Highlights
- The tribocorrosion behavior of the WC/DLC coating was systematically investigated.
- The dense and chemically inert DLC barrier prevents solution penetration.
- The high-hardness and self-lubricating DLC enhances the tribological properties.
- The tribocorrosion resistance of duplex coating stems from the functional top layer.
Abstract
To gain deeper insight into the protective mechanism of tungsten carbide/diamond-like carbon (WC/DLC) duplex coatings, this study employed high-velocity oxygen-fuel (HVOF) combined with linear ion source (LIS) technology to deposit the WC/DLC duplex coating on the Ti6Al4V substrate. Their tribocorrosion behaviors were thoroughly investigated. The results show that the dense, uniform, and chemically inert DLC top layer acts as an effective barrier, preventing the corrosive medium from penetrating into the underlying WC cermet layer through pores. Consequently, the duplex coating exhibits a lower Icorr of 3.54 × 10−8 A/cm2, compared with that of the single WC coating (1.08 × 10−6 A/cm2), demonstrating significantly improved corrosion resistance. Moreover, the DLC coating offers excellent tribological performance owing to the high hardness and self-lubricating characteristics. After depositing the DLC top layer on the HVOF-sprayed WC cermet, the COF is reduced to ~0.08, and the wear rate reaches only 5.64 × 10−8 mm3/N·m, indicating notably enhanced tribocorrosion resistance. In short, in such HVOF-PVD/CVD duplex coating systems, the PVD/CVD functional layer can improve the tribocorrosion performance of the HVOF interlayer by leveraging its intrinsic advantages, such as high hardness, low friction, dense and uniform microstructure, and chemical inertness.
1. Introduction
The ocean harbors abundant oil and gas resources, and advanced marine engineering equipment serves as the core guarantee for exploiting such resources [1,2]. However, in harsh marine corrosive environments, the operational stability and service life of metal moving components face severe challenges [3]. Typical examples include seawater hydraulic piston pumps, buoyancy regulation systems, and drilling equipment, which commonly suffer from coupled electrochemical–tribological damage under multi-field conditions.
As a protective layer for metal substrates, surface coatings can extend the service limits while preserving the intrinsic properties of the materials, thereby providing long-term and reliable protection for key components of marine engineering equipment. As mainstream surface modification techniques, high-velocity oxygen-fuel (HVOF) and physical/chemical vapor deposition (PVD/CVD) enable the tailored fabrication of functional protective coatings. HVOF enables rapid deposition of thick, hard cermet coatings. However, its inherent high porosity and the corrosion susceptibility of the binder phase make it challenging to meet the corrosion resistance and lubrication requirements in marine environments [4,5]. In contrast, PVD/CVD techniques, particularly those producing chemically inert amorphous carbon-based coatings, provide coatings with self-lubricity, dense and uniform microstructure, and strong corrosion resistance [6]. Nevertheless, the high-energy ion bombardment during deposition tends to generate significant internal stress and insufficient adhesion, which can lead to sudden coating delamination under high contact-stress conditions [7]. Thus, for marine engineering equipment operating under demanding conditions such as hard abrasive media, intense fluid erosion, and high-frequency movement, existing single-surface technologies struggle to deliver the combined protective properties of high load-bearing capacity, wear resistance, low friction, and strong corrosion resistance required for long service life and high reliability.
In recent years, researchers have proposed a novel duplex coating system combining an HVOF underlayer with a PVD/CVD top layer [8]. It has been suggested that this composite structure can overcome the limitations of individual surface techniques while combining their advantages, resulting in a synergistic effect. For instance, a CrN/Cr3C2-NiCr duplex coating fabricated by Li et al. was shown to synergistically improve both the mechanical strength and wear resistance of the substrate [9]. In a related study, finite element modeling conducted by Zheng et al. on an AlCrN/Cr3C2-NiCr coating revealed that its composite architecture redirects the Mises stress concentration away from the AlCrN layer. This stress redistribution mitigates deformation and delamination caused by high stress, thereby substantially increasing the coating’s load-bearing capacity and service life [10]. Beyond mechanical properties, such designs also improve chemical stability. Zhang et al. confirmed that in a CrN/WC-Co coating system, the CrN overlayer acts as a sealant for the pores in the underlying WC-Co, resulting in significantly enhanced corrosion resistance [11]. Furthermore, the versatility of composite coatings is highlighted by work such as that of Pougoum et al., who utilized combined HVOF-PVD/CVD techniques to deposit CrN(DLC)/Fe3Al and Cr(Nb)N/WC-12Co duplex coatings. These coatings were reported to exhibit exceptional resistance to tribocorrosion [12,13]. In our previous studies [14,15,16], WC/DLC duplex coatings were successfully prepared via the combined HVOF-PVD/CVD approach. It was demonstrated that the HVOF-PVD/CVD synergistic strategy ingeniously achieves dual benefits, offering a new paradigm for designing coatings with high strength-toughness, wear resistance, and corrosion resistance. From the above studies, it is evident that the HVOF-PVD/CVD strategy endows metallic substrates with superior corrosion and tribocorrosion performance compared to single-layer coating technologies. However, the tribocorrosion protection mechanism of such duplex coatings in marine environments still lacks systematic and in-depth exploration. In particular, the protective mechanism of the PVD/CVD top layer over the HVOF underlayer still lacks detailed and intuitive characterization, necessitating a comprehensive elucidation.
Thus, this study systematically investigates the tribocorrosion behaviors of a WC/DLC duplex coating fabricated by HVOF-PVD/CVD. The microstructure and chemical bonding states of the duplex coating were comprehensively characterized using FIB-SEM, TEM, XPS, and Raman spectroscopy to elucidate its tribocorrosion protection mechanism. It should be emphasized that, compared to prior work, this study is the first to provide direct visualization of the barrier effect of the DLC top layer against the corrosive medium by employing FIB technology. This work is expected to provide not only comprehensive and universal theoretical guidance for understanding the corrosion and tribocorrosion behaviors of HVOF-PVD/CVD duplex coatings, but also important references for designing tribocorrosion-resistant coatings for critical metal components in marine engineering equipment.
2. Experimental Procedure
2.1. Coating Preparation
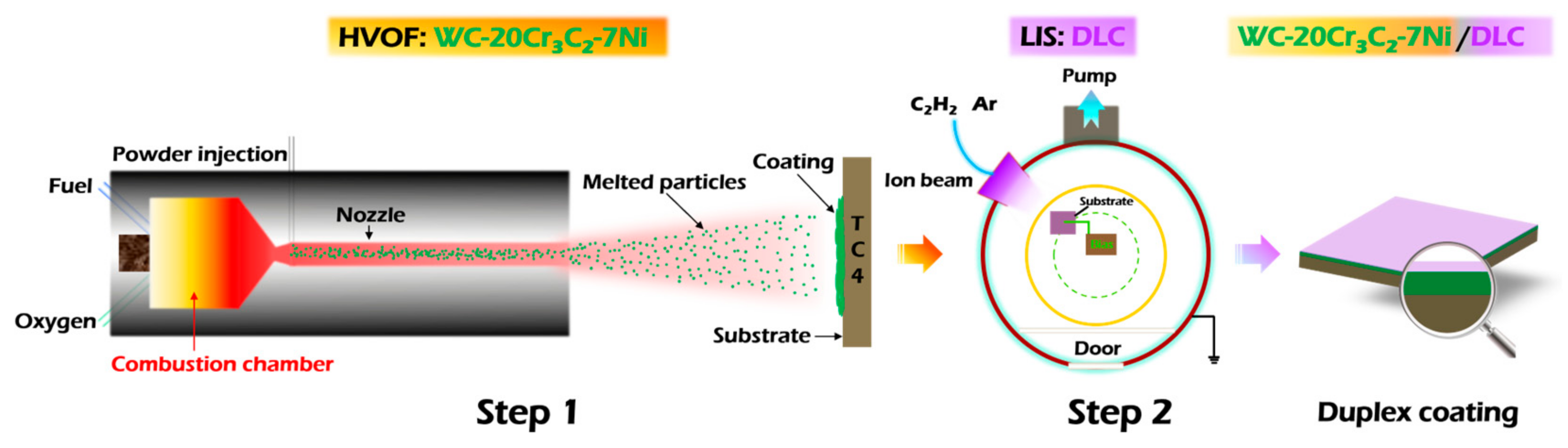
A commercially available Ti6Al4V alloy (TC4, Baoti Group, Baoji, China) was used as the substrate material. It is a typical α-β dual-phase titanium alloy, with a hardness exceeding 30 HRC after stress-relief annealing. The chemical composition (wt.%) of the TC4 substrate is as follows: Al: 6.25, V: 4.31, Fe: 0.17, O: 0.12, and balance Ti. Prior to the deposition of the HVOF underlayer, the substrate was sequentially ground using 400, 1000, 3000, and 5000-grit SiC abrasive papers, followed by polishing with diamond paste. The resulting surface roughness (Ra) was approximately 8.1 nm. Finally, the substrate was ultrasonically cleaned in ethanol for 10 min to remove any surface oils and contaminants. The preparation scheme of the Duplex coating is shown in Figure 1.
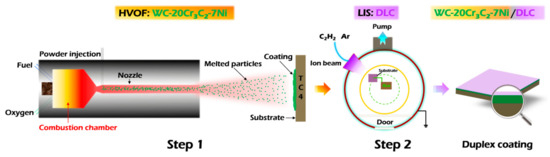
Figure 1.
Preparation scheme of Duplex coating on the TC4 substrate.
The first step involves preparing the HVOF coating. The commercially available WC-20Cr3C2-7Ni (wt.%, WC: 73, Cr3C2: 20, Ni: 7) powder was used as the feed-stock material, which was sprayed on the sandblasted TC4 substrate by using a JP8000 HVOF system (Praxair, Eau Claire, WI, USA). The spray area on the TC4 substrate measured 200 mm × 200 mm. After spraying, the coated sample was cut into specified dimensions of Ø17 mm × 3 mm. An image of the as-prepared sample is presented in Figure 2. The spray parameters are presented in Table 1.
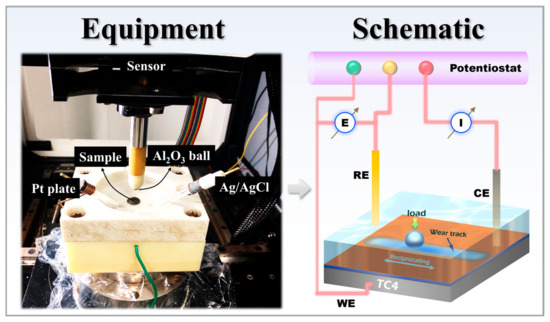
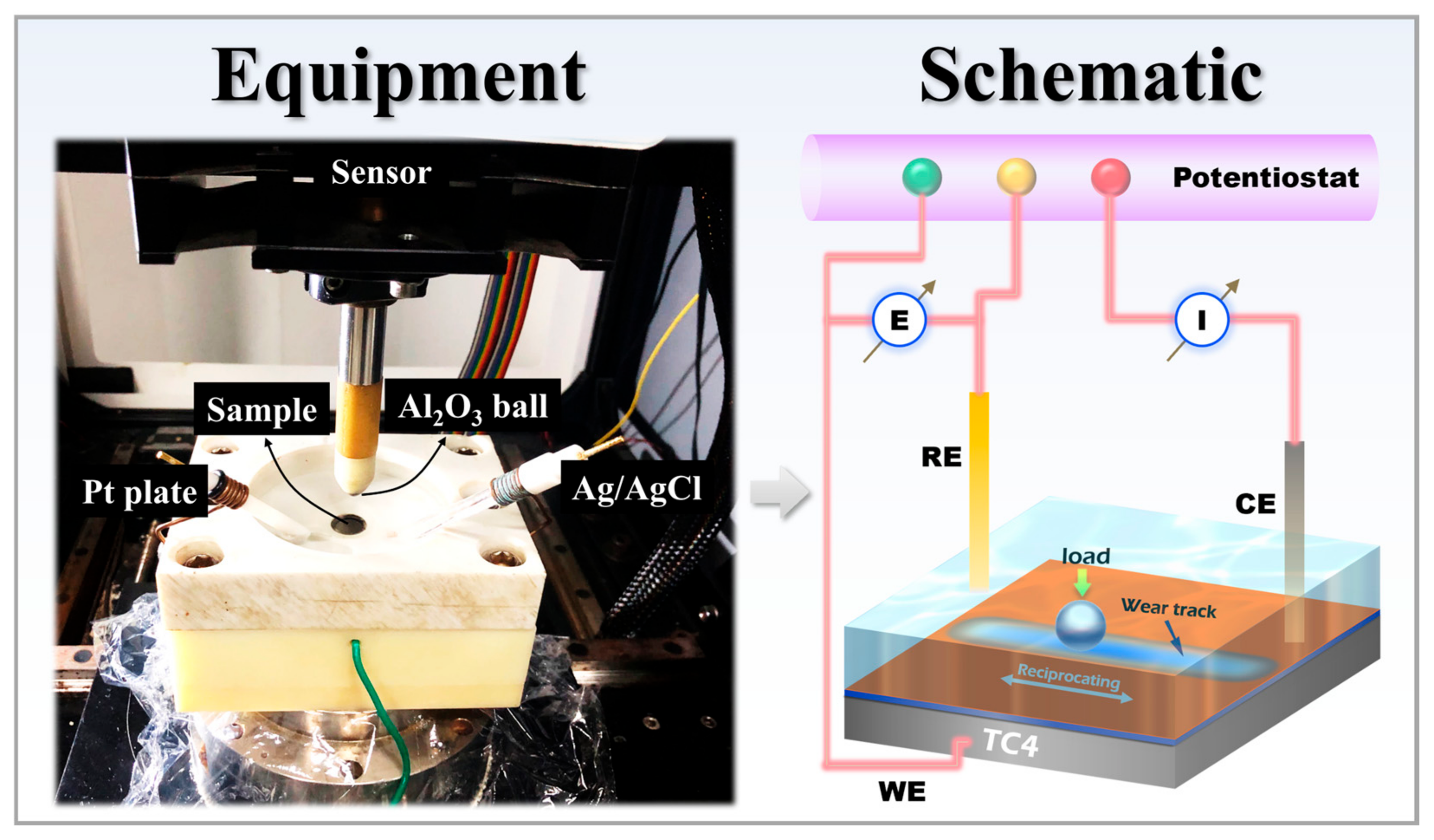
Figure 2.
Equipment and schematic diagram of the tribocorrosion experiment device.

Table 1.
The deposition conditions of the HVOF coating.
The second step involves covering the HVOF coating with the DLC film. Prior to DLC film deposition, the HVOF-coated surface was sequentially ground and polished using diamond abrasive papers (400, 800, 1200, and 2000 grit) followed by diamond lapping films (600, 1200, and 2000 grit). This process resulted in a final surface roughness (Ra) of approximately 10.4 nm. The sample was then ultrasonically cleaned in acetone and ethanol for 10 min each. After cleaning, it was dried with a lint-free cloth and a stream of clean air, before being placed into the vapor deposition system for DLC top-layer fabrication. The DLC film was deposited using a P600 chemical vapor deposition (CVD) system (J&L), with high-purity acetylene (C2H2, 99.99%) employed as the carbon precursor. During deposition, a linear ion source (LIS) was utilized, operating at parameters of 0.2 A and 1200 V. The complete set of deposition parameters is detailed in Table 2.

Table 2.
The process conditions of the DLC film.
2.2. Electrochemical Corrosion and Tribocorrosion Tests
Electrochemical corrosion performance was evaluated in accordance with the ASTM G59-97 standard. Tests were conducted using a Gamry Reference 600+ electrochemical workstation at room temperature (Gamry Instruments, Warminster, PA, USA), with a 3.5 wt% NaCl solution serving as the electrolyte. A standard three-electrode cell configuration was employed: the TC4 substrate, HVOF coating, and Duplex coating served as the working electrodes, a platinum plate as the counter electrode, and a KCl-saturated Ag/AgCl electrode as the reference electrode. Prior to testing, samples were immersed in the electrolyte for 2 h to allow the open circuit potential (OCP) to stabilize, during which the OCP was continuously monitored. Electrochemical impedance spectroscopy (EIS) measurements were then performed over a frequency range of 105 to 10−2 Hz, applying a sinusoidal amplitude of 10 mV. Subsequently, potentiodynamic polarization (PDP) scans were carried out, scanning from −0.5 V to +1.0 V relative to the OCP at a constant sweep rate of 0.5 mV/s. Electrochemical corrosion tests were performed in triplicate for each sample group to ensure data reliability. Figure 2 shows the equipment and schematic diagram of the tribocorrosion test apparatus.
The tribocorrosion test setup primarily consisted of an MFT-5000 reciprocating friction and wear tester (Rtec) integrated with a ModuLab XM ECS electrochemical workstation (Modulab, Oak Ridge, TN, USA). As shown, the sample was secured in a specifically designed holder. This holder featured a reservoir to contain the corrosive solution. The sample had a total diameter of 17 mm, with an exposed area of 15 mm in diameter within the holder. Reciprocating sliding was applied to the central region of this exposed surface. A loading rod, positioned above the holder, was equipped with a grinding ball at its lower end and connected to a force sensor at its upper end. The electrochemical setup employed a standard three-electrode configuration: the sample itself served as the working electrode (WE), a platinum plate as the counter electrode (CE), and an Ag/AgCl electrode as the reference electrode (RE). All electrodes were connected to the testing circuit via interfaces incorporated into the sample holder. All tests in this work were conducted in a 3.5 wt.% NaCl electrolyte under stabilized OCP conditions. Prior to tribocorrosion testing, the samples were immersed in the solution for 3600 s to allow the OCP to reach a steady state. Subsequently, continuous sliding was initiated and maintained for another 3600 s to monitor the evolution of the OCP. The sliding was performed under the following specific parameters: a normal load of 2 N was applied through an Al2O3 ball (6 mm in diameter) serving as the counterpart, with a reciprocating stroke length of 4 mm and a sliding speed of 20 mm/s. It is noteworthy that a normal load of 2 N was selected based on findings from prior research, which reported varying degrees of delamination in the DLC top layer when sliding under loads exceeding 5 N [14]. This choice was made to prevent premature failure of the DLC coating, thereby enabling a focused investigation into its protective mechanism against the tribocorrosion of the WC-based cermet underlayer. Following the cessation of sliding, the OCP of the samples continued to be monitored for an additional 3600 s. All tribocorrosion tests were performed in triplicate to ensure the reliability of the results.
2.3. Characterization Method
The microstructural characteristics of the coatings were analyzed by TF20 transmission electron microscopy (TEM, FEI, Hillsboro, OR, USA). TEM specimens were prepared using a focused ion beam (FIB, Carl Zeiss, Jena, Germany) system, a protective Pt layer was first deposited on the area of interest, followed by cutting, extraction, and final thinning to electron transparency using a Ga+ ion beam at 30 kV. TEM observations were conducted at an accelerating voltage of 200 kV. The chemical composition of the coatings was examined by X-ray photoelectron spectroscopy (XPS, Kratos AXIS SUPRA, Manchester, UK) using a monochromatic Al Kα X-ray source (1486.6 eV). Survey and high-resolution spectra were acquired with pass energies of 160 eV and 40 eV, respectively. Binding energies were calibrated to the C 1s peak at 284.8 eV, and chemical state analysis was performed using Shirley background subtraction and peak fitting with Gaussian-Lorentzian line shapes. The surface and cross-sectional morphologies of the coatings and substrates were examined using field-emission scanning electron microscopy (SEM, FEI Quanta FEG 250, Hillsboro, OR, USA) at an accelerating voltage of 10 kV with a secondary electron detector. The phase composition of the coatings was determined by a D8 Advance DaVinci X-ray diffraction (XRD, Bruker, Ettlingen, Germany) performed with Cu-Kα radiation (λ = 1.5418 Å, 40 kV, 15 mA), scanning from 20° to 80° (2θ) with a step size of 0.02° and a scan speed of 2°/min. The structure of the DLC films was characterized using a Renishaw inVia Reflex micro-confocal Raman spectrometer (Bruker, Ettlingen, Germany). A 532 nm laser source was used for excitation at 1% of its maximum power to avoid thermal damage to the samples. The spectra were collected in the range of 1000–1800 cm−1, with each spectrum accumulated over 3 scans. Mechanical properties were assessed via microhardness testing with an HV1000IS image analysis system (Shanghai Jvjing, Shanghai, China); for each sample, hardness values were obtained from an average of ten individual measurements to ensure statistical reliability. Quantitative analysis of the wear track profiles (including width, depth, and cross-sectional area) was conducted using an UP-Lambda 3D optical profiler (Rtec Instrument, San Jose, CA, USA). To minimize random errors, three distinct regions near the center of each track were measured, with the average value reported. The wear rate (W) was evaluated according to the following formula [17]: W = (S × L)/(F × D), where S and L are the cross-sectional area and length of the wear track, respectively, F is the applied normal load, and D is the total sliding distance. It is noted that all measurements were performed in the central region of the samples to avoid potential edge effects that could influence the test results.
3. Results and Discussion
3.1. Microstructure and Phase Composition
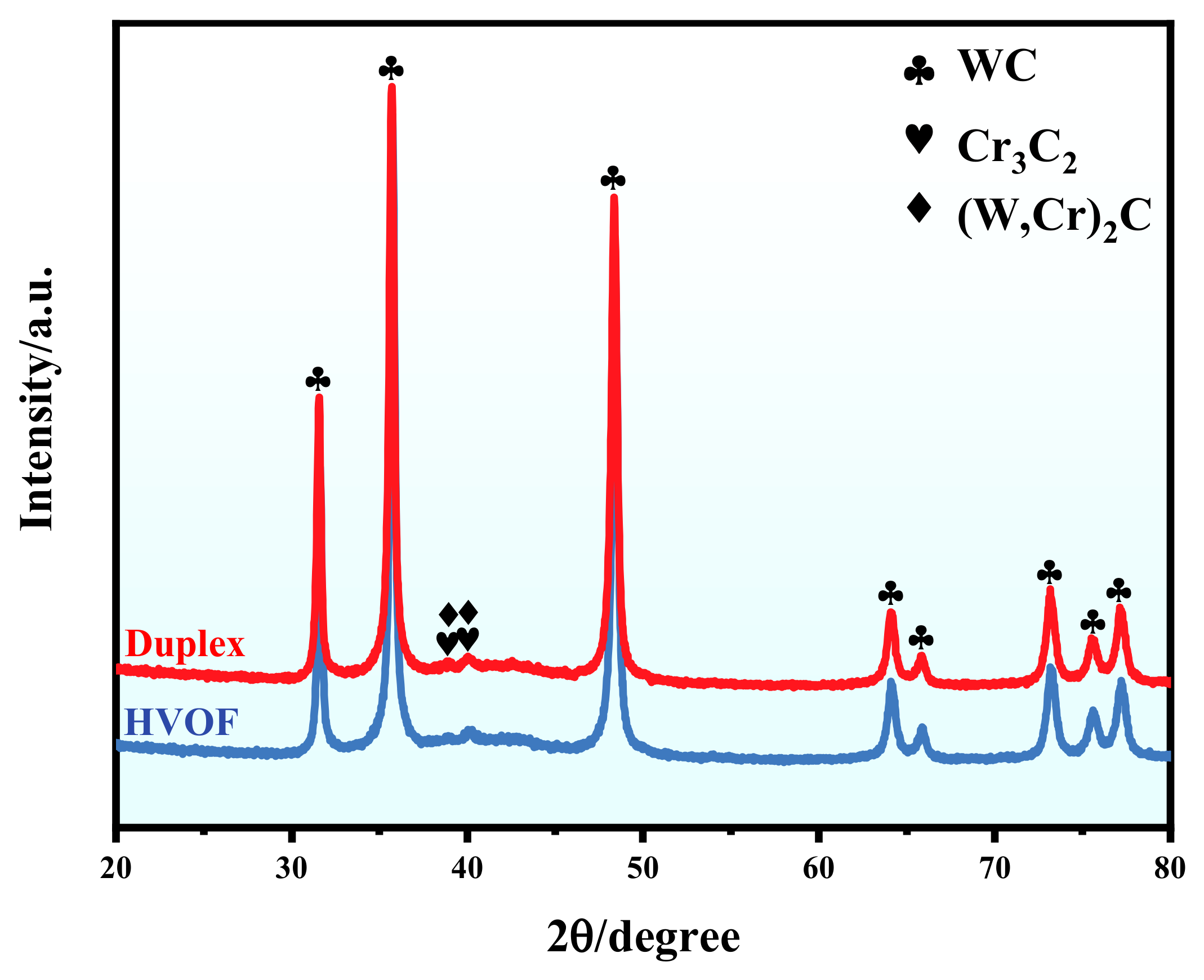
The XRD patterns of the HVOF and Duplex coatings are shown in Figure 3. The HVOF coating is mostly constituted of the WC phase and the minor Cr3C2 and (W, Cr)2C phases. The formation of the hard brittle (W, Cr)2C phase was caused by the high-temperature decarburization of the powder during the HVOF spraying process. It should be noted that the diffraction peaks of the Duplex coating are almost identical to that of the HVOF coating. It means that X-rays penetrate the DLC film and into the underlying HVOF coating. In addition, no crystalline carbon phase was observed in the XRD pattern, which indicates that the carbon contained in the DLC film is amorphous.
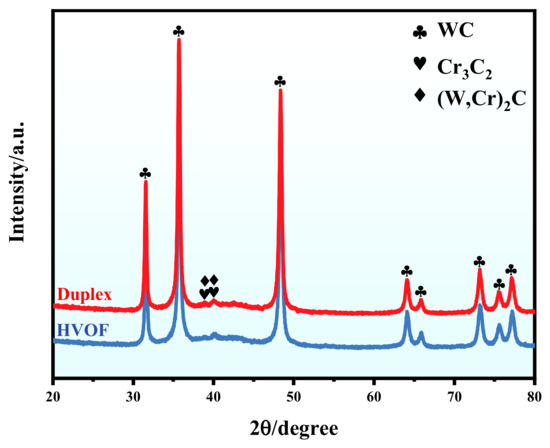
Figure 3.
XRD patterns of the HVOF and Duplex coatings.
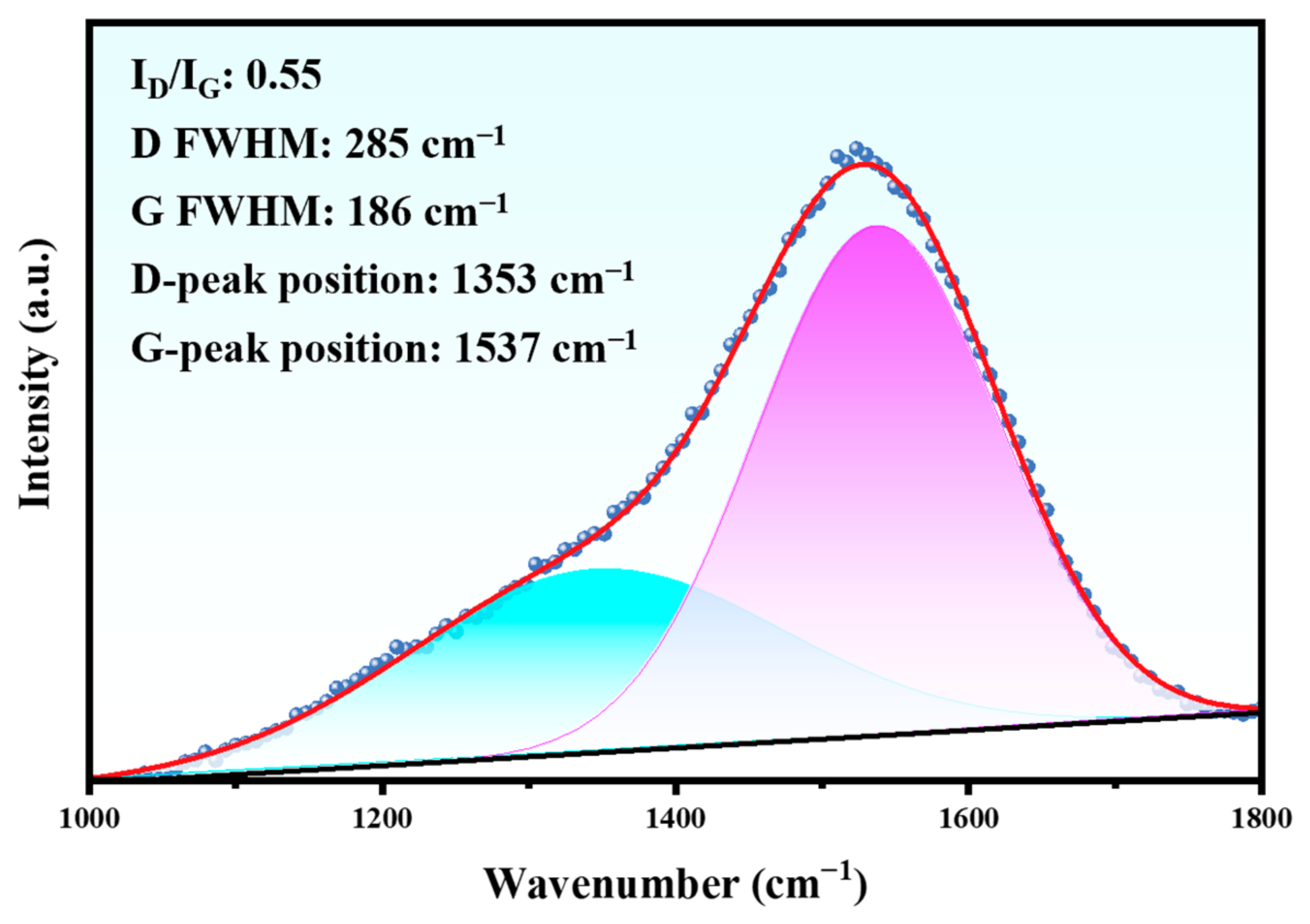
To further comprehend the structural characteristics of the DLC film, the chemical bonding of carbon was analyzed by Raman spectroscopy (Figure 4). It can be observed that an asymmetric broad peak exists near the 1500 cm−1 wave number, which is a typical Raman peak of a DLC film. The broad peak was decomposed into two peaks by Gaussian function, which were D peak and G peak, respectively. The G peak near 1540 cm−1 was produced by the central vibration mode of the E2g region of crystalline graphite optics, which confirms the existence of the sp2-C hybrid bond. The D peak near 1350 cm−1 was derived from the T2g symmetric vibration mode, which was attributed to the disorder of the sp2 hybrid bond angle in the carbon material [18]. Generally, the D band corresponds to the disorder, while the G band is related to the properties of graphite [19]. The DLC film was composed of sp3 carbon atoms with a diamond like structure and sp2 carbon atoms with a graphitic structure [20]. The value of ID/IG indirectly indicates the sp3 hybrid bond content in the DLC film, and a small ID/IG ratio implies that the sp3 hybrid bond content in the film is high.
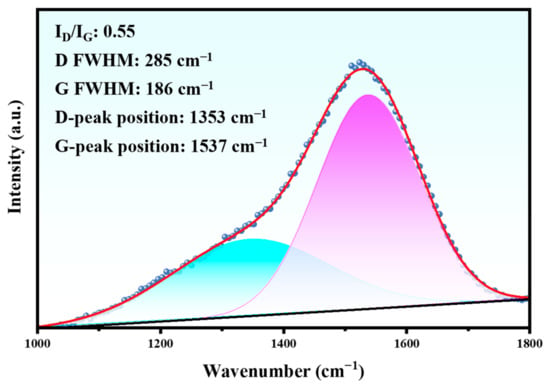
Figure 4.
Raman spectrum of the initial Duplex coating.
It can be seen from Figure 4 that the intensity ratio (ID/IG) of the DLC film is 0.55. This value can reflect the physical and chemical properties of the DLC film on the Duplex coating to a certain extent. The cross-sectional characteristics of the Duplex coating are shown in Figure 5.

Figure 5.
(a,b) SEM image and (c,d) FIB-STEM characterization (selected area) of the Duplex coating (Pt denotes the platinum protective layer).
It is observed from Figure 5a that the HVOF coating and the TC4 substrate are in close contact without obvious gaps. High-velocity particles impact the soft TC4 substrate and produce plastic deformation during the thermal spraying process. Therefore, the HVOF coating exhibits good contiguous mechanical contact with the substrate. From Figure 5c (partial enlargement and thinning of Figure 5b), it can be clearly seen that the thickness of the DLC film is about 2.25 μm, accompanied by a dense and uniform structure. The EDS mappings of C, W, Cr, and Ni are also displayed in Figure 5c. The C gathered on the top represents the DLC film, and the evenly dispersed C at the bottom comes from WC-20Cr3C2-7Ni. It should be pointed out that the missing area of the W element corresponds to the enriched area of the Cr elements. Furthermore, in conjunction with the SAED results (Figure 5d), it can be demonstrated that the HVOF coating consists mainly of tungsten carbide and chromium carbide phases. Unlike this, the DLC film exhibits a typical amorphous structure, which aligns with the XRD results.
3.2. Surface Hardness Test
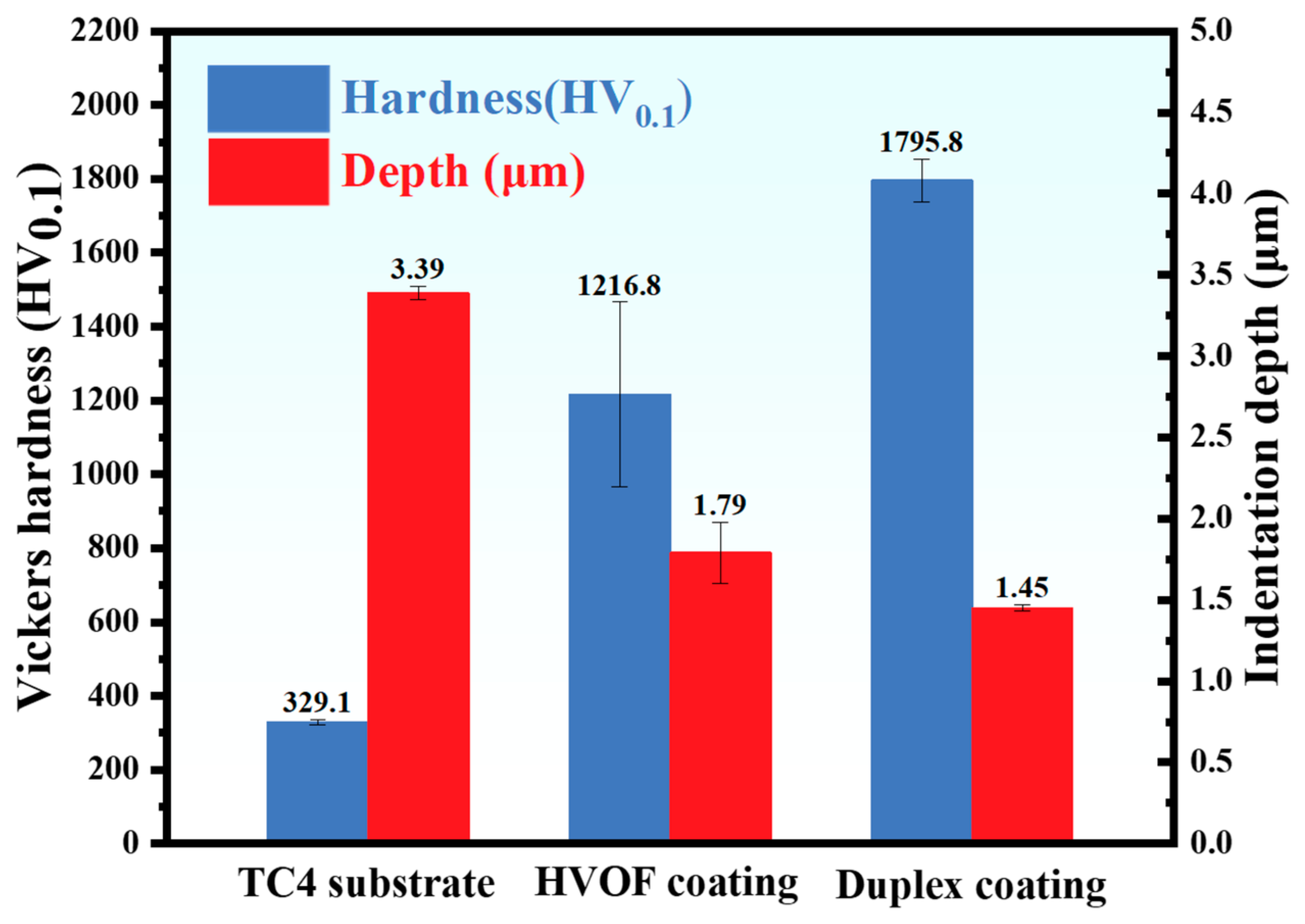
The Vickers hardness of the TC4 substrate, HVOF coating, and Duplex coating was 329.1 ± 7.8 HV0.1, 1216.8 ± 249.7 HV0.1, and 1795.8 ± 57.9 HV0.1, respectively, as shown in Figure 6. In addition, the indentation depths of TC4 substrate, HVOF coating, and Duplex coating were 3.39 ± 0.04 μm, 1.79 ± 0.19 μm, and 1.45 ± 0.02 μm, respectively. It can be observed that the sequence of indentation depth in the specimens is negatively correlated with hardness. As shown in Figure 5c, the DLC coating possesses a thickness of approximately 2 µm, which is sufficient to prevent it from being penetrated during the mechanical test. Notably, the HVOF coating exhibits an intermediate hardness value between the hard DLC top layer and the soft TC4 substrate. This graded mechanical response is believed to facilitate a smooth transition in stress distribution and provide effective support to the overlying DLC film, thereby enhancing the system’s integrity under load [21]. Details regarding the mechanical properties of the WC/DLC duplex coating, such as adhesion strength and residual stress, can be found in our previous publication [14].
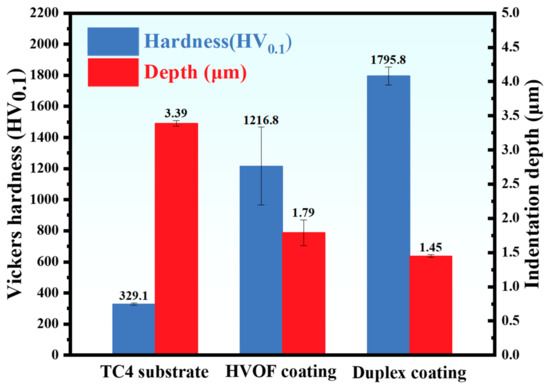
Figure 6.
Vickers hardness and indentation depth of TC4 substrate, HVOF coating, and Duplex coating.
3.3. Electrochemical Corrosion Performance
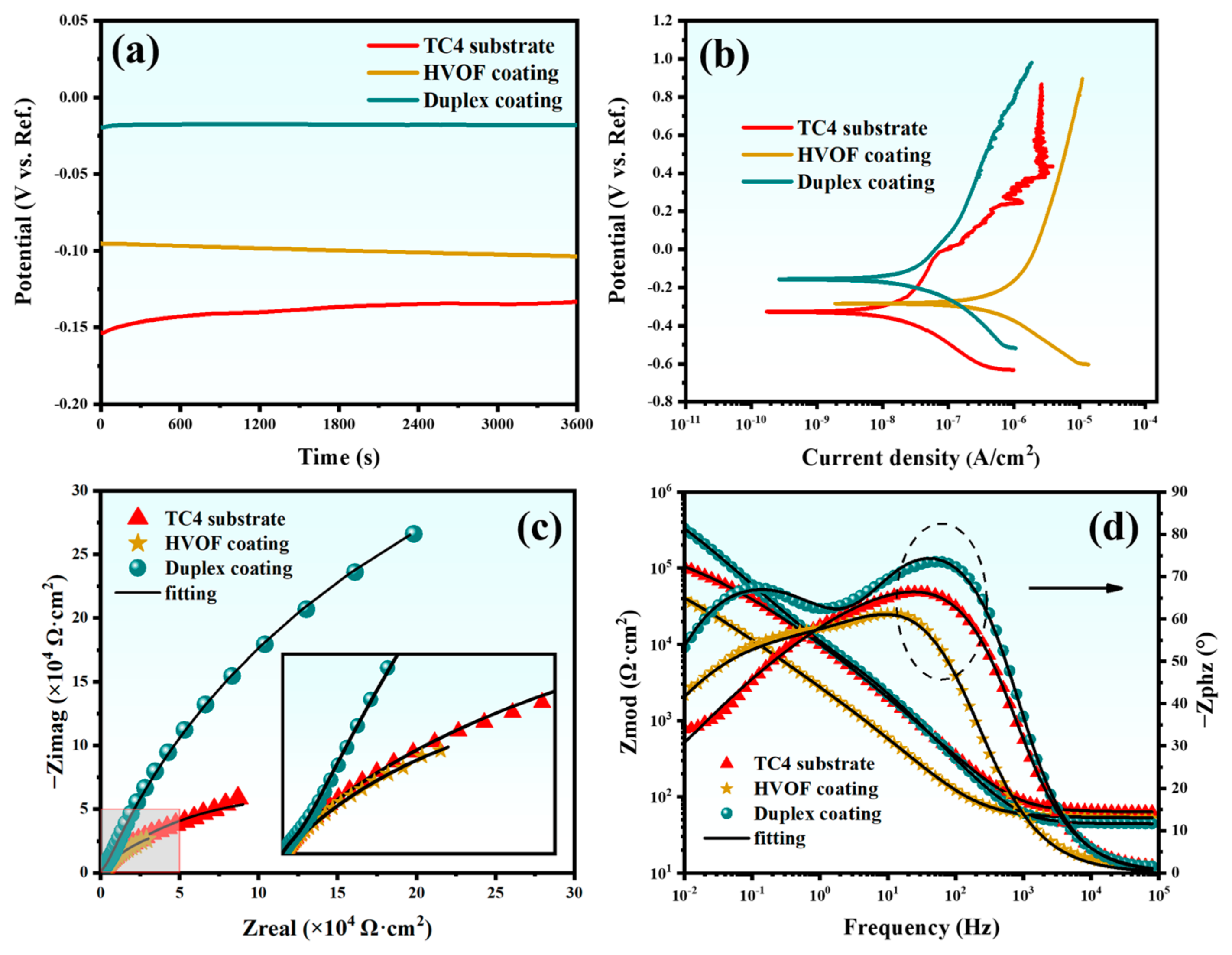
The OCP, PDP, Nyquist plot, and Bode plot of the TC4 substrate, HVOF, and Duplex coatings are shown in Figure 7, respectively. OCP is an important parameter to evaluate the corrosion tendency of the coating. Generally, a high OCP symbolizes a low corrosion tendency [22]. It can be inferred from Figure 7a that the OCP of the TC4 substrate is lower than that of the HVOF coating and the Duplex coating. It indicates that the corrosion tendency of the TC4 substrate is reduced after the deposition of the HVOF and Duplex coatings. In addition, the OCP of the Duplex coating is enormously higher than that of the HVOF coating, which means that the coverage of the DLC film significantly improves the corrosion resistance of the HVOF coating. Notably, the OCP of the TC4 substrate exhibited a continuous increase with immersion time, manifesting a clear passivation behavior. In contrast, the OCP of the HVOF coating showed a slight decrease over time, suggesting a tendency for active corrosion. In stark contrast to both, the OCP of the duplex coating remained nearly unchanged throughout the immersion period, demonstrating exceptional stability. Figure 7b shows the PDP curve of the sample, with the Tafel fitting results presented in Table 3.
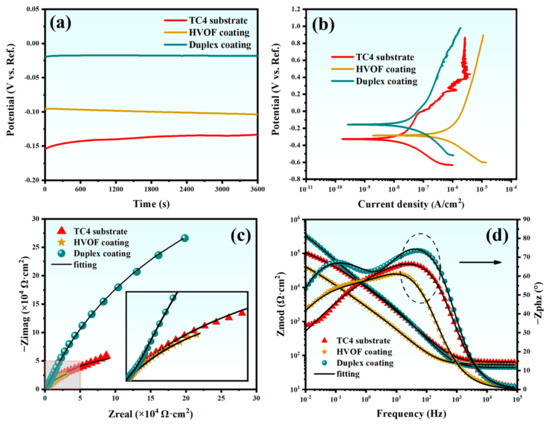
Figure 7.
(a) OCP-time response, (b) PDP curves, (c) Nyquist plots, and (d) Bode plots of the TC4 substrate, HVOF, and Duplex coatings.

Table 3.
Tafel fitting results obtained from the PDP curve.
The Duplex coating showed the highest corrosion potential (Ecorr, −0.156 V) and the lowest corrosion current density (Icorr, 3.54 × 10−8 A/cm2) compared to the other samples. This indicates that the Duplex coating exhibits excellent corrosion resistance.
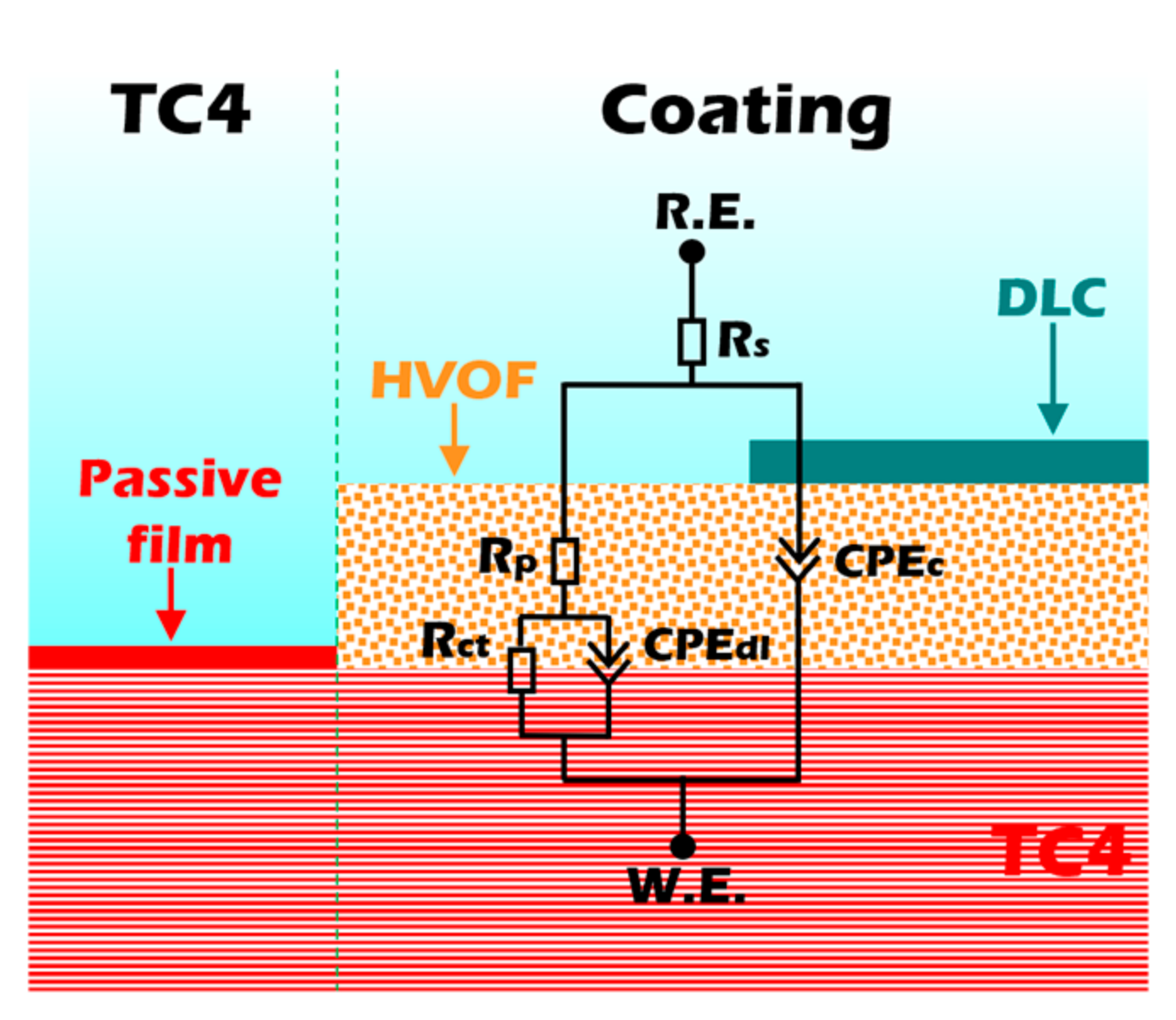
The EIS was further employed to evaluate the corrosion resistance of the samples. In EIS analysis, a larger capacitive arc radius in the Nyquist plot typically corresponds to superior corrosion resistance. As presented in Figure 7c, the Duplex coating exhibits a significantly larger capacitive arc radius compared to both the TC4 substrate and the standalone HVOF coating. This result clearly demonstrates that the Duplex coating possesses the highest corrosion resistance among the three materials tested. The Bode impedance plot (Figure 7d) indicates that the impedance modulus (|Z|0.01Hz) of the Duplex coating is much higher than that of the other samples, showing an exceptional corrosion resistance. In addition, the Bode phase plot (Figure 7d) testifies that the Duplex coating always maintains a higher value compared to the TC4 substrate and HVOF coating, indicating that it has an outstanding barrier that prevents the penetration of corrosive media. An equivalent circuit (Figure 8) was used to fit the EIS data of the TC4 substrate, HVOF coating, and Duplex coating.
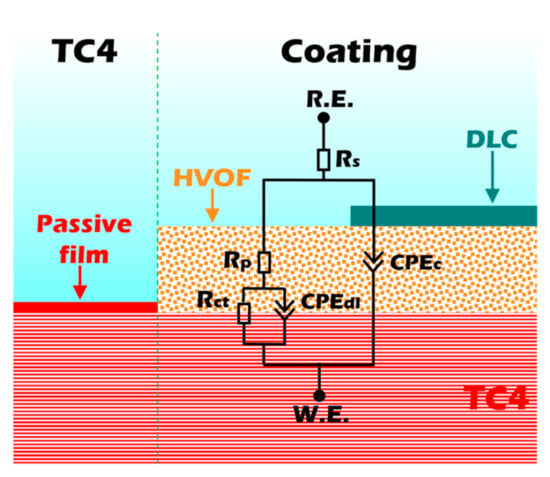
Figure 8.
Equivalent circuits of the TC4 substrate, HVOF, and Duplex coatings.
The electronic components in the equivalent circuit are composed of Rs (solution resistance), Rp (pore resistance in the coating/passivation film), Rct (charge transfer resistance), CPEc, and CPEdl (constant phase element for coating and electric double layer). The CPE was used to represent the rough and porous surface of the sample. CPE is usually used in electrochemical models instead of capacitors to compensate for heterogeneity in the system [23]. CPE contains two parameters, CPE-T and CPE-P, respectively. CPE-P can be considered as the degree to which CPE-T is close to pure capacitance, and the value range of CPE-P is 0–1. Therefore, if the value of CPE-P is 0 or 1, then CPE-T represents an ideal resistor or capacitor, respectively. When CPE-P is between 0 and 1, CPE-T appears as a pseudo-capacitance. Therefore, the larger the CPE-P, the closer the CPE-T is to the ideal capacitor, which means that the sample is denser [24]. The fitted curves obtained from this equivalent circuit model are presented in Figure 7c,d. As shown, the modeled curves show excellent agreement with the experimental data points, which is further supported by a low Chi-Squared value (on the order of 10−4). This indicates a high degree of consistency between the model and the experimental results. The detailed fitting results are listed in Table 4.

Table 4.
The fitting results of the TC4 substrate, HVOF coating, and Duplex coating according to the EIS plots.
It can be seen from Table 4 that the CPEc-P of the Duplex coating (0.91) is higher than that of the HVOF coating (0.80), which indicates that the Duplex coating is more compact. Moreover, compared with the HVOF coating, the Duplex coating has lower CPEc-T (9.37 × 10−6 F·cm−2·sn−1) and higher Rp (9.56 × 103 Ω·cm2). It is further confirmed that the Duplex coating exhibits a better corrosion shielding effect. Rct is an important parameter to evaluate the electrochemical performance, which directly represents the electrochemical dissolution rate of the electrode (sample) [25]. Among all samples, the Duplex coating displayed the highest Rct (1.01 × 106 Ω·cm2) and the lowest CPEdl-T (1.53 × 10−5 F·cm−2·sn−1). Therefore, the Duplex coating acts as a shielding material with prevailing resistances and inferior capacitances. According to electrochemical tests, it is concluded that the Duplex coating has the most outstanding corrosion resistance compared to the TC4 substrate and HVOF coating.
3.4. Tribocorrosion Resistance
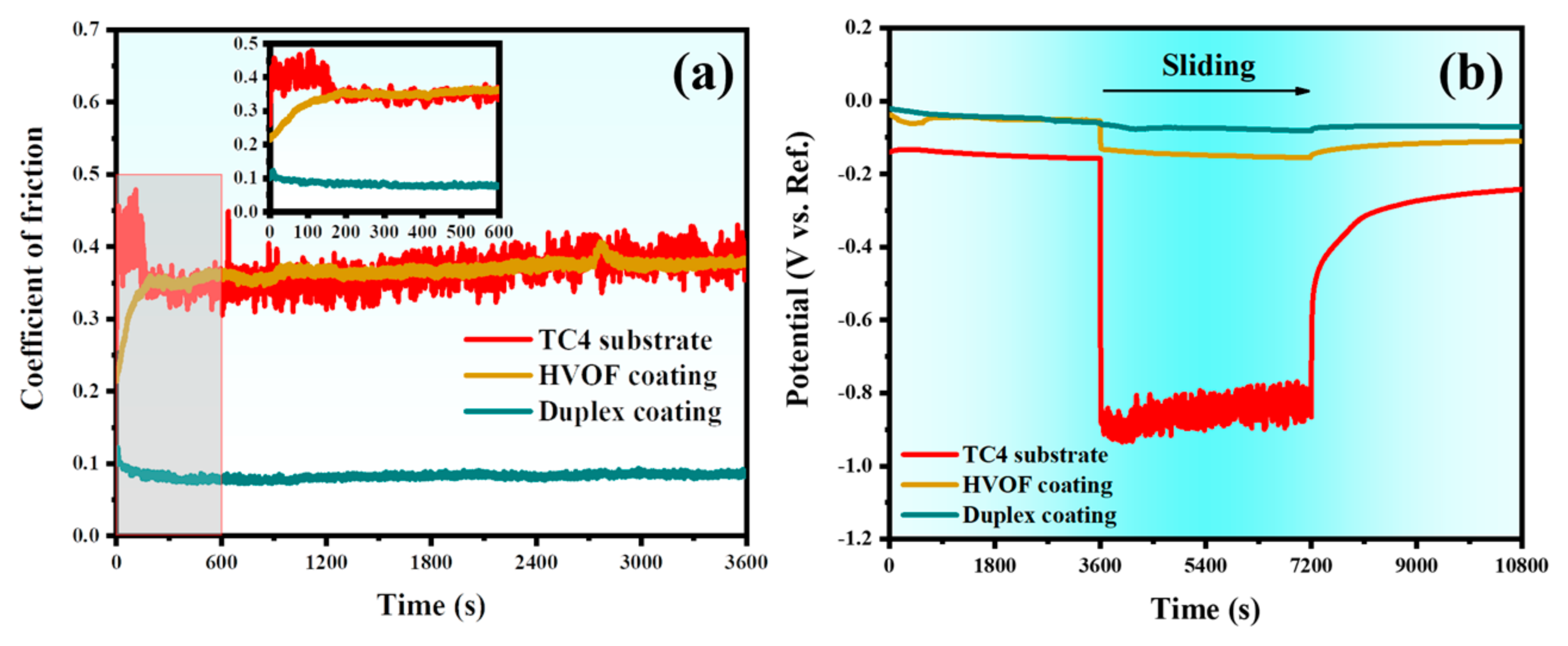
The coefficient of friction (COF) and OCP of the TC4 substrate, HVOF coating, and Duplex coating measured during the tribocorrosion tests are shown in Figure 9. It can be seen from Figure 9a that the COF of the TC4 substrate increases rapidly in the initial stage of sliding and then changes drastically between 0.25 and 0.55 with pronounced fluctuations. The COF value of the HVOF coating is similar to that of the TC4 substrate, except that the amplitude of fluctuation is slightly reduced. It is worth noting that the COF of the Duplex coating shows a completely different trend compared to the TC4 substrate and the HVOF coating. The COF of the Duplex coating decreased slightly during the start of the sliding and then stabilized around ~0.08 with very little fluctuation. Interestingly, it was observed that the COF of both the TC4 substrate and the HVOF coating increased gradually at the initial stage of sliding, then stabilized with varying degrees of fluctuation. In contrast, the COF of the Duplex coating exhibited a decreasing trend at the onset, eventually reaching a stable and smooth plateau. This behavior is likely attributed to the progressive graphitization of the DLC top layer under shear stress from the Al2O3 ball during the running-in period, leading to a reduction in COF. Subsequently, the graphitized DLC film provided persistent lubrication, maintaining the COF of the Duplex coating at a consistently low and stable value. The phenomenon of DLC graphitization will be discussed in detail in the following section. The OCP of the TC4 substrate, HVOF, and Duplex coatings measured during the entire tribocorrosion test (including before and after sliding), are illustrated in Figure 9b. Before sliding, all the samples were immersed in 3.5 wt% NaCl for 3600 s to obtain a stable OCP. When the sliding starts, it was observed that the OCP of the TC4 substrate dropped from −0.15 V to −0.88 V, with a huge diminution. It implies that the corrosion tendency of the TC4 substrate had increased significantly. After that, the OCP remained relatively stable at a low level with a weak amplitude. When the sliding stopped, the OCP quickly rose to about -0.5 V, and then slowly rose to about −0.24 V. In a static environment, TC4 substrate had excellent corrosion resistance due to the protection offered by the surficial passivation film [26]. However, the passivation film on the surface of the TC4 substrate is removed rapidly during sliding. Thus, its potential dropped significantly. Subsequently, the exposed TC4 substrate was re-passivated in 3.5 wt% NaCl, resulting in a slight recovery of potential within a short period. However, under the action of sliding wear, the new passivation film was rapidly removed. This repassivation/depassivation situation is the reason for the fluctuations in the potentials of the TC4 substrate. After the sliding, the passivation film was found to form swiftly on the surface of the fresh TC4 substrate, resulting in the potential returning to a higher value.
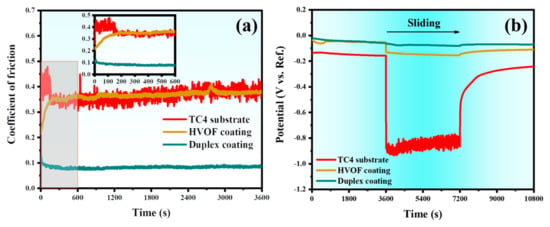
Figure 9.
(a) COF and (b) OCP of the TC4 substrate, HVOF, and Duplex coatings during the tribocorrosion tests.
On the other hand, the potential of the HVOF coating drops from −0.06 V to −0.13 V during sliding. It can be seen from Figure 9a that the tribological performance of the HVOF coating is poor. The exposed fresh surface in the wear track is more prone to corrosion, compared to the oxidized surface [27]. Therefore, the potential of the HVOF coating dropped. Unlike the TC4 substrate, the HVOF coating did not display an obvious passivation. Therefore, the OCP of the HVOF coating stabilized at the lowest value and remained constant during the sliding process. After the sliding, the fresh surface of the HVOF coating was re-oxidized, and then the potential was found to rebound. It should be noted that there is almost no potential change in the Duplex coating during the whole tribocorrosion process. It indicates that the Duplex coating was virtually undamaged during the sliding process. In addition, the protection offered by the chemically inert DLC film was another linchpin reason for the non-decreasing potential of the Duplex coating during the sliding process. Hence, it can be summarised that the Duplex coating exhibits excellent tribocorrosion resistance.
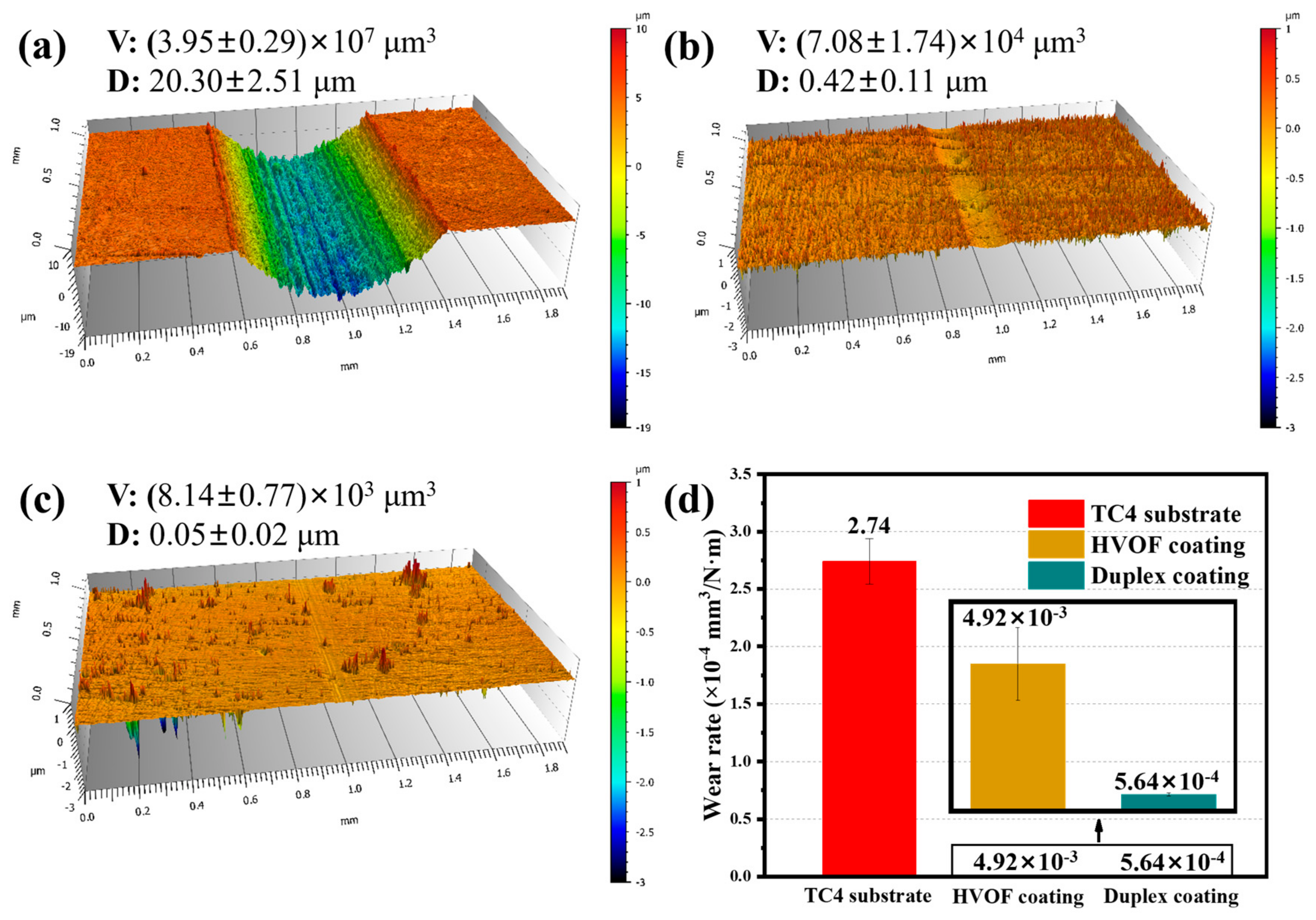
The 3D contours of the wear tracks of the TC4 substrate, HVOF coating, and Duplex coating are shown in Figure 10. It can be clearly seen that the TC4 substrate (20.30 ± 2.51 μm, (3.95 ± 0.29) × 107 μm3) had the deepest and widest wear track compared to the coated samples. Moreover, a pronounced wear track was also observed on the HVOF coating (0.42 ± 0.11 μm, (7.08 ± 1.74) × 104 μm3). However, there was almost no apparent wear track on the Duplex coating (0.05 ± 0.02 μm, (8.14 ± 0.77) × 103 μm3) surface. The volume wear rate of the samples is shown in Figure 10d. It can be observed that the wear rate of the Duplex coating was only (5.64 ± 0.53) × 10−8 mm3/N·m, which was about four and an order of magnitude lower than that of the TC4 substrate ((2.74 ± 0.20) × 10−4 mm3/N·m) and HVOF coating ((4.92 ± 1.21) × 10−7 mm3/N·m), respectively. It confirms the outstanding wear resistance of the Duplex coating.
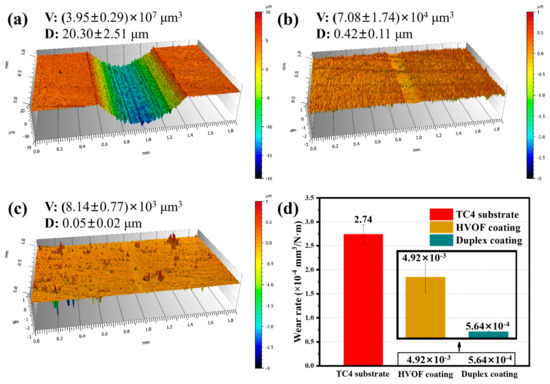
Figure 10.
(a–c) 3D profile diagram and (d) wear rate of the TC4 substrate, HVOF, and Duplex coatings after tribocorrosion.
3.5. Static Electrochemical Corrosion Mechanism
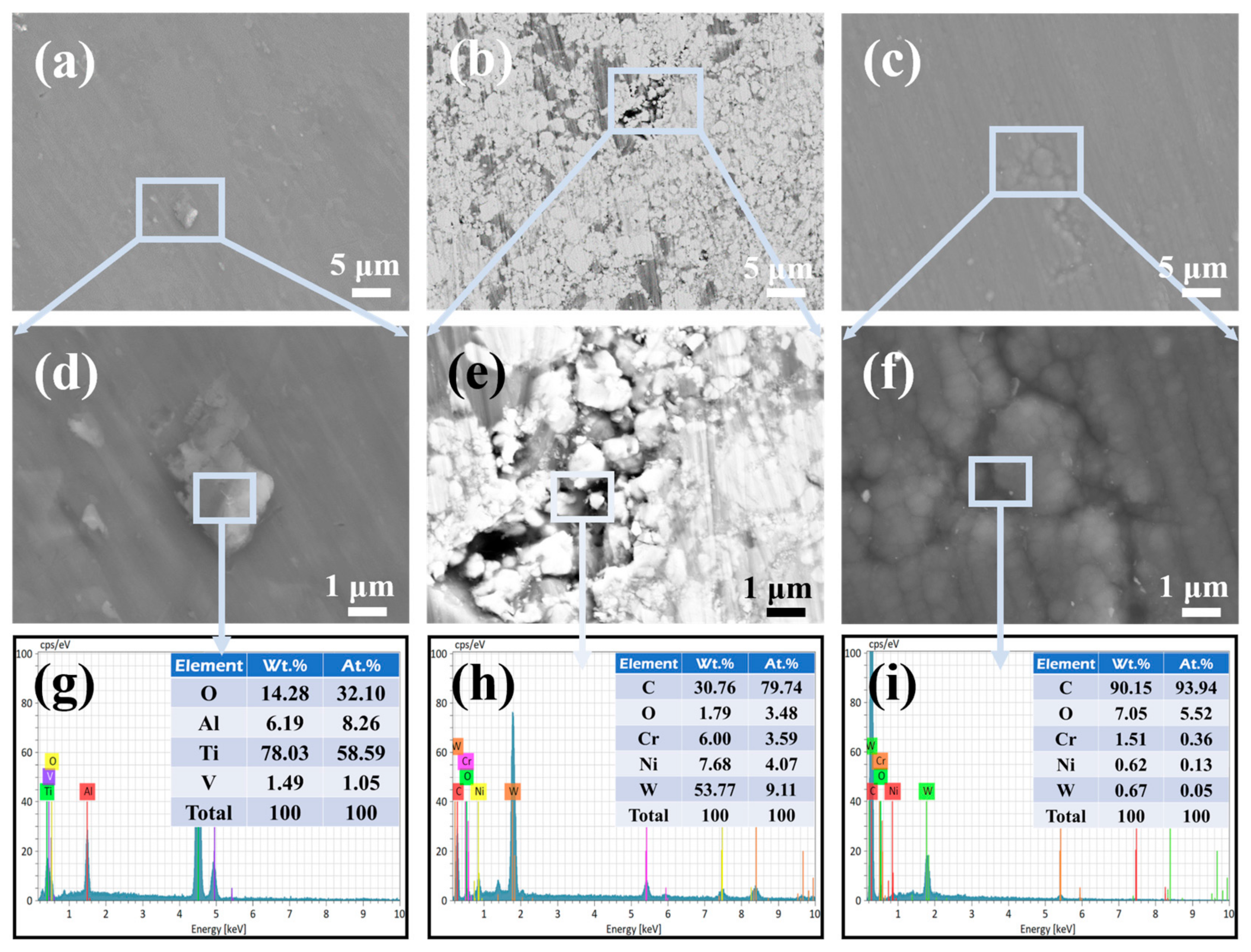
The surface morphologies of the TC4 substrate, HVOF coating, and Duplex coating after corrosion are displayed in Figure 11. As seen in Figure 11a, tiny irregular particles were found on the TC4 substrate after electrochemical corrosion. The EDS probing of the enlarged area (Figure 11d,g) found that the main elements of the particles comprised Ti and O. It means that the corrosion product formed by the TC4 substrate is mainly TiO2. The high-stability of the TiO2 film provides shielding for the TC4 substrate, which gives it an excellent corrosion resistance under static conditions [28].
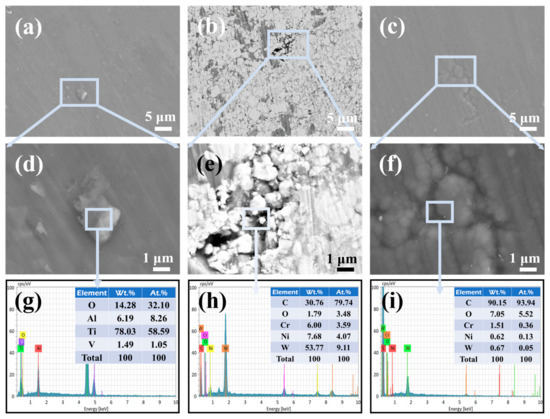
Figure 11.
The surface morphology and elemental composition (selected area) of the (a,d,g) TC4 substrate, (b,e,h) HVOF coating, and (c,f,i) Duplex coating after electrochemical corrosion.
In addition, no obvious corrosion products were observed on the surface of the HVOF coating (Figure 11b,e); however, microcracks and defects were found after corrosion. It is caused by the intrinsic characteristics of thermal spraying [29]. The EDS result (Figure 11h) shows that the oxygen content is very small at the crack. According to reports [28,30], the electrochemical corrosion performance of the HVOF coating is more controlled by the pores and defects in the coating. It is worth noting that the defects similar to the ones seen on the HVOF coating were also found on the corroded Duplex coating (Figure 11c). The difference is that the defect is evenly filled with dark material in the Duplex coating (Figure 11f). The EDS (Figure 11i) inspection revealed that the main element at the crack was carbon element. To further elucidate the superior corrosion resistance of the Duplex coating relative to the HVOF coating, the post-corrosion cross-sectional morphology of the Duplex coating was examined using a combined focused ion beam and scanning electron microscopy (FIB-SEM) system. Figure 12a,b shows FIB selection area and EDS layered image, respectively.

Figure 12.
The (a) cross-sectional morphology, (b) EDS layered image, and (c–e) ion thinning of the Duplex coating after electrochemical corrosion.
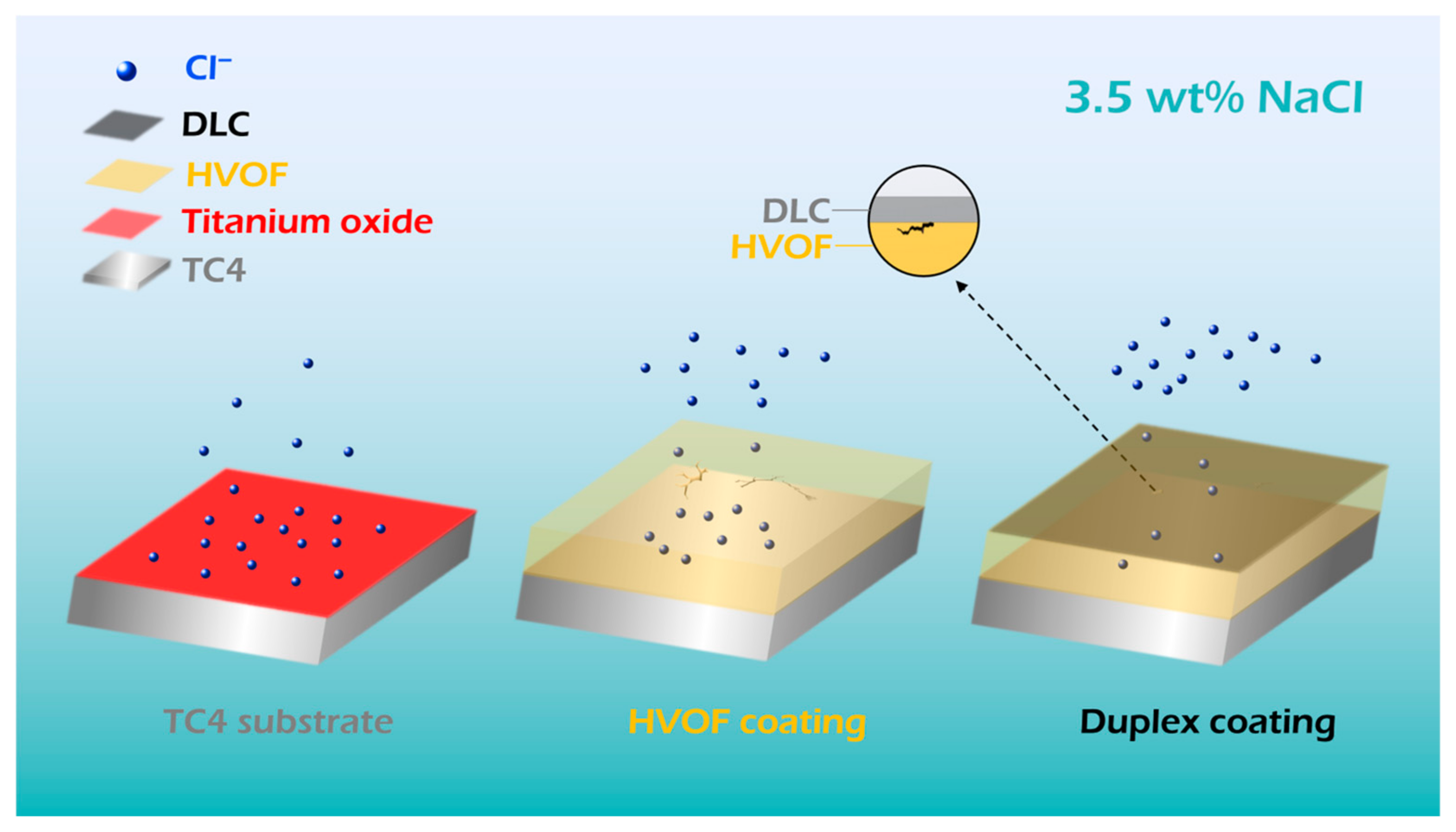
After thinning the area in turn (Figure 12c–e), it was found that there are obvious holes in the HVOF coating. However, the DLC covered coating still maintains a dense state. The top DLC coating acts as an effective barrier, preventing corrosive liquids from penetrating the underlying HVOF coating through surface defects. This conclusion is corroborated by both the electrochemical equivalent circuit data and the post-corrosion surface morphology of the Duplex coating (Figure 11). The electrochemical corrosion diagram of the TC4 substrate, HVOF, and Duplex coatings is shown in Figure 13. It can be understood that the chemically inert DLC film uniformly covers the cracks and holes of the HVOF coating. Therefore, the current density of the Duplex coating (3.54 × 10−8 A/cm2) decreased by two orders of magnitude compared to the HVOF coating (1.08 × 10−6 A/cm2), thus exhibiting outstanding corrosion resistance. In addition, the excellent corrosion resistance of the TC4 substrate is attributed to the surface-stable TiO2 passivation film.
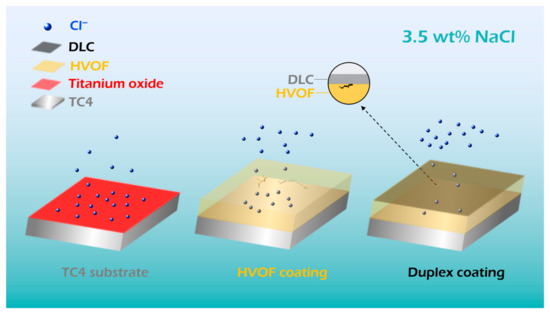
Figure 13.
Schematic of the static electrochemical corrosion process of the TC4 substrate, HVOF coating, and Duplex coating.
3.6. Tribocorrosion Mechanism
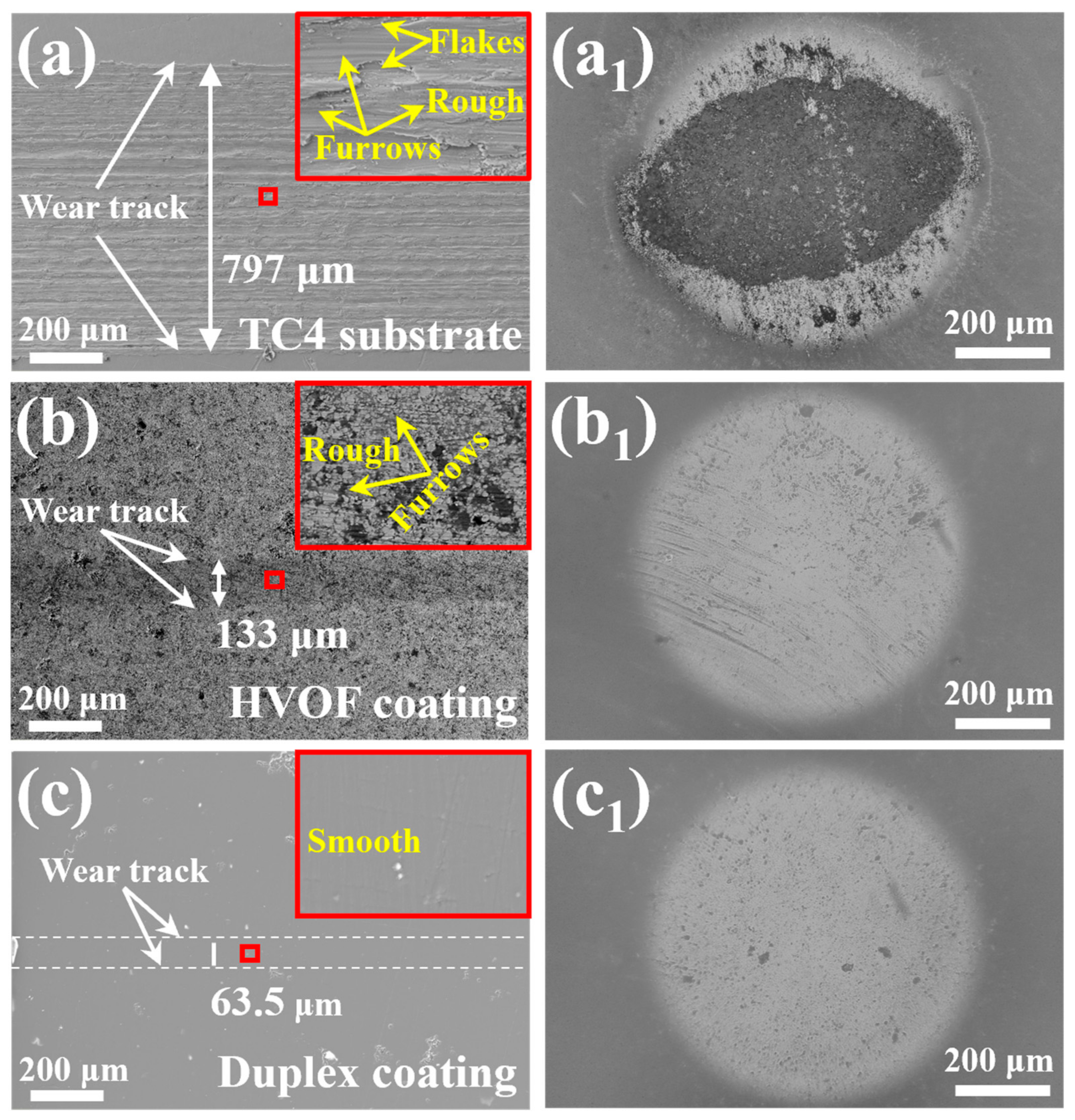
The wear track morphologies of the TC4 substrate, HVOF coating, and Duplex coating following tribocorrosion tests are presented in Figure 14a–c. As depicted in Figure 14a,b, the wear tracks on both the TC4 substrate and the HVOF coating exhibit scattered furrows of varying severity. Notably, the TC4 substrate demonstrates more pronounced furrowing and material flaking relative to the HVOF coating. Under the cutting action of the high hardness Al2O3 ball, the soft TC4 substrate suffered a severe abrasive wear [31]. Therefore, the material loss of the HVOF coating is less than that of the TC4 substrate. It is worth mentioning that the wear track of the Duplex coating (63.5 μm) is the narrowest and smoothest compared to the TC4 substrate (797 μm) and the HVOF coating (133 μm). Moreover, the enlarged image of the wear track (upper right corner of Figure 14c) had no obvious scratches. It indicates that the Duplex coating underwent only a slight fatigue wear, which lead to a very little material loss. Generally, the coatings with high hardness have better wear resistance [22]. Therefore, it is not difficult to understand that the wear rate of the HVOF coating (4.92 × 10−7 mm3/N·m) is much lower than that of the TC4 substrate (2.74 × 10−4 mm3/N·m).
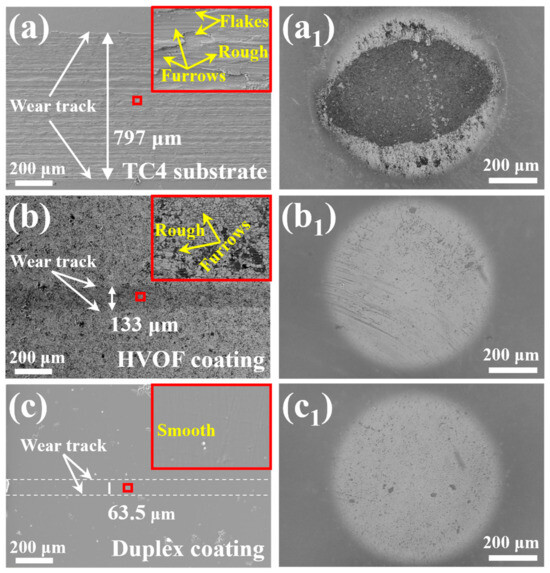
Figure 14.
The wear tracks morphology (the high-magnification image of the selected area is in the upper right corner) and their corresponding Al2O3 balls of the (a,a1) TC4 substrate, (b,b1) HVOF coating, and (c,c1) Duplex coating.
In addition, Duplex coating had the lowest COF (~0.08) and wear rate (5.64 × 10−8 mm3/N·m) among all samples. The Duplex coating exhibited an approximately one-order-of-magnitude lower wear rate and a significantly reduced COF compared to the HVOF coating. This marked improvement in tribological performance is attributed to the application of a dense DLC film, which provides high hardness and self-lubricating properties [32]. Furthermore, while the corrosion potential of the HVOF coating decreased during tribocorrosion, that of the Duplex coating remained stable. This stability can be ascribed to the additional protection imparted by the compact and chemically inert DLC film over the underlying HVOF coating. Figure 14a1–c1 displays images of the Al2O3 counterpart balls paired with the TC4 substrate, HVOF coating, and duplex coating, respectively. A substantial amount of wear debris was found adhered to the surface of the ball paired with the TC4 substrate (Figure 14a1). In contrast, no significant debris was observed on the ball from the HVOF coating pair (Figure 14b1), with only some scratching present. These observations suggest that the sliding of the soft TC4 substrate against the Al2O3 ball involved both adhesive and abrasive wear mechanisms, while material removal from the HVOF coating occurred predominantly by abrasive wear. In distinct contrast, neither debris nor scratches were detected on the ball paired with the Duplex coating (Figure 14c1). This indicates that the Al2O3 ball remained virtually intact after sliding, and consequently, its wear did not significantly influence the test results. The outer/inner wear tracks of the samples (labeled as sampleO/sampleI, respectively) are analyzed by XPS spectra after tribocorrosion. The relative atomic concentrations of the elements on the sample surface are listed in Table 5.

Table 5.
The atomic concentration of elements outside/inside the wear track obtained by XPS detection.
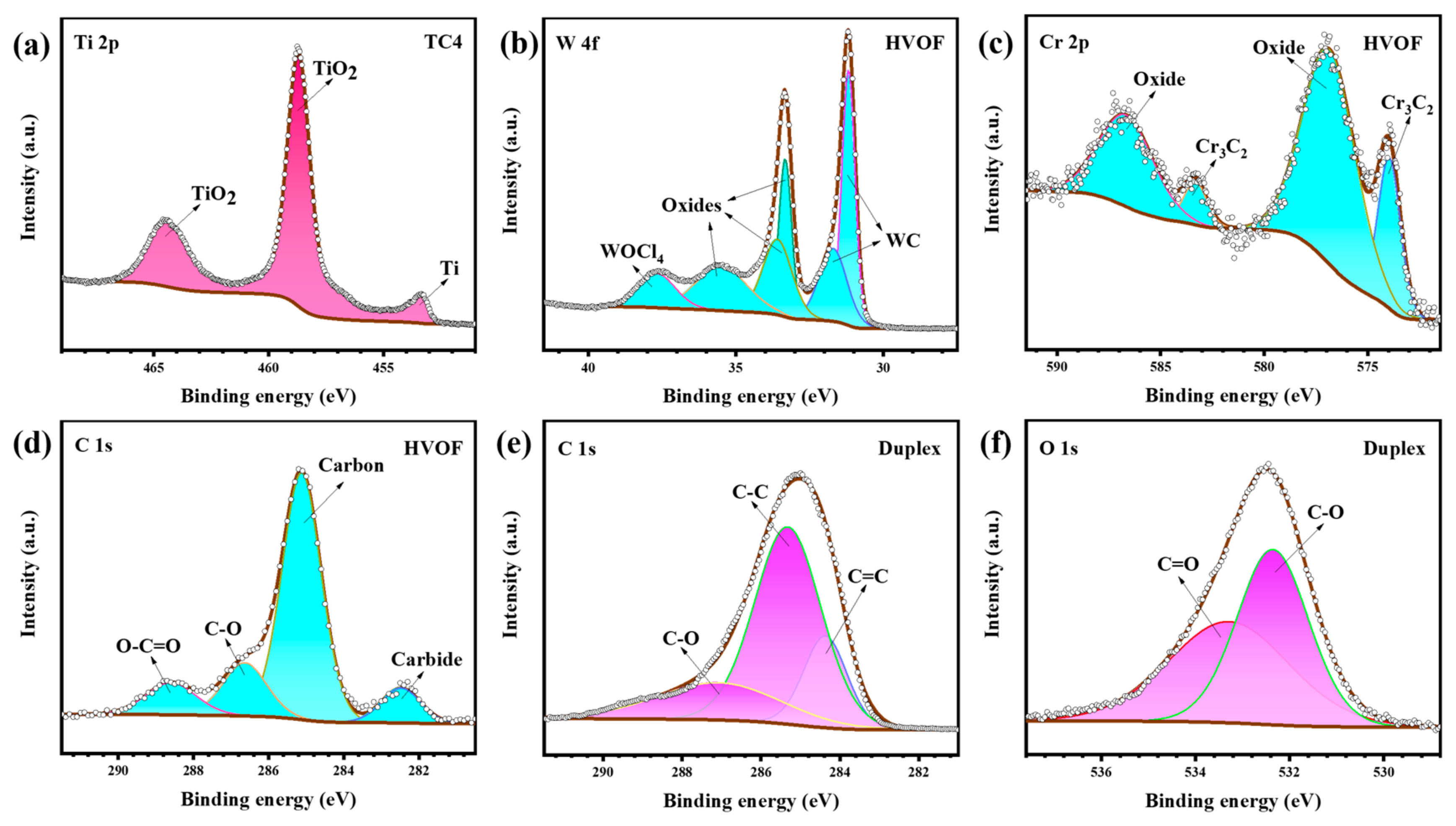
It can be seen that the content of elements inside and outside the wear track of the sample are not much different. According to the tribocorrosion results (Figure 9), the samples were in a depassivated/repassivated state during the entire sliding process. Upon the completion of sliding, a reformed oxide film renders the elemental composition largely uniform across and outside the wear track. Notably, the oxygen (O) content within the TC4 substrate’s wear track is lower than in the surrounding area, suggesting the formation of a thinner passivation film post-sliding. In contrast, the HVOF and Duplex coatings exhibit a slight enrichment of O after sliding. This is hypothesized to result from shear-induced instability in the near-surface region, promoting mechanical mixing between oxygen and elements such as W, Cr, and C [33]. The XPS high-resolution spectra of the samples within the wear track are shown in Figure 15.
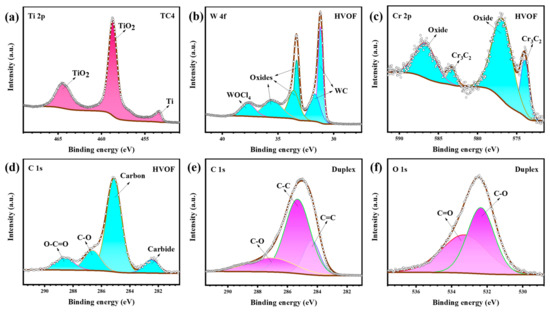
Figure 15.
XPS high-resolution spectra of the (a) TC4 substrate, (b–d) HVOF coating, and (e,f) Duplex coating (inside the wear track) after tribocorrosion.
From Figure 15a, it can be seen that the wear track of the TC4 substrate is mainly composed of TiO2 (464.5 eV, 458.7 eV) and a tiny amount of Ti (453.4 eV). Figure 15b–d presents the results of the XPS peak fitting for the HVOF coating. The W 4f peak of the HVOF coating can be convolved as WOCl4 (37.7 eV), oxides (35.5 eV, 33.6 eV, 33.3 eV), and WC (31.7 eV, 31.2 eV). And its Cr 2p peak contains both oxide (586.8 eV, 577.0 eV) and Cr3C2 (583.3 eV, 574.0 eV) forms [34]. It can be seen that the W and Cr elements in the wear track of the HVOF coating mainly contain carbide (carbide is further illustrated by Figure 15d) and oxide phases. It is reported that an appropriate amount of oxide on the surface of HVOF coating contributes to the wear performance [35]. The C 1s peak of the Duplex coating (Figure 15e) were fitted to C-O (287.1 eV), C-C (285.3 eV) and C=C (284.4 eV) bonds. Moreover, it is noted that the major peaks are still C-C and C=C bonds, except for the low-intensity C-O bonds (further confirmed to be C-O/C=O bonds in Figure 15f). This carbon peak, which is mainly composed of sp3-C bond and sp2-C bond, is a typical characteristic of DLC [36]. It shows that the DLC coating on top of the Duplex coating doesn’t suffer much damage after tribocorrosion. The structure evolution of the DLC coating after tribocorrosion was further studied by Raman spectroscopy. As shown in Figure 16, it can be seen that it is similar to the Raman characteristics of the initial DLC coating (Figure 4).
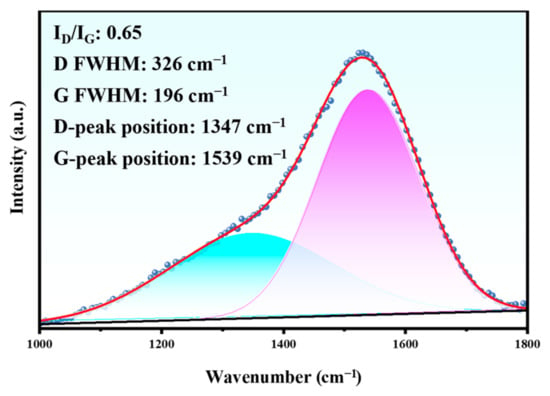
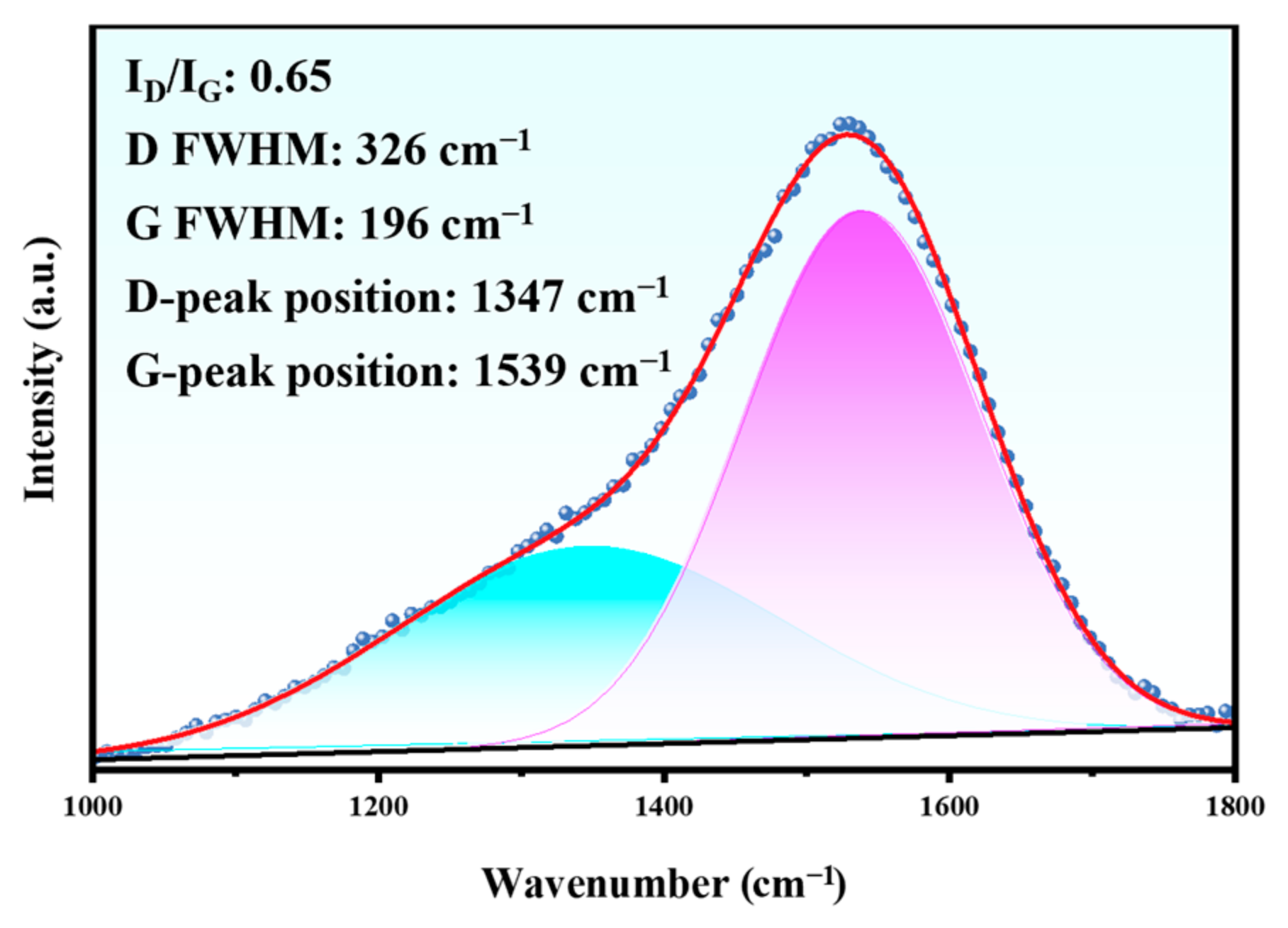
Figure 16.
Raman spectrum in the wear track of the Duplex coating.
This indicates that the tribocorrosion effect did not significantly affect the DLC coating. However, the ID/IG value of the tribocorrosion sample (0.65) is slightly higher than the initial value (0.55), which implies that the DLC coating had somewhat undergone a graphitized transformation [37]. It is not difficult to understand that the hard DLC coating when subjected to the reciprocating friction of the Al2O3 ball, resulted in the transformation of the diamond phase to the graphite phase on the surface.
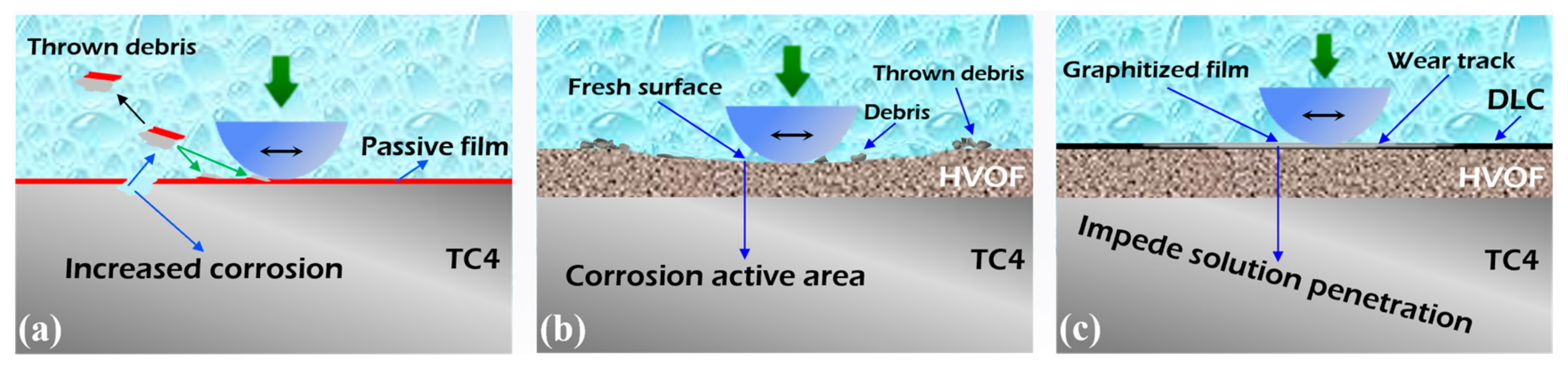
The schematic diagram describing the tribocorrosion process of the TC4 substrate, HVOF coating, and HVOF coating is shown in Figure 17. Due to the low hardness of the TC4 substrate (329.09 HV0.1), severe abrasive wear and adhesive wear occurred under the friction of the hard Al2O3 ball. Therefore, the passivation film on the surface is directly removed to expose the TC4 substrate. After losing the protection of the surface passivation film, its corrosion rate was seriously intensified. Similarly to the TC4 substrate, the HVOF (WC-20Cr3C2-7Ni) coating underwent a slight abrasive wear during tribocorrosion. The exposed fresh surface became an active corrosion area, which exhibited a decrease in the OCP. Unlike the TC4 substrate and the HVOF coating, the chemically inert DLC coating was extremely difficult to be corroded by the electrolyte [38]. Moreover, the DLC coating had excellent tribological properties due to its high hardness and self-lubricating characteristics [39,40]. Simultaneously, the DLC coating was more dense and uniform compared to the HVOF coating, which further impedes the penetration of the corrosive liquid. Therefore, the tribocorrosion performance of the HVOF coating was significantly improved after covering it with the DLC coating.
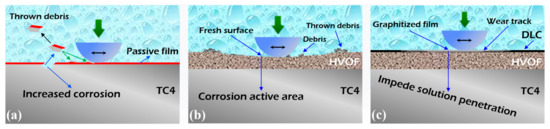
Figure 17.
Schematic diagram of tribocorrosion for (a) TC4 substrate, (b) HVOF coating, and (c) Duplex coating.
3.7. Limitations of the Tribocorrosion Analysis
Although this work investigated the corrosion and tribocorrosion behavior of the DLC-modified WC cermet duplex coating and provided preliminary insights into its underlying protection mechanism, certain limitations remain. It is widely recognized that materials often experience more severe degradation under combined corrosion–wear conditions than the simple sum of individual effects, a phenomenon typically attributed to a synergistic interaction that accelerates material loss [3]. Conversely, antagonistic effects between corrosion and wear have also been reported, leading to a total material loss that is less than the additive sum from individual corrosion and wear tests [41]. However, since pure wear tests (e.g., in a non-corrosive medium) were not conducted in this study, a further discussion on the specific nature of the corrosion–wear interaction (synergistic or antagonistic) is constrained. Future work should address this limitation by performing controlled wear tests in environments such as distilled water to decouple the mechanisms and enable a deeper exploration of the tribocorrosion coupling effects.
4. Conclusion
In summary, it was demonstrated that the Duplex coating was successfully deposited on the TC4 substrate via HVOF coupled LIS system, and its corrosion and tribocorrosion behavior in 3.5 wt% NaCl was investigated. The key findings are as follows:
- (1)
- In a static environment, the Duplex coating had the outstanding corrosion resistance compared to the HVOF coating and TC4 substrate, which is attributed to the protection offered by the dense, uniform, and chemically inert DLC coating.
- (2)
- Under the dynamic (tribocorrosion) conditions, the Duplex coating exhibited no significant decrease in potential and had an extremely low COF (~0.08) and wear rate (5.64 × 10−8 mm3/N·m). This is due to the reinforcement of the high hardness, self-lubricating, and corrosion-resistant DLC top layer, which provided the Duplex coating with a superb tribocorrosion resistance.
- (3)
- The novel HVOF-PVD/CVD duplex coating represents a promising and viable universal strategy for protecting metal components/equipment exposed to saline/marine environments.
Author Contributions
Investigation, data curation, writing—original draft, Y.W.; methodology, Z.L.; writing—review and editing, funding acquisition, supervision, Y.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the Zhejiang Provincial Natural Science Foundation of China (LQN26E010018), Specialty Fund of Zhejiang Institute of Tianjin University (ZITJU2024-ZYHT006), and Start-up Fund from Ningbo University of Technology (24KQ088).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the articles. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Liu, X.-P.; Jin, Z.-J.; Bai, G.-P.; Guan, M.; Liu, J.; Pan, Q.-H.; Li, T.; Xing, Y.-J. Formation and distribution characteristics of Proterozoic–Lower Paleozoic marine giant oil and gas fields worldwide. Pet. Sci. 2017, 14, 237–260. [Google Scholar] [CrossRef]
- Oliveira-Pinto, S.; Rosa-Santos, P.; Taveira-Pinto, F. Assessment of the potential of combining wave and solar energy resources to power supply worldwide offshore oil and gas platforms. Energy Convers. Manag. 2020, 223, 113299. [Google Scholar] [CrossRef]
- Wood, R.J. Marine wear and tribocorrosion. Wear 2017, 376, 893–910. [Google Scholar] [CrossRef]
- Wang, Q.; Luo, S.; Wang, S.; Wang, H.; Ramachandran, C.S. Wear, erosion and corrosion resistance of HVOF-sprayed WC and Cr3C2 based coatings for electrolytic hard chrome replacement. Int. J. Refract. Met. Hard Mater. 2019, 81, 242–252. [Google Scholar] [CrossRef]
- Tang, P.; He, D.; Li, W.; Shang, L.; Zhai, H.; Wang, L.; Zhang, G. Achieving superior hot corrosion resistance by PVD/HVOF duplex design. Corros. Sci. 2020, 175, 108845. [Google Scholar] [CrossRef]
- Li, S.; Li, H.; Ma, G.; Wei, J.; Zhou, G.; Zhang, Y.; Guo, P.; Ke, P.; Wang, A. Dense Cr/GLC multilayer coating by HiPIMS technique in high hydrostatic pressure: Microstrusctural evolution and galvanic corrosion failure. Corros. Sci. 2023, 225, 111618. [Google Scholar] [CrossRef]
- Shiri, S.; Ashtijoo, P.; Odeshi, A.; Yang, Q. Evaluation of Stoney equation for determining the internal stress of DLC thin films using an optical profiler. Surf. Coat. Technol. 2016, 308, 98–100. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Q.; Ramachandran, C.S.; Guo, P.; Wang, A. Microstructure and performance of high-velocity oxygen-fuel coupled physical vapor deposition (HVOF-PVD) duplex protective coatings: A review. Coatings 2022, 12, 1395. [Google Scholar] [CrossRef]
- Li, W.; Tang, P.; Shang, L.; He, D.; Wang, L.; Zhang, G.; Jin, K. Tribological behaviors of CrN/Cr3C2-NiCr duplex coating at elevated temperatures. Surf. Coat. Technol. 2019, 378, 124926. [Google Scholar] [CrossRef]
- Zheng, W.; He, D.; Li, W.; Shang, L.; Song, Q.; Zhang, G.; Zhai, H.; Cheng, B. AlCrN/Cr3C2–NiCr duplex coating towards high load-bearing and dry sliding antiwear applications. Ceram. Int. 2022, 48, 18933–18943. [Google Scholar] [CrossRef]
- Zhang, Q.; Shao, L.; Li, W.; Cui, S.; Shang, L.; Wang, C.; Song, Q.; Zhang, C. Comparative study of anti-corrosion properties in different CrN/WC-Co duplex coatings processed by PVD/HVOF. Surf. Coat. Technol. 2024, 483, 130799. [Google Scholar] [CrossRef]
- Pougoum, F.; Qian, J.; Martinu, L.; Klemberg-Sapieha, J.; Zhou, Z.; Li, K.Y.; Savoie, S.; Lacasse, R.; Potvin, E.; Schulz, R. Study of corrosion and tribocorrosion of Fe3Al-based duplex PVD/HVOF coatings against alumina in NaCl solution. Surf. Coat. Technol. 2019, 357, 774–783. [Google Scholar] [CrossRef]
- Monticelli, C.; Balbo, A.; Zucchi, F. Corrosion and tribocorrosion behaviour of cermet and cermet/nanoscale multilayer CrN/NbN coatings. Surf. Coat. Technol. 2010, 204, 1452–1460. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H.; Cui, L.; Yang, W.; Ma, G.; Chen, R.; Guo, P.; Ke, P.; Wang, A. Comparative study on tribocorrosion behavior of hydrogenated/hydrogen-free amorphous carbon coated WC-based cermet in 3.5 wt% NaCl solution. Corros. Sci. 2024, 227, 111738. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, X.; Chen, R.; Yang, W.; Guo, P.; Nishimura, K.; Li, X.; Wang, A. Ultrahigh tribocorrosion resistance of hydrogenated amorphous carbon coating via trace gradient W incorporation. Corros. Sci. 2024, 240, 112504. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Li, L.; Li, H.; Guo, P.; Wang, A. Effect of alternating hydrostatic pressure (AHP) on the corrosion and tribocorrosion behavior of gradient W-DLC coated WC-based cermet. Corros. Sci. 2025, 256, 113190. [Google Scholar] [CrossRef]
- Jiang, X.; Meng, L.; Guo, P.; Chen, R.; Wang, Z.; Li, H.; Nishimura, K.; Wang, A.; Ke, P. Improved tribological performance of aC: H/aC: H: Si: O coated polyether ether ketone by introducing graded interfacial structure. Wear 2025, 582-583, 206326. [Google Scholar] [CrossRef]
- Wang, Y.; Alsmeyer, D.C.; McCreery, R.L. Raman spectroscopy of carbon materials: Structural basis of observed spectra. Chem. Mater. 1990, 2, 557–563. [Google Scholar] [CrossRef]
- Valencia, F.J.; Santiago, J.; González, R.I.; González-Arrabal, R.; Ruestes, C.; Díaz, M.P.; Monclus, M.A.; Molina-Aldareguia, J.; Nuñez, P.D.; Munoz, F. Nanoindentation of Amorphous Carbon: A combined experimental and simulation approach. Acta Mater. 2021, 203, 116485. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Raman spectroscopy of amorphous, nanostructured, diamond–like carbon, and nanodiamond. Philos. Trans. R. Soc. London. Ser. A Math. Phys. Eng. Sci. 2004, 362, 2477–2512. [Google Scholar] [CrossRef]
- Bolelli, G.; Lusvarghi, L.; Mantini, F.P.; Pitacco, F.; Volz, H. Enhanced tribological properties of PECVD DLC coated thermally sprayed coatings. Surf. Coat. Technol. 2008, 202, 4382–4386. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Q.; Chen, G.; Ramachandran, C.S. Mechanical, tribological and corrosion physiognomies of CNT-Al metal matrix composite (MMC) coatings deposited by cold gas dynamic spray (CGDS) process. Surf. Coat. Technol. 2020, 403, 126380. [Google Scholar] [CrossRef]
- Hirschorn, B.; Orazem, M.E.; Tribollet, B.; Vivier, V.; Frateur, I.; Musiani, M. Determination of effective capacitance and film thickness from constant-phase-element parameters. Electrochim. Acta 2010, 55, 6218–6227. [Google Scholar] [CrossRef]
- Daroonparvar, M.; Khan, M.F.; Saadeh, Y.; Kay, C.; Kasar, A.; Kumar, P.; Esteves, L.; Misra, M.; Menezes, P.; Kalvala, P. Modification of surface hardness, wear resistance and corrosion resistance of cold spray Al coated AZ31B Mg alloy using cold spray double layered Ta/Ti coating in 3.5 wt% NaCl solution. Corros. Sci. 2020, 176, 109029. [Google Scholar] [CrossRef]
- Lu, F.-F.; Ma, K.; Li, C.-X.; Yasir, M.; Luo, X.-T.; Li, C.-j. Enhanced corrosion resistance of cold-sprayed and shot-peened aluminum coatings on LA43M magnesium alloy. Surf. Coat. Technol. 2020, 394, 125865. [Google Scholar] [CrossRef]
- Manhabosco, T.M.; Tamborim, S.; Dos Santos, C.; Müller, I. Tribological, electrochemical and tribo-electrochemical characterization of bare and nitrided Ti6Al4V in simulated body fluid solution. Corros. Sci. 2011, 53, 1786–1793. [Google Scholar] [CrossRef]
- Mindivan, H. Corrosion and tribocorrosion behaviour of WC/C coating on beryllium-copper mould alloy. Mater. Today Proc. 2020, 27, 3114–3118. [Google Scholar] [CrossRef]
- Totolin, V.; Pejaković, V.; Csanyi, T.; Hekele, O.; Huber, M.; Ripoll, M.R. Surface engineering of Ti6Al4V surfaces for enhanced tribocorrosion performance in artificial seawater. Mater. Des. 2016, 104, 10–18. [Google Scholar] [CrossRef]
- Zhou, Z.; Wang, L.; Wang, F.; Zhang, H.; Liu, Y.; Xu, S. Formation and corrosion behavior of Fe-based amorphous metallic coatings by HVOF thermal spraying. Surf. Coat. Technol. 2009, 204, 563–570. [Google Scholar] [CrossRef]
- Guilemany, J.; Espallargas, N.; Suegama, P.; Benedetti, A.V. Comparative study of Cr3C2–NiCr coatings obtained by HVOF and hard chromium coatings. Corros. Sci. 2006, 48, 2998–3013. [Google Scholar] [CrossRef]
- Zhang, M.; Zhou, F.; Fu, Y.; Wang, Q.; Kong, J. Electrochemical corrosion and tribological properties of CrMoCN coatings sliding against Al2O3 balls in artificial seawater. Surf. Coat. Technol. 2021, 417, 127225. [Google Scholar] [CrossRef]
- Bewilogua, K.; Hofmann, D. History of diamond-like carbon films—From first experiments to worldwide applications. Surf. Coat. Technol. 2014, 242, 214–225. [Google Scholar] [CrossRef]
- Chen, X.; Ma, Y.; Yang, Y.; Meng, A.; Han, Z.; Han, Z.; Zhao, Y. Revealing tribo–oxidation mechanisms of the copper–WC system under high tribological loading. Scr. Mater. 2021, 204, 114142. [Google Scholar] [CrossRef]
- Jiang, K.; Zhao, D.; Jiang, X.; Huang, Q.; Miao, L.; Lu, H.; Li, Y. Electronic-structure, corrosion and mechanical properties of nc-CrC/aC: H films deposited by multi-arc ion plating. J. Alloys Compd. 2018, 750, 560–569. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Kuroda, S.; Kawakita, J.; Sakamoto, Y.; Takaya, M. Sliding wear properties of HVOF sprayed WC–20% Cr3C2–7% Ni cermet coatings. Surf. Coat. Technol. 2007, 201, 4718–4727. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, J.; Luo, T.; Cao, B.; Xu, J.; Chen, X.; Luo, J. Tribochemical mechanism of superlubricity in graphene quantum dots modified DLC films under high contact pressure. Carbon 2021, 173, 329–338. [Google Scholar] [CrossRef]
- Bueno, A.; Solis, J.; Zhao, H.; Wang, C.; Simoes, T.; Bryant, M.; Neville, A. Tribocorrosion evaluation of hydrogenated and silicon DLC coatings on carbon steel for use in valves, pistons and pumps in oil and gas industry. Wear 2018, 394, 60–70. [Google Scholar] [CrossRef]
- Cai, W.; Sui, J. Effect of working pressure on the structure and the electrochemical corrosion behavior of diamond-like carbon (DLC) coatings on the NiTi alloys. Surf. Coat. Technol. 2007, 201, 5194–5197. [Google Scholar] [CrossRef]
- Tillmann, W.; Dias, N.F.L.; Stangier, D.; Hagen, L.; Schaper, M.; Hengsbach, F.; Hoyer, K.-P. Tribo-mechanical properties and adhesion behavior of DLC coatings sputtered onto 36NiCrMo16 produced by selective laser melting. Surf. Coat. Technol. 2020, 394, 125748. [Google Scholar] [CrossRef]
- Masripan, N.A.B.; Ohara, K.; Umehara, N.; Kousaka, H.; Tokoroyama, T.; Inami, S.; Zushi, K.; Fujita, M. Hardness effect of DLC on tribological properties for sliding bearing under boundary lubrication condition in additive-free mineral base oil. Tribol. Int. 2013, 65, 265–269. [Google Scholar] [CrossRef]
- Wood, R.J.; Herd, S.; Thakare, M.R. A critical review of the tribocorrosion of cemented and thermal sprayed tungsten carbide. Tribol. Int. 2018, 119, 491–509. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.