Abstract
A 78-year-old woman with a history of rheumatoid arthritis (treated with methotrexate) developed disturbed consciousness, emesis, and intestinal perforation. Initial labs revealed hyperammonemia (189 μg/dL) and hypertonic dehydration. Despite ammonia normalization, her neurological status improved only slightly, necessitating additional tests. Cerebrospinal fluid analysis showed no pleocytosis but positive oligoclonal bands and markedly elevated myelin basic protein (>500 pg/mL). Serum autoimmune markers were negative, including anti-aquaporin-4 (AQP4), anti-myelin oligodendrocyte glycoprotein (MOG), and anti-glial fibrillary acidic protein (GFAP) antibodies. MRI revealed T2/DWI-hyperintense lesions in the left parietal lobe and cerebellum. Crucially, extensive T2/DWI-hyperintense lesions with diffusion restriction spanned the white matter from the medulla oblongata to the lumbar spinal cord. Axial spinal DWI demonstrated diffuse hyperintensity throughout the entire white matter, accompanied by gray matter atrophy. Subsequent metabolic screening revealed low folate and hypocupremia (34 μg/dL) as well as urinary orotic acid and low serum citrulline, suggesting late-onset ornithine transcarbamylase (OTC) deficiency. Given the clinical context, this was interpreted as a metabolic breakdown rather than an established genetic diagnosis. This case is characterized by a long, diffusion-restricted lesion from the brainstem to the spinal cord that does not correspond to vascular territories. She experienced sudden death. We hypothesize that an underlying metabolic disorder, nutritional deficiencies and drug-induced neurotoxicity contributed to lesion formation.
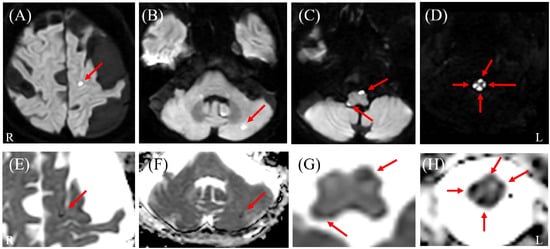
Figure 1.
Brain magnetic resonance imaging (MRI) results. Diffusion-weighted imaging (DWI) (A–D) and corresponding apparent diffusion coefficient (ADC) maps (E–H) reveal multiple scattered lesions with diffusion restriction in the cerebral hemispheres, cerebellum, and brainstem (arrows). Notably, the brainstem lesion extends longitudinally and is continuous with the spinal cord involvement. L: left; R: right.
Figure 1.
Brain magnetic resonance imaging (MRI) results. Diffusion-weighted imaging (DWI) (A–D) and corresponding apparent diffusion coefficient (ADC) maps (E–H) reveal multiple scattered lesions with diffusion restriction in the cerebral hemispheres, cerebellum, and brainstem (arrows). Notably, the brainstem lesion extends longitudinally and is continuous with the spinal cord involvement. L: left; R: right.
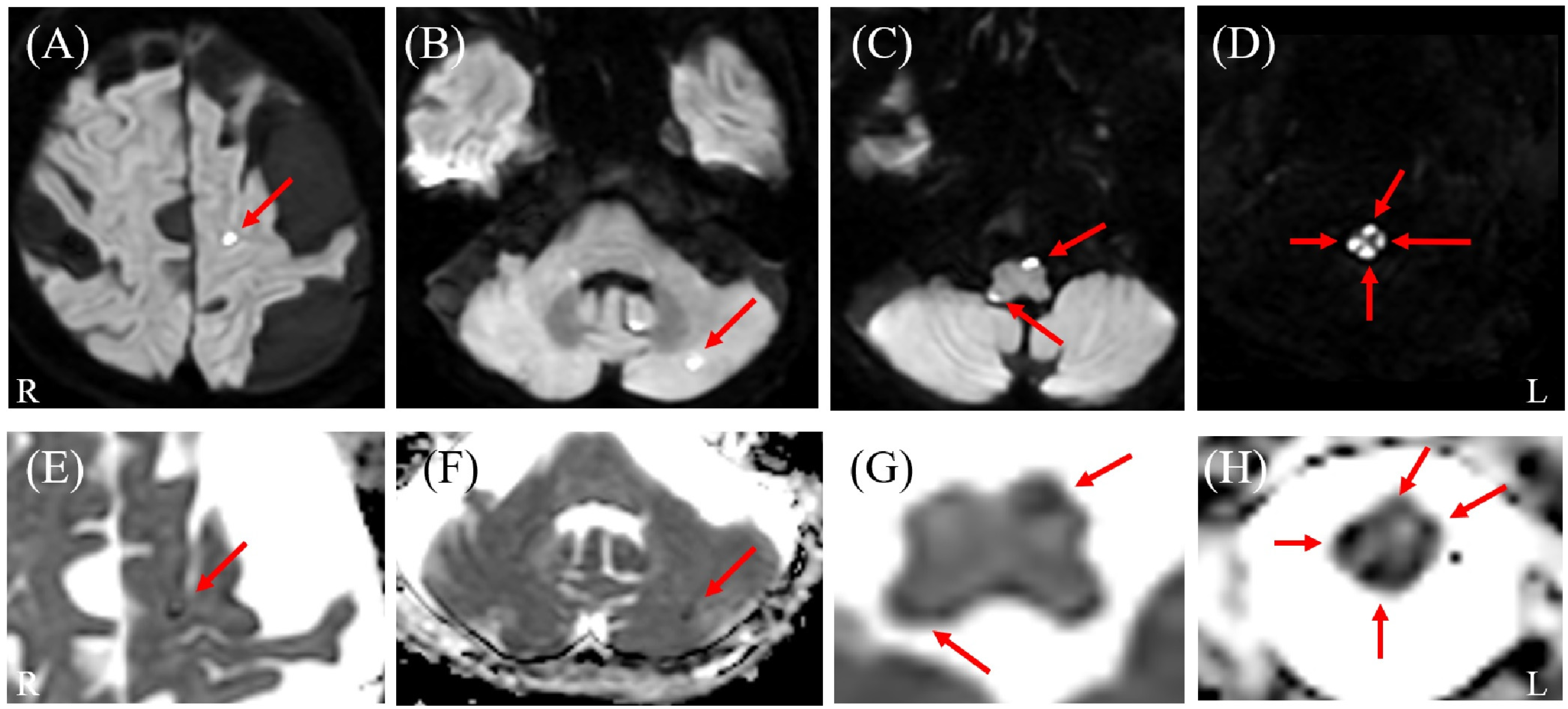
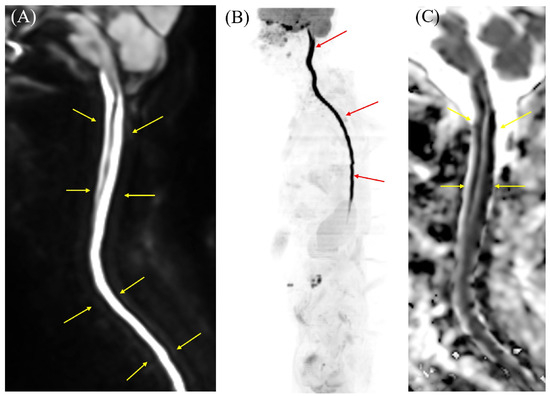
Figure 2.
Cervicothoracic MRI on DWI, diffusion-weighted whole-body imaging with background body signal (DWIBS), and ADC map results. (A) DWI demonstrates longitudinal hyperintensity extending from the cervical to the lumbar spinal cord (arrows). Similarly to the intracranial findings, this lesion demonstrated restricted diffusion. (B) DWIBS vividly displays high signal intensity along the entire cervicothoracolumbar spinal cord (arrows). DWIBS, first described by Takahara et al. in 2004 [1,2], is a sophisticated MRI technique rooted in DWI, which probes tissue microstructure via water molecule Brownian motion [2,3]. Employing short tau inversion recovery (STIR) echo-planar imaging (EPI) [1,2,3], it applies fat suppression and intense diffusion weighting to mute signals from healthy tissues, generating a stark “black background” [2,3]. Regions of restricted diffusion—e.g., malignancies, abscesses, or acute inflammation—stand out as bright hyperintensities [1,2,3,4,5]. These can be rendered as 3D PET-like images and co-registered with T2-weighted sequences for superior anatomical correlation [2,3]. (C) ADC maps show low ADC values corresponding to the areas of high signal intensity on DWI (arrows).
Figure 2.
Cervicothoracic MRI on DWI, diffusion-weighted whole-body imaging with background body signal (DWIBS), and ADC map results. (A) DWI demonstrates longitudinal hyperintensity extending from the cervical to the lumbar spinal cord (arrows). Similarly to the intracranial findings, this lesion demonstrated restricted diffusion. (B) DWIBS vividly displays high signal intensity along the entire cervicothoracolumbar spinal cord (arrows). DWIBS, first described by Takahara et al. in 2004 [1,2], is a sophisticated MRI technique rooted in DWI, which probes tissue microstructure via water molecule Brownian motion [2,3]. Employing short tau inversion recovery (STIR) echo-planar imaging (EPI) [1,2,3], it applies fat suppression and intense diffusion weighting to mute signals from healthy tissues, generating a stark “black background” [2,3]. Regions of restricted diffusion—e.g., malignancies, abscesses, or acute inflammation—stand out as bright hyperintensities [1,2,3,4,5]. These can be rendered as 3D PET-like images and co-registered with T2-weighted sequences for superior anatomical correlation [2,3]. (C) ADC maps show low ADC values corresponding to the areas of high signal intensity on DWI (arrows).
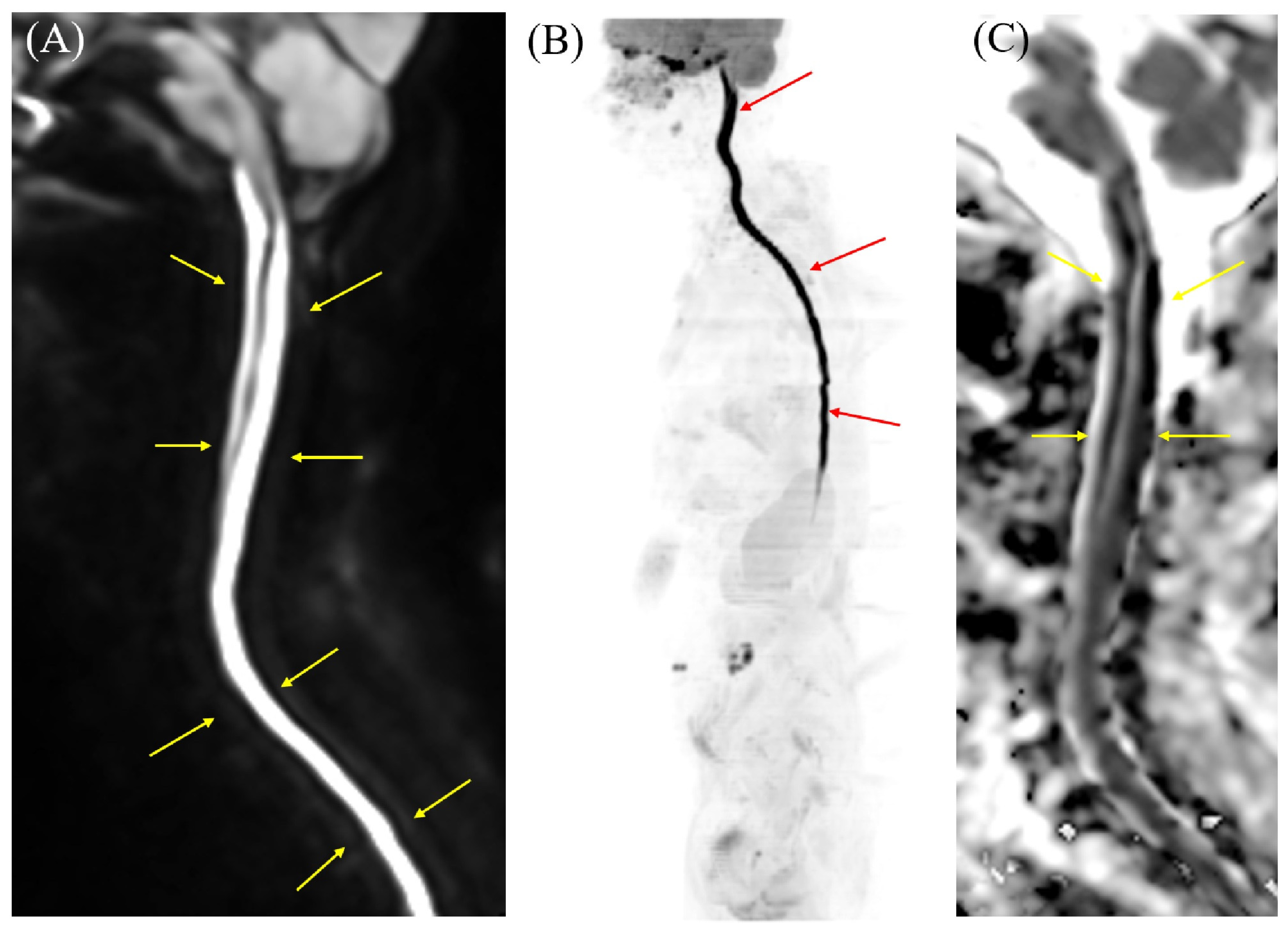
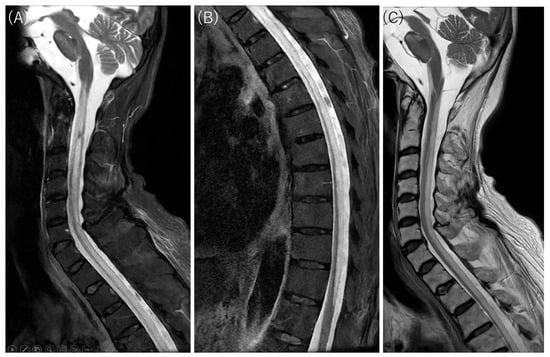
Figure 3.
Cervicothoracic MRI findings. Hyperintensity on short tau inversion recovery fast spin-echo (STIR FSE) (A,B) and T2-weighted imaging (C) corresponds to the longitudinally extensive diffusion-restricted lesions observed along the spinal cord. These axial sections reveal diffuse involvement of the entire white matter circumference and robust diffusion restriction (low ADC), distinguishing the pathology from the central gray matter target and isointense or increased ADC values typical of neuromyelitis optica spectrum disorder (NMOSD) [6,7]. Although the massive elevation of CSF myelin basic protein (>500 pg/mL) and positive oligoclonal bands indicate an extensive breakdown of the myelin sheath, the diagnosis of NMOSD and other inflammatory myelopathies was excluded by negative serology for anti-aquaporin-4 (AQP4), anti-myelin oligodendrocyte glycoprotein (MOG), and anti-glial fibrillary acidic protein (GFAP) antibodies, combined with a lack of cerebrospinal fluid (CSF) pleocytosis and the clinical attribution of emesis to intestinal perforation rather than an area postrema lesion.
Figure 3.
Cervicothoracic MRI findings. Hyperintensity on short tau inversion recovery fast spin-echo (STIR FSE) (A,B) and T2-weighted imaging (C) corresponds to the longitudinally extensive diffusion-restricted lesions observed along the spinal cord. These axial sections reveal diffuse involvement of the entire white matter circumference and robust diffusion restriction (low ADC), distinguishing the pathology from the central gray matter target and isointense or increased ADC values typical of neuromyelitis optica spectrum disorder (NMOSD) [6,7]. Although the massive elevation of CSF myelin basic protein (>500 pg/mL) and positive oligoclonal bands indicate an extensive breakdown of the myelin sheath, the diagnosis of NMOSD and other inflammatory myelopathies was excluded by negative serology for anti-aquaporin-4 (AQP4), anti-myelin oligodendrocyte glycoprotein (MOG), and anti-glial fibrillary acidic protein (GFAP) antibodies, combined with a lack of cerebrospinal fluid (CSF) pleocytosis and the clinical attribution of emesis to intestinal perforation rather than an area postrema lesion.
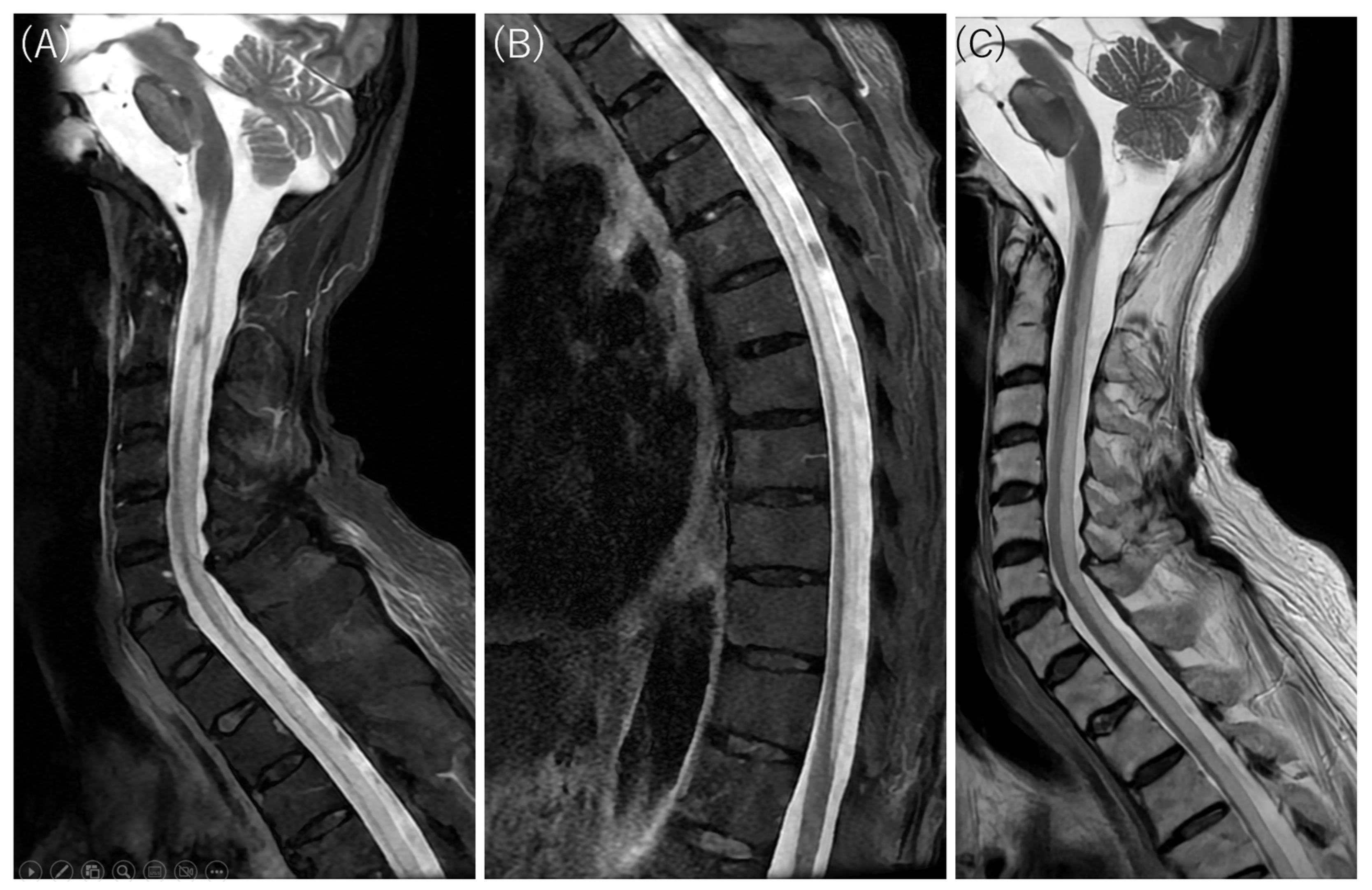
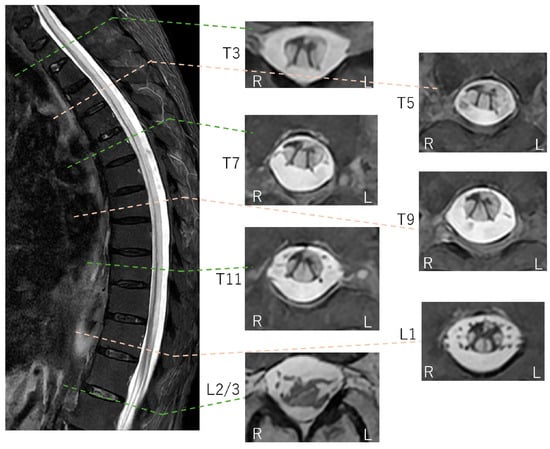
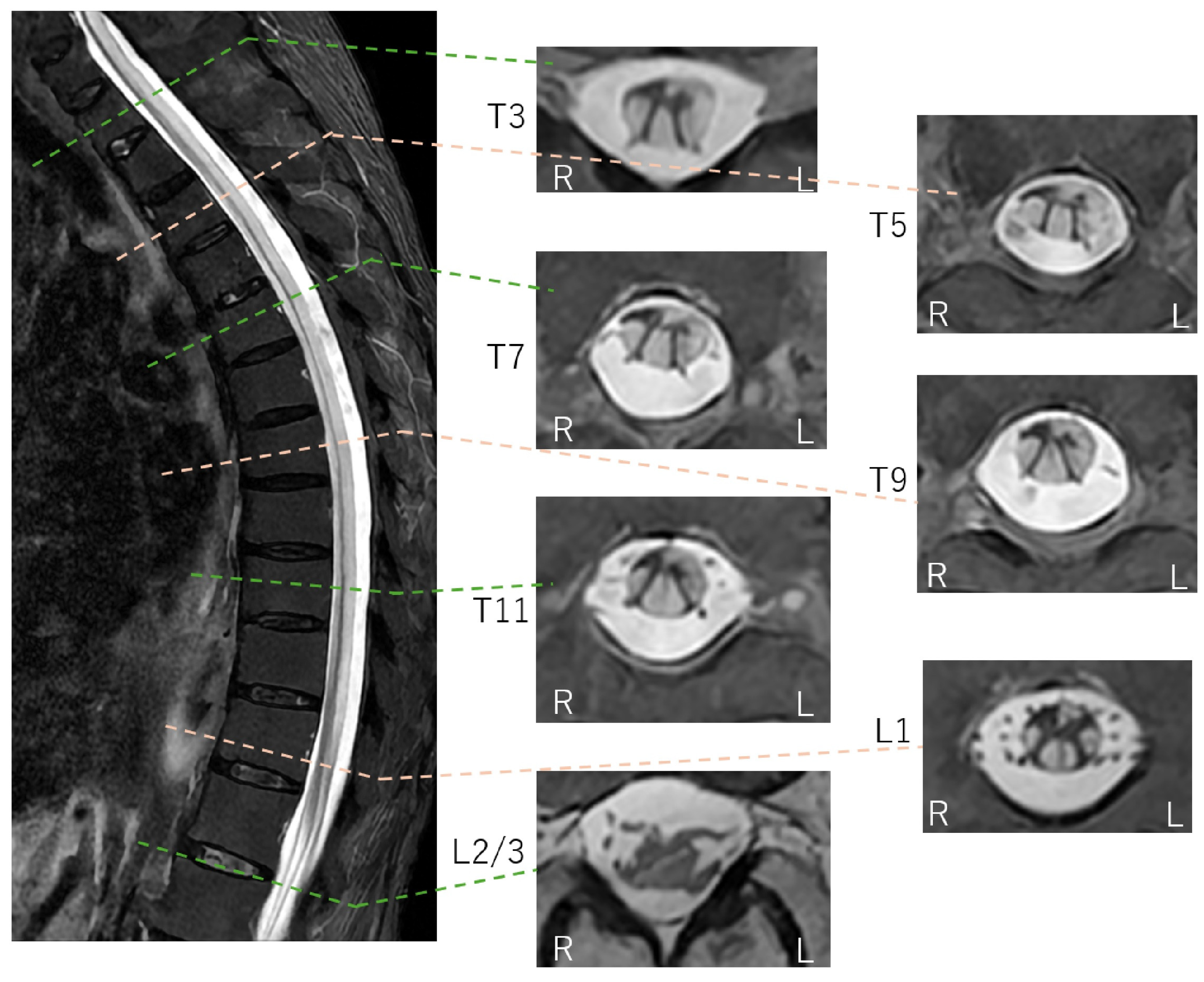
Figure 4.
Thoracolumbar STIR FSE MRI findings (axial section). Corresponding to the hyperintensities seen on DWI, high signal intensities are observed within the spinal cord extending down to the lumbar level on STIR-FSE images. In the axial sections, diffuse hyperintensities are noted in the anterior, lateral, and posterior columns. Conversely, the areas with preserved signal intensity are consistent with the spinal gray matter, which appears markedly atrophic. This tract-predominant distribution is a key imaging signature that distinguishes this case from typical spinal cord infarction (SCI), which follows specific arterial territories (anterior or posterior spinal arteries). While venous infarction similarly lacks arterial territory adherence, it is excluded here due to its rarity and the absence of predisposing vascular malformations. Methotrexate (MTX) neurotoxicity is a potential synergistic factor in this case. Although typically associated with intrathecal administration, oral MTX used for the patient’s rheumatoid arthritis can induce white matter vacuolar degeneration [8,9,10]. The observed tract-predominant distribution—specifically involving the posterior and lateral columns—aligns with the known patterns of MTX-induced cord injury, albeit rarely manifesting as such longitudinally extensive involvement from oral dosing. Suspected late-onset ornithine transcarbamylase (OTC) deficiency is supported by hallmark biomarkers, including hyperammonemia, urinary orotic acid, and low citrulline [11,12], though definitive genetic or enzymatic confirmation was not performed. Mechanistically, hyperammonemia-induced glutamine accumulation leads to astrocyte swelling and cytotoxic edema [11,12,13], potentially explaining the diffuse, tract-predominant diffusion restriction observed from the brainstem to the spinal cord. Folate deficiency (potentially due to MTX’s side effect) and hypocupremia (34 μg/dL) might act as synergistic factors in this LESCL, as copper is essential for mitochondrial electron transport and myelin maintenance [14,15,16,17]. Resulting enzymatic dysfunction typically manifests as symmetric T2 hyperintensities in the dorsal and lateral columns [14,15,16,17]. Collectively, the imaging and biochemical profile suggests a catastrophic metabolic breakdown of the spinal white matter architecture driven by the combined effects of suspected late-onset OTC deficiency, hypocupremia, and MTX-induced neurotoxicity. When neuroimaging patterns mismatch recognized vascular or inflammatory territories, complex metabolic and toxic etiologies must be prioritized in the differential diagnosis of LESCL. L: left; R: right.
Figure 4.
Thoracolumbar STIR FSE MRI findings (axial section). Corresponding to the hyperintensities seen on DWI, high signal intensities are observed within the spinal cord extending down to the lumbar level on STIR-FSE images. In the axial sections, diffuse hyperintensities are noted in the anterior, lateral, and posterior columns. Conversely, the areas with preserved signal intensity are consistent with the spinal gray matter, which appears markedly atrophic. This tract-predominant distribution is a key imaging signature that distinguishes this case from typical spinal cord infarction (SCI), which follows specific arterial territories (anterior or posterior spinal arteries). While venous infarction similarly lacks arterial territory adherence, it is excluded here due to its rarity and the absence of predisposing vascular malformations. Methotrexate (MTX) neurotoxicity is a potential synergistic factor in this case. Although typically associated with intrathecal administration, oral MTX used for the patient’s rheumatoid arthritis can induce white matter vacuolar degeneration [8,9,10]. The observed tract-predominant distribution—specifically involving the posterior and lateral columns—aligns with the known patterns of MTX-induced cord injury, albeit rarely manifesting as such longitudinally extensive involvement from oral dosing. Suspected late-onset ornithine transcarbamylase (OTC) deficiency is supported by hallmark biomarkers, including hyperammonemia, urinary orotic acid, and low citrulline [11,12], though definitive genetic or enzymatic confirmation was not performed. Mechanistically, hyperammonemia-induced glutamine accumulation leads to astrocyte swelling and cytotoxic edema [11,12,13], potentially explaining the diffuse, tract-predominant diffusion restriction observed from the brainstem to the spinal cord. Folate deficiency (potentially due to MTX’s side effect) and hypocupremia (34 μg/dL) might act as synergistic factors in this LESCL, as copper is essential for mitochondrial electron transport and myelin maintenance [14,15,16,17]. Resulting enzymatic dysfunction typically manifests as symmetric T2 hyperintensities in the dorsal and lateral columns [14,15,16,17]. Collectively, the imaging and biochemical profile suggests a catastrophic metabolic breakdown of the spinal white matter architecture driven by the combined effects of suspected late-onset OTC deficiency, hypocupremia, and MTX-induced neurotoxicity. When neuroimaging patterns mismatch recognized vascular or inflammatory territories, complex metabolic and toxic etiologies must be prioritized in the differential diagnosis of LESCL. L: left; R: right.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/diagnostics16030504/s1, File S1: The CARE reporting checklist.
Author Contributions
Conceptualization, Y.N., K.H. (Koji Hayashi) and Y.K.; methodology, Y.N. and K.H. (Koji Hayashi); validation, Y.N., K.H. (Koji Hayashi), M.S., Y.M., T.M., H.H., K.H. (Kouji Hayashi) and Y.K.; investigation (clinical care), Y.N., K.H. (Koji Hayashi), M.S. and Y.M.; resources, Y.N. and K.H. (Koji Hayashi); data curation, Y.N. and K.H. (Koji Hayashi); writing—original draft preparation, Y.N. and K.H. (Koji Hayashi); writing—review and editing, Y.N., K.H. (Koji Hayashi), M.S., Y.M., T.M., H.H., K.H. (Kouji Hayashi) and Y.K.; visualization, Y.N. and K.H. (Koji Hayashi); supervision, Y.K.; project administration, K.H. (Koji Hayashi). All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
In accordance with the Ethical Guidelines for Medical and Health Research Involving Human Subjects issued by the Ministry of Health, Labor and Welfare of Japan, case reports involving a single patient typically do not require formal approval from an Institutional Review Board (IRB). Therefore, the ethical review and approval were waived for this study by the Ethics Committee of Fukui General Hospital. Additionally, this case report has been prepared in accordance with the CARE guidelines (File S1) to ensure a comprehensive and transparent description of the clinical findings and diagnostic process.
Informed Consent Statement
Written informed consent for the publication of this case report and the accompanying neuroimaging was obtained from the patient’s family.
Data Availability Statement
The data presented in this study is available on request from the corresponding author. Due to patient privacy and ethical considerations, the data is not publicly accessible.
Acknowledgments
During the preparation of this manuscript, the authors used ChatGPT (GPT-4.1) for the purposes of language editing only. No data or conclusions were generated by AI. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CT | Computed tomography |
| MRI | Magnetic resonance imaging |
| DWIBS | Diffusion-weighted whole-body imaging with background body signal suppression |
| LESCL | Longitudinally extensive spinal cord lesion |
| MBP | Myelin basic protein |
| MTX | Methotrexate |
| OTC | Ornithine transcarbamylase |
| NMOSD | Neuromyelitis optica spectrum disorder |
| EPI | Echo-planar imaging |
| DWI | Diffusion-weighted imaging |
| ADC | Apparent diffusion coefficient |
| MOG | Myelin oligodendrocyte glycoprotein |
| AQP4 | Aquaporin-4 |
| GFAP | Glial fibrillary acidic protein |
| SCI | Spinal cord infarction |
References
- Suzuki, A.; Hayashi, K.; Sato, M.; Nakaya, Y.; Miura, T.; Takaku, N.; Iwasaki, T.; Kobayashi, Y. Visualizing Aortic Inflammation by Diffusion-Weighted Whole-Body Imaging with Background Body Signal Suppression (DWIBS). Diagnostics 2025, 15, 1151. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Hayashi, K.; Sato, M.; Iwasaki, T.; Kobayashi, Y. Uncovering sternoclavicular arthritis, suspected pseudogout, in a fever of unknown origin by whole-body MRI. Diagnostics 2025, 15, 2032. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Hayashi, K.; Misaki, K.; Sato, M.; Nakaya, Y.; Iwasaki, T.; Kobayashi, Y. A case of pyogenic vertebral osteomyelitis and iliopsoas abscess caused by invasive pneumococcal disease serotype 35F: Utility of diffusion-weighted whole-body imaging with background body signal suppression as an adjunctive diagnostic tool. Cureus 2025, 17, e87770. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Hayashi, K.; Suzuki, A.; Sato, M.; Nakaya, Y.; Takaku, N.; Miura, T.; Kobayashi, Y. A case of synovitis acne pustulosis hyperostosis osteitis (SAPHO) syndrome with myeloperoxidase anti-neutrophil cytoplasmic antibody: Exploring an association or coincidence. Cureus 2025, 17, e83866. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Hayashi, M.; Izumi, R.; Sato, M.; Hayashi, S.; Iwasaki, T.; Sakamaki, I.; Kobayashi, Y. Unveiling hidden abscesses: The clinical utility of diffusion-weighted whole-body imaging with background suppression (DWIBS) in metastatic abscess screening. Diagnostics 2026, 16, 223. [Google Scholar] [CrossRef] [PubMed]
- Wingerchuk, D.M.; Banwell, B.; Bennett, J.L.; Cabre, P.; Carroll, W.; Chitnis, T.; de Seze, J.; Fujihara, K.; Greenberg, B.; Jacob, A.; et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015, 85, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M. The utility of diffusion-weighted imaging in patients with spinal cord infarction: Difference from the findings of neuromyelitis optica spectrum disorder. BMC Neurol. 2022, 22, 382. [Google Scholar] [CrossRef] [PubMed]
- Shintaku, M.; Toyooka, N.; Koyama, T.; Teraoka, S.; Tsudo, M. Methotrexate myelopathy with extensive transverse necrosis: Report of an autopsy case. Neuropathology 2014, 34, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Satomi, K.; Yoshida, M.; Matsuoka, K.; Okita, H.; Hosoya, Y.; Shioda, Y.; Kumagai, M.-A.; Mori, T.; Morishita, Y.; Noguchi, M.; et al. Myelopathy mimicking subacute combined degeneration in a Down syndrome patient with methotrexate treatment for B lymphoblastic leukemia: Report of an autopsy case. Neuropathology 2014, 34, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.H.; Yang, E.; Chen, Y.C. Ascending myelopathy after intrathecal methotrexate. Kaohsiung J. Med. Sci. 2021, 37, 1018–1019. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Lu, G.; Mowshica, R.; Cheng, Y.; Zhao, M. Clinical and cranial MRI features of female patients with ornithine transcarbamylase deficiency: Two case reports. Medicine 2019, 98, e16827. [Google Scholar] [CrossRef] [PubMed]
- Sen, K.; Anderson, A.A.; Whitehead, M.T.; Gropman, A.L. Review of multi-modal imaging in urea cycle disorders: The old, the new, the borrowed, and the blue. Front. Neurol. 2021, 12, 632307. [Google Scholar] [CrossRef] [PubMed]
- Takanashi, J.I.; Barkovich, A.J.; Cheng, S.F.; Kostiner, D.; Baker, J.C.; Packman, S. Brain MR imaging in acute hyperammonemic encephalopathy arising from late-onset ornithine transcarbamylase deficiency. AJNR Am. J. Neuroradiol. 2003, 24, 390–393. [Google Scholar] [PubMed]
- Gabreyes, A.A.; Abbasi, H.N.; Forbes, K.P.; McQuaker, G.; Duncan, A.; Morrison, I. Hypocupremia associated cytopenia and myelopathy: A national retrospective review. Eur. J. Haematol. 2013, 90, 1–9. [Google Scholar] [CrossRef]
- Marotta, D.A.; Mason, M.C.; Abraham, B.M.; Kesserwani, H. Myeloneuropathy in the setting of hypocupremia: An overview of copper-related pathophysiology. Cureus 2021, 13, e16254. [Google Scholar] [CrossRef] [PubMed]
- Pantbalekundri, N.; Kumar, S.; Acharya, S.; Bedi, G. Postpartum hypocupric myelopathy masquerading as acute transverse myelitis: A case report and literature review of a rare presentation. Cureus 2024, 16, e52149. [Google Scholar] [CrossRef] [PubMed]
- Tornabene, D.; Bini, P.; Gastaldi, M.; Vegezzi, E.; Asteggiano, C.; Marchioni, E.; Diamanti, L. Neurological complications due to copper deficiency in the context of Wilson disease treatment: A case report with long-term follow-up and review of the literature. Neurol. Sci. 2024, 45, 987–996. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
