-strength correction function based on the Debye-Hückel model. This configuration enabled the simultaneous quantification of K+ (0.1–100 mM) and Cl− (LOD 0.16 mM) in sweat and serum samples while minimizing matrix-related deviations (Figure 13a) [159]. In addition, the redox buffering provided by the polymer backbone ensures prolonged stability of the electrode potential [160].
Portable and disposable ISE platforms have also been actively developed to meet the requirements for on-site analysis and cost-effective operation [161]. A representative example is a thermoplastic microfluidic chip integrating a solid-contact K+-selective electrode and an organic light-emitting diode-based readout system for field-portable potentiometric analysis (Figure 13b) [162]. The decoration of laser-induced graphene/TiO2 onto MXene/PVDF nanofiber composite significantly reduced charge-transfer resistance and maintained potential stability under varying humidity conditions (~ 80–85% using a humidifier) [163]. Furthermore, the emergence of biodegradable conductive polymer electrodes has opened new opportunities for environmentally sustainable, single-use electrochemical sensors [164].
The technology for multiplexed ISE arrays capable of detecting multiple ions, such as Na+, K+, and Ca2+, has also advanced rapidly [165]. A smartphone-based potentiometric system coupled with these arrays achieved correlation coefficients above 0.99 compared to reference inductively coupled plasma–optical emission spectrometry measurements (Figure 13c). Moreover, machine-learning-based pattern-recognition algorithms have been employed to quantify cross-sensitivity among ionophore membranes and construct ion-selectivity maps [166]. These artificial intelligence-driven approaches enhance the accuracy of simultaneous multi-ion detection and pave the way for quasi-calibration-free sensor design.
Wearable ISEs are being developed using composite materials such as PEDOT: PSS, MXene, graphene, and conductive hydrogels, which simultaneously improve mechanical flexibility and aqueous stability [167]. For instance, a Bluetooth low energy-enabled K+ monitoring wristband achieved potential drift stability within ±1 mV even under humid and sweat-rich environments (Figure 13d). In addition, leaching of ionophores from ISM is a well-known issue that can cause cytotoxicity in biomedical or wearable applications. Currently, plasticizer-free poly(decyl methacrylate) (PDMA) membranes incorporating covalently attachable ionophores with pyridine or tertiary amino groups were suggested, showing a significant reduction in leaching [168]. This strategy provides a solution to mitigate the loss of ionophores while simultaneously improving the stability and lifetime of the sensor.
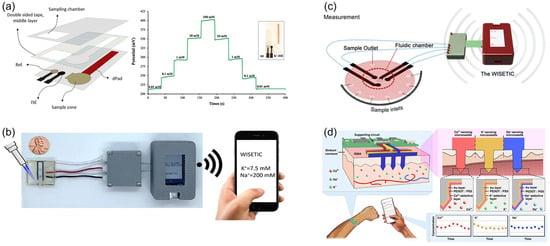
Figure 13.
Recent research trends in ISE-based electrochemical sensors: (a) hybrid platform. Reprinted with permission from Ref. [159]. Copyright 2022 Elsevier. (b) portable and disposable sensors. Reprinted with permission from Ref. [162]. Copyright 2022 Springer Nature. (c) multiplexed detection. Reprinted with permission from Ref. [165]. Copyright 2023 Springer Nature. (d) wearable sensors. Reprinted with permission from Ref. [167]. Copyright 2023 Nature.
Figure 13.
Recent research trends in ISE-based electrochemical sensors: (a) hybrid platform. Reprinted with permission from Ref. [159]. Copyright 2022 Elsevier. (b) portable and disposable sensors. Reprinted with permission from Ref. [162]. Copyright 2022 Springer Nature. (c) multiplexed detection. Reprinted with permission from Ref. [165]. Copyright 2023 Springer Nature. (d) wearable sensors. Reprinted with permission from Ref. [167]. Copyright 2023 Nature.
6. Challenges and Future Perspectives
Despite these advances, several key challenges emerge. First, regarding stability, ionophore leaching and the degradation of solid-contact layers induced by oxidation or humidity represent major sources of long-term potential drift. Therefore, high-capacitance/low-resistance contacts utilizing conducting polymers, carbon/metal-oxide composites, or molecular redox couples are strongly recommended. In addition, standardized validation methods, such as the water-layer test, EIS, and chronopotentiometry, are also advised [91,169]. Second, matrix effects, including those arising from ionic strength, pH, protein adsorption, and surfactants, distort selectivity patterns. Thus, it is essential that the co-optimization of ionophore and membrane composition be coupled with mechanistic analyses of interferents (e.g., the unusual behavior observed for ammonium) [170]. Third, standardization is needed to ensure reproducibility and cross-study comparability. Consensus checklists should comprehensively address selectivity-coefficient determination, water-layer inspection, and the detailed configuration and sequence of both reference electrodes and measurement protocols [119].
Looking ahead, three strategic directions are pivotal. First, implementing reference-electrode-free differential measurements using quasi-reference transistors is recommended. Second, the further development of modular integration between OECT/ISFET transducers and both microfluidics and wireless telemetry should be pursued. Third, the introduction of on-chip calibration processes that incorporate kinetic models and compensate for variations in temperature and ionic strength is essential. Concurrently, integration with low-cost IoT technologies is expected to drive progress toward large-scale applications in population and environmental monitoring.
Funding
This work was supported by the Gachon University Research Fund of 2025 (GCU-202503300001).
Data Availability Statement
No data was generated in this work.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| Abbreviation | Full form |
| COF | covalent organic framework |
| DBP | dibutyl phthalate |
| EDLT | electric double-layer transistor |
| EGFET | extended-gate field-effect transistor |
| EIS | electrochemical impedance spectroscopy |
| FET | field-effect transistor |
| HOMO | highest occupied molecular orbital |
| HSAB | hard and soft acids and bases |
| ISE | ion-selective electrode |
| ISFET | ion-sensitive field-effect transistor |
| ISM | ion-selective membrane |
| ITIES | immiscible electrolyte solutions |
| LOD | limit of detection |
| LUMO | lowest unoccupied molecular orbital |
| MXene | 2D transition metal carbide/nitride |
| NaTPB | sodium tetraphenylborate |
| OECT | organic electrochemical transistor |
| o-NPOE | o-nitrophenyl octyl ether |
| PEC | photoelectrochemical |
| PEDOT:PSS | poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) |
| PVC | polyvinyl chloride |
| SC-ICE | solid-contact ion-selective electrode |
| TEMPO | (2,2,6,6-tetramethylpiperidin-1-yl)oxyl |
Nomenclature
| Symbol | Description | Units |
| selectivity coefficient | ||
| ΔG | Gibbs free energy change | J mol−1 |
| β | stability constant | |
| R | ideal gas constant | J mol−1 K−1 |
| T | absolute temperature | K |
| I | controlled current | A |
| n | electron transfer number | |
| A | surface area of the electrode | cm2 |
| D | analyte diffusion coefficient diffusion | cm2 s−1 |
| F | Faraday constant | C mol−1 |
| Cbulk | concentration of bulk solution | mol cm−3 |
| Cx=0 | concentration at the surface of electrode | mol cm−3 |
| δ | thickness of the diffusion layer | cm |
| Rs | solution resistance | Ω |
| Rct | charge-transfer resistance | Ω |
| ISD | drain current | A |
| VG | gate voltage | V |
| Nernstian slope | for Nernstian response | mV·dec−1 |
References
- Malik, L.A.; Bashir, A.; Qureashi, A.; Pandith, A.H. Detection and removal of heavy metal ions: A review. Environ. Chem. Lett. 2019, 17, 1495–1521. [Google Scholar] [CrossRef]
- Aragay, G.; Pons, J.; Merkoçi, A. Recent Trends in Macro-, Micro-, and Nanomaterial-Based Tools and Strategies for Heavy-Metal Detection. Chem. Rev. 2011, 111, 3433–3458. [Google Scholar] [CrossRef]
- Abdelmonem, B.H.; Kamal, L.T.; Elbaz, R.M.; Khalifa, M.R.; Abdelnaser, A. From contamination to detection: The growing threat of heavy metals. Heliyon 2025, 11, e41713. [Google Scholar] [CrossRef]
- Ciopec, M.; Pascu, B.; Negrea, P. Microbial Electrochemical Technologies; Wiley: Hoboken, NJ, USA, 2023; pp. 201–228. [Google Scholar] [CrossRef]
- Sulthana, S.F.; Iqbal, U.M.; Suseela, S.B.; Anbazhagan, R.; Chinthaginjala, R.; Chitathuru, D.; Ahmad, I.; Kim, T.-h. Electrochemical Sensors for Heavy Metal Ion Detection in Aqueous Medium: A Systematic Review. ACS Omega 2024, 9, 25493–25512. [Google Scholar] [CrossRef]
- Troudt, B.K.; Rousseau, C.R.; Dong, X.I.N.; Anderson, E.L.; Bühlmann, P. Recent progress in the development of improved reference electrodes for electrochemistry. Anal. Sci. 2022, 38, 71–83. [Google Scholar] [CrossRef] [PubMed]
- de la Asunción-Nadal, V.; Crespo, G.A.; Cuartero, M. Light-induced Delivery of Charged Species using Ion-selective Core–Shell Nanoparticles. Angew. Chem. Int. Ed. 2024, 136, e202403756. [Google Scholar] [CrossRef]
- Pressman, B.C.; Painter, G.; Fahim, M. Inorganic Chemistry in Biology and Medicine; American Chemical Society: Washington, DC, USA, 1980; Volume 140, pp. 3–22. [Google Scholar] [CrossRef]
- Guo, C.; Cui, E.; Xu, X.; Yang, D. Ionophore-based nanospheres enable selective and sensitive fluorescence detection of copper ions. Talanta 2025, 281, 126855. [Google Scholar] [CrossRef]
- Iurgenson, N.; Vladimirova, N.; Polukeev, V.; Mikhelson, K.; Kirsanov, D. Designing new sulfate ionophores for potentiometric membrane sensors: Selectivity assessment and practical application. Sens. Actuators B Chem. 2025, 422, 136663. [Google Scholar] [CrossRef]
- Bühlmann, P.; Pretsch, E.; Bakker, E. Carrier-Based Ion-Selective Electrodes and Bulk Optodes. 2. Ionophores for Potentiometric and Optical Sensors. Chem. Rev. 1998, 98, 1593–1688. [Google Scholar] [CrossRef]
- Mao, C.; Robinson, K.J.; Yuan, D.; Bakker, E. Ion–ionophore interactions in polymeric membranes studied by thin layer voltammetry. Sens. Actuators B Chem. 2022, 358, 131428. [Google Scholar] [CrossRef]
- Johnson, R.D.; Bachas, L.G. Ionophore-based ion-selective potentiometric and optical sensors. Anal. Bioanal. Chem. 2003, 376, 328–341. [Google Scholar] [CrossRef]
- Xiang, Z.; Wang, H.; Zhao, P.; Fa, X.; Wan, J.; Wang, Y.; Xu, C.; Yao, S.; Zhao, W.; Zhang, H.; et al. Hard Magnetic Graphene Nanocomposite for Multimodal, Reconfigurable Soft Electronics. Adv. Mater. 2024, 36, 2308575. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.N.; Thomas, D.W. T Lymphocyte Integrated Endoplasmic Reticulum Ca2+ Store Signaling Functions Are Linked to Sarco/Endoplasmic Reticulum Ca2+-ATPase Isoform-Specific Levels of Regulation. Int. J. Mol. Sci. 2025, 26, 4147. [Google Scholar] [CrossRef] [PubMed]
- Migdalski, J.; Lewenstam, A. Electrically Enhanced Sensitivity (EES) of Ion-Selective Membrane Electrodes and Membrane-Based Ion Sensors. Membranes 2022, 12, 763. [Google Scholar] [CrossRef]
- Soda, Y.; Gao, W.; Bosset, J.; Bakker, E. Emulsion Doping of Ionophores and Ion-Exchangers into Ion-Selective Electrode Membranes. Anal. Chem. 2020, 92, 14319–14324. [Google Scholar] [CrossRef]
- Schaller, U.; Bakker, E.; Pretsch, E. Carrier mechanism of acidic ionophores in solvent polymeric membrane ion-selective electrodes. Anal. Chem. 1995, 67, 3123–3132. [Google Scholar] [CrossRef]
- Xue, R.; Liu, Y.-S.; Huang, S.-L.; Yang, G.-Y. Recent Progress of Covalent Organic Frameworks Applied in Electrochemical Sensors. ACS Sens. 2023, 8, 2124–2148. [Google Scholar] [CrossRef]
- Zhang, W.; Li, X.; Ding, X.; Hua, K.; Sun, A.; Hu, X.; Nie, Z.; Zhang, Y.; Wang, J.; Li, R.; et al. Progress and opportunities for metal–organic framework composites in electrochemical sensors. RSC Adv. 2023, 13, 10800–10817. [Google Scholar] [CrossRef]
- Casanova, A.; Iniesta, J.; Gomis-Berenguer, A. Recent progress in the development of porous carbon-based electrodes for sensing applications. Analyst 2022, 147, 767–783. [Google Scholar] [CrossRef] [PubMed]
- Bondar, A.V.; Keresten, V.M.; Mikhelson, K.N. Ionophore-Based Ion-Selective Electrodes in Non-Zero Current Modes: Mechanistic Studies and the Possibilities of the Analytical Application. J. Anal. Chem. 2022, 77, 145–154. [Google Scholar] [CrossRef]
- Bahathiq, A.O.S.; Babalghith, A.O.; Amin, A.S.; Askar, A.M. Fabrication of a novel palladium membrane sensor for its determination in environmental and biological samples. Environ. Sci. Adv. 2024, 3, 776–788. [Google Scholar] [CrossRef]
- Li, X.; Zhang, C.; Cai, S.; Lei, X.; Altoe, V.; Hong, F.; Urban, J.J.; Ciston, J.; Chan, E.M.; Liu, Y. Facile transformation of imine covalent organic frameworks into ultrastable crystalline porous aromatic frameworks. Nat. Commun. 2018, 9, 2998. [Google Scholar] [CrossRef]
- Vezse, P.; Gede, M.; Golcs, A.; Huszthy, P.; Toth, T. Synthetic Modifications of a Pb2+-Sensor Acridono-Crown Ether for Covalent Attachment and Their Effects on Selectivity. Molecules 2024, 29, 1121. [Google Scholar] [CrossRef]
- Liu, Z.; Nalluri, S.K.M.; Stoddart, J.F. Surveying macrocyclic chemistry: From flexible crown ethers to rigid cyclophanes. Chem. Soc. Rev. 2017, 46, 2459–2478. [Google Scholar] [CrossRef]
- Gokel, G.W. Crown Ethers and Cryptands; Royal Society of Chemistry: London, UK, 2016; Volume 3. [Google Scholar] [CrossRef]
- Sayed, M.; Pal, H. An overview from simple host–guest systems to progressively complex supramolecular assemblies. Phys. Chem. Chem. Phys. 2021, 23, 26085–26107. [Google Scholar] [CrossRef]
- Oral, I.; Ott, F.; Abetz, V. Thermodynamic study of crown ether–lithium/magnesium complexes based on benz-1,4-dioxane and its homologues. Phys. Chem. Chem. Phys. 2022, 24, 11687–11695. [Google Scholar] [CrossRef]
- Nasri, F. A Review of the Thermodynamics of Complexation of Crown Ethers with Metal Ion. J. Adv. Environ. Health Res. 2022, 10, 263–272. [Google Scholar] [CrossRef]
- Ghildiyal, N. Experimental investigations of complexes of some small ring fluorescent benzo oxa-crown ethers with alkali metal ions in solution phase: A review. J. Mol. Struct. 2024, 1312, 138603. [Google Scholar] [CrossRef]
- Wang, S.; Rong, M.; Sun, L.-B.; Zhao, Y.; Xing, H.; Yang, L. Benzo-12-crown-4 and β-diketone bi-functionalized ionic liquids for highly selective lithium extraction from alkaline solution. Chem. Eng. J. 2024, 497, 154341. [Google Scholar] [CrossRef]
- Katsuta, S.; Ino, Y.; Wakabayashi, H. Silicon-Bridged Bis(12-crown-4) Ethers as Ionophores for Sodium Ion-Selective Electrodes. Molecules 2025, 30, 925. [Google Scholar] [CrossRef]
- Hollstein, S.; von Delius, M. The Dynamic Chemistry of Orthoesters and Trialkoxysilanes: Making Supramolecular Hosts Adaptive, Fluxional, and Degradable. Acc. Chem. Res. 2024, 57, 602–612. [Google Scholar] [CrossRef]
- Begel, S.; Puchta, R.; van Eldik, R. Host–guest complexes of mixed glycol-bipyridine cryptands: Prediction of ion selectivity by quantum chemical calculations, part V. Beilstein J. Org. Chem. 2013, 9, 1252–1268. [Google Scholar] [CrossRef] [PubMed]
- Hu, A.; Wilson, J.J. Advancing Chelation Strategies for Large Metal Ions for Nuclear Medicine Applications. Acc. Chem. Res. 2022, 55, 904–915. [Google Scholar] [CrossRef] [PubMed]
- Morawska, K.; Malinowski, S.; Wardak, M.; Wardak, C. Comparative Study of Potassium Ion-Selective Electrodes with Solid Contact: Impact of Intermediate Layer Material on Temperature Resistance. Molecules 2024, 29, 5803. [Google Scholar] [CrossRef]
- Saris, N.-E.L.; Andersson, M.A.; Mikkola, R.; Andersson, L.C.; Teplova, V.V.; Grigoriev, P.A.; Salkinoja-Salonen, M.S. Microbial toxin’s effect on mitochondrial survival by increasing K uptake. Toxicol. Ind. Health 2009, 25, 441–446. [Google Scholar] [CrossRef]
- Prestegard, J.H. Proton magnetic resonance studies of the cation-binding properties of nonactin. II. Comparison of the sodium ion, potassium ion, and cesium ion complexes. J. Am. Chem. Soc. 1970, 92, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhong, L.; Tang, Y.; Han, T.; Liu, S.; Sun, Z.; Bao, Y.; Wang, H.; He, Y.; Wang, W.; et al. Beyond Nonactin: Potentiometric Ammonium Ion Sensing Based on Ion-selective Membrane-free Prussian Blue Analogue Transducers. Anal. Chem. 2022, 94, 10487–10496. [Google Scholar] [CrossRef]
- Anker, P.; Wieland, E.; Ammann, D.; Dohner, R.E.; Asper, R.; Simon, W. Neutral carrier based ion-selective electrode for the determination of total calcium in blood serum. Anal. Chem. 1981, 53, 1970–1974. [Google Scholar] [CrossRef]
- Habata, Y.; Fukuda, Y.; Akabori, S.; Bradshaw, J.S. Alkali metal ion selectivities of podands that form pseudo-cyclic structures by intramolecular hydrogen bonding. J. Chem. Soc. 2002, 1, 865–869. [Google Scholar] [CrossRef]
- Brachvogel, R.-C.; Hampel, F.; von Delius, M. Self-assembly of dynamic orthoester cryptates. Nat. Commun. 2015, 6, 7129. [Google Scholar] [CrossRef]
- Dabrowa, K.; Niedbala, P.; Majdecki, M.; Duszewski, P.; Jurczak, J. A General Method for Synthesis of Unclosed Cryptands via H-Bond Templated Macrocyclization and Subsequent Mild Postfunctionalization. Org. Lett. 2015, 17, 4774–4777. [Google Scholar] [CrossRef] [PubMed]
- Gutsche, C.D. Calixarenes Revisited; Royal Society of Chemistry: London, UK, 1998; p. 248. [Google Scholar] [CrossRef]
- Rodik, R.; Cherenok, S.; Kalchenko, O.; Yesypenko, O.; Lipkowski, J.; Kalchenko, V. Functional calixarenes for material and life science. Curr. Org. Chem. 2018, 22, 2200–2222. [Google Scholar] [CrossRef]
- Deligöz, H.; Karakuş, Ö.Ö.; Çilgi, G.K. A Brief Review on the Thermal Behaviors of Calixarene-Azocalixarene Derivatives and Their Complexes. J. Macromol. Sci. Chem. A. 2012, 49, 259–274. [Google Scholar] [CrossRef]
- Simões, J.B.; Leite da Silva, D.; Fernandes, S.A.; de Fátima, Â. Calix[n]arenes in Action: Recent Applications in Organocatalysis. Eur. J. Org. Chem. 2022, 38, e202200532. [Google Scholar] [CrossRef]
- Sliwa, W.; Deska, M. Functionalization reactions of calixarenes. ARKIVOC 2011, 1, 496–551. [Google Scholar] [CrossRef]
- Eddaif, L.; Shaban, A.; Telegdi, J. Sensitive detection of heavy metals ions based on the calixarene derivatives-modified piezoelectric resonators: A review. Int. J. Environ. Anal. Chem. 2019, 99, 824–853. [Google Scholar] [CrossRef]
- Peng, S.; He, Q.; Vargas-Zúñiga, G.I.; Qin, L.; Hwang, I.; Kim, S.K.; Heo, N.J.; Lee, C.-H.; Dutta, R.; Sessler, J.L. Strapped calix [4] pyrroles: From syntheses to applications. Chem. Soc. Rev. 2020, 49, 865–907. [Google Scholar] [CrossRef] [PubMed]
- Guérineau, V.; Rollet, M.; Viel, S.; Lepoittevin, B.; Costa, L.; Saint-Aguet, P.; Laurent, R.; Roger, P.; Gigmes, D.; Martini, C.; et al. The synthesis and characterization of giant Calixarenes. Nat. Commun. 2019, 10, 113. [Google Scholar] [CrossRef]
- Mei, C.J.; Ahmad, S.A.A. A review on the determination heavy metals ions using calixarene-based electrochemical sensors. Arab. J. Chem. 2021, 14, 103303. [Google Scholar] [CrossRef]
- Ali, M.B.; Chabanne, R.B.; Vocanson, F.; Dridi, C.; Jaffrezic, N.; Lamartine, R. Comparison study of evaporated thiacalix [4] arene thin films on gold substrates as copper ion sensing. Thin Solid Film. 2006, 495, 368–371. [Google Scholar] [CrossRef]
- Mahiuddin, M.; Banerjee, M.; Shaikh, A.; Shyam, T.; Taniya, S.; Ghosh, A.; Adhikary, A.; Brandão, P.; Félix, V.; Das, D. Optical sensors for detection of nano-molar Zn2+ in aqueous medium: Direct evidence of probe- Zn2+ binding by single crystal X-ray structures. J. Photochem. Photobiol. A 2019, 368, 52–61. [Google Scholar] [CrossRef]
- Ngounoue Kamga, F.A.; Hrubaru, M.-M.; Enache, O.; Diacu, E.; Draghici, C.; Tecuceanu, V.; Ungureanu, E.-M.; Nkemone, S.; Ndifon, P.T. Ni(II)-Salophen—Comprehensive Analysis on Electrochemical and Spectral Characterization and Biological Studies. Molecules 2023, 28, 5464. [Google Scholar] [CrossRef]
- Niklas, J.E. Redox-Active α-Diimines and Novel Schiff Base Ligands for Uranium Coordination Chemistry. Ph.D. Thesis, Auburn University, Auburn, AL, USA, 2020. [Google Scholar]
- Tsantis, S.T.; Tzimopoulos, D.I.; Holynska, M.; Perlepes, S.P. Oligonuclear Actinoid Complexes with Schiff Bases as Ligands—Older Achievements and Recent Progress. Int. J. Mol. Sci. 2020, 21, 555. [Google Scholar] [CrossRef]
- Revanna, B.N.; Madegowda, M.; Neelufar; Swamynayaka, A.; Rangaswamy, J.; Naik, N. A multi-ion detection Schiff base chemosensor: Turn-off and ratiometric fluorescence sensing of Ni2+, Co2+, Cd2+, and Cu2+ ions. J. Mol. Struct. 2025, 1337, 142183. [Google Scholar] [CrossRef]
- Feng, Q.; Chen, Q.; Zhao, J. Excited state intramolecular proton transfer (ESIPT) termination enables “one-to-two” sensing property of an indolocarbazole-based Schiff base. Spectrochim. Acta—A Mol. Biomol. Spectrosc. 2025, 329, 125574. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, N.; Chakraborty, A.; Das, S. A pyrene based fluorescent turn on chemosensor for detection of Cu2+ ions with antioxidant nature. J. Lumin. 2018, 199, 302–309. [Google Scholar] [CrossRef]
- Luo, M.; Liu, Y.; Zhao, J.; Jiang, L.; Chen, X.; Li, W.; Yang, Z.; Yan, Q.; Wang, S.; Chi, Z. Magic tetraphenylethene Schiff base derivatives with AIE, liquid crystalline and photochromic properties. Dye. Pigment. 2022, 202, 110222. [Google Scholar] [CrossRef]
- Hiroto, S.; Miyake, Y.; Shinokubo, H. Synthesis and Functionalization of Porphyrins through Organometallic Methodologies. Chem. Rev. 2017, 117, 2910–3043. [Google Scholar] [CrossRef]
- Paolesse, R.; Nardis, S.; Monti, D.; Stefanelli, M.; Di Natale, C. Porphyrinoids for Chemical Sensor Applications. Chem. Rev. 2017, 117, 2517–2583. [Google Scholar] [CrossRef]
- Negut, C.C.; van Staden, R.-I.S.; van Staden, J.F. Porphyrins-as Active Materials in the Design of Sensors. An Overview. ECS J. Solid State Sci. Technol. 2020, 9, 051005. [Google Scholar] [CrossRef]
- Zha, Q.; Rui, X.; Wei, T.; Xie, Y. Recent advances in the design strategies for porphyrin-based coordination polymers. CrystEngComm 2014, 16, 7371–7384. [Google Scholar] [CrossRef]
- Naithani, S.; Kumar, P.; Dubey, R.; Thetiot, F.; Layek, S.; Goswami, T.; Kumar, S. Design principles for metal–organic receptors targeting optical recognition of Pd(ii) in environmental matrices. J. Mater. Chem. C 2025, 13, 11562–11585. [Google Scholar] [CrossRef]
- Mack, J.; Kobayashi, N. Low Symmetry Phthalocyanines and Their Analogues. Chem. Rev. 2011, 111, 281–321. [Google Scholar] [CrossRef]
- Sekaran, B.; Misra, R. β-Pyrrole functionalized porphyrins: Synthesis, electronic properties, and applications in sensing and DSSC. Coord. Chem. Rev. 2022, 453, 214312. [Google Scholar] [CrossRef]
- Pschepiurca, M.E.R.; Vadra, N.; Suarez, S.A. Versatile Metalloporphyrin-Based Electrochemical Sensing Applications: From Small Gasotransmitters to Macromolecules. Eur. J. Inorg. Chem. 2023, 26, e202300005. [Google Scholar] [CrossRef]
- Kumar, P.; Shim, Y.-B. A novel Mg(II)-selective sensor based on 5,10,15,20-tetrakis(2-furyl)-21,23-dithiaporphyrin as an electroactive material. J. Electroanal. Chem. 2011, 661, 25–30. [Google Scholar] [CrossRef]
- Halder, S.; Bhavana, P. Role of conformational and electronic effects on various thienylporphyrinic ionophores in the detection of metal ions—A potentiometric investigation. J. Mol. Struct. 2017, 1150, 206–213. [Google Scholar] [CrossRef]
- Vlascici, D.; Fagadar-Cosma, E.; Pica, E.M.; Cosma, V.; Bizerea, O.; Mihailescu, G.; Olenic, L. Free Base Porphyrins as Ionophores for Heavy Metal Sensors. Sensors 2008, 8, 4995–5004. [Google Scholar] [CrossRef]
- Tatu, G.-L.A.; Van Staden, J.F. Phthalocyanine Modified Electrodes Based on Reduced Graphene Oxide for Determination of Lead in Different Types of Water. Rev. Roum. Chim. 2019, 64, 887–892. [Google Scholar] [CrossRef]
- Sajjan, V.A.; Aralekallu, S.; Nemakal, M.; Palanna, M.; Prabhu, C.P.K.; Sannegowda, L.K. Nanomolar detection of lead using electrochemical methods based on a novel phthalocyanine. Inorg. Chim. Acta 2020, 506, 119564. [Google Scholar] [CrossRef]
- Mudasir, M.; Baskara, R.A.; Suratman, A.; Yunita, K.S.; Perdana, R.; Puspitasari, W. Simultaneous Adsorption of Zn(II) and Hg(II) Ions on Selective Adsorbent of Dithizone-Immobilized Bentonite in the Presence of Mg(II) Ion. J. Environ. Chem. Eng. 2020, 8, 104002. [Google Scholar] [CrossRef]
- Aprilita, N.H.; Luqman, M.; Suratman, A. Removal of cobalt (II) by dithizone-immobilized nickel slag. Results Chem. 2023, 5, 100698. [Google Scholar] [CrossRef]
- Song, J.-Y.; Liu, Y.; Zhao, H.-Y.; Han, H.-T.; Li, Z.-F.; Guo, W.-H.; Chu, W.-Y.; Sun, Z.-Z. Efficient nickel(ii) naringenin-oxime complex catalyzed Mizoroki–Heck cross-coupling reaction in the presence of hydrazine hydrate. New J. Chem. 2017, 41, 12288–12292. [Google Scholar] [CrossRef]
- Shayesteh, T.H.; Khajavi, F.; Khosroshahi, A.G.; Mahjub, R. Development and validation of a novel, simple, and accurate spectrophotometric method for the determination of lead in human serum. Environ. Monit. Assess. 2015, 188, 7. [Google Scholar] [CrossRef]
- Ganjali, M.R.; Mizani, F.; Emami, M.; Darjezini, M.; Darvich, M.R.; Yousefi, M. Synthesis of a New Oxime and Its Application to the Construction of a Highly Selective and Sensitive Co(II) PVC-Based Membrane Sensor. Anal. Sci. 2004, 20, 531–535. [Google Scholar] [CrossRef]
- Yari, A.; Azizi, S.; Kakanejadifard, A. An electrochemical Ni(II)-selective sensor-based on a newly synthesized dioxime derivative as a neutral ionophore. Sens. Actuators B Chem. 2006, 119, 167–173. [Google Scholar] [CrossRef]
- Abbaspour, A.; Izadyar, A. Highly selective electrode for Nickel (II) ion based on 1.5-diphenylthiocarbazone (dithizone). Microchem. J. 2001, 69, 7–11. [Google Scholar] [CrossRef]
- Dudik, O.; Gil, R.L.; Queirós, R.B. Critical assessment of different ion-to-electron transducers in modified screen-printed electrodes for potentiometric lithium sensing. Microchem. J. 2025, 215, 114195. [Google Scholar] [CrossRef]
- Morf, W.E. The Principles of Ion-Selective Electrodes and of Membrane Transport; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1981; ISBN 978-0-444-99749-4. [Google Scholar]
- Bakker, E.; Bühlmann, P.; Pretsch, E. Carrier-Based Ion-Selective Electrodes and Bulk Optodes. 1. General Characteristics. Chem. Rev. 1997, 97, 3083–3132. [Google Scholar] [CrossRef]
- Kim, Y.; Seo, M.; Baek, S. Ion-selective electrode-based sensors from the macro- to the nanoscale. Sens. Actuators Rep. 2025, 9, 100258. [Google Scholar] [CrossRef]
- Tatara, R.; Shibasaki, Y.; Igarashi, D.; Osada, H.; Aoki, K.; Miyamoto, Y.; Takayama, T.; Matsui, T.; Komaba, S. All-Solid-State Ion-Selective Electrode Inspired from All-Solid-State Li-Ion Batteries. Anal. Chem. 2025, 97, 4819–4823. [Google Scholar] [CrossRef]
- Zhai, J.; Yuan, D.; Xie, X. Ionophore-based ion-selective electrodes: Signal transduction and amplification from potentiometry. Sens. Diagn. 2022, 1, 213–221. [Google Scholar] [CrossRef]
- Rousseau, C.R.; Chipangura, Y.E.; Stein, A.; Bühlmann, P. Effect of Ion Identity on Capacitance and Ion-to-Electron Transduction in Ion-Selective Electrodes with Nanographite and Carbon Nanotube Solid Contacts. Langmuir 2024, 40, 1785–1792. [Google Scholar] [CrossRef]
- Hu, J.; Stein, A.; Bühlmann, P. Rational design of all-solid-state ion-selective electrodes and reference electrodes. TrAC Trends. Anal. Chem. 2016, 76, 102–114. [Google Scholar] [CrossRef]
- Gao, L.; Tian, Y.; Gao, W.; Xu, G. Recent Developments and Challenges in Solid-Contact Ion-Selective Electrodes. Sensors 2024, 24, 4289. [Google Scholar] [CrossRef] [PubMed]
- Wardak, C.; Pietrzak, K.; Morawska, K.; Grabarczyk, M. Ion-Selective Electrodes with Solid Contact Based on Composite Materials: A Review. Sensors 2023, 23, 5839. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Mi, Y.; Bakker, E. Determination of complex formation constants of 18 neutral alkali and alkaline earth metal ionophores in poly(vinyl chloride) sensing membranes plasticized with bis(2-ethylhexyl)sebacate and o-nitrophenyloctylether. Anal. Chim. Acta 2000, 421, 207–220. [Google Scholar] [CrossRef]
- Kumar, C.D.; Sharma, B.; Soujanya, Y.; Chary, V.N.; Patpi, S.; Kantevari, S.; Sastry, G.N. Evaluating the cation binding strength and selectivity of calix[4]pyrroles: A computational and ESI-MS/MS study. Phys. Chem. Chem. Phys. 2014, 16, 17266–17271. [Google Scholar] [CrossRef]
- Katsuta, S.; Maeda, K. Extraction spectrophotometry using a lithium-ion selective metallacrown: Temperature effect on extraction reaction and application to determination of lithium in serum and seawater. Anal. Sci. 2024, 40, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Wagay, S.A.; Hasnain, M.S.; Ali, R. New dimensions in calix[4]pyrrole: The land of opportunity in supramolecular chemistry. RSC Adv. 2019, 9, 38309–38344. [Google Scholar] [CrossRef]
- Pietrzak, M.; Meyerhoff, M.E.; Malinowska, E. Polymeric membrane electrodes with improved fluoride selectivity and lifetime based on Zr(IV)- and Al(III)-tetraphenylporphyrin derivatives. Anal. Chim. Acta 2007, 596, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Bühlmann, P.; Chen, L.D. Supramolecular Chemistry; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Marcos, P.M.; Cragg, P.J. Homooxacalixarene-based supramolecular receptors for cations and anions. Coord. Chem. Res. 2025, 2, 100012. [Google Scholar] [CrossRef]
- Konovalov, A.I.; Antipin, I.S.; Mustafina, A.R.; Solov’eva, S.E.; Pod’yachev, S.N. Design and Ionophore Properties of Some Macrocyclic Calixarene-Based Ligands. Russ. J. Coord. Chem. 2004, 30, 227–244. [Google Scholar] [CrossRef]
- Leko, K.; Usenik, A.; Cindro, N.; Modrušan, M.; Požar, J.; Horvat, G.; Stilinović, V.; Hrenar, T.; Tomišić, V. Enhancing the Cation-Binding Ability of Fluorescent Calixarene Derivatives: Structural, Thermodynamic, and Computational Studies. ACS Omega 2023, 8, 43074–43087. [Google Scholar] [CrossRef] [PubMed]
- Ohms, L.; Eickhoff, L.; Kramer, P.; Villinger, A.; Bresien, J.; Schulz, A. Coinage Metal Complexes of Multidentate Pacman Phosphane Ligands. Chem. Commun. 2023, 59, 5245–5248. [Google Scholar] [CrossRef] [PubMed]
- Sappino, C.; Stefano, S.D.; Lanzalunga, O.; Zonta, C.; Licini, G. Transition Metal Catalysts Bearing Multidentate Ligands for Efficient and Environmental Benign Oxidations by H2O2. Eur. J. Inorg. Chem. 2023, 26, e202200782. [Google Scholar] [CrossRef]
- Sittel, T.; Becker, K.; Polly, R.; Müllich, U.; Geist, A.; Panak, P.J. The Role of Ion Size andπ-Interaction in Stabilizing Calix[4]arene Crown Ether Metal Complexes. Chem. Eur. J. 2025, 31, e202501065. [Google Scholar] [CrossRef]
- Nikšic-Franjic, I.; Saftic, D.P.; Smrecki, V.; Colasson, B.; Reinaud, O.; Piantanida, I.; Višnjevac, A. Transition Metals Coordination by Bis-imidazole-calix[4]arene Ligands with and Without Pyrene Units Grafted at the Large Rim. Int. J. Mol. Sci. 2024, 25, 11314. [Google Scholar] [CrossRef]
- Mulaudzi, I.; Zhang, Y.; Ndlovu, G.F.; Wu, Y.; Legodi, M.A.; van Ree, T. Copper Doped Li3VO4 as Anode Material for Lithium-ion Batteries. Electroanalysis 2020, 32, 2635–2641. [Google Scholar] [CrossRef]
- Kozma, J.; Papp, S.; Gyurcsányi, R.E. TEMPO-Functionalized Carbon Nanotubes for Solid-Contact Ion-Selective Electrodes with Largely Improved Potential Reproducibility and Stability. Anal. Chem. 2022, 94, 8249–8257. [Google Scholar] [CrossRef]
- Cuartero, M.; Parrilla, M.; Crespo, G.A. Wearable Potentiometric Sensors for Medical Applications. Sensors 2019, 19, 363. [Google Scholar] [CrossRef]
- Kaushik, V.; Yakisich, J.S.; Kumar, A.; Azad, N.; Iyer, A.K.V. Ionophores: Potential Use as Anticancer Drugs and Chemosensitizers. Cancers 2018, 10, 360. [Google Scholar] [CrossRef]
- Telting-Diaz, M.; Bakker, E. Effect of Lipophilic Ion-Exchanger Leaching on the Detection Limit of Carrier-Based Ion-Selective Electrodes. Anal. Chem. 2001, 73, 5582–5589. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Jain, S.; Khurana, U. A PVC-based pentathia-15-crown-5 membrane potentiometric sensor for mercury(II). Electroanalysis 1997, 9, 478–480. [Google Scholar] [CrossRef]
- Lyu, Y.; Gan, S.; Bao, Y.; Zhong, L.; Xu, J.; Wang, W.; Liu, Z.; Ma, Y.; Yang, G.; Niu, L. Solid-Contact Ion-Selective Electrodes: Response Mechanisms, Transducer Materials and Wearable Sensors. Membranes 2020, 10, 128. [Google Scholar] [CrossRef]
- Ni, Z.; Wei, C.; Xu, K.; He, J.; Tang, S.; Zhou, S.; Yang, J.; Yang, W.; Zhang, Y.; Li, X. Achieving preeminent stability and reproducibility in an ion-selective electrode for copper ion via introducing a hydrophobic NiCo2S4/PFOA solid-contact. Microchem. J. 2024, 200, 110324. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Y.; Liang, R.; Zhong, L.; Xu, J.; Lu, H.; Xu, X.; Han, T.; Bao, Y.; Ma, Y.; et al. Carbon-Based Transducers for Solid-Contact Calcium Ion-Selective Electrodes: Mesopore and Nitrogen-Doping Effects. Membranes 2022, 12, 903. [Google Scholar] [CrossRef]
- Wang, R.; Sun, L.; Zhang, L.; Zou, C.; Liu, X.; Zhang, H.; Han, Y. In Situ Cation-Switched Organometallic Cage Reconfiguration Enables Highly Selective Li+ Recognition and Extraction. Angew. Chem. Int. Ed. 2025, 64, e202513692. [Google Scholar] [CrossRef]
- Chen, K.-Y.; Kachhadiya, J.; Muhtasim, S.; Cai, S.; Huang, J.; Andrews, J. Underground Ink: Printed Electronics Enabling Electrochemical Sensing in Soil. Micromachines 2024, 15, 625. [Google Scholar] [CrossRef] [PubMed]
- Podrażka, M.; Maciejewska, J.; Adamiak, W.; Witkowska Nery, E.; Jönsson-Niedziółka, M. Facilitated cation transfer at a three-phase junction and its applicability for ionophore evaluation. Electrochim. Acta 2019, 307, 326–333. [Google Scholar] [CrossRef]
- Sabela, A.; Koryta, J.; Valent, O. Ion carrier properties of nigericin studied by voltammetry at the interface of two immiscible electrolyte solutions. J. Electroanal. Chem. Interfacial Electrochem. 1986, 204, 267–272. [Google Scholar] [CrossRef]
- Quentel, F.; Mirčeski, V.; L’Her, M. Kinetics of Anion Transfer across the Liquid|Liquid Interface of a Thin Organic Film Modified Electrode, Studied by Means of Square-Wave Voltammetry. Anal. Chem. 2005, 77, 1940–1949. [Google Scholar] [CrossRef] [PubMed]
- Mei, C.J.; Yusof, N.A.; Alang Ahmad, S.A. Electrochemical Determination of Lead & Copper Ions Using Thiolated Calix[4]arene-Modified Screen-Printed Carbon Electrode. Chemosensors 2021, 9, 157. [Google Scholar] [CrossRef]
- Podrażka, M.; Witkowska Nery, E.; Henares, T.G.; Jönsson-Niedziółka, M.; Arrigan, D.W.M. Ion Transfer Voltammetry with an Electrochemical Pen. Anal. Chem. 2020, 92, 15997–16004. [Google Scholar] [CrossRef]
- Liu, Y.; Crespo, G.A.; Cuartero, M. Voltammetric Ion-Selective Electrodes in Thin-Layer Samples: Absolute Detection of Ions Using Ultrathin Membranes. Anal. Chem. 2024, 96, 1147–1155. [Google Scholar] [CrossRef]
- Jetmore, H.D.; Anupriya, E.S.; Cress, T.J.; Shen, M. Interface between Two Immiscible Electrolyte Solutions Electrodes for Chemical Analysis. Anal. Chem. 2022, 94, 16519–16527. [Google Scholar] [CrossRef]
- Magar, H.S.; Hassan, R.Y.A.; Mulchandani, A. Electrochemical Impedance Spectroscopy (EIS): Principles, Construction, and Biosensing Applications. Sensors 2021, 21, 6578. [Google Scholar] [CrossRef]
- Braik, M.; Dridi, C.; Ali, A.; Abbas, M.N.; Ben Ali, M.; Errachid, A. Development of a perchlorate sensor based on Co-phthalocyanine derivative by impedance spectroscopy measurements. Org. Electron. 2015, 16, 77–86. [Google Scholar] [CrossRef]
- Khripoun, G.A.; Korchak, P.A.; Mikhelson, K.N. Charge transfer resistance at the interface between valinomycin-based potassium-selective membranes and KCl aqueous solutions. Sens. Actuators B Chem. 2025, 424, 136934. [Google Scholar] [CrossRef]
- Moradi, N.; Shamsipur, M.; Taherpour, A.; Pashabadi, A. Impedimetric determination of Cs(I) using AuNPs@PoPD-DB24C8: A targeted molecular-scale perturbation. Anal. Chim. Acta 2020, 1108, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Touzi, H.; Chevalier, Y.; Martin, M.; Ben Ouada, H.; Jaffrezic-Renault, N. Detection of Gadolinium with an Impedimetric Platform Based on Gold Electrodes Functionalized by 2-Methylpyridine-Substituted Cyclam. Sensors 2021, 21, 1658. [Google Scholar] [CrossRef]
- Wang, M.; Wu, Y.; Lou, F.; Cui, W.; Chen, D.; Zhang, X.; Jin, D.; Hun, X. Photoelectrochemical signal for anion and cation detections with photoactive material. Appl. Nanosci. 2022, 12, 2987–2995. [Google Scholar] [CrossRef]
- Zhou, B.; Jiang, Y.; Guo, Q.; Das, A.; Sobrido, A.B.J.; Hing, K.A.; Zayats, A.V.; Krause, S. Photoelectrochemical Detection of Calcium Ions Based on Hematite Nanorod Sensors. ACS Appl. Nano Mater. 2022, 5, 17087–17094. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Li, Y.; Qin, Y.; Jiang, D.; Chen, H.-Y. Ion-selective polymer dots for photoelectrochemical detection of potassium ions. Analyst 2021, 146, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Imali, D.Y.; Perera, E.C.J.; Kaumal, M.N.; Dissanayake, D.P. Conducting polymer functionalization in search of advanced materials in ionometry: Ion-selective electrodes and optodes. RSC Adv. 2024, 14, 25516–25548. [Google Scholar] [CrossRef] [PubMed]
- Tao, B.; Gao, B.; Li, J.; Miao, F.; Zhang, P.; Zang, Y. Photoelectrochemical sensor based on Au/ZnS/ZnO nanomaterials for selective detection of copper ions. Vaccum 2022, 204, 111378. [Google Scholar] [CrossRef]
- Bergveld, P. Development of an Ion-Sensitive Solid-State Device for Neurophysiological Measurements. IEEE Trans. Biomed. Eng. 1970, 17, 70–71. [Google Scholar] [CrossRef]
- Buck, R.P.; Hackleman, D.E. Field Effect Potentiometric Sensors. Anal. Chem. 1977, 49, 2315–2321. [Google Scholar] [CrossRef]
- Li, C.; Li, Z.; Nguyen, T.D.D.; Ingebrandt, S.; Vu, X.T. Real-time and multiplexed detection of sodium and potassium ions using PEDOT:PSS OECT microarrays integrated with ion-selective membranes. Electrochim. Acta 2024, 507, 145111. [Google Scholar] [CrossRef]
- Liu, N.; Chen, R.; Wan, Q. Recent Advances in Electric-Double-Layer Transistors for Bio-Chemical Sensing Applications. Sensors 2019, 19, 3425. [Google Scholar] [CrossRef]
- Yamashita, Y.; Hayakawa, H.; Wang, P.; Makita, T.; Kumagai, S.; Watanabe, S.; Takeya, J. Ion sensors based on organic semiconductors acting as quasi-reference electrodes. Proc. Natl. Acad. Sci. USA 2024, 121, e2405933121. [Google Scholar] [CrossRef]
- Ghittorelli, M.; Lingstedt, L.; Romele, P.; Crăciun, N.I.; Kovács-Vajna, Z.M.; Blom, P.W.M.; Torricelli, F. High-sensitivity ion detection at low voltages with current-driven organic electrochemical transistors. Nat. Commun. 2018, 9, 1441. [Google Scholar] [CrossRef]
- Demuru, S.; Kunnel, B.P.; Briand, D. Real-Time Multi-Ion Detection in the Sweat Concentration Range Enabled by Flexible, Printed, and Microfluidics-Integrated Organic Transistor Arrays. Adv. Mater. Technol. 2020, 5, 2000328. [Google Scholar] [CrossRef]
- Poursaberi, T.; Hajiagha-Babaei, L.; Yousefi, M.; Rouhani, S.; Shamsipur, M.; Kargar-Razi, M.; Moghimi, A.; Aghabozorg, H.; Ganjali, M.R. The Synthesis of a New Thiophene-Derivative Schiff’s Base and Its Use in Preparation of Copper-Ion Selective Electrodes. Electroanalysis 2001, 13, 1513–1517. [Google Scholar] [CrossRef]
- Mahajan, R.K.; Kamal, A.; Kumar, N.; Bhalla, V.; Kumar, M. Selective sensing of mercury(II) using PVC-based membranes incorporating recently synthesized 1,3-alternate thiacalix[4]crown ionophore. Environ. Sci. Pollut. Res. 2013, 20, 3086–3097. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.; Gao, Y.; Wang, P. A general approach to one-step fabrication of single-piece nanocomposite membrane based Pb2+-selective electrodes. Sens. Actuators B Chem. 2019, 281, 705–712. [Google Scholar] [CrossRef]
- Rajagopalan, A.K.; Varghese, S.; Mathew, J. Arsenic detection down to the picomolar level: A low-tech potentiometric approach. Microchem. J. 2025, 210, 112929. [Google Scholar] [CrossRef]
- Gholivand, M.B.; Sharifpour, F. Chromium(III) ion selective electrode based on glyoxal bis(2-hydroxyanil). Talanta 2003, 60, 707–713. [Google Scholar] [CrossRef]
- Mirzaee, E.; Zamani, H.A.; Mohammadhosseini, M. Construction of a New Selective and Sensitive Nd3+ ion-selective Electrode Based on 1-nitroso-2-naphtol as a Neutral Ion Carrier. Int. J. Electrochem. Sci. 2019, 14, 693–704. [Google Scholar] [CrossRef]
- Gupta, V.K.; Jain, A.K.; Agarwal, S.; Maheshwari, G. An iron(III) ion-selective sensor based on a μ-bis(tridentate) ligand. Talanta 2007, 71, 1964–1968. [Google Scholar] [CrossRef]
- Mahajan, R.K.; Kaur, I.; Kaur, R.; Bhalla, V.; Kumar, M. Calix[4]arene Derivatives: Efficient Ionophores for Silver(I) Ion Sensors. Bull. Chem. Soc. Jpn. 2005, 78, 1635–1640. [Google Scholar] [CrossRef]
- Arfin, T.; Tarannum, A. Rapid determination of lead ions using polyaniline-zirconium (IV) iodate-based ion selective electrode. J. Environ. Chem. Eng. 2019, 7, 102811. [Google Scholar] [CrossRef]
- Shamsipur, M.; Mashhadizadeh, M.H. Cadmium ion-selective electrode based on tetrathia-12-crown-4. Talanta 2001, 53, 1065–1071. [Google Scholar] [CrossRef]
- Day, C.; Søpstad, S.; Ma, H.; Jiang, C.; Nathan, A.; Elliott, S.R.; Karet Frankl, F.E.; Hutter, T. Impedance-based sensor for potassium ions. Anal. Chim. Acta 2018, 1034, 39–45. [Google Scholar] [CrossRef]
- Keresten, V.; Mikhelson, K. Voltammetric Ion Sensing with Ionophore-Based Ion-Selective Electrodes Containing Internal Aqueous Solution, Improving Lifetime of Sensors. Membranes 2022, 12, 1048. [Google Scholar] [CrossRef]
- Mattos, G.J.; Rothen, J.A.; Tiuftiakov, N.Y.; Bakker, E. Ion transfer mediated by TEMPO in ionophore-doped thin films for multi-ion sensing by cyclic voltammetry. Anal. Chim. Acta 2024, 1299, 342388. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Kaur, J.; Sharma, J.; Kaur, I. S–S bridged Schiff bases as versatile ionophores: Synthesis and application for electrochemical sensing of Copper(II) and Mercury(II). Anal. Chim. Acta 2024, 1287, 342122. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Cuartero, M.; Crespo, G.A. Lowering the limit of detection of ion-selective membranes backside contacted with a film of poly(3-octylthiophene). Sens. Actuators B Chem. 2019, 297, 126781. [Google Scholar] [CrossRef]
- Pan, D.; Wang, Y.; Chen, Z.; Lou, T.; Qin, W. Nanomaterial/Ionophore-Based Electrode for Anodic Stripping Voltammetric Determination of Lead: An Electrochemical Sensing Platform toward Heavy Metals. Anal. Chem. 2009, 81, 5088–5094. [Google Scholar] [CrossRef] [PubMed]
- Shavokshina, V.A.; Nikitina, V.N.; Karyakin, A.A. Improved performance of Prussian blue solid contact allowing flow injection amperometric detection of potassium ions in the excess of sodium. J. Electroanal. Chem. 2024, 962, 118264. [Google Scholar] [CrossRef]
- Sun, X.; Yin, T.; Zhang, Z.; Qin, W. Redox probe-based amperometric sensing for solid-contact ion-selective electrodes. Talanta 2022, 239, 123114. [Google Scholar] [CrossRef] [PubMed]
- Phoonsawat, K.; Agir, I.; Dungchai, W.; Ozer, T.; Henry, C.S. A smartphone-assisted hybrid sensor for simultaneous potentiometric and distance-based detection of electrolytes. Anal. Chim. Acta 2022, 1226, 340245. [Google Scholar] [CrossRef]
- Stelmach, E.; Maksymiuk, K.; Michalska, A. Dual Sensitivity─Potentiometric and Fluorimetric─Ion-Selective Membranes. Anal. Chem. 2021, 93, 14737–14742. [Google Scholar] [CrossRef]
- Wu, X.; Ma, X.; Liu, Z.; Yu, X.; Yu, H. Porous Core–Shell Yarn in Wearable Electrochemical Sensors for Real-Time Sweat Monitoring. ACS Appl. Mater. Interfaces 2024, 16, 66727–66737. [Google Scholar] [CrossRef]
- Ozer, T.; Henry, C.S. Microfluidic-based ion-selective thermoplastic electrode array for point-of-care detection of potassium and sodium ions. Microchim. Acta 2022, 189, 152. [Google Scholar] [CrossRef]
- Kim, D.Y.; Reza, M.S.; Samad, A.A.; Islam, Z.; Go, J.W.; Park, J.Y. A highly stable and flexible ion-selective patch sensor for real-time sweat Na+ and K+ monitoring. Micro Nano Syst. Lett. 2025, 13, 15. [Google Scholar] [CrossRef]
- Jadoun, S.; Riaz, U.; Budhiraja, V. Biodegradable conducting polymeric materials for biomedical applications: A review. Med. Devices Sens. 2021, 4, e10141. [Google Scholar] [CrossRef]
- Teekayupak, K.; Lomae, A.; Agir, I.; Chuaypen, N.; Dissayabutra, T.; Henry, C.S.; Chailapakul, O.; Ozer, T.; Ruecha, N. Large-scale fabrication of ion-selective electrodes for simultaneous detection of Na+, K+, and Ca2+ in biofluids using a smartphone-based potentiometric sensing platform. Microchim. Acta 2023, 190, 237. [Google Scholar] [CrossRef]
- Gabrieli, G.; Hu, R.; Matsumoto, K.; Temiz, Y.; Bissig, S.; Cox, A.; Heller, R.; Lopez, A.; Barroso, J.; Kaneda, K.; et al. Combining an integrated sensor array with machine learning for the simultaneous quantification of multiple cations in aqueous mixtures. Anal. Chem. 2021, 93, 16853–16861. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zheng, S.; Liang, B.; He, M.; Wu, F.; Yang, J.; Chen, H.-j.; Xie, X. 3D-assembled microneedle ion sensor-based wearable system for the transdermal monitoring of physiological ion fluctuations. Microsyst. Nanoeng. 2023, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.R.; Honig, M.L.; Buhlmann, P. Covalently attached ionophores extend the working range of potentiometric pH sensors with poly(decyl methacrylate) sensing membranes. Analyst. 2024, 149, 1132–1140. [Google Scholar] [CrossRef] [PubMed]
- Abdollahzadeh, M.; Zhu, Y.; Bayatsarmadi, B.; Vepsäläinen, M.; Razmjou, A.; Murugappan, K.; Rodopoulos, T.; Asadnia, M. Portable multiplexed ion-selective sensor for long-term and continuous irrigation water quality monitoring. Comput. Electr. Agric. 2024, 227, 109455. [Google Scholar] [CrossRef]
- Bakker, E. Electroanalysis with Membrane Electrodes and Liquid–Liquid Interfaces. Anal. Chem. 2016, 88, 395–413. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).