Ultra-Processed Foods and Inflammatory Bowel Disease: A Narrative Review of Epidemiology, Mechanisms, and Dietary Implications
Highlights
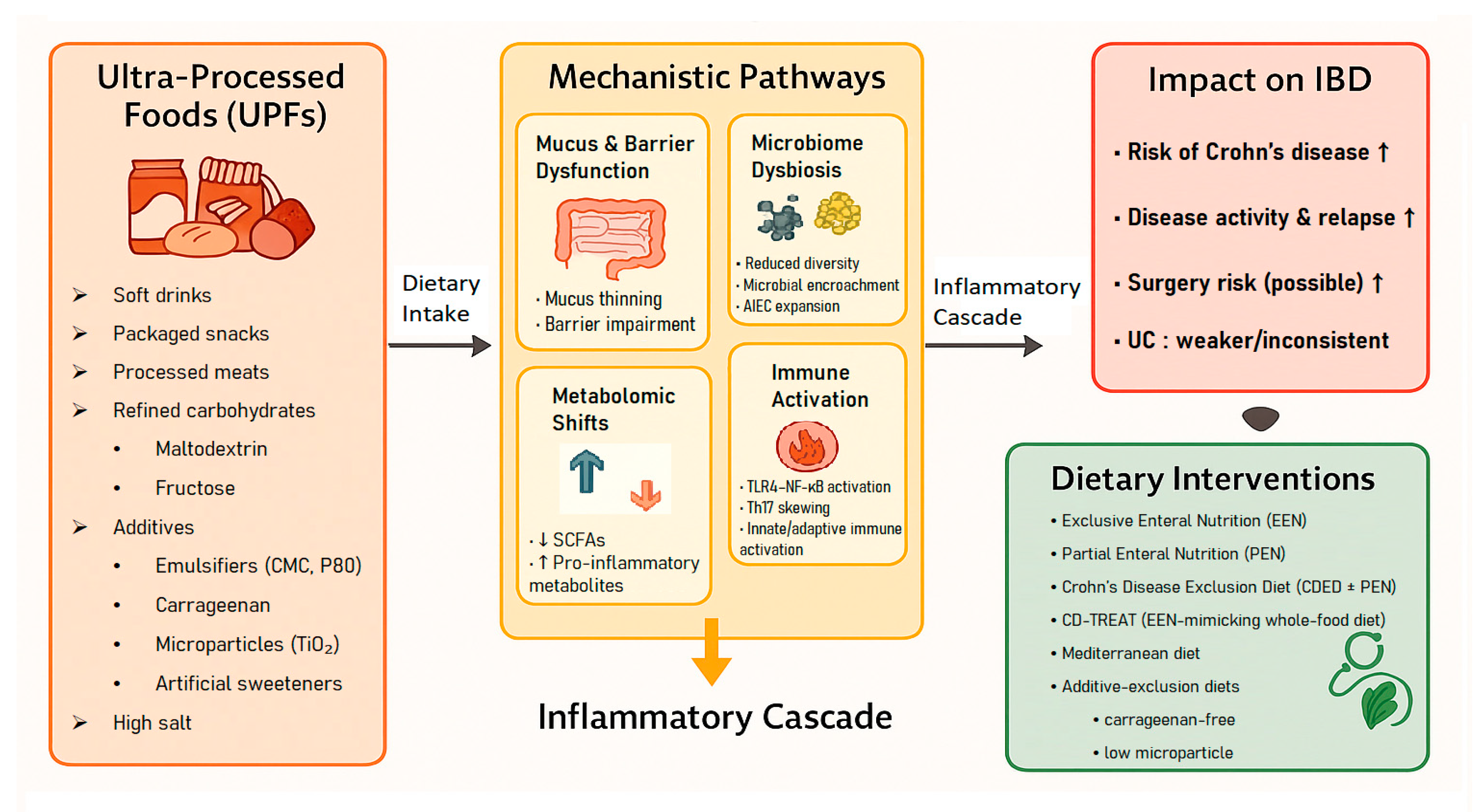
- A higher intake of ultra-processed foods (UPFs) was consistently associated with an increased risk of Crohn’s disease, while its association with ulcerative colitis was weaker or inconsistent.
- Specific components of UPFs (e.g., emulsifiers, carrageenan, maltodextrin, excess salt) can disrupt intestinal barrier integrity, alter gut microbiota, and activate immune pathways that promote intestinal inflammation.
- Dietary interventions that reduce UPF exposure—such as exclusive enteral nutrition (EEN) and the Crohn’s disease exclusion diet (CDED)—demonstrated clinical benefits, particularly in pediatric Crohn’s disease.
- The findings highlight the potential role of dietary processing and food additives as modifiable environmental factors contributing to the development and progression of inflammatory bowel disease (IBD).
- This study suggests that reducing the consumption of ultra-processed foods may serve as a practical dietary strategy for preventing or managing IBD, although further human clinical trials are required to establish causality and long-term effectiveness.
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Search Strategy
- Primary Search: We broadly searched for combinations of “ultra-processed foods,” “NOVA classification,” and “inflammatory bowel disease” (including “Crohn’s disease” and “ulcerative colitis”).
- Targeted Secondary Search: Based on initial findings, we performed targeted searches for specific UPF-associated components identified in the literature, such as “food additives,” “emulsifiers” (e.g., carboxymethylcellulose), “carrageenan,” and “maltodextrin.”
- Reference Screening: We manually screened the reference lists of retrieved articles and relevant systematic reviews to identify additional pivotal studies not captured by the initial electronic search.
2.3. Eligibility Criteria
2.4. Study Selection and Synthesis
3. Epidemiology
3.1. Prospective Cohorts
3.2. Patient Cohorts
3.3. Meta-Analyses
3.4. Complementary Evidence from Dietary Pattern Studies
3.5. Synthesis and Methodological Considerations
4. Mechanistic Pathways
4.1. Emulsifiers (Carboxymethylcellulose, Polysorbate-80)
4.2. Carrageenan (CGN)
4.3. Refined Carbohydrate Additives: Maltodextrin and Added Sugars
4.4. Non-Nutritive Sweeteners (NNS)
4.5. Microparticles and Colorants (Titanium Dioxide, E171)
4.6. High Salt as a UPF-Linked Property
4.7. Translational Gaps and Limitations
4.8. Integrated Mechanistic Synthesis
5. Therapeutic Implications
5.1. Exclusive Enteral Nutrition (EEN) and Partial Enteral Nutrition (PEN)
5.2. Crohn’s Disease Exclusion Diet (CDED)
5.3. Individualized Food-Based Diet (CD-TREAT)
5.4. Specific Carbohydrate Diet (SCD) and Mediterranean Diet (MedDiet)
5.5. Additive-Exclusion Approaches
5.6. Guideline and Consensus Perspectives
5.7. Therapeutic Synthesis
6. Conclusions
7. Future Directions
- Standardization of exposure: Development of harmonized definitions of UPFs and validated tools. including AI-assisted dietary monitoring and specific biomarkers of food processing, to enable cross-study comparability.
- Mechanism-focused trials: Well-designed randomized controlled trials targeting specific additives (e.g., emulsifiers, carrageenan) at physiologically relevant doses, utilizing endoscopic, histologic, and multi-omics outcomes to bridge the translational gap between animal models and human disease.
- Personalized nutrition strategies: Integration of individual microbiome characteristics, host genetics, and baseline immune signatures to tailor dietary interventions for specific IBD phenotypes.
- Long-term feasibility and safety: Rigorous evaluation of adherence, nutritional adequacy, cost-effectiveness, and real-world barriers to implementing UPF-restricted diets in diverse real-world clinical settings.
- Policy and clinical translation: Development of public health strategies for reducing population-level UPF intake, regulatory guidance for industry reformulation to minimize harmful additives, and clear pathways for integrating dietitian-led counseling into routine IBD care.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| IBD | Inflammatory bowel disease |
| CD | Crohn’s disease |
| UC | Ulcerative colitis |
| UPF | Ultra-processed food |
| NOVA | NOVA food classification system |
| FFQ | Food frequency questionnaire |
| HR | Hazard ratio |
| OR | Odds ratio |
| RR | Relative risk |
| SES | Socioeconomic status |
| SCFA | Short-chain fatty acid |
| AIEC | Adherent-invasive Escherichia coli |
| CMC | Carboxymethylcellulose |
| P80 | Polysorbate-80 |
| CGN | Carrageenan |
| MDX | Maltodextrin |
| NNS | Non-nutritive sweetener |
| IEC | Intestinal epithelial cell |
| TLR | Toll-like receptor |
| NF-κB | Nuclear factor kappa B |
| Th17 | T helper 17 cell |
| EFSA | European Food Safety Authority |
References
- Narula, N.; Wong, E.C.L.; Dehghan, M.; Mente, A.; Rangarajan, S.; Lanas, F.; Lopez-Jaramillo, P.; Rohatgi, P.; Lakshmi, P.V.M.; Varma, R.P.; et al. Association of ultra-processed food intake with risk of inflammatory bowel disease: Prospective cohort study. Bmj 2021, 374, n1554. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Moubarac, J.C.; Levy, R.B.; Louzada, M.L.C.; Jaime, P.C. The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018, 21, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.; Machado, P.; Santos, T.; Sievert, K.; Backholer, K.; Hadjikakou, M.; Russell, C.; Huse, O.; Bell, C.; Scrinis, G.; et al. Ultra-processed foods and the nutrition transition: Global, regional and national trends, food systems transformations and political economy drivers. Obes. Rev. 2020, 21, e13126. [Google Scholar] [CrossRef]
- Elizabeth, L.; Machado, P.; Zinöcker, M.; Baker, P.; Lawrence, M. Ultra-Processed Foods and Health Outcomes: A Narrative Review. Nutrients 2020, 12, 1955. [Google Scholar] [CrossRef] [PubMed]
- Srour, B.; Fezeu, L.K.; Kesse-Guyot, E.; Allès, B.; Méjean, C.; Andrianasolo, R.M.; Chazelas, E.; Deschasaux, M.; Hercberg, S.; Galan, P.; et al. Ultra-processed food intake and risk of cardiovascular disease: Prospective cohort study (NutriNet-Santé). Bmj 2019, 365, l1451. [Google Scholar] [CrossRef]
- Lo, C.H.; Khandpur, N.; Rossato, S.L.; Lochhead, P.; Lopes, E.W.; Burke, K.E.; Richter, J.M.; Song, M.; Ardisson Korat, A.V.; Sun, Q.; et al. Ultra-processed Foods and Risk of Crohn’s Disease and Ulcerative Colitis: A Prospective Cohort Study. Clin. Gastroenterol. Hepatol. 2022, 20, e1323–e1337. [Google Scholar] [CrossRef]
- Chen, J.; Wellens, J.; Kalla, R.; Fu, T.; Deng, M.; Zhang, H.; Yuan, S.; Wang, X.; Theodoratou, E.; Li, X.; et al. Intake of Ultra-processed Foods Is Associated with an Increased Risk of Crohn’s Disease: A Cross-sectional and Prospective Analysis of 187 154 Participants in the UK Biobank. J. Crohns Colitis 2023, 17, 535–552. [Google Scholar] [CrossRef] [PubMed]
- Sarbagili-Shabat, C.; Zelber-Sagi, S.; Isakov, N.F.; Hirsch, A.; Ron, Y.; Grinshpan, L.S.; Anbar, R.; Bromberg, A.; Thurm, T.; Maharshak, N. Ultra-Processed Foods Consumption Is Positively Associated with the Clinical Activity of Inflammatory Bowel Diseases: A Cross-Sectional Single-Center Study. Inflamm. Intest. Dis. 2024, 9, 241–251. [Google Scholar] [CrossRef]
- Sarbagili-Shabat, C.; Zelber-Sagi, S.; Fliss Isakov, N.; Hirsch, A.; Ron, Y.; Sol Grinshpan, L.; Aviv Cohen, N.; Leibovitzh, H.; Thurm, T.; Maharshak, N. High Ultra-Processed Food Consumption Is Associated with Clinical Exacerbation in Patients with Crohn’s Disease in Remission: A Prospective Cohort Study. Dig. Dis. 2025, 43, 466–475. [Google Scholar] [CrossRef]
- Chassaing, B.; Koren, O.; Goodrich, J.K.; Poole, A.C.; Srinivasan, S.; Ley, R.E.; Gewirtz, A.T. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 2015, 519, 92–96. [Google Scholar] [CrossRef]
- Bancil, A.S.; Sandall, A.M.; Rossi, M.; Chassaing, B.; Lindsay, J.O.; Whelan, K. Food Additive Emulsifiers and Their Impact on Gut Microbiome, Permeability, and Inflammation: Mechanistic Insights in Inflammatory Bowel Disease. J. Crohns Colitis 2021, 15, 1068–1079. [Google Scholar] [CrossRef] [PubMed]
- Vasseur, P.; Dugelay, E.; Benamouzig, R.; Savoye, G.; Lan, A.; Srour, B.; Hercberg, S.; Touvier, M.; Hugot, J.P.; Julia, C.; et al. Dietary Patterns, Ultra-processed Food, and the Risk of Inflammatory Bowel Diseases in the NutriNet-Santé Cohort. Inflamm. Bowel Dis. 2021, 27, 65–73. [Google Scholar] [CrossRef]
- Narula, N.; Chang, N.H.; Mohammad, D.; Wong, E.C.L.; Ananthakrishnan, A.N.; Chan, S.S.M.; Carbonnel, F.; Meyer, A. Food Processing and Risk of Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. 2023, 21, 2483–2495.e2481. [Google Scholar] [CrossRef]
- Amre, D.K.; D’Souza, S.; Morgan, K.; Seidman, G.; Lambrette, P.; Grimard, G.; Israel, D.; Mack, D.; Ghadirian, P.; Deslandres, C.; et al. Imbalances in dietary consumption of fatty acids, vegetables, and fruits are associated with risk for Crohn’s disease in children. Am. J. Gastroenterol. 2007, 102, 2016–2025. [Google Scholar] [CrossRef]
- D’Souza, S.; Levy, E.; Mack, D.; Israel, D.; Lambrette, P.; Ghadirian, P.; Deslandres, C.; Morgan, K.; Seidman, E.G.; Amre, D.K. Dietary patterns and risk for Crohn’s disease in children. Inflamm. Bowel Dis. 2008, 14, 367–373. [Google Scholar] [CrossRef]
- Sakamoto, N.; Kono, S.; Wakai, K.; Fukuda, Y.; Satomi, M.; Shimoyama, T.; Inaba, Y.; Miyake, Y.; Sasaki, S.; Okamoto, K.; et al. Dietary risk factors for inflammatory bowel disease: A multicenter case-control study in Japan. Inflamm. Bowel Dis. 2005, 11, 154–163. [Google Scholar] [CrossRef]
- Ananthakrishnan, A.N.; Khalili, H.; Konijeti, G.G.; Higuchi, L.M.; de Silva, P.; Korzenik, J.R.; Fuchs, C.S.; Willett, W.C.; Richter, J.M.; Chan, A.T. A prospective study of long-term intake of dietary fiber and risk of Crohn’s disease and ulcerative colitis. Gastroenterology 2013, 145, 970–977. [Google Scholar] [CrossRef]
- Christensen, C.; Knudsen, A.; Arnesen, E.K.; Hatlebakk, J.G.; Sletten, I.S.; Fadnes, L.T. Diet, Food, and Nutritional Exposures and Inflammatory Bowel Disease or Progression of Disease: An Umbrella Review. Adv. Nutr. 2024, 15, 100219. [Google Scholar] [CrossRef] [PubMed]
- Babaei, A.; Pourmotabbed, A.; Talebi, S.; Mehrabani, S.; Bagheri, R.; Ghoreishy, S.M.; Amirian, P.; Zarpoosh, M.; Mohammadi, H.; Kermani, M.A.H.; et al. The association of ultra-processed food consumption with adult inflammatory bowel disease risk: A systematic review and dose-response meta-analysis of 4,035,694 participants. Nutr. Rev. 2024, 82, 861–871. [Google Scholar] [CrossRef]
- Hou, J.K.; Abraham, B.; El-Serag, H. Dietary intake and risk of developing inflammatory bowel disease: A systematic review of the literature. Am. J. Gastroenterol. 2011, 106, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.; Phillipson, M.; Petersson, J.; Velcich, A.; Holm, L.; Hansson, G.C. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 15064–15069. [Google Scholar] [CrossRef] [PubMed]
- Wardle, R.A.; Wardle, A.J.; Charadva, C.; Ghosh, S.; Moran, G.W. Literature review: Impacts of socioeconomic status on the risk of inflammatory bowel disease and its outcomes. Eur. J. Gastroenterol. Hepatol. 2017, 29, 879–884. [Google Scholar] [CrossRef]
- Chassaing, B.; Compher, C.; Bonhomme, B.; Liu, Q.; Tian, Y.; Walters, W.; Nessel, L.; Delaroque, C.; Hao, F.; Gershuni, V.; et al. Randomized Controlled-Feeding Study of Dietary Emulsifier Carboxymethylcellulose Reveals Detrimental Impacts on the Gut Microbiota and Metabolome. Gastroenterology 2022, 162, 743–756. [Google Scholar] [CrossRef]
- Borthakur, A.; Bhattacharyya, S.; Dudeja, P.K.; Tobacman, J.K. Carrageenan induces interleukin-8 production through distinct Bcl10 pathway in normal human colonic epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G829–G838. [Google Scholar] [CrossRef]
- Tobacman, J.K. Review of harmful gastrointestinal effects of carrageenan in animal experiments. Environ. Health Perspect. 2001, 109, 983–994. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Gill, R.; Chen, M.L.; Zhang, F.; Linhardt, R.J.; Dudeja, P.K.; Tobacman, J.K. Toll-like receptor 4 mediates induction of the Bcl10-NFkappaB-interleukin-8 inflammatory pathway by carrageenan in human intestinal epithelial cells. J. Biol. Chem. 2008, 283, 10550–10558. [Google Scholar] [CrossRef]
- Borsani, B.; De Santis, R.; Perico, V.; Penagini, F.; Pendezza, E.; Dilillo, D.; Bosetti, A.; Zuccotti, G.V.; D’Auria, E. The Role of Carrageenan in Inflammatory Bowel Diseases and Allergic Reactions: Where Do We Stand? Nutrients 2021, 13, 3402. [Google Scholar] [CrossRef]
- Shang, Q.; Sun, W.; Shan, X.; Jiang, H.; Cai, C.; Hao, J.; Li, G.; Yu, G. Carrageenan-induced colitis is associated with decreased population of anti-inflammatory bacterium, Akkermansia muciniphila, in the gut microbiota of C57BL/6J mice. Toxicol. Lett. 2017, 279, 87–95. [Google Scholar] [CrossRef]
- Liu, F.; Hou, P.; Zhang, H.; Tang, Q.; Xue, C.; Li, R.W. Food-grade carrageenans and their implications in health and disease. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3918–3936. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Borthakur, A.; Dudeja, P.K.; Tobacman, J.K. Carrageenan induces cell cycle arrest in human intestinal epithelial cells in vitro. J. Nutr. 2008, 138, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Xue, L.; Devkota, S.; Chang, E.; Morris, S.; Tobacman, J.K. Carrageenan-induced colonic inflammation is reduced in Bcl10 null mice and increased in IL-10-deficient mice. Mediat. Inflamm. 2013, 2013, 397642. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Shumard, T.; Xie, H.; Dodda, A.; Varady, K.A.; Feferman, L.; Halline, A.G.; Goldstein, J.L.; Hanauer, S.B.; Tobacman, J.K. A randomized trial of the effects of the no-carrageenan diet on ulcerative colitis disease activity. Nutr. Healthy Aging 2017, 4, 181–192. [Google Scholar] [CrossRef]
- Nickerson, K.P.; McDonald, C. Crohn’s disease-associated adherent-invasive Escherichia coli adhesion is enhanced by exposure to the ubiquitous dietary polysaccharide maltodextrin. PLoS ONE 2012, 7, e52132. [Google Scholar] [CrossRef] [PubMed]
- Nickerson, K.P.; Chanin, R.; McDonald, C. Deregulation of intestinal anti-microbial defense by the dietary additive, maltodextrin. Gut Microbes 2015, 6, 78–83. [Google Scholar] [CrossRef]
- Laudisi, F.; Di Fusco, D.; Dinallo, V.; Stolfi, C.; Di Grazia, A.; Marafini, I.; Colantoni, A.; Ortenzi, A.; Alteri, C.; Guerrieri, F.; et al. The Food Additive Maltodextrin Promotes Endoplasmic Reticulum Stress-Driven Mucus Depletion and Exacerbates Intestinal Inflammation. Cell Mol. Gastroenterol. Hepatol. 2019, 7, 457–473. [Google Scholar] [CrossRef]
- Zangara, M.T.; Ponti, A.K.; Miller, N.D.; Engelhart, M.J.; Ahern, P.P.; Sangwan, N.; McDonald, C. Maltodextrin Consumption Impairs the Intestinal Mucus Barrier and Accelerates Colitis Through Direct Actions on the Epithelium. Front. Immunol. 2022, 13, 841188. [Google Scholar] [CrossRef] [PubMed]
- Montrose, D.C.; Nishiguchi, R.; Basu, S.; Staab, H.A.; Zhou, X.K.; Wang, H.; Meng, L.; Johncilla, M.; Cubillos-Ruiz, J.R.; Morales, D.K.; et al. Dietary Fructose Alters the Composition, Localization, and Metabolism of Gut Microbiota in Association With Worsening Colitis. Cell Mol. Gastroenterol. Hepatol. 2021, 11, 525–550. [Google Scholar] [CrossRef]
- Nishiguchi, R.; Basu, S.; Staab, H.A.; Ito, N.; Zhou, X.K.; Wang, H.; Ha, T.; Johncilla, M.; Yantiss, R.K.; Montrose, D.C.; et al. Dietary interventions to prevent high-fructose diet-associated worsening of colitis and colitis-associated tumorigenesis in mice. Carcinogenesis 2021, 42, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Cohen, Y.; Valdés-Mas, R.; Mor, U.; Dori-Bachash, M.; Federici, S.; Zmora, N.; Leshem, A.; Heinemann, M.; Linevsky, R.; et al. Personalized microbiome-driven effects of non-nutritive sweeteners on human glucose tolerance. Cell 2022, 185, 3307–3328.e3319. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Korem, T.; Zeevi, D.; Zilberman-Schapira, G.; Thaiss, C.A.; Maza, O.; Israeli, D.; Zmora, N.; Gilad, S.; Weinberger, A.; et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 2014, 514, 181–186. [Google Scholar] [CrossRef]
- Bettini, S.; Boutet-Robinet, E.; Cartier, C.; Coméra, C.; Gaultier, E.; Dupuy, J.; Naud, N.; Taché, S.; Grysan, P.; Reguer, S.; et al. Food-grade TiO2 impairs intestinal and systemic immune homeostasis, initiates preneoplastic lesions and promotes aberrant crypt development in the rat colon. Sci. Rep. 2017, 7, 40373. [Google Scholar] [CrossRef]
- Lomer, M.C.; Hutchinson, C.; Volkert, S.; Greenfield, S.M.; Catterall, A.; Thompson, R.P.; Powell, J.J. Dietary sources of inorganic microparticles and their intake in healthy subjects and patients with Crohn’s disease. Br. J. Nutr. 2004, 92, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, N.S.; de Kok, T.M.; Sijm, D.; van Breda, S.G.; Briedé, J.J.; Castenmiller, J.J.M.; Opperhuizen, A.; Chirino, Y.I.; Dirven, H.; Gott, D.; et al. Possible Adverse Effects of Food Additive E171 (Titanium Dioxide) Related to Particle Specific Human Toxicity, Including the Immune System. Int. J. Mol. Sci. 2020, 22, 207. [Google Scholar] [CrossRef] [PubMed]
- Lomer, M.C.; Harvey, R.S.; Evans, S.M.; Thompson, R.P.; Powell, J.J. Efficacy and tolerability of a low microparticle diet in a double blind, randomized, pilot study in Crohn’s disease. Eur. J. Gastroenterol. Hepatol. 2001, 13, 101–106. [Google Scholar] [CrossRef]
- Younes, M.; Aquilina, G.; Castle, L.; Engel, K.H.; Fowler, P.; Frutos Fernandez, M.J.; Fürst, P.; Gundert-Remy, U.; Gürtler, R.; Husøy, T.; et al. Safety assessment of titanium dioxide (E171) as a food additive. Efsa J. 2021, 19, e06585. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EU) 2022/63 of 14 January 2022 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council as regards the food additive titanium dioxide (E171). Off. J. Eur. Union 2022, L11, 1–5. Available online: https://eur-lex.europa.eu/eli/reg/2022/63/oj (accessed on 5 December 2025).
- Tubbs, A.L.; Liu, B.; Rogers, T.D.; Sartor, R.B.; Miao, E.A. Dietary Salt Exacerbates Experimental Colitis. J. Immunol. 2017, 199, 1051–1059. [Google Scholar] [CrossRef]
- Miranda, P.M.; De Palma, G.; Serkis, V.; Lu, J.; Louis-Auguste, M.P.; McCarville, J.L.; Verdu, E.F.; Collins, S.M.; Bercik, P. High salt diet exacerbates colitis in mice by decreasing Lactobacillus levels and butyrate production. Microbiome 2018, 6, 57. [Google Scholar] [CrossRef]
- Kleinewietfeld, M.; Manzel, A.; Titze, J.; Kvakan, H.; Yosef, N.; Linker, R.A.; Muller, D.N.; Hafler, D.A. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 2013, 496, 518–522. [Google Scholar] [CrossRef]
- Wei, Y.; Lu, C.; Chen, J.; Cui, G.; Wang, L.; Yu, T.; Yang, Y.; Wu, W.; Ding, Y.; Li, L.; et al. High salt diet stimulates gut Th17 response and exacerbates TNBS-induced colitis in mice. Oncotarget 2017, 8, 70–82. [Google Scholar] [CrossRef]
- Ruemmele, F.M.; Veres, G.; Kolho, K.L.; Griffiths, A.; Levine, A.; Escher, J.C.; Amil Dias, J.; Barabino, A.; Braegger, C.P.; Bronsky, J.; et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J. Crohns Colitis 2014, 8, 1179–1207. [Google Scholar] [CrossRef]
- Levine, A.; Sigall Boneh, R.; Wine, E. Evolving role of diet in the pathogenesis and treatment of inflammatory bowel diseases. Gut 2018, 67, 1726–1738. [Google Scholar] [CrossRef]
- Diederen, K.; Li, J.V.; Donachie, G.E.; de Meij, T.G.; de Waart, D.R.; Hakvoort, T.B.M.; Kindermann, A.; Wagner, J.; Auyeung, V.; Te Velde, A.A.; et al. Exclusive enteral nutrition mediates gut microbial and metabolic changes that are associated with remission in children with Crohn’s disease. Sci. Rep. 2020, 10, 18879. [Google Scholar] [CrossRef] [PubMed]
- Wall, C.L.; Gearry, R.B.; Day, A.S. Treatment of Active Crohn’s Disease with Exclusive and Partial Enteral Nutrition: A Pilot Study in Adults. Inflamm. Intest. Dis. 2018, 2, 219–227. [Google Scholar] [CrossRef]
- Nardone, O.M.; Calabrese, G.; La Mantia, A.; Testa, A.; Rispo, A.; Alfonsi, L.; Pasanisi, F.; Castiglione, F. Effectiveness of Partial Enteral Nutrition as Add-On to Biologics in Patients With Refractory and Difficult-to-Treat Crohn’s Disease: A Pilot Study. Crohns Colitis 360 2024, 6, otae011. [Google Scholar] [CrossRef]
- Levine, A.; Wine, E.; Assa, A.; Sigall Boneh, R.; Shaoul, R.; Kori, M.; Cohen, S.; Peleg, S.; Shamaly, H.; On, A.; et al. Crohn’s Disease Exclusion Diet Plus Partial Enteral Nutrition Induces Sustained Remission in a Randomized Controlled Trial. Gastroenterology 2019, 157, 440–450.e448. [Google Scholar] [CrossRef]
- Sigall Boneh, R.; Sarbagili Shabat, C.; Yanai, H.; Chermesh, I.; Ben Avraham, S.; Boaz, M.; Levine, A. Dietary Therapy With the Crohn’s Disease Exclusion Diet is a Successful Strategy for Induction of Remission in Children and Adults Failing Biological Therapy. J. Crohns Colitis 2017, 11, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Svolos, V.; Hansen, R.; Nichols, B.; Quince, C.; Ijaz, U.Z.; Papadopoulou, R.T.; Edwards, C.A.; Watson, D.; Alghamdi, A.; Brejnrod, A.; et al. Treatment of Active Crohn’s Disease With an Ordinary Food-based Diet That Replicates Exclusive Enteral Nutrition. Gastroenterology 2019, 156, 1354–1367.e1356. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.A.; Gold, B.D.; Oliva, S.; Lewis, J.; Stallworth, A.; Koch, B.; Eshee, L.; Mason, D. Clinical and mucosal improvement with specific carbohydrate diet in pediatric Crohn disease. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 516–521. [Google Scholar] [CrossRef]
- Lewis, J.D.; Sandler, R.S.; Brotherton, C.; Brensinger, C.; Li, H.; Kappelman, M.D.; Daniel, S.G.; Bittinger, K.; Albenberg, L.; Valentine, J.F.; et al. A Randomized Trial Comparing the Specific Carbohydrate Diet to a Mediterranean Diet in Adults With Crohn’s Disease. Gastroenterology 2021, 161, 837–852.e839. [Google Scholar] [CrossRef]
- Hashash, J.G.; Elkins, J.; Lewis, J.D.; Binion, D.G. AGA Clinical Practice Update on Diet and Nutritional Therapies in Patients With Inflammatory Bowel Disease: Expert Review. Gastroenterology 2024, 166, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Sigall-Boneh, R.; Levine, A.; Lomer, M.; Wierdsma, N.; Allan, P.; Fiorino, G.; Gatti, S.; Jonkers, D.; Kierkus, J.; Katsanos, K.H.; et al. Research Gaps in Diet and Nutrition in Inflammatory Bowel Disease. A Topical Review by D-ECCO Working Group [Dietitians of ECCO]. J. Crohns Colitis 2017, 11, 1407–1419. [Google Scholar] [CrossRef] [PubMed]
- Svolos, V.; Gordon, H.; Lomer, M.C.E.; Aloi, M.; Bancil, A.; Day, A.S.; Day, A.S.; Fitzpatrick, J.A.; Gerasimidis, K.; Gkikas, K.; et al. European Crohn’s and Colitis Organisation consensus on dietary management of inflammatory bowel disease. J. Crohn’s Colitis 2025, 19, jjaf122. [Google Scholar] [CrossRef]
- Bischoff, S.C.; Bager, P.; Escher, J.; Forbes, A.; Hébuterne, X.; Hvas, C.L.; Joly, F.; Klek, S.; Krznaric, Z.; Ockenga, J.; et al. ESPEN guideline on Clinical Nutrition in inflammatory bowel disease. Clin. Nutr. 2023, 42, 352–379. [Google Scholar] [CrossRef] [PubMed]
| Study/ Country | Sample Size/Follow-Up | Exposure Assessment | UPF Definition | Outcome | Key Findings |
|---|---|---|---|---|---|
| PURE [1] 21 countries | 116,087/ median 9.7 years | Baseline FFQ | NOVA-defined UPF (servings/day) | Incident IBD | Consumption of ≥5 UPF servings/day was associated with a higher risk of incident IBD (HR 1.82; CD and UC combined). The strongest associations were observed for soft drinks, refined sweetened foods, salty snacks, and processed meats. |
| NHS I, NHS II, HPFS [6] USA | 245,112/ 5,468,444 person-years | Repeated FFQs every 2–4 years | NOVA-defined UPF | CD, UC | Higher UPF intake was associated with an increased risk of CD in a dose–response manner. No consistent association was observed between UPF intake and UC. |
| NutriNet-Santé [12] France | 105,832/ mean 2.3 years | Repeated web-based 24 h dietary records | Dietary UPF proportion (NOVA) | Incident IBD | No significant association was observed between UPF consumption and incident IBD. The authors noted a limited number of cases and relatively short follow-up duration. |
| UK Biobank [7] UK | 187,854/ median 9.84 years | Web-based 24 h dietary recall questionnaires | NOVA-defined UPF intake | CD, UC; IBD-related surgery | Higher UPF consumption was associated with an increased risk of CD (HR 2.00 for highest vs. lowest intake), but not UC. Among individuals with IBD, higher UPF intake was also associated with an increased risk of IBD-related surgery. |
| Study/ Country | Population | Design | UPF Exposure | Outcome | Key Findings |
|---|---|---|---|---|---|
| Sarbagili-Shabat 2024 [8] Israel | 242 IBD patients | Cross- sectional | NOVA-defined UPF intake | Disease activity | Higher UPF intake was associated with active disease; minimally processed foods showed protective associations. |
| Sarbagili-Shabat 2025 [9] Israel | 111 CD patients in remission | Prospective (1 year) | UPF intake tertiles (NOVA) | Clinical relapse | Higher UPF intake was associated with increased risk of relapse. |
| UK Biobank IBD subgroup [7] UK | Individuals with IBD | Prospective | NOVA-defined UPF intake | IBD-related surgery | Higher UPF intake was associated with increased risk of IBD-related surgery. |
| Christensen et al. [18] Multinational | IBD populations | Umbrella review | Various dietary exposures | Disease activity and progression | Processed food–rich diets were associated with worse outcomes, whereas whole-food–based patterns were protective. |
| Component/ Additive | Model | Dose & Exposure | Mechanistic Pathway | Key Findings | References |
|---|---|---|---|---|---|
| Emulsifiers (CMC, P80) | Mouse; human RCT | Mouse: 1.0% (w/v) in water, 12 weeks Human: CMC 15 g/day, 11 days | Mucus thinning; Microbial encroachment | Induces low-grade inflammation in WT mice; Human RCT showed microbiota depletion and reduced SCFAs. | [10,23] |
| Carrageenan (CGN) | Human IECs; mouse; RCT in UC | Cell: 1–10 µg/mL Human: 200 mg/day capsule | TLR4–Bcl10–NF-κB activation | Promoted epithelial inflammation in experimental models; accelerated clinical relapse in UC patients in remission. | [24,26,30,31,32] |
| Maltodextrin (MDX) | IECs; macrophages; IL-10−/− mice | Mouse: 5% (w/v) in water | Biofilm formation; AIEC adhesion; impaired antimicrobial defense | Enhanced AIEC expansion, disrupted mucosal defense, and exacerbated colitis severity. | [33,34,35,36] |
| Fructose (high-fructose diets) | Mouse | High-fructose diet (e.g., 60%) | Dysbiosis; Barrier disruption | Worsened colitis; effects were reversible after dietary normalization or psyllium supplementation. | [37,38] |
| Titanium dioxide (E171) | Mouse; in vitro; pilot RCT | Mouse: 10 mg/kg body weight/day Human: Dietary exclusion | Microparticle uptake; Biofilm modulation; immune activation | Induced preneoplastic and inflammatory changes in animal models; pilot RCT showed reduced inflammation with a microparticle-free diet; safety concerns highlighted in regulatory assessment. | [41,43,44,45] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, S.Y.; Moon, W. Ultra-Processed Foods and Inflammatory Bowel Disease: A Narrative Review of Epidemiology, Mechanisms, and Dietary Implications. Nutrients 2025, 17, 3852. https://doi.org/10.3390/nu17243852
Choi SY, Moon W. Ultra-Processed Foods and Inflammatory Bowel Disease: A Narrative Review of Epidemiology, Mechanisms, and Dietary Implications. Nutrients. 2025; 17(24):3852. https://doi.org/10.3390/nu17243852
Chicago/Turabian StyleChoi, So Yoon, and Won Moon. 2025. "Ultra-Processed Foods and Inflammatory Bowel Disease: A Narrative Review of Epidemiology, Mechanisms, and Dietary Implications" Nutrients 17, no. 24: 3852. https://doi.org/10.3390/nu17243852
APA StyleChoi, S. Y., & Moon, W. (2025). Ultra-Processed Foods and Inflammatory Bowel Disease: A Narrative Review of Epidemiology, Mechanisms, and Dietary Implications. Nutrients, 17(24), 3852. https://doi.org/10.3390/nu17243852







