Simple Summary
Cervical cancer treatment is often complicated by drug resistance, which limits the success of chemotherapy and contributes to poor patient outcomes. Medicinal plants have been investigated as potential therapeutic options to enhance drug responsiveness and reduce resistance. Recent preclinical evidence indicates that plant-derived compounds may improve treatment efficacy and provide alternative strategies for patients with limited treatment options. Although these approaches are promising, challenges such as poor bioavailability and limited clinical trial data remain. Addressing these gaps is essential for translating laboratory findings into meaningful clinical benefit.
Abstract
Drug resistance remains a significant challenge in cancer therapy, accounting for most relapses and contributing substantially to cancer-related mortality worldwide. Several molecular processes are linked to the development of resistance to anticancer drugs, with the most studied mechanisms including epigenetic changes, drug efflux, cell survival signalling pathways, and inactivation of anticancer drugs. Both intrinsic and acquired forms of resistance hinder tumour cell elimination, reducing treatment success. This translates to poorer patient outcomes and the need for more aggressive treatment regimens. Therefore, understanding these molecular processes is crucial for enhancing the efficacy of anticancer therapy. Medicinal plants offer potential to counter various resistance mechanisms through their diverse phytocompounds. These compounds may offer benefits including consistent availability, anticancer potency, few side effects, and minimal drug resistance. However, the bioavailability of these phytochemicals and the lack of extensive clinical trials remain key challenges. Therefore, this review provides in-depth information on the mechanisms that lead to drug resistance during cervical cancer therapy, the challenges related to phytochemical bioavailability, the current status, and future needs for clinical trials evaluating the application of medicinal plants to combat drug resistance in cancer cells.
1. Introduction
Cervical cancer is a prevalent gynaecological malignancy, forming inside the epithelium of the uterine cervix. An estimation of around 604,127 new cases and 341,831 fatalities attributed to the disease were reported in 2020, with nearly 90% of them arising in low- and middle-income countries. This high prevalence in these regions is largely driven by inadequate access to advanced healthcare services and late diagnosis, which contribute to increased mortality among underprivileged women [1,2]. Cervical cancer ranks fourth in global female cancer prevalence and is a leading cause of death [3]. Drug resistance is a major obstacle in cervical cancer management [4]. It drives relapse and contributes to rising mortality by reducing tumour sensitivity to antineoplastic drugs. As tumour cells acquire resistance, drug potency declines, leading to poor therapeutic responses and eventual treatment failure [5,6]. Drug resistance in cancer is remarkably similar to that of infectious illnesses, in that both complications are confronted by several rapidly expanding intrinsic and/or extrinsic factors [7].
The development of multidrug resistance is attributed to mutations and metabolic abnormalities that accumulate over time in tumours initially sensitive to chemotherapy [8]. Chemotherapy employs drugs, such as cisplatin, paclitaxel, and others, to inhibit the growth of malignant tumours and induce their eradication. The initial chemotherapeutic intervention in cervical cancer patients frequently demonstrates significant efficacy; however, as treatment progresses, drug resistance often develops in most patients, thereby constraining the overall effectiveness of the therapy [5,9].
Over the last few decades, medicinal plants have been extensively studied for cancer treatment, offering an alternative to conventional treatments with fewer adverse effects. Phytochemicals, which are secondary metabolites found in medicinal plants, may offer an alternative treatment strategy to combat drug resistance [10]. Moreover, they offer a viable therapy option for patients with multidrug resistance cancers by targeting multiple molecular processes, including blocking ABC transporters, sensitisation of cancer cells, induction of apoptosis, and modulation of pathways underlying multidrug resistance [11].
2. Methodology
2.1. Review Design
This study was conducted as a systematic review in accordance with the PRISMA 2020 guidelines to ensure transparency and reproducibility.
2.2. Search Strategy
A systematic literature search was conducted from the following databases: PubMed, Scopus and Google Scholar. The search strategy was designed to identify relevant studies published up to February 2025. The search strategy included the use of keywords such as ‘medicinal plants’, ‘cervical cancer’, ‘drug resistance’, ‘phytochemicals’, ‘anticancer drugs’ and ‘bioavailability’. The Boolean operators used included AND and OR to refine the search results. The reference lists of key articles were also manually screened to identify additional relevant studies.
2.3. Language Restrictions
Only articles published in English were considered for review. No lower limit was placed on the publication year, allowing for the inclusion of both foundational studies and recent advances.
2.4. Inclusion Criteria
Studies were included if they focused on the role of medicinal plants or their phytochemicals in overcoming drug resistance in cervical cancer, investigated the molecular mechanisms underlying drug resistance, and considered the potential of using medicinal plants to enhance tumour cell response to chemotherapy.
2.5. Exclusion Criteria
Studies that did not focus on drug resistance in cervical cancer were excluded. Furthermore, studies lacking sufficient methodological details or experimental evidence, as well as those that were case reports or opinions without original data, were excluded.
2.6. Screening and Selection
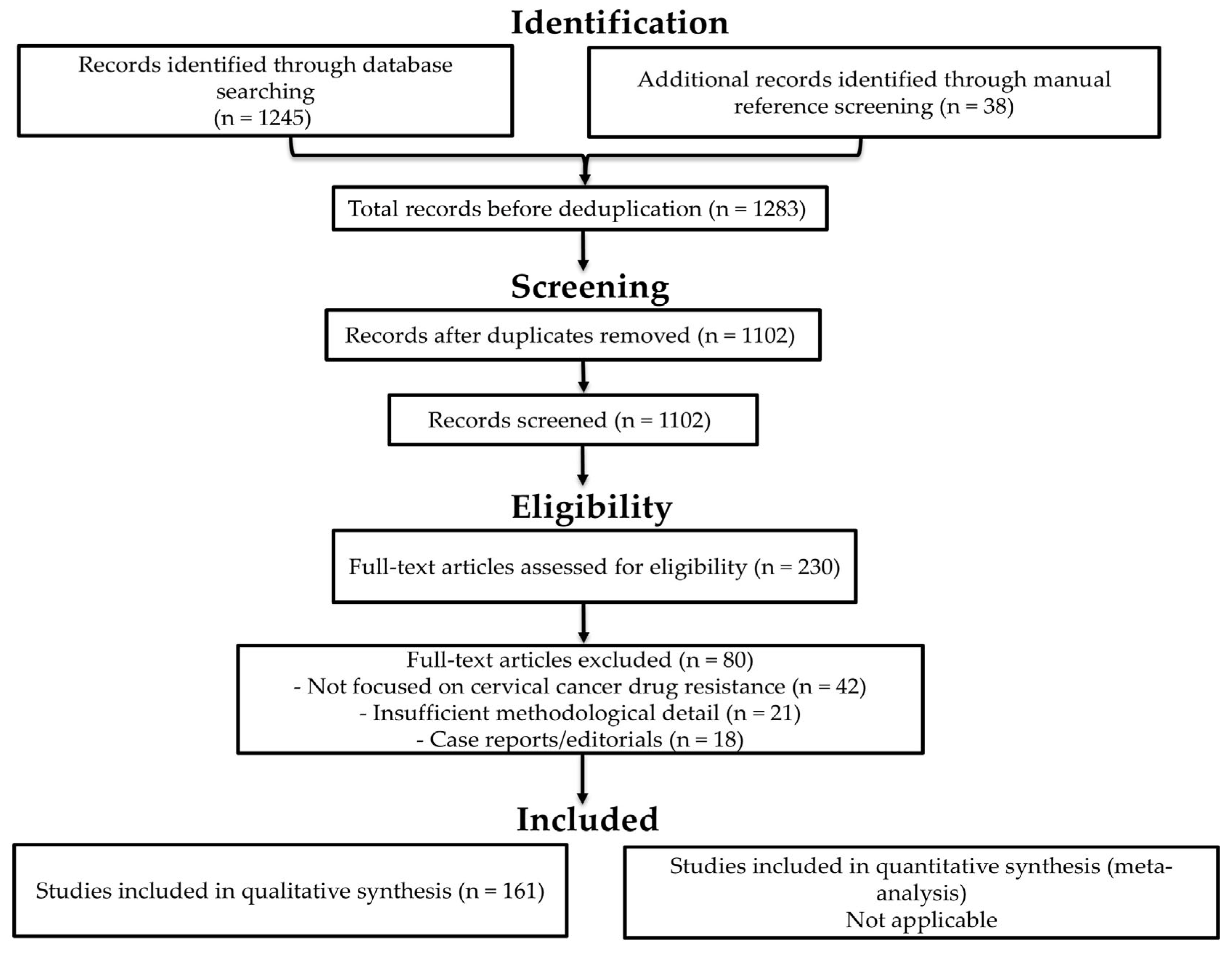
Titles and abstracts were screened by the lead author (T.P.M.), using predefined inclusion and exclusion criteria. Other co-authors subsequently cross-checked screening decisions to ensure consistency and accuracy. All retrieved records were compiled into a Microsoft Word document and sorted alphabetically by author’s name. Duplicates were identified manually and removed, with verification against publication year and title to ensure accuracy. Full-text articles were then retrieved and assessed for eligibility. Each study was evaluated for methodological detail, relevance to drug resistance mechanisms, and reporting quality. Studies lacking sufficient experimental or mechanistic data were excluded. Although primary screening was conducted by a single reviewer, cross-checking by co-authors provided an additional layer of verification to enhance consistency and reduce the likelihood of oversight. This approach was adopted due to resource constraints but was supported by standardised criteria and documentation to maintain methodological rigour. A PRISMA 2020 flow diagram summarising the screening and selection process is provided in Figure 1. This includes the number of records identified, screened, excluded, and included in the final synthesis.
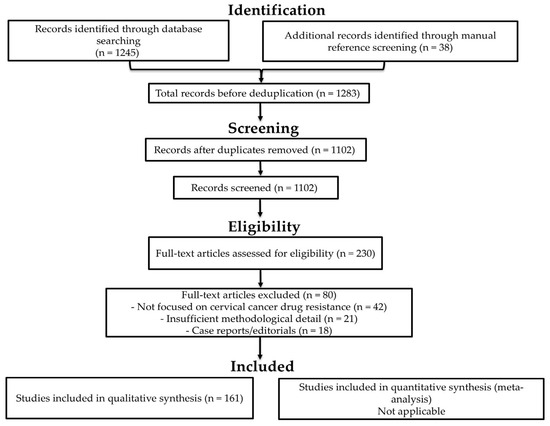
Figure 1.
PRISMA 2020 flow diagram illustrating the study selection process.
2.7. Data Extraction and Synthesis
Data extraction was performed using a structured template developed before the review. For each eligible study, information was collected on the study design, experimental model (in vitro, in vivo, or clinical), the medicinal plant or phytochemical investigated, and the specific mechanism of drug resistance addressed. Outcomes related to chemosensitisation, reversal of resistance, or enhancement of therapeutic efficacy were systematically recorded. Where available, details on dosage, bioavailability, and molecular pathways were noted to facilitate comparison across studies. Extraction was conducted independently by two reviewers to minimise bias, and discrepancies were resolved through discussion or consultation with a third reviewer. The extracted data were subsequently synthesised narratively, with findings grouped according to the resistance mechanism targeted (e.g., oxidative stress, epithelial–mesenchymal transition, epigenetic modulation) and mapped to the corresponding phytochemical interventions.
3. Processes Underlying Multidrug Resistance
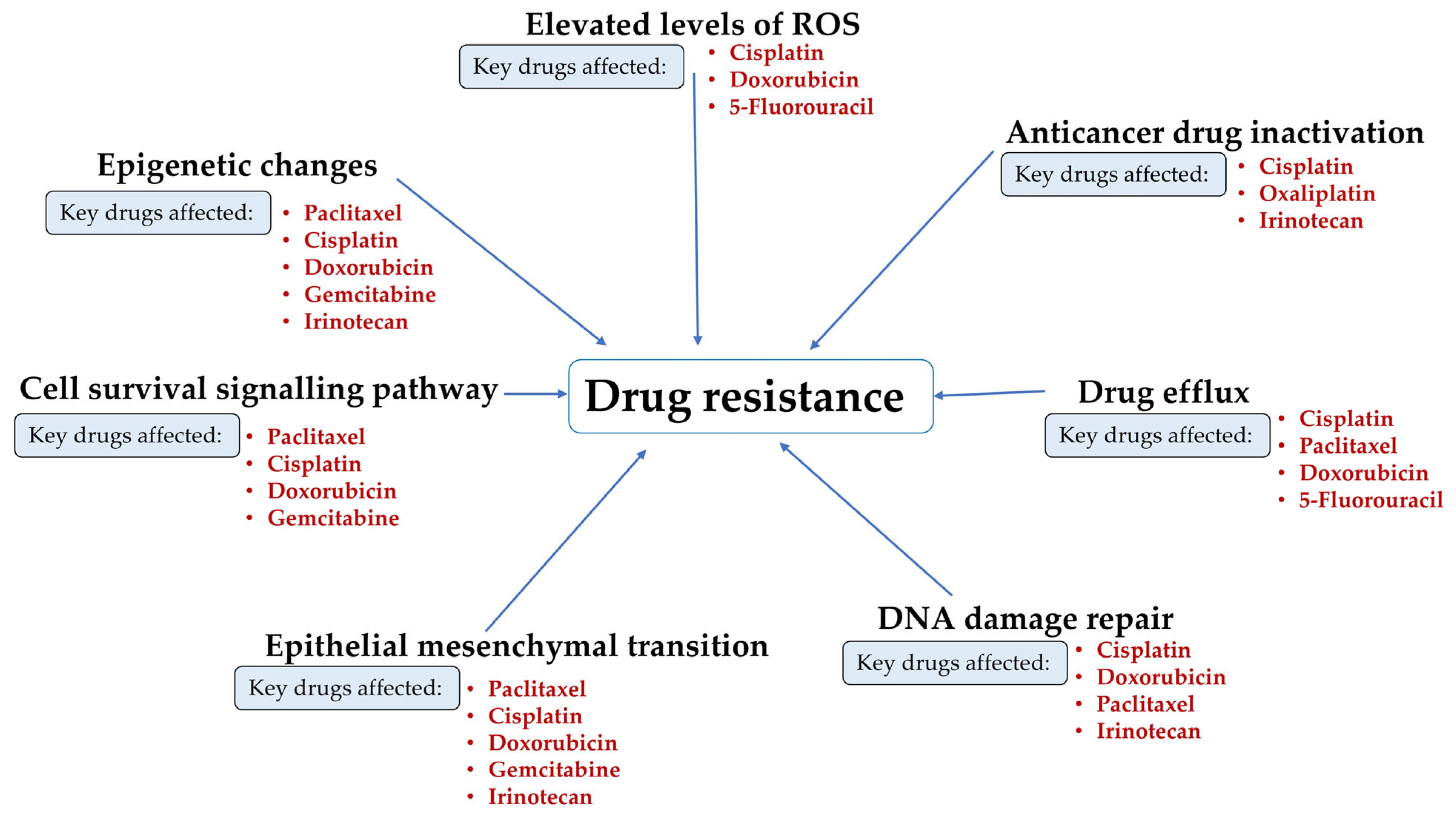
Due to unsuccessful therapies and advancement of the disease, drug resistance is accountable for approximately 90% of cancer-related fatalities, including those from cervical cancer [5]. Several mechanisms contribute to drug resistance, including drug efflux [12], epigenetic changes [13], elevated amounts of reactive oxygen species (ROS) [14], enhanced DNA damage repair mechanisms [15], epithelial–mesenchymal transition (EMT), anticancer drug inactivation [16], and cell survival signalling pathways [17]. Various anticancer drugs that are used in cervical cancer treatment, such as cisplatin, doxorubicin, 5-fluorouracil (5-FU), icaritin, vincristine, irinotecan, gemcitabine, and paclitaxel, are significantly impacted by each of these processes (Figure 2). This results in reduced effectiveness of the drug delivered, making cancer treatment more challenging [18].
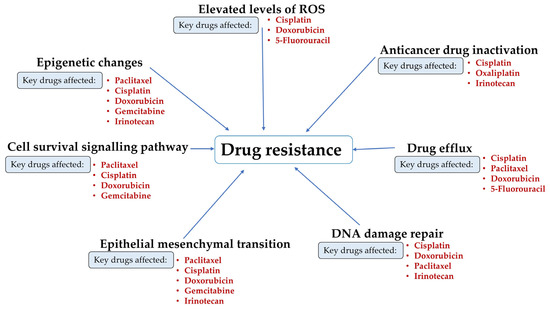
Figure 2.
Mechanisms driving drug resistance in cervical cancer. Processes such as ROS elevation, epigenetic changes, signalling dysregulation, EMT, enhanced DNA repair, drug efflux, and drug inactivation reduce the efficacy of drugs, including cisplatin, doxorubicin, 5-fluorouracil, icaritin, vincristine, irinotecan, gemcitabine, and paclitaxel.
Drug resistance can be categorised as either innate (intrinsic) or acquired, depending on the timing of its onset. Intrinsic resistance emerges in a cluster of cancerous cells before drug administration and reduces the effectiveness of the treatment. In contrast, acquired resistance develops when an anticancer agent loses its effectiveness following repeated drug administrations [5,19,20].
3.1. Intrinsic Drug Resistance
Some of the characteristics associated with intrinsic drug resistance include drug deterioration; altered drug receptors leading to unresponsiveness of tumour cells towards a specific treatment and signalling pathways, due to weak affinity of the drug to its molecular targets; and rapid progression of the tumour cells resulting from preceding mutations or other cellular activities associated with clusters of tumour cells [6,21]. Various mechanisms contribute to intrinsic drug resistance, such as enhanced cell survival signalling pathways, EMT, and elevated levels of ROS [22].
3.1.1. Evidence Scope
The majority of studies discussed in this section are based on in vitro or animal models. While these findings provide valuable mechanistic insights and identify potential therapeutic targets, they should be interpreted as preclinical evidence unless otherwise supported by clinical trial data.
3.1.2. ROS Induces Intrinsic Drug Resistance
ROS are oxygen-containing, unstable compounds such as superoxide (O2−*), hydrogen peroxide (H2O2) and hydroxyl radicals (HO*) [23]. Elevated levels of ROS in cancer cells drive DNA damage [24], genetic instability [25], and pro-tumourigenic signalling [26]. Aerobic respiration produces ROS largely via the electron transport chain in the mitochondria and ß-oxidation of fatty acids in the peroxisomes [27,28]. In some cancers, electron transport chain activation contributes to drug resistance. In cervical tumour cells, electron leakage occurring during oxidative phosphorylation triggers the production of ROS and other radicals, leading to oxidative DNA damage of the uterine epithelial cells [29]. Furthermore, elevated levels of ROS promote the triggering of cellular receptor tyrosine kinase (RTK) signalling, leading to upregulation of cell survival processes such as MAPK, PI3K/AKT, and JAK/STAT, which induce intrinsic drug resistance [30,31]. Additionally, CaSki cancer cells exposed to cisplatin activate EphA4 (Ephrin type-A receptor 4) receptor tyrosine kinase signalling via the production of reactive oxygen species, thus inducing chemoresistance [32]. Some of the ROS production is mediated by oxidoreductase enzymes such as lipoxygenase, cyclooxygenases (COXs), and NADPH oxidases (NOXs), as well as external stimuli such as UV radiation and host defence mechanisms [33].
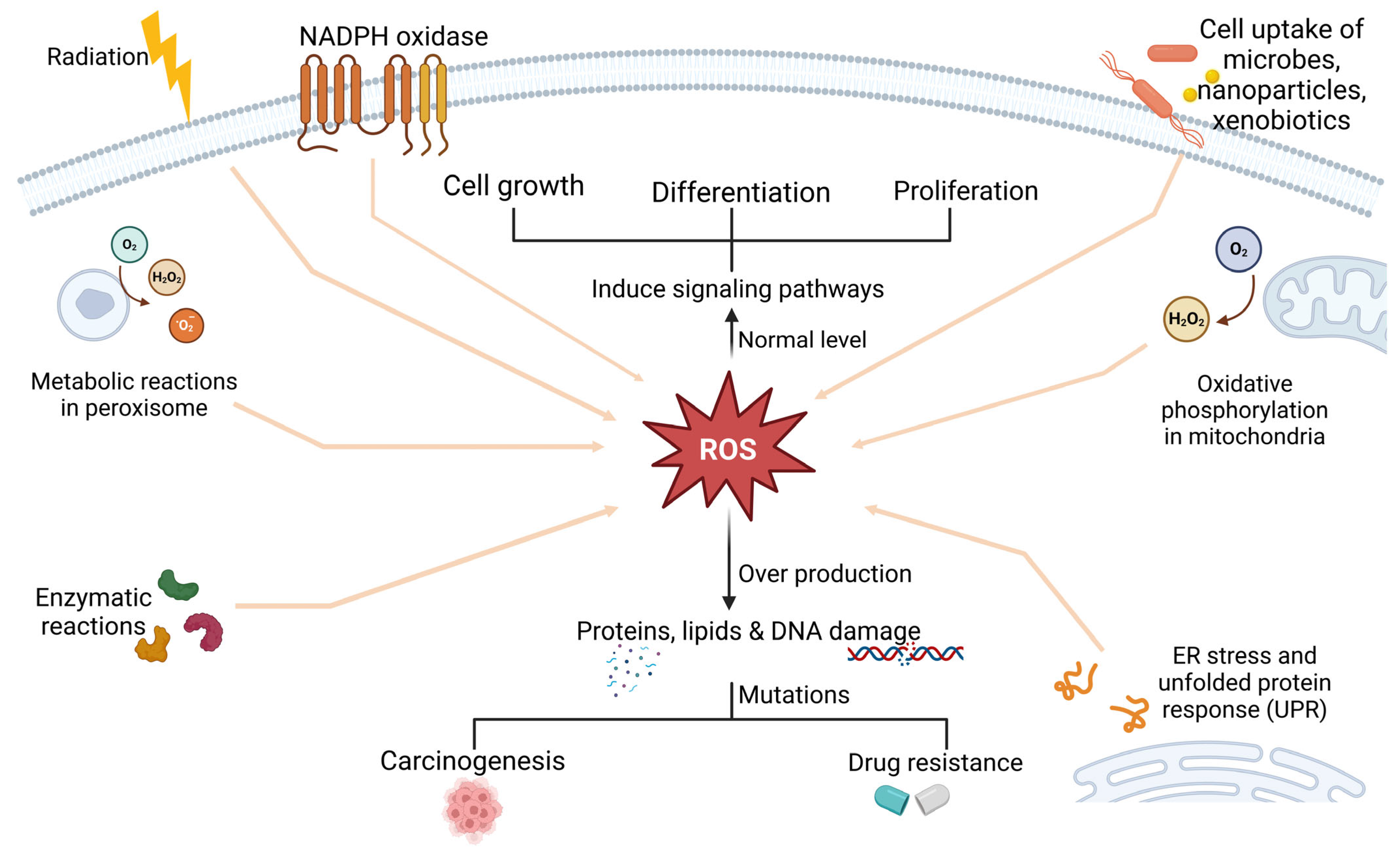
Minimal ROS levels modulate proliferation and differentiation in non-cancerous and cancerous cells. For instance, low-to-moderate amounts of ROS under physiological circumstances interact with macromolecules through reversible oxidative changes, and this process is essential for cell growth [34]. Elevated ROS levels are associated with anticancer drug resistance, as they promote tumourigenesis and metastasis during or after treatment. Cervical cancer is mainly attributed to infections caused by the human papillomavirus (HPV). The insertion of HPV into the cellular genome causes oncoprotein expression, which is linked to persistent inflammation and increased ROS production [35]. Elevated intracellular ROS can disrupt lipids, proteins, and DNA, thus causing mutations that increase carcinogenesis in non-cancerous cells or multidrug resistance in cancer cells, as demonstrated in Figure 3 [14,36]. The interaction of ROS with proteins such as Heat Shock Protein 70 (HSP70) has been the subject of several studies [37]. For instance, ROS can increase the expression of HSP70 to provide a cytoprotective effect. In non-cancerous cells, expression of HSP70 effectively induces their response to external stresses and prevents cell death; however, when HSP70 is overexpressed in tumour cells, resistance to treatment intensifies [38,39]. Most cancer cells may survive apoptosis and develop resistance to various chemotherapeutic treatments when they are exposed to intrinsic oxidative stress. A range of chemotherapy medications utilised for treating cervical cancer may be rendered much less effective if ROS levels are elevated [40]. One common chemotherapy drug for cervical cancer is cisplatin. Elevated amounts of ROS can promote cellular processes that repair DNA damage, which can lead to resistance and eventually reduce the efficacy of cisplatin [41]. One way that doxorubicin induces cell death is by generating ROS. Conversely, tumour cells can become resistant to high ROS environments by upregulating antioxidant defences or strengthening DNA repair mechanisms, among other strategies. These changes can greatly minimise the cytotoxic effects of doxorubicin. Another chemotherapy drug that is susceptible to changes in ROS levels is 5-FU. Elevated levels of ROS in cancer cells may improve repair systems and help the cells to withstand 5-FU treatment, leading to drug resistance [11,42].
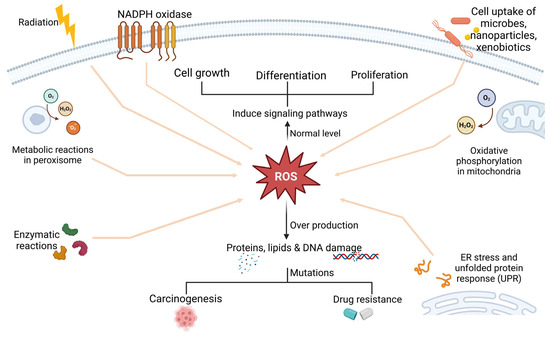
Figure 3.
Reactive oxygen species (ROS) arise from enzymatic reactions, peroxisomal metabolism, external stimuli such as radiation or microbial invasion, mitochondrial oxidative phosphorylation, and ER stress-induced UPR. Low ROS levels regulate pathways involved in cell proliferation and differentiation, whereas elevated ROS promote carcinogenesis and resistance to anticancer therapy. This figure was created in BioRender.com.
Several medicinal plants have been shown to lower ROS levels, perhaps reversing multidrug resistance or making cervical cancer cells more sensitive to anticancer drugs. For instance, Cassia tora (Figure 4) contains emodin (an anthraquinone) that has demonstrated substantial antiproliferative effects against HeLa cells, inducing apoptosis and lowering DNA content through its antioxidant capabilities, which help to mitigate oxidative stress [43]. This process is linked to the cleavage of poly [adenosine 5′-diphosphate (ADP)-ribose] polymerase and the activation of caspase-9.

Figure 4.
Cassia tora plant with flowers: The plant contains bioactive compounds such as emodin, alaternin and nor-rubrofusarin glucose that induce apoptosis and mitigate ROS. This image was adopted from https://pixabay.com/photos/flower-yellow-petals-cassia-5474624/ (accessed on 12 January 2026) by DEZALB.
Moreover, emodin also leads to cell cycle arrest at the G2/M phase and inhibits cell growth and differentiation by downregulating cyclin A and cyclin-dependent kinase 2 (CDK2) [44,45]. Peroxynitrite (ONOO−) is a ROS produced when superoxide anion radicals (O2−) react with nitric oxide (NO) [46]. In the study conducted by Jain and Patil (2010) [47], alaternin and nor-rubrofusarin glucose (phenolic active compounds) extracted from Cassia tora demonstrated potent ONOO− scavenging activity and led to a decrease in ONOO− mediated nitration of tyrosine through electron donation.
Another interesting medicinal plant is Scutellaria baicalensis (Figure 5), whose flavonoid constituents, such as baicalein, targeted cyclin D1 and regulated the Wnt/β-catenin signalling cascade, suppressing cell proliferation and triggering apoptosis [48].

Figure 5.
Scutellaria baicalensis plant: It contains a flavonoid called baicalein that demonstrated a chemosensitising effect in cervical cancer cells to cisplatin and induced apoptosis and cuproptosis. This image was adapted from Wikipedia, https://en.wikipedia.org/wiki/Scutellaria_baicalensis (accessed on 12 January 2026).
In another preclinical study conducted by Jin et al. [49], baicalein enhanced the sensitivity of cervical cancer cells to cisplatin by inducing cuproptosis, a regulated form of cell death. In vivo experiments demonstrated that the combination of baicalein and cisplatin exerted a synergistic effect, promoting apoptosis and suppressing tumour progression. This potentiation of cisplatin cytotoxicity was mediated via the Akt signalling pathway, suggesting that baicalein may serve as a promising adjuvant for overcoming cisplatin resistance in cervical cancer therapy. In addition to the role of medicinal plants in regulating ROS levels, it is crucial to consider the impact of cell survival pathways on the efficacy of anticancer drugs.
3.1.3. Cell Survival Pathways and Their Contribution to Drug Resistance
The efficiency of chemotherapeutics is greatly impacted by cell survival pathways, which promote cell proliferation and interfere with apoptosis, thereby contributing to intrinsic drug resistance. The transforming Growth Factor Beta (TGF-β) cascade regulates several cellular activities, affecting the tumour microenvironment and cancer development. It is well recognised that TGF-β may promote tumour growth in more advanced cancers or suppress tumours in early stages. It suppresses immune surveillance while concurrently promoting cell proliferation and metastasis [50]. Tumours can evade treatment because of the pleiotropic nature of TGF-β signalling, leading to resistance to drugs [51].
The antioxidant pathway in cervical epithelial tissue is governed by the transcription factor Nuclear Factor Erythroid 2-related Factor 2 (NRF2), which controls gene transcription and regulates free radicals. Factors such as oxidative stress trigger the expression of p21 and NRF2 genes, enhancing glutathione metabolism and thereby reducing the efficiency of anticancer drugs [52]. Under normal conditions, Kelch-like ECH-associated protein 1 (Keap1) interacts with NRF2, promoting its proteasomal degradation and thereby reducing its concentration. However, during oxidative stress, Keap1-NRF2 interaction weakens, forcing them to dissociate and sequester NRF2 in the nucleus. This process stimulates the transcription of genes responsible for cytoprotection and metabolic processes. In tumour cells, the degradation of NRF2 is often disrupted, resulting in its overexpression and accumulation, which can trigger drug resistance [17].
Moreover, abnormal oncogenes alter pathways including MAPK/JNK, Ras/Raf/ERK, PI3K/AKT, and JAK/STAT, further establishing drug resistance [53]. The PI3K/AKT/mTOR and the JAK/STAT survival pathways play roles in different biological processes such as cell survival, apoptosis, tumour development, and cell proliferation [54]. These signalling pathways are activated by HPV infection and E6/E7 expression, which disrupts several cellular and molecular processes to promote tumourigenesis. Activation of the PI3K/AKT/mTOR pathway impacts the efficacy of various anticancer drugs through various mechanisms [5,55]. For instance, when AKT is activated, it suppresses p53 activity, thereby decreasing the apoptotic response to cisplatin therapy [56]. Additionally, activation of the PI3K/AKT/mTOR signalling cascade results in the overproduction of BCL-2, diminishing the efficacy of doxorubicin, thus contributing to doxorubicin resistance [57]. Resistance to gemcitabine has been associated with overexpression of the Ras/Raf/ERK and PI3K/AKT signalling pathways. These pathways interact and increase the expression of genes that prevent drug-induced apoptosis while also promoting cell survival and proliferation [48,58]. Furthermore, paclitaxel resistance can be triggered by transcription factors belonging to the Fox family. These factors regulate proteins such as BH3-interacting domain death agonist (Bid) and express anti-apoptotic proteins like BCL-2, initiating pathways such as MAPK/JNK that promote cell survival [59].
Numerous plant extracts have been shown to affect the TGF-β pathway, which can be very important for treating cervical cancer. Studies have demonstrated that some plant extracts can either stimulate or inhibit TGF-β activity, thereby influencing the susceptibility of tumours to drugs. For instance, Curcuma longa (Figure 6) extract (curcumin) and emodin have been shown to lower the expression of TGF-β receptor II and related Smad proteins, thus limiting tumour metastasis and invasion [60].

Figure 6.
Curcuma longa plant and root: It contains curcumin, which has demonstrated the ability to counteract resistance to chemotherapy. These images were adopted from https://unsplash.com/s/photos/curcuma-longa (accessed on 12 January 2026) and https://en.wikipedia.org/wiki/File:Curcuma_longa_roots.jpg (accessed on 12 January 2026).
Additionally, resveratrol has been linked to anticancer benefits by modulating the TGF-β pathway, enhancing the effectiveness of chemotherapeutic drugs and inhibiting tumour growth [61]. By disrupting mitochondrial membrane potential and increasing intracellular calcium levels, kaempferol (a type of flavonol) has demonstrated effectiveness in inhibiting cell growth and triggering apoptosis. Interestingly, it increases susceptibility to paclitaxel by decreasing P-glycoprotein activity [48,62]. Diosgenin (a saponin) and oridonin (a diterpenoid) extracted from Rabdosia rubescens (Figure 7) have been shown to suppress cervical cancer cell growth through numerous pathways, such as PI3K/AKT and MAPK signalling, inducing apoptosis, and lowering drug resistance mechanisms [63,64]. In addition to these pathways, the EMT is a fundamental process that also contributes to tumour progression and metastasis.

Figure 7.
Rabdosia rubescens plant: It contains phytocompounds such as diosgenin and oridonin that have been demonstrated to suppress cervical cancer growth via PI3K/AKT and MAPK signalling pathways in preclinical models. This image was adapted from Chen et al., Pathogens 2023, CC BY licence [65].
3.1.4. The Role of Epithelial–Mesenchymal Transition (EMT) in Drug Resistance
Epithelial cells lose their epithelial phenotype through a physiological process known as EMT. The cells exhibit a mesenchymal phenotype, losing cadherin-mediated cell–cell adhesion and its polarity; however, they become more invasive [66]. During EMT, transcription factors Ovo-Like Zinc Finger 1 (OVOL1) and Ovo-Like Zinc Finger 2 (OVOL2) bind to the epithelial markers E-cadherin and cytokeratin, as well as junctional proteins like catenins and claudins. This interaction enhances the production of mesenchymal markers such as vimentin, N-cadherin and fibronectin [67,68]. Overexpression of EMT transcription factors such as Snail Family Transcriptional Repressor 1 (SNAI1) triggers drug resistance by regulating the synthesis of ATP-binding cassette transport proteins. Research has demonstrated that overproduction of Snail and Twist enhances the activity of ABC transporter promoters, leading to an increased synthesis of these proteins in tumour cells [69,70]. Overexpression of ABC transporters facilitates the removal of chemotherapeutic agents from cells, leading to a reduction in their intracellular levels and, as a result, contributing to resistance against drugs such as doxorubicin, cisplatin, gemcitabine, and irinotecan [70,71]. Cells undergoing EMT frequently evade apoptosis, which is critical for the efficacy of several chemotherapeutics. For instance, during EMT, BCL-2 and BCL-XL become overexpressed, allowing cells to survive during chemotherapy treatment and resulting in drug resistance against agents that induce cell death, such as gemcitabine, irinotecan and cisplatin [67,70,72].
Numerous medicinal plants have demonstrated the ability to downregulate EMT, thereby reversing drug resistance. For instance, bergenin (a polyphenol), a phytochemical isolated from Bergenia crassifolia (Figure 8), has been shown to inhibit the growth of cells such as SiHa and C33A by inducing autophagy and cell death. It may also reverse EMT-related drug resistance by lowering the expression of angiogenic proteins implicated in tumour growth and metastasis, such as galectin-3 and matrix metalloproteinase-9 (MMP-9) [48,73].

Figure 8.
Bergenia crassifolia: It contains a polyphenol called bergenin that has been shown to lower the expression of angiogenic proteins such as galectin-3 and MMP-9 and reverse EMT-related drug resistance. This image was adapted from https://commons.wikimedia.org/wiki/File:Bergenia_crassifolia_a1.jpg (accessed on 12 January 2026) by Jerzy Opioła.
Curcumin has demonstrated the ability to counteract resistance to chemotherapy by inhibiting the expression of anti-apoptotic proteins and modulating the AKT/mTOR and NF-κB signalling pathways, thereby increasing the vulnerability of tumour cells to treatments including doxorubicin and cisplatin [74,75]. Berries and grapes contain resveratrol, which suppresses regulatory proteins, including Twist and Snail, to prevent EMT and sensitise cancer cells to treatment [76]. Tumour cells can acquire resistance after being exposed to an anticancer drug.
3.2. Acquired Drug Resistance
The term “acquired resistance” refers to cancer drug resistance that results in tumourigenesis after the initial drug administration. Genetic factors, such as mutations, increase the emergence of acquired resistance to treatment in tumour cells by altering genes that control metabolic pathways [77]. For example, amplification of the ATP-binding cassette subfamily B member 1 (ABCB1) gene, which encodes proteins associated with multidrug resistance, such as P-glycoprotein, can trigger resistance to lipophilic cytotoxic medications, including docetaxel and camptothecin, thereby reducing the chances of survival for cancer patients [78]. In genotype-matched targeted therapy, the development of acquired resistance limits the effectiveness of tumour response to treatment while also creating a significant barrier to achieving improved long-term survival outcomes [79]. Most research on acquired drug resistance has focused on understanding the molecular resistance mechanisms in cervical cancer cells. Epigenetic modification, drug efflux, and alterations in the DNA damage repair mechanism contribute to the emergence of acquired drug resistance.
3.2.1. The Role of Epigenetics in Acquired Drug Resistance
Epigenetics refers to biological phenomena where phenotypic changes are passed down through generations without altering the DNA sequence. These phenomena encompass DNA methylation, acetylation, and RNA silencing. Within the context of cervical cancer, epigenetics significantly influences the emergence of drug resistance, particularly against drugs such as gemcitabine, cisplatin, doxorubicin, 5-fluorouracil, irinotecan, and paclitaxel [80]. High-risk HPV strains, including HPV-16 and HPV-18, are key factors influencing epigenetic changes in cervical cancer. The HPV oncoproteins E6 and E7 produced by these viruses alter DNA methylation patterns, accelerating tumourigenesis and promoting drug resistance. For example, E6 can disrupt the activity of p53, a crucial tumour suppressor involved in DNA damage response, permitting cells with damaged DNA to continue growing even during chemotherapeutic treatment. This interplay not only encourages carcinogenesis but also promotes drug resistance [81,82]. The synergistic interaction between viral proteins E6 and E7 and cellular resistance mechanisms significantly contributes to the increased drug resistance observed in cervical cancer cells. These viral proteins induce hypermethylation of CpG islands within tumour suppressor gene promoter regions, thereby silencing their expression, while concurrently promoting hypomethylation that activates oncogenes. Viral proteins extensively alter the transcriptional capacity of infected cells by influencing chromatin-remodelling proteins and histone modifications. These alterations not only play a crucial role in carcinogenesis but also enhance the ability of cancer cells to evade therapeutic interventions, ultimately contributing to drug resistance [83,84].
Chemotherapy resistance in cervical cancer is also influenced by factors such as Toll-like receptors (TLRs), particularly TLR4. In HPV-positive cervical cancer cells, TLR4 activation is associated with increased cell survival and proliferation, which modulates the expression of genes associated with cell cycle control and apoptosis [85]. Studies show that TLR4 expression levels are often greater in HPV-positive cells than in HPV-negative cells, particularly in cervical cancer. This enhanced expression may result in higher cell survival and proliferation, potentially promoting chemoresistance [86].
DNA methylation, which suppresses transcription processes, is a key mechanism underlying drug resistance. For instance, DNA methylation of the Caspase 8 Associated Protein 2 (Casp8AP2) gene in cervical cancer reduces its expression and induces cisplatin and taxol resistance. Moreover, genetic research has uncovered that hypermethylation of DAPK is linked to drug resistance in various malignancies [87]. Essential tumour suppressor genes, such as CDKN2A, are commonly found to be hypermethylated, thereby disrupting cellular mechanisms such as cell cycle control and apoptosis, allowing tumour cells to survive doxorubicin therapy. Acetylation and methylation of histones play an important role in modulating the expression of drug resistance genes. Genes involved in apoptosis or drug sensitivity may be suppressed as a result of histone deacetylase (HDAC) dysregulation [81,88,89]. Furthermore, histone modifications can alter chromatin structure, leading to increased transcription of DNA repair genes such as Xeroderma Pigmentosum Group F (XPF) and Excision Repair Cross-Complementation Group 1 (ERCC1), which repair cisplatin-induced DNA damage, thereby promoting cancer survival and increasing resistance to cisplatin during treatment [90]. DNA demethylation in CpGs of promoter sequences, along with histone methylation, can trigger NRF2 expression. For instance, DNA methylation and histone methylation status in the context of oxidative stress induced by 5-FU influence the expression of NRF2, which then induces drug resistance to 5-FU [91]. Epigenetic modifications that enhance topoisomerase II expression or affect its activity have been shown to induce resistance to irinotecan by decreasing its ability to generate DNA breaks [90,92]. Resistance to paclitaxel has been associated with epigenetic changes that increase the expression of proteins that are responsible for microtubule stability and dynamics. Additionally, epigenetic changes can modify the expression of enzymes that regulate nucleotide metabolism, impacting gemcitabine’s activation and efficiency [20,88].
Alternative splicing is a molecular mechanism that allows the synthesis of a variety of mature mRNAs from a single gene [93]. Defects occurring in the process result in the synthesis of non-functional or unfavourable products that may initiate cancer development and progression. Drug resistance against anticancer agents may develop due to defects in the splicing process. BCL-2–interacting mediator of cell death (BIM) is a group of proteins involved in essential biological processes such as maintaining haematopoietic balance, restriction of autoimmune diseases, and initiation of cancer. When the BIM proteins are upregulated, inhibitors of cell death pathways such as cellular inhibitor of apoptosis proteins (cIAP1 and cIAP2), survivin, X-linked inhibitor of apoptosis protein (XIAP), and livin accumulate in the tumours, promoting drug resistance [94,95].
Numerous medicinal plants and their phytochemicals have been shown to affect epigenetic modifications in preclinical models, which can be very important for treating cervical cancer. For instance, Salvia miltiorrhiza (Figure 9) has been associated with changes in cytosine DNA methylation and the control of secondary metabolites, which can improve its therapeutic properties. The phytochemicals in this plant, such as salvianolic acid B and tanshinone IIA, may reverse epigenetic silencing, though clinical efficacy has not yet been demonstrated [90,96].

Figure 9.
Salvia miltiorrhiza flowers: they contain salvianolic acid B and tanshinone IIA, which have the potential to reverse abnormal epigenetic changes. This image was adopted from https://pixabay.com/photos/salvia-white-sage-bloom-meadow-4262870/ (accessed on 12 January 2026) by silviarita.
Rhaponticum Carthamoides (Figure 10) has demonstrated the ability to induce apoptosis in cancer cells via epigenetic pathways. The anticancer properties of its extracts, such as tricaffeoylquinic acid derivatives, may be linked to their ability to regulate histone modifications and induce DNA damage [97,98].

Figure 10.
Rhaponticum Carthamoides plant: It contains tricaffeoylquinic acid derivatives that may regulate histone modifications and induce DNA damage. This image was adapted from https://en.wikipedia.org/wiki/Rhaponticum_carthamoides#/media/File:Rhaponticum_carthamoides.jpg (accessed on 12 January 2026) by Doronenko.
Through processes such as DNA methylation and histone modification, several phytochemicals interact with the epigenetic machinery, influencing gene expression. For instance, epigallocatechin-3-gallate (a type of flavonoid) has been shown to alter microRNA expression and DNA methylation, which can suppress oncogene expression and reactivate silenced tumour suppressor genes. It can modify gene expression patterns, increasing apoptosis and promoting cell cycle arrest, thereby improving chemotherapeutic efficacy and reversing resistance [99].
The most studied polyphenols, such as curcumin, quercetin, and resveratrol, have been shown to regulate both DNA methylation and histone acetylation in preclinical models. Moreover, studies have indicated that they inhibit histone deacetylases and DNA methyltransferases, leading to the downregulation of oncogenes and the reactivation of previously silenced tumour suppressor genes. This modulation can enhance the susceptibility of cervical cancer cells to chemotherapy while also overcoming drug resistance [100]. Numerous natural compounds have been identified as effective inhibitors of HPV E6 and E7 activity, which are crucial for the progression of cervical cancer. For instance, luteolin has been shown to bind to the E6 protein, preventing the degradation of p53, a vital tumour suppressor, thereby reducing the viability of HPV-infected cervical cancer cells [101]. Similarly, curcumin has demonstrated the ability to inhibit both E6 and E7 transcripts and proteins, thereby obstructing the translocation of NF-κB and AP-1 transcription factors and ultimately inducing apoptosis in HeLa, SiHa, and C33A cancer cell lines [102]. Additionally, tanshinone IIA has been reported to downregulate E6 and E7 expression while promoting apoptosis and cell cycle arrest in various cervical cancer cell lines, including HeLa, SiHa, CaSki, and C33A [103].
3.2.2. Drug Efflux and Its Role in Drug Resistance
Drug efflux mechanisms have a major impact on the development of multidrug resistance. Multidrug resistance is associated with upregulated ATP-binding cassette (ABC) transporters, such as P-glycoprotein (P-gp), as well as members of the multidrug resistance protein (MRP) family, including MRP1 and MRP2, which are located on the tumour cell membranes and play a crucial role in mediating drug efflux [104]. P-glycoprotein, commonly known as ABCB1, is a chemotherapeutic drug-binding protein transcribed by ATP-binding cassette subfamily B member 1. The frequent administration of chemo-drugs that are ABCB1 substrates, such as doxorubicin, gemcitabine, and paclitaxel, leads to overexpression of ABCB1. When ABCB1 is overexpressed, it expels chemotherapy drugs from tumour cells, decreasing their intracellular concentration and effectiveness, ultimately resulting in multidrug resistance [105,106,107]. MRP1 has been linked to a higher cisplatin efflux in cervical cancer cells. MRP1 overexpression may result in lower intracellular concentrations of cisplatin, which could reduce the cytotoxic effects of the drug [108]. MRP2 contributes to drug resistance as well; however, its expression patterns could vary. In several studies, MRP2 was found to enhance cisplatin efflux in some cancer cell lines, with some resistant cell lines exhibiting lower expression and susceptible cell lines showing greater levels [109].
Studies have shown that phytochemicals such as curcumin, resveratrol, epigallocatechin gallate, and quercetin can modify the expression and activity of ABC transporters, which can improve treatment outcomes by increasing drug retention [11]. In various cancer cell lines, curcumin, epigallocatechin gallate, and resveratrol have been shown to reduce the expression of P-glycoprotein (P-gp) and other ABC transporters such as MRP1. This downregulation leads to increased intracellular levels of chemotherapeutic drugs. By inhibiting P-gp, these phytochemicals can reverse MDR, thereby enhancing the sensitivity of cancer cells to treatments such as vincristine and doxorubicin [11,110]. Quercetin has been reported to inhibit several ABC transporters, including MRP, P-gp, and breast cancer resistance protein (BCRP). It can also increase the bioavailability of chemotherapy agents by preventing their efflux. This results in a greater drug accumulation within tumour cells, thereby amplifying the cytotoxic effects of drugs such as doxorubicin and cisplatin [111]. The increased cytotoxic effects of such drugs are closely linked to their ability to induce DNA damage, necessitating further exploration of how DNA damage repair induces drug resistance.
3.2.3. DNA Damage Repair
When DNA damage occurs, DNA repair mechanisms are triggered to repair the damaged DNA using various repair proteins. The risk of cancer development and drug resistance increases when mutations arise in genes that code for DNA repair proteins like O-methylguanyl DNA methyltransferase (MGMT) and Ataxia telangiectasia mutated (ATM) protein kinase [112,113]. The DNA repair mechanism is precisely regulated by DNA repair-associated genes through DNA strand breaks and homologous end-joining pathways. If repair enzymes, such as DNA polymerases, are inhibited, cells containing damaged DNA may erroneously duplicate, thereby promoting tumourigenesis and increasing the chances of acquiring drug resistance [114,115]. For instance, cervical cancer cells with upregulated DNA damage repair enzyme activity could swiftly repair cisplatin-DNA adducts. Cisplatin is recognised as the most effective drug for cervical cancer treatment. It induces intra-strand DNA crosslinks that activate the nucleotide excision repair (NER) mechanism. It has been suggested that NER prevents apoptosis in cells treated with cisplatin by activating components of the ATM pathway and recruiting them to the DNA damage site. Activated ATM quickly degrades drug-DNA adducts, resulting in drug resistance [116,117]. Doxorubicin treatment forms double-strand breaks, which are normally repaired by the non-homologous end-joining (NHEJ) and homologous recombination mechanisms. Overexpression of these DNA damage response mechanisms allows cells to resist doxorubicin treatment by successfully repairing the generated damage [118]. 5-fluorouracil (5-FU) disrupts DNA synthesis and causes strand breaks by misincorporating uracil into the DNA. The cell’s capacity to repair this damage may affect the efficacy of 5-FU; hence, improved repair mechanisms may induce resistance to 5-FU [119]. Irinotecan induces DNA damage predominantly by inhibiting topoisomerase I, leading to single-stranded breaks repaired through the base excision repair pathway. Tumours with strong DNA damage response capacities are more likely to survive irinotecan treatment because their lesions can be repaired effectively [120].
Numerous medicinal plants have demonstrated potential in modulating DNA damage repair mechanisms in cancer cells, which could improve chemotherapy efficacy and help combat drug resistance. For example, curcumin has been shown to impact several processes, such as non-homologous end joining and base excision repair, which lowers the accumulation of mutations and sensitises cancer cells to chemotherapy. Furthermore, it can cause cancer cells to undergo apoptosis, which increases their vulnerability to therapies such as doxorubicin and cisplatin [121]. Additionally, resveratrol is known to modify various DNA repair mechanisms, such as homologous recombination and mismatch repair [122]. In addition to DNA damage repair, tumour cells could resist chemotherapeutic agents due to inactivation of the anticancer drug.
3.2.4. Anticancer Drug Inactivation
The interactions between the drug and various cellular proteins may reduce their efficiency and potency, thereby inactivating them [123]. For instance, protein-bound cisplatin has far lower absorption into cells and tissues than free cisplatin and is considerably less cytotoxic. Plasma proteins, particularly those with thiol groups such as human serum albumin, are irreversibly bound to cisplatin, inactivating it, reducing its efficacy and leading to cisplatin resistance. The Phase II glucuronidation pathway, responsible for the detoxification of various drugs, is one of the processes involved in drug inactivation. In the Glucuronidation pathway, various types of uridine diphosphate glucuronosyltransferase catalyse the transfer of uridine-5′-diphospho-α-D-glucuronic acid to the substrate, increasing its solubility and facilitating its degradation. Overexpression of the glucuronidation pathway results in the degradation of anti-tumour drugs, thereby inactivating them [16,124,125]. One typical detoxifying mechanism is glutathione (GSH) conjugation catalysed by glutathione S-transferases (GSTs). When platinum-based drugs such as cisplatin and oxaliplatin are conjugated with GSH, they become substrates for ATP-binding cassette (ABC) transporters. This process induces resistance by drastically lowering the intracellular concentration of these drugs [8,125]. Cytochrome P450 3A4 (CYP3A4) plays a role in drug metabolism. When CYP3A4 enzymes are high, they metabolise drugs such as irinotecan into inactive metabolites, reducing the levels of SN-38 (the active form of irinotecan), compromising treatment efficacy, and leading to drug resistance [126,127].
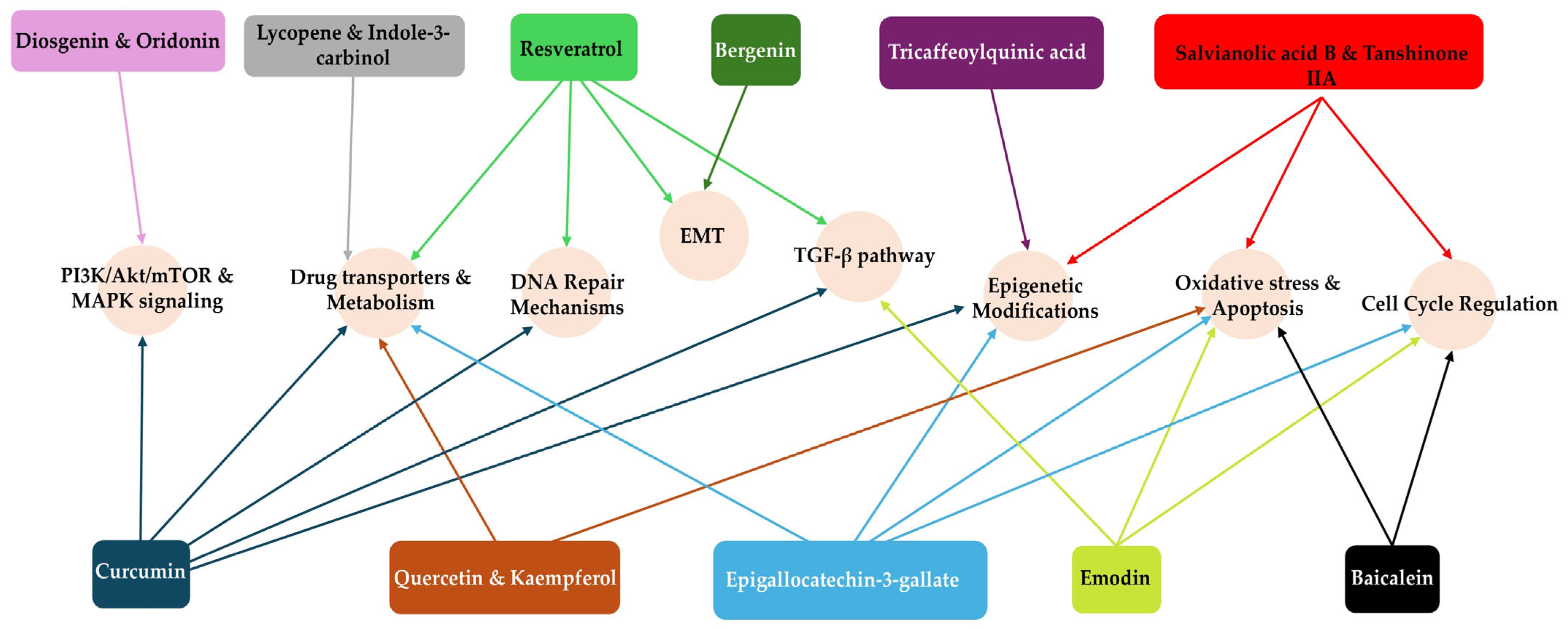
Numerous phytochemicals have been reported to reverse drug resistance mechanisms in tumour cells, especially by focusing on pathways related to drug inactivation. For instance, curcumin and quercetin improve the efficacy of certain chemotherapy drugs such as cisplatin and doxorubicin by regulating mitochondrial activities. They have been shown to reduce the activity of cytochrome P450 enzymes, which are involved in the metabolism of several anticancer drugs, enhancing their bioavailability and inducing apoptosis [128,129]. Certain phytochemicals, including epigallocatechin gallate, resveratrol, lycopene, and indole-3-carbinol, play a role in regulating GSTs and causing cancer cells to undergo apoptosis. These phytochemicals may improve chemotherapeutic efficacy by decreasing GST activity while simultaneously activating apoptotic pathways that result in cancer cell death. Integrating such phytochemicals into cancer treatments might enhance therapeutic outcomes by overcoming drug resistance mechanisms induced by excessive GST levels [44,130,131]. The summary of medicinal plants/phytochemicals and their target mechanisms is provided in Table 1 and Figure 11.

Table 1.
Summary of medicinal plants targeting drug resistance in cervical cancer and their target mechanisms.
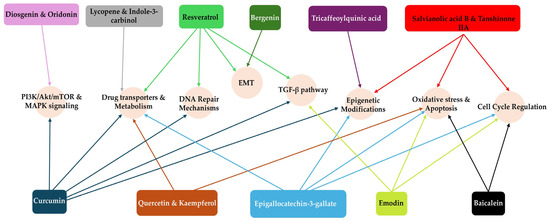
Figure 11.
Phytochemicals and their targeted pathways: The diagram shows phytochemicals acting on major molecular processes—oxidative stress, apoptosis, cell cycle, TGF-β, DNA repair, PI3K/Akt/mTOR, MAPK, epigenetic regulation, EMT, and drug transport and metabolism. Colour-coded arrows indicate activation, inhibition, or modulation. Detailed mechanisms are provided in Table 1.
4. Critical Synthesis of Findings
4.1. Comparative Analysis of Mechanisms
Among the mechanisms identified in the reviewed studies, drug efflux mediated by ABC transporters and the activation of survival signalling pathways, including PI3K/AKT/mTOR and JAK/STAT, were the most commonly reported. While efflux is consistently linked to reduced intracellular drug accumulation, its translational relevance is limited by the absence of clinically approved inhibitors with acceptable toxicity. In contrast, EMT and epigenetic modulation, though less frequently studied, appear more promising for clinical translation because they are reversible processes and can be targeted by phytochemicals with established safety records, such as curcumin and resveratrol. Therefore, these mechanisms vary not only in their frequency of occurrence but also in their potential utility for therapeutic intervention.
4.2. Contradictions and Limitations Across Studies
Several contradictions emerged across the literature. For example, quercetin was reported as a potent chemosensitiser in some studies, while others found minimal or no effect, likely due to differences in cell line models, dosing regimens, and bioavailability. Similarly, the modulation of reactive oxygen species (ROS) is described variably as either protective or sensitising, contingent upon the experimental context. This variability underscores the necessity for standardised assays and more clearly defined mechanistic endpoints.
Notable limitations across the reviewed studies include a predominance of in vitro models, inconsistent documentation of phytochemical concentrations, and a lack of pharmacokinetic validation. Numerous studies utilised supraphysiological doses without adequate justification or failed to disclose formulation strategies aimed at enhancing solubility and absorption. Mechanistic assertions were frequently based on single biomarkers without corroborative evidence from upstream or downstream pathways or rescue experiments. Furthermore, few studies incorporated in vivo validation, and those that did rarely included pharmacokinetic profiling, toxicity assessment, or clinically relevant dosing. The absence of standardised protocols, inconsistent use of controls, and limited reporting of negative results suggest potential publication bias and hinder reproducibility. Collectively, these inconsistencies emphasise the need for cautious interpretation of existing data and highlight the imperative for harmonised methodologies to facilitate translational advancement.
4.3. Mechanisms Exhibiting Potential for Clinical Translation
From a translational research standpoint, the reversal of epithelial–mesenchymal transition (EMT) and the modulation of epigenetic processes emerge as particularly promising therapeutic strategies. Phytochemicals such as curcumin, resveratrol, and bergenin demonstrated dual functionality by targeting EMT-associated markers and concurrently downregulating efflux transporters in preclinical models. Epigenetic modulators have shown potential in restoring chemosensitivity through the reversal of HPV-induced gene silencing; however, their clinical implementation necessitates the development of enhanced delivery systems to address challenges related to limited bioavailability. Additionally, inhibitors of the PI3K/AKT/mTOR signalling pathway represent a viable avenue for intervention, although comprehensive in vivo validation remains essential.
5. Anticancer Potential of Medicinal Plants and Clinical Trial Issues
Evidence Transition
While Section 3 and Section 4 primarily synthesised findings from in vitro and animal models, clinical evidence for phytochemicals in cervical cancer remains limited. Most reported effects should therefore be interpreted as preclinical insights rather than established therapeutic efficacy. This section highlights the few available clinical studies, discusses barriers to translation, and outlines the challenges of moving from mechanistic promise to patient benefit.
In addition to anticancer drug resistance, the non-specificity of anticancer drugs that induce apoptosis has been reported in several studies. Medicinal plants have been shown to interact with multiple resistance mechanisms since they contain a variety of phytocomponents. Therefore, compared to single conventional drugs, medicinal plants could offer complementary strategies for addressing chemoresistance, particularly in preclinical models. It is suggested that these compounds may offer benefits, including consistent availability, ensuring therapeutic effects, and fewer side effects [132,133,134]. While medicinal plants show promising preclinical activity, their safety and specificity in humans remain to be validated through rigorous clinical trials [135]. Plants can synthesise substances that are challenging to synthesise by chemical synthesis, rendering them a valuable source for the formulation of innovative anticancer agents that may specifically target cancer cells or sensitise tumour cells to chemotherapy [136]. Plants have also been shown to be therapeutic and efficient in extending patient survival rates, minimising chemotherapy side effects and chemoresistance, and increasing cancer patients’ quality of life, although further trials are needed to confirm effects on patient survival [137]. As a result, implementing a therapeutic approach that utilises medicinal plants could offer a strategy that inhibits cancer cell proliferation while reversing drug resistance. More focus has been placed on using medicinal plants as innovative, independent anticancer medications or in combination with already-approved anticancer agents to circumvent resistance processes to effectively treat cancer. Some clinical studies suggest that integrating phytochemicals with conventional chemotherapy can improve patient outcomes. For instance, curcumin has demonstrated improved efficacy when combined with gemcitabine [129]. Despite this potential, the number of clinical trials remains limited compared to preclinical studies. Phytochemicals can synergise with traditional chemotherapeutic agents, potentially increasing their effectiveness while reducing toxicity. This synergy is supported by various studies, showing that combinations of phytochemicals may induce cell death and inhibit tumour growth in preclinical models [138,139,140].
Although several studies demonstrate the anticancer potential of medicinal plants, only a few of the phytochemicals reviewed have documented human clinical trials (Table 2); most remain supported by preclinical evidence. Cervical-specific trials are scarce, with curcumin being the only compound currently under investigation in invasive cervical cancer.
The bioavailability of plant-derived compounds can be a significant challenge since many active compounds may not be easily absorbed or metabolised in the body. The body’s capacity to absorb and use these phytochemicals can be influenced by variables including solubility and stability. The water solubility of phytochemicals, particularly phenolics and flavonoids, hinders their ability to pass through intestinal lipid membranes, leading to inadequate bioavailability and absorption. For instance, curcumin has limited bioavailability since it is poorly soluble in water [48,141,142]. Furthermore, phenolic aglycone metabolism can reduce bioavailability by increasing their water solubility and facilitating excretion. Phytochemicals such as epigallocatechin gallate and resveratrol confront similar issues due to low absorption as well as fast first-pass metabolism [143,144].
Plant-derived compounds face substantial regulatory and translational barriers. Unlike conventional pharmaceuticals, phytochemicals often lack standardised formulations, leading to batch variability and inconsistent therapeutic effects. Regulatory frameworks for herbal compounds remain fragmented, delaying trial approval and market access. Funding constraints further limit trial size and duration, reducing statistical power and generalisability [145,146,147]. Additionally, many studies lack pharmacokinetic data, toxicity profiling, and reproducible formulations, impeding clinical advancement. Although some are generally considered safe, phytochemicals can become toxic at higher doses or with overexposure. For instance, a study by Liu et al. (2022) has demonstrated dose-dependent adverse reactions for curcumin [148]. The reproducibility of research findings is further confounded by differences in the chemical composition of plant extracts, which are impacted by parameters such as geographic origin as well as preparation methods. This discrepancy may result in different batches of the same plant extract exhibiting varying therapeutic effects. Furthermore, reproducibility is further impeded by inadequate reporting guidelines and methodological errors [145,149].
There is conflicting evidence regarding the safety and effectiveness of phytochemicals in humans, despite several studies suggesting their potential to overcome chemoresistance. Due to variations in patient metabolism, bioavailability, and tumour microenvironments, some research indicates that the benefits seen in vitro may not fully translate to clinical outcomes. Furthermore, different study methods and phytochemical formulations may produce different results [11,150]. Despite promising preclinical data, many phytochemicals fail to advance clinically due to poor solubility, rapid metabolism, and low tissue penetration. Without formulation strategies to improve bioavailability, therapeutic concentrations cannot be achieved in vivo [151]. Moreover, the lack of intellectual property protection for natural compounds reduces commercial incentive for pharmaceutical investment, further slowing translation [152].

Table 2.
Clinical trial evidence for selected phytochemicals.
Table 2.
Clinical trial evidence for selected phytochemicals.
| Phytochemical | Cervical Cancer Trial | CIN/HPV Human Study | Trials in Other Cancers | Outcome Summary | Reference |
|---|---|---|---|---|---|
| Curcumin | Recruiting trial adjunct to chemotherapy (Phase II, NCT06080841) | Capsule for CIN (Phase II, NCT02554344) | Pancreatic cancer (Phase II, NCT00192842); Breast cancer (Phase I/II, NCT01740323) | Safe; poor bioavailability; cervical trial ongoing; CIN regression signals; safe adjunct in pancreatic and breast | [153,154,155,156] |
| Epigallocatechin-3-gallate | None | None | Prostate cancer (Phase II, NCT00596011) | Tested as Polyphenon E in men undergoing prostatectomy; safe; showed biomarker modulation (PSA, tissue EGCG levels) | [157] |
| Resveratrol | None | None | Colon cancer prevention (Phase I, NCT00256334) | Safe; modest antiproliferative effects in colorectal mucosa | [158] |
| Diindolylmethane (Indole-3-carbinol derivative) | None | CIN regression (not formally registered) | Breast cancer prevention (Phase I, NCT01391689) | Safe; exploratory efficacy | [159] |
| Indole-3-carbinol (I3C) | None | CIN regression (Phase II, NCT00988845) | None | Regression in subset; safe | [160] |
| Active Hexose Correlated Compound | None | Persistent HPV clearance (Phase II, NCT02405533) | None | Increased HPV clearance rates; safe | [161] |
6. Study Limitations
The current literature on the clinical efficacy and safety of natural products in overcoming drug resistance in cervical cancer is limited, with most evidence derived from preclinical studies. Well-designed clinical trials are needed to validate these findings. A major challenge is the poor bioavailability of many promising phytochemicals, which may limit their therapeutic use. Approaches such as nanoformulations, liposomes, micelles, and polymer-based delivery systems are under investigation to improve absorption, stability, and tumour-specific targeting. These strategies aim to overcome solubility barriers, protect compounds from first-pass metabolism, and enhance intracellular delivery, but further work is needed in this area. Given the scarcity of clinical data, conclusions regarding therapeutic efficacy must be interpreted cautiously. This review is limited by the availability of published clinical studies and may not capture all relevant information on the topic.
7. Conclusions
Drug resistance accounts for many cancer treatment failures. Regardless of efforts to combat drug resistance, there is currently no viable solution. Consequently, uncovering the underlying molecular processes of drug resistance is crucial for the development of various cervical cancer treatments. Medicinal plants may offer a complementary strategy for addressing drug resistance during cervical cancer treatment due to their diverse phytochemical profiles that show anticancer activity in preclinical models. However, these findings are primarily based on in vitro and in vivo studies, and their translation to clinical success remains uncertain. Future research should focus on well-designed clinical trials to determine the efficacy and safety of specific phytochemicals in reversing drug resistance. Given the promising anticancer properties and drug-sensitising effects observed in vitro, potential phytochemicals such as curcumin, resveratrol, and kaempferol should be further investigated in clinical trials. More studies are needed to address their bioavailability and examine nanoformulations, liposomes and other drug delivery systems that can improve the absorption, distribution, and targeted delivery of phytochemicals to the cancer cells, as well as clarify their mechanisms of action. Additionally, investigating the synergistic effect of combining phytochemicals with conventional chemotherapy drugs could enhance therapeutic efficacy while reducing toxicity, though this requires further validation and robust clinical evidence. By addressing these challenges, the therapeutic relevance of medicinal plants may be clarified, contributing to future strategies for overcoming drug resistance in cervical cancer.
Author Contributions
Conceptualisation, T.P.M. and Z.M.; methodology, T.P.M.; validation, K.L.; formal analysis, T.P.M.; investigation, T.P.M.; resources, Z.M.; data curation, T.P.M.; writing—original draft preparation, T.P.M.; writing—review and editing, K.L. and Z.M.; visualisation, T.P.M.; supervision, Z.M.; project administration, Z.M. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Research Foundation (NRF), South Africa, under grant number CSUR240406212512.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analysed in this study. Data sharing is not applicable to this article.
Acknowledgments
The authors would like to express their gratitude to the University of Limpopo for institutional support.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Hull, R.; Mbele, M.; Makhafola, T.; Hicks, C.; Wang, S.; Reis, R.M.; Mehrotra, R.; Mkhize-Kwitshana, Z.; Kibiki, G.; Bates, D.O.; et al. Cervical Cancer in Low and Middle-income Countries (Review). Oncol. Lett. 2020, 20, 2058–2074. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.; Jhingran, A.; Oaknin, A.; Denny, L. Cervical cancer. Lancet 2019, 393, 169–182. [Google Scholar] [CrossRef]
- Sazonova, E.V.; Yapryntseva, M.A.; Pervushin, N.V.; Tsvetcov, R.I.; Zhivotovsky, B.; Kopeina, G.S. Cancer drug resistance: Targeting proliferation or programmed cell death. Cells 2024, 13, 388. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Chen, X. Drug resistance and combating drug resistance in cancer. Cancer Drug Resist. 2019, 2, 141–160. [Google Scholar] [CrossRef]
- Nikolaou, M.; Pavlopoulou, A.; Georgakilas, A.; Kyrodimos, E. The challenge of drug resistance in cancer treatment: A current overview. Clin. Exp. Metastasis 2018, 35, 309–318. [Google Scholar] [CrossRef]
- Vasan, N.; Baselga, J.; Hyman, D. A view on drug resistance in cancer. Nature 2019, 575, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Housman, G.; Byler, S.; Heerboth, S.; Lapinska, K.; Longacre, M.; Snyder, N.; Sarkar, S. Drug Resistance in Cancer: An Overview. Cancers 2014, 6, 1769–1792. [Google Scholar] [CrossRef] [PubMed]
- Alam, A.; Farooq, U.; Singh, R.; Dubey, V.; Kumar, S.; Kumari, R.; Naik, K.K.; Tripathi, B.; Dhar, K. Chemotherapy Treatment and Strategy Schemes: A Review. J. Toxicol. 2018, 2, 555600. [Google Scholar] [CrossRef]
- More, M.P.; Pardeshi, S.R.; Pardeshi, C.V.; Sonawane, G.A.; Shinde, M.N.; Deshmukh, P.K.; Naik, J.B.; Kulkarni, A.D. Recent advances in phytochemical-based Nano-formulation for drug-resistant Cancer. Med. Drug Discov. 2021, 10, 100082. [Google Scholar] [CrossRef]
- Tinoush, B.; Shirdel, I.; Wink, M. Phytochemicals: Potential Lead Molecules for MDR Reversal. Front. Pharmacol. 2020, 11, 832. [Google Scholar] [CrossRef]
- Nishino, K.; Yamasaki, S.; Nakashima, R.; Zwama, M.; Hayashi-Nishino, M. Function and inhibitory mechanisms of multidrug efflux pumps. Front. Microbiol. 2021, 12, 737288. [Google Scholar] [CrossRef]
- Sadida, H.Q.; Abdulla, A.; Marzooqi, S.A.; Hashem, S.; Macha, M.A.; Akil, A.S.; Bhat, A.A. Epigenetic modifications: Key players in cancer heterogeneity and drug resistance. Transl. Oncol. 2024, 39, 101821. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Song, L.; Hou, Y.; Li, F. Reactive oxygen species induced by Icaritin promote DNA strand breaks and apoptosis in human cervical cancer cells. Oncol. Rep. 2018, 41, 765–778. [Google Scholar] [CrossRef] [PubMed]
- Tiek, D.; Cheng, S.Y. DNA damage and metabolic mechanisms of cancer drug resistance. Cancer Drug Resist. 2022, 5, 368–379. [Google Scholar] [CrossRef]
- Ghosh, S. Cisplatin: The first metal based Anticancer Drug. Bioorg. Chem. 2019, 88, 102925. [Google Scholar] [CrossRef]
- Jung, B.; Yoo, H.; Shin, S.; Park, Y.; Jeon, S. Dysregulation of NRF2 in Cancer: From Molecular Mechanisms to Therapeutic Opportunities. Biomol. Ther. 2018, 26, 57–68. [Google Scholar] [CrossRef]
- Bukowski, K.; Kciuk, M.; Kontek, R. Mechanisms of multidrug resistance in cancer chemotherapy. Int. J. Mol. Sci. 2020, 21, 3233. [Google Scholar] [CrossRef] [PubMed]
- Holohan, C.; Van Schaeybroeck, S.; Longley, D.; Johnston, P. Cancer drug resistance: An evolving paradigm. Nat. Rev. Cancer 2013, 13, 714–726. [Google Scholar] [CrossRef] [PubMed]
- Emran, T.B.; Shahriar, A.; Mahmud, A.R.; Rahman, T.; Abir, M.H.; Siddiquee, M.F.R.; Ahmed, H.; Rahman, N.; Nainu, F.; Wahyudin, E.; et al. Multidrug resistance in cancer: Understanding molecular mechanisms, immunoprevention and therapeutic approaches. Front. Oncol. 2022, 12, 891652. [Google Scholar] [CrossRef]
- Hammerlindl, H.; Schaider, H. Tumour cell-intrinsic phenotypic plasticity facilitates adaptive cellular reprogramming driving acquired drug resistance. J. Cell Commun. Signal. 2017, 12, 133–141. [Google Scholar] [CrossRef]
- Vaidya, F.U.; Chhipa, A.S.; Mishra, V.; Gupta, V.K.; Rawat, S.G.; Kumar, A.; Pathak, C. Molecular and cellular paradigms of multidrug resistance in cancer. Cancer Rep. 2020, 5, e1291. [Google Scholar] [CrossRef]
- Weinberg, F.; Ramnath, N.; Nagrath, D. Reactive oxygen species in the tumour microenvironment: An overview. Cancers 2019, 11, 1191. [Google Scholar] [CrossRef] [PubMed]
- Pelicano, H.; Carney, D.; Huang, P. Ros stress in cancer cells and therapeutic implications. Drug Resist. Updat. 2004, 7, 97–110. [Google Scholar] [CrossRef]
- Renaudin, X. Reactive oxygen species and DNA damage response in cancer. Int. Rev. Cell Mol. Biol. 2021, 364, 139–161. [Google Scholar] [CrossRef]
- Sieber, O.M.; Heinimann, K.; Tomlinson, I.P. Genomic instability—The engine of tumorigenesis? Nat. Rev. Cancer 2003, 3, 701–708. [Google Scholar] [CrossRef]
- Zhao, R.Z.; Jiang, S.; Zhang, L.; Yu, Z.B. Mitochondrial electron transport chain, ROS generation and uncoupling (review). Int. J. Mol. Med. 2019, 44, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Jo, D.S.; Park, N.Y.; Cho, D. Peroxisome quality control and dysregulated lipid metabolism in neurodegenerative diseases. Exp. Mol. Med. 2020, 52, 1486–1495. [Google Scholar] [CrossRef]
- Cruz-Gregorio, A.; Aranda-Rivera, A.; Pedraza-Chaverri, J. Human Papillomavirus-related Cancers and Mitochondria. Virus Res. 2020, 286, 198016. [Google Scholar] [CrossRef] [PubMed]
- Paletta-Silva, R.; Rocco-Machado, N.; Meyer-Fernandes, J. NADPH Oxidase Biology and the Regulation of Tyrosine Kinase Receptor Signalling and Cancer Drug Cytotoxicity. Int. J. Mol. Sci. 2013, 14, 3683–3704. [Google Scholar] [CrossRef]
- Dev, S.S.; Abidin, S.A.Z.; Farghadani, R.; Othman, I.; Naidu, R. Receptor tyrosine kinases and their signalling pathways as therapeutic targets of curcumin in cancer. Front. Pharmacol. 2021, 12, 772510. [Google Scholar] [CrossRef]
- Kina, S.; Kinjo, T.; Liang, F.; Nakasone, T.; Yamamoto, H.; Arasaki, A. Targeting EphA4 abrogates intrinsic resistance to chemotherapy in well-differentiated cervical cancer cell line. Eur. J. Pharmacol. 2018, 84, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, V.; Rajaram, R. A paradoxical role of reactive oxygen species in cancer signalling pathway: Physiology and pathology. Process. Biochem. 2021, 100, 69–81. [Google Scholar] [CrossRef]
- Shi, X.; Zhang, Y.; Zheng, J.; Pan, J. Reactive oxygen species in cancer stem cells. Antioxid. Redox Signal. 2012, 16, 1215–1228. [Google Scholar] [CrossRef] [PubMed]
- Preci, D.P.; Almeida, A.; Weiler, A.L.; Mukai Franciosi, M.L.; Cardoso, A.M. Oxidative damage and antioxidants in cervical cancer. Int. J. Gynecol. Cancer 2020, 31, 265–271. [Google Scholar] [CrossRef]
- Perillo, B.; Di Donato, M.; Pezone, A.; Di Zazzo, E.; Giovannelli, P.; Galasso, G.; Castoria, G.; Migliaccio, A. Ros in cancer therapy: The bright side of the moon. Exp. Mol. Med. 2020, 52, 192–203. [Google Scholar] [CrossRef]
- Kumari, S.; Badana, A.K.; Murali, M.G.; Shailender, G.; Malla, R. Reactive oxygen species: A key constituent in cancer survival. Biomark. Insights 2018, 13, 1177271918755391. [Google Scholar] [CrossRef]
- Harahap, N.S.; Amelia, R.; Sibuea, A.; Manalu, N.; Novita, N. Hsp70 (heat shock protein 70) expression and antioxidant as a protective against oxidative stress triggered by sub-maximal physical activity. IOP Conf. Ser. Earth Environ. Sci. 2021, 713, 012051. [Google Scholar] [CrossRef]
- Peinado-Ruiz, I.C.; Burgos-Molina, A.M.; Sendra-Portero, F.; Ruiz-Gómez, M.J. Relationship between heat shock proteins and cellular resistance to drugs and ageing. Exp. Gerontol. 2022, 167, 111896. [Google Scholar] [CrossRef]
- Van Loenhout, J.; Peeters, M.; Bogaerts, A.; Smits, E.; Deben, C. Oxidative Stress-Inducing Anticancer Therapies: Taking a Closer Look at Their Immunomodulating Effects. Antioxidants 2020, 9, 1188. [Google Scholar] [CrossRef]
- Chen, X.; Yang, N.; Wang, Y.; Yang, S.; Peng, Y. PCK1-mediated glycogenolysis facilitates ROS clearance and chemotherapy resistance in cervical cancer stem cells. Sci. Rep. 2024, 14, 13670. [Google Scholar] [CrossRef]
- Jiang, H.; Zuo, J.; Li, B.; Chen, R.; Luo, K.; Xiang, X.; Lu, S.; Huang, C.; Liu, L.; Tang, J.; et al. Drug-induced oxidative stress in cancer treatments: Angel or devil? Redox Biol. 2023, 63, 102754. [Google Scholar] [CrossRef]
- Joseph, A.; Dhanraj, K.; Ramalingam, S.; Saravanan, R. Treating cervical cancer with herbs: A review. Asian J. Pharm. Sci. 2018, 11, 4–10. [Google Scholar] [CrossRef]
- AL-Ishaq, R.K.; Overy, A.J.; Büsselberg, D. Phytochemicals and Gastrointestinal Cancer: Cellular Mechanisms and Effects to Change Cancer Progression. Biomolecules 2020, 10, 105. [Google Scholar] [CrossRef] [PubMed]
- Lande, P.S.; Adhao, V.S.; Ambhore, J.P.; Gaikwad, K.P.; Chandak, C.S.; Joge, L.P. Anticancer action of naturally occurring emodin for the controlling of cervical cancer. Explor. Target. Anti-Tumor Ther. 2023, 4, 690–698. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, L.; Wang, Y.; Wang, C.; Guo, Y.; Yuan, Z.; Jia, Y.; Li, P.; Sun, S.; Zhao, G. A highly selective red-emitting fluorescent probe and its micro-nano-assembly for imaging endogenous peroxynitrite (ONOO−) in living cells. Anal. Chim. Acta 2023, 1241, 340778. [Google Scholar] [CrossRef]
- Jain, S.; Patil, U. Phytochemical and pharmacological profile of Cassia tora Linn. An Overview. Indian J. Nat. Prod. Resour. 2010, 1, 430–437. [Google Scholar]
- He, M.; Xia, L.; Li, J. Potential Mechanisms of Plant-Derived Natural Products in the Treatment of Cervical Cancer. Biomolecules 2021, 11, 1539. [Google Scholar] [CrossRef]
- Jin, Y.; Wu, Q.; Pan, S.; Zhou, Q.; Liu, H.; Zhang, Q.; Zhang, J.; Zhu, X. Baicalein enhances cisplatin sensitivity in cervical cancer cells by promoting cuproptosis through the Akt pathway. Biomed. Pharmacother. 2024, 179, 117415. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.G.; Malek, E.; Choi, S.H.; Ignatz-Hoover, J.J.; Driscoll, J.J. Novel therapies emerging in oncology to target the TGF-β pathway. J. Hematol. Oncol. 2021, 14, 55. [Google Scholar] [CrossRef]
- Seoane, J.; Gomis, R.R. TGF-β Family Signalling in Tumour Suppression and Cancer Progression. Cold Spring Harb. Perspect. Biol. 2017, 9, a022277. [Google Scholar] [CrossRef]
- Oshimori, N.; Oristian, D.; Fuchs, E. TGF-β promotes heterogeneity and drug resistance in squamous cell carcinoma. Cell 2015, 160, 963–976. [Google Scholar] [CrossRef] [PubMed]
- Barouch-Bentov, R.; Sauer, K. Mechanisms of drug resistance in kinases. Expert Opin. Investig. Drugs. 2011, 20, 153–208. [Google Scholar] [CrossRef]
- Bou Antoun, N.; Chioni, A.M. Dysregulated Signalling Pathways Driving Anticancer Drug Resistance. Int. J. Mol. Sci. 2023, 24, 12222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wu, J.; Ling, M.T.; Zhao, L.; Zhao, K.N. The role of the PI3K/Akt/mTOR signalling pathway in human cancers induced by infection with human papillomaviruses. Mol. Cancer 2015, 14, 87. [Google Scholar] [CrossRef] [PubMed]
- Navaei, Z.N.; Khalili-Tanha, G.; Zangouei, A.S.; Abbaszadegan, M.R.; Moghbeli, M. PI3K/AKT signalling pathway as a critical regulator of Cisplatin response in tumour cells. Oncol. Res. 2022, 29, 235–250. [Google Scholar] [CrossRef]
- Kciuk, M.; Gielecińska, A.; Mujwar, S.; Kołat, D.; Kałuzińska-Kołat, Ż.; Celik, I.; Kontek, R. Doxorubicin—An Agent with Multiple Mechanisms of Anticancer Activity. Cells 2023, 12, 659. [Google Scholar] [CrossRef]
- Rascio, F.; Spadaccino, F.; Rocchetti, M.T.; Castellano, G.; Stallone, G.; Netti, G.S.; Ranieri, E. The Pathogenic Role of PI3K/AKT Pathway in Cancer Onset and Drug Resistance: An Updated Review. Cancers 2021, 13, 3949. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, X.; Jiang, L.; Zhang, L.; Xiang, M.; Ren, H. FoxM1 Induced Paclitaxel Resistance via Activation of the FoxM1/PHB1/RAF-MEK-ERK Pathway and Enhancement of the ABCA2 Transporter. Mol. Ther. Oncolytics 2019, 14, 196–212. [Google Scholar] [CrossRef]
- Yang, C.; Mai, Z.; Liu, C.; Yin, S.; Cai, Y.; Xia, C. Natural Products in Preventing Tumour Drug Resistance and Related Signalling Pathways. Molecules 2022, 27, 3513. [Google Scholar] [CrossRef]
- Ghasemi, F.; Shafiee, M.; Banikazemi, Z.; Pourhanifeh, M.H.; Khanbabaei, H.; Shamshirian, A.; Moghadam, S.A.; Aref Nezhad, R.; Sahebkar, A.; Avan, A.; et al. Curcumin inhibits NF-kB and Wnt/β-catenin pathways in cervical cancer cells. Pathol. Res. Pract. 2019, 215, 152556. [Google Scholar] [CrossRef]
- Kumar, A.; Kalra, S.; Kailash, J.; Vikas, J. Flavonoids as P-glycoprotein inhibitors for multidrug resistance in cancer: An in-silico approach. J. Biomol. Struct. Dyn. 2022, 41, 7627–7639. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Ding, X. Dioscin suppresses the viability of ovarian cancer cells by regulating the VEGFR2 and PI3K/AKT/MAPK signalling pathways. Oncol. Lett. 2018, 15, 9537–9542. [Google Scholar] [CrossRef] [PubMed]
- Shi, A.; Liu, L.; Li, S.; Qi, B. Natural products targeting the MAPK-signalling pathway in cancer: Overview. J. Cancer Res. Clin. Oncol. 2024, 150, 6. [Google Scholar] [CrossRef]
- Chen, G.; Yang, Z.; Wen, D.; Li, P.; Xiong, Q.; Wu, C. Oridonin Inhibits Mycobacterium marinum Infection-Induced Oxidative Stress In Vitro and In Vivo. Pathogens 2023, 12, 799. [Google Scholar] [CrossRef]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2018, 20, 69–84. [Google Scholar] [CrossRef]
- Seo, J.; Ha, J.; Kang, E.; Cho, S. The role of epithelial–mesenchymal transition-regulating transcription factors in anti-cancer drug resistance. Arch. Pharm. Res. 2021, 44, 281–292. [Google Scholar] [CrossRef]
- Huang, Y.; Hong, W.; Wei, X. The molecular mechanisms and therapeutic strategies of EMT in tumour progression and metastasis. J. Hematol. Oncol. 2022, 15, 129. [Google Scholar] [CrossRef]
- Saxena, M.; Stephens, M.A.; Pathak, H.; Rangarajan, A. Transcription factors that mediate epithelial-mesenchymal transition lead to multidrug resistance by upregulating ABC transporters. Cell Death Dis. 2011, 2, e179. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Shim, J. Targeting Epithelial–Mesenchymal Transition (EMT) to Overcome Drug Resistance in Cancer. Molecules 2016, 21, 965. [Google Scholar] [CrossRef]
- De Las Rivas, J.; Brozovic, A.; Izraely, S.; Casas-Pais, A.; Witz, I.P.; Figueroa, A. Cancer drug resistance induced by EMT: Novel therapeutic strategies. Arch. Toxicol. 2021, 95, 2279–2297. [Google Scholar] [CrossRef]
- Song, K.A.; Faber, A.C. Epithelial-to-mesenchymal transition and drug resistance: Transitioning away from death. J. Thorac. Dis. 2019, 11, 82–85. [Google Scholar] [CrossRef]
- Chauhan, R.; Malhotra, L.; Gupta, A.; Dagar, G.; Mendiratta, M.; Masoodi, T.; Hashem, S.; Al Marzooqi, S.; Das, D.; Uddin, S.; et al. Bergenin inhibits growth of human cervical cancer cells by decreasing Galectin-3 and MMP-9 expression. Sci. Rep. 2024, 14, 15287. [Google Scholar] [CrossRef]
- Shaikh, S.; Shaikh, J.; Naba, Y.S.; Doke, K.; Ahmed, K.; Yusufi, M. Curcumin: Reclaiming the lost ground against cancer resistance. Cancer Drug Resist. 2021, 4, 298–320. [Google Scholar] [CrossRef]
- Nunzio, A.C.; Cuciniello, R.; Gianluigi, D.P.; Piccioni, M.; Filosa, S.; Crispi, S. An Overview of the Enhanced Effects of Curcumin and Chemotherapeutic Agents in Combined Cancer Treatments. Int. J. Mol. Sci. 2023, 24, 12587. [Google Scholar] [CrossRef] [PubMed]
- Naujokat, C.; McKee, D.L. The ‘Big Five’ Phytochemicals Targeting Cancer Stem Cells: Curcumin, EGCG, Sulforaphane, Resveratrol and Genistein. Curr. Med. Chem. 2020, 28, 4321–4342. [Google Scholar] [CrossRef] [PubMed]
- Gottesman, M. Mechanisms of Cancer Drug Resistance. Annu. Rev. Med. 2002, 53, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.; Curry, E.; Magnani, L.; Wilhelm-Benartzi, C.; Borley, J. Poised epigenetic states and acquired drug resistance in cancer. Nat. Rev. Cancer 2014, 14, 747–753. [Google Scholar] [CrossRef]
- Lim, Z.; Ma, P. Emerging insights of tumour heterogeneity and drug resistance mechanisms in lung cancer targeted therapy. J. Hematol. Oncol. 2019, 12, 134. [Google Scholar] [CrossRef]
- Lumpp, T.; Stößer, S.; Fischer, F.; Hartwig, A.; Köberle, B. Role of Epigenetics for the Efficacy of Cisplatin. Int. J. Mol. Sci. 2024, 25, 1130. [Google Scholar] [CrossRef]
- Feng, C.; Dong, J.; Chang, W.; Cui, M.; Xu, T. The Progress of Methylation Regulation in Gene Expression of Cervical Cancer. Int. J. Genom. 2018, 2011, 8260652. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zhu, H.; Tian, M.; Wang, D.; He, J.; Xu, T. DNA Methylation and Hydroxymethylation in Cervical Cancer: Diagnosis, Prognosis and Treatment. Front. Genet. 2020, 11, 347. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.L.R.; De Albuquerque, B.H.D.R.; Allyrio, T.A.D.M.F.; De Almeida, V.D.; Cobucci, R.N.D.O.; Bezerra, F.L.; Andrade, V.S.; Lanza, D.C.F.; De Azevedo, J.C.V.; De Araújo, J.M.G.; et al. The role of HPV-induced epigenetic changes in cervical carcinogenesis (Review). Biomed. Rep. 2021, 15, 60. [Google Scholar] [CrossRef]
- Albulescu, A.; Plesa, A.; Fudulu, A.; Iancu, I.; Anton, G.; Botezatu, A. Epigenetic approaches for cervical neoplasia screening (Review). Exp. Ther. Med. 2021, 22, 1481. [Google Scholar] [CrossRef]
- Morale, M.G.; Tamura, R.E.; Cintra, R.; Araújo, N.M.; Villa, L.L. TLR4 and SARM1 modulate survival and chemoresistance in an HPV-positive cervical cancer cell line. Sci. Rep. 2022, 12, 6714. [Google Scholar] [CrossRef]
- Jiang, N.; Xie, F.; Guo, Q.; Li, M.Q.; Xiao, J.; Sui, L. Toll-like receptor 4 promotes proliferation and apoptosis resistance in human papillomavirus–related cervical cancer cells through the Toll-like receptor 4/nuclear factor-κB pathway. Tumor Biol. 2017, 39, 1010428317710586. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Lee, K.-D.; Pai, M.-Y.; Chu, P.-Y.; Hsu, C.-C.; Chiu, C.-C.; Chen, L.-T.; Chang, J.-Y.; Hsiao, S.-H.; Leu, Y.-W. Changes in DNA methylation are associated with the development of drug resistance in cervical cancer cells. Cancer Cell Int. 2015, 15, 98. [Google Scholar] [CrossRef]
- Dueñas-González, A.; Lizano, M.; Candelaria, M.; Cetina, L.; Arce, C.; Cervera, E. Epigenetics of cervical cancer. An overview and therapeutic perspectives. Mol. Cancer 2005, 4, 38. [Google Scholar] [CrossRef]
- Kang, K.; Hyun, J. Oxidative Stress, Nrf2, and Epigenetic Modification Contribute to Anticancer Drug Resistance. Toxicol. Res. 2017, 33, 1–5. [Google Scholar] [CrossRef]
- Guo, L.; Lee, Y.T.; Zhou, Y.; Huang, Y. Targeting epigenetic regulatory machinery to overcome cancer therapy resistance. Semin. Cancer Biol. 2021, 83, 487–502. [Google Scholar] [CrossRef]
- Kang, K.A.; Piao, M.J.; Kim, K.C.; Kang, H.K.; Chang, W.Y.; Park, I.C.; Keum, Y.S.; Surh, Y.J.; Hyun, J.W. Epigenetic modification of Nrf2 in 5-fluorouracil-resistant colon cancer cells: Involvement of TET-dependent DNA demethylation. Cell Death Dis. 2014, 5, e1183. [Google Scholar] [CrossRef]
- Jeong, H.M.; Kwon, M.J.; Shin, Y.K. Overexpression of Cancer-Associated Genes via Epigenetic Derepression Mechanisms in Gynecologic Cancer. Front. Oncol. 2014, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, D.; Soreq, H. Alternative Splicing. In Brenner’s Encyclopedia of Genetics; Elsevier Inc.: Amsterdam, The Netherlands, 2013; pp. 97–98. [Google Scholar]
- de Necochea-Campion, R.; Shouse, G.; Zho, Q.; Mirshahidi, S.; Chen, C. Aberrant splicing and drug resistance in AML. J. Hematol. Oncol. 2016, 9, 85. [Google Scholar] [CrossRef][Green Version]
- Hughes, P.; Bouillet, P.; Strasser, A. Role of Bim and other Bcl-2 Family Members in Autoimmune and Degenerative Diseases. In Current Directions in Autoimmunity; Karger Publisher: Basel, Switzerland, 2005; pp. 74–94. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, R.; Li, P.; Zheng, D.; Zhou, J.; Wang, J.; Zhang, B.; Zhu, C. Traditional Chinese Medicine: Salvia miltiorrhiza Enhances Survival Rate of Autologous Adipose Tissue Transplantation in Rabbit Model. Aesthetic Plast. Surg. 2015, 39, 985–991. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Xu, Y.M.; Lau, A.T.Y. Epigenetic effects of herbal medicine. Clin. Epigenet. 2023, 15, 85. [Google Scholar] [CrossRef]
- Skała, E.; Toma, M.; Kowalczyk, T.; Tomasz, Ś.; Sitarek, P. Rhaponticum carthamoides transformed root extract inhibits human glioma cells viability, induces double strand DNA damage, H2A.X phosphorylation, and PARP1 cleavage. Cytotechnology 2018, 70, 1585–1594. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wang, J.; Momeni, M.R. Epigallocatechin-3-gallate and its nanoformulation in cervical cancer therapy: The role of genes, MicroRNA and DNA methylation patterns. Cancer Cell Int. 2023, 23, 335. [Google Scholar] [CrossRef] [PubMed]
- Carlos-Reyes, Á.; López-González, J.S.; Meneses-Flores, M.; Gallardo-Rincón, D.; Ruíz-García, E.; Marchat, L.A.; la Vega, H.A.-D.; de la Cruz, O.N.H.; López-Camarillo, C. Dietary Compounds as Epigenetic Modulating Agents in Cancer. Front. Genet. 2019, 10, 79. [Google Scholar] [CrossRef]
- Pal, A.; Kundu, R. Human Papillomavirus E6 and E7: The Cervical Cancer Hallmarks and Targets for Therapy. Front. Microbiol. 2020, 10, 3116. [Google Scholar] [CrossRef]
- Divya, C.S.; Pillai, M.R. Antitumor action of curcumin in human papillomavirus associated cells involves downregulation of viral oncogenes, prevention of NFkB and AP-1 translocation, and modulation of apoptosis. Mol. Carcinog. 2006, 45, 320–332. [Google Scholar] [CrossRef]
- Munagala, R.; Aqil, F.; Jeyabalan, J.; Gupta, R.C. Tanshinone IIA inhibits viral oncogene expression leading to apoptosis and inhibition of cervical cancer. Cancer Lett. 2015, 356, 536–546. [Google Scholar] [CrossRef]
- Tian, Y.; Yuan, L.; Wang, Y.; Lai, J.; Wang, J.; Xia, F. Mechanism of multidrug resistance to chemotherapy mediated by P-glycoprotein (Review). Int. J. Oncol. 2023, 63, 119. [Google Scholar] [CrossRef]
- Choi, Y.; Yu, A. ABC Transporters in Multidrug Resistance and Pharmacokinetics, and Strategies for Drug Development. Curr. Pharm. Des. 2014, 20, 793–807. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ling, K.; Zhu, Y.; Deng, L.; Li, Y.; Liang, Z. Rucaparib antagonize multidrug resistance in cervical cancer cells through blocking the function of ABC transporters. Gene 2020, 759, 145000. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.-D.; Zhang, M.; Cai, C.-Y.; Teng, Q.-X.; Wang, J.-Q.; Fu, Y.-G.; Cui, Q.; Patel, K.; Wang, D.-T.; Chen, Z.-S. Overexpression of ABCB1 associated with the resistance to the kras-G12C specific inhibitor ARS-1620 in cancer cells. Front. Pharmacol. 2022, 13, 843829. [Google Scholar] [CrossRef]
- Zhu, X.; Zhu, H.; Luo, H.; Zhang, W.; Shen, Z.; Hu, X. Molecular mechanisms of cisplatin resistance in cervical cancer. Drug Des. Dev. Ther. 2016, 10, 1885–1895. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; König, J.; Buchholz, J.K.; Spring, H.; Leier, I.; Keppler, D. Drug resistance and ATP-dependent conjugate transport mediated by the apical multidrug resistance protein, MRP2, permanently expressed in human and canine cells. Mol. Pharmacol. 1999, 55, 926–937. [Google Scholar] [CrossRef]
- Wen, C.; Fu, L.; Huang, J.; Dai, Y.; Wang, B.; Xu, G.; Wu, L.; Zhou, H. Curcumin reverses doxorubicin resistance via inhibition the efflux function of ABCB4 in doxorubicin-resistant breast cancer cells. Mol. Med. Rep. 2019, 19, 5162–5168. [Google Scholar] [CrossRef]
- Morris, M.E.; Zhang, S. Flavonoid–drug interactions: Effects of flavonoids on ABC transporters. Life Sci. 2006, 78, 2116–2130. [Google Scholar] [CrossRef]
- Soejima, H.; Zhao, W.; Mukai, T. Epigenetic silencing of the MGMT gene in cancer. Biochem. Cell Biol. 2005, 83, 429–437. [Google Scholar] [CrossRef]
- Stucci, L.S.; Internò, V.; Tucci, M.; Perrone, M.; Mannavola, F.; Palmirotta, R.; Porta, C. The ATM gene in breast cancer: Its relevance in clinical practice. Genes 2021, 12, 727. [Google Scholar] [CrossRef]
- Rodrigues, A.S.; Gomes, B.C.; Martins, C.; Gromicho, M.; Oliveira, N.G.; Guerreiro, P.S.; Rueff, J. DNA Repair and Resistance to Cancer Therapy. In New Research Directions in DNA Repair; OpenIntech: London, UK, 2013; pp. 1–674. [Google Scholar] [CrossRef][Green Version]
- Chatterjee, N.; Walker, G. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagen. 2017, 58, 235–263. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, E.; Das, S.; Biswas, S.; Ghosh, A.; Sengupta, D.; Roy, M.; Mukherjee, S. Insights of Cisplatin Resistance in Cervical Cancer: A Decision Making for Cellular Survival. In Cervical Cancer—A Global Public Health Treatise; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Winterman, S.; Playe, M.; Benbara, A.; Zelek, L.; Pamoukdjian, F.; Bousquet, G. Dose-intense cisplatin-based neoadjuvant chemotherapy increases survival in advanced cervical cancer: An up-to-date meta-analysis. Cancers 2022, 14, 842. [Google Scholar] [CrossRef]
- Li, L.Y.; Guan, Y.; Chen, X.S.; Yang, J.M.; Cheng, Y. DNA Repair Pathways in Cancer Therapy and Resistance. Front. Pharmacol. 2020, 11, 629266. [Google Scholar] [CrossRef]
- Zhang, N.; Yin, Y.; Xu, S.J.; Chen, W.S. 5-Fluorouracil: Mechanisms of Resistance and Reversal Strategies. Molecules 2008, 13, 1551–1569. [Google Scholar] [CrossRef]
- Gilbert, D.C.; Chalmers, A.J.; El-Khamisy, S.F. Topoisomerase I inhibition in colorectal cancer: Biomarkers and therapeutic targets. Br. J. Cancer 2011, 106, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Lagunas-Rangel, F.A.; Bermúdez-Cruz, R.M. Natural Compounds That Target DNA Repair Pathways and Their Therapeutic Potential to Counteract Cancer Cells. Front. Oncol. 2020, 10, 598174. [Google Scholar] [CrossRef]
- Wang, L.; Long, L.; Wang, W.; Liang, Z. Resveratrol, a potential radiation sensitizer for glioma stem cells both in vitro and in vivo. J. Pharmacol. Sci. 2015, 129, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Druker, B.J.; Sawyers, C.L.; Kantarjian, H.; Resta, D.J.; Reese, S.F.; Ford, J.M.; Capdeville, R.; Talpaz, M. Activity of a Specific Inhibitor of the BCR-ABL Tyrosine Kinase in the Blast Crisis of Chronic Myeloid Leukemia and Acute Lymphoblastic Leukemia with the Philadelphia Chromosome. N. Engl. J. Med. 2001, 344, 1038–1042. [Google Scholar] [CrossRef]
- Stewart, D.J. Mechanisms of resistance to Cisplatin and Carboplatin. Crit. Rev. Oncol. Hematol. 2007, 63, 12–31. [Google Scholar] [CrossRef]
- Chai, J.Y.; Sugumar, V.; Alshanon, A.F.; Wong, W.F.; Fung, S.Y.; Looi, C.Y. Defining the role of GLI/hedgehog signalling in chemoresistance: Implications in therapeutic approaches. Cancers 2021, 13, 4746. [Google Scholar] [CrossRef]
- Zahreddine, H.; Borden, K.L.B. Mechanisms and insights into drug resistance in cancer. Front. Pharmacol. 2013, 4, 28. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, X.; Wang, Y.; Chen, Y.; Lu, H.; Meng, X.; Ye, X.; Chen, W. Activation/Inactivation of Anticancer Drugs by CYP3A4: Influencing Factors for Personalized Cancer Therapy. Drug Metab. Dispos. 2023, 51, 543–559. [Google Scholar] [CrossRef] [PubMed]
- Das, S. Phytochemicals Modify the Action of Cancer Cells Mitochondrial Drug-Resistance Mechanism. Sci. Pharm. 2023, 2, 9–105. [Google Scholar] [CrossRef]
- Zou, J.-Y.; Chen, Q.-L.; Luo, X.-C.; Damdinjav, D.; Abdelmohsen, U.R.; Li, H.-Y.; Battulga, T.; Chen, H.-B.; Wang, Y.-Q.; Zhang, J.-Y. Natural products reverse cancer multidrug resistance. Front. Pharmacol. 2024, 15, 1348076. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.T. Phytochemicals and cancer. Proc. Nutr. Soc. 2007, 66, 207–215. [Google Scholar] [CrossRef]
- Kwon, K.T.; Barve, A.; Yu, S.; Huang, M.T.; Kong, A.N.T. Cancer chemoprevention by phytochemicals: Potential molecular targets, biomarkers and animal models. Acta Pharm. Sin. 2007, 28, 1409–1421. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, C.; Huang, Y.; Lam, C.W.; Chow, M.S. Overcoming chemotherapy resistance with herbal medicines: Past, present and future perspectives. Phytochem. Rev. 2013, 13, 323–337. [Google Scholar] [CrossRef]
- Mbaveng, A.T.; Manekeng, H.T.; Nguenang, G.S.; Dzotam, J.K.; Kuete, V.; Efferth, T. Cytotoxicity of 18 Cameroonian medicinal plants against drug sensitive and multi-factorial drug resistant cancer cells. J. Ethnopharmacol. 2018, 222, 21–33. [Google Scholar] [CrossRef]
- Hull, R.; Mabeta, P.; Chauke-Malinga, N.; Molefi, T.; Chatziioannou, A.; Dlamini, Z. Reversal of resistance mechanisms in cervical cancer. In Strategies for Overcoming Chemotherapy Resistance in Cervical Cancer; Elsevier: Amsterdam, The Netherlands, 2024; pp. 71–93. [Google Scholar] [CrossRef]
- Jamshidi-Kia, F.; Lorigooini, Z.; Amini-Khoei, H. Medicinal plants: Past history and future perspective. J. Herbmed Pharmacol. 2018, 7, 1–7. [Google Scholar] [CrossRef]
- Sponchiado, G.; Adam, M.L.; Silva, C.D.; Soley, B.S.; de Mello-Sampayo, C.; Cabrini, D.A.; Correr, C.J.; Otuki, M.F. Quantitative genotoxicity assays for analysis of medicinal plants: A systematic review. J. Ethnopharmacol. 2016, 178, 289–296. [Google Scholar] [CrossRef]
- Monteiro, L.D.; Bastos, K.X.; Barbosa-Filho, J.M.; de Athayde-Filho, P.F.; Diniz, M.D.; Sobral, M.V. Medicinal plants and other living organisms with antitumour potential against lung cancer. Evid. Based Complement. Altern. Med. 2014, 2014, 604152. [Google Scholar] [CrossRef]
- Rizeq, B.; Gupta, I.; Ilesanmi, J.; AlSafran, M.; Rahman, M.M.; Ouhtit, A. The Power of Phytochemicals Combination in Cancer Chemoprevention. J. Cancer 2020, 11, 4521–4533. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Cheung, M. Combination of Phytochemicals as Adjuvants for Cancer Therapy. Recent Pat. Anticancer. Drug Discov. 2014, 9, 297–302. [Google Scholar] [CrossRef]
- Ayaz, M.; Nawaz, A.; Ahmad, S.; Mosa, O.F.; Hamdoon, A.A.E.; Khalifa, M.A.; Sadiq, A.; Ullah, F.; Wadood, A.; Kabra, A.; et al. Underlying Anticancer Mechanisms and Synergistic Combinations of Phytochemicals with Cancer Chemotherapeutics: Potential Benefits and Risks. J. Food Qual. 2022, 2022, 1189034. [Google Scholar] [CrossRef]
- Lino, J.; Nyamai, D.W.; Njeru, S.N. Phytochemical analysis, in-vitro and in-silico study of antiproliferative activity of ethyl acetate fraction of Launaea cornuta (Hochst. ex Oliv. & Hiern) C. Jeffrey against human cervical cancer cell line. Front. Pharmacol. 2024, 15, 1399885. [Google Scholar] [CrossRef]
- Kesarwani, K.; Gupta, R. Bioavailability enhancers of herbal origin: An overview. Asian Pac. J. Trop. Biomed. 2013, 3, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; da Silva Pinto, M. Bioavailability of bioactive food compounds: A challenging journey to bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602. [Google Scholar] [CrossRef]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Vadhanam, M.V. Bioavailability of phytochemicals and its enhancement by drug delivery systems. Cancer Lett. 2013, 334, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Funk, J.L.; Schneider, C. Perspective on Improving the Relevance, Rigor, and Reproducibility of Botanical Clinical Trials: Lessons Learned From Turmeric Trials. Front. Nutr. 2021, 8, 782912. [Google Scholar] [CrossRef]
- Sangho, A.; Ouoba, K.; Dori, D.; Sanogo, R.; Semdé, R. Regulation and practice of clinical trials of herbal medicines in French-speaking West African countries (WAEMU): Current status and challenges. PAMJ One Health 2024, 15, 8. [Google Scholar] [CrossRef]
- Parveen, A.; Parveen, B.; Parveen, R.; Ahmad, S. Challenges and guidelines for clinical trial of herbal drugs. J. Pharm. Bioallied. Sci. 2015, 7, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, J.; He, L.; Liu, L.; Cheng, B.; Zhou, F.; Cao, D.; He, Y. A Comprehensive Review on the Benefits and Problems of Curcumin with Respect to Human Health. Molecules 2022, 27, 4400. [Google Scholar] [CrossRef]
- Panossian, A. Challenges in phytotherapy research. Front. Pharmacol. 2023, 14, 1199516. [Google Scholar] [CrossRef]
- Rudzińska, A.; Juchaniuk, P.; Oberda, J.; Wiśniewska, J.; Wojdan, W.; Szklener, K.; Mańdziuk, S. Phytochemicals in Cancer Treatment and Cancer Prevention—Review on Epidemiological Data and Clinical Trials. Nutrients 2023, 15, 1896. [Google Scholar] [CrossRef] [PubMed]
- Asano, T. Investigating the Pharmacokinetics of Natural Products: Challenges and Advances in Bioavailability. J. Pharmacogn. Nat. Prod. 2024, 10, 326. [Google Scholar] [CrossRef]
- Patwardhan, B.; Mashelkar, R.A. Traditional medicine-inspired approaches to drug discovery: Can Ayurveda show the way forward? Drug Discov. Today 2009, 14, 804–811. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Curcumin Adjunct to Chemoradiotherapy in Cervical Cancer; Identifier NCT06080841; National Library of Medicine (US): Bethesda, MD, USA, 2023. Available online: https://clinicaltrials.gov/study/NCT06080841 (accessed on 12 January 2026).
- ClinicalTrials.gov. Curcumin Vaginal Capsule for Cervical Intraepithelial Neoplasia; Identifier NCT02554344; National Library of Medicine (US): Bethesda, MD, USA, 2015. Available online: https://clinicaltrials.gov/study/NCT02554344 (accessed on 12 January 2026).
- ClinicalTrials.gov. Curcumin in Advanced Pancreatic Cancer; Identifier NCT00192842; National Library of Medicine (US): Bethesda, MD, USA, 2005. Available online: https://clinicaltrials.gov/study/NCT00192842 (accessed on 12 January 2026).
- ClinicalTrials.gov. Curcumin in Breast Cancer Patients; Identifier NCT01740323; National Library of Medicine (US): Bethesda, MD, USA, 2012. Available online: https://clinicaltrials.gov/study/NCT01740323 (accessed on 12 January 2026).
- ClinicalTrials.gov. Green Tea Extract in Prostate Cancer Prevention; Identifier NCT00596011; National Library of Medicine (US): Bethesda, MD, USA, 2007. Available online: https://clinicaltrials.gov/study/NCT00596011 (accessed on 12 January 2026).
- ClinicalTrials.gov. Resveratrol in Colon Cancer Prevention; Identifier NCT00256334; National Library of Medicine (US): Bethesda, MD, USA, 2005. Available online: https://clinicaltrials.gov/study/NCT00256334 (accessed on 12 January 2026).
- ClinicalTrials.gov. Diindolylmethane in Breast Cancer Prevention; Identifier NCT01391689; National Library of Medicine (US): Bethesda, MD, USA, 2011. Available online: https://clinicaltrials.gov/study/NCT01391689 (accessed on 12 January 2026).
- ClinicalTrials.gov. Indole-3-Carbinol in Cervical Intraepithelial Neoplasia; Identifier NCT00988845; National Library of Medicine (US): Bethesda, MD, USA, 2009. Available online: https://clinicaltrials.gov/study/NCT00988845 (accessed on 12 January 2026).
- ClinicalTrials.gov. Active Hexose Correlated Compound (AHCC) for HPV Clearance; Identifier NCT02405533; National Library of Medicine (US): Bethesda, MD, USA, 2015. Available online: https://clinicaltrials.gov/study/NCT02405533 (accessed on 12 January 2026).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.