Abstract
This study focused on investigating the influence of zinc on tin pest, both alone and in combination with lead and copper. Based on the known composition of the organ pipe from Trpín, five model alloys were prepared, from which model samples were produced. The model alloys were exposed to low temperatures for 100 days or until complete degradation occurred. The kinetics of the transformation were compared for annealed and non-annealed samples. It was confirmed that the transformation is much faster in samples with retained internal stress. A comparison of the Avrami coefficients indicated similar nucleation behavior for both sample types. Phase transformation was observed in samples containing tin, copper, zinc, and lead, as well as those containing only copper and lead. This suggests that even a relatively small amount of zinc (0.25 wt.%) and copper (0.9 wt.%) can affect the course of tin pest in an alloy containing 13 wt.% lead. Transformation progressed more slowly in samples with only 0.25 wt.% zinc than in pure tin, likely due to the limited solubility of zinc in a tin with low concentrations of alloying elements. The crystallographic structure of both the model alloys and the original historical pipe material was studied using transmission electron microscopy (TEM). In almost all model samples, zinc was uniformly dissolved in the tin matrix. However, in the original pipe, zinc was primarily located at grain boundaries and in association with copper. This indicates that zinc was not intentionally added to the historical alloy but likely appeared in the alloy as a contaminant of impure copper.
1. Introduction
The organ is one of the oldest musical instruments, with records dating back to the third millennium BC. The basic parts of an organ are air management, a playing table (which may include keyboards, hinges, buttons, and pedals), an air box (air distributor), an organ case (which is usually made of oak) and pipes [1]. The organ in the church of St. Wenceslas in Trpín was built in 1840 by František Svítil the Elder. Due to neglected regular maintenance, its condition significantly deteriorated in the 20th century, as a result of which an extensive restoration of the organ took place in 2006–2008 [2]. Organ pipes are usually made from Sn-Pb alloys, where the Sn:Pb ratio can reach almost any value. A special case is the visible pipes, so-called prospect organ pipes, with their high Sn content causing a pleasant shiny appearance to the object. They might be endangered by the degradation process known as a tin pest. The tin pest is a phase transformation of white tin (β-Sn) to gray tin (α-Sn) [3,4,5]. This phenomenon occurs when the temperature drops below the thermodynamic stability threshold of 13.2 °C [6]. During this transformation, the crystal structure changes—the tetragonal structure rearranges into a cubic structure upon cooling [7]. As a result, the material undergoes severe mechanical damage, including deformation, cracking, and, in extreme cases, disintegration into powder. A typical indicator of the initial onset of tin pest is the formation of dark blisters on the surface of the “infected” object. Although the signs of tin pest are unique, it may be that some other low-temperature degradation mechanism is mistaken for tin pest. Among these mechanisms are surface oxidation [8], chemical reactions with formation of perovskite [9,10], electrochemical corrosion caused by different potentials of the tin matrix and intermetallic phases [11,12] and mechanical disintegration caused by crystallization pressure of intermetallic particles, usually in the form of whiskers [13,14].
The other pipes should be protected against tin pest due to the lead content. It is believed that even 0.1 wt.% of Pb successfully inhibits the low-temperature phase transformation [8,15]. This idea is not accepted generally, and some publications have reported occurrence of tin pest events in alloys with a high Pb content [16,17,18]. Other elements like Zn are known to accelerate the process [19]. Copper has shown contradictory results—in some studies, it has acted as an inhibitor [16,20], in others it has been an accelerator [7,21,22], had no effect at all [23] or caused growth of whiskers [13,14,24]. The discrepancy in the described Cu effect may be caused by several factors—real historical materials can exhibit heterogenous composition with depleted zones of Cu [25] compared to modern soldering materials that usually contain eutectic 0.7 wt.% of Cu. The cold working of the material also plays a crucial role [22]. As the solubility of Cu in the Sn matrix is extremely low [26], the state of the studied materials can vary due to possible supersaturation, and the initial stage includes the formation of precipitates corresponding to internal stresses in the material. Cu and Zn, together with Fe, were not added intentionally but were included in material of organ pipes from ores or from casting dies. The interactions of individual elements are not sufficiently described, but it is generally believed that the inhibiting influence of Pb overcomes the accelerating influence of other elements that are present [8].
This article describes the extraordinary behavior of one of the pipes from Trpín. The pipe material was composed of Sn–13 Pb–0.9 Cu–0.25 Zn (given in wt.%). The pipe exhibited severe damage, and the material was extremely brittle, as we already described in [27]. The typical blisters of tin pest were missing, but the material underwent extreme embrittlement. To clarify the mechanism, the microstructure of the original historical material was studied in detail by TEM. Subsequently, modern analogs were prepared, and their properties and microstructures were compared to those of the original material.
Understanding the various degradation mechanisms of tin alloys will not only help obtain information about historical materials. It is also necessary to identify modern lead-free solders that are, by their chemical composition, close to the original tin-rich materials.
2. Materials and Methods
For this study, historical organ pipe material from Trpín was used that was over 200 years old. The main elements were Sn–13 wt.%, Pb–0.9 wt.%, and Cu–0.25 wt.% Zn, with traces of other elements of minor impurity and elements for surface contamination, as detailed in [27].
To cast model samples, tin with 99.999% purity, copper, zinc, and lead with 99.99% purity were used. All samples were evacuated in quartz ampoules and melted in a muffle furnace for 48 h at 400 °C with repeated stirring and cooling in a furnace. They were subsequently cold-worked to a thickness of 0.1 to 0.2 mm (corresponding to 90% reduction). Selected samples were additionally annealed at 120 °C for 6 h. The release of internal stress slowed down the tin pest process [7], and this part of the experiment served to confirm the proper preparation of the samples.
One α-Sn particle was pressed into each sample (with a size of about 1 cm2) with a force of 100 N to inoculate the phase transformation [28]. The samples were stored at −50 °C in a TEFCOLD freezer (TEFCOLD, Vítkovice, Czech Republic) and observed using an optical microscope OLYMPUS SZX10 (Olympus, Tokyo, Japan) to evaluate the unaffected surface. Temperature of −50 °C was chosen as the condition of the highest phase transformation rate [29,30].
Chemical compositions of samples were analyzed using XRF (Axios spectrometer, PANanalytical, Almelo, Holland). The detailed microstructure was studied using a transmission electron microscope (TEM) Jeol 2200 FS (Jeol, Tokyo, Japan) operated at 200 kV and equipped with an Oxford Instrument EDS analyzer (Oxford Instruments, Abingdon, UK). The EDS data were measured in STEM mode with a step size of 1 nm.
The intended composition of model alloys derived from the composition of the original historical materials is given in Table 1.

Table 1.
Intended composition of model alloys per 1 g of alloy (given in g), __ means not detected.
Although the material was melted in the evacuated quartz ampoules, the composition varies, as shown in Table 2. The composition should be homogenous due to stirring; thus, divergences in compositions might be caused by errors during encapsulation.

Table 2.
Real composition of model alloys (given in wt.%), __ means not detected.
3. Results and Discussion
The alloys were cold-worked into small sheets, indented with α-Sn particles and exposed to a temperature of −50 °C. The samples were periodically observed, and Table 3 shows the records of the first occurrence of tin pest in the sample. The first occurrence of tin pest for SnCu1 and SnPb0.1 [31] is included in Table 3 to obtain complex information on the influence of system behavior, despite their composition not being relevant to the composition of historical material.

Table 3.
Time to occurrence of the first sign of tin pest (dark blisters) in the cold-worked and annealed materials; “__” means not detected.
The data in Table 3 provides information with high uncertainty, as the occurrence of the tin pest is dependent on several factors, like crystallography and environmental humidity [32]. On the other hand, once the process has started, the increase in degradation is uniform.
Our results are in good agreement with published observations [16,17,28] where binary Sn-Cu alloys underwent phase transformation and also where annealing slowed down the occurrence of tin pest. The discrepancies in actual time to tin pest occurrence are most likely caused by different experimental setups.
Figure 1 shows macrographs from the SnZn sample. On the left-hand side, we can see the initial materials just after inoculation by the α-Sn particle. The documentation of degradation after 100 days at a temperature of −50 °C is documented on the right-hand side of Figure 1. The degradation to cold-worked sheet is shown in the upper part of Figure 1, while in the lower part, the degradation of the annealed sheet is shown. Annealing influences the kinetic aspect when tin pest occurs, but not its growth.

Figure 1.
Macrographs from SnZn sample: (a) cold-worked initial sample; (b) cold-worked sample after 100 days at −50 °C; (c) annealed initial sample; (d) annealed sample after 100 days at −50 °C.
Figure 2 shows the macrographs of the SnPbCu alloy after inoculation and 100 days of exposure at a temperature of −50 °C. Surprisingly, the 13 wt.% of Pb did not protect the alloy from the tin pest. Let us assume that the influence on tin pest involves only those atoms that are dissolved in the matrix. Those that are present in separate phases, such as Pb or bound in the intermetallic or Cu in Cu6Sn5, play no role in the degradation of the matrix. Maximum solubility of Cu in the Sn matrix is only 0.0063 wt.% [26], while the Pb value is 1.3 wt.% [33]. Based on empirical observation, we can postulate that if the atomic radius of the alloying element is smaller than the Sn radius, then the element will serve as a tin pest accelerator [19]. Vice versa, larger atoms inhibit phase transformation. As the equilibrium solubility of Cu is lower than that of Pb, it might have a greater influence on the tin pest process.

Figure 2.
Macrographs from SnPbCu sample: (a) cold-worked initial sample; (b) cold-worked sample after 100 days at −50 °C.
Figure 3 shows the low-temperature transformation of the SnPbCuZn alloy, which, by its composition, is almost identical to the original historical material. Because of this, both cold-worked and annealed sheet degradation are shown. Although the degradation of the annealed sheet was slower, traces of tin pest (blisters) are visible in Figure 3d and are labeled by red arrows. The time to tin pest occurrence given in Table 3 is 3 days longer, but, as already mentioned, it is affected by many factors; let us assume that it is approximately the same for SnPbCuZn and SnPbCu alloys.

Figure 3.
Macrographs from SnPbCuZn sample: (a) cold-worked initial sample; (b) cold-worked sample after 100 days at −50 °C; (c) annealed initial sample; (d) annealed sample after 100 days at −50 °C (red arrows labeling blisters).
The low-temperature degradation of the SnCuZn alloy was complete in 100 days at a temperature of −50 °C, as shown in Figure 4, as both elements act as tin pest accelerators.

Figure 4.
Macrographs from SnCuZn sample: (a) cold-worked initial sample; (b) cold-worked sample after 100 days at −50 °C.
The influence of Zn on acceleration of tin pest was not observed in the SnPbZn alloy, as documented in Figure 5. The solubility of Zn in the Sn matrix is not described sufficiently. Some sources [34] state that there is almost no solubility, but others [35] estimate that the solubility is approx. 1 at%.

Figure 5.
Macrographs from SnPbZn sample: (a) cold-worked initial sample; (b) cold-worked sample after 100 days at −50 °C.
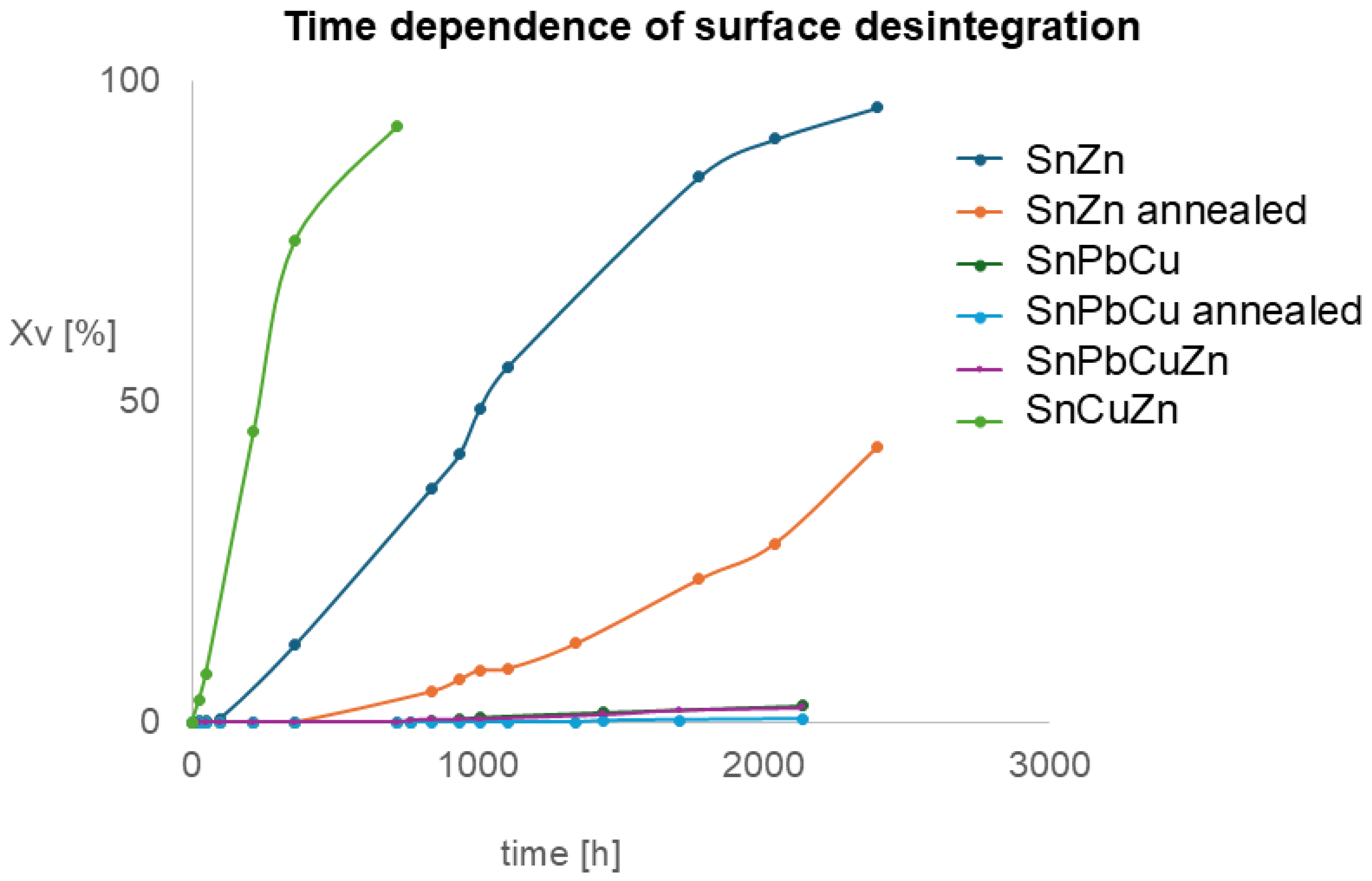
The degradation process was observed and recorded as the plot shown in Figure 6. Sample SnPbZn is not included as no degradation was observed.
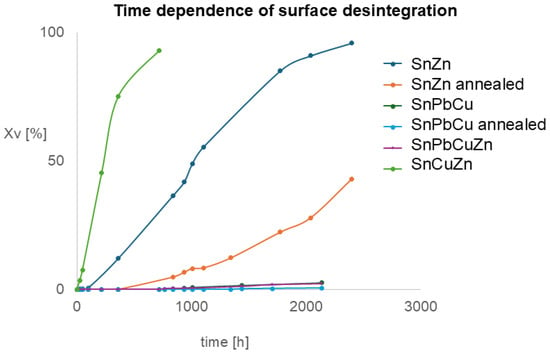
Figure 6.
Time dependence of surface decomposition measured by image analysis from macrographs: y-axis—surface affected by tin pest.
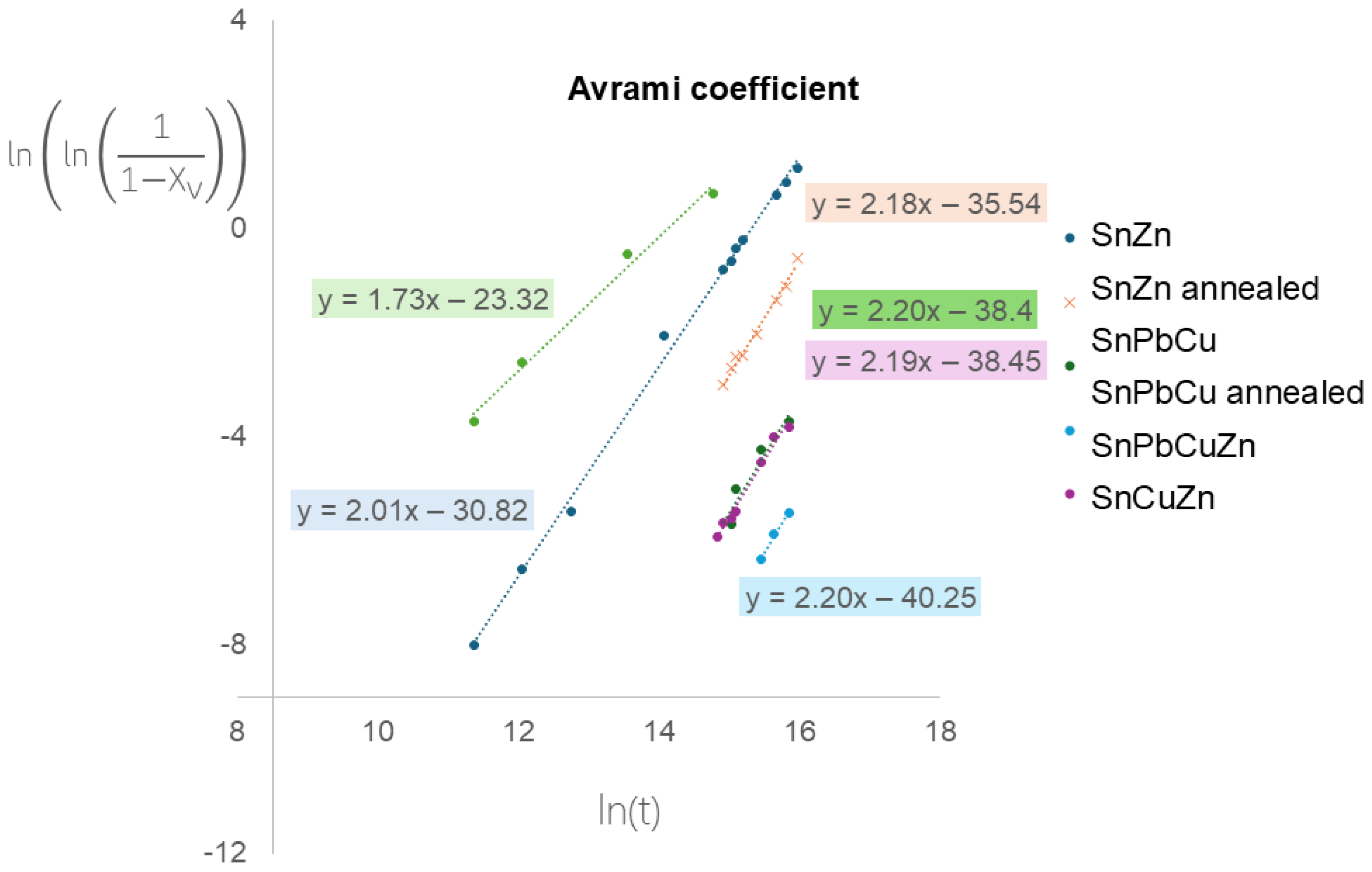
The data from Figure 6 were processed and are presented in Figure 7. The Avrami coefficients were estimated, which are a combination of unfirm nucleation and growth rates, n = D + 1, where D is the dimensionality of space in which crystallization occurs [36]. Interestingly, the values of Avrami coefficients are comparable with values of approximately two for all samples. It means that alloying and annealing change the initiation time but not the mechanism of progression. Our observations are in slight disagreement with published observations [28], where numerically obtained coefficients were influenced by annealing. This might be caused by different sample geometries, temperatures of experiments and detection techniques. The Avrami coefficient for tin pest was described to reach a value of three, which indicates a 3D process [37,38]. In our case, the value is about two, which comes from the sheet (2D) geometry of our samples.
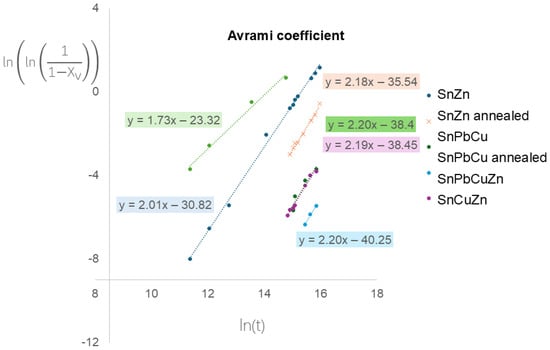
Figure 7.
Avrami coefficient plots.
To clarify the influence of alloying elements on matrix behavior, detailed observations were performed using STEM/EDS. Elemental maps in Figure 8 show presence of Zn particles in the Sn matrix. Based on this observation, we can claim that the solubility of Zn is strongly limited. It also explains negligible influence of Zn in Table 3.

Figure 8.
TEM/EDS elemental map from sample SnZn.
In the SnPbCu alloy shown in Figure 9, large particles of Pb and Cu are presented in the form of Cu6Sn5, as previously described [25]. In our case, both particles are large as the cooling in quartz ampules was slow.

Figure 9.
TEM/EDS elemental map from sample SnPbCu.
A similar distribution of Pb and Cu was observed in the SnPbCuZn alloy shown in Figure 10. No Zn particles were observed and Zn was distributed homogenously. Proving this idea using EDS point analysis was not possible as the concentration of Zn was too low for its reliable determination.
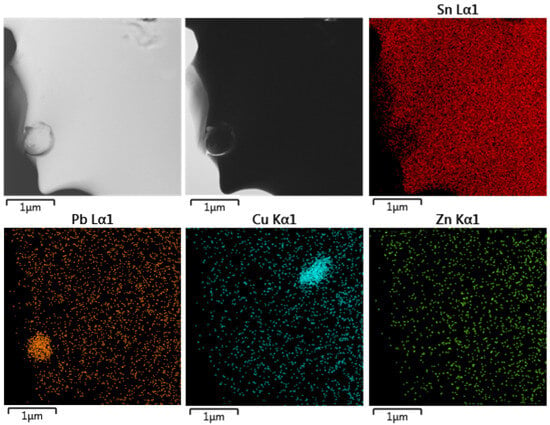
Figure 10.
TEM/EDS elemental map from sample SnPbCuZn.
Homogenous distribution of Zn was also observed in SnCuZn, as shown in Figure 11. In this case, the shortest time was needed for evolution of tin pest; see Table 3.

Figure 11.
TEM/EDS elemental map from sample SnCuZn.
Figure 12 and Figure 13 show the distribution of elements in SnPbZn material. In this case, it is possible to see Pb particles (Figure 12) and Zn particles (Figure 13). Unlike Cu, Pb most likely does not increase solubility of Zn in the Sn matrix. This might be the reason why this material was not affected by the tin pest process during exposure to low temperatures.

Figure 12.
TEM/EDS elemental map from sample SnPbZn—location 1.

Figure 13.
TEM/EDS elemental map from sample SnPbZn—location 2.
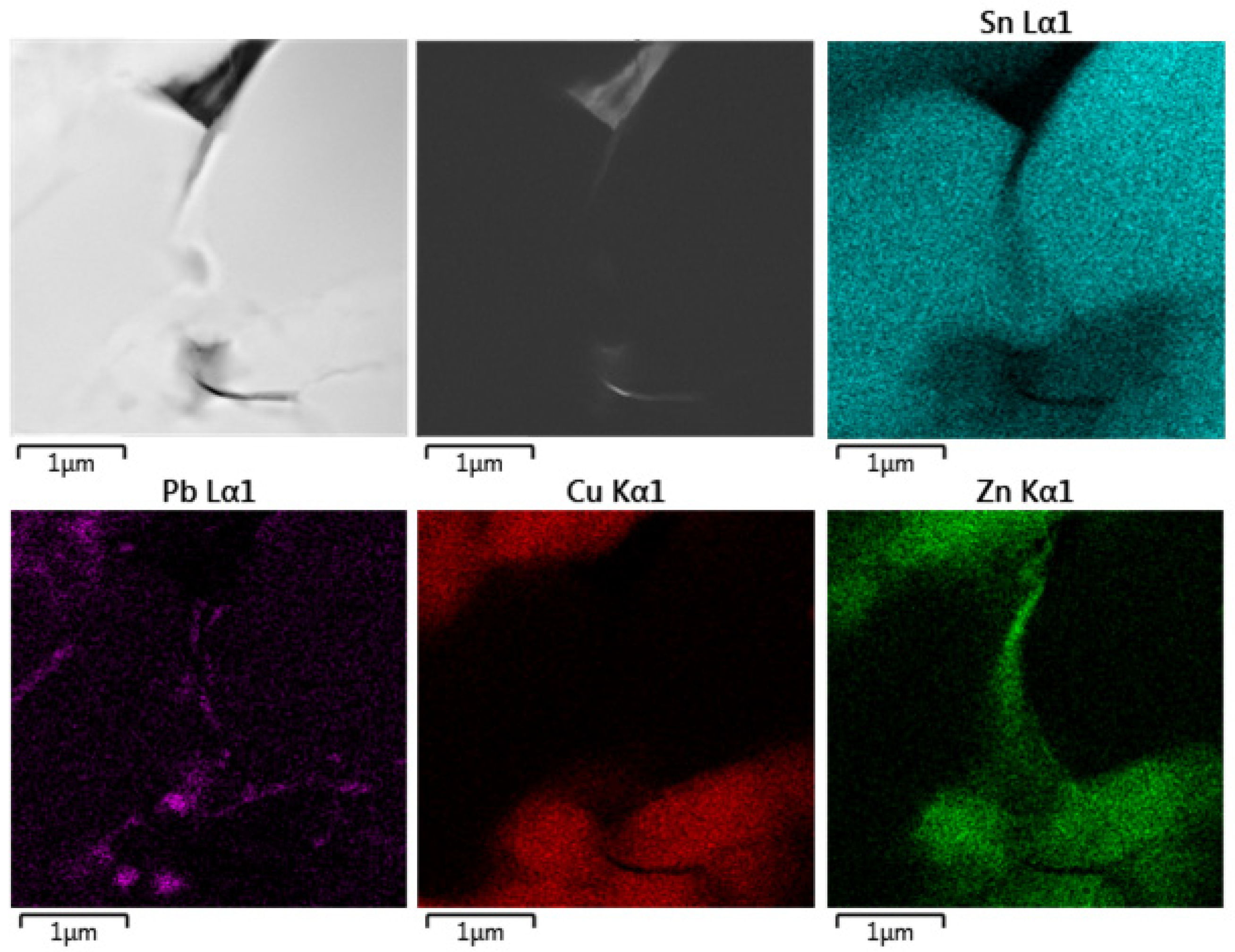
Figure 14 shows distribution of elements in the original historical material. It is clearly visible that the microstructure differs from the above-mentioned models. In this case, the Zn distribution overlaps with the Cu distribution, and the Zn is present in decorations at grain boundaries together with a small amount of Pb.
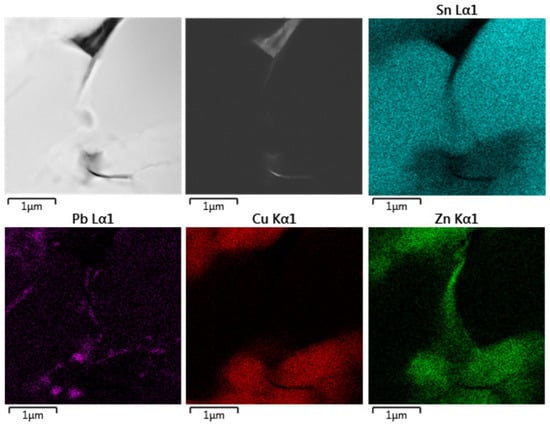
Figure 14.
TEM/EDS elemental map from original historical sample of organ pipe material.
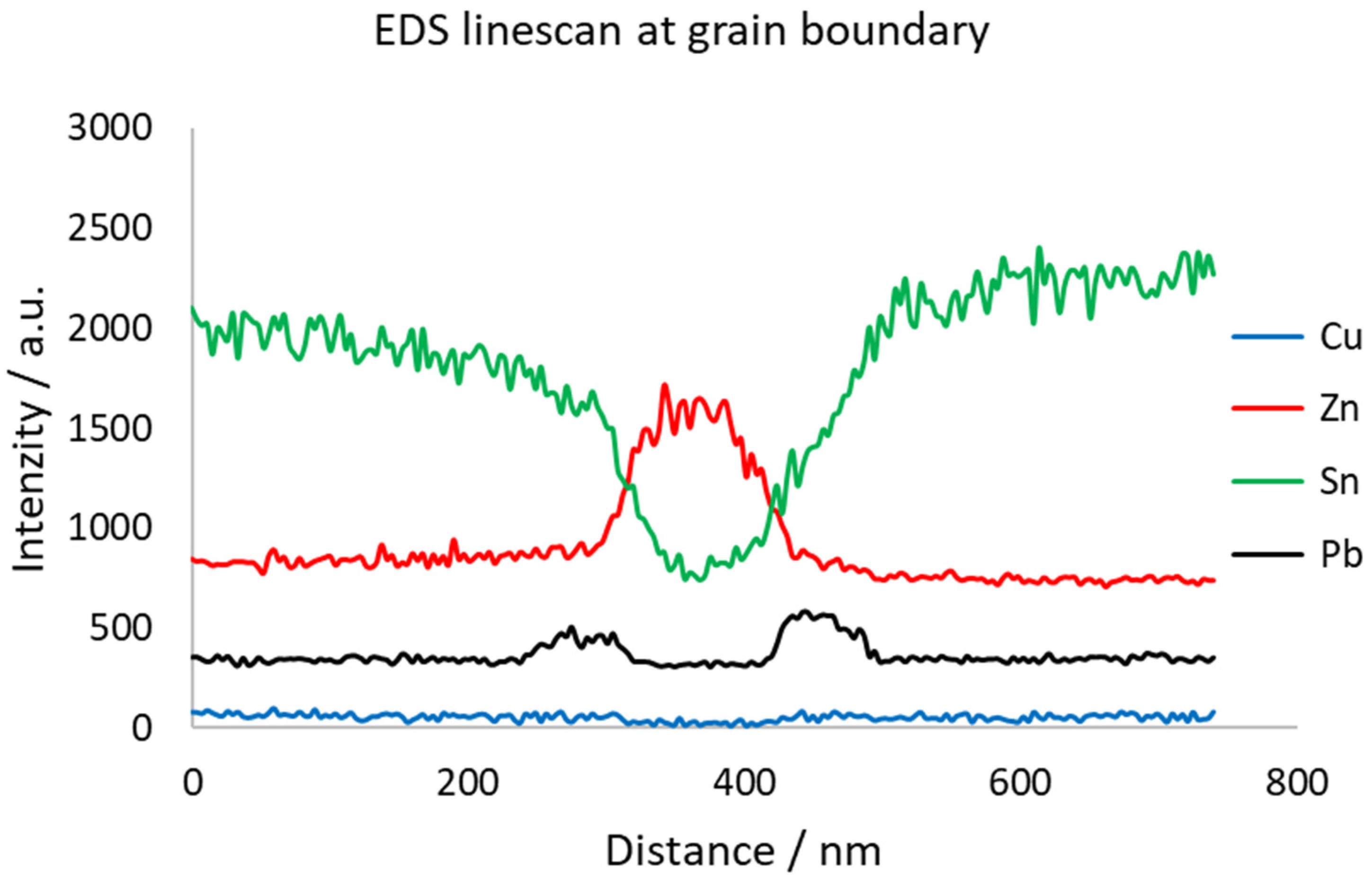
As shown in Figure 15, the grain boundary is significantly enriched by Zn. In this case, Zn is slightly oxidized, which might come from storing the thin TEM sample in the air’s atmosphere. On both sides of the Zn peak, there are Pb peaks. This sandwich-like structure is probably the weakest point of the material. Grain boundary diffusion of Zn and Pb caused weakening of the grain boundaries, resulting in extremely brittle behavior, as shown in Figure 16.
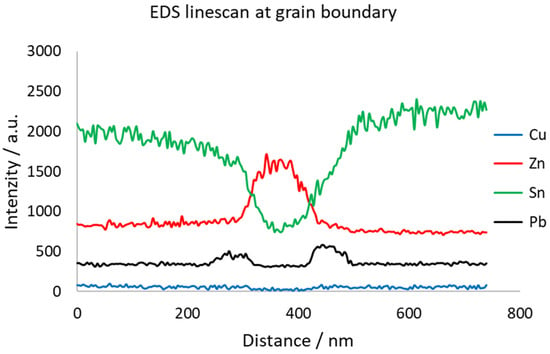
Figure 15.
EDS line profile over Zn-rich boundary in original historical sample of organ pipe material.

Figure 16.
Brittle damage of the pipe material, arrow showing crack in material.
Point EDS analysis was performed in the Cu- and Zn-rich particles, and the results are given in Table 4. The Cu:Zn ratio corresponds to common brass with Zn content at around 36 wt.% when taken as a Cu-Zn binary alloy. Unlike in model alloys, where the alloying elements were added separately, in the historical alloy, both Cu and Zn probably originated from the casting die. Pb detection was identified in the decoration of grain boundaries and Cu6Sn5 particle boundaries, as illustrated in Figure 14. Fe and Al probably represent contamination from sample preparation.

Table 4.
Composition of Cu- and Zn-rich particles in original historical sample of organ pipe material.
4. Conclusions
This study described mechanisms of damage in historical organ pipes from a church in Trpín (Czech Republic). The organ pipe was heavily deformed and exhibited severe embrittlement. Tin pest is a popular degradation mechanism and usually comes to the minds of researchers first when dealing with tin degradation. We have proven that in this case, the damage mechanism was caused by grain boundary diffusion.
A study of model analog alloys has proven that the rule that 0.1 wt.% of Pb protects tin alloy from tin pest is not always true. Tin pest accelerators might have a greater influence. This is probably in connection with their lower solubility in the Sn matrix. A lower content of the alloying element is needed to influence the properties of the material.
The influence of single elements was already described, although the published data is contradictory. We have proven that in the case of complex alloys, the interaction of alloying elements is key to alloy behavior. It was proven that presence of Cu in the alloy leads to an increase in Zn solubility in the Sn matrix. The increased solubility of tin pest accelerators (atoms with a smaller atomic radius than Sn) enhances tin pest propagation. A probable explanation for this might be induction of internal stresses in the material. The role of internal stresses on acceleration of tin pest propagation was documented during observation of cold-worked and annealed samples. Presence of Pb has no influence on Zn distribution in materials, and in this case, the retarding influence of Pb overcomes the accelerating influence of Zn.
Author Contributions
Conceptualization, D.F., Š.M. and A.M.; methodology, A.M., Š.M. and P.J.; validation, Š.M.; formal analysis, A.M.; investigation, E.G., D.F., A.M. and P.J.; resources, A.M.; data curation, A.M. and E.G.; writing—original draft preparation, A.M.; writing—review and editing, A.M.; supervision, A.M. and Š.M.; project administration, Š.M.; funding acquisition, Š.M. All authors have read and agreed to the published version of the manuscript.
Funding
The authors acknowledge the Czech Science Foundation for the financial support received under Project No. 22-05801S. The TEM sample preparation was done within Research Infrastructure NanoEnviCz, supported by the Ministry of Education, Youth and Sports of the Czech Republic under Project No. LM2023066.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Krátký, J.; Svoboda, Š. Nejvýznamnější Varhany České Republiky; Cpress: Brno, Czech Republic, 2019; ISBN 978-80-264-2859-6. [Google Scholar]
- Obec Trpín. Available online: https://www.trpin.cz/obec/pamatky (accessed on 19 January 2025).
- Peng, W.Q. An investigation of Sn pest in pure Sn and Sn-based solders. Microelectron. Reliab. 2009, 49, 86–91. [Google Scholar] [CrossRef]
- Eckert, A. Organ Pipes and Tin Pest. Mater. Corros.—Werkst. Und Korros. 2008, 59, 254–260. [Google Scholar] [CrossRef]
- Skorupa, W. Short Time Thermal Processing: From Electronics via Photonics to Pipe Organs of the 17th Century. Mater. Sci. Forum 2008, 573–574, 417–428. [Google Scholar] [CrossRef]
- Seitz, F.; Turnbull, D. Solid State Physics. Elsevier Sci. Technol. 1960, 11, 1–40. [Google Scholar] [CrossRef]
- Plumbridge, W.J. Tin pest issues in lead-free electronic solders. J. Mater. Sci. Mater. Electron. 2007, 18, 307–318. [Google Scholar] [CrossRef]
- Czerwiński, A.; Skwarek, A.; Płuska, M.; Ratajczak, J.; Witek, K. Tin Pest and Tin Oxidation on Tin-Rich Lead-Free Alloys Investigated by Electron Microscopy Methods. Solid State Phenom. 2012, 186, 275–278. [Google Scholar] [CrossRef]
- Machado, J.F.; Hieulle, J.; Vanderhaegen, A.; Redinger, A. Light-induced Degradation of Methylammonium Tin Iodide Absorber Layers. J. Mater. Chem. A 2025, 13, 517–525. [Google Scholar] [CrossRef]
- Lanzetta, L.; Webb, T.; Zibouche, N.; Liang, X.; Ding, D.; Min, G.; Westbrook, R.J.E.; Gaggio, B.; Macdonald, T.J.; Islam, M.S.; et al. Degradation mechanism of hybrid tin-based perovskite solar cells and the critical role of tin (IV) iodide. Nat. Commun. 2021, 12, 2853. [Google Scholar] [CrossRef]
- Stoulil, J.; Msallamová, Š.; Dušek, D.; Aliger, T.; Michalcová, A. Influence of intermetallic particles on localized corrosion of tin alloy organ pipes. Mater. Corros.—Werkst. Und Korros. 2025, 76, 1676–1683. [Google Scholar] [CrossRef]
- van de Ryck, I.; Biezen, E.; Leysen, E.; Adriaens, K.; Storme, A.; Adams, P.; Freddy. Study of tin corrosion: The influence of alloying elements. J. Cult. Herit. 2004, 5, 189–195. [Google Scholar] [CrossRef]
- Illés, B.; Krammer, O.; Hurtony, T.; Dušek, K.; Bušek, D.; Skwarek, A. Kinetics of Sn whisker growth from Sn thin-films on Cu substrate. J. Mater. Sci. Mater. Electron. 2020, 31, 16314–16323. [Google Scholar] [CrossRef]
- Williams, M.E.; Moon, K.-W.; Boettinger, W.J.; Josell, D.; Deal, A.D. Hillock and whisker growth on Sn and SnCu electrodeposits on a substrate not forming interfacial intermetallic compounds. J. Electron. Mater. 2007, 36, 214–219. [Google Scholar] [CrossRef]
- Msallamová, Š.; Jindrová, E.; Urbánek, Š. Kinetics of the Tin Phase Transformation. In Proceedings of the 23rd International Conference on Metallurgy and Materials (METAL), Brno, Czech Republic, 21–23 May 2014; pp. 1322–1327. [Google Scholar]
- Kariya, Y.; Williams, N.; Gagg, C.; Plumbridge, W. Tin pest in Sn-0.5 wt.% Cu lead-free solder. JOM 2001, 53, 39–41. [Google Scholar] [CrossRef]
- Plumbridge, W.J. Recent Observations on Tin Pest Formation in Solder Alloys. J. Electron. Mater. 2008, 37, 218–223. [Google Scholar] [CrossRef]
- Plumbridge, W.J. Further Observations on Tin Pest Formation in Solder Alloys. J. Electron. Mater. 2010, 39, 433–440. [Google Scholar] [CrossRef]
- Tin Pest, A Review. Available online: https://www.electronics.org/system/files/technical_resource/E29%26S05-6.pdf (accessed on 12 January 2026).
- Zenh, G.; McDonald, S.D.; Gu, Q.; Sweatman, K.; Nogita, K. Effects of element addition on the β→α transformation in tin. Philos. Mag. Lett. 2013, 94, 53–62. [Google Scholar] [CrossRef]
- Rogers, R.R.; Fydell, J. Effect of germanium on the transformation of white gray tin, at comparatively low temperature. J. Electrochem. Soc. 1953, 100, 161–164. [Google Scholar] [CrossRef]
- Joo, Y.J.; Takemoto, T. Transformation of Sn–Cu alloy from white tin to gray tin. Mater. Lett. 2002, 56, 793–796. [Google Scholar] [CrossRef]
- Cohen, E.; Van Lieshout, A.K.W.A. Effect of mechanical deformation on the velocity of transformation of polymorphic metals. III. Effect of metallic additions. II. Proc. K. Akad. Wet. Amsterdam. 1936, 39, 1174–1179. [Google Scholar]
- Hillman, D.; Wilcoxon, R.; Wieland, A. An Examination of the Tin Pest Phenomenon Over a 10-Year Period. J. Electron. Mater. 2022, 51, 6492–6502. [Google Scholar] [CrossRef]
- Michalcová, A.; Msallamová, Š.; Fink, D.; Friák, M. Minor Phases in Tin Rich Historical Materials. In Proceedings of the 32nd International Conference on Metallurgy and Materials, Orea Congress Hotel Brno, Brno, Czech Republic, 17–19 May 2023; pp. 579–582. [Google Scholar] [CrossRef]
- Snugovsky, L.; Cermignani, C.; Perovic, D.D.; Rutter, J.W. The Solid Solubility of Ag and Cu in the Sn Phase of Eutectic and Near-Eutectic Sn-Ag-Cu Solder Alloys. J. Electron. Mater. 2004, 33, 1313–1315. [Google Scholar] [CrossRef]
- Michalcová, A.; Fink, D.; Msallamová, Š.; Paleček, J.; Friák, M. The Zn Influence on Degradation Behaviour of Sn-based Materials. In Proceedings of the 33rd International Conference on Metallurgy and Materials (METAL 2024), Orea Congress Hotel Brno, Brno, Czech Republic, 22–24 May 2024; pp. 432–438. [Google Scholar] [CrossRef]
- Skwarek, A.; Illés, B.; Hurtony, T.; Bušek, D.; Dušek, K. Effect of Recrystallization on β to α-Sn Allotropic Transition in 99.3Sn–0.7Cu wt.% Solder Alloy Inoculated with InSb. Materials 2020, 13, 968. [Google Scholar] [CrossRef] [PubMed]
- Roupcová, P.; Švábenská, E.; Schneeweiss, O.; Havlíček, L.; Michalcová, A.; Msallamová, Š.; Friák, M. Low Temperature Investigations of Phase Transformation in Pure Tin. In Proceedings of the 33rd International Conference on Metallurgy and Materials (METAL), Brno, Czech Republic, 22–24 May 2024; pp. 581–586. [Google Scholar] [CrossRef]
- Cornelius, B.; Treivish, S.; Rosenthal, Y.; Pecht, M. The phenomenon of tin pest: A review. Microelectron. Reliab. 2017, 79, 175–192. [Google Scholar] [CrossRef]
- Fink, D.; Michalcová, A.; Msallamová, Š. Characterization ofhistorical tin materials from the perspective of tin pest. In Proceedings of the 29th International Conference on Materials and Technology (ICMT29), Portorož, Slovenia, 2-4 October 2024; Institute of Metals and Technology: Ljubljana, Slovenia, 2024; Volume 58, p. 1316, (Book of Abstracts). [Google Scholar] [CrossRef]
- Michalcová, A.; Msallamová, Š.; Fink, D.; Kubásek, J.; Friák, M. The microscopic study of the evolution of the phase transformation in the tin after the indentation of an inoculator. Manuf. Technol. 2024, 24, 83–86. [Google Scholar] [CrossRef]
- Karakaya, I.; Thompson, W.T. The Pb–Sn (Lead-Tin) system. J. Phase Equilib. 1988, 9, 144–152. [Google Scholar] [CrossRef]
- Manasijević, D.; Balanović, L.; Marković, I.; Gorgievski, M.G.; Stamenković, U.; Božinović, K. Microstructure, melting behavior and thermal conductivity of the Sn–Zn alloys. Thermochim. Acta 2021, 702, 178978. [Google Scholar] [CrossRef]
- Wei, X.; Huang, H.; Zhou, L.; Zhang, M.; Liu, X. On the advantages of using a hypoeutectic Sn–Zn as lead-free solder material. Mater. Lett. 2007, 61, 655–658. [Google Scholar] [CrossRef]
- Cahn, J.W. Transformation kinetics during continuous cooling. Acta Metall. 1956, 4, 572–575. [Google Scholar] [CrossRef]
- Irzhak, T.F.; Mezhikovskii, S.M.; Irzhak, V.I. The Physical Meaning of the Avrami Equation in Oligomer Curing Reactions. Polym. Sci. Ser. B 2008, 50, 201–203. [Google Scholar] [CrossRef]
- Zeng, G.; McDonald, S.D.; Gu, Q.; Matsumura, S.; Nogita, K. Kinetics of the β→α Transformation of Tin: Role of α-Tin Nucleation. Cryst. Growth Des. 2015, 15, 5767–5773. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.