Abstract
Caffeine is widely consumed and generally considered safe at customary doses. How-ever, high-dose preparations available online pose a risk of severe and potentially fatal intoxication. Although uncommon, lethal caffeine poisoning is associated with profound cardiovascular and neurological toxicity. A rare case of intentional acute caffeine intoxication with fatal outcome is presented. A 25-year-old woman ingested an estimated 60 tablets containing 200 mg of caffeine each, purchased online. She was admitted to hospital shortly after ingestion of the caffeine tablets with palpitations, agitation, dizziness, and repeated vomiting. On examination, she presented with arterial hypotension (90/60 mmHg) and marked sinus tachycardia (150 beats/min), accompanied by psychomotor agitation. Her blood caffeine concentration measured by means of high-performance liquid chromatography (HPLC) was 177 µg/mL. The patient’s condition rapidly deteriorated, with the development of convulsive syndrome progressing to coma, extreme ventricular tachycardia, exotoxic shock, and toxic cardiomyopathy. Despite intensive care management, including mechanical ventilation and advanced cardiopulmonary resuscitation, the patient died several hours after admission. In conclusion, this case underscores the life-threatening potential of acute high-dose caffeine ingestion and highlights the risk associated with unrestricted access to concentrated caffeine products. Early recognition and aggressive management are crucial, yet may be insufficient in cases of massive overdose.
1. Introduction
Caffeine (1,3,7-trimethylxanthine) is the most widely consumed psychoactive substance worldwide and is generally regarded as safe when ingested at customary dietary doses [1]. Its pharmacological activity is primarily mediated through nonselective antagonism of adenosine A1 and A2A receptors, resulting in disinhibition of neuronal activity and enhanced release of excitatory neurotransmitters, including catecholamines. At higher concentrations, caffeine additionally inhibits phosphodiesterase enzymes and promotes intracellular calcium mobilization, thereby augmenting myocardial excitability and sympathetic stimulation. These mechanisms underlie its central nervous system (CNS) stimulant properties as well as its cardiovascular effects, including positive chronotropic and inotropic responses [2,3]. At moderate doses, caffeine improves alertness, vigilance, and cognitive performance. It has established medical applications, most notably as caffeine citrate in the management of apnea of prematurity, where it reduces apnea frequency and enhances respiratory drive [4,5]. Furthermore, caffeine is widely used as an adjuvant in combination with non-opioid analgesics and non-steroidal anti-inflammatory drugs, potentiating their analgesic efficacy [6].
Daily caffeine intake of up to 400 mg in healthy adults is considered safe by major regulatory authorities, including the European Food Safety Authority (EFSA) and the U.S. Food and Drug Administration (FDA). This intake roughly corresponds to four cups of brewed coffee and is not typically associated with clinically significant adverse reactions [2,7,8]. Nevertheless, even at therapeutic doses, caffeine may cause tachycardia, anxiety, tremor, gastrointestinal discomfort, and sleep disturbances [4]. In contrast, excessive caffeine exposure can result in severe toxicity. Clinical manifestations of acute caffeine poisoning include pronounced agitation, persistent vomiting, seizures, refractory ventricular tachyarrhythmias, and hemodynamic instability. Therapeutic plasma caffeine concentrations typically range between 4 and 8 µg/mL. Symptomatic toxicity is generally observed at plasma concentrations above 15–20 µg/mL, whereas levels exceeding 80–100 µg/mL have been associated with coma and fatal outcomes. Reported fatal ingestions commonly exceed 5–10 g of caffeine, although significant interindividual variability exists [9,10].
Although severe caffeine intoxication remains uncommon, its reported incidence appears to be increasing, likely reflecting the expanding availability of high-dose caffeine products, including concentrated tablets and powdered formulations marketed online. Parallel concerns have emerged regarding energy drink consumption, particularly among children and adolescents. Many commercially available products contain between 80 and 200 mg of caffeine per serving, with some exceeding 300 mg per container. These beverages are frequently consumed rapidly and may contain additional stimulants such as guarana (a supplementary caffeine source), taurine, and other sympathomimetic compounds, potentially amplifying cardiovascular and neuropsychiatric effects [11,12]. A recent systematic review of published case reports spanning more than a century identified over 200 documented cases of acute caffeine toxicity, a substantial proportion involving intentional overdose and severe cardiotoxic or neurotoxic manifestations. While many patients recover with supportive treatment, a subset develops life-threatening arrhythmias, refractory shock, and multiorgan dysfunction, underscoring the importance of early recognition and aggressive management [10].
In this report, we present a rare case of intentional acute caffeine intoxication with fatal outcome in a 25-year-old woman who ingested an unknown quantity of caffeine-containing tablets (estimated 12 g of caffeine according to the available data). This case highlights the life-threatening potential of massive caffeine overdose and emphasizes the public health risks associated with unrestricted access to concentrated caffeine products, especially among young individuals. Early recognition and aggressive management are crucial, yet may be insufficient in cases of massive overdose.
2. Detailed Case Description
2.1. Medical History
A 25-year-old woman was admitted to the Emergency Consultative Unit of the Military Medical Academy–Varna after a suicide attempt. According to the information obtained from the emergency medical services (EMS) team and partially from the patient herself, approximately one hour prior to hospital admission, the patient intentionally ingested an unknown quantity of caffeine-containing tablets (presumably 60 tablets containing 200 mg of caffeine each, according to the found bottle of tablets) with suicidal intent. It was reported that the tablets had been purchased online. After ingestion, the patient informed her sister about the incident. She experienced multiple episodes of vomiting and was subsequently transported to the hospital by an EMS team. Upon initial examination, the patient complained of palpitations, psychomotor agitation, and dizziness. The patient denied any known medical or psychiatric disorders and reported no regular medication use. However, additional information provided by her parents revealed a previous suicide attempt approximately five years earlier, involving an attempted self-strangulation. Furthermore, two years prior to the current incident, the patient had consulted a psychiatrist for depressive symptoms and had been prescribed homeopathic treatment, which reportedly resulted in clinical improvement, leading to discontinuation of therapy.
2.2. Physical Examination and Clinical Course
On admission, the patient was in moderately impaired general condition and was able to maintain an active position in bed. The patient was conscious, communicative, agitated, dysthymic, and oriented, with intermittent episodes of psychomotor agitation. Pupils were equal and miotic, with a mildly delayed light reflex.
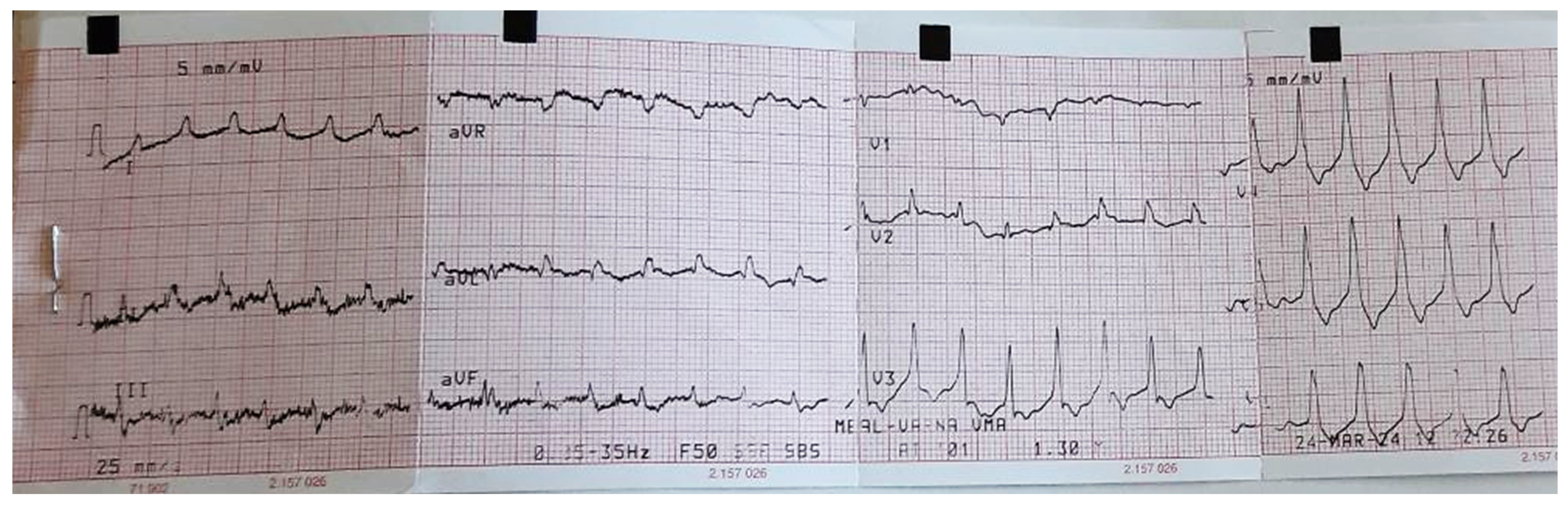
Cardiovascular examination demonstrated regular tachycardia rhythm (150 beats per minute), muffled heart sounds, and no pathological murmurs (Figure 1). Blood pressure was 90/60 mmHg.
Figure 1.
Patient’s ECG result on admission.
The exact duration over which the tablets were ingested could not be reliably established; however, based on the available history and clinical presentation, the ingestion is presumed to have occurred over a short period of time, consistent with an acute overdose. The patient’s condition on admission is described in detail in Table 1, which summarizes the patient’s clinical course by hours.

Table 1.
Clinical timeline.
Gastric lavage followed by administration of activated charcoal was performed immediately after hospital admission in an attempt to remove the ingested caffeine. Gastrointestinal decontamination represents a key early intervention in cases of acute caffeine intoxication, particularly when patients present shortly after ingestion. Activated charcoal is widely recommended due to its capacity to adsorb caffeine within the gastrointestinal tract and thereby limit further systemic absorption. Moreover, given that caffeine undergoes enterohepatic recirculation, repeated doses of activated charcoal may further enhance elimination by reducing reabsorption. Gastric lavage may be considered in life-threatening ingestions if performed within a narrow time window after exposure; however, its use remains controversial and must be carefully weighed against the potential risk of complications [9,13,14]. Despite its benefits, in massive overdoses such as the present case, gastrointestinal decontamination alone is often insufficient to prevent severe systemic toxicity due to the rapid absorption of caffeine and the large ingested dose.
2.3. Toxicological Screening and Laboratory Results
2.3.1. Urine Drug Screening
Blood and urine samples from the patient were analyzed at the Laboratory of Analytical Toxicology. A 10-panel urine drug screening test was performed (Table 2). The results were negative for all substances tested, except for benzodiazepines, for which a positive result was yielded in the urine. This finding is expected and fully consistent with intravenous diazepam administration after hospital admission. Table 2 presents toxicological findings from the urine sample.

Table 2.
Results from the 10-panel urine drug screening test (Hangzhou Alltest Biotech Co., Ltd., Hangzhou, Zhejiang Province, China).
2.3.2. Laboratory Analysis of Blood and Urine Samples
The laboratory findings are indicative of the severity and dynamic progression of the clinical course and are consistent with the underlying pathophysiological mechanisms of caffeine toxicity (Table 3). Significant metabolic and electrolyte disturbances were documented, manifested by severe hyperglycemia, hypokalemia, and lactic acidosis.

Table 3.
Monitoring of Laboratory Parameters.
The observed marked deviations of some laboratory biomarkers from the listed reference ranges highlight the severity of intoxication.
2.3.3. Toxicological Analysis of Blood Sample
High-performance liquid chromatography (HPLC) analysis was performed using an Agilent 1260 Infinity Binary LC system equipped with a Zorbax Extend-C18 column (150 × 4.6 mm, 5 μm) and a 1260 Infinity diode array detector (DAD) for quantitative determination of caffeine.
The pre-analytical procedure began with 500 μL of plasma sample, alkalinized with 500 μL of 1 M NaOH. Deproteinization was achieved by the addition of 1.5 mL acetonitrile, followed by a two-step liquid–liquid extraction using 2 × 3 mL ethyl acetate. The organic phase was evaporated to dryness, and the residue was reconstituted in 500 μL of mobile phase and filtered through a 0.22 μm nylon syringe filter prior to analysis. Chromatographic separation was carried out under isocratic conditions using a methanol–water mobile phase (25:75, v/v) at 25 °C with a flow rate of 1.0 mL/min. The injection volume was 20 μL. Detection was performed at a wavelength of 274 nm.
Data acquisition and processing were carried out using the Agilent OpenLAB (ChemStation edition, rev. C.01.05) and MassHunter (rev. B.07.00)software package. All chemicals and reagents were of analytical grade or higher. Mobile phases and standard solutions were prepared using HPLC-grade solvents and purified deionized water (0.067–0.100 μS cm−1) obtained from a TKA™ Pacific water purification system.
Under the described chromatographic conditions, the retention time of caffeine was approximately 5.02 min. The measured plasma caffeine concentration was 177 µg/mL. The result of the HPLC analysis was ready on the next day at 10:59 AM and was immediately reported to the Department of Toxicology.
2.4. Diagnosis
The patient was diagnosed with severe acute caffeine intoxication (ICD-10 code T43.6) as a result of intentional ingestion of caffeine tablets. The clinical course was complicated by cerebrototoxic syndrome, marked psychomotor agitation, and status epilepticus progressing to coma. The patient developed exotoxic shock, extreme ventricular tachycardia, and toxic cardiomyopathy.
She underwent endotracheal intubation and invasive mechanical ventilation, as well as advanced cardiopulmonary resuscitation. Despite full intensive care support, the clinical course was refractory, and a fatal outcome was recorded approximately six hours after admission to the hospital. No autopsy was performed.
2.5. Therapeutic Course
The therapeutic course during the patient’s hospitalization is presented in Table 4 by hours. It reflects a multidisciplinary approach focused on stabilizing the patient’s condition as well as preventing complications and fatal outcome.

Table 4.
Patient’s therapeutic course and interventions by hours.
3. Discussion
The present case illustrates a fulminant course of massive caffeine intoxication characterized by rapidly progressive cardiovascular collapse, refractory ventricular tachyarrhythmia, severe metabolic acidosis, and toxic cardiomyopathy. The measured plasma caffeine concentration of 177 µg/mL, confirmed by means of HPLC analysis, is markedly above levels commonly associated with fatal outcomes (>80–100 µg/mL), providing objective toxicological confirmation of extreme systemic exposure [10,15]. Therefore, the biochemical data corroborate the clinical severity and support the causal relationship between massive caffeine ingestion and the fatal outcome.
Under physiological conditions, caffeine is rapidly and almost completely absorbed from the gastrointestinal tract, with peak plasma concentrations typically occurring within 30–120 min. It is primarily metabolized in the liver by cytochrome P450 1A2 (CYP1A2), with an elimination half-life of approximately 3–7 h in healthy adults. However, in massive overdose, caffeine metabolism may become saturated, resulting in non-linear kinetics and significant prolongation of elimination half-life [3,9,16]. Omi (2021) reported that in severe intoxication, the half-life may extend beyond 15 h due to metabolic pathway saturation [17]. This pharmacokinetic feature may partly explain the rapid escalation of toxicity observed in massive ingestions, as sustained high serum concentrations perpetuate adrenergic stimulation, intracellular calcium overload, and progressive myocardial instability. In the present case, the measured plasma caffeine concentration of 177 µg/mL strongly suggests metabolic saturation, as such levels far exceed the threshold typically associated with severe toxicity.
In the present case, the early clinical presentation of marked sinus tachycardia, hypotension, agitation, vomiting, and subsequent progression to ventricular tachycardia (VT) is consistent with profound sympathetic overstimulation. At toxic concentrations, caffeine exerts competitive antagonism of adenosine A1- and A2A-receptors, inhibits phosphodiesterase enzymes, and enhances intracellular calcium release through ryanodine receptor activation. The resulting catecholamine surge and calcium overload increase myocardial automaticity and triggered activity, predisposing to malignant ventricular arrhythmias. Thus, malignant arrhythmias may be refractory to standard antiarrhythmic therapy even when administered promptly and appropriately [2,3,9]. Severe hypokalemia likely further amplified arrhythmogenic susceptibility. β2-adrenergic stimulation induces intracellular potassium shift, while vomiting and osmotic diuresis contribute to additional losses. Additionally, hypokalemia shortens repolarization and promotes re-entry mechanisms, lowering the threshold for sustained VT [18]. Concurrent severe metabolic acidosis reflects systemic hypoperfusion and β-adrenergic-mediated hyperlactatemia, both recognized features of severe methylxanthine toxicity [9,15,19]. Moreover, at extreme concentrations, caffeine disrupts intracellular calcium homeostasis within cardiomyocytes, impairing excitation–contraction coupling and contributing to acute toxic cardiomyopathy and hemodynamic collapse [2,3]. Elevated intracellular calcium may contribute to triggered activity and complex arrhythmogenic substrates, as evidenced by reported bidirectional VT in heavy caffeine poisoning [20]. The progression from VT to asystole, with only transient return of spontaneous circulation despite advanced cardiopulmonary resuscitation, suggests profound electrical instability rather than primary hypoxic arrest. This pattern has been described in severe caffeine intoxication cases complicated by refractory ventricular arrhythmias and shock [10,21].
Management of severe caffeine intoxication remains primarily supportive and symptom-directed, as there is no universally accepted antidote or standardized treatment protocol due to the rarity of such cases. Initial therapy focuses on stabilization of airway, breathing, and circulation, correction of metabolic derangements such as acidosis and electrolyte imbalances, and control of seizures and arrhythmias with appropriate anticonvulsants and cardiovascular support agents. Beta-adrenergic blockade may be considered for persistent tachyarrhythmias, while correction of hypotension often requires vasoactive support such as dopamine infusion. Continuous monitoring in an intensive care setting is essential given the risk of rapid deterioration [9,22]. Adjunctive measures such as forced diuresis and enhanced elimination have been described in case reports, reflecting attempts to augment toxin clearance; however, the evidence base consists predominantly of individual cases rather than controlled trials [9].
In the management of severe caffeine intoxication complicated by malignant ventricular arrhythmias, antiarrhythmic therapy is foundational, yet its efficacy may be limited by the underlying pathophysiology of methylxanthine toxicity [9,15]. In this case, the patient was treated with intravenous amiodarone and magnesium sulfate, consistent with advanced cardiac life support strategies for refractory ventricular arrhythmias. Amiodarone’s multi-channel blocking effects can suppress ventricular ectopy and reentrant circuits, while magnesium stabilizes cardiomyocyte membranes and may blunt catecholamine-mediated excitability [23,24]. However, case series and toxicology reviews indicate that severe caffeine-induced ventricular arrhythmias can be refractory to standard antiarrhythmic interventions, likely due to persistent adrenergic stimulation and intracellular calcium overload that diminish the effectiveness of pharmacologic suppression. One case report describes persistent ventricular fibrillation following massive caffeine overdose that required prolonged supportive care and ultimately responded only after extracorporeal support modalities were initiated [23]. In our case, guideline-based antiarrhythmic therapy was initiated, including bolus and continuous intravenous amiodarone and intravenous magnesium sulfate to address myocardial electrical instability and catecholamine-mediated arrhythmogenicity [25]. This highlights a mechanistic limitation of conventional antiarrhythmics in the setting of profound methylxanthine toxicity.
Considering the mechanisms of caffeine toxicity, β-adrenergic blockade represents a rational therapeutic approach, as it directly counteracts catecholamine-mediated cardiotoxicity. Several toxicology reviews support the use of β-blockers in cases of severe caffeine intoxication complicated by refractory tachyarrhythmias [9,26,27]. In the present case, bisoprolol was administered. Despite aggressive antiarrhythmic management and the administration of magnesium sulfate and bisoprolol, the arrhythmia proved refractory and was accompanied by progressive hemodynamic instability requiring cardiopulmonary resuscitation and mechanical ventilation.
In addition to cardiovascular stabilization and antiarrhythmic therapy, the patient received comprehensive supportive treatment, including benzodiazepine administration, electrolyte correction, vitamin supplementation, and neuroprotective measures. The use of diazepam is consistent with current toxicology recommendations for the management of agitation and seizure activity in caffeine intoxication, as benzodiazepines are considered first-line agents for methylxanthine-induced CNS hyperexcitability. Correction of electrolyte disturbances is particularly important in severe caffeine poisoning, as hypokalemia and metabolic derangements contribute significantly to arrhythmogenesis and myocardial instability [9]. Supportive adjuncts such as B-group vitamins and neuroprotective agents are frequently employed in critical care settings to mitigate secondary metabolic and neurologic injury, particularly in patients with convulsive syndromes and evolving cerebral toxicity. Although direct evidence for outcome modification in caffeine poisoning is limited, their use aligns with established principles of neuroprotective supportive care in toxic encephalopathy [28,29].
Extracorporeal elimination techniques, particularly hemodialysis and hemoperfusion, have been reported in severe or life-threatening caffeine overdoses and may be effective in reducing serum concentrations. Caffeine possesses several physicochemical properties that make it amenable to extracorporeal removal, such as low molecular weight (194 Da), relatively low protein binding (≈15–35%), small volume of distribution (~0.6 L/kg), and water solubility [30]. In published case series, hemodialysis has been associated with clinical improvement and reduced toxin levels, although timing and indications remain case-specific rather than guideline-driven [31,32]. One such report demonstrates venoarterial extracorporeal membrane oxygenation (ECMO) support in the context of caffeine-induced ventricular fibrillation with subsequent favorable outcome [33]. In the present case, extracorporeal elimination was not pursued. The rapid clinical deterioration with early onset of asystole likely precluded the time necessary for vascular access and initiation of dialysis or ECMO. This underscores a major practical limitation in caffeine poisoning management, as even theoretically effective treatments may not be implementable.
Intravenous lipid emulsion (ILE) has been proposed as an adjunctive therapy in cases of severe drug intoxication, particularly for highly lipophilic agents, based on the “lipid sink” theory. Although caffeine is only moderately lipophilic and exhibits relatively low protein binding, isolated case reports have described the use of ILE in massive caffeine poisoning complicated by refractory cardiovascular instability [34]. Kohl et al. (2020) discuss the potential role of lipid emulsion therapy in non-local anesthetic toxicities, noting that evidence outside classical lipophilic cardiotoxins remains limited and largely anecdotal [35]. Similarly, Muraro et al. (2016) emphasize that lipid emulsion therapy should be considered a rescue intervention when conventional resuscitative measures fail, particularly in life-threatening cardiotoxicity [36]. Controlled clinical data, however, remain lacking. In the present case, ILE was also administered to the patient.
An overall comparison with previously reported cases of severe caffeine intoxication reveals several consistent clinical patterns. Most fatal or near-fatal cases involve ingestion of large quantities of caffeine in the form of tablets or powder, frequently exceeding 5–10 g, and are characterized by rapid onset of tachyarrhythmias, central nervous system excitation, and metabolic derangements. Ventricular tachycardia and ventricular fibrillation are among the most commonly reported life-threatening complications [9,10,15]. A systematic review by Uehlein et al. (2025) identified more than 200 published cases of acute caffeine toxicity, with a significant proportion presenting with malignant arrhythmias and hemodynamic instability. Importantly, cases with plasma caffeine concentrations exceeding 100 µg/mL were strongly associated with severe outcomes, including cardiac arrest and death [10].
Despite these common features, the available literature demonstrates considerable heterogeneity in both clinical course and therapeutic response, reflecting differences in ingested dose, timing of intervention, and availability of advanced supportive measures. Similar to our patient, several case reports describe refractory ventricular arrhythmias despite aggressive antiarrhythmic therapy [21,34,35,36,37,38,39]. Unlike our case, Bioh et al. (2013) performed continuous venovenous hemodiafiltration, which, according to the authors, contributed to the patient’s survival [38]. Additionally, Elbokl et al. (2021) reported the use of prolonged hemodialysis for caffeine elimination, facilitating patient recovery [30]. Moreover, Harsten et al. (2020) demonstrated that the combined use of intravenous lipid emulsion and hemodialysis may represent an effective strategy in life-threatening caffeine poisoning [34]. However, Han et al. (2022) as well as Pina Cabral et al. (2022) reported successful outcomes with supportive care alone, without the use of active elimination methods such as hemodialysis or ECMO [21,39].
This variability highlights a critical gap in the current evidence base, as treatment decisions are largely guided by case reports rather than standardized protocols. In particular, the timing of extracorporeal elimination appears to be a key determinant of outcome, with favorable results more often reported when such interventions are initiated before the onset of profound hemodynamic collapse. In contrast, rapidly progressive toxicity, as observed in the present case, may preclude the timely implementation of these potentially life-saving measures.
Although rare, fatal outcomes following caffeine ingestion have been documented in association with a variety of formulations, including tablets, powders, energy drinks, and concentrated caffeine products. Intentional overdoses involving caffeine tablets or bulk powder are the most commonly reported causes of lethal intoxication, often due to the ease of access to high-dose preparations via online markets [40]. Case series and individual reports indicate that fatal intoxication typically presents with sudden cardiac arrest, seizures, and malignant arrhythmias, frequently occurring shortly after ingestion of large quantities of caffeine. In a series of fatal cases, postmortem blood concentrations commonly exceeded 80 mg/L, with deaths primarily attributed to arrhythmogenic mechanisms [41,42,43,44]. More recent reports confirm that fatal outcomes continue to occur, including a case involving combined caffeine and methamphetamine intoxication, highlighting that although rare, caffeine-related deaths remain consistently documented in the literature [45].
Overall, the available evidence indicates that while moderate caffeine consumption is generally safe, high-dose exposure carries a substantial risk of life-threatening toxicity and sudden cardiac death.
In conclusion, the present case illustrates the fulminant clinical course that may follow massive caffeine ingestion and emphasize that therapeutic success may be limited by rapid toxic progression and refractory cardiovascular instability. The case further highlights the importance of early consideration of extracorporeal removal techniques in patients with markedly elevated serum concentrations of caffeine, provided that clinical conditions permit timely initiation.
Finally, an additional noteworthy aspect in this case is the patient’s documented history of depressive symptoms and a prior suicide attempt, for which psychiatric consultation had been sought. According to available information, treatment consisted solely of homeopathic preparations. It is important to emphasize that current evidence does not support the clinical effectiveness of homeopathy in the management of depression [46]. Untreated or inadequately treated depression is a well-established risk factor for suicidal behavior, and failure to implement evidence-based psychiatric management may increase vulnerability to future suicide attempts. In this context, reliance on non-evidence-based therapeutic approaches may represent a missed opportunity for effective intervention and long-term risk reduction.
4. Conclusions
Acute massive caffeine ingestion can result in rapidly progressive, life-threatening cardiotoxicity and neurotoxicity, even when prompt and aggressive supportive treatment is provided. The present case underscores the potential for fatal outcome at extremely elevated plasma concentrations and highlights the limitations of current therapeutic options in fulminant intoxication. Early recognition, intensive monitoring, and timely consideration of extracorporeal elimination strategies are essential, although they may not always be feasible in rapidly deteriorating patients. Furthermore, the unrestricted availability of high-dose caffeine products, including energy drinks, represents an emerging public health concern and underscores the need for greater awareness and regulatory oversight.
Author Contributions
Conceptualization, M.R.-I., S.S.-G., I.Y., G.B., S.Z. and P.M.; methodology, M.R.-I., S.S.-G., I.Y., I.P., N.H., G.B., S.M., P.M. and S.Z.; software, N.H., S.S.-G. and I.Y.; validation, M.R.-I., S.S.-G. and I.Y.; formal analysis, S.S.-G., I.Y., I.P., G.B. and S.M.; investigation, M.R.-I., S.S.-G., I.Y., I.P., G.B., N.H. and S.M.; resources, N.H., M.M., P.M. and S.Z.; data curation, S.S.-G. and I.Y.; writing—original draft preparation, M.R.-I. and S.S.-G.; writing—review and editing, M.R.-I., S.S.-G. and S.Z.; visualization, N.H., S.S.-G. and I.Y.; supervision, G.B., P.M. and S.Z.; project administration, M.R.-I., S.S.-G., M.M. and P.M.; funding acquisition, M.M., P.M. and S.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the European Union–NextGenerationEU through the National Recovery and Resilience Plan of the Republic of Bulgaria [project No. BG-RRP-2.004-0009-C02, Research Group 3.2.2. GOSACD, which is part of the MUVE-TEAM project No. BG-RRP-2.004-0009-C02].
Institutional Review Board Statement
This study is retrospective and relies solely on medical data that had already been collected for other clinical purposes. Therefore, no approval was required from an ethics committee or an institutional review board, as no new interventions were performed on patients and all data were obtained from existing medical records.
Informed Consent Statement
Informed consent was not applicable, as the subject was deceased and all data were anonymized. Informed consent was obtained from the patient’s family to obtain her clinical history.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| HPLC | High-Performance Liquid Chromatography |
| CNS | Central Nervous System |
| EFSA | European Food Safety Authority |
| FDA | Food and Drug Administration |
| EMS | Emergency Medical Services |
| ICU | Intensive Care Unit |
| VT | Ventricular Tachycardia |
| ECMO | Extracorporeal Membrane Oxygenation |
| ILE | Intravenous Lipid Emulsion |
References
- Rodak, K.; Kokot, I.; Kratz, E.M. Caffeine as a Factor Influencing the Functioning of the Human Body—Friend or Foe? Nutrients 2021, 13, 3088. [Google Scholar] [CrossRef]
- Temple, J.L.; Bernard, C.; Lipshultz, S.E.; Czachor, J.D.; Westphal, J.A.; Mestre, M.A. The Safety of Ingested Caffeine: A Comprehensive Review. Front. Psychiatry 2017, 8, 80. [Google Scholar] [CrossRef]
- Nehlig, A. Interindividual Differences in Caffeine Metabolism and Factors Driving Caffeine Consumption. Pharmacol. Rev. 2018, 70, 384–411. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.; Richards, J.R.; Battisti, A.S. Caffeine. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK519490/ (accessed on 21 February 2026).
- Schmidt, B.; Roberts, R.S.; Davis, P.; Doyle, L.W.; Barrington, K.J.; Ohlsson, A.; Solimano, A.; Tin, W.; Caffeine for Apnea of Prematurity Trial Group. Long-term effects of caffeine therapy for apnea of prematurity. N. Engl. J. Med. 2007, 357, 1893–1902. [Google Scholar] [CrossRef]
- Derry, C.J.; Derry, S.; Moore, R.A. Caffeine as an analgesic adjuvant for acute pain in adults. Cochrane Database Syst. Rev. 2014, 2014, CD009281. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on the safety of caffeine. EFSA J. 2015, 13, 4102. [Google Scholar] [CrossRef]
- Wikoff, D.; Welsh, B.T.; Henderson, R.; Brorby, G.P.; Britt, J.; Myers, E.; Goldberger, J.; Lieberman, H.R.; O’Brien, C.; Peck, J.; et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem. Toxicol. 2017, 109, 585–648. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.; Traylor, J. Caffeine Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK532910/?utm_source=chatgpt.com (accessed on 21 February 2026).
- Uehlein, S.; Dechant, K.; Stahl, K.; Schneider, R.; Wedemeyer, H.; Schäfer, A. Caffeine Intoxication: An Analysis of Published Case Reports, 1883–2023. Dtsch. Arztebl. Int. 2025, 122, 523–528. [Google Scholar] [CrossRef]
- Medrano-Sanchez, E.J.; Gutierrez-Berrocal, C.A.; Gonzales-Aguilar, L.C.; Huaman, M.A.; Monteza, K.C.; Ayllon, M.L. Energy Drinks and Cardiovascular Health: A Critical Review of Recent Evidence. Beverages 2026, 12, 4. [Google Scholar] [CrossRef]
- Li, P.; Haas, N.A.; Dalla-Pozza, R.; Jakob, A.; Oberhoffer, F.S.; Mandilaras, G. Energy Drinks and Adverse Health Events in Children and Adolescents: A Literature Review. Nutrients 2023, 15, 2537. [Google Scholar] [CrossRef]
- Silberman, J.; Galuska, M.A.; Taylor, A. Activated Charcoal. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2026. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482294/ (accessed on 18 March 2026).
- Ajjampur, K.; Subramaniam, A. The importance of early use of beta blockers and gastric decontamination in caffeine overdose: A case report. Aust. Crit. Care 2021, 34, 395–400. [Google Scholar] [CrossRef]
- Willson, C. The clinical toxicology of caffeine: A review and case study. Toxicol. Rep. 2018, 5, 1140–1152. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US) Committee on Military Nutrition Research. Pharmacology of Caffeine. In Caffeine for the Sustainment of Mental Task Performance: Formulations for Military Operations; National Academies Press: Washington, DC, USA, 2001. Available online: https://www.ncbi.nlm.nih.gov/books/NBK223808/ (accessed on 21 February 2026).
- Omi, T. Protracted effects of caffeine after hemodialysis for severe caffeine intoxication. Toxicol. Commun. 2021, 5, 73–75. [Google Scholar] [CrossRef]
- Osadchii, O.E. Mechanisms of hypokalemia-induced ventricular arrhythmogenicity. Fundam. Clin. Pharmacol. 2010, 24, 547–559. [Google Scholar] [CrossRef]
- Baddam, S.; Tubben, R.E. Lactic Acidosis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470202/ (accessed on 24 February 2026).
- Toya, N.; Isokawa, S.; Suzuki, A.; Otani, N.; Ishimatsu, S. Bidirectional ventricular tachycardia induced by caffeine poisoning. Am. J. Emerg. Med. 2019, 37, 2118.e1–2118.e3. [Google Scholar] [CrossRef]
- Han, K.; You, K.M.; Jung, J.H. A case of refractory ventricular fibrillation after caffeine poisoning successfully treated by supportive care. Toxicol. Rep. 2022, 9, 1710–1712. [Google Scholar] [CrossRef] [PubMed]
- Papadakis, M.A.; Rabow, M.W.; McQuaid, K.R.; Nadler, P.L.; Price, E.L. Poisoning. In Current Medical Diagnosis & Treatment, 2026; Nadler, P.L., Price, E.L., Eds.; McGraw Hill: New York, NY, USA, 2026; Available online: https://accessmedicine.mhmedical.com/content.aspx?bookid=3594§ionid=299555250 (accessed on 21 February 2026).
- Van Herendael, H.; Dorian, P. Amiodarone for the treatment and prevention of ventricular fibrillation and ventricular tachycardia. Vasc. Health Risk Manag. 2010, 6, 465–472. [Google Scholar] [CrossRef]
- Baker, W.L. Treating arrhythmias with adjunctive magnesium: Identifying future research directions. Eur. Heart J. Cardiovasc. Pharmacother. 2017, 3, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Panchal, A.R.; Bartos, J.A.; Cabañas, J.G.; Donnino, M.W.; Drennan, I.R.; Hirsch, K.G.; Kudenchuk, P.J.; Kurz, M.C.; Lavonas, E.J.; Morley, P.T.; et al. Part 3: Adult Basic and Advanced Life Support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2020, 142, S366–S468. [Google Scholar] [CrossRef]
- Miyamura, Y.; Takei, T.; Suzuki, T.; Tachibana, T.; Sasamoto, I. Successful β-blocker usage to treat a patient with hemodynamic instability caused by severe caffeine poisoning. Resusc. Plus 2024, 20, 100749. [Google Scholar] [CrossRef]
- Voskoboinik, A.; Kalman, J.M.; Kistler, P.M. Caffeine and Arrhythmias: Time to Grind the Data. JACC Clin. Electrophysiol. 2018, 4, 425–432. [Google Scholar] [CrossRef]
- Calderón-Ospina, C.A.; Nava-Mesa, M.O. B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS Neurosci. Ther. 2020, 26, 5–13. [Google Scholar] [CrossRef]
- Grieb, P. Neuroprotective properties of citicoline: Facts, doubts and unresolved issues. CNS Drugs 2014, 28, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Elbokl, M.; Randall, I.; Lok, C. Severe Caffeine Intoxication Treated with Hemodialysis: A Case Report. Kidney Med. 2021, 3, 299–302. [Google Scholar] [CrossRef]
- Gahona, C.C.T.; Bharadwaj, A.K.; Shah, M.; Bhagat, U.; Sterman, P.; Vasquez, W. Treatment of Lethal Caffeine Overdose with Haemodialysis: A Case Report and Review. J. Crit. Care Med 2022, 8, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Ishigaki, S.; Fukasawa, H.; Kinoshita-Katahashi, N.; Yasuda, H.; Kumagai, H.; Furuya, R. Caffeine Intoxication Successfully Treated by Hemoperfusion and Hemodialysis. Intern. Med. 2014, 53, 2745–2747. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Hisamura, M.; Hirano, T.; Kukihara, Y.; Kodama, K.; Konishi, K.; Nakamura, M.; Hiramatsu, G.; Koshimizu, K. Caffeine poisoning successfully treated by venoarterial extracorporeal membrane oxygenation and emergency hemodialysis. Acute Med. Surg. 2020, 8, e627. [Google Scholar] [CrossRef]
- Harsten, R.; Tetlow, S.J.; Chan, T.; Ankuli, A. Intralipid and haemodialysis in caffeine overdose. BMJ Case Rep. 2020, 13, e234256. [Google Scholar] [CrossRef]
- Kohl, B.A.; Kaur, K.; Dincher, N.; Schumann, J.; Carachilo, T.; Komurek, C. Acute intentional caffeine overdose treated preemptively with hemodialysis. Am. J. Emerg. Med. 2020, 38, 692.e1–692.e3. [Google Scholar] [CrossRef]
- Muraro, L.; Longo, L.; Geraldini, F.; Bortot, A.; Paoli, A.; Boscolo, A. Intralipid in acute caffeine intoxication: A case report. J. Anesth. 2016, 30, 895–899. [Google Scholar] [CrossRef]
- Riku, S.; Yamamoto, T.; Kubota, Y.; Ichii, T.; Jinno, Y.; Suzuki, S.; Murohara, T. Refractory ventricular fibrillation caused by caffeine intoxication. J. Cardiol. Cases 2018, 18, 210–212. [Google Scholar] [CrossRef] [PubMed]
- Bioh, G.; Gallagher, M.M.; Prasad, U. Survival of a highly toxic dose of caffeine. BMJ Case Rep. 2013, bcr2012007454. [Google Scholar] [CrossRef]
- Pina Cabral, J.; Sousa, D.L.; Carvalho, C.; Girao, A.; Pacheco Mendes, A.; Pina, R. Caffeine Intoxication: Unregulated, Over-the-Counter Sale of Potentially Deadly Supplements. Cureus 2022, 14, e21045. [Google Scholar] [CrossRef]
- Cappelletti, S.; Piacentino, D.; Fineschi, V.; Frati, P.; Cipolloni, L.; Aromatario, M. Caffeine-Related Deaths: Manner of Deaths and Categories at Risk. Nutrients 2018, 10, 611. [Google Scholar] [CrossRef]
- Banerjee, P.; Ali, Z.; Levine, B.; Fowler, D.R. Fatal caffeine intoxication: A series of eight cases from 1999 to 2009. J. Forensic Sci. 2014, 59, 865–868. [Google Scholar] [CrossRef]
- Cannon, M.E.; Cookie, C.T.; McCarthy, J.S. Caffeine-induced cardiac arrhythmia: An unrecognised danger of healthfood products. Med. J. Aust. 2001, 174, 520–521. [Google Scholar]
- Ishikawa, T.; Yuasa, I.; Endoh, M. Non specific drug distribution in an autopsy case report of fatal caffeine intoxication. Leg. Med. 2015, 17, 535–538. [Google Scholar] [CrossRef]
- Magdalan, J.; Zawadzki, M.; Skowronek, R.; Czuba, M.; Porębska, B.; Sozański, T.; Szpot, P. Nonfatal and fatal intoxications with pure caffeine—Report of three different cases. Forensic Sci. Med. Pathol. 2017, 13, 355–358. [Google Scholar] [CrossRef]
- Kim, D.G.; Powers, S.; Gitto, L. Death Due to Caffeine and Methamphetamine Toxicity: A Case Report. Cureus 2024, 16, e66634. [Google Scholar] [CrossRef] [PubMed]
- Pilkington, K.; Kirkwood, G.; Rampes, H.; Fisher, P.; Richardson, J. Homeopathy for depression: A systematic review of the research evidence. In Database of Abstracts of Reviews of Effects (DARE): Quality-Assessed Reviews; Centre for Reviews and Dissemination (UK): York, UK, 2005. Available online: https://www.ncbi.nlm.nih.gov/books/NBK71860/ (accessed on 24 February 2026).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.