Abstract
Staphylococcus aureus is a major foodborne pathogen associated with contamination of dairy and meat products, posing a persistent challenge to food safety due to its biofilm formation and resistance to multiple antibiotics. In this review, we summarize recent advances in the use of bacteriophages and phage-derived endolysins as targeted biocontrol agents against S. aureus in food systems. Bacteriophages exhibit host specificity and self-replicating capacity, while endolysins provide rapid lytic activity, minimal resistance development, and effectiveness against biofilm-embedded cells. Studies demonstrate significant microbial reductions in milk, cheese, and meat matrices, although factors such as pH, salt, and fat content can influence their efficacy. The integration of these biocontrol tools into food preservation represents a sustainable and safe alternative to conventional antimicrobials. Finally, we discuss current limitations and the need for optimizing formulations, stability, and regulatory frameworks to facilitate the adoption of phage and endolysin-based products in the food industry.
1. Introduction
Staphylococcus aureus is a Gram-positive anaerobic bacterium that groups in clusters and is β-hemolytic, catalase and coagulase-positive. It is widely distributed in various environments and present in the microbial flora of humans, which increases the risk of its dissemination [1]. It is one of the etiological agents frequently found in clinical settings and food processing plants. Furthermore, it produces various enterotoxins that are heat stable, capable of forming biofilms and evolving into strains resistant to multiple drugs such as antibiotics, such as methicillin-resistant S. aureus strains (MRSA) and Vancomycin-Resistant S. aureus (VRSA) [2].
MRSA and VRSA exhibit a high mutation rate and adaptation related to a short duplication period in the environment in which they are found. MRSA causes ten times more infections than all multi-resistant Gram-negative pathogens combined [3]. According to the report published by the European Food Safety Authority and the European Centre for Disease Prevention and Control, S. aureus is one of the main pathogens that caused hospitalizations due to foodborne outbreaks in 36 European countries in 2018 [4]. Therefore, prevention of food contamination by S. aureus has generated growing interest in different areas, such as the food industry [5].
Among the traditional treatments used to control S. aureus are antibiotics, resins, essential oils, and peptides with antimicrobial properties [6]. Currently, one of the proposed alternatives to deal with the emergence of multi-resistant strains is the use of bacteriophages. Bacteriophages are natural antimicrobials that exist widely in the environment. They can overcome the barriers generated by bacterial defense mechanisms and have a high specificity to bind to their host through receptor-binding proteins (RBPs) that recognize specific molecules present on the surface of bacteria. The high specificity of bacteriophages allows them to attack pathogenic bacteria without affecting the microbiota [7,8,9].
Bacteriophages can be lytic or lysogenic, with the difference being found in the cycle they use to infect the bacterial host. In the lytic cycle, holins perforate the cytoplasmic membrane of bacteria, thus generating access to the peptidoglycan so that endolysins can degrade the bacterial cell wall, lyse the host, and release new bacteriophages into the external environment [10]. In the lysogenic cycle, viral genetic material is incorporated into the bacterial chromosome, with which it replicates and then is transferred to new bacteria without lysis of the host. During the phage-host interaction, virulent genes can be expressed, and the lysogenic life cycle can be converted to a lytic cycle [11,12]. In bacteriophage treatment to control bacterial contamination, the use of lysogenic bacteriophages is avoided due to the possibility of horizontal transfer of resistance or virulence genes to other bacteria lacking these genes [13].
Most S. aureus bacteriophages are lysogenic. They have been identified in the DNA of the microorganism, contributing to bacterial resistance due to the encoding of virulence factors. However, it is important to mention that exclusively lytic phages can also be found [14]. Bacteriophage JD419 for the biocontrol of S. aureus strains N315, MR-84, and MS-41, which was found to be a lysogenic phage by genomic analysis, had a latency period of 50 min and burst size of 33 PFU/infected cell; therefore, to improve its bactericidal activity and avoid genes related to the lysogenic cycle, it is necessary to make modifications through genetic engineering [15].
On the other hand, endolysins produced by bacteriophages that infect S. aureus generally have catalytic domains, such as an N-terminal, cysteine, histidine-dependent amidohydrolase/peptidase domain and a central N-acetylmuramoyl-l-alanine domain [16]. Endolysins have been used for the biocontrol of S. aureus in vitro and in foods contaminated with the microorganism, mainly in meat and dairy products. The endolysin LysSAP27 obtained from the phage vB_SauS-SAP27 (ϕSAP27), which had previously shown little efficiency in inhibiting the growth of S. aureus, was used in milk samples contaminated with S. aureus ATCC 6538, where biological control could be determined during treatment [17].
The use of bacteriophages and phage-derived endolysins represents a potential alternative for the biocontrol of S. aureus strains in food, which cause food spoilage and foodborne illnesses. Previous reviews on S. aureus bacteriophages have reported their genomic and morphological characteristics and their efficiency in the treatment of skin infections [18,19]. Therefore, this review focuses on the applications of bacteriophages and endolysins for the biocontrol of S. aureus in food, the characteristics of the microorganism’s bacteriophages, and the importance of endolysins in the food industry. Additionally, it covers commercial phage products for the biocontrol of S. aureus and the main limitations of S. aureus bacteriophages and endolysins in their application in food products.
2. S. aureus in the Food Industry
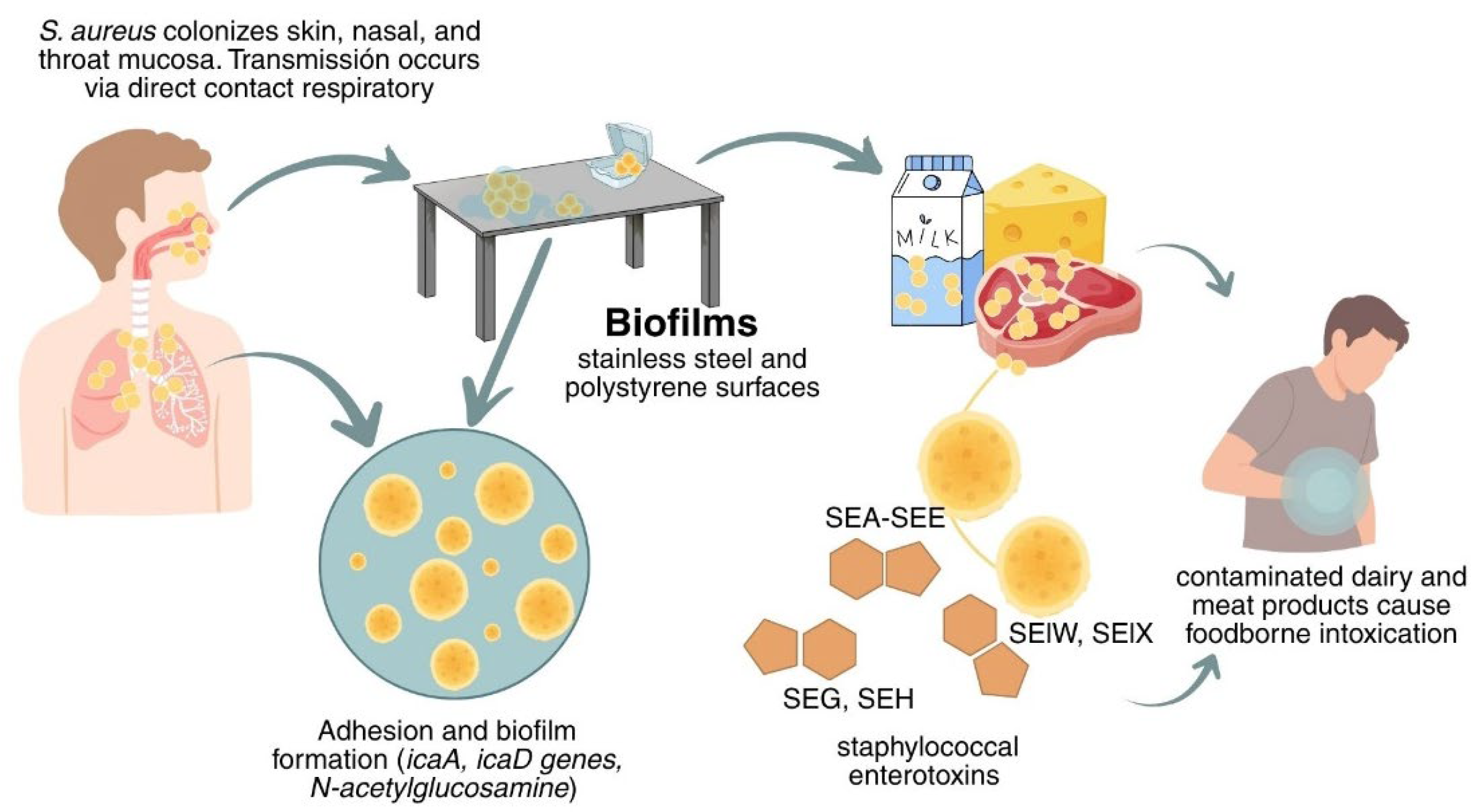
S. aureus is a pathogen that can be transmitted through the consumption of food contaminated during preparation or processing, either by direct contact or by respiratory secretions from food workers. It grows at temperatures between 7 and 48.5 °C, pH 4.2 and 9.3, and aw as low as 0.83; it grows optimally near 30–37 °C, pH 7.0–7.5, and aw 0.99, conditions frequently encountered in many food matrices [20,21,22].
Specific processing conditions (temperature, salt, and nutrient availability) can promote S. aureus growth and toxin expression (leukocidins, hemolysins, and staphylococcal enterotoxins, classical: SEA-SEE, non-classical: SEG, SHE; and enterotoxin-like: SElW, SElX, etc.), which underlie host survival and staphylococcal food poisoning with emesis [22,23,24]. These enterotoxins are pyrogenic and highly resistant to acidic pH, heat, and proteolysis, so the risk may persist even after certain treatments [25,26].
S. aureus has the ability to adhere, colonize, and form biofilms throughout the food production chain. This bacterium exhibits a strong adhesion capacity to various surfaces, particularly stainless steel and polystyrene. The extensive use of polystyrene in food processing environments increases the risk of recurrent cross-contamination [27,28]. Biofilm development in S. aureus is largely regulated by the intracellular adhesion (ica) locus, which encodes enzymes involved in the synthesis of the polysaccharide intercellular adhesin (PIA). The icaA and icaD genes play a central role in the process, catalyzing the polymerization of N-acetylglucosamine, the main exopolysaccharide that mediates intracellular adhesion and biofilm accumulation. The expression of the ica operon is further modulated by global regulators such as sarA, which enhances biofilm formation, and agr, which typically represses it in favor of acute virulence. Nevertheless, biofilm mass can vary even among strains harboring the ica locus, suggesting the involvement of ica-independent mechanisms, including surface proteins such as protein A and fibronectin-binding proteins [29]. Strains of S. aureus isolated from foods, processing facilities, and food handlers have demonstrated the ability to form biofilms on both stainless steel and polystyrene surfaces [30].
Infections and staphylococcal food poisoning linked to dairy and meat products contaminated with S. aureus have been reported, often associated with poor hygiene during milking, handling, transportation, and storage. These foods can serve as vehicles for transmission from farm to consumer; therefore, preventing and controlling S. aureus outbreaks along the food chain is essential [31,32]. Figure 1 shows the transmission route and the key problem points associated with S. aureus contamination in the industry.
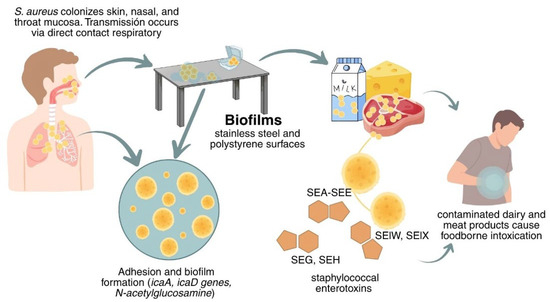
Figure 1.
Transmission and persistence of Staphylococcus aureus.
3. Characteristics of S. aureus Bacteriophages
Staphylococcus bacteriophages have an icosahedral capsid and a tail, carry linear double-stranded DNA, and belong to the class Caudoviricetes according to the recent update of the International Committee on Taxonomy of Viruses (ICTV). Electron microscopy reveals tail morphologies that correlated with typical genome sizes: 16–18 kb with short tails, 39–43 kb with long non-contractile tails, and 120–140 kb with long contractile tails, often with double sheath [19,33].
Prophages in S. aureus have been reported with a 39–43 kb genome and a long non-contractile tail. Their genomes display a modular organization, including lysogeny, DNA replication, transcription regulation, packaging, and head, tail, and lysis modules [34]. The lysogeny module encodes integrase (int) genes that recognize attP (phage) and attB (bacteria) sites and mediate unidirectional site-specific integration of the phage genome into the bacterial chromosome [35]. Prophage carriage can modulate host fitness, pathogenicity, and survival [36].
Among the lysogenic bacteriophages of S. aureus most widely studied are ϕ11 and ϕ80α, which show high transduction efficiency and are used for genetic engineering between S. aureus strains [37]. Phages in the same group have also been reported to carry lysogeny genes within the DNA regulation/manipulation module but lack the cI gene; consequently, they do not establish lysogeny and do not mediate horizontal gene transfer to S. aureus strains. The SA97 phage encodes an integrase (SA97_040); however, no virulence or drug-resistance genes were detected, supporting its use as a biocontrol agent against S. aureus [38].
On the other hand, bacteriophages belonging to group A represent a highly conserved lineage in terms of nucleotide and amino acid homology, lytic infection mechanism, genome size, GC content (27–29%), and predicted gene number (20–29%). Approximately 60% of their genes are associated with essential functions such as DNA replication (DNA-binding proteins, DNA polymerase), virion morphogenesis (DNA packaging, tail fibers, collar, and major capsid proteins), and host cell lysis (holin and endolysin) [36]. These phages exhibit remarkable genome compaction (<20 kb) and a streamlined genetic organization primarily dedicated to replication and structural functions, leaving minimal space for accessory or virulence-related genes. Their compact and well-defined genomes, together with a strictly lytic lifestyle, restrict uncharacterized regions and mobile genetic elements, thereby reducing the potential for horizontal gene transfer and enhancing their biosafety for therapeutic and biocontrol applications [39]. Among the most extensively studied members of this group are phages S13′ and S24-1, which have been effectively used against infections caused by S. aureus strains [40].
Long-tailed contractile staphylococcal phages encode 200–250 open reading frames and several tRNA genes, showing high nucleotide similarity (88.3–99.9% identity). They carry linear dsDNA of approximately 127–145 kbp with direct terminal repeats; notable examples include Stau2, StAP1, vB_SauM_Remus, vB_SauM_Romulus, SA11, and qdsa001, with high average nucleotide identity values (>95%) and shared gene content (>77%) [36]. Their genomes display a modular organization encompassing morphogenesis, cell lysis, nucleic-acid metabolism, and host metabolic reprogramming [41]. Infections typically show average latency periods of 30–50 min [42]. Table 1 below shows S. aureus bacteriophages and their main characteristics.

Table 1.
Staphylococcus aureus-related phages.
4. Biocontrol of S. aureus with Bacteriophages
In recent years, various investigations have been carried out where the biocontrol of S. aureus has been evaluated, especially in antibiotic-resistant strains. Specifically using lytic bacteriophages classified in groups A and C, which exhibit broader host ranges than those with a lysogenic nature (group B) [49]. Among the highlighted factors that have been evaluated for the biocontrol of S. aureus are the characterization of bacteriophages, the optimization of temperature conditions, and the time in which maximum reductions in the bacteria are generated, the MOI of the bacteriophages used to lyse S. aureus strains.
The vB_SauS_IMEP5 phage showed a 30 min latency and burst size of 272 PFU/infected cell; its lytic activity against S. aureus (6.6 × 108 CFU/mL) was tested across MOIs 10−4–100, with MOI 10−3 as the optimum; the authors reported high bacteriolytic potential and good efficiency in limiting bacterial growth [50]. The vB_SauS_SA2 phage exhibited a 10 min latency and a burst size of 293 PFU/infected cell; its bactericidal effect was assessed in S. aureus F2 (5.5 × 108 CFU/mL) at MOIs 10−5 and 1 for 24 h at 3 °C. At MOI 1, lysis began 2 h after co-incubation, and by the end of treatment, most of the bacteria were lysed at all MOIs, indicating a strong inhibitory effect and high replication rate in S. aureus F2 [51].
The lytic activity of two lytic bacteriophages, vB_SauM-515A1 and vB_SauP-436A, of the commercial Staphylococcus bacteriophage cocktail produced by Microgen (Russia) was evaluated at MOI 0.1 and 1. The bactericidal effect of bacteriophage vB_SauM-515A1 was evaluated on S. aureus SA515 cultures, while the inhibitory effect of bacteriophage vB_SauP-436A1 was determined on S. aureus SA436. The results indicated that lysis of bacterial culture of the host strain S. aureus SA515 was faster than that of the S. aureus SA436 strain for approximately 3 h, which is attributed to the shorter latency period and larger burst size of bacteriophage vB_SauM-515A1 (40 min and 185 PFU/infected cell) compared to bacteriophage vB_SauP-436A1 (50 min and 94 PFU/infected cell) [52].
The UPMK_1 phage (MOI 1) exhibited a 20 min latency and a burst size of 32 PFU/infected cell. In S. aureus MRSA t127/4, treatment reduced optical density versus the untreated control, with a marked effect in the first 4 h and persistence up to 8 h [43]. In a separate assay, CSA13 (MOI 1) against S. aureus (2.6 × 108 CFU/mL) decreased optical density to undetectable from the first hour, remaining so up to 20 h; CSA13 showed a 20 min latency and a burst size of 230 PFU/infected cell [33]. Collectively, these studies confirm the promising potential of lytic bacteriophages as biocontrol tools against S. aureus, including antibiotic-resistant strains.
Beyond lytic efficiency, the stability of S. aureus bacteriophage under varying physicochemical and environmental conditions is a key determinant of their applicability in food systems. Phages generally remain viable between 4 °C and 50 °C and within a pH range of 4.0–11.0, but they are rapidly inactivated at temperatures ≥ 70 °C or under extreme acidic (pH ≤ 3.0) environments [53]. Complex foods denature phage particles, diminishing infectivity [54]. To enhance stability, preservation strategies such as lyophilization with cryoprotectants (e.g., trehalose, sucrose) or microencapsulation within polymeric matrices (e.g., alginate, chitosan) have been successfully applied to maintain structural integrity and lytic functionality under adverse conditions [55].
Importantly, the environmental impact of phage-based interventions is considered minimal, as bacteriophages specifically infect bacterial hosts and do not affect eukaryotic cells. Their high specificity and natural biodegradability position them as sustainable alternatives to chemical disinfectants or antibiotics, minimizing non-target effects and ecological disturbances. To ensure biosafety, only strictly lytic phages should be employed, thereby avoiding risks of lysogeny and horizontal gene transfer [56]. Supporting this, in situ assessments demonstrated that phages such as FoX2 and FoX4 did not alter local biomass or microbial diversity in soil microbiomes, confirming their environmental compatibility and alignment with sustainable biotechnological [57].
The results obtained in these studies confirm the potential of using bacteriophages for the biocontrol of S. aureus in strains resistant to antibiotics. Recently, several investigations have been published related to the isolation, evaluation of the lytic activity, and the efficiency of phages to lyse S. aureus present in foods.
5. Application of Bacteriophages That Infect S. aureus in Foods
In the food industry, bacteriophages and bacteriophage cocktails have been used for biocontrol of S. aureus strains present in dairy and meat products in order to decrease the likelihood of bacterial infection and disease, prevent biofilm formation, preserve the product, and promote safe environments in the production, processing, and handling of these products [58].
5.1. Milk Products
Milk is one of the most consumed foods worldwide. Through its processing, dairy products such as yogurt, cheese, whey, and butter can be obtained. During processing, thermal treatments are used that reduce the bacterial load and prolong the shelf life of the food; however, in some cases, the presence of microorganisms in these products has been determined [59] because milk and dairy products are often nutrient-rich and pH neutral, allowing the growth of various bacteria such as S. aureus [60]. In a study, the prevalence of S. aureus in samples of commercial pasteurized milk was determined, indicating that 75% of the isolates were resistant to multiple antibiotics at the different concentrations tested and were capable of producing biofilms [61].
Multiple studies report effective biocontrol of S. aureus in whole, skimmed, pasteurized, and UHT milk using bacteriophages, aided by the liquid matrix that facilitates phage-bacterium interaction [62]. In UHT milk inoculated at 108 CFU/mL, adding vB_SauM_ME126 and vB_SauM_ME18 at 25 °C for 6 h showed no control at MOI 1 [63], likely because milk proteins coat the bacterial surface and block host receptors [64]. At MOI 10, bactericidal activity was evident: vB_SauM_ME18 fully reduced the bacteria by 4 h, and vB_SauM_ME126 by 6 h.
In pasteurized milk inoculated with S. aureus (106 CFU/mL), phages SA2 and SANF applied at MOI 102 achieved a 90–99% growth reduction after 6 h at 25 °C; by contrast, at 37 °C, only SA2 maintained a >65% reduction over the same period [64]. In UHT milk inoculated at 104 CFU/mL, phage M8 at MOI 102 produced a 3 log10 CFU/mL decrease after 8 h at 37 °C [65]. These findings are noteworthy because low host densities typically require a higher phage-to-bacterium ratio to ensure efficient infection and sustained biocontrol [54].
The evaluation of the bactericidal effect of phage SA13m in pasteurized whole milk, previously inoculated with S. aureus ATCC 29213 (105 CFU/mL), indicated that adding the phage at MOI 102 achieved complete lysis at 4 °C, lowering counts by 4.1 log10 CFU/mL within 24 h with no regrowth up to 48 h. At 25 °C, the reduction reached 4.0 log10 CFU/mL by 48 h. Increasing the dose to MOI 103 at 25 °C produced the fastest and largest decline (4.2 log10 CFU/mL in 3 h) compared with 4 °C (4.2 log10 CFU/mL in 12 h) [66]. In skimmed milk containing S. aureus BM001 (103 CFU/mL), addition of bacteriophage LSA2308 under the same temperature–time conditions showed no evident control at MOI of 102–103 at 4 °C; by contrast, at 25 °C, biological control was observed between 6 and 48 h. The best results occurred with an MOI of 104 at 4 °C, where S. aureus BM001 was gradually reduced by 99.95% at 48 h [9].
The addition of bacteriophage SA46-CTH2 with MOI 104 in pasteurized milk containing S. aureus SA46 (10 5 CFU/mL) at 4 °C for 24 h, reduced the viable counts of the microorganism by 2.2 log10 CFU/mL. An increased lytic activity was also reported for a bacteriophage cocktail composed of STA1.ST29, EB1.ST11 and EB1.ST27, applied at an MOI of 104 against S. aureus 10614 (1.1 × 105 CFU/mL) in pasteurized milk at 4 °C for 8 h, resulting in a reduction of approximately 2 log10 CFU/mL [67]. Similarly, when evaluated in raw milk under the same conditions, the maximum reduction achieved was around 1 log10 CFU/mL [68]. The use of bacteriophage cocktails offers an effective alternative for controlling foodborne pathogens such as S. aureus. Their combined action broadens the host range and decreases the likelihood of bacterial resistance. Optimizing their formulation involves selecting complementary phages that maximize bacterial reduction while maintaining manageable complexity. This strategy enhances robustness and ensures sustained lytic efficacy across diverse strains and food matrices [69]
5.2. Cheese
S. aureus has been identified in dairy products and high-salt foods such as cheese [70]. The biocontrol of S. aureus present in cheeses has been studied mainly in cheddar cheese during its ripening and in the commercial product. One study evaluated the addition of a cocktail of three bacteriophages, phi812, 44AHJD, and phi2, of different morphologies (belonging to Groups A, B, and C) with MOIs of 15, 45, and 150 in S. aureus SMQ-1320 (106 CFU/mL) during the elaboration of cheddar cheese. This reduced the contamination to 4 log10 CFU/mL after a ripening period of 14 days at 4 °C, transforming the contaminated milk samples into pasteurized cheese with controlled ripening [71]. On the other hand, the lytic effect of bacteriophage KMSP1 was evaluated with MOI 103 and 104 on S. aureus (104 CFU/cm2) present in 3 × 3 cm2 slices of cheddar cheese, where reductions between 0.6 and 1.4 log10 CFU/mL were presented after 30 min at 25 °C; this result was maintained without bacterial multiplication for 24 h [72].
In fresh cheese, gelatin-glycerol films loaded with phiIPLA-RODI (1.75 × 108, 1.16 × 108 and 6.35 × 107 PFU/mL) were tested against S. aureus (105 CFU/mL). The highest phage dose yielded the lowest counts (44 CFU/g), whereas the lowest dose sustained control up to day 3 [73]. By day 6, bacterial levels were similar to the control, likely because phages adsorb to the food surface and, given their non-motile phenotype, limit contact with cells [74]. Accordingly, higher phage concentrations and full surface coverage are advised to maximize phage-host contact [75].
The phage cocktail (vB_SauS-phi-IPLA35 and vB_SauS-phi-IPLA88) effectively reduced S. aureus in cheese. In fresh cheese, counts decreased by 3.83 log10 CFU/g within 3 h, reaching undetectable levels at 6 h. In hard cheese, reductions reached 4.64 log10 CFU/g in the curd and 1.24 log10 CFU/g after ripening, demonstrating consistent efficacy during processing and storage [76]. Similarly, the S. aureus phage cocktail (EBTH and K2, 108 PFU/mL) efficiently controlled both inoculated and native S. aureus in traditional Egyptian cheeses. In soft varieties (Karish and Domiati), bacterial counts dropped below detection (≤10 CFU/g) within 3–24 h, preventing enterotoxin accumulation, whereas in hard Ras cheese, complete elimination occurred after 60–90 days of ripening [77]. More recently, a biopreservation strategy for hard Dutch-type cheese using two virulent phages (No. 4 and No. 8) applied post-pasteurization (108 PFU/mL) completely prevented S. aureus growth during 60 days of ripening, while the control reached 154.2 CFU/g, confirming the strong protective capacity of the phage treatment [78].
Collectively, these studies demonstrate that the use of lytic bacteriophage and phage cocktails can effectively reduce or eliminate S. aureus in both fresh and hard cheeses without compromising the activity of starter cultures or altering the physicochemical properties of the final product. This highlights their potential as safe and efficient biocontrol agents in the dairy industry.
5.3. Meat Products
S. aureus has also been identified in beef, lamb, chicken, and meat products, causing staphylococcal food poisoning with vomiting and diarrhea [79]. Moreover, the biocontrol of the microorganism (108 CFU/mL) was evaluated in samples of raw chicken filets using the bacteriophage vB_SauM_CP9 with MOI 1 and 10, which presented lytic activity in ten strains of S. aureus, with a burst size of 228 PFU/infected cell and a latent period of 45 min. With MOI 1 and 10, the reductions in S. aureus were 73.06% and 76.94%, respectively. In the research, they also evaluated the bactericidal effect of 1% thyme oil (Thymus vulgaris) in combination with the bacteriophage with an MOI of 10. A reduction of 87.22% of the microorganism was presented for 120 min, which corresponded to the treatment with the greatest inhibition [80]. This result indicates the potential of combined treatment with bacteriophages and antimicrobial products to eliminate foodborne pathogens.
Biocontrol of S. aureus in dairy and meat products with bacteriophages has shown varying levels of success. In this regard, the application of endolysins obtained from bacteriophages has been explored as an alternative for biocontrol of the microorganism to overcome the barriers developed by S. aureus against phage activity.
6. Importance of Endolysins in the Food Industry
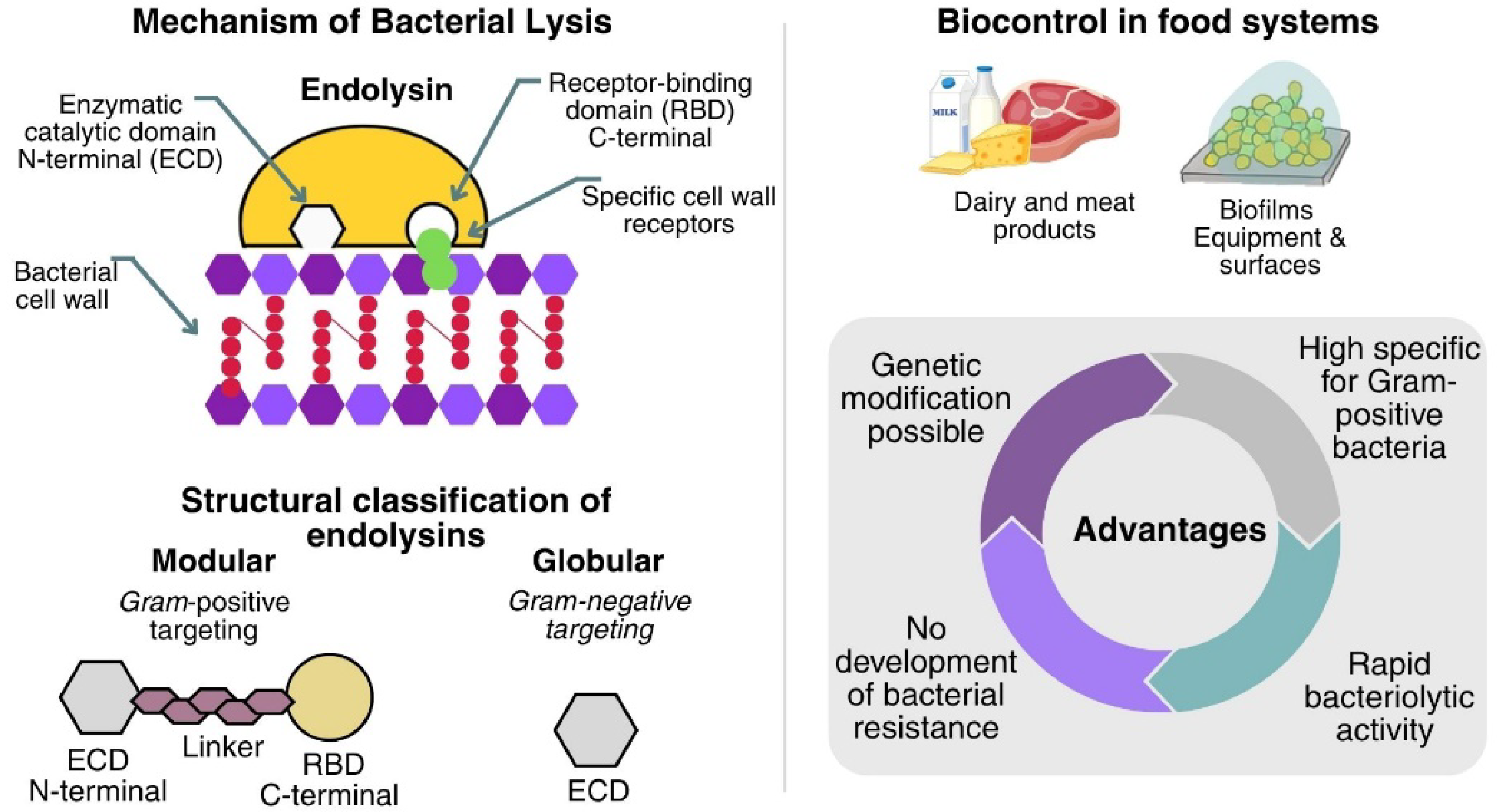
The study of the mechanisms by which bacteriophages lyse bacteria has made it possible to determine the antimicrobial potential of endolysins. Endolysins are phage enzymes synthesized during the final stage of the lytic cycle. These enzymes cause the rupture of the bacterial cell wall, thus releasing the newly formed virions so that they can infect new host cells [81]. Endolysins are enzymes composed of the catalytic domain present in the N-terminal region, which provides the catalytic activity, and the binding domain. This domain specifically recognizes and binds to its target bacterial cell wall receptors, present in the C-terminal region. Structurally, endolysins are classified into modular, bacteriophage-encoded, Gram-positive-targeting, bacteria-targeting, and globular, Gram-negative-targeting, which comprise a single enzymatically active domain and lack a modular structure [82,83].
Endolysins are a promising option for food-industry decontamination; they can be produced and purified at scale at low cost, display high specificity for Gram-positive bacteria (acting externally in the absence of an outer membrane), and can be protein-engineered to enhance bacteriolytic activity, solubility, and other physicochemical traits. They also show a lower propensity for resistance because they cleave highly conserved bonds in the peptidoglycan. Taken together, these advantages support their use as biocontrol tools in foods [84,85,86].
Another advantage of endolysins for food industry decontamination is that they spare the beneficial microbiota owing to their host specificity. They also achieved rapid lysis of Gram-positive bacteria in foods within minutes using low doses [87]. Endolysins are likewise promising against biofilms, surface-attached microbial communities, of concern because they drive food contamination, contribute to the pathogenesis of foodborne diseases, and show tolerance to antibiotics and sanitation. The endolysin LysCSA13, derived from the virulent CSA13 phage of S. aureus, demonstrated high efficiency (80–90%) in reducing biofilm biomass on polystyrene, glass, and stainless Steel [88].
Figure 2 presents the main advantages of studying and applying endolysins in the food industry. Therefore, endolysins can be used as biocontrol agents for various uses in the food industry, covering applications in equipment, surfaces, and foods.
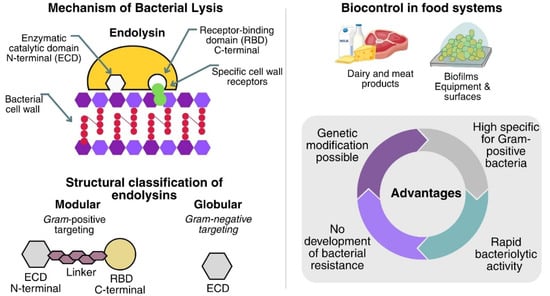
Figure 2.
Mechanism, structural organization, and advantages of endolysins.
7. Biocontrol of S. aureus with Endolysins
Regarding the studies carried out evaluating the lytic activity of endolysins in S. aureus strains, the endolysin LysSAP8 derived from the bacteriophage SAP8 presented high efficiency in the inhibition of S. aureus at temperatures between 15 and 30 °C, pH between 7 and 8, in a medium supplemented with calcium ions and a NaCl concentration of 500 mM. The evaluation of the antimicrobial effect on S. aureus KCCM 12103 (>108 CFU/mL) by LysSAP8 (1 µM) resulted in a reduction of 3.46 log10 (CFU/mL) in 30 min, evidencing that endolysin has a good potential for applications in foods containing NaCl or neutral/subacidic foods [89].
Endolysin HY-133 showed a Minimum Inhibitory Concentration (MIC) of 0.12–0.25 µg/mL against methicillin-resistant S. aureus (MRSA), methicillin-susceptible S. aureus (MSSA), and mupirocin-resistant S. aureus (MupRSA) tested at 5 × 105 CFU/mL. Maximal inhibition occurred at 2 h, comparable to daptomycin (a lipopeptide antibiotic). However, regrowth by 48 h was observed for MSSA ATCC 29213 and MRSA ATCC 43300 [90]. For CF-301 (exebacase), MICs of 0.2–0.4 µg/mL were reported with lysis of S. aureus ATCC 29213 (4.8 × 105 CFU/mL) [91]. In another study, recombinant LysSAP26 showed an MIC of 20 µg/mL across 20 oxacillin-resistant S. aureus (ORSA) isolates [92].
The lytic activity of the CHAP-amidase domain of endolysin Lysk (1, 5, and 10 µg/mL) was evaluated in MRSA strains (8 × 108 CFU/mL). With 10 µg/mL of Lysk, the greatest reduction in MRSA (3.2 Log10 CFU/mL) was observed during 50 min of incubation at 37 °C, while a MIC of 1 µg/mL reduced MRSA by more than 1 log10 CFU/mL. Regarding temperature, there was no significant alteration in CHAP-amidase activity between 5 and 60 °C for 15 min. Furthermore, endolysin remained active for 1 month and 1 year when stored at 4 °C and −20 °C, respectively [93].
The evaluation of the lytic activity of the CHAPk endolysin was evaluated in MRSA 252 (7 × 105 CFU/mL) at temperatures of 32 and 37 °C, the latter being the one in which the greatest decrease in the bacterial optical density of MRSA 252 was observed [94]. The evaluation of the bacteriolytic effect of endolysin LysRODI (13.44 μg/mL) derived from the bacteriophage phiIPLA-RODI was determined in 67 MRSA strains. The results obtained showed that endolysin had a lower inhibitory effect in 62.69% of the strains, such as MRSA-IPLA 4, 20, 22, 26, 30, 32, 35, 43, 50, and 58. The medium and high lytic activity was present in the remaining 37.31% of the strains, among which the activity in MRSA-IPLA 28 and 46 stood out, respectively [95].
8. Application of Endolysins Infecting S. aureus in Foods
The lytic activity of endolysins for the biocontrol of S. aureus has been studied mainly in dairy products such as pasteurized and UHT whole milk, skimmed milk, cheese, and in meat products such as pork, beef, ham, and bacon. The lytic activity of the endolysins evaluated has shown variability in the reduction of the microorganism count in foods. The variability observed is mainly due to the protein stability and antimicrobial efficiency of the endolysins used. In turn, this is closely related to various factors such as the structure of the enzyme, the use of cofactors, the specificity, and affinity that endolysins present with foods, related to their composition of fats, proteins, minerals, among others, in addition to biochemical factors such as temperature, pH, and ionic strength that occur when carrying out the treatments [96].
8.1. Endolysin-Based Control in Milk Products
Regarding the biocontrol of S. aureus in milk using endolysins, the antimicrobial effect of LysGH15 (25 nM) was determined in whole and skimmed milk samples contaminated with MRSA 2701 (108 CFU/mL). This was performed to evaluate the effect of fat on the antimicrobial efficiency of LysGH15 in the milk samples evaluated at 37 °C for 24 h. The results indicated that LysGH15 did not present a significant bactericidal activity, and the growth rate of MRSA increased slightly, similar to the behavior in the control group. The bacteriolytic action occurred more quickly in skimmed milk than in whole milk due to endolysin, signifying that the lower amount of fat was more favorable for the bactericidal activity; this is attributed to the fact that fat affected the binding of LysGH15 to MRSA 2701 cells [97].
Similar results were presented in the study of the antimicrobial activity of endolysin LysSA97 (1.88 μM) in previously pasteurized skimmed and whole milk samples inoculated with S. aureus RN4220 (105 CFU/mL). The reduction in bacterial cell count was less than 2 log10 CFU/mL in skimmed milk for 3 h, while in whole milk samples, there was no evident control of S. aureus. Therefore, endolysin may be more effective in low-fat food products [98].
The lytic activity of Lys109 (30, 300, 900, and 1500 nM) was determined against the MRSA strain CCARM 3090 (105 CFU/mL) previously inoculated in pasteurized whole milk at 37 °C for 1 h. After treatment with 300 nM of Lys109, the control of the bacteria was evident, taking into account that the reduction had a value of 2 log10 CFU/mL of S. aureus at the end of the evaluation. Regarding the results related to the concentration of 900 nM of lysine, there was a total elimination of the microorganism after 45 min of testing [99].
The inhibitory effect of LysSA11 at concentrations of 1.125, 2.25, 3.375, 4.5, and 9 μM on MRSA CCARM 3089 (2 × 105 CFU/mL) present in commercial pasteurized milk was evaluated for 1 h at 4 °C and 25 °C, respectively. Endolysin showed greater activity at 25 °C, since bacterial cells were reduced to undetectable levels with 9 μM of lysine in 30 min. At the same concentration at 4 °C, the same results were obtained after 60 min of treatment. This indicates an adequate temperature for applying the enzyme [100]. Therefore, it should be used after heat treatments in milk or other foods and should not be applied to refrigerated products [101].
Comparison of the potential of LysRODI lysine (4 μM, 8 μM, or 16 μM) obtained from phage phiIPLA-RODI and its derivative chimeric lysine LysRODIΔAmi, lacking the amidase domain, to lyse S. aureus cells in pasteurized whole milk, UHT whole milk, and UHT skim milk (103, 104, and 105 CFU/mL) was evaluated at 37 °C for 2 h. LysRODI showed a reduction between 1 and 2 log10 CFU/mL of S. aureus in the milk samples evaluated. LysRODIΔAmi was more effective in reducing S. aureus because there was an elimination of bacterial contamination below the detection levels in all samples, except for the UHT whole milk sample that contained 105 CFU/mL of S. aureus, where there was a reduction of 2.82 log10 CFU/mL [81].
8.2. Endolysin-Based Control in Cheese
The biocontrol of S. aureus with endolysins has been little studied in cheese. However, the studies carried out have shown the potential that endolysins have to lyse the microorganism. The reduction in the bacterial cell count of S. aureus ATCC 33591 (104 CFU/mL) during the production of ripened cheese was less than 2 log10 CFU/mL after 6 h of adding the endolysin Lysdb from the phage Lactobacillus delbrueckii at 38 °C. During cheese ripening at 10 °C after 6 weeks, the S. aureus counts were 2.7 × 103 CFU/g [102]. On the other hand, Endolysin LysRODI (4 μM) reduced viable S. aureus cells during cheese production by approximately 2 log10 CFU/mL after 2 h incubation at 32 °C. For the sample stored for 7 days at 12 °C and 90% relative humidity, the reduction in S. aureus was 0.67 ± 0.44 log10 CFU/mL compared to the untreated control sample [81].
8.3. Endolysin-Based Control in Meat Products
LysSA11 endolysin at concentrations of 1.125, 2.25, 3.375, 4.5, and 9 μM showed lytic activity against MRSA CCARM 3089 (2 × 105 CFU/mL) in 0.5 cm3 ham cubes at 4 °C and 25 °C for one hour. The evaluation of the lytic activity shown by LysSA11 was more efficient at 25 °C since the concentrations 2.25, 3.375, 4.5, and 9 μM completely reduced the bacteria in 30 min. In the evaluation of the inhibitory effect at 4 °C, the concentration of 2.25 μM of LysSA11 reduced viable cells to undetectable levels in 45 min [100]. The results obtained indicate that LysSA11 has greater lytic activity in the microorganism present in ham at 25 °C than at 4 °C, so treatment with endolysin can be used at 25 °C and 30 min before refrigeration of this product.
The evaluation of the lytic activity of endolysin LysSA97 (18.8 μM), carvacrol (6.66 mM) and the combination of LysSA97-carvacrol (18.8 μM and 6.66 mM) in 2 cm3 beef cubes contaminated with S. aureus (105 CFU/mL) did not show a significant reduction in the live bacterial cell counts in the meat samples treated with LysSA97 and carvacrol compared to the control sample. The combination LysSA97-carvacrol reduced 2.1 ± 0.5 log10 CFU/cm2 of S. aureus in the meat samples for 3 h [98]. Therefore, a significant synergistic effect was presented between lysine and carvacrol, which is a monoterpenoid phenol present in the essential oils of oregano (Origanum vulgare), thyme (Thymus vulgaris), and peppermint (Lepidium flavum) with antimicrobial properties [103].
Biocontrol of S. aureus (104 CFU/cm2) present in Chinese bacon samples (5 cm × 5 cm × 0.5 cm) containing approximately 10.52 ± 1.21% salt (180 mM) and low-salt fresh pork was carried out with LysGH15 concentrations of 0.1 nM/cm2, 0.2 nM/cm2, and 0.4 nM/cm2 at 4 °C for 2 h. The results indicated that LysGH15 increased its inhibitory effect on S. aureus as its concentration in Chinese bacon increased, such that the microorganism was completely reduced at the highest concentration. However, in the pork samples, there was an evident control of S. aureus, observing significant differences in relation to the evaluated samples, considering that with the highest concentration of LysGH15, the reduction was 1 log10 CFU/cm2 [97].
The evaluation of the inhibitory effect of the LysGH15 CHAP domain on S. aureus was carried out under the same conditions as previously mentioned in the same foods. However, the lytic activity of LysGH15 CHAP on S. aureus in Chinese bacon was lower, and the highest reduction had a value of 0.7 log10 CFU/cm2 (0.4 nM/cm2 of LysGH15 CHAP). In fresh pork samples, a reduction in S. aureus was seen as the concentration of LysGH15 CHAP increased, which completely eliminated S. aureus from pork. Therefore, the LysGH15 CHAP domain could be an interesting strategy for low-salt and non-acidic foods [97].
Table 2 presents the results obtained in different investigations related to the biocontrol of S. aureus strains and some food matrices.

Table 2.
Biocontrol studies of S. aureus with bacteriophages and endolysins.
9. Commercial Phage Products for Biocontrol of S. aureus
For the biocontrol of microorganisms, mixtures of bacteriophages and endolysins known as phage cocktails have been widely applied, and several commercial products are currently available for food safety applications. Among them are ListShield™ and Listex P-100™ (for Listeria monocytogenes), SalmoFresh™ and Salmonelex™ (Salmonella enterica), all approved by the United States Department of Agriculture (USDA) for use in ready-to-eat or raw foods [109]. In agriculture, the first phage-based product formally approved by U.S. regulatory agencies (U.S. Environmental Protection Agency) was Agriphage™, developed by OmniLytics Inc. in 2005 to control bacterial diseases in crops [110].
Despite the growing global interest in bacteriophages as a sustainable alternative to antibiotics, their generalized use in food for biocontrol or biopreservation remains restricted in the European Union (EU). This limitation arises because phages are not included in the list of approved food additives under current EU legislation [111]. The European Food Safety Authority (EFSA) has shown caution in endorsing phage-based applications for human consumption, mainly due to concerns regarding the potential transfer of undesirable genes and challenges in their taxonomic classifications. Consequently, bacteriophages have not been granted the Qualified Presumption of Safety (QPS) status, which has delayed regulatory acceptance compared to other regions [112].
Currently, the only phage-based preparation to obtain full regulatory approval within the EU is Bafasal®, authorized for use exclusively as an additive in poultry feed starting in July 2025. Meanwhile, European biotechnology companies such as Micreos have developed innovative formulations, such as PhageGuard STM, designed to reduce Salmonella in raw and ready-to-eat meats, and PhageGuard LTM, targeted against Listeria monocytogenes. PhageGuard LTM is one of the most advanced candidates under EFSA evaluation and is expected to become the first phage product authorized for direct application on food surfaces in the EU [111].
In addition to regulatory progress, several studies have demonstrated the high efficacy of commercial phage cocktails designed for S. aureus control. These formulations offer broader host ranges and enhanced lytic performance due to the synergistic interaction among multiple phages, which surpasses the inhibitory capacity of individual phages or endolysins [113]. For instance, the commercial product SASPject PT1.2 showed strong antibacterial activity, achieving reductions of 3–5 log10 CFU/mL in 93.7% of MRSA and 67.6% of MSSA isolates, as well as in mixed MRSA/MSSA cultures after only 3 h of treatment [114].
Commercial phage products specific for the control of S. aureus are summarized in Table 3.

Table 3.
Characterization of phage products used for the biocontrol of S. aureus.
10. Limitations of S. aureus Bacteriophages and Endolysins in Foods
Various researchers have studied the use of bacteriophages and endolysins for the biocontrol of S. aureus. Significant results have been reported in the reduction in microorganisms. Nevertheless, the application of phages and endolysins in foods presents some difficulties related to the isolation of S. aureus bacteriophages because they present a high level of non-specific adsorption of dead bacteria, molds, yeasts, milk proteins, carbohydrate fractions, etc., present in various samples, which generates their inactivation. Likewise, staphylococcal phages usually form small or even cloudy punctate plaques, which are difficult to enumerate or visualize, which decreases the probability of their isolation and purification [123,124,125]
Regarding the difficulty in isolating S. aureus bacteriophages, the isolation of phage from wastewater samples was reported after a total of 117 enrichment attempts, in contrast to the bacteriophage isolation trials for E. coli, K. pneumoniae, P. aeruginosa, and Salmonella, where there was a high success rate from the same samples [118]. In the isolation of bacteriophages of Enterococcus faecium, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter cloacae from 16 samples from different sources, the isolation of bacteriophages of S. aureus was not carried out [126].
Despite the multiple advantages presented by S. aureus bacteriophages and endolysins, such as their host specificity without affecting the normal microflora, self-replication in the presence of the microorganism, their use in bacterial bio-detection and as biocontrol products of S. aureus in foods, they have structures composed of proteins, which are affected by factors such as temperature, pH, acidity and composition of food matrices, such as fat and salt content [32,98,127]. On the other hand, horizontal gene transfer is frequently observed in S. aureus and is associated with the presence of prophages (group B), so the use of these bacteriophages without previously performing their genomic characterization can promote the transfer of virulent genes or resistance to antibiotics [19,36].
The implementation of bacteriophage-based products faces persistent limitations in regulation, scalability, and food matrix variability. Regulatory disparities remain a critical obstacle; although several formulations have obtained GRAS status in the U.S., the EFSA excludes phages from QPS due to classification issues, lysogenic potential, and gene transfer risks. Industrial scalability is constrained by the biological complexity of phage-host systems, complicating standardized large-scale production. Moreover, phage efficacy decreases in solid foods, where limited diffusion and adverse physicochemical conditions (pH, temperature) reduce activity. Typical bacterial reductions (1–3 log10 CFU/mL highlight the need for improved stabilization methods, such as encapsulation and harmonized international regulatory frameworks, to ensure broader practical adoption
In the case of endolysins, it is important to mention that their production requires the isolation, propagation, and purification of bacteriophages. In addition to the cloning of the enzymes and their purification, some endolysins show low levels of expression and insolubility [128]. In addition, the enzymatic activity of endolysins is influenced by the composition of the cell walls, so to be used in the biocontrol of Gram-negative bacteria, which have a cell wall composed of an internal cytoplasmic membrane, a peptidoglycan layer and an external membrane that has a complex lipopolysaccharide layer, the use of membrane permeabilizers such as peptides, nanoparticles, aminoglycosides, EDTA, among others, is necessary to facilitate the degradation of the cell wall of Gram-negative bacteria [129,130,131].
11. Conclusions
Determining the efficiency of the application of bacteriophages and endolysins for the biocontrol of S. aureus is of great importance in the food industry due to the advantages that phage products have compared to antibiotics and the effects on food safety associated with contamination caused by the microorganism. In the reported research, the most evaluated means for the biocontrol of S. aureus by bacteriophages and endolysins were in cultures of antibiotic-resistant strains and in foods such as dairy and meat products.
Bacteriophages and endolysins are made up of protein structures, so they depend on the treatment conditions for the biocontrol of S. aureus, such as pH, acidity, and affinity with the composition of food matrices. In addition, the lytic activity of endolysins in S. aureus occurs in less time compared to the use of bacteriophages and has lower bacterial resistance. It is important to note that one of the disadvantages of enzymes is that they do not have the ability to replicate as bacteriophages do.
Finally, the importance of biocontrol of S. aureus by bacteriophages and endolysins is highlighted, which largely inhibited the growth of the bacteria under different conditions and even in dairy and meat products, characterized by being low or high in sodium and fat.
Author Contributions
Conceptualization, M.E.S.L. and F.M.-C.; literature search, A.M.O.-H., R.L.-V., F.M.-C. and P.M.P.V.; original draft preparation, M.E.S.L., F.M.-C., A.M.O.-H., J.J.S.-B. and A.M.H.-A.; review and editing, M.E.S.L., F.M.-C., D.C. and P.M.P.V.; supervision, M.E.S.L. and O.P.-S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ministerio de Ciencia Tecnología e Innovación (MINCIENCIAS) of Colombia, through the project “Desarrollo y transferencia de conocimiento para la innovación de productos biocontroladores en queso costeño para atender las necesidades del sector derivadas de la emergencia económica y social causada por el COVID-19 en el departamento de Córdoba” (funding code: BPIN 2020000100697). P.M.P.V. is supported by a fellowship from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), grant number 200773/2024-0.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Acknowledgments
The authors thank the Universidad de Córdoba and Grupo de Investigaciones en Propiedades y Procesos Alimentarios (GIPPAL), for providing the space and resources used in this study. They also acknowledge the support of the Ministerio de Ciencia Tecnología e Innovación (MINCIENCIAS) of Colombia, and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| S. aureus | Staphylococcus aureus |
| MRSA | Methicillin-Resistant Staphylococcus aureus |
| MSSA | Methicillin-Susceptible S. aureus |
| VRSA | Vancomycin-Resistant S. aureus |
| CFU | Colony Forming Unit |
| PFU | Plaque Forming Unit |
| MOI | Multiplicity of Infection |
| Aw | Water activity |
| DNA | Deoxyribonucleic Acid |
| dsDNA | Double-Stranded DNA |
| RBPs | Receptor-Binding proteins |
| PIA | Polysaccharide Intercellular Adhesin |
| MIC | Minimum Inhibitory Concentration |
| UHT | Ultra-High Temperature |
| ICTV | International Committee on Taxonomy of Viruses |
| GC | Guanine - Cytosine |
| GRAS | Generally Recognized As Safe |
| QPS | Qualified Presumption of Safety |
| OECD | Organization for Economic Co-operation and Development |
| EFSA | European Food Safety Authority |
| EU | European Union |
| USDA | United States Department of Agriculture |
References
- Pasachova Garzón, J.; Ramírez Martínez, S.; Muñoz Molina, L. Staphylococcus aureus: Generalidades, mecanismos de patogenicidad y colonización celular. Nova 2019, 32, 25–38. [Google Scholar] [CrossRef]
- Méndez, I.A.; Holguín-Riaño, D.F.; Pachón-Barinas, D.P.; Africano, F.J.; González, I.M.; Rojas, N.A. Prevalence and antimicrobial susceptibility of Staphylococcus aureus methicilin resistant isolated from medical students. CES Med. 2013, 27, 21–30. Available online: http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S0120-87052013000100003&lng=en&nrm=iso&tlng=en (accessed on 10 February 2025).
- Craft, K.M.; Nguyen, J.M.; Berg, L.J.; Townsend, S.D. Methicillin-resistant Staphylococcus aureus (MRSA): Antibiotic-resistance and the biofilm phenotype. Medchemcomm 2019, 10, 1231–1241. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority; European Centre for Disease Prevention and Control. The European Union One Health 2018 Zoonoses Report. EFSA J. 2019, 17, e05926. [Google Scholar] [CrossRef] [PubMed]
- Pipiya, S.O.; Mokrushina, Y.A.; Gabibov, A.G.; Smirnov, I.V.; Terekhov, S.S. Selective Eradication of Staphylococcus aureus by the Designer Genetically Programmed Yeast Biocontrol Agent. Antibiotics 2020, 9, 527. [Google Scholar] [CrossRef]
- Carneiro Aguiar, R.A.; Ferreira, F.A.; Dias, R.S.; Nero, L.A.; Miotto, M.; Verruck, S.; De Marco, I.; De Dea Lindner, J. Graduate Student Literature Review: Enterotoxigenic potential and antimicrobial resistance of staphylococci from Brazilian artisanal raw milk cheeses. J. Dairy Sci. 2022, 105, 5685–5699. [Google Scholar] [CrossRef]
- Ringaci, A.; Shevchenko, K.; Zelepukin, I.; Popova, A.; Nikitin, M. Phage-mimicking nanoagents for rapid depolymerase specificity screening against multidrug resistant bacteria. Biosens. Bioelectron. 2022, 213, 114444. [Google Scholar] [CrossRef]
- Dunne, M.; Prokhorov, N.S.; Loessner, M.J.; Leiman, P.G. Reprogramming bacteriophage host range: Design principles and strategies for engineering receptor binding proteins. Curr. Opin. Biotechnol. 2021, 68, 272–281. [Google Scholar] [CrossRef]
- Ma, F.; Ning, Y.; Wan, Q.; Zou, L.; Liu, Y.; Chen, S.; Li, J.; Zeng, Z.; Yang, Y.; Chen, H.; et al. Bacteriophages LSA2308 and LSA2366 infecting drug-resistant Staphylococcus aureus: Isolation, characterization and potential application for milk safety. LWT 2021, 152, 112298. [Google Scholar] [CrossRef]
- Cisek, A.A.; Dąbrowska, I.; Gregorczyk, K.P.; Wyżewski, Z. Phage Therapy in Bacterial Infections Treatment: One Hundred Years After the Discovery of Bacteriophages. Curr. Microbiol. 2017, 74, 277–283. [Google Scholar] [CrossRef]
- Briot, T.; Kolenda, C.; Ferry, T.; Medina, M.; Laurent, F.; Leboucher, G.; Pirot, F. Paving the way for phage therapy using novel drug delivery approaches. J. Control. Release 2022, 347, 414–424. [Google Scholar] [CrossRef]
- Han, S.; Byun, K.H.; Mizan, M.F.R.; Kang, I.; Ha, S. Do Bacteriophage and their lysins: A new era of biocontrol for inactivation of pathogenic bacteria in poultry processing and production—A review. Food Control 2022, 137, 108976. [Google Scholar] [CrossRef]
- Essa, N.; Rossitto, M.; Fiscarelli, E.V. Phages and phage therapy: Past, present and future. Microbiol. Medica 2020, 35, 8709. [Google Scholar] [CrossRef]
- Picoli, S.U.; Röhnelt, N.M.S.; Schenkel, T.S.; Picoli, S.U.; Röhnelt, N.M.S.; Schenkel, T.S. Bacteriophages as Anti-Methicillin Resistant Staphylococcus aureus Agents; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Feng, T.; Leptihn, S.; Dong, K.; Loh, B.; Zhang, Y.; Stefan, M.I.; Li, M.; Guo, X.; Cui, Z. JD419, a Staphylococcus aureus Phage with a Unique Morphology and Broad Host Range. Front. Microbiol. 2021, 12, 602902. [Google Scholar] [CrossRef] [PubMed]
- Schmelcher, M.; Shen, Y.; Nelson, D.C.; Eugster, M.R.; Eichenseher, F.; Hanke, D.C.; Loessner, M.J.; Dong, S.; Pritchard, D.G.; Lee, J.C.; et al. Evolutionarily distinct bacteriophage endolysins featuring conserved peptidoglycan cleavage sites protect mice from MRSA infection. J. Antimicrob. Chemother. 2015, 70, 1453–1465. [Google Scholar] [CrossRef]
- Park, D.W.; Lee, Y.D.; Park, J.H. Characteristics for phage-encoded cell wall hydrolase of LysSAP27 to reduce staphylococcal food poisoning. Food Sci. Biotechnol. 2021, 30, 745–753. [Google Scholar] [CrossRef]
- Hatoum-Aslan, A. The phages of staphylococci: Critical catalysts in health and disease. Trends Microbiol. 2021, 29, 1117–1129. [Google Scholar] [CrossRef]
- Ingmer, H.; Gerlach, D.; Wolz, C. Temperate Phages of Staphylococcus aureus. Microbiol. Spectr. 2019, 7, 10–1128. [Google Scholar] [CrossRef]
- Mahros, M.A.; Abd-Elghany, S.M.; Sallam, K.I. Multidrug-, methicillin-, and vancomycin-resistant Staphylococcus aureus isolated from ready-to-eat meat sandwiches: An ongoing food and public health concern. Int. J. Food Microbiol. 2021, 346, 109165. [Google Scholar] [CrossRef]
- Bencardino, D.; Amagliani, G.; Brandi, G. Carriage of Staphylococcus aureus among food handlers: An ongoing challenge in public health. Food Control 2021, 130, 108362. [Google Scholar] [CrossRef]
- Wan, Y.; Yang, L.; Li, Q.; Wang, X.; Zhou, T.; Chen, D.; Li, L.; Wang, Y.; Wang, X. Stability and emetic activity of enterotoxin like X (SElX) with high carrier rate of food poisoning Staphylococcus aureus. Int. J. Food Microbiol. 2023, 404, 110352. [Google Scholar] [CrossRef]
- Spanu, V.; Scarano, C.; Virdis, S.; Melito, S.; Spanu, C.; De Santis, E.P.L. Population structure of Staphylococcus aureus isolated from bulk tank goat’s milk. Foodborne Pathog. Dis. 2013, 10, 310–315. [Google Scholar] [CrossRef]
- Soltan-Dallal, M.M.; Rajabi, Z.; Mirbagheri, S.Z.; Keyvanlou, M.; Didar, Z. Characterisation of antibiotic resistant Staphylococcus species and molecular identification of mecA and toxic shock syndrome toxin-1 (tsst-1) genes of Staphylococcus aureus isolates from cows’ milk. Int. Dairy J. 2023, 147, 105762. [Google Scholar] [CrossRef]
- Argudín, M.Á.; Mendoza, M.C.; Rodicio, M.R. Food Poisoning and Staphylococcus aureus Enterotoxins. Toxins 2010, 2, 1751–1773. [Google Scholar] [CrossRef] [PubMed]
- Fisher, E.L.; Otto, M.; Cheung, G.Y.C. Basis of Virulence in Enterotoxin-Mediated Staphylococcal Food Poisoning. Front. Microbiol. 2018, 9, 436. [Google Scholar] [CrossRef] [PubMed]
- Doulgeraki, A.I.; Di Ciccio, P.; Ianieri, A.; Nychas, G.J.E. Methicillin-resistant food-related Staphylococcus aureus: A review of current knowledge and biofilm formation for future studies and applications. Res. Microbiol. 2017, 168, 1–15. [Google Scholar] [CrossRef]
- Pacha, P.A.; Munoz, M.A.; González-Rocha, G.; San Martín, I.; Quezada-Aguiluz, M.; Aguayo-Reyes, A.; Bello-Toledo, H.; Latorre, A.A. Molecular diversity of Staphylococcus aureus and the role of milking equipment adherences or biofilm as a source for bulk tank milk contamination. J. Dairy Sci. 2021, 104, 3522–3531. [Google Scholar] [CrossRef]
- Azmi, K.; Qrei, W.; Abdeen, Z. Screening of genes encoding adhesion factors and biofilm production in methicillin resistant strains of Staphylococcus aureus isolated from Palestinian patients. BMC Genom. 2019, 20, 578. [Google Scholar] [CrossRef]
- Di Ciccio, P.; Vergara, A.; Festino, A.R.; Paludi, D.; Zanardi, E.; Ghidini, S.; Ianieri, A. Biofilm formation by Staphylococcus aureus on food contact surfaces: Relationship with temperature and cell surface hydrophobicity. Food Control 2015, 50, 930–936. [Google Scholar] [CrossRef]
- Chon, J.; Sung, K.; Khan, S.; Chon, J.; Sung, K.; Khan, S. Methicillin-Resistant Staphylococcus aureus (MRSA) in Food-Producing and Companion Animals and Food Products. In Frontiers in Staphylococcus aureus; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Yan, J.; Yang, R.; Yu, S.; Zhao, W. The application of the lytic domain of endolysin from Staphylococcus aureus bacteriophage in milk. J. Dairy Sci. 2021, 104, 2641–2653. [Google Scholar] [CrossRef]
- Cha, Y.; Chun, J.; Son, B.; Ryu, S. Characterization and Genome Analysis of Staphylococcus aureus Podovirus CSA13 and Its Anti-Biofilm Capacity. Viruses 2019, 11, 54. [Google Scholar] [CrossRef]
- Kahánková, J.; Pantůček, R.; Goerke, C.; Růžičková, V.; Holochová, P.; Doškař, J. Multilocus PCR typing strategy for differentiation of Staphylococcus aureus siphoviruses reflecting their modular genome structure. Environ. Microbiol. 2010, 12, 2527–2538. [Google Scholar] [CrossRef]
- Merrick, C.A.; Zhao, J.; Rosser, S.J. Serine Integrases: Advancing Synthetic Biology. ACS Synth. Biol. 2018, 7, 299–310. [Google Scholar] [CrossRef]
- Oliveira, H.; Sampaio, M.; Melo, L.D.R.; Dias, O.; Pope, W.H.; Hatfull, G.F.; Azeredo, J. Staphylococci phages display vast genomic diversity and evolutionary relationships. BMC Genom. 2019, 20, 357. [Google Scholar] [CrossRef]
- Xia, G.; Wolz, C. Phages of Staphylococcus aureus and their impact on host evolution. Infect. Genet. Evol. 2014, 21, 593–601. [Google Scholar] [CrossRef]
- Chang, Y.; Shin, H.; Lee, J.H.; Park, C.J.; Paik, S.Y.; Ryu, S. Isolation and Genome Characterization of the Virulent Staphylococcus aureus Bacteriophage SA97. Viruses 2015, 7, 5225–5242. [Google Scholar] [CrossRef] [PubMed]
- Culbertson, E.K.; Bari, S.M.N.; Dandu, V.S.; Kriznik, J.M.; Scopel, S.E.; Stanley, S.P.; Lackey, K.; Hernandez, A.C.; Hatoum-Aslan, A. Draft Genome Sequences of Staphylococcus Podophages JBug18, Pike, Pontiff, and Pabna. Microbiol. Resour. Announc. 2019, 8, e00054-19. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, J.; Taniguchi, M.; Kurokawa, K.; Takemura-Uchiyama, I.; Ujihara, T.; Shimakura, H.; Sakaguchi, Y.; Murakami, H.; Sakaguchi, M.; Matsuzaki, S. Adsorption of Staphylococcus viruses S13’ and S24-1 on Staphylococcus aureus strains with different glycosidic linkage patterns of wall teichoic acids. J. Gen. Virol. 2017, 98, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Łobocka, M.; Hejnowicz, M.S.; Dabrowski, K.; Gozdek, A.; Kosakowski, J.; Witkowska, M.; Ulatowska, M.I.; Weber-Dabrowska, B.; Kwiatek, M.; Parasion, S.; et al. Genomics of staphylococcal Twort-like phages-potential therapeutics of the post-antibiotic era. Adv. Virus Res. 2012, 83, 143–216. [Google Scholar] [CrossRef]
- Kornienko, M.; Bespiatykh, D.; Gorodnichev, R.; Abdraimova, N.; Shitikov, E. Transcriptional Landscapes of Herelleviridae Bacteriophages and Staphylococcus aureus during Phage Infection: An Overview. Viruses 2023, 15, 1427. [Google Scholar] [CrossRef]
- Dakheel, K.H.; Rahim, R.A.; Neela, V.K.; Al-Obaidi, J.R.; Hun, T.G.; Isa, M.N.M.; Yusoff, K. Genomic analyses of two novel biofilm-degrading methicillin-resistant Staphylococcus aureus phages. BMC Microbiol. 2019, 19, 114. [Google Scholar] [CrossRef]
- Vybiral, D.; Takáč, M.; Loessner, M.; Witte, A.; Von Ahsen, U.; Bläsi, U. Complete nucleotide sequence and molecular characterization of two lytic Staphylococcus aureus phages: 44AHJD and P68. FEMS Microbiol. Lett. 2003, 219, 275–283. [Google Scholar] [CrossRef]
- D’Souza, R.; White, R.C.; Buzzeo, R.; Goglin, K.; Vashee, S.; Lee, Y.; Son, B.; Ryu, S.; Fouts, D.E. Complete Genome Sequence of Staphylococcus aureus Phage SA75, Isolated from Goat Feces. Microbiol. Resour. Announc. 2020, 9, e00114-20. [Google Scholar] [CrossRef] [PubMed]
- Iandolo, J.J.; Worrell, V.; Groicher, K.H.; Qian, Y.; Tian, R.; Kenton, S.; Dorman, A.; Ji, H.; Lin, S.; Loh, P.; et al. Comparative analysis of the genomes of the temperate bacteriophages φ11, φ12 and φ13 of Staphylococcus aureus 8325. Gene 2002, 289, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Feng, T.; Gu, F.; Li, Q.; Dong, K.; Zhang, Y.; Zhu, Y.; Han, L.; Qin, J.; Guo, X. Characterization and complete genome of the virulent Myoviridae phage JD007 active against a variety of Staphylococcus aureus isolates from different hospitals in Shanghai, China. Virol. J. 2017, 14, 26. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.G.; Jun, J.W.; Giri, S.S.; Yun, S.; Kim, H.J.; Chi, C.; Kim, S.W.; Park, S.C. Complete Genome Sequence of Staphylococcus aureus Bacteriophage pSa-3. Genome Announc. 2017, 5, e00182-17. [Google Scholar] [CrossRef]
- Moller, A.G.; Lindsay, J.A.; Read, T.D. Determinants of phage host range in Staphylococcus species. Appl. Environ. Microbiol. 2019, 85, e00209-19. [Google Scholar] [CrossRef]
- Zhang, Q.; Xing, S.; Sun, Q.; Pei, G.; Cheng, S.; Liu, Y.; An, X.; Zhang, X.; Qu, Y.; Tong, Y. Characterization and complete genome sequence analysis of a novel virulent Siphoviridae phage against Staphylococcus aureus isolated from bovine mastitis in Xinjiang, China. Virus Genes 2017, 53, 464–476. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, F.; Sun, H.; Wang, Q.; Zhang, C.; Liu, W.; Zou, L.; Pan, Q.; Ren, H. Isolation and characterization of the Staphylococcus aureus bacteriophage vB_SauS_SA2. AIMS Microbiol. 2019, 5, 285. [Google Scholar] [CrossRef]
- Kornienko, M.; Kuptsov, N.; Gorodnichev, R.; Bespiatykh, D.; Guliaev, A.; Letarova, M.; Kulikov, E.; Veselovsky, V.; Malakhova, M.; Letarov, A.; et al. Contribution of Podoviridae and Myoviridae bacteriophages to the effectiveness of anti-staphylococcal therapeutic cocktails. Sci. Rep. 2020, 10, 18612. [Google Scholar] [CrossRef]
- Liu, S.; Hon, K.; Bouras, G.S.; Psaltis, A.J.; Shearwin, K.; Wormald, P.-J.; Vreugde, S. APTC-C-SA01: A Novel Bacteriophage Cocktail Targeting Staphylococcus aureus and MRSA Biofilms. Int. J. Mol. Sci. 2022, 23, 6116. [Google Scholar] [CrossRef]
- García, P.; Madera, C.; Martínez, B.; Rodríguez, A.; Evaristo Suárez, J. Prevalence of bacteriophages infecting Staphylococcus aureus in dairy samples and their potential as biocontrol agents. J. Dairy Sci. 2009, 92, 3019–3026. [Google Scholar] [CrossRef]
- Wdowiak, M.; Paczesny, J.; Raza, S. Enhancing the Stability of Bacteriophages Using Physical, Chemical, and Nano-Based Approaches: A Review. Pharmaceutics 2022, 14, 1936. [Google Scholar] [CrossRef]
- Organisation for Economic Co-operation and Development (OECD). Guidance Document for the Regulatory Framework for the Microorganism Group: Bacteriophages; Paris. 2022. Available online: https://www.oecd.org/content/dam/oecd/en/publications/reports/2022/11/guidance-document-for-the-regulatory-framework-for-the-microorganism-group-bacteriophages_92fbf702/706035c7-en.pdf (accessed on 15 January 2025).
- Fortuna, K.J.; Szoboszlay, M.; Holtappels, D.; Lavigne, R.; Tebbe, C.C.; Wagemans, J. Assessing the environmental biosafety of phage-based biocontrol applications. Biol. Control 2023, 187, 105375. [Google Scholar] [CrossRef]
- Cristobal-Cueto, P.; García-Quintanilla, A.; Esteban, J.; García-Quintanilla, M. Phages in Food Industry Biocontrol and Bioremediation. Antibiotics 2021, 10, 786. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hameid Ahmed, A.; Saad Maharik, N.M.; Valero, A.; Kamal, S.M. Incidence of enterotoxigenic Staphylococcus aureus in milk and Egyptian artisanal dairy products. Food Control 2019, 104, 20–27. [Google Scholar] [CrossRef]
- Osman, K.M.; Pires, Á.D.S.; Franco, O.L.; Orabi, A.; Hanafy, M.H.; Marzouk, E.; Hussien, H.; Alzaben, F.A.; Almuzaini, A.M.; Elbehiry, A. Enterotoxigenicity and Antibiotic Resistance of Coagulase-Negative Staphylococci Isolated from Raw Buffalo and Cow Milk. Microb. Drug Resist. 2020, 26, 520–530. [Google Scholar] [CrossRef]
- Dai, J.; Wu, S.; Huang, J.; Wu, Q.; Zhang, F.; Zhang, J.; Wang, J.; Ding, Y.; Zhang, S.; Yang, X.; et al. Prevalence and Characterization of Staphylococcus aureus Isolated from Pasteurized Milk in China. Front. Microbiol. 2019, 10, 641. [Google Scholar] [CrossRef]
- García-Anaya, M.C.; Sepulveda, D.R.; Sáenz-Mendoza, A.I.; Rios-Velasco, C.; Zamudio-Flores, P.B.; Acosta-Muñiz, C.H. Phages as biocontrol agents in dairy products. Trends Food Sci. Technol. 2020, 95, 10–20. [Google Scholar] [CrossRef]
- Ali Gharieb, R.M.; Saad, M.F.; Mohamed, A.S.; Tartor, Y.H. Characterization of two novel lytic bacteriophages for reducing biofilms of zoonotic multidrug-resistant Staphylococcus aureus and controlling their growth in milk. LWT 2020, 124, 109145. [Google Scholar] [CrossRef]
- Tahir, A.; Asif, M.; Abbas, Z.; ur Rehman, S. Three Bacteriophages SA, SA2 and SNAF can Control Growth of Milk Isolated Staphylococcal Species. Pak. J. Zool. 2017, 49, 493–496. [Google Scholar] [CrossRef]
- Mohammadian, F.; Rahmani, H.K.; Bidarian, B.; Khoramian, B. Isolation and evaluation of the efficacy of bacteriophages against multidrug-resistant (MDR), methicillin-resistant (MRSA) and biofilm-producing strains of Staphylococcus aureus recovered from bovine mastitis. BMC Vet. Res. 2022, 18, 406. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Bai, J.; Lee, J.H.; Ryu, S. Mutation of a Staphylococcus aureus temperate bacteriophage to a virulent one and evaluation of its application. Food Microbiol. 2019, 82, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Titze, I.; Lehnherr, T.; Lehnherr, H.; Krömker, V. Efficacy of Bacteriophages Against Staphylococcus aureus Isolates from Bovine Mastitis. Pharmaceuticals 2020, 13, 35. [Google Scholar] [CrossRef]
- Duc, H.M.; Son, H.M.; Ngan, P.H.; Sato, J.; Masuda, Y.; Honjoh, K.I.; Miyamoto, T. Isolation and application of bacteriophages alone or in combination with nisin against planktonic and biofilm cells of Staphylococcus aureus. Appl. Microbiol. Biotechnol. 2020, 104, 5145–5158. [Google Scholar] [CrossRef]
- Mani, I. Chapter Seven—Phage and Phage Cocktails Formulations. In Phage Therapy—Part A; Singh, V., Ed.; Academic Press: Amsterdam, The Netherlands, 2023; Volume 200, pp. 159–169. ISBN 1877-1173. [Google Scholar]
- Rodas Pazmiño, K.; Pazmiño Gómez, B.; Rodas Neira, E.; Cagua Montaño, L. Presencia de Staphylococcus aureus en quesos comercializados en la ciudad de Milagro, octubre—noviembre 2013. Cumbres 2016, 2, 25–29. [Google Scholar] [CrossRef]
- El Haddad, L.; Roy, J.P.; Khalil, G.E.; St-Gelais, D.; Champagne, C.P.; Labrie, S.; Moineau, S. Efficacy of two Staphylococcus aureus phage cocktails in cheese production. Int. J. Food Microbiol. 2016, 217, 7–13. [Google Scholar] [CrossRef]
- Kwak, H.; Kim, J.; Ryu, S.; Bai, J. Characterization of KMSP1, a newly isolated virulent bacteriophage infecting Staphylococcus aureus, and its application to dairy products. Int. J. Food Microbiol. 2023, 390, 110119, Erratum in Int. J. Food Microbiol. 2024, 412, 110558. [Google Scholar] [CrossRef]
- Weng, S.; López, A.; Sáez-Orviz, S.; Marcet, I.; García, P.; Rendueles, M.; Díaz, M. Effectiveness of bacteriophages incorporated in gelatine films against Staphylococcus aureus. Food Control 2021, 121, 107666. [Google Scholar] [CrossRef]
- Guenther, S.; Huwyler, D.; Richard, S.; Loessner, M.J. Virulent bacteriophage for efficient biocontrol of Listeria monocytogenes in ready-to-eat foods. Appl. Environ. Microbiol. 2009, 75, 93–100. [Google Scholar] [CrossRef]
- Oliveira, M.; Viñas, I.; Colàs, P.; Anguera, M.; Usall, J.; Abadias, M. Effectiveness of a bacteriophage in reducing Listeria monocytogenes on fresh-cut fruits and fruit juices. Food Microbiol. 2014, 38, 137–142. [Google Scholar] [CrossRef]
- Bueno, E.; García, P.; Martínez, B.; Rodríguez, A. Phage inactivation of Staphylococcus aureus in fresh and hard-type cheeses. Int. J. Food Microbiol. 2012, 158, 23–27. [Google Scholar] [CrossRef]
- Kandil, S.T. Biocontrol of Pathogenic Bacteria in Egyptian Cheeses by Bacteriophages; Harper Adams University: Newport, UK, 2023. [Google Scholar]
- Kukhtyn, M.; Kremenchuk, I.; Horiuk, Y.; Salata, V.; Kochetova, H.; Kladnytska, L.; Kozhyn, V.; Matviishyn, T. Development and evaluation of technology for preserving hardcheese with staphylococcal bacteriophage. Scifood 2025, 19, 208–223. [Google Scholar] [CrossRef]
- Ali, S.S.; Moawad, M.S.; Hussein, M.A.; Azab, M.; Abdelkarim, E.A.; Badr, A.; Sun, J.; Khalil, M. Efficacy of metal oxide nanoparticles as novel antimicrobial agents against multi-drug and multi-virulent Staphylococcus aureus isolates from retail raw chicken meat and giblets. Int. J. Food Microbiol. 2021, 344, 109116. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, K.; Tharwat, A.; Gharieb, R. High efficacy of a characterized lytic bacteriophage in combination with thyme essential oil against multidrug-resistant Staphylococcus aureus in chicken products. Iran. J. Vet. Res. 2021, 22, 24. [Google Scholar] [CrossRef] [PubMed]
- Agún, S.; Fernández, L.; Rodríguez, A.; García, P. Deletion of the amidase domain of endolysin LysRODI enhances antistaphylococcal activity in milk and during fresh cheese production. Food Microbiol. 2022, 107, 104067. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, H.; Azeredo, J.; Lavigne, R.; Kluskens, L.D. Bacteriophage endolysins as a response to emerging foodborne pathogens. Trends Food Sci. Technol. 2012, 28, 103–115. [Google Scholar] [CrossRef]
- Schmelcher, M.; Donovan, D.M.; Loessner, M.J. Bacteriophage endolysins as novel antimicrobials. Future Microbiol. 2012, 7, 1147–1171. [Google Scholar] [CrossRef]
- São-José, C. Engineering of Phage-Derived Lytic Enzymes: Improving Their Potential as Antimicrobials. Antibiotics 2018, 7, 29, Erratum in Antibiotics 2018, 7, 56. [Google Scholar] [CrossRef]
- Xu, Y. Phage and phage lysins: New era of bio-preservatives and food safety agents. J. Food Sci. 2021, 86, 3349–3373. [Google Scholar] [CrossRef]
- Harshitha, N.; Rajasekhar, A.; Saurabh, S.; Sonalkar, R.; Tejashwini, M.; Mitra, S. Das Bacteriophages: Potential Biocontrol Agents and Treatment Options for Bacterial Pathogens. Clin. Microbiol. Newsl. 2022, 44, 41–50. [Google Scholar] [CrossRef]
- Lee, C.; Kim, H.; Ryu, S. Bacteriophage and endolysin engineering for biocontrol of food pathogens/pathogens in the food: Recent advances and future trends. Crit. Rev. Food Sci. Nutr. 2022, 63, 8919–8938. [Google Scholar] [CrossRef] [PubMed]
- Cha, Y.; Son, B.; Ryu, S. Effective removal of staphylococcal biofilms on various food contact surfaces by Staphylococcus aureus phage endolysin LysCSA13. Food Microbiol. 2019, 84, 103245. [Google Scholar] [CrossRef]
- Yu, J.H.; Lim, J.A.; Chang, H.J.; Park, J.H. Characteristics and Lytic Activity of Phage-Derived Peptidoglycan Hydrolase, LysSAP8, as a Potent Alternative Biocontrol Agent for Staphylococcus aureus. J. Microbiol. Biotechnol. 2019, 29, 1916–1924. [Google Scholar] [CrossRef] [PubMed]
- Knaack, D.; Idelevich, E.A.; Schleimer, N.; Molinaro, S.; Kriegeskorte, A.; Peters, G.; Becker, K. Bactericidal activity of bacteriophage endolysin HY-133 against Staphylococcus aureus in comparison to other antibiotics as determined by minimum bactericidal concentrations and time-kill analysis. Diagn. Microbiol. Infect. Dis. 2019, 93, 362–368. [Google Scholar] [CrossRef]
- Traczewski, M.M.; Ambler, J.E.; Schuch, R. Determination of mic quality control parameters for exebacase, a novel lysin with antistaphylococcal activity. J. Clin. Microbiol. 2021, 59, 10–1128. [Google Scholar] [CrossRef]
- Kim, S.; Jin, J.S.; Choi, Y.J.; Kim, J. LysSAP26, a New Recombinant Phage Endolysin with a Broad Spectrum Antibacterial Activity. Viruses 2020, 12, 1340. [Google Scholar] [CrossRef]
- Kashani, H.H.; Fahimi, H.; Goli, Y.D.; Moniri, R. A novel chimeric endolysin with antibacterial activity against methicillin-resistant Staphylococcus aureus. Front. Cell. Infect. Microbiol. 2017, 7, 290. [Google Scholar] [CrossRef]
- Hathaway, H.; Ajuebor, J.; Stephens, L.; Coffey, A.; Potter, U.; Sutton, J.M.; Jenkins, A.T.A. Thermally triggered release of the bacteriophage endolysin CHAPK and the bacteriocin lysostaphin for the control of methicillin resistant Staphylococcus aureus (MRSA). J. Control. Release 2017, 245, 108–115. [Google Scholar] [CrossRef]
- Salas, M.; Wernecki, M.; Fernández, L.; Iglesias, B.; Gutiérrez, D.; Álvarez, A.; García, L.; Prieto, E.; García, P.; Rodríguez, A. Characterization of Clinical MRSA Isolates from Northern Spain and Assessment of Their Susceptibility to Phage-Derived Antimicrobials. Antibiotics 2020, 9, 447. [Google Scholar] [CrossRef]
- Shannon, R.; Radford, D.R.; Balamurugan, S. Impacts of food matrix on bacteriophage and endolysin antimicrobial efficacy and performance. Crit. Rev. Food Sci. Nutr. 2020, 60, 1631–1640. [Google Scholar] [CrossRef]
- Yan, J.; Yang, R.; Yu, S.; Zhao, W. The strategy of biopreservation of meat product against MRSA using lytic domain of lysin from Staphylococcus aureus bacteriophage. Food Biosci. 2021, 41, 100967. [Google Scholar] [CrossRef]
- Chang, Y.; Yoon, H.; Kang, D.H.; Chang, P.S.; Ryu, S. Endolysin LysSA97 is synergistic with carvacrol in controlling Staphylococcus aureus in foods. Int. J. Food Microbiol. 2017, 244, 19–26. [Google Scholar] [CrossRef]
- Son, B.; Kong, M.; Lee, Y.; Ryu, S. Development of a Novel Chimeric Endolysin, Lys109 with Enhanced Lytic Activity Against Staphylococcus aureus. Front. Microbiol. 2021, 11, 615887. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Kim, M.; Ryu, S. Characterization of a novel endolysin LysSA11 and its utility as a potent biocontrol agent against Staphylococcus aureus on food and utensils. Food Microbiol. 2017, 68, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Obeso, J.; Martínez, B.; Rodríguez, A.; García, P. Lytic activity of the recombinant staphylococcal bacteriophage ΦH5 endolysin active against Staphylococcus aureus in milk. Int. J. Food Microbiol. 2008, 128, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Xin, Y.P.; Zhang, C.; Ouyang, X.; Kong, J. The potential of the endolysin Lysdb from Lactobacillus delbrueckii phage for combating Staphylococcus aureus during cheese manufacture from raw milk. Appl. Microbiol. Biotechnol. 2016, 100, 3545–3554. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Varoni, E.M.; Iriti, M.; Martorell, M.; Setzer, W.N.; del Mar Contreras, M.; Salehi, B.; Soltani-Nejad, A.; Rajabi, S.; Tajbakhsh, M.; et al. Carvacrol and human health: A comprehensive review. Phytother. Res. 2018, 32, 1675–1687. [Google Scholar] [CrossRef]
- Tabla, R.; Martínez, B.; Rebollo, J.E.; González, J.; Ramírez, M.R.; Roa, I.; Rodríguez, A.; García, P. Bacteriophage performance against Staphylococcus aureus in milk is improved by high hydrostatic pressure treatments. Int. J. Food Microbiol. 2012, 156, 209–213. [Google Scholar] [CrossRef]
- Han, J.E.; Kim, J.H.; Hwang, S.Y.; Choresca, C.H.; Shin, S.P.; Jun, J.W.; Chai, J.Y.; Park, Y.H.; Park, S.C. Isolation and characterization of a Myoviridae bacteriophage against Staphylococcus aureus isolated from dairy cows with mastitis. Res. Vet. Sci. 2013, 95, 758–763. [Google Scholar] [CrossRef]
- Kim, S.G.; Giri, S.S.; Yun, S.; Kim, H.J.; Kim, S.W.; Kang, J.W.; Han, S.J.; Kwon, J.; Oh, W.T.; Jun, J.W.; et al. Synergistic phage-surfactant combination clears IgE-promoted Staphylococcus aureus aggregation in vitro and enhances the effect in vivo. Int. J. Antimicrob. Agents 2020, 56, 105997. [Google Scholar] [CrossRef]
- Son, B.; Kong, M.; Cha, Y.; Bai, J.; Ryu, S. Simultaneous Control of Staphylococcus aureus and Bacillus cereus Using a Hybrid Endolysin LysB4EAD-LysSA11. Antibiotics 2020, 9, 906. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Huang, C.; Qiu, C.; Wang, Z.; Cheng, M.; Zhang, Y.; Qiao, Y.; Guan, Y.; Feng, X.; Sun, C.; et al. Rapid and sensitive detection of Staphylococcus aureus using biolayer interferometry technology combined with phage lysin LysGH15. Biosens. Bioelectron. 2022, 198, 113799. [Google Scholar] [CrossRef] [PubMed]
- Rogovski, P.; Cadamuro, R.D.; da Silva, R.; de Souza, E.B.; Bonatto, C.; Viancelli, A.; Michelon, W.; Elmahdy, E.M.; Treichel, H.; Rodríguez-Lázaro, D.; et al. Uses of Bacteriophages as Bacterial Control Tools and Environmental Safety Indicators. Front. Microbiol. 2021, 12, 793135. [Google Scholar] [CrossRef]
- De Melo, A.G.; Levesque, S.; Moineau, S. Phages as friends and enemies in food processing. Curr. Opin. Biotechnol. 2018, 49, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Efenberger-Szmechtyk, M.; Nowak, A. Bacteriophage Power: Next-Gen Biocontrol Strategies for Safer Meat. Molecules 2025, 30, 3641. [Google Scholar] [CrossRef]
- Dhulipalla, H.; Basavegowda, N.; Haldar, D.; Syed, I.; Ghosh, P.; Rana, S.S.; Somu, P.; Naidu, R.; Yadav, A.K.; Lee, M.-J.; et al. Integrating phage biocontrol in food production: Industrial implications and regulatory overview. Discov. Appl. Sci. 2025, 7, 314. [Google Scholar] [CrossRef]
- Anyaegbunam, N.J.; Anekpo, C.C.; Anyaegbunam, Z.K.G.; Doowuese, Y.; Chinaka, C.B.; Odo, O.J.; Sharndama, H.C.; Okeke, O.P.; Mba, I.E. The resurgence of phage-based therapy in the era of increasing antibiotic resistance: From research progress to challenges and prospects. Microbiol. Res. 2022, 264, 127155. [Google Scholar] [CrossRef]
- Cass, J.; Barnard, A.; Fairhead, H. Engineered Bacteriophage as a Delivery Vehicle for Antibacterial Protein, SASP. Pharmaceuticals 2021, 14, 1038. [Google Scholar] [CrossRef]
- Fabijan, A.P.; Lin, R.C.; Ho, J.; Maddocks, S.; Zakour, N.L.b.; Iredell, J.R. Safety of bacteriophage therapy in severe Staphylococcus aureus infection. Nat. Microbiol. 2020, 5, 465–472, Erratum in Nat. Microbiol. 2020, 5, 652. [Google Scholar] [CrossRef]
- Kifelew, L.G.; Warner, M.S.; Morales, S.; Vaughan, L.; Woodman, R.; Fitridge, R.; Mitchell, J.G.; Speck, P. Efficacy of phage cocktail AB-SA01 therapy in diabetic mouse wound infections caused by multidrug-resistant Staphylococcus aureus. BMC Microbiol. 2020, 20, 204. [Google Scholar] [CrossRef] [PubMed]
- Połaska, M.; Sokołowska, B. Bacteriophages—A new hope or a huge problem in the food industry. AIMS Microbiol. 2019, 5, 324–346. [Google Scholar] [CrossRef] [PubMed]
- Alomari, M.M.M.; Dec, M.; Urban-Chmiel, R. Bacteriophages as an alternative method for control of zoonotic and foodborne pathogens. Viruses 2021, 13, 2348. [Google Scholar] [CrossRef] [PubMed]
- Zschach, H.; Joensen, K.G.; Lindhard, B.; Lund, O.; Goderdzishvili, M.; Chkonia, I.; Jgenti, G.; Kvatadze, N.; Alavidze, Z.; Kutter, E.M.; et al. What Can We Learn from a Metagenomic Analysis of a Georgian Bacteriophage Cocktail? Viruses 2015, 7, 6570–6589. [Google Scholar] [CrossRef]
- Erol, H.B.; Kaskatepe, B.; Bakkaloglu, Z.; Suzuk Yildiz, S. The evaluation of five commercial bacteriophage cocktails against methicillin-resistant Staphylococcus aureus isolated from nasal swab samples. Arch. Microbiol. 2021, 203, 5735–5743. [Google Scholar] [CrossRef]
- Kim, N.H.; Park, W.B.; Cho, J.E.; Choi, Y.J.; Choi, S.J.; Jun, S.Y.; Kang, C.K.; Song, K.H.; Choe, P.G.; Bang, J.H.; et al. Effects of Phage Endolysin SAL200 Combined with Antibiotics on Staphylococcus aureus Infection. Antimicrob. Agents Chemother. 2018, 62, e00731-18. [Google Scholar] [CrossRef]
- Totté, J.E.E.; van Doorn, M.B.; Pasmans, S.G.M.A. Successful Treatment of Chronic Staphylococcus aureus-Related Dermatoses with the Topical Endolysin Staphefekt SA.100: A Report of 3 Cases. Case Rep. Dermatol. 2017, 9, 19–25. [Google Scholar] [CrossRef]
- Kaur, S.; Harjai, K.; Chhibber, S. Methicillin-Resistant Staphylococcus aureus Phage Plaque Size Enhancement Using Sublethal Concentrations of Antibiotics. Appl. Environ. Microbiol. 2012, 78, 8227–8233. [Google Scholar] [CrossRef]
- Rakieten, M.L.; Rakieten, T.L.; Doff, S. Absorption of Staphylococcus Bacteriophages. J. Bacteriol. 1936, 32, 505–518. [Google Scholar] [CrossRef]
- Pattee, P.A. Use of tetrazolium for improved resolution of bacteriophage plaques. J. Bacteriol. 1966, 92, 787–788. [Google Scholar] [CrossRef]
- Mattila, S.; Ruotsalainen, P.; Jalasvuori, M. On-Demand Isolation of Bacteriophages Against Drug-Resistant Bacteria for Personalized Phage Therapy. Front. Microbiol. 2015, 6, 1271. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Xu, W.; Lei, L.; Huang, J.; Feng, X.; Sun, C.; Du, C.; Zuo, J.; Li, Y.; Du, T.; et al. LysGH15, a novel bacteriophage lysin, protects a murine bacteremia model efficiently against lethal methicillin-resistant Staphylococcus aureus infection. J. Clin. Microbiol. 2011, 49, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Son, B.; Cha, Y.; Ryu, S. Characterization and Genomic Analysis of PALS2, a Novel Staphylococcus Jumbo Bacteriophage. Front. Microbiol. 2021, 12, 622755. [Google Scholar] [CrossRef] [PubMed]
- Klobucar, K.; Brown, E.D. New potentiators of ineffective antibiotics: Targeting the Gram-negative outer membrane to overcome intrinsic resistance. Curr. Opin. Chem. Biol. 2022, 66, 102099. [Google Scholar] [CrossRef]
- Jiang, Y.; Xu, D.; Wang, L.; Qu, M.; Li, F.; Tan, Z.; Yao, L. Characterization of a broad-spectrum endolysin LysSP1 encoded by a Salmonella bacteriophage. Appl. Microbiol. Biotechnol. 2021, 105, 5461–5470. [Google Scholar] [CrossRef]
- Rahman, M.U.; Wang, W.; Sun, Q.; Shah, J.A.; Li, C.; Sun, Y.; Li, Y.; Zhang, B.; Chen, W.; Wang, S. Endolysin, a Promising Solution against Antimicrobial Resistance. Antibiotics 2021, 10, 1277. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).