Management of Neuroinflammatory Responses to AAV-Mediated Gene Therapies for Neurodegenerative Diseases
Abstract
1. Introduction
2. Neuroinflammation in Adeno-Associated Virus (AAV)-Mediated Gene Therapies
2.1. Systemic Delivery
2.2. Central Nervous System (CNS)-Directed Delivery
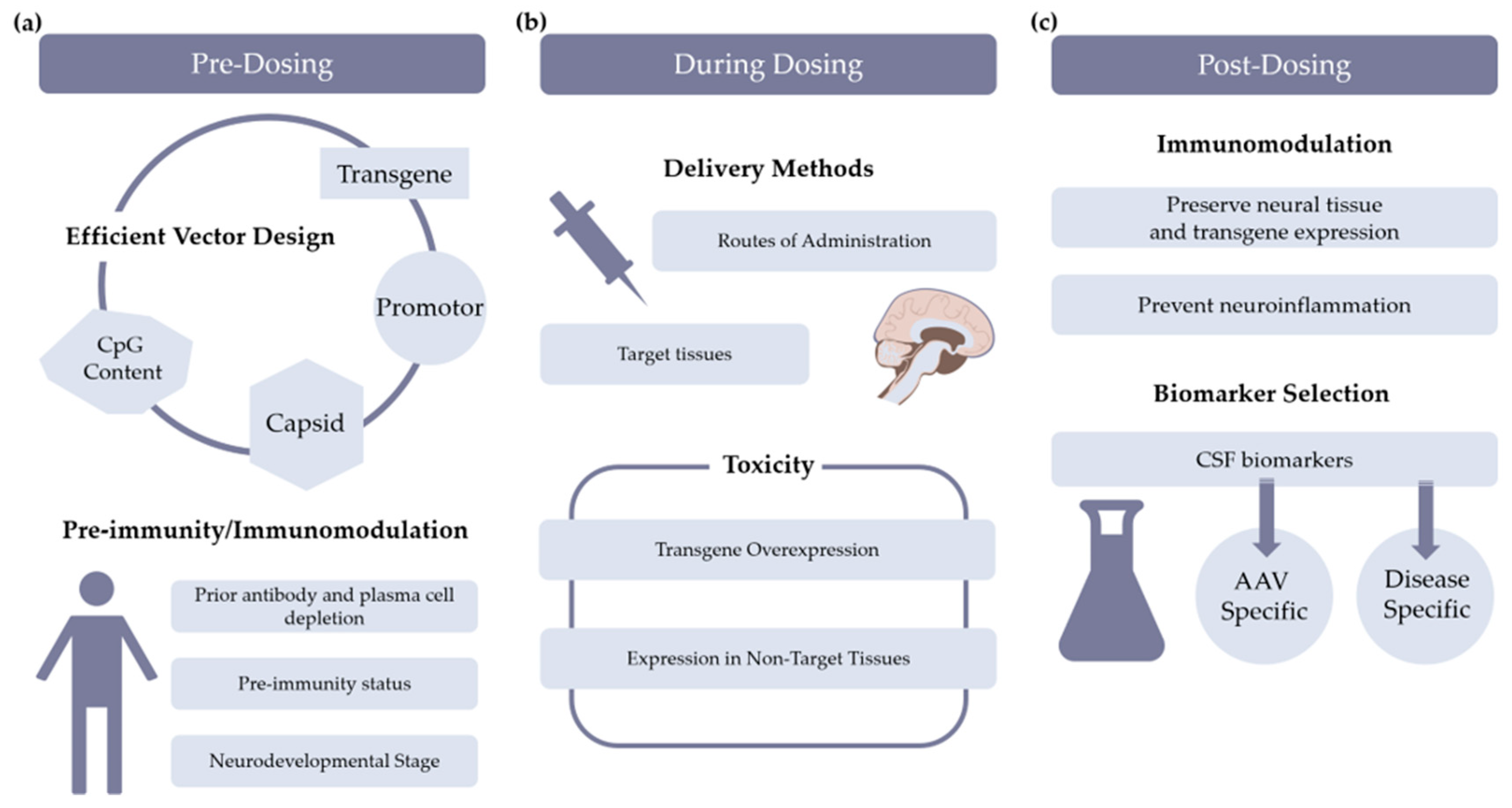
3. Managing AAV-Mediated Neuroinflammation
3.1. Choice of AAV Capsid Serotype and Promoter
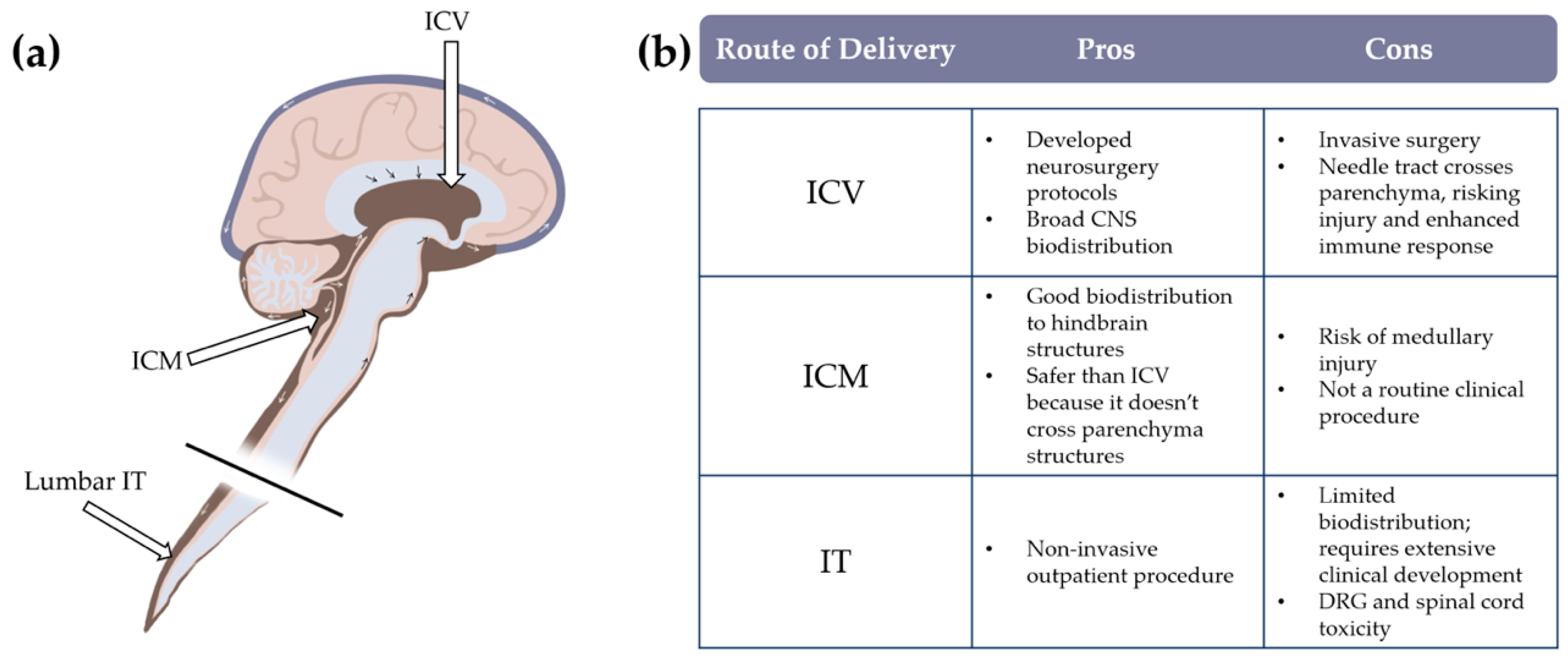
3.2. Route of Administration
3.3. Genetic Manipulations to Decrease TLR9-Mediated Immune Responses
3.4. Immunomodulation Strategies and Their Effect on CNS
4. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Corti, M.; Norman, S.; Coleman, K.; Liberati, C.; Elder, M.; Escolar, M.; Clement, N.; Clever, B.; Byrne, B.; Gao, G. Immune Blockade in CNS Gene Therapy Improves Safety and Clinical Outcome; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2018; Volume 26, p. 32. [Google Scholar]
- Nair, J.; Freeman, D., Jr.; Davis, I.; McParland, T.; Pope, M.; Perez, B.; Rodriguez-Lebron, E.; Coleman, K.; Subramony, S.; Byrne, B.; et al. AAV9 Biodistribution with Different Routes of CNS Administration in Rodents; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2019; Volume 27, p. 259. [Google Scholar]
- Perez, B.A.; Coleman, K.E.; Wichman, M.B.; Futch, K.N.; Mandolini, K.E.; Cravey, L.D.; Nair, J.; Cloutier, D.A.; Lynch, D.; Byrne, B.J.; et al. Management of Preexisting Immunity to AAV9 in Friedreich’s Ataxia; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2019; Volume 27, p. 140. [Google Scholar]
- Perdomini, M.; Belbellaa, B.; Monassier, L.; Reutenauer, L.; Messaddeq, N.; Cartier, N.; Crystal, R.G.; Aubourg, P.; Puccio, H. Prevention and reversal of severe mitochondrial cardiomyopathy by gene therapy in a mouse model of Friedreich’s ataxia. Nat. Med. 2014, 20, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Mendell, J.R.; Al-Zaidy, S.; Shell, R.; Arnold, W.D.; Rodino-Klapac, L.R.; Prior, T.W.; Lowes, L.; Alfano, L.; Berry, K.; Church, K.; et al. Single-Dose Gene-Replacement Therapy for Spinal Muscular Atrophy. N. Engl. J. Med. 2017, 377, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, V.; Twisk, J.; Kwikkers, K.; Aronica, E.; Brisson, D.; Methot, J.; Petry, H.; Gaudet, D. Immune responses to intramuscular administration of alipogene tiparvovec (AAV1-LPL(S447X)) in a phase II clinical trial of lipoprotein lipase deficiency gene therapy. Hum. Gene Ther. 2014, 25, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Manno, C.S.; Pierce, G.F.; Arruda, V.R.; Glader, B.; Ragni, M.; Rasko, J.J.; Rasko, J.; Ozelo, M.C.; Hoots, K.; Blatt, P.; et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat. Med. 2006, 12, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Ertl, H.C.J.; High, K.A. Impact of AAV Capsid-Specific T-Cell Responses on Design and Outcome of Clinical Gene Transfer Trials with Recombinant Adeno-Associated Viral Vectors: An Evolving Controversy. Hum. Gene Ther. 2017, 28, 328–337. [Google Scholar] [CrossRef] [PubMed]
- McPhee, S.W.; Janson, C.G.; Li, C.; Samulski, R.J.; Camp, A.S.; Francis, J.; Shera, D.; Lioutermann, L.; Feely, M.; Freese, A.; et al. Immune responses to AAV in a phase I study for Canavan disease. J. Gene Med. 2006, 8, 577–588. [Google Scholar] [CrossRef]
- Faust, S.M.; Bell, P.; Cutler, B.J.; Ashley, S.N.; Zhu, Y.; Rabinowitz, J.E.; Wilson, J.M. CpG-depleted adeno-associated virus vectors evade immune detection. J. Clin. Investig. 2013, 123, 2994–3001. [Google Scholar] [CrossRef]
- Rabinowitz, J.; Chan, Y.K.; Samulski, R.J. Adeno-associated Virus (AAV) versus Immune Response. Viruses 2019, 11, 102. [Google Scholar] [CrossRef]
- Letizia, A.; Chan, Y.K.; Graveline, A.; Church, G. Engineering AAV Vectors to Reduce CD8+ T Cell Responses Following Intramuscular Injection in Mice; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2019; Volume 27, p. 301. [Google Scholar]
- Chan, Y.K.; Wang, S.; Letizia, A.; Chan, Y.; Lim, E.; Graveline, A.; Verdera, H.C.; Alphonse, P.; Xue, Y.; Chiang, J.; et al. Reducing AAV-Mediated Immune Responses and Pathology in a Subretinal Pig Model by Engineering the Vector Genome; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2019; Volume 27, p. 298. [Google Scholar]
- Clément, N.; Grieger, J.C. Manufacturing of recombinant adeno-associated viral vectors for clinical trials. Mol. Ther. Methods Clin. Dev. 2016, 3, 16002. [Google Scholar] [CrossRef]
- Corti, M.; Elder, M.; Falk, D.; Lawson, L.; Smith, B.; Nayak, S.; Conlon, T.; Clément, N.; Erger, K.; Lavassani, E.; et al. B-Cell Depletion is Protective Against Anti-AAV Capsid Immune Response: A Human Subject Case Study. Mol. Ther. Methods Clin. Dev. 2014, 1, 14033. [Google Scholar] [CrossRef]
- Corti, M.; Liberati, C.; Smith, B.K.; Lawson, L.A.; Tuna, I.S.; Conlon, T.J.; Coleman, K.E.; Islam, S.; Herzog, R.W.; Fuller, D.D.; et al. Safety of Intradiaphragmatic Delivery of Adeno-Associated Virus-Mediated Alpha-Glucosidase (rAAV1-CMV-hGAA) Gene Therapy in Children Affected by Pompe Disease. Hum. Gene Ther. Clin. Dev. 2017, 28, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Mingozzi, F.; High, K.A. Overcoming the Host Immune Response to Adeno-Associated Virus Gene Delivery Vectors: The Race Between Clearance, Tolerance, Neutralization, and Escape. Annu. Rev. Virol. 2017, 4, 511–534. [Google Scholar] [CrossRef] [PubMed]
- Mingozzi, F.; High, K.A. Immune responses to AAV vectors: Overcoming barriers to successful gene therapy. Blood 2013, 122, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Doerfler, P.A.; Nayak, S.; Corti, M.; Morel, L.; Herzog, R.W.; Byrne, B.J. Targeted approaches to induce immune tolerance for Pompe disease therapy. Mol. Ther. Methods Clin. Dev. 2016, 3, 15053. [Google Scholar] [CrossRef] [PubMed]
- Hordeaux, J.; Hinderer, C.; Goode, T.; Katz, N.; Buza, E.L.; Bell, P.; Calcedo, R.; Richman, L.K.; Wilson, J.M. Toxicology Study of Intra-Cisterna Magna Adeno-Associated Virus 9 Expressing Human Alpha-L-Iduronidase in Rhesus Macaques. Mol. Ther. Methods Clin. Dev. 2018, 10, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Hordeaux, J.; Hinderer, C.; Goode, T.; Buza, E.L.; Bell, P.; Calcedo, R.; Richman, L.K.; Wilson, J.M. Toxicology Study of Intra-Cisterna Magna Adeno-Associated Virus 9 Expressing Iduronate-2-Sulfatase in Rhesus Macaques. Mol. Ther. Methods Clin. Dev. 2018, 10, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Muldoon, L.L.; Alvarez, J.I.; Begley, D.J.; Boado, R.J.; Del Zoppo, G.J.; Doolittle, N.D.; Engelhardt, B.; Hallenbeck, J.M.; Lonser, R.R.; Ohlfest, J.R.; et al. Immunologic privilege in the central nervous system and the blood-brain barrier. J. Cereb. Blood Flow Metab. 2013, 33, 13–21. [Google Scholar] [CrossRef]
- Lowenstein, P.R.; Mandel, R.J.; Xiong, W.D.; Kroeger, K.; Castro, M.G. Immune responses to adenovirus and adeno-associated vectors used for gene therapy of brain diseases: The role of immunological synapses in understanding the cell biology of neuroimmune interactions. Curr. Gene Ther. 2007, 7, 347–360. [Google Scholar] [CrossRef]
- Ransohoff, R.M.; Brown, M.A. Innate immunity in the central nervous system. J. Clin. Investig. 2012, 122, 1164–1171. [Google Scholar] [CrossRef]
- Golebiowski, D.; van der Bom, I.M.J.; Kwon, C.S.; Miller, A.D.; Petrosky, K.; Bradbury, A.M.; Maitland, S.; Kühn, A.L.; Bishop, N.; Curran, E.; et al. Direct Intracranial Injection of AAVrh8 Encoding Monkey β-N-Acetylhexosaminidase Causes Neurotoxicity in the Primate Brain. Hum. Gene Ther. 2017, 28, 510–522. [Google Scholar] [CrossRef]
- Novartis Announces AVXS-101 Intrathecal Study Update. Available online: https://www.novartis.com/news/media-releases/novartis-announces-avxs-101-intrathecal-study-update (accessed on 28 January 2020).
- Ransohoff, R.M. How neuroinflammation contributes to neurodegeneration. Science 2016, 353, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Ransohoff, R.M.; Schafer, D.; Vincent, A.; Blachère, N.E.; Bar-Or, A. Neuroinflammation: Ways in Which the Immune System Affects the Brain. Neurotherapeutics 2015, 12, 896–909. [Google Scholar] [CrossRef] [PubMed]
- Ransohoff, R.M.; Cardona, A.E. The myeloid cells of the central nervous system parenchyma. Nature 2010, 468, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Saade, D.; Bhurucha-Goebel, D.; Foley, A.R.; Neuhaus, S.B.; Soldatos, A.; Hu, Y.; Hewitt, M.; Calcedo del Hoyo, R.; Jacobson, S.; Liebovitch, E.; et al. Review of CSF and Peripheral Immune Responses Following Intrathecal Gene Transfer for Giant Axonal Neuropathy; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2018; Volume 26, pp. 456–457. [Google Scholar]
- Bharucha-Goebel, D.; Saade, D.; Soldatos, A.; Kang, E.; Del Calcedo, R.; Hu, Y.; Liebovitch, E.; Jacobson, S.; Rybin, D.; Foley, A.R.; et al. Immune Analysis Following Intrathecal Gene Transfer: 3-Year Data from Clinical Intrathecal Gene Transfer Trial for Patients with Giant Axonal Neuropathy; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2019; Volume 27, pp. 298–299. [Google Scholar]
- Stephenson, J.; Nutma, E.; van der Valk, P.; Amor, S. Inflammation in CNS neurodegenerative diseases. Immunology 2018, 154, 204–219. [Google Scholar] [CrossRef]
- Lepore, S.; Waiczies, H.; Hentschel, J.; Ji, Y.; Skodowski, J.; Pohlmann, A.; Millward, J.M.; Paul, F.; Wuerfel, J.; Niendorf, T.; et al. Enlargement of cerebral ventricles as an early indicator of encephalomyelitis. PLoS ONE 2013, 8, e72841. [Google Scholar] [CrossRef]
- Ku, M.C.; Waiczies, S.; Niendorf, T.; Pohlmann, A. Assessment of Blood Brain Barrier Leakage with Gadolinium-Enhanced MRI. Methods Mol. Biol. 2018, 1718, 395–408. [Google Scholar] [CrossRef]
- Pascual, B.; Prieto, E.; Arbizu, J.; Marti-Climent, J.M.; Peñuelas, I.; Quincoces, G.; Zarauza, R.; Pappatà, S.; Masdeu, J.C. Decreased carbon-11-flumazenil binding in early Alzheimer’s disease. Brain 2012, 135, 2817–2825. [Google Scholar] [CrossRef][Green Version]
- Edison, P.; Donat, C.K.; Sastre, M. Imaging of Glial Activation in Alzheimer’s Disease. Front. Neurol. 2018, 9, 625. [Google Scholar] [CrossRef]
- Woodcock, T.; Morganti-Kossmann, M.C. The role of markers of inflammation in traumatic brain injury. Front. Neurol. 2013, 4, 18. [Google Scholar] [CrossRef]
- Fitzpatrick, Z.; Leborgne, C.; Barbon, E.; Masat, E.; Ronzitti, G.; van Wittenberghe, L.; Vignaud, A.; Collaud, F.; Charles, S.; Simon Sola, M.; et al. Influence of Pre-existing Anti-capsid Neutralizing and Binding Antibodies on AAV Vector Transduction. Mol. Ther. Methods Clin. Dev. 2018, 9, 119–129. [Google Scholar] [CrossRef]
- Peden, C.S.; Burger, C.; Muzyczka, N.; Mandel, R.J. Circulating anti-wild-type adeno-associated virus type 2 (AAV2) antibodies inhibit recombinant AAV2 (rAAV2)-mediated, but not rAAV5-mediated, gene transfer in the brain. J. Virol. 2004, 78, 6344–6359. [Google Scholar] [CrossRef] [PubMed]
- Elder, M.E.; Nayak, S.; Collins, S.W.; Lawson, L.A.; Kelley, J.S.; Herzog, R.W.; Modica, R.F.; Lew, J.; Lawrence, R.M.; Byrne, B.J. B-Cell depletion and immunomodulation before initiation of enzyme replacement therapy blocks the immune response to acid alpha-glucosidase in infantile-onset Pompe disease. J. Pediatr. 2013, 163, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Murrey, D.A.; Naughton, B.J.; Duncan, F.J.; Meadows, A.S.; Ware, T.A.; Campbell, K.J.; Bremer, W.G.; Walker, C.M.; Goodchild, L.; Bolon, B.; et al. Feasibility and safety of systemic rAAV9-hNAGLU delivery for treating mucopolysaccharidosis IIIB: Toxicology, biodistribution, and immunological assessments in primates. Hum. Gene Ther. Clin. Dev. 2014, 25, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Foust, K.D.; Wang, X.; McGovern, V.L.; Braun, L.; Bevan, A.K.; Haidet, A.M.; Le, T.T.; Morales, P.R.; Rich, M.M.; Burghes, A.H.; et al. Rescue of the spinal muscular atrophy phenotype in a mouse model by early postnatal delivery of SMN. Nat. Biotechnol. 2010, 28, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Zolgensma [package insert]; AveXis Inc.: Bannockburn, IL, USA, 2019.
- Kruzik, A.; Fetahagic, D.; Hartlieb, B.; Dorn, S.; Koppensteiner, H.; Horling, F.M.; Scheiflinger, F.; Reipert, B.M.; de la Rosa, M. Prevalence of Anti-Adeno-Associated Virus Immune Responses in International Cohorts of Healthy Donors. Mol. Ther. Methods Clin. Dev. 2019, 14, 126–133. [Google Scholar] [CrossRef]
- Kuntz, N.; Shieh, P.B.; Smith, B.; Bonnemann, C.G.; Dowling, J.J.; Lawson, M.W.; Muller-Felber, W.; Noursalehi, M.; Rico, S.; Servais, L.; et al. Aspiro Phase 1/2 Gene Therapy Trial in X-Linked Myotubular Myopathy (Xlmtm): Preliminary Safety and Efficacy Findings; Molecular Therapy; Cell Press: Cambridge, MA, USA, 2018; Volume 26, p. 4. [Google Scholar]
- Pfizer Presents Initial Clinical Data on Phase 1B Gene Therapy Study for Duchenne Muscular Dystrophy (DMD). Available online: https://investors.pfizer.com/investor-news/press-release-details/2019/Pfizer-Presents-Initial-Clinical-Data-on-Phase-1b-Gene-Therapy-Study-for-Duchenne-Muscular-Dystrophy-DMD/default.aspx (accessed on 28 January 2020).
- Solid Biosciences Provides Data Update from SGT-001 Development Plan. Available online: https://investors.solidbio.com/news-releases/news-release-details/solid-biosciences-provides-data-update-sgt-001-development (accessed on 28 January 2020).
- Samaranch, L.; Salegio, E.A.; San Sebastian, W.; Kells, A.P.; Foust, K.D.; Bringas, J.R.; Lamarre, C.; Forsayeth, J.; Kaspar, B.K.; Bankiewicz, K.S. Adeno-associated virus serotype 9 transduction in the central nervous system of nonhuman primates. Hum. Gene Ther. 2012, 23, 382–389. [Google Scholar] [CrossRef]
- Ciron, C.; Desmaris, N.; Colle, M.A.; Raoul, S.; Joussemet, B.; Vérot, L.; Ausseil, J.; Froissart, R.; Roux, F.; Chérel, Y.; et al. Gene therapy of the brain in the dog model of Hurler’s syndrome. Ann. Neurol. 2006, 60, 204–213. [Google Scholar] [CrossRef]
- Ellinwood, N.M.; Ausseil, J.; Desmaris, N.; Bigou, S.; Liu, S.; Jens, J.K.; Snella, E.M.; Mohammed, E.E.; Thomson, C.B.; Raoul, S.; et al. Safe, efficient, and reproducible gene therapy of the brain in the dog models of Sanfilippo and Hurler syndromes. Mol. Ther. 2011, 19, 251–259. [Google Scholar] [CrossRef]
- Meyer, K.; Ferraiuolo, L.; Schmelzer, L.; Braun, L.; McGovern, V.; Likhite, S.; Michels, O.; Govoni, A.; Fitzgerald, J.; Morales, P.; et al. Improving single injection CSF delivery of AAV9-mediated gene therapy for SMA: A dose-response study in mice and nonhuman primates. Mol. Ther. 2015, 23, 477–487. [Google Scholar] [CrossRef]
- Ribera, A.; Haurigot, V.; Garcia, M.; Marcó, S.; Motas, S.; Villacampa, P.; Maggioni, L.; León, X.; Molas, M.; Sánchez, V.; et al. Biochemical, histological and functional correction of mucopolysaccharidosis type IIIB by intra-cerebrospinal fluid gene therapy. Hum. Mol. Genet. 2015, 24, 2078–2095. [Google Scholar] [CrossRef]
- Hinderer, C.; Bell, P.; Gurda, B.L.; Wang, Q.; Louboutin, J.P.; Zhu, Y.; Bagel, J.; O’Donnell, P.; Sikora, T.; Ruane, T.; et al. Intrathecal gene therapy corrects CNS pathology in a feline model of mucopolysaccharidosis I. Mol. Ther. 2014, 22, 2018–2027. [Google Scholar] [CrossRef] [PubMed]
- Davidson, B.L.; Stein, C.S.; Heth, J.A.; Martins, I.; Kotin, R.M.; Derksen, T.A.; Zabner, J.; Ghodsi, A.; Chiorini, J.A. Recombinant adeno-associated virus type 2, 4, and 5 vectors: Transduction of variant cell types and regions in the mammalian central nervous system. Proc. Natl. Acad. Sci. USA 2000, 97, 3428–3432. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.P.; Alvira, M.R.; Wang, L.; Calcedo, R.; Johnston, J.; Wilson, J.M. Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc. Natl. Acad. Sci. USA 2002, 99, 11854–11859. [Google Scholar] [CrossRef]
- Petrosyan, H.A.; Alessi, V.; Singh, V.; Hunanyan, A.S.; Levine, J.M.; Arvanian, V.L. Transduction efficiency of neurons and glial cells by AAV-1, -5, -9, -rh10 and -hu11 serotypes in rat spinal cord following contusion injury. Gene Ther. 2014, 21, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Cearley, C.N.; Wolfe, J.H. Transduction characteristics of adeno-associated virus vectors expressing cap serotypes 7, 8, 9, and Rh10 in the mouse brain. Mol. Ther. 2006, 13, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Schuster, D.J.; Dykstra, J.A.; Riedl, M.S.; Kitto, K.F.; Belur, L.R.; McIvor, R.S.; Elde, R.P.; Fairbanks, C.A.; Vulchanova, L. Biodistribution of adeno-associated virus serotype 9 (AAV9) vector after intrathecal and intravenous delivery in mouse. Front. Neuroanat. 2014, 8, 42. [Google Scholar] [CrossRef]
- Murlidharan, G.; Samulski, R.J.; Asokan, A. Biology of adeno-associated viral vectors in the central nervous system. Front. Mol. Neurosci. 2014, 7, 76. [Google Scholar] [CrossRef]
- Rosario, A.M.; Cruz, P.E.; Ceballos-Diaz, C.; Strickland, M.R.; Siemienski, Z.; Pardo, M.; Schob, K.L.; Li, A.; Aslanidi, G.V.; Srivastava, A.; et al. Microglia-specific targeting by novel capsid-modified AAV6 vectors. Mol. Ther. Methods Clin. Dev. 2016, 3, 16026. [Google Scholar] [CrossRef]
- Foust, K.D.; Nurre, E.; Montgomery, C.L.; Hernandez, A.; Chan, C.M.; Kaspar, B.K. Intravascular AAV9 preferentially targets neonatal neurons and adult astrocytes. Nat. Biotechnol. 2009, 27, 59–65. [Google Scholar] [CrossRef]
- Duque, S.; Joussemet, B.; Riviere, C.; Marais, T.; Dubreil, L.; Douar, A.M.; Fyfe, J.; Moullier, P.; Colle, M.A.; Barkats, M. Intravenous administration of self-complementary AAV9 enables transgene delivery to adult motor neurons. Mol. Ther. 2009, 17, 1187–1196. [Google Scholar] [CrossRef]
- Ayers, J.I.; Fromholt, S.; Sinyavskaya, O.; Siemienski, Z.; Rosario, A.M.; Li, A.; Crosby, K.W.; Cruz, P.E.; DiNunno, N.M.; Janus, C.; et al. Widespread and efficient transduction of spinal cord and brain following neonatal AAV injection and potential disease modifying effect in ALS mice. Mol. Ther. 2015, 23, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Dirosario, J.; Killedar, S.; Zaraspe, K.; McCarty, D.M. Correction of neurological disease of mucopolysaccharidosis IIIB in adult mice by rAAV9 trans-blood-brain barrier gene delivery. Mol. Ther. 2011, 19, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, P.; Rosario, A.; Cruz, P.; Siemienski, Z.; Ceballos-Diaz, C.; Crosby, K.; Jansen, K.; Borchelt, D.R.; Kim, J.Y.; Jankowsky, J.L.; et al. Capsid serotype and timing of injection determines AAV transduction in the neonatal mice brain. PLoS ONE 2013, 8, e67680. [Google Scholar] [CrossRef] [PubMed]
- Gray, S.J.; Matagne, V.; Bachaboina, L.; Yadav, S.; Ojeda, S.R.; Samulski, R.J. Preclinical differences of intravascular AAV9 delivery to neurons and glia: A comparative study of adult mice and nonhuman primates. Mol. Ther. 2011, 19, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Deverman, B.E.; Pravdo, P.L.; Simpson, B.P.; Kumar, S.R.; Chan, K.Y.; Banerjee, A.; Wu, W.L.; Yang, B.; Huber, N.; Pasca, S.P.; et al. Cre-dependent selection yields AAV variants for widespread gene transfer to the adult brain. Nat. Biotechnol. 2016, 34, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Hordeaux, J.; Wang, Q.; Katz, N.; Buza, E.L.; Bell, P.; Wilson, J.M. The Neurotropic Properties of AAV-PHP.B Are Limited to C57BL/6J Mice. Mol. Ther. 2018, 26, 664–668. [Google Scholar] [CrossRef]
- Zlokovic, B.V. The blood-brain barrier in health and chronic neurodegenerative disorders. Neuron 2008, 57, 178–201. [Google Scholar] [CrossRef]
- Piguet, F.; Alves, S.; Cartier, N. Clinical Gene Therapy for Neurodegenerative Diseases: Past, Present, and Future. Hum. Gene Ther. 2017, 28, 988–1003. [Google Scholar] [CrossRef]
- Gray, S.J.; Foti, S.B.; Schwartz, J.W.; Bachaboina, L.; Taylor-Blake, B.; Coleman, J.; Ehlers, M.D.; Zylka, M.J.; McCown, T.J.; Samulski, R.J. Optimizing promoters for recombinant adeno-associated virus-mediated gene expression in the peripheral and central nervous system using self-complementary vectors. Hum Gene Ther. 2011, 22, 1143–1153. [Google Scholar] [CrossRef]
- Wang, L.; Yi, R. 3’UTRs take a long shot in the brain. Bioessays 2014, 36, 39–45. [Google Scholar] [CrossRef]
- Mayr, C. Regulation by 3’-Untranslated Regions. Annu. Rev. Genet. 2017, 51, 171–194. [Google Scholar] [CrossRef] [PubMed]
- Gadalla, K.K.E.; Vudhironarit, T.; Hector, R.D.; Sinnett, S.; Bahey, N.G.; Bailey, M.E.S.; Gray, S.J.; Cobb, S.R. Development of a Novel AAV Gene Therapy Cassette with Improved Safety Features and Efficacy in a Mouse Model of Rett Syndrome. Mol. Ther. Methods Clin. Dev. 2017, 5, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Pfeffer, J.L.; Gururangan, S.; Lester, T.; Lim, D.A.; Shaywitz, A.J.; Westphal, M.; Slavc, I. Intracerebroventricular Delivery as a Safe, Long-Term Route of Drug Administration. Pediatr Neurol 2017, 67, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Slavc, I.; Cohen-Pfeffer, J.L.; Gururangan, S.; Krauser, J.; Lim, D.A.; Maldaun, M.; Schwering, C.; Shaywitz, A.J.; Westphal, M. Best practices for the use of intracerebroventricular drug delivery devices. Mol. Genet. Metab. 2018, 124, 184–188. [Google Scholar] [CrossRef]
- Vuillemenot, B.R.; Korte, S.; Wright, T.L.; Adams, E.L.; Boyd, R.B.; Butt, M.T. Safety Evaluation of CNS Administered Biologics-Study Design, Data Interpretation, and Translation to the Clinic. Toxicol. Sci. 2016, 152, 3–9. [Google Scholar] [CrossRef]
- Bottros, M.M.; Christo, P.J. Current perspectives on intrathecal drug delivery. J. Pain Res. 2014, 7, 615–626. [Google Scholar] [CrossRef]
- Hinderer, C.; Bell, P.; Vite, C.H.; Louboutin, J.P.; Grant, R.; Bote, E.; Yu, H.; Pukenas, B.; Hurst, R.; Wilson, J.M. Widespread gene transfer in the central nervous system of cynomolgus macaques following delivery of AAV9 into the cisterna magna. Mol. Ther. Methods Clin. Dev. 2014, 1, 14051. [Google Scholar] [CrossRef]
- Samaranch, L.; Bringas, J.; Pivirotto, P.; Sebastian, W.S.; Forsayeth, J.; Bankiewicz, K. Cerebellomedullary Cistern Delivery for AAV-Based Gene Therapy: A Technical Note for Nonhuman Primates. Hum. Gene Ther. Methods 2016, 27, 13–16. [Google Scholar] [CrossRef]
- Wright, B.L.; Lai, J.T.; Sinclair, A.J. Cerebrospinal fluid and lumbar puncture: A practical review. J. Neurol. 2012, 259, 1530–1545. [Google Scholar] [CrossRef]
- Hardcastle, N.; Boulis, N.M.; Federici, T. AAV gene delivery to the spinal cord: Serotypes, methods, candidate diseases, and clinical trials. Expert Opin. Biol. Ther. 2018, 18, 293–307. [Google Scholar] [CrossRef]
- Spronck, E.A.; Brouwers, C.C.; Vallès, A.; de Haan, M.; Petry, H.; van Deventer, S.J.; Konstantinova, P.; Evers, M.M. AAV5-miHTT Gene Therapy Demonstrates Sustained Huntingtin Lowering and Functional Improvement in Huntington Disease Mouse Models. Mol. Ther. Methods Clin. Dev. 2019, 13, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Mittermeyer, G.; Christine, C.W.; Rosenbluth, K.H.; Baker, S.L.; Starr, P.; Larson, P.; Kaplan, P.L.; Forsayeth, J.; Aminoff, M.J.; Bankiewicz, K.S. Long-term evaluation of a phase 1 study of AADC gene therapy for Parkinson’s disease. Hum. Gene Ther. 2012, 23, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Huang, X.; Yang, Y. The TLR9-MyD88 pathway is critical for adaptive immune responses to adeno-associated virus gene therapy vectors in mice. J. Clin. Investig. 2009, 119, 2388–2398. [Google Scholar] [CrossRef]
- Martino, A.T.; Suzuki, M.; Markusic, D.M.; Zolotukhin, I.; Ryals, R.C.; Moghimi, B.; Ertl, H.C.; Muruve, D.A.; Lee, B.; Herzog, R.W. The genome of self-complementary adeno-associated viral vectors increases Toll-like receptor 9-dependent innate immune responses in the liver. Blood 2011, 117, 6459–6468. [Google Scholar] [CrossRef]
- Tang, S.C.; Arumugam, T.V.; Xu, X.; Cheng, A.; Mughal, M.R.; Jo, D.G.; Lathia, J.D.; Siler, D.A.; Chigurupati, S.; Ouyang, X.; et al. Pivotal role for neuronal Toll-like receptors in ischemic brain injury and functional deficits. Proc. Natl. Acad. Sci. USA 2007, 104, 13798–13803. [Google Scholar] [CrossRef]
- Olson, J.K.; Miller, S.D. Microglia initiate central nervous system innate and adaptive immune responses through multiple TLRs. J. Immunol. 2004, 173, 3916–3924. [Google Scholar] [CrossRef]
- Bowman, C.C.; Rasley, A.; Tranguch, S.L.; Marriott, I. Cultured astrocytes express toll-like receptors for bacterial products. Glia 2003, 43, 281–291. [Google Scholar] [CrossRef]
- Bsibsi, M.; Ravid, R.; Gveric, D.; van Noort, J.M. Broad expression of Toll-like receptors in the human central nervous system. J. Neuropathol. Exp. Neurol. 2002, 61, 1013–1021. [Google Scholar] [CrossRef]
- Sørensen, L.N.; Reinert, L.S.; Malmgaard, L.; Bartholdy, C.; Thomsen, A.R.; Paludan, S.R. TLR2 and TLR9 synergistically control herpes simplex virus infection in the brain. J. Immunol. 2008, 181, 8604–8612. [Google Scholar] [CrossRef]
- Liu, Y.; Walter, S.; Stagi, M.; Cherny, D.; Letiembre, M.; Schulz-Schaeffer, W.; Heine, H.; Penke, B.; Neumann, H.; Fassbender, K. LPS receptor (CD14): A receptor for phagocytosis of Alzheimer’s amyloid peptide. Brain 2005, 128, 1778–1789. [Google Scholar] [CrossRef] [PubMed]
- Butchi, N.B.; Woods, T.; Du, M.; Morgan, T.W.; Peterson, K.E. TLR7 and TLR9 trigger distinct neuroinflammatory responses in the CNS. Am. J. Pathol. 2011, 179, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Monteilhet, V.; Saheb, S.; Boutin, S.; Leborgne, C.; Veron, P.; Montus, M.F.; Moullier, P.; Benveniste, O.; Masurier, C. A 10 patient case report on the impact of plasmapheresis upon neutralizing factors against adeno-associated virus (AAV) types 1, 2, 6, and 8. Mol. Ther. 2011, 19, 2084–2091. [Google Scholar] [CrossRef] [PubMed]
- Chicoine, L.G.; Montgomery, C.L.; Bremer, W.G.; Shontz, K.M.; Griffin, D.A.; Heller, K.N.; Lewis, S.; Malik, V.; Grose, W.E.; Shilling, C.J.; et al. Plasmapheresis eliminates the negative impact of AAV antibodies on microdystrophin gene expression following vascular delivery. Mol. Ther. 2014, 22, 338–347. [Google Scholar] [CrossRef]
- Holt, P.G.; Sedgwick, J.D.; O’Leary, C.; Krska, K.; Leivers, S. Long-lived IgE- and IgG-secreting cells in rodents manifesting persistent antibody responses. Cell. Immunol. 1984, 89, 281–289. [Google Scholar] [CrossRef]
- Slifka, M.K.; Antia, R.; Whitmire, J.K.; Ahmed, R. Humoral immunity due to long-lived plasma cells. Immunity 1998, 8, 363–372. [Google Scholar] [CrossRef]
- Zand, M.S.; Vo, T.; Huggins, J.; Felgar, R.; Liesveld, J.; Pellegrin, T.; Bozorgzadeh, A.; Sanz, I.; Briggs, B.J. Polyclonal rabbit antithymocyte globulin triggers B-cell and plasma cell apoptosis by multiple pathways. Transplantation 2005, 79, 1507–1515. [Google Scholar] [CrossRef]
- Jayne, D.; Tyndall, A. Autologous stem cell transplantation for systemic lupus erythematosus. Lupus 2004, 13, 359–365. [Google Scholar] [CrossRef]
- Karman, J.; Gumlaw, N.K.; Zhang, J.; Jiang, J.L.; Cheng, S.H.; Zhu, Y. Proteasome inhibition is partially effective in attenuating pre-existing immunity against recombinant adeno-associated viral vectors. PLoS ONE 2012, 7, e34684. [Google Scholar] [CrossRef]
- Goldshmit, Y.; Kanner, S.; Zacs, M.; Frisca, F.; Pinto, A.R.; Currie, P.D.; Pinkas-Kramarski, R. Rapamycin increases neuronal survival, reduces inflammation and astrocyte proliferation after spinal cord injury. Mol. Cell. Neurosci. 2015, 68, 82–91. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perez, B.A.; Shutterly, A.; Chan, Y.K.; Byrne, B.J.; Corti, M. Management of Neuroinflammatory Responses to AAV-Mediated Gene Therapies for Neurodegenerative Diseases. Brain Sci. 2020, 10, 119. https://doi.org/10.3390/brainsci10020119
Perez BA, Shutterly A, Chan YK, Byrne BJ, Corti M. Management of Neuroinflammatory Responses to AAV-Mediated Gene Therapies for Neurodegenerative Diseases. Brain Sciences. 2020; 10(2):119. https://doi.org/10.3390/brainsci10020119
Chicago/Turabian StylePerez, Barbara A., Alison Shutterly, Ying Kai Chan, Barry J. Byrne, and Manuela Corti. 2020. "Management of Neuroinflammatory Responses to AAV-Mediated Gene Therapies for Neurodegenerative Diseases" Brain Sciences 10, no. 2: 119. https://doi.org/10.3390/brainsci10020119
APA StylePerez, B. A., Shutterly, A., Chan, Y. K., Byrne, B. J., & Corti, M. (2020). Management of Neuroinflammatory Responses to AAV-Mediated Gene Therapies for Neurodegenerative Diseases. Brain Sciences, 10(2), 119. https://doi.org/10.3390/brainsci10020119




