Abstract
Stimulating electronic transitions and promoting exciton dissociation are key to enhancing the photocatalytic performance of polymer carbon nitride (PCN). Herein, a controllable synthesis strategy based on supramolecular self-assembly and mild salt melting crystallization has been developed, successfully preparing carbon nitride-based photocatalytic materials with tunable crystal phase composition. The mixed crystal phases effectively induced significant n→π* electronic transition, expanding the material’s light response range to the near-infrared region (700 nm). Meanwhile, the homojunction promoted the efficient separation of photogenerated carriers through the built-in electric field. Under visible-light excitation, this material exhibits excellent selective catalytic performance, over 99% for the oxidation and removal of H2S into elemental sulfur. This synergistic mechanism of crystal phase engineering in regulating electronic structure and interface charge dynamics provides a new material design strategy for efficient non-metallic photocatalysts.
1. Introduction
Photocatalysis is a technology that uses solar energy to drive redox reactions; it has attracted extensive research interest for its potential to address global energy challenges and environmental pollution issues [1,2]. The systematic investigation of diverse materials, including metal oxides [3], sulfides [4], nitrogen heterocycles [5], and organic polymers [6], has been previously conducted. Among these materials, polymeric carbon nitride (PCN) is regarded as a highly promising photocatalytic material, offering a compelling combination of visible-light response, ease of functionalization, and environmental benignity [7]. However, the inherent limitations of PCN, such as its low specific surface area, high recombination of photoinduced charge, slow charge mobility, and relatively wide bandgap, which leads to low solar light utilization efficiency, significantly restrict its photocatalytic performance. In recent years, the development of various modification strategies, including morphology control, elemental doping, surface engineering, and heterojunction construction, has been actively pursued to extend the photo-response ability and accelerate the charge carrier transport of PCN [8,9,10]. However, achieving sufficient photocatalytic performance in PCN under longer wavelength visible-light irradiation remains a significant challenge.
The fundamental absorption bands in PCN originate largely from the lone pairs of N atoms and the p orbitals of C and N atoms. Theoretical calculations indicate that its light absorption is mainly influenced by π-π* and n-π* electronic transitions, corresponding to absorption edges at approximately 460 nm and 600 nm, respectively [11]. This π-π* transition plays a central role in PCN’s visible-light absorption, forming the foundation of its photocatalytic activity. Although the n-π* electronic transition could, in principle, extend the absorption edge of PCN by reducing the energy gap between these orbitals, it is typically forbidden in pristine PCN due to the orthogonality between the non-bonding (ψn) and anti-bonding (ψπ*) orbitals [12,13]. However, thermal vibrations in practical PCN, driven by entropy, destabilize its perfectly planar geometry, leading to slight structural distortions. This distortion reduces the strict orthogonality of ψn and ψπ*, thereby enabling the activation of the previously prohibited n-π* electronic transition [14]. Based on this theoretical support, various methods such as atomic doping [15], vacancy engineering [16], structural distortions [17], and organic modification [18] have been developed to activate the n-π* electronic transition of PCN. However, many modification methods, while enhancing the activity, sacrifice the chemical or structural stability of the material (such as the leaching of dopants, degradation caused by defects). The newly introduced charge recombination centers (such as impurity atoms, vacancies) may suppress charge carrier mobility. The long-term stability of functional molecular modifications is often compromised by the inherently weak host–guest binding affinity. The relaxation of purely physically modulated structures during catalytic processes, coupled with the challenge of fabricating materials with uniform strain on a large scale, needs to be addressed. Therefore, it is imperative to develop a method that activates the n-π* electronic transitions of PCN while simultaneously ensuring material stability and superior charge transfer kinetics.
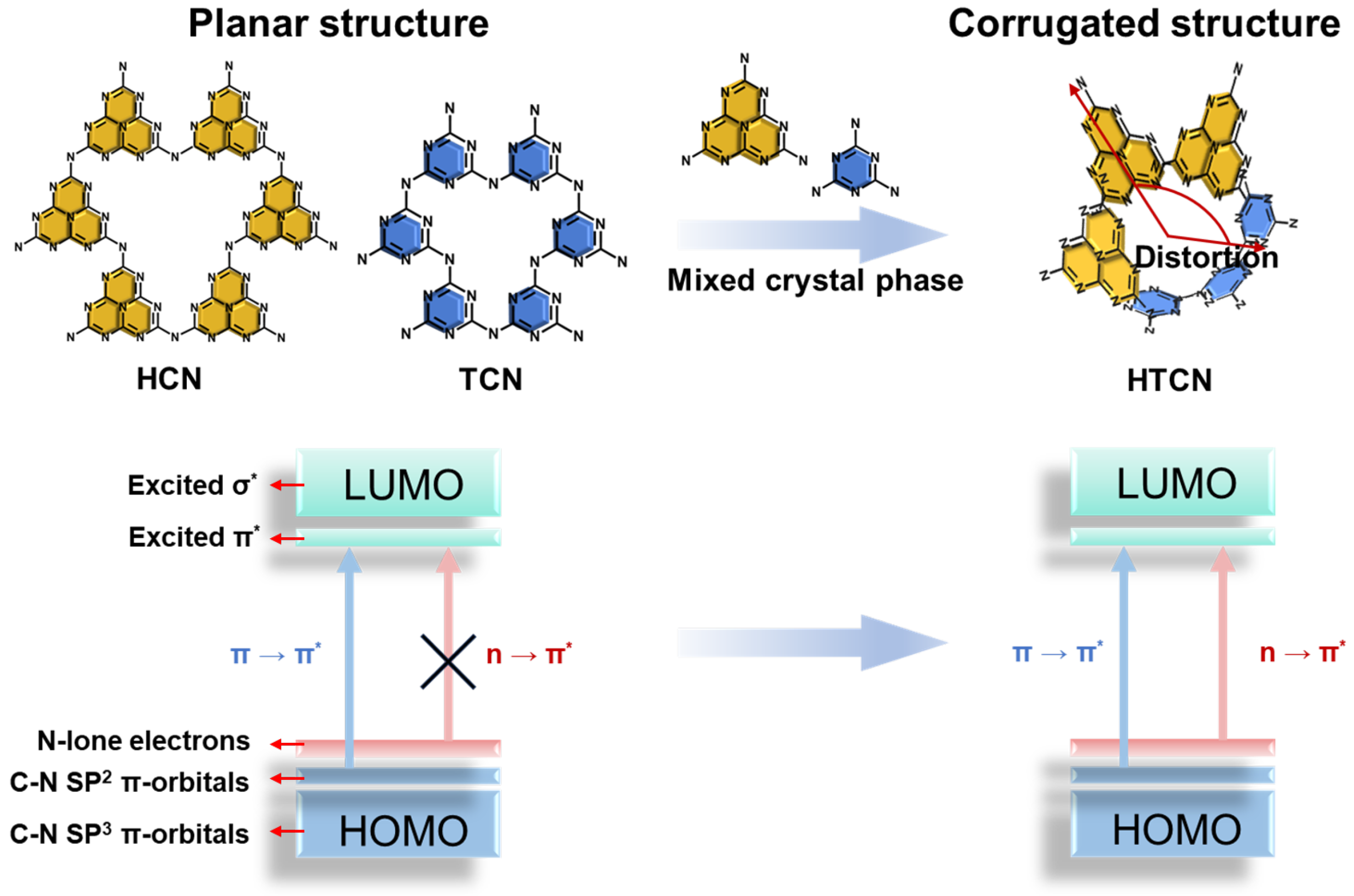
The structure of polymeric carbon nitride is constructed from stable covalent triazine (C3N3) or heptazine (C6N7) units interconnected through terminal nitrogen atoms. When composed exclusively of a single building block, heptazine-based carbon nitride (HCN) and triazine-based carbon nitride (TCN) typically adopt a planar conjugated framework (Figure 1). In contrast, the coexistence and intergrowth of heptazine and triazine units within a single lattice introduce intrinsic lattice mismatch at the molecular scale, giving rise to structural distortion and a corrugated configuration. Such geometric perturbation not only modifies the electronic coupling between adjacent units but also alters the π-conjugation system, thereby critically influencing the optical and electronic properties of the framework [7,19]. In this work, a heptazine/triazine mixed-phase carbon nitride (HTCN) was synthesized via a supramolecular molten salt copolymerization strategy.
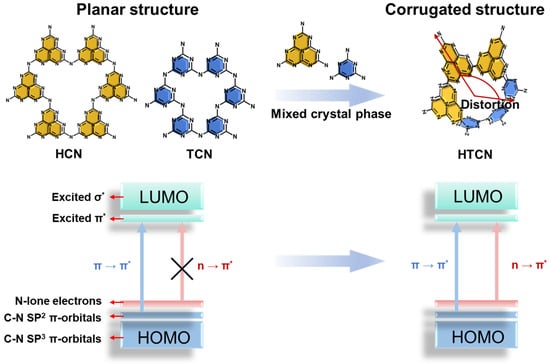
Figure 1.
Electronic transitions in different crystal phase carbon nitrides: planar (HCN and TCN) and corrugated (HTCN) structures of carbon nitride.
The synthesis was rationally decoupled into two independently controllable steps. At room temperature, the precursors (ATZ and CA) were pre-organized into a specific supramolecular stacking configuration through self-assembly, which predetermined the subsequent formation and homogeneous intergrowth of heptazine and triazine phases. Upon thermal treatment, a KCl/LiCl eutectic molten salt served as a confined and homogeneous reaction medium with efficient mass and heat transfer, facilitating polymerization and crystallization while promoting sufficient interconnection between the two distinct units. The intimate intergrowth of triazine and heptazine rings induced stable, intrinsic structural distortion and non-planarity originating from lattice mismatch, ultimately forming a corrugated HTCN architecture. This structural modulation led to a synergistic enhancement in photocatalytic performance. The distortion-induced extension of π-conjugation activated the n-π* electronic transition and red-shifted the absorption edge, enabling broader visible-light harvesting. Simultaneously, the endogenous homojunction formed between the two phase domains facilitated spatial charge separation and reduced interfacial transport resistance, thereby improving charge utilization efficiency. Consequently, HTCN achieved a synergistic integration of widened light absorption and more efficient charge separation and utilization. Compared with thermocatalytic desulfurization under harsh high-temperature and high-pressure conditions and traditional metal-based catalysts with high cost and easy deactivation, this work precisely modulates the intrinsic structure and electronic properties of carbon nitride, overcomes the bottlenecks of light absorption and charge carrier dynamics, significantly improves the efficiency of low-energy photocatalytic H2S decontamination, and provides a new strategy for the construction of highly efficient and green desulfurization systems.
2. Results and Discussion
2.1. Structure Construction and Physicochemical Properties
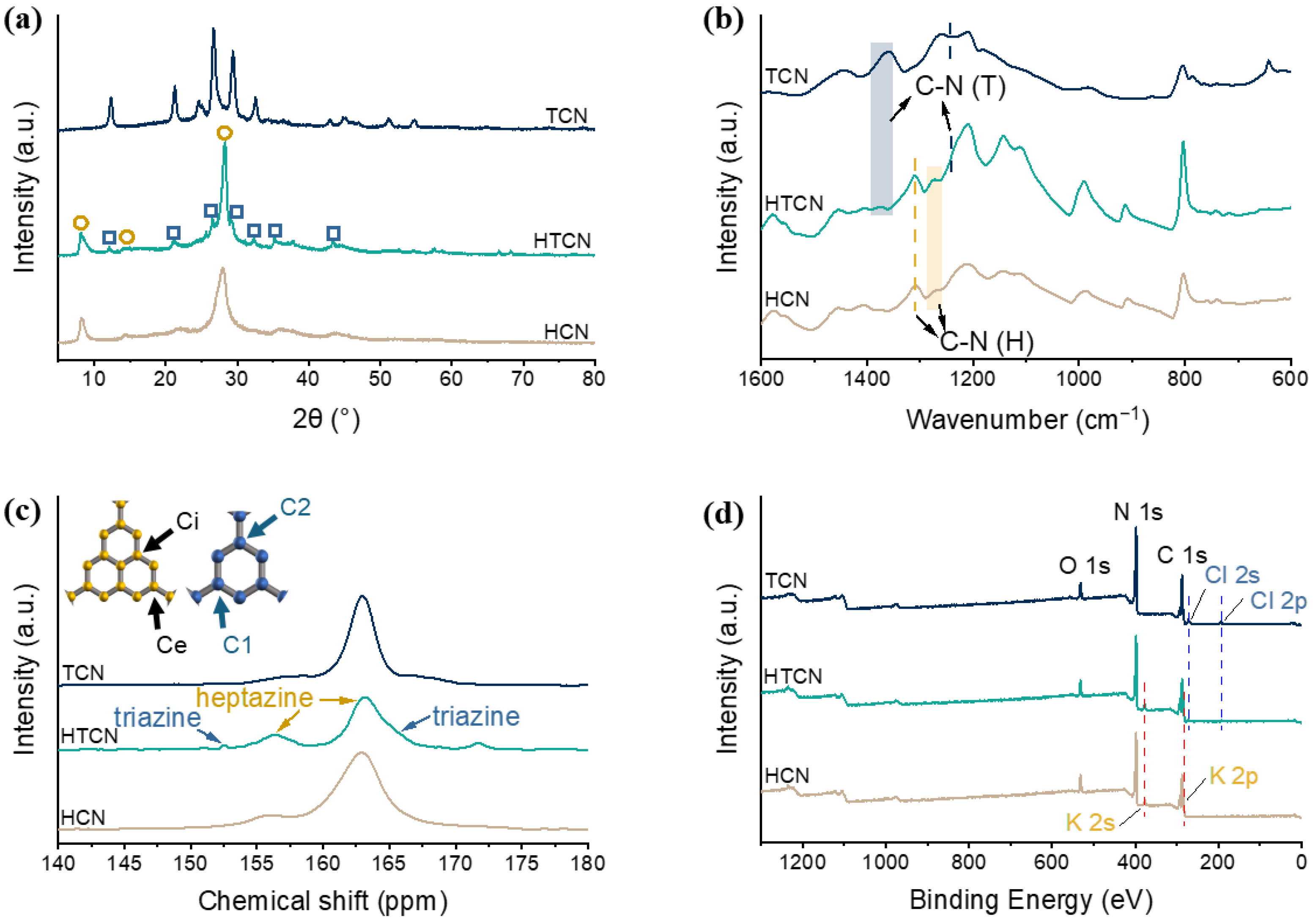
To verify the phase composition of g-C3N4 samples prepared from different precursors via a supramolecular self-assembly molten salt thermal polymerization strategy, X-ray diffraction (XRD) patterns of TCN, HCN, and HTCN were analyzed, as shown in Figure 2a. Here, TCN and HCN served as single-crystal reference phases for triazine-based and heptazine-based carbon nitride, respectively. The XRD pattern of HTCN exhibited two characteristic peaks marked by yellow circles at 27.5° and 8°, which were attributed to the interplanar π-π* stacking of aromatic systems and the in-plane structural ordering of poly heptazine imide (PHI) motifs, respectively [20]. Additionally, five other distinct peaks located at 12.0°, 21.0°, 26.4°, 29.2°, and 32.2° (marked by blue squares) matched well with the crystalline features of poly triazine imides (PTIs), indicating the coexistence of the PTI phase in HTCN [21]. These results confirmed that HTCN comprises a multiphase composite consisting of both PHI and PTI domains, demonstrating that the integration of distinct structural units was successfully achieved through the applied synthesis route.
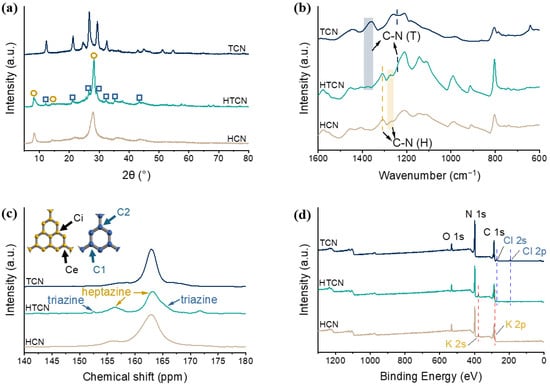
Figure 2.
(a) Powder XRD patterns, (b) FT-IR spectra, (c) 13C NMR spectra, and (d) XPS surveys of TCN, HCN, and HTCN samples.
To further investigate the groups of the as-obtained g-C3N4 samples, the FTIR spectra of the TCN, HCN, and HTCN systems are presented in Figure 2b. The strong absorption bands at 1200–1600 cm−1 correspond to the typical skeletal stretching vibration modes of aromatic carbon nitride heterocycles [22]. The peaks at 1257–1288 and 1310 cm−1 can be attributed to the stretching vibration modes of the connected units of N-(C)3 (full condensation) or CNH-C (partial condensation), which belong to the heptazine-based units [23]. The peaks at 1264–1270 and 1361–1376 cm−1 belong to the vibrations of aromatic N-C groups in triazine-based units [24]. These spectral features collectively confirm the coexistence of both triazine and heptazine structural units in HTCN, providing strong evidence for the successful crystal phase transition.
To gain deeper insight into the chemical structure of the material, solid-state NMR was employed. As shown in the 13C NMR spectrum (Figure 2c), the characteristic chemical shifts observed at 156.4 ppm and 163.2 ppm were assigned to the carbon atoms at the Ci and Ce sites, respectively, within the heptazine-based units [25]. Meanwhile, the signals appearing at 152.5 ppm and 165.0 ppm were correspondingly attributed to the C1 [CN2(NH2)] and C2 [CN2(NH2)] atoms of the triazine-based units [26]. The clear presence of these characteristic peaks from distinct structural units collectively confirms the successful coexistence of both triazine and heptazine building blocks in HTCN.
The X-ray photoelectron spectroscopy (XPS) results confirm the presence of various elements (Figure 2d and Figure S2). The C 1s spectra of HTCN, TCN and HCN could be divided into three peaks (Figure S2a). The peaks at 288.2 eV and 286.8 eV correspond to the sp2-hybridized carbons in the heptazine/triazine rings (N-C=N) and the sp-hybridized carbons in the cyano group, respectively. The peak of 284.8 eV was attributed to exogenous contaminated carbon [27]. The N 1s spectra (Figure S2b) could be deconvoluted into three peaks. The main peak at 398.6 eV belonged to sp2-hybridized nitrogen (C-N=C), while the peaks at 400.4 eV and 401.2 eV corresponded to tertiary nitrogen N(C)3 groups and surface amino groups (C-N-H) [28]. Previously published research mentioned that PHI synthesized from the KCl/LiCl eutectic salt contained K elements [29], while PTI synthesized from the KCl/LiCl eutectic salt contained not only C, N, and H elements, but also Cl and Li elements [30]. From the XPS results, the HTCN sample in this work was found to contain elements including C, N, H, and O, in addition to Cl, K, and Li. In the high-resolution XPS spectra, K was primarily detected in HCN and HTCN (Figure S2c), while Cl (Figure S2d) and Li (Figure S2e) were only observed in TCN and HTCN. Therefore, the K in HTCN primarily originated from the PHI component, whereas the Li and Cl elements were associated with the PTI component. These results provide strong evidence for the formation of a PHI/PTI hybrid crystalline phase in HTCN.
HR-TEM images of the samples provide clear evidence of the formation of a heterojunction between PHI and PTI. As shown in Figure 3a, a heterojunction revealed lattice fringes with spacings of approximately 1.05 nm and 0.73 nm. These lattice fringes were assigned to the (100) planes of PHI and PTI, corresponding to the in-plane periodicity of the heptazine and triazine units, respectively [20,21]. The contact between these two lattices appeared to establish a semi-coherent interface. Furthermore, the dark-field image in Figure 3b displays the morphology of HTCN. Energy-dispersive X-ray spectroscopy (EDX) elemental mapping images demonstrate that HTCN consists of C, N, K, and Cl elements. This observation is consistent with the XPS results.

Figure 3.
(a) HR-TEM image (The red box shows the interlaced growth of the two phases) and (b) dark-field image and element mapping images of HTCN.
2.2. Photoelectric Characteristics and Charge Separation Effect
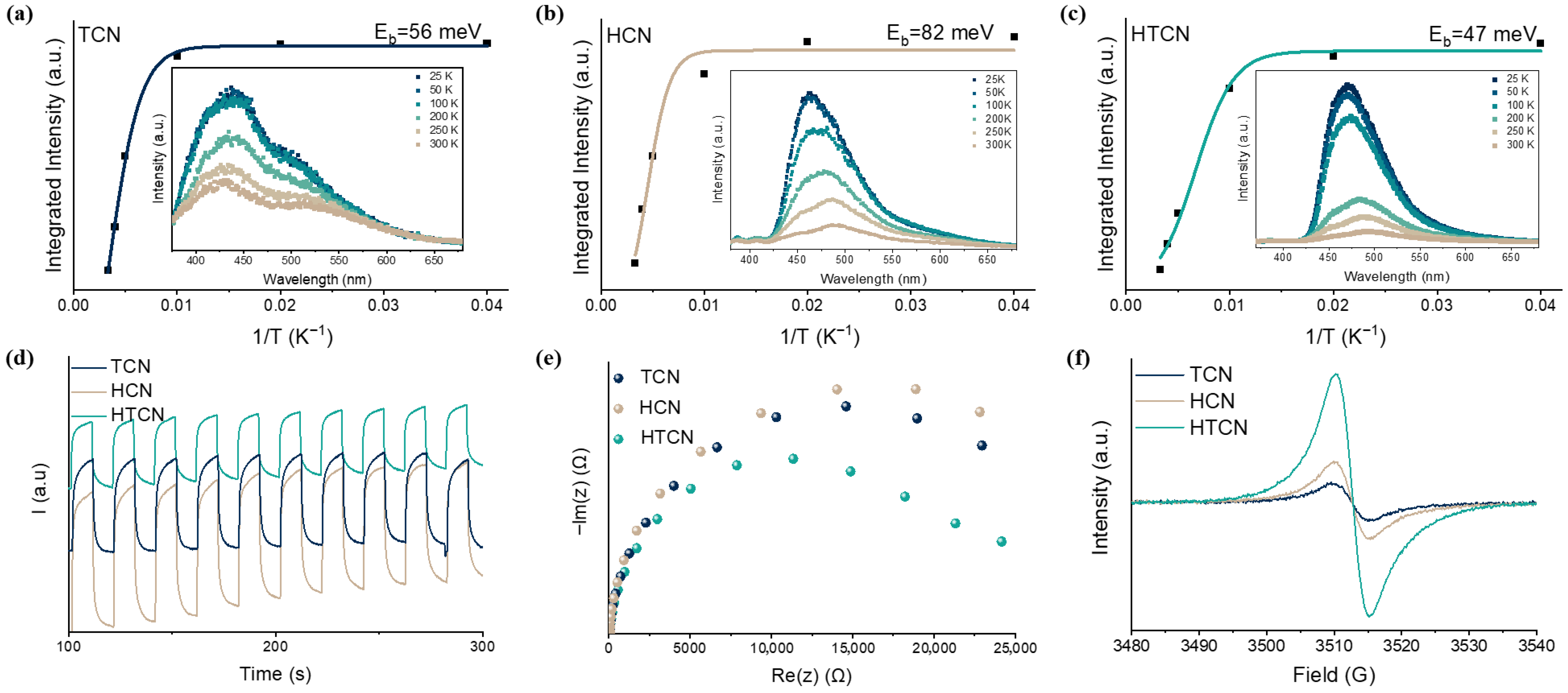
The Eb of these polymers could be estimated from the temperature-dependent photoluminescence (PL) spectra (Figure 4a–c). Upon a decrease in the temperature, the integrated PL peak intensity increases monotonically, and the corresponding Eb can be experimentally obtained by fitting these data using the Arrhenius equation, I(T) = I0/(1 + Aexp (−Eb/kBT)) [31]. The Eb of HCN was 82 meV, that of TCN was 56 meV, while HTCN had a much lower Eb (47 meV). This occurred because the strong localization of energy states within the carbon–nitrogen framework hindered the dissociation of photogenerated electron–hole pairs into free charges, leading instead to the formation of bound excitons. Consequently, the probability of photogenerated charge recombination was significantly increased. Simultaneously, the highly crystalline nature of the HCN and TCN polymeric structures resulted in symmetrical carbon–nitrogen frameworks. This high symmetry inherently limited anisotropic charge transport and spatial charge separation [32]. Through crystal phase tuning in PHI/PTI hybrids, the lattice planes of HTCN undergo distortion. This structural modification enhances delocalization of energy states across the carbon–nitrogen skeleton while inducing spatial heterogeneity in their distribution. Therefore, the planar torsion generated by crystal phase mixing facilitates Eb minimization through the construction of more delocalized charge transfer pathways.
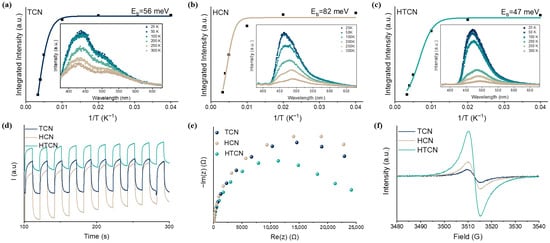
Figure 4.
(a–c) Integrated PL emission intensity as a function of temperature (inset: temperature-dependent PL spectra, λexcitation = 375 nm) of TCN, HCN, and HTCN samples. (d) Transient photocurrent under visible-light irradiation. (e) Electrochemical impedance spectroscopy (EIS) and (f) EPR spectra of HCN, TCN, and HTCN.
Photoelectrochemical measurements provided further evidence for the modulation of charge mobility in these polymers. The transient photocurrent curves (Figure 4d) show that HTCN exhibited a markedly higher transient photocurrent density than those of TCN and HCN, thereby providing compelling evidence that the fabricated PTI/PHI homojunction significantly enhances charge separation. Additionally, the interfacial charge migration efficiency was evaluated by electrochemical impedance spectroscopy (EIS). As demonstrated in Figure 4e, HTCN shows the smallest radii of the semicircular Nyquist plots, indicating that it has the lowest charge transfer resistance and a significantly improved charge mobility. Room-temperature electron paramagnetic resonance (EPR) spectra of TCN, HCN, and HTCN are shown in Figure 4f. Compared to TCN and HCN, almost no change in g-value but a greatly enhanced EPR intensity is observed in HTCN, suggesting the presence of plenty of free charge carriers in the aromatic system. This is ascribed to the symbiosis of PTI and PHI, which caused the structure of the crystalline surface to stretch and deform, leading to a more extended π-conjugated aromatic system, greater delocalization, and greater mobility of the spins [33].
To adjust the ratio of PTI/PHI in the HTCN samples, we obtained HTCNx (x = 0.125, 0.25, 0.5, 1, and 2) samples by regulating the ratio of the two precursors. As shown in Figure S4a, the XRD patterns of the HTCNx samples confirm the coexistence of both triazine and heptazine-based g-C3N4 phases. To accurately ascertain the contents of triazine and heptazine-based g-C3N4 in these samples, the XRD peaks at 25.5~30.5° are fitted into independent ones, as shown in Figure S5. Two of them at about 27.8 and 26.5° are (002) crystal planes of heptazine and triazine-based g-C3N4, respectively, and their peak areas are used to estimate the contents of these two phases [34]. Clearly, the addition of ATZ in ATZ/CA precursors results in an increase in the triazine-based phase. More precisely, increasing incorporation of ATZ led to a higher content in the triazine-based phase, accompanied by a gradual reduction in the overall heptazine-to-triazine group ratio. Notably, the PHI-to-PTI crystal phase ratio in HTCN1 was closest to 1:1 (Table S1). A quantitative analysis was performed by deconvoluting the 13C solid-state NMR spectra (140–180 ppm region) of the HTCNx samples (Figure S6, Table S2). The deconvolution resolved characteristic peaks belonging to triazine and heptazine structures. Analysis of the relative peak areas showed an evident dependency on the ATZ doping level: the PHI (heptazine) phase content systematically decreased as the PTI (triazine) phase content in-creased. This phase evolution was in full agreement with the trends indicated by the XRD analysis.
2.3. Activation and Tuning of n-π* Electronic Transitions
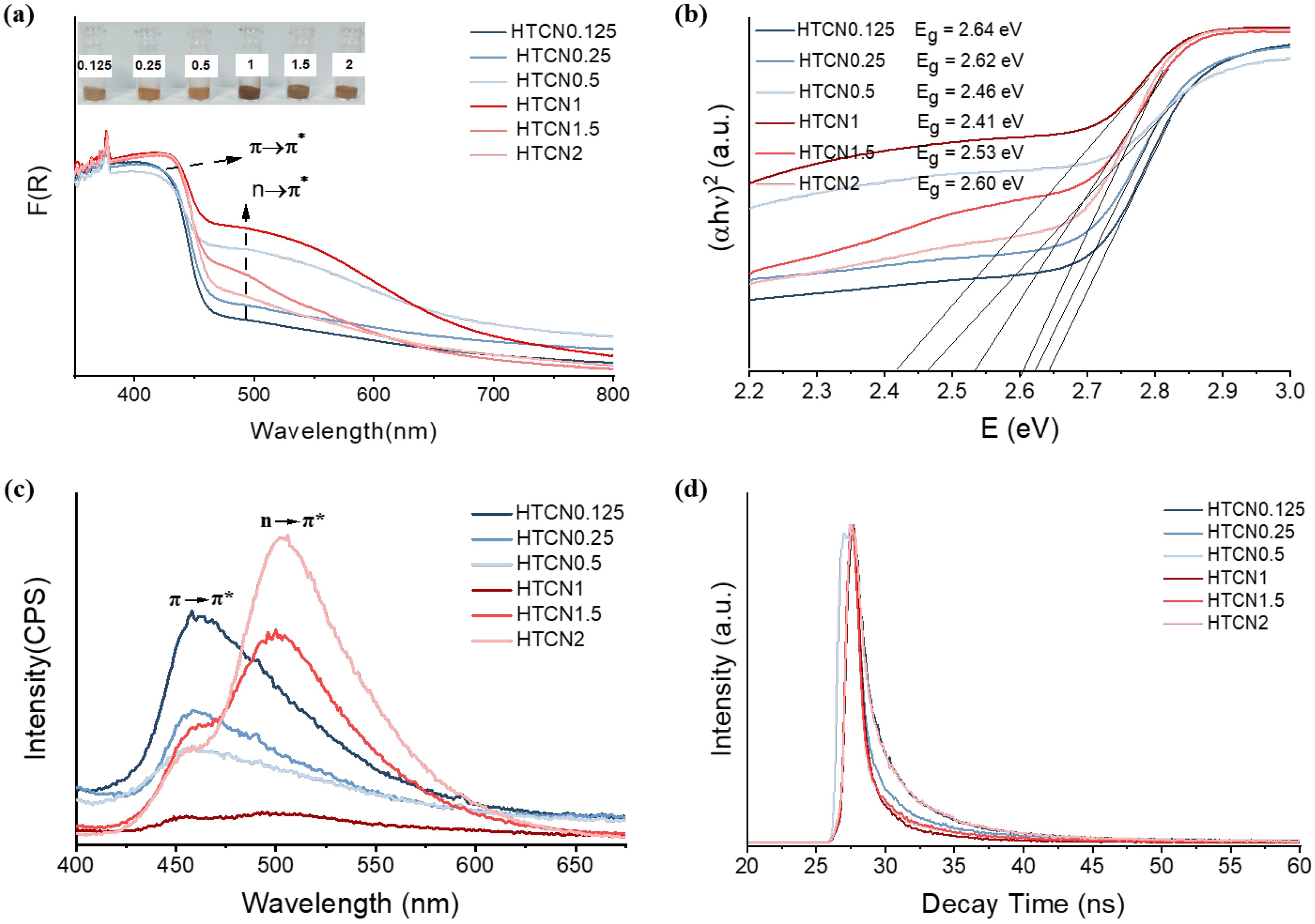
It is noteworthy that the variation in the relative content of the two phases concurrently modulated the optical properties of the materials. The optical absorption of PCN samples is characterized by the UV-Vis spectra displayed in Figure 5. Compared to TCN and HCN (Figure S3), the HTCNx (x = 0.5–1.5) samples show enhanced light absorption in both UV and visible-light regions. A typical absorption edge around 460 nm, identified as the intrinsic electronic transition (π→π*) of PCN, exhibits a redshift to the visible-light region. The reinforced π→π* electronic transition can be attributed to the expansive π-conjugated aromatic framework [35]. More importantly, a new absorption peak at ≈490 nm appears in the UV-Vis spectrum of HTCNx (x = 0.5–2), which is ascribed to the n→π* electronic transition (Figure 5a) [36]. The internal distortion of the originally symmetrical planar structure, attributed to the hybrid growth of the two phases, successfully activated additional n→π* electronic transitions in HTCNx, thereby enabling the utilization of visible light at wavelengths greater than 700 nm. However, the new optical absorption peaks in HTCN0.125 and HTCN2 were exceedingly weak. This indicates that the extent of n→π* electronic transitions was determined by the relative content ratio of the two crystalline phases. Specifically, when the proportion of triazine to heptazine moieties approached unity, the resultant framework exhibited more pronounced structural distortion, which consequently facilitated the occurrence of n→π* electronic transitions. Furthermore, the visible color variation among the HTCNx samples was consistent with their UV-vis spectra. Specifically, HTCN1 exhibited a distinct brick-red color, corresponding to its optimal visible-light absorption.
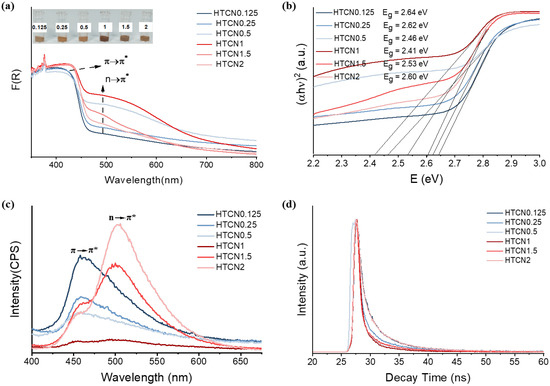
Figure 5.
(a) The UV-Vis absorption spectra and corresponding color spectra of HTCNx. (b) The bandgap of HTCNx, (c) PL spectra, and (d) time-resolved transient PL spectra of HTCNx (x = 0.125, 0.25, 0.5, 1, 1.5, and 2).
Figure 5b presents the bandgaps of HTCNx, which decrease first and then increase with the change in the ratio of crystal phase content. The observed non-monotonic trend was attributed to the competing effects of interfacial distortion and the intrinsic band structures of the constituent phases, explainable across two stages. Initially, bandgap narrowing stemmed from the structural distortion generated at the phase boundaries during hybridization (e.g., between PTI and PHI). This distortion disrupted long-range order, potentially creating defect states near the valence band edge and facilitating orbital hybridization between previously separated levels, which collectively reduced the bandgap [37]. The distortion was maximized at an optimal mixing ratio (near 1:1), leading to the most pronounced absorption edge redshift and the strongest visible-light absorption, corresponding to the condition in HTCN1. In the bandgap increase stage, as the proportion of one phase continued to rise and became dominant, the overall electronic structure of the material began to approximate the intrinsic characteristics of that major phase. Although interfacial distortion still existed, its influence became limited relative to the homogeneous matrix of the dominant phase. Thus, the overall bandgap gradually recovered from its minimum value. In summary, the initial decrease in the bandgap was primarily governed by band modulation induced by interfacial structural distortion, while the subsequent increase reflected the restoration of the intrinsic wide bandgap feature of the dominant phase. This non-monotonic change revealed that precise control over the phase content ratio enables the optimization of the electronic structure in carbon nitride.
The contribution of the n→π* electronic transition to charge transport was also investigated by using the stable and transient photoluminescence (PL) spectrum. As shown in Figure 5c, the emission peaks at 460 nm are related to the π→π* electronic transition in the π-conjugated aromatic ring system. The stronger PL intensity usually represents a higher charge recombination rate. In contrast, in the HTCN1 sample, a nearly quenched PL intensity was observed, signifying highly suppressed recombination of electron–hole pairs. It is worth mentioning that with the adjustment of the crystal phase ratio, the PL peak redshifts from 460 nm to 520 nm, showing that the n→π* electronic transition alters the charge transport pathway [38]. The time-resolved transient PL spectra of HTCNx fitted by the two-exponential decay are shown in Figure 5d. The results reveal that the average lifetime progressively decreased from HTCN0.125 to HTCN1, and then increased again from HTCN1 to HTCN2, suggesting a more efficient non-radiative decay pathway in the HTCN1 sample [39]. Therefore, the internal distortion and homojunction induced by crystal phase engineering can promote band bending and construct an internal electric field for charge separation, achieving a synergistic enhancement of light capture and charge utilization.
2.4. DFT Calculations
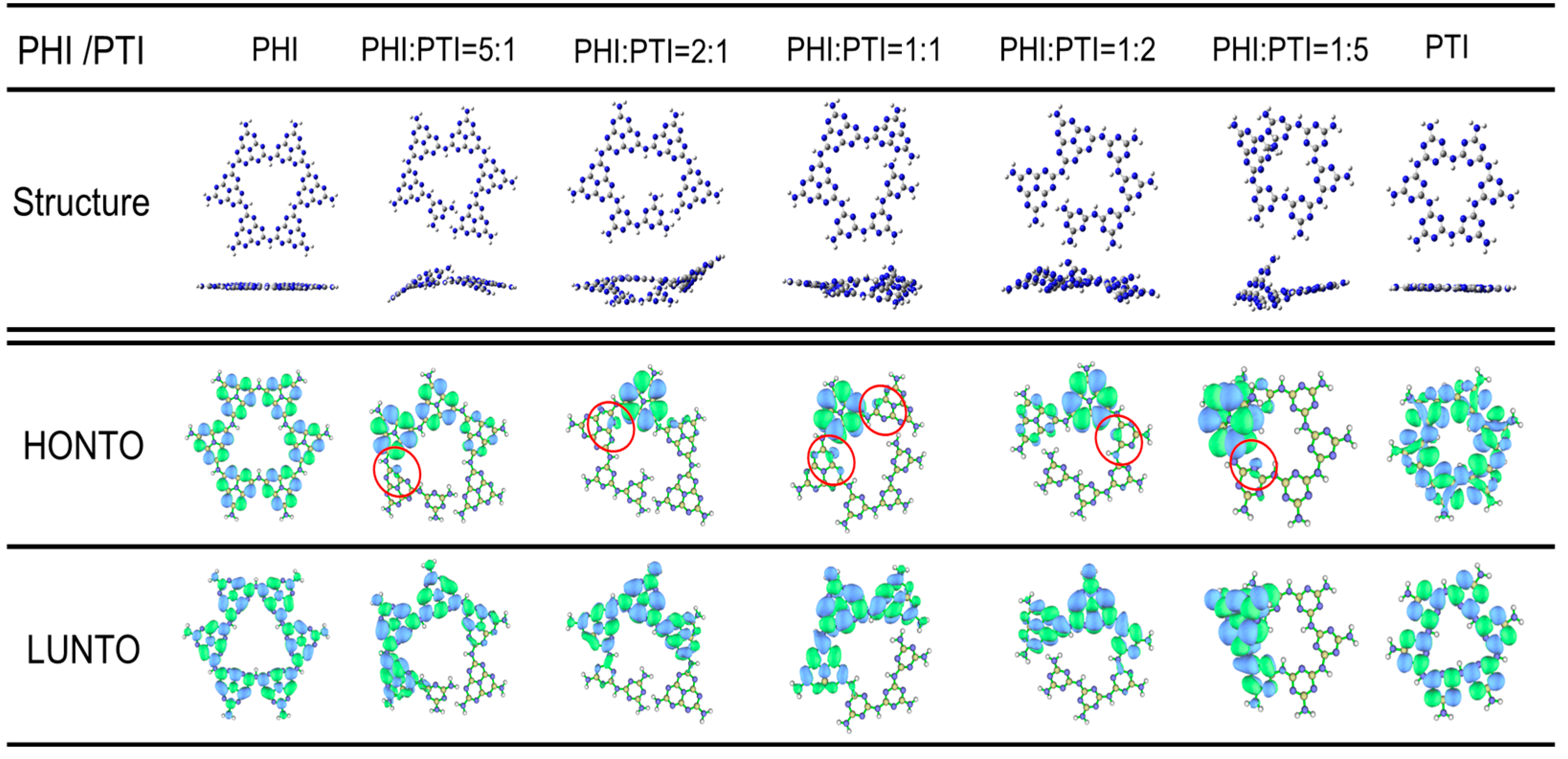
To investigate the effect of symmetry breaking, induced by the hybrid growth of PTI and PHI, on the redistribution of electronic states, simplified models consisting of six basic units (PTI or PHI) were constructed via theoretical calculations to evaluate the electronic structures of samples with different phase ratios (Figure 6). The results indicate that pure PTI (TCN) and PHI (HCN), with their highly symmetric structures, exhibited centrosymmetric charge distributions. In contrast, when their ratio was altered to form an asymmetric hybrid structure (HTCN), charge redistribution occurred, effectively separating the e− and h+. This separation induced the formation of a built-in polarization electric field. This field served as a crucial dynamic factor driving the separation of photogenerated charges, directly influencing photocatalytic performance. Specifically, the HOMO and LUMO of pure PTI and PHI were predominantly localized on N orbitals and C, N orbitals, exhibiting significant spatial overlap, which is unfavorable for the effective separation of electrons (e−) and holes (h+). Conversely, in the HTCN models, the HOMO and LUMO showed spatially separated characteristics. The HOMO was concentrated on nitrogen atoms with lone electron pairs at sites of structural distortion, while the LUMO was mainly distributed on the adjacent π-conjugated skeleton. Consequently, the polarization field at the homojunction interface effectively drove the migration of e- from the HOMO to the LUMO, helping to overcome the Coulombic attraction between e- and h+, thereby suppressing their recombination. More importantly, the n→π* electronic transition is formally forbidden by orbital symmetry in perfectly symmetric structures. The structural distortion introduced by adjusting the PHI:PTI ratio broke this symmetry, enabling partial overlap between the lone pair of electrons (n-orbital) on nitrogen atoms and the neighboring π* orbital (the red circle), thus successfully activating the otherwise forbidden n→π* electronic transition.
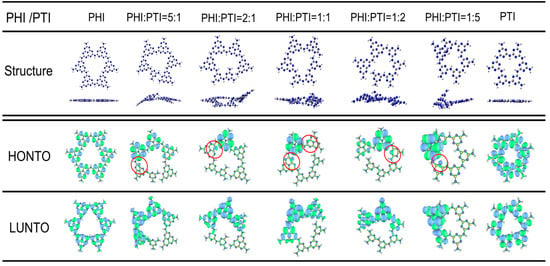
Figure 6.
Different ratios of the triazine and heptazine junction structure and the change in electron transition simulation (Color code: C in grey, H in white, N in blue).
2.5. Photocatalytic Performance
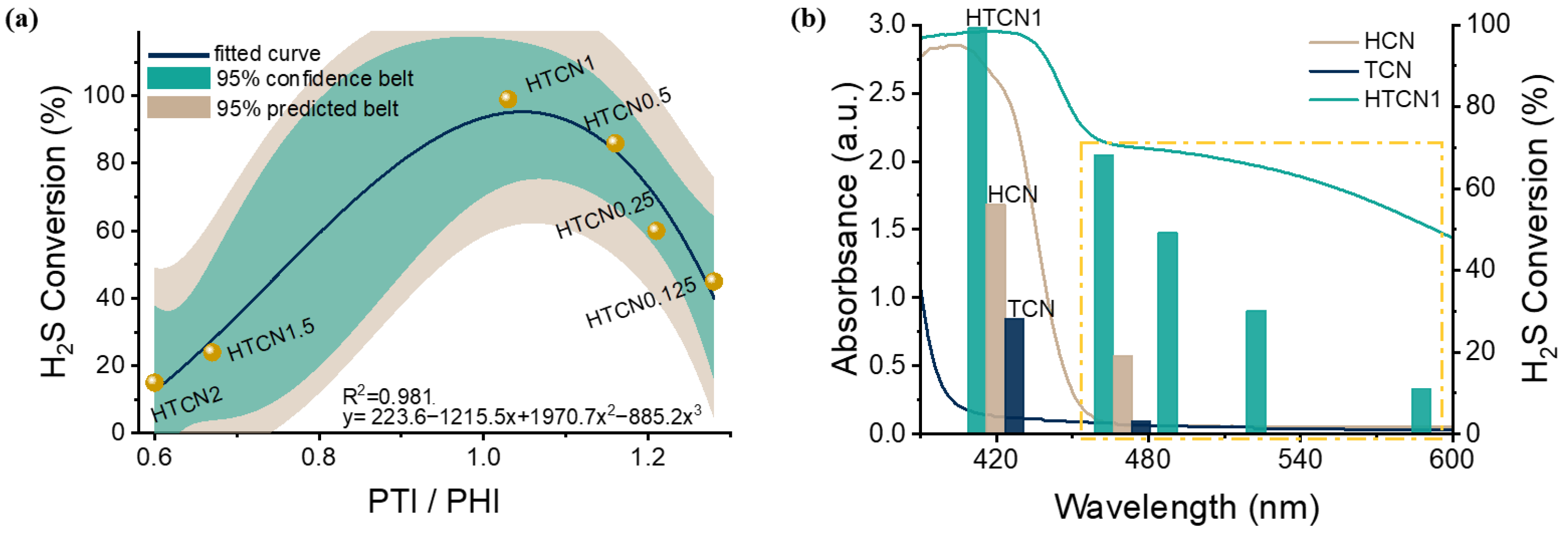
Photocatalytic oxidation of H2S over HTCNx catalysts was evaluated in a photocatalytic reaction flow bed fed with 20 ppm H2S mixed gas (Flow rate: 20 mL/min; CH2S:CO2 = 2:1) under an LED lamp (λ = 420 nm) (Figure 7a). Using the PTI/PHI content ratio as the horizontal coordinate and the H2S conversion rate as the vertical coordinate, the photocatalytic activity of the samples showed a typical volcanic curve that first increased and then decreased with the increase in the crystal phase mixing ratio (data distributed within a 95% confidence belt) [40]. This trend was consistent with the variation in n→π* electronic transition absorption observed in UV-Vis spectroscopy. HTCN1 exhibited the best photocatalytic activity, achieving a hydrogen sulfide conversion rate of over 99%. The catalytic activity of the modified sample far exceeds that of commercial Fe2O3 [41]. Compared with metal cocatalysts (e.g., Mo cocatalyst), our catalyst exhibits superior H2S conversion efficiency under visible-light irradiation [42]. These results clearly highlight the advantages of the modified g-C3N4 for photocatalytic H2S decontamination. The superior catalytic activity was ascribed to the internal electric field established via homojunction construction, which effectively promoted the separation of photogenerated charge carriers [43]. Additionally, structural distortion-induced n→π* electronic transitions enhanced light absorption efficiency. These two factors collectively endowed the catalyst with excellent photocatalytic activity.
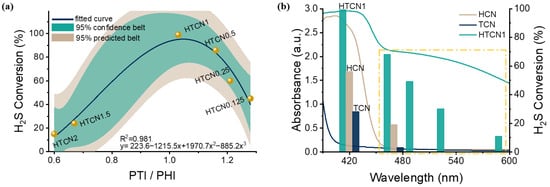
Figure 7.
(a) Correlation graphs of photocatalytic desulfurization activity of HTCNx (x = 0.125, 0.25, 0.5, 1, 1.5, and 2) with PTI/PHI (homojunction). (b) The wavelength-dependent H2S conversion of HCN, TCN, and HTCN1.
The positive contributions of the n→π* electron transition and the homojunction to enhancing photocatalytic activity were further confirmed by the wavelength-dependent H2S conversion experiments conducted on HCN, TCN, and HTCN1. As shown in Figure 7b, HTCN1 exhibited a significant improvement in H2S conversion within the 420–600 nm visible-light region compared to HCN and TCN. At 420 nm, the conversion efficiencies of hydrogen sulfide over HCN and TCN were below 60% and 30%, respectively, and no activity was observed beyond 480 nm (the yellow dashed box). Combined with the photoelectrochemical characterization, these results strongly demonstrate that the formation of the n→π* electron transition and the homojunction could markedly enhance the photocatalytic activity of carbon nitride in the visible-light region, especially at wavelengths greater than 500 nm.
The stability and reproducibility of the catalyst are key factors in the evaluation of the catalyst system. Structural characterization of the used catalysts confirmed that their core structure was retained after the reaction (Figure S9). The high-resolution S 2p XPS spectrum of the used HTCN1 was further analyzed, as shown in Figure S10. The two peaks correspond to the S 2p3/2 and S 2p1/2 orbitals of elemental sulfur (S0) [44]. The collected reaction products are consistent with the XRD pattern of elemental sulfur, demonstrating that HTCN successfully oxidized H2S to elemental sulfur (Figure S11). These results confirm that the catalytic system exhibits excellent selectivity. The deposition of reaction product S on the surface of the catalyst will damage the overall catalytic performance. The reacted samples were exposed to the N2 atmosphere and heat-treated at 200 °C for 2 h to remove the product attached to the surface. As can be observed from Figure S12, HTCN1 can still maintain almost the same conversion rate as the initial irradiation after four rounds of cyclic testing. It is suggested that continuous photocatalytic removal of H2S could be achieved by heat treatment of the post-reaction catalyst.
3. Materials and Methods
3.1. Materials
Detailed information on the materials is provided in the Supplementary Materials.
3.2. Synthesis of HCN (Heptazine-Based Carbon Nitride)
A total of 1 g of melamine was thoroughly ground with KCl (6.6 g) and LiCl (5.4 g). The mixture was then treated at 550 °C for 4 h with N2 at a flow rate of 500 mL/min. After cooling down to room temperature, the bulk was thoroughly washed with deionized water to remove the alkali metal salts. The obtained product is HCN. Melamine, KCl and LiCl were obtained from Sigma-Aldrich (Shanghai Macklin Biochemical Co., Ltd., Shanghai, China).
3.3. Synthesis of TCN (Triazine-Based Carbon Nitride)
In a glove box, 1 g of DCDA and 10 g of molten salt (KCl/LiCl eutectic mixture (6.6 g/5.4 g) were ground evenly and then transferred to a quartz tube. The mixture was placed in a muffle furnace and reacted at 400 °C in air for 4 h with N2 at a flow rate of 500 mL/min and a heating rate of 2 °C/min. After natural cooling to room temperature, we extracted the gas into the quartz tube and sealed it. Then, is the tube was placed in a muffle furnace for further heating at a rate of 2 °C/min. The reaction temperature was 550 °C and the reaction time was 4 h. After the reaction was completed, the product was allowed to cool to room temperature and then washed with deionized water. The obtained product is TCN. DCDA, KCl and LiCl were obtained from Sigma-Aldrich (Shanghai Macklin Biochemical Co., Ltd., Shanghai, China).
3.4. Synthesis of HTCN (Heptazine/Triazine Mixed-Phase Carbon Nitride)
HTCN photocatalysts were designed and synthesized by using the supramolecular molten salt method. As shown in Scheme S1, a certain amount of tricyanic acid (CA) and pentamino1h-tetrazolium (ATZ) were oscillated in 120 mL of deionized water at a certain molar ratio to form supramolecular precursors. Then, they were uniformly mixed with KCl/LiCl eutectic mixture (6.6 g/5.4 g), heated to 550 °C in a nitrogen tube furnace, and held for 4 h. Before collecting the final powder, the above-mentioned products were ground in mortar and washed with deionized water. The final products were denoted as HTCNx (x = 0.125, 0.25, 0.5, 1.0, 1.5, 2), where x represents the molar ratio of ATZ to CA. ATZ, CA, KCl and LiCl were obtained from Sigma-Aldrich (Shanghai Macklin Biochemical Co., Ltd., Shanghai, China).
3.5. Catalyst Characterization
Detailed information on the testing instruments and conditions is provided in the Supplementary Materials.
3.6. Photocatalytic Activity Test
Detailed information on the photocatalytic removal of hydrogen sulfide is provided in the Supplementary Materials.
4. Conclusions
In summary, this study developed a controllable synthesis strategy based on supramolecular self-assembly and salt melt crystallization, successfully preparing carbon nitride-based photocatalytic materials with tunable crystalline phase composition and distinct n→π* electron transition characteristics. The built-in electric field formed by the homojunction effectively promoted the separation of photogenerated charge carriers, while the structurally distorted-induced n→π* electron transition extended the light-response range into the near-infrared region (up to 700 nm). These features collectively enhanced the visible-light utilization efficiency and significantly improved the performance of photocatalytic oxidation removal of H2S. It achieved the ordered assembly and synergistic coupling of different functional units at the molecular scale, establishing a controllable preparation paradigm for rationally designing high-performance and multifunctional polymer photocatalysts, deepening the understanding of the structure–performance relationship of non-metallic catalysts at the microstructure and macroscopic levels, and providing important theoretical support and technical routes for the development of new photocatalytic materials for air pollution control and clean energy conversion.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/catal16030286/s1.
Author Contributions
Conceptualization, investigation, data curation, methodology, formal analysis, writing—original draft, Y.R.; writing—review and editing, data curation, R.X.; project administration, funding acquisition, validation, supervision, C.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the National Key Technologies R & D Program of China (2018YFA0209300), the National Natural Science Foundation of China (22272027, 52201006, U21A20326, and U24A20567), the Natural Science Foundation of Fujian Province (2022J01534), the Chang Jiang Scholars Program of China (T2016147), and the 111 Project (D16008). C.Y. also thanks the support from the Eyas Program of Fujian Province.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| PCN | Polymer carbon nitride |
| HCN | Heptazine-based carbon nitride |
| TCN | Triazine-based carbon nitride |
| HTCN | Heptazine/triazine mixed-phase carbon nitride |
| ATZ | 5-Aminotetrazole |
| CA | Cyanuric acid |
| PTI | Poly triazine imide |
| PHI | Poly heptazine imide |
| XRD | X-ray diffraction |
| FTIR | Fourier Transform infrared spectroscopy |
| XPS | X-ray photoelectron spectroscopy |
| SEM | Scanning electron microscope |
| TEM | Transmission electron microscope |
| DFT | Density functional theory |
| NMR | Nuclear magnetic resonance |
References
- Zheng, K.; Wu, M.; Zhu, J.; Zhang, W.; Liu, S.; Zhang, X.; Wu, Y.; Li, L.; Li, B.; Liu, W.; et al. Breaking the Activity–Selectivity Trade-off for CH4-to-C2H6 Photoconversion. J. Am. Chem. Soc. 2024, 146, 12233–12242. [Google Scholar] [CrossRef]
- Gunawan, D.; Zhang, J.; Li, Q.; Toe, C.Y.; Scott, J.; Antonietti, M.; Guo, J.; Amal, R. Materials Advances in Photocatalytic Solar Hydrogen Production: Integrating Systems and Economics for a Sustainable Future. Adv. Mater. 2024, 36, 2404618. [Google Scholar] [CrossRef]
- Zheng, K.; Liu, S.; Li, B.; Zhu, J.; Zhang, X.; Wu, M.; Li, L.; Zhu, S.; Liu, W.; Hu, J.; et al. Atomically Precise Pd Species Accelerating CO2 Hydrodeoxygenation into CH4 with 100% Selectivity. Precis. Chem. 2023, 1, 530–537. [Google Scholar] [CrossRef]
- Paraschoudi, E.N.; Bairamis, F.; Sygellou, L.; Andrikopoulos, K.S.; Konstantinou, I.; Tasis, D. Construction of Pd-Co-Doped CdS Heterojunctions as Efficient Platforms in Photocatalysis. Chem. A Eur. J. 2023, 29, e202300568. [Google Scholar] [CrossRef]
- Haider, Z.; Archana, R.; Ju, H. Recent Advancements in Photocatalytic Synthesis of Five Membered Nitrogen Heterocycles and Their Derivatives. Molecules 2025, 30, 3490. [Google Scholar] [CrossRef]
- Wang, S.; Xie, Z.; Zhu, D.; Fu, S.; Wu, Y.; Yu, H.; Lu, C.; Zhou, P.; Bonn, M.; Wang, H.I.; et al. Efficient Photocatalytic Production of Hydrogen Peroxide Using Dispersible and Photoactive Porous Polymers. Nat. Commun. 2023, 14, 6891. [Google Scholar] [CrossRef]
- Li, J.; Wu, D.; Iocozzia, J.; Du, H.; Liu, X.; Yuan, Y.; Zhou, W.; Li, Z.; Xue, Z.; Lin, Z. Achieving Efficient Incorporation of π-Electrons into Graphitic Carbon Nitride for Markedly Improved Hydrogen Generation. Angew. Chem. Int. Ed. 2019, 131, 2007–2011. [Google Scholar] [CrossRef]
- Khan, M.S.; Zhang, F.; Osada, M.; Mao, S.S.; Shen, S. Graphitic Carbon Nitride-Based Low-Dimensional Heterostructures for Photocatalytic Applications. Solar RRL 2020, 4, 1900435. [Google Scholar] [CrossRef]
- Wang, Y.; Du, P.; Pan, H.; Fu, L.; Zhang, Y.; Chen, J.; Du, Y.; Tang, N.; Liu, G. Increasing Solar Absorption of Atomically Thin 2D Carbon Nitride Sheets for Enhanced Visible-Light Photocatalysis. Adv. Mater. 2019, 31, 1807540. [Google Scholar] [CrossRef]
- Ran, J.; Guo, W.; Wang, H.; Zhu, B.; Yu, J.; Qiao, S. Metal-Free 2D/2D Phosphorene/g-C3 N4 Van Der Waals Heterojunction for Highly Enhanced Visible-Light Photocatalytic H2 Production. Adv. Mater. 2018, 30, 1800128. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, B.; Lin, S.; Zhang, Y.; Wang, X. Activation of n→π* Transitions in Two-Dimensional Conjugated Polymers for Visible Light Photocatalysis. J. Phys. Chem. C 2014, 118, 29981–29989. [Google Scholar] [CrossRef]
- Guseinov, I.I.; Mamedov, B.A. Accurate Evaluation of Overlap Integrals of Slater Type Orbitals with Noninteger Principal Quantum Numbers Using Complete Orthonormal Sets of Ψα-Exponential Type Orbitals. J. Math. Chem. 2008, 43, 1527–1532. [Google Scholar] [CrossRef]
- Öztekin, E. Overlap Integrals with Respect to Quantum Numbers over Slater-type Orbitals via the Fourier-transform Method. Int. J. Quantum Chem. 2004, 100, 236–243. [Google Scholar] [CrossRef]
- Hu, S.; Jiang, D.; Gu, L.; Xu, G.; Li, Z.; Yuan, Y. Awakening n→π* Electronic Transition by Breaking Hydrogen Bonds in Graphitic Carbon Nitride for Increased Photocatalytic Hydrogen Generation. Chem. Eng. J. 2020, 399, 125847. [Google Scholar] [CrossRef]
- Zhai, B.; Li, H.; Gao, G.; Wang, Y.; Niu, P.; Wang, S.; Li, L. A Crystalline Carbon Nitride Based Near-Infrared Active Photocatalyst. Adv. Funct. Mater. 2022, 32, 2207375. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Hu, Z.; Huang, J.; Yang, S.; Li, H. High-Efficiency Photocatalytic CO2 Reduction Enabled by Interfacial Ov and Isolated Ti3+ of g-C3N4/TiO2 Z-Scheme Heterojunction. J. Colloid Interface Sci. 2024, 663, 891–901. [Google Scholar] [CrossRef]
- Li, X.; Bai, J.; Li, J.; Li, C.; Zhong, X.; Deng, S. The Effect of n–π* Electronic Transitions on the N2 Photofixation Ability of Carbon Self-Doped Honeycomb-like g-C3N4 Prepared via Microwave Treatment. RSC Adv. 2020, 10, 7019–7025. [Google Scholar] [CrossRef]
- Tashiro, K. Structural Science of Crystalline Polymers: Basic Concepts and Practices; Springer Nature: Singapore, 2022. [Google Scholar]
- Ong, W.-J.; Tan, L.-L.; Ng, Y.H.; Yong, S.-T.; Chai, S.-P. Graphitic Carbon Nitride (g-C3 N4)-Based Photocatalysts for Artificial Photosynthesis and Environmental Remediation: Are We a Step Closer To Achieving Sustainability? Chem. Rev. 2016, 116, 7159–7329. [Google Scholar] [CrossRef]
- Lin, L.; Ou, H.; Zhang, Y.; Wang, X. Tri-s-Triazine-Based Crystalline Graphitic Carbon Nitrides for Highly Efficient Hydrogen Evolution Photocatalysis. ACS Catal. 2016, 6, 3921–3931. [Google Scholar] [CrossRef]
- Lin, L.; Lin, Z.; Zhang, J.; Cai, X.; Lin, W.; Yu, Z.; Wang, X. Molecular-Level Insights on the Reactive Facet of Carbon Nitride Single Crystals Photocatalysing Overall Water Splitting. Nat. Catal. 2020, 3, 649–655. [Google Scholar] [CrossRef]
- Cui, L.; Song, J.; McGuire, A.F.; Kang, S.; Fang, X.; Wang, J.; Yin, C.; Li, X.; Wang, Y.; Cui, B. Constructing Highly Uniform Onion-Ring-like Graphitic Carbon Nitride for Efficient Visible-Light-Driven Photocatalytic Hydrogen Evolution. ACS Nano 2018, 12, 5551–5558. [Google Scholar] [CrossRef]
- Du, X.; Zou, G.; Wang, Z.; Wang, X. A Scalable Chemical Route to Soluble Acidified Graphitic Carbon Nitride: An Ideal Precursor for Isolated Ultrathin g-C3N4 Nanosheets. Nanoscale 2015, 7, 8701–8706. [Google Scholar] [CrossRef]
- Zhao, T.; Zhang, S.; Guo, Y.; Wang, Q. TiC2: A New Two-Dimensional Sheet beyond MXenes. Nanoscale 2016, 8, 233–242. [Google Scholar] [CrossRef]
- Holst, J.R.; Gillan, E.G. From Triazines to Heptazines: Deciphering the Local Structure of Amorphous Nitrogen-Rich Carbon Nitride Materials. J. Am. Chem. Soc. 2008, 130, 7373–7379. [Google Scholar] [CrossRef]
- Lan, Z.; Fang, Y.; Zhang, Y.; Wang, X. Photocatalytic Oxygen Evolution from Functional Triazine-Based Polymers with Tunable Band Structures. Angew. Chem. Int. Ed. 2018, 57, 470–474. [Google Scholar] [CrossRef]
- Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A Metal-Free Polymeric Photocatalyst for Hydrogen Production from Water under Visible Light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef]
- Zhang, J.; Liang, X.; Zhang, C.; Lin, L.; Xing, W.; Yu, Z.; Zhang, G.; Wang, X. Improved Charge Separation in Poly(Heptazine-triazine) Imides with Semi-coherent Interfaces for Photocatalytic Hydrogen Evolution. Angew. Chem. Int. Ed. 2022, 61, e202210849. [Google Scholar] [CrossRef]
- Barrio, J.; Volokh, M.; Shalom, M. Polymeric Carbon Nitrides and Related Metal-Free Materials for Energy and Environmental Applications. J. Mater. Chem. A 2020, 8, 11075–11116. [Google Scholar] [CrossRef]
- Li, H.; Zhou, Y.; Tu, W.; Ye, J.; Zou, Z. State-of-the-Art Progress in Diverse Heterostructured Photocatalysts toward Promoting Photocatalytic Performance. Adv. Funct. Mater. 2015, 25, 998–1013. [Google Scholar] [CrossRef]
- Lan, Z.; Zhang, G.; Chen, X.; Zhang, Y.; Zhang, K.A.I.; Wang, X. Reducing the Exciton Binding Energy of Donor–Acceptor-Based Conjugated Polymers to Promote Charge-Induced Reactions. Angew. Chem. Int. Ed. 2019, 58, 10236–10240. [Google Scholar] [CrossRef]
- Bai, K.; Yu, X.; Wen, G.; Yang, Y.; Lin, Y.; Zhang, L.; Rong, J.; Yin, L.-C.; Qi, W.; Bonn, M.; et al. Spontaneous Dissociation of Excitons in Polymeric Photocatalysts for Overall Water Splitting. Nat. Commun. 2025, 16, 8577. [Google Scholar] [CrossRef]
- Li, F.; Tang, M.; Li, T.; Zhang, L.; Hu, C. Two-Dimensional Graphene/g-C3N4 in-Plane Hybrid Heterostructure for Enhanced Photocatalytic Activity with Surface-Adsorbed Pollutants Assistant. Appl. Catal. B Environ. Energy 2020, 268, 118397. [Google Scholar] [CrossRef]
- Cao, S.; Low, J.; Yu, J.; Jaroniec, M. Polymeric Photocatalysts Based on Graphitic Carbon Nitride. Adv. Mater. 2015, 27, 2150–2176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, X.; Takanabe, K.; Maeda, K.; Domen, K.; Epping, J.D.; Fu, X.; Antonietti, M.; Wang, X. Synthesis of a Carbon Nitride Structure for Visible-Light Catalysis by Copolymerization. Angew. Chem. Int. Ed. 2010, 49, 441–444. [Google Scholar] [CrossRef] [PubMed]
- An, S.; Zhang, G.; Li, K.; Huang, Z.; Wang, X.; Guo, Y.; Hou, J.; Song, C.; Guo, X. Self-Supporting 3D Carbon Nitride with Tunable n→π* Electronic Transition for Enhanced Solar Hydrogen Production. Adv. Mater. 2021, 33, 2104361. [Google Scholar] [CrossRef]
- Wang, K.; Fu, J.; Zheng, Y. Insights into Photocatalytic CO2 Reduction on C3N4: Strategy of Simultaneous B, K. Co-Doping and Enhancement by N Vacancies. Appl. Catal. B Environ. 2019, 254, 270–282. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, J.; Che, H.; Liu, B.; Ao, Y. n→π* Electron Transitions and Directional Charge Migration Synergistically Promoting O2 Activation and Holes Utilization on Carbon Nitride for Efficiently Photocatalytic Degradation of Organic Contaminants. Small 2023, 19, 2302510. [Google Scholar] [CrossRef]
- Xing, W.; Li, C.; Wang, Y.; Han, Z.; Hu, Y.; Chen, D.; Meng, Q.; Chen, G. A Novel 2D/2D Carbonized Poly-(Furfural Alcohol)/g-C3N4 Nanocomposites with Enhanced Charge Carrier Separation for Photocatalytic H2 Evolution. Carbon 2017, 115, 486–492. [Google Scholar] [CrossRef]
- Wang, W.; Shan, G. Exact Confidence Intervals for the Relative Risk and the Odds Ratio. Biometrics 2015, 71, 985–995. [Google Scholar] [CrossRef]
- Shen, L.; Lei, G.; Fang, Y.; Cao, Y.; Wang, X.; Jiang, L. Polymeric carbon nitride nanomesh as an efficient and durable metal-free catalyst for oxidative desulfurization. Chem. Commun. 2018, 54, 2475–2478. [Google Scholar] [CrossRef]
- Katayama, T.; Nagata, M. Photocatalytic Gas-Phase Hydrogen Sulfide Removal Using Mo Cocatalyst: Implementation of Counter-Poisoning Photocycle. ACS Omega 2025, 10, 4670–4678. [Google Scholar] [CrossRef]
- Deng, A.; Sun, Y.; Gao, Z.; Yang, S.; Liu, Y.; He, H.; Zhang, J.; Liu, S.; Sun, H.; Wang, S. Internal electric field in carbon nitride-based heterojunctions for photocatalysis. Nano Energy 2023, 108, 108228. [Google Scholar] [CrossRef]
- Chen, Y.; Tong, S.; Li, W.; Liu, Y.; Tan, F.; Ge, M.; Xie, X.; Sun, J. Photocatalytic Oxidation of SO2 by TiO2: Aerosol Formation and the Key Role of Gaseous Reactive Oxygen Species. Environ. Sci. Technol. 2021, 55, 9784–9793. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.