Simple Summary
Pseudomyxoma peritonei is a rare condition in which mucinous tumor cells spread throughout the abdomen, most often originating from a ruptured mucinous tumor of the appendix. The principal therapeutic approach, combining cytoreductive surgery to remove visible disease with hyperthermic intraperitoneal chemotherapy, has greatly improved patient survival. However, recurrence remains a major concern, making careful selection of patients essential. This meta-analysis synthesizes the role of prognostic clinical-pathological, (immuno-) histological, and molecular parameters in pseudomyxoma peritonei patients undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. The findings emphasize the importance of using detailed tumor and patient characteristics to guide personalized, multidisciplinary treatment decisions. Looking ahead, future research should integrate molecular and genetic biomarkers to further improve patient selection and optimize treatment results for pseudomyxoma peritonei patients.
Abstract
Background/Objectives: While cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) significantly improve the outcome of pseudomyxoma peritonei (PMP) patients, recurrence remains a concern, highlighting the need for careful patient selection. This systematic review and meta-analysis evaluated the clinical-pathological, (immuno-) histological, and molecular prognostic factors in PMP patients undergoing CRS and HIPEC. Methods: Databases including PubMed, MEDLINE, Cochrane library, Embase, Scopus, and Web of Science were searched up to July 2024. Studies assessing clinical-pathological, (immuno-) histological, and molecular parameters influencing overall, progression-free, and/or disease-free survival were included. Statistical analysis was performed using R (Version 4.4.3) with random-effect models employed to address heterogeneity. Subsequent subgroup analyses were conducted to investigate potential sources of publication bias (Egger test). Results: A total of 16 studies involving 4009 PMP patients were included. Age, sex (male), completeness of cytoreduction score, peritoneal cancer index, tumor markers (CEA, CA 19-9, CA-125), tumor grade (high), histological subtype, and Ki-67 were significantly associated with prognosis prediction. Conclusions: Despite the presence of heterogeneity and publication bias, clinical-pathological and (immuno-) histological parameters were significantly associated with prognosis in PMP. The findings highlight the importance of integrating clinical-pathological and (immuno-) histological parameters in the individualized multidisciplinary management of PMP patients undergoing CRS and HIPEC. Future research should focus on incorporating molecular and genetic biomarkers to further refine patient selection and improve treatment outcomes.
1. Introduction
Pseudomyxoma peritonei (PMP) is characterized by the accumulation and extensive colonization of mucinous tumor cells, frequently resulting from a perforated mucinous appendiceal tumor [1,2].
PMP is a rare disease with a recently estimated incidence of 3.2 per million and a prevalence of 22 per million in Europe [3]. Females are affected two to three times more frequently than males [4,5,6]. Although the intra-abdominal mucinous tumor masses may vary among different patients, without a therapeutic surgical approach, progressive intestinal obstruction, followed by nutritional deficiencies, will inevitably lead to the patient’s death.
Cytoreductive surgery (CRS) combined with hyperthermic intraperitoneal chemotherapy (HIPEC) is regarded as the principal therapeutic approach, significantly improving long-term prognosis in case of complete cytoreduction (completeness of cytoreduction score CC0 or CC1) [7,8,9,10,11]. CRS-HIPEC significantly improves the overall survival (OS) of PMP patients up to 196 months, with 5- and 10-year survival rates reaching up to 74% and 63%, respectively [12]. However, approximately one-quarter to nearly half of patients experience tumor recurrence and progression following CRS-HIPEC [13,14,15,16,17]. In such instances, the efficacy of repeat CRS-HIPEC is often limited, and not all patients derive benefit from the procedure, thereby necessitating meticulous patient selection. Furthermore, CRS-HIPEC is a multifaceted surgical intervention characterized by extensive resection and intricate reconstructive procedures, resulting in prolonged operative durations and potential adverse events, including intra-abdominal infections, renal and hematologic toxicity, and thromboembolic events [18,19,20]. The major complication rate is 20–34%, while perioperative mortality is reported between 1.5% and 4% in patients with PMP undergoing CRS-HIPEC. However, outcomes may vary based on patient selection and center experience [7,12]. Notably, significant postoperative complications are associated with diminished OS and disease-free survival (DFS) rates, as well as extended hospital stays [21,22].
To date, individual small-scale retrospective analyses have identified certain risk factors associated with OS, progression-free (PFS), and DFS in PMP patients undergoing CRS and HIPEC.
In the dynamic field of personalized multimodal oncological treatment, the identification of predictive clinical-pathological, (immuno-) histological, and molecular markers is crucial for effective patient selection, particularly for those who may benefit from CRS-HIPEC. This systematic review and meta-analysis aimed to synthesize and evaluate the current evidence of prognostic clinical-pathological, (immuno-) histological, and molecular parameters in pseudomyxoma peritonei patients undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy, thus guiding clinical decision-making.
2. Materials and Methods
A systematic literature review was performed in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [23]. The study protocol has been registered in the PROSPERO international prospective register of systematic reviews (PROSPERO 2025 CRD420251148221).
2.1. Search Strategy
The literature search was conducted by two independent reviewers. The electronic databases PubMed, MEDLINE, Cochrane library, Embase, Scopus, and Web of Science were included for literature search. The search strategy included the following key words in adaptation to the specific syntax of each database: (HIPEC AND prognosis AND biomarker) OR (HIPEC AND prognosis AND histochemistry) OR (HIPEC AND prognosis AND immunohistochemistry) OR (HIPEC AND prognosis AND molecular pathology).
The search was performed from 1 October 2023 to 30 July 2024. Additionally, a snowball search of the included references was conducted [24]. Initially, titles and abstracts were screened by two independent reviewers according to the predefined inclusion criteria. Subsequently, a full-text evaluation was conducted, and data were extracted. In total, 16 studies were identified and included in the analysis (Table 1).

Table 1.
Basic clinical characteristics of the included studies of the meta-analysis.
2.2. Inclusion and Exclusion Criteria
Following an initial literature screening of studies concerning patients with peritoneal malignancies undergoing CRS and HIPEC, only those research papers that met specific inclusion criteria were incorporated into this systematic review. The inclusion criteria were as follows: retro- and prospective studies involving patients with histologically confirmed PMP; studies evaluating the prognostic value of clinical-pathological, (immuno-) histological, and molecular markers on CRS and HIPEC efficacy based on OS, PFS, and DFS; and studies reporting survival outcomes with hazard ratios (HRs), 95% confidence intervals (CIs). The exclusion criteria encompassed studies involving tumors other than PMP, studies not explicitly assessing the impact of prognostic factors on CRS and HIPEC efficacy, studies lacking survival outcomes and adequate statistical data (e.g., HRs), and studies that did not involve surgical treatment based on the combined approach of CRS and HIPEC. Non-English articles, conference papers, unpublished works, and animal studies were also excluded from this systematic review.
2.3. Data Extraction
Data were extracted from each article by two independent reviewers, encompassing the following variables: The first author’s name, publication year, country, study design, sample size, patients’ baseline characteristics, and clinical-pathological, (immuno-) histological, and molecular factors associated with OS, PFS, and/or DFS prediction, as well as reported HRs, 95% CIs, and p-values. In instances of discrepancies, a third reviewer was consulted to reach a final decision and achieve consensus. If multiple HRs were reported for the same parameter, the HR corresponding to the highest-risk category (the most clinically relevant contrast) was selected for statistical analysis; this situation occurred in only one study [25].
2.4. Outcome
The primary objective was to assess the impact of clinical-pathological, (immuno-) histological, and molecular parameters influencing OS, PFS, and/or DFS in patients suffering from PMP undergoing CRS and HIPEC.
2.5. Statistical Analysis
Statistical analysis was performed using R (Version 4.4.3) with the metafor package [41]. For statistical analysis, all parameters influencing OS, PFS, and/or DFS were categorized as follows: age, CC score, sex, PCI, performance status, symptoms, therapy, tumor marker (category: clinical-pathological); grading, histology, and immunohistochemistry (category: [immuno-] histological); and molecular pathology (category: molecular).
Statistical Heterogeneity and Publication Bias
Survival data were analyzed utilizing HRs and their corresponding 95% CIs through multivariate regression techniques. Statistical heterogeneity among studies was assessed using I2 statistics, with thresholds for low, moderate, and high heterogeneity set at 25%, 50%, and 75%, respectively [42]. To accommodate potential variability across studies, a random-effects model and Restricted Maximum Likelihood (REML) were consistently utilized, irrespective of the degree of heterogeneity. This approach was adopted to more accurately capture the true variations among studies and to extend our conclusions to a broader patient population. Furthermore, to obtain a more conservative estimate of the effect size, the Hartung–Knapp–Sidik–Jonkman (HKSJ) adjustment was applied for indicators with greater heterogeneity to correct the test statistics and CIs.
Subsequent subgroup analyses were conducted to investigate potential sources of publication bias. The Egger test, as an indicator of funnel plot asymmetry, was employed for each category to assess the correlation between the effect estimate and its variance, with a p value of less than 0.1 indicating a significant difference [43]. Statistical significance was set at a threshold of p < 0.05. The trim-and-fill method was applied as a sensitivity analysis to further estimate the potential impact of publication bias and to provide bias-adjusted pooled estimates.
2.6. Methodological Quality and Grade of Evidence Assessment
The quality and the grade of evidence of the included studies were assessed by two independent investigators based on the Newcastle Ottawa Scale (NOS) [44] and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) Working Group grades of evidence (high, moderate, low, very low quality) [45], respectively. Concerning NOS scaling, all studies were evaluated based on patient selection, comparability of cohorts based on the design or analysis, and assessment of measured outcomes. The studies were then rated as high (7–9), moderate (4–6), or low quality (0–3).
3. Results
3.1. Study Selection Process and Characteristics of Included Studies
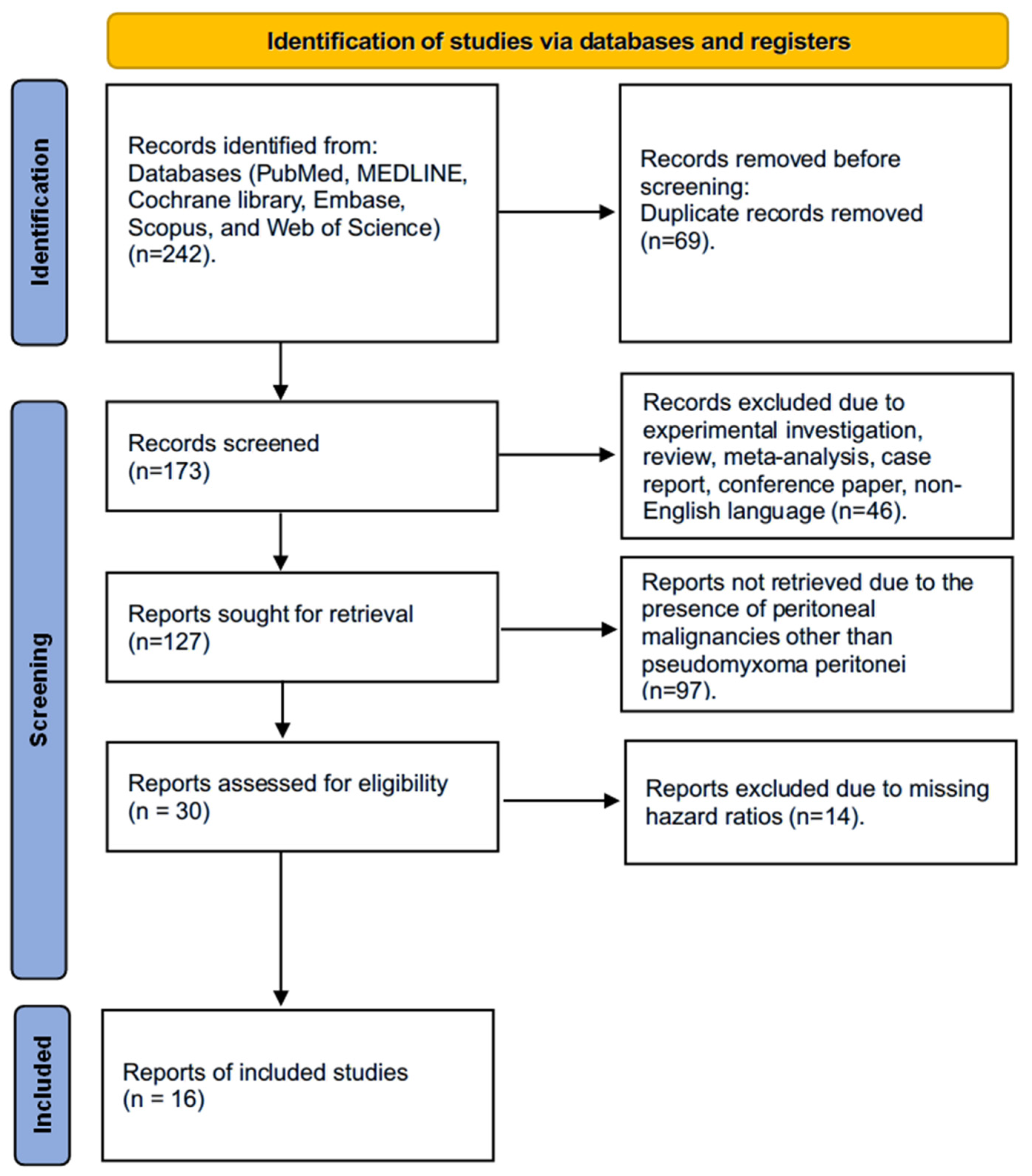
We identified 242 studies using the search strategy. Finally, this meta-analysis includes a total of 4009 patients diagnosed with PMP, derived from 16 studies published between 2002 and 2023, following the application of inclusion criteria. The detailed PRISMA flowchart illustrating the article selection process is presented in Figure 1. Excluded studies due to missing HRs for OS, PFS, or DFS are presented in Table S1.
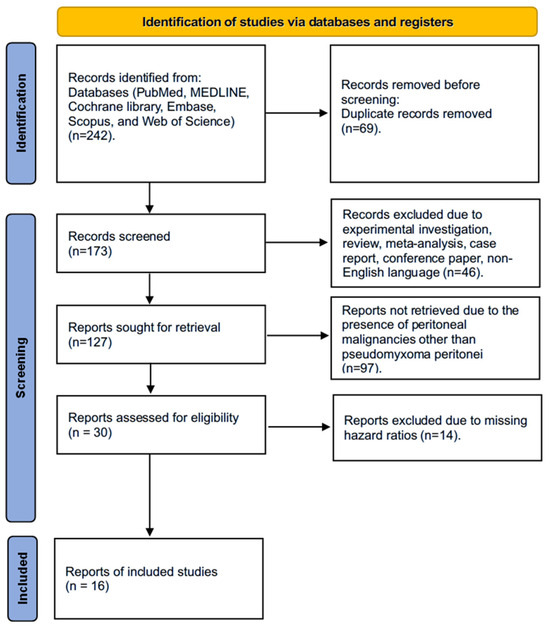
Figure 1.
PRISMA flowchart showing the article selection process.
Among the 16 studies included, all examined the impact of clinical-pathological parameters, ten assessed histological parameters, two evaluated immunohistochemical parameters, and one investigated molecular parameters on the prognosis of CRS-HIPEC in PMP patients. Twelve studies reported OS, while six studies provided data on PFS and DFS, respectively. The female-to-male ratio was approximately 1:1.5, with a median age of 51 years. Most of the included studies (n = 15) were retrospective cohort studies. No randomized controlled trial was identified. Most studies were conducted in Europe (n = 12). Further details of the studies included are presented in Table 1. Detailed surgical and perioperative characteristics of the included studies are described in Table 2.

Table 2.
Surgical and perioperative characteristics of the included studies of the meta-analysis.
3.2. Study Quality and Grade of Evidence Assessment
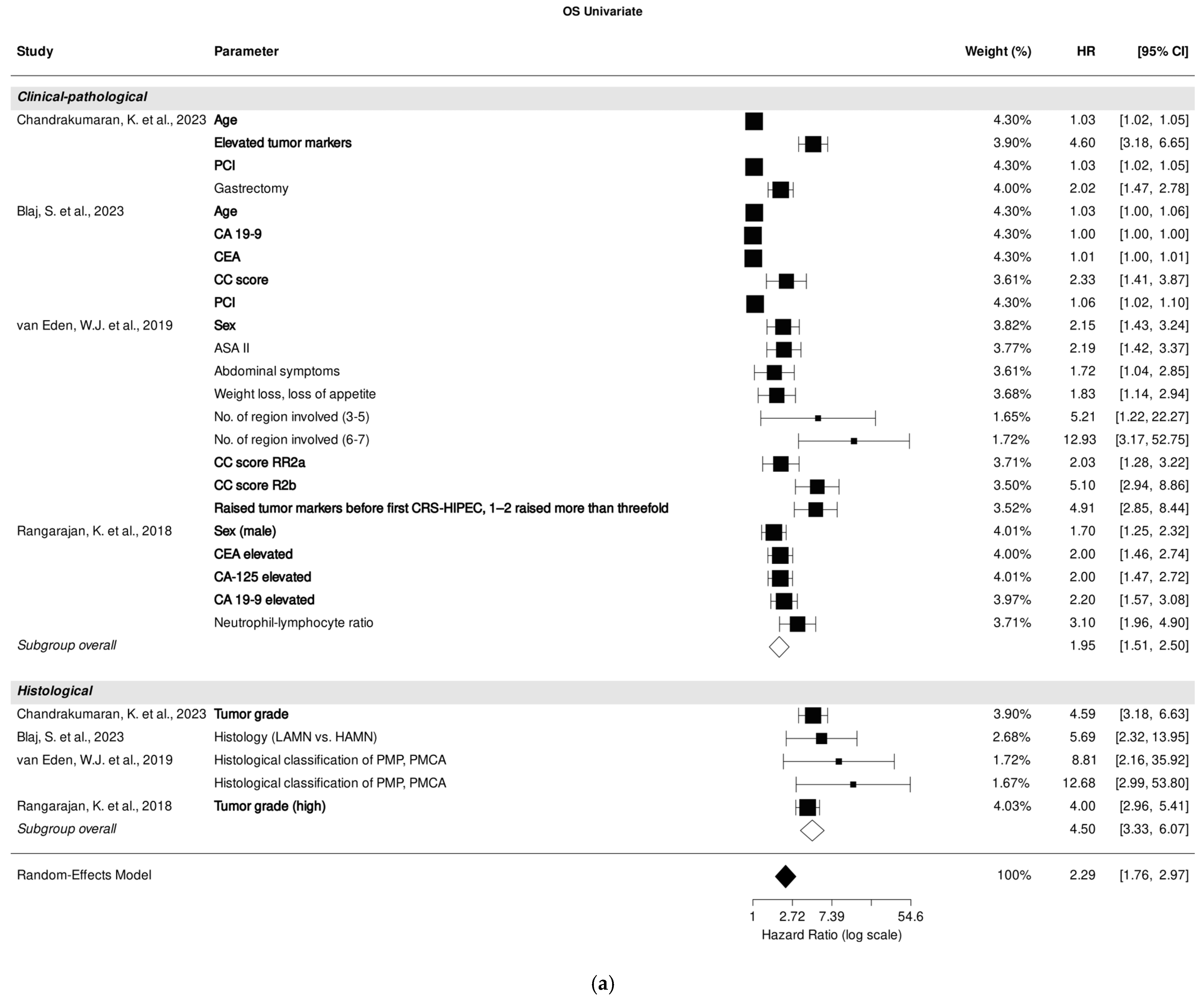
3.3. Association of Clinical-Pathological and Histological Parameters with OS in Univariate Analysis
Four studies investigated the correlation between clinical-pathological and histological parameters and OS through univariate analysis [25,26,30,31].
The pooled analysis of all included studies indicated a strong correlation between clinical-pathological parameters and OS prediction (HR, 1.95; 95% CI: 1.51–2.50; p < 0.001), despite high heterogeneity (I2 = 99.98%). Additionally, histological parameters demonstrated a strong association with OS prediction (HR, 4.50; 95% CI: 3.33–6.07; p < 0.001) with low heterogeneity (I2 = 0.00%).
Within the studies focusing on clinical-pathological parameters, age [25,26], male sex [30,31], CC score [26,30], PCI [25,26], and tumor markers carcinoembryonic antigen (CEA), cancer antigen (CA)-125, and CA 19-9 [25,26,30,31] emerged as the most significant predictors of OS in univariate analysis. Regarding studies that investigated histological parameters, high tumor grade was identified as having the most substantial effect on OS prediction [25,31] (Figure 2a).
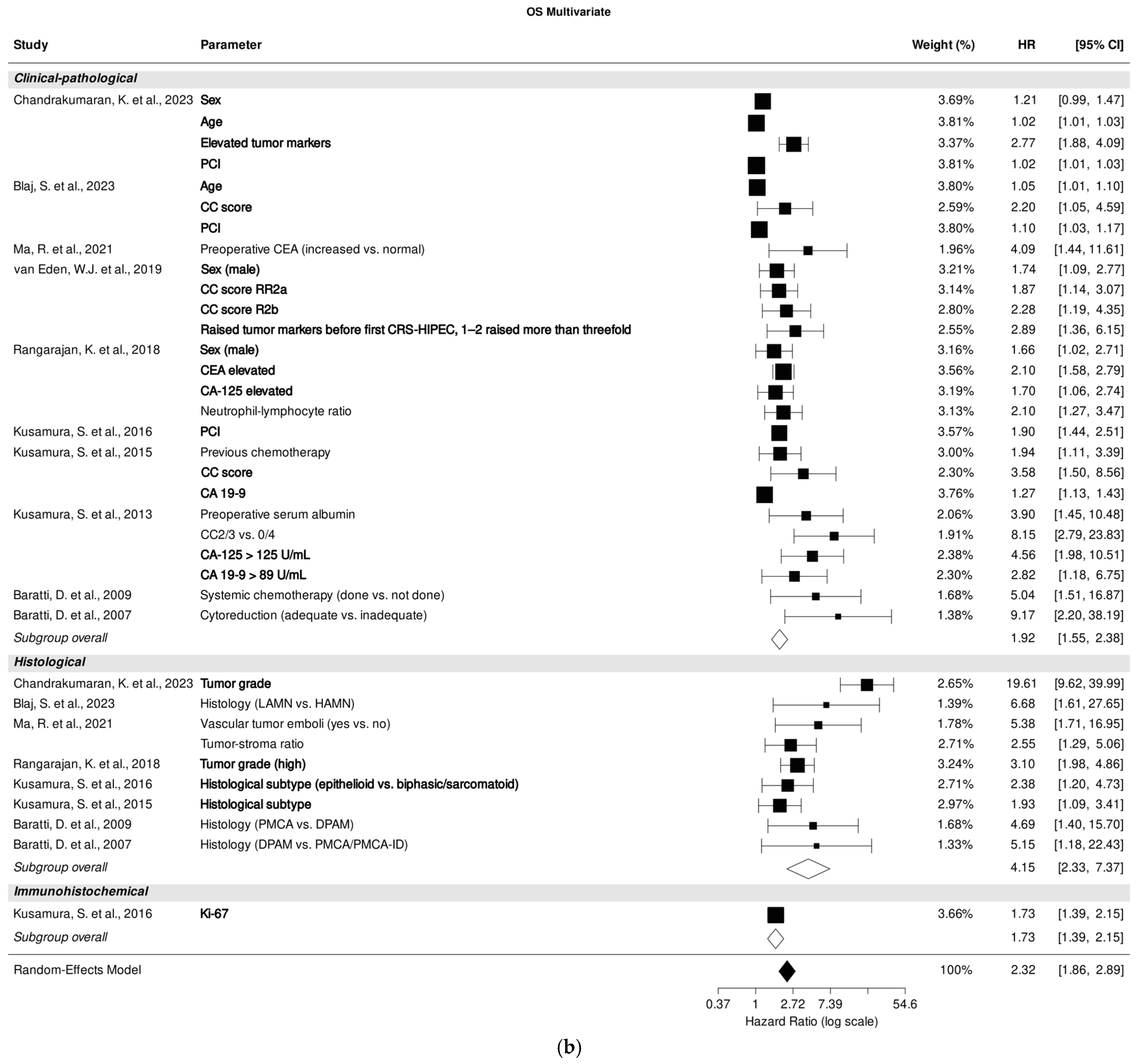
3.4. Association of Clinical-Pathological, Histological, and Immunohistochemical Parameters with OS in Multivariate Analysis
Ten studies included in this analysis examined the correlation between clinical-pathological parameters and OS through multivariate analysis [25,26,28,30,31,33,34,35,37,39]. Eight studies focused on the correlation between histological parameters and OS [25,26,28,31,33,34,37,39]. Additionally, one study examined immunohistochemical parameters [33].
A pooled analysis of all included studies revealed a strong correlation between clinical-pathological parameters and OS prediction (HR, 1.92; 95% CI: 1.55–2.38; p < 0.001), despite high heterogeneity (I2 = 99.69%). Histological parameters were also strongly associated with OS prediction (HR, 4.15; 95% CI: 2.33–7.37; p < 0.001), with moderate heterogeneity (I2 = 74.09%). Immunohistochemical parameters showed a correlation with OS prediction as well (HR, 1.73; 95% CI: 1.39–2.15; p < 0.001).
Among the studies examining the role of clinical-pathological parameters, factors such as age [25,26], male sex [25,30,31], CC score [26,30,34], PCI [25,26,33], and tumor markers CEA, CA-125, and CA 19-9 [25,30,31,34,35] were most significantly associated with OS prediction in multivariate analysis.
Furthermore, high tumor grade [25,31], histological subtype [33,34], and Ki-67 [33] revealed the highest impact in multivariate OS prediction regarding (immuno-) histological parameters (Figure 2b).
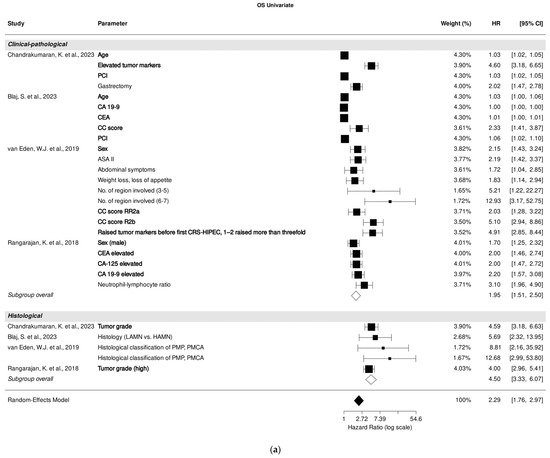
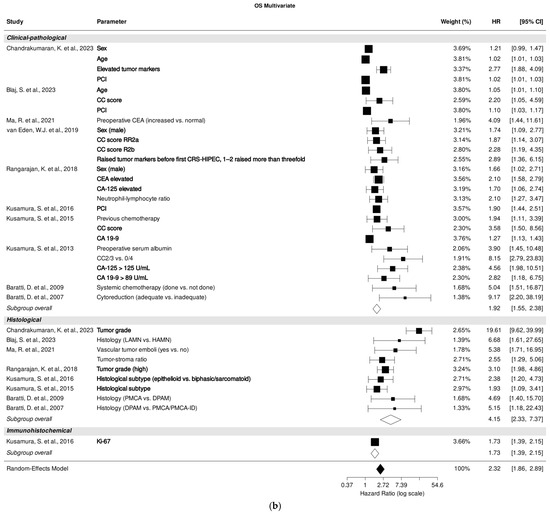
Figure 2.
Forest plot of the association between clinical-pathological, histological, and immunohistochemical parameters with overall survival in univariate (a) and multivariate (b) analysis. Most relevant parameters are shown in bold (based on HRs, weights, and the number of mentions across the different studies). HR, hazard ratio; CI, confidence interval.
Figure 2.
Forest plot of the association between clinical-pathological, histological, and immunohistochemical parameters with overall survival in univariate (a) and multivariate (b) analysis. Most relevant parameters are shown in bold (based on HRs, weights, and the number of mentions across the different studies). HR, hazard ratio; CI, confidence interval.
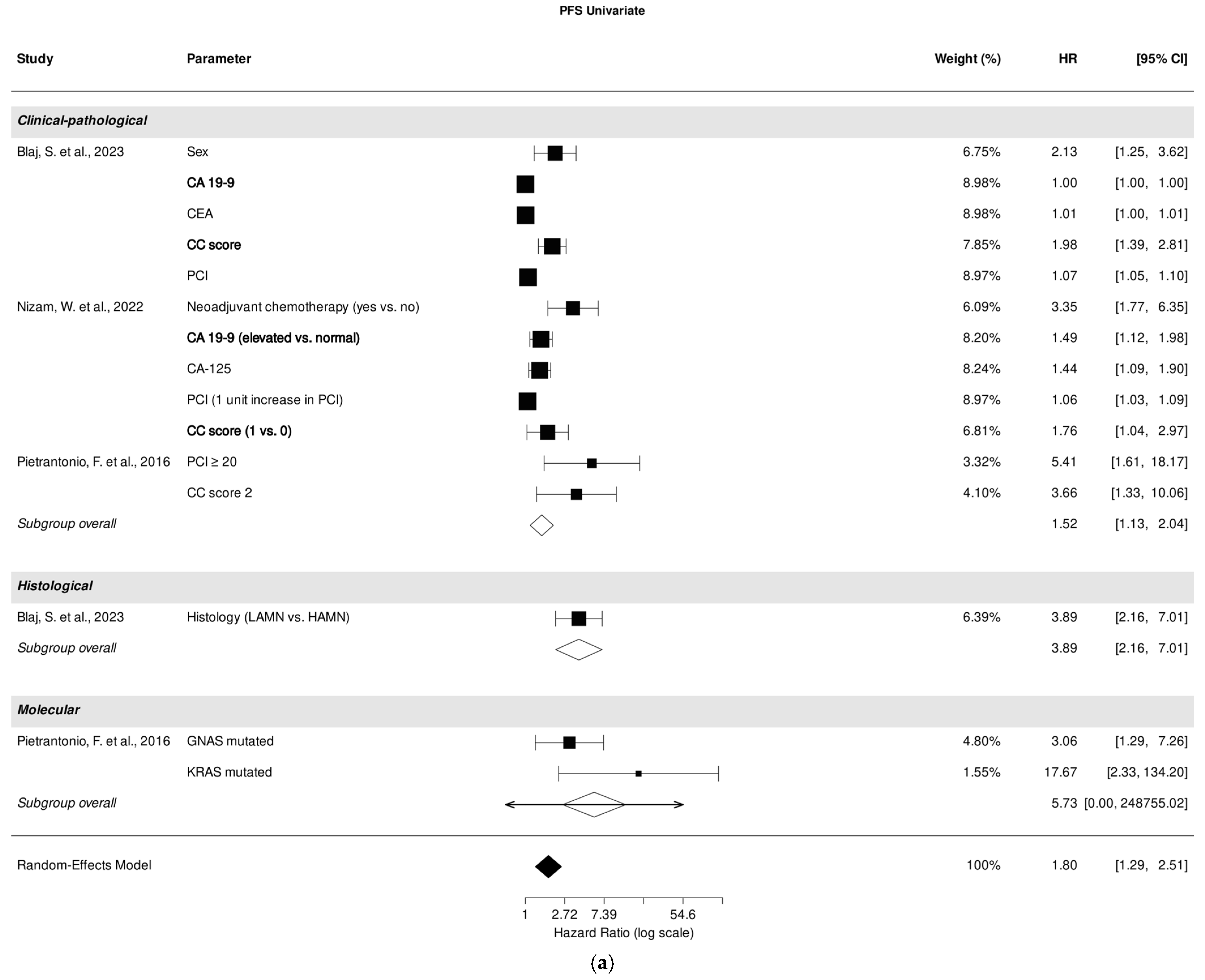
3.5. Association of Clinical-Pathological, Histological, and Molecular Parameters with PFS in Univariate Analysis
Three studies investigated the correlation between clinical-pathological parameters [26,27,32], one study [26] focused on the correlation between histological parameters, while another study examined the correlation between molecular parameters [32] with PFS in univariate analysis.
A pooled analysis of all included studies revealed that clinical-pathological parameters were associated with PFS prediction (HR, 1.52; 95% CI: 1.13–2.04; p = 0.01), despite high heterogeneity (I2 = 99.99%). Histological parameters demonstrated a strong association with PFS prediction (HR, 3.89; 95% CI: 2.16–7.01; p < 0.001). Among all included studies examining the role of clinical-pathological parameters, CC score [26,27], PCI [26,27], and tumor marker CA 19-9 [26,27] were most significantly associated with PFS prediction (Figure 3a).
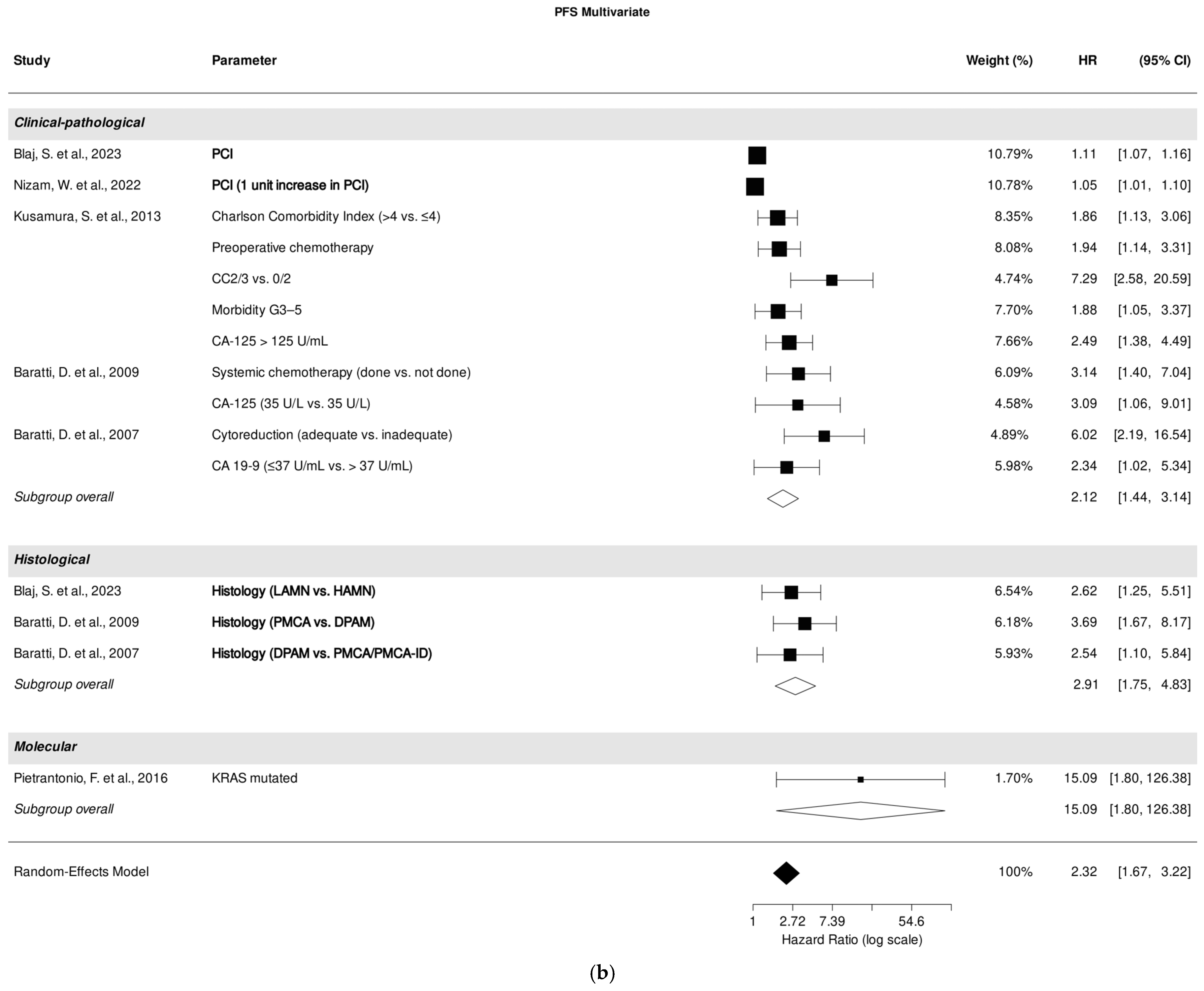
3.6. Association of Clinical-Pathological, Histological, and Molecular Parameters with PFS in Multivariate Analysis
Five studies investigated the correlation between clinical-pathological parameters [26,27,35,37,39], while three studies [26,37,39] focused on the correlation between histological parameters. Additionally, one study [32] examined the correlation between molecular parameters and PFS through multivariate analysis. A pooled analysis of all included studies demonstrated a strong association between clinical-pathological parameters and PFS prediction (HR, 2.12; 95% CI: 1.44–3.14; p = 0.002), despite high heterogeneity (I2 = 98,29%). Histological parameters also revealed a strong association with PFS prediction (HR, 2.91; 95% CI: 1.75–4.83; p = 0.012) with a low heterogeneity (I2 = 0.00%).
Within the analyzed studies, PCI emerged as the most significant clinical-pathological parameter associated with PFS prediction in multivariate analysis [26,27]. Additionally, histological subtypes [26,37,39] were also significantly correlated with PFS prediction (Figure 3b).
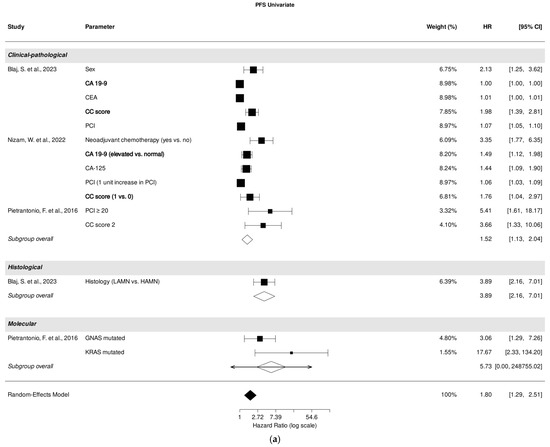
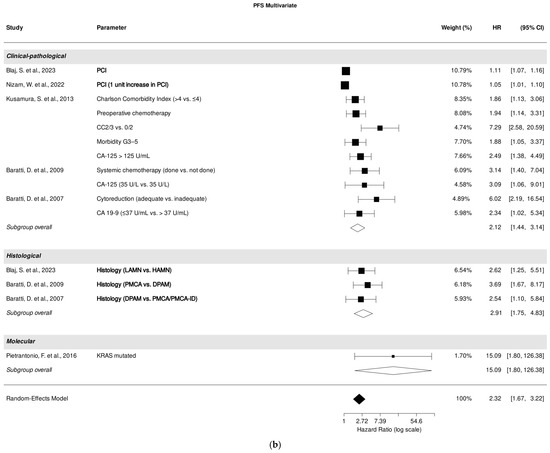
Figure 3.
Forest plot of the association between clinical-pathological, histological, and molecular parameters with progression-free survival in univariate (a) and multivariate (b) analysis. Most relevant parameters (based on HRs, weights, and the number of mentions across the different studies) are shown in bold. HR, hazard ratio; CI, confidence interval.
Figure 3.
Forest plot of the association between clinical-pathological, histological, and molecular parameters with progression-free survival in univariate (a) and multivariate (b) analysis. Most relevant parameters (based on HRs, weights, and the number of mentions across the different studies) are shown in bold. HR, hazard ratio; CI, confidence interval.
3.7. Association of Clinical-Pathological and Histological Parameters with DFS in Univariate Analysis
Seven studies [25,29,30,31,36,38,40] investigated the association between clinical-pathological parameters and DFS through univariate analysis, while three studies focused on histological parameters [25,30,31].
A pooled analysis of all included studies demonstrated a strong association between clinical-pathological (HR, 2.09; 95% CI: 1.59–2.75; p < 0.001) and DFS prediction, despite high heterogeneity (I2 = 99.99%). PCI [25,29], CC score [29,30], and tumor markers CEA, CA-125, and CA 19-9 [25,29,30,31,36,38,40] were most significantly associated with DFS prediction in univariate analysis (Figure 4a).
3.8. Association of Clinical-Pathological and Histological Parameters with DFS in Multivariate Analysis
Four studies [25,29,30,31] investigated the correlation between clinical-pathological parameters, while two studies [25,31] focused on the correlation between histological parameters and DFS in multivariate analysis. A pooled analysis of all included studies revealed an association between clinical-pathological parameters and DFS prediction (HR, 1.85; 95% CI: 1.45–2.36; p < 0.001), despite high heterogeneity (I2 = 96.89%). Among the studies assessing the role of clinical-pathological parameters, PCI [25,29] and tumor markers CEA, CA-125, and CA 19-9 [25,30,31] were most significantly associated with DFS prediction in multivariate analysis (Figure 4b).
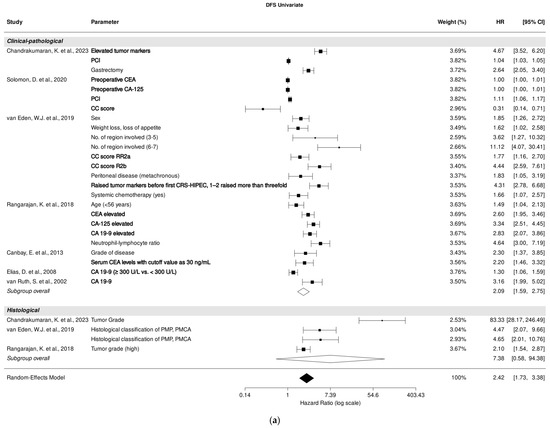
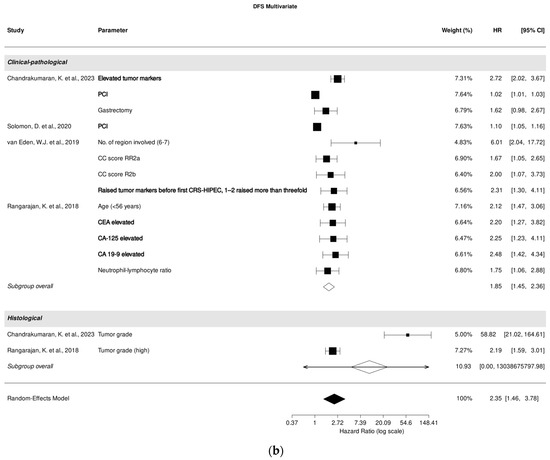
Figure 4.
Forest plot of the association between clinical-pathological and histological parameters with disease-free survival in univariate (a) and multivariate (b) analysis. Most relevant parameters (based on HRs, weights, and the number of mentions across the different studies) are shown in bold. HR, hazard ratio; CI, confidence interval.
Figure 4.
Forest plot of the association between clinical-pathological and histological parameters with disease-free survival in univariate (a) and multivariate (b) analysis. Most relevant parameters (based on HRs, weights, and the number of mentions across the different studies) are shown in bold. HR, hazard ratio; CI, confidence interval.
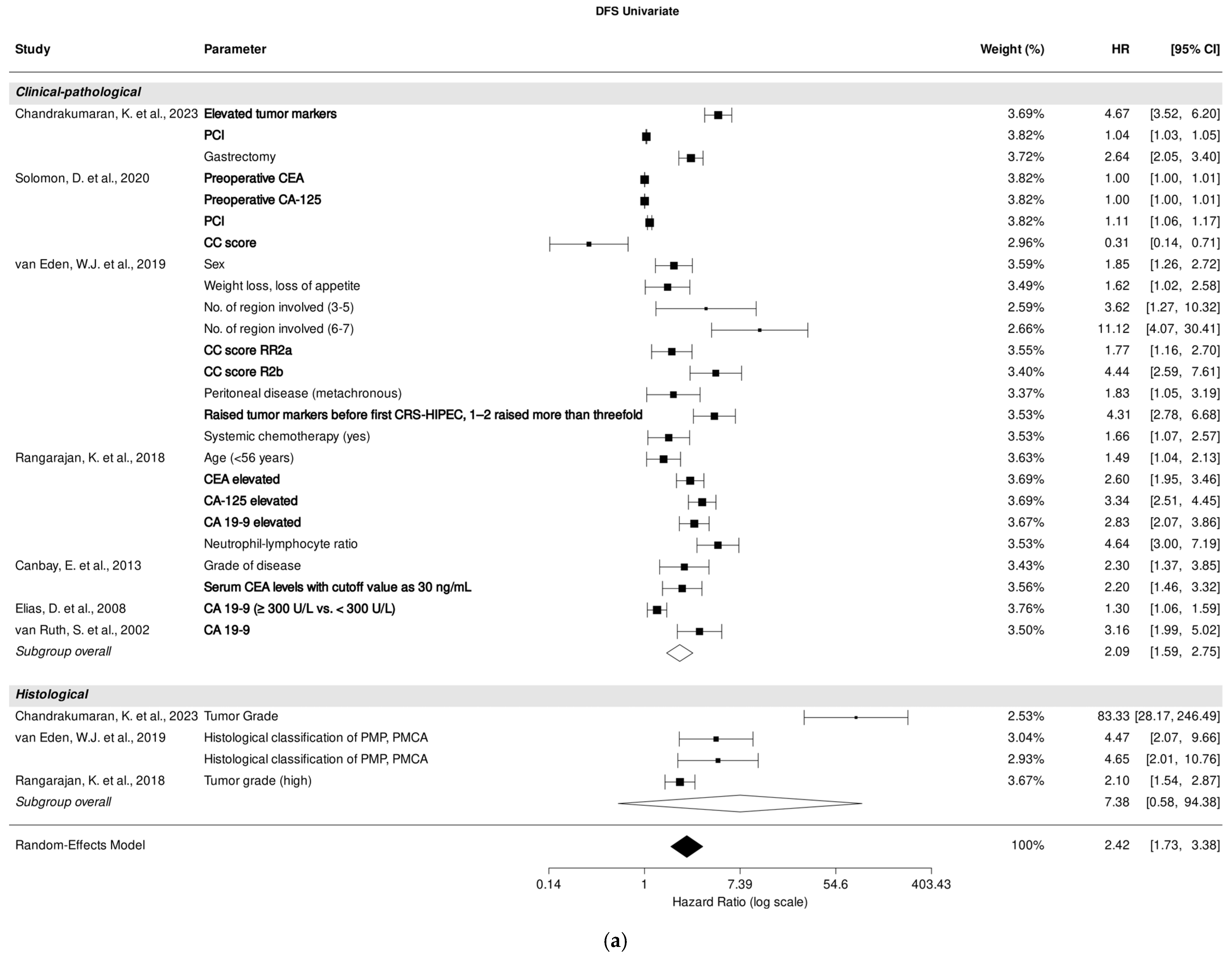
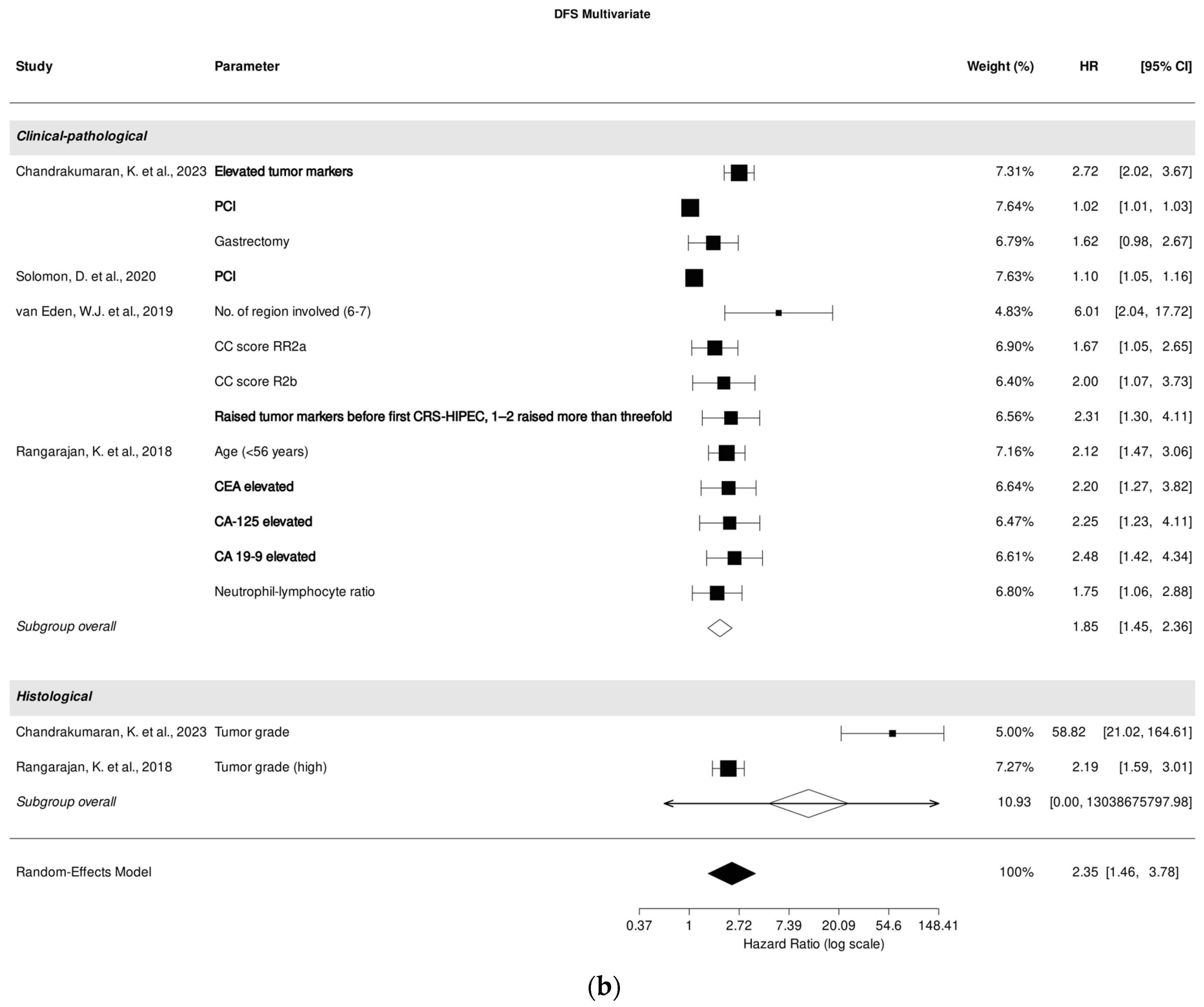
The key findings of this meta-analysis are summarized in Table 3.

Table 3.
Key findings overview of this study: Significant clinical-pathological, (immuno-) histological, and molecular parameters associated with overall, progression-free, and disease-free survival prediction in univariate and multivariate analysis. HR, hazard ratio; CI, confidence interval; OS, overall survival; PFS, progression-free survival; DFS, disease-free survival; CC, completeness of cytoreduction; PCI, peritoneal cancer index; CA, cancer antigen.
3.9. Publication Bias
3.9.1. Publication Bias OS Univariate Analysis
In studies investigating the association between clinical-pathological and histological parameters and OS through univariate analysis, the asymmetrical distribution of studies suggested a significant risk of publication bias. This was corroborated by Egger’s regression test with a p value of <0.0001 (Supplementary Figure S1a). The trim-and-fill method identified eight missing studies on the left side of the funnel plot, suggesting a potential overestimation of effect sizes due to small study effects (Supplementary Figure S1b; Table S4).
3.9.2. Publication Bias OS Multivariate Analysis
In studies examining the association between clinical-pathological, histological and immunohistochemical parameters and OS through multivariate analysis, the asymmetrical distribution of studies indicated the presence of publication bias, which was supported by Egger’s regression test (p < 0.0001) (Supplementary Figure S1c). The trim-and-fill method identified 14 missing studies on the left side of the funnel plot, suggesting a potential overestimation of effect sizes due to small study effects (Supplementary Figure S1d; Table S4).
3.9.3. Publication Bias PFS Univariate Analysis
In studies investigating the association between clinical-pathological, histological, and molecular parameters and PFS through univariate analysis, the asymmetrical distribution of studies indicated the presence of publication bias, which was further corroborated by Egger’s regression test with a p value of <0.0001 (Supplementary Figure S2a). The trim-and-fill method identified six missing studies on the left side of the funnel plot, suggesting a potential overestimation of effect sizes due to small study effects (Supplementary Figure S2b, Table S4).
3.9.4. Publication Bias PFS Multivariate Analysis
In studies examining the association between clinical-pathological, histological, and molecular parameters and PFS through multivariate analysis, the asymmetrical distribution of studies indicated the presence of publication bias, which was further corroborated by Egger’s regression test with a p value of <0.0001 (Supplementary Figure S2c). The trim-and-fill method identified six missing studies on the left side of the funnel plot, suggesting a potential overestimation of effect sizes due to small study effects (Supplementary Figure S2d; Table S4).
3.9.5. Publication Bias DFS Univariate Analysis
In studies investigating the association between clinical-pathological and histological parameters and DFS through univariate analysis, the asymmetrical distribution of studies indicated the presence of publication bias, which was further corroborated by Egger’s regression test with a p value of <0.0001 (Supplementary Figure S3a). Trim-and-fill analyses showed no imputed studies, implying a lower likelihood of publication bias, but still a high heterogeneity (Table S4).
3.9.6. Publication Bias DFS Multivariate Analysis
In studies examining the association between clinical-pathological and histological parameters and DFS through multivariate analysis, the asymmetrical distribution of studies indicated the presence of publication bias, which was further corroborated by Egger’s regression test with a p value of <0.0001 (Supplementary Figure S3b). Trim-and-fill analyses showed no imputed studies, implying a lower likelihood of publication bias, but still a high heterogeneity (Table S4).
4. Discussion
PMP is a rare malignancy frequently resulting from a perforated mucinous appendiceal tumor [1,2]. CRS and HIPEC remain the main therapeutic interventions. Nevertheless, there is potential to further refine eligibility criteria, as post-surgical tumor recurrence and progression significantly impact long-term survival [7,8,9,10,11]. Until now, the traditional criteria such as operability (based on patient’s ability to tolerate major surgery) and resectability (based on the extent and distribution of peritoneal disease) seem to appear insufficient.
In this study, we provide one of the first comprehensive analyses of predictive markers affecting OS, PFS, and DFS following CRS and HIPEC in patients with PMP. The pooled analysis, including 16 studies, identified the following risk factors: three patient-related risk factors (age, male sex, and PCI), three tumor-markers (CEA, CA-125, CA 19-9), two histological parameters (high tumor grade and histological subtype), one immunohistochemical parameter (Ki-67), and one surgery-related risk factor (CC score).
4.1. Prognostic Factors on CRS and HIPEC Efficacy
4.1.1. Clinical-Pathological Risk Factors
Patient-Related Risk Factors
In this study, age, male sex, PCI, and tumor markers CEA, CA-125, and CA 19-9 emerged as the most significant factors associated with prognosis. Epidemiological data indicate that both the prevalence and incidence of PMP increase with advancing age, with the highest rates observed in the oldest demographic groups. In a nationwide Chinese cohort, both prevalence and incidence were found to rise across age strata, reaching a peak in individuals over 80 years of age [46]. Moreover, advanced age is directly correlated with an increase in comorbidities and a diminished capacity for recovery following major surgical procedures, potentially negating any long-term survival benefits conferred by CRS and HIPEC [47]. In this context, the age threshold most consistently associated with increased postoperative complications after CRS-HIPEC in PMP patients is ≥65 years [7,25,48,49,50].
The association of male patients with OS prediction aligns with findings from one of the largest studies evaluating outcomes and long-term survival following CRS-HIPEC in PMP patients [12] and a recent meta-analysis [51]. The existing literature suggests that male patients may exhibit symptom onset at a more advanced stage compared to female patients [4]. So far, there is no definitive hypothesis to explain this discrepancy. It is possible that male patients may delay seeking medical attention and overlook symptoms, a pattern observed in other cancers, such as colorectal cancer [52,53].
Furthermore, the PCI score, as the most widely utilized tool for determining the extent of peritoneal disease, has been associated with OS, PFS, and DFS prediction. A practical prognostic dichotomy at a PCI of approximately ≤21 for low-grade and ≤25 for high-grade tumors is currently supported [54]. It is acknowledged that histologic grade and the completeness of cytoreduction influence risk, and that selected patients with a high PCI may still derive benefit from CRS and HIPEC when complete cytoreduction (CC0/1) is achievable [25,26,54,55]. This aligns with a recent meta-analysis, which indicates that a PCI exceeding 20 points is associated with poor surgical outcomes and a higher risk of recurrence [51]. In a large international registry, Chua et al. demonstrated that a high PCI is an independent predictor of poor PFS [12].
Our meta-analysis indicated that tumor markers CEA, CA-125, and CA 19-9 are associated with OS, PFS, and DFS prediction, which is in line with a recent meta-analysis [51]. In the context of PMP, these tumor markers could be crucial for baseline risk stratification assessing the predictability of resection. Regarding the initial evaluation of disease burden and resectability, elevated preoperative levels of CEA, CA 19-9, and CA-125 correlate with higher PCI and lower likelihood of achieving complete cytoreduction [27,35,39,56,57]. Notably, CA 19-9 frequently demonstrates the strongest association with earlier progression, while both CA-125 and CA 19-9 are linked to inferior OS in several cohorts [26,35,39,40,51,56,57,58,59,60,61].
Surgery-Related Risk Factors
Achieving complete cytoreduction (CC0/1) is the primary determinant of long-term survival, with HIPEC commonly integrated [12]. Patients with CCR2 or CCR3, characterized by gross residual disease, exhibit a 5-year survival rate of 24% in contrast to 85% in CCR0 patients [12]. In this study, complete/incomplete cytoreduction was associated with OS, PFS, and DFS prediction. These results are in line with a recent meta-analysis by Wei et al. who identified the CC score as a risk factor associated with surgery [51]. Even histologically bland or low-grade appearance PMP often necessitates highly aggressive and extensive surgical intervention due to the biological behavior of PMP, which is not reflected by its histology [62].
4.1.2. (Immuno-) Histological Parameters
Concerning histological parameters, both high tumor grade and histological subtype exhibited the most significant associations with prognosis, including OS and PFS prediction. The Peritoneal Surface Oncology Group International (PSOGI) consensus categorizes PMP disease into acellular mucin (acellular mucinous peritoneal deposits), low-grade mucinous carcinoma peritonei/disseminated peritoneal adenomucinosis (DPAM), high-grade mucinous carcinoma peritonei/peritoneal mucinous carcinomatosis (PMCA), and high-grade mucinous carcinoma peritonei with signet ring cells/peritoneal mucinous carcinomatosis with signet ring cells (PMCA-S) [63,64], in alignment with the World Health Organization (WHO) two-tier system (low vs. high grade) [65]. Recent studies involving CRS and HIPEC indicate that survival outcomes are most favorable for acellular mucin, moderate for low-grade in comparison with less favorable for high-grade, and finally to poorest in the presence of signet ring cells [12,63,64]. Multivariable analyses consistently identified the histologic grade of the peritoneum as an independent predictor of OS, with risk increasing from low-grade to high-grade to signet ring cell categories [25,26,66].
In the context of immunohistochemical parameters, our meta-analysis identified Ki-67 as the sole predictor of OS in patients with PMP undergoing CRS and HIPEC. For high-grade PMP, a Ki-67 proliferation index threshold of approximately 15% has been shown to effectively stratify prognosis. Patients with high-grade PMP exhibiting a Ki-67 index exceeding 15% experience significantly poorer OS and DFS compared to those with an index of 15% or less. This finding has prompted a proposed modification of the PSOGI classification, which further subdivides high-grade PMP based on Ki-67 levels [67,68].
4.1.3. Molecular Parameters
Our meta-analysis did not identify any significant prognostic molecular markers; however, this likely reflects the limited availability of data rather than a true lack of biological relevance. Molecular markers in PMP primarily function to elucidate pathogenesis and provide prognostic stratification. The genes most mutated in PMP are KRAS and GNAS, with KRAS mutations occurring in 38–100% of cases and GNAS in 17–100%, often appearing together. These mutations are crucial to the pathogenesis of PMP and are linked to mucin overproduction through the PKA pathway, a characteristic feature of the disease [32,69,70,71,72,73,74]. So far, no clinical study has reported data on KRAS inhibition, but in vitro and in vivo models have shown promising results in targeting KRAS in PMP [75,76].
4.2. Strengths and Limitations of the Study
In contrast to two earlier systematic reviews [77,78] and one recent meta-analysis [51], our meta-analysis neither assessed the efficacy or safety of cytoreductive surgery and HIPEC in patients with pseudomyxoma peritonei nor focused exclusively on clinical prognostic factors. Instead, it comprehensively integrated (immuno-) histological and molecular parameters, thereby enabling a broader and more in-depth evaluation of prognostic determinants.
However, it is important to acknowledge certain limitations inherent in our study. Firstly, most of the studies included were retrospective in nature, which may introduce selection bias and impact the overall quality of the evidence. To date, no randomized controlled trials have been conducted. Due to high heterogeneity, the pooled estimates should be interpreted with caution.
Secondly, the results of the meta-analysis may be influenced by the variability in patient selection criteria across the included studies, such as age, systemic anticancer treatments, comorbidities, and performance status.
Thirdly, this review revealed that many studies report only a limited set of key surgical characteristics, and there is substantial variability in the HIPEC procedures themselves, particularly regarding duration, temperature, drug choice, and drug concentration. The studies included were conducted over a span of more than two decades, which may contribute to a significant publication bias. During this period, there have been changes in surgical teams, HIPEC regimens, and the classification and awareness of PMP. Lastly, the restriction to English-language studies, predominantly conducted in Europe, and the use of different statistical methods in the included studies may further limit the reliability and generalizability of our meta-analysis.
5. Conclusions
Despite significant heterogeneity and possible selection bias due to mostly retrospective included studies, our meta-analysis primarily identified clinical-pathological parameters such as age, male sex, PCI, tumor markers, high tumor grade, histological subtype, and CC score as the most pertinent indicators associated with OS, PFS, and DFS prediction in PMP patients undergoing CRS and HIPEC. Currently, immunohistochemical and molecular markers only play a minor prognostic role. Effective individualized multidisciplinary management of PMP requires the integration of molecular and genetic biomarkers [79]; therefore, the current scarcity of molecular studies must be urgently addressed through well-designed, adequately powered investigations of markers such as KRAS and GNAS to clarify their prognostic and potential therapeutic relevance.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cancers18050795/s1, Figure S1: Funnel plot of studies examining the association between clinical-pathological, histological, and immunohistochemical parameters with overall survival in univariate (a), trim-and-fill adapted univariate (b), multivariate (c), and trim-and-fill adapted multivariate (d) analysis; Figure S2: Funnel plot of studies examining the association between clinical-pathological, histological, and molecular parameters with progression-free survival in univariate (a), trim-and-fill adapted univariate (b), multivariate (c), and trim-and-fill adapted multivariate (d) analysis; Figure S3: Funnel plot of studies examining the association between clinical-pathological and histological parameters with disease-free survival in univariate (a) and multivariate (b) analysis; Table S1: Excluded studies due to missing HRs for OS, PFS, or DFS; Table S2: The quality of the included studies (Newcastle-Ottawa Quality Assessment Scale); Table S3: Grading of Recommendations, Assessment, Development and Evaluation (GRADE) Working Group grades of evidence of the included studies; Table S4: Trim-and-fill sensitivity analysis results.
Author Contributions
Conceptualization, J.P.R., D.N. and T.J.; methodology, J.P.R., D.N. and T.J.; validation, L.S.-C., P.S., K.E., D.B., J.P. and A.P.; formal analysis, J.P.R., A.K. and D.N.; investigation, J.P.R., D.N. and T.J.; resources, J.P.R., A.K., D.B. and D.N.; data curation, J.P.R., A.K., L.S.-C., P.S., K.E., D.B., J.P., A.P., D.N. and T.J.; writing—original draft preparation, J.P.R., D.N. and T.J.; writing—review and editing, J.P.R., A.K., L.S.-C., P.S., K.E., D.B., J.P., A.P., D.N. and T.J.; visualization, J.P.R., A.K. and D.N.; supervision, D.N. and T.J.; project administration, J.P.R., D.N. and T.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
As this was a systematic literature review, no institutional review board statement was required.
Informed Consent Statement
As this was a systematic literature review, no informed patient consent was required.
Data Availability Statement
Data for the systematic literature review and meta-analysis were obtained from published sources. Data are available upon request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CA-125 | Cancer antigen 125 |
| CC | Completeness of cytoreduction score |
| CEA | Carcinoembryonic antigen |
| CI | Confidence interval |
| CRS | Cytoreductive surgery |
| DFS | Disease-free survival |
| DPAM | Disseminated peritoneal adenomucinosis |
| GRADE | Grading of Recommendations, Assessment, Development and Evaluation |
| HIPEC | Hyperthermic intraperitoneal chemotherapy |
| HKJS | Hartung–Knapp–Sidik–Jonkman |
| HR | Hazard ratio |
| NOS | Newcastle Ottawa Scale |
| OS | Overall survival |
| PCI | Peritoneal cancer index |
| PFS | Progression-free survival |
| PMCA | Peritoneal mucinous carcinomatosis |
| PMCA-S | Peritoneal mucinous carcinomatosis with signet ring cells |
| PMP | Pseudomyxoma peritonei |
| PRISMA | Preferred reporting items for systematic reviews and meta-analyses |
| PSOGI | Peritoneal Surface Oncology Group International |
| REML | Restricted Maximum Likelihood |
| WHO | World Health Organization |
References
- Sugarbaker, P.H. Pseudomyxoma peritonei. Cancer Treat. Res. 1996, 81, 105–119. [Google Scholar] [CrossRef]
- Moran, B.J.; Cecil, T.D. The etiology, clinical presentation, and management of pseudomyxoma peritonei. Surg. Oncol. Clin. N. Am. 2003, 12, 585–603. [Google Scholar] [CrossRef]
- Patrick-Brown, T.; Carr, N.J.; Swanson, D.M.; Larsen, S.; Mohamed, F.; Flatmark, K. Estimating the Prevalence of Pseudomyxoma Peritonei in Europe Using a Novel Statistical Method. Ann. Surg. Oncol. 2021, 28, 252–257. [Google Scholar] [CrossRef]
- Mittal, R.; Chandramohan, A.; Moran, B. Pseudomyxoma peritonei: Natural history and treatment. Int. J. Hyperth. 2017, 33, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Smeenk, R.M.; van Velthuysen, M.L.; Verwaal, V.J.; Zoetmulder, F.A. Appendiceal neoplasms and pseudomyxoma peritonei: A population based study. Eur. J. Surg. Oncol. 2008, 34, 196–201. [Google Scholar] [CrossRef]
- Zhong, Y.; Deng, M.; Xu, R.; Kokudo, N.; Tang, W. Pseudomyxoma peritonei as an intractable disease and its preoperative assessment to help improve prognosis after surgery: A review of the literature. Intractable Rare Dis. Res. 2012, 1, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Kusamura, S.; Barretta, F.; Yonemura, Y.; Sugarbaker, P.H.; Moran, B.J.; Levine, E.A.; Goere, D.; Baratti, D.; Nizri, E.; Morris, D.L.; et al. The Role of Hyperthermic Intraperitoneal Chemotherapy in Pseudomyxoma Peritonei After Cytoreductive Surgery. JAMA Surg. 2021, 156, e206363. [Google Scholar] [CrossRef] [PubMed]
- Moran, B.; Baratti, D.; Yan, T.D.; Kusamura, S.; Deraco, M. Consensus statement on the loco-regional treatment of appendiceal mucinous neoplasms with peritoneal dissemination (pseudomyxoma peritonei). J. Surg. Oncol. 2008, 98, 277–282. [Google Scholar] [CrossRef]
- Delhorme, J.B.; Villeneuve, L.; Bouché, O.; Averous, G.; Dohan, A.; Gornet, J.M.; You, B.; Bibeau, F.; Dartigues, P.; Eveno, C.; et al. Appendiceal tumors and pseudomyxoma peritonei: French Intergroup Clinical Practice Guidelines for diagnosis, treatments and follow-up (RENAPE, RENAPATH, SNFGE, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO, ACHBT, SFR). Dig. Liver Dis. 2022, 54, 30–39. [Google Scholar] [CrossRef]
- Kusamura, S.; Delhorme, J.B.; Taibi, A.; Villeneuve, L.; Deraco, M.; Dico, R.L.; Glehen, O.; Moran, B. The 2022 PSOGI International Consensus on HIPEC Regimens for Peritoneal Malignancies: Pseudomyxoma Peritonei. Ann. Surg. Oncol. 2024, 31, 6262–6273. [Google Scholar] [CrossRef]
- Govaerts, K.; Lurvink, R.J.; De Hingh, I.; Van der Speeten, K.; Villeneuve, L.; Kusamura, S.; Kepenekian, V.; Deraco, M.; Glehen, O.; Moran, B.J. Appendiceal tumours and pseudomyxoma peritonei: Literature review with PSOGI/EURACAN clinical practice guidelines for diagnosis and treatment. Eur. J. Surg. Oncol. 2021, 47, 11–35. [Google Scholar] [CrossRef] [PubMed]
- Chua, T.C.; Moran, B.J.; Sugarbaker, P.H.; Levine, E.A.; Glehen, O.; Gilly, F.N.; Baratti, D.; Deraco, M.; Elias, D.; Sardi, A.; et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J. Clin. Oncol. 2012, 30, 2449–2456. [Google Scholar] [CrossRef]
- Ahmadi, N.; Kostadinov, D.; Sakata, S.; Ball, W.R.; Gandhi, J.; Carr, N.J.; Tzivanakis, A.; Dayal, S.P.; Mohamed, F.; Cecil, T.D.; et al. Managing Recurrent Pseudomyxoma Peritonei in 430 Patients After Complete Cytoreduction and HIPEC: A Dilemma for Patients and Surgeons. Ann. Surg. Oncol. 2021, 28, 7809–7820. [Google Scholar] [CrossRef]
- Baratti, D.; Kusamura, S.; Guaglio, M.; Milione, M.; Pietrantonio, F.; Cavalleri, T.; Morano, F.; Deraco, M. Relapse of Pseudomyxoma Peritonei After Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy: Pattern of Failure, Clinical Management and Outcomes. Ann. Surg. Oncol. 2023, 30, 404–414. [Google Scholar] [CrossRef]
- Lord, A.C.; Shihab, O.; Chandrakumaran, K.; Mohamed, F.; Cecil, T.D.; Moran, B.J. Recurrence and outcome after complete tumour removal and hyperthermic intraperitoneal chemotherapy in 512 patients with pseudomyxoma peritonei from perforated appendiceal mucinous tumours. Eur. J. Surg. Oncol. 2015, 41, 396–399. [Google Scholar] [CrossRef]
- Smeenk, R.M.; Verwaal, V.J.; Antonini, N.; Zoetmulder, F.A. Survival analysis of pseudomyxoma peritonei patients treated by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann. Surg. 2007, 245, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Mercier, F.; Dagbert, F.; Pocard, M.; Goéré, D.; Quenet, F.; Wernert, R.; Dumont, F.; Brigand, C.; Passot, G.; Glehen, O. Recurrence of pseudomyxoma peritonei after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. BJS Open 2019, 3, 195–202. [Google Scholar] [CrossRef]
- Carboni, F.; Valle, M.; Vaira, M.; Sammartino, P.; Federici, O.; Robella, M.; Deraco, M.; Framarini, M.; Macrì, A.; Sassaroli, C.; et al. Complications and Mortality Rate of Cytoreductive Surgery with Hyperthermic Intraperitoneal Chemotherapy: Italian Peritoneal Surface Malignancies Oncoteam Results Analysis. Cancers 2022, 14, 5824. [Google Scholar] [CrossRef]
- Baratti, D.; Kusamura, S.; Mingrone, E.; Balestra, M.R.; Laterza, B.; Deraco, M. Identification of a subgroup of patients at highest risk for complications after surgical cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann. Surg. 2012, 256, 334–341. [Google Scholar] [CrossRef]
- Cripe, J.; Tseng, J.; Eskander, R.; Fader, A.N.; Tanner, E.; Bristow, R. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for recurrent ovarian carcinoma: Analysis of 30-day morbidity and mortality. Ann. Surg. Oncol. 2015, 22, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Gamboa, A.C.; Lee, R.M.; Turgeon, M.K.; Zaidi, M.Y.; Kimbrough, C.W.; Grotz, T.E.; Leiting, J.; Fournier, K.; Lee, A.J.; Dineen, S.P.; et al. Implications of Postoperative Complications for Survival After Cytoreductive Surgery and HIPEC: A Multi-Institutional Analysis of the US HIPEC Collaborative. Ann. Surg. Oncol. 2020, 27, 4980–4995. [Google Scholar] [CrossRef] [PubMed]
- Choudry, M.H.A.; Shuai, Y.; Jones, H.L.; Pai, R.K.; Pingpank, J.F.; Ahrendt, S.S.; Holtzman, M.P.; Zeh, H.J.; Bartlett, D.L. Postoperative Complications Independently Predict Cancer-Related Survival in Peritoneal Malignancies. Ann. Surg. Oncol. 2018, 25, 3950–3959. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Peacock, R. Effectiveness and efficiency of search methods in systematic reviews of complex evidence: Audit of primary sources. BMJ 2005, 331, 1064–1065. [Google Scholar] [CrossRef] [PubMed]
- Chandrakumaran, K.; Carr, N.J.; Mohamed, F.; Cecil, T.D.; Moran, B.J. Development and Validation of Nomograms to Predict Survival in Patients Undergoing Complete Cytoreduction and Hyperthermic Intraperitoneal Chemotherapy for Pseudomyxoma Peritonei of Appendiceal Origin. JAMA Surg. 2023, 158, 522–530. [Google Scholar] [CrossRef]
- Blaj, S.; Dora, D.; Lohinai, Z.; Herold, Z.; Szasz, A.M.; Herzberg, J.; Kodacsi, R.; Baransi, S.; Schlitt, H.J.; Hornung, M.; et al. Prognostic Factors in Pseudomyxoma Peritonei with Emphasis on the Predictive Role of Peritoneal Cancer Index and Tumor Markers. Cancers 2023, 15, 1326. [Google Scholar] [CrossRef]
- Nizam, W.; Fackche, N.; Pessoa, B.; Kubi, B.; Cloyd, J.M.; Grotz, T.; Fournier, K.; Dineen, S.; Veerapong, J.; Baumgartner, J.M.; et al. Prognostic Significance of Preoperative Tumor Markers in Pseudomyxoma Peritonei from Low-Grade Appendiceal Mucinous Neoplasm: A Study from the US HIPEC Collaborative. J. Gastrointest. Surg. 2022, 26, 414–424. [Google Scholar] [CrossRef]
- Ma, R.; Lin, Y.L.; Li, X.B.; Yan, F.C.; Xu, H.B.; Peng, Z.; Li, Y. Tumor-stroma ratio as a new prognosticator for pseudomyxoma peritonei: A comprehensive clinicopathological and immunohistochemical study. Diagn. Pathol. 2021, 16, 116. [Google Scholar] [CrossRef]
- Solomon, D.; Bekhor, E.; Leigh, N.; Maniar, Y.M.; Totin, L.; Hofstedt, M.; Aycart, S.N.; Carr, J.; Ballentine, S.; Magge, D.R.; et al. Surveillance of Low-Grade Appendiceal Mucinous Neoplasms with Peritoneal Metastases After Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy: Are 5 Years Enough? A Multisite Experience. Ann. Surg. Oncol. 2020, 27, 147–153. [Google Scholar] [CrossRef]
- van Eden, W.J.; Kok, N.F.M.; Snaebjornsson, P.; Jóźwiak, K.; Woensdregt, K.; Bottenberg, P.D.; Boot, H.; Aalbers, A.G.J. Factors influencing long-term survival after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for pseudomyxoma peritonei originating from appendiceal neoplasms. BJS Open 2019, 3, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Rangarajan, K.; Chandrakumaran, K.; Dayal, S.; Mohamed, F.; Moran, B.J.; Cecil, T.D. The pre-operative neutrophil-lymphocyte ratio predicts overall and disease-free survival following cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) in patients with pseudomxyoma peritonei of appendiceal origin. Int. J. Hyperth. 2018, 34, 559–563. [Google Scholar] [CrossRef]
- Pietrantonio, F.; Perrone, F.; Mennitto, A.; Gleeson, E.M.; Milione, M.; Tamborini, E.; Busico, A.; Settanni, G.; Berenato, R.; Caporale, M.; et al. Toward the molecular dissection of peritoneal pseudomyxoma. Ann. Oncol. 2016, 27, 2097–2103. [Google Scholar] [CrossRef]
- Kusamura, S.; Torres Mesa, P.A.; Cabras, A.; Baratti, D.; Deraco, M. The Role of Ki-67 and Pre-cytoreduction Parameters in Selecting Diffuse Malignant Peritoneal Mesothelioma (DMPM) Patients for Cytoreductive Surgery (CRS) and Hyperthermic Intraperitoneal Chemotherapy (HIPEC). Ann. Surg. Oncol. 2016, 23, 1468–1473. [Google Scholar] [CrossRef]
- Kusamura, S.; Baratti, D.; Hutanu, I.; Gavazzi, C.; Morelli, D.; Iusco, D.R.; Grassi, A.; Bonomi, S.; Virzì, S.; Haeusler, E.; et al. The role of baseline inflammatory-based scores and serum tumor markers to risk stratify pseudomyxoma peritonei patients treated with cytoreduction (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC). Eur. J. Surg. Oncol. 2015, 41, 1097–1105. [Google Scholar] [CrossRef]
- Kusamura, S.; Hutanu, I.; Baratti, D.; Deraco, M. Circulating tumor markers: Predictors of incomplete cytoreduction and powerful determinants of outcome in pseudomyxoma peritonei. J. Surg. Oncol. 2013, 108, 1–8. [Google Scholar] [CrossRef]
- Canbay, E.; Ishibashi, H.; Sako, S.; Mizumoto, A.; Hirano, M.; Ichinose, M.; Takao, N.; Yonemura, Y. Preoperative carcinoembryonic antigen level predicts prognosis in patients with pseudomyxoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. World J. Surg. 2013, 37, 1271–1276. [Google Scholar] [CrossRef] [PubMed]
- Baratti, D.; Kusamura, S.; Nonaka, D.; Cabras, A.D.; Laterza, B.; Deraco, M. Pseudomyxoma peritonei: Biological features are the dominant prognostic determinants after complete cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann. Surg. 2009, 249, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Elias, D.; Honoré, C.; Ciuchendéa, R.; Billard, V.; Raynard, B.; Lo Dico, R.; Dromain, C.; Duvillard, P.; Goéré, D. Peritoneal pseudomyxoma: Results of a systematic policy of complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Br. J. Surg. 2008, 95, 1164–1171. [Google Scholar] [CrossRef] [PubMed]
- Baratti, D.; Kusamura, S.; Martinetti, A.; Seregni, E.; Laterza, B.; Oliva, D.G.; Deraco, M. Prognostic value of circulating tumor markers in patients with pseudomyxoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann. Surg. Oncol. 2007, 14, 2300–2308. [Google Scholar] [CrossRef]
- van Ruth, S.; Hart, A.A.; Bonfrer, J.M.; Verwaal, V.J.; Zoetmulder, F.A. Prognostic value of baseline and serial carcinoembryonic antigen and carbohydrate antigen 19.9 measurements in patients with pseudomyxoma peritonei treated with cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann. Surg. Oncol. 2002, 9, 961–967. [Google Scholar] [CrossRef]
- Viechtbauer, W. Conducting Meta-Analyses in R with the metafor Package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Duval, S.; Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 8 December 2025).
- Atkins, D.; Best, D.; Briss, P.A.; Eccles, M.; Falck-Ytter, Y.; Flottorp, S.; Guyatt, G.H.; Harbour, R.T.; Haugh, M.C.; Henry, D.; et al. Grading quality of evidence and strength of recommendations. BMJ 2004, 328, 1490. [Google Scholar] [CrossRef]
- Feng, J.N.; Hu, A.J.; Xu, L.; Liu, L.L.; Liu, G.Z.; Wang, J.X.; Gao, P.; Liu, C.R.; Wang, S.F.; Zhan, S.Y. Prevalence and incidence of pseudomyxoma peritonei in urban China: A nationwide retrospective cohort study. Eur. J. Surg. Oncol. 2022, 48, 2226–2232. [Google Scholar] [CrossRef] [PubMed]
- Oemrawsingh, A.; de Boer, N.L.; Brandt-Kerkhof, A.R.M.; Verhoef, C.; Burger, J.W.A.; Madsen, E.V.E. Short-term complications in elderly patients undergoing CRS and HIPEC: A single center’s initial experience. Eur. J. Surg. Oncol. 2019, 45, 383–388. [Google Scholar] [CrossRef]
- Turgeon, M.K.; Gamboa, A.C.; Lee, R.M.; Zaidi, M.Y.; Kimbrough, C.; Grotz, T.; Fournier, K.; Powers, B.; Dineen, S.; Veerapong, J.; et al. The Intersection of Age and Tumor Biology with Postoperative Outcomes in Patients After Cytoreductive Surgery and HIPEC. Ann. Surg. Oncol. 2020, 27, 4894–4907. [Google Scholar] [CrossRef]
- Saxena, A.; Yan, T.D.; Chua, T.C.; Morris, D.L. Critical assessment of risk factors for complications after cytoreductive surgery and perioperative intraperitoneal chemotherapy for pseudomyxoma peritonei. Ann. Surg. Oncol. 2010, 17, 1291–1301. [Google Scholar] [CrossRef]
- Youssef, H.; Newman, C.; Chandrakumaran, K.; Mohamed, F.; Cecil, T.D.; Moran, B.J. Operative findings, early complications, and long-term survival in 456 patients with pseudomyxoma peritonei syndrome of appendiceal origin. Dis. Colon Rectum 2011, 54, 293–299. [Google Scholar] [CrossRef]
- Wei, X.; Liao, P.; Chen, X.; Wu, W. Risk factors affecting prognosis in pseudomyxoma peritonei patients after hyperthermic intraperitoneal chemotherapy and cytoreductive surgery: A systematic review and meta-analysis. Int. J. Surg. 2025, 111, 9622–9633. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Wagle, N.S.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 233–254. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, G.; He, J.; Ren, S.; Wu, F.; Zhang, J.; Wang, F. Gender differences in colorectal cancer survival: A meta-analysis. Int. J. Cancer 2017, 141, 1942–1949. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Li, Y.; Pu, H.; Xu, Y.; Chen, J.; Xu, H.; Wei, H.; Liang, G.; Ma, R.; Feng, J. Optimal peritoneal cancer index cutoff point for predicting surgical resectability of pseudomyxoma peritonei in treatment-naive patients. World J. Surg. Oncol. 2024, 22, 39. [Google Scholar] [CrossRef]
- Flicek, K.; Ashfaq, A.; Johnson, C.D.; Menias, C.; Bagaria, S.; Wasif, N. Correlation of Radiologic with Surgical Peritoneal Cancer Index Scores in Patients with Pseudomyxoma Peritonei and Peritoneal Carcinomatosis: How Well Can We Predict Resectability? J. Gastrointest. Surg. 2016, 20, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Wagner, P.L.; Austin, F.; Sathaiah, M.; Magge, D.; Maduekwe, U.; Ramalingam, L.; Jones, H.L.; Holtzman, M.P.; Ahrendt, S.A.; Zureikat, A.H.; et al. Significance of serum tumor marker levels in peritoneal carcinomatosis of appendiceal origin. Ann. Surg. Oncol. 2013, 20, 506–514. [Google Scholar] [CrossRef]
- Lopes, A.; de Mello, E.S.; Mendoza Lopez, R.V.; Leonardi, P.C.; Ribeiro, U., Jr. Prognostic Impact of Pathology, Cytoreduction, and Tumor Markers in Pseudomyxoma Peritonei. J. Surg. Res. 2022, 274, 68–76. [Google Scholar] [CrossRef]
- Liang, L.; Fang, J.; Han, X.; Zhai, X.; Song, Y.; Lu, Y.; Zhang, Q.; Ma, R. Prognostic Value of CEA, CA19-9, CA125, CA724, and CA242 in Serum and Ascites in Pseudomyxoma Peritonei. Front. Oncol. 2021, 11, 594763. [Google Scholar] [CrossRef]
- Wang, B.; Ma, R.; Rao, B.; Xu, H. Serum and ascites tumor markers in the diagnostic and prognostic prediction for appendiceal pseudomyxoma peritonei. BMC Cancer 2023, 23, 90. [Google Scholar] [CrossRef]
- Kozman, M.A.; Fisher, O.M.; Rebolledo, B.J.; Valle, S.J.; Alzahrani, N.; Liauw, W.; Morris, D.L. CA 19-9 to peritoneal carcinomatosis index (PCI) ratio is prognostic in patients with epithelial appendiceal mucinous neoplasms and peritoneal dissemination undergoing cytoreduction surgery and intraperitoneal chemotherapy: A retrospective cohort study. Eur. J. Surg. Oncol. 2017, 43, 2299–2307. [Google Scholar] [CrossRef]
- Nummela, P.; Leinonen, H.; Järvinen, P.; Thiel, A.; Järvinen, H.; Lepistö, A.; Ristimäki, A. Expression of CEA, CA19-9, CA125, and EpCAM in pseudomyxoma peritonei. Hum. Pathol. 2016, 54, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Moran, B.; Samuel, M.V.; Lord, A. ASO Author Reflections: The Paradox of Surgery for Pseudomyxoma Peritonei of Appendiceal Origin-Sometimes the Most Extensive Operations for Histologically Bland Disease. Ann. Surg. Oncol. 2025, 32, 249–250. [Google Scholar] [CrossRef]
- Carr, N.J.; Cecil, T.D.; Mohamed, F.; Sobin, L.H.; Sugarbaker, P.H.; González-Moreno, S.; Taflampas, P.; Chapman, S.; Moran, B.J. A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am. J. Surg. Pathol. 2016, 40, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Baratti, D.; Kusamura, S.; Milione, M.; Bruno, F.; Guaglio, M.; Deraco, M. Validation of the Recent PSOGI Pathological Classification of Pseudomyxoma Peritonei in a Single-Center Series of 265 Patients Treated by Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy. Ann. Surg. Oncol. 2018, 25, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Nagtegaal, I.D.; Odze, R.D.; Klimstra, D.; Paradis, V.; Rugge, M.; Schirmacher, P.; Washington, K.M.; Carneiro, F.; Cree, I.A. The 2019 WHO classification of tumours of the digestive system. Histopathology 2020, 76, 182–188. [Google Scholar] [CrossRef]
- Yan, F.; Lin, Y.; Zhou, Q.; Chang, H.; Li, Y. Pathological prognostic factors of pseudomyxoma peritonei: Comprehensive clinicopathological analysis of 155 cases. Hum. Pathol. 2020, 97, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Arjona-Sanchez, A.; Martinez-López, A.; Moreno-Montilla, M.T.; Mulsow, J.; Lozano-Lominchar, P.; Martínez-Torres, B.; Rau, B.; Canbay, E.; Sommariva, A.; Milione, M.; et al. External multicentre validation of pseudomyxoma peritonei PSOGI-Ki67 classification. Eur. J. Surg. Oncol. 2023, 49, 1481–1488. [Google Scholar] [CrossRef]
- Arjona-Sánchez, Á.; Martínez-López, A.; Valenzuela-Molina, F.; Rufián-Andújar, B.; Rufián-Peña, S.; Casado-Adam, Á.; Sánchez-Hidalgo, J.M.; Rodríguez-Ortiz, L.; Medina-Fernández, F.J.; Díaz-López, C.; et al. A Proposal for Modification of the PSOGI Classification According to the Ki-67 Proliferation Index in Pseudomyxoma Peritonei. Ann. Surg. Oncol. 2022, 29, 126–136. [Google Scholar] [CrossRef]
- Murage, N.W.; Ahmed, N.M.; Underwood, T.J.; Walters, Z.S.; Breininger, S.P. The genetic profile and molecular subtypes of human pseudomyxoma peritonei and appendiceal mucinous neoplasms: A systematic review. Cancer Metastasis Rev. 2023, 42, 335–359. [Google Scholar] [CrossRef]
- Nummela, P.; Saarinen, L.; Thiel, A.; Järvinen, P.; Lehtonen, R.; Lepistö, A.; Järvinen, H.; Aaltonen, L.A.; Hautaniemi, S.; Ristimäki, A. Genomic profile of pseudomyxoma peritonei analyzed using next-generation sequencing and immunohistochemistry. Int. J. Cancer 2015, 136, E282–E289. [Google Scholar] [CrossRef]
- Saarinen, L.; Nummela, P.; Thiel, A.; Lehtonen, R.; Järvinen, P.; Järvinen, H.; Aaltonen, L.A.; Lepistö, A.; Hautaniemi, S.; Ristimäki, A. Multiple components of PKA and TGF-β pathways are mutated in pseudomyxoma peritonei. PLoS ONE 2017, 12, e0174898. [Google Scholar] [CrossRef]
- Gibson, J.; Pengelly, R.J.; Mirandari, A.; Boukas, K.; Stanford, S.; Cecil, T.D.; Mohamed, F.; Dayal, S.P.; Tzivanakis, A.; Moran, B.J.; et al. Targeted Genetic Sequencing Analysis of 223 Cases of Pseudomyxoma Peritonei Treated by Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy Shows Survival Related to GNAS and KRAS Status. Cancer Med. 2024, 13, e70340. [Google Scholar] [CrossRef]
- Noguchi, R.; Yano, H.; Gohda, Y.; Suda, R.; Igari, T.; Ohta, Y.; Yamashita, N.; Yamaguchi, K.; Terakado, Y.; Ikenoue, T.; et al. Molecular profiles of high-grade and low-grade pseudomyxoma peritonei. Cancer Med. 2015, 4, 1809–1816. [Google Scholar] [CrossRef]
- Gleeson, E.M.; Feldman, R.; Mapow, B.L.; Mackovick, L.T.; Ward, K.M.; Morano, W.F.; Rubin, R.R.; Bowne, W.B. Appendix-derived Pseudomyxoma Peritonei (PMP): Molecular Profiling Toward Treatment of a Rare Malignancy. Am. J. Clin. Oncol. 2018, 41, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Borrego, M.C.; Granados-Rodríguez, M.; Bura, F.I.; Martínez-López, A.; Rufián-Andújar, B.; Valenzuela-Molina, F.; Rodríguez-Ortiz, L.; Haro-Yuste, S.; Moreno-Serrano, A.; Ortega-Salas, R.; et al. Antitumor effect of a small-molecule inhibitor of KRAS(G12D) in xenograft models of mucinous appendicular neoplasms. Exp. Hematol. Oncol. 2023, 12, 102. [Google Scholar] [CrossRef]
- Martínez-Quintanilla, J.; Cabot, D.; Sabia, D.; Arqués, O.; Vergés, J.; Chicote, I.; Bijelic, L.; Cabellos, L.; Alcántara, A.M.; Ramos, I.; et al. Precision Oncology and Systemic Targeted Therapy in Pseudomyxoma Peritonei. Clin. Cancer Res. 2024, 30, 4082–4099. [Google Scholar] [CrossRef]
- Yan, T.D.; Black, D.; Savady, R.; Sugarbaker, P.H. A systematic review on the efficacy of cytoreductive surgery and perioperative intraperitoneal chemotherapy for pseudomyxoma peritonei. Ann. Surg. Oncol. 2007, 14, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Floriano, I.; Silvinato, A.; Reis, J.C.; Cafalli, C.; Bernardo, W.M. Efficacy and safety in the use of intraperitoneal hyperthermia chemotherapy and peritoneal cytoreductive surgery for pseudomyxoma peritonei from appendiceal neoplasm: A systematic review. Clinics 2022, 77, 100039. [Google Scholar] [CrossRef]
- Kepenekian, V.; Bhatt, A.; Péron, J.; Alyami, M.; Benzerdjeb, N.; Bakrin, N.; Falandry, C.; Passot, G.; Rousset, P.; Glehen, O. Advances in the management of peritoneal malignancies. Nat. Rev. Clin. Oncol. 2022, 19, 698–718. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.