Melanin and Melanin-Functionalized Nanoparticles as Promising Tools in Cancer Research—A Review
Abstract
:Simple Summary
Abstract
1. Introduction
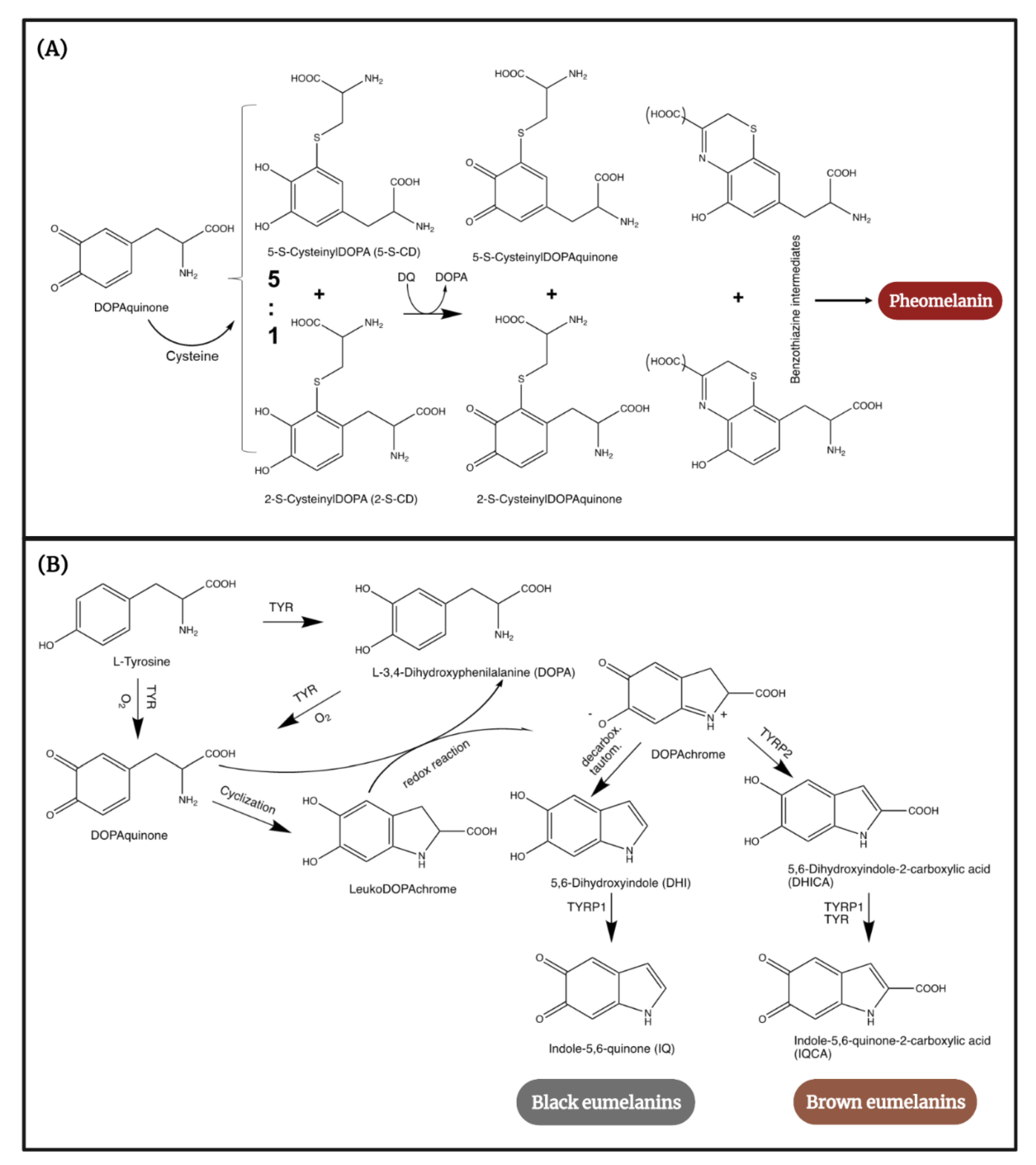
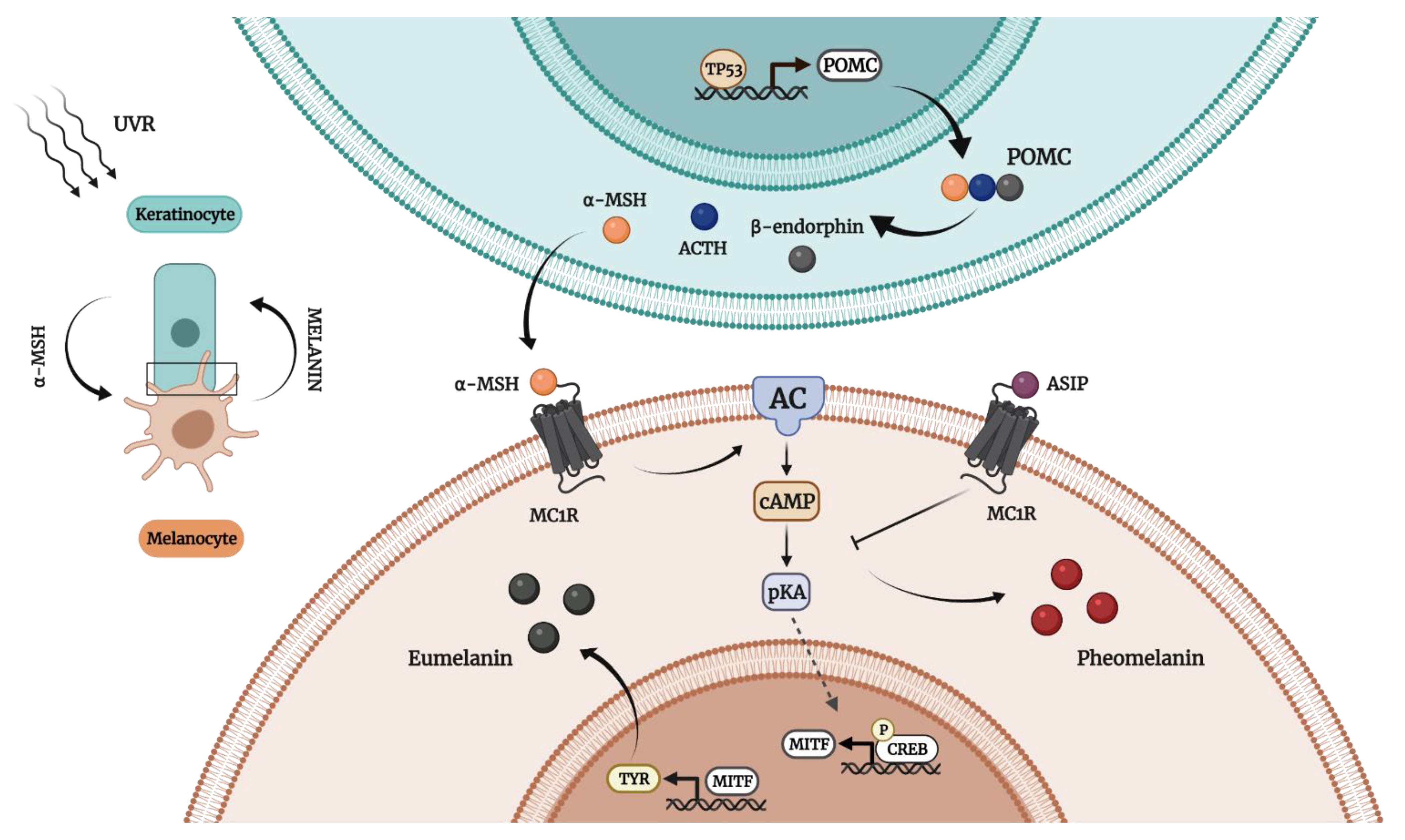
2. Human Melanogenesis in a Nutshell
3. Therapeutic Properties
3.1. Antioxidant Effect
3.2. Photoprotective Capacity
3.3. Anti-Inflammatory Properties
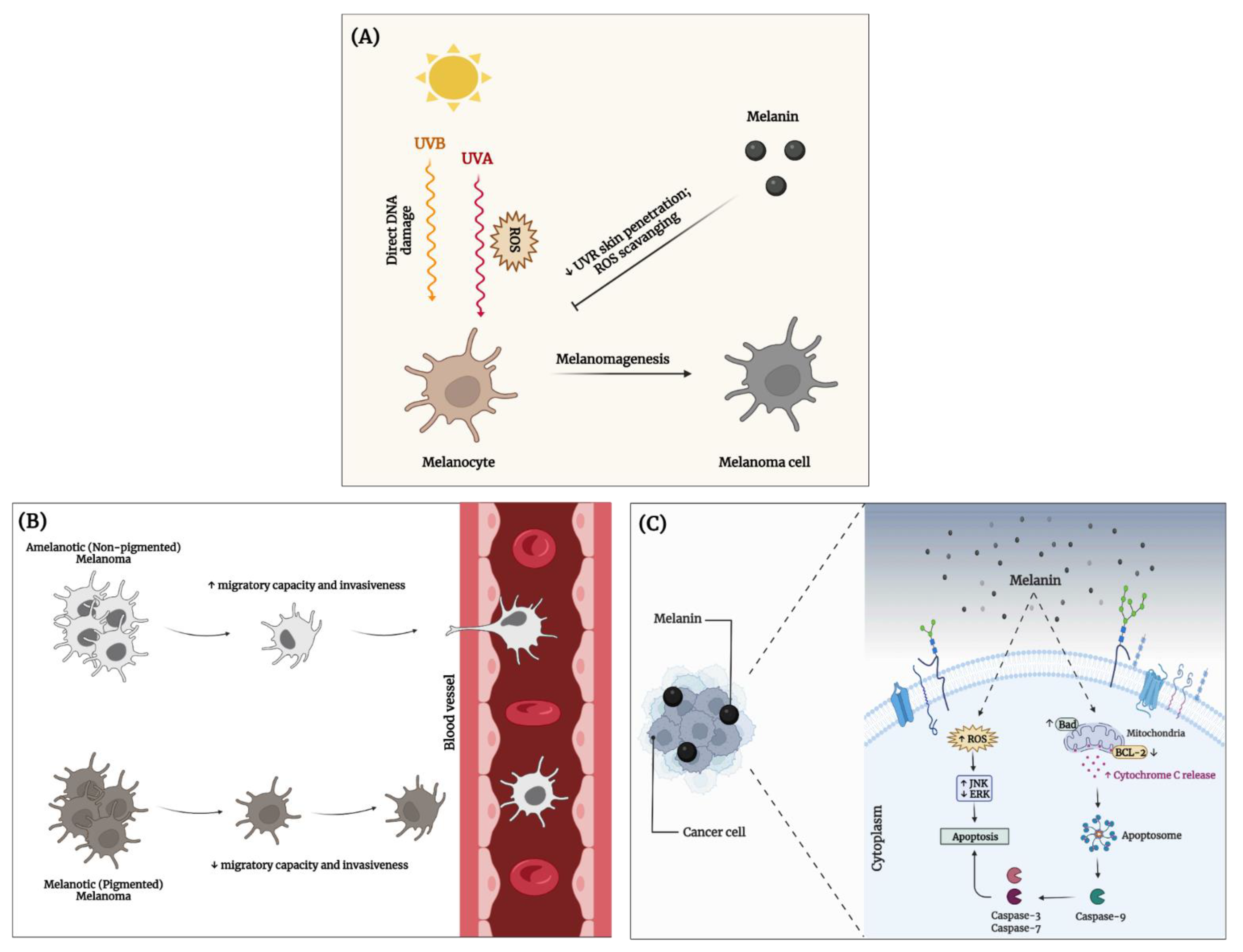
3.4. Anticancer Activity
4. Melanin-Based Targeted Cancer Therapy
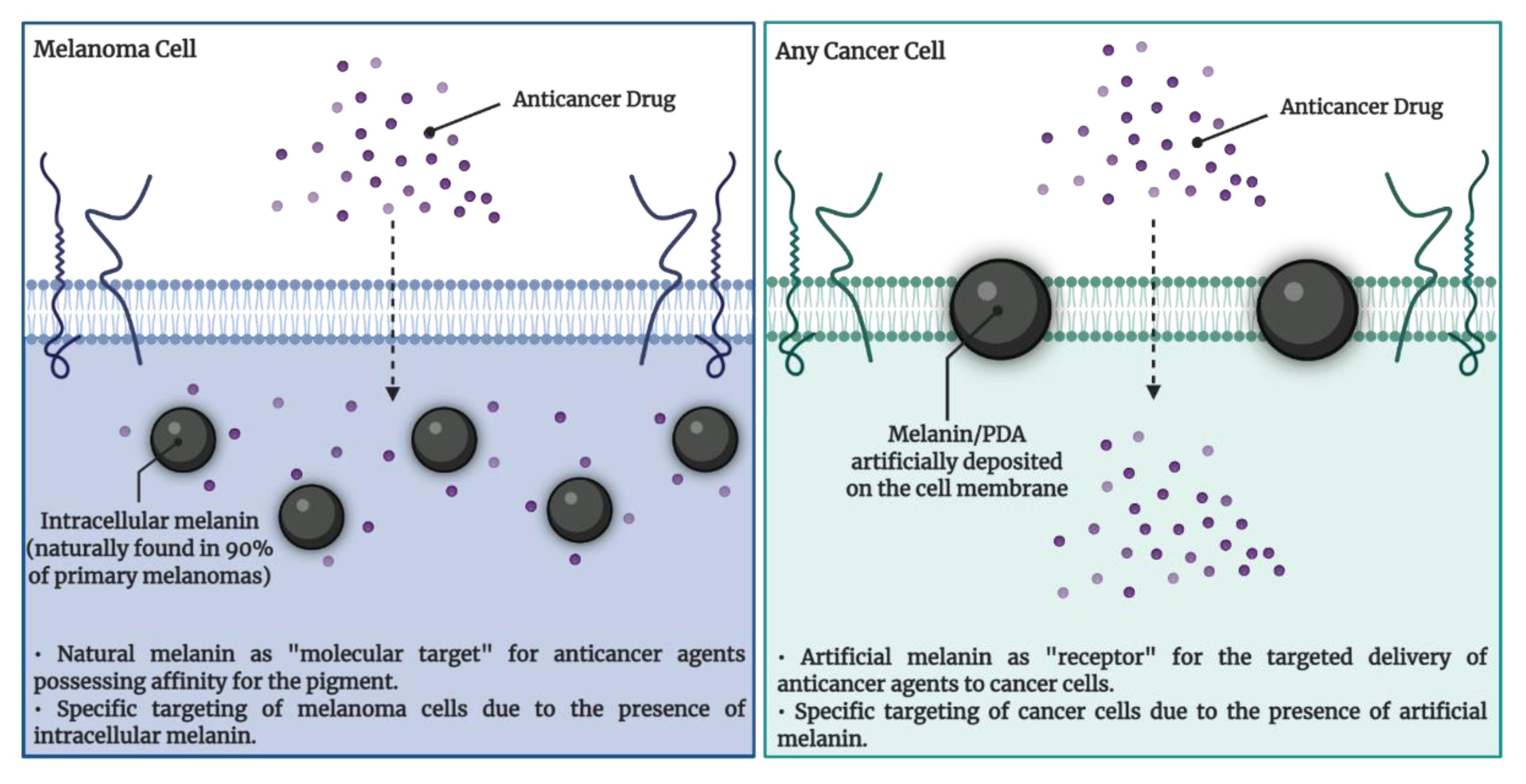
4.1. Melanin Targeting in Cancer Therapy
4.2. Melanin Nanoparticles (MEL-NPs) in Cancer Therapy
4.2.1. Synthesis of MEL-NPs
4.2.2. Applications of MEL-NPs in Cancer Therapy
Chemotherapy
Radio(pharmaceutical) Therapy
Phototherapy
Immunotherapy
Gene Therapy
Cancer Detection and Bio-Imaging
Nanotheranostics
4.2.3. Biosafety and Metabolism of MEL-NPs
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Hassanpour, S.H.; Dehghani, M. Review of cancer from perspective of molecular. J. Cancer Res. Pract. 2017, 4, 127–129. [Google Scholar] [CrossRef]
- Arem, H.; Loftfield, E. Cancer Epidemiology: A Survey of Modifiable Risk Factors for Prevention and Survivorship. Am. J. Lifestyle Med. 2018, 12, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Gavas, S.; Quazi, S.; Karpiński, T.M. Nanoparticles for cancer therapy: Current progress and challenges. Nanoscale Res. Lett. 2021, 16, 173. [Google Scholar] [CrossRef] [PubMed]
- Ulbrich, K.; Holá, K.; Šubr, V.; Bakandritsos, A.; Tuček, J.; Zbořil, R. Targeted drug delivery with polymers and magnetic nanoparticles: Covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 2016, 116, 5338–5431. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iranzo, J.; Martincorena, I.; Koonin, E.V. Cancer-mutation network and the number and specificity of driver mutations. Proc. Natl. Acad. Sci. USA 2018, 115, E6010–E6019. [Google Scholar] [CrossRef] [Green Version]
- Rangel-Sosa, M.M.; Aguilar-Córdova, E.; Rojas-Martínez, A. Immunotherapy and gene therapy as novel treatments for cancer. Colomb. Med. (Cali) 2017, 48, 138–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, S.; Zhao, P.; Yu, T.; Gu, N. Current applications and future prospects of nanotechnology in cancer immunotherapy. Cancer Biol. Med. 2019, 16, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Roma-Rodrigues, C.; Rivas-García, L.; Baptista, P.V.; Fernandes, A.R. Gene Therapy in Cancer Treatment: Why Go Nano? Pharmaceutics 2020, 12, 233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, A.; Kaur, K. Biological and Physical Applications of Silver Nanoparticles with Emerging Trends of Green Synthesis. In Engineered Nanomaterials—Health and Safety; Avramescu, S.M., Akhtar, K., Fierascu, I., Khan, S.B., Ali, F., Asiri, A.M., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Arya, G.; Mankamna Kumari, R.; Sharma, N.; Gupta, N.; Chandra, R.; Nimesh, S. Chapter 18—Polymeric nanocarriers for site-specific gene therapy. In Drug Targeting and Stimuli Sensitive Drug Delivery Systems; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2018; pp. 689–714. ISBN 9780128136898. [Google Scholar]
- Sousa, D.; Ferreira, D.; Rodrigues, J.L.; Rodrigues, L.R. Chapter 14—Nanotechnology in Targeted Drug Delivery and Therapeutics. In Applications of Targeted Nano Drugs and Delivery Systems; Mohapatra, S.S., Ranjan, S., Dasgupta, N., Mishra, R.K., Thomas, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 357–409. ISBN 9780128140291. [Google Scholar]
- Cheng, Z.; Li, M.; Dey, R.; Chen, Y. Nanomaterials for cancer therapy: Current progress and perspectives. J. Hematol. Oncol. 2021, 14, 1–27. [Google Scholar] [CrossRef] [PubMed]
- D’Ischia, M.; Napolitano, A.; Ball, V.; Chen, C.T.; Buehler, M.J. Polydopamine and eumelanin: From structure-property relationships to a unified tailoring strategy. Acc. Chem. Res. 2014, 47, 3541–3550. [Google Scholar] [CrossRef] [PubMed]
- Solano, F. Melanins: Skin Pigments and Much More—Types, Structural Models, Biological Functions, and Formation Routes. New J. Sci. 2014, 2014, 1–28. [Google Scholar] [CrossRef] [Green Version]
- Roy, S.; Rhim, J.W. New insight into melanin for food packaging and biotechnology applications. Crit. Rev. Food Sci. Nutr. 2021, 1, 1–27. [Google Scholar] [CrossRef] [PubMed]
- ElObeid, A.S.; Kamal-Eldin, A.; Abdelhalim, M.A.K.; Haseeb, A.M. Pharmacological Properties of Melanin and its Function in Health. Basic Clin. Pharmacol. Toxicol. 2017, 120, 515–522. [Google Scholar] [CrossRef] [PubMed]
- D’Ischia, M.; Wakamatsu, K.; Napolitano, A.; Briganti, S.; Garcia-Borron, J.C.; Kovacs, D.; Meredith, P.; Pezzella, A.; Picardo, M.; Sarna, T.; et al. Melanins and melanogenesis: Methods, standards, protocols. Pigment. Cell Melanoma Res. 2013, 26, 616–633. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, Y.; Liu, Y.; Pan, J.; Wang, J.; Man, F.; Zhang, W.; Liu, G. Melanin-Like Nanomaterials for Advanced Biomedical Applications: A Versatile Platform with Extraordinary Promise. Adv. Sci. 2020, 7, 1903129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solano, F. Photoprotection and skin pigmentation: Melanin-related molecules and some other new agents obtained from natural sources. Molecules 2020, 25, 1537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, C.; Fu, L.H.; Xu, H.; Wang, T.F.; Lin, J.; Huang, P. Melanin/polydopamine-based nanomaterials for biomedical applications. Sci. China Chem. 2019, 62, 162–188. [Google Scholar] [CrossRef]
- Blázquez-Castro, A.; Stockert, J.C. Biomedical overview of melanin. 1. Updating melanin biology and chemistry, physico-chemical properties, melanoma tumors, and photothermal therapy. Biocell 2021, 45, 849–862. [Google Scholar] [CrossRef]
- Cheng, W.; Zeng, X.; Chen, H.; Li, Z.; Zeng, W.; Mei, L.; Zhao, Y. Versatile Polydopamine Platforms: Synthesis and Promising Applications for Surface Modification and Advanced Nanomedicine. ACS Nano 2019, 13, 8537–8565. [Google Scholar] [CrossRef]
- Lynge, M.E.; Van Der Westen, R.; Postma, A.; Städler, B. Polydopamine—A nature-inspired polymer coating for biomedical science. Nanoscale 2011, 3, 4916–4928. [Google Scholar] [CrossRef] [PubMed]
- Ball, V. Polydopamine nanomaterials: Recent advances in synthesis methods and applications. Front. Bioeng. Biotechnol. 2018, 6, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kohri, M. Progress in polydopamine-based melanin mimetic materials for structural color generation. Sci. Technol. Adv. Mater. 2020, 21, 833–848. [Google Scholar] [CrossRef]
- Mrowczynski, R. Polydopamine-Based Multifunctional (Nano)materials for Cancer Therapy. ACS Appl. Mater. Interfaces 2018, 10, 7541–7561. [Google Scholar] [CrossRef] [PubMed]
- Cichorek, M.; Wachulska, M.; Stasiewicz, A.; Tymińska, A. Skin melanocytes: Biology and development. Postep. Dermatol. I Alergol. 2013, 30, 30–41. [Google Scholar] [CrossRef]
- Schroeder, R.L.; Double, K.L.; Gerber, J.P. Using Sepia melanin as a PD model to describe the binding characteristics of neuromelanin—A critical review. J. Chem. Neuroanat. 2015, 64–65, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Nasti, T.H.; Timares, L. Invited Review MC1R, Eumelanin and Pheomelanin: Their role in determining the susceptibility to skin cancer. Photochem. Photobiol. 2015, 91, 188–200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haining, R.L.; Achat-Mendes, C. Neuromelanin, one of the most overlooked molecules in modern medicine, is not a spectator. Neural Regen. Res. 2017, 12, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Zucca, F.A.; Basso, E.; Cupaioli, F.A.; Ferrari, E.; Sulzer, D.; Casella, L.; Zecca, L. Neuromelanin of the human substantia Nigra: An update. Neurotox. Res. 2014, 25, 13–23. [Google Scholar] [CrossRef]
- Pralea, I.E.; Moldovan, R.C.; Petrache, A.M.; Ilieș, M.; Hegheș, S.C.; Ielciu, I.; Nicoară, R.; Moldovan, M.; Ene, M.; Radu, M.; et al. From extraction to advanced analytical methods: The challenges of melanin analysis. Int. J. Mol. Sci. 2019, 20, 3943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Bino, S.; Duval, C.; Bernerd, F. Clinical and biological characterization of skin pigmentation diversity and its consequences on UV impact. Int. J. Mol. Sci. 2018, 19, 2668. [Google Scholar] [CrossRef] [Green Version]
- d’Ischia, M.; Wakamatsu, K.; Cicoira, F.; Di Mauro, E.; Garcia-Borron, J.C.; Commo, S.; Galván, I.; Ghanem, G.; Kenzo, K.; Meredith, P.; et al. Melanins and melanogenesis: From pigment cells to human health and technological applications. Pigment Cell Melanoma Res. 2015, 28, 520–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swope, V.B.; Abdel-Malek, Z.A. MC1R: Front and center in the bright side of dark eumelanin and DNA repair. Int. J. Mol. Sci. 2018, 19, 2667. [Google Scholar] [CrossRef] [Green Version]
- Maresca, V.; Flori, E.; Picardo, M. Skin phototype: A new perspective. Pigment Cell Melanoma Res. 2015, 28, 378–389. [Google Scholar] [CrossRef] [Green Version]
- Oren, M.; Bartek, J. The Sunny Side of p53. Cell 2007, 128, 826–828. [Google Scholar] [CrossRef] [Green Version]
- Meredith, P.; Sarna, T. The physical and chemical properties of eumelanin. Pigment Cell Res. 2006, 19, 572–594. [Google Scholar] [CrossRef] [PubMed]
- Barabanov, M.; Martyanov, G.S.; Pestov, A.V. Biological activity and synthesis of 5,6-dihydroxyindole-2-carboxylic acid—biosynthetic precursor of melanins (microreview). Chem. Heterocycl. Compd. 2021, 57, 417–419. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Shawkey, M.D.; Dhinojwala, A. Bioinspired Melanin-Based Optically Active Materials. Adv. Opt. Mater. 2020, 8, 1–15. [Google Scholar] [CrossRef]
- Zonios, G.; Dimou, A. Melanin optical properties provide evidence for chemical and structural disorder in vivo. Opt. Express 2008, 16, 8263. [Google Scholar] [CrossRef] [PubMed]
- Mostert, A.B.; Powell, B.J.; Pratt, F.L.; Hanson, G.R.; Sarna, T.; Gentle, I.R.; Meredith, P. Role of semiconductivity and ion transport in the electrical conduction of melanin. Proc. Natl. Acad. Sci. USA 2012, 109, 8943–8947. [Google Scholar] [CrossRef] [Green Version]
- Ou-Yang, H.; Stamatas, G.; Kollias, N. Spectral Responses of Melanin to Ultraviolet a Irradiation. J. Invest. Dermatol. 2004, 122, 492–496. [Google Scholar] [CrossRef] [Green Version]
- Zdybel, M.; Pilawa, B.; Buszman, E.; Wrześniok, D.; Krzyminiewski, R.; Kruczyński, Z. EPR Studies of DOPA-Melanin Complexes with Netilmicin and Cu(II) at Temperatures in the Range of 105–300 K. Appl. Magn. Reson. 2012, 43, 341–351. [Google Scholar] [CrossRef] [Green Version]
- De Goncalves, R.C.R.; Pombeiro-Sponchiado, S.R. Antioxidant activity of the melanin pigment extracted from Aspergillus nidulans. Biol. Pharm. Bull. 2005, 28, 1129–1131. [Google Scholar] [CrossRef] [Green Version]
- Tu, Y.G.; Sun, Y.Z.; Tian, Y.G.; Xie, M.Y.; Chen, J. Physicochemical characterisation and antioxidant activity of melanin from the muscles of Taihe Black-bone silky fowl (Gallus gallus domesticus Brisson). Food Chem. 2009, 114, 1345–1350. [Google Scholar] [CrossRef]
- El-Naggar, N.E.A.; El-Ewasy, S.M. Bioproduction, characterization, anticancer and antioxidant activities of extracellular melanin pigment produced by newly isolated microbial cell factories Streptomyces glaucescens NEAE-H. Sci. Rep. 2017, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Geng, J.; Tang, W.; Wan, X.; Zhou, Q.; Wang, X.J.; Shen, P.; Lei, T.C.; Chen, X.D. Photoprotection of bacterial-derived melanin against ultraviolet A-induced cell death and its potential application as an active sunscreen. J. Eur. Acad. Dermatol. Venereol. 2008, 22, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Seelam, S.D.; Agsar, D.; Halmuthur, M.S.K.; Reddy Shetty, P.; Vemireddy, S.; Reddy, K.M.; Umesh, M.K.; Rajitha, C.H. Characterization and photoprotective potentiality of lime dwelling Pseudomonas mediated melanin as sunscreen agent against UV-B radiations. J. Photochem. Photobiol. B Biol. 2021, 216, 112126. [Google Scholar] [CrossRef]
- El-Obeid, A.; Al-Harbi, S.; Al-Jomah, N.; Hassib, A. Herbal melanin modulates tumor necrosis factor alpha (TNF-α), interleukin 6 (IL-6) and vascular endothelial growth factor (VEGF) production. Phytomedicine 2006, 13, 324–333. [Google Scholar] [CrossRef] [PubMed]
- El-Obeid, A.; Hassib, A.; Pontén, F.; Westermark, B. Effect of herbal melanin on IL-8: A possible role of Toll-like receptor 4 (TLR4). Biochem. Biophys. Res. Commun. 2006, 344, 1200–1206. [Google Scholar] [CrossRef]
- Al-Obeed, O.; El-Obeid, A.S.; Matou-Nasri, S.; Vaali-Mohammed, M.A.; Alhaidan, Y.; Elwatidy, M.; Al Dosary, H.; Alehaideb, Z.; Alkhayal, K.; Haseeb, A.; et al. Herbal melanin inhibits colorectal cancer cell proliferation by altering redox balance, inducing apoptosis, and modulating MAPK signaling. Cancer Cell Int. 2020, 20, 126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Obeid, A.; Alajmi, H.; Harbi, M.; Yahya, W.B.; Al-Eidi, H.; Alaujan, M.; Haseeb, A.; Trivilegio, T.; Alhallaj, A.; Alghamdi, S.; et al. Distinct anti-proliferative effects of herbal melanin on human acute monocytic leukemia thp-1 cells and embryonic kidney hek293 cells. BMC Complement. Med. Ther. 2020, 20, 154. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Wang, C.; Zhang, X.; Hu, Q.; Zhang, Y.; Liu, Q.; Wen, D.; Milligan, J.; Bellotti, A.; Huang, L.; et al. A melanin-mediated cancer immunotherapy patch. Sci. Immunol. 2017, 2, eaan5692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carpentier, A.F.; Geinguenaud, F.; Tran, T.; Sejalon, F.; Martin, A.; Motte, L.; Tartour, E.; Banissi, C. Synthetic melanin bound to subunit vaccine antigens significantly enhances CD8+ T-cell responses. PLoS ONE. 2017, 12, e0181403. [Google Scholar] [CrossRef] [Green Version]
- Jiang, S.; Liu, X.M.; Dai, X.; Zhou, Q.; Lei, T.C.; Beermann, F.; Wakamatsu, K.; Xu, S.Z. Regulation of DHICA-mediated antioxidation by dopachrome tautomerase: Implication for skin photoprotection against UVA radiation. Free Radic. Biol. Med. 2010, 48, 1144–1151. [Google Scholar] [CrossRef]
- Yao, Z.Y.; Qi, J.H. Comparison of antioxidant activities of melanin fractions from chestnut shell. Molecules 2016, 21, 487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Micillo, R.; Panzella, L.; Koike, K.; Monfrecola, G.; Napolitano, A.; D’Ischia, M. “Fifty shades” of black and red or how carboxyl groups fine tune eumelanin and pheomelanin properties. Int. J. Mol. Sci. 2016, 17, 746. [Google Scholar] [CrossRef]
- Kumar, C.G.; Mongolla, P.; Pombala, S.; Kamle, A.; Joseph, J. Physicochemical characterization and antioxidant activity of melanin from a novel strain of Aspergillus bridgeri ICTF-201. Lett. Appl. Microbiol. 2011, 53, 350–358. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV radiation and the skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hart, P.H.; Norval, M. Ultraviolet radiation-induced immunosuppression and its relevance for skin carcinogenesis. Photochem. Photobiol. Sci. 2018, 17, 1872–1884. [Google Scholar] [CrossRef] [PubMed]
- Panich, U.; Sittithumcharee, G.; Rathviboon, N.; Jirawatnotai, S. Ultraviolet radiation-induced skin aging: The role of DNA damage and oxidative stress in epidermal stem cell damage mediated skin aging. Stem Cells Int. 2016, 2016, 7370642. [Google Scholar] [CrossRef] [Green Version]
- Mohania, D.; Chandel, S.; Kumar, P.; Verma, V.; Digvijay, K.; Tripathi, D.; Choudhury, K.; Mitten, S.K.; Shah, D. Ultraviolet radiations: Skin defense-damage mechanism. Adv. Exp. Med. Biol. 2017, 996, 71–87. [Google Scholar] [CrossRef]
- Sample, A.; He, Y.Y. Mechanisms and prevention of UV-induced melanoma. Photodermatol. Photoimmunol. Photomed. 2018, 34, 13–24. [Google Scholar] [CrossRef]
- Kobayashi, N.; Nakagawa, A.; Muramatsu, T.; Yamashina, Y.; Shirai, T.; Hashimoto, M.W.; Ishigaki, Y.; Ohnishi, T.; Mori, T. Supranuclear melanin caps reduce ultraviolet induced DNA photoproducts in human epidermis. J. Invest. Dermatol. 1998, 110, 806–810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solano, F. Photoprotection versus photodamage: Updating an old but still unsolved controversy about melanin. Polym. Int. 2016, 65, 1276–1287. [Google Scholar] [CrossRef]
- Brenner, M.; Hearing, V.J. The protective role of melanin against UV damage in human skin. Photochem. Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, J.J.; Kim, J.Y.; Son, S.H.; Jung, W.J.; Kim, D.H.; Seo, J.W.; Kim, G.H. Fungal melanin as a biocompatible broad-spectrum sunscreen with high antioxidant activity. RSC Adv. 2021, 11, 19682–19689. [Google Scholar] [CrossRef]
- Corani, A.; Huijser, A.; Gustavsson, T.; Markovitsi, D.; Malmqvist, P.Å.; Pezzella, A.; D’Ischia, M.; Sundström, V. Superior photoprotective motifs and mechanisms in eumelanins uncovered. J. Am. Chem. Soc. 2014, 136, 11626–11635. [Google Scholar] [CrossRef] [PubMed]
- Kulka, M. Mechanisms and Treatment of Photoaging and Photodamage. In Using Old Solutions to New Problems—Natural Drug Discovery in the 21st Century; Kulka, M., Ed.; IntechOpen: London, UK, 2013. [Google Scholar] [CrossRef] [Green Version]
- Fu, C.; Chen, J.; Lu, J.; Yi, L.; Tong, X.; Kang, L.; Pei, S.; Ouyang, Y.; Jiang, L.; Ding, Y.; et al. Roles of inflammation factors in melanogenesis (Review). Mol. Med. Rep. 2020, 21, 1421–1430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lawrence, E.; Al Alboud, K.M. Postinflammatory Hyperpigmentation; StatPearls: Treasure Island, FL, USA, 9 October 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559150/ (accessed on 5 January 2022).
- Belemets, N.; Kobyliak, N.; Falalyeyeva, T.; Kuryk, O.; Sulaieva, O.; Vovk, T.; Beregova, T.; Ostapchenko, L. Polyphenol compounds melanin prevented hepatic inflammation in rats with experimental obesity. Nat. Prod. Commun. 2018, 13, 1485–1488. [Google Scholar] [CrossRef] [Green Version]
- Belemets, N.; Kobyliak, N.; Virchenko, O.; Falalyeyeva, T.; Olena, T.; Bodnar, P.; Savchuk, O.; Galenova, T.; Caprnda, M.; Rodrigo, L.; et al. Effects of polyphenol compounds melanin on NAFLD/NASH prevention. Biomed. Pharmacother. 2017, 88, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Kurian, N.K.; Nair, H.P.; Bhat, S.G. Evaluation of anti-inflammatory property of melanin from marine bacillus spp. BTCZ31. Asian J. Pharm. Clin. Res. 2015, 8, 251–255. [Google Scholar]
- Leupold, D.; Pfeifer, L.; Hofmann, M.; Forschner, A.; Wessler, G.; Haenssle, H. From melanocytes to melanoma cells: Characterization of the malignant transformation by four distinctly different melanin fluorescence spectra (review). Int. J. Mol. Sci. 2021, 22, 5265. [Google Scholar] [CrossRef]
- Slominski, R.M.; Zmijewski, M.A.; Slominski, A.T. The role of melanin pigment in melanoma. Exp. Dermatol. 2015, 24, 258–259. [Google Scholar] [CrossRef] [Green Version]
- Burns, D.; George, J.; Aucoin, D.; Bower, J.; Burrell, S.; Gilbert, R.; Bower, N. The Pathogenesis and Clinical Management of Cutaneous Melanoma: An Evidence-Based Review. J. Med. Imaging Radiat. Sci. 2019, 50, 460–469.e1. [Google Scholar] [CrossRef] [Green Version]
- Sarna, M.; Krzykawska-Serda, M.; Jakubowska, M.; Zadlo, A.; Urbanska, K. Melanin presence inhibits melanoma cell spread in mice in a unique mechanical fashion. Sci. Rep. 2019, 9, 9280. [Google Scholar] [CrossRef]
- Sarna, M.; Zadlo, A.; Czuba-Pelech, B.; Urbanska, K. Nanomechanical phenotype of melanoma cells depends solely on the amount of endogenous pigment in the cells. Int. J. Mol. Sci. 2018, 19, 607. [Google Scholar] [CrossRef] [Green Version]
- Shi, F.; Li, J.; Ye, Z.; Yang, L.; Chen, T.; Chen, X.; Ye, M. Antitumor effects of melanin from: Lachnum YM226 and its derivative in H22 tumor-bearing mice. MedChemComm 2018, 9, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.T.; Tan, Y.J.; Oon, C.E. Molecular targeted therapy: Treating cancer with specificity. Eur. J. Pharmacol. 2018, 834, 188–196. [Google Scholar] [CrossRef] [PubMed]
- AL-Busairi, W.; Khajah, M. The Principles behind Targeted Therapy for Cancer Treatment. In Tumor Progression and Metastasis; Lasfar, A., Cohen-Solal, K., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Rouanet, J.; Quintana, M.; Auzeloux, P.; Cachin, F.; Degoul, F. Benzamide derivative radiotracers targeting melanin for melanoma imaging and therapy: Preclinical/clinical development and combination with other treatments. Pharmacol. Ther. 2021, 224. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, M.; Mishellany, F.; Papon, J.; Cayre, A.; Penault-Llorca, F.; Madelmont, J.C.; Miot-Noirault, E.; Chezal, J.M.; Moins, N. Anti-melanoma efficacy of internal radionuclide therapy in relation to melanin target distribution. Pigment Cell Melanoma Res. 2010, 23, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dadachova, E.; Casadevall, A. Melanin as a potential target for radionuclide therapy of metastatic melanoma. Future Oncol. 2005, 1, 541–549. [Google Scholar] [CrossRef]
- Link, E.M. Targeting melanoma with 211At/131I-methylene blue: Preclinical and clinical experience. Hybridoma 1999, 18, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Degoul, F.; Borel, M.; Jacquemot, N.; Besse, S.; Communal, Y.; Mishellany, F.; Papon, J.; Penault-Llorca, F.; Donnarieix, D.; Doly, M.; et al. In vivo efficacy of melanoma internal radionuclide therapy with a 131I-labelled melanin-targeting heteroarylcarboxamide molecule. Int. J. Cancer 2013, 133, 1042–1053. [Google Scholar] [CrossRef] [PubMed]
- Viallard, C.; Perrot, Y.; Boudhraa, Z.; Jouberton, E.; Miot-Noirault, E.; Bonnet, M.; Besse, S.; Mishellany, F.; Cayre, A.; Maigne, L.; et al. [123 I]ICF01012 melanoma imaging and [131 I]ICF01012 dosimetry allow adapted internal targeted radiotherapy in preclinical melanoma models. Eur. J. Dermatol. 2015, 25, 29–35. [Google Scholar] [CrossRef]
- Safari Yazd, H.; Yang, Y.; Li, L.; Yang, L.; Li, X.; Pan, X.; Chen, Z.; Jiang, J.; Cui, C.; Tan, W. Precise Deposition of Polydopamine on Cancer Cell Membrane as Artificial Receptor for Targeted Drug Delivery. iScience 2020, 23, 101750. [Google Scholar] [CrossRef]
- Le Na, N.T.; Loc, S.D.; Tri, N.L.M.; Loan, N.T.B.; Son, H.A.; Toan, N.L.; Thu, H.P.; Nhung, H.T.M.; Thanh, N.L.; Van Anh, N.T.; et al. Nanomelanin potentially protects the spleen from radiotherapy-associated damage and enhances immunoactivity in tumor-bearing mice. Materials (Basel) 2019, 12, 1725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yue, Y.; Zhao, X. Melanin-like nanomedicine in photothermal therapy applications. Int. J. Mol. Sci. 2021, 22, 399. [Google Scholar] [CrossRef]
- Lemaster, J.E.; Jeevarathinam, A.S.; Kumar, A.; Chandrasekar, B.; Chen, F.; Jokerst, J.V. Synthesis of Ultrasmall Synthetic Melanin Nanoparticles by UV Irradiation in Acidic and Neutral Conditions. ACS Appl. Bio Mater. 2019, 2, 4667–4674. [Google Scholar] [CrossRef]
- Yan, J.; Yang, L.; Lin, M.F.; Ma, J.; Lu, X.; Lee, P.S. Polydopamine spheres as active templates for convenient synthesis of various nanostructures. Small 2013, 9, 596–603. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Zarei, M.; Hashemi, S.A.R. Polydopamine for Biomedical Application and Drug Delivery System. Med. Chem. (Los. Angeles) 2018, 08, 218–229. [Google Scholar] [CrossRef]
- Mavridi-Printezi, A.; Guernelli, M.; Menichetti, A.; Montalti, M. Bio-Applications of Multifunctional Melanin Nanoparticles: From Nanomedicine to Nanocosmetics. Nanomaterials 2020, 10, 2276. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Wang, S.; Chen, K.; Zhao, Y.; Ma, X.; Wang, L. Doxorubicin-Loaded Melanin Particles for Enhanced Chemotherapy in Drug-Resistant Anaplastic Thyroid Cancer Cells. J. Nanomater. 2018, 2018, 2603712. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Bi, D.; Qi, X.; Guo, Y.; Yue, F.; Wang, X.; Han, M. Polydopamine-based surface modification of paclitaxel nanoparticles for osteosarcoma targeted therapy. Nanotechnology 2019, 30, 255101. [Google Scholar] [CrossRef]
- Nieto, C.; Vega, M.A.; Del Valle, E.M. Nature-inspired nanoparticles as paclitaxel targeted carrier for the treatment of her2-positive breast cancer. Cancers (Basel) 2021, 13, 2526. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Zhong, C.; Zhang, Q.; Wang, L.; Wang, L.; Liu, Y.; Zhang, X.; Zhao, X. pH-responsive delivery vehicle based on RGD-modified polydopamine-paclitaxel-loaded poly (3-hydroxybutyrate-co-3-hydroxyvalerate) nanoparticles for targeted therapy in hepatocellular carcinoma. J. Nanobiotechnology 2021, 19, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Ozlu, B.; Kabay, G.; Bocek, I.; Yilmaz, M.; Piskin, A.K.; Shim, B.S.; Mutlu, M. Controlled release of doxorubicin from polyethylene glycol functionalized melanin nanoparticles for breast cancer therapy: Part I. Production and drug release performance of the melanin nanoparticles. Int. J. Pharm. 2019, 570, 118613. [Google Scholar] [CrossRef]
- Song, Y.; Cai, L.; Tian, Z.; Wu, Y.; Chen, J. Phytochemical Curcumin-Coformulated, Silver-Decorated Melanin-like Polydopamine/Mesoporous Silica Composites with Improved Antibacterial and Chemotherapeutic Effects against Drug-Resistant Cancer Cells. ACS Omega 2020, 5, 15083–15094. [Google Scholar] [CrossRef]
- Wang, B.; Yuan, T.; Zha, L.; Liu, Y.; Chen, W.; Zhang, C.; Bao, Y.; Dong, Q. Oral Delivery of Gambogenic Acid by Functional Polydopamine Nanoparticles for Targeted Tumor Therapy. Mol. Pharm. 2021, 18, 1470–1479. [Google Scholar] [CrossRef]
- Li, X.J.; Li, W.T.; Li, Z.H.R.; Zhang, L.P.; Gai, C.C.; Zhang, W.F.; Ding, D.J. Iron-chelated polydopamine decorated doxorubicin-loaded nanodevices for reactive oxygen species enhanced cancer combination therapy. Front. Pharmacol. 2019, 10, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Li, W.Q.; Wang, Z.; Hao, S.; He, H.; Wan, Y.; Zhu, C.; Sun, L.P.; Cheng, G.; Zheng, S.Y. Mitochondria-Targeting Polydopamine Nanoparticles to Deliver Doxorubicin for Overcoming Drug Resistance. ACS Appl. Mater. Interfaces 2017, 9, 16793–16802. [Google Scholar] [CrossRef]
- Schweitzer, A.D.; Revskaya, E.; Chu, P.; Pazo, V.; Friedman, M.; Nosanchuk, J.D.; Cahill, S.; Frases, S.; Casadevall, A.; Dadachova, E. Melanin-covered nanoparticles for protection of bone marrow during radiation therapy of cancer. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 1494–1502. [Google Scholar] [CrossRef] [Green Version]
- Rageh, M.M.; El-Gebaly, R.H. Melanin nanoparticles: Antioxidant activities and effects on γ-ray-induced DNA damage in the mouse. Mutat Res Genet Toxicol Environ Mutagen 2018, 828, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, B.; Zhang, Z.; Wang, B.; Xu, Q.; Mao, W.; Tian, J.; Yang, K.; Wang, F. Radionuclide Imaging-Guided Chemo-Radioisotope Synergistic Therapy Using a 131I-Labeled Polydopamine Multifunctional Nanocarrier. Mol. Ther. 2018, 26, 1385–1393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, X.; Yang, K.; Dong, Z.; Yi, X.; Wang, Y.; Ge, C.; Zhao, Y.; Liu, Z. Polydopamine as a Biocompatible Multifunctional Nanocarrier for Combined Radioisotope Therapy and Chemotherapy of Cancer. Adv. Funct. Mater. 2015, 25, 7327–7336. [Google Scholar] [CrossRef]
- Yang, P.; Zhang, S.; Zhang, N.; Wang, Y.; Zhong, J.; Sun, X.; Qi, Y.; Chen, X.; Li, Z.; Li, Y. Tailoring Synthetic Melanin Nanoparticles for Enhanced Photothermal Therapy. ACS Appl. Mater. Interfaces 2019, 11, 42671–42679. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Yan, Y.; Wang, L.; Zhang, Q.; Cheng, Y. Melanin-like nanoparticles decorated with an autophagy-inducing peptide for efficient targeted photothermal therapy. Biomaterials 2019, 203, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, X.; Guo, H. Synergic highly effective photothermal-chemotherapy with platinum prodrug linked melanin-like nanoparticles. Artif. Cells Nanomed. Biotechnol. 2018, 46, 356–363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Chai, J.; Liu, L.; Cui, Z.; Duan, D.; Shi, R.; Zhang, Y. Dual-functional melanin-based nanoliposomes for combined chemotherapy and photothermal therapy of pancreatic cancer. RSC Adv. 2019, 9, 3012–3019. [Google Scholar] [CrossRef] [Green Version]
- Kong, N.; Deng, M.; Sun, X.N.; Chen, Y.D.; Sui, X.B. Polydopamine-functionalized CA-(PCL-ran-PLA) nanoparticles for target delivery of docetaxel and chemo-photothermal therapy of breast cancer. Front. Pharmacol. 2018, 9, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hauser, D.; Estermann, M.; Milosevic, A.; Steinmetz, L.; Vanhecke, D.; Septiadi, D.; Drasler, B.; Petri-Fink, A.; Ball, V.; Rothen-Rutishauser, B. Polydopamine/transferrin hybrid nanoparticles for targeted cell-killing. Nanomaterials 2018, 8, 1065. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Ouyang, X.; Chen, J.; Hu, Y.; Sun, X.; Yu, Z. Nanoparticulate photosensitizer decorated with hyaluronic acid for photodynamic/photothermal cancer targeting therapy. Nanomedicine 2019, 14, 151–167. [Google Scholar] [CrossRef]
- Li, X.; Zou, Q.; Zhang, J.; Zhang, P.; Zhou, X.; Yalamarty, S.S.K.; Liang, X.; Liu, Y.; Zheng, Q.; Gao, J. Self-assembled dual-targeted epirubicin-hybrid polydopamine nanoparticles for combined chemo-photothermal therapy of triple-negative breast cancer. Int. J. Nanomed. 2020, 15, 6791–6811. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Huang, Q.; Chen, J.; Song, X.; Chen, Z.; Huang, M.; Xu, P.; Zhang, J. Tumor-targeting photodynamic therapy based on folate-modified polydopamine nanoparticles. Int. J. Nanomed. 2019, 14, 6799–6812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, D.; Wu, M.; Zeng, Y.; Wu, L.; Wang, Q.; Han, X.; Liu, X.; Liu, J. Chlorin e6 conjugated poly(dopamine) nanospheres as PDT/PTT dual-modal therapeutic agents for enhanced cancer therapy. ACS Appl. Mater. Interfaces 2015, 7, 8176–8187. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, N.; Yang, Y.; Wang, X.; Liang, J.; Tian, X.; Zhang, H.; Leng, X. Polydopamine nanoparticles carrying tumor cell lysate as a potential vaccine for colorectal cancer immunotherapy. Biomater. Sci. 2019, 7, 3062–3075. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Zhang, L.; Guo, Q.; Zuo, Y.; Wang, N.; Wang, H.; Kong, D.; Zhu, D.; Zhang, L. Robust Nanovaccine Based on Polydopamine-Coated Mesoporous Silica Nanoparticles for Effective Photothermal-Immunotherapy Against Melanoma. Adv. Funct. Mater. 2021, 31, 2010637. [Google Scholar] [CrossRef]
- Wang, N.; Yang, Y.; Wang, X.; Tian, X.; Qin, W.; Wang, X.; Liang, J.; Zhang, H.; Leng, X. Polydopamine as the Antigen Delivery Nanocarrier for Enhanced Immune Response in Tumor Immunotherapy. ACS Biomater. Sci. Eng. 2019, 5, 2330–2442. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, X.; Pan, W.; Li, N.; Tang, B. Photothermal therapy-induced immunogenic cell death based on natural melanin nanoparticles against breast cancer. Chem. Commun. 2020, 56, 1389–1392. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Xu, Q.; Li, X.; Wang, Y. pH-responsive polydopamine nanoparticles for photothermally promoted gene delivery. Mater. Sci. Eng. C 2020, 108, 110396. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, Z.; Zuo, D.; Li, L.; Li, F.; Yang, D. A Synergistic DNA-polydopamine-MnO2Nanocomplex for Near-Infrared-Light-Powered DNAzyme-Mediated Gene Therapy. Nano Lett. 2021, 21, 5377–5385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Xu, Q.; Du, J.; Wang, Y. Polydopamine-based nanoparticles with excellent biocompatibility for photothermally enhanced gene delivery. RSC Adv. 2018, 8, 34596–34602. [Google Scholar] [CrossRef] [Green Version]
- Guan, Q.; Guo, R.; Huang, S.; Zhang, F.; Liu, J.; Wang, Z.; Yang, X.; Shuai, X.; Cao, Z. Mesoporous polydopamine carrying sorafenib and SPIO nanoparticles for MRI-guided ferroptosis cancer therapy. J. Control Release 2020, 320, 392–403. [Google Scholar] [CrossRef]
- Ha, S.W.; Cho, H.S.; Yoon, Y.I.; Jang, M.S.; Hong, K.S.; Hui, E.; Lee, J.H.; Yoon, T.J. Ions doped melanin nanoparticle as a multiple imaging agent. J. Nanobiotechnology 2017, 15, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, W.; Hu, C.; Zheng, H.; Xie, J.; Shi, X.; Du, Y.; Wang, F. A Functionalized Polydopamine Theranostic Nanoprobe for Efficient Imaging of miRNA-21 and In Vivo Synergetic Cancer Therapy. Mol. Ther—Nucleic Acids 2020, 22, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, L.; Lin, M.; Wang, D.; Song, Z.; Li, S.; Ge, R.; Zhang, X.; Liu, Y.; Li, Z.; et al. Cu(II)-Doped Polydopamine-Coated Gold Nanorods for Tumor Theranostics. ACS Appl. Mater. Interfaces 2017, 9, 44293–44306. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Da, L.; Yang, C.; Chen, R.; Gao, L.; Fan, L.; Han, J. Mn2+-coordinated PDA@DOX/PLGA nanoparticles as a smart theranostic agent for synergistic chemo-photothermal tumor therapy. Int. J. Nanomed. 2017, 12, 3331–3345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Falzone, L.; Salomone, S.; Libra, M. Evolution of cancer pharmacological treatments at the turn of the third millennium. Front. Pharmacol. 2018, 9, 1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vega-Vásquez, P.; Mosier, N.S.; Irudayaraj, J. Nanoscale Drug Delivery Systems: From Medicine to Agriculture. Front. Bioeng. Biotechnol. 2020, 8, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Fan, Q.; Yang, M.; Cheng, K.; Lu, X.; Zhang, L.; Huang, W.; Cheng, Z. Engineering Melanin Nanoparticles as an Efficient Drug-Delivery System for Imaging-Guided Chemotherapy. Adv. Mater. 2015, 27, 5063–5069. [Google Scholar] [CrossRef] [PubMed]
- Cuzzubbo, S.; Carpentier, A.F. Applications of melanin and melanin-like nanoparticles in cancer therapy: A review of recent advances. Cancers (Basel) 2021, 13, 1463. [Google Scholar] [CrossRef]
- Nieto, C.; Vega, M.A.; Marcelo, G.; Martín Del Valle, E.M. Polydopamine nanoparticles kill cancer cells. RSC Adv. 2018, 8, 36201–36208. [Google Scholar] [CrossRef] [Green Version]
- Perring, J.; Crawshay-Williams, F.; Huang, C.; Townley, H.E. Bio-inspired melanin nanoparticles induce cancer cell death by iron adsorption. J. Mater. Sci. Mater. Med. 2018, 29, 181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gabriele, V.R.; Mazhabi, R.M.; Alexander, N.; Mukherjee, P.; Seyfried, T.N.; Nwaji, N.; Akinoglu, E.M.; Mackiewicz, A.; Zhou, G.; Giersig, M.; et al. Light-and melanin nanoparticle-induced cytotoxicity in metastatic cancer cells. Pharmaceutics 2021, 13, 965. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.G.; Surendran, S.P.; Jeong, Y.Y. Tumor Microenvironment-Stimuli Responsive Nanoparticles for Anticancer Therapy. Front. Mol. Biosci. 2020, 7, 610533. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Chen, L.; Chen, X.; Xiao, B. Current strategies to enhance the targeting of polydopamine-based platforms for cancer therapeutics. J. Drug Target. 2020, 28, 142–153. [Google Scholar] [CrossRef]
- Xie, J.; Wang, C.; Zhao, F.; Gu, Z.; Zhao, Y. Application of Multifunctional Nanomaterials in Radioprotection of Healthy Tissues. Adv. Healthc. Mater. 2018, 7, 1800421. [Google Scholar] [CrossRef]
- Baker, C.H. Radiation Protection and Nanoparticles. JSM Nanotechnol Nanomed 2014, 2, 1019. [Google Scholar]
- Huang, Y.; Li, Y.; Hu, Z.; Yue, X.; Proetto, M.T.; Jones, Y.; Gianneschi, N.C. Mimicking Melanosomes: Polydopamine Nanoparticles as Artificial Microparasols. ACS Cent. Sci. 2017, 3, 564–569. [Google Scholar] [CrossRef] [Green Version]
- Sgouros, G.; Bodei, L.; McDevitt, M.R.; Nedrow, J.R. Radiopharmaceutical therapy in cancer: Clinical advances and challenges. Nat. Rev. Drug Discov. 2020, 19, 589–608. [Google Scholar] [CrossRef]
- Altıparmak Güleç, B.; Yurt, F. Treatment with Radiopharmaceuticals and Radionuclides in Breast Cancer: Current Options. Eur. J. Breast Health 2021, 17, 214–219. [Google Scholar] [CrossRef]
- James, S.S.; Bednarz, B.; Benedict, S.; Buchsbaum, J.C.; Dewaraja, Y.; Frey, E.; Hobbs, R.; Grudzinski, J.; Roncali, E.; Sgouros, G.; et al. Current Status of Radiopharmaceutical Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 891–901. [Google Scholar] [CrossRef]
- Zmerli, I.; Ibrahim, N.; Cressey, P.; Denis, S.; Makky, A. Design and Synthesis of New PEGylated Polydopamine-Based Nanoconstructs Bearing ROS-Responsive Linkers and a Photosensitizer for Bimodal Photothermal and Photodynamic Therapies against Cancer. Mol. Pharm. 2021, 18, 3623–3637. [Google Scholar] [CrossRef]
- Lee, Y.E.; Kopelman, R. Polymeric Nanoparticles for Photodynamic Therapy. Methods Mol. Biol. 2011, 726, 151–178. [Google Scholar] [CrossRef] [PubMed]
- Vines, J.B.; Lim, D.J.; Park, H. Contemporary polymer-based nanoparticle systems for photothermal therapy. Polymers (Basel) 2018, 10, 1357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dias, L.D.; Buzzá, H.H.; Stringasci, M.D.; Bagnato, V.S. Recent Advances in Combined Photothermal and Photodynamic Therapies against Cancer Using Carbon Nanomaterial Platforms for In Vivo Studies. Photochem 2021, 1, 26. [Google Scholar] [CrossRef]
- Jiang, Q.; Luo, Z.; Men, Y.; Yang, P.; Peng, H.; Guo, R.; Tian, Y.; Pang, Z.; Yang, W. Red blood cell membrane-camouflaged melanin nanoparticles for enhanced photothermal therapy. Biomaterials 2017, 143, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Colombo, L.L.; Vanzulli, S.I.; Blázquez-Castro, A.; Terrero, C.S.; Stockert, J.C. Photothermal effect by 808-nm laser irradiation of melanin: A proof-of-concept study of photothermal therapy using B16-F10 melanotic melanoma growing in BALB/c mice. Biomed. Opt. Express 2019, 10, 2932. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ai, K.; Liu, J.; Deng, M.; He, Y.; Lu, L. Dopamine-melanin colloidal nanospheres: An efficient near-infrared photothermal therapeutic agent for in vivo cancer therapy. Adv. Mater. 2013, 25, 1353–1359. [Google Scholar] [CrossRef]
- Poinard, B.; Neo, S.Z.Y.; Yeo, E.L.L.; Heng, H.P.S.; Neoh, K.G.; Kah, J.C.Y. Polydopamine Nanoparticles Enhance Drug Release for Combined Photodynamic and Photothermal Therapy. ACS Appl. Mater. Interfaces 2018, 10, 21125–21136. [Google Scholar] [CrossRef] [PubMed]
- Thakur, N.; Thakur, S.; Chatterjee, S.; Das, J.; Sil, P.C. Nanoparticles as Smart Carriers for Enhanced Cancer Immunotherapy. Front. Chem. 2020, 8, 597806. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, M.S. Improving cancer immunotherapy through nanotechnology. Nat. Rev. Cancer 2019, 19, 587–602. [Google Scholar] [CrossRef] [PubMed]
- Velpurisiva, P.; Gad, A.; Piel, B.; Jadia, R.; Rai, P. Nanoparticle Design Strategies for Effective Cancer Immunotherapy. J. Biomed. (Syd) 2017, 2, 64–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, J.; Safdari, H.A.; Hoque, M. Nanoparticle mediated cancer immunotherapy. Semin. Cancer Biol. 2021, 69, 307–324. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Guo, Z.; Tian, H.; Chen, X. Production and clinical development of nanoparticles for gene delivery. Mol. Ther.—Methods Clin. Dev. 2016, 3, 16023. [Google Scholar] [CrossRef] [PubMed]
- Roacho-Perez, J.A.; Gallardo-Blanco, H.L.; Sanchez-Dominguez, M.; Garcia-Casillas, P.; Chapa-Gonzalez, C.; Sanchez-Dominguez, C.N. Nanoparticles for death-induced gene therapy in cancer (Review). Mol. Med. Rep. 2018, 17, 1413–1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bulcha, J.T.; Wang, Y.; Ma, H.; Tai, P.W.L.; Gao, G. Viral vector platforms within the gene therapy landscape. Signal Transduct. Target. Ther. 2021, 6, 53. [Google Scholar] [CrossRef]
- Bolat, G.; Vural, O.A.; Yaman, Y.T.; Abaci, S. Polydopamine nanoparticles-assisted impedimetric sensor towards label-free lung cancer cell detection. Mater. Sci. Eng. C 2021, 119, 111549. [Google Scholar] [CrossRef]
- Chapman, S.; Dobrovolskaia, M.; Farahani, K.; Goodwin, A.; Joshi, A.; Lee, H.; Meade, T.; Pomper, M.; Ptak, K.; Rao, J.; et al. Nanoparticles for cancer imaging: The good, the bad, and the promise. Nano Today 2013, 8, 454–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Li, M.; Gao, X.; Chen, Y.; Liu, T. Nanotechnology in cancer diagnosis: Progress, challenges and opportunities. J. Hematol. Oncol. 2019, 12, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ji, L.; Yan, T.; Li, Y.; Gao, J.; Wang, Q.; Hu, L.; Wu, D.; Wei, Q.; Du, B. Preparation of Au-polydopamine functionalized carbon encapsulated Fe3O4 magnetic nanocomposites and their application for ultrasensitive detection of carcino-embryonic antigen. Sci. Rep. 2016, 6, 21017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, W.K.; Wong, S.H.D.; Bian, L. Long-Term Detection of Oncogenic MicroRNA in Living Human Cancer Cells by Gold@ Polydopamine-Shell Nanoprobe. ACS Biomater. Sci. Eng. 2020, 6, 3778–3783. [Google Scholar] [CrossRef]
- Jin, C.; Wang, K.; Oppong-Gyebi, A.; Hu, J. Application of nanotechnology in cancer diagnosis and therapy—A mini-review. Int. J. Med. Sci. 2020, 17, 2964–2973. [Google Scholar] [CrossRef] [PubMed]
- Toy, R.; Bauer, L.; Hoimes, C.; Ghaghada, K.B.; Karathanasis, E. Targeted nanotechnology for cancer imaging. Adv. Drug Deliv. Rev. 2014, 76, 79–97. [Google Scholar] [CrossRef] [Green Version]
- Smith, L.; Kuncic, Z.; Ostrikov, K.; Kumar, S. Nanoparticles in cancer imaging and therapy. J. Nanomater. 2012, 2012. [Google Scholar] [CrossRef] [Green Version]
- Dong, Z.; Gong, H.; Gao, M.; Zhu, W.; Sun, X.; Feng, L.; Fu, T.; Li, Y.; Liu, Z. Polydopamine nanoparticles as a versatile molecular loading platform to enable imaging-guided cancer combination therapy. Theranostics 2016, 6, 1031–1042. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.; Jain, S.; Kumar, D.; Soni, S.L.; Sharma, M. A Review on—Theranostics: An Approach to Targeted Diagnosis and Therapy. Asian J. Pharm. Res. Dev. 2019, 7, 63–69. [Google Scholar] [CrossRef]
- Wong, X.Y.; Sena-Torralba, A.; Álvarez-Diduk, R.; Muthoosamy, K.; Merkoçi, A. Nanomaterials for Nanotheranostics: Tuning Their Properties According to Disease Needs. ACS Nano 2020, 14, 2585–2627. [Google Scholar] [CrossRef] [PubMed]
- Indoria, S.; Singh, V.; Hsieh, M.F. Recent advances in theranostic polymeric nanoparticles for cancer treatment: A review. Int. J. Pharm. 2020, 582, 119314. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Zhang, J.; Li, J.; Fan, Y.; Wang, J.; Sun, W.; Yang, H.; Peng, C.; Shen, M.; Shi, X. Polydopamine-coated magnetic mesoporous silica nanoparticles for multimodal cancer theranostics. J. Mater. Chem. B 2019, 7, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects Jayanta. J. Nanobiotechnology 2018, 16, 1–33. [Google Scholar] [CrossRef] [Green Version]
- Chu, M.; Hai, W.; Zhang, Z.; Wo, F.; Wu, Q.; Zhang, Z.; Shao, Y.; Zhang, D.; Jin, L.; Shi, D. Melanin nanoparticles derived from a homology of medicine and food for sentinel lymph node mapping and photothermal in vivo cancer therapy. Biomaterials 2016, 91, 182–199. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Kim, K.Y.; Wook, H.J.; Park, S.Y.; Lee, K.D.; Lee, D.Y.; Lee, H. Attenuation of the in vivo toxicity of biomaterials by polydopamine surface modification. Nanomedicine 2011, 6, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, Y.; Higashisaka, K.; Tsutsumi, Y. Biocompatibility of Nanomaterials. In Nanomaterials in Pharmacology; Lu, Z.-R., Sakuma, S., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany; Humana Press: Totowa, NJ, USA, 2016. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Fang, H.; Gai, Y.; Lan, X. pH-Triggered Assembly of Natural Melanin Nanoparticles for Enhanced PET Imaging. Front. Chem. 2020, 8, 1–9. [Google Scholar] [CrossRef]
- Ruiz, J.J.; Marro, M.; Galván, I.; Bernabeu-Wittel, J.; Conejo-Mir, J.; Zulueta-Dorado, T.; Guisado-Gil, A.B.; Loza-Álvarez, P. Novel Non-Invasive Quantification and Imaging of Eumelanin and DHICA Subunit in Skin Lesions by Raman Spectroscopy and MCR Algorithm: Improving Dysplastic Nevi Diagnosis. Cancers 2022, 14, 1056. [Google Scholar] [CrossRef] [PubMed]

| Source | Effect | Observations | Tested Concentrations | Reference |
|---|---|---|---|---|
| Fungal melanin from Aspergillus nidulans | Antioxidant | inhibition of 5-thio-2-nitrobenzoic acid (TNB) oxidation; scavenging activity on the tested oxidants (i.e., H2O2 and HOCl) | 25, 50, and 100 µg/mL (in vitro) | [46] |
| Melanin from the muscles of Gallus domesticus Brisson | Antioxidant | concentration-dependent scavenging of DPPH and superoxide radicals; inhibition of lipid peroxidation | 20–3000 µg/mL (in vitro) | [47] |
| Bacterial melanin from Streptomyces glaucescens NEAE-H | Antioxidant Anticancer Anti-hemolytic | scavenging and neutralization of ABTS radical; ↑ mortality and cytotoxicity in HFB4 skin cancer cells; neutralization of free radicals and protection of erythrocytes from membrane destruction/lysis | 1.56–100 µg/mL; IC50 = 16.34 ± 1.31 µg/mL (in vitro) | [48] |
| Bacterial melanin from Pseudomonas maltophilia AT18 | Photoprotective | ↑ viability of normal fibroblasts (NL-FB) post-UVA irradiation; inhibition of UVA-induced apoptosis; suppression of intracellular ROS generated by UVA | 25–800 µg/mL (in vitro) | [49] |
| Bacterial melanin from Pseudomonas otitidis DDB2 | Photoprotective | protection of NIH 3T3 mouse fibroblasts against UVB radiation; scavenging of ROS generated upon UVB irradiation | 15.625–500 µg/mL (in vitro) | [50] |
| Herbal melanin from Nigella sativa seed coats | Immunomodulatory | ↑ TNF-α, IL-6, and VEGF mRNA expression in human monocytic THP-1 cells and peripheral blood mononuclear cells (PBMC) | 50 and 100 µg/mL (in vitro) | [51] |
| Immunomodulatory | ↑ IL-8 expression and production in human monocytic THP-1 cells and peripheral blood mono- nuclear cells (PBMC) | 5–50 µg/mL (in vitro) | [52] | |
| Anticancer | ↓ cell viability; ↑ generation of cellular ROS; apoptosis induction; ↓ Bcl-2 expression; ↑ Bad expression; ↑ cytochrome c expression; activation of caspase-3 and -7; ↑ JNK, cJun and ATF2 phosphorylation; ↓ ERK phosphorylation in HT-29 and SW620 colorectal adenocarcinoma cells | 5–200 µg/mL (in vitro) | [53] | |
| Anticancer | ↓ cell viability; cell growth arrest in G0/G1 and G2 phases; ↑ TLR4 protein expression; apoptosis induction in human acute monocytic leukemia THP-1 and human embryonic kidney HEK293 cells | 7.8–500 µg/mL (in vitro) | [54] | |
| B16F10 melanoma tumor lysates containing melanin (microneedle patch) | Anticancer | Melanin-mediated heat generation; promotion of tumor-antigen uptake by dendritic cells; ↑ antitumor vaccination against B16F10 tumors; complete tumor remission in BRAFV600E-mutated BP melanoma- and 4T1 breast carcinoma-bearing mice | around 50 µg of melanin/patch (in vivo) | [55] |
| Synthetic melanin | Immunomodulatory | ↑ CD8+ T-cell responses and inhibition of tumor growth in BALB/c mice; ↑ efficiency of melanin as adjuvant in anticancer vaccines | 0.5 μg of melanin bound to the gp100 epitope (gp100-melanin) (in vivo) | [56] |
| Application | Melanin Nanoplatform Type | Cancer Type | Observations | Concentration/Dosage | Reference |
|---|---|---|---|---|---|
| Chemotherapy | Doxorubicin-loaded MEL-NPs | Thyroid cancer | Chelating of doxorubicin (DOX) through π-π stacking and hydrogen bonding; ↓ viability of HTh74 and HTh74R thyroid cancer cells; ↑ therapeutic efficacy and cell internalization compared to free doxorubicin. | 10, 20, 40, 80, 160 mg/L (in vitro) | [98] |
| PDA-coated and alendronate-grafted paclitaxel (PTX) nanoparticles | Osteosarcoma | Targeted cancer treatment; sustained drug release; ↑ cytotoxicity against K7M2 wt osteosarcoma cells; ↑ accumulation in tumor, and ↓ side effects of PTX in K7M2 wt tumor-bearing mice | 1, 5, 10, 50, 100 µg/mL (in vitro) 8 mg/kg equivalent concentrations of PTX (in vivo) | [99] | |
| PTX-loaded trastuzumab-decorated PDA-NPs (PDA NPs•Tmab@PTX) | Breast cancer | ↓ viability of BT474, SKBR3, and HS5 cells HER2+ breast cancer cells; ↑ number of early apoptotic HER2+ breast cancer cells BT474; disintegration and ↓ viability BT474 spheroids | 0.035, and 0.042 mg/mL (2D in vitro model) 0.035 mg/mL (3D in vitro model) | [100] | |
| RGD-modified polydopamine-paclitaxel-loaded poly (3-hydroxybutyrate-co-3-hydroxyvalerate) nanoparticles | Hepatocellular carcinoma | ↓ cytotoxicity against L02 of PTX-free NPs; ↓ viability of HepG2 and SMMC-7721 cells; ↑ inhibitory effect on HepG2 and SMMC-7721 cell proliferation compared to free PTX; ↑ cellular uptake in HepG2 cells; ↑ PTX release at pH values of 5.0–6.5; ↓ tumor volume and weight in HepG2 tumor-bearing mice | 0.1, 0.5, 1, 2.5, 5, 10 µg/mL (in vitro) 4 mg/kg (in vivo) | [101] | |
| Doxorubicin-loaded polyethylene glycol functionalized MEL-NPs | Breast cancer | Sustained and extended release of doxorubicin; ↓ proliferation of MDA-MB-231 breast cancer cells; | 0.125, 0.250, 0.500 mg (in vitro) | [102] | |
| Curcumin-loaded silver-decorated melanin-like polydopamine/mesoporous silica composites | Cervical and Taxol-resistant non-small cell lung cancers | ↓ hemolytic activity and biocompatibility; pH- and ROS-responsive release of curcumin; prolonged inhibition of Escherichia coli and Staphylococcus aureus bacterial growth; ↑ chemotherapeutic efficiency against HeLa (human cervical) and A549/TAX (Taxol-resistant non-small cell lung) cancer cells compared to free curcumin. | ≤ 500 µg/mL (in vitro) | [103] | |
| Gambogenic acid-loaded functional polydopamine nanoparticles (GNA@PDA-FA SA NPs) | Breast cancer | ↓ 4T1 (breast cancer cells) cell viability; ↓ IC50 value compared to raw GNA; ↑ targeting effect of GNA against 4T1 cells; inhibition of tumor growth in 4T1 xenograft-bearing BALB/C mice | 0.78–310 µM (in vitro) 24 mg/kg (in vivo) | [104] | |
| Iron-chelated doxorubicin-loaded folic acid-conjugated polyethylene glycol (PEG)-coated polydopamine nanoparticles (DOX@Fe-PDA/FA-PEG NPs) | Breast cancer | ↑ pH responsiveness of the PDA-modified NPs and pH-dependent release of DOX; ↑ DOX release under acidic conditions; sustained DOX release; ↑ cell uptake compared to free DOX; ↓ MCF7 (breast cancer cells) cell viability; ↑ intracellular ROS in MCF7 cells | 0.1093–3.5 µg/mL (in vitro) | [105] | |
| Doxorubicin-loaded polyethylene glycol-modified polydopamine nanoparticles (PDA-PEG-DOX) | Breast cancer | ↓ MDA-MB-231 (breast cancer cells) cell viability; ↓ pro-caspase 3 expression level; accumulation within the MDA-MB-231 cell nucleus and lysosomes; ↓ mitochondrial membrane potential | 0.5, 1, and 5 µg/mL (in vitro) | [106] | |
| Doxorubicin-loaded triphenylphosphonium- functionalized polyethylene glycol-modified polydopamine nanoparticles (PDA-PEG-TPP-DOX) | Breast cancer | ↓ MDA-MB-231 cell viability; ↓ pro-caspase 3 expression level; ↓ mitochondrial membrane potential; mitochondria-targeted anticancer effect; ↓ long-term drug resistance. | 0.5, 1, and 5 µg/mL (in vitro) | [106] | |
| Radio (pharmaceutical) Therapy | Melanin-covered silica nanoparticles (MNs) | - | ↓ hematologic toxicity in mice exposed to external gamma radiation and radioimmunotherapy | 50 mg/kg (in vivo) | [107] |
| Melanin nanoparticles (MNPs) | - | ↓ gamma radiation-induced cytotoxicity in Chinese hamster ovary cells | 6.25, 12.5, 25 and 50 µg/mL (in vitro) | [108] | |
| 131I-labeled PEGylated polydopamine nanoparticles loaded with sanguinarine and metformin (131I-PDA- PEG-SAN-MET) | Breast cancer | ↓ 4T1 cell viability; induction of 4T1 cell apoptosis; relieved tumor hypoxia in 4T1 tumor-bearing nude mice. | NPs containing 4 mg/kg of SAN, 8 mg/kg of MET, 200 mCi of 131I (in vivo) | [109] | |
| PEGylated polydopamine nanoparticles loaded with 131I and DOX (131I-PDA-PEG/DOX) | Breast cancer | ↓ 4T1 cell viability; ↑ cellular 131I uptake delivered by PDA-PEG; inhibited tumor growth, reduced tumor size, and prolonged survival rate in BALB/c mice bearing 4T1 xenografts. | 10 mg/kg of PDA-PEG, 20 mCi of 131I (in vivo) | [110] | |
| Phototherapy | Arginine-doped synthetic melanin nanoparticles (SMNPs) | Breast cancer | ↑ photothermal efficiency following arginine introduction within the PDA structures of SMNPs; ↓ 4T1 cell viability; ↓ tumor volume and weight in 4T1 xenograft-bearing female BALB/c nude mice. | 12.5, 25, 50, 100, 200 μg/mL (in vitro) 10.0 and 20.0 mg/kg (in vivo) | [111] |
| RGD- and beclin 1-modified and PEGylated MEL-NPs | Anticancer | Induced autophagy and cytotoxicity; ↓ cell viability upon NIR irradiation in cancer cells; tumor regression in BALB/c nude mice at 43 °C | 50 μg/mL (in vitro) 34 mg/kg (in vivo) | [112] | |
| Cisplatin prodrug Pt (IV) MEL-NPs | Prostate cancer | ↓ viability of PC3, DU145, and LNCaP prostate cancer cells; induction of mitochondrial membrane depolarization in PC3 cells; ↑ cell uptake ability; synergistic photothermal therapy and chemotherapy properties; potent photothermal conversion efficiency (29.6%); biocompatibility; prolonged the blood circulation time and ↓ tumor growth in BALB/c mice. | 10, 20, 30 µM (in vitro) 200 µL (in vivo) | [113] | |
| Gemcitabine-loaded dual-functional melanin-based nanoliposomes | Pancreatic cancer | Synergistic antitumor effect between melanin and gemcitabine; ↑ therapeutic efficiency; potent conversion of NIR light into thermal energy in the presence of MEL; photothermal conversion efficiency of MEL uninfluenced by liposomal encapsulation and drug loading; ↓ cell viability of BxPC-3 pancreatic cancer cells; controlled and enhanced drug release to the tumor sites via hyperthermia; no systemic toxicity to BxPC-3 tumor-bearing mice | 50 mg/kg (in vivo) | [114] | |
| Docetaxel-loaded polydopamine-functionalized CA-(PCL-ran-PLA) nanoparticles | Breast cancer | ↑ drug loading content, and encapsulation efficiency; effective target delivery of drugs to tumor sites by incorporating AS1411 aptamers; synergistic chemo-photothermal effect; ↓ proliferation of MCF-7 breast cancer cells; ↑ survival time, and ↓ side effects in mice; ↓ tumor volume in vivo | 0.25–25 μg/mL (in vitro) 10 mg/kg (in vivo) | [115] | |
| PDA/transferrin hybrid NPs | Malignant melanoma | ↑ apoptosis when associated with UV irradiation in B16F10 mouse melanoma cells, J774A.1 mouse macrophages, and in an organotypic melanoma spheroid model; lack of cytotoxicity or proliferation impairment of PDA-NPs in B16F10 and J774A.1; | 5–160 µg/mL (in vitro) | [116] | |
| Hyaluronic acid-decorated polydopamine nanoparticles with conjugated chlorin e6 (HA–PDA–Ce6) | Colorectal carcinoma | ↓ dark toxicity; ↑ photodynamic and photothermal activities upon laser illumination; ↑ uptake and penetration in vitro and in vivo; ↑ cytotoxicity and apoptosis in HCT-116 cells following the combined laser treatment; inhibited tumor growth in HCT-116 tumor-bearing mice. | IC50 = 33.07 ± 12.92 μg/mL (in vitro) 0.65 mg/kg (in vivo) | [117] | |
| Epirubicin-hybrid polydopamine nanoparticles (E/PCF-NPs) | Breast cancer | pH sensitive drug release; ↑ cytotoxicity against 4T1 cells; inhibited survival rate and induced cell apoptosis 4T1 cells; ↑ ROS generation; ↓ NAD+/NADH; complete tumor regression in 4T1 tumor-bearing mice | IC50 = 1.3 ± 0.2 μg/mL (in vitro) 5 mg/kg drug dose (in vivo) | [118] | |
| Folate-modified PDA nanoparticles loaded with a cationic phthalocyanine-type photosensitizer (PDA-FA-Pc) | Breast cancer Cervical cancer | Non-measurable toxicity of PDA-FA-Pc without illumination; ↓ dose-dependent survival rate of MCF-7, HeLa, HELF, and L02 cells following illumination; ↑ cytotoxicity against tumor cells (MCF-7, HeLa) compared to healthy cells (HELF, L02); ↓ tumor volume and weight in MCF-7 and HeLa xenograft-bearing female Kunming mice. | 0.15, 0.3, 0.6, 1.2 and 2.4 mg/mL (in vitro) 43.5 mg/kg (in vivo) | [119] | |
| Chlorin e6-conjugated PDA nanospheres | Hepatocellular carcinoma | Simultaneous PTT and PDT therapy; ↑ internalization within HepG2 cells; ↓ cell viability of HepG2 cells; tumor regression in HepG2 tumor-bearing male BALB/c-nude mice | Ce6 concentration 0.1–8 μg/mL (in vitro) 20 μg/mL PDA and 5 μg/mL Ce6 (in vivo) | [120] | |
| PDA nanoparticles carrying tumor cell lysate (TLC) (TCL@PDA NPs) | Delayed cancer progression in tumor-bearing mice; ↑ antigen uptake, BMDCs (bone-marrow-derived dendritic cells) maturation, and Th1-related cytokines secretion; ↑ CD4+ and CD8+ T cells; delayed tumor development by empty PDA-NPs | 300 μg TLC | [121] | ||
| Immunotherapy | Polydopamine-coated mesoporous silica nanoparticles containing thiolated ovalbumin and ammonium bicarbonate (MSNs-ABC@PDA-OVA) | Malignant melanoma | Rapid antigen release and endosome escape under laser illumination; ↑ activation and maturation of dendritic cells; antigen specific CD8+ and Th1 CD4+ T cell responses; melanoma eradication with a cure rate of 75%; strong immunological memory; inhibition of tumor recurrence and metastasis in C57BL/6 mice. | 25 µg OVA/mouse (in vivo) | [122] |
| Antigen-ovalbumin-loaded polydopamine nanoparticles (OVA@Pdop-NPs) | Colon cancer | Lack of cytotoxicity and ↑ cellular uptake in bone marrow-derived dendritic cells (BMDCs); ↑ maturation of dendritic cells; ↑ expression of major histocompatibility complex, costimulatory molecules, and cytokines; activation of OVA-specific cytotoxic CD8+ T cells; ↑ production of memory CD4+ and CD8+ T cells; ↓ tumor growth in OVA-MC38 colon tumor-bearing mice | 0.5–100 μg/mL (in vitro) 100 μg/mice OVA content (in vivo) | [123] | |
| Natural melanin nanoparticles coated with cancer cell membrane (M@C NPs) | Breast cancer | ↑ antitumor activity; ↑ levels of CD8+ T cells and cytokines; ↑ 4T1 cell cytotoxicity and ↓ cell invasion under laser radiation; ↑ expression of calreticulin proteins under irradiation suggesting immunogenic cell death of 4T1 cells; ↑ tumor targeting ability, ↓ levels of IL- 12 and IL-6, and synergistic effect with immunoblocking inhibitors (IDOi) leading to ↓ tumor volume and growth in mice. | ≤ 1000 μg/mL (in vitro) | [124] | |
| Gene Therapy | pH-responsive polydopamine nanoparticles modified with polyethylenimine and polyethylene glycol-phenylboronic acid (PDANP-PEI-rPEG) | Hepatocellular carcinoma (in vitro) Malignant melanoma (in vivo) | Stability to physiological pH (7.4); ↑ gene transfection levels; ↑ photothermal conversion ability; quick endosomal escape; | 0.4–1.5 mass ratio PDANP to DNA (in vitro) 50 μL (in vivo) | [125] |
| DNA-polydopamine-MnO2 nanocomplex (DP-PM) | Breast cancer | ↓ viability of MCF7 cells; ↓ tumor volume and weight in MCF7 xenograft-bearing BALB/c nude mice; glutathione-triggered release of Mn2+ to activate intracytoplasmic DNAzyme ↑ Egr-1 mRNA cleavage activity of DNAzyme and ↓ of Egr-1 protein in tumor cells; synergistic tumor ablation upon NIR irradiation. | 5–50 μg/mL (in vitro) | [126] | |
| Polyethylenimine-modified polydopamine nanoparticles (PPNPs) | Hepatocellular carcinoma | ↓ cytotoxicity to HepG2 cells; ↑ gene transfection levels compared to Lipofectamine 2000 at mass ratios of 23 and 30; tripled gene transfection levels following NIR irradiation; lack of hemolytic effect. | 10–30 mass ratio PPNPs to DNA (in vitro) | [127] | |
| Cancer Detection and Bio-Imaging | Mesoporous polydopamine carrying sorafenib and SPIO nanoparticles (SRF@MPDA-SPIO NPs) | Hepatocellular carcinoma | ↑ MRI contrast; ↑ R2 (1/T2) values; MRI-guided ferroptosis; responsive release of ferric ions and sorafenib to stimuli (pH, temperature); effectively conversion ability of NIR light; reduced tumor volume and weight in HCT-116 tumor-bearing mice | 100 μL (in vivo) | [128] |
| Ions (Fe3+, Bi3+, I+)-doped melanin nanoparticles conjugated with EGFR antibody (iMNPs) | Hepatocellular carcinoma | ↑ contrast intensity in T1-w MRI and CT; specific targeting of EGFR-overexpressed HepG2 cells observed by MRI and CT imaging; ↑ contrast of MRI/CT/SPECT images in xenograft-bearing-mice | 200 μL (in vivo) | [129] | |
| Nanotheranostics | PDA-based theranostic nanoprobe loaded with fluorescein isothiocyanate (FITC)-labeled hairpin DNA (hpDNA) and doxorubicin | Breast cancer | ↓ viability of 4T1 breast cancer cells; real-time detection of the dynamic expression of specific miRNAs; ↓ tumor volume in 4T1 xenograft-bearing male BALB/c-nu mice | 2.5, 5, 10, 20 μg/mL of doxorubicin (in vitro) | [130] |
| Cu (II)-doped polydopamine-coated gold nanorods | Squamous cell carcinoma | ↑ physiological stability, biocompatibility, photothermal performance, and blood circulation time; computer tomography imaging and magnetic resonance imaging functions; ↓ tumor volume and weight; lack of short-term toxicity against liver and renal functions in BALB/c mice | 25–500 µg/mL (in vitro) 50 μL of 5 mg/mL (in vivo) | [131] | |
| Mn2+ -coordinated PDA-modified doxorubicin-loaded poly (lactic- co-glycolic acid) (PLGA) NPs | Colon cancer | ↑ permeability and retention; ↑ ability of NIR photothermal transduction in vitro and in vivo; chemo-photothermal synergistic effect; ↑ DOX release; ↓ viability of CT26 murine colorectal carcinoma cells; stronger efficacy in killing cancer cells under NIR irradiation; efficient cellular uptake; ↓ tumor growth in CT26 tumor-bearing mice; no acute side effects in vivo | ≤ 200 µg/mL (in vitro) 20 mg/kg (in vivo) | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcovici, I.; Coricovac, D.; Pinzaru, I.; Macasoi, I.G.; Popescu, R.; Chioibas, R.; Zupko, I.; Dehelean, C.A. Melanin and Melanin-Functionalized Nanoparticles as Promising Tools in Cancer Research—A Review. Cancers 2022, 14, 1838. https://doi.org/10.3390/cancers14071838
Marcovici I, Coricovac D, Pinzaru I, Macasoi IG, Popescu R, Chioibas R, Zupko I, Dehelean CA. Melanin and Melanin-Functionalized Nanoparticles as Promising Tools in Cancer Research—A Review. Cancers. 2022; 14(7):1838. https://doi.org/10.3390/cancers14071838
Chicago/Turabian StyleMarcovici, Iasmina, Dorina Coricovac, Iulia Pinzaru, Ioana Gabriela Macasoi, Roxana Popescu, Raul Chioibas, Istvan Zupko, and Cristina Adriana Dehelean. 2022. "Melanin and Melanin-Functionalized Nanoparticles as Promising Tools in Cancer Research—A Review" Cancers 14, no. 7: 1838. https://doi.org/10.3390/cancers14071838
APA StyleMarcovici, I., Coricovac, D., Pinzaru, I., Macasoi, I. G., Popescu, R., Chioibas, R., Zupko, I., & Dehelean, C. A. (2022). Melanin and Melanin-Functionalized Nanoparticles as Promising Tools in Cancer Research—A Review. Cancers, 14(7), 1838. https://doi.org/10.3390/cancers14071838










