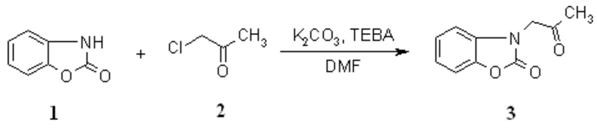
The title compound is an important intermediate for the preparation of 3,4- and 3,4,5-substituted 1-(2-hydroxy-phenyl)imidazolin-2-ones [1,2] and 6-methyl-4-(2-hydroxyphenyl)-1,2,4-triazin-3-ones [3]. According to literature data, the product 3 is prepared from 2(3H)-benzoxazolone with commercially unavailable bromoacetone in abs. ethanol in the presence of sodium ethoxide [4,5]. We present an improve method for the synthesis of 3-(2-oxopropyl)-2(3H)-benzoxazolone (3) from available starting materials 1 and 2 under PTC conditions in higher yield.
To a mixture of 2(3H)-benzoxazolone (6.75 g, 50 mmol), fine powdered potassium carbonate (4.14 g, 30 mmol) and TEBA (200 mg) in DMF (15 ml), chloroacetone (4 ml, 50 mmol) was added. The reaction mixture was stirred for 1 h at 20-25 0C and then poured under stirring into 150 ml of water. The resulting precipitate was filtered and washed with water. Recrystallization from ethanol gave the product 3 as colorless needles. Yield: 90%.
Melting point: 119-120 0C
Elemental Analysis: Calculated for C10H9NO3: C 62.82%, H 4.74%, N 7.33%. Found: C 62.95%, H 4.53%, N 7.17%.
IR (nujol, cm-1): 1770, 1720 (CO).
1H-NMR (250 MHz, CDCl3): 2.25 (s, 3H, CH3); 4.61 (s, 2H, CH2); 6.77-7.21 (m, 4H, arom. H).
13C-NMR (63 MHz, CDCl3): 26.9 (CH3CO); 50.7 (CH2CO); 108.2 (C7); 109.9 (C4); 122.7 (C5); 123.8 (C6); 142.4 (C7a); 154.3 (C2); 199.8 (CH2CO).
MS (m/z, %): 191 [M+] (47); 148 (67); 120 (12); 104 (11); 77 (100); 65 (10); 50 (19).
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References
- Lozanova, Ch.; Kalcheva, V.; Simov, D. Khim. Geterosikl. Soedin. 1988, 10, 1362.
- Lozanova, Ch.; Kalcheva, V. J. Prakt. Chem. 1989, 331(6), 1007.
- Bojtscheva, Ch.; Simov, D.; Kaltscheva, V. J. Prakt. Chem. 1979, 321(2), 226.
- Zinner, H.; Randow, F. J. Prakt. Chem. 1961, 4, 144.
- Simov, D. A.; Kalcheva, V.B.; Boycheva, H.S. Compt. Rend. Acad. Bulg. Sci. 1974, 27, 1073.
© 2007 by MDPI (http://www.mdpi.org/). Reproduction is permitted for noncommercial purposes.
