Study of Reaction Parameters for the Precise Synthesis of Low-Molecular-Weight Oligosiloxanes
Abstract
1. Introduction
2. Materials and Methods
2.1. Equipment
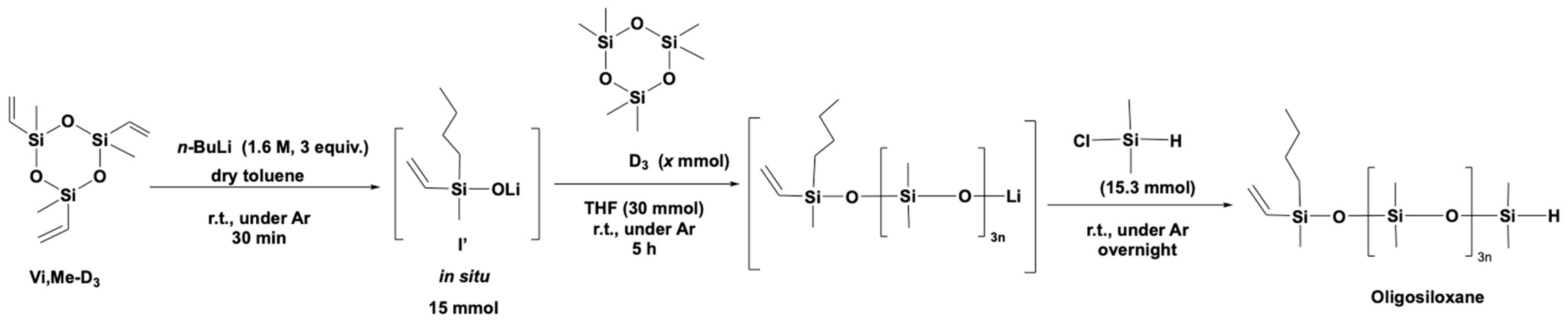
2.2. General Consideration of Synthesis
2.3. Optimization of Reaction Time for Initiator Formation

2.4. Typical Procedure for the Synthesis of Oligosiloxane (e.g., Table 3, Entry 3)

3. Results and Discussion
3.1. Optimization of Reaction Time for Initiator Formation
3.2. Investigation of Reaction Conditions for Propagation of Ring-Opening Polymerization
3.3. Variation in Molecular Ratio of Cyclotrisiloxane
3.4. Study of the Solvent Composition for Ring-Opening Polymerization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jones, G.R.; Ando, W.; Chojnowski, J. Silicon-Containing Polymers; Kluwer: Dordrecht, The Netherlands, 2000. [Google Scholar]
- Brook, A.M. Silicon in Organic, Organometallic, and Polymer Chemistry; Wiley-Inter Science: New York, NY, USA, 2000. [Google Scholar]
- Pienkowska, M.L. Review of Current Pharmaceutical Applications of Polysiloxanes (Silicones). In Handbook of Polymers for Pharmaceutical Technologies; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; Volume 2, pp. 363–382. [Google Scholar]
- Chen, G.; Feng, J.; Qiu, W.; Zhao, Y. Eugenol-modified polysiloxanes as effective anticorrosion additives for epoxy resin coatings. RSC Adv. 2017, 7, 55967. [Google Scholar] [CrossRef]
- Stadtmüller, S. Siloxanes as Additives for Plastics. Polym. Polym. Compos. 2002, 10, 49–62. [Google Scholar] [CrossRef]
- Saotome, S.; Kuang, J.; Akashi, R.; Takahashi, M.; Liu, Y.; Iijima, T.; Unno, M. Functionalization of Oligosiloxane for Polyester Comonomer. Molecules 2025, 13, 2775. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Osby, J. Siloxane modification of polycarbonate for superior flow and impact toughness. Polymer 2010, 9, 1990–1999. [Google Scholar] [CrossRef]
- Grushevenkoa, A.E.; Borisova, L.I.; Volkov, V.A. High-Selectivity Polysiloxane Membranes for Gases and Liquids Separation (A Review). Pet. Chem. 2021, 61, 959–976. [Google Scholar] [CrossRef]
- Karol Zalewski, K.; Chyłek, Z.; Trzci´nski, A.W. A Review of Polysiloxanes in Terms of Their Application in Explosives. Polymers 2021, 13, 1080. [Google Scholar] [CrossRef]
- Kumar, V.; Alam, N.M.; Park, S.S. Review of Recent Progress on Silicone Rubber Composites for Multifunctional Sensor Systems. Polymers 2024, 16, 1841. [Google Scholar] [CrossRef]
- Brinker, J.C.; Scherer, W.G. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing; Academic Press: New York, NY, USA, 1990. [Google Scholar]
- Abe, Y.; Gunji, T. Oligo- and Polysiloxanes. Prog. Polym. Sci. 2004, 29, 149–182. [Google Scholar] [CrossRef]
- Bezlepkina, A.K.; Milenin, A.S.; Vasilenko, G.N.; Muzafarov, M.A. Ring-Opening Polymerization (ROP) and Catalytic Rearrangement as a Way to Obtain Siloxane Mono- and Telechelics, as Well as Well-Organized Branching Centers: History and Prospects. Polymers 2022, 14, 2408. [Google Scholar] [CrossRef]
- Goff, J.; Sulaiman, S.; Arkles, B.; Lewicki, P.J. Soft Materials with Recoverable Shape Factors from Extreme Distortion States. Adv. Mater. 2016, 28, 2393–2398. [Google Scholar] [CrossRef]
- Fuchise, K.; Sato, K.; Igarashi, M. Precise Synthesis of Side-Chain-Functionalized Linear Polysiloxanes by Organocatalytic Ring-Opening Polymerization of Monofunctional Cyclotrisiloxanes. Macromolecules 2021, 54, 5204–5217. [Google Scholar] [CrossRef]
- Shi, J.; Liu, Z.; Zhao, N.; Liu, S.; Li, Z. Controlled Ring-Opening Polymerization of Hexamethylcyclotrisiloxane Catalyzed by Trisphosphazene Organobase to Well-defined Poly(dimethylsiloxane)s. Macromolecules 2022, 55, 2844–2853. [Google Scholar] [CrossRef]
- Sato, K.; Henmi, Y.; Kato, A.; Matsui, H.; Fuchise, K.; Higashihara, T. Precise synthesis of a,w-chain-end-functionalized poly(dimethylsiloxane) with bromoaryl groups for incorporation in naphthalene-diimide-based N-type semiconducting polymers. Polymer 2022, 252, 124934. [Google Scholar] [CrossRef]
- Okamoto, H.; Sogabe, A.; Honda, S. Synergetic binary organocatalyzed ring opening polymerization for the precision synthesis of polysiloxanes. Commun. Chem. 2024, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Corfa, A.; Caillol, S.; Pinaud, J.; Ladmiral, V. TBD-catalyzed anionic ring-opening polymerization of hexamethylcyclotrisiloxane: A new route for the controlled synthesis of PDMS. Polym. Chem. 2025, 16, 577–588. [Google Scholar] [CrossRef]
- Longin, Y.P.; Verdier, C.; Piau, M. Dynamic shear rheology of high molecular weight polydimethylsiloxanes: Comparison of rheometry and ultrasound. J. Non-Newton. Fluid Mech. 1998, 76, 213–232. [Google Scholar] [CrossRef]
- Riehle, N.; Thude, S.; Götz, T.; Kandelbauer, A.; Thanos, S.; Tovar, E.M.G.; Lorenz, G. Influence of PDMS molecular weight on transparency and mechanical properties of soft polysiloxane-urea-elastomers for intraocular lens application. Euro. Polym. J. 2018, 101, 190–201. [Google Scholar] [CrossRef]
- Matsumoto, K.; Oba, Y.; Nakajima, Y.; Shimada, S.; Sato, K. One-Pot Sequence-Controlled Synthesis of Oligosiloxanes. Angew. Chem. Int. Ed. 2018, 57, 4637–4641. [Google Scholar] [CrossRef]
- Matsumoto, K.; Shimada, S.; Sato, K. Sequence-Controlled Catalytic One-Pot Synthesis of Siloxane Oligomers. Chem. Eur. J. 2019, 25, 920–928. [Google Scholar] [CrossRef]
- Kawatsu, T.; Choi, J.-C.; Sato, K.; Matsumoto, K. Facile Synthesis of Sequence-Defined Oligo(Dimethylsiloxane-co-Diphenylsiloxane)s. Macromol. Rapid Commun. 2021, 42, 2000593. [Google Scholar] [CrossRef]
- Matsumoto, K. Precise Synthesis of Discrete Sequence-defined Oligosiloxanes. J. Jpn. Pet. Inst. 2021, 64, 307–316. [Google Scholar] [CrossRef]
- Kawatsu, T.; Minamikawa, H.; Sato, K.; Matsumoto, K. Building-block approach to discrete and sequence-specific oligosiloxanes. Polym. Chem. 2024, 15, 2740–2746. [Google Scholar] [CrossRef]
- Fuchise, K.; Kobayashi, T.; Sato, K.; Igarashi, M. Organocatalytic ring-opening polymerization of cyclotrisiloxanes using silanols as initiators for the precise synthesis of asymmetric linear polysiloxanes. Polym. Chem. 2020, 11, 7625–7636. [Google Scholar] [CrossRef]
- Ottou, N.W.; Mondière, B.A.; Parisot, H.; Blanc, D.; Portinha, D.; Fleury, E. Imidazolium triflimide-based Brønsted acidic ionic liquid as organocatalyst to trigger the cationic ring-opening polymerization of cyclotrisiloxanes. Polym. Chem. 2023, 14, 4693–4703. [Google Scholar]
- Fuchise, K.; Igarashi, M.; Sato, K.; Shimada, S. Organocatalytic controlled/living ring-opening polymerization of cyclotrisiloxanes initiated by water with strong organic base catalysts. Chem. Sci. 2018, 9, 2879–2891. [Google Scholar] [CrossRef]
- Zhang, W.; Li, S.; Liu, S.; Wang, T.-T.; Luo, Z.-H.; Bian, C.; Zhou, Y.-N. Photomediated Cationic Ring-Opening Polymerization of Cyclosiloxanes with Temporal Control. JACS Au 2024, 11, 4317–4327. [Google Scholar] [CrossRef]
- Goff, J.; Sulaiman, S.; Arkles, B. Applications of Hybrid Polymers Generated from Living Anionic Ring Opening Polymerization. Molecules 2021, 26, 2755. [Google Scholar] [CrossRef]
- Shi, L.; Boulègue-Mondière, A.; Blanc, D.; Bacelredo, A.; Branchadell, V.; Kato, T. Ring-opening polymerization of cyclic oligosiloxanes without producing cyclic oligomers. Science 2023, 381, 1011–1014. [Google Scholar] [CrossRef]
- Fei, H.F.; Xie, W.; Wang, Q.; Gao, X.; Hu, T.; Zhang, Z.; Xie, Z. Controlled synthesis and characterization of poly [methyl(3,3,3-trifluoropropyl)siloxane] with selective end groups. RSC Adv. 2014, 4, 56279. [Google Scholar] [CrossRef]





| Entry | Reaction Time t [h] | 1H NMR Integral Ratio [Methyl:Vinyl] |
|---|---|---|
| 1 | 0.5 | 2:1 |
| 2 | 1 | 2:1 |
| 3 | 2 | 2:1 |
| Entry | x (Equiv.) | [THF]/[I] | t (h) | Average Si Number * |
|---|---|---|---|---|
| 1 | 9 | 2 | 5 | 6.8 |
| 2 | 9 | 2 | 24 | 5.8 |
| 3 | 0.9 | 0.2 | 5 | 6.3 |
| 4 | 0.9 | 0.2 | 24 | 5.5 |
| Entry | D3 (x mmol) | [D3]/[I] | Estimated Si Number [a] | Calculated Si Number [b] |
|---|---|---|---|---|
| 1 | 17.2 | 1.15 | 4.6 | 5.5 |
| 2 | 27 | 1.80 | 7.6 | 7.4 |
| 3 | 60 | 4.00 | 12.2 | 14.0 |
| Entry | Initiator (I′) [mmol] | D3 [mmol] | Hexane [mmol] | Toluene [mmol] | THF [mmol] | [THF]/[I′] | [D3]/[I′] | Np/P [a] | Si [b] | Si * [c] |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 15 | 22.5 | 72 | 14 | 838 | 55.9 | 1.5 | 0.10 | 11.4 | 6.5 |
| 2 | 15 | 22.5 | 72 | 14 | 170 | 11.3 | 1.5 | 0.50 | 11.2 | 6.5 |
| 3 | 15 | 22.5 | 72 | 14 | 55 | 3.7 | 1.5 | 1.55 | 10.4 | 6.5 |
| 4 | 15 | 22.5 | 80 | 23 | 32 | 2.1 | 1.5 | 3.20 | 8.3 | 6.5 |
| 5 | 6 | 6 | 29 | 6 | 2 | 0.33 | 1 | 17.5 | 6.5 | 5 |
| 6 | 6 | 6 | 44 | 15 | 2 | 0.33 | 1 | 29.5 | 3.9 | 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saotome, S.; Kuang, J.; Liu, Y.; Iijima, T.; Unno, M. Study of Reaction Parameters for the Precise Synthesis of Low-Molecular-Weight Oligosiloxanes. Materials 2025, 18, 5677. https://doi.org/10.3390/ma18245677
Saotome S, Kuang J, Liu Y, Iijima T, Unno M. Study of Reaction Parameters for the Precise Synthesis of Low-Molecular-Weight Oligosiloxanes. Materials. 2025; 18(24):5677. https://doi.org/10.3390/ma18245677
Chicago/Turabian StyleSaotome, Satoru, Jiaorong Kuang, Yujia Liu, Takayuki Iijima, and Masafumi Unno. 2025. "Study of Reaction Parameters for the Precise Synthesis of Low-Molecular-Weight Oligosiloxanes" Materials 18, no. 24: 5677. https://doi.org/10.3390/ma18245677
APA StyleSaotome, S., Kuang, J., Liu, Y., Iijima, T., & Unno, M. (2025). Study of Reaction Parameters for the Precise Synthesis of Low-Molecular-Weight Oligosiloxanes. Materials, 18(24), 5677. https://doi.org/10.3390/ma18245677









