Modified mRNA-Based Therapeutic Strategies for Myocardial Ischemia–Reperfusion Injury
Abstract
1. Introduction
2. Mechanisms of Myocardial Ischemia–Reperfusion Injury
2.1. Oxidative Stress
2.2. Inflammatory Response
2.3. Autophagy, Apoptosis, and Necrosis
3. Structural Basis of modRNA
4. Delivery Vehicles for modRNA
| Delivery Vehicle | Composition/Properties | Advantages | Limitations | References |
|---|---|---|---|---|
| Lipid Nanoparticles (LNPs) | Cationic lipids, phospholipids, cholesterol, PEG-lipids forming uniform lipid core | High encapsulation efficiency; good cellular uptake; scalable production (e.g., COVID-19 mRNA vaccines); clinically validated | Limited cardiomyocyte targeting (primarily accumulates in fibroblasts); may cause mild inflammation | [98,99,100] |
| Polymeric Nanoparticles | PEI, etc., via electrostatic encapsulation | Tunable structure; biodegradable (good biocompatibility); sustained release | Lower transfection efficiency than LNPs; some polymers (e.g., PEI) show cytotoxicity | [101,102] |
| Viral Vectors | AAV, lentiviruses carrying modRNA (non-genomic integration) | High transduction efficiency | Immunogenicity risk; limited cargo capacity (<4.7 kb); preexisting antibodies may neutralize vectors (30–50% AAV neutralization rate) | [88,103] |
| Exosomes | Natural nanovesicles (30–150 nm) with targetable surface peptides | Extremely low immunogenicity; natural membrane penetration; endogenous (avoids phagocytic clearance) | Low yield/purification challenges; inconsistent drug loading | [104,105,106] |
| Naked modRNA + Buffer | Sucrose-citrate buffer system (unencapsulated) | Fastest translation (protein expression in 10 min); no carrier toxicity; lowest cost | Highly susceptible to nuclease degradation; lacks targeting; only suitable for local injection | [91] |
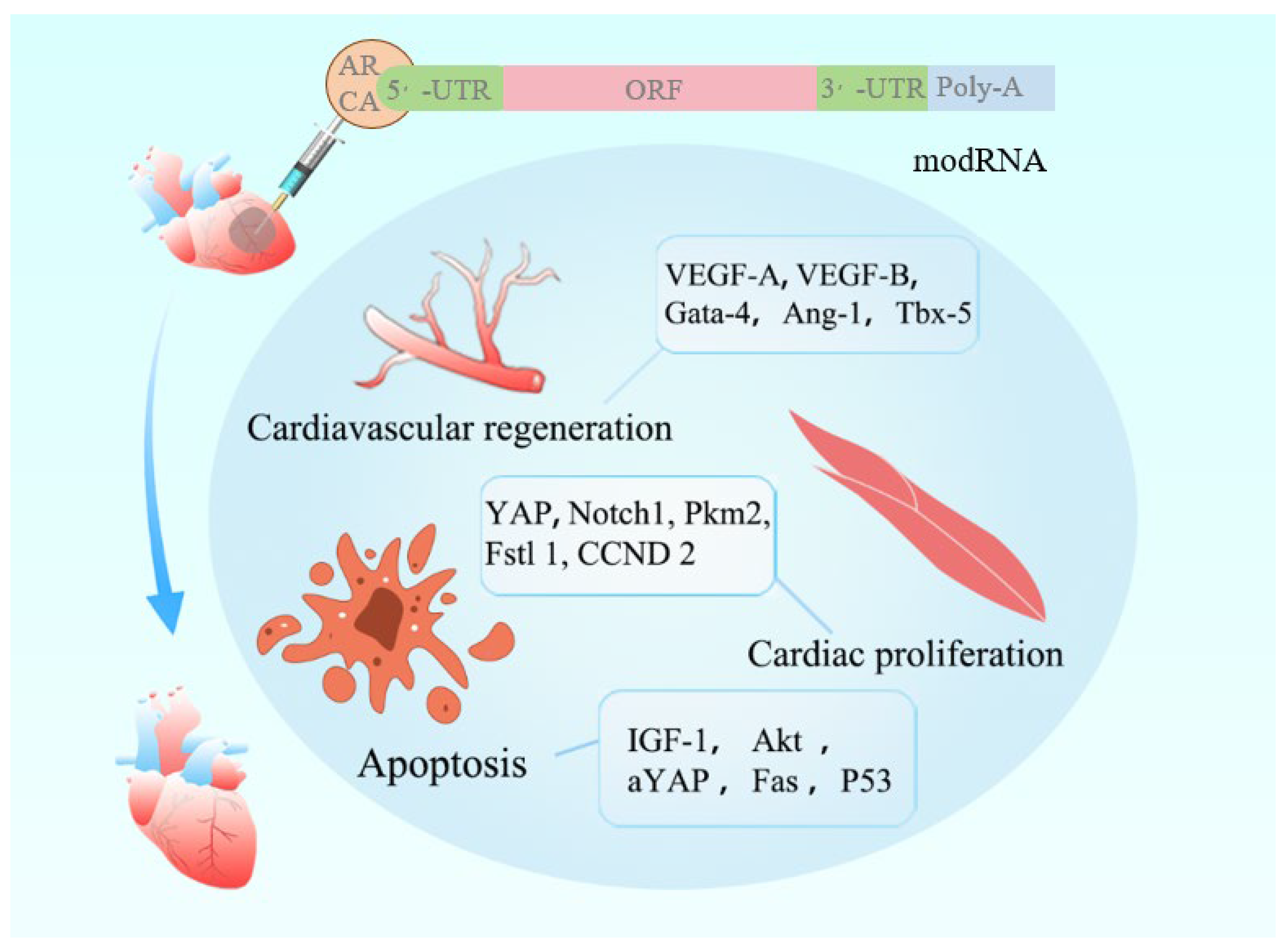
5. Applications of modRNA in MIRI Therapy
5.1. Cardiomyocyte Proliferation and Regeneration
5.2. Cardiovascular Regeneration
5.3. Anti-Apoptotic Strategies
5.4. Anti-Fibrotic Strategies
6. Challenges and Future Perspectives of modRNA in MIRI Therapy
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Wang, W.; Hu, M.; Liu, H.; Zhang, X.; Li, H.; Zhou, F.; Liu, Y.M.; Lei, F.; Qin, J.J.; Zhao, Y.C.; et al. Global Burden of Disease Study 2019 suggests that metabolic risk factors are the leading drivers of the burden of ischemic heart disease. Cell Metab. 2021, 33, 1943–1956.e2. [Google Scholar] [CrossRef]
- An, X.; Liu, Z.; Zhang, L.; Zhao, J.; Gu, Q.; Han, W.; Hu, Y.; Xue, F.; Zhang, F.; Wang, S.; et al. Co-occurrence patterns and related risk factors of ischaemic heart disease and ischaemic stroke across 203 countries and territories: A spatial correspondence and systematic analysis. Lancet Glob. Health 2025, 13, e808–e819, Erratum in Lancet Glob. Health 2025, 13, e1174. [Google Scholar] [CrossRef]
- Chang, H.; Chen, E.; Zhu, T.; Liu, J.; Chen, C. Communication Regarding the Myocardial Ischemia/Reperfusion and Cognitive Impairment: A Narrative Literature Review. J. Alzheimer’s Dis. 2024, 97, 1545–1570. [Google Scholar] [CrossRef]
- Ahmad, Y.; Howard, J.P.; Arnold, A.D.; Cook, C.M.; Prasad, M.; Ali, Z.A.; Parikh, M.A.; Kosmidou, I.; Francis, D.P.; Moses, J.W.; et al. Mortality after drug-eluting stents vs. coronary artery bypass grafting for left main coronary artery disease: A meta-analysis of randomized controlled trials. Eur. Heart J. 2020, 41, 3228–3235. [Google Scholar] [CrossRef]
- Buja, M.L.; Heide, V.S.R. Pathobiology of Ischemic Heart Disease: Past, Present and Future. Cardiovasc. Pathol. 2016, 25, 214–220. [Google Scholar] [CrossRef]
- Tasoulis, M.-K.; Douzinas, E.E. Hypoxemic reperfusion of ischemic states: An alternative approach for the attenuation of oxidative stress mediated reperfusion injury. J. Biomed. Sci. 2016, 23, 7. [Google Scholar] [CrossRef]
- Chen, W.; Liu, L.; Tang, M.; Li, J.; Yuan, W.; Yin, D.; Cao, Y.; Tian, J. Type I collagen-targeted liposome delivery of Serca2a modulates myocardium calcium homeostasis and reduces cardiac fibrosis induced by myocardial infarction. Mater. Today Bio 2024, 28, 101162. [Google Scholar] [CrossRef]
- Zou, R.; Shi, W.; Qiu, J.; Zhou, N.; Du, N.; Zhou, H.; Chen, X.; Ma, L. Empagliflozin attenuates cardiac microvascular ischemia/reperfusion injury through improving mitochondrial homeostasis. Cardiovasc. Diabetol. 2022, 21, 106. [Google Scholar] [CrossRef]
- Tawfik, M.K.; Abo-Elmatty, D.M.; Ahmed, A.A.M. The role of ATP-sensitive potassium channel blockers in ischemia-reperfusion-induced renal injury versus their effects on cardiac ischemia reperfusion in rats. Eur. Rev. Med. Pharmacol. Sci. 2009, 13, 81–93. [Google Scholar]
- Liao, X.; Tang, M.; Li, J.; Guo, R.; Zhong, C.; Chen, X.; Zhang, X.; Mo, H.; Que, D.; Yu, W.; et al. Acid-Triggered Cascaded Responsive Supramolecular Peptide Alleviates Myocardial Ischemia—Reperfusion Injury by Restoring Redox Homeostasis and Protecting Mitochondrial Function. Adv. Healthc. Mater. 2025, 14, e2404319. [Google Scholar] [CrossRef]
- Bassino, E.; Fornero, S.; Gallo, M.P.; Gallina, C.; Femminò, S.; Levi, R.; Tota, B.; Alloatti, G. Catestatin exerts direct protective effects on rat cardiomyocytes undergoing ischemia/reperfusion by stimulating PI3K-Akt-GSK3β pathway and preserving mitochondrial membrane potential. PLoS ONE 2017, 10, e0119790. [Google Scholar] [CrossRef]
- Lim, S.Y.; Hsiao, S.T.; Lokmic, Z.; Sivakumaran, P.; Dusting, G.J.; Dilley, R.J. Ischemic preconditioning promotes intrinsic vascularisation and enhances growth of cardiac tissue. J. Hypertens. 2012, 30 (Suppl. S1), e14. [Google Scholar] [CrossRef]
- Yu, W.; Meiping, W.; Haidong, G. Modified mRNA as a Treatment for Myocardial Infarction. Int. J. Mol. Sci. 2023, 24, 4737. [Google Scholar] [CrossRef]
- Andries, O.; De Filette, M.; De Smedt, S.C.; Demeester, J.; Van Poucke, M.; Peelman, L.; Sanders, N.N. Innate immune response and programmed cell death following carrier-mediated delivery of unmodified mRNA to respiratory cells. J. Control Release 2013, 167, 157–166. [Google Scholar] [CrossRef]
- Kaur, K.; Sultana, N.; Hadas, Y.; Magadum, A.; Sharkar, M.T.K.; Chepurko, E.; Zangi, L. Delivery of Modified mRNA in a Myocardial Infarction Mouse Model. J. Vis. Exp. 2020, 11, 160. [Google Scholar]
- Mathias, B.; Michael, S.; Ulrik, S. 2021 FDA approvals: Value driven by COVID-19 vaccines. Nat. Rev. Drug Discov. 2022, 21, 90. [Google Scholar] [CrossRef]
- Jahanafrooz, Z.; Baradaran, B.; Mosafer, J.; Hashemzaei, M.; Rezaei, T.; Mokhtarzadeh, A.; Hamblin, M.R. Comparison of DNA and mRNA vaccines against cancer. Drug Discov. Today 2020, 25, 552–560. [Google Scholar] [CrossRef]
- Li, K.; Luo, R.; Yu, X.; Dong, W.; Hao, G.; Hu, D.; Yu, Z.; Liu, M.; Lu, T.; Wang, X.; et al. Enhanced human adipose-derived stem cells with VEGFA and bFGF mRNA promote stable vascular regeneration and improve cardiac function following myocardial infarction. Clin. Transl. Med. 2025, 15, e70250. [Google Scholar] [CrossRef]
- Kowalski, P.S.; Rudra, A.; Miao, L.; Anderson, D.G. Delivering the Messenger: Advances in Technologies for Therapeutic mRNA Delivery. Mol. Ther. 2019, 27, 710–728. [Google Scholar] [CrossRef]
- Cojocaru, K.A.; Luchian, I.; Goriuc, A.; Antoci, L.M.; Ciobanu, C.G.; Popescu, R.; Vlad, C.E.; Blaj, M.; Foia, L.G. Mitochondrial dysfunction, oxidative stress, and therapeutic strategies in diabetes, obesity, and cardiovascular disease. Antioxidants 2023, 12, 658. [Google Scholar] [CrossRef]
- Lian, D.; Chen, M.M.; Wu, H.; Deng, S.; Hu, X. The Role of Oxidative Stress in Skeletal Muscle Myogenesis and Muscle Disease. Antioxidants 2022, 11, 755. [Google Scholar] [CrossRef]
- Zou, J.; Fei, Q.; Xiao, H.; Wang, H.; Liu, K.; Liu, M.; Zhang, H.; Xiao, X.; Wang, K.; Wang, N. VEGF-A promotes angiogenesis after acute myocardial infarction through increasing ROS production and enhancing ER stress-mediated autophagy. J. Cell. Physiol. 2019, 234, 17690–17703. [Google Scholar] [CrossRef]
- Verma, P.; Rishi, B.; George, N.G.; Kushwaha, N.; Dhandha, H.; Kaur, M.; Jain, A.; Jain, A.; Chaudhry, S.; Singh, A.; et al. Recent advances and future directions in etiopathogenesis and mechanisms of reactive oxygen species in cancer treatment. Pathol. Oncol. Res. 2023, 29, 1611415. [Google Scholar] [CrossRef]
- Franchina, G.D.; Dostert, C.; Brenner, D. Reactive Oxygen Species: Involvement in T Cell Signaling and Metabolism. Trends Immunol. 2018, 39, 489–502. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, Q.; Guo, Y.; Wu, J.; Zhao, L.; Wang, Z.; Gong, Z.; Kan, K.; Wang, Z. ROS-related nanoparticles for the management of Alzheimer’s disease: Wielding the double-edged sword. Chem. Eng. J. 2025, 511, 161784. [Google Scholar] [CrossRef]
- Hu, Y.; Huang, Y.; Zong, L.; Lin, J.; Liu, X.; Ning, S. Emerging roles of ferroptosis in pulmonary fibrosis: Current perspectives, opportunities and challenges. Cell Death Discov. 2024, 10, 301. [Google Scholar] [CrossRef]
- Li, X.; Ou, W.; Xie, M.; Yang, J.; Li, Q.; Li, T. Nanomedicine-Based Therapeutics for Myocardial Ischemic/Reperfusion Injury. Adv. Healthc. Mater. 2025, 12, e2300161. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, S.; Du, Y.; Gao, Z.; Li, Y.; Ma, H.; Li, H.; Ren, X.; Fan, Q.; Wu, D.; et al. Inverse Oxide/Alloy-Structured Nanozymes with NIR-Triggered Enzymatic Cascade Regulation of ROS Homeostasis for Efficient Wound Healing. Adv. Mater. 2025, 37, e2418731. [Google Scholar] [CrossRef]
- Paola, I.; Gianfranco, S. Enzymatic and Non-Enzymatic Molecules with Antioxidant Function. Antioxidants 2021, 10, 579. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, C.; Guo, M.; Zhu, F.; Yu, Z.; Zhang, W.; Li, W.; Zhang, Y.; Tian, W. Circadian Rhythm-Dependent Therapy by Composite Targeted Polyphenol Nanoparticles for Myocardial Ischemia-Reperfusion Injury. ACS Nano 2024, 18, 28154–28169. [Google Scholar] [CrossRef]
- Zhu, K.; Wang, K.; Zhang, R.; Zhu, Z.; Wang, W.; Yang, B.; Zhao, J.; Shen, Y. Iron chelators loaded on myocardiocyte mitochondria-targeted nanozyme system for treating myocardial ischemia-reperfusion injury in mouse models. J. Nanobiotechnol. 2025, 23, 112. [Google Scholar] [CrossRef]
- Nasiri, R.; Arefnezhad, R.; Baniasad, K.; Hosseini, S.A.; Jeshari, A.S.; Miri, M.; Lotfi, A.; Ghaemi, M.S.; Amini-Salehi, E.; Fatemian, H.; et al. Baicalin and baicalein against myocardial ischemia-reperfusion injury: A review of the current documents. Tissue Cell 2025, 93, 102772. [Google Scholar] [CrossRef]
- Xu, G.; Sun, X.; An, J.; Sun, F.; Zhang, C.; Williams, J.P. Ozone Protects from Myocardial Ischemia-Reperfusion Injury via Inhibition of the NLRP3 Inflammasome. Eur. J. Pharmacol. 2025, 997, 177631. [Google Scholar] [CrossRef]
- He, M.; Yang, Y.; He, X.; Lei, R.; Liu, H.; Yang, M. Melatonin inhibits ferroptosis through the ATF3/GPX4 signaling pathway to relieve myocardial ischemia-reperfusion injury in rats. Vitr. Cell. Dev. Biol. Anim. 2025, 61, 135–148. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, W.J.; Liu, X.Y.; Hu, T.P.; Gao, N.; Li, Z.H.; Wang, Y.; Zhang, G.Q. KW-2449 Ameliorates Cardiac Dysfunction in a Rat Model of Sepsis-Induced Cardiomyopathy. Inflammation 2025, 48, 2732–2744. [Google Scholar] [CrossRef]
- Deng, Y.; Liu, L.; Li, Y.; Ma, H.; Li, C.; Yan, K.; Tian, J.; Li, C. pH-sensitive nano-drug delivery systems dual-target endothelial cells and macrophages for enhanced treatment of atherosclerosis. Drug Deliv. Transl. Res. 2025, 15, 2924–2940. [Google Scholar] [CrossRef]
- Childers, G.M.; Perry, C.A.; Blachut, B.; Martin, N.; Bortner, C.D.; Sieber, S.; Li, J.L.; Fessler, M.B.; Harry, G.J. Assessing the Association of Mitochondrial Function and Inflammasome Activation in Murine Macrophages Exposed to Select Mitotoxic Tri-Organotin Compounds. Environ. Health Perspect. 2021, 129, 47015. [Google Scholar] [CrossRef]
- Aleynick, M.; Svensson-Arvelund, Z.; Flowers, C.R.; Marabelle, A.; Brody, J.D. Pathogen Molecular Pattern Receptor Agonists: Treating Cancer by Mimicking Infection. Clin. Cancer Res. 2019, 25, 6283–6294. [Google Scholar] [CrossRef]
- Linfert, D.; Chowdhry, T.; Rabb, H. Lymphocytes and ischemia-reperfusion injury. Transplant. Rev. 2008, 23, 1–10. [Google Scholar] [CrossRef]
- Ulrich, H.; Stefan, F. Role of T-cells in myocardial infarction. Eur. Heart J. 2016, 37, 873–879. [Google Scholar]
- Qin, Y.; Li, M.; Liu, H. Regulatory T cells: A promising new therapeutic target in ventricular remodeling after myocardial infarction. Front. Immunol. 2025, 16, 1514335. [Google Scholar] [CrossRef]
- Zhang, M.; Michael, L.H.; Grosjean, S.A.; Kelly, R.A.; Carroll, M.C.; Entman, M.L. The role of natural IgM in myocardial ischemia–reperfusion injury. J. Mol. Cell. Cardiol. 2006, 41, 62–67. [Google Scholar] [CrossRef]
- Garlanda, C.; Dinarello, A.C.; Mantovani, A. The Interleukin-1 Family: Back to the Future. Immunity 2013, 39, 1003–1018. [Google Scholar] [CrossRef]
- Liao, W.; Lin, J.; Leonard, J.W. IL-2 family cytokines: New insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Curr. Opin. Immunol. 2011, 23, 598–604. [Google Scholar] [CrossRef]
- Gu, C.; Wu, L.; Li, X. IL-17 family: Cytokines, receptors and signaling. Cytokine 2013, 64, 477–485. [Google Scholar] [CrossRef]
- Gong, G.; Wan, W.; Zhang, X.; Chen, X.; Yin, J. Management of ROS and Regulatory Cell Death in Myocardial Ischemia–Reperfusion Injury. Mol. Biotechnol. 2025, 67, 1765–1783. [Google Scholar] [CrossRef]
- Tai, P.; Chen, X.; Jia, G.; Chen, G.; Gong, L.; Cheng, Y.; Li, Z.; Wang, H.; Chen, A.; Zhang, G.; et al. WGX50 mitigates doxorubicin-induced cardiotoxicity through inhibition of mitochondrial ROS and ferroptosis. J. Transl. Med. 2023, 21, 823. [Google Scholar] [CrossRef]
- Ren, Y.; Wang, R.; Weng, S.; Xu, H.; Zhang, Y.; Chen, S.; Liu, S.; Ba, Y.; Zhou, Z.; Luo, P.; et al. Multifaceted role of redox pattern in the tumor immune microenvironment regarding autophagy and apoptosis. Mol. Cancer 2023, 22, 130. [Google Scholar] [CrossRef]
- Rusiecka, O.M.; Molica, F.; Nielsen, M.S.; Tollance, A.; Morel, S.; Frieden, M.; Chanson, M.; Boengler, K.; Kwak, B.R. Mitochondrial pannexin1 controls cardiac sensitivity to ischaemia/reperfusion injury. Cardiovasc. Res. 2023, 119, 2342–2354. [Google Scholar] [CrossRef]
- Swirski, F.K.; Nahrendorf, M. Cardioimmunology: The immune system in cardiac homeostasis and disease. Nat. Rev. Immunol. 2018, 18, 733–744. [Google Scholar] [CrossRef]
- Peng, J.F.; Salami, O.M.; Habimana, O.; Xie, Y.X.; Yao, H.; Yi, G.H. Targeted Mitochondrial Drugs for Treatment of Ischemia-Reperfusion Injury. Curr. Drug Targets 2022, 23, 1526–1536. [Google Scholar] [CrossRef]
- Mastoor, Y.; Murphy, E.; Roman, B. Mechanisms of postischemic cardiac death and protection following myocardial injury. J. Clin. Investig. 2025, 135, e184134. [Google Scholar] [CrossRef]
- Singh, G.; Vengayil, V.; Khanna, A.; Adhikary, S.; Laxman, S. Active control of mitochondrial network morphology by metabolism-driven redox state. Proc. Natl. Acad. Sci. USA 2025, 122, e2421953122. [Google Scholar] [CrossRef]
- Rozich, E.; Ozkurede, U.; Pakkiriswami, S.; Gemilere, R.; Azarin, S.M.; Liu, J.C. Mitochondrial oxidative stress, calcium and dynamics in cardiac ischaemia-reperfusion injury. J. Physiol. 2025. ahead of print. [Google Scholar] [CrossRef]
- Bai, Y.; Wu, J.; Yang, Z.; Wang, X.; Zhang, D.; Ma, J. Mitochondrial quality control in cardiac ischemia/reperfusion injury: New insights into mechanisms and implications. Cell Biol. Toxicol. 2022, 39, 33–51. [Google Scholar] [CrossRef]
- He, Y.; Ren, S.; Liu, C.; Zheng, X.; Zhu, C. Targeting mitochondria quality control for myocardial ischemia-reperfusion injury. Mitochondrion 2025, 84, 102046. [Google Scholar] [CrossRef]
- Li, C.; Ma, Q.; Toan, S.; Wang, J.; Zhou, H.; Liang, J. SERCA overexpression reduces reperfusion-mediated cardiac microvascular damage through inhibition of the calcium/MCU/mPTP/necroptosis signaling pathways. Redox Biol. 2020, 36, 101659. [Google Scholar] [CrossRef]
- Grandi, E.; Navedo, M.F.; Saucerman, J.J.; Bers, D.M.; Chiamvimonvat, N.; Dixon, R.E.; Dobrev, D.; Gomez, A.M.; Harraz, O.F.; Hegyi, B.; et al. Diversity of Cells and Signals in the Cardiovascular System. J. Physiol. 2023, 601, 2547–2592. [Google Scholar] [CrossRef]
- Tsurusaki, S.; Kizana, E. Mechanisms and Therapeutic Potential of Multiple Forms of Cell Death in Myocardial Ischemia–Reperfusion Injury. Int. J. Mol. Sci. 2024, 25, 13492. [Google Scholar] [CrossRef]
- Yin, F.; Zhou, H.; Fang, Y.; Li, C.; He, Y.; Yu, L.; Wan, H.; Yang, J. Astragaloside IV alleviates ischemia reperfusion-induced apoptosis by inhibiting the activation of key factors in death receptor pathway and mitochondrial pathway. J. Ethnopharmacol. 2020, 248, 112319. [Google Scholar] [CrossRef]
- Luo, Y.; Liao, S.; Wu, J.; Maneechote, C.; Arunsak, B.; Apaijai, N.; Benjanuwattra, J.; Chattipakorn, S.; Chattipakorn, N. Abstract 9481: Inhibition of Apoptosis and Ferroptosis Signaling Pathways Alleviates Myocardial Ischemia-Reperfusion Injury in Rats Through Modulation of Mitochondrial Function. Circulation 2021, 144, A9481. [Google Scholar] [CrossRef]
- Liu, H.; Li, S.; Jiang, W.; Li, Y. MiR-484 Protects Rat Myocardial Cells from Ischemia-Reperfusion Injury by Inhibiting Caspase-3 and Caspase-9 during Apoptosis. Korean Circ. J. 2020, 50, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Palikaras, K.; Lionaki, E.; Tavernarakis, N.J.C.D. Balancing mitochondrial biogenesis and mitophagy to maintain energy metabolism homeostasis. Cell Death Differ. 2015, 22, 1399–1401. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.M.; Hurley, H.J. Structure and function of the ULK1 complex in autophagy. Curr. Opin. Cell Biol. 2016, 39, 3961–3968. [Google Scholar] [CrossRef]
- Tian, J.; Niu, Z.; Yang, H.; Wang, C.; Guan, L.; Zhao, L.; Shi, D.; Zhang, Z. PERK/Sestrin2 Signaling Pathway Mediated Autophagy Regulates Human Cardiomyocytes Apoptosis Induced by Traffic-Related PM2.5 and Diverse Constituents. Int. J. Mol. Sci. 2025, 26, 3784. [Google Scholar] [CrossRef]
- Claudia, R.; Mercedes, M.; Cristina, C.; Prieto, D. AMPK, metabolism, and vascular function. FEBS J. 2021, 288, 3746–3771. [Google Scholar] [CrossRef]
- Xia, B.; Liang, J.; Lu, Y.; Ding, J.; Peng, J.; Li, F.; Dai, J.; Liu, Y.; Wang, J.; Wan, C.; et al. Lactoferrin influences atherosclerotic progression by modulating macrophagic AMPK/mTOR signaling-dependent autophagy. Sci. Rep. 2025, 15, 10585. [Google Scholar] [CrossRef]
- Ying, Z.; Yue, W.; Junnan, X.; Tian, F.; Hu, S.; Chen, Y.; Fu, Z. Melatonin attenuates myocardial ischemia-reperfusion injury via improving mitochondrial fusion/mitophagy and activating the AMPK-OPA1 signaling pathways. J. Pineal Res. 2019, 66, e12542. [Google Scholar]
- Poole, L.P.; Bock-Hughes, A.; Berardi, D.E.; Macleod, K.F. ULK1 promotes mitophagy via phosphorylation and stabilization of BNIP3. Sci. Rep. 2021, 11, 20526. [Google Scholar] [CrossRef]
- Zhian, C.; Tianying, L.; Hao, Y.; Sun, H.; Liu, S.; Zhang, S.; Liu, L.; Jiang, S.; Tang, Y.; Liu, Z. Bioinformatics integration reveals key genes associated with mitophagy in myocardial ischemia-reperfusion injury. BMC Cardiovasc. Disord. 2024, 24, 183. [Google Scholar] [CrossRef]
- Yuping, L.; Ting, L.; Tiantian, L.; Zhang, D.; Du, S.; Girani, L.; Qi, D.; Lin, C.; Tong, R.; Wang, Y. RIP1/RIP3-regulated necroptosis as a target for multifaceted disease therapy (Review). Int. J. Mol. Med. 2019, 44, 771–786. [Google Scholar]
- Yang, Z.; Li, C.; Xue, H.; Wang, Y.; Ma, H. Abstract 558: Melatonin Exerts Cardioprotective Effects Under Chronic Neuropathic Pain Through Rip1-rip3-mlkl and Rip3-camkii Pathway Mediating Myocardial Necroptosis in Ischemia Reperfusion Injury. Circ. Res. 2018, 123, A558. [Google Scholar] [CrossRef]
- Meade, J.J.; Stuart, S.; Zenevich, N.J.; Krustev, C.; Girardin, S.E.; Mogridge, J. Activation of the NLRP1B inflammasome by caspase-8. Commun. Biol. 2024, 7, 1164. [Google Scholar] [CrossRef] [PubMed]
- Bei, Y.; Das, S.; Rodosthenous, R.S.; Holvoet, P.; Vanhaverbeke, M.; Monteiro, M.C.; Monteiro, V.V.S.; Radosinska, J.; Bartekova, M.; Jansen, F.; et al. Extracellular Vesicles in Cardiovascular Theranostics. Theranostics 2017, 7, 4168–4182. [Google Scholar] [CrossRef]
- Ingrid, G.; Jifu, J.; Dameng, L.; Huang, X.; Fuhrmann, B.; Liu, W.; Haig, A.; Jevnikar, A.M.; Zhang, Z.X. Mitochondrial permeability regulates cardiac endothelial cell necroptosis and cardiac allograft rejection. Am. J. Transplant. 2019, 19, 686–698. [Google Scholar]
- Poulos, M.G.; Batra, R.; Charizanis, K.; Swanson, M.S. Developments in RNA splicing and disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a000778. [Google Scholar] [CrossRef]
- Sultana, N.; Hadas, Y.; Sharkar, K.T.M.; Kaur, K.; Magadum, A.; Kurian, A.A.; Hossain, N.; Alburquerque, B.; Ahmed, S.; Chepurko, E.; et al. Optimization of 5′ Untranslated Region of Modified mRNA for Use in Cardiac or Hepatic Ischemic Injury. Mol. Ther. Methods Clin. Dev. 2020, 17, 622–633. [Google Scholar] [CrossRef]
- Dirisala, A.; Uchida, S.; Toh, K.; Li, J.; Liu, X.; Wen, P.; Fukushima, S.; Kataoka, K. Structural stability and RNase resistance of mRNA Polyplex micelles for systemic delivery. J. Control Release 2025, 384, 113935. [Google Scholar] [CrossRef]
- Longfei, J.; Yuanhui, M.; Quanquan, J.; Dersh, D.; Yewdell, J.W.; Qian, S.B. Decoding mRNA translatability and stability from the 5’ UTR. Nat. Struct. Mol. Biol. 2020, 27, 814–821. [Google Scholar] [CrossRef]
- Kran, S.; Lihong, T.; Vera, B.; D’Souza, S.S.; Kumar, A.; Slukvin, I. Optimization of Synthetic mRNA for Highly Efficient Translation and its Application in the Generation of Endothelial and Hematopoietic Cells from Human and Primate Pluripotent Stem Cells. Stem Cell Rev. Rep. 2018, 14, 525–534. [Google Scholar] [CrossRef]
- Imbriano, C.; Moresi, V.; Belluti, S.; Renzini, A.; Cavioli, G.; Maretti, E.; Molinari, S. Epitranscriptomics as a New Layer of Regulation of Gene Expression in Skeletal Muscle: Known Functions and Future Perspectives. Int. J. Mol. Sci. 2023, 24, 15161. [Google Scholar] [CrossRef]
- Chen, R.; Wang, S.K.; Belk, J.A.; Amaya, L.; Li, Z.; Cardenas, A.; Abe, B.T.; Chen, C.K.; Wender, P.A.; Chang, H. Engineering circular RNA for enhanced protein production. Nat. Biotechnol. 2022, 41, 262–272, Erratum in Nat. Biotechnol. 2023, 41, 293. [Google Scholar] [CrossRef] [PubMed]
- Eisen, J.T.; Eichhorn, W.S.; Subtelny, O.A.; Lin, K.S.; McGeary, S.E.; Gupta, S.; Bartel, D.P. The Dynamics of Cytoplasmic mRNA Metabolism. Mol. Cell 2020, 77, 786–799.e10. [Google Scholar] [CrossRef] [PubMed]
- Mangus, D.A.; Evans, M.C.; Jacobson, A. Poly(A)-binding proteins: Multifunctional scaffolds for the post-transcriptional control of gene expression. Genome Biol. 2003, 4, 223. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Liu, D.; Guo, J.; Aditham, A.; Zhou, Y.; Tian, J.; Luo, S.; Ren, J.; Hsu, A.; Huang, J.; et al. Branched chemically modified poly(A) tails enhance the translation capacity of mRNA. Nat. Biotechnol. 2024, 43, 194–203. [Google Scholar] [CrossRef]
- Ibrahim, A.; Twizeyimana, E.; Lu, N.; Ke, W.; Mukerabigwi, J.F.; Mohammed, F.; Japir, A.A.M.M.; Ge, Z. Reduction-Responsive Polymer Prodrug Micelles with Enhanced Endosomal Escape Capability for Efficient Intracellular Translocation and Drug Release. ACS Appl. Bio Mater. 2019, 2, 5099–5109. [Google Scholar] [CrossRef]
- Xin, M.; Sun, H. Immunogenicity of In Vitro-Transcribed RNA. Acc. Chem. Res. 2021, 54, 4012–4023. [Google Scholar]
- Ni, R.; Zhou, J.; Hossain, N.; Chau, Y. Virus-inspired nucleic acid delivery system: Linking virus and viral mimicry. Adv. Drug Deliv. Rev. 2016, 106, 3–26. [Google Scholar] [CrossRef]
- Yanfang, W.; Ernst, W. Non-Viral Targeted Nucleic Acid Delivery: Apply Sequences for Optimization. Pharmaceutics 2020, 12, 888. [Google Scholar] [CrossRef]
- Jia, F.; Huang, W.; Yin, Y.; Jiang, Y.; Yang, Q.; Huang, H.; Nie, G.; Wang, H. Stabilizing RNA Nanovaccines with Transformable Hyaluronan Dynamic Hydrogel for Durable Cancer Immunotherapy. Adv. Funct. Mater. 2022, 33, 2204636. [Google Scholar] [CrossRef]
- Sultana, N.; Magadum, A.; Hadas, Y.; Kondrat, J.; Singh, N.; Youssef, E.; Calderon, D.; Chepurko, E.; Dubois, N.; Hajjar, R.J.; et al. Optimizing Cardiac Delivery of Modified mRNA. Mol. Ther. 2017, 25, 1306–1315. [Google Scholar] [CrossRef] [PubMed]
- Ting, L.; Gangfeng, L.; Guolong, B.; Xu, Y.; He, C.; Zhao, G. Optimizing mRNA translation efficiency through rational 5′UTR and 3′UTR combinatorial design. Gene 2025, 942, 149254. [Google Scholar] [CrossRef] [PubMed]
- Setare, A.; Majid, F.; Ali, M.T.; Miri, M.R.; Rafiei Dehbidi, G.; Farhadi, A.; Ranjbaran, R.; Alavi, P.; Nikouyan, N.; Seyyedi, N.; et al. Enhancing Stability of Destabilized Green Fluorescent Protein Using Chimeric mRNA Containing Human Beta-Globin 5’ and 3’ Untranslated Regions. Avicenna J. Med. Biotechnol. 2019, 11, 112–117. [Google Scholar]
- Mokhtari, B.; Aboutaleb, N.; Nazarinia, D.; Nikougoftar, M.; Razavi Tousi, S.M.T.; Molazem, M.; Azadi, M.R. Comparison of the effects of intramyocardial and intravenous injections of human mesenchymal stem cells on cardiac regeneration after heart failure. Iran. J. Basic. Med. Sci. 2020, 23, 879–885. [Google Scholar]
- Dror, L.; Lipinski, M.J.; Westman, P.C.; Glover, D.K.; Dimastromatteo, J.; Frias, J.C.; Albelda, M.T.; Sikora, S.; Kharazi, A.; Vertelov, G.; et al. Intravenously Delivered Mesenchymal Stem Cells: Systemic Anti-Inflammatory Effects Improve Left Ventricular Dysfunction in Acute Myocardial Infarction and Ischemic Cardiomyopathy. Circ. Res. 2017, 120, 1598–1613. [Google Scholar]
- Michael, D.; Michael, U.; Rolf, B.; Randau, L. RIP-Seq Suggests Translational Regulation by L7Ae in Archaea. mBio 2017, 8, e00730-17. [Google Scholar]
- Ajit, M.; Yishu, D.; Lan, H.; Kim, T.; Vasudevarao, M.D.; Long, Q.; Yang, K.; Wickramasinghe, N.; Renikunta, H.V.; Dubois, N.; et al. Live cell screening platform identifies PPARδ as a regulator of cardiomyocyte proliferation and cardiac repair. Cell Res. 2017, 27, 1002–1019. [Google Scholar] [CrossRef]
- Kang, D.D.; Hou, X.; Wang, L.; Xue, Y.; Li, H.; Zhong, Y.; Wang, S.; Deng, B.; McComb, D.W.; Dong, Y. Engineering LNPs with polysarcosine lipids for mRNA delivery. Bioact. Mater. 2024, 37, 3786–3793. [Google Scholar] [CrossRef]
- Kong, W.; Wei, Y.; Dong, Z.; Liu, W.; Zhao, J.; Huang, Y.; Yang, J.; Wu, W.; He, H.; Qi, J. Role of size, surface charge, and PEGylated lipids of lipid nanoparticles (LNPs) on intramuscular delivery of mRNA. J. Nanobiotechnol. 2024, 22, 553. [Google Scholar] [CrossRef]
- Labonia, I.C.M.; Senti, E.M.; Kraak, D.V.H.P.; Brans, M.A.D.; Dokter, I.; Streef, T.J.; Smits, A.M.; Deshantri, A.K.; de Jager, S.C.A.; Schiffelers, R.M.; et al. Cardiac delivery of modified mRNA using lipid nanoparticles: Cellular targets and biodistribution after intramyocardial administration. J. Control Release 2024, 369, 734–745. [Google Scholar] [CrossRef]
- van den Berg, A.I.S.; Yun, C.O.; Schiffelers, R.M.; Hennink, W.E. Polymeric delivery systems for nucleic acid therapeutics: Approaching the clinic. J. Control Release 2021, 331, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Zheng, T.; Li, M.; Zhong, X.; Tang, Y.; Qin, M.; Sun, X. Optimization of an mRNA vaccine assisted with cyclodextrin-polyethyleneimine conjugates. Drug Deliv. Transl. Res. 2020, 10, 678–689. [Google Scholar] [CrossRef] [PubMed]
- Maguire, C.A.; Balaj, L.; Sivaraman, S.; Crommentuijn, M.H.; Ericsson, M.; Mincheva-Nilsson, L.; Baranov, V.; Gianni, D.; Tannous, B.A.; Sena-Esteves, M.; et al. Microvesicle-associated AAV vector as a novel gene delivery system. Mol. Ther. J. Am. Soc. Gene Ther. 2012, 20, 960–971. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Guangyu, X.; Zhidi, L.; Zou, F.; Liu, S.; Zhang, Y.; Fu, W.; Jiang, J.; Ma, X.; Song, J. Engineered exosomes enriched in netrin-1 modRNA promote axonal growth in spinal cord injury by attenuating inflammation and pyroptosis. Biomater. Res. 2023, 27, 3. [Google Scholar] [CrossRef]
- Ma, T.; Xu, G.; Gao, T.; Zhao, G.; Huang, G.; Shi, J.; Chen, J.; Song, J.; Xia, J.; Ma, X. Engineered Exosomes with ATF5-Modified mRNA Loaded in Injectable Thermogels Alleviate Osteoarthritis by Targeting the Mitochondrial Unfolded Protein Response. ACS Appl. Mater. Interfaces 2024, 16, 21383–21399. [Google Scholar] [CrossRef]
- You, Y.; Tian, Y.; Guo, R.; Shi, J.; Kwak, K.J.; Tong, Y.; Estania, A.P.; Hsu, W.H.; Liu, Y.; Hu, S.; et al. Extracellular vesicle-mediated VEGF-A mRNA delivery rescues ischaemic injury with low immunogenicity. Eur. Heart J. 2025, 46, 1662–1676. [Google Scholar] [CrossRef]
- Iwona, D. Stem Cell-Based Therapy and Cell-Free Therapy as an Alternative Approach for Cardiac Regeneration. Stem Cells Int. 2023, 2023, 2729377. [Google Scholar]
- Trounson, A.; McDonald, C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem Cell 2015, 17, 11–22. [Google Scholar] [CrossRef]
- Magadum, A.; Kaur, K.; Zangi, L. mRNA-Based Protein Replacement Therapy for the Heart. Mol. Ther. 2019, 27, 785–793. [Google Scholar] [CrossRef]
- Magadum, A.; Singh, N.; Kurian, A.A.; Sharkar, M.T.K.; Sultana, N.; Chepurko, E.; Kaur, K.; Żak, M.M.; Hadas, Y.; Lebeche, D.; et al. Therapeutic Delivery of Pip4k2c-Modified mRNA Attenuates Cardiac Hypertrophy and Fibrosis in the Failing Heart. Adv. Sci. 2021, 8, 2004661. [Google Scholar] [CrossRef]
- Gao, J.; Liu, J.; Meng, Z.; Li, Y.; Hong, Y.; Wang, L.; He, L.; Hu, B.; Zheng, Y.; Li, T.; et al. Ultrasound-assisted C3F8-filled PLGA nanobubbles for enhanced FGF21 delivery and improved prophylactic treatment of diabetic cardiomyopathy. Acta Biomater. 2021, 130, 395–408. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jiang, H.; Xie, W.; Qian, B.; Long, Q.; Qi, Z.; Huang, S.; Zhong, Y.; Zhang, Y.; Chang, L.; et al. LNPs-mediated VEGF-C mRNA delivery promotes heart repair and attenuates inflammation by stimulating lymphangiogenesis post-myocardial infarction. Biomaterials 2025, 322, 123410. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Deng, X.; Liu, Y.; Sun, L.; Chen, F. PKM2, function and expression and regulation. Cell Biosci. 2019, 9, 52. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.C.; Sofia, M.D.; Thomas, B.; Qin, J.; van Mil, A.; Doevendans, P.A.; Chamuleau, S.A.J.; Sluijter, J.P.G.; Neef, K. Follistatin-like 1 promotes proliferation of matured human hypoxic iPSC-cardiomyocytes and is secreted by cardiac fibroblasts. Mol. Ther. Methods Clin. Dev. 2022, 25, 13–16. [Google Scholar] [CrossRef]
- Ajit, M.; Neha, S.; Anu, A.K.; Munir, I.; Mehmood, T.; Brown, K.; Sharkar, M.T.K.; Chepurko, E.; Sassi, Y.; Oh, J.G.; et al. Pkm2 Regulates Cardiomyocyte Cell Cycle and Promotes Cardiac Regeneration. Circulation 2020, 141, 1249–1265. [Google Scholar] [CrossRef]
- Magadum, A.; Sun, J.; Singh, N.; Kurian, A.A.; Chepurko, E.; Fargnoli, A.; Hajjar, R.; Zhang, J.; Zangi, L. Lin28a cardiomyocyte-specific modified mRNA translation system induces cardiomyocyte cell division and cardiac repair. J. Mol. Cell. Cardiol. 2024, 188, 61–64. [Google Scholar] [CrossRef]
- Collén, A.; Bergenhem, N.; Carlsson, L.; Chien, K.R.; Hoge, S.; Gan, L.M.; Fritsche-Danielson, R. VEGFA mRNA for regenerative treatment of heart failure. Nat. Rev. Drug Discov. 2022, 21, 79–80. [Google Scholar] [CrossRef]
- Carlsson, L.; Clarke, C.J.; Yen, C.; Gregoire, F.; Albery, T.; Billger, M.; Egnell, A.C.; Gan, L.M.; Jennbacken, K.; Johansson, E.L.; et al. Biocompatible, Purified VEGF-A mRNA Improves Cardiac Function after Intracardiac Injection 1 Week Post-myocardial Infarction in Swine. Mol. Ther. Methods Clin. Dev. 2018, 9, 330–346. [Google Scholar] [CrossRef]
- Lui, K.O.; Zangi, L.; Silva, E.A.; Bu, L.; Sahara, M.; Li, R.A.; Mooney, D.J.; Chien, K.R. Driving vascular endothelial cell fate of human multipotent Isl1+ heart progenitors with VEGF modified mRNA. Cell Res. 2013, 23, 1172–1186. [Google Scholar] [CrossRef]
- Huang, C.-L.; Leblond, A.-L.; Turner, E.C.; Kumar, A.H.; Martin, K.; Whelan, D.; O’Sullivan, D.M.; Caplice, N.M. Synthetic chemically modified mrna-based delivery of cytoprotective factor promotes early cardiomyocyte survival post-acute myocardial infarction. Mol. Pharm. 2015, 12, 991–996. [Google Scholar] [CrossRef]
- Jung, H.J.; Suh, Y. Regulation of IGF -1 signaling by microRNAs. Front. Genet. 2014, 5, 472. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; John, L.; Jun, W.; Martin, J.F. Hippo/Yap Signaling in Cardiac Development and Regeneration. Curr. Treat. Opt. Cardiovasc. Med. 2016, 18, 38. [Google Scholar]
- Chen, C.; Ma, Q.; King, J.S.; Sun, Y.; Xu, B.; Zhang, X.; Zohrabian, S.; Guo, H.; Cai, W.; Li, G.; et al. aYAP modRNA reduces cardiac inflammation and hypertrophy in a murine ischemia-reperfusion model. Life Sci. Alliance 2020, 3, e201900424. [Google Scholar] [CrossRef] [PubMed]
- Yunan, G.; Yan, S.; Gulhan, A.E.; King, J.S.; Akerberg, B.N.; Ma, Q.; Kontaridis, M.I.; Pu, W.T.; Lin, Z. YAP/TEAD1 Complex Is a Default Repressor of Cardiac Toll-Like Receptor Genes. Int. J. Mol. Sci. 2021, 22, 6649. [Google Scholar]
- Zhang, Z.; Ma, B.; Li, B.; Li, Z.; Gao, M.; Zhao, H.; Peng, R.; Hu, J.; Wang, Y.; You, W.; et al. Cardiolipin-mimic lipid nanoparticles without antibody modification delivered senolytic in vivo CAR-T therapy for inflamm-aging. Cell Rep. Med. 2025, 6, 102209. [Google Scholar] [CrossRef]
- Aghajanian, H.; Kimura, T.; Rurik, J.G.; Hancock, A.S.; Leibowitz, M.S.; Li, L.; Scholler, J.; Monslow, J.; Lo, A.; Han, W.; et al. Targeting cardiac fibrosis with engineered T cells. Nature 2019, 573, 430–433, Erratum in Nature 2019, 576, E2. [Google Scholar] [CrossRef]
- Rurik, J.G.; Tombácz, I.; Yadegari, A.; Méndez Fernández, P.O.; Shewale, S.V.; Li, L.; Kimura, T.; Soliman, O.Y.; Papp, T.E.; Tam, Y.K.; et al. CAR T cells produced in vivo to treat cardiac injury. Science 2022, 375, 91–96. [Google Scholar] [CrossRef]
- Du, H.; You, X.; Zhang, J.; Liu, S.; Zhou, Y.; Wang, Y.; Yang, C.; Meng, Y.; Liu, X.; Zhang, H.; et al. CAR Macrophages Engineered In Vivo for Attenuating Myocardial Ischemia-Reperfusion Injury. Circ. Res. 2025, 137, 846–859. [Google Scholar] [CrossRef]
- Wang, A.Y.L.; Chang, Y.-C.; Che, K.-H.; Loh, C.Y.Y. Potential Application of Modified mRNA in Cardiac Regeneration. Cell Transplant. 2024, 33, 9636897241248956. [Google Scholar] [CrossRef]
- Degors, I.M.S.; Wang, C.; Rehman, Z.R.; Zuhorn, I.S. Carriers Break Barriers in Drug Delivery: Endocytosis and Endosomal Escape of Gene Delivery Vectors. Acc. Chem. Res. 2019, 52, 1750–1760. [Google Scholar] [CrossRef]
- Haoyang, L.; Hui, X.; Manyu, D.; Xue, Y.; Zhao, R. Britanin relieves ferroptosis-mediated myocardial ischaemia/reperfusion damage by upregulating GPX4 through activation of AMPK/GSK3β/Nrf2 signaling. Pharm. Biol. 2022, 60, 38–45. [Google Scholar]
- Wang, X.; Chen, T.; Chen, S.; Zhang, J.; Cai, L.; Liu, C.; Zhang, Y.; Wu, X.; Li, N.; Ma, Z.; et al. STING aggravates ferroptosis-dependent myocardial ischemia-reperfusion injury by targeting GPX4 for autophagic degradation. Signal Transduct. Target. Ther. 2025, 10, 136. [Google Scholar] [CrossRef]
- Zhang, T.; Zhu, B.; Deng, S.; Qin, J.; Zhang, J.; Meng, S. SDF-1α mRNA therapy in peripheral artery disease. Angiogenesis 2025, 28, 26. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cai, T.; Yang, X.-Q. Modified mRNA-Based Therapeutic Strategies for Myocardial Ischemia–Reperfusion Injury. Int. J. Mol. Sci. 2026, 27, 55. https://doi.org/10.3390/ijms27010055
Cai T, Yang X-Q. Modified mRNA-Based Therapeutic Strategies for Myocardial Ischemia–Reperfusion Injury. International Journal of Molecular Sciences. 2026; 27(1):55. https://doi.org/10.3390/ijms27010055
Chicago/Turabian StyleCai, Ting, and Xiang-Qun Yang. 2026. "Modified mRNA-Based Therapeutic Strategies for Myocardial Ischemia–Reperfusion Injury" International Journal of Molecular Sciences 27, no. 1: 55. https://doi.org/10.3390/ijms27010055
APA StyleCai, T., & Yang, X.-Q. (2026). Modified mRNA-Based Therapeutic Strategies for Myocardial Ischemia–Reperfusion Injury. International Journal of Molecular Sciences, 27(1), 55. https://doi.org/10.3390/ijms27010055





