Prediction of 177Lu-DOTATATE Therapy Outcomes in Neuroendocrine Tumor Patients Using Semi-Automatic Tumor Delineation on 68Ga-DOTATATE PET/CT
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Patient Assessments
2.3. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Imaging Parameters
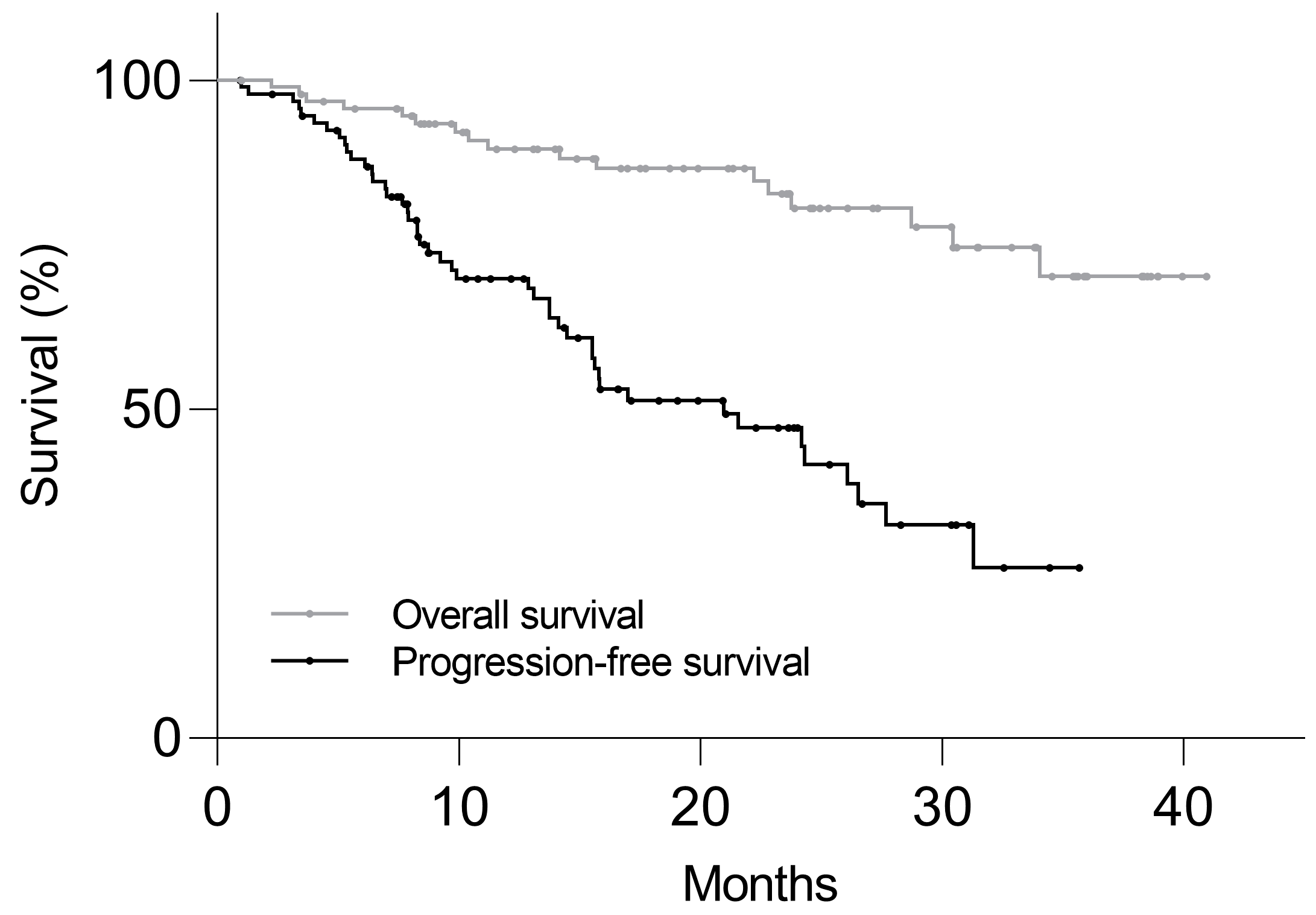
3.3. Follow-Up
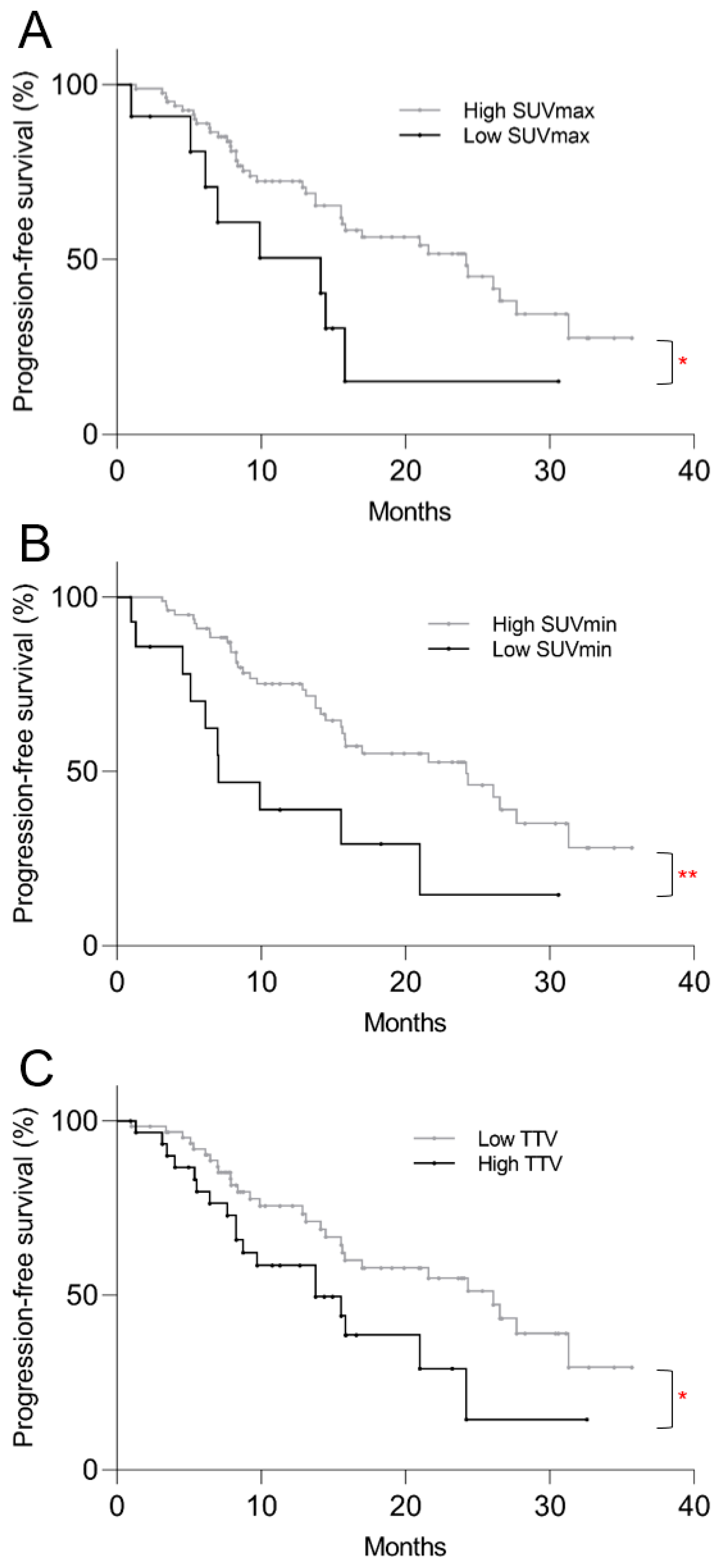
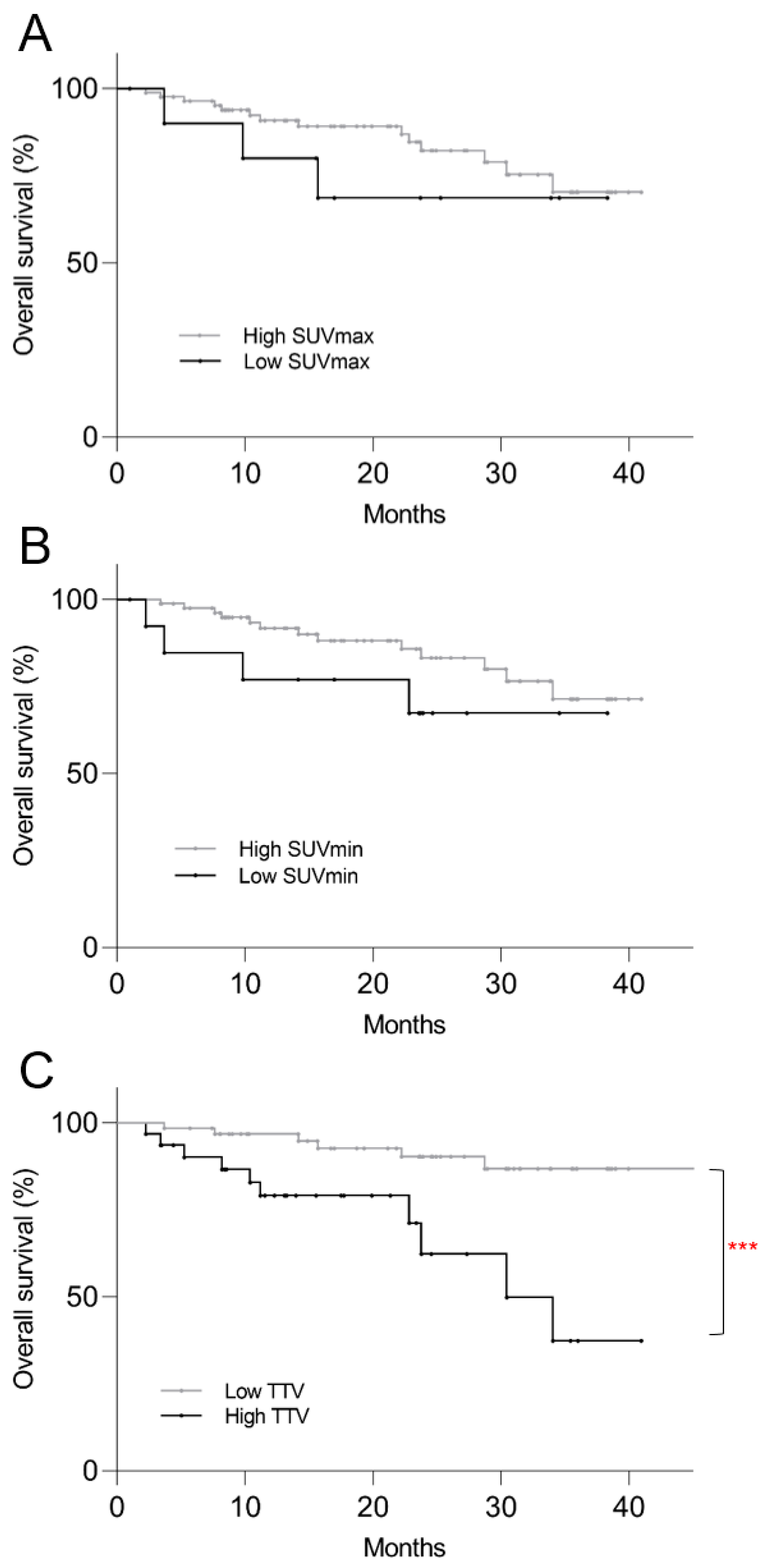
3.4. Survival Analysis
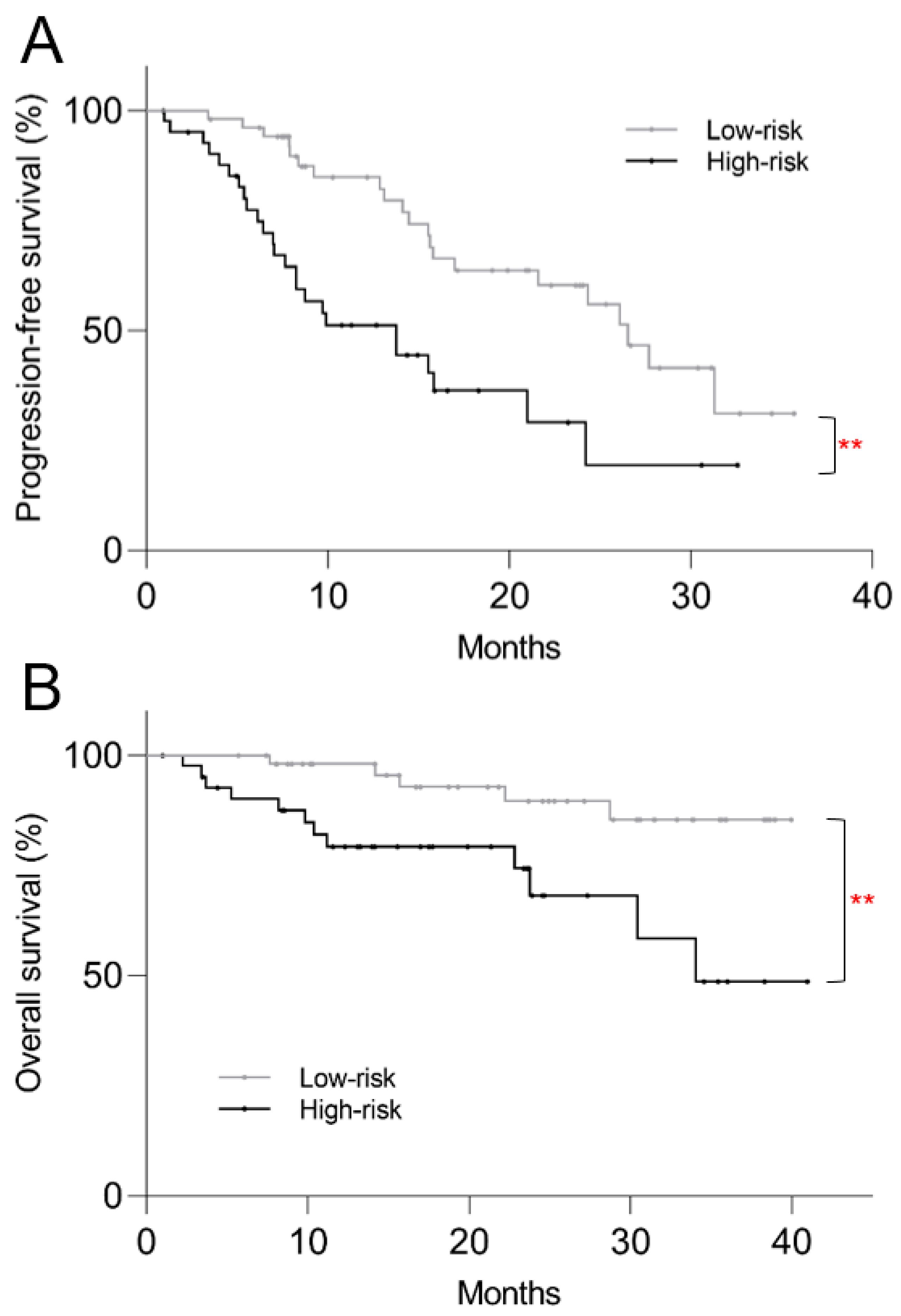
3.5. Post Hoc Kaplan–Meier Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients with Neuroendocrine Tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Hassan, M.; Phan, A.; Dagohoy, C.; Leary, C.; Mares, J.E.; Abdalla, E.K.; Fleming, J.B.; Vauthey, J.N.; Rashid, A.; et al. One hundred years after “carcinoid”: Epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J. Clin. Oncol. 2008, 26, 3063–3072. [Google Scholar] [CrossRef] [PubMed]
- Del Arco, C.D.; Sastre, J.; Peinado, P.; Díaz, Á.; Medina, L.O.; Fernández Aceñero, M.J. Neuroendocrine Neoplasms in Rare Locations: Clinicopathological Features and Review of the Literature. Indian J. Endocrinol. Metab. 2018, 22, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Klöppel, G. Neuroendocrine Neoplasms: Dichotomy, Origin and Classifications. Visc. Med. 2017, 33, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.R.; Li, T.; Ter-Minassian, M.; Yang, J.; Chan, J.A.; Brais, L.K.; Masugi, Y.; Thiaglingam, A.; Brooks, N.; Nishihara, R.; et al. Association between Somatostatin Receptor Expression and Clinical Outcomes in Neuroendocrine Tumors. Pancreas 2016, 45, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.H.; Goldner, W.S.; Benson, A.B.; Bergsland, E.; Blaszkowsky, L.S.; Brock, P.; Chan, J.; Das, S.; Dickson, P.V.; Fanta, P.; et al. Neuroendocrine and Adrenal Tumors, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2021, 19, 839–868. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of (177)Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Kipnis, S.T.; Hung, M.; Kumar, S.; Heckert, J.M.; Lee, H.; Bennett, B.; Soulen, M.C.; Pryma, D.A.; Mankoff, D.A.; Metz, D.C.; et al. Laboratory, Clinical, and Survival Outcomes Associated with Peptide Receptor Radionuclide Therapy in Patients with Gastroenteropancreatic Neuroendocrine Tumors. JAMA Netw. Open 2021, 4, e212274. [Google Scholar] [CrossRef]
- Binderup, T.; Knigge, U.; Johnbeck, C.B.; Loft, A.; Berthelsen, A.K.; Oturai, P.; Mortensen, J.; Federspiel, B.; Langer, S.W.; Kjaer, A. (18)F-FDG PET is Superior to WHO Grading as a Prognostic Tool in Neuroendocrine Neoplasms and Useful in Guiding PRRT: A Prospective 10-Year Follow-up Study. J. Nucl. Med. 2021, 62, 808–815. [Google Scholar] [CrossRef]
- Bodei, L.; Kidd, M.S.; Singh, A.; van der Zwan, W.A.; Severi, S.; Drozdov, I.A.; Cwikla, J.; Baum, R.P.; Kwekkeboom, D.J.; Paganelli, G.; et al. PRRT genomic signature in blood for prediction of (177)Lu-octreotate efficacy. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1155–1169. [Google Scholar] [CrossRef]
- Das, S.; Chauhan, A.; Du, L.; Thomas, K.E.; Jacob, A.; Schad, A.; Jain, S.; Jessop, A.; Shah, C.; Eisner, D.; et al. External Validation of a Clinical Score for Patients with Neuroendocrine Tumors Under Consideration for Peptide Receptor Radionuclide Therapy. JAMA Netw. Open 2022, 5, e2144170. [Google Scholar] [CrossRef] [PubMed]
- Bodei, L.; Raj, N.; Do, R.K.; Mauguen, A.; Krebs, S.; Reidy-Lagunes, D.; Schöder, H. Interim Analysis of a Prospective Validation of 2 Blood-Based Genomic Assessments (PPQ and NETest) to Determine the Clinical Efficacy of (177)Lu-DOTATATE in Neuroendocrine Tumors. J. Nucl. Med. 2023, 64, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Lee, O.N.Y.; Tan, K.V.; Tripathi, V.; Yuan, H.; Chan, W.W.; Chiu, K.W.H. The Role of 68 Ga-DOTA-SSA PET/CT in the Management and Prediction of Peptide Receptor Radionuclide Therapy Response for Patients with Neuroendocrine Tumors: A Systematic Review and Meta-analysis. Clin. Nucl. Med. 2022, 47, 781–793. [Google Scholar] [CrossRef] [PubMed]
- Hope, T.A.; Allen-Auerbach, M.; Bodei, L.; Calais, J.; Dahlbom, M.; Dunnwald, L.K.; Graham, M.M.; Jacene, H.A.; Heath, C.L.; Mittra, E.S.; et al. SNMMI Procedure Standard/EANM Practice Guideline for SSTR PET: Imaging Neuroendocrine Tumors. J. Nucl. Med. 2023, 64, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Olsen, I.H.; Langer, S.W.; Federspiel, B.H.; Oxbøl, J.; Loft, A.; Berthelsen, A.K.; Mortensen, J.; Oturai, P.; Knigge, U.; Kjær, A. (68)Ga-DOTATOC PET and gene expression profile in patients with neuroendocrine carcinomas: Strong correlation between PET tracer uptake and gene expression of somatostatin receptor subtype 2. Am. J. Nucl. Med. Mol. Imaging 2016, 6, 59–72. [Google Scholar] [PubMed]
- Öksüz, M.; Winter, L.; Pfannenberg, C.; Reischl, G.; Müssig, K.; Bares, R.; Dittmann, H. Peptide receptor radionuclide therapy of neuroendocrine tumors with (90)Y-DOTATOC: Is treatment response predictable by pre-therapeutic uptake of (68)Ga-DOTATOC? Diagn. Interv. Imaging 2014, 95, 289–300. [Google Scholar] [CrossRef]
- Sharma, R.; Wang, W.M.; Yusuf, S.; Evans, J.; Ramaswami, R.; Wernig, F.; Frilling, A.; Mauri, F.; Al-Nahhas, A.; Aboagye, E.O.; et al. (68)Ga-DOTATATE PET/CT parameters predict response to peptide receptor radionuclide therapy in neuroendocrine tumours. Radiother. Oncol. 2019, 141, 108–115. [Google Scholar] [CrossRef]
- Carlsen, E.A.; Johnbeck, C.B.; Loft, M.; Pfeifer, A.; Oturai, P.; Langer, S.W.; Knigge, U.; Ladefoged, C.N.; Kjaer, A. Semiautomatic Tumor Delineation for Evaluation of (64)Cu-DOTATATE PET/CT in Patients with Neuroendocrine Neoplasms: Prognostication Based on Lowest Lesion Uptake and Total Tumor Volume. J. Nucl. Med. 2021, 62, 1564–1570. [Google Scholar] [CrossRef]
- Pauwels, E.; Dekervel, J.; Verslype, C.; Clement, P.M.; Dooms, C.; Baete, K.; Goffin, K.; Jentjens, S.; Van Laere, K.; Van Cutsem, E.; et al. [(68)Ga]Ga-DOTATATE-avid tumor volume, uptake and inflammation-based index correlate with survival in neuroendocrine tumor patients treated with [(177)Lu]Lu-DOTATATE PRRT. Am. J. Nucl. Med. Mol. Imaging 2022, 12, 152–162. [Google Scholar]
- Ebbers, S.C.; Heimgartner, M.; Barentsz, M.W.; van Leeuwaarde, R.S.; van Treijen, M.J.C.; Lam, M.; Braat, A. Gallium-68-somatostatin receptor PET/CT parameters as potential prognosticators for clinical time to progression after peptide receptor radionuclide therapy: A cohort study. Eur. J. Hybrid Imaging 2021, 5, 22. [Google Scholar] [CrossRef]
- Niman, R.; Buteau, J.P.; Kruzer, A.; Turcotte, É.; Nelson, A. Evaluation of a semi-automated whole body PET segmentation method applied to Diffuse Large B Cell Lymphoma. J. Nucl. Med. 2018, 59, 592. [Google Scholar]
- Wahl, R.L.; Jacene, H.; Kasamon, Y.; Lodge, M.A. From RECIST to PERCIST: Evolving Considerations for PET response criteria in solid tumors. J. Nucl. Med. 2009, 50 (Suppl. S1), 122s–150s. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.Y.; Kim, S. Determination of cutoff values for biomarkers in clinical studies. Precis. Future Med. 2020, 4, 2–8. [Google Scholar] [CrossRef]
- Kratochwil, C.; Stefanova, M.; Mavriopoulou, E.; Holland-Letz, T.; Dimitrakopoulou-Strauss, A.; Afshar-Oromieh, A.; Mier, W.; Haberkorn, U.; Giesel, F.L. SUV of [68Ga]DOTATOC-PET/CT Predicts Response Probability of PRRT in Neuroendocrine Tumors. Mol. Imaging Biol. 2015, 17, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Wulfert, S.; Kratochwil, C.; Choyke, P.L.; Afshar-Oromieh, A.; Mier, W.; Kauczor, H.U.; Schenk, J.P.; Haberkorn, U.; Giesel, F.L. Multimodal imaging for early functional response assessment of (90)Y-/ (177)Lu-DOTATOC peptide receptor targeted radiotherapy with DW-MRI and (68)Ga-DOTATOC-PET/CT. Mol. Imaging Biol. 2014, 16, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Schmid, H.A.; Lambertini, C.; van Vugt, H.H.; Barzaghi-Rinaudo, P.; Schäfer, J.; Hillenbrand, R.; Sailer, A.W.; Kaufmann, M.; Nuciforo, P. Monoclonal antibodies against the human somatostatin receptor subtypes 1-5: Development and immunohistochemical application in neuroendocrine tumors. Neuroendocrinology 2012, 95, 232–247. [Google Scholar] [CrossRef]
- Ohlendorf, F.; Henkenberens, C.; Brunkhorst, T.; Ross, T.L.; Christiansen, H.; Bengel, F.M.; Derlin, T. Volumetric 68Ga-DOTA-TATE PET/CT for assessment of whole-body tumor burden as a quantitative imaging biomarker in patients with metastatic gastroenteropancreatic neuroendocrine tumors. Q. J. Nucl. Med. Mol. Imaging 2022, 66, 361–371. [Google Scholar] [CrossRef]
- Pauwels, E.; Van Binnebeek, S.; Vandecaveye, V.; Baete, K.; Vanbilloen, H.; Koole, M.; Mottaghy, F.M.; Haustermans, K.; Clement, P.M.; Nackaerts, K.; et al. Inflammation-Based Index and (68)Ga-DOTATOC PET-Derived Uptake and Volumetric Parameters Predict Outcome in Neuroendocrine Tumor Patients Treated with (90)Y-DOTATOC. J. Nucl. Med. 2020, 61, 1014–1020. [Google Scholar] [CrossRef]
- Durmo, R.; Filice, A.; Fioroni, F.; Cervati, V.; Finocchiaro, D.; Coruzzi, C.; Besutti, G.; Fanello, S.; Frasoldati, A.; Versari, A. Predictive and Prognostic Role of Pre-Therapy and Interim 68Ga-DOTATOC PET/CT Parameters in Metastatic Advanced Neuroendocrine Tumor Patients Treated with PRRT. Cancers 2022, 14, 592. [Google Scholar] [CrossRef]
- Zhang, J.; Kulkarni, H.R.; Singh, A.; Niepsch, K.; Müller, D.; Baum, R.P. Peptide Receptor Radionuclide Therapy in Grade 3 Neuroendocrine Neoplasms: Safety and Survival Analysis in 69 Patients. J. Nucl. Med. 2019, 60, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Kinahan, P.E.; Fletcher, J.W. Positron emission tomography-computed tomography standardized uptake values in clinical practice and assessing response to therapy. Semin. Ultrasound CT MR. 2010, 31, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, C.; Ganeshan, B.; Endozo, R.; Wan, S.; Aldridge, M.D.; Groves, A.M.; Bomanji, J.B.; Gaze, M.N. Radiomics-Based Texture Analysis of (68)Ga-DOTATATE Positron Emission Tomography and Computed Tomography Images as a Prognostic Biomarker in Adults with Neuroendocrine Cancers Treated with (177)Lu-DOTATATE. Front. Oncol. 2021, 11, 686235. [Google Scholar] [CrossRef] [PubMed]
- Ortega, C.; Wong, R.K.S.; Schaefferkoetter, J.; Veit-Haibach, P.; Myrehaug, S.; Juergens, R.; Laidley, D.; Anconina, R.; Liu, A.; Metser, U. Quantitative (68)Ga-DOTATATE PET/CT Parameters for the Prediction of Therapy Response in Patients with Progressive Metastatic Neuroendocrine Tumors Treated with (177)Lu-DOTATATE. J. Nucl. Med. 2021, 62, 1406–1414. [Google Scholar] [CrossRef] [PubMed]
- Werner, R.A.; Ilhan, H.; Lehner, S.; Papp, L.; Zsótér, N.; Schatka, I.; Muegge, D.O.; Javadi, M.S.; Higuchi, T.; Buck, A.K.; et al. Pre-therapy Somatostatin Receptor-Based Heterogeneity Predicts Overall Survival in Pancreatic Neuroendocrine Tumor Patients Undergoing Peptide Receptor Radionuclide Therapy. Mol. Imaging Biol. 2019, 21, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Opalińska, M.; Morawiec-Sławek, K.; Kania-Kuc, A.; Al Maraih, I.; Sowa-Staszczak, A.; Hubalewska-Dydejczyk, A. Potential value of pre- and post-therapy [68Ga]Ga-DOTA-TATE PET/CT in the prognosis of response to PRRT in disseminated neuroendocrine tumors. Front. Endocrinol. 2022, 13, 929391. [Google Scholar] [CrossRef]
- Gabriel, M.; Oberauer, A.; Dobrozemsky, G.; Decristoforo, C.; Putzer, D.; Kendler, D.; Uprimny, C.; Kovacs, P.; Bale, R.; Virgolini, I.J. 68Ga-DOTA-Tyr3-octreotide PET for assessing response to somatostatin-receptor-mediated radionuclide therapy. J. Nucl. Med. 2009, 50, 1427–1434. [Google Scholar] [CrossRef]
- Soydal, Ç.; Peker, A.; Özkan, E.; Küçük, Ö.N.; Kir, M.K. The role of baseline Ga-68 DOTATATE positron emission tomography/computed tomography in the prediction of response to fixed-dose peptide receptor radionuclide therapy with Lu-177 DOTATATE. Turk. J. Med. Sci. 2016, 46, 409–413. [Google Scholar] [CrossRef]
- Weber, M.; Kessler, L.; Schaarschmidt, B.; Fendler, W.P.; Lahner, H.; Antoch, G.; Umutlu, L.; Herrmann, K.; Rischpler, C. Treatment-related changes in neuroendocrine tumors as assessed by textural features derived from (68)Ga-DOTATOC PET/MRI with simultaneous acquisition of apparent diffusion coefficient. BMC Cancer 2020, 20, 326. [Google Scholar] [CrossRef]
- Huizing, D.M.V.; Aalbersberg, E.A.; Versleijen, M.W.J.; Tesselaar, M.E.T.; Walraven, I.; Lahaye, M.J.; de Wit-van der Veen, B.J.; Stokkel, M.P.M. Early response assessment and prediction of overall survival after peptide receptor radionuclide therapy. Cancer Imaging 2020, 20, 57. [Google Scholar] [CrossRef]
- Marquis, H.; Willowson, K.P.; Bailey, D.L. Partial volume effect in SPECT & PET imaging and impact on radionuclide dosimetry estimates. Asia Ocean. J. Nucl. Med. Biol. 2023, 11, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Simpson, G.; Young, L.; Ford, J.; Dogan, N.; Wang, L. Impact of contouring variability on oncological PET radiomics features in the lung. Sci. Rep. 2020, 10, 369. [Google Scholar] [CrossRef] [PubMed]
- Kothari, G.; Woon, B.; Patrick, C.J.; Korte, J.; Wee, L.; Hanna, G.G.; Kron, T.; Hardcastle, N.; Siva, S. The impact of inter-observer variation in delineation on robustness of radiomics features in non-small cell lung cancer. Sci. Rep. 2022, 12, 12822. [Google Scholar] [CrossRef] [PubMed]
- Sadaghiani, M.S.; Rowe, S.P.; Sheikhbahaei, S. Applications of artificial intelligence in oncologic (18)F-FDG PET/CT imaging: A systematic review. Ann. Transl. Med. 2021, 9, 823. [Google Scholar] [CrossRef]
- Lee, H.; Murphy, C.; Mortani Barbosa, E.J., Jr. Prediction of Complication Risk in Computed Tomography-guided Thoracic Biopsy: A Prescription for Improving Procedure Safety. J. Thorac. Imaging 2023, 38, 88–96. [Google Scholar] [CrossRef]
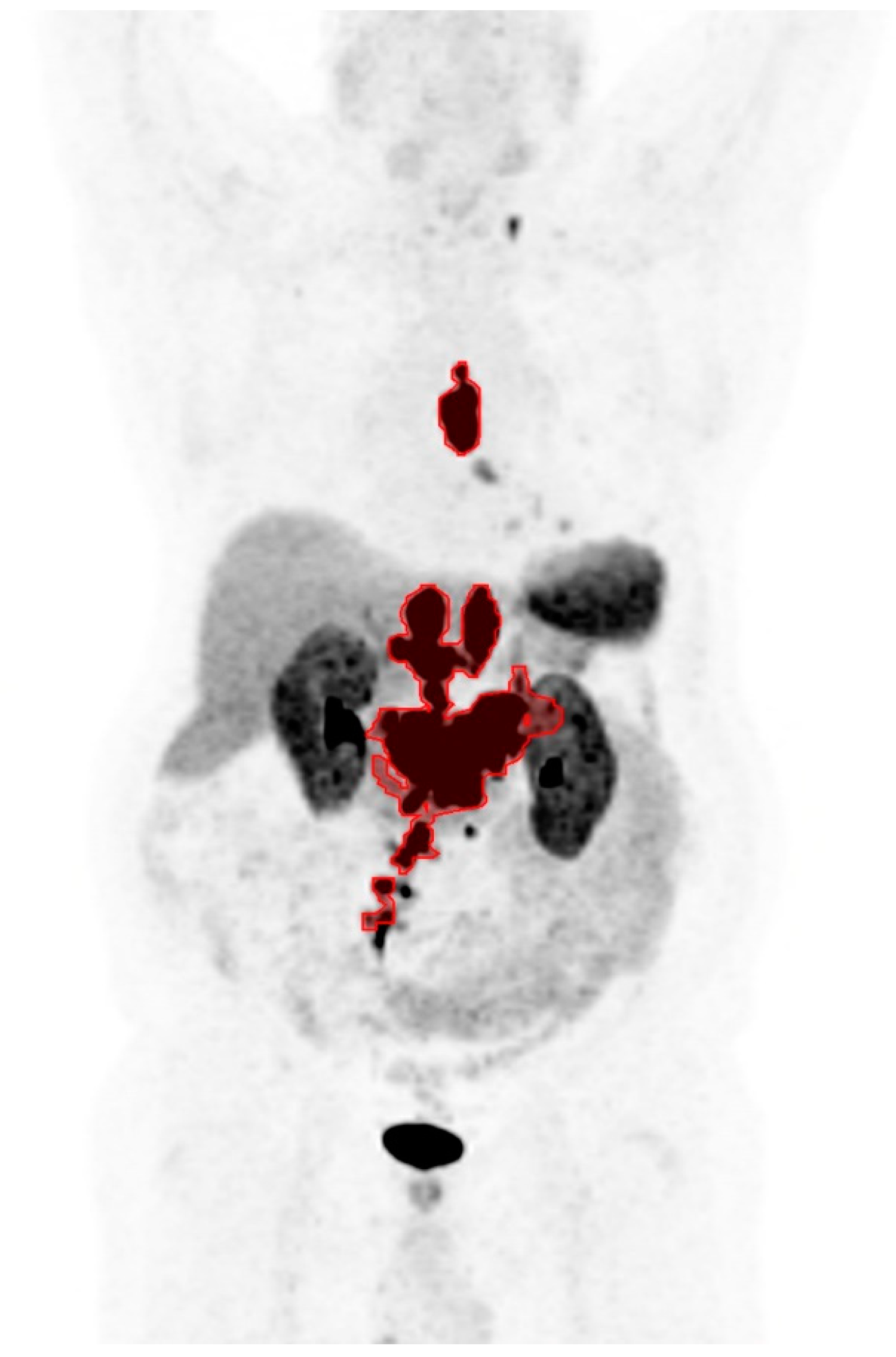
| Baseline Characteristics | Percentage (n = 94) |
|---|---|
| Gender | |
| Male | 50% |
| Female | 50% |
| Age (years) | |
| ≤50 | 17% |
| >50 and ≤60 | 31% |
| >60 and ≤70 | 26% |
| >70 | 27% |
| Primary tumor site | |
| Small intestine | 36% |
| Pancreas | 35% |
| Colon | 9% |
| Lung | 9% |
| Other gastrointestinal | 3% |
| Pheochromocytoma/Paraganglioma | 2% |
| Unknown | 6% |
| Grade for gastroenteropancreatic tumors | |
| G1 | 28% |
| G2 | 61% |
| G3 | 11% |
| Ki-67 index | |
| Median | 8% |
| Range | 0–95% |
| Number of 177Lu-DOTATATE cycles | |
| 4 | 74% |
| 3 | 11% |
| 2 | 7% |
| 1 | 7% |
| Parameter | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Low SUVmax | 2.3 | 1.0–4.9 | 0.037 | 1.5 | 0.6–4.0 | 0.418 |
| Low SUVmin | 2.8 | 1.3–6.1 | 0.008 | 3.1 | 1.1–8.7 | 0.030 |
| High TTV | 1.9 | 1.0–3.5 | 0.037 | 2.4 | 1.3–4.7 | 0.007 |
| Parameter | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Low SUVmax | 1.7 | 0.5–5.9 | 0.412 | 1.7 | 0.3–9.1 | 0.530 |
| Low SUVmin | 1.7 | 0.5–6.0 | 0.393 | 3.0 | 0.5–18.1 | 0.233 |
| High TTV | 3.9 | 1.5–10.3 | 0.006 | 5.9 | 1.8–18.7 | 0.003 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Kipnis, S.T.; Niman, R.; O’Brien, S.R.; Eads, J.R.; Katona, B.W.; Pryma, D.A. Prediction of 177Lu-DOTATATE Therapy Outcomes in Neuroendocrine Tumor Patients Using Semi-Automatic Tumor Delineation on 68Ga-DOTATATE PET/CT. Cancers 2024, 16, 200. https://doi.org/10.3390/cancers16010200
Lee H, Kipnis ST, Niman R, O’Brien SR, Eads JR, Katona BW, Pryma DA. Prediction of 177Lu-DOTATATE Therapy Outcomes in Neuroendocrine Tumor Patients Using Semi-Automatic Tumor Delineation on 68Ga-DOTATATE PET/CT. Cancers. 2024; 16(1):200. https://doi.org/10.3390/cancers16010200
Chicago/Turabian StyleLee, Hwan, Sarit T. Kipnis, Remy Niman, Sophia R. O’Brien, Jennifer R. Eads, Bryson W. Katona, and Daniel A. Pryma. 2024. "Prediction of 177Lu-DOTATATE Therapy Outcomes in Neuroendocrine Tumor Patients Using Semi-Automatic Tumor Delineation on 68Ga-DOTATATE PET/CT" Cancers 16, no. 1: 200. https://doi.org/10.3390/cancers16010200
APA StyleLee, H., Kipnis, S. T., Niman, R., O’Brien, S. R., Eads, J. R., Katona, B. W., & Pryma, D. A. (2024). Prediction of 177Lu-DOTATATE Therapy Outcomes in Neuroendocrine Tumor Patients Using Semi-Automatic Tumor Delineation on 68Ga-DOTATATE PET/CT. Cancers, 16(1), 200. https://doi.org/10.3390/cancers16010200






