Abstract
Monkeypox (Mpox) disease, caused by the Monkeypox virus (Mpox virus), emerged as a significant global health threat during the 2022 outbreak, prompting the World Health Organisation (WHO) to declare it a Public Health Emergency of International Concern (PHEIC). Rapid evolution through genomic modifications enhanced its outbreak potential. Zoonotic transmission occurs through close contact with infected rodents or primates; human-to-human transmission occurs via close contact or homosexual intercourse. The virus disseminates via the lymphatic system, causing symptoms ranging from mild skin lesions to severe multi-system complications or even death. Diagnosis incorporates clinical symptoms as well as advanced molecular and immunological methods. Currently, no specific antiviral medications or vaccines are available for Mpox, necessitating reliance on conventional therapeutic supports and treatments developed for smallpox. Raising awareness, promoting protective practices, implementing surveillance, enabling rapid diagnosis, ensuring timely treatment, and promoting mass vaccination are crucial to curb Mpox transmission. This narrative review provides a comprehensive overview of the current knowledge on epidemiology, evolution, transmission, pathogenesis, clinical signs, diagnosis, treatment, vaccination, and prevention strategies for Mpox.
Keywords:
epidemiology; transmission; diagnosis; vaccine; monkeypox; mpox; evolution; pathogenesis; treatment; prevention 1. Introduction
The Monkeypox virus (Mpox virus), belonging to the Orthopoxvirus genus and Poxviridae family, causes a zoonotic illness that can be transmitted from animals to humans and from person to person. This disease is characterised by the appearance of a pustular rash [1] and exhibits signs similar to those of smallpox [2]. The virus was first identified in Denmark in 1958, with the first human case reported in the Democratic Republic of Congo in 1970. Initially confined to remote regions of Africa, the Mpox virus first appeared in the United States during an outbreak in 2003. Concerns have emerged regarding evolving epidemiological trends and global health threats, as Mpox cases increased significantly from 2018 to 2024 [3,4]. According to a report published by the World Health Organisation (WHO) on 29 April 2025, 137,892 Mpox cases have been confirmed globally between 1 January 2022, and 31 March 2025, with 317 deaths across 132 countries [4]. Notably, on 14 August 2024, the WHO classified the Mpox virus, a zoonotic pathogen, as a public health emergency of international concern (PHEIC) [5].
The distribution of Mpox cases has been predominantly observed in Africa from 2022 to 2025, with the Democratic Republic of the Congo (DRC) being significantly impacted by the Ia and Ib clades of the virus. Conversely, Clade IIb has been primarily concentrated in Sierra Leone. The WHO assessed the public health risk as high for clade Ib and moderate for other clades, underscoring the need for continued surveillance and response measures [6,7]. Mpox infection is heightened in rural, wooded areas of Central and Western Africa, particularly among individuals who handle bush meat, caregivers of infected persons, unvaccinated individuals, and men who hunt and interact with wild animals [8]. Infected individuals typically display distinct symptoms, which include fever, multiple papular lesions, vesiculopustular lesions, and ulcerative lesions on the body and face, accompanied by lymphadenopathy [7,9,10,11,12,13]. Upon entry, the Mpox virus replicates primarily within immune cells and disseminates to regional lymph nodes. After lymphatic spread, the virus infiltrates various tissues throughout the body [14].
Symptoms typically onset between 7 and 14 days post-infection, with prodromal signs including headaches, fever, and chills that may appear 1 to 2 days before the onset of a facial rash. In more severe cases, the infection can lead to critical complications such as pneumonia, encephalitis, or septicemia, which may result in fatal outcomes. The infected individuals remain contagious throughout this period [15,16]. The clinical features of Mpox are similar to those of smallpox; however, certain symptoms help distinguish the two diseases. Notably, the presence of lymphadenopathy, or swollen lymph nodes, is a defining characteristic of Mpox that is absent in smallpox [17]. Mpox infections are usually self-limiting, and milder cases can be managed with supportive care [18]. People with weakened immune systems, aged less than 8 years, pregnant women, or those with an immunocompromised state or damaged skin are at greater risk of severe Mpox. Currently, no specific treatment exists for Mpox. Various antiviral medications and vaccines originally designed and approved for smallpox or other orthopoxviruses have been used in the treatment of Mpox [18].
The potential for asymptomatic transmission of the Mpox virus is a major concern, especially since the routine smallpox vaccination program ended in 1980, which may have altered the virus’s structure. Changes in human behaviour and movement contributed to the 2022 outbreaks, yet limited research exists on their epidemiology, clinical features, and transmission, unlike previous rare outbreaks [2,7,19]. The WHO (2022) noted cases without travel or contact from endemic areas, indicating unknown routes and a pandemic risk [20]. Additionally, a thorough investigation has shown that the fatality rate for Mpox cases ranges from 1% to 11% [21].
Currently, there are few up-to-date, Mpox-specific vaccines or treatments available, highlighting the need for immunity-boosting vaccines and better professional knowledge, as confidence in diagnosis and management remains low [22,23]. Increased awareness allows for quick diagnosis, contact tracing, isolation, and outbreak control, even though diverse symptoms can delay testing and increase patient anxiety [24]. This highlights the urgent need for educational initiatives and training to prevent and effectively manage the current outbreak [18]. The surge in cases has prompted a greater focus on investigating the clinical features and transmission of the outbreak to attain a clearer understanding of the situation.
This narrative review aims to present an in-depth overview of the genomic characteristics, evolution, and current epidemiological landscape of the Mpox outbreak, and to explore various signs and symptoms associated with the infection, as well as the modes of transmission and the underlying mechanisms of its pathogenesis. Additionally, the review will discuss the major diagnostic methods available, covering both the conventional approaches and innovative detection techniques. A comprehensive analysis of treatment options will be included, emphasising both traditional approaches and newer antiviral medications. Finally, the review will address preventive strategies, including vaccines from diverse platforms, designed to combat the spread of Mpox.
2. Methodology
To gather relevant data for this review, articles were retrieved from three major databases: Google Scholar, PubMed, and ScienceDirect. Specific search keywords such as “genome”, “evolution”, “epidemiology,” “signs,” “symptoms,” “complication”, “transmission,” “pathogenesis,” “diagnosis,” “detection,” “treatment,” “antiviral,” “drug,” “vaccine,” and “prevention” were combined with “Mpox” and “mpox” using appropriate Boolean operators, i.e., AND and OR. Additionally, “Title and abstract” and “Title, abstract, or author-specified keywords” filters were employed in the advanced search options on PubMed and ScienceDirect, respectively, while the term “allintitle” was used prior to the specific search keywords during searches in Google Scholar. Additional filters, such as “Full text,” were applied in PubMed and ScienceDirect, respectively. Relevant data were also gathered from articles available on ResearchGate, as well as from various recommendations, guidelines, and reports published by the World Health Organisation (WHO) and the Centres for Disease Control and Prevention (CDC) (Figure 1).
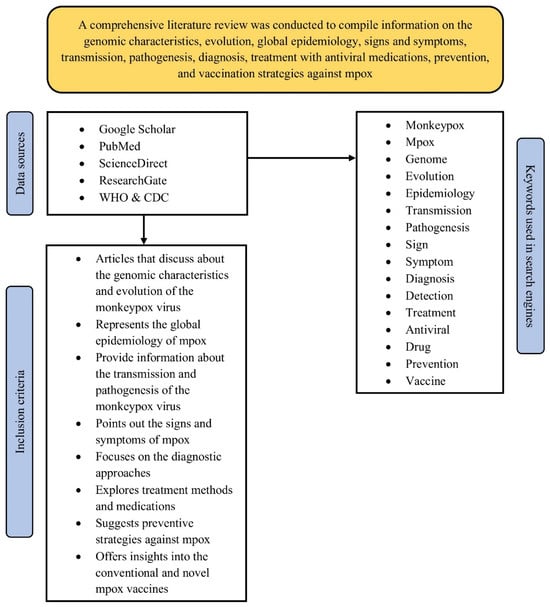
Figure 1.
Schematic flowchart presenting the literature search strategy of the review.
3. Review of Literature
3.1. Evolution of Mpox Virus and Global Distribution
Various genomic modifications, including variations in mutation types and rates, genetic recombination, and the loss of specific genes, have contributed to Mpox virus evolution and changes in viral host specificity. One significant driver of Mpox virus evolution is the increased frequency of G>A/C>T substitutions, which can be attributed to the action of the host apolipoprotein B mRNA editing enzyme, catalytic subunit 3 (APOBEC3) cytidine deaminase. This activity generates mutations that enhance adaptability while potentially reducing viral fitness. Notably, APOBEC3 is responsible for 87% of the alterations observed in strains from the 2022 outbreak, particularly in genes that regulate host interactions. Furthermore, evidence suggests that undetected transmission has occurred since 2016, and this mutational bias underscores the virus’s prolonged circulation within human populations [25,26].
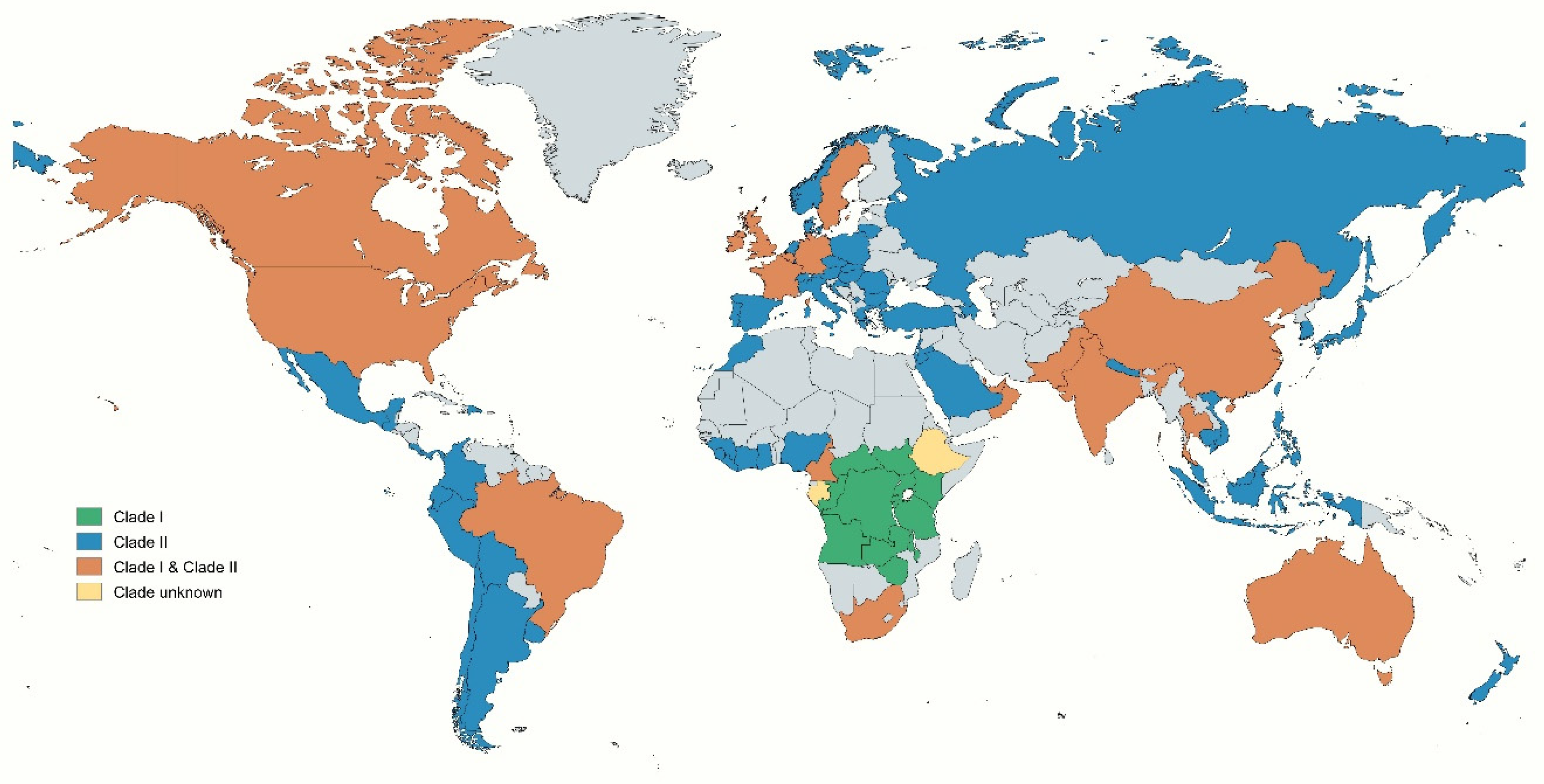
Phylogenomic analysis reveals that clade I of the Mpox virus, particularly in South Kivu, exhibits greater genetic diversity and is evolving towards enhanced human-to-human transmission through APOBEC3-type mutations. These mutations not only promote viral replication but also enable the virus to evade the host’s immune system. In contrast, clade II b, linked to the 2022–2023 outbreak, demonstrates more stable genetic variation and a slower evolutionary rate [27]. The Mpox virus is classified into two clades: clade I, known as the Central African/Congo Basin clade, and clade II, the West African clade, which is further divided into IIa and IIb. Clade IIb exhibits a lower mortality rate of less than 1% but demonstrates more efficient dissemination than clade I, which has a mortality rate of 10–15% [28,29]. Clade I of the Mpox virus has been identified in several Central African nations, including the Democratic Republic of Congo, the Central African Republic, Kenya, Uganda, Rwanda, Malawi, and Tanzania, among others. In contrast, Clade II is prevalent across various countries on multiple continents, including Asia, Europe, Africa, and the Americas. Both clade I and clade II Mpox viruses have been detected in a range of countries, such as the USA, Canada, Brazil, France, Germany, Switzerland, the UK, Ireland, China, Pakistan, India, Thailand, South Africa, Oman, and Australia, among others [30] (Figure 2).
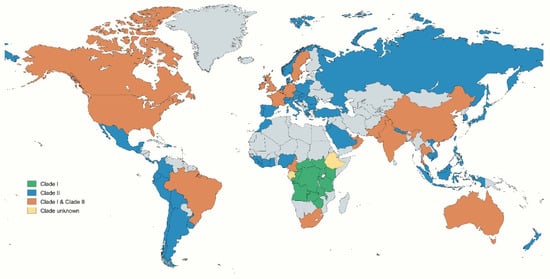
Figure 2.
The world map illustrates the global distribution of different clades of the Mpox virus. Distinct clades are represented by individual colours, while grey-shaded regions indicate areas with no reported cases.
Since 2024, the majority of Mpox cases have continued to be reported from the African continent, largely driven by outbreaks of Mpox virus Ib in East African countries, including the DRC, where clade Ia is co-circulating. Sierra Leone, however, is experiencing a rapidly evolving outbreak that, based on available genomic sequencing results, appears to be driven by the Mpox virus clade IIb. Outside the African region, most cases reflect ongoing circulation of Mpox clade IIb among men who have sex with men (MSM) [6,31].
A significant factor influencing the evolution of the Mpox virus is the recombination between different strains of Mpox and other orthopoxviruses, such as the vaccinia virus. This recombination may enhance the Mpox virus’s ability to evade immune responses or adapt to new hosts. Additionally, compromises between human adaptation and the retention of zoonotic capabilities are evident in gene loss or pseudogenization, as seen with the D10L gene in Clade IIb [28,32].
Although Mpox is a DNA virus with a relatively stable genome, prolonged human transmission has led to a surge in its substitution rate in Clade IIb, up to 6–12 times that of strains before 2018. This acceleration underscores the importance of human reservoirs in viral development, marking a departure from historical trends [32,33].
3.2. Genomic Characteristics of Mpox Virus
The genome of the Mpox virus consists of approximately 197 kilobases of linear, double-stranded DNA, characterised by covalently closed hairpin ends and inverted terminal repeats (ITRs) of approximately 10 kilobases at both termini. It encodes a total of 181 proteins. The central conserved region contains housekeeping genes crucial for replication, transcription, and virion assembly, while genes at the ends are variable and play roles in the virus’s pathogenesis and host range [34]. The central region remains relatively conserved across Mpox virus strains and shows over 90% identity with analogous genes in other orthopoxviruses [31,32]. In contrast, the more variable terminal regions include genes linked to pathogenesis and host range, which facilitate modulation of the immune response and the development of species-specific characteristics, and show significant variation among different strains of Mpox virus [35]. Within the ITR region of the Mpox virus, there are at least four open reading frames (ORFs) [36], which may contribute to maintaining genome stability and influencing evolutionary processes [35].
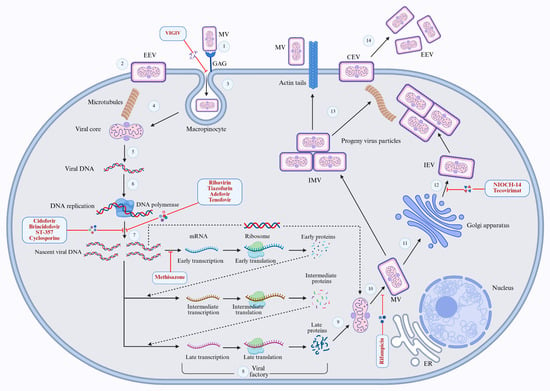
The central genomic regions of the Mpox virus genome contain the open reading frames (ORFs) A25R and C10L, which exhibit a 96.3% similarity to the corresponding regions in the smallpox virus [37,38]. The Mpox virus genome encodes all the proteins necessary for DNA replication, including the helicase-primase E5, which is vital for genome uncoating and DNA replication [39]. The F8–A22–E4 polymerase holoenzyme is crucial for the accurate replication of the viral genome. Unlike other DNA polymerases, this holoenzyme incorporates a multifunctional cofactor, A22–E4, which is involved in processivity, proofreading, and repair during replication [40]. After replication occurs in the cytoplasm, the virally encoded RNA polymerase generates two distinct infectious forms i.e., the extracellular enveloped virus (EEV), which exits the cell through exocytosis and possesses a lipid envelope derived from the Golgi complex or endosomes, and the intracellular mature virus (IMV), which is released upon cell lysis and features a robust lipoprotein membrane that facilitates its spread between animals [41].
The Mpox virus genome encodes a variety of proteins critical to viral pathogenesis [42]. The conserved central genomic region encodes structural proteins that appear in various forms within virions. The enveloped extracellular virus (EEV) form generates membrane proteins such as C19L, A35R, and B6R, while the intracellular mature virus (IMV) form produces proteins including A29, M1R, E8L, H3L, and L1R. Among these, proteins such as A35 are vital for virulence, whereas L1R plays a key role in facilitating viral entry, highlighting their roles in cellular invasion, viral transmission, and pathogenicity [42]. The A29L protein, a homolog of the vaccinia virus A27L protein, is crucial for the virus’s attachment to host cell membranes. Consequently, A29L is considered a promising target for diagnostics and a potential vaccine candidate, capable of eliciting neutralising antibodies and robust immune responses [43]. The A35R gene encodes the A35R protein, which is crucial for the virus’s pathogenicity; its absence results in significant attenuation [42]. In a study examining eight different host-derived cell lines, it was determined that while the A35 protein is not necessary for viral replication, its removal from the viral genome resulted in a 1000-fold reduction in virulence in a mouse model [44].
3.3. Global Epidemiology of Mpox
The discovery of Mpox infection dates back to the summer and fall of 1958, marked by two consecutive outbreaks of a skin disease resembling smallpox among captive cynomolgus monkeys used for polio vaccine research at Statens Serum Institut in Copenhagen, Denmark [45]. This was followed by similar occurrences at research institutes in The Netherlands, the United States, and France [46,47]. According to reports from the WHO, several countries in the rainforests of West and Central Africa, particularly within the Congo Basin, are recognised as endemic areas for Mpox. Here, the zoonotic transmission of the Mpox virus occurs among small rodents and various primates [20,48,49]. Over time, facilitated by advancements in global transport and trade, this infection has spread beyond African borders, leading to sporadic outbreaks and epidemics that pose a significant threat to healthcare systems and humanity.
3.3.1. Epidemiology of Mpox Within the African Continent
The first reported case of Mpox transmitted from animals to humans occurred in a nine-month-old child in the Democratic Republic of the Congo (DRC) in 1970. The child was admitted to a hospital, displaying centrifugal rashes and clinical symptoms resembling those of smallpox [50]. Shortly after this report and the subsequent virological confirmation by the WHO, cases of Mpox were diagnosed both virologically and serologically in various countries within the African Congo basin and West Africa, including Liberia, Nigeria, and Sierra Leone, between 1970 and 1971 [17,51]. The infected populations were predominantly children who had close contact with wild primates or monkeys, which are believed to be the primary intermediate hosts for Mpox virus transmission [52]. The cessation of the global smallpox vaccination campaign, following the complete eradication of smallpox, led to a sudden increase in Mpox incidences, with 47 reported cases in Central and West Africa from 1970 to 1979 [53,54]. Notably, 38 of these cases occurred in the DRC, and 12 additional cases were reported in the DRC and other Central and West African countries between 1980 and 1981. Of those infected, 80% were unvaccinated children under the age of 10, with eight deaths (17%) reported among children aged between 7 months and 7 years [17,55]. In response to the rise in Mpox cases, an active surveillance program was initiated in the DRC to monitor the spread of the virus, ensure accurate diagnosis, and implement preventive measures. This effort led to identifying a significant number of cases-specifically, 386 new Mpox infections in the DRC alone, compared to just 18 cases in other Central and West African countries, with a notably higher proportion of infected children than adults [17,54,56,57]. However, in 1986, Mpox surveillance programs in the DRC were discontinued due to the widespread HIV epidemic across Africa, which led the WHO to shift its resources away from Mpox [58]. As a result, routine Mpox diagnosis was discontinued, leading to a decline in reported cases, ultimately falling to zero by the 1980s. Reported Mpox cases remained very low, nearly zero, in the DRC and other countries in Central and West Africa until 1996, when a cluster of 344 new cases was identified among an unvaccinated population in the DRC between 1996 and 1997 [59]. This resurgence brought global attention to Mpox in Africa, with cases eventually rising to 500 by 1999. Between 2001 and 2004, 136 suspected cases were reported in the DRC, of which 51 were later confirmed as Mpox-positive. Following that period, thousands of new cases were reported annually in the DRC until 2005 [60]. Other countries in West and Central Africa reported infrequent Mpox cases, likely due to a decline in the disease diagnosis [52]. Between 2010 and 2018, several African countries experienced outbreaks of Mpox, including the Democratic Republic of Congo, Cameroon, Liberia, the Central African Republic, Sierra Leone, the Ivory Coast, and Nigeria. In 2017, Nigeria reported an outbreak with 122 confirmed cases of Mpox across 17 states within a year, resulting in six confirmed deaths [61,62,63]. The incidence of Mpox fluctuated in endemic regions across the continent, with sporadic outbreaks attributed to irregular Mpox diagnoses, weakened healthcare facilities, other infectious diseases, and inadequate surveillance systems.
3.3.2. Epidemiology of Mpox Outside of the African Continent
The first known case of Mpox outside the endemic region of Africa was identified in May 2003 in a 3-year-old girl from Wisconsin, USA, who was hospitalised due to cellulitis in her hand resulting from a bite by her pet prairie dog [64]. Subsequently, additional laboratory-confirmed cases of Mpox were reported in several states, including Illinois, Missouri, Ohio, and Indiana. These cases were suspected to have arisen from scratches or bites from pet prairie dogs imported along with Mpox-infected Gambian rats from Ghana, West Africa [65,66]. In total, 47 laboratory-diagnosed or suspected cases were reported that year across various states in the USA. Fortunately, no deaths occurred, and there was no evidence of direct human-to-human transmission [67]. Between 2018 and 2021, both virologically confirmed and suspected cases of Mpox were reported in the United States, the United Kingdom, Israel, and Singapore [68,69,70,71]. This spread was largely attributed to travellers returning from Africa, especially Nigeria, where an ongoing outbreak was ongoing [72]. The infected individuals received prompt medical attention, which halted the spread of Mpox [60]. However, Mpox transmission did not end there, leading to an alarming global outbreak in 2022.
3.3.3. Global Outbreak of Mpox in 2022–2025
In 2022, the world experienced a global outbreak of Mpox, first highlighted by the UK Health Security Agency and the WHO after a human Mpox infection was diagnosed in a traveller returning to the UK from Nigeria in May [73]. Shortly thereafter, Italy and Portugal also confirmed their first cases of Mpox in May [74]. By the end of May 2022, the UK had reported 86 laboratory-confirmed cases linked to the initial case [75]. Mpox spreads from person to person through direct contact, with new cases continuing to emerge across the UK, various European countries, and beyond. By the first week of October 2022, Mpox had been reported in approximately 100 non-endemic countries, according to the CDC, with outbreaks occurring in a range of nations across Europe, Asia, Australia, North America, and South America, as well as in various countries on the African continent where the disease is endemic [52,76]. Public health experts initially believed that direct person-to-person transmission through close proximity was the primary mode of transmission. Events such as popular festivals and saunas were considered significant sources of Mpox transmission, particularly in countries such as Germany and France [52,77]. However, subsequent research from Italy, Spain, and Germany revealed multiple Mpox cases where Mpox DNA was identified in human semen samples. This led to the conclusion that homosexual activities among men were a major route of transmission during the 2022 mass outbreak [78,79,80].
According to a 2023 report by the WHO, 96.6% of the reported cases were male, with 84.4% of these individuals participating in homosexual intercourse, predominantly aged between 18 and 44 years, indicating sexual transmission as a major route during the 2022 outbreak [81]. From 1 January 2022, to 30 September 2024, a total of 109,699 laboratory-confirmed Mpox cases and 236 deaths have been reported to the WHO from its 123 member states across all six regions. The top ten countries with the highest reported cases globally since the onset of the Mpox outbreaks in 2022 are as follows: the USA (34,063 cases), Brazil (12,722), Spain (8282), the Democratic Republic of the Congo (7095), France (4334), Colombia (4269), Mexico (4177), the UK (4100), Peru (3948), and Germany (3932). African countries, being the endemic region and a focal point for the Mpox outbreak, are facing a significantly worse situation than the rest of the world, with 11,724 confirmed cases and 57 deaths reported from 1 January 2022, to 20 October 2024, across 22 WHO member states in Africa [81].
As of 12 October 2025, a total of 59,213 laboratory-confirmed cases, including 245 deaths, have been reported to WHO. Regionally, from January 2022 to September 2025, the African Region reported 53,647 cases and 227 deaths, showing a notable 82% decrease in September 2025 compared to August. The Region of the Americas recorded 70,451 cases and 154 deaths, while the Western Pacific Region reported 6488 cases and 18 deaths, indicated 97% monthly decrease. Other WHO regions reported comparatively fewer cases, suggesting the outbreak is declining worldwide [81].
3.4. Transmission
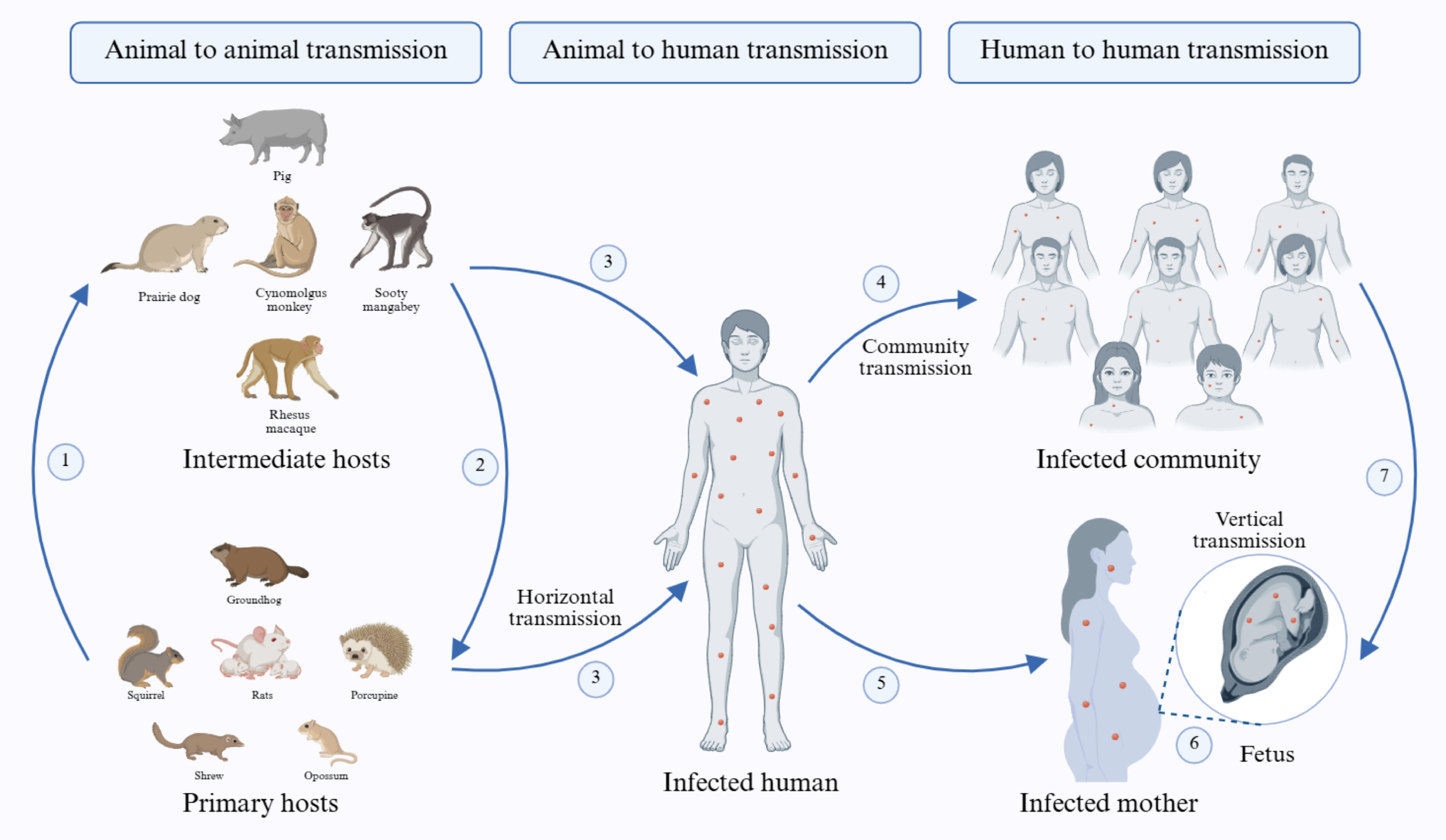
Although the name of the infection and its causative agent were derived initially from monkeys, the virus is believed to have its natural reservoir in various species of rodents and small primates inhabiting the forests of central Africa [82,83]. This zoonotic virus is transmitted from these animals, such as rats, squirrels, opossums, jerboas, elephant shrews, porcupines, and woodchucks, to different incidental intermediate hosts, typically primates including various species of macaques like rhesus and cynomolgus macaques, as well as monkeys like Sooty mangabey and Asian monkeys, domestic pigs, prairie dogs, and others, before ultimately reaching humans [84] (Figure 3). However, the exact natural reservoirs and the specific modes of animal-to-human transmission remain elusive [7,45]. Exposure to the virus can occur through various means, including direct contact with infected primary or intermediate hosts, bites or skin scratches, consumption of undercooked meat of infected animals, viral shedding from respiratory exudates or pustules, respiratory droplets, bodily fluids, particularly saliva or excrement of infected animals, and the international trade of exotic pets [57,85,86,87]. In the past, cases of Mpox were primarily associated with individuals who had been in contact with wild rodents and primates suspected of being infected with the virus, or with those who had returned from regions where Mpox is endemic [88,89]. However, in recent years, Mpox outbreaks have predominantly occurred through sexual contact with infected individuals [20]. Human-to-human transmission primarily occurs through direct contact with infected individuals, sharing personal belongings or food, and viral shedding from respiratory droplets, body fluids, and lesions [90,91,92,93]. Studies indicate that sexual activity is suspected as a mode of transmission in 95% of infected individuals, 98% of whom are either homosexual or bisexual [94,95]. Allan-Blitz et al. concluded that Mpox is a sexually transmitted disease due to the high consistency of its current transmission dynamics with those of STDs. Additionally, vertical transmission from mother to fetus may occur during delivery or close contact between mother and newborn during or after delivery [29,96,97,98] (Figure 3).
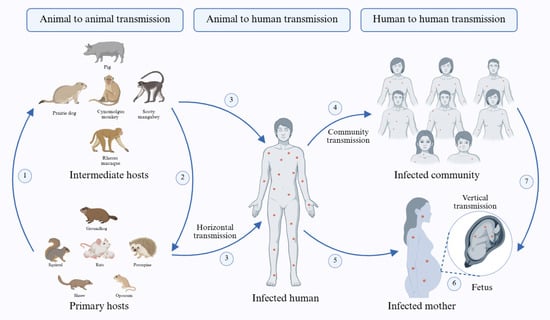
Figure 3.
Schematic illustration showings the major sources of the Mpox virus and the potential routes of transmission. Steps: 1. Transmission of the Mpox virus from its infected primary hosts to the intermediate hosts; 2. Reverse transmission from the infected intermediate hosts to the primary hosts; 3. Horizontal transmission of the Mpox virus from infected animals to humans; 4. Transmission of the virus from infected individuals to the community; 5. Horizontal transmission of Mpox from an infected person to a pregnant woman; 6. Vertical transmission of the Mpox virus from the infected mother to the fetus; 7. Transmission of the Mpox virus to the pregnant woman from the infected individuals of the community. Created in BioRender. Adnan, N. (2026) https://BioRender.com/45zmjlr.
3.5. Pathogenesis
The respiratory route is the primary portal of entry for the Mpox virus, with the mucous membranes of the nasopharynx and oropharynx serving as its principal site of replication [37,99]. Additionally, Mpox may enter the body through various other routes, including the ocular, oral, rectal, genitourinary tracts, and subcutaneous portals [100]. The Mpox virus demonstrates broad host tissue and organ tropism, indicating its ability to effectively invade and utilise a wide range of animal reservoirs in non-endemic regions [101]. Various organs, such as the liver, kidneys, pancreas, ovaries, heart, lungs, eyes, and genitourinary organs, exhibit tissue tropism for the Mpox virus [102]. However, the specific mechanisms of cellular invasion and replication remain unknown. The tissue tropism of the Mpox virus is believed to be influenced by particular host–cell receptors, as well as factors such as the Mpox inhibitor of complement enzymes (MOPICE), complement control protein (CCP), and various viral ligands. These components play a role in the virus’s ability to bind host cell receptors, facilitating cell invasion and the initiation of replication. While the specific functions of these ligands remain elusive, it is suggested that the virus may employ a range of alternative ligands to interact with host cell receptors effectively [103]. Following successful entry of the mature virion, which consists of a single membrane, into the body, Mpox infects local cutaneous or mucosal cells, which serve as the primary replication sites for the virus [104,105]. This process releases various cytokines, ultimately attracting professional phagocytes, particularly macrophages and dendritic cells [17]. These phagocytic cells engulf the cells and the mature virion, allowing the virus to survive. This latent stage of Mpox pathogenesis is known as primary viremia, a period during which no symptoms or lesions are present and can last up to 2 weeks [74,106]. The Mpox virus is transmitted to local lymph nodes via phagocytic cells, leading to secondary viremia with symptoms such as atypical flu-like characteristics [37,107]. Following secondary viremia, the contagious prodromal stage begins as the virus spreads from the lymph nodes to other organs, such as the eyes, lungs, liver, and genitourinary organs, where it proliferates [18].
3.5.1. Signs and Symptoms in Phases of Mpox Infection
Common signs seen in individuals infected with Mpox include rashes on various parts of the body, lesions in the anogenital and mucosal regions, fever, lethargy, muscle pain (myalgia), headaches, and lymphadenopathy (swelling of lymph nodes located in the neck, armpits, and groin). Additional symptoms may include sore throat, nasal congestion, cough, proctitis, tonsillitis, bacterial skin abscesses, ulcerated ventral tongue, genital vesicles, painful swallowing (odynophagia), perianal and tonsillar abscesses, rectal discomfort (which can lead to rectal perforation), and penile swelling [108]. Certain sexually transmitted infections (STIs), such as granuloma inguinale, molluscum contagiosum, chancres, and herpes simplex virus infections, can often be confused with Mpox [109].
The Mpox infection is generally divided into the invasive and cutaneous phases [110], and progresses through four distinct phases: incubation, prodromal, rash, and crusting [105] (Table 1).

Table 1.
Signs and symptoms of Mpox in different phases.
Incubation Period
During the incubation period, the Mpox virus initially proliferates at the entry site, including the mouth, nose, throat, or damaged skin. It subsequently spreads to nearby lymph nodes, leading to viremia, and can also affect other tissues and organs. The incubation period typically lasts 6 to 13 days, but can range from 5 to 21 days in some cases, and often occurs without noticeable symptoms [105,111]. The onset of the illness is characterised by symptoms such as fever, chills, swollen lymph nodes (lymphadenopathy), headaches, back pain, muscle soreness, and fatigue (Table 1).
Prodromal Period
The prodromal phase lasts 1 to 4 days. It is characterised by fever, headaches, muscle pain, back discomfort, chills, fatigue, sore throat, difficulty breathing (dyspnea), and swollen lymph nodes, all occurring before rash onset. In rare cases, additional symptoms such as sweating and skin-related issues may occur [113]. Fever is the most common prodromal symptom, with body temperatures typically ranging from 101.3 °F (38.5 °C) to 104.9 °F (40.5 °C), peaking around the second day of fever [105] (Table 1).
Rash Period
The rash typically becomes visible 1 to 3 days after the onset of fever and spreads outward from the centre of the body. Initially, it appears as flat red spots that persist for 2 to 4 weeks before evolving into raised bumps, blisters, and, eventually, pus-filled lesions, crusts, and scabs [114]. The area’s most frequently affected by skin lesions include the face (97.5%), torso (92.5%), arms (87.5%), legs (85%), genitals (67.5%), scalp (62.5%), palms (55%), soles of the feet (50%), mouth (37.5%), and eyes (25%) [105]. These lesions crust over within 5 to 7 days and fall off within 7 to 14 days [115]. The resultant scars are often indented, resembling a naval, and may cause significant itching and discomfort. The number of lesions can vary widely, ranging from just a few to thousands. Lesion count classifies the severity of human Mpox: 1–25 as mild, 26–100 as moderate, 101–250 as severe, and over 250 as very severe [105] (Table 1).
Crusting Period
The pustules eventually develop crusts that change colour within one to two weeks [116]. The time from lesion appearance to scab shedding typically ranges from 2 to 4 weeks. The scabs that fall away may be significantly smaller than the original lesions, and erythema or pigmentation can persist even after the scabs have detached, often leaving more than half of the scar still visible [105]. In males, genital lesions may lead to complications such as paraphimosis and necrotic crusts. Lesions in the rectal area can cause proctitis, with pain during bowel movements being the primary symptom. Additionally, ulcers with central depressions may emerge on the oral mucosa, tongue, pharyngeal wall, and tonsils [112] (Table 1).
3.5.2. Clinical Complications of Mpox Infection in Different Body Parts
Neurologic Complications
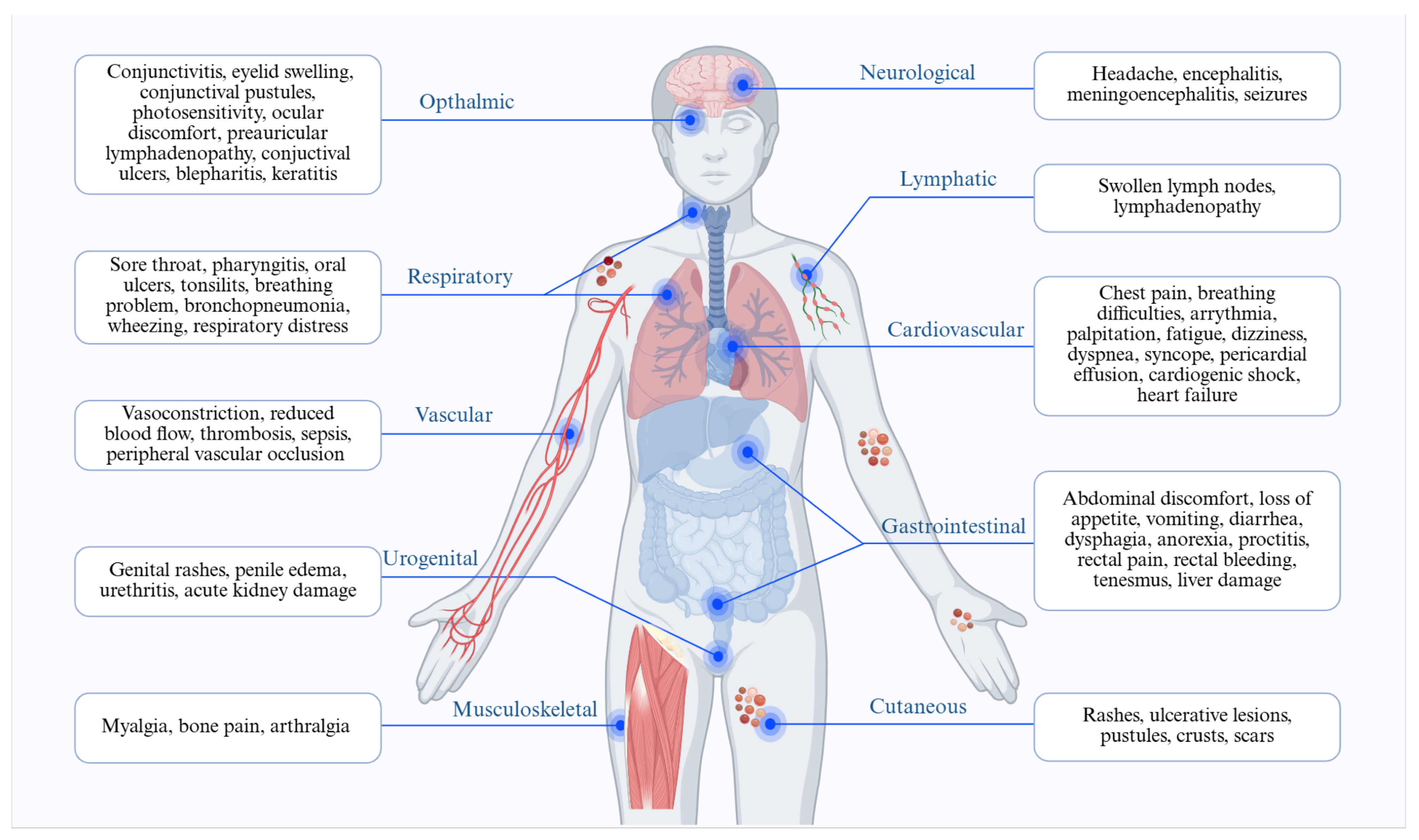
The most frequent neurological symptom is headache, often seen during the initial stage of the infection. Severe neurological issues, such as encephalitis, meningoencephalitis, and seizures, can arise after this initial phase [52] (Figure 4).
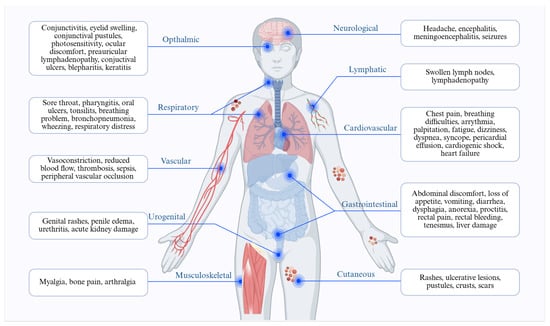
Figure 4.
Schematic illustration outlining the major clinical manifestations and complications of Mpox. Created in BioRender. Adnan, N. (2026) https://BioRender.com/uek6dcu.
Ophthalmic Complications
The primary ophthalmic symptoms associated with Mpox infection include distinctive rashes and lesions, frontal headaches localised to the periorbital and orbital regions, conjunctivitis, and eyelid swelling [117,118]. The pustular rash, discharge, and crusted secretions may lead to an inability to open the eyelids for several days. Additionally, conjunctival pustules can develop, accompanied by excessive tearing, light sensitivity, and discomfort [118] (Figure 4). Kaufman et al. introduced the term Mpox-related ophthalmic disease (MPOXROD) to describe the range of eye-related manifestations that may occur with Mpox infection [119]. Ulcerative lesions and swollen eyelids typically appear at the margins of the eyelids and conjunctiva. Many patients have exhibited preauricular lymphadenopathy, along with vesicular and infiltrative lesions. Large conjunctival ulcers may have smooth edges and a pale colouration [119]. Symptoms related to pox-induced conjunctivitis can include conjunctival ulcers, pseudomembranous or subconjunctival nodules, extensive blistering, papular lesions on the conjunctiva, and follicular reactions of the conjunctiva. Secondary symptoms accompanying Mpox patients experiencing conjunctivitis include nausea, light sensitivity, mouth sores, swollen lymph nodes, sore throat, fatigue, chills, and sweating [117]. Blepharitis, characterised by inflammation of both eyelids, may develop due to a blockage in the small oil glands at the bases of the eyelashes, leading to irritation and redness [120].
Cardiovascular Complications
Cardiovascular effects associated with Mpox infection can vary in both type and severity. These effects may affect various aspects of the cardiovascular system, including arrhythmias, myocarditis, and pericarditis. Myocarditis can range from mild inflammation to severe heart muscle damage. The primary mechanism underlying Mpox-related myocarditis is lymphocytic inflammation, typically occurring 10 to 14 days after the initial infection, followed by myonecrosis [121,122]. Common symptoms include chest pain, breathing difficulties, irregular heartbeats, fatigue, and diminished exercise capacity. In severe cases, myocarditis can lead to acute heart failure, cardiogenic shock, or even sudden cardiac death [123].
Mpox can lead to pericarditis, an inflammation of the pericardium that often causes sharp chest pain, particularly when lying down or deep breathing. Additional symptoms may include a low-grade fever, cough, and a distinctive friction rub detectable during a chest examination. Symptoms associated with pericarditis encompass chest pain, fatigue, shortness of breath (dyspnea), palpitations, and, in severe cases, cardiogenic shock [124]. The inflamed pericardium often intensifies chest pain with deep breaths or when positioned horizontally. Moreover, in more critical scenarios, fluid accumulation in the pericardial space, known as pericardial effusion, may impair cardiac function [125] (Figure 4).
Mpox may also disrupt the heart’s regular electrical conduction system, leading to arrhythmias. During Mpox infection, symptoms of arrhythmia may include palpitations, dizziness or lightheadedness, fainting (syncope), and chest pain [123]. This interference can occur directly, disrupting the rhythm and timing of heartbeats [123]. Inflammation and damage to the heart muscle or its conduction pathways can disrupt electrical signals, resulting in irregular heart rhythms [125].
Gastrointestinal Complications
The gastrointestinal complications associated with Mpox infection may encompass abdominal discomfort, anorexia, diarrhoea, nausea, vomiting, proctitis, rectal pain, painful bowel movements, and the sensation of incomplete defecation (tenesmus) [126,127] (Figure 4). There have also been reported cases of liver damage linked to this condition. Loss of appetite is the most common gastrointestinal symptom of Mpox, affecting 47% of patients, as reported by Simadibrata et al. Other commonly reported symptoms include vomiting (12%), nausea (10%), abdominal discomfort (9%), and diarrhoea (5%). Additionally, proctitis, along with rectal or anal discomfort and rectal bleeding, is prevalent, with relative frequencies of 11%, 25%, and 12%, respectively [127].
Pulmonary Complications
The respiratory system may be affected, leading to severe coughing, difficulty breathing, or bronchopneumonia [108]. The impacted areas within the respiratory organs can be divided into three categories: upper respiratory (which includes symptoms such as a runny nose, sore throat, pharyngitis, oral ulcers, and tonsillitis), lower respiratory (characterized by wheezing, coughing, and respiratory distress), and general respiratory (which encompasses a combination of symptoms from both the upper and lower respiratory categories) [128] (Figure 4).
3.6. Diagnosis of Mpox
Mpox is primarily diagnosed based on a combination of clinical indicators and epidemiological factors, with definitive confirmation obtained by nucleic acid amplification testing (NAAT) [74]. The primary method for identifying suspected cases involves detecting viral DNA from swabs collected from the patient’s skin lesions and wounds. A comprehensive diagnosis should incorporate both thorough clinical evaluations and laboratory analyses [129,130,131]. A range of advanced laboratory techniques is currently utilised for Mpox diagnosis. Key methods include cell culture, polymerase chain reaction (PCR), and enzyme-linked immunosorbent assay (ELISA). Additionally, tests include immunohistochemistry, electron microscopy, virus isolation, culture and cytopathic effect (CPE) screening, immunofluorescent antibody testing, and in situ histopathological examination. Biosensors are cutting-edge tools designed for rapid viral detection, while Western blot analysis and genetic sequencing further characterise the virus’s structural and genetic properties [129,132,133,134,135,136].
To perform these diagnostic procedures, a variety of clinical samples are necessary, including fluid from lesions, dried scab material, crusts, oropharyngeal and nasopharyngeal swabs, tonsillar tissue, punch biopsy specimens, whole blood, and serum collected from both the acute and convalescent phases of infection. The most effective specimens for laboratory confirmation are those obtained directly from skin lesions. All samples that could potentially contain the Mpox virus should be processed in Biosafety Level 2 (BSL-2) facilities, which minimise exposure risks and safeguard the integrity of diagnostic testing [133].
3.6.1. Polymerase Chain Reaction (PCR)
Real-time polymerase chain reaction (RT-PCR) is an advanced genetic technique used to detect the Mpox virus. This method emphasises amplifying specific target genes typically located within the conserved regions of the virus’s genetic material. Key genes selected for PCR amplification include the extracellular envelope protein gene (B6R), the E9L gene, which encodes DNA polymerase, and the RPO18 gene, an essential subunit of DNA-dependent RNA polymerase. Additional genes of interest include the complement-binding protein genes, namely C3L, F3L, and N3R [109]. Both RT-PCR and conventional PCR tests must be conducted in a laboratory environment classified as Biosafety Level 2 (BSL-2) to ensure safety, given the virus’s infectious nature [137]. To further confirm the presence of the Mpox virus, PCR-amplified genes or gene fragments may be analysed using restriction length fragment polymorphism (RFLP) [134]. A positive result from an orthopoxvirus PCR test, followed by a confirmatory Mpox-specific PCR and sequencing, provides strong evidence of an active Mpox infection. Notably, the PCR test is recognised as the gold standard in laboratory examinations due to its outstanding specificity and sensitivity, making it essential for rapidly and accurately detecting the monkeypox virus [133,135,138,139] (Table 2).

Table 2.
Laboratory test methods available for the precise diagnosis of Mpox.
3.6.2. Immunologic Assays
Serological methods detect Mpox antigens or IgG and IgM antibodies using techniques such as immunohistochemistry, ELISA, and rapid tests such as lateral flow and dot blot assays. ELISA is the preferred method for serum antibody detection, with specific IgM appearing around 7 days and IgG around 21 days post-rash onset [143,146]. Immunochemistry analysis can employ either polyclonal or monoclonal antibodies targeting orthopoxviruses to differentiate between poxvirus and herpesvirus infections. Levels of antiviral antibodies and T-cell responses have been observed to increase around the time the illness manifests. If an individual without a history of rash or severe illness tests positive for both IgM and IgG antibodies, an indirect diagnosis of Mpox may be considered [134]. Serological testing for Mpox can reinforce a diagnosis, especially when NAAT is not feasible. Detecting IgM in acutely ill patients (within 4 to 56 days post-rash onset) or measuring IgG in paired serum samples (collected at least 21 days apart, with the first taken during the first week of illness) can further assist the diagnosis [74]. The ELISA can detect specific IgM and IgG antibodies in the serum of patients infected with Mpox approximately 5 and 8 days after infection, respectively, making it a valuable tool for serological testing. A fourfold increase in serum antibodies can be utilised to diagnose Mpox infection during both the acute and convalescent phases [109]. A CDC study analysed sera from 37 confirmed Mpox cases during the 2003 outbreak in the USA using IgM-capture and IgG ELISAs targeting orthopoxvirus antigens. It detected IgM in 94.5% of cases (optimal ≥5 days after rash onset) and IgG in 80.5% (optimal ≥8 days), with high sensitivity, e.g., 94.8% for IgM and 100% for IgG, and good specificity of 94.5% for IgM and 88.5% for IgG, correlating well with PCR [155]. Various immunological methods are available for detecting IgG and IgM antibodies, as well as immunohistochemistry for identifying viral antigens to detect orthopoxvirus DNA. However, due to the serological cross-reactivity of orthopoxviruses, none of these tests can provide specific confirmation of Mpox, as this phenomenon was seen in the case of the detection assay of other viruses [156] (Table 2).
3.6.3. Electron Microscopy
Electron microscopy is a crucial tool for detecting poxvirus infections and is one of the early indicators of rash-related illnesses. When examined under an electron microscope, the distinctive morphology of poxvirus virions becomes apparent. A notable instance of this was observed during the recent Mpox outbreak in the United States, where electron imaging revealed keratinocytes containing a significant number of mature virions alongside immature virions in the synthesis phase, often referred to as “viral factories”, situated within the cytoplasm of infected cells [134,140,141]. However, relying exclusively on electron microscopy for poxvirus identification is not entirely reliable, as virions cannot be definitively identified by their shape alone [109], and it is important to distinguish the Mpox virus from other virus families, such as Herpes and Parapox viruses [133,142]. Moreover, the need for a complex laboratory setup, specialised, trained personnel, specific reagents, and high operating and maintenance costs limits its practicality and usefulness as a general diagnostic method for Mpox. Therefore, it can serve as an additional technique to support other diagnostic methods [142,157] (Table 2).
3.6.4. Other Diagnostic Approaches
Multiple nucleic acid amplification tests have been developed to identify Mpox. Combining two distinct viral gene targets, such as E9L and B6R, can provide a reliable, sensitive, and rapid diagnostic method. However, access to these tests remains limited. Recently, a self-contained cartridge, the Cepheid GeneXpert, was developed as an alternative to traditional PCR detection methods. The GeneXpert assay has demonstrated high sensitivity, specificity, negative predictive value, and positive predictive value in suspected specimens, regardless of specimen type (crust or vesicular swab) [154,158]. As with electron microscopy, the cultivation of viruses for detection poses various limitations and challenges. Key among these are the need to identify suitable cells for virus replication, isolate the virus, and have a laboratory equipped with the necessary tools. However, cytopathic effect (CPE) screening methods are employed for certain viruses. Research has shown that the Mpox virus replicates rapidly in cell cultures, displaying detectable CPE in Rhesus Monkey Kidney (RMK), Buffalo Green Monkey (BGM), A549, and Medical Research Council cell strain 5 (MRC-5) cell lines within approximately two days. Despite this, cell culture and electron microscopy techniques remain critical in vaccine preparation, but not for diagnostics. Today, viral cultures have primarily been replaced by nucleic acid amplification tests and antigen detection methods [129]. Individuals infected with Mpox typically exhibit detectable levels of anti-orthopoxvirus antibodies. Skin tissue biopsies can also serve as viable clinical samples for diagnostic evaluation when clinically indicated [74]. Furthermore, various methods, including recombinase polymerase amplification (RPA), CRISPR-Cas-based technique, loop-mediated isothermal amplification (LAMP), and restriction length fragment polymorphism (RFLP), have been developed to detect Mpox virus DNA [109,144,145,147,148,149,150,151] (Table 2). The World Health Organisation recommends nucleic acid amplification testing (NAAT) for detecting the Mpox viral genome, hemagglutinin, acidophilic-type inclusion bodies, and the crmB genes [152,159]. Immunohistochemical examination with monoclonal and polyclonal antibodies can aid in distinguishing a herpesvirus infection from a poxvirus disease [134,146].
A flexible lateral flow immunoassay (LFIA) has been developed, featuring robust colourimetric capabilities and enhanced dual-signal fluorescence output. This innovation enables rapid, on-site, highly sensitive detection of the Mpox virus antigen (A29L) [160]. Utilising readily accessible materials and demonstrating no cross-reactivity, an electrochemical biosensor based on electrochemical impedance spectroscopy (EIS) has been shown to effectively identify the A29 viral protein from Mpox virus antigens in saliva and plasma samples within 15 min [161]. The MAGLUMI® Mpox virus Ag Chemiluminescence Immunoassay (CLIA) assay kit, produced by Shenzhen New Industries Biomedical Engineering Co., Ltd. (Snibe), employs a double-Mpox virus monoclonal antibody that selectively targets the A29 and A35R epitopes [162]. This represents the first application of the Helicase-Dependent Amplification (HDA) method, combined with a lateral flow test (LFT), to detect Mpox infection. The HDA-LFT assay exhibited 94% specificity and 86% sensitivity in identifying the Mpox virus-specific conserved fragment F3L [163]. Additionally, a highly sensitive and specific MPXV-MCDA-LFB assay was developed, integrating isothermal multiple cross-displacement amplification (MCDA) with a nanoparticle-based lateral flow biosensor (LFB) for the rapid detection of Mpox virus target genes (D14L and ATI). This assay demonstrated no cross-reactivity with non-Mpox virus templates, establishing it as a reliable diagnostic tool for Mpox infection [164].
3.7. Treatment Approaches and Medications for Mpox
3.7.1. Supportive Care and Conventional Therapeutic Supports
The management of Mpox has integrated supportive care with targeted antiviral therapies and vaccination strategies, especially following the 2022 outbreaks [165]. In most cases, supportive care is sufficient to meet patients’ clinical needs. Since there are no Food and Drug Administration (FDA)-approved antivirals outside of special circumstances, the main focus of treatment is symptom management. Although many antiviral agents are in clinical trials or used under compassionate use protocols, none are FDA-approved for Mpox [29,166]. On the other hand, for mild infections, symptomatic treatment is recommended to relieve symptoms; however, for severe Mpox, the focus shifts to supportive care to manage systemic complications, including secondary bacterial infections [167].
Therapeutic interventions may include administering antipyretics to reduce fever and flu-like symptoms, analgesics for pain relief, and antibiotics when secondary infections occur. Additionally, treatment should be customised to meet the specific needs of vulnerable groups, such as individuals with serious health conditions, immunocompromised patients, people living with HIV, as well as pregnant women, children, and the elderly [29].
3.7.2. Antiviral Drugs to Treat Mpox
It is essential to note that antiviral medications specifically approved for the treatment of Mpox have not yet received final authorisation [168,169]. Several studies are currently underway to evaluate the effectiveness of treatment approaches and therapies traditionally used for smallpox in managing Mpox [37,170]. The role of antibody responses in protecting individuals against infections caused by poxviruses has been documented [171]. Several antiviral agents, including tecovirimat, cidofovir, and brincidofovir, originally developed for the treatment of smallpox and related infections caused by other orthopoxviruses, are now being repurposed for Mpox [129,172,173,174,175,176]. The Centres for Disease Control and Prevention (CDC) has implemented a new protocol permitting the emergency use of tecovirimat for non-variola infections, including those diagnosed with Mpox. However, there is limited evidence supporting the effectiveness of cidofovir and, even less so, tecovirimat in treating severe cases of Mpox [177,178,179].
Cidofovir has been utilised in various studies as a treatment option for severe cases of Mpox, albeit with a limited patient cohort. Research conducted by Raccagni et al. reported no documented side effects or significant changes in creatinine levels following cidofovir administration. Additionally, the article indicated that patients did not exhibit any new symptoms after treatment [180]. Tecovirimat has been used in a randomised controlled trial (RCT) for endemic Mpox (Clade I MPXV) and in a phase-3, double-blinded RCT for Clade II mpox. However, the results did not show significance for early clinical resolution of lesions, pain reduction, or viral clearance among participants [181,182]. Immunotherapy for Mpox shows promise in mitigating the severity of the disease. The potential benefits of combining antiviral medications with immunotherapy may exceed those achieved through antiviral treatment alone, thereby enhancing overall therapeutic efficacy [183,184,185].
3.7.3. Antimicrobial Drugs to Treat Mpox
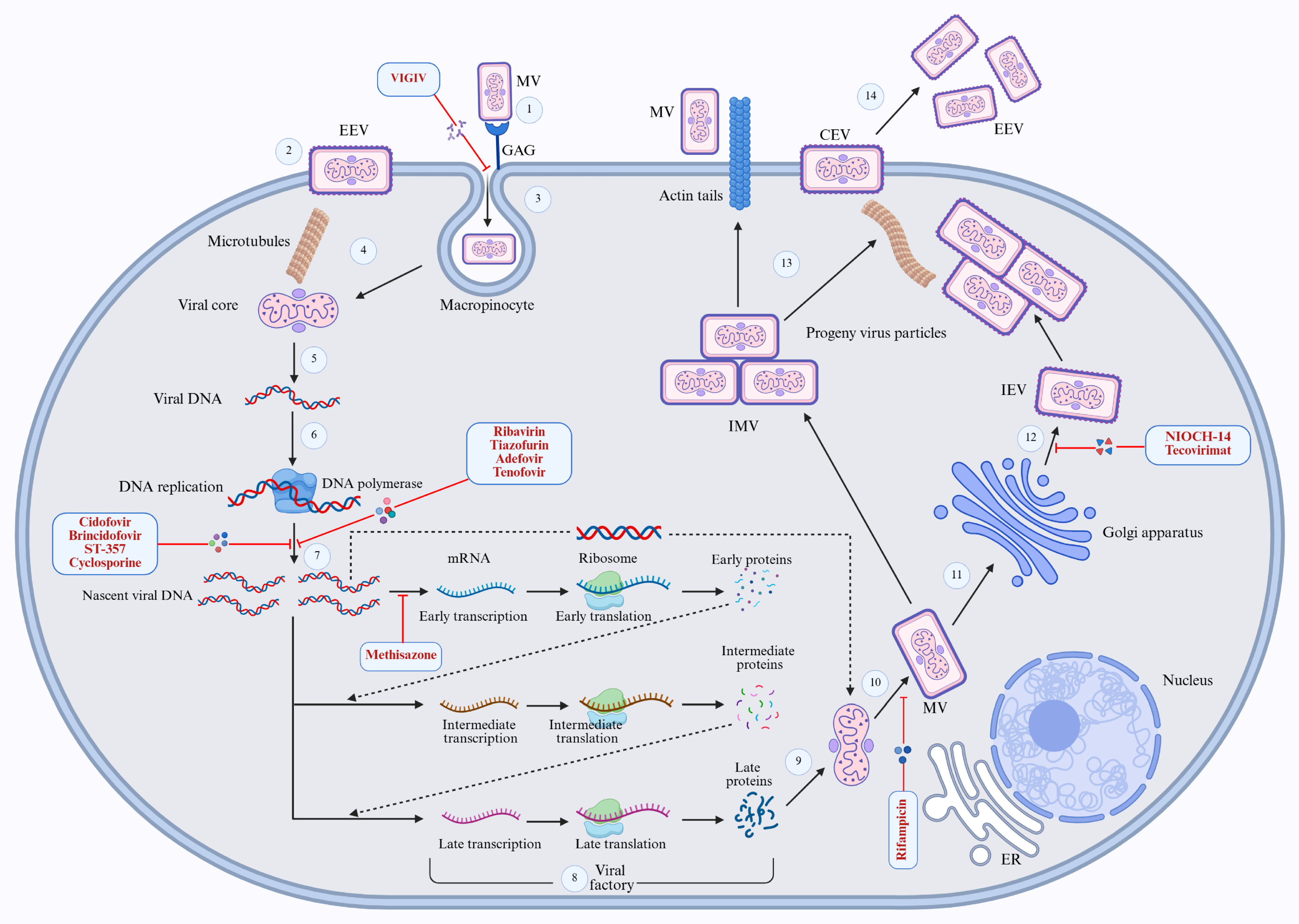
Conventional antibiotics, cyclosporine, and rifampicin are being used to some extent to treat Mpox. Cyclosporine inhibits poxvirus DNA replication by incorporating into the nascent DNA chain [194]. In contrast, the rifampicin antibiotic, known to be effective against poxviruses for a long time, exerts its antiviral activity against poxviruses by blocking the interaction between the viral proteins D13 and A17, which is necessary for the synthesis, assembly, and maturation of membrane precursor proteins [193,202] (Figure 5).
In addition to these drugs, immunoglobulin therapy, such as Vaccinia Immune Globulin Intravenous (VIGIV), is approved by the FDA and recommended by the CDC [203]. It is administered as an intravenous immunoglobulin solution containing high titers of IgG against vaccinia virus derived from recovered patients. This therapy is used to manage complications, such as severe generalised vaccinia, following smallpox vaccination [60,204] (Figure 5).
3.8. Vaccines Against Mpox
Currently, no vaccine specifically designed for Mpox is available, necessitating reliance on vaccinia virus-based vaccines, initially developed for smallpox, since members of the Orthopoxvirus genus are capable of inducing serological cross-protection against Mpox [205,206]. The history of smallpox vaccination dates back to the use of bovine pox pustules, long before the discovery of viruses and the development of modern immunology and vaccination methods [207]. Over time, this approach evolved to include the use of attenuated viral strains, leading to the development of safer, more effective vaccines, driven by advancements in virology, immunology, and vaccine research.
3.8.1. Traditional Mpox Vaccine Candidates
Traditional Mpox vaccine candidates, ranging from inactivated whole-cell vaccines to recent attenuated vaccines, have been developed, approved, and administered in different countries to provide immunity against various orthopoxviruses [208,209]. Attenuated strain-based vaccines can be categorised into three generations based on the attenuation technique, cultivation methods, and replication status of the vaccine strains [210,211].
The initial smallpox vaccine, developed in the previous century, utilised live vaccinia virus strains such as Lister/Elstree, EM-63, or Tian-Tian. These strains were obtained from calf lymph nodes, purified, and lyophilised before being administered by scarification using bifurcated needles [210]. The Dryvax vaccine, developed and administered in the USA, is a first-generation, live, replication-competent vaccinia virus-based vaccine that offers protection against both smallpox and Mpox [212,213,214,215] (Table 4). While demonstrating notable effectiveness against Mpox, the vaccine was associated with occasional severe adverse effects, including Stevens-Johnson syndrome, myo/pericarditis, fetal vaccinia, encephalitis, progressive vaccinia, eczema vaccinatum, and occasional fatalities among patients, particularly among the immunocompromised [216].

Table 4.
Major vaccines of different generations are used to prevent Mpox.
The second-generation vaccines include several different live attenuated vaccines, such as the Lister-based vaccine (RIVM), Elstree-BN, the Aventis Pasteur Smallpox Vaccine (APSV), the vaccinia virus-based Lister vaccine, and the advanced live attenuated vaccine ACAM2000 [218,219,230]. The Lister-based vaccine (RIVM) is a single-passage, live-attenuated, replication-competent vaccinia virus derived from strains isolated from calf lymph nodes and directly transferred to rabbit kidney cells for replication [231]. The Elstree-BN smallpox vaccine, a second-generation vaccine, was developed using the Lister strain, replicated in chicken embryonic cells [232]. Additionally, the ACAM2000 vaccine, derived from Dryvax, demonstrated greater immunogenicity and fewer adverse effects than first-generation vaccines [220,221,222,233,234] (Figure 6) (Table 4).
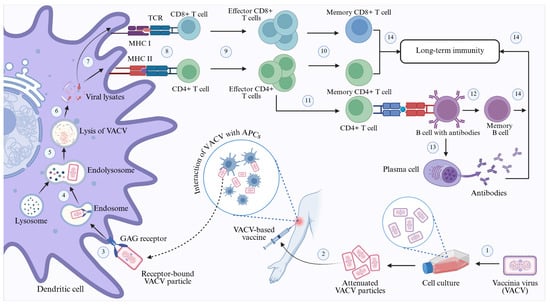
Figure 6.
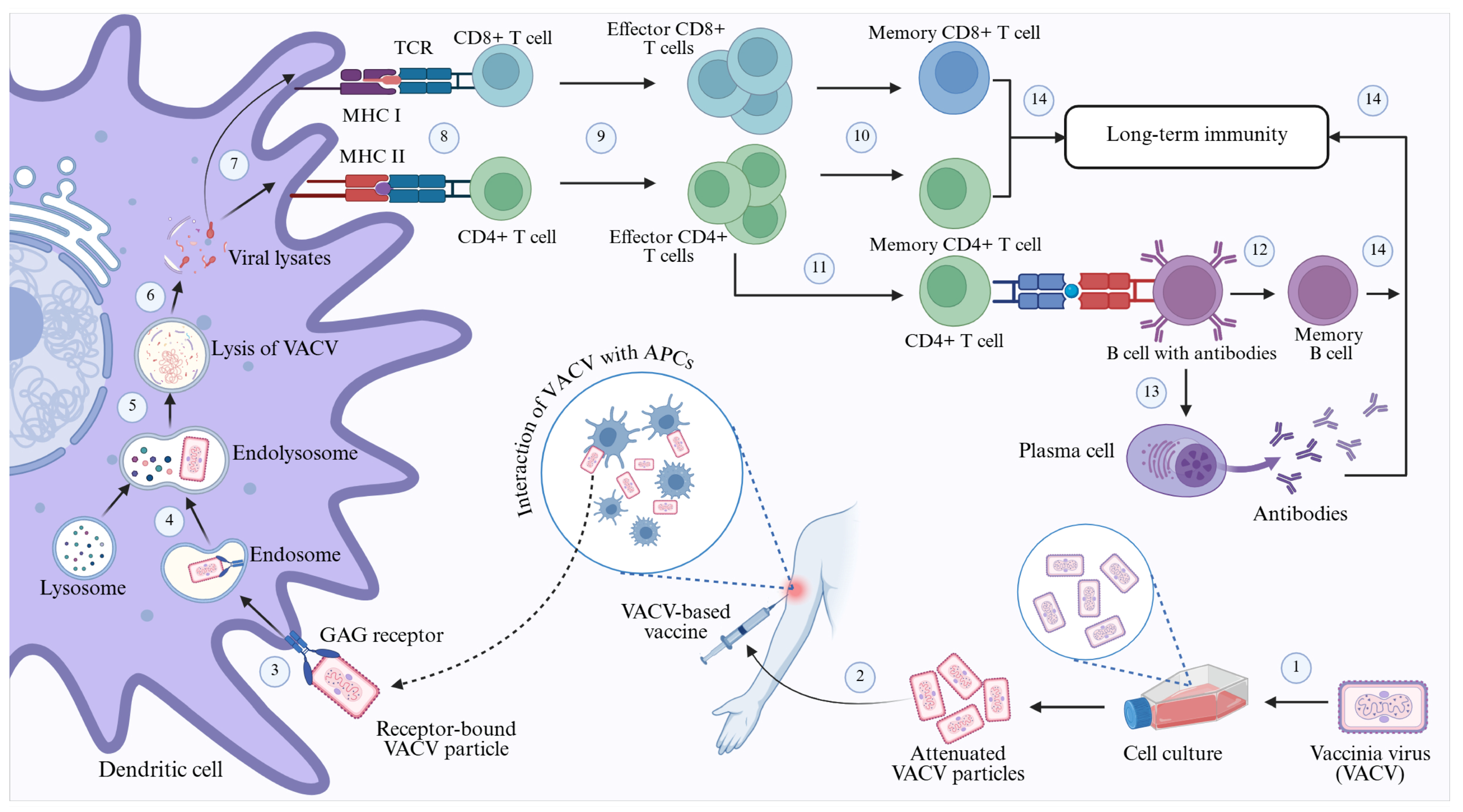
Mechanism of action of the attenuated Vaccinia virus-based vaccine used for immunisation against Mpox. Steps: 1. Attenuation of live VACV particles through repeated cell culture; 2. Preparation and administration of VACV-based vaccine; 3. Attachment and subsequent entry of VACV particles into dendritic cells through GAG receptor-mediated macropinocytosis; 4. Formation of the endolysosome through fusion of the VACV-containing endosome with lysosome; 5. Endosomal lysis of VACV particles by lysozyme; 6. Release of the VACV lysates into the cytoplasm of the dendritic cell; 7. Presentation of the antigenic viral lysates on the dendritic cell through the interaction with MHC class I and MHC class II molecules; 8. Attachment of CD8+ and CD4+ T cells with antigen-bound MHC I and MHC II receptors, respectively, through their interaction with T cell receptors; 9. Activation of the CD4+ and CD8+ T cells and production of the effector T cells; 10. Generation of memory CD4+ and CD8+ T cells from the effector cells; 11. Attachment and CD4+ mediated activation of B cells; 12. Conversion of effector B cell into memory B cell; 13. Generation of antibody-secreting plasma cells from the effector B cells; 14. Memory T cells, memory B cells, and plasma cells provide long-term immunity. Created in BioRender. Adnan, N. (2026) https://BioRender.com/exe6ksk. Abbreviations: VACV—vaccinia virus; APC—antigen-presenting cell; GAG—glycosaminoglycan; MHC—major histocompatibility complex; TCR—T cell receptor; CD—cluster of differentiation.
Additionally, the Aventis Pasteur smallpox vaccine, a second-generation vaccine based on replication-competent vaccinia virus, exhibits safety and immunogenic properties similar to those of ACAM2000 [217,235]. Although effective against smallpox, this vaccine has not yet been used for Mpox.
The third-generation live attenuated smallpox vaccines were developed using replication-deficient, modified vaccinia virus strains to mitigate adverse effects stemming from contamination with animal lymph nodes or complications arising from autoinoculation during vaccine strain development. The LC16m8 vaccine, a representative of third-generation attenuated vaccines, was developed in Japan in 1970. The vaccine was derived from the Lister strain, which underwent passage in primary rabbit kidney cells and Vero cells. chorioallantoic membrane (CAM) to obtain the desired clone, designated LC16m8. This strain exhibited complete non-replicating and non-infectious characteristics, rendering it suitable for use in the vaccine [223,224,225,226,236]. The recently developed live attenuated vaccine, IMVAMUNE, also known as JYNNEOS or IMVANEX, represents a cutting-edge, cell culture-based, third-generation smallpox vaccine. This advanced vaccine was developed using a modified vaccinia Ankara strain that underwent 596 passages in chick embryo fibroblasts and was subsequently attenuated through multiple rounds of dilution. IMVAMUNE, is authorised by the Advisory Committee on Immunisation Practices (ACIP) and the US FDA for pre-exposure prophylaxis against smallpox and Mpox [237,238,239] (Table 4).
Upon administration, the attenuated strains, combined with vaccine adjuvants, promote the uptake of specific viral surface antigens by dendritic cells at the vaccination site. Subsequently, these dendritic cells are activated by pattern recognition receptors in response to cytokine signals generated by the adjuvants. Upon recognition by dendritic cells, the vaccine strains’ antigens are transported to the lymph nodes, where they are presented to T cells by MHC I and MHC II molecules. Subsequently, T cells are activated by binding to the antigen-presenting MHC molecules through their T cell receptors. The MHC class I molecule activates CD8+ T cells, assisted by helper T cells, which initiate cytotoxic activity and generate memory T cells. Conversely, when presenting antigens, the MHC class II molecule stimulates CD4+ T cells, also known as helper T cells, by releasing cytokines. This activation of CD4+ T cells subsequently triggers B cells to differentiate into plasma cells, thereby increasing antibody production and forming memory B cells. The resulting memory B and T cells persist in the body, conferring long-lasting immunity against the antigens and orchestrating a response to neutralise or eliminate poxviruses upon re-entry [240,241] (Figure 6).
3.8.2. Novel Mpox Vaccine Candidates
Multiple novel Mpox vaccine candidates, including protein subunit, recombinant protein subunit, virus-like particle (VLP)- based, and nucleic acid-based (e.g., DNA and mRNA) vaccines, have been designed and are currently undergoing in silico, molecular docking, or animal trials [230]. The novel vaccine platforms were designed to boost both humoral and cellular immune responses while reducing the likelihood of adverse side effects, such as reactogenicity. Additionally, they aim to minimise labour, expenses, and, most importantly, the risks associated with developing traditional live-virus-based vaccines [242]. Several protein-subunit vaccines have been developed utilising distinct immunogenic glycoprotein fragments of the vaccinia virus, combined with various adjuvants to augment immune responses [243]. Notably, Buchman et al. detailed the design of a protein-based smallpox vaccine. This vaccine incorporates purified ectodomains of A33 and B5, extracted from the enveloped vaccinia virion, and L1 and A27 proteins from the mature vaccinia virion. Furthermore, the vaccine formulation included aluminium hydroxide and CpG as adjuvants. The immunogenicity and safety profile of this vaccine were assessed through animal model studies involving cynomolgus macaques [244].
Bhattacharya et al. have outlined a novel multi-epitopic peptide-based potential vaccine candidate against Mpox. This vaccine incorporates 10 B- and T-cell-derived epitopes spanning 147 amino acid residues. Additionally, the CTxB adjuvant is attached to the N-terminus via the EAAAK peptide linker, and the PADRE sequence (AKFVAAWTLKAAA) is attached to the C-terminus to enhance the vaccine’s immunogenicity [245].
Shantier et al. developed a 275-amino-acid, multi-epitopic protein subunit vaccine. This vaccine was constructed using MHC-1 and MHC-2 molecules, B cell epitopes, appropriate linkers (e.g., EAAAK), and adjuvants, including the PADRE sequence and RS09 [246]. Similarly, Heraud et al. designed a subunit recombinant vaccine using plasmid DNA encoding the Mpox orthologs of the vaccine virus proteins L1R, A27L, A33R, and B5R. This vaccine has demonstrated the ability to elicit helper T-cell responses and to generate binding antibodies against all four proteins. Presently, it is undergoing animal model trials involving Rhesus macaques [247].
Among the DNA-based nucleic acid vaccines, the intradermal multivalent smallpox DNA vaccine, developed by Hirao et al., is notable for its utilisation of chemically synthesised and human codon-optimised VACV Western Reserve (WR) Strain genes, including A27, F9, H3, L1, A33, A56, and B5, in conjunction with the core antigen A4. This approach aims to enhance the vaccine’s cytotoxic T-cell activity [248].
In contrast, Mucker et al. have developed an mRNA-based vaccine utilising lipid nanoparticle delivery to administer unmodified mRNA encoding three monoclonal antibodies: c8A, c6C, and c7D11. This vaccine is undergoing preclinical animal studies to assess its immunogenicity and safety profile [249].
3.9. Preventive Measures of Mpox
The Mpox virus primarily spreads through direct contact with infected animals or person-to-person via respiratory droplets, bodily fluids, and skin lesions [61]. Understanding these transmission routes is essential for prevention, especially as the virus appears in non-endemic countries [7]. Effective prevention depends on rapid detection, which enables the isolation of infected individuals and reduces spread through symptom recognition and contact tracing [30,76]. Infected individuals should be isolated until they are no longer contagious, which is vital for outbreak control [250]. To reduce transmission and alleviate symptoms, consulting a healthcare provider, staying in well-ventilated areas, and practising good hygiene and sanitation are recommended.
Over-the-counter pain relievers such as paracetamol and ibuprofen can help relieve pain [251]. To prevent infections and the spread of viruses, popping blisters or scratching sores should be avoided, and maintaining isolation until all lesions have crusted over and scabs have fallen off is essential. Individuals should remain alert for symptoms for up to 21 days after exposure and take appropriate precautions [29]. Those with close contact with infected individuals should be quarantined to monitor for symptoms to prevent secondary transmission during outbreaks [30,252].
Healthcare professionals and individuals in close contact with infected persons should use PPE, including masks, gloves, and gowns [76,168,253]. It is essential to strictly adhere to disinfection protocols to prevent the spread of these infections. Orthopoxviruses can be inactivated by agents such as 70% ethanol (≤1 min), 0.2% peracetic acid (≤10 min), 1–10% probiotic cleanser (1 h), sodium hypochlorite (0.25–2.5%; 1 min), 2% glutaraldehyde (10 min), and 0.55% ortho-phthalaldehyde (5 min) [254].
Public health agencies must strengthen surveillance in both endemic and non-endemic areas [7,255], along with adequate environmental controls, such as educating communities to avoid contact with wild animals and to practice sanitation, to help reduce zoonotic Mpox transmission in endemic regions. Governments should issue travel advisories and provide guidance on preventive measures, such as avoiding contact with animals and maintaining good hygiene. Travellers must receive guidance on preventive measures, including avoiding contact with animals and maintaining strict hygiene practices [1,81]. Promoting behavioural changes, such as avoiding contact with suspected cases and practising proper hygiene and sanitation, is also essential [256,257]. Public awareness is essential; health campaigns should educate about symptoms, transmission, and prevention, increasing awareness and encouraging early healthcare visits [81,258,259].
In endemic areas, contact with animals such as primates and rodents that may carry Mpox should be strictly avoided to reduce the risk [260]. Educating the general public raises awareness and encourages safe hunting and handling [57]. Collaborating with veterinary services can help track and control Mpox in animals. Regular health checks and population monitoring are key to identifying potential sources of the virus [258]. Community leaders and healthcare professionals should work together to dispel misinformation and provide clear guidance on protective measures [259].
Limited public awareness of Mpox worsens due to social media misinformation, leading to fear, panic, and discrimination against key populations. This phenomenon, known as an ‘infodemic’, hinders response efforts by obscuring facts and marginalising experts, much as in the early stages of the COVID-19 pandemic [261,262,263,264]. A strategic approach involving policymakers, healthcare providers, the media, and social platforms is essential for raising awareness and reducing stigma. Public figures and medical professionals play crucial roles in reassuring the public and promoting health guidelines. Addressing research gaps and improving healthcare communication can build trust and encourage timely care. Emphasising a One Health approach is vital to combat stigma and strengthen public health efforts [261].
Effectively controlling Mpox mandates a multifaceted approach that includes public health education, vaccination, surveillance, and community involvement [7]. The successful implementation of these measures will reduce the likelihood of Mpox outbreaks and safeguard public health [76]. Furthermore, continuous research and effective coordination among health sectors are crucial for adapting and updating strategies as new information becomes available [255,265].
4. Future Perspectives and Recommendations
The ongoing global spread of Mpox, particularly highlighted by the 2022 outbreak, underscores significant deficiencies in diagnosis, treatment, and prevention strategies, as well as in healthcare infrastructure and facilities. These deficiencies pose formidable challenges for healthcare organisations and researchers alike. The absence of Mpox-specific antiviral drugs exacerbates these challenges, underscoring the urgent need for randomised controlled trials of promising antiviral candidates, including brincidofovir, tecovirimat, and novel agents such as ST-357. Special attention must be directed towards high-risk populations, including immunocompromised individuals, pregnant women, and communities in endemic regions, particularly in Africa. Moreover, vaccine development must extend beyond repurposing existing smallpox vaccines to encompass robust research initiatives and clinical trials focused on Mpox-specific vaccine candidates. This includes, but is not limited to, advancements in virus-like particles, mRNA vaccine technologies, and multi-epitope subunit vaccines targeting key antigens such as A29L, A35R, and L1R. Furthermore, ensuring equitable access to antiviral medications and vaccines globally is critical to mitigating the Mpox crisis.
The acceleration of research to develop more precise and rapid diagnostic approaches is paramount, particularly given the current reliance on clinical manifestations. While PCR remains the gold standard, its limited availability in resource-constrained settings underscores the need for rapid point-of-care testing platforms, including loop-mediated isothermal amplification (LAMP), CRISPR-Cas12 assays, and lateral flow tests targeting specific viral antigens. Strengthening real-time whole-genome sequencing capacities, particularly in endemic regions, is crucial for ongoing surveillance of viral lineages, tracking APOBEC3 mutations, and monitoring potential antiviral resistance.
Additionally, there is an urgent need to enhance public awareness and understanding of Mpox transmission, treatment, and prevention strategies. This is especially vital for populations in low- and middle-income countries, where vaccination rates may be suboptimal and reliance on personal protective measures remains critical. Healthcare professionals must receive improved training focused on both Mpox diagnosis and management, supported by community-oriented, non-stigmatising communication strategies to mitigate misinformation within high-risk groups. Further empirical research, including statistical and meta-analytic analyses in Mpox hotspots, is essential for systematically evaluating existing control strategies, therapeutic interventions, and vaccine efficacy.
Lastly, the establishment of effective global coordination, along with prioritisation of pre-positioned vaccines and antiviral supplies for endemic countries, is imperative. This should include incorporating Mpox-specific guidelines into national pandemic preparedness frameworks and investing in long-term healthcare infrastructure based on a One Health approach that integrates human, animal, and environmental health surveillance systems. Such comprehensive strategies are crucial for reducing transmission and preventing future outbreaks.
5. Limitations of This Review
As this article was intended to be a narrative review, a systematic review approach was not employed to select appropriate studies as data sources. This approach may have led to the omission of information that could have been relevant to the review. Additionally, the absence of quantitative data and statistical analysis may have limited the conclusions that could be drawn from the review.
6. Conclusions
Mpox, caused by the zoonotic Mpox virus, has been reported worldwide, particularly in West and Central Africa, since its discovery in 1958. The virus gained global attention during the worldwide outbreak in 2022, which resulted in millions of clinically diagnosed cases and hundreds of deaths. Mpox spreads through close contact with infected animals or humans, and after infection, the virus travels through the lymphatic system, causing symptoms that range from mild skin lesions to severe, potentially fatal complications affecting multiple organ systems. Diagnosis involves several laboratory techniques, including polymerase chain reaction (PCR), enzyme-linked immunosorbent assay (ELISA), immunohistochemical methods, and rapid detection techniques utilising biosensor devices. Treatment primarily consists of supportive care along with antiviral agents such as cidofovir, brincidofovir, and tecovirimat. For prevention, several vaccines originally developed for smallpox, such as Dryvax, LC16m8, and IMVAMUNE (also known as JYNNEOS or IMVANEX), are currently in use. In addition, preventive strategies include routine surveillance, increased public awareness, accurate diagnosis, strengthening healthcare systems, and implementing updated guidelines for the control, treatment, and prevention of Mpox, all of which are essential to limit the spread and progression of the disease.
Author Contributions
Conceptualization, D.Z.I. and N.A. (Nihad Adnan); methodology, N.A. (Nihad Adnan) and D.Z.I.; software, M.S.R. and S.S.K.; writing—original draft preparation, D.Z.I., F.S.T., M.F., M.S.A.S., A.K., M.M.H., M.S.I.S. and S.S.K.; writing—review and editing, N.A. (Nihad Adnan), N.A. (Nafisa Azmuda), M.F.A., S.A., A.A.I.S., S.S.K. and D.Z.I.; visualization, D.Z.I. and S.S.K. and N.A. (Nihad Adnan); supervision, N.A. (Nihad Adnan) and A.A.I.S. All authors have agreed on the journal to which the article has been submitted, and decided to be accountable for all aspects of the work. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by SAGE Fund PS76828-UNSW School of Biotechnology & Biomolecular sciences.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
This article is a narrative review based exclusively on previously published literature. No new datasets were generated or analyzed during this study; therefore, data sharing is not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- WHO. Mpox (Monkeypox) Outbreak. Available online: https://www.who.int/emergency/situations/monkeypox-oubreak-2022 (accessed on 16 December 2025).
- Luna, N.; Muñoz, M.; Bonilla-Aldana, D.K.; Patiño, L.H.; Kasminskaya, Y.; Paniz-Mondolfi, A.; Ramírez, J.D. Monkeypox Virus (Mpxv) Genomics: A Mutational and Phylogenomic Analyses of B. 1 Lineages. Travel Med. Infect. Dis. 2023, 52, 102551. [Google Scholar] [CrossRef] [PubMed]
- Cuetos-Suárez, D.; Gan, R.K.; Cuetos-Suárez, D.; Arcos González, P.; Castro-Delgado, R. A Review of Mpox Outbreak and Public Health Response in Spain. Risk Manag. Healthc. Policy 2024, 17, 297–310. [Google Scholar] [CrossRef] [PubMed]
- WHO. Multi-Country Outbreak of Mpox, External Situation Report #51-29 April 2025. Available online: https://www.who.int/publications/m/item/multi-country-outbreak-of-mpox--external-situation-report--51---29-april-2025 (accessed on 1 December 2025).
- Yadav, R.; Chaudhary, A.A.; Srivastava, U.; Gupta, S.; Rustagi, S.; Rudayni, H.A.; Kashyap, V.K.; Kumar, S. Mpox 2022 to 2025 Update: A Comprehensive Review on Its Complications, Transmission, Diagnosis, and Treatment. Viruses 2025, 17, 753. [Google Scholar] [CrossRef] [PubMed]
- WHO. Fourth Meeting of the International Health Regulations (2005) Emergency Committee Regarding the Upsurge of Mpox 2024. Available online: https://www.who.int/news/item/10-07-2025-fourth-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-upsurge-of-mpox-2024 (accessed on 1 December 2025).
- Bunge, E.M.; Hoet, B.; Chen, L.; Lienert, F.; Weidenthaler, H.; Baer, L.R.; Steffen, R. The Changing Epidemiology of Human Monkeypox—A Potential Threat? A Systematic Review. PLoS Negl. Trop. Dis. 2022, 16, e0010141. [Google Scholar] [CrossRef]
- Petersen, B.W.; Kabamba, J.; McCollum, A.M.; Lushima, R.S.; Wemakoy, E.O.; Tamfum, J.-J.M.; Nguete, B.; Hughes, C.M.; Monroe, B.P.; Reynolds, M.G. Vaccinating Against Monkeypox in the Democratic Republic of the Congo. Antivir. Res. 2019, 162, 171–177. [Google Scholar] [CrossRef]
- Raccagni, A.R.; Candela, C.; Mileto, D.; Canetti, D.; Bruzzesi, E.; Rizzo, A.; Castagna, A.; Nozza, S. Monkeypox Infection among Men Who Have Sex with Men: Pcr Testing on Seminal Fluids. J. Infect. 2022, 85, 573. [Google Scholar] [CrossRef]
- Hoffmann, C.; Jessen, H.; Wyen, C.; Grunwald, S.; Noe, S.; Teichmann, J.; Krauss, A.S.; Kolarikal, H.; Scholten, S.; Schuler, C. Clinical Characteristics of Monkeypox Virus Infections Among Men with and Without Hiv: A Large Outbreak Cohort in Germany. HIV Med. 2023, 24, 389–397. [Google Scholar] [CrossRef]
- Vivancos-Gallego, M.J.; Sánchez-Conde, M.; Rodríguez-Domínguez, M.; Fernandez-Gonzalez, P.; Martínez-García, L.; Garcia-Mouronte, E.; Martínez-Sanz, J.; Moreno-Zamora, A.M.; Casado, J.L.; Ron, R. Human Monkeypox in People with Hiv: Transmission, Clinical Features, and Outcome. Open Forum Infect. Dis. 2022, 9, ofac557. [Google Scholar] [CrossRef]
- Orviz, E.; Negredo, A.; Ayerdi, O.; Vázquez, A.; Muñoz-Gomez, A.; Monzón, S.; Clavo, P.; Zaballos, A.; Vera, M.; Sánchez, P. Monkeypox Outbreak in Madrid (Spain): Clinical and Virological Aspects. J. Infect. 2022, 85, 412–417. [Google Scholar] [CrossRef]
- Patel, A.; Bilinska, J.; Tam, J.C.; Fontoura, D.D.S.; Mason, C.Y.; Daunt, A.; Snell, L.B.; Murphy, J.; Potter, J.; Tuudah, C. Clinical Features and Novel Presentations of Human Monkeypox in a Central London Centre During the 2022 Outbreak: Descriptive Case Series. BMJ 2022, 378, e072410. [Google Scholar] [CrossRef]
- Ogoina, D.; Damon, I.; Nakoune, E. Clinical Review of Human Mpox. Clin. Microbiol. Infect. 2023, 29, 1493–1501. [Google Scholar] [CrossRef] [PubMed]
- Soheili, M.; Nasseri, S.; Afraie, M.; Khateri, S.; Moradi, Y.; Mortazavi, S.M.M.; Gilzad-Kohan, H. Monkeypox: Virology, Pathophysiology, Clinical Characteristics, Epidemiology, Vaccines, Diagnosis, and Treatments. J. Pharm. Pharm. Sci. 2022, 25, 297–322. [Google Scholar] [CrossRef] [PubMed]
- Damon, I.K. Status of Human Monkeypox: Clinical Disease, Epidemiology and Research. Vaccine 2011, 29, D54–D59. [Google Scholar] [CrossRef] [PubMed]
- McCollum, A.M.; Damon, I.K. Human Monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Ophinni, Y.; Megawati, D.; Frediansyah, A.; Mamada, S.S.; Salampe, M.; Bin Emran, T.; Winardi, W.; Fathima, R.; Sirinam, S. Monkeypox: A Comprehensive Review. Viruses 2022, 14, 2155. [Google Scholar] [CrossRef]
- Wang, L.; Shang, J.; Weng, S.; Aliyari, S.R.; Ji, C.; Cheng, G.; Wu, A. Genomic Annotation and Molecular Evolution of Monkeypox Virus Outbreak in 2022. J. Med. Virol. 2023, 95, e28036. [Google Scholar] [CrossRef]
- WHO. Multi-Country Monkeypox Outbreak in Non-Endemic Countries. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON385 (accessed on 16 December 2025).
- Beer, E.M.; Rao, V.B. A Systematic Review of the Epidemiology of Human Monkeypox Outbreaks and Implications for Outbreak Strategy. PLoS Negl. Trop. Dis. 2019, 13, e0007791. [Google Scholar] [CrossRef]
- Hayat, C.; Shahab, M.; Khan, S.A.; Liang, C.; Duan, X.; Khan, H.; Zheng, G.; Ul-Haq, Z. Design of a Novel Multiple Epitope-Based Vaccine: An Immunoinformatics Approach to Combat Monkeypox. J. Biomol. Struct. Dyn. 2023, 41, 9344–9355. [Google Scholar] [CrossRef]
- Sallam, M.; Al-Mahzoum, K.; Al-Tammemi, A.a.B.; Alkurtas, M.; Mirzaei, F.; Kareem, N.; Al-Naimat, H.; Jardaneh, L.; Al-Majali, L.; AlHadidi, A. Assessing Healthcare Workers’ Knowledge and Their Confidence in the Diagnosis and Management of Human Monkeypox: A Cross-Sectional Study in a Middle Eastern Country. Healthcare 2022, 10, 1722. [Google Scholar] [CrossRef] [PubMed]
- Melendez-Rosado, J.; Thompson, K.M.; Cowdell, J.C.; Sanchez Alvarez, C.; Ung, R.L.; Villanueva, A.; Jeffers, K.B.; Imam, J.S.; Mitkov, M.V.; Kaleem, T.A. Reducing Unnecessary Testing: An Intervention to Improve Resident Ordering Practices. Postgrad. Med. J. 2017, 93, 476–479. [Google Scholar] [CrossRef] [PubMed]
- Dumonteil, E.; Herrera, C.; Sabino-Santos, G. Monkeypox Virus Evolution before 2022 Outbreak. Emerg. Infect. Dis. 2023, 29, 451. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhang, X.; Liu, J.; Xiang, L.; Huang, S.; Xie, X.; Fang, L.; Lin, Y.; Zhang, M.; Wang, L. Phylogeny and Molecular Evolution of the First Local Monkeypox Virus Cluster in Guangdong Province, China. Nat. Commun. 2023, 14, 8241. [Google Scholar] [CrossRef] [PubMed]
- Scarpa, F.; Azzena, I.; Ciccozzi, A.; Branda, F.; Locci, C.; Perra, M.; Pascale, N.; Romano, C.; Ceccarelli, G.; Terrazzano, G. Update of the Genetic Variability of Monkeypox Virus Clade Iib Lineage B. 1. Microorganisms 2024, 12, 1874. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Shi, H.; Cheng, G. Mpox Virus: Its Molecular Evolution and Potential Impact on Viral Epidemiology. Viruses 2023, 15, 995. [Google Scholar] [CrossRef]
- WHO. Mpox. Available online: https://www.who.int/news-room/fact-sheets/detail/mpox (accessed on 16 December 2025).
- CDC. Mpox in the United States and around the World: Current Situation. Available online: https://www.cdc.gov/mpox/situation-summary/index.html (accessed on 10 July 2025).
- Isidro, J.; Borges, V.; Pinto, M.; Sobral, D.; Santos, J.D.; Nunes, A.; Mixão, V.; Ferreira, R.; Santos, D.; Duarte, S. Phylogenomic Characterization and Signs of Microevolution in the 2022 Multi-Country Outbreak of Monkeypox Virus. Nat. Med. 2022, 28, 1569–1572. [Google Scholar] [CrossRef]
- Lu, J.; Xing, H.; Wang, C.; Tang, M.; Wu, C.; Ye, F.; Yin, L.; Yang, Y.; Tan, W.; Shen, L. Mpox (Formerly Monkeypox): Pathogenesis, Prevention, and Treatment. Signal Transduct. Target. Ther. 2023, 8, 458. [Google Scholar] [CrossRef]
- Nakweya, G. Mpox Virus Evolving Due to Sustained Human Transmission. Available online: https://www.nature.com/articles/d44148-023-00318-6 (accessed on 1 December 2025).
- Karagoz, A.; Tombuloglu, H.; Alsaeed, M.; Tombuloglu, G.; AlRubaish, A.A.; Mahmoud, A.; Smajlović, S.; Ćordić, S.; Rabaan, A.A.; Alsuhaimi, E. Monkeypox (Mpox) Virus: Classification, Origin, Transmission, Genome Organization, Antiviral Drugs, and Molecular Diagnosis. J. Infect. Public Health 2023, 16, 531–541. [Google Scholar] [CrossRef]
- Monzón, S.; Varona, S.; Negredo, A.; Vidal-Freire, S.; Patiño-Galindo, J.A.; Ferressini-Gerpe, N.; Zaballos, A.; Orviz, E.; Ayerdi, O.; Muñoz-Gómez, A. Monkeypox Virus Genomic Accordion Strategies. Nat. Commun. 2024, 15, 3059. [Google Scholar] [CrossRef]
- Kugelman, J.R.; Johnston, S.C.; Mulembakani, P.M.; Kisalu, N.; Lee, M.S.; Koroleva, G.; McCarthy, S.E.; Gestole, M.C.; Wolfe, N.D.; Fair, J.N.; et al. Genomic Variability of Monkeypox Virus Among Humans, Democratic Republic of the Congo. Emerg. Infect. Dis. 2014, 20, 232. [Google Scholar] [CrossRef]
- Di Giulio, D.B.; Eckburg, P.B. Human Monkeypox: An Emerging Zoonosis. Lancet Infect. Dis. 2004, 4, 15–25. [Google Scholar] [CrossRef]
- Berthet, N.; Descorps-Declère, S.; Besombes, C.; Curaudeau, M.; Nkili Meyong, A.A.; Selekon, B.; Labouba, I.; Gonofio, E.C.; Ouilibona, R.S.; Simo Tchetgna, H.D. Genomic History of Human Monkey Pox Infections in the Central African Republic between 2001 and 2018. Sci. Rep. 2021, 11, 13085. [Google Scholar] [CrossRef]
- Xu, Y.; Wu, Y.; Zhang, Y.; Gao, K.; Wu, X.; Yang, Y.; Li, D.; Yang, B.; Zhang, Z.; Dong, C. Essential and Multifunctional Mpox Virus E5 Helicase-Primase in Double and Single Hexamer. Sci. Adv. 2024, 10, eadl1150. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Kuai, L.; Peng, Q.; Wang, Q.; Wang, H.; Li, X.; Qi, J.; Ding, Q.; Shi, Y.; Gao, G.F. Structural Basis of DNA Replication Fidelity of the Mpox Virus. Proc. Natl. Acad. Sci. USA 2025, 122, e2411686122. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; He, P.; Song, Y.; Cheng, S.; Linhardt, R.J.; Dordick, J.S.; Chi, L.; Zhang, F. Kinetic and Structural Aspects of Glycosaminoglycan–Monkeypox Virus Protein A29 Interactions Using Surface Plasmon Resonance. Molecules 2022, 27, 5898. [Google Scholar] [CrossRef] [PubMed]
- Sagdat, K.; Batyrkhan, A.; Kanayeva, D. Exploring Monkeypox Virus Proteins and Rapid Detection Techniques. Front. Cell. Infect. Microbiol. 2024, 14, 1414224. [Google Scholar] [CrossRef]
- Liang, C.-Y.; Chao, T.-L.; Chao, C.S.; Liu, W.-D.; Cheng, Y.-C.; Chang, S.-Y.; Chang, S.-C. Monkeypox Virus A29l Protein as the Target for Specific Diagnosis and Serological Analysis. Appl. Microbiol. Biotechnol. 2024, 108, 522. [Google Scholar] [CrossRef]
- Rehm, K.E. The Poxvirus A35 Protein Promotes Virulence by Regulating the Host Adaptive Immune Response; East Carolina University: Greenville, NC, USA, 2010. [Google Scholar]
- von Magnus, P.; Andersen, E.K.; Petersen, K.B.; Birch Andersen, A. A Pox-Like Disease in Cynomolgus Monkeys. Acta Pathol. Microbiol. Scand. 1959, 46, 156–176. [Google Scholar] [CrossRef]
- Arita, I.; Henderson, D. Smallpox and Monkeypox in Non-Human Primates. Bull. World Health Organ. 1968, 39, 277. [Google Scholar]
- Arita, I.; Gispen, R.; Kalter, S.; Wah, L.T.; Marennikova, S.; Netter, R.; Tagaya, I. Outbreaks of Monkeypox and Serological Surveys in Nonhuman Primates. Bull. World Health Organ. 1972, 46, 625. [Google Scholar]
- Durski, K.N. Emergence of Monkeypox—West and Central Africa, 1970–2017. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 306–310, Erratum in MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 479. [Google Scholar] [CrossRef] [PubMed]
- Osadebe, L.; Hughes, C.M.; Shongo Lushima, R.; Kabamba, J.; Nguete, B.; Malekani, J.; Pukuta, E.; Karhemere, S.; Muyembe Tamfum, J.-J.; Wemakoy Okitolonda, E. Enhancing Case Definitions for Surveillance of Human Monkeypox in the Democratic Republic of Congo. PLoS Negl. Trop. Dis. 2017, 11, e0005857. [Google Scholar] [CrossRef] [PubMed]
- Ladnyj, I.; Ziegler, P.; Kima, E. A Human Infection Caused by Monkeypox Virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593. [Google Scholar]
- Cho, C.T.; Wenner, H.A. Monkeypox Virus. Bacteriol. Rev. 1973, 37, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, S.; Bondy, L.; Hanage, W.P. Monkeypox Virus Infections in Humans. Clin. Microbiol. Rev. 2022, 35, e0009222. [Google Scholar] [CrossRef] [PubMed]
- Rimoin, A.W.; Mulembakani, P.M.; Johnston, S.C.; Lloyd Smith, J.O.; Kisalu, N.K.; Kinkela, T.L.; Blumberg, S.; Thomassen, H.A.; Pike, B.L.; Fair, J.N. Major Increase in Human Monkeypox Incidence 30 Years After Smallpox Vaccination Campaigns Cease in the Democratic Republic of Congo. Proc. Natl. Acad. Sci. USA 2010, 107, 16262–16267. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Damon, I.K. Outbreaks of Human Monkeypox After Cessation of Smallpox Vaccination. Trends Microbiol. 2012, 20, 80–87. [Google Scholar] [CrossRef]
- Jezek, Z.; Khodakevich, L.; Wickett, J.F. Smallpox and Its Post-Eradication Surveillance. Bull. World Health Organ. 1987, 65, 425. [Google Scholar]
- Heymann, D.L.; Szczeniowski, M.; Esteves, K. Re-Emergence of Monkeypox in Africa: A Review of the Past Six Years. Br. Med. Bull. 1998, 54, 693–702. [Google Scholar] [CrossRef]
- Sklenovská, N.; Van Ranst, M. Emergence of Monkeypox as the Most Important Orthopoxvirus Infection in Humans. Front. Public Health 2018, 6, 383729. [Google Scholar] [CrossRef]
- Obi, C.L.; Mkolo, N.M.; Mugivhisa, L.L.; Ogunrombi, M.O.; Mphephu, M.M.; Naidoo, C.M. Mpox: An Emerging or Re-Emerging Infection with a Potential Colossal Burden on Healthcare Globally. Afr. J. Lab. Med. 2025, 14, 2644. [Google Scholar] [CrossRef]
- Hutin, Y.; Williams, R.J.; Malfait, P.; Pebody, R.; Loparev, V.N.; Ropp, S.L.; Rodriguez, M.; Knight, J.C.; Tshioko, F.K.; Khan, A.S. Outbreak of Human Monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg. Infect. Dis. 2001, 7, 434. [Google Scholar] [CrossRef]
- Huang, Y.; Mu, L.; Wang, W. Monkeypox: Epidemiology, Pathogenesis, Treatment and Prevention. Signal Transduct. Target. Ther. 2022, 7, 373. [Google Scholar] [CrossRef] [PubMed]
- Yinka-Ogunleye, A.; Aruna, O.; Dalhat, M.; Ogoina, D.; McCollum, A.; Disu, Y.; Mamadu, I.; Akinpelu, A.; Ahmad, A.; Burga, J. Outbreak of Human Monkeypox in Nigeria in 2017–18: A Clinical and Epidemiological Report. Lancet Infect. Dis. 2019, 19, 872–879. [Google Scholar] [CrossRef] [PubMed]
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef]
- Ogoina, D.; Iroezindu, M.; James, H.I.; Oladokun, R.; Yinka-Ogunleye, A.; Wakama, P.; Otike-Odibi, B.; Usman, L.M.; Obazee, E.; Aruna, O. Clinical Course and Outcome of Human Monkeypox in Nigeria. Clin. Infect. Dis. 2020, 71, e210–e214. [Google Scholar] [CrossRef]
- Farahat, R.A.; Sah, R.; El-Sakka, A.A.; Benmelouka, A.Y.; Kundu, M.; Labieb, F.; Shaheen, R.S.; Abdelaal, A.; Abdelazeem, B.; Bonilla-Aldana, D.K. Human Monkeypox Disease (Mpx). Infez. Med. 2022, 30, 372. [Google Scholar]
- CDC, MMWR. Multistate Outbreak of Monkeypox—Illinois, Indiana, and Wisconsin, 2003. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5223a1.htm (accessed on 16 December 2025).
- Update: Multistate Outbreak of Monkeypox—Illinois, Indiana, Kansas, Missouri, Ohio, and Wisconsin, 2003. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5224a1.htm (accessed on 16 December 2025).
- Xiang, Y.; White, A. Monkeypox Virus Emerges from the Shadow of Its More Infamous Cousin: Family Biology Matters. Emerg. Microbes Infect. 2022, 11, 1768–1777. [Google Scholar] [CrossRef]
- Vaughan, A.; Aarons, E.; Astbury, J.; Brooks, T.; Chand, M.; Flegg, P.; Hardman, A.; Harper, N.; Jarvis, R.; Mawdsley, S. Human-to-Human Transmission of Monkeypox Virus, United Kingdom, October 2018. Emerg. Infect. Dis. 2020, 26, 782. [Google Scholar] [CrossRef]
- Ng, O.T.; Lee, V.; Marimuthu, K.; Vasoo, S.; Chan, G.; Lin, R.T.P.; Leo, Y.S. A Case of Imported Monkeypox in Singapore. Lancet Infect. Dis. 2019, 19, 1166. [Google Scholar] [CrossRef]
- Costello, V.; Sowash, M.; Gaur, A.; Cardis, M.; Pasieka, H.; Wortmann, G.; Ramdeen, S. Imported Monkeypox from International Traveler, Maryland, USA, 2021. Emerg. Infect. Dis. 2022, 28, 1002. [Google Scholar]
- Erez, N.; Achdout, H.; Milrot, E.; Schwartz, Y.; Wiener-Well, Y.; Paran, N.; Politi, B.; Tamir, H.; Israely, T.; Weiss, S. Diagnosis of Imported Monkeypox, Israel, 2018. Emerg. Infect. Dis. 2019, 25, 980. [Google Scholar] [CrossRef] [PubMed]
- Mauldin, M.R.; McCollum, A.M.; Nakazawa, Y.J.; Mandra, A.; Whitehouse, E.R.; Davidson, W.; Zhao, H.; Gao, J.; Li, Y.; Doty, J. Exportation of Monkeypox Virus from the African Continent. J. Infect. Dis. 2022, 225, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- WHO. Monkeypox-United Kingdom of Great Britain and Northern Ireland. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON381 (accessed on 16 December 2025).
- Gessain, A.; Nakoune, E.; Yazdanpanah, Y. Monkeypox. N. Engl. J. Med. 2022, 387, 1783–1793. [Google Scholar] [CrossRef] [PubMed]
- Vivancos, R.; Anderson, C.; Blomquist, P.; Balasegaram, S.; Bell, A.; Bishop, L.; Brown, C.S.; Chow, Y.; Edeghere, O.; Florence, I. Community Transmission of Monkeypox in the United Kingdom, April to May 2022. Eurosurveillance 2022, 27, 2200422, Erratum in Eurosurveillance 2022, 27, 220609c. [Google Scholar] [CrossRef]
- CDC. Ongoing 2022 Global Outbreak Cases and Data. Available online: https://archive.cdc.gov/#/details?archive_url=https://archive.cdc.gov/www_cdc_gov/poxvirus/mpox/response/2022/index.html (accessed on 16 December 2025).
- Thornhill, J.P.; Barkati, S.; Walmsley, S.; Rockstroh, J.; Antinori, A.; Harrison, L.B.; Palich, R.; Nori, A.; Reeves, I.; Habibi, M.S. Monkeypox Virus Infection in Humans across 16 Countries—April–June 2022. N. Engl. J. Med. 2022, 387, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Noe, S.; Zange, S.; Seilmaier, M.; Antwerpen, M.H.; Fenzl, T.; Schneider, J.; Spinner, C.D.; Bugert, J.J.; Wendtner, C.-M.; Wölfel, R. Clinical and Virological Features of First Human Monkeypox Cases in Germany. Infection 2023, 51, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Antinori, A.; Mazzotta, V.; Vita, S.; Carletti, F.; Tacconi, D.; Lapini, L.E.; D’Abramo, A.; Cicalini, S.; Lapa, D.; Pittalis, S. Epidemiological, Clinical and Virological Characteristics of Four Cases of Monkeypox Support Transmission through Sexual Contact, Italy, May 2022. Eurosurveillance 2022, 27, 2200421. [Google Scholar] [CrossRef]
- Heskin, J.; Belfield, A.; Milne, C.; Brown, N.; Walters, Y.; Scott, C.; Bracchi, M.; Moore, L.S.; Mughal, N.; Rampling, T. Transmission of Monkeypox Virus Through Sexual Contact—A Novel Route of Infection. J. Infect. 2022, 85, 334–363. [Google Scholar] [CrossRef]
- WHO. 2022-24 Mpox (Monkeypox) Outbreak: Global Trends. Available online: https://worldhealthorg.shinyapps.io/mpx_global/ (accessed on 16 December 2025).
- Harris, E. What to Know About Monkeypox. JAMA 2022, 327, 2278–2279. [Google Scholar] [CrossRef]
- Li, K.; Yuan, Y.; Jiang, L.; Liu, Y.; Liu, Y.; Zhang, L. Animal Host Range of Mpox Virus. J. Med. Virol. 2023, 95, e28513. [Google Scholar] [CrossRef]
- Alakunle, E.; Kolawole, D.; Diaz-Canova, D.; Alele, F.; Adegboye, O.; Moens, U.; Okeke, M.I. A Comprehensive Review of Monkeypox Virus and Mpox Characteristics. Front. Cell. Infect. Microbiol. 2024, 14, 1360586. [Google Scholar] [CrossRef] [PubMed]
- Lum, F.-M.; Torres-Ruesta, A.; Tay, M.Z.; Lin, R.T.; Lye, D.C.; Rénia, L.; Ng, L.F. Monkeypox: Disease Epidemiology, Host Immunity and Clinical Interventions. Nat. Rev. Immunol. 2022, 22, 597–613. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.; Kantele, A.; Koopmans, M.; Asogun, D.; Yinka-Ogunleye, A.; Ihekweazu, C.; Zumla, A. Human Monkeypox: Epidemiologic and Clinical Characteristics, Diagnosis, and Prevention. Infect. Dis. Clin. 2019, 33, 1027–1043. [Google Scholar]
- Kaler, J.; Hussain, A.; Flores, G.; Kheiri, S.; Desrosiers, D. Monkeypox: A Comprehensive Review of Transmission, Pathogenesis, and Manifestation. Cureus 2022, 14, e26531. [Google Scholar] [CrossRef]
- Wang, Y.; Leng, P.; Zhou, H. Global Transmission of Monkeypox Virus—A Potential Threat under the COVID-19 Pandemic. Front. Immunol. 2023, 14, 1174223, Erratum in Front. Immunol. 2025, 16, 1592351. [Google Scholar] [CrossRef]
- Kumar, P.; Chaudhary, B.; Yadav, N.; Devi, S.; Pareek, A.; Alla, S.; Kajal, F.; Nowrouzi-Kia, B.; Chattu, V.K.; Gupta, M.M. Recent Advances in Research and Management of Human Monkeypox Virus: An Emerging Global Health Threat. Viruses 2023, 15, 937. [Google Scholar] [CrossRef]
- Beeson, A.; Styczynski, A.; Hutson, C.L.; Whitehill, F.; Angelo, K.M.; Minhaj, F.S.; Morgan, C.; Ciampaglio, K.; Reynolds, M.G.; McCollum, A.M. Mpox Respiratory Transmission: The State of the Evidence. Lancet Microbe 2023, 4, e277–e283. [Google Scholar] [CrossRef]
- Walter, K.; Malani, P.N. What Is Monkeypox? JAMA 2022, 328, 222. [Google Scholar] [CrossRef]
- Simpson, K.; Heymann, D.; Brown, C.S.; Edmunds, W.J.; Elsgaard, J.; Fine, P.; Hochrein, H.; Hoff, N.A.; Green, A.; Ihekweazu, C. Human Monkeypox–After 40 Years, an Unintended Consequence of Smallpox Eradication. Vaccine 2020, 38, 5077–5081. [Google Scholar] [CrossRef]
- O’Neill, M.; LePage, T.; Bester, V.; Yoon, H.; Browne, F.; Nemec, E.C. Mpox (Formally Known as Monkeypox). Physician Assist. Clin. 2023, 8, 483. [Google Scholar] [CrossRef]
- Guarner, J.; Del Rio, C.; Malani, P.N. Monkeypox in 2022—What Clinicians Need to Know. JAMA 2022, 328, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Sah, R.; Abdelaal, A.; Reda, A.; Katamesh, B.E.; Manirambona, E.; Abdelmonem, H.; Mehta, R.; Rabaan, A.A.; Alhumaid, S.; Alfouzan, W.A. Monkeypox and Its Possible Sexual Transmission: Where Are We Now with Its Evidence? Pathogens 2022, 11, 924. [Google Scholar] [CrossRef] [PubMed]
- Baud, D.; Nielsen-Saines, K.; Mattar, C.; Musso, D.; Tambyah, P.; Dashraath, P. Monkeypox Exposure During Pregnancy: What Does Uk Public Health Guidance Advise?—Authors’ Reply. Lancet 2022, 400, 1509–1510. [Google Scholar] [CrossRef]
- Khalil, A.; Samara, A.; O’Brien, P.; Morris, E.; Draycott, T.; Lees, C.; Ladhani, S. Monkeypox and Pregnancy: What Do Obstetricians Need to Know? Ultrasound Obs. Gynecol. 2022, 60, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Allan-Blitz, L.-T.; Gandhi, M.; Adamson, P.; Park, I.; Bolan, G.; Klausner, J.D. A Position Statement on Mpox as a Sexually Transmitted Disease. Clin. Infect. Dis. 2023, 76, 1508–1512. [Google Scholar] [CrossRef]
- Saied, A.A.; Dhawan, M.; Metwally, A.A.; Fahrni, M.L.; Choudhary, P.; Choudhary, O.P. Disease History, Pathogenesis, Diagnostics, and Therapeutics for Human Monkeypox Disease: A Comprehensive Review. Vaccines 2022, 10, 2091. [Google Scholar] [CrossRef]
- Kumar, N.; Acharya, A.; Gendelman, H.E.; Byrareddy, S.N. The 2022 Outbreak and the Pathobiology of the Monkeypox Virus. J. Autoimmun. 2022, 131, 102855. [Google Scholar] [CrossRef]
- Kmiec, D.; Kirchhoff, F. Monkeypox: A New Threat? Int. J. Mol. Sci. 2022, 23, 7866. [Google Scholar] [CrossRef] [PubMed]
- Upadhayay, S.; Arthur, R.; Soni, D.; Yadav, P.; Navik, U.; Singh, R.; Singh, T.G.; Kumar, P. Monkeypox Infection: The Past, Present, and Future. Int. Immunopharmacol. 2022, 113, 109382. [Google Scholar] [CrossRef] [PubMed]
- Hudson, P.N.; Self, J.; Weiss, S.; Braden, Z.; Xiao, Y.; Girgis, N.M.; Emerson, G.; Hughes, C.; Sammons, S.A.; Isaacs, S.N. Elucidating the Role of the Complement Control Protein in Monkeypox Pathogenicity. PLoS ONE 2012, 7, e35086. [Google Scholar] [CrossRef] [PubMed]
- Moss, B. Poxvirus Cell Entry: How Many Proteins Does It Take? Viruses 2012, 4, 688–707. [Google Scholar] [CrossRef]
- Wang, X.; Lun, W. Skin Manifestation of Human Monkeypox. J. Clin. Med. 2023, 12, 914. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Ahmed, A.; Ahsan, O.; Muhammad, K.; Waheed, Y. Monkeypox Virus: A Comprehensive Overview of Viral Pathology, Immune Response, and Antiviral Strategies. Vaccines 2023, 11, 1345. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; McCollum, A.M.; Nguete, B.; Shongo Lushima, R.; Petersen, B.W. Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research. Viruses 2017, 9, 380. [Google Scholar] [CrossRef] [PubMed]
- Chenchula, S.; Ghanta, M.K.; Chavan, M.; Amerneni, K.C.; Padmavathi, R.; Gupta, R. Novel Clinical Manifestations of Human Monkeypox Virus Infection and Current Therapeutic and Preventive Strategies: A Systematic Review. medRxiv 2023. [Google Scholar] [CrossRef]
- Anwar, F.; Haider, F.; Khan, S.; Ahmad, I.; Ahmed, N.; Imran, M.; Rashid, S.; Ren, Z.-G.; Khattak, S.; Ji, X.-Y. Clinical Manifestation, Transmission, Pathogenesis, and Diagnosis of Monkeypox Virus: A Comprehensive Review. Life 2023, 13, 522. [Google Scholar] [CrossRef]
- Babu, A.; Nair, C.V.; Moni, M.; Sathyapalan, D.T. Re-Emergence of Monkeypox: Changing Epidemiology, Clinical Manifestations, and Preventive Strategies. Amrita J. Med. 2023, 19, 2–8. [Google Scholar] [CrossRef]
- Shuvo, P.A.; Roy, A.; Dhawan, M.; Chopra, H.; Emran, T.B. Recent Outbreak of Monkeypox: Overview of Signs, Symptoms, Preventive Measures, and Guideline for Supportive Management. Int. J. Surg. 2022, 105, 106877. [Google Scholar] [CrossRef]
- Sharma, A.; Prasad, H.; Kaeley, N.; Bondalapati, A.; Edara, L.; Kumar, Y.A. Monkeypox Epidemiology, Clinical Presentation, and Transmission: A Systematic Review. Int. J. Emerg. Med. 2023, 16, 20. [Google Scholar] [CrossRef]
- Altindis, M.; Puca, E.; Shapo, L. Diagnosis of Monkeypox Virus—An Overview. Travel Med. Infect. Dis. 2022, 50, 102459. [Google Scholar] [CrossRef]
- Hatmal, M.m.M.; Al-Hatamleh, M.A.; Olaimat, A.N.; Ahmad, S.; Hasan, H.; Ahmad Suhaimi, N.A.; Albakri, K.A.; Abedalbaset Alzyoud, A.; Kadir, R.; Mohamud, R. Comprehensive Literature Review of Monkeypox. Emerg. Microbes Infect. 2022, 11, 2600–2631. [Google Scholar] [CrossRef] [PubMed]
- Bryer, J.; Freeman, E.E.; Rosenbach, M. Monkeypox Emerges on a Global Scale: A Historical Review and Dermatologic Primer. J. Am. Acad. Dermatol. 2022, 87, 1069–1074. [Google Scholar] [CrossRef] [PubMed]
- Cheema, A.Y.; Ogedegbe, O.J.; Munir, M.; Alugba, G.; Ojo, T.K. Monkeypox: A Review of Clinical Features, Diagnosis, and Treatment. Cureus 2022, 14, e26756. [Google Scholar] [CrossRef] [PubMed]
- Zong, Y.; Kamoi, K.; Zhang, J.; Yang, M.; Ohno-Matsui, K. Mpox (Monkeypox) and the Eye: Ocular Manifestation, Diagnosis, Treatment and Vaccination. Viruses 2023, 15, 616. [Google Scholar] [CrossRef] [PubMed]
- Lucena-Neto, F.D.; Falcão, L.F.; Vieira-Junior, A.S.; Moraes, E.C.; David, J.P.; Silva, C.C.; Sousa, J.R.; Duarte, M.I.; Vasconcelos, P.F.; Quaresma, J.A. Monkeypox Virus Immune Evasion and Eye Manifestation: Beyond Eyelid Implications. Viruses 2023, 15, 2301. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, A.R.; Chodosh, J.; Pineda, R. Monkeypox Virus and Ophthalmology—A Primer on the 2022 Monkeypox Outbreak and Monkeypox-Related Ophthalmic Disease. JAMA Ophthalmol. 2023, 141, 78–83, Erratum in JAMA Ophthalmol. 2023, 141, 104. https://doi.org/10.1001/jamaophthalmol.2022.6121. [Google Scholar] [CrossRef]
- Chakravarty, N.; Hemani, D.; Paravastu, R.; Ahmad, Z.; Palani, S.N.; Arumugaswami, V.; Kumar, A. Mpox Virus and Its Ocular Surface Manifestations. Ocul. Surf. 2024, 34, 108–121. [Google Scholar] [CrossRef]
- Jaiswal, V.; Sultana, Q.; Lahori, S.; Mukherjee, D.; Agrawal, V.; Doshi, N.; Shrestha, A.B.; Huang, H.; Nasir, Y.M.; Naz, S. Monkeypox-Induced Myocarditis: A Systematic Review. Curr. Probl. Cardiol. 2023, 48, 101611. [Google Scholar] [CrossRef]
- El-Qushayri, A.E.; Tawfik, A.G.; Mahmoud-Elsayed, H. Cardiovascular Manifestations of Monkeypox Virus Outbreak: An Overview of the Reported Cases. Heart Lung 2023, 59, 67–72. [Google Scholar] [CrossRef]
- Maqbool, K.U.; Arsh, H.; Kumar, D.; Veena, F.; Punshi, A.K.; Payal, F.; Kumar, S.; Kumar, S.; Rani, D.; Malik, J. Cardiovascular Manifestations of Human Monkeypox Virus: An Updated Review. Curr. Probl. Cardiol. 2023, 48, 101869. [Google Scholar] [CrossRef]
- Shaik, T.A.; Voloshyna, D.; Nasr, T.H.; Makki, A.; Kosuru, S.H.; Khan, M.H.; Ghobriel, N.G.; Sandhu, Q.I.; Saleem, F. Monkeypox-Associated Pericarditis: A Maiden Case. Cureus 2022, 14, e29638. [Google Scholar] [CrossRef] [PubMed]
- Maqbool, K.U.; Akhtar, M.T.; Ayub, S.; Simran, F.; Malik, J.; Malik, M.; Zubair, R.; Mehmoodi, A. Role of Vaccination in Patients with Human Monkeypox Virus and Its Cardiovascular Manifestations. Ann. Med. Surg. 2024, 86, 1506–1516. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, R.; Shenoy, A.; Madhavan, R.; Meyer, D. Mpox Gastrointestinal Manifestations: A Systematic Review. BMJ Open Gastroenterol. 2024, 11, e001266. [Google Scholar] [CrossRef] [PubMed]
- Simadibrata, D.M.; Lesmana, E.; Pratama, M.I.A.; Annisa, N.G.; Thenedi, K.; Simadibrata, M. Gastrointestinal Symptoms of Monkeypox Infection: A Systematic Review and Meta-Analysis. J. Med. Virol. 2023, 95, e28709. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Yorita, K.L.; Kuehnert, M.J.; Davidson, W.B.; Huhn, G.D.; Holman, R.C.; Damon, I.K. Clinical Manifestations of Human Monkeypox Influenced by Route of Infection. J. Infect. Dis. 2006, 194, 773–780. [Google Scholar] [CrossRef]
- Ghaseminia, M. Preventing Monkeypox Outbreaks: Focus on Diagnosis, Care, Treatment, and Vaccination. J. Clin. Transl. Sci. 2023, 7, e60. [Google Scholar] [CrossRef]
- Subbaram, K.; Naher, Z.U.; Faiz, R.; Huda, A.; Manandhar, P.L.; Ali, S.; Tazerji, S.S.; Duarte, P.M. Re-Emerging Disease: Detection and Laboratory Diagnosis of Monkeypox Virus—A Narrative Review. J. Basic Clin. Microbiol. 2024, 1, 19. [Google Scholar] [CrossRef]
- He, S.; Zhao, J.; Chen, J.; Liang, J.; Hu, X.; Zhang, X.; Zeng, H.; Sun, G. Urogenital Manifestations in Mpox (Monkeypox) Infection: A Comprehensive Review of Epidemiology, Pathogenesis, and Therapeutic Approaches. Infect. Drug Resist. 2025, 18, 209–226. [Google Scholar] [CrossRef]
- Licheri, M.; Licheri, M.F.; Probst, L.; Sägesser, C.; Bittel, P.; Suter-Riniker, F.; Dijkman, R. A Novel Isothermal Whole Genome Sequencing Approach for Monkeypox Virus. Sci. Rep. 2024, 14, 22333, Erratum in Sci. Rep. 2025, 15, 4346. [Google Scholar] [CrossRef]
- Ferdous, J.; Barek, M.A.; Hossen, M.S.; Bhowmik, K.K.; Islam, M.S. A Review on Monkeypox Virus Outbreak: New Challenge for World. Health Sci. Rep. 2023, 6, e1007. [Google Scholar] [CrossRef] [PubMed]
- Khattak, S.; Rauf, M.A.; Ali, Y.; Yousaf, M.T.; Liu, Z.; Wu, D.-D.; Ji, X.-Y. The Monkeypox Diagnosis, Treatments and Prevention: A Review. Front. Cell. Infect. Microbiol. 2023, 12, 1088471. [Google Scholar] [CrossRef] [PubMed]
- Burnette, W.N. “Western Blotting”: Electrophoretic Transfer of Proteins from Sodium Dodecyl Sulfate-Polyacrylamide Gels to Unmodified Nitrocellulose and Radiographic Detection with Antibody and Radioiodinated Protein A. Anal. Biochem. 1981, 112, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Farlow, J.; Ichou, M.A.; Huggins, J.; Ibrahim, S. Comparative Whole Genome Sequence Analysis of Wild-Type and Cidofovir-Resistant Monkeypoxvirus. Virol. J. 2010, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Sajal, S.S.A.; Islam, D.Z.; Khandker, S.S.; Solórzano-Ortiz, E.; Fardoun, M.; Ahmed, M.F.; Jamiruddin, M.R.; Azmuda, N.; Mehta, M.; Kumar, S. Strategies to Overcome Erroneous Outcomes in Reverse Transcription-Polymerase Chain Reaction (Rt-Pcr) Testing: Insights from the COVID-19 Pandemic. Cureus 2024, 16, e72954. [Google Scholar]
- Li, Y.; Zhao, H.; Wilkins, K.; Hughes, C.; Damon, I.K. Real-Time Pcr Assays for the Specific Detection of Monkeypox Virus West African and Congo Basin Strain DNA. J. Virol. Methods 2010, 169, 223–227. [Google Scholar] [CrossRef]
- Li, Y.; Olson, V.A.; Laue, T.; Laker, M.T.; Damon, I.K. Detection of Monkeypox Virus with Real-Time Pcr Assays. J. Clin. Virol. 2006, 36, 194–203. [Google Scholar] [CrossRef]
- Müller, M.; Ingold-Heppner, B.; Stocker, H.; Heppner, F.L.; Dittmayer, C.; Laue, M. Electron Microscopy Images of Monkeypox Virus Infection in 24-Year-Old Man. Lancet 2022, 400, 1618. [Google Scholar] [CrossRef]
- Gentile, M.; Gelderblom, H.R. Rapid Viral Diagnosis: Role of Electron Microscopy. New Microbiol. 2005, 28, 1–12. [Google Scholar]
- Gelderblom, H.R.; Madeley, D. Rapid Viral Diagnosis of Orthopoxviruses by Electron Microscopy: Optional or a Must? Viruses 2018, 10, 142. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, Z. Mpox: A Review of Laboratory Detection Techniques. Arch. Virol. 2023, 168, 221. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Gul, I.; Liu, C.; Lei, Z.; Li, X.; Raheem, M.A.; He, Q.; Haihui, Z.; Leeansyah, E.; Zhang, C.Y. Crispr–Cas12-Based Field-Deployable System for Rapid Detection of Synthetic DNA Sequence of the Monkeypox Virus Genome. J. Med. Virol. 2023, 95, e28385. [Google Scholar] [CrossRef]
- Mao, L.; Ying, J.; Selekon, B.; Gonofio, E.; Wang, X.; Nakoune, E.; Wong, G.; Berthet, N. Development and Characterization of Recombinase-Based Isothermal Amplification Assays (Rpa/Raa) for the Rapid Detection of Monkeypox Virus. Viruses 2022, 14, 2112. [Google Scholar] [CrossRef]
- da Silva, S.J.R.; Kohl, A.; Pena, L.; Pardee, K. Clinical and Laboratory Diagnosis of Monkeypox (Mpox): Current Status and Future Directions. iScience 2023, 26, 106759. [Google Scholar] [CrossRef]
- Becherer, L.; Borst, N.; Bakheit, M.; Frischmann, S.; Zengerle, R.; von Stetten, F. Loop-Mediated Isothermal Amplification (Lamp)–Review and Classification of Methods for Sequence-Specific Detection. Anal. Methods 2020, 12, 717–746. [Google Scholar] [CrossRef]
- Stefano, J.S.; e Silva, L.R.G.; Kalinke, C.; de Oliveira, P.R.; Crapnell, R.D.; Brazaca, L.C.; Bonacin, J.A.; Campuzano, S.; Banks, C.E.; Janegitz, B.C. Human Monkeypox Virus: Detection Methods and Perspectives for Diagnostics. TrAC Trends Anal. Chem. 2023, 167, 117226. [Google Scholar] [CrossRef]
- Fan, G.; Kuang, J.; Zhang, S.; Yang, Y.; Liu, Y.; Lu, H. Diagnostic Approaches for Monkeypox Virus. iLABMED 2024, 2, 6–13. [Google Scholar] [CrossRef]
- Davi, S.D.; Kissenkötter, J.; Faye, M.; Böhlken-Fascher, S.; Stahl-Hennig, C.; Faye, O.; Faye, O.; Sall, A.A.; Weidmann, M.; Ademowo, O.G. Recombinase Polymerase Amplification Assay for Rapid Detection of Monkeypox Virus. Diagn. Microbiol. Infect. Dis. 2019, 95, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Macdonald, J.; Von Stetten, F. A Comprehensive Summary of a Decade Development of the Recombinase Polymerase Amplification. Analyst 2019, 144, 31–67, Erratum in Analyst 2020, 145, 1950–1960. [Google Scholar] [CrossRef]
- Berson, S.A.; Yalow, R.S. General Principles of Radioimmunoassay. Clin. Chim. Acta 1968, 22, 51–69. [Google Scholar] [CrossRef]
- Houldcroft, C.J.; Beale, M.A.; Breuer, J. Clinical and Biological Insights from Viral Genome Sequencing. Nat. Rev. Microbiol. 2017, 15, 183–192. [Google Scholar] [CrossRef]
- Li, D.; Wilkins, K.; McCollum, A.M.; Osadebe, L.; Kabamba, J.; Nguete, B.; Likafi, T.; Balilo, M.P.; Lushima, R.S.; Malekani, J. Evaluation of the Genexpert for Human Monkeypox Diagnosis. Am. J. Trop. Med. Hyg. 2017, 96, 405. [Google Scholar] [CrossRef]
- Karem, K.L.; Reynolds, M.; Braden, Z.; Lou, G.; Bernard, N.; Patton, J.; Damon, I.K. Characterization of Acute-Phase Humoral Immunity to Monkeypox: Use of Immunoglobulin M Enzyme-Linked Immunosorbent Assay for Detection of Monkeypox Infection During the 2003 North American Outbreak. Clin. Vaccine Immunol. 2005, 12, 867–872. [Google Scholar] [CrossRef]
- Adnan, N.; Haq, M.A.; Tisha, T.A.; Khandker, S.S.; Jamiruddin, M.R.; Sajal, S.S.A.; Akter, S.; Ahmed, M.F.; Raqib, R.; Khondoker, M.U. Optimizing SARS-CoV-2 Immunoassays for Specificity in Dengue-Co-Endemic Areas. Cureus 2023, 15, e47683. [Google Scholar] [CrossRef]
- Osborn, L.J.; Villarreal, D.; Wald-Dickler, N.; Bard, J.D. Monkeypox: Clinical Considerations, Epidemiology, and Laboratory Diagnostics. Clin. Microbiol. Newsl. 2022, 44, 199–208, Erratum in Clin. Microbiol. Newsl. 2024, 46, 1–2. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.W. Recent Advances in the Diagnosis Monkeypox: Implications for Public Health. Expert Rev. Mol. Diagn. 2022, 22, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Adnan, M.; Aldarhami, A.; Bazaid, A.S.; Saeedi, N.H.; Alkayyal, A.A.; Saleh, F.M.; Awadh, I.B.; Saeed, A.; Alshaghdali, K. Current Insights into Diagnosis, Prevention Strategies, Treatment, Therapeutic Targets, and Challenges of Monkeypox (Mpox) Infections in Human Populations. Life 2023, 13, 249. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Cheng, X.; Wei, H.; Tu, Z.; Rong, Z.; Wang, C.; Wang, S. Fluorescence-Enhanced Dual Signal Lateral Flow Immunoassay for Flexible and Ultrasensitive Detection of Monkeypox Virus. J. Nanobiotechnology 2023, 21, 450. [Google Scholar] [CrossRef]
- de Lima, L.F.; Barbosa, P.P.; Simeoni, C.L.; de Paula, R.F.d.O.; Proenca-Modena, J.L.; de Araujo, W.R. Electrochemical Paper-Based Nanobiosensor for Rapid and Sensitive Detection of Monkeypox Virus. ACS Appl. Mater. Interfaces 2023, 15, 58079–58091. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Zhang, X.; Liu, K.; Li, Y.; Wang, T.; Fang, Z.; Chen, C.; Tan, X.; Lin, Y.; Xu, Q. A Comparative Evaluation of Three Diagnostic Assays for the Detection of Human Monkeypox. Viruses 2024, 16, 1286. [Google Scholar] [CrossRef]
- Gulinaizhaer, A.; Yang, C.; Zou, M.; Ma, S.; Fan, X.; Wu, G. Detection of Monkeypox Virus Using Helicase Dependent Amplification and Recombinase Polymerase Amplification Combined with Lateral Flow Test. Virol. J. 2023, 20, 274. [Google Scholar] [CrossRef]
- Zhou, J.; Xiao, F.; Fu, J.; Jia, N.; Huang, X.; Sun, C.; Liu, C.; Huan, H.; Wang, Y. Rapid Detection of Monkeypox Virus by Multiple Cross Displacement Amplification Combined with Nanoparticle-Based Biosensor Platform. J. Med. Virol. 2023, 95, e28479. [Google Scholar] [CrossRef]
- Adler, H.; Gould, S.; Hine, P.; Snell, L.B.; Wong, W.; Houlihan, C.F.; Osborne, J.C.; Rampling, T.; Beadsworth, M.B.; Duncan, C.J. Clinical Features and Management of Human Monkeypox: A Retrospective Observational Study in the UK. Lancet Infect. Dis. 2022, 22, 1153–1162, Erratum in Lancet Infect. Dis. 2022, 22, e177. [Google Scholar] [CrossRef]
- CDC. What to Do If You Are Sick. Available online: https://www.cdc.gov/mpox/caring/index.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fmpox%2Fcaring%2Fpatients-guide-to-mpox-treatment-with-tpoxx-tecovirimat.html#cdc_caring_for_yourself_and_others_what_to_watch_out_for-when-extra-help-is-needed (accessed on 16 December 2025).
- Rao, A.S.C.; Alarcon, J.; Dretler, A. Situational Update for Clinicians About Severe Monkeypox Virus Infections. Available online: https://restoredcdc.org/www.cdc.gov/coca/hcp/trainings/severe-monkeypox-virus-infections.html (accessed on 16 December 2025).
- CDC. Monkeypox Virus Infection in the United States and Other Non-Endemic Countries—2022. Available online: https://archive.cdc.gov/#/details?url=https://www.cdc.gov/han/2022/han00466.html (accessed on 16 December 2025).
- CDC. Clinical Treatment of Mpox. Available online: https://www.cdc.gov/mpox/hcp/clinical-care/?CDC_AAref_Val=https://www.cdc.gov/poxvirus/mpox/clinicians/treatment.html (accessed on 16 December 2025).
- Brown, K.; Leggat, P.A. Human Monkeypox: Current State of Knowledge and Implications for the Future. Trop. Med. Infect. Dis. 2016, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Pütz, M.M.; Midgley, C.M.; Law, M.; Smith, G.L. Quantification of Antibody Responses against Multiple Antigens of the Two Infectious Forms of Vaccinia Virus Provides a Benchmark for Smallpox Vaccination. Nat. Med. 2006, 12, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- O’Laughlin, K. Clinical Use of Tecovirimat (Tpoxx) for Treatment of Monkeypox under an Investigational New Drug Protocol—United States, May–August 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 1190–1195. [Google Scholar] [CrossRef] [PubMed]
- Titanji, B.K.; Hazra, A.; Zucker, J. Mpox Clinical Presentation, Diagnostic Approaches, and Treatment Strategies: A Review. JAMA 2024, 332, 1652–1662. [Google Scholar] [CrossRef]
- Food and Drug Administration. FDA Approves Cidofovir for Treatment of CMV Retinitis. J. Int. Assoc. Physicians AIDS Care 1996, 2, 30. [Google Scholar]
- Hutson, C.L.; Kondas, A.V.; Mauldin, M.R.; Doty, J.B.; Grossi, I.M.; Morgan, C.N.; Ostergaard, S.D.; Hughes, C.M.; Nakazawa, Y.; Kling, C. Pharmacokinetics and Efficacy of a Potential Smallpox Therapeutic, Brincidofovir, in a Lethal Monkeypox Virus Animal Model. mSphere 2021, 6, e00927-20, Erratum in mSphere 2021, 6, e00126-21. https://doi.org/10.1128/msphere.00126-21. [Google Scholar] [CrossRef]
- Center for Drug Evaluation and Research. 2021. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2021/214460Orig1s000,%20214461Orig1s000Approv.pdf (accessed on 16 December 2025).
- Hoy, S.M. Tecovirimat: First Global Approval. Drugs 2018, 78, 1377–1382. [Google Scholar] [CrossRef]
- Baker, R.O.; Bray, M.; Huggins, J.W. Potential Antiviral Therapeutics for Smallpox, Monkeypox and Other Orthopoxvirus Infections. Antivir. Res. 2003, 57, 13–23. [Google Scholar] [CrossRef]
- Center for Drug Evaluation and Research. 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2018/208627Orig1s000MedR.pdf (accessed on 16 December 2025).
- Raccagni, A.R.; Candela, C.; Bruzzesi, E.; Mileto, D.; Canetti, D.; Rizzo, A.; Castagna, A.; Nozza, S. Real-Life Use of Cidofovir for Treatment of Severe Monkeypox Cases. J. Med. Virol. 2022, 95, e28218. [Google Scholar] [CrossRef] [PubMed]
- Ali, R.; Alonga, J.; Biampata, J.-L.; Basika, M.K.; Berry, I.M.; Bisento, N.; Blum, E.; Bonnett, T.; Cone, K.; Crozier, I. Placebo-Controlled Trial of Tecovirimat for Endemic (Clade I MPXV) Mpox, Drc. N. Engl. J. Med. 2025, 392, 1484. [Google Scholar]
- Zucker, J.; Fischer, W.A.; Zheng, L.; McCarthy, C.; Saha, P.T.; Javan, A.C.; Greninger, A.; Hamill, M.M.; Leslie, K.; Brooks, K.M. Tecovirimat for the Treatment of Mpox. N. Engl. J. Med. 2026, 394, 884–895. [Google Scholar] [CrossRef] [PubMed]
- Bloch, E.M.; Sullivan, D.J.; Shoham, S.; Tobian, A.A.; Casadevall, A.; Gebo, K.A. The Potential Role of Passive Antibody-Based Therapies as Treatments for Monkeypox. mBio 2022, 13, e0286222. [Google Scholar] [CrossRef] [PubMed]
- Saghazadeh, A.; Rezaei, N. Insights on Mpox Virus Infection Immunopathogenesis. Rev. Med. Virol. 2023, 33, e2426. [Google Scholar] [CrossRef]
- Li, H.; Huang, Q.-Z.; Zhang, H.; Liu, Z.-X.; Chen, X.-H.; Ye, L.-L.; Luo, Y. The Land-Scape of Immune Response to Monkeypox Virus. eBioMedicine 2023, 87, 104424. [Google Scholar] [CrossRef]
- Siegrist, E.A.; Sassine, J. Antivirals with Activity Against Mpox: A Clinically Oriented Review. Clin. Infect. Dis. 2023, 76, 155–164. [Google Scholar] [CrossRef]
- De Clercq, E. Cidofovir in the Treatment of Poxvirus Infections. Antivir. Res. 2002, 55, 1–13. [Google Scholar] [CrossRef]
- Andrei, G.; Snoeck, R. Cidofovir Activity Against Poxvirus Infections. Viruses 2010, 2, 2803. [Google Scholar] [CrossRef]
- Smee, D.F.; Sidwell, R.W.; Kefauver, D.; Bray, M.; Huggins, J.W. Characterization of Wild-Type and Cidofovir-Resistant Strains of Camelpox, Cowpox, Monkeypox, and Vaccinia Viruses. Antimicrob. Agents Chemother. 2002, 46, 1329–1335. [Google Scholar] [CrossRef]
- Magee, W.C.; Hostetler, K.Y.; Evans, D.H. Mechanism of Inhibition of Vaccinia Virus DNA Polymerase by Cidofovir Diphosphate. Antimicrob. Agents Chemother. 2005, 49, 3153–3162. [Google Scholar] [CrossRef]
- Jesus, D.M.; Costa, L.T.; Gonçalves, D.L.; Achete, C.A.; Attias, M.; Moussatché, N.; Damaso, C.R. Cidofovir Inhibits Genome Encapsidation and Affects Morphogenesis During the Replication of Vaccinia Virus. J. Virol. 2009, 83, 11477–11490. [Google Scholar] [CrossRef]
- Cihlar, T.; Chen, M.S. Identification of Enzymes Catalyzing Two-Step Phosphorylation of Cidofovir and the Effect of Cytomegalovirus Infection on Their Activities in Host Cells. Mol. Pharmacol. 1996, 50, 1502–1510. [Google Scholar] [CrossRef]
- Parker, S.; Handley, L.; Buller, R.M. Therapeutic and Prophylactic Drugs to Treat Orthopoxvirus Infections. Future Virol. 2008, 3, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Saalbach, K.P. Treatment and Vaccination for Smallpox and Monkeypox. Poxviruses 2024, 1451, 301–316. [Google Scholar]
- Merchlinsky, M.; Albright, A.; Olson, V.; Schiltz, H.; Merkeley, T.; Hughes, C.; Petersen, B.; Challberg, M. The Development and Approval of Tecoviromat (Tpoxx®), the First Antiviral Against Smallpox. Antivir. Res. 2019, 168, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Pevear, D.C.; Davies, M.H.; Collett, M.S.; Bailey, T.; Rippen, S.; Barone, L.; Burns, C.; Rhodes, G.; Tohan, S. An Orally Bioavailable Antipoxvirus Compound (St-246) Inhibits Extracellular Virus Formation and Protects Mice from Lethal Orthopoxvirus Challenge. J. Virol. 2005, 79, 13139–13149. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.T.; Grosenbach, D.W.; Chinsangaram, J.; Honeychurch, K.M.; Long, P.G.; Lovejoy, C.; Maiti, B.; Meara, I.; Hruby, D.E. An Overview of Tecovirimat for Smallpox Treatment and Expanded Anti-Orthopoxvirus Applications. Expert Rev. Anti-Infect. Ther. 2021, 19, 331–344. [Google Scholar] [CrossRef]
- Mazurkov, O.Y.; Kabanov, A.S.; Shishkina, L.N.; Sergeev, A.A.; Skarnovich, M.O.; Bormotov, N.I.; Skarnovich, M.A.; Ovchinnikova, A.S.; Titova, K.A.; Galahova, D.O.; et al. New Effective Chemically Synthesized Anti-Smallpox Compound Nioch-14. J. Gen. Virol. 2016, 97, 1229–1239. [Google Scholar] [CrossRef]
- Delaune, D.; Iseni, F. Drug Development Against Smallpox: Present and Future. Antimicrob. Agents Chemother. 2020, 64, e01683-19. [Google Scholar] [CrossRef]
- WHO. Who Advisory Committee on Variola Virus Research: Report of the Twenty-Third Meeting, Virtual Meeting, 3–4 November 2021. Available online: https://iris.who.int/handle/10665/354260 (accessed on 16 December 2024).
- Kolodziej, M.; Joniec, J.; Bartoszcze, M.; Gryko, R.; Kocik, J.; Knap, J. Research on Substances with Activity Against Orthopoxviruses. Ann. Agric. Environ. Med. 2013, 20, 1–7. [Google Scholar]
- Garriga, D.; Headey, S.; Accurso, C.; Gunzburg, M.; Scanlon, M.; Coulibaly, F. Structural Basis for the Inhibition of Poxvirus Assembly by the Antibiotic Rifampicin. Proc. Natl. Acad. Sci. USA 2018, 115, 8424–8429. [Google Scholar] [CrossRef]
- Wittek, R. Vaccinia Immune Globulin: Current Policies, Preparedness, and Product Safety and Efficacy. Int. J. Infect. Dis. 2006, 10, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Rizk, J.G.; Lippi, G.; Henry, B.M.; Forthal, D.N.; Rizk, Y. Prevention and Treatment of Monkeypox. Drugs 2022, 82, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gu, Z.; Sheng, S.; Song, R.; Jin, R. The Current State and Progress of Mpox Vaccine Research. China CDC Wkly. 2024, 6, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Gieryńska, M.; Szulc-Dąbrowska, L.; Struzik, J.; Gregorczyk-Zboroch, K.P.; Mielcarska, M.B.; Toka, F.N.; Schollenberger, A.; Biernacka, Z. Orthopoxvirus Zoonoses—Do We Still Remember and Are Ready to Fight? Pathogens 2023, 12, 363. [Google Scholar] [CrossRef] [PubMed]
- Martín-Delgado, M.C.; Sánchez, F.J.M.; Martínez-Sellés, M.; García, J.M.M.; Guillén, S.M.; Rodríguez-Artalejo, F.; Ruiz-Galiana, J.; Cantón, R.; Ramos, P.D.L.; García-Botella, A.; et al. Monkeypox in Humans: A New Outbreak. Rev. Esp. Quimioter. 2022, 35, 509. [Google Scholar] [CrossRef]
- Lozano, J.M.; Muller, S. Monkeypox: Potential Vaccine Development Strategies. Trends Pharmacol. Sci. 2023, 44, 15–19. [Google Scholar] [CrossRef]
- Hajra, D.; Datey, A.; Chakravortty, D. Attenuation Methods for Live Vaccines. Vaccine Deliv. Technol. Methods Protoc. 2021, 2183, 331–356. [Google Scholar]
- Reina, J.; Iglesias, C. Vaccines against Monkeypox. Med. Clín. (Engl. Ed.) 2023, 160, 305–309. [Google Scholar] [CrossRef]
- Jandrasits, D.; Züst, R.; Siegrist, D.; Engler, O.B.; Weber, B.; Schmidt, K.M.; Jonsdottir, H.R. Third-Generation Smallpox Vaccines Induce Low-Level Cross-Protecting Neutralizing Antibodies Against Monkeypox Virus in Laboratory Workers. Heliyon 2024, 10, e31490. [Google Scholar] [CrossRef] [PubMed]
- Meseda, C.A.; Garcia, A.D.; Kumar, A.; Mayer, A.E.; Manischewitz, J.; King, L.R.; Golding, H.; Merchlinsky, M.; Weir, J.P. Enhanced Immunogenicity and Protective Effect Conferred by Vaccination with Combinations of Modified Vaccinia Virus Ankara and Licensed Smallpox Vaccine Dryvax in a Mouse Model. Virology 2005, 339, 164–175. [Google Scholar] [CrossRef] [PubMed]
- WHO. Summary Report on First, Second and Third Generation Smallpox Vaccines; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- Pugh, C.; Keasey, S.; Korman, L.; Pittman, P.R.; Ulrich, R.G. Human Antibody Responses to the Polyclonal Dryvax Vaccine for Smallpox Prevention Can Be Distinguished from Responses to the Monoclonal Replacement Vaccine Acam2000. Clin. Vaccine Immunol. 2014, 21, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Dryvax® (Smallpox Vaccine, Dried, Calf Lymph Type) Wyeth Laboratories, Inc. Available online: https://biotech.law.lsu.edu/blaw/bt/smallpox/dryvax_label.htm (accessed on 16 December 2025).
- Hatch, G.J.; Graham, V.A.; Bewley, K.R.; Tree, J.A.; Dennis, M.; Taylor, I.; Funnell, S.G.; Bate, S.R.; Steeds, K.; Tipton, T.; et al. Assessment of the Protective Effect of Imvamune and Acam2000 Vaccines Against Aerosolized Monkeypox Virus in Cynomolgus Macaques. J. Virol. 2013, 87, 7805–7815. [Google Scholar] [CrossRef]
- Islam, M.R.; Hossain, M.J.; Roy, A.; Hasan, A.N.; Rahman, M.A.; Shahriar, M.; Bhuiyan, M.A. Repositioning Potentials of Smallpox Vaccines and Antiviral Agents in Monkeypox Outbreak: A Rapid Review on Comparative Benefits and Risks. Health Sci. Rep. 2022, 5, e798. [Google Scholar] [CrossRef]
- Rock, M.T.; Yoder, S.M.; Talbot, T.R.; Edwards, K.M.; Crowe, J.E. Cellular Immune Responses to Diluted and Undiluted Aventis Pasteur Smallpox Vaccine. J. Infect. Dis. 2006, 194, 435–443. [Google Scholar] [CrossRef]
- WHO. Overview of Available Vaccines against Smallpox & Monkeypox; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- Greenberg, R.N.; Kennedy, J.S. Acam2000: A Newly Licensed Cell Culture-Based Live Vaccinia Smallpox Vaccine. Expert Opin. Investig. Drugs 2008, 17, 555–564. [Google Scholar] [CrossRef]
- Nalca, A.; Zumbrun, E.E. Acam2000™: The New Smallpox Vaccine for United States Strategic National Stockpile. Drug Des. Dev. Ther. 2010, 4, 71–79. [Google Scholar] [CrossRef] [PubMed]
- CDC. About Smallpox Vaccines. Available online: https://www.cdc.gov/smallpox/hcp/vaccines/index.html (accessed on 16 December 2025).
- WHO. Recommendation for an Emergency Use Listing of “Freeze-Dried Smallpox Vaccine Prepared in Cell Culture Lc16 “Kmb” (Lc16m8 Vaccine) Submitted by Km Biologics, Japan; WHO: Geneva, Switzerland, 2024. [Google Scholar]
- Kenner, J.; Cameron, F.; Empig, C.; Jobes, D.V.; Gurwith, M. Lc16m8: An Attenuated Smallpox Vaccine. Vaccine 2006, 24, 7009–7022. [Google Scholar] [CrossRef]
- Kennedy, J.S.; Gurwith, M.; Dekker, C.L.; Frey, S.E.; Edwards, K.M.; Kenner, J.; Lock, M.; Empig, C.; Morikawa, S.; Saijo, M.; et al. Safety and Immunogenicity of Lc16m8, an Attenuated Smallpox Vaccine in Vaccinia-Naive Adults. J. Infect. Dis. 2011, 204, 1395–1402. [Google Scholar] [CrossRef]
- WHO. Policy Brief on Vaccination against Monkeypox in the Who European Region; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- Rao, A.K. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for Preexposure Vaccination of Persons at Risk for Occupational Exposure to Orthopoxviruses: Recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 886. [Google Scholar] [CrossRef] [PubMed]
- Full Prescribing Information. Available online: https://www.fda.gov/media/131078/download (accessed on 16 December 2025).
- Frey, S.E.; Winokur, P.L.; Salata, R.A.; El-Kamary, S.S.; Turley, C.B.; Walter, E.B., Jr.; Hay, C.M.; Newman, F.K.; Hill, H.R.; Zhang, Y. Safety and Immunogenicity of Imvamune® Smallpox Vaccine Using Different Strategies for a Post Event Scenario. Vaccine 2013, 31, 3025–3033. [Google Scholar] [CrossRef] [PubMed]
- Saadh, M.J.; Ghadimkhani, T.; Soltani, N.; Abbassioun, A.; Pecho, R.D.C.; Kazem, T.J.; Yasamineh, S.; Gholizadeh, O.; Taha, A. Progress and Prospects on Vaccine Development Against Monkeypox Infection. Microb. Pathog. 2023, 180, 106156. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Sampedro, L.; Perdiguero, B.; Mejías-Pérez, E.; García-Arriaza, J.; Di Pilato, M.; Esteban, M. The Evolution of Poxvirus Vaccines. Viruses 2015, 7, 1726–1803. [Google Scholar] [CrossRef] [PubMed]
- Wiser, I.; Balicer, R.D.; Cohen, D. An Update on Smallpox Vaccine Candidates and Their Role in Bioterrorism Related Vaccination Strategies. Vaccine 2007, 25, 976–984. [Google Scholar] [CrossRef]
- de la Calle-Prieto, F.; Muñoz, M.E.; Ramírez, G.; Díaz-Menéndez, M.; Velasco, M.; Galparsoro, H.A.; Lletí, M.S.; Forte, T.M.; Blanco, J.L.; Mora-Rillo, M. Treatment and Prevention of Monkeypox. Enferm. Infecc. Microbiol. Clín. (Engl. Ed.) 2023, 41, 629–634. [Google Scholar] [CrossRef]
- Monath, T.P.; Caldwell, J.R.; Mundt, W.; Fusco, J.; Johnson, C.S.; Buller, M.; Liu, J.; Gardner, B.; Downing, G.; Blum, P.S. Acam2000 Clonal Vero Cell Culture Vaccinia Virus (New York City Board of Health Strain)—A Second-Generation Smallpox Vaccine for Biological Defense. Int. J. Infect. Dis. 2004, 8, 31–44. [Google Scholar] [CrossRef]
- Ferrier-Rembert, A.; Drillien, R.; Meignier, B.; Garin, D.; Crance, J.-M. Safety, Immunogenicity and Protective Efficacy in Mice of a New Cell-Cultured Lister Smallpox Vaccine Candidate. Vaccine 2007, 25, 8290–8297. [Google Scholar] [CrossRef]
- Eto, A.; Saito, T.; Yokote, H.; Kurane, I.; Kanatani, Y. Recent Advances in the Study of Live Attenuated Cell-Cultured Smallpox Vaccine Lc16m8. Vaccine 2015, 33, 6106–6111. [Google Scholar] [CrossRef]
- Kennedy, J.S.; Greenberg, R.N. Imvamune®: Modified Vaccinia Ankara Strain as an Attenuated Smallpox Vaccine. Expert Rev. Vaccines 2009, 8, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.W. Clinical Guidance for the Use of JYNNEOS. Advisory Committee on Immunization Practices. Meeting: Orthopoxvirus. 2021. Available online: https://stacks.cdc.gov/view/cdc/111305 (accessed on 16 December 2025).
- Rao, A.K.; Minhaj, F.S.; Carter, R.J.; Duffy, J.; Satheshkumar, P.S.; Delaney, K.P.; Quilter, L.A.S.; Kachur, R.E.; McLean, C.; Moulia, D.L.; et al. Use of JYNNEOS (Smallpox and Mpox Vaccine, Live, Nonreplicating) for Persons Aged ≥18 Years at Risk for Mpox During an Mpox Outbreak: Recommendations of the Advisory Committee on Immunization Practices—United States, 2023. MMWR Morb. Mortal. Wkly. Rep. 2025, 74, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Vetter, V.; Denizer, G.; Friedland, L.R.; Krishnan, J.; Shapiro, M. Understanding Modern-Day Vaccines: What You Need to Know. Ann. Med. 2018, 50, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Pollard, A.J.; Bijker, E.M. A Guide to Vaccinology: From Basic Principles to New Developments. Nat. Rev. Immunol. 2021, 21, 83–100, Erratum in Nat. Rev. Immunol. 2021, 21, 129. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Shahid, F.A.; Haq, M.U.; Qamar, M.T.U.; Irfan, M.; Shaker, B.; Ahmad, S.; Alrumaihi, F.; Allemailem, K.S.; Almatroudi, A. An Integrative Reverse Vaccinology, Immunoinformatic, Docking and Simulation Approaches Towards Designing of Multi-Epitopes Based Vaccine Against Monkeypox Virus. J. Biomol. Struct. Dyn. 2023, 41, 7821–7834. [Google Scholar] [CrossRef] [PubMed]
- Golden, J.W.; Hooper, J.W. The Strategic Use of Novel Smallpox Vaccines in the Post-Eradication World. Expert Rev. Vaccines 2011, 10, 1021–1035. [Google Scholar] [CrossRef]
- Buchman, G.W.; Cohen, M.E.; Xiao, Y.; Richardson-Harman, N.; Silvera, P.; DeTolla, L.J.; Davis, H.L.; Eisenberg, R.J.; Cohen, G.H.; Isaacs, S.N. A Protein-Based Smallpox Vaccine Protects Non-Human Primates from a Lethal Monkeypox Virus Challenge. Vaccine 2010, 28, 6627–6636. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Chatterjee, S.; Nag, S.; Dhama, K.; Chakraborty, C. Designing, Characterization, and Immune Stimulation of a Novel Multi-Epitopic Peptide-Based Potential Vaccine Candidate Against Monkeypox Virus Through Screening Its Whole Genome Encoded Proteins: An Immunoinformatics Approach. Travel Med. Infect. Dis. 2022, 50, 102481. [Google Scholar] [CrossRef]
- Shantier, S.W.; Mustafa, M.I.; Abdelmoneim, A.H.; Fadl, H.A.; Elbager, S.G.; Makhawi, A.M. Novel Multi Epitope-Based Vaccine Against Monkeypox Virus: Vaccinomic Approach. Sci. Rep. 2022, 12, 15983. [Google Scholar] [CrossRef]
- Heraud, J.-M.; Edghill-Smith, Y.; Ayala, V.; Kalisz, I.; Parrino, J.; Kalyanaraman, V.S.; Manischewitz, J.; King, L.R.; Hryniewicz, A.; Trindade, C.J.; et al. Subunit Recombinant Vaccine Protects Against Monkeypox. J. Immunol. 2006, 177, 2552–2564. [Google Scholar] [CrossRef]
- Hirao, L.A.; Draghia-Akli, R.; Prigge, J.T.; Yang, M.; Satishchandran, A.; Wu, L.; Hammarlund, E.; Khan, A.S.; Babas, T.; Rhodes, L.; et al. Multivalent Smallpox DNA Vaccine Delivered by Intradermal Electroporation Drives Protective Immunity in Nonhuman Primates against Lethal Monkeypox Challenge. J. Infect. Dis. 2011, 203, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Mucker, E.M.; Thiele-Suess, C.; Baumhof, P.; Hooper, J.W. Lipid Nanoparticle Delivery of Unmodified Mrnas Encoding Multiple Monoclonal Antibodies Targeting Poxviruses in Rabbits. Mol. Ther.-Nucleic Acids 2022, 28, 847–858. [Google Scholar] [CrossRef]
- Nuzzo, J.B.; Borio, L.L.; Gostin, L.O. The Who Declaration of Monkeypox as a Global Public Health Emergency. JAMA 2022, 328, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.-M.; Zheng, Y.-J.; Zhou, L.; Feng, L.-Z.; Ma, L.; Xu, B.-P.; Xu, H.-M.; Liu, W.; Xie, Z.-D.; Deng, J.-K.; et al. Diagnosis, Treatment, and Prevention of Monkeypox in Children: An Experts’ Consensus Statement. World J. Pediatr. 2023, 19, 231–242. [Google Scholar] [CrossRef]
- Maldonado, M.S.; Lucchetti, A.J.; Pacheco, R.A.P.; Cevallos, L.C.M.; Saavedra, E.U.Z.; Zapata, L.R.P.; Huayta, F.A.L.; Prado, E.D.M. Epidemiologic Characteristics and Clinical Features of Patients with Monkeypox Virus Infection from a Hospital in Peru between July and September 2022. Int. J. Infect. Dis. 2023, 129, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Decousser, J.-W.; Romano-Bertrand, S.; Glele, L.A.; Baron, R.; Carre, Y.; Cassier, P.; Dananche, C.; Depaix-Champagnac, F.; Fournier, S.; Racaud, J.; et al. Healthcare Worker Protection Against Mpox Contamination: Position Paper of the French Society for Hospital Hygiene. J. Hosp. Infect. 2023, 140, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Kampf, G. Efficacy of Biocidal Agents and Disinfectants Against the Monkeypox Virus and Other Orthopoxviruses. J. Hosp. Infect. 2022, 127, 101–110. [Google Scholar] [CrossRef]
- Hasan, S.; Saeed, S. Monkeypox Disease: An Emerging Public Health Concern in the Shadow of COVID-19 Pandemic: An Update. Trop. Med. Infect. Dis. 2022, 7, 283. [Google Scholar] [CrossRef]
- CDC. Mpox Monitoring and Risk Assessment for People Exposed in the Community. Available online: https://www.cdc.gov/mpox/php/monitoring/index.html (accessed on 16 December 2024).
- CDC. Interim Guidance for Prevention and Treatment of Monkeypox in Persons with HIV Infection—United States, August 2022. Available online: https://www.cdc.gov/mmwr/volumes/71/wr/mm7132e4.htm (accessed on 16 December 2024).
- Sah, R.; Mohanty, A.; Hada, V.; Singh, P.; Govindaswamy, A.; Siddiq, A.; Reda, A.; Dhama, K. The Emergence of Monkeypox: A Global Health Threat. Cureus 2022, 14, e29304. [Google Scholar] [CrossRef]
- Nolen, L.D.; Osadebe, L.; Katomba, J.; Likofata, J.; Mukadi, D.; Monroe, B.; Doty, J.; Malekani, J.; Kabamba, J.; Bomponda, P.L.; et al. Introduction of Monkeypox into a Community and Household: Risk Factors and Zoonotic Reservoirs in the Democratic Republic of the Congo. Am. J. Trop. Med. Hyg. 2015, 93, 410. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Carroll, D.S.; Karem, K.L. Factors Affecting the Likelihood of Monkeypox’s Emergence and Spread in the Post-Smallpox Era. Curr. Opin. Virol. 2012, 2, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Farahat, R.A.; Head, M.G.; Tharwat, S.; Alabdallat, Y.; Essar, M.Y.; Abdelazeem, B.; Ould Setti, M. Infodemic and the Fear of Monkeypox: Call for Action. Trop. Med. Health 2022, 50, 63. [Google Scholar] [CrossRef] [PubMed]
- Emery, N.; Dugerdil, A.; Flahault, A. Infodemics in an Era of Pandemics. Int. J. Public Health 2022, 67, 1605209. [Google Scholar] [CrossRef] [PubMed]
- Alam, A.M.; Azim Majumder, M.A.; Haque, M.; Ashraf, F.; Khondoker, M.U.; Mashreky, S.R.; Wahab, A.; Siddiqui, T.H.; Uddin, A.; Joarder, T.; et al. Disproportionate COVID-19 Vaccine Acceptance Rate Among Healthcare Professionals on the Eve of Nationwide Vaccine Distribution in Bangladesh. Expert Rev. Vaccines 2021, 20, 1167–1175. [Google Scholar] [CrossRef]
- Mou, T.J.; Afroz, K.A.; Haq, M.A.; Jahan, D.; Ahmad, R.; Islam, T.; Chowdhury, K.; Kumar, S.; Irfan, M.; Islam, M.S. The Effect of Socio-Demographic Factors in Health-Seeking Behaviors Among Bangladeshi Residents During the First Wave of COVID-19. Healthcare 2022, 10, 483. [Google Scholar] [CrossRef]
- Deputy, N.P.; Deckert, J.; Chard, A.N.; Sandberg, N.; Moulia, D.L.; Barkley, E.; Dalton, A.F.; Sweet, C.; Cohn, A.C.; Little, D.R.; et al. Vaccine Effectiveness of JYNNEOS Against Mpox Disease in the United States. N. Engl. J. Med. 2023, 388, 2434–2443. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.