Oxygen-Mediated Molecular Mechanisms Involved in Intestinal Ischemia and Reperfusion Injury
Abstract
1. Introduction
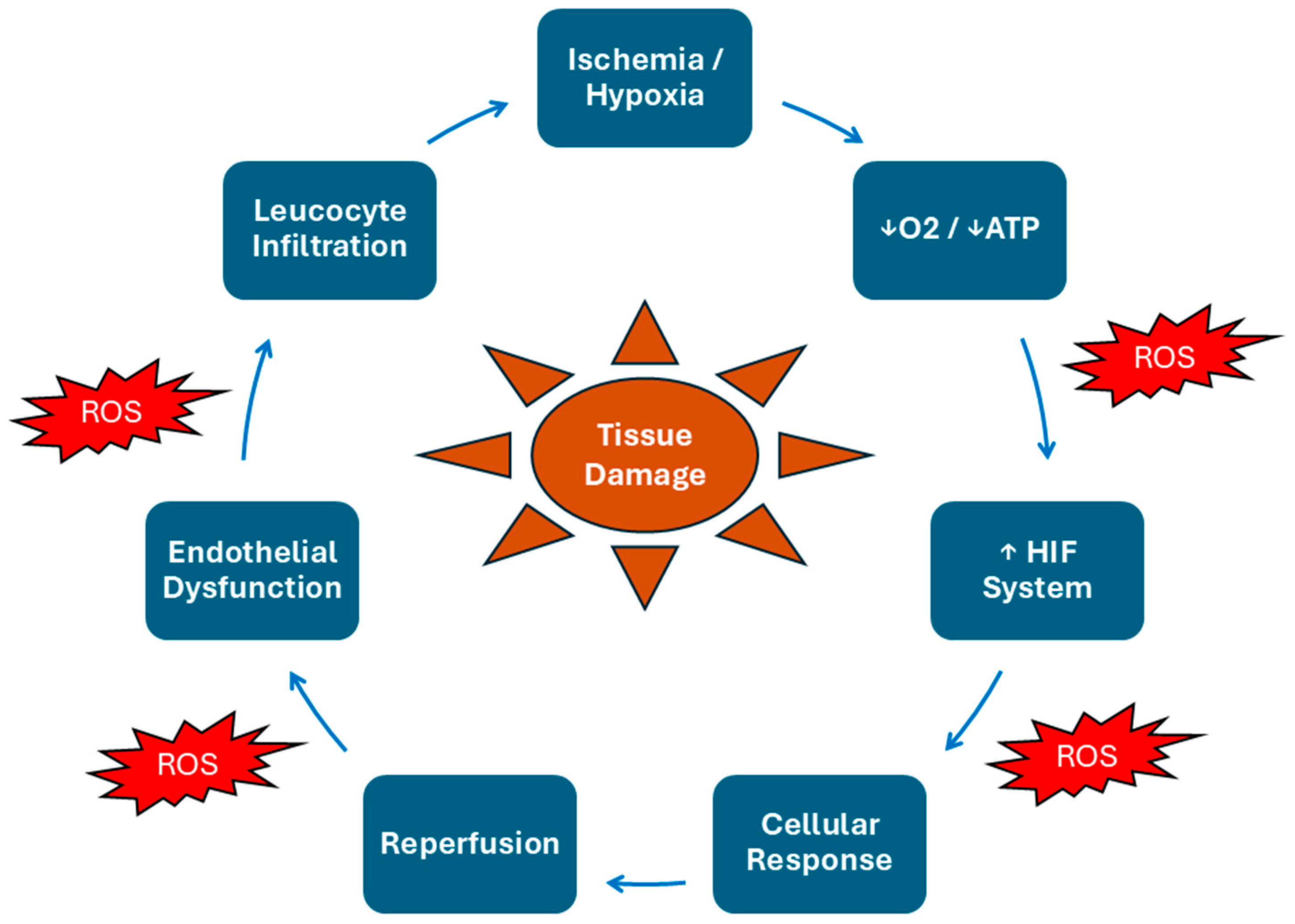
2. Natural History of IRI
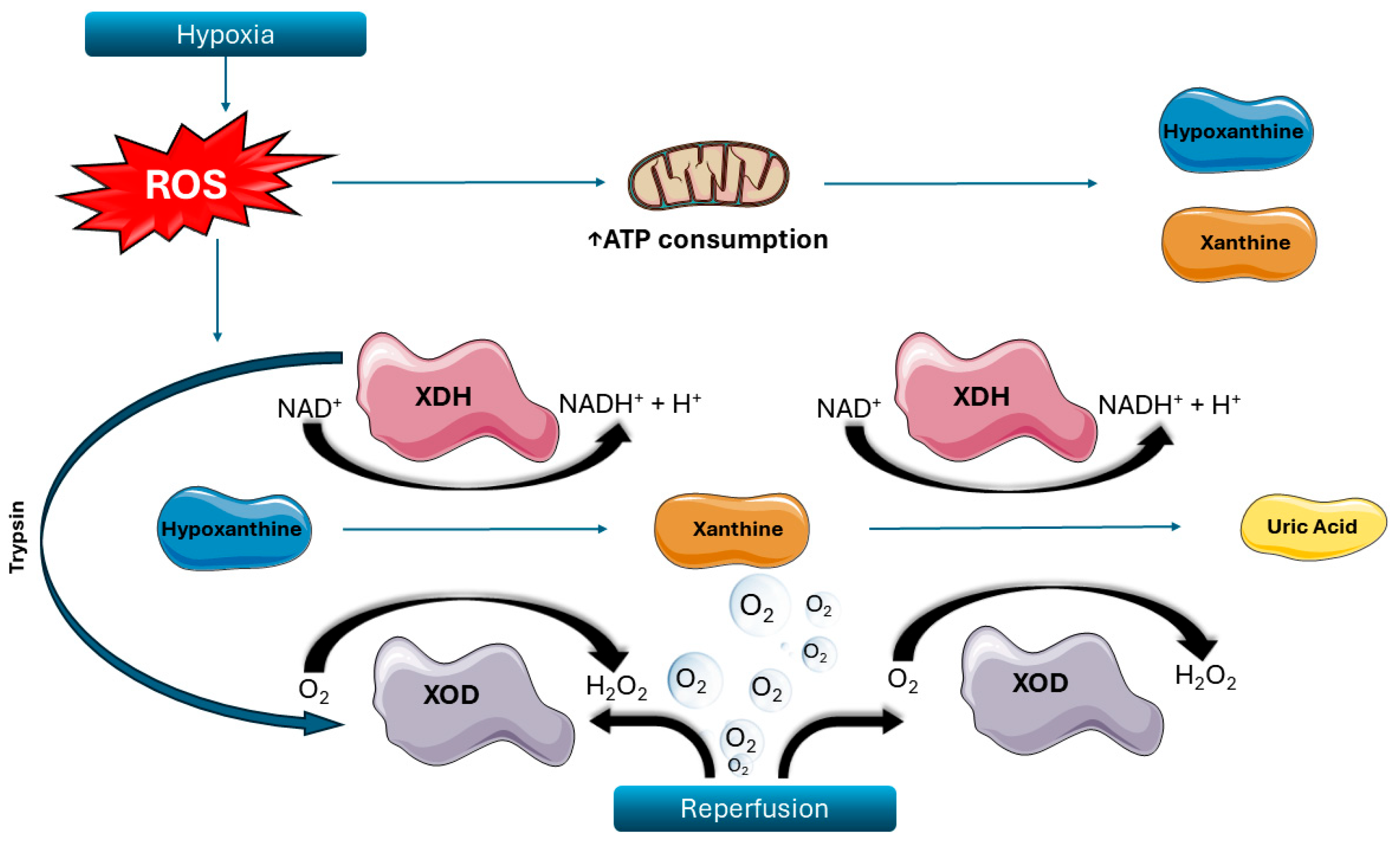
3. Biochemical Mechanisms in IRI
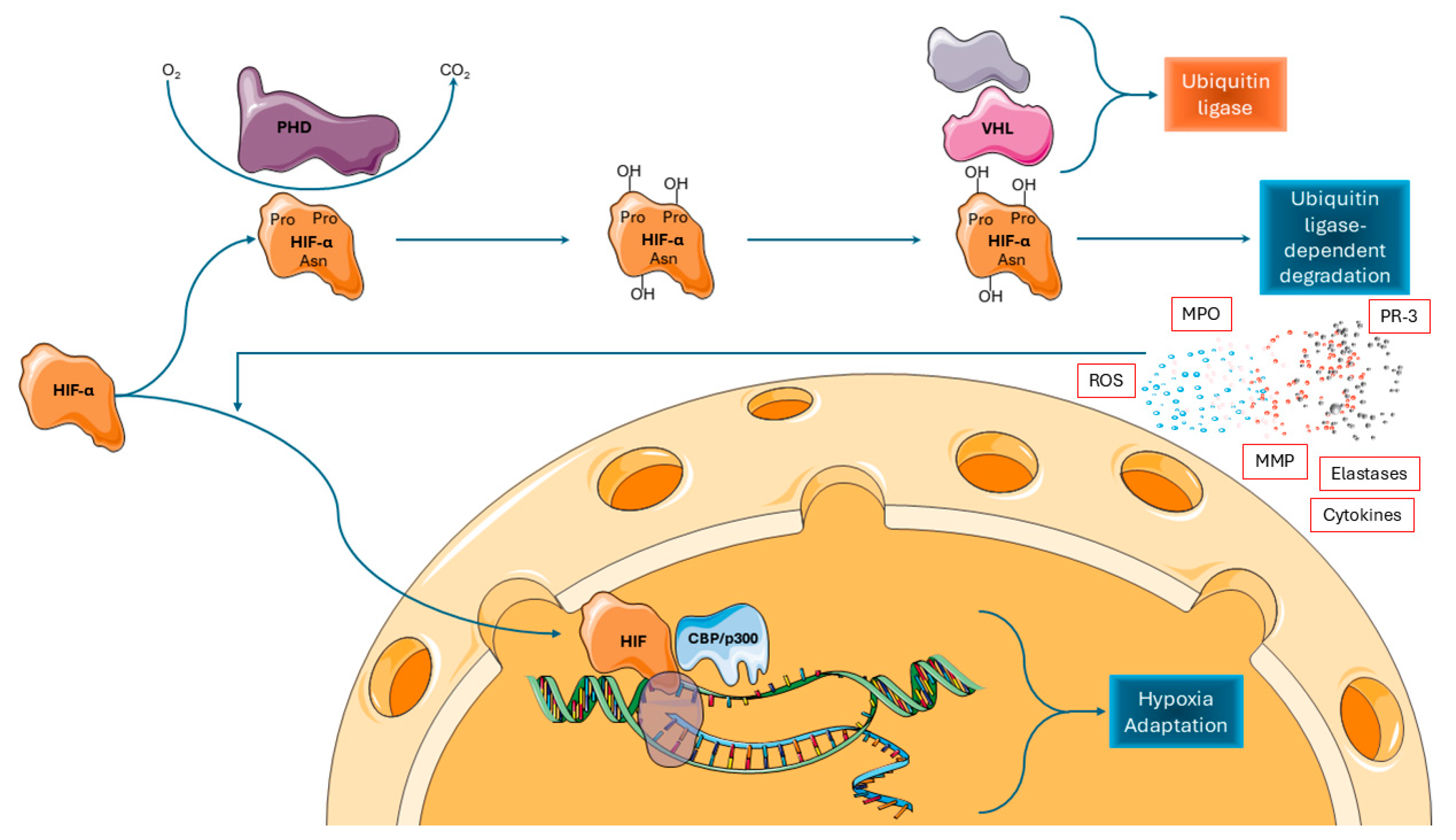
4. The Hypoxia-Inducible Factor System
5. Intestinal Epithelial Cellular Mechanisms in IRI
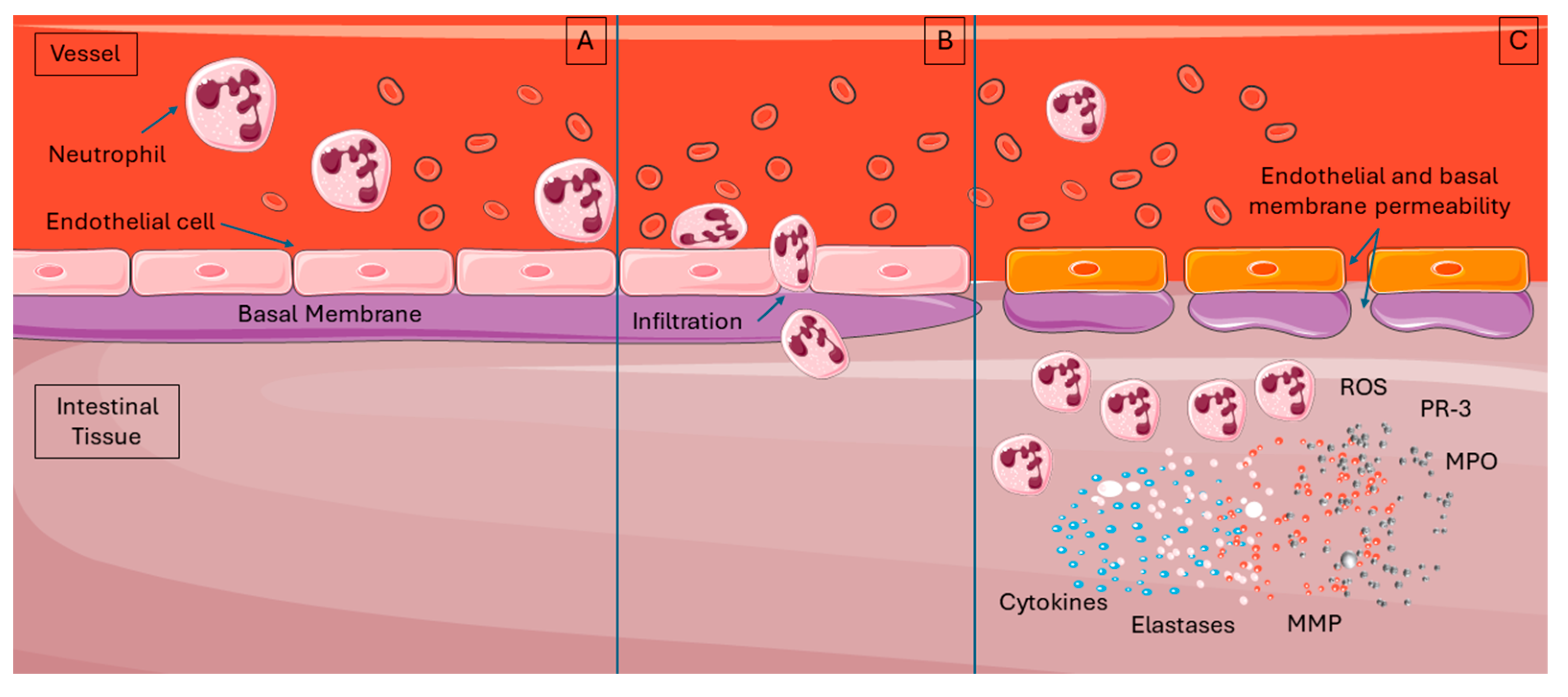
6. Immune Cellular Mechanisms in IRI
7. Microcirculation and Tissue Changes in IRI
8. Clinical and Experimental Indicators for Diagnosing and Monitoring Intestinal Ischemia and Reperfusion
8.1. Criteria for Defining the Ischemic Phase
8.2. Criteria for Irreversible Ischemia
8.3. Monitoring Intestinal Oxygenation During Ischemic Insult
8.4. Diagnostic Indicators of Reperfusion Quality
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kalogeris, T.; Baines, C.P.; Krenz, M.; Korthuis, R.J. Ischemia/Reperfusion. Compr. Physiol. 2016, 7, 113–170. [Google Scholar] [CrossRef]
- Zhao, W.; Gan, X.; Su, G.; Wanling, G.; Li, S.; Hei, Z.; Yang, C.; Wang, H. The interaction between oxidative stress and mast cell activation plays a role in acute lung injuries induced by intestinal ischemia-reperfusion. J. Surg. Res. 2014, 187, 542–552. [Google Scholar] [CrossRef] [PubMed]
- Deitch, E.A.; Xu, D.; Kaise, V.L. Role of the gut in the development of injury- and shock induced SIRS and MODS: The gut-lymph hypothesis, a review. Front. Biosci. 2006, 11, 520–528. [Google Scholar] [CrossRef]
- Huang, C.Y.; Hsiao, J.K.; Lu, Y.Z.; Lee, T.C.; Yu, L.C. Anti-apoptotic PI3K/Akt signaling by sodium/glucose transporter 1 reduces epithelial barrier damage and bacterial translocation in intestinal ischemia. Lab. Investig. 2011, 91, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Puleo, F.; Arvanitakis, M.; Van Gossum, A.; Preiser, J.C. Gut failure in the ICU. Semin. Respir. Crit. Care Med. 2011, 32, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Swank, G.M.; Deitch, E.A. Role of the gut in multiple organ failure: Bacterial translocation and permeability changes. World J. Surg. 1996, 20, 411–417. [Google Scholar] [CrossRef]
- Yasuhara, H. Acute mesenteric ischemia: The challenge of gastroenterology. Surg. Today 2005, 35, 185–195. [Google Scholar] [CrossRef]
- Archontakis-Barakakis, P.; Mavridis, T.; Chlorogiannis, D.D.; Barakakis, G.; Laou, E.; Sessler, D.I.; Gkiokas, G.; Chalkias, A. Intestinal oxygen utilisation and cellular adaptation during intestinal ischaemia-reperfusion injury. Clin. Transl. Med. 2025, 15, e70136. [Google Scholar] [CrossRef]
- Mallick, I.H.; Yang, W.; Winslet, M.C.; Seifalian, A.M. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig. Dis. Sci. 2004, 49, 1359–1377. [Google Scholar] [CrossRef]
- Sastry, P.; Hardman, G.; Page, A.; Parker, R.; Goddard, M.; Large, S.; Jenkins, D.P. Mesenteric ischaemia following cardiac surgery: The influence of intraoperative perfusion parameters. Interact. Cardiovasc. Thorac. Surg. 2014, 19, 419–424. [Google Scholar] [CrossRef]
- Corcos, O.; Nuzzo, A. Gastro-intestinal vascular emergencies. Best. Pract. Res. Clin. Gastroenterol. 2013, 27, 709–725. [Google Scholar] [CrossRef]
- Fishman, J.E.; Sheth, S.U.; Levy, G.; Alli, V.; Lu, Q.; Xu, D.; Qin, Y.; Qin, X.; Deitch, E.A. Intraluminal nonbacterial intestinal components control gut and lung injury after trauma hemorrhagic shock. Ann. Surg. 2014, 260, 1112–1120. [Google Scholar] [CrossRef]
- Brandt, L.J.; Feuerstadt, P.; Longstreth, G.F.; Boley, S.J.; American College of Gastroenterology. ACG clinical guideline: Epidemiology, risk factors, patterns of presentation, diagnosis, and management of colon ischemia (CI). Am. J. Gastroenterol. 2015, 110, 18–44, quiz 45. [Google Scholar] [CrossRef]
- Acosta, S. Epidemiology of mesenteric vascular disease: Clinical implications. Semin. Vasc. Surg. 2010, 23, 4–8. [Google Scholar] [CrossRef]
- Darien, B.J.; Sims, P.A.; Stone, W.C.; Schilly, D.R.; Dubielzig, R.R.; Albrecht, R.M. Ischemia/reperfusion injury of the ascending colon in ponies: A correlative study utilizing microvascular histopathology and corrosion casting. Scanning Microsc. 1993, 7, 1311–1319, discussion 1320. [Google Scholar]
- Granger, D.N.; Korthuis, R.J. Physiologic mechanisms of postischemic tissue injury. Annu. Rev. Physiol. 1995, 57, 311–332. [Google Scholar] [CrossRef] [PubMed]
- Blikslager, A.T. Life in the gut without oxygen: Adaptive mechanisms and inflammatory bowel disease. Gastroenterology 2008, 134, 346–348. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.J.; McArdle, A.H.; Brown, R.; Scott, H.J.; Gurd, F.N. Intestinal mucosal lesion in low-flow states. I. A morphological, hemodynamic, and metabolic reappraisal. Arch. Surg. 1970, 101, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Derikx, J.P.; Matthijsen, R.A.; de Bruine, A.P.; van Bijnen, A.A.; Heineman, E.; van Dam, R.M.; Dejong, C.H.; Buurman, W.A. Rapid reversal of human intestinal ischemia-reperfusion induced damage by shedding of injured enterocytes and reepithelialisation. PLoS ONE 2008, 3, e3428. [Google Scholar] [CrossRef]
- Hernandez, G.; Bruhn, A.; Luengo, C.; Regueira, T.; Kattan, E.; Fuentealba, A.; Florez, J.; Castro, R.; Aquevedo, A.; Pairumani, R.; et al. Effects of dobutamine on systemic, regional and microcirculatory perfusion parameters in septic shock: A randomized, placebo-controlled, double-blind, crossover study. Intensive Care Med. 2013, 39, 1435–1443. [Google Scholar] [CrossRef]
- Sinaasappel, M.; van Iterson, M.; Ince, C. Microvascular oxygen pressure in the pig intestine during haemorrhagic shock and resuscitation. J. Physiol. 1999, 514 Pt 1, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Boros, M.; Takaichi, S.; Hatanaka, K. Ischemic time-dependent microvascular changes and reperfusion injury in the rat small intestine. J. Surg. Res. 1995, 59, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, D.R.; Mirochnitchenko, O.; Ghole, V.S.; Agnese, D.; Shah, P.C.; Reddell, M.; Brolin, R.E.; Inouye, M. Intestinal ischemia and reperfusion injury in transgenic mice overexpressing copper-zinc superoxide dismutase. Am. J. Physiol. 1997, 273, C1130–C1135. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.N.; Benoit, J.N.; Suzuki, M.; Grisham, M.B. Leukocyte adherence to venular endothelium during ischemia-reperfusion. Am. J. Physiol. 1989, 257, G683–G688. [Google Scholar] [CrossRef]
- Granger, D.N.; Hollwarth, M.E.; Parks, D.A. Ischemia-reperfusion injury: Role of oxygen-derived free radicals. Acta Physiol. Scand. Suppl. 1986, 548, 47–63. [Google Scholar]
- Granger, D.N.; Rutili, G.; McCord, J.M. Superoxide radicals in feline intestinal ischemia. Gastroenterology 1981, 81, 22–29. [Google Scholar] [CrossRef]
- Grisham, M.B.; Hernandez, L.A.; Granger, D.N. Xanthine oxidase and neutrophil infiltration in intestinal ischemia. Am. J. Physiol. 1986, 251, G567–G574. [Google Scholar] [CrossRef]
- Grum, C.M.; Gross, T.J.; Mody, C.H.; Sitrin, R.G. Expression of xanthine oxidase activity by murine leukocytes. J. Lab. Clin. Med. 1990, 116, 211–218. [Google Scholar]
- Haglind, E.; Haglund, U.; Lundgren, O.; Stenberg, B. Mucosal lesions of the small intestine after intestinal vascular obstruction in the rat. Acta Chir. Scand. 1985, 151, 147–150. [Google Scholar]
- Hernandez, L.A.; Grisham, M.B.; Granger, D.N. A role for iron in oxidant-mediated ischemic injury to intestinal microvasculature. Am. J. Physiol. 1987, 253, G49–G53. [Google Scholar] [CrossRef]
- Kubes, P.; Hunter, J.; Granger, D.N. Ischemia/reperfusion-induced feline intestinal dysfunction: Importance of granulocyte recruitment. Gastroenterology 1992, 103, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Kubes, P.; Hunter, J.; Granger, D.N. Effects of cyclosporin A and FK506 on ischemia/reperfusion-induced neutrophil infiltration in the cat. Dig. Dis. Sci. 1991, 36, 1469–1472. [Google Scholar] [CrossRef] [PubMed]
- Kurtel, H.; Tso, P.; Granger, D.N. Granulocyte accumulation in postischemic intestine: Role of leukocyte adhesion glycoprotein CD11/CD18. Am. J. Physiol. 1992, 262, G878–G882. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, U.A.; Aberg, J.; Aneman, A.; Lundgren, O. Feline intestinal ischemia and reperfusion: Relation between radical formation and tissue damage. Eur. Surg. Res. 1993, 25, 20–29. [Google Scholar] [CrossRef]
- Nilsson, U.A.; Lundgren, O.; Haglind, E.; Bylund-Fellenius, A.C. Radical production during in vivo intestinal ischemia and reperfusion in the cat. Am. J. Physiol. 1989, 257, G409–G414. [Google Scholar] [CrossRef]
- Nilsson, U.A.; Schoenberg, M.H.; Aneman, A.; Poch, B.; Magadum, S.; Beger, H.G.; Lundgren, O. Free radicals and pathogenesis during ischemia and reperfusion of the cat small intestine. Gastroenterology 1994, 106, 629–636. [Google Scholar] [CrossRef]
- Parks, D.A.; Bulkley, G.B.; Granger, D.N.; Hamilton, S.R.; McCord, J.M. Ischemic injury in the cat small intestine: Role of superoxide radicals. Gastroenterology 1982, 82, 9–15. [Google Scholar] [CrossRef]
- Parks, D.A.; Granger, D.N. Contributions of ischemia and reperfusion to mucosal lesion formation. Am. J. Physiol. 1986, 250, G749–G753. [Google Scholar] [CrossRef]
- Parks, D.A.; Granger, D.N. Ischemia-induced vascular changes: Role of xanthine oxidase and hydroxyl radicals. Am. J. Physiol. 1983, 245, G285–G289. [Google Scholar] [CrossRef]
- Schoenberg, M.H.; Poch, B.; Younes, M.; Schwarz, A.; Baczako, K.; Lundberg, C.; Haglund, U.; Beger, H.G. Involvement of neutrophils in postischaemic damage to the small intestine. Gut 1991, 32, 905–912. [Google Scholar] [CrossRef]
- Suzuki, M.; Grisham, M.B.; Granger, D.N. Leukocyte-endothelial cell adhesive interactions: Role of xanthine oxidase-derived oxidants. J. Leukoc. Biol. 1991, 50, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Chaudry, I.H. Use of ATP following shock and ischemia. Ann. N. Y. Acad. Sci. 1990, 603, 130–140, discussion 140–131. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.Y.; Baek, B.S.; Song, S.H.; Kim, M.S.; Huh, J.I.; Shim, K.H.; Kim, K.W.; Lee, K.H. Xanthine dehydrogenase/xanthine oxidase and oxidative stress. Age 1997, 20, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.N.; McCord, J.M.; Parks, D.A.; Hollwarth, M.E. Xanthine oxidase inhibitors attenuate ischemia-induced vascular permeability changes in the cat intestine. Gastroenterology 1986, 90, 80–84. [Google Scholar] [CrossRef]
- Hayes, J.D.; Flanagan, J.U.; Jowsey, I.R. Glutathione transferases. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 51–88. [Google Scholar] [CrossRef]
- Jaiswal, A.K. Characterization and partial purification of microsomal NAD(P)H:quinone oxidoreductases. Arch. Biochem. Biophys. 2000, 375, 62–68. [Google Scholar] [CrossRef]
- Granger, D.N.; Kvietys, P.R. Reperfusion injury and reactive oxygen species: The evolution of a concept. Redox Biol. 2015, 6, 524–551. [Google Scholar] [CrossRef]
- Tissier, R.; Chenoune, M.; Pons, S.; Zini, R.; Darbera, L.; Lidouren, F.; Ghaleh, B.; Berdeaux, A.; Morin, D. Mild hypothermia reduces per-ischemic reactive oxygen species production and preserves mitochondrial respiratory complexes. Resuscitation 2013, 84, 249–255. [Google Scholar] [CrossRef]
- Garcia, N.; Chavez, E. Mitochondrial DNA fragments released through the permeability transition pore correspond to specific gene size. Life Sci. 2007, 81, 1160–1166. [Google Scholar] [CrossRef]
- Kuwano, Y.; Tominaga, K.; Kawahara, T.; Sasaki, H.; Takeo, K.; Nishida, K.; Masuda, K.; Kawai, T.; Teshima-Kondo, S.; Rokutan, K. Tumor necrosis factor alpha activates transcription of the NADPH oxidase organizer 1 (NOXO1) gene and upregulates superoxide production in colon epithelial cells. Free Radic. Biol. Med. 2008, 45, 1642–1652. [Google Scholar] [CrossRef]
- El Hassani, R.A.; Benfares, N.; Caillou, B.; Talbot, M.; Sabourin, J.C.; Belotte, V.; Morand, S.; Gnidehou, S.; Agnandji, D.; Ohayon, R.; et al. Dual oxidase2 is expressed all along the digestive tract. Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 288, G933–G942. [Google Scholar] [CrossRef]
- Ha, E.M.; Oh, C.T.; Bae, Y.S.; Lee, W.J. A direct role for dual oxidase in Drosophila gut immunity. Science 2005, 310, 847–850. [Google Scholar] [CrossRef]
- Kubes, P.; McCafferty, D.M. Nitric oxide and intestinal inflammation. Am. J. Med. 2000, 109, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, B.C.; Chang, C.J. Chemistry and biology of reactive oxygen species in signaling or stress responses. Nat. Chem. Biol. 2011, 7, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Imlay, J.A. The molecular mechanisms and physiological consequences of oxidative stress: Lessons from a model bacterium. Nat. Rev. Microbiol. 2013, 11, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, J.; Gong, N. Role of the PI3K/Akt signaling pathway in liver ischemia reperfusion injury: A narrative review. Ann. Palliat. Med. 2022, 11, 806–817. [Google Scholar] [CrossRef]
- Wang, F.S.; Wang, C.J.; Chen, Y.J.; Chang, P.R.; Huang, Y.T.; Sun, Y.C.; Huang, H.C.; Yang, Y.J.; Yang, K.D. Ras induction of superoxide activates ERK-dependent angiogenic transcription factor HIF-1alpha and VEGF-A expression in shock wave-stimulated osteoblasts. J. Biol. Chem. 2004, 279, 10331–10337. [Google Scholar] [CrossRef]
- Fiorini, G.; Schofield, C.J. Biochemistry of the hypoxia-inducible factor hydroxylases. Curr. Opin. Chem. Biol. 2024, 79, 102428. [Google Scholar] [CrossRef]
- Singhal, R.; Shah, Y.M. Oxygen battle in the gut: Hypoxia and hypoxia-inducible factors in metabolic and inflammatory responses in the intestine. J. Biol. Chem. 2020, 295, 10493–10505. [Google Scholar] [CrossRef]
- Heir, P.; Ohh, M. Hydroxylation-Dependent Interaction of Substrates to the Von Hippel-Lindau Tumor Suppressor Protein (VHL). Methods Mol. Biol. 2016, 1458, 87–94. [Google Scholar] [CrossRef]
- Ohh, M.; Park, C.W.; Ivan, M.; Hoffman, M.A.; Kim, T.Y.; Huang, L.E.; Pavletich, N.; Chau, V.; Kaelin, W.G. Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel-Lindau protein. Nat. Cell Biol. 2000, 2, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, P.H.; Wiesener, M.S.; Chang, G.W.; Clifford, S.C.; Vaux, E.C.; Cockman, M.E.; Wykoff, C.C.; Pugh, C.W.; Maher, E.R.; Ratcliffe, P.J. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 1999, 399, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Jewell, U.R.; Kvietikova, I.; Scheid, A.; Bauer, C.; Wenger, R.H.; Gassmann, M. Induction of HIF-1alpha in response to hypoxia is instantaneous. FASEB J. 2001, 15, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Watts, E.R.; Walmsley, S.R. Inflammation and Hypoxia: HIF and PHD Isoform Selectivity. Trends Mol. Med. 2019, 25, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Lando, D.; Peet, D.J.; Whelan, D.A.; Gorman, J.J.; Whitelaw, M.L. Asparagine hydroxylation of the HIF transactivation domain a hypoxic switch. Science 2002, 295, 858–861. [Google Scholar] [CrossRef]
- Wenger, R.H.; Stiehl, D.P.; Camenisch, G. Integration of oxygen signaling at the consensus HRE. Sci. STKE Signal Transduct. Knowl. Environ. 2005, 2005, re12. [Google Scholar] [CrossRef]
- Smythies, J.A.; Sun, M.; Masson, N.; Salama, R.; Simpson, P.D.; Murray, E.; Neumann, V.; Cockman, M.E.; Choudhry, H.; Ratcliffe, P.J.; et al. Inherent DNA-binding specificities of the HIF-1alpha and HIF-2alpha transcription factors in chromatin. EMBO Rep. 2019, 20, e46401. [Google Scholar] [CrossRef]
- Kaelin, W.G., Jr.; Ratcliffe, P.J. Oxygen sensing by metazoans: The central role of the HIF hydroxylase pathway. Mol. Cell 2008, 30, 393–402. [Google Scholar] [CrossRef]
- Taylor, C.T.; Scholz, C.C. The effect of HIF on metabolism and immunity. Nat. Rev. Nephrol. 2022, 18, 573–587. [Google Scholar] [CrossRef]
- Ramakrishnan, S.K.; Shah, Y.M. Role of Intestinal HIF-2alpha in Health and Disease. Annu. Rev. Physiol. 2016, 78, 301–325. [Google Scholar] [CrossRef]
- Sun, L.; Li, T.; Tang, H.; Yu, K.; Ma, Y.; Yu, M.; Qiu, Y.; Xu, P.; Xiao, W.; Yang, H. Intestinal Epithelial Cells-Derived Hypoxia-Inducible Factor-1alpha Is Essential for the Homeostasis of Intestinal Intraepithelial Lymphocytes. Front. Immunol. 2019, 10, 806. [Google Scholar] [CrossRef]
- Volkova, Y.L.; Pickel, C.; Jucht, A.E.; Wenger, R.H.; Scholz, C.C. The Asparagine Hydroxylase FIH: A Unique Oxygen Sensor. Antioxid. Redox Signal. 2022, 37, 913–935. [Google Scholar] [CrossRef] [PubMed]
- Feinman, R.; Deitch, E.A.; Watkins, A.C.; Abungu, B.; Colorado, I.; Kannan, K.B.; Sheth, S.U.; Caputo, F.J.; Lu, Q.; Ramanathan, M.; et al. HIF-1 mediates pathogenic inflammatory responses to intestinal ischemia-reperfusion injury. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G833–G843. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Reyes, I.; Diebold, L.P.; Kong, H.; Schieber, M.; Huang, H.; Hensley, C.T.; Mehta, M.M.; Wang, T.; Santos, J.H.; Woychik, R.; et al. TCA Cycle and Mitochondrial Membrane Potential Are Necessary for Diverse Biological Functions. Mol. Cell 2016, 61, 199–209. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Berean, K.J.; Burgell, R.E.; Muir, J.G.; Gibson, P.R. Intestinal gases: Influence on gut disorders and the role of dietary manipulations. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 733–747. [Google Scholar] [CrossRef]
- Nizet, V.; Johnson, R.S. Interdependence of hypoxic and innate immune responses. Nat. Rev. Immunol. 2009, 9, 609–617. [Google Scholar] [CrossRef]
- Hellwig-Burgel, T.; Rutkowski, K.; Metzen, E.; Fandrey, J.; Jelkmann, W. Interleukin-1beta and tumor necrosis factor-alpha stimulate DNA binding of hypoxia-inducible factor-1. Blood 1999, 94, 1561–1567. [Google Scholar] [CrossRef]
- Scharte, M.; Han, X.; Bertges, D.J.; Fink, M.P.; Delude, R.L. Cytokines induce HIF-1 DNA binding and the expression of HIF-1-dependent genes in cultured rat enterocytes. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G373–G384. [Google Scholar] [CrossRef]
- Koury, J.; Deitch, E.A.; Homma, H.; Abungu, B.; Gangurde, P.; Condon, M.R.; Lu, Q.; Xu, D.Z.; Feinman, R. Persistent HIF-1alpha activation in gut ischemia/reperfusion injury: Potential role of bacteria and lipopolysaccharide. Shock 2004, 22, 270–277. [Google Scholar] [CrossRef]
- Huang, L.E.; Gu, J.; Schau, M.; Bunn, H.F. Regulation of hypoxia-inducible factor 1alpha is mediated by an O2-dependent degradation domain via the ubiquitin-proteasome pathway. Proc. Natl. Acad. Sci. USA 1998, 95, 7987–7992. [Google Scholar] [CrossRef]
- Scharte, M.; Han, X.; Uchiyama, T.; Tawadrous, Z.; Delude, R.L.; Fink, M.P. LPS increases hepatic HIF-1alpha protein and expression of the HIF-1-dependent gene aldolase A in rats. J. Surg. Res. 2006, 135, 262–267. [Google Scholar] [CrossRef]
- Niu, G.; Briggs, J.; Deng, J.; Ma, Y.; Lee, H.; Kortylewski, M.; Kujawski, M.; Kay, H.; Cress, W.D.; Jove, R.; et al. Signal transducer and activator of transcription 3 is required for hypoxia-inducible factor-1alpha RNA expression in both tumor cells and tumor-associated myeloid cells. Mol. Cancer Res. MCR 2008, 6, 1099–1105. [Google Scholar] [CrossRef]
- D’Hulst, G.; Soro-Arnaiz, I.; Masschelein, E.; Veys, K.; Fitzgerald, G.; Smeuninx, B.; Kim, S.; Deldicque, L.; Blaauw, B.; Carmeliet, P.; et al. PHD1 controls muscle mTORC1 in a hydroxylation-independent manner by stabilizing leucyl tRNA synthetase. Nat. Commun. 2020, 11, 174. [Google Scholar] [CrossRef]
- Masoud, G.N.; Li, W. HIF-1alpha pathway: Role, regulation and intervention for cancer therapy. Acta Pharm. Sin. B 2015, 5, 378–389. [Google Scholar] [CrossRef] [PubMed]
- McGettrick, A.F.; O’Neill, L.A.J. The Role of HIF in Immunity and Inflammation. Cell Metab. 2020, 32, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Laukoetter, M.G.; Bruewer, M.; Nusrat, A. Regulation of the intestinal epithelial barrier by the apical junctional complex. Curr. Opin. Gastroenterol. 2006, 22, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, B.J.; Kao, D.J.; Kitzenberg, D.A.; Dobrinskikh, E.; Schwisow, K.D.; Masterson, J.C.; Kendrick, A.A.; Kelly, C.J.; Bayless, A.J.; Kominsky, D.J.; et al. HIF-dependent regulation of claudin-1 is central to intestinal epithelial tight junction integrity. Mol. Biol. Cell 2015, 26, 2252–2262. [Google Scholar] [CrossRef]
- Birchenough, G.M.; Johansson, M.E.; Gustafsson, J.K.; Bergstrom, J.H.; Hansson, G.C. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 2015, 8, 712–719. [Google Scholar] [CrossRef]
- Bin Hafeez, A.; Jiang, X.; Bergen, P.J.; Zhu, Y. Antimicrobial Peptides: An Update on Classifications and Databases. Int. J. Mol. Sci. 2021, 22, 11691. [Google Scholar] [CrossRef]
- Louis, N.A.; Hamilton, K.E.; Canny, G.; Shekels, L.L.; Ho, S.B.; Colgan, S.P. Selective induction of mucin-3 by hypoxia in intestinal epithelia. J. Cell. Biochem. 2006, 99, 1616–1627. [Google Scholar] [CrossRef]
- Krzywinska, E.; Stockmann, C. Hypoxia, Metabolism and Immune Cell Function. Biomedicines 2018, 6, 56. [Google Scholar] [CrossRef] [PubMed]
- Kistler, E.B.; Alsaigh, T.; Chang, M.; Schmid-Schonbein, G.W. Impaired small-bowel barrier integrity in the presence of lumenal pancreatic digestive enzymes leads to circulatory shock. Shock 2012, 38, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.; Alsaigh, T.; Kistler, E.B.; Schmid-Schonbein, G.W. Breakdown of mucin as barrier to digestive enzymes in the ischemic rat small intestine. PLoS ONE 2012, 7, e40087. [Google Scholar] [CrossRef] [PubMed]
- Alsaigh, T.; Chang, M.; Richter, M.; Mazor, R.; Kistler, E.B. In vivo analysis of intestinal permeability following hemorrhagic shock. World J. Crit. Care Med. 2015, 4, 287–295. [Google Scholar] [CrossRef]
- Blikslager, A.T.; Roberts, M.C.; Rhoads, J.M.; Argenzio, R.A. Is reperfusion injury an important cause of mucosal damage after porcine intestinal ischemia? Surgery 1997, 121, 526–534. [Google Scholar] [CrossRef]
- Park, P.O.; Haglund, U.; Bulkley, G.B.; Falt, K. The sequence of development of intestinal tissue injury after strangulation ischemia and reperfusion. Surgery 1990, 107, 574–580. [Google Scholar]
- DeLano, F.A.; Hoyt, D.B.; Schmid-Schonbein, G.W. Pancreatic digestive enzyme blockade in the intestine increases survival after experimental shock. Sci. Transl. Med. 2013, 5, 169ra111. [Google Scholar] [CrossRef]
- Ma, H.; Tao, W.; Zhu, S. T lymphocytes in the intestinal mucosa: Defense and tolerance. Cell Mol. Immunol. 2019, 16, 216–224. [Google Scholar] [CrossRef]
- Michalsky, M.P.; Deitch, E.A.; Ding, J.; Lu, Q.; Huang, Q. Interleukin-6 and tumor necrosis factor production in an enterocyte cell model (Caco-2) during exposure to Escherichia coli. Shock 1997, 7, 139–146. [Google Scholar] [CrossRef]
- Yeh, K.Y.; Yeh, M.; Glass, J.; Granger, D.N. Rapid activation of NF-kappaB and AP-1 and target gene expression in postischemic rat intestine. Gastroenterology 2000, 118, 525–534. [Google Scholar] [CrossRef]
- Hierholzer, C.; Harbrecht, B.G.; Billiar, T.R.; Tweardy, D.J. Hypoxia-inducible factor-1 activation and cyclo-oxygenase-2 induction are early reperfusion-independent inflammatory events in hemorrhagic shock. Arch. Orthop. Trauma. Surg. 2001, 121, 219–222. [Google Scholar] [CrossRef]
- Liaudet, L.; Szabo, A.; Soriano, F.G.; Zingarelli, B.; Szabo, C.; Salzman, A.L. Poly (ADP-ribose) synthetase mediates intestinal mucosal barrier dysfunction after mesenteric ischemia. Shock 2000, 14, 134–141. [Google Scholar] [CrossRef]
- Grootjans, J.; Lenaerts, K.; Derikx, J.P.; Matthijsen, R.A.; de Bruine, A.P.; van Bijnen, A.A.; van Dam, R.M.; Dejong, C.H.; Buurman, W.A. Human intestinal ischemia-reperfusion-induced inflammation characterized: Experiences from a new translational model. Am. J. Pathol. 2010, 176, 2283–2291. [Google Scholar] [CrossRef]
- Louis, K.; Netea, M.G.; Carrer, D.P.; Kotsaki, A.; Mylona, V.; Pistiki, A.; Savva, A.; Roditis, K.; Alexis, A.; Van der Meer, J.W.; et al. Bacterial translocation in an experimental model of multiple organ dysfunctions. J. Surg. Res. 2013, 183, 686–694. [Google Scholar] [CrossRef]
- Podolsky, D.K. Mucosal immunity and inflammation. V. Innate mechanisms of mucosal defense and repair: The best offense is a good defense. Am. J. Physiol. 1999, 277, G495–G499. [Google Scholar] [CrossRef] [PubMed]
- Riley, J.S.; Tait, S.W. Mitochondrial DNA in inflammation and immunity. EMBO Rep. 2020, 21, e49799. [Google Scholar] [CrossRef] [PubMed]
- Hemmi, H.; Takeuchi, O.; Kawai, T.; Kaisho, T.; Sato, S.; Sanjo, H.; Matsumoto, M.; Hoshino, K.; Wagner, H.; Takeda, K.; et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000, 408, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Rose, W.A., 2nd; Sakamoto, K.; Leifer, C.A. TLR9 is important for protection against intestinal damage and for intestinal repair. Sci. Rep. 2012, 2, 574. [Google Scholar] [CrossRef]
- Liu, C.Y.; Cham, C.M.; Chang, E.B. Epithelial wound healing in inflammatory bowel diseases: The next therapeutic frontier. Transl. Res. 2021, 236, 35–51. [Google Scholar] [CrossRef]
- Prochnicki, T.; Latz, E. Inflammasomes on the Crossroads of Innate Immune Recognition and Metabolic Control. Cell Metab. 2017, 26, 71–93. [Google Scholar] [CrossRef]
- Zhen, Y.; Zhang, H. NLRP3 Inflammasome and Inflammatory Bowel Disease. Front. Immunol. 2019, 10, 276. [Google Scholar] [CrossRef] [PubMed]
- Doedens, A.L.; Phan, A.T.; Stradner, M.H.; Fujimoto, J.K.; Nguyen, J.V.; Yang, E.; Johnson, R.S.; Goldrath, A.W. Hypoxia-inducible factors enhance the effector responses of CD8(+) T cells to persistent antigen. Nat. Immunol. 2013, 14, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.H.; Barbi, J.; Pan, F. Hypoxia-inducible factors in T lymphocyte differentiation and function. A Review in the Theme: Cellular Responses to Hypoxia. Am. J. Physiol. Cell Physiol. 2015, 309, C580–C589. [Google Scholar] [CrossRef]
- Palazon, A.; Tyrakis, P.A.; Macias, D.; Velica, P.; Rundqvist, H.; Fitzpatrick, S.; Vojnovic, N.; Phan, A.T.; Loman, N.; Hedenfalk, I.; et al. An HIF-1alpha/VEGF-A Axis in Cytotoxic T Cells Regulates Tumor Progression. Cancer Cell 2017, 32, 669–683.e665. [Google Scholar] [CrossRef]
- Mecklenburgh, K.I.; Walmsley, S.R.; Cowburn, A.S.; Wiesener, M.; Reed, B.J.; Upton, P.D.; Deighton, J.; Greening, A.P.; Chilvers, E.R. Involvement of a ferroprotein sensor in hypoxia-mediated inhibition of neutrophil apoptosis. Blood 2002, 100, 3008–3016. [Google Scholar] [CrossRef]
- Kong, T.; Eltzschig, H.K.; Karhausen, J.; Colgan, S.P.; Shelley, C.S. Leukocyte adhesion during hypoxia is mediated by HIF-1-dependent induction of beta2 integrin gene expression. Proc. Natl. Acad. Sci. USA 2004, 101, 10440–10445. [Google Scholar] [CrossRef]
- Suzuki, M.; Inauen, W.; Kvietys, P.R.; Grisham, M.B.; Meininger, C.; Schelling, M.E.; Granger, H.J.; Granger, D.N. Superoxide mediates reperfusion-induced leukocyte-endothelial cell interactions. Am. J. Physiol. 1989, 257, H1740–H1745. [Google Scholar] [CrossRef]
- Walmsley, S.R.; Chilvers, E.R.; Thompson, A.A.; Vaughan, K.; Marriott, H.M.; Parker, L.C.; Shaw, G.; Parmar, S.; Schneider, M.; Sabroe, I.; et al. Prolyl hydroxylase 3 (PHD3) is essential for hypoxic regulation of neutrophilic inflammation in humans and mice. J. Clin. Investig. 2011, 121, 1053–1063. [Google Scholar] [CrossRef]
- Linfert, D.; Chowdhry, T.; Rabb, H. Lymphocytes and ischemia-reperfusion injury. Transplant. Rev. 2009, 23, 1–10. [Google Scholar] [CrossRef]
- Zimmerman, B.J.; Granger, D.N. Reperfusion-induced leukocyte infiltration: Role of elastase. Am. J. Physiol. 1990, 259, H390–H394. [Google Scholar] [CrossRef]
- Hickey, M.J.; Kubes, P. Intravascular immunity: The host-pathogen encounter in blood vessels. Nat. Rev. Immunol. 2009, 9, 364–375. [Google Scholar] [CrossRef]
- Fluck, K.; Breves, G.; Fandrey, J.; Winning, S. Hypoxia-inducible factor 1 in dendritic cells is crucial for the activation of protective regulatory T cells in murine colitis. Mucosal Immunol. 2016, 9, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Kohler, T.; Reizis, B.; Johnson, R.S.; Weighardt, H.; Forster, I. Influence of hypoxia-inducible factor 1alpha on dendritic cell differentiation and migration. Eur. J. Immunol. 2012, 42, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Kuhlicke, J.; Frick, J.S.; Morote-Garcia, J.C.; Rosenberger, P.; Eltzschig, H.K. Hypoxia inducible factor (HIF)-1 coordinates induction of Toll-like receptors TLR2 and TLR6 during hypoxia. PLoS ONE 2007, 2, e1364. [Google Scholar] [CrossRef] [PubMed]
- Wobben, R.; Husecken, Y.; Lodewick, C.; Gibbert, K.; Fandrey, J.; Winning, S. Role of hypoxia inducible factor-1alpha for interferon synthesis in mouse dendritic cells. Biol. Chem. 2013, 394, 495–505. [Google Scholar] [CrossRef]
- Mills, E.L.; O’Neill, L.A. Reprogramming mitochondrial metabolism in macrophages as an anti-inflammatory signal. Eur. J. Immunol. 2016, 46, 13–21. [Google Scholar] [CrossRef]
- Barrero, C.A.; Datta, P.K.; Sen, S.; Deshmane, S.; Amini, S.; Khalili, K.; Merali, S. HIV-1 Vpr modulates macrophage metabolic pathways: A SILAC-based quantitative analysis. PLoS ONE 2013, 8, e68376. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, Z.; Zhang, W.; Meng, X.; Zhu, Y.; Han, P.; Zhou, X.; Hu, Y.; Wang, R. Nuclear translocation of HIF-1alpha induced by influenza A (H1N1) infection is critical to the production of proinflammatory cytokines. Emerg. Microbes Infect. 2017, 6, e39. [Google Scholar] [CrossRef]
- Staples, K.J.; Sotoodehnejadnematalahi, F.; Pearson, H.; Frankenberger, M.; Francescut, L.; Ziegler-Heitbrock, L.; Burke, B. Monocyte-derived macrophages matured under prolonged hypoxia transcriptionally up-regulate HIF-1alpha mRNA. Immunobiology 2011, 216, 832–839. [Google Scholar] [CrossRef]
- Tannahill, G.M.; Curtis, A.M.; Adamik, J.; Palsson-McDermott, E.M.; McGettrick, A.F.; Goel, G.; Frezza, C.; Bernard, N.J.; Kelly, B.; Foley, N.H.; et al. Succinate is an inflammatory signal that induces IL-1beta through HIF-1alpha. Nature 2013, 496, 238–242. [Google Scholar] [CrossRef]
- Filippi, M.D. Neutrophil transendothelial migration: Updates and new perspectives. Blood 2019, 133, 2149–2158. [Google Scholar] [CrossRef]
- Gayle, J.; Jones, S.L.; Argenzio, R.A.; Blikslager, A.T. Neutrophils increase paracellular permeability of restituted ischemic-injured porcine ileum. Surgery 2002, 132, 461–470. [Google Scholar] [CrossRef]
- Campbell, E.L.; Bruyninckx, W.J.; Kelly, C.J.; Glover, L.E.; McNamee, E.N.; Bowers, B.E.; Bayless, A.J.; Scully, M.; Saeedi, B.J.; Golden-Mason, L.; et al. Transmigrating neutrophils shape the mucosal microenvironment through localized oxygen depletion to influence resolution of inflammation. Immunity 2014, 40, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Harrois, A.; Baudry, N.; Huet, O.; Kato, H.; Lohez, M.; Ziol, M.; Duranteau, J.; Vicaut, E. Synergistic deleterious effect of hypoxemia and hypovolemia on microcirculation in intestinal villi*. Crit. Care Med. 2013, 41, e376–e384. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, B.G.; Kloner, R.A. Coronary no reflow. J. Mol. Cell Cardiol. 2012, 52, 873–882. [Google Scholar] [CrossRef] [PubMed]
- del Zoppo, G.J.; Schmid-Schonbein, G.W.; Mori, E.; Copeland, B.R.; Chang, C.M. Polymorphonuclear leukocytes occlude capillaries following middle cerebral artery occlusion and reperfusion in baboons. Stroke 1991, 22, 1276–1283. [Google Scholar] [CrossRef]
- Hernandez, L.A.; Grisham, M.B.; Twohig, B.; Arfors, K.E.; Harlan, J.M.; Granger, D.N. Role of neutrophils in ischemia-reperfusion-induced microvascular injury. Am. J. Physiol. 1987, 253, H699–H703. [Google Scholar] [CrossRef]
- Biancheri, P.; Di Sabatino, A.; Corazza, G.R.; MacDonald, T.T. Proteases and the gut barrier. Cell Tissue Res. 2013, 351, 269–280. [Google Scholar] [CrossRef]
- Maronek, M.; Marafini, I.; Gardlik, R.; Link, R.; Troncone, E.; Monteleone, G. Metalloproteinases in Inflammatory Bowel Diseases. J. Inflamm. Res. 2021, 14, 1029–1041. [Google Scholar] [CrossRef]
- Cui, N.; Hu, M.; Khalil, R.A. Biochemical and Biological Attributes of Matrix Metalloproteinases. Prog. Mol. Biol. Transl. Sci. 2017, 147, 1–73. [Google Scholar] [CrossRef]
- Dejonckheere, E.; Vandenbroucke, R.E.; Libert, C. Matrix metalloproteinases as drug targets in ischemia/reperfusion injury. Drug Discov. Today 2011, 16, 762–778. [Google Scholar] [CrossRef]
- Ito, K.; Kitajima, Y.; Kai, K.; Matsufuji, S.; Yamada, K.; Egawa, N.; Kitagawa, H.; Okuyama, K.; Tanaka, T.; Noshiro, H. Matrix metalloproteinase-1 expression is regulated by HIF-1-dependent and epigenetic mechanisms and serves a tumor-suppressive role in gastric cancer progression. Int. J. Oncol. 2021, 59, 102. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Zhou, Y.; Wang, L.; Zhang, J.; Wu, H.; Xiong, J.; Zhang, J.; Tian, Y.; Wang, C.; Wu, H. Transcriptional upregulation of MT2-MMP in response to hypoxia is promoted by HIF-1alpha in cancer cells. Mol. Carcinog. 2011, 50, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Yang, W.H.; Chang, S.Y.; Tai, S.K.; Tzeng, C.H.; Kao, J.Y.; Wu, K.J.; Yang, M.H. Regulation of membrane-type 4 matrix metalloproteinase by SLUG contributes to hypoxia-mediated metastasis. Neoplasia 2009, 11, 1371–1382. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, S.; Meng, X.P.; Ramasamy, S.; Harrison, D.G.; Galis, Z.S. Reactive oxygen species produced by macrophage-derived foam cells regulate the activity of vascular matrix metalloproteinases in vitro. Implications for atherosclerotic plaque stability. J. Clin. Investig. 1996, 98, 2572–2579. [Google Scholar] [CrossRef]
- Shah, S.V.; Baricos, W.H.; Basci, A. Degradation of human glomerular basement membrane by stimulated neutrophils. Activation of a metalloproteinase(s) by reactive oxygen metabolites. J. Clin. Investig. 1987, 79, 25–31. [Google Scholar] [CrossRef]
- Weiss, S.J.; Peppin, G.; Ortiz, X.; Ragsdale, C.; Test, S.T. Oxidative autoactivation of latent collagenase by human neutrophils. Science 1985, 227, 747–749. [Google Scholar] [CrossRef]
- Haglund, U.; Bulkley, G.B.; Granger, D.N. On the pathophysiology of intestinal ischemic injury. Clinical review. Acta Chir. Scand. 1987, 153, 321–324. [Google Scholar]
- Lima, A.; Bakker, J. Noninvasive monitoring of peripheral perfusion. Intensive Care Med. 2005, 31, 1316–1326. [Google Scholar] [CrossRef]
- Sowa, M.G.; Kohlenberg, E.; Payette, J.R.; Leonardi, L.; Levasseur, M.A.; Riley, C.B. Detecting Intestinal Ischemia Using near Infrared Spectroscopy. J. Near Infrared Spectrosc. 2006, 14, 1–7. [Google Scholar] [CrossRef]
- Fiddian-Green, R.G.; Baker, S. Predictive value of the stomach wall pH for complications after cardiac operations: Comparison with other monitoring. Crit. Care Med. 1987, 15, 153–156. [Google Scholar] [CrossRef]
| Parameter | Typical Indicator in Ischemia |
|---|---|
| Lactate | >2 mmol/L (severe cases often >4 mmol/L) |
| pH/Base deficit | pH < 7.35, base deficit > 4 mmol/L |
| Intestinal fatty acid-binding protein (I-FABP) | Elevated compared to baseline—no single cutoff; used as an early marker of mucosal damage |
| D-lactate | Elevated; typical serum levels rise above 0.25 mmol/L, but ranges vary |
| Cytokines (e.g., TNF-α, IL-6) | Elevated; used in research, no diagnostic cutoff |
| Parameter | Approximate Irreversible Range |
|---|---|
| SMA occlusion time | ≥90–120 min (animal models) |
| Tissue ATP | ≤10–30% of baseline (~0.3–0.5 µmol/g) |
| Mucosal PO2 | ~0–5 mmHg for >60 min |
| Serum lactate (clinical) | >4–6 mmol/L → high risk of transmural necrosis |
| Histopathology | Full-thickness (transmural) necrosis, loss of mucosal architecture |
| Parameter | Approximate Irreversible Range |
|---|---|
| Systemic SpO2 | Not useful for local ischemia |
| Hb concentration | Not useful by itself |
| Lactate/base deficit | Good general markers of anaerobic metabolism |
| StO2 (NIRS) | Most practical noninvasive method to estimate local tissue oxygenation |
| Mucosal PO2/PCO2 | Excellent but more invasive, used in research/ICU |
| Imaging | CT angiography |
| Intraoperative NIRS for Gut StO2 |
|---|
| Purpose |
| To assess local tissue oxygen saturation (StO2) in the intestinal wall during surgery for suspected ischemia (e.g., bowel resection, mesenteric thrombectomy). |
| Equipment |
| Near-Infrared Spectroscopy (NIRS) monitor, e.g., INVOS™, NIRO™, or equivalent. |
| Sterile adhesive sensor/probe for intraoperative use. |
| Optional: Doppler probe to confirm perfusion. |
| Protocol Steps |
| 1. Patient prep |
| Standard anesthesia, maintain normoxia (SpO2 > 95%), normotension. |
| Baseline arterial blood gas, lactate, base deficit. |
| 2. Probe placement |
| Sterilize probe surface if reusable. |
| Gently place the NIRS probe directly onto the serosal surface of the bowel loop in question. |
| Avoid compressing the bowel wall to prevent false readings. |
| 3. Measurement |
| Wait 30–60 s for signal stabilization. |
| Record baseline StO2. |
| Take readings every 5–10 min, or continuously if device allows. |
| Repeat after interventions: e.g., SMA release, anastomosis. |
| 4. Interpretation |
| Normal StO2 for gut wall: ~70–80% |
| StO2 < 60–70% → suspicious for inadequate perfusion. |
| Persistent StO2 < 50–60% despite revascularization → consider nonviable segment. |
| 5. Documentation |
| Note probe site, time, and conditions. |
| Correlate with clinical judgement (color, peristalsis, Doppler flow) |
| Mucosal Tonometry for Gastric or Intestinal Mucosal Oxygenation |
|---|
| Purpose |
| To indirectly assess mucosal hypoxia by measuring local mucosal PCO2 or mucosal pH (pHi) → increased mucosal PCO2 means hypoperfusion. |
| Equipment |
| Gastric or intestinal tonometer catheter (e.g., TRIP tonometer or balloon tonometry). |
| Standard blood gas analyzer. |
| Protocol Steps |
| 1. Patient prep |
| Insert tonometry catheter into stomach or directly into bowel lumen if intraoperative. |
| Fill balloon with air or saline according to device instructions. |
| 2. Equilibration |
| 3. Sampling |
| Aspirate sample gas from balloon. |
| Measure PCO2 using standard ABG machine. |
| Simultaneously measure arterial blood PCO2. |
| 4. Calculations |
| Calculate mucosal–arterial CO2 gap (ΔPCO2): ΔPCO2 = PCO2(mucosa)—PCO2(arterial) |
| Calculate gastric pHi: pHi = 6.1 + log([HCO3−]/(PCO2 mucosal × 0.03)) |
| 5. Interpretation |
| Normal mucosal–arterial CO2 gap: <15 mmHg. |
| ΔPCO2 > 20 mmHg or pHi < 7.3 → significant mucosal ischemia. |
| Persistent elevation → high risk of irreversible damage. |
| Parameter | Controlled (Smooth) Reperfusion | Risk of Reperfusion Injury |
|---|---|---|
| StO2 (NIRS) | Gradual rise to 60–80% over 5–10 min | Sudden spike > 20–30% in 1–2 min |
| Mucosal–arterial CO2 gap | Normalizes to <15 mmHg | Remains > 20 mmHg |
| Plasma MDA | Stable, low | Peaks > 2–3 µmol/L |
| XOD activity | Mild increase | High levels |
| MPO | Mild increase | High levels |
| Serum lactate | Steady fall | Increased Paradoxical rebound |
| Doppler flow Microvascular flow | Laminar, adequate | Turbulent flow Microvascular no-reflow pattern |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Archontakis-Barakakis, P.; Mavridis, T.; Chalkias, A. Oxygen-Mediated Molecular Mechanisms Involved in Intestinal Ischemia and Reperfusion Injury. Int. J. Mol. Sci. 2025, 26, 8398. https://doi.org/10.3390/ijms26178398
Archontakis-Barakakis P, Mavridis T, Chalkias A. Oxygen-Mediated Molecular Mechanisms Involved in Intestinal Ischemia and Reperfusion Injury. International Journal of Molecular Sciences. 2025; 26(17):8398. https://doi.org/10.3390/ijms26178398
Chicago/Turabian StyleArchontakis-Barakakis, Paraschos, Theodoros Mavridis, and Athanasios Chalkias. 2025. "Oxygen-Mediated Molecular Mechanisms Involved in Intestinal Ischemia and Reperfusion Injury" International Journal of Molecular Sciences 26, no. 17: 8398. https://doi.org/10.3390/ijms26178398
APA StyleArchontakis-Barakakis, P., Mavridis, T., & Chalkias, A. (2025). Oxygen-Mediated Molecular Mechanisms Involved in Intestinal Ischemia and Reperfusion Injury. International Journal of Molecular Sciences, 26(17), 8398. https://doi.org/10.3390/ijms26178398









