1. Introduction
Nitrogen oxides (NO
x) are one of the main air pollutants that form during fuel burning. Their Selective Catalytic Reduction (SCR) with ammonia is applied to remove them from off-gases from the stationary sources of emission [
1]. During this process, nitrogen and water are expected products like in the following reactions:
Unfortunately, nitrous oxide is often formed as well, according to the reaction equations:
This greenhouse gas influences the global warming about 300 hundred times stronger than carbon dioxide. Its lifetime in atmosphere is ca. 180 years and can be decomposed only in the stratosphere according to the reaction equation:
Ozone is taken from the ozone layer damaging Earth’s protection [
2]. The amount of nitrous oxide emitted from anthropogenic sources has increased and now is assessed as 40% of total N
2O emission [
3]. Therefore, in order to design catalysts for SCR of NOx, it is important to remember their high selectivity to avoid forming this by-product. Such catalysts can be obtained by the deposition of an active phase on a support or by its coprecipitation with metal ions improving its properties.
Coprecipitation is regarded as a simple synthesis method of industrial catalysts [
4]. The oxide catalysts obtained by this method turned out to be efficient in SCR of NO
x due to the good dispersion of an active phase, its high concentration [
5], as well as a strong interaction between active metals [
6]. In such systems, aluminum oxide has often been the dispersive phase. Moreover, its acidic properties also improve resistance to sulfur poisoning [
6]. The aluminum oxide often crystallizes with other metals in the spinel structure [
5,
7]. It was suggested that the spinel structure can be responsible for the high catalytic activity of copper–aluminum oxide systems in SCR of NO [
7]. It is related to the large adsorption of NO due to the high number of oxygen vacancies [
1,
6]. The spinel composition can be designed in such a way to obtain the catalyst active in a broad temperature range of 100–400 °C [
8]. In automotive catalysts, the alumina support is promoted with ceria, which can store oxygen, develop their surface area, and improve thermal stability. The key factor in the CeO
2 efficiency is its dispersion [
4]. The lattice oxygen of ceria takes part in the oxidation reaction, and the reduced oxide is reoxidized by NO or N
2O with the formation of nitrogen without N
2O production [
1]. CeO
2 inhibits also the formation of NH species, which are intermediates of N
2O [
6]. Cerium and copper can form a redox system which inhibits SO
2 oxidation or can form sulfates protecting the active copper centers and can accelerate its decomposition as well [
6]. Ceria also inhibits the formation of ammonium nitrate and protects the catalyst from the alkali poisoning providing the Lewis acid sites for NH
3 activation [
6].
The aim of the presented paper was to obtain a copper aluminum spinel catalyst and then to modify its catalytic properties by substituting a part of copper by cerium and/or manganese. The relationship between the sample catalytic activity in SCR of NO with ammonia and the amount of cerium was examined. Factors determining the catalytic activity were identified by a physicochemical characterization of solids.
2. Results and Discussion
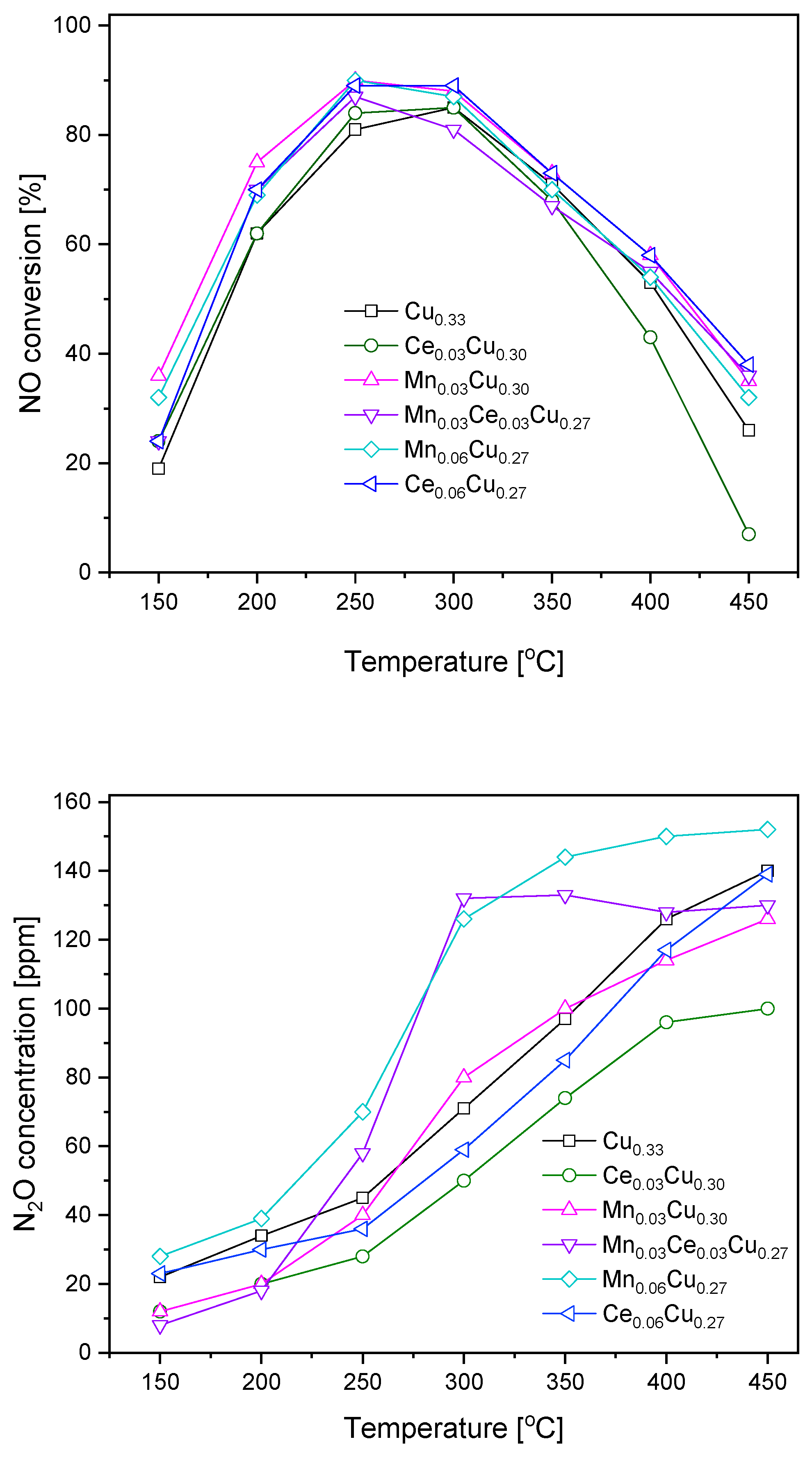
Obtained copper–aluminium oxide and its derivatives doped with cerium and manganese (0.03 and 0.06 atomic ratio) were subjected to catalytic tests in SCR of NO with ammonia (
Figure 1). For all samples, the NO conversion was in the range 20–40% at 150 °C, attained its maximum at 250 or 300 °C, and decreased below 40% at 450 °C. Over the Cu
0.33 sample, the maximum of NO conversion of ca. 85% was observed at 300 °C. The replacement of copper by cerium in the Ce
0.03Cu
0.30 sample caused a slight increase in the catalytic efficiency at lower temperatures (150–250 °C) and a more distinct decrease above 400 °C. The analogous substitution with manganese brought about an increase in NO conversion in the whole temperature range. The mixed Mn
0.03Ce
0.03Cu
0.27 sample was less efficient than that with manganese but slightly better than with cerium one, particularly at higher temperatures of 400–450 °C. The increase in manganese content did not strengthen the catalytic efficiency. In contrast, the same increase in the cerium concentration caused improvement in the catalyst activity making the Ce
0.06Cu
0.27 sample only slightly less active than the Mn
0.03Cu
0.30 sample at low temperatures of 150–200 °C.
During the catalytic tests, the concentration of N
2O forming the undesired by-product was measured as well (
Figure 1). Over the undoped catalyst, the concentration of N
2O achieved 45 ppm at 250 °C and started to rise more rapidly above this temperature reaching a level of 140 ppm at 450 °C. The admixture of Ce resulted in significant lowering of N
2O formation, namely, at 300 °C, the N
2O concentration exceeded the detection limit of 30 ppm and reached 100 ppm at 450 °C. In the temperature range of 300–350 °C, the presence of manganese increased the formation of by-product in comparison to the Cu
0.33 sample; however, at higher temperatures (400–450 °C), the opposite was true. The equimolar Ce
0.03Mn
0.03Cu
0.27 catalyst caused the formation of 60 ppm of nitrous oxide at 250 °C, while above 300 °C, the concentration of N
2O was constant at the level of 130 ppm. The increase of the Mn amount resulted in the highest N
2O concentration in the whole temperature range. In the case of higher cerium content, the amount of N
2O formed was about 10 ppm higher than over the Ce
0.03Cu
0.30 sample up to 350 °C.
To determine the crystallographic structure of catalysts, powder XRD patterns were registered as seen in
Figure 2. For the Cu
0.33 sample, the diffraction lines at a 2θ angle of ca. 19.0, 31.2, 36.9, 44.9, 49.2, 55.8, 59.4, 65.2, 74.2, 77.4, 78.5, and 82.7° can be ascribed to (111), (220), (311), (400), (331), (422), (511), (440), (620), (533), (622), and (444) planes of CuAl
2O
4 spinel, respectively [01-071-0967]. Main reflections are narrow, which suggests that crystallites were rather large. Their average size was estimated based on the (311) plane from the Scherrer equation as equal to 31 nm (
Table 1). After an admixture of cerium, new reflections at a 2θ angle of ca. 28.6, 33.1, 47.5, 56.3, 69.5, 76.7, and 88.5° appear, which origin from (111), (200), (220), (311), (400), (331), and (422) planes of CeO
2, respectively [03-065-2975]. At the same time, the average crystallite size decreased to 21 nm. The double increase in the cerium content did not change the CeO
2 crystallite size (calculated for the (111) plane) in contrast to a noticeable decrease of spinel crystallite size to 18 nm (
Table 1). In the case of manganese doping, only reflections from the spinel phase are present. It suggests that Mn formed a solid solution with copper–aluminum oxide in the spinel structure or an amorphous oxide. Manganese admixture caused a decrease of the average crystallite size to 18 nm in the Mn
0.03Cu
0.30 sample and to 21 nm in the Mn
0.06Cu
0.27 one. The doubly doped sample Ce
0.03Mn
0.03Cu
0.27 crystallized in the spinel structure, although some cerium oxide was also detected. It seems that manganese ions have built into the spinel phase as well. The ceria crystallites were about 12 nm large and the spinel ones were ca. 20 nm.
The chemical composition and textural properties of oxide catalysts are compared in
Table 1. The measured metal contents for the first six samples were very close to the intended ones. The Cu
0.33 sample exhibited a SSA
BET of 19 m
2 g
−1 and a total pore volume of 0.445 cm
3 g
−1. The replacement of some copper by cerium or manganese resulted in an increase in the specific surface area and a decrease of pore volume. The doping with Ce (0.03) caused almost 50% growth in SSA
BET, whereas the same amount of Mn brought about almost a double increase in this value. The pore volume decreased above two times. Further increase in Mn and Ce content (to 0.06) did not result in a change of SSA
BET. The total pore volume decreased with manganese amount and increased with cerium concentration.
Taking into account that the Ce
0.06Cu
0.27 sample was only slightly less catalytically active than the Mn
0.03Cu
0.30 one at lower temperatures, the increase in Mn content did not improve catalytic efficiency, and less N
2O was formed over cerium containing catalysts. We prepared some additional samples with a higher content of Ce. Over such catalysts, NO conversion increased with the cerium content; simultaneously, N
2O formation grew (
Figure 3). The NO conversion at 150 °C was an exception where the sequence of catalyst activity was different. Below 250 °C, the concentration of nitrous oxide changed from 40 to 50 ppm. Above this temperature, the growth was faster and linearly achieved ca. 150 ppm at 450 °C. The increase in nitrous oxide formation with Ce content was small for higher cerium concentrations. NO conversion and N
2O formation over the Ce
0.09Cu
0.24 catalyst did not follow that trend. The Ce
0.15Cu
0.18 sample exhibited the highest catalytic activity in the whole range of tested temperatures attaining the maximum at 250–300 °C and a NO conversion above 80% in the temperature range of 200–350 °C, this activity is worse at higher temperatures than observed for the zeolite catalyst containing copper—Cu-SSZ-13, which achieved such activity in the range of 175–450 °C [
9]. The comparable efficiency was attained for copper–alumina coprecipitated in the presence of carbon nanotubes—CuAlLDOCNT, NO conversion higher than 80% was measured for the temperature range of 180–330 °C [
10]. For this catalyst, selectivity to N
2 was about 95%. While for the zeolite materials, N
2O yield, calculated as Y
N2O = (2xc
N2O)/(c
NOin + c
NH3in), was ca. 6–10% [
9]. We estimated this value for our catalyst in the range of 7–11%.
As seen from
Table 1 and
Figure 4, the average size of spinel crystallites decreased with the cerium content. Much weaker growth of spinel crystallites in the presence of ceria was described earlier [
11]. On the other hand, the size of CeO
2 crystallites remained almost the same. Only for the Ce
0.15Cu
0.18 sample a small increase in the crystallites size was observed. The metal concentration for the Ce
0.09Cu
0.24 and Ce
0.12Cu
0.21 samples were very similar, which could result from an experimental error during synthesis. The measured composition of the Ce
0.15Cu
0.18 catalyst was close to intended. The specific surface area lowered for the Ce
0.09Cu
0.24 sample to ca. 25 m
2 g
−1, and for the Ce
0.12Cu
0.21, catalyst remained similar as for the Ce
0.06Cu
0.27 sample. The total pore volume did not change significantly. The catalyst with the highest cerium content exhibited the largest SSA
BET and a pore volume ca. 44 m
2 g
−1 and 0.348 cm
3 g
−1, respectively (
Table 1).
To shed some light on the catalytic activity of copper aluminum spinel and the role of Ce dopant, the XPS measurements were taken for parent Cu
0.33 and samples with various Ce contents before and after SCR of NO. The surface concentration of cerium and copper was calculated from the XPS survey spectra (
Table 2). All samples were analyzed in detail in O 1s, C 1s, Al 2p, Cu 2p, and Ce 3d regions.
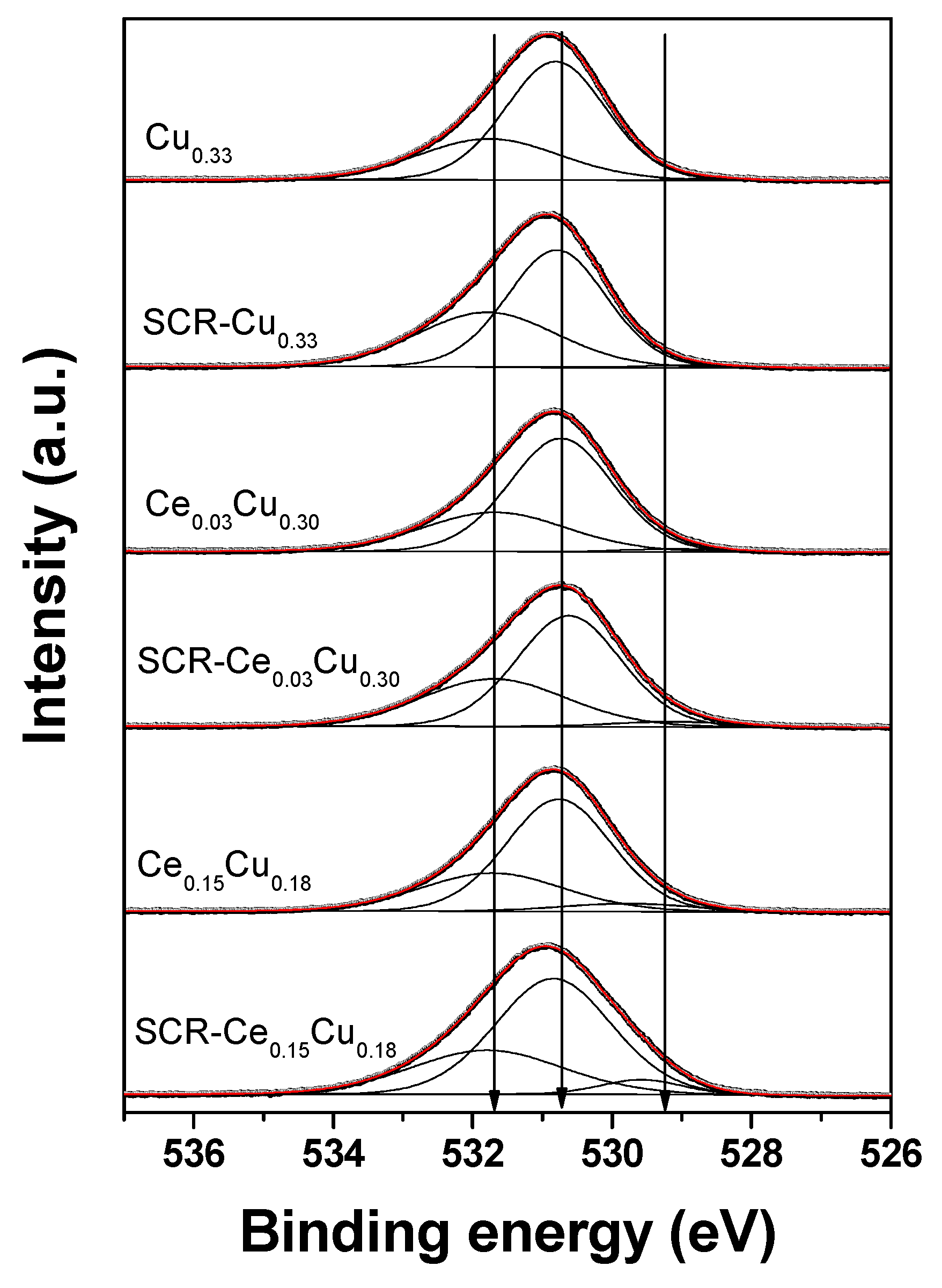
The O 1s spectra (
Figure 5) were well-decomposed into three components: (i) a most intense peak (>60%) located at 530.6–530.8 eV referred to the O
2− ions in the matrix of CuAl
2O
4 [
12]; (ii) a small peak (<3%) at 528.5–529.8 eV due to oxygen–metal bonds (CuO (528.6 eV–529.8 eV) [
13,
14], CeO
2 (528.7–529.2 eV) [
15,
16], Al
2O
3 (528.3–528.7 eV)) [
17]; (iii) a peak at BE of 531.7–531.8 eV assigned to OH groups, adsorbed water, and oxygen of organic contaminants. It can be seen from
Table 3 that there are some small differences (<5%) of relative amount of oxygen species between the fresh and used samples. However, one cannot exclude that these differences could be larger, but samples after the SCR reaction were stored under air atmosphere and could reoxidized before XPS studies.
Three peaks at 285.0 eV (organic contaminants), 285.8–286.0 eV (C–O groups), and 289.2–289.5 eV (O–C=O groups) can be distinguished in the C 1s spectra (
Figure 6). The hydrocarbon contamination was used as an internal calibration for our insulating samples, as we mentioned below. The binding energy of the Al 2p
3/2 core line is close to 74.1 eV suggesting Al
3+ in tetrahedral sites of the spinel structure [
12,
18].
Three doublets were used to properly fit the Cu 2p spectra (
Figure 7 and
Table 4). The spin-orbit splittings of all doublets were in the range of 19.7–19.9 eV. The weakest doublet (<5%) with Cu 2p
3/2 BE values of 930.7–931.2 eV is associated with the presence of reduced Cu
+ and/or Cu
0 [
12,
19]. Such a component can be related to the limited reduction of highly dispersed small copper particles under high-vacuum conditions or to the differential charging caused by some structural damages in the spinel structure. Some additional clue to distinguish between Cu
+ and Cu
0 species can be drawn from the copper Wagner plot [
20]. The kinetic energy of maximum of Cu L
3M
45M
45 Auger peak was read from the survey spectra, whereas binding energy of low-energy component from the Cu 2p high-resolution spectra. The copper Auger parameter for such contribution is below 1847.6 eV for all measured samples. Thus, based on the copper Wagner plot, we can expect that a low-energy component is related more to Cu
0 than Cu
+ [
20]. However, one can point out that such a procedure is a major approximation only and results should be treated with caution. The most intense components origin from the octahedral Cu
2+ (932.7–932.9 eV) [
21] and tetrahedral Cu
2+ species (933.9–934.5 eV) [
19] in Cu–Al spinel structure and/or CuO particles. The main photoelectron Cu 2p peaks are accompanied by strong shake-up satellites confirming that copper is present in all samples as Cu
2+ species mainly.
In the parent Cu
0.33 sample, copper was observed in the concentration twice lower than intended in the synthesis. The majority of Cu
+2 species occupied tetrahedral sites in the amount ca. twice higher than found in our recent paper [
22]. The ratio of tetrahedral to octahedral copper cations was close to typical of bulk partially inverse CuAl
2O spinel (60:40) [
23]. In copper–alumina systems, the spinel phase was the only one observed [
11], and in the Cu
0.33 sample, the reduced Cu
+ cations can occur in octahedrons which favor forming such species [
23]. The SCR conditions resulted in a small decrease (10%) in the presence of Cu
2+ cations in octahedral sites as well as in the amount of reduced copper. Cerium addition caused a significant growth of the octahedral component mostly for Ce
0.15Cu
0.18 (over 63%). It was shown [
23] that Cu
2+ ions in the octahedral positions are important in catalytic reactions due to undergoing Cu
2+ → Cu
+ transitions. Moreover, the reduced copper is crucial in dissociative NO adsorption [
11].
The Ce 3d XPS spectra of Ce-doped Cu–Al catalysts as received and after SCR are presented in
Figure 8. According to the common approach [
24,
25,
26,
27], Ce 3d spectra can be fitted with eight or ten components marked ’v’ for Ce 3d
5/2 and ‘u’ for Ce 3d
3/2 multiplets. The multiplicity of these states comes from different Ce 4f level occupancies in the final state. Here, we have chosen 10-components approach; however, v
0 and u
0 peaks are difficult to resolve because the energy separation with v and u peaks is small. The peaks at 882.3 (v), 889.1 (v’’), and 898.4 eV (v’’’) as well as 900.9 (u), 907.6 (u’’), and 916.7 eV (u’’’) can be assigned to Ce
4+, whereas 880.4 (v
0), 884.5 (v’), 896.9 (u
0), and 902.9 eV (u’) to Ce
3+. The position of the Ce
4+ reference line (u’’’) was 916.7 eV which is in perfect agreement with literature data [
28]. The surface concentration of Ce
3+ was estimated from the Ce
3+/(Ce
3+ + Ce
4+) peak areas ratio [
29]. Because Ce 3d spectra are quite similar to reference CeO
2 spectrum [
30], a Ce
3+ percentage of 29% (Ce
0.03-SCR), 43% (Ce
0.03), 36% (Ce
0.15-SCR), and 30% (Ce
0.15) found in our samples is very close to pure CeO
2 [
20], and is connected with the copper content on the surface as expected. However, these values are much higher than found in Cu/CeO
2 [
19]. The presence of trivalent Ce
3+ is closely related to the oxygen vacancies in the spinel structure, and as a result, influences the redox properties and catalytic performance of Ce-based systems. The vicinity of ceria is necessary for Cu
+ ions to take part in redox reactions. On the other hand, the presence of cerium retards the sintering of the Cu particles and ensures its good dispersion [
11]. In the case of the Ce
0.03Cu
0.30 sample, the concentration of copper was retained at the surface, although its amount was decreased by 10% compared to the Cu
0.33 sample. The same ratio of the surface copper amount to the intended Cu concentration was observed for the Ce
0.15Cu
0.18 sample. Copper covers preferentially ceria [
11] like in the Ce
0.15Cu
0.18 sample, where the ratio of Ce/Cu was twice lower than intended. The amount of reduced copper increased significantly after the SCR reaction, which could be the result of reducing conditions and the presence of cerium, because it was not observed for copper–aluminum oxide. During catalytic tests of Ce-containing samples, the concentration of copper in tetrahedral sites increased by about 25% at the cost of octahedral Cu, not observed to such an extent for the Cu
0.33 sample. This tetrahedral copper could form CuO [
11] and pure Al
2O
3 spinel as observed in the Ce
0.15Cu
0.18 sample. In such copper–ceria–alumina systems, dissociative adsorption of NO and surface mobility of N atoms increase which can result in higher efficiency in SCR [
11] observed for the Ce
0.15Cu
0.18 sample.
3. Materials and Methods
Samples were obtained by coprecipitatation of metal nitrates with ammonium carbonate. The copper to aluminium ratio was equal to 1:2—typical of spinel compounds. The 1M solutions of copper (Cu(NO3)2·3H2O) and aluminum (Al(NO3)3·9H2O) nitrates were mixed and dropped into a beaker simultaneously with the 1M solution of (NH4)2CO3 taken in a 20% excess to a stoichiometric amount. The solutions were added at a rate of 4 mL min−1 under stirring and the obtained precipitation was aged at 50 °C for one hour. Then, the solid was filtered with distilled water and dried overnight at room temperature. The sample was calcined at 900 °C for 6 h. The sample was marked as Cu0.33. Next, samples were promoted with manganese or cerium and prepared analogously using Ce(NO3)3·6H2O and Mn(NO3)2·4H2O as substrates. The amount of copper was lowered and another metal was added to obtain the samples with the Cu:Ce(Mn) ratio equal to 0.30:0.03 and 0.27:0.06, the sample where Cu:Ce:Mn was 0.27:0.03:0.03 was prepared as well. They were denoted as Ce0.03Cu0.30, Ce0.06Cu0.27, Mn0.03Cu0.30, Mn0.06Cu0.27, and Ce0.03Mn0.03Cu0.27. The samples with higher cerium concentrations—Cu:Ce equal to 0.24:0.09, 0.21:0.12, and 0.18:0.15—were synthesized and marked as Ce0.09Cu0.24, Ce0.12Cu0.21, and Ce0.15Cu0.18.
The samples were subjected to catalytic tests in selective catalytic reduction of NO with ammonia. The concentrations of NO and NH3 were equal to 800 ppm and the oxygen concentration was 3.5%. These reagents were diluted in helium and passed at a rate of 100 mL min−1 through a quartz reactor with 0.2 g of a catalyst. Its 0.25–1 mm fraction was placed on quartz wool. The concentrations of NO and N2O were measured in the temperature range of 150–450 °C at a step of 50 °C with the IR analyzers—serie 2020 A0, produced by ABB.
The crystallographic structure of the samples was determined with the powder X-ray diffraction. XRD patterns were recorded at 2θ angles from 10 to 90°, with a step of 0.013° by a PANalytical-Empyrean diffractometer. As a radiation source, the copper lamp was used with λ = 1.5406 Å.
To perform the quantitative analysis of the metals contained in the materials, the XRF method was used. An EDX 3600H apparatus from Skyray Instruments with a tungsten lamp enabled probing with the depth larger than 50 μm.
The textural properties of samples were examined by the low temperature sorption of nitrogen. The isotherms were registered within the relative pressure range of 0–0.98 with a Gemini V 2.00 2380 (Micromeritics) analyzer. The specific surface area was measured using the BET method—SSABET. The total pore volume was assessed with the BJH model from the desorption branch.
Cu–Al spinel (Cu0.33) and the samples with minimum and maximum cerium concentrations (Ce0.03Cu0.30 and Ce0.15Cu0.18) were subjected to surface analysis with X-ray Photoelectron Spectroscopy (XPS). The experiments were performed with a SES R4000 hemispherical analyzer (Gammadata Scienta) and non-monochromatic MgKα X-ray radiation (1253.6 eV). The X-ray anode operated at 12 kV and 15 mA. For pass energy of 100 eV, the system resolution (FWHM) measured for the Ag 3d5/2 line was 0.9 eV. The spectrometer calibration was conducted according to ISO 15472:2001. During the experiment, the pressure in the analysis chamber was close to 3 × 10−10 mbar. Samples in the powder form were pressed into In foil and seated on a special holder. The analysis area was about 4 mm2 (5 × 0.8 mm). The survey scans were obtained at pass energy of 200 eV (with 250 meV step), whereas the high-resolution spectra were obtained at a pass energy of 100 eV (with 25 meV step). The spectra deconvolution into individual components were performed with using the CasaXPS 2.3.19 software. We used a Shirley-type background algorithm and pseudo-Voigt line profiles (mixture of Gaussian and Lorentzian lines) with 70:30 variable proportions. The charging was corrected to the carbon C 1s line at 285.0 eV.