Abstract
Perfluoroalkyl and polyfluoroalkyl substances (PFASs) have become a global environmental concern due to their extreme persistence and toxicity. In this study, perfluorooctanoic acid (PFOA) was removed from aqueous solutions using porous carbon adsorbents synthesized from peach stones. The novelty of this work lies in the development of a procedure for obtaining a suitable carbon adsorbent, whose properties are consistent with the properties of the adsorbate. An appropriate activation was used, allowing the preparation of an adsorbent with a highly developed porous texture and a large surface area, which is a prerequisite for a significant adsorption capacity of the obtained adsorbents towards PFOA. Both carbon adsorbents obtained from peach pits, with clearly different surface chemistry—KOH-activated carbon (ACKOH) and its nitric acid-oxidized derivative (ACHNO3)—for PFOA adsorption were compared, along with the clarification of the relationship between the graphitic structure, pore development, surface functionality and adsorption characteristics. The first adsorbent was produced by chemical activation with KOH at 800 °C, while the second was obtained by oxidative modification of the activated sample with 12% HNO3. Characterization by Raman spectroscopy, SEM, and nitrogen physisorption revealed a highly graphitized structure (ID/IG = 0.86) and well-developed porosity. Adsorption experiments were carried out at PFOA concentrations from 8 to 40 µmol/L using a spectrophotometric method based on methylene blue ion-pair extraction into chloroform. The results showed that ACKOH exhibited a high maximum adsorption capacity of 1660 µmol/g (687.36 mg/g) and followed the Langmuir isotherm model, indicating monolayer adsorption. In contrast, ACHNO3 showed a significantly lower adsorption capacity of 398.36 µmol/g (164.95 mg/g), which was attributed to electrostatic repulsion caused by acidic oxygen-containing surface groups. These findings demonstrate that peach stone-derived activated carbon is a promising, sustainable, and efficient adsorbent for the removal of PFOA from water.
1. Introduction
Over the last two decades, perfluoroalkyl and polyfluoroalkyl substances (PFASs) have emerged as one of the most critical and widespread environmental challenges of the modern era [1,2]. This group of over 10,000 synthetic compounds is defined by a unique chemical structure dominated by the carbon–fluorine (C–F) bond, which is one of the shortest and most energetically stable bonds in organic chemistry, and provides these molecules with exceptional resistance to thermal, chemical, and biological degradation. This persistence, combined with their amphiphilic nature—the ability to repel both water and fats—has led to their extensive industrial application, ranging from aqueous film-forming foams and textiles to food packaging and cosmetics.
The degree of PFAS contamination extends far beyond the fixed borders of industrial zones [3,4,5]. Due to their high mobility in aquatic environments and capacity for long-range atmospheric transport, these substances are detected in the most remote ecosystems, including the blood of Arctic wildlife and within the global hydrological cycle. Unlike classical persistent organic pollutants, PFASs exhibit a strong tendency toward bioaccumulation by binding to blood proteins, resulting in prolonged half-lives within the human body. Toxicological studies reveal a concerning correlation between chronic PFAS exposure and severe health outcomes [6,7,8].
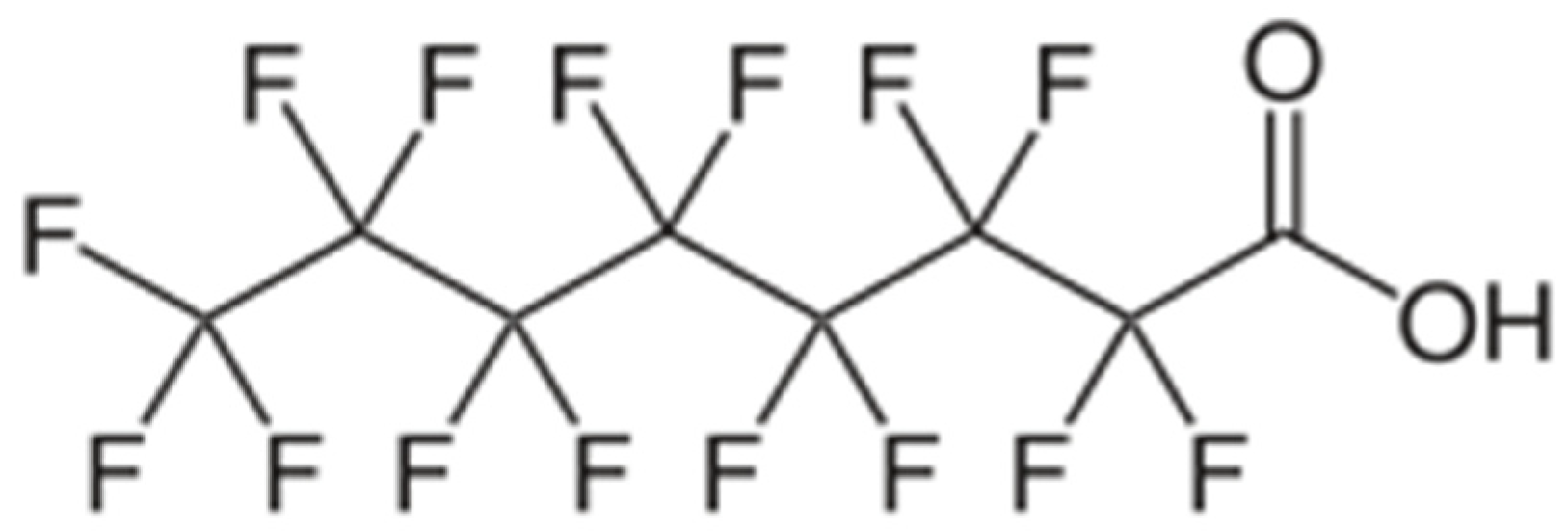
The World Health Organization (WHO) and the International Agency for Research on Cancer (IARC) have recently classified perfluorooctanoic acid (PFOA) as a well-known human carcinogen (Group 1), highlighting risks such as immunosuppression, endocrine disruption, and reproductive abnormalities. PFOA is classified as a persistent organic pollutant due to its exceptional stability; it does not degrade in the environment and bioaccumulates within living organisms. Its molecular formula is C7F15COOH, with a molecular weight of 414.07 g/mol [7,8,9,10,11,12]. The perfluorinated “tail” (C7F15) consists of seven carbon atoms where all hydrogen atoms have been substituted with fluorine. The C–F bond is one of the shortest and most energetically robust bonds in organic chemistry. Due to their atomic radius and high electronegativity, the fluorine atoms create a dense “electron shell” around the carbon skeleton, rendering it inert to chemical and biological degradation. The carboxylic “head” (–COOH/–COO–) provides the molecule’s hydrophilicity and is responsible for its negative charge at neutral pH (Figure 1).
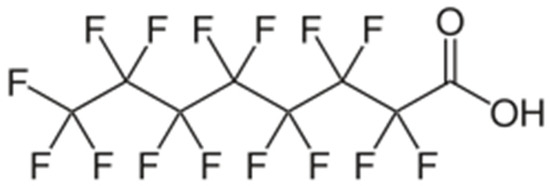
Figure 1.
Structural formula of perfluorooctanoic acid [7].
The molecule exhibits an elongated, rod-like morphology. Its length varies between 1.2 and 1.6 nm, while its width ranges from 0.5 to 0.6 nm. Due to the significant atomic volume of the fluorine atoms, the carbon chain often adopts a slightly helical configuration to minimize steric hindrance. In this case, the colorimetric content determination in aqueous solutions by UV/VIS spectroscopy is suitable [13]. This growing body of evidence regarding toxicity has prompted a significant change in global regulatory frameworks, including the implementation of strict drinking water limits in both the European Union and the United States [6,10,11,12].
In its recent updates, the WHO introduced provisional reference values, establishing a threshold of 0.1 µg/L individually for the two most extensively studied representatives—PFOA and PFOS. In comparison, a limit of 0.5 µg/L was set for the total concentration of all identified PFAS compounds in drinking water. These values reflect the organization’s effort to balance toxicological risks with the technological feasibility of global monitoring [12].
During the 2025–2026 period, the WHO entered a new phase of activity, focused on so-called Phases 2 and 3. These initiatives include expanding the monitoring scope to less-studied PFAS compounds and developing innovative methodologies for cumulative risk assessment [14,15]. A primary priority is understanding their effect—the interaction of various PFAS types entering the human body not only through water but also through the food chain. In this manner, the WHO aims to establish a more comprehensive health protection strategy that is both scientifically grounded and globally applicable [16].
Conventional wastewater treatment plants are primarily designed to remove biological contaminants and readily degradable organic compounds through standard sedimentation and biological oxidation processes. Due to their exceptional chemical stability, PFAS molecules pass through these facilities nearly intact, rendering drinking water a major source of chronic human exposure. In response to this challenge, specialized technologies such as ion-exchange resins and membrane filtration (reverse osmosis) are being implemented; however, these often require substantial initial investment and high maintenance costs [7,17].
Against this backdrop, activated carbon adsorption emerges as the most suitable and effective method for the large-scale treatment of PFOA-contaminated water. This technology stands out as highly cost-effective, requiring relatively simple infrastructure and allowing for easy integration into existing treatment systems without the need for radical reconstruction. The principal advantage of activated carbon lies in its high accessibility and the low cost of the filter media compared to alternatives [18,19,20].
Although filtration methods are often criticized for merely “transferring” the contaminant from the aqueous phase to a solid phase, activated carbon offers a unique advantage in the waste treatment cycle. Once saturated, it can undergo high-temperature thermal regeneration. This process not only restores the adsorption capacity of the carbon but also ensures the definitive destructive mineralization of PFOA molecules by breaking the resilient carbon–fluorine bonds. Consequently, this method is not merely a temporary capture solution but a sustainable and financially accessible strategy for the long-term protection of water resources [21,22].
Despite the proven efficiency of commercial activated carbon brands, the growing global demand for PFOA water remediation necessitates the development of a new generation of adsorbents with optimized characteristics. Most conventional carbon materials are designed for general purification and often exhibit limited capacity toward the specific structure of PFOA, particularly when competing with other organic matter present in the water [23].
Driven by these challenges, the primary objective of the present work is the development of an innovative synthesis method for a specialized carbon adsorbent characterized by high selectivity and efficiency in the removal of PFOA from aqueous environments. The research focuses on the precise control of textural parameters—specifically surface area, pore size distribution, and surface chemistry—to achieve maximum affinity toward fluorinated compounds.
The development of such an adsorbent aims not only to enhance process kinetics but also to provide a cost-effective solution based on accessible raw materials and energy-efficient synthesis methods. By optimizing adsorption capacity, the proposed method seeks to reduce the frequency of filter media replacement, which is critical for the large-scale application of this technology in industrial and municipal settings. Ultimately, this work endeavors to provide a sustainable tool for combating contamination that meets the strict health and environmental protection requirements established by international organizations such as the WHO.
Among various biomass precursors, peach stones (Prunus persica) stand out due to their high lignin content and dense lignocellulosic matrix. These properties are fundamental prerequisites for the production of activated carbons with high mechanical strength and a well-developed micropore structure. The hierarchical pore architecture of peach stone-derived carbons is particularly suitable for the adsorption of perfluorinated compounds like PFOA, as it provides the necessary surface area for hydrophobic interactions while maintaining favorable diffusion kinetics.
The main objective of the work is to select a suitable processing of the raw material, as a result of which to obtain activated carbon with a very large surface area. Further, we determine the influence of the chemical nature of the surface of the adsorbent, obtained as a result of oxidative treatment, on the adsorption capacity towards PFOA.
2. Materials and Methods
2.1. Synthesis of Carbon Adsorbent
A KOH solution (≥85%, CAS № 1310-58-3, Valerus Ltd., Sofia, Bulgaria) was mixed with peach stones with a grain size of 1–5 mm. The ratio of raw material and activating reagent was 1:1. After impregnation of the raw material with KOH, the resulting product was dried at a temperature of 150 °C. The dried product was placed in a stainless steel reactor and subjected to temperature treatment in a nitrogen atmosphere to a temperature of 800 °C, holding at the final temperature for 1 h, with a temperature increase rate of 15 °C/min (Sample 1). The solid product thus obtained was treated with 12% HNO3, 65%, CAS № 7697-37-2, Valerus Ltd., Sofia, Bulgaria (Sample 2) [24]. The samples are labeled in the text respectively: Sample 1—peach stones treated with KOH–ACKOH, and Sample 2— peach stones treated with 12% HNO3–ACHNO3.
2.2. Methods for Characterization of Raw Materials and Resulting Products
Elemental analysis was performed on a Vario Macro Cube (Elementar Analyzensysteme GmbH, Langenselbold, Germany) apparatus for the determination of C, H, N, S. The oxygen content was determined by difference. Carbon was determined as CO2, hydrogen was determined as H2O, while sulfur was determined as SO2. This system utilizes a precise TCD detector to measure gas flow conductivity, with a standard deviation for C, H, N, and S determination of less than 0.1%.
The sample (1–50 mg), covered with tin foil, falls from the autosampler into an oxidation column, where it is heated to 1150 °C in an oxygen atmosphere. After combustion, a gas mixture containing CO2, H2O, SO2 and nitrogen oxides is formed, and the ash remains in a quartz ash tube. The gas mixture passes through a reduction column containing a copper catalyst, which converts nitrogen oxides into nitrogen. The gas mixture then passes through adsorption columns for CO2, H2O and SO2. The remaining nitrogen passes through a precise TCD detector, which determines the gas flow by measuring its conductivity. Then, CO2, H2O and SO2 are passed one after the other, sequentially, through a CVD detector and measured. Based on these results, the instrument calculates the content (wt.%) of N, C, H and S in the sample.
The texture of the synthesized carbon material was characterized by N2 adsorption at −196 °C, carried out in a Quantachrome Autosorb iQ-C-XR/MP automatic volumetric apparatus (Quantachrome Inc., Boynton Beach, FL, USA). This automatic volumetric apparatus provides pressure measurement accuracy of 0.1% of the full scale, ensuring high reproducibility in calculating BET surface area and pore volume. Before the experiments, the sample was degassed under vacuum at 350 °C overnight. The isotherms were used to calculate the specific surface area SBET, the total pore volume Vtotal, and the micropore volume Vmicro using the T-plot method [25,26,27,28,29].
The amount of oxygen-containing surface functional groups with increasing acidity was determined by Boehm’s method of titration with basic solutions of different base strength (NaHCO3 (≥99.5%), Na2CO3 (≥99.8%), NaOH (98–99%, pellets), and C2H5ONa (96% ethanol, NaOH 98–99%), Valerus Ltd., Sofia, Bulgaria) [30]. For this purpose, the samples were agitated for at least 16 h with 0.05 N solutions of four bases. The amount of OH− anions remaining in the solution is determined by adding an excess of standard HCl solution and back titration. The basic surface group content of the samples is determined with 0.05 N HCl. Every measurement was performed three times and the average was taken [31].
The materials were analyzed with a FEI Quanta 250 FEG (2016, FEI Company, Thermo Fisher Scientific, Hillsboro, OR, USA) scanning electron microscope (SEM). Samples with a width of 3 mm and a length of 5 mm were used to observe the fractured surface. The microscope allows for high-resolution visualization down to 3.0 nm, providing a detailed and accurate representation of the pore morphology. The samples were prepared using a sputtering device that applied a 10 Å-thick gold layer in the presence of a flow of argon gas. Vacuum pressure was applied to the samples to improve the visualization of the detectors and to obtain representative images of the samples.
Raman spectroscopy is an extremely important modern technique for the analysis of carbon-based materials, providing unique insight into various aspects of their microstructure, such as orientation, number of layers, defects, and doping. It is a multiscale, high-resolution, and non-destructive method that offers the opportunity to study the specific peculiarities of carbon materials and their composites. In this study, Raman spectra were recorded using a Raman Spectrometer Senterra II (Bruker GmbH, Denkendorf, Germany). Samples (approximately 10 mg) were placed onto a glass surface and analyzed using a vertical 20× objective in a 180° backscattering arrangement. The experimental parameters were as follows: a 532 nm laser wavelength, an exposure time of 100 s, a laser power of 6.5 mW, and a spectral resolution of 1 cm−1 for all samples.
The proximate analysis of the peach stone precursor and the resulting activated carbons was conducted according to standard methods to determine the content of moisture (W-ASTM D2867 [32]), ash (Ash-ASTM D2866 [33]), and volatile matter (Vol.-ASTM D5832 [34]), with results expressed in weight percentages (wt.%). Moisture content was determined by the mass loss after drying to a constant weight, ash content was calculated as the inorganic residue following complete combustion in a muffle furnace, and volatile matter was established through heating in the absence of air. This analysis is essential for characterizing the purity of the precursor and evaluating the impact of the carbonization and activation processes on the structure of the final adsorbents.
The procedure for pH measurements was performed as follows: 4.0 g of carbon (ground, not dried) was weighed into a 250 mL beaker and 100 mL of distilled water was added. After boiling for 5 min, the mixture was set aside and the supernatant liquid was poured off at 60 °C. The decanted portion was set aside to cool down to 25 °C and the pH value was measured with an error of 0.1 (standard NORIT).
A commercially available activated carbon, NORIT RO 0.8 (Cabot Corporation, Boston, MA, USA), was used as a reference standard to evaluate the performance of the peach stone-derived carbon. NORIT is widely recognized in water treatment research as a benchmark material due to its high surface area, standardized pore structure, and proven effectiveness in removing a broad range of organic micropollutants, including perfluorinated compounds. By comparing our synthesized adsorbents with this industry standard, we can better assess the practical applicability and efficiency of the biomass-derived samples.
2.3. Adsorption
A stock solution of perfluorooctanoic acid (PFOA, CAS № 335-67-1) with a concentration of 80 µmol/L was prepared, from which working solutions of 8, 12, 20, and 40 µmol/L were obtained by dilution. Adsorption experiments were conducted by mixing 50 mL of each PFOA solution with 0.05 g of activated carbon. The mixtures were homogenized for 1 h on a shaker at room temperature to reach equilibrium. Subsequently, the solutions were filtered through a 0.45 μm membrane filter to ensure the complete removal of the adsorbent particles.
To determine the residual PFOA concentration in the filtrate, an indirect spectrophotometric method was employed. Specifically, 2.5 mL of methylene blue (MB, CAS № 122965-43-9) and 100 mL of chloroform (CAS № 67-66-3) were added to the filtrates. Methylene blue, a cationic dye, reacts with the anionic PFOA to form a hydrophobic ion-pair complex, which is then extracted into the organic chloroform phase. The mixtures were left to stand for 72 h (Figure 2) to ensure complete phase separation. The absorbance of the chloroform layer was measured at 630 nm using a Pharo 300 UV-Vis spectrometer. The equilibrium adsorption capacity, qe (µmol/g), was calculated according to the following mass balance equation [29]:
where C0 and Ce are the initial and equilibrium concentrations of PFOA (µmol/L), V is the solution volume (L), and m is the mass of the activated carbon (g).
qe = m(C0 − Ce) × V

Figure 2.
Simulated illustration of the experiment conducted (PFOA, chloroform and methylene blue).
Preparation of methylene blue solution (dye): a 100 mL volumetric flask was filled with approximately 60 mL of deionized water. Methylene blue (37.4 mg) was added into the flask. The flask was filled to exactly 100 mL with deionized water.
3. Results and Discussion
3.1. Chemical Composition of the Raw Materials and the Solid Products
Table 1 presents the results of the technical and elemental analysis of raw peach pits and the obtained activated carbons. The raw material is characterized by a very low ash content (0.33 wt.%), which remains relatively low in the final adsorbents (3.50 wt.% for ACKOH and 3.80 wt.% for ACHNO3), confirming the purity of the precursor. During the carbonization process, a significant increase in the carbon content was observed from 51.52% in the raw material to 84.69% for ACKOH. This process was accompanied by a sharp decrease in oxygen (from 41.94% to 11.02%) and hydrogen (from 6.13% to 1.35%), which is evidence of the destruction of oxygen-containing structures and the formation of a stable aromatic matrix. The lowest hydrogen content (1.35%) was observed in ACKOH, which correlates with the higher degree of carbonization in this sample.

Table 1.
Chemical composition of the raw materials and the solid products obtained at 800 °C flash pyrolysis.
3.2. Oxygen Functional Groups Content
The main active adsorption centers on the surface of carbon materials are the various functional groups and paramagnetic centers (unpaired electrons). These centers play an important role in the chemical interaction between the carbon adsorbent and the adsorbate. In the present work, the amount of oxygen groups (with acidic and alkaline character) on the surface of the obtained carbon adsorbent were determined. Oxygen-containing functional groups are extremely important specific characteristics of carbon adsorbents, since they have a strong influence on the adsorption properties as active centers for chemical adsorption.
Table 2 represents the type and quantitative determination of oxygen groups in the synthesized carbon material. On the surface of the activated carbon, various oxygen-containing groups with different chemical properties were found by titration using the Boehm method.

Table 2.
Quantification of oxygen groups on activated carbon surface (mmol/g).
The surface chemistry of the synthesized adsorbents plays a decisive role in the PFOA capture mechanism. The sample obtained by KOH activation (ACKOH) exhibits a predominantly basic surface character, which is consistent with its measured pH value. This basicity is attributed to the presence of pyrone-like structures and basic oxygen-containing groups that facilitate the adsorption of negatively charged PFOA molecules through strong electrostatic attraction forces.
In contrast, as demonstrated in Table 2, the oxidative surface treatment with HNO3 (ACHNO3) leads to the formation of a high concentration of oxygen-containing functional groups, which are predominantly acidic in nature (e.g., carboxyl and lactonic groups). This shift in surface chemistry substantially influences the adsorption process; due to the negative charge of the PFOA molecules at the working pH, they experience electrostatic repulsion from the negatively charged acidic sites on the ACHNO3 surface. Consequently, this leads to a reduction in the overall adsorption efficiency compared to the non-oxidized sample.
3.3. Raman Structural Analysis of ACHNO3 and ACKOH
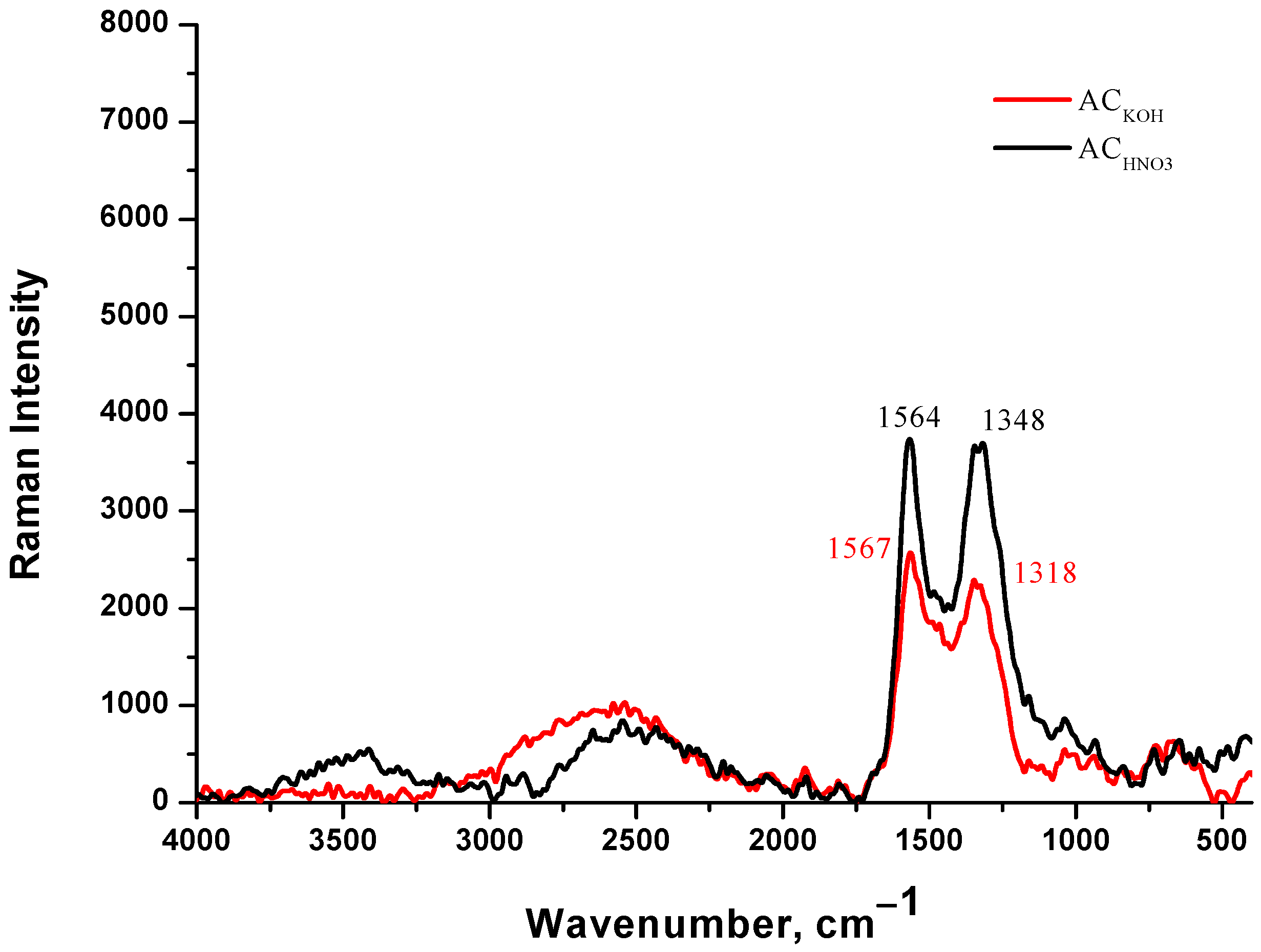
The Raman spectrum of AC from peach stones with KOH is shown in Figure 3. The bands at around 1324 cm−1 and 1574 cm−1 are the G-peak and D-peak of carbon material, respectively. The D-peak represents the defect of the C atom lattice which represents disordered vibration carbon, and the G-peak represents the in-plane tensile vibration of the sp2 hybridization of the C atom which represents the ideal graphite lattices. The intensity ratio of D and G peaks (ID/IG) was calculated to quantify the graphitized degree of AC. The ID/IG ratio of the ACKOH is 1.0, and for ACHNO3 it is 0.86. The ratio is essential for determining the quality of synthetic carbons, for our case suggests highly amorphous or disordered carbons [35].
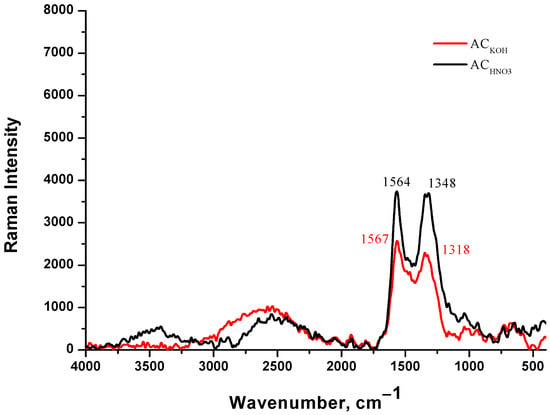
Figure 3.
Raman spectrum on carbon adsorbent obtained from peach stones with KOH (red), and carbon adsorbent obtained from peach stones with HNO3 (black).
3.4. Porous Texture of the Obtained Carbon Adsorbents
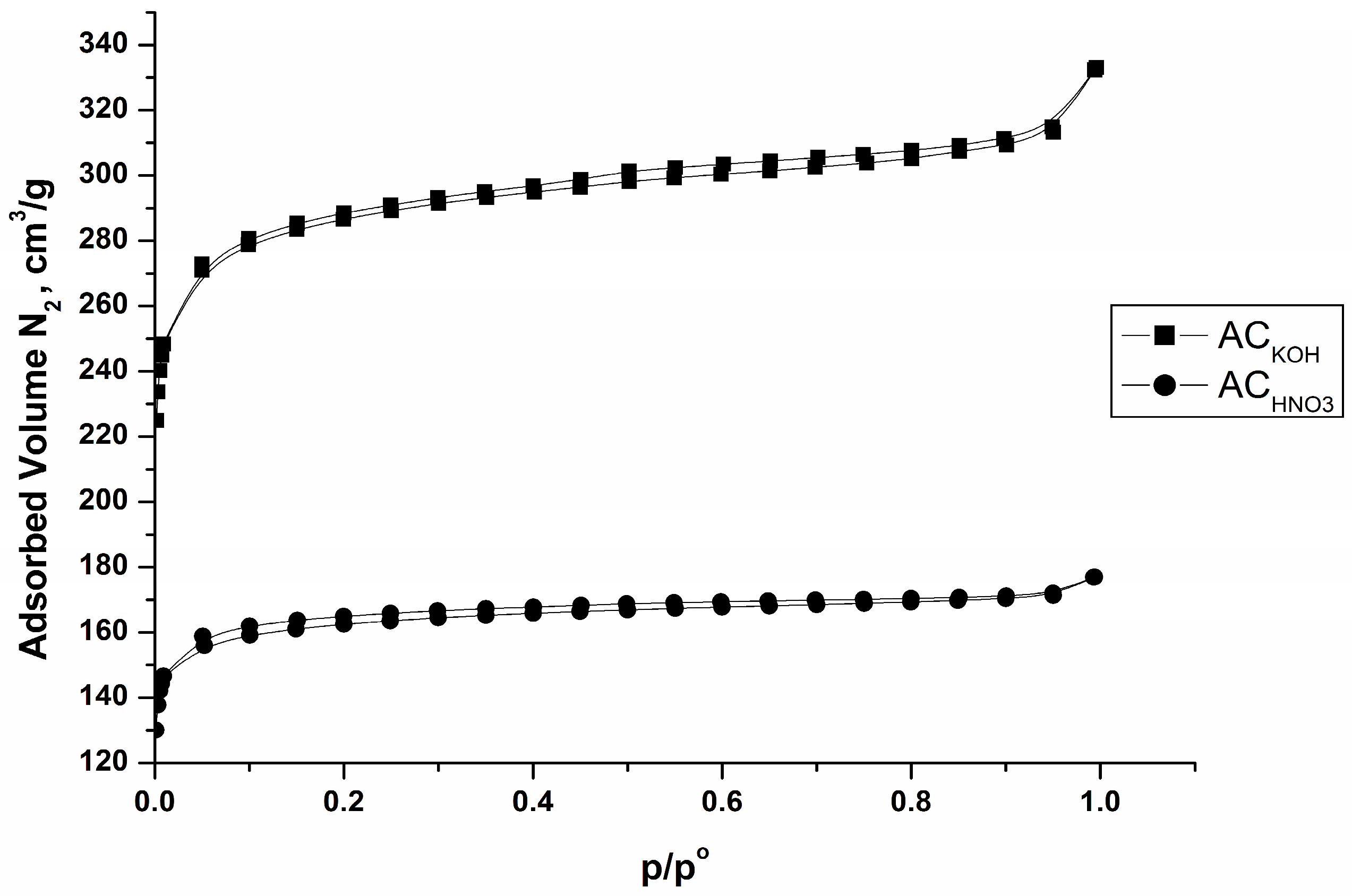
Figure 4 and Table 3 represent data from nitrogen physisorption experiments at −196 °C. Data show that both adsorbents have a microporous texture. The isotherms of Figure 4 are closer to type I (ACKOH) and type II (ACHNO3), indicating presence of micropores, with strong adsorbent–adsorptive interactions according to IUPAC classification [28,29] in the low-pressure region. The variations in the isotherm profiles of the two samples are attributed to the modification of the porous texture in the oxidized specimen resulting from the oxidative treatment. Pores up to 2 nm in size predominate, and there are a small amount of mesopores with a size between 2 and 50 nm. The data also show that the obtained carbon adsorbents have a large surface area, which decreases in the case of the sample oxidized with nitric acid (Table 3). The reason for this is steric hindrance from the large number of oxygen-containing groups, mainly of acidic nature, formed as a result of the oxidation treatment.
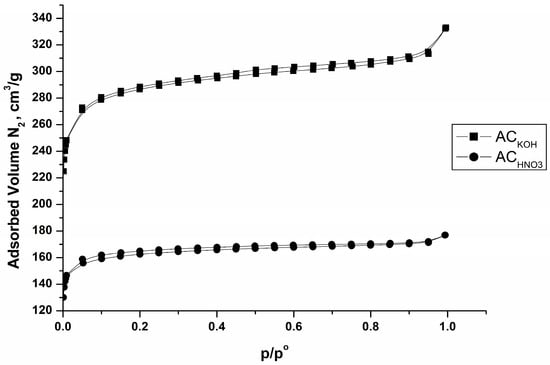
Figure 4.
N2 (−196 °C) adsorption isotherms of carbon adsorbents obtained from peach stones by flash pyrolysis.

Table 3.
Main textural parameters of the obtained activated carbons.
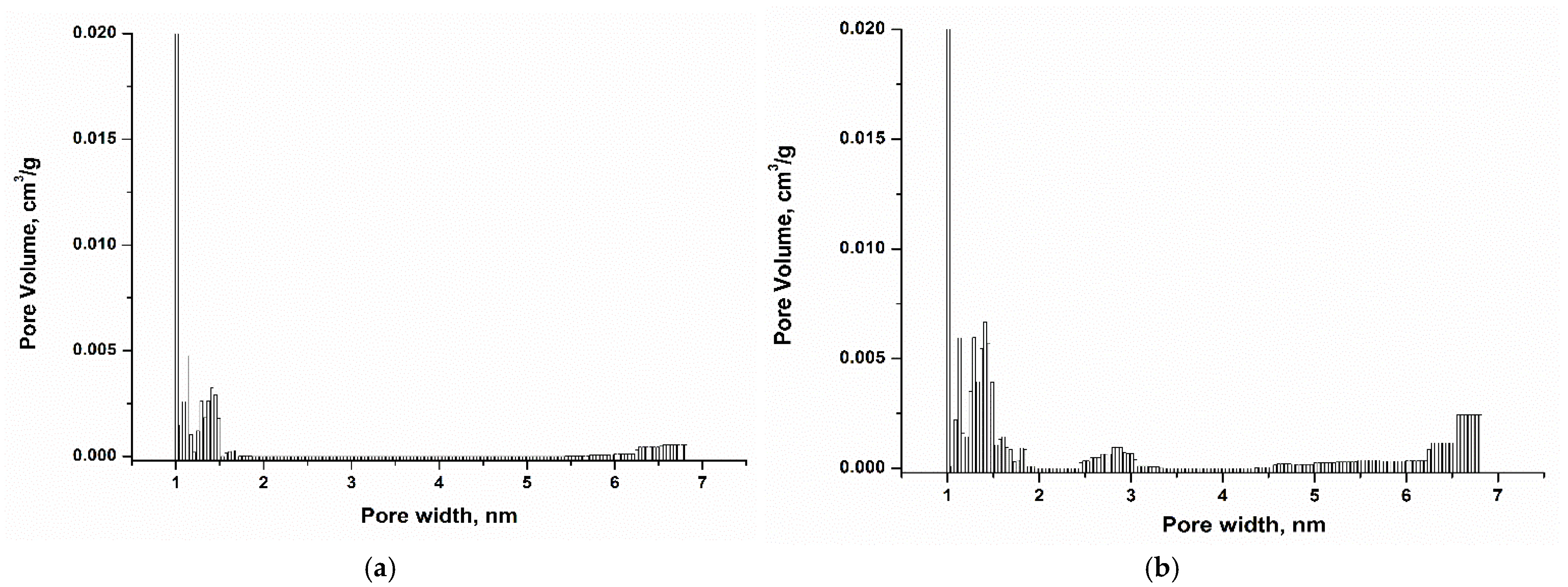
Pore size distribution [36] data are presented in Figure 5 and the results show the presence of micropores in the range 1.5–2.0 nm, and small mesopores in the range 2.0–7.0 nm. Comparison of ACHNO3 and ACKOH samples show that ACKOH possesses higher surface area, total pore volume, micropore volume and mesopore volume (Table 3). This phenomenon is attributed to the influence of the high density of oxygen-containing functional groups on the surface of the oxidized sample. Due to steric hindrance, these groups restrict access to a portion of the adsorbent pores. While this may reduce the extent of physical adsorption to some degree, it significantly enhances the progression of chemisorption and the formation of complexes between the generated oxygen groups and the PFAS molecules.
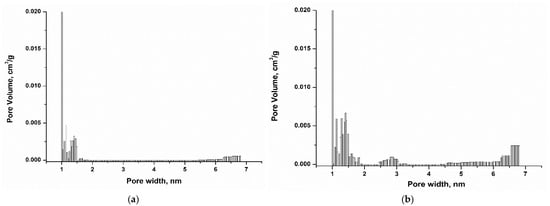
Figure 5.
Pore size distribution of ACHNO3 (a), ACKOH (b).
3.5. Scanning Electronic Microscopy (SEM)
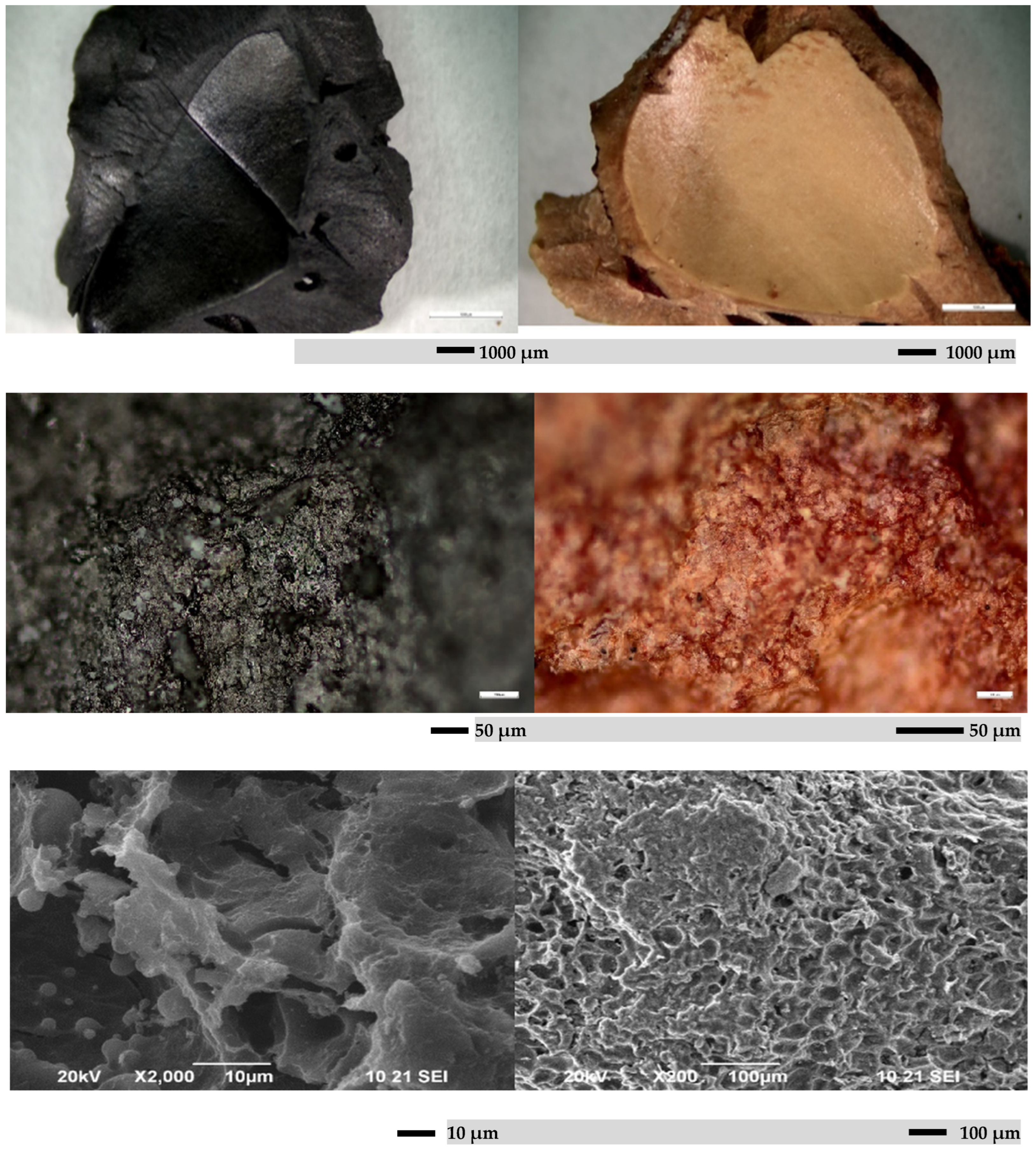
Figure 6 and Figure 7 show SEM micrographs of activated carbon obtained from peach stones. SEM micrographs show that the obtained carbon adsorbent is characterized by the presence of well-developed pores, which is due to the processing method of the raw material and its composition. The SEM data are consistent with the pore distribution data from nitrogen physisorption.
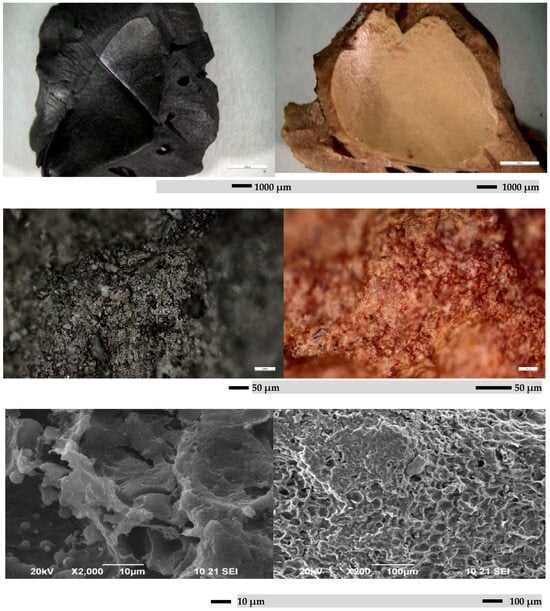
Figure 6.
SEM micrograph of sample from raw material peach stone, and sample treated with KOH.

Figure 7.
SEM micrograph of sample treated with HNO3.
3.6. Adsorption Investigations
PFOA in aqueous solutions react with the dye methylene blue to form a blue-colored ion complex, which is then extracted into chloroform (Figure 8). Subsequently, the absorbance at 630 nm of the chloroform solution is measured with an UV/VIS spectrophotometer(Merck KGaA, Darmstadt, Germany).
C7F15COO− (aq) + MB+ (aq) ⇌ [C7F15COO−·MB+](org)
- C7F15COO−: Perfluorooctanoate (the anionic form of PFOA).
- MB+: Methylene blue cation.
- [C7F15COO−·MB+]: Electrically neutral ion pair (blue-colored complex).
- (aq): Aqueous phase (water).
- (org): Organic phase (chloroform).

Figure 8.
Schematic representation of the ion-pair extraction of PFOA with methylene blue. The phases are labeled as follows: aqueous phase (PFOA + methylene blue + water), and organic phase (chloroform).
Figure 8.
Schematic representation of the ion-pair extraction of PFOA with methylene blue. The phases are labeled as follows: aqueous phase (PFOA + methylene blue + water), and organic phase (chloroform).

The principle of the method is based on the reaction between the cationic dye methylene blue, and the anionic PFOA molecules in an acidic medium. The resulting ion pair is electroneutral and hydrophobic, enabling its separation from the aqueous phase into an organic solvent (chloroform). The intensity of the blue coloration in the organic phase is directly proportional to the analyte concentration, in accordance with the Beer–Lambert law [20]:
A = ϵ × l × c.
- A (Absorbance): also known as optical density or extinction. A dimensionless quantity that measures the amount of light absorbed by the sample at a specific wavelength.
- ϵ (Molar absorptivity/Molar extinction coefficient): measured in L mol−1 cm−1. A constant characteristic of a specific substance (in this case, the PFOA-MB complex) at a given wavelength, indicating how strongly it absorbs light.
- l (Path length): usually measured in cm. The distance the light travels through the cuvette containing the solution.
- c (Concentration): molar concentration measured in mol/L. The amount of the analyte (PFAS) present in the solution.
Since ϵ and l are constants under standard laboratory conditions, a linear relationship is established: the higher the concentration of PFOA in the sample, the more intensely blue the organic phase (chloroform) becomes, resulting in a higher absorbance value (A) recorded by the spectrophotometer.
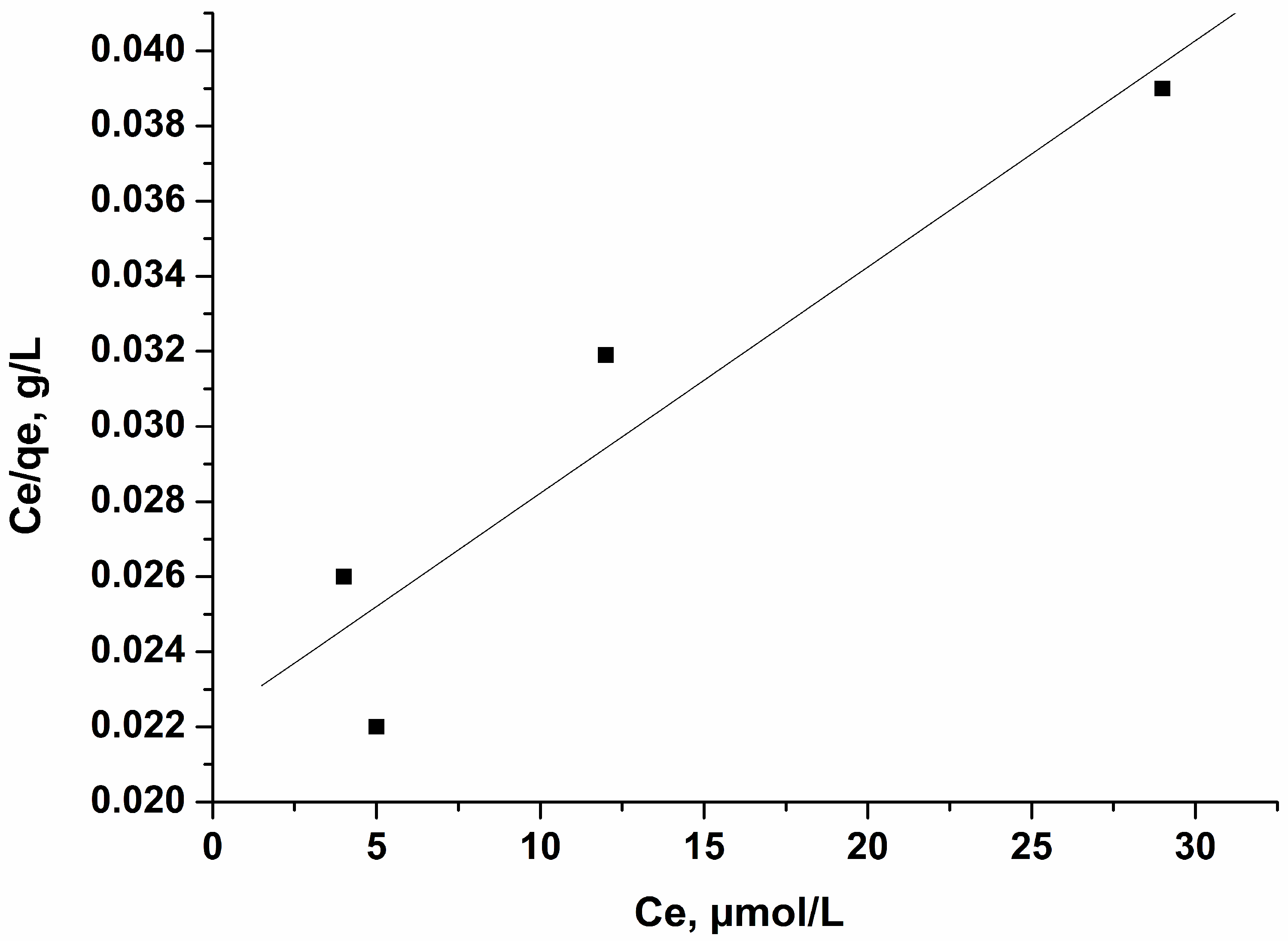
3.6.1. Langmuir Isotherm
The adsorption of perfluorooctanoic acid (PFOA) from aqueous solutions by carbon adsorbents was investigated for different concentrations: 8, 12, 20, and 40 µmol/L. The adsorption isotherm linear form of PFOA on carbon adsorbent treated with KOH is presented in Figure 9. The isotherm is associated with type L of the Giles classification [37], where adsorption proceeds by forming a monolayer in the range of concentrations used. The obtained results demonstrate that the adsorption mechanism of perfluorooctanoic acid (PFOA) on the surface of adsorbent is similar. The experimental data coincide very well with the Langmuir equation [29]:
where Ce (relative error of the measurement 1.0%) is the equilibrium pollutant concentration remaining in solution after adsorption (mg/L), Qe (relative error 2.01%) is the amount of pollutant bound to the adsorbent (mg/g), Qo (relative error 0.01%) is the maximal amount of the pollutant per unit weight of adsorbent (mg/g), and b (relative error 1.0%) is a constant related to the affinity of binding sites (L/mg).
Qe = Qo × b × Ce/(1 + b × Ce),
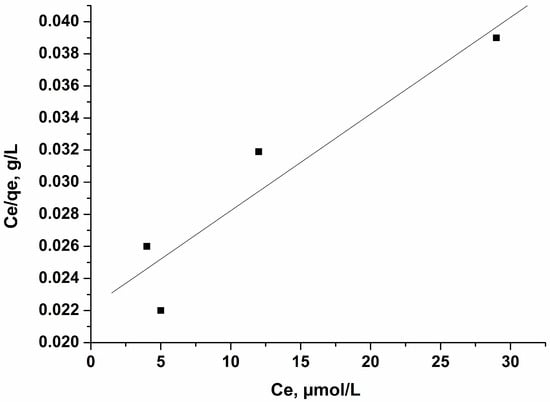
Figure 9.
Langmuir isotherm of PFOA on activated carbon treated with KOH. Conditions: adsorbent amount 0.05 g per 50 mL solution, concentration 8, 12, 20, 40, µmol/L, time of treatment 60 min, 25 °C.
The calculated experimental values of the parameters and the relative affinity of the adsorbates towards the surface of the adsorbents—parameter b, Qo, and the correlation coefficients—are presented in Table 4. The very strong fit shows that the Langmuir model is excellent for predicting the adsorptive behavior of these carbons toward perfluorooctanoic acid (PFOA), which is indicated by the formation of a monomolecular layer. The adsorption capacity of the activated carbon activated with KOH for perfluorooctanoic acid (PFOA) is very high, most probably due to the hydrophobic character of the carbon surface and the porous texture of the carbon. The maximal value calculated by Langmuir equation for this carbon is 1660 µmol/g (687.36 mg/g).

Table 4.
Data for PFOA adsorption obtained from Langmuir (Equation (3)) and Freundlich (Equation (4)) plot.
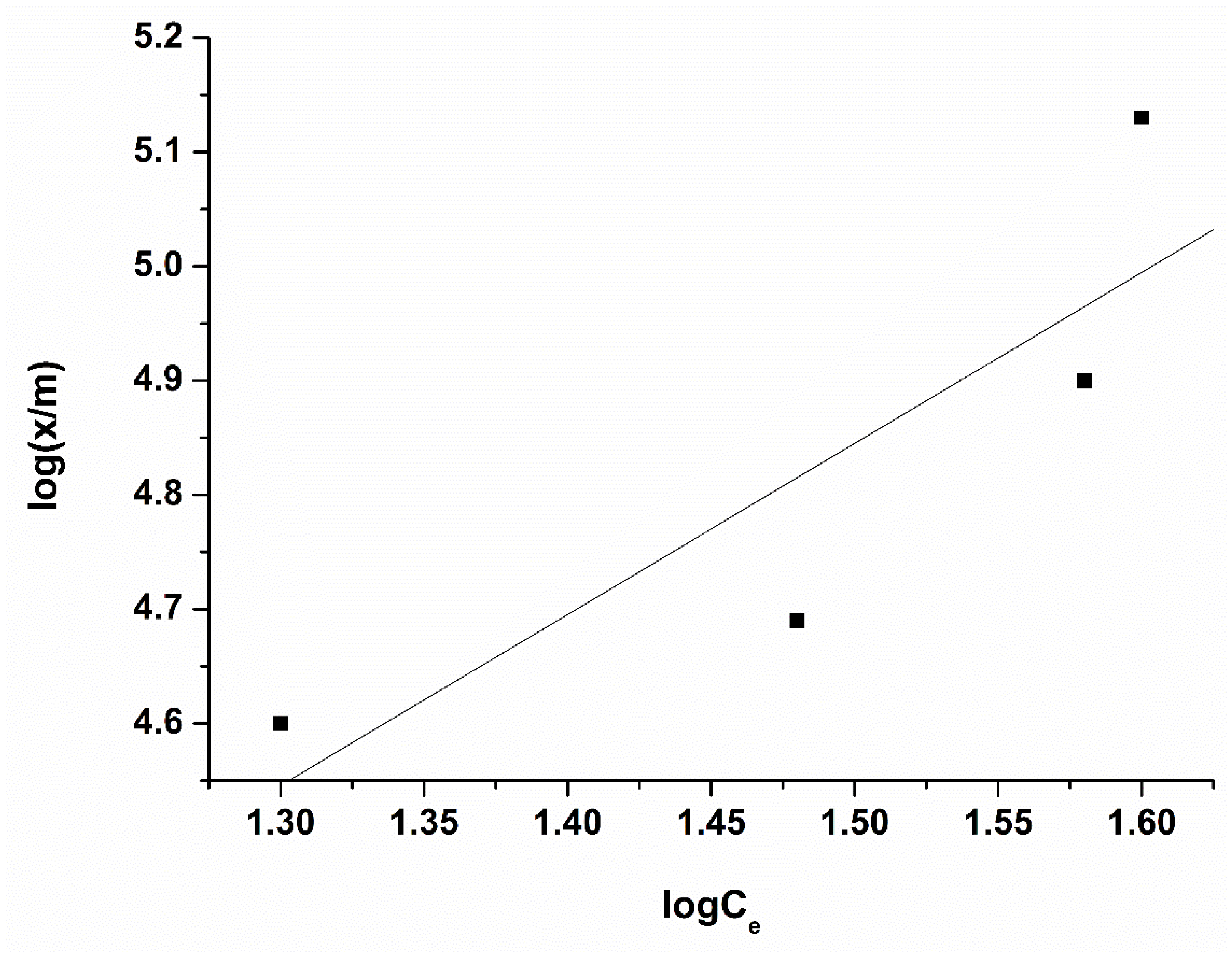
3.6.2. Freundlich Isotherm
The linear form of Freundlich equation [29]
is applied to the experimental results presented in Figure 10, where x is the amount of the adsorbed solute (mg), m is the weight of the adsorbent (g), Ce is the equilibrium concentration (mg/L), n is the slope, showing the variation in the adsorption with concentration, and Kf is the intercept, showing the adsorption capacity of the adsorbent.
log(x/m) = lg Kf + lgCe/n
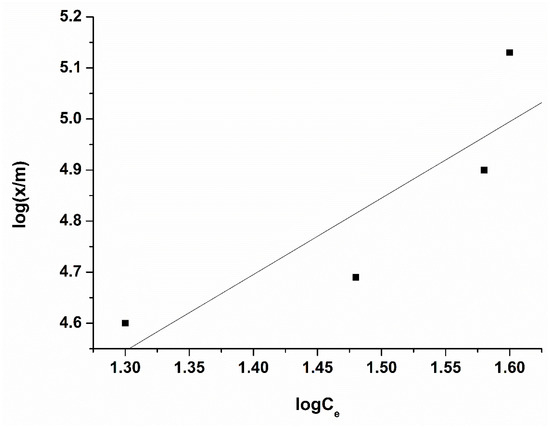
Figure 10.
Freundlich isotherms of PFOA onto activated carbon treated with HNO3. Conditions: adsorbent amount 0.01–0.1 g per 50 mL solution, concentration 40 µmol/L, time of treatment 60 min, 25 °C.
The adsorption capacity for the sample oxidized with nitric acid towards PFOA was calculated by Freundlich equation (398.36 µmol/g ≈ 164.95 mg/g). We believe that this low value for the adsorption capacity of the adsorbent treated with nitric acid is due to the fact that the surface of the activated carbon is dominated by oxygen-containing groups of acidic nature, which repel (electrostatically) negatively charged perfluorooctanoic acid (PFOA), and thus interferes with the adsorption interaction between them. This conclusion is supported by the fact that at the working pH of the solution (pH > pKa of PFOA), PFOA molecules exist predominantly in the anionic form. Simultaneously, Boehm titration confirmed a significant increase in carboxyl and phenolic groups with ACHNO3. As these groups are deprotonated at neutral pH, the carbon surface acquires a negative potential, leading to electrostatic repulsion of PFOA anions. Similar effects have been reported by other authors [8,9], who noted that the introduction of oxygen-containing groups by strong oxidants such as HNO3 reduced the adsorption of hydrophobic organic acids due to steric hindrance and surface repulsion.
The values of maximal adsorption capacity for our samples are much higher than those from the scarce literature data for adsorption of PFOA reported by other researchers (Table 5).

Table 5.
Comparative data on PFOA adsorption on adsorbents.
The fit of experimental data to the Temkin and Dubinin-Radushkevich models, along with the calculated mean free energy (E), can be found in the Supplementary Materials (Table S1).
4. Conclusions
The present study demonstrates the successful conversion of peach stone biomass into high-performance activated carbon for environmental water remediation. The adsorbent obtained via chemical activation with KOH (ACKOH) is characterized by excellent textural properties, low ash content, and a high degree of graphitization (ID/IG = 0.86), ensuring both structural stability and a high affinity for fluorinated organic compounds.
The results indicate that adsorption efficiency is determined by the synergy between the material’s porous texture and its surface chemistry. It was established that micropores (especially those in the 1.5–2.0 nm range) serve as the primary active sites, allowing optimal spatial orientation of the rod-like PFOA molecule. Concurrently, the presence of mesopores facilitates mass transfer and prevents diffusion limitations.
Surface modification plays a decisive role: while the basic functional groups in ACKOH favor adsorption, the introduction of acidic oxygen-containing groups via HNO3 oxidation leads to the electrostatic repulsion of PFOA anions, drastically reducing the capacity to 398.36 µmol/g (164.95 mg/g). The impressive maximum capacity of 1660 µmol/g (687.36 mg/g), calculated according to the Langmuir model, establishes the developed carbon as a competitive and sustainable alternative to commercial adsorbents. In conclusion, the proposed method for utilizing peach stone waste offers a cost-effective strategy for the efficient removal of PFOA from aquatic ecosystems.
The main contribution of the work compared to previous studies is the finding of a suitable processing of the raw material and the obtaining of an adsorbent with a very large surface area of 1100 m2/g, which is an important condition for its adsorption capacity towards the studied adsorbate. The additional oxidative treatment of the adsorbent carried out in order to change the chemical nature of the coal showed that the change does not lead to an increase in its adsorption capacity, but is reduced as a result of the treatment. This statement provides ground to conclude that a large adsorption capacity is due to the positively charged surface of the adsorbent.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/environments13030174/s1, Supplementary Equation: S1: Temkin Isotherm Model—Used to account for adsorbent–adsorbate interactions; it assumes that the heat of adsorption of all molecules in the layer decreases linearly with surface coverage. S2: Polanyi Potential (ϵ)—A fundamental parameter in the Dubinin-Radushkevich model, related to the adsorption free energy. S3: Mean Free Adsorption Energy (E)—Formula for calculating the mean free energy, used to distinguish between physical adsorption (E < 8 kJ/mol) and chemical adsorption or ion exchange (E between 8 and 16 kJ/mol). Table S1: Isotherm Parameters for PFOA Adsorption—List of calculated constants for the Temkin (AT, B) and Dubinin-Radushkevich (β, Qm, E) models for the samples ACKOH and ACHNO3.
Author Contributions
Conceptualization, I.S. and N.P.; methodology, I.S. and N.P.; investigation, I.S., B.R., B.P., A.K. and N.P.; writing—original draft preparation, I.S., A.K., B.R., P.D. and N.P.; writing—review and editing, I.S., A.K. and N.P.; funding acquisition, I.S., B.T. and P.D. All authors have read and agreed to the published version of the manuscript.
Funding
The authors gratefully acknowledge the financial support for this work from the project BG-RRP-2.017-0006 from the Recovery Plan for Europe (NextGenerationEU) and project No. BG16RFPR002-1.014-0015: “Clean Technologies for Sustainable Environment—Water, Waste, Energy for Circular Economy”, funded by the European Regional Development Fund through the Bulgarian program “Research, Innovation and Digitalization for Smart Transformation”. The authors also gratefully thank the Bulgarian Science Fund, grant number KP-06-M77/2.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Jha, G.; Kankarla, V.; McLennon, E.; Pal, S.; Sihi, D.; Dari, B.; Diaz, D.; Nocco, M. Per-and polyfluoroalkyl substances (PFAS) in integrated crop–livestock systems: Environmental exposure and human health risks. Int. J. Environ. Res. Public Health 2021, 18, 12550. [Google Scholar] [CrossRef] [PubMed]
- Panieri, E.; Baralic, K.; Djukic-Cosic, D.; Buha Djordjevic, A.; Saso, L. PFAS molecules: A major concern for the human health and the environment. Toxics 2022, 10, 44. [Google Scholar] [CrossRef]
- Verma, S.; Lee, T.; Sahle-Demessie, E.; Ateia, M.; Nadagouda, M.N. Recent advances on PFAS degradation via thermal and nonthermal methods. Chem. Eng. J. Adv. 2023, 13, 100421. [Google Scholar] [CrossRef]
- Qi, L.; Li, R.; Wu, Y.; Lin, X.; Chen, G. Effect of solution chemistry on the transport of short-chain and long-chain perfluoroalkyl carboxylic acids (PFCAs) in saturated porous media. Chemosphere 2022, 303, 135160. [Google Scholar] [CrossRef]
- Costello, M.C.S.; Lee, L.S. Sources, fate, and plant uptake in agricultural systems of per-and polyfluoroalkyl substances. Curr. Pollut. Rep. 2024, 10, 799–819. [Google Scholar] [CrossRef]
- Podder, A.; Sadmani, A.A.; Reinhart, D.; Chang, N.-B.; Goel, R. Per and poly-fluoroalkyl substances (PFAS) as a contaminant of emerging concern in surface water: A transboundary review of their occurrences and toxicity effects. J. Hazard. Mater. 2021, 419, 126361. [Google Scholar] [CrossRef]
- PubChem. Perfluorooctanoic Acid; CID 9554; National Center for Biotechnology Information, U.S. National Library of Medicine: Bethesda, MD, USA, 2005. [Google Scholar]
- Zhu, X.; Zhang, M.; Zhang, Z.; Li, M.; Liu, M.; Jing, H.; Tan, J.; Jia, H.; Wang, J. Electrochemical sensor with redox-active poly (acridine orange) imprinted film for ultrasensitive and selective detection of perfluorooctanoic acid: A binding-induced electroactivity suppression strategy. J. Hazard. Mater. 2026, 505, 141442. [Google Scholar] [CrossRef]
- Misal, S.N.; Ruiz, R.A.; Duan, J.; Choi, Y.J.; Adkins, J.W.; Chen, C.; Elam, J.W.; Abiade, J.T.; Schaefer, C.E.; Lee, L.S.; et al. Electrochemical Oxidation of Perfluorooctanoic Acid on a Bi2O3–SnO2/Ti4O7 Reactive Electrochemical Membrane: Application to Groundwater Treatment with Mechanistic Insights. ACS EST Eng. 2025, 5, 3591–3606. [Google Scholar] [CrossRef]
- Brake, H.D.; Wilkins, M.J.; Kaneene, J.B. Per-and polyfluoroalkyl substances: Using comparative medicine to understand exposure and adverse health outcomes in people and their pets. Am. J. Vet. Res. 2023, 84, ajvr.22.12.0221. [Google Scholar] [CrossRef] [PubMed]
- Cohen, N.J.; Yao, M.; Midya, V.; India-Aldana, S.; Mouzica, T.; Andra, S.S.; Narasimhan, S.; Meher, A.K.; Arora, M.; Chan, J.K.Y. Exposure to perfluoroalkyl substances and women’s fertility outcomes in a Singaporean population-based preconception cohort. Sci. Total Environ. 2023, 873, 162267. [Google Scholar] [CrossRef]
- Coffin, E.S.; Reeves, D.M.; Cassidy, D.P. PFAS in municipal solid waste landfills: Sources, leachate composition, chemical transformations, and future challenges. Curr. Opin. Environ. Sci. Health 2023, 31, 100418. [Google Scholar] [CrossRef]
- Giles, C.H.; Smith, D.; Huitson, A. A general treatment and classification of the solute adsorption isotherm. I. Theoretical. J. Colloid Interface Sci. 1974, 47, 755–765. [Google Scholar] [CrossRef]
- WHO. Call for Experts: Technical Advisory Group on PFAS Assessment (TAG-PFAS-2); World Health Organization: Geneva, Switzerland, 2025; Available online: https://www.who.int/news-room/articles-detail/request-for-expressions-of-interest--who-initiatives-to-evaluate-pfas-(phase-2) (accessed on 16 January 2026).
- Rampazzo, G.; Arioli, F.; Pagliuca, G.; Depau, G.; Zironi, E.; Gazzotti, T. Cumulative Exposure and Health Risk Assessment of PFAS in Animal-Derived Foods Using the Relative Potency Factor Approach. Toxics 2025, 13, 931. [Google Scholar] [CrossRef]
- Pozzebon, E.A.; Seifert, L. Emerging environmental health risks associated with the land application of biosolids: A scoping review. Environ. Health 2023, 22, 57. [Google Scholar] [CrossRef]
- Lenka, S.P.; Kah, M.; Padhye, L.P. A review of the occurrence, transformation, and removal of poly- and perfluoroalkyl substances (PFAS) in wastewater treatment plants. Water Res. 2021, 199, 117187. [Google Scholar] [CrossRef]
- Ochoa-Herrera, V.; Sierra-Alvarez, R. Removal of perfluorinated surfactants by sorption onto granular activated carbon, zeolite and sludge. Chemosphere 2008, 72, 1588–1593. [Google Scholar] [CrossRef]
- Qian, J.; Shen, M.; Wang, P.; Wang, C.; Li, K.; Liu, J.; Lu, B.; Tian, X. Perfluorooctane sulfonate adsorption on powder activated carbon: Effect of phosphate (P) competition, pH, and temperature. Chemosphere 2017, 182, 215–222. [Google Scholar] [CrossRef]
- Qu, Y.; Zhang, C.; Li, F.; Bo, X.; Liu, G.; Zhou, Q. Equilibrium and kinetics study on the adsorption of perfluorooctanoic acid from aqueous solution onto powdered activated carbon. J. Hazard. Mater. 2009, 169, 146–152. [Google Scholar] [CrossRef]
- Zhi, Y.; Liu, J. Surface modification of activated carbon for enhanced adsorption of perfluoroalkyl acids from aqueous solutions. Chemosphere 2016, 144, 1224–1232. [Google Scholar] [CrossRef]
- Laghlimi, C.; Moutcine, A.; Chtaini, A.; Isaad, J.; Zannou, A.; Ziat, Y.; Ahari, M. Voltammetric investigation of the complexing effect of Capparis spinosa on heavy metals: Application in the treatment of water. Ionics 2023, 29, 5441–5452. [Google Scholar] [CrossRef]
- Rigby, S.; Mousa, S. Structural Characterisation of Disordered Porous Materials Using Gas Sorption and Complementary Techniques. Surfaces 2026, 9, 20. [Google Scholar] [CrossRef]
- Stoycheva, I.; Petrova, B.; Tsyntsarski, B.; Petrov, N.; Ranguelov, B. Influence of Peach Stone Composition, Pretreatment and Processing Method on the Properties of the Resulting Carbon Adsorbent. Biomass 2026, 6, 14. [Google Scholar] [CrossRef]
- Rouqueol, J.; Rouquerol, F.; Sing, K.; Llewellyn, P.; Maurin, G.; Denoyel, R.; Coulet, M.V.; Beurroies, I. Adsorption by Powders and Porous Solids—Principles, Methodology and Applications; Elsevier: Amsterdam, The Netherlands, 2026. [Google Scholar]
- Menéndez-Díaz, J.A.; Martín-Gullón, I. Activated Carbon: From Synthesis to Applications; Elsevier: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Gomez-Serrano, V.; Gonzalez-Garcia, C.; Gonzalez-Martin, M. Nitrogen adsorption Isotherms on ca bonaceous materials. Comparison of BET and Langmuir surface areas. Powder Technol. 2001, 116, 103–108. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report) Pure. Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Ehiomogue, P.; Ahuchaogu, I.I.; Ahaneku, I.E. Review of adsorption isotherms models. Acta Tech. Corviniensis-Bull. Eng. 2021, 14, 87–96. [Google Scholar]
- Schönherr, J.; Buchheim, J.R.; Scholz, P.; Adelhelm, P. Boehm Titration Revisited (Part II): A Comparison of Boehm Titration with Other Analytical Techniques on the Quantification of Oxygen-Containing Surface Groups for a Variety of Carbon Materials. J. Carbon Res. 2018, 4, 22. [Google Scholar] [CrossRef]
- Peng, H.; Ma, G.; Sun, K.; Mu, J.; Zhang, Z.; Lei, Z. Formation of Carbon Nanosheets via Simultaneous Activation and Catalytic Carbonization of Macroporous Anion-Exchange Resin for Supercapacitors Application. ACS Appl. Mater. Interfaces 2014, 6, 20795–20803. [Google Scholar] [CrossRef]
- ASTM D2867; Standard Test Methods for Moisture in Activated Carbon. ASTM International: West Conshohocken, PA, USA, 2024.
- ASTM D2866; Standard Test Method for Total Ash Content of Activated Carbon. ASTM International: West Conshohocken, PA, USA, 2018.
- ASTM D5832; Standard Test Method for Volatile Matter Content of Activated Carbon Samples. ASTM International: West Conshohocken, PA, USA, 2022.
- Li, Z.; Deng, L.; Kinloch, I.A.; Young, R.J. Raman spectroscopy of carbon materials and their composites: Graphene, nanotubes and fibres. Prog. Mater. Sci. 2023, 135, 101089. [Google Scholar] [CrossRef]
- Groen, J.C.; Peffer, L.A.A.; Pérez-Ramírez, J. Pore size determination in modified micro- and mesoporous materials. Pitfalls and limitations in gas adsorption data analysis. Microporous Mesoporous Mater. 2003, 60, 1–17. [Google Scholar] [CrossRef]
- Giles, C.H.; D’Silva, A.P.; Easton, I.A. A general treatment and classification of the solute adsorption isotherm part. II. Experimental interpretation. J. Colloid Interface Sci. 1974, 47, 766–778. [Google Scholar] [CrossRef]
- Saawarn, B.; Mahanty, B.; Subrata, H. Adsorption of perfluorooctanoic acid from aqueous matrices onto chitosan-modified magnetic biochar: Response surface methodology-based modeling, performance, and mechanism. Environ. Pollut. 2025, 368, 125734. [Google Scholar] [CrossRef]
- Meegoda, J.N.; Mudalige, R.N.; Perera, D.C. Maximum Adsorption Capacity of Perfluorooctanoic Acid (PFOA) on Clays. Environments 2026, 13, 37. [Google Scholar] [CrossRef]
- Zhang, Z.; Sarkar, D.; Datta, R.; Deng, Y. Adsorption of perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) by aluminum-based drinking water treatment residuals. J. Hazard. Mater. Lett. 2021, 2, 100034. [Google Scholar] [CrossRef]
- Saawarn, B.; Mahanty, B.; Hait, S. Adsorptive removal of perfluorooctanoic acid from aqueous matrices using peanut husk-derived magnetic biochar: Statistical and artificial intelligence approaches, kinetics, isotherm, and thermodynamics. Chemosphere 2024, 360, 142397. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.