Simple Summary
Renal cell carcinoma with upper-level (level III/level IV) tumor thrombus of the inferior vena cava is rare, and surgical management remains a mainstay of treatment. Cardiopulmonary bypass (CPB) is often used in these patients for hemodynamic management; however, concerns remain regarding its associated risks. Previous work has questioned whether CPB increases complication rates or worsens long-term outcomes for patients undergoing nephrectomy with upper-level thrombectomy. In this single-center study, we found no difference in complication rates or overall survival between patients managed with and without CPB. Although comparative data on surgical techniques are lacking, our findings add to the growing evidence that CPB use is not independently associated with complications or survival in patients undergoing nephrectomy with level III/IV thrombectomy.
Abstract
Radical nephrectomy with inferior vena cava (IVC) thrombectomy is a technically complex procedure. Cardiopulmonary bypass (CPB) is frequently employed for managing high-level thrombi, yet its impact on surgical outcomes remains uncertain. This study evaluated the outcomes of radical nephrectomy with level III/IV thrombectomy with or without CPB. We retrospectively reviewed records of patients with renal cell carcinoma and level III/IV (Mayo classification) thrombi who underwent open radical nephrectomy and IVC thrombectomy at our center between January 2000 and December 2023. Perioperative and survival outcomes were compared between patients in the CPB and non-CPB groups. Multivariable regression identified clinical factors associated with all-grade complications and survival. Primary and secondary outcomes were 90-day complications and overall survival. Fifty-seven patients were included: 30 (53%) in the CPB group and 27 (47%) in the non-CPB group. Within 90 days, overall complication and mortality rates were 49% and 10.5%; no statistically significant differences were observed between groups. In multivariable models, CPB was not independently associated with 90-day complications (odds ratio [OR] 0.55, 95% CI 0.13–2.12, p = 0.4) or overall survival (hazard ratio [HR] 1.34, 95% CI 0.65–2.78, p = 0.41). In our cohort, we did not find CPB use to independently influence perioperative complications or survival outcomes in radical nephrectomy with level III/IV IVC thrombectomy.
1. Introduction
Venous tumor invasion is present in 4–10% of renal cell carcinomas (RCC), with fewer than 1% of patients having tumor thrombus extension at or above the hepatic veins (level III/level IV tumor thrombus) [1]. The prognosis of RCC with tumor thrombus is grim, with a reported median survival of 5 months in an untreated population [2], though aggressive surgical management with complete tumor resection has been shown to provide long-term survival benefit and oncologic control in nonmetastatic patients [3]. Surgical management of these higher-level thrombi is technically complex and requires meticulous hemodynamic control. Cardiopulmonary bypass (CPB) is often employed in these cases, though data vary as to whether CPB has an impact on morbidity or mortality [4,5,6,7,8]. Various non-CPB techniques for management of higher-level thrombi [9,10,11] have been described, though comparative data among surgical techniques are lacking.
To address this question, we evaluated surgical and survival outcomes of patients undergoing open radical nephrectomy with IVC thrombectomy for RCC with level III/IV caval thrombus, comparing cases managed with and without CPB.
2. Materials and Methods
After institutional review board approval, we retrospectively reviewed the records of 57 patients with RCC and level III or IV caval thrombi who underwent open radical nephrectomy and IVC thrombectomy. The study cohort included patients treated at Duke University Medical Center (Durham, NC, USA) between January 2000 and December 2023. Pathologic staging was determined utilizing the 2009 AJCC TNM staging criteria, and patients treated prior to this were reclassified per criteria. Tumor thrombus level was determined per Mayo classification system [12] and confirmed on pre-operative cross-sectional imaging and/or intraoperative transesophageal echocardiography.
Use of CPB was determined on a case-by-case basis by a multidisciplinary team, and all CPB cases were performed jointly with both cardiothoracic suregeons and urologic oncologists. In total, cases represented patients from eight urologic oncologists and four cardiothoracic surgeons. Technique for CPB was as follows: After exposure of the kidney and intrabdominal IVC, including hepatic mobilization, a median sternotomy was made. Central aortic or axillary arterial cannulation was performed for inflow, and bicaval venous cannulation via peripheral femoral vein and superior vena cava was then performed for outflow. The SVC and IVC below the tumor thrombus are snared, and the patient is then placed on cardiopulmonary bypass. Resection of the tumor thrombus is performed via right atriotomy and vena cavotomy without aortic cross-clamp. A graphic depicting our typical exposure is provided in Figure 1. Deep hypothermic circulatory arrest (DHCA) was not utilized in any of the CPB cases. Operative technique for non-CPB cases was by surgeon preference; no patients were placed on venovenous bypass in our cohort.
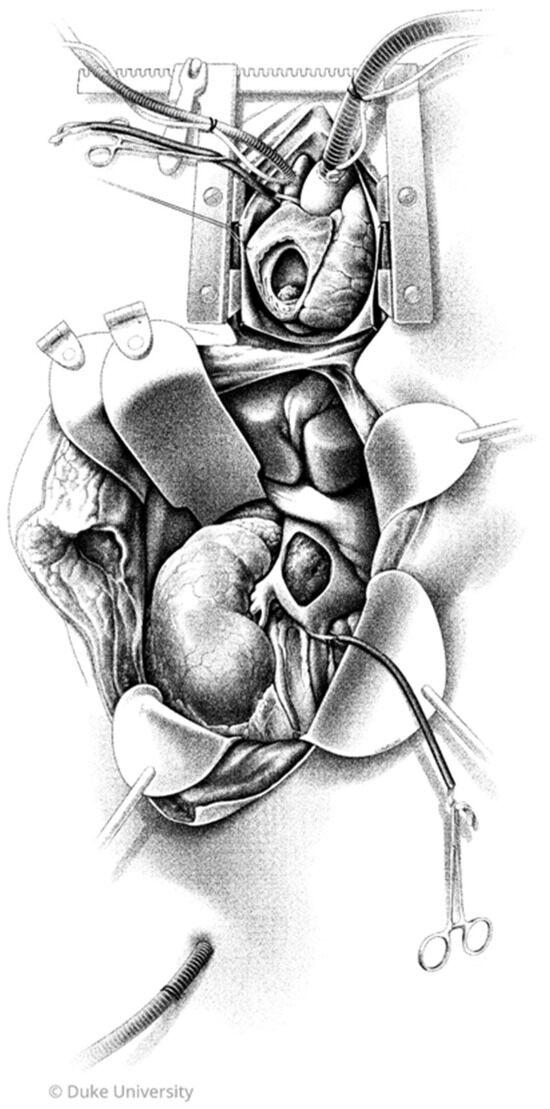
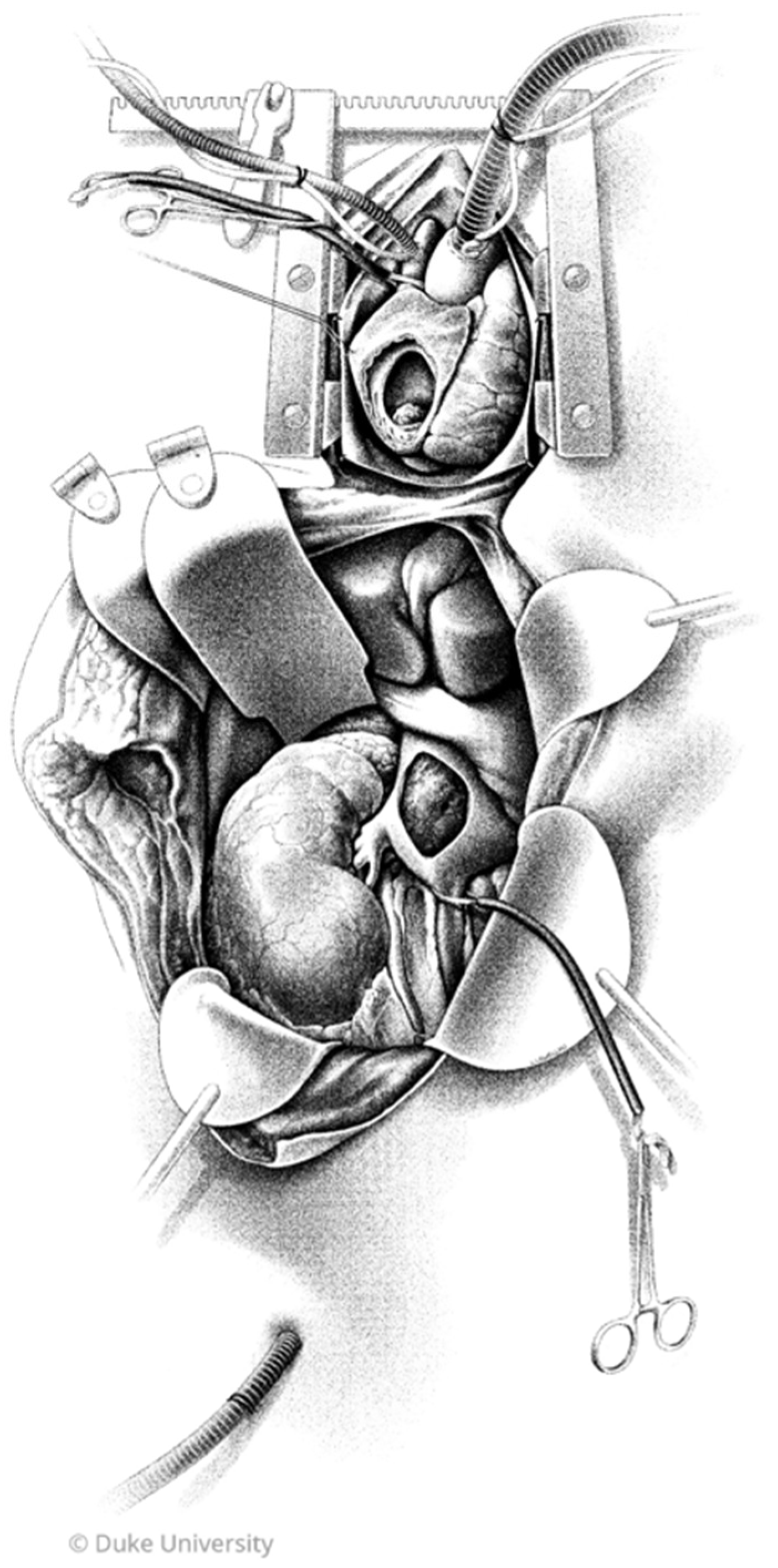
Figure 1.
Operative exposure for resection of a right renal cell carcinoma with tumor thrombus (TT) extension to right atrium. A Cattell–Braasch maneuver exposes the right kidney and IVC prior to initiating CPB. Central aortic cannulation and bicaval venous cannulation via peripheral femoral vein and superior vena cava are used for CPB. The SVC and IVC below the TT are snared. Inferior vena cavotomy and right atriotomy are performed to expose and remove the TT en bloc with radical nephrectomy. TT: tumor thrombus; RA: right atrium; IVC: inferior vena cava; CPB: cardiopulmonary bypass; SVC: superior vena cava. Illustrated by Megan Llewellyn, MSMI; copyright Duke University; with permission under a CC BY-ND 4.0 license.
Pre-operative angioembolization and regional lymphadenectomy were performed at surgeon discretion. Follow-up intervals were at the discretion of the treatment team and consisted of interval physical exams, laboratory testing, and cross-sectional imaging. Surgical complications were recorded within 90 days and classified with the Clavien–Dindo grading system and further grouped into low-grade (Clavien I–II) and high-grade (Clavien III–IV) complications [13].
Demographic and clinical variables included age, sex, body mass index (BMI), smoking status (current/former), pre-operative hemoglobin, pre-operative serum creatinine, estimated glomerular filtration rate (eGFR), Charlson Comorbidity Index, IVC thrombus level, tumor laterality, pre-operative embolization, metastasis on pre-operative imaging, year of surgery, CPB use, operative time in minutes, estimated blood loss (EBL), intraoperative blood product transfusion, length of hospital stay after index operation (days), tumor size, pathologic staging, 90-day post-operative complication rate, and 90-day mortality. The primary outcome was rates of overall and high-grade ninety-day complications between the CPB and non-CPB groups. The secondary outcome was comparison of overall survival between groups.
Clinical variables and peri-operative outcomes were compared between the CPB and non-CPB groups using the Wilcoxon rank-sum test for continuous variables and the Pearson chi-square test or Fisher’s exact test for categorical variables. Median follow-up was estimated with the reverse Kaplan–Meier method. Overall survival was compared between groups with the Kaplan–Meier (log-rank) method. Cox proportional hazards models with Firth’s penalized likelihood method were used to assess factors associated with overall survival. Firth’s penalized logistic regression was used to assess clinical factors associated with 90-day surgical complications. Candidate variables for multivariable models were selected based on clinical relevance and p < 0.10 on univariable analysis. CPB utilization was included in final models regardless of univariate results. All p-values reported were two-sided, and a p < 0.05 was considered statistically significant.
Analyses were performed using R version 4.5.1 with RStudio 2025.5.1.513 with the following packages installed: coxphf, ggplot2, ggpubr, ggsurvfit, gtsummary, logistf, lubridate, MASS, tidyverse, tableone, survival, survminer.
3. Results
A total of 57 patients were included, with 30 (53%) in the CPB group and 27 in the non-CPB group. Overall, 36 patients had level III and 21 had level IV IVC thrombi. Pre-operative characteristics are summarized in Table 1. All patients in the non-CPB group had a level III thrombus, whereas 9 of 30 (30%) of the CPB cohort had a level III thrombus and 21 of 30 (70%) had a level IV thrombus (p < 0.001). The proportion of patients with a Charlson Comorbidity Index score ≥ 1 was higher in the non-CPB cohort (56% vs. 27%; p = 0.03). The majority (80%) of CPB patients were treated between 2012 and 2023, compared to 52% of the non-CPB cohort (p = 0.049). No other demographic or pre-operative characteristic showed a statistically significant difference between groups.

Table 1.
Clinical features of patients, stratified by CPB use.
Intraoperative variables, pathologic outcomes, and complication rates are summarized in Table 2. Median operative time was longer in the CPB group (466 min) compared with the non-CPB group (317 min; p < 0.001). Transfusion requirements were similar between groups (median 2200 mL vs. 1400 mL; p = 0.2). No intraoperative deaths occurred.

Table 2.
Intra-operative variables and peri-operative outcomes stratified by CPB use.
Within 90 days post-surgery, the overall complication and mortality rates were 49% and 10.5%, respectively. Compared with non-CPB patients, those undergoing CPB showed no statistically significant difference in 90-day mortality (10% vs. 11%; p = 1.0) or in overall 90-day complications (50% vs. 48%; p = 1.0), including low-grade (27% vs. 26%) and high-grade (23% vs. 22%) complications. Counts of complications stratified by afflicted system are available in Table S1.
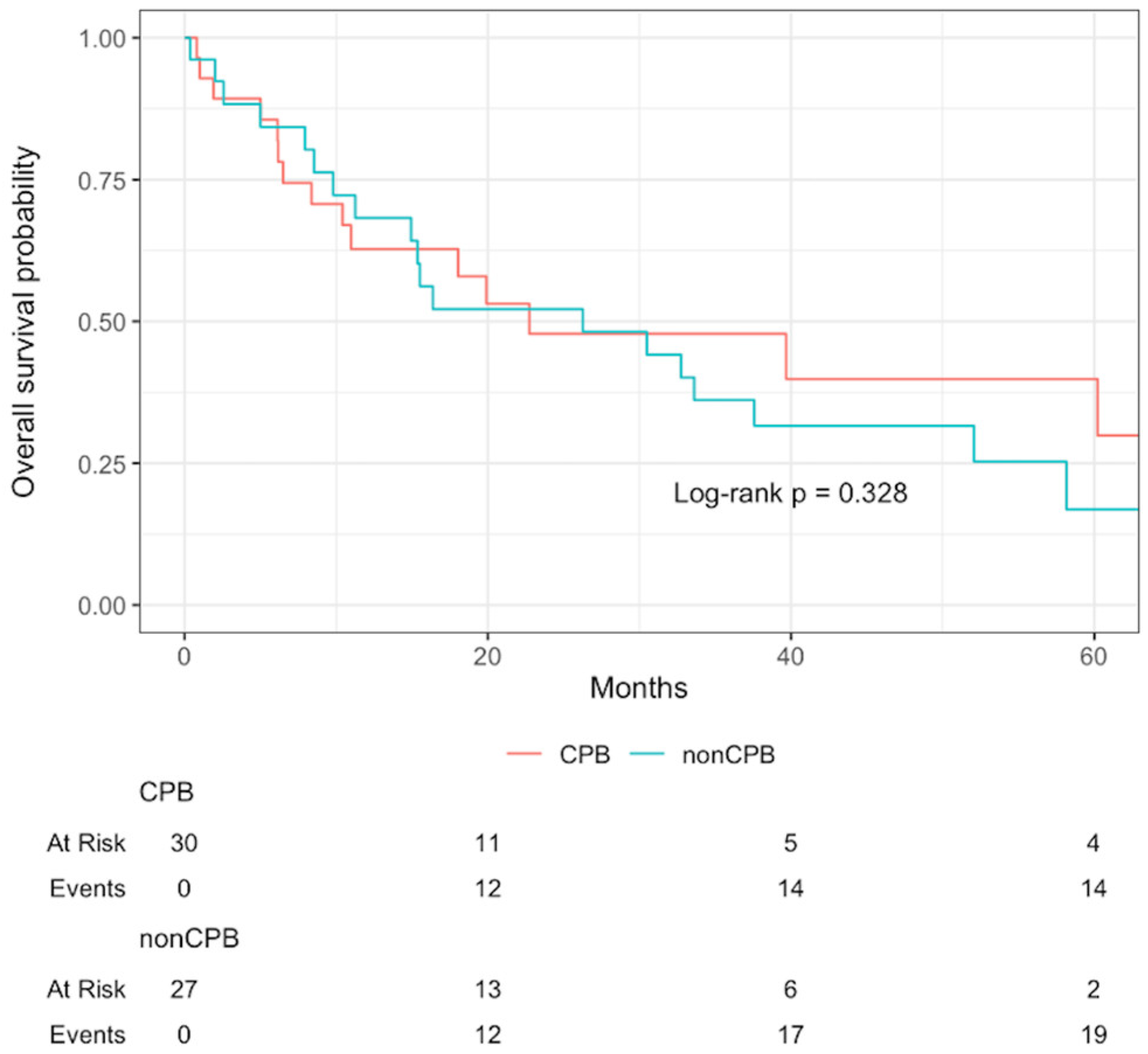
Overall median follow-up, estimated by reverse Kaplan–Meier, was 54.7 months (IQR 33.0–87.7). Three-year overall survival was 47.8% for patients undergoing CPB and 36.1% for those without CPB; the corresponding 5-year survival was 39.8% versus 16.9%, respectively. These differences were not statistically significant (p = 0.33), and Kaplan–Meier survival estimates are shown in Figure 2.
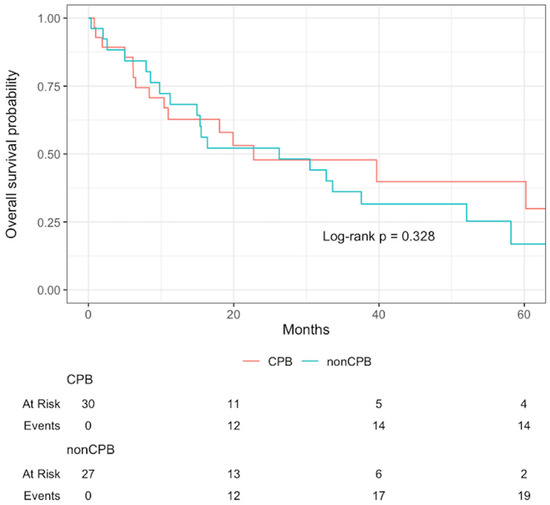
Figure 2.
Kaplan–Meier survival curves stratified by CPB use (univariate).
On univariate Cox analysis, clinical metastasis, pre-operative hemoglobin, pre-operative creatinine, and pathologic T stage were each significantly associated with increased hazard of mortality and included in the final multivariable model. Only pre-operative metastasis (hazard ratio [HR] 2.31, 95% CI: 1.05–4.93, p = 0.039) was found to be an independent predictor for increased hazard of mortality. Use of CPB was not associated with an increased hazard of mortality (HR 1.34, 95% CI 0.65–2.78, p = 0.41) (Table 3). This analysis was then repeated to include only patients treated between 2012 aand 2023, and no statistically significant association between OS and CPB use was seen (Table S2).

Table 3.
Multivariable Cox proportional hazards model with Firth’s penalized likelihood for predictors of mortality.
In analyses of 90-day complications, univariable logistic regression identified higher pre-operative serum creatinine and longer operative time as significant predictors. The final multivariable model included pre-operative serum creatinine, operative time, and CPB use. CPB was not independently associated with increased odds of a 90-day complication (odds ratio [OR] 0.55, 95% CI 0.13–2.12, p = 0.4). Longer operative time remained a significant predictor (OR 1.01, 95% CI: 1.0001–1.01, p = 0.03) (Table 4). A separate analysis was completed, including only patients treated between 2012 and 2023, and no statistically significant association between CPB use and post-operative complications was found (Table S3).

Table 4.
Multivariable Firth’s penalized logistic regression for variables associated with 90-day complication.
4. Discussion
Managing level III/IV tumor thrombus is one of the greatest challenges in urologic oncology, with continued debate over the risks and benefits of CPB. Concerns over the morbidity and mortality risks of CPB have led to non-CPB management strategies for hemodynamic control in high-level thrombectomy, including venovenous bypass [14,15], liver transplantation techniques, and intrapericardial control [10,11,16]. Though these alternatives to CPB have demonstrated success, it is unclear if CPB is associated with increased risk in RCC with higher-level thrombi. Our findings add to the evidence that CPB is not independently associated with increased complications or mortality in nephrectomy with upper-level thrombectomy.
Previous reports have called into question the risk of CPB’s impact on outcomes. Ngyeun et al. analyzed 362 patients with level III/IV tumor thrombus, comparing outcomes with and without CPB, and found no differences in complication or survival rates based on CPB use, consistent with our results [8]. Our observed high-grade complication rate of 22% aligns with prior reports of 19.5–45% [8,10,17,18,19]. We additionally did not find CPB to be associated with complication rates on multivariable analysis, in line with prior reports [5,8,20]. These findings are contrasted by work from Abel et al., who, in a multicenter study of 162 patients undergoing nephrectomy with upper-level thrombectomy, found CPB to be associated with major complications on univariate analysis (OR 2.16, 95% CI: 1.11–4.22; p = 0.02), though they did not include CBP in multivariable analysis [17]. Huang et al. have described a technique for robotic intrapericardial control for level IV tumor thrombi not involving the atria and reported a non-statistically significant lower rate of high-grade complication in their non-CPB group, though in a limited CPB-free sample (n = 8) [16]. A recent study by Cai et al. evaluated post-operative neurologic outcomes of patients undergoing nephrectomy with tumor thrombectomy and did not find CPB to be associated with post-operative cognitive impairment [21]. Our findings add to the growing body of evidence that for high-level tumors, CPB is not independently associated with peri-operative complications. These findings persisted when examining patients treated in the contemporary era, providing novel insight given the more historic cohorts reported in prior work.
While higher-level thrombus has been shown to be associated with worse survival outcomes, the contribution of CPB, if any, is undetermined. In multiple previous reports assessing survival outcomes, utilization of CPB was not associated with overall or cancer-specific survival for patients with level III/IV thrombus [5,8,15]. We found similar rates of 90-day mortality among our CPB and non-CBP cohorts (10% vs. 11%) and did not find CPB to be a predictor of mortality on either univariate or multivariate analysis. While not reaching statistical significance, we interestingly did see increased overall survival in our CPB cohort. Larger studies are needed, but this potentially could be related to pre-operative fitness for CPB or intensive intraoperative and post-operative monitoring of patients who went on CPB.
Advancements in CPB, including improvements in neurologic and hemodynamic monitoring, have been well described [22]. Shuch et al. demonstrated that deep hypothermic circulatory arrest (DHCA) was protective against perioperative mortality in patients with level IV thrombus (HR 0.13, 95% CI: 0.036–0.510; p = 0.003), though perioperative mortality in their non-DHCA group was 37.5%, notably higher than mortality rates found in our study [6]. These discrepancies may be explained by a more historic cohort, with their study population dating from 1983 to 2007. DHCA was not utilized in our cohort, and further work will be needed to assess its role.
There is no consensus on selecting candidates for CPB or non-CPB approaches for upper-level thrombectomy. Decisions are typically multidisciplinary and institution-dependent. In the case of level III thrombus, some authors advocate only for the use of CPB in patients who do not tolerate IVC clamping and have described non-CPB methods for selected level IV thrombi [23]. Advanced imaging may help elucidate thrombus characteristics pre-operatively and aid in surgical modality selection [24]. Collaboration between the urologic and cardiothoracic surgery teams remains essential. It should be noted that contemporary practice at our center has been to perform cardiac catheterization in all patients potentially undergoing CPB, aiming to optimize surgical candidacy. Given the rarity of these cases, comparative analysis between surgical techniques is limited. Future studies in larger cohorts may be able to directly compare non-CPB modalities both amongst each other and with CPB. Additionally, the role of neoadjuvant systemic immunotherapy with immune checkpoint inhibitors (ICI) prior to nephrectomy and thrombectomy continues to be defined, with a recent series reporting longer operative times but comparable perioperative morbidity among recipients of neoadjuvant ICI compared with a matched cohort of nonrecipients [25]. Further work should clarify the safety of CPB in patients who underwent neoadjuvant immunotherapy, with known increases in systemic inflammation after ICI that potentially could alter surgical outcomes.
Our study is not without limitations, given its nonrandomized, retrospective design and relatively small cohort with limited statistical power. Patients were treated at a single academic tertiary medical center, limiting generalizability, though given the complexity and rarity of these cases requiring multidisciplinary care, outcomes have been shown to be improved at high-volume centers [26]. Despite these limitations, we present contemporary results consistent with prior work, demonstrating that CPB was not independently associated with an increased risk of 90-day perioperative complications, short-term mortality, or overall survival. These findings support individualized decision-making regarding CPB use, informed by perioperative factors and multidisciplinary evaluation.
5. Conclusions
Cardiopulmonary bypass was not associated with 90-day postoperative complications or overall survival in our cohort of patients undergoing radical nephrectomy with caval thrombectomy for level III/IV tumor thrombus. Surgical decision-making in this setting is highly complex and multidisciplinary, and future work should focus on identifying optimal candidates for cardiopulmonary bypass.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/curroncol32120671/s1, Table S1: Post-operative complications stratified by CPB use. Table S2. Multivariable Cox proportional hazards model with Firth’s penalized likelihood for predictors of mortality for patients treated from 2012 to 2023. Table S3: Multivariable Firth’s penalized logistic regression for variables associated with 90-day complication for patients treated from 2012 to 2023.
Author Contributions
Conceptualization, J.V.D., A.G., J.R., F.S.M., A.R.B., J.J.F., A.R.W., A.S. and M.R.A.; Data curation, J.V.D., A.G., S.D., M.I., A.E.H. and F.S.M.; Formal analysis, J.V.D. and A.G.; Methodology, J.V.D., A.G., J.R., F.S.M., A.R.B., A.S. and M.R.A.; Supervision, A.R.W. and A.S.; Writing–original draft, J.V.D., A.G. and J.R.; Writing—review and editing, J.V.D., A.G., J.R., S.D., M.I., A.E.H., F.S.M., A.R.B., J.J.F., A.R.W., A.S. and M.R.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved on 6/28/2024 by the Institutional Review Board of Duke University Health System and was determined IRB exempt (protocol code Pro00116176) for studies involving humans.
Informed Consent Statement
Patient consent was waived due to the retrospective nature of the study with de-identified data.
Data Availability Statement
The datasets presented in this article are not readily available because of the protection of personal health information, with the dataset stored behind an institutional firewall. Requests to access the datasets should be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest related to this work. M.R.A. has consulted for Medtronic Inc. A.R.W. has consulted for Stryker Corporation. A.S. has received funding from Pfizer for unrelated research.
Abbreviations
The following abbreviations are used in this manuscript:
| BMI | Body Mass Index |
| CCI | Charlson Comorbidity Index |
| CPB | Cardiopulmonary Bypass |
| IVC | Inferior Vena Cava |
| HR | Hazard Ratio, OR: Odds Ratio |
| RCC | Renal Cell Carcinoma |
| SVC | Superior Vena Cava |
References
- Martínez-Salamanca, J.I.; Huang, W.C.; Millán, I.; Bertini, R.; Bianco, F.J.; Carballido, J.A.; Ciancio, G.; Hernández, C.; Herranz, F.; Haferkamp, A.; et al. Prognostic impact of the 2009 UICC/AJCC TNM staging system for renal cell carcinoma with venous extension. Eur. Urol. 2011, 59, 120–127. [Google Scholar] [CrossRef]
- Reese, A.C.; Whitson, J.M.; Meng, M.V. Natural history of untreated renal cell carcinoma with venous tumor thrombus. Urol. Oncol. Semin. Orig. Investig. 2013, 31, 1305–1309. [Google Scholar] [CrossRef] [PubMed]
- Ciancio, G.; Manoharan, M.; Katkoori, D.; De Los Santos, R.; Soloway, M.S. Long-term survival in patients undergoing radical nephrectomy and inferior vena cava thrombectomy: Single-center experience. Eur. Urol. 2010, 57, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Almatari, A.L.; Sathe, A.; Wideman, L.; Dewan, C.A.; Vaughan, J.P.; Bennie, I.C.; Buscarini, M. Renal cell carcinoma with tumor thrombus: A review of relevant anatomy and surgical techniques for the general urologist. Urol. Oncol. Semin. Orig. Investig. 2023, 41, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Suk-Ouichai, C.; Huang, M.M.; Neill, C.; Mehta, C.K.; Ross, A.E.; Kundu, S.D.; Perry, K.T., Jr.; Pham, D.T.; Patel, H.D. Utilization of cardiopulmonary bypass at radical nephrectomy for renal cell carcinoma with tumour thrombus. BJUI Compass 2025, 6, e460. [Google Scholar] [CrossRef]
- Shuch, B.; Crispen, P.L.; Leibovich, B.C.; Larochelle, J.C.; Pouliot, F.; Pantuck, A.J.; Liu, W.; Crepel, M.; Schuckman, A.; Rigaud, J.; et al. Cardiopulmonary bypass and renal cell carcinoma with level IV tumour thrombus: Can deep hypothermic circulatory arrest limit perioperative mortality? BJU Int. 2011, 107, 724–728. [Google Scholar] [CrossRef]
- Elahi, M.M.; Khan, J.S.; Matata, B.M. Deleterious effects of cardiopulmonary bypass in coronary artery surgery and scientific interpretation of off-pump’s logic. Acute Card. Care 2006, 8, 196–209. [Google Scholar] [CrossRef]
- Nguyen, H.G.; Tilki, D.; Dall’Era, M.A.; Durbin-Johnson, B.; Carballido, J.A.; Chandrasekar, T.; Chromecki, T.; Ciancio, G.; Daneshmand, S.; Gontero, P.; et al. Cardiopulmonary Bypass has No Significant Impact on Survival in Patients Undergoing Nephrectomy and Level III–IV Inferior Vena Cava Thrombectomy: Multi-Institutional Analysis. J. Urol. 2015, 194, 304–309. [Google Scholar] [CrossRef]
- Ciancio, G.; Shirodkar, S.P.; Soloway, M.S.; Livingstone, A.S.; Barron, M.; Salerno, T.A. Renal Carcinoma with Supradiaphragmatic Tumor Thrombus: Avoiding Sternotomy and Cardiopulmonary Bypass. Ann. Thorac. Surg. 2010, 89, 505–510. [Google Scholar] [CrossRef]
- Patil, M.B.; Montez, J.; Loh-Doyle, J.; Cai, J.; Skinner, E.C.; Schuckman, A.; Thangathurai, D.; Skinner, D.G.; Daneshmand, S. Level III–IV inferior vena caval thrombectomy without cardiopulmonary bypass: Long-term experience with intrapericardial control. J. Urol. 2014, 192, 682–689. [Google Scholar] [CrossRef]
- Gorin, M.A.; González, J.; Garcia-Roig, M.; Ciancio, G. Transplantation techniques for the resection of renal cell carcinoma with tumor thrombus: A technical description and review. Urol. Oncol. Semin. Orig. Investig. 2013, 31, 1780–1787. [Google Scholar] [CrossRef] [PubMed]
- Neves, R.J.; Zincke, H. Surgical treatment of renal cancer with vena cava extension. Br. J. Urol. 1987, 59, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Simon, R.M.; Kim, T.; Espiritu, P.; Kurian, T.; Sexton, W.J.; Pow-Sang, J.M.; Sverrisson, E.; Spiess, P.E. Effect of utilization of veno-venous bypass vs. cardiopulmonary bypass on complications for high level inferior vena cava tumor thrombectomy and concomitant radical nephrectomy. Int. Braz. J. Urol. 2015, 41, 911–919. [Google Scholar] [CrossRef]
- Granberg, C.F.; Boorjian, S.A.; Schaff, H.V.; Orszulak, T.A.; Leibovich, B.C.; Lohse, C.M.; Cheville, J.C.; Blute, M.L. Surgical management, complications, and outcome of radical nephrectomy with inferior vena cava tumor thrombectomy facilitated by vascular bypass. Urology 2008, 72, 148–152. [Google Scholar] [CrossRef]
- Huang, Q.; Zhao, G.; Chen, Y.; Wu, P.; Li, S.; Peng, C.; Liu, K.; Yu, H.; Gao, Y.; Xiao, C.; et al. Robotic Level IV Inferior Vena Cava Thrombectomy Using an Intrapericardial Control Technique: Is It Safe Without Cardiopulmonary Bypass? J. Urol. 2023, 209, 99–110. [Google Scholar] [CrossRef]
- Abel, E.J.; Thompson, R.H.; Margulis, V.; Heckman, J.E.; Merril, M.M.; Darwish, O.M.; Krabbe, L.M.; Boorjian, S.A.; Leibovich, B.C.; Wood, C.G. Perioperative outcomes following surgical resection of renal cell carcinoma with inferior vena cava thrombus extending above the hepatic veins: A contemporary multicenter experience. Eur. Urol. 2014, 66, 584–592. [Google Scholar] [CrossRef]
- Nini, A.; Capitanio, U.; Larcher, A.; Dell’Oglio, P.; Dehò, F.; Suardi, N.; Muttin, F.; Carenzi, C.; Freschi, M.; Lucianò, R.; et al. Perioperative and Oncologic Outcomes of Nephrectomy and Caval Thrombectomy Using Extracorporeal Circulation and Deep Hypothermic Circulatory Arrest for Renal Cell Carcinoma Invading the Supradiaphragmatic Inferior Vena Cava and/or Right Atrium. Eur. Urol. 2018, 73, 793–799. [Google Scholar] [CrossRef]
- Casey, R.G.; Raheem, O.A.; Elmusharaf, E.; Madhavan, P.; Tolan, M.; Lynch, T.H. Renal cell carcinoma with IVC and atrial thrombus: A single centre’s 10 year surgical experience. Surgeon 2013, 11, 295–299. [Google Scholar] [CrossRef]
- Haddad, A.Q.; Leibovich, B.C.; Abel, E.J.; Luo, J.-H.; Krabbe, L.-M.; Thompson, R.H.; Heckman, J.E.; Merrill, M.M.; Gayed, B.A.; Sagalowsky, A.I.; et al. Preoperative multivariable prognostic models for prediction of survival and major complications following surgical resection of renal cell carcinoma with suprahepatic caval tumor thrombus. Urol. Oncol. Semin. Orig. Investig. 2015, 33, 388.e1–388.e9. [Google Scholar] [CrossRef]
- Cai, X.; Huang, J.; Yao, X.; Qian, H.; Zhang, J.; Kong, W.; Wu, X.; Huang, Y.; Chen, Y.; Xue, W. Cognitive Function After Cardiopulmonary Bypass and Deep Hypothermic Circulatory Arrest in Management of Renal Cell Carcinoma with Vena Caval Thrombus. Urology 2022, 167, 144–151. [Google Scholar] [CrossRef]
- Ripoll, J.G.; Bittner, E.A.; Zaremba, S.; Nabzdyk, C.S.; Seelhammer, T.G.; Wieruszewski, P.M.; Chang, M.G.; Ramakrishna, H. Analysis of 2024 EACTS/EACTAIC/EBCP Guidelines on Cardiopulmonary Bypass in Adult Cardiac Surgery. J. Cardiothorac. Vasc. Anesthesia 2025, 39, 1853–1865. [Google Scholar] [CrossRef]
- Tabbara, M.M.; González, J.; Ciancio, G. The surgical evolution of radical nephrectomy and tumor thrombectomy: A narrative review. Ann. Transl. Med. 2023, 11, 262. [Google Scholar] [CrossRef]
- Kowal, P.; Ratajczyk, K.; Bursiewicz, W.; Trzciniecki, M.; Marek-Bukowiec, K.; Rogala, J.; Kowalskyi, V.; Dragasek, J.; Botikova, A.; Kruzliak, P.; et al. Differentiation of solid and friable tumour thrombus in patients with renal cell carcinoma: The role of MRI apparent diffusion coefficient. Adv. Med. Sci. 2024, 69, 434–442. [Google Scholar] [CrossRef]
- Khene, Z.E.; Bhanvadia, R.; Tachibana, I.; Issa, W.; Graber, W.; Trevino, I.; Woldu, S.L.; Gaston, K.; Zafar, A.; Hammers, H.; et al. Surgical Outcomes of Radical Nephrectomy and Inferior Vena Cava Thrombectomy Following Preoperative Systemic Immunotherapy: A Propensity Score Analysis. Clin. Genitourin. Cancer 2025, 23, 102307. [Google Scholar] [CrossRef]
- Freifeld, Y.; Woldu, S.L.; Singla, N.; Clinton, T.; Bagrodia, A.; Hutchinson, R.; Lotan, Y.; Margulis, V. Impact of Hospital Case Volume on Outcomes Following Radical Nephrectomy and Inferior Vena Cava Thrombectomy. Eur. Urol. Oncol. 2019, 2, 691–698. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).