Abstract
Analyses of the oxidation characteristics of HR3C austenitic steel exposed to supercritical carbon dioxide were carried out at temperatures ranging from 600 to 650 °C under 25 MPa. It was observed that the weight gain increased with increases in temperature and time. The oxide morphology and phase were characterized using scanning electron microscopy (SEM) combined with energy-dispersive X-ray spectroscopy (EDS), and X-ray diffraction (XRD). Furthermore, the three-dimensional morphology and chemical composition of the surface oxide were inspected using a secondary ion mass spectrometer (SIMS). The majority of the oxide formed on HR3C at 600–650 °C was Cr2O3. Carbon enrichment occurs on the surface of the oxide scale and the oxide–substrate interface due to a carbonization reaction. The corrosion mechanism is also discussed in this paper.
1. Introduction
The power generation efficiency of a supercritical carbon dioxide (S-CO2) Brayton cycle is higher than that of a steam cycle with the same parameters [1]. In practical S-CO2 power cycles and coupled energy systems, corrosion products, metal ions, and fine particulates released from structural materials can influence emulsion stability, phase separation efficiency, and fouling behavior in separators and heat exchangers [2]. In Rankine cycle power generation systems, a reheat heat exchanger is often applied that includes a high-pressure path between a high-pressure inlet and a high-pressure outlet. The high-temperature environment causes severe corrosion to take place. Corrosion of high-temperature surface materials has strongly affected the safe operation of such units, and this issue has attracted significant attention from researchers.
HR3C stainless steel tubes are used as superheaters and reheater boiler tube sections in ultra-supercritical coal-fired power plants due to their relatively high creep strength [3]. A duplex scale with an outer iron-rich oxide and an inner oxide layer containing Fe, Ni, and Cr developed on ferritic–martensitic steels during high-temperature oxidation in S-CO2 [4,5]. Rouillard et al. [5] studied the influence of pressure on the corrosion behavior of 9Cr and 12Cr ferritic–martensitic steels. Increasing CO2 pressure is beneficial in terms of increasing the carburization rate. Cao et al. [6] compared the oxidation resistance of stainless steel and nickel-based alloys in S-CO2 at 650 °C and 20 MPa. The lower corrosion rate of the nickel-based alloys, as compared to the stainless steel, is attributed to the formation of a chromium-rich oxide. Zhu et al. [7] carried out corrosion tests of T22 and P92 in S-CO2 and compared the corrosion rate with data for supercritical water (SCW). The weight gain indicated that the oxidation rates of T22 and P92 in the SCW are much higher than those in the S-CO2. Liu et al. [8] investigated the influence of O2, SO2, or H2S impurities on the corrosion of T91 in S-CO2. The carburization of the alloy in an environment containing impurities is inhibited due to the reduced carbon activity or a competitive adsorption process. Li et al. [9] found that the presence of O2 in S-CO2 weakened the oxidation of SS310 austenite steel and Alloy 740 due to the higher stability of the surface Cr2O3 layer. Quadakkers et al. [10] also reported that the addition of O2 to S-CO2 promoted the formation of protective chromia scale. The corrosion behavior of HR3C was investigated in S-CO2 with 3.6 mol.% O2 and 5.3 mol.% H2O, and it was found that HR3C suffered from severe oxidation and carburization [11]. Moreover, the corrosion and creep behavior of different materials at different S-CO2 temperatures were also investigated [12,13,14,15], and the corrosion mechanisms of the materials were discussed.
Due to the insufficient strength and poor corrosion resistance of carbon steel, it is not suitable as a material for the superheaters and reheaters of supercritical boilers. A more appropriate and efficient alternative material is HR3C austenitic steel, which is widely used as a structural material in ultra-supercritical power plants due to its excellent creep strength and steam oxidation resistance [16]. However, the corrosion behavior of HR3C austenite steel in high-temperature S-CO2 is still not fully understood. This paper aims to study the corrosion properties of HR3C steel exposed to a 600–650 °C S-CO2 environment. The microstructure of HR3C, including its surface and cross-sectional morphologies, was analyzed using scanning electron microscopy (SEM). The elemental compositions of the oxide layer and oxide component were investigated by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and energy-dispersive spectroscopy (EDS). To further determine the distribution of elements along the thickness direction, particularly the distribution of carbon, depth profiling and 3D imaging analysis of the oxide structure were conducted using a secondary ion mass spectrometer (SIMS).
2. Experimental Design
A commercial austenitic stainless steel, HR3C, was used in the tests. The chemical composition of the HR3C steel was provided by the manufacturers and is listed in Table 1. Specimens with the dimensions 20 mm × 10 mm × 2 mm were obtained using an electron discharge machine. The samples were polished by SiC abrasive paper from 200# to 1000#, followed by alcohol cleaning in an ultrasonic cleaner. The samples were then loaded onto a ceramic multilayer triangular bracket. The corrosion test was performed in CO2 at 600–650 °C and 25 MPa. Figure 1 shows a schematic of this SC-CO2 system and the 2 L autoclave. The experimental system is mainly composed of gas cylinder, metering pump, preheater, autoclave, cooling machine and counterbalance valve. The samples were positioned in the autoclave, and the flow rate was maintained at about 5 g/min by the S-CO2 pump. The purity of the carbon dioxide used for the corrosion test was 99.998%. The concentration of oxygen, the main impurity, was 2 ppm by volume. The weight of the samples was recorded before and after the experiment using an electronic balance with a precision of 0.1 mg. In each experiment, three parallel samples were used to obtain weight gain data per oxidation time by averaging the results. In order to further analyze layer structure and composition depth distribution information, depth profiling and 3D imaging of the different elements were performed using an SIMS equipped with cesium sputtering.

Table 1.
The chemical composition of HR3C (wt.%).

Figure 1.
Schematic diagram of the corrosion test platform.
3. Results and Discussion
3.1. Weight Gain
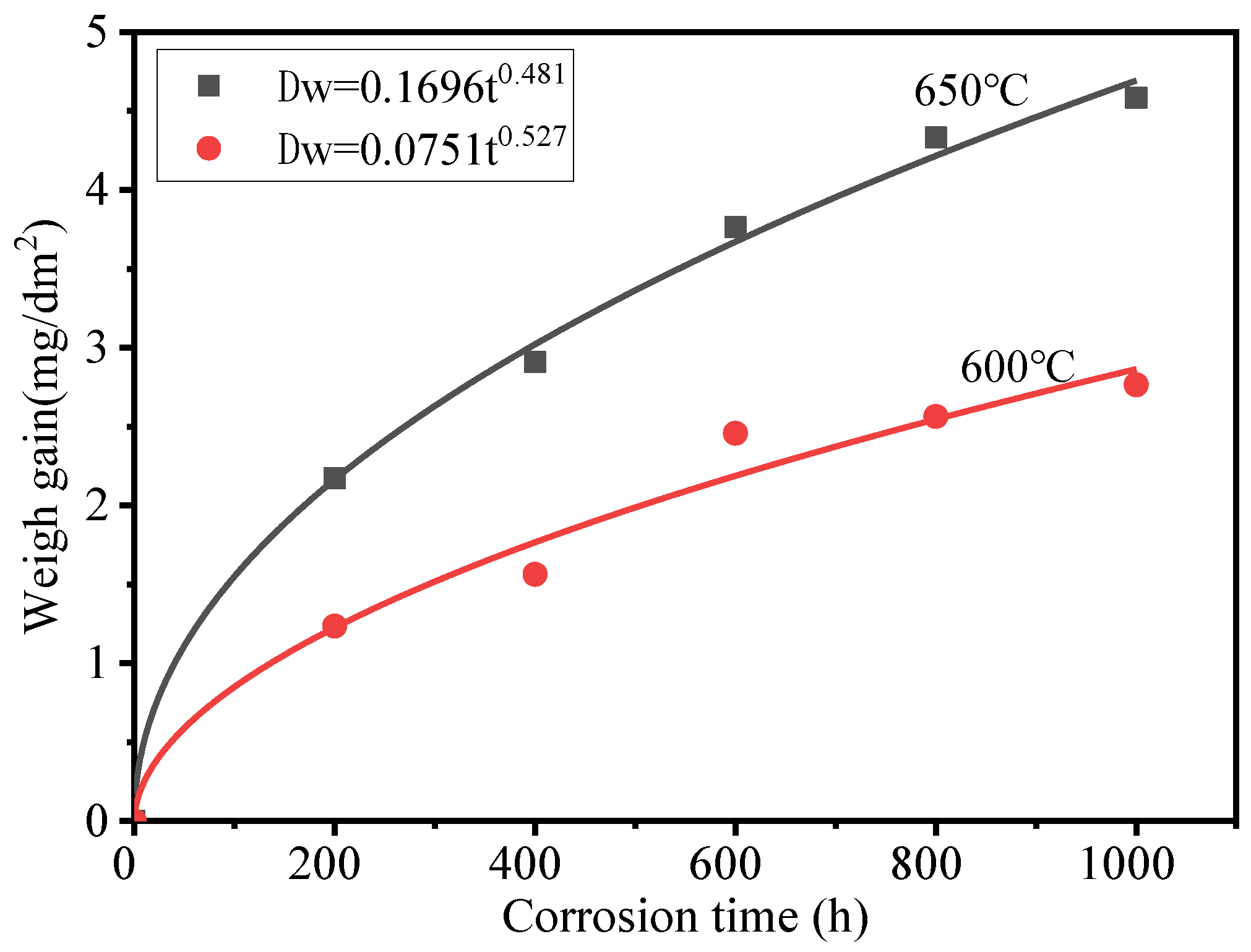
Figure 2 shows the weight gain of HR3C exposed to 600–650 °C S−CO2 at 25 MPa. The weight gain can be fitted using oxidation laws, as shown in the following equation:
where is the weight gain of HR3C (mg dm−2), is an oxidation rate constant (mg dm−2 h−n), n is a time exponent, and t is the oxidation time (h). The time exponents are 0.527 and 0.481 for 600 and 650 °C, respectively, and are represented by a diffusion-controlled oxide growth rate. The largest weight gain was 4.583 mg dm−2, recorded when the oxidation time was 1000 h at 650 °C. HR3C showed good corrosion resistance in S−CO2 due to its high Cr concentration. The weight gain of HR3C in the tests is greater than that of Sanicro 25 in 600 °C S-CO2 [14], but lower than that of 800H in 650 °C S-CO2 [17].
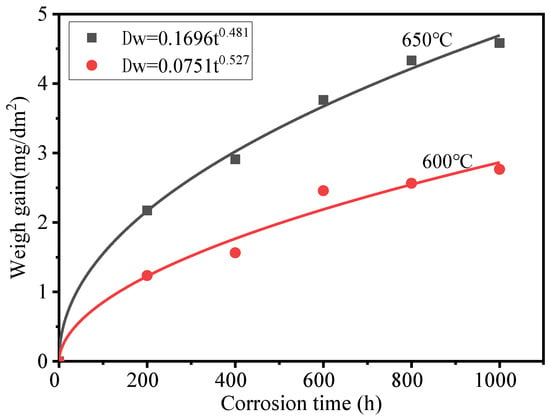
Figure 2.
Weight gain of HR3C in S-CO2 at 600–650 °C and 25 MPa.
3.2. Surface Morphology and Phase Composition
The oxides formed on austenitic steel are thin compared to those formed on the ferritic–martensitic steel. Tan [4] reported the corrosion behavior of F91 and HCM12A at 650 °C and 20.7 MPa, and found that angular oxide grains and some cracks covered the surface of ferritic–martensitic steels. F-M steels also exhibit a similar structure under supercritical water [18,19]. A needle-shaped morphology forms on the surface of 316 austenitic stainless steel exposed to S-CO2 for short periods [6]. The surfaces of Alloy 800H samples exposed to S-CO2 are covered by clustered oxide nodules, which tend to overlap as the exposure time increases [17]. Discontinuous oxides and numerous micrometer-sized nodular pores form on Sanicro 25 [14].
As can be observed in Figure 3, the morphology of the surface oxide is highly dependent on both temperature and time. Figure 3 shows the surface morphology of HR3C steel after different oxidation durations in 600 and 650 °C S-CO2. As can be seen from Figure 3, a nodular oxide composed of particles of different sizes can be observed, while the other formation is a dense oxide film composed of fine particles. With increases in temperature and oxidation time, the outer nodular oxide film gradually grows along the surface, but even if the oxidation time reaches 1000 h, the nodular outer oxide film does not completely cover the whole matrix surface.


Figure 3.
Surface morphological features of HR3C samples after exposure to S-CO2 at 600–650 °C for different durations. (a) 600 °C for 200 h, (b) 600 °C for 600 h, (c) 600 °C for 1000 h, (d) 650 °C for 200 h, (e) 650 °C for 600 h, (f) 650 °C for 1000 h, and (g) higher magnification view of region in (f).
As shown in Figure 3a,d, the alloy surface is covered with uniformly small oxide particles. With prolonged oxidation time, larger oxide particles form on the metal surface (Figure 3b,e). Even larger oxide particles are observed on the alloy surface after 1000 h of oxidation (Figure 3c,f). Corresponding to the blue box shown in Figure 3f, Figure 3g presents a high-magnification morphology image of the nodular oxide formed on HR3C. From this surface morphology image, it can be observed that after 1000 h of oxidation in S-CO2 at 650 °C, the oxide exhibits a polyhedral grain morphology and pores are noted.
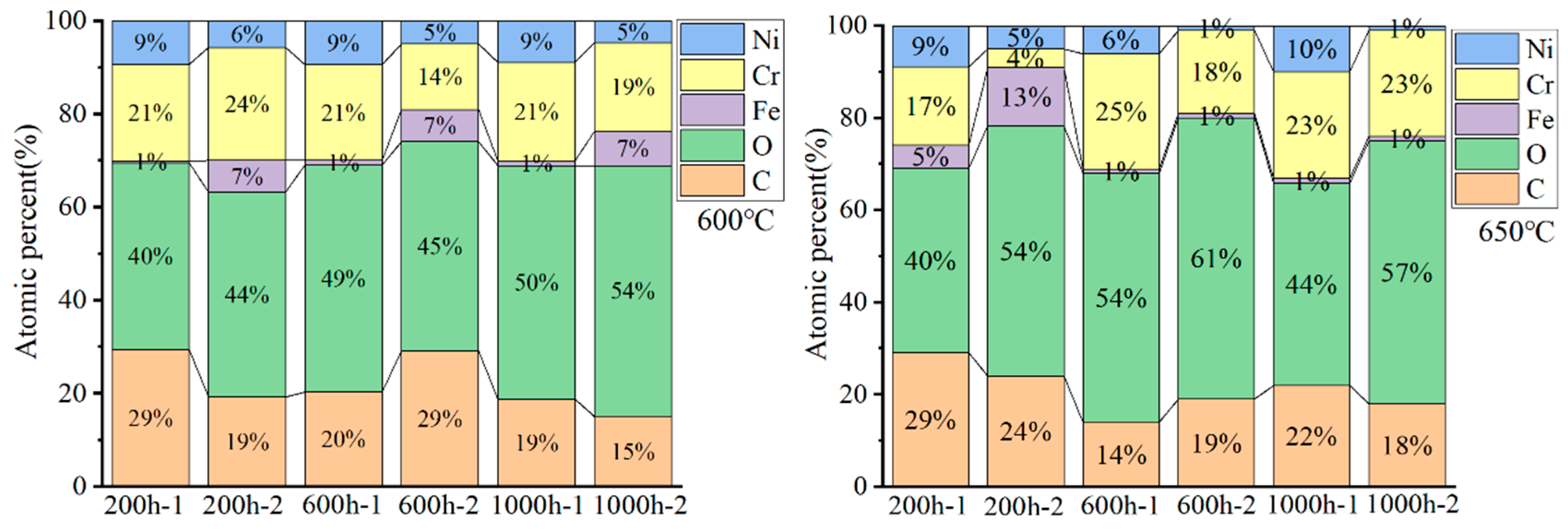
The chemical compositions of the oxides are presented in Figure 4, marked with the numbers 1 and 2. As shown in Figure 3 and Figure 4, the nodular oxides formed on HR3C steel, marked by the number two, are primarily composed of Cr, O, and C, with a low concentration of Ni and Fe. The flat oxide layer marked by the number one also is primarily composed of Cr, O, and C. The large particles in Figure 3 (location 2) exhibit a relatively low Ni content. In comparison, the Ni content is higher in the region denoted as position 1 in Figure 3, compared to that in the nodular oxides.
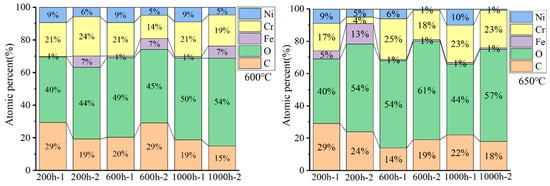
Figure 4.
Energy-dispersive X-ray spectroscopy analysis of the surface composition of the oxide scale formed on HR3C exposed to 600–650 °C S-CO2 for different durations (the numbers 1 and 2 represent the positions marked in Figure 3).
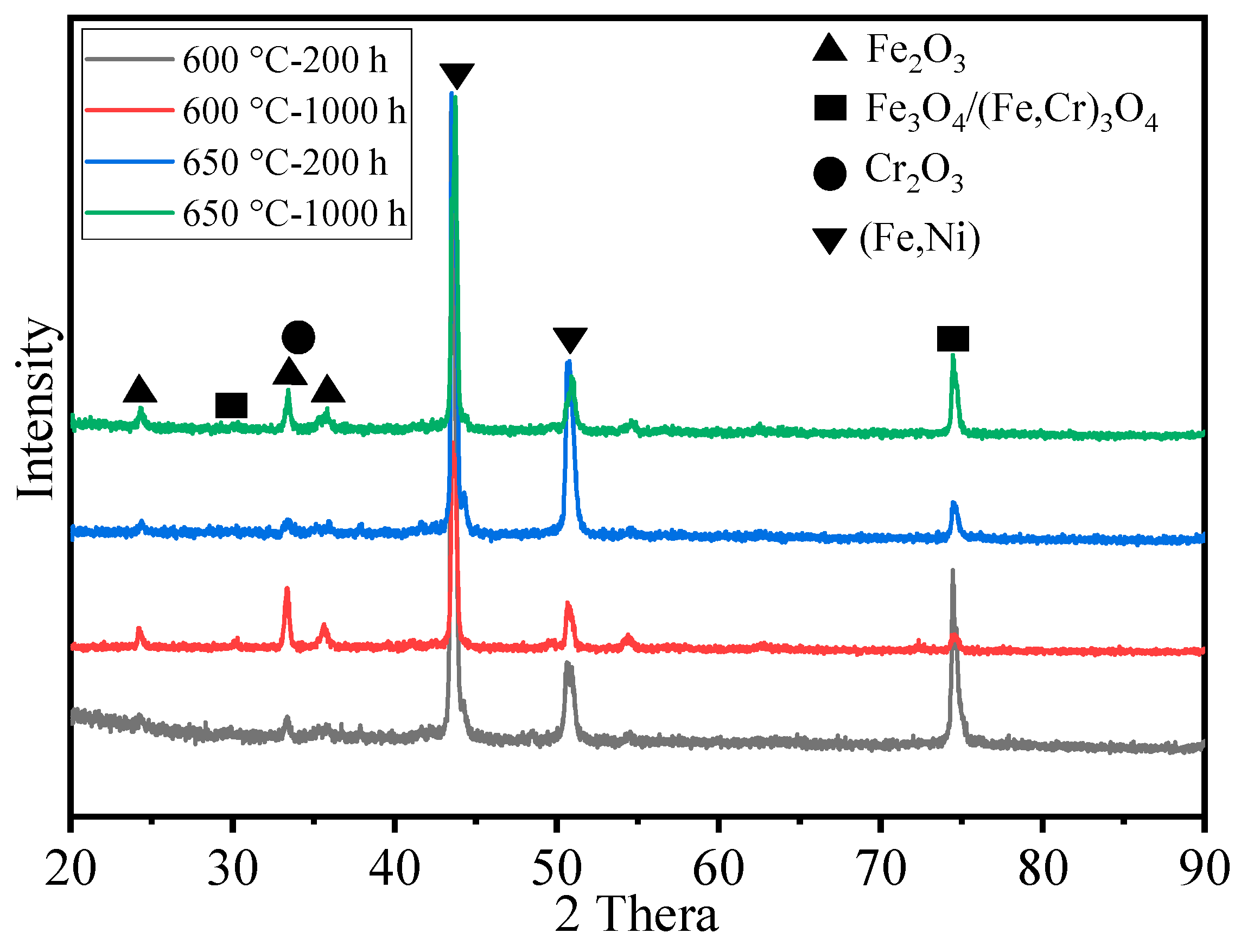
Figure 5 presents the XRD spectra of HR3C steel oxidized in S-CO2 at 600 °C and 650 °C for 200 h and 1000 h. As shown in Figure 5, in the early stages of oxidation, i.e., at 200 h, the oxide phases on the alloy surface at different temperatures are not pronounced. However, at prolonged oxidation times, the characteristic peaks of the oxide phases become more prominent, indicating an increase in the oxide content. At 1000 h of oxidation, HR3C steel oxidized at 600 °C and 650 °C exhibits similar oxide types, primarily composed of Fe2O3, Cr2O3, and the spinel phases Fe3O4/(Fe,Cr)3O4. Additionally, characteristic peaks of Ni in the HR3C alloy matrix are also detected, suggesting the formation of a relatively thin oxide film on the alloy surface.
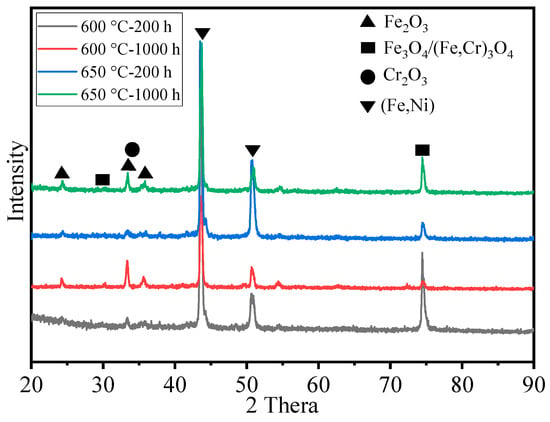
Figure 5.
XRD spectra of HR3C steel after oxidation for 200 h and 1000 h in S-CO2 at 600 °C and 650 °C.
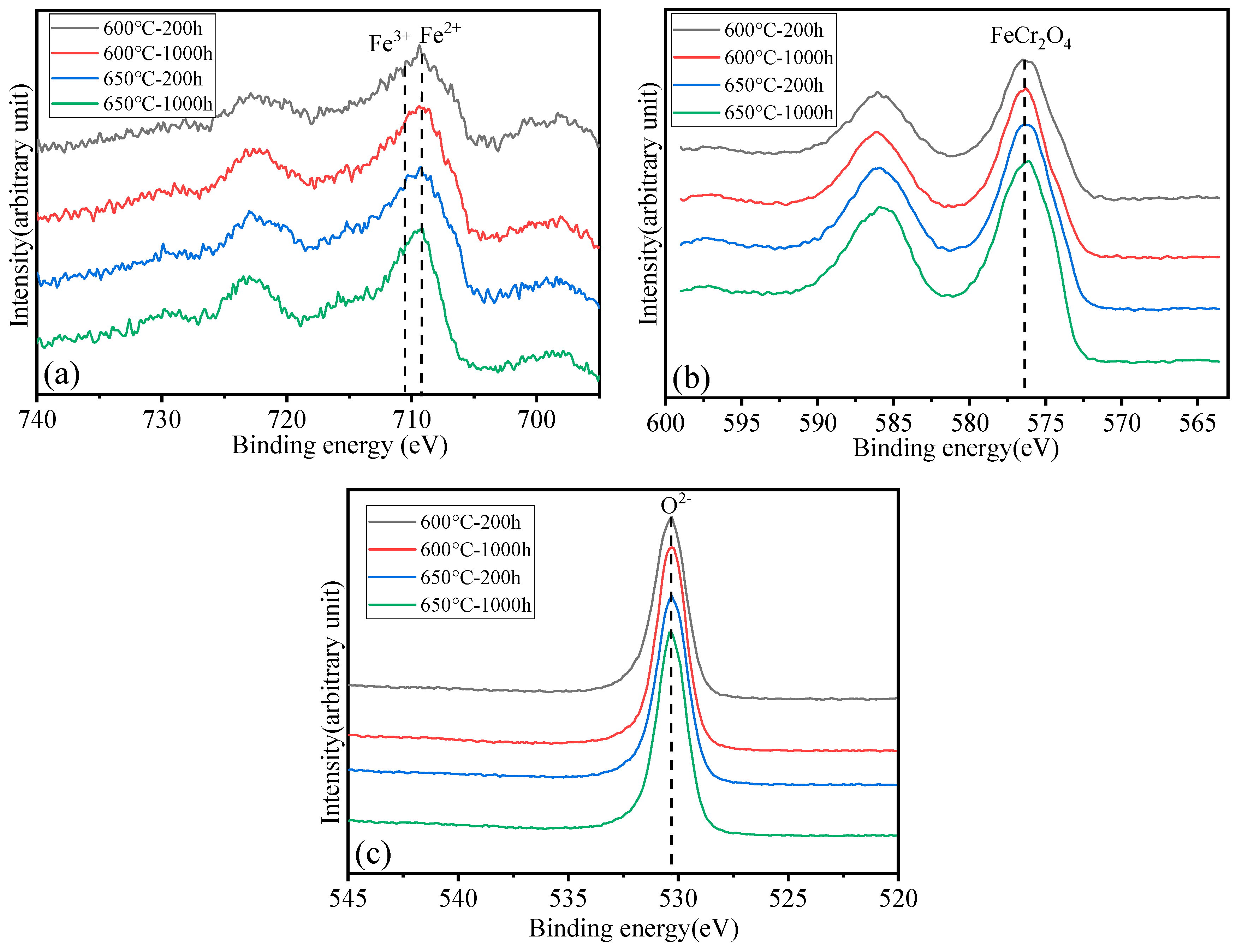
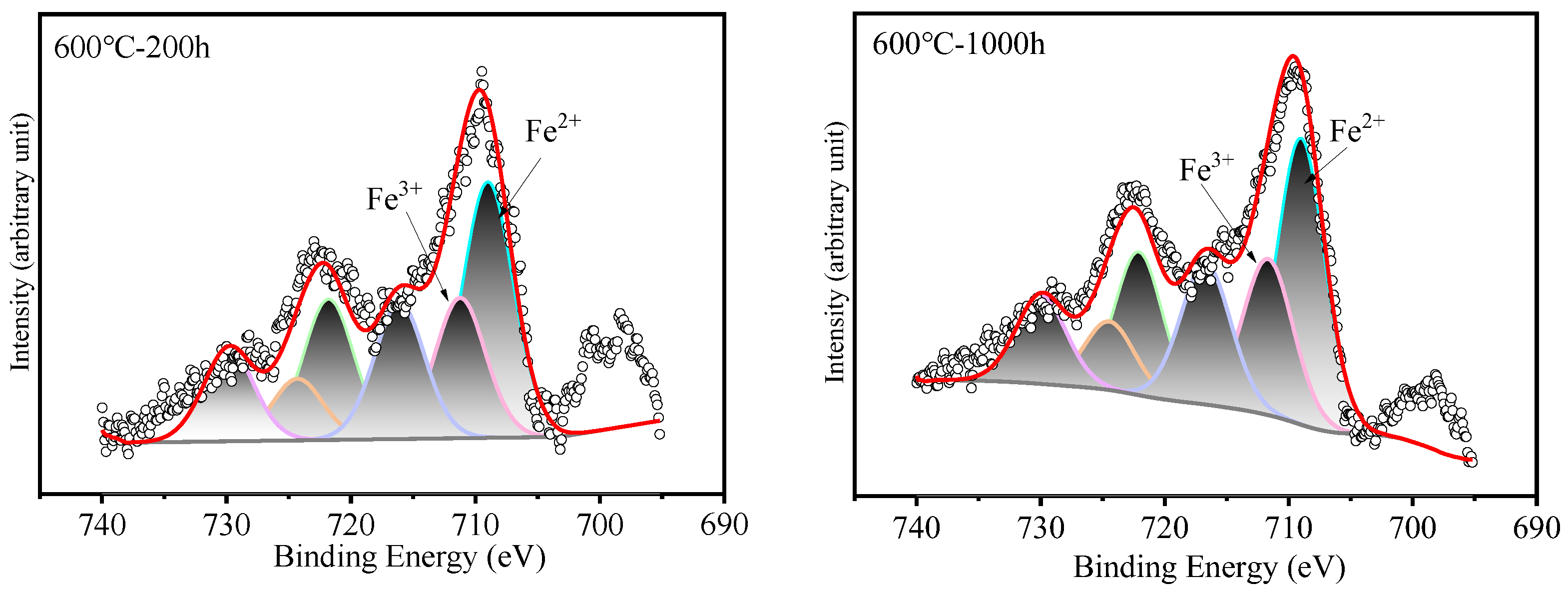
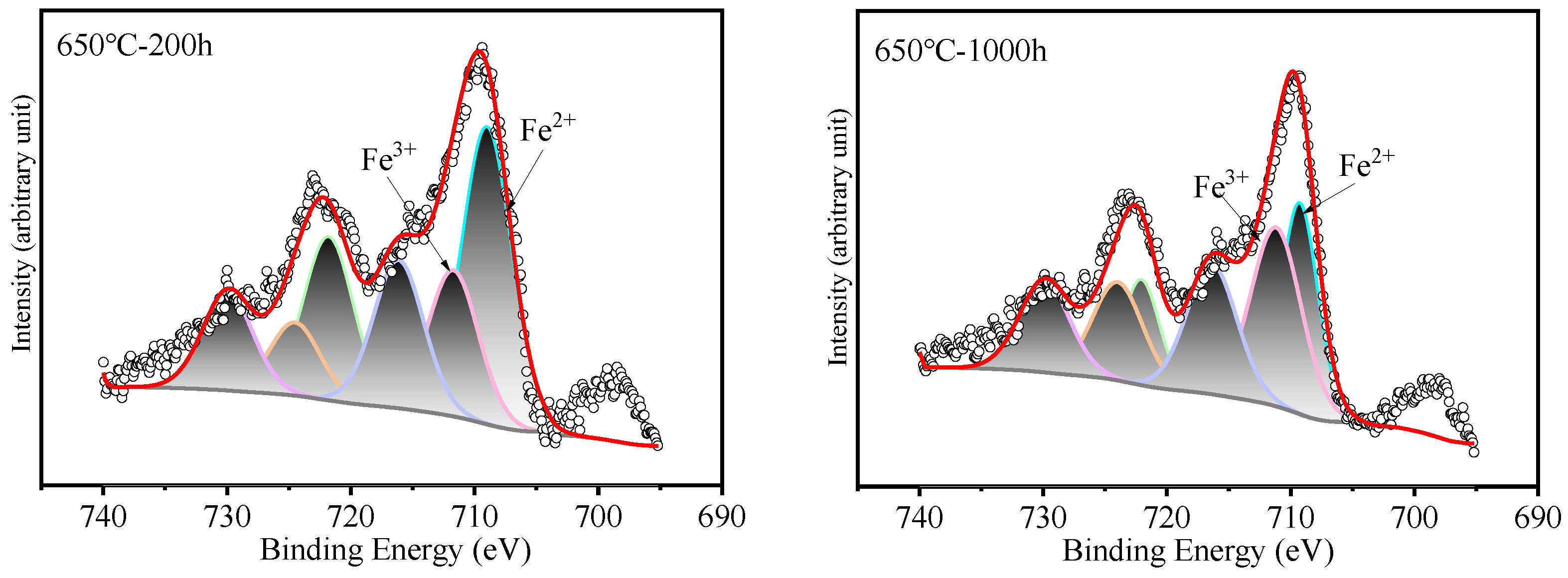
Surface analysis of the HR3C alloy oxidized in S-CO2 at 600 °C and 650 °C for varying durations was conducted using XPS to examine the elemental states of the oxide film components, including Fe, Cr, and O, and the results are presented in Figure 6. Figure 7 shows XPS patterns of Fe on HR3C steel after exposure to S-CO2 at 600–650 for 200 and 1000 h. In Figure 6a and Figure 7, the peak positions of Fe are at (709.2 ± 0.3) eV and (710.2 ± 0.3) eV, corresponding to Fe2+ and Fe3+. The corresponding oxide phase may be Fe3O4 and Fe2O3. Figure 6b displays the narrow XPS scan spectra of Cr after different sputtering times. On the surface of the oxide film, the Cr peak is located at (576.6 ± 0.2) eV, corresponding to FeCr2O4. Figure 6c shows the O spectrum with a binding energy of (530.5 ± 0.2) eV, corresponding to O2−.
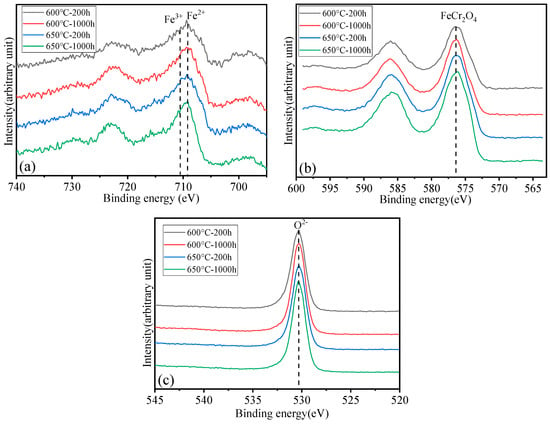
Figure 6.
XPS patterns of the oxide film on HR3C steel after exposure to S-CO2 at 600–650 °C for 200 and 1000 h. (a) Fe 2p core-level spectra, (b) Cr 2p core-level spectra, and (c) O 1s core-level spectra.
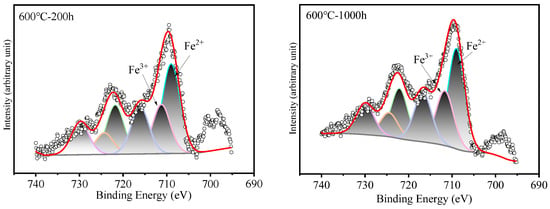
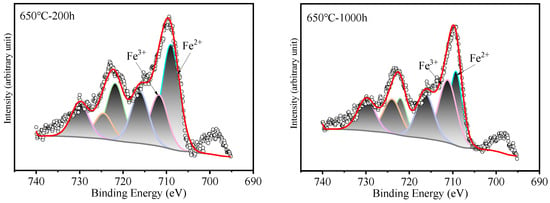
Figure 7.
XPS patterns of Fe on HR3C steel after exposure to S-CO2 at 600–650 °C for 200 and 1000 h.
3.3. Analysis of Element Distribution Characteristics
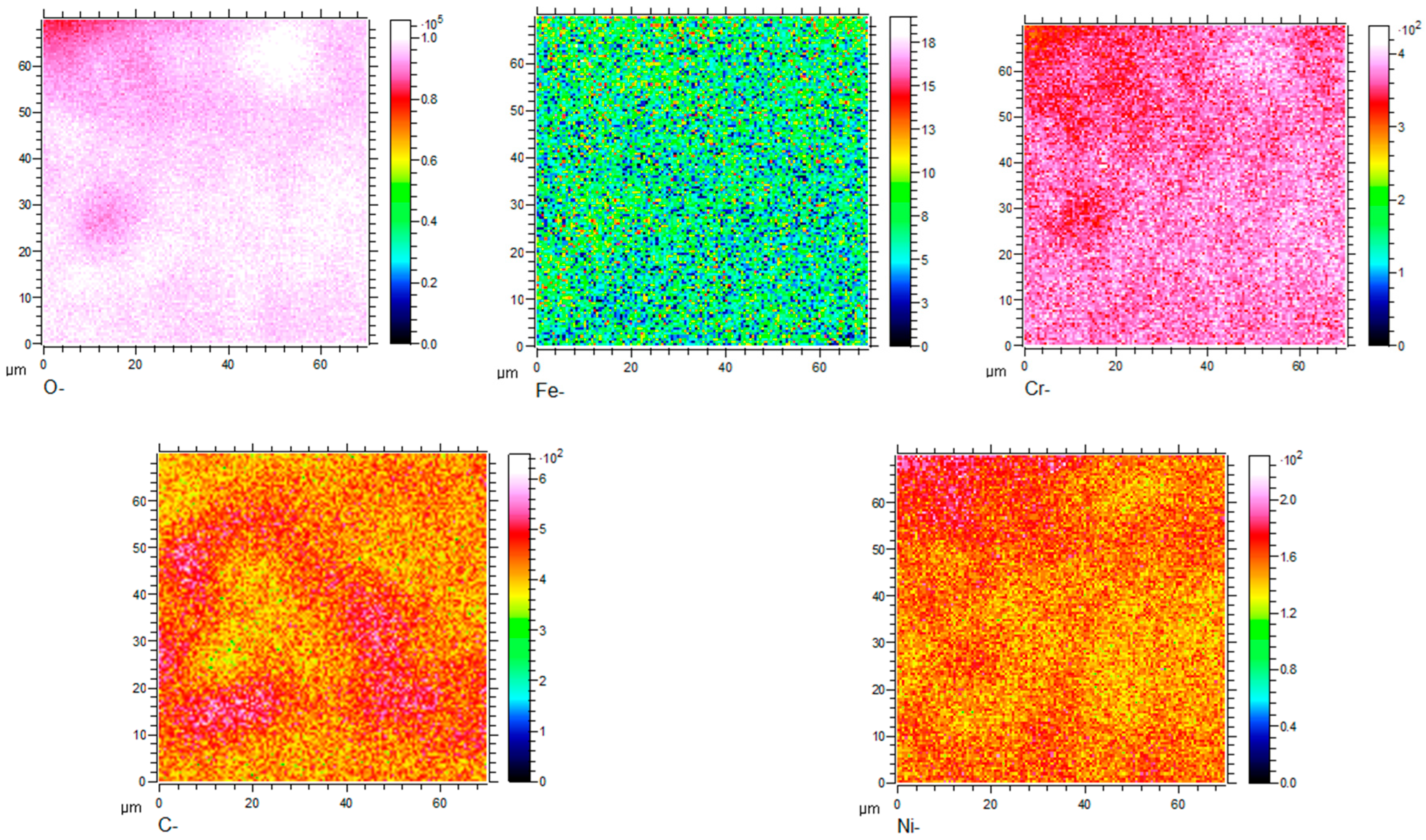
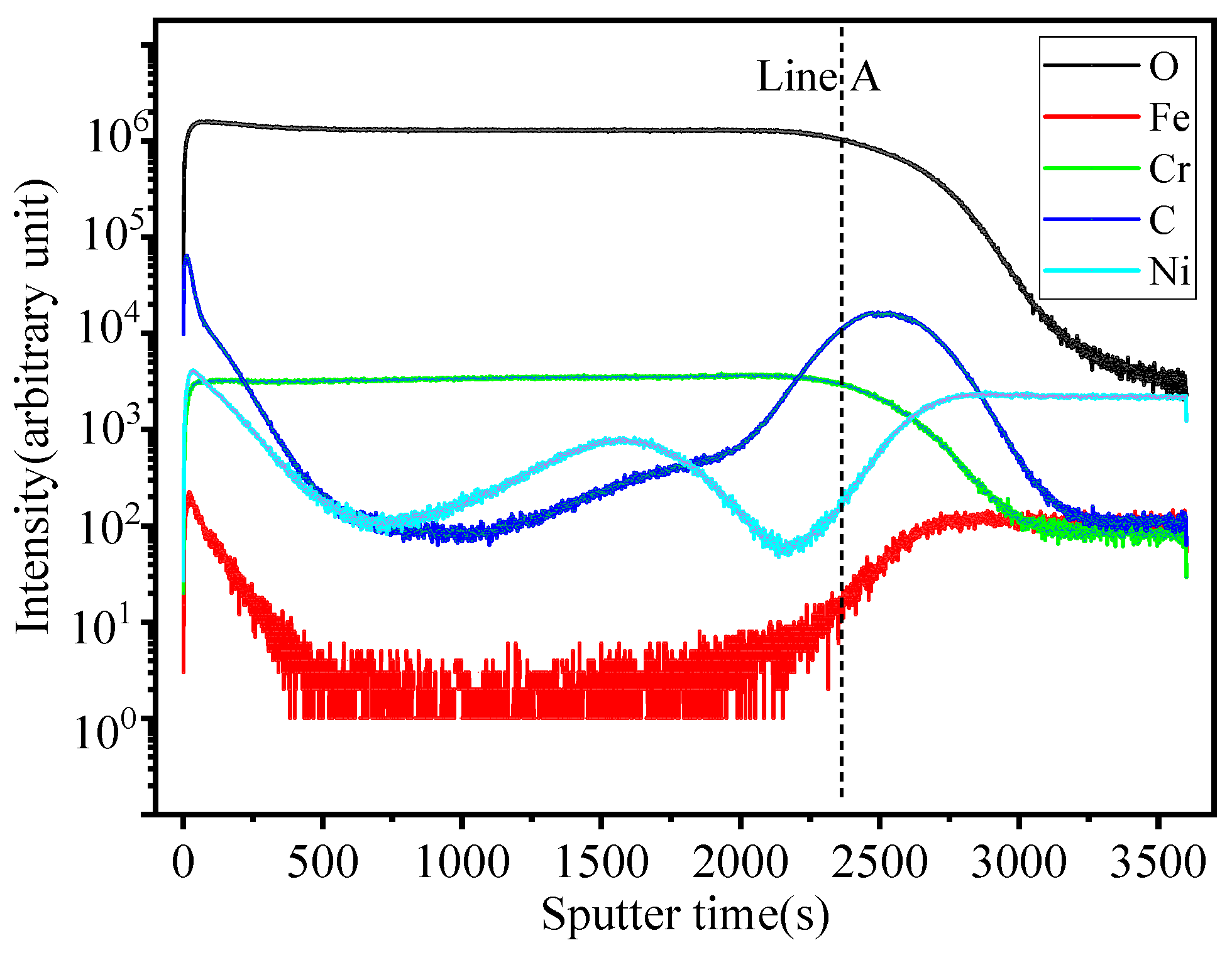
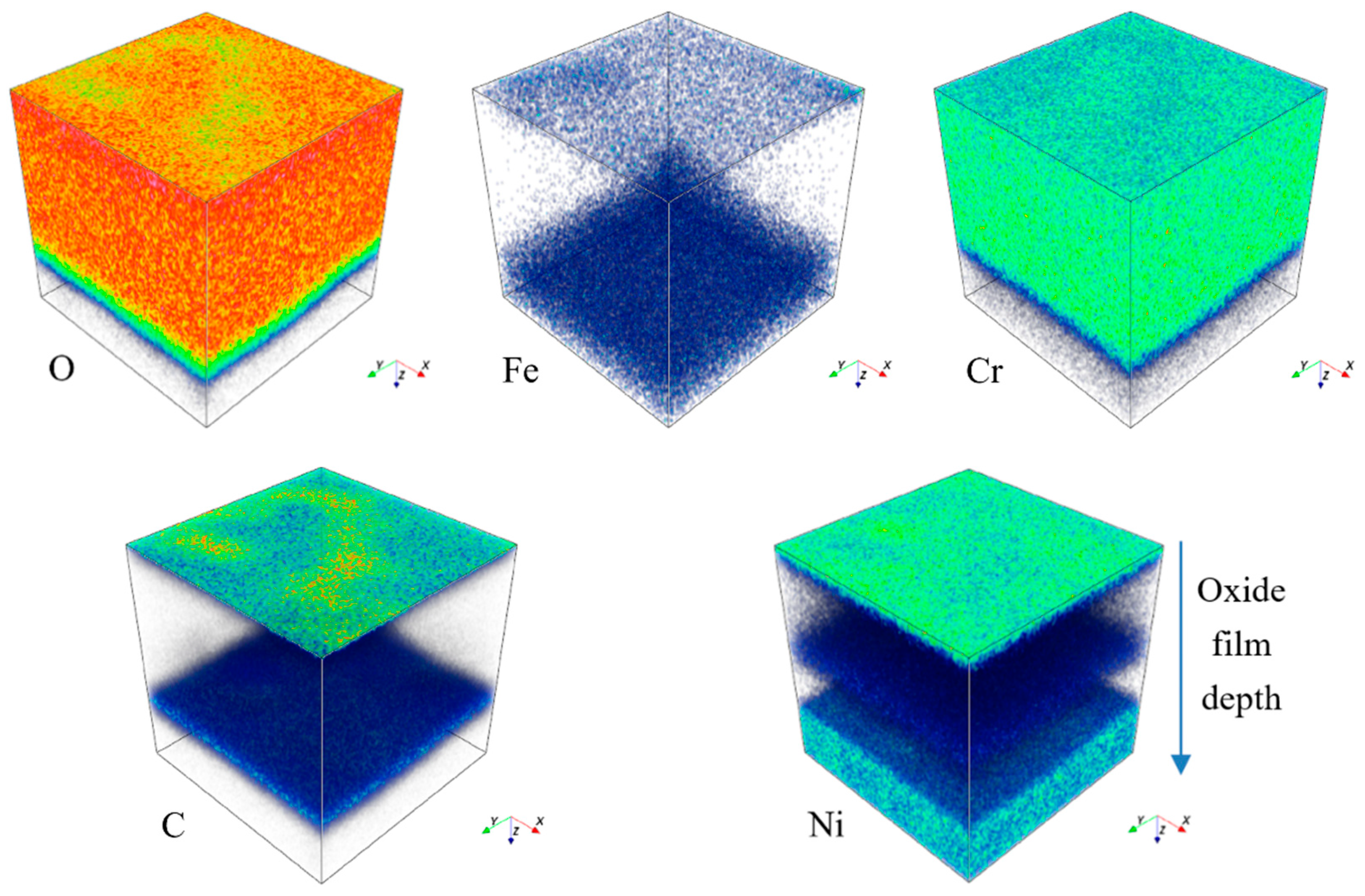
Figure 8, Figure 9, Figure 10 and Figure 11 show the element distribution obtained by selecting a 70 × 70 μm2 area of the sample for scanning using Cs with a 2 keV sputtering ion beam to conduct in-depth analysis. The concentration profiles of the sample surface are indicated in Figure 8, which shows that the oxide surface is rich in Cr and O, while the content of Fe is low. C and Ni are also observed to be present in the oxide surface. Figure 9 shows the relationship between sputtering time and elemental distribution intensity. The sputtering time can be regarded as a proxy for the depth of the specimen, which means that as the sputtering time increases deeper elemental compositions of the material can be detected. It can be seen from Figure 9 that the content of O is high and remains constant for sputtering times of less than 2370 s. The oxygen content continuously decreases from the location of the line marked A to the base alloy, indicating the existence of an inward gradient of diffusion of oxygen. A small quantity of the element Fe was detected at the outermost layer, while it was absent from the oxide scale. The highest carbon content is located at the interface between the oxide and the metallic bulk and at the outermost oxide layer. The distribution of Cr in the two-dimensional diagram is relatively uniform, and the concentration of Cr has an obvious peak in the intermediate region, which indicates that chromium enrichment at a certain depth of the oxide layer is present. In HR3C, a small amount of Cr scale can be observed under the oxide, indicating an outward diffusion of Cr. Combined with the data in Figure 5, it can be concluded that the peak of Cr is related to the formation of Cr2O3, i.e., it is related to stable oxides that can improve the corrosion resistance of a material [20].
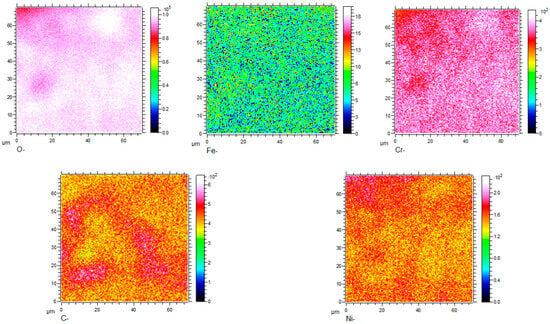
Figure 8.
Two-dimensional element distribution diagram of HR3C steel after oxidation in S-CO2 at 650 °C for 1000 h.
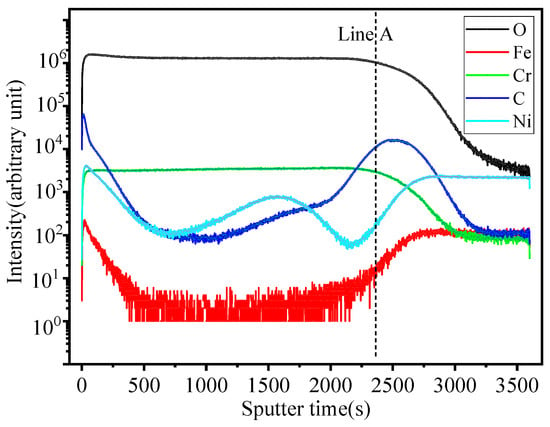
Figure 9.
Elemental depth profile of HR3C steel after oxidation in S-CO2 at 650 °C for 1000 h.
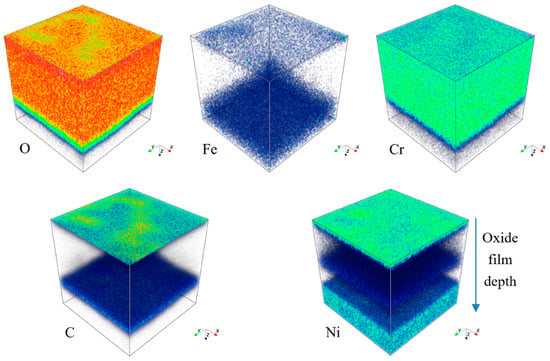
Figure 10.
Three-dimensional element distribution diagram of HR3C steel after oxidation in S-CO2 at 650 °C for 1000 h.

Figure 11.
SEM morphology of the oxide layer formed on an HR3C steel surface exposed to S-CO2 at 650 °C for 1000 h.
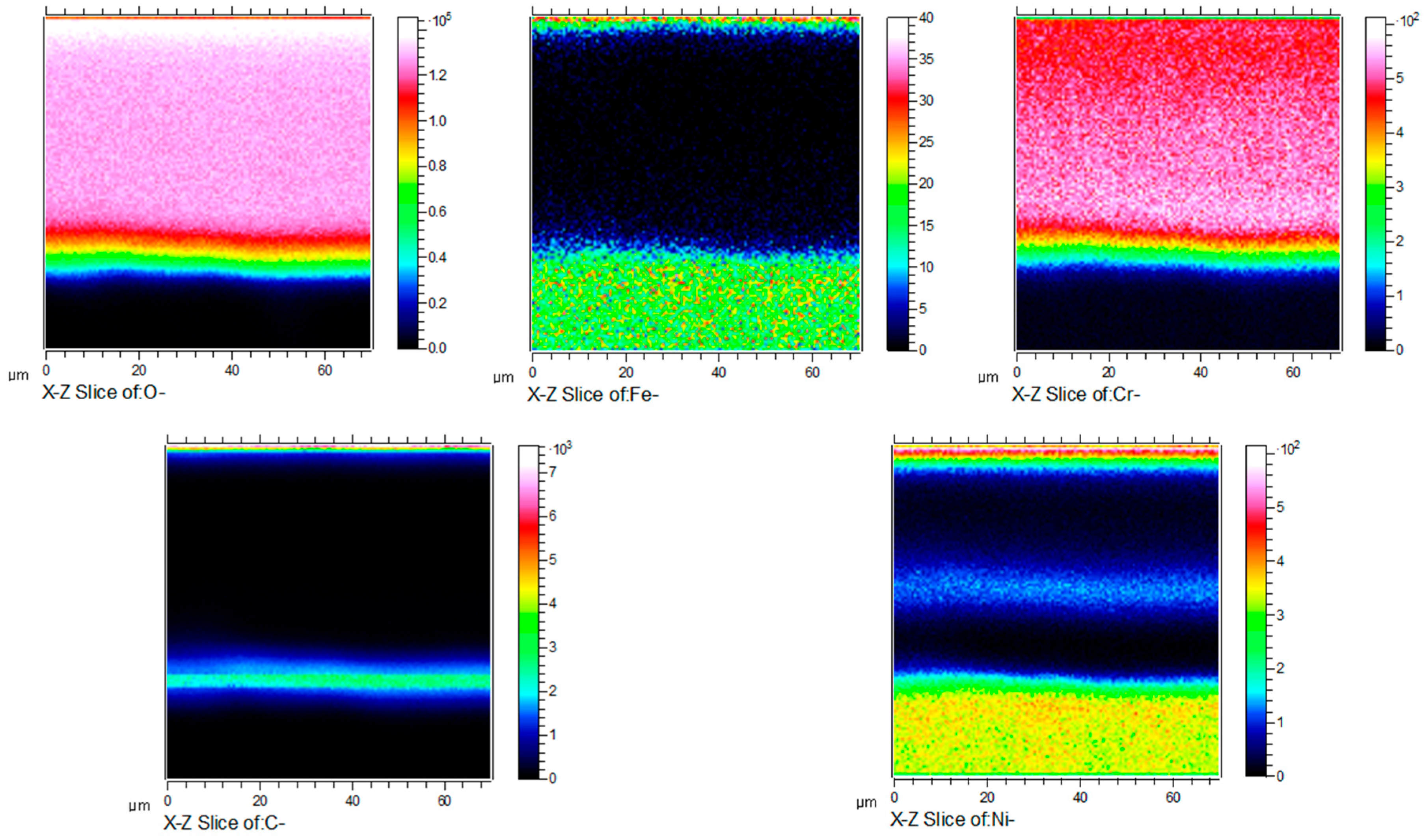
Figure 11 shows the cross-sectional micrograph of HR3C in an S-CO2 environment for 1000 h at 650 °C. According to Figure 11, the thickness of the oxide scale developed on HR3C is about 1.2 μm. Combined with the results of XRD shown in Figure 5, it can be concluded that a relatively thin oxide film forms on the alloy’s surface. Figure 10 and Figure 12 show, respectively, the dimensional elemental distributions and cross-section mapping in the scanning area after exposure to S-CO2 at 650 °C for 1000 h. This shows that the concentrations of O and Cr are high throughout the scale, and a transition layer can be detected that is possibly caused by inward oxygen diffusion and outward Cr diffusion. The results of XRD and elemental distribution mapping indicated that the chromium-rich oxide is mainly composed of Cr2O3, which provides good protection in supercritical CO2 environments. Due to the thermodynamic stability of the lattice structure of Cr2O3, it can effectively prevent further oxidation. In addition, the formation of a Cr2O3 layer may also benefit from the higher proportion of Cr in HR3C steel and its rapid diffusion behavior at high temperatures. A number of studies in the literature indicate that Cr2O3 forms at the oxide–S-CO2 interface by the outward migration of Cr cations [21,22,23]. Cr interstitial and Cr vacancies have, respectively, been found to be the dominant defects in Cr2O3 at low and high oxygen partial pressures. Gulsoy [24] believed that the grain boundary diffusion of Cr cations plays a role in the Cr oxidation kinetics of Alloy 617 in He-CO-CO2 environments. These defects can provide a path for Cr diffusion. The content of Cr2O3 is greater, in relative terms, than that of the other oxide phases, as shown in Figure 9. HR3C’s excellent corrosion resistance in S-CO2 environments can be attributed to a dense and continuous Cr2O3 oxide layer. The sufficient Cr content of the alloy is a key factor in maintaining the stability of the Cr2O3 layer [9].
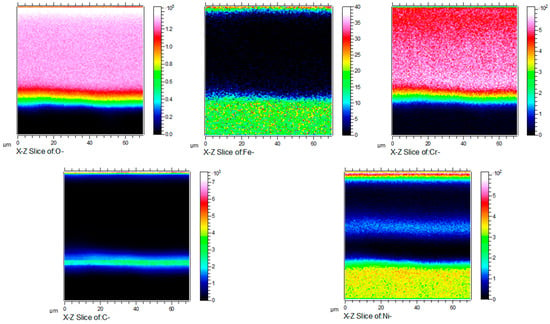
Figure 12.
X-Z plane element concentration profile of HR3C steel after oxidation in S-CO2 at 650 °C for 1000 h.
The formation of an iron-rich oxide during the corrosion of HR3C steel indicates that iron ions can diffuse outward to the oxide–S-CO2 interface, i.e., the following reaction occurs [25,26,27]:
The formation of the protective layer is indicated by the passivation Reaction (5). Considering the formation of mixed spinels, which are more stable and adherent than pure magnetite, the Reaction (6) is given as
Based on analysis of the results of XPS, XRD, and EDS, it can further be confirmed that the iron-rich oxide in the outermost layer is formed mainly of Fe2O3, followed by Fe3O4 and (Fe,Cr)3O4. Theoretical calculation shows that the partial pressure of oxygen in carbon is much higher than that of Fe2O3 [28]. Based on the equilibrium oxygen partial pressure needed for oxide stability, both Fe oxide phases appear in the order of Fe3O4 followed by Fe2O3. However, Fe2O3 was not detected in high-purity carbon dioxide environments, except for when oxygen is added to the carbon dioxide. For Sanicro 25, Gui reports that the oxide scale mainly consists of Cr oxides and carbides [14]. The oxides that form on 800H and AL-6XN austenitic steel exposed to high-purity CO2 at 650 °C are primarily composed of Cr2O3 and (Fe,Cr,Mn)-rich spinel oxide [4]. In this study, Fe2O3 was also detected, in addition to Fe3O4, Cr2O3, and (Fe,Cr)-rich spinel. The presence of Fe2O3 indicates that the high oxygen partial pressure at the oxide–S-CO2 interface is due to the added oxygen. Compared with Cr, Fe cations can easily diffuse through the oxide scale to form a multi-layer oxide. Furthermore, the high solubility of Fe in Cr2O3 permits their outward diffusion, resulting in the formation of more Fe-rich oxides at the scale surface.
The data in Figure 9, Figure 10 and Figure 11, obtained by SIMS, clearly show that the enrichment of C occurred on the surface of the corrosion product and at the oxide film–substrate interface. It has been proposed that the diffusion of gaseous species such as CO and CO2 takes place through the grain boundary, through cracks or pores in the scale, or through nanochannels [29,30,31,32]. The formation of carbon may occur through Reaction (7) between carbon-containing gas and metal cations at the oxide–metal interface. Carbon reacts with chromium in the austenitic matrix to form carbides, resulting in the formation of a carburized region.
In addition, in regions where carbide and oxide coexist, carbides are very likely oxidized to form oxide [33]. The outward cation migration may be the rate-limiting step due to the existence of a diffusion layer (as shown in Figure 9). If oxygen diffusion were the rate-limiting step, it is unlikely that a diffusion layer would form because the oxygen would not diffuse before the oxide.
4. Conclusions
This study investigated in detail the oxidation behavior of HR3C steel in S-CO2 at 600 °C and 650 °C. Through a series of analyses, including depth profiling, two-dimensional distribution mapping, and three-dimensional visualization, it provides insight into the complex interplay between the material components and the corrosive environment. The outer layer predominantly consists of Fe2O3, Fe3O4, and (Fe,Cr)3O4. The impurity oxygen promotes the formation of Fe2O3. The inner layer is mainly composed of Cr2O3, which plays a key role in improving the corrosion resistance of HR3C steel. The carbon distribution indicates the outward diffusion of cations and inward diffusion of carbon-containing gas, and that the reactions occur at the intersection of the S-CO2–corrosion product interface and the oxide–metal substrate interface.
Author Contributions
Methodology, K.L. and S.M.; formal analysis, K.L.; investigation, K.Y.; resources, S.Z.; data curation, S.Z.; writing—original draft, Z.Z.; writing—review and editing, Z.Z.; supervision, K.Y.; project administration, S.M.; funding acquisition, K.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This work is financially supported by the Science and Technology Program of CSEI (2023youth16).
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Halimi, B.; Suh, K.Y. Computational analysis of supercritical CO2 Brayton cycle power conversion system for fusion reactor. Energ. Convers. Manag. 2012, 63, 38–43. [Google Scholar] [CrossRef]
- Khormali, A. Increasing the Efficiency of Demulsification Treatment in Petroleum Industry Using a Multicomponent Demulsifier Package. Pet. Chem. 2023, 63, 539–552. [Google Scholar] [CrossRef]
- Scheffknecht, G.; Chen, Q. Material Issues for Supercritical Boilers. In Parsons 2000: Advanced Materials for 21st Century Turbines and Power plant; IOM Communications Ltd.: Cambridge, UK, 2000. [Google Scholar]
- Tan, L.; Anderson, M.; Taylor, D.; Allen, T.R. Corrosion of austenitic and ferritic-martensitic steels exposed to supercritical carbon dioxide. Corros Sci. 2011, 53, 3273–3280. [Google Scholar]
- Rouillard, F.; Furukawa, T. Corrosion of 9-12Cr ferritic–martensitic steels in high-temperature CO2. Corros Sci. 2016, 105, 120–132. [Google Scholar] [CrossRef]
- Cao, G.; Firouzdor, V.; Sridharan, K.; Anderson, M.; Allen, T.R. Corrosion of austenitic alloys in high temperature supercritical carbon dioxide. Corros Sci. 2012, 60, 246–255. [Google Scholar]
- Zhu, Z.; Xu, H.; Khan, H.I.; Jiang, D.; Zhang, N. Effect of Exposure Temperature on Oxidation of Austenitic Steel HR3C in Supercritical Water. Oxid Met. 2019, 91, 77–93. [Google Scholar] [CrossRef]
- Liu, Y.; Xiao, G.; Wang, M.; Guo, Q.; Wang, Z.; Wu, Y.; Xu, H.; Chen, D. Corrosion behavior of heat-resistant steel T91 in high-temperature supercritical carbon dioxide with impurity O2, SO2 or H2S. J. Supercrit. Fluid. 2023, 198, 105936. [Google Scholar]
- Li, K.; Zeng, Y.; Luo, J.L. Corrosion of SS310 and Alloy 740 in high temperature supercritical CO2 with impurities H2O and O2. Corros. Sci. 2021, 184, 109350. [Google Scholar] [CrossRef]
- Quadakkers, W.; Olszewski, T.; Piron-Abellan, J.; Shemet, V.; Singheiser, L. Oxidation of Metallic Materials in Simulated CO2/H2O-rich Service Environments Relevant to an Oxyfuel Plant. Materials Science Forum. Trans. Tech. Publ. 2011, 696, 194–199. [Google Scholar] [CrossRef]
- Kung, S.C.; Wright, I.G.; Shingledecker, J.P.; Tossey, B.M. Oxidation and carburization of alloys exposed to impure supercritical CO2. In Proceedings of the CORROSION 2017, New Orleans, LA, USA, 26–30 March 2017; p. 2017. [Google Scholar]
- Yang, Y.; Mao, S.; Yang, Q.; Xu, Z.; Ren, Y.; Wu, W. Effect of stress on corrosion behavior of martensitic and austenitic steels in supercritical carbon dioxide at 550 °C and 20 MPa. J. Supercrit. Fluid. 2023, 192, 105805. [Google Scholar]
- Chae, H.; Seo, S.; Jung, Y.C.; Huang, E.-W.; Jain, J.; Han, J.H.; Lee, S.Y. Evaluation of Supercritical Carbon Dioxide Corrosion by High Temperature Oxidation Experiments and Machine Learning Models. Metall. Mater. Trans. A 2022, 53, 2614–2626. [Google Scholar] [CrossRef]
- Gui, Y.; Liang, Z.; Shao, H.; Zhao, Q. Corrosion behavior and lifetime prediction of VM12, Sanicro 25 and Inconel 617 in supercritical carbon dioxide at 600 °C. Corros Sci. 2020, 175, 108870. [Google Scholar] [CrossRef]
- Brittan, A.; Mahaffey, J.; Adam, D.; Anderson, M. Mechanical and corrosion response of 316SS in supercritical CO2. Oxid Met. 2021, 95, 409–425. [Google Scholar] [CrossRef]
- Wang, X.; Wan, X.; Liu, Y.; Zhu, Y.; Zhang, F.; Lv, S.; Zhang, Y.; Du, Z. Failure mechanism of HR3C austenitic steel during creep at 650 °C. Fatigue Fract. Eng. M 2023, 46, 2017–2022. [Google Scholar] [CrossRef]
- Adam, B.; Teeter, L.; Mahaffey, J.; Anderson, M.; Árnadóttir, L.; Tucker, J.D. Effects of corrosion in supercritical CO2 on the microstructural evolution in 800H alloy. Oxid Met. 2018, 90, 453–468. [Google Scholar]
- Tan, L.; Ren, X.; Allen, T.R. Corrosion behavior of 9–12% Cr ferritic–martensitic steels in supercritical water. Corros Sci. 2010, 52, 1520–1528. [Google Scholar] [CrossRef]
- Ren, X.; Sridharan, K.; Allen, T.R. Corrosion of ferritic–martensitic steel HT9 in supercritical water. J. Nucl. Mater. 2006, 358, 227–234. [Google Scholar] [CrossRef]
- Wright, I.G.; Dooley, R.B. A review of the oxidation behaviour of structural alloys in steam. Int. Mater. Rev. 2010, 55, 129–167. [Google Scholar] [CrossRef]
- Kofstad, P.; Lillerud, K.P. On high temperature oxidation of chromium: II. Properties of and the oxidation mechanism of chromium. J. Electrochem. Soc. 1980, 127, 2410. [Google Scholar] [CrossRef]
- Kofstad, P.; Lillerud, K.P. Chromium transport through Cr2O3 scales I. On lattice diffusion of chromium. Oxid. Met. 1982, 17, 177–194. [Google Scholar] [CrossRef]
- Cotell, C.M.; Yurek, G.J.; Hussey, R.J.; Mitchell, D.F.; Graham, M.J. The influence of grain-boundary segregation of Y in Cr2O3 on the oxidation of Cr metal. Oxid. Met. 1990, 34, 173–200. [Google Scholar] [CrossRef]
- Guillou, S.; Cabet, C.; Desgranges, C.; Marchetti, L.; Wouters, Y. Influence of hydrogen and water vapour on the kinetics of chromium oxide growth at high temperature. Oxid. Met. 2011, 76, 193–214. [Google Scholar] [CrossRef]
- Stellwag, B. The mechanism of oxide film formation on austenitic stainless steels in high temperature water. Corros. Sci. 1998, 40, 337–370. [Google Scholar] [CrossRef]
- He, L.F.; Roman, P.; Leng, B.; Sridharan, K.; Anderson, M.; Allen, T.R. Corrosion behavior of an alumina forming austenitic steel exposed to supercritical carbon dioxide. Corros. Sci. 2014, 82, 67–76. [Google Scholar] [CrossRef]
- Rouillard, F.; Moine, G.; Tabarant, M.; Ruiz, J.C. Corrosion of 9Cr steel in CO2 at intermediate temperature II: Mechanism of carburization. Oxid. Met. 2012, 77, 57–70. [Google Scholar] [CrossRef]
- Liang, Z.; Gui, Y.; Wang, Y.; Zhao, Q. Corrosion performance of heat-resisting steels and alloys in supercritical carbon dioxide at 650 °C; and 15 MPa. Energy 2019, 175, 345–352. [Google Scholar]
- Costa, G.; Garg, A.; Brady, M.P. Corrosion of alumina-forming austenitic alloys under the supercritical carbon dioxide-based venus atmospheric surface conditions. Corros. Sci. 2023, 211, 110882. [Google Scholar] [CrossRef]
- Kofstad, P. On the formation of porosity and microchannels in growing scales. Oxid. Met. 1985, 24, 265–276. [Google Scholar] [CrossRef]
- Tan, L.; Ren, X.; Sridharan, K.; Allen, T. Effect of shot-peening on the oxidation of alloy 800H exposed to supercritical water and cyclic oxidation. Corros. Sci. 2008, 50, 2040–2046. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Zhang, J.; Young, D.J. Effect of Mn on oxide formation by Fe-Cr and Fe-Cr-Ni alloys in dry and wet CO2 gases at 650 °C. Corros Sci. 2016, 112, 110–127. [Google Scholar] [CrossRef]
- Young, D.; Huczkowski, P.; Olszewski, T.; Hüttel, T.; Singheiser, L.; Quadakkers, W. Non-steady state carburisation of martensitic 9–12%Cr steels in CO2 rich gases at 550 °C. Corros. Sci. 2014, 88, 161–169. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.