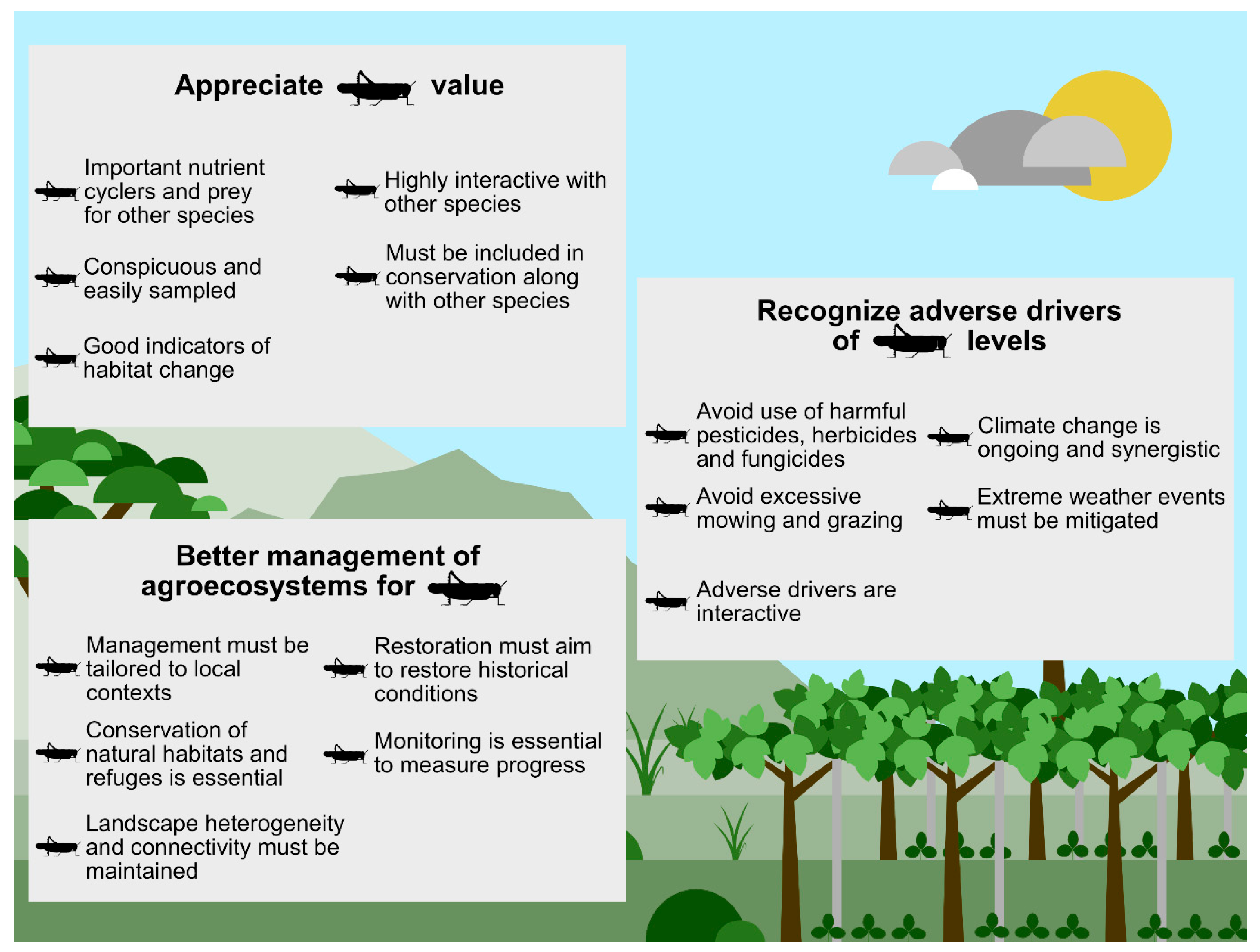
Abstract
Grasshoppers and their allies (Orthoptera) are numerous and diverse insects globally, while being significant components of biodiversity and nutrient cycling. They are variously responsive to environmental change but are paradoxical, as some species are major pests while others are threatened or even extinct. Most orthopteran species are somewhere in between, with their assemblage composition shifting in response to changing conditions and according to the response traits of the constituent species. With global concern over the impact of conventional agriculture on biodiversity, there is currently a strong transition to agroecology. The agroecological approach is two-fold: to set aside land and to better manage the overall landscape. Both approaches aim to boost the numbers and diversity of most orthopterans, while reducing the impact of the pest species using biologically based pesticides instead of chemical pesticides as part of an integrated pest management program. Set-aside land is present at various spatial scales for conservation action, involving a diversity of practical approaches. Management depends on understanding orthopteran responses to change, and harnessing the positive responses using, for example, improved grazing, fire management, and vegetation diversification for maximizing habitat heterogeneity. These initiatives also recognize the additional interactive effect of climate change and extreme weather events. Importantly, improvement of orthopteran abundance and diversity is an integral component of overall biodiversity conservation. New technologies, both aerial and genomic, are advancing the field of orthopteran biology and ecology greatly. We review advances being made in the field that hold the most promise and suggest ways forward based on three themes: appreciating orthopteran value, recognizing the adverse drivers of orthopteran abundance and diversity, and better design and management of agroecosystems.
1. Introduction
The growing need for sustainable agriculture has led to an increased interest in agroecological farming as a holistic framework integrating ecological principles into agricultural practices in a wide range of production systems ranging from arable and perennial crop systems, through to plantation forestry and peri-urban systems [1]. Assessing and monitoring biodiversity and ecosystem health is critical to this transition. In turn, bioindicators are essential tools for assessing the impact of agricultural management on ecological processes, as well as the effects of other natural or human disturbances on ecosystem integrity, whether in natural habitats or in agricultural systems [2].
Grasshoppers and their allies (Orthoptera), owing to their strong responses to environmental gradients, short life cycles, large size, and ease of sampling, are emerging as robust indicators in temperate and tropical agroecosystems. Their diversity, abundance, and species-specific habitat preferences enable Orthoptera to serve as sensitive indicators of subtle changes in vegetation composition, land-use intensity, ecosystem resilience, soil quality, microclimatic variations, habitat fragmentation, and the impacts of pollution or of invasive species [3,4]. However, Orthoptera are too often viewed through the narrow lens of economic impact, particularly as agricultural pests, while their ecological diversity and bioindicator potential remain underappreciated.
This intentionally comprehensive review aims to reframe orthopterans not solely as targets of pest management, but as valuable tools for ecological understanding and environmental assessment and monitoring, especially for underpinning the move away from conventional agriculture towards a more agroecological approach in a wide range of biomes and human environments where food and fiber production take place.
The insect order Orthoptera consists of two main lineages, Caelifera (grasshoppers and locusts, with short antennae) and Ensifera (bush crickets (katydids), crickets, and others, with long antennae) [5,6]. Currently, just over 29,400 extant species have been described [7]. The group has a global distribution [8], except in permanently frozen areas, with a roughly estimated global dry biomass of ≈30 Mt [9], making orthopterans a significant portion of terrestrial functional biodiversity. They inhabit all the major types of ecosystems from desert and grasslands to temperate and tropical forests as well as some species inhabiting freshwater ecosystems and caves.
Abundance of some orthopterans, especially among acridids, can reach extremely high levels. Desert locust (Schistocerca gregaria (Forskål, 1775)) (Orthoptera: Acrididae) adult density can reach about 60 million individuals/km2, which produce around 2.814 billion hoppers. In one generation, 18,240,000 kg of adults and 7,941,799 kg of hoppers consume about 26,182 t of plant material [10]. However, most orthopteran species do not reach anywhere near these density levels. For example, in Californian native grasslands, among the five acridid species present, average density over two years of all grasshoppers was 2.3 nymphs/m2, consuming 140 kg/ha of plant material [11]. Among three Chorthippus sensu lato species in England, the maximum density of any of the species recorded over many sites was 3.6 individuals/m2 for the ‘common’ Pseudochorthippus parallelus (Zetterstedt, 1821) (Orthoptera: Acrididae) [12]. Furthermore, some orthopteran species can survive remarkably well over many years in very small and isolated populations, seemingly without risk from the human footprint [13,14,15].
Population fluctuations among insects may be driven by exogenous factors (environmental stochasticity coupled with weak density dependence) and/or endogenous factors (over-compensatory density dependence), often leading to boom-bust population dynamics [16]. Orthopteran population levels vary over time, and sometimes greatly according to changing environmental conditions, especially rainfall. Some species, the locusts, swarm following heavy and extensive rainfall events, and then migrate [17]. Populations of other species, although still varying in levels over time, especially from one season to the next, remain overall, consistently common or rare [18], although there can be differences in the population levels among species, even at nearby sites, due to different field ecological characteristics and from different grasshopper associations [19]. Currently, relative population levels are also changing in response to climate change [20].
Orthopteran species are widely recognized as variously sensitive to habitat type. Some species, like Gryllus bimaculatus De Geer, 1773 (Orthoptera: Gryllidae), are highly tolerant of various types of human landscape transformation [21] making this species, for example, eligible as a potentially invasive species with ongoing climate change [22]. In contrast, other species are highly sensitive to change in their habitat, such as the hygrophilous Stethophyma grossum (L., 1758) (Orthoptera: Acrididae), the sub-populations of which naturally inhabit stable, long-standing naturally isolated habitats, which nevertheless are subject to change through human activity [23]. Overall, orthopteran species with low mobility and fertility, and narrower, more specialized climatic niches tend to show higher probability of local extinction [24].
Owing to the biomass, ubiquity, and diversity of orthopterans globally, they are major players in ecological processes. However, some species are pests requiring management and others are of conservation concern, and in need of protection, especially in this age of considerable landscape and climate change. Here, we take a global perspective to compare orthopteran responses to landscape transformation and pollution to ascertain differences versus similarities in different biomes and biogeographic realms that lead to a set of principles that pertain to their use in agroecological advancement. We first review some of the major traits responsible for the various sensitivities to changing environmental conditions among orthopterans. We then focus on the human drivers of orthopteran population and assemblage change in various parts of the world and consider the role of various orthopterans as bioindicators in agroecological improvement. Finally, given their valuable utility in being large insects and readily sampled, we make recommendations for their greater use in environmental and biodiversity assessment and monitoring in the context of agroecology, while also focusing on recent technological advances.
2. Orthopteran Biological and Ecological Traits
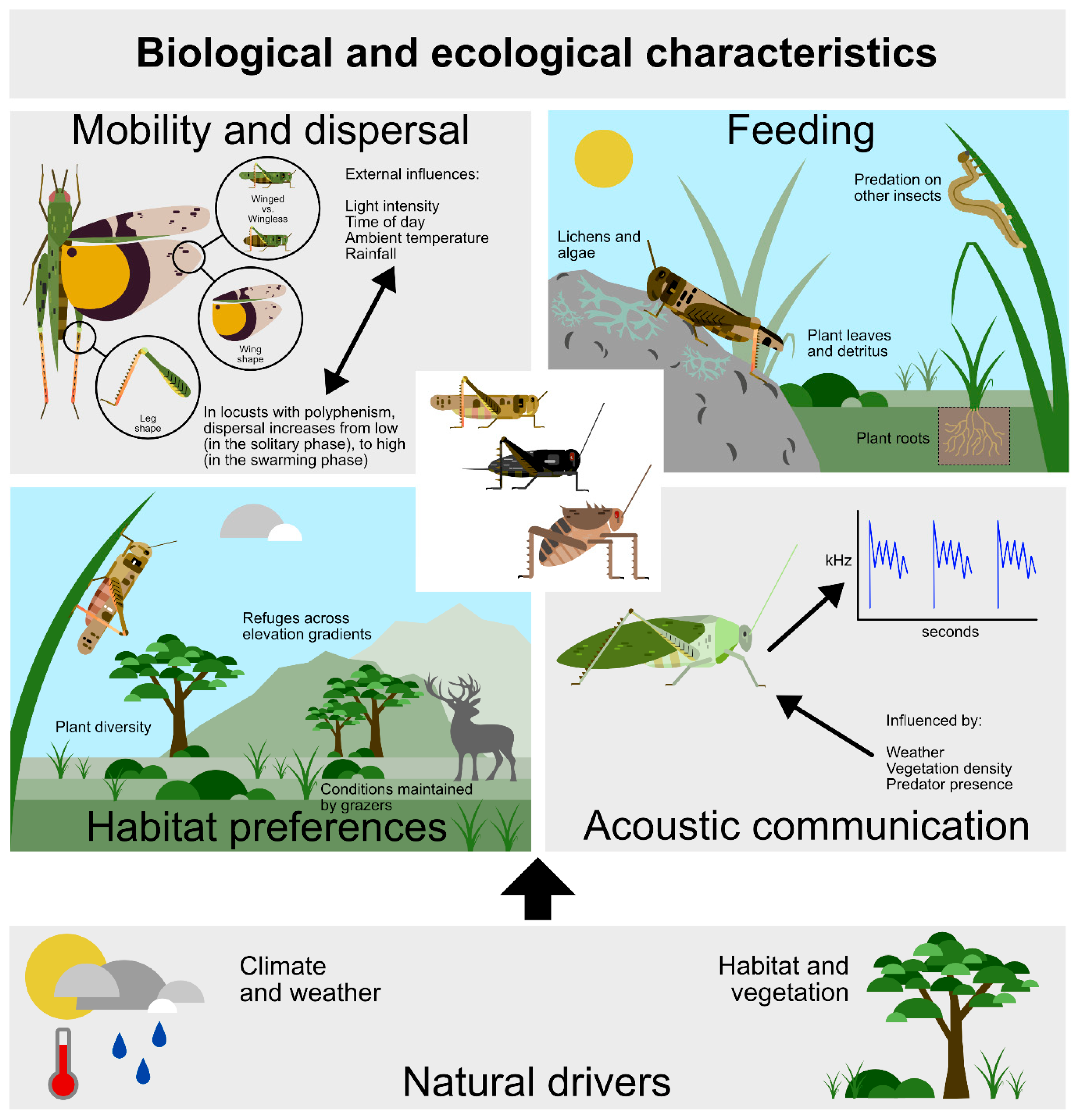
Orthopterans have a characteristic set of biological and ecological traits determined strongly by historical climate type and prevailing weather conditions, moderated by other factors, especially topography. In turn, these abiotic drivers determine the extent of habitat availability as manifested by vegetation type and composition. In broad terms, orthopterans characteristically respond according to four main biological traits: their mobility and dispersal capabilities, feeding behavior, habitat preferences, and acoustic behavior (Figure 1). We firstly consider these four fundamental orthopteran biological traits (Section 2.1.), followed by a discussion of ecological response traits to significant natural drivers (Section 2.2.). This trait perspective is an essential baseline for use of orthopterans as indicators of environmental assessment and for measuring the intensity of human activity (Section 3). In turn, this helps determine steps for agroecological assessment and monitoring, as well as for orthopteran conservation management (Section 4), all based on much insightful research undertaken in recent years (referenced in Section 3 and Section 4).
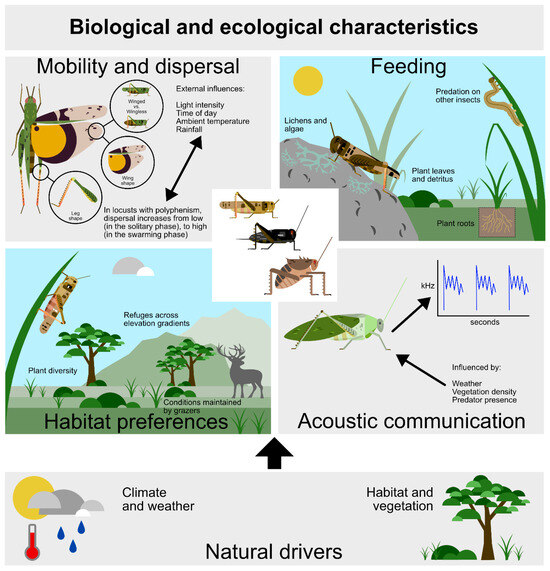
Figure 1.
Significant orthopteran biological traits relate to four main aspects of their biology on the base of two significant natural drivers.
2.1. Main Orthopteran Biological Traits
2.1.1. Mobility and Dispersal Traits
Insect movement enables individuals to locate optimal habitat for body maintenance and reproduction, as well as for escaping predators [25]. Orthopterans move by walking, jumping, and/or flying. In turn, movement depends on external factors such as light intensity and ambient temperature, and internal factors such as leg and wing morphology and associated musculature [26], body size [27,28], and response traits of individuals (see below).
Orthopterans have various levels of morphologically associated mobility which can differ greatly among species even at the same site [29]. There can also be differences among individuals within a species [30], or between different sub-populations [31]. In turn, the differential mobility among different species can sort assemblages across landscapes, with the sedentary species contributing most to species turnover [32]. However, there are multiple traits beyond those that are associated with dispersal that determine the response of orthopterans to landscape change. Furthermore, different existing traits are favored towards survivability according to the type of landscape disturbance [33].
One of the great differences among orthopteran species lies in whether they are flighted or flightless [34]. The term ‘movement’ refers to displacement that may occur over short and/or long distances, often in any direction whereas ‘dispersal’ denotes a directional movement toward a new location, usually aimed at enhancing survivability. Individuals of a species may display regular high levels of mobility but do not disperse far, while those in another species may move considerable distances to a new locality over largely straight-line distances in a day. The adult desert locust (Schistocerca gregaria) in its swarming phase, may cover an average of 183 km/day [35], whereas most species cover distances of only a few meters to reach an optimal microhabitat, procure food, a mate, or avoid intra- and interspecific competition, or seek enemy-free space [36].
In locusts—a limited group of grasshopper species exhibiting density-dependent phase polyphenism—individuals that are normally solitary may, under certain conditions, undergo relatively rapid and pronounced increases in aggregation and abundance, forming gregarious swarms [10]. This density-dependent transition is accompanied by coordinated changes in biochemistry, physiology, morphology, and behavior [37,38]. Such increases in population density typically occur following periods of high rainfall in dry areas, when vegetation flushes [39] and may be more prevalent because of human activity on the landscape [17]. This gregarious phase—responsible for major economic losses to crops—is highly nomadic in both nymphal (hopper) and adult stages, and when climatic and vegetation conditions are optimal. Dispersal may be over several generations and cover vast distances and areas where climatic and vegetation conditions remain suitable [40,41,42,43,44] (Figure 1).
2.1.2. Food Preference Traits
There are many types of feeding strategies among orthopterans overall. Most species, especially in the Caelifera, are herbivorous, with various species feeding on a wide range of living plants. Some, such as Tetrigidae, may feed on algae, mosses, and liverworts, and Gryllotalpidae eat roots, as well as grasses. Other species, notably in the Tettigoniidae, are predacious, mostly on other insects, as well as feeding on plant material, including seeds. Others, like the Gryllidae, are largely omnivorous or detritivorous. In turn, some may scavenge plant debris on the ground, and in the case of some Anostostomatidae, on fallen rotting fruit. Rhaphidophoridae may eat decaying plant material and even fungi. There are also cases of orthopteran individuals eating cadavers of their own species [45,46].
On the grasslands of Texas, grasshoppers range from monophagous to polyphagous feeders, although most species are oligophagous to polyphagous, with gomphocerines being mostly grass feeders while melanoplines feed largely on forbs. The oedipodines are less specific. Importantly, feeding patterns are remarkably constant across sites, with overall assemblage niche breadth distributions between sites being similar. Individual species tend to eat the same plant species at various sites and maintain similar niche breadths, while species with relatively specialized diets tend to feed on specific plant species such as grasses and long-lived perennial forbs [47] (Figure 1).
2.1.3. Traits Relating to Habitat Selection and Life Cycle Requirements
The habitat of a species is the total area where it can perform all its life functions and can grow into an adult. This involves seeking optimal food resources, whether nymph or adult, finding a mate, laying eggs, and developing into healthy adults that are free to disperse to maintain an effective population size. Egg-laying sites are chosen by the mature female, with the nymph then developing in an optimal microhabitat with suitable environmental conditions that ideally are also enemy- and pathogen-free, while facing minimum intra- and interspecific competition. There can of course be unforeseen adverse environmental factors such as floods, drought, or fire.
The egg, nymph, and adult may require different microhabitats suitable for each stage. In turn, the adult may have different feeding sites and perch sites from which to proclaim their mating song [36]. For example, at the small spatial scale, in mixed dipterocarp lowland Bornean rain forest and among 49 arboreal mostly ensiferan morphospecies, 96% were found to be new to science. Almost 50% of the sampled individuals were singletons, with only three species sampled more than once. The orthopterans were seemingly randomly distributed across the tree canopies, while most of the trunk specimens (92%) were small nymphs. This suggests that there is migration into the canopy after egg development in the soil or from lower forest strata [48].
With species that have a very wide geographic range, there may be biological variation across the range. For example, Decticus verrucivorus (L., 1758) (Orthoptera: Tettigoniidae) is widespread across the Euro-Siberia region, where it can tolerate well-below zero temperatures and reach an elevation of 2600 m a.s.l. It selects its habitat according to food availability, humidity, temperature, and where the eggs are in direct contact with water, and its eco-behavioral versatility has enabled it to occupy a vast area in post-glacial times [49]. However, in Britain, where this species is threatened, management must consider the microhabitat requirements of all life stages. For successful development and reproduction, this species requires a mosaic of vegetation heights. Both nymphs and male adults prefer taller, tussocky vegetation. But adult females prefer short, open turf for oviposition in the soil. The vegetation mosaic is also important for enhancing mate location as the male song is more effective over short turf than through dense vegetation [50].
There are many morphological forms of D. verrucivorus across its geographic range, with certain distinct adaptations to local environments [51]. Interestingly, the variable morphology depends on population isolation and density. In the late 1940s, the short-winged, large-bodied D. v. monspeliensis Rambur, 1838 reached pest levels in the Mediterranean-climate Montpellier area of southern France [52], but it is very rare today [53] (Figure 1).
2.1.4. Acoustic Communication Traits
Acoustic communication involves the evolution of both sound producing and sound reception organs. With about 16,000 orthopteran species primarily using acoustic signaling as a main mode of sexual communication, orthopterans are the most species-rich group of all sound-communicating animals [54].
Besides the calling song being energetically expensive to produce, there are other constraints. Firstly, atmospheric conditions, such as heat, humidity, and air movement (which can also rustle the vegetation) can affect the song broadcast. Secondly, vegetation, especially broad-leaved plants, can attenuate the song, especially the higher frequencies. Thirdly, there are also the faunal components (intraspecific and interspecific song interference, as well as listening predators) that may change song broadcast behavior and affect the position on the vegetation from where the song is proclaimed [55].
Orthopteran sound signals travel fast over long distances and are easily located and detected by potential predators at any time of day. While effective for locating mates, calls also enhance the chances that the signaler will be detected by a predator. Non-calling females approaching a male may also produce noise and vibration, making them more susceptible to predation [56].
Suffice here to note that orthopteran songs can have great value in the assessment of certain orthopteran species and assemblages in response to human-induced landscape change (see Section 4.8) (Figure 1).
2.2. Major Orthopteran Ecological Traits Relative to Natural and Semi-Natural Drivers of Populations and Assemblages
Environmental drivers include abiotic variables such as underlying geological structure and chemistry, climate, weather, temperature, along with occasional extreme events like drought, fire, or floods. Each variable has a spatial extent, severity, and timing, often moderated by topography. In addition, there are biotic environmental variables that range from the effects of food abundance and quality, pathogens, parasites, parasitoids, and predators through to interspecific and intraspecific interactions. Importantly, many of these environmental variables, both abiotic and biotic, are synergistic with each other and can change species dynamics and assemblage structures over both space and time.
2.2.1. Orthopteran Responses to Climate and Weather
Among the various abiotic factors shaping orthopteran ecology, climate and weather stand out as the most pervasive and influential. Acting across multiple spatial and temporal scales, they determine the physiological performance, phenology, and distribution of individual species, as well as the structure and diversity of entire assemblages [57,58,59]. Climatic influences operate through direct physiological pathways (temperature-dependent development, desiccation tolerance) and indirect ecological pathways (vegetation growth, food quality, predation). These drivers are often intertwined with other important factors such as microclimate, topography, and land-use intensity, together forming the environmental template that determines local orthopteran assemblage structure.
Temperature exerts a strong influence on the developmental rates, metabolic efficiency, and life-cycle duration of grasshoppers [57]. Warmer conditions generally accelerate egg/nymphal development and advance the timing of adult emergence.
Rainfall patterns regulate primary productivity, food availability, and microhabitat quality, especially in semi-arid and savanna ecosystems [58,60]. These patterns are especially strong where monsoons alternate with dry periods, while being exacerbated by land-use change [61]. Short-term extremes—droughts, heatwaves, or floods—impose strong selective pressures that can rapidly alter assemblage structure [62]. Furthermore, seasonality shapes the temporal organization of orthopteran life cycles and their synchrony with vegetation dynamics [63].
Long-term climatic trends and associated biogeographical shifts have had a profound influence on grasshopper populations. Over recent decades, global warming has driven marked northward and elevational range shifts in numerous orthopteran species [20,64,65,66,67].
2.2.2. Orthopteran Responses to Habitat and Vegetation
Grasslands, which cover about 40% of the Earth’s terrestrial extent [68], are important for orthopterans, particularly grasshoppers, while bushy and wooded patches are important for many tettigoniid species. High densities of grassland grasshoppers can play a major role in nutrient cycling, increasing primary production [69], while also serving as a critical food supply for other organisms, especially grassland birds [70].
Assemblage structures of grasshoppers in both the Asian steppe and North American Great Plains are determined by the flora. However, there are taxonomic differences. The Asian fauna is dominated by gomphocerines such as grass-feeding Chorthippus spp., while the North American fauna is dominated by both the gomphocerines and melanoplines (mostly Melanoplus spp.), which together are grass- and mixed-feeders. While the population dynamics in both regions are linked to weather events, the North American grasslands maintain about four times the grasshopper biomass per forage unit than the Asian steppe. Predators and parasites regulate grasshopper populations at low population densities in both regions, although pathogens have more effect in North America [71]. Furthermore, there are more flightless species in Asia than in North America [72].
Chinese grasslands support high and extensive populations of grasshoppers, with changes in their levels and distribution altering grassland dynamics [73]. Furthermore, these grassland ecosystems are not uniform owing to variation in soil type, above-ground biomass, elevation, and temperature that together mainly determine grasshopper diversity and abundance in typical steppe and meadow steppe [74]. The level of livestock grazing can also affect grasshopper prevalence, along with soil temperature in the egg stage, vegetation type, soil type, and rainfall amount in the nymph stage being significant factors in both meadow and typical steppes [75] and relative to elevation [76]. Also on the Chinese steppes, total grasshopper abundance and diversity largely increased with moderate grazing intensity but declined with high grazing intensity by Mongolian sheep (Ovis aries L.) (Artiodactyla: Bovidae), with species-specific responses prevalent [77].
In Central Europe, certain environmental variables (moisture, vegetation cover, vegetation height, direct radiation, and average seasonal temperature) determined niche widths relative to the threat status of orthopteran species. Vegetation cover and height, followed by moisture, affected the relative importance of orthopteran species. Average seasonal temperature, which is closely related to elevation, was generally of lower importance. Furthermore, threatened species had narrower niches than species of lesser concern [78]. In the Pyrenees mountain range, orthopteran species richness decreased with elevation. However, the elevation range distribution was hump-shaped (i.e., orthopterans at mid-elevations displayed a wider elevation range than those at lower and higher elevations), driven not only by elevation but also by vegetation structure (height and cover) and sampling effort. Interestingly, the shorter-winged localized endemic species were apparent at higher elevations, while the longer-winged species were more prevalent at lower elevations. The important point here is that clarifying the biotic and abiotic predictors of species distribution is important for informing conservation efforts and predicting consequences of climate change [79].
Grass height is a significant variable for orthopterans in Hungarian grasslands [80], while in Romania, plant species richness is an important component of landscape heterogeneity. However, orthopteran species richness and abundance negatively correlate with plant species richness but increase with the proportion of grassland in the landscape and local vegetation height. Overall, species-rich grassland orthopteran assemblages are significantly affected by both local and landscape scale land-use changes, but effects can vary according to orthopteran taxonomic group [81].
An experimental study of acridids in the Netherlands indicated the major role that vegetation plays in egg development and hence hatching rate, and subsequently on life cycle completion. This has implications when there is nitrogen loading of the grasslands, leading to an increase in vegetation density, reduced soil surface temperatures, a delay in hatching, and consequently a loss of thermophilous species [82].
Grasshopper interactions with grazing by indigenous megaherbivores and fire are ecologically significant. There are certain characteristics associated with responses of insect species or populations to fire. These are related to the degree and extent of exposure to, or shelter from, lethal temperatures, amount of stress experienced in the post-fire environment before full vegetational regrowth, suitability of this post-fire vegetation as habitat, and ability to increase numbers via survivors or colonizers. Factors such as mortality during a fire event, stress following a fire, ability of vegetation to resprout, and ability to repopulate are all important in determining the response dynamics of grasshoppers to fire, notwithstanding other effects such as mammal grazing [83]. Effects of grazing by large mammals on arthropods and orthopterans have been reviewed by [84] and [85], respectively.
In the tallgrass prairie of Kansas, there are no significant interactions between fire and American bison (Bison bison L., 1758 (Artiodactyla: Bovidae)) grazing. However, localized disturbances from grazing, and to a lesser extent fire frequency, influence grasshopper densities. Bison grazing activity results in significantly increased grasshopper densities, while infrequent fires (4 yr intervals) do not result in more grasshoppers as is the case in annually burned sites, although numbers of grasshoppers drop in relatively unburned sites (20 yr intervals). Overall, grasshopper densities respond negatively to increased canopy height and total grass biomass, and positively to increased spatial variability in canopy structure, with no detectable response to plant species richness and forb biomass. Recent bison grazing activity and recent fire following a long period of no burning also result in higher overall grasshopper densities, suggesting that grasshoppers can respond rapidly to altered habitat characteristics [63], with topographic position (upland vs. lowland) and fire frequency (1-, 2-, and 4-year intervals, and unburned) having little effect on grasshopper species richness or diversity [86]. However, long-term studies reveal that local weather events occurring early in grasshopper life cycles play a major role in determining grasshopper densities, especially of forb- and mixed-feeding species. In short, fire, grazing, and weather all modulate long-term fluctuations in grasshopper abundances and assemblage structure [87].
Grazing lawns are grassland areas where regular grazing by mammalian herbivores creates patches of short-statured, high nutrient vegetation. Grazing lawns are small sites that modify microclimate, plant structure, community composition, and nutrient availability. In Kansas tallgrass prairie, mixed-feeding grasshopper abundances are consistently higher on bison grazing lawns, while grass-feeder and forb-feeder abundances are higher on lawns only in the wetter years [88].
At a coastal dune community in California, exclusion experiments showed that grasshopper abundances were reduced by the presence of Black-tailed jackrabbits (Lepus californicus Gray, 1837 (Lagomorpha: Leporidae)) but not by the presence of Columbian black-tailed deer (Odocoileus hemionus columbianus (Richardson, 1829) (Artiodactyla: Cervidae)) [89]. Differences in grass sward height in the UK favor different Chorthippus sensu lato species [12], with grazing by Red deer (Cervus elaphus Linnaeus, 1758 (Artiodactyla: Cervidae)) reducing grasshopper densities in the UK [90]. In a German national park, Wild boar (Sus scrofa L., 1758 (Artiodactyla: Suidae)) rooting behavior on calcareous grassland positively influences orthopteran assemblages by creating a successional mosaic of microhabitats that supports different habitat specialists with various life cycle traits, leading to high levels of orthopteran diversity [91].
In subalpine grasslands in the Swiss Alps, where grazing by red deer and chamois (Rupicapra rupicapra (L., 1758) (Artiodactyla: Bovidae)) has created a mosaic of short- and tall-grass patches, there is an average density of 2.6 m2 orthopteran individuals. Neither abundance nor diversity differed between short- and tall-grass patches created by the ungulates. However, both abundance and diversity were positively influenced by increasing vegetation height, but negatively by increasing habitat diversity within patches. Increasing plant nitrogen content promoted a more even spread of species within the orthopteran assemblage on short- but not on tall-grass patches. Overall, large-scale habitat alteration by wild ungulates had no direct effect on abundance and diversity of the orthopterans in this system, although there were indirect effects on abundance and diversity by altering plant nitrogen content and structure of the habitat at small spatial scales [92].
Large ungulates also trample the vegetation as well as feeding on it, as seen for grasshoppers and other arthropods in a savanna protected area [93]. However, in these savanna areas the ungulates make distinct paths, particularly towards water holes, creating diverse habitats for grasshoppers [94].
Ecosystem types within a region may support very different levels of grasshopper biomass, related to both mammalian grazing pressure and fire. The southern African savanna supports 0.73 kg/ha grasshopper biomass, along with limited dietary overlap between the grasshoppers and large grazing mammals, with the grasshoppers transferring 77% of energy removed from plants directly to the decomposer chain as wastage or feces [95]. In contrast, the Mediterranean-type climate at the southern tip of Africa, with its sclerophyllous fynbos and very little grazing by mammals supports a grasshopper biomass of 0.08 kg/ha. However, in seven months post-fire there is an increase in grasshopper biomass to 0.12 kg/ha following regrowth of the fire-resilient grasses and restioids, along with an increase in grasshopper diversity [96]. In both cases, grasshopper assemblages show considerable behavioral and morphological adaptation [97], such as fire melanism, in fire-prone environments, suggesting camouflage and predation as important drivers of fire melanism in some orthopteran species [98,99].
In turn, fire alone can have a major impact on orthopteran assemblages. For example, in the dry, sunny winter in the Drakensberg Mountains, South Africa, where adults of 19 grasshopper species remain active mid-winter, they seek warm refuges on hillsides. However, fire removes the grass cover, leaving ash on the soil surface. Burning of the grass insulating layer results in a night-time temperature inversion directly interfacing with the soil. Artificial translocation of grasshoppers to these burned areas left them exposed to a temperature drop of about 10 °C, with all individuals dying [100].
In northern Arizona, Arroyo willow (Salix lasiolepis Bentham, 1857 (Malpighiales: Salicaceae)) readily resprouts after fire, with these vigorous resprouts especially attractive to both certain grasshoppers and Elk (Cervus canadensis (Erxleben, 1777) (Artiodactyla: Cervidae)), and which are nutrient rich [101]. In a protected area in Arizona, fire reduced grasshopper density in an ungrazed semiarid grassland, and changed the relative abundances of species and subfamilies, but only for 1 or 2 years after the fire. In contrast, plots that had burned for >20 years supported similar plant cover and grasshopper populations as plots only in their 3rd year postfire. While the grassland community is highly fire-tolerant, it is not fire-dependent, with grasshopper assemblages being remarkably resilient [102]. Similar results come from the mixed grasslands of North Dakota, where fire negatively affected grasshopper population densities in the year after a late autumn wildfire but did not significantly affect green plant biomass or nitrogen content of grasses. Grasshopper decline was mostly due to a drop in densities of gomphocerine species, with the assemblage overall recovering two years later [103].
In a tall-grass prairie in Kansas, experimental fire regimes affected grasshopper assemblage structure hatching several weeks later after spring fires. There were shifts in local species composition as nymphs matured, with fire adversely affecting survivorship of forb- but not grass-feeding nymphs. However, because forb feeders were most common on burned sites that had previously been protected from fire longest, change in grasshopper assemblages after fire was especially pronounced at these sites. This meant that overall greatest species diversity was at sites subjected to intermediate frequency of fire, i.e., every four years [104]. On short-grass prairie in Colorado, there were differences among grasshopper assemblage responses to fire according to location, resulting from pre-existing differences among the locations and sites. This suggests that artificial spring burns in the short-grass prairie do not affect the grasshopper assemblage beyond the natural variability occurring within the grasshopper assemblage [105].
3. Human Drivers of Orthopteran Population and Assemblage Change
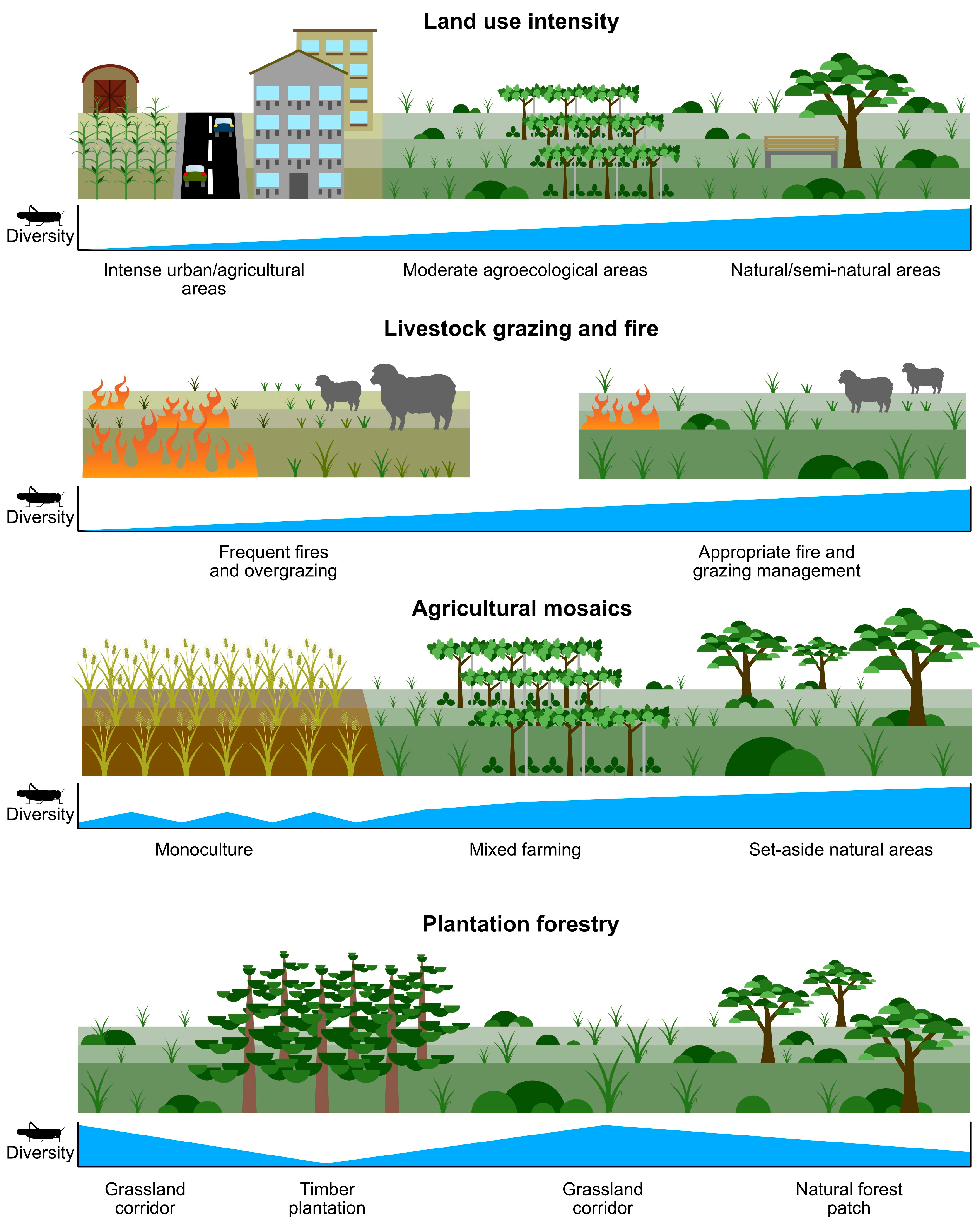
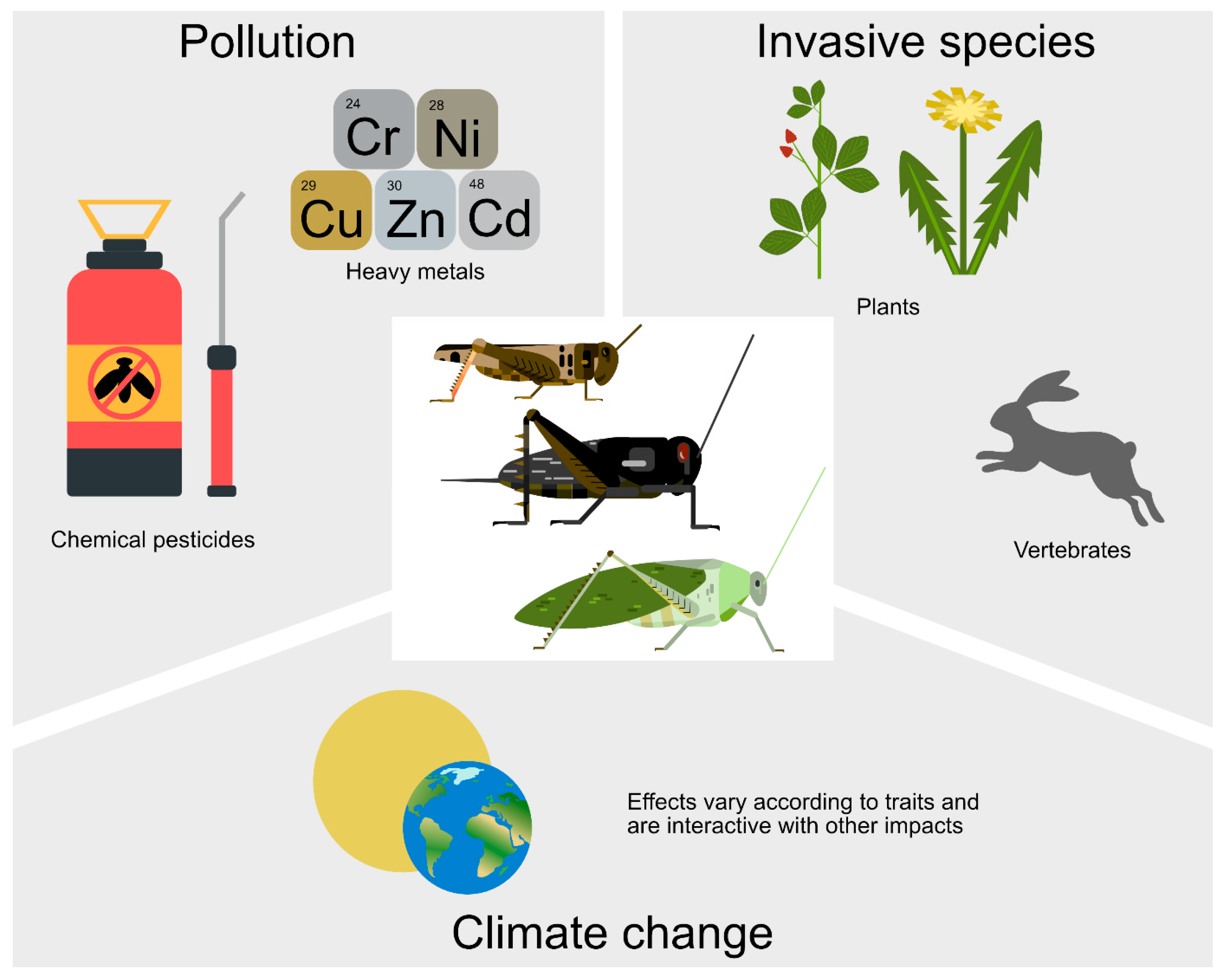
In this section, we go beyond the natural and semi-natural drivers of orthopteran population and assemblage change to consider the additional major human drivers of change. This section is divided into two parts: human-induced landscape change (Section 3.1, Section 3.2, Section 3.3 and Section 3.4) (Figure 2), (while also mentioning urban environments where agroecological advancement is also taking place (Section 3.5)), and some of the distinct adverse drivers of othopteran populations and assemblage composition (Section 3.6, Section 3.7 and Section 3.8) (Figure 3).
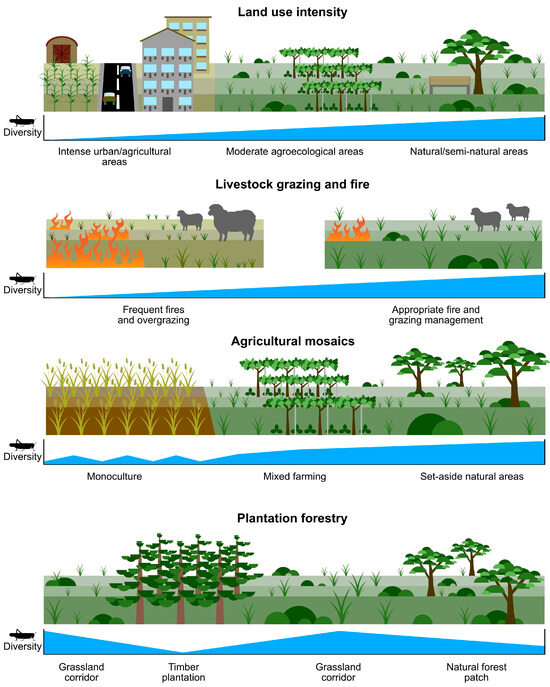
Figure 2.
Orthopterans respond, often strongly, to various aspects of human-induced landscape change.
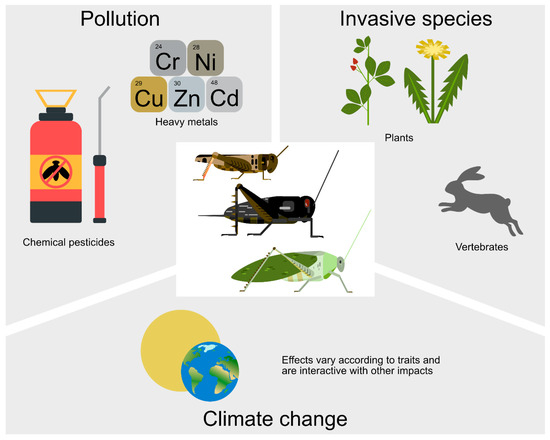
Figure 3.
Some adverse drivers on orthopteran populations and assemblage composition. Climate change also affects orthopterans, with a positive effect on some but a negative effect on others and can also be adversely synergistic with the other drivers.
3.1. Landscape Transformation and Habitat Loss
There is great concern over the loss of insect species globally [106], which includes orthopteran species, and where landscape transformation and habitat loss are among the greatest drivers of extinction risk (www.iucn.redlist.org). But among Orthoptera, as well as other insect taxa, there can be major differences among species in their response to human-induced landscape change. We now consider these responses.
A first consideration is the taxon/taxa being studied, where responses to modified habitat can vary among families and even within a family, such as the Acrididae [107]. Secondly, results can vary greatly according to the spatial scale of observations. As Cherrill [108] points out, using, for example, 100 km2 sampling areas across a wide area of southern England leads to areas of arable land being associated with decreasing orthopteran species frequency. But, nevertheless, there are always small areas of natural or seminatural habitat suitable for various species within these large quadrats, even where there is agricultural intensification.
When point sampling was used over a large area of Germany, high land-use intensity across various ecosystems negatively affected orthopteran diversity across years and regions, corresponding to decreases associated with high fertilizer use, and to the intensity of mowing and livestock grazing, high levels of which also reduced abundance. Overall, 15 of 29 orthopteran species were losers due to intense land use and yet were more abundant in grasslands with low-intensity land use. Two species were winners of high land-use intensity, whereas the remaining twelve were opportunists. Interestingly, omnivorous, herbivorous, and graminivorous species did not differ in their response to land-use intensity, whereas the Tetrix species that fed on mosses or lichens showed consistently negative responses to intensive land use. These results suggest that low intensity fertilizer use, infrequent mowing, and variable grazing levels will help maintain high orthopteran diversity [109].
Also in Germany, orthopterans showed a clear response to plant community succession, with each successional stage supporting a unique assemblage. Although species richness among habitat specialists was highest in the earliest seral stages, the density of all species peaked at the intermediate successional stage. Consequently, conservation management should aim at reintroduction of traditional, low-intensity land use for abandoned steppe grasslands using traditional rough grazing with sheep and goats, which creates a heterogeneous vegetational habitat structure along with some bare ground, without the accumulation of litter which would otherwise be inhibitory [110].
In another German study, land-use intensity was the only predictor of Orthoptera species richness. Furthermore, the mean number of all species and of threatened species was highest in grassland with low land-use intensity, in dry grasslands, and in those differing from the mesic (high land-use intensity) and wet (mostly abandoned) grasslands. Additionally, dry grasslands had the highest number of all threatened and exclusive indicator species. Intensive land use in mesic grasslands and abandonment of land use in wet grasslands led to a homogenization of habitat structures and of the assemblages. In contrast, dry grasslands with low land-use intensity and high habitat heterogeneity were hotspots of Orthoptera species richness [111]. In France, elevation is an additional strong driver of orthopteran assemblage trait composition, and plant species richness correlates with orthopteran alpha diversity. In turn, less intensive agricultural practices enhance orthopteran diversity [112].
Among central European grasslands, climate warming has led to biotic homogenization of orthopteran assemblages associated with abandoned wet grasslands and interacts with land-use intensification in mesic grasslands. The outcome is a tendency towards dominance by more widespread species. In contrast, common pastures are characterized by high heterogeneity of landscapes and low-intensity management and, as a result, are more resilient to the effects of climate warming [65].
Widely distributed grasshopper species in the Inner Mongolian grassland likely will extend their distribution northward with climate change. However, because of differential responses to warming, different species are predicted to aggregate toward the middle period of the growing season, potentially increasing interspecific competition and grazing pressure on grasslands [113]. Over time, it is now known that certain grasshopper species have already adapted to climate change through mechanisms such as diapause. By serving as ecological indicators, grasshoppers offer valuable insights for monitoring climatic and environmental shifts [114].
In Europe, there have also been long-term effects of landscape fragmentation. The oedipodine grasshopper Bryodemella tuberculata (Fabricius, 1775) (Orthoptera: Acrididae) was a widely distributed species prior to the mid-19th century. Since then, many European populations have declined greatly, with some populations becoming either highly threatened or extinct. While Europe still has highly suitable habitats close to the climatic optimum, changes in land cover and other human impacts on habitats at the local scale seem to be responsible for local extinctions. Genetic analysis suggests that the declining European populations are relics of a formerly wider distribution, subsequently fragmented by changes in land use. These relics are now threatened by limited connectivity and small effective population sizes [115].
In Indian dry forest, acridid abundance and species diversity were highest in natural habitat with high plant diversity and an absence of human disturbance, including livestock grazing. Increasingly more intense human disturbance caused a drop in grasshopper species richness, diversity, and abundance [116].
A 37-year study over a wider area of the Pampas showed that agricultural intensification in the Pampas has modified the distribution pattern and abundance of grasshoppers, seemingly favoring generalist species over others. Assemblages are dominated by a few species, with three or four species representing over 50% of the total. Some abundant species declined or disappeared over time, while others appeared later. There were varying abundances of species across zones, with some species, such as Dichroplus maculipennis (Blanchard, 1851) (Orthoptera: Acrididae) and Borellia bruneri (Rehn, 1906) (Orthoptera: Acrididae), being more abundant in specific areas in the south-east [117].
In tropical Cameroon, slash-and-burn/planting of cash crops greatly changes the naturally forested landscape. Among the ground and low canopy acridids, abundance and species diversity increase with level of land transformation, with a tendency towards homogenization of the fauna with increased conversion to agriculture. However, species diversity is highest in the more moderately transformed forests [118]. Also in Cameroon, comparison of the acridid fauna farmland (with short grasses), fallows (with tall grasses), and forest (with tall trees), illustrated that abundance, species richness, and diversity were highest in the farmlands than fallows or forest, although one catantopine grasshopper species, Serpusia opacula Karsch, 189 (Orthoptera: Acrididae), increased in abundance with the degree of naturalness in the forest [119].
However, these two studies did not focus on the great changes in orthopteran assemblages following tropical forest loss. In Malaysian forests, orthopteran assemblages were affected by habitat degradation through logging and the conversion of forest to oil palm. The greatest difference in species composition was between plantations and primary forests. Logged forests had the lowest species richness of the three habitats investigated [120].
Landscape transformation, sometimes coupled with extreme weather events, can cause huge surges in certain potentially pestiferous species. Not surprisingly, expansion of arable agriculture provides a novel food resource in times of locust outbreaks. For example, Locusta migratoria (L., 1758) (Orthoptera: Acrididae) swarms in maize and wheat cropping areas of South Africa, where it is difficult to control and has taken advantage of crop production to produce an extra generation per year that was not previously possible in the original grasslands [121]. Forest loss, in combination with broader environmental changes, can also trigger outbreaks of the tropical migratory locust L. migratoria. The 1997 drought in Indonesia was intense, seemingly due to extensive deforestation for agricultural expansion, and had adverse effects on the locust’s natural enemies [122]. This locust is also subject to outbreaks, especially in Mali, Chad, Sudan, and Madagascar, resulting from the introduction of agricultural and pastoral activities, especially extensive cultivation of monocotyledon summer rainfall crops. Some locust outbreaks follow initial ground disturbance without subsequent population resurgence, while other populations surge following high rainfall events in the novel agricultural areas. Continued agricultural development in tropical and subtropical areas of the Old World will bring the risk of further upsurges of the tropical migratory locust [123]. In recent years, intensification of extreme weather events has favored Schistocerca gregaria swarm formation in North Africa, which is erratic and difficult to forecast. For example, despite a significant long-term trend of increased incidence of drought in Africa, formation of swarms in 2018 was stimulated by heavy rains from two tropical cyclones in the Arabian Peninsula. These extreme weather events led to three generations of the desert locust developing and remaining undetected until they spilled over into a much wider region. This situation constitutes considerable agricultural risk when rainfall is combined with favorable vegetation and soil conditions, which are themselves also the result of weather events. This situation is also aggravated by human-induced changes in land use (e.g., agriculture, wood extraction, urbanization), for which there are no reliable forecasts (Figure 2).
3.2. Livestock Grazing and Fire
There can be very different orthopteran species-specific responses to different grazing intensities according to the requirements of the different life stages and associated traits [124]. Furthermore, the effects of livestock grazing and fire in grasslands are often interactive [85]. In northern mixed-grass prairie in the USA, the interaction of the two drivers had a cumulative effect on grasshopper density and assemblage composition, with burned and grazed pastures having higher grasshopper densities than unburned pastures. However, these effects are highly dynamic over space and time, with the grasshopper populations being highly resilient to the environmental changes of the landscape [125,126].
In the dry grasslands of Arizona, the effect of livestock grazing alone on grasshopper abundances and assemblage composition varies greatly from one year to the next [127]. While in Idaho rangelands, heavy invasion by alien grasses and other plants, coupled with drought, diminishes acridid population levels [128]. Further north, in the Canadian fescue grasslands, the relative densities and assemblage composition of grasshoppers changed considerably with intensity of cattle grazing, with heavy grazing in this cool, wet environment not being adverse to grasshoppers overall [129].
In Montana, experimental manipulation of grasslands (supporting several pestiferous grasshopper species) with different livestock densities, showed that increased grazing intensity reduced overall grasshopper densities. Ungrazed sites supported dense vegetation, which took away ground-level basking sites for some species. In contrast, heavily grazed habitats have less shade and fewer elevated perches for cooling. In short, grazing affects the microclimate, which can affect hatching date, early season survival, rates of development, susceptibility to pathogenic fungi, and availability of oviposition sites, with some species preferring to lay eggs in soil trampled by livestock, while others prefer to oviposit within grass clumps, or in litter-free patches of bare soil [130]. Further studies in the same area showed that some pest grasshopper species tended to be more abundant on ungrazed plots, whereas others had greater densities on heavily grazed plots, but grasshopper densities were reduced by the presence of the invasive Crested wheatgrass Agropyron cristatum (L., 1770) Gaertner 1770 (Poales: Poaceae) [131].
Increased livestock grazing intensity also reduces grasshopper diversity on Inner Mongolian grasslands [76]. Furthermore, rainfall levels interact with the effect of grazing on grasshopper assemblages. Desert steppe was low in grasshopper species richness and diversity, but abundance was not different from wetter sites. At dry sites, livestock grazing decreased grasshopper diversity and increased abundance of the main pest species. At the wettest sites, grasshopper assemblages did not differ relative to grazing levels. Across all sites and grazing intensities, grasshopper abundance tended to be lowest and diversity highest in plant communities with intermediate levels of biomass and plant species [132]. Interestingly, in these same grasslands there is a significant correlation between variation in the gut microbiota of Calliptamus abbreviatus Ikonnikov, 1913 (Orthoptera: Acrididae) and changes in plant species under different sheep grazing intensities, with structural shifts between the gut microbial communities of grasshoppers with dietary regimes subject to resource constraints (i.e., under grazing) and those with abundant resources (i.e., no grazing) [133].
In the arid Karoo of South Africa, long-term rotationally grazed sites by cattle and sheep supported the highest overall grasshopper abundance and species richness compared to those in continuously grazed sites [134]. In Spain, grazing pressure homogenizes the orthopteran assemblage. However, even during the first year after abandonment of grazing, orthopteran abundance, species richness, and diversity increased [135] (Figure 2).
3.3. Agricultural Mosaics
The agricultural landscape is a mosaic of different landscape elements, which can roughly be divided into the agricultural patches (plus associated infrastructure) and the non-production, set-aside areas. The amount of suitable habitat in the form of these set-asides can be a major determinant of the level of prevalence of, for example, gomphocerine grasshoppers across an agricultural landscape [136]. In China, mixed farming, which included lac farming plantations, secondary natural lac forest, crop lands, and rice paddies, supported 31 orthopteran species in five families. Grasshopper abundance and richness correlated with plant resources, with mixed farming providing an optimal approach for balancing conservation and development, compared to any single type of land use [137].
At the boundary between any two types of landscape element, such as agricultural and natural patches, there can be spillover effects from agricultural activities, which might not necessarily be negative. Fertilizer application to the native North American grass Calamovilfa longifolia increased the total biomass of Ageneotettix deorum (Scudder, 1876) (Orthoptera: Acrididae) [138]. In turn, bush crickets of the tettigoniid genus Platycleis in southern France benefitted from spillover of alien wild oat grass (Avena spp.) on which they will feed [139].
The agricultural patches have varying degrees of agricultural intensification, both in terms of design and management. A crucifer field, with its intense annual tilling, planting, and harvesting, is very different from a permanently planted vineyard that may have natural vegetation established in the inter-rows and set-aside patches.
There is also the issue of functional connectivity. Agricultural patches, especially intensively managed ones, can isolate natural or semi-natural remnants, affecting orthopteran dispersal in the search for optimal conditions. In Hungary, for example, intensive land use in sunflower, maize, and wheat fields led to less diverse orthopteran assemblages with lower species diversity and less conservation value. A continuous three-year monoculture of maize and/or sunflower resulted in especially low average species richness and, in some cases, there were no orthopterans at all in these fields [140].
In a tropical climate, agricultural intensification compared to natural sites can cause greater fluctuations in orthopteran diversity due to periodic disturbances and habitat modifications [141]. Establishment of rice paddies results in most orthopteran species being confined to the semi-natural margins, although some orthopteran species can feed on the rice and can even become pestiferous [142].
In the Indian dry forest, acridid abundance and species diversity were highest in natural habitat with its high plant diversity and absence of human disturbance, including livestock grazing. Increasingly more intense human disturbance caused a drop in grasshopper species richness, diversity, and abundance [116]. Among disturbed sites in Indonesia, post-harvest maize fields that are not treated with pesticides and are overgrown with wild plants, especially natural grasses, and, as a result, support the most diverse and abundant grasshopper assemblages [143] (Figure 2).
3.4. Plantation Forestry and Timber Harvesting
Planted patches of alien Cupressus and Pinus species in a natural grassland matrix in South Africa affected grassland orthopterans differentially. While the edge effects of the Cupressus patches were negligible, those of Pinus were significant due to their much denser canopies. The sunny side of the tree patches hosted significantly richer orthopteran assemblages than the shady side. The Pinus patches had large distances of negative influence, extending 30 m or more on the northern, sunnier side, and more than 30 m on the southern, shady side. In turn, the northern ecotones hosted significantly richer assemblages than the control sites of contiguous grassland, while the assemblages on the austral southern shady ecotone were significantly poorer [144].
Also in South Africa, edge effects in natural grassland conservation corridors separating alien Pinus timber compartments were compared to edges of natural forest. There were different orthopteran assemblages in the two types of edges. The edge effect of both Pinus and natural woodland on the grassland orthopterans was similar, and although depauperate adjacent to the pine trees, they increased away from the tree line, reaching a peak at 16 m into the grassland away from the timber line [145].
In Austria, orthopteran species richness at the Pinus–grassland edge was low. Into the grassland, orthopteran species numbers increased steadily with increasing distance from the forest edge up to about 30 m into the grassland, exclusively due to an increase in the number of acridid species, while tettigoniid species numbers showed no clear relation to edge distance. Increased sunny conditions and soil temperatures were driving factors [146].
In Slovakia, clear cuts of natural beech forest affected orthopteran assemblages differentially over time since tree harvest. Grasshopper species were more abundant than bush crickets in first year clear-cuts. Flighted species occurred in all age categories of clear-cuts, with an asymptote for all species locally present appearing two years after timber harvesting, with assemblages varying among sites according to the composition and structure of the successional vegetation. There was also a positive correlation between the number of orthopteran species and the area of the clear-cut regardless of its age, as well as between the number of species and elevation [147] (Figure 2).
3.5. Urban Environments
Urbanization is a localized increase in human density and activity, which, in general, has an attritional effect of most major insect groups [148]. But often there are urban vegetated green spaces in the form of reserves and parks that support certain levels of insect diversity [149]. Additionally, particularly in suburban areas, gardens are used not only for aesthetic purposes but also for production of human food [150].
Around Berlin, Germany, levels of urbanization and the presence of remnant grasslands were the main drivers of orthopteran species richness, species diversity, and functional diversity. All these metrics decreased with increasing proportion of surrounding hard surfaces, which also changed assemblage composition due to effects of the urban matrix and vegetation characteristics, along with a tendency for the more mobile species to dominate in most urban environments [151].
Similarly, around Paris, France, urbanization had a negative effect on total bush cricket species richness, abundance, and habitat specialization, especially at larger spatial scales. There were differences according to species’ functional traits, with mobile species being more sensitive to urbanization at larger scales than sedentary species, but no scale effect on habitat specialization was detected [152].
In suburban Basel, Switzerland, the number of orthopteran species in moderately disturbed meadows and in intensely disturbed ruderal sites increased with habitat size. In general, large-sized species preferred larger meadows and ruderal sites, with small-sized species less affected. Moderate dispersers and omnivorous species also benefited from increasing habitat size. There was no effect of habitat size on the number of low-dispersal orthopteran species [153].
Around London, UK, at large spatial scales, the number of orthopteran species declined with increasing dominance of arable and urban land uses but not correlated with the area of an individual land cover representing cultivated land, and only weakly correlated with a second land cover representing urban development. Few orthopteran species occupy arable crops or highly urbanized areas. Importantly, while measurement of areas of proportionate land cover is interesting, these area values do not capture ecologically relevant information about variation in the composition of the uncultivated and non-urban land-cover matrices within which the species reside [154].
The importance of small-scale effects was emphasized by Huchler et al. [155], who showed that orthopterans overall are highly affected by several different site-level drivers. These drivers influence the habitats through management-related factors, having a stronger impact on local urban orthopteran assemblages than urban landscape characteristics. This is likely due in part to historical sorting and local survival of the more mobile species with strong dispersal abilities and high habitat tolerance persisting in such disturbed landscapes. In Rome, Italy, orthopteran spatial data were compared over time (1990s and 2010–2022), revealing that local extinction in urban areas is affected by species’ traits. Those with low mobility and fertility, and narrower, more specialized, climate specific niches were more likely to go locally extinct [156].
Among sub-urban grasshopper assemblages in Chiba Prefecture, Japan, three quarters of grasshopper species almost exclusively occurred in semi-natural grasslands and vacant lots, while habitat generalists were largely at the cropland margins, correlating with plant community characteristics and associated current disturbance regimes. Vacant lots, as well as semi-natural grasslands, had the highest conservation value for grasshoppers [157].
3.6. Pollution and Pesticides
As many insect species have essential roles in agriculture, it means that there must be control of pest species. However, in the transition to agroecology there must be concern for the wider insect assemblage with its many non-target organisms. In addition, there must be an awareness that pesticides might be only one of several adverse drivers [158] (Figure 3).
The fundamental issue is that the high sensitivity of many non-target insects to pesticide exposure raises concerns about its impacts and the cascading effects on biotic communities and therefore ecosystem function [159]. Besides concern over pesticides permeating ecosystems beyond the focal crop area, there is the issue of sublethal effects on agricultural beneficial insects through adverse effects on the biology of these organisms in many ways. This requires integrated pest management programs to be directed to the use of safer pesticides or those with lower risk to the insects that participate in agroecosystem functioning. Furthermore, climate change is exacerbating pest infestations and challenging environmentally safe control methods [160,161].
Currently, there are >10,000 major pest insect species globally, with herbivorous insects damaging 18% of world crops [162], with many species having already developed resistance to chemical pesticides [163]. Conventional pesticide risk assessments rely on surrogate species with a primary focus on acute lethality, thus failing to capture the broader impacts on non-target organisms and overall biodiversity while also overlooking the complex interactions between multiple stressors such as pesticide contamination, landscape transformation, and climate change [164]. This means that there is an urgent need for better pesticide risk assessments to include complex sets of interactions, especially those involving non-target organisms, and that integrate ecological and evolutionary complexities, especially with ongoing climate change [165].
There are essentially three issues regarding the role of orthopterans in the transition from conventional agriculture to agroecology. Firstly, some orthopteran species are pests that require control without harming other orthopteran species and other biodiversity (discussed in Section 4.5). Secondly, control of other pest species can be harmful to indigenous orthopteran species and assemblages. Thirdly, the spectrum of orthopteran species is an indicator of good or bad agroecological practice.
Various pesticides are used against the Nantucket pine tip moth (Rhyacionia frustrana (Scudder in Comstock, 1880)) (Lepidoptera: Tortricidae), a native regeneration pest on young loblolly pines (Pinus taeda), and which negatively impacts pine growth. All four pesticides (chlorantraniliprole, dinotefuran, fipronil, or imidacloprid) were tested and found to differentially but significantly reduce ground-inhabiting orthopterans [166].
In Germany, reducing the number of chemical pesticide applications and associated tractor passages, at least in organic viticulture, is important and shows a clear benefit for some orthopterans [167]. Also in Germany, field monitoring revealed adverse effects of pesticide drift on the density of grasshopper assemblages in margins around fields by up to 20 m [168].
Orthoptera individuals can accumulate heavy metals such as Cd, Cr, Cu, Ni, and Zn more than some other insect orders [169], although the extent of accumulation of these metals may vary among orthopteran species [170]. In short, orthopterans can be good indicators of environmental pollution, including when contaminants are passed from plants to them [171] and further up the food chain [172].
There can be an interaction between phenoxy herbicides and loss of plant diversity on the polyphagous grasshopper Pseudochorthippus parallelus. In females, grass diversity increased herbivory, insect nitrogen content, and egg load, while herbicides reduced herbivory but increased the number of offspring, likely mediated by altered plant community composition. In males, grass diversity also increased herbivory, and had positive effects on fat body, muscle volume, and lifespan, but negative effects on body mass. Herbicide negatively affected herbivory in both females and males. In sum, plant diversity and herbicides may shift resource allocation in this grasshopper, indicating complex and unexpected effects of human-induced environmental change through chemical herbicide use [173].
3.7. Impact of Invasive Alien Plants and Animals
Alien plant invasions can impact indigenous orthopteran assemblages (Figure 3). In Arizona, invasion by Lehmann’s lovegrass Eragrostis lehmanniana Nees, 1841 (Poales: Poaceae) reduced overall orthopteran population levels by 10%, a lower impact than on many other insect taxa [174]. Similar results come from Japan, where alien E. curvula reduced the densities of 12 species of grasshoppers with the greatest impact on the habitat specialists [175].
Ecological pressure from alien plants on arthropods overall has the potential to alter broadscale ecological processes, such as energy flow and nutrient cycling, and reduce food resources for insectivores, which can have adverse, cascading effects on threatened grassland ecosystems [176]. This is especially so when the alien plant canopy is dense and shades the grassland, leading to differential species responses according to their specific traits [177].
On the Great Plains, USA, most grasshopper species are widely distributed, and numerically responsive to combinations of environmental conditions expressed at local scales [178]. In Utah, there are complex indirect effects of cheatgrass (Bromus tectorum L. 1753 (Poales: Poaceae)) invasion and grasshopper (Xanthippus corallipes (Haldeman, 1852) (Orthoptera: Acrididae) and Melanoplus confuses Scudder, 1897 (Orthoptera: Acrididae)) herbivory on native bunchgrasses (Poa secunda Presl, 1830 and Elymus elymoides (Rafinesque, 1819) (Poales: Poaceae)), which are determined by neighboring cheatgrass stands [179].
However, certain invasive plants may not necessarily be disadvantageous to grasshopper assemblages. On Idaho rangelands, areas that had been severely disturbed by wildfires and invasion of alien annual vegetation (B. tectorum) had significantly higher grasshopper densities compared to less severely disturbed areas that retained some indigenous sagebrush (Artemisia spp.) cover [180]. Overall, both herbivory and grasshopper performance (body length) were greater on alien compared to native grasses. However, alien grasses that do not share a coevolutionary history with native generalist Melanoplus spp. grasshoppers appear to have lower physical and chemical defenses than native plants. This means that Melanoplus grasshoppers may provide biotic resistance to these alien grasses should they invade natural areas [181,182,183].
In the USA, choice tests using Melanoplus borealis (Fieber, 1853) (Orthoptera: Acrididae) showed that it preferred the alien Dandelion Taraxacum officinale Wiggers, 1780 (Asterales: Asteraceae) and Crepis tectorum L., 1753 (Asterales: Asteraceae) over two native forbs, and a grass species. The grasshoppers reduced the biomass of mature plants, flowers, and seedlings of C. tectorum, but not other forbs, with this alien readily providing a new food resource for generalist-feeding grasshoppers. However, grasshopper densities are not high enough to have significant impact on the weed populations in the wild [184]. However, herbivory on invasive versus non-invasive plants from the genus Centaurea (Asteraceae) feeding trials using M. femurrubrum (De Geer, 1773) (Orthoptera: Acrididae), indicated that the highly invasive plants were fed on the least by grasshoppers, suggesting that they contain feeding deterrents that render them less acceptable than non-invasive plants [185].
However, in South African grassland, grasshopper species richness and evenness responded strongly to local vegetation height and bare ground, while grasshopper assemblage composition also responded to plant species richness. However, there was a major negative impact from invasive alien Sand blackberry (Rubus cuneifolius Pursh, 1813 (Rosales: Rosaceae)) on large-sized grasshoppers and species of conservation concern. Overall, it is important to maintain natural levels of habitat heterogeneity while at the same time control or remove the invasive bramble [186].
Invasive alien vertebrates can also affect orthopteran assemblages (Figure 3). The European rabbit (Oryctolagus cuniculus L., 1758 (Lagomorpha: Leporidae)) has been an invasive species in England for centuries, and its high grazing levels lead to reduced sward height with abundant bare earth areas on high slopes. The intensive grazing resulted in Chorthippus brunneus (Thunberg, 1815) (Orthoptera: Acrididae) adults being the only species that was abundant in these areas through provision of short swards and bare earth as basking and egg-laying microhabitats. Aspect was also important, with significantly more grasshopper nymphs and adults on the warmer, south-facing slope than on the northern slope, promoted by soil slippage creating warm microclimates preferred by the nymphs. However, other species like Pseudochorthippus parallelus and Conocephalus fuscus (Fabricius, 1793) (Orthoptera: Tettigoniidae) were disadvantaged through loss of taller vegetation from overgrazing [187].
3.8. Climate Change and Synergism with Other Change Drivers
In central Europe between the years 1980 and 2019, orthopterans decreased in land occupancy by 41%. These changes were due to recent climate and land-use changes, illustrating the importance of temperature preferences and habitat specificity as drivers of species trends [188] (Figure 3). Also, in central Europe, prior to 1990, most orthopteran species suffered a large amount of range loss, with habitat specialists declining more than generalists with equal mobility. Range retractions were mostly related to species associated with farmland. Since then, climate change has been the main driver, with the distribution of less mobile species generally remaining stable, and highly mobile species even expanding their ranges, irrespective of their habitat specificity, especially among thermophilic species [189].
Also in central Europe, a comparison of grasshopper assemblages between 1995 and 2012 showed strong changes in environmental conditions and grasshopper assemblages. In contrast to grassland verges, grassland patches suffered from severe habitat loss. More than a quarter of all grassland patches were converted to other uses such as maize, which homogenized the landscape. Furthermore, summer temperatures increased by 1.1 °C. Owing to high connectivity of grasslands, thermophilous and habitat generalist orthopteran species with low or high dispersal ability were able to track global warming. Overall, availability of suitable habitats and climate warming were the major drivers of grasshopper assemblage shifts [190]. Furthermore, in the remaining grasslands in 2012, irrespective of their protection status, orthopteran species richness mostly increased, except for low dispersal species in unprotected grasslands [191]. Among central European grasslands, climate warming has led to biotic homogenization of orthopteran assemblages associated with abandoned wet grasslands and interacts with land-use intensification in mesic grasslands. The outcome is a tendency towards a dominance by more widespread species. In contrast, common pastures are characterized by high heterogeneity of landscapes and low-intensity management and, as a result, are more resilient to the effects of climate warming [65].
In Italy, the spatial responses to climate change in two Oecanthus tree crickets (O. pellucens (Scopoli, 1763) and O. dulcisonans Gorochov, 1993 (Orthoptera: Oecanthidae)) show ecological and morphological resemblance, and partial range overlap, although with species-specific associations with bioclimatic variables related to temperature and rainfall. Climatic modeling predicted responses between the two species, with great range loss for O. pellucens and slight expansion for O. dulcisonans under future scenarios. Importantly, O. pellucens is a significantly more specialized species in its climatic niche. At a regional scale, interspecific competition likely will play a strong and asymmetrical role in determining species’ presence, with only O. pellucens being significantly affected by O. dulcisonans, but not vice versa. These findings underscore how, within cryptic species complexes, competition dynamics and niche specialization may be key in determining winners and losers in the face of climate change [192].
El Niño Southern Oscillation events can also affect the interaction between livestock grazing and vegetation communities and grasshopper assemblages. In the arid southwest of the USA an evaluation of the interactive effects of short-term livestock grazing and climate variation on plant and grasshopper assemblages and species abundance indicated that livestock grazing greatly changed plant and grasshopper species assemblages during the wet years, following shifts in plant communities. Overall, grazing effects on vegetation and grasshoppers were significant during years with high rainfall, plant production, and grasshopper abundance, but not in years when rainfall, plant production and grasshopper abundance were all low [193].
Climate change in the USA has delayed grasshopper development in high-elevation, season-limited grasshopper populations, especially for early-season species, but has advanced development in populations at lower elevations. Population, elevation, and temperature interact to determine development time. Population differences in developmental plasticity often result in variability in phenological shifts among adults. These results suggest that it is important to consider the full life cycle relative to local adaptation and plasticity when assessing the effects of climate change [194].
As regards individual grasshopper species and their specific traits, increased temperature has greatest effect on mid- and late-season species, resulting in certain grasshopper species aggregating toward the center of the developmental season. Increased rainfall may offset the effect of warming on egg development and balance warming-induced risks to grasshopper populations [113]. Furthermore, there can be differences in response to warming among different populations in a species. For example, Chorthippus dubius (Zubovski, 1898) (Orthoptera: Acrididae) showed distinct thermal adaptations across latitudinal gradients, affecting the number of accumulated degree days required to offset the effect of elevated temperatures on egg development at lower latitudes. This adaptive response nevertheless led to the preservation of a univoltine life cycle across all populations [195].
4. Orthopteran Conservation in a Rapidly Changing World
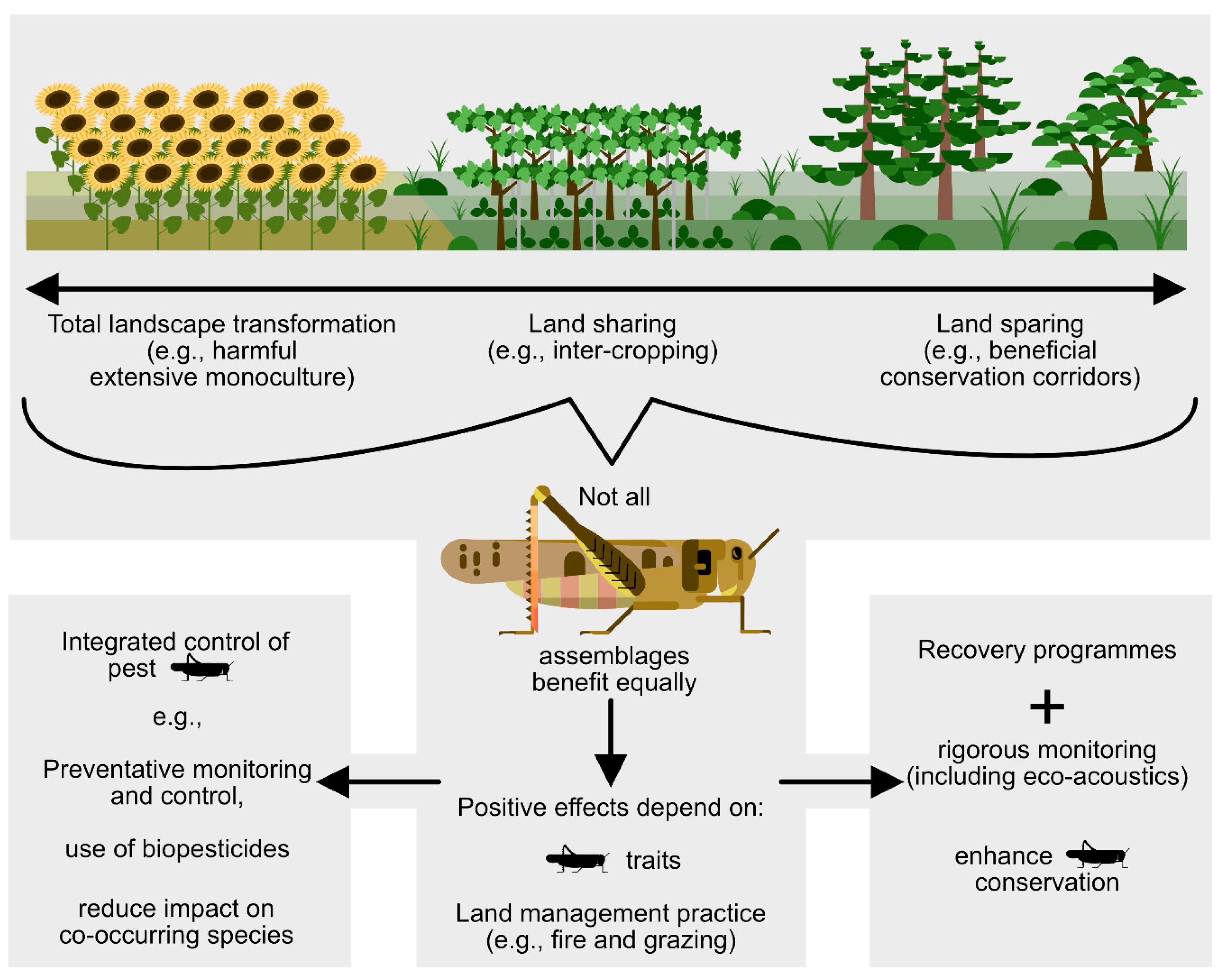
Spatially immediate human impacts (e.g., landscape transformation, pesticide use) can be addressed at various local spatial scales, with due consideration given to orthopteran species traits. However, climate change and attendant extreme weather events, as with all biodiversity conservation, are best addressed at the global scale. Figure 4 is a summary and guide for Section 4.1, Section 4.2, Section 4.3, Section 4.4 and Section 4.5.
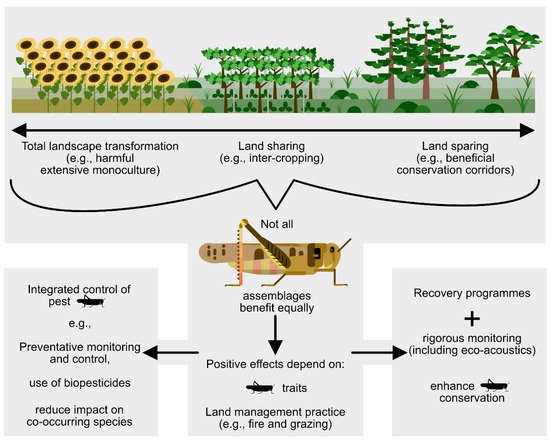
Figure 4.
Management for orthopteran conservation is relative to spatial and temporal scales of activity and the traits of the various species.
4.1. A Question of Spatial and Temporal Scales
In the simplest terms, there are two main issues associated with the integration of agriculture and orthopteran conservation. The first is to set-aside land, and the second is to manage host systems optimally to maintain both agricultural production and orthopteran assemblages.
Considering the spatial and temporal scales at which both set-aside initiatives and management take place is important. At large spatial scales, larger areas can be set aside as land sparing, principally involving the establishment of protected areas. Further valuable information is obtained when data are gathered at small spatial scales (i.e.,1 km2) and where feasible [196].
At smaller spatial scales land sharing can be practiced at the field level when certain areas are set aside for production and other areas for maintenance of near-natural levels of biodiversity. However, land sharing and land sparing are not a binary concept but a spectrum, and they can also be applied simultaneously in a region. For example, biosphere reserves are composed of three zones: a central core devoted to biodiversity conservation, a surrounding buffer zone where there are limited agroecological and sustainable production systems, and an outer transition area where there is more extensive agroecological cultivation as well as patches of remnant habitat.
In addition to spatial land planning, there is also management of the landscape. Besides the mechanical practice of cultivation, there are other factors to consider, which include, for example, the use of environmentally sensitive integrated pest management procedures and control of invasive alien plants.
Temporal aspects to consider include the transition period from conventional agricultural practices to more environmentally sensitive ones. A fundamental aim is to improve sustainability and ecological resilience in a changing world to provide a means for natural processes to continue. These include, for example, hydrological and soil processes, as well as ensuring the presence of well-vegetated refuges to support natural levels of biodiversity and biotic interactions.
4.2. Importance of Set-Aside Areas at Various Spatial Scales
Biogeographical studies on orthopterans have great value in determining hotspots of orthopteran species diversity, including identifying areas where locust control may have to consider risks to rare species [197]. Across Switzerland, a hotspot study using 1 ha quadrats compared the extent to which common and threatened grasshopper species represent overall grasshopper species richness. Overall, grasshopper species richness was well represented by both the number of common and of threatened grasshopper species. In turn, species richness hotspots not only featured the most common grasshopper species, but they even supported more threatened species than the rarity hotspots. The combination of rarity hotspots and hotspots of common species featured more species than the other combinations of hotspot types. However, the gain from combining two hotspot types compared to the single-hotspot approach was low (maximum of three species). As 24% of the species were not present in any of the hotspots, these species would require species-specific action plans. As rarity hotspots were in areas that are subject to landscape change, species richness in rarity hotspots may decrease in the future in the absence of species action plans [196].
Although formally protected areas play a crucial role in the conservation of orthopteran species, particularly narrow range endemic species or as refuges for declining species, it does not mean that protected areas necessarily afford total protection of all orthopteran species present in a specific region [156,191,198,199,200,201,202,203,204,205].
Biosphere reserves provide great opportunities for the conservation of orthopterans. In Kogelberg Biosphere Reserve, South Africa, all three zones (core, buffer, and transition) were equal in tettigoniid diversity, illustrating that agroecological farming in the buffer and transition zones was not adversely affecting the assemblages [206].
In the Cape Winelands Biosphere Reserve, South Africa, grasshopper abundance was highest in agroecological vineyards with well-vegetated inter-rows compared to either natural fynbos or deciduous fruit orchards. Nevertheless, orthopteran species richness, diversity, and evenness were highest in fynbos, followed by vineyards and then orchards. But significantly, orchards had no unique species, vineyards had a few, and the fynbos many, composed mostly of flightless endemics. This means that retaining patches of good-quality fynbos in transformed land mosaics is essential for maintaining the endemic orthopteran species [207]. However, with increasingly high levels of fynbos transformation, there is replacement of endemic grasshopper species with more widespread generalist species [29].
As well as land-use type and species traits, seasonality also plays a significant role in these orthopteran spatial patterns, with highly mobile, generalist feeders being abundant and widely dispersed across the landscape, especially late-season when they utilize high plant productivity in the vineyards. These actively mobile generalists benefit by being able to occupy transformed areas, with populations greatly increasing during the late season. The low mobility specialist feeders only preferentially occupy natural fynbos vegetation and are disadvantaged by not being able to move far, while also lacking their specific host plants in the transformed areas. Overall, this suggests that it is essential in the Cape Winelands Biosphere Reserve to maintain as much fynbos functional connectivity as possible to maintain effective population sizes of endemic grasshoppers [208]. There are also differences among the local endemic grasshopper species in the genus Euloryma (Orthoptera: Acrididae), with two species, E. umoja Spearman, 2013 and E. ottei Spearman, 2013, able to tolerate some landscape transformation away from natural fynbos. However, E. larsenorum Spearman, 2013 and E. lapollai Spearman, 2013 only live in fully natural fynbos [209] (Figure 5).

Figure 5.
Conventional vineyard (left) almost devoid of orthopterans, compared with an agroecological vineyard (right) in the Cape Winelands Biosphere Reserve, rich in orthopteran species. Photos: Michael Samways.
Choice of set-aside procedure can depend on the local topographical context. In contrast to the South African vineyards, those on steep slopes in central Europe support woody vegetation. Vineyard abandonment reduced orthopteran diversity at the local and landscape scales through a decrease in habitat quality, especially for open-habitat species. Actively managing inter-rows through retention of open vegetation supported heat-adapted grasshoppers, while on terrace embankments, extensive management for taller vegetation benefited ensiferans. However, short and mulched vegetation in vertically oriented vineyards favored dominance by just one grasshopper species [210].
In a South African summer rainfall area, networks of large conservation corridors can effectively extend the reach of protected areas into agricultural or afforested landscapes, with orthopteran species richness and abundance significantly higher in the edge zones of both corridors and protected areas compared to sites of similar size in the interior zones of both land types [145], with increased landscape heterogeneity elevating orthopteran species richness, assemblage composition, and species turnover [211], overall suggesting that these conservation corridors should be as wide as possible [212].
In these same conservation corridors, grasshopper species responded strongly and consistently to site area size, time since the last fire, and inconsistently to the proportion of short grasses, although varying among species. But as orthopteran assemblages are more sensitive to habitat quality rather than habitat isolation, and when high habitat quality is being maintained, these conservation corridors are highly effective in conserving the overall orthopteran assemblage [213], with certain orthopteran species being particularly good indicators of conservation corridor quality [214]. However, these corridors have constrictions, or some may be blocked off at one end, for various production reasons. Neither the pinch points nor the cul-de-sacs had any adverse effect on orthopteran abundance or species richness [215].
In the same system, wide corridors along an elevation gradient with different aspects supported different grasshopper assemblages and enhanced species turnover. Sensitive species occurred mainly at high elevations on northwest-facing slopes in wide corridors that are functionally connected to large high-quality remnant grassland patches [216]. Overall, the various studies above clearly indicate that good-quality conservation corridors play a significant role in maintaining orthopteran assemblages in a timber plantation mosaic, especially when landscape heterogeneity is high (Figure 6).

Figure 6.
Agroecological pine plantation forestry in South Africa, where large networks of wide remnant grassland corridors are rich in orthopteran species, with their diversity increasing when rivers (left) and naturally small indigenous forest patches (right) are also included in the corridors. Photos: Michael Samways.
Small refuges in agricultural landscapes can also play an important role in supporting orthopteran assemblages. For example, in Hungary, ditch banks are a suitable habitat for most local orthopterans, including rare and threatened species, with abundance levels being especially significant in determining the quality of the ditch bank. While caeliferans preferred more open habitats, ensiferans favored wooded areas [217].
4.3. Maintaining Orthopteran Populations and Assemblages Through Better Land Management
Design and management of set-aside land are entwined, with subtle differences in either or both leading to changes in orthopteran assemblages, even in the same geographical area, as in the plantation mosaics with conservation corridors in South Africa [145,213]. Furthermore, maintenance of functional connectivity is dependent on the availability of high-quality habitat, and when shaped by fires, can have a significant effect on range-restricted species. However, not all high-quality habitat is functionally connected, often because of fire exclusion or the effect of invasive alien plants. This means that prescribed burning should be used to maintain functional connectivity through burns every 2–3 years, guided by vegetation assessment. While wide and extensive corridors at high elevation are a conservation priority, narrow corridors and stepping-stone habitats, as well as grassland buffers next to dirt roads, also help improve functional connectivity [216].
Timing of burns is also important in this summer rainfall area. Burning mid-winter is more favorable for orthopteran assemblages than burning in autumn or after the first spring rains. Mean grasshopper species richness is highest following mowing after the first spring rains, and the mean number of individuals highest following mowing in early summer. When comparing annually burned plots with annually mown plots, orthopteran abundance and species richness are highest in the burned plots. These results suggest that a rotational winter burning program, which is practical under African conditions, is recommended for orthopteran conservation [218].
Due consideration of both landscape design and management is seen in the rapidly declining European grasshopper Arcyptera fusca (Pallas, 1773) (Orthoptera: Acrididae) in Hungary. Traditional extensive habitat management (grazing and mowing) and the leaving of uncut refuges in mountain meadows are essential for maintaining effective populations. This also opens a way for translocation of the species to these well-designed and managed sites [219]. Traditional grazing at moderate densities in the Bavarian pre-Alps also contributes greatly to the conservation of two threatened species, Pseudochorthippus montanus (Charpentier, 1825) (Orthoptera: Acrididae) and Stethophyma grossum [220]. In the Białowieża Forest, Poland, most orthopteran and other insect species diversity improves when human land use is replaced by grazing and browsing by wild herbivores [221].
In a mountainous part of Germany, site conditions and management go together for orthopteran diversity and abundance conservation. Warm, south-facing pastures maintained more diversity than north-facing pastures, but both had greater diversity than extensively used meadows. Bare ground cover and forb cover were also important, especially for south-facing pastures. Furthermore, grazing by horses and cattle provided better management for maintaining high orthopteran diversity in calcareous grasslands than did mowing [222].
A hydrological spectrum of peat bog, fen, mesic grassland, semi-dry grassland, and dry grassland in Central Europe revealed strong differences in environmental conditions and in assemblage composition of threatened orthopteran species in mire and grassland habitats. Land-use intensity was the key driver of habitat heterogeneity and of orthopteran species richness. These findings suggest that the way forward is to instigate continuous large-scale, low-intensity cattle grazing from spring to autumn, as it promotes high habitat heterogeneity for these sensitive orthopterans [223].
In a mountainous area of Greece, the three main environmental factors shaping diversity patterns are grass height, woody vegetation cover, and bare ground cover. The implications for conservation are that it is important to focus on areas with high species richness, with attention given to biodiversity-inclusive grazing schemes to reduce forest encroachment and to restore mountain grassland quality following cattle overgrazing [224].
Similarly, in the arid Karoo of South Africa, seasonal and rotational grazing maintained the highest level of grasshopper diversity [134], notwithstanding that topographic heterogeneity has a remarkably strong effect on local grasshopper spatial distribution [225]. An intense study of a highly responsive grasshopper species, Orthochtha dasycnemis (Gerstaecker, 1869) (Orthoptera: Acrididae), showed that the summit of a mesa, through inaccessibility to livestock grazing, was effectively a conservation refugium for the species, albeit at different abundance levels among years, due to varying rainfall levels. In effect, the mesa acts as a conservation island and refuge that would otherwise be altered by heavy livestock grazing on the surrounding flatlands [226]. Similarly, in the Drakensberg Mountains, rocky hilltops are a major refuge for grasshopper assemblages, as the rockiness preserves relatively ungrazed vegetation patches, while also conferring protection from predators. In the dry winter, when many grasshopper species are still active, the rocky hillsides and summits also provide thermal refuges [100].
In the Carpathian lowlands, linear ruderal habitats and less intensively cultivated arable lands (at least to 5%, optimally 8–10%) maintain orthopteran diversity, especially through landscape- and crop rotation planning that considers spatial and temporal distribution of refuges [140]. In southern Italy, habitat loss from conversion to arable fields leads to local loss of orthopteran species, especially those that are sedentary and require large habitat patches. The best management response is to improve habitat diversity along with cutting or burning small areas while also reintroducing sheep to abandoned grasslands. These actions improve orthopteran species diversity in both small and large patches [227]. In the Italian Alps, mown meadows and young abandoned meadows (3–5 years since last cut) result in colonization by forest trees and reduced orthopteran diversity. Furthermore, effective agri-environment schemes help maintain extensive semi-natural hay meadows employing a mowing regime of 3–5 years, which reduces mortality due to mechanical mowing and halts forest invasion [228].
But sometimes mowing must not be too frequent, as shown by findings from Swiss mountains where frequent mowing is one of the main threats to orthopteran assemblages in managed grasslands. Also, it is important to retain habitat refuges (e.g., woody vegetation or unmown areas) close to grassland patches, to enhance orthopteran species richness and to compensate for the detrimental effect of mowing [229]. Similar findings come from the Poľana Biosphere Reserve in the Western Carpathians, Central Slovakia [230] and the Hrubý Jeseník Mountains, Czechia [231]. Furthermore, orthopteran species mostly readily colonize newly available optimal habitats when embedded in species-rich grasslands [232]. In Cyprus, rural mosaics and forests are best for orthopteran conservation [204]. In tropical Cameroon, forest management by reforestation, reduction in slash-and-burn agriculture and wood cutting, restores the original grasshopper assemblages and general environmental health [233].
In northeastern Italy, at the large spatial scale, orthopteran diversity would benefit from maintaining extensively managed meadows throughout the whole region to improve the regional orthopteran species pool. This increased connectivity encourages species occupancy and population persistence in patches, particularly for sedentary species [32].
Changing crop variety can also make a difference. In southwestern Germany, fungus-resistant grape varieties have reduced pesticide applications by 44% in the transition away from conventional and towards organic production. Although total densities of herb-dwelling orthopterans did not differ between grape varieties in conventional vineyards, they were 2.9 times higher under organic management. In contrast, total densities of vine-dwelling orthopterans were mainly driven by the dominant species Phaneroptera falcata (Poda, 1761) (Orthoptera: Acrididae). Densities were similar between grape varieties in organic vineyards but were 1.4 times higher in classic grapes under conventional management. Furthermore, the type of management system in the semi-natural habitats within a radius of 500 m in the surrounding landscape influenced species composition. Overall, these results indicate that under organic viticulture, reducing the use of non-specific pesticides is important for biodiversity conservation and that the cultivation of fungus-resistant grape varieties can play a significant role in this direction [167].
Overall, the findings above align with Gardiner’s [84] conclusion that the over-arching principle for grazing management should be to establish a heterogeneous grass sward with a range of sward heights and bare earth for oviposition and basking. In more extensive systems, patches of scrub provide woody vegetation habitat for species such as bush crickets. This means that high habitat heterogeneity should lead to the highest species richness.
4.4. Recovery Programs
Active recovery programs are intimately related to the set-aside and management actions discussed above. Here, though, we focus on further actions where targeted conservation actions are undertaken for orthopteran assemblage and species recovery.
In Switzerland, improving agricultural field margins by using seed mixtures with 60 to 80% forbs, increased orthopteran abundance and species richness considerably, approaching levels in biodiversity conservation areas [234]. Similarly, in Hungary, sown grass strips in intensive farmland benefited orthopteran assemblages by substituting for loss of prime grassland habitats and are high-quality habitats compared to remaining disturbed grasslands. Sown mixtures and grass cutting and harvesting were the main factors driving orthopteran density. Mixtures composed of mixed grasses without legumes were the best, complemented by hay harvesting. Grasshopper species richness also increased in the sown grass strips when they were in landscapes with low grassland field availability but high connectivity. Enhanced use of sown grass strips by grasshoppers can be seen as a direct effect of habitat improvement and management actions, but also a response to the rarity and instability of the remaining grassland habitats in these agricultural landscapes [136].
A large, protected area in the South African Karoo was allowed to recover after replacing cattle-grazed farmland with an indigenous mammal parkland. The grasshopper assemblage was assessed 62 years later and compared with the livestock-grazed area outside the park. While species richness inside and outside the park was not significantly different, abundance inside the park was significantly higher, indicating that long-term restoration using indigenous ungulates provided a better environment for grasshoppers to flourish than did livestock. The problem with intensive livestock grazing and trampling is that they lead to bush encroachment and reduction in grass cover and/or disappearance of several grass species. However, inside the park over time the indigenous herbivores improved vegetation composition and structure, particularly grass height and percentage cover, especially beneficial to the grasshoppers [235].
In the montane heathlands of Germany, orthopterans responded rapidly to restoration activities following establishment of Ericaceae dwarf shrubs. Most species that occurred on the restoration sites were early and mid-successional species. However, later colonization by late-successional species depends on further increases in density of the heathland vegetation following appropriate management [236].
Fifteen years of natural forest restoration on Rodrigues Island, Mauritius, increased by four to seven times the density of the endemic orthopterans, depending on the restoration scheme. However, restoration of an oak forest in southern Spain only benefitted the ensiferans, not the caeliferans [237].
It is possible in some cases that certain grasshopper species may inhibit restoration activities through preferential feeding on native grasses over alien ones [238,239]. However, research to date overall suggests that acridid grasshoppers have the potential to contribute to biotic resistance in native plant communities through grasshoppers preferentially feeding on invasive alien plants [183], even when the plants are closely related [240].
At the single-species level, Hochkirch et al. [241] translocated individuals of the local threatened Gryllus campestris L., 1758 (Orthoptera: Gryllidae) in Germany. Success depended on the recipient locality containing sufficiently large areas of suitable habitat and a high number of wild juveniles from a closely located and large source population that were released during a climatically favorable period. Furthermore, management and restoration of habitats, as well as continuous monitoring, were of crucial importance to ensure the success of the translocation project, along with good co-operation between conservationists, authorities, foresters, farmers, financiers, and scientists. Importantly, the translocation did not lead to a significant loss of genetic diversity, when compared to the source or other natural populations. This suggests that translocation of many nymphs from different subpopulations is a suitable method to decrease the loss of genetic diversity and reduce the risk of inbreeding. Furthermore, the translocation had no negative effect on the source population [242].
4.5. Integrated Locust and Grasshopper Management
While several well-known acridid species (locusts) swarm and migrate, or can reach high densities on rangelands, other orthopterans (e.g., Ruspolia differens (Serville, 1838) (Orthoptera: Tettigoniidae) in East Africa; [243]) also cause economic damage and must be subject to control measures. In addition to the 24 locust and non-model locust species listed by Song [244], about 400 grasshopper species can be considered as potential agricultural pests, although to very different degrees [245]. The problem with traditional control using chemical pesticides is the impact on non-target organisms. However, more environmentally benign options are available. This was seen in the case of attempts to control Schistocerca gregaria during massive outbreaks in the Horn of Africa in 2019–2021. Firstly, environmental monitoring was largely neglected, with only limited surveys conducted in Kenya and Ethiopia. Overdosing with fenitrothion with a 960 g/L formulation in Kenya led to non-target mortality, including birds and honeybees. In Ethiopia, chlorpyrifos and malathion applications coincided with a honey production decline of 78% in 2020 compared to prior locust control. However, application of the fungal biocontrol agent Metarhizium acridum (Driver and Milner) J.F. Bisch., Rehner and Humber, 2009 (Hypocreales: Clavicipitaceae) on nearly 253,000 ha in Somalia was a breakaway from previous campaigns and successfully controlled both hopper bands and swarms with no environmental effects detected [246].
Metarhizium acridum has been recorded to have no adverse effect on ants, beetles, and grasshopper-feeding birds, which will even feed on M. acridum-treated cadavers. In contrast, these birds move away from plots treated with chlorpyrifos and fenitrothion, risking a secondary locust outbreak [247]. In Ethiopia, M. acridum was target-specific against Zonocerus variegatus (L., 1758) (Orthoptera: Pyrgomorphidae), Cyrtacanthacris spp., Diabolocatantops spp., Oedaleus spp., while non-target bugs, lepidopterans, mantids, and spiders showed no infection [248]. Other non-target taxa such as hymenopterans, homopterans, asilid flies, and ground-scavenging arthropods are also not directly infected and killed under field conditions. Furthermore, large-scale mycopesticide treatment appears to enable natural control to act as a buffer against the recolonization of treated areas once pest populations have been reduced. This contrasts with the effect of chemical pesticides, such as fenitrothion, which may cause grasshopper resurgences. In short, biopesticides offer a more environmentally safe alternative to chemical pesticides for locust and grasshopper control [249,250].
Langewald et al. [251] provide an extensive list of non-target (excluding orthopterans) species in response to M. acridum application and dosages. However, of concern for orthopteran assemblages, is that non-target orthopteran species were not included in the assessments. Prior (1997) though, provides information on effects of M. acridum on some non-target orthopterans, concluding that there is none, low, or moderate susceptibility in the tested non-target species, while it is high for the target species.
Furthermore, M. acridum can remain viable in the environment in the USA for >30 days [251,252], in Mexico for 66 weeks and only disappearing after 8 months [253], and in Australia for 3.5 years [254]. While such figures are beneficial for controlling the pest species, they are of concern for the overall orthopteran assemblage, especially as more persistent formulations are being sought [255,256].
Besides M. acridum, the microsporidian Nosema locustae Canning, 1953 (Protozoa: Microsporidia) (synonym: Paranosema locustae) is also being used for locust control with reduced environmental and non-target damage compared to chemical insecticides [257,258]. Combinations of the two pathogens have also been used to control target orthopterans [259].
Shi et al. [260] investigated the non-target effects of N. locustae and concluded that application of the pathogen had no effect or increased population levels of the natural enemies of grasshopper pests in Chinese grasslands. Although the general mode of transmission of the pathogen is cannibalism of infected grasshoppers, healthy grasshoppers could also be infected in the presence of the predatory ground beetle Pterostichus gebleri (Dejean, 1828) (Coleoptera: Carabidae) which carried microsporidian spores, despite the spores being non-pathogenic to the beetle. Significantly, this mechanism aids spread of the pathogen into untreated areas [261]. Of concern, however, at least for non-target indigenous species, is that the pathogen remains infective in application areas and beyond for several years in China [262], while also having a persistent adverse effect on indigenous melanoplines in Argentina for several years after the application of the alien pathogen [263].
Lockwood [264,265] has argued that biocontrol on North American grasslands, using foreign natural enemies against pestiferous grasshoppers, is not advisable as the risk of upsetting general orthopteran assemblage composition, abundances, and nutrient cycling is too high. Alternatives have since been implemented. The first consideration is that grasshopper outbreaks are intermittent and cyclical, and once these outbreaks have occurred, there is little chance of reducing grasshopper damage. It is better to minimize pest grasshopper damage by acting before outbreaks occur using a multidisciplinary management system. Preventative measures include good grazing management and prescribed fires that improve grassland condition, although they must be tailored to each ecosystem type [266].
In turn, as emphasized for Chinese grasslands, monitoring and prediction of potential outbreak areas can be improved by identifying major environmental factors that drive outbreaks between low- and high-risk areas [267]. Furthermore, this type of preventive monitoring, which is designed to enable intervention at the earliest signs of an outbreak and before a population begins to cover larger areas, underpins the current strategy for locust control and emphasizes the importance of early detection in minimizing ecological and agricultural impacts [17].
In Patagonian wetlands, feeding experiments were used to determine orthopteran vegetation consumption rate and preferences, leading to a spatially explicit stochastic model based on individuals. The model demonstrates that the potential impact of these species depends on both their densities and the wetland’s vegetal biomass. Knowing this background enables determination of pest thresholds and development of early warning strategies that promote the conservation and management of the natural environment [268].
In difficult terrain, any form of insecticide application may be difficult. In a mountainous part of China, the achievement of a balance between control of orthopteran pests while being cognizant of the needs of rare orthopterans, a method termed ‘controlling grasshoppers and rearing birds’ was introduced. The method involves using chickens to reduce grasshopper populations to control the pest species while avoiding local extinction of non-target species, such as endemic or threatened orthopteran species, while also providing free-range chickens for human consumption [269]. However, further research is required to ascertain whether certain orthopteran species are at risk of decline from free-range chicken farming.
4.6. Congruency of Orthopterans with Other Animal Taxa
Land set aside specifically for other animal taxa can often benefit orthopteran assemblages and specific species. The first French Natural Asset Reserve was created in the plain of La Crau (Southeastern France), the last semi-arid steppe in Western Europe. The initial emphasis was to conserve rare and protected steppe birds. Rehabilitation of the area saw rapid recovery of typical orthopteran assemblages measured in terms of species richness, abundance, and composition. Importantly, the project also helped restore habitat of two rare and threatened species, the Crau Plain grasshopper (Prionotropis rhodanica Uvarov, 1923 (Orthoptera: Pamphagidae) and the Predatory bush cricket (Saga pedo (Pallas, 1771) (Orthoptera: Tettigoniidae)) [270].
The Dadia reserve, Greece, was established to protect the Black vulture (Aegypius monachus) and other raptors. This bird protection project also conserved 39 orthopteran species, including the rare species, Paranocarodes chopardi Peshev, 1965 (Orthoptera: Pamphagidae). Interestingly, the surrounding buffer zone of the reserve was far more important for orthopteran conservation. Management recommendations in the buffer zone include maintenance of forest heterogeneity, the enhancement of periodical livestock grazing, and the use of nine indicator species and P. chopardi in the reserve monitoring program [271].
Prescribed burning is practiced as a management tool to improve habitat of the threatened caprine species, Nilgiri tahr (Nilgiritragus hylocrius Ogilby) (Artiodactyla: Bovidae), at a large scale (50 ha grids) in Eravikulam National Park, Western Ghats, India. When comparing this large scale burning with small scale burning (10 m2), there was a negative response of orthopterans to burning of larger contiguous areas in terms of their recovery after fire events. In contrast, burning of small patches in a mosaic pattern enabled rapid recovery of grasshopper assemblages, with the possibility of optimizing burn management to the benefit of both the tahr and the orthopterans [272].
Stemming from congruency at the local scale, conservation of butterflies can go hand in hand with orthopteran conservation in remnant corridors in a plantation mosaic. In a summer rainfall mountainous area of South Africa, plants, but particularly degree of rockiness, greatly influences grasshopper local distribution patterns [273]. In turn, these landscape features produced congruence between grasshoppers and butterflies, modified by elevation [274]. Similar findings come from a nearby less mountainous area. However, environmental variables were less influential on the congruency between butterflies and orthopterans, which showed in-step turnover of assemblage composition with differences in vegetation types [275].
The effects of five management regimes associated with German fenland were investigated. Butterflies and grasshoppers showed greatest species richness, diversity, and numbers of threatened species on moist meadows, but were adversely affected by tall vegetation. Overall, drained, intensive grassland was not suitable for preserving fen-specific assemblages, while extensively managed moist meadows harbored overall the highest numbers of threatened species [276]. Yet elsewhere in Germany, in a grassland/forest mosaic subject to different grazing intensities, increased vegetation height benefited butterflies, whereas grasshopper species richness was more strongly related to vegetation heterogeneity [277].
Lastly, and importantly, we must consider the importance of multi-taxon approaches for the assessment and monitoring of levels of ecological integrity. Not least because the move to agroecology and improved conservation generally considers biodiversity in its entirety. Thus, the use of a range of carefully selected taxa may be the most expeditious way forward and may well include orthopterans along with other taxa [278,279].
4.7. Non-Acoustic Sampling of Orthopterans
4.7.1. Standard Sampling Methods
The assessment and monitoring of changes in state or status of environmental conditions, population levels, species dispersion patterns, and assemblage composition over space and time are fundamental for guiding decisions and management for an improved agroecological approach. In this regard, practical sampling methods for orthopterans are discussed by Sperber et al. [280] and Giuliano and Rizzioli [281], determination of main environmental drivers of orthoptera assemblages change by Wei et al. [282], and development of a generalized assessment and monitoring method for tettigoniids by Thompson et al. [283].
4.7.2. Emergent Airborne Technologies
Novel airborne technologies are enabling great advances for improved assessment and monitoring. Among these is the use of drones and satellite mapping for insect conservation [284]. The advantage of satellite mapping is that it can be used to determine environmental variables, such as habitat, microclimate, and light pollution, while capturing data on topography, vegetation structure and composition, and luminosity at spatial scales appropriate to insect studies [285].
In the case of orthopterans, remote sensing and machine learning can identify threats to them from managerial legacies [186] and provide strategies to conserve irreplaceable species through identifying optimal habitat relative to the level of functional connectivity [216], and challenges to this connectivity [215].
Use of remote sensing for locust management has advanced greatly, mostly using earth-observation sensor systems, with the focus mainly on the desert locust (S. gregaria), the migratory locust (L. migratoria), and the Australian plague locust (Chortoicetes terminifera (Walker, 1870) (Orthoptera: Acrididae)). In terms of applied sensors, mostly the Advanced Very-High-Resolution Radiometer (AVHRR), Satellite Pour l’Observation de la Terre VEGETATION (SPOT-VGT), and Moderate-Resolution Imaging Spectroradiometer (MODIS) have been used, as well as Landsat data focusing mainly on vegetation monitoring or land cover mapping [286]. Application of geomorphological metrics and radar-based soil moisture data is comparably rare despite previous recognition of their importance for detecting locust outbreaks [287,288]. Recently, Soil Moisture and Ocean Salinity (SMOS) satellite imagery was used for soil moisture estimates for desert locust preventive management, and a Soil Moisture product at 1 km resolution was integrated into the national and global Desert Locust early warning systems [289,290].
Early intervention effectively prevents the development of locust plagues [291]. However, detection of small but increasing large swarms is challenging, but is being overcome, for example, by using a space-borne hyperspectral sensor. It is an optical sensor that measures the reflectance over hundreds of wavelengths and can identify significant components associated with swarm development beyond that of the human eye [292].
4.7.3. Emergent Genomic Advances
Genomic advancement provides much insight into orthopteran conservation initiatives. Improved phylogeny elucidating orthopteran taxonomic relationships and resolving clade divergent dates [5] rests on increasingly refined genetic barcode data gathered and housed in genomic libraries [293,294,295,296,297]. Meanwhile, whole mitochondrial genomes of certain species have been resolved, helping determine the genetic relationships between certain species [298].
Biogeographic distributions of putative morphological species are also being resolved using genomics [299], leading at times to the discovery of genetically differentiated cryptic species in different taxa in various parts of the world [300,301,302], refugial effects on speciation [303], and ongoing speciation among certain species [304].
Importantly for conservation, species are being rediscovered through genomic investigation after being thought to be locally extinct, underscoring the need for preserving certain ecosystems [305]. Also, genomic studies on orthopteran populations of a species can be used to predict potential changes in abundance [306]. Furthermore, risks associated with sampling live individuals of rare and threatened species can be overcome by extracting genomic DNA from exuviae [307]. Orthopteran gut microbiomes are also being identified using metabarcoding [308].
Genomic studies relevant to agroecological advancement also include risks of genotoxicity by the herbicide paraquat [309], the organophosphate dimethoate [310], fertilizer pollutants [311], and from radioactive contamination [312]. Furthermore, the grasshopper Aiolopus thalassinus Fabricius, 1781) (Orthoptera: Acrididae), while having been widely used as an ecotoxicity indicator species of heavy metal pollution [313], has now been identified according to its complete mitochondrial genome, useful for environmental assessment and monitoring [314].
Understanding the diet composition of herbivorous orthopterans is essential for the conservation of threatened species and their habitats. By DNA metabarcoding the feces of the Critically Endangered Isophya beybienkoi Maran, 1958 (Orthoptera: Tettigoniidae) it was possible, through its diet, to determine its exact habitat requirements [315]. However, it is also important to consider the composition and activity of the gut microbiota. This can be achieved using high-throughput sequencing employing the 16S rRNA gene and shotgun metagenomic sequencing methods to provide insights into microbial community composition and function. Using this approach on three species of ensiferans with very different feeding preferences, Zheng et al. [316] showed that the gut microbial communities of these species were significantly different, with more diverse feeding preferences associated with gut microorganisms with less specific functions. But other microbial functional genes, related to energy and nutrition acquisition, are also involved and affect the host species survival.
Metabarcoding (the combination of DNA taxonomy and high-throughput sequencing) among orthopterans has also been used in other contexts, including diet shift in the conversion of tropical forest to oil palm production [120]. In warm Chinese forests where rubber and tea had replaced natural forest, results from metabarcoding showed that orthopteran diversity was high in tea and higher even than mixed plantation/natural forest [317].
A further technological advance is the practical implementation of environmental DNA (eDNA). The method can be applied to rare or elusive species as it is highly sensitive in detecting minute traces of the genome left behind by insects such as orthopterans on the surface of plant material [318] and in river systems [319]. Application of this highly sensitive method can be used for detecting nascent invasive populations, delimiting invasion fronts, as well as for detecting optimal habitats [320]. The method can be applied to various types of ecosystems and may uncover the presence of previously locally unknown species [321] and plant–animal interactions [322]. Further application is for monitoring over space and time [323], although it should use standardized eDNA workflows for effective conservation and management [324].
Orthopterans were a component of an arthropod study that archived leaf material to characterize plant-associated arthropod assemblages over several decades. The study used leaf time series from tree canopies in four land-use types across Germany. Results indicated that, rather than widespread declines in α-diversity or genetic variation within sites, there has been a gradual assemblage turnover, manifesting as temporal and spatial biotic homogenization across all land-use types and arthropod orders. These findings suggest that insect decline is more complex than only α-diversity loss and instead is driven by β-diversity decay across space and time [325].
4.8. Role of Ecoacoustics in Orthopteran Conservation
As sound production is so prevalent in orthopterans, use of their song for assessment and monitoring is a valuable tool. However, from a practical point of view, the human ear detects only those song frequencies in the lower, sonic range, which varies according to a person’s age. Nevertheless, unaided song detection can still be valuable in orthopteran assessment and conservation [326,327]. But for detection of higher frequency components under field conditions, it is necessary to use ultrasonic wavelength-detection apparatus [328,329,330].
The value of an acoustic approach is that the exact location of individual orthopterans can be determined, especially for those that sing at dusk or during the night when other methods may not be suitable [331]. It is an approach that can also determine assemblage rules [332]. Furthermore, otherwise morphologically indistinguishable species can be detected [333]. However, as not all orthopteran species sing, for a comprehensive assessment of all orthopteran species present it may be necessary to supplement sound assessment with visual surveys (or other methods), as in the Austrian Alps [334].
In the transition from conventional to sustainable agriculture, ecoacoustic assessment was used to determine the relative value of the zones of a biosphere reserve in South Africa and showed that agroecological approaches are supporting the indigenous tettigoniid fauna rather well [206]. Also, in South Africa, orthopteran acoustic assessment was used to determine the quality of large-scale conservation corridors across a plantation mosaic. Habitat heterogeneity in terms of varied vegetation types and structures emerged as the most important features for determining the effectiveness of these corridors as natural refuges for improved orthopteran conservation [335].
In the same area, the orthopteran soundscape illustrated that ecotones between wooded and grassland areas played an important role in supporting the sound-emitting orthopteran assemblages [336]. Furthermore, acoustic interference between tettigoniid species was low in the area as species co-existed by having distinct ecological resource requirements and inhabited different biotopes, preventing acoustic interference from other species. In sum, acoustic and environmental factors play interactive roles in enabling sympatric species to co-exist across these complex landscapes [337].
When these results are compared with earlier findings from southern France, the effect of agricultural practices becomes apparent. Platycleis affinis Fieber, 1853 and P. intermedia (Serville, 1838) (Orthoptera: Tettigoniidae) nymphs readily inhabited barley fields. However, harvesting of the crop led to the young adults moving to the surrounding meager natural patches, causing acoustic interference through insufficient natural habitat [338]. This interference on the landscape can be so severe that P. intermedia’s song is greatly distorted by P. affinis’ song and especially by that of another species in the area, P. falx (Fabricius, 1775), with its intense song [339].
5. Discussion
5.1. Some Fundamentals Associated with the Transition to Agroecology
The transition from conventional agriculture to an agroecological approach involves circular practices that focus on creating closed-loop systems where waste is minimized and resources are reused and recycled within the agricultural ecosystem. This way forward aims to reduce reliance on external inputs, improve resource efficiency, and promote more sustainable and resilient food systems. It also considers the wider domain of a farm, ecologically, socially, and economically, with attention given to ecological integrity and resilience in these times of rapid climate change. A protocol has been developed to achieve these goals [340].
In effect, the human-altered landscape is a huge filter that causes shifts in population levels, dispersion patterns, and assemblage structures. While some species may respond positively to these changes, others do not. One result is the homogenization of communities where there is loss of some specialist species in favor of ecologically tolerant and opportunistic species. The risk is that some species will be lost entirely, firstly locally and then more widely. However, tipping points remain difficult to predict (let alone manage) where or when they are most likely to occur in complex real-world settings [341].
As agriculture is globally dominant and essential, it is urgent that there is more action towards the transition away from conventional resource-hungry agriculture and a move towards a more environmentally sensitive way forward. Grasshoppers and crickets can help us do this in a practical way, as well as by us appreciating their intrinsic value.
However, there are three major issues facing this initiative. The first is the ‘shifting baseline syndrome’ [342]. This is about recognizing environmental deterioration, people’s valuing of nature, and even appreciation of its crucial role in our wellbeing and survival. With biodiversity continually being eroded, current environmental conditions are often accepted without being questioned. What was considered as a normal environment and levels of biodiversity by past human generations is continually giving way with each new generation to a new normal. That means we are living in a continually depleted biotic world while depending on a more precarious environmental state.
The second issue is the ‘pest-preservation paradox’ which is highly significant among orthopterans. As orthopteran species and assemblages vary enormously in biology, abundance, population variability, and geographic range, it means that some are major pests while others are threatened with extinction, with some species already extinct through human agency. While most agricultural pest species are acridids, many other species in all families face some level of threat, especially those confined to small geographical areas and where the human footprint coincides with their small ranges. As pestiferous species of grasshoppers and crickets are widespread and numerous, it means their economic control often leads to an umbrella of threat to the many non-target species [343].
The third issue, and related to the second, is that of human perception impediment. This is illustrated by a survey among 41 respondents in rural communities in Wyoming rangeland. Results found that most respondents (51%) chose ‘grasshoppers are not a problem unless in large numbers (outbreak)’, while 39% selected ‘all should be killed’ and only 10% chose ‘they are beneficial to grasslands and should only be controlled if in large numbers’, while none chose ‘that grasshoppers should not be controlled’. Overall, these responses are either neutral or negative, inferring that civil society, at least in this community, does not hold an overly positive perception of grasshoppers [344].
5.2. Natural Orthopteran Population and Assemblage Dynamics
Among orthopteran species there are great differences in dispersal ability, largely related to locating optimal resources, completing their life cycle, and escaping predators. The most obvious feature is either being able to fly or not. While flying enables more rapid location of optimal conditions than walking or hopping, flightless species may also readily move to avoid adversity and procure optimal conditions at the local scale [29].
Natural drivers can vary in extent. Grazing by mammalian herbivores is localized on a daily basis, with most orthopteran species well adapted to seek optimal conditions where such grazing occurs [85]. Fire on the other hand, can be rapid, destructive, and extensive. Good fliers may be able to readily flee a fire, but this is more difficult for less mobile species. These poor dispersers may either perish in a fire or seek refuges in unburned patches. In the highly fire-prone Cape Floristic Region of South Africa, many flightless species have not only survived numerous past fires but have also undergone significant diversification in some genera [300]. They appear able to readily seek refuges, possibly by first detecting smoke [345]. Some also display melanism, for better crypsis against a burned and blackened background, where they take advantage of rapid plant regrowth while remaining relatively inconspicuous to visually oriented predators [99].
For the excellent fliers such as locusts, they travel great distances to intergenerationally take advantage of optimal food resources and breeding sites, usually following high rainfall in dry areas. From an evolutionary perspective swarming and long-distance flights have been an effective strategy [346].
Grazing by indigenous mammalian herbivores important for creating gaps in a forested landscape. Orthopterans are well able to take advantage of not only the opportunistic herbaceous vegetation but also the ecotones between woody and low-canopy vegetation [333]. In response, many orthopteran species can maintain effective population sizes in relatively small patches of optimal habitat [23]. Nevertheless, when the opportunity arises from improved functional connectivity, they readily take advantage of it [213].
In short, landscape and habitat heterogeneity have been an important driver of orthopteran assemblages, with the extant species composed of individuals that can readily and continuously locate optimal conditions both for bodily maintenance and life cycle completion.
5.3. Orthopteran Survival in Transformed Landscapes
Agriculture has been gradually transforming landscapes over many centuries. The transformation has been both extensive and intensive, with the first adverse effects on insects noted already in the late 19th century [347]. However, it was the development of mechanization, synthetic products, and increased infrastructure that radically transformed vast areas of land with huge consequences for insects [348].
From the evidence available, it appears that with loss of indigenous grazing and browsing animals, and subsequent replacement by livestock, that the former natural grazing effects are being simulated to some extent by the presence of livestock. But there are optimal levels of herbivory by large animals, as shown by both abandonment of traditional livestock grazing lands and, at the other extreme, heavy grazing that reduces orthopteran abundance and diversity, especially in dry areas [75,109]. As a result, there have been, across continents, a return to moderate levels of grazing. This approach increases vegetation heterogeneity, both by feeding and trampling, which benefits the various life stages [130].
There are often other impacts, such as crop and timber production, that affect orthopterans. But the footprint of these novel landscape transformations varies according to the type of production. Intensively managed arable fields are particularly devastating for orthopterans [108,140]. The only valuable space left is set-aside land and refuges, which are more feasible and prevalent in mountainous areas than on fertile flatlands [349]. In turn, perennial production systems offer more opportunities for orthopteran preservation than do arable fields. Such long-lasting systems can be modified through establishment of vegetation-rich inter-rows or set-aside patches, especially in the case of crops like vines and fruit trees (deciduous fruit, citrus, and olives) [207]. Even terraced agriculture can provide refuges on the revetments when well managed [210].
In the case of timber plantations, the landscape can be managed through setting aside large areas of remnant habitat in the form of extensive networks of conservation corridors. When these are managed in an environmentally effective way, through careful prescribed burning for example, they can maintain the orthopteran fauna as well as protected areas do. These corridors have excellent functional connectivity, allowing the natural ebb and flow of the orthopteran inhabitants. When the corridors are adjacent to protected areas, they extend the reach of the protected areas and provide considerable ecological resilience against climate change and extreme weather events. This has proved to be an excellent approach for harmonizing orthopteran and other biodiversity conservation alongside timber production [350].
One of the greatest challenges in the move to agroecological management has been to move away from the use of chemical pesticides. Not only do they have a huge impact on insects in general within the area of application, but also beyond it, in the margins and refuges, mostly because of pesticide drift [168]. This is a challenge greater than just for orthopteran conservationists, but for all biodiversity practitioners. However, research on orthopterans can really help in transitioning away from conventional applications to a much more ecologically viable integrated pest programs, especially when applied area-wide [263]. Together with this progression, are other methodological improvements, which include, for example, improving the heterogeneity of indigenous vegetation, and use of improved crop varieties [65,111,167,186,211].
One of the great challenges for orthopteran conservation is the control of grasshopper outbreaks and locust swarms. Conventional pesticides in most of the main registered chemical groups have been used against grasshoppers and locusts for many years. But outbreaks and swarms still appear to cause alarm, triggering an insecticide blitz. However, the advent of specific biopesticides has proven to be a great advance in having a more environmentally safe means of outbreak and swarm control [246]. While this new generation of orthopteran control is mostly safe for most non-Orthoptera taxa tested [251,351], there is still concern over its impact on non-target orthopterans. This is especially the case as new biopesticide strains are being produced which can be environmentally highly persistent [263].
The current overarching driver on all biodiversity and farming methods is global change. Besides a warming world, there is the great effect on insects as ectotherms and as sensitive responders to even small differences in environmental conditions [352,353]. Coupled with the effects of increased global temperatures are increasingly intense and variable weather events, whether drought and fire, or precipitation and floods, all of which are currently affecting insects [354]. Furthermore, climate change is directly synergistic with other drivers and can have an adverse effect on, for example, small and isolated refuges to which a population may be confined. Another important development is that climate change has overtaken landscape transformation in central Europe as the main driver of orthopteran assemblage shifts [189].
6. The Way Forward for Integration of Orthopterans in the Shift to Agroecology
From the evidence to date on the transition from conventional agriculture to agroecology, it is possible to identify some distinct pointers on the value of orthopterans in agroecological advancement, as well as identifying some of the main drivers affecting changes in their populations and assemblages. This leads to suggestions relating to the value of orthopterans towards better design and management of the agroecological landscape. We now summarize the various approaches for improved sustainability.
6.1. Appreciating Orthopteran Value
- Orthopterans do not live in isolation, interacting with many other organisms, both positively and negatively.
- Orthopterans play a vital ecological role in cycling nutrients and as prey that supports many other organisms.
- Orthopterans ideally should be considered with other biodiversity to attain more holism in the transition to agroecology that is more acceptable to farmers, managers, and policy makers.
- All successful agroecological advances consider the whole life cycle of orthopterans and the special needs of each life stage.
- Changes in both abundance and relative species frequency (assemblage composition) are strong indicators of changes in landscape quality for better or worse.
- Orthopterans are largely conspicuous and easily sampled in low-canopy systems.
- In forests (as well as in low canopies) their acoustic emissions can be used to record species’ presence.
- In low canopies, both acoustic and other sampling methods can be used.
6.2. Recognizing the Adverse Drivers of Orthopteran Abundance and Diversity Levels
- Excessive use of chemical pesticides, herbicides, and fungicides is unacceptable. Their use must be carefully integrated into a sustainable farm management program, preferably area-wide.
- Excessive grazing or mowing is detrimental to overall orthopteran assemblages and reduces ecological resilience to climate change.
- Arable crop fields have an impact on most biodiversity and must be managed with this view in mind, and in the case of orthopterans by instigating natural or semi-natural set-aside land.
- Adverse drivers can be synergistic, and these synergisms must be understood for agroecological advancement.
- Ongoing climate change is an overarching challenge that interacts with other adverse drivers. However, some adaptation by orthopterans to climate change has already been recorded.
- Increased intensity and extent of extreme weather events must be recognized and measures put in place to mitigate them.
6.3. Better Design and Management of Agroecosystems
- All effective design and management activities that improve the transition to agroecology must be tailored to local geographic, climatic, topographic, and socio-ecological contexts.
- At the large spatial scale, protected areas and biosphere reserves play a major role in maintaining orthopteran and other biodiversity.
- At the landscape scale, presence of networks of well-connected, high-quality conservation corridors are of great value for orthopterans and other biodiversity, especially as they provide high levels of functional connectivity.
- In the case of intensive arable fields, which can greatly fragment orthopteran populations, use of set-aside refuges in the form of vegetation patches, field margins, or ditches promote orthopteran species presence and abundance.
- Refuges that support isolated populations, endemic species, and/or threatened species are of especially high value.
- In orchards and vineyards, vegetal improvement of inter-rows and set-aside areas can improve orthopteran diversity and functional connectivity. Additional refuges in the matrix are complementary to inter-row improvement.
- In the tropics, where forests have been replaced by farming and have led to concurrent loss of habitat for forest orthopterans, certain species benefit from mixed cropping. Sometimes to such an extent that insecticides are then used against certain species, to the detriment of other non-target orthopteran species.
- As much natural forest must be maintained as intact as possible, especially in warmer climates. However, when only patches are left, these should be as large as possible and functionally connected. Where natural megaherbivores have been lost, as in Europe for example, their re-introduction can help create forest mosaics and provide landscape heterogeneity, beneficial to orthopteran assemblages.
- Maintaining or rewilding with indigenous megaherbivores greatly benefits orthopterans and other biodiversity through moderate grazing, browsing, and trampling of vegetation. Protection of these large animals can be an important conservation umbrella for orthopterans.
- Where wild herbivores have been lost, domestic livestock can also create a heterogenous landscape, greatly beneficial to a wide range of orthopterans. This heterogeneity includes some bare ground as oviposition sites for many species, especially caeliferans. A healthy sward through appropriate levels of grazing also provides some resilience against the effect of climate change.
- Avoid overgrazing by livestock as this reduces ecological integrity and resilience, especially with the ongoing pressure from climate change and extreme weather events, especially drought.
- Excessive grazing and mowing are mostly contrary to maintenance of orthopteran diversity, meaning that sustainable traditional methods of grazing should be considered. This must be carried out according to local context, and employ, for example, rotational approaches that may have to be executed according to the right season of the year. Wet environments in generally dry areas may allow for slightly more intense grazing.
- Prescribed burning of the landscape can also promote orthopteran abundance and diversity but must be implemented at optimal intervals and extent by skilled practitioners.
- Restoration activities should consider maintaining intermediate seral stages in certain areas, as this can often provide much landscape heterogeneity.
- Where feasible, control or removal of invasive alien plants is important. This is more easily achieved in the case of alien trees, which can shade out orthopteran habitat.
- Orthopterans benefit from environmentally sensitive integrated pest management programs on any type of crop, especially in terms of reducing insecticide input and drift. Fertilizer input should also be moderated to maintain vegetation and orthopteran diversity.
- Use of chemical insecticides for control of pestiferous orthopterans is environmentally hazardous and must be replaced by use of much more environmentally safe biopesticides. These products are largely safe for non-orthopteran, non-target organisms in many taxa. However, there is concern that biopesticides can be harmful to some non-target orthopterans.
- Monitoring populations of pest orthopterans is essential to better predict and then manage outbreaks and swarms before they become too large.
7. Conclusions
Orthopterans are virtually ubiquitous across all but permanently frozen areas, have diverse lifestyles, and overall have a wide dietary base. In total, their high abundance also means that they are of great functional significance. Furthermore, their densities change over time, sometimes greatly in certain species. Overall, orthopterans are also relatively mobile and respond quickly to deteriorating local environmental conditions, moving to optimal habitat. The outcome is not only dynamic populations but also constantly changing assemblage compositions over space and time.
Owing to their investigative tractability, orthopterans are ideal for tracking landscape change while also making an important contribution to assessing the state of biodiversity in various contexts. This is why they should be included in assessments and in monitoring the transition to agroecology. This review suggests that we have enough evidence from orthopteran studies to date to estimate how well we are performing in this respect. But inevitably there are some knowledge gaps, especially in warmer regions.
While much progress has been made in temperate North America, Europe, and Asia, there is still relatively little information available from the tropics where orthopteran species richness is very high. As with other insect taxa, tropical forest deforestation is having a major impact on the orthopteran inhabitants, but this is very poorly investigated.
The temperate northern hemisphere biomes have been transformed by humans over many centuries meaning that we must assume that a shifting baseline has already taken place. But there are glimpses of this from studies in central Europe where formerly landscape transformation was the major driver but today it is climate change, notwithstanding that there has been landscape improvement in the region.
Assessment and monitoring are critical for measuring how well or not we are performing in the transition to agroecology. One problem is that it is expensive to undertake, especially on a long-term basis. However, it is being advanced by improved technology from satellite imagery and machine learning, used in some of the studies reviewed here. There are other areas where new technology may help. While environmental DNA studies have moved forward in watery environments, there is potential for using it for orthopteran assessment and monitoring.
Lastly, there are some basic issues that need to be further resolved, especially in warmer areas of the world. Better identification guides and applications are needed in many geographic areas. There is also a communication issue, not least because while civil society is largely familiar with ‘a grasshopper’, there is very poor appreciation of the value of orthopterans as a group. This is partly because outbreak grasshoppers and locust swarms dominate the messaging in the media as well as among farmers, managers, and policy makers. Clearly, there is still much more to be achieved with better communication in schools, colleges, farmers’ groups, and particularly civil society.
These conclusions are summarized in Figure 7.
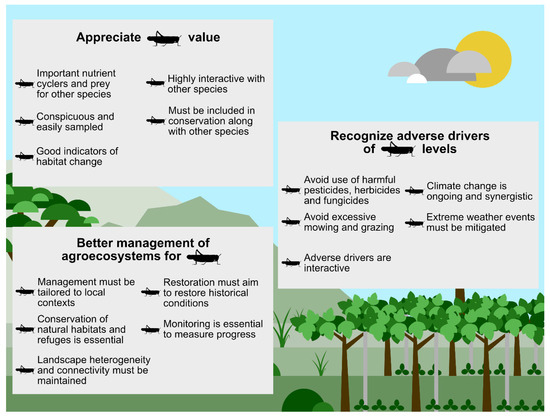
Figure 7.
A summary of the conclusions which relate to three main themes: appreciating the value of orthopterans, recognizing the significant drivers causing their decline, and aiming to instigate better agroecological methods.
Author Contributions
Conceptualization, M.J.S.; investigation, M.J.S. and M.L.; writing—original draft preparation, M.J.S.; writing—review and editing, M.L. and C.D.; graphics, C.D. All authors have read and agreed to the published version of the manuscript.
Funding
M.J.S. and C.D. acknowledge funding from Stellenbosch University, National Research Foundation, and Mondi Group over many years.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Gaigher, R.; van den Berg, J.; Batáry, P.; Grass, I. Agroecological Farming for Insect Conservation. In Routledge Handbook of Insect Conservation; Pryke, J.S., Samways, M.J., New, T.R., Cardoso, P., Gaigher, R., Eds.; Routledge: Abingdon, UK; New York, NY, USA, 2024; pp. 132–145. [Google Scholar] [CrossRef]
- Samways, M.J. Insect Conservation: A Global Synthesis; CABI: Wallingford, UK, 2020; ISBN 9781789241686. [Google Scholar]
- Gerlach, J.; Samways, M.; Pryke, J.S. Terrestrial Invertebrates as Bioindicators: An Overview of Available Taxonomic Groups. J. Insect Conserv. 2013, 17, 831–850. [Google Scholar] [CrossRef]
- Bharamal, D.L.; Koli, Y.J. Grasshoppers as Multidisciplinary Model Organisms in Changing Environments. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 372–380. [Google Scholar]
- Song, H.; Amédégnato, C.; Cigliano, M.M.; Desutter-Grandcolas, L.; Heads, S.W.; Huang, Y.; Otte, D.; Whiting, M.F. 300 Million Years of Diversification: Elucidating the Patterns of Orthopteran Evolution Based on Comprehensive Taxon and Gene Sampling. Cladistics 2015, 31, 621–651. [Google Scholar] [CrossRef]
- Song, H.; Mariño-Pérez, R.; Woller, D.A.; Cigliano, M.M. Evolution, Diversification, and Biogeography of Grasshoppers (Orthoptera: Acrididae). Insect Syst. Divers. 2018, 2, 3. [Google Scholar] [CrossRef]
- Cigliano, M.M.; Braun, H.; Eades, D.C.; Otte, D. Orthoptera Species File. Available online: https://orthoptera.speciesfile.org (accessed on 21 May 2025).
- Song, H. Biodiversity of Orthoptera. In Insect Biodiversity: Science and Society, 2nd ed.; Foottit, R.G., Adler, P.H., Eds.; Wiley-Blackwell: Oxford, UK, 2018; pp. 245–279. [Google Scholar]
- Rosenberg, Y.; Bar-On, Y.; Fromm, Y.M.; Ostikar, M.; Shoshany, A.; Milo, R. The Global Biomass and Number of Terrestrial Arthropods. Sci. Adv. 2023, 9, eabq4049. [Google Scholar] [CrossRef]
- Kietzka, G.J.; Lecoq, M.; Samways, M.J. Ecological and Human Diet Value of Locusts in a Changing World. Agronomy 2021, 11, 1856. [Google Scholar] [CrossRef]
- Porter, E.E.; Redak, R.A.; Braker, H.E. Density, Biomass, and Diversity of Grasshoppers (Orthoptera: Acrididae) in a California Native Grassland. Great Basin Nat. 1996, 56, 172–176. [Google Scholar]
- Gardiner, T.; Pye, M.; Field, R.; Hill, J. The Influence of Sward Height and Vegetation Composition in Determining the Habitat Preferences of Three Chorthippus Species (Orthoptera: Acrididae) in Chelmsford, Essex, UK. J. Orthoptera Res. 2002, 11, 207–213. [Google Scholar] [CrossRef]
- Appelt, M. Elements of Population Vulnerability of the Blue-Winged Grasshopper, Oedipoda caerulescens (Linnaeus, 1758) (Caelifera, Acrididae). In Species Survival in Fragmented Landscapes; Settle, J., Margules, C., Poschlod, P., Henle, K., Eds.; Kluwer: Dordrecht, The Netherlands, 1996; pp. 320–323. [Google Scholar]
- Köhler, G. The Ecological Background of Population Vulnerability in Central European Grasshoppers and Bush Crickets: A Brief Review. In Species Survival in Fragmented Landscapes; Settle, J., Margules, C., Poschlod, P., Henle, K., Eds.; Kluwer: Dordrecht, The Netherlands, 1996; pp. 290–298. [Google Scholar]
- Wagner, G.; Berger, U. A Population Vulnerability Analysis of the Red-Winged Grasshoppers, Oedipoda germanica (Caelifera: Acrididae). In Species Survival in Fragmented Landscapes; Settle, J., Margules, C., Poschlod, P., Henle, K., Eds.; Kluwer: Dordrecht, The Netherlands, 1996; pp. 312–319. [Google Scholar]
- Himes Boor, G.K.; Schultz, C.B.; Crone, E.E.; Morris, W.F. Mechanism Matters: The Cause of Fluctuations in Boom–Bust Populations Governs Optimal Habitat Restoration Strategy. Ecol. Appl. 2018, 28, 356–372. [Google Scholar] [CrossRef]
- Zhang, L.; Lecoq, M.; Latchininsky, A.; Hunter, D. Locust and Grasshopper Management. Annu. Rev. Entomol. 2019, 64, 15–34. [Google Scholar] [CrossRef]
- Antonatos, S.; Emmanouel, N.; Fantinou, A.; Tsagkarakis, A.; Anagnostopoulos, A.; Ntampakis, D. Seasonal Population Fluctuation and Spatial Distribution of Orthoptera in Two Grassland Areas of Attica, Greece. J. Nat. Hist. 2014, 48, 661–674. [Google Scholar] [CrossRef]
- Badenhausser, I.; Amouroux, P.; Lerin, J.; Bretagnolle, V. Acridid (Orthoptera: Acrididae) Abundance in Western European Grasslands: Sampling Methodology and Temporal Fluctuations. J. Appl. Entomol. 2009, 133, 720–732. [Google Scholar] [CrossRef]
- Illich, I.; Zuna-Kratky, T. Population Dynamics of an Alpine Grasshopper (Orthoptera) Community over 30 Years and the Effects of Climate Warming and Grazing. J. Insect Conserv. 2022, 26, 435–451. [Google Scholar] [CrossRef]
- Magoba, R.N.; Samways, M.J. Comparative Footprint of Alien, Agricultural and Restored Vegetation on Surface-Active Arthropods. Biol. Invasions 2012, 14, 165–177. [Google Scholar] [CrossRef]
- Kulessa, A.K.; Cuthbert, R.N.; Soto, I.; Balzani, P.; Toutain, M.; Kouba, A.; Haubrock, P.J. Assessing the Phytosanitary Threats of Two Non-Native Crickets under Temperature Change. J. Pest Sci. 2025, 98, 1629–1643. [Google Scholar] [CrossRef]
- Bönsel, A.B.; Sonneck, A.-G. Habitat Use and Dispersal Characteristics of Stethophyma grossum: The Role of Habitat Isolation and Stable Habitat Conditions towards Low Dispersal. J. Insect Conserv. 2011, 15, 455–463. [Google Scholar] [CrossRef]
- Ancillotto, L.; Labadessa, R. Functional Traits Drive the Fate of Orthoptera in Urban Areas. Insect Conserv. Divers. 2024, 17, 304–311. [Google Scholar] [CrossRef]
- Lagos, P.A. A Review of Escape Behaviour in Orthopterans. J. Zool. 2017, 303, 165–177. [Google Scholar] [CrossRef]
- Leubner, F.; Wipfler, B.; Bradler, S. Comparative Morphology of the Orthoptera Thorax with a Discussion of Phylogenetically Relevant Characters. Insect Syst. Divers. 2017, 1, 29–47. [Google Scholar] [CrossRef]
- Whitman, D.W. The Significance of Body Size in the Orthoptera: A Review. J. Orthoptera Res. 2008, 17, 117–134. [Google Scholar] [CrossRef]
- García-Navas, V.; Noguerales, V.; Cordero, P.J.; Ortego, J. Ecological Drivers of Body Size Evolution and Sexual Size Dimorphism in Short-Horned Grasshoppers (Orthoptera: Acrididae). J. Evol. Biol. 2017, 30, 1592–1608. [Google Scholar] [CrossRef]
- Adu-Acheampong, S.; Samways, M.J. Feeding and Mobility Traits Influence Grasshopper Vulnerability to Agricultural Production in the Cape Floristic Region Biodiversity Hotspot. Neotrop. Entomol. 2019, 48, 992–1000. [Google Scholar] [CrossRef]
- Ren, Y.-S.; Zhang, B.; Zeng, Y.; Zhu, D.-H. Effects of Flight on Reproductive Development in Long-Winged Female Crickets (Velarifictorus aspersus Walker; Orthoptera: Gryllidae) with Differences in Flight Behavior. Insects 2023, 14, 79. [Google Scholar] [CrossRef] [PubMed]
- Chapuis, M.-P.; Bazelet, C.S.; Blondin, L.; Foucart, A.; Vitalis, R.; Samways, M.J. Subspecific Taxonomy of the Desert Locust, Schistocerca gregaria (Orthoptera: Acrididae), Based on Molecular and Morphological Characters. Syst. Entomol. 2016, 41, 516–530. [Google Scholar] [CrossRef]
- Marini, L.; Öckinger, E.; Battisti, A.; Bommarco, R. High Mobility Reduces Beta-Diversity among Orthopteran Communities—Implications for Conservation. Insect Conserv. Divers. 2012, 5, 37–45. [Google Scholar] [CrossRef]
- Dziock, F.; Gerisch, M.; Siegert, M.; Hering, I.; Scholz, M.; Ernst, R. Reproducing or Dispersing? Using Trait-Based Habitat Templet Models to Analyse Orthoptera Response to Flooding and Land Use. Agric. Ecosyst. Environ. 2011, 145, 85–94. [Google Scholar] [CrossRef]
- Palacios-Gimenez, O.M.; Casillo, E.R.D.; Schielzeth, H. Karyotype Evolution and Speciation in Orthoptera. J. Evol. Biol. 2025, 38, 516–529. [Google Scholar] [CrossRef]
- Krogh, A.; Weis-Fogh, T.A. Roundabout for Studying Sustained Flight of Locusts. J. Exp. Biol. 1952, 29, 211–219. [Google Scholar] [CrossRef]
- Nickle, D.A.; Castner, J.L. Strategies Utilized by Katydids (Orthoptera: Tettigoniidae) against Diurnal Predators in Rainforests of Northeastern Peru. J. Orthoptera Res. 1995, 4, 75–88. [Google Scholar] [CrossRef]
- Ariel, G.; Ayali, A. Locust Collective Motion and Its Modeling. PLoS Comput. Biol. 2015, 11, e1004522. [Google Scholar] [CrossRef]
- Cullen, D.A.; Cease, A.J.; Latchininsky, A.V.; Ayali, A.; Berry, K.; Buhl, J.; De Keyser, R.; Foquet, B.; Hadrich, J.C.; Matheson, T.; et al. From Molecules to Management: Mechanisms and Consequences of Locust Phase Polyphenism. Adv. Insect Physiol. 2017, 53, 167–285. [Google Scholar] [CrossRef]
- van der Werf, W.; Woldewahid, G.; van Huis, A.; Butrous, M.; Sykora, K. Plant Communities Can Predict the Distribution of Solitarious Desert Locust Schistocerca gregaria. J. Appl. Ecol. 2005, 42, 989–997. [Google Scholar] [CrossRef]
- Skaf, R.; Popov, G.; Roffey, J.; Scorer, R.; Hewitt, J. The Desert Locust: An International Challenge. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1990, 328, 525–538. [Google Scholar] [CrossRef]
- Le Gall, M.; Touré, M.; Lecoq, M.; Marescot, L.; Cease, A.; Maiga, I. Chapter 4—Senegalese Grasshopper: A Major Pest of the Sahel. In Biological and Environmental Hazards, Risks, and Disasters, 2nd ed.; Sivanpillai, R., Shroder, F.J., Eds.; Hazards and Disasters Series; Elsevier: Boston, MA, USA, 2023; pp. 77–96. [Google Scholar] [CrossRef]
- Lecoq, M. Schistocerca gregaria (Desert Locust). In Crop Protection Compendium; CAB International: Wallingford, UK, 2022. [Google Scholar] [CrossRef]
- Lecoq, M. Locusta migratoria (Migratory Locust). In Crop Protection Compendium; CAB International: Wallingford, UK, 2023. [Google Scholar] [CrossRef]
- Cease, A.; Lecoq, M.; Word Ries, M.; Therville, C. Locusts as pests: Past, present, and future. In Grasshoppers, Locusts, and Crickets of the World; Husemann, M., Hawlitschek, O., Eds.; Princeton University Press: Princeton, NJ, USA; Oxford, UK, 2025. [Google Scholar]
- Gangwere, S.K. A Monograph on Food Selection in Orthoptera. Trans. Am. Entomol. Soc. 1961, 87, 67–230. [Google Scholar]
- Lockwood, J.A. Cannibalism in Rangeland Grasshoppers (Orthoptera: Acrididae): Attraction to Cadavers. J. Kans. Entomol. Soc. 1988, 61, 379–387. [Google Scholar]
- Joern, A. Feeding Patterns in Grasshoppers (Orthoptera: Acrididae): Factors Influencing Diet Specialization. Oecologia 1979, 38, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Floren, A.; Riede, K.; Ingrisch, S. Diversity of Orthoptera from Bornean Lowland Rainforest Trees. Ecotropica 2001, 7, 33–42. [Google Scholar]
- Weidemann, S.; Steidl, O.; Kalmring, K. Distribution and Population Density of the Bushcricket Decticus verrucivorus in a Damp-Meadow Biotope. Oecologia 1990, 82, 369–374. [Google Scholar] [CrossRef]
- Cherrill, A.J.; Brown, V.K. The habitat requirements of adults of the wart-biter Decticus verrucivorus (L.) (Orthoptera: Tettigoniidae) in southern England. Biol. Conserv. 1990, 53, 145–157. [Google Scholar] [CrossRef]
- Samways, M.J.; Harz, K. Biogeography of Intraspecific Morphological Variation in the Bush Crickets Decticus verrucivorus (L.) and D. albifrons (F.) (Orthoptera: Tettigoniidae). J. Biogeogr. 1982, 9, 243–254. [Google Scholar] [CrossRef]
- Chopard, L. Orthoptères, Faune de France; Lechevalier: Paris, France, 1951; Volume 56. [Google Scholar]
- Ton, L.; Nabholz, B. Le Dectique Verrucivore, Decticus verrucivorus (Linnaeus, 1758), en France Continentale: Étude Morphologique et Implications Taxonomiques (Orthoptera: Tettigoniidae). Matériaux Orthoptériques Entomocénotiques 2023, 28, 45–68. [Google Scholar]
- Song, H.; Béthoux, O.; Shin, S.; Donath, A.; Letsch, H.; Liu, S.; McKenna, D.D.; Meng, G.; Misof, B.; Podsiadlowski, L.; et al. Phylogenomic Analysis Sheds Light on the Evolutionary Pathways towards Acoustic Communication in Orthoptera. Nat. Commun. 2020, 11, 4939. [Google Scholar] [CrossRef]
- Römer, H. Environmental and Biological Constraints for the Evolution of Long-Range Signalling and Hearing in Acoustic Insects. Philos. Trans. R. Soc. Lond. B 1993, 340, 179–185. [Google Scholar] [CrossRef]
- Robinson, D.J.; Hall, M.J. Sound Signalling in Orthoptera. Adv. Insect Physiol. 2002, 29, 151–278. [Google Scholar] [CrossRef]
- Uvarov, B.P. Dynamics of Populations: Outbreaks and Plagues. In Grasshoppers and Locusts: A Handbook of General Acridology; The Centre for Overseas Pest Research: London, UK, 1977; Volume 2, pp. 445–523. [Google Scholar]
- Joern, A.; Gaines, S.B. Population Dynamics and Regulation in Grasshoppers. In Biology of Grasshoppers; Chapman, R.F., Joern, A., Eds.; Wiley: Hoboken, NJ, USA, 1990; pp. 415–482. [Google Scholar]
- Joern, A.; Laws, A.N. Ecological Mechanisms Underlying Arthropod Species Diversity in Grasslands. Annu. Rev. Entomol. 2013, 58, 19–36. [Google Scholar] [CrossRef]
- Cease, A.J.; Elser, J.J.; Ford, C.F.; Hao, S.; Kang, L.; Harrison, J.F. Heavy Livestock Grazing Promotes Locust Outbreaks by Lowering Plant Nitrogen Content. Science 2012, 335, 467–469. [Google Scholar] [CrossRef]
- Pati, A.; Paul, I.; Dutta, S. Drylands under Pressure: Responses of Insect Density to Land-Use Change in a Tropical Desert. Insects 2025, 16, 1043. [Google Scholar] [CrossRef]
- Branson, D.H. Influence of Individual Body Size on Reproductive Traits in Melanopline Grasshoppers (Orthoptera: Acrididae). J. Orthoptera Res. 2008, 17, 259–263. [Google Scholar] [CrossRef]
- Joern, A. Variation in Grasshopper (Acrididae) Densities in Response to Fire Frequency and Bison Grazing in Tallgrass Prairie. Environ. Entomol. 2004, 33, 1617–1625. [Google Scholar] [CrossRef]
- Hochkirch, A.; Damerau, M. Rapid Range Expansion of a Wing-Dimorphic Bush-Cricket after the 2003 Climatic Anomaly. Biol. J. Linn. Soc. 2009, 97, 118–127. [Google Scholar] [CrossRef]
- Fumy, F.; Löffler, F.; Samways, M.J.; Fartmann, T. Response of Orthoptera Assemblages to Environmental Change in a Low-Mountain Range Differs among Grassland Types. J. Environ. Manag. 2020, 256, 109919. [Google Scholar] [CrossRef]
- Bourhis, Y.; Milne, A.E.; Shortall, C.R.; Beckman, B.; Blumgart, D.; Edwards, R.; Evans, L.C.; Foster, C.W.; Fox, R.; Botham, M.S.; et al. Trait Mediation Explains Decadal Distributional Shifts for a Wide Range of Insect Taxa. Nat. Commun. 2025, 30, e8131. [Google Scholar] [CrossRef] [PubMed]
- Fartmann, T.; Poniatowski, D. (Eds.) Distribution Atlas of Grasshoppers and Praying Mantises in Germany; Ulmer: Stuttgart, Germany, 2025. [Google Scholar]
- Squires, V.R.; Dengler, J.; Feng, H.; Hua, L. (Eds.) Grasslands of the World: Diversity, Management and Conservation; CRC Press: Boca Raton, FL, USA, 2018; ISBN 978-1-4987-9626-2. [Google Scholar] [CrossRef]
- Belovsky, G.E. Do Grasshoppers Diminish Productivity? A New Perspective for Control Based on Conservation. In Grasshoppers and Grassland Health: Managing Grasshopper Outbreaks Without Risking Environmental Disaster; Lockwood, J.A., Latchininsky, A.V., Sergeev, M.G., Eds.; Kluwer Academic: Boston, MA, USA, 2000; pp. 7–29. [Google Scholar]
- McEwen, L.C.; DeWeese, L.R. Wildlife and Pest Control in the Sagebrush Ecosystem: Ecology and Management Considerations. In Integrated Pest Management on Rangeland: State of the Art in the Sagebrush Ecosystem; Onsager, J.A., Ed.; Bulletin ARS-50; U.S. Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 1987; pp. 76–85. [Google Scholar]
- Lockwood, J.A.; Li, H.-C.; Dodd, J.L.; Williams, S.E. Comparison of Grasshopper (Orthoptera: Acrididae) Ecology on the Grasslands of the Asian Steppe in Inner Mongolia and the Great Plains of North America. J. Orthoptera Res. 1994, 4, 4–14. [Google Scholar] [CrossRef]
- Lockwood, J.A.; Sergeev, M.G. Comparative Biogeography of Grasshoppers (Orthoptera: Acrididae) in North America and Siberia: Applications to the Conservation of Biodiversity. J. Insect Conserv. 2000, 4, 161–172. [Google Scholar] [CrossRef]
- Guo, Z.-W.; Li, H.-C.; Gan, Y.-L. Grasshopper (Orthoptera: Acrididae) Biodiversity and Grassland Ecosystems. Insect Sci. 2006, 13, 221–227. [Google Scholar] [CrossRef]
- Lu, L.; Kong, W.; Eerdengqimuge; Ye, H.; Sun, Z.; Wang, N.; Du, B.; Zhou, Y.; Weijun; Huang, W. Detecting Key Factors of Grasshopper Occurrence in Typical Steppe and Meadow Steppe by Integrating Machine Learning Model and Remote Sensing Data. Insects 2022, 13, 894. [Google Scholar] [CrossRef]
- Guo, J.; Lu, L.; Dong, Y.; Huang, W.; Zhang, B.; Du, B.; Ding, C.; Ye, H.; Wang, K.; Huang, Y.; et al. Spatiotemporal Distribution and Main Influencing Factors of Grasshopper Potential Habitats in Two Steppe Types of Inner Mongolia, China. Remote Sens. 2023, 5, 866. [Google Scholar] [CrossRef]
- Wang, M.; Song, X.; Li, C.; Jashenko, R.; Cao, Z.; Liu, H.; Lin, J.; Ji, R. Diversity of Grasshoppers in Different Grassland Types in the Western Tianshan Mountains of China and Influencing Factors. Environ. Entomol. 2025, 54, nvaf065. [Google Scholar] [CrossRef] [PubMed]
- Shen, T.; Wu, Q.; Jin, Y.; Wang, Y.; Zhu, A.; Ju, X.; Tan, Y.; Han, G. Grazing-induced Changes in the Abundance and Diversity of Grasshoppers is Mediated through Changes in Plant Functional Groups. Insect Conserv. Divers. 2025. [Google Scholar] [CrossRef]
- Dvořák, T.; Hadrava, J.; Knapp, M. The Ecological Niche and Conservation Value of Central European Grassland Orthopterans: A Quantitative Approach. Biol. Conserv. 2022, 265, 109406. [Google Scholar] [CrossRef]
- Thomas, J.; Segar, S.T.; Cherrill, A.J. Species richness of Orthoptera declines with elevation while elevation range of individual species peaks at mid elevation. Ecol. Evol. 2024, 14, e10985. [Google Scholar] [CrossRef]
- Kisbenedek, T.; Baldí, A. What Factors Govern Orthopteran Community Structure and Species Prevalence? In Grasslands and Grassland Health; Lockwood, J.A., Latchininsky, A.V., Sergeev, M.G., Eds.; Kluwer: Boston, MA, USA, 2000; pp. 97–107. [Google Scholar]
- Sutcliffe, L.M.E.; Batáry, P.; Becker, T.; Orci, K.M.; Leuschner, C. Both Local and Landscape Factors Determine Plant and Orthoptera Diversity in the Semi-Natural Grasslands of Transylvania, Romania. Biodivers. Conserv. 2015, 24, 229–245. [Google Scholar] [CrossRef]
- van Wingerden, W.K.R.E.; Musters, J.C.M.; Maaskamp, F.I.M. The influence of temperature on the duration of egg development in West European grasshoppers (Orthoptera: Tettigoniidae). Oecologia 1991, 87, 417–423. [Google Scholar] [CrossRef]
- Swengel, A.B. A Literature Review of Insect Responses to Fire, Compared to Other Conservation Managements of Open Habitat. Biodivers. Conserv. 2001, 10, 1141–1169. [Google Scholar] [CrossRef]
- van Klink, R.; van der Plas, F.; van Noordwijk, G.C.E.; WallisDeVries, M.F.; Olff, H. Effects of Large Herbivores on Grassland Arthropod Diversity. Biol. Rev. 2015, 90, 347–366. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, T. Orthoptera Response to Grazing. J. Orthoptera Res. 2018, 27, 3–11. [Google Scholar] [CrossRef]
- Joern, A. Disturbance by Fire Frequency and Bison Modulate Grasshopper Assemblages in Tallgrass Prairie. Ecology 2005, 86, 861–873. [Google Scholar] [CrossRef]
- Jonas, J.L.; Joern, A. Grasshopper (Orthoptera: Acrididae) Communities Respond to Fire, Bison Grazing and Weather in North American Tallgrass Prairie: A Long-Term Study. Oecologia 2007, 153, 699–711. [Google Scholar] [CrossRef]
- Ozment, K.A.; Welti, E.A.R.; Shaffer, M.; Kaspari, M. Tracking Nutrients in Space and Time: Interactions between Grazing Lawns and Drought Drive Abundances of Tallgrass Prairie Grasshoppers. Ecol. Evol. 2021, 11, 5413–5423. [Google Scholar] [CrossRef]
- Huntzinger, M.; Karban, R.; Hall Cushman, J. Negative Effects of Vertebrate Herbivores on Invertebrates in a Coastal Dune Community. Ecology 2008, 89, 1972–1980. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, T. Altitudinal Limits of Grasshoppers in the Cotswolds and Malvern Hills in Relation to Livestock Grazing of Hilltops. Bull. Amat. Entomol. Soc. 2011, 70, 77–81. [Google Scholar]
- Tuinder, Q.A.; van Leeuwen, B.O.; Vergeer, P.; Scherer, G.; Fartmann, T.; Jansen, P.A. Impact of Wild Boar (Sus scrofa) Rooting Succession on Grasshoppers (Orthoptera) in Abandoned Calcareous Grasslands. J. Insect Conserv. 2025, 29, 78. [Google Scholar] [CrossRef]
- Spalinger, L.C.; Haynes, A.G.; Schütz, M.; Risch, A.C. Impact of Wild Ungulate Grazing on Orthoptera Abundance and Diversity in Subalpine Grasslands. Insect Conserv. Divers. 2012, 5, 444–452. [Google Scholar] [CrossRef]
- Rivers-Moore, N.A.; Samways, M.J. Game and Cattle Trampling, and Impacts of Human Dwellings on Arthropods at a Game Park Boundary. Biodivers. Conserv. 1996, 5, 1545–1556. [Google Scholar] [CrossRef]
- Samways, M.J.; Kreuzinger, K. Vegetation, Ungulate and Grasshopper Interactions inside vs. outside an African Savanna Game Park. Biodivers. Conserv. 2001, 10, 1963–1981. [Google Scholar] [CrossRef]
- Gandar, M.V. Trophic Ecology of Grasshoppers (Acridoidea) in a South African Savanna. Oecologia 1982, 54, 370–378. [Google Scholar] [CrossRef]
- Schlettwein, C.H.G.; Giliomee, J.H. Comparison of Insect Biomass and Community Structure between Fynbos Sites of Different Ages after Fire with Particular Reference to Ants, Leafhoppers and Grasshoppers. Ann. Univ. Stellenbosch Ser. A3 Landbouwetenskappe 1987, 2, 1–76. [Google Scholar]
- Chapman, K. Ecological Studies on Solitary Acrididae in England and South Africa. J. Entomol. Soc. S. Afr. 1952, 25, 165–203. [Google Scholar]
- Hocking, B. Fire Melanism in Some African Grasshoppers. Evolution 1964, 18, 332–335. [Google Scholar] [CrossRef]
- Karpestam, E.; Merilaita, S.; Forsman, A. Reduced Predation Risk for Melanistic Pygmy Grasshoppers in Post-Fire Environments. Ecol. Evol. 2012, 2, 2204–2212. [Google Scholar] [CrossRef]
- Samways, M.J. Land Forms and Winter Habitat Refugia in the Conservation of Montane Grasshoppers in Southern Africa. Conserv. Biol. 1990, 4, 375–382. [Google Scholar] [CrossRef]
- Stein, S.J.; Price, P.W.; Abrahamson, W.G.; Sacchi, C.F. The Effect of Fire on Stimulating Willow Regrowth and Subsequent Attack by Grasshoppers and Elk. Oikos 1992, 65, 190–196. [Google Scholar] [CrossRef]
- Bock, C.E.; Bock, J.H. Response of Grasshoppers (Orthoptera: Acrididae) to Wildfire in a Southeastern Arizona Grassland. Am. Midl. Nat. 1991, 125, 162–167. [Google Scholar] [CrossRef]
- Branson, D.H. Effects of Fire on Grasshopper Assemblages in a Northern Mixed-Grass Prairie. Environ. Entomol. 2005, 34, 1109–1113. [Google Scholar] [CrossRef]
- Evans, E.W. Fire as a Natural Disturbance to Grasshopper Assemblages of Tallgrass Prairie. Oikos 1984, 43, 9–16. [Google Scholar] [CrossRef]
- Nadeau, L.; Cushing, P.E.; Kondratieff, B.C. Effects of Fire Disturbance on Grasshopper (Orthoptera: Acrididae) Assemblages of the Comanche National Grasslands, Colorado. J. Kans. Entomol. Soc. 2006, 79, 2–12. [Google Scholar] [CrossRef]
- Cardoso, P.; Barton, P.S.; Birkhofer, K.; Chichorro, F.; Deacon, C.; Fartmann, T.; Fukushima, C.S.; Gaigher, R.; Habel, J.C.; Hallmann, C.A.; et al. Scientists’ Warning to Humanity on Insect Extinctions. Biol. Conserv. 2020, 242, e108426. [Google Scholar] [CrossRef]
- Nufio, C.R.; McClenahan, J.L.; Bowers, M.D. Grasshopper Response to Reductions in Habitat Area as Mediated by Subfamily Classification and Life History Traits. J. Insect Conserv. 2011, 15, 409–419. [Google Scholar] [CrossRef]
- Cherrill, A. Species Richness of Orthoptera along Gradients of Agricultural Intensification and Urbanisation. J. Orthoptera Res. 2010, 19, 293–301. [Google Scholar] [CrossRef]
- Chisté, M.N.; Mody, K.; Gossner, M.M.; Simons, N.K.; Köhler, G.; Weisser, W.W.; Blüthgen, N. Losers, Winners, and Opportunists: How Grassland Land-Use Intensity Affects Orthopteran Communities. Ecosphere 2016, 7, e01545. [Google Scholar] [CrossRef]
- Fartmann, T.; Krämer, B.; Stelzner, F.; Poniatowski, D. Orthoptera as Ecological Indicators for Succession in Steppe Grassland. Ecol. Indic. 2012, 20, 337–344. [Google Scholar] [CrossRef]
- Fumy, F.; Kaempfer, S.; Fartmann, T. Land-use Intensity Determines Grassland Orthoptera Assemblage Composition across a Moisture Gradient. Agric. Ecosyst. Environ. 2021, 315, e107424. [Google Scholar] [CrossRef]
- Fournier, B.; Mouly, A.; Moretti, M.; Gillet, F. Contrasting Processes Drive Alpha and Beta Taxonomic, Functional and Phylogenetic Diversity of Orthopteran Communities in Grasslands. Agric. Ecosyst. Environ. 2017, 242, 43–52. [Google Scholar] [CrossRef]
- Guo, K.; Hao, S.-G.; Sun, O.J.; Kang, L. Differential Responses to Warming and Increased Precipitation among Three Contrasting Grasshopper Species. Glob. Change Biol. 2009, 15, 2539–2548. [Google Scholar] [CrossRef]
- Guo, W.; Ma, C.; Kang, L. Community Change and Population Outbreak of Grasshoppers Driven by Climate Change. Curr. Opin. Insect Sci. 2024, 61, 101154. [Google Scholar] [CrossRef]
- Dey, L.S.; Simoes, M.V.P.; Hawlitschek, O.; Sergeev, M.G.; Xu, S.-Q.; Lkhagvasuren, D.; Husemann, M. Analysis of Geographic Centrality and Genetic Diversity in the Declining Grasshopper Species Bryodemella tuberculata (Orthoptera: Oedipodinae). Biodivers. Conserv. 2021, 30, 2773–2796. [Google Scholar] [CrossRef]
- Saha, H.K.; Haldar, P. Acridids as Indicators of Disturbance in Dry Deciduous Forest of West Bengal in India. Biodivers. Conserv. 2009, 18, 2343–2350. [Google Scholar] [CrossRef]
- Mariottini, Y.; De Wysiecki, M.L.; Cepeda, R.; Marinelli, C.; Bardi, C.J.; Lange, C.E. Grasshopper (Orthoptera: Acridoidea) Diversity in the Pampas Region of Argentina: Status as Revealed by Long-Term Sampling. J. Insect Conserv. 2024, 28, 1265–1283. [Google Scholar] [CrossRef]
- Ngoute, C.O.; Kekeunou, S.; Lecoq, M.; Fiemapong, A.R.N.; Um Nyobe, P.C.A.; Bilong, C.F.B. Effect of Anthropogenic Pressure on Grasshopper (Orthoptera: Acridomorpha) Species Diversity in Three Forests in Southern Cameroon. J. Orthoptera Res. 2020, 29, 25–34. [Google Scholar] [CrossRef]
- Yetchom-Fondjo, J.A.; Kekeunou, S.; Kenne, M.; Missoup, A.D.; Shen-Quan, X. Diversity, Abundance and Distribution of Grasshopper Species (Orthoptera: Acridea) in Three Different Types of Vegetation with Different Levels of Anthropogenic Disturbances in the Littoral Region of Cameroon. J. Insect Biodivers. 2020, 14, 16–33. [Google Scholar]
- Hardwick, J.L. The Community Responses of Orthoptera to Tropical Forest Degradation with Applications of DNA Barcoding for Food Web Analysis. Ph.D. Thesis, Griffith University, Brisbane, Australia, 2018. [Google Scholar] [CrossRef]
- Price, R.E. Invasions and Local Outbreaks of Four Species of Plague Locusts in South Africa: A Historical Review of Outbreak Dynamics and Patterns. Insects 2023, 14, 846. [Google Scholar] [CrossRef] [PubMed]
- Lecoq, M.; Sukirno. Drought and an Exceptional Outbreak of the Oriental Migratory Locust, Locusta migratoria manilensis (Meyen 1835) in Indonesia (Orthoptera: Acrididae). J. Orthoptera Res. 1999, 8, 153–161. [Google Scholar] [CrossRef]
- Farrow, R.A. Effects of Changing Land Use on Outbreaks of Tropical Migratory Locust, Locusta migratoria migratorioides. Insect Sci. Appl. 1987, 8, 969–975. [Google Scholar] [CrossRef]
- Kang, L.; Yonglin, C. Dynamics of Grasshopper Communities under Different Grazing Intensities in Inner Mongolian Steppes. Entomol. Sin. 1995, 2, 265–281. [Google Scholar] [CrossRef]
- Welch, J.L.; Redak, R.; Kondratieff, B.C. Effect of Cattle Grazing on the Density and Species of Grasshoppers (Orthoptera: Acrididae) of the Central Plains Experimental Range, Colorado: A Reassessment After Two Decades. J. Kans. Entomol. Soc. 1991, 64, 337–343. [Google Scholar]
- Branson, D.H.; Sword, G.A. An Experimental Analysis of Grasshopper Community Responses to Fire and Livestock Grazing in a Northern Mixed-Grass Prairie. Environ. Entomol. 2010, 39, 1441–1446. [Google Scholar] [CrossRef]
- Jepson-Innes, K.; Bock, C.E. Response of Grasshoppers (Orthoptera: Acrididae) to Livestock Grazing in Southeastern Arizona: Differences between Seasons and Subfamilies. Oecologia 1989, 78, 430–431. [Google Scholar] [CrossRef]
- Fielding, D.J.; Brusven, M.A. Grasshopper Densities on Grazed and Ungrazed Rangeland under Drought Conditions in Southern Idaho. Great Basin Nat. 1995, 55, 352–358. [Google Scholar]
- Holmes, N.D.; Smith, D.S.; Johnston, A. Effect of Grazing by Cattle on the Abundance of Grasshoppers on Fescue Grassland. J. Range Manag. 1979, 32, 310–311. [Google Scholar] [CrossRef]
- O’Neill, K.M.; Olson, B.E.; Rolston, M.G.; Wallander, R.; Larson, D.P.; Seibert, C.E. Effects of Livestock Grazing on Rangeland Grasshopper (Orthoptera: Acrididae) Abundance. Agric. Ecosyst. Environ. 2003, 97, 51–64. [Google Scholar] [CrossRef]
- O’Neill, K.M.; Olson, B.E.; Wallander, R.; Rolston, M.G.; Seibert, C.E. Effects of Livestock Grazing on Grasshopper Abundance on a Native Rangeland in Montana. Environ. Entomol. 2010, 39, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Wang, S.; Cease, A.; Kang, L. Landscape-Level Patterns of Grasshopper Communities in Inner Mongolia: Interactive Effects of Livestock Grazing and a Precipitation Gradient. Landsc. Ecol. 2015, 30, 1657–1668. [Google Scholar] [CrossRef]
- Shen, T.; Wu, Q.; Tan, Y.; Jua, X.; Han, G. Diet-Induced Gut Microbiota Shifts in Grasshoppers: Ecological Implications for Management and Adaptation under Varying Grazing Intensities. Pest Manag. Sci. 2025, 81, e8953. [Google Scholar] [CrossRef]
- Gebeyehu, S.; Samways, M.J. Responses of Grasshopper Assemblages to Long-Term Grazing Management in a Semi-Arid African Savanna. Agric. Ecosyst. Environ. 2003, 95, 613–622. [Google Scholar] [CrossRef]
- Isern-Vallverdu, J.; Pedrocchi, C. Effect of the Abandonment of Mountain Pastures on the Orthoptera Populations in the Northwest of Spain. Articulata 1994, 9, 15–23. [Google Scholar]
- Badenhausser, I.; Cordeau, S. Sown Grass Strip—A Stable Habitat for Grasshoppers (Orthoptera: Acrididae) in Dynamic Agricultural Landscapes. Agric. Ecosyst. Environ. 2012, 159, 105–111. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Q.; Chen, Y.; Chen, Z. Effects of Different Land-Use on Grasshopper Diversity in Lac Agroecosystems. Terr. Arthropod Rev. 2011, 4, 255–269. [Google Scholar] [CrossRef]
- Heidorn, T.J.; Joern, A. Feeding Preference and Spatial Distribution of Grasshoppers (Acrididae) in Response to Nitrogen Fertilization of Calamovilfa longifolia. Funct. Ecol. 1987, 1, 369–375. [Google Scholar] [CrossRef]
- Samways, M.J. Insect Conservation and Landscape Ecology: A Case-History of Bush Crickets (Tettigoniidae) in Southern France. Environ. Conserv. 1989, 16, 217–226. [Google Scholar] [CrossRef]
- Jakab, D.A.; Nagy, A. How Can an Intensively Used Agricultural Landscape Preserve Diversity of Orthoptera Assemblages? J. Insect Conserv. 2022, 26, 947–958. [Google Scholar] [CrossRef]
- Merugu, L.; Narayana, E. Orthoptera Diversity and Its Seasonal Fluctuations in Agricultural and Forest Ecosystems of Warangal District, Telangana State, India. Cah. Magellanes-NS 2024, 6, 4096–4103. [Google Scholar] [CrossRef]
- Akhtar, M.H.; Usmani, M.K.; Nayeem, M.R.; Kumar, H. Species Diversity and Abundance of Grasshopper Fauna (Orthoptera) in Rice Ecosystem. Ann. Biol. Res. 2012, 3, 2190–2193. [Google Scholar]
- Khatimah, A.; Leksono, A.S.; Yanuwiadi, B. Diversity of Grasshoppers on Agricultural Land and Savanna in Dompu Regency, Indonesia. J. Trop. Biol. 2022, 10, 203–210. [Google Scholar] [CrossRef]
- Samways, M.J.; Moore, S.D. Influence of Exotic Conifer Patches on Grasshopper (Orthoptera) Assemblages in a Grassland Matrix at a Recreational Resort, Natal, South Africa. Biol. Conserv. 1991, 57, 117–137. [Google Scholar] [CrossRef]
- Pryke, J.S.; Samways, M.J. Conservation Management of Complex Natural Forest and Plantation Edge Effects. Landsc. Ecol. 2012, 27, 73–85. [Google Scholar] [CrossRef]
- Bieringer, G.; Zulka, K.P. Shading Out Species Richness: Edge Effect of a Pine Plantation on the Orthoptera (Tettigoniidae and Acrididae) Assemblage of an Adjacent Dry Grassland. Biodivers. Conserv. 2003, 12, 1481–1495. [Google Scholar] [CrossRef]
- Sliacka, A.; Krištín, A.; NAĎO, L. Response of Orthoptera to Clear-Cuts in Beech Forests. Eur. J. Entomol. 2013, 110, 319–326. [Google Scholar] [CrossRef]
- Fenoglio, M.S.; Rossetti, M.R.; Videla, M. Negative Effects of Urbanization on Terrestrial Arthropod Communities: A Meta-Analysis. Glob. Ecol. Biogeogr. 2020, 29, 1412–1429. [Google Scholar] [CrossRef]
- Tamara, M.E.L.; Latty, T.; Threlfall, C.G.; Hochuli, D.F. Major Insect Groups Show Distinct Responses to Local and Regional Attributes of Urban Green Spaces. Landsc. Urban Plan. 2021, 216, 104238. [Google Scholar] [CrossRef]
- New, T.R. Insect Conservation and Urban Environments; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- Pernat, N.; Buchholz, S.; Schirmel, J. Urbanization Reduces Orthoptera Diversity and Changes Community Structure towards Mobile Species. Insect Conserv. Divers. 2024, 17, 259–272. [Google Scholar] [CrossRef]
- Penone, C.; Kerbiriou, C.; Julien, J.-F.; Julliard, R.; Machon, N.; Le Viol, I. Urbanisation Effect on Orthoptera: Which Scale Matters? Insect Conserv. Divers. 2012, 6, 319–327. [Google Scholar] [CrossRef]
- Melliger, R.L.; Rusterholz, H.-P.; Baur, B. Habitat- and Matrix-Related Differences in Species Diversity and Trait Richness of Vascular Plants, Orthoptera and Lepidoptera in an Urban Landscape. Urban Ecosyst. 2017, 20, 1095–1107. [Google Scholar] [CrossRef]
- Cherrill, A. Large-Scale Spatial Patterns in Species Richness of Orthoptera in the Greater London Area, United Kingdom: Relationships with Land Cover. Landsc. Res. 2015, 40, 476–485. [Google Scholar] [CrossRef][Green Version]
- Huchler, K.; Pachinger, B.; Kropf, M. Management Is More Important than Urban Landscape Parameters in Shaping Orthopteran Assemblages across Green Infrastructure in a Metropole. Urban Ecosyst. 2023, 26, 209–222. [Google Scholar] [CrossRef]
- Ancillotto, L.; Labadessa, R. Can Protected Areas and Habitats Preserve the Vulnerable Predatory Bush Cricket Saga pedo? J. Insect Conserv. 2023, 27, 615–624. [Google Scholar] [CrossRef]
- Nakajima, K.; Miyashita, T. Close Association between Grasshopper and Plant Communities in Suburban Secondary Grasslands and the Indicator Value of Grasshoppers for Conservation. Landsc. Ecol. Eng. 2021, 17, 363–374. [Google Scholar] [CrossRef]
- Oku, E.E.; Arong, G.A.; Bassey, D.A. Species Composition of Grasshoppers (Orthoptera) in Open Plots and Farmlands in Calabar Metropolis, Southern Nigeria. Pak. J. Biol. Sci. 2011, 14, 507–510. [Google Scholar] [CrossRef]
- Quandahor, P.; Kim, L.; Kim, M.; Lee, K.; Kusi, F.; Jeong, I.-H. Effects of Agricultural Pesticides on Decline in Insect Species and Individual Numbers. Environments 2024, 11, 182. [Google Scholar] [CrossRef]
- Serrão, J.E.; Plata Rueda, A.; Martínez, L.C.; Zanuncio, J.C. Side Effects of Pesticides on Non-Target Insects in Agriculture: A Mini Review. Sci. Nat. 2022, 109, 17. [Google Scholar] [CrossRef]
- Gandara, L.; Jacoby, R.; Laurent, F.; Spatuzzi, M.; Vlachopoulos, N.; Borst, N.O.; Ekmen, G.; Potel, C.M.; Garrido-Rodriguez, M.; Böhmert, A.L.; et al. Pervasive Sublethal Effects of Agrochemicals on Insects at Environmentally Relevant Concentrations. Science 2024, 386, 446–453. [Google Scholar] [CrossRef]
- Jankielsohn, A. The Importance of Insects in Agricultural Ecosystems. Adv. Entomol. 2018, 6, 62–73. [Google Scholar] [CrossRef]
- Metcalf, R.L.; Horowitz, A.R. Insect Control, 1. Fundamentals. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2014. [Google Scholar] [CrossRef]
- Brühl, C.A.; Zaller, J.G. Biodiversity Decline as a Consequence of an Inappropriate Environmental Risk Assessment of Pesticides. Front. Environ. Sci. 2019, 7, 2013–2016. [Google Scholar] [CrossRef]
- Tissier, M.L.; Shahmohamadloo, R.S.; Guzman, L.M. Pesticide Risk Assessment in a Changing World. Glob. Change Biol. 2025, 31, 70203. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, V.C.; McCarty, E.P.; Asaro, C. Limited Scope Risk Assessment for Nontarget Ground-Dwelling Arthropods from Systemic Insecticide Applications to Young Pines. Environ. Entomol. 2021, 50, 359–366. [Google Scholar] [CrossRef]
- Kaczmarek, M.; Gillich, M.; Entling, M.H.; Hoffmann, C.; Schirmel, J. Differential Responses of Orthoptera in Vineyards to Organic Farming, Pesticide Reduction, and Landscape Heterogeneity. J. Insect Conserv. 2023, 27, 729–741. [Google Scholar] [CrossRef]
- Bundschuh, R.; Schmitz, J.; Bundschuh, M.; Brühl, C.A. Does Insecticide Drift Adversely Affect Grasshoppers (Orthoptera: Saltatoria) in Field Margins? A Case Study Combining Laboratory Acute Toxicity Testing with Field Monitoring Data. Environ. Toxicol. Chem. 2012, 31, 1874–1879. [Google Scholar] [CrossRef] [PubMed]
- Azam, I.; Afsheen, S.; Zia, A.; Javed, M.; Saeed, R.; Sarwar, M.K.; Munir, B. Evaluating Insects as Bioindicators of Heavy Metal Contamination and Accumulation near Industrial Area of Gujrat, Pakistan. Biomed. Res. Int. 2015, 2015, 942751. [Google Scholar] [CrossRef]
- Devkota, B.; Schmidt, G.H. Accumulation of Heavy Metals in Food Plants and Grasshoppers from the Taigetos Mountains, Greece. Agric. Ecosyst. Environ. 2020, 78, 85–91. [Google Scholar] [CrossRef]
- Esteves Aguilar, J.; Mussali Galante, P.; Valencia Cuevas, L.; Garcia Cigarrero, A.A.; Rodríguez, A.; Castrejon Godinez, M.L.; Tovar Sanchez, E. Ecotoxicological Effects of Heavy Metal Bioaccumulation in Two Trophic Levels. Environ. Sci. Pollut. Res. 2023, 30, 49840–49855. [Google Scholar] [CrossRef]
- Liaqat, I.; Virk, N.; Ali, N.M. Recent Advances in Evaluating Insects as Bioindicators of Heavy Metal Pollution. In Heavy Metals—Recent Advances; Almayyahi, B.A., Ed.; IntechOpen: London, UK, 2023; pp. 1–17. [Google Scholar] [CrossRef]
- Gutiérrez, Y.; Ott, D.; Scherber, C. Direct and Indirect Effects of Plant Diversity and Phenoxy Herbicide Application on the Development and Reproduction of a Polyphagous Herbivore. Sci. Rep. 2020, 10, 7300. [Google Scholar] [CrossRef]
- Litt, A.R.; Steidl, R.J. Insect Assemblages Change along a Gradient of Invasion by a Nonnative Grass. Biol. Invasions 2010, 12, 3449–3463. [Google Scholar] [CrossRef]
- Yoshioka, A.; Kadoya, T.; Suda, S.-I.; Washitani, I. Impacts of Weeping Lovegrass (Eragrostis curvula) Invasion on Native Grasshoppers: Responses of Habitat Generalist and Specialist Species. Biol. Invasions 2010, 12, 531–539. [Google Scholar] [CrossRef]
- Andersen, E.M.; Cambrelin, M.N.; Steidl, R.J. Responses of Grassland Arthropods to an Invasion by Nonnative Grasses. Biol. Invasions 2019, 21, 405–416. [Google Scholar] [CrossRef]
- Samways, M.J.; Caldwell, P.M.; Osborn, R. Ground-Living Invertebrate Assemblages in Native, Planted and Invasive Vegetation in South Africa. Agric. Ecosyst. Environ. 1996, 59, 19–32. [Google Scholar] [CrossRef]
- Craig, D.P.; Bock, C.E.; Bennett, B.C.; Bock, J.H. Habitat Relationships among Grasshoppers (Orthoptera: Acrididae) at the Western Limit of the Great Plains in Colorado. Am. Midl. Nat. 1999, 142, 314–327. [Google Scholar] [CrossRef]
- Beckstead, J.; Meyer, S.E.; Augsperger, C.K. The Indirect Effects of Cheatgrass Invasion: Grasshopper Herbivory on Native Grasses Determined by Neighboring Cheatgrass. USDA For. Serv. Proc. 2008, RMRS-P-52, 41–48. [Google Scholar]
- Fielding, D.J.; Brusven, M.A. Spatial Analysis of Grasshopper Density and Ecological Disturbance on Southern Idaho Rangeland. Agric. Ecosyst. Environ. 1993, 43, 31–47. [Google Scholar] [CrossRef]
- Avanesyan, A.; Culley, T.M. Feeding Preferences of Melanoplus femurrubrum Grasshoppers on Native and Exotic Grasses: Behavioral and Molecular Approaches. Entomol. Exp. Appl. 2015, 157, 152–163. [Google Scholar] [CrossRef]
- Avanesyan, A.; Culley, T.M. Herbivory of Native and Exotic North American Prairie Grasses by Nymph Melanoplus Grasshoppers. Plant Ecol. 2015, 216, 451–464. [Google Scholar] [CrossRef]
- Avanesyan, A. Should I Eat or Should I Go? Acridid Grasshoppers and Their Novel Host Plants: Potential for Biotic Resistance. Plants 2018, 7, 83. [Google Scholar] [CrossRef]
- Fielding, D.J.; Conn, J.S. Feeding Preference for and Impact on an Invasive Weed (Crepis tectorum) by a Native, Generalist Insect Herbivore, Melanoplus borealis (Orthoptera: Acrididae). Ann. Entomol. Soc. Am. 2011, 104, 1303–1308. [Google Scholar] [CrossRef]
- Jogesh, T.; Carpenter, D.; Cappuccino, N. Herbivory on Invasive Exotic Plants and Their Non-Invasive Relatives. Biol. Invasions 2008, 10, 797–804. [Google Scholar] [CrossRef]
- Theron, K.J.; Pryke, J.S.; Samways, M.J. Identifying Managerial Legacies within Conservation Corridors Using Remote Sensing and Grasshoppers as Bioindicators. Ecol. Appl. 2022, 32, e02496. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, T. Hillside Lagomorph Grazing and Its Influence on Orthoptera. J. Orthoptera Res. 2022, 31, 157–162. [Google Scholar] [CrossRef]
- Engelhardt, E.K.; Biber, M.F.; Dolek, M.; Fartmann, T.; Hochkirch, A.; Leidinger, J.; Löffler, F.; Pinkert, S.; Poniatowski, D.; Voith, J.; et al. Consistent Signals of a Warming Climate in Occupancy Changes of Three Insect Taxa over 40 Years in Central Europe. Glob. Change Biol. 2022, 28, 3998–4012. [Google Scholar] [CrossRef] [PubMed]
- Poniatowski, D.; Beckmann, C.; Löffler, F.; Münsch, T.; Helbing, F.; Samways, M.J.; Fartmann, T. Relative Impacts of Land-Use and Climate Change on Grasshopper Range Shifts Have Changed over Time. Glob. Ecol. Biogeogr. 2020, 29, 2190–2202. [Google Scholar] [CrossRef]
- Fartmann, T.; Poniatowski, D.; Holtmann, L. Habitat Availability and Climate Warming Drive Changes in the Distribution of Grassland Grasshoppers. Agric. Ecosyst. Environ. 2021, 320, 107565. [Google Scholar] [CrossRef]
- Fartmann, T.; Poniatowski, D.; Holtmann, L. Effects of Land-Use and Climate Change on Grasshopper Assemblages Differ between Protected and Unprotected Grasslands. Basic Appl. Ecol. 2022, 63, 83–92. [Google Scholar] [CrossRef]
- Labadessa, R.; Ancillotto, L. A Tale of Two Crickets: Global Climate and Local Competition Shape the Distribution of European Oecanthus Species (Orthoptera, Gryllidae). Front. Biogeogr. 2022, 14, 57538. [Google Scholar] [CrossRef]
- Lightfoot, D.C. The Effects of Livestock Grazing and Climate Variation on Vegetation and Grasshopper Communities in the Northern Chihuahuan Desert. J. Orthoptera Res. 2018, 27, 35–51. [Google Scholar] [CrossRef]
- Buckley, L.B.; Nufio, C.R.; Kirk, E.M.; Kingsolver, J.G. Elevational Differences in Developmental Plasticity Determine Phenological Responses of Grasshoppers to Recent Climate Warming. Proc. R. Soc. B 2015, 282, e20150441. [Google Scholar] [CrossRef]
- Hao, S.; Liu, C.; Ma, C.; Guo, W.; Kang, L. Embryonic Development of Grasshopper Populations along Latitudinal Gradients Reveal Differential Thermoaccumulation for Adaptation to Climate Warming. Front. Ecol. Evol. 2021, 9, 736456. [Google Scholar] [CrossRef]
- Steck, C.E.; Bürgi, M.; Coch, T.; Duelli, P. Hotspots and Richness Pattern of Grasshopper Species in Cultural Landscapes. Biodivers. Conserv. 2007, 16, 2075–2086. [Google Scholar] [CrossRef]
- Sergeev, M.G. Conservation of Orthopteran Biological Diversity Relative to Landscape Change in Temperate Eurasia. J. Insect Conserv. 1998, 2, 247–252. [Google Scholar] [CrossRef]
- Matenaar, D.; Bazelet, C.S.; Hochkirch, A. Simple Tools for the Evaluation of Protected Areas for the Conservation of Grasshoppers. Biol. Conserv. 2015, 192, 192–199. [Google Scholar] [CrossRef]
- Pina, S.; Vasconcelos, S.; Reino, L.; Santana, J.; Beja, P.; Sánchez-Oliver, J.S.; Catry, I.; Moreira, F.; Ferreira, S. The Orthoptera of Castro Verde Special Protection Area (Southern Portugal): New Data and Conservation Value. ZooKeys 2017, 691, 19–48. [Google Scholar] [CrossRef][Green Version]
- Ruchin, A.B.; Mikhailenko, A.P. Fauna of Mantids and Orthopterans (Insecta: Mantodea, Orthoptera) of the Mordovia State Nature Reserve, Russia. Biodiversitas 2018, 19, 1194–1206. [Google Scholar] [CrossRef]
- Ogan, S.; Paulus, C.; Froehlich, C.; Renker, C.; Kolwelter, C.; Schendzielorz, M.; Danielczak, A.; Müller, K.; Eulering, H.; Hochkirch, A. Re-Surveys Reveal Biotic Homogenization of Orthoptera Assemblages as a Consequence of Environmental Change. Divers. Distrib. 2022, 28, 1795–1809. [Google Scholar] [CrossRef]
- Nagy, A.; Rácz, A.I.; Jakab, D.A.; Szanyi, S. Setting Priorities and Evaluation of Habitats for the Conservation of Orthopterans: Case Study in the Aggtelek National Park (N Hungary). Biol. Futur. 2023, 74, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Krpach, V. Revised List of Strictly Protected Insects (Lepidoptera, Coleoptera, Orthoptera and Diptera) Species in North Macedonia. ECOTEC Int. J. Sci. Environ. Technol. 2024, 4, 71–92. [Google Scholar] [CrossRef]
- Tzirkalli, E.; Zografou, K.; Willemse, L.; Vogiatzakis, I.N.; Kati, V. Conservation Prioritization of Orthoptera Assemblages on a Mediterranean Island. Diversity 2024, 16, 347. [Google Scholar] [CrossRef]
- Stefanidis, A.; Kougioumoutzis, K.; Zografou, K.; Fotiadis, G.; Tzortzakaki, O.; Willemse, L.; Kati, V. Mitigating the Extinction Risk of Globally Threatened and Endemic Mountainous Orthoptera Species: Parnassiana parnassica and Oropodisma parnassica. Insect Conserv. Divers. 2025, 18, 54–68. [Google Scholar] [CrossRef]
- Thompson, A.C.; Samways, M.J.; Bazelet, C.S. Biosphere Reserve Zones Are Equal in Terms of Katydid Ecoacoustics. Bioacoustics 2020, 29, 249–265. [Google Scholar] [CrossRef]
- Adu-Acheampong, S.; Bazelet, C.S.; Samways, M.J. Extent to Which an Agricultural Mosaic Supports Endemic Species-Rich Grasshopper Assemblages in the Cape Floristic Region Biodiversity Hotspot. Agric. Ecosyst. Environ. 2016, 227, 52–60. [Google Scholar] [CrossRef]
- Adu-Acheampong, S.; Samways, M.J. Traits and Land Transformation Change the Fortunes of Grasshopper Generalists vs. Specialists in a Biodiversity Hotspot. Biosyst. Divers. 2019, 27, 26–32. [Google Scholar] [CrossRef]
- Adu-Acheampong, S.; Samways, M.J.; Landmann, T.; Kyerematen, R.; Minkah, R.; Mukundamago, M.; Moshobane, C.M. Endemic Grasshopper Species Distribution in an Agronatural Landscape of the Cape Floristic Region, South Africa. Ecol. Eng. 2017, 105, 133–140. [Google Scholar] [CrossRef]
- Wersebeckmann, V.; Biegerl, C.; Leyer, I.; Mody, K. Orthopteran Diversity in Steep Slope Vineyards: The Role of Vineyard Type and Vegetation Management. Insects 2023, 14, 83. [Google Scholar] [CrossRef] [PubMed]
- Pryke, J.S.; Samways, M.J. Conserving Natural Heterogeneity Is Crucial for Designing Effective Ecological Networks. Landsc. Ecol. 2015, 30, 595–607. [Google Scholar] [CrossRef]
- van Schalkwyk, J.; Pryke, J.S.; Samways, M.J.; Gaigher, R. Corridor Width Determines Strength of Edge Influence on Arthropods in Conservation Corridors. Landsc. Ecol. 2020, 35, 1175–1185. [Google Scholar] [CrossRef]
- Bazelet, C.S.; Samways, M.J. Relative Importance of Management vs. Design for Implementation of Large-Scale Ecological Networks. Landsc. Ecol. 2011, 26, 341–353. [Google Scholar] [CrossRef]
- Bazelet, C.S.; Samways, M.J. Grasshopper Assemblage Response to Conservation Ecological Networks in a Timber Plantation Matrix. Agric. Ecosyst. Environ. 2011, 144, 124–129. [Google Scholar] [CrossRef]
- Davids, L.; Pryke, J.S.; Samways, M.J. Do Pinch Points in Conservation Corridors Matter? A Test Case Using Localized Butterflies and Grasshoppers. Biol. Conserv. 2025, 305, 111073. [Google Scholar] [CrossRef]
- Theron, G.J.; Pryke, J.S.; Samways, M.J. Maintaining Functional Connectivity in Grassland Corridors between Plantation Forests Promotes High-Quality Habitat and Conserves Range-Restricted Grasshoppers. Landsc. Ecol. 2022, 37, 2081–2097. [Google Scholar] [CrossRef]
- Torma, A.; Bozsó, M.; Gallé, R. Secondary Habitats Are Important in Biodiversity Conservation: A Case Study on Orthopterans along Ditch Banks. Anim. Biodivers. Conserv. 2018, 41, 97–108. [Google Scholar] [CrossRef]
- Chambers, B.Q.; Samways, M.J. Grasshopper Response to a 40-Year Experimental Burning and Mowing Regime, with Recommendations for Invertebrate Conservation Management. Biodivers. Conserv. 1998, 7, 985–1012. [Google Scholar] [CrossRef]
- László, M.; Laczi, M.; Szövényi, G. Conservation on the Edge: Unravelling Critical Drivers behind the Decline of a Mountain Grasshopper’s Marginal Populations. J. Insect Conserv. 2025, 29, 44. [Google Scholar] [CrossRef]
- Schwarz, C.; Fartmann, T. Traditional Grazing Management Creates Heterogeneous Swards and Fosters Grasshopper Densities. Insect Sci. 2022, 29, 1805–1818. [Google Scholar] [CrossRef]
- Theuerkauf, J.; Rouys, S. Do Orthoptera Need Human Land Use in Central Europe? The Role of Habitat Patch Size and Linear Corridors in the Białowieża Forest, Poland. Biodivers. Conserv. 2006, 15, 1497–1508. [Google Scholar] [CrossRef]
- Weiss, N.; Zucchi, H.; Hochkirch, A. The Effects of Grassland Management and Aspect on Orthoptera Diversity and Abundance: Site Conditions Are as Important as Management. Biodivers. Conserv. 2013, 22, 2167–2178. [Google Scholar] [CrossRef]
- Fumy, F.; Fartmann, T. Low-Intensity Land Use Fosters Species Richness of Threatened Butterflies and Grasshoppers in Mires and Grasslands. Glob. Ecol. Conserv. 2023, 41, e02357. [Google Scholar] [CrossRef]
- Stefanidis, A.; Zografou, K.; Tzortzakaki, O.; Kati, V. Orthoptera Community Dynamics and Conservation in a Natura 2000 Site (Greece): The Role of Beta Diversity. Diversity 2024, 16, 11. [Google Scholar] [CrossRef]
- Gebeyehu, S.; Samways, M.J. Topographic Heterogeneity Plays a Crucial Role for Grasshopper Diversity in a Southern African Megabiodiversity Hotspot. Biodivers. Conserv. 2006, 15, 231–244. [Google Scholar] [CrossRef]
- Gebeyehu, S.; Samways, M.J. Conservation Refugium Value of a Large Mesa for Grasshoppers in South Africa. Biodivers. Conserv. 2006, 15, 717–734. [Google Scholar] [CrossRef]
- Marini, L.; Bommarco, R.; Fontana, P.; Battisti, A. Disentangling Effects of Habitat Diversity and Area on Orthopteran Species with Contrasting Mobility. Biol. Conserv. 2010, 143, 2164–2171. [Google Scholar] [CrossRef]
- Marini, L.; Fontana, P.; Battisti, A.; Gaston, K.J. Response of Orthopteran Diversity to Abandonment of Semi-Natural Meadows. Agric. Ecosyst. Environ. 2009, 132, 232–236. [Google Scholar] [CrossRef]
- Braschler, B.; Marini, L.; Thommen, G.H.; Baur, B. Effects of Small-Scale Grassland Fragmentation and Frequent Mowing on Population Density and Species Diversity of Orthopterans: A Long-Term Study. Ecol. Entomol. 2009, 34, 321–329. [Google Scholar] [CrossRef]
- Fabriciusová, V.; Kaňuch, P.; Krištín, A. Response of Orthoptera Assemblages to Management of Montane Grasslands in the Western Carpathians. Biologia 2011, 66, 1127–1133. [Google Scholar] [CrossRef]
- Rada, S.; Mazalová, M.; Šipaš, J.; Kuras, S. Impacts of Mowing, Grazing and Edge Effect on Orthoptera of Submontane Grasslands: Perspectives for Biodiversity Protection. Pol. J. Ecol. 2014, 62, 123–138. [Google Scholar] [CrossRef]
- Helbing, F.; Fartmann, T.; Morkel, C.; Poniatowski, D. Rapid Response of Vascular Plants and Insects to Restoration of Montane Grasslands. Front. Ecol. Evol. 2023, 11, 1148266. [Google Scholar] [CrossRef]
- Ngoute, C.O.; Kekeunou, S.; Bilong Bilong, C.F. Grasshopper (Orthoptera: Acridomorpha) Diversity in Response to Fallow-Land Use in Southern Cameroon with Recommendations for Land Management. J. Insect Sci. 2023, 23, 5. [Google Scholar] [CrossRef]
- Jacot, K.; Eggenschwiler, L.; Junge, X.; Luka, H.; Bosshard, A. Improved Field Margins for a Higher Biodiversity in Agricultural Landscapes. Asp. Appl. Biol. 2007, 81, 277–281. [Google Scholar]
- Gebeyehu, S.; Samways, M.J. Grasshopper Assemblage Response to a Restored National Park (Mountain Zebra National Park, South Africa). Biodivers. Conserv. 2002, 11, 283–304. [Google Scholar] [CrossRef]
- Borchard, F.; Schulte, A.M.; Fartmann, T. Rapid Response of Orthoptera to Restoration of Montane Heathland. Biodivers. Conserv. 2013, 22, 687–700. [Google Scholar] [CrossRef]
- Moyano, L.; Cárdenas, A.M.; Gallardo, P.; Presa, J.J. Short-Term Effects of a Revegetation Program on the Orthopteran Diversity in Oak Forests of the Southern Iberian Peninsula. J. Insect Sci. 2014, 14, 290. [Google Scholar] [CrossRef]
- Branson, D.H.; Sword, G.A. Grasshopper Herbivory Affects Native Plant Diversity and Abundance in a Grassland Dominated by the Exotic Grass Agropyron cristatum. Restor. Ecol. 2009, 17, 89–96. [Google Scholar] [CrossRef]
- Cumberland, C.; Jonas, J.L.; Paschke, M.W. Impact of Grasshoppers and an Invasive Grass on Establishment and Initial Growth of Restoration Plant Species. Restor. Ecol. 2017, 25, 385–395. [Google Scholar] [CrossRef]
- Fan, S.; Yu, D.; Liu, C. The Invasive Plant Alternanthera philoxeroides Was Suppressed More Intensively than Its Native Congener by a Native Generalist: Implications for the Biotic Resistance Hypothesis. PLoS ONE 2013, 8, e83619. [Google Scholar] [CrossRef]
- Hochkirch, A.; Witzenberger, K.A.; Teerling, A.; Niemeyer, F. Translocation of an Endangered Insect Species, the Field Cricket (Gryllus campestris Linnaeus, 1758) in Northern Germany. Biodivers. Conserv. 2007, 16, 3597–3607. [Google Scholar] [CrossRef]
- Witzenberger, K.A.; Hochkirch, A. Genetic Consequences of Animal Translocations: A Case Study Using the Field Cricket, Gryllus campestris L. Biol. Conserv. 2008, 141, 3059–3068. [Google Scholar] [CrossRef]
- Matojo, N.D.; Njau, M.A. Plasticity and Biosystematics of Swarming of the Conehead Ruspolia differens Serville (Orthoptera: Conocephalidae). Int. J. Integr. Biol. 2010, 9, 97–103. [Google Scholar]
- Song, H. Density-Dependent Phase Polyphenism in Nonmodel Locusts: A Minireview. Psyche 2011, 2011, 741769. [Google Scholar] [CrossRef]
- COPR. The Locust and Grasshopper Agricultural Manual; Centre for Overseas Pest Research: London, UK, 1982. [Google Scholar]
- Mullié, W.C.; Prakash, A.; Müller, A.; Lazutkaite, E. Insecticide Use against Desert Locust in the Horn of Africa 2019–2021 Reveals a Pressing Need for Change. Agronomy 2023, 13, 819. [Google Scholar] [CrossRef]
- Mullié, W.C.; Cheke, R.A.; Young, S.; Ibrahim, A.B.; Murk, A.J. Increased and Sex-Selective Avian Predation of Desert Locusts Schistocerca gregaria Treated with Metarhizium acridum. PLoS ONE 2021, 16, e0244733. [Google Scholar] [CrossRef]
- Seyoum, E.; Negash, M. Studies on the Field Performance of Metarhizium anisopliae var. acridum (Green Muscle®) against Mixed Populations of Grasshoppers in Ethiopia. Ethiop. J. Sci. 2007, 30, 55–64. [Google Scholar] [CrossRef]
- Milner, R.J.; Hunter, D.M. Recent Developments in the Use of Fungi as Biopesticides against Locusts and Grasshoppers in Australia. J. Orthoptera Res. 2001, 10, 271–276. [Google Scholar] [CrossRef]
- Arthurs, S.; Thomas, M.B.; Langewald, J. Field Observations of the Effects of Fenitrothion and Metarhizium anisopliae var. acridum on Non-Target Ground-Dwelling Arthropods in the Sahel. Biol. Control 2003, 26, 333–340. [Google Scholar] [CrossRef]
- Langewald, J.; Stolz, I.; Everts, J.; Peveling, R. Towards the Registration of Microbial Insecticides in Africa: Non-Target Arthropod Testing on Green Muscle™, a Grasshopper and Locust Control Product Based on the Fungus Metarhizium anisopliae var. acridum. In Biological Control in IPM Systems in Africa; Neuenschwander, P., Borgemeister, C., Langewald, J., Eds.; CABI: Wallingford, UK, 2007; pp. 207–225. [Google Scholar]
- Arthurs, S.P.; Thomas, M.B.; Lawton, J.L. Seasonal Patterns of Persistence and Infectivity of Metarhizium anisopliae var. acridum in Grasshopper Cadavers in the Sahel. Entomol. Exp. Appl. 2001, 100, 69–76. [Google Scholar] [CrossRef]
- Guerrero-Guerra, C.; Reyes-Montes, M.D.R.; Toriello, C.; Hernández-Velázquez, V.; Santiago-López, I.; Mora-Palomino, L.; Calderón-Segura, M.E.; Fernández, S.D.; Calderón-Ezquerro, C. Study of the Persistence and Viability of Metarhizium acridum in Mexico’s Agricultural Area. Aerobiologia 2013, 29, 249–261. [Google Scholar] [CrossRef]
- Milner, R.J.; Samson, P.; Morton, R. Persistence of Conidia of Metarhizium anisopliae in Sugarcane Fields: Effect of Isolate and Formulation on Persistence over 3.5 Years. Biocontrol Sci. Technol. 2003, 13, 507–516. [Google Scholar] [CrossRef]
- Kassa, A.; Stephan, D.; Vidal, S.; Zimmermann, G. Laboratory and Field Evaluation of Different Formulations of Metarhizium anisopliae var. acridum Submerged Spores and Aerial Conidia for the Control of Locusts and Grasshoppers. BioControl 2004, 49, 63–81. [Google Scholar] [CrossRef]
- Aw, K.M.S.; Hue, S.M. Mode of Infection of Metarhizium spp. Fungus and Their Potential as Biological Control Agents. J. Fungi 2017, 3, 30. [Google Scholar] [CrossRef]
- Bomar, C.R.; Lockwood, J.A.; Pomerinke, M.A.; French, J.D. Multiyear Evaluation of the Effects of Nosema locustae (Microsporidia: Nosematidae) on Rangeland Grasshopper (Orthoptera: Acrididae) Population Density and Natural Biological Controls. Environ. Entomol. 1993, 22, 489–497. [Google Scholar] [CrossRef]
- Zhang, L.; Lecoq, M. Nosema locustae (Protozoa, Microsporidia), a Biological Agent for Locust and Grasshopper Control. Agronomy 2021, 11, 711. [Google Scholar] [CrossRef]
- Tounou, A.K.; Kooyman, C.; Douro-Kpindou, O.K.; Poehling, H.M. Combined Field Efficacy of Paranosema locustae and Metarhizium anisopliae var. acridum for the Control of Sahelian Grasshoppers. BioControl 2008, 53, 813–828. [Google Scholar] [CrossRef]
- Shi, W.P.; Wang, X.Y.; Yin, Y.; Zhang, Y.X.; Rizvi, U.E.H.; Tan, S.Q.; Cao, C.; Yu, H.Y.; Ji, R. Dynamics of Aboveground Natural Enemies of Grasshoppers, and Biodiversity after Application of Paranosema locustae in Rangeland. Insects 2019, 10, 224. [Google Scholar] [CrossRef]
- Wang-Peng, S.; Zheng, X.; Jia, W.T.; Li, A.M.; Camara, I.; Chen, H.X.; Tan, S.Q.; Liu, Y.Q.; Ji, R. Horizontal Transmission of Paranosema locustae (Microsporidia) in Grasshopper Populations via Predatory Natural Enemies. Pest Manag. Sci. 2018, 74, 2589–2593. [Google Scholar] [CrossRef]
- Miao, J.; Guo, Y.; Shi, W. The Persistence of Paranosema locustae after Application in Qinhai Plateau, China. Biocontrol Sci. Technol. 2012, 22, 733–735. [Google Scholar] [CrossRef]
- Lange, C.E.; Azzaro, F.G. New Case of Long-Term Persistence of Paranosema locustae (Microsporidia) in Melanopline Grasshoppers (Orthoptera: Acrididae: Melanoplinae) of Argentina. J. Invertebr. Pathol. 2008, 99, 357–359. [Google Scholar] [CrossRef]
- Lockwood, J.A. Environmental Issues Involved in Biological Control of Rangeland Grasshoppers (Orthoptera: Acrididae) with Exotic Agents. Environ. Entomol. 1993, 22, 503–518. [Google Scholar] [CrossRef]
- Lockwood, J.A. Management of Orthopteran Pests: A Conservation Perspective. J. Insect Conserv. 1998, 2, 253–261. [Google Scholar] [CrossRef]
- Branson, D.H.; Joern, A.; Sword, G.A. Sustainable Management of Insect Herbivores in Grassland Ecosystems: New Perspectives in Grasshopper Control. BioScience 2006, 56, 743–755. [Google Scholar] [CrossRef]
- Du, B.; Wei, J.; Lin, K.; Lu, L.; Ding, X.; Ye, H.; Huang, W.; Wang, N. Spatial and Temporal Variability of Grassland Grasshopper Habitat Suitability and Its Main Influencing Factors. Remote Sens. 2022, 14, 3910. [Google Scholar] [CrossRef]
- Serrano, L.S.; Pietrantuono, A.L.; Laguna, M.F.; Weigandt, M.; Amadio, M.E.; Fernández-Arhex, V. Assessing the Potential Impact of Grasshopper Outbreaks on Patagonian Wetlands through Mathematical Modelling. Sci. Rep. 2025, 15, 682. [Google Scholar] [CrossRef]
- Sun, T.; Liu, X.; Sun, G.; Long, R.; Liu, Z. Grasshopper Plague Control in the Alpine Rangelands of the Qilian Mountains, China: A Socio-Economic and Biological Approach. Land Degrad. Dev. 2016, 27, 1763–1770. [Google Scholar] [CrossRef]
- Alignan, J.-F.; Debras, J.-F.; Dutoit, T. Orthoptera Prove Good Indicators of Grassland Rehabilitation Success in the First French Natural Asset Reserve. J. Nat. Conserv. 2018, 44, 1–11. [Google Scholar] [CrossRef]
- Kati, V.; Dufrêne, M.; Legakis, A.; Grill, A.; Lebrun, P. Conservation Management for Orthoptera in the Dadia Reserve, Greece. Biol. Conserv. 2004, 115, 33–44. [Google Scholar] [CrossRef]
- Bhaskar, D.; Easa, P.S.; Sreejith, K.A.; Skejo, J.; Hochkirch, A. Large-Scale Burning for a Threatened Ungulate in a Biodiversity Hotspot Is Detrimental for Grasshoppers (Orthoptera: Caelifera). Biodivers. Conserv. 2019, 28, 3221–3237. [Google Scholar] [CrossRef]
- Crous, C.J.; Samways, M.J.; Pryke, J.S. Grasshopper Assemblage Response to Surface Rockiness in Afro-Montane Grasslands. Insect Conserv. Divers. 2014, 7, 185–194. [Google Scholar] [CrossRef]
- Crous, C.J.; Samways, M.J.; Pryke, J.S. Exploring the Mesofilter as a Novel Operational Scale in Conservation Planning. J. Appl. Ecol. 2013, 50, 205–214. [Google Scholar] [CrossRef]
- Bazelet, C.S.; Samways, M.J. Grasshopper and Butterfly Local Congruency in Grassland Remnants. J. Insect Conserv. 2012, 16, 71–85. [Google Scholar] [CrossRef]
- Görn, S.; Dobner, B.; Suchanek, A.; Fischer, K. Assessing Human Impact on Fen Biodiversity: Effects of Different Management Regimes on Butterfly, Grasshopper, and Carabid Beetle Assemblages. Biodivers. Conserv. 2014, 23, 309–326. [Google Scholar] [CrossRef]
- Kruess, A.; Tscharntke, T. Grazing Intensity and the Diversity of Grasshoppers, Butterflies, and Trap-Nesting Bees and Wasps. Conserv. Biol. 2002, 16, 1570–1580. [Google Scholar] [CrossRef]
- Pryke, J.S.; Samways, M.J. Importance of Using Many Taxa and Having Adequate Controls for Monitoring Impacts of Fire for Arthropod Conservation. J. Insect Conserv. 2012, 16, 177–185. [Google Scholar] [CrossRef]
- Yekwayo, I.; Pryke, J.S.; Gaigher, R.; Samways, M.J. Only Multi-Taxon Studies Show the Full Range of Arthropod Responses to Fire. PLoS ONE 2018, 13, e0195414. [Google Scholar] [CrossRef]
- Sperber, C.F.; Zefa, E.; de Oliveira, E.C.; de Campos, L.D.; Bolfarini, M.P.; Fianco, M.; Lhano, M.G.; Vicente, N.; Szinwelski, N.; Barrios de Souza Dias, P.G.; et al. Measuring Orthoptera Diversity. In Measuring Arthropod Biodiversity; Santos, J.C., Fernandes, G.W., Eds.; Springer Nature: Cham, Switzerland, 2021; pp. 257–287. [Google Scholar] [CrossRef]
- Giuliano, D.; Rizzioli, B. Mini-Round Box as Standardized Sampling Method for Orthopterans in Alpine and Subalpine Grasslands: A Field Study to Highlight Strengths and Weaknesses. Contrib. Entomol. 2025, 75, 213–222. [Google Scholar] [CrossRef]
- Wei, S.; Liu, X.; McNeill, M.R.; Wang, Y.; Sun, W.; Tu, X.; Wang, G.; Ban, L.; Zhang, Z.; Zhang, R. Identification of Spatial Distribution and Drivers for Grasshopper Populations Based on Geographic Detectors. Ecol. Indic. 2023, 154, 110500. [Google Scholar] [CrossRef]
- Thompson, A.C.; Bazelet, C.S.; Naskrecki, P.; Samways, M.J. Adapting the Dragonfly Biotic Index to a Katydid (Tettigoniidae) Rapid Assessment Technique: Case Study of a Biodiversity Hotspot, the Cape Floristic Region, South Africa. J. Orthoptera Res. 2017, 26, 63–71. [Google Scholar] [CrossRef]
- Landmann, T.; Ochungo, P.; Habel, J.C. Drones and Satellite Mapping for Insect Conservation. In Routledge Handbook of Insect Conservation; Pryke, J.S., Samways, M.J., New, T.R., Cardoso, P., Gaigher, R., Eds.; Routledge: Abingdon, UK; New York, NY, USA, 2024; pp. 461–474. [Google Scholar] [CrossRef]
- Rhodes, M.W.; Bennie, J.J.; Spalding, A.; Ffrench-Constant, R.H.; Maclean, I.M. Recent advances in the remote sensing of insects. Biol. Rev. 2022, 97, 343–360. [Google Scholar] [CrossRef] [PubMed]
- Cressman, K. Role of remote sensing in desert locust early warning. J. Appl. Remote Sens. 2013, 7, 075098. [Google Scholar] [CrossRef]
- Klein, I.; Oppelt, N.; Kuenzer, C. Application of Remote Sensing Data for Locust Research and Management—A Review. Insects 2021, 12, 233. [Google Scholar] [CrossRef]
- Klein, I.; Uereyen, S.; Eisfelder, C.; Pankov, V.; Oppelt, N.; Kuenzer, C. Application of geospatial and remote sensing data to support locust management. Int. J. Appl. Earth Obs. Geoinf. 2023, 117, 103212. [Google Scholar] [CrossRef]
- Escorihuela, M.J.; Merlin, O.; Stefan, V.; Moyano, G.; Eweys, O.A.; Zribi, M.; Kamara, S.; Benahi, A.S.; Ebbe, M.A.B.; Chihrane, J.; et al. SMOS based high resolution soil moisture estimates for desert locust preventive management. Remote Sens. Appl. Soc. Environ. 2018, 11, 40–50. [Google Scholar] [CrossRef]
- Piou, C.; Gay, P.E.; Benahi, A.S.; Ebbe, M.A.O.B.; Chihrane, J.; Ghaout, S.; Cisse, S.; Diakite, F.; Lazar, M.; Cressman, K.; et al. Soil moisture from remote sensing to forecast desert locust presence. J. Appl. Ecol. 2019, 56, 966–975. [Google Scholar] [CrossRef]
- Magor, J.I.; Lecoq, M.; Hunter, D.M. Preventive Control and Desert Locust Plagues. Crop Protect. 2008, 27, 1527–1533. [Google Scholar] [CrossRef]
- Yamazaki, A.; Iwasaki, S. Detection of Locust Swarms Using a Space-borne Hyperspectral Sensor: Theory and Simulation. Int. J. Remote Sens. 2025, 46, 3004–3021. [Google Scholar] [CrossRef]
- Hawlitschek, O.; Morinière, J.; Lehmann, G.U.C.; Lehmann, A.W.; Kropf, M.; Dunz, A.; Glaw, F.; Detcharoen, M.; Schmidt, S.; Hausmann, A.; et al. DNA Barcoding of Crickets, Katydids and Grasshoppers (Orthoptera) from Central Europe with Focus on Austria, Germany and Switzerland. Mol. Ecol. Resour. 2017, 17, 1037–1053. [Google Scholar] [CrossRef]
- Chen, L.P.; Chang, Y.G.; Li, J.; Wang, J.M.; Liu, J.L.; Zhi, Y.C.; Li, X.J. Application of DNA Barcoding in the Classification of Grasshoppers (Orthoptera: Acridoidea)—A Case Study of Grasshoppers from Hebei Province, China. Zootaxa 2018, 4497, 99–110. [Google Scholar] [CrossRef]
- Timm, V.F.; Gonçalves, L.T.; Valente, V.L.D.S.; Deprá, M. The Efficiency of the COI Gene as a DNA Barcode and an Overview of Orthoptera (Caelifera and Ensifera) Sequences in the BOLD System. Can. J. Zool. 2022, 100, 710–718. [Google Scholar] [CrossRef]
- Gutiérrez-Rodríguez, J.; Zaldívar-Riverón, A.; López-Estrada, E.K.; Barranco, P.; García-París, M. DNA Barcode Reference Library of Bush-crickets (Orthoptera, Tettigoniidae) from the Iberian Peninsula. Sci. Rep. 2025, 15, e33883. [Google Scholar] [CrossRef] [PubMed]
- Ragazzini, M.; Kleukers, R.M.J.C.; Willemse, L.; Ode, B.; Dey, L.-S.; Hawlitschek, O. DNA Barcoding and Species Delimitation of Crickets, Katydids, and Grasshoppers (Orthoptera) from Central and Southern Europe, with Focus on the Mediterranean Basin. Arthropod System. Phylogeny 2025, 83, 211–230. [Google Scholar] [CrossRef]
- Liu, J.; Lu, X.; Bian, X. The Complete Mitochondrial Genome of Ruspolia yunnana (Orthoptera: Tettigoniidae: Conocephalinae). Mitochondrial DNA Part B 2022, 7, 1682–1684. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Cai, Y.; Ma, L.; Chai, J.; Zhou, Z. DNA Barcoding of the Genus Gampsocleis (Orthoptera, Tettigoniidae) from China. Arch. Insect Biochem. Physiol. 2024, 115, e22070. [Google Scholar] [CrossRef]
- Matenaar, D.; Fingerle, M.; Heym, E.; Wirtz, S.; Hochkirch, A. Phylogeography of the Endemic Grasshopper Genus Betiscoides (Lentulidae) in the South African Cape Floristic Region. Mol. Phylogenet. Evol. 2018, 118, 318–329. [Google Scholar] [CrossRef]
- Kim, S.Y.; Kang, T.H.; Kim, T.W.; Seo, H.Y. DNA Barcoding of the South Korean Tettigoniidae (Orthoptera) using Collection Specimens reveals Three Potential Species Complexes. Entomol. Res. 2020, 5, 267–281. [Google Scholar] [CrossRef]
- Kundu, S.; Kumar, H.; Tyagi, K.; Chandra, K.; Kumar, K. DNA Barcoding of Selected Short-horned Grasshoppers (Orthoptera: Acrididae) from Indian Himalayan region. Mitochondrial DNA Part B 2020, 5, 3600–3605. [Google Scholar] [CrossRef]
- Ritchie, M.G.; Kidd, D.M.; Gleason, J.M. Mitochondrial DNA Variation and GIS Analysis Confirm a Secondary Origin of Geographical Variation in the Bushcricket Ephippiger ephippiger (Orthoptera: Tettigonioidea), and Resurrect Two Subspecies. Mol. Ecol. 2001, 10, 603–611. [Google Scholar] [CrossRef]
- Kaczmarczyk-Ziemba, A.; Wagner, G.K.; Staniec, B.; Zagaja, M.; Pietrykowska-Tudruj, E.; Iorgu, E.I.; Iorgu, I.Ş. Intraspecific Diversity of Myrmecophilus acervorum (Orthoptera: Myrmecophilidae) Indicating an Ongoing Cryptic Speciation. Sci. Rep. 2024, 14, e23984. [Google Scholar] [CrossRef]
- Uchida, K.; Shinohara, T.; Takahashi, S.; Nakahama, N.; Takami, Y.; Ushimaru, A. Rediscovery of Celes akitanus (Orthoptera: Acrididae) from Semi-natural Grasslands in Japan. Entomol. Sci. 2016, 19, 89–96. [Google Scholar] [CrossRef]
- Romero, M.L.; Colombo, P.C.; Remis, M.I. Microsatellite DNA Analysis of Population Structure in Cornops aquaticum (Orthoptera: Acrididae), over a Cline for Three Robertsonian Translocations. Evol. Ecol. 2017, 31, 937–953. [Google Scholar] [CrossRef]
- Kim, M.; Lee, H.R.; Lee, J.E.; Cha, D. Identification of the Endangered Species Haplotropis brunneriana (Insecta: Orthoptera) from South Korea using Exuviae. J. Insect Conserv. 2024, 28, 869–876. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, L.; Qiu, Z.; Yuan, H. The Gut Microbiota Diversity of Five Orthoptera (Insecta, Polyneoptera) Insects Determined by DNA Metabarcoding. Biodivers. Data J. 2023, 11, e98162. [Google Scholar] [CrossRef]
- Augustyniak, M.; Kędziorski, A.; Łaszczyca, P.; Sawczyn, T.; Tarnawska, M.; Zawisza-Raszka, A. DNA Damage in Grasshopper Chorthippus brunneus (Orthoptera) Hatchlings Following Paraquat Exposure. Chemosphere 2015, 125, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Karpeta-Kaczmarek, J.; Kubok, M.; Dziewięcka, M.; Sawczyn, T.; Augustyniak, M. The Level of DNA Damage in Adult Grasshoppers Chorthippus biguttulus (Orthoptera, Acrididae) Following Dimethoate Exposure is Dependent on the Insects’ Habitat. Environ. Pollut. 2016, 215, 266–272. [Google Scholar] [CrossRef]
- Abdelfattah, E.A.; Augustyniak, M.; Yousef, H.A. Biomonitoring of Genotoxicity of Industrial Fertilizer Pollutants in Aiolopus thalassinus (Orthoptera: Acrididae) using Alkaline Comet Assay. Chemosphere 2017, 182, 762–770. [Google Scholar] [CrossRef]
- Gustavino, B.; Meschini, R.; Franzetti, G.; Gratton, P.; Allegrucci, G.; Sbordoni, V. Genotoxicity Testing for Radon Exposure: Dolichopoda (Orthoptera, Rhaphidophoridae) as Potential Bio-indicator of Confined Environments. Curr. Zool. 2014, 60, 299–307. [Google Scholar] [CrossRef]
- Yousef, H.A.; Abdelfattah, E.A.; Augustyniak, M. Antioxidant Enzyme Activity in Responses to Environmentally Induced Oxidative Stress in the 5th instar Nymphs of Aiolopus thalassinus (Orthoptera: Acrididae). Environ. Sci. Pollut. Res. 2019, 26, 3823–3833. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.X.; Guan, D.L.; Xu, S.Q.; Wang, W.Q. Characterization of the Complete Mitochondrial Genome of the Ecotoxicity Indicator Insect Aiolopus thalassinus (Orthoptera; Acrididae; Oedipodinae). Conserv. Genet. Resour. 2017, 9, 423–426. [Google Scholar] [CrossRef]
- Svetlíková, S.; Svetlík, J.; Šuvada, R.; Šibíková, M.; Šibík, J.; Jarolímek, I.; Pekárová, S.; Hawlitschek, O. DNA Metabarcoding of Faecal Samples Reveals Life-history Traits in the Critically Endangered Bush-cricket. J. Insect Conserv. 2025, 29, 63. [Google Scholar] [CrossRef]
- Zheng, X.; Zhu, Q.; Qin, M.; Zhou, Z.; Liu, C.; Wang, L.; Shi, F. The Role of Feeding Characteristics in Shaping Gut Microbiota Composition and Function of Ensifera (Orthoptera). Insects 2022, 13, 719. [Google Scholar] [CrossRef]
- Beng, K.C.; Tomlinson, K.W.; Shen, X.H.; Surget-Groba, Y.; Hughes, A.C.; Corlett, R.T.; Slik, J.F. The Utility of DNA Metabarcoding for Studying the Response of Arthropod Diversity and Composition to Land-use Change in the Tropics. Sci. Rep. 2016, 6, e24965. [Google Scholar] [CrossRef]
- Weber, S.; Stothut, M.; Mahla, L.; Kripp, A.; Hirschler, L.; Lenz, N.; Junker, A.; Künzel, S.; Krehenwinkel, H. Plant-derived Environmental DNA Complements Diversity Estimates from Traditional Arthropod Monitoring Methods but Outperforms them Detecting Plant–arthropod Interactions. Mol. Ecol. Resour. 2024, 24, e13900. [Google Scholar] [CrossRef]
- Hupało, K.; Schmidt, S.; Macher, T.H.; Weiss, M.; Leese, F. Fresh Insights into Mediterranean Biodiversity: Environmental DNA Reveals Spatio-temporal Patterns of Stream Invertebrate Communities on Sicily. Hydrobiologia 2022, 849, 155–173. [Google Scholar] [CrossRef]
- Allen, M.C.; Lockwood, J.L.; Kwait, R.; Vastano, A.R.; Peterson, D.L.; Tkacenko, L.A.; Kisurin, A.; Stringham, O.; Angle, J.; Jaffe, B. Using surface environmental DNA to assess arthropod biodiversity within a forested ecosystem. Environ. DNA 2023, 5, 1652–1666. [Google Scholar] [CrossRef]
- Pelletier, T.A.; Stefaniak, K.R.; Vigilante, T.E.; Reavis, D.; Mekus, A.; Mohamed, D.A.; Lau, J.K. Documenting Emerging Insects, Environmental DNA, and Metal Concentrations in a Small Appalachian Stream. Northeast. Nat. 2022, 29, 171–197. [Google Scholar] [CrossRef]
- Banerjee, P.; Stewart, K.A.; Antognazza, C.M.; Bunholi, I.V.; Deiner, K.; Barnes, M.A.; Saha, S.; Verdier, H.; Doi, H.; Maity, J.P.; et al. Plant–animal Interactions in the Era of Environmental DNA (eDNA)—A Review. Environ. DNA 2022, 4, 987–999. [Google Scholar] [CrossRef]
- Mathieu, C.; Hermans, S.M.; Lear, G.; Buckley, T.R.; Lee, K.C.; Buckley, H.L. A Systematic Review of Sources of Variability and Uncertainty in eDNA Data for Environmental Monitoring. Front. Ecol. Evol. 2020, 8, e135. [Google Scholar] [CrossRef]
- Harper, L.R.; Buxton, A.S.; Rees, H.C.; Bruce, K.; Brys, R.; Halfmaerten, D.; Read, D.S.; Watson, H.V.; Sayer, C.D.; Jones, E.P.; et al. Prospects and Challenges of Environmental DNA (eDNA) Monitoring in Freshwater Ponds. Hydrobiologia 2019, 826, 25–41. [Google Scholar] [CrossRef]
- Krehenwinkel, H.; Weber, S.; Broekmann, R.; Melcher, A.; Hans, J.; Wolf, R.; Hochkirch, A.; Kennedy, S.R.; Koschorreck, J.; Künzel, S.; et al. Environmental DNA from Archived Leaves Reveals Widespread Temporal Turnover and Biotic Homogenization in Forest Arthropod Communities. eLife 2022, 11, e78521. [Google Scholar] [CrossRef]
- Riede, K. Acoustic Monitoring of Orthoptera and its Potential for Conservation. J. Insect Conserv. 1998, 2, 217–223. [Google Scholar] [CrossRef]
- Diwakar, S.; Jain, M.; Balakrishnan, R. Psychoacoustic Sampling as a Reliable, Non-invasive Method to Monitor Orthopteran Species Diversity in Tropical Forests. Biodivers. Conserv. 2007, 16, 4081–4093. [Google Scholar] [CrossRef]
- Tan, M.K.; Duncan, J.; Wahab, R.H.A.; Lee, C.-Y.; Japir, R.; Chung, A.Y.C.; Baroga-Barbecho, J.B.; Yap, S.A.; Montealegre-Z, F. The Calling Songs of Some Katydids (Orthoptera: Tettigonioidea) from the Tropical Forests of Southeast Asia. J. Orthoptera Res. 2023, 32, 1–24. [Google Scholar] [CrossRef]
- Bennett, D.; Nissen, H.; Maschke, M.A.; Reck, H.; Diekötter, T. Recent Technological Developments Allow for Passive Acoustic Monitoring of Orthoptera (Grasshoppers and Crickets) in Research and Conservation across a Broad Range of Temporal and Spatial Scales. Basic Appl. Ecol. 2025; in press. [Google Scholar] [CrossRef]
- Riede, K.; Balakrishnan, R. Acoustic Monitoring for Tropical Insect Conservation. Philos. Trans. R. Soc. B 2025, 380, 20240046. [Google Scholar] [CrossRef]
- McNeil, D.J.; Grozinger, C.M. Singing in the Suburbs: Point Count Surveys Efficiently Reveal Habitat Associations for Nocturnal Orthoptera across an Urban to Rural Gradient. J. Insect Conserv. 2020, 24, 1031–1043. [Google Scholar] [CrossRef]
- Roca, I.T.; Proulx, R. Acoustic Assessment of Species Richness and Assembly Rules in Ensiferan Communities from Temperate Ecosystems. Ecology 2016, 97, 116–123. [Google Scholar] [CrossRef]
- Nityananda, V.; Balakrishnan, R. A Diversity of Songs among Morphologically Indistinguishable Katydids of the Genus Mecopoda (Orthoptera: Tettigoniidae) from Southern India. Bioacoustics 2006, 15, 223–250. [Google Scholar] [CrossRef]
- Walcher, R.; Hussain, R.I.; Sachslehner, L.; Zaller, J.G.; Arnberger, A.; Frank, T. Assessing Grasshopper Communities in Mountainous Meadows: A Comparison of a Visual-Acoustic and a Novel Purely Acoustic Soundscape Method. Entomol. Exp. Appl. 2022, 170, 895–901. [Google Scholar] [CrossRef]
- Grant, P.B.C.; Samways, M.J. Use of Ecoacoustics to Determine Biodiversity Patterns across Ecological Gradients. Conserv. Biol. 2016, 30, 1320–1329. [Google Scholar] [CrossRef] [PubMed]
- van der Mescht, A.C.; Pryke, J.S.; Gaigher, R.; Samways, M.J. Ecological and Acoustic Responses of Bush Crickets to Anthropogenic and Natural Ecotones. Biodivers. Conserv. 2021, 30, 3859–3878. [Google Scholar] [CrossRef]
- van der Mescht, A.; Pryke, J.S.; Gaigher, R.; Samways, M.J. Sympatric Bush Cricket Species Co-Exist across a Complex Landscape by Optimising Both Acoustic and Ecological Space. Bioacoustics 2022, 31, 241–260. [Google Scholar] [CrossRef]
- Samways, M.J. Effect of Farming on Population Movements and Acoustic Behaviour of Two Bush Crickets (Orthoptera, Tettigoniidae). Bull. Entomol. Res. 1977, 67, 471–481. [Google Scholar] [CrossRef]
- Samways, M.J. Bush Cricket Interspecific Acoustic Interactions in the Field (Orthoptera, Tettigoniidae). J. Nat. Hist. 1977, 11, 155–168. [Google Scholar] [CrossRef]
- O‘Keeffe, S.; Stein, S.; Curran, M.; Baumgart, L.; Zikeli, S.; Siegmund-Schultze, M. How to Square the Circle? A Conceptual Framework Synergising Strategies for Circular Agriculture to Tackle Climate Change and Enhance Overall On-Farm Sustainability. Ambio 2025, 54, 1334–1352. [Google Scholar] [CrossRef] [PubMed]
- Kéfi, S.; Saade, C.; Berlow, E.L.; Cabral, J.S.; Fronhofer, E.A. Scaling Up Our Understanding of Tipping Points. Philos. Trans. R. Soc. B 2022, 377, 20210386. [Google Scholar] [CrossRef] [PubMed]
- Pauly, D. Anecdotes and the Shifting Baseline Syndrome of Fisheries. Trends Ecol. Evol. 1995, 10, 430. [Google Scholar] [CrossRef]
- Samways, M.J.; Lockwood, J.A. Orthoptera Conservation: Pests and Paradoxes. J. Insect Conserv. 1998, 2, 143–149. [Google Scholar] [CrossRef]
- Andujar, A. White, Black, and Grey: The Role of Human Perception in Grasshopper Management Strategies. Bachelor’s Thesis, University of Colorado at Boulder, Boulder, CO, USA, 2013; 54p. [Google Scholar]
- Miles, A. Evolution of a Response to Smoke Within the Arthropoda. Master’s Thesis, Western Kentucky University, Bowling Green, KY, USA, 2024. [Google Scholar]
- Simpson, S.J.; Sword, G.A. Locusts. Curr. Biol. 2008, 18, R364–R366. [Google Scholar] [CrossRef] [PubMed]
- Swinton, A.H. Insect Variety: Its Propagation and Distribution; Cassell, Petter, Galpin and Co.: London, UK, 1880. [Google Scholar]
- Cardoso, P.; Fukushima, C.S.; Grames, E.M.; Kwak, M.L.; Milčić, M.; Wagner, D. Global Threats to Insects. In Routledge Handbook of Insect Conservation; Pryke, J.S., Samways, M.J., New, T.R., Cardoso, P., Gaigher, R., Eds.; Routledge: Abingdon, UK; New York, NY, USA, 2024; pp. 63–76. [Google Scholar] [CrossRef]
- Badenhausser, I.; Gouat, M.; Goarant, A.; Cornulier, T.; Bretagnolle, V. Spatial Autocorrelation in Farmland Grasshopper Assemblages (Orthoptera: Acrididae) in Western France. Environ. Entomol. 2012, 41, 1050–1061. [Google Scholar] [CrossRef]
- Samways, M.J.; Gaigher, R.; Deacon, C.; Pryke, J.S. Heterogeneous Conservation Corridors of Remnant Vegetation Protect Biodiversity in South African Timber Mosaics. Biodivers. Conserv. 2025, 34, 357–379. [Google Scholar] [CrossRef]
- Prior, C. Susceptibility of Target Acridoids and Non-Target Organisms to Metarhizium anisopliae and M. flavoviride. In New Strategies in Locust Control; Krall, S., Peveling, R., Ba Diallo, D., Eds.; Birkhäuser: Basel, Switzerland, 1997; pp. 369–375. [Google Scholar]
- Outhwaite, C.L.; Cooke, R.; Millard, J.; Bladon, A.J. Pervasive and Synergistic Effects of Climate Change. In Routledge Handbook of Insect Conservation; Pryke, J.S., Samways, M.J., New, T.R., Cardoso, P., Gaigher, R., Eds.; Routledge: Abingdon, UK; New York, NY, USA, 2024; pp. 92–107. [Google Scholar] [CrossRef]
- Bladon, A.J.; Cooke, R.; Millard, J.; Outhwaite, C.L. Practical Solutions to Climate Change for Insect Conservation. In Routledge Handbook of Insect Conservation; Pryke, J.S., Samways, M.J., New, T.R., Cardoso, P., Gaigher, R., Eds.; Routledge: Abingdon, UK; New York, NY, USA, 2024; pp. 108–117. [Google Scholar] [CrossRef]
- Harvey, J.A.; Tougeron, K.; Gols, R.; Heinen, R.; Abarca, M.; Abram, P.K.; Basset, Y.; Berg, M.; Boggs, C.; Brodeur, J.; et al. Scientists’ Warning on Climate Change and Insects. Ecol. Monogr. 2023, 93, e1553. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.