Abstract
Background and Objectives: Biological therapies improve disease severity and quality of life in patients with psoriasis, but data on Romanian patients remain limited. Our study aimed to characterize patients with psoriasis from Transylvania and to evaluate the impact of biologics on disease severity, treatment switching, affected special areas response, quality of life, and laboratory biomarkers. Materials and Methods: We conducted a retrospective exploratory study at two centers in Cluj-Napoca, Romania, using routinely collected medical data. Results: One-hundred and fifteen patients (aged 2–72 years) were evaluated; 45 patients received anti-TNF, 43 received anti-IL-17, and 27 received anti-IL-23. Patients treated with anti-IL-17 or anti-IL-23 were older at diagnosis than those treated with anti-TNF (p = 0.0001). Psoriatic lesions were prevalent in the scalp (58.3%) and nails (36.5%). Methotrexate was the most common prior systemic therapy (87.8%), with no difference between the groups (p = 0.7668). Patients receiving anti-TNF therapy (46.7%) or anti-IL-17 therapy (20.9%) also most frequently received prior treatment with systemic retinoids. Cardiometabolic comorbidities, including hypertension (40.9%) and diabetes mellitus (20.9%), were prevalent. Anti-IL-17 therapies were used more frequently in patients with hypertension (46.5%), diabetes mellitus (34.9%), and psoriatic arthritis (34.9%). Baseline severity scores were comparable across the groups (p > 0.10). A therapeutic switch occurred in approximately one-quarter of the patients, most frequently in the anti-TNF group (57.8%), which also showed higher PASI and DLQI scores at switching (p < 0.0001). At 36 weeks, anti-IL-17 and anti-IL-23 therapies demonstrated superior outcomes compared to anti-TNF therapy (p = 0.045). All patients receiving anti-IL-23 therapy achieved a PASI 100 at the 60-week follow-up. Significant improvements in PASI and DLQI were observed for all biologics (p < 0.0001). Conclusions: Biological therapies were associated with significant improvements in disease severity and quality of life. Anti-TNF therapies were switched more frequently due to reduced efficacy, while clinical improvement was observed regardless of lesion localization.
1. Introduction
Psoriasis is a chronic, immune-mediated inflammatory disease that primarily affects the skin and joints [1]. The prevalence of psoriasis in Europe, North America, and Africa is 4.6%, 3.7%, and 1.7%, respectively [2]. Differences also exist in psoriasis prevalence across Europe, with higher rates in high-income countries and in Central and Western Europe [3]. In Poland, psoriasis affects 1.7% of the population, with slightly higher rates in women than in men (1.8% in Polish women vs. 1.58% in Polish men) [4]. In Romania, the prevalence of psoriasis is 4.2–5.18% [5,6], while the highest reported rate is in Norway at 8.50% [7].
Plaque-type psoriasis is the most common form of the disease [8], whereas other variants include generalized pustular psoriasis (von Zumbusch type), localized pustular psoriasis of the palms and soles, acrodermatitis continua of Hallopeau, inverse psoriasis, guttate psoriasis, and erythrodermic psoriasis [8].
The diagnosis is clinical [9], and severity is assessed using the Psoriasis Area Severity Index (PASI) and Body Surface Area (BSA) [9]. Localizations of lesions are assessed using the Psoriasis Scalp Severity Index (PSSI) [10], Nail Psoriasis Severity Index (NAPSI) [11], and Genital Psoriasis Symptoms Scale (GPSS) [12]. Quality of life is evaluated using the Dermatology Life Quality Index (DLQI) [13]. The disease is considered severe when psoriatic lesions are localized in specific areas or when PASI and DLQI exceed 10 points [14].
Psoriasis etiopathogenesis involves an interaction between genetic predisposition and environmental triggers [15], affecting not only the skin but also the joints, cardiovascular and metabolic systems, and psychological well-being [1,16]. Over 109 susceptibility loci have been identified [17], and triggers include infections, trauma, drugs, and autoantigens [18]. Immune dysregulation in psoriasis involves both the innate and adaptive immunity [19]. The key pathway that perpetuates inflammation is the TNFα-IL-23-Th17 axis [20]. Interleukins that play a significant role in pathogenesis are targets of biological therapies [21].
Management includes topical treatment, phototherapy, systemic conventional agents, and biologics [14]. Biological therapy targets TNFα (Tumor Necrosis Factor-alpha), IL-17 (IL-interleukin), IL-23, or IL-12/23, which are key interleukins implicated in etiopathogenesis [14]. Treatment choices depend on severity, site involvement, and comorbidities, and must address both skin and joint disease, as well as associated conditions [14,22,23].
Patients with psoriasis have an increased risk of atherosclerotic disease compared to the general population [24], with higher intima-media thickness (IMT) and more frequent atherosclerosis than controls [25]. Early onset and severe psoriasis further increase cardiovascular risk [26], likely via the IL-23–IL-17 axis [27].
Few original studies have been conducted on patients with psoriasis in Romania. Dascălu et al. found that metabolic syndrome (MetS) is more common in patients with psoriasis [28]. Additionally, patients with psoriasis had a higher IMT and more frequent atherosclerotic plaques [28]. The percentage of carotid artery plaques is higher in patients with psoriasis and MetS [28]. Cardiovascular disease is the most frequent comorbidity, followed by hypertension, dyslipidemia, and diabetes mellitus [5,6]. Regarding treatment response, Bucur et al. reported greater early PASI improvement with Ixekizumab than Secukinumab [29]. Psoriasis also carries a substantial psychological burden [30,31], including depression, low self-esteem, anger [30], anxiety [31], stigma [6], and a reduced quality of life [6,32].
Studies on patients with psoriasis in Romania have focused on the prevalence, characteristics, and quality of life [4,5] without evaluation of evolution under biological therapies. In the context of this gap, our retrospective study had twofold aims: to evaluate the therapy-switching rate and evolution of biologic-naïve patients under biological therapy using severity scores over time, and to analyze lesions from special areas response and the effects, if any, of biological treatment on measured laboratory biomarkers and of the skin condition on the patient’s life reflected by the DLQI score. The primary endpoints were the proportion of biological-naïve patients with psoriasis who required a switch in biological therapy during follow-up and treatment effectiveness, assessed by PASI response (PASI 100, PASI ≥ 90, PASI ≥ 75, PASI ≥ 50, and PASI < 50) and changes in DLQI scores evaluated at 36 and 60 weeks of treatment. Secondary endpoints included time to clinical response of psoriatic lesions located in special areas and changes in laboratory parameters (glycemia, creatinine, serum urea, and serum lipid levels) from baseline to 36 and 60 weeks of follow-up.
2. Materials and Methods
The study has been approved by the following Ethics Committees: Infectious Diseases Clinical Hospital, Cluj-Napoca Ethics Committee, approval 20948/29.11.2024; “Iuliu Hațieganu” University of Medicine and Pharmacy, Cluj-Napoca Ethics Committee, approval 55460/5.07.2023; County Emergency Hospital, Cluj-Napoca Ethics Committee, approval 55460/27.11.2023. This study was conducted in accordance with the principles of the Declaration of Helsinki.
2.1. Study Design and Variables
This retrospective study was conducted at the Dermatology Department of the County Emergency Hospital, Cluj-Napoca, and the Integrated Ambulatory-Infectious Diseases Clinical Hospital, Cluj-Napoca. Biologically naïve patients with histopathologically proven psoriasis vulgaris who received biological treatment with anti-TNF, anti-IL-17, or anti-IL-23, between 2010 and 2025, were eligible for the study.
Data were retrospectively collected from hospital and electronic medical records, laboratory databases, and the Romanian National Registry for Dermato-Venereological Disorders between January 2025 and October 2025. In Romania, treatment decisions are individualized and guided by the national protocol, the “Therapeutic protocol for severe chronic psoriasis (plaque psoriasis)—biological agents and small molecule therapies with intracellular action” [33]. The choice of biological agent is made in compliance with current legislation, based on the clinical characteristics of the disease, the patient’s age, pre-existing comorbidities, and the treating dermatologist’s experience and local facilities’ capabilities [33]. For the initiation of biological treatment, the histopathological certification of psoriasis is mandatory [33]. Additionally, for the initiation of treatment and monitoring of patients undergoing biological therapy, several laboratory investigations are necessary (i.e., complete blood count, erythrocyte sedimentation rate, liver and kidney function tests, electrolytes (sodium, potassium), tuberculin skin test/Interferon-Gamma Release Assay (except for apremilast), lung X-ray, screening for hepatitis B and C) [33]. The patients must also present a certificate from their family doctor stating the chronic diseases they have [33]. According to the national protocol, psoriasis vulgaris is classified as severe when PASI > 10 or lesions are located in topographic regions associated with significant functional impairment and/or high level of suffering and/or difficult to treat: face, scalp, palms, soles, nails, genital region, large folds, quantified by area scores [33]. The inclusion criteria for the initiation of biological treatment for adult patients with psoriasis vulgaris are: the presence of severe plaque psoriasis defined as (involvement of more than 10% of the body or PASI ≥ 10 or lesions located in topographic regions associated with significant functional impairment and/or high level of suffering and/or difficult to treat: face, scalp, palms, soles, nails, genital region, large folds) for more than 6 months, together with a DLQI ≥ 10, eligibility for biological therapy and failure/intolerance/contraindication for conventional systemic therapy [33]. The therapeutic target is defined as a decrease in PASI score of 50% from baseline (including 50% of the specific scores for the special areas affected) with a long-term objective of reaching an average remission of lesions of 90% [33]. Regarding DLQI, a minimum 5-point decrease from baseline is expected, with a long-term objective of reaching an absolute value of no more than 2 [33]. Discontinuation of biological treatment is indicated when the therapeutic target has not been achieved, as assessed by the therapeutic target [33]. Discontinuation of treatment is also indicated in the event of a severe adverse reaction [33]. Biological treatment switch is required in the case of patients who, upon evaluation, do not reach or maintain the therapeutic target during treatment or who have developed an adverse reaction [33]. The biological agent can be changed to another biological agent from the same therapeutic class only once in succession [33]. The national protocol also includes recommendations for the pediatric population (age 4 to 18) for biological therapy for psoriasis vulgaris [33].
The extracted variables included demographic data (sex, age, ethnicity), clinical data (body mass index [BMI], presence of overweight or obesity, age at diagnosis, comorbidities at biological treatment initiation, treatment details, severity scores (PASI), and effect of skin condition on the patient’s life reflected by DLQI score), and routinely measured laboratory biomarkers (at the time of biological treatment initiation, at 36 weeks, and at 60 weeks). The raw data for laboratory measurements were collected as continuous variables and then transformed into a nominal variable, with values classified as normal, low, or high based on the normal values of the two laboratories, because different devices, laboratory kits, and ranges were used.
All data were anonymized before statistical analysis to maintain patient confidentiality.
2.2. Analysis of Data
An exploratory statistical analysis was conducted using the Jamovi (v. 2.6.26.0) software at a level of significance of 5%. Qualitative data are reported as numbers (percentages), and differences between groups (biological therapy) were tested using Fisher’s exact test. Distribution was tested using the Shapiro–Wilk test prior to analysis for all quantitative variables, and data were reported following a free-distribution analysis when the normality assumption was not met.
The PASI and DLQI scores were evaluated at different time points using Friedman’s test, followed by post hoc analysis using Dwass–Steel–Critchlow–Fligner for pairwise comparisons when Friedman’s test was statistically significant. The psoriasis area and severity index (PASI) score (range, 0–72) was used to classify disease severity as mild (0 < PASI ≤ 5), moderate (5 < PASI ≤ 10), or severe (PASI ≥ 11) at baseline. The effect of the skin condition on the patient’s life was reported to have no effect (0 < DLQI ≤ 1), small effect (2 < DLQI ≤ 5), moderate effect (6 < DLQI ≤ 10), very large effect (11 < DLQI ≤ 20), and extremely large effect (21 < DLQI ≤ 30). Correlation analysis between baseline BMI, baseline PASI, and DLQI scores was performed using Spearman’s correlation coefficient. Graphical representations were constructed using Microsoft Excel.
No imputation method was used to handle the missing data, and the number of patients per group was reported whenever missing data were present.
3. Results
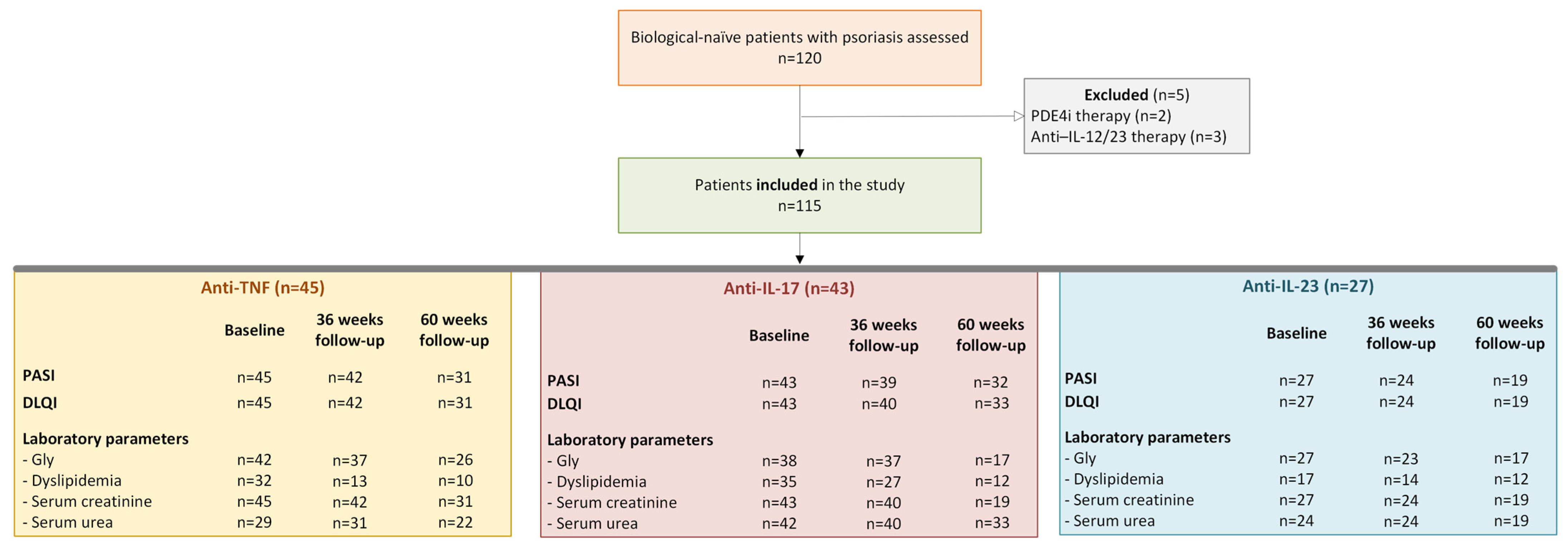
One hundred and twenty biologic-naïve patients with a histopathological diagnosis of psoriasis and eligible for biological therapy were assessed for eligibility. Of these, 115 patients aged 2–72 years were included in the study. Five patients were excluded: two because of prior PDE4 inhibitor therapy and three because of prior anti-IL-12/23 biological treatment. The included patients were allocated to the following biological treatment groups: anti-TNF (n = 45), anti-IL-17 (n = 43), and anti-IL-23 (n = 27). Fifty-four patients received biological therapy within 6 months of disease onset, with a time span of 1–38 years from disease onset to biological therapy initiation. The flow diagram of patient selection and follow-up is presented in Figure 1.
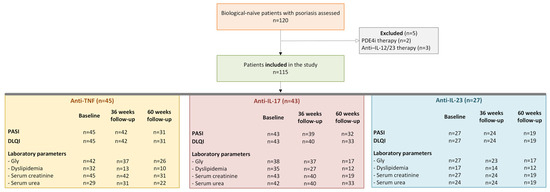
Figure 1.
Flow diagram of patient selection and follow-up. n- the number of patients; Gly—glycemia; PASI—Psoriasis Area Severity Index; DLQI—The Dermatology Life Quality Index; Anti-TNF—anti-tumor necrosis factor alpha biological therapy; Anti-IL-17—anti-interleukin 17 biological therapy; Anti-IL-23—anti-interleukin 23 biological therapy.
3.1. Characteristics of the Evaluated Cohort
The characteristics of the patients undergoing biological treatment are presented in Table 1. The most prevalent comorbidity was hypertension, followed by diabetes mellitus (Table 1), with a heterogeneous distribution according to biological therapy class. A limited number of patients had heart failure (two patients), ischemic cardiomyopathy (five patients), myocardial infarction (one patient), stroke (one patient), peripheral artery disease (one patient), neoplasia (five patients), chronic kidney disease (one patient), tuberculosis (six patients), or infection (five patients). All patients received topical treatment before biologics, except for three; the rest received systemic conventional therapy.

Table 1.
Baseline patient characteristics by initial biological treatment class.
Baseline BMI data was available for 88 patients. No statistically significant associations were found between baseline BMI and baseline PASI (Spearman’s ρ = −0.14, p = 0.2053) or between baseline BMI and baseline DLQI (Spearman’s ρ = −0.05, p = 0.6422)
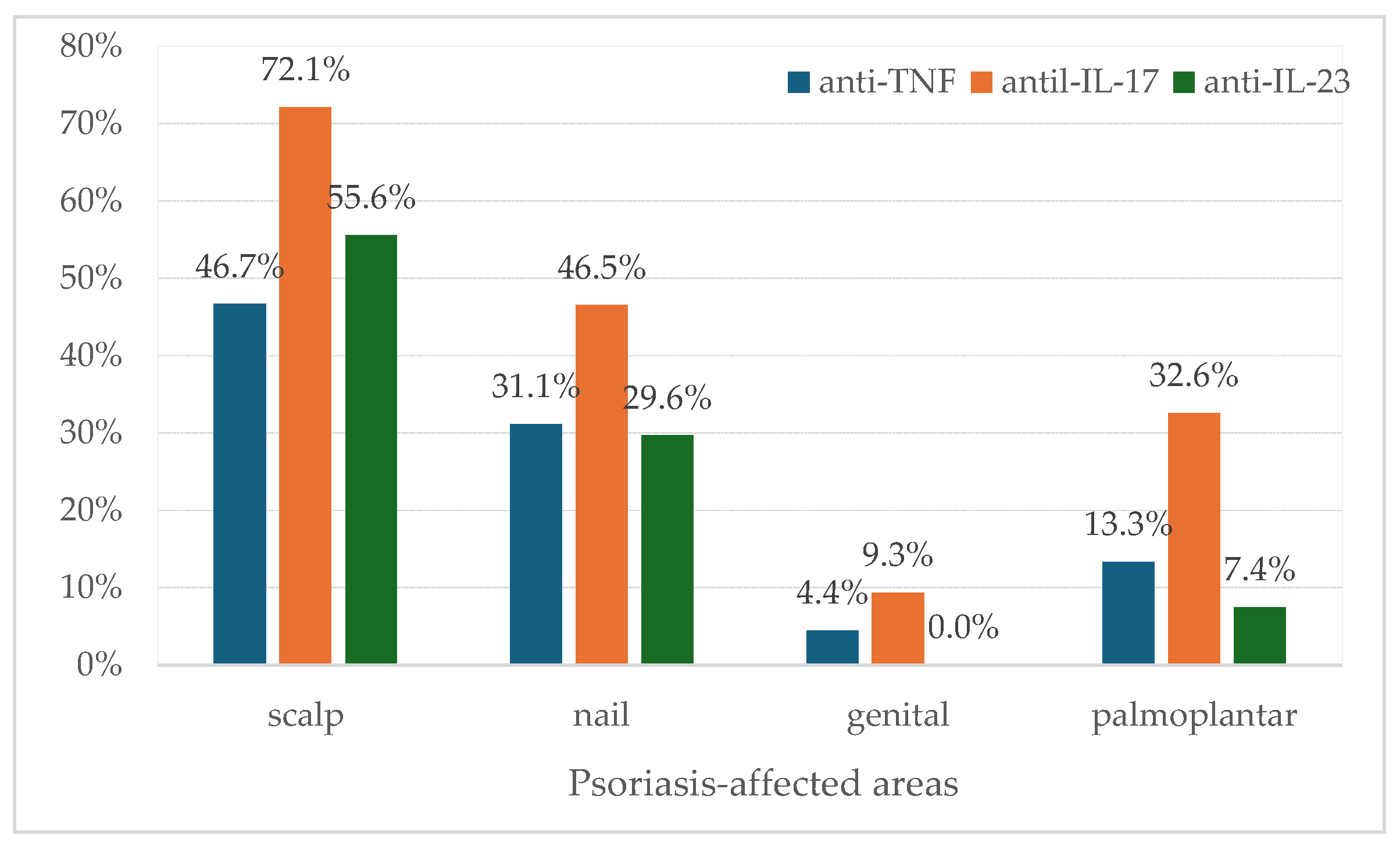
The most prevalent psoriatic lesions were localized to the scalp (58.3%) and nails (36.5%) (Figure 2).
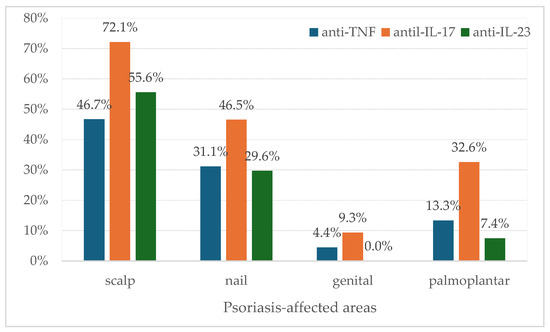
Figure 2.
Distribution of psoriasis-affected regions before initiation of biological therapy. Statistical significance between groups was observed only for palmoplantar (Fisher’s exact test: p = 0.0191) and for scalp (Fisher’s exact test: p = 0.0501).
3.2. Disease Severity Dynamics Under Biological Treatment
A biological switch was observed in 29 patients: 26 (57.8%) had initial anti-TNF (17 cases with one switch and nine cases with two switches) and three (7.0%, with a single switch per case) anti-IL-17 (Fisher’s exact test: p < 0.0001). The switch was due to ineffectiveness (26 cases), multiple sclerosis (one case, under anti-TNF therapy), and paradoxical psoriasis (another case, under anti-TNF therapy). The need for switching occurred between 1 and 12 months after initiation, with a median of 6 months and an interquartile range (IQR) of 5 to 10 months.
Similar baseline PASI scores were observed across the biological treatment groups, with higher values at the time of therapy switch and at 36 and 60 weeks of follow-up for patients with anti-TNF (Table 2). A change towards mild PASI scores and no effects on quality of life were observed in all groups (Table 2).

Table 2.
Severity scores by type of initial biological treatment.
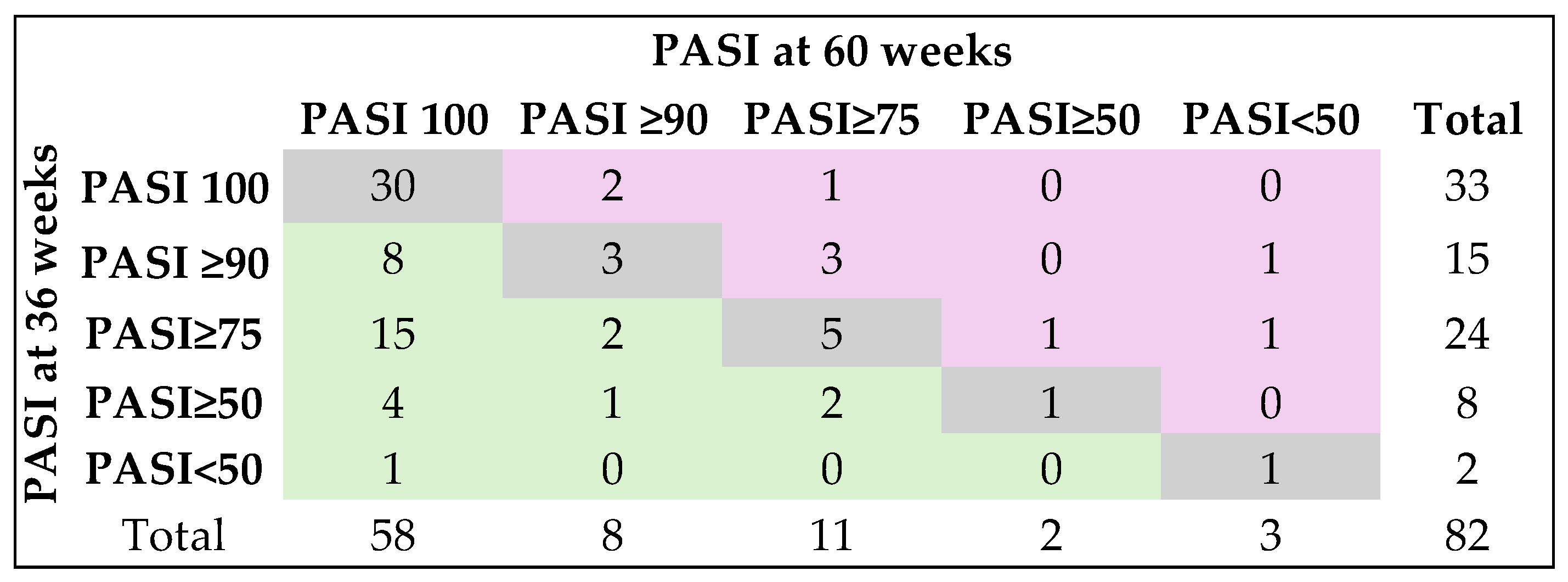
Regardless of the biological treatment group, the PASI score changed significantly from baseline to 60 weeks follow-up, with statistically significant differences between pairs of determinations (p < 0.0001), with one exception observed in the anti-IL-17 group, where PASI scores at 36 weeks were not statistically significant compared to those at 60 weeks follow-up (Durbin-Conover Pairwise comparisons: p = 0.1295). PASI remained the same at 60 weeks compared to 36 weeks for 40 (48.8%) patients, changed positively in 33 (40.2%) patients, and negatively in nine (11%) patients (analysis conducted on 82 patients) (Fisher’s exact test: p = 0.0201) (Figure 3).
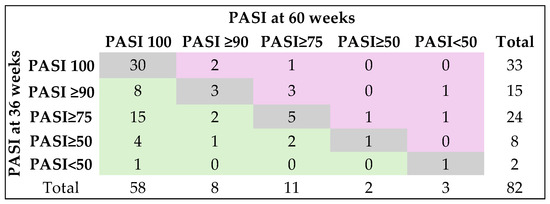
Figure 3.
Changes in PASI between weeks 36 and 60 in the evaluated cohort. Gray color (diagonal cells) indicates no change in response category (patients stayed in the same PASI response group at week 60 as they were at week; Green color (cells to the left of the diagonal) indicates improvement (patients moved to a better PASI category by week 60 of follow-up; Pink color (cells to the right of the diagonal) indicates worsening (patients moved to a lower PASI category by week 60 compared with week 36 of follow-up).
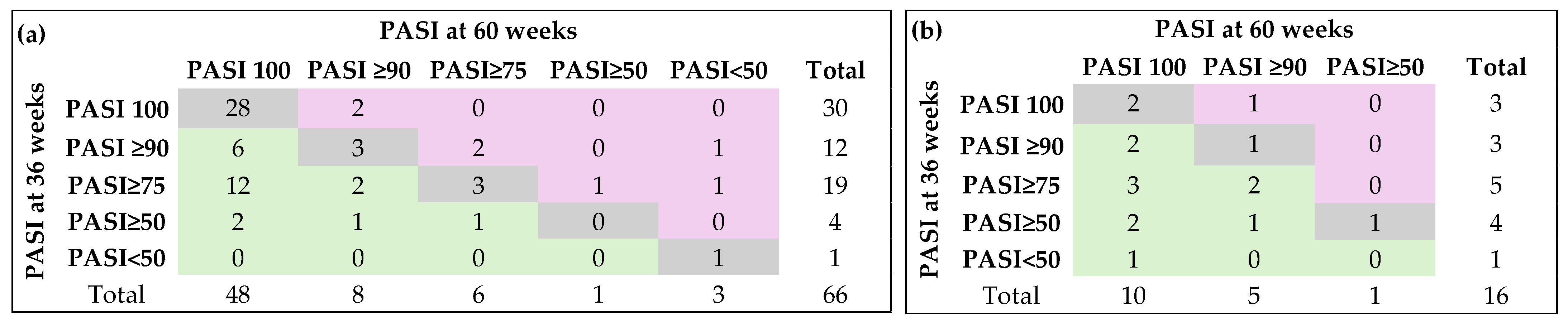
The change in PASI score was statistically significant only in patients who did not switch to therapy (Figure 4a, Fisher’s exact test: p = 0.0046).
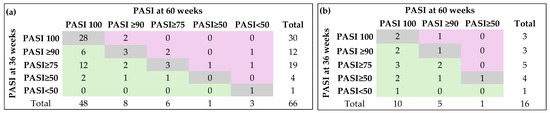
Figure 4.
Dynamics of PASI score: (a) patients without biological therapy switching (n = 66); (b) patients with biological therapy switching (n = 16). Gray color (diagonal cells) indicates no change in response category (patients stayed in the same PASI response group at week 60 as they were at week; Green color (cells to the left of the diagonal) indicates improvement (patients moved to a better PASI category by week 60 of follow-up; Pink color (cells to the right of the diagonal) indicates worsening (patients moved to a lower PASI category by week 60 compared with week 36 of follow-up).
The PASI scores decreased over time, but the values remained higher in patients with lesions in specific locations than in those without lesions (Table 3).

Table 3.
Dynamics of PASI scores by lesion localization.
Adverse events were observed in two patients receiving topical therapy, in three patients who developed pruritus and erythema after phototherapy, and in 24 patients receiving systemic therapy. Treatment with MTX was associated with cytopenia (one case), digestive intolerance (three cases), hepatic cytolysis (eight cases), nausea (two cases), cholestasis (one case), and pulmonary fibrosis (two cases). Treatment with cyclosporine was associated with increased creatinine (one case), hypertension (four cases), and osteodynia (one case). Retinoid treatment was associated with metabolic syndrome (three cases), osteodynia (one case), abdominal pain (two cases), digestive adverse events (one case), and hepatic cytolysis (one case).
3.3. Special Areas Response, Patient-Reported Outcomes, and Biomarker Modulation
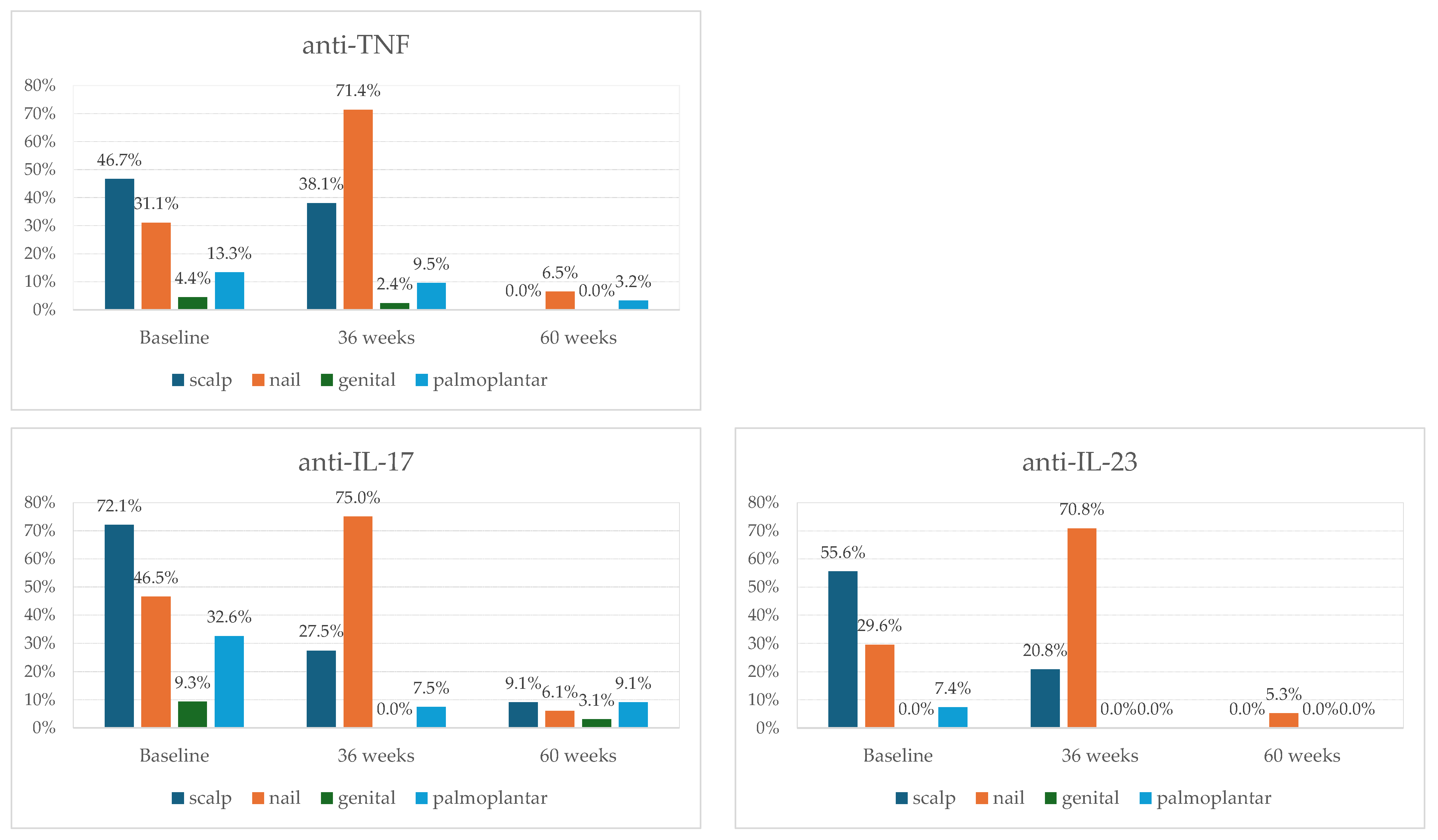
Biological therapy improved disease manifestations, independent of the initial biological agent used (Figure 5).
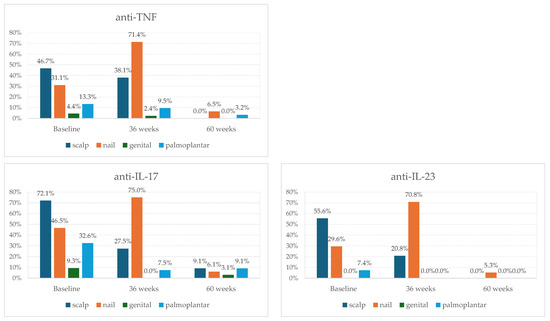
Figure 5.
Dynamics of psoriasis-affected special areas by class of the initial biological therapy (n = 115 baseline, n = 106 at 36 weeks of follow-up, and n = 83 at 60 weeks of follow-up).
Fridman’s ANOVA identified significant changes in the DLQI scores at baseline, at the switch, and at 60 weeks of follow-up (Table 4). A change towards no effects on quality of life at 36 and 60 weeks of follow-up was observed in all groups (Table 4).

Table 4.
Dynamics of DLQI scores by type of initial biological treatment.
The DLQI scores decreased over time, regardless of lesion localization, with higher scores observed in patients with lesions localized to special areas than in those without lesions (Table 5).

Table 5.
Dynamics of DLQI scores by lesion localization.
Routinely assessed biomarkers showed a similar proportion of elevated values across all initial biological therapy classes, except for glucose metabolism at baseline (see GLY-serum glucose level, Table 6).

Table 6.
Distribution of patients with elevated biomarker levels by initial biological therapy class.
4. Discussion
The evaluated biological therapies were associated with significant improvements in disease severity and quality of life. Anti-TNF therapies were switched more frequently due to reduced efficacy, while clinical improvement was observed regardless of lesion localization.
4.1. Findings Interpretation
Patients treated with anti-IL-17 and anti-IL-23 in our cohort were older at diagnosis than those treated with anti-TNF (Table 1). Hypertension was the most common comorbidity among the patients with psoriasis, followed by diabetes mellitus (Table 1). Most of the patients with psoriatic arthritis received anti-IL-17 therapy (Table 1). Psoriasis is a chronic inflammatory disease that affects the body beyond the skin [34], and its management is multidisciplinary, with attention to comorbidities [35]. Biological therapies targeting key interleukins influence comorbidities and mortality risk, and improve quality of life and psychological burden [36,37,38]. Genetic and environmental factors can trigger or worsen the disease [15].
In our cohort, BMI was not significantly associated with baseline PASI or DLQI scores, in contrast to other studies that reported that higher BMI correlates with greater disease severity and lower quality-of-life scores [39,40]. Wang et al. [39] reported in a multicenter study of 1979 patients that each 1-point increase in BMI is associated with higher DLQI, PASI, and BSA scores [39]. Obesity and overweight are linked to increased disease severity, as reflected by higher PASI scores [40] and diminished efficacy of biological therapies [41]. Obesity is recognized as both a cause and a modifier of psoriatic diseases [42]. Baseline BMI did not show a significant association with PASI or DLQI in our cohort, potentially due to variable overweight and obesity prevalence and missing data, considering that we had data only for 61.7% of the evaluated cohort.
Cardiometabolic disorders were the most common comorbidities in the evaluated cohort (Table 1), consistent with a study conducted by Almenara-Blasco et al. [43]. Hypertension was the most common comorbidity observed in a Romanian study by Nicolescu et al. [5], in which infections ranked second, and dyslipidemia third [5]. Diabetes mellitus was the second most prevalent comorbidity in our cohort, compared to the third position reported by Boca et al. [6].
Topical therapy, which is considered safe [44], caused reactions in only two patients in our cohort. Phototherapy led to three acute events (pruritus and erythema). Conventional systemic therapy was associated with the highest number of adverse reactions. In the case of methotrexate, which was prescribed to the majority of patients, the most frequent adverse reactions were digestive, and hepatic cytolysis syndrome was the most frequent. West et al. ranked infections as the most common adverse reaction in patients treated with methotrexate in a meta-analysis [45]; this was not described in the cohort of this study.
Severity scores were comparable across treatment groups at baseline, while at the therapeutic switch, the anti-TNF group had higher PASI and DLQI scores than the anti-IL-17 group (Table 2 and Table 4). No therapeutic switch was required for patients treated with anti-IL-23 biologics. In our study, 29 treatment switches were recorded: 26 with anti-TNF and three with anti-IL-17 therapies, mainly due to loss or lack of efficacy. Anti-TNF-α was also the most frequently switched class in real-world data from Armstrong et al. [46]. The mean time to the first switch was 6 months, similar to the 6.5 months reported by Mease et al. [47], with inefficacy as the leading cause [47].
At 36 weeks, the anti-IL-17 and anti-IL-23 classes were associated with greater benefits than anti-TNF (Table 2). All patients who received anti-IL-23 therapy reached PASI 100 at 60 weeks of follow-up and PASI ≥ 50 at 36 weeks of follow-up (Table 2). In contrast, two patients with anti-IL-17 had a PASI < 50 at 60 weeks of follow-up. In the anti-TNF-α group, only 21.4% reached PASI 100 at 36 weeks, compared to 43.6% for anti-IL-17 and 54.2% for anti-IL-23. Two anti-TNF patients showed increased PASI scores at 36 weeks (Table 2, Figure 2 and Figure 3). The literature demonstrates that anti-IL-17 and anti-IL-23 biologics most effectively reduce PASI in biologically naïve patients at 3, 6, and 12 months [48], with Ixekizumab and Secukinumab providing superior 24-week responses [49]. Ntawuyamara et al. [49] reported no statistically significant differences in DLQI scores among biologics. In our study, the DLQI showed the best results at 36 and 60 weeks of follow-up in patients receiving anti-IL-23 biologics (Table 4). Despite the favorable therapeutic outcomes observed in the anti-IL-23 group, consistent with the literature, this group included the smallest number of patients compared with the other two groups.
Our results indicated that scalp lesions responded more favorably to anti-TNF and anti-IL-23 therapies (Table 3 and Table 5, Figure 5). It is well known that the most significant impact of biological therapies is on lesion localization, given their effects on functionality and quality of life [50]. Identifying a therapy that leads to the fastest and sustained improvement is essential [50]. Khan et al. [51] reported the greatest mean NAPSI improvement at week 24 with Brodalumab, followed by Etanercept and Ixekizumab [51]. The highest probability of complete nail clearance was achieved with Adalimumab (44%), followed by Ixekizumab (41%), and Brodalumab (31.6%) [51]. At the 52-week evaluation, the greatest mean NAPSI improvement was observed in patients who received Ixekizumab (83.3% of patients), followed by Brodalumab (83.1% of patients), and Adalimumab (70.2% of patients) [51]. The highest proportion of patients with NAPSI 0 was for those who received Brodalumab, followed by Adalimumab (58.8%), Ixekizumab (57.5%), and Ustekinumab (33.9%) [51]. For scalp psoriasis, PSSI 100 was achieved by week 16 with Ixekizumab (74.6% of patients) and Secukinumab (35% of patients), while PSSI75 was achieved at week 8 in >75% of patients on Ixekizumab and Infliximab [52]. Ustekinumab and Risankizumab led to 94% and 90% reductions in PSSI at the 12-week evaluation, and Adalimumab reduced PSSI by 77.2% after 16 weeks of treatment [52]. In our cohort, nail improvement was the greatest in patients who were treated with anti-IL-23 and anti-IL-17 (Figure 5).
Regarding the laboratory parameters, at baseline, most patients with higher glycemia received anti-IL-17 and anti-IL-23 therapies (Table 6) without any other impact on the evaluated biomarkers, regardless of the biologicals. Guselkumab is known to lower non-HDL cholesterol, whereas Secukinumab and Adalimumab affect glucose levels [49]. In our study, biologics did not significantly affect the lipid profile, glycemia, or renal function (Table 6), but the generalizability of the findings is limited due to the existence of missing data.
4.2. Strengths and Limitations of the Study
One of the strengths of this study is that the patients were recruited from two medical centers, reflecting prescribing patterns across multiple physicians regarding patients’ comorbidities at the initiation of biological therapy. The cohort included patients from both urban and rural areas in Central and Northern Romania, thus enhancing the representativeness of the results for the psoriasis population in Transylvania. Additionally, this study provides insight into real-world prescription patterns of biological therapies, an area with limited published data, particularly in Eastern Europe and Romania.
Although this study provides valuable insights, certain limitations must be acknowledged. The study design, based on routinely collected medical data, is limited by available data and associated quality, leading to potential biases, such as selection bias, measurement or instrument, misclassification, and admission rate bias. Consequently, missing and incomplete data reduced the statistical power of our study and the generalizability of the findings outside the evaluated cohort. Moreover, in the absence of control for confounding factors, considering that certain clinically relevant variables are not routinely documented, an adjusted analysis was not conducted. The small number of patients allowed only analysis by therapy class and not by individual drugs. Given the retrospective nature of the study and the fact that the subjects were selected from two medical centers, the exact timing of patient assessment may vary; therefore, patients may not be assessed exactly at 36 and 60 weeks of follow-up. Since biomarkers were evaluated in different laboratories, we reported nominal values rather than measured values, which decreased the power of the reported results. In addition, lesion localization was recorded only as present and not according to severity. Finally, Romanian national legislation and guidelines for biological treatments for psoriasis changed over time, which also affected access to biologics and the use of specific scoring systems for lesion localization [33,53,54,55,56]. Potential selection bias, center-specific factors, and attrition during follow-up may affect the generalizability of our findings.
5. Conclusions
In the evaluated cohort, patients with psoriasis who received biological therapy showed a favorable clinical course. The limited number of patients receiving anti-IL-23 therapy achieved the highest PASI 100 rates, with all patients reaching this by week 60 of follow-up. Anti-TNF was the most frequently switched treatment, primarily due to loss of efficacy, whereas no anti-IL-23 patients required a switch. Biological therapy had no significant impact on the lipid profile, blood glucose, or renal function.
Author Contributions
Conceptualization A.-L.L., S.D.B. and A.C.; methodology A.-L.L., S.D.B. and A.C.; software S.D.B.; validation A.-L.L., S.D.B. and A.C.; formal analysis A.-L.L., S.D.B. and A.C.; investigation A.-L.L.; resources A.-L.L., P.I., D.V.C., A.-L.B., C.-I.B., E.C., S.D. and I.S.; data curation A.-L.L.; writing—original draft preparation A.-L.L. and S.D.B.; writing—review and editing A.-L.L., P.I., D.V.C., A.-L.B., C.-I.B., E.C., S.D., I.S., S.D.B., A.C., O.H.O., A.-V.S.-T. and R.V.; visualization A.-L.L., P.I., D.V.C., A.-L.B., C.-I.B., E.C., S.D., I.S., S.D.B., A.C., O.H.O., A.-V.S.-T. and R.V.; supervision S.D.B. and A.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the following Ethics Committees: Infectious Diseases Clinical Hospital, Cluj-Napoca (approval number 20948/29.11.2024,date of approval: 29 November 2024), “Iuliu Hațieganu” University of Medicine and Pharmacy, Cluj-Napoca (approval number 55460/5.07.2023, date of approval: 5 July 2023), and County Emergency Hospital, Cluj-Napoca, Ethics Committee (approval 55460/27.11.2023, date of approval: 27 November 2023).
Informed Consent Statement
The Ethics Committee waived the requirement for informed consent because this study was retrospective, involved no direct patient contact, and used de-identified data.
Data Availability Statement
The dataset analyzed in the current study is part of an ongoing Ph.D. study and is available from the corresponding author upon reasonable request.
Acknowledgments
The authors acknowledge the use of ChatGPT (OpenAI, free version 4o) for assistance with editing and reducing the abstract to meet word-count requirements, and of Paperpal for English language review and linguistic refinement. The authors confirm that these tools were used solely for language and editorial support, and that all scientific content, data interpretation, and conclusions are those of the authors.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| PASI | Psoriasis Area Severity Index |
| DLQI | The Dermatology Life Quality Index |
| BSA | Body Surface Area |
| PSSI | Psoriasis Scalp Severity Index |
| NAPSI | Nail Psoriasis Severity Index |
| GPSS | Genital Psoriasis Symptoms Scale |
| TNFα-IL23-Th17 axis | Tumor Necrosis Factor alpha -Interleukin-23 (IL-23)-T helper 17 (Th17) cells axis |
| anti-TNF | Anti-Tumor Necrosis Factor alpha biological therapy |
| anti-IL-17 | Anti-Interleukin 17 biological therapy |
| anti-IL-23 | Anti-Interleukin 23 biological therapy |
| anti-IL-12/23 | Anti- interleukin 12/23 biological therapy |
| BMI | Body Mass Index |
| IMT | Intima-media thickness |
| MetS | Metabolic syndrome |
| PDE4i | Phosphodiesterase-4 inhibitor |
| MTX | Methotrexate |
References
- Boehncke, W.H.; Schönm, M.P. Psoriasis. Lancet 2015, 386, 983–994. [Google Scholar] [CrossRef]
- Skayem, C.; Taieb, C.; Halioua, B.; Baissac, C.; Saint Aroman, M. Epidemiology of Psoriasis: A Worldwide Global Study. Acta Derm. Venereol. 2025, 105, adv42945. [Google Scholar] [CrossRef]
- Parisi, R.; Iskandar, I.Y.K.; Kontopantelis, E.; Augustin, M.; Griffiths, C.E.M.; Ashcroft, D.M.; Global Psoriasis Atlas. National, regional, and worldwide epidemiology of psoriasis: Systematic analysis and modelling study. BMJ 2020, 369, m1590. [Google Scholar] [CrossRef] [PubMed]
- Bartosińska, J.; Szepietowski, J.C.; Raczkiewicz, D.; Griffiths, C.E.M.; Ashcroft, D.M.; Wright, A.K.; Podwójcic, K.; Turcza, J.; Maluchnik, M.; Chłoń-Domińczak, A.; et al. Epidemiology of Psoriasis in Poland: Prevalence, Incidence, and Mortality Rates. Int. J. Dermatol. 2025, 64, 1599–1606. [Google Scholar] [CrossRef] [PubMed]
- Nicolescu, A.C.; Bucur, Ș.; Giurcăneanu, C.; Gheucă-Solovăstru, L.; Constantin, T.; Furtunescu, F.; Ancuța, I.; Constantin, M.M. Prevalence and Characteristics of Psoriasis in Romania—First Study in Overall Population. J. Pers. Med. 2021, 11, 523. [Google Scholar] [CrossRef] [PubMed]
- Boca, A.N.; Ilies, R.F.; Vesa, S.; Pop, R.; Tataru, A.D.; Buzoianu, A.D. The first nation-wide study revealing epidemiologic data and life quality aspects of psoriasis in Romania. Exp. Ther. Med. 2019, 18, 900–904. [Google Scholar] [CrossRef]
- Parisi, R.; Symmons, D.P.; Griffiths, C.E.; Ashcroft, D.M.; IMPACT Project Team. Global epidemiology of psoriasis: A systematic review of incidence and prevalence. J. Investig. Dermatol. 2013, 133, 377–385. [Google Scholar] [CrossRef]
- Sarac, G.; Koca, T.T.; Baglan, T. A brief summary of clinical types of psoriasis. North Clin. Istanb. 2016, 3, 79–82. [Google Scholar] [CrossRef]
- Kimmel, G.W.; Lebwohl, M. Psoriasis: Overview and Diagnosis. In Evidence-Based Psoriasis; Bhutani, T., Liao, W., Nakamura, M., Eds.; Springer: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Wozel, G.; Klein, E.; Mrowietz, U.; Reich, K.; Sebastian, M.; Streit, V. Scalp psoriasis. J. Dtsch. Dermatol. Ges. 2011, 9, 70–74, Erratum in J. Dtsch. Dermatol. Ges. 2011, 9, 872. [Google Scholar] [CrossRef]
- Rich, P.; Scher, R.K. Nail Psoriasis Severity Index: A useful tool for evaluation of nail psoriasis. J. Am. Acad. Dermatol. 2003, 49, 206–212. [Google Scholar] [CrossRef]
- Gottlieb, A.B.; Kirby, B.; Ryan, C.; Naegeli, A.N.; Burge, R.; Potts Bleakman, A.; Anatchkova, M.D.; Yosipovitch, G. The Development of a Patient-Reported Outcome Measure for Assessment of Genital Psoriasis Symptoms: The Genital Psoriasis Symptoms Scale (GPSS). Dermatol. Ther. 2018, 8, 45–56. [Google Scholar] [CrossRef]
- Liluashvili, S.; Kituashvili, T. Dermatology Life Quality Index and disease coping strategies in psoriasis patients. Postep. Dermatol. Alergol. 2019, 36, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Nast, A.; Smith, C.; Spuls, P.I.; Avila Valle, G.; Bata-Csörgö, Z.; Boonen, H.; De Jong, E.; Garcia-Doval, I.; Gisondi, P.; Kaur-Knudsen, D.; et al. EuroGuiDerm Guideline on the systemic treatment of Psoriasis vulgaris—Part 1. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 2461–2498. [Google Scholar] [CrossRef]
- Sugumaran, D.; Yong, A.C.H.; Stanslas, J. Advances in psoriasis research: From pathogenesis to therapeutics. Life Sci. 2024, 355, 122991. [Google Scholar] [CrossRef]
- Armstrong, A.W.; Blauvelt, A.; Callis Duffin, K.; Huang, H.Y.; Savage, J.L.; Guo, L.; Merola, J.F. Psoriasis. Nat. Rev. Dis. Primers 2025, 11, 45. [Google Scholar] [CrossRef]
- Dand, N.; Stuart, P.E.; Bowes, J.; Ellinghaus, D.; Nititham, J.; Saklatvala, J.R.; Teder-Laving, M.; Thomas, L.F.; Traks, T.; Uebe, S.; et al. GWAS meta-analysis of psoriasis identifies new susceptibility alleles impacting disease mechanisms and therapeutic targets. Nat. Commun. 2025, 16, 2051. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; He, M.; Jiang, J.; Duan, X.; Chai, B.; Zhang, J.; Tao, Q.; Chen, H. Triggers for the onset and recurrence of psoriasis: A review and update. Cell Commun. Signal. 2024, 22, 108. [Google Scholar] [CrossRef] [PubMed]
- Rendon, A.; Schäkel, K. Psoriasis Pathogenesis and Treatment. Int. J. Mol. Sci. 2019, 20, 1475. [Google Scholar] [CrossRef]
- Ten Bergen, L.L.; Petrovic, A.; Krogh Aarebrot, A.; Appel, S. The TNF/IL-23/IL-17 axis—Head-to-head trials comparing different biologics in psoriasis treatment. Scand. J. Immunol. 2020, 92, e12946. [Google Scholar] [CrossRef]
- Brownstone, N.D.; Hong, J.; Mosca, M.; Hadeler, E.; Liao, W.; Bhutani, T.; Koo, J. Biologic Treatments of Psoriasis: An Update for the Clinician. Biologics 2021, 15, 39–51. [Google Scholar] [CrossRef]
- Floris, A.; Mugheddu, C.; Sichi, L.; Anedda, J.; Frau, A.; Sorgia, J.; Li Volsi, L.; Paladino, M.T.; Congia, M.; Chessa, E.; et al. Treatment of psoriasis with different classes of biologics reduces the likelihood of peripheral and axial psoriatic arthritis development. Rheumatology 2025, 64, 1131–1137. [Google Scholar] [CrossRef]
- Amara, S.; Pasumarthi, A.; Parikh, N.; Kodali, N.; Lebwohl, M.; Monks, G. Psoriasis management tree based on comorbidity. Int. J. Dermatol. 2025, 64, 229–245. [Google Scholar] [CrossRef]
- Osigwe, P.C.; Agomoh, C.E.; Osigwe, I.S.; Akumiah, F.K. The Association Between Psoriasis and Atherosclerotic Cardiovascular Disease: A Systematic Review and Meta-Analysis of Observational Studies. Cureus 2024, 16, e63379. [Google Scholar] [CrossRef]
- Dattola, A.; Manenti, G.; Ferrari, D.; Vollono, L.; Marsico, S.; Lamacchia, F.; Esposito, M.; Marchesano, M.; Zangrilli, A.; Floris, R.; et al. Prevalence of Atherosclerosis in Psoriatic Patients Detected with Epiaortic Color Doppler Ultrasound and Computed Tomography Angiography. Dermatol. Pract. Concept. 2022, 12, e2022011. [Google Scholar] [CrossRef]
- Liu, C.; Chen, H.; Liu, Y.; Huang, H.; Yu, W.; Du, T.; Long, X.; Chen, X.; Chen, Z.; Guo, S.; et al. Immunity: Psoriasis comorbid with atherosclerosis. Front. Immunol. 2022, 13, 1070750. [Google Scholar] [CrossRef]
- Su, W.; Zhao, Y.; Wei, Y.; Zhang, X.; Ji, J.; Yang, S. Exploring the Pathogenesis of Psoriasis Complicated with Atherosclerosis via Microarray Data Analysis. Front. Immunol. 2021, 12, 667690. [Google Scholar] [CrossRef]
- Dascălu, R.C.; Bărbulescu, A.L.; Dinescu, Ș.C.; Biță, C.E.; Stoica, L.E.; Sandu, R.E.; Vreju, F.A. Metabolic Syndrome in Psoriasis Patients—An Observational Study. Curr. Health Sci. J. 2024, 50, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Bucur, S.; Serban, E.D.; Ileanu, B.V.; Costache, R.S.; Nicolescu, A.C.; Constantin, T.; Costache, D.O.; Constantin, M.M. Effectiveness and Drug Survival of Ixekizumab and Secukinumab in Patients with Moderate to Severe Plaque Psoriasis. Psoriasis 2024, 14, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Mihu, C.; Popescu, C.A.; Neag, M.A.; Bocșan, I.C.; Melincovici, C.S.; Baican, A.L.; Năsui, B.A.; Buzoianu, A.D. The Psoriasis Disability Index in Romanian Psoriasis Patients during COVID-19 Pandemic. J. Clin. Med. 2023, 12, 6000. [Google Scholar] [CrossRef]
- Decean, L.; Badea, M.; Ilies, R.; Sasu, A.; Rus, V.; Mihai, A. Psoriasis-Related Stigma: Is There More to Uncover? J. Interdiscip. Med. 2022, 7, 56–59. [Google Scholar] [CrossRef]
- Burlacu, G.; Virag-Iorga, C.; Radu, M.O.; Giurcaneanu, C. Strategies to improve quality of life in psoriasis. DermatoVenerologia 2025, 70, 7–12. [Google Scholar]
- COD PROTOCOL DENUMIRE [Internet]. [Cited 18 January 2026]. Available online: https://cnas.ro/wp-content/uploads/2025/06/lista-protocoalelor-terapeutice-iunie-2025-site_all.pdf (accessed on 18 January 2026).
- Raharja, A.; Mahil, S.K.; Barker, J.N. Psoriasis: A brief overview. Clin. Med. 2021, 21, 170–173. [Google Scholar] [CrossRef]
- Daugaard, C.; Iversen, L.; Hjuler, K.F. Comorbidity in Adult Psoriasis: Considerations for the Clinician. Psoriasis 2022, 12, 139–150. [Google Scholar] [CrossRef]
- Cai, J.; Cui, L.; Wang, Y.; Li, Y.; Zhang, X.; Shi, Y. Cardiometabolic Comorbidities in Patients with Psoriasis. Front. Pharmacol. 2021, 12, 774808. [Google Scholar] [CrossRef]
- de Ruiter, C.C.; Rustemeyer, T. Biologics Can Significantly Improve Dermatology Life Quality Index in Psoriatic Patients. Psoriasis 2022, 12, 99–112. [Google Scholar] [CrossRef]
- Timis, T.L.; Beni, L.; Mocan, T.; Florian, I.A.; Orasan, R.I. Biologic Therapies Decrease Disease Severity and Improve Depression and Anxiety Symptoms in Psoriasis Patients. Life 2023, 13, 1219. [Google Scholar] [CrossRef]
- Wang, H.; Hou, S.; Kang, X.; Yu, C.; Yang, B.; Shi, Y.; Li, F.; Li, W.; Gu, J.; Lei, M.; et al. BMI matters: Understanding the link between weight and severe psoriasis. Sci. Rep. 2025, 15, 11158. [Google Scholar] [CrossRef]
- Li, L.; Liu, K.; Duan, X.; Xu, L.; Yang, Q.; Liu, F. A Comparison of Clinical Characteristics in Overweight/Obese and Normal Weight Patients with Psoriasis Vulgaris. Clin. Cosmet. Investig. Dermatol. 2023, 16, 1377–1385. [Google Scholar] [CrossRef]
- Bardazzi, F.; Balestri, R.; Baldi, E.; Antonucci, A.; De Tommaso, S.; Patrizi, A. Correlation between BMI and PASI in patients with moderate to severe psoriasis undergoing biological therapy. Dermatol. Ther. 2010, 23, S14–S19. [Google Scholar] [CrossRef]
- Haberman, R.H.; Ogdie, A.; Merola, J.F.; Scher, J.U.; Eder, L. The Obesity-Inflammation Axis in Psoriatic Disease: Mechanisms and Therapeutic Strategies. Nat. Rev. Rheumatol. 2025; in press. [Google Scholar] [CrossRef]
- Almenara-Blasco, M.; Gracia-Cazaña, T.; Poblador-Plou, B.; Laguna-Berna, C.; Carmona-Pírez, J.; Navarro-Bielsa, A.; Prados-Torres, A.; Gimeno-Miguel, A.; Gilaberte, Y. Multimorbidity of Psoriasis: A Large-Scale Population Study of Its Associated Comorbidities. J. Clin. Med. 2024, 13, 492. [Google Scholar] [CrossRef]
- Bruner, C.R.; Feldman, S.R.; Ventrapragada, M.; Fleischer, A.B., Jr. A systematic review of adverse effects associated with topical treatments for psoriasis. Dermatol. Online J. 2003, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Ogston, S.; Foerster, J. Safety and Efficacy of Methotrexate in Psoriasis: A Meta-Analysis of Published Trials. PLoS ONE 2016, 11, e0153740, Erratum in: PLoS ONE 2016, 11, e0158928. https://doi.org/10.1371/journal.pone.0158928. [Google Scholar] [CrossRef]
- Armstrong, A.W.; Patel, M.; Li, C.; Garg, V.; Mandava, M.R.; Wu, J.J. Real-world switching patterns and associated characteristics in patients with psoriasis treated with biologics in the United States. J. Dermatolog. Treat. 2023, 34, 2200870. [Google Scholar] [CrossRef]
- Mease, P.J.; Blauvelt, A.; Sima, A.P.; Beaty, S.W.; Low, R.; Gomez, B.; Gurrola, M.; Lebwohl, M.G. Impact of Disease Factors of Patients with Psoriasis and Psoriatic Arthritis on Biologic Therapy Switching. Dermatol. Ther. 2024, 14, 2805–2825. [Google Scholar] [CrossRef]
- Graier, T.; Salmhofer, W.; Jonak, C.; Weger, W.; Zikeli, C.; Gruber, B.; Sator, P.; Prillinger, K.; Mlynek, A.; Schütz-Bergmayr, M.; et al. Evolution of characteristics and biologic treatment effectiveness in patients of the Austrian psoriasis registry from 2004–2022. J. Dtsch. Dermatol. Ges. 2023, 21, 1513–1523. [Google Scholar] [CrossRef] [PubMed]
- Ntawuyamara, E.; Deng, B.; Liang, Y. Cutaneous and systemic improvements in psoriasis patients after different biologic treatments. Sci. Rep. 2025, 15, 15528. [Google Scholar] [CrossRef]
- Blauvelt, A.; Gondo, G.C.; Bell, S.; Echeverría, C.; Schmitt-Egenolf, M.; Skov, L.; van de Kerkhof, P.; McCormick Howard, L.; Strober, B. Psoriasis Involving Special Areas Is Associated with Worse Quality of Life. J. Psoriasis Psoriatic Arthritis 2023, 8, 100–106. [Google Scholar] [CrossRef]
- Khan, M.; Wallace, C.E.; Ahmed, F.; Rahman, S.M.; Memon, N.; Haque, A. Assessing Comparative Efficacy of Biologics for the Treatment of Psoriasis with Nail Involvement. J. Psoriasis Psoriatic Arthritis 2024, 9, 61–68. [Google Scholar] [CrossRef]
- Papadimitriou, I.; Bakirtzi, K.; Katoulis, A.; Ioannides, D. Scalp Psoriasis and Biologic Agents: A Review. Skin Appendage Disord. 2021, 7, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Ordin 1218/16.09.2010. Portal Legislativ. Available online: https://legislatie.just.ro/public/DetaliiDocument/123112 (accessed on 30 November 2025).
- Eli Lilly. România Anunță Lansarea TALTZ în Tratamentul Psoriazisului Moderat-Sever. MedicalManager 2018. Available online: https://www.medicalmanager.ro/eli-lilly-romania-anunta-lansarea-taltz-in-tratamentul-psoriazis-ului-moderat-sever/ (accessed on 30 November 2025).
- Anexa 28/10/2022. Portal Legislativ. Available online: https://legislatie.just.ro/Public/DetaliiDocumentAfis/261213 (accessed on 30 November 2025).
- Casa Națională de Asigurări de Sănătate (CNAS). Protocol List. Available online: https://cnas.ro/wp-content/uploads/2022/04/Binder1-3.pdf (accessed on 30 November 2025).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.