Abstract
Chalcopyrite leaching in sulfuric acid with chloride-based oxidizing agents (NaCl, NaClO, NaClO3) was investigated to optimize copper recovery. The influence of sulfuric acid concentration, chloride concentration, and temperature on copper dissolution was systematically evaluated through experimental tests. A Gaussian Process Regression (GPR) model was developed to predict copper recovery, integrating experimental data with Partial Rank Correlation Coefficient (PRCC) analysis to assess the impact of key variables. The results showed that NaClO and NaClO3 significantly improved copper recovery, with NaClO3 achieving nearly 100% copper recovery in under 30 min at higher temperatures. Maximum recovery of 45.5% was achieved with NaCl at 1 M concentration, 3 M H2SO4, and 80 °C. The GPR model demonstrated superior predictive accuracy, achieving RMSE = 4.0028 and R2 = 0.99, outperforming Support Vector Machine Regression (SVMR) and Ensemble Regression (ER) models. The GPR model accurately predicted recovery under conditions not tested experimentally, providing a robust tool for process optimization. The results confirm the effectiveness of chloride-based oxidizers in enhancing copper dissolution and demonstrate the practical application of GPR for optimizing leaching conditions, ensuring maximum copper recovery in hydrometallurgical processes.
1. Introduction
Copper extraction from chalcopyrite (CuFeS2) remains a critical challenge in hydrometallurgy due to the mineral’s refractory nature and slow dissolution kinetics [1,2,3]. Conventional leaching in sulfuric acid often results in limited copper recovery, prompting the development of alternative methods such as chloride-assisted leaching, iodine-assisted processes, and bioleaching systems [1,4,5]. Recent studies have highlighted the significance of oxidizing agents, temperature, and solution chemistry on chalcopyrite dissolution [1,6,7,8,9,10].
Scale-up studies of iodine-assisted leaching from laboratory reactors to heap columns have demonstrated that larger-scale operations face slower copper dissolution due to particle size effects and partial liberation of chalcopyrite grains [1,11]. Dual-scale pore network modeling further revealed that heterogeneous flow pathways and micro-porosity significantly affect leach solution distribution and mass transfer in heap systems [9,12,13,14]. Similarly, auto-thermal bioleaching models integrating microbial kinetics, oxygen transport, and energy balances showed that temperature-driven thermophilic activity critically influences copper recovery [4,15,16,17].
Deterministic modeling approaches, such as HeapSim, have successfully coupled chemical, biological, and hydrodynamic phenomena to simulate heap leaching, emphasizing the role of oxygen transfer, microbial competition, and kinetic constraints at different temperature regimes [3,18,19]. These models provide mechanistic insights but are often limited by assumptions of homogeneity, the reliance on laboratory-calibrated parameters, and computational complexity when scaling to industrial heaps [20].
In recent years, artificial intelligence (AI) and machine learning (ML) approaches, including artificial neural networks (ANN), support vector machines (SVM), and Gaussian Process Regression (GPR), have emerged as promising tools for predicting complex, nonlinear leaching processes without the need for explicit mechanistic assumptions [21,22,23,24]. Data-driven models have been successfully applied to simulate dynamic leachate behavior, optimize operational parameters, and forecast industrial copper production with high accuracy [23,25]. Hybrid frameworks combining mechanistic and ML-based models can further enhance predictive power while maintaining interpretability, offering potential for real-time process control and digital twins of hydrometallurgical operations [21].
Despite these advances, significant knowledge gaps remain in accurately predicting chalcopyrite dissolution under combined sulfate-chloride systems, particularly regarding the influence of chloride-based oxidizers on copper recovery kinetics and the interactions between operational variables [2,26]. Furthermore, most previous ML applications in leaching have focused on bioleaching or industrial-scale prediction, leaving a gap for integrating experimental optimization with predictive ML models in sulfate-chloride leaching systems [21,22].
The present study addresses these gaps by combining controlled experimental evaluation of chalcopyrite leaching in sulfuric acid with chloride-based oxidizers (NaCl, NaClO, NaClO3) and a Gaussian Process Regression (GPR) model. The model integrates Partial Rank Correlation Coefficient (PRCC) analysis to identify the most influential variables, enabling accurate prediction of copper recovery under both tested and untested conditions. The novelty of this work lies in (i) applying chloride-based oxidizers under systematically varied chemical and thermal conditions, (ii) developing a GPR-based predictive model with superior accuracy compared to traditional regression and ensemble methods, and (iii) providing a robust data-driven framework for optimizing sulfate-chloride leaching of chalcopyrite, bridging experimental insights with predictive machine learning to enhance copper recovery and operational efficiency.
2. Materials and Methods
2.1. Sample Preparation
The chalcopyrite concentrate used in this study was sourced from the Sonqon copper mine in East Azerbaijan Province, Iran. A total of 10 kg of concentrate was collected over five days for chemical and mineralogical analysis. The material was washed with deionized water to remove flotation reagents and then filtered.
Chemical analysis revealed the following composition: 23.59% total copper, 0.36% copper oxide, 30.21% total iron, 36% sulfur, and 0.099% molybdenum. X-ray diffraction analysis showed that the sample was primarily composed of chalcopyrite (42%) and pyrite (35%), with minor amounts of covellite (10%), molybdenite (0.1%), chalcocite (1%), sphalerite (1%), tennantite (1%), and other minerals (10%).
2.2. Chemicals and Equipment for Leaching Experiments
Leaching experiments were performed using analytical-grade reagents, including sulfuric acid (H2SO4, 98%), sodium chloride (NaCl, 99%), sodium chlorate (NaClO3, 100%), sodium hypochlorite (NaClO, 10%), and deionized water.
All experiments were conducted in 500 mL two-neck round-bottom flasks placed in an oil bath for uniform heat distribution. Mechanical stirring and heating were provided by a hot plate, with temperature monitored by a GHZEAL thermometer. A spiral condenser was used to prevent solution evaporation.
2.3. Experimental Design and Procedure
Leaching experiments were optimized using Minitab software (Statistical, Version 19) with a Taguchi mixed-level design to assess the impact of different chloride salts (NaCl, NaClO, NaClO3) on copper dissolution. Three key factors were considered for each chloride reagent: chloride salt concentration, sulfuric acid concentration, and temperature, while the solution volume was fixed at 400 mL. The experimental design and combinations are summarized in Table 1, Table 2 and Table 3.

Table 1.
Taguchi mixed-level design for NaCl leaching system.

Table 2.
Taguchi mixed-level design for NaClO leaching system.

Table 3.
Taguchi mixed-level design for NaClO3 leaching system.
In each experiment, 400 mL of sulfuric acid solution was prepared, and the selected chloride reagent was added under mechanical stirring. After stabilizing the temperature in an oil bath, 6 mL samples were taken at fixed time intervals (0.5, 1, 2, 4, 8, and 24 h). These samples were centrifuged, filtered, and analyzed for copper concentration using atomic absorption spectroscopy (AAS). Experiments were performed once under each condition. The remaining solution was returned to the reactor to maintain the solid liquid ratio. The leaching time was set to 24 h, which was sufficient to capture the main dissolution trends, as preliminary tests showed that copper recovery reached near-maximum levels within this period, especially for NaClO3. Longer-term stability and reaction behavior after potential oxidant depletion were not investigated and are recommended for future work.
2.4. Modeling Approach and Data Analysis
Copper recovery predictions under different leaching conditions were made using Gaussian Process Regression (GPR). This model was used to simulate the complex, nonlinear relationships between various process variables (such as sulfuric acid concentration, chloride additives, temperature, and reaction time) and copper recovery. To enhance the accuracy of the model, experimental data collected from the leaching tests were input into the GPR model, which was then trained using data-driven learning algorithms.
To assess the relative importance of each variable on copper recovery, Partial Rank Correlation Coefficient (PRCC) analysis was applied. The variable importance derived from PRCC analysis was systematically incorporated into the GPR kernel, assigning higher weights to more influential parameters. This integration enables the model to account for nonlinear and interactive effects among acid concentration, temperature, reaction time, and different chloride-based oxidizers, enhancing predictive reliability. Consequently, the GPR framework can accurately estimate copper recovery not only under conditions included in the experimental dataset but also for untested scenarios, providing a robust and interpretable tool for process optimization. This method helped identify nonlinear and interactive effects of the variables on recovery, providing valuable insights for the optimization of input parameters.
The results from the GPR model were used to predict copper recovery under conditions that were not included in the experimental tests. Additionally, the model was validated by comparing the predictions with the actual experimental data.
3. Results and Discussions
3.1. Chalcopyrite Leaching in Sulfuric Acid Without Chloride Additives
Experiments were conducted to establish a baseline for chalcopyrite leaching in sulfuric acid without chloride additives. Acid concentrations ranged from 0.4 to 0.8 M, and temperatures were set between 40 and 80 °C, with constant stirring and fixed solid-to-liquid ratios. Copper recoveries obtained under these conditions are summarized in Table 4.

Table 4.
Baseline experiments on the effective parameters on H2SO4 leaching (copper recovery, %) without additives.
The results indicate that increasing the temperature significantly enhances copper recovery. At 40 °C with 0.4 M H2SO4, the 24 h recovery reached only 12.4%, whereas at 80 °C it increased to 16.1%. Increasing the acid concentration from 0.4 M to 0.8 M at 80 °C slightly reduced recovery to 14.9%, suggesting the possible formation of a passivating surface layer.
Analysis of the more extensive data in Table 5 and Table 6 further clarifies these trends. Raising the acid concentration from 0.5 M to 3 M gradually increased the 24 h copper recovery from 15.3% to 16.8%, while increasing the temperature from 21 to 100 °C improved recovery from 11.2% to 16.6%. These findings confirm that both temperature and acid concentration are key factors controlling chalcopyrite dissolution, but even under the most favorable conditions without chloride, final copper recovery remained below 17%, highlighting the refractory nature of chalcopyrite in pure sulfuric acid.

Table 5.
Effect of H2SO4 concentration on copper recovery (%) without additives.

Table 6.
Effect of temperature on H2SO4 leaching (copper recovery, %) without additives.
Overall, higher temperatures accelerate the initial dissolution rate and increase the final recovery, with the most pronounced effects observed between 40 and 80 °C. Similarly, increasing acid concentration up to approximately 3 M gradually improves recovery, whereas further increases result in diminishing returns, likely due to surface passivation. The maximum copper recovery observed without chloride additives is limited to 16–17%, emphasizing the need for optimized conditions or the use of additives to enhance leaching efficiency.
3.2. Chalcopyrite Leaching with Sulfuric Acid and Chloride Additives
Leaching experiments were designed using the Taguchi mixed-level method to evaluate the effects of sodium chloride (NaCl), sodium hypochlorite (NaClO), and sodium chlorate (NaClO3) on copper dissolution from chalcopyrite concentrate. The tests were carried out for 24 h under identical hydrodynamic conditions (impeller speed: 800 rpm) with varying concentrations of each chloride reagent, sulfuric acid concentration, and temperature.
NaCl was used to disrupt the passivating film on chalcopyrite, improving copper recovery. The highest recovery (45.5%) occurred at 1 M NaCl, 3 M H2SO4, and 80 °C. However, increasing NaCl concentration beyond 1.0 M led to diminishing returns, suggesting excessive chloride ions may hinder dissolution. Table 7 shows the effect of NaCl concentration on copper recovery.

Table 7.
Copper recovery (%) of Taguchi mixed-level chalcopyrite leaching with sulfuric acid and NaCl.
In NaClO experiments, increasing temperature significantly enhanced copper recovery. The highest recovery occurred at 1 M NaClO and 65 °C, achieving complete dissolution in under 2 h. After 2 h, the dissolution rate decreased due to depletion of active chlorine species. Table 8 illustrates the effect of NaClO concentration.

Table 8.
Copper recovery (%) of Taguchi mixed-level chalcopyrite leaching with sulfuric acid and NaClO.
NaClO3 led to rapid chalcopyrite dissolution, with 100% copper recovery achieved in less than 30 min at elevated temperatures. Table 9 shows the effect of NaClO3 concentration.

Table 9.
Copper recovery (%) of Taguchi mixed-level chalcopyrite leaching with sulfuric acid and NaClO3.
In comparison, NaCl showed moderate improvement in copper recovery, while NaClO and NaClO3 provided faster and more complete dissolution, with NaClO3 being the most efficient at higher temperatures.
During the leaching experiments, the solution redox potential was monitored and remained within a narrow range of 420–440 mV, indicating relatively stable oxidizing conditions. Although the chloride ion consumption rate was not quantified, the enhanced copper recovery observed in the presence of chloride-based oxidants is consistent with previously reported chloride-assisted activation effects on chalcopyrite dissolution. Therefore, the discussion of passivation layer modification is based on recovery trends and literature evidence rather than direct electrochemical measurements.
Previous studies on chloride-assisted leaching of sulfide minerals have demonstrated that chloride ions can form soluble metal-chloro complexes, such as CuCl2−, which enhance copper solubility, and can also weaken or disrupt passive sulfur-rich surface films on chalcopyrite, facilitating higher metal dissolution [27,28,29]. In agreement with these mechanistic insights, our experimental results show that stronger oxidizing chloride species, specifically hypochlorite (NaClO) and chlorate (NaClO3), accelerate the breakdown of surface passivation and markedly increase copper recovery compared to simple chloride (NaCl). These observations confirm that both the oxidative strength and the chemical nature of the chloride species play critical roles in promoting chalcopyrite leaching efficiency.
Recent research has also highlighted the importance of optimizing oxidant type and concentration, as well as leveraging data-driven approaches for process prediction and control [6,12,23,24]. These works indicate that careful selection of chloride oxidants can accelerate dissolution and improve extraction efficiency without relying solely on prolonged reaction times.
Overall, the results of this study are in agreement with established literature and provide additional experimental evidence on the effectiveness of chloride-based oxidants for chalcopyrite leaching. By systematically comparing different chloride systems, this work complements previous mechanistic insights and offers practical guidance for optimizing leaching conditions in industrial applications [7,8].
3.3. Data-Driven Sensitivity and Regression Analysis for Predictive Estimation of Copper Recovery
3.3.1. Sensitivity Analysis of Copper Recovery
The Partial Rank Correlation Coefficient (PRCC) analysis revealed that NaClO3 (PRCC = 0.834) and o assess the relative influence of process variables on copper recovery, both Morris and Partial Rank Correlation Coefficient (PRCC) analyses were conducted. The PRCC results indicated that NaClO (r = 0.81, p < 0.001) and NaClO3 (r = 0.83, p < 0.001) were the most significant contributors to copper recovery, followed by reaction time (r = 0.66) and H2SO4 concentration (r = 0.47), while temperature and NaCl showed weaker but still positive correlations. Consistently, the Morris method identified temperature (μ* = 19.19, σ = 24.48), NaCl (μ* = 18.40, σ = 22.58), and NaClO3 (μ* = 16.59, σ = 25.30) as dominant and highly nonlinear factors, confirming the strong and interactive effects of these parameters. Together, these analyses demonstrate that oxidizing agents (NaClO, NaClO3) and temperature play the primary roles in enhancing copper dissolution, while acid concentration and leaching time exert secondary but consistent influences on recovery efficiency. The results are shown in Table 10.

Table 10.
Sensitivity Analysis of Copper Recovery Using PRCC and Morris Methods.
3.3.2. Predictive Modeling of Copper Recovery Using Gaussian Process Regression
To assess the predictive capability of different data-driven approaches, three regression models, Gaussian Process Regression (GPR), Support Vector Machine Regression (SVMR), and Ensemble Regression (ER), were trained and evaluated using five-fold cross-validation. The comparative results, presented in Table 11, demonstrate that all models effectively captured the nonlinear dependencies between leaching parameters and copper recovery. Among them, the GPR model exhibited the highest predictive accuracy, achieving the lowest RMSE and the highest R2 values.

Table 11.
Comparative results.
The PRCC-informed GPR framework advances previous ML applications by incorporating experimentally derived variable importance into the kernel structure, allowing the model to capture nonlinear interactions among critical process parameters. This results in more interpretable and reliable predictions, extending the applicability of the model beyond the conditions directly tested experimentally. The GPR kernel function was selected and optimized using PRCC results to incorporate the relative influence of each variable, and hyperparameters were tuned via five-fold cross-validation.
The GPR model was implemented in MATLAB using the fitrgp function with an Automatic Relevance Determination (ARD) Matérn 5/2 kernel, which allows for anisotropic smoothness and variable-specific length scales. For each input vector corresponds to one experimental leaching condition ( leaching time, H2SO4 Concentration, Temperature, NaCl Concentration, NaClO Concentration and NaClO3 Concentration) the kernel function is defined as
where represents the characteristic length scale of the -th input variable and denotes the signal variance. The initial kernel hyperparameters were initialized as , reflecting the priority ranking obtained from the sensitivity analysis presented in Table 10, and the initial signal standard deviation was fixed at 1 prior to optimization. Hyperparameter optimization was carried out by maximizing the log-marginal likelihood using L-BFGS with exact inference.
To complement the five-fold cross-validation employed for hyperparameter tuning and model selection, an independent holdout validation strategy was additionally implemented to provide an unbiased estimate of final model’s generalization performance on unseen data. The full dataset of n observations was randomly partitioned using MATLAB’s (Statistical, Version 19) cvpartition function with a holdout fraction of 0.1, resulting in a non-stratified split that allocated approximately 90% of the observations to the training set and the remaining 10% to an independent test set. This random partition was performed once after completing the five-fold cross-validation-based optimization phase. The final optimized model—trained on the larger training portion—was then evaluated on the test set to assess predictive accuracy, uncertainty quantification, and generalization beyond the cross-validation folds. This combined approach aligns with common best practices in data-driven studies, particularly when dataset sizes are moderate and a reliable, low-variance estimate of real-world performance is required, while avoiding overly optimistic bias from evaluating on the same data used during tuning.
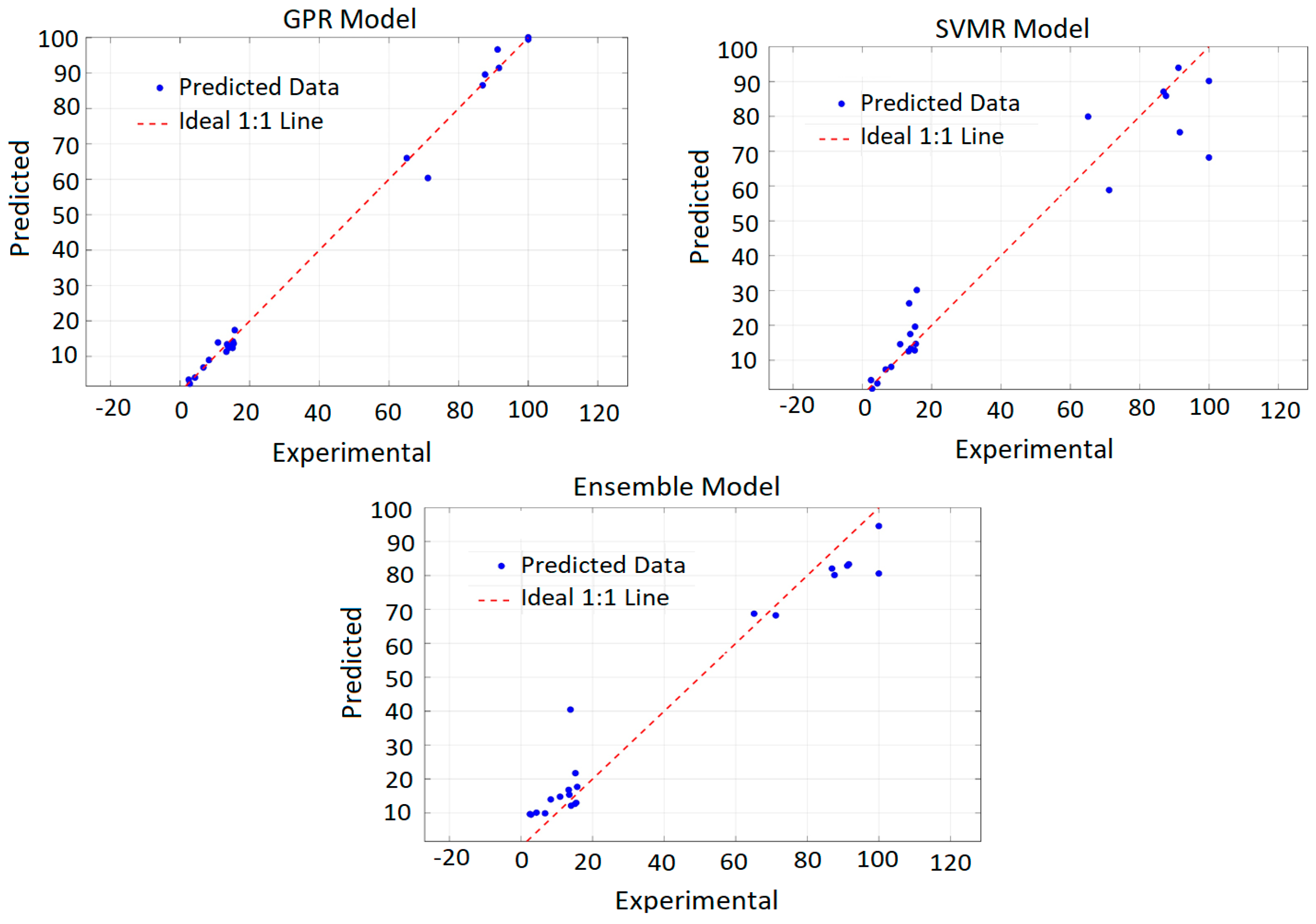
In addition to RMSE and R2, model training efficiency and prediction confidence intervals were examined, confirming the robustness and generalization capability of the model. As illustrated in Figure 1, the GPR predictions showed excellent agreement with experimental recovery values, closely following the ideal 1:1 line. Overall, the PRCC-informed GPR model provides a reliable and interpretable framework for predicting copper recovery under diverse leaching conditions.
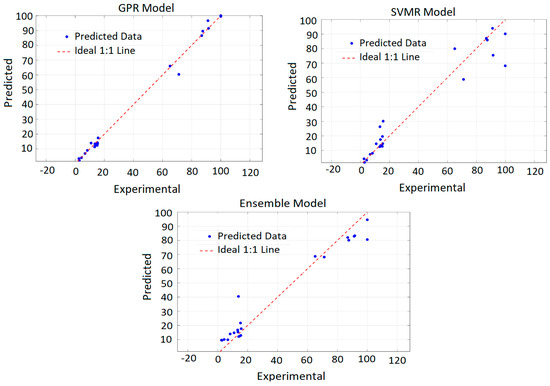
Figure 1.
Predicted vs. actual copper recovery (%) obtained from the developed models.
To ensure reliable generalization, cross-validation was employed during training, which also helped mitigate potential overfitting associated with experimental variability. The PRCC-informed GPR model was subsequently applied to predict copper recovery across the entire experimental domain, producing smooth and physically consistent response surfaces that capture the complex nonlinear relationships governing the leaching process. This comprehensive model thus provides a reliable predictive tool for identifying optimal combinations of temperature and leaching duration to maximize copper recovery.
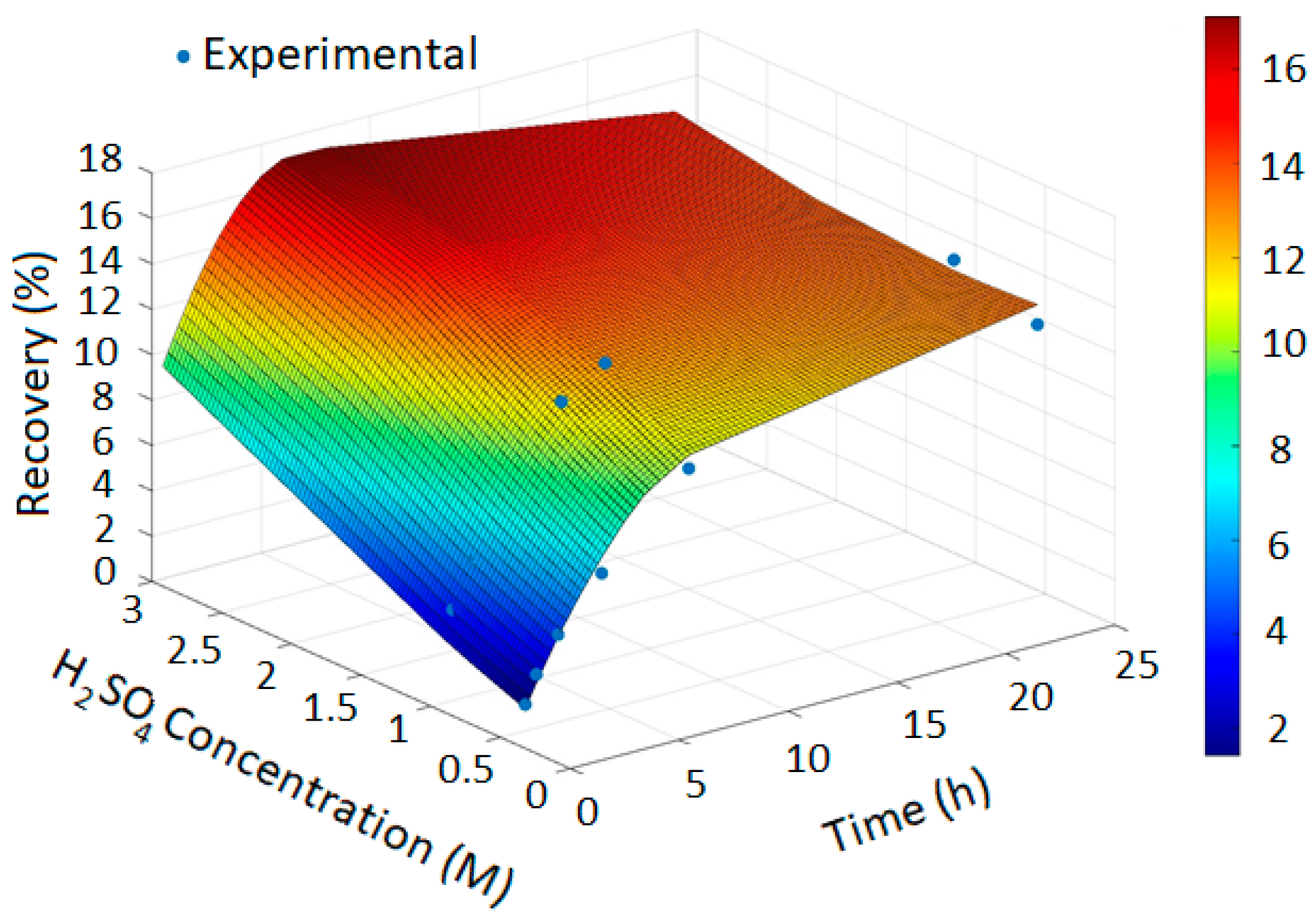
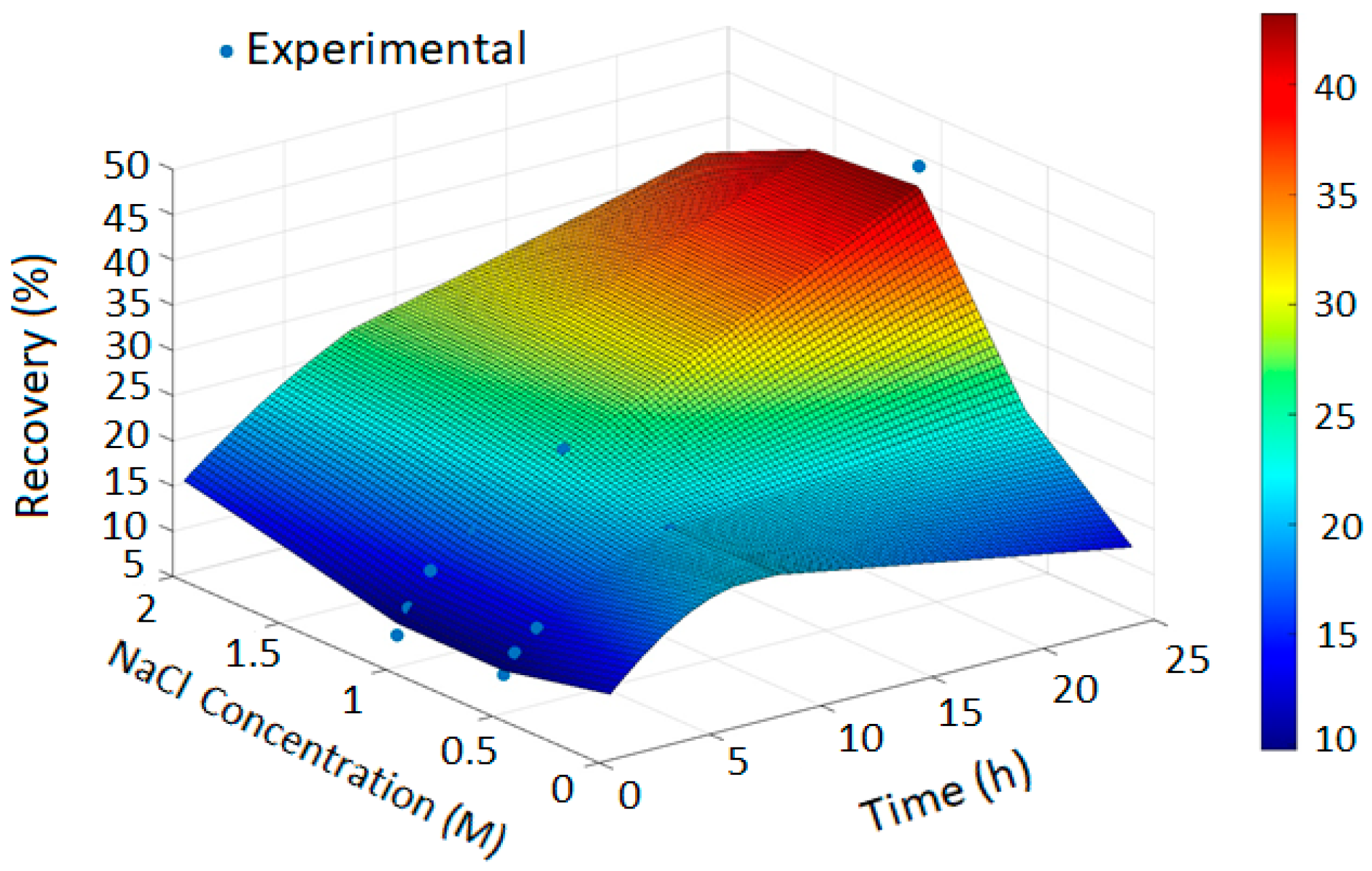
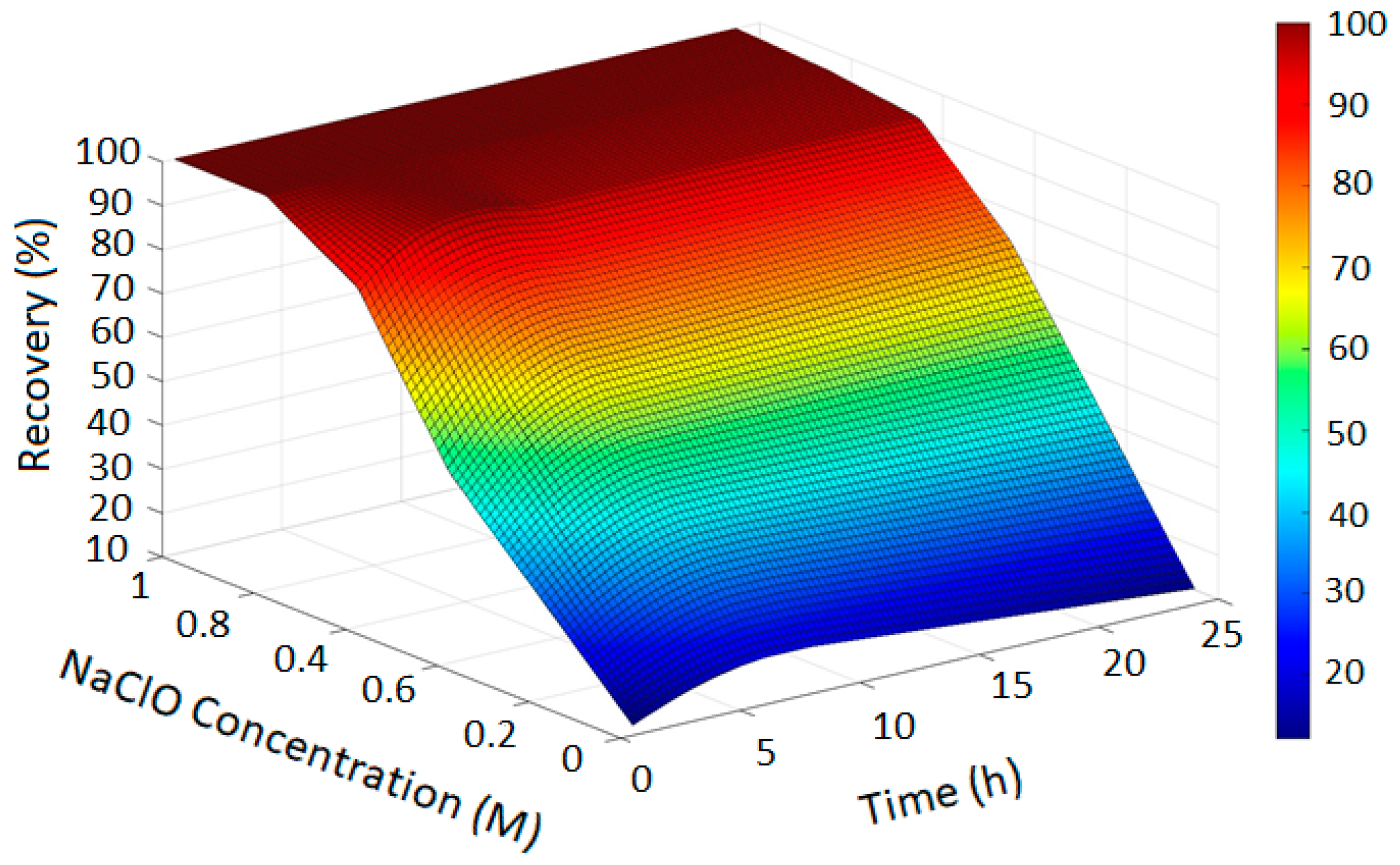
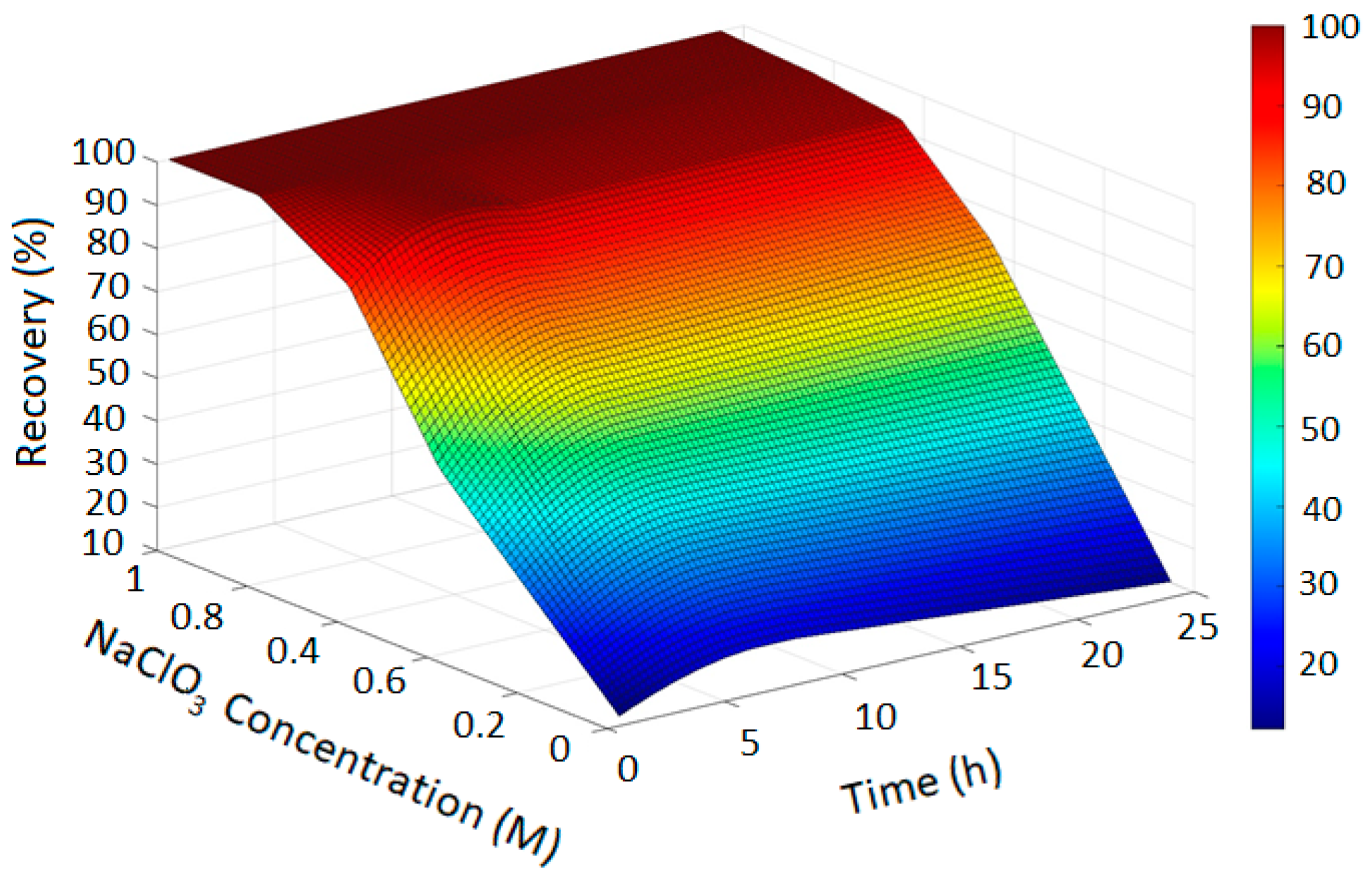
The PRCC-informed GPR models generated accurate prediction surfaces for copper recovery under different oxidizing chloride systems, as illustrated in the four 3D plots (Figure 2, Figure 3, Figure 4 and Figure 5). The x- and y-axes represent leaching time and chloride additive dosage, respectively, while the z-axis indicates the predicted copper recovery (%), with a color scale ranging from blue (minimum recovery) to red (maximum recovery).
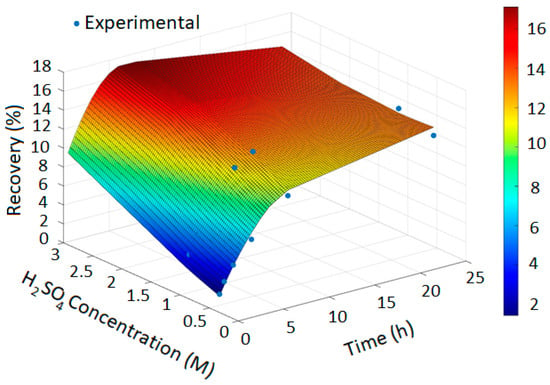
Figure 2.
Copper Recovery Predicted by PRCC-informed GPR (Temperature = 40 °C, NaCl = 0, NaClO = 0 and NaClO3 = 0).
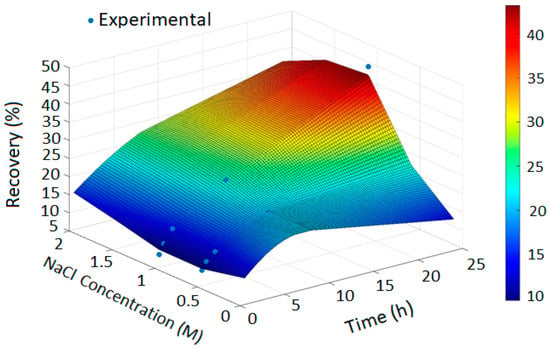
Figure 3.
Copper Recovery Predicted by PRCC-informed GPR (H2SO4 = 3, Temperature = 80 °C, NaClO = 0 and NaClO3 = 0).
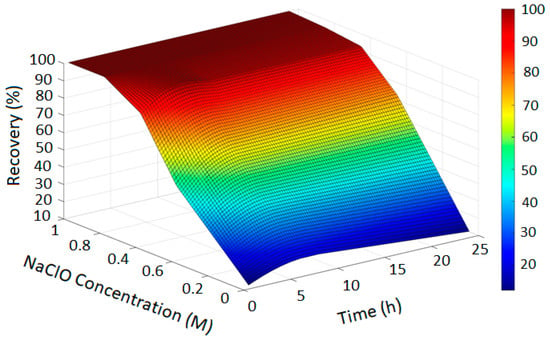
Figure 4.
Copper Recovery Predicted by PRCC-informed GPR (H2SO4 = 3, Temperature = 80 °C, NaCl = 0 and NaClO3 = 0).
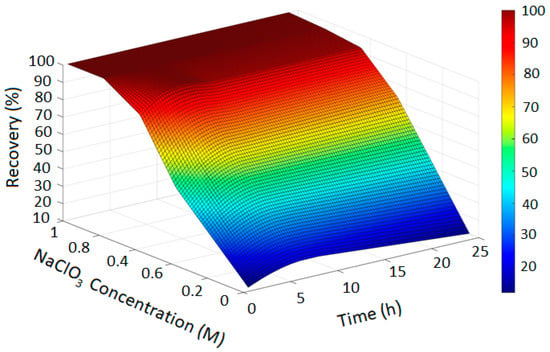
Figure 5.
Copper Recovery Predicted by PRCC-informed GPR (H2SO4 = 3, Temperature = 80 °C, NaCl = 0 and NaClO = 0).
In Figure 2, the GPR surface shows a monotonic increase in recovery with leaching time, reflecting baseline dissolution behavior. In Figure 3, the surface reveals a moderate enhancement in extraction at elevated NaCl levels, consistent with the limited oxidizing capacity of chloride alone. Figure 4 demonstrates a pronounced improvement in recovery induced by hypochlorite, while Figure 5 exhibits near-complete dissolution due to the strong oxidizing effect of chlorate. Taken together, these figures demonstrate that the PRCC-informed GPR model reliably captures the nonlinear leaching behavior under different oxidizing conditions and provides meaningful predictive insight into enhanced dissolution mechanisms.
3.4. Chemical Reactions and Phase Analysis
To clarify the chemical mechanisms of chalcopyrite leaching in sulfuric acid and in the presence of chloride-based oxidizing agents, the principal reactions can be represented as follows (Equations (2)–(7)). These reactions show the formation of soluble copper–chloride complexes and sulfur-containing products, explaining the observed enhancement of leaching.
The experimental results indicate a distinct shift in dissolution kinetics upon the addition of chloride oxidants, which can be explained by the solution redox potential (Eh) and the stability of chlorine species. During the leaching experiments, the solution redox potential was monitored and remained within a narrow range of 420–440 mV (vs. Ag/AgCl). In sulfate-chloride systems, this potential range is critical as it favors the stability of strong oxidants, specifically hypochlorous acid (HOCl) and molecular chlorine (Cl2), rather than the less reactive chloride ions (Cl−). The presence of these active species is pivotal for the oxidative breakdown of the chalcopyrite lattice.
For the NaClO system, hypochlorite exists in equilibrium with hypochlorous acid (ClO− + H+ ⟷ HClO). At the acidic pH values used in this study, HClO becomes the dominant species. Being a much stronger oxidant than dissolved oxygen, HClO rapidly attacks the metal-sulfur bonds, preventing the formation of a continuous passivating sulfur layer and leading to the high recoveries observed within 2 h.
In the case of NaClO3, the unusually rapid and complete dissolution (100% in <30 min) is attributed to the thermodynamic instability of chlorate ions in hot concentrated sulfuric acid. Under these conditions, chlorate (ClO−3) decomposes to form intermediate perchlorate or reactive chlorine dioxide/oxygen species, significantly raising the local oxidative potential. This aggressive oxidizing environment not only dissolves copper but also rapidly oxidizes the iron component of chalcopyrite. The XRD analysis of leach residues supports this, showing a significant reduction in chalcopyrite peaks and the appearance of iron-containing phases (such as jarosite or iron oxides/hydroxides), indicating that iron precipitates out of the solution once the mineral structure is destroyed.
Regarding secondary reactions, the high acidity and elevated temperature promote the evolution of chlorine gas (2HCl + [O]→H2O + Cl2↑), particularly in the NaClO3 system. While the evolution of Cl2 gas represents a reagent loss, it confirms the generation of highly oxidative conditions necessary for the extreme kinetics observed. Furthermore, the formation of soluble copper–chloride complexes (CuCln(n−2)) stabilizes Cu+ ions in solution, preventing reprecipitation and shifting the equilibrium towards complete dissolution. Thus, the synergy between high Eh, active chlorine species generation, and complexation reactions explains the superior performance of chloride-based oxidants over pure sulfuric acid leaching. Mass balance calculations confirmed that the total copper recovered in the leachate and that remaining in the solid residue matched the initial copper content within the experimental error (<2%), validating the reported recovery values in Table 8 and Table 9.
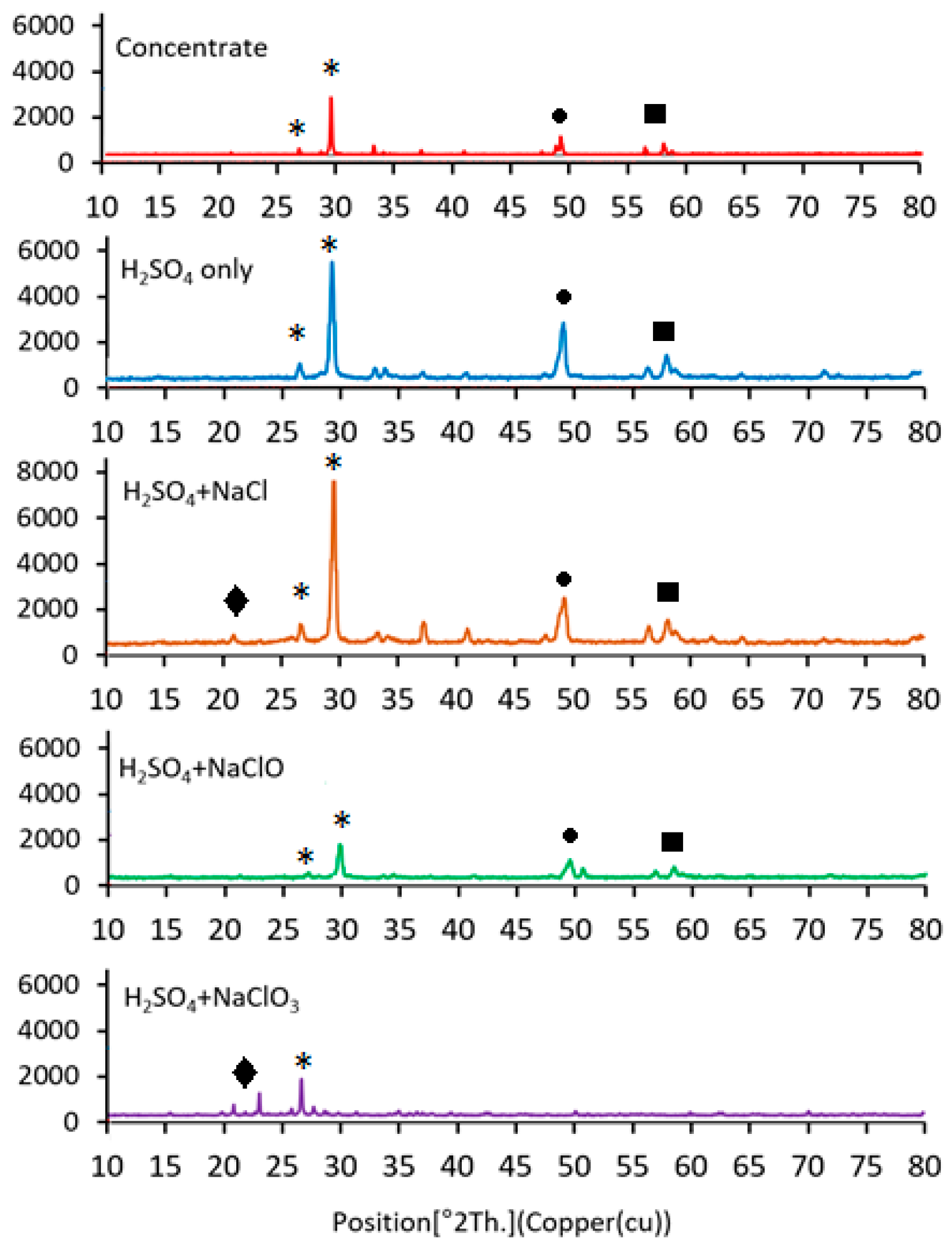
X-ray diffraction (XRD) analyses were performed on both the initial chalcopyrite concentrate and the leach residues to examine phase changes after leaching with different chloride-based additives (Figure 6). The initial concentrate showed characteristic peaks of chalcopyrite and pyrite. After leaching, the intensity of chalcopyrite peaks decreased, indicating partial mineral dissolution.
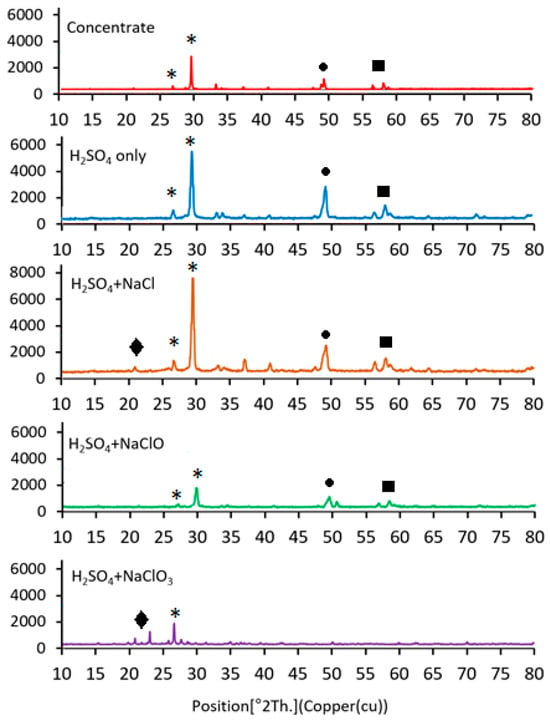
Figure 6.
XRD patterns of initial and leached residues illustrating phase changes due to additive-assisted leaching (Symbols: star: chalcopyrite, circle: pyrite, square: minor sulfide phases and rhomb: sulfur).
Treatment with NaCl led to a modest reduction in chalcopyrite peaks, while NaClO caused a more pronounced decrease, consistent with the faster dissolution observed experimentally. The most substantial effect was seen with NaClO3, where chalcopyrite peaks nearly disappeared, suggesting near-complete dissolution. An increase in sulfur-related phases was also observed, indicating accumulation of residual sulfur on the solid surface.
These XRD observations support the experimental trends in copper recovery and provide indirect evidence of surface modifications during leaching with chloride-based oxidants, without making strong mechanistic claims.
3.5. Industrial Feasibility and Environmental Considerations
While NaClO and NaClO3 demonstrate high technical efficiency, their industrial scalability requires careful consideration of economic and environmental factors. Economically, these chemical oxidants are more expensive than conventional agents like ferric iron. However, the drastic reduction in leaching time could offset operational costs by increasing throughput, potentially making the process viable for high-grade concentrates or specific pre-treatment stages. Regarding safety and operations, the generation of chlorine gas in acidic media necessitates closed reactor systems equipped with alkaline scrubbers to manage off-gas emissions and corrosion. Environmentally, the effluent contains high chloride levels and residual oxidants, requiring robust treatment protocols, such as neutralization and advanced filtration, to ensure compliance with discharge standards. Furthermore, the management of soluble copper-chloro complexes is crucial for downstream purification. In conclusion, while effective, industrial application depends on optimizing reagent consumption and integrating efficient gas and effluent treatment systems. Future research should focus on hybrid approaches, such as electrochemical regeneration or lower-dose combinations, to enhance sustainability.
4. Conclusions
This study successfully developed a PRCC-informed Gaussian Process Regression (GPR) model to predict copper recovery during advanced chalcopyrite leaching. The integration of sensitivity analysis results into the kernel design enabled the model to capture complex nonlinear interactions among critical process variables, such as acid concentration, temperature, and oxidizing agents, leading to highly accurate predictions. The model outperformed traditional regression methods, demonstrating its robustness and exceptional predictive accuracy across all tested conditions.
The results indicate that NaClO3 was the most effective chloride-based oxidizing agent, achieving near-complete copper recovery (100%) under optimal conditions. Temperature and reaction time were identified as key factors that significantly enhance copper recovery, while oxidizing agent strength and sulfuric acid concentration were found to govern overall dissolution efficiency. Specifically, the model predicted that recovery improves progressively with increasing temperature and leaching time, confirming the influence of thermal and temporal conditions on chalcopyrite dissolution kinetics.
The PRCC-informed GPR model was validated through excellent agreement with experimental data, achieving an RMSE of 4.0028 and an R2 of 0.98803, outperforming other models such as Support Vector Machine Regression (SVMR) and Ensemble Regression (ER). The model’s ability to predict copper recovery under untested conditions further establishes its reliability and versatility.
These findings highlight the effectiveness of the PRCC-guided GPR framework in optimizing chalcopyrite leaching processes, providing a reliable, interpretable tool for improving copper recovery. This model serves as a powerful tool for industrial applications, enabling better process design and operational optimization for hydrometallurgical copper extraction. The insights gained from this study offer valuable guidance for future experimental and industrial work, contributing to more efficient and sustainable copper recovery strategies.
It should be noted that the experimental dataset used in this study was obtained from a single, well-characterized chalcopyrite concentrate to ensure controlled and reproducible leaching conditions. Although chalcopyrite from different geological origins may exhibit variations in mineralogy and texture, the PRCC-informed GPR framework developed here is process-oriented and scalable. By incorporating additional experimental data from concentrates of different grades and origins, the model can be readily retrained and extended, providing a flexible and transferable tool for broader hydrometallurgical applications.
While the influence of oxidant concentration on copper recovery was systematically studied, a detailed economic assessment of chloride oxidant usage was not conducted. Future studies should evaluate the balance between reagent dosage, cost, and industrial applicability to guide practical implementation.
Author Contributions
Conceptualization, M.A., A.E.K. and B.S.; methodology, M.A., A.E.K. and B.S.; software, H.S., A.E.K.; validation, M.A., A.E.K. and B.S.; formal analysis, M.A.; investigation, M.A. and B.S.; resources, M.A. and H.S.; data curation, M.A. and A.E.K.; writing—original draft preparation, B.S. and A.E.K.; writing—review and editing, B.S. and A.E.K.; visualization, M.A., B.S., A.E.K. and H.S.; supervision, M.A., B.S.; project administration, M.A., B.S.; funding acquisition, H.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors gratefully acknowledge the Mineral Processing Department, Mining and Materials Engineering Faculty, Tarbiat Modares University, Tehran, Iran, for providing laboratory facilities and technical support required to conduct the experimental work of this study. The authors also acknowledge the Department of Computing Sciences, The University of Scranton, Pennsylvania, USA, for the financial support and computational assistance that contributed to the development of the modeling and data analysis components of this research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Winarko, R.; Dreisinger, D.B.; Miura, A.; Yuken, F.; Liu, W. Modelling the scale-up of the iodine-assisted chalcopyrite leaching in ferric sulfate media from reactors to columns and simulating heap operations. Hydrometallurgy 2023, 222, 106203. [Google Scholar] [CrossRef]
- Watling, H.R. Chalcopyrite hydrometallurgy at atmospheric pressure: 1. Review of acidic sulfate, sulfate–chloride and sulfate–nitrate process options. Hydrometallurgy 2013, 140, 163–180. [Google Scholar] [CrossRef]
- Bartlett, R.W. Simulation of ore heap leaching using deterministic models. Hydrometallurgy 1992, 29, 231–260. [Google Scholar] [CrossRef]
- Vilcáez, J.; Suto, K.; Inoue, C. Modeling the auto-thermal performance of a thermophilic bioleaching heap employing mesophilic and thermophilic microbes. Hydrometallurgy 2008, 94, 82–92. [Google Scholar] [CrossRef]
- Dixon, D.G.; Afewu, K.I. HeapSim2D—A 2D axisymmetric model of heap leaching under drip emitters I: Hydrology and solute transport. Miner. Eng. 2022, 183, 107555. [Google Scholar] [CrossRef]
- Jumbo-Pacheco, P.; Townley, B.; Kelm, U.; Vargas, T. Chloride leaching of copper ores: Influence of NaCl on gangue dissolution and its effect on copper leaching kinetics. Miner. Eng. 2025, 233, 109640. [Google Scholar] [CrossRef]
- Machaca, D.M.C.; Botelho, A.B.J.; Carvalho, T.C.; Tenório, J.A.S.; Espinosa, D.C.R. Use of hydrogen peroxide as oxidizing agent in chalcopyrite leaching: A review. Metals 2025, 15, 531. [Google Scholar] [CrossRef]
- Machaca, D.M.C.; Botelho, A.B.J.; Carvalho, T.C.; Tenório, J.A.S.; Espinosa, D.C.R. Hydrometallurgical processing of chalcopyrite: A review of leaching techniques. Int. J. Miner. Metall. Mater. 2024, 31, 2537–2555. [Google Scholar] [CrossRef]
- Miao, X.; Narsilio, G.A.; Wu, A.; Yang, B. A 3D dual pore-system leaching model. Part 1: Study on fluid flow. Hydrometallurgy 2016, 167, 136–149. [Google Scholar] [CrossRef]
- Winarko, R.; Dreisinger, D.B.; Miura, A.; Fukano, Y.; Liu, W. Iodine-assisted chalcopyrite leaching in ferric sulfate media: Kinetic study under fully controlled redox potential and pH. Hydrometallurgy 2022, 208, 105797. [Google Scholar] [CrossRef]
- Dixon, D.G.; Lizama, H.M. Design, construction, and modelling of bioheaps. In Biomining Technologies; Johnson, D.B., Bryan, C.G., Schlömann, M., Roberto, F.F., Eds.; Springer: Cham, Switzerland, 2023; pp. 15–40. [Google Scholar] [CrossRef]
- Ghadamgahi, S.M.; Babakhani, A.; Barati Darband, G.; Shalchian, H.; Behmadi, R. Solvometallurgical properties of choline chloride-based deep eutectic solvents for copper extraction from chalcopyrite: Optimization and analysis. Mining 2025, 5, 8. [Google Scholar] [CrossRef]
- Narsilio, G.A.; Buzzi, O.; Fityus, S.; Yun, T.S.; Smith, D.W. Upscaling of Navier–Stokes equations in porous media: Theoretical, numerical and experimental approach. Comput. Geotech. 2009, 36, 1200–1206. [Google Scholar] [CrossRef]
- Ochoa-Tapia, J.A.; Whitaker, S. Momentum transfer at the boundary between a porous medium and a homogeneous fluid I: Theoretical development. Int. J. Heat Mass Transf. 1995, 38, 2635–2646. [Google Scholar] [CrossRef]
- Xie, S.; Yu, Y.; Xie, Y.; Chen, X.; Tang, Z. Overview and prospects of modeling and optimal control for non-ferrous metallurgical processes and mineral processing. Green Smart Min. Eng. 2025, 2, 440–458. [Google Scholar] [CrossRef]
- Petersen, J.; Dixon, D.G. Thermophilic heap leaching of chalcopyrite concentrate. Miner. Eng. 2002, 15, 777–785. [Google Scholar] [CrossRef]
- Gericke, M.; Pinches, A.; van Rooyen, J.V. Bioleaching of a chalcopyrite concentrate using an extremely thermophilic culture. Int. J. Miner. Process. 2001, 62, 243–255. [Google Scholar] [CrossRef]
- Bouffard, S.C.; Dixon, D.G. Modeling pyrite bioleaching in isothermal test columns with the HeapSim model. Hydrometallurgy 2007, 88, 225–240. [Google Scholar] [CrossRef]
- Bennett, C.; Cross, M.; Croft, T.; Uhrie, J.; Green, C.; Gebhardt, J. A comprehensive copper stockpile leach model: Background and model formulation. In Hydrometallurgy 2003—Volume 1: Leaching and Solution Purification; Young, C., Alfantazi, A., Anderson, C., Dreisinger, D., Harris, B., James, A., Eds.; TMS: Warrendale, PA, USA, 2003; pp. 315–328. [Google Scholar]
- Bouffard, S.C. Understanding the Heap Biooxidation of Sulfidic Refractory Gold Ores. Ph.D. Thesis, University of British Columbia, Vancouver, BC, Canada, 2003. [Google Scholar]
- Saldaña, M.; Neira, P.; Gallegos, S.; Salinas-Rodríguez, E.; Pérez-Rey, I.; Toro, N. Mineral leaching modeling through machine learning algorithms—A review. Front. Earth Sci. 2022, 10, 816751. [Google Scholar] [CrossRef]
- Kang, J.-K.; Cho, K.-H.; Kim, S.-B.; Choi, N.-C. Artificial neural network modeling for prediction of dynamic changes in solution from bioleaching by indigenous acidophilic bacteria. Appl. Sci. 2020, 10, 7569. [Google Scholar] [CrossRef]
- Bagdaulet, K.; Azatbekuly, N.; Aibagarov, S.; Amangeldy, B.; Koizhanova, A.; Magomedov, D. Predicting industrial copper hydrometallurgy output with deep learning approach using data augmentation. Minerals 2025, 15, 702. [Google Scholar] [CrossRef]
- Behnajady, B.; Najafi, M.; Karimi, S. A new approach to direct chemical leaching of Sungun chalcopyrite concentrate via green deep eutectic solvent choline chloride–p-toluenesulfonic acid and MD simulation. J. Taiwan Inst. Chem. Eng. 2025, 172, 106118. [Google Scholar] [CrossRef]
- Saldaña, M.; Gálvez, E.; Robles, P.; Castillo, J.; Toro, N. Copper mineral leaching mathematical models—A review. Materials 2023, 15, 1757. [Google Scholar] [CrossRef] [PubMed]
- Winarko, R.; Dreisinger, D.B.; Miura, A.; Tokoro, C.; Liu, W. Kinetic modelling of chalcopyrite leaching assisted by iodine in ferric sulfate media. Hydrometallurgy 2020, 197, 105481. [Google Scholar] [CrossRef]
- Dutrizac, J.E. The leaching of sulfide minerals in chloride media. Hydrometallurgy 1992, 29, 1–45. [Google Scholar] [CrossRef]
- Arslan, F.; Kangal, M.O.; Bulut, G.; Gül, A. Leaching of massive rich copper ore with acidified ferric chloride leaching. Miner. Process. Extr. Metall. Rev. 2004, 25, 143–158. [Google Scholar] [CrossRef]
- Kangal, O.; Bulut, G.; Gül, A.; Ozaksoy, G.; Arslan, F. Cupric chloride leaching of sulphidic copper ores. In Proceedings of the VI SHMMT/XVIII ENTMH, Rio de Janeiro, Brazil, 2001; pp. 445–450. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.