Patient-to-patient variability in response to drugs creates a significant challenge for the safe and effective treatment of many human diseases. Pharmacogenomics seeks to address this challenge by linking drug response to patient genotypes at important loci in order to better customize patient treatments. While many diverse ‘pharmacogenes’ are recognized (e.g., TPMT, G6PD, HLA, etc.), a thorough understanding of the implications of genetic variation in the CYP superfamily, especially, provides a golden opportunity to practice precision medicine and improve therapeutic outcomes. Rare variation in the steroidogenic cytochrome P450 (CYP) enzymes has long been associated with some inborn errors of metabolism [1]. However, it is an appreciation of the consequences of more commonly occurring variation in the drug-metabolizing CYPs—especially the large CYP2 gene family—that has been a driver in the development of contemporary pharmacogenomics.
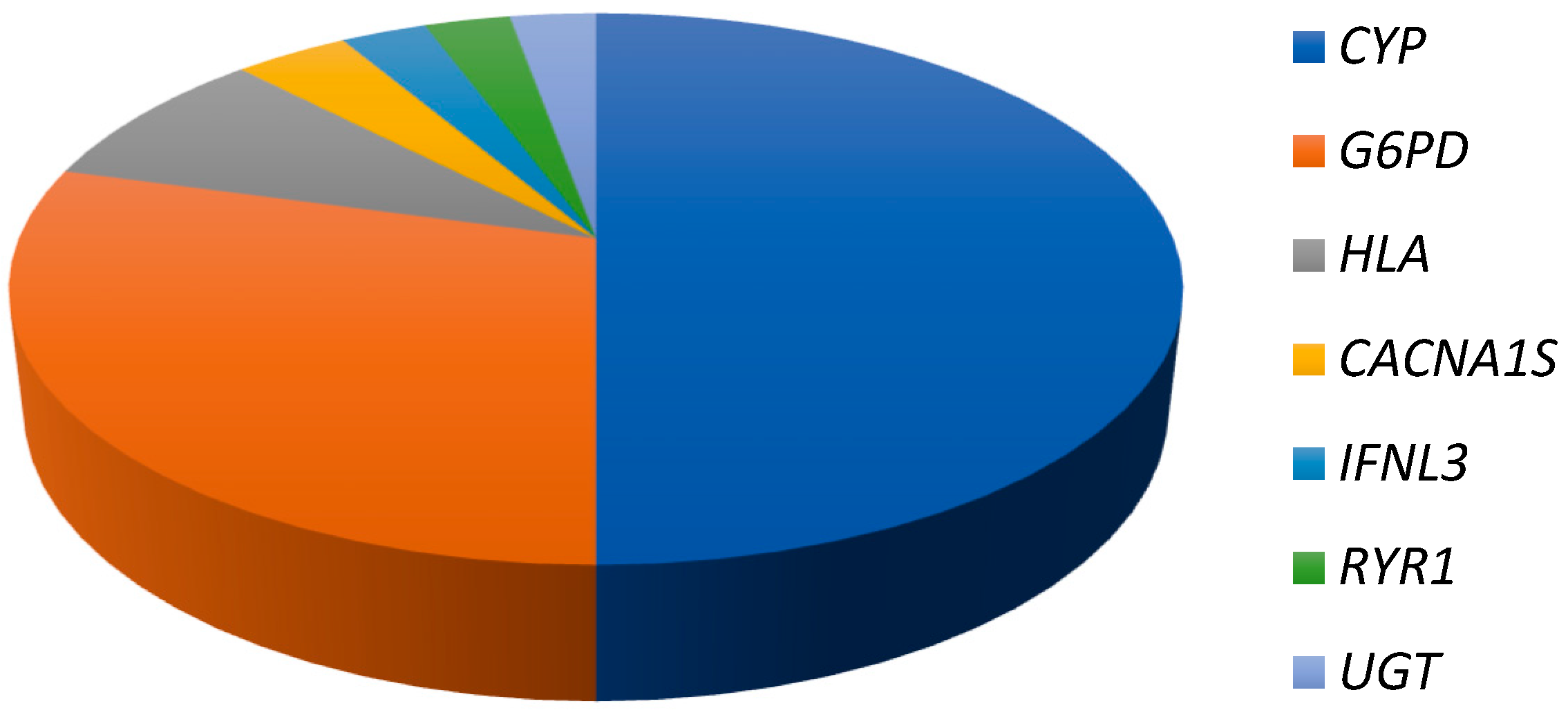
The importance of CYP-based pharmacogenomics can be gleaned from Figure 1 below, which depicts the relative percentages of the most frequently identified genes/gene families on the Clinical Pharmacogenetics and Implementation Consortium (CPIC) prioritized drug–gene pairs list (https://cpicpgx.org/genes-drugs/). CPIC identifies Level A or B drug–gene pairs as those where prescribing action based on pharmacogenetic information is recommended. Of the 153 Level A or B gene pairs (accessed February 2018), there were 106 instances where the gene features in an interaction with more than two prescription drugs. Within this subset, CYP genes constituted 50% of the entries. The next most important gene/gene family is G6PD, followed distantly by HLA genes.
Figure 1.
Importance of CYP pharmacogenomics. The pie chart depicts the percentage that each set of pharmacogenes contributes in priority drug–gene pair interactions with two or more drugs.
Worldwide, the distribution of CYP alleles varies substantially between populations, with attendant consequences for global health [2]. CYP variation, and its functional effects on enzyme activity (where known), is cataloged at the PharmVar web-site (https://www.pharmvar.org/). This repository continues to grow and to be a valuable resource to inform clinical decision-making [3].
Against this backdrop, it felt particularly timely to devote a Special Issue of the Journal of Personalized Medicine to the topic of ‘Cytochrome P450 Variation in Pharmacogenomics’. This issue, which comprises eight original articles, begins by reviewing the fundamental biochemistry, genetics and clinical importance of CYP2A6 [4], CYP2C9 [5] CYP2C19 [6] and CYP2D6 [7]. This is followed with an article on translational applications of CYP pharmacogenomics in guiding tamoxifen and warfarin therapy in Canada [8] and two papers on CYP pharmacogenomics in special populations [9,10]. The issue closes with a commentary on the future of warfarin pharmacogenomics, long heralded as the ‘poster-child’ of pharmacogenomics [11].
We hope this collection of papers will prove a useful introduction to CYP pharmacogenetics at large, provide an update on contemporary translational efforts in the clinic and stimulate further basic research in the field.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pikuleva, I.A.; Waterman, M.R. Cytochromes p450: Roles in diseases. J. Biol. Chem. 2013, 288, 17091–17098. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ingelman-Sundberg, M.; Lauschke, V.M. Worldwide distribution of cytochrome P450 alleles: A meta-analysis of population-scale sequencing projects. Clin. Pharmacol. Ther. 2017, 102, 688–700. [Google Scholar] [CrossRef] [PubMed]
- Gaedigk, A.; Ingelman-Sundberg, M.; Miller, N.A.; Leeder, J.S.; Whirl-Carrillo, M.; Klein, T.E.; PharmVar Steering Committee. The Pharmacogene Variation (PharmVar) Consortium: Incorporation of the human cytochrome P450 (CYP) allele nomenclature database. Clin. Pharmacol. Ther. 2018, 103, 399–401. [Google Scholar] [CrossRef] [PubMed]
- Tanner, J.-A.; Tyndale, R.F. Variation in CYP2A6 Activity and personalized medicine. J. Pers. Med. 2017, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Daly, A.K.; Rettie, A.E.; Fowler, D.M.; Miners, J.O. Pharmacogenomics of CYP2C9: Functional and clinical considerations. J. Pers. Med. 2018, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.-A.; Pereira, N. Pharmacogenomic Impact of CYP2C19 Variation on clopidogrel therapy in precision cardiovascular medicine. J. Pers. Med. 2018, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Gaedigk, A.; Dinh, J.C.; Jeong, H.; Prasad, B.; Leeder, J.S. Ten years’ experience with the CYP2D6 activity score: A perspective on future investigations to improve clinical predictions for precision therapeutics. J. Pers. Med. 2018, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Wigle, T.J.; Jansen, L.E.; Teft, W.A.; Kim, R.B. Pharmacogenomics guided-personalization of warfarin and tamoxifen. J. Pers. Med. 2017, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Aka, I.; Bernal, C.J.; Carroll, R.; Maxwell-Horn, A.; Oshikoya, K.A.; Van Driest, S.L. Clinical pharmacogenetics of cytochrome P450-associated drugs in children. J. Pers. Med. 2017, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Henderson, L.M.; Claw, K.G.; Woodahl, E.L.; Robinson, R.F.; Boyer, B.B.; Burke, W.; Thummel, K.E. P450 Pharmacogenetics in indigenous North American populations. J. Pers. Med. 2018, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Pirmohamed, M. Warfarin: The end or the end of one size fits all therapy? J. Pers. Med. 2018, 8, 22. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).