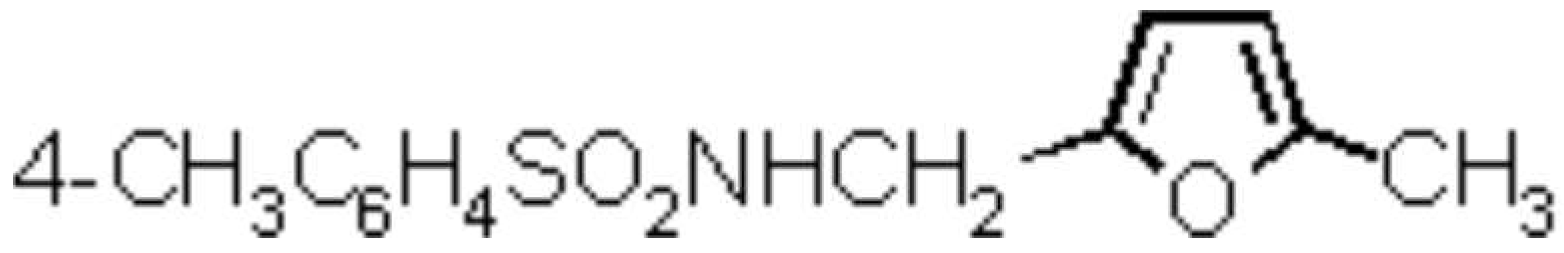
5-Methylfurfurylamine was prepared from commercially available 5-methylfurfural via the oxime, followed by LiAlH4 reduction [1]. A solution of 5-methylfurfurylamine (12.2 g, 100 mmol) in tert-butylmethyl ether[2] (MTBE, 50 mL) was made in a 250-mL four-necked flask, fitted with two dropping funnels and an immersion thermometer, and cooled in an ice/salt bath to -15 °C. With vigorous stirring, solutions of p-toluenesulfonyl chloride (20.97 g, 110 mmol) in MTBE (80 mL) and 20% aqueous sodium hydroxide (22 mL, 110 mmol) were added simultaneously and cautiously. The temperatur of the reaction mixture was not allowed to exceed –5 °C. When the addition was completed (60–90 min), the cooling bath was removed, and the mixture stirred for 2–3 hours. The MTBE layer was separated, and the aqueous layer acidified to pH 3–4 and extracted with MTBE (4 ’ 40 mL). The combined organic solutions were washed with saturated brine (2 ’ 40 mL) and dried overnight with magnesium sulfate. After filtration, the solvent was removed in a rotary evaporator. The remaining pale yellow oil solidified in a refrigerator. This raw product was recrystallized from ethanol/pentane (1:1 v/v, 150 mL) to yield 23.44 g (88%) of a colourless crystalline solid.
Melting Point: 82.5–83.5 °C [3].
1H-NMR (250 MHz, CDCl3): δ= 2.11 (s, 3 H, 5-CH3); 2.41 (s, 3 H, CH3C6H4SO2); 4.10 (d, J = 5.9 Hz, 2 H, CH2-N); 4.98 (t, J = 5.9 Hz, 1 H, NH); AB sub-spectrum with δ A = 5.95, δ B = 5.76, JAB = 3.1 Hz, the B part is split (5 lines, 2 H, H-3 and H-4); AA’BB’ sub-spectrum with δ A = 7.71, δ B = 7.26, JAB = 8.0 Hz, (4 H, H-2/6 of CH3C6H4SO2, HAA’ and H-3/5 of CH3C6H4SO2, HBB’).
13C-NMR (62.9 MHz, CDCl3): δ= 12.3 (5-CH3); 20.5 (CH3C6H4SO2); 39.2 (CH2-N); 105.15 (C-4); 108.1 (C-3); 126.1 (Tos-C-2/6); 128.5 (Tos-C-3/5); 136.0 (Tos-C-4); 142.3 (Tos-C-1); 146.6 (C-5); 151.2 (C-2).
IR (CHCl3 film, cm-1): 3380, 3280 (NH); 2930, 2880 (C-H); 1598 (C=C); 1555, 1492, 1410, 1330, 1160 cm-1 (SO2).
Elemental Analysis: Calculated for C13H15NO3S (265.3): C, 58.85%; H, 5.70%; N, 5.28%; S, 12.08%. Found: C, 58.61%; H, 5.68%; N, 5.24%; S 12.03%.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes
- a) Mndzhoian, A. L.; Afrikian, V. G.; Khorenian, G. A. Izv. Akad. Nauk. Arm. SSR Khim. Nauki 1961, 14, 363–368, Chem. Abstr. 1962, 57, 11136g. b) Mndzhoian, A. L. Synthesis of Heterocyclic Compounds; Consultants Bureau: London, 1959; Vol. 2, p. 60. [Google Scholar]
- tert-Butylmethyl ether was recommended as an inexpensive solvent when this experiment was carried out (June 1991, Diploma thesis of H.M.). Nowadays, it is considered environmentally problematic. We also used 1,2-dichloroethane instead of the ether.
- For a more expensive micro-scale preparation with selective annihilation of contaminants (‘impurity annihilation’), see: Barrett, A. G. M.; Smith, M. L.; Zecri, F. J. J. Chem. Soc., Chem. Commun. 1998, 2317–2318. However, physical data and combustion analysis are not given in this paper.
- Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.
