Abstract
The cosmetic and personal care industry is increasingly shifting toward plant-derived ingredients that combine multifunctional biological activity with favorable safety and sustainability profiles. Trigonella foenum-graecum L. (fenugreek), a leguminous plant traditionally used in food and medicine, has recently attracted growing attention as a potential cosmetic and cosmeceutical ingredient. This review provides a comprehensive and critical synthesis of current knowledge on fenugreek in the context of skin and hair care applications. First, the phytochemical profile of fenugreek relevant to cosmetics is examined, with emphasis on polyphenols and flavonoids, steroidal saponins, alkaloids (notably trigonelline), and mucilage-rich galactomannans. The biological activities of these constituents are then discussed in relation to key cutaneous and scalp mechanisms, including antioxidant defense, anti-inflammatory and antimicrobial effects, anti-glycation and dermal matrix protection, skin hydration and barrier support, as well as hair growth promotion and scalp health. Particular attention is given to recent in vitro, ex vivo, and emerging clinical evidence supporting anti-aging, moisturizing, and hair-care claims. Current formulation strategies and green extraction technologies enabling the incorporation of fenugreek-derived ingredients into topical products are reviewed, alongside considerations of safety, toxicity, and regulatory status under EU cosmetic legislation. Finally, existing research gaps are identified, highlighting the need for standardized extracts, robust clinical validation, and advanced omics-based approaches. This review highlights fenugreek’s strong potential as a versatile plant-based ingredient in cosmetics, despite remaining understudied in topical applications.
1. Introduction
Over the past decade, the cosmetics and personal care industry has undergone a marked shift towards naturally derived and plant-based ingredients, driven by consumer concerns about safety, sustainability, and transparency, as well as by the search for multifunctional “cosmeceutical” actives with scientifically demonstrated efficacy. Recent reviews on natural products and plant extracts in skin care underline that botanical ingredients now play a central role in formulations targeting photoprotection, anti-inflammatory effects, wound healing, anti-aging, and skin barrier support [1,2]. In parallel, cosmetic science is increasingly integrating concepts and tools from phytochemistry, dermatology, and biotechnology to rationally select and standardize plant-derived actives, while also acknowledging important challenges related to stability, bioavailability, and safety assessment [3,4]. Within this context, plant-based ingredients are no longer seen as simple marketing add-ons, but rather as complex mixtures of bioactive molecules that can modulate well-defined skin targets such as oxidative stress, inflammation, extracellular matrix degradation and melanogenesis [2].
In this context, fenugreek (Trigonella foenum-graecum L.) stands out as an interesting plant with real potential for cosmetic and cosmeceutical uses. Fenugreek is an annual legume widely used as a food, spice, and medicinal plant across South Asia, North Africa and the Middle East, with a long history of traditional use for metabolic, digestive and women’s health conditions [5,6]. Pharmacological and nutraceutical reviews consistently describe fenugreek seeds and leaves as rich sources of steroidal saponins (e.g., diosgenin), alkaloids (e.g., trigonelline), flavonoids, polyphenols, and galactomannan-type mucilage, which together confer antioxidant, anti-inflammatory, hypoglycemic, hypolipidemic and gastroprotective effects in a variety of experimental models [5,6]. Although these properties have been mainly explored in the context of systemic health, they are also highly relevant to skin and hair physiology, where oxidative stress, chronic low-grade inflammation, extracellular matrix breakdown, and microcirculatory changes contribute to intrinsic and extrinsic aging, barrier dysfunction, scalp disorders and hair loss [2].
Only in recent years has fenugreek been more explicitly investigated as a cosmetic active. A 2025 study in Cosmetics optimized ultrasound-assisted extraction of fenugreek seeds to obtain a phenolic-rich extract with high antioxidant capacity, with the explicit aim of developing an eco-designed cosmetic formulation; the authors reported conditions that maximized total phenolic content and radical-scavenging activity while maintaining physicochemical stability compatible with topical products, thereby supporting the feasibility of fenugreek as a skin-care ingredient [7]. In Pharmaceutics, ethanolic fenugreek seed extract was shown to exert potent anti-aging effects in vitro, including collagenase inhibition more pronounced than vitamin C and protection of dermal fibroblasts against oxidative and inflammatory stress, suggesting a direct relevance for anti-wrinkle and dermal matrix-preserving formulations [8]. Other recent work has reported favorable antioxidant and anti-inflammatory profiles for fenugreek-based creams enriched in flavonoid glycosides, as well as beneficial effects in topical applications aimed at improving skin texture and signs of aging [9]. In parallel, nutraceutical and topical uses of fenugreek in hair-care contexts including supplements for hair loss and traditional preparations for scalp health have been documented, further supporting its potential as a dual skin- and hair-care active [10]. This review aims to gather and evaluate the available data on fenugreek’s role in skin and hair care products. The term “Skin–hair benefits” refers to improvements in cutaneous homeostasis (hydration, barrier integrity, anti-aging, soothing) and scalp/hair physiology (follicle protection, reduced shedding, conditioning, microbiome balance), mediated by key pathways including oxidative stress reduction, inflammation modulation, extracellular matrix protection (collagenase inhibition), glycation prevention, and antimicrobial or microbiome-supporting actions [2].
Specifically, we aim to (i) summarize the phytochemical constituents of T. foenum-graecum that are most relevant to skin and hair applications, including phenolic compounds, flavonoids, saponins, alkaloids and mucilaginous polysaccharides; (ii) analyze experimental evidence on cutaneous and scalp-related mechanisms, such as antioxidant, anti-inflammatory, anti-glycation, photoprotective, barrier-supporting and hair-growth-modulating effects; (iii) review current formulation approaches and extraction technologies used to incorporate fenugreek derivatives into topical products, with attention to eco-design, stability and delivery issues; and (iv) identify knowledge gaps and future research directions, including needs for clinical studies, standardized characterization, and regulatory alignment in the context of natural and plant-based cosmetics. By integrating data from pharmacological, cosmetic science and formulation research, this article seeks to position fenugreek within the broader landscape of plant-derived cosmetic actives and to clarify its realistic opportunities and limitations for skin and hair care.
2. Botanical, Agronomic, and Ethnopharmacological Overview
Trigonella foenum-graecum L. (fenugreek) is an annual herb of the Fabaceae family, featuring an erect branched stem (20–60 cm), trifoliate leaves with obovate leaflets, papilionaceous flowers (pale yellow-white), and narrow curved pods containing 10–20 angular seeds [11]. Key bioactive-rich parts include seeds (steroidal saponins, alkaloids like trigonelline, galactomannans) and leaves (phenolics, carotenoids, vitamins). Specialized structures include glandular trichomes on leaves/stems (phenolic secretion) and seed endosperm (galactomannan storage). These organs contribute differentially to cosmetic-relevant bioactives [5,11,12].
Fenugreek is considered native to the Eastern Mediterranean and Western Asia but is now widely cultivated across South Asia, the Middle East, North Africa, and parts of Europe [12]. Recent agronomic reviews indicate that India remains the largest producer, followed by Pakistan, Ethiopia, and Egypt, and that the crop is well adapted to semi-arid conditions, low-input systems, and diverse cropping patterns, including intercropping with cereals and legumes [13]. Ongoing breeding and omics-based programs are focusing on improving stress tolerance, seed yield, and the content of bioactive metabolites, underscoring fenugreek’s strategic value as a multipurpose industrial and medicinal crop [14].
Ethnopharmacologically, fenugreek occupies a prominent place in Ayurveda, Unani, and Mediterranean folk medicine, where seeds and leaves are used internally for metabolic disorders (diabetes, hyperlipidemia), digestive complaints, and women’s health, and externally for inflammatory skin conditions, wounds, and musculoskeletal pain [15]. Traditional topical preparations typically consist of pastes made from crushed seeds or leaf powders mixed with water, yogurt, or oils and applied as poultices to erythematous, edematous, or infected skin. Historical and contemporary ethnobotanical surveys describe their use to soften rough skin, alleviate acne and minor infections, and promote wound healing [16]. In hair care, fenugreek seeds are frequently macerated in oils or water and applied as masks or rinses to strengthen hair shafts, reduce dandruff, and mitigate hair shedding practices now echoed in commercial “methi” hair oils and herbal hair tonics [17].
3. Phytochemical Profile Relevant to Cosmetic Applications
3.1. Polyphenols and Flavonoids
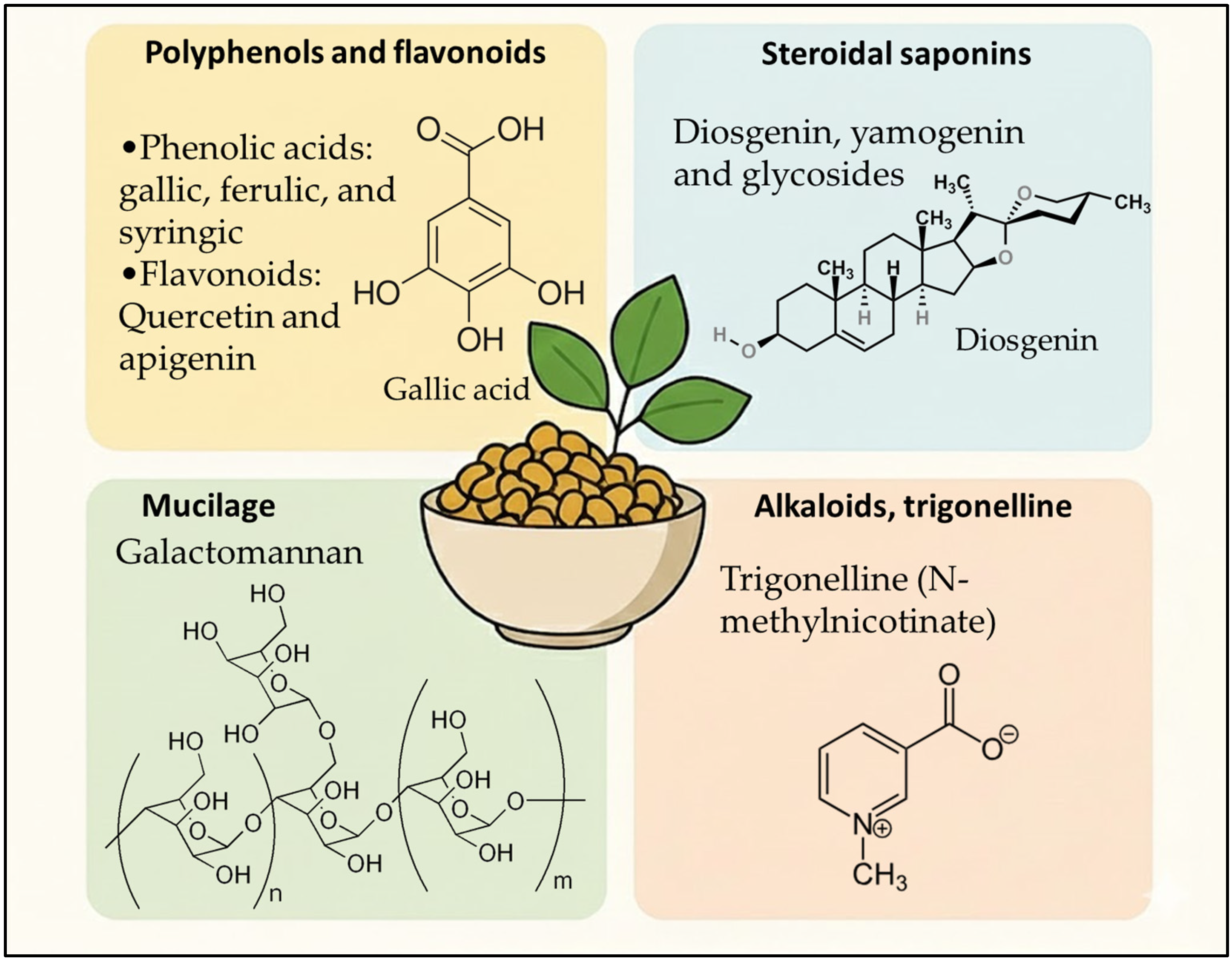
Fenugreek contains a diverse phenolic fraction that is highly relevant for cosmetic performance because many of its constituents target oxidative stress, inflammation, and extracellular-matrix degradation key drivers of skin aging and scalp dysfunction. As already framed in Figure 1, the most recurrent families are phenolic acids (hydroxybenzoic and hydroxycinnamic acids) and flavonoids (mainly flavone/flavonol glycosides). Across seed, leaf, and microgreen matrices, analytical studies consistently report gallic, vanillic, syringic, caffeic, ferulic, chlorogenic, and p-coumaric acids, alongside flavonoids such as quercetin, rutin, kaempferol, and multiple apigenin/luteolin glycosides.
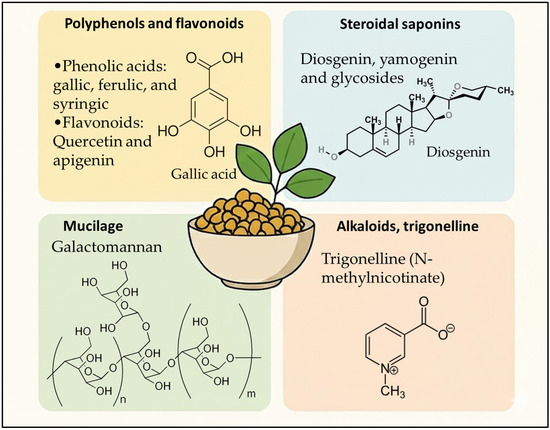
Figure 1.
Phytochemical profile of Trigonella foenum-graecum relevant to cosmetic applications.
These profiles are typically established using HPLC-PDA/DAD and LC–MS(/MS) workflows, enabling both tentative annotation (accurate mass/fragmentation) and targeted quantification. For example, UPLC-MS work highlighted apigenin-7-O-glycoside and luteolin-7-O-glycoside among abundant fenugreek flavonoids, supporting a plausible mechanistic link to antioxidant/anti-inflammatory activity in topical contexts [18]. Importantly, seed-specific profiling also indicates that fenugreek polyphenols are not limited to simple aglycones: comprehensive HPLC-DAD-ESI-MS characterization showed predominantly flavone derivatives with apigenin as a major aglycone, including acylated/non-acylated glycosides chemotypes that matter for formulation because glycosylation can improve water-compatibility but may reduce permeability unless delivery is optimized [19] (Table 1).

Table 1.
Reported identification of fenugreek polyphenols/flavonoids.
3.2. Steroidal Saponins
Steroidal saponins are among the most characteristic secondary metabolites of fenugreek seeds and represent a key fraction when positioning fenugreek as a multifunctional cosmetic active. Chemically, fenugreek saponins are described as a complex mixture of spirostanol and furostanol glycosides, with sugar chains that may be straight or branched, creating a broad “saponin fingerprint” that is highly dependent on genotype and origin [25]. Across analytical characterizations, the dominant aglycones reported for fenugreek saponins are diosgenin and its 25-epimer yamogenin, which act as core steroidal backbones to which diverse glycosidic motifs are attached [25]. A recurrent challenge in the literature is nomenclature overlap, where identical structures have been reported under different common names (e.g., protodioscin/compound C; protoneodioscin/trigonelloside C), emphasizing the need for harmonized marker selection and structure-confirmed reference standards when translating these extracts into cosmetic-grade ingredients [25]. From a formulation perspective, steroidal saponins are relevant because they combine biological bioactivity potential with surface-active behavior (amphiphilic structure), which can influence cleansing, foaming, and dispersion properties features frequently leveraged in botanical hair-care and scalp-care products. While cosmetic mechanisms must be supported by topical studies, the broader pharmacological literature shows that fenugreek seed extracts enriched in steroidal saponins can be prepared to high purity (≥90% steroid saponins reported) and that these saponins represent a well-defined bioactive fraction within the plant matrix [26]. Extraction technology strongly impacts saponin recovery. A green-oriented study reported microwave-assisted extraction optimized to maximize total saponins (alongside phenolics and antioxidant capacity), with LC–MS-QTOF used to identify bioactives in the optimized extract an approach aligned with eco-designed cosmetic ingredient development [27]. Finally, quantitative data underline substantial geographic and genetic variability: one HPLC-ELSD-ESI-MS study reported a total steroidal saponin content of ~0.14% (dry matter basis) for Polish fenugreek seeds, contrasted with markedly higher ranges reported for Asian materials (e.g., ~0.92–1.68 g/100 g dry matter; up to ~6% dry matter in other reports cited within the same paper) [25]. This variability is directly relevant to cosmetics, where batch-to-batch standardization (e.g., using diosgenin/yamogenin-linked markers or major glycosides such as protodioscin) becomes critical for consistent efficacy and sensorial performance (Table 2).

Table 2.
Reported identification of fenugreek steroidal saponins.
3.3. Mucilage and Galactomannans
Fenugreek seed mucilage is a polysaccharide-rich hydrocolloid whose dominant structural component is galactomannan, a water-soluble heteropolysaccharide with a mannan backbone and galactose substitutions. A recent Journal of Agricultural and Food Chemistry review emphasizes that fenugreek mucilage stands out from many conventional hydrocolloids by its high degree of galactose substitution and abundance of hydroxyl groups, features that rationalize its strong hydration/interaction capacity and its technological value as a natural thickener and stabilizer [28]. Beyond compositional identity, the functional profile of fenugreek galactomannan maps directly onto cosmetic formulation needs (hydration, texture, film formation, sensory feel). A Carbohydrate Polymers study isolating and purifying fenugreek galactomannan reported characteristic FTIR bands and quantified several performance-relevant attributes, including very high oil-holding capacity (626.46%), foaming capacity (25.5%), swelling index (86%), and foaming stability (18.55), alongside notable emulsion performance and defined thermal transitions (melting range 66–139 °C, degradation temperature 296.45 °C) [29] (Table 3). These parameters support the use of fenugreek mucilage/galactomannan as a rheology modifier, stabilizer, and film-forming matrix in “natural hydrogel”, mask, scalp-gel, and conditioner-type formats.
Processing strongly influences the final polymer properties. Work in Food and Bioprocess Technology examined stepwise extraction strategies (e.g., ultrasound followed by microwave) and reported thermal events consistent with moisture loss and polysaccharide backbone degradation (e.g., a moisture-loss region spanning ~60–135 °C with a peak at ~71.54 °C, and subsequent higher-temperature events reflecting polymer integrity changes) [30]. Such data are valuable for cosmetics because they inform drying, heating, and stability windows during manufacturing (Table 3).
Finally, fenugreek mucilage’s high swelling/wetting behavior is corroborated by excipient-focused studies. In a formulation study evaluating fenugreek seed mucilage as a disintegrant, the authors reported that extracted mucilage was “devoid of toxicity” in their testing and that a 4% level produced rapid hydration-linked performance (short disintegration time and complete release in their model system) [30]. While pharmaceutical endpoints do not directly translate to skin, they reinforce the mucilage’s rapid water uptake and biocompatibility-oriented positioning for topical textures.

Table 3.
Key Fenugreek mucilage/galactomannan evidence from the provided sources.
Table 3.
Key Fenugreek mucilage/galactomannan evidence from the provided sources.
| Publication | Material | What Was Characterized? | Key Data/Takeaways Relevant to Cosmetics |
|---|---|---|---|
| Urooj et al., (2025) [28] | Fenugreek seed mucilage (FSM) | Galactomannan structure & substitution degree | High galactose substitution → excellent water-binding & hydration capacity; ideal for moisturizing gels, leave-on products and skin conditioning agents [28] |
| Nalbantova et al. (2024) [31] | Fenugreek galactomannan | Review of structure–property relationships and applications | Consolidates multifunctionality (hydration, film-forming, rheology modification) → broad utility in natural hydrogels, masks, scalp gels and conditioners [31] |
| Rashid et al., (2018) [29] | Purified fenugreek galactomannan | Swelling index, oil-holding capacity, foaming properties | Swelling index 86%, oil-holding capacity 626.46%, foaming capacity 25.5% → superior hydration, emollience and film-forming; excellent for masks, conditioners and scalp care products [29] |
| Kumar et al., (2009) [30] | Galactomannan extracted from fenugreek seeds | Thermal stability (DSC/TGA) | Moisture loss region 60–135 °C, polymer degradation > 296 °C → compatible with cosmetic heating/drying processes (typically 60–140 °C); stable during manufacturing of creams, gels and masks [30] |
3.4. Alkaloids, Trigonelline
Among the alkaloidal constituents of Fenugreek, trigonelline (N-methylnicotinic acid) is by far the most abundant and best characterized, and it represents a compound of growing interest for cosmetic and cosmeceutical applications. Trigonelline is a pyridine alkaloid biosynthetically derived from niacin (vitamin B3) methylation, and it is widely distributed in fenugreek seeds [32], where it has been consistently identified and quantified using HPLC, LC–MS, and NMR-based approaches in phytochemical profiling studies [33,34,35].
Standardization is critical for cosmetic consistency. Due to significant batch-to-batch variability in fenugreek phytochemical profiles (influenced by genotype, origin, agronomic conditions, and extraction methods), the use of validated marker compounds and analytical protocols is essential. Trigonelline serves as a key alkaloid marker and is precisely quantified by RP-HPLC-UV. For steroidal saponins, diosgenin and protodioscin are targeted using HPLC-ELSD-ESI-MS or HPTLC, with typical contents ranging from 0.12–0.18% in raw seeds to up to 0.5–1.7% in enriched extracts [25]. These standardized approaches ensure reproducible bioactivity (e.g., antioxidant, anti-inflammatory) and facilitate regulatory compliance in cosmetic formulations.
4. Biological Activities and Cosmetic Mechanisms of Fenugreek
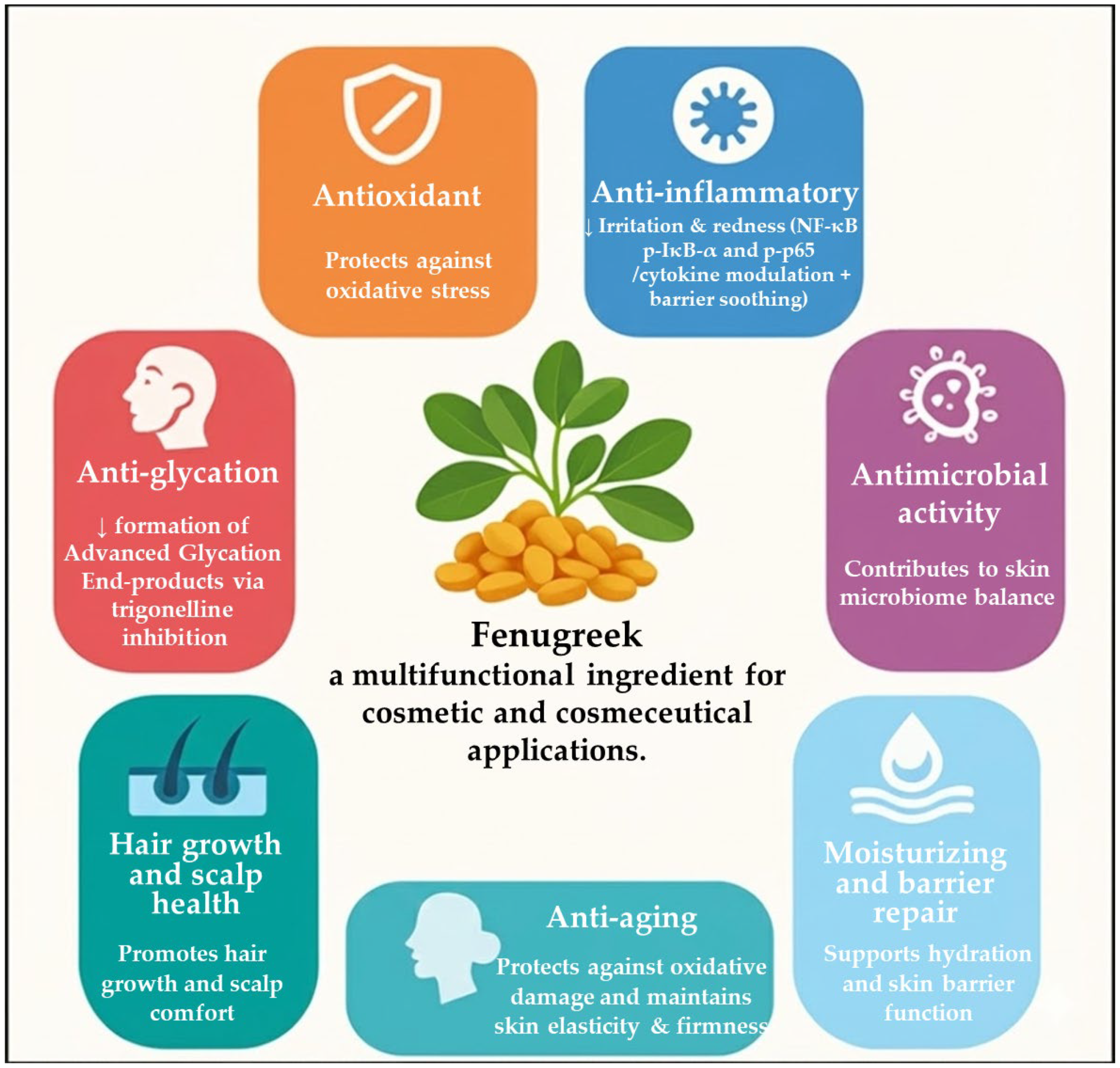
Fenugreek is a multifunctional medicinal and culinary plant whose biological activities are largely attributable to its rich and diverse phytochemical profile, including steroidal saponins, flavonoids, alkaloids (notably trigonelline), polyphenols, and polysaccharides such as galactomannan and mucilage. These constituents collectively underpin a broad spectrum of biological effects that are directly relevant to skin and hair physiology, positioning fenugreek as a promising ingredient for cosmetic and cosmeceutical applications (Figure 2).
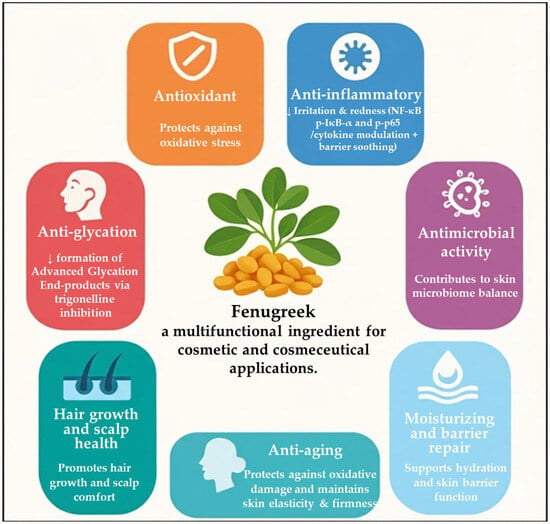
Figure 2.
Biological activities and cosmetic mechanisms of Trigonella foenum-graecum.
4.1. Antioxidant Mechanisms and Protection Against Oxidative Stress
Oxidative stress plays a central role in cutaneous aging, inflammation, barrier disruption, and hair follicle dysfunction [36,37,38]. Fenugreek seeds and their extracts exhibit strong antioxidant activity, consistently demonstrated through their ability to scavenge reactive oxygen species (ROS), inhibit lipid peroxidation, and neutralize hydroxyl radicals. These effects are driven by phenolic compounds and flavonoids (e.g., ferulic acid, quercetin, apigenin glycosides), which donate electrons or hydrogen atoms to stabilize free radicals [7,39]. Pterocarpans from fenugreek aerial parts further exhibit chain-breaking antioxidant potential by donating hydrogen atoms to peroxyl radicals, thereby interrupting lipid peroxidation chains and complementing the ROS scavenging of phenolic/flavonoid compounds [39]. Polysaccharide fractions also contribute significantly, as demonstrated by the potent in vitro and in vivo antioxidant capacity of fenugreek water-extractable polysaccharide (FWEP) [40]. Several studies show that phenolic-rich extracts upregulate endogenous antioxidant enzymes such as superoxide dismutase (SOD) and catalase (CAT), while reducing lipid peroxidation markers [39,41]. Phenolic-rich extracts obtained through optimized extraction strategies displayed high total phenolic content and strong radical-scavenging capacity, reinforcing the relevance of fenugreek for anti-aging and anti-pollution cosmetic formulations [8]. These mechanisms collectively limit oxidative damage to lipids, proteins, and DNA in cutaneous tissues, supporting fenugreek’s role in preventing premature skin aging, hyperpigmentation, and loss of dermal integrity [7,8,41] (Table 4).

Table 4.
Reported Antioxidant activities and protection against oxidative stress.
4.2. Anti-Inflammatory Activity and Skin-Soothing Effects
Chronic low-grade inflammation is a hallmark of sensitive skin, inflammatory dermatoses, and photoaging [47]. Fenugreek demonstrates potent anti-inflammatory activity, attributed to the synergistic action of its phytochemicals [7,48]. In particular, pterocarpans isolated from fenugreek aerial parts have shown marked anti-inflammatory effects, while other constituents modulate inflammatory pathways involved in cytokine production and oxidative stress [49] (Table 5). Fenugreek demonstrates potent anti-inflammatory activity through synergistic actions of its phytochemicals, particularly pterocarpans from aerial parts and saponins/flavonoids from seeds [48,49]. Key mechanisms include modulation of the NF-κB signaling pathway, with reduced nuclear translocation of p65 subunit and downregulation of pro-inflammatory cytokines such as TNF-α and IL-6 [48,50,51]. Pterocarpans and other constituents further inhibit inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) expression, thereby decreasing nitric oxide and prostaglandin production [49]. These pathways are supported by in vivo models showing suppression of adjuvant-induced inflammation via mucilage fractions [52] and by studies linking fenugreek extracts to reduced NF-κB activation in various inflammatory contexts [50,51]. Combined with strong antioxidant effects, these mechanisms provide a scientific basis for fenugreek’s traditional use in soothing irritated or inflamed skin and its inclusion in formulations for sensitive, reactive, or redness-prone skin [7,10,52]. These anti-inflammatory properties support fenugreek’s traditional topical use for soothing irritated or inflamed skin and provide a scientific rationale for its inclusion in cosmetic products designed for sensitive, reactive, or redness-prone skin. The combined antioxidant and anti-inflammatory actions enable fenugreek extracts to mitigate inflammation induced by UV exposure, pollution, or microbiome imbalance [10].

Table 5.
Reported Anti-inflammatory Effects of Fenugreek.
4.3. Antimicrobial Activity and Skin Microbiome Balance
Fenugreek has been shown in multiple in vitro studies to exert antimicrobial effects against both Gram-positive and Gram-negative bacteria, supporting its potential role in cosmetic formulations targeting microbial imbalance, acne-prone skin, and scalp disorders [54,55]. The antimicrobial activity of fenugreek seed extracts has been reported using standard agar diffusion and broth microdilution methods, with methanolic extracts frequently showing greater inhibitory effects than aqueous preparations. For example, the methanolic extract of fenugreek seeds inhibited the growth of Staphylococcus aureus, Escherichia coli, and other pathogenic bacteria in a concentration-dependent manner, while aqueous extracts showed comparatively weaker activity [22]. A recent in vitro study demonstrated that methanol extracts of fenugreek seeds inhibited multidrug-resistant S. aureus and E. coli, highlighting its relevance against strains associated with skin infections and bacterial dysbiosis [56]. Similarly, comparative antimicrobial assays documented zones of inhibition against Pseudomonas aeruginosa, S. aureus, Bacillus subtilis, and E. coli when using methanolic and other organic solvent extracts [57]. These antimicrobial effects are attributed to fenugreek’s diverse phytochemicals, including flavonoids, saponins, and alkaloids, which may disrupt microbial cell membranes and interfere with key bacterial processes. Thus, fenugreek’s antimicrobial effects (against S. aureus, E. coli, etc. [54,57]) support skin microbiome balance by reducing pathogenic overgrowth while preserving commensals, potentially preventing dysbiosis-related irritation or scalp disorders (e.g., dandruff). This contributes to skin–hair benefits via healthier scalp environment, reduced inflammation, and improved follicle anchorage/hair strength [10,56].
Collectively, these findings indicate that fenugreek extracts can contribute to maintaining skin microbiome balance, reduce pathogenic colonization, and offer a natural antimicrobial component in facial cleansers, tonics, and scalp formulations designed for acne-prone or compromised skin (Table 6).

Table 6.
Antimicrobial Activity of Trigonella foenum-graecum Extracts.
4.4. Anti-Aging, Anti-Glycation, and Dermal Matrix Protection
Skin aging is strongly driven by oxidative stress, chronic low-grade inflammation, and extracellular matrix (ECM) remodeling, particularly through the activation of matrix-degrading enzymes such as collagenases (MMPs). Fenugreek has gained interest as a cosmeceutical active because several of its constituents (polyphenols/flavonoids, steroidal saponins/sapogenins, and alkaloids) can converge on these core aging pathways. A key mechanistic evidence comes from an in vitro anti-aging study showing that an ethanolic fenugreek seed extract inhibited collagenase activity with an IC50 of 0.57 ± 0.02 mg/mL, reported as ~2.6-fold more potent than vitamin C in the same assay, and also provided cellular protection under oxidative/inflammatory stress conditions; the same work further explored nanoencapsulation to improve extract stability and performance consistency an important consideration for cosmetic formulation [8].
The inhibitory effect targets primarily interstitial collagenases, including MMP-1 (interstitial collagenase), MMP-8 (neutrophil collagenase), and MMP-13 (collagenase-3), which are key enzymes responsible for extracellular matrix degradation in photoaged and intrinsically aged skin [8]. By reducing the activity of these specific MMPs, fenugreek extract helps preserve dermal collagen integrity and limits wrinkle formation.
Recent clinical evidence strengthens these findings: A 2025 randomized, double-blind, placebo-controlled trial on 75 women (35–55 years) with mild to moderate wrinkles demonstrated that two standardized fenugreek seed extract creams (flavonoid glycosides-based and eleutheroside-oligosaccharide-based) applied topically for 60 days significantly improved skin hydration, reduced wrinkle depth, decreased transepidermal water loss, and showed trends toward reduced roughness and increased collagen distribution, with no adverse events [9]. Beyond enzymatic inhibition, fenugreek’s steroidal sapogenin diosgenin has been associated with improved skin collagen content in experimental work, supporting the plausibility of dermal matrix-supporting effects through modulation of fibroblast dynamics and collagen turnover (even though this evidence is not topical and should be interpreted cautiously for cosmetics) [61]. Anti-glycation activity is also relevant because advanced glycation end-products (AGEs) contribute to collagen stiffening and loss of elasticity. Fenugreek’s alkaloid trigonelline has been reported to reduce AGE-related endpoints and/or show antiglycating effects in experimental systems, providing a rationale for investigating fenugreek-derived fractions in anti-glycation cosmetic strategies [62,63]. Taken together, these findings suggest that fenugreek could act on multiple aging pathways, making it an attractive option for anti-aging formulations, though further topical studies are needed [7].
4.5. Moisturizing, Barrier Repair, and Soothing Effects
Skin hydration and barrier integrity are fundamental determinants of cutaneous health, comfort, and resilience. Disruption of the stratum corneum lipid–protein matrix leads to increased transepidermal water loss (TEWL), dryness, irritation, and heightened sensitivity to environmental stressors [64]. Fenugreek exhibits significant potential in this context, largely due to its high content of mucilage and galactomannan polysaccharides, complemented by anti-inflammatory and antioxidant constituents [50].
Fenugreek seed mucilage is a hydrophilic polysaccharide system dominated by galactomannans, which display strong water-binding, swelling, and film-forming properties. Physicochemical studies have shown that fenugreek galactomannan exhibits high swelling indices, oil-holding capacity, and viscosity, enabling it to form a protective, semi-occlusive film on biological surfaces. Such films reduce water evaporation from the skin surface and contribute to sustained hydration, a mechanism directly relevant to moisturizing cosmetic formulations [28,65]. Beyond hydration, these polysaccharides play a role in barrier repair and reinforcement. By forming a continuous hydrophilic network, fenugreek mucilage can support the stratum corneum barrier, potentially reducing TEWL and improving skin softness and elasticity [28,65].
Fenugreek seed mucilage, rich in galactomannan polysaccharides, exhibits strong water-binding, swelling, and film-forming properties, enabling the formation of a protective semi-occlusive film on the skin surface that reduces transepidermal water loss (TEWL) and sustains hydration [28,29,65,66]. This hydrophilic matrix supports stratum corneum barrier reinforcement and improves skin softness and elasticity [28,65]. The soothing and calming effects arise from the synergy between mucilage’s physical barrier function and the anti-inflammatory/antioxidant actions of flavonoids and saponins, which collectively mitigate irritation, cytokine-mediated inflammation, and oxidative stress-induced barrier disruption [48,50,52]. Experimental evidence further shows that polysaccharide fractions promote wound healing and epithelial regeneration, reinforcing barrier repair beyond simple moisturization [67].
Fenugreek also exhibits soothing and calming effects, which are essential for sensitive or compromised skin. The combination of mucilage with anti-inflammatory phytochemicals (flavonoids and saponins) contributes to reduced irritation and enhanced skin comfort [50]. Processing-oriented studies have highlighted the thermal and structural stability of fenugreek galactomannan, supporting its compatibility with cosmetic manufacturing conditions and its use in hydrogels, masks, and leave-on products [66]. Collectively, these properties position fenugreek-derived mucilage and polysaccharide-rich extracts as valuable natural ingredients for moisturizing, barrier-supporting, and soothing cosmetic products, particularly those targeting dry, sensitive, or environmentally stressed skin, while aligning with current trends toward biocompatible and plant-based hydrocolloids [7,28,66].
4.6. Hair Growth Promotion and Scalp Health
Fenugreek has a long-standing reputation in traditional hair care, and recent scientific evidence provides mechanistic support for these uses. One of the primary mechanisms underlying its hair-supporting effects is its antioxidant and anti-inflammatory activity, which help protect hair follicles from oxidative stress and inflammatory microenvironments known to contribute to hair thinning and alopecia [68].
Oxidative stress can impair dermal papilla cell function and disrupt the hair growth cycle, while chronic scalp inflammation is increasingly recognized as a contributor to hair loss [38]. Fenugreek seed extracts, rich in flavonoids, polyphenols, and alkaloids, have demonstrated strong antioxidant activity and the ability to modulate inflammatory responses, thereby creating a more favorable scalp environment for hair growth [6,10]. Steroidal saponins and sapogenins, particularly diosgenin derivatives, have been proposed to influence hormonal and inflammatory pathways involved in hair follicle regulation.
Mechanistically, these effects likely involve modulation of dermal papilla cell signaling pathways, including inhibition of inflammatory cytokine signaling (e.g., IL-1β, TNF-α) that induces premature catagen entry, and potential indirect support of Wnt/β-catenin and Sonic Hedgehog (Shh) pathways that promote anagen phase maintenance and follicular proliferation. These actions collectively contribute to extended anagen duration, reduced telogen effluvium, and improved hair anchorage [69,70]. These compounds may indirectly modulate dermal papilla cell function and prolong the anagen phase by reducing cytokine-mediated induction of catagen [69]. Although direct clinical evidence demonstrating dermal papilla cell modulation remains limited, experimental studies suggest improved follicular anchorage and reduced hair breakage following fenugreek-based treatments [70]. Emerging clinical data include a randomized, double-blind, placebo-controlled trial reporting that a topical fenugreek seed gel significantly reduced hair loss and trichoptilosis after two months of application [71]. Fenugreek’s mucilage and galactomannan polysaccharides also play a functional role in hair care. These polymers exhibit excellent film-forming and conditioning properties, allowing them to coat the hair shaft, enhance smoothness, reduce friction, and improve manageability [72]. On the scalp, their moisturizing and soothing effects can help alleviate dryness, irritation, and dandruff-associated discomfort, indirectly supporting healthy hair growth [73]. Fenugreek mucilage further contributes film-forming and conditioning properties that improve hair fiber manageability, while its antimicrobial activity may support scalp microbiome balance and scalp health [56,74]. Despite these promising findings, well-designed clinical studies are still required to substantiate specific hair growth claims in humans. Future research should prioritize standardized topical formulations and objective endpoints, including phototrichograms, anagen/telogen ratios, and direct assessments of dermal papilla cell proliferation using in vitro or ex vivo follicle models.
5. Cosmetic Formulation Opportunities
The translation of T. foenum-graecum L. from a bioactive botanical to a cosmetic ingredient has begun to be addressed in the scientific literature, although the number of studies explicitly formulating fenugreek into cosmetic products remains limited. One of the most direct demonstrations of cosmetic relevance is provided by Eaknai et al. [8], who formulated an ethanolic fenugreek seed extract into a topical system and further developed a nanoencapsulated delivery approach to enhance stability and performance. In this study, the formulated extract retained strong collagenase inhibitory activity and protected human dermal fibroblasts from oxidative and inflammatory stress, supporting its suitability for anti-aging cosmetic applications rather than merely pharmacological use [8].
In a more explicitly cosmetic-oriented study, Ben Abdennebi et al. [7] optimized ultrasound-assisted extraction of fenugreek seeds specifically for cosmetic application, with formulation compatibility and antioxidant performance as explicit objectives. The resulting phenolic-rich extract was successfully integrated into a model cosmetic cream, retaining physicochemical stability (pH ~6.2–6.4, homogeneous texture, no phase separation after accelerated stability testing) and strong radical-scavenging performance, thereby providing practical proof-of-concept for fenugreek as a functional cosmetic raw material [7]. Beyond extract-based actives, fenugreek seeds are a rich source of galactomannan mucilage, whose physicochemical and functional properties have been extensively characterized. Although most galactomannan studies are conducted in food or pharmaceutical contexts, their relevance to cosmetics lies in documented hydrogel formation, film-forming ability, water retention, and rheological modification, properties that are directly transferable to cosmetic emulsions, gels, and masks. Structural and functional analyses reported by Nalbantova et al. [31] and Rashid et al. [29] demonstrate that fenugreek galactomannan exhibits strong hydration, viscosity, and stability, supporting its use as a natural thickener or moisturizing matrix in topical systems, even if cosmetic products were not explicitly formulated in these studies [29,31]. Taken together, the available literature indicates that fenugreek has already been successfully incorporated into cosmetic formulations in a limited but growing number of studies, while additional work on its polysaccharide fraction provides robust functional justification for its broader cosmetic application. However, the field would benefit from more formulation-focused and clinical cosmetic studies to fully substantiate performance, stability, and consumer-relevant outcomes.
Practical formulation considerations are essential to translate these promising bioactives into stable, consumer-acceptable products. Stability optimization includes maintaining a pH range of approximately 4.5–6.5 (commonly ~6.2–6.4 in tested prototypes) to preserve sensitive phenolic compounds and galactomannan integrity while preventing microbial growth or hydrolysis; incorporation of natural antioxidants such as tocopherol or ascorbic acid derivatives helps mitigate oxidation of polyphenols during storage. For mucilage and polysaccharide fractions, processing should respect thermal windows of ~60–140 °C to preserve hydration capacity and avoid backbone degradation (as indicated by DSC/TGA profiles showing moisture loss ~60–135 °C and polymer degradation above ~140–150 °C) [29,30,66]. Sensory challenges, notably the characteristic fenugreek odor, can be effectively addressed through micro or nanoencapsulation techniques [8] or by strategic blending with masking essential oils or fragrance compounds (e.g., rosemary, lavender, citrus, or vanilla accords). Advanced delivery systems such as liposomes, nanoemulsions, or hybrid carriers further enhance skin penetration and bioavailability of key lipophilic actives like diosgenin and trigonelline, thereby improving anti-aging efficacy while maintaining elegant sensorial profiles in creams, serums, gels, scalp tonics, and hair masks. Taken together, the emerging literature shows that fenugreek derivatives can be successfully incorporated into cosmetic formulations with retained bioactivity and acceptable stability. However, the field would greatly benefit from additional formulation-focused studies addressing long-term stability (e.g., 3–6 months at 40 °C/75% RH), consumer sensory panels, scale-up feasibility, and comparative performance against benchmark actives to fully unlock fenugreek’s potential in next-generation skin and hair care products.
6. Safety and Toxicity
In vitro safety screens available for fenugreek include human keratinocytes (HaCaT) and human dermal fibroblasts. Eaknai et al. [8] evaluated an ethanolic fenugreek extract and a nanoencapsulated system in skin-relevant models, including HaCaT keratinocytes and human dermal fibroblasts, and reported concentration-dependent cytotoxicity testing alongside efficacy endpoints in a co-cultured skin model [8]. Independent cytotoxicity profiling by Stefanowicz-Hajduk et al. [75] assessed standardized fenugreek extracts/fractions and individual metabolites using HaCaT keratinocytes (among other lines), providing quantitative cytotoxicity metrics useful for setting non-cytotoxic in-use ranges during formulation development [75].
Clinical safety data specifically for topical fenugreek cosmetics remains limited, but a controlled human study is available: Yusharyahya et al. (2021) investigated a fenugreek cream in a randomized, double-blind controlled trial design reporting clinical monitoring consistent with cosmetic tolerability evaluation in the context of facial anti-aging assessment [76]. In parallel, clinicians have highlighted that fenugreek can act as an emerging allergen in sensitized individuals (mainly food/spice exposure), which is relevant to cosmetic risk management (e.g., labeling, patch testing strategy, and avoidance for known legume/spice-allergic users) [77].
In addition to in vitro cytotoxicity data in HaCaT keratinocytes and dermal fibroblasts [8] and limited clinical tolerability [78], fenugreek presents an emerging allergenic risk, particularly in sensitized individuals. Recent reviews highlight rare but documented cases of Type IV delayed contact dermatitis following topical application of fenugreek pastes or preparations, as well as IgE-mediated reactions (urticaria, angioedema) more commonly from ingestion or inhalation [77]. Thus, cross-reactivity with other Fabaceae (e.g., peanuts, chickpeas) has been reported, warranting caution in legume-allergic users. For cosmetic risk management, recommend patch testing in sensitive populations, clear INCI labeling (e.g., “Trigonella foenum-graecum Seed Extract”), and avoidance claims for known spice/legume-allergic consumers [79].
7. Cosmetic Regulatory Position
Under EU law, fenugreek ingredients are regulated as cosmetic ingredients within the framework of Regulation (EC) No 1223/2009 [80], which requires a pre-market Cosmetic Product Safety Report (CPSR) and compliance with safety, labeling, and claims rules. Fenugreek seed extract is listed in the European Commission’s CosIng database as an INCI ingredient with functions such as skin conditioning and perfuming, supporting its recognized cosmetic-use positioning (not an authorization of safety by itself, but a harmonized reference for ingredient identity/function) [78].
In the EU, “authorized claims” are not granted ingredient-by-ingredient; instead, claims are permitted if they comply with Article 20 of 1223/2009 and the common criteria in Regulation (EU) 655/2013 (legal compliance, truthfulness, evidential support, honesty, fairness, informed decision-making) [81]. For fenugreek-based products, claims typically need to remain in the cosmetic domain (e.g., “helps moisturize,” “helps soothe,” “helps improve the appearance of fine lines,” “helps reduce the appearance of redness,” “scalp comfort,” “hair feels stronger/softer”), supported by appropriate evidence (in vitro/ex vivo/clinical, depending on the strength of the claim), and must avoid medicinal/therapeutic disease-treatment wording [82].
For claim substantiation under Regulation (EU) 655/2013, evidence must support cosmetic wording, with the level of substantiation proportionate to the claim. Examples of compliant claims for fenugreek-based products include: “helps moisturize and soften skin” (supported by mucilage/galactomannan film-forming, high swelling index, and hydration data); “helps reduce the appearance of fine lines and improve skin elasticity” (via in vitro collagenase inhibition and emerging clinical wrinkle/TEWL reductions [9]; “contributes to scalp comfort and healthier-looking hair” (via soothing, anti-inflammatory, and antimicrobial effects on scalp microbiome). Avoid therapeutic or disease-related claims (e.g., “treats hair loss,” “cures aging,” “prevents alopecia,” or “reduces dandruff as a medical condition”), as these fall outside the cosmetic scope and could trigger medicinal product classification. Product-specific dossiers with in vitro, ex vivo, consumer perception tests, and/or clinical evidence are essential to demonstrate compliance with the common criteria, particularly evidential support and truthfulness. Guidance from the Technical Document on Cosmetic Claims further emphasizes that claims must reflect the perception of the average consumer and avoid exaggeration [83].
8. Research Gaps and Future Directions
Despite the growing body of preclinical and formulation-oriented research on Trigonella foenum-graecum L., several critical gaps remain before its full potential as a cosmetic and cosmeceutical ingredient can be realized. One of the most significant limitations is the scarcity of robust clinical evidence. Although in vitro and ex vivo studies consistently demonstrate antioxidant, anti-inflammatory, antimicrobial, and anti-aging related activities, well-designed human clinical trials remain limited as of 2023–2024, particularly for topical cosmetic applications. Future studies should prioritize randomized, controlled clinical trials evaluating skin hydration, barrier function (e.g., TEWL), wrinkle appearance, pigmentation, scalp condition, and hair parameters to substantiate cosmetic claims under real-use conditions [7,8]. Although ultrasound and microwave-assisted extraction methods have been shown to improve yield and preserve bioactivity, further work is required to optimize scalability, solvent systems, and energy efficiency, while ensuring batch-to-batch reproducibility for cosmetic manufacturing [7,66]. A second major research gap concerns the standardization of fenugreek extracts. The phytochemical composition of fenugreek varies significantly depending on geographic origin, cultivar and processing conditions, which can affect batch-to-batch consistency. To address this, validated marker compounds should be established, such as or diosgenin-linked saponins [25]. Reliable quantification can be achieved using RP-HPLC-UV/MS for trigonelline and HPLC-ELSD or HPTLC for saponins [25]. Such standardization is essential to guarantee reproducible bioactivity, consistent product performance and robust claim substantiation in cosmetic applications [23]. Furthermore, integrating advanced omics approaches particularly metabolomics for comprehensive extract fingerprinting and proteomics for elucidating skin-relevant interaction pathways would significantly strengthen mechanistic understanding and translational potential [83].
In addition, fenugreek-derived peptides and glycoproteins remain largely unexplored in skin and hair research. Given the growing importance of bioactive peptides in barrier repair, signaling, and regeneration, targeted studies on these higher-molecular-weight fractions could open new formulation opportunities [31]. Finally, the application of omics-based approaches, particularly metabolomics and proteomics, represents a promising direction to comprehensively characterize fenugreek extracts, elucidate skin-relevant molecular pathways, and better understand interactions with the cutaneous microbiome and dermal cells [83]. Such integrative strategies will be critical to strengthening the mechanistic and translational foundation of fenugreek-based cosmetic innovation.
9. Conclusions
Collectively, the available evidence positions Trigonella foenum-graecum L. as a multifunctional botanical ingredient with significant potential for cosmetic and cosmeceutical applications. Fenugreek’s rich phytochemical composition encompassing polyphenols and flavonoids, steroidal saponins, alkaloids such as trigonelline, and mucilage-rich galactomannans provides a strong mechanistic basis for its reported antioxidant, anti-inflammatory, antimicrobial, moisturizing, anti-aging, and hair-supporting effects. These activities target key biological processes involved in skin aging, barrier dysfunction, inflammatory conditions, scalp imbalance, and hair fiber degradation. Importantly, recent formulation-oriented studies demonstrate that fenugreek extracts can be successfully incorporated into topical cosmetic systems while retaining bioactivity, particularly in anti-aging and antioxidant contexts. In parallel, extensive physicochemical characterization of fenugreek galactomannan supports its dual role as both a functional excipient and a skin-conditioning agent, aligning well with current clean beauty and sustainability-driven formulation trends. Available in vitro and limited clinical safety data suggest an overall favorable safety profile when appropriately formulated, although allergenic potential in sensitized individuals warrants careful risk assessment and labeling. Despite these strengths, fenugreek remains insufficiently validated from a cosmetic science perspective. The field would benefit from robust, well-controlled clinical studies, improved extract standardization, and deeper exploration of under-investigated fractions such as peptides and glycoproteins. Integrating advanced omics approaches could further clarify skin- and scalp-relevant mechanisms and support evidence-based claim substantiation. With such advances, fenugreek has the potential to evolve from a traditionally valued medicinal plant into a scientifically grounded, high-value ingredient for next-generation skin and hair care products.
Author Contributions
Conceptualization, I.B.R., O.M. and A.B.A.; methodology, I.B.R., E.C. and O.M.; software, I.B.R. and M.H.; validation, I.B.R., E.C., W.Y., O.M. and M.S.T.; formal analysis, I.B.R., A.B.A. and M.H., investigation, I.B.R., A.B.A., W.Y., M.H. and O.M.; resources, I.B.R.; data curation, I.B.R.; writing—original draft preparation, I.B.R. and A.B.A.; writing—review and editing I.B.R., A.B.A., O.M. and M.S.T.; visualization, I.B.R.; supervision, M.S.T.; project administration, I.B.R., M.S.T.; funding acquisition, I.B.R., O.M. and M.S.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding authors.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ECM | Extracellular Matrix |
| AGEs | Advanced Glycation End-products |
| TEWL | Transepidermal Water Loss |
| ROS | Reactive Oxygen Species |
| MMPs | Matrix Metalloproteinases |
| INCI | International Nomenclature of Cosmetic Ingredients |
| CPSR | Cosmetic Product Safety Report |
| HPLC | High-Performance Liquid Chromatography |
| PDA/DAD | Photodiode Array Detection |
| ESI-MS | Electrospray Ionization Mass Spectrometry |
| LC–MS(/MS) | Liquid Chromatography–Mass Spectrometry (tandem MS) |
| UPLC-MS | Ultra-Performance Liquid Chromatography–Mass Spectrometry |
| ELSD | Evaporative Light Scattering Detection |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| FWEP | Fenugreek Water-Extracted Polysaccharide |
| IC50 | Half-Maximal Inhibitory Concentration |
| HaCaT | Human immortalized keratinocytes |
References
- Liu, J.-K. Natural Products in Cosmetics. Nat. Prod. Bioprospect. 2022, 12, 40. [Google Scholar] [CrossRef]
- Michalak, M. Plant Extracts as Skin Care and Therapeutic Agents. Int. J. Mol. Sci. 2023, 24, 15444. [Google Scholar] [CrossRef] [PubMed]
- Giménez Martínez, R.J.; Rivas García, F.; March Cerdá, J.C.; Hernández-Ruíz, Á.; González Castro, M.I.; Valverde-Merino, M.-I.; Huertas Camarasa, F.J.; Lloris Meseguer, F.; López-Viota Gallardo, M. Bioactive Substances and Skin Health: An Integrative Review from a Pharmacy and Nutrition Perspective. Pharmaceuticals 2025, 18, 373. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, A.; Selvan, S.A.; Patial, P.K.; Pal, T. Plant-Based Ingredients in Cosmetic Science: Current Applications, Limitations, and Prospects. Int. J. Cosmet. Sci. 2025, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Visuvanathan, T.; Than, L.T.L.; Stanslas, J.; Chew, S.Y.; Vellasamy, S. Revisiting Trigonella foenum-graecum L.: Pharmacology and Therapeutic Potentialities. Plants 2022, 11, 1450. [Google Scholar] [CrossRef]
- Ruwali, P.; Pandey, N.; Jindal, K.; Singh, R.V. Fenugreek (Trigonella foenum-graecum): Nutraceutical Values, Phytochemical, Ethnomedicinal and Pharmacological Overview. S. Afr. J. Bot. 2022, 151, 423–431. [Google Scholar] [CrossRef]
- Ben Abdennebi, A.; Chaabani, E.; Bourgou, S.; Hammami, M.; Tounsi, M.S.; Merah, O.; Bettaieb Rebey, I. Fenugreek as a Potential Active Ingredient for the Development of Innovative Cosmetic Formulation. Cosmetics 2025, 12, 21. [Google Scholar] [CrossRef]
- Eaknai, W.; Bunwatcharaphansakun, P.; Phungbun, C.; Jantimaporn, A.; Chaisri, S.; Boonrungsiman, S.; Nimmannit, U.; Khongkow, M. Ethanolic Fenugreek Extract: Its Molecular Mechanisms against Skin Aging and the Enhanced Functions by Nanoencapsulation. Pharmaceuticals 2022, 15, 254. [Google Scholar] [CrossRef]
- Thakurdesai, P.; Deshpande, P.; Nimse, S.; Nimse, S. Anti-Aging Efficacy and Safety of Topical Application of Two Standardized Fenugreek Seed Extracts on Facial Skin in Women: Randomized, Double-Blind, Placebo-Controlled, Clinical Study. Pharmacogn. J. 2025, 17, 470–479. [Google Scholar] [CrossRef]
- Faisal, Z.; Irfan, R.; Akram, N.; Manzoor, H.M.I.; Aabdi, M.A.; Anwar, M.J.; Khawar, S.; Saif, A.; Shah, Y.A.; Afzaal, M.; et al. The Multifaceted Potential of Fenugreek Seeds: From Health Benefits to Food and Nanotechnology Applications. Food Sci. Nutr. 2024, 12, 2294–2310. [Google Scholar] [CrossRef]
- Singh, N.; Yadav, S.S.; Kumar, S.; Narashiman, B. Ethnopharmacological, Phytochemical and Clinical Studies on Fenugreek (Trigonella foenum-graecum L.). Food Biosci. 2022, 46, 101546. [Google Scholar] [CrossRef]
- Shahrajabian, M.H.; Sun, W.; Magadlela, A.; Hong, S.; Cheng, Q. Fenugreek Cultivation in the Middle East and Other Parts of the World with Emphasis on Historical Aspects and Its Uses in Traditional Medicine and Modern Pharmaceutical Science. In Fenugreek: Biology and Applications; Naeem, M., Aftab, T., Khan, M.M.A., Eds.; Springer: Singapore, 2021; pp. 13–30. [Google Scholar]
- Kartal, S.; Baturay, Z.; Efe, L. Effect of Planting Times on the Yield and Quality of Fenugreek (Trigonella foenum-graecum L.) Under the Conditions of Kahramanmaraş. Turk. J. Agric. Food Sci. Technol. 2023, 11, 296–302. [Google Scholar] [CrossRef]
- Bal, S.; Sharangi, A.B.; Saeed, M.; Alkhathami, A.; Siddiqui, S.; Alabdallah, N.M. Advances in Fenugreek Breeding: Novel Genetic and Omic Approaches. Front. Plant Sci. 2025, 16, 1674013. [Google Scholar] [CrossRef]
- Varshney, H.; Siddique, Y. Medicinal Properties of Fenugreek: A Review. Open Biol. J. 2023, 11, e187503622302220. [Google Scholar] [CrossRef]
- Hina, S.; Mustafa, S.; Ismail, A.M.; Mahmood, S.; Matar, A.; Alharbi, A.S.; Saleh, H.A.F.; Ge, W. Therapeutic and Protective Valuation of Fenugreek (Trigonella foenum-graecum). Ital. J. Food Sci. 2025, 37, 1–17. [Google Scholar] [CrossRef]
- Bouissane, L.; Elfardi, Y.; Khatib, S.; Fatimi, A.; Pereira, C.; Cruz-Martins, N. Medicinal Plants and Their Derivatives for Skin and Hair: A Mediterranean Perspective of Women Care. Arch. Dermatol. Res. 2025, 317, 710. [Google Scholar] [CrossRef] [PubMed]
- Kenny, O.; Smyth, T.J.; Hewage, C.M.; Brunton, N.P. Antioxidant Properties and Quantitative UPLC-MS Analysis of Phenolic Compounds from Extracts of Fenugreek (Trigonella foenum-graecum) Seeds and Bitter Melon (Momordica charantia) Fruit. Food Chem. 2013, 141, 4295–4302. [Google Scholar] [CrossRef]
- Benayad, Z.; Gómez-Cordovés, C.; Es-Safi, N.E. Characterization of Flavonoid Glycosides from Fenugreek (Trigonella foenum-graecum) Crude Seeds by HPLC–DAD–ESI/MS Analysis. Int. J. Mol. Sci. 2014, 15, 20668–20685. [Google Scholar] [CrossRef]
- Alu’datt, M.H.; Rababah, T.; Al-ali, S.; Tranchant, C.C.; Gammoh, S.; Alrosan, M.; Kubow, S.; Tan, T.-C.; Ghatasheh, S. Current Perspectives on Fenugreek Bioactive Compounds and Their Potential Impact on Human Health: A Review of Recent Insights into Functional Foods and Other High Value Applications. J. Food Sci. 2024, 89, 1835–1864. [Google Scholar] [CrossRef]
- Khenifi, M.L.; Serseg, T.; Migas, P.; Krauze-Baranowska, M.; Özdemir, S.; Bensouici, C.; Alghonaim, M.I.; Al-Khafaji, K.; Alsalamah, S.A.; Boudjeniba, M.; et al. HPLC-DAD-MS Characterization, Antioxidant Activity, α-Amylase Inhibition, Molecular Docking, and ADMET of Flavonoids from Fenugreek Seeds. Molecules 2023, 28, 7798. [Google Scholar] [CrossRef]
- Salam, S.G.A.; Rashed, M.M.; Ibrahim, N.A.; Rahim, E.A.A.; Aly, T.A.A.; AL-Farga, A. Phytochemical Screening and In-Vitro Biological Properties of Unprocessed and Household Processed Fenugreek (Trigonella foenum-graecum Linn.) Seeds and Leaves. Sci. Rep. 2023, 13, 7032. [Google Scholar] [CrossRef]
- Farag, M.A.; Rasheed, D.M.; Kropf, M.; Heiss, A.G. Metabolite Profiling in Trigonella Seeds via UPLC-MS and GC-MS Analyzed Using Multivariate Data Analyses. Anal. Bioanal. Chem. 2016, 408, 8065–8078. [Google Scholar] [CrossRef]
- Keskes, H.; Belhadj, S.; Jlail, L.; El Feki, A.; Sayadi, S.; Allouche, N. LC–MS–MS and GC–MS Analyses of Biologically Active Extracts of Tunisian Fenugreek (Trigonella foenum-graecum L.) Seeds. Food Meas. 2018, 12, 209–220. [Google Scholar] [CrossRef]
- Król-Kogus, B.; Głód, D.; Krauze-Baranowska, M. Qualitative and Quantitative HPLC-ELSD-ESI-MS Analysis of Steroidal Saponins in Fenugreek Seed. Acta Pharm. 2020, 70, 89–99. [Google Scholar] [CrossRef]
- Petit, P.R.; Sauvaire, Y.D.; Hillaire-Buys, D.M.; Leconte, O.M.; Baissac, Y.G.; Ponsin, G.R.; Ribes, G.R. Steroid Saponins from Fenugreek Seeds: Extraction, Purification, and Pharmacological Investigation on Feeding Behavior and Plasma Cholesterol. Steroids 1995, 60, 674–680. [Google Scholar] [CrossRef]
- Akbari, S.; Abdurahman, N.H.; Yunus, R.M. Optimization of Saponins, Phenolics, and Antioxidants Extracted from Fenugreek Seeds Using Microwave-Assisted Extraction and Response Surface Methodology as an Optimizing Tool. Comptes Rendus Chim. 2019, 22, 714–727. [Google Scholar] [CrossRef]
- Urooj, I.; Muhammad, G.; Irfan, T.; Suyama, T.L.; Hussain, M.A.; Amin, M. Galactomannan-Based Mucilage from Trigonella foenum-graecum L.: Structural Insights, Extraction Strategies, and Applications in Food and Sustainable Biomaterials. J. Agric. Food Chem. 2025, 73, 27855–27890. [Google Scholar] [CrossRef]
- Rashid, F.; Hussain, S.; Ahmed, Z. Extraction Purification and Characterization of Galactomannan from Fenugreek for Industrial Utilization. Carbohydr. Polym. 2018, 180, 88–95. [Google Scholar] [CrossRef]
- Kumar, R.; Patil, S.; Patil, M.; Patil, S.; Paschapur, M.; Parade, M. Isolation and Evaluation of Disintegrant Properties of Fenugreek Seed Mucilage. Int. J. PharmTech Res. 2009, 1, 982–996. [Google Scholar]
- Nalbantova, V.; Benbassat, N.; Delattre, C. Fenugreek Galactomannan and Its Versatile Applications. Polysaccharides 2024, 5, 478–492. [Google Scholar] [CrossRef]
- Ashihara, H.; Ludwig, I.A.; Katahira, R.; Yokota, T.; Fujimura, T.; Crozier, A. Trigonelline and Related Nicotinic Acid Metabolites: Occurrence, Biosynthesis, Taxonomic Considerations, and Their Roles in Planta and in Human Health. Phytochem. Rev. 2015, 14, 765–798. [Google Scholar] [CrossRef]
- Gonda, S.; Szűcs, Z.; Plaszkó, T.; Cziáky, Z.; Kiss-Szikszai, A.; Sinka, D.; Bácskay, I.; Vasas, G. Quality-Controlled LC-ESI-MS Food Metabolomics of Fenugreek (Trigonella foenum-graecum) Sprouts: Insights into Changes in Primary and Specialized Metabolites. Food Res. Int. 2023, 164, 112347. [Google Scholar] [CrossRef] [PubMed]
- Joshi, D.D. NMR Spectroscopy: Herbal Drugs and Fingerprints. In Herbal Drugs and Fingerprints: Evidence Based Herbal Drugs; Joshi, D.D., Ed.; Springer: New Delhi, India, 2012; pp. 163–186. [Google Scholar]
- Singh, P.; Bajpai, V.; Gond, V.; Kumar, A.; Tadigoppula, N.; Kumar, B. Determination of Bioactive Compounds of Fenugreek (Trigonella foenum-graecum) Seeds Using LC-MS Techniques. In Legume Genomics: Methods and Protocols; Jain, M., Garg, R., Eds.; Springer: New York, NY, USA, 2020; pp. 377–393. [Google Scholar]
- Papaccio, F.; D′Arino, A.; Caputo, S.; Bellei, B. Focus on the Contribution of Oxidative Stress in Skin Aging. Antioxidants 2022, 11, 1121. [Google Scholar] [CrossRef] [PubMed]
- Trüeb, R.M. Oxidative Stress in Ageing of Hair. Int. J. Trichology 2009, 1, 6. [Google Scholar] [CrossRef]
- Trüeb, R.M. Oxidative Stress and Its Impact on Skin, Scalp and Hair. Int. J. Cosmet. Sci. 2021, 43, S9–S13. [Google Scholar] [CrossRef]
- Al-Dabbagh, B.; Elhaty, I.A.; Al Hrout, A.; Al Sakkaf, R.; El-Awady, R.; Ashraf, S.S.; Amin, A. Antioxidant and Anticancer Activities of Trigonella foenum-graecum, Cassia acutifolia and Rhazya stricta. BMC Complement. Altern. Med. 2018, 18, 240. [Google Scholar] [CrossRef]
- Ktari, N.; Trabelsi, I.; Bardaa, S.; Triki, M.; Bkhairia, I.; Ben Slama-Ben Salem, R.; Nasri, M.; Ben Salah, R. Antioxidant and Hemolytic Activities, and Effects in Rat Cutaneous Wound Healing of a Novel Polysaccharide from Fenugreek (Trigonella foenum-graecum) Seeds. Int. J. Biol. Macromol. 2017, 95, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Hazel, A.; Moumen, O.; Ali, O.O.; Ouajed, H.; Mechrour, A. Exploring the Antioxidant Potential and HPLC Profile of Fenugreek (Trigonella foenum-graecum L.) Seed Extracts. N. Afr. J. Food Nutr. Res. 2025, 9, 203–212. [Google Scholar] [CrossRef]
- Ghevariya, H.; Vatukiya, V.; Mistry, N.; Jain, N. Comparative Evaluation of Bioactive Compounds and Antioxidant Properties of Fenugreek (Trigonella foenum-graecum L.) Seed, Stem, Leaf, and Microgreens. Third Concept 2023, 37, 62–75. [Google Scholar]
- Dhull, S.B.; Kaur, M.; Sandhu, K.S. Antioxidant Characterization and in Vitro DNA Damage Protection Potential of Some Indian Fenugreek (Trigonella foenum-graecum) Cultivars: Effect of Solvents. J. Food Sci. Technol. 2020, 57, 3457–3466. [Google Scholar] [CrossRef]
- Akbari, S.; Abdurahman, N.H.; Yunus, R.M.; Alara, O.R.; Abayomi, O.O. Extraction, Characterization and Antioxidant Activity of Fenugreek (Trigonella-foenum graecum) Seed Oil. Mater. Sci. Energy Technol. 2019, 2, 349–355. [Google Scholar] [CrossRef]
- Setti, K.; Kachouri, F.; Hamdi, M. Improvement of the Antioxidant Activity of Fenugreek Protein Isolates by Lactococcus Lactis Fermentation. Int. J. Pept. Res. Ther. 2018, 24, 499–509. [Google Scholar] [CrossRef]
- Priya, V.; Jananie, R.K.; Vijayalakshmi, K. Studies on Anti-Oxidant Activity of Trigonella foenum graecum Seed Using in Vitro Models. Int. J. Pharm. Sci. Res. 2011, 35, 2704–2708. [Google Scholar] [CrossRef]
- Guimarães, G.R.; Almeida, P.P.; de Oliveira Santos, L.; Rodrigues, L.P.; de Carvalho, J.L.; Boroni, M. Hallmarks of Aging in Macrophages: Consequences to Skin Inflammaging. Cells 2021, 10, 1323. [Google Scholar] [CrossRef]
- Fatima, H.; Shahid, M.; Pruitt, C.; Pung, M.A.; Mills, P.J.; Riaz, M.; Ashraf, R. Chemical Fingerprinting, Antioxidant, and Anti-Inflammatory Potential of Hydroethanolic Extract of Trigonella foenum-graecum. Antioxidants 2022, 11, 364. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Cai, Y.-S.; Yuan, R.; Wan, Q.; Xiao, D.; Lei, J.; Yu, J. Bioactive Pterocarpans from Trigonella foenum-graecum L. Food Chem. 2020, 313, 126092. [Google Scholar] [CrossRef]
- Alsuliam, S.M.; Albadr, N.A.; Almaiman, S.A.; Al-Khalifah, A.S.; Alkhaldy, N.S.; Alshammari, G.M. Fenugreek Seed Galactomannan Aqueous and Extract Protects against Diabetic Nephropathy and Liver Damage by Targeting NF-κB and Keap1/Nrf2 Axis. Toxics 2022, 10, 362. [Google Scholar] [CrossRef]
- Huang, H.; Wang, X.; Yang, L.; He, W.; Meng, T.; Zheng, K.; Xia, X.; Zhou, Y.; He, J.; Liu, C.; et al. The Effects of Fenugreek Extract on Growth Performance, Serum Biochemical Indexes, Immunity and NF-κB Signaling Pathway in Broiler. Front. Vet. Sci. 2022, 9, 882754. [Google Scholar] [CrossRef]
- Sindhu, G.; Ratheesh, M.; Shyni, G.L.; Nambisan, B.; Helen, A. Anti-Inflammatory and Antioxidative Effects of Mucilage of Trigonella foenum graecum (Fenugreek) on Adjuvant Induced Arthritic Rats. Int. Immunopharmacol. 2012, 12, 205–211. [Google Scholar] [CrossRef]
- Bordia, A.; Verma, S.K.; Srivastava, K.C. Effect of Ginger (Zingiber officinale Rosc.) and Fenugreek (Trigonella foenumgraecum L.) on Blood Lipids, Blood Sugar and Platelet Aggregation in Patients with Coronary Artery Disease. Prostaglandins Leukot. Essent. Fat. Acids 1997, 56, 379–384. [Google Scholar] [CrossRef]
- Al-Timimi, L.A.N. Antibacterial and Anticancer Activities of Fenugreek Seed Extract. Asian Pac. J. Cancer Prev. 2019, 20, 3771–3776. [Google Scholar] [CrossRef]
- Sethi, G.; Sood, S.; Bhardwaj, S.B.; Jain, A. In Vitro Evaluation of Anti-Microbial Efficacy of Trigonella foenum-graecum and Its Constituents on Oral Biofilms. J. Indian Soc. Periodontol. 2024, 28, 304–311. [Google Scholar] [CrossRef]
- Alenazy, R. Antimicrobial Activities and Biofilm Inhibition Properties of Trigonella foenum graecum Methanol Extracts against Multidrug-Resistant Staphylococcus Aureus and Escherichia Coli. Life 2023, 13, 703. [Google Scholar] [CrossRef]
- Alwan, A.M.; Jassim, I.M.; Jasim, G.M. Study of Antibacterial Activities of Seeds Extract of Fenugreek (Trigonella foenum-graecum). Diyala J. Med. 2017, 13, 63–67. [Google Scholar] [CrossRef]
- Ahmad, R.; Alqathama, A.; Al-Maimani, R.; Al-Said, H.M.; Ashgar, S.S.; Althubiti, M.; Jalal, N.A.; Khan, M.; Algarzai, M. Exploring the Role of Phytochemical Classes in the Biological Activities of Fenugreek (Trigonella feonum graecum): A Comprehensive Analysis Based on Statistical Evaluation. Foods 2025, 14, 933. [Google Scholar] [CrossRef]
- Abu-Zaid, A.A.; Al-Barty, A.; Morsy, K.; Hamdi, H. In Vitro Study of Antimicrobial Activity of Some Plant Seeds against Bacterial Strains Causing Food Poisoning Diseases. Braz. J. Biol. 2022, 82, e256409. [Google Scholar] [CrossRef]
- Walli, R.R.; Musrati, R.A.A.; Eshtewi, H.M.; Fathi, M. Sherif Screening of Antimicrobial Activity of Fenugreek Seeds. Pharm. Pharmacol. Int. J. 2015, 2, 122–124. [Google Scholar] [CrossRef]
- Haratake, A.; Watase, D.; Setoguchi, S.; Nagata-Akaho, N.; Matsunaga, K.; Takata, J. Effect of Orally Ingested Diosgenin into Diet on Skin Collagen Content in a Low Collagen Skin Mouse Model and Its Mechanism of Action. Life Sci. 2017, 174, 77–82. [Google Scholar] [CrossRef]
- Chowdhury, A.A.; Gawali, N.B.; Bulani, V.D.; Kothavade, P.S.; Mestry, S.N.; Deshpande, P.S.; Juvekar, A.R. In Vitro Antiglycating Effect and in Vivo Neuroprotective Activity of Trigonelline in D-Galactose Induced Cognitive Impairment. Pharmacol. Rep. 2018, 70, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.C.; Lima, T.F.O.; Arcaro, C.A.; Inacio, M.D.; Batista-Duharte, A.; Carlos, I.Z.; Spolidorio, L.C.; Assis, R.P.; Brunetti, I.L.; Baviera, A.M. Trigonelline and Curcumin Alone, but Not in Combination, Counteract Oxidative Stress and Inflammation and Increase Glycation Product Detoxification in the Liver and Kidney of Mice with High-Fat Diet-Induced Obesity. J. Nutr. Biochem. 2020, 76, 108303. [Google Scholar] [CrossRef]
- Maarouf, M.; Maarouf, C.L.; Yosipovitch, G.; Shi, V.Y. The Impact of Stress on Epidermal Barrier Function: An Evidence-based Review. Br. J. Dermatol. 2019, 181, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.; Zakaria, S.; Syed Jaafar, S.N. Enhanced Mechanical Properties of Hydrothermal Carbamated Cellulose Nanocomposite Film Reinforced with Graphene Oxide. Carbohydr. Polym. 2017, 172, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Niknam, R.; Mousavi, M.; Kiani, H. New Studies on the Galactomannan Extracted from Trigonella foenum-graecum (Fenugreek) Seed: Effect of Subsequent Use of Ultrasound and Microwave on the Physicochemical and Rheological Properties. Food Bioprocess Technol. 2020, 13, 882–900. [Google Scholar] [CrossRef]
- Yetiskin, B.; Okay, O. High-Strength and Self-Recoverable Silk Fibroin Cryogels with Anisotropic Swelling and Mechanical Properties. Int. J. Biol. Macromol. 2019, 122, 1279–1289. [Google Scholar] [CrossRef]
- Trüeb, R.M. Value of Nutrition-Based Therapies for Hair Growth, Color, and Quality. In Nutrition for Healthy Hair: Guide to Understanding and Proper Practice; Trüeb, R.M., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 225–255. [Google Scholar]
- Semwal, P.; Painuli, S.; Abu-Izneid, T.; Rauf, A.; Sharma, A.; Daştan, S.D.; Kumar, M.; Alshehri, M.M.; Taheri, Y.; Das, R.; et al. Diosgenin: An Updated Pharmacological Review and Therapeutic Perspectives. Oxid. Med. Cell. Longev. 2022, 2022, 1035441. [Google Scholar] [CrossRef]
- Xiao, L.; Zhang, X.; Chen, Z.; Li, B.; Li, L. A Timosaponin B-II Containing Scalp Care Solution for Improvement of Scalp Hydration, Dandruff Reduction, and Hair Loss Prevention: A Comparative Study on Healthy Volunteers before and after Application. J. Cosmet. Dermatol. 2021, 20, 819–824. [Google Scholar] [CrossRef]
- Hajizadeh, M.; Azadbakht, M.; Asgarirad, H.; Rahmatpour Rokni, G.; Fakhri, M.; Mirzaee, F. Efficacy of a Gel from the Seed of Trigonella foenum-graceum L. in Preventing Trichoptilosis: A Randomized Placebo-Controlled Clinical Trial. J. Maz. Univ. Med. Sci. 2022, 32, 25–36. [Google Scholar]
- Lochhead, R.Y. A Review of Recent Advances in the Polymeric Delivery of Attributes in Cosmetics and Personal Care Products. In Polymeric Delivery of Therapeutics; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2010; Volume 1053, pp. 3–22. [Google Scholar]
- Kumari, K.U.; Yadav, N.P.; Luqman, S. Promising Essential Oils/Plant Extracts in the Prevention and Treatment of Dandruff Pathogenesis. Curr. Top. Med. Chem. 2022, 22, 1104–1133. [Google Scholar] [CrossRef]
- Sakthi, D. Effectiveness of Fenugreek Seed Paste on Dandruff among Adolescent Girls in Selected Women’s Hostel, Coimbatore. Int. J. Nurs. Educ. Res. 2014, 2, 147–150. [Google Scholar]
- Stefanowicz-Hajduk, J.; Król-Kogus, B.; Sparzak-Stefanowska, B.; Kimel, K.; Ochocka, J.R.; Krauze-Baranowska, M. Cytotoxic Activity of Standardized Extracts, a Fraction, and Individual Secondary Metabolites from Fenugreek Seeds against SKOV-3, HeLa and MOLT-4 Cell Lines. Pharm. Biol. 2021, 59, 422–435. [Google Scholar] [CrossRef] [PubMed]
- Yusharyahya, S.N.; Bramono, K.; Indriatmi, W.; Prasetyo, M.; Ascobat, P.; Hestiantoro, A.; Wiraguna, A.A.G.P. Anti-Aging Effects of Fenugreek Cream on Postmenopausal Skin: A Randomized Controlled Trial. J. Appl. Pharm. Sci. 2021, 11, 095–103. [Google Scholar] [CrossRef]
- Alessandrello, C.; Sanfilippo, S.; Gangemi, S.; Pioggia, G.; Minciullo, P.L. Fenugreek: New Therapeutic Resource or Emerging Allergen? Appl. Sci. 2024, 14, 9195. [Google Scholar] [CrossRef]
- CE CosIng—Cosmetics—GROWTH—European Commission. Available online: https://ec.europa.eu/growth/tools-databases/cosing/details/80358?utm_source=chatgpt.com (accessed on 14 December 2025).
- Bruusgaard-Mouritsen, M.A. Allergy to Natural Ingredients and Polyethylene Glycols in Cosmetic and Pharmaceutical Products. 2021. Available online: https://www.videncenterforallergi.dk/wp-content/uploads/files/ph.d-afhandlinger/phd-Bruusgaard-Mouritsen.pdf (accessed on 10 November 2025).
- European Union. CE Regulation (EC) No 1223/2009 of the European Parliament and of the Council. Off. J. Eur. Union 2009, L 342, 59. Available online: https://health.ec.europa.eu/system/files/2016-11/cosmetic_1223_2009_regulation_en_0.pdf (accessed on 10 November 2025).
- CE Commission Regulation (EU) No 655/2013 of 10 July 2013 Laying Down Common Criteria for the Justification of Claims Used in Relation to Cosmetic Products Text with EEA Relevance. 2013. Volume 190. Available online: https://www.legislation.gov.uk/eur/2013/655/introduction (accessed on 10 November 2025).
- CE Technical Document on Cosmetic Claims. Available online: https://ec.europa.eu/docsroom/documents/24847 (accessed on 14 December 2025).
- Huang, K.; Li, F.; Liu, Y.; Liang, B.; Qu, P.; Yang, L.; Han, S.; Li, W.; Mo, X.; Dong, L.; et al. Multi-Omics Analyses Reveal Interactions between the Skin Microbiota and Skin Metabolites in Atopic Dermatitis. Front. Microbiol. 2024, 15, 1349674. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.