Abstract
Background: Chronic ethanol consumption is a major global health concern traditionally associated with liver disease. Ethanol disrupts gut microbial communities, compromises intestinal barrier function, and contributes to hepatic, metabolic, and neurocognitive disorders. Methods: We conducted a systematic PubMed search and meta-analysis of 11 human and 19 animal studies evaluating ethanol-induced gut microbiota alterations. Studies were assessed for microbial diversity, taxonomic shifts, barrier integrity, and systemic effects. Effect sizes were calculated where possible, and interventional outcomes were examined. Results: Across species, ethanol exposure was consistently associated with reduced microbial diversity and depletion of beneficial commensals such as Faecalibacterium, Lactobacillus, Akkermansia, and Bifidobacterium, alongside an expansion of proinflammatory taxa (Proteobacteria, Enterococcus, Veillonella). Our analysis uniquely highlights discrepancies between human and animal studies, including opposite trends in specific genera (e.g., Akkermansia and Bifidobacterium) and the impact of confounders such as antibiotic exposure in human cohorts. We also demonstrate that microbiota-targeted interventions can partially restore diversity and improve clinical or behavioral outcomes. Conclusions: This meta-analysis highlights reproducible patterns of ethanol-induced gut dysbiosis across both human and animal studies.
1. Introduction
Alcohol consumption is deeply embedded in many cultures around the world. With more than 2.3 billion global consumers and approximately 75 million individuals suffering from alcohol use disorders (AUDs), ethanol (ethyl alcohol) poses a formidable burden on public health systems worldwide [1].
Figure 1 displays the top nine countries ranked by total per capita alcohol consumption in 2022, based on WHO data. On the left, it shows the global average alcohol consumption (liters per capita) from 2000 to 2022, peaking around 2012 at nearly 6.5 L, followed by a gradual decline to about 5 L by 2022. The shaded area represents a confidence interval or range of variation across countries. On the right, a bar chart ranks countries by their 2022 per capita alcohol consumption, with Romania leading at 17.1 L, followed by Georgia (15.5), Latvia (14.7), and Moldova (14.1).
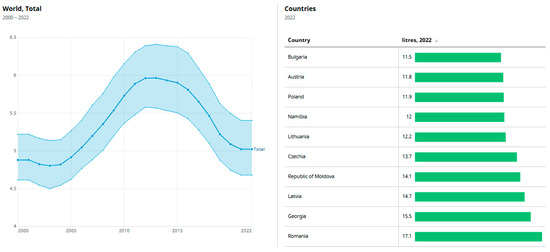
Figure 1.
Total amount of alcohol consumed per adult (15+ years) over a calendar year, in liters of pure alcohol [1]. Data from WHO (Indicators).
Although the liver is classically recognized as the primary target organ in alcohol-related morbidity, emerging research has accentuated the crucial role of the gastrointestinal (GI) tract, particularly the gut microbiome, in the initiation and progression of alcohol-related diseases [2].
1.1. Ethanol and Its Metabolic Impact on the GI Tract
Following oral ingestion, ethanol is rapidly absorbed in the stomach and proximal small intestine via simple diffusion and distributed throughout the body, including the distal GI tract, where its concentration mirrors that in systemic circulation [3]. Ethanol metabolism occurs via oxidative and non-oxidative pathways (Figure 2) [4].

Figure 2.
Oxidative and non-oxidative metabolism of ethanol. Data from Biorender [5].
Oxidative metabolism, mediated primarily by alcohol dehydrogenase (ADH) and cytochrome P450 2E1 (CYP2E1), yields acetaldehyde—a highly reactive and toxic intermediate implicated in tissue injury and carcinogenesis [6]. This is followed by conversion to acetate via aldehyde dehydrogenase (ALDH). While the liver carries out the majority of ethanol metabolism, significant enzymatic activity has also been observed in the epithelial cells of the small and large intestine [7]. The presence of metabolic machinery within the gut mucosa exposes the intestinal epithelium to locally generated acetaldehyde, particularly in the colon and rectum (Figure 3).

Figure 3.
Ethanol and its metabolic impact. Created with Biorender [5]. Data from Madigan et al. [8].
This localized metabolic activity is associated with the disruption of epithelial barrier function, or “leaky gut,” and has been shown to contribute to an increased risk of gastrointestinal cancers [7].
1.2. Alcohol-Induced Gut Barrier Dysfunction
One of the most deleterious effects of chronic ethanol consumption is the compromise of the gut epithelial barrier. Both ethanol and its primary metabolite, acetaldehyde, have been implicated in the disruption of tight junction proteins such as occludin and zonula occludens-1 (ZO-1), thereby increasing intestinal permeability [8]. Experimental studies in humans and rodents have demonstrated that both acute and chronic ethanol exposure leads to enhanced translocation of bacterial endotoxins such as lipopolysaccharides (LPSs) from the gut lumen into the portal circulation [9].
This breach of the epithelial barrier facilitates the entry of microbial-associated molecular patterns (MAMPs) into systemic circulation, which, in turn, activates hepatic Kupffer cells via toll-like receptors (e.g., TLR4), driving proinflammatory cascades that culminate in hepatic inflammation, fibrosis, and eventually alcoholic liver disease (ALD) [10].
1.3. Gut Microbiota Dysbiosis in Alcohol-Related Disease
The gut microbiota, a complex ecosystem of trillions of microorganisms, plays an indispensable role in host metabolism, immune modulation, and barrier integrity [11]. This microbial imbalance has been implicated in the pathogenesis of several immune-mediated and metabolic diseases [12], including multiple sclerosis, autoimmune hepatitis, rheumatoid arthritis, type 1 diabetes, colorectal cancer, and other systemic disorders (Figure 4).

Figure 4.
Microbial dysbiosis and its disease associations. Created with Biorender [5]. Data from Szychlinska et al. [13].
Chronic alcohol intake has been shown to significantly alter the composition, diversity, and function of the gut microbiome, a phenomenon broadly described as dysbiosis [10]. Both preclinical and clinical models have demonstrated ethanol-induced microbial shifts characterized by a decline in beneficial commensals, such as Faecalibacterium prausnitzii, Lactobacillus, and Bifidobacterium, accompanied by an overgrowth of potentially pathogenic genera such as Proteobacteria, Clostridium, and Fusobacterium [14,15].
Preclinical studies using ethanol-fed rodent models have revealed marked increases in bacterial overgrowth in the upper small intestine and significant alterations in the cecal microbiota, including elevated levels of Bacteroides and Verrucomicrobia alongside reduced Firmicutes [16]. These microbial shifts have been associated with suppressed expression of host antimicrobial peptides such as Reg3β and Reg3γ, exacerbating mucosal vulnerability and bacterial translocation [17]. In humans, ethanol consumption has similarly been linked to decreased fecal concentrations of butyrate-producing bacteria and short-chain fatty acids (SCFAs), which are critical for maintaining intestinal homeostasis and mucosal healing [18].
1.4. The Gut–Liver–Brain Axis: A Triangular Pathophysiological Circuit
Beyond its hepatic consequences, ethanol-induced dysbiosis and gut barrier dysfunction appear to play central roles in the systemic complications of alcohol use disorder [19]. The gut–liver axis is now expanded to include the brain, forming a complex gut–liver–brain axis [20], in which microbial dysbiosis, intestinal permeability, and inflammatory signaling converge to influence neuropsychiatric outcomes (Figure 5).
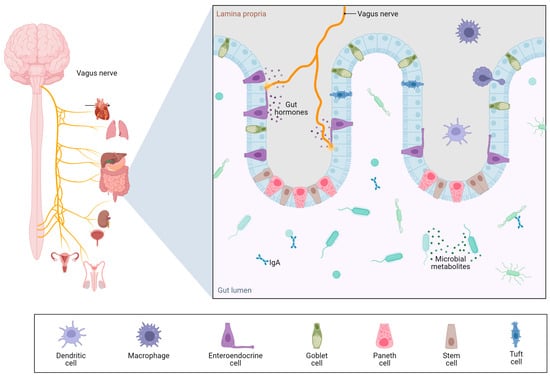
Figure 5.
Gut–brain axis. Created with Biorender [5]. Data from Gonzalez-Santana et al. [21].
Evidence indicates that microbial-derived products crossing a compromised gut barrier not only exacerbate liver injury but also reach the central nervous system (CNS), where they may trigger neuroinflammation and contribute to cognitive impairments observed in AUD [22]. Furthermore, ethanol-induced reductions in microbial synthesis of vitamins such as thiamine can lead to Wernicke–Korsakoff syndrome and other neurodegenerative manifestations [23].
1.5. Therapeutic Potential of Targeting the Gut Microbiota
Given the intimate interplay between ethanol metabolism, gut microbiota dysbiosis, and systemic inflammation, restoring microbial homeostasis has emerged as a promising therapeutic avenue. Several studies have demonstrated that administration of probiotics or fecal microbiota transplantation (FMT) from healthy donors can ameliorate ethanol-induced gut dysbiosis and its sequelae. In murine models, treatment with Lactobacillus rhamnosus or dietary oats improved microbial balance and intestinal barrier integrity, while in humans, probiotic supplementation increased beneficial bacterial counts and improved liver enzyme profiles [24].
These interventions hold promise not only for mitigating the progression of ALD but also for alleviating neurocognitive symptoms in patients with AUD [25]. The metabolomic profile of the gut microbiota, including SCFAs and bile acids, may also serve as biomarkers for disease severity and response to therapy [26].
Thus, ethanol-induced disruptions in gut microbiota composition and function constitute a key mechanism linking alcohol consumption to systemic diseases, particularly liver injury and neurocognitive disorders. Through alterations in microbial taxa, metabolite profiles, and intestinal permeability, ethanol sets the stage for a cascade of inflammatory and fibrotic events with implications that extend far beyond the liver. This meta-analysis seeks to consolidate current evidence on the bidirectional relationship between ethanol intake and gut microbiota dysbiosis with the aim of elucidating mechanistic pathways and evaluating the therapeutic potential of microbiota-targeted interventions [27].
Unlike prior reviews on ethanol-induced dysbiosis, our study integrates data from both human and animal studies to generate meta-aggregated effect sizes and highlight translational patterns. Additionally, we provide a direct comparative analysis between human and animal models and evaluate the impact of microbiota-targeted interventions, offering insights into mechanistic pathways and therapeutic potential that were not addressed in previous reviews.
2. Materials and Methods
2.1. Strategy for Searching the Literature
A systematic search was conducted using the PubMed database to identify original research articles evaluating the effects of ethanol exposure on gut microbiota in both human and animal studies. The search included combinations of MeSH terms and free-text keywords such as “ethanol”, “alcohol”, “gut microbiota”, “intestinal microbiome”, “dysbiosis”, “humans”, “mice”, “rats”, and “rodent models” [28]. Boolean operators “AND” and “OR” were applied to optimize the yield. Searches were limited to articles published in English [29]. No restrictions were placed on publication date.
The full selection process is detailed in the PRISMA flow diagram (Figure 6) [30].

Figure 6.
PRISMA flow diagram. Created with Biorender [5]. Data from www.prisma-statement.org, accessed on 12 June 2025 [30].
The search initially identified a total of 112 records, including 70 human studies and 42 animal studies. After the removal of duplicates (4 human, 1 animal), 107 records were screened by title. This was followed by abstract screening (humans: 54; animals: 30) and subsequent full-text review (humans: 33; animals: 25) to determine final eligibility. Based on predefined inclusion and exclusion criteria, 11 human studies and 19 animal studies were included in the final analysis.
2.2. Inclusion and Exclusion Criteria
Inclusion criteria were defined separately for human and animal studies:
- Human studies: eligible if they investigated individuals with chronic alcohol use or alcohol dependence, assessed gut microbiota composition using sequencing or molecular tools, and included a comparator group (e.g., healthy controls or non-drinkers).
- Animal studies: included if the animals were exposed to ethanol via drinking water, liquid diet, vapor exposure, or gavage and reported microbiota outcomes assessed via validated methods (e.g., 16S rRNA sequencing, qPCR).
- There were no geographical restrictions in study selection.
Exclusion criteria applied to both domains:
- Lack of a control group, absence of microbiota outcome data;
- Non-ethanol-related interventions (e.g., antibiotics alone, prebiotics);
- Language other than English;
- Limited methodological rigor;
- Inaccessible full text.
Specifically, 12 human titles and 14 animal titles were excluded during the title screening phase. Abstract-level exclusions included scope mismatches, inaccessible papers, language barriers, and insufficient study design. At the full-text level, 22 human and 6 animal studies were excluded primarily due to insufficient control, unclear ethanol effect attribution, or poor methodological quality.
Ethanol was selected as the focus of this analysis because it represents the predominant form of alcohol consumed globally and is the principal agent responsible for alcohol-related disease. Moreover, ethanol-based rodent models are well characterized and have been extensively validated to reproduce key features of alcohol-induced dysbiosis.
2.3. Screening and Data Extraction
All screening steps—titles, abstracts, and full texts—were performed independently by two reviewers, with disagreements resolved by discussion. Data extracted included population characteristics (species, strain, age, sex), ethanol exposure parameters (dose, route, and duration), microbiota analysis method (e.g., sequencing platform, region targeted), and key outcomes (diversity metrics, taxonomic shifts, functional alterations).
For human studies, additional variables, such as alcohol consumption history, diagnostic criteria, sample source (e.g., feces, mucosal biopsy), and reported comorbidities, were recorded.
The number of included studies was limited by the challenge of identifying randomized controlled trials and high-quality observational studies with sufficient methodological rigor and heterogeneity to allow for meaningful synthesis. Many studies were excluded due to the absence of control groups, inadequate microbiota outcome data, or failure to meet minimum quality criteria. The final 11 human and 19 animal studies therefore represent the best available evidence suitable for robust comparative analysis.
2.4. Quality Assessment
Animal studies were assessed using the SYRCLE risk of bias tool [31]. Most studies reported randomization, ethical compliance, and sample size estimates but often lacked details on blinding and environmental controls.
Human studies were evaluated using the Newcastle–Ottawa scale, focusing on study selection, comparability, and outcome assessment [32]. While most human studies adequately defined exposure and outcome, variability was noted in controlling for confounders such as diet and medication use.
2.5. Data Synthesis and Analysis
Given the diversity in experimental designs and microbiota outcome reporting, both narrative synthesis and quantitative comparison were employed. Microbial diversity (alpha and beta), shifts in major phyla (e.g., Firmicutes, Bacteroidetes, Proteobacteria), and functional signatures (e.g., SCFA production, bile acid metabolism) were synthesized descriptively.
Where applicable, summary measures such as mean differences and confidence intervals were calculated for comparative analysis between ethanol-exposed and control groups. Results from both human and animal studies were tabulated and analyzed separately to preserve biological relevance.
No funding was utilized in the design, conduct, analysis, or preparation of this article. All research activities were carried out independently by the authors without financial support from public or private institutions.
3. Results
Table 1 and Table 2 below present the core studies included in this meta-analysis, offering a structured overview of ethanol-induced gut microbiota alterations in both humans and animal models. Table 1 summarizes 11 human studies, highlighting participant characteristics, diagnostic categories, intervention types, and key microbiota outcomes. Table 2 compiles findings from 19 animal studies involving rodent models, detailing ethanol exposure protocols, microbial shifts, and associated physiological or behavioral effects.

Table 1.
Selected studies on humans.

Table 2.
Selected studies on animals.
3.1. Human Cohort Findings: Microbiota Diversity and Clinical Outcomes
To assess the impact of ethanol exposure on gut microbiota in humans, we included 11 eligible studies that met all inclusion criteria. These studies spanned various designs, including randomized controlled trials, observational studies, and interventional protocols involving fecal microbiota transplantation, probiotics, abstinence programs, or pharmaceutical agents.
3.1.1. Population and Study Characteristics
The selected studies involved a total of 702 participants aged between 18 to 65 years, with most subjects being male (Table 3). Participants included individuals diagnosed with AUD, ARC, AH, and related comorbidities. Control groups varied across studies, comprising healthy individuals, placebo groups, or standard-of-care cohorts (Figure 7).

Table 3.
Percentage breakdown by diagnosis; proportion of total participants across the 11 human studies.

Figure 7.
Breakdown of participant diagnoses across the 11 human studies.
3.1.2. Effect Sizes for Human Studies
Table 4 summarizes the calculated effect size estimates for the included human studies.

Table 4.
Effect size estimates for human studies evaluating interventions or outcomes related to alcohol use and microbiota.
Among the studies with sufficient data, Du et al. [33] demonstrated large negative effect sizes for cognitive outcomes (MoCA: d = −0.98; MMSE: d = −1.25), indicating worse performance in the AUD group compared to healthy controls. Dedon et al. [34] showed a small-to-moderate effect (d = 0.39) in favor of placebo for percent drinking reduction, while Bajaj et al. [36] reported a small effect (d = 0.29) favoring FMT over placebo for alcohol craving (ACQ-SF). Philips et al. [38] revealed a large effect (d = 0.69) for improved 90-day survival with FMT compared to high-dose probiotic infusion. Similarly, Zhang et al. [40] reported moderate-to-large effects for reductions in liver enzymes (AST: d = 0.66; γ-GT: d = 0.87) with BC99 treatment compared to placebo. Han et al. [43] observed a small effect size for serum LPS reduction with probiotics (d = 0.18). The remaining studies (Zhang et al. [35], Amadieu et al. [37], Muthiah et al. [39], Lang et al. [41], and Haas et al. [42]) did not provide sufficient outcome data to compute effect sizes.
In most cases, beneficial shifts, such as increased abundance of Lactobacillus or decreased Enterobacteriaceae, were observed during or immediately after the intervention period, with limited follow-up extending beyond 4–8 weeks (Table 5).

Table 5.
Summary of human studies based on type of intervention and primary microbiota findings.
As such, the long-term persistence of these effects remains uncertain. Notably, Wang et al. [62] provided limited post-treatment data suggesting partial reversion of microbial profiles, implying that colonization by introduced or promoted taxa may be transient in the absence of sustained intervention.
3.1.3. Microbiota Alterations
Human studies reported a consistent pattern of dysbiosis following ethanol exposure (Table 6):

Table 6.
Microbial changes by study (humans).
- Decreases: Ruminococcaceae, Lachnospiraceae, Faecalibacterium, Akkermansia, Bifidobacterium;
- Increases: Enterococcus, Streptococcus, Veillonella, Proteobacteria (especially Enterobacteriaceae) (Figure 8).
 Figure 8. Meta-aggregated taxonomic alterations across all human studies. Data from [33,34,35,36,37,38,39,40,41,42,43].
Figure 8. Meta-aggregated taxonomic alterations across all human studies. Data from [33,34,35,36,37,38,39,40,41,42,43].
Changes in microbial diversity (α-diversity and β-diversity) were often correlated with clinical severity, systemic inflammation (↑ IL-6, TNF-α, LBP), and psychological symptoms (↑ craving, ↓ cognition).
3.2. Animal Model Findings
The studies analyzed employed a diverse array of animal models, with mice being the predominant species (n = 13), particularly the C57BL/6J, BALB/c, and ICR strains. Rats were used in five studies, most commonly the Wistar and Sprague Dawley strains. One study uniquely utilized human fecal microbiota transferred into rats, bridging preclinical and translational paradigms.
Ethanol administration routes varied, with the Lieber–DeCarli liquid diet being the most commonly employed method for chronic exposure. Other models included drinking water supplementation (gradually increasing or fixed concentrations), oral gavage, vapor exposure, intermittent binge-like exposure, and ethanol combined with a high-fat diet (HFD). The ethanol doses ranged from 5% to 56%, with exposure durations from 4 weeks to 12 weeks, or longer for chronic use. One study mimicked acute exposure using a binge protocol, while another included subacute vapor exposure to simulate inhalation effects (Table 7, Figure 9).

Table 7.
Microbial changes by study (animals).

Figure 9.
Meta-aggregated taxonomic alterations across all animal studies. Data from [45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62].
A consistent observation across experimental models was the reduction in beneficial commensal bacteria. Notably, Lactobacillus species were significantly depleted in at least six studies, suggesting a reproducible pattern of sensitivity to ethanol’s disruptive effects. Other commensals, such as Akkermansia and Allobaculum, were frequently altered, with some interventions promoting their abundance. These bacteria are known for their roles in maintaining mucosal integrity, modulating immune responses, and supporting metabolic homeostasis. Additionally, short-chain fatty-acid-producing bacteria, including Butyricimonas and members of the Ruminococcaceae family, were found to decline in abundance. The loss of these SCFA producers likely contributes to impaired gut barrier function and heightened susceptibility to inflammation.
Conversely, ethanol exposure favored the overgrowth of potentially pathogenic or proinflammatory taxa. An increase in the phylum Proteobacteria, particularly Enterobacteriaceae family members, was a prominent finding, especially in the studies by Hendrikx et al. [53] and Chen et al. [54]. Similarly, other opportunistic bacteria, such as Helicobacter, Fusobacterium, and Escherichia coli, exhibited increased relative abundance under ethanol influence. Several studies also reported an elevated Firmicutes-to-Bacteroidetes ratio, most notably in the work by Daaz-Ubilla et al. [44].
Only Wang et al. [47] investigated fungal components, identifying ↑ Saccharomyces and Kurtzmaniella, ↓ Candida, and a significantly reduced fungal-to-bacterial ratio.
In parallel, ethanol-induced dysbiosis was frequently associated with compromised intestinal barrier integrity. Elevated levels of biomarkers like intestinal fatty acid binding protein (i-FABP) and LPS were observed, suggesting increased gut permeability and endotoxin translocation into systemic circulation. This barrier dysfunction likely contributes to the systemic inflammation and hepatic damage documented in multiple studies, including those by Daaz-Ubilla [44], Hendrikx [53], and Wang [47].
Table 8 highlights notable discrepancies in microbial responses to ethanol exposure between human and animal studies.

Table 8.
Discrepancies in microbial findings.
Akkermansia decreased in humans [34] but increased in several animal studies [47,56,57,59,61]. In his research, Yan et al. [63] mentions that this might be due to species-specific microbiome dynamics or the absence of confounding clinical factors in animal models. Similarly, Bacteroides and E. coli decreased in human studies [34,42,43] but increased in rodents [48,62]. Nguyen et al. [64] concluded that this might be because of various confounding factors ranging from diet to exposure to pathogens. Contrasting trends were also seen for Lachnospiraceae, Prevotella, and Roseburia.
3.2.1. Effect Sizes for Animal Model Studies
Table 9 presents the calculated effect size estimates for the 19 animal studies included in this analysis.

Table 9.
Effect size estimates for animal model studies assessing behavioral, biochemical, and microbiota-related outcomes in alcohol exposure models.
Several studies demonstrated very large effect sizes, particularly those measuring ethanol intake, liver injury markers, and behavioral outcomes. For instance, Díaz-Ubilla et al. [44], Wang et al. [47], Xia et al. [51], and Thoen et al. [58] reported Cohen’s d values > 4 for key outcomes such as ethanol intake and serum ALT. Behavioral assessments, including the open-field test, elevated plus maze, forced swim test, and tail suspension test, also consistently showed large effects (e.g., d > 1) in studies such as those by Xiao et al. [46], Xu et al. [45], and Wang et al. [62], indicating pronounced anxiety- and depression-like behaviors associated with alcohol exposure or fecal microbiota transplantation from alcohol-dependent subjects. Biochemical markers also revealed large differences across groups (Han et al. [55], Li et al. [48], Cunningham et al. [61]).
3.2.2. Intervention Outcomes
Several studies evaluated interventions aimed at reversing or mitigating alcohol-induced dysbiosis (Figure 10).

Figure 10.
Venn diagram showing differences and similarities in the change in intestinal microbiota between humans and animals. ↑ indicates an increase in bacterial abundance or clinical parameter; ↓ indicates a decrease.
Probiotic therapy: Lactobacillus casei supplementation reversed microbial shifts (↑ Lactobacillus, ↓ E. coli) and corrected iron metabolism disturbances by reducing ferritin and hepcidin and restoring transport proteins (DMT1, FPN1) [48]. In the study by Jiang et al. [59], co-treatment with probiotics partially restored the gut microbiota and improved barrier function.
Studies incorporating ethanol withdrawal showed partial or complete recovery of microbiota profiles. For example, Xia et al. [51] documented reversibility of SCFA-producing bacteria, while Yang Fan et al. [52] demonstrated colonic restoration of beneficial taxa and functional metabolic pathways after ethanol cessation.
FXR-deficient mice [53] exhibited worsened liver pathology and microbial imbalance, pointing to nuclear receptor pathways as therapeutic targets.
4. Discussion
This meta-analysis consolidates evidence from 11 human and 19 animal studies to highlight the consistent, ethanol-induced perturbations in gut microbiota composition, diversity, and function. Notably, both categories of studies demonstrated convergence on hallmark microbial changes, suggesting robust and translationally relevant biological patterns.
Multiple human studies (Table 1) have demonstrated consistent taxonomic alterations associated with ethanol exposure. Specifically, a significant reduction in Lactobacillus abundance was reported by Du et al. [33], Philips et al. [38], and Bajaj et al. [36]. Comparable findings were also observed in animal models, as evidenced by the results of Li et al. [48] and Yang et al. [55] (Table 2). These reductions are significant given Lactobacillus’s role in maintaining mucosal integrity and immune regulation. Consistent with our findings, Chancharoenthana et al. [65] reported that chronic alcohol exposure in mice suppressed Lactobacillus abundance and impaired tight junction expression, leading to increased gut permeability and endotoxemia.
Similarly, Akkermansia, a mucin-degrading genus linked to metabolic and gut barrier health, declined in both Dedon et al. [34] and Jiang et al. [59], while probiotic or dietary interventions restored its abundance. These trends align with observations by Wei et al. [66], who found that Akkermansia supplementation attenuated ethanol-induced steatosis and inflammation in murine models.
Microbial diversity also emerged as a central theme. Decreased alpha-diversity was reported in severe AUD cohorts [37,41], echoing findings in several animal models where chronic ethanol exposure reduced microbial richness [47,60]. Parallel work by Capurso et al. [67] supports these outcomes, showing that long-term alcohol use leads to ecological instability in the microbiome and favors pathogenic overgrowth.
A notable distinction between human and animal studies lies in the complexity of comorbid conditions. While animal models often isolate ethanol as the primary variable, human studies [36,38] contend with layered pathologies such as hepatic encephalopathy, cirrhosis, or psychiatric symptoms. Despite these confounders, both domains recorded elevated Proteobacteria, especially Enterobacteriaceae, a marker of dysbiosis and inflammation. This is consistent with findings by Smirnova et al. [68], who demonstrated increased Proteobacteria in alcoholic hepatitis patients and linked it to systemic inflammation.
A notable divergence was noted in the abundance of Escherichia coli between human and animal studies. In humans, a reduction in fecal E. coli counts has been consistently observed, particularly in hospitalized patients with alcoholic hepatitis, which may be attributed to prior exposure to antibiotics, altered bile acid profiles, or impaired mucosal immunity secondary to liver dysfunction. These factors can selectively suppress facultative anaerobes such as E. coli within the gut lumen. In contrast, rodent models exhibited an increase in E. coli abundance following chronic ethanol exposure. This discrepancy may be attributed to species-specific differences in intestinal physiology and microbiota resilience, as well as the more controlled experimental conditions in animal studies. In rodents, ethanol-induced disruption of tight junctions and increased intestinal permeability may facilitate colonization and expansion of E. coli and other opportunistic taxa, which thrive in the inflamed and oxygen-enriched microenvironment of a compromised gut barrier.
Interventional outcomes offer additional insights. In humans, FMT significantly improved microbial diversity and clinical endpoints [36,38], while in animals, similar benefits were seen with Lactobacillus casei or Clostridium butyricum supplementation [48,49]. Comparable benefits have been documented by Pu et al. [69], who reported that Clostridium butyricum ameliorated ethanol-induced gut and liver injury via TLR4/NF-κB pathway inhibition.
Another important factor contributing to variability in the findings is the heterogeneity in microbiota sequencing techniques and analysis pipelines across the included studies. While the majority of both human and animal studies utilized 16S rRNA gene sequencing, there was significant inconsistency in the specific hypervariable regions targeted (V3–V4, V4 alone, or V1–V3). Moreover, differences in DNA extraction methods, sequencing platforms, and downstream bioinformatics pipelines (OTU vs. ASV-based clustering, use of different reference databases) further complicate direct cross-study comparisons. A small number of studies employed shotgun metagenomic sequencing, offering higher resolution but introducing additional variability in functional and compositional analyses.
While several studies in this review reported neurobehavioral alterations associated with dysbiosis, it is important to note that most of these findings remain correlative. Direct evidence linking specific microbial metabolites to neurobehavioral outcomes is still limited. A few preclinical studies have demonstrated potential causality; for instance, SCFA supplementation has been shown to restore GABAergic signaling and ameliorate anxiety-like behavior in germ-free or antibiotic-treated rodents. However, such mechanistic insights have not yet been conclusively validated in human cohorts.
Observed behavioral outcomes associated with microbial alterations support the translational relevance of findings between preclinical and clinical studies. Rodents exposed to dysbiotic microbiota displayed depressive and anxiety-like behaviors [45,46], paralleling observations of increased craving and reduced cognition in AUD patients [34,37]. These findings are echoed by Nikel et al. [70], who identified correlations between gut dysbiosis and anxiety scores in AUD cohorts.
5. Limitations
One important limitation of this meta-analysis lies in the heterogeneity of control groups across included studies. Control cohorts ranged from healthy individuals with no alcohol exposure to placebo-treated or standard-of-care populations, introducing variability in baseline microbiota composition and systemic parameters. While this diversity does not invalidate the findings, it represents a potential source of bias that must be acknowledged in the interpretation of results. In addition, significant heterogeneity was observed in other aspects of study design, including ethanol dosing regimens, intervention durations, and microbiota analysis techniques. Specifically, studies employed differing routes and concentrations of ethanol administration (e.g., oral gavage vs. voluntary intake), and microbiota characterization ranged from 16S rRNA gene sequencing (with different hypervariable regions) to shotgun metagenomics.
6. Conclusions
This meta-analysis provides compelling evidence that ethanol exposure induces consistent and functionally significant alterations in the gut microbiota across both human and animal studies. Despite differences in methodology, host physiology, and experimental design, the findings converge on key microbial signatures: a depletion of beneficial commensals such as Faecalibacterium, Akkermansia, Lactobacillus, and Bifidobacterium, alongside an expansion of proinflammatory taxa like Proteobacteria, Enterococcus, Veillonella, and Escherichia-Shigella. These microbial alterations are accompanied by reduced diversity and a depletion of short-chain fatty-acid-producing organisms, which collectively contribute to compromised gut barrier integrity, increased intestinal permeability, and systemic inflammation.
In humans, dysbiosis was associated with alcohol use disorder, alcoholic hepatitis, cirrhosis, and neurocognitive symptoms such as increased craving and cognitive impairment. In animal models, microbial shifts were not only reproducible but also shown to mediate behavioral changes, including anxiety and depressive-like states.
Interventional studies further reinforce the modifiability of the gut microbiome. Probiotic supplementation, fecal microbiota transplantation, and ethanol abstinence were shown to restore microbial diversity, rebalance key taxa, and improve clinical or behavioral outcomes.
Collectively, these findings position the gut microbiota as a central mediator in alcohol-related pathophysiology, spanning hepatic, gastrointestinal, and neuropsychiatric domains. Future research should focus on precision microbiome interventions and integrative biomarker development to personalize treatment strategies for AUD and related disorders.
Author Contributions
Conceptualization, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H. and methodology, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H., I.T.T., A.D.N., E.R. and B.C., software, L.A., A.D., E.D., C.T., I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H. and validation, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H., I.T.T., A.D.N., E.R., B.C. and formal analysis L.A., A.D., E.D., C.T., I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H. and investigation, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H., I.T.T., A.D.N., E.R., B.C. and resources, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H. and data curation, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H. and writing—original draft preparation, L.A., A.D., E.D., C.T., I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H. and writing—review and editing, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H., visualization, L.A., A.D., E.D., C.T., I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H. and supervision, L.A., A.D., E.D., C.T., I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H., I.T.T., A.D.N., E.R., B.C. and project administration, L.A., A.D., E.D., C.T., I.P. (Ioana Popescu), D.E.T., A.N.T., D.M.A., A.H. and funding acquisition, L.A., A.D., E.D., C.T., I.P. (Ionela Preotesoiu), D.E.T., A.N.T., D.M.A., A.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- EE6F72A. Datadot. Available online: https://data.who.int/indicators/i/EF38E6A/EE6F72A (accessed on 12 June 2025).
- Jew, M.H.; Hsu, C.L. Alcohol, the gut microbiome, and liver disease. J. Gastroenterol. Hepatol. 2023, 38, 1205–1210. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maccioni, L.; Fu, Y.; Horsmans, Y.; Leclercq, I.; Stärkel, P.; Kunos, G.; Gao, B. Alcohol-associated bowel disease: New insights into pathogenesis. Egastroenterology 2023, 1, e100013. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lieber, C.S. Alcohol metabolism. In Elsevier eBooks; Elsevier: Amsterdam, The Netherlands, 2004; pp. 28–32. [Google Scholar] [CrossRef]
- Scientific Image and Illustration Software|BioRender. Available online: https://www.biorender.com/ (accessed on 12 June 2025).
- Chirila, S.; Hangan, T.; Gurgas, L.; Costache, M.G.; Vlad, M.A.; Nitu, B.F.; Bittar, S.M.; Craciun, A.; Condur, L.; Bjørklund, G. Pharmacy-Based Influenza Vaccination: A Study of Patient Acceptance in Romania. Risk Manag. Healthc. Policy 2024, 17, 1005–1013. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Surdu, T.-V.; Surdu, M.; Surdu, O.; Franciuc, I.; Tucmeanu, E.-R.; Tucmeanu, A.-I.; Serbanescu, L.; Tica, V.I. Microvascular Responses in the Dermis and Muscles After Balneotherapy: Results from a Prospective Pilot Histological Study. Water 2025, 17, 1830. [Google Scholar] [CrossRef]
- Madigan. Brock Biology of Microorganisms: (International Edition): With How to Write about Biology; Prentice Hall: Hoboken, NJ, USA, 2003. [Google Scholar]
- Voiosu, T.; Voiosu, A.; Danielescu, C.; Popescu, D.; Puscasu, C.; State, M.; Chiricuţă, A.; Mardare, M.; Spanu, A.; Bengus, A.; et al. Unmet needs in the diagnosis and treatment of Romanian patients with bilio-pancreatic tumors: Results of a prospective observational multicentric study. Rom. J. Intern. Med. 2021, 59, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Bansal, M.B. Role of kupffer cells in driving hepatic inflammation and fibrosis in HIV infection. Front. Immunol. 2020, 11, 1086. [Google Scholar] [CrossRef] [PubMed]
- Colella, M.; Charitos, I.A.; Ballini, A.; Cafiero, C.; Topi, S.; Palmirotta, R.; Santacroce, L. Microbiota revolution: How gut microbes regulate our lives. World J. Gastroenterol. 2023, 29, 4368–4383. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mousa, W.K.; Chehadeh, F.; Husband, S. Microbial dysbiosis in the gut drives systemic autoimmune diseases. Front. Immunol. 2022, 13, 906258. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Szychlinska, M.A.; Di Rosa, M.; Castorina, A.; Mobasheri, A.; Musumeci, G. A correlation between intestinal microbiota dysbiosis and osteoarthritis. Heliyon 2019, 5, e01134. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sosnowski, K.; Przybyłkowski, A. Ethanol-induced changes to the gut microbiome compromise the intestinal homeostasis: A review. Gut Microbes 2024, 16, 2393272. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Islam, M.M.; Mahbub, N.U.; Hong, S.; Chung, H. Gut bacteria: An etiological agent in human pathological conditions. Front. Cell. Infect. Microbiol. 2024, 14, 1291148. [Google Scholar] [CrossRef]
- Daniel-MacDougall, C. How Does Alcohol Affect the Microbiome? Available online: https://www.mdanderson.org/cancerwise/how-does-alcohol-affect-the-microbiome.h00-159696756.html (accessed on 12 June 2025).
- Sugisawa, E.; Kondo, T.; Kumagai, Y.; Kato, H.; Takayama, Y.; Isohashi, K.; Shimosegawa, E.; Takemura, N.; Hayashi, Y.; Sasaki, T.; et al. Nociceptor-derived Reg3γ prevents endotoxic death by targeting kynurenine pathway in microglia. Cell Rep. 2022, 38, 110462. [Google Scholar] [CrossRef] [PubMed]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277, Erratum in Front. Immunol. 2019, 10, 1486. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Koutromanos, I.; Legaki, E.; Gazouli, M.; Vasilopoulos, E.; Kouzoupis, A.; Tzavellas, E. Gut microbiome in alcohol use disorder: Implications for health outcomes and therapeutic strategies-a literature review. World J. Methodol. 2024, 14, 88519. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ashique, S.; Mohanto, S.; Ahmed, M.G.; Mishra, N.; Garg, A.; Chellappan, D.K.; Omara, T.; Iqbal, S.; Kahwa, I. Gut-brain axis: A cutting-edge approach to target neurological disorders and potential synbiotic application. Heliyon 2024, 10, e34092. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Santana, A.; Diaz Heijtz, R. Bacterial Peptidoglycans from Microbiota in Neurodevelopment and Behavior. Trends Mol. Med. 2020, 26, 729–743. [Google Scholar] [CrossRef] [PubMed]
- Di Vincenzo, F.; Del Gaudio, A.; Petito, V.; Lopetuso, L.R.; Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: A narrative review. Intern. Emerg. Med. 2024, 19, 275–293. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kalapatapu, N.; Skinner, S.G.; D’Addezio, E.G.; Ponna, S.; Cadenas, E.; Davies, D.L. Thiamine Deficiency and Neuroinflammation Are Important Contributors to Alcohol Use Disorder. Pathophysiology 2025, 32, 34. [Google Scholar] [CrossRef]
- Pisarello, M.J.L.; Marquez, A.; Chaia, A.P.; Babot, J.D. Targeting gut health: Probiotics as promising therapeutics in alcohol-related liver disease management. AIMS Microbiol. 2025, 11, 410–435. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Anouti, A.; Kerr, T.A.; Mitchell, M.C.; Cotter, T.G. Advances in the management of alcohol-associated liver disease. Gastroenterol. Rep. 2024, 12, goae097. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jiang, Y.; Qu, Y.; Shi, L.; Ou, M.; Du, Z.; Zhou, Z.; Zhou, H.; Zhu, H. The role of gut microbiota and metabolomic pathways in modulating the efficacy of SSRIs for major depressive disorder. Transl. Psychiatry 2024, 14, 493. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rathore, K.; Shukla, N.; Naik, S.; Sambhav, K.; Dange, K.; Bhuyan, D.; Imranul Haq, Q.M. The Bidirectional Relationship Between the Gut Microbiome and Mental Health: A Comprehensive Review. Cureus 2025, 17, e80810. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- DeMars, M.M.; Perruso, C. MeSH and text-word search strategies: Precision, recall, and their implications for library instruction. J. Med. Libr. Assoc. 2022, 110, 23–33. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- MacFarlane, A.; Russell-Rose, T.; Shokraneh, F. Search strategy formulation for systematic reviews: Issues, challenges and opportunities. Intell. Syst. Appl. 2022, 15, 200091. [Google Scholar] [CrossRef]
- PRISMA 2020 Flow Diagram—PRISMA Statement. PRISMA Statement. Available online: https://www.prisma-statement.org/prisma-2020-flow-diagram (accessed on 12 June 2025).
- Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE). Available online: https://norecopa.no/3r-guide/systematic-review-centre-for-laboratory-animal-experimentation-syrcle (accessed on 12 June 2025).
- Ottawa Hospital Research Institute. Copyright 2011 Ottawa Hospital Research Institute. All Rights Reserved. Available online: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 12 June 2025).
- Du, Y.; Li, L.; Gong, C.; Li, T.; Xia, Y. The diversity of the intestinal microbiota in patients with alcohol use disorder and its relationship to alcohol consumption and cognition. Front. Psychiatry 2022, 13, 1054685. [Google Scholar] [CrossRef]
- Dedon, L.R.; Yuan, H.; Chi, J. Hu, H.; Arias, A.J.; Covault, J.M.; Zhou, Y. Baseline gut microbiome and metabolites are correlated with changes in alcohol consumption in participants in a randomized Zonisamide clinical trial. Sci. Rep. 2025, 15, 10486. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, R.; Deng, H.; Cui, P.; Li, C.; Yang, F.; Leong Bin Abdullah, M.F.I. Research protocol of the efficacy of probiotics for the treatment of alcohol use disorder among adult males: A comparison with placebo and acceptance and commitment therapy in a randomized controlled trial. PLoS ONE 2023, 18, e0294768. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bajaj, J.S.; Gavis, E.A.; Fagan, A.; Wade, J.B.; Thacker, L.R.; Fuchs, M.; Patel, S.; Davis, B.; Meador, J.; Puri, P.; et al. A Randomized Clinical Trial of Fecal Microbiota Transplant for Alcohol Use Disorder. Hepatology 2021, 73, 1688–1700. [Google Scholar] [CrossRef] [PubMed]
- Amadieu, C.; Ahmed, H.; Leclercq, S.; Koistinen, V.; Leyrolle, Q.; Stärkel, P.; Bindels, L.B.; Layé, S.; Neyrinck, A.M.; Kärkkäinen, O.; et al. Effect of inulin supplementation on fecal and blood metabolome in alcohol use disorder patients: A randomised, controlled dietary intervention. Clin. Nutr. ESPEN 2025, 66, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Philips, C.A.; Ahamed, R.; Oommen, T.T.; Nahaz, N.; Tharakan, A.; Rajesh, S.; Augustine, P. Clinical outcomes and associated bacterial and fungal microbiota changes after high dose probiotic therapy for severe alcohol-associated hepatitis: An observational study. Medicine 2024, 103, e40429. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Muthiah, M.D.; Smirnova, E.; Puri, P.; Chalasani, N.; Shah, V.H.; Kiani, C.; Taylor, S.; Mirshahi, F.; Sanyal, A.J. Development of Alcohol-Associated Hepatitis Is Associated With Specific Changes in Gut-Modified Bile Acids. Hepatol. Commun. 2022, 6, 1073–1089. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, J.; Li, C.; Duan, M.; Qu, Z.; Wang, Y.; Dong, Y.; Wu, Y.; Fang, S.; Gu, S. The Improvement Effects of Weizmannia coagulans BC99 on Liver Function and Gut Microbiota of Long-Term Alcohol Drinkers: A Randomized Double-Blind Clinical Trial. Nutrients 2025, 17, 320. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lang, S.; Fairfied, B.; Gao, B.; Duan, Y.; Zhang, X.; Fouts, D.E.; Schnabl, B. Changes in the fecal bacterial microbiota associated with disease severity in alcoholic hepatitis patients. Gut Microbes 2020, 12, 1785251. [Google Scholar] [CrossRef] [PubMed Central]
- Haas, E.A.; Saad, M.J.A.; Santos, A.; Vitulo, N.; Lemos, W.J.F.; Martins, A.M.A.; Picossi, C.R.C.; Favarato, D.; Gaspar, R.S.; Magro, D.O.; et al. A red wine intervention does not modify plasma trimethylamine N-oxide but is associated with broad shifts in the plasma metabolome and gut microbiota composition. Am. J. Clin. Nutr. 2022, 116, 1515–1529. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Han, S.H.; Suk, K.T.; Kim, D.J.; Kim, M.Y.; Baik, S.K.; Kim, Y.D.; Cheon, G.J.; Choi, D.H.; Ham, Y.L.; Shin, D.H.; et al. Effects of probiotics (cultured Lactobacillus subtilis/Streptococcus faecium) in the treatment of alcoholic hepatitis: Randomized-controlled multicenter study. Eur. J. Gastroenterol. Hepatol. 2015, 27, 1300–1306. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Ubilla, M.; Figueroa-Valdés, A.I.; Tobar, H.E.; Quintanilla, M.E.; Díaz, E.; Morales, P.; Berríos-Cárcamo, P.; Santapau, D.; Gallardo, J.; de Gregorio, C.; et al. Gut Microbiota-Derived Extracellular Vesicles Influence Alcohol Intake Preferences in Rats. J. Extracell. Vesicles 2025, 14, e70059. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Xu, Z.; Wang, C.; Dong, X.; Hu, T.; Wang, L.; Zhao, W.; Zhu, S.; Li, G.; Hu, Y.; Gao, Q.; et al. Chronic alcohol exposure induced gut microbiota dysbiosis and its correlations with neuropsychic behaviors and brain BDNF/Gabra1 changes in mice. Biofactors 2019, 45, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.W.; Ge, C.; Feng, G.X.; Li, Y.; Luo, D.; Dong, J.L.; Li, H.; Wang, H.; Cui, M.; Fan, S.J. Gut microbiota modulates alcohol withdrawal-induced anxiety in mice. Toxicol. Lett. 2018, 287, 23–30. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, G.; Liu, Q.; Guo, L.; Zeng, H.; Ding, C.; Zhang, W.; Xu, D.; Wang, X.; Qiu, J.; Dong, Q.; et al. Gut Microbiota and Relevant Metabolites Analysis in Alcohol Dependent Mice. Front. Microbiol. 2018, 9, 1874. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, X.; Liang, H. Effects of Lactobacillus casei on Iron Metabolism and Intestinal Microflora in Rats Exposed to Alcohol and Iron. Turk. J. Gastroenterol. 2022, 33, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, H.; Lin, X.; Liu, J.; Feng, Y.; Bai, Y.; Liang, H.; Hu, T.; Wu, Z.; Lai, J.; et al. Gut microbiota dysbiosis induced by alcohol exposure in pubertal and adult mice. mSystems 2024, 9, e0136624. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yi, S.; Zhang, G.; Liu, M.; Yu, W.; Cheng, G.; Luo, L.; Ning, F. Citrus Honey Ameliorates Liver Disease and Restores Gut Microbiota in Alcohol-Feeding Mice. Nutrients 2023, 15, 1078. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Xia, T.; Duan, W.; Zhang, Z.; Li, S.; Zhao, Y.; Geng, B.; Zheng, Y.; Yu, J.; Wang, M. Polyphenol-rich vinegar extract regulates intestinal microbiota and immunity and prevents alcohol-induced inflammation in mice. Food Res. Int. 2021, 140, 110064. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Ya-E, Z.; Ji-Dong, W.; Yu-Fan, L.; Ying, Z.; Ya-Lun, S.; Meng-Yu, M.; Rui-Ling, Z. Comparison of Microbial Diversity and Composition in Jejunum and Colon of the Alcohol-dependent Rats. J. Microbiol. Biotechnol. 2018, 28, 1883–1895. [Google Scholar] [CrossRef] [PubMed]
- Hendrikx, T.; Duan, Y.; Wang, Y.; Oh, J.H.; Alexander, L.M.; Huang, W.; Stärkel, P.; Ho, S.B.; Gao, B.; Fiehn, O.; et al. Bacteria engineered to produce IL-22 in intestine induce expression of REG3G to reduce ethanol-induced liver disease in mice. Gut 2019, 68, 1504–1515. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, P.; Miyamoto, Y.; Mazagova, M.; Lee, K.C.; Eckmann, L.; Schnabl, B. Microbiota Protects Mice Against Acute Alcohol-Induced Liver Injury. Alcohol. Clin. Exp. Res. 2015, 39, 2313–2323. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, F.; Wei, J.; Shen, M.; Ding, Y.; Lu, Y.; Ishaq, H.M.; Li, D.; Yan, D.; Wang, Q.; Zhang, R. Integrated Analyses of the Gut Microbiota, Intestinal Permeability, and Serum Metabolome Phenotype in Rats with Alcohol Withdrawal Syndrome. Appl. Environ. Microbiol. 2021, 87, e0083421. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Xue, M.; Liu, Y.; Lyu, R.; Ge, N.; Liu, M.; Ma, Y.; Liang, H. Protective effect of aplysin on liver tissue and the gut microbiota in alcohol-fed rats. PLoS ONE 2017, 12, e0178684. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mittal, A.; Choudhary, N.; Kumari, A.; Yadav, K.; Maras, J.S.; Sarin, S.K.; Sharma, S. Protein supplementation differentially alters gut microbiota and associated liver injury recovery in mouse model of alcohol-related liver disease. Clin. Nutr. 2025, 46, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Thoen, R.U.; Longo, L.; Leonhardt, L.C.; Pereira, M.H.M.; Rampelotto, P.H.; Cerski, C.T.S.; Álvares-da-Silva, M.R. Alcoholic liver disease and intestinal microbiota in an experimental model: Biochemical, inflammatory, and histologic parameters. Nutrition 2023, 106, 111888. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liu, Y.; Gao, M.; Xue, M.; Wang, Z.; Liang, H. Nicotinamide riboside alleviates alcohol-induced depression-like behaviours in C57BL/6J mice by altering the intestinal microbiota associated with microglial activation and BDNF expression. Food Funct. 2020, 11, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yasuda, K.; Gilmore, R.A.; Westmoreland, S.V.; Platt, D.M.; Miller, G.M.; Vallender, E.J. Alcohol-induced changes in the gut microbiome and metabolome of rhesus macaques. Psychopharmacology 2019, 236, 1531–1544. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cunningham, K.C.; Smith, D.R.; Villageliú, D.N.; Ellis, C.M.; Ramer-Tait, A.E.; Price, J.D.; Wyatt, T.A.; Knoell, D.L.; Samuelson, M.M.; Molina, P.E.; et al. Human Alcohol-Microbiota Mice have Increased Susceptibility to Bacterial Pneumonia. Cells 2023, 12, 2267. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, C.; Yan, J.; Du, K.; Liu, S.; Wang, J.; Wang, Q.; Zhao, H.; Li, M.; Yan, D.; Zhang, R.; et al. Intestinal microbiome dysbiosis in alcohol-dependent patients and its effect on rat behaviors. mBio 2023, 14, e0239223. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yan, J.; Sheng, L.; Li, H. Akkermansia muciniphila: Is it the Holy Grail for ameliorating metabolic diseases? Gut Microbes 2021, 13, 1984104. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nguyen, T.L.; Vieira-Silva, S.; Liston, A.; Raes, J. How informative is the mouse for human gut microbiota research? Dis. Model. Mech. 2015, 8, 1–16. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chancharoenthana, W.; Kamolratanakul, S.; Udompornpitak, K.; Wannigama, D.L.; Schultz, M.J.; Leelahavanichkul, A. Alcohol-induced gut permeability defect through dysbiosis and enterocytic mitochondrial interference causing pro-inflammatory macrophages in a dose dependent manner. Sci. Rep. 2025, 15, 14710. [Google Scholar] [CrossRef]
- Wei, L.; Pan, Y.; Guo, Y.; Zhu, Y.; Jin, H.; Gu, Y.; Li, C.; Wang, Y.; Lin, J.; Chen, Y.; et al. Symbiotic combination of Akkermansia muciniphila and inosine alleviates alcohol-induced liver injury by modulating gut dysbiosis and immune responses. Front. Microbiol. 2024, 15, 1355225. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Capurso, G.; Lahner, E. The interaction between smoking, alcohol and the gut microbiome. Best. Pract. Res. Clin. Gastroenterol. 2017, 31, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, E.; Puri, P.; Muthiah, M.D.; Daitya, K.; Brown, R.; Chalasani, N.; Liangpunsakul, S.; Shah, V.H.; Gelow, K.; Siddiqui, M.S.; et al. Fecal Microbiome Distinguishes Alcohol Consumption From Alcoholic Hepatitis But Does Not Discriminate Disease Severity. Hepatology 2020, 72, 271–286. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pu, W.; Zhang, H.; Zhang, T.; Guo, X.; Wang, X.; Tang, S. Inhibitory effects of Clostridium butyricum culture and supernatant on inflammatory colorectal cancer in mice. Front. Immunol. 2023, 14, 1004756. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nikel, K.; Stojko, M.; Smolarczyk, J.; Piegza, M. The Impact of Gut Microbiota on the Development of Anxiety Symptoms—A Narrative Review. Nutrients 2025, 17, 933. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).