Abstract
The paper focuses on the improved process of metal recovery from lithium-ion batteries (LIBs) lithium nickel manganese cobalt oxide (NMC) cathode waste materials by using hydrometallurgical methods. In the acid leaching step, the essential effects of acidity concentration, H2O2 concentration, leaching time, liquid-solid mass ratio, and reaction temperature with the leaching percentage were investigated in detail. The cathode material was leached with 2M H2SO4 and 10 vol. % H2O2 at 70 °C and 300 rpm using a liquid-solid mass ratio of 30 mL/g. In order to complete the recovery process, this paper designs the proper separation process to recover valuable metals. The leach liquor in the recovery process uses Cyanex 272 to first extract Co and Mn to the organic phase. Secondly, Co and Mn are separated by using D2EHPA, and a high purity of Co is obtained. Thirdly, Ni is selectively precipitated by using DMG, and Ni is completely formed as a solid complex. Finally, in the chemical precipitation process, the remaining Li in the leach liquor is recovered as Li2CO3 precipitated by saturated Na2CO3, and Co, Mn, and Ni are recovered as hydroxides by NaOH. This hydrometallurgical process may provide an effective separation and recovery of valuable metals from LIBs waste cathode materials.
1. Introduction
Nowadays, as a result of the rapid development of modern society and technology, the use of lithium-ion batteries (LIBs) has become indispensable. These are commonly applied in our lives and play an important role in power sources and diverse devices such as mobile phones, laptop computers, digital cameras, and even the developing electric vehicles (EVs) and hybrid electric vehicles (HEVs) [1,2,3,4]. In the period between 2000 and 2010, the annual production of LIBs increased by 800% worldwide [5]. With the popularity of LIBs’ development, the resulting use of LIBs is also growing prominently [6,7]. Consequently, the recycling of spent LIBs by means of the recovery of the valuable metals contained in the cathode material, such as lithium, cobalt, nickel, and manganese, is considered as a progressively more substantial process to prevent environmental problems and meet sustainable and environmentally friendly regulations.
LIBs are frequently classified according to their cathode materials, into lithium cobalt oxide (LiCoO2) batteries, lithium manganese oxide (LiMn2O4) batteries, and lithium iron phosphate (LiFePO4) batteries, for example [8,9,10,11,12]. However, these types of batteries are gradually being replaced by lithium nickel manganese cobalt oxide (LiNiMnCoO2 or NMC) batteries. Hence, in this study, we focus on dealing with the NMC cathode waste materials.
At present, the recycling process of LIBs has been investigated in several studies [13,14]. In order to recover the valuable metals from several types of LIBs, measures have been reported involving mechanical processes [15], mechano-chemical processes [16], thermal treatment [17,18], and dissolution processes. Subsequently, chemical processes mainly involve the use of hydrometallurgical operations to carry out the recovery of valuable metals; these include acid leaching [19,20,21], chemical precipitation [22,23], solvent extraction [24,25], ion-exchange [26], and electrochemistry [27]. Thus far, approaches for recycling valuable metals in LIBs mainly use pyrometallurgical and hydrometallurgical processes. In hydrometallurgical processes, the recovery of valuable metals from spent LIB cathode materials is dealt with via acid leaching. According to different leaching ways to cope with spent LIBs, this is mostly carried out using an inorganic acid as an acidic leaching agent, such as H2SO4 [28,29,30], HCl [31], or HNO3 [32]. Compared with those given in the literature [33], H2SO4 has a great effect on lithium cobalt oxide batteries and is cheaper than others. Therefore, we chose H2SO4 as the leaching agent to process the NMC cathode materials. The research focused on finding the most suitable acid concentration, reaction temperature, liquid-solid mass ratio, and other parameters.
In the separation process, mainly used are solvent extraction, ion-exchange, and chemical precipitation to cope with the waste materials. Because several extractants, resins, and precipitating agents, such as PC88A [34], D2EHPA [35], Cyanex 272/Cyanex 301/Cyanex 302 [36,37], Mextral272P [38], Dowex M4195, Diaion CR-11, Lewatit TP-272 [26], and dimethylglyoxime (DMG) [39,40], were experimented with, we found that the most common way to separate cobalt, nickel, and manganese from other sources is solvent extraction. In comparison with literature, as an extractant, Cyanex 272 has a great selectivity between nickel and cobalt; D2EHPA has the effect of separating cobalt and manganese. However, when the target materials are no longer only two metals, such as nickel/cobalt or cobalt/manganese, but four metals, such as nickel/cobalt/manganese/lithium, the effect of separation is limited by co-extraction. Furthermore, as a precipitating agent, DMG precipitates nickel very selectively in the absence of cobalt. Hence, we designed an improved recovery process and combined the advantages of the extractants and precipitating agent mentioned above to overcome the inadequate abilities, achieving effective results. In this study, we concentrate on discussing the experimental parameters and design a recycling process for LIB NMC cathode waste materials.
2. Materials and Methods
2.1. Materials and Reagents
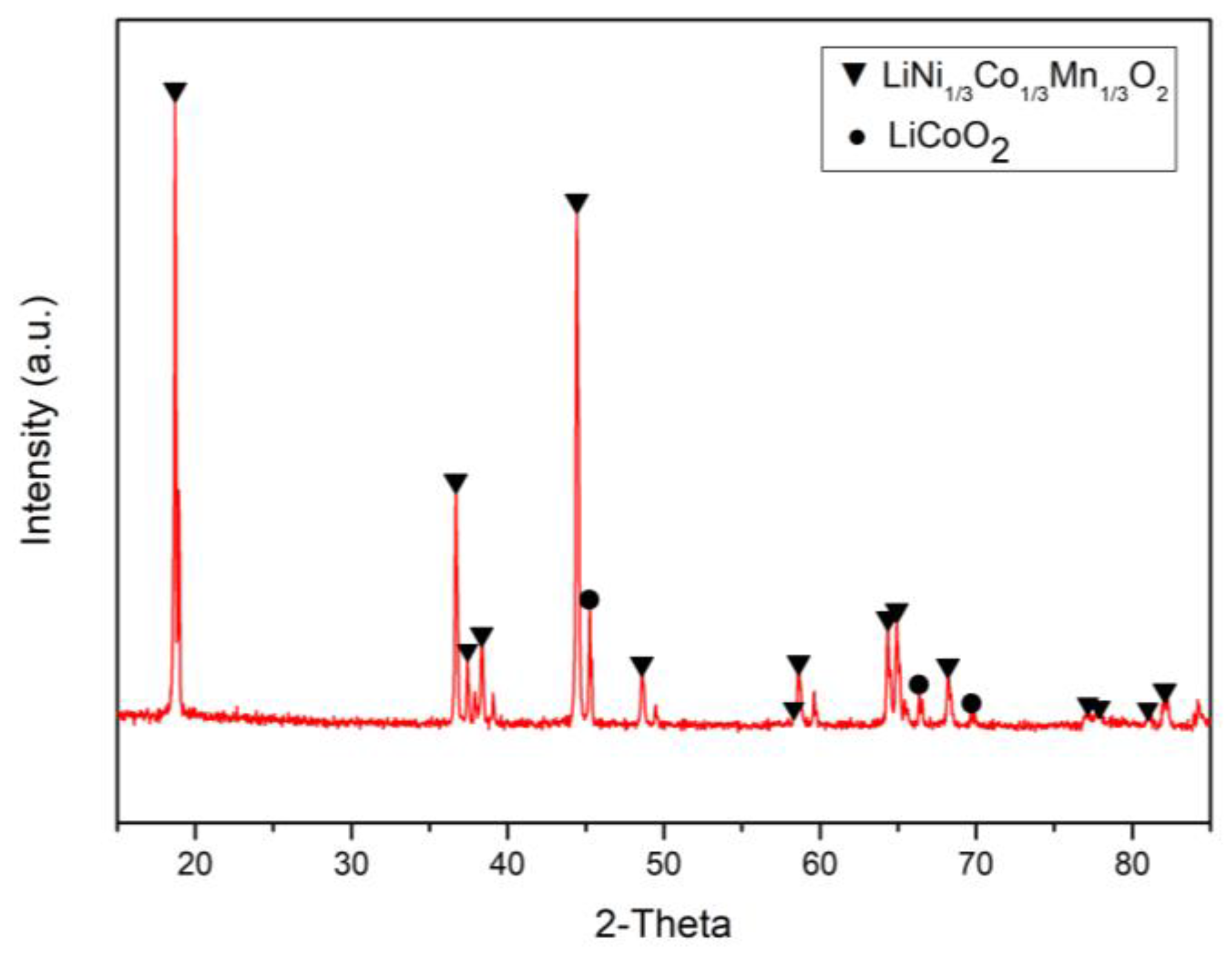
The NMC cathode waste materials mainly contain cobalt, nickel, manganese, and lithium. In our experiment, the source of materials was from the LIB industry, and these were produced and acquired during the manufacture of LIBs. The materials were analyzed by scanning electron microscopy (SEM; Hitachi, S-3000N), energy-dispersive X-ray spectroscopy (EDS; Bruker, XFlash6110), X-ray diffraction (XRD; Dandong DX-2700), and inductively coupled plasma optical emission spectrometry (ICP-OES; Varian, Vista-MPX). Figure 1 shows that the valuable metals in the cathode materials were present as LiCoO2 and LiNi1/3Co1/3Mn1/3O2. Figure 2a,b shows the result of the cathode material analysis by SEM and EDS. It was found that the metals were distributed evenly, and we could also determine the presence of LiCoO2 and LiNi1/3Co1/3Mn1/3O2. The chemical composition of the LIB cathode materials was analyzed by ICP-OES and mainly contained 25.83% Co, 26.29% Ni, 14.41% Mn, and 8.31% Li. Compared with previous literature [41], we found that the total amount of Co and Ni in this study was nearly 15% higher than in previous literature. Because of the similar chemical properties of Co and Ni, a large proportion of Co and Ni causes serious co-precipitation and co-extraction, as well as other negative effects. Therefore, an improved separation process is required in subsequent studies.
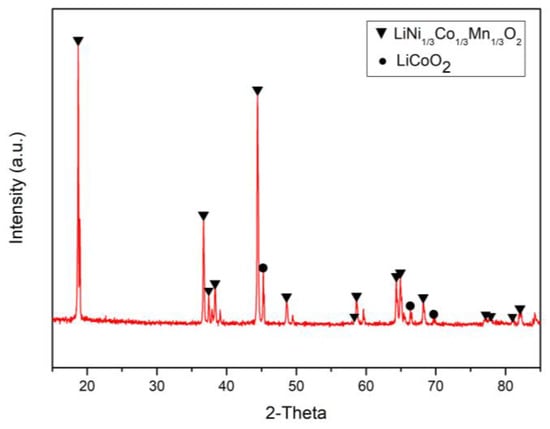
Figure 1.
The X-ray diffraction (XRD) pattern of waste lithium nickel manganese cobalt oxide (NMC) cathode materials.

Figure 2.
(a) The scanning electron microscopy (SEM) analysis of cathode materials; (b) the energy-dispersive X-ray spectroscopy (EDS) analysis of cathode materials.
The sulfuric acid used as the leaching agent and stripping agent in the experiment was from Sigma-Aldrich (St. Louis, MO, USA) (H2SO4, 98%) and was diluted in deionized water. The commercial extractants Cyanex 272 (CYTEC, 85%) and D2EHPA (Alfa Aesar, 95%) were diluted in kerosene, and both were saponified by the addition of a stoichiometric amount of sodium hydroxide solution. In this study, the preparation of Cyanex 272 and D2EHPA was saponified to 60% Na-Cyanex 272 and 50% Na-D2EHPA. Sodium hydroxide was from Showa (NaOH, 97%), and the selective precipitation reagent in this experiment was dimethylglyoxime (DMG, Sigma-Aldrich, ≥99%). All other chemical reagents used in the experiment were of analytical grade and were prepared or diluted with deionized water.
2.2. Leaching
Leaching procedures were carried out using standard laboratory leaching equipment. The cathode material was dissolved in sulfuric acid. The leaching parameters, such as the acid concentration, reaction temperature, reaction time, and liquid-solid mass ratio, were investigated. The acidity was set from 0.25 M to 8 M, the reducing reagent concentration was set from 0.2–21%, and the liquid-solid mass ratio was set from 2.5 mL/g to 50 mL/g. The effect of temperature was tested at different temperatures from 25 °C to 85 °C to achieve a better leaching percentage. The leaching percentage was calculated according to Equation (1):
where XB is the leaching percentage, m1 is the measured quantity of metal leached, and m2 is the quantity of metal in the raw material.
The following chemical equations of dissolution demonstrated that the cathode waste materials from LIBs were dissolved in the sulfuric acid solution with hydrogen peroxide:
2LiCoO2(s) + 3H2SO4(aq) + H2O2(aq)→2CoSO4(aq) + Li2SO4(aq) + 4H2O(g) + O2(g)
6LiNi1/3Mn1/3Co1/3O2(s) + 9H2SO4(aq) + H2O2(aq)→2MnSO4(aq) + 2NiSO4(aq) + 2CoSO4(aq) + 3Li2SO4(aq) + 10H2O(g) + 2O2(g)
2.3. Solvent Extraction
In the experiment, Na-Cyanex 272 was used as the extractant to efficiently separate cobalt and nickel in the sulfate solution. Na-D2EHPA was used as the extractant to entirely separate cobalt and manganese in the sulfate solution. The extractant was diluted into kerosene and was partially saponified by NaOH. The saponification reaction of Cyanex 272 and D2EHPA can be written as Equation (4), and the extraction mechanism of Na-Cyanex 272 and Na-D2EHPA can be written as Equation (5) [42,43,44]:
Na+(aq) + 1/2 (HA)2(org)→NaA(org) + H+(aq)
M2+(aq) + NaA(org) + 2(HA)2(org)↹(MA2•3HA)(org) + Na+(aq) + H+(aq)
The distribution ratio, D, was calculated as the concentration ratio of the metal present in the organic phase to that in the aqueous phase at equilibrium:
where C0 is an initial total concentration of metal ions in an aqueous phase; C is the equilibrium concentration of metal ions in an aqueous phase; and Vaq and Vorg are the volumes of the aqueous and organic phases, respectively.
From the distribution ratio, D, the extraction percentage, %E, could be calculated by Equation (7):
where D is the distribution ratio; and Vaq and Vorg are the volumes of the aqueous and organic phases, respectively.
2.4. Stripping Process
The stripping agent in the experiment was H2SO4, which was mixed with the organic phases after the solvent extraction step. The metals, such as cobalt ions and manganese ions, were stripped into the aqueous phase owing to their high solubility in H2SO4. After the first solvent extraction, the acidity of H2SO4 (0.01–0.15 mol/L) and the organic-aqueous ratio (0.5–4) of the stripped cobalt and manganese were investigated. After the second solvent extraction, the acidity of H2SO4 (0.005–0.15 mol/L) and the organic-aqueous ratio (1–8) of the stripped manganese were investigated.
2.5. Selective Precipitation and Chemical Precipitation
The selective precipitation process used DMG (C4H8N2O2) as the reagent to separate nickel and lithium. The molar ratio of C4H8N2O2 to nickel (MRDN) and the equilibrium pH value were adjusted respectively from 1 to 3 and 3 to 10 to obtain the best precipitation percentage under optimal parameters. In the chemical precipitation process, sodium hydroxide was dissolved in deionized water and modified the pH value. In addition, the best precipitation percentages of cobalt hydroxide, manganese hydroxide, and nickel hydroxide were investigated. The precipitation percentages were calculated by Equation (8):
where P is the precipitation percentage, is the metal concentration of the leach liquor, and is the metal concentration of the leach liquor after precipitation.
3. Results and Discussion
3.1. Leaching Process
3.1.1. Effect of Acid Concentration and H2O2 Concentration
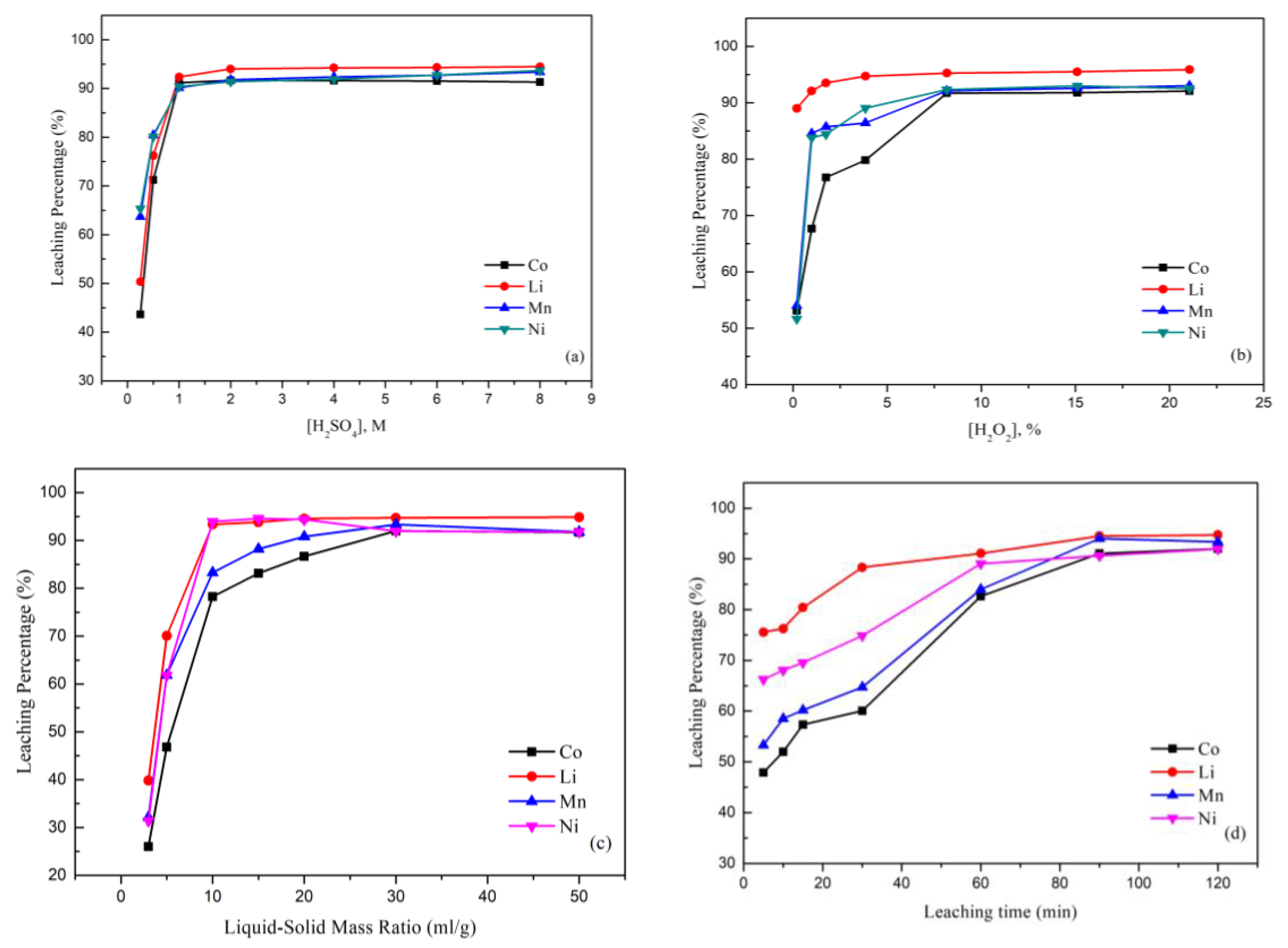
Figure 3a shows the leaching behavior of the metals cobalt, nickel, lithium, and manganese from spent LIB cathode materials by sulfuric acid and hydrogen peroxide. The effect of the H2SO4 concentration was investigated by varying the H2SO4 concentration from 0.25 M to 8.0 M. The results indicated that Co increased steeply from 43.7% to 91.6% as the H2SO4 concentration increased up to 2.0 M, while Mn, Ni, and Li respectively increased to 91.8%, 91.4%, and 94.0%. The effect was ascribed to the fact that a higher acid concentration assisted and speeded up the forward reaction, resulting in a higher leaching percentage [45].
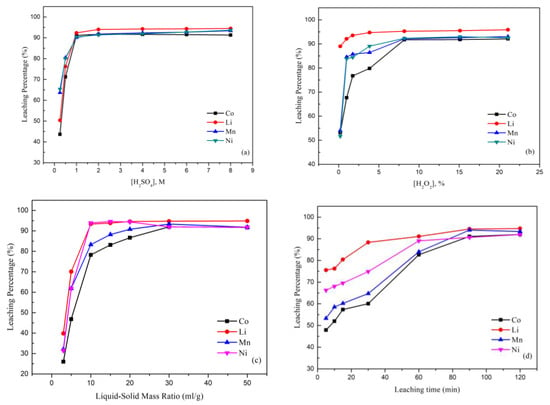
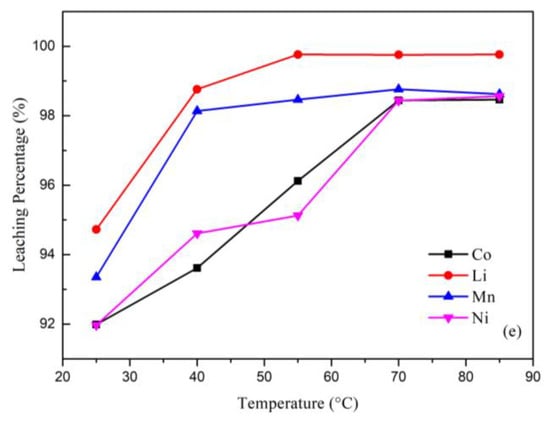
Figure 3.
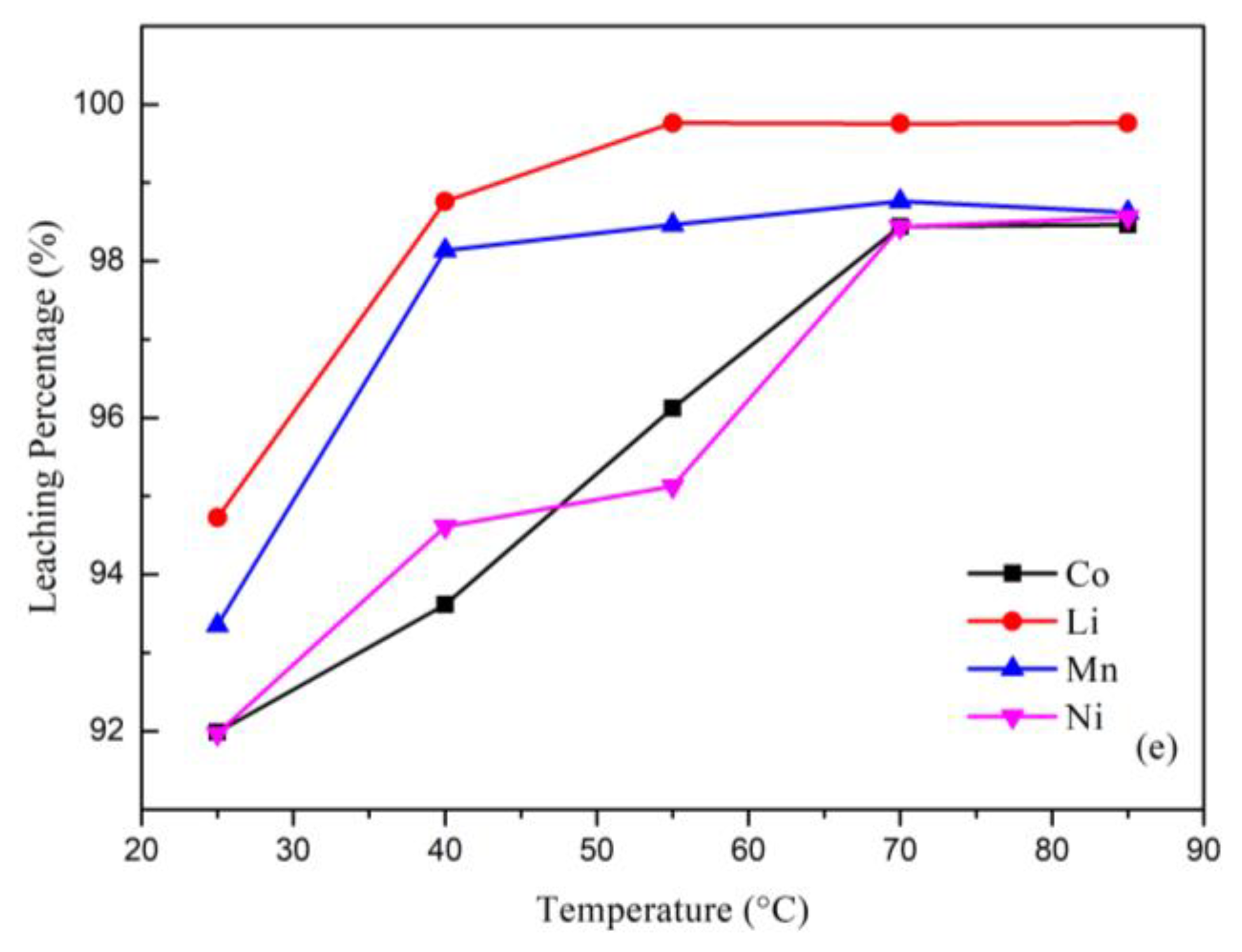
(a) Effect of the leaching percentage on H2SO4 concentration (reaction conditions: liquid-solid mass ratio of 50, 10.0% H2O2, 120 min, and 25 °C). (b) Effect of the leaching percentage on H2O2 concentration (reaction conditions: 2.0 M H2SO4, liquid-solid mass ratio of 50, 120 min, and 25 °C). (c) Effect of the leaching percentage on liquid-solid mass ratio (reaction conditions: 2.0 M H2SO4, 10.0% H2O2, 120 min, and 25 °C). (d) Effect of the leaching percentage on reaction time (reaction conditions: 2.0 M H2SO4, 10.0% H2O2, liquid-solid mass ratio of 30, and 25 °C). (e) Effect of the leaching percentage on temperature (reaction conditions: 2.0 M H2SO4, 10.0% H2O2, liquid-solid mass ratio of 30, and 90 min).
In order to examine the effect of the H2O2 concentration on the leaching process, the concentration of H2O2 was varied in the range from 0.2% to 21%. Figure 3b illustrates that the leaching efficiency of the metal significantly increased when the H2O2 concentration was 8.16%. The results indicated that the Co, Ni, Mn, and Li percentages steeply increased up to 91.72%, 92.34%, 92.12%, and 95.27%. This phenomenon was attributed to the fact that the reductions of Co3+ to Co2+ and Mn4+ to Mn2+ would help these metals to dissolve more readily. However, there was no apparent influence when H2O2 was added with a concentration of more than 10%. Therefore, the concentration of H2O2 was chosen as 10% to be optimal in the leaching process.
3.1.2. Effect of Liquid-Solid Mass Ratio
The effect of the liquid-solid mass ratio is shown in Figure 3c. The leaching percentages of all metals investigated were generally increased, while the liquid-solid mass ratio increased from 3/1 to 30/1 and percentages were increased to 91.99% for Co, 94.72% for Li, 93.35% for Mn, and 91.97% for Ni. The reason was that when the liquid-solid mass ratio was low, there was insufficient acid to react in the process. In other words, when the liquid-solid mass ratio was high, there was more acid readily able to react and available to obtain a higher leaching percentage. Hence, the liquid-solid mass ratio was chosen to be 30 mL/g as optimal.
3.1.3. Effect of Reaction Time and Temperature
Figure 3d shows the effect of the leaching percentage with the reaction time. The leaching percentages of Co, Li, Ni, and Mn substantially increased by about 44%, 19.8%, 25.7%, and 40.1% when the reaction time was increased from 5 to 90 min. The reason was that with the increase in the leaching time, a greater and greater surface area of the unreacted particle cores could react with the sulfuric acid.
The effect of the temperature is shown in Figure 3e. The leaching percentage increased with the increasing temperature because the temperature has a great effect on the leaching process. A higher temperature can increase the speed of the molecular motion and increase the energy of the particles’ collisions. The optimal leaching parameters from this study are illustrated in Table 1. The leaching percentages under the optimal conditions were Co: 98.46%; Ni: 98.56%; Li: 99.76%; and Mn: 98.62%.

Table 1.
The optimal parameters of leaching process.
3.2. Solvent Extraction with Na-Cyanex 272
Because NMC cathode waste material contains an extraordinarily large proportion of cobalt and nickel, we used Na-Cyanex 272 to first separate the nickel and cobalt. If the nickel-cobalt separation was not treated first, it would have been difficult to achieve a high purity of the product and would have impeded the following processes. Hence in this study, we used Na-Cyanex 272 to handle this problem. The extraction pH value was considered the key variable for separation in the extraction process. The metals’ concentration was set as the concentration ratio of Co/Ni/Mn/Li = 2590:2610:1400:800 mg/L; this was set according to the leaching condition and was analyzed by ICP-OES to calculate the extraction percentage.
3.2.1. Effect of Equilibrium pH Value
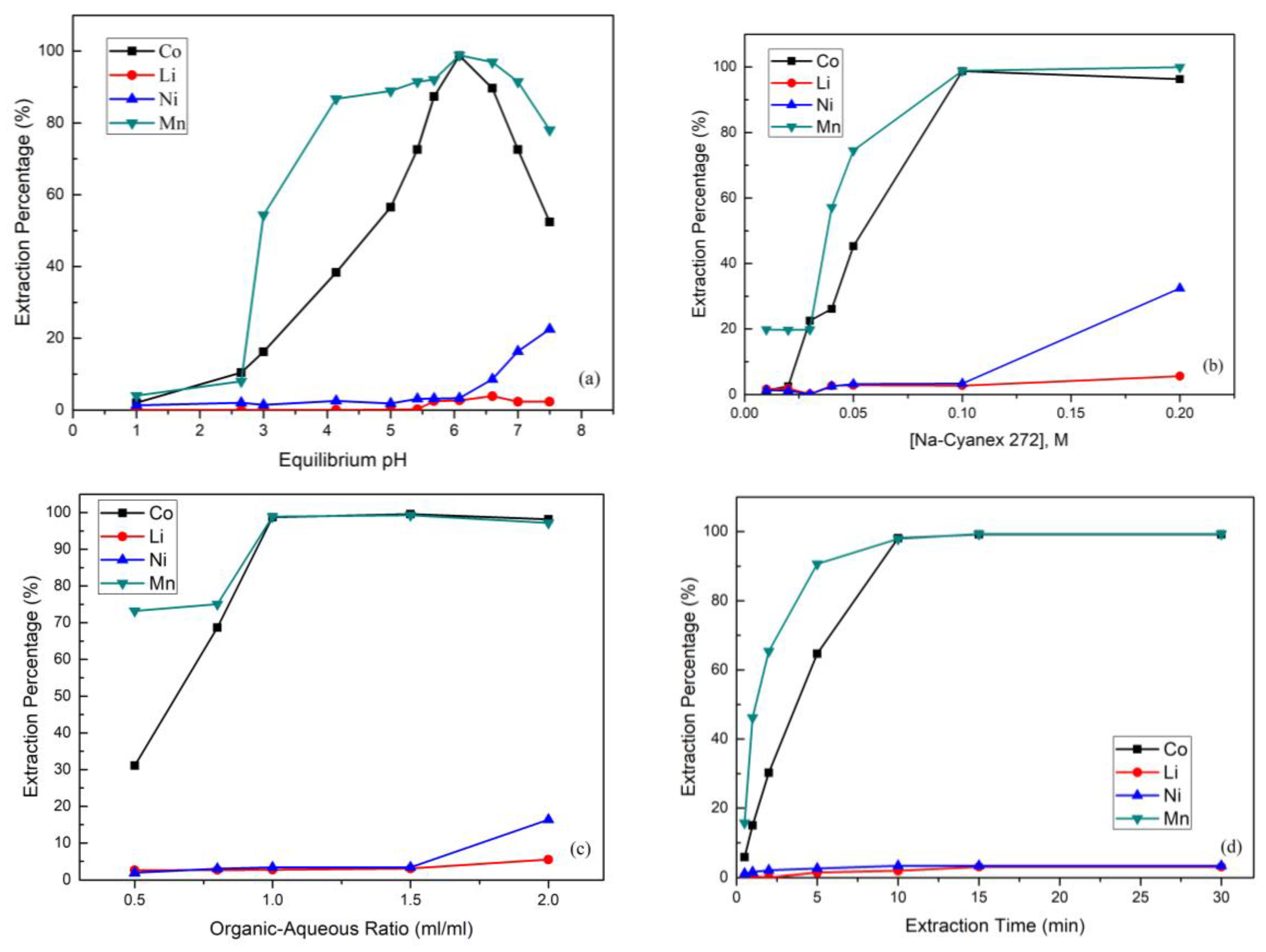
The equilibrium pH value was set from 1 to 7.5 by using 0.1 M 60% Na-Cyanex 272 with an organic-aqueous ratio of 1:1 over 15 min. Figure 4a shows that the extraction percentages of cobalt and manganese were observed to increase significantly from almost 0% to 98.8% and 98.9% respectively when the pH value was raised from 4 to 6. The extraction was not desirable when the pH value was higher than 6, as the extraction percentage of nickel began to increase rapidly. Moreover, when the pH value was higher than 7, cobalt began to partially precipitate to cobalt hydroxide. Hence, in this step, the equilibrium pH value equal set to 6 was optimal.
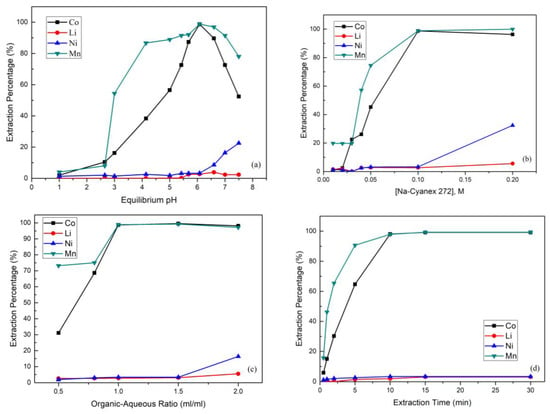
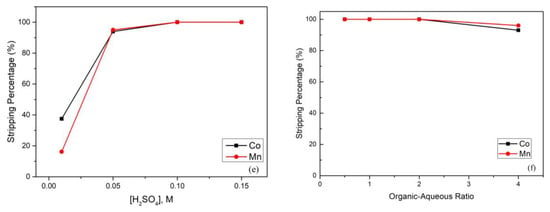
Figure 4.
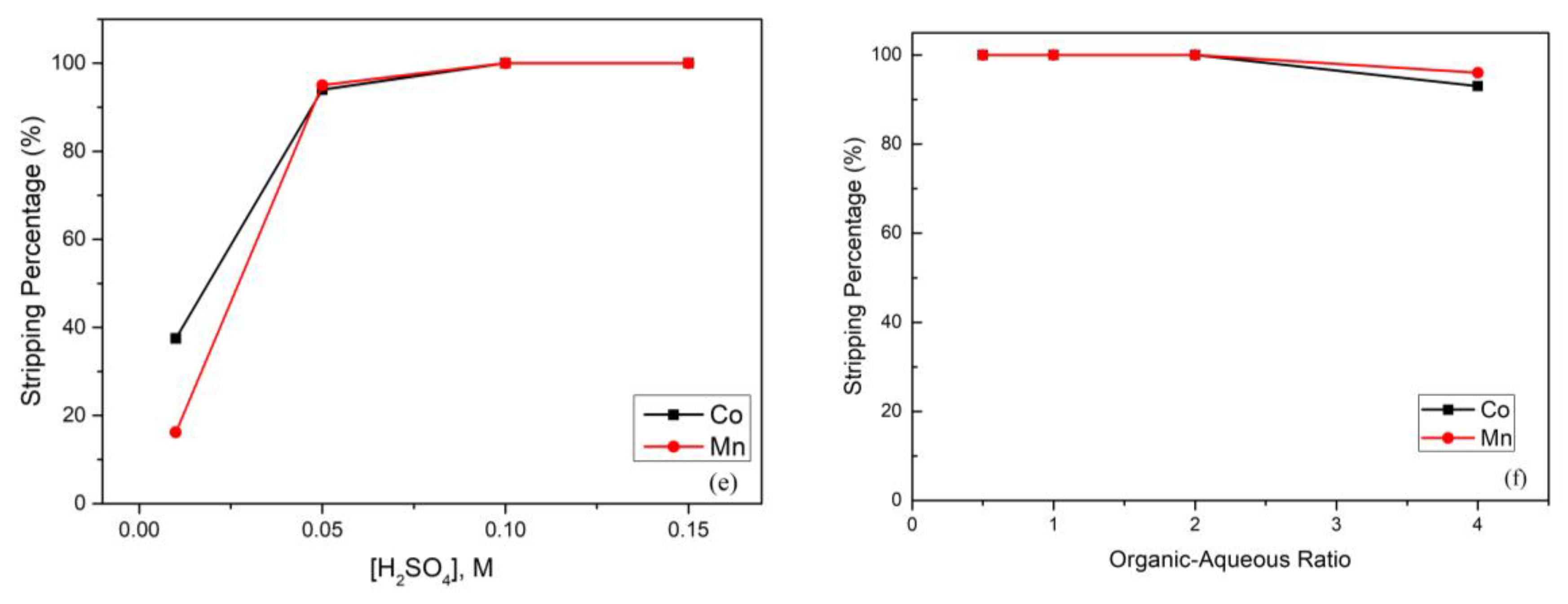
(a) Effect of the extraction percentage on equilibrium pH value (reaction conditions: 0.1 M Na-Cyanex 272, organic-aqueous ratio of 1, and 15 min). (b) Effect of the extraction percentage on Na-Cyanex 272 concentration (reaction conditions: equilibrium pH value of 6, organic-aqueous ratio of 1, and 15 min). (c) Effect of the extraction percentage on organic-aqueous ratio (reaction conditions: equilibrium pH value of 6, 0.1 M Na-Cyanex 272, and 15 min). (d) Effect of the extraction percentage on extraction time (reaction conditions: equilibrium pH value of 6, 0.1 M Na-Cyanex 272, and organic-aqueous ratio of 1.5). (e) Effect of the stripping percentage on sulfuric acid concentration (reaction conditions: organic-aqueous ratio of 1 and 10 min). (f) Effect of the stripping percentage on organic-aqueous ratio (reaction conditions: 0.1 M H2SO4 and 10 min).
3.2.2. Effect of Na-Cyanex 272 Concentration
The extraction of Co, Mn, Ni, and Li from the leach liquor of spent NMC batteries was studied with conditions of Na-Cyanex 272 concentration from 0.01 to 0.2 M at pH 6 and an organic-aqueous ratio of 1:1 over 15 min. Figure 4b shows that by increasing the Na-Cyanex 272 concentration from 0.01 M to 0.1 M, the extraction percentages of cobalt and manganese increased strictly. The reason was that a higher concentration of the extractant enabled more Co2+ and Mn2+ ions to be caught. However, when the Na-Cyanex 272 concentration was higher than 0.1 M, the extraction percentage of nickel started to increase. This was because the excess extractant resulted in an extraction effect that was too strong and that thus had an adverse effect on the separation process.
3.2.3. Effect of Organic-aqueous Ratio
Figure 4c shows that an organic-aqueous ratio from 0.5 to 2.0 was studied using 0.1 M Na-Cyanex 272 at pH 6 over 15 min. The result shows that the extraction percentages of cobalt and manganese increased as the organic-aqueous ratio increased, which means that the cobalt and manganese were not yet extracted completely. However, when the ratio was greater than 1.5, the extraction percentage of nickel increased rapidly. Hence, in order to avoid the extraction problem of nickel, an organic-aqueous ratio of 1.5 was better for the cobalt and nickel separation.
3.2.4. Effect of Extraction Time
In Figure 4d, it is clear that the extraction time was a significant influence in the extraction process. The effect of the extraction time was studied using 0.1 M Na-Cyanex 272 when the pH was 6 and the organic-aqueous ratio was 1.5. The extraction percentage increased substantially from 0.5 to 15 min, and the reaction was balanced after 15 min. In this case, the extraction percentages of lithium and nickel were found to have almost no increases with the increasing extraction time. Finally, the extraction percentages of Co, Mn, Ni, and Li were about 99.2%, 99.3%, 3.3%, and 3.0%, respectively.
3.2.5. Stripping of Co and Mn from the Organic Phase by Sulfuric Acid
After extraction, the cobalt and manganese in the organic phase continued to the stripping process. In this process, we chose H2SO4 as the stripping agent, and the effect of the H2SO4 concentration is presented in Figure 4e. As the figure shows, when the H2SO4 concentration increased, the stripping percentage increased simultaneously. It was clear that the reason was the shortage of H2SO4. Hence, we found that when the H2SO4 concentration was increased up to 0.1 M, the stripping percentages of cobalt and manganese achieved almost 100%. Figure 4f shows that the stripping percentage declined as the organic-aqueous ratio increased, which means that the cobalt and manganese were not yet stripped completely when the organic phase increased. We found that when the organic–aqueous ratio was 2:1, the stripping percentage started to decrease. Hence, in the stripping process, an organic-aqueous ratio of 2 was optimal, and the stripping percentage of cobalt and manganese was almost 100%.
3.3. Solvent Extraction with Na-D2EHPA
After the separation by using Na-Cyanex 272 as the extractant, the metals were separated into two sides. One side contained cobalt with manganese, and the other side contained nickel with lithium. In the previous step, most of the nickel in the material had been separated. Compared with previous literature [46,47], the biggest problem in the recovery process was the poor separation effect resulting from the co-extraction of cobalt and nickel. Hence, the problem of the high proportion of cobalt and nickel had been already solved. In this step, in order to separate the cobalt and manganese effectively, Na–D2EHPA was used as the extractant.
3.3.1. Effect of Equilibrium pH Value
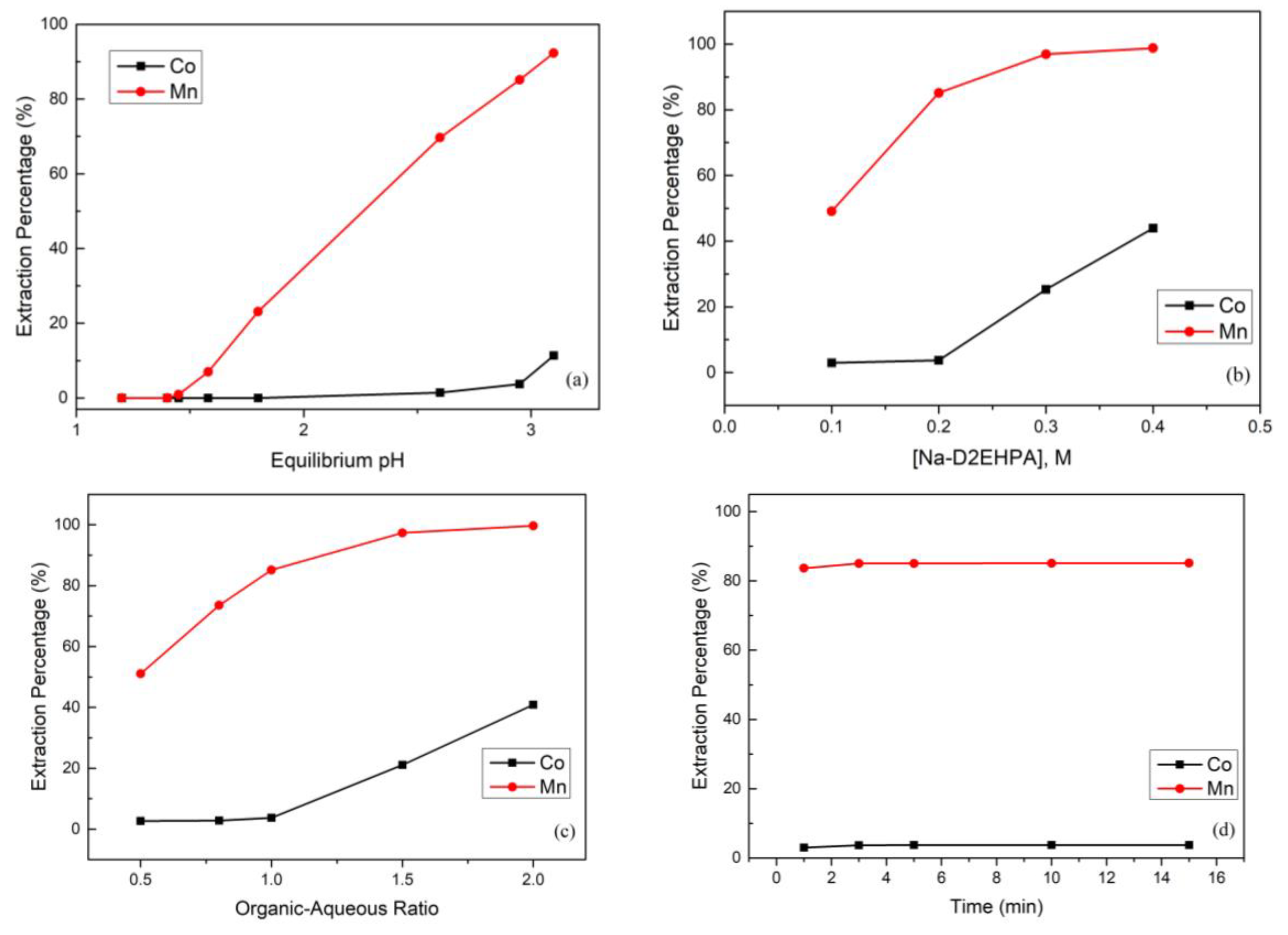
The effect of the equilibrium pH value in the extraction and separation of cobalt and manganese from the sulfate solution is shown in Figure 5a. The extraction percentage of manganese increased as the equilibrium pH value increased. However, when the equilibrium pH value was greater than 2.95, the extraction percentage of cobalt started to increase rapidly. In order to obtain a good recovery of cobalt, we chose a pH value of 2.95 as the optimal.
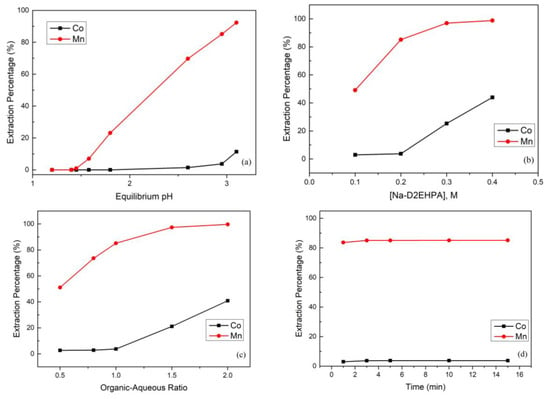
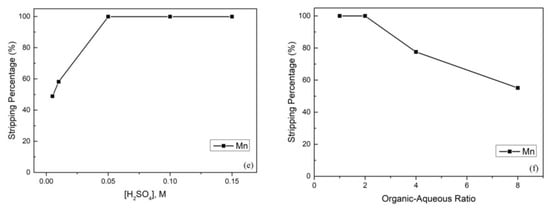
Figure 5.
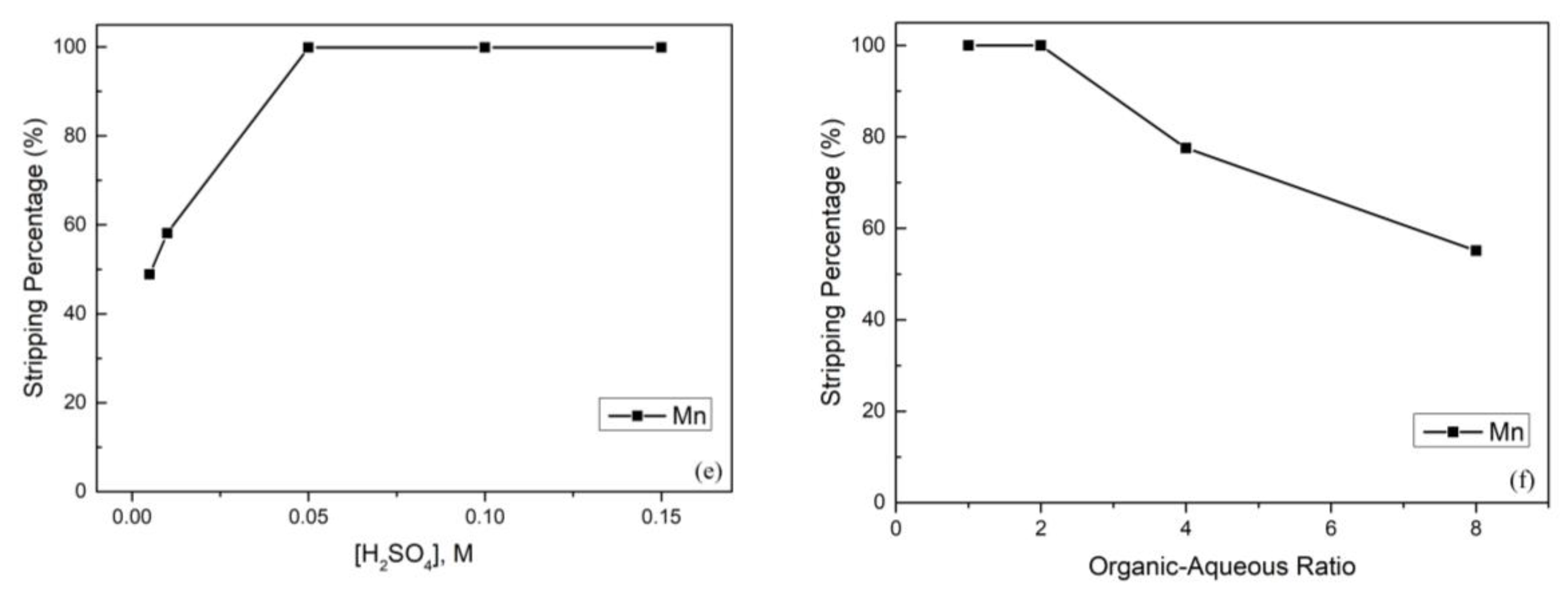
(a) Effect of the extraction percentage on equilibrium pH value (reaction conditions: 0.2 M Na-D2EHPA, organic-aqueous ratio of 1, and 15 min). (b) Effect of the extraction percentage on Na-D2EHPA concertation (reaction conditions: equilibrium pH value of 2.95, organic-aqueous ratio of 1, and 15 min). (c) Effect of the extraction percentage on organic-aqueous ratio (reaction conditions: equilibrium pH value of 2.95, 0.2 M Na-D2EHPA, and 15 min). (d) Effect of the extraction percentage on reaction time (reaction conditions: equilibrium pH value of 2.95, 0.2 M Na-D2EHPA, and organic-aqueous ratio of 1.0). (e) Effect of the stripping percentage on sulfuric acid concentration (reaction conditions: organic-aqueous ratio of 1 and 5 min). (f) Effect of the stripping percentage on organic-aqueous ratio (reaction conditions: 0.05 M H2SO4 and 5 min).
3.3.2. Effect of Na-D2EHPA Concentration
The competitive extraction of cobalt and manganese was studied with Na-D2EHPA concentrations from 0.1 M to 0.4 M. Figure 5b shows that the extraction percentage of manganese increased simultaneously with the Na-D2EHPA concentration. However, when the Na-D2EHPA concentration was higher than 0.2 M, owing to the extraction capacity becoming too strong, the extraction percentage of cobalt also started to increase. Therefore, the extraction percentages of manganese and cobalt were 85.1% and 3.7% respectively under 0.2 M Na-D2EHPA at the equilibrium pH value of 2.95 and organic-aqueous ratio of 1.0 over 15 min.
3.3.3. Effect of Organic-Aqueous Ratio and Extraction Time
In the experiment, the organic-aqueous ratio also influenced the extraction percentage. Figure 5c shows that when the organic-aqueous ratio increased from 0.5 to 1.0, the extraction percentage of manganese increased to 85.1% and the extraction percentage of cobalt only slightly increased. However, when the organic-aqueous ratio was increased up to 1.5, the extraction percentage of cobalt increased to 21.1%. The reason was that the organic phase was too great or the aqueous phase was too little to selectively extract quantities of metal ions. Therefore, an organic-aqueous ratio of 1.0 was chosen as optimal.
In order to avoid consuming too much energy, we controlled the reaction time and investigated the interaction between the organic and aqueous phases. The effect of the extraction time was investigated with the optimized parameters. Figure 5d shows that the extraction time needed to approach the reaction balance was very short. Hence, an extraction time of 5 min was chosen as optimal.
According to the above studies, under optimal parameters, the extraction efficiency of manganese was up to 85.14%. On the basis of this distribution ratio, the efficiency could also increase to over 99.6% by three stages of extraction, retaining cobalt in the aqueous phase. The optimal parameters of both extractions are illustrated in Table 2.

Table 2.
The optimal parameters of solvent extraction.
3.3.4. Stripping of Mn from the Organic Phase by Sulfuric Acid
After extraction, the manganese in the organic phase continued to the stripping process. In this process, we chose H2SO4 as the stripping agent, and the effect of the H2SO4 concentration is presented in Figure 5e. To obtain the best stripping percentage, the H2SO4 concentration was analyzed from 0.005 M to 0.15 M, and the stripping percentage of manganese achieved almost 100% when the H2SO4 concentration increased up to 0.05 M. Furthermore, the effect of the organic-aqueous ratio was also important. The organic-aqueous ratio was analyzed from 1 to 8, and the stripping percentage of manganese started to decline when the organic-aqueous ratio was greater than 2. The reason was that the acidity was insufficient to strip metal ions from the organic phase. Hence, we chose 0.05 M H2SO4 and an organic-aqueous ratio of 2 as optimal parameters.
3.4. Selective Precipitation with DMG
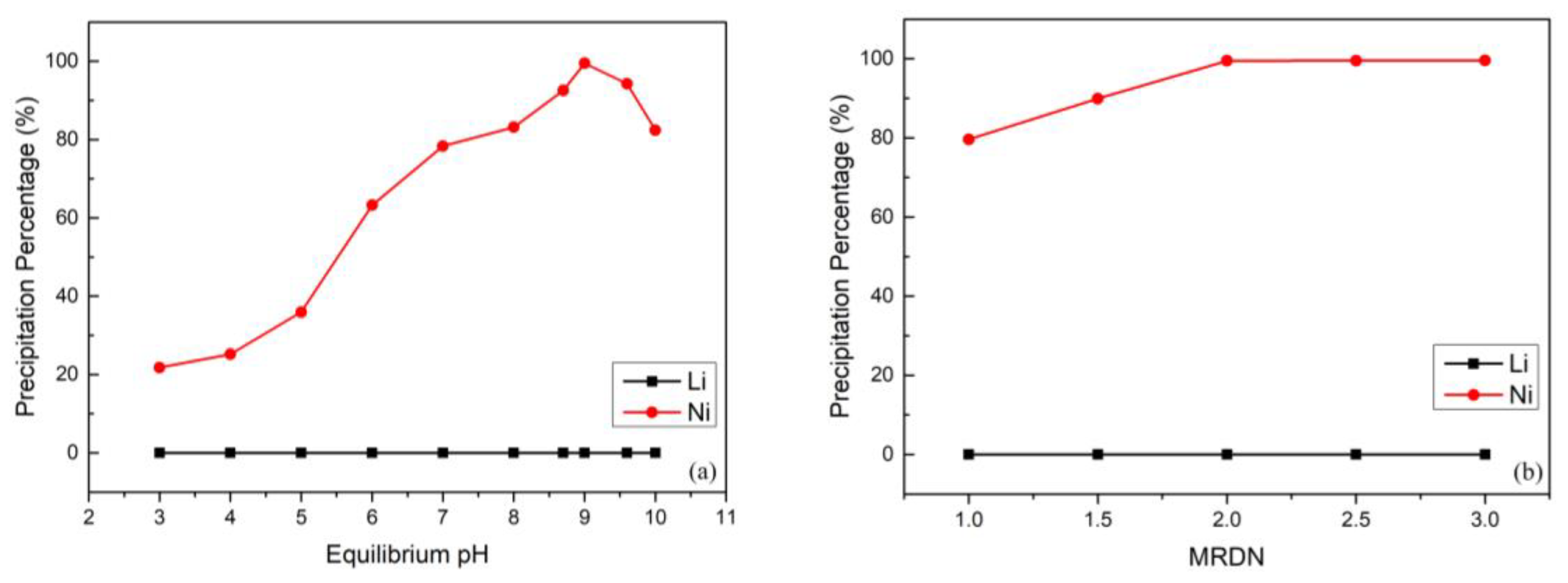
After the separation by using Na-Cyanex 272 as the extractant, the cobalt and manganese were extracted from the leach liquor; on the other hand, nickel and lithium were retained in the aqueous phase. The experiment was designed to employ DMG reagent (C4H8N2O2) to separate the nickel and lithium efficiently. Compared with other methods [48,49], using DMG produces excellent selectivity of Ni2+. DMG is often used as an analytical chemistry reagent and reacts with Ni2+ to form a nickel DMG chelating precipitate. According to previous literature [39], DMG slightly precipitates cobalt at higher pH value; however, we extracted nearly all of the cobalt in the previous solvent extraction process to prevent this problem. Additionally, lithium cannot react with DMG, and thus the separation could be carried out completely. The equilibrium pH value and the MRDN were investigated under conditions of 25 °C, 300 rpm, and 30 min. Figure 6a shows the effect of the equilibrium pH value on the selective precipitation of nickel and lithium. It is clear from this data that DMG was completely unreactive toward lithium, and thus the highest precipitation percentage of nickel was the optimal parameter. When the equilibrium pH was increased, the precipitation percentage of nickel also increased gradually and reached almost 99.5% when the equilibrium pH value was 9; the precipitation percentage of lithium was almost 0% relatively. However, the precipitation percentage of nickel slightly decreased at a higher equilibrium pH value. The reason was attributed to an inadequate reaction between the nickel and DMG chelating precipitate [39]. Figure 6b shows the effect of MRDN in the selective precipitation process. The results indicate that when the MRDN was lower than 2, Ni2+ was not completely reacting with DMG and only formed a small amount of red complex. Hence, the optimal MRDN was 2. This also represented the theoretical ratio of 0.5 for the nickel DMG chelating precipitate in the process. Furthermore, the red complex could be dissolved easily using 4 M HCl solution, and almost 100% of the nickel was dissolved back to the solution. The acid dissolution reaction was the reverse reaction, and DMG is essentially insoluble in strong acid; thus the DMG could be recovered by filtration and reused as a reagent in the selective precipitation process.
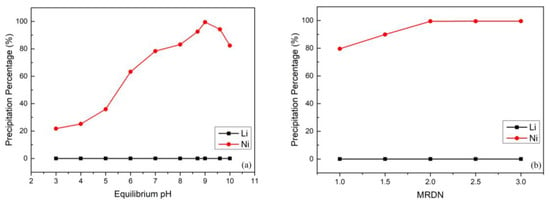
Figure 6.
(a) Effect of dimethylglyoxime (DMG) precipitation percentage on equilibrium pH value (reaction conditions: molar ratio of C4H8N2O2 to nickel (MRDN) of 2.5, 300 rpm, and 30 min). (b) Effect of DMG precipitation percentage on MRDN (reaction conditions: equilibrium pH value of 9, 300 rpm, and 30 min).
3.5. Chemical Precipitation
After the solvent extraction, stripping process, and selective precipitation, four elements had already been separated. Then, chemical precipitation was conducted to obtain the final product under the best operational conditions found in the study. In order to obtain the highest precipitation percentage in the experiment, pH values from 7 to 13 was investigated. In this case, the pH was adjusted to 11 by using the saturated solution of NaOH, and cobalt was precipitated as a red precipitate, cobalt hydroxide. The solution of manganese was precipitated by adding the saturated solution of NaOH at pH 13, and the manganese ions eventually totally transferred to manganese hydroxide. The nickel ion solution could be recovered as nickel hydroxide by using the saturated solution of NaOH when the pH value was increased up to 12. On the other hand, the solution of lithium could be recovered as Li2CO3 by adding a saturated solution of Na2CO3; moreover, hot water could wash out the remaining sodium ions. Finally, the purity analysis was conducted by using ICP-OES; the purity of cobalt, nickel, and lithium products was over 99.5%, and the manganese product also achieved over 93.3% purity.
4. Conclusions
The recovery and separation processes of metals from NMC cathode waste materials has been proven in this work to be successful and effective. The suggested recovery process is shown in Figure 7. The NMC cathode waste materials were treated by leaching, solvent extraction, stripping, selective precipitation, and chemical precipitation processes to recover cobalt, manganese, nickel, and lithium. The optimal parameters obtained in leaching were 2.0 mol/L of H2SO4, 30 mL/g, 70 °C, and 90 min. In this study, we used several agents and combined the advantages of each extractant and precipitating agent to improve the recovery process. The results showed that 0.1 M Na-Cyanex 272 should first be used as the extractant to separate cobalt and nickel under the optimal condition of pH 6, with an organic-aqueous ratio of 1.5 and over 15 min. Then, cobalt and manganese should be separated by using 0.2 M Na-D2EHPA at equilibrium pH 2.95, with an organic-aqueous ratio of 1.0 and over 5 min. On the other hand, nickel and lithium can be separated by using DMG at pH 9, with the molar ratio of DMG to Ni2+ (MRDN) of 2. Finally, the four elements can be precipitated separately by using a saturated solution of NaOH and Na2CO3. By this process, the purity of the cobalt, nickel, and lithium products produced was over 99.5%, and the manganese product also achieved over 90%.
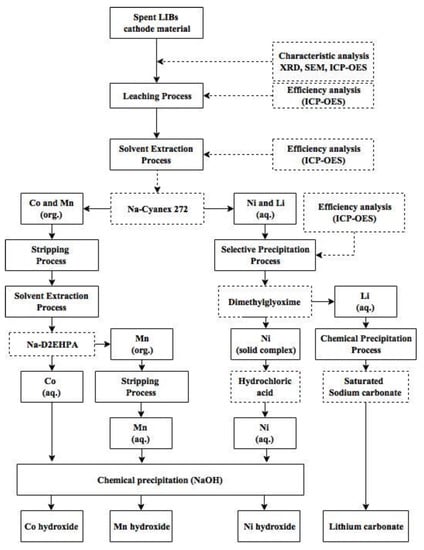
Figure 7.
Suggested recovery process to separate the four metals.
Author Contributions
W.-S.C. and H.-J.H. conceived and designed the experiments; H.-J.H. performed the experiments, analyzed the data, and wrote the paper.
Funding
This research was funded by NCKU Research and Development Foundation (106S281).
Acknowledgments
We wish to acknowledge the support of the Laboratory of Resources Circulation (LRC) at National Cheng Kung University.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, X.; Gaustad, G.; Babbit, C.W.; Richa, K. Economies of scale for future lithium-ion battery recycling infrastructure. Resour. Conserv. Recycl. 2014, 83, 53–62. [Google Scholar] [CrossRef]
- Yamaji, Y.; Dodbiba, G.; Matsuo, S.; Okaya, K.; Shibayama, A.; Fujita, T. A Novel Flow Sheet for Processing of Used Lithium-ion Batteries for Recycling. Resour. Process 2011, 58, 9–11. [Google Scholar] [CrossRef]
- Jha, M.K.; Kumari, A.; Jha, A.K.; Kumar, V.; Hait, J.; Pandey, B.D. Recovery of lithium and cobalt from waste lithium ion batteries of mobile phone. Waste Manag. 2013, 33, 1890–1897. [Google Scholar] [CrossRef] [PubMed]
- Scrosati, B.; Garche, J. Lithium batteries: Status, prospects and future. J. Power Sources 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Zeng, X.L.; Li, J.H.; Singh, N. Recycling of spent lithium-ion battery: A critical review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1129–1165. [Google Scholar] [CrossRef]
- Bernardes, A.M.; Espinosa, D.C.R.; Tenório, J.A.S. Recycling of batteries: A review of current processes and technologies. J. Power Sources 2004, 130, 288–293. [Google Scholar] [CrossRef]
- Shin, S.M.; Kim, N.H.; Sohn, J.S.; Yang, D.H.; Kim, Y.H. Development of a metal recovery process from Li-ion battery wastes. Hydrometallurgy 2005, 79, 172–181. [Google Scholar] [CrossRef]
- Zhao, J.M.; Shen, X.Y.; Deng, F.L.; Wang, F.C.; Wu, Y.; Liu, H.Z. Synergistic extraction and separation of valuable metals from waste cathodic material of lithium ion batteries using Cyanex272 and PC-88A. Sep. Purif. Technol. 2011, 78, 345–351. [Google Scholar] [CrossRef]
- Nayaka, G.P.; Pai, K.V.; Santhosh, G.; Manjanna, J. Recovery of cobalt as cobalt oxalate from spent lithium ion batteries by using glycine as leaching agent. J. Environ. Chem. Eng. 2016, 4, 2378–2383. [Google Scholar] [CrossRef]
- Fouad, O.A.; Farghaly, F.I.; Bahgat, M. A novel approach for synthesis of nanocrystalline γ-LiAlO2 from spent lithium-ion batteries. J. Anal. Appl. Pyrolysis 2007, 78, 65–69. [Google Scholar] [CrossRef]
- Kang, J.G.; Senanayake, G.; Sohn, J.S.; Shin, S.M. Recovery of cobalt sulfate from spent lithium ion batteries by reductive leaching and solvent extraction with Cyanex 272. Hydrometallurgy 2010, 100, 168–171. [Google Scholar] [CrossRef]
- Paulino, J.F.; Busnardo, N.G.; Afonso, J.C. Recovery of valuable elements from spent Li-batteries. J. Hazard. Mater. 2008, 150, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review. Hydrometallurgy 2014, 150, 192–208. [Google Scholar] [CrossRef]
- Xu, J.; Thomas, H.R.; Francis, R.W.; Lum, K.R.; Wang, J.; Liang, B. A review of processes and technologies for the recycling of lithium-ion secondary batteries. J. Power Sources 2008, 177, 512–527. [Google Scholar] [CrossRef]
- Diekmanna, J.; Hanisch, C.; Froböse, L.; Schälicke, G.; Loellhoeffel, T.; Fölster, A.-S.; Kwade, A. Ecological Recycling of Lithium-Ion Batteries from Electric Vehicles with Focus on Mechanical Processes. J. Electrochem. Soc. 2017, 164, A6184–A6191. [Google Scholar] [CrossRef]
- Pagnanelli, F.; Moscardini, E.; Altimari, P.; Atia, T.A.; Toro, L. Leaching of electrodic powders from lithium ion batteries: Optimization of operating conditions and effect of physical pretreatment for waste fraction retrieval. Waste Manag. 2017, 60, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Barik, S.P.; Prabaharan, G.; Kumar, B. An innovative approach to recover the metal values from spent lithium-ion batteries. Waste Manag. 2016, 51, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xue, Q.; Li, L.; Fan, E.; Wu, F.; Chen, R. Sustainable Recycling and Regeneration of Cathode Scraps from Industrial Production of Lithium-Ion Batteries. ACS Sustain. Chem. Eng. 2016, 4, 7041–7049. [Google Scholar] [CrossRef]
- Nayl, A.A.; Elkhashab, R.A.; Badawy, S.M.; El-Khateeb, M.A. Acid leaching of mixed spent Li-ion batteries. Arab. J. Chem. 2014, 10, 3632S–S3639. [Google Scholar] [CrossRef]
- Takacova, Z.; Havlik, T.; Kukurugya, F.; Orac, D. Cobalt and lithium recovery from active mass of spent Li-ion batteries: Theoretical and experimental approach. Hydrometallurgy 2016, 163, 9–17. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R.; Deveci, H. Acid baking of spent lithium ion batteries for selective recovery of major metals: A two-step process. J. Ind. Eng. Chem. 2016, 43, 117–126. [Google Scholar] [CrossRef]
- Zhang, X.; Xie, Y.; Cao, H.; Nawaz, F.; Zhang, Y. A novel process for recycling and resynthesizing LiNi1/3Co1/3Mn1/3O2 from the cathode scraps intended for lithium-ion batteries. Waste Manag. 2014, 34, 1715–1724. [Google Scholar] [CrossRef] [PubMed]
- Pant, D.; Dolker, T. Green and facile method for the recovery of spent Lithium Nickel Manganese Cobalt Oxide (NMC) based Lithium ion batteries. Waste Manag. 2017, 60, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Nayl, A.A.; Hamed, M.M.; Rizk, S.E. Selective extraction and separation of metal values from leach liquor of mixed spent Li-ion batteries. J. Taiwan Inst. Chem. Eng. 2015, 55, 119–125. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Lee, J.C.; Jeong, J.; Kim, B.S.; Pandey, B.D. Selective recovery of cobalt, nickel and lithium from sulfate leachate of cathode scrap of Li-ion batteries using liquid-liquid extraction. Met. Mater. Int. 2014, 20, 357–365. [Google Scholar] [CrossRef]
- Chiu, K.L.; Chen, W.S. Recovery and Separation of Valuable Metals from Cathode Materials of Spent Lithium-Ion Batteries (LIBs) by Ion Exchange. Sci. Adv. Mater. 2017, 9, 2155–2160. [Google Scholar] [CrossRef]
- Song, D.; Wang, X.; Zhou, E.; Hou, P.Y.; Guo, F.X.; Zhang, L.Q. Recovery and heat treatment of the Li(Ni1/3Co1/3Mn1/3)O2 cathode scrap material for lithium ion battery. J. Power Sources 2013, 232, 348–352. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Hydrometallurgical processing of spent lithium ion batteries (LIBs) in the presence of a reducing agent with emphasis on kinetics of leaching. Chem. Eng. J. 2015, 281, 418–427. [Google Scholar] [CrossRef]
- Zhang, X.; Cao, H.B.; Xie, Y.B.; Ning, P.G.; An, H.J.; You, H.X.; Nawaz, F. A closed-loop process for recycling LiNi1/3Co1/3Mn1/3O2 from the cathode scraps of lithium-ion batteries: Process optimization and kinetics analysis. Sep. Purif. Technol. 2015, 150, 186–195. [Google Scholar] [CrossRef]
- He, L.P.; Sun, S.Y.; Song, X.F.; Yu, J.G. Leaching process for recovering valuable metals from the LiNi1/3Co1/3Mn1/3O2 cathode of lithium-ion batteries. Waste Manag. 2017, 64, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gaustad, G.; Babbitt, C.W.; Bailey, C.; Ganter, M.J.; Landi, B.J. Economic and environmental characterization of an evolving Li-ion battery waste stream. J. Environ. Manag. 2014, 135, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, D.A.; Prados, L.M.Z.; Majuste, D.; Mansur, M.B. Hydrometallurgical separation of aluminium, cobalt, copper and lithium from spent Li-ion batteries. J. Power Sources 2009, 187, 238–246. [Google Scholar] [CrossRef]
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Recovery of valuable metals from cathodic active material of spent lithium ion batteries: Leaching and kinetic aspects. Waste Manag. 2015, 45, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Sun, R.; Xu, J.; Chen, Z.; Kang, M. Recovery of cobalt from spent lithium ion batteries using sulphuric acid leaching followed by solid-liquid separation and solvent extraction. RSC Adv. 2016, 88, 85303–85313. [Google Scholar] [CrossRef]
- Hung, S.H.; Lin, C.F.; Chiang, P.C.; Tsai, T.H.; Peng, C.Y. Recovery of metal ions from spent Lithium Ion Batteries (LIBs) using sodium salts of D2EHPA or P507: Performance evaluation and life cycle assessment. Res. J. Chem. Environ. 2014, 18, 39–47. [Google Scholar] [CrossRef]
- Swain, B.; Mishra, C.; Jeong, J.; Lee, J.C.; Hong, H.S.; Pandey, B.D. Separation of Co(II) and Li(I) with Cyanex 272 using hollow fiber supported liquid membrane: A comparison with flat sheet supported liquid membrane and dispersive solvent extraction process. Chem. Eng. J. 2015, 271, 61–70. [Google Scholar] [CrossRef]
- Mantuano, D.P.; Dorella, G.; Elias, R.C.A.; Mansur, M.B. Analysis of a hydrometallurgical route to recover base metals from spent rechargeable batteries by liquid-liquid extraction with Cyanex 272. J. Power Sources 2006, 159, 1510–1518. [Google Scholar] [CrossRef]
- Chen, X.P.; Xu, B.; Zhou, T.; Liu, D.; Hu, H.; Fan, S.Y. Separation and recovery of metal values from leaching liquor of mixed-type of spent lithium-ion batteries. Sep. Purif. Technol. 2015, 144, 197–205. [Google Scholar] [CrossRef]
- Wang, R.C.; Lin, Y.C.; Wu, S.H. A novel recovery process of metal values from the cathode active materials of the lithium-ion secondary batteries. Hydrometallurgy 2009, 99, 194–201. [Google Scholar] [CrossRef]
- Chen, X.P.; Zhou, T.; Kong, J.R.; Fang, H.X.; Chen, Y.B. Separation and recovery of metal values from leach liquor of waste lithium nickel cobalt manganese oxide based cathodes. Sep. Purif. Technol. 2015, 141, 76–83. [Google Scholar] [CrossRef]
- Joo, S.H.; Shin, D.J.; Oh, C.H.; Wang, J.P.; Park, J.T.; Shin, S.M. Application of Co and Mn for a Co-Mn-Br or Co-Mn-C2H3O2 Petroleum Liquid Catalyst from the Cathode Material of Spent Lithium Ion Batteries by Hydrometallurgical Route. Metals 2017, 7, 439. [Google Scholar] [CrossRef]
- Devi, N.B.; Nathsarma, K.C.; Chakravortty, V. Separation and recovery of cobalt (II) and nickel (II) from sulphate solutions using sodium salts of D2EHPA, PC 88A and Cyanex 272. Hydrometallurgy 1998, 49, 47–61. [Google Scholar] [CrossRef]
- Mohapatra, D.; Kim, H.I.; Nam, C.W.; Park, K.H. Liquid-liquid extraction of aluminium(III) from mixed sulphate solutions using sodium salts of Cyanex 272 and D2EHPA. Sep. Purif. Technol. 2007, 56, 311–318. [Google Scholar] [CrossRef]
- Sarangi, K.; Reddy, B.R.; Das, R.P. Extraction studies of cobalt(II) and nickel(II) from chloride solutions using Na-Cyanex 272. Separation of Co(II)/Ni(II) by the sodium salts of D2EHPA, PC88A and Cyanex 272 and their mixtures. Hydrometallurgy 1999, 52, 253–265. [Google Scholar] [CrossRef]
- Park, K.H.; Kim, H.I.; Parhi, P.K.; Mishra, D.; Nam, C.W.; Park, J.T.; Kim, D.J. Extraction of metals from Mo-Ni/Al2O3 spent catalyst using H2SO4 baking-leaching-solvent extraction technique. J. Ind. Eng. Chem. 2012, 18, 2036–2045. [Google Scholar] [CrossRef]
- Mubarok, M.Z.; Hanif, L.I. Cobalt and Nickel Separation in Nitric Acid Solution by Solvent Extraction Using Cyanex 272 and Versatic 10. Procedia Chem. 2016, 19, 743–750. [Google Scholar] [CrossRef]
- Flett, D.S. Cobalt-Nickel Separation in Hydrometallurgy: A Review. Chem. Sustain. Dev. 2004, 12, 81–91. [Google Scholar]
- Lewis, A.; Van Hille, R. An exploration into the sulphide precipitation method and its effect on metal sulphide removal. Hydrometallurgy 2006, 81, 197–204. [Google Scholar] [CrossRef]
- Hammack, R.W.; Edenborn, H.M. The removal of nickel from mine waters using bacterial sulfate reduction. Appl. Microbiol. Biotechnol. 1992, 37, 674–678. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).