Diagnosis and Improvement of Combustion Characteristics of Methanol Miniature Reciprocating Piston Internal Combustion Engine
Abstract
1. Introduction
2. Combustion Characteristics Test of Methanol Fuel in Micro-Space
3. Effect of Additives on the Combustion Characteristics of the Platinum Wire Glow Ignition in Micro-Space
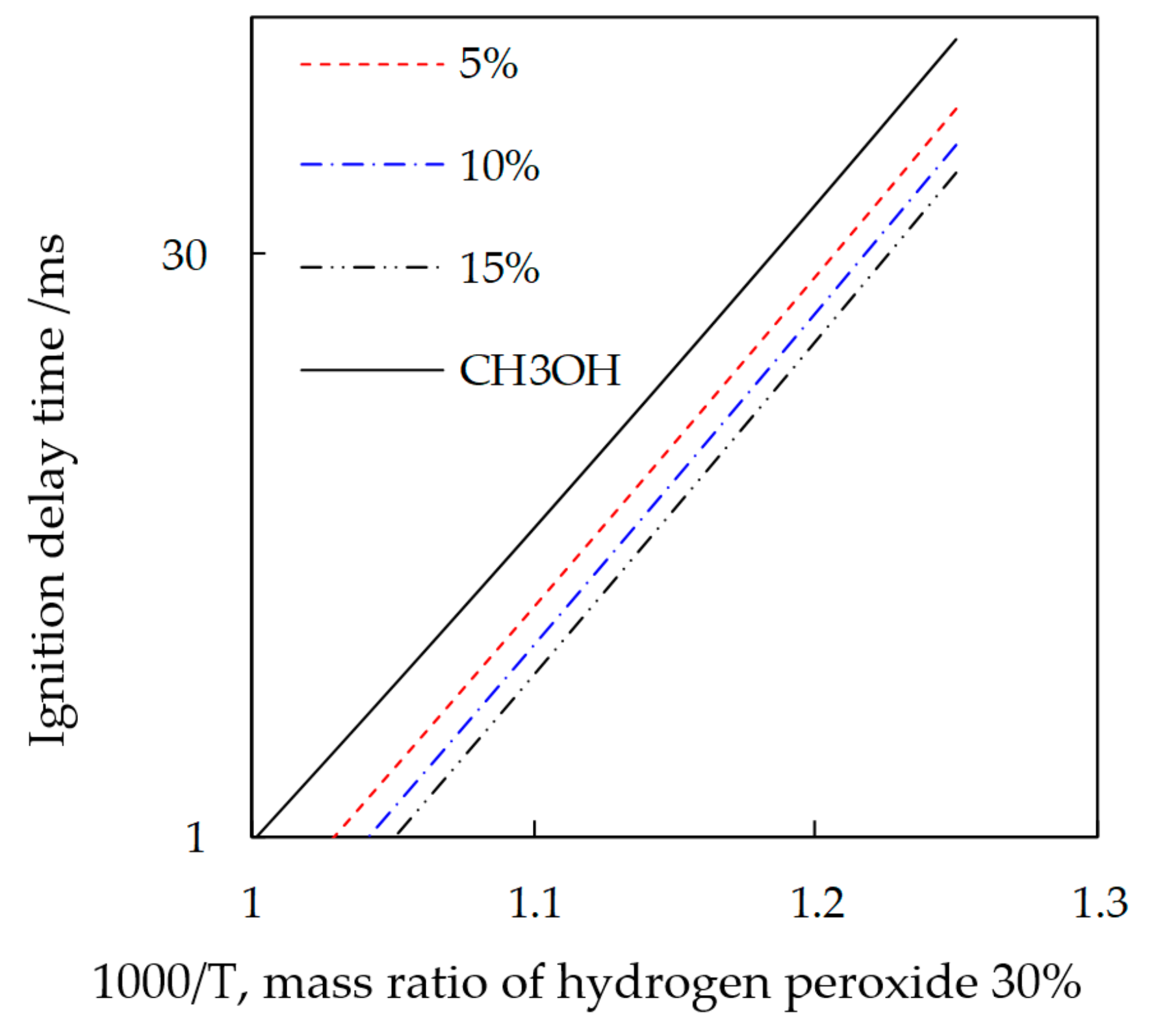
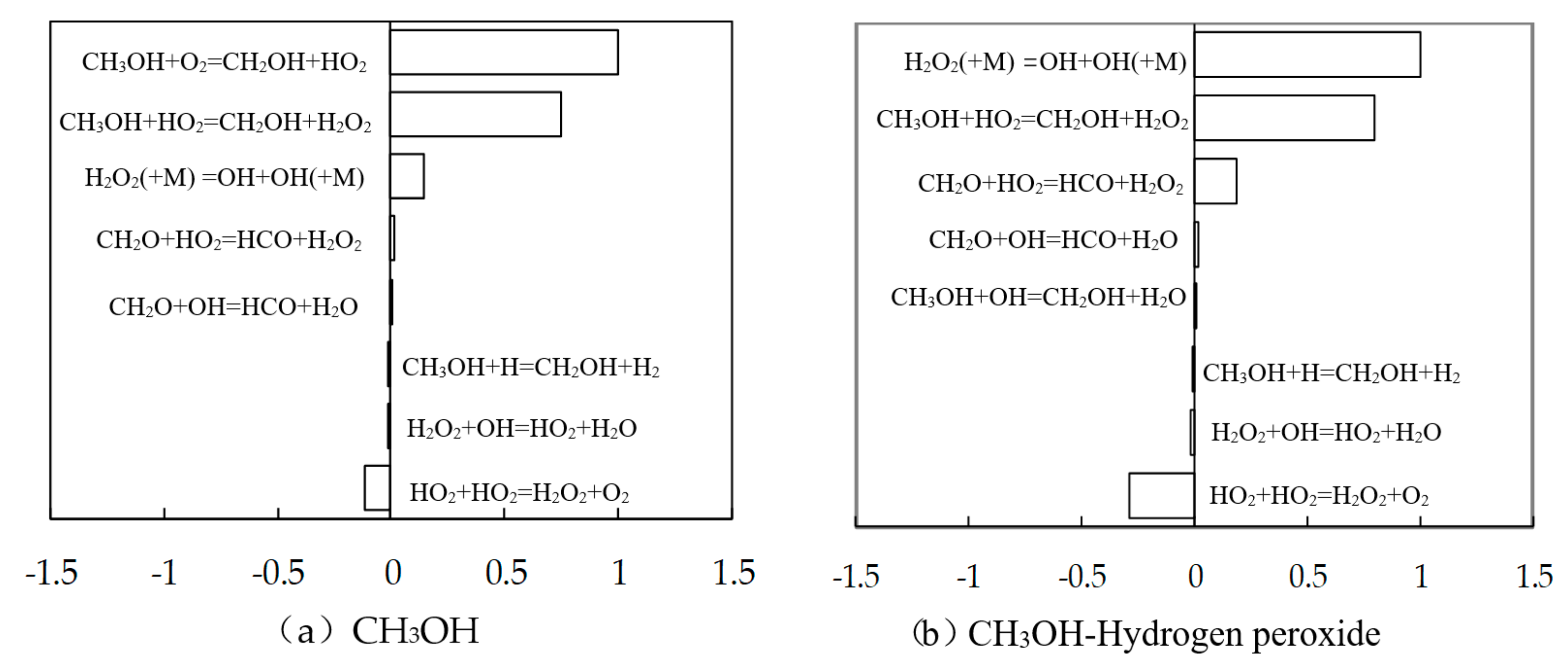
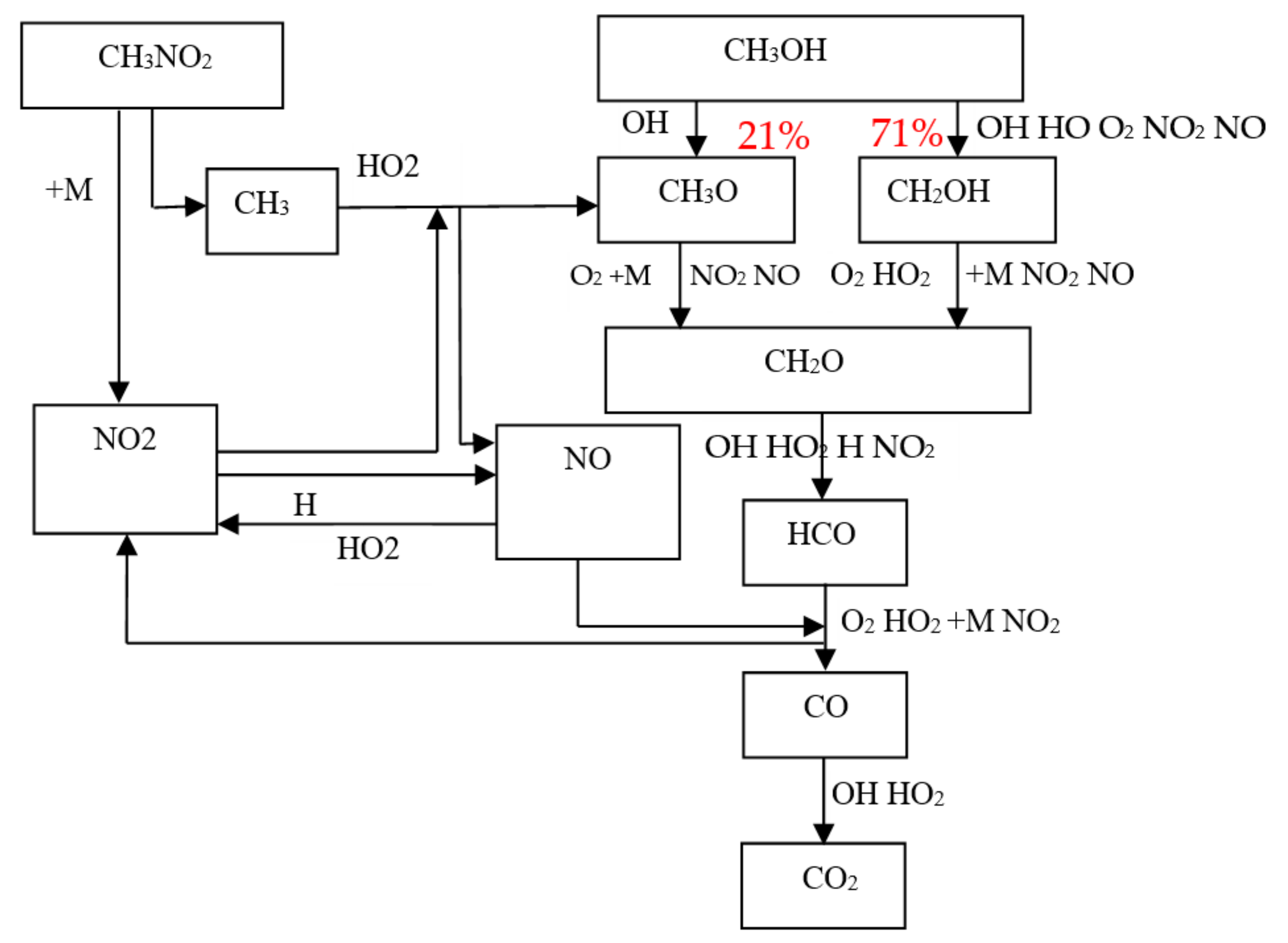
3.1. Study on the Promotion Mechanism of Nitromethane and Hydrogen Peroxide to Methanol Combustion
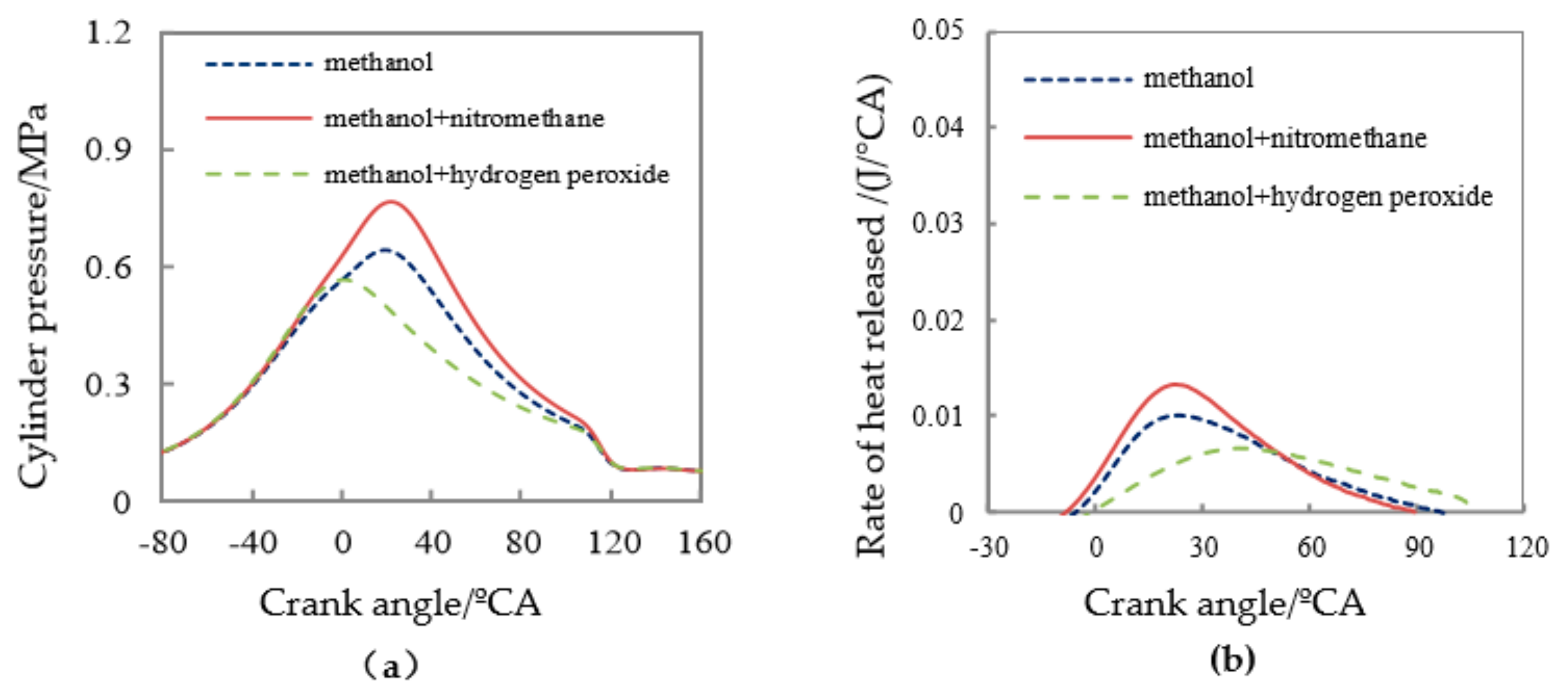
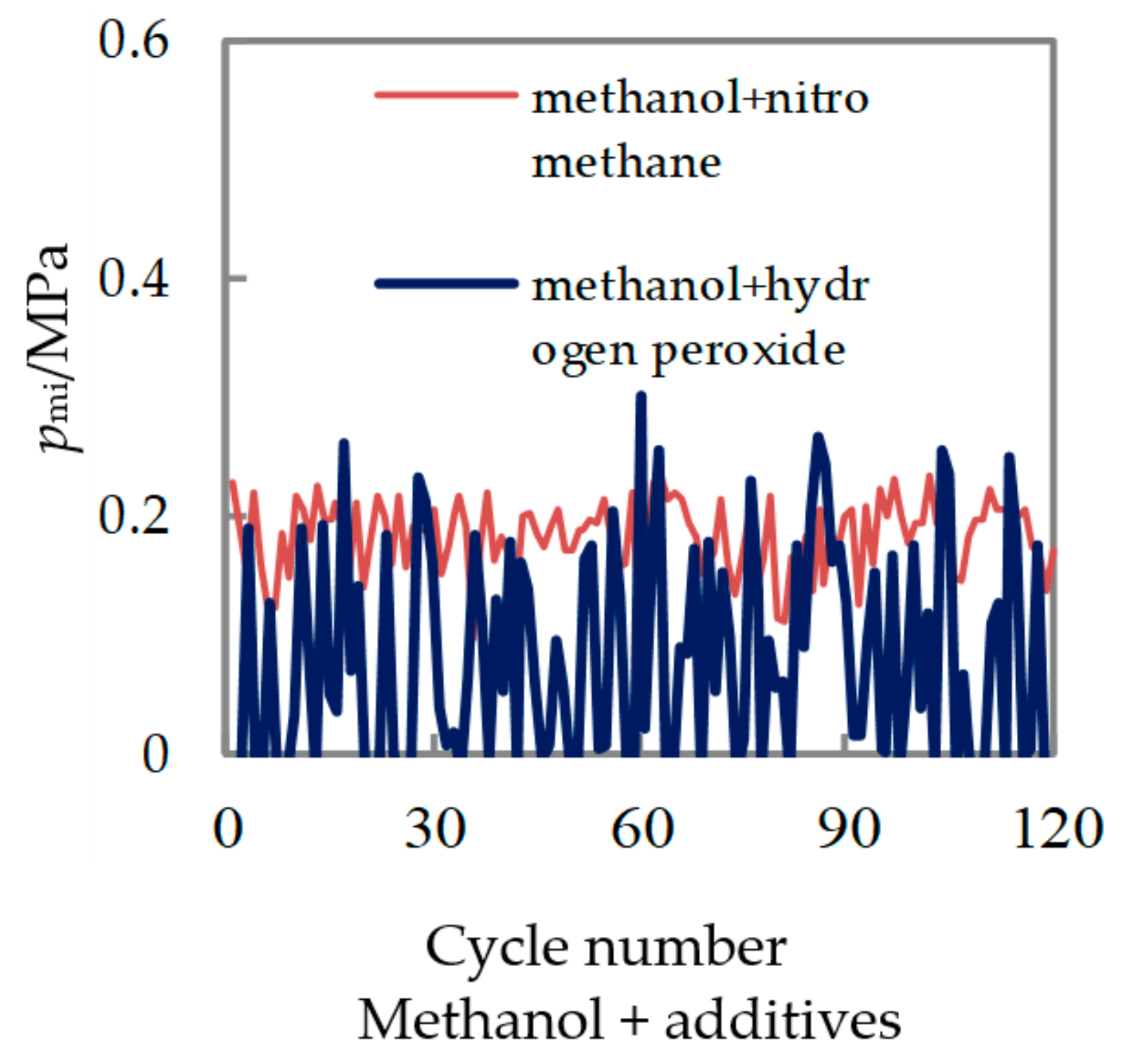
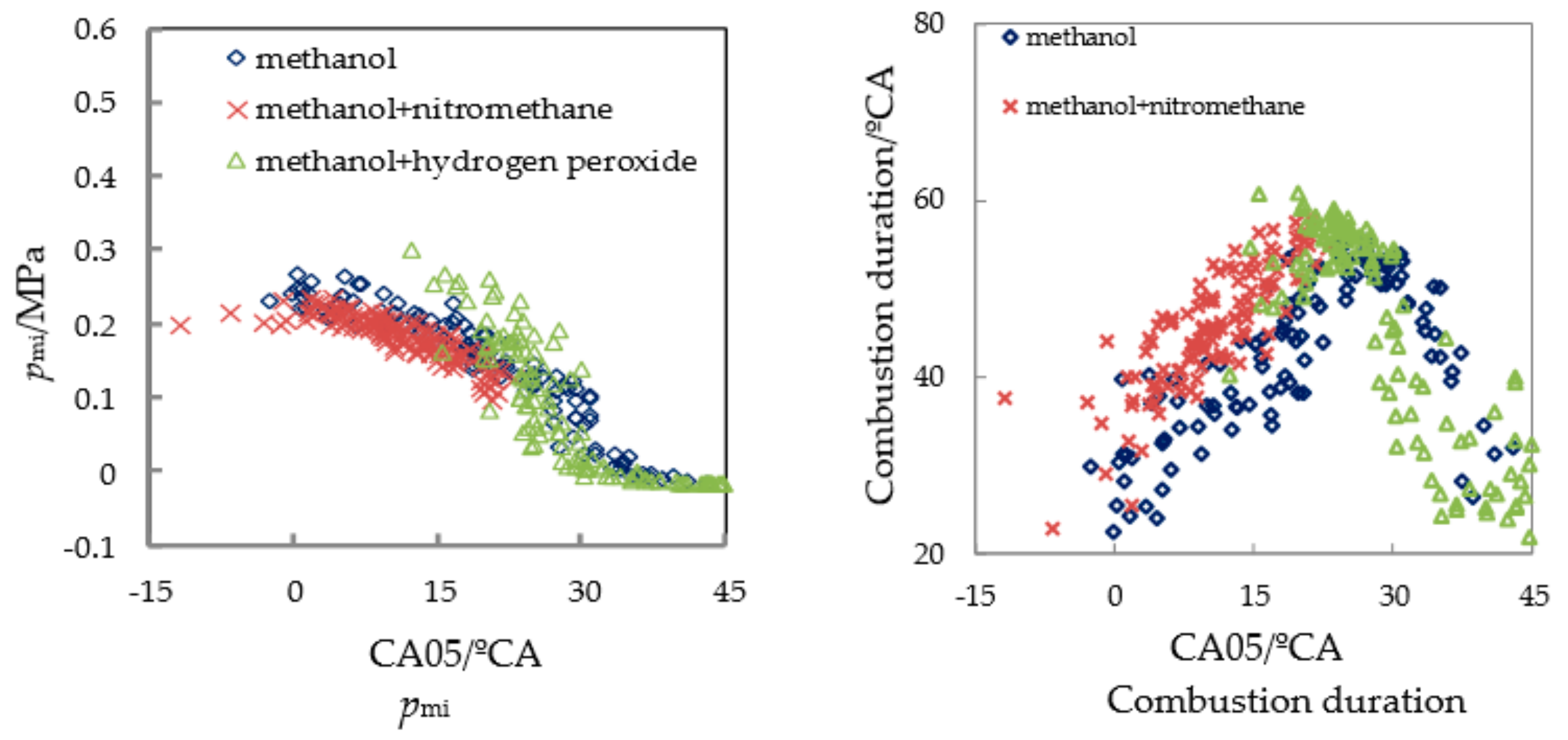
3.2. Test of Methanol Combustion Enhanced by Nitromethane and Hydrogen Peroxide
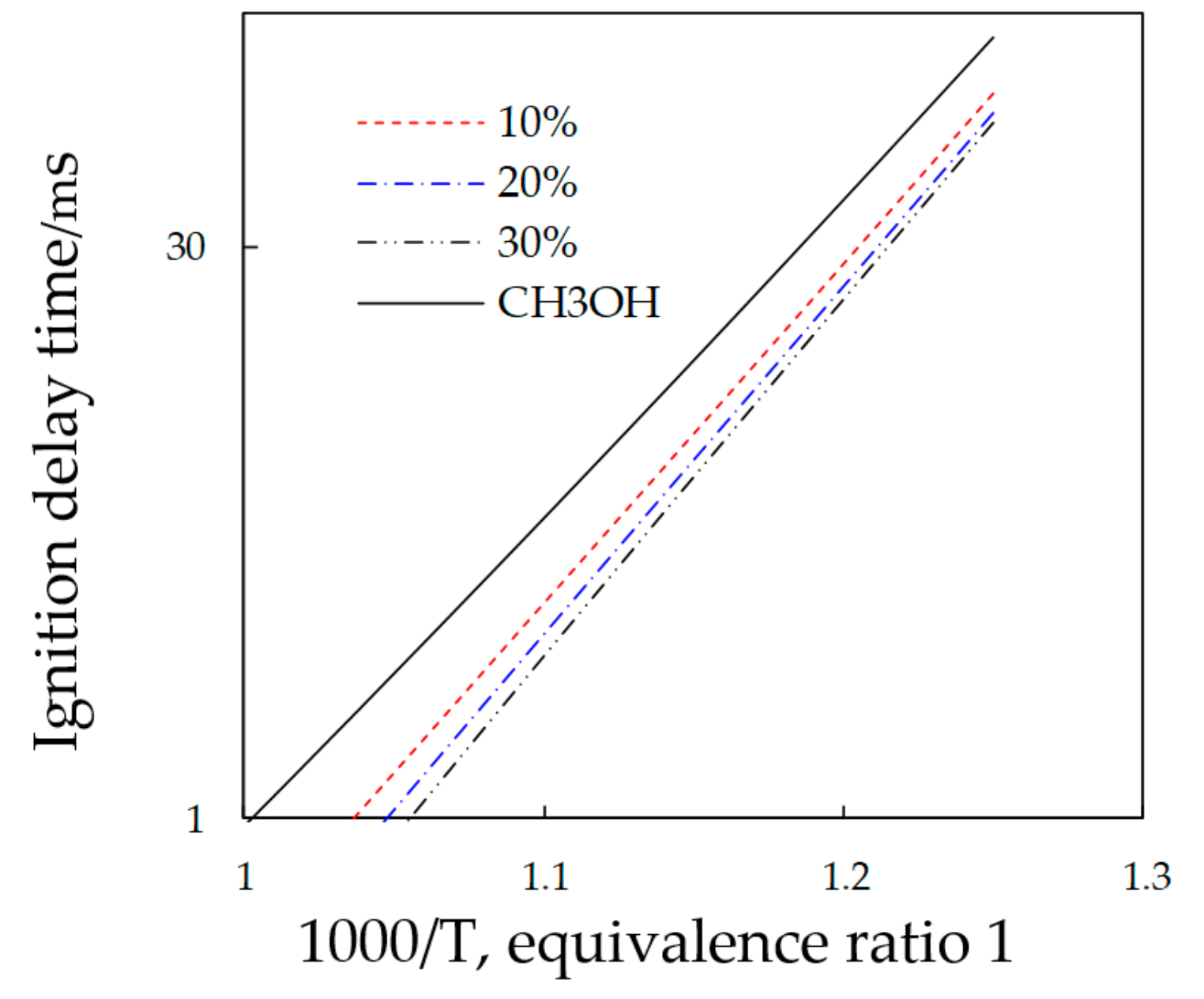
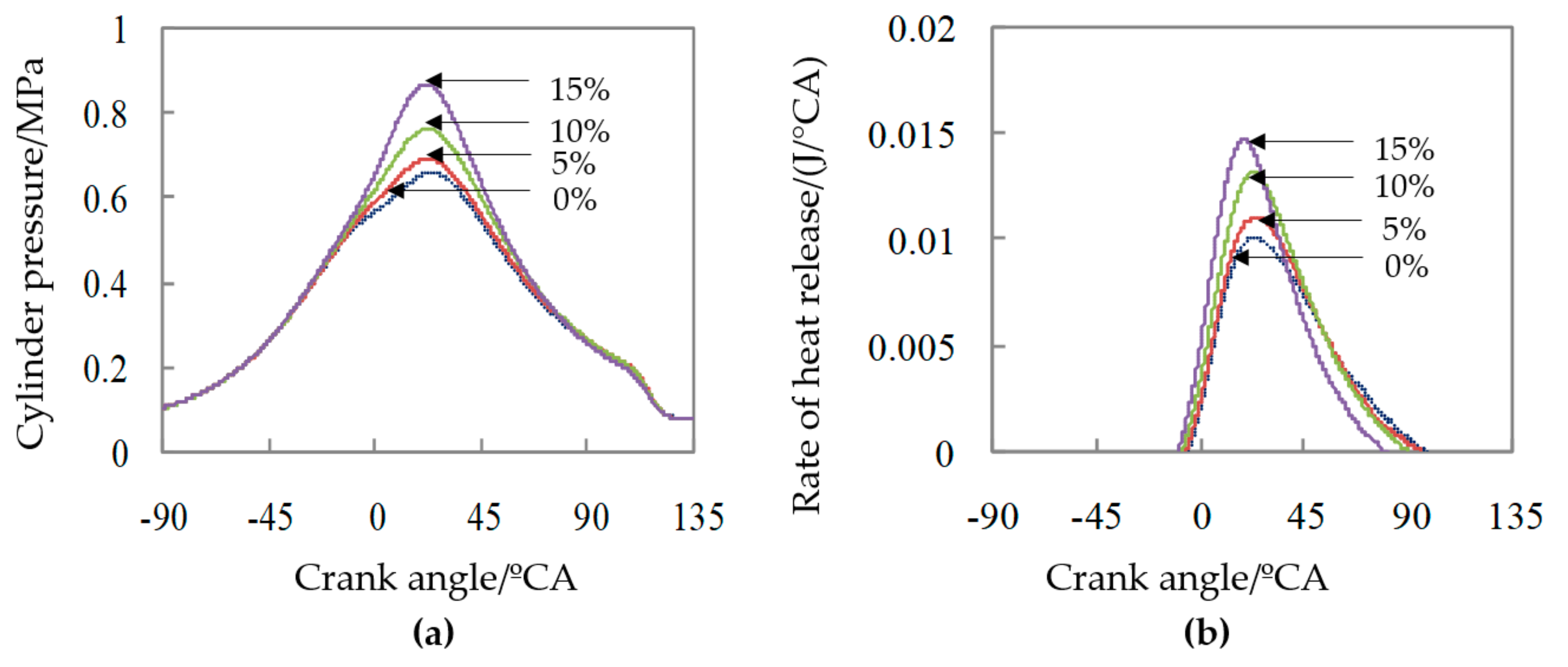
4. Effect of Nitromethane with Different Proportions on the Methanol Combustion Characteristics in Micro-Space
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chou, S.K.; Yang, W.M.; Chua, K.J.; Li, J.; Zhang, K.L. Development of micro power generators-A review. Appl. Energy 2011, 88, 1–16. [Google Scholar] [CrossRef]
- Sher, E.; Sher, I. Theoretical limits of scaling-down internal combustion engines. Chem. Eng. Sci. 2011, 66, 260–267. [Google Scholar] [CrossRef]
- Epstein, A.H. Millimeter-scale, micro-electro-mechanical systems gas turbine engines. J. Eng. gas Turbines Power 2004, 126, 205–226. [Google Scholar] [CrossRef]
- Fernandez-Pello, A.C.; Pisano, A.P.; Fu, K.; Walther, D.C.; Knobloch, A.; Martinez, F.; Senesky, M.; Stoldt, C.; Maboudian, R.; Sanders, S.; et al. MEMS rotary engine power system. IEEJ Trans. Sens. Micromach. 2003, 123, 326–330. [Google Scholar] [CrossRef]
- Fréchette, L.G.; Lee, C.; Arslan, S. Development of a MEMS-based rankine cycle steam turbine for power generation: Project status. In Proceedings of the Power MEMS 2004, 4th Int’l Workshop on Micro and Nano Technology for Power Gene. & Energy Conv. Apps (Power MEMS’04), Kyoto, Japan, 28–30 November 2004; pp. 92–95. [Google Scholar]
- Guo, Z.P.; Wang, Y.F.; Zhang, H.J.; Lu, Y.Z.; Li, G.F.; Guo, W. Transient dynamics analysis of micro free piston swing engine. Chin. Intern. Combust. Engine Eng. 2014, 35, 119–124. [Google Scholar]
- Yang, W.M.; Chou, S.K.; Li, J. Micro thermophotovoltaic power generator with high power density. Appl. Therm. Eng. 2009, 29, 3144–3148. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Cleary, M.; Schoensee, L.; Kempf, N.; Richardson, J. High-performance nanostructured thermoelectric generators for micro combined heat and power systems. Appl. Therm. Eng. 2016, 96, 83–87. [Google Scholar] [CrossRef]
- Chiabrera, F.; Garbayo, I.; Alayo, N.; Tarancón, A. Micro solid oxide fuel cells: A new generation of micro-power sources for portable applications. In Proceedings of the SPIE 2017, 10246: Smart Sensors, Actuators, and MEMS VIII, Barcelona, Spain, 8–11 May 2017; p. 102460S. [Google Scholar]
- Menon, S.; Cadou, P.C. Scaling of miniature piston-engine performance, Part 1: Overall Engine Performance. J. Propul. Power 2013, 29, 774–787. [Google Scholar] [CrossRef]
- Shirsat, V.; Gupta, A.K. A review of progress in heat recirculating meso-scale combustors. Appl. Energy 2011, 88, 4294–4309. [Google Scholar] [CrossRef]
- David, C.W.; Jeongmin, A. Advances and challenges in the development of power-generation systems at small scales. Prog. Energy Combust. Sci. 2011, 37, 583–610. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Q.; Xie, Q.; Bai, J.; Zhang, P.G.; Wu, K. Experimental study of homogeneous charge compression ignition using DME in micro power device. J. Eng. Thermophys. 2014, 35, 814–818. [Google Scholar]
- Chen, J.; Yan, L.; Song, W. Study on catalytic combustion characteristics of the micro-engine with detailed chemical kinetic model of methane-air mixture. Combust. Sci. Technol. 2015, 187, 505–524. [Google Scholar] [CrossRef]
- Zhou, M.Y. Experimental study on premixed catalytic combustion characteristics of hydrogen and alkanes in micro tubes. Master’s Thesis, Zhe Jiang University, Hang Zhou, China, 2016. [Google Scholar]
- Faravelli, T.; Frassoldati, A.; Ranzi, E. Kinetic modeling of the interactions between NO and hydrocarbons in the oxidation of hydrocarbons at low temperatures. Combust. Flame 2003, 132, 188–207. [Google Scholar] [CrossRef]
- Rasmussen, C.L.; Wassard, K.H.; Dam-Johansen, K.; Glarborg, P. Methanol Oxidation in a Flow Reactor: Implications for the Branching Ratio of the CH3OH+OH Reaction. Int. Chem. Kinet. 2010, 40, 423–441. [Google Scholar] [CrossRef]
- Zhang, K.; Li, Y.; Yuan, T.; Cai, J.; Glarborg, P.; Qi, F. An experimental and kinetic modeling study of premixed nitromethane flames at low pressure. Proc. Combust. Inst. 2011, 33, 407–414. [Google Scholar] [CrossRef]
- Li, J.; Zhao, Z.; Kazakov, A.; Chaos, M.; Dryer, F.L.; Scire, J.J., Jr. A comprehensive kinetic mechanism for CO, CH2O, and CH3OH combustion. Int. Chem. Kinet. 2007, 39, 109–136. [Google Scholar] [CrossRef]
- Eric, B.; Kenneth, K.K. Modeling of nitromethane flame structure and burning behavior. Proc. Combust. Inst. 2007, 31, 2045–2053. [Google Scholar]
- Glarborg, P.; Bendtsen, A.B.; Miller, J.A. Nitromethane dissociation: Implications for the CH3+ NO2 Reaction. Int. Chem. Kinet. 1999, 31, 591–602. [Google Scholar] [CrossRef]
- Burcat, A. Third Millennium Ideal Gas and Condensed Phase Thermochemical Database for Combustion; Technion Aerospace Engineering (TAE) Report # 867; Technion-Israel Institute of Technology: Haifa, Israel, 2001. [Google Scholar]
- Melius, F. Thermochemistry and reaction mechanisms of nitromethane ignition. J. Phys. 1995, 5, 535–547. [Google Scholar] [CrossRef]
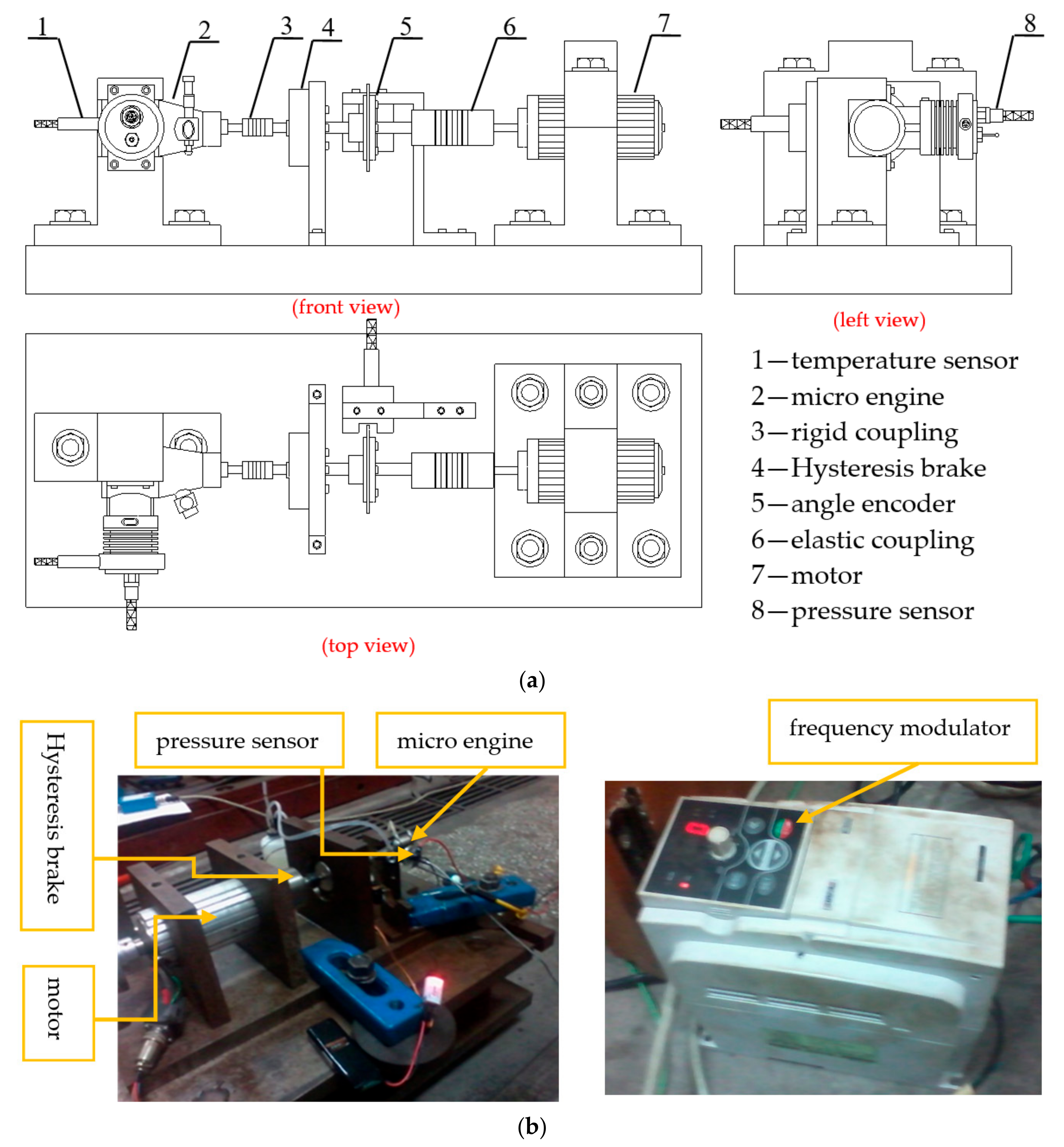
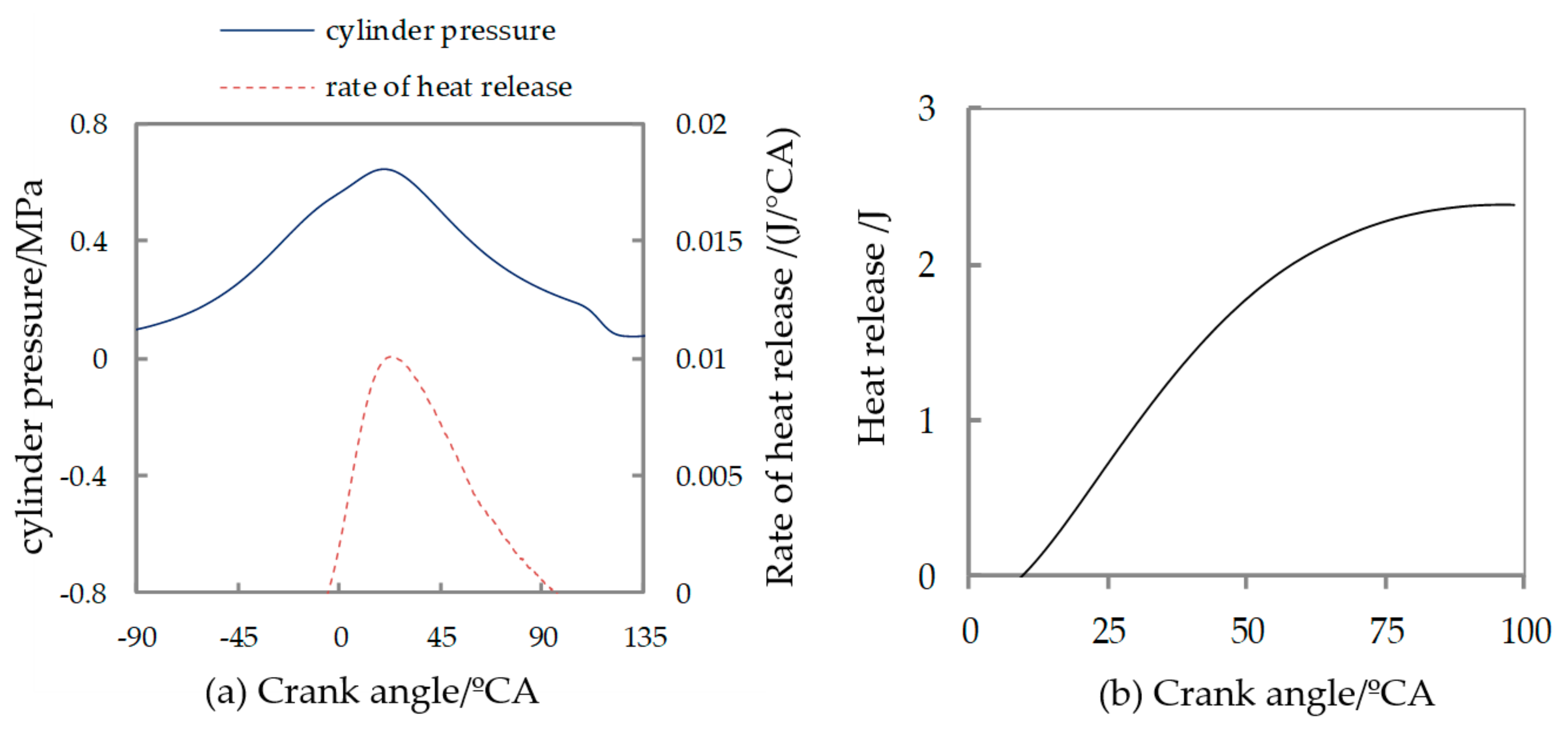

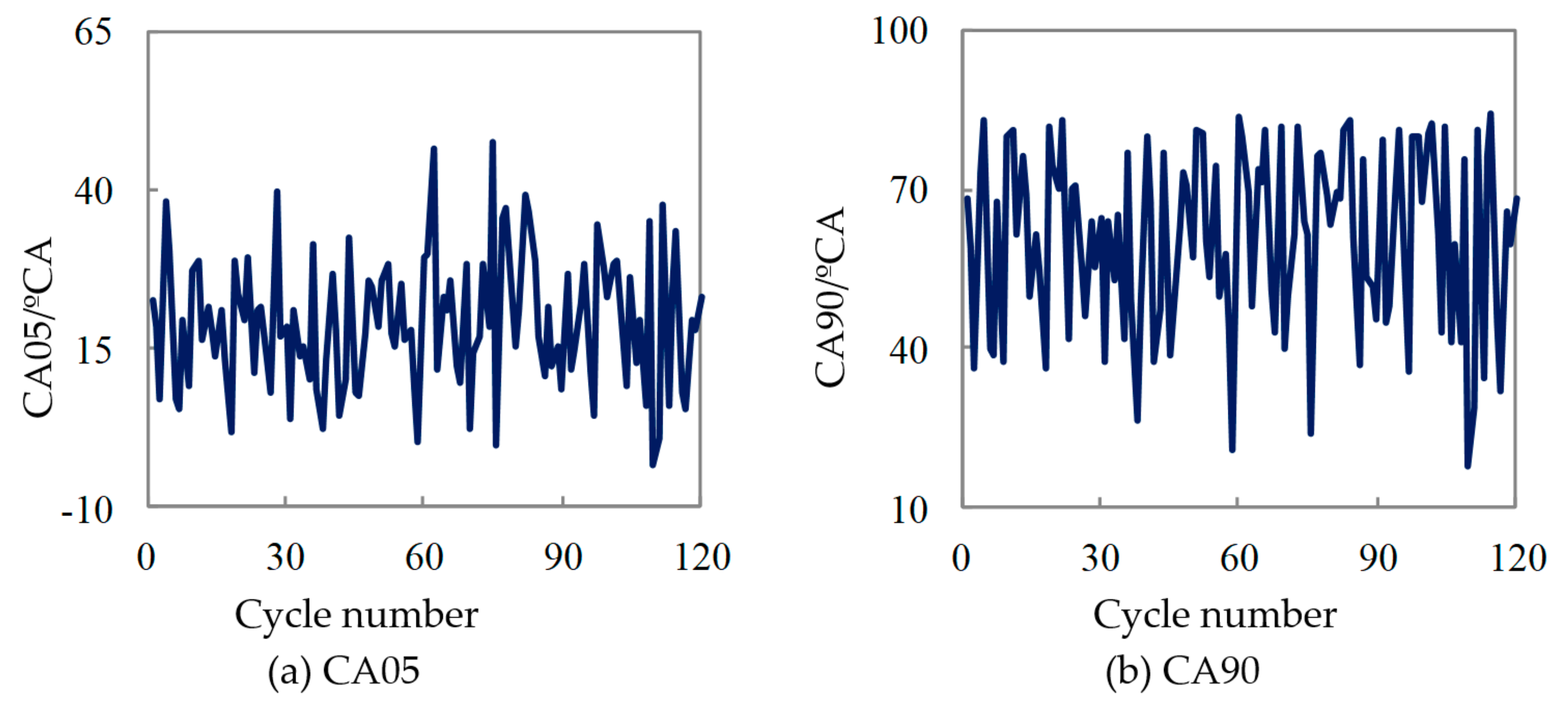
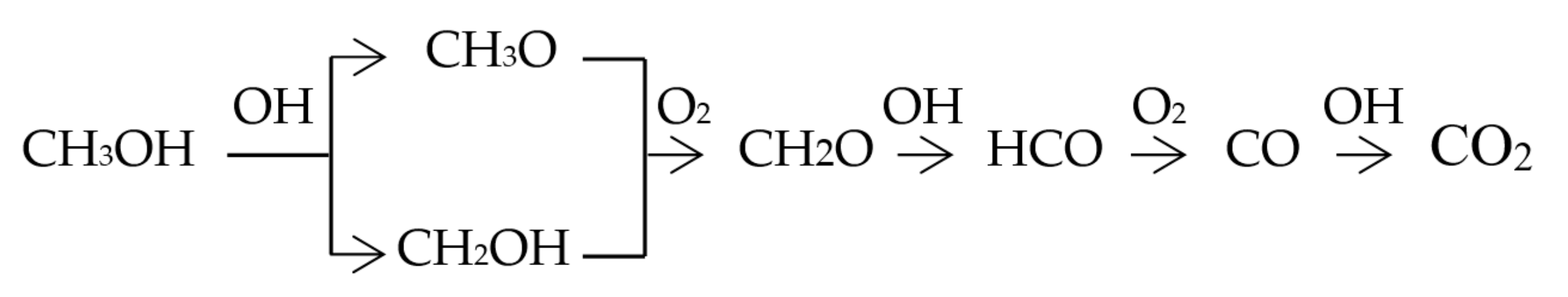
| Displacement/L | Cylinder Bore/mm | Stroke/mm | Compression Ratio |
|---|---|---|---|
| 0.001 | 11.25 | 10 | 8 |
| Parameters | Argument Value |
|---|---|
| Pressure range/bar | 0–250 |
| Limit value/bar | 300 |
| Natural frequency/kHz | 130 |
| Operating temperature range/°C | −50–400 |
| Linear error/%FSO | ≤±0.4 |
| Sensitivity/pC/bar | 16 |
| Thermal impact error/bar | ≤±0.5 |
| Impact-resistant/g | 2000 |
| Capacity/PF | 5 |
| Install threads | M5 × 0.5 |
| Fuel | Pmax/MPa | APmax/°CA | Pmi/MPa | CA05/°CA | CA10/°CA | CA50/°CA | CA90/°CA |
|---|---|---|---|---|---|---|---|
| methanol | 0.65 | 19.2 | 0.137 | 12.4 | 15.1 | 34.5 | 65.2 |
| Fuel | Pmax/MPa | APmax/°CA | Pmi/MPa | CA05/°CA | CA10/°CA | CA50/°CA | CA90/°CA |
|---|---|---|---|---|---|---|---|
| methanol/10% nitromethane | 0.767 | 22.6 | 0.182 | 8.5 | 11.5 | 29.7 | 58.2 |
| methanol/10% hydrogen peroxide solution | 0.569 | 1.0 | 0.078 | 24.7 | 27.8 | 50.2 | 78.5 |
| Ratio of Nitromethane/% | Pmax/MPa | APmax/°CA | Pmi/MPa | CA05/°CA | CA10/°CA | CA50/°CA | CA90/°CA |
|---|---|---|---|---|---|---|---|
| 0 | 0.646 | 19.2 | 0.137 | 12.4 | 15.1 | 34.5 | 65.2 |
| 5 | 0.687 | 20.4 | 0.153 | 11.1 | 13.8 | 32.7 | 61.7 |
| 10 | 0.767 | 22.6 | 0.182 | 8.5 | 11.5 | 29.7 | 58.2 |
| 15 | 0.874 | 21.4 | 0.193 | 5.3 | 8.0 | 24.3 | 50.2 |
| Ratio of Nitromethane/% | Pmax | Pmi | ||||
|---|---|---|---|---|---|---|
| σPmax/MPa | max/MPa | CoVPmax/% | σPmi/MPa | mi/MPa | CoVPmi/% | |
| 0% | 0.25 | 0.71 | 35.8 | 0.08 | 0.14 | 61.1 |
| 5% | 0.20 | 0.72 | 27.9 | 0.05 | 0.15 | 31.8 |
| 10% | 0.19 | 0.79 | 23.9 | 0.03 | 0.18 | 16.9 |
| 15% | 0.17 | 0.89 | 19.5 | 0.02 | 0.19 | 9.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, G.; Wang, S.; Zhang, L.; Shang, H. Diagnosis and Improvement of Combustion Characteristics of Methanol Miniature Reciprocating Piston Internal Combustion Engine. Micromachines 2020, 11, 96. https://doi.org/10.3390/mi11010096
Tang G, Wang S, Zhang L, Shang H. Diagnosis and Improvement of Combustion Characteristics of Methanol Miniature Reciprocating Piston Internal Combustion Engine. Micromachines. 2020; 11(1):96. https://doi.org/10.3390/mi11010096
Chicago/Turabian StyleTang, Gangzhi, Shuaibin Wang, Li Zhang, and Huichao Shang. 2020. "Diagnosis and Improvement of Combustion Characteristics of Methanol Miniature Reciprocating Piston Internal Combustion Engine" Micromachines 11, no. 1: 96. https://doi.org/10.3390/mi11010096
APA StyleTang, G., Wang, S., Zhang, L., & Shang, H. (2020). Diagnosis and Improvement of Combustion Characteristics of Methanol Miniature Reciprocating Piston Internal Combustion Engine. Micromachines, 11(1), 96. https://doi.org/10.3390/mi11010096





