Abstract
Prunus sibirica L. is a key ecological and economic tree species in northern China that is threatened by habitat degradation due to climate change and human activities. To address the gaps of incomplete historical dynamics and lack of conservation integration in existing studies, we integrated MaxEnt and Zonation v4.0 to predict its suitable habitat across five periods (LIG to 2090s) and three CMIP6 SSP scenarios, identifying key drivers and priority conservation zones. The model showed high prediction accuracy (mean AUC > 0.9). Results indicated that Human Footprint (HFP), Precipitation Seasonality (Bio15), Annual Mean Temperature (Bio1), Elevation (ELEV), and Mean Temperature of the Coldest Quarter (Bio11) were the key environmental factors (cumulative contribution 91.4%), with Bio1, Bio15, Temperature Seasonality (Bio4), and HFP confirmed as major drivers (AUC > 0.8) via jackknife test. Spatiotemporally, the species’ suitable habitat contracted from the Last Interglacial to the Last Glacial Maximum and expanded to the current total suitable area of 506,620.1 km2. Under future SSP scenarios, suitable habitats expanded continuously under SSP126 and SSP245 but showed a “first expansion then contraction” trend under SSP585, with a persistent northeastward migration of the habitat centroid. The vertical (altitudinal) distribution of P. sibirica showed a trend of moving to higher elevations under future warming scenarios, especially in the SSP585 scenario. High-priority conservation zones are concentrated in northern China with insufficient existing protection. It is emphasized that this study contributes to improving the adaptive capacity and genetic characterization of P. sibirica almond populations to future climate.
1. Introduction
The response of vegetation to climate and environmental changes has emerged as a focal issue in global change and biogeography research. Climate is widely recognized as one of the key factors regulating vegetation distribution across regional and global scales [1]. The interactions between climate, topography, soil, and other environmental factors not only shape the physiological traits of plants but also modulate their growth and geographical distribution [2]. Leveraging vegetation adaptability to these factors to investigate their potential geographical distribution facilitates the enhancement of vegetation protection and management strategies [3]. Additionally, shifts in potential geographical distribution can indirectly indicate variations in climate and other environmental drivers.
Numerous Species Distribution Models (SDMs) have been extensively applied in diverse research fields worldwide, including HABITAT, DOMAIN, Ecological Niche Factor Analysis (ENFA), Mahalanobis Distance, Border Function, and Maximum Entropy (MaxEnt) [4]. Among the commonly used SDMs for predicting species’ potential geographical distribution, MaxEnt stands out due to its convenience, efficiency, and practicality, with superior performance in estimating potential suitable ranges [5]. The model operates by integrating species occurrence data and environmental variables. It simulates the potential geographical distribution of target species based on the constraints of their current habitat conditions, ultimately outputting predictions in the form of probability values [6]. After years of optimization and validation by researchers, MaxEnt has achieved satisfactory results in species distribution prediction. It accurately estimates suitable habitats while offering user-friendly operation and reliable simulation effects [7]. Consequently, MaxEnt has been widely adopted in studies related to species distribution and potential suitable area assessment. It provides scientific support for ecosystem conservation, understanding species formation mechanisms, tracking geographical distribution changes, and formulating germplasm resource management strategies and climate change adaptation measures [8,9].
Vegetation response to climate change is a focal issue in global change research, with climate as a key regulator of plant distribution [1]. Species Distribution Models (SDMs) such as MaxEnt are widely used for predicting potential habitats due to high accuracy and operability [5]. Prunus sibirica a plant belonging to Prunoideae of Rosaceae, Armeniaca, is commonly known as mountain apricot or Mongolian apricot [10]. Possessing strong adaptability, cold resistance, and drought tolerance, P. sibirica serves as a key ecological tree species for windbreak and sand fixation. It is naturally distributed in China’s Three-North Regions [11], where it plays a vital role in maintaining regional ecological balance and preventing desertification. Additionally, its almonds are rich in proteins, vitamins, dietary fiber, minerals, and various essential trace elements [12]. This makes P. sibirica a valuable economic tree species and a popular commercial dried fruit, driving the rapid development of related industries in northern China [13]. However, P. sibirica habitats—primarily distributed in semi-arid sandy plains, mountains, and hills with inherently poor soil conditions [14]—have been severely degraded in recent years. Climate change-induced temperature increases and precipitation variability, coupled with human activities such as infrastructure construction and deforestation, have led to a continuous decline in its distribution range and population size [15]. The growing market demand for P. sibirica products further intensifies the pressure on its natural resources, creating an urgent need to balance conservation and industrial development.
Existing studies on P. sibirica distribution focus primarily on current or single-scenario future predictions, lacking a coherent analysis of historical migration. No future multi-scenario dynamics or targeted conservation zoning have been designed [16]. This study addresses three core objectives using the MaxEnt model and Zonation v4.0: (1) identify the key environmental factors regulating P. sibirica geographical distribution; (2) Providing a comprehensive historical dynamic trajectory of P. sibirica through five key climatic periods, including Last Interglacial, Last Glacial Maximum, Middle Holocene, Current Climate, and Future climate—this study comprehensively reveals the historical dynamic trajectory of P. sibirica. The selection of these periods is based on two considerations: first, these periods represent critical climate turning points in the Quaternary, which have had profound impacts on the distribution of temperate plant species [17,18]; second, these periods are widely used in species distribution modeling studies, facilitating comparison with the results of similar studies [19,20]. (3) Determine priority conservation zones and propose targeted strategies for germplasm resource protection, industrial development, and protected area network optimization. This research enhances the understanding of P. sibirica adaptive responses to climate change and provides a scientific basis for formulating evidence-based protection policies, guiding rational plantation layout, and promoting the sustainable development of the P. sibirica industry.
2. Data Source and Preprocessing
2.1. Data Collection and Screening
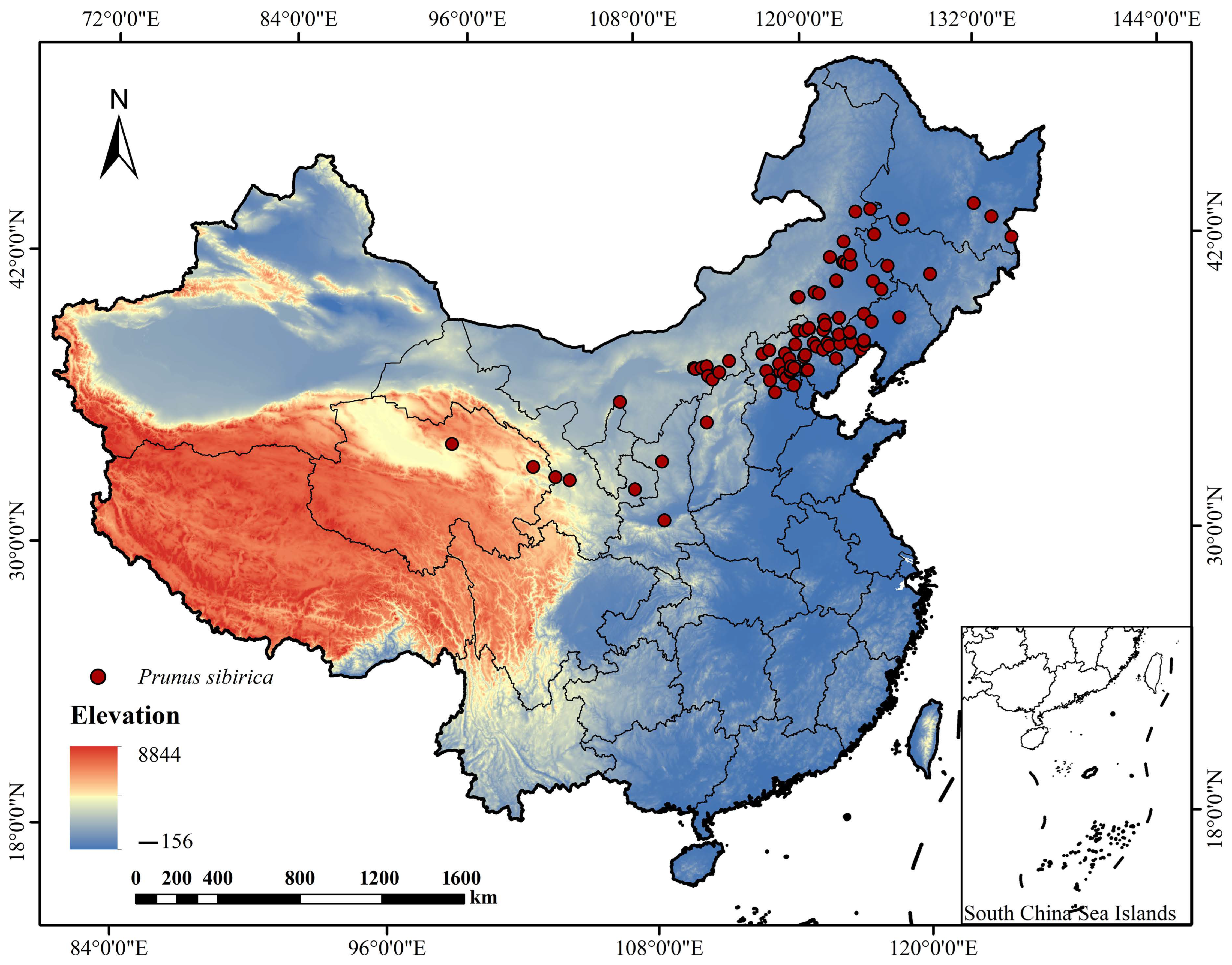
To obtain accurate and representative distribution data of P. sibirica, we combined literature review, herbarium database query, and standardized field surveys. Through querying relevant literature and field investigation, the main geographical distribution samples of P. sibirica in China were collected. The data came from plant specimens such as National Herbarium NSII, China Digital Herbarium CHV, the global biodiversity information network database GBIF (https://www.gbif.org/citation-guidelines, last accessed on 1 April 2025), and the Chinese Plant Image Library [21]. The criteria for selecting specimen data are as follows: (1) only specimens with clear collection locations (accuracy to the county level) and collection dates (after 1950) were retained; (2) cultivated individuals were excluded to ensure the data represented natural distribution populations. Combined with our research group, field surveys were conducted from 2021 to 2023 in the main natural distribution areas of P. sibirica, including Inner Mongolia, Shaanxi, Hebei, Shanxi, Liaoning, and Jilin. The survey method adopted a grid sampling strategy with a grid size of 10 km × 10 km, and the presence/absence of P. sibirica was recorded in each grid. A total of 116 distribution data were systematically investigated and recorded from the natural distribution area of P. sibirica in Inner Mongolia, Shaanxi, Hebei Shanxi, and other places; referring to the method of Zhang [22], all the above records are filtered, except for natural records and redundant data, which were deleted (Supplementary Table S1). To reduce the errors caused by the cluster effect, in each grid (2.5′, 2.5 km × 2.5 km) only one distribution point was retained, and 87 effective samples were finally obtained (Figure 1).
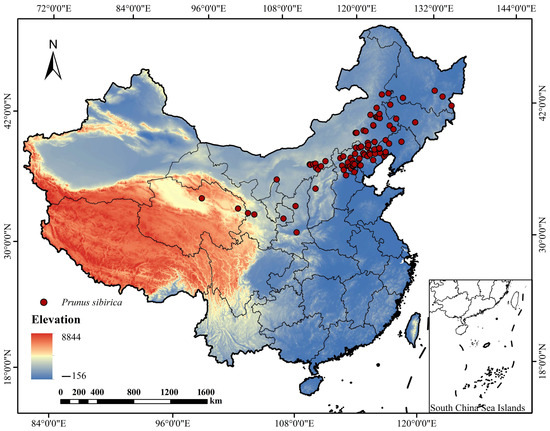
Figure 1.
Location of P. sibirica sampling site distribution.
2.2. Environmental Variable Screening and Distribution Point Data Processing
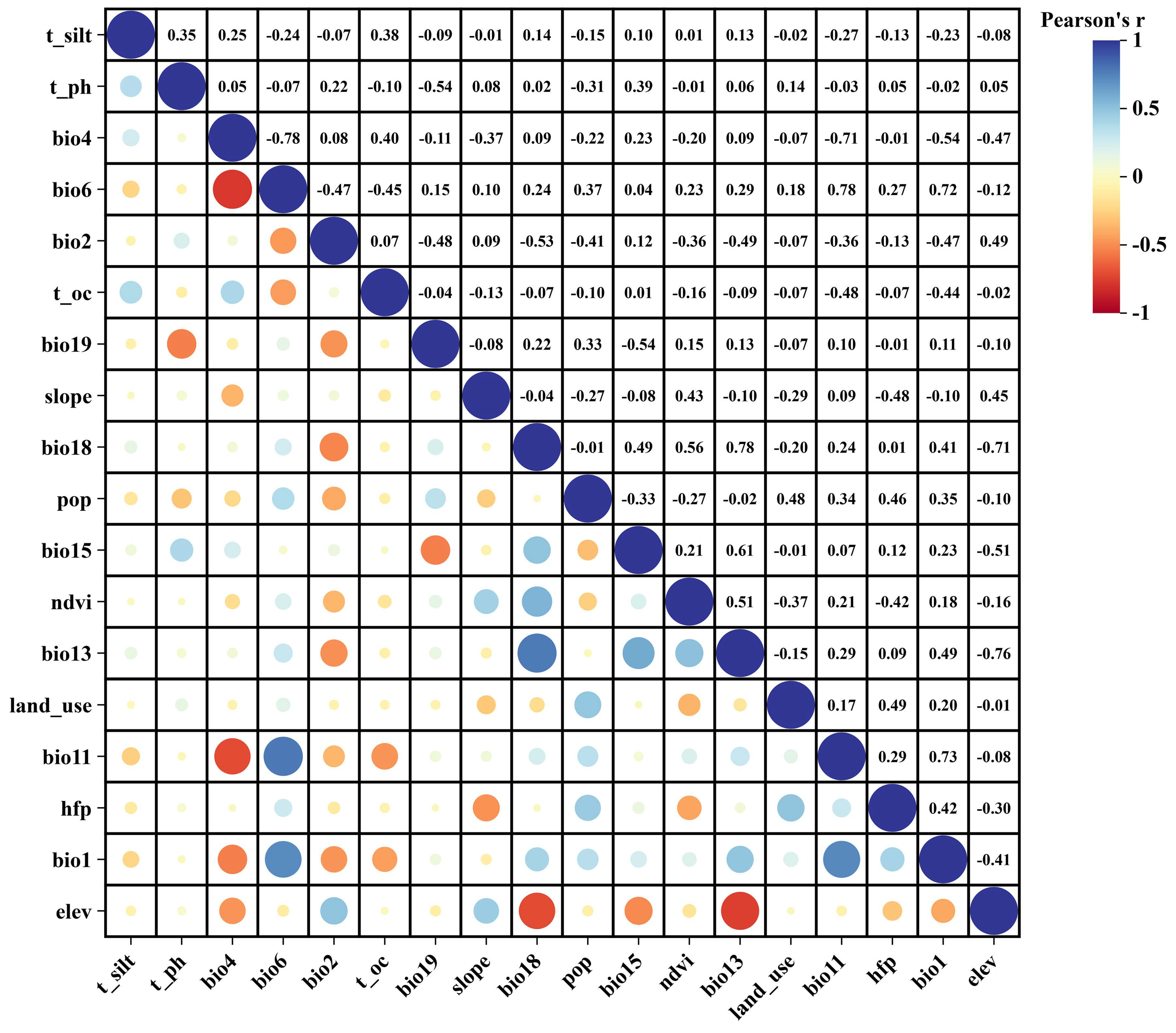
Based on existing P. sibirica habitat studies, 40 environmental variables were considered, including 19 climatic factors, 14 soil factors, 3 topographic variables (Elevation, Slope, Aspect), two vegetation variables (NDVI, land-use type) and two anthropogenic variables (Human footprint, Population density). Current, Last Inter Glacial (Last Inter Glacial, 120,000–140,000 years before present), Last Glacial Maximum (Last Glacial Maximum, 22,000 years before present), Mid-Holocene (about 6000 years ago), and the future-2050s (2041−2050s) and 2090s (2051−2090s) information was obtained from the WorldClim database [23,24]. The Beijing Climate Center Climate System Model (BCC-CSM2-MR) provides future climate data for three shared socioeconomic pathway (SSP) scenarios: SSP126, SSP245, and SSP585, which correspond to low, medium, and high levels of greenhouse gas emissions, respectively [25]. The Food and Agriculture Organization of the United Nations World Soil Database (HWSD) provided the soil and topography information used in this study [26]. All variables had a standardized spatial resolution of 2.5 arcmin, representing an approximate coverage of 2.5 km. To mitigate the effects of multicollinearity on the accuracy of predictions, we used the R software (version 4.1.3) to perform Spearman correlation analysis and Variance Inflation Factor (VIF) evaluations on all examined environmental variables. This study chose those environmental variables that exhibited a Spearman correlation coefficient below 0.7 and a VIF lower than 5 [27,28,29], this step retained 21 variables (Supplementary Table S2). The variable set was further refined by a strict correlation threshold. To reduce the potential impact of residual collinearity on model accuracy, we calculated the Spearman’s correlation coefficient of the 21 variables retained in Step 1 again and excluded variables with |r| > 0.80, resulting in a final selection of 18 environmental variables for predicting the habitat distribution of P. sibirica (Figure 2).
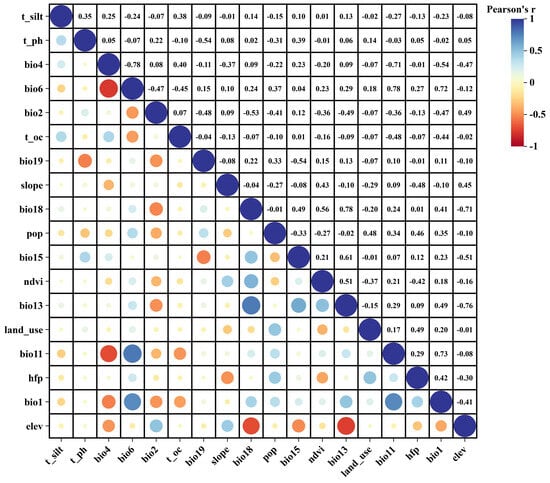
Figure 2.
Spearman correlation test among the 18 selected environmental variables. Positive correlations are displayed in blue and negative correlations in a red color. The color intensity and the size of the circle are proportional to the correlation coefficients.
2.3. Model Optimization and Accuracy Evaluation
A total of 87 valid in situ occurrence points for P. sibirica were compiled from field surveys and the published literature as species distribution data. To enhance prediction reliability and mitigate overfitting, the MaxEnt (v3.4.4) model was optimized using the R-package ‘ENMeval’ (v2.0.5) by adjusting two key parameters: Feature Combination (FC) and Regularization Multiplier (RM) [30,31,32]. Six FC combinations (L, LQ, H, LQH, LQHP, LQHPT) were selected from the five MaxEnt feature types (linear L, quadratic Q, hinge H, product P, threshold T), and RM was set to range from 0.5 to 6 with 0.5 increments. The model underwent 10 iterations with 10-fold cross-validation, employing a standard 75% training/25% testing data split, 10,000 background points, and 5000 iterations. Model performance was evaluated using delta Akaike Information Criterion (AIC) for fit and complexity, and the area under the receiver operating characteristic (ROC) curve (AUC) for predictive accuracy [7]. The average results of the 10 runs were employed for subsequent analysis to ensure robustness.
2.4. Potentially Suitable Area Partitions
To analyze the dynamic changes in P. sibirica distribution range across different periods, the average simulation results were imported into ArcGIS 10.4 software and converted into raster layers. The classification threshold was determined by combining the natural break method with the maximum training sensitivity plus specificity (MTSS) criterion—a widely adopted approach for optimizing suitability classification in MaxEnt studies. Using the “Reclassify” tool in Spatial Analyst, the prediction results (probability values: 0–1) were categorized into four suitability levels: (1) Unsuitable area (p < 0.1); (2) Low-suitability area (0.1 ≤ p < 0.3); (3) Medium-suitability area (0.3 ≤ p < 0.5); (4) High-suitable area (0.5 ≤ p ≤ 1) [33,34,35,36]. This classification yielded the potential geographical distribution of P. sibirica in China, with the generally suitable and highly suitable areas collectively defined as the “total suitable area” for quantitative analysis of distribution changes. To analyze the vertical (altitudinal) shift in P. sibirica under different climate scenarios, we extracted the elevation information of the suitable habitats in each period using ArcGIS 10.4 and calculated the average elevation of high-suitability areas.
2.5. Priority Conservation Areas
Priority conservation zones for P. sibirica were identified systematically using Zonation v4.0 software to ensure reproducibility [37]. The analysis scope was restricted to the prefecture-level city ranges where P. sibirica occurs, based on the current and future binary distribution maps mentioned above, to reduce model computational load. Input data included the current-period potential suitable habitat map of P. sibirica and spatial distribution data of China’s existing protected areas (unified to 2.5 arcmin resolution). The core-area removal rule was adopted to sequentially eliminate low-priority suitable habitat units, retaining core areas with high connectivity and suitability; the edge removal rule was further applied to remove edge regions of remaining grids, ensuring landscape continuity during the removal process. Other parameters followed default settings, and the resulting priority conservation index (0–1) was classified into five levels via the natural break method [38].
3. Results
3.1. The Accuracy of the MaxEnt Model
We integrated 87 distribution records and environmental variables into the MaxEnt model to predict the potential geographic distribution of P. sibirica. Under default model parameters (FC = LQ, RM = 1), the omission rate reached 2.6% with the minimum delta AICc value. Ten replicate runs were performed for accuracy validation, yielding training- and test-set AUC values all above 0.9 (mean training AUC = 0.962; Supplementary Figure S1). Across all replicates, omission rates and omission rate curves remained consistently low with minimal fluctuations across different threshold values. These results indicate strong correlations between the selected environmental variables and the potential distribution of P. sibirica, as well as high prediction accuracy and robustness of the MaxEnt model.
3.2. Environmental Variable Analysis
In these 18 environmental variables, Human Footprint, Precipitation Seasonality-Bio15, Annual Mean Temperature-Bio1, Elevation, and Mean temperature of coldest quarter-Bio11 contributed significantly to the MaxEnt model predictions, with a cumulative contribution of 91.4% (Table 1). The jackknife test (Supplementary Figure S2) indicated that Annual Mean Temperature-Bio1, Precipitation Seasonality-Bio15, Temperature Seasonality-Bio4 and Human Footprint had a substantial impact on the distribution of P. sibirica (AUC > 0.8). Based on the environmental factor response curves (Supplementary Figure S3), P. sibirica exhibits unimodal response curves for all six primary environmental factors; the aforementioned six factors represent the primary environmental determinants influencing the optimal distribution of P. sibirica, indicating that the modeled predictions are relatively accurate.

Table 1.
Contribution and importance of 18 environment factor variables.
3.3. Distribution of P. sibirica in Our Current Climate
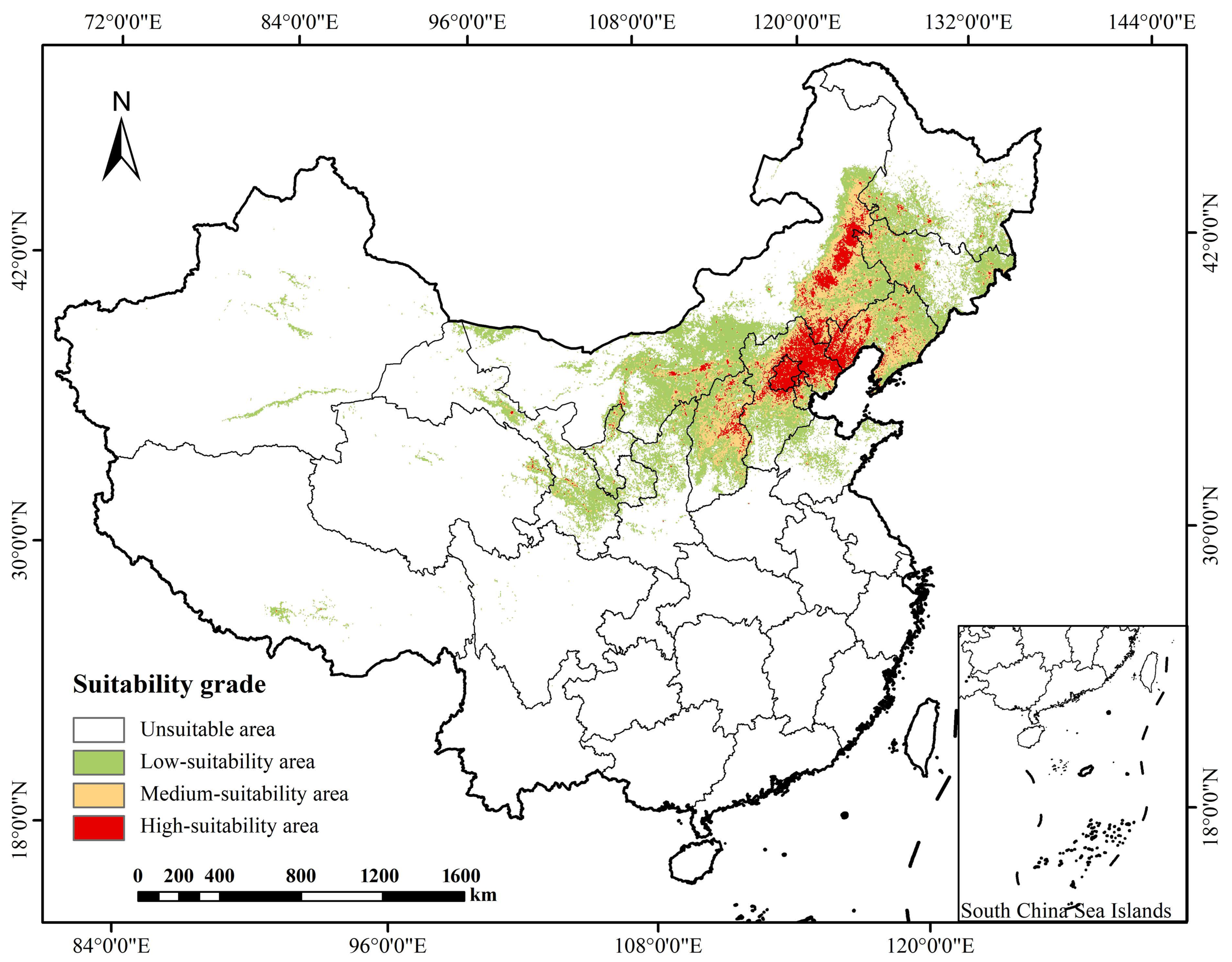
The MaxEnt model simulation revealed the spatial pattern and area characteristics of the current suitable habitats for P. sibirica (Table 2 and Figure 3). The total area of potential suitable habitats (high-suitability + medium-suitability) is 506,620.2 km2, accounting for 5.28% of China’s total land area. Among them, the high-suitability area covers 170,974.1 km2, and the medium-suitability area is 335,646.1 km2. Spatially, the suitable habitats exhibit a concentrated distribution pattern, primarily located in Northwest, North and Northeast China, and in high-suitability regions in the north and northeast. This spatial distribution is closely linked to the species physiological adaptations, reflecting the decisive role of regional hydrothermal conditions in shaping its current distribution range.

Table 2.
Potential distribution area of P. sibirica in China in different periods.
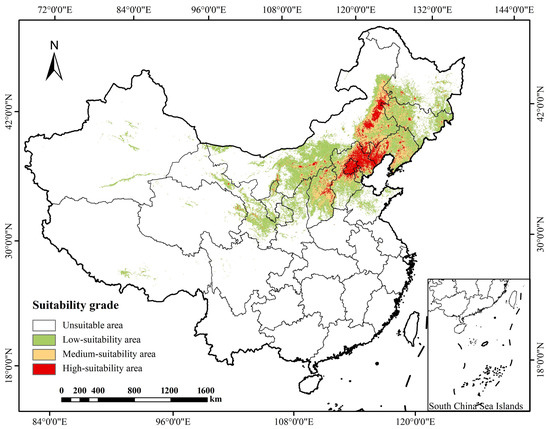
Figure 3.
The current period P. sibirica distribution map of suitable areas.
3.4. Historical Suitable Habitats and Shifting Trends of P. sibirica Under Past Climates
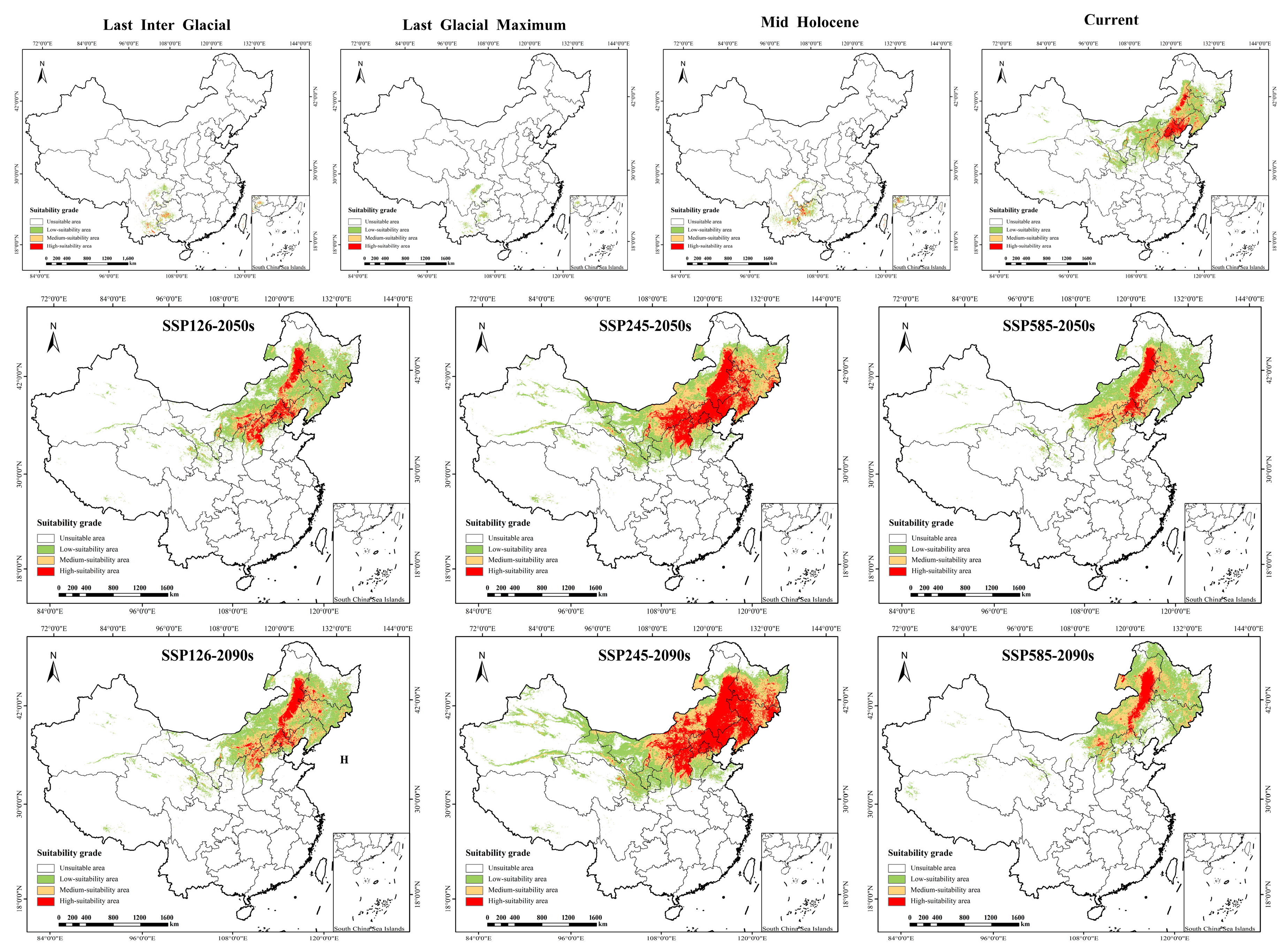
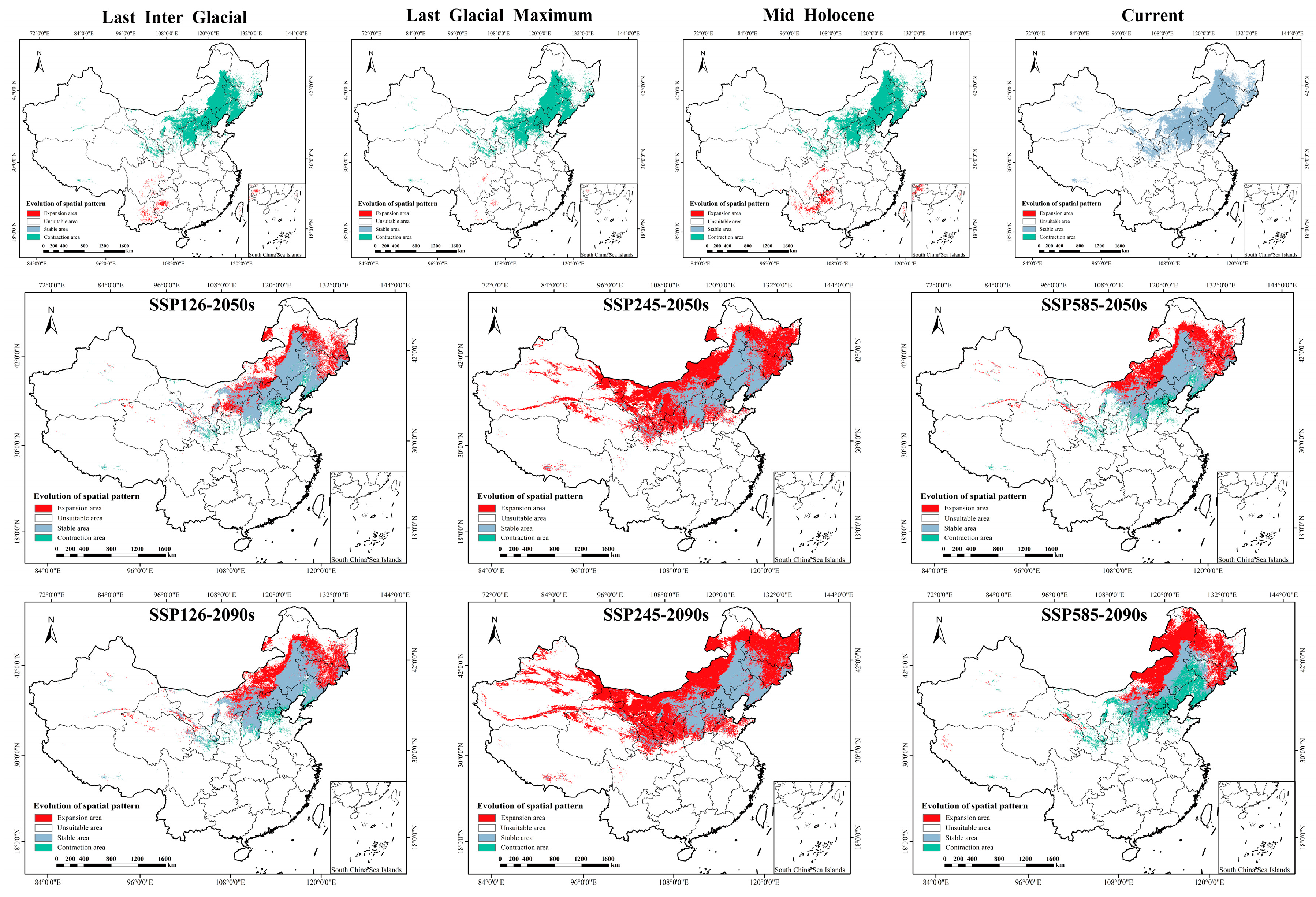
Reconstruction of historical potential distribution revealed significant spatiotemporal dynamics of P. sibirica suitable habitats across the Last Interglacial (LIG), Last Glacial Maximum (LGM), and Mid-Holocene (MH) (Table 2 and Table 3; Figure 4 and Figure 5). Temporally, the suitable area showed a “contraction followed by expansion” trend: it drastically contracted by 74.79% from 26,306.8 km2 (LIG) to 6632.8 km2 (LGM), with no stable habitat retained between the two periods and a contraction area of 940,782.1 km2; by the MH, the suitable area expanded to 50,608.0 km2, and a small stable habitat area (26.1 km2) emerged. Spatially, the distribution center shifted dramatically: from the LIG to the LGM, suitable habitats retreated from high and low latitudes to mid-high latitudes; by the MH, the core distribution migrated to Southwest China (Sichuan, Yunnan, Guizhou), where the high-suitability area (10,428.0 km2) accounted for only 6% of the current high-suitability area. These results confirm that glacial–interglacial climate fluctuations drove the extreme contraction and subsequent expansion of the species’ historical suitable habitats, with minimal habitat stability between key periods.

Table 3.
Dynamic changes in the suitable area for P. sibirica under different combination of climate scenarios/years.
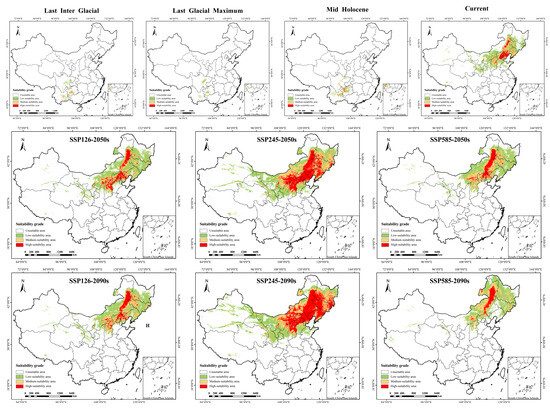
Figure 4.
Distribution of suitable areas of P. sibirica under historical, current and future climate scenarios in China. SSP: Shared socio-economic pathways.
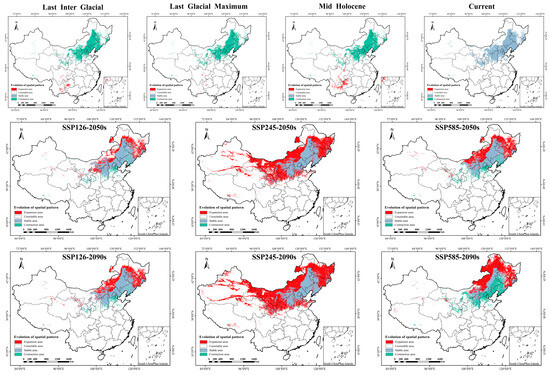
Figure 5.
The spatial pattern of P. sibirica in China under historical, current and future climate scenarios. SSP: Shared socio-economic pathways.
3.5. Future Suitable Habitats and Shifting Trends of P. sibirica Under Future Climates
Under three future shared socioeconomic pathway (SSP) scenarios (SSP126, SSP245, SSP585), the suitable habitats of P. sibirica exhibited distinct scenario-dependent expansion/contraction dynamics, area changes, and stability characteristics (Table 2 and Table 3; Figure 4 and Figure 5). In the SSP126 scenario, the suitable area continuously expanded from 63,882.4 km2 (2050s) to 695,804.1 km2 (2090s), the stable habitat area reached 821,957.5 km2 (2090s), while the contraction area was 118,805.2 km2, and the high-suitability area extended from North China to northeastern and northwestern peripheries. The SSP245 scenario showed the most prominent expansion: the total suitable area surged from 1,419,201.4 km2 (2050s) to 1,742,417.5 km2 (2090s); notably, there was no significant contraction area across both periods, and the stable habitat area remained 940,762.6 km2, with the high-suitability area expanding northwestward and northeastward with enhanced spatial continuity. In contrast, the SSP585 scenario presented a “first expansion then contraction” trend: the suitable area expanded to 681,876.8 km2 (2050s) but contracted to 617,463.4 km2 (2090s), the contraction area increased sharply to 530,501.3 km2 (2090s), while the stable habitat area decreased to 410,261.3 km2, and the high-suitability area shrank in the northeast due to extreme climate stress. Overall, the SSP245 scenario is the most suitable for habitat expansion and stability, while the late SSP585 scenario is characterized by significant habitat contraction and reduced stability, reflecting the adverse impacts of extreme climate change on species’ suitable habitats.
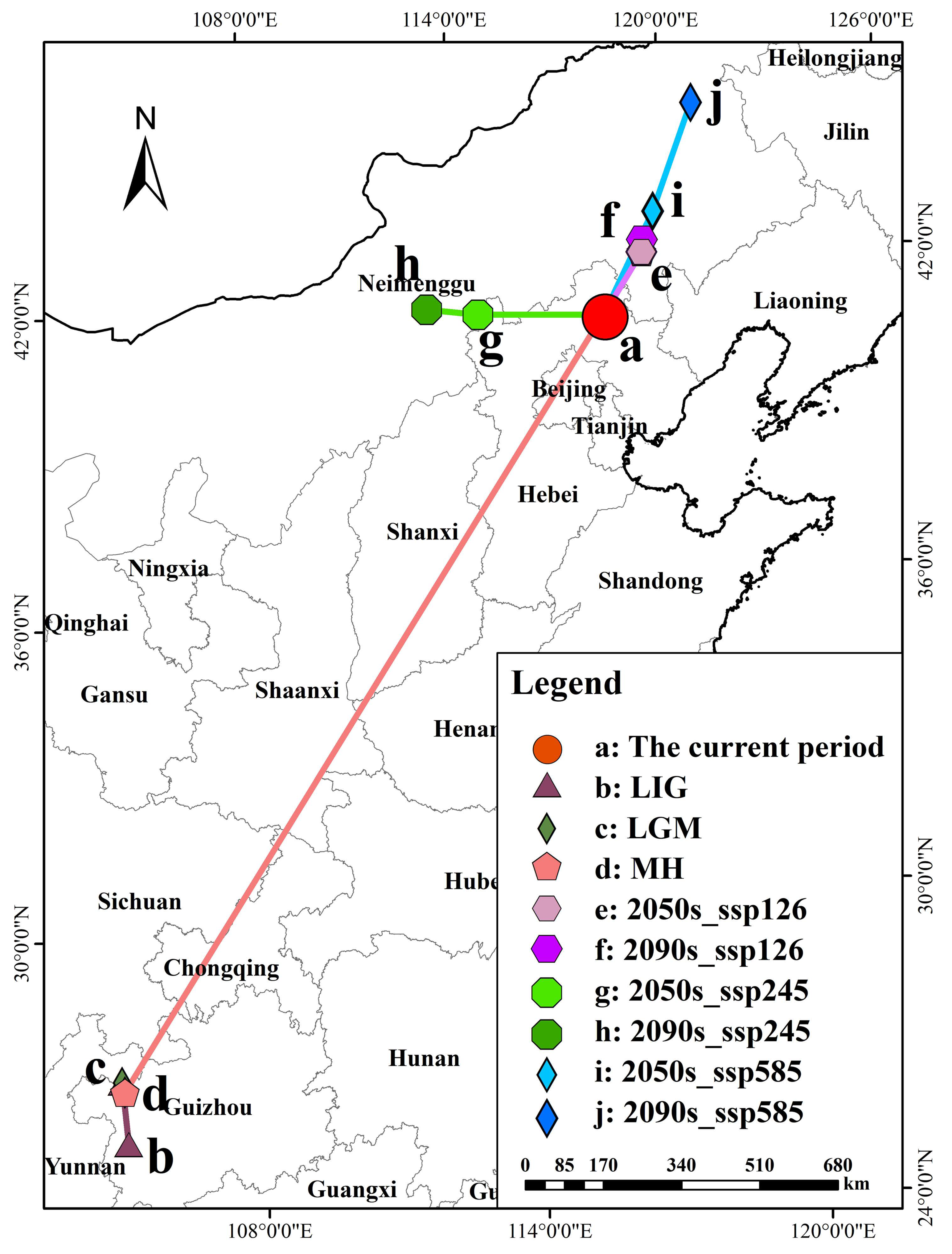
3.6. The Change in Center-of-Mass Migration of P. sibirica in Different Periods
This study used the geometric centroid method to analyze the spatial distribution of potential habitats of P. sibirica across different periods in China and also examined the altitudinal shifts in its suitable habitats (Supplementary Table S3). Results showed that the present-day centroid is located in Chengde City, Hebei Province (Figure 6), with the current average elevation of suitable habitats at 552.68 m. From the Last Interglacial (LIG, 1540.33 m) to the Last Glacial Maximum (LGM, 918.55 m), the centroid only shifted a short distance within southwestern Guizhou, while the average elevation of suitable habitats decreased. In contrast, the centroid migrated 2137.06 km northeastward from the Mid-Holocene (MH, 1607.94 m) to the present, accompanied by a notable downward shift in average elevation (Table 4). Under future climate scenarios, the optimal habitat centroid tended to move northeastward overall as greenhouse gas concentrations increased (to Chifeng and Tongliao under SSP126 and SSP585), and the average elevation showed a slight upward trend in the late high-emission scenario (SSP585 in 2090s, 823.47 m). Specifically, under SSP126 (2050s/2090s) and SSP585 (2050s/2090s) scenarios, the centroid migrated northeast to Chifeng and Tongliao, while under SSP245 (2050s/2090s), it shifted west to Ulanqab with a gradual decrease in average elevation. Historically, the optimal habitat of P. sibirica shifted from southwestern China (higher elevations) to the current North China Plain (lower elevations). Under future climate scenarios, its suitable habitats will further migrate northeastward toward higher-elevation areas.
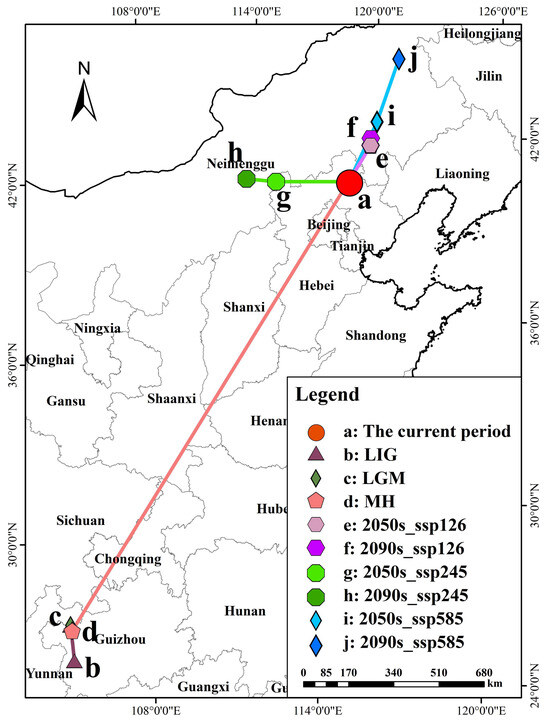
Figure 6.
The change in center-of-mass migration of P. sibirica in different periods.

Table 4.
Geometric center (centroid) migration distance in different periods.
3.7. Analysis of the P. sibirica Priority Conservation Area
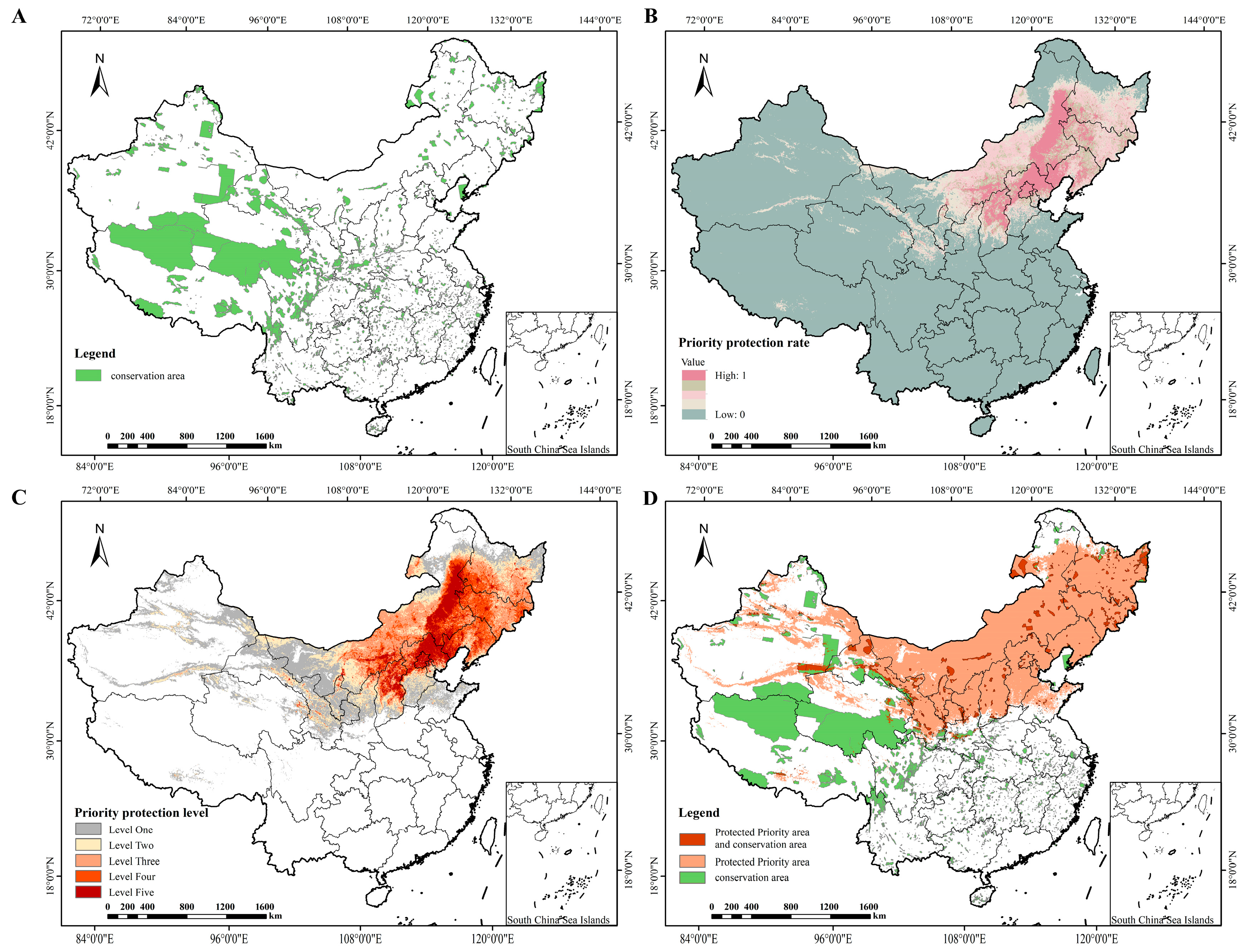
This study employed Zonation v4.0 software to systematically identify priority conservation areas, with conservation status analyzed via area statistics (Table 5) and spatial patterns (Figure 7). Existing conservation areas (Figure 7A) are fragmented and distributed in southwestern, northwestern, and partial northern China, covering 147.17 × 104 km2 (15.33% of the study area, Table 5). Priority protection levels (Figure 7C and Table 5) showed distinct regional differentiation: the first-level priority zone (135.96 × 104 km2, 14.16%) concentrated in northern China (Inner Mongolia, Hebei, Liaoning), matching the high priority protection rate in this region (Figure 7B). Overlay analysis (Figure 7D and Table 5) revealed that only 19.65 × 104 km2 of the identified priority conservation zones overlapped with existing protected areas. Notably, 320.32 × 104 km2 of the priority conservation zones fall outside the scope of current protected areas, including the core first-level priority zones in northern China that lack adequate coverage. These results highlight significant gaps in the existing fragmented protection system which fails to fully safeguard the critical high-priority habitats of P. sibirica.

Table 5.
Area statistics of conservation zones for P. sibirica.
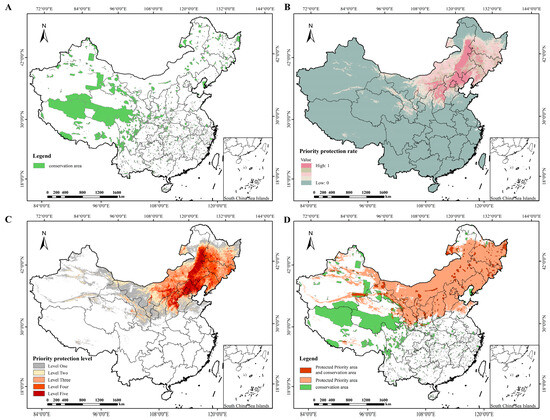
Figure 7.
Priority conservation areas for P. sibirica. in China. (A): Distribution of protected areas in China (B): Priority conservation rate for P. sibirica. (C): Priority protection level for P. sibirica (D): Overlap between priority conservation areas and protected areas for P. sibirica.
4. Discussion
4.1. Bioclimatic Predictors and Model Performance
Prunus sibirica has strong resistance to cold, drought, and barrenness and prefers to grow in arid and desertified areas [39]. Our research team found through field investigation that most of the wild seed sources of P. sibirica in China grow in Qinghai, Gansu, Inner Mongolia, Shaanxi, Shanxi, Hebei, Liaoning, Jilin, and Heilongjiang. Among the 18 environmental variables integrated into the MaxEnt model, three key factors collectively drive its distribution with a cumulative contribution of 71.8%: Human Footprint (HFP, 27.7%), Precipitation Seasonality (Bio15, 26.9%), and Annual Mean Temperature (Bio1, 17.2%). Notably, HFP showed the highest percentage contribution but relatively low permutation importance, a pattern that may stem from the distinct ecological meanings of these two metrics. Percentage contribution reflects the variable role in model training, while permutation importance indicates the impact of randomizing the variable on prediction accuracy [32]. This discrepancy likely arises because HFP, as a background disturbance factor, shapes habitat suitability over long timescales but has weaker marginal effects on fine-scale predictions once climate and topographic variables are incorporated [29,40]. This finding aligns with prior studies highlighting seasonal precipitation and cool temperate conditions as pivotal for P. sibirica [41] but extends existing knowledge by emphasizing HFP as a dominant driver, reflecting the significant impact of anthropogenic disturbances (overgrazing, agricultural expansion, infrastructure construction) on local habitat suitability [40]. Notably, this contrasts with earlier research that focused solely on climatic factors [42], underscoring the need to integrate human activities for accurate distribution modeling of semi-arid zone species. The MaxEnt model demonstrated excellent predictive performance, with a mean AUC > 0.9, consistent with Li’s [43] results for P. sibirica habitat modeling. The high congruence between predicted suitable areas and the actual distribution of P. sibirica sources further verifies the model reliability. However, potential uncertainties should be acknowledged: the WorldClim database used in this study may have limited accuracy in arid northern China due to sparse meteorological station coverage [44] which could affect fine-scale predictions of edge habitats. Additionally, the model equilibrium assumption may underestimate the impacts of rapid climate change, as P. sibirica may not migrate fast enough to track optimal hydrothermal conditions [19]. These limitations highlight avenues for future improvement, such as integrating high-resolution in situ climate data and dynamic land-use scenarios.
4.2. Changes in Potential Distribution Areas
The analysis of P. sibirica potential distribution dynamics across historical and current periods demonstrates a significant response to global climate change, particularly global warming [42,45,46,47,48]. Our results reveal a distinct “contraction followed by expansion” trend in its historical suitable habitats: the total suitable area was 26,306.8 km2 during the Last Interglacial (LIG) but contracted drastically by 74.79% to 6632.8 km2 during the Last Glacial Maximum (LGM), with no stable habitat retained and a contraction area of 940,782.1 km2. This severe habitat loss is tightly linked to the highly unstable LGM climate, characterized by alternating warm-wet, warm-dry, and cool-dry phases [16,18,49]. Inherently adapted to semi-arid and semi-humid habitats [35], P. sibirica likely lost low-latitude suitable ranges (southwestern Shaanxi, central Sichuan, southwest Guizhou, southeastern Yunnan, southeastern Tibet) under LGM extreme conditions, leading to a shift toward mid-high latitudes. By the Mid-Holocene, with climate stabilization, the suitable area expanded to 50,608.0 km2, with the core distribution shifting to Southwest China (Sichuan, Yunnan, Guizhou), laying the foundation for its subsequent northward migration to the current northern China core. This historical dynamic fully reflects the high sensitivity of P. sibirica’s habitat to climate fluctuations, with climate change acting as a key driver of its range contraction, expansion, and spatial migration.
Compared with the Mid-Holocene, the suitable habitat and distribution centroid of P. sibirica have undergone dramatic changes, driven primarily by post-MH climate drying and the species’ ecological adaptability. Our results show the total suitable area expanded nearly 10-fold from 50,608.0 km2 (MH) to 506,620.2 km2 at present, with the core distribution shifting sharply from Southwest to northern China arid and semi-arid regions. This shift aligns with East Asia post-MH climate evolution. Northern China gradually formed a temperate continental climate characterized by low precipitation and large seasonal temperature differences [50], which perfectly matches P. sibirica’s inherent cold and drought tolerance. Notably, the distribution centroid migrated 2137.06 km northeastward from Bijie in the MH to Chengde. This displacement is linked to key environmental drivers: post-MH monsoon weakening led to increased humidification in parts of Southwest China [51], reducing habitat suitability, while northern China’s precipitation showed a long-term decreasing trend, forming a stable semi-arid environment [52]. Phylogeographic evidence confirms P. sibirica expanded northward from glacial refugia post MH, with climatic factors shaping its eastern and western genetic groups [39]. These changes reflect P. sibirica’s high sensitivity to long-term climate change, as its ecological adaptability enabled tracking suitable habitats northward, consistent with the general response of temperate arid zone plants to post-Holocene climate shifts.
Under three future CMIP6 shared socioeconomic pathway (SSP) scenarios, the potential distribution of P. sibirica will exhibit distinct scenario-dependent dynamics, driven by continued climate warming and associated changes in key environmental factors [34,40]. Our results show that SSP245 is the most suitable for habitat expansion: the total suitable area will surge from 1,419,201.4 km2 (2050s) to 1,742,417.5 km2 (2090s) with no significant contraction, and the high-suitability area will expand northwestward and northeastward. This aligns with the species drought and cold tolerance [53], as SSP245 moderate warming maintains stable semi-arid conditions in northern China, which is core to P. sibirica’s ecological niche. In contrast, SSP126 will see gradual expansion (63,882.4 km2 to 695,804.1 km2) with limited contraction in peripheral regions, while SSP585 presents a “first expansion then contraction” trend: the total suitable area expands to 681,876.8 km2 (2050s) but shrinks to 617,463.4 km2 (2090s), with northeast high-suitability areas contracting due to extreme climate stress [54]. These dynamics are closely linked to key drivers including precipitation seasonality (Bio15) and annual mean temperature (Bio1), which collectively contribute 44.1% to distribution predictions [55]. Under SSP585, intensified precipitation fluctuations and excessive warming may exceed the species thermal and moisture niche thresholds, leading to habitat degradation [56]. Additionally, centroid migration reflects scenario-specific shifts: northeastward movement under SSP126 and SSP585 and westward shift under SSP245, consistent with temperate arid species tracking optimal hydrothermal conditions. These findings highlight that P. sibirica’s future range is tightly tied to emission trajectories. Conservation strategies should prioritize protecting stable core habitats under SSP245, mitigating contraction risks in SSP585 northeast regions, and facilitating natural migration to emerging suitable areas. The vertical migration trend of P. sibirica is consistent with the results of studies on other temperate tree species, such as Larix gmelinii (Rupr.) Kuzen. and Betula platyphylla Suk. [16,24]. Larix gmelinii migrated upward in the Greater Khingan Mountains under climate warming, which is similar to the altitudinal migration trend of P. sibirica (Supplementary Table S3). This indicates that altitudinal migration is a universal adaptive strategy for temperate trees to cope with climate change and should be considered in the design of protected areas.
4.3. Priority Protected Areas of P. sibirica
Based on Zonation v4.0 identification of P. sibirica, priority conservation areas reveals critical mismatches: first-level zones overlap with only 19.65 × 104 km2 of existing fragmented, southwest/northwest-focused protections, leaving 320.32 × 104 km2 of key northern habitats incompletely protected. This gap stems from historical biases toward high-biodiversity regions over species-specific core habitats [20], and given northern China’s stable semi-arid conditions [57], optimizing the conservation network is urgent. To enhance practical application, we propose targeted strategies: (1) expand northern protected areas by establishing nature reserves/ecological corridors in Level 1 zones (Inner Mongolia, Hebei, Liaoning) to connect fragmented habitats; (2) implement adaptive management (artificial afforestation, soil moisture conservation) in SSP585 high-risk areas (northeast China) to boost population resilience; (3) balance conservation and industrial development by promoting sustainable cultivation in medium-priority zones, using almond industry income to support protection. Consistent with China’s national ecological policies (Three-North Shelterbelt Program), these measures will safeguard germplasm resources and arid/semi-arid ecosystem functions.
5. Conclusions
This study clarifies the bioclimatic adaptation and spatiotemporal distribution dynamics of P. sibirica. under climate change, using MaxEnt and Zonation v4.0 to identify Human Footprint, precipitation seasonality (Bio15), and annual mean temperature (Bio1) as core distribution drivers. Historical dynamics show the species underwent range contraction–expansion, with a 2137.06 km northeastward centroid shift to northern China’s arid/semi-arid regions. Future SSP scenarios indicate SSP245 favors habitat stability, while SSP585 brings contraction risks; vertical migration analysis also reveals a trend toward higher elevations (informing protected area design). Critically, northern China priority conservation zones overlap with only 2.05% of existing protections, highlighting urgent gaps. We propose targeted strategies (expanding northern protected areas, adaptive management in high-risk zones, balancing conservation–industry) to support core habitat protection, protected area optimization, and adaptive germplasm management for P. sibirica under climate change.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/f17020266/s1.
Author Contributions
J.C.: Editing, writing—original draft, resources, methodology, investigation. L.W.: Methodology, writing—review. D.A.: Methodology, conceptualization. M.M.: Methodology. R.Y.: Validation, investigation. S.Z.: Resources, methodology. W.B.: Supervision, project administration, funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This study was jointly supported by the National Natural Science Foundation of China (32360400) and the Outstanding Youth Fund for Natural Science of Inner Mongolia Autonomous Region (2024JQ13).
Institutional Review Board Statement
Not applicable. This manuscript does not include human or animal research. If this manuscript involves research on animals or humans, it is imperative to disclose all approval details.
Data Availability Statement
The data and materials underlying this article are available in the article and in its Supplementary Materials.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Zhang, H.; Zhao, H.X.; Xu, C.G. The potential geographical distribution of Alsophila spinulosain under climate change in China. Chin. J. Ecol. 2021, 40, 968–979. [Google Scholar] [CrossRef]
- Taonda, A.; Zerbo, I.; Da, S.S.; Traoré, I.C.E.; N’Guessan, A.E.; Kassi, J.N.; Thiombiano, A. Potential distribution of Detarium microcarpum under different climate change scenarios in Burkina Faso. J. Nat. Conserv. 2024, 82, 126737. [Google Scholar] [CrossRef]
- Ding, J.J.; Zhuo, Y.Y.; Xu, W.X.; Kessler, M.; Wang, M.Y.; Yang, W.K. Synergistic effects of climate and land use change on khulan (Equus hemionus hemionus) habitat in China. Glob. Ecol. Conserv. 2024, 54, e03181. [Google Scholar] [CrossRef]
- Xu, Z.L.; Peng, H.H.; Peng, S.Z. The development and evaluation of species distribution models. Acta Ecol. Sin. 2015, 35, 557–567. [Google Scholar] [CrossRef]
- Lu, Z.R.; Shan, Y.X.; Shan, H.J.; Wen, H.C.; Wu, Y.N.; Han, R.C.; Tong, X.H. Effect of climate change on Clinopodium polycephalum (Vaniot) C. Y. Wu & S. J. Hsuan distribution adopting temporal data, ArcGIS, and the MaxEnt model. Front. Plant Sci. 2024, 15, 1445764. [Google Scholar] [CrossRef]
- Huan, Z.Q.; Geng, X.M.; Xu, X.R.; Liu, W.; Zhu, Z.L.; Tang, M. Potential Geographical Distribution of Michelia martinii under Different Climate Change Scenarios Based on MaxEnt Model. J. Ecol. Rural Environ. 2023, 39, 1277–1287. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Z.Z.; Meng, S.; Jiang, Q.; Hu, G.M.; Zhang, L.; Yao, X.H. Potential distribution under climate change and ecological niche differences between Actinidia chinensis complex. Sci. Hortic. 2024, 337, 113533. [Google Scholar] [CrossRef]
- Wang, Z.H.; Chang, N.; Li, H.Y.; Wei, X.H.; Shi, Y.; Li, K.; Li, J.Y.; Guo, C.R.; Liu, Q.Y. Impacts of climate change on the potential distribution of Pulex simulans and Polygenis gwyni. Ecol. Evol. 2024, 14, e11621. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Liu, H.L.; Cui, X.J.; Peng, J.J.; Wang, X.; Shen, L.; Zhang, M.Q.; Chen, L.X.; Li, X. Predicting the Population Size and Potential Habitat Distribution of Moschus berezovskii in Chongqing Based on the MaxEnt Model. Forests 2024, 5, 1449. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, P.; Zhang, C.; Li, B.; Kang, Y.; Yang, J.; Ren, T.; Chen, J.; Dong, S. Growth and development characteristics of fruit and vegetative bud outgrowth of Prunus sibirica L. in relation to physiological fruit drop. BMC Plant Biol. 2025, 25, 958. [Google Scholar] [CrossRef]
- Liu, Q.; Li, W.; Zhao, X.; Zhang, H.; Chen, J.; Liu, Q.; Li, T.; Dong, S. Transcriptomic Profiling Analyses Revealed Candidate Genes Under Freezing Stress in Siberian Apricot (Prunus sibirica). Forests 2024, 15, 1870. [Google Scholar] [CrossRef]
- Bernoussi, S.E.; Boujemaa, I.; Guezzane, C.E.; Bou-Ouzoukni, Y.; Nounah, I.; Bouyahya, A.; Ullah, R.; Iqbal, Z.; Maggi, F.; Caprioli, G.; et al. Comparative analysis of nutritional value and antioxidant activity in sweet and bitter almonds. LWT-Food Sci. Technol. 2024, 206, 116587. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, S.; Zhao, X.; Dong, S.; Chen, J.; Sun, Y.; Sun, Q.; Liu, Q. Genome-wide identification and comprehensive analysis of the AP2/ERF gene family in Prunus sibirica under low-temperature stress. BMC Plant Biol. 2024, 24, 883. [Google Scholar] [CrossRef]
- Liu, R.L. Physiological and Biochemical Characteristics of Delayed Flowering in Armeniaca sibirica. Master’s Thesis, Shenyang Agricultural University, Shenyang, China, 2023. [Google Scholar] [CrossRef]
- Liu, S.; Veronique, D.; Zhang, Y.J.; Liu, N.; Ma, X.X.; Zhang, Q.P.; Xu, M.; Zhang, Y.P.; Liu, W.S. Study on the Relationship between Genetic Diversity and Geographical Distribution of Wild Common Apricots and Siberian apricots. J. Plant Genet. Resour. 2020, 21, 1527–1538. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, J.; Zhang, C.; Feng, Y.; Jin, L.; Wei, S.; Yang, H.; Cao, Z.; Peng, J.; Luo, Y. Prediction of the Future Evolution Trends of Prunus sibirica in China Based on the Key Climate Factors Using MaxEnt Modeling. Biology 2024, 13, 973. [Google Scholar] [CrossRef]
- Wang, T.; Tian, Z.P.; Jiang, D.B. Changes in the Asian ITCZ During the Last Interglacial, the Last Glacial Maximum, and the Mid-Holocene. J. Geophys. Res. Atmos. 2024, 129, 16. [Google Scholar] [CrossRef]
- Ge, Y.G.; Wei, M.J. Palaeo-vegetation and Palaeo-environment Change of Last Inter-Glacial Period in Xishan Mountain of Beijing. Geol. Sci. Technol. Inf. 2008, 27, 5–12. Available online: http://ir.imde.ac.cn/handle/131551/279 (accessed on 8 October 2024).
- Araújo, M.B.; Guisan, A. Five (or so) challenges for species distribution modelling. J. Biogeogr. 2006, 33, 1677–1688. [Google Scholar] [CrossRef]
- Thuiller, W.; Lavorel, S.; Araújo, M.B.; Sykes, M.T.; Prentice, I.C. Climate change threats to plant diversity in Europe. Proc. Natl. Acad. Sci. USA 2005, 102, 8245–8250. [Google Scholar] [CrossRef]
- Zhang, S.N.; Chen, J.X.; Ao, D.; Hong, M.; Zhang, Y.Q.; Bao, F.H.; Wang, L.; Wu, Y.T.N.; Bai, Y.E.; Bao, W.Q. Prediction of Potential Suitable Areas of Amygdalus pedunculata in China under Climate Change. Sci. Silvae Sin. 2023, 59, 25–36. Available online: http://www.linyekexue.net/CN/Y2023/V59/I12/25 (accessed on 8 October 2024).
- Zhang, T.; Huang, Z.H.; Peng, Y.J.; Wang, Y.T.; Wang, P.; Wang, S.T.; Cui, G.F. Prediction of potential suitable areas of Actinidia arguta in China based on Maxent model. Acta Ecol. Sin. 2020, 40, 4921–4928. [Google Scholar]
- Wang, P.; Luo, W.X.; Zhang, Q.Y.; Han, S.X.; Jin, Z.; Liu, J.C.; Li, Z.F.; Tao, J.P. Assessing the impact of climate change on three Populus species in China: Distribution patterns and implications. Glob. Ecol. Conserv. 2024, 50, e02853. [Google Scholar] [CrossRef]
- Zhao, X.H.; Hao, Y.S.; Wang, T.; Dong, L.H.; Li, F.R. Competition is critical to the growth of Larix gmelinii and Betula platyphylla in secondary forests in Northeast China under climate change. Glob. Ecol. Conserv. 2024, 51, e02935. [Google Scholar] [CrossRef]
- Gütschow, J.; Jeffery, M.L.; Günther, A.; Meinshausen, M. Country-resolved combined emission and socio-economic pathways based on the representative concentration pathway (RCP) and shared socio-economic pathway (SSP) scenarios. Earth Syst. Sci 2021, 13, 1005–1040. [Google Scholar] [CrossRef]
- OuYang, L.; Chen, S.; Liu, X.; He, S.; Zhang, W. Suitable geographic Range for Eucalyptus camaldulensis in China and its Response to climate change. Sci. Silvae Sin. 2019, 55, 1–11. [Google Scholar] [CrossRef]
- Gao, M.; Zhao, G.; Zhang, S.; Wang, Z.; Wen, X.; Liu, L.; Zhang, C.; Tie, N.; Sa, R. Priority Conservation Area of Larix gmelinii under Climate Change: Application of an Ensemble Modeling. Front. Plant Sci. 2023, 14, 1177307. [Google Scholar] [CrossRef]
- Elith, J.H.; Graham, C.P.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel Methods Improve Prediction of Species’ Distributions from Occurrence Data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carré, G.; Marquéz, J.R.G.; Gruber, B.; Lafourcade, B.; Leitão, P.J.; et al. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Jueterbock, A.; Smolina, I.; Coyer, J.A.; Hoarau, G. The fate of the arctic seaweed Fucus distichus under climate change: An ecological niche modeling approach. Ecol. Evol. 2016, 6, 1712–1724. [Google Scholar] [CrossRef]
- Muscarella, R.; Galante, P.J.; Soley-Guardia, M.; Boria, R.A.; Kass, J.M.; Uriarte, M.; Anderson, R.P. ENMeval: An R package for conducting spatially independent evaluations and estimating optimal model complexity for maxent ecological niche models. Methods Ecol. Evol. 2014, 5, 1198–1205. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Dudík, M.; Schapire, R.E.; Blair, M.E. Opening the black box: An open-source release of maxent. Ecography 2017, 40, 887–893. [Google Scholar] [CrossRef]
- Zhang, T.; Lv, X.Y.; Zhao, Q.; Zhang, C.J.; Yin, H.L.; Deng, S.Y.; Yan, G.; Wang, G.Z.; Cao, X.Y.; Ou, H.; et al. Assessment of suitable region of Asparagus cochinchinensis (Lour.) Merr. under different climatic conditions in China by the MaxEnt model and HPLC analysis. Ecol. Evol. 2024, 14, e70354. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.T.; Huang, Y.; Su, M.M.; Liu, M.; Yang, J.X.; Wu, Q.G. Spatial Distribution Patterns of the Key Afforestation Species Cupressus funebris: Insights from an Ensemble Model under Climate Change Scenarios. Forests 2024, 15, 1280. [Google Scholar] [CrossRef]
- Guo, Y.X.; Zhang, S.Y.; Ren, L.H.; Tian, X.; Tang, S.C.; Xian, Y.S.; Wu, X.J.; Zhang, Z.L. Prediction of Chinese suitable habitats of Panax notoginseng under climate change based on MaxEnt and chemometric methods. Sci. Rep. 2024, 14, 16434. [Google Scholar] [CrossRef]
- Duan, J.P.; Liu, J.; Huang, Z.H. Predicting the distribution pattern changes of dye plant habitats caused by climate change. Front. Plant Sci. 2024, 15, 1364481. [Google Scholar] [CrossRef]
- Lehtomki, J.; Moilanen, A. Methods and workflow for spatial conservation prioritization using Zonation. Environ. Model. Softw. 2013, 47, 128–137. [Google Scholar] [CrossRef]
- Jenks, G.F. The Data Model Concept in Statistical Mapping. In International Yearbook of Cartography; C. Vertelsmanns Verlag: Gütersloh, Germany, 1967; Volume 7, pp. 186–190. Available online: https://cir.nii.ac.jp/crid/1573668925394541312 (accessed on 11 December 2025).
- Wang, P.K. Study on fruit growth and physiological fruit drop characteristics of Armeniaca sibirica. Master’s Thesis, Shenyang Agricultural University, Shenyang, China, 2023. [Google Scholar]
- Li, K.; Wu, W.; Tian, S.; Li, Z.; Wu, Y.; Cao, Y.; Li, L.; Xiao, W. Divergent spatiotemporal interactions between human footprint and ecosystem services: Multi-scenario analysis of protected areas and edge zones. Environ. Impact Assess. Rev. 2025, 18, 108312. [Google Scholar] [CrossRef]
- Wang, Z.; Zeng, Y.; Zhang, Z.; Sheng, S.; Tian, J.; Wu, R.; Pang, X. Phylogeography Study of the Siberian Apricot (Prunus sibirica L.) in Northern China Assessed by Chloroplast Microsatellite and DNA Makers. Front. Plant Sci. 2017, 8, 1989. [Google Scholar] [CrossRef]
- Lu, Q.; Li, J.; Liu, G.; Zhang, Y.R.; Li, G.Q. Multi-component assessment of the impact of climate change on species vulnerability: A case study of Armeniaca sibirica in China. Acta Ecol. Sin. 2023, 43, 3181–3191. [Google Scholar] [CrossRef]
- Li, J.W.; Guo, H.; Wang, Y.S.; Xin, Z.M.; Lü, Y.J. Identification of Potential Distribution Area for Populus euphratica by the MaxEnt Ecologic Niche Model. Sci. Silvae Sin. 2019, 55, 133–139. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Liu, J.Q.; Wei, G.K.; Shi, C.Q.; Zhao, T.N.; Qian, Y.K. Suitable distribution area of drought-resistant afforestation tree species in north China based on MaxEnt model. J. Beijing For. Univ. 2022, 44, 63–77. [Google Scholar] [CrossRef]
- Zhang, J.C.; Dang, Y.W.; Chen, Y.H. Ecological Suitability Regionalization of Armeniaca sibirica in Beiliuhe Region of Fuping County Based on GIS. J. Southwest For. Univ. 2021, 41, 98–104. [Google Scholar]
- Jiang, L.; Wei, T.X.; Li, Y.R.; Wei, A.Q. Effects of topographical factors on tree species distribution of shelter forest in Loess hilly region of northern Shaanxi. Arid Land Geogr. 2021, 44, 1763–1771. [Google Scholar] [CrossRef]
- Xu, H.; Liu, M.G.; Dong, S.J.; Wu, Y.L.; Zhang, H.K. Diversity and geographical variations of germplasm resources of Armeniaca mandshurica. Chin. J. Plant Ecol. 2019, 43, 585–600. [Google Scholar] [CrossRef]
- Li, M.Y.; Zhang, S.R.; Xu, Q.H.; Xiao, J.L.; Wen, R.L. Spatial patterns of vegetation and climate in the North China Plain during the Last Glacial Maximum and Holocene climatic optimum. Sci. China Earth Sci. 2018, 61, 1279–1287. [Google Scholar] [CrossRef]
- Li, X.; Liu, X.; Pan, Z.; Xie, X.; Shi, Z.; Wang, Z.; Bai, A. Orbital-scale dynamic vegetation feedback caused the Holocene precipitation decline in northern China. Commun. Earth Environ. 2022, 3, 257. [Google Scholar] [CrossRef]
- Yue, Y.; Yang, S.; He, L.; Zheng, Z.; Zhang, X.; Chen, C.; Wan, Q.; Tang, Y.; Huang, K.; Gu, F. Vegetation ecosystem responses to climate-human interactions since the Late Holocene from peat record of Niangniang Mountain, western Guizhou. Glob. Planet. Change 2026, 257, 105238. [Google Scholar] [CrossRef]
- Yang, Z.; Gao, G.; Jie, D.; Wang, J.; Niu, H.; Liu, Z.; Meng, M.; Song, L.; Chen, N.; Wei, Y.; et al. Holocene vegetation dynamics of Horqin Sandy Land in northern China inferred from the phytolith record of a sand-paleosol section. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2023, 622, 111571. [Google Scholar] [CrossRef]
- Liu, R.; Chen, J.; Zhang, Y.; Wang, P.; Kang, Y.; Li, B.; Dong, S. Physiological and Biochemical Characteristics of Prunus sibirica during Flowering. Sci. Hortic. 2023, 321, 112358. [Google Scholar] [CrossRef]
- McDonald, R.I.; Mansur, A.V.; Ascensão, F.; Colbert, M.; Crossman, K.; Elmqvist, T.; Gonzalez, A.; Güneralp, B.; Haase, D.; Hamann, M.; et al. Research gaps in knowledge of the impact of urban growth on biodiversity. Nat. Sustain. 2020, 3, 16–24. [Google Scholar] [CrossRef]
- Bai, X.; Zhao, W.; Wang, J.; Ferreira, C.S.S. Precipitation drives the floristic composition and diversity of temperate grasslands in China. Glob. Ecol. Conserv. 2021, 32, e01933. [Google Scholar] [CrossRef]
- Chen, P.; Shen, C.; Tao, Z.; Qin, W.; Huang, W.; Siemann, E. Deterministic responses of biodiversity to climate change through exotic species invasions. Nat. Plants 2024, 10, 1464–1472. [Google Scholar] [CrossRef]
- Wang, Z.; Kang, M.; Liu, H.; Gao, J.; Zhang, Z.; Li, Y.; Wu, R.; Pang, X. High-Level Genetic Diversity and Complex Population Structure of Siberian Apricot (Prunus sibirica L.) in China as Revealed by Nuclear SSR Markers. PLoS ONE 2014, 9, e87381. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.