2.3.1. Setting-Up of Micro-Kinetic Models
The temperature programmed desorption of CO from Fe(100) was modeled using two mean-field micro-kinetic models, viz. Bragg–Williams approximation (BWA) and the quasi-chemical approach (QCA), and a kinetic Monte Carlo (kMC) method [
34]. The mean-field approach presumes a desorption process as a result of a single averaged effect. In the Bragg–Williams approximation (BWA) the likelihood for a certain species at an adjacent site is given by the fractional coverage of that species. This kind of model may work well for systems with low or negligible mobility of the adsorbed species (either through diffusion or via desorption and re-adsorption) resulting in a ‘frozen’ surface structure since surface species cannot diffuse to an energetically more favorable site. In the quasi-chemical approach (QCA), the likelihood for a certain species at an adjacent site is dependent on the interaction energy between the species at the central site and the species at the neighboring sites. This type of models may work sufficiently well in systems with high mobility of surface species. The more complex kinetic Monte Carlo (kMC) model can capture subtleties in the catalytic process better, e.g., induced effects by finite rates of diffusion.
Essentially only three reaction steps need to be considered when describing temperature programmed desorption of CO taking place in a vacuum, i.e.,
In addition to these reaction steps, diffusion of the various surface species is considered in the kMC-model:
The rate constants for these steps are given by the transition state theory. A thermodynamic consistent expression for the rate constant for CO desorption is obtained from the equilibrium constant for adsorption of CO and the rate constant for the adsorption of CO. The latter is given by the impingement and sticking of CO on to a surface site with the area, A
site. Assuming a sticking probability of one for the adsorption of CO, the rate constant for desorption of CO is then given by:
where Q
CO(g) and Q
CO* are partition functions for CO in the gas phase and adsorbed state, respectively, taking into account the vibrational and for CO in the gas phase also the translational and rotational partition function and E
des,CO(θ) is the coverage dependent desorption energy for CO. The rate constants for the surface reactions and diffusion steps are given by
with
and
are partition functions for the transition state and initial state, respectively, only considering the vibrational contribution while h is Planck’s constant and E
a(θ) the coverage dependent activation energy. Hence, the pre-exponential factors in all our models are dependent on temperature. It should be noted that in principle lateral interactions also affect the pre-exponential factor due to a change in the vibrational partition function, but this can be neglected seeing the relatively small change in the partition function.
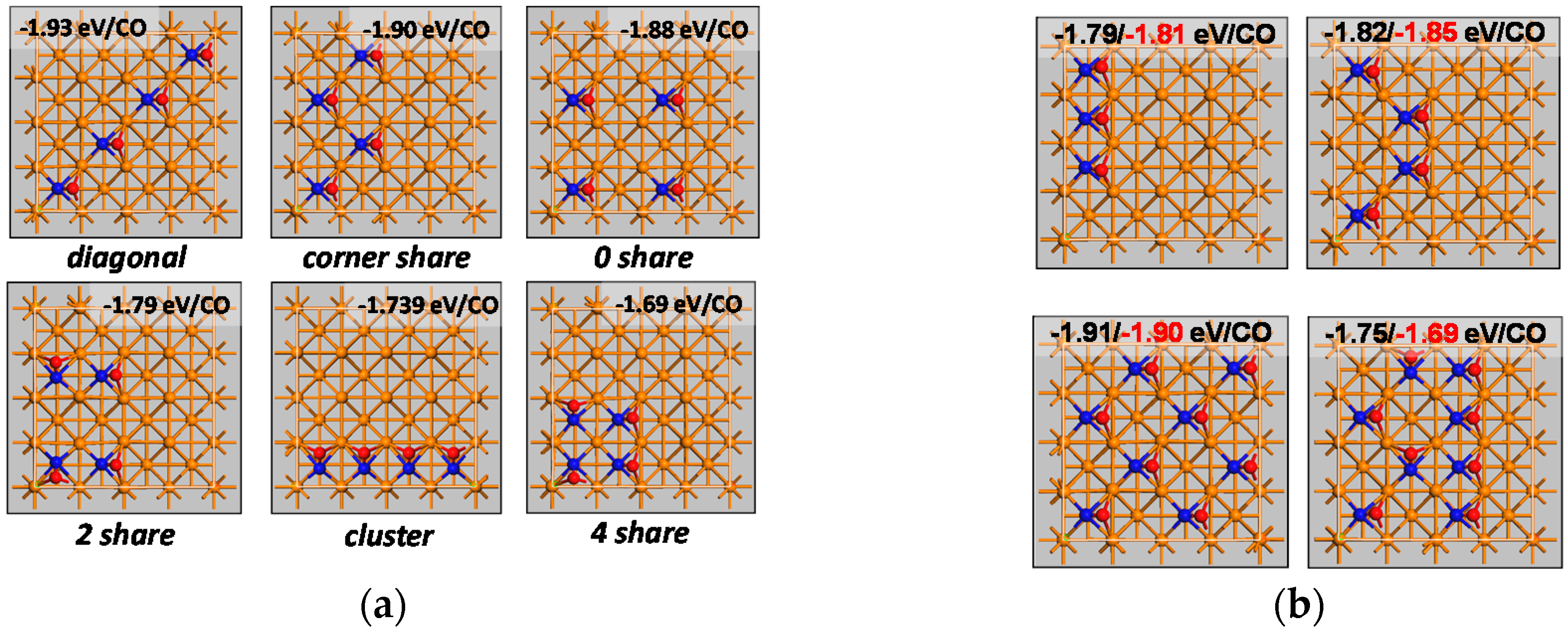
The coverage dependent reaction energies can be obtained from the energy without lateral interaction (here taken as the reaction energy obtained from DFT at a coverage of 0.25 ML—see
Table 3) and the interaction energy, ε
i-j (see
Table 4). The interaction energy is dependent on the orientation of the species involved and their environment. This complex relationship may be simplified by considering only the interaction energy obtained at a coverage of 0.5 ML. This may be a suitable simplification for the description of the CO-desorption process if interactions are repulsive and diffusion of the surface species is sufficiently high resulting in a low probability of nearest neighbor interactions at low coverage. Under these assumptions, the interaction energy for the nearest neighbor interaction is obtained from the excess energy obtained in the co-adsorption of CO-CO, CO-C, CO-O, and C-O at 0.5 ML in the p(2x2) unit cell with the adsorbates in adjacent positions. The interaction energy for the next-nearest neighbor interaction is obtained from the excess energy in the co-adsorbed systems with the adsorbates diagonally orientated.
The interaction energies, εi-j, are in principle dependent on temperature. The temperature dependency of CO-CO and the C-O interaction may be neglected (vide supra). During CO-TPD, the system is expected to have an initially a high coverage with CO and relatively low coverage at high temperature. Hence, the temperature dependency is not incorporated.
The coverage dependent activation barriers can be obtained by invoking the Brønsted−Evans−Polanyi correlation and Hammond’s postulate [
35], i.e., implying that the curvature of the potential energy surface does not change significantly due to lateral interactions [
36]:
with
and
the activation barrier and the reaction energy respectively in the absence of lateral interactions,
the reaction energy in the presence of lateral interactions. Hence, it is assumed that the change in the activation energy due to lateral interactions is proportional to the change in the reaction energy due to lateral interactions.
The factor α is in the limit case 0 for reactions with an early transition state, such as CO dissociation, and 1 for reactions with a late transition state, such as C/O recombination. The factor α is taken to be 0.5 for the diffusion of CO, C, and O.
The coverage dependent change in energy upon reaction is given by the difference in the energy of the product and reacting species involved in the reaction taking into account their interaction with neighboring species, and thus the difference in the cluster energy of the product and the reacting species. According to the Bragg–Williams approximation (BWA) the coverage dependent cluster energy for a system containing m species is determined from the energy of the species without lateral interactions (
) and the interaction of a central adsorbate i with the nearest neighbor j (
) and its interaction with the next-nearest neighbor k (
):
with n
NN the number of nearest neighboring sites (n
NN = 4 on Fe(100)) and n
NNN the number of next-nearest neighboring sites (n
NNN = 4 on Fe(100)).
In the quasi-chemical approach (QCA) the coverage dependent cluster energy for a system containing m species is given by [
37]:
where
and
, are fractional coverage of species-
and the total number of surface species, respectively.
is the probability that the neighboring site is occupied by species-
given that the central site is occupied by species
. This probability is dependent on the probability of finding a pair of sites occupied by species
and species
. A detailed description to calculate these probability functions is given by Hellman and Honkala [
37] based on the work by Zhdanov [
3].
The determination of the CO-TPD-profile using mean-field models involves solving a set of three ODEs. In contrast, the kinetic Monte Carlo (kMC) algorithms identify all possible sets of events which may occur within a given system state, <σ>. An event, reaction step, is chosen depending on its kinetic rate constant and its multiplicity—faster reactions are chosen more frequently. The algorithm then evolves from a given initial state, <σ
0>. In this study, we used the
Zacros, version 2.0, software package [
38,
39,
40,
41] which is based on the graph-theoretical kinetic Monte Carlo framework [
39]. Detailed descriptions of this package and its implementation can be found elsewhere [
40,
41]. Briefly, the total lattice energy Hamiltonian, H(σ), is calculated using the cluster expansion approach [
40]:
where
describes the occupancy of each lattice site, N
c is the number of clusters considered to account for lateral interaction effects on the total lattice energy. The summation is done over all
clusters where ECI
k, GM
k and NCE
k are the effective cluster energy, cluster multiplicity and the number of occurrences of cluster k within a given lattice state, <σ> [
42]. The cluster energy, ECI
k, is obtained from the energy of the central adsorbate (see
Table 3) taking lateral interactions with the adjacent species into account (see
Table 4). The cluster multiplicity, GM
k, the number of occurrences of cluster k are determined for a given lattice state using a detection algorithm [
40].
2.3.2. Describing CO-TPD Using Mean Field Models
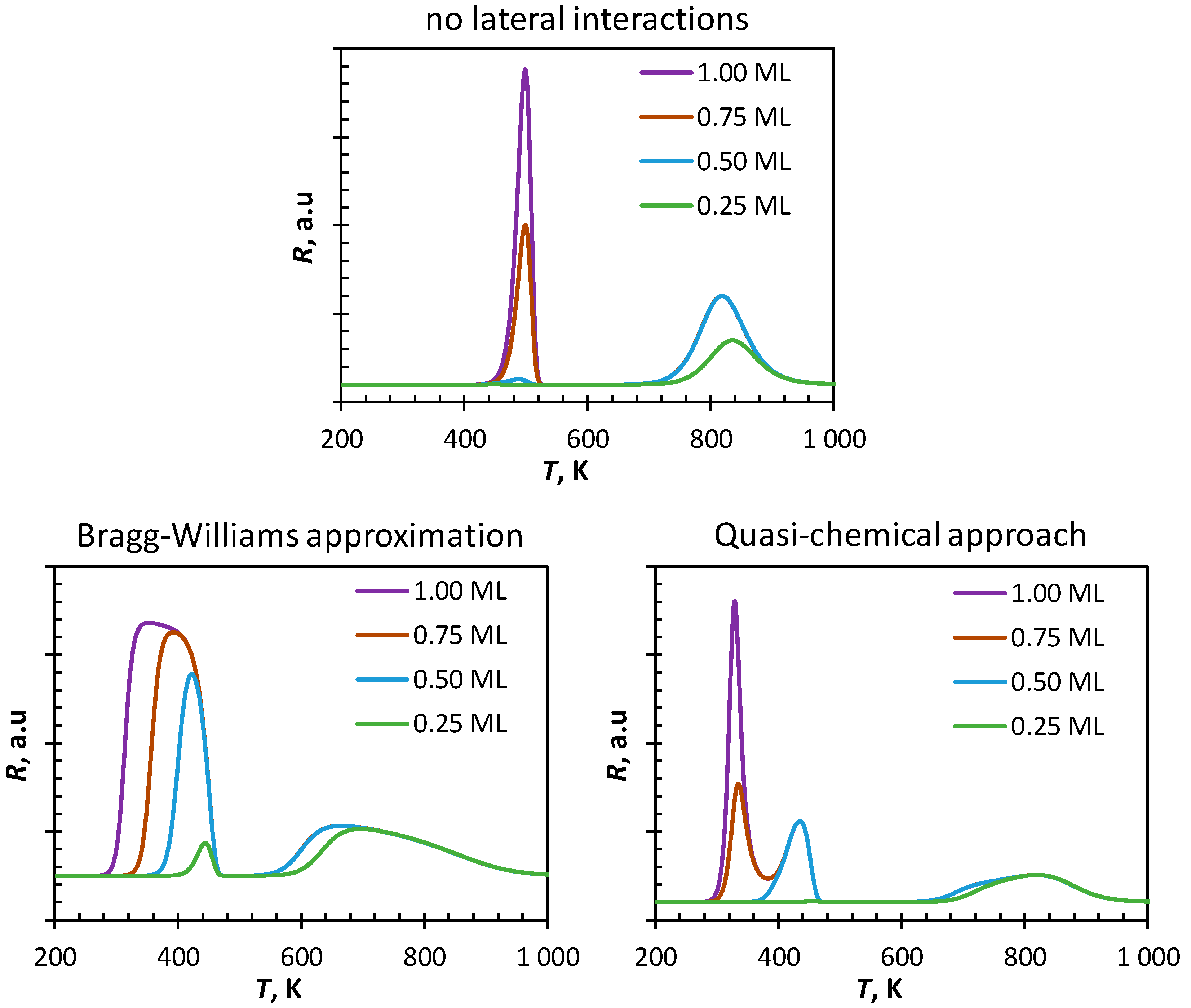
Figure 2 shows simulated spectra of temperature programmed desorption of CO from Fe(100) obtained from different starting coverages of CO using a mean-field approach. In the absence of lateral interactions, the TPD-profile starting with an initially saturated surface is characterized by two peaks with a peak maximum at 500 K and 810 K, respectively. These peaks can be attributed to the desorption of molecularly adsorbed CO and the desorption of CO after re-combination of surface carbon and surface oxygen, which have been formed upon dissociation of adsorbed CO. Decreasing the initial coverage of the surface to Θ
CO,initial = 0.75 ML decreases the magnitude of the first peak, but not its position. It can be further noted that the maximum in the high temperature region remains unchanged in both its magnitude and its position. The first peak almost completely disappears upon decreasing the initial coverage further to Θ
CO,initial = 0.50 ML and the second peak remains again unaltered. This can be rationalized since the rate of CO-dissociation at high coverage is limited by the availability of vacant sites. At sufficiently low initial coverage, CO will dissociate before it desorbs as the activation barrier for CO dissociation is lower than the activation barrier for CO desorption. Dissociation of CO for experiments started with an initial coverage of Θ
CO,initial > 0.50 ML will result in a surface almost saturated with atomic carbon and atomic oxygen. Decreasing the initial coverage to Θ
CO,initial = 0.25 ML results in a small shift of the high temperature peak to ca. 830 K. Starting with a low initial coverage of CO results in only a partial coverage of the surface with carbon and oxygen, thus limiting the rate of recombination and thus the desorption of CO from the surface.
The simulated TPD-spectra of CO from Fe(100) using the Bragg–Williams approximation also shows in principle two maxima. Starting from ΘCO,initial = 1.00 ML the on-set of the first peak is estimated to be at ca. 300 K showing the highest rate of desorption at 360 K. Decreasing the initial CO-coverage to ΘCO,initial = 0.75 ML and to ΘCO,initial = 0.50 ML increases the onset temperature for the first peak to 330 K and 370 K, respectively. The maximum rate of desorption is then obtained at 403 K and 416 K, respectively. The occurrence of CO-desorption at significantly lower temperatures predicted by the Bragg–Williams approximation in comparison to the simulated CO-TPD spectra in the absence of lateral interactions is attributed to the repulsive nearest-neighbor interactions in this system, and in particular to the repulsive nature of the nearest neighbor CO-CO interaction. The increased broadness of the peak is attributed to the gradual change in the strength of adsorption due to the change in the surface coverage. It can be further noted that the tail of the first peak coincides for the experiments with an initial coverage of CO of 1.00 ML, 0.75 ML, and 0.50 ML. At these temperatures, the coverage of the surface is independent of the initial coverage with CO, and hence the desorption profiles become indistinguishable from this temperature onwards.
In contrast to the simulated CO-TPD profile with ΘCO,initial = 0.25 ML in the absence of lateral interactions, the same system simulated using the Bragg–Williams approximation does show a low temperature peak with a peak maximum at ca. 450 K. CO-dissociation taking place at these temperatures result in a higher surface coverage (with atomic carbon and atomic oxygen) thereby increasing lateral interactions thus favoring desorption of some CO from the surface.
The high temperature peak in the simulated CO-TPD profile using the Bragg–Williams approximation is strongly shifted towards lower temperatures in comparison to the simulated CO-TPD profile in the absence of lateral interactions. Strong repulsive nearest-neighbor interactions between in particular C and O will favor the recombination of C and O, a reaction with a late transition state, resulting in the formation and desorption of CO at relatively low temperatures. Reduction of the lateral interactions, through desorption of CO, will thus reduce the rate of C-O recombination and slow down the rate of CO-desorption. The second peak maximum in the simulated CO-TPD profile for the experiment with ΘCO,initial = 0.25 ML is again shifted slightly to higher temperatures. This can be attributed to the reduced likelihood of nearest neighbor interactions in this system due to the lower surface coverage. This was confirmed by reducing the initial coverage to 0.05 ML (not shown), which results in peak maximum at high temperature close to the one obtained ignoring lateral interactions.
The simulated spectra of CO-TPD from Fe(100) starting from an initially saturated surface using the quasi-chemical approach (QCA) shows three maxima at 327 K, 440 K and a broad maximum around 820 K. The appearance of two separate peaks at low temperature rather than a broad peak as observed in the simulated CO-TPD using the Bragg–Williams approximation, can be attributed to the instantaneous rearrangement of the surface upon generation of vacancies on the surface to the lowest energy state minimizing strongly repulsive nearest neighbor interactions. Decreasing the initial coverage to ΘCO,initial = 0.75 ML results in a strong decrease of the first peak (as well as a slight shift in the peak maximum to 338 K) as the number of nearest neighbor interactions would have been lower from the start of the experiment. Decreasing the initial coverage further to ΘCO,initial = 0.50 ML results in the disappearance of the low temperature peak. Only a rather small amount of CO desorbs at a low temperature (440 K) if the initial coverage is lowered to ΘCO,initial = 0.25 ML.
The observed new low temperature peak in addition to the 2
nd low temperature peak in the simulated CO-TPD using the quasi-chemical method starting with an initially saturated surface compare well with the appearance of a new peak with a maximum at 306 K (α
2-CO) in addition to a peak at 440 K (α
3-CO) in the CO-TPD from Fe(100) with increasing dosing of CO [
18]. The tentative assignment of α
2-CO in CO-TPD from Fe(100) to CO bonded to a bridge site [
19] may rather be related to lateral interactions in the system.
The high temperature peak in the simulated CO-TPD using the quasi-chemical method is broad, and its maximum is shifted significantly towards higher temperatures in comparison to the simulated TPD using the Bragg–Williams approximation. Strong repulsive interactions can be minimized in systems with sufficient vacant sites, thus minimizing the rate of desorption from the surface.
The input parameters in the kinetic models are associated with the uncertainty inherent to the method of determining these factors.
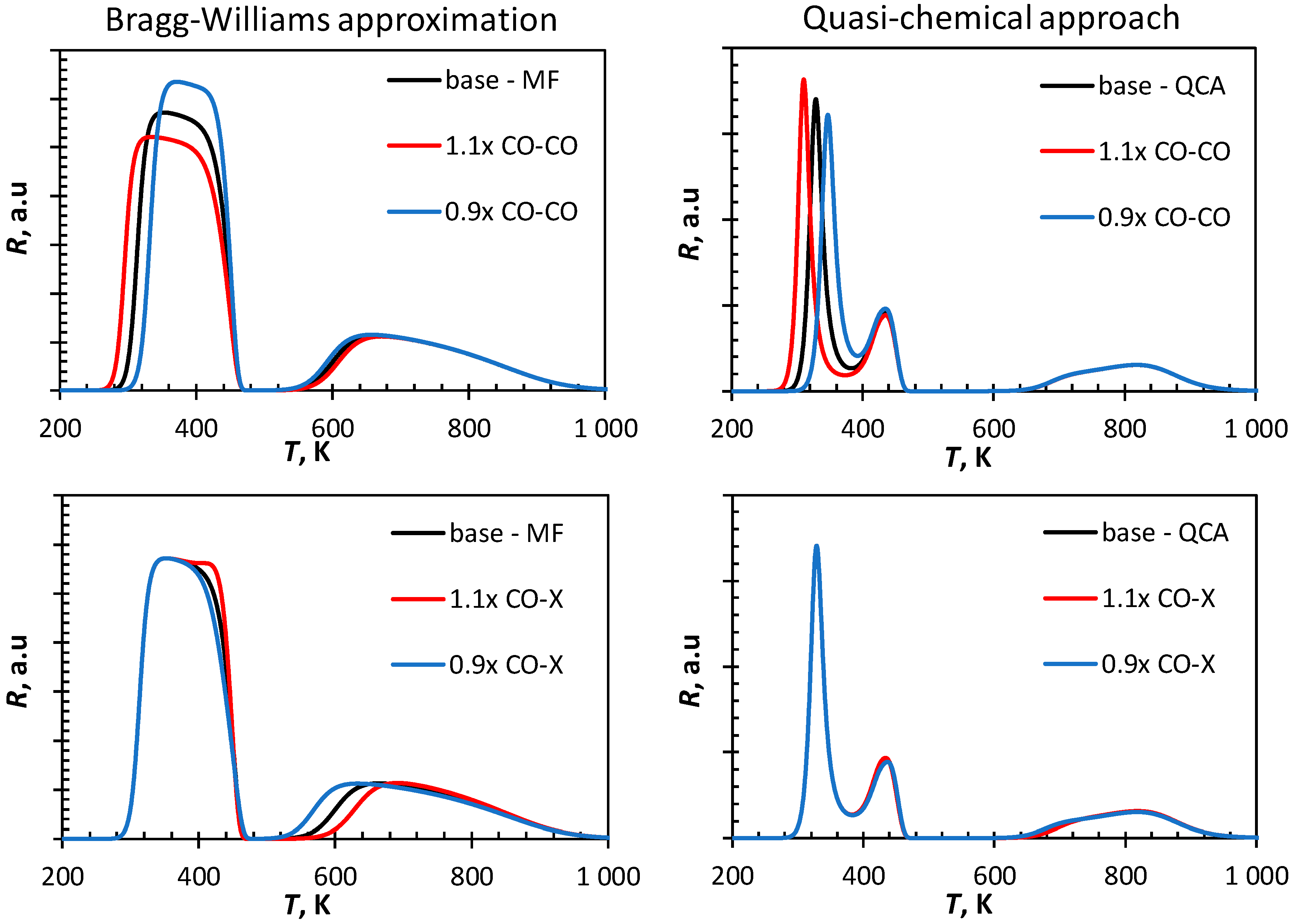
Figure 3 shows the influence of the variation of the strength of the nearest neighbor interaction on the simulated CO-TPD-profile. Increasing the strength of the lateral interactions between CO and CO by 10% shifts the broad, low temperature peak obtained using the Bragg–Williams approach to lower temperatures, but the essential feature in this profile, i.e., the broad single peak at a low temperature remains. It can be further noted that the tail of the first peak is hardly affected. A change in the CO-CO lateral interaction shifts only the low temperature peak in the CO-TPD profile simulated using the quasi-chemical approach, but not the peak with a maximum at ca. 440 K. This implies that at the adsorption of CO, which desorbs at this temperature is no longer affected by lateral interactions between adsorbed CO and CO on adjacent sites.
The low temperature region of the CO-TPD simulated using the Bragg–Williams approximation is only marginally affected by a change in the lateral interactions between CO and C and the lateral interactions between CO and O. Increasing the lateral interactions forces the desorption of some more CO at lower temperatures starting to result in the formation of a new maximum in the rate of desorption at ca. 440 K. The recombination of C and O is affected by the lateral interaction as indicated by a shift in the peak maximum at high temperature region towards higher temperatures with increasing strength of CO-X lateral interactions. This is not seen in the CO-TPD simulated using the quasi-chemical approach as these lateral interactions are minimized when applying this framework.
The CO-TPD profiles were simulated assuming an early transition state for CO dissociation. A shift in the transition state from a state resembling more the initial state to a state resembling more the product state will affect the energy barrier for CO-dissociation through a change in the difference in the reaction energy incorporating lateral interactions and the reaction energy in the absence of lateral interactions (see Equation (9)). This difference in the reaction energy for CO-dissociation becomes higher if the extent of CO dissociation increases whilst keeping the fraction of surface which is vacant constant, and upon decreasing the fraction of the surface which is vacant keeping the ratio of CO adsorbed to C/O adsorbed constant. Hence, the activation energy for CO dissociation will become higher for a late transition state. Similar reasoning can be put forward for the reverse reaction taking into account that the difference in the reaction energy incorporating lateral interactions is less than the reaction energy in the absence of lateral interactions. Hence, the activation energy for C/O recombination is lower with an early transition state than with a late transition state.
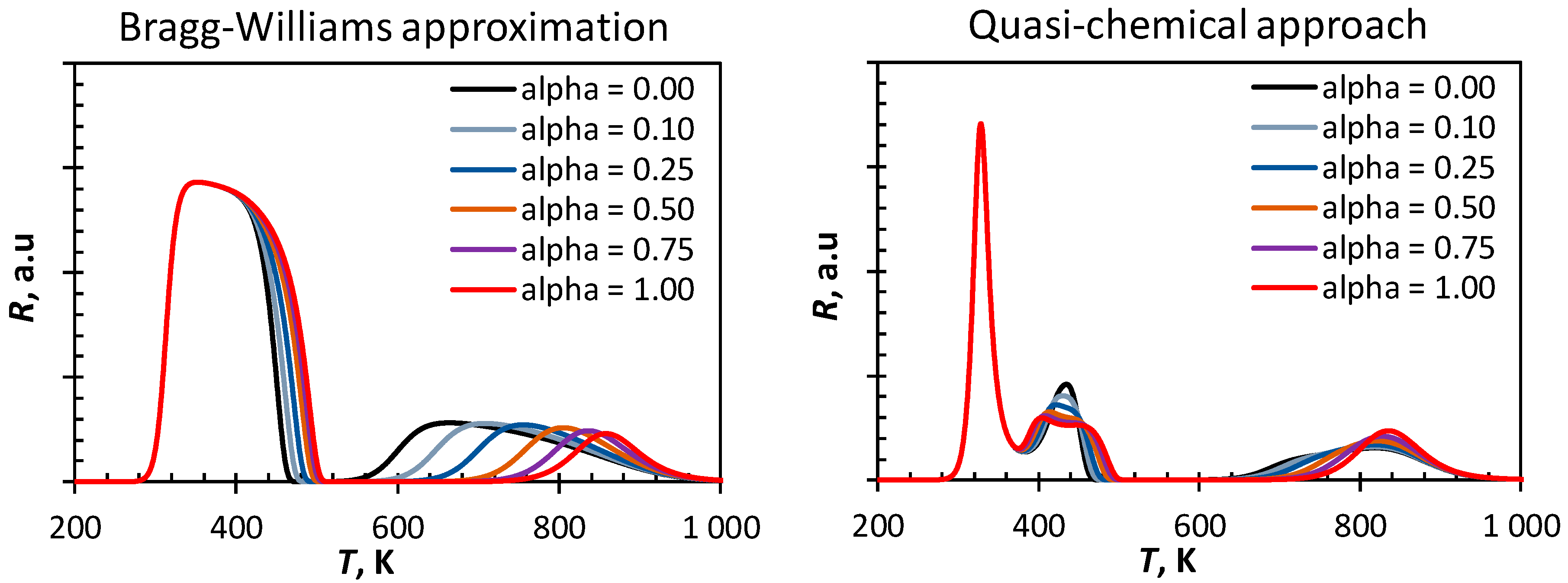
The tail of the first peak in the CO-TPD profile simulated using the Bragg–Williams approximation shifts towards higher temperatures upon changing from an early to a late transition state (see
Figure 4). This can be attributed to an increase in the activation energy for CO-dissociation upon increasing the α value. The increase in the activation energy for CO-dissociation also results in a decrease of the surface coverage with C and O, e.g., at temperatures around 500 K. A higher coverage with atomic C and O on the surface will according to the Bragg–Williams approximation result in higher repulsive lateral interactions between the adsorbed species and thus favoring the C/O recombination. Hence, the high temperature peak (β) shifts towards lower temperatures upon decreasing α.
The observed variations in the CO-TPD profiles simulated using the quasi-chemical approach upon changing the transition state from an early transition state to a late transition state are much subtler than in the same variation in the CO-TPD profile simulated using the Bragg–Williams approximation. The first peak is not affected by changing the assumption on the transition state for CO-dissociation. Interestingly, the second peak with a maximum at ca. 440 K becomes broader and start showing features of two separate events assuming a late transition state. Increasing the value of α, whilst keeping the fraction of vacant sites approximately constant, will increase the reaction barrier for CO dissociation. As a consequence, the fractional coverage of the surface with CO at 400 K is slightly higher if CO dissociation has a late transition state, which thus yields a slightly higher rate of desorption of CO at this temperature. The delay in the build-up of appreciable amounts of carbon and oxygen on the surface further implies that the influence of those lateral interactions on the low temperature peak will be shifted towards slightly higher temperatures.
The high temperature peak in the CO-TPD profile becomes sharper upon changing from an early to a late transition state. Increasing the value of α will result in a shift from an early transition state to a later transition state increasing the energy barrier for C/O recombination.
2.3.3. Describing CO-TPD Using Kinetic Monte-Carlo (kMC)
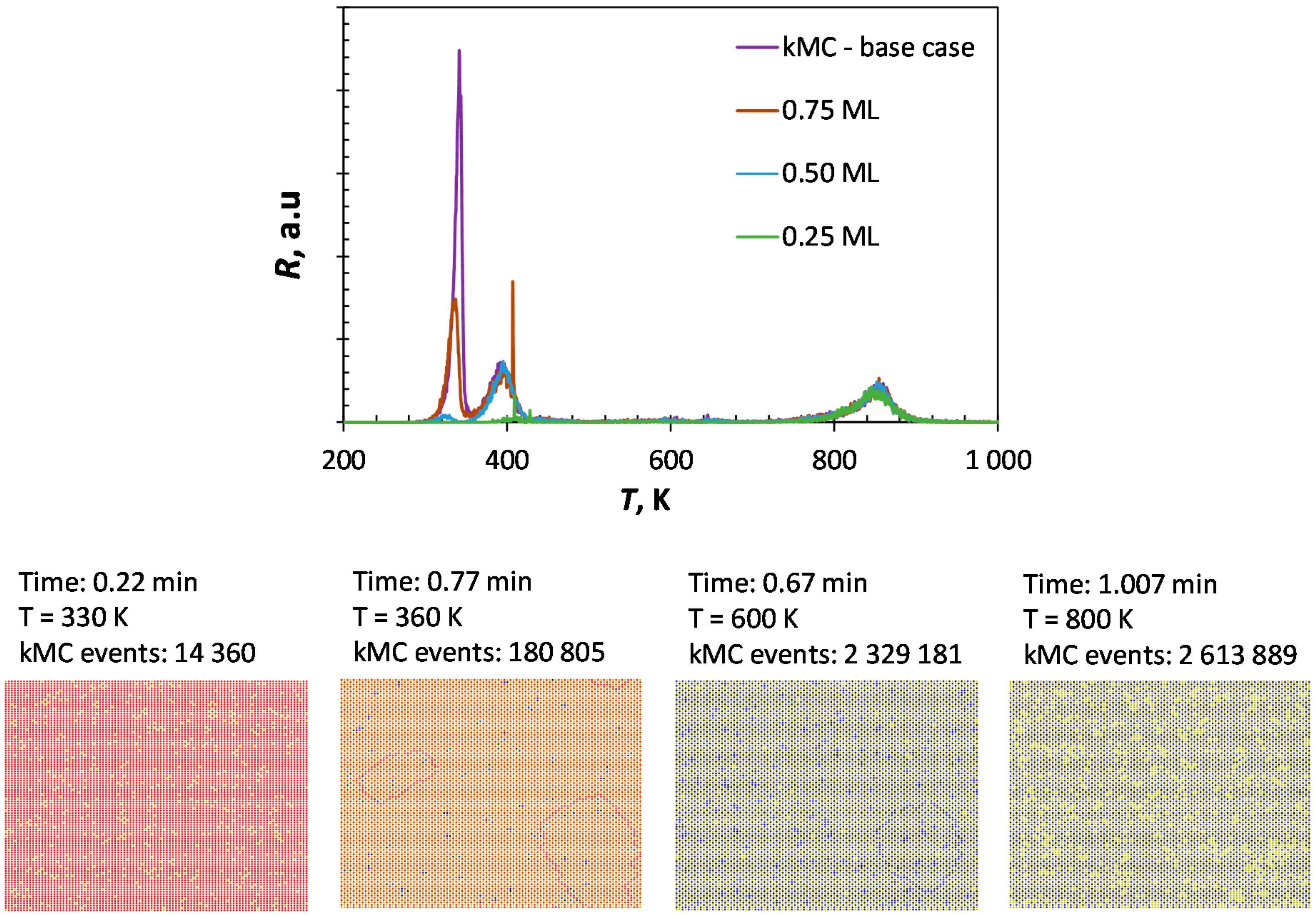
The applied kinetic Monte Carlo explicitly considers diffusion of the species on the surface and as a consequence the local structure. The simulated TPD-profile of CO from Fe(100) using kinetic Monte Carlo (see
Figure 5) shows similarity to the profile generated using a mean-field quasi-chemical approach (cf.
Figure 3) with three main desorption maxima. However, there are some differences between the two CO-TPD profiles. The maximum of the first peak is shifted towards slightly higher temperatures at high coverage (from 320 K to 330 K). This might be attributed to the local local stabilization accounted for in the kinetic Monte Carlo simulation through a diffusional shift of adsorbed CO. The second peak maximum is shifted to a significantly lower desorption temperature (from 430 K to 390 K). At this temperature, CO starts to dissociate. The slow diffusion of C/O from adjacent CO will increase the rate of desorption of CO.
A particular feature of kinetic Monte Carlo is the inclusion of local effects rather than global averaging effect. It can be noted that upon dissociation (and given sufficient time), the system seems to prefer C-C and O-O neighboring couples rather than C-O as nearest neighbors (as could be deduced the data given in
Table 4). Thus, CO desorption at temperatures higher than 700 K thus requires first diffusion of the reactants to adjacent positions. This slows down the desorption of dissociated CO and the maximum in the rate of desorption is seen to be shifted towards higher temperatures in the kinetic Monte Carlo simulation compared to the simulation using the mean-field, quasi-chemical approach.
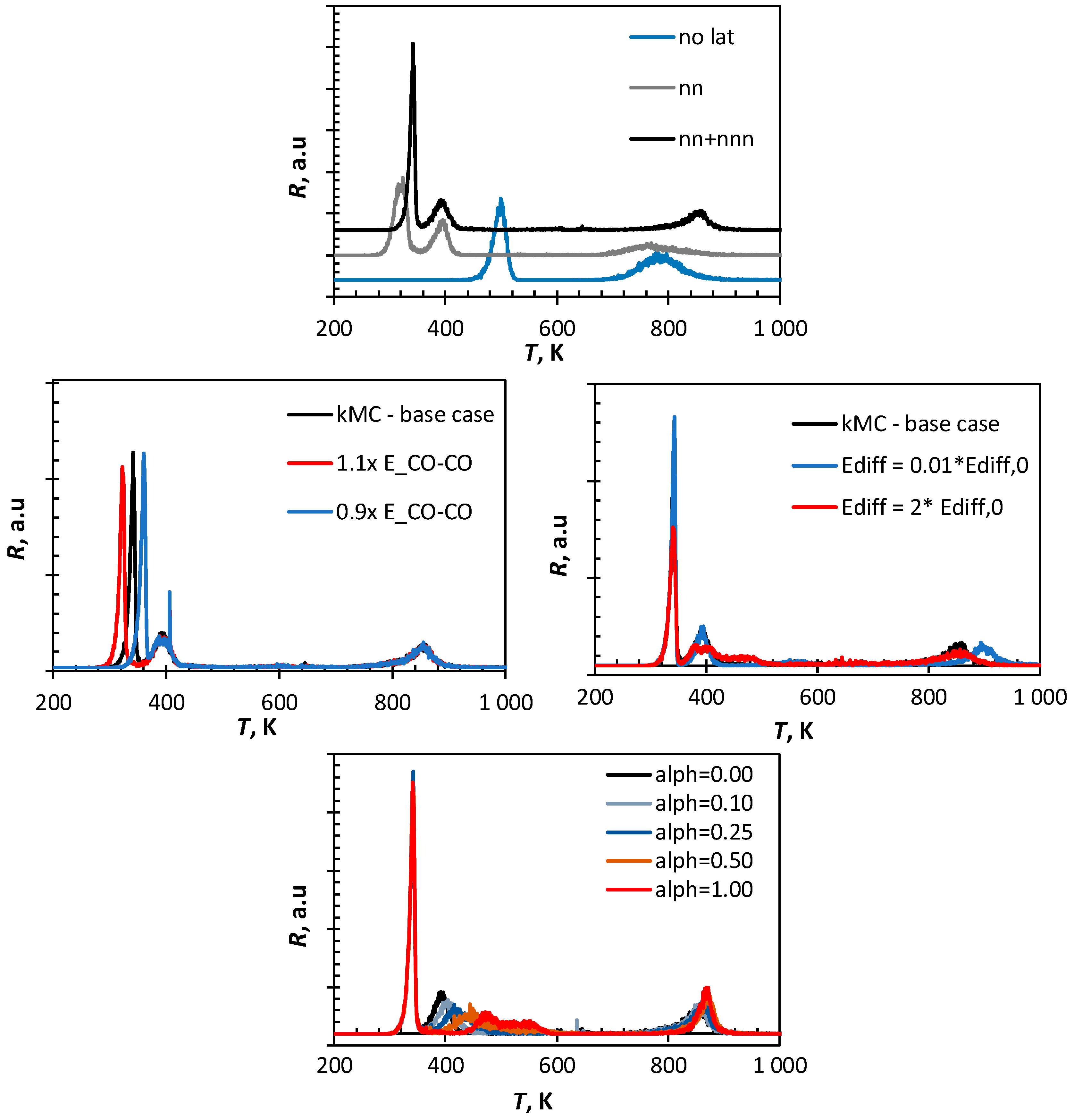
Figure 6 shows the sensitivity of the simulated CO-TPD profile using the kinetic Monte Carlo method on the choice of the various parameters. The absence of lateral interactions results in a CO-TPD profile with only two maxima. This profile is similar to the one calculated using a mean-field approach, although the high temperature peak (β-CO) is shifted to a slightly lower temperature when using kMC. This can be attributed to a dilution effect. In the mean-field approach, the average fractional coverage of C and O is taken into account to estimate the rate for the C/O recombination, whereas kMC takes local coverages into account. The incorporation of nearest neighbor lateral interactions results in the low temperature region of the CO-TPD profile in a low temperature peak, as a result of CO desorption experiencing strong lateral interaction with co-adsorbed CO, and a peak with a maximum at ca. 405 K, due to the desorption of more strongly bound CO. The low temperature peak in the CO-TPD profile shifts towards higher temperatures upon incorporation of the slightly attractive, next-nearest neighbor interactions. The high temperature peak (β-CO) is also affected by the inclusion of lateral interactions. The inclusion of the repulsive, nearest-neighbor interactions results in the CO-desorption already at significantly lower temperatures: the repulsive interaction forces the C/O recombination. This is countered by the attractive next-nearest neighbor interactions.
Increasing the lateral interaction by 10% results in a shift in the low temperature peak in the CO-TPD with 19 K towards lower temperature. Similarly, a decrease in energy associated with CO-CO interaction causes the low temperature peak to shift to a higher temperature by ca. 19 K. A similar effect was observed in the sensitivity analysis using the quasi-chemical approach (QCA), but the position of the peak maximum was ca. 10 K higher in the kinetic Monte Carlo simulation in comparison to the simulation using the quasi-chemical approach (QCA). This is a result of the finite rate of diffusion of the species in the Monte Carlo simulation compared to the instantaneous equilibration of the lateral interactions on the surface assumed in the QCA-simulation.
The CO-TPD profile simulated using kinetic Monte Carlo was virtually unaffected by the strength of the lateral interactions between CO and C and CO and O (as also noted in the CO-TPD profile simulated using the quasi-chemical approach). The diffusion of the various species is fast enough at the temperatures where these interactions start to play a role to ensure that the repulsive interactions can be minimized.
The energy barrier for the diffusion over the surface of the various species does not affect the first peak in the TPD profile of CO from Fe(100). Also, the second peak in the CO-TPD profile is not affected by increasing the rate of diffusion (i.e., by decreasing the energy barrier for diffusion). However, slowing down the diffusion by increasing the energy barrier for the diffusion with a factor of 2 results in a significant broadening of the second peak with some evidence for peak splitting. CO on a site is then more likely to have a species on an adjacent site (e.g., formed upon dissociation of another CO), since it will diffuse away rather slowly. Thus, the increased coverage surrounding adsorbed CO may induce further CO-desorption. Furthermore, co-adsorbed C and O formed in adjacent sites upon CO-dissociation will not separate easily, which may result in recombination and subsequent desorption. The high temperature peak in the CO-TPD profile (β-CO) hardly affected by increasing the energy barrier for diffusion by a factor of two. Interestingly, the high temperature peak is affected by lowering the diffusion barrier. This may be attributed to the difference in the mobility of C and O, with surface carbon being much less mobile than surface oxygen (see
Table 3). Decreasing the energy barrier for surface diffusion implies that both species will have similar, high mobility. They will try to avoid each other to minimize lateral interactions, thus resulting in a shift of the high temperature peak further upon increasing the rate of diffusion.
Shifting the assumption of an early transition state for CO dissociation to a late transition state affects mainly the second peak in the CO-TPD profile. A late transition state implies higher activation energy for CO dissociation, whilst at the same time, CO on adjacent sites will diffuse to energetically more favorable sites. This will lead to an increase in the desorption energy shifting the maximum for the second peak. It can be further noted that the second peak is significantly broadened since the stability of adsorbed CO depends on its immediate neighborhood (with the absence of nearest neighbors resulting in the strongest CO adsorption).
The experimentally observed CO-TPD profile from Fe(100) [
18] seems to be modeled best using either the quasi-chemical approach (QCA) or the kinetic Monte Carlo method (kMC). The quasi-chemical approach is a mean-field model assuming a rapid equilibration between the interacting species. The kinetic Monte Carlo method accounts for both local effects and the finite rate of diffusion. Reducing the activation energy for the diffusion of CO did not change the low temperature region of the simulated CO-TPD profile using kMC significantly implying that the origin of the low temperature desorption maximum in the kMC model is due the local effect (i.e., the presence of CO still surrounded by co-adsorbed CO at the nearest neighbor position, which has relatively low activation energy for desorption). The local effect simulated in the kMC model is further demonstrated by slowing down the diffusion of in particular surface oxygen (vide supra). The similarity between the CO-TPD profile from Fe(100) simulated using the quasi-chemical approach (QCA), and the kinetic Monte Carlo method (kMC) may seem, therefore, fortuitous. However, the energy as modeled using the quasi-chemical approach (QCA—see Equation (11)) is in principle a sigmoid function rather than a linear function as predicted by the Bragg–Williams approximation. Hence, the increase in the strength of adsorption with decreasing coverage will be initially less in the QCA model compared to the BWA-model favoring desorption of less strongly adsorbed species. The strength of adsorption as predicted using increases rapidly upon decreasing the coverage further bringing the desorption process virtually to a halt. Thus, QCA starts mimicking the behavior observed with kMC for this system with repulsive lateral interactions and a low rate of surface diffusion. QCA appears to be the next best approach if the required parameter set for kMC is not available or if the kMC-model becomes computationally too cumbersome.