Predicting the Temperature Evolution during Nanomilling of Drug Suspensions via a Semi-Theoretical Lumped-Parameter Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Wet Stirred Media Milling
2.2.2. Formulation of the Lumped-Parameter Model (LPM)
2.2.3. Fits by the LPM and Predictions by the LPM Augmented with the PL and ML Models
3. Results and Discussion
3.1. Properties of the Milled Suspensions and Particles
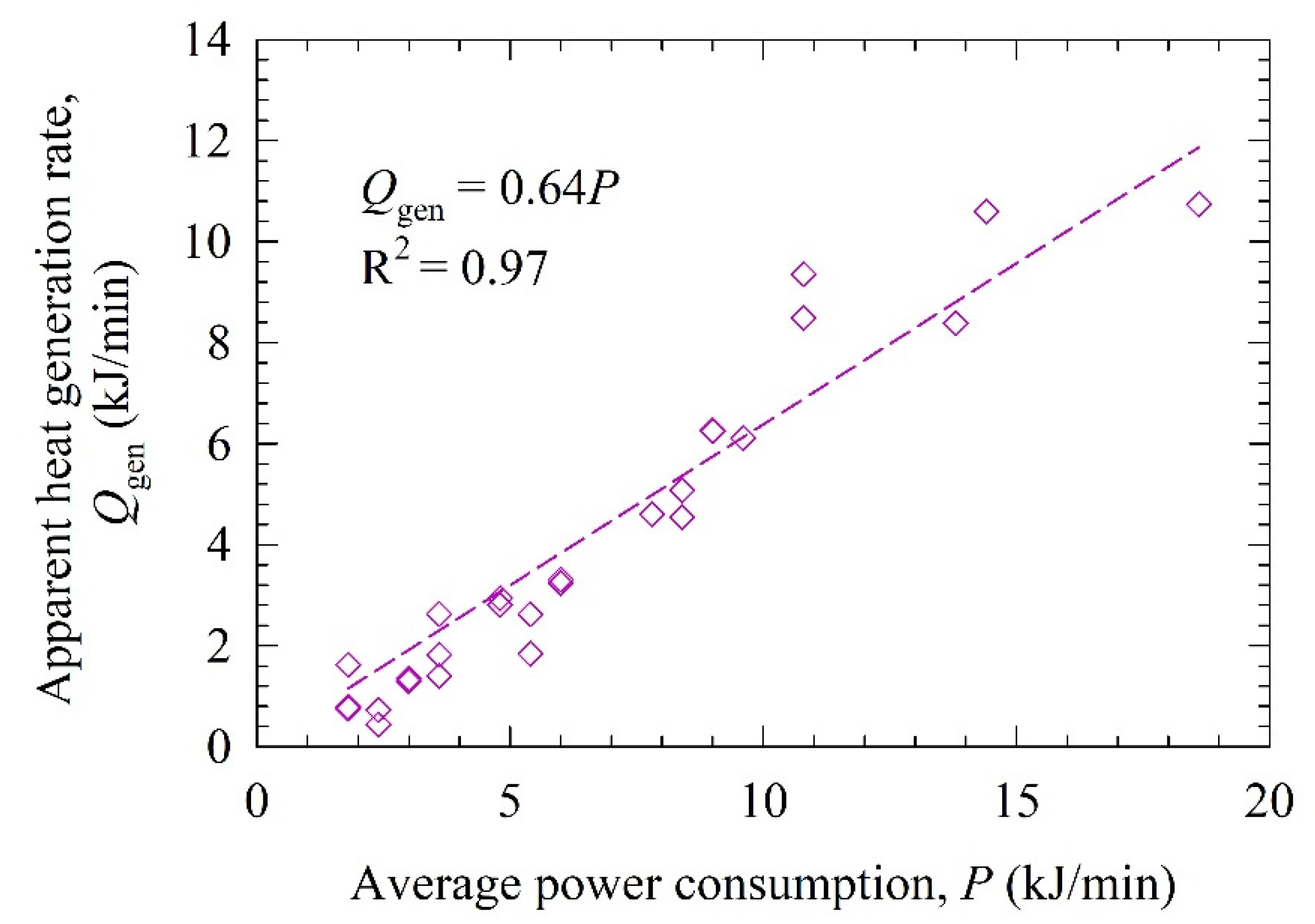
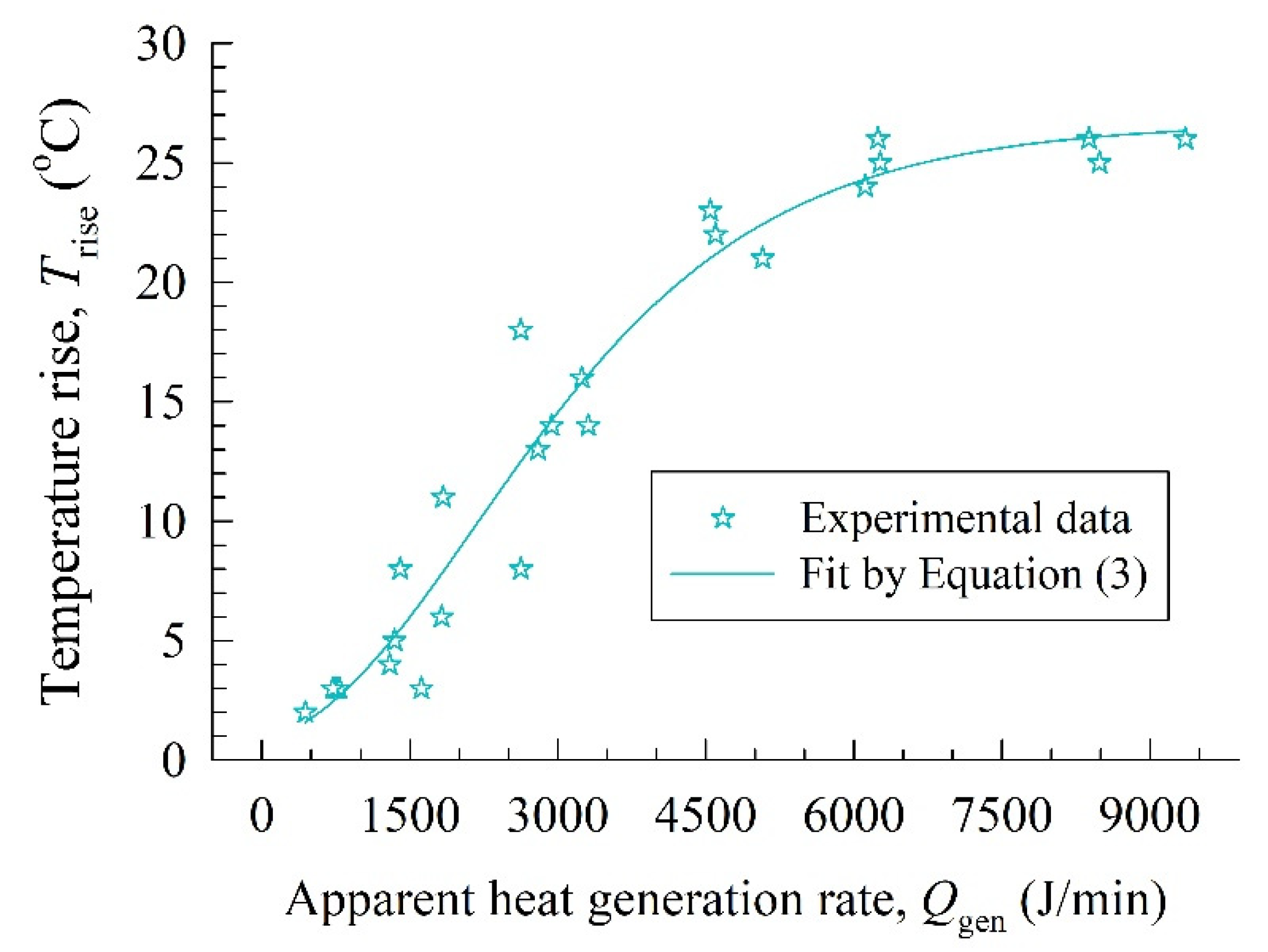
3.2. Fitted LPM Parameters and the Origin of Temperature Rise during the Milling
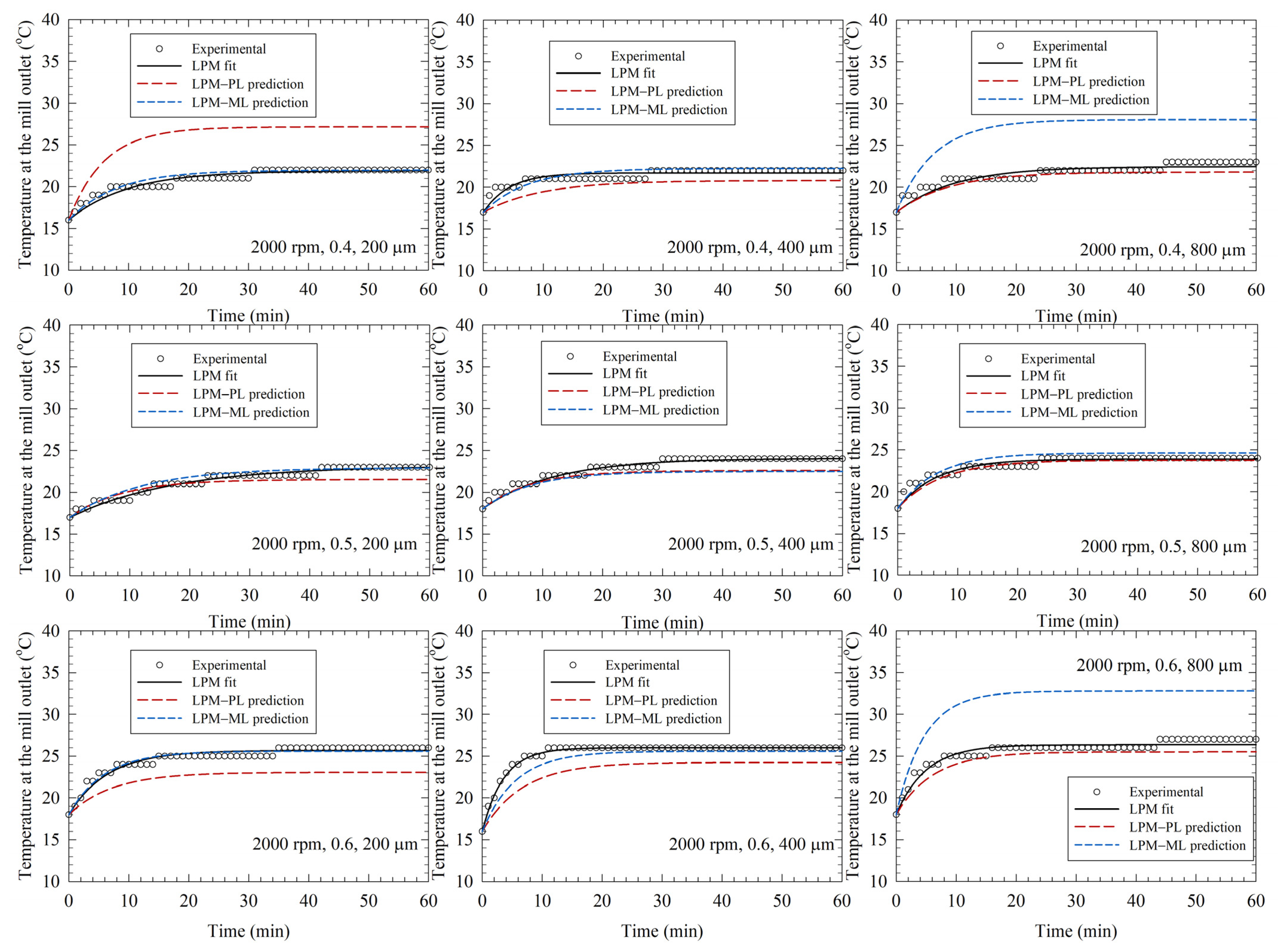
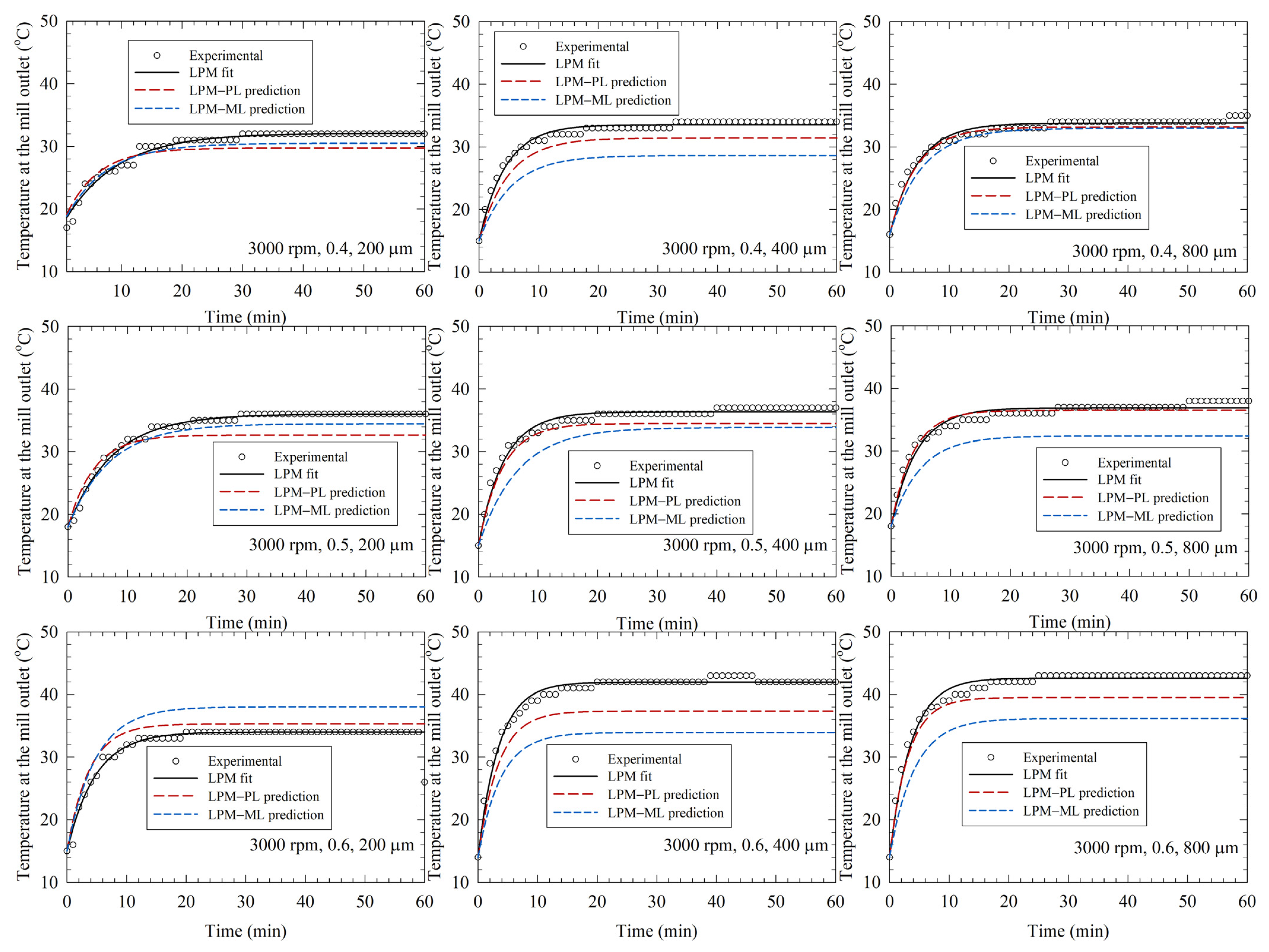
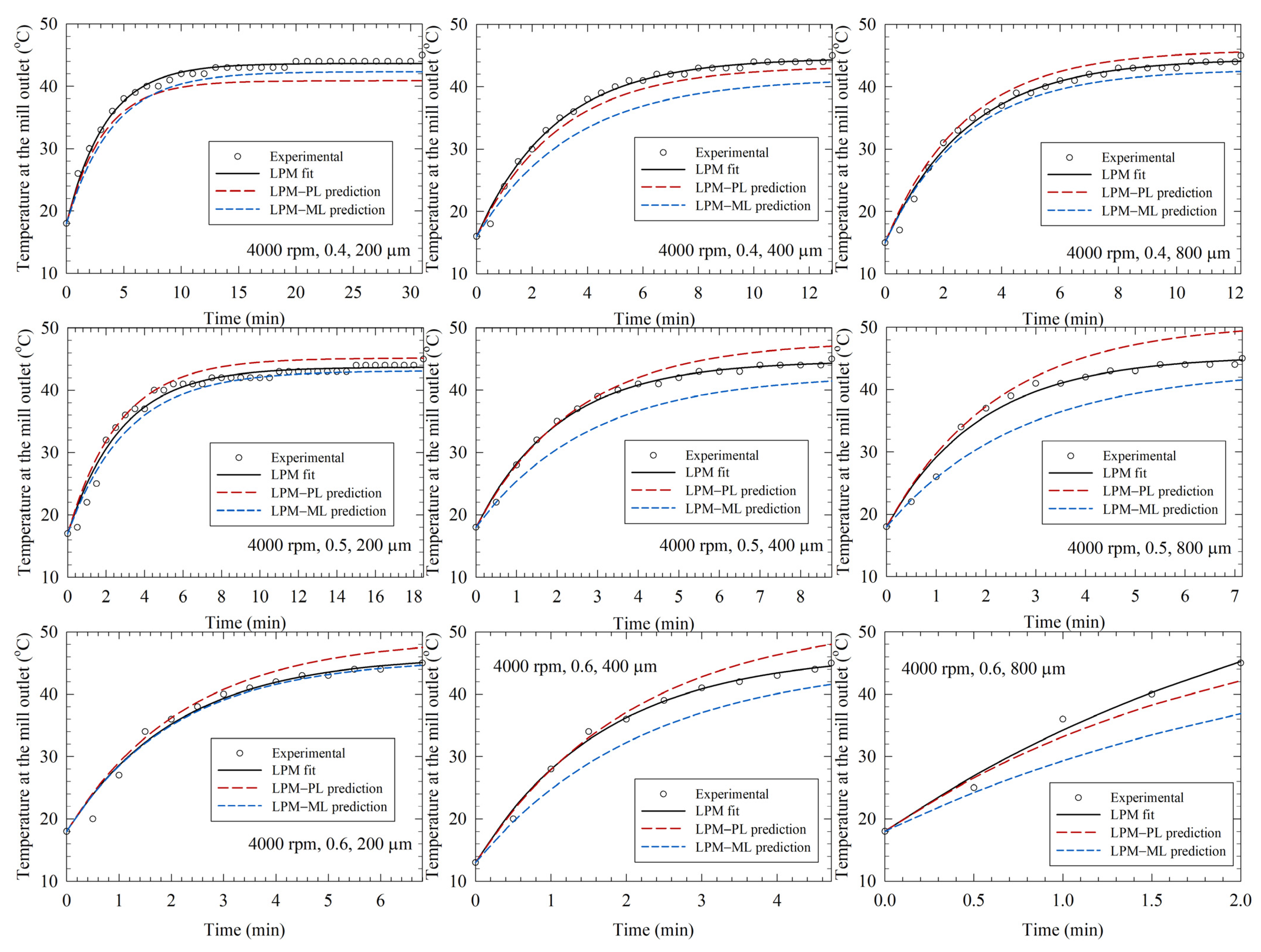
3.3. LPM-Fitted Temperature Profiles and LPM–PL/LPM-ML Predictions in the Training Runs
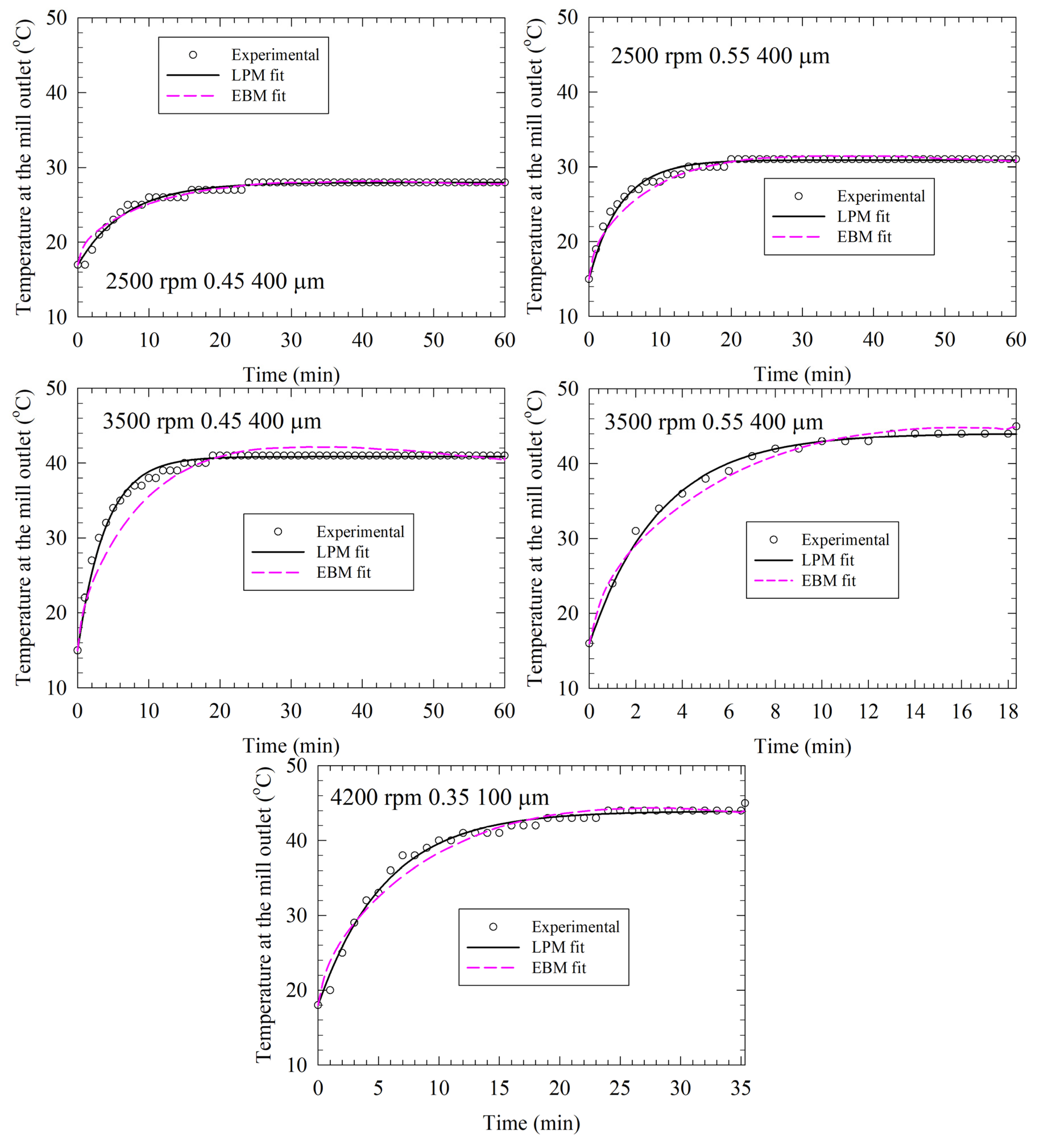
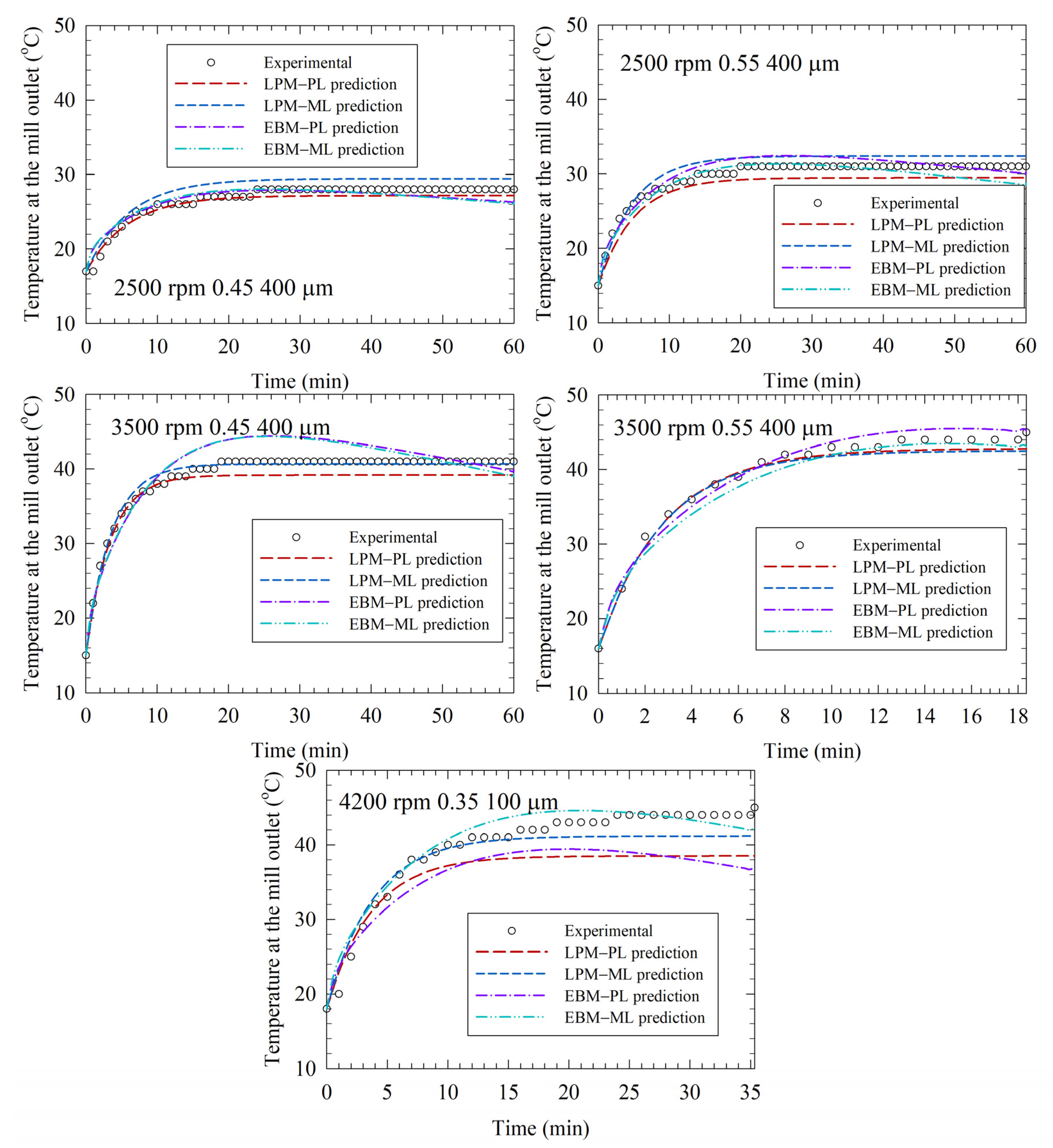
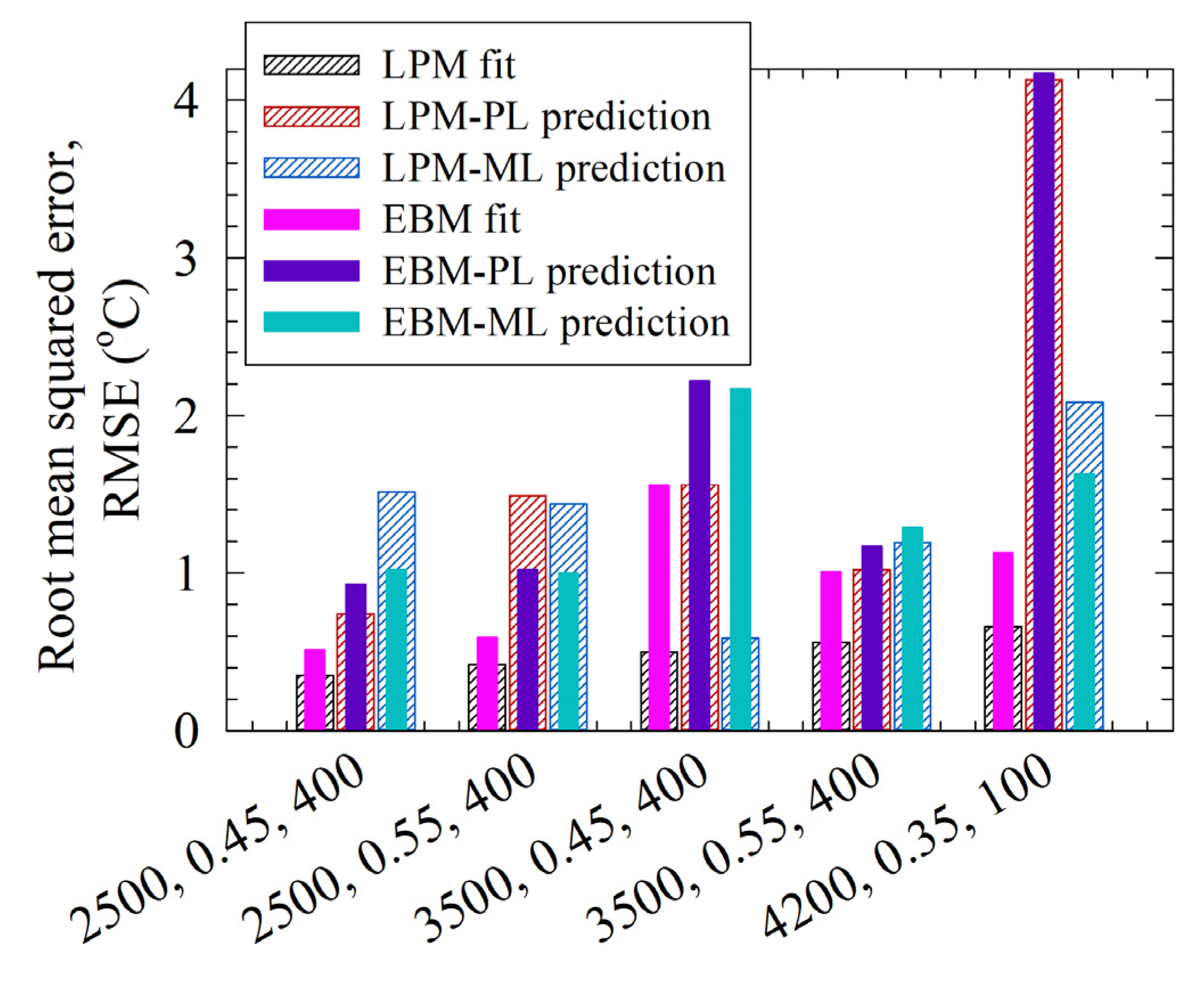
3.4. Comparative Analysis of LPM and EBM Fits and Their Predictions for the Test Runs
3.5. The LPM and the EBM Comparison and the Limitations of the LPM
3.6. A holistic Perspective on the Impact of Process Parameters
4. Conclusions and Future Outlook
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| A | heat transfer surface area, m2 |
| Am | heat transfer surface area of the mill chamber, m2 |
| c | bead loading or fractional volumetric concentration of the beads, – |
| Cp | specific heat capacity, J/g °C |
| EBM | enthalpy balance model |
| Db | bead size, µm |
| LPM | lumped-parameter model |
| m | mass in the mill chamber, g |
| ML | machine learning |
| PL | power law |
| P | power consumption, J/min |
| Pξ | heat generation rate during milling, retrieved from EBM study, J/min |
| Qgen | apparent heat generation during milling, J/min |
| RMSE | root-mean-squared error, °C |
| T | temperature at the mill outlet, °C |
| Tch | chiller temperature, °C |
| Trise | temperature rise at 6 min of milling, °C |
| T0 | initial temperature at the mill outlet, °C |
| t | milling time, min |
| U | overall heat transfer coefficient, W/m2 °C |
| UA | apparent overall heat transfer coefficient times surface area, J/min °C |
| UAm | apparent overall heat transfer coefficient times surface area in the mill chamber, J/min °C |
| ξ | fraction of mechanical power converted into heat, – |
| ω | stirrer (rotational) speed, rpm |
References
- Jermain, S.V.; Brough, C.; Williams, R.O. Amorphous solid dispersions and nanocrystal technologies for poorly water-soluble drug delivery—An update. Int. J. Pharm. 2018, 535, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Ku, M.S.; Dulin, W. A biopharmaceutical classification-based right-first-time formulation approach to reduce human pharmacokinetic variability and project cycle time from first-in-human to clinical proof-of-concept. Pharm. Dev. Technol. 2012, 17, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Takagi, T.; Ramachandran, C.; Bermejo, M.; Yamashita, S.; Yu, L.X.; Amidon, G.L. A provisional biopharmaceutical classification of the top 200 oral drug products in the United States, great Britain, Spain, and Japan. Mol. Pharm. 2006, 3, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Kipp, J. The role of solid nanoparticle technology in the parenteral delivery of poorly water-soluble drugs. Int. J. Pharm. 2004, 284, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Anane-Adjei, A.B.; Jacobs, E.; Nash, S.C.; Askin, S.; Soundararajan, R.; Kyobula, M.; Booth, J.; Campbell, A. Amorphous solid dispersions: Utilization and challenges in preclinical drug development within astrazeneca. Int. J. Pharm. 2022, 614, 121387. [Google Scholar] [CrossRef]
- Keserü, G.M.; Makara, G.M. The influence of lead discovery strategies on the properties of drug candidates. Nat. Rev. Drug Discov. 2009, 8, 203–212. [Google Scholar] [CrossRef]
- Ricarte, R.G.; van Zee, N.J.; Li, Z.; Johnson, L.M.; Lodge, T.P.; Hillmyer, M.A. Recent advances in understanding the micro- and nanoscale phenomena of amorphous solid dispersions. Mol. Pharm. 2019, 16, 4089–4103. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
- Liu, H.; Taylor, L.S.; Edgar, K.J. The role of polymers in oral bioavailability enhancement; a review. Polymer 2015, 77, 399–415. [Google Scholar] [CrossRef]
- Tan, J.; Liu, J.; Ran, L. A review of pharmaceutical nano-cocrystals: A novel strategy to improve the chemical and physical properties for poorly soluble drugs. Crystals 2021, 11, 463. [Google Scholar] [CrossRef]
- Bhujbal, S.V.; Mitra, B.; Jain, U.; Gong, Y.; Agrawal, A.; Karki, S.; Taylor, L.S.; Kumar, S.; Zhou, Q.T. Pharmaceutical amorphous solid dispersion: A review of manufacturing strategies. Acta Pharm. Sin. B 2021, 11, 2505–2536. [Google Scholar] [CrossRef] [PubMed]
- Schittny, A.; Huwyler, J.; Puchkov, M. Mechanisms of increased bioavailability through amorphous solid dispersions: A review. Drug Deliv. 2020, 27, 110–127. [Google Scholar] [CrossRef] [PubMed]
- Sathisaran, I.; Dalvi, S.V. Engineering cocrystals of poorly water-soluble drugs to enhance dissolution in aqueous medium. Pharmaceutics 2018, 10, 108. [Google Scholar] [CrossRef]
- Kumar, S. Pharmaceutical cocrystals: An overview. Indian J. Pharm. Sci. 2018, 79, 858–871. [Google Scholar] [CrossRef]
- Bhakay, A.; Rahman, M.; Dave, R.N.; Bilgili, E. Bioavailability enhancement of poorly water-soluble drugs via nanocomposites: Formulation–processing aspects and challenges. Pharmaceutics 2018, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Azad, M.; Davé, R.; Bilgili, E. Nanomilling of drugs for bioavailability enhancement: A holistic formulation-process perspective. Pharmaceutics 2016, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Bilgili, E.; Guner, G. Mechanistic modeling of wet stirred media milling for production of drug nanosuspensions. AAPS PharmSciTech 2020, 22, 2. [Google Scholar] [CrossRef] [PubMed]
- Lehocký, R.; Pěček, D.; Štěpánek, F. Scale-up from batch to flow-through wet milling process for injectable depot formulation. Eur. J. Pharm. Sci. 2016, 95, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Nakach, M.; Authelin, J.-R.; Agut, C. New approach and practical modelling of bead milling process for the manufacturing of nanocrystalline suspensions. J. Pharm. Sci. 2017, 106, 1889–1904. [Google Scholar] [CrossRef]
- Siewert, C.; Moog, R.; Alex, R.; Kretzer, P.; Rothenhäusler, B. Process and scaling parameters for wet media milling in early phase drug development: A knowledge based approach. Eur. J. Pharm. Sci. 2018, 115, 126–131. [Google Scholar] [CrossRef]
- Breitung-Faes, S.; Kwade, A. Nano particle production in high-power-density mills. Chem. Eng. Res. Des. 2008, 86, 390–394. [Google Scholar] [CrossRef]
- Hennart, S.L.A.; Domingues, M.C.; Wildeboer, W.J.; van Hee, P.; Meesters, G.M.H. Study of the process of stirred ball milling of poorly water soluble organic products using factorial design. Powder Technol. 2010, 198, 56–60. [Google Scholar] [CrossRef]
- Juhnke, M.; Märtin, D.; John, E. Generation of wear during the production of drug nanosuspensions by wet media milling. Eur. J. Pharm. Biopharm. 2012, 81, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Singare, D.S.; Marella, S.; Gowthamrajan, K.; Kulkarni, G.T.; Vooturi, R.; Rao, P.S. Optimization of formulation and process variable of nanosuspension: An industrial perspective. Int. J. Pharm. 2010, 402, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Srinivasan, K.; Gowthamarajan, K.; Singare, D.S.; Prakash, D.; Gaikwad, N.B. Investigation of preparation parameters of nanosuspension by top-down media milling to improve the dissolution of poorly water-soluble glyburide. Eur. J. Pharm. Biopharm. 2011, 78, 441–446. [Google Scholar] [CrossRef]
- Patel, P.J.; Gajera, B.Y.; Dave, R.H. A quality-by-design study to develop nifedipine nanosuspension: Examining the relative impact of formulation variables, wet media milling process parameters and excipient variability on drug product quality attributes. Drug Dev. Ind. Pharm. 2018, 44, 1942–1952. [Google Scholar] [CrossRef]
- Ahuja, B.K.; Jena, S.K.; Paidi, S.K.; Bagri, S.; Suresh, S. Formulation, optimization and in vitro–in vivo evaluation of febuxostat nanosuspension. Int. J. Pharm. 2015, 478, 540–552. [Google Scholar] [CrossRef]
- Medarević, D.; Djuriš, J.; Ibrić, S.; Mitrić, M.; Kachrimanis, K. Optimization of formulation and process parameters for the production of carvedilol nanosuspension by wet media milling. Int. J. Pharm. 2018, 540, 150–161. [Google Scholar] [CrossRef]
- Afolabi, A.; Akinlabi, O.; Bilgili, E. Impact of process parameters on the breakage kinetics of poorly water-soluble drugs during wet stirred media milling: A microhydrodynamic view. Eur. J. Pharm. Sci. 2014, 51, 75–86. [Google Scholar] [CrossRef]
- Guner, G.; Kannan, M.; Berrios, M.; Bilgili, E. Use of bead mixtures as a novel process optimization approach to nanomilling of drug suspensions. Pharm. Res. 2021, 38, 1279–1296. [Google Scholar] [CrossRef]
- Eskin, D.; Zhupanska, O.; Hamey, R.; Moudgil, B.; Scarlett, B. Microhydrodynamics of stirred media milling. Powder Technol. 2005, 156, 95–102. [Google Scholar] [CrossRef]
- Wylie, J.J.; Koch, D.L.; Ladd, A.J. Rheology of suspensions with high particle inertia and moderate fluid inertia. J. Fluid Mech. 2003, 480, 95–118. [Google Scholar] [CrossRef]
- Guner, G.; Elashri, S.; Mehaj, M.; Seetharaman, N.; Yao, H.F.; Clancy, D.J.; Bilgili, E. An enthalpy-balance model for timewise evolution of temperature during wet stirred media milling of drug suspensions. Pharm. Res. 2022, 39, 2065–2082. [Google Scholar] [CrossRef] [PubMed]
- Guner, G.; Seetharaman, N.; Elashri, S.; Mehaj, M.; Bilgili, E. Analysis of heat generation during the production of drug nanosuspensions in a wet stirred media mill. Int. J. Pharm. 2022, 624, 122020. [Google Scholar] [CrossRef] [PubMed]
- Bitterlich, A.; Laabs, C.; Krautstrunk, I.; Dengler, M.; Juhnke, M.; Grandeury, A.; Bunjes, H.; Kwade, A. Process parameter dependent growth phenomena of naproxen nanosuspension manufactured by wet media milling. Eur. J. Pharm. Biopharm. 2015, 92, 171–179. [Google Scholar] [CrossRef]
- Knieke, C.; Azad, M.; Davé, R.; Bilgili, E. A study of the physical stability of wet media-milled fenofibrate suspensions using dynamic equilibrium curves. Chem. Eng. Res. Des. 2013, 91, 1245–1258. [Google Scholar] [CrossRef]
- Verma, S.; Kumar, S.; Gokhale, R.; Burgess, D.J. Physical stability of nanosuspensions: Investigation of the role of stabilizers on ostwald ripening. Int. J. Pharm. 2011, 406, 145–152. [Google Scholar] [CrossRef]
- Aleandri, S.; Schönenberger, M.; Niederquell, A.; Kuentz, M. Temperature-induced surface effects on drug nanosuspensions. Pharm. Res. 2018, 35, 69. [Google Scholar] [CrossRef]
- Descamps, M.; Willart, J. Perspectives on the amorphisation/milling relationship in pharmaceutical materials. Adv. Drug Deliv. Rev. 2016, 100, 51–66. [Google Scholar] [CrossRef]
- Coelho, A.; Schenck, L.; Guner, G.; Punia, A.; Bilgili, E. A combined isolation and formulation approach to convert nanomilled suspensions into high drug-loaded composite particles that readily reconstitute. Powders 2022, 1, 88–110. [Google Scholar] [CrossRef]
- Khuman, P.; Singh, W.B.K.; Devi, S.D.; Naorem, H. Viscosity-temperature behavior of hydroxypropyl cellulose solution in presence of an electrolyte or a surfactant: A convenient method to determine the cloud point of polymer solutions. J. Macromol. Sci. Part A 2014, 51, 924–930. [Google Scholar] [CrossRef]
- Frances, C. On modelling of submicronic wet milling processes in bead mills. Powder Technol. 2004, 143, 253–263. [Google Scholar] [CrossRef]
- Sommer, M.; Stenger, F.; Peukert, W.; Wagner, N.J. Agglomeration and breakage of nanoparticles in stirred media mills—a comparison of different methods and models. Chem. Eng. Sci. 2006, 61, 135–148. [Google Scholar] [CrossRef]
- Gudin, D.; Turczyn, R.; Mio, H.; Kano, J.; Saito, F. Simulation of the movement of beads by the DEM with respect to the wet grinding process. AIChE J. 2006, 52, 3421–3426. [Google Scholar] [CrossRef]
- Jayasundara, C.T.; Yang, R.Y.; Yu, A.B. Discrete particle simulation of particle flow in a stirred mill: Effect of mill properties and geometry. Ind. Eng. Chem. Res. 2012, 51, 1050–1061. [Google Scholar] [CrossRef]
- Varinot, C.; Berthiaux, H.; Dodds, J. Prediction of the product size distribution in associations of stirred bead mills. Powder Technol. 1999, 105, 228–236. [Google Scholar] [CrossRef]
- Stražišar, J.; Runovc, F. Kinetics of comminution in micro- and sub-micrometer ranges. Int. J. Miner. Process. 1996, 44–45, 673–682. [Google Scholar] [CrossRef]
- Azad, M.; Guner, G.; Afolabi, A.; Davé, R.; Bilgili, E. Impact of solvents during wet stirred media milling of cross-linked biopolymer suspensions. Adv. Powder Technol. 2021, 32, 4562–4575. [Google Scholar] [CrossRef]
- Guner, G.; Yilmaz, D.; Bilgili, E. Kinetic and microhydrodynamic modeling of fenofibrate nanosuspension production in a wet stirred media mill. Pharmaceutics 2021, 13, 1055. [Google Scholar] [CrossRef]
- Guner, G.; Yilmaz, D.; Eskin, D.; Bilgili, E. Effects of bead packing limit concentration on microhydrodynamics-based prediction of breakage kinetics in wet stirred media milling. Powder Technol. 2022, 403, 117433. [Google Scholar] [CrossRef]
- Elsheikh, A.H.; Saba, A.I.; Panchal, H.; Shanmugan, S.; Alsaleh, N.A.; Ahmadein, M. Artificial intelligence for forecasting the prevalence of covid-19 pandemic: An overview. Healthcare 2021, 9, 1614. [Google Scholar] [CrossRef] [PubMed]
- Ahmadein, M.; Elsheikh, A.H.; Alsaleh, N.A. Modeling of cooling and heat conduction in permanent mold casting process. Alex. Eng. J. 2022, 61, 1757–1768. [Google Scholar] [CrossRef]
- Janssen, A.; Bennis, F.C.; Mathôt, R.A. Adoption of machine learning in pharmacometrics: An overview of recent implementations and their considerations. Pharmaceutics 2022, 14, 1814. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Ma, X.; Ouyang, D.; Williams III, R.O. Emerging artificial intelligence (ai) technologies used in the development of solid dosage forms. Pharmaceutics 2022, 14, 2257. [Google Scholar] [CrossRef] [PubMed]
- Jamzad, S.; Fassihi, R. Role of surfactant and pH on dissolution properties of fenofibrate and glipizide—A technical note. AAPS PharmSciTech 2006, 7, E17–E22. [Google Scholar] [CrossRef] [PubMed]
- Bilgili, E.; Rahman, M.; Palacios, D.; Arevalo, F. Impact of polymers on the aggregation of wet-milled itraconazole particles and their dissolution from spray-dried nanocomposites. Adv. Powder Technol. 2018, 29, 2941–2956. [Google Scholar] [CrossRef]
- NISSO. Nisso HPC, Nippon Soda Co., Ltd. Nissoexcipients.Com. Available online: https://www.nissoexcipients.com/hpc-e/care_stable (accessed on 12 December 2022).
- Bilgili, E.; Li, M.; Afolabi, A. Is the combination of cellulosic polymers and anionic surfactants a good strategy for ensuring physical stability of BCS class II drug nanosuspensions? Pharm. Dev. Technol. 2016, 21, 499–510. [Google Scholar] [CrossRef]
- Azad, M.; Afolabi, A.; Bhakay, A.; Leonardi, J.; Davé, R.; Bilgili, E. Enhanced physical stabilization of fenofibrate nanosuspensions via wet co-milling with a superdisintegrant and an adsorbing polymer. Eur. J. Pharm. Biopharm. 2015, 94, 372–385. [Google Scholar] [CrossRef]
- Li, M.; Yaragudi, N.; Afolabi, A.; Dave, R.; Bilgili, E. Sub-100 nm drug particle suspensions prepared via wet milling with low bead contamination through novel process intensification. Chem. Eng. Sci. 2015, 130, 207–220. [Google Scholar] [CrossRef]
- Li, M.; Alvarez, P.; Bilgili, E. A microhydrodynamic rationale for selection of bead size in preparation of drug nanosuspensions via wet stirred media milling. Int. J. Pharm. 2017, 524, 178–192. [Google Scholar] [CrossRef]
- Guner, G. Microhydrodynamic, Kinetic and Thermal Modeling of Wet Media Milling for Process Optimization and Intensification. Ph.D. Dissertation, New Jersey Institute of Technology, Newark, NJ, USA, 2022. [Google Scholar]
- Geankoplis, C. Transport Processes and Separation Process Principles, 4th ed.; Prentice Hall: Hoboken, NJ, USA, 2003. [Google Scholar]
- Annapragada, A.; Adjei, A. Numerical simulation of milling processes as an aid to process design. Int. J. Pharm. 1996, 136, 1–11. [Google Scholar] [CrossRef]
- Tojo, T.; Atake, T.; Mori, T.; Yamamura, H. Heat capacity and thermodynamic functions of zirconia and yttria-stabilized zirconia. J. Chem. Thermodyn. 1999, 31, 831–845. [Google Scholar] [CrossRef]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning; Springer: Berlin/Heidelberg, Germany, 2013; Volume 112. [Google Scholar]
- Bilgili, E.; Afolabi, A. A combined microhydrodynamics–polymer adsorption analysis for elucidation of the roles of stabilizers in wet stirred media milling. Int. J. Pharm. 2012, 439, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Cerdeira, A.M.; Gander, B.; Mazzotti, M. Role of milling parameters and particle stabilization on nanogrinding of drug substances of similar mechanical properties. Chem. Eng. Technol. 2011, 34, 1427–1438. [Google Scholar] [CrossRef]
- Fogel’Son, R.; Likhachev, E. Temperature dependence of viscosity. Tech. Phys. 2001, 46, 1056–1059. [Google Scholar] [CrossRef]
- Mannheim, V. Empirical and scale-up modeling in stirred ball mills. Chem. Eng. Res. Des. 2011, 89, 405–409. [Google Scholar] [CrossRef]
- Barrios, A.N.S.; Silva, J.B.C.; Rodrigues, A.R.; Coelho, R.T.; Braghini Junior, A.; Matsumoto, H. Modeling heat transfer in die milling. Appl. Therm. Eng. 2014, 64, 108–116. [Google Scholar] [CrossRef]
- Xi, S.Q.; Zhou, J.G.; Wang, X.T. Research on temperature rise of powder during high energy ball milling. Powder Metall. 2007, 50, 367–373. [Google Scholar] [CrossRef]
- Hennart, S.; Wildeboer, W.; van Hee, P.; Meesters, G. Identification of the grinding mechanisms and their origin in a stirred ball mill using population balances. Chem. Eng. Sci. 2009, 64, 4123–4130. [Google Scholar] [CrossRef]
- Li, H.; Ndjamo, A.; Sauriol, P.; Patience, G.S. Optimization of lifepo4 wet media milling and regressive population balance modeling. Adv. Powder Technol. 2017, 28, 1000–1007. [Google Scholar] [CrossRef]
| Run No. | Stirrer Speed, ω (rpm) | Bead Loading, c (-) | Bead Size, Db (µm) |
|---|---|---|---|
| 1 1 | 2000 | 0.4 | 200 |
| 2 1 | 2000 | 0.4 | 400 |
| 3 1 | 2000 | 0.4 | 800 |
| 4 1 | 2000 | 0.5 | 200 |
| 5 1 | 2000 | 0.5 | 400 |
| 6 1 | 2000 | 0.5 | 800 |
| 7 1 | 2000 | 0.6 | 200 |
| 8 1 | 2000 | 0.6 | 400 |
| 9 1 | 2000 | 0.6 | 800 |
| 10 1 | 3000 | 0.4 | 200 |
| 11 1 | 3000 | 0.4 | 400 |
| 12 1 | 3000 | 0.4 | 800 |
| 13 1 | 3000 | 0.5 | 200 |
| 14 1 | 3000 | 0.5 | 400 |
| 15 1 | 3000 | 0.5 | 800 |
| 16 1 | 3000 | 0.6 | 200 |
| 17 1 | 3000 | 0.6 | 400 |
| 18 1 | 3000 | 0.6 | 800 |
| 19 1 | 4000 | 0.4 | 200 |
| 20 1 | 4000 | 0.4 | 400 |
| 21 1 | 4000 | 0.4 | 800 |
| 22 1 | 4000 | 0.5 | 200 |
| 23 1 | 4000 | 0.5 | 400 |
| 24 1 | 4000 | 0.5 | 800 |
| 25 1 | 4000 | 0.6 | 200 |
| 26 1 | 4000 | 0.6 | 400 |
| 27 1 | 4000 | 0.6 | 800 |
| 28 2 | 2500 | 0.45 | 400 |
| 29 2 | 2500 | 0.55 | 400 |
| 30 2 | 3500 | 0.45 | 400 |
| 31 2 | 3500 | 0.55 | 400 |
| 32 2 | 4000 | 0.35 | 100 |
| Run No: Identifier | Qgen (J/min) | UA (J/min °C) | RMSE (°C) |
|---|---|---|---|
| 1: 2000 0.4 200 | 755.2 | 47.79 | 0.40 |
| 2: 2000 0.4 400 | 1616 | 103.5 | 0.46 |
| 3: 2000 0.4 800 | 787.4 | 48.08 | 0.56 |
| 4: 2000 0.5 200 | 441.4 | 25.82 | 0.32 |
| 5: 2000 0.5 400 | 720.8 | 40.18 | 0.33 |
| 6: 2000 0.5 800 | 1296 | 72.88 | 0.39 |
| 7: 2000 0.6 200 | 1343 | 68.63 | 0.44 |
| 8: 2000 0.6 400 | 2625 | 131.9 | 0.15 |
| 9: 2000 0.6 800 | 1822 | 89.95 | 0.50 |
| 10: 3000 0.4 200 | 1402 | 53.92 | 0.55 |
| 11: 3000 0.4 400 | 2938 | 107.0 | 0.57 |
| 12: 3000 0.4 800 | 2798 | 101.0 | 0.66 |
| 13: 3000 0.5 200 | 1837 | 61.49 | 0.40 |
| 14: 3000 0.5 400 | 3243 | 107.3 | 0.72 |
| 15: 3000 0.5 800 | 3307 | 107.4 | 0.83 |
| 16: 3000 0.6 200 | 2624 | 94.08 | 0.43 |
| 17: 3000 0.6 400 | 4598 | 128.2 | 0.76 |
| 18: 3000 0.6 800 | 4542 | 124.4 | 0.80 |
| 19: 4000 0.4 200 | 5075 | 135.1 | 0.42 |
| 20: 4000 0.4 400 | 6266 | 162.7 | 0.41 |
| 21: 4000 0.4 800 | 6245 | 162.5 | 0.51 |
| 22: 4000 0.5 200 | 6116 | 162.7 | 0.90 |
| 23: 4000 0.5 400 | 8490 | 220.0 | 0.30 |
| 24: 4000 0.5 800 | 9359 | 238.1 | 0.60 |
| 25: 4000 0.6 200 | 8383 | 208.9 | 0.62 |
| 26: 4000 0.6 400 | 10,600 | 261.7 | 0.28 |
| 27: 4000 0.6 800 | 10,740 | 171.9 | 0.34 |
| Runs | Direct Fitting | PL Prediction | ML Prediction | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Qgen (J/min) | UA (J/min °C) | LPM RMSE (°C) | EBM RMSE (°C) 1 | Qgen (J/min) | UA (J/min °C) | LPM RMSE (°C) | EBM RMSE (°C) 1 | Qgen (J/min) | UA (J/min °C) | LPM RMSE (°C) | EBM RMSE (°C) 1 | |
| 2500 0.45 400 | 1481 | 68 | 0.35 | 0.51 | 1634 | 78 | 0.74 | 0.93 | 1792 | 77 | 1.51 | 1.02 |
| 2500 0.55 400 | 2495 | 101 | 0.42 | 0.59 | 2118 | 91 | 1.49 | 1.02 | 2506 | 95 | 1.44 | 1.00 |
| 3500 0.45 400 | 4084 | 118 | 0.50 | 1.56 | 4519 | 137 | 1.56 | 2.22 | 4555 | 132 | 0.59 | 2.17 |
| 2500 0.55 400 | 5567 | 147 | 0.56 | 1.01 | 5856 | 160 | 1.02 | 1.17 | 5911 | 162 | 1.19 | 1.29 |
| 4200 0.35 100 | 3185 | 84 | 0.66 | 1.13 | 4180 | 129 | 4.13 | 4.17 | 4359 | 124 | 2.08 | 1.63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guner, G.; Yilmaz, D.; Yao, H.F.; Clancy, D.J.; Bilgili, E. Predicting the Temperature Evolution during Nanomilling of Drug Suspensions via a Semi-Theoretical Lumped-Parameter Model. Pharmaceutics 2022, 14, 2840. https://doi.org/10.3390/pharmaceutics14122840
Guner G, Yilmaz D, Yao HF, Clancy DJ, Bilgili E. Predicting the Temperature Evolution during Nanomilling of Drug Suspensions via a Semi-Theoretical Lumped-Parameter Model. Pharmaceutics. 2022; 14(12):2840. https://doi.org/10.3390/pharmaceutics14122840
Chicago/Turabian StyleGuner, Gulenay, Dogacan Yilmaz, Helen F. Yao, Donald J. Clancy, and Ecevit Bilgili. 2022. "Predicting the Temperature Evolution during Nanomilling of Drug Suspensions via a Semi-Theoretical Lumped-Parameter Model" Pharmaceutics 14, no. 12: 2840. https://doi.org/10.3390/pharmaceutics14122840
APA StyleGuner, G., Yilmaz, D., Yao, H. F., Clancy, D. J., & Bilgili, E. (2022). Predicting the Temperature Evolution during Nanomilling of Drug Suspensions via a Semi-Theoretical Lumped-Parameter Model. Pharmaceutics, 14(12), 2840. https://doi.org/10.3390/pharmaceutics14122840









