Evaluation and Screening of Rapeseed Varieties (Brassica napus L.) Suitable for Mechanized Harvesting with High Yield and Quality
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Materials
2.2. Experimental Design
2.3. Measurement Index and Methods
2.3.1. Plant Biomass and Leaf Area Index
2.3.2. Plant Lodging Investment
2.3.3. Plant Architecture, Seed Yield, and Quality Measurement
2.3.4. Evaluation of the Comprehensive Performance of Variety Suitable for Machine Harvest with High Yield
2.4. Statistical Analysis
3. Results
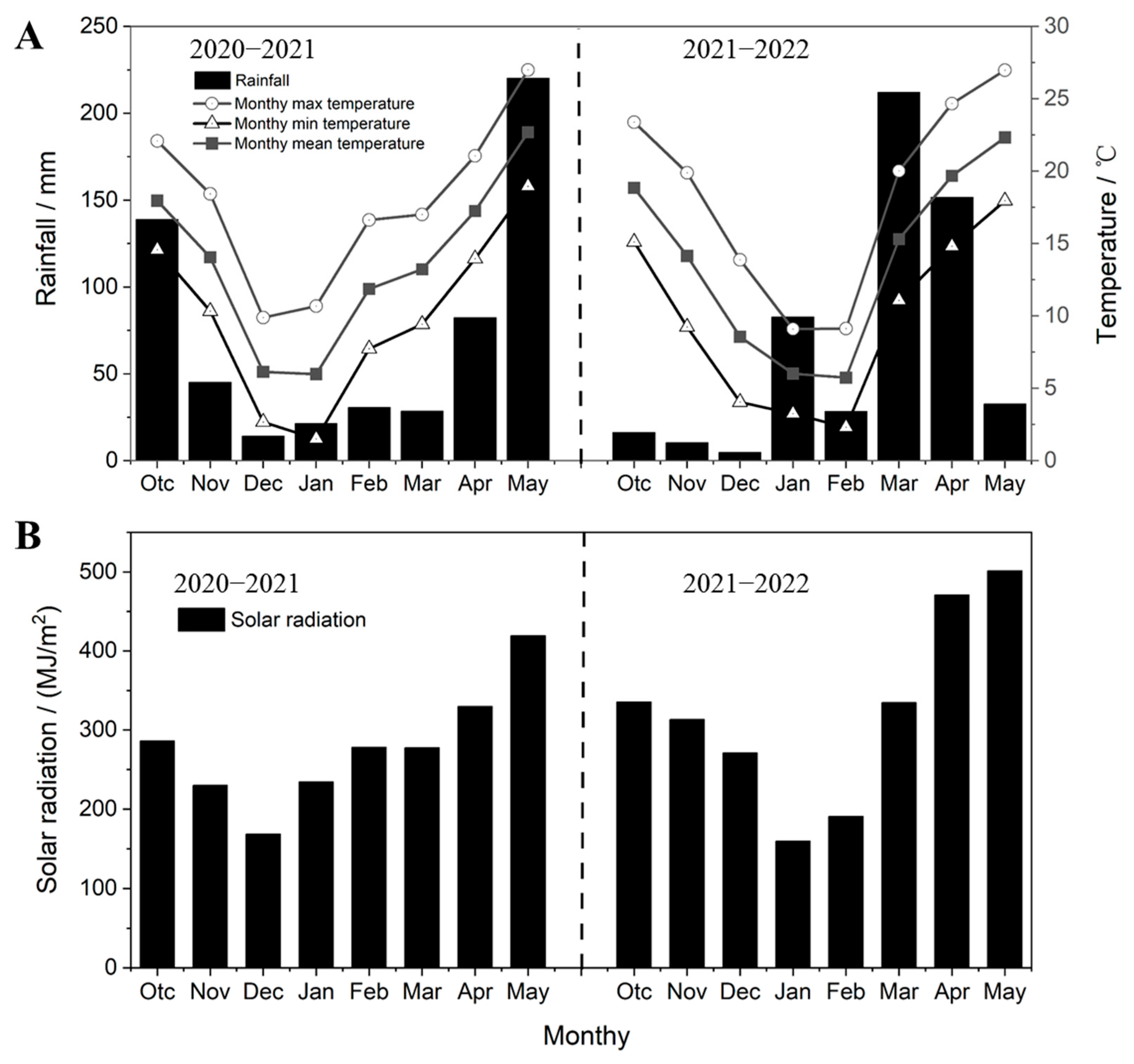
3.1. Climate Data
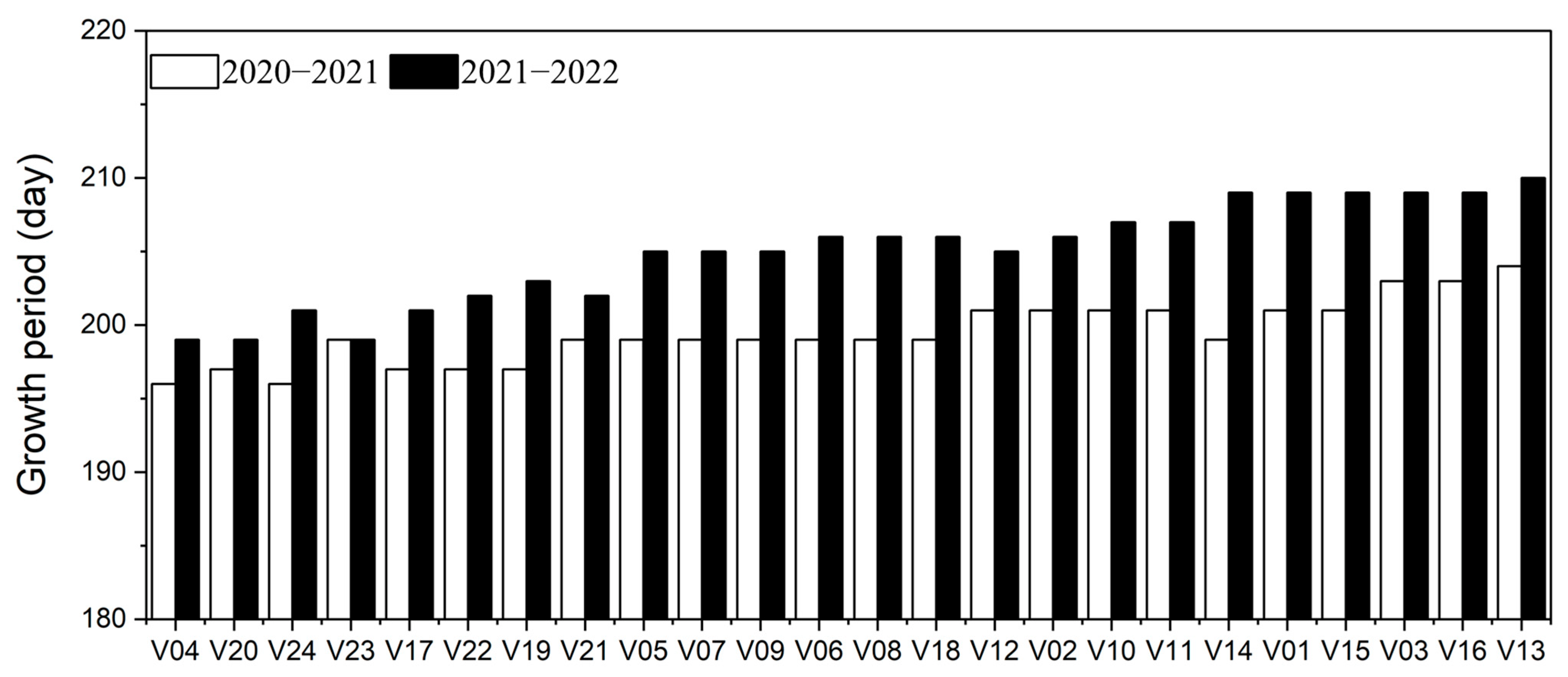
3.2. Growth Period
3.3. Yield Component and Seed Quality
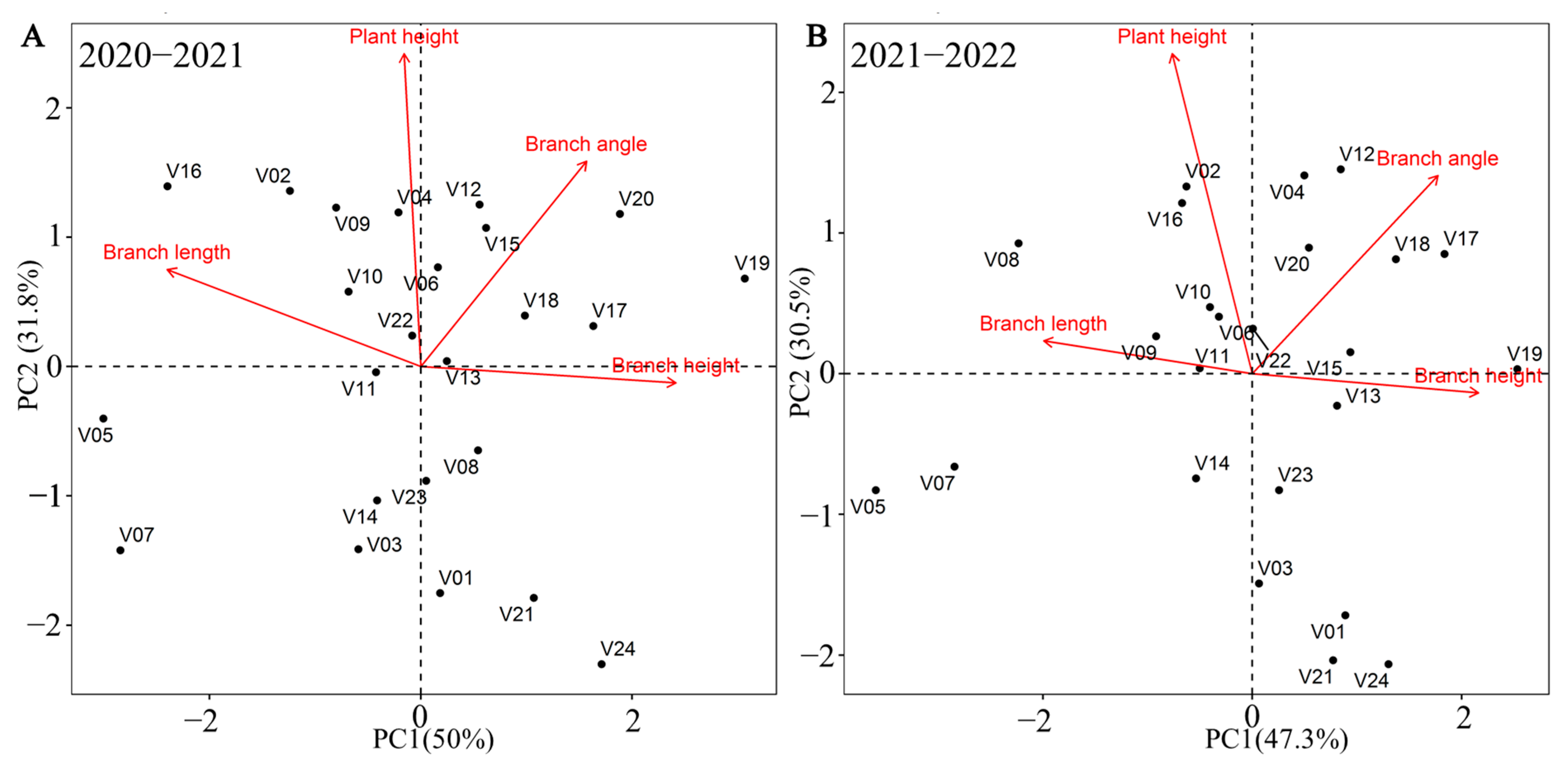
3.4. Plant Architecture
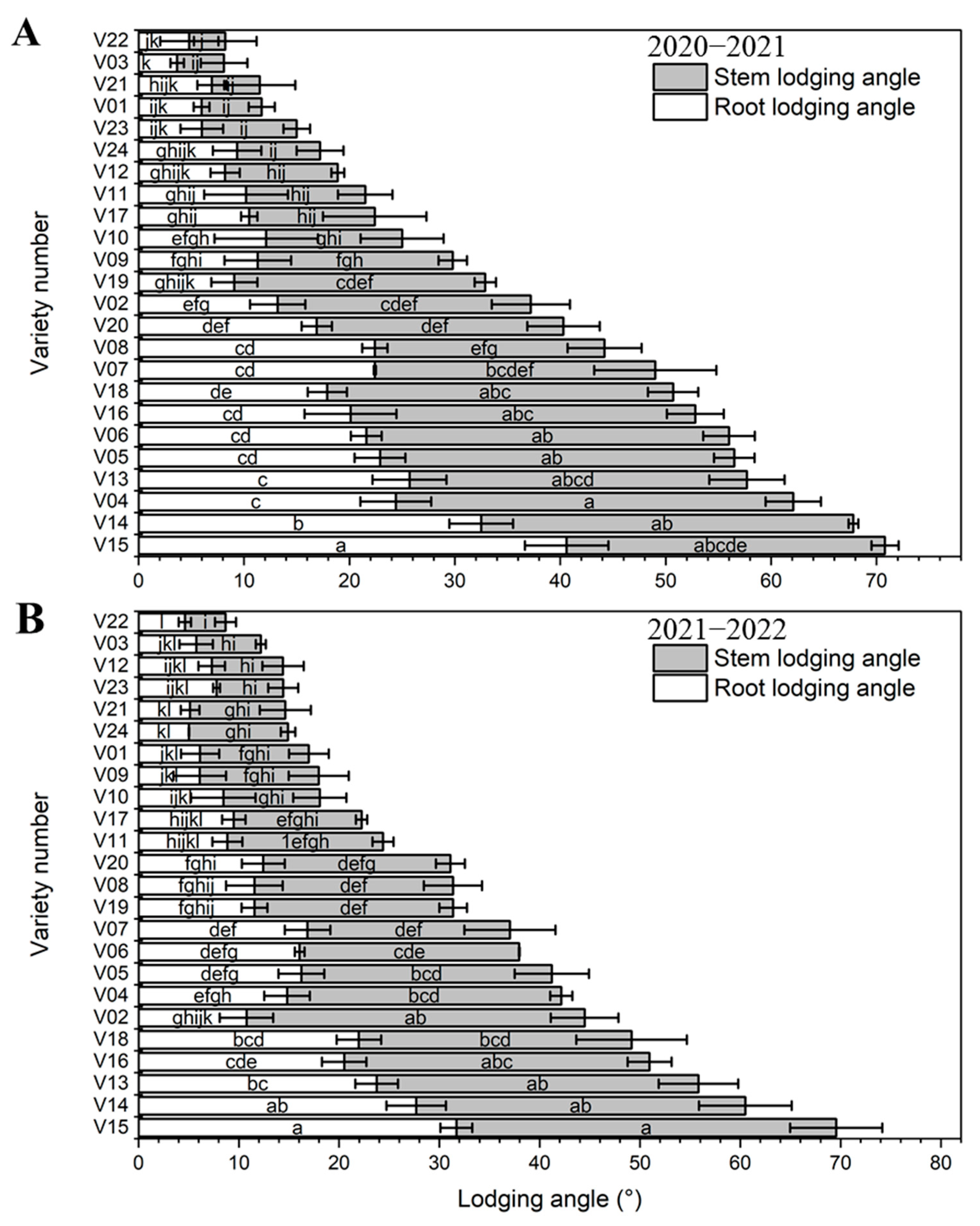
3.5. Lodging Resistance
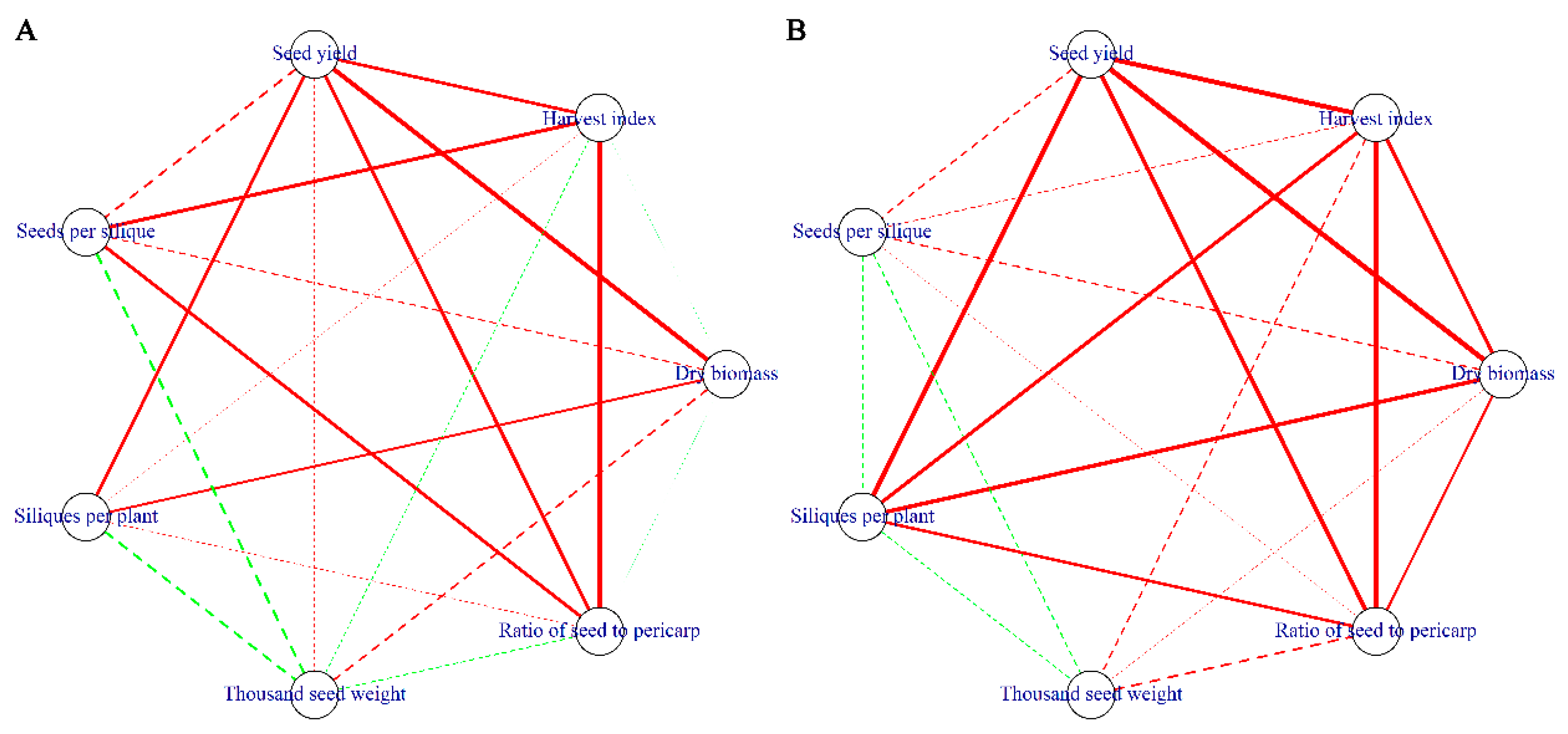
3.6. Path Analysis for the Seed Yield, Plant Architecture, Lodging, and Growth Rate
3.7. A Comprehensive Evaluation for Mechanized Harvesting and High Yield
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bonjean, A.P.; Dequidt, C.; Sang, T. Rapeseed in China. OCL 2016, 23, D605. [Google Scholar] [CrossRef]
- Wang, H. Strategy for rapeseed industry development based on the analysis of rapeseed production and demand in China. Chin. J. Oil Crop Sci. 2007, 29, 101–105. [Google Scholar]
- Lu, C.; Napier, J.A.; Clemente, T.E.; Cahoon, E.B. New frontiers in oilseed biotechnology: Meeting the global demand for vegetable oils for food, feed, biofuel, and industrial applications. Curr. Opin. Biotechnol. 2011, 22, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Zhang, J.; Khan, M.N.; Liu, J.; Xu, Z.; Hu, L. Temperature variation caused by sowing dates significantly affects floral initiation and floral bud differentiation processes in rapeseed (Brassica napus L.). Plant Sci. 2018, 271, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Lin, R.; Cheng, T.; Hu, L. Low temperature rather than nitrogen application mainly modulates the floral initiation of different ecotypes of rapeseed (Brassica napus L.). Agronomy 2022, 12, 1624. [Google Scholar] [CrossRef]
- Lopez, F.; Barclay, G. Plant anatomy and physiology. In Pharmacognosy; Elsevier: Amsterdam, The Netherlands, 2017; pp. 45–60. [Google Scholar]
- Barthélémy, D.; Caraglio, Y. Plant architecture: A dynamic, multilevel and comprehensive approach to plant form, structure and ontogeny. Ann. Bot. 2007, 99, 375–407. [Google Scholar] [CrossRef]
- Liu, H. Genetics and breeding of rapeseed. In Genetics and Breeding of Rapeseed; Liu, H., Ed.; Shanghai Scientific and Technical Publishers: Shanghai, China, 1985; pp. 338–339. [Google Scholar]
- Qing, Y.; Li, Y.; Xu, L.; Ma, Z. Screen oilseed rape (Brassica napus) suitable for low-loss mechanized harvesting. Agriculture 2021, 11, 504. [Google Scholar] [CrossRef]
- Ren, R.; Liu, W.; Yao, M.; Jia, Y.; Huang, L.; Li, W.; He, X.; Guan, M.; Liu, Z.; Guan, C. Regional association and transcriptome analysis revealed candidate genes controlling plant height in Brassica napus. Mol. Breed. 2022, 42, 69. [Google Scholar] [CrossRef]
- Qing, Y.; Li, Y.; Xu, L.; Ma, Z.; Tan, X.; Wang, Z. Oilseed rape (Brassica napus L.) pod shatter resistance and its relationship with whole plant and pod characteristics. Ind. Crops Prod. 2021, 166, 113459. [Google Scholar] [CrossRef]
- Khan, S.; Anwar, S.; Kuai, J.; Noman, A.; Shahid, M.; Din, M.; Ali, A.; Zhou, G. Alteration in yield and oil quality traits of winter rapeseed by lodging at different planting density and nitrogen rates. Sci. Rep. 2018, 8, 634. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, K.; An, T.; Tian, Z.; Dun, X.; Shi, J.; Wang, X.; Deng, J.; Wang, H. Genetic dissection of branch architecture in oilseed rape (Brassica napus L.) germplasm. Front. Plant Sci. 2022, 4443, 1053459. [Google Scholar] [CrossRef]
- Sun, C.; Wang, B.; Wang, X.; Hu, K.; Li, K.; Li, Z.; Li, S.; Yan, L.; Guan, C.; Zhang, J. Genome-wide association study dissecting the genetic architecture underlying the branch angle trait in rapeseed (Brassica napus L.). Sci. Rep. 2016, 6, 33673. [Google Scholar] [CrossRef]
- Wang, W.; Qiong, H.; Mei, D.; Li, Y.; Hui, W.; Jun, W.; Li, F.; Jia, L. Evaluation of branch and pod angle measurement based on digital images from Brassica napus L. Chin. J. Oil Crop Sci. 2015, 37, 566. [Google Scholar]
- Wang, W.; Chu, W.; Mei, D.; Cheng, H.; Zhu, L.; Fu, L.; Hu, Q.; Liu, J. Quantitative trait loci mapping for branch angle and candidate gene screening in Brassica napus L. Acta Agron. Sin. 2019, 45, 37–45. [Google Scholar] [CrossRef]
- Li, H.; Cheng, X.; Zhang, L.; Hu, J.; Zhang, F.; Chen, B.; Xu, K.; Gao, G.; Li, H.; Li, L. An integration of genome-wide association study and gene co-expression network analysis identifies candidate genes of stem lodging-related traits in Brassica napus. Front. Plant Sci. 2018, 9, 796. [Google Scholar] [CrossRef]
- Liu, S.; Raman, H.; Xiang, Y.; Zhao, C.; Huang, J.; Zhang, Y. De novo design of future rapeseed crops: Challenges and opportunities. Crop J. 2022, 10, 587–596. [Google Scholar] [CrossRef]
- Alizadeh, M.R.; Bagheri, I.; Payman, M.H. Evaluation of a rice reaper used for rapeseed harvesting. Am.-Eurasian J. Agric. Environ. Sci. 2007, 2, 388–397. [Google Scholar]
- Zheng, M.; Terzaghi, W.; Wang, H.; Hua, W. Integrated strategies for increasing rapeseed yield. Trends Plant Sci. 2022, 27, 742–745. [Google Scholar] [CrossRef]
- Kuai, J.; Sun, Y.; Guo, C.; Zhao, L.; Zuo, Q.; Wu, J.; Zhou, G. Root-applied silicon in the early bud stage increases the rapeseed yield and optimizes the mechanical harvesting characteristics. Field Crops Res. 2017, 200, 88–97. [Google Scholar] [CrossRef]
- Zuo, Q.; Kuai, J.; Zhao, L.; Hu, Z.; Wu, J.; Zhou, G. The effect of sowing depth and soil compaction on the growth and yield of rapeseed in rice straw returning field. Field Crops Res. 2017, 203, 47–54. [Google Scholar] [CrossRef]
- Krishnan, A.R.; Kasim, M.M.; Hamid, R.; Ghazali, M.F. A modified CRITIC method to estimate the objective weights of decision criteria. Symmetry 2021, 13, 973. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Ftatistical Fomputing: Vienna, Austria, 2018. [Google Scholar]
- Hu, Q.; Hua, W.; Yin, Y.; Zhang, X.; Liu, L.; Shi, J.; Zhao, Y.; Qin, L.; Chen, C.; Wang, H. Rapeseed research and production in China. Crop J. 2017, 5, 127–135. [Google Scholar] [CrossRef]
- Pilarczyk, W. The relation between GxE interaction and the number of locations in series of oilseed rape trials. Biom. Lett. 2013, 50, 53–60. [Google Scholar] [CrossRef]
- Puhl, L.E.; Miralles, D.J.; López, C.G.; Iriarte, L.B.; Rondanini, D.P. Genotype × environment interaction on the yield of spring oilseed rape (Brassica napus) under rainfed conditions in Argentine Pampas. J. Agric. Sci. 2019, 157, 235–244. [Google Scholar] [CrossRef]
- Zhang, H.; Berger, J.D.; Herrmann, C. Yield stability and adaptability of canola (Brassica napus L.) in multiple environment trials. Euphytica 2017, 213, 155. [Google Scholar] [CrossRef]
- Zhang, H.; Turner, N.C.; Poole, M.L. Source–sink balance and manipulating sink–source relations of wheat indicate that the yield potential of wheat is sink-limited in high-rainfall zones. Crop Pasture Sci. 2010, 61, 852–861. [Google Scholar] [CrossRef]
- Li, S.; Zhu, Y.; Varshney, R.K.; Zhan, J.; Zheng, X.; Shi, J.; Wang, X.; Liu, G.; Wang, H. A systematic dissection of the mechanisms underlying the natural variation of silique number in rapeseed (Brassica napus L.) germplasm. Plant Biotechnol. J. 2020, 18, 568–580. [Google Scholar] [CrossRef]
- Wang, C.; Hai, J.; Yang, J.; Tian, J.; Chen, W.; Chen, T.; Luo, H.; Wang, H. Influence of leaf and silique photosynthesis on seeds yield and seeds oil quality of oilseed rape (Brassica napus L.). Eur. J. Agron. 2016, 74, 112–118. [Google Scholar] [CrossRef]
- Habibi, M.; Majidian, M.; Shoja, T.; Rabiee, M. Effects of S, B and Zn, on seed yield, nutrient concentration and seed quality of rapeseed (Brassica napus L.). J. Oil Plants Prod. 2016, 2, 1–12. [Google Scholar]
- Fang, C.; Wang, Z.; Wang, P.; Song, Y.; Ahmad, A.; Dong, F.; Hong, D.; Yang, G. Heterosis derived from nonadditive effects of the BnFLC homologs coordinates early flowering and high yield in rapeseed (Brassica napus l.). Front. Plant Sci. 2022, 12, 3351. [Google Scholar] [CrossRef]
- Lu, G.-Y.; Zhang, F.; Zheng, P.-Y.; Cheng, Y.; Feng-Ian, L.; Fu, G.-P.; Zhang, X.-K. Relationship among yield components and selection criteria for yield improvement in early rapeseed (Brassica napus L.). Agric. Sci. China 2011, 10, 997–1003. [Google Scholar] [CrossRef]
- Zhou, Q.; Han, D.; Mason, A.S.; Zhou, C.; Zheng, W.; Li, Y.; Wu, C.; Fu, D.; Huang, Y. Earliness traits in rapeseed (Brassica napus): SNP loci and candidate genes identified by genome-wide association analysis. DNA Res. 2018, 25, 229–244. [Google Scholar] [CrossRef]
- Huang, L.; Min, Y.; Schiessl, S.; Xiong, X.; Jan, H.U.; He, X.; Qian, W.; Guan, C.; Snowdon, R.J.; Hua, W. Integrative analysis of GWAS and transcriptome to reveal novel loci regulation flowering time in semi-winter rapeseed. Plant Sci. 2021, 310, 110980. [Google Scholar] [CrossRef]
- Wei, S.; Li, X.; Lu, Z.; Zhang, H.; Ye, X.; Zhou, Y.; Li, J.; Yan, Y.; Pei, H.; Duan, F. A transcriptional regulator that boosts grain yields and shortens the growth duration of rice. Science 2022, 377, eabi8455. [Google Scholar] [CrossRef]
- Zhang, C.; Luo, T.; Liu, J.; Xian, M.; Yuan, J.; Hu, L.; Xu, Z. Evaluation of the low-temperature tolerance of rapeseed genotypes at the germination and seedling emergence stages. Crop Sci. 2019, 59, 1709–1717. [Google Scholar] [CrossRef]
- Marjanović-Jeromela, A.; Marinković, R.; Mijić, A.; Zdunić, Z.; Ivanovska, S.; Jankulovska, M. Correlation and path analysis of quantitative traits in winter rapeseed (Brassica napus L.). Agric. Conspec. Sci. 2008, 73, 13–18. [Google Scholar]
- Armstrong, E.; Nicol, H. Reducing height and lodging in rapeseed with growth regulators. Aust. J. Exp. Agric. 1991, 31, 245–250. [Google Scholar] [CrossRef]
- Wu, W.; Ma, B.-L. Understanding the trade–off between lodging resistance and seed yield, and developing some non–destructive methods for predicting crop lodging risk in canola production. Field Crops Res. 2022, 288, 108691. [Google Scholar] [CrossRef]
- Zhang, L.; Miao, L.; He, J.; Li, H.; Li, M. The transcriptome and metabolome reveal the potential mechanism of lodging resistance in intergeneric hybrids between Brassica napus and Capsella bursa-pastoris. Int. J. Mol. Sci. 2022, 23, 4481. [Google Scholar] [CrossRef]
- Shah, L.; Yahya, M.; Shah, S.M.A.; Nadeem, M.; Ali, A.; Ali, A.; Wang, J.; Riaz, M.W.; Rehman, S.; Wu, W. Improving lodging resistance: Using wheat and rice as classical examples. Int. J. Mol. Sci. 2019, 20, 4211. [Google Scholar] [CrossRef]
- Kaur, H.; Kaur, R.; Manchanda, G.; Bindra, S.; Sharma, A. Screening of Cicer arietinum L. genotypes under combined presence of NaCl and anthracene using membership function value of stress tolerance. Plant Gene 2022, 31, 100371. [Google Scholar] [CrossRef]
- Yan, C.; Song, S.; Wang, W.; Wang, C.; Li, H.; Wang, F.; Li, S.; Sun, X. Screening diverse soybean genotypes for drought tolerance by membership function value based on multiple traits and drought-tolerant coefficient of yield. BMC Plant Biol. 2020, 20, 321. [Google Scholar] [CrossRef] [PubMed]


| Variety Name | Variety Code | Breeding Institution | Variety Name | Variety Code | Breeding Institution |
|---|---|---|---|---|---|
| Shuangyou 195 | V01 | Henan-AAS | Xiangyouza 787 | V13 | HAU |
| Shuangyou 123 | V02 | Henan-AAS | Xiangyouza 512 | V14 | HAU |
| Shuangyou 1918 | V03 | Henan-AAS | Nongxiangyou 409 | V15 | HAU |
| Shuang you 1973 | V04 | Henan-AAS | Nongxiangyou 510 | V16 | HAU |
| Fengyou 737 | V05 | Hunan-AAS | Ganyouza 8 | V17 | JX-AAS |
| Fengyou 845 | V06 | Hunan-AAS | Ganyouza 9 | V18 | JX-AAS |
| Fengyou 306 | V07 | Hunan-AAS | Ganyouza 10 | V19 | JX-AAS |
| FY509 | V08 | Hunan-AAS | Ganfengyou 3 | V20 | JX-AAS |
| Huayouza 9 | V09 | HAZU | Zhongyouza 19 | V21 | OCR-CAAS |
| Huayouza 62 | V10 | HAZU | Zhongyouza 39 | V22 | OCR-CAAS |
| Huayouza 69 | V11 | HAZU | GDP-10 | V23 | OCR-CAAS |
| H20-09 | V12 | HAZU | Dadi 199 | V24 | OCR-CAAS |
| Variety Number | Seed Yield (kg/ha) | Siliques per Plant | Seeds per Silique | 1000-Seed Weight (g) | Harvest Index | Seed-to-Pericarp Ratio | Oil Content (%) | Oleic Acid (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Y1 | Y2 | Y1 | Y2 | Y1 | Y2 | Y1 | Y2 | Y1 | Y2 | Y1 | Y2 | Y1 | Y2 | Y1 | Y2 | |
| V01 | 2628hi | 2446o | 113jk | 115j | 20.95cdefgh | 18.12hi | 3.48efg | 3.27fgh | 0.32ab | 0.27ef | 1.18ab | 0.91defg | 47.25g | 47.04hi | 68.08fghi | 69.09d |
| V02 | 2554i | 2574mn | 106k | 105l | 22.54abcd | 18.23hi | 3.49efg | 3.50bcdef | 0.32abc | 0.28def | 0.90cdef | 0.92defg | 50.43cde | 47.41gh | 68.09fghi | 70.15cd |
| V03 | 2864def | 2814ij | 131fghi | 118hij | 21.20cdefgh | 21.5bcdef | 3.40fgh | 3.29fgh | 0.30abcde | 0.31abcd | 1.05abcde | 0.99bcdef | 49.52def | 48.23gh | 69.19efgh | 69.22d |
| V04 | 2820def | 2608mn | 122hij | 117ij | 17.78i | 17.44hi | 4.80a | 4.20a | 0.27ef | 0.30abcde | 0.82efg | 1.05abcde | 48.70f | 50.84de | 67.8fghij | 71.81bcd |
| V05 | 2854def | 2745jk | 154bc | 140d | 18.70ghi | 17.02i | 3.41fgh | 3.43cdef | 0.30abcde | 0.28def | 0.90cdef | 0.90defg | 45.11j | 47.07hi | 67.9fghij | 69.76d |
| V06 | 3127a | 3283a | 156bc | 160b | 21.86cdef | 19.48efgh | 3.39fgh | 3.31efg | 0.32abcd | 0.33abc | 1.17ab | 1.11abcd | 47.24g | 48.31gh | 66.79hijk | 69.26d |
| V07 | 2937bcd | 3209b | 164b | 153c | 21.34cdefgh | 21.83bcde | 3.16hij | 3.06gh | 0.32ab | 0.31abcde | 1.02abcde | 1.05abcde | 47.06gh | 48.72fg | 71.44cde | 73.75ab |
| V08 | 3047ab | 3061cde | 144cdef | 142d | 21.62cdefg | 22.67abcd | 3.56efg | 3.34defg | 0.31abcd | 0.33abc | 1.03abcde | 1.13abc | 45.70ij | 47.81gh | 67.88fghij | 71.13bcd |
| V09 | 3106a | 3023.e | 178a | 169a | 25.16a | 20.72defg | 2.92j | 3.07gh | 0.32abc | 0.33abc | 1.12abcd | 1.08abcd | 46.67ghi | 47.61gh | 68.64fgh | 69.76d |
| V10 | 2774efg | 2907fg | 146cde | 130f | 20.97cdefgh | 20.89cdefg | 3.14hij | 3.65bc | 0.33a | 0.29cde | 1.15abc | 0.87efgh | 45.27j | 46.96hi | 65.6jk | 65.67e |
| V11 | 2859def | 2791ij | 143cdef | 124g | 21.83cdef | 21.94bcde | 3.09ij | 3.26fgh | 0.31abcd | 0.28def | 1.06abcde | 0.83fgh | 44.84j | 46.07i | 65.36k | 63.41e |
| V12 | 3133a | 3111cd | 145cdef | 130f | 21.42cdefgh | 23.21abc | 3.67ef | 3.60bcd | 0.28cde | 0.33ab | 0.95bcdef | 1.05abcde | 52.27a | 54.27a | 68.33fghi | 74.25ab |
| V13 | 2742fgh | 2583mn | 133efgh | 115jk | 24.78ab | 21.63bcde | 2.93j | 3.02h | 0.28def | 0.28def | 0.97bcdef | 0.86efgh | 49.04f | 50.81de | 65.95ijk | 69.19d |
| V14 | 2876def | 2847ghi | 135efgh | 112k | 21.59cdefg | 22.55abcd | 3.53efg | 3.53bcdef | 0.29bcde | 0.27ef | 0.87def | 0.83fgh | 50.93bc | 51.2cd | 68.13fghi | 69.99cd |
| V15 | 2581i | 2691kl | 112jk | 101m | 22.67abcd | 21.88bcde | 3.49efg | 3.41cdef | 0.25f | 0.25f | 0.74fg | 0.73fg | 47.44g | 50.21de | 67.93fghij | 70.20cd |
| V16 | 2427j | 2644lm | 152bc | 118hi | 11.54j | 18.44ghi | 3.48efg | 3.42cdef | 0.21g | 0.28def | 0.64g | 0.68g | 50.68bcd | 52.39bc | 76.63a | 75.85a |
| V17 | 2948bcd | 2934f | 124ghij | 121h | 23.85abc | 23.62ab | 3.61ef | 3.59bcde | 0.32abc | 0.33a | 1.10abcd | 1.14ab | 47.29g | 50.42de | 68.70fgh | 68.63d |
| V18 | 2813def | 2761jk | 129ghi | 113k | 20.81defgh | 21.65bcde | 3.49efg | 3.5bcdef | 0.28cde | 0.29def | 0.90cdef | 0.94cdef | 45.84hij | 49.58ef | 67.03ghijk | 69.79d |
| V19 | 2910cde | 2893fgh | 124ghij | 107l | 22.87abcd | 24.99a | 3.73de | 3.62bcd | 0.31abcd | 0.31abcd | 1.04abcde | 0.99bcdef | 48.82f | 50.34de | 71.97bc | 71.85bcd |
| V20 | 2943bcd | 2917fg | 124ghij | 117hij | 23.19abcd | 23.37ab | 3.92cd | 3.75b | 0.33a | 0.31abcd | 1.26a | 1.04abcde | 49.16ef | 50.64de | 69.76cdef | 73.25abc |
| V21 | 2663ghi | 2549n | 119ijk | 93i | 18.48hi | 23.04abcd | 3.64ef | 3.59bcde | 0.29bcde | 0.29bcde | 0.94bcdef | 0.93cdef | 50.36cde | 51.4bcd | 68.41fghi | 70.37cd |
| V22 | 2813def | 2884fgh | 129ghi | 121h | 22.21bcde | 22.49bcd | 3.30ghi | 3.28fgh | 0.31abcd | 0.30abcde | 1.04abcde | 0.94bcdef | 51.06abc | 52.57b | 71.52cd | 73.95ab |
| V23 | 3130a | 3136bc | 149cd | 133ef | 18.89fghi | 19.16fghi | 4.11bc | 4.3a | 0.32ab | 0.33ab | 1.07abcde | 1.24a | 50.43cde | 50.92de | 69.38defg | 69.95cd |
| V24 | 3022abc | 3039de | 137defj | 135e | 19.26efghi | 19.08fghi | 4.21b | 4.25a | 0.3abcde | 0.33abc | 1.00bcde | 1.05abcde | 51.86ab | 52.4bc | 73.99b | 74.37ab |
| Significant analyses | ||||||||||||||||
| Y | 0.22 NS | 94.35 ** | 0.01 NS | 1.07 NS | 0.12 NS | 0.41 NS | 109.62 ** | 46.06 ** | ||||||||
| V | 60.88 ** | 35.36 ** | 12.05 ** | 25.47 ** | 7.71 ** | 3.15 ** | 50.60 ** | 17.83 ** | ||||||||
| Y × V | 5.47 ** | 2.46 ** | 4.09 ** | 1.80 * | 3.04 ** | 1.58 NS | 5.60 ** | 2.07 ** | ||||||||
| Index | 2020–2021 | 2021–2022 | ||||
|---|---|---|---|---|---|---|
| Standard Deviation | Information Content | Objective Weight | Standard Deviation | Information Content | Objective Weight | |
| Seeds yield | 0.271 | 2.601 | 18.54% | 0.267 | 2.518 | 20.34% |
| Growth period | 0.273 | 2.708 | 19.45% | 0.317 | 2.280 | 21.86% |
| Lodging angle | 0.318 | 2.647 | 22.09% | 0.282 | 2.250 | 19.20% |
| Plant architecture | 0.282 | 2.692 | 19.96% | 0.254 | 2.523 | 19.39% |
| Seed quality | 0.274 | 2.773 | 19.96% | 0.242 | 2.623 | 19.21% |
| Variety Number | 2020–2021 | 2021–2022 | Averaged Comprehensive Score | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Seed Yield | Calendar Days | Lodging Angle | Plant Architecture | Seed Quality | Comprehensive Score | Seed Yield | Calendar Days | Lodging Angle | Plant Architecture | Seed Quality | Comprehensive Score | ||
| V01 | 0.28 | 0.38 | 0.93 | 0.82 | 0.33 | 0.56 | 0.00 | 0.09 | 0.86 | 0.77 | 0.29 | 0.39 | 0.48 |
| V02 | 0.18 | 0.38 | 0.53 | 0.01 | 0.61 | 0.35 | 0.15 | 0.36 | 0.41 | 0.00 | 0.36 | 0.26 | 0.30 |
| V03 | 0.62 | 0.13 | 0.98 | 0.64 | 0.57 | 0.60 | 0.44 | 0.09 | 0.94 | 0.58 | 0.38 | 0.48 | 0.54 |
| V04 | 0.56 | 1.00 | 0.14 | 0.05 | 0.44 | 0.43 | 0.19 | 1.00 | 0.45 | 0.15 | 0.66 | 0.50 | 0.46 |
| V05 | 0.60 | 0.63 | 0.22 | 0.14 | 0.13 | 0.34 | 0.36 | 0.45 | 0.47 | 0.16 | 0.32 | 0.35 | 0.35 |
| V06 | 0.99 | 0.63 | 0.23 | 0.19 | 0.27 | 0.45 | 1.00 | 0.36 | 0.52 | 0.27 | 0.39 | 0.51 | 0.48 |
| V07 | 0.72 | 0.63 | 0.34 | 0.16 | 0.46 | 0.45 | 0.91 | 0.45 | 0.53 | 0.26 | 0.59 | 0.55 | 0.50 |
| V08 | 0.88 | 0.63 | 0.42 | 0.64 | 0.18 | 0.54 | 0.73 | 0.36 | 0.63 | 0.51 | 0.42 | 0.53 | 0.54 |
| V09 | 0.96 | 0.63 | 0.64 | 0.15 | 0.30 | 0.53 | 0.69 | 0.45 | 0.85 | 0.30 | 0.36 | 0.53 | 0.53 |
| V10 | 0.49 | 0.38 | 0.72 | 0.19 | 0.05 | 0.37 | 0.55 | 0.27 | 0.85 | 0.26 | 0.15 | 0.41 | 0.39 |
| V11 | 0.61 | 0.38 | 0.77 | 0.54 | 0.00 | 0.46 | 0.41 | 0.27 | 0.74 | 0.38 | 0.00 | 0.36 | 0.41 |
| V12 | 1.00 | 0.38 | 0.82 | 0.19 | 0.78 | 0.63 | 0.79 | 0.45 | 0.91 | 0.18 | 1.00 | 0.66 | 0.65 |
| V13 | 0.45 | 0.00 | 0.21 | 0.48 | 0.39 | 0.30 | 0.16 | 0.00 | 0.23 | 0.44 | 0.56 | 0.27 | 0.29 |
| V14 | 0.64 | 0.63 | 0.05 | 0.67 | 0.65 | 0.51 | 0.48 | 0.09 | 0.15 | 0.51 | 0.62 | 0.36 | 0.44 |
| V15 | 0.22 | 0.38 | 0.00 | 0.31 | 0.34 | 0.24 | 0.29 | 0.09 | 0.00 | 0.41 | 0.56 | 0.27 | 0.25 |
| V16 | 0.00 | 0.13 | 0.28 | 0.00 | 1.00 | 0.29 | 0.24 | 0.09 | 0.31 | 0.03 | 0.93 | 0.31 | 0.30 |
| V17 | 0.74 | 0.88 | 0.76 | 0.45 | 0.36 | 0.64 | 0.58 | 0.82 | 0.78 | 0.29 | 0.51 | 0.60 | 0.62 |
| V18 | 0.55 | 0.63 | 0.32 | 0.36 | 0.16 | 0.40 | 0.38 | 0.36 | 0.34 | 0.25 | 0.50 | 0.36 | 0.38 |
| V19 | 0.69 | 0.88 | 0.59 | 0.42 | 0.63 | 0.64 | 0.53 | 0.64 | 0.63 | 0.57 | 0.63 | 0.60 | 0.62 |
| V20 | 0.73 | 0.88 | 0.48 | 0.17 | 0.57 | 0.56 | 0.56 | 1.00 | 0.63 | 0.22 | 0.71 | 0.63 | 0.60 |
| V21 | 0.33 | 0.63 | 0.93 | 0.90 | 0.61 | 0.69 | 0.12 | 0.73 | 0.90 | 0.92 | 0.65 | 0.66 | 0.67 |
| V22 | 0.55 | 0.88 | 1.00 | 0.38 | 0.81 | 0.73 | 0.52 | 0.73 | 1.00 | 0.34 | 0.87 | 0.69 | 0.71 |
| V23 | 1.00 | 0.63 | 0.88 | 0.52 | 0.66 | 0.74 | 0.82 | 1.00 | 0.91 | 0.59 | 0.60 | 0.79 | 0.76 |
| V24 | 0.84 | 1.00 | 0.84 | 1.00 | 0.99 | 0.93 | 0.71 | 0.82 | 0.90 | 1.00 | 0.87 | 0.86 | 0.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Luo, T.; Cheng, T.; Yang, S.; She, H.; Li, J.; Wang, B.; Kuai, J.; Wang, J.; Xu, Z.; et al. Evaluation and Screening of Rapeseed Varieties (Brassica napus L.) Suitable for Mechanized Harvesting with High Yield and Quality. Agronomy 2023, 13, 795. https://doi.org/10.3390/agronomy13030795
Li Q, Luo T, Cheng T, Yang S, She H, Li J, Wang B, Kuai J, Wang J, Xu Z, et al. Evaluation and Screening of Rapeseed Varieties (Brassica napus L.) Suitable for Mechanized Harvesting with High Yield and Quality. Agronomy. 2023; 13(3):795. https://doi.org/10.3390/agronomy13030795
Chicago/Turabian StyleLi, Qin, Tao Luo, Tai Cheng, Shuting Yang, Huijie She, Jun Li, Bo Wang, Jie Kuai, Jing Wang, Zhenghua Xu, and et al. 2023. "Evaluation and Screening of Rapeseed Varieties (Brassica napus L.) Suitable for Mechanized Harvesting with High Yield and Quality" Agronomy 13, no. 3: 795. https://doi.org/10.3390/agronomy13030795
APA StyleLi, Q., Luo, T., Cheng, T., Yang, S., She, H., Li, J., Wang, B., Kuai, J., Wang, J., Xu, Z., & Zhou, G. (2023). Evaluation and Screening of Rapeseed Varieties (Brassica napus L.) Suitable for Mechanized Harvesting with High Yield and Quality. Agronomy, 13(3), 795. https://doi.org/10.3390/agronomy13030795










