In-Situ Catalytic Fast Pyrolysis of Pinecone over HY Catalysts
Abstract
1. Introduction
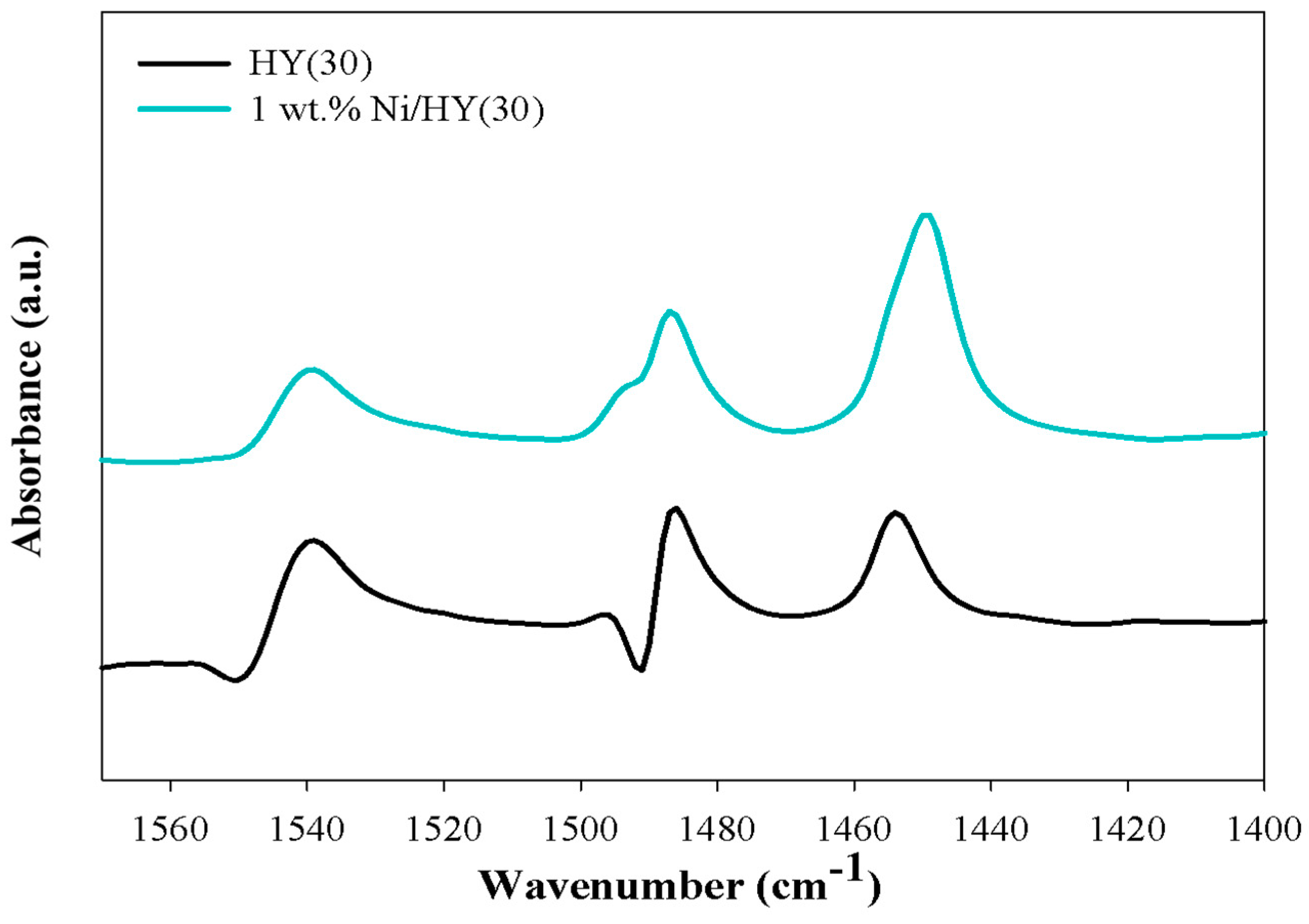
2. Results and Discussion
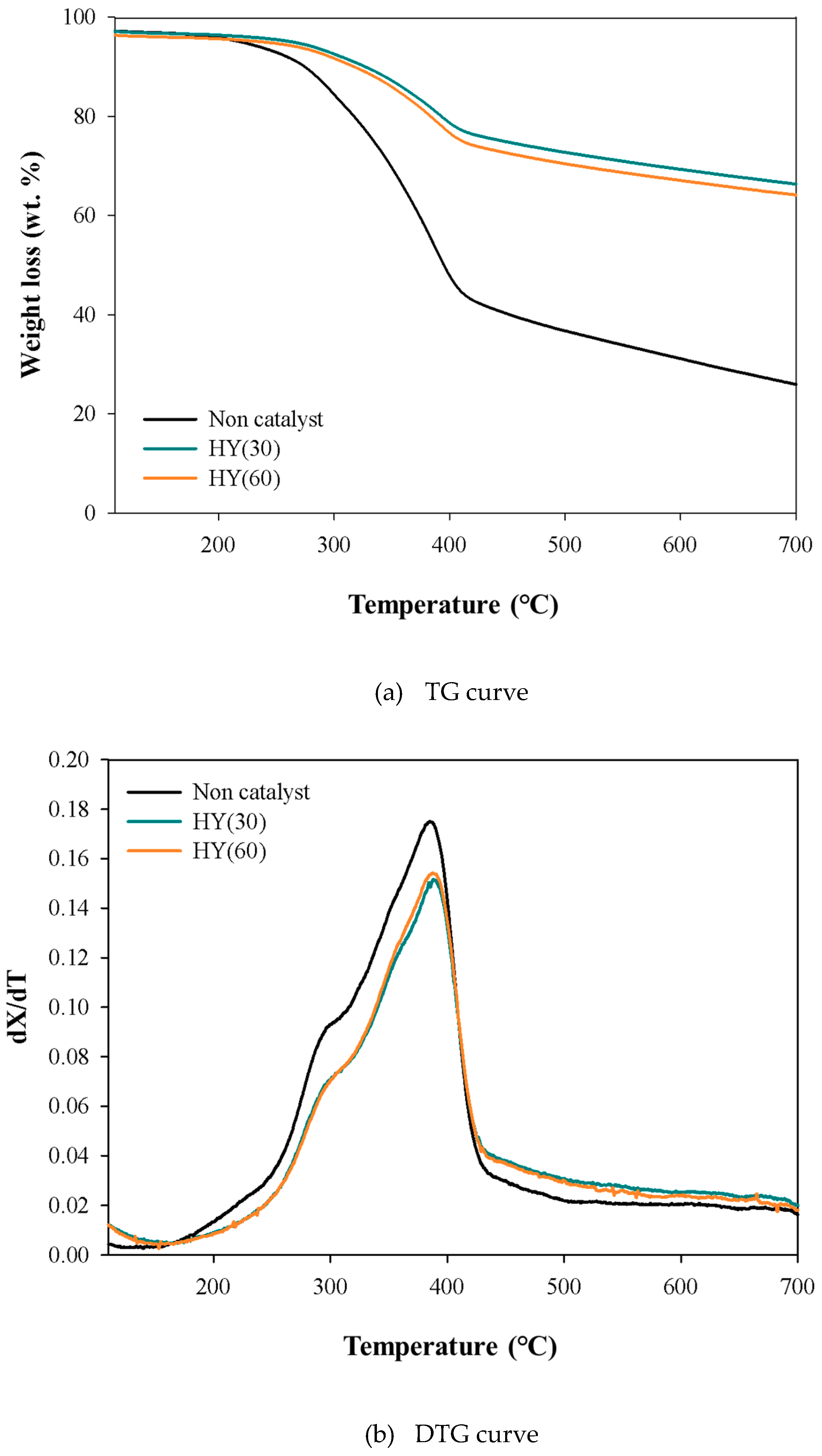
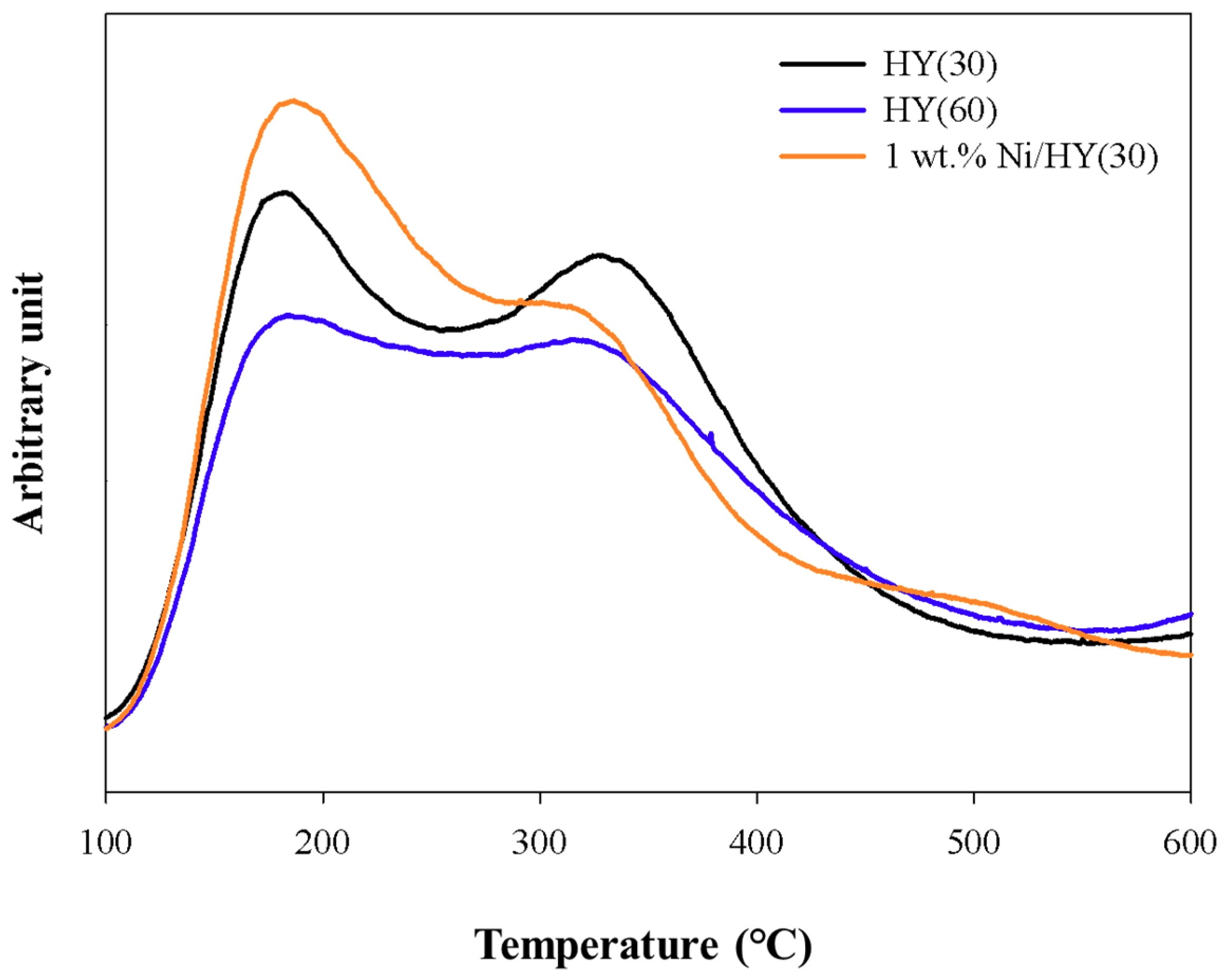
2.1. TG Analysis
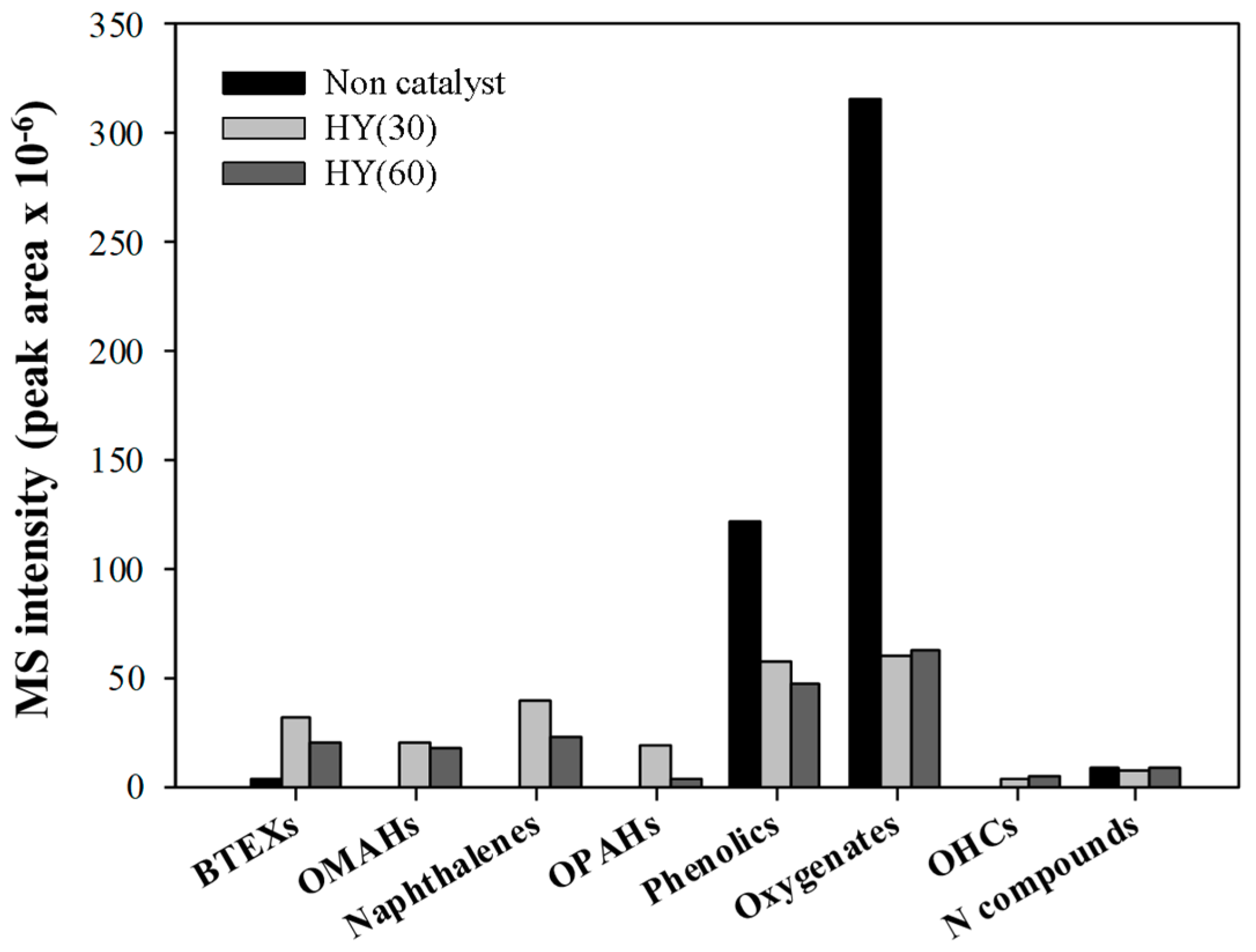
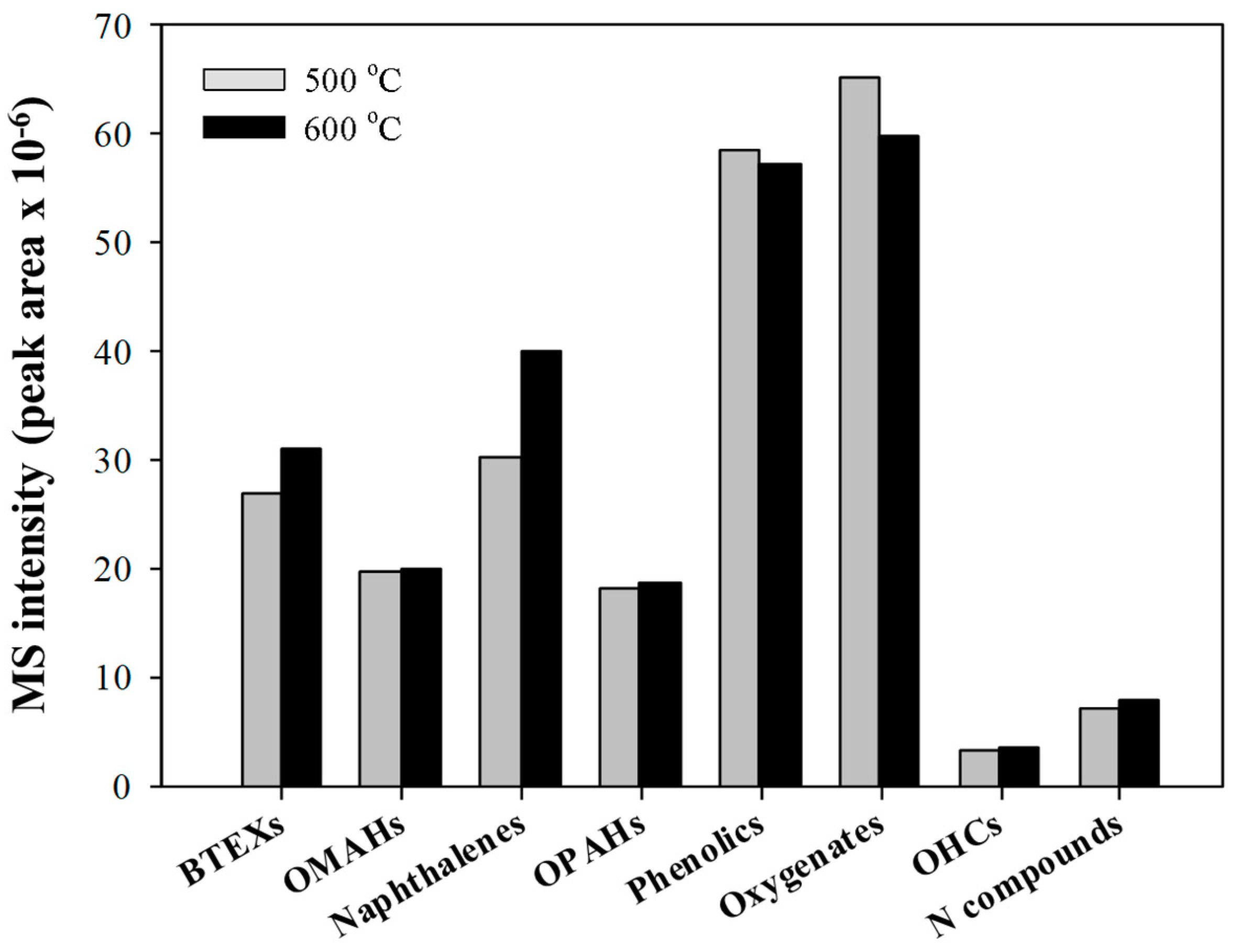
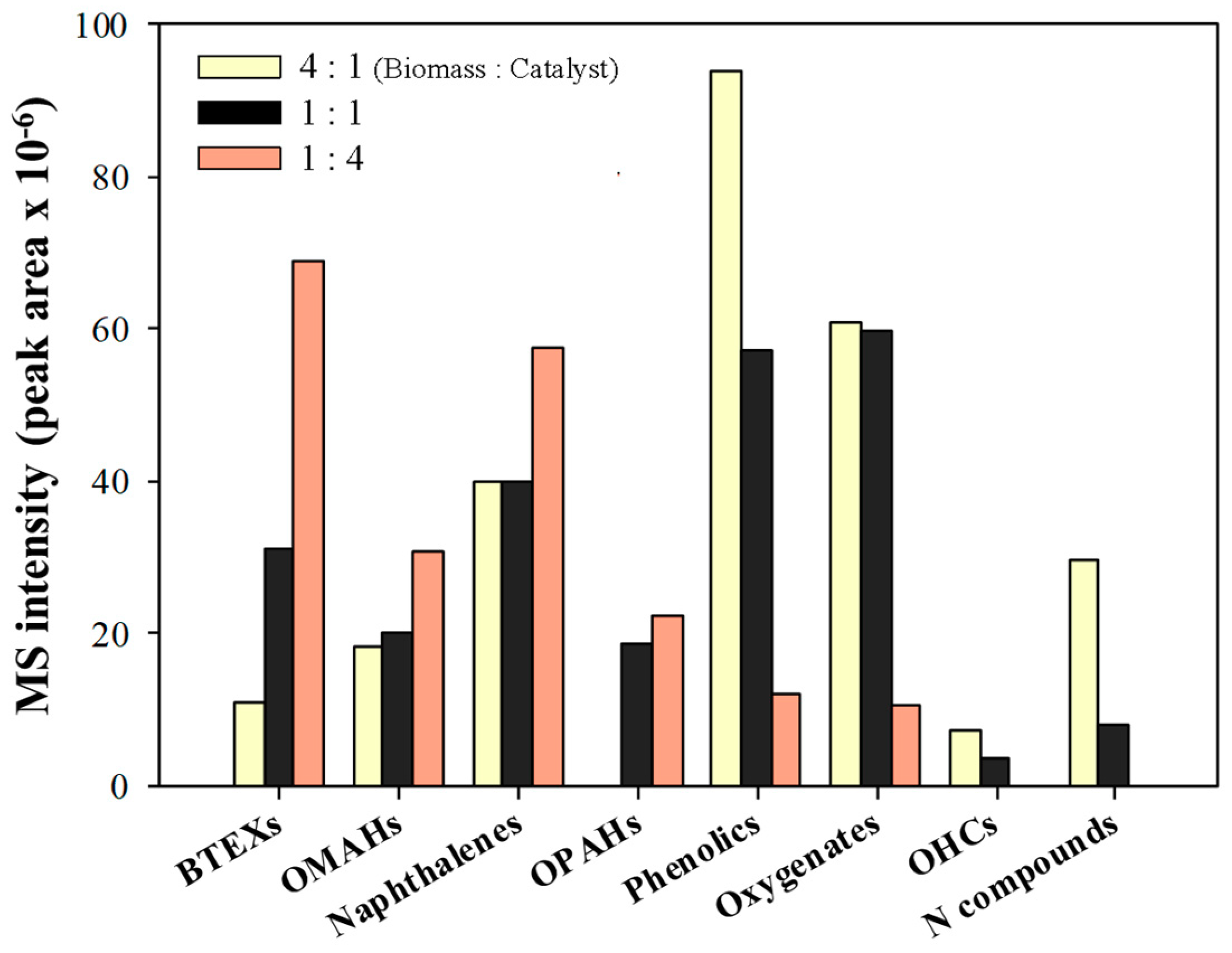
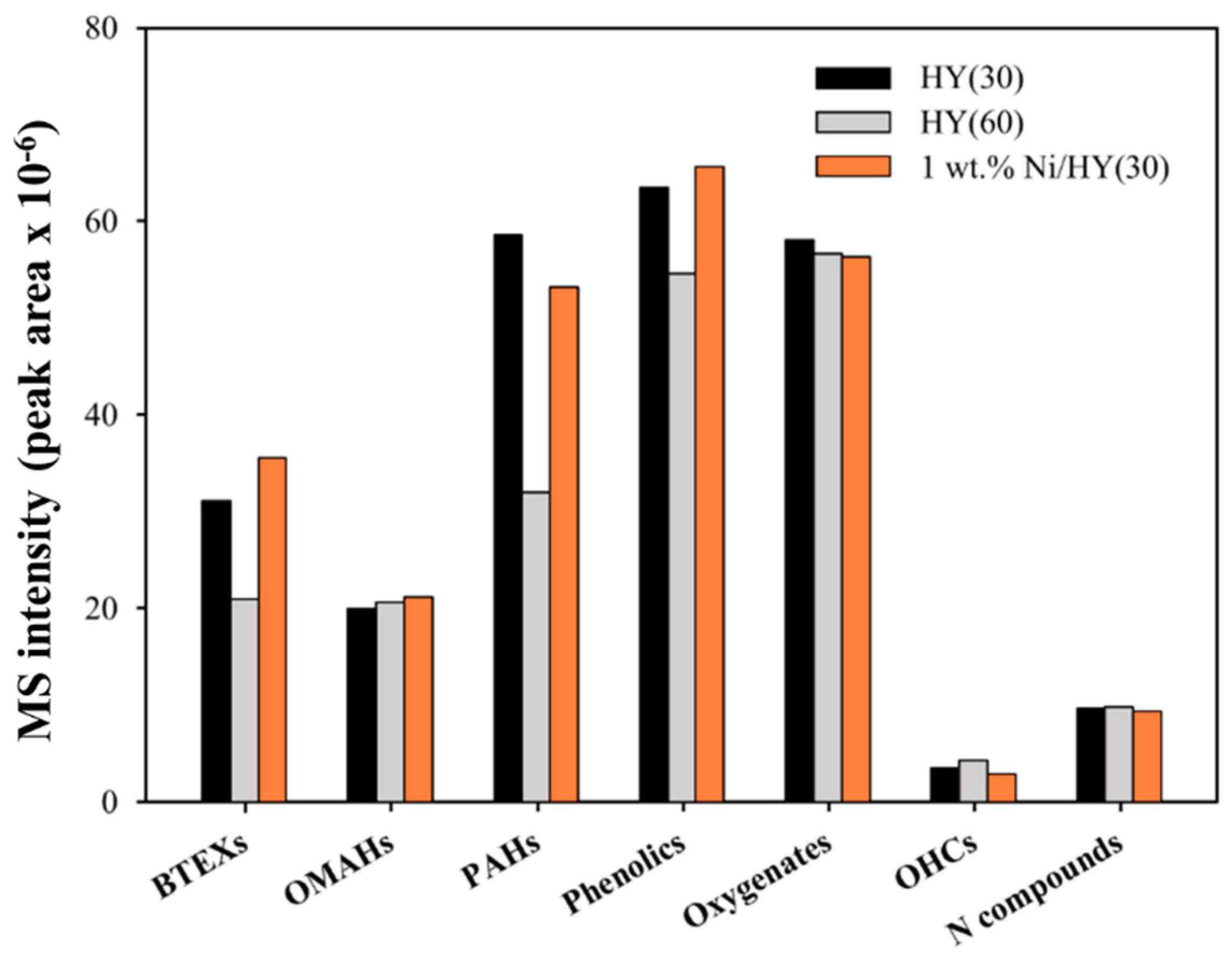
2.2. Py-GC/MS Analysis
3. Materials and Methods
3.1. Pinecone
3.2. Catalysts
3.3. TGA
3.4. Py-GC/MS Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wang, K.; Kim, K.H.; Brown, R.C. Catalytic pyrolysis of individual components of lignocellulosic biomass. Green Chem. 2014, 16, 727–735. [Google Scholar] [CrossRef]
- Mamaeva, A.; Tahmasebi, A.; Yu, J. The effects of mineral salt catalysts on selectivity of phenolic compounds in bio-oil during microwave pyrolysis of peanut shell. Korean J. Chem. Eng. 2017, 34, 672–680. [Google Scholar] [CrossRef]
- Devarajan, Y.; Nagappan, B.K.; Munuswamy, D.B. Performance and emissions analysis on diesel engine fuelled with cashew nut shell biodiesel and pentanol blends. Korean J. Chem. Eng. 2017, 34, 1021–1026. [Google Scholar] [CrossRef]
- Yadav, A.K.; Khan, M.E.; Pal, A. Biodiesel production from oleander (Thevetia Peruviana) oil and its performance testing on a diesel engine. Korean J. Chem. Eng. 2017, 34, 340–345. [Google Scholar] [CrossRef]
- Cherikkallinmel, S.K.; Sugunan, S.; Narayanan, B.N.; Faisal, P.A. Statistical optimization for lithium silicate catalysed production of biodiesel from waste cooking oil. Korean J. Chem. Eng. 2017, 34, 2840–2851. [Google Scholar] [CrossRef]
- Xie, H.; Yu, Q.; Duan, W.; Wang, K.; Li, X.; Shi, X. Pyrolysis characteristics and kinetics of lignin derived from three agricultural wastes. J. Renew. Sustain. Energy 2013, 5, 063119. [Google Scholar] [CrossRef]
- Yang, J.-H.; Shin, H.-Y.; Ryu, Y.-J.; Lee, C.-G. Hydrothermal liquefaction of Chlorella vulgaris: Effect of reaction temperature and time on energy recovery and nutrient recovery. J. Ind. Eng. Chem. 2018, 68, 267–273. [Google Scholar] [CrossRef]
- Phusunti, N.; Phetwarotai, W.; Tekasakul, S. Effects of torrefaction on physical properties, chemical composition and reactivity of microalgae. Korean J. Chem. Eng. 2018, 35, 503–510. [Google Scholar] [CrossRef]
- Kim, I.; Dwiatmoko, A.A.; Choi, J.W.; Suh, D.J.; Jae, J.; Ha, J.M.; Kim, J.K. Upgrading of sawdust pyrolysis oil to hydrocarbon fuels using tungstate-zirconia-supported Ru catalysts with less formation of cokes. J. Ind. Eng. Chem. 2017, 56, 74–81. [Google Scholar] [CrossRef]
- Pang, S. Advances in thermochemical conversion of woody biomass to energy, fuels and chemicals. Biotechnol. Adv. 2019, 37, 589–597. [Google Scholar] [CrossRef]
- Boutaieb, M.; Guiza, M.; Roman, S.; Nogales, S.; Ledesma, B.; Ouederni, A. Pine cone pyrolysis: Optimization of temperature for energy recovery. Environ. Prog. Sustain. Energy 2019, e13272. [Google Scholar] [CrossRef]
- Park, J.-H.; Wang, J.J.; Kim, S.-H.; Jeong, C.Y.; Jeon, J.-R.; Park, K.H.; Cho, J.-S.; Delaune, R.D.; Sep, D.-C. Cadmiun adsorption characteristics of biochars derived using various pine tree residues and pyrolysis. J. Colloid Interface Sci. 2019, 553, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Ng, W.; Wong, B.S.E.; Baeg, G.H.; Wang, C.-H.; Ok, Y.S. Characterization and ecotoxicological investigation of biochar produced via slow pyrolysis: Effect of feedstock composition and pyrolysis conditions. J. Hazard. Mater. 2019, 365, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Shafahat, H.; Kim, J.; Kang, B.S.; Jeon, J.K.; Jung, S.C.; Lee, I.G.; Park, Y.K. Stabilization of bio-oil over a low cost dolomite catalyst. Korean J. Chem. Eng. 2018, 35, 922–925. [Google Scholar] [CrossRef]
- Xue, Y.; Yan, C.; Zhao, X.; Huang, S.; Guo, C. Ni/La2O3-ZrO2 catalyst for hydrogen production from steam reforming of acetic acid as a model compounds of bio-oil. Korean J. Chem. Eng. 2017, 34, 305–313. [Google Scholar] [CrossRef]
- Lee, Y.; Shafaghat, H.; Kim, J.; Jeon, J.K.; Jung, S.C.; Lee, I.G.; Park, Y.K. Upgrading of pyrolysis bio-oil using WO3/ZrO2 and Amberlyst catalysts: Evaluation of acid number and viscosity. Korean J. Chem. Eng. 2017, 34, 2180–2187. [Google Scholar] [CrossRef]
- Zhao, S.; Liu, M.; Zhao, L.; Lu, J. Effects of organic and inorganic metal salts on thermogravimetric pyrolysis of biomass components. Korean J. Chem. Eng. 2017, 34, 3077–3084. [Google Scholar] [CrossRef]
- Zhou, H.; Long, Y.; Meng, A.; Chen, S.; Li, Q.; Zhang, Y. A novel method for kinetics analysis of pyrolysis of hemicellulose, cellulose, and lignin in TGA and macro-TGA. RSC Adv. 2015, 5, 26509–26516. [Google Scholar] [CrossRef]
- Yao, C.; Tian, H.; Hu, Z.; Yin, Y.; Chen, D.; Yan, X. Characteristics and analyses of different genus biomass pyrolysis. Korean J. Chem. Eng. 2018, 35, 511–517. [Google Scholar] [CrossRef]
- French, R.; Czernik, S. Catalytic pyrolysis of biomass for biofuels production. Fuel Process. Technol. 2010, 91, 25–32. [Google Scholar] [CrossRef]
- Lazaridis, P.A.; Fotopoulos, A.A.P.; Karakoulia, S.A.; Triantafyllidis, K.S. Catalytic Fast Pyrolysis of Kraft Lignin with Conventional, Mesoporous and Nanosized ZSM-5 Zeolite for the Production of Alkyl-Phenols and Aromatics. Front. Chem. 2018, 6, 295. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Kim, Y.-M.; Lee, H.W.; Jae, J.; Kim, D.H.; Jung, S.C.; Watanabe, C.; Park, Y.-K. Catalytic Copyrolysis of Cellulose and Thermoplastics over HZSM-5 and HY. ACS Sustain. Chem. Eng. 2016, 4, 1354–1363. [Google Scholar] [CrossRef]
- Choi, J.H.; Kim, S.-S.; Suh, D.J.; Jang, E.-J.; Min, K.-I.; Woo, H.C. Characterization of the bio-oil and bio-char produced by fixed bed pyrolysis of the brown alga Saccharina japonica. Korean J. Chem. Eng. 2016, 33, 2691–2698. [Google Scholar] [CrossRef]
- Shen, D.; Jin, W.; Hu, J.; Xiao, R.; Luo, K. An overview on fast pyrolysis of the main constituents in lignocellulosic biomass to valued-added chemicals: Structures, pathways and interactions. Renew. Sustain. Energy Rev. 2015, 51, 761–774. [Google Scholar] [CrossRef]
- Ma, Z.; Sun, Q.; Ye, J.; Yao, Q.; Zhao, C. Study on the thermal degradation behaviors and kinetics of alkali lignin for production of phenolic-rich bio-oil using TGA-FTIR and Py-GC/MS. J. Anal. Appl. Pyrolysis 2016, 117, 116–124. [Google Scholar] [CrossRef]
- Bai, X.; Kim, K.H.; Brown, R.C.; Dalluge, E.; Hutchinson, C.; Lee, Y.J.; Dalluge, D. Formation of phenolic oligomers during fast pytolysis of lignin. Fuel 2014, 128, 170–179. [Google Scholar] [CrossRef]
- Rezaei, P.S.; Shafaghat, H.; Daud, W.M.A.W. Production of green aromatics and olefins by catalytic cracking of oxygenate compounds derived from biomass pyrolysis: A review. Appl. Catal. A-Gen. 2014, 469, 490–511. [Google Scholar] [CrossRef]
- Cheng, Y.-T.; Huber, G.W. Production of targeted aromatics by using Diels-Alder classes of reactions with Furans and olefins overZSM-5. Green Chem. 2012, 14, 3114–3125. [Google Scholar] [CrossRef]
- Ben, H.; Ragauskas, A.J. Influence of Si/Al ratio of ZSM-5 Zeolite on the Properties of Lignin pyrolysis Products. ACS Sustain. Chem. Eng. 2013, 1, 316–324. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.H.; Park, J.; Kim, J.K.; An, D.; Song, I.K.; Choi, J.W. Catalytic pyrolysis of lignin over HZSM-5 catalysts: Effect of various parameters on the production of aromatic hydrocarbon. J. Anal. Appl. Pyrolysis 2015, 114, 273–280. [Google Scholar] [CrossRef]
- Chung, K.H.; Jeong, S.; Kim, H.; Kim, S.J.; Park, Y.-K.; Jung, S.-C. Highly Selective Catalytic Properties of HZSM-5 Zeolite in the Synthesis of Acetyl Triethyl Citrate by the Acetylation of Triethyl Citrate with Acetic Anhydride. Cataylsts 2017, 7, 321. [Google Scholar] [CrossRef]
- Usman, M.; Li, D.; Razzaq, R.; Yaseen, M.; Li, C.; Zhang, S. Novel MoP/HY catalyst for the selective conversion of naphthalene to tetralin. J. Ind. Eng. Chem. 2015, 23, 21–26. [Google Scholar] [CrossRef]
- Malik, G.M.; Patel, P.C.; Tailor, J.H.; Patel, S.S. Synthesis, Characterization, and Dyeing Performance of Thiadiazole Derivatives. Fiber Polym. 2018, 19, 1670–1677. [Google Scholar] [CrossRef]
- Ben, H.; Ragauskas, A.J. Pyrolysis of Kraft lignin with additives. Energy Fuels 2011, 25, 4662–4668. [Google Scholar] [CrossRef]
- Dai, L.; Wang, Y.; Liu, Y.; Ruan, R.; Duan, D.; Zhao, Y.; Yu, Z.; Jiang, L. Catalytic fast pyrolysis of torrefied corn cob to aromatic hydrocarbons over Ni-modified hierarchical ZSM-5 catalyst. Bioresour. Technol. 2019, 272, 407–414. [Google Scholar] [CrossRef]
- Almendros, A.O.I.; Martin-Lara, M.A.; Ronda, A.; Perez, A.; Blazquez, G.; Calero, M. Physico-chemical characterization of pine cone shell and its use as biosorbent and fuel. Bioresour. Technol. 2015, 196, 406–412. [Google Scholar] [CrossRef]
- Vijaykumar, S.M.; Halgeri, A.B. Metal ion-exchanged zeolites as highly active solid acid catalysts for the green synthesis of glycerol carbonate from glycerol. RSC Adv. 2015, 5, 14286–14293. [Google Scholar]
- Busca, G. Acidity and basicity of zeolites: A fundamental approach. Microporous Mesoporous Mater. 2017, 254, 3–16. [Google Scholar] [CrossRef]
| Compound | Group | Peak Area (×10−6) | |
|---|---|---|---|
| 1 | Methyl furan | Furans | 2.41 |
| 2 | Acetic acid | Acids | 30.05 |
| 3 | Hydroxy propanone | Ketones | 6.22 |
| 4 | Toluene | BTEXs | 3.81 |
| 5 | Methyl pyruvate | Esters | 5.02 |
| 6 | Furfural | Furans | 6.39 |
| 7 | Hydroxy cyclopentenone | Ketones | 10 |
| 8 | Furanone | Ketones | 5.22 |
| 9 | Methyl pyrimidinone | N compounds | 8.22 |
| 10 | Phenol | Phenolics | 4.23 |
| 11 | Methylimino perhydro oxazine | N compounds | 8.82 |
| 12 | Hydroxy methyl cyclopentenone | Ketones | 3.26 |
| 13 | Methyl phenol | Phenolics | 3.68 |
| 14 | Dimethyl hydroxyl furanone | Ketones | 3.16 |
| 15 | Methoxy phenol | Phenolics | 7.47 |
| 16 | 1,2-Benzenediol | Phenolics | 25.74 |
| 17 | Methyl methoxy phenol | Phenolics | 23.5 |
| 18 | Hydroxymethyl furancarboxaldehyde | Aldehydes | 8.85 |
| 19 | Ethyl methoxy phenol | Phenolics | 3.07 |
| 20 | Methyl benzenediol | Phenolics | 10.87 |
| 21 | Vinyl methoxy phenol | Phenolics | 17.46 |
| 22 | Eugenol | Phenolics | 3.16 |
| 23 | Hexanoic acid | Acids | 26.71 |
| 24 | Vanillin | Phenolics | 5.36 |
| 25 | Propenyl methoxy phenol | Phenolics | 17.43 |
| 26 | Levoglucosan | Levoglucosan | 195.10 |
| 27 | Hydroxy methoxy cinnamaldehyde | Aldehydes | 4.57 |
| Proximate Analysis (wt.%) | Water | 2.1 |
| Volatiles | 83.0 | |
| Fixed carbon | 13.4 | |
| Ash | 1.5 | |
| Sum | 100.0 | |
| Ultimate Analysis a (wt.%) | C | 43.9 |
| H | 5.9 | |
| O b | 49.9 | |
| N | 0.3 | |
| Sum | 100.0 |
| Amount of Weak Acid Sites (mmol/g) | Amount of Medium Brönsted Acid Sites (mmol/g) | Amount of Total Acid Sites (mmol/g) | |
|---|---|---|---|
| HY(60) | 5.649 | 5.885 | 11.534 |
| HY(30) | 6.402 | 7.184 | 13.586 |
| 1 wt.% Ni/HY(30) | 7.710 | 4.657 | 12.367 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, J.; Lee, H.W.; Jang, S.H.; Ryu, S.; Kim, Y.-M.; Park, R.-s.; Jung, S.-C.; Jeon, J.-K.; Park, Y.-K. In-Situ Catalytic Fast Pyrolysis of Pinecone over HY Catalysts. Catalysts 2019, 9, 1034. https://doi.org/10.3390/catal9121034
Jeong J, Lee HW, Jang SH, Ryu S, Kim Y-M, Park R-s, Jung S-C, Jeon J-K, Park Y-K. In-Situ Catalytic Fast Pyrolysis of Pinecone over HY Catalysts. Catalysts. 2019; 9(12):1034. https://doi.org/10.3390/catal9121034
Chicago/Turabian StyleJeong, Jaehun, Hyung Won Lee, Seong Ho Jang, Sumin Ryu, Young-Min Kim, Rae-su Park, Sang-Chul Jung, Jong-Ki Jeon, and Young-Kwon Park. 2019. "In-Situ Catalytic Fast Pyrolysis of Pinecone over HY Catalysts" Catalysts 9, no. 12: 1034. https://doi.org/10.3390/catal9121034
APA StyleJeong, J., Lee, H. W., Jang, S. H., Ryu, S., Kim, Y.-M., Park, R.-s., Jung, S.-C., Jeon, J.-K., & Park, Y.-K. (2019). In-Situ Catalytic Fast Pyrolysis of Pinecone over HY Catalysts. Catalysts, 9(12), 1034. https://doi.org/10.3390/catal9121034








