ARFID—Strategies for Dietary Management in Children
Abstract
:1. Introduction
2. Epidemiology of ARFID
3. Diagnostic Criteria
4. Consequences of ARFID
5. Strategies with ARFID
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Katzman, D.K.; Norris, M.L.; Zucker, N. Avoidant Restrictive Food Intake Disorder. Psychiatr. Clin. N. Am. 2019, 42, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Cañas, L.; Palma, C.; Molano, A.M.; Domene, L.; Carulla-Roig, M.; Cecilia-Costa, R.; Dolz, M.; Serrano-Troncoso, E. Avoidant/restrictive food intake disorder: Psychopathological similarities and differences in comparison to anorexia nervosa and the general population. Eur. Eat. Disord. Rev. 2021, 29, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.M.; Rosen, D.S.; Ornstein, R.M.; Mammel, K.A.; Katzman, D.K.; Rome, E.S.; Callahan, S.T.; Malizio, J.; Kearney, S.; Walsh, B.T. Characteristics of Avoidant/Restrictive Food Intake Disorder in Children and Adolescents: A “New Disorder” in DSM-5. J. Adolesc. Health 2014, 55, 49–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zimmerman, J.; Fisher, M. Avoidant/Restrictive Food Intake Disorder (ARFID). Curr. Issues Pediatric Adolesc. Health Care 2017, 47, 95–103. [Google Scholar] [CrossRef]
- Kurz, S.; Van Dyck, Z.; Dremmel, D.; Munsch, S.; Hilbert, A. Early-onset restrictive eating disturbances in primary school boys and girls. Eur. Child Adolesc. Psychiatry 2015, 24, 779–785. [Google Scholar] [CrossRef] [Green Version]
- Spicer, L.; Strudwick, K.; Kelly, V. GP82 Prevalence rates for avoidant restrictive food intake disorder (ARFID) in tertiary feeding clinic in UK. Arch. Dis. Child. 2019, 104, A63. [Google Scholar]
- Norris, M.; Spettigue, W.; Katzman, D. Update on eating disorders: Current perspectives on avoidant/restrictive food intake disorder in children and youth. Neuropsychiatr. Dis. Treat. 2016, 12, 213–218. [Google Scholar] [CrossRef] [Green Version]
- Nicely, T.A.; Lane-Loney, S.; Masciulli, E.; Hollenbeak, C.S.; Ornstein, R.M. Prevalence and characteristics of avoidant/restrictive food intake disorder in a cohort of young patients in day treatment for eating disorders. J. Eat. Disord. 2014, 2, 1–8. [Google Scholar] [CrossRef]
- Thomas, J.J.; Lawson, E.; Micali, N.; Misra, M.; Deckersbach, T.; Eddy, K.T. Avoidant/Restrictive Food Intake Disorder: A Three-Dimensional Model of Neurobiology with Implications for Etiology and Treatment. Curr. Psychiatry Rep. 2017, 19, 1–9. [Google Scholar] [CrossRef]
- Hilbert, A.; Zenger, M.; Eichler, J.; Brähler, E. Psychometric evaluation of the Eating Disorders in Youth-Questionnaire when used in adults: Prevalence estimates for symptoms of avoidant/restrictive food intake disorder and population norms. Int. J. Eat. Disord. 2021, 54, 399–408. [Google Scholar] [CrossRef]
- Nakai, Y.; Nin, K.; Noma, S.; Teramukai, S.; Wonderlich, S.A. Characteristics of Avoidant/Restrictive Food Intake Disorder in a Cohort of Adult Patients. Eur. Eat. Disord. Rev. 2016, 24, 528–530. [Google Scholar] [CrossRef] [PubMed]
- Dinkler, L.; Yasumitsu-Lovell, K.; Eitoku, M.; Fujieda, M.; Suganuma, N.; Hatakenaka, Y.; Hadjikhani, N.; Bryant-Waugh, R.; Råstam, M.; Gillberg, C. Development of a parent-reported screening tool for avoidant/restrictive food intake disorder (ARFID): Initial validation and prevalence in 4–7-year-old Japanese children. Appetite 2021, 168, 105735. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.; Stone, E.L. Avoidant Restrictive Food Intake Disorder—More Than Just Picky Eating: A Case Discussion and Literature Review. J. Nurse Pract. 2020, 16, 713–717. [Google Scholar] [CrossRef]
- World Health Organization. International Classification of Diseases for Mortality and Morbidity Statistics (11th Revision). 2018. Available online: https://icd.who.int/browse11/l-m/en#/http://id.who.int/icd/entity/129180281 (accessed on 7 March 2022).
- Zucker, N.L.; LaVia, M.C.; Craske, M.G.; Foukal, M.; Harris, A.A.; Datta, N.; Savereide, E.; Maslow, G.R. Feeling and body investigators (FBI): ARFID division-Anacceptance-based interoceptive exposure treatment for children with ARFID. Int. J. Eat. Disord. 2019, 52, 466–472. [Google Scholar] [CrossRef]
- Brigham, K.S.; Manzo, L.D.; Eddy, K.T.; Thomas, J.J. Evaluation and Treatment of Avoidant/Restrictive Food Intake Disorder (ARFID) in Adolescents. Curr. Pediatr. Rep. 2018, 6, 107–113. [Google Scholar] [CrossRef]
- Eddy, K.T.; Thomas, J.J.; Bs, E.H.; Ba, K.E.; Lamont, E.; Ba, C.M.N.; Ba, R.M.P.; Ba, H.B.M.; Bryant-Waugh, R.; Becker, A. Prevalence of DSM-5 avoidant/restrictive food intake disorder in a pediatric gastroenterology healthcare network. Int. J. Eat. Disord. 2015, 48, 464–470. [Google Scholar] [CrossRef]
- Hay, P.; Mitchison, D.; Collado, A.E.L.; González-Chica, D.A.; Stocks, N.; Touyz, S. Burden and health-related quality of life of eating disorders, including Avoidant/Restrictive Food Intake Disorder (ARFID), in the Australian population. J. Eat. Disord. 2017, 5, 1–10. [Google Scholar] [CrossRef]
- Forman, S.F.; McKenzie, N.; Hehn, R.; Monge, M.C.; Kapphahn, C.J.; Mammel, K.A.; Callahan, S.T.; Sigel, E.J.; Bravender, T.; Romano, M.; et al. Predictors of Outcome at 1 Year in Adolescents With DSM-5 Restrictive Eating Disorders: Report of the National Eating Disorders Quality Improvement Collaborative. J. Adolesc. Health 2014, 55, 750–756. [Google Scholar] [CrossRef]
- Norris, M.L.; Robinson, A.; Obeid, N.; Harrison, M.; Spettigue, W.; Henderson, K. Exploring avoidant/restrictive food intake disorder in eating disordered patients: A descriptive study. Int. J. Eat. Disord. 2014, 47, 495–499. [Google Scholar] [CrossRef]
- Ornstein, R.M.; Rosen, D.S.; Mammel, K.A.; Callahan, S.T.; Forman, S.; Jay, M.S.; Fisher, M.; Rome, E.; Walsh, B.T. Distribution of Eating Disorders in Children and Adolescents Using the Proposed DSM-5 Criteria for Feeding and Eating Disorders. J. Adolesc. Health 2013, 53, 303–305. [Google Scholar] [CrossRef]
- Ornstein, R.M.; Essayli, J.H.; Nicely, T.A.; Masciulli, E.; Lane-Loney, S. Treatment of avoidant/restrictive food intake disorder in a cohort of young patients in a partial hospitalization program for eating disorders. Int. J. Eat. Disord. 2017, 50, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Feillet, F.; Bocquet, A.; Briend, A.; Chouraqui, J.-P.; Darmaun, D.; Frelut, M.-L.; Girardet, J.-P.; Guimber, D.; Hankard, R.; Lapillonne, A.; et al. Nutritional risks of ARFID (avoidant restrictive food intake disorders) and related behavior. Archives de Pédiatrie 2019, 26, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Sharp, W.G.; Postorino, V.; McCracken, C.E.; Berry, R.C.; Criado, K.K.; Burrell, T.L.; Scahill, L. Dietary Intake, Nutrient Status, and Growth Parameters in Children with Autism Spectrum Disorder and Severe Food Selectivity: An Electronic Medical Record Review. J. Acad. Nutr. Diet. 2018, 118, 1943–1950. [Google Scholar] [CrossRef] [PubMed]
- Eddy, K.T.; Harshman, S.G.; Becker, K.R.; Bern, E.; Bryant-Waugh, R.; Hilbert, A.; Katzman, D.K.; Lawson, E.A.; Manzo, L.D.; Menzel, J.; et al. Radcliffe ARFID Workgroup: Toward operationalization of research diagnostic criteria and directions for the field. Int. J. Eat. Disord. 2019, 52, 361–366. [Google Scholar] [CrossRef]
- Dovey, T.M.; Kumari, V.; Blissett, J. Eating behaviour, behavioural problems and sensory profiles of children with avoidant/restrictive food intake disorder (ARFID), autistic spectrum disorders or picky eating: Same or different? Eur. Psychiatry 2019, 61, 56–62. [Google Scholar] [CrossRef]
- Mairs, R.; Nicholls, D. Assessment and treatment of eating disorders in children and adolescents. Arch. Dis. Child. 2016, 101, 1168–1175. [Google Scholar] [CrossRef]
- Cooney, M.; Lieberman, M.; Guimond, T.; Katzman, D.K. Clinical and psychological features of children and adolescents diagnosed with avoidant/restrictive food intake disorder in a pediatric tertiary care eating disorder program: A descriptive study. J. Eat. Disord. 2018, 6, 7. [Google Scholar] [CrossRef]
- Bryant-Waugh, R. Avoidant/Restrictive Food Intake Disorder. Child Adolesc. Psychiatr. Clin. N. Am. 2019, 28, 557–565. [Google Scholar] [CrossRef]
- Schmidt, R.; Hiemisch, A.; Kiess, W.; von Klitzing, K.; Schlensog-Schuster, F.; Hilbert, A. Macro- and Micronutrient Intake in Children with Avoidant/Restrictive Food Intake Disorder. Nutrients 2021, 13, 400. [Google Scholar] [CrossRef]
- Jarosz, M.; Rychlik, E.; Stoś, K.; Charzewska, J. Normy Żywienia Dla Populacji Polski I Ich Zastosowanie; Narodowy Instytut Zdrowia Publicznego-Państwowy Zakład Higieny: Warsaw, Poland, 2020.
- Mueller, C. (Ed.) The ASPEN Adult Nutrition Support Core Curriculum, 3rd ed.; American Society for Parenteral and Enteral Nutrition: Silver Spring, MD, USA, 2017. [Google Scholar]
- Takeshima, M.; Ishikawa, H.; Kitadate, A.; Sasaki, R.; Kobayashi, T.; Nanjyo, H.; Kanbayashi, T.; Shimizu, T. Anorexia nervosa-associated pancytopenia mimicking idiopathic aplastic anemia: A case report. BMC Psychiatry 2018, 18, 150. [Google Scholar] [CrossRef]
- Mammel, K.A.; Ornstein, R.M. Avoidant/restrictive food intake disorder: A new eating disorder diagnosis in the diagnostic and statistical manual 5. Curr. Opin. Pediatr. 2017, 29, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Claudino, A.M.; Pike, K.M.; Hay, P.; Keeley, J.W.; Evans, S.C.; Rebello, T.J.; Bryant-Waugh, R.; Dai, Y.; Zhao, M.; Matsumoto, C.; et al. The classification of feeding and eating disorders in the ICD-11: Results of a field study comparing proposed ICD-11 guidelines with existing ICD-10 guidelines. BMC Med. 2019, 17, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Ziółkowska, B. Food restriction/avoidance disorder-considerations in light of research findings. Pediatr. Pol. 2017, 92, 733–738. [Google Scholar] [CrossRef]
- Tamás, D.S.; Melinda, C. Treatment methods of avoidant/restrictive food intake disorder: A review with therapeutic implications. Psihijatr. Danas 2017, 49, 5–24. [Google Scholar]
- Zickgraf, H.F.; Ellis, J.M. Initial validation of the Nine Item Avoidant/Restrictive Food Intake disorder screen (NIAS): A measure of three restrictive eating patterns. Appetite 2018, 123, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Hilbert, A.; van Dyck, Z. Eating Disorders in Youth-Questionnaire. English Version. 21 June 2016. Available online: http://nbn-resolving.de/urn:nbn:de:bsz:15-qucosa-197246 (accessed on 7 February 2022).
- Herpertz-Dahlmann, B. Treatment of eating disorders in child and adolescent psychiatry. Curr. Opin. Psychiatry 2017, 30, 438–445. [Google Scholar] [CrossRef]
- Fraker, C.; Fishbein, M.; Walbert, L.; Cox, S. Food Chaining: The Proven 6-Step Plan to Stop Picky Eating, Solve Feeding Problems, and Expand Your Child’s Diet; Da Capo Press: Boston, MA, USA, 2007; Volume 1. [Google Scholar]
- Chatoor, I. Lack of Interest in Eating or Food, A Subtype of Avoidant/Restrictive Food Intake Disorder (ARFID): Diagnosis, Treatment, and Follow-Up from Infancy to Late Childhood. J. Am. Acad. Child Adolesc. Psychiatry 2019, 58, S85. [Google Scholar] [CrossRef]
- Rosania, K.; Lock, J. Family-Based Treatment for a Preadolescent with Avoidant/Restrictive Food Intake Disorder with Sensory Sensitivity: A Case Report. Front. Psychiatry 2020, 11. [Google Scholar] [CrossRef]
- Lock, J.; Robinson, A.; Sadeh-Sharvit, S.; Rosania, K.; Osipov, L.; Kirz, N.; Derenne, J.; Utzinger, L. Applying family-based treatment (FBT) to three clinical presentations of avoidant/restrictive food intake disorder: Similarities and differences from FBT for anorexia nervosa. Int. J. Eat. Disord. 2019, 52, 439–446. [Google Scholar] [CrossRef]
- Sikora, D. The treatment of avoidant/restrictive food intake disorder (ARFID) with predominance of anxiety presentation. A proposal of a protocol for therapeutic procedure. Psychotherapy 2021, 198, 33–47. [Google Scholar] [CrossRef]
- Thomas, J.J.; Wons, O.B.; Eddy, K.T. Cognitive–behavioral treatment of avoidant/restrictive food intake disorder. Curr. Opin. Psychiatry 2018, 31, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, K.; Forsberg, S.; Colborn, D. Family-Based Treatment for Avoidant Restrictive Food Intake Disorder: Families Facing Neophobias. In Family Therapy for Adolescent Eating and Weight Disorders: New Applications; Loeb, K., Le Grange, D., Lock, J., Eds.; Routledge: New York, NY, USA, 2015; pp. 256–276. [Google Scholar]
- O’Connor, G.; Nicholls, D.; Hudson, L.; Singhal, A. Refeeding Low Weight Hospitalized Adolescents with Anorexia Nervosa: A Multicenter Randomized Controlled Trial. Nutr. Clin. Pract. 2016, 31, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Strandjord, S.E.; Sieke, E.; Richmond, M.; Rome, E.S. Avoidant/Restrictive Food Intake Disorder: Illness and Hospital Course in Patients Hospitalized for Nutritional Insufficiency. J. Adolesc. Health 2015, 57, 673–678. [Google Scholar] [CrossRef] [PubMed]
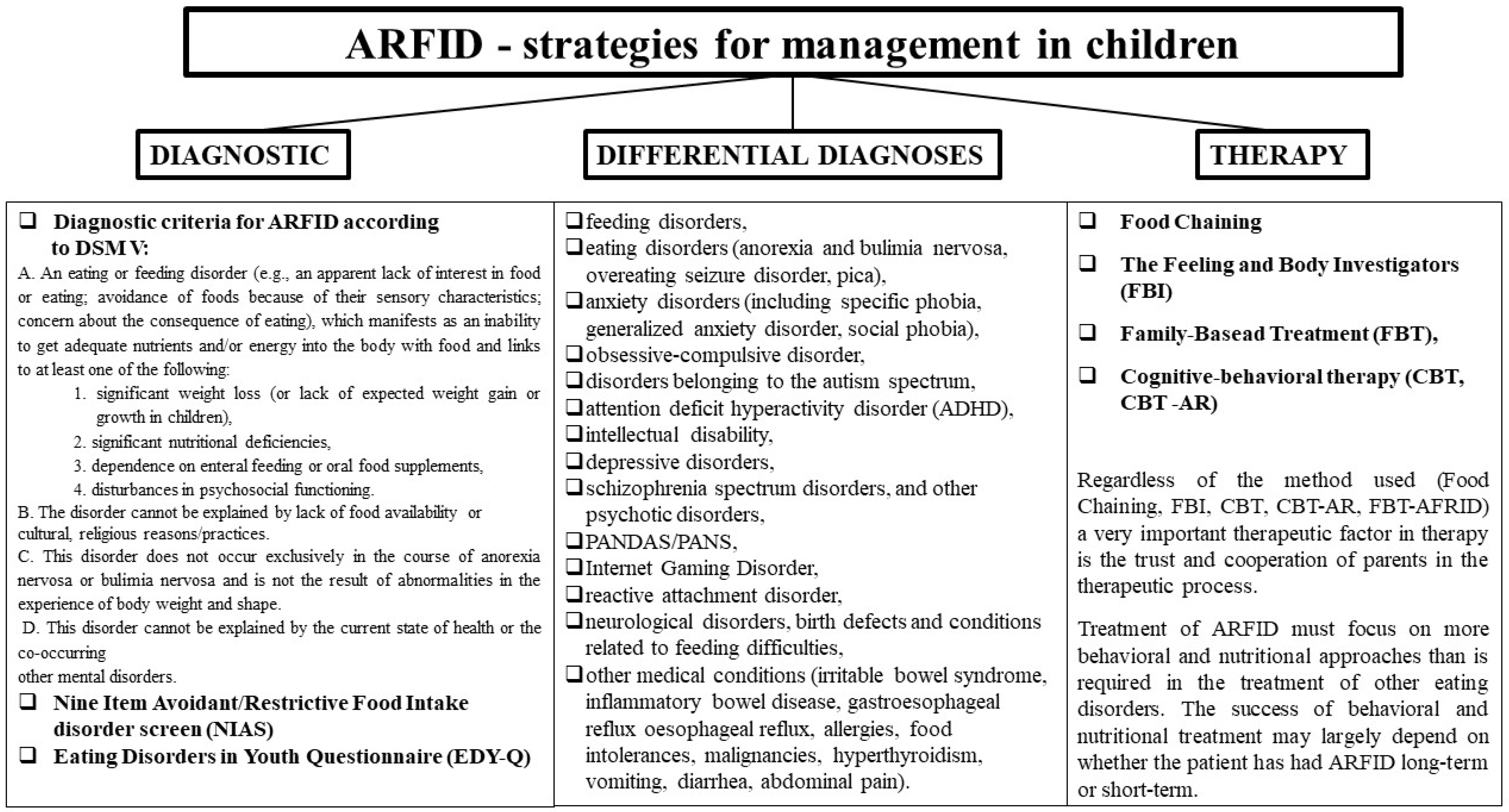
| A. An eating or feeding disorder (e.g., an apparent lack of interest in food or eating; avoidance of foods because of their sensory characteristics; concern about the consequence of eating), which manifests as an inability to get adequate nutrients and/or energy into the body with food and links to at least one of the following: 1. Significant weight loss (or lack of expected weight gain or growth in children); 2. Significant nutritional deficiencies; 3. Dependence on enteral feeding or oral food supplements; 4. Disturbances in psychosocial functioning. |
| B. The disorder cannot be explained by lack of food availability or cultural and religious reasons/practices. |
| C. This disorder does not occur exclusively in the course of anorexia nervosa or bulimia nervosa and is not the result of abnormalities in the experience of body weight and shape. |
| D. This disorder cannot be explained by the current state of health or other co-occurring mental disorders. |
| Type of Food Avoided | Nutrient Deficiency | Basic Parameter | Health Consequences of Deficiency |
|---|---|---|---|
| Cereal products | carbohydrates | body weight and height | hypotrophy |
| fiber | e.g., screening for cancer, atherosclerosis, cholelithiasis | Atherosclerosis, gallstones, diverticulosis, and colorectal cancer, breast cancer in women. | |
| Milk and milk products | calcium | PTH, alkaline phosphatases | rickets, hypocalciuria, reduced bone mineral density, osteopenia, bone weakness or fractures, and osteoporosis. |
| Animal products and dairy products | Riboflavin/Vitamin B2 | the serum concentration of vit. B2 | Low energy levels, poor growth, dry skin/skin problems, hair loss, dry cracked lips or cracks at the corners of the mouth, magenta tongue swelling, itchy and/or red eyes, sore throat, loss of lean body mass, anemia, and cataracts |
| total protein | Plasma protein, albumin, prealbumin | malnutrition, edema | |
| vitamin B12 Cobalamin | plasma cobalamin | Hyperhomocysteinemia, megaloblastic or macrocytic anemia, low energy, weakness, numbness or tingling in hands or feet, difficulty walking or instability, constipation, anorexia, confusion, poor memory, mood changes, psychosis, and mouth/tongue discomfort | |
| Iron | Plasma ferritin, the plasma iron | Microcytic anemia, pallor, weakness, fatigue or drowsiness, irritability, poor concentration, learning, cognitive difficulties, mood changes, reduced exercise endurance, headaches, temperature intolerance, weakened immune system, and reduced appetite due to mucosal changes (disappearance of tongue papillae with taste buds, reduced saliva production) | |
| Selenium | Selenium in plasma | Oxidative stress | |
| Zinc | Plasma zinc | Oxidative stress, poor growth, and development, anorexia, weakened immune system, impaired night vision, taste and smell changes, hair loss, diarrhea, and poor wound healing | |
| Fish | omega-3 acids | omega-3 acids in plasma | central nervous system disorders and cardiovascular disorders |
| vitamin D3 | plasma vitamin D3 | rickets, osteomalacia, and osteopenia | |
| Vegetables and fruits | folates | plasma folate | Hyperhomocysteinemia, megaloblastic or macrocytic anemia, persistent fatigue, pallor, palpitations, dyspnoea, headache, mouth ulcers, increased risk of birth defects, poor concentration, increased irritability, and weight loss |
| Vitamin C | Vitamin C in plasma | Microcytic anemia, scurvy, petechiae, easy bruising, bleeding, and swollen gums, anorexia, anemia, malaise, muscle, joint pains, corkscrew, perianal hemorrhage, wound healing disorders, hyperkeratosis, weakness, and mood disorders | |
| Animal and vegetable fats | Vitamin E | Vitamin E/lipids | Oxidative stress |
| Vitamin A | Plasma vitamin A | Hemeralopia, poor night vision/night blindness, weakened immune system, hyperkeratosis, and impaired wound healing | |
| Vitamin K | Plasma vitamin K | Bruising and easy bleeding and prolonged prothrombin time | |
| Fat | observation | Weight loss and absence of menstruation |
| STRONGLY DISAGREE | DISAGREE | SLIGHTLY DISAGREE | SLIGHTLY AGREE | AGREE | STRONGLY AGREE | ||
|---|---|---|---|---|---|---|---|
| 1 | I am a picky eater | □ | □ | □ | □ | □ | □ |
| 2 | I dislike most of the foods that other people eat | □ | □ | □ | □ | □ | □ |
| 3 | The lisf of foods that I like and will eat is shorter that the list of foods I won’t eat | □ | □ | □ | □ | □ | □ |
| 4 | I am not very interester in eatingl I seem to have a smaller appetite than other people | □ | □ | □ | □ | □ | □ |
| 5 | I have to push myself to eat regular meals throughout the day, or to eat a large enough amount of food at meals | □ | □ | □ | □ | □ | □ |
| 6 | Even when I am eating a food I really like, it is hard for me to eat a large enough volume at meals | □ | □ | □ | □ | □ | □ |
| 7 | I avoid of put off eating because I am afraid of GI discomfort, choking, or vomiting | □ | □ | □ | □ | □ | □ |
| 8 | I restrict myself to certain food because I am afraid that other foods will cause GI discomfort, choking, or vomiting | □ | □ | □ | □ | □ | □ |
| 9 | I eat small portion because I am afraid that other foods will cause GI discomfort, choking, or vomiting | □ | □ | □ | □ | □ | □ |
| Please Read Through the Following Statements and Check the Box that Describes You Best. |  | ||||||
|---|---|---|---|---|---|---|---|
| Never True | Always True | ||||||
| 1 | If I was allowed to, I would not eat. | □ | □ | □ | □ | □ | □ |
| 2 | Food/eating does not interest me. | □ | □ | □ | □ | □ | □ |
| 3 | I do not eat when I’m sad, worried, or anxious. | □ | □ | □ | □ | □ | □ |
| 4 | Other people think that I weigh too little. | □ | □ | □ | □ | □ | □ |
| 5 | I would like to weigh more. | □ | □ | □ | □ | □ | □ |
| 6 | I feel fat, even if other people do not agree with me. | □ | □ | □ | □ | □ | □ |
| 7 | As long as I do not look too fat or weigh too much, everything else does not matter. | □ | □ | □ | □ | □ | □ |
| 8 | I am a picky eater. | □ | □ | □ | □ | □ | □ |
| 9 | I do not like to try new food. | □ | □ | □ | □ | □ | □ |
| 10 | I am afraid of choking or vomiting while eating. | □ | □ | □ | □ | □ | □ |
| 11 | I am afraid of swallowing food. | □ | □ | □ | □ | □ | □ |
| 12 | I do not like to try food with a specific smell, taste, appearance, or a certain consistency (e.g., crispy or soft). | □ | □ | □ | □ | □ | □ |
| 13 | I like to eat things that are not meant for eating (e.g., sand). | □ | □ | □ | □ | □ | □ |
| 14 | I regurgitate food that I have already swallowed. | □ | □ | □ | □ | □ | □ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Białek-Dratwa, A.; Szymańska, D.; Grajek, M.; Krupa-Kotara, K.; Szczepańska, E.; Kowalski, O. ARFID—Strategies for Dietary Management in Children. Nutrients 2022, 14, 1739. https://doi.org/10.3390/nu14091739
Białek-Dratwa A, Szymańska D, Grajek M, Krupa-Kotara K, Szczepańska E, Kowalski O. ARFID—Strategies for Dietary Management in Children. Nutrients. 2022; 14(9):1739. https://doi.org/10.3390/nu14091739
Chicago/Turabian StyleBiałek-Dratwa, Agnieszka, Dorota Szymańska, Mateusz Grajek, Karolina Krupa-Kotara, Elżbieta Szczepańska, and Oskar Kowalski. 2022. "ARFID—Strategies for Dietary Management in Children" Nutrients 14, no. 9: 1739. https://doi.org/10.3390/nu14091739
APA StyleBiałek-Dratwa, A., Szymańska, D., Grajek, M., Krupa-Kotara, K., Szczepańska, E., & Kowalski, O. (2022). ARFID—Strategies for Dietary Management in Children. Nutrients, 14(9), 1739. https://doi.org/10.3390/nu14091739











