Abstract
Black shale represents a distinctive and critical mineral resource in China, harboring approximately 90% of the nation’s recoverable vanadium reserves while concurrently containing abundant strategic metal elements such as nickel and molybdenum. However, the complex occurrence states of vanadium, nickel, and molybdenum within black shale pose significant challenges to their efficient extraction. Conventional metallurgical processes—including calcination-leaching and hydrometallurgical leaching—primarily target vanadium recovery, exhibiting limited efficiency for comprehensive utilization of valuable metals. Against the backdrop of green metallurgy and carbon neutrality objectives, bioleaching techniques have garnered extensive research attention. This study developed a specialized consortium of ore-leaching microorganisms, designated WZ-Q, tailored to the mineralogical characteristics of black shale, demonstrating effective leaching capabilities for vanadium, nickel, and molybdenum. Furthermore, the enhancing effects of Fe2+, elemental sulfur (S0), and pyrite as energy substrates on bioleaching efficiency were investigated. Upon incorporating these energy materials, maximum leaching efficiencies reached 66.5% for vanadium, 82.5% for nickel, and 29.7% for molybdenum. Analysis through leaching process monitoring and multi-characterization of both raw ore and residues revealed that supplemental energy substrates intensify shifts in solution potential and pH, thereby promoting elemental oxidation and mineral decomposition. Nevertheless, critical impediments to leaching efficiency include the encapsulation of target elements within silicate matrices and incomplete dissolution of oxidized species. Subsequent research should prioritize methodologies to intensify silicate mineral dissolution and enhance the release of oxidized compounds during microbial leaching processes.
1. Introduction
Vanadium stands as one of China’s 55 strategic emerging industry minerals recognized in the Classification of Strategic Emerging Industries [1], renowned as the “modern industrial catalyst” for its exceptionally comprehensive properties. Due to the enormous reserves of black shale in China, which also contain considerable amounts of vanadium, statistics show that the V2O5 reserves in black shale exceed 100 million tons, accounting for nearly 90% of China’s vanadium resources. However, the grade is usually low, the processing difficulty is high, and there are significant regional differences, which have not been effectively utilized [2,3]. In addition to vanadium, black shale also contains key metal elements such as nickel and molybdenum [4,5]. Therefore, developing a green, clean, low-cost, and widely applicable comprehensive recovery process for valuable metals in black shale is of great significance for the utilization of shale resources in China.
The traditional methods for vanadium recovery in black shale mainly involve roasting leaching [6,7] or enhanced whole hydrometallurgical leaching with leaching aids [8,9], and there are many problems that need to be solved: (1) high energy consumption (requiring 700–900 °C roasting temperature), (2) high acid consumption (25–40% sulfuric acid relative to ore weight), and (3) environmental pollution from SO2 emissions, acidic wastewater, and hazardous residues. In addition, nickel and molybdenum in black shale are usually not effectively utilized, and only high-grade shale minerals are subjected to beneficiation for nickel and molybdenum enrichment before further recovery [10,11,12].
The fundamental objective of metal recovery lies in oxidizing and transforming vanadium, nickel, and molybdenum from diverse occurrence states into soluble forms for subsequent concentration and separation. Against the backdrop of China’s 14th Five-Year Plan advocating “enhanced mineral resource conservation, green mining initiatives, and sustainable mine development”—coupled with carbon neutrality imperatives for nonferrous metallurgy—innovative leaching technologies are emerging to reduce environmental impact, lower energy consumption, improve vanadium recovery rates, and realize comprehensive polymetallic extraction. In the context of green metallurgy and “carbon peak and carbon neutrality”, new roasting processes, whole hydrometallurgical leaching vanadium extraction processes, and biohydrometallurgy processes have been extensively studied. These methods have improved the leaching rate and leaching percentage of vanadium in vanadium-bearing mica by promoting solid–liquid reactions, breaking covalent bonds in vanadium-bearing mica, and oxidizing low valence vanadium to promote dissolution. In the roasting process, the emergence of new sodium roasting additives [13] has reduced environmental pollution, low-temperature sulfuric acid roasting [14] has reduced energy consumption, while microwave roasting [15], blank roasting and fluidized bed suspension oxidation roasting [16] have further reduced pollutant emissions and fully utilized the original heat value in black shale, which is currently widely used. The whole hydrometallurgical leaching process promotes the dissolution of vanadium-containing mica by adding leaching aids, mainly including fluorides and oxidants [9,17]. This process eliminates the roasting section, resulting in a shorter process and a higher leaching percentage. However, the introduction of fluoride also leads to environmental hazards and the complexity of subsequent processes.
Biohydrometallurgy has been extensively applied for leaching low-grade ores, tailings, and wastes of copper, nickel, cobalt, zinc, uranium, and gold [18]. Research on black shale bioleaching, however, remains limited and focuses on three domains: (1) Utilizing autotrophic bacteria like Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans to achieve 62–69% vanadium leaching after 25 days [19]; (2) Bioleaching and chemical leaching combined leaching to reduce acid and energy consumption, while increasing the leaching rate of vanadium in black shale [20]; (3) Bioleaching by using heterotrophic microorganisms silicate bacteria (e.g., Bacillus mucilaginosus), the highest vanadium leaching percentage can reach 67% [21,22,23,24].
Adding energy substrates required for microbial survival and activities is an important way to strengthen microbial leaching. Because the content of energy substances necessary for the growth of leaching microorganisms, such as Fe2+, S0, in black shale is not sufficient, adding a certain amount of energy substances to the system will promote the bioleaching of black shale. For instance: Fe2+ supplementation during A. ferrooxidans leaching elevated vanadium recovery to 48.92% versus 13.81% by sulfuric acid alone after 30 days [19]. Similarly, adding 8 g/L sulfur powder boosted vanadium extraction to 74.22% [25].
In this study, the bioleaching experiment of vanadium-nickel-molybdenum-bearing black shale was carried out by using the medium-temperature bacteria obtained by breeding and domestication as leaching microorganisms, and the effect of energy substrates on the leaching efficiency of vanadium, nickel, and molybdenum in black shale was studied by adding different types of energy substrates as strengthening agents. Complementary SEM-EDS, FTIR, and XPS analyses of residues elucidate elemental dissolution mechanisms and microbial leaching pathways, establishing a foundation for enhanced polymetallic bioleaching.
2. Materials and Methods
2.1. Experimental Minerals
The main elemental composition of the black shale used in this study is detailed in Table 1. The ore exhibits relatively low grades of vanadium (0.71%), nickel (0.44%), and molybdenum (0.12%), alongside trace elements including iron, sulfur, magnesium, silicon, and aluminum.

Table 1.
Principal Elemental Composition of Black Shale.
The main elemental composition of the molybdenite concentrate used in this study is detailed in Table 2.

Table 2.
Principal Elemental Composition of Molybdenite Concentrate.
2.2. Experimental Bacteria
The bacteria selected in this experiment are medium temperature bacteria designated WZ-Y, comprising Acidithiobacillus and Leptospirillum, cultivated and mineral-adapted in 9 K medium (The medium is divided into two parts: (a) 0 K medium (NH4)2SO4 3.0 g, MgSO4·7H2O 0.5 g, K2HPO4 0.5 g, KCl 0.1 g, Ca(NO3)2 0.01 g, distilled water 1000 mL, (b) FeSO4·7H2O 44.6 g). Adaptation was achieved through sequential subculturing in 250 mL flasks containing 90 mL medium and 10 mL inoculum. Black shale was added progressively to attain a pulp density of 10%, with transfers initiated when the redox potential exceeded 600 mV. System pH and redox potential were monitored daily to assess microbial activity.
In view of the toxic effect of molybdenum on bacteria, bacteria were further adapted to higher molybdenum concentrations through domestication. The initial molybdenum concentration was set at 20 mg/L, and then increased by 280 mg/L during the transfer until the target molybdenum tolerance was achieved. This adapted bacteria, designated WZ-Q, served as the leaching bacteria in all subsequent experiments.
2.3. Experimental Conditions
Bioleaching experiment of black shale was conducted under standardized conditions: temperature 30 °C, initial pH 1.4, pulp density 10% (w/v), inoculum volume 10% (v/v), and agitation at 150 rpm using 0 K medium. To evaluate energy substrate enhancement, treatments included: Control (no additives), Fe2+ (added as FeSO4), Elemental sulfur (S0), Pyrite (FeS2).
Triplicate experiments were performed per condition (Table 3).

Table 3.
Bioleaching Conditions with Energy Substrate Augmentation.
Bioleaching experiment of molybdenite concentrate was conducted with the moderately thermophilic consortium WZ-Q. The experiments were carried out in 250 mL conical flasks under the following conditions: 10% pulp density (w/v), inoculum volume 10% (v/v), initial pH 1.4, and 9 K medium.
2.4. Analytical Methods
pH and Eh were respectively tracked using an Orion 3 Star pH meter (Thermo Fisher Scientific, Waltham, MA, USA) and a PC-350 potentiometer (SUNTEX, Taipei, China). Post-leaching residues were digested and analyzed via inductively coupled plasma optical emission spectrometry (ICP-OES). Vanadium, nickel, and molybdenum leaching efficiencies were calculated from ICP-OES data.
The elemental phase associations in black shale raw ore and leaching residues were quantified via stepwise chemical dissolution, and the detailed experimental procedures were carried out as follows:
- Vanadium phases
Black shale raw ore or leaching residue was leached in a boiling water bath with 30% HCl to dissolve vanadium in iron oxides and clay minerals. After solid–liquid separation, the residue was designed to be leached with 80% HF to extract vanadium from vanadium-bearing mica. The remaining residue after re-separation contained vanadium bound to tourmaline and garnet.
- 2.
- Nickel phases
Raw ore or leaching residue was leached with water and calcite for 5 min to dissolve nickel sulfate. After separation, the residue was leached with dilute H2SO4-HF-CuSO4 solution to extract nickel from silicates (Cu2+ forms a copper sulfide film to protect nickel sulfide). The residual fraction after separation contained nickel in sulfide minerals.
- 3.
- Molybdenum phases
Raw ore or leaching residue was leached in boiling Na2CO3 solution to dissolve molybdenum oxides. After separation, the residue with filter paper was ashed and melted to determine molybdenum in sulfide form.
X-ray photoelectron spectroscopy (XPS) was used to analyze the valence changes in vanadium, nickel, and molybdenum in the original ore sample and the bioleaching residue. Fourier transform infrared spectroscopy (FT-IR) was used to analyze the chemical bond changes in the ore sample. Scanning electron microscopy and energy dispersive spectroscopy (SEM-EDS) were used to observe the morphological changes in the mineral surface, and the elemental composition of the specific area on the sample surface was analyzed.
3. Experimental Procedure
3.1. Microorganism Adaptation and Monitoring
A mixed consortium of iron- and sulfur-oxidizing microorganisms was selected as the initial inoculum, aligned with the mineralogical characteristics of black shale. To mitigate molybdenum-derived microbial inhibition [26], the consortium underwent sequential adaptation to increasing molybdenum concentrations.
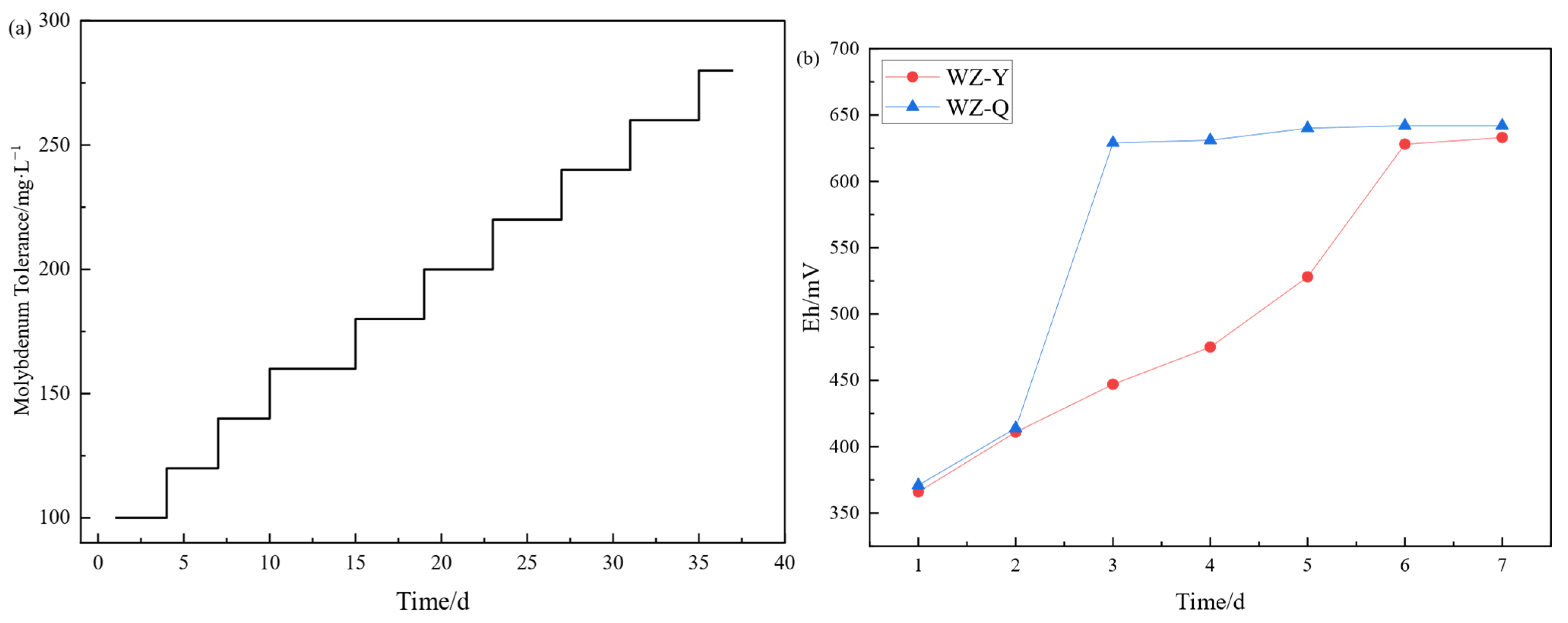
The domestication situation is shown in Figure 1. After 37 days of step domestication of molybdenum content, a strain with a tolerance of 280 mg/L molybdenum content is obtained, and its tolerance is much greater than the theoretical molybdenum content of the leaching system. As shown in the figure, when conducting the microbial culture experiment under the molybdenum containing system, it was found that the potential growth of the non domesticated strain system was slower than that of the domesticated strain, which proved that molybdenum had an inhibitory effect on the redox ability of microorganisms, and the molybdenum resistant strain had been able to adapt to the molybdenum containing environment, and could be used for the bioleaching of black shale.
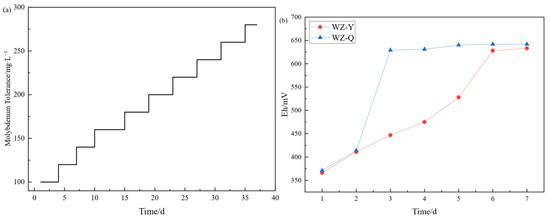
Figure 1.
(a) Progressive Acclimatization of Microbial Molybdenum Tolerance; (b) Comparative Analysis of Microbial Proliferation in Non-Acclimated (WZ-Y) versus Acclimated (WZ-Q) Systems.
The molybdenum-adapted consortium was inoculated into culture medium containing progressively increased black shale pulp densities. Commencing at 1% mineral concentration, pulp density was escalated in 1% increments every third day until reaching 10%, enabling stepwise bacterial acclimation to the mineral-laden environment. This mineral-adapted consortium, designated as WZ-Q, served as the specialized hyper-efficient bioleaching agent for black shale under mesophilic conditions in subsequent leaching experiments.
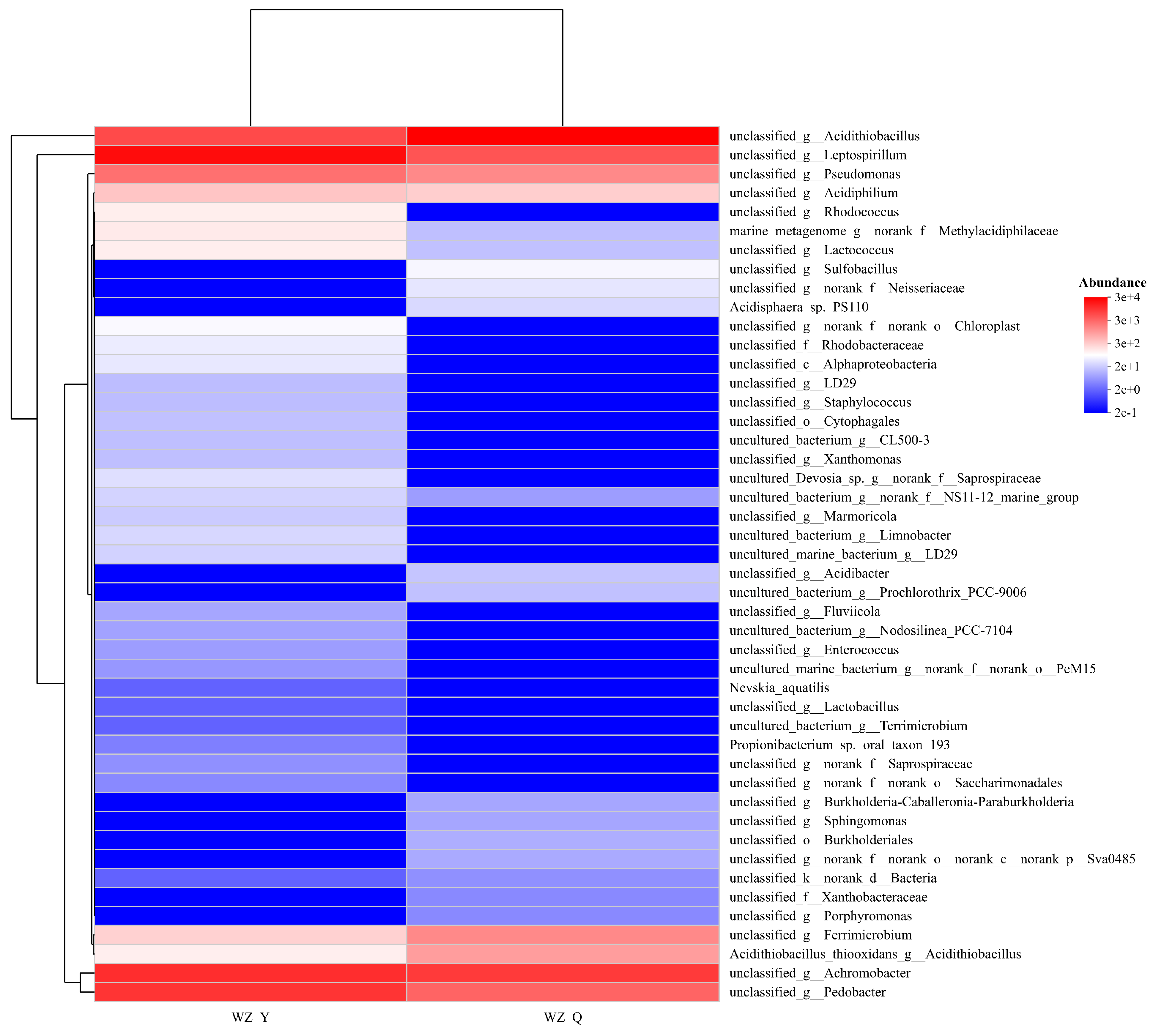
Comparative high-throughput sequencing analyses of pre- and post-adaptation microbial communities (Figure 2) revealed a post-adaptation consortium dominated by Leptospirillum and Acidithiobacillus, with significant presence of Achromobacter and Acinetobacter, alongside minor proportions of Pseudomonas and Ferrimicrobium. Iron and sulfur constituents serve as essential energy substrates, enhancing bioleaching efficacy. Figure 2 further demonstrates increased proportions of Acidithiobacillus and sulfur-oxidizing bacteria following adaptation. Functional annotations indicate enriched metabolic pathways, including energy transduction, biosynthesis, and transport of secondary metabolites—functions mechanistically associated with optimized mineral bioleaching performance.
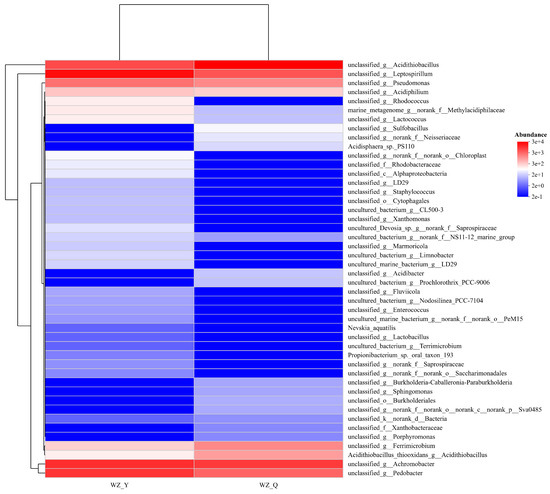
Figure 2.
Community heatmap of Bioleaching Microorganisms in Black Shale (WZ-Y: Initial Microorganism Community, WZ-Q: Domesticated microbial community).
3.2. Evolution of Potential and pH Under Enhanced Energy Substrate Regimes
The changes in potential and pH of the leaching system reflect the metabolic activity of microorganisms during the leaching process. Iron-oxidizing microorganisms catalyze the oxidation of Fe2+ to Fe3+, concomitant with elevated redox potential. Concurrently, sulfur-oxidizing species mediate sulfide-to-sulfate conversion, generating acidic byproducts that depress system pH.
Compounding these biologically driven processes, mineral dissolution releases alkaline gangue constituents—oxide ores such as calcium and magnesium—that neutralize acidity through proton exchange. This counterbalance modulates both electrochemical parameters throughout leaching. Figure 3 delineates the complex interplay between microbially mediated redox acceleration and mineralogically induced acid consumption, as manifested through temporal fluctuations in potential and pH.
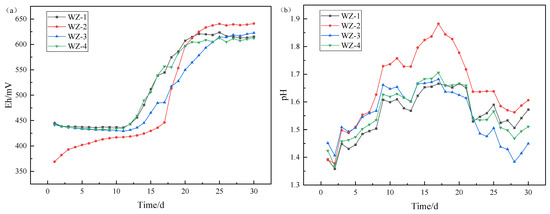
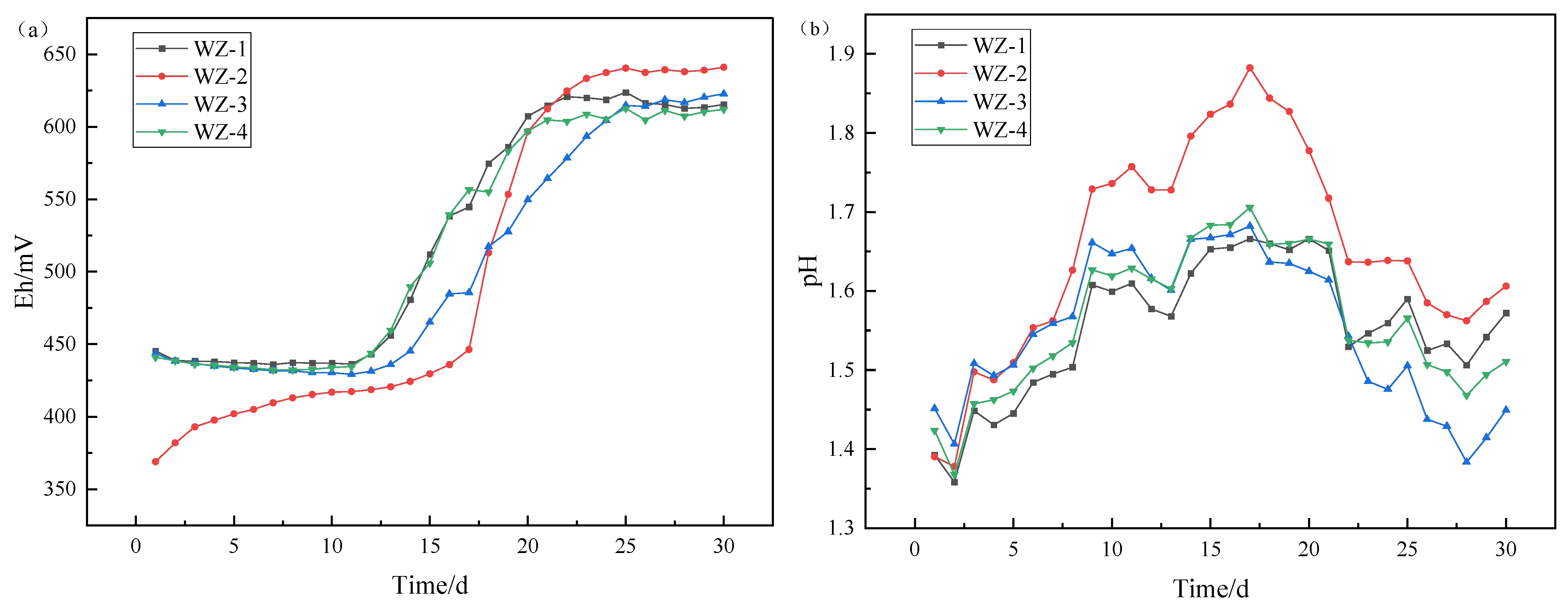
Figure 3.
Changes of (a) Eh/(b) pH under different energy substances.
Detection of system potential revealed that the initial potential of the WZ-2 system (with added Fe2+) is lower. The reason is that the ratio of Fe2+/Fe3+ is the main factor controlling the system potential, while the addition of Fe2+ causes a larger ratio of Fe2+/Fe3+ in the solution, resulting in a decrease in potential. During the leaching process, the system potential continues to show an upward trend, and the potential at the end of leaching is higher than that of other groups, indicating that iron-oxidizing bacteria gradually oxidize Fe2+ to Fe3+. The potential of the group WZ-1 (without additives), WZ-3 (with the addition of S0), and WZ-4 (with the addition of pyrite) changes slowly at the beginning of leaching. This is because the initial solution does not contain iron ions, and the subsequent increase in potential is due to the dissolution and oxidation of iron in the mineral. By the end of leaching, the iron in the mineral is almost completely dissolved and oxidized.
The pH of each leaching system showed a trend of first increasing and then decreasing. At the stage of pH rising, the main reaction was the acid consumption reaction of oxidized ore (containing calcium and magnesium), and reached equilibrium after a certain time of leaching. At the later stage of leaching, the decrease in pH value was mainly due to the acid production of sulfur-oxidizing bacteria. Due to the addition of additional sulfur sources in the WZ-3 system, microorganisms produce more acids, which is reflected in the fact that the pH value of this group is lower than that of other groups. Since pyrite contains more sulfur than black shale, the pH of group WZ-4 is lower than that of the control group and ferrous group.
3.3. Bioleaching Dynamics of Molybdenite
Given the predominance of molybdenite as the primary host of molybdenum in black shale, targeted bioleaching experiments were conducted on molybdenite concentrate obtained through mineral processing.
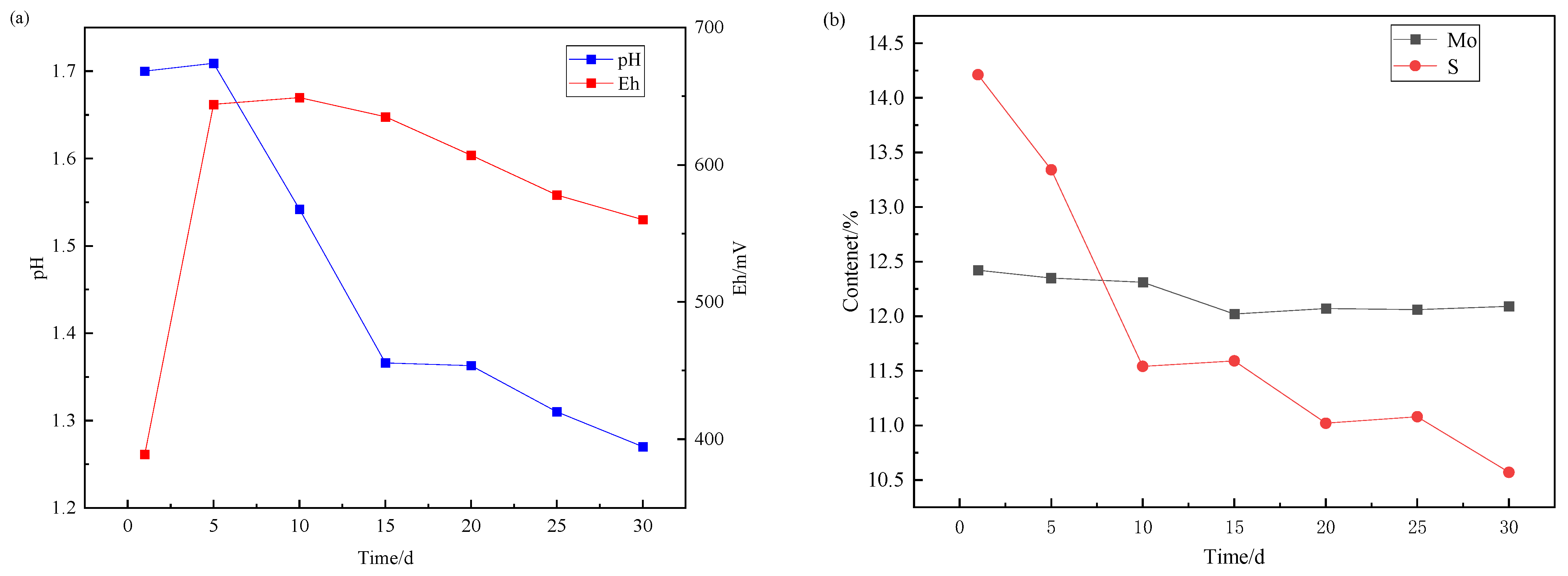
Over a 30-day leaching, samples from one flask were collected at 5-day intervals for vacuum filtration and residue analysis. This methodology enabled phase transformation tracking of molybdenum-bearing species in leached residues across progressive stages. Concurrent monitoring of potential, pH evolution, and aqueous molybdenum/sulfur concentrations was performed throughout the experiment, with comprehensive temporal profiles illustrated in Figure 4.
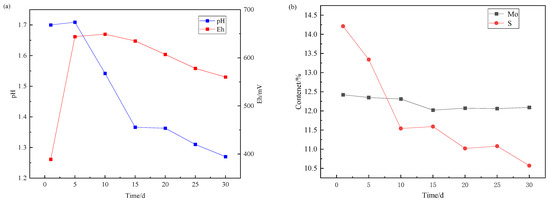
Figure 4.
Change of (a) Eh/pH and (b) element content of leaching residue during molybdenite leaching.
The absence of acid-consuming oxidized minerals in the molybdenite concentrate yielded sustained acidification throughout leaching. Microbial catalysis by iron-oxidizing bacteria triggered rapid anodic polarization during initial stages; however, progressive cathodic drift emerged in later phases with conspicuous ochreous jarosite precipitates. During the bioleaching, the content of molybdenum decreased slightly, while the content of sulfur decreased significantly, which proved that the transformation of molybdenum from sulfide state to oxidation state occurred in the bioleaching process, but the leaching of molybdenum oxide did not proceed further.
4. Results and Discussion
4.1. Leaching Efficiency Evolution with Energy Substrates
During bioleaching, pH and redox potential are the main factors affecting the extraction efficiency. Therefore, it is of great significance to investigate the correlation between the leaching percentage of vanadium, nickel, and molybdenum and the potential and pH for the optimization of the enhanced bioleaching of black shale, and to preliminarily explore the behavior of vanadium, nickel, and molybdenum in the process of bioleaching.
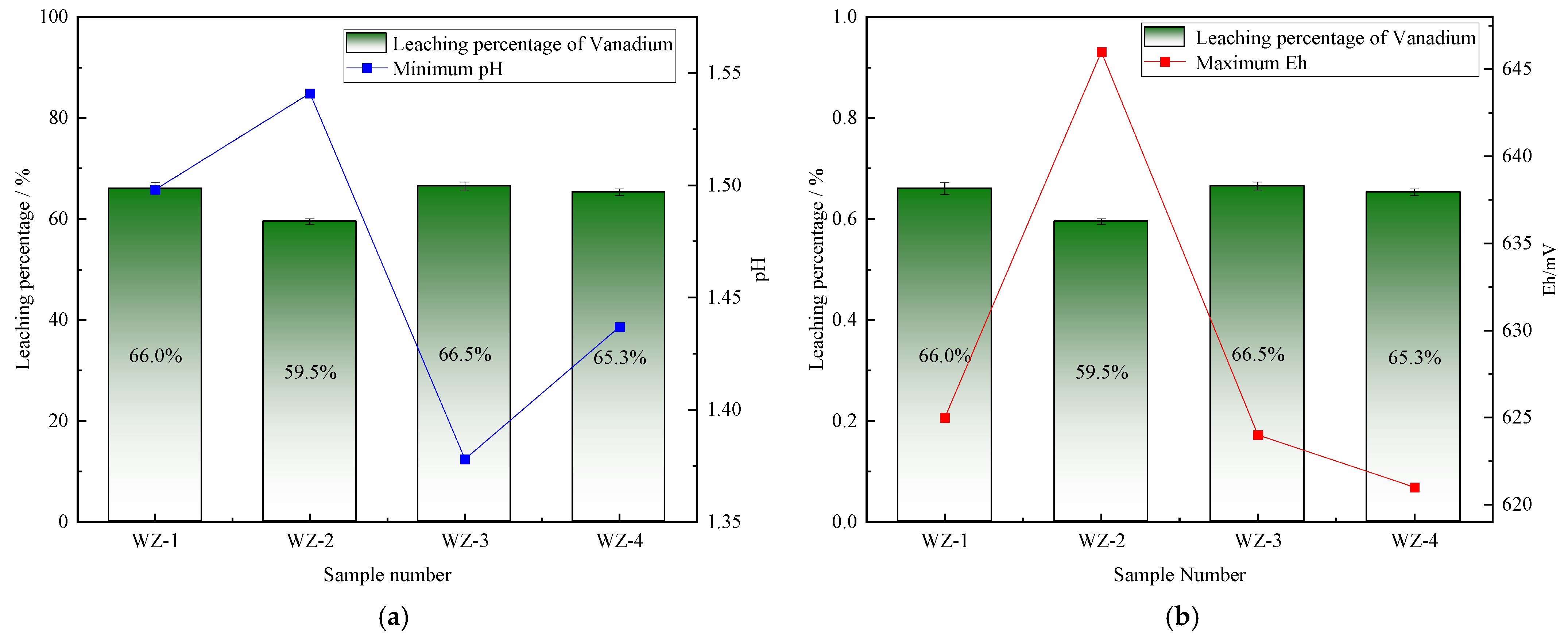
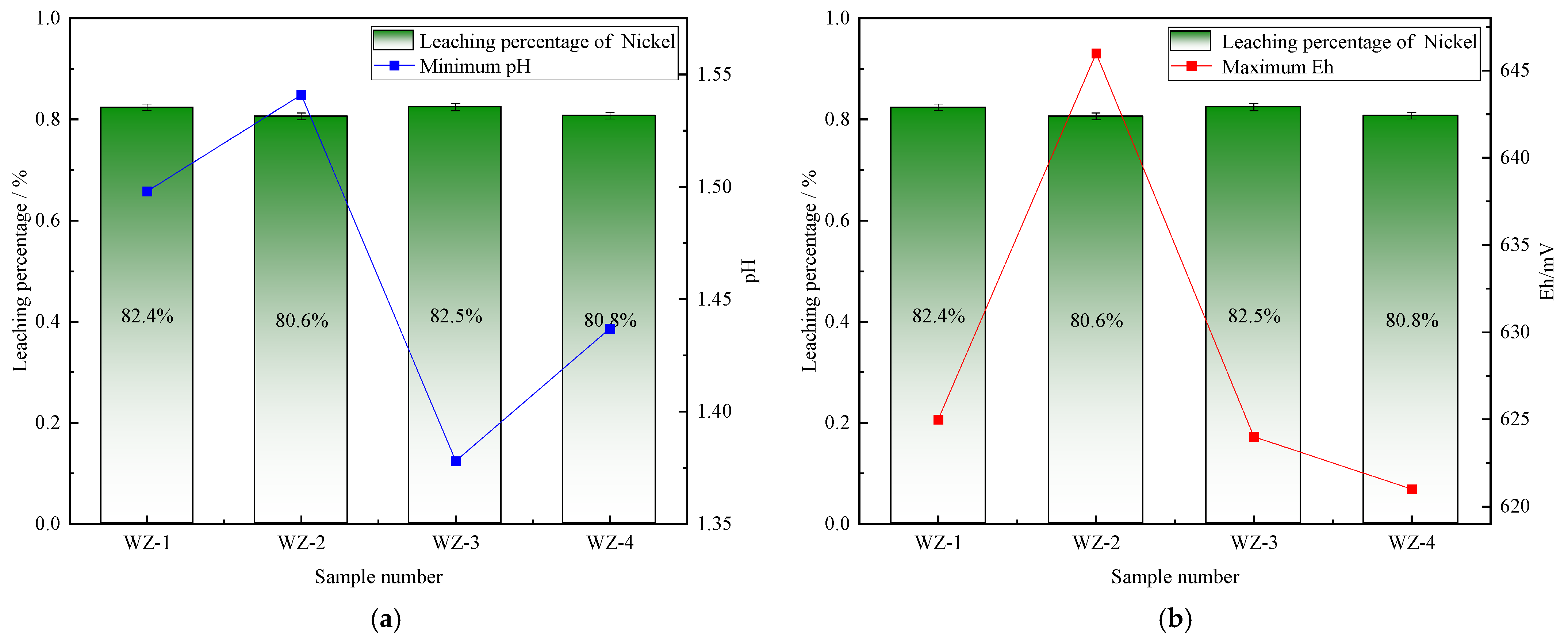
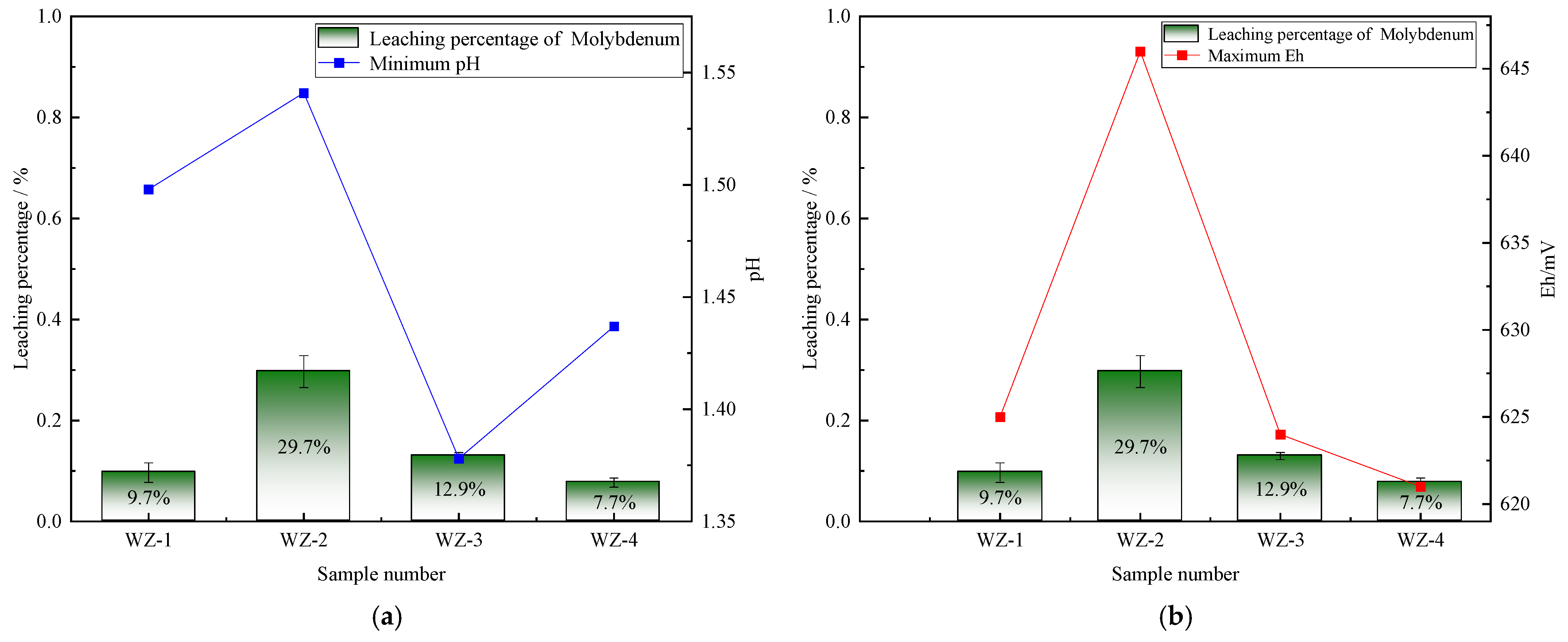
The extraction of vanadium in black shale is mainly affected by pH, and the leaching percentage of vanadium is higher under lower pH conditions. As shown in Figure 5, the vanadium leaching percentage of the group added with S0 increased slightly, peaking at 66.5%. Under the action of sulfur-oxidizing bacteria, the pH decreased to 1.39, indicating that the addition of S0 is more favorable for vanadium extraction, while the pH of the group added with Fe2+ is the highest, but the vanadium extraction yield shows a certain reduction. The dissolution of nickel is less affected by experimental conditions. As depicted in Figure 6, it remains above 80% under the strengthening effect of different energy substances. The extraction of molybdenum in black shale is mainly affected by the potential. As illustrated in Figure 7, the molybdenum leaching percentage of the group added with Fe2+ is significantly higher than that of other groups, reaching up to 29.7%. Combined with the potential data, the redox potential of this group exceeded 640 mV, confirming that the high potential exerts a significant promoting effect on molybdenum dissolution. A comparison of the extraction yields of the three elements revealed that those of vanadium, nickel, and molybdenum in the pyrite group were lower than those in the control group. Unreacted pyrite was observed in the leaching residues, suggesting that microorganisms primarily acted on the exposed pyrite and thus inhibited the dissolution of target metals from black shale.
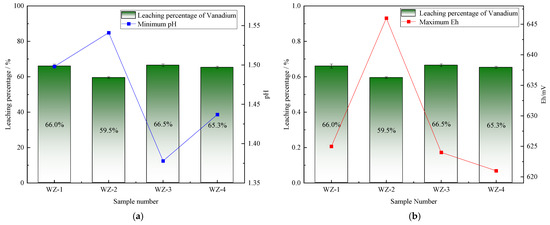
Figure 5.
Relationship between vanadium leaching percentage and changes in (a) pH and (b) Eh.
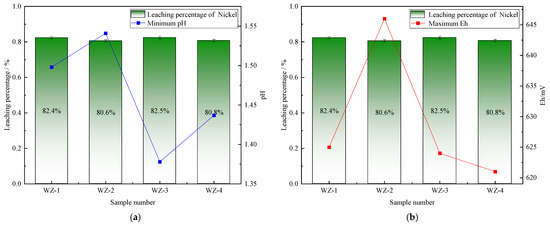
Figure 6.
Relationship between nickel leaching percentage and changes in (a) pH and (b) Eh.
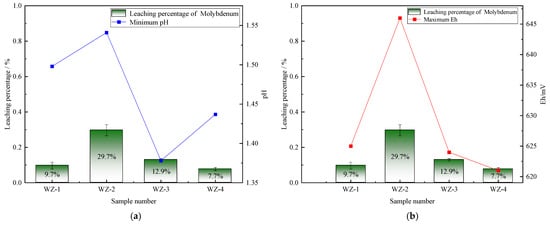
Figure 7.
Relationship between molybdenum leaching percentage and changes in (a) pH and (b) Eh.
Collectively, reducing the pH of the leaching system and elevating the redox potential of the system are important ways to comprehensively and efficiently recover vanadium, nickel, and molybdenum from black shale. Augmenting oxidative capacity through tailored energy substrates—activating both iron-oxidizing bacteria and sulfur-oxidizers—serves as a determinant methodology. However, Fe2+ and S0 amendments exhibit pronounced counteractive proton-modulation behaviors. The simultaneous utilization of Fe2+, which facilitates high-potential molybdenite oxidation, and S0, which intensifies acid-promoted vanadium leaching, demonstrates substantial application potential.
It is the direction and focus of the follow-up research to explore the strengthening methods combining the effects of the two energy substances, reasonably regulate the potential and pH of the leaching system, and strengthen the bioleaching of vanadium, nickel, and molybdenum from black shale.
4.2. Phase Transformation of V-Ni-Mo in Black Shale Pre-/Post-Bioleaching
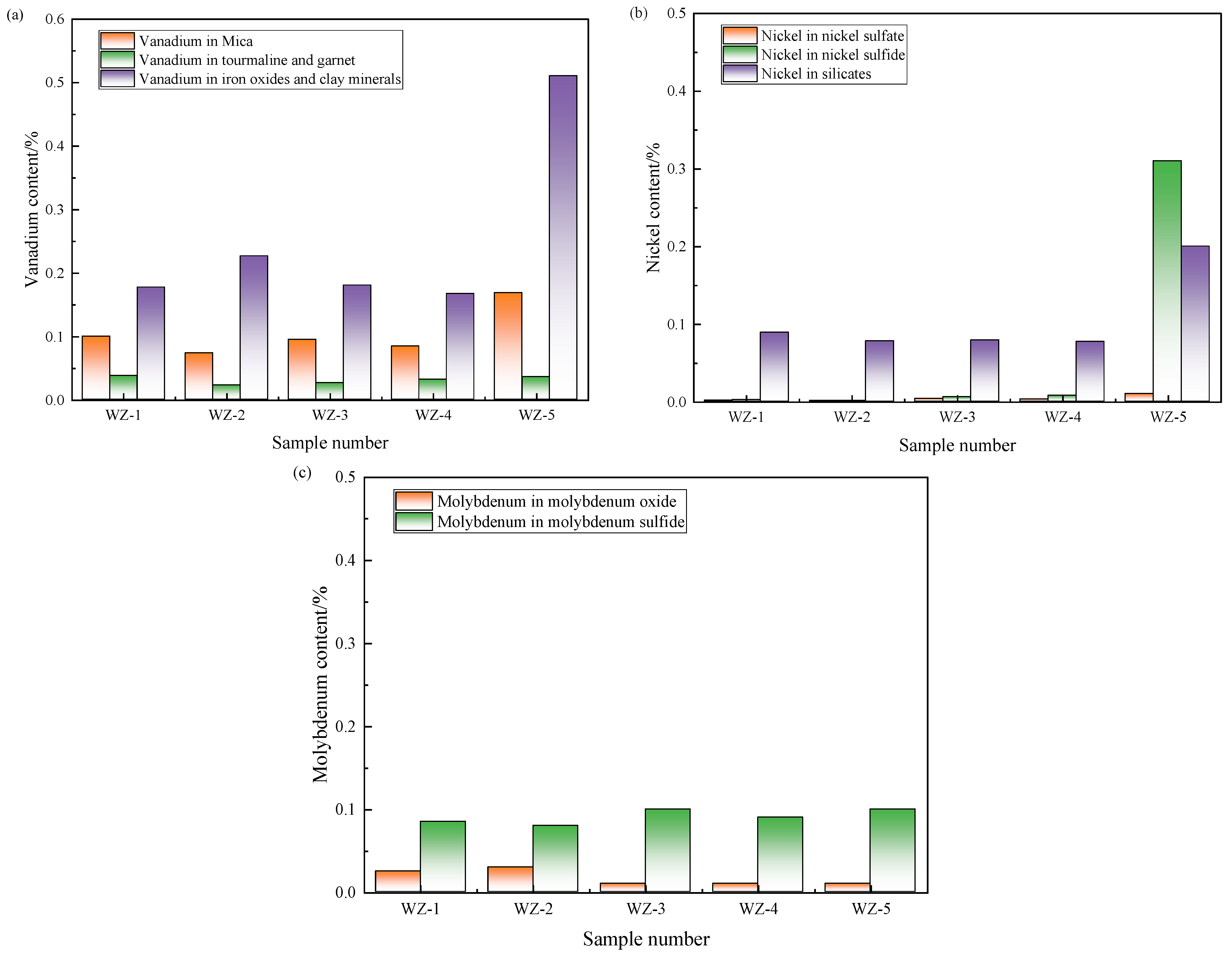
The occurrence states of each element in black shale are complex. By detecting the phase change in elements, we can explore the object and effect of microbial leaching, analyze the occurrence state of undissolved elements, and guide the optimization direction of the subsequent process. The phase analysis results of vanadium, nickel, and molybdenum in black shale raw ore and leaching residues are shown in Figure 8.
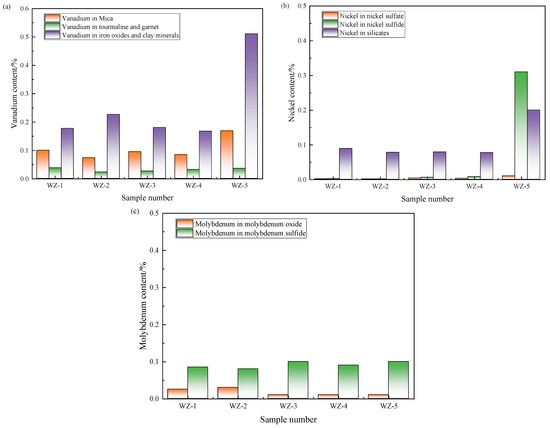
Figure 8.
Content changes of (a) vanadium, (b) nickel and (c) molybdenum in different phases before and after black shale leaching.
Vanadium predominantly occurs within vanadium-bearing mica, iron oxides, and clay minerals in black shale, with trace quantities hosted in other silicate phases. Experimental systems amended with Fe2+ exhibited significant depletion of vanadium in vanadium-mica lattices, while concurrently demonstrating elevated vanadium retention within iron oxides and clay matrices relative to the raw ore. This redistribution indicates that although vanadium in mica structures undergoes oxidative dissolution, its complete leaching into the leachate remains unachieved.
Nickel primarily resides in nickel sulfides and silicate forms. Across all leaching systems, sulfide-bound nickel phases (including nickel sulfides and sulfates) experienced near-total extraction, whereas nickel occlusion within silicate frameworks persisted as the principal determinant constraining overall recovery efficiency.
In raw black shale, molybdenum predominantly exists as molybdenite (MoS2). Following bioleaching under elevated electrochemical potentials, molybdenum speciation transitions toward oxidized molybdenum minerals. Nevertheless, these secondary molybdenum oxides exhibit recalcitrance to complete solubilization, presenting a critical kinetic barrier to comprehensive recovery.
4.3. SEM-EDS Characterization of Black Shale Raw Ore and Leached Residues
Conduct SEM-EDS testing on black shale to explore the actual occurrence state of various valuable metal elements in minerals.
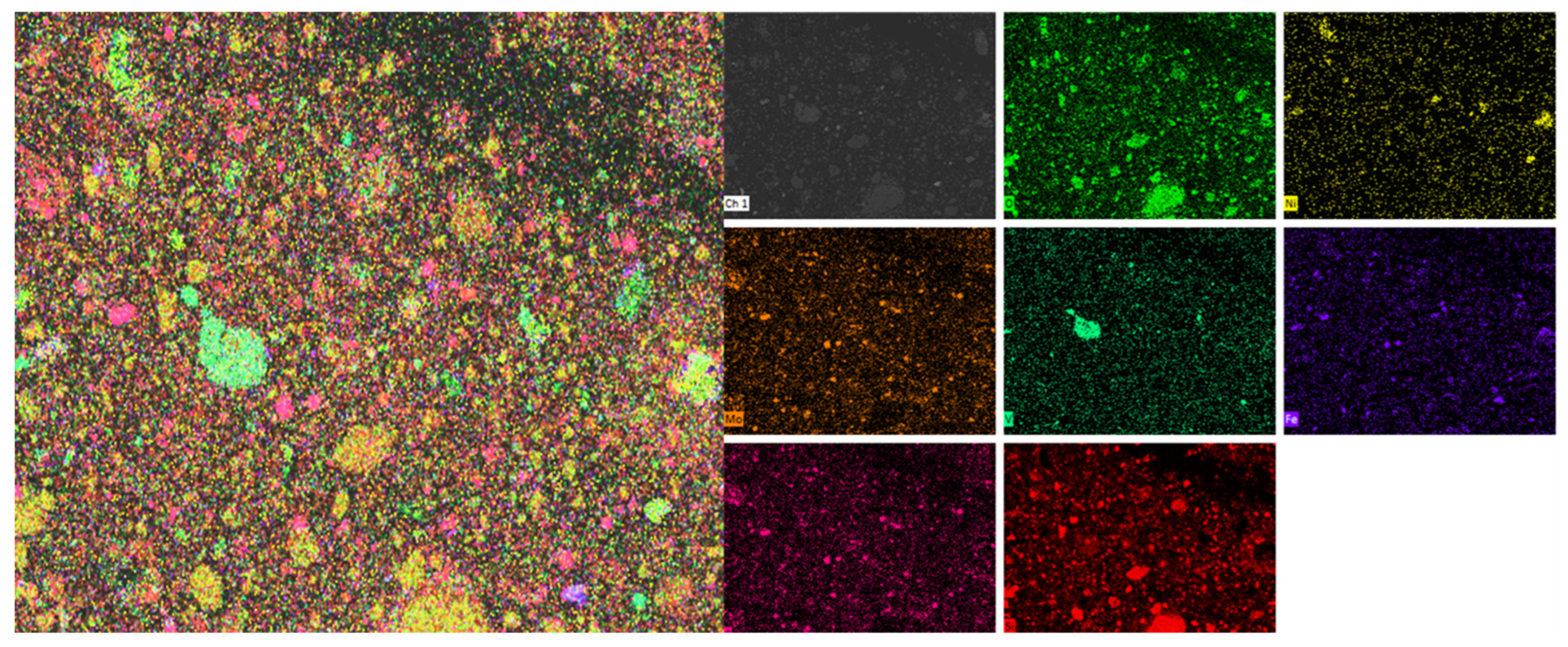
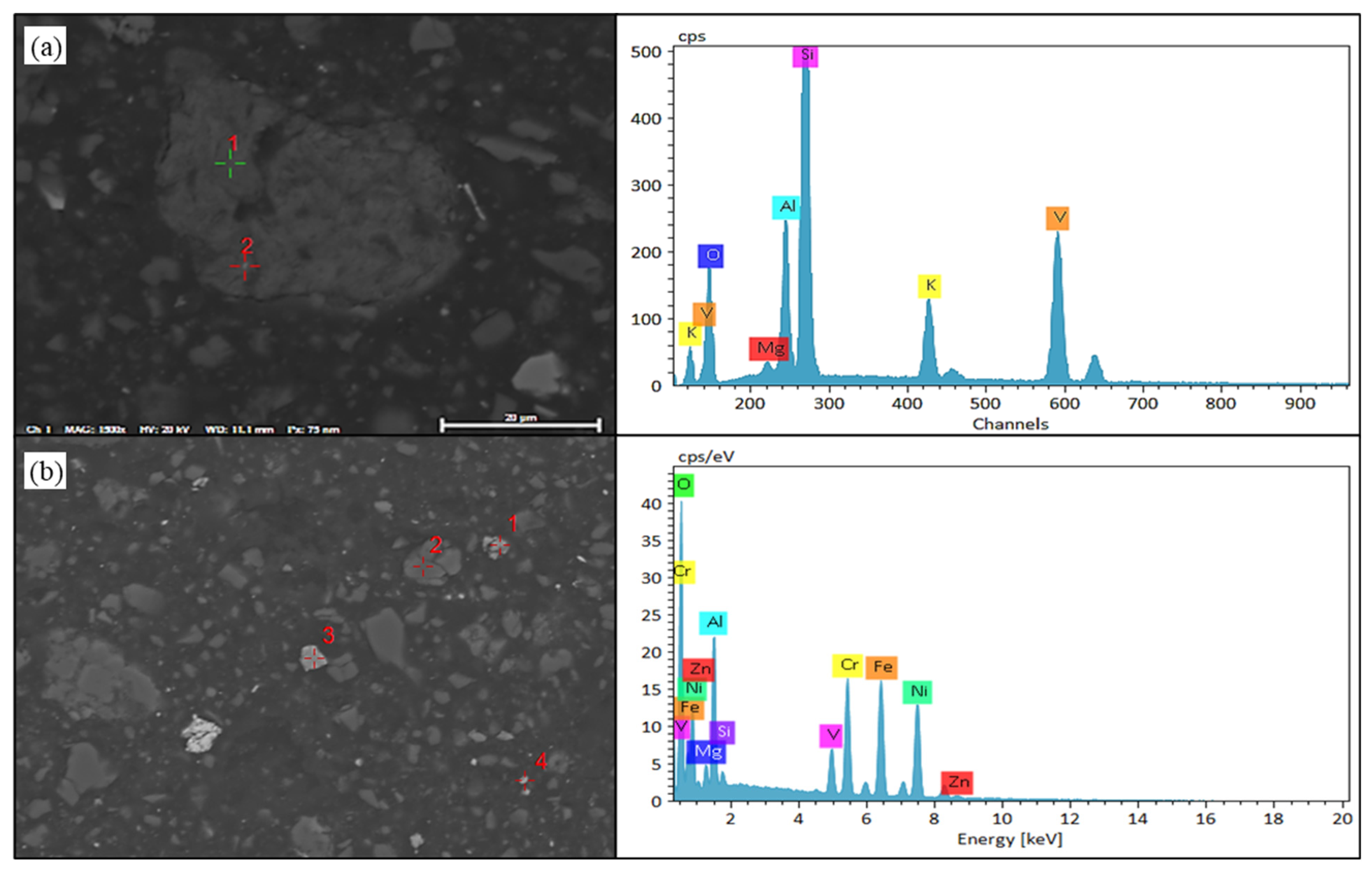
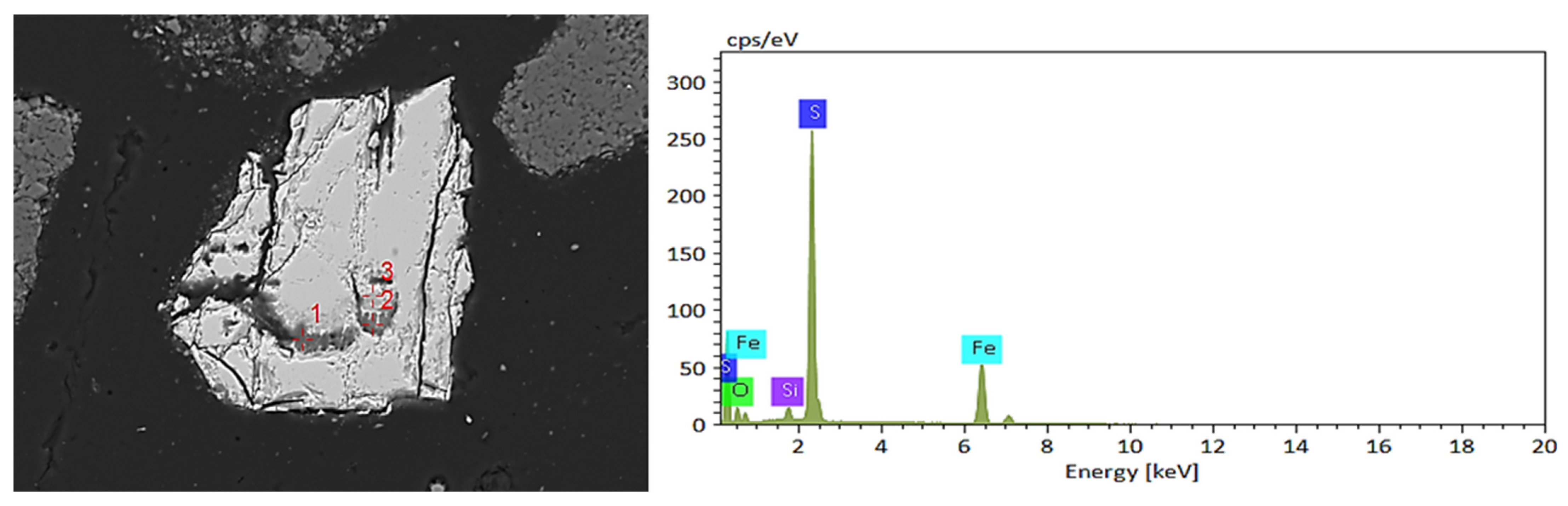
Firstly, the distribution areas of vanadium, nickel, molybdenum, silicon, iron, sulfur, oxygen, and other elements in the black shale ore were obtained by surface scanning, as shown in Figure 9. EDS detection was performed on the vanadium-containing areas in the leaching residue of black shale, and the results are shown in Figure 10. Under the microscope, it was observed that the surface of the black shale leaching residue appeared uneven after dissolution. EDS results showed that the vanadium in the leaching residue existed in the form of vanadium-bearing mica and was associated with iron oxides. The vanadium present in vanadium-bearing mica usually came in two forms: muscovite and phlogopite. The specific form of mica needs further testing; Vanadium compounds that are associated with iron oxides can coexist with various metal oxides (e.g., Fe and Ni) and are encapsulated or adsorbed by silicate minerals.
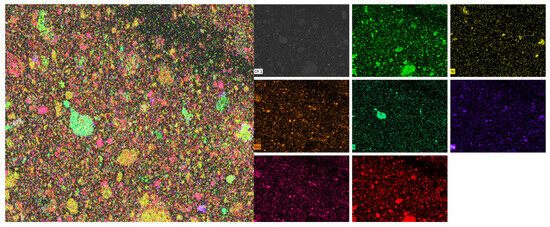
Figure 9.
Multi-element SEM-EDS results of black shale raw ore.
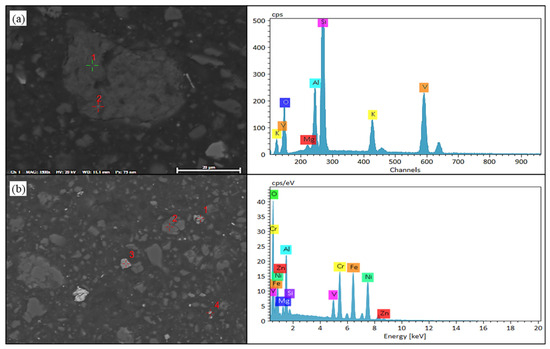
Figure 10.
SEM-EDS results of vanadium oxides in black shale biological leaching residue. (a) SEM image of vanadium-bearing shale and EDS result of Point 1. (b) SEM image of vanadium compounds and EDS result of Point 1.
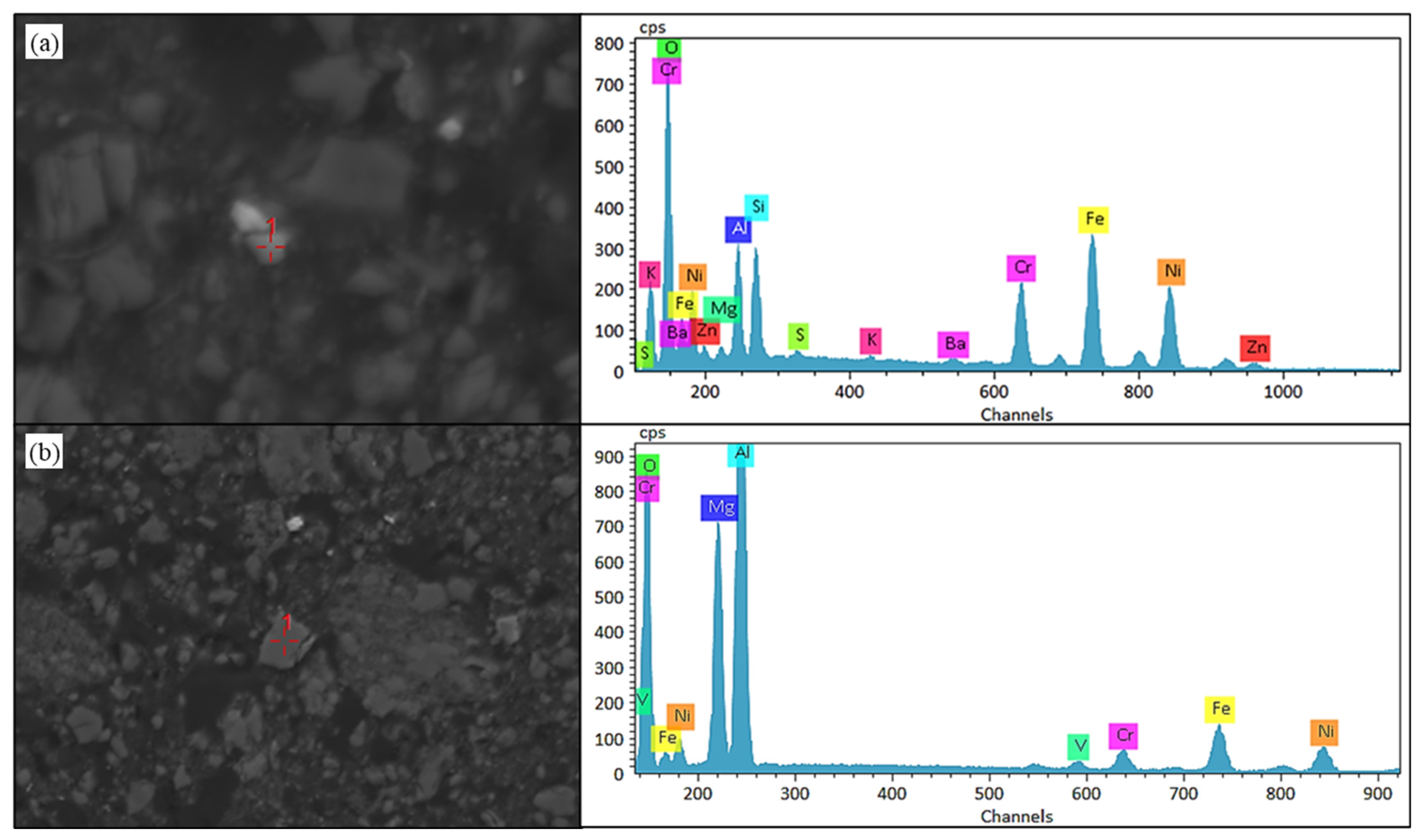
Microstructural evidence from Figure 11 indicates that nickel within black shale leach residues predominantly exists as oxide phases. Notably, substantial fractions of nickel are either adsorbed onto or encapsulated within silicate minerals alongside multimetallic oxides (zinc, iron, chromium), or coalesced into oxide polymers with magnesium and aluminum. This refractory occurrence state constitutes the primary constraint limiting further enhancement of nickel leaching efficiency.
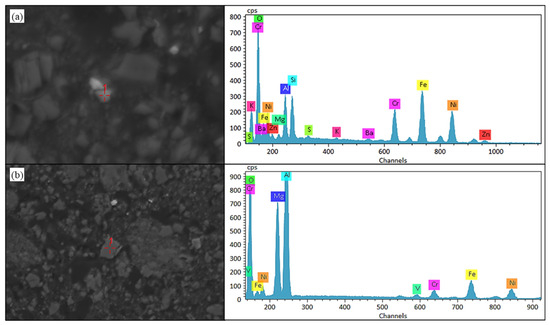
Figure 11.
SEM-EDS results of nickel and other oxides present in silicate minerals and the oxide polymerization in black shale biological leaching residue. (a) SEM image and EDS result of nickel in silicate minerals. (b) SEM image and EDS result of nickel coalesced into oxide polymers.
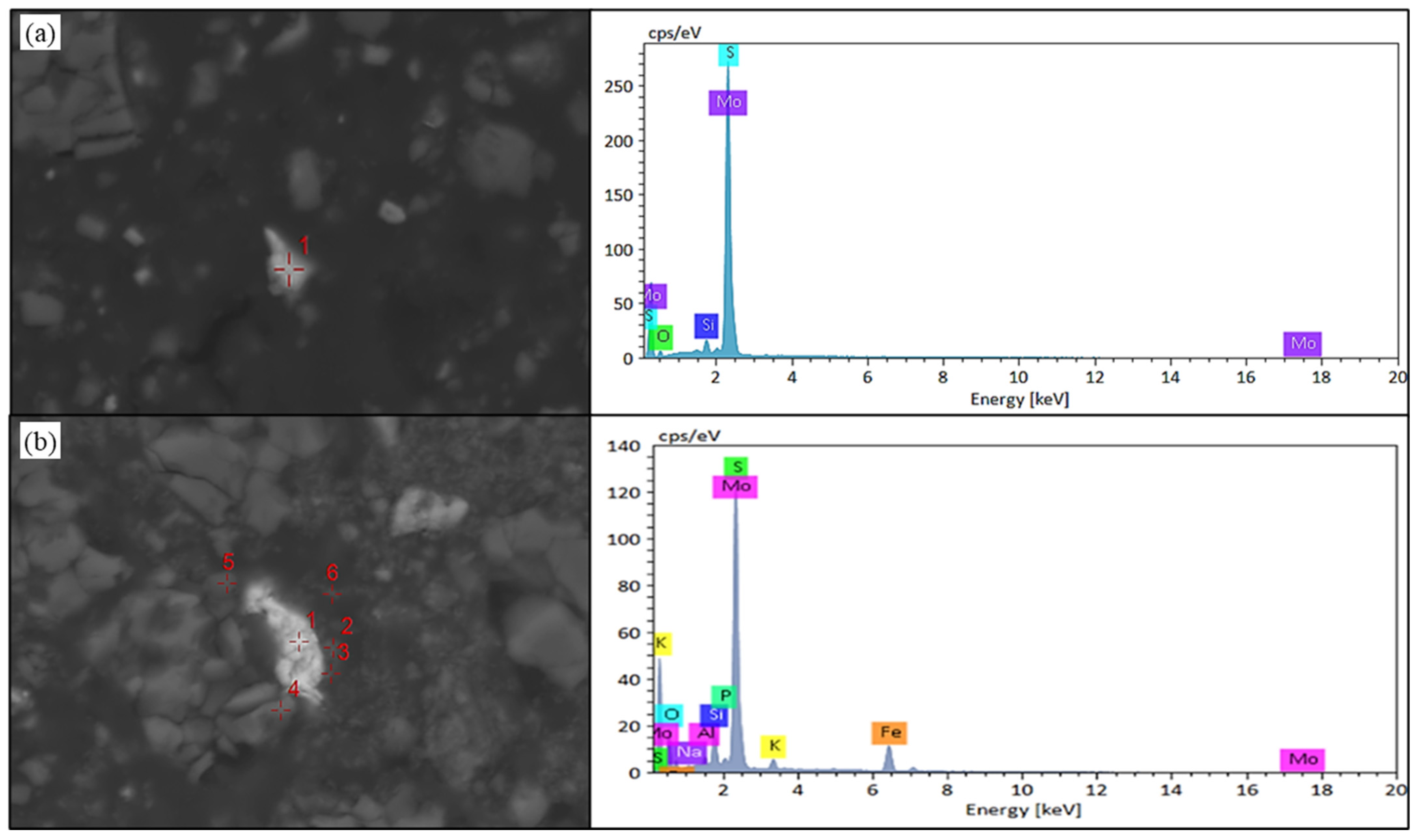
As illustrated in Figure 12, sulfidic molybdenum predominantly manifests as molybdenite within the raw ore—the principal occurrence state for this metal. Crucially, molybdenite assemblages in black shale substrates exhibit pronounced susceptibility to quartz encapsulation, thereby impeding microbial consortia or leaching solutions from interfacing with the mineral surface. This entrapment mechanism critically undermines molybdenum dissolution kinetics. Concomitantly, Figure 12 reveals the persistence of molybdenite as the dominant host phase in leach residues; however, peripheral microanalyses detected minor occurrences of iron molybdate and oxidized molybdenum species. Significantly, iron molybdate precipitates form tenacious coatings upon molybdenite crystallites, thus inhibiting subsequent oxidative degradation and leaching processes. This physical barrier detrimentally compromises the prospects for enhanced molybdenum recovery.
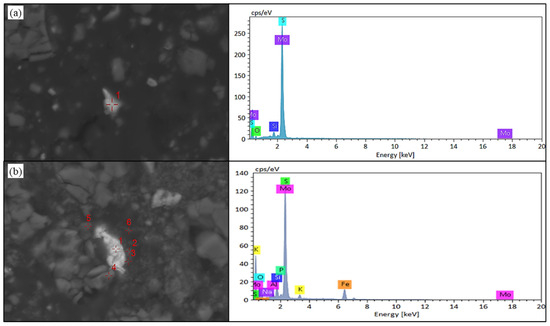
Figure 12.
SEM-EDS results of molybdenite in black shale raw ore and biological leaching residue. (a) SEM image and EDS result of molybdenite in black shale raw ore. (b) SEM image of molybdenum in the leaching residue of black shale, Point 1 is molybdenite, Point 2 and 3 are ferric molybdate, and Point 4–5 is molybdenum oxide.
Microstructural characterization of pyrite utilized as an energy substrate revealed, as demonstrated in Figure 13, that portions of the added pyrite exhibited negligible bio-oxidative degradation. Moreover, EDS analyses confirmed the presence of trace concentrations of jarosite species and iron oxide residues within adjacent matrices.
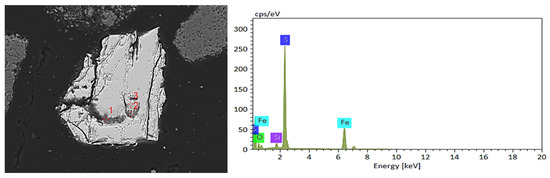
Figure 13.
SEM image of the addition of pyrite in black shale bioleaching residue and EDS result of Point 1.
4.4. Analysis of Roscoelite Structure and V, Ni, Mo Speciation
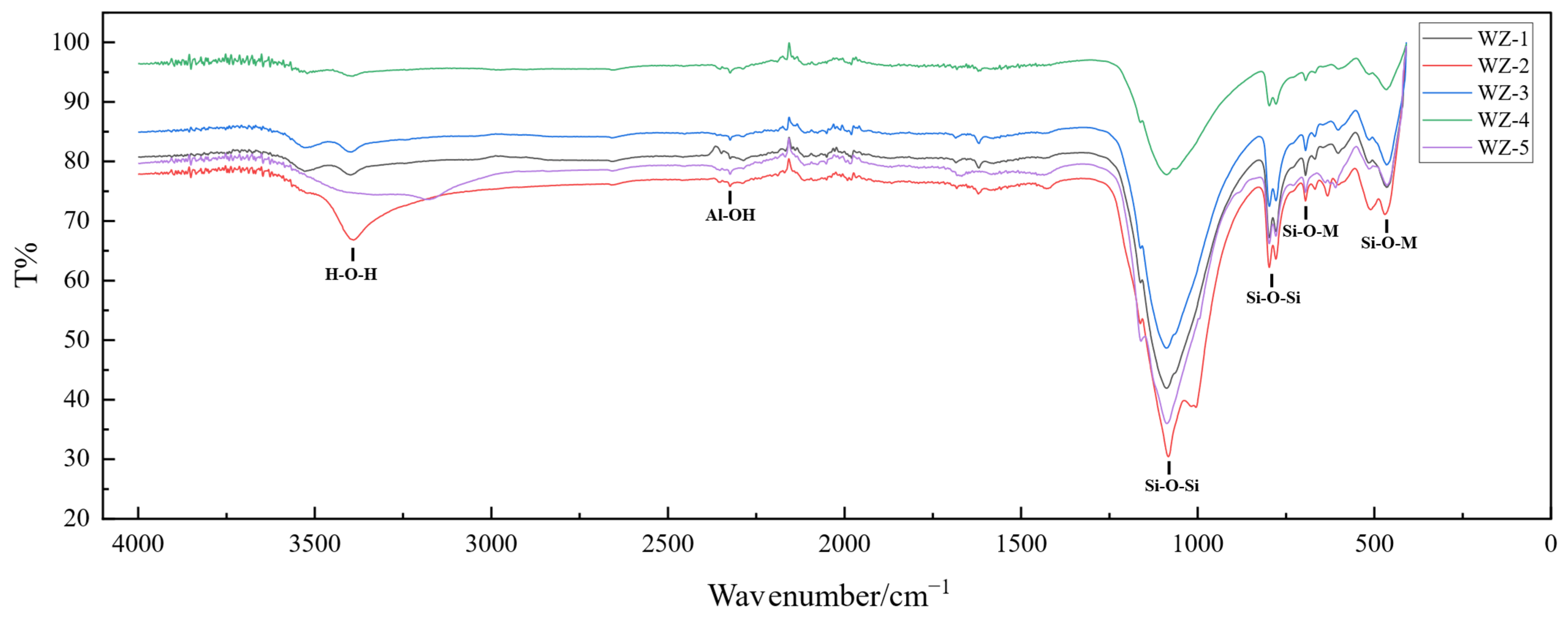
Fourier-transform infrared spectroscopy (FTIR) and X-ray photoelectron spectroscopy (XPS) were employed to characterize black shale bioleaching residues. These analytical techniques elucidated critical alterations in vanadium valence states and bonding configurations—notably Si-O and Al-O bonds—to evaluate the mineralogical liberation of vanadium-bearing phases. The results are presented in Figure 14 and Figure 15.
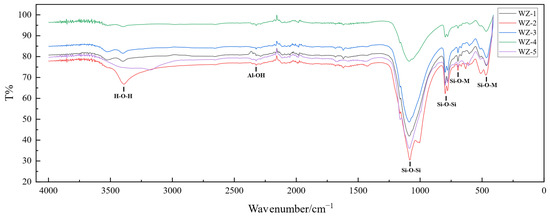
Figure 14.
Infrared spectra of black shale raw ore and leach residue (WZ-1-4 bioleach residue, WZ-5 raw ore).
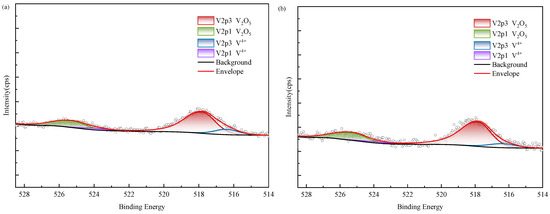
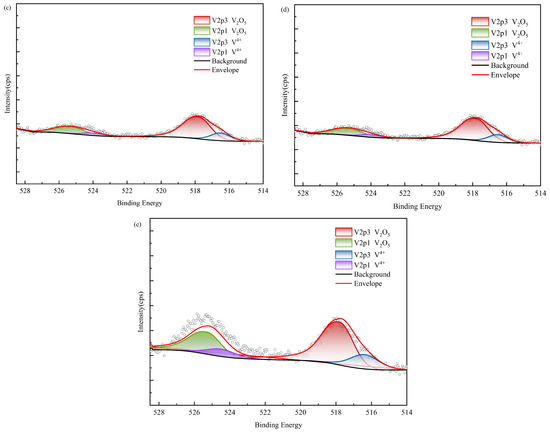
Figure 15.
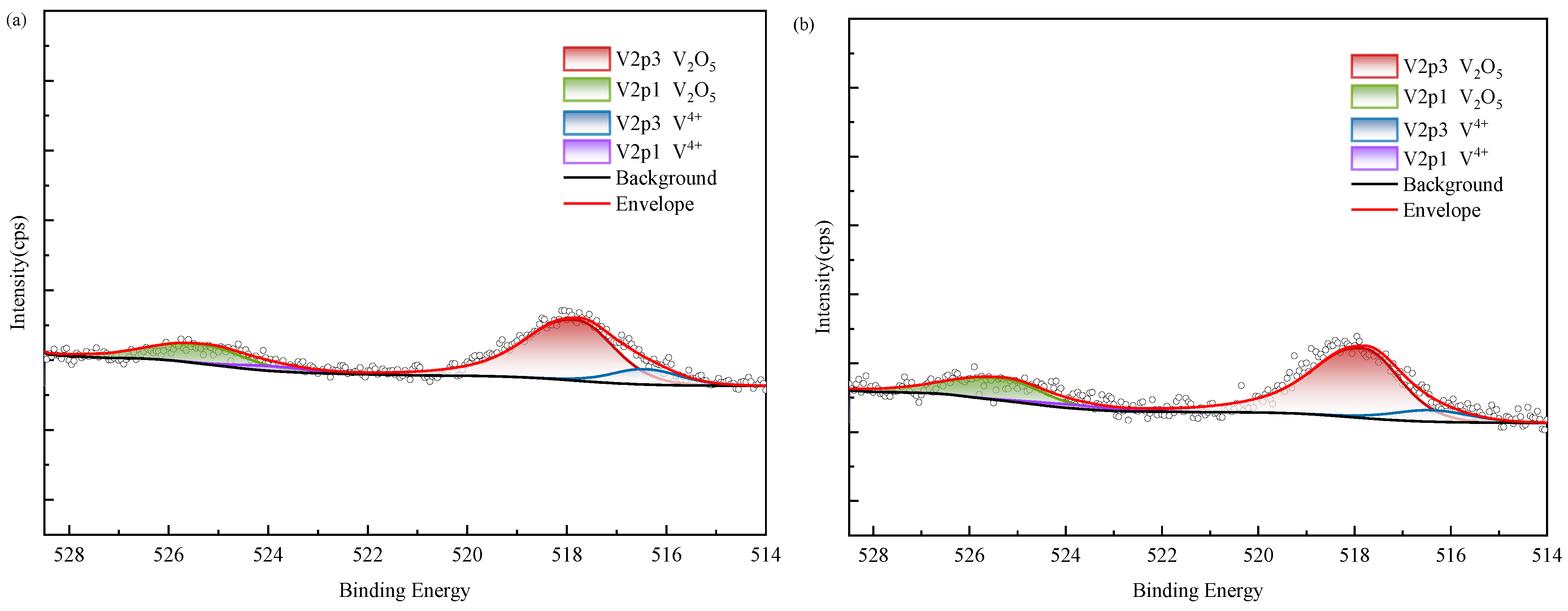
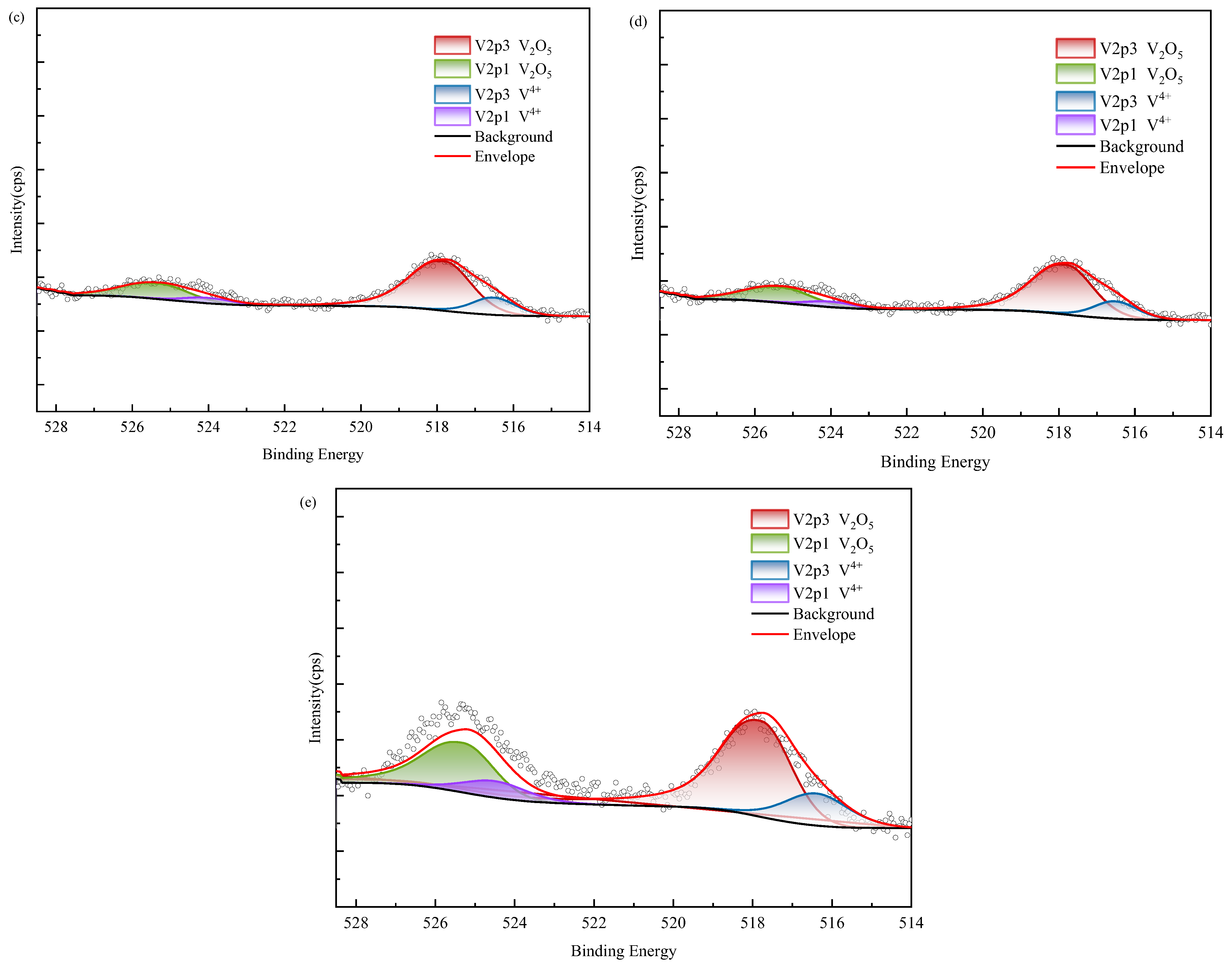
XPS results of vanadium in black shale and bioleach residue ((a–d): WZ-1~4 bioleach residue, (e): WZ-5 raw ore).
Post-bioleaching mineralogical analysis via FTIR spectroscopy revealed structural modifications. The altered absorption at 3409 cm−1 signifies hydrogen-bond restructuring within intercalated hydrous complexes of mica, indicative of disrupted -OH configurations. This confirms partial decomposition of the layered silicate framework. Additionally, minor shifts in Si-O-M bending vibration peaks suggest diminished bond symmetry within mica phases of bioleached black shale, although the fundamental tetrahedral-octahedral structural units maintained crystallographic integrity without detectable lattice distortion or collapse.
According to XPS results, vanadium in black shale raw ore mainly exists in the form of V(IV) and V(V). Tetravalent vanadium usually exists in the form of isomorphic substitution in vanadium mica, while pentavalent vanadium usually exists in the form of vanadium oxide. According to SEM analysis, it is present in oxide polymers. After biological leaching, the content of V(IV) decreases and is oxidized into V(V), which exhibits higher decomposability. The proportion of V(V) in the group with added ferrous is relatively higher than in other groups; however, correlation analysis indicated no corresponding increase in vanadium leaching amount, suggesting that the dissolution of oxidized vanadium remained incomplete.
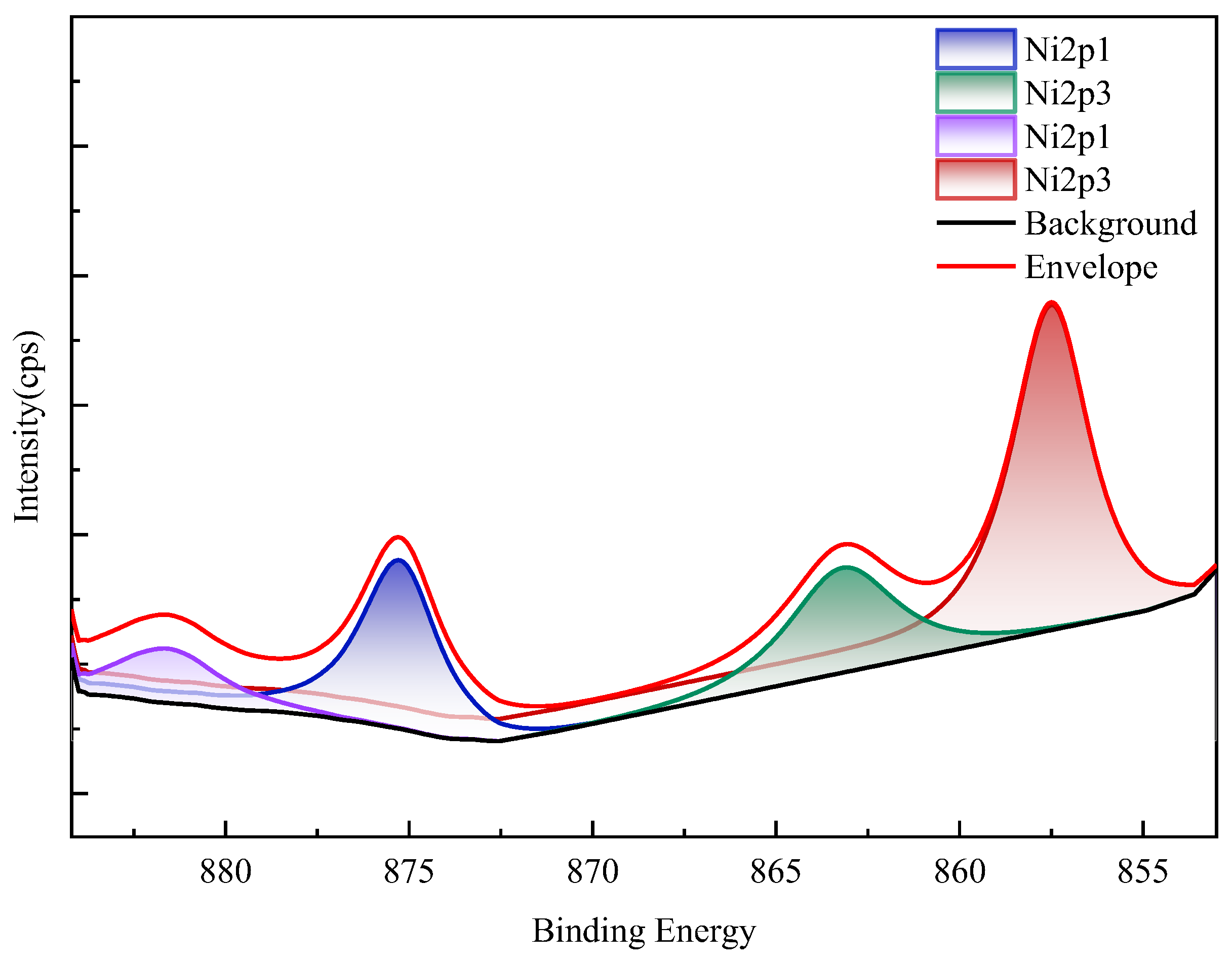
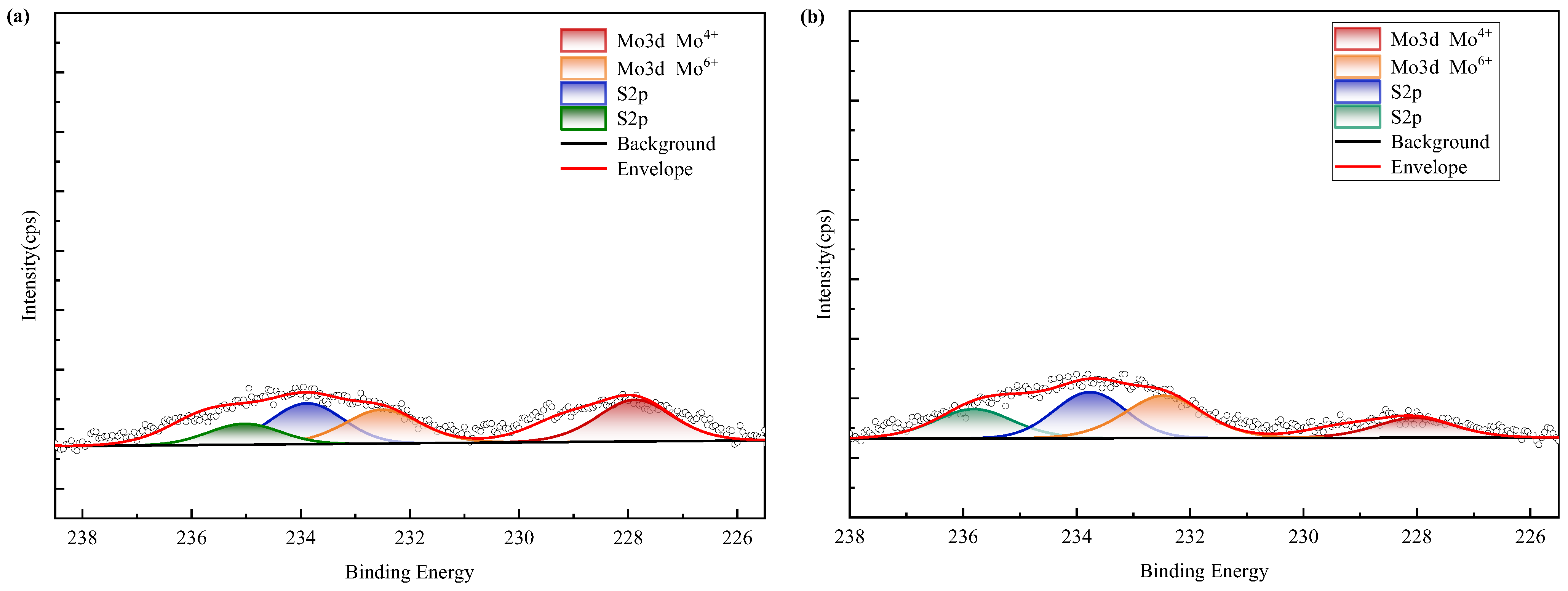
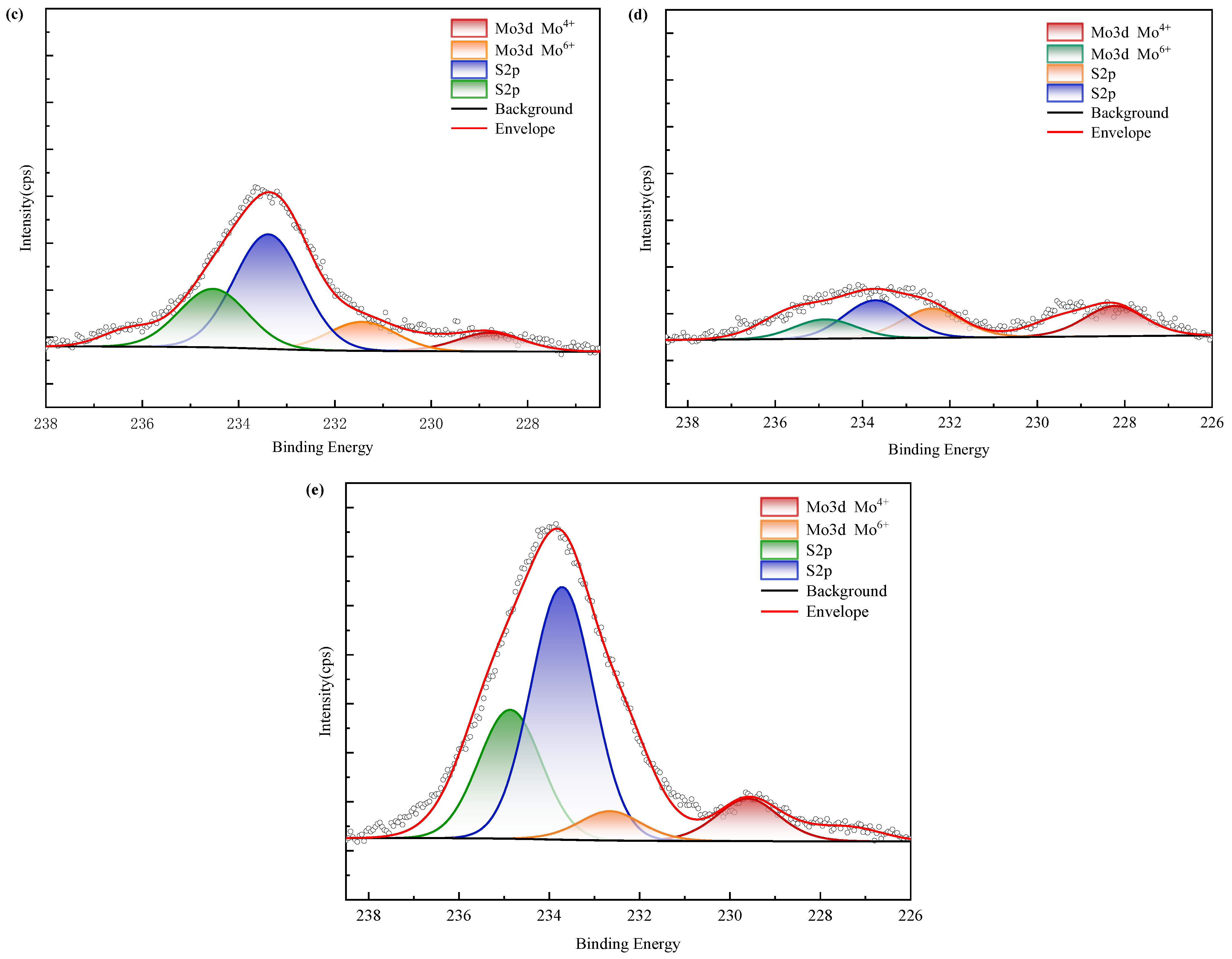
XPS spectra (Figure 16) confirm nickel predominantly occurs as refractory sulfide phases in raw ore. Post-bioleaching residues exhibited negligible Ni 2p signals, consistent with near-complete dissolution. Deconvolution of Mo 3d/S 2p spectra (Figure 17) revealed the coexistence of Mo(IV) and Mo(VI) species across all samples. Raw ore dominantly contained tetravalent molybdenum (e.g., molybdenite), whereas bioleaching triggered partial oxidation to hexavalent molybdate phases—particularly enhanced in ferrous-amended systems where elevated Mo leaching efficiencies correlated with greater Mo(VI) conversion. Notably, exogenous sulfate introduction in WZ-3/WZ-4 cohorts intensified S 2p spectral signatures despite unaltered sulfur speciation.
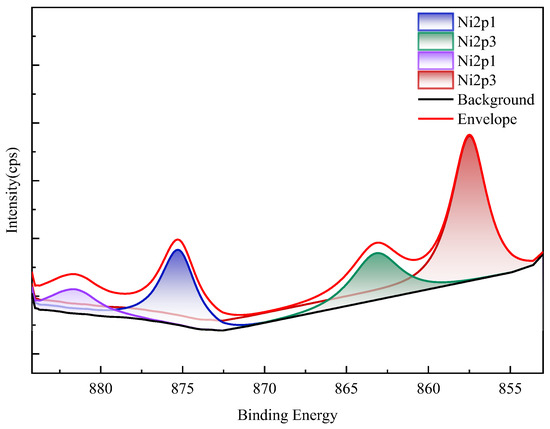
Figure 16.
XPS results of nickel in black shale and bioleach residue (WZ-5 raw ore).
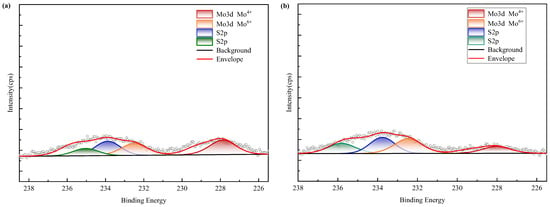
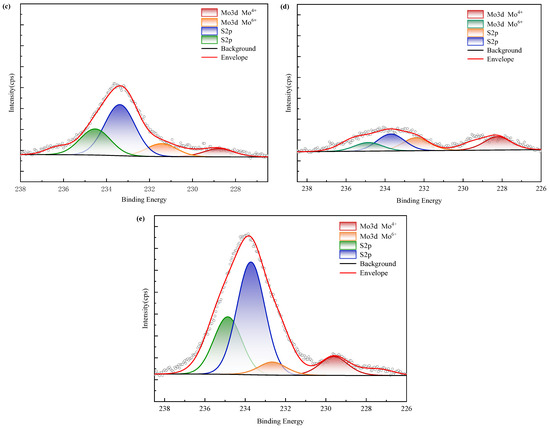
Figure 17.
XPS results of molybdenum in black shale and bioleach residue ((a–d): WZ-1~4 bioleach residue, (e): WZ-5 raw ore).
4.5. Discussion of Bioleaching Mechanism
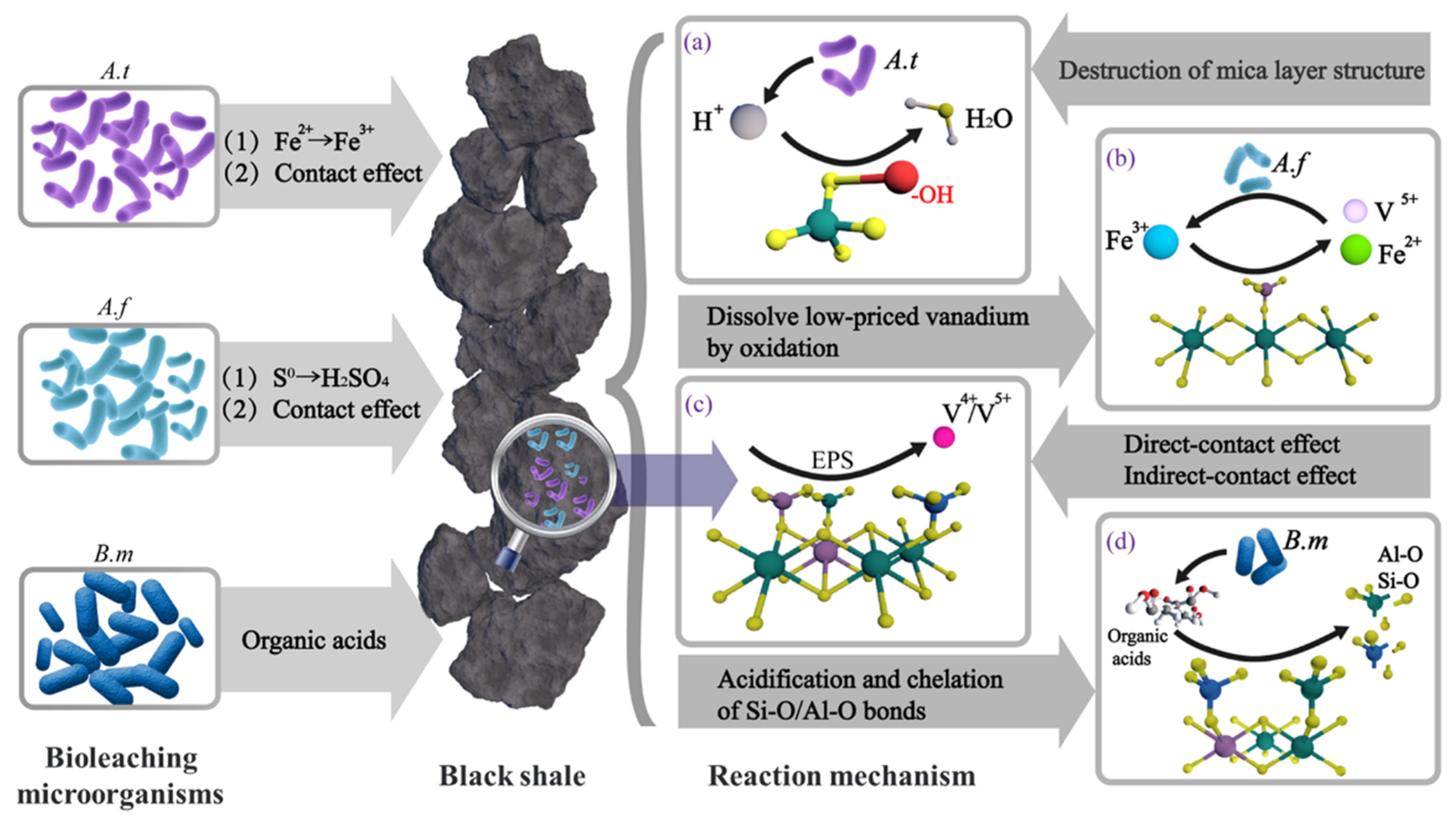
The autotrophic bacteria mainly employed for bioleaching of black shale are A. ferrooxidans and A. thiooxidans, which are currently the two most widely used microorganisms in the field of bioleaching. Since the discovery of A. ferrooxidans (formerly named Thiobacillus ferrooxidans), the mechanism of bacterial action on minerals has been a key focus of scholarly research. There are three main explanations for its action mechanism: direct-contact effect (microorganisms adsorb on target mineral surfaces and oxidize reduced iron and sulfur via specific cellular enzymes, disrupting the mineral lattice and releasing embedded valuable metals [27]), indirect-contact effect (microorganisms secrete extracellular polymers on mineral surfaces that enrich trivalent iron ions and form stable complexes with Fe3+, facilitating mineral matrix oxidation [28]), and indirect effect (leaching microorganisms oxidize leaching solution Fe2+ to Fe3+ or generate H+ while metabolically oxidizing S0; Fe3+ and H+ then synergistically promote mineral oxidative decomposition, converting insoluble valuable metals into soluble leaching solution ions [29]). Based on the above analysis results, the leaching of vanadium, nickel, and molybdenum from black shale mainly consists of two processes: the decomposition of mineral structures and the oxidation of valuable metal elements.
The dominant structural component in black shale is vanadium-bearing silicate minerals, and their decomposition is mainly affected by H+. According to the research findings of Zheng et al. [30], H+ reacts with hydroxyl groups between the layers of the mica structure, resulting in the initial destruction of the mica framework. This exposes vanadium to an oxidizing environment, where it is oxidized to high-valence vanadium; the latter then dissolves into the solution, thereby promoting vanadium leaching. The H+ in the system is mainly derived from the oxidation of elemental sulfur (S0) to sulfuric acid (H2SO4) by sulfur-oxidizing bacteria, and the massive generation of H+ is also reflected by a decrease in the system pH. This constitutes the primary role of sulfur-oxidizing bacteria in the leaching process.
On the other hand, under the action of ferrous-oxidizing bacteria, Fe2+ in iron-bearing minerals (e.g., pyrite) within black shale is oxidized to Fe3+, establishing an oxidative environment for the leaching system. Given that the redox potential φ (Fe3+/Fe2+) = 0.770 V is higher than φ (VO2+/V3+) = 0.680 V, the generated Fe3+ can oxidize vanadium to the more soluble V(V). Therefore, vanadium leaching can be attributed to the synergistic effect of these two types of leaching microorganisms. Their functions and the overall leaching process are illustrated in (a–c) of Figure 18.
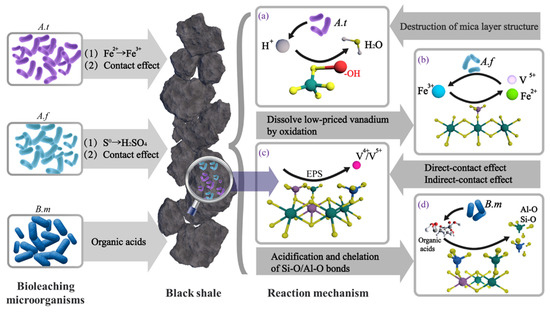
Figure 18.
Leaching mechanism of vanadium from black shale [31]. (A.f: A. ferrooxidans, A.t: A. thiooxidans, B.M: Bacillus mucilaginosus) (a) A. thiooxidans produce acid to promote mica decomposition; (b) A. ferrooxidans produce Fe3+ and oxidize lower-valence vanadium; (c) direct/indirect contact effect of A. thiooxidans/A. ferrooxidans; (d) organic acids produced by Bacillus mucilaginosus break Al-O and Si-O bonds.
Nickel and molybdenum, as traditional sulfide ores, follow the typical bioleaching mechanism of sulfide ores. They are oxidized to a more soluble valence state under the contact and indirect oxidation of microorganisms, and then leached in a sulfuric acid system. However, during this process, due to the encapsulation effect of silicate minerals and silica, and the inability of silica to decompose rapidly in this environment, a small amount of nickel and most of molybdenum are difficult to dissolve.
In response to the precipitation phenomenon of iron molybdate observed in SEM, the calculation results based on the water system potential pH diagram of molybdenum show that iron molybdate precipitation will occur in the leaching system at pH values greater than 1.5, system potentials less than 600 mV, and high concentrations of iron and molybdenum. Molybdate is a relatively stable structure that is insoluble except for a few specific salts. Therefore, the precipitation of iron molybdate generated is a critical barrier to leaching. In the future, the ion concentration of the system should be adjusted, and the system potential should be increased to ensure that the form of molybdenum is transformed into a more soluble form.
5. Conclusions
A specialized bioleaching consortium (designated WZ-Q), acclimatized for black shale processing, integrates Leptospirillum and Acidithiobacillus, with significant presence of Achromobacter and Acinetobacter, alongside minor proportions of Pseudomonas and Ferrimicrobium. It has obvious tolerance to the leaching system containing molybdenum, and has an effective leaching effect on vanadium, nickel, and molybdenum in black shale, which can be applied to the enhanced bioleaching of black shale.
To enhance microbial activity, energy substrates including Fe2+, S0, and pyrite were selected based on the dominant microbial community structure determined via high-throughput sequencing, to strengthen bioleaching performance and reveal the corresponding leaching mechanism.
In the process of bioleaching, it was found that the leaching percentage of vanadium is mainly related to the pH of the system; nickel always maintains a high leaching rate, while the leaching percentage of molybdenum is mainly related to the potential. The leaching of vanadium can be enhanced by the acid production of sulfur-oxidizing bacteria, the leaching of molybdenum can be enhanced by the oxidation capacity of iron-oxidizing bacteria, and the oxidation capacity of leaching microorganisms can be enhanced by adding corresponding energy substances. Under the optimized conditions, the maximum leaching percentage of vanadium can reach 66.5%, the leaching percentage of nickel can reach 82.5%, and the leaching percentage of molybdenum can reach 29.7%.
Combined phase analysis, FTIR analysis, XPS analysis, and SEM-EDS analysis of black shale raw ore and leaching residue, it was found that although the elements were oxidized under the action of microorganisms in the leaching process, they were not completely leached into the solution, and the encapsulation of silicate minerals was an important factor in hindering the leaching. The subsequent research should focus on the dissolution of silicate minerals such as mica and clay, and the methods to enhance the dissolution of oxidized minerals in the process of microbial leaching.
Author Contributions
Conceptualization, X.Y.; methodology, Y.L.; investigation, Y.L.; resources, H.S.; data curation, Y.L.; writing—original draft preparation, Y.L.; writing—review and editing, J.W. and X.M.; supervision, H.Y.; project administration, H.S.; funding acquisition, X.L. and J.S. All authors have read and agreed to the published version of the manuscript.
Funding
This paper was funded by the National Natural Science Foundation of China (52274354), the National Natural Science Foundation of China (52304297), and the National Natural Science Foundation of China (52074243).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The co-authors Yuanhang Lu, Jiankang Wen, Xiaolan Mo, Xinlong Yang, He Shang, Xue Liu, and Jianzhi Sun are affiliated with the companies National Engineering Research Center for Environment-Friendly Metallurgy in Producing Premium Non-Ferrous Metals, China GRINM Group Co., Ltd., GRINM Resources and Environment Tech. Co., Ltd., and GRIMAT Engineering Institute Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Ju, J.H.; Zhang, Z.Z.; Pan, Z.S.; Che, D.; Li, H.M. Determination of mineral resources in China’s strategic emerging industries andanalysis of the demand of the “14th five year plan”. China Min. Mag. 2022, 31, 1–11. [Google Scholar] [CrossRef]
- Dai, S.F.; Zheng, X.; Wang, X.B.; Finkelman, R.B.; Jiang, Y.F.; Ren, D.Y.; Yan, X.Y.; Zhou, Y.P. Stone coal in China: A review. Int. Geol. Rev. 2018, 60, 736–753. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, D.H.; Liu, W.H.; Zhang, B.X.; He, R. 2021 Global Vanadium industry Development Report. Vanadium Titan. Iron Steel 2021, 43, 1–9. [Google Scholar] [CrossRef]
- Coveney, R.M.; Nansheng, C. Ni-Mo-PGE-Au-rich ores in Chinese black shales and speculations on possible analogues in the United States. Miner. Depos. 1991, 26, 83–88. [Google Scholar] [CrossRef]
- Dai, S.F.; Seredin, V.V.; Ward, C.R.; Hower, J.C.; Xing, Y.W.; Zhang, W.G.; Song, W.J.; Wang, P.P. Enrichment of U–Se–Mo–Re–V in coals preserved within marine carbonate successions: Geochemical and mineralogical data from the Late Permian Guiding Coalfield, Guizhou, China. Miner. Depos. 2015, 50, 159–186. [Google Scholar] [CrossRef]
- Bie, S.; Wang, Z.J.; Li, Q.H.; Zhang, Y.G. Review of Vanadium Extraction from Stone Coal by Roasting Technique with Sodium Chloride and Calcium Oxide. Chin. J. Rare Met. 2010, 34, 291–297. [Google Scholar] [CrossRef]
- Zhou, Z.Y.; Zhu, Y.M.; Jin, J.P.; Han, Y.X.; Bai, Z.; Tang, Z.D. Enhanced vanadium extraction from Muscovite-type Vanadium-bearing shale by suspension oxidation roasting pretreatment-acid leaching. Sep. Purif. Technol. 2023, 309, 123066. [Google Scholar] [CrossRef]
- Li, M.T.; Wei, C.; Fan, G.; Wu, H.L.; Li, C.X.; Li, X.B. Acid leaching of black shale for the extraction of vanadium. Int. J. Miner. Process. 2010, 95, 62–67. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, Y.M.; Huang, J.; Liu, T.; Wang, Y.; Yang, X.; Zhao, J. Mechanisms of aid-leaching reagent calcium fluoride in the extracting vanadium processes from stone coal. Rare Met. 2013, 32, 57–62. [Google Scholar] [CrossRef]
- Wang, M.; Wen, J.K.; Li, W.J.; Yang, X.L.; Lai, Y.C.; Yang, H.Y. Research Progress on Recovery of Associated Strategic Metals Vanadium, Nickel and Molybdenum from Stone Coal by Combined Process of Beneficiation and Metallurgy. Chin. J. Rare Met. 2023, 47, 881–892. [Google Scholar] [CrossRef]
- Wang, M.; Cai, L.L.; Wen, J.K.; Li, W.J.; Yang, X.L.; Yang, H.Y. The Prospect of Recovering Vanadium, Nickel, and Molybdenum from Stone Coal by Using Combined Beneficiation and Metallurgy Technology Based on Mineralogy Feature. Minerals 2023, 13, 21. [Google Scholar] [CrossRef]
- Xian, P.F.; Zhou, S.F.; Wang, M.Y.; Wang, X.W.; Chen, B.F. Extraction of molybdenum and nickel from roasted Ni–Mo ore by hydrochloric acid leaching, sulphation roasting and water leaching. Trans. Nonferrous Met. Soc. China 2017, 27, 220–226. [Google Scholar] [CrossRef]
- Lu, M.; Zhang, Y.M.; Liu, T.; Yang, D. Sintering action of NaCl calcination during extracting vanadium from stone coal. Chin. J. Rare Met. 2009, 33, 894–897. [Google Scholar] [CrossRef]
- Wang, M.; Cheng, Q.; Qi, J.Y.; Li, J.; Ning, X.X.; Jin, J.P. Process of vanadium extraction from stone coal vanadium ore by sulfuric acid low temperature curing and column leaching. Min. Metall. 2020, 29, 62. [Google Scholar] [CrossRef]
- Wang, J.P.; Zhang, Y.M.; Liu, T.; Huang, J. Evaluation of microwave intensified vanadium bearing-high carbonaceous shale acid extraction process. Minerals 2018, 8, 113. [Google Scholar] [CrossRef]
- Zeng, X.; Wang, F.; Zhang, H.F.; Cui, L.J.; Yu, J.; Xu, G.W. Extraction of vanadium from stone coal by roasting in a fluidized bed reactor. Fuel 2015, 142, 180. [Google Scholar] [CrossRef]
- Chen, B.; Bao, S.X.; Zhang, Y.M. Synergetic strengthening mechanism of ultrasound combined with calcium fluoride towards vanadium extraction from low-grade vanadium-bearing shale. Int. J. Min. Sci. Technol. 2021, 31, 1095–1106. [Google Scholar] [CrossRef]
- Wen, J.K.; Chen, B.W.; Shang, H.; Zhang, G.C. Research progress in biohydrometallurgy of rare metals and heavy nonferrous metals with an emphasis on China. Rare Met. 2016, 35, 433. [Google Scholar] [CrossRef]
- Liao, M.X.; Zhang, X.Y.; Zhang, M.X.; Deng, R.; Zeng, Z.N. Study on the bioleaching conditions of vanadium from vanadium-bearing stone coal by Acidthiobacillus ferrooxidans. Fine Chem. 2015, 32, 144. [Google Scholar] [CrossRef]
- Wei, D.P.; Liu, T.; Zhang, Y.M.; Cai, Z.L.; He, J.T.; Xu, C.B. Vanadium bioleaching behavior by Acidithiobacillus ferrooxidans from a vanadium-bearing Shale. Minerals 2018, 8, 24. [Google Scholar] [CrossRef]
- Tang, H.Q.; Cai, Z.L.; Zhang, Y.M.; Zheng, Q.S. Chemical mutation of Bacillus mucilaginosus genes to enhance the bioleaching of vanadium-bearing shale. Biochem. Eng. J. 2023, 197, 108962. [Google Scholar] [CrossRef]
- Dong, Y.B.; Lin, H.; Liu, Y.; Zhao, Y. Blank roasting and bioleaching of stone coal for vanadium recycling. J. Clean. Prod. 2020, 243, 118625. [Google Scholar] [CrossRef]
- Dong, Y.B.; Chong, S.J.; Lin, H. Bioleaching and biosorption behavior of vanadium-bearing stone coal by Bacillus mucilaginosus. Int. J. Miner. Metall. Mater. 2023, 30, 283–292. [Google Scholar] [CrossRef]
- Wang, Y.; Cai, Z.L.; Zhang, Y.M.; Zheng, Q.S. Green recovery of vanadium from vanadium-bearing shale under the biological action by Bacillus mucilaginosus and its effect on mineral dissolution. J. Environ. Chem. Eng. 2022, 10, 107048. [Google Scholar] [CrossRef]
- He, J.T.; Cai, Z.L.; Zhang, Y.M.; Xue, N.N.; Wang, X.J.; Zheng, Q.S. Effects of energy source on bioleaching of vanadium-bearing shale by Acidithiobacillus ferrooxidans. Biochem. Eng. J. 2019, 151, 107355. [Google Scholar] [CrossRef]
- Yu, J.; Yang, H.Y.; Tong, L.L.; Zhu, J. Intensified bioleaching of low-grade molybdenite concentrate by ferrous sulfate and pyrite. Rare Met. 2015, 34, 207–214. [Google Scholar] [CrossRef]
- Pogliani, C.; Curutchet, G.; Donati, E.; Tedesco, P.H. A need for direct contact with particle surfaces in the bacterial oxidation of covellite in the absence of a chemical lixiviant. Biotechnol. Lett. 1990, 12, 515–518. [Google Scholar] [CrossRef]
- Yu, R.L.; Tan, J.X.; Yang, P.; Sun, J.; Ouyang, X.J.; Dai, Y.J. EPS-contact-leaching mechanism of chalcopyrite concentrates by A. ferrooxidans. Trans. Nonferrous Met. Soc. China 2008, 18, 1427–1432. [Google Scholar] [CrossRef]
- Tributsch, H. Direct versus indirect bioleaching. Hydrometallurgy 2001, 59, 177–185. [Google Scholar] [CrossRef]
- Zheng, Q.S.; Zhang, Y.M.; Xue, N.N.; Liu, T.; Huang, J. Vanadium occupation and its leachability differences in trioctahedral and dioctahedral mica. RSC Adv. 2019, 9, 27615–27624. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.H.; Wen, J.K.; Mo, X.L.; Yang, X.L.; Gao, W.C.; Yang, H.Y. A review of mechanism and technology of vanadium extraction from strategic mineral black shale. Rare Met. 2024, 43, 6183–6200. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.