Insect and Plant Diversity in Hot-Spring Ecosystems during the Jurassic-Cretaceous Boundary from Spain (Aguilar Fm., Palencia)
Abstract
:Simple Summary
Abstract
1. Introduction
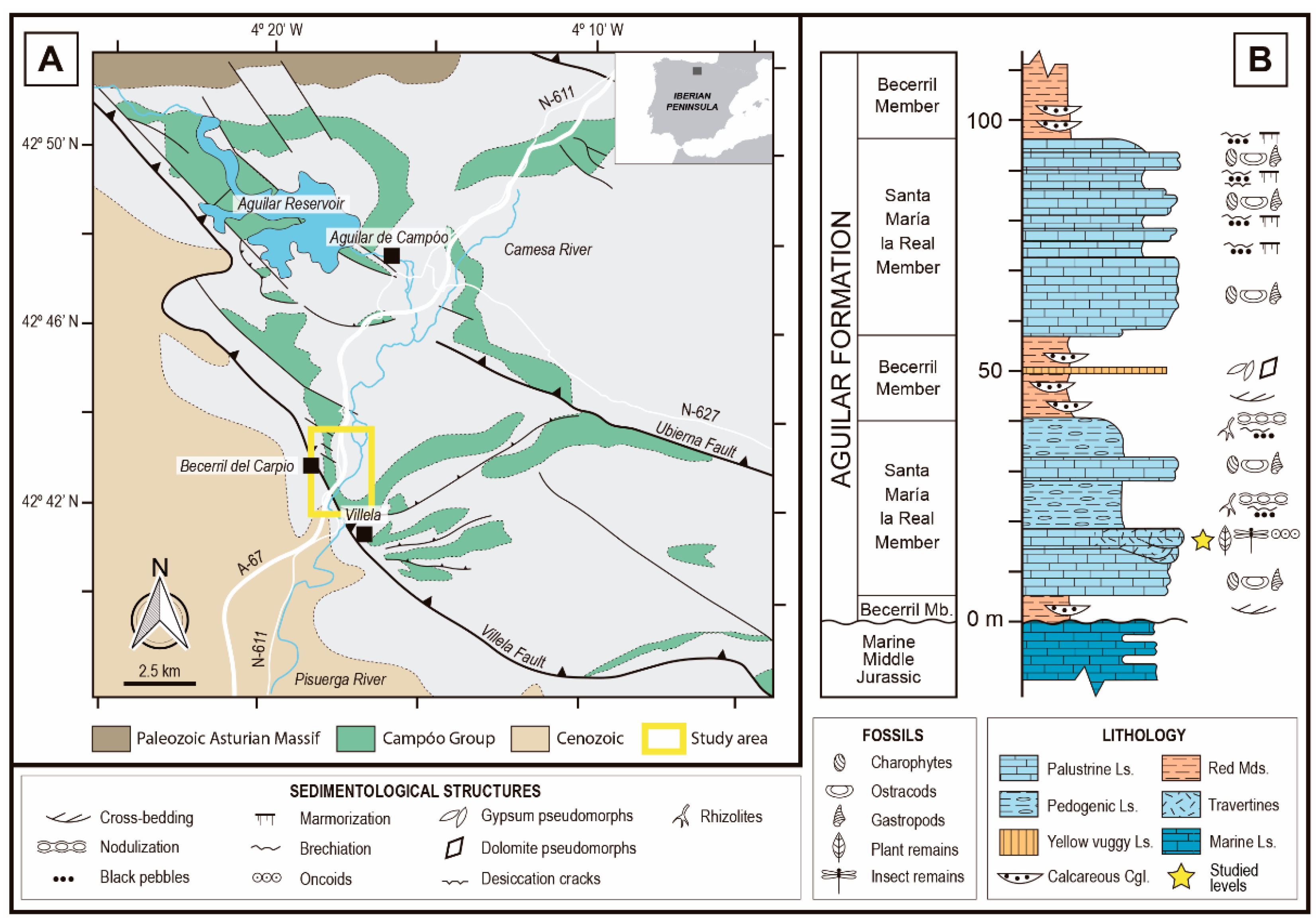
2. Geographic and Geological Setting
3. Material and Methods
4. Systematic Palaeontology
4.1. Insects

4.2. Plants


5. Discussion
5.1. Entomofauna
5.2. Palaeobotany: Palaeoenvironmental, Taphonomical, and Palaeogeographical Implications
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mora, C.; Tittensor, D.P.; Adl, S.; Simpson, A.G.; Worm, B. How many species are there on Earth and in the ocean? PLoS Biol. 2011, 9, e1001127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.Q. Animal Biodiversity: An Outline of Higher-Level Classification and Survey of Taxonomic Richness; Zootaxa: Auckland, New Zealand, 2013; 82p. [Google Scholar]
- Roskov, Y.; Kunze, T.; Paglinawan, L.; Orrell, T.; Nicolson, D.; Culham, A.; Bailly, N.; Kirk, P.; Bourgoin, T.; Baillargeon, G.; et al. Species 2000 & ITIS Catalogue of Life. Available online: www.catalogueoflife.org/col/ (accessed on 11 March 2013).
- Stork, N.E. How many species of insects and other terrestrial arthropods are there on Earth? Annu. Rev. Entomol. 2018, 63, 31–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bar-On, Y.M.; Phillips, R.; Milo, R. The biomass distribution on Earth. Proc. Natl. Acad. Sci. USA 2018, 115, 6506–6511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Channing, A.; Zamuner, A.; Edwards, D.; Guido, D. Equisetum thermale sp. nov. (Equisetales) from the Jurassic San Agustín hot spring deposit, Patagonia: Anatomy, paleoecology, and inferred paleoecophysiology. Am. J. Bot. 2011, 98, 680–697. [Google Scholar] [CrossRef]
- Guido, D.M.; Campbell, K.A.M.; Foucher, F.; Westall, F. Life is everywhere in sinters: Examples from Jurassic hot-spring environments of Argentine Patagonia. Geol. Mag. 2019, 156, 1631–1638. [Google Scholar] [CrossRef]
- Aksenova, O.V.; Potapov, G.S.; Bespalaya, Y.V.; Kolosova, Y.S.; Vikhrev, I.V.; Kondakov, A.V.; Gofarov, M.Y.; Bolotov, I.N. Dragonflies from hot springs in Russia with a country-level checklist of species known to occur in geothermal environments. Ecol. Montenegrina 2020, 34, 49–63. [Google Scholar] [CrossRef]
- Corbet, P.S. Dragonflies: Behavior and Ecology of Odonata; Harley Books: Colchester, UK, 2004; 829p. [Google Scholar]
- Cox, A.; Shock, E.L.; Havig, J.R. The transition to microbial photosynthesis in hot spring ecosystems. Chem. Geol. 2011, 280, 344–351. [Google Scholar] [CrossRef]
- O’Gorman, E.J.; Pichler, D.E.; Adams, G.; Benstead, J.P.; Cohen, H.; Craig, N.; Cross, W.F.; Demars, B.; Friberg, N.; Gislason, G.M.; et al. Impacts of warming on the structure and functioning of aquatic communities: Individual-to ecosystem-level responses. Adv. Ecol. Res. 2012, 47, 81–176. [Google Scholar]
- Pleshanov, A.S.; Pleshanova, G.I.; Shamanova, S.I. Landscape-climatic patterns of spatial distribution of refugia in the Baikal Region. Contemp. Probl. Ecol. 2002, 5, 603–610. (In Russian) [Google Scholar]
- Kornobis, E.; Pálsson, S.; Kristjansson, B.K.; Svavarsson, J. Molecular evidence of the survival of subterranean amphipods (Arthropoda) during Ice Age underneath glaciers in Iceland. Mol. Ecol. 2010, 19, 2516–2530. [Google Scholar] [CrossRef]
- Bolotov, I.N.; Bespalaya, Y.V.; Usacheva, O.V. Ecology and evolution of hydrobionts in hot springs of the Subarctic and Arctic: Formation of similar assemblages, adaptation of species, and microevolutionary processes. Biol. Bull. Rev. 2012, 2, 340–348. [Google Scholar] [CrossRef]
- Jumawan, K.; Medina, M.; Villanueva, R.J. Annotated list of Odonata from Mainit Hot Spring Protected Landscape, Compostella Valley, Mindanao Island, Philippines. Philipp. J. Syst. Biol. 2012, 6, 14–27. [Google Scholar]
- Borisov, S.N. Dragonflies (Odonata) of thermal springs in Central Asia. Entomol. Rev. 2015, 95, 1203–1211. [Google Scholar] [CrossRef]
- Ramírez del Pozo, J. Algunas observaciones sobre el Jurásico de Álava, Burgos y Santander. Cuad. Geol. Iber. 1972, 4, 491–508. [Google Scholar]
- Schaaf, D. Der Jura der Kantabrischen Ketten (Nordspanien). Genese und Evolution eines Speziellen Marinen Sedimentationsraumes. Master’s Thesis, Universität Tübingen, Tübingen, Germany, 1986; p. 190. [Google Scholar]
- Schudack, M. Charophytenflora und fazi elle Entwicklung der Grenzschichten mariner Jura/Wealden in den Nordwestlichen Iberischen Ketten (mit Vergleichen zu Asturien und Kantabrien). Palaeontogr. Abt. B Palaeophytol. 1987, 204, 1–180. [Google Scholar]
- Hernández, J.M.; Diéguez, C.; Pujalte, V.; Robles, S.; Wright, V.P. Reconocimiento de acumulaciones travertínicas fósiles en la Fm. A guilar (Kimmeridgiense-Berriasiense de Palencia y Burgos): Implicaciones paleoecológicas y paleohidrológicas. Geogaceta 1998, 24, 167–170. [Google Scholar]
- Hernández, J.M.; Pujalte, V.; Robles, S.; Martin-Closas, C. División estratigráfica genética del Grupo Campóo (Malm-Cretácico inferior, SW Cuenca Vascocantábrica). Rev. Soc. Geol. Esp. 1999, 12, 377–396. [Google Scholar]
- Hernández, J.M. Sedimentología, Paleogeografía Y Relaciones Tectónica/Sedimentación de Los Sistemas Fluviales, Aluviales y Palustres de la Cuenca Rift Aguilar (Grupo Campóo, Jurásico superior-Cretácico Inferior de Palencia, Burgos y Cantabria). Ph.D. Thesis, Universidad del País Vasco, País Vasco, Spain, 2000; p. 324, unpublished. [Google Scholar]
- Diéguez, C.; Hernández, J.M.; Pujalte, V. A fern-bennettitalean assemblage in Tithonian-Berriasian travertine deposits (Aguilar Formation, Burgos-Palencia, N Spain) and its palaeoclimatic and vegetational implications. J. Iber. Geol. 2009, 35, 127–140. [Google Scholar]
- García-Ramos, J.C.; Piñuela, L.; Uzqueda, H.; Poblet, J.; Bulnes, M.; Alonso, J.L.; Suárez-Verga, L.C. Travertinos ricos en Oncoides Asociados a Paleomanantiales y Lagos Efímeros Próximos a Fallas Sinsedimentarias en el Jurásico Superior de Asturias. In Proceedings of the V Congreso del Jurásico de España, Colunga, Spain, 8–11 September 2010; pp. 83–91. [Google Scholar]
- Arenas, C.; Piñuela, L.; García-Ramos, J.C. Climatic and tectonic controls on carbonate deposition in syn-rift siliciclastic fluvial systems: A case of microbialites and associated facies in the Late Jurassic. Sedimentology 2015, 62, 1149–1183. [Google Scholar] [CrossRef] [Green Version]
- Lozano, R.P.; Delvene, G.; Piñuela, L.; García-Ramos, J.C. Late Jurassic biogeochemical microenvironments associated with microbialite-coated unionids (Bivalvia), Asturias (N Spain). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2016, 443, 80–97. [Google Scholar] [CrossRef]
- Echeveste, H. Travertines and jasperoids of the Manantial Espejo, a Jurassic hot spring environment. Macizo del Deseado, Santa Cruz province, Argentina. Lat. Am. J. Sedimentol. Basin Anal. 2015, 12, 23–39. [Google Scholar]
- Channing, A.; Zamuner, A.B.; Zuniga, A. A new Middle–Late Jurassic flora and hot spring chert deposit from the Deseado Massif, Santa Cruz province, Argentina. Geol. Mag. 2007, 144, 401–411. [Google Scholar] [CrossRef]
- Guido, D.M.; Campbell, K.A. Jurassic hot-spring activity in a fluvial setting at La Marciana, Patagonia, Argentina. Geol. Mag. 2009, 146, 617–622. [Google Scholar] [CrossRef] [Green Version]
- Guido, D.M.; Channing, A.; Campbell, K.A.; Zamuner, A. Jurassic geothermal landscapes and fossil ecosystems at San Agustín, Patagonia, Argentina. Geol. Soc. Spec. Publ. 2010, 167, 11–20. [Google Scholar] [CrossRef]
- Guido, D.M.; Campbell, K.A. Jurassic hot spring deposits of the Deseado Massif (Patagonia, Argentina): Characteristics and controls on regional distribution. J. Volcanol. Geotherm. Res. 2011, 203, 35–47. [Google Scholar] [CrossRef]
- Guido, D.M.; Campbell, K.A. Diverse subaerial and sublacustrine hot spring settings of the Cerro Negro epithermal system (Jurassic, Deseado Massif), Patagonia, Argentina. J. Volcanol. Geotherm. Res. 2012, 229, 1–12. [Google Scholar] [CrossRef]
- García-Massini, J.G.; Falaschi, P.; Zamuner, A.B. Fungal–arthropod–plant interactions from the Jurassic petrified forest Monumento Natural Bosques Petrificados, Patagonia, Argentina. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2012, 329, 37–46. [Google Scholar] [CrossRef]
- Parrish, J.T.; Falcon-Lang, H.J. Coniferous trees associated with interdune deposits in the Jurassic Navajo Sandstone Formation, Utah, USA. Palaeontology 2007, 50, 829–843. [Google Scholar] [CrossRef]
- Whiteside, J.H.; Olsen, P.E. Arboreal Stromatolites from the Triassic and Jurassic of Eastern North America: Implications for Environmental Change. In Geological Society of America, Proceedings of the Northeastern Section—40th Annual Meeting, Salt Lake City, UT, USA, 14–16 March 2005; Geological Society of America: Boulder, CO, USA, 2005; p. 8. [Google Scholar]
- De Wet, C.C.B.; Hubert, J.F. The Scots Bay Formation, Nova Scotia, Canada, a Jurassic carbonate lake with silica-rich hydrothermal springs. Sedimentology 1989, 36, 857–873. [Google Scholar] [CrossRef]
- Steinen, R.P.; Gray, N.H.; Mooney, J. A Mesozoic carbonate hot-spring deposit in the Hartford Basin of Connecticut. J. Sediment. Res. 1987, 57, 319–326. [Google Scholar]
- Nel, A.; Blot, P. Paléoentomologie de la paléotufière éocène de Sézanne (Marne, France) (Insectes Odonata, Trichoptera, Hemiptera, Diptera). Entomol. Gall. 1990, 2, 26–31. [Google Scholar]
- O’Flynn, R.J.; Audo, D.; Kawai, T. Systematic revision and palaeobiology of Emplastron edwardsi (Van Straelen, 1928) gen. et comb. nov. (Crustacea, Decapoda, Astacidae) entombed within travertine, from Sézanne, France. Paleontol. Res. 2021, 25, 379–388. [Google Scholar] [CrossRef]
- Zeuner, F.E. Die Insektenfauna des Böttinger Marmors. Eine systematische und paläobiologische Studie. Fortschrit. Geol. Paläontol. 1931, 9, 247–406. [Google Scholar]
- Schwenkel, H. Der Böttinger Marmor, ein Naturdenkmal. Jahrb. Staatl. Mus. Nat. 1935, 12, 142–166. [Google Scholar]
- Bleich, K.E. Soil displacement with regard to calcareous deposits and fossil accumulations in the “Böttinger Marmor” (Swabian Alb, Southwestern Germany). Jahresh. Ver. Vaterl. Naturkd. Wb. 1998, 154, 43–50. [Google Scholar]
- Pujalte, V.; Hernández, J.M.; Robles, S. Control tectónico de los ciclos fluvio-palustres del Grupo Campóo en la Cuenca de Aguilar (Titoniense superior-Berriasiense, Palencia). Geogaceta 2015, 58, 19–22. [Google Scholar]
- Mendes, M.M.; Friis, E.M.; Pais, J. Erdtmanispermum juncalense sp. nov., a new species of the extinct order Erdtmanithecales from the Early Cretaceous (probably Berriasian) of Portugal. Rev. Palaeobot. Palynol. 2008, 149, 50–56. [Google Scholar] [CrossRef]
- Mendes, M.M.; Pais, J.; Friis, E.M. Raunsgaardispermum lusitanicum gen. et sp. nov., a new seed with in situ pollen from the Early Cretaceous (probably Berriasian) of Portugal: Further support for the Bennettitales-Erdtmanithecales-Gnetales link. Grana 2008, 47, 211–219. [Google Scholar] [CrossRef] [Green Version]
- Mendes, M.M.; Dinis, J.; Pais, J. Lower Cretaceous Pollen–Spore and Mesofossil Associations of the Bombarral Formation (Lusitanian Basin, Western Portugal). In STRATI 2013, 1st ed.; Rocha, R., Pais, J., Kullberg, J.C., Finney, S., Eds.; Springer: Cham, Switzerland, 2014; pp. 1129–1133. [Google Scholar]
- Santos, A.A.; Villanueva-Amadoz, U.; Royo-Torres, R.; Sender, L.M.; Cobos, A.; Alcala, L.; Diez, J.B. Palaeobotanical records associated with the first dinosaur defined in Spain: Palynostratigraphy, taxonomy and palaeoenvironmental remarks. Cretac. Res. 2018, 90, 318–334. [Google Scholar] [CrossRef]
- Dilcher, D.L.; Hill, C.R. A heterophyllous fern from the Lower Cretaceous of Northern Spain. Cour. Forsch. Inst. Senckenberg. 2003, 241, 111–118. [Google Scholar]
- Sillitoe, R.H. Epithermal models—Genetic types, geometric controls and shallow features. Geol. Soc. Am. Spec. Pap. 1993, 40, 403–417. [Google Scholar]
- Santos, A.A.; Sender, L.M.; Wappler, T.; Engel, M.S.; Diez, J.B. A Robinson Crusoe story in the fossil record: Plant-insect interactions from a Middle Jurassic ephemeral volcanic island (Eastern Spain). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2021, 583, 110655. [Google Scholar] [CrossRef]
- Martinez-Delclos, X. Chresmoda aquatica n. sp. insecto Chresmodidae del Cretácico inferior de la Sierra del Montsec (Lleida, España). Rev. Esp. Paleontol. 1989, 4, 67–74. [Google Scholar]
- Martinez-Delclos, X. Blátidos (Insecta, Blattodea) del Cretácico Inferior de España. Familias Mesoblattinidae, Blattulidae y Poliphagidae. Bol. Geol. Min. 1993, 104, 516–538. [Google Scholar]
- Nel, A.; Martínez-Delclòs, X.; Escuillié, F.; Brisac, P. Les Aeshnidae fossiles: État actuel des connaissances (Odonata, Anisoptera). Neues Jahrb. Geol. Palaontol. Abh. 1994, 194, 143–186. [Google Scholar]
- Nel, A.; Delclòs Martínez, X. Nuevos Zygoptera y Anisoptera (Insecta: Odonata) en el Cretácico inferior de España. Estud. Geol. 1993, 49, 351–359. [Google Scholar] [CrossRef]
- Martínez-Delclòs, X.; Nel, A. Los Goinphidae (Insecta, Odonata, Anisoptera) del Cretácico Inferior de España. Rev. Esp. Paleontol. 1994, 9, 176–184. [Google Scholar]
- Peñalver, E.; Martínez, X.D.; Arillo, A. Yacimientos con insectos fósiles en España. Rev. Esp. Paleontol. 1999, 14, 231–246. [Google Scholar]
- Rasnitsyn, A.P.; Ansorge, J. New Early Cretaceous hymenopterous insects (Insecta: Hymenoptera) from Sierra del Montsec (Spain). PalZ 2000, 74, 335–341. [Google Scholar] [CrossRef]
- Szwedo, J.; Ansorge, J. The first Mimarachnidae (Hemiptera: Fulgoromorpha) from lower cretaceous lithographic limestones of the Sierra del Montsec in Spain. Cret. Res. 2015, 52, 390–401. [Google Scholar] [CrossRef]
- Pujalte, V.; Robles, S.; Hernández, J.M. La sedimentación continental del Grupo Campóo (Malm-Cretácico basal de Cantabria, Burgos y Palencia): Testimonio de un reajuste hidrográfico al inicio de una fase rift. Cuad. Geol. Iber. 1996, 21, 227–251. [Google Scholar]
- Espina, R.J. La estructura y Evolución Tectono-Estratigráfica del Borde Occidental de la Cuenca Vasco-Cantábrica (Cordillera Cantábrica, NO de España). Ph.D. Thesis, Oviedo University, Oviedo, Spain, 1997. [Google Scholar]
- Pujalte, V.; Hernández, J.M.; Robles, S.; Alonso, A. Geología de España; Vera, J.A., Ed.; SGE-IGME: Madrid, Spain, 2004; pp. 339–341. [Google Scholar]
- Hernández, J.M.; Pujalte, V.; Robles, S. Tipología y significado de los fragmentos de calcretas laminares acumulados en la base de la Formación Aguilar (Titoniense terminal–Berriasiense inferior, Grupo Campóo, Palencia, Burgos y Cantabria). Geogaceta 2015, 58, 15–18. [Google Scholar]
- Hernández, J.M.; Pujalte, V.; Robles, S. Los rizolitos de la Fm. Aguilar (Kimmeridgiense-Berriasiense, Palencia, Burgos y Cantabria): Caracterización, génesis y significado. Geogaceta 1997, 22, 93–96. [Google Scholar]
- Bechly, G.; Nel, A.; Martínez-Delclòs, X.; Jarzembowski, E.A.; Coram, R.; Martill, D.; Fleck, G.; Escuillié, F.; Wisshak, M.M.; Maisch, M. A revision and phylogenetic study of Mesozoic Aeshnoptera, with description of several new families, genera and species (Insecta: Odonata: Anisoptera). N. Jb. Geol. Paläont. 2001, 4, 1–219. [Google Scholar]
- Nel, A.; Bechly, G.; Jarzembowski, E.A.; Martínez-Delclòs, X. A revision of the fossil petalurid dragonflies (Insecta: Odonata: Anisoptera: Petalurida). Paleont. Lomb. 1998, 10, 1–68. [Google Scholar]
- Hagen, H.A. Die fossilen Libellen Europas. Stett. Ent. Ztg. 1848, 9, 6–13. [Google Scholar]
- Fleck, G.; Nel, A. Revision of the Mesozoic family Aeschnidiidae (Odonata: Anisoptera). Zoologica 2003, 153, 1–180. [Google Scholar]
- Suarez-Tovar, C.M.; Sarmiento, C.E. Beyond the wing planform: Morphological differentiation between migratory and nonmigratory dragonfly species. J. Evol. Biol. 2016, 29, 690–703. [Google Scholar] [CrossRef] [Green Version]
- Zheng, D.; Jarzembowski, E.A.; Chang, S.C.; Wang, B.; Zhang, H. New cymatophlebiid dragonflies from the Lower Cretaceous of China and England (Odonata: Anisoptera: Cymatophlebiinae, Valdaeshninae). Cret. Res. 2018, 90, 311–317. [Google Scholar] [CrossRef]
- Pritchard, G. Insects in thermal springs. Mem. Ent. Soc. 1991, 123, 89–106. [Google Scholar] [CrossRef]
- Vakhrameev, V.A. Jurassic and Cretaceous Floras and Climates of the Earth; Cambridge University Press: Cambridge, UK, 1991; p. 340. [Google Scholar]
- Campos-Soto, S.; Benito, M.I.; Cobos, A.; Caus, E.; Quijada, I.E.; Suarez-Gonzalez, P.; Mayoral, J.R.; Royo-Torres, R.; Alcalá, L. Revisiting the age and palaeoenvironments of the Upper Jurassic–Lower Cretaceous? Dinosaur-bearing sedimentary record of eastern Spain: Implications for Iberian palaeogeography. J. Iber. Geol. 2019, 45, 471–510. [Google Scholar] [CrossRef] [Green Version]
- MacLeod, S.E.; Hills, L.V. Late Jurassic to Early Cretaceous (Tithonian to pre-Albian) plant macrofossils, northern Bowser Basin, British Columbia, Canada. Rev. Palaeobot. Palynol. 1991, 70, 9–45. [Google Scholar] [CrossRef]
- van Konijnenburg-van Cittert, J.H.; Van der Burgh, J. Review of the Kimmeridgian flora of Sutherland, Scotland, with reference to the ecology and in situ pollen and spores. Proc. Geol. Assoc. 1996, 107, 97–105. [Google Scholar] [CrossRef]
- Van Konijnenburg-Van Cittert, J.H. Ecology of some late Triassic to early Cretaceous ferns in Eurasia. Rev. Palaeobot. Palynol. 2002, 119, 113–124. [Google Scholar] [CrossRef]
- Blanco-Moreno, C.; Buscalioni, Á.D. Quantitative and qualitative frond analysis of the fern Cladophlebis: Distinguishing morphotypes from the Barremian locality of Las Hoyas (southwestern Iberia Basin, Cuenca, Spain). Rev. Palaeobot. Palynol. 2021, 292, 104476. [Google Scholar] [CrossRef]
- Van Konijnenburg-van Cittert, J.H.; Pott, C.; Schmeißner, S.; Dütsch, G.; Kustatscher, E. The Rhaetian flora of Wüstenwelsberg, Bavaria, Germany: Description of selected gymnosperms (Ginkgoales, Cycadales, Coniferales) together with an ecological assessment of the locally prevailing vegetation. Rev. Palaeobot. Palynol. 2021, 288, 104398. [Google Scholar] [CrossRef]
- Pott, C.; Krings, M.; Kerp, H. The Carnian (Late Triassic) flora from Lunz in Lower Austria: Paleoecological considerations. Palaeoworld 2008, 17, 172–182. [Google Scholar] [CrossRef]
- McLoughlin, S.; Carpenter, R.J.; Pott, C. Ptilophyllum muelleri (Ettingsh.) comb. nov. from the Oligocene of Australia: Last of the Bennettitales? Int. J. Plant Sci. 2011, 172, 574–585. [Google Scholar] [CrossRef] [Green Version]
- Butzmann, R.; Fischer, T.; Röper, M.; Rothgaenger, M. Neu in Angriff genommen: Die Bearbeitung der Flora der »Fossillagerstätte Brunn. Freunde der Bayerischen Staatssammlung für Paläontologie und Historische Geologie München. Jahresber. Mitt. 2015, 43, 52–64. [Google Scholar]
- Barthel, K.W.; Swinburne, N.H.M.; Conway Morris, S. Solnhofen, a Study in Mesozoic Palaeontology; Cambridge University Press: Cambridge, UK, 1994; p. 236. [Google Scholar]
- Frickhinger, K.A. Die Fossilien Von Solnhofen. The Fossils of Solnhofen; Goldschneck-Verlag: Korb, Austria, 1994; p. 336. [Google Scholar]
- Bernier, P.; Barale, G.; Bourseau, J.P.; Buffetaut, E.; Gaillard, C.; Gall, J.C.; Wenze, S. The lithographic limestones of Cerin (southern Jura Mountains, France). A synthetic approach and environmental interpretation. C. R. Palevol. 2014, 13, 383–402. [Google Scholar] [CrossRef]
- Nel, A.; Nel, P.; Krieg-Jacquier, R.; Pouillon, J.M.; Garrouste, R. Exceptionally preserved insect fossils in the Late Jurassic lagoon of Orbagnoux (Rhône Valley, France). PeerJ 2014, 2, e510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peyer, K.; Charbonnier, S.; Allain, R.; Läng, E.; Vacant, R. A new look at the Late Jurassic Canjuers conservation Lagerstätte (Tithonian, Var, France). C. R. Palevol. 2014, 13, 403–420. [Google Scholar] [CrossRef]
- Moreau, J.D.; Baret, L.; Lafaurie, G.; Chateau-Smith, C. Terrestrial plants and marine algae from the Late Jurassic lithographic limestone of the Causse Méjean (Lozère, southern France). BSGF Earth Sci. B. 2016, 187, 121–127. [Google Scholar] [CrossRef]
- Barale, G. La paléoflore jurassique du Jura français. Étude systéma-tique, aspects stratigraphiques et paléoécologiques. Doc. Lab. Geol. Lyon 1981, 81, 1–467. [Google Scholar]
- Roman, J.; Atrops, F.; Arnaud, M.; Barale, G.; Barrat, J.M.; Boullier, A.; de Broin, F.; Gill, G.A.; Michard, J.G.; Taquet, P.; et al. Le gisement Tithonien inférieur des calcaires lithographiques de Canjuers (Var, France): État actuel des connaissances. Geobios Mem. Spec. 1994, 16, 126–135. [Google Scholar]
- Pott, C.; Guhl, M.; Lehmann, J. The Early Cretaceous flora from the Wealden facies at Duingen, Germany. Rev. Palaeobot. Palynol. 2014, 201, 75–105. [Google Scholar] [CrossRef]
- Watson, J.; Alvin, K.L. An English Wealden floral list, with comments on possible environmental indicators. Cret. Res. 1996, 17, 5–26. [Google Scholar] [CrossRef]
| Location | Formation/Basin | Age | Geological Setting | Fossil Remains | References |
|---|---|---|---|---|---|
| Spain (Asturias) | Vega Formation (Asturian Basin) | Late Jurassic (early Kimmeridgian) | Oncoid-rich travertines associated with springs and ephemeral lakes near synsedimentary faults (lacustrine environment) | Roots, wood remains, charophytes, and ostracods | García-Ramos et al. (2010), Arenas et al. (2015), Lozano et al. (2016) |
| Spain (Palencia and Burgos provinces, Castilla y León) | Aguilar Formation (Aguilar Basin) | Jurassic-Cretaceous boundary (Tithonian-Berriasian) | Travertine facies that have been associated with hot springs probably linked to the Villela fault (fluvial/lacustrine environment) | Plant macro-remains (pteridophytes and Bennettitales), and insects (Odonata) | Ramírez del Pozo (1972), Schaaf, (1986), Schudack (1987), Hernández et al. (1998, 1999), Hernández (2000), Diéguez et al. (2009), This work |
| Argentina (Patagonia, Santa Cruz province) | Macizo del Deseado | Middle to Late Jurassic | Subaerial and sublacustrine hot spring environments (lacustrine environment) | Plants, arthropods, microbial, and fungal fossils | Echeveste (2005), Channig et al. (2007), Guido and Campbell (2009), Guido et al. (2010), Guido and Campbell (2011, 2012), García-Massini et al. (2012), Channing et al. (2011), Guido et al. (2019) |
| USA (Utah) | Navajo Sandstone Formation | Early Jurassic | Shallow spring-fed lakes ponded between aeolian dunes (desert oases environment) | Conifer trunks, ostracodes, charophytes, fish, mollusks, possible freshwater sponge, trace fossils, fragments of vascular plants | Parrish and Falcon-Lang (2007) |
| Canada (Nova Scotia) | Scots Bay Formation | Early Jurassic | Silica-rich hydrothermal springs and seeps around the floor of an aerobic lake | Algae, plants (wood remains, oncolites, charophytes, ostracods, gastropods, conchostraceans, fish bones | De Wet and Hubert (1989) |
| USA (Connecticut) | Coe’s Quarry (Hartford Basin) | Early Jurassic | Boiling hot spring setting characterized by micritic and banded travertines, cellular tufa, and abundant spherulites | Algae or bacteria evidence (stromatolitic structures) | Steinen et al. (1987) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, A.A.; Nel, A.; Rodríguez-Barreiro, I.; Sender, L.M.; Wappler, T.; Diez, J.B. Insect and Plant Diversity in Hot-Spring Ecosystems during the Jurassic-Cretaceous Boundary from Spain (Aguilar Fm., Palencia). Biology 2022, 11, 273. https://doi.org/10.3390/biology11020273
Santos AA, Nel A, Rodríguez-Barreiro I, Sender LM, Wappler T, Diez JB. Insect and Plant Diversity in Hot-Spring Ecosystems during the Jurassic-Cretaceous Boundary from Spain (Aguilar Fm., Palencia). Biology. 2022; 11(2):273. https://doi.org/10.3390/biology11020273
Chicago/Turabian StyleSantos, Artai A., André Nel, Iván Rodríguez-Barreiro, Luis M. Sender, Torsten Wappler, and José B. Diez. 2022. "Insect and Plant Diversity in Hot-Spring Ecosystems during the Jurassic-Cretaceous Boundary from Spain (Aguilar Fm., Palencia)" Biology 11, no. 2: 273. https://doi.org/10.3390/biology11020273
APA StyleSantos, A. A., Nel, A., Rodríguez-Barreiro, I., Sender, L. M., Wappler, T., & Diez, J. B. (2022). Insect and Plant Diversity in Hot-Spring Ecosystems during the Jurassic-Cretaceous Boundary from Spain (Aguilar Fm., Palencia). Biology, 11(2), 273. https://doi.org/10.3390/biology11020273








