Abstract
Oak trees are widely distributed nationwide and account for approximately 24% of the total forest area in South Korea. However, these species are currently threatened by oak wilt disease caused by Dryadomyces quercus-mongolicae, leading to significant economic and ecological losses in the forestry industry. This study evaluated the effectiveness of culture suspension and lyophilized powder formulations of Streptomyces blastmyceticus in controlling oak wilt disease on Mongolian oak (Quercus mongolica). Field experiments were conducted using trunk and root injection methods in Q. mongolica plantations. The non-conductive area (NCA) of sapwood and colonization rate of the oak wilt fungus were analyzed and compared across treatments. In the Chuncheon experiment, Kangwon province, only the root injection of fungicide showed a significant difference compared to the culture suspension treatments. There were no significant differences between culture suspension and lyophilized powder treatments in Uiwang, Gyeonggi Province. Specifically, both preventive and curative treatments using culture suspension and lyophilized powder of S. blastmyceticus resulted in significantly different NCA values compared to the negative control (8.7%) and positive control (88.5%). The NCA for culture suspension ranged from 33.3% to 49.9%, and for lyophilized powder, from 37.3% to 43.9%. The colonization rate of the oak wilt fungus was lowest (9.72%) in the preventive treatment using lyophilized powder via trunk injection. For the culture suspension, the lowest colonization rate (20.83%) was observed in the curative treatment using trunk injection. These findings suggest that the lyophilized powder formulation of S. blastmyceticus efficiently suppresses the progression of oak wilt disease under field conditions.
1. Introduction
The biocontrol of pests and diseases using beneficial microorganisms has been widely studied worldwide [1,2]. For insect pest management, species of Beauveria and Metarhizium have been utilized due to their ability to invade insect tissues and produce insecticidal toxins [2,3,4]. Other beneficial microorganisms, including fungi, bacteria, and actinomycetes, have been applied to control plant pathogens [5,6,7,8]. For instance, the endophytic bacteria Paenibacillus polymyxa inhibited 8 different Fusarium pathogens [9]. Additionally, the endophytic fungus Yamadazyma mexicana, isolated from avocado fruit epicarp, exhibited antifungal activity against Colletotrichum gloeosporioides [10,11] and endophytic actinomycetes Streptomyces spp. from sorghum stems showed inhibitory ability against the ubiquitous phytopathogen Rhizoctonia solani [12]. Currently, the formulation of microbial biocontrol and botanical agents is considered a sustainable strategy for managing plant pathogens [13,14,15]. A powder formulation of Streptomyces corchorusii strain UCR3-16, isolated from rice rhizospheric soils, has been developed as both a biocontrol and biofertilizer agent for rice cultivation [15]. Another study demonstrated that the dry powder formulation of Talaromyces tratensis KUFA0091 efficiently controlled dirty panicle disease in rice under field conditions [16]. Endophytic Trichoderma strains obtained from healthy corn leaves effectively controlled corn leaf blight caused by Exserohilum turcicum; however, fresh formulations of each Trichoderma strain exhibited higher efficacy than their corresponding dry powder formulations [14]. Moreover, a powder formulation of Meyerozyma caribbica was applied to protect postharvest mango fruit from C. gloeosporioides, a primary cause of postharvest anthracnose in mango [17,18]. This formulation was most effective when mango fruit was stored at 25 °C, resulting in reductions in disease incidence and severity by up to 53.4% and 23.9%, respectively [18].
Since 2004, oak wilt disease associated with Dryadomyces quercus-mongolicae has been a threat to Mongolian oak (Quercus mongolica) populations in South Korea. This fungal pathogen was initially described as Raffaelea quercus-mongolicae but was subsequently reclassified as Dryadomyces quercus-mongolicae [19,20]. This disease induces systematic wilting of oak trees (Figure 1) and has spread throughout the country via the insect vector, Platypus koryoensis [19]. Several biocontrol strategies for the oak wilt fungus have been evaluated in South Korea [21,22,23,24]. These approaches involve the selection of endophytic fungi with antifungal properties against D. quercus-mongolicae, and some promising isolates have been identified as potential biocontrol agents [23,24]. For example, the culture filtrate of Nectria balsamea E282 and its extracts completely inhibited mycelial growth, sporulation, and spore germination of the oak wilt fungus under laboratory conditions [24]. Similarly, the culture filtrate of Streptomyces blastmyceticus exhibited strong antifungal activity against D. quercus-mongolicae, and tree injection with its culture suspension effectively prevented oak wilt disease [21,22]. The preventive efficacy of the culture suspension was comparable to that of the fungicide Propiconazole (14.3%, Alamo®) in suppressing colonization of the injected oak wilt fungus [22]. Other methods have been used to manage oak wilt disease in Korea, including the application of silver nanoparticles to control the oak wilt fungus and insect vector trapping to limit disease spread [19,25].

Figure 1.
The typical symptoms of oak wilt disease, caused by Dryadomyces quercus-mongolicae, include the following: (a) wilting leaves on oak trees; (b) sawdust from the holes created by insect vectors; (c) the insect vector (Platypus koryoensis); (d) discoloration of the sapwood caused by the pathogen; and (e) mycelia of D. quercus-mongolicae growing on potato dextrose agar (PDA) medium.
To date, no biological formulations, such as lyophilized powder products, have been developed for controlling oak wilt disease; therefore, it is essential to explore this potential bioproduct. Streptomyces species are well known to function as plant growth promoters and biocontrol agents through the production of volatile organic compounds, antibiotics, and other secondary metabolites [7]. Additionally, Streptomyces spp. can enhance induced resistance against oak powdery mildew and activate systemic defense responses in oak trees [26]. Our previous study demonstrated that treatment with a culture suspension of S. blastmyceticus effectively prevented oak wilt disease [22]. However, its potential use as a curative treatment to suppress the oak wilt fungus has not yet been investigated. Long-term preservation of liquid culture suspensions is challenging and logistically inconvenient. Although freeze-drying may reduce spore viability over time, in our previous study, we found that lyophilized spores and mycelia of S. blastmyceticus remained viable for up to 240 days when stored at 4 °C [21]. Therefore, this study aimed to compare the preventive and curative efficacy of culture suspension treatments and to evaluate the effectiveness of a lyophilized powder formulation of S. blastmyceticus in controlling oak wilt disease. Field experiments were conducted using trunk and root flare injection methods. The efficacy of the lyophilized powder formulation was compared with that of the culture suspension by analyzing the non-conductive area (NCA) of sapwood and the colonization rate of the oak wilt fungus in wood samples collected from the treated trees.
2. Materials and Methods
2.1. Study Site and Experimental Design
The study sites were located in Mongolian oak (Quercus mongolica) plantations in Chuncheon, Kangwon province (site 1: 37°52′21″ N, 127°48′25″ E; 423 m elevation) and Uiwang, Gyeonggi province (site 2: 37°21′47″ N, 126°59′50″ E; 196 m elevation). At each site, thirty healthy trees with a diameter at breast height (1.3 m above the ground) of 8–12 cm were randomly selected for the experiment. The treatments were designed based on a previous study [22]. A completely randomized design was applied, with three biological replicates (trees) per treatment. Trees were spatially distributed within each site to minimize micro-site effects; however, formal blocking was not applied due to homogeneous stand structure and uniform tree size. Treatment included preventive and curative applications using trunk and root flare injection methods. For preventive treatment, test substances were injected into trees one month prior to inoculation with D. quercus-mongolicae. For curative treatments, substances were injected into trees one month after pathogen inoculation. Trunk injections were performed using a Chemjet® tree injector (Brisbane, Australia), whereas root flare injections were conducted using the macro-infusion method. Negative and positive controls consisted of sterilized distilled water and pathogen inoculation alone, respectively. Control treatments were applied using an injection into the trees using Chemjet® tree injectors without additional substances. In the Chuncheon experiment (site 1), the tested substances included the fungicide Alamo® (propiconazole 14.3%; Syngenta Crop Protection, Greensboro, NC, USA) and a culture suspension of S. blastmyceticus. In the Uiwang experiment (site 2), treatments consisted of lyophilized powder and culture suspension of S. blastmyceticus. All treatments at each site were conducted with three biological replicates and the detailed treatments are summarized as follows:
Negative control: distilled water performed at site 1 and site 2.
Positive control: pathogen (D. quercus-mongolicae) performed at site 1 and site 2.
Alamo preventive trunk injection: Alamo injection + pathogen inoculation (site 1).
Alamo curative trunk injection: pathogen inoculation + Alamo injection (site 1).
Alamo preventive root injection: Alamo injection + pathogen inoculation (site 1).
Alamo curative root injection: pathogen inoculation + Alamo injection (site 1).
Culture suspension preventive trunk injection: culture suspension injection + pathogen inoculation (site 1 and site 2).
Culture suspension curative trunk injection: pathogen inoculation + culture suspension injection (site 1 and site 2).
Culture suspension preventive root injection: culture suspension injection + pathogen inoculation (site 1 and site 2).
Culture suspension curative root injection: pathogen inoculation + culture suspension injection (site 1 and site 2).
Lyophilized powder preventive trunk injection: lyophilized powder injection + pathogen inoculation (site 2).
Lyophilized powder curative trunk injection: pathogen inoculation + lyophilized powder injection (site 2).
Lyophilized powder preventive root injection: lyophilized powder injection + pathogen inoculation (site 2).
Lyophilized powder curative root injection: pathogen inoculation + lyophilized powder injection (site 2).
2.2. Preparation of Lyophilized Powder and Culture Suspension of S. blastmyceticus
The culture filtrate of Streptomyces blastmyceticus isolate (TPML13085) completely inhibited the mycelial growth of D. quercus-mongolicae under unheated conditions [21] and this isolate was cultured on Potato Dextrose Agar (PDA) at 25 °C for 5 days in the dark. The isolate was subsequently cultured in Potato Dextrose Broth (PDB) at 25 °C with shaking at 160 rpm for 7 days in the dark to obtain a culture suspension of S. blastmyceticus (108 cfu/mL). The culture suspension was centrifuged at 9000 rpm for 20 min to harvest 100 mg of mycelia and spores. A solution of skimmed milk was sterilized at 110 °C for 15 min. The harvested mycelia and spores were blended with the skimmed milk solution to a final concentration of 12% (w/v) [21]. The mixture was transferred into test tubes, sealed with sterilized cotton plugs, and pre-cooled at 4 °C for 3 h, followed by freezing at −20 °C for 3 h and subsequently at −86 °C for 6 h. The frozen samples were lyophilized for 24 h to obtain a lyophilized powder of S. blastmyceticus, which was vacuum-sealed and stored at 4 °C in a dark room. The lyophilized powder was dissolved in distilled water to a final concentration of 1% (w/v) prior to trunk and root flare injection into trees.
2.3. Injection of Culture Suspension, Lyophilized Powder, Fungicide and Pathogen
During the Chuncheon experiment, trunk injections were performed using four Chemjet® injectors to inject a total of 40 mL of culture suspension per tree. Additionally, 40 mL of Alamo® fungicide was injected into each tree using the same method. For root flare injections, the macro-infusion method was used to inject the same volumes (40 mL) of culture suspension and Alamo® fungicide at the root flare of each tree. Preventive treatments were administered one month prior to pathogen inoculation, whereas curative treatments were applied one month after inoculation. Oak wilt pathogen, D. quercus-mongolicae, was cultured on PDA medium and incubated at 25 °C in the dark for 7 days, followed by an additional 7 days at room temperature to induce spores [23]. A spore suspension of the pathogen (106 cfu/mL) was injected into each tree (40 mL per tree) via trunk injection at 15 cm above ground level for the positive control treatment. The culture suspension and fungicide treatments were injected at positions lower than the pathogen injection point. Sterilized distilled water was injected into each tree using the same trunk injection method to serve as the negative control. After injection, all injection holes were sealed with petroleum jelly to prevent secondary pathogen contamination. The experiment in Uiwang followed the same protocol, except lyophilized powder of S. blastmyceticus was used instead of the Alamo® fungicide.
2.4. Analysis of Non-Conductive Area in Sapwood
Eight months after inoculating the trees with the pathogen, treated trees were cut below the pathogen inoculation points. The cut surfaces were immediately stained by dipping them into baskets filled with a 1% fuchsin acid solution overnight. On the following day, cut wood discs were collected at 0, 10, 20, 30, 40, and 50 cm above and below the inoculation point along the trunk of each treated tree. The wood discs were transported to the laboratory for cross-sectional imaging. The non-conductive area (NCA) of the sapwood was quantified as the proportion of the transverse section using Adobe Photoshop CS6 program version 13.0 (Adobe Systems, San Jose, CA, USA) [22,27].
2.5. Colonization Rate of Oak Wilt Fungus
Four wood chips were collected from the transverse section of each wood disc to determine the colonization rate of D. quercus-mongolicae in treated trees from the Uiwang experiment. These wood chips were surface-sterilized with 2% sodium hypochlorite (NaOCl) followed by ethanol and then placed on potato dextrose agar (PDA) supplemented with streptomycin sulfate (100 mg/L). The plates were incubated at 25 °C in the dark, and the fungal growth of the fungal pathogen was monitored daily for 10 days. A total of 24 wood samples per tree were analyzed to calculate the colonization rate for each treatment. The colonization rate for each tree was calculated using the following formula:
where n is the number of wood chips infected with the oak wilt pathogen, while N is the total number of samples collected from each tree (n = 24).
2.6. Statistical Analysis
A two-way analysis of variance (ANOVA) was conducted to evaluate differences in the non-conductive areas between treatments and controls based on the distance from the inoculation point of the pathogen. One-way ANOVA was performed to compare differences in non-conductive areas among treatments within each experimental site. Model residuals were assessed for normality using the Shapiro–Wilk test. When necessary, data were log-transformed to meet assumptions of normality. Homogeneity of variances across treatments was evaluated using Levene’s test. When significant differences were detected, Tukey’s honestly significant difference (HSD) test was applied for multiple comparisons, and statistical significance was considered at p-value < 0.05. All statistical analyses and computations were performed using IBM SPSS Statistics version 27 for Windows (IBM Corp., Armonk, NY, USA).
3. Results
3.1. Reisolation of D. quercus-mongolicae
The re-isolation results of D. quercus-mongolicae from wood samples of treated trees are presented in Table 1. The number of positive isolates obtained from the different treatments ranged from 0 (negative control) to 41 (positive control). No pathogenic isolates were obtained from the negative control trees, confirming that infections in the experimental trees resulted solely from the inoculated pathogen. Colonization rates differed significantly among treatments (p < 0.05). The highest colonization rate was observed in the positive control (56.95%). No positive isolates were recovered from the negative control treatment. Among the treatments, preventive trunk injection of lyophilized powder showed the lowest colonization rate (9.72%). Consequently, this treatment exhibited the highest control value (82.91%). Curative trunk injection of the culture suspension resulted in a colonization rate of 20.83%, corresponding to a control value of 63.49%. The remaining treatments showed colonization rates ranging from 33.33% to 41.67%, with no significant differences among these treatments (Table 1).

Table 1.
Colonization rate of D. quercus-mongolicae in trees injected with culture suspension and lyophilized powder of S. blastmyceticus before and after pathogen inoculation.
3.2. Comparison of Non-Conductive Area in Sapwood Between Fungicide and Culture Suspension Injections
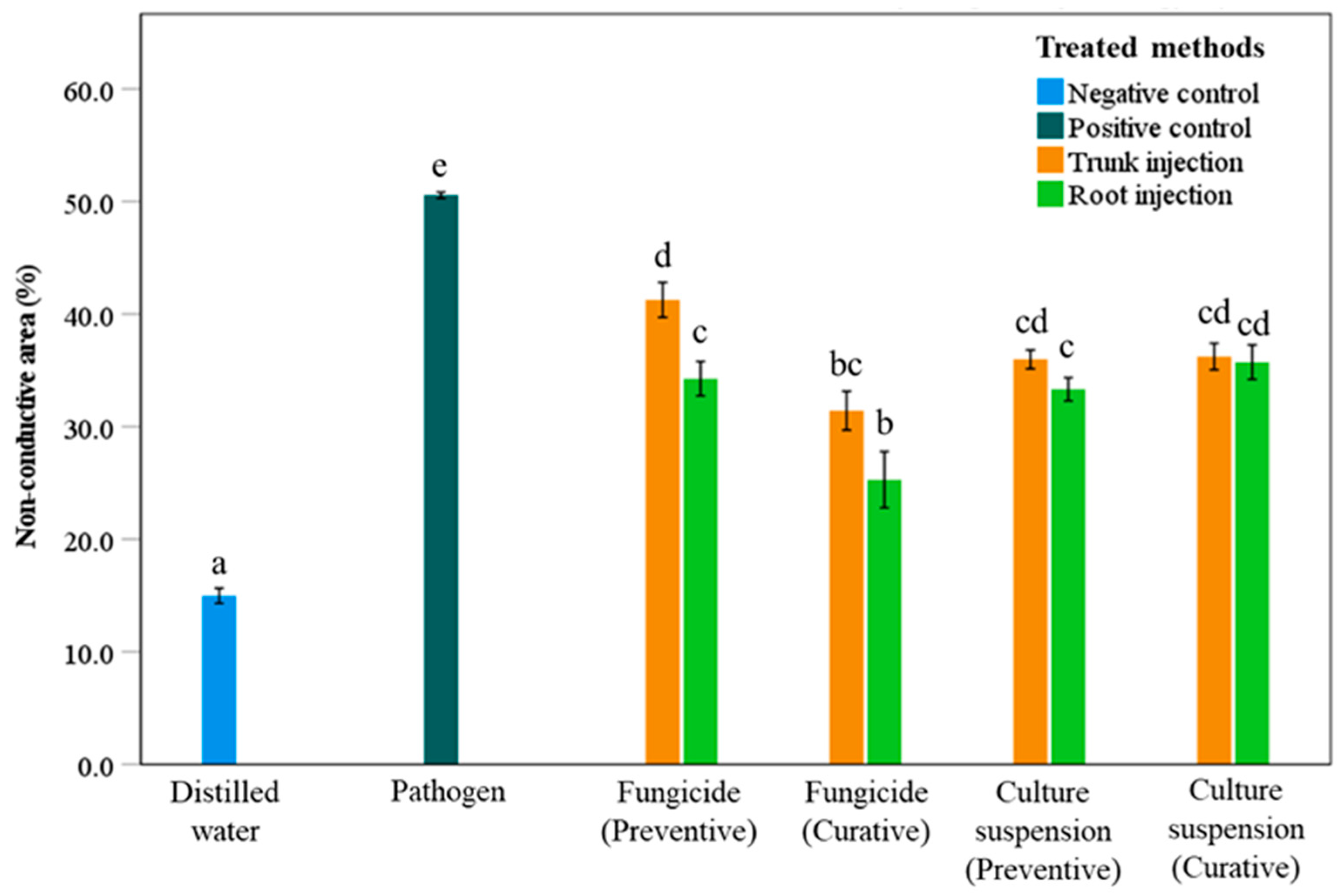
The non-conductive area (NCA) of the sapwood exhibited significant differences among treatments compared with the negative and positive controls (Figure 2). The NCA of the positive control was highest at 50.6%, while the NCA of the negative control was lowest at 15.0%. These results indicate that higher NCA values are associated with greater fungal colonization of the stem, whereas lower NCA values reflect reduced pathogen spread within the sapwood, effectively preventing infection and development of the pathogenic fungi. The trunk injection method generally resulted in higher NCA values than the root injection method under the same treatment conditions (Figure 2). For example, when using the preventive fungicide treatment, the trunk injection method produced a NCA of 41.3%, whereas the root injection method produced a NCA of 34.3%, indicating a significant difference between the two injection methods. In the curative fungicide treatment, the NCA values for trunk and root injection methods were 31.4% and 25.3%, respectively; however, there was no significant difference between the two injection methods. For the culture suspension of S. blastmyceticus, no significant differences in NCA were observed between trunk and root injections in either preventive or curative treatments, with values ranging from 33.3% to 36.0% (Figure 2).
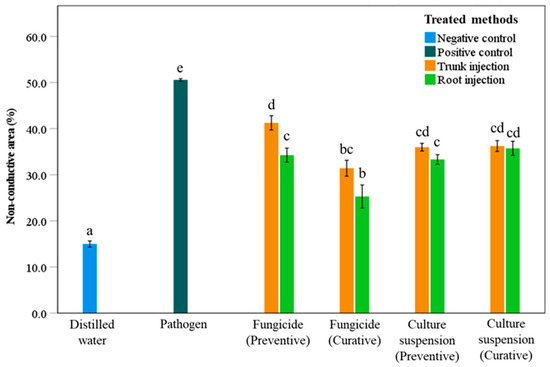
Figure 2.
Percentage of non-conductive areas in the sapwood of Mongolian oak trees after preventive and curative treatments with Alamo fungicide or culture suspension of S. blastmyceticus, performed as trunk or root injections in Chuncheon, Kangwon Province. The vertical bars illustrate the values of the standard error of the means. Different letters indicate a significant difference (p < 0.05) among treatments by Tukey’s HSD test.
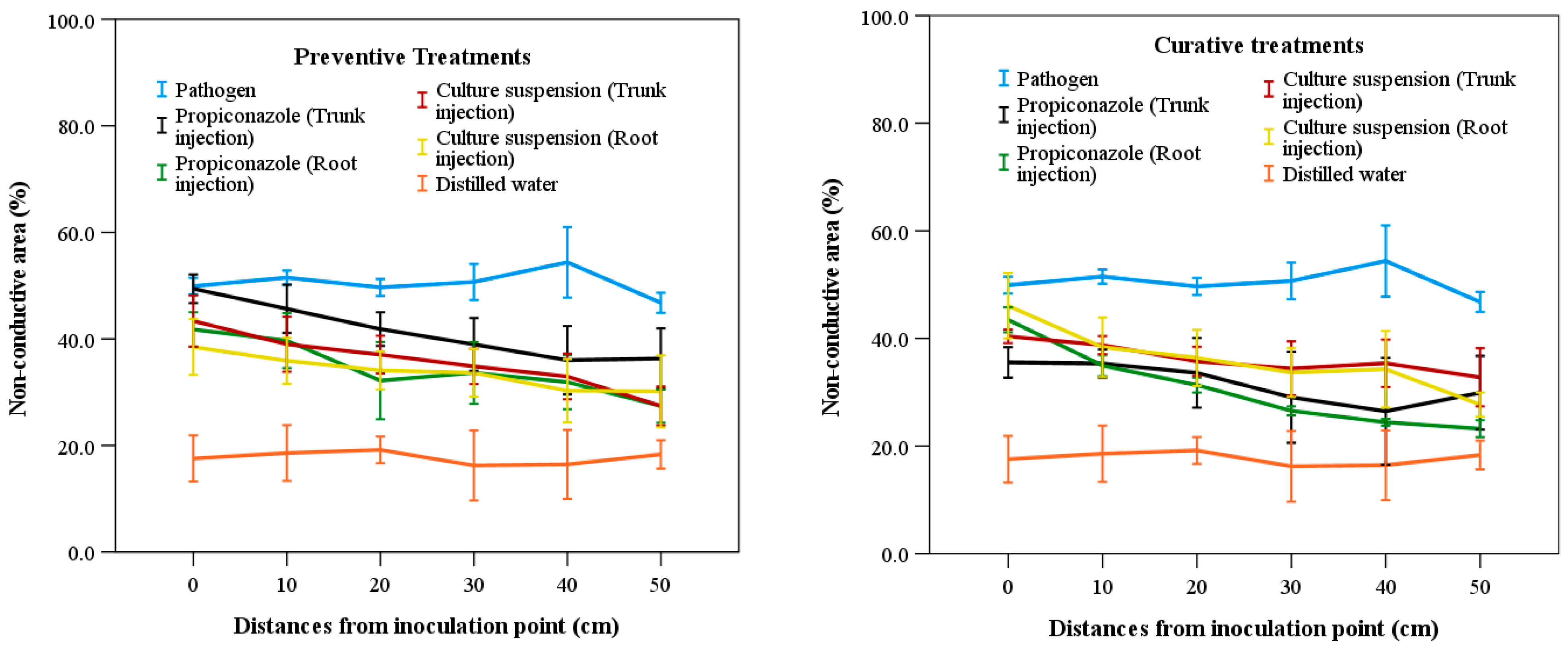
The results of evaluating the non-conductive area (NCA) values at different distances from the inoculation point are presented in Figure 3. In general, NCA values decreased as the distance from the inoculation point of the pathogen increased for all treatments except the negative control. There were no significant differences (p > 0.05) among preventive treatments across the distances from the inoculation point of the pathogen. However, significant differences (p < 0.05) were observed among curative treatments. The treatment with pathogen-only injection showed the highest NCA values at three distance intervals (0–10 cm, 20–30 cm, and 40–50 cm), with values of 50.17%, 49.95%, and 48.93%, respectively. In contrast, the treatment with distilled water exhibited the lowest NCA values of 13.94% and 14.55% at distances of 0–10 cm and 20–30 cm, respectively (Figure 3). The lowest and highest efficiency for different treatments were found at 0–10 cm and 40–50 cm from the inoculation points of the pathogen, respectively. Specifically, in the preventive treatments, trunk injection of fungicide showed the highest NCA value of 48.03%, whereas root injection showed the lowest NCA value of 28.79%. For the culture suspension treatment, the highest and lowest NCA values were 40.37% and 29.77%, respectively. In the curative treatments, root injection of fungicide produced the highest and lowest NCA values of 39.05% and 12.46%, respectively. The highest and lowest NCA values of culture suspension treatment with root injection were 41.24% and 29.58%, respectively (Figure 3).
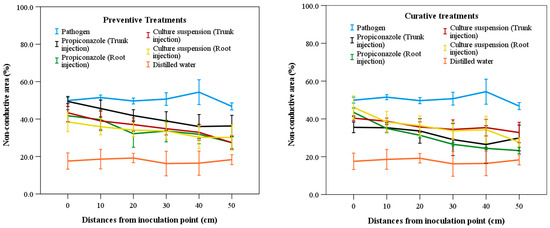
Figure 3.
Non-conductive area in the sapwood according to distances from the inoculation position of the pathogen among treatments of fungicide and culture suspensions of S. blastmyceticus in Chuncheon, Kangwon province. The vertical bars illustrate the values of the standard error of the means. Preventive treatments (left) and curative treatments (right).
3.3. Comparison of Non-Conductive Area in Sapwood Between Culture Suspension and Lyophilized Powder
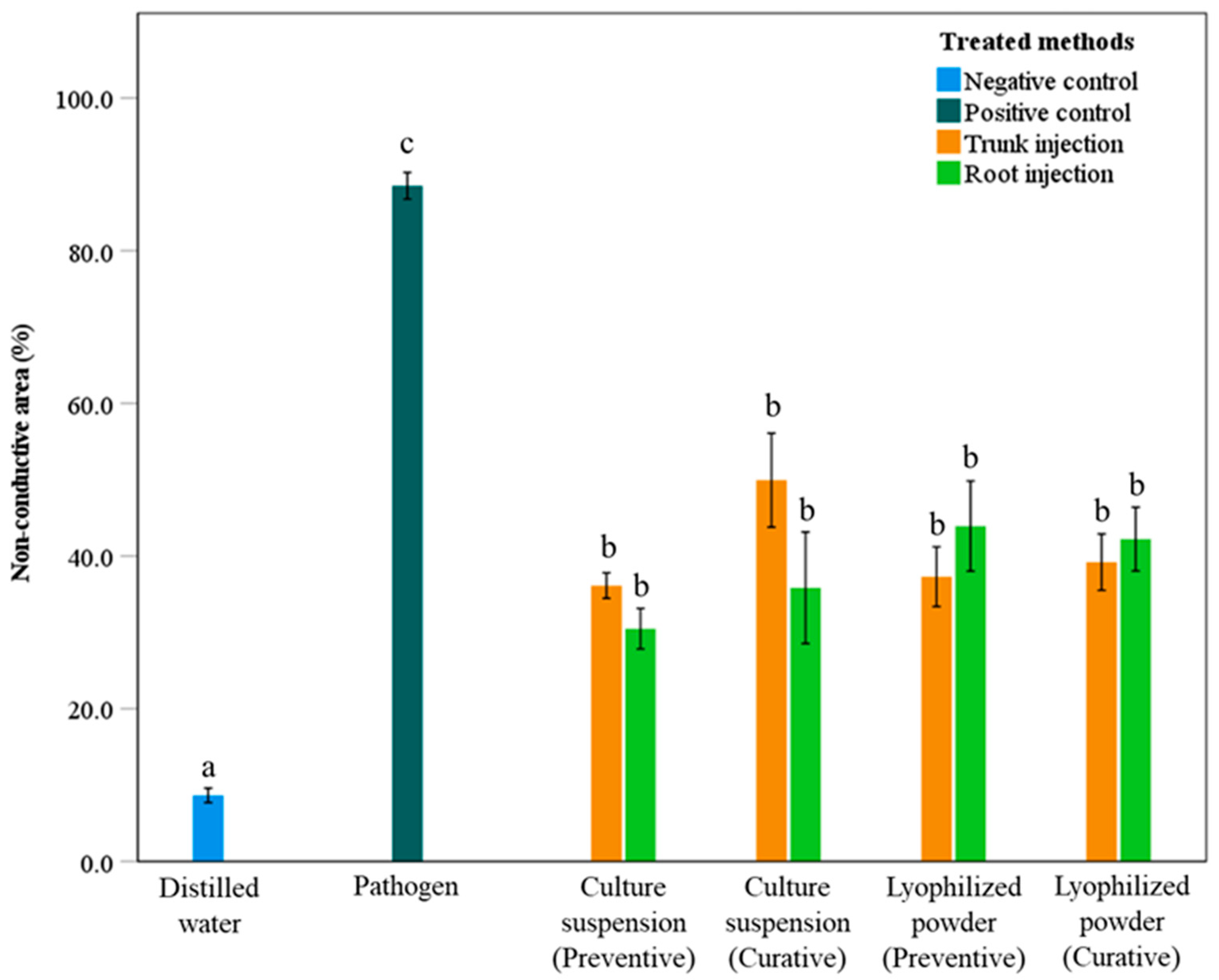
The NCA values for treatments with culture suspension and lyophilized powder of S. blastmyceticus are presented in Figure 4. Treatments using culture suspension and lyophilized powder showed significant differences (p < 0.05) compared with the positive and negative controls (Figure 4). The pathogen and distilled water treatments exhibited the highest and lowest NCA values of 88.5% and 8.7%, respectively. The remaining treatments showed NCA values ranging from 30.5% to 49.9%. There were no significant differences (p > 0.05) between preventive and curative treatments. In the trunk injection, curative treatments of the culture suspension and lyophilized powder resulted in higher NCA values than preventive treatments, with NCA values of 49.9% and 36.1% for curative treatments and 39.2% and 37.3% for preventive treatments, respectively (Figure 4). In root injection, the curative treatment using culture suspension showed a higher NCA value (35.9%) than the preventive treatment (30.5%). On the other hand, the curative treatment using lyophilized powder showed a slightly lower NCA values (42.2%) than the preventive treatment (43.9%) (Figure 4).
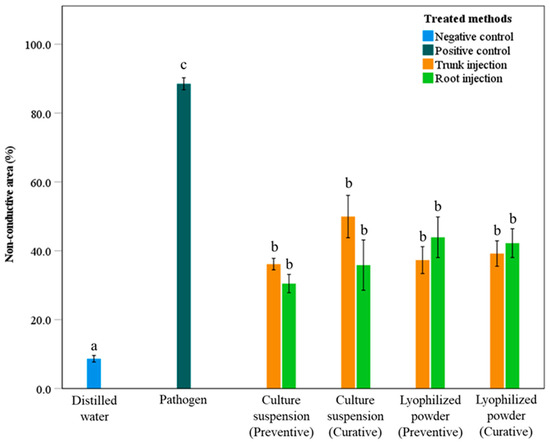
Figure 4.
Percentage of non-conductive areas in the sapwood of Mongolian oak trees after preventive and curative treatments with culture suspension or lyophilized powder of S. blastmyceticus, performed as trunk or root injections in Uiwang, Gyeonggi province. The vertical bars illustrate the values of the standard error of the means. Different letters indicate a significant difference (p < 0.05) among treatments by Tukey’s HSD test.
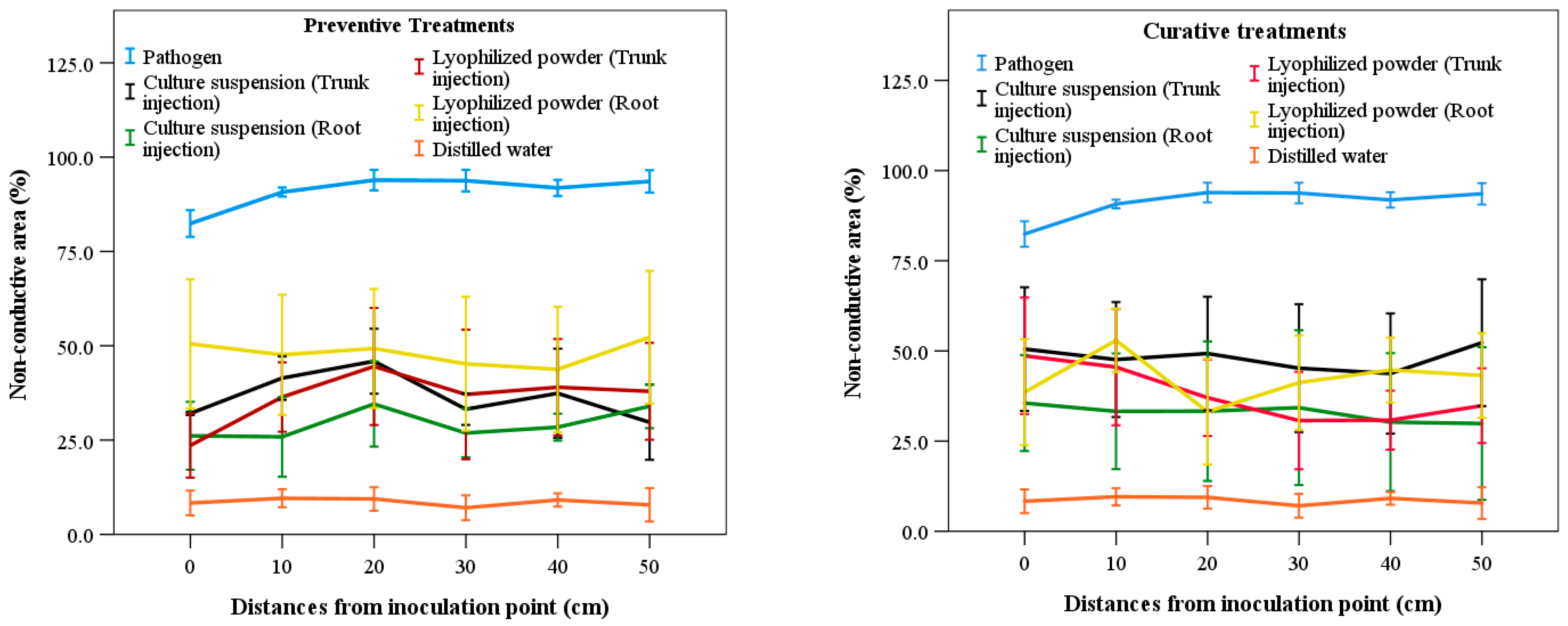
Analysis indicated no significant differences (p > 0.05) among treatments in terms of different distances from the pathogen inoculation point, regardless of whether treatments were preventive or curative (Figure 5). Compared with the distilled water treatment, all treatments using the culture suspension and lyophilized powder of S. blastmyceticus produced higher NCA values but lower values than the pathogen-only treatment. As the distance from the inoculation point increased, NCA values fluctuated among preventive and curative treatments. The pathogen-only injection treatments showed the highest NCA values at three different distance intervals (0–10 cm, 20–30 cm, and 40–50 cm), with NCA values of 70.32%, 73.26%, and 74.87%, respectively. In contrast, the distilled water treatment showed the lowest NCA values of 9.40%, 7.60%, and 7.84% at the same distances (Figure 5). In preventive treatments, the lyophilized powder produced the highest NCA value (48.54%) at 40–50 cm, while the culture suspension showed the lowest NCA value (24.47%) at 0–10 cm in the root injection method. In curative treatments, the culture suspension showed the highest and lowest NCA values at 40–50 cm, with 48.54% in trunk injection and 25.88% in root injection (Figure 5).
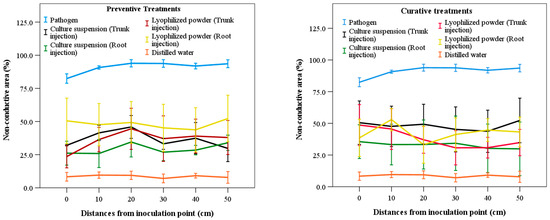
Figure 5.
Non-conductive area according to distances from the inoculation position of the pathogen among treatments of culture suspension and lyophilized powder of S. blastmyceticus in Uiwang, Gyeonggi province. The vertical bars illustrate the values of the standard error of the means. Preventive treatments (left) and curative treatments (right).
4. Discussion
In the experiment conducted in Chuncheon, Kangwon Province, preventive treatment with propiconazole showed significant differences between trunk and root flare injection methods. Curative injection of propiconazole was more effective than preventive injection, and root injection showed greater efficacy than trunk injection (Figure 2). Several treatments using the culture suspension of S. blastmyceticus showed no significant differences compared with propiconazole treatments. Furthermore, there were no significant differences among the various culture suspension treatments (Figure 2). These suggest that the culture suspension of S. blastmyceticus could serve as a potential biocontrol agent against the oak wilt pathogen, D. quercus-mongolicae. The ability of S. blastmyceticus culture suspension to suppress oak wilt disease through tree injection has also been demonstrated previously [21]. However, climatic characteristics and ecological conditions may influence the inhibitory effects of microbial metabolites against the oak wilt pathogen. For example, secondary metabolites derived from Streptomyces spp. have shown variable inhibitory effects on the plant pathogen Botrytis cinerea depending on environmental conditions such as temperature, light-dark cycles, and culture media used in the experiments [28]. Similarly, differences in NCA values between the Chuncheon (423 m elevation) and Uiwang (196 m elevation) sites may be partially attributed to site-specific environmental factors such as altitude, temperature regime, and microclimatic conditions. Higher elevation sites may experience cooler temperatures and different moisture dynamics, which can influence both pathogen colonization and host vascular responses. Such environmental variability may have affected the spatial progression of non-conductive sapwood formation and treatment efficacy. In the experiment conducted in Uiwang, Gyeonggi Province, similar efficacy was observed between treatments using culture suspension and lyophilized powder of S. blastmyceticus, with no significant differences detected among treatments. However, the lyophilized powder showed a weaker inhibitory effect when applied through root injection compared with trunk injection (Figure 4). Additionally, the lyophilized powder of S. blastmyceticus remained viable for up to 8 months when stored at 4 °C [21]. Therefore, these results indicate that the lyophilized powder formulation of S. blastmyceticus shows promise as a potential biocontrol formulation; however, comprehensive shelf-life, stability, and large-scale field validation studies are required before commercialization can be considered.
Powder formulations of microorganisms have also been used to control plant pathogens in various agricultural systems worldwide. For example, powdered Meyerozyma caribbica has been used to control Colletotrichum gloeosporioides, the causal agent of anthracnose in mango fruit. The powdered formulation showed significant disease suppression compared with the control treatment; however, it produced a smaller reduction in lesion diameter on mango fruit than fresh yeast cells of M. caribbica, with reductions of 23.90% and 36.33%, respectively [18]. The effectiveness of fresh cells or powder formulations strongly depends on storage temperature and conditions, which influence the population dynamics and viability of microbial agents [18]. Another study utilized spray-dried spores of Candida sake to control Penicillium expansum, the causal agent of blue mold rot disease in apples. However, the powder formulation was less effective than fresh cells because spore survival was negatively affected by drying temperature [29]. The efficacy of a dry powder formulation of Talaromyces tratensis against dirty panicle disease in rice varied depending on whether the formulation was applied as a single or double application under field conditions [16]. The T. tratensis powder formulation reduced disease incidence by 44.62% and 41.08% when applied at concentrations of 5% and 1%, respectively [16]. However, the disease suppression achieved by the powder formulation was significantly lower than that of the mixed fungicide (difenoconazole + propiconazole). Nevertheless, there were no significant differences in rice yield obtained from these two treatments [16].
Streptomyces blastmyceticus has been reported as a potential biological control agent against various plant pathogens. S. blastmyceticus can colonize plant vascular tissues and protect host plants through multiple mechanisms, including antibiotic production, secretion of lytic enzymes, induction of systemic resistance (ISR), and modulation of plant hormone pathways [30]. Previous studies showed that S. blastmyceticus strongly inhibits peach brown rot caused by Monilinia fructicola and produces hydrolytic enzymes such as chitinase, cellulase, and protease [31]. S. blastmyceticus isolated from forest soil has been shown to inhibit several plant pathogens, including Colletotrichum acutatum, C. coccodes, C. gleosporioides, Fusarium oxysporum, and Trichothecium roseum [32]. In particular, the n-butanol extract of S. blastmyceticus completely inhibited spore germination of C. acutatum and F. oxysporum at a concentration of 0.8 µg/mL [32]. Streptomyces noursei, a remarkable species within the Streptomyces genus, is known to produce diverse bioactive compounds. These compounds exhibit strong efficacy against a wide range of plant pathogens, such as Alternaria alternata, Botrytis cinerea, F. oxysporum, and Phytophthora capsici. This highlights the potential of S. noursei as a valuable resource for combating plant diseases and improving crop health [33]. Two natural compounds, phenylpropanoid and cis-p-menthane-7,8-diol, were isolated from Streptomyces sp. BC129–2 and showed strong antifungal activity against C. gloeosporioides, Fusarium sp. and Neopestalotiopsis sp. [34]. Most previous studies have focused on the secondary metabolites produced by Streptomyces species. However, recent studies have shown that Streptomyces species can also inhibit plant pathogens through volatile secondary metabolites. For example, volatile compounds produced by S. philanthi have been reported to effectively suppress rice sheath blight disease caused by Rhizoctonia solani [35]. Another study indicated that the level of antifungal activity of volatile organic compounds (VOCs) depends on culture conditions such as incubation volume and duration [36].
In this study, both the culture suspension and lyophilized powder of S. blastmyceticus showed comparable efficacy in controlling the oak wilt pathogen D. quercus-mongolicae. In the trunk injection treatment, the colonization rate of oak wilt fungus was lowest with the preventive treatment using lyophilized powder (Table 1). Consequently, the effectiveness of lyophilized powder in controlling the colonization rate of oak wilt fungus was highest at 82.91% (Table 1). These findings suggest that the lyophilized powder formulation of S. blastmyceticus has potential for development as a biological control product with long-term storage stability. However, comprehensive shelf-life, stability, and large-scale field validation studies are required before commercialization can be considered. Further studies are also required to elucidate the inhibitory mechanism and to identify antifungal compounds produced by S. blastmyceticus that are active against the oak wilt pathogen.
5. Conclusions
The culture suspension and lyophilized powder formulations of S. blastmyceticus efficiently suppressed the growth of the oak wilt pathogen in trees and may serve as an alternative management strategy. Among the tested treatments, preventive trunk injection with lyophilized powder was the most effective, resulting in a colonization rate of oak wilt fungus of only 9.72% and a control value reaching 82.91%. Overall, trunk injection method was more effective than root injection, and preventive treatment showed greater efficacy than curative treatments. The lyophilized powder formulation of S. blastmyceticus has potential for development into commercial biological products with long-term storage stability. This formulation could therefore serve as a promising biocontrol agent against Dryadomyces quercus-mongolicae.
Author Contributions
Conceptualization, D.-H.L. and K.S.; methodology, J.H.L., M.H.N., D.-H.L. and K.S.; software, J.H.L., M.H.N. and K.S.; validation, J.H.L., M.H.N., D.-H.L. and K.S.; formal analysis, J.H.L. and M.H.N.; investigation, J.H.L. and M.H.N.; resources, J.K.L. and K.S.; data curation, J.H.L., M.H.N. and D.-H.L.; writing—original draft preparation, J.H.L. and M.H.N.; writing—review and editing, D.-H.L. and K.S.; supervision, J.K.L., D.-H.L. and K.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Research Foundation of Korea (NRF) (No. RS-2023-00250589).
Data Availability Statement
The data presented in this study are available upon request from the corresponding authors. The data are not publicly available due to institutional policy.
Conflicts of Interest
The authors declare that they have no competing financial interests related to personal relationships that could have influenced the work reported in this study.
References
- Jaber, L.R.; Ownley, B.H. Can We Use Entomopathogenic Fungi as Endophytes for Dual Biological Control of Insect Pests and Plant Pathogens? Biol. Control 2018, 116, 36–45. [Google Scholar] [CrossRef]
- Reverchon, F.; Contreras-Ramos, S.M.; Eskalen, A.; Guerrero-Analco, J.A.; Quiñones-Aguilar, E.E.; Rios-Velasco, C.; Velázquez-Fernández, J.B. Microbial Biocontrol Strategies for Ambrosia Beetles and Their Associated Phytopathogenic Fungi. Front. Sustain. Food Syst. 2021, 5, 737977. [Google Scholar] [CrossRef]
- Imoulan, A.; Hussain, M.; Kirk, P.M.; El Meziane, A.; Yao, Y.-J. Entomopathogenic Fungus Beauveria: Host Specificity, Ecology and Significance of Morpho-Molecular Characterization in Accurate Taxonomic Classification. J. Asia-Pac. Entomol. 2017, 20, 1204–1212. [Google Scholar] [CrossRef]
- Karthi, S.; Vasantha-Srinivasan, P.; Senthil-Nathan, S.; Han, Y.S.; Shivakumar, M.S.; Murali-Baskaran, R.K.; Kalaivani, K.; Radhakrishnan, N.; Park, K.B.; Malafaia, G. Entomopathogenic Fungi Promising Biocontrol Agents for Managing Lepidopteran Pests: Review of Current Knowledge. Biocatal. Agric. Biotechnol. 2024, 58, 103146. [Google Scholar] [CrossRef]
- Ali, M.A.; Ahmed, T.; Ibrahim, E.; Rizwan, M.; Chong, K.P.; Yong, J.W.H. A Review on Mechanisms and Prospects of Endophytic Bacteria in Biocontrol of Plant Pathogenic Fungi and Their Plant Growth-Promoting Activities. Heliyon 2024, 10, e31573. [Google Scholar] [CrossRef]
- Etesami, H.; Jeong, B.R.; Glick, B.R. Biocontrol of Plant Diseases by Bacillus spp. Physiol. Mol. Plant Pathol. 2023, 126, 102048. [Google Scholar] [CrossRef]
- Nazari, M.T.; Schommer, V.A.; Braun, J.C.A.; Dos Santos, L.F.; Lopes, S.T.; Simon, V.; Machado, B.S.; Ferrari, V.; Colla, L.M.; Piccin, J.S. Using Streptomyces spp. as Plant Growth Promoters and Biocontrol Agents. Rhizosphere 2023, 27, 100741. [Google Scholar] [CrossRef]
- Poveda, J.; Baptista, P. Filamentous Fungi as Biocontrol Agents in Olive (Olea europaea L.) Diseases: Mycorrhizal and Endophytic Fungi. Crop Prot. 2021, 146, 105672. [Google Scholar] [CrossRef]
- Cai, F.; Yang, C.; Ma, T.; Osei, R.; Jin, M.; Zhang, C.; Wang, Y. An Endophytic Paenibacillus polymyxa Hg18 and Its Biocontrol Potential against Fusarium oxysporum f. sp. cucumerinum. Biol. Control 2024, 188, 105380. [Google Scholar] [CrossRef]
- González-Gutiérrez, K.N.; Ragazzo-Sánchez, J.A.; Calderón-Santoyo, M. Bioformulation of Yamadazyma mexicana LPa14 by Electrospraying Process: Anthracnose Control and Effect on Postharvest Quality of Avocado Fruit. Biol. Control 2024, 190, 105449. [Google Scholar] [CrossRef]
- González-Gutiérrez, K.N.; Ragazzo-Sánchez, J.A.; Barros-Castillo, J.C.; Narváez-Zapata, J.A.; Calderón-Santoyo, M. Yeasts with Potential Biocontrol of Colletotrichum gloeosporioides in Avocado (Persea americana Mill. Cv. Hass) and Characterization of Yamadazyma mexicana Mechanisms. Eur. J. Plant Pathol. 2023, 165, 525–543. [Google Scholar] [CrossRef]
- Patel, J.K.; Madaan, S.; Archana, G. Antibiotic Producing Endophytic Streptomyces spp. Colonize above-Ground Plant Parts and Promote Shoot Growth in Multiple Healthy and Pathogen-Challenged Cereal Crops. Microbiol. Res. 2018, 215, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Borges, D.F.; Lopes, E.A.; Fialho Moraes, A.R.; Soares, M.S.; Visôtto, L.E.; Oliveira, C.R.; Moreira Valente, V.M. Formulation of Botanicals for the Control of Plant-Pathogens: A Review. Crop Prot. 2018, 110, 135–140. [Google Scholar] [CrossRef]
- Limdolthamand, S.; Songkumarn, P.; Suwannarat, S.; Jantasorn, A.; Dethoup, T. Biocontrol Efficacy of Endophytic Trichoderma spp. in Fresh and Dry Powder Formulations in Controlling Northern Corn Leaf Blight in Sweet Corn. Biol. Control 2023, 181, 105217. [Google Scholar] [CrossRef]
- Tamreihao, K.; Ningthoujam, D.S.; Nimaichand, S.; Singh, E.S.; Reena, P.; Singh, S.H.; Nongthomba, U. Biocontrol and Plant Growth Promoting Activities of a Streptomyces corchorusii Strain UCR3-16 and Preparation of Powder Formulation for Application as Biofertilizer Agents for Rice Plant. Microbiol. Res. 2016, 192, 260–270. [Google Scholar] [CrossRef]
- Eakjamnong, W.; Keawsalong, N.; Dethoup, T. Novel Ready-to-Use Dry Powder Formulation of Talaromyces tratensis KUFA0091 to Control Dirty Panicle Disease in Rice. Biol. Control 2021, 152, 104454. [Google Scholar] [CrossRef]
- Aguirre-Güitrón, L.; Calderón-Santoyo, M.; Ortiz-Basurto, R.I.; Bautista-Rosales, P.U.; Ragazzo-Sánchez, J.A. Optimisation of the Spray Drying Process of Formulating the Post-Harvest Biocontrol Agent Meyerozyma caribbica. Biocontrol Sci. Technol. 2018, 28, 574–590. [Google Scholar] [CrossRef]
- Aguirre-Güitrón, L.; Calderón-Santoyo, M.; Bautista-Rosales, P.U.; Ragazzo-Sánchez, J.A. Application of Powder Formulation of Meyerozyma Caribbica for Postharvest Control of Colletotrichum Gloeosporioides in Mango (Mangifera indica L.). LWT 2019, 113, 108271. [Google Scholar] [CrossRef]
- Kim, K.-H.; Choi, Y.-J.; Seo, S.-T.; Shin, H.-D. Raffaelea quercus-mongolicae sp. Nov. Associated with Platypus koryoensis on Oak in Korea. Mycotaxon 2009, 110, 189–197. [Google Scholar] [CrossRef]
- De Beer, Z.W.; Procter, M.; Wingfield, M.J.; Marincowitz, S.; Duong, T.A. Generic Boundaries in the Ophiostomatales Reconsidered and Revised. Stud. Mycol. 2022, 101, 57–120. [Google Scholar] [CrossRef]
- Hong, A.R.; Yun, J.H.; Yi, S.H.; Lee, J.H.; Seo, S.T.; Lee, J.K. Screening of Antifungal Microorganisms with Strong Biological Activity against Oak Wilt Fungus, Raffaelea quercus-mongolicae. J. For. Environ. Sci. 2018, 34, 395–404. [Google Scholar] [CrossRef]
- Lee, J.H.; Hong, A.R.; Yun, J.H.; Seo, S.T.; Lee, J.K. Prevention of Oak Wilt by Tree Injection of Culture Suspension of an Antifungal Microorganism, Streptomyces blastmyceticus against Oak Wilt Fungus, Raffaelea quercus-mongolicae. J. For. Environ. Sci. 2018, 34, 376–381. [Google Scholar] [CrossRef]
- Nguyen, M.H.; Yong, J.H.; Sung, H.J.; Lee, J.K. Screening of Endophytic Fungal Isolates Against Raffaelea quercus-mongolicae Causing Oak Wilt Disease in Korea. Mycobiology 2020, 48, 484–494. [Google Scholar] [CrossRef]
- Nguyen, M.H.; Park, I.-K.; Lee, J.K.; Lee, D.-H.; Shin, K. Antifungal Activity of Culture Filtrate from Endophytic Fungus Nectria balsamea E282 and Its Fractions against Dryadomyces quercus-mongolicae. Forests 2024, 15, 332. [Google Scholar] [CrossRef]
- Park, I.-K.; Nam, Y.; Seo, S.-T.; Kim, S.-W.; Jung, C.-S.; Han, H.-R. Development of a Mass Trapping Device for the Ambrosia Beetle, Platypus koryoensis, an Insect Vector of Oak Wilt Disease in Korea. J. Asia-Pac. Entomol. 2016, 19, 39–43. [Google Scholar] [CrossRef]
- Kurth, F.; Mailänder, S.; Bönn, M.; Feldhahn, L.; Herrmann, S.; Große, I.; Buscot, F.; Schrey, S.D.; Tarkka, M.T. Streptomyces-Induced Resistance Against Oak Powdery Mildew Involves Host Plant Responses in Defense, Photosynthesis, and Secondary Metabolism Pathways. Mol. Plant-Microbe Interact. 2014, 27, 891–900. [Google Scholar] [CrossRef]
- Torii, M.; Matsuda, Y.; Seo, S.T.; Kim, K.H.; Ito, S.; Moon, M.J.; Kim, S.H.; Yamada, T. The Effect of Raffaelea quercus-mongolicae Inoculations on the Formation of Non-Conductive Sapwood of Quercus Mongolica. Mycobiology 2014, 42, 210–214. [Google Scholar] [CrossRef]
- Boukaew, S.; Yossan, S.; Cheirsilp, B.; Prasertsan, P. Impact of Environmental Factors on Streptomyces spp. Metabolites against Botrytis cinerea. J. Basic Microbiol. 2022, 62, 611–622. [Google Scholar] [CrossRef]
- Abadias, M.; Teixidó, N.; Usall, J.; Solsona, C.; Viñas, I. Survival of the Postharvest Biocontrol Yeast Candida sake CPA-1 after Dehydration by Spray-Drying. Biocontrol Sci. Technol. 2005, 15, 835–846. [Google Scholar] [CrossRef]
- Khan, S.; Srivastava, S.; Karnwal, A.; Malik, T. Streptomyces as a Promising Biological Control Agents for Plant Pathogens. Front. Microbiol. 2023, 14, 1285543. [Google Scholar] [CrossRef]
- Ni, M.; Wu, Q.; Wang, H.; Liu, W.; Hu, B.; Zhang, D.; Zhao, J.; Liu, D.; Lu, C. Identification of a Novel Strain, Streptomyces blastmyceticus JZB130180, and Evaluation of Its Biocontrol Efficacy against Monilinia fructicola. J. Zhejiang Univ. Sci. B 2019, 20, 84–94. [Google Scholar] [CrossRef]
- Kim, Y.J.; Kim, J.; Rho, J.-Y. Antifungal Activities of Streptomyces blastmyceticus Strain 12-6 Against Plant Pathogenic Fungi. Mycobiology 2019, 47, 329–334. [Google Scholar] [CrossRef]
- Lu, Y.; Song, W.; Wang, J.; Cao, Y.; Han, X.; Xu, C.; Wang, F.; Ge, B. Biocontrol of Botrytis cinerea by Streptomyces noursei C27 and Preliminary Identification of Antimicrobial Metabolites. Biol. Control 2024, 196, 105561. [Google Scholar] [CrossRef]
- Cheng, M.-J.; Chen, J.-J.; Liu, H.-W.; Leu, J.-Y.; Wu, M.-D.; Tseng, M. Isolation and Antifungal Activity Assessment of Natural Compounds from the Culture Medium of Actinobacterium Streptomyces sp. BC129-2. Phytochem. Lett. 2024, 64, 1–5. [Google Scholar] [CrossRef]
- Boukaew, S.; Plubrukam, A.; Prasertsan, P. Effect of Volatile Substances from Streptomyces philanthi RM-1-138 on Growth of Rhizoctonia solani on Rice Leaf. BioControl 2013, 58, 471–482. [Google Scholar] [CrossRef]
- Li, Q.; Ning, P.; Zheng, L.; Huang, J.; Li, G.; Hsiang, T. Fumigant Activity of Volatiles of Streptomyces Globisporus JK-1 against Penicillium italicum on Citrus microcarpa. Postharvest Biol. Technol. 2010, 58, 157–165. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.