Omega-3 Long-Chain Polyunsaturated Fatty Acids Intake in Children with Attention Deficit and Hyperactivity Disorder
Abstract
1. Introduction
- 1)
- Evaluation of the pattern of consumption of the main dietary sources of food containing omega-3 LC-PUFA in children with ADHD and in a control group.
- 2)
- Estimation of the daily intake of omega-3 LC-PUFA (EPA + DHA) in the two groups.
- 3)
- Evaluation of the influence of age, sex, and body mass index (BMI) upon omega-3 LC-PUFA intake.
2. Materials and Methods
2.1. Study Design
2.2. Diet Assessment
2.3. Estimation of Omega-3 LC-PUFA Intake from Fish and Nuts
2.4. Statistical Analysis
3. Results
3.1. Description of the Sample
3.2. Energy Intake and Frequency of Seafood Consumption
3.3. Estimation of Omega-3 LC-PUFA (EPA + DHA) Intake
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sciberras, E.; Mulraney, M.; Silva, D.; Coghill, D. Prenatal Risk Factors and the Etiology of ADHD—Review of Existing Evidence. Curr. Psychiatry Rep. 2017, 19. [Google Scholar] [CrossRef]
- Nigg, J.; Nikolas, M.; Burt, S.A. Measured Gene by Environment Interaction in Relation to Attention-Deficit/Hyperactivity Disorder (ADHD). J. Am. Acad. Child Adolesc. Psychiatry 2010, 49, 863–873. [Google Scholar] [CrossRef]
- Sarris, J.; Logan, A.C.; Akbaraly, T.N.; Amminger, G.P.; Balanzá-Martínez, V.; Freeman, M.P.; Hibbeln, J.; Matsuoka, Y.; Mischoulon, D.; Mizoue, T.; et al. International Society for Nutritional Psychiatry Research. Nutritional medicine as mainstream in psychiatry. Lancet Psychiatry 2015, 2, 271–274. [Google Scholar] [CrossRef]
- Yan, X.; Zhao, X.; Li, J.; He, L.; Xu, M. Effects of early-life malnutrition on neurodevelopment and neuropsychiatric disorders and the potential mechanisms. Prog. Neuro Psychopharmacol. Boil. Psychiatry 2018, 83, 64–75. [Google Scholar] [CrossRef]
- Gow, R.V.; Hibbeln, J.R. Omega-3 Fatty Acid and Nutrient Deficits in Adverse Neurodevelopment and Childhood Behaviors. Child Adolesc. Psychiatr. Clin. Psychiatry 2014, 23, 555–590. [Google Scholar] [CrossRef]
- Grosso, G.; Galvano, F.; Marventano, S.; Malaguarnera, M.; Bucolo, C.; Drago, F.; Caraci, F. Omega-3 Fatty Acids and Depression: Scientific Evidence and Biological Mechanisms. Oxidative Med. Cell. Longev. 2014, 2014, 1–16. [Google Scholar] [CrossRef]
- Parletta, N.; Milte, C.M.; Meyer, B.J. Nutritional modulation of cognitive function and mental health. J. Nutr. Biochem. 2013, 24, 725–743. [Google Scholar] [CrossRef]
- Schuchardt, J.P.; Huss, M.; Stauss-Grabo, M.; Hahn, A. Significance of long-chain polyunsaturated fatty acids (PUFAs) for the development and behaviour of children. Eur. J. Pediatr. 2010, 169, 149–164. [Google Scholar] [CrossRef]
- Cooper, R.E.; Tye, C.; Kuntsi, J.; Vassos, E.; Asherson, P. Omega-3 polyunsaturated fatty acid supplementation and cognition: A systematic review and meta-analysis. J. Psychopharmacol. 2015, 29, 753–763. [Google Scholar] [CrossRef]
- Mischoulon, D.; Freeman, M.P. Omega-3 fatty acids in psychiatry. Psychiatr. Clin. North Am. 2013, 36, 15–23. [Google Scholar] [CrossRef]
- Bloch, M.H.; Hannestad, J. Omega-3 fatty acids for the treatment of depression: Systematic review and meta-analysis. Mol. Psychiatry 2012, 17, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Politi, P.; Rocchetti, M.; Emanuele, E.; Rondanelli, M.; Barale, F. Randomized Placebo-Controlled Trials of Omega-3 Polyunsaturated Fatty Acids in Psychiatric Disorders: A Review of the Current Literature. Drug Discov. Technol. 2013, 10, 245–253. [Google Scholar] [CrossRef]
- Bloch, M.H.; Qawasmi, A. Omega-3 Fatty Acid Supplementation for the Treatment of Children with Attention-Deficit/Hyperactivity Disorder Symptomatology: Systematic Review and Meta-Analysis. J. Am. Acad. Child Adolesc. Psychiatry 2011, 50, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Ramalho, R.; Pereira, A.C.; Vicente, F.; Pereira, P. Docosahexaenoic acid supplementation for children with attention deficit hyperactivity disorder: A comprehensive review of the evidence. Clin. Nutr. ESPEN 2018, 25, 1–7. [Google Scholar] [CrossRef]
- Agostoni, C.; Nobile, M.; Ciappolino, V.; Delvecchio, G.; Tesei, A.; Turolo, S.; Crippa, A.; Mazzocchi, A.; Altamura, C.A.; Brambilla, P. The Role of Omega-3 Fatty Acids in Developmental Psychopathology: A Systematic Review on Early Psychosis, Autism and ADHD. Int. J. Mol. Sci. 2017, 18, 2608. [Google Scholar] [CrossRef]
- Lange, K.W.; Hauser, J.; Makulska-Gertruda, E.; Nakamura, Y.; Reissmann, A.; Sakaue, Y.; Takano, T.; Takeuchi, Y. The Role of Nutritional Supplements in the Treatment of ADHD: What the Evidence Says. Curr. Psychiatry Rep. 2017, 19, 8. [Google Scholar] [CrossRef]
- Königs, A.; Kiliaan, A.J. Critical appraisal of omega-3 fatty acids in attention-deficit/hyperactivity disorder treatment. Neuropsychiatr. Dis. Treat. 2016, 12, 1869–1882. [Google Scholar]
- Arnold, L.E. Fish oil is not snake oil. J. Am. Acad. Child Adolesc. Psychiatry 2011, 50, 969–971. [Google Scholar] [CrossRef]
- Nigg, J.T.; Lewis, K.; Edinger, T.; Falk, M. Meta-analysis of attention-deficit/hyperactivity disorder symptoms, restriction diet and synthetic food color additives. J. Am. Acad. Child Adolesc. Psychiatry 2012, 21, 86–89. [Google Scholar] [CrossRef]
- Stevenson, J.; Buitelaar, J.; Cortese, S.; Ferrin, M.; Konofal, E.; Lecendreux, M.; Simonoff, E.; Wong, I.C.; Sonuga-Barke, E. Research review: The role of diet in the treatment of attention-deficit/hyperactivity disorder—An appraisal of the evidence on efficacy and recommendations on the design of future studies. J. Child Psychol. Psychiatry 2014, 55, 416–427. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar]
- Morgane, P.J.; Austin-LaFrance, R.; Bronzino, J.; Tonkiss, J.; Díaz-Cintra, S.; Cintra, L.; Kemper, T.; Galler, J.R.; Kemper, T. Prenatal malnutrition and development of the brain. Neurosci. Biobehav. Rev. 1993, 17, 91–128. [Google Scholar] [CrossRef]
- Bourre, J.M.; Dumont, O.; Piciotti, M.; Clément, M.; Chaudière, J.; Bonneil, M.; Nalbone, G.; Lafont, H.; Pascal, G.; Durand, G. Essentiality of omega 3 fatty acids for brain structure and function. World Rev. Nutr. Diet. 1991, 66, 103–117. [Google Scholar]
- Burgess, J.R.; Stevens, L.; Zhang, W.; Peck, L. Long-chain polyunsaturated fatty acids in children with attention-deficit hyperactivity disorder. Am. J. Clin. Nutr. 2000, 71, 327S–330S. [Google Scholar] [CrossRef] [PubMed]
- Pusceddu, M.M.; Kelly, P.; Stanton, C.; Cryan, J.F.; Dinan, T.G. N-3 Polyunsaturated Fatty Acids through the Lifespan: Implication for Psychopathology. Int. J. Neuropsychopharmacol. 2016, 19. [Google Scholar] [CrossRef] [PubMed]
- Kuczmarski, R.J.; Ogden, C.L.; Guo, S.S.; Grummer-Strawn, L.M.; Flegal, K.M.; Mei, Z.; Wei, R.; Curtin, L.R.; Roche, A.F.; Johnson, C.L. CDC Growth Charts for the United States: Methods and development. Vital Health Stat. 2002, 246, 147–148. [Google Scholar]
- Domingo-Salvany, A.; Regidor, E.; Alonso, J.; Alvarez-Dardet, C. Proposal for a social class measure. Working Group of the Spanish Society of Epidemiology and the Spanish Society of Family and Community Medicine. Aten Primaria 2000, 25, 350. [Google Scholar] [PubMed]
- Martin-Moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-Rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and Validation of a Food Frequency Questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef]
- Le Moullec, N.; Deheeger, M.; Preziosi, P.; Monteiro, P.; Valeix, P.; Rolland-Cachera, M.F.; Potier De Courcy, G.; Christides, J.P.; Cherouvrier, F.; Galan, P.; et al. Validation du manuel-photos utilisé pour l’enquête alimentaire de l’étude SU. VI. MAX. Cahiers de Nutrition et de Diététique 1996, 31, 158–164. [Google Scholar]
- Moreiras, O.; Carbajal, A.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos (Food Composition Tables); Ediciones Piramide: Madrid, Spain, 2005. [Google Scholar]
- Fernandez-Ballart, J.D.; Piñol, J.L.; Zazpe, I.; Corella, D.; Carrasco, P.; Toledo, E.; Perez-Bauer, M.; Martínez-González, M.Á.; Salas-Salvadó, J.; Martín-Moreno, J.M. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br. J. Nutr. 2010, 103, 1808–1816. [Google Scholar] [CrossRef]
- Hepburn, F.N.; Exler, J.; Weihrauch, J.L. Provisional tables on the content of omega-3 fatty acids and other fat components of selected foods. J. Am. Diet. Assoc. 1986, 86, 788–793. [Google Scholar]
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1228S. [Google Scholar] [CrossRef]
- Willcutt, E.G. The Prevalence of DSM-IV Attention-Deficit/Hyperactivity Disorder: A Meta-Analytic Review. Neurotherapeutics 2012, 9, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Siró, I.; Kápolna, E.; Lugasi, A. Functional food. Product development, marketing and consumer acceptance. A review. Appetite 2008, 51, 456–467. [Google Scholar] [CrossRef] [PubMed]
- SENC, Sociedad Española de Nutrición Comunitaria. Objetivos nutricionales para la población española. Rev. Esp. Nutr. Comunitaria 2011, 4, 178–199. [Google Scholar]
- Kris-Etherton, P.; Taylor, D.S.; Yu-Poth, S.; Huth, P.; Moriarty, K.; Fishell, V.; Hargrove, R.L.; Zhao, G.; Etherton, T.D. Polyunsaturated fatty acids in the food chain in the United States. Am. J. Clin. Nutr. 2000, 71, 179S–188S. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.J.; Yu, Y.H.; Fu, M.L.; Yeh, W.T.; Hsu, J.L.; Yang, Y.H.; Yang, H.T.; Huang, S.Y.; Wei, I.L.; Chen, W.J.; et al. Dietary Profiles, Nutritional Biochemistry Status, and Attention-Deficit/Hyperactivity Disorder: Path Analysis for a Case-Control Study. J. Clin. Med. 2019, 8, 709. [Google Scholar] [CrossRef]
- FAO/FINUT. Grasas y ácidos grasos en Nutrición Humana. Available online: www.fao.org/3/i1953s/i1953s.pdf (accessed on 10 March 2019).
- Hawkey, E.; Nigg, J.T. Omega-3 fatty acid and ADHD, blood level analysis and meta-analytic extension of suplementation trials. Clin. Psychol. Rev. 2014, 34, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Guesnet, P.; Tressou, J.; Buaud, B.; Simon, N.; Pasteau, S. Inadequate daily intakes of n-3 polyunsaturated fatty acids (PUFA) in the general French population of children (3–10 years), the INCA2 survey. Eur. J. Nutr. 2019, 58, 895–903. [Google Scholar] [CrossRef]
- Marí-Bauset, S.; Llopis-González, A.; Zazpe-García, I.; Marí-Sanchis, A.; Morales-Suárez-Varela, M. Nutritional status of children with autism spectrum disorders (ASDs), a case control study. J. Autism. Dev. Disord. 2015, 45, 203–212. [Google Scholar]
- Bowling, A.B.; Tiemeier, H.W.; Jaddoe, V.W.V.; Barker, E.D.; Jansen, P.W. ADHD symptoms and body composition changes in childhood: A longitudinal study evaluating directionality of associations. Pediatr. Obes. 2018, 13, 567–575. [Google Scholar] [CrossRef]
- Hosomi, R.; Yoshida, M.; Fukunaga, K. Seafood Consumption and Components for Health. J. Heal. Sci. 2012, 4, 72–86. [Google Scholar] [CrossRef]
- Ouellet, V.; Weisnagel, S.J.; Marois, J.; Bergeron, J.; Julien, P.; Gougeon, R.; Tchernof, A.; Holub, B.J.; Jacques, H. Dietary Cod Protein Reduces Plasma C-Reactive Protein in Insulin-Resistant Men and Women. J. Nutr. 2008, 138, 2386–2391. [Google Scholar] [CrossRef]
- Ouellet, V.; Marois, J.; Weisnagel, S.J.; Jacques, H. Dietary cod protein improves insulin sensitivity in insulin-resistant men and women: A randomized controlled trial. Diabetes Care 2007, 30, 2816–2821. [Google Scholar] [CrossRef] [PubMed]
- Jerlich, A.; Fritz, G.; Kharrazi, H.; Hammel, M.; Tschabuschnig, S.; Glatter, O.; Schaur, R. Comparison of HOCl traps with myeloperoxidase inhibitors in prevention of low density lipoprotein oxidation. Biochim. Biophys. Acta 2000, 1481, 109–118. [Google Scholar] [CrossRef]
- Karppi; Rissanen; Nyyssönen; Kaikkonen; Olsson; Voutilainen; Salonen; Karppi, J.; Rissanen, T.H.; Nyyssönen, K.; et al. Effects of Astaxanthin Supplementation on Lipid Peroxidation. Int. J. Vitam. Nutr. 2007, 77, 3–11. [Google Scholar] [CrossRef]
- Yoshida, H.; Yanai, H.; Ito, K.; Tomono, Y.; Koikeda, T.; Tsukahara, H.; Tada, N. Administration of natural astaxanthin increases serum HDL-cholesterol and adiponectin in subjects with mild hyperlipidemia. Atherosclerosis 2010, 209, 520–523. [Google Scholar] [CrossRef]
- Chung, S.Y.; Moriyama, T.; Uezu, E.; Uezu, K.; Hirata, R.; Yohena, N.; Masuda, Y.; Kokubu, T.; Yamamoto, S. Administration of phosphatidylcholine increases brain acetylcholine concentration and improves memory in mice with dementia. J. Nutr. 1995, 125, 1484–1489. [Google Scholar] [PubMed]
- Cortese, S.; Moreira-Maia, C.R.; Fleur, D.S.; Morcillo-Peñalver, C.; Rohde, L.A.; Faraone, S.V. Association Between ADHD and Obesity: A Systematic Review and Meta-Analysis. Am. J. Psychiatry 2016, 173, 34–43. [Google Scholar] [CrossRef]
- Nigg, J.T.; Johnstone, J.M.; Musser, E.D.; Long, H.G.; Willoughby, M.T.; Shannon, J. Attention-deficit/hyperactivity disorder (ADHD) and being overweight/obesity, new data and meta-analysis. Clin. Psychol. Rev. 2016, 43, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Bonaventura, I.; Granero, R.; Ezpeleta, L. The relationship between weight status and emotional and behavioral problems in Spanish preschool children. J. Pediatr. Psychol. 2015, 40, 455–463. [Google Scholar] [CrossRef]
- Bass, J.L.; Corwin, M.; Gozal, D.; Moore, C.; Nishida, H.; Parker, S.; Schonwald, A.; Wilker, R.E.; Stehle, S.; Kinane, T.B. The effect of chronic or intermittent hypoxia on cognition in childhood: A review of the evidence. Pediatrics 2004, 114, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Albayrak, Ö.; Pütter, C.; Volckmar, A.L.; Cichon, S.; Hoffmann, P.; Nöthen, M.M.; Jöckel, K.H.; Schreiber, S.; Wichmann, H.E.; Faraone, S.V.; et al. Common obesity risk alleles in childhood attention-deficit/hyperactivity disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2013, 162, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.L.; Li, B.M.; Yang, J.; Wang, Y.W. Does dopaminergic reward system contribute to explaining comorbidity obesity and ADHD? Med. Hypotheses. 2008, 70, 1118–1120. [Google Scholar] [CrossRef] [PubMed]
- Ghanadri, Y.; Eisenberg, I.; Ben Neriah, Z.; Agranat-Meged, A.; Kieselstein-Gross, E.; Mitrani-Rosenbaum, S.; Agranat-Meged, A.; Kieselstein-Gross, E.; Mitrani-Rosenbaum, S. Attention deficit hyperactivity disorder in obese melanocortin-4-receptor (MC4R) deficient subjects: A newly described expression of MC4R deficiency. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2008, 147, 1547–1553. [Google Scholar]
- Meyer, B.J.; Mann, N.J.; Lewis, J.L.; Milligan, G.C.; Sinclair, A.J.; Howe, P.R.C. Dietary intakes and food sources of omega-6 and omega-3 polyunsaturated fatty acids. Lipids 2003, 38, 391–398. [Google Scholar] [CrossRef]
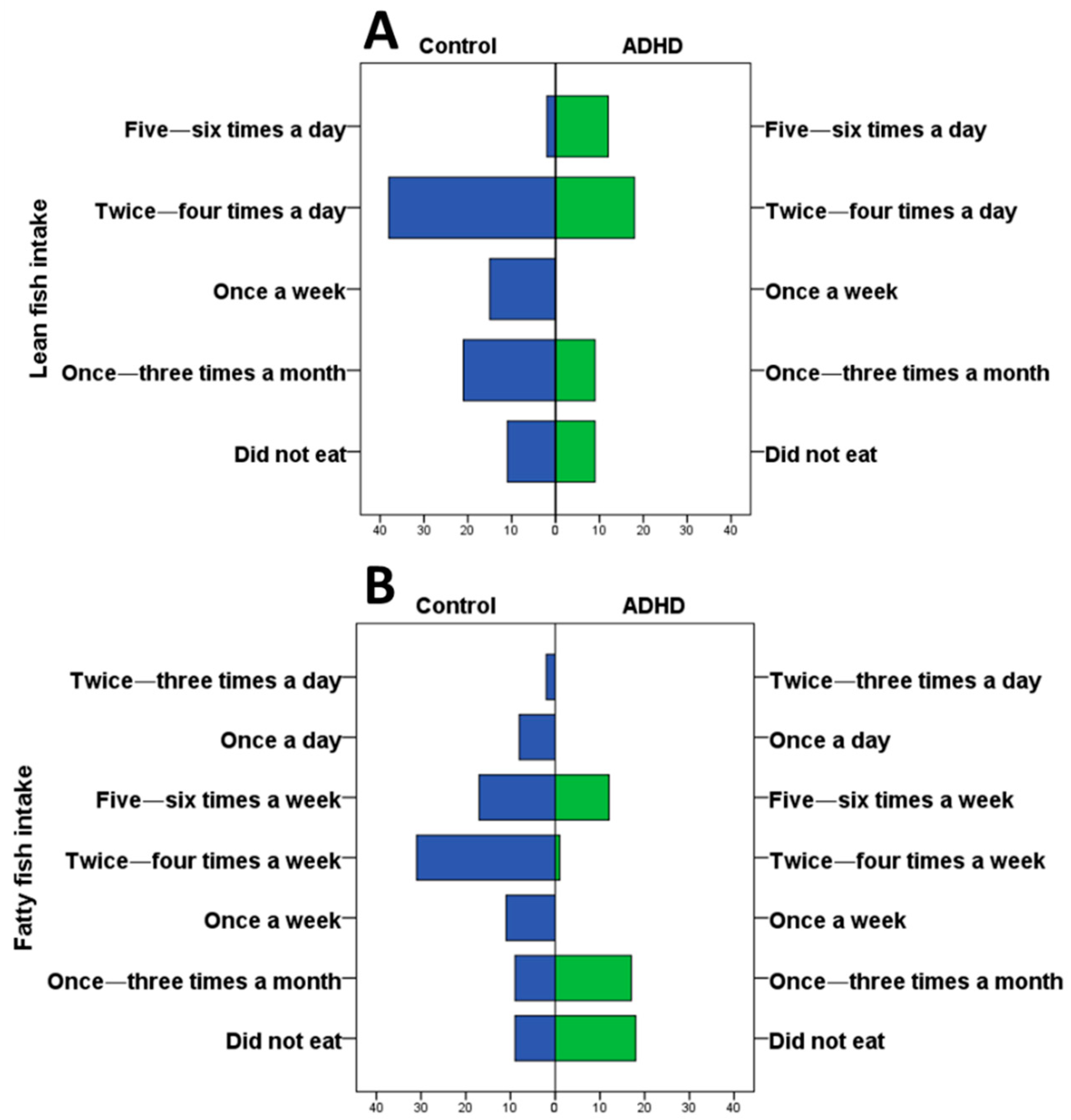
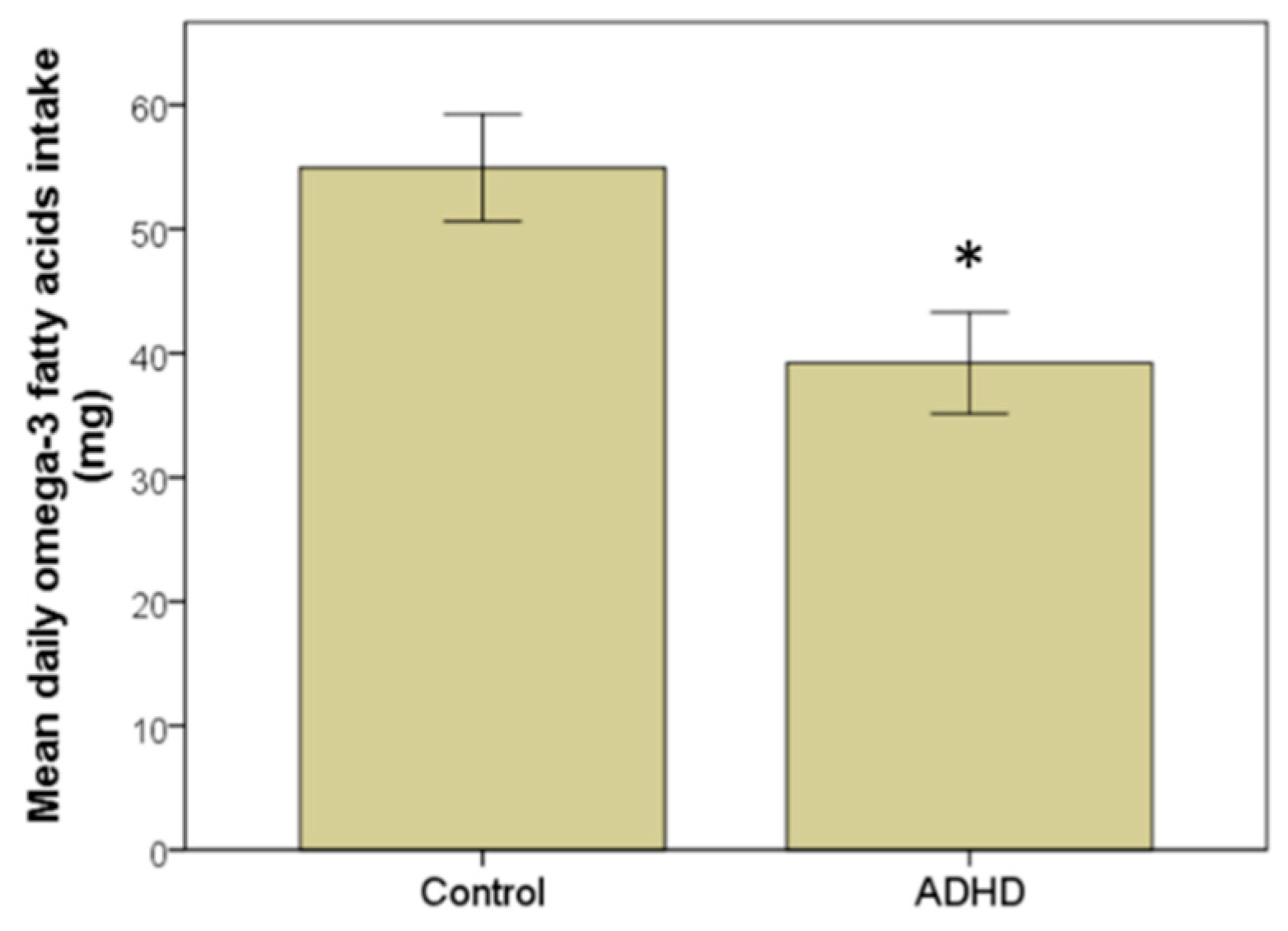
| Variable | Control | ADHD | p-Value |
|---|---|---|---|
| Age | 10.00 ± 0.27 (range 4–13) | 9.54 ± 0.31 (5–14) | p = 0.86 (Mann–Whitney test) |
| Sex | Female n = 25 Male n = 62 | Female n = 12 Male n = 36 | p = 0.64 (Chi-squared test) |
| BMI | 18.69 ± 0.39 (range 10.65–30.44) | 20.89 ± 0.44 (range 15.50–28.31) | p = 0.04 (Mann–Whitney test) |
| Low weight | 26.4% | 4.2% | p < 0.001 (Chi-squared test) |
| Normal weight | 36.8% | 35.4% | |
| Over weight | 25.3% | 20.8% | |
| Obesity | 11.5% | 39.6% | |
| Social class | Higher: 26.4% | Higher: 31.3% | p = 0.88 (Chi-squared test) |
| Medium: 55.2% | Medium: 52.1% | ||
| Lower: 18.4 | Lower: 16.6% | ||
| Employment situation | Father Employed: 97.7% Unemployed: 2.3% | Father Employed: 95.8% Unemployed: 4.2% | p = 0.95 (Chi-squared test) |
| Mother Employed: 50.6% Unemployed: 16.1% Homemaker: 33.3% | Mother Employed: 58.3% Unemployed: 10.4% Homemaker: 31.3% | p = 0.78 (Chi-squared test) | |
| Educational level | Father Primary school: 23.0% Secondary school: 54% University: 23.0% | Father Primary school: 20.8% Secondary school: 54.2 University: 25.0% | p = 0.89 (Chi-squared test) |
| Mother Primary school: 17.2% Secondary school: 56.4% University: 26.4% | Mother Primary school: 12.5% Secondary school: 56.2% University: 31.3% | p = 0.84 (Chi-squared test) |
| Group | Lean Fish (mg/day) | Fatty Fish (mg/day) | Mollusks (mg/day) | Crustaceans (mg/day) | Other Types (mg/day) |
|---|---|---|---|---|---|
| Control | 45.56 ± 19.81 | 40,63 ± 33.6 | 18.28 ± 18.20 | 3.21 ± 6.22 | 2.20 ± 5.11 |
| ADHD | 38.51 ± 19.22 * | 26.42 ± 20.30 ** | 10.21 ± 15.4 * | 3.0 ± 6.43 | 0.29 ± 2.62 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuentes-Albero, M.; Martínez-Martínez, M.I.; Cauli, O. Omega-3 Long-Chain Polyunsaturated Fatty Acids Intake in Children with Attention Deficit and Hyperactivity Disorder. Brain Sci. 2019, 9, 120. https://doi.org/10.3390/brainsci9050120
Fuentes-Albero M, Martínez-Martínez MI, Cauli O. Omega-3 Long-Chain Polyunsaturated Fatty Acids Intake in Children with Attention Deficit and Hyperactivity Disorder. Brain Sciences. 2019; 9(5):120. https://doi.org/10.3390/brainsci9050120
Chicago/Turabian StyleFuentes-Albero, Milagros, María Isabel Martínez-Martínez, and Omar Cauli. 2019. "Omega-3 Long-Chain Polyunsaturated Fatty Acids Intake in Children with Attention Deficit and Hyperactivity Disorder" Brain Sciences 9, no. 5: 120. https://doi.org/10.3390/brainsci9050120
APA StyleFuentes-Albero, M., Martínez-Martínez, M. I., & Cauli, O. (2019). Omega-3 Long-Chain Polyunsaturated Fatty Acids Intake in Children with Attention Deficit and Hyperactivity Disorder. Brain Sciences, 9(5), 120. https://doi.org/10.3390/brainsci9050120






