Abstract
Alumina (Al2O3) reinforced copper matrix composites are widely used in the electronic industry, rail transit, and other fields due to their excellent electrical conductivity, ductility, and wear resistance. However, due to problems such as non-wetting and thermal expansion differences between alumina and Cu, weak interfacial bonding can easily reduce physical and thermal properties. A uniform silver layer was deposited on Al2O3 via chemical plating to enhance interface bonding with copper. Al2O3@Ag/Cu composites with 1–3 wt.% Al2O3 were prepared by rapid hot-press sintering. The effects of plating temperature and Al2O3 content on microstructure and properties were investigated. The results show that the optimum coating temperature is 25 °C, and a thin and uniform silver coating can be formed. This effectively improved Al2O3–Cu interface bonding while maintaining 77.8% of copper’s thermal conductivity (320.7 W/(m·K)). The composites showed improved wear resistance with increasing Al2O3 content. At 3 wt.% Al2O3@Ag, the wear rate was 3.36 × 10−5 mm3/(N·m), 84.4% lower than pure copper, with plow groove wear as the main mechanism.
1. Introduction
Cu composites have been widely applied in electrical contact materials and friction materials due to their excellent electrical properties, thermal properties, mechanical properties, and wear resistance [1,2,3,4,5]. Nowadays, the development of Cu composite materials requires high mechanical strength, high electrical and thermal conductivity, as well as excellent thermal stability and wear resistance [6,7,8,9]. Cu is considered the most important basic material in industrial applications. It is common to add hard ceramic particles (such as alumina, zirconia, silicon carbide, etc.) to Cu and other soft metal phases to improve the strength and wear resistance of the materials [10,11,12,13,14,15,16].
Alumina (Al2O3) possesses excellent characteristics such as low density, high hardness, high melting point, and good chemical stability. The addition of alumina particles to a Cu matrix can enhance the material’s strength and serve as a support for the matrix, thereby improving the material’s resistance to plastic deformation. The addition of alumina particles affects the deformation and recrystallization of the Cu matrix. The fine and closely arranged alumina particles exert a pinning effect on the movement of dislocations, significantly reducing grain size and inhibiting grain growth. They also enhance microhardness, thus strengthening the matrix [17]. In recent years, the characterization and application of alumina-reinforced Cu composite materials have attracted widespread attention. However, there is a significant difference in density and coefficient of thermal expansion between alumina and Cu. The poor wetting property at the alumina–Cu interface results in a weak interface bond. Therefore, the interface bond strength between Al2O3 and Cu and the uniform distribution of alumina are the primary factors affecting the performance of alumina/Cu composite materials [18].
Surface modification is a major approach to improving interface bonding. Surface metallization can not only enhance the interface bond strength, but also inhibit particle agglomeration to some extent. Shang et al. [19] obtained a nickel coating on the surface of Al2O3 through chemical deposition. The nickel-coated Al2O3 particles (Al2O3@Ni) were used as particle reinforcements in an iron matrix, and Al2O3@Ni/Fe composite materials were prepared by spark plasma sintering. Results showed that by optimizing the chemical deposition process, the Al2O3 surface was uniformly covered with Ni, and the Ni coating tightly bonded with Al2O3. The Ni coating not only improved the wettability between Al2O3 and the iron matrix, but also promoted the diffusion and reaction at the Al2O3–iron interface. Güler et al. [20] fabricated silver-plated and uncoated Cu composite materials reinforced with Al2O3 particles (0.5, 1, 2, 3, and 5 wt.%) using hot pressing. They revealed the positive impact of silver coating and Cu particle grinding on the physical properties (such as density, hardness, and electrical conductivity) of Al2O3-reinforced Cu composite materials. Moustafa et al. [21] applied a chemical deposition method to deposit a layer of nickel metal on SiC and Al2O3 reinforcement particles. They prepared nickel-coated and uncoated SiC and Al2O3 particle-reinforced Cu composite materials using powder metallurgy forming methods. Results showed that the Cu composite materials reinforced with nickel-coated particles exhibited higher relative density and lower porosity compared to the uncoated composite materials. The coated composite materials also demonstrated superior yield strength and compressive fracture strength. Cherng-Yuh et al. [22] utilized a thin layer of La-doped Ti for metallization treatment on the surface of alumina, achieving bonding within the temperature range of 250–350 °C. They produced uniform, thermally stable, and high-strength alumina/Cu joints after the vacuum hot pressing process. Yan et al. [23] developed a convenient and environmentally friendly method for the deposition of high-quality Cu layers on ceramics through surface modification and chemical deposition. By evaluating water contact angle, XPS, FT-IR, SEM, EDS, and adhesion tests, they found that the modified treatment significantly enhanced the adhesion between the substrate and Cu layer. The resulting deposited Cu layer exhibited excellent performance in terms of adhesion and conductivity.
The size and content of alumina particles also significantly affect the performance of composite materials. As the alumina content in the Cu matrix increases, the composite materials demonstrate higher wear resistance, hardness, and thermal stability [24,25]. Sedighi et al. [26] prepared alumina particle-reinforced aluminum-based composite materials through the process of thermal accumulative rolling. They tested the microstructure and mechanical properties of the composite materials with different alumina particle contents using tensile tests, Vickers microhardness tests, and scanning electron microscopy. The results showed that increasing the volume percentage of alumina particles improved the strength and average microhardness of the composite materials. Vogel et al. [27] produced aluminum-based composite materials containing a large amount of alumina through powder metallurgy. They systematically investigated the influence of alumina content and morphology on the microstructure and mechanical properties of the composite materials. The research indicated that alumina content, morphology, and particle size play important roles in determining the strength and deformation mechanisms of the composite materials.
In this study, Al2O3@Ag/Cu composites with different reinforcement contents (1–3 wt.%) were fabricated via rapid hot-press sintering. The core innovation lies in employing an electroless silver-plated layer as an interfacial engineering strategy to address the poor wettability between Al2O3 and the Cu matrix. The influence of chemical deposition temperature on the silver coating’s characteristics was first investigated to determine the optimal process. Subsequently, the phase composition, microstructure, mechanical properties, thermal conductivity, and tribological behavior of the composites were systematically studied, with a particular focus on elucidating the synergistic effect of the Ag coating and Al2O3 content on comprehensive performance. This work aims to provide valuable insights for designing high-performance copper matrix composites with balanced strength, conductivity, and wear resistance.
2. Experimental Procedure
2.1. Preparation
In this study, ultrafine Cu powder with an average particle size of 6–7 μm (purity > 99.7%, density 8.96 g/cm3) was produced by Beijing Xingrongyuan Technology Co, Ltd. Beijing, China. The alumina powder, with an average particle size of 10 μm (Al2O3, purity > 99.9%), was produced by Naibo Detection Technology Co., Ltd., Shanghai, China. Silver nitrate was used as a surface modifier, and formaldehyde was used as a reducing agent. In addition, the chemical reagents used in this experiment were modified on the surface of alumina powder through chemical deposition process. All chemicals used in the experiment (including tartaric acid sodium hydroxide, sodium hydroxide, hydrochloric acid, and ethanol) were of analytical grade quality.
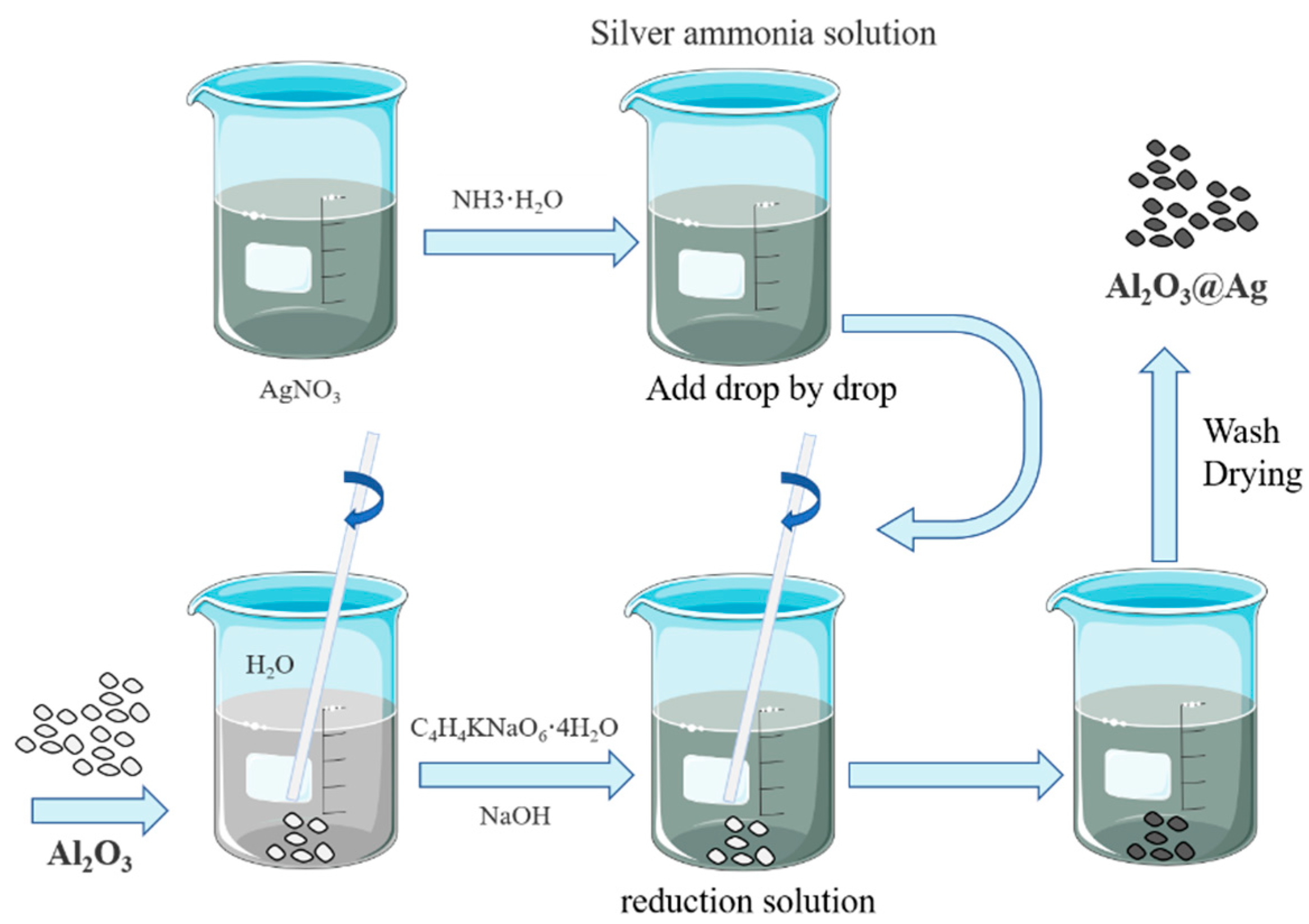
Alumina silver plating powder was prepared by electroless plating method, and the preparation flow chart is shown in Figure 1.
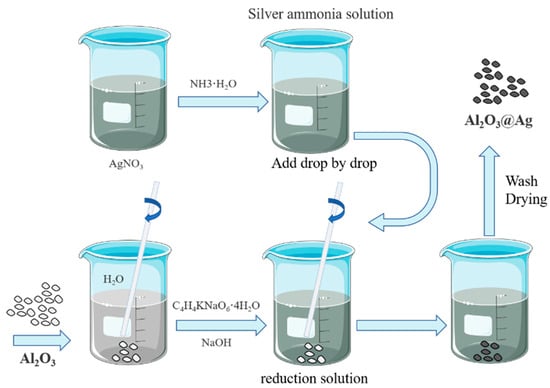
Figure 1.
Alumina silver plating powder preparation flow chart.
It mainly includes the following steps: (1) Preparation of silver-ammonia solution: Dissolve 20 g of silver nitrate in 500 mL of deionized water, slowly add 30 mL of ammonia solution to the solution while stirring continuously. The solution will initially become turbid and then gradually become clear and transparent. Add deionized water to make a total volume of 1 L. (2) Preparation of reducing solution: Add 8 g of Alumina to a beaker, then add 500 mL of deionized water. Use magnetic stirring to ensure complete dispersion of Alumina in the water. Add 100 g of potassium sodium tartrate and dissolve completely while stirring with a magnetic stirrer. Add deionized water to make a total volume of 1 L. (3) Chemical silver plating: To investigate the effect of temperature on silver plating on the surface of alumina, the experiment is conducted at 25 °C, 30 °C, and 40 °C while keeping other parameters constant. The silver–ammonia solution is added dropwise into the reducing solution at a rate of 200 mL/min, while the alumina is kept suspended in the solution by magnetic stirring. The reaction starts immediately upon the addition of the solution, and the milky white solution gradually turns dark brown and then silver-gray. The reaction stops once all the solutions are added. A faint silver mirror reaction can be observed on the walls of the beaker. A small amount of the solution is added to hydrochloric acid, and no white precipitate is formed. Then, the solution is filtered and washed repeatedly with deionized water and anhydrous ethanol until the pH value approaches neutral. Finally, the alumina coated with silver powder (Al2O3@Ag) is obtained by vacuum drying at 60 °C.
O. Güler et al. [20] explored the effect of Al2O3 content (0.5, 1, 2, 3, and 5 wt.%) on Cu composites. Studies have shown that when the amount of Al2O3 added exceeds 3 wt.%, the mechanical properties of the composites will be greatly reduced. Therefore, the alumina contents of 1, 2, and 3 wt.% were selected for this study. Silver-plated alumina/Cu composite material was prepared by rapid hot-press sintering. Firstly, the silver-plated alumina powder (Al2O3@Cu = 1 wt.%, 2 wt.%, 3 wt.%) and Cu powder were mixed using a planetary ball mill. The mixing was performed in a stainless-steel ball milling tank with 6 mm diameter steel balls. The ball milling time was 3 h at a rotation speed of 300 rpm. The powder-to-ball ratio was 5:1. After thorough mixing, the mixture was placed into a cylindrical graphite mold with an inner diameter of 40 mm. A certain pressure was applied for a period to allow the composite powder to uniformly fill the graphite mold. After the pre-pressing step, the cylindrical samples with a diameter of 40 mm and a thickness of approximately 3 mm were prepared under the conditions of 40 MPa, vacuum of 10 Pa, and insulation and pressure holding at 750 °C for 5 min. The samples were heated to 600 °C at a rate of 100 °C/min, and then heated to 750 °C at a rate of 75 °C/min to reduce the temperature difference between the actual temperature and the heating temperature. After the insulation stage, the samples were kept under pressure for 10 min during the furnace cooling period to improve the density. As a comparison, pure Cu and alumina/Cu (Al2O3 = 1 wt.%, 2 wt.%, 3 wt.%) composite materials without chemical silver plating were prepared using the same raw materials and fabrication process.
2.2. Characterization
The composite powders and bulk composite materials were observed for their microstructure, elemental distribution, and surface wear morphology using a scanning electron microscope (SEM, JSM-IT500 A, JEOL Ltd., Tokyo, Japan) equipped with an energy-dispersive X-ray spectrometer (EDS). X-ray diffraction analysis (XRD, Dmax/2500 PC, Rigaku, Tokyo, Japan) was conducted on alumina, silver-plated alumina, and their composite materials to determine the phase composition. The wavelength used in the XRD instrument was 1.5406 Å, with a tube voltage of 40 KV and tube current of 200 mA. The scanning speed was set at 5°/min. The densities of sintered samples were measured by means of an Anton Paar true-density instrument (Ultrapyc 5000, Anton Paar, Graz, Austria). By calculating the mass fraction of Al2O3, Ag coating, and Cu matrix in the composite, the theoretical density of the composite was obtained. The actual density of the composites was further measured by the Anton Paar true-density instrument. The average value was obtained by multiple measurements. The relative density is equal to the real density divided by the theoretical density. Reciprocating friction-wear tests were performed using a multifunctional tribometer (Rtec MFT-5000-H, Rtec instruments, San Jose, CA, USA). To minimize experimental errors, the frictional properties of the samples were tested three times. GCr15 with a diameter of 6.35 mm was used as the counter material for friction. The single reciprocating motion had a stroke of 5 mm, and the total running time was 10 min. The worn surface 3D profiles of the samples were observed using a laser scanning confocal microscope (LSM 700, Zeiss, Jena, Germany), and the wear volume of the samples was calculated from the cross-sectional profile of the wear track. Each group of experiments was tested 5 times to take the average value, and the error bar was drawn by analyzing the data fluctuation. The tribological test parameters are shown in Table 1.

Table 1.
Tribometer testing parameters (ball-on-disk configuration).
3. Results and Discussion
3.1. Microstructure Analysis of Al2O3 and Al2O3@Ag/Cu Composites
Figure 2 shows SEM images of the alumina powder before and after silver plating at different temperatures. As shown in Figure 2a, the alumina powder used in this experiment was irregularly shaped with a particle size of approximately 10 μm. Figure 2b–d shows the silver-plated alumina powder obtained by chemical plating at different temperatures. The microstructure in the figure indicates that the silver particles were uniformly deposited on the surface of the alumina at room temperature, and the silver layer had good uniformity and coverage. To accurately determine the thickness of the deposited silver layer, a comprehensive analysis was conducted by combining the mass change of the powders before and after electroless plating with a systematic comparison of the particle size distributions of the pristine and silver-coated alumina. The coating thickness was derived from the correlation between the measured mass gain and the increase in median particle size, yielding an average silver layer thickness of approximately 245 nm. A well-plated layer has a positive effect on improving the performance of composite materials. No free silver particles were observed except on the surface of the alumina. As the temperature increased, the silver layer on the surface of the alumina became thinner, and the size of the silver particles decreased. At 40 °C, the silver particles were deposited on the surface of the alumina in the form of aggregated particles without forming a well-covered silver film. A large area of exposed alumina was still visible in the field of view. In addition, a large number of free silver particles were present around the alumina.

Figure 2.
SEM images of alumina and silver-plated alumina. (a) Al2O3 raw powder; silver-plated alumina at different temperatures: (b) 25 °C; (c) 30 °C; (d) 40 °C.
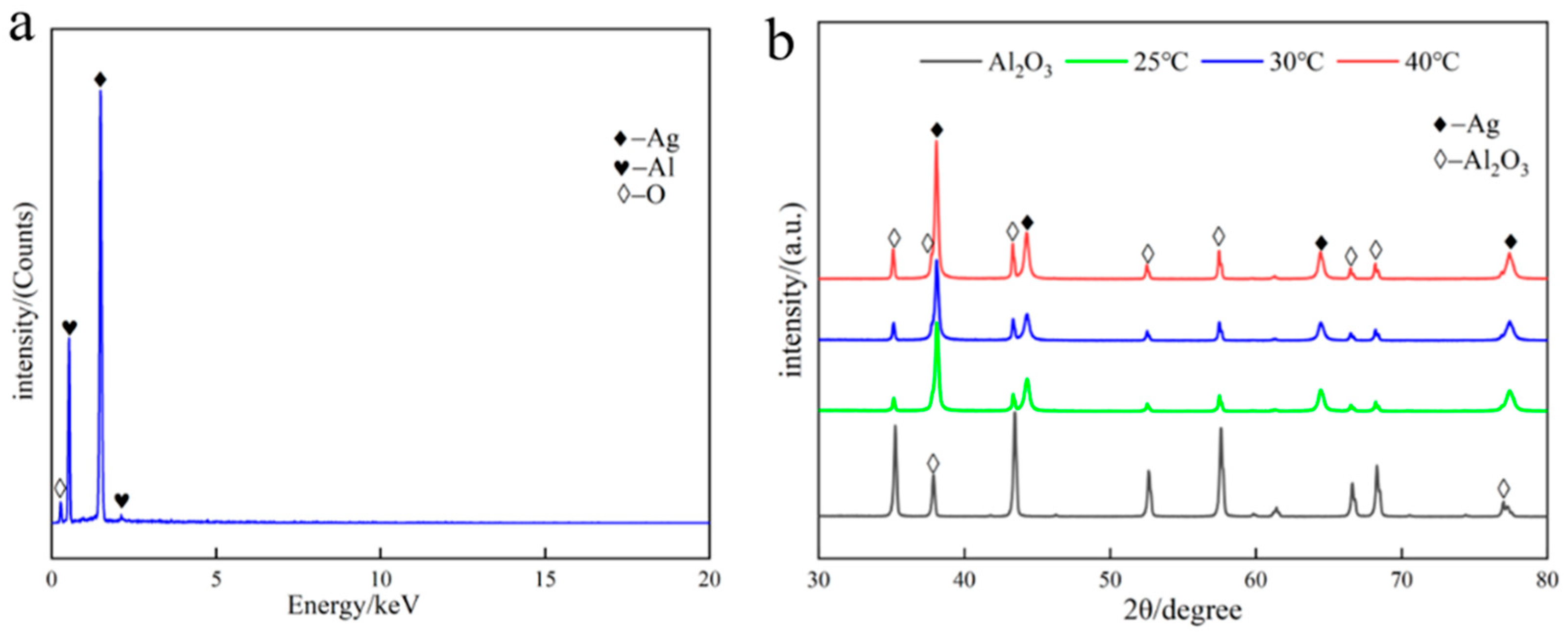
The silver-plated alumina powder obtained at different temperatures was analyzed by EDS spectroscopy and characterized by XRD, as shown in Figure 3. The EDS spectra and XRD patterns indicate the presence of characteristic peaks for silver particles, and the results show that no impurity phase was formed. The XRD results show characteristic diffraction peaks corresponding to the different crystallographic planes of silver, namely, the (111) plane at 2θ = 38.0°, the (220) plane at 2θ = 44.24°, the (220) plane at 2θ = 64.4°, and the (311) plane at 2θ = 77.36°. Moreover, with increasing temperature, the relative peak intensity of the alumina powder in the XRD spectrum increases, indicating an increase in the exposed area of the alumina. This suggests a decrease in the coverage of the silver-plated layer, which is consistent with the SEM results shown in Figure 2.
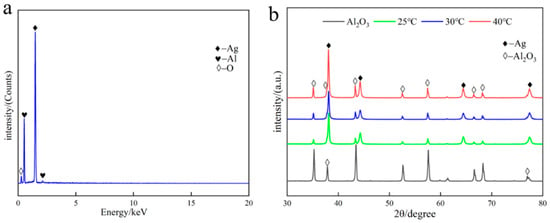
Figure 3.
EDS spectra and XRD patterns of silver-plated alumina. (a) EDS spectra and (b) XRD patterns of silver-plated alumina at different temperatures.
Alumina-reinforced Cu composites with untreated and silver-plated surfaces were prepared by powder metallurgy. As shown in Figure 4, the composite material was polished by sandpaper, polished, and then corroded with the grain boundary, and the metallographic SEM morphology after corrosion was obtained. As shown in Figure 4b, when the content of Al2O3@Ag is 3 wt.%, the distribution of Al and O elements in Cu is uniform. In addition, the uniform distribution of Ag element is basically consistent with the distribution of Al and O elements. Figure 4c shows the high-magnification SEM morphology of the Al2O3/Cu composite material. The black particles are alumina, and the light color area is Cu matrix. The presence of voids at the interface indicates poor interfacial bonding. Figure 4d shows the SEM image of the Al2O3@Ag/Cu composite material. It can be seen that the bonding interface between the alumina and the Cu substrate is good, and no obvious pores are observed. Compared with the alumina without electroless plating (Figure 4c), the interface bonding quality is significantly improved. The improvement of the bonding interface has a positive effect on the performance of the composite material.

Figure 4.
Microstructure of alumina–Cu composites. (a) Al2O3@Ag/Cu composites (Al2O3 = 3 wt.%); (b) EDS results in a; (c,d) high-magnification SEM morphology of Al2O3/Cu and Al2O3@Ag/Cu composites.
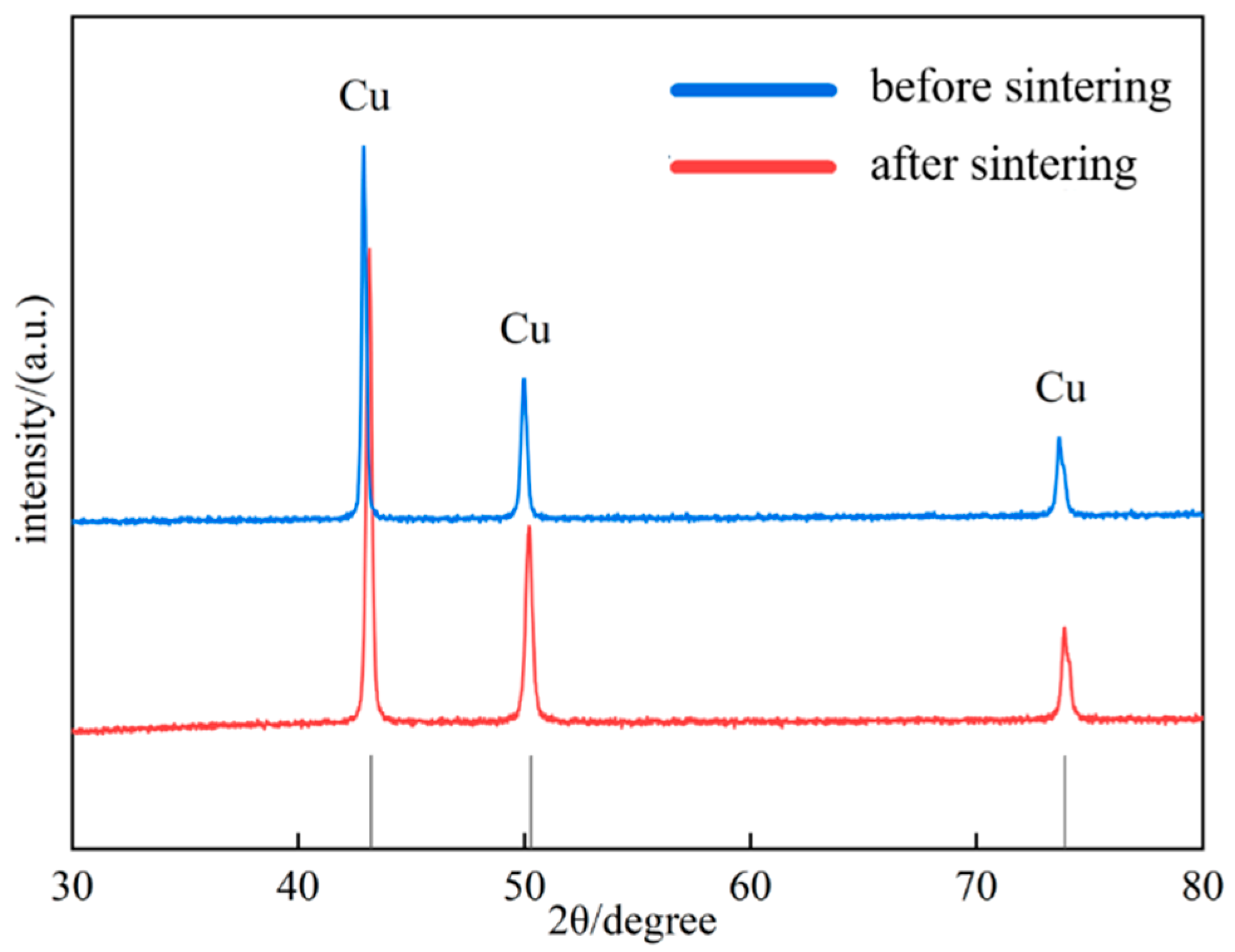
XRD analysis was performed on the Al2O3@Ag/Cu composite material, and the results are shown in Figure 5. From the figure, all peaks correspond to the characteristic peaks of Cu, and a comparison with the standard PDF card indicates that the characteristic peak positions of the composite powder before sintering correspond one-to-one with those of the PDF card. After sintering, a blue shift in the characteristic peaks of the composite material appears. This phenomenon is caused by the diffusion of silver atoms from the coating into the Cu lattice during sintering, resulting in lattice expansion and a consequent shift of the diffraction peaks towards lower angles [28]. Meanwhile, we calculated the lattice constants of the composite material before and after sintering, which were 3.6286 Å and 3.64276 Å, respectively, consistent with the above theory.
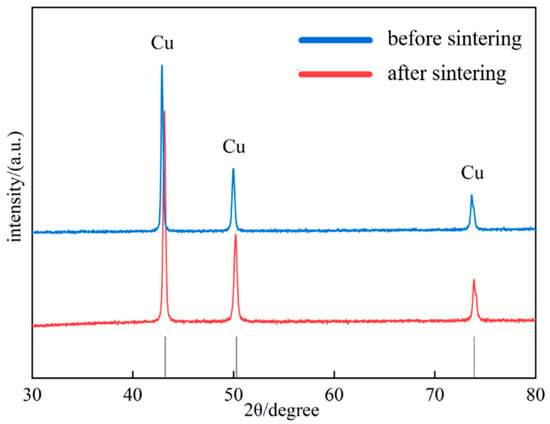
Figure 5.
XRD patterns of Al2O3@Ag/Cu composites.
3.2. Physical and Mechanical Properties
The physical and mechanical properties of Al2O3/Cu and Al2O3@Ag/Cu composite materials (Al2O3, Al2O3@Ag = 1–3 wt.%) are shown in Table 2. The results show that with the increase of alumina content, the relative density and conductivity of the composites decrease gradually, and the Vickers hardness increases gradually. Moreover, after silver plating on the surface of Al2O3, the relative density, Vickers hardness, and conductivity of the composite material are higher than those of the non-silver-plated composite material. First, during the powder sintering process, micro-pores are easily formed at the grain boundary due to the density and intrinsic differences between alumina and Cu. In addition, due to the low relative density of Al2O3/Cu composites, micro-pores are easily formed inside the material. The higher the Al2O3 content, the lower the relative density. During the sintering process, the significant difference in the coefficient of thermal expansion (CTE) between Al2O3 and Cu can induce interfacial thermal stress, potentially damaging the interface bonding, and promote decohesion or micro-crack formation, particularly at triple points. Furthermore, increasing the Al2O3 content introduces a greater number of these incompatible interfaces and triple junctions into the Cu matrix. The rigid Al2O3 particles hinder full densification during sintering, making it difficult to eliminate pores that are initially trapped at these locations, which consequently leads to an increase in overall porosity and a decrease in the composite density. On the other hand, the silver plating on the alumina surface acts as a compliant and wettable interlayer. It effectively mitigates the CTE mismatch stress, improves the interfacial bonding strength, reduces particle aggregation, and promotes a more continuous Cu matrix [29]. This, in turn, facilitates densification and significantly reduces the formation and retention of pores at the interfaces and triple points. At the same time, the rapid sintering process, under the influence of high temperature and pressure, promotes rapid densification and the formation of sintering necks between particles. Crucially, the short sintering duration effectively limits the subsequent grain growth and coarsening, thereby facilitating the formation of a fine-grained microstructure, which contributes to strengthening via the Hall–Petch mechanism [15,18]. Finally, the presence of Ag coating fills the gap between the alumina and Cu matrix, which contributes to the densification of Cu composites and improves their comprehensive properties.

Table 2.
Physical and mechanical properties of Al2O3/Cu and Al2O3@Ag/Cu composites.
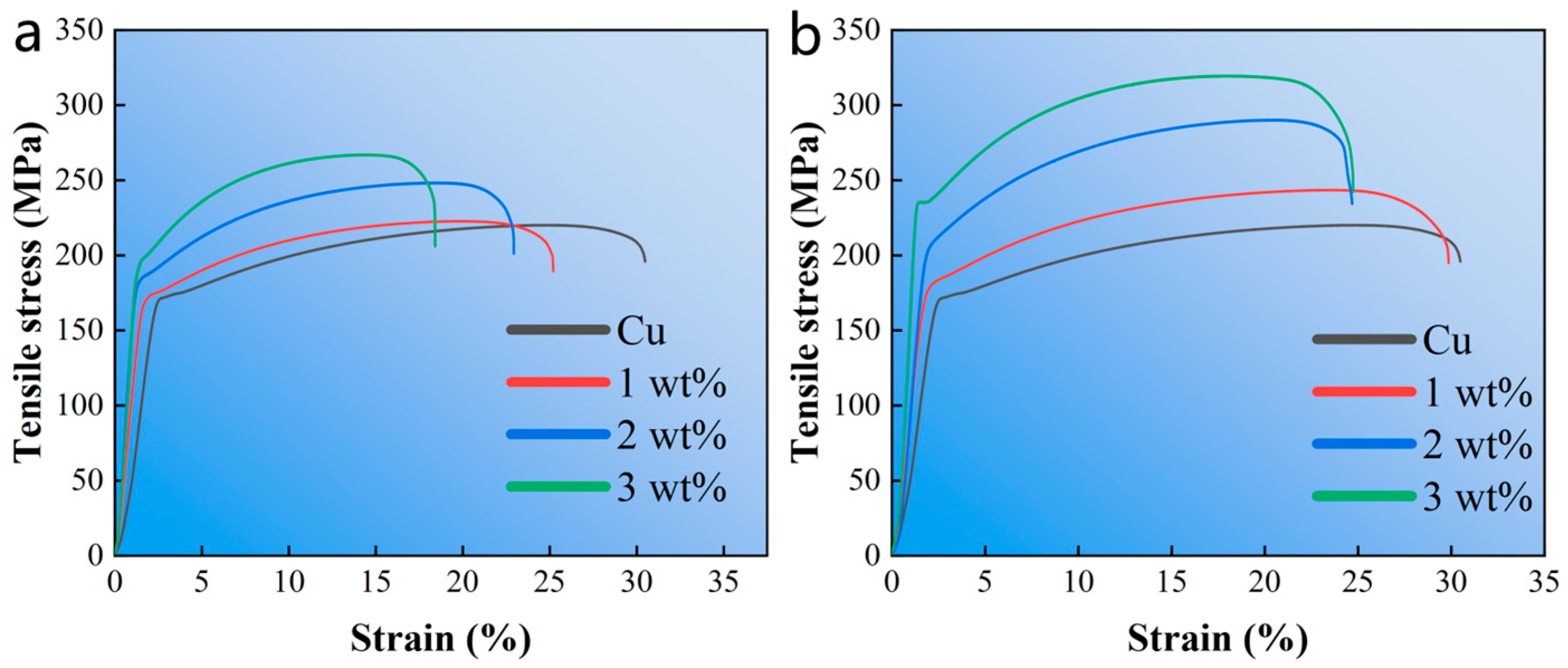
Figure 6 shows the engineering stress–strain curve of the composite material. With the increase of alumina content, the tensile strength of Cu composites has a slight increasing trend. However, the elongation of the composites is greatly reduced due to the weak interfacial bonding between alumina and Cu, and the presence of voids reduces the ability of alumina to withstand load. In addition, with the increase of silver-plated alumina content, the ultimate tensile strength of the composite material is greatly improved, which is due to the effect of alumina on refining Cu grains. At the same time, the elongation decreases slowly, and the elongation of Cu can be mostly retained. Therefore, alumina silver plating can not only improve the interface bonding, retain the physical properties of Cu, but also greatly improve the mechanical properties of the composites.
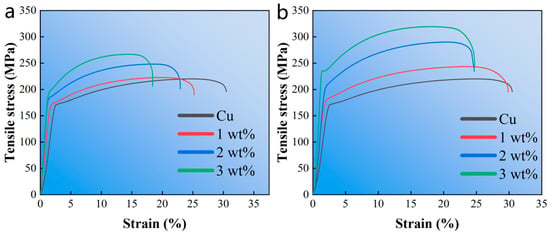
Figure 6.
The engineering stress–strain curves of specimens. (a) Al2O3/Cu composites with different Al2O3 content; (b) Al2O3@Ag/Cu composites with different Al2O3@Ag content.
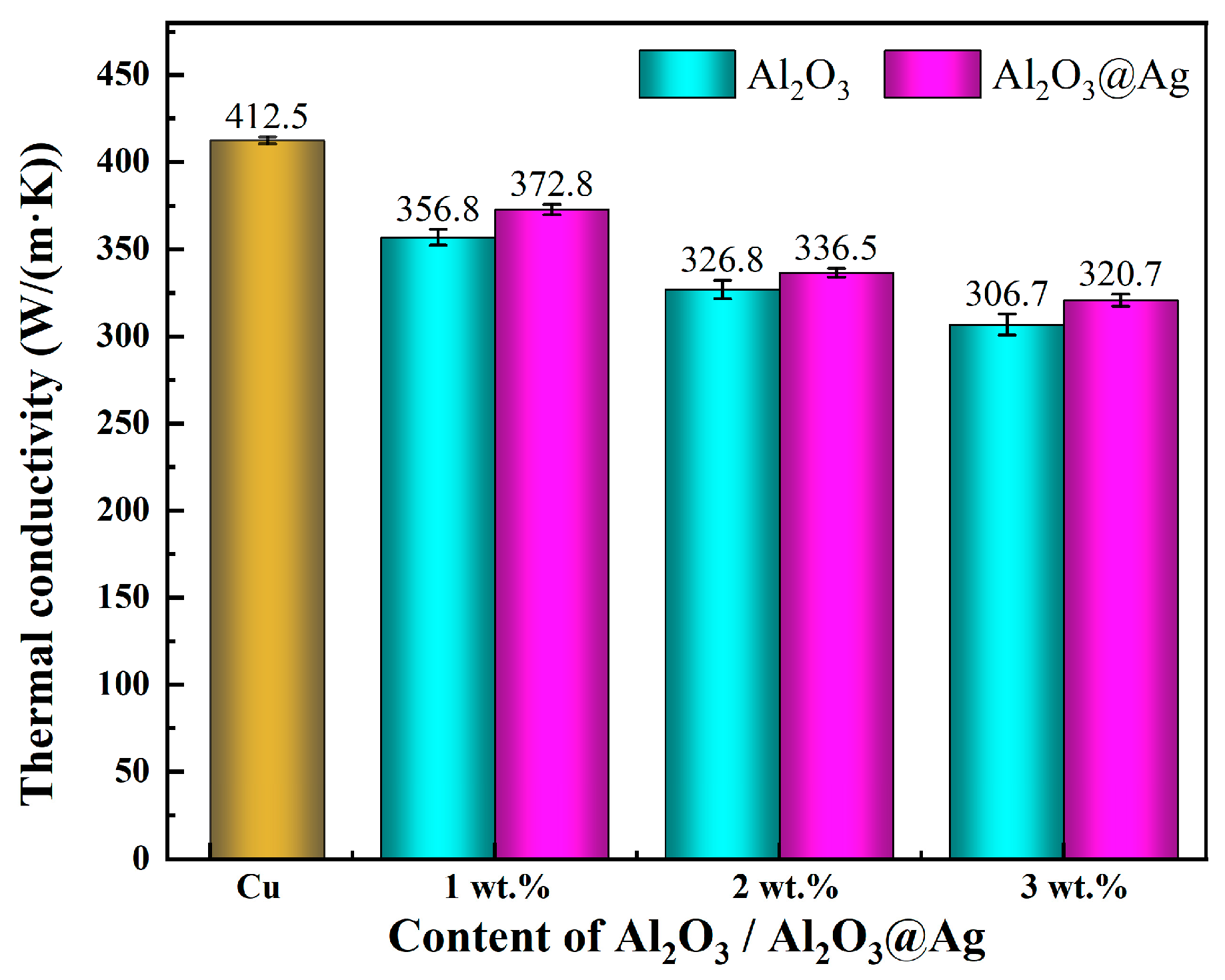
Figure 7 shows the statistical chart of thermal conductivity of Al2O3@Ag/Cu and Al2O3/Cu composite materials with varying alumina content. From the graph, the following observations can be made: (1) With increasing alumina content, the thermal conductivity of both composite materials decreases. This is because, on one hand, as the alumina content increases, the relative density of the composite material gradually decreases, resulting in small pores between the alumina and Cu matrix, creating thermal insulation barriers that increase the interfacial thermal resistance between the alumina and Cu. On the other hand, the introduction of alumina leads to the formation of more bonding interfaces within the composite material, which further increases the interfacial thermal resistance and results in a decrease in thermal conductivity. (2) Under the same alumina content, the thermal conductivity of the Al2O3@Ag/Cu composite material is significantly higher than that of the Al2O3/Cu composite material. When the alumina content reaches 3%, the thermal conductivity of the Al2O3/Cu composite material is 306.7 W/(m∙K), which is only 74.4% of pure Cu, while the thermal conductivity of the Al2O3@Ag/Cu composite material is 320.7 W/(m∙K), reaching 77.8% of pure Cu. This phenomenon can be attributed to the silver-plating treatment on the surface of the alumina [30]. The addition of Ag as an impurity increases the number of free electrons, enhances the conduction path of free electrons, and strengthens phonon scattering, thereby increasing the electrical and thermal conductivity. Additionally, the inclusion of Ag improves the densification of the composite material, which has a positive effect on phonon conduction.
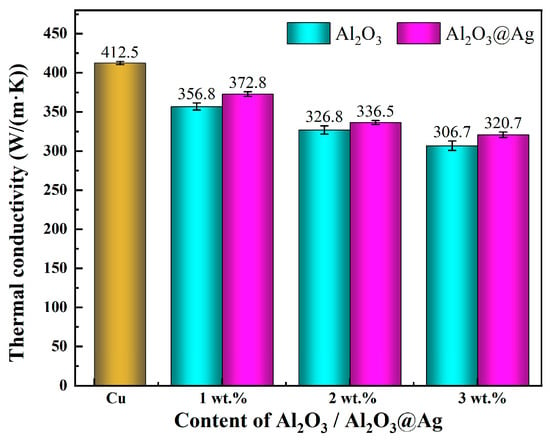
Figure 7.
Thermal conductivity of composites with different alumina content.
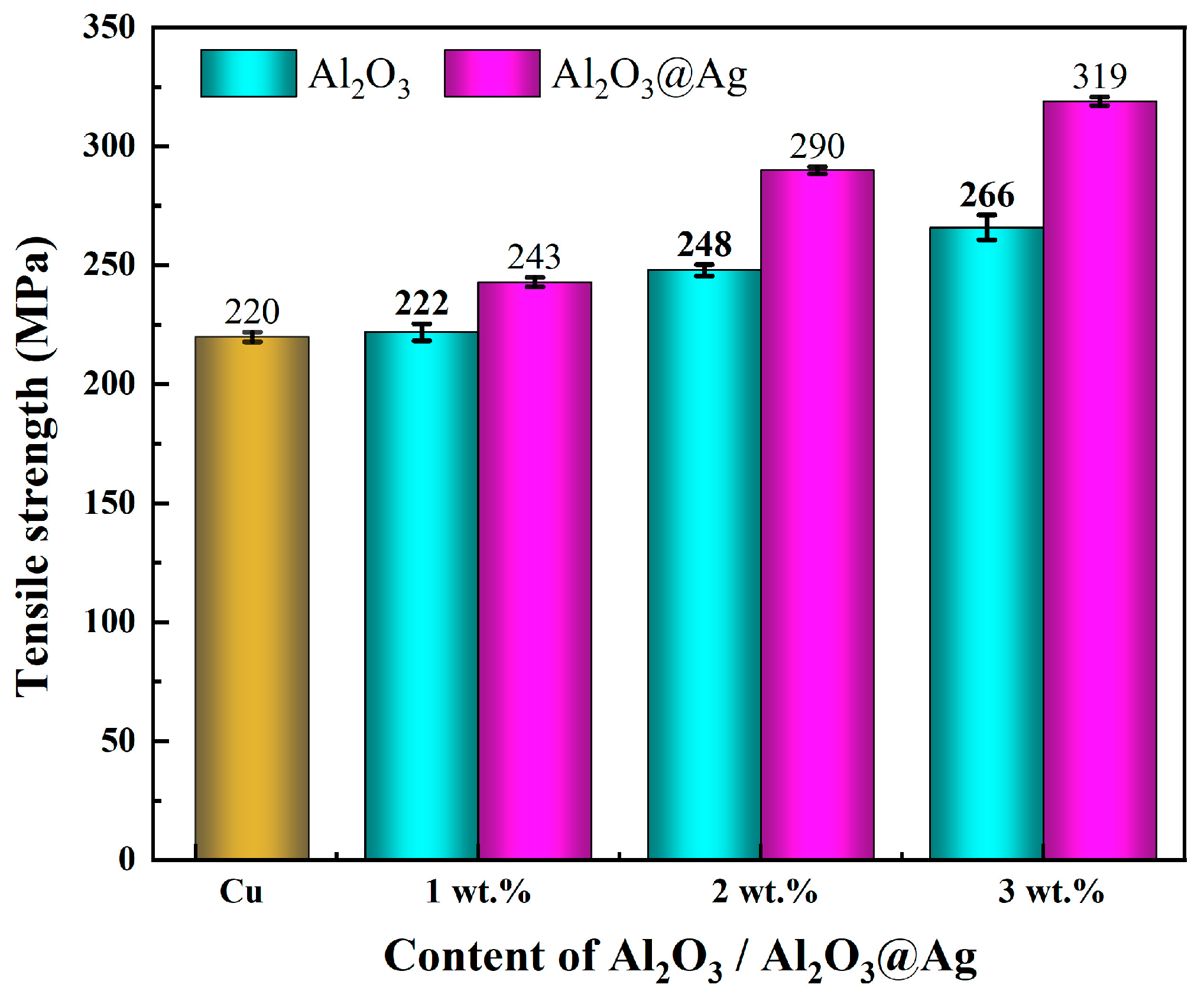
Figure 8 shows the tensile strength of Al2O3/Cu and Al2O3@Ag/Cu composite materials with different alumina contents. When the alumina content reaches 3 wt.%, the tensile strength of the untreated composite material is 266.35 MPa, which is approximately 21.1% higher than that of the Cu matrix. The silver-plated composite materials exhibit even higher tensile strength. When the alumina content reaches 3 wt.%, the tensile strength of the Al2O3@Ag/Cu composite material is 318.56 MPa, which is approximately 19.6% higher than the untreated material and approximately 53.9% higher than the Cu matrix.
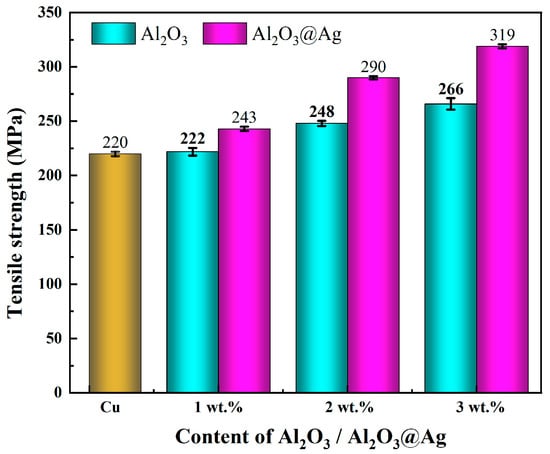
Figure 8.
The tensile strength of Al2O3/Cu and Al2O3@Ag/Cu composites were obtained.
When alumina is added to the Cu matrix, thermal mismatch occurs easily during sintering due to the different coefficients of thermal expansion, causing large dislocation accumulations at the interface and affecting dislocation movement. At the same time, the non-shearable alumina particles penetrate and anchor in the Cu matrix, which effectively blocks dislocation movement and results in a higher tensile strength for the composite material [31]. For Al2O3@Ag/Cu composite material, strong bonding can be formed between Ag and Cu at the interface, which has a positive effect on the physical and mechanical properties of the composite material and leads to better tensile strength performance. Moreover, because the deposition rates of Cu and silver are different during sintering, segregation can occur at the eutectic phase, which helps improve the mechanical strength.
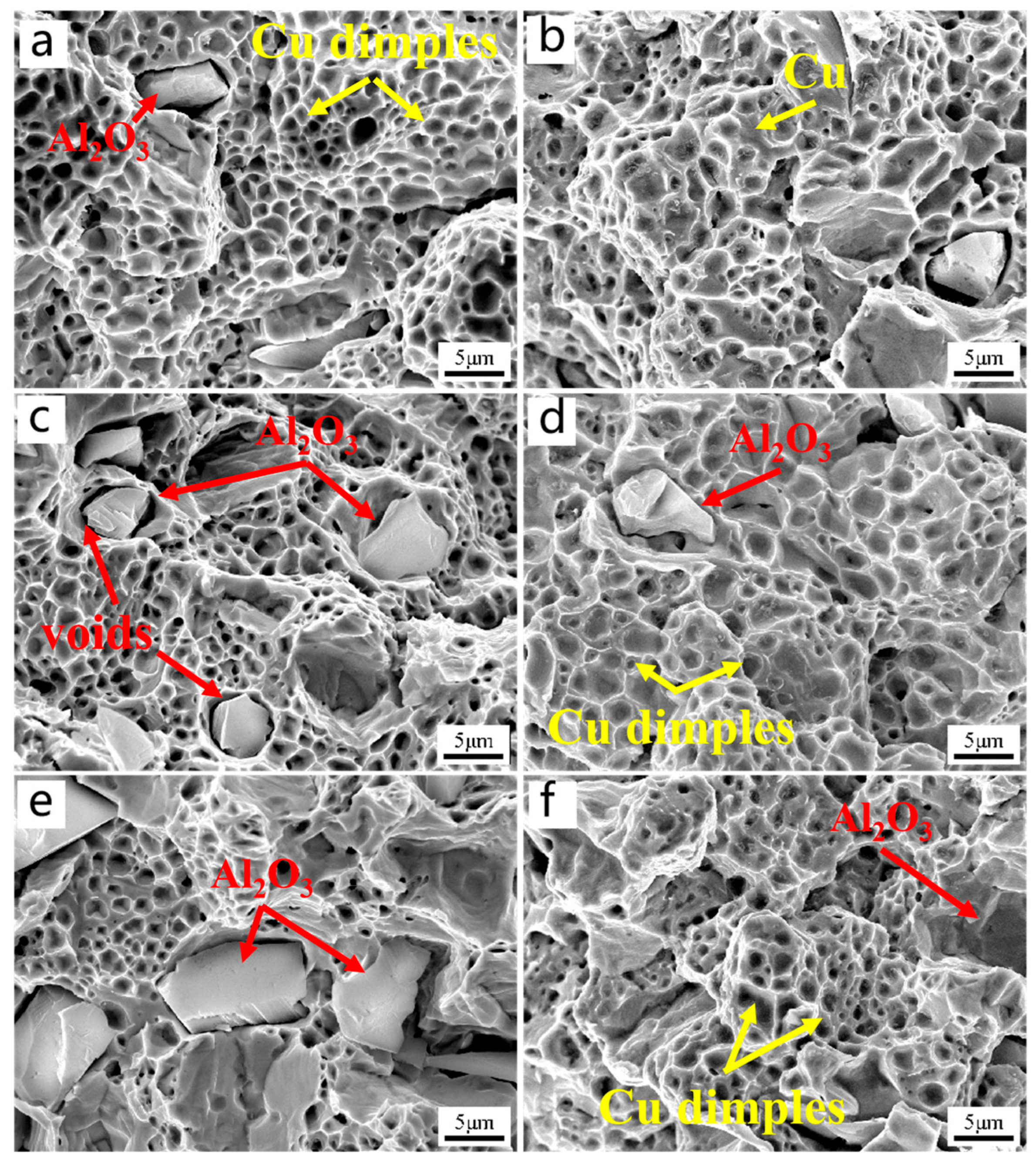
To further investigate the strengthening mechanism of the two types of composite materials, the microstructure of the tensile fracture surface was observed. Figure 9a,c,e shows the microstructures of the tensile fracture surfaces of Al2O3/Cu composite materials with different alumina contents. It can be seen that the fracture surfaces are mainly ductile fracture surfaces, indicating that the fracture mode is mainly ductile fracture. The alumina particles are either completely embedded in the Cu matrix or completely pulled out. The ductile dimples around the alumina particles are relatively flat, which is because the interface binding between the two materials is weak, and the load transmission is subject to less surrounding dislocation bending. Figure 9b,d,f shows the microstructures of the tensile fracture surfaces of Al2O3@Ag/Cu composite materials, which also exhibit ductile fracture.
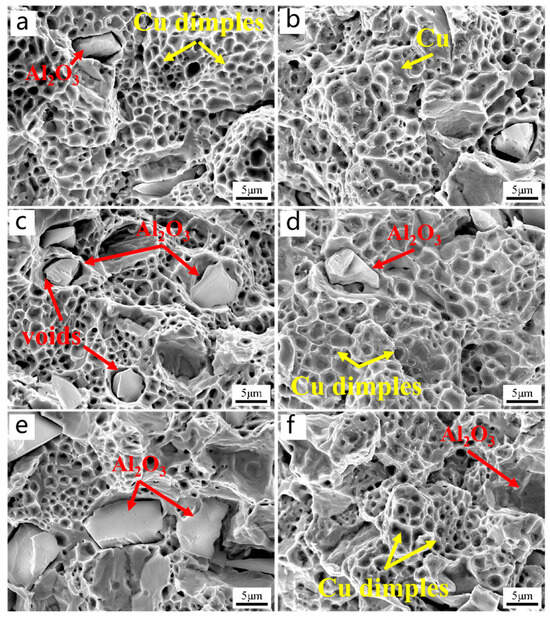
Figure 9.
The tensile fracture morphology of Al2O3/Cu and Al2O3@Ag/Cu composites. (a,c,e) Al2O3 content were 1 wt.%, 2 wt.%, 3 wt.%; (b,d,f) the contents of Al2O3@Ag were 1 wt.%, 2 wt.%, 3 wt.%.
3.3. Tribological Behavior
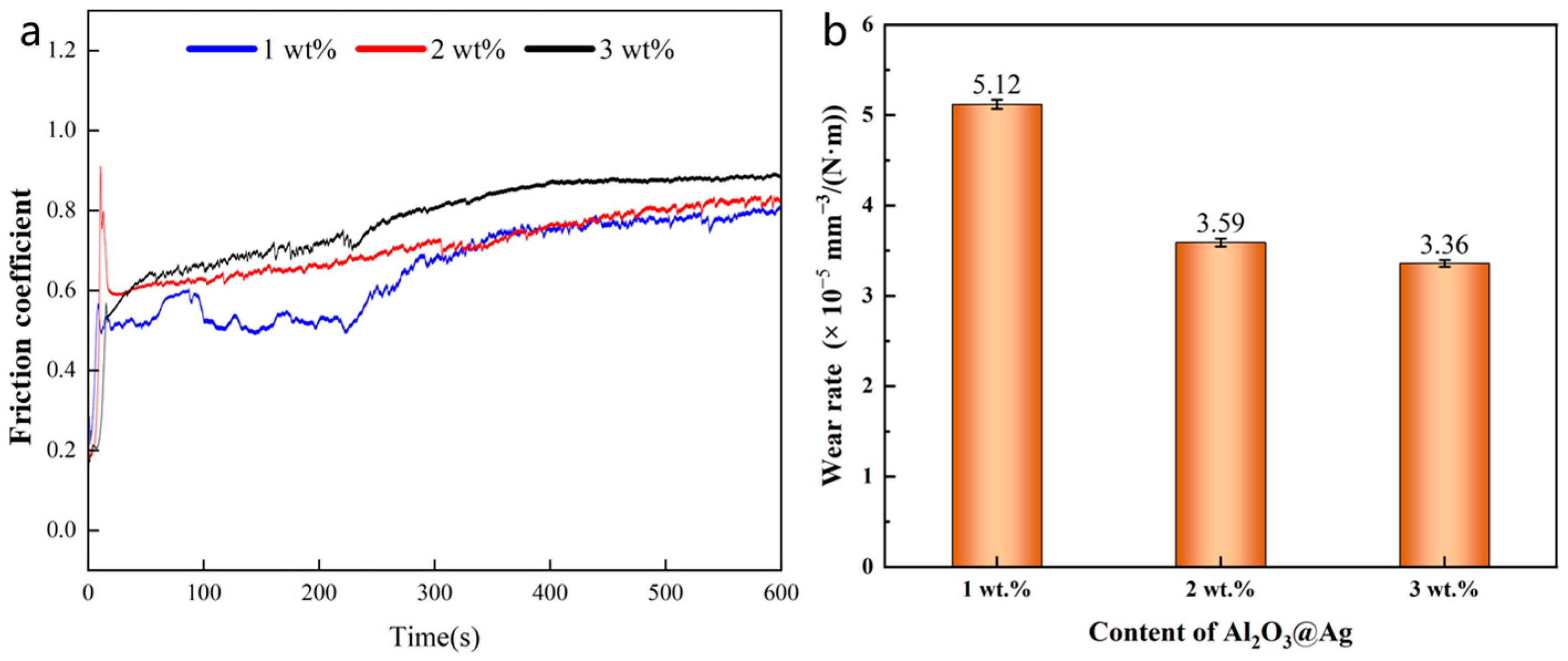
The friction coefficient–time curve and wear rate of the composite material are shown in Figure 10a. From the Figure, it can be seen that as the alumina content increases, the friction coefficient of the composite material also increases, indicating that the addition of alumina can increase the friction force of the material to some extent. At the same time, from Figure 10a, it can be observed that the friction coefficient changes in three stages over time. In the first stage, the friction coefficient rapidly increases but lasts for a short duration. During this stage, the composite material surface experiences the combined effect of horizontal shear force and vertical load, resulting in plastic deformation and the formation of wear marks. In the second stage, the friction coefficient begins to fluctuate with small-scale decreases. This is because as the width of wear marks increases, the surface tends to become smoother. The third stage is the stable wear stage, where the friction coefficient stabilizes.
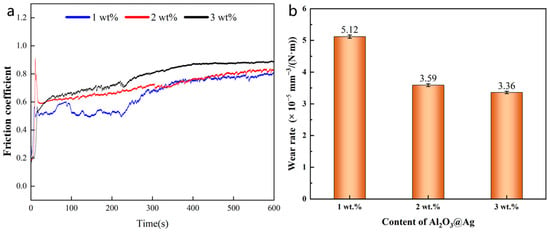
Figure 10.
(a) Friction coefficient and (b) wear rate of Al2O3@Ag/Cu composites with different Al2O3@Ag content.
Figure 10b shows the wear rate of Al2O3@Ag/Cu composite materials with different alumina content. The wear volume of the composite material was measured by integrating the cross-sectional areas of three scratches using laser confocal microscopy and calculating their arithmetic mean. From the figure, it can be observed that the wear rate decreases as the Al2O3@Ag content increases. When the Al2O3@Ag content is 3%, the wear rate is only 3.36 × 10−5 mm3/(N∙m), which is approximately an 84.4% reduction compared to pure Cu. This indicates that the addition of alumina can significantly improve the wear resistance of the Cu composite material [32]. This is because the addition of alumina increases the hardness of the composite material, enhancing its ability to resist plastic deformation, thereby reducing the contact area between abrasive balls and the worn surface, and increasing its resistance to scratches and wear. Additionally, the chemical silver-plating process helps to uniformly disperse alumina particles in the Cu matrix and enhances the interface bonding between alumina particles and the Cu matrix, positively affecting the wear resistance of the composite material.
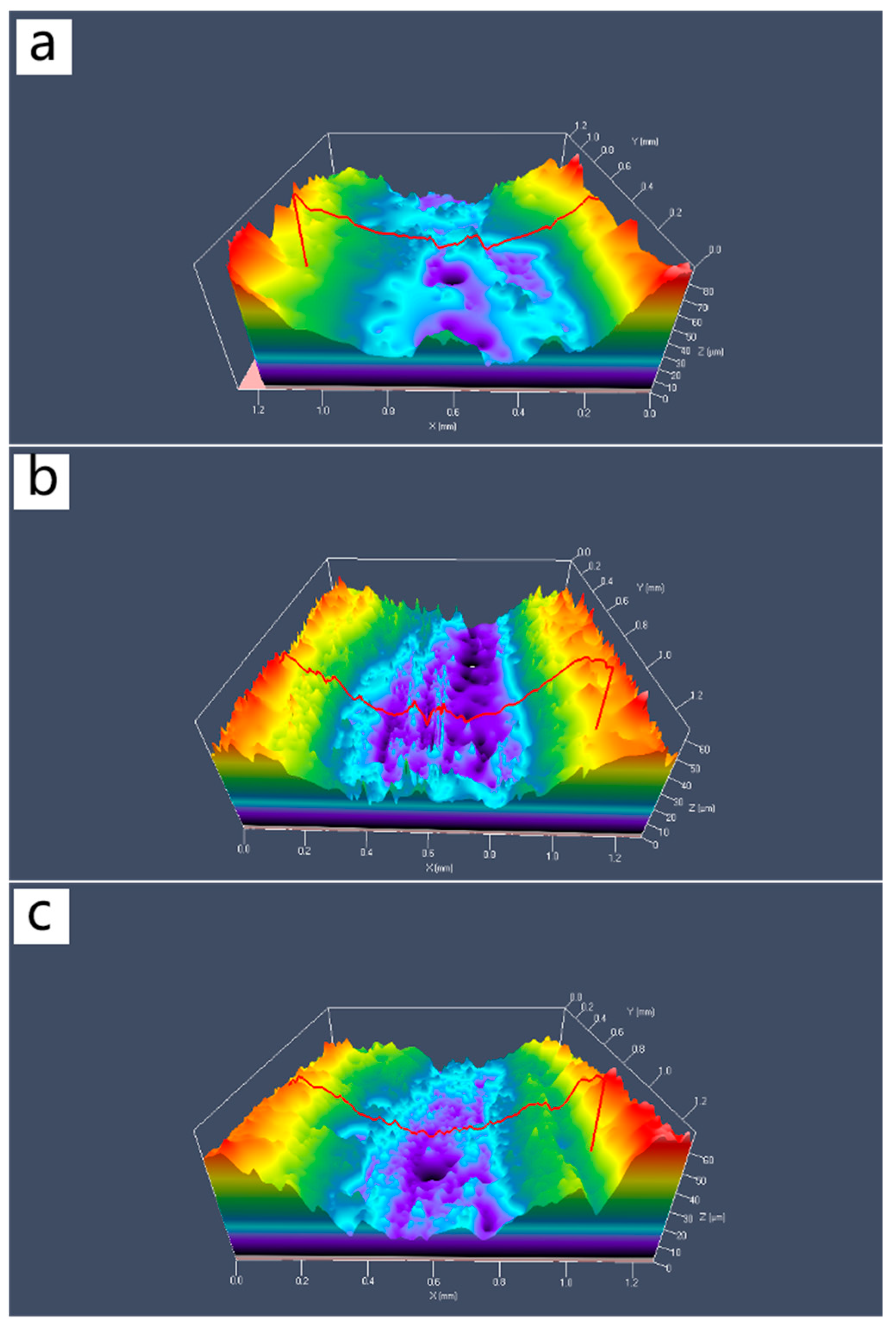
Figure 11 shows the three-dimensional contour maps of the wear tracks of composite materials with different Al2O3@Ag content. From the Figure 11, it can be seen that as the Al2O3@Ag content increases, the wear track depth gradually decreases, and the width tends to narrow. This indicates that the amount of wear of the composite material gradually decreases, which is consistent with the above experimental results.
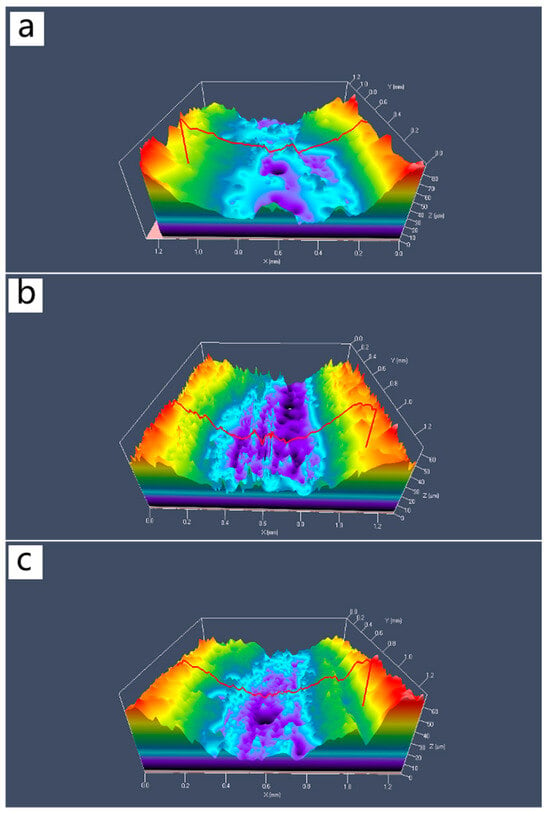
Figure 11.
Three-dimensional wear morphology of Al2O3@Ag/Cu composites. The contents of Al2O3@Ag are (a) 1 wt.%, (b) 2 wt.%, and (c) 3 wt.%.
To further investigate the effect of Al2O3@Ag content on the wear morphology of composite materials, the wear morphology of the composite materials was observed. The wear morphology of the Al2O3@Ag/Cu composite material is shown in Figure 11, where furrows, peeling layers, and cracks can be observed on the worn surface. These defects are produced by the combined action of transverse shear force and normal load during the friction process, resulting in deformation of the friction surface, exposing the alumina particles, and further compacting them into the Cu matrix, gradually increasing the friction coefficient of the composite material. Meanwhile, these particles, as load-bearing components, can effectively protect the soft matrix from damage during the friction process, reducing the wear amount of the composite material with an increase of alumina content.
As shown in Figure 12a, when the alumina content is 1 wt.%, the main feature of the worn surface is deep furrows and a small number of spalling pits. This is because when the alumina content is low, alumina cannot provide sufficient support, and a large amount of Cu falls off along the friction direction. At the same time, under the action of normal load, plastic deformation occurs on the surface of the composite material, forming furrow-shaped wear. At this time, the primary wear mechanism is furrowing wear. As the alumina content gradually increases, reaching 2 wt.%, as shown in Figure 12b, the surface furrows become shallower, and spalling pits appear on the worn surface. This is because the supporting role of alumina in the composite material gradually appears, the surface deformation decreases, and the material peels off along the defect at the interface. Currently, the primary wear mechanism is spalling wear. When the alumina content reaches 3 wt.%, as shown in Figure 12c, a large amount of debris can be observed on the surface. This is because ceramic particles can be divided into two processes during friction: One is that alumina particles are embedded into the composite material under the action of normal load and undergo micro-cutting of the soft matrix on the surface of the composite material under the action of transverse shear force [33]. In addition, alumina particles are exposed on the surface, which hinders the micro-cutting of the abrasive and reduces the wear rate of the composite material. Consequently, the wear mechanism is debris wear and spalling wear.

Figure 12.
The wear morphology of Al2O3@Ag/Cu composites. Al2O3@Ag content is (a) 1 wt.%; (b) 2 wt.%; (c) 3 wt.%.
4. Conclusions
In this study, Ag-coated alumina-reinforced Cu composites were successfully prepared by rapid hot-press sintering. The presence of a Ag layer not only enhances the interface bonding between the alumina and Cu matrix, but also improves the mechanical properties of the composites. Based on greatly retaining the physical properties of Cu, the comprehensive performance is improved. Based on the findings, the main conclusions are as follows.
- Comparing the morphology of the electroless Ag coating at different temperatures, a thin and uniform Ag coating (230 nm) was obtained at 25 °C. It fills the gap between the interface of alumina and Cu, and greatly improves the interface bonding force. It provides some insights for electroless plating Ag on the surface of ceramic particles.
- With the increase of Al2O3 and Al2O3@Ag content, the microhardness and tensile strength of Cu composites gradually increase, while the conductivity and thermal conductivity gradually decrease. The mechanical properties of Al2O3/Cu composites did not increase significantly, and the physical properties decreased significantly. The Al2O3@Ag/Cu composite (Al2O3@Ag = 3 wt.%) retains about 80% conductivity and 83% elongation of Cu, while the ultimate tensile strength reaches 318.6 MPa (145% of Cu). The composite exhibits a relatively balanced combination of hardness, strength, plasticity, and thermal conductivity.
- The addition of hard abrasive Al2O3 can improve the wear resistance of Cu composites. With the increase of Al2O3@Ag content, the uniform distribution of alumina in the Cu matrix, the continuity in the Cu matrix gradually increased, the friction coefficient of the composite gradually stabilized, and the wear rate decreased to 3.36 ×10−5 mm3/(N·m). The wear rate is only 25% of Cu. The main wear mechanism is abrasive wear.
The research results will provide a new strategy for the surface treatment of ceramic particles and the design of high-strength, high-conductivity, and thermally conductive copper matrix composites.
Author Contributions
Conceptualization, H.Y. and H.Z.; methodology, K.L.; validation, Y.C.; investigation, X.Z., K.L. and P.D.; data curation, P.D. and Y.C.; writing—original draft preparation, X.Z.; writing—review and editing, H.Y.; supervision, H.Z.; funding acquisition, H.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This work is supported by Jilin Provincial Scientific and Technological Development Program (No. YDZJ202301ZYTS493).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ho, V.; Ladani, L.; Razmi, J.; Gruber, S.; Murphy, A.B.; Chen, C.; East, D.; Lopez, E. Powder Bed Fabrication of Copper: A Comprehensive Literature Review. Metals 2025, 15, 1114. [Google Scholar] [CrossRef]
- Meng, F.; Zhou, Y.; Zhang, H.; Wang, Z.; Liu, D.; Cao, S.; Cui, X.; Nong, Z.; Man, T.; Liu, T. Development and Research Status of Wear-Resistant Coatings on Copper and Its Alloys: Review. Crystals 2025, 15, 204. [Google Scholar] [CrossRef]
- Shu, D.; Li, X.; Yang, Q. Effect on Microstructure and Performance of B4C Content in B4C/Cu Composite. Metals 2021, 11, 1250. [Google Scholar] [CrossRef]
- Liu, H.; Teng, X.; Wu, W.; Wu, X.; Leng, J.; Geng, H. Effect of graphene addition on properties of Cu-based composites for electrical contacts. Mater. Res. Express. 2017, 4, 066506. [Google Scholar] [CrossRef]
- Pan, Y.; Lu, X.; Volinsky, A.A.; Liu, B.; Xiao, S.; Zhou, C.; Li, Y.; Chen, M.; Qu, X. Tribological and mechanical properties of copper matrix composites reinforced with carbon nanotube and alumina nanoparticles. Mater. Res. Express. 2019, 6, 116524. [Google Scholar] [CrossRef]
- Cao, H.; Tan, Z.; Fan, G.; Guo, Q.; Su, Y.; Li, Z.; Xiong, D.-B. Wide and fine alignment control and interface modification for high-performance thermally conductive graphite/copper composite. Compos. Part B-Eng. 2020, 191, 107965. [Google Scholar] [CrossRef]
- Ordoñez, M.F.C.; Farias, M.C.M.; Descartes, S.; Machado, I.F.; Souza, R.M. Tribofilm formation during dry sliding of graphite- and MoS2- based composites obtained by spark plasma sintering. Tribol. Int. 2021, 160, 107035. [Google Scholar] [CrossRef]
- Wang, X.; Liu, K.; Su, Y.; Wang, X.; Cao, H.; Hua, A.; Ouyang, Q.; Zhang, D. Synergistic enhancing effect of tungsten-copper coated graphite flakes and aluminum nitride nanoparticles on microstructure, mechanical and thermal properties of copper matrix composites. Mater. Sci. Eng. A 2022, 857, 143987. [Google Scholar] [CrossRef]
- Malaki, M.; Fadaei Tehrani, A.; Niroumand, B.; Gupta, M. Wettability in Metal Matrix Composites. Metals 2021, 11, 1034. [Google Scholar] [CrossRef]
- Korać, M.; Kamberović, Ž.; Anđić, Z.; Stopić, S. Advances in Thermochemical Synthesis and Characterization of the Prepared Copper/Alumina Nanocomposites. Metals 2020, 10, 719. [Google Scholar] [CrossRef]
- Abu-Oqail, A.; Wagih, A.; Fathy, A.; Elkady, O.; Kabeel, A.M. Effect of high energy ball milling on strengthening of Cu-ZrO2 nanocomposites. Ceram. Int. 2019, 45, 5866–5875. [Google Scholar] [CrossRef]
- Şenel, M.C.; Gürbüz, M.; Koç, E. Fabrication and characterization of synergistic Al-SiC-GNPs hybrid composites. Compos. Part B-Eng. 2018, 154, 1–9. [Google Scholar] [CrossRef]
- Wallstabe, R. Laser-Assisted Surface Modification of Alumina and Its Tribological Behavior. J. Mate. Eng. Perform. 2013, 22, 223–235. [Google Scholar] [CrossRef]
- Konopka, K. Alumina Composites with Metal Particles in Ceramic Matrix. Powder Metall. Met. Ceram. 2015, 54, 374–379. [Google Scholar] [CrossRef]
- Phuong, D.D.; Van Trinh, P.; Minh, P.N.; Shtertser, A.A.; Ulianitsky, V.Y. Effect of Powder Preparation Techniques on Microstructure, Mechanical Properties, and Wear Behaviors of Graphene-Reinforced Copper Matrix Composites. Crystals 2024, 14, 1000. [Google Scholar] [CrossRef]
- Su, Y.; Zhang, Y.; Song, J.; Hu, L. Tribological behavior and lubrication mechanism of self-lubricating ceramic/metal composites: The effect of matrix type on the friction and wear properties. Wear 2017, 372, 130–138. [Google Scholar] [CrossRef]
- Rajkovic, V.; Bozic, D.; Stasic, J.; Wang, H.; Jovanovic, M.T. Processing, characterization and properties of copper-based composites strengthened by low amount of alumina particles. Powder Technol. 2014, 268, 392–400. [Google Scholar] [CrossRef]
- Feng, T.; Zheng, W.; Chen, W.; Shi, Y.; Fu, Y.Q. Enhanced interfacial wettability and mechanical properties of Ni@Al2O3/Cu ceramic matrix composites using spark plasma sintering of Ni coated Al2O3 powders. Vacuum 2021, 184, 109938. [Google Scholar] [CrossRef]
- Shang, F.; Wang, W.; Yang, T.; Liu, R.; Zhou, J. Interaction Mechanism and Wear Resistance of Ni- encapsulated Al2O3 Particles Reinforced Iron Matrix Composites. Rare Metal Mat. Eng. 2022, 51, 422–428. [Google Scholar]
- Güler, O.; Varol, T.; Alver, Ü.; Canakci, A. Effect of Al2O3 content and milling time on the properties of silver coated Cu matrix composites fabricated by electroless plating and hot pressing. Mater. Today Commun. 2020, 24, 101153. [Google Scholar] [CrossRef]
- Moustafa, S.F.; Abdel-Hamid, Z.; Abd-Elhay, A.M. Copper matrix SiC and Al2O3 particulate composites by powder metallurgy technique. Mater. Lett. 2002, 53, 244–249. [Google Scholar] [CrossRef]
- Su, C.-Y.; Huang, J.-L.; Chen, P.-C.; Yu, H.-J.; Ma, D.-L.; Yu, B.-Y. A Low-Temperature Alumina/Copper Diffusion Bonding Process using La-Doped Titanium Interlayers. Coatings 2018, 8, 401. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, Y.; Cao, Z.; Yan, C.; Wang, K.; Chen, J.; Cheng, S.; Feng, Z. A facile process to manufacture high performance copper layer on ceramic material via biomimetic modification and electroless plating. Compos. Part B-Eng. 2019, 157, 123–130. [Google Scholar] [CrossRef]
- Shehata, F.; Fathy, A.; Abdelhameed, M.; Moustafa, S.F. Preparation and properties of Al2O3 nanoparticle reinforced copper matrix composites by in situ processing. Mater. Design. 2009, 30, 2756–2762. [Google Scholar] [CrossRef]
- Radhika, N.; Subramanian, R.; Prasat, S. Tribological Behaviour of Aluminium/Alumina/Graphite Hybrid Metal Matrix Composite Using Taguchi’s Techniques. J. Min. Mater. Charact. Eng. 2011, 10, 427–443. [Google Scholar] [CrossRef]
- Sedighi, M.; Vini, M.H.; Farhadipour, P. Effect of Alumina Content on the Mechanical Properties of AA5083/Al2O3 Composites Fabricated by Warm Accumulative Roll Bonding. Powder Metall Met. Ceram. 2016, 55, 413–418. [Google Scholar] [CrossRef]
- Vogel, T.; Ma, S.; Liu, Y.; Guo, Q.; Zhang, D. Impact of alumina content and morphology on the mechanical properties of bulk nanolaminated Al2O3-Al composites. Compos. Commun. 2020, 22, 100462. [Google Scholar] [CrossRef]
- Chen, S.; Yu, H.; An, J.; Shang, Z.; Wang, Z.; Ding, W. Regulating the GP Zone to T′ Phase Evolution and Achieving Strength–Ductility Synergy in an Al-Mg-Zn-Cu Alloy via a Two-Step Aging (T4P-BH) Process. Metals 2025, 15, 1347. [Google Scholar] [CrossRef]
- Sobhani, M.; Mirhabibi, A.; Arabi, H.; Brydsonr, M.D. Effects of in situ formation of TiB2 particles on age hardening behavior of Cu–1 wt% Ti–1 wt% TiB2. Mater. Sci. Eng. A 2013, 577, 16–22. [Google Scholar] [CrossRef]
- Zuo, C.H.; Xu, L.; Sun, Y.F.; Xia, H.P.; Tang, Z.M.; Zheng, G.; Han, Z.H. Effect of interface bonding and properties of graphite/copper composites by microwave pressure sintering. J. Alloys Compd. 2024, 976, 173009. [Google Scholar] [CrossRef]
- Jung, T.K.; Lim, S.C.; Kwon, H.C.; Kim, M.S. Fabrication and properties of TiB2 reinforced Cu composites by electromagnetic stirring. Mater. Sci. Forum 2004, 449–452, 297–300. [Google Scholar] [CrossRef]
- Zhang, S.R.; Kang, H.J.; Li, R.G.; Zou, C.L.; Guo, E.Y.; Chen, Z.N.; Wang, T.M. Microstructure evolution, electrical conductivity and mechanical properties of dual-scale Cu5Zr/ZrB2 particulate reinforced copper matrix composites. Mater. Sci. Eng. A 2019, 762, 138108. [Google Scholar] [CrossRef]
- Ye, H.; Zhu, W.; Zou, H.; Han, Y.; Ran, X. Preparation, microstructure and properties of titanium-aluminum intermetallic compound reinforced copper matrix composites. Mater. Charact. 2025, 229, 115519. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.