Abstract
Infant feeding guidelines provide evidence-based recommendations to support optimal infant health, growth, and development, and exploring adherence to guidelines is a useful way of assessing diet quality. The aim of this study was to determine adherence to the recently updated Ministry of Health “Healthy Eating Guidelines for New Zealand Babies and Toddlers (0–2 years old)”. Data were obtained from First Foods New Zealand, a multicentre observational study of 625 infants aged 7.0–10.0 months. Caregivers completed two 24-h diet recalls and a demographic and feeding questionnaire. Nearly all caregivers (97.9%) initiated breastfeeding, 37.8% exclusively breastfed to around six months of age, and 66.2% were currently breastfeeding (mean age 8.4 months). Most caregivers met recommendations for solid food introduction, including appropriate age (75.4%), iron-rich foods (88.3%), puréed textures (80.3%), and spoon-feeding (74.1%). Infants consumed vegetables (63.2%) and fruit (53.9%) more frequently than grain foods (49.5%), milk and milk products (38.6%), and meat and protein-rich foods (31.8%). Most caregivers avoided inappropriate beverages (93.9%) and adding salt (76.5%) and sugar (90.6%). Our findings indicated that while most infants met the recommendations for the introduction of appropriate solid foods, the prevalence of exclusive breastfeeding could be improved, indicating that New Zealand families may need more support.
Keywords:
infant; feeding guidelines; adherence; breastfeeding; complementary feeding; food group; salt; sugar; beverages 1. Introduction
Rapid dietary changes are observed as an infant transitions from an exclusive milk diet to one that resembles that of the family [1,2,3,4]. To support those involved in the care of infants, guidelines have been developed to provide evidence-based nutrition recommendations. Guidelines provide population health advice on meeting nutrition requirements for growth, development, and the establishment of healthy behaviours [5]. Most infant feeding guidelines include recommendations for breastfeeding, complementary feeding, dietary variety (food groups), and appropriate foods and beverages.
In New Zealand (NZ), the Ministry of Health (MoH) infant feeding guidelines “Healthy Eating Guidelines for New Zealand Babies and Toddlers (0 to 2 years old)” were recently revised and updated to include six statements on breastfeeding and formula feeding, complementary feeding, food groups, appropriate foods and beverages, and feeding environments [5]. These guidelines are available to NZ whānau online and are promoted by Whānau Āwhina Plunket, which provides Well Child services to approximately 85% of NZ infants and their whānau [6]. Mothers are recommended to exclusively breastfeed (EBF) their infant until they are around six months of age, in accordance with the World Health Organization’s (WHO) recommendations [7]. Exclusive breastfeeding has benefits for both mothers and their infants, including the provision of all of the energy and nutrients that an infant needs; it also supports infant gastrointestinal development, reduces the risk of infant mortality, supports mother–infant bonding, and reduces the risk of maternal breast and ovarian cancer [7]. At six months of age, breast milk is not able to provide adequate energy and nutrients, and complementary feeding should be initiated [5]. Iron-rich foods (including meat, poultry, fish, seafood, and iron-fortified infant cereals), vegetables, and fruits are suitable first foods and should each be offered daily once complementary feeding has started. Infants should be offered a wide variety of foods, especially during the first weeks and months, as this helps to establish healthy taste preferences. There is no need to introduce new foods one at a time unless the food is a common allergen. A variety of foods from the main food groups—vegetables and fruit; grain foods; milk and milk products; and legumes, nut butters, eggs, fish, seafood and chicken or lean red meat (referred to as meat and protein-rich foods from here)—should be included in an infant’s diet every day, increasing the amount consumed and food texture as developmentally appropriate [5,8]. Inappropriate foods and beverages, such as foods with added sugar and salt (e.g., confectionery, biscuits, ice cream, processed meats, and fast foods) and beverages other than breast milk, infant formula, and water (such as juice, cordial, fruit drink, flavoured milk, soft drinks, tea, coffee, and alcohol) should be avoided [5].
Investigating adherence to feeding guidelines is a common method of measuring diet quality. Golley et al. [9] were the first to develop and utilise an index that measured adherence to infant feeding guidelines in a high-income country (England). Growing Up in New Zealand (GUiNZ), a longitudinal study of 6470 ethnically diverse infants, measured adherence to the 2008 MoH infant feeding guidelines [10]. The GUiNZ study collected data on infant feeding at nine months of age between 2009 and 2011 and found low levels of adherence (defined as less than 50%) to breastfeeding recommendations [10,11]. Exclusive breastfeeding to around six months of age was met by 35%, while 37% continued to breastfeed to 12 months or beyond. Similar relatively low adherence to EBF recommendations has been observed in other high-income countries [12,13,14,15,16], indicating a global issue. In some studies, low adherence to EBF recommendations can be explained by an early introduction of solid foods [9,10,17], with recommendations for EBF and introducing solid foods often overlapping. In NZ and Australia, recent studies have suggested that the age of introduction of solid foods has increased to align with recommendations [13,18]. The impact this has on EBF adherence is yet to be explored.
Studies have consistently shown high adherence levels for exposure to iron-rich foods, even though assessment methods have varied. In GUiNZ, high levels (80% of infants aged nine months of age) of adherence were found for offering iron-rich foods daily [10], while other studies have reported adherence levels for starting iron-rich foods of 89% and 87% at six [9] and seven [17] months, respectively. By contrast, adherence to beverage recommendations appears to be more variable, depending on what beverages are reported [9,10,17]. In GUiNZ, a moderate (defined as 50–80%) adherence to avoiding inappropriate drinks (61%) was reported at nine months of age [10], with juice reported as the most commonly consumed inappropriate beverage [11,19,20].
Adherence to food group recommendations is typically assessed by the consumption of vegetables, fruit, grain foods, milk and milk products, and meat and protein-rich foods, with varying levels of adherence being found between food groups [9,10,17]. The GUiNZ study found high levels of adherence for the daily consumption of vegetables (91%), fruit (87%), and grain foods (90%) in infants [10,11]. The study also found high adherence levels for the consumption of breast milk or infant formula (96%), although it did not report all milk and milk products [10,11]. Meat and protein-rich foods were less frequently consumed daily (61%). In other high-income countries, similar trends have been observed [9,21]. Although different dietary assessment methods have been used, meat and protein-rich foods are often reported as the least-frequently-consumed food group by infants [9,21]. However, the intake of vegetables and fruits appears to be lower than that reported in NZ [9,17,21]. Mixed results have been found for the offering of foods with added salt and sugar to infants, with high adherence levels for avoiding added sugar (86%) and salt (84%) in GUiNZ [10] but low adherence in the United States of America (USA) [17].
Various sociodemographic characteristics have been associated with adherence to infant feeding guidelines. In GUiNZ, mothers of European ethnicity, of older age, with a higher education, who attended antenatal classes, and who had a partner, as well as those living in the least deprived neighbourhoods, were more likely to meet recommendations [10]. Similar trends have been observed in the USA [22], the United Kingdom (UK) [23,24], Ireland [25], and other European countries [26]. Maternal adherence to adult feeding guidelines [3,21,24] and having fewer children [24,27,28] have also been associated with greater adherence to infant feeding guidelines.
In recent years, there have been notable changes in how infants are fed. Examples of these changes include the increasing availability of commercial infant foods [29,30] and trends surrounding baby-led weaning (BLW) [31,32], where an infant is given whole pieces of food from the start of complementary feeding [18,33]. The MoH does not currently recommend BLW in New Zealand, as more research is required to determine potential risks [5]. The practice also conflicts with recommendations for spoon-feeding puréed foods when starting complementary feeding. The advent of these approaches to infant feeding may have influenced adherence to other guidelines as well.
This study aimed to investigate adherence to the key indicators from the 2021 “Healthy Eating Guidelines for New Zealand Babies and Toddlers (0 to 2 years old)” in infants. Associations between sociodemographic characteristics and adherence to the guidelines were also explored.
2. Materials and Methods
2.1. Study Design
Data for the current study were obtained as part of the First Foods New Zealand (FFNZ) observational study. A detailed description of FFNZ’s methods can be found elsewhere [34]. FFNZ recruited 625 infants from the Auckland and Dunedin regions of NZ using word of mouth and Facebook advertisements. To ensure sufficient representation of Māori and Pacific infants, advertisements were promoted in Māori and Pasifika people’s community groups. Data were collected between July 2020 and February 2022. Ethical approval was obtained from the Health and Disability Ethics Committee New Zealand (19/STH/151), and written consent was obtained from parent participants before data collection. The study was registered with the Australian New Zealand Clinical Trials Registry (www.anzctr.org.au, accessed on 29 October 2023, registration number: ACTRN12620000459921).
2.2. Participants
To meet eligibility criteria, infants were required to live in the Auckland or Dunedin regions of NZ, be 7.0 to 10.0 months of age at the time of participation, and not have taken part in a nutrition intervention study that might have influenced their diet. Caregivers were required to be 16 years or older and able to communicate in English. The screening questionnaire was available on the FFNZ website and generated 1424 responses. Of those, 1198 were eligible, and 630 provided written consent. Five participants did not meet the eligibility criteria, leaving a final sample of 625 infants. Questionnaire data were available from all infants, and two days of diet recall data were available from 614 infants, with the remaining 11 infants providing one day of dietary data.
2.3. Data Collection
Participants were invited to attend two study appointments at their home or closest research centre approximately one week apart. Due to COVID-19 restrictions, some second appointments were conducted online. At the first appointment, participants completed a questionnaire investigating demographics, infant health, breastfeeding, and complementary feeding practices. Participants who reported two or more ethnic groups were assigned to a single group using the MoH prioritisation system (order of ranking: Māori, Pacific, Asian, Others, European) [35]. Socioeconomic deprivation levels were assigned according to the NZ Index of Deprivation, which uses the participant’s home address [36] and is displayed as deciles, with low deprivation being classified as deciles 1–3 and high deprivation as deciles 8–10. At each of the two appointments, caregivers completed a 24-h diet recall using a multiple-pass method. Caregivers recalled all breastfeeds, foods, and beverages offered, and the amount consumed the day before their appointment. To aid in portion size estimation, caregivers were asked to take photographs of foods and drinks when they were offered. Measurement prompts (grid sheets (square and circle), measuring cups and spoons, exemplar infant food pouches and jars, infant bowls, and thickness sticks) were also available to guide portion size estimation. Open questions were administered to determine the addition of salt and sugar to infant foods. If infants attended an early child education centre (ECE) or were cared for by someone other than their main caregiver, a food diary with written prompts was provided. Telephone or email contact was made, with caregiver consent, if additional details were required. After their second appointment, caregivers were emailed a final questionnaire, which included the Paediatric Eating Assessment Tool (PediEAT) [37]. Responses for “eats a variety of foods (fruits, vegetables, proteins, etc.) (always, almost always, often, sometimes, almost never, and never)” were used to describe caregiver perceptions of diet variety.
2.4. Adherence to National Infant Feeding Guidelines
Indicators presented in this paper were based on the “Healthy Eating Guidelines for New Zealand Babies and Toddlers (0 to 2 years old)” [5]. Recommendations that were measurable from FFNZ data and applied to those aged 7.0 to 10.0 months were selected (Table 1). Mothers reported breastfeeding initiation rates and when breastfeeding was stopped or if they continued to breastfeed. Exclusive breastfeeding to “around six months” [5] was defined as “5 months” or “6 months” (from a pull-down menu of months) being the age when something other than breast milk, i.e., either another drink, or solid foods, was first introduced. Due to the age of infants recruited, breastfeeding to two years of age was not assessed. Instead, current breastfeeding status at the time of participation (7.0 to 10.0 months of age) was reported. Food variety, or the consumption of diverse food groups, was determined by whether each food group was included at least once per day in both diet recalls, in accordance with the MoH recommendation for daily consumption of each food group [5].

Table 1.
Indicators developed from the Ministry of Health infant feeding guidelines [5].
2.5. Food Coding
All foods consumed in diet recalls (n = 20,975) were individually coded into food groups, using the MoH infant and adult guidelines as a guide (Table 2). The food groups were as follows: vegetables, fruit, grain foods, milk and milk products, and meat and protein-rich foods. Ingredients in recipes or mixed dishes (e.g., baby food pouches) containing multiple food groups were individually assigned to a food group, regardless of the amount consumed. Foods were only assigned to a food group if they complied with the guidelines. This meant that foods such as sour cream and fruit juice, for example, were not considered to contribute to intake from the “milk and milk products” and “fruit” food groups, respectively. Iron-rich foods included meat, poultry, fish, seafood, and iron-fortified infant cereals, as per MoH guidelines [5].

Table 2.
Foods included and excluded in food group analyses.
2.6. Statistical Analysis
Variables were reported as percentages and means (standard deviation (SD)) from the study population. The proportions of the sample that met the indicator were also adjusted by weighting for ethnicity and socioeconomic deprivation to represent the New Zealand population more closely [38] and estimate adherence with a 95% logit-transformed confidence interval. Logistic regression was used to estimate odds ratios, 95% confidence intervals, and p-values for associations between sociodemographic characteristics and adherence to different indicators, and p-values < 0.05 indicated statistical significance. Odds ratios were only calculated for variables with at least 10 participants in each cell and indicators with more than 10% of the sample in each category. Associations between sociodemographic variables (caregiver age, education, employment status; maternal parity; number of children living in household; use of childcare/ECE; and socioeconomic deprivation) and MoH recommendations (exclusive breastfeeding, current breastfeeding, solids introduction between 5 and 7 months, puréed foods introduced first, iron-rich foods introduced first, food variety (iron-rich foods, vegetables, and fruit consumed on both recall days), no salt or sugar added, and infants not fully spoon-fed at current age) were investigated. Analyses were performed using Stata Statistical Software (version 17, StataCorp LP, College Station, TX, USA) and Microsoft Excel (version 16.66).
3. Results
3.1. Maternal and Infant Characteristics
Maternal and infant demographic characteristics are summarised in Table 3. Infants and caregivers were on average (mean (SD)) 8.4 (0.8) months and 32.7 (4.9) years of age, respectively. The majority (98%) of caregivers were the infant’s mother. Six respondents were fathers, one was a grandparent, and one was a guardian.

Table 3.
Demographic characteristics of infants and caregivers (n = 625).
3.2. Adherence to Infant Feeding Guidelines
Adherence to the MoH infant feeding guidelines (Table 1) is shown in Table 4, with further detail regarding each indicator and sociodemographic predictors provided below. The proportions of the sample that met the indicator were also weighted for ethnicity and socioeconomic deprivation in Table 4 to more closely represent the New Zealand population [38], allowing wider conclusions to be made.

Table 4.
Summary of adherence to indicators developed from the Ministry of Health infant feeding guidelines (n = 625).
3.2.1. Breastfeeding
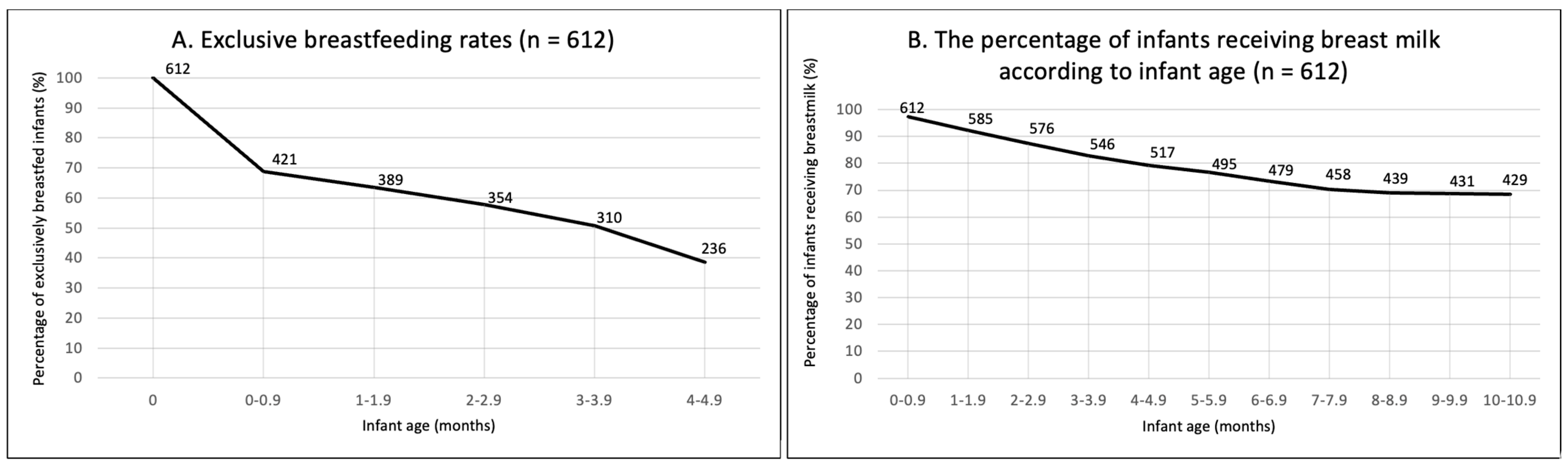
Breastfeeding had been initiated by 97.9% of women. At around six months of age, 37.8% met EBF recommendations (Table 4). Of those who did not meet the recommendations, 51% stopped breastfeeding before one month of age (Figure 1A). At the time of participation, 66.2% were still breastfeeding. A progressive decline in breastfeeding was observed between birth and seven months; however, it seemed to be stabilising around 9–10 months of age (Figure 1B).
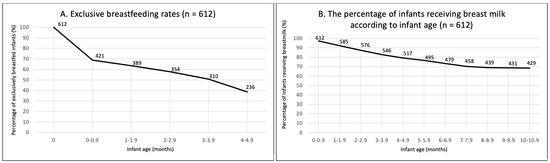
Figure 1.
(A) Exclusive breastfeeding prevalence (between birth and 4.9 months). (B) The percentage of infants receiving breast milk according to infant age. N = 612.
Infants had a higher odds of meeting EBF recommendations if their caregivers had the following characteristics: they were older (1.05 (1.01, 1.08), p = 0.008) vs. young; their highest qualification was university vs. school (0.37 (0.22, 0.64), p < 0.001); they were not currently working vs. employed full-time (0.54 (0.31, 0.96), p = 0.035); they were multiparous mothers vs. primiparous (0.67 (0.48, 0.93), p = 0.016); they had two children in the household (1.56 (1.08, 2.27), p = 0.019) vs. one child; and they did not use ECE vs. those who did (0.60 (0.38, 0.95), p = 0.028) (Table 5).

Table 5.
Sociodemographic characteristics associated with adherence to breastfeeding recommendations.
Caregivers were more likely to be breastfeeding at the time of the study (mean infant age 8.4 months) if their highest qualification was university vs. school (0.41 (0.25, 0.64), p < 0.001) or polytechnic (0.47 (0.31, 0.72), p < 0.001) only; they were not currently working vs. employed full-time (0.41 (0.25, 0.64), p = 0.014); they had two children (1.52 (1.03, 2.26), p = 0.037) vs. one child; their infant did not attend ECE vs. those whose infant did attend ECE (0.54 (0.35, 0.81), p = 0.004); or they lived in low vs. high socioeconomic deprivation areas (0.61 (0.39, 0.96), p = 0.032).
3.2.2. Introduction of Solids
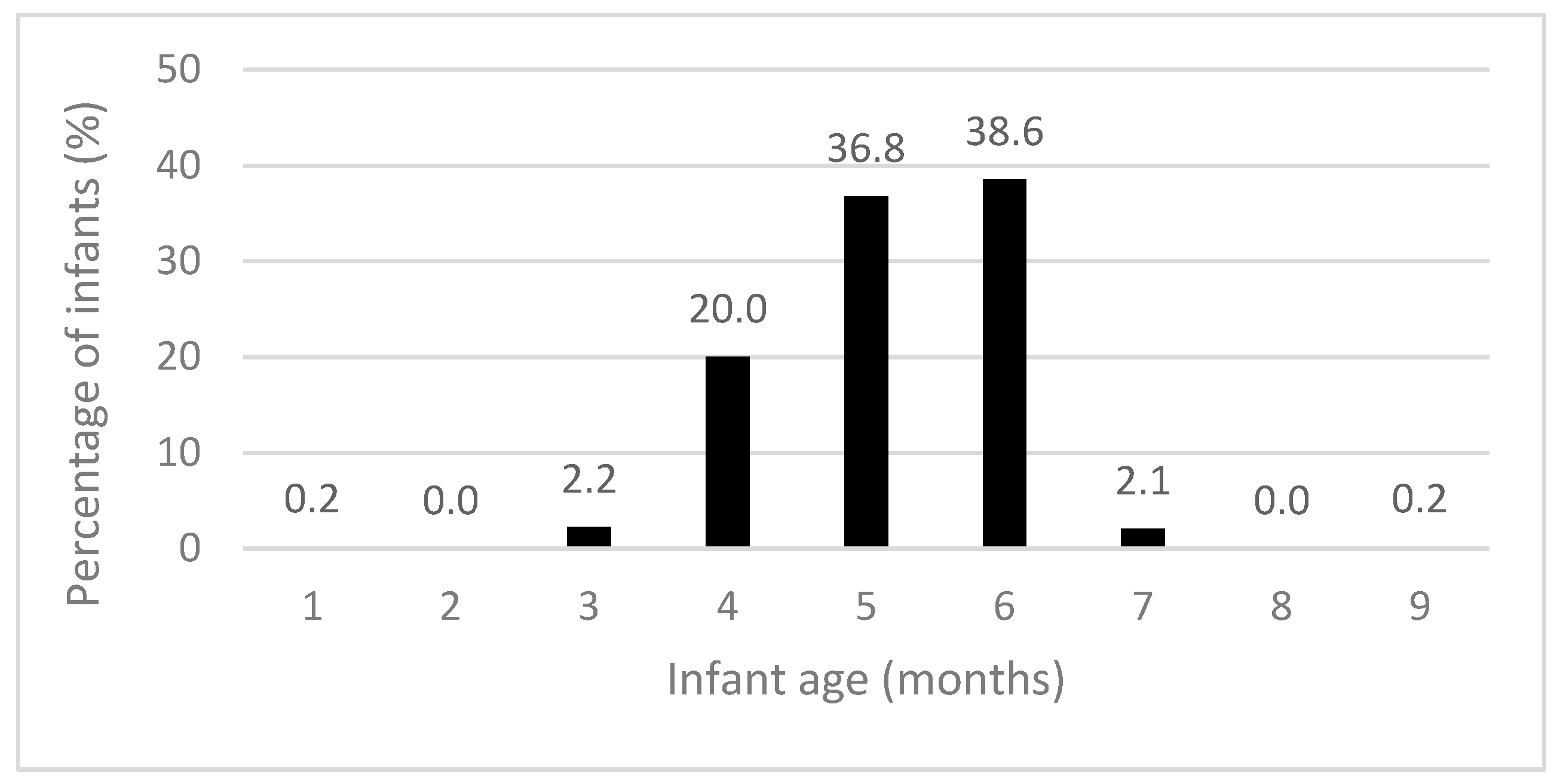
The recommendation for introducing solid foods “around” six months of age was met by 75.4% of participants (Table 4). Solid foods were introduced at a mean age (SD) of 5.18 (0.89) months, with the majority starting by six months (Figure 2). Most infants met the recommendations for puréed food textures (80.3%), spoon-feeding (74.1%), and the introduction of iron-rich foods (88.3%) (Table 4). Few infants consumed an iron-rich food, vegetables, and fruit during both diet recall days (13.4%). During the time when solid foods were introduced, 61.4% and 65.6% of infants consumed infant rice cereal and red meat, respectively.
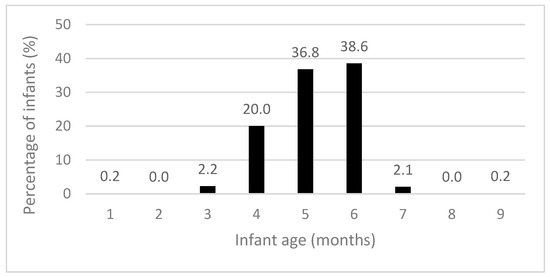
Figure 2.
Infant age when solid foods were introduced (n = 625).
Infants were more likely to be introduced to solid foods “around” six months of age if they had younger (1.12 (1.08, 0.02), p < 0.001) vs. older caregivers; their caregiver’s highest qualification was university level vs. school (0.21 (0.13, 0.34), p < 0.001) or polytechnic (0.53 (0.33, 0.84), p = 0.007); their caregiver had one child in the household vs. four or more (0.44 (0.23, 0.84), p = 0.013); they did not attend ECE vs. those who did (0.54 (0.35, 0.81), p = 0.004); or they were living in a low vs. high socioeconomic deprivation area (0.47 (0.28, 0.77), p = 0.003) (Table 6). Infants were more likely to be spoon-fed when starting solid foods if their caregiver’s highest qualification was school-level (2.08 (1.16, 3.70), p = 0.013) or polytechnic-level (1.70 (1.05, 2.77), p = 0.032) vs. university-level and if the infant attended ECE (2.29 (1.30, 4.03), p = 0.004) vs. those who did not.

Table 6.
Sociodemographic characteristics associated with adherence to solid food introduction recommendations.
3.2.3. Food Group Intake of Infants
Vegetables (63.1%) and fruit (53.9%) were the most commonly consumed food groups. Fewer than half of infants consumed grain foods (49.5%), milk and milk products (38.6%), and meat and protein-rich foods (31.8%) on both diet recall days. A small proportion (6.5%) of infants consumed each food group on both diet recall days. When asked if caregivers felt that their infant consumed a variety of foods, 144 of 146 (98.6%) participants reported “sometimes or more”.
Infants were more likely to consume iron-rich foods and vegetables on both diet recall days if caregivers were older (iron-rich: 1.06 (1.02, 1.10), p = 0.003; vegetables: 1.07 (1.04, 1.12), p <0.001) vs. younger, primiparous (iron-rich: 1.44 (1.01, 2.04), p = 0.042; vegetables: 1.81 (1.29, 2.52), p = 0.001) vs. multiparous, and had a university qualification as their highest level of education vs. a polytechnic qualification (iron-rich: 0.58 (0.36, 0.94), p = 0.020; vegetables: 0.52 (0.35, 0.79), p < 0.001). Infants were also more likely to consume vegetables on both diet recall days if the caregiver’s highest qualification was university vs. school (0.42 (0.27, 0.68), p < 0.001), there was one child in the household vs. three (0.53 (0.33, 0.86), p = 0.010) and four or more (0.23 (0.12, 0.45), p < 0.001), and they lived in an area of low vs. high socioeconomic deprivation (0.45 (0.29, 0.70), p < 0.001) (Table 7). Infants were more likely to consume fruit if their caregiver’s highest qualification was university vs. school (0.47 (0.29, 0.75), p = 0.002).

Table 7.
Sociodemographic characteristics associated with adherence to food variety recommendations on both 24-h recall days.
3.2.4. Appropriate Foods and Beverages
Most participants met the recommendations for avoiding inappropriate foods, including salt (75.7%) and sugar (90.9%) (Table 4). For those using salt, this was typically added to roasted meat/vegetables, vegetable mash, family meals, and potato fries. Sugar was added to Weet-Bix, baking, fruit purées, and family meals. Inappropriate beverages were rarely consumed, and 93.9% of infants met the recommendation for not offering juice, cordial, fruit drink, flavoured milk, soft drinks, tea, coffee, or alcohol (Table 4). Juice was the predominant inappropriate beverage reported in diet recalls (6.6%). Tea and coffee were not reported in any diet recall.
The associations between sociodemographic characteristics and not adding salt and sugar to infant food are shown in Table 8. Primiparous mothers (0.53 (0.36, 0.78), p = 0.001) were less likely to add salt to infant foods than multiparous mothers. Those with two (1.64 (1.06, 2.54), p = 0.026) or three (1.87 (1.09, 3.18), p = 0.022) children in the household were more likely to add salt to infant foods than those with one child. Younger caregivers (0.94 (0.88, 0.99), p = 0.019) were less likely to add sugar than older caregivers, and those with a school qualification only (3.20 (1.65, 6.19), p = 0.001) were more likely to add sugar than university-educated caregivers.

Table 8.
Sociodemographic characteristics associated with adherence to the recommendation of not adding salt and sugar.
3.2.5. Feeding Environment
At the age of participation, 86% of infants were no longer fully spoon-fed, aligning with the MoH recommendation that encourages self-feeding from a young age (Table 4). Infants were more likely to meet the recommendation for self-feeding if there was one child in the household vs. four or more children in the household (0.41 (0.20, 0.87), p = 0.020) (Table 9). Infants of caregivers with part-time employment had a higher odds of self-feeding than those whose caregivers were not currently working (2.21 (1.10, 4.44), p = 0.026).

Table 9.
Sociodemographic characteristics associated with adherence to self-feeding from a young age recommendation.
4. Discussion
The current study has provided insight into adherence to the 2021 NZ MoH infant feeding guidelines in an ethnically diverse infant population. High levels of adherence to recommendations for the introduction of solid foods, appropriate foods and beverages, and eating environments were observed, whereas breastfeeding and food group recommendations were less commonly met. Socioeconomic deprivation, caregiver education, parity, and the number of children in the household were key sociodemographic characteristics associated with adherence to the NZ guidelines.
4.1. Adherence to Breastfeeding Recommendations
Breastfeeding initiation rates in NZ are high [3,19]; however, as seen in this study and previous NZ research, many women do not meet EBF and continued breastfeeding recommendations [10,15,16,39,40]. Although most women did not meet the EBF recommendation, our cohort’s response was higher (37.8%) than that observed in GUiNZ (34.3%) in 2009–2011, suggesting that adherence might be improving. Despite higher adherence, further promotion is required if NZ is to meet the 2030 global nutrition target of at least 70% of women EBF to six months of age [41]. A possible area to target is EBF during the first month—the vast majority of those who did not meet the EBF recommendation in our study stopped during this time. These results indicate a crucial time window that needs further investigation. Although we did not investigate the reasons why women stopped EBF in our study, we found that primiparous women were less likely to meet the EBF recommendation. Similar patterns have been observed in NZ [10] and other high-income countries [42], with strong associations between EBF and previous breastfeeding experience [42]. Early intervention through breastfeeding support programmes has shown beneficial outcomes [43] and could be targeted towards those, such as primiparous mothers, who are at higher risk of not meeting EBF recommendations. We also observed that caregivers who were older, more highly educated, not currently working, and whose infants did not attend ECE were more likely to meet the EBF recommendation. This agrees with global findings [44], with factors such as education, employment status, and use of ECE often being interconnected with socioeconomic status, with those who are more highly educated having greater opportunity to take paid maternity leave than those who are less highly educated and require ECE support when returning to work [42,45]. Sustaining breastfeeding is typically harder for women who return to work in the first twelve months postpartum, with common reports of inadequate support and facilities for breastfeeding [46,47]. Finding ways to support women who are required to return to work, such as encouraging peer support and introducing regulations for the provision of appropriate facilities, has improved the incidence of EBF in other countries [48] and would be beneficial in NZ. Further—qualitative—input from mothers would also be helpful to determine what support is required to achieve the EBF recommendations.
After six months of age, breastfeeding continued to decline. Rates of decline, however, seem to have reduced since those reported in GUiNZ (48%) [11] and to have plateaued around 9–10 months of age, with 70.4% of women (n = 431) continuing to breastfeed at nine months of age in our study. As in GUiNZ [10], women in the current study with higher levels of education were more likely to meet the continued breastfeeding recommendations. As shown for EBF, education, returning to work, use of ECE, and socioeconomic status are often interconnected, making these complex markers to investigate [42,45]. The positive influence of previous experience was also evident for continued breastfeeding, as seen for EBF, with caregivers of two children having higher adherence, suggesting that experience and education are key factors in sustaining breastfeeding. Further support to sustain breastfeeding for first-time mothers and those who are required to return to work may improve rates of adherence in NZ.
4.2. Adherence to Complementary Feeding Recommendation
The recommendation to introduce solid foods “around six months of age” was met by most FFNZ participants (75.4%, average age of 5.18 months); this was notably higher than for participants in the GUiNZ study (56.9%), which reported that 39.4% of infants had started solid foods before or at four months of age [10,49]. Other recent NZ studies have also suggested that the average age of solid food introduction has increased since GUiNZ, with an average age of 5.2 months reported in 2018 [18] and few infants being offered solid foods before four months (5.4%) in the 2020–2021 NZ Health Survey [50]. However, sample bias within studies focusing on nutrition may explain differences, with a common limitation being that those who are interested in nutrition take part in those studies, compared to GUiNZ, which included a wider range of health and environmental variables beyond nutrition. A higher number of Pacific mothers, who were less likely to meet solid food introduction recommendations than European mothers, also took part in GUiNZ compared to the current study. Similar trends exist in Australia, with survey results suggesting that the average age of solid food introduction is moving towards “around six months” (5.3 months [3], 5.1 months [51], 5.0 months [13]), with few caregivers providing solid foods before four months [51]. As in GUiNZ, caregivers with lower education levels and those living in areas of high socioeconomic deprivation were less likely to meet this recommendation [10], identifying key population groups who require additional support. The relationship between low adherence and attending ECE was a new finding and may suggest an area of influence that needs additional investigation.
Previously reported trends for the increasing number of caregivers following BLW [52] did not appear to influence food textures or feeding methods in the current study. Most infants consumed puréed foods when solids were introduced and were spoon-fed by an adult. However, university-educated women were less likely to spoon feed their infant when starting solid foods, agreeing with findings that maternal education may influence BLW trends [53]. Attendance at ECE appears to be a positive influence for spoon-feeding, a finding which has not been reported before. The rate of introduction of iron-rich foods when starting solids (88.3%) was similar to that observed in recent studies [18], suggesting that many infants are offered iron-rich foods in the early complementary feeding period. We also observed a higher likelihood that iron-fortified infant cereals were consumed (64.1%) than that reported in a recent online survey (12%) [18] and GUiNZ (47.9%) [11]. This may be secondary to the increased availability of these products in NZ [29]. However, the continuation of offering iron-fortified cereals in addition to other food groups was low, with only 13.4% of infants consuming an iron-rich food, vegetables, and fruit on both recall days. Further promotion of the importance of offering these foods daily during complementary feeding is required in NZ to increase diet diversity and the provision of a wide range of nutritious foods.
4.3. Adherence to Food Group Recommendations
Few infants consumed a food from each food group on both diet recall days (6.5%), increasing the risk of nutritional inadequacies. Our results were lower than those reported by GUiNZ [11]. In GUiNZ, most infants consumed vegetables (91% vs. 63.2% in the current study), fruit (87% vs. 53.9%), grain foods (90% vs. 49.5%), and meat and protein-rich foods (61% vs. 31.8%) daily at nine months of age [10,11]. GUiNZ did not report the consumption of milk and milk products. The different (and mainly younger) infant ages we recruited could be a factor that explains why fewer infants met the recommendations in our study, with foods often being gradually added to infants’ diets during the complementary feeding transition [54]. The difference of approximately one month between average ages (8.4 months in the current study vs. 9.0 months in GUiNZ) in the studies, therefore, must be considered. This, however, was not the case when comparing our results to those reported by Morison et al. [33], in which the majority of 6–8-month-old infants consumed vegetables (96%) and fruit (96%) at least once during a three-day food diary. Again, however, differences in methodological approaches need to be considered: in the Morison et al. study [33], intake was calculated across a three-day period, which is likely to have contributed to higher results than when assessing daily intake by 24-h recall, as in our study. Therefore, this is the first study to suggest that most infants aged 7.0 to 10.0 months are not meeting MoH food group recommendations, and further investigation into adherence of infants at different ages is required. Investigation into the nutritional implications of not meeting MoH recommendations is also required to determine the impact that current dietary practices have on nutrient intake. Given that 98.6% of parents reported that their infants consumed a varied diet “sometimes or more”, further promotion about the optimal provision of food groups is likely to be required to support understanding of the recommendations promoting daily consumption of each food group from the start of complementary feeding.
4.4. Adherence to Appropriate Food and Beverage Recommendations
Research on the use of added salt and sugar in the infant’s diet has provided conflicting results in NZ. The absence of sugar added to food in the current study (90.6%) is the highest adherence observed in NZ to date, higher than in GUiNZ (86%) [10] and in Morison et al. (55%) [33]. Adherence to the salt recommendation, however, is lower (76.5%) than in GUiNZ (84%) [9] but higher than that reported by Morison et al. (22%) [33]. In the current study, the addition of salt appeared to come from family meals, becoming more frequent with higher parity. This is consistent with results observed in other high-income countries [3,4,55,56,57], indicating a need for education about sources of salt to improve recommendation adherence. Sugar was added to a range of foods, indicating that promotion strategies should focus on key messages (e.g., that added sugar is not required in an infant’s diet).
Before the current study, GUiNZ was the only study that had reported inappropriate drinks consumed by NZ infants [10]. The GUiNZ study found a moderate adherence (61%) to recommendations, whereas adherence in the current study was considerably higher (93.9%). In alignment with GUiNZ [10] and studies from the UK [42], juice continues to be the most commonly consumed inappropriate drink. Further research into why these beverages continue to be offered would allow targeted approaches to improve recommendation adherence.
4.5. Strengths, Limitations, and Future Research
The present findings should be interpreted within the context of the study’s strengths and limitations. While the FFNZ study did not have a representative sample, the cohort recruited was diverse in many respects, including a large number of usually unrepresented participants, including the Māori population and those living in higher deprivation areas. Weighted estimates have been provided where appropriate. Our results provide important updated information about adherence to the MoH infant feeding guidelines. A potential limitation is that the key indicators used in this study are those from the 2021 guideline release, and our data were collected between June 2020 and February 2022; therefore, a portion of the reported data were collected before the guidelines were available to the public. The decision to compare to the 2021 guidelines was made after comparisons between guidelines showed minimal changes to the key indicators discussed. Although a full analysis of breastfeeding to two years could not be completed, FFNZ provides a snapshot of “continued breastfeeding” from 7.0 to 10.0 months of age. A small number of infants were born preterm (n = 46), and it is not known whether caregivers reported corrected age for starting solid foods or not. A three-pass assessment approach and photographic memory prompts were used to ensure accuracy of diet recalls, but the 24-h diet recalls relied on caregiver-reported data and have the potential for misreporting. Although a full representation of the diet, including the usual intake of foods, cannot be achieved from two diet recalls, low daily consumption of MoH food groups is evident.
Further research is now required to investigate why early breastfeeding cessation is occurring and what interventions can be implemented to support breastfeeding. Investigation into the nutritional implications of not meeting food group recommendations during complementary feeding will be important to determine the seriousness of these dietary shortcomings.
5. Conclusions
The current study results provide an important update on adherence to the newest MoH infant feeding guidelines in NZ. The initiation of breastfeeding continues to be high; however, EBF to “around” six months of age is low. Most infants met recommendations for starting solid foods, but many did not consume foods from the MoH food groups daily. The consumption of inappropriate foods and beverages has improved since previous assessments; however, a small number of caregivers continue to offer salt, sugar, and inappropriate beverages. As seen previously, there are associations between sociodemographic characteristics and adherence, identifying key groups (primiparous mothers, caregivers with lower levels of education, those living with multiple children, and those living in areas of high socioeconomic deprivation) that require additional support with infant feeding. Further research is now required to identify reasons for these findings and the most appropriate support for NZ whānau so that they can adhere to current recommendations.
Author Contributions
Conceptualisation, A.-L.H. and R.T.; methodology, K.L.B., P.v.H., A.-L.H., R.T., J.H., L.D., L.T.M. and C.C.; formal analysis, K.J.B. and J.H.; investigation, K.J.B., J.M., R.P., M.C., N.M., A.C., E.J., I.K., M.R., B.B., R.J. and A.W.; data curation, K.J.B., J.H., E.F. and E.J.; writing—original draft preparation, K.J.B.; writing—review and editing, K.L.B., P.v.H., A.-L.H., R.T., J.H., L.D., L.T.M. and C.C.; visualisation, K.J.B.; supervision, K.L.B., P.v.H. and C.C.; project administration, J.M. and R.P.; funding acquisition, A.-L.H. and R.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Health Research Council (HRC) of New Zealand (19/172). N.M. and K.J.B. were supported by the Health Research Council of New Zealand grant; L.D. was supported by a Lotteries Health Research Postdoctoral Fellowship; N.M., A.C. and I.K. were supported by University of Otago doctoral scholarships; E.J., R.J. and M.C. were supported by doctoral scholarships from Massey University.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Health and Disability Ethics Committee New Zealand (19/STH/151), and written consent was obtained from participants before data collection. The study was registered with the Australian New Zealand Clinical Trials Registry (www.anzctr.org.au, accessed on 29 October 2023, registration number: ACTRN12620000459921).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data used in the present study are not publicly available due to ethical restrictions related to the consent provided by participants. An ethically compliant dataset may be made available by the corresponding author upon reasonable request and upon approval by the Health and Disability Ethics Committees New Zealand.
Acknowledgments
The authors would like to acknowledge the families who took part in the study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Smithers, L.G.; Golley, R.K.; Brazionis, L.; Emmett, P.; Northstone, K.; Lynch, J.W. Dietary patterns of infants and toddlers are associated with nutrient intakes. Nutrients 2012, 4, 935–948. [Google Scholar]
- Birch, L.; Savage, J.S.; Ventura, A. Influences on the development of children’s eating behaviours: From infancy to adolescence. Can. J. Diet. Pract. Res. 2007, 68, s1. [Google Scholar] [PubMed]
- Lioret, S.; McNaughton, S.; Spence, A.; Crawford, D.; Campbell, K. Tracking of dietary intakes in early childhood: The Melbourne InFANT Program. Eur. J. Clin. Nutr. 2013, 67, 275–281. [Google Scholar] [CrossRef]
- Grummer-Strawn, L.M.; Scanlon, K.S.; Fein, S.B. Infant feeding and feeding transitions during the first year of life. Pediatrics 2008, 122, S36–S42. [Google Scholar] [CrossRef]
- Ministry of Health. Healthy Eating Guidelines for New Zealand Babies and Toddlers (0–2 Years Old); Ministry of Health: Wellington, New Zealand, 2021.
- Plunket. What We Offer. Available online: https://www.plunket.org.nz/ (accessed on 26 October 2023).
- World Health Organization. Infant and Young Child Feeding. Available online: https://www.who.int/news-room/fact-sheets/detail/infant-and-young-child-feeding (accessed on 5 October 2022).
- United Nations International Children’s Emergency Fund. Too Many Children Are Not Eating the Nutrient-Rich Foods They Need to Grow and Develop. Available online: https://data.unicef.org/topic/nutrition/diets/ (accessed on 5 October 2022).
- Golley, R.K.; Smithers, L.G.; Mittinty, M.N.; Brazionis, L.; Emmett, P.; Northstone, K.; Campbell, K.; McNaughton, S.A.; Lynch, J.W. An index measuring adherence to complementary feeding guidelines has convergent validity as a measure of infant diet quality. J. Nutr. 2012, 142, 901–908. [Google Scholar] [PubMed]
- Gontijo de Castro, T.; Gerritsen, S.; Wall, C.; Grant, C.; Teixeira, J.; Marchioni, D.; Pillai, A.; Morton, S. Infant Feeding in New Zealand: Adherence to Food and Nutrition Guidelines among the Growing up in New Zealand Cohort; Ministry of Social Development: Wellington, New Zealand, 2018.
- Morton, S.M.B.; Atatoa Carr, P.E.; Grant, C.C.; Lee, A.C.; Bandara, D.K.; Mohal, J.; Kinloch, J.M.; Schmidt, J.M.; Hedges, M.R.; Ivory, V.C.; et al. Growing up in New Zealand report 2: Now we are born. In Growing up in New Zealand; University of Auckland: Auckland, New Zealand, 2012. [Google Scholar]
- United Nations International Children’s Emergency Fund. Breastfeeding: A Mother’s Gift, for Every Child; Unicef: Geneva, Switzerland, 2018. [Google Scholar]
- Netting, M.J.; Moumin, N.A.; Knight, E.J.; Golley, R.K.; Makrides, M.; Green, T.J. The Australian Feeding Infants and Toddler Study (OzFITS 2021): Breastfeeding and Early Feeding Practices. Nutrients 2022, 14, 206. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Breastfeeding among U.S. Children Born 2011–2018, CDC National Immunization Survey. 2021. Available online: https://www.cdc.gov/breastfeeding/data/nis_data/results.html (accessed on 16 September 2020).
- Castro, T.; Grant, C.; Wall, C.; Welch, M.; Marks, E.; Fleming, C.; Teixeira, J.; Bandara, D.; Berry, S.; Morton, S. Breastfeeding indicators among a nationally representative multi-ethnic sample of New Zealand children. N. Z. Med. J. 2017, 130, 34–44. [Google Scholar]
- Manhire, K.M.; Williams, S.M.; Tipene-Leach, D.; Baddock, S.A.; Abel, S.; Tangiora, A.; Jones, R.; Taylor, B.J. Predictors of breastfeeding duration in a predominantly Māori population in New Zealand. BMC Pediatr. 2018, 18, 299. [Google Scholar]
- Au, L.E.; Gurzo, K.; Paolicelli, C.; Whaley, S.E.; Weinfield, N.S.; Ritchie, L.D. Diet quality of US infants and toddlers 7–24 months old in the WIC Infant and Toddler Feeding Practices Study-2. J. Nutr. 2018, 148, 1786–1793. [Google Scholar] [CrossRef]
- Fu, X.; Conlon, C.A.; Haszard, J.J.; Beck, K.L.; von Hurst, P.R.; Taylor, R.W.; Heath, A.-L.M. Food fussiness and early feeding characteristics of infants following Baby-Led Weaning and traditional spoon-feeding in New Zealand: An internet survey. Appetite 2018, 130, 110–116. [Google Scholar]
- Grimes, C.A.; Szymlek-Gay, E.A.; Campbell, K.J.; Nicklas, T.A. Food sources of total energy and nutrients among US infants and toddlers: National Health and Nutrition Examination Survey 2005–2012. Nutrients 2015, 7, 6797–6836. [Google Scholar] [CrossRef] [PubMed]
- Roess, A.A.; Jacquier, E.F.; Catellier, D.J.; Carvalho, R.; Lutes, A.C.; Anater, A.S.; Dietz, W.H. Food consumption patterns of infants and toddlers: Findings from the Feeding Infants and Toddlers Study (FITS) 2016. J. Nutr. 2018, 148, 1525S–1535S. [Google Scholar] [PubMed]
- Fox, M.K.; Pac, S.; Devaney, B.; Jankowski, L. Feeding infants and toddlers study: What foods are infants and toddlers eating? J. Am. Diet. Assoc. 2004, 104, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Pak-Gorstein, S.; Haq, A.; Graham, E.A. Cultural influences on infant feeding practices. Pediatr. Rev. 2009, 30, e11–e21. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Lee, M. An exploration of experiences of mothers following a baby-led weaning style: Developmental readiness for complementary foods. Matern. Child Nutr. 2013, 9, 233–243. [Google Scholar] [CrossRef]
- Robinson, S.; Marriott, L.; Poole, J.; Crozier, S.; Borland, S.; Lawrence, W.; Law, C.; Godfrey, K.; Cooper, C.; Inskip, H. Dietary patterns in infancy: The importance of maternal and family influences on feeding practice. Br. J. Nutr. 2007, 98, 1029–1037. [Google Scholar] [CrossRef]
- O’Donovan, S.M.; Murray, D.M.; Hourihane, J.O.B.; Kenny, L.C.; Irvine, A.D.; Kiely, M. Adherence with early infant feeding and complementary feeding guidelines in the Cork Baseline birth cohort study. Public Health Nutr. 2015, 18, 2864–2873. [Google Scholar] [CrossRef]
- Gage, H.; Williams, P.; Rosen-Von Hoewel, V.; Laitinen, K.; Jakobik, V.; Martin-Bautista, E.; Schmid, M.; Egan, B.; Morgan, J.; Decsi, T. Influences on infant feeding decisions of first-time mothers in five European countries. Eur. J. Clin. Nutr. 2012, 66, 914–919. [Google Scholar] [CrossRef]
- Synnott, K.; Bogue, J.; Edwards, C.; Scott, J.; Higgins, S.; Norin, E.; Frias, D.; Amarri, S.; Adam, R. Parental perceptions of feeding practices in five European countries: An exploratory study. Eur. J. Clin. Nutr. 2007, 61, 946–956. [Google Scholar] [CrossRef]
- Anderson, A.S.; Guthrie, C.-A.; Alder, E.M.; Forsyth, S.; Howie, P.W.; Williams, F.L. Rattling the plate: Reasons and rationales for early weaning. Health Educ. Res. 2001, 16, 471–479. [Google Scholar] [CrossRef]
- Katiforis, I.; Fleming, E.A.; Haszard, J.J.; Hape-Cramond, T.; Taylor, R.W.; Heath, A.-L.M. Energy, Sugars, Iron, and Vitamin B12 Content of Commercial Infant Food Pouches and Other Commercial Infant Foods on the New Zealand Market. Nutrients 2021, 13, 657. [Google Scholar] [CrossRef]
- Padarath, S.; Gerritsen, S.; Mackay, S. Nutritional aspects of commercially available complementary foods in New Zealand supermarkets. Nutrients 2020, 12, 2980. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Jones, S.W.; Rowan, H. Baby-led weaning: The evidence to date. Curr. Nutr. Rep. 2017, 6, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Cameron, S.L.; Taylor, R.W.; Heath, A.-L.M. Parent-led or baby-led? Associations between complementary feeding practices and health-related behaviours in a survey of New Zealand families. BMJ Open 2013, 3, e003946. [Google Scholar] [CrossRef] [PubMed]
- Morison, B.J.; Taylor, R.W.; Haszard, J.J.; Schramm, C.J.; Erickson, L.W.; Fangupo, L.J.; Fleming, E.A.; Luciano, A.; Heath, A.-L.M. How different are baby-led weaning and conventional complementary feeding? A cross-sectional study of infants aged 6–8 months. BMJ Open 2016, 6, e010665. [Google Scholar] [CrossRef]
- Taylor, R.W.; Conlon, C.A.; Beck, K.L.; von Hurst, P.R.; Te Morenga, L.A.; Daniels, L.; Haszard, J.J.; Meldrum, A.M.; McLean, N.H.; Cox, A.M. Nutritional implications of baby-led weaning and baby food pouches as novel methods of infant feeding: Protocol for an observational study. JMIR Res. Protoc. 2021, 10, e29048. [Google Scholar] [CrossRef]
- Ministry of Health. HISO 10001: 2017 Ethnicity Data Protocols; Ministry of Health: Wellington, New Zealand, 2017.
- Atkinson, J.; Salmond, C.; Crampton, P. NZDep2013 Index of Deprivation; University of Otago: Wellington, New Zealand, 2014. [Google Scholar]
- Thoyre, S.M.; Pados, B.F.; Park, J.; Estrem, H.; Hodges, E.A.; McComish, C.; Van Riper, M.; Murdoch, K. Development and content validation of the pediatric eating assessment tool (Pedi-EAT). Am. J. Speech-Lang. Pathol. 2014, 23, 46–59. [Google Scholar] [CrossRef]
- Daniels, L.; Haszard, J.J.; Taylor, R.W.; Taylor, B.J. Prevalence of low and high BMI during the first 3 years of life: Using New Zealand national electronic health data. Pediatr. Obes. 2023, 18, e13013. [Google Scholar] [CrossRef]
- Plunket. Annual Breastfeeding Statistics. Available online: https://www.plunket.org.nz/news-and-research/research-from-plunket/plunket-breastfeeding-data-analysis/annual-breastfeeding-statistics/ (accessed on 16 September 2020).
- Ministry of Health. Release of the Regional Results: 2017–2020: New Zealand Health Survey; Ministry of Health: Wellington, New Zealand, 2021.
- United Nations Children’s Fund. The Extension of the 2025 Maternal, Infant and Young Child Nutrition Targets to 2030. Available online: https://data.unicef.org/resources/who-unicef-discussion-paper-nutrition-targets/ (accessed on 15 August 2023).
- McAndrew, F.; Thompson, J.; Fellows, L.; Large, A.; Speed, M.; Renfrew, M.J. Infant Feeding Survey 2010; Health and Social Care Information Centre: Leeds, UK, 2012.
- Wong, M.S.; Mou, H.; Chien, W.T. Effectiveness of educational and supportive intervention for primiparous women on breastfeeding related outcomes and breastfeeding self-efficacy: A systematic review and meta-analysis. Int. J. Nurs. Stud. 2021, 117, 103874. [Google Scholar] [CrossRef]
- Mangrio, E.; Persson, K.; Bramhagen, A.C. Sociodemographic, physical, mental and social factors in the cessation of breastfeeding before 6 months: A systematic review. Scand. J. Caring Sci. 2018, 32, 451–465. [Google Scholar] [CrossRef]
- Gallegos, D.; Parkinson, J.; Duane, S.; Domegan, C.; Jansen, E.; Russell-Bennett, R. Understanding breastfeeding behaviours: A cross-sectional analysis of associated factors in Ireland, the United Kingdom and Australia. Int. Breastfeed. J. 2020, 15, 103. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, E.; Hiller, J.E.; Turnbull, D. Determinants of infant feeding practices in a low socio-economic area: Identifying environmental barriers to breastfeeding. Aust. N. Z. J. Public Health 1999, 23, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.K.; Khoury, A.J.; Hinton, A.W.; Carothers, C. Predictors of breastfeeding intention among low-income women. Matern. Child Health J. 2004, 8, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Scott, V.C.; Taylor, Y.J.; Basquin, C.; Venkitsubramanian, K. Impact of key workplace breastfeeding support characteristics on job satisfaction, breastfeeding duration, and exclusive breastfeeding among health care employees. Breastfeed. Med. 2019, 14, 416–423. [Google Scholar] [CrossRef]
- Morton, S.M.B.; Atatoa Carr, P.E.; Grant, C.C.; Berry, S.D.; Bandara, D.K.; Mohal, J.; Tricker, P.J.; Ivory, V.C.; Kingi, T.R.; Liang, R.; et al. Now we are two: Describing our first 1000 days. In Growing Up in New Zealand; Ministry of Health: Wellington, New Zealand, 2014. [Google Scholar]
- Ministry of Health. Indicator: Given Solids before 4 Months (among Children Aged 4 Months to <5 Years). Available online: https://minhealthnz.shinyapps.io/nz-health-survey-2021-22-annual-data-explorer/_w_81846aaf/#!/explore-indicators (accessed on 15 August 2023).
- Arora, A.; Manohar, N.; Hector, D.; Bhole, S.; Hayen, A.; Eastwood, J.; Scott, J.A. Determinants for early introduction of complementary foods in Australian infants: Findings from the HSHK birth cohort study. Nutr. J. 2020, 19, 16. [Google Scholar] [CrossRef]
- Rapley, G. Baby-led weaning: The theory and evidence behind the approach. J. Health Visit. 2015, 3, 144–151. [Google Scholar] [CrossRef]
- Brown, A. Differences in eating behaviour, well-being and personality between mothers following baby-led vs. traditional weaning styles. Matern. Child Nutr. 2016, 12, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Nicklaus, S. Development of food variety in children. Appetite 2009, 52, 253–255. [Google Scholar] [CrossRef]
- Bournez, M.; Ksiazek, E.; Charles, M.-A.; Lioret, S.; Brindisi, M.-C.; de Lauzon-Guillain, B.; Nicklaus, S. Frequency of use of added sugar, salt, and fat in infant foods up to 10 months in the nationwide ELFE cohort study: Associated infant feeding and caregiving practices. Nutrients 2019, 11, 733. [Google Scholar] [CrossRef]
- Wang, Y.; Guglielmo, D.; Welsh, J.A. Consumption of sugars, saturated fat, and sodium among US children from infancy through preschool age, NHANES 2009–2014. Am. J. Clin. Nutr. 2018, 108, 868–877. [Google Scholar] [CrossRef]
- Kay, M.C.; Welker, E.B.; Jacquier, E.F.; Story, M.T. Beverage consumption patterns among infants and young children (0–47.9 months): Data from the Feeding Infants and Toddlers Study, 2016. Nutrients 2018, 10, 825. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).