Fibrotic Disease of the Skin and Lung: Shared Pathways, Environmental Drivers, and Therapeutic Opportunities in a Changing Climate
Abstract
1. Introduction
2. Pathophysiology and Cellular Mechanisms
2.1. Shared Fibrotic Pathways in Skin and Lung Fibrosis
2.2. Fibroinflammatory Remodeling in Lung and Skin Fibrosis
2.3. Clinical Characteristics of Skin and Lung Fibrosis: Focus on Systemic Sclerosis
3. Epigenetic Regulation in Skin and Lung Fibrosis
3.1. Epigenetics of Skin Fibrosis
3.2. Epigenetics of Lung Fibrosis
4. MicroRNAs in Skin and Lung Fibrosis: Shared Mechanisms and Molecular Pathways
5. The Exposome Driving Skin and Lung Fibrosis
5.1. Climate Change and Pollution
5.2. UV Radiation, Wildfires, and Burns
5.2.1. UV Radiation-Induced Fibrosis
5.2.2. Wildfires and Thermal Injury
5.3. Occupational Exposome
6. Therapeutic Opportunities and Future Directions
6.1. Evidence from SSc with Nintedanib
6.2. From the Lung to the Skin: Expanding Antifibrotic Therapies
6.3. Fibroinflammatory Skin Diseases: A Paradigm Shift
6.4. RNA and Epigenetic Therapeutics
7. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wynn, T.A. Fibrotic Disease and the TH1/TH2 Paradigm. Nat. Rev. Immunol. 2004, 4, 583–594. [Google Scholar] [CrossRef]
- Thannickal, V.J.; Zhou, Y.; Gaggar, A.; Duncan, S.R. Fibrosis: Ultimate and Proximate Causes. J. Clin. Investig. 2014, 124, 4673–4677. [Google Scholar] [CrossRef]
- Ku, J.C.; Raiten, J.; Li, Y. Understanding Fibrosis: Mechanisms, Clinical Implications, Current Therapies, and Prospects for Future Interventions. Biomed. Eng. Adv. 2024, 7, 100118. [Google Scholar] [CrossRef]
- Rieder, F.; Nagy, L.E.; Maher, T.M.; Distler, J.H.W.; Kramann, R.; Hinz, B.; Prunotto, M. Publisher Correction: Fibrosis: Cross-Organ Biology and Pathways to Development of Innovative Drugs. Nat. Rev. Drug Discov. 2025, 24, 399. [Google Scholar] [CrossRef]
- Jiang, M.; Bu, W.; Wang, X.; Ruan, J.; Shi, W.; Yu, S.; Huang, L.; Xue, P.; Tang, J.; Zhao, X.; et al. Pulmonary Fibrosis: From Mechanisms to Therapies. J. Transl. Med. 2025, 23, 515. [Google Scholar] [CrossRef] [PubMed]
- Henderson, N.C.; Rieder, F.; Wynn, T.A. Fibrosis: From Mechanisms to Medicines. Nature 2020, 587, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, S.A.; Mendoza, F.A.; Piera-Velazquez, S. A Review of Recent Studies on the Pathogenesis of Systemic Sclerosis: Focus on Fibrosis Pathways. Front. Immunol. 2025, 16, 1551911. [Google Scholar] [CrossRef]
- Nanthakumar, C.B.; Hatley, R.J.D.; Lemma, S.; Gauldie, J.; Marshall, R.P.; Macdonald, S.J.F. Dissecting Fibrosis: Therapeutic Insights from the Small-Molecule Toolbox. Nat. Rev. Drug Discov. 2015, 14, 693–720. [Google Scholar] [CrossRef]
- Distler, J.H.W.; Györfi, A.-H.; Ramanujam, M.; Whitfield, M.L.; Königshoff, M.; Lafyatis, R. Shared and Distinct Mechanisms of Fibrosis. Nat. Rev. Rheumatol. 2019, 15, 705–730. [Google Scholar] [CrossRef]
- Herriges, M.; Morrisey, E.E. Lung Development: Orchestrating the Generation and Regeneration of a Complex Organ. Development 2014, 141, 502–513. [Google Scholar] [CrossRef]
- Hu, M.S.; Borrelli, M.R.; Hong, W.X.; Malhotra, S.; Cheung, A.T.M.; Ransom, R.C.; Rennert, R.C.; Morrison, S.D.; Lorenz, H.P.; Longaker, M.T. Embryonic Skin Development and Repair. Organogenesis 2018, 14, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Truchetet, M.E.; Brembilla, N.C.; Chizzolini, C. Current Concepts on the Pathogenesis of Systemic Sclerosis. Clin. Rev. Allergy Immunol. 2023, 64, 262–283. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.; Tonelli, R.; Murray, M.; Samarelli, A.V.; Spagnolo, P. Environmental Causes of Idiopathic Pulmonary Fibrosis. Int. J. Mol. Sci. 2023, 24, 16481. [Google Scholar] [CrossRef]
- Miller, G.W.; Jones, D.P. The Nature of Nurture: Refining the Definition of the Exposome. Toxicol. Sci. 2014, 137, 1–2. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Wei, J.; Varga, J. Understanding Fibrosis in Systemic Sclerosis: Shifting Paradigms, Emerging Opportunities. Nat. Rev. Rheumatol. 2011, 8, 42–54. [Google Scholar] [CrossRef]
- Wynn, T.A. Cellular and Molecular Mechanisms of Fibrosis. J. Pathol. 2008, 214, 199–210. [Google Scholar] [CrossRef]
- Martinez, F.J.; Collard, H.R.; Pardo, A.; Raghu, G.; Richeldi, L.; Selman, M.; Swigris, J.J.; Taniguchi, H.; Wells, A.U. Idiopathic Pulmonary Fibrosis. Nat. Rev. Dis. Primers 2017, 3, 17074. [Google Scholar] [CrossRef]
- Li, F.; Wan, D.W.; Hu, J.; Qin, R. Effect of Artificial Skin Membrane on the Expression of miR-155 and miR-506-3p in Patients with Second-Degree Burns. J. Clin. Lab. Anal. 2022, 36, e24564. [Google Scholar] [CrossRef]
- Wu, W.; Jordan, S.; Graf, N.; de Oliveira Pena, J.; Curram, J.; Allanore, Y.; Matucci-Cerinic, M.; Pope, J.E.; Denton, C.P.; Khanna, D.; et al. Progressive Skin Fibrosis Is Associated with a Decline in Lung Function and Worse Survival in Patients with Diffuse Cutaneous Systemic Sclerosis in the European Scleroderma Trials and Research (EUSTAR) Cohort. Ann. Rheum. Dis. 2019, 78, 648–656. [Google Scholar] [CrossRef]
- Li, D.J.; Berry, C.E.; Wan, D.C.; Longaker, M.T. Clinical, Mechanistic, and Therapeutic Landscape of Cutaneous Fibrosis. Sci. Transl. Med. 2024, 16, eadn7871. [Google Scholar] [CrossRef] [PubMed]
- Condorelli, A.G.; El Hachem, M.; Zambruno, G.; Nystrom, A.; Candi, E.; Castiglia, D. Notch-Ing up Knowledge on Molecular Mechanisms of Skin Fibrosis: Focus on the Multifaceted Notch Signalling Pathway. J. Biomed. Sci. 2021, 28, 36. [Google Scholar] [CrossRef]
- Jin, J.; Wang, Z.; Liu, Y.; Chen, J.; Jiang, M.; Lu, L.; Xu, J.; Gao, F.; Wang, J.; Zhang, J.; et al. miR-143-3p Boosts Extracellular Vesicles to Improve the Dermal Fibrosis of Localized Scleroderma. J. Autoimmun. 2025, 153, 103422. [Google Scholar] [CrossRef]
- Xie, L.; Long, X.; Mo, M.; Jiang, J.; Zhang, Q.; Long, M.; Li, M. Bone Marrow Mesenchymal Stem Cell-Derived Exosomes Alleviate Skin Fibrosis in Systemic Sclerosis by Inhibiting the IL-33/ST2 Axis via the Delivery of microRNA-214. Mol. Immunol. 2023, 157, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Hou, J.; Zeng, S.; Wang, X.; Liang, Y.; Zhou, R. METTL3-Mediated m 6A Modification of Pri-miRNA-31 Promotes Hypertrophic Scar Progression. Acta Biochim. Biophys. Sin. 2025, 57, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Spagnolo, P.; Kropski, J.A.; Jones, M.G.; Lee, J.S.; Rossi, G.; Karampitsakos, T.; Maher, T.M.; Tzouvelekis, A.; Ryerson, C.J. Idiopathic Pulmonary Fibrosis: Disease Mechanisms and Drug Development. Pharmacol. Ther. 2021, 222, 107798. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Sun, J.; Zhang, S.; Nie, Y.; Zhou, S.; Zeng, Y. Progress in Understanding and Treating Idiopathic Pulmonary Fibrosis: Recent Insights and Emerging Therapies. Front. Pharmacol. 2023, 14, 1205948. [Google Scholar] [CrossRef]
- Todd, N.W.; Luzina, I.G.; Atamas, S.P. Molecular and Cellular Mechanisms of Pulmonary Fibrosis. Fibrogenesis Tissue Repair. 2012, 5, 11. [Google Scholar] [CrossRef]
- Tanguy, J.; Pommerolle, L.; Garrido, C.; Kolb, M.; Bonniaud, P.; Goirand, F.; Bellaye, P.-S. Extracellular Heat Shock Proteins as Therapeutic Targets and Biomarkers in Fibrosing Interstitial Lung Diseases. Int. J. Mol. Sci. 2021, 22, 9316. [Google Scholar] [CrossRef]
- Selman, M.; Pardo, A. Fibroageing: An Ageing Pathological Feature Driven by Dysregulated Extracellular Matrix-Cell Mechanobiology. Ageing Res. Rev. 2021, 70, 101393. [Google Scholar] [CrossRef]
- Ansary, T.M.; Hossain, M.R.; Kamiya, K.; Komine, M.; Ohtsuki, M. Inflammatory Molecules Associated with Ultraviolet Radiation-Mediated Skin Aging. Int. J. Mol. Sci. 2021, 22, 3974. [Google Scholar] [CrossRef]
- Panich, U.; Sittithumcharee, G.; Rathviboon, N.; Jirawatnotai, S. Ultraviolet Radiation-Induced Skin Aging: The Role of DNA Damage and Oxidative Stress in Epidermal Stem Cell Damage Mediated Skin Aging. Stem Cells Int. 2016, 2016, 7370642. [Google Scholar] [CrossRef]
- Hsieh, C.-C.; Yi, T.-K.; Kao, Y.-F.; Lin, S.-P.; Tu, M.-C.; Chou, Y.-C.; Lu, J.-J.; Chai, H.-J.; Cheng, K.-C. Comparative Efficacy of Botryocladia Leptopoda Extracts in Scar Inhibition and Skin Regeneration: A Study on UV Protection, Collagen Synthesis, and Fibroblast Proliferation. Molecules 2024, 29, 5688. [Google Scholar] [CrossRef]
- Boothe, P.F.; Kumar, V.P.; Kong, Y.; Wang, K.; Levinson, H.; Mu, D.; Brown, M.L. Radiation Induced Skin Fibrosis (RISF): Opportunity for Angiotensin II-Dependent Intervention. Int. J. Mol. Sci. 2024, 25, 8261. [Google Scholar] [CrossRef]
- Rognoni, E.; Goss, G.; Hiratsuka, T.; Sipilä, K.H.; Kirk, T.; Kober, K.I.; Lui, P.P.; Tsang, V.S.; Hawkshaw, N.J.; Pilkington, S.M.; et al. Role of Distinct Fibroblast Lineages and Immune Cells in Dermal Repair Following UV Radiation-Induced Tissue Damage. Elife 2021, 10, e71052. [Google Scholar] [CrossRef]
- Li, C.; Xie, R.; Zhang, S.; Yun, J.; Zhao, T.; Zhong, A.; Zhang, J.; Chen, J. Selective Inhibition of HSF1 Expression in the Heat Shock Pathway of Keloid Fibroblasts Reduces Excessive Fibrosis in Keloid. Arch. Dermatol. Res. 2025, 317, 204. [Google Scholar] [CrossRef]
- Faour, S.; Farahat, M.; Aijaz, A.; Jeschke, M.G. Fibrosis in Burns: An Overview of Mechanisms and Therapies. Am. J. Physiol. Cell Physiol. 2023, 325, C1545–C1557. [Google Scholar] [CrossRef]
- Zhao, W.; Ye, J.; Yang, X.; Wang, J.; Cong, L.; Zhang, Q.; Li, J. Rynchopeterine Inhibits the Formation of Hypertrophic Scars by Regulating the miR-21/HIF1AN Axis. Exp. Cell Res. 2024, 440, 114114. [Google Scholar] [CrossRef]
- Liu, F.; Li, T.; Zhan, X. Silencing Circular RNAPTPN12 Promoted the Growth of Keloid Fibroblasts by Activating Wnt Signaling Pathway via Targeting microRNA-21-5p. Bioengineered 2022, 13, 3503–3515. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Fang, L.; Chen, J.; Zhou, S.; Zhou, K.; Cheng, F.; Cen, Y.; Qing, Y.; Wu, J. Exosomal MicroRNA-21 Promotes Keloid Fibroblast Proliferation and Collagen Production by Inhibiting Smad7. J. Burn Care Res. 2021, 42, 1266–1274. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Wang, L.-Z.; Xiao, R.; Cao, R.; Pan, B.; Lv, X.-Y.; Jiao, H.; Zhuang, Q.; Sun, X.-J.; Liu, Y.-B. Inhibition of microRNA-21-5p Reduces Keloid Fibroblast Autophagy and Migration by Targeting PTEN after Electron Beam Irradiation. Lab. Investig. 2020, 100, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Fang, L.; Cen, Y.; Qing, Y.; Chen, J.; Li, Z. MiR-21 Regulates Keloid Formation by Downregulating Smad7 via the TGF-β/Smad Signaling Pathway. J. Burn Care Res. 2019, 40, 809–817. [Google Scholar] [CrossRef]
- Mehal, W.Z.; Iredale, J.; Friedman, S.L. Scraping Fibrosis: Expressway to the Core of Fibrosis. Nat. Med. 2011, 17, 552–553. [Google Scholar] [CrossRef]
- Massagué, J. TGFβ Signalling in Context. Nat. Rev. Mol. Cell Biol. 2012, 13, 616–630. [Google Scholar] [CrossRef]
- Deng, Z.; Fan, T.; Xiao, C.; Tian, H.; Zheng, Y.; Li, C.; He, J. TGF-β Signaling in Health, Disease, and Therapeutics. Signal Transduct. Target. Ther. 2024, 9, 61. [Google Scholar] [CrossRef]
- Liu, C.; Li, Y.; Semenov, M.; Han, C.; Baeg, G.H.; Tan, Y.; Zhang, Z.; Lin, X.; He, X. Control of Beta-Catenin Phosphorylation/Degradation by a Dual-Kinase Mechanism. Cell 2002, 108, 837–847. [Google Scholar] [CrossRef] [PubMed]
- van Noort, M.; Meeldijk, J.; van der Zee, R.; Destree, O.; Clevers, H. Wnt Signaling Controls the Phosphorylation Status of Beta-Catenin. J. Biol. Chem. 2002, 277, 17901–17905. [Google Scholar] [CrossRef] [PubMed]
- Bhanot, P.; Brink, M.; Samos, C.H.; Hsieh, J.C.; Wang, Y.; Macke, J.P.; Andrew, D.; Nathans, J.; Nusse, R. A New Member of the Frizzled Family from Drosophila Functions as a Wingless Receptor. Nature 1996, 382, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Cong, F.; Schweizer, L.; Varmus, H. Wnt Signals across the Plasma Membrane to Activate the Beta-Catenin Pathway by Forming Oligomers Containing Its Receptors, Frizzled and LRP. Development 2004, 131, 5103–5115. [Google Scholar] [CrossRef]
- Roose, J.; Molenaar, M.; Peterson, J.; Hurenkamp, J.; Brantjes, H.; Moerer, P.; van de Wetering, M.; Destrée, O.; Clevers, H. The Xenopus Wnt Effector XTcf-3 Interacts with Groucho-Related Transcriptional Repressors. Nature 1998, 395, 608–612. [Google Scholar] [CrossRef]
- Hecht, A.; Vleminckx, K.; Stemmler, M.P.; van Roy, F.; Kemler, R. The P300/CBP Acetyltransferases Function as Transcriptional Coactivators of Beta-Catenin in Vertebrates. EMBO J. 2000, 19, 1839–1850. [Google Scholar] [CrossRef]
- Rim, E.Y.; Clevers, H.; Nusse, R. The Wnt Pathway: From Signaling Mechanisms to Synthetic Modulators. Annu. Rev. Biochem. 2022, 91, 571–598. [Google Scholar] [CrossRef]
- van Tienen, L.M.; Mieszczanek, J.; Fiedler, M.; Rutherford, T.J.; Bienz, M. Correction: Constitutive Scaffolding of Multiple Wnt Enhanceosome Components by Legless/BCL9. Elife 2017, 6, e27150, Erratum in Elife 2017, 6, e27150. [Google Scholar] [CrossRef]
- Barker, N.; Hurlstone, A.; Musisi, H.; Miles, A.; Bienz, M.; Clevers, H. The Chromatin Remodelling Factor Brg-1 Interacts with Beta-Catenin to Promote Target Gene Activation. EMBO J. 2001, 20, 4935–4943. [Google Scholar] [CrossRef]
- Maurice, M.M.; Angers, S. Mechanistic Insights into Wnt-β-Catenin Pathway Activation and Signal Transduction. Nat. Rev. Mol. Cell Biol. 2025, 26, 371–388. [Google Scholar] [CrossRef]
- Gumede, D.B.; Abrahamse, H.; Houreld, N.N. Targeting Wnt/β-Catenin Signaling and Its Interplay with TGF-β and Notch Signaling Pathways for the Treatment of Chronic Wounds. Cell Commun. Signal 2024, 22, 244. [Google Scholar] [CrossRef]
- Hynes, R.O. Stretching the Boundaries of Extracellular Matrix Research. Nat. Rev. Mol. Cell Biol. 2014, 15, 761–763. [Google Scholar] [CrossRef] [PubMed]
- Herrera, J.; Henke, C.A.; Bitterman, P.B. Extracellular Matrix as a Driver of Progressive Fibrosis. J. Clin. Investig. 2018, 128, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.W.; Rossi, D.; Peterson, M.; Smith, K.; Sikström, K.; White, E.S.; Connett, J.E.; Henke, C.A.; Larsson, O.; Bitterman, P.B. Fibrotic Extracellular Matrix Activates a Profibrotic Positive Feedback Loop. J. Clin. Investig. 2014, 124, 1622–1635. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Y.; Liu, Y. Fibroblast Activation and Heterogeneity in Fibrotic Disease. Nat. Rev. Nephrol. 2025, 21, 613–632. [Google Scholar] [CrossRef]
- Yang, P.; Luo, Q.; Wang, X.; Fang, Q.; Fu, Z.; Li, J.; Lai, Y.; Chen, X.; Xu, X.; Peng, X.; et al. Comprehensive Analysis of Fibroblast Activation Protein Expression in Interstitial Lung Diseases. Am. J. Respir. Crit. Care Med. 2023, 207, 160–172. [Google Scholar] [CrossRef]
- Gieseck, R.L.; Wilson, M.S.; Wynn, T.A. Type 2 Immunity in Tissue Repair and Fibrosis. Nat. Rev. Immunol. 2018, 18, 62–76. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.E. IL-4 and IL-13: Regulators and Effectors of Wound Repair. Annu. Rev. Immunol. 2023, 41, 229–254. [Google Scholar] [CrossRef] [PubMed]
- Mattoo, H.; Bangari, D.S.; Cummings, S.; Humulock, Z.; Habiel, D.; Xu, E.Y.; Pate, N.; Resnick, R.; Savova, V.; Qian, G.; et al. Molecular Features and Stages of Pulmonary Fibrosis Driven by Type 2 Inflammation. Am. J. Respir. Cell Mol. Biol. 2023, 69, 404–421. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.K.; Austin, E.; Huang, A.; Mamalis, A.; Jagdeo, J. The IL-4/IL-13 Axis in Skin Fibrosis and Scarring: Mechanistic Concepts and Therapeutic Targets. Arch. Dermatol. Res. 2020, 312, 81–92. [Google Scholar] [CrossRef]
- Hu, B.; Phan, S.H. Notch in Fibrosis and as a Target of Anti-Fibrotic Therapy. Pharmacol. Res. 2016, 108, 57–64. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, Z.; Chen, Q.; Zhou, Z. Notch Signaling Regulates Pulmonary Fibrosis. Front. Cell Dev. Biol. 2024, 12, 1450038. [Google Scholar] [CrossRef]
- Shaw, T.J.; Kishi, K.; Mori, R. Wound-Associated Skin Fibrosis: Mechanisms and Treatments Based on Modulating the Inflammatory Response. Endocr. Metab. Immune Disord. Drug Targets 2010, 10, 320–330. [Google Scholar] [CrossRef]
- Smith, G.P.; Chan, E.S.L. Molecular Pathogenesis of Skin Fibrosis: Insight from Animal Models. Curr. Rheumatol. Rep. 2010, 12, 26–33. [Google Scholar] [CrossRef]
- Wang, K.; Wen, D.; Xu, X.; Zhao, R.; Jiang, F.; Yuan, S.; Zhang, Y.; Gao, Y.; Li, Q. Extracellular Matrix Stiffness-The Central Cue for Skin Fibrosis. Front. Mol. Biosci. 2023, 10, 1132353. [Google Scholar] [CrossRef]
- Seguro Paula, F.; Delgado Alves, J. The Role of the Notch Pathway in the Pathogenesis of Systemic Sclerosis: Clinical Implications. Expert Rev. Clin. Immunol. 2021, 17, 1257–1267. [Google Scholar] [CrossRef]
- Aoyagi-Ikeda, K.; Maeno, T.; Matsui, H.; Ueno, M.; Hara, K.; Aoki, Y.; Aoki, F.; Shimizu, T.; Doi, H.; Kawai-Kowase, K.; et al. Notch Induces Myofibroblast Differentiation of Alveolar Epithelial Cells via Transforming Growth Factor-{beta}-Smad3 Pathway. Am. J. Respir. Cell Mol. Biol. 2011, 45, 136–144. [Google Scholar] [CrossRef]
- Blokzijl, A.; Dahlqvist, C.; Reissmann, E.; Falk, A.; Moliner, A.; Lendahl, U.; Ibáñez, C.F. Cross-Talk between the Notch and TGF-Beta Signaling Pathways Mediated by Interaction of the Notch Intracellular Domain with Smad3. J. Cell Biol. 2003, 163, 723–728. [Google Scholar] [CrossRef]
- Dees, C.; Zerr, P.; Tomcik, M.; Beyer, C.; Horn, A.; Akhmetshina, A.; Palumbo, K.; Reich, N.; Zwerina, J.; Sticherling, M.; et al. Inhibition of Notch Signaling Prevents Experimental Fibrosis and Induces Regression of Established Fibrosis. Arthritis Rheum. 2011, 63, 1396–1404. [Google Scholar] [CrossRef] [PubMed]
- Zmorzyński, S.; Styk, W.; Filip, A.A.; Krasowska, D. The Significance of NOTCH Pathway in the Development of Fibrosis in Systemic Sclerosis. Ann. Dermatol. 2019, 31, 365–371. [Google Scholar] [CrossRef]
- Uchida, M.; Shiraishi, H.; Ohta, S.; Arima, K.; Taniguchi, K.; Suzuki, S.; Okamoto, M.; Ahlfeld, S.K.; Ohshima, K.; Kato, S.; et al. Periostin, a Matricellular Protein, Plays a Role in the Induction of Chemokines in Pulmonary Fibrosis. Am. J. Respir. Cell Mol. Biol. 2012, 46, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Takayama, G.; Arima, K.; Kanaji, T.; Toda, S.; Tanaka, H.; Shoji, S.; McKenzie, A.N.J.; Nagai, H.; Hotokebuchi, T.; Izuhara, K. Periostin: A Novel Component of Subepithelial Fibrosis of Bronchial Asthma Downstream of IL-4 and IL-13 Signals. J. Allergy Clin. Immunol. 2006, 118, 98–104. [Google Scholar] [CrossRef]
- Nanri, Y.; Nunomura, S.; Honda, Y.; Takedomi, H.; Yamaguchi, Y.; Izuhara, K. A Positive Loop Formed by SOX11 and Periostin Upregulates TGF-β Signals Leading to Skin Fibrosis. J. Investig. Dermatol. 2023, 143, 989–998.e7. [Google Scholar] [CrossRef]
- Conway, S.J.; Izuhara, K.; Kudo, Y.; Litvin, J.; Markwald, R.; Ouyang, G.; Arron, J.R.; Holweg, C.T.J.; Kudo, A. The Role of Periostin in Tissue Remodeling across Health and Disease. Cell. Mol. Life Sci. 2014, 71, 1279–1288. [Google Scholar] [CrossRef]
- Wang, X.; Huang, W.; Li, Y.; Zhu, C. The Fibroblast Heterogeneity across Keloid, Normal and Tumor Samples from Single-Cell Resolution. Cell. Mol. Biol. 2024, 70, 200–205. [Google Scholar] [CrossRef]
- De Luca, G.; Campochiaro, C.; Burastero, S.E.; Matucci-Cerinic, M.; Doglioni, C.; Dagna, L. Periostin Expression in Uninvolved Skin as a Potential Biomarker for Rapid Cutaneous Progression in Systemic Sclerosis Patients: A Preliminary Explorative Study. Front. Med. 2023, 10, 1214523. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, Z.; Yang, H.; Zhu, J.; Hu, Z. Bioinformatics Analysis and Identification of Dysregulated POSTN in the Pathogenesis of Keloid. Int. Wound J. 2023, 20, 1700–1711. [Google Scholar] [CrossRef]
- Okamoto, M.; Izuhara, K.; Ohta, S.; Ono, J.; Hoshino, T. Ability of Periostin as a New Biomarker of Idiopathic Pulmonary Fibrosis. Adv. Exp. Med. Biol. 2019, 1132, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Hoshino, T.; Kitasato, Y.; Sakazaki, Y.; Kawayama, T.; Fujimoto, K.; Ohshima, K.; Shiraishi, H.; Uchida, M.; Ono, J.; et al. Periostin, a Matrix Protein, Is a Novel Biomarker for Idiopathic Interstitial Pneumonias. Eur. Respir. J. 2011, 37, 1119–1127. [Google Scholar] [CrossRef]
- Sheng, X.R.; Gao, X.; Schiffman, C.; Jiang, J.; Ramalingam, T.R.; Lin, C.J.F.; Khanna, D.; Neighbors, M. Biomarkers of Fibrosis, Inflammation, and Extracellular Matrix in the Phase 3 Trial of Tocilizumab in Systemic Sclerosis. Clin. Immunol. 2023, 254, 109695. [Google Scholar] [CrossRef]
- Adams, T.S.; Schupp, J.C.; Poli, S.; Ayaub, E.A.; Neumark, N.; Ahangari, F.; Chu, S.G.; Raby, B.A.; DeIuliis, G.; Januszyk, M.; et al. Single-Cell RNA-Seq Reveals Ectopic and Aberrant Lung-Resident Cell Populations in Idiopathic Pulmonary Fibrosis. Sci. Adv. 2020, 6, eaba1983. [Google Scholar] [CrossRef]
- Rustam, S.; Hu, Y.; Mahjour, S.B.; Rendeiro, A.F.; Ravichandran, H.; Urso, A.; D’oVidio, F.; Martinez, F.J.; Altorki, N.K.; Richmond, B.; et al. A Unique Cellular Organization of Human Distal Airways and Its Disarray in Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2023, 207, 1171–1182. [Google Scholar] [CrossRef]
- Jaeger, B.; Schupp, J.C.; Plappert, L.; Terwolbeck, O.; Artysh, N.; Kayser, G.; Engelhard, P.; Adams, T.S.; Zweigerdt, R.; Kempf, H.; et al. Airway Basal Cells Show a Dedifferentiated KRT17highPhenotype and Promote Fibrosis in Idiopathic Pulmonary Fibrosis. Nat. Commun. 2022, 13, 5637. [Google Scholar] [CrossRef]
- Danielidi, A.; Lygeros, S.; Anastogianni, A.; Danielidis, G.; Georgiou, S.; Stathopoulos, C.; Grafanaki, K. Genetic and Epigenetic Interconnections Between Atopic Dermatitis, Allergic Rhinitis, and Rhinitis with Nasal Polyps. Allergies 2025, 5, 9. [Google Scholar] [CrossRef]
- Fiuza, B.S.D.; Fonseca, H.F.; Meirelles, P.M.; Marques, C.R.; da Silva, T.M.; Figueiredo, C.A. Understanding Asthma and Allergies by the Lens of Biodiversity and Epigenetic Changes. Front. Immunol. 2021, 12, 623737. [Google Scholar] [CrossRef] [PubMed]
- Potaczek, D.P.; Harb, H.; Michel, S.; Alhamwe, B.A.; Renz, H.; Tost, J. Epigenetics and Allergy: From Basic Mechanisms to Clinical Applications. Epigenomics 2017, 9, 539–571. [Google Scholar] [CrossRef]
- Rock, J.; Königshoff, M. Endogenous Lung Regeneration: Potential and Limitations. Am. J. Respir. Crit. Care Med. 2012, 186, 1213–1219. [Google Scholar] [CrossRef]
- Lafyatis, R. Transforming Growth Factor β--at the Centre of Systemic Sclerosis. Nat. Rev. Rheumatol. 2014, 10, 706–719. [Google Scholar] [CrossRef]
- Jinnin, M. Mechanisms of Skin Fibrosis in Systemic Sclerosis. J. Dermatol. 2010, 37, 11–25. [Google Scholar] [CrossRef]
- Denton, C.P.; Khanna, D. Systemic Sclerosis. Lancet 2017, 390, 1685–1699. [Google Scholar] [CrossRef] [PubMed]
- King, T.E.; Bradford, W.Z.; Castro-Bernardini, S.; Fagan, E.A.; Glaspole, I.; Glassberg, M.K.; Gorina, E.; Hopkins, P.M.; Kardatzke, D.; Lancaster, L.; et al. A Phase 3 Trial of Pirfenidone in Patients with Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2014, 370, 2083–2092. [Google Scholar] [CrossRef]
- Tsukui, T.; Sun, K.-H.; Wetter, J.B.; Wilson-Kanamori, J.R.; Hazelwood, L.A.; Henderson, N.C.; Adams, T.S.; Schupp, J.C.; Poli, S.D.; Rosas, I.O.; et al. Collagen-Producing Lung Cell Atlas Identifies Multiple Subsets with Distinct Localization and Relevance to Fibrosis. Nat. Commun. 2020, 11, 1920. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Wang, Y.; Deng, N.; Huang, G.; Taghavifar, F.; Geng, Y.; Liu, N.; Kulur, V.; Yao, C.; Chen, P.; et al. Single-Cell Deconvolution of Fibroblast Heterogeneity in Mouse Pulmonary Fibrosis. Cell Rep. 2018, 22, 3625–3640. [Google Scholar] [CrossRef]
- Schafer, M.J.; White, T.A.; Iijima, K.; Haak, A.J.; Ligresti, G.; Atkinson, E.J.; Oberg, A.L.; Birch, J.; Salmonowicz, H.; Zhu, Y.; et al. Cellular Senescence Mediates Fibrotic Pulmonary Disease. Nat. Commun. 2017, 8, 14532. [Google Scholar] [CrossRef] [PubMed]
- Kisseleva, T.; Brenner, D.A. Mechanisms of Fibrogenesis. Exp. Biol. Med. 2008, 233, 109–122. [Google Scholar] [CrossRef]
- Horowitz, J.C.; Thannickal, V.J. Mechanisms for the Resolution of Organ Fibrosis. Physiology 2019, 34, 43–55. [Google Scholar] [CrossRef]
- D’Angelo, W.A.; Fries, J.F.; Masi, A.T.; Shulman, L.E. Pathologic Observations in Systemic Sclerosis (Scleroderma). A Study of Fifty-Eight Autopsy Cases and Fifty-Eight Matched Controls. Am. J. Med. 1969, 46, 428–440. [Google Scholar] [CrossRef]
- Lescoat, A.; Huscher, D.; Schoof, N.; Airò, P.; de Vries-Bouwstra, J.; Riemekasten, G.; Hachulla, E.; Doria, A.; Rosato, E.; Hunzelmann, N.; et al. Systemic Sclerosis-Associated Interstitial Lung Disease in the EUSTAR Database: Analysis by Region. Rheumatology 2023, 62, 2178–2188, Erratum in Rheumatology 2023, 62, 3219–3220. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann-Vold, A.-M.; Fretheim, H.; Halse, A.-K.; Seip, M.; Bitter, H.; Wallenius, M.; Garen, T.; Salberg, A.; Brunborg, C.; Midtvedt, Ø.; et al. Tracking Impact of Interstitial Lung Disease in Systemic Sclerosis in a Complete Nationwide Cohort. Am. J. Respir. Crit. Care Med. 2019, 200, 1258–1266. [Google Scholar] [CrossRef]
- Varga, J.; Abraham, D. Systemic Sclerosis: A Prototypic Multisystem Fibrotic Disorder. J. Clin. Investig. 2007, 117, 557–567. [Google Scholar] [CrossRef]
- Milano, A.; Pendergrass, S.A.; Sargent, J.L.; George, L.K.; McCalmont, T.H.; Connolly, M.K.; Whitfield, M.L. Molecular Subsets in the Gene Expression Signatures of Scleroderma Skin. PLoS ONE 2008, 3, e2696, Erratum in PLoS ONE 2008; 3. [Google Scholar] [CrossRef]
- Pendergrass, S.A.; Lemaire, R.; Francis, I.P.; Mahoney, J.M.; Lafyatis, R.; Whitfield, M.L. Intrinsic Gene Expression Subsets of Diffuse Cutaneous Systemic Sclerosis Are Stable in Serial Skin Biopsies. J. Investig. Dermatol. 2012, 132, 1363–1373. [Google Scholar] [CrossRef] [PubMed]
- Hinchcliff, M.; Huang, C.-C.; Wood, T.A.; Matthew Mahoney, J.; Martyanov, V.; Bhattacharyya, S.; Tamaki, Z.; Lee, J.; Carns, M.; Podlusky, S.; et al. Molecular Signatures in Skin Associated with Clinical Improvement during Mycophenolate Treatment in Systemic Sclerosis. J. Investig. Dermatol. 2013, 133, 1979–1989. [Google Scholar] [CrossRef]
- Franks, J.M.; Toledo, D.M.; Martyanov, V.; Wang, Y.; Huang, S.; Wood, T.A.; Spino, C.; Chung, L.; Denton, C.P.; Derrett-Smith, E.; et al. A Genomic Meta-Analysis of Clinical Variables and Their Association with Intrinsic Molecular Subsets in Systemic Sclerosis. Rheumatology 2022, 62, 19–28. [Google Scholar] [CrossRef]
- Johnson, M.E.; Mahoney, J.M.; Taroni, J.; Sargent, J.L.; Marmarelis, E.; Wu, M.-R.; Varga, J.; Hinchcliff, M.E.; Whitfield, M.L. Experimentally-Derived Fibroblast Gene Signatures Identify Molecular Pathways Associated with Distinct Subsets of Systemic Sclerosis Patients in Three Independent Cohorts. PLoS ONE 2015, 10, e0114017. [Google Scholar] [CrossRef]
- Zugmaier, G.; Klinger, M.; Subklewe, M.; Zaman, F.; Locatelli, F. B-Cell-Depleting Immune Therapies as Potential New Treatment Options for Systemic Sclerosis. Sclerosis 2025, 3, 5. [Google Scholar] [CrossRef]
- Perelas, A.; Silver, R.M.; Arrossi, A.V.; Highland, K.B. Systemic Sclerosis-Associated Interstitial Lung Disease. Lancet Respir. Med. 2020, 8, 304–320. [Google Scholar] [CrossRef]
- Raghu, G.; Montesi, S.B.; Silver, R.M.; Hossain, T.; Macrea, M.; Herman, D.; Barnes, H.; Adegunsoye, A.; Azuma, A.; Chung, L.; et al. Treatment of Systemic Sclerosis-Associated Interstitial Lung Disease: Evidence-Based Recommendations. An Official American Thoracic Society Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2024, 209, 137–152. [Google Scholar] [CrossRef]
- Rahaghi, F.F.; Hsu, V.M.; Kaner, R.J.; Mayes, M.D.; Rosas, I.O.; Saggar, R.; Steen, V.D.; Strek, M.E.; Bernstein, E.J.; Bhatt, N.; et al. Expert Consensus on the Management of Systemic Sclerosis-Associated Interstitial Lung Disease. Respir. Res. 2023, 24, 6. [Google Scholar] [CrossRef]
- Khanna, D.; Tashkin, D.P.; Denton, C.P.; Renzoni, E.A.; Desai, S.R.; Varga, J. Etiology, Risk Factors, and Biomarkers in Systemic Sclerosis with Interstitial Lung Disease. Am. J. Respir. Crit. Care Med. 2020, 201, 650–660. [Google Scholar] [CrossRef]
- Vonk, M.C.; Walker, U.A.; Volkmann, E.R.; Kreuter, M.; Johnson, S.R.; Allanore, Y. Natural Variability in the Disease Course of SSc-ILD: Implications for Treatment. Eur. Respir. Rev. 2021, 30, 200340. [Google Scholar] [CrossRef]
- Liaskos, C.; Marou, E.; Simopoulou, T.; Barmakoudi, M.; Efthymiou, G.; Scheper, T.; Meyer, W.; Bogdanos, D.P.; Sakkas, L.I. Disease-Related Autoantibody Profile in Patients with Systemic Sclerosis. Autoimmunity 2017, 50, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Liem, S.I.E.; Neppelenbroek, S.; Fehres, C.M.; Wevers, B.A.; Toes, R.E.M.; Allaart, C.F.; Huizinga, T.W.J.; Scherer, H.U.; De Vries-Bouwstra, J.K. Progression from Suspected to Definite Systemic Sclerosis and the Role of Anti-Topoisomerase I Antibodies. RMD Open 2023, 9, e002827. [Google Scholar] [CrossRef] [PubMed]
- Wortel, C.M.; Liem, S.I.; van Leeuwen, N.M.; Boonstra, M.; Fehres, C.M.; Stöger, L.; Huizinga, T.W.; Toes, R.E.; De Vries-Bouwstra, J.; Scherer, H.U. Anti-Topoisomerase, but Not Anti-Centromere B Cell Responses in Systemic Sclerosis Display Active, Ig-Secreting Cells Associated with Lung Fibrosis. RMD Open 2023, 9, e003148. [Google Scholar] [CrossRef] [PubMed]
- van Oostveen, W.M.; Huizinga, T.W.J.; Fehres, C.M. Pathogenic Role of Anti-Nuclear Autoantibodies in Systemic Sclerosis: Insights from Other Rheumatic Diseases. Immunol. Rev. 2024, 328, 265–282. [Google Scholar] [CrossRef]
- Raschi, E.; Privitera, D.; Bodio, C.; Lonati, P.A.; Borghi, M.O.; Ingegnoli, F.; Meroni, P.L.; Chighizola, C.B. Scleroderma-Specific Autoantibodies Embedded in Immune Complexes Mediate Endothelial Damage: An Early Event in the Pathogenesis of Systemic Sclerosis. Arthritis Res. Ther. 2020, 22, 265. [Google Scholar] [CrossRef]
- Sgonc, R.; Gruschwitz, M.S.; Dietrich, H.; Recheis, H.; Gershwin, M.E.; Wick, G. Endothelial Cell Apoptosis Is a Primary Pathogenetic Event Underlying Skin Lesions in Avian and Human Scleroderma. J. Clin. Investig. 1996, 98, 785–792. [Google Scholar] [CrossRef]
- van Leeuwen, N.M.; Liem, S.I.E.; Maurits, M.P.; Ninaber, M.; Marsan, N.A.; Allaart, C.F.; Huizinga, T.W.J.; Knevel, R.; de Vries-Bouwstra, J.K. Disease Progression in Systemic Sclerosis. Rheumatology 2021, 60, 1565–1567. [Google Scholar] [CrossRef]
- Grafanaki, K.; Grammatikakis, I.; Ghosh, A.; Gopalan, V.; Olgun, G.; Liu, H.; Kyriakopoulos, G.C.; Skeparnias, I.; Georgiou, S.; Stathopoulos, C.; et al. Noncoding RNA Circuitry in Melanoma Onset, Plasticity, and Therapeutic Response. Pharmacol. Ther. 2023, 248, 108466. [Google Scholar] [CrossRef]
- Ciechomska, M.; van Laar, J.M.; O’Reilly, S. Emerging Role of Epigenetics in Systemic Sclerosis Pathogenesis. Genes Immun. 2014, 15, 433–439. [Google Scholar] [CrossRef]
- Liu, Y.; Wen, D.; Ho, C.; Yu, L.; Zheng, D.; O’Reilly, S.; Gao, Y.; Li, Q.; Zhang, Y. Epigenetics as a Versatile Regulator of Fibrosis. J. Transl. Med. 2023, 21, 164. [Google Scholar] [CrossRef]
- Altorok, N.; Tsou, P.-S.; Coit, P.; Khanna, D.; Sawalha, A.H. Genome-Wide DNA Methylation Analysis in Dermal Fibroblasts from Patients with Diffuse and Limited Systemic Sclerosis Reveals Common and Subset-Specific DNA Methylation Aberrancies. Ann. Rheum. Dis. 2015, 74, 1612–1620. [Google Scholar] [CrossRef] [PubMed]
- Baker Frost, D.; da Silveira, W.; Hazard, E.S.; Atanelishvili, I.; Wilson, R.C.; Flume, J.; Day, K.L.; Oates, J.C.; Bogatkevich, G.S.; Feghali-Bostwick, C.; et al. Differential DNA Methylation Landscape in Skin Fibroblasts from African Americans with Systemic Sclerosis. Genes 2021, 12, 129. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Gao, Y.; Yu, D.; Zhong, L.; Cai, W.; Ji, J.; Geng, F.; Tang, G.; Zhang, H.; Cao, J.; et al. Genome-Wide Analysis Reveals Zinc Transporter ZIP9 Regulated by DNA Methylation Promotes Radiation-Induced Skin Fibrosis via the TGF-β Signaling Pathway. J. Investig. Dermatol. 2020, 140, 94–102.e7. [Google Scholar] [CrossRef] [PubMed]
- Huber, L.C.; Distler, J.H.W.; Moritz, F.; Hemmatazad, H.; Hauser, T.; Michel, B.A.; Gay, R.E.; Matucci-Cerinic, M.; Gay, S.; Distler, O.; et al. Trichostatin A Prevents the Accumulation of Extracellular Matrix in a Mouse Model of Bleomycin-Induced Skin Fibrosis. Arthritis Rheum. 2007, 56, 2755–2764. [Google Scholar] [CrossRef]
- Roso-Mares, A.; Andújar, I.; Díaz Corpas, T.; Sun, B.K. Non-Coding RNAs as Skin Disease Biomarkers, Molecular Signatures, and Therapeutic Targets. Hum. Genet. 2024, 143, 801–812. [Google Scholar] [CrossRef]
- Ling, H.; Wang, X.-C.; Liu, Z.-Y.; Mao, S.; Yang, J.-J.; Sha, J.-M.; Tao, H. Noncoding RNA Network Crosstalk in Organ Fibrosis. Cell Signal 2024, 124, 111430. [Google Scholar] [CrossRef]
- Wasson, C.W.; Ross, R.L.; Wells, R.; Corinaldesi, C.; Georgiou, I.C.; Riobo-Del Galdo, N.A.; Del Galdo, F. Long Non-Coding RNA HOTAIR Induces GLI2 Expression through Notch Signalling in Systemic Sclerosis Dermal Fibroblasts. Arthritis Res. Ther. 2020, 22, 286. [Google Scholar] [CrossRef] [PubMed]
- Wasson, C.W.; Abignano, G.; Hermes, H.; Malaab, M.; Ross, R.L.; Jimenez, S.A.; Chang, H.Y.; Feghali-Bostwick, C.A.; Del Galdo, F. Long Non-Coding RNA HOTAIR Drives EZH2-Dependent Myofibroblast Activation in Systemic Sclerosis through miRNA 34a-Dependent Activation of NOTCH. Ann. Rheum. Dis. 2020, 79, 507–517. [Google Scholar] [CrossRef]
- Yu, X.; Zhu, X.; Xu, H.; Li, L. Emerging Roles of Long Non-Coding RNAs in Keloids. Front. Cell Dev. Biol. 2022, 10, 963524. [Google Scholar] [CrossRef]
- Pan, L.; Sun, C.; Jin, H.; Lv, S. LINC01711 Modulates Proliferation, Migration, and Extracellular Matrix Deposition of Hypertrophic Scar Fibroblasts by Targeting miR-34a-5p. Arch. Dermatol. Res. 2025, 317, 736. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yin, Y.; Li, J.; Li, Q.; Zhu, Z.; Li, J. LINC00525 Promotes Cell Proliferation and Collagen Expression through Feedforward Regulation of TGF-β Signaling in Hypertrophic Scar Fibroblasts. Burns 2025, 51, 107353. [Google Scholar] [CrossRef]
- Bu, W.; Fang, F.; Zhang, M.; Zhou, W. Long Non-Coding RNA Uc003jox.1 Promotes Keloid Fibroblast Proliferation and Invasion Through Activating the PI3K/AKT Signaling Pathway. J. Craniofacial Surg. 2023, 34, 556–560. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, B.; Lu, J.; Li, A.; Wa, Q. LncRNA SNHG1/miR-320b/CTNNB1 Axis Regulating the Collective Migration of Fibroblasts in the Formation of Keloid. Cutan. Ocul. Toxicol. 2025, 44, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, L.; Wang, J.-Y.; Gao, F.; Lin, X.; Lin, S.-S.; Qiu, Z.-Y.; Liang, Z.-H. LncRNA GNAS-AS1 Knockdown Inhibits Keloid Cells Growth by Mediating the miR-188-5p/RUNX2 Axis. Mol. Cell. Biochem. 2023, 478, 707–719. [Google Scholar] [CrossRef]
- Zhou, Q.; Chung, A.C.K.; Huang, X.R.; Dong, Y.; Yu, X.; Lan, H.Y. Identification of Novel Long Noncoding RNAs Associated with TGF-β/Smad3-Mediated Renal Inflammation and Fibrosis by RNA Sequencing. Am. J. Pathol. 2014, 184, 409–417. [Google Scholar] [CrossRef]
- Yang, Z.; Jiang, S.; Shang, J.; Jiang, Y.; Dai, Y.; Xu, B.; Yu, Y.; Liang, Z.; Yang, Y. LncRNA: Shedding Light on Mechanisms and Opportunities in Fibrosis and Aging. Ageing Res. Rev. 2019, 52, 17–31. [Google Scholar] [CrossRef]
- Sanders, Y.Y.; Ambalavanan, N.; Halloran, B.; Zhang, X.; Liu, H.; Crossman, D.K.; Bray, M.; Zhang, K.; Thannickal, V.J.; Hagood, J.S. Altered DNA Methylation Profile in Idiopathic Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2012, 186, 525–535. [Google Scholar] [CrossRef]
- Luo, Q.-K.; Zhang, H.; Li, L. Research Advances on DNA Methylation in Idiopathic Pulmonary Fibrosis. In Single-Cell Sequencing and Methylation: Methods and Clinical Applications; Yu, B., Zhang, J., Zeng, Y., Li, L., Wang, X., Eds.; Springer: Singapore, 2020; pp. 73–81. ISBN 978-981-15-4494-1. [Google Scholar]
- Korytina, G.F.; Markelov, V.A.; Gibadullin, I.A.; Zulkarneev, S.R.; Nasibullin, T.R.; Zulkarneev, R.H.; Avzaletdinov, A.M.; Avdeev, S.N.; Zagidullin, N.S. The Relationship Between Differential Expression of Non-Coding RNAs (TP53TG1, LINC00342, MALAT1, DNM3OS, miR-126-3p, miR-200a-3p, miR-18a-5p) and Protein-Coding Genes (PTEN, FOXO3) and Risk of Idiopathic Pulmonary Fibrosis. Biochem. Genet. 2025; epub ahead of print. [Google Scholar] [CrossRef]
- Zhang, Y.-S.; Tu, B.; Song, K.; Lin, L.-C.; Liu, Z.-Y.; Lu, D.; Chen, Q.; Tao, H. Epigenetic Hallmarks in Pulmonary Fibrosis: New Advances and Perspectives. Cell Signal 2023, 110, 110842. [Google Scholar] [CrossRef]
- Qin, W.; Spek, C.A.; Scicluna, B.P.; van der Poll, T.; Duitman, J. Myeloid DNA Methyltransferase3b Deficiency Aggravates Pulmonary Fibrosis by Enhancing Profibrotic Macrophage Activation. Respir. Res. 2022, 23, 162. [Google Scholar] [CrossRef]
- Liu, M.; Song, L.; Lai, Y.; Gao, F.; Man, J. LncRNA FEZF1-AS1 Promotes Pulmonary Fibrosis via up-Regulating EZH2 and Targeting miR-200c-3p to Regulate the ZEB1 Pathway. Sci. Rep. 2024, 14, 26044. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.K.; Gomer, R.H. Translational Regulators in Pulmonary Fibrosis: MicroRNAs, Long Non-Coding RNAs, and Transcript Modifications. Cells 2025, 14, 536. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Wei, Y.; Li, C.; Xie, B.; Zhang, X.; Cui, Y.; Dai, H. A Novel lncRNA ABCE1-5 Regulates Pulmonary Fibrosis by Targeting KRT14. Am. J. Physiol. Cell Physiol. 2025, 328, C1487–C1500. [Google Scholar] [CrossRef]
- Takashima, T.; Zeng, C.; Murakami, E.; Fujiwara, N.; Kohara, M.; Nagata, H.; Feng, Z.; Sugai, A.; Harada, Y.; Ichijo, R.; et al. Involvement of lncRNA MIR205HG in Idiopathic Pulmonary Fibrosis and IL-33 Regulation via Alu Elements. JCI Insight 2025, 10, e187172. [Google Scholar] [CrossRef]
- Wu, W.; Yu, N.; Chen, W.; Zhu, Y. ANRIL Upregulates TGFBR1 to Promote Idiopathic Pulmonary Fibrosis in TGF-Β1-Treated Lung Fibroblasts via Sequestering Let-7d-5p. Epigenetics 2024, 19, 2435682. [Google Scholar] [CrossRef] [PubMed]
- Alzahrani, A.R.; Mohamed, D.I.; Abo Nahas, H.H.; Alaa El-Din Aly El-Waseef, D.; Altamimi, A.S.; Youssef, I.H.; Ibrahim, I.A.A.; Mohamed, S.M.Y.; Sabry, Y.G.; Falemban, A.H.; et al. Trimetazidine Alleviates Bleomycin-Induced Pulmonary Fibrosis by Targeting the Long Noncoding RNA CBR3-AS1-Mediated miRNA-29 and Resistin-Like Molecule Alpha 1: Deciphering a Novel Trifecta Role of LncRNA CBR3-AS1/miRNA-29/FIZZ1 Axis in Lung Fibrosis. Drug Des. Devel. Ther. 2024, 18, 3959–3986. [Google Scholar] [CrossRef]
- López-Martínez, A.; Santos-Álvarez, J.C.; Velázquez-Enríquez, J.M.; Ramírez-Hernández, A.A.; Vásquez-Garzón, V.R.; Baltierrez-Hoyos, R. lncRNA-mRNA Co-Expression and Regulation Analysis in Lung Fibroblasts from Idiopathic Pulmonary Fibrosis. Non-Coding RNA 2024, 10, 26. [Google Scholar] [CrossRef]
- Song, X.; Cao, G.; Jing, L.; Lin, S.; Wang, X.; Zhang, J.; Wang, M.; Liu, W.; Lv, C. Analysing the Relationship between lncRNA and Protein-Coding Gene and the Role of lncRNA as ceRNA in Pulmonary Fibrosis. J. Cell. Mol. Med. 2014, 18, 991–1003. [Google Scholar] [CrossRef]
- Wei, Y.; Hong, M.; Zhu, H.; Li, F. Recent Progress in Exosomal Non-Coding RNAs Research Related to Idiopathic Pulmonary Fibrosis. Front. Genet. 2025, 16, 1556495. [Google Scholar] [CrossRef] [PubMed]
- Dirol, H.; Toylu, A.; Ogus, A.C.; Cilli, A.; Ozbudak, O.; Clark, O.A.; Ozdemir, T. Alterations in Plasma miR-21, miR-590, miR-192 and miR-215 in Idiopathic Pulmonary Fibrosis and Their Clinical Importance. Mol. Biol. Rep. 2022, 49, 2237–2244. [Google Scholar] [CrossRef]
- Lu, Y.; Liu, Z.; Zhang, Y.; Wu, X.; Bian, W.; Shan, S.; Yang, D.; Ren, T. METTL3-Mediated m6A RNA Methylation Induces the Differentiation of Lung Resident Mesenchymal Stem Cells into Myofibroblasts via the miR-21/PTEN Pathway. Respir. Res. 2023, 24, 300. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Su, Y.; Hsia, I.; Xu, Y.; Vincent-Chong, V.K.; Mojica, W.; Seshadri, M.; Zhao, R.; Wu, Y. Delivery of Anti-microRNA-21 by Lung-Targeted Liposomes for Pulmonary Fibrosis Treatment. Mol. Ther. Nucleic Acids 2023, 32, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Maurer, B.; Stanczyk, J.; Jüngel, A.; Akhmetshina, A.; Trenkmann, M.; Brock, M.; Kowal-Bielecka, O.; Gay, R.E.; Michel, B.A.; Distler, J.H.W.; et al. MicroRNA-29, a Key Regulator of Collagen Expression in Systemic Sclerosis. Arthritis Rheum. 2010, 62, 1733–1743. [Google Scholar] [CrossRef]
- Xiao, J.; Meng, X.-M.; Huang, X.R.; Chung, A.C.; Feng, Y.-L.; Hui, D.S.; Yu, C.-M.; Sung, J.J.; Lan, H.Y. miR-29 Inhibits Bleomycin-Induced Pulmonary Fibrosis in Mice. Mol. Ther. 2012, 20, 1251–1260. [Google Scholar] [CrossRef]
- Yamada, Y.; Takanashi, M.; Sudo, K.; Ueda, S.; Ohno, S.-I.; Kuroda, M. Novel Form of miR-29b Suppresses Bleomycin-Induced Pulmonary Fibrosis. PLoS ONE 2017, 12, e0171957. [Google Scholar] [CrossRef]
- Gallant-Behm, C.L.; Piper, J.; Lynch, J.M.; Seto, A.G.; Hong, S.J.; Mustoe, T.A.; Maari, C.; Pestano, L.A.; Dalby, C.M.; Jackson, A.L.; et al. A MicroRNA-29 Mimic (Remlarsen) Represses Extracellular Matrix Expression and Fibroplasia in the Skin. J. Investig. Dermatol. 2019, 139, 1073–1081. [Google Scholar] [CrossRef]
- Li, Y.; Xiao, Y.; Han, Y.; Zhu, H.; Han, J.; Wang, H. Blocking the MIR155HG/miR-155 Axis Reduces CTGF-Induced Inflammatory Cytokine Production and α-SMA Expression via Upregulating AZGP1 in Hypertrophic Scar Fibroblasts. Cell. Signal. 2024, 120, 111202. [Google Scholar] [CrossRef]
- Wang, C.-R.; Zhu, H.-F.; Zhu, Y. Knockout of MicroRNA-155 Ameliorates the Th17/Th9 Immune Response and Promotes Wound Healing. Curr. Med. Sci. 2019, 39, 954–964. [Google Scholar] [CrossRef]
- Wu, X.; Li, J.; Yang, X.; Bai, X.; Shi, J.; Gao, J.; Li, Y.; Han, S.; Zhang, Y.; Han, F.; et al. miR-155 Inhibits the Formation of Hypertrophic Scar Fibroblasts by Targeting HIF-1α via PI3K/AKT Pathway. J. Mol. Histol. 2018, 49, 377–387. [Google Scholar] [CrossRef]
- Yang, L.-L.; Liu, J.-Q.; Bai, X.-Z.; Fan, L.; Han, F.; Jia, W.-B.; Su, L.-L.; Shi, J.-H.; Tang, C.-W.; Hu, D.-H. Acute Downregulation of miR-155 at Wound Sites Leads to a Reduced Fibrosis through Attenuating Inflammatory Response. Biochem. Biophys. Res. Commun. 2014, 453, 153–159. [Google Scholar] [CrossRef]
- Christmann, R.B.; Wooten, A.; Sampaio-Barros, P.; Borges, C.L.; Carvalho, C.R.R.; Kairalla, R.A.; Feghali-Bostwick, C.; Ziemek, J.; Mei, Y.; Goummih, S.; et al. miR-155 in the Progression of Lung Fibrosis in Systemic Sclerosis. Arthritis Res. Ther. 2016, 18, 155. [Google Scholar] [CrossRef] [PubMed]
- Condorelli, A.G.; Logli, E.; Cianfarani, F.; Teson, M.; Diociaiuti, A.; El Hachem, M.; Zambruno, G.; Castiglia, D.; Odorisio, T. MicroRNA-145-5p Regulates Fibrotic Features of Recessive Dystrophic Epidermolysis Bullosa Skin Fibroblasts. Br. J. Dermatol. 2019, 181, 1017–1027. [Google Scholar] [CrossRef]
- Sun, W.; Zhou, S.; Peng, L.; Liu, Y.; Cheng, D.; Wang, Y.; Ni, C. CircZNF609 Regulates Pulmonary Fibrosis via miR-145-5p/KLF4 Axis and Its Translation Function. Cell. Mol. Biol. Lett. 2023, 28, 105. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Wu, Y.X.; Sun, J.X.; Wang, F.C.; Cui, Z.Q.; Xu, X.H. The Role of miR-145 in Promoting the Fibrosis of Pulmonary Fibroblasts. J. Biol. Regul. Homeost. Agents 2019, 33, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Cui, H.; Xie, N.; Icyuz, M.; Banerjee, S.; Antony, V.B.; Abraham, E.; Thannickal, V.J.; Liu, G. miR-145 Regulates Myofibroblast Differentiation and Lung Fibrosis. FASEB J. 2013, 27, 2382–2391. [Google Scholar] [CrossRef]
- Chen, L.; Yang, Y.; Peng, X.; Yan, H.; Zhang, X.; Yin, L.; Yu, H. Transcription Factor YY1 Inhibits the Expression of THY1 to Promote Interstitial Pulmonary Fibrosis by Activating the HSF1/miR-214 Axis. Aging 2020, 12, 8339–8351. [Google Scholar] [CrossRef]
- Vrijheid, M. The Exposome: A New Paradigm to Study the Impact of Environment on Health. Thorax 2014, 69, 876–878. [Google Scholar] [CrossRef]
- Grafanaki, K.; Bania, A.; Kaliatsi, E.G.; Vryzaki, E.; Vasilopoulos, Y.; Georgiou, S. The Imprint of Exposome on the Development of Atopic Dermatitis across the Lifespan: A Narrative Review. J. Clin. Med. 2023, 12, 2180. [Google Scholar] [CrossRef]
- Park, Y.; Ahn, C.; Kim, T.-H. Occupational and Environmental Risk Factors of Idiopathic Pulmonary Fibrosis: A Systematic Review and Meta-Analyses. Sci. Rep. 2021, 11, 4318. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, S.; Bao, S.; Yao, L.; Wen, Z.; Xu, L.; Chen, X.; Guo, S.; Pang, H.; Zhou, Y.; et al. Deciphering the Fibrotic Process: Mechanism of Chronic Radiation Skin Injury Fibrosis. Front. Immunol. 2024, 15, 1338922. [Google Scholar] [CrossRef]
- Salminen, A.; Kaarniranta, K.; Kauppinen, A. Photoaging: UV Radiation-Induced Inflammation and Immunosuppression Accelerate the Aging Process in the Skin. Inflamm. Res. 2022, 71, 817–831. [Google Scholar] [CrossRef]
- Isler, M.F.; Coates, S.J.; Boos, M.D. Climate Change, the Cutaneous Microbiome and Skin Disease: Implications for a Warming World. Int. J. Dermatol. 2023, 62, 337–345. [Google Scholar] [CrossRef]
- Belzer, A.; Parker, E.R. Climate Change, Skin Health, and Dermatologic Disease: A Guide for the Dermatologist. Am. J. Clin. Dermatol. 2023, 24, 577–593. [Google Scholar] [CrossRef]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The Skin Aging Exposome. J. Dermatol. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef]
- Krutmann, J.; Schikowski, T.; Morita, A.; Berneburg, M. Environmentally-Induced (Extrinsic) Skin Aging: Exposomal Factors and Underlying Mechanisms. J. Investig. Dermatol. 2021, 141, 1096–1103. [Google Scholar] [CrossRef]
- He, J.; Cheng, X.; Fang, B.; Shan, S.; Li, Q. Mechanical Stiffness Promotes Skin Fibrosis via Piezo1-Wnt2/Wnt11-CCL24 Positive Feedback Loop. Cell Death Dis. 2024, 15, 84. [Google Scholar] [CrossRef]
- Grafanaki, K.; Antonatos, C.; Maniatis, A.; Petropoulou, A.; Vryzaki, E.; Vasilopoulos, Y.; Georgiou, S.; Gregoriou, S. Intrinsic Effects of Exposome in Atopic Dermatitis: Genomics, Epigenomics and Regulatory Layers. J. Clin. Med. 2023, 12, 4000. [Google Scholar] [CrossRef]
- Tahara, M.; Fujino, Y.; Yamasaki, K.; Oda, K.; Kido, T.; Sakamoto, N.; Kawanami, T.; Kataoka, K.; Egashira, R.; Hashisako, M.; et al. Exposure to PM2.5 Is a Risk Factor for Acute Exacerbation of Surgically Diagnosed Idiopathic Pulmonary Fibrosis: A Case-Control Study. Respir. Res. 2021, 22, 80. [Google Scholar] [CrossRef]
- Bezerra, F.S.; Lanzetti, M.; Nesi, R.T.; Nagato, A.C.; Silva, C.P.e.; Kennedy-Feitosa, E.; Melo, A.C.; Cattani-Cavalieri, I.; Porto, L.C.; Valenca, S.S. Oxidative Stress and Inflammation in Acute and Chronic Lung Injuries. Antioxidants 2023, 12, 548. [Google Scholar] [CrossRef]
- Lechowicz, K.; Drożdżal, S.; Machaj, F.; Rosik, J.; Szostak, B.; Zegan-Barańska, M.; Biernawska, J.; Dabrowski, W.; Rotter, I.; Kotfis, K. COVID-19: The Potential Treatment of Pulmonary Fibrosis Associated with SARS-CoV-2 Infection. J. Clin. Med. 2020, 9, 1917. [Google Scholar] [CrossRef]
- Balmes, J.R.; Hicks, A.; Johnson, M.M.; Nadeau, K.C. The Effect of Wildfires on Asthma and Allergies. J. Allergy Clin. Immunol. Pract. 2025, 13, 280–287. [Google Scholar] [CrossRef]
- Reid, C.E.; Maestas, M.M. Wildfire Smoke Exposure under Climate Change: Impact on Respiratory Health of Affected Communities. Curr. Opin. Pulm. Med. 2019, 25, 179–187. [Google Scholar] [CrossRef]
- Wilgus, M.-L.; Merchant, M. Clearing the Air: Understanding the Impact of Wildfire Smoke on Asthma and COPD. Healthcare 2024, 12, 307. [Google Scholar] [CrossRef] [PubMed]
- Hong, D.S.; Banerji, U.; Tavana, B.; George, G.C.; Aaron, J.; Kurzrock, R. Targeting the Molecular Chaperone Heat Shock Protein 90 (HSP90): Lessons Learned and Future Directions. Cancer Treat. Rev. 2013, 39, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Bellaye, P.-S.; Burgy, O.; Causse, S.; Garrido, C.; Bonniaud, P. Heat Shock Proteins in Fibrosis and Wound Healing: Good or Evil? Pharmacol. Ther. 2014, 143, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, X.; Huang, W.; Ge, X. The Role of Heat Shock Proteins in the Regulation of Fibrotic Diseases. Biomed. Pharmacother. 2021, 135, 111067. [Google Scholar] [CrossRef]
- Dragon, J.; Hoaglund, M.; Badireddy, A.R.; Nielsen, G.; Schlezinger, J.; Shukla, A. Perfluoroalkyl Substances (PFAS) Affect Inflammation in Lung Cells and Tissues. Int. J. Mol. Sci. 2023, 24, 8539. [Google Scholar] [CrossRef]
- Solan, M.E.; Park, J.-A. Per- and Poly-Fluoroalkyl Substances (PFAS) Effects on Lung Health: A Perspective on the Current Literature and Future Recommendations. Front. Toxicol. 2024, 6, 1423449. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Zou, Y.; Wei, G.; Zhang, C.; Zhang, K.; Guo, H.; Xiong, W. Moderate-Intensity Physical Activity Reduces the Role of Serum PFAS on COPD: A Cross-Sectional Analysis with NHANES Data. PLoS ONE 2024, 19, e0308148. [Google Scholar] [CrossRef]
- Kornher, K.; Gould, C.F.; Manzano, J.M.; Baines, K.; Kayser, G.; Tu, X.; Suarez-Torres, J.; Martinez, D.; Peterson, L.A.; Huset, C.A.; et al. Associations of PFAS and Pesticides with Lung Function Changes from Adolescence to Young Adulthood in the ESPINA Study. Int. J. Hyg. Environ. Health 2025, 265, 114526. [Google Scholar] [CrossRef]
- Karakoltzidis, A.; Karakitsios, S.P.; Gabriel, C.; Sarigiannis, D.A. Integrated PBPK Modelling for PFOA Exposure and Risk Assessment. Environ. Res. 2025, 282, 121947. [Google Scholar] [CrossRef]
- Wright, R.O.; Makris, K.C.; Natsiavas, P.; Fennell, T.; Rushing, B.R.; Wilson, A.; Members of the Exposomics Consortium. A Long and Winding Road: Culture Change on Data Sharing in Exposomics. Exposome 2024, 4, osae004. [Google Scholar] [CrossRef]
- Chung, M.K.; House, J.S.; Akhtari, F.S.; Makris, K.C.; Langston, M.A.; Islam, K.T.; Holmes, P.; Chadeau-Hyam, M.; Smirnov, A.I.; Du, X.; et al. Decoding the Exposome: Data Science Methodologies and Implications in Exposome-Wide Association Studies (ExWASs). Exposome 2024, 4, osae001. [Google Scholar] [CrossRef]
- Bertino, L.; Guarneri, F.; Cannavò, S.P.; Casciaro, M.; Pioggia, G.; Gangemi, S. Oxidative Stress and Atopic Dermatitis. Antioxidants 2020, 9, 196. [Google Scholar] [CrossRef]
- Mousavi, S.E.; Delgado-Saborit, J.M.; Godderis, L. Exposure to Per- and Polyfluoroalkyl Substances and Premature Skin Aging. J. Hazard. Mater. 2021, 405, 124256. [Google Scholar] [CrossRef]
- Denisow-Pietrzyk, M. Human Skin Reflects Air Pollution—A Review of the Mechanisms and Clinical Manifestations of Environment-Derived Skin Pathologies. Pol. J. Environ. Stud. 2021, 30, 3433–3444. [Google Scholar] [CrossRef]
- Araviiskaia, E.; Berardesca, E.; Bieber, T.; Gontijo, G.; Sanchez Viera, M.; Marrot, L.; Chuberre, B.; Dreno, B. The Impact of Airborne Pollution on Skin. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1496–1505. [Google Scholar] [CrossRef] [PubMed]
- Passeron, T.; Zouboulis, C.C.; Tan, J.; Andersen, M.L.; Katta, R.; Lyu, X.; Aguilar, L.; Kerob, D.; Morita, A.; Krutmann, J.; et al. Adult Skin Acute Stress Responses to Short-Term Environmental and Internal Aggression from Exposome Factors. J. Eur. Acad. Dermatol. Venereol. JEADV 2021, 35, 1963–1975. [Google Scholar] [CrossRef]
- Gu, X.; Li, Z.; Su, J. Air Pollution and Skin Diseases: A Comprehensive Evaluation of the Associated Mechanism. Ecotoxicol. Environ. Saf. 2024, 278, 116429. [Google Scholar] [CrossRef]
- Dijkhoff, I.M.; Drasler, B.; Karakocak, B.B.; Petri-Fink, A.; Valacchi, G.; Eeman, M.; Rothen-Rutishauser, B. Impact of Airborne Particulate Matter on Skin: A Systematic Review from Epidemiology to In Vitro Studies. Part. Fibre Toxicol. 2020, 17, 35. [Google Scholar] [CrossRef]
- Abolhasani, R.; Araghi, F.; Tabary, M.; Aryannejad, A.; Mashinchi, B.; Robati, R.M. The Impact of Air Pollution on Skin and Related Disorders: A Comprehensive Review. Dermatol. Ther. 2021, 34, e14840. [Google Scholar] [CrossRef]
- Haykal, D.; Lim, H.W.; Calzavara-Pinton, P.; Fluhr, J.; Cartier, H.; Berardesca, E. The Impact of Pollution and Climate Change on Skin Health: Mechanisms, Protective Strategies, and Future Directions. JAAD Rev. 2025, 6, 1–11. [Google Scholar] [CrossRef]
- Pan, Y.; Mei, J.; Jiang, J.; Xu, K.; Gao, X.; Jiang, S.; Liu, Y. PFAS in PMs Might Be the Escalating Hazard to the Lung Health. Nano Res. 2023, 16, 13113–13133. [Google Scholar] [CrossRef]
- Mariscal-Aguilar, P.; Gómez-Carrera, L.; Bonilla, G.; Díaz-Almirón, M.; Gayá, F.; Carpio, C.; Zamarrón, E.; Fernández-Velilla, M.; Torres, I.; Esteban, I.; et al. Air Pollution Exposure and Its Effects on Idiopathic Pulmonary Fibrosis: Clinical Worsening, Lung Function Decline, and Radiological Deterioration. Front. Public Health 2023, 11, 1331134. [Google Scholar] [CrossRef]
- Lan, D.; Fermoyle, C.C.; Troy, L.K.; Knibbs, L.D.; Corte, T.J. The Impact of Air Pollution on Interstitial Lung Disease: A Systematic Review and Meta-Analysis. Front. Med. 2023, 10, 1321038. [Google Scholar] [CrossRef] [PubMed]
- Goobie, G.C.; Carlsten, C.; Johannson, K.A.; Khalil, N.; Marcoux, V.; Assayag, D.; Manganas, H.; Fisher, J.H.; Kolb, M.R.J.; Lindell, K.O.; et al. Association of Particulate Matter Exposure with Lung Function and Mortality Among Patients with Fibrotic Interstitial Lung Disease. JAMA Intern. Med. 2022, 182, 1248–1259. [Google Scholar] [CrossRef] [PubMed]
- Sack, C.; Wojdyla, D.M.; MacMurdo, M.G.; Gassett, A.; Kaufman, J.D.; Raghu, G.; Redlich, C.A.; Li, P.; Olson, A.L.; Leonard, T.B.; et al. Long-Term Air Pollution Exposure and Severity of Idiopathic Pulmonary Fibrosis: Data from the Idiopathic Pulmonary Fibrosis Prospective Outcomes (IPF-PRO) Registry. Ann. Am. Thorac. Soc. 2025, 22, 378–386. [Google Scholar] [CrossRef]
- Wang, Q.; Goracci, C.; Sundar, I.K.; Rahman, I. Environmental Tobacco Smoke Exposure Exaggerates Bleomycin-Induced Collagen Overexpression during Pulmonary Fibrogenesis. J. Inflamm. 2024, 21, 9. [Google Scholar] [CrossRef]
- Majewski, S.; Piotrowski, W.J. Air Pollution-An Overlooked Risk Factor for Idiopathic Pulmonary Fibrosis. J. Clin. Med. 2020, 10, 77. [Google Scholar] [CrossRef]
- Mariscal-Aguilar, P.; Gómez-Carrera, L.; Bonilla, G.; Carpio, C.; Zamarrón, E.; Fernández-Velilla, M.; Díaz-Almirón, M.; Gayá, F.; Villamañán, E.; Prados, C.; et al. Impact of Air Pollution on the Long-Term Decline of Non-Idiopathic Pulmonary Fibrosis Interstitial Lung Disease. Atmosphere 2024, 15, 1405. [Google Scholar] [CrossRef]
- Tomos, I.; Dimakopoulou, K.; Manali, E.D.; Papiris, S.A.; Karakatsani, A. Long-Term Personal Air Pollution Exposure and Risk for Acute Exacerbation of Idiopathic Pulmonary Fibrosis. Environ. Health 2021, 20, 99. [Google Scholar] [CrossRef]
- Díaz-Gay, M.; Zhang, T.; Hoang, P.H.; Khandekar, A.; Zhao, W.; Steele, C.D.; Otlu, B.; Nandi, S.P.; Vangara, R.; Bergstrom, E.N.; et al. The Mutagenic Forces Shaping the Genomic Landscape of Lung Cancer in Never Smokers. medRxiv 2024. [Google Scholar] [CrossRef] [PubMed]
- Mookherjee, N.; Piyadasa, H.; Ryu, M.H.; Rider, C.F.; Ezzati, P.; Spicer, V.; Carlsten, C. Inhaled Diesel Exhaust Alters the Allergen-Induced Bronchial Secretome in Humans. Eur. Respir. J. 2018, 51, 1701385. [Google Scholar] [CrossRef] [PubMed]
- Goobie, G.C.; Nouraie, M.; Zhang, Y.; Kass, D.J.; Ryerson, C.J.; Carlsten, C.; Johannson, K.A. Air Pollution and Interstitial Lung Diseases: Defining Epigenomic Effects. Am. J. Respir. Crit. Care Med. 2020, 202, 1217–1224. [Google Scholar] [CrossRef]
- Zhao, Z.; Rong, Y.; Yin, R.; Zeng, R.; Xu, Z.; Lv, D.; Hu, Z.; Cao, X.; Tang, B. Skin Microbiota, Immune Cell, and Skin Fibrosis: A Comprehensive Mendelian Randomization Study. Biomedicines 2024, 12, 2409. [Google Scholar] [CrossRef]
- Siu, M.C.; Voisey, J.; Zang, T.; Cuttle, L. MicroRNAs Involved in Human Skin Burns, Wound Healing and Scarring. Wound Repair. Regen. 2023, 31, 439–453. [Google Scholar] [CrossRef]
- Afthab, M.; Hambo, S.; Kim, H.; Alhamad, A.; Harb, H. Particulate Matter-Induced Epigenetic Modifications and Lung Complications. Eur. Respir. Rev. 2024, 33, 240129. [Google Scholar] [CrossRef]
- Zhang, N.; Liu, K.; Wang, K.; Zhou, C.; Wang, H.; Che, S.; Liu, Z.; Yang, H. Dust Induces Lung Fibrosis through Dysregulated DNA Methylation. Environ. Toxicol. 2019, 34, 728–741. [Google Scholar] [CrossRef]
- Goobie, G.C.; Li, X.; Ryerson, C.J.; Carlsten, C.; Johannson, K.A.; Fabisiak, J.P.; Lindell, K.O.; Chen, X.; Gibson, K.F.; Kass, D.J.; et al. PM2.5 and Constituent Component Impacts on Global DNA Methylation in Patients with Idiopathic Pulmonary Fibrosis. Environ. Pollut. 2023, 318, 120942. [Google Scholar] [CrossRef]
- Tang, B.; Shi, Y.; Zeng, Z.; He, X.; Yu, J.; Chai, K.; Liu, J.; Liu, L.; Zhan, Y.; Qiu, X.; et al. Silica’s Silent Threat: Contributing to Skin Fibrosis in Systemic Sclerosis by Targeting the HDAC4/Smad2/3 Pathway. Environ. Pollut. 2024, 355, 124194. [Google Scholar] [CrossRef]
- Ramos, P.S. Epigenetics of Scleroderma: Integrating Genetic, Ethnic, Age, and Environmental Effects. J. Scleroderma Relat. Disord. 2019, 4, 238–250. [Google Scholar] [CrossRef]
- Bridges, J.P.; Vladar, E.K.; Kurche, J.S.; Krivoi, A.; Stancil, I.T.; Dobrinskikh, E.; Hu, Y.; Sasse, S.K.; Lee, J.S.; Blumhagen, R.Z.; et al. Progressive Lung Fibrosis: Reprogramming a Genetically Vulnerable Bronchoalveolar Epithelium. J. Clin. Investig. 2025, 135, e183836. [Google Scholar] [CrossRef]
- Tang, X.; Yang, T.; Yu, D.; Xiong, H.; Zhang, S. Current Insights and Future Perspectives of Ultraviolet Radiation (UV) Exposure: Friends and Foes to the Skin and beyond the Skin. Environ. Int. 2024, 185, 108535. [Google Scholar] [CrossRef]
- Berry, C.E.; Kendig, C.B.; An, N.; Fazilat, A.Z.; Churukian, A.A.; Griffin, M.; Pan, P.M.; Longaker, M.T.; Dixon, S.J.; Wan, D.C. Role of Ferroptosis in Radiation-Induced Soft Tissue Injury. Cell Death Discov. 2024, 10, 313. [Google Scholar] [CrossRef]
- Fijardo, M.; Kwan, J.Y.Y.; Bissey, P.-A.; Citrin, D.E.; Yip, K.W.; Liu, F.-F. The Clinical Manifestations and Molecular Pathogenesis of Radiation Fibrosis. EBioMedicine 2024, 103, 105089. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, M.R.; Shen, A.H.; Lee, G.K.; Momeni, A.; Longaker, M.T.; Wan, D.C. Radiation-Induced Skin Fibrosis: Pathogenesis, Current Treatment Options, and Emerging Therapeutics. Ann. Plast. Surg. 2019, 83, S59–S64. [Google Scholar] [CrossRef]
- Käsmann, L.; Dietrich, A.; Staab-Weijnitz, C.A.; Manapov, F.; Behr, J.; Rimner, A.; Jeremic, B.; Senan, S.; De Ruysscher, D.; Lauber, K.; et al. Radiation-Induced Lung Toxicity—Cellular and Molecular Mechanisms of Pathogenesis, Management, and Literature Review. Radiat. Oncol. 2020, 15, 214. [Google Scholar] [CrossRef]
- Wei, J.; Meng, L.; Hou, X.; Qu, C.; Wang, B.; Xin, Y.; Jiang, X. Radiation-Induced Skin Reactions: Mechanism and Treatment. Cancer Manag. Res. 2019, 11, 167–177. [Google Scholar] [CrossRef]
- Jarzebska, N.; Karetnikova, E.S.; Markov, A.G.; Kasper, M.; Rodionov, R.N.; Spieth, P.M. Scarred Lung. An Update on Radiation-Induced Pulmonary Fibrosis. Front. Med. 2020, 7, 585756. [Google Scholar] [CrossRef] [PubMed]
- Savin, I.A.; Zenkova, M.A.; Sen’kova, A.V. Pulmonary Fibrosis as a Result of Acute Lung Inflammation: Molecular Mechanisms, Relevant In Vivo Models, Prognostic and Therapeutic Approaches. Int. J. Mol. Sci. 2022, 23, 14959. [Google Scholar] [CrossRef]
- Guillamat-Prats, R. The Role of MSC in Wound Healing, Scarring and Regeneration. Cells 2021, 10, 1729. [Google Scholar] [CrossRef]
- Dos Santos, C.C. Advances in Mechanisms of Repair and Remodelling in Acute Lung Injury. Intensive Care Med. 2008, 34, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Blondonnet, R.; Constantin, J.-M.; Sapin, V.; Jabaudon, M. A Pathophysiologic Approach to Biomarkers in Acute Respiratory Distress Syndrome. Dis. Markers 2016, 2016, 3501373. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cone, J.E.; Brackbill, R.M.; Giesinger, I.; Yung, J.; Farfel, M.R. Pulmonary Fibrosis among World Trade Center Responders: Results from the WTC Health Registry Cohort. Int. J. Environ. Res. Public Health 2019, 16, 825. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.T.; Ventura, I.B.; Phillips, E.K.; Leahy, A.; Jablonski, R.; Montner, S.; Chung, J.H.; Vij, R.; Adegunsoye, A.; Strek, M.E. Interstitial Lung Disease in Firefighters: An Emerging Occupational Hazard. Front. Med. 2022, 9, 864658. [Google Scholar] [CrossRef]
- Muntyanu, A.; Milan, R.; Rahme, E.; LaChance, A.; Ouchene, L.; Cormier, M.; Litvinov, I.V.; Hudson, M.; Baron, M.; Netchiporouk, E.; et al. Exposure to Silica and Systemic Sclerosis: A Retrospective Cohort Study Based on the Canadian Scleroderma Research Group. Front. Med. 2022, 9, 984907. [Google Scholar] [CrossRef]
- Alahmari, H.; Ahmad, Z.; Johnson, S.R. Environmental Risks for Systemic Sclerosis. Rheum. Dis. Clin. N. Am. 2022, 48, 845–860. [Google Scholar] [CrossRef]
- Andersson, M.; Blanc, P.D.; Torén, K.; Järvholm, B. Smoking, Occupational Exposures, and Idiopathic Pulmonary Fibrosis among Swedish Construction Workers. Am. J. Ind. Med. 2021, 64, 251–257. [Google Scholar] [CrossRef]
- Ferri, C.; Arcangeletti, M.-C.; Caselli, E.; Zakrzewska, K.; Maccari, C.; Calderaro, A.; D’Accolti, M.; Soffritti, I.; Arvia, R.; Sighinolfi, G.; et al. Insights into the Knowledge of Complex Diseases: Environmental Infectious/Toxic Agents as Potential Etiopathogenetic Factors of Systemic Sclerosis. J. Autoimmun. 2021, 124, 102727. [Google Scholar] [CrossRef]
- Lazzaroni, M.-G.; Piantoni, S.; Angeli, F.; Bertocchi, S.; Franceschini, F.; Airò, P. A Narrative Review of Pathogenetic and Histopathologic Aspects, Epidemiology, Classification Systems, and Disease Outcome Measures in Systemic Sclerosis. Clin. Rev. Allergy Immunol. 2023, 64, 358–377. [Google Scholar] [CrossRef] [PubMed]
- Walecka, I.; Roszkiewicz, M.; Malewska, A. Potential Occupational and Environmental Factors in SSc Onset. Ann. Agric. Environ. Med. 2018, 25, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Kosarek, N.N.; Romano, M.E.; Moen, E.L.; Simms, R.W.; Erickson, A.; Khanna, D.; Pioli, P.A.; Whitfield, M.L. Geographic Clustering of Systemic Sclerosis in Areas of Environmental Pollution. Arthritis Care Res. 2025, 77, 855–866. [Google Scholar] [CrossRef]
- Glazer, C.S.; Newman, L.S. Occupational Interstitial Lung Disease. Clin. Chest Med. 2004, 25, 467–478. [Google Scholar] [CrossRef]
- Spagnolo, P.; Ryerson, C.J.; Guler, S.; Feary, J.; Churg, A.; Fontenot, A.P.; Piciucchi, S.; Udwadia, Z.; Corte, T.J.; Wuyts, W.A.; et al. Occupational Interstitial Lung Diseases. J. Intern. Med. 2023, 294, 798–815. [Google Scholar] [CrossRef]
- Taskar, V.S.; Coultas, D.B. Is Idiopathic Pulmonary Fibrosis an Environmental Disease? Proc. Am. Thorac. Soc. 2006, 3, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.A.; Min, B.; Fazio, J.C.; Johannson, K.A.; Steinmaus, C.; Reynolds, C.J.; Cummings, K.J. The Impact of Occupational Exposures on the Risk of Idiopathic Pulmonary Fibrosis: A Systematic Review and Meta-Analysis. Ann. Am. Thorac. Soc. 2024, 21, 486–498. [Google Scholar] [CrossRef] [PubMed]
- Rajnoveanu, A.-G.; Rajnoveanu, R.-M.; Motoc, N.S.; Postolache, P.; Gusetu, G.; Man, M.A. COPD in Firefighters: A Specific Event-Related Condition Rather than a Common Occupational Respiratory Disorder. Medicina 2022, 58, 239. [Google Scholar] [CrossRef]
- 9/11 First Responder: Toxic Dust Led to Cancer, Then Treatment Made It Worse. Available online: https://www.today.com/health/9-11-first-responder-diagnosed-cancer-then-pulmonary-fibrosis-t162167 (accessed on 15 July 2025).
- American Lung Association. Firefighters. Available online: https://www.lung.org/lung-health-diseases/lung-disease-lookup/occupational-lung-diseases/firefighters-lung-health (accessed on 15 July 2025).
- Leeming, D.J.; Genovese, F.; Sand, J.M.B.; Rasmussen, D.G.K.; Christiansen, C.; Jenkins, G.; Maher, T.M.; Vestbo, J.; Karsdal, M.A. Can Biomarkers of Extracellular Matrix Remodelling and Wound Healing Be Used to Identify High Risk Patients Infected with SARS-CoV-2?: Lessons Learned from Pulmonary Fibrosis. Respir. Res. 2021, 22, 38. [Google Scholar] [CrossRef] [PubMed]
- Ramli, I.; Cheriet, T.; Posadino, A.M.; Giordo, R.; Zayed, H.; Eid, A.H.; Pintus, G. Potential Therapeutic Targets of Resveratrol in the Prevention and Treatment of Pulmonary Fibrosis. Front. Biosci. (Landmark Ed.) 2023, 28, 198. [Google Scholar] [CrossRef]
- Trethewey, S.P.; Walters, G.I. The Role of Occupational and Environmental Exposures in the Pathogenesis of Idiopathic Pulmonary Fibrosis: A Narrative Literature Review. Medicina 2018, 54, 108. [Google Scholar] [CrossRef]
- Wick, G.; Grundtman, C.; Mayerl, C.; Wimpissinger, T.-F.; Feichtinger, J.; Zelger, B.; Sgonc, R.; Wolfram, D. The Immunology of Fibrosis. Annu. Rev. Immunol. 2013, 31, 107–135. [Google Scholar] [CrossRef] [PubMed]
- Paolocci, G.; Folletti, I.; Torén, K.; Ekström, M.; Dell’Omo, M.; Muzi, G.; Murgia, N. Occupational Risk Factors for Idiopathic Pulmonary Fibrosis in Southern Europe: A Case-Control Study. BMC Pulm. Med. 2018, 18, 75. [Google Scholar] [CrossRef]
- Mosconi, G.; Zanelli, R.; Migliori, M.; Seghizzi, P.; Michetti, G.; Nicoli, D.; Poma, M. Study of Lung Reactions in Six Asymptomatic Workers Occupationally Exposed to Hard Metal Dusts. Med. Lav. 1991, 82, 131–136. [Google Scholar]
- Oo, T.W.; Thandar, M.; Htun, Y.M.; Soe, P.P.; Lwin, T.Z.; Tun, K.M.; Han, Z.M. Assessment of Respiratory Dust Exposure and Lung Functions among Workers in Textile Mill (Thamine), Myanmar: A Cross-Sectional Study. BMC Public Health 2021, 21, 673. [Google Scholar] [CrossRef]
- Pauchet, A.; Chaussavoine, A.; Pairon, J.C.; Gabillon, C.; Didier, A.; Baldi, I.; Esquirol, Y. Idiopathic Pulmonary Fibrosis: What Do We Know about the Role of Occupational and Environmental Determinants? A Systematic Literature Review and Meta-Analysis. J. Toxicol. Environ. Health B Crit. Rev. 2022, 25, 372–392. [Google Scholar] [CrossRef] [PubMed]
- Bast, A.; Semen, K.O.; Drent, M. Pulmonary Toxicity Associated with Occupational and Environmental Exposure to Pesticides and Herbicides. Curr. Opin. Pulm. Med. 2021, 27, 278–283. [Google Scholar] [CrossRef]
- D’Amico, R.; Monaco, F.; Fusco, R.; Siracusa, R.; Impellizzeri, D.; Peritore, A.F.; Crupi, R.; Gugliandolo, E.; Cuzzocrea, S.; Di Paola, R.; et al. Atrazine Inhalation Worsen Pulmonary Fibrosis Regulating the Nuclear Factor-Erythroid 2-Related Factor (Nrf2) Pathways Inducing Brain Comorbidities. Cell Physiol. Biochem. 2021, 55, 704–725. [Google Scholar] [CrossRef] [PubMed]
- St Pierre, L.; Berhan, A.; Sung, E.K.; Alvarez, J.R.; Wang, H.; Ji, Y.; Liu, Y.; Yu, H.; Meier, A.; Afshar, K.; et al. Integrated Multiomic Analysis Identifies TRIP13 as a Mediator of Alveolar Epithelial Type II Cell Dysfunction in Idiopathic Pulmonary Fibrosis. Biochim. Biophys. Acta Mol. Basis Dis. 2025, 1871, 167572. [Google Scholar] [CrossRef]
- Sozeri, B.; Gulez, N.; Aksu, G.; Kutukculer, N.; Akalın, T.; Kandiloglu, G. Pesticide-Induced Scleroderma and Early Intensive Immunosuppressive Treatment. Arch. Environ. Occup. Health 2012, 67, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Madray, V.M.; Liles, J.E.; Davis, L.S. Chemical-Induced Sclerodermoid Disease Triggered by Pressure Washing Bleach Solution. JAAD Case Rep. 2020, 6, 1330–1332. [Google Scholar] [CrossRef]
- Rubio-Rivas, M.; Moreno, R.; Corbella, X. Occupational and Environmental Scleroderma. Systematic Review and Meta-Analysis. Clin. Rheumatol. 2017, 36, 569–582. [Google Scholar] [CrossRef] [PubMed]
- Darr, D.; Combs, S.; Murad, S.; Pinnell, S. Studies on the Inhibition of Collagen Synthesis in Fibroblasts Treated with Paraquat. Arch. Biochem. Biophys. 1993, 306, 267–271. [Google Scholar] [CrossRef]
- Zhou, Q.; Kan, B.; Jian, X.; Zhang, W.; Liu, H.; Zhang, Z. Paraquat Poisoning by Skin Absorption: Two Case Reports and a Literature Review. Exp. Ther. Med. 2013, 6, 1504–1506. [Google Scholar] [CrossRef]
- Papiris, S.A.; Maniati, M.A.; Kyriakidis, V.; Constantopoulos, S.H. Pulmonary Damage Due to Paraquat Poisoning through Skin Absorption. Respiration 1995, 62, 101–103. [Google Scholar] [CrossRef]
- Hadimioglu, N.; Dosemeci, L.; Arici, G.; Ramazanoglu, A. Systemic Organophosphate Poisoning Following the Percutaneous Injection of Insecticide. Case Report. Ski. Pharmacol. Appl. Ski. Physiol. 2002, 15, 195–199. [Google Scholar] [CrossRef]
- Majumder, S.; Chakraborty, A.K.; Bhattacharyya, A.; Mandal, T.K.; Basak, D.K. Effect of Short-Term Dermal Toxicity of Fenvalerate on Residue, Cell Architecture and Biochemical Profiles in Broiler Chicks. Indian J. Exp. Biol. 1997, 35, 162–167. [Google Scholar] [PubMed]
- Skibba, M.; Drelich, A.; Poellmann, M.; Hong, S.; Brasier, A.R. Nanoapproaches to Modifying Epigenetics of Epithelial Mesenchymal Transition for Treatment of Pulmonary Fibrosis. Front. Pharmacol. 2020, 11, 607689. [Google Scholar] [CrossRef]
- Distler, O.; Highland, K.B.; Gahlemann, M.; Azuma, A.; Fischer, A.; Mayes, M.D.; Raghu, G.; Sauter, W.; Girard, M.; Alves, M.; et al. Nintedanib for Systemic Sclerosis-Associated Interstitial Lung Disease. N. Engl. J. Med. 2019, 380, 2518–2528. [Google Scholar] [CrossRef] [PubMed]
- Maher, T.M.; Mayes, M.D.; Kreuter, M.; Volkmann, E.R.; Aringer, M.; Castellvi, I.; Cutolo, M.; Stock, C.; Schoof, N.; Alves, M.; et al. Effect of Nintedanib on Lung Function in Patients With Systemic Sclerosis-Associated Interstitial Lung Disease: Further Analyses of a Randomized, Double-Blind, Placebo-Controlled Trial. Arthritis Rheumatol. 2021, 73, 671–676. [Google Scholar] [CrossRef]
- Flaherty, K.R.; Wells, A.U.; Cottin, V.; Devaraj, A.; Walsh, S.L.F.; Inoue, Y.; Richeldi, L.; Kolb, M.; Tetzlaff, K.; Stowasser, S.; et al. Nintedanib in Progressive Fibrosing Interstitial Lung Diseases. N. Engl. J. Med. 2019, 381, 1718–1727. [Google Scholar] [CrossRef] [PubMed]
- Matteson, E.L.; Kelly, C.; Distler, J.H.W.; Hoffmann-Vold, A.-M.; Seibold, J.R.; Mittoo, S.; Dellaripa, P.F.; Aringer, M.; Pope, J.; Distler, O.; et al. Nintedanib in Patients With Autoimmune Disease-Related Progressive Fibrosing Interstitial Lung Diseases: Subgroup Analysis of the INBUILD Trial. Arthritis Rheumatol. 2022, 74, 1039–1047. [Google Scholar] [CrossRef]
- Wells, A.U.; Flaherty, K.R.; Brown, K.K.; Inoue, Y.; Devaraj, A.; Richeldi, L.; Moua, T.; Crestani, B.; Wuyts, W.A.; Stowasser, S.; et al. Nintedanib in Patients with Progressive Fibrosing Interstitial Lung Diseases-Subgroup Analyses by Interstitial Lung Disease Diagnosis in the INBUILD Trial: A Randomised, Double-Blind, Placebo-Controlled, Parallel-Group Trial. Lancet Respir. Med. 2020, 8, 453–460. [Google Scholar] [CrossRef]
- Huang, J.; Beyer, C.; Palumbo-Zerr, K.; Zhang, Y.; Ramming, A.; Distler, A.; Gelse, K.; Distler, O.; Schett, G.; Wollin, L.; et al. Nintedanib Inhibits Fibroblast Activation and Ameliorates Fibrosis in Preclinical Models of Systemic Sclerosis. Ann. Rheum. Dis. 2016, 75, 883–890. [Google Scholar] [CrossRef]
- Karampitsakos, T.; Sotiropoulou, V.; Katsaras, M.; Tzouvelekis, A. Improvement of Psoriatic Skin Lesions Following Pirfenidone Use in Patients with Fibrotic Lung Disease. BMJ Case Rep. 2023, 16, e252591. [Google Scholar] [CrossRef]
- Elias, J.A.; Zhu, Z.; Chupp, G.; Homer, R.J. Airway Remodeling in Asthma. J. Clin. Investig. 1999, 104, 1001–1006. [Google Scholar] [CrossRef]
- Holgate, S.T.; Polosa, R. The Mechanisms, Diagnosis, and Management of Severe Asthma in Adults. Lancet 2006, 368, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Cytokine Modulators as Novel Therapies for Asthma. Annu. Rev. Pharmacol. Toxicol. 2002, 42, 81–98. [Google Scholar] [CrossRef]
- Zimmermann, N.; Hershey, G.K.; Foster, P.S.; Rothenberg, M.E. Chemokines in Asthma: Cooperative Interaction between Chemokines and IL-13. J. Allergy Clin. Immunol. 2003, 111, 227–242; quiz 243. [Google Scholar] [CrossRef]
- Varricchi, G.; Brightling, C.E.; Grainge, C.; Lambrecht, B.N.; Chanez, P. Airway Remodelling in Asthma and the Epithelium: On the Edge of a New Era. Eur. Respir. J. 2024, 63, 2301619. [Google Scholar] [CrossRef]
- Wang, H.; Yip, K.H.; Keam, S.P.; Vlahos, R.; Nichol, K.; Wark, P.; Toubia, J.; Kral, A.C.; Cildir, G.; Pant, H.; et al. Dual Inhibition of Airway Inflammation and Fibrosis by Common β Cytokine Receptor Blockade. J. Allergy Clin. Immunol. 2024, 153, 672–683.e6. [Google Scholar] [CrossRef]
- Oh, M.-H.; Oh, S.Y.; Yu, J.; Myers, A.C.; Leonard, W.J.; Liu, Y.J.; Zhu, Z.; Zheng, T. IL-13 Induces Skin Fibrosis in Atopic Dermatitis by Thymic Stromal Lymphopoietin. J. Immunol. 2011, 186, 7232–7242. [Google Scholar] [CrossRef]
- Beck, L.A.; Cork, M.J.; Amagai, M.; De Benedetto, A.; Kabashima, K.; Hamilton, J.D.; Rossi, A.B. Type 2 Inflammation Contributes to Skin Barrier Dysfunction in Atopic Dermatitis. JID Innov. 2022, 2, 100131. [Google Scholar] [CrossRef]
- Facheris, P.; Jeffery, J.; Del Duca, E.; Guttman-Yassky, E. The Translational Revolution in Atopic Dermatitis: The Paradigm Shift from Pathogenesis to Treatment. Cell. Mol. Immunol. 2023, 20, 448–474. [Google Scholar] [CrossRef]
- Flora, A.; Jepsen, R.; Kozera, E.K.; Woods, J.A.; Cains, G.D.; Radzieta, M.; Jensen, S.O.; Malone, M.; Frew, J.W. Human Dermal Fibroblast Subpopulations and Epithelial Mesenchymal Transition Signals in Hidradenitis Suppurativa Tunnels Are Normalized by Spleen Tyrosine Kinase Antagonism In Vivo. PLoS ONE 2023, 18, e0282763. [Google Scholar] [CrossRef]
- Krueger, J.G.; Frew, J.; Jemec, G.B.E.; Kimball, A.B.; Kirby, B.; Bechara, F.G.; Navrazhina, K.; Prens, E.; Reich, K.; Cullen, E.; et al. Hidradenitis Suppurativa: New Insights into Disease Mechanisms and an Evolving Treatment Landscape. Br. J. Dermatol. 2024, 190, 149–162. [Google Scholar] [CrossRef]
- Sabat, R.; Alavi, A.; Wolk, K.; Wortsman, X.; McGrath, B.; Garg, A.; Szepietowski, J.C. Hidradenitis Suppurativa. Lancet 2025, 405, 420–438. [Google Scholar] [CrossRef]
- Ashley, S.L.; Wilke, C.A.; Kim, K.K.; Moore, B.B. Periostin Regulates Fibrocyte Function to Promote Myofibroblast Differentiation and Lung Fibrosis. Mucosal Immunol. 2017, 10, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Naik, P.K.; Bozyk, P.D.; Bentley, J.K.; Popova, A.P.; Birch, C.M.; Wilke, C.A.; Fry, C.D.; White, E.S.; Sisson, T.H.; Tayob, N.; et al. Periostin Promotes Fibrosis and Predicts Progression in Patients with Idiopathic Pulmonary Fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2012, 303, L1046–L1056. [Google Scholar] [CrossRef]
- Okamoto, M.; Fujimoto, K.; Johkoh, T.; Kawaguchi, A.; Mukae, H.; Sakamoto, N.; Ogura, T.; Ikeda, S.; Kondoh, Y.; Yamano, Y.; et al. A Prospective Cohort Study of Periostin as a Serum Biomarker in Patients with Idiopathic Pulmonary Fibrosis Treated with Nintedanib. Sci. Rep. 2023, 13, 22977. [Google Scholar] [CrossRef] [PubMed]
- Makino, K.; Makino, T.; Stawski, L.; Mantero, J.C.; Lafyatis, R.; Simms, R.; Trojanowska, M. Blockade of PDGF Receptors by Crenolanib Has Therapeutic Effect in Patient Fibroblasts and in Preclinical Models of Systemic Sclerosis. J. Investig. Dermatol. 2017, 137, 1671–1681. [Google Scholar] [CrossRef]
- van Straalen, K.R.; Ma, F.; Tsou, P.-S.; Plazyo, O.; Gharaee-Kermani, M.; Calbet, M.; Xing, X.; Sarkar, M.K.; Uppala, R.; Harms, P.W.; et al. Single-Cell Sequencing Reveals Hippo Signaling as a Driver of Fibrosis in Hidradenitis Suppurativa. J. Clin. Investig. 2024, 134, e169225. [Google Scholar] [CrossRef]
- Gasparini, G.; Cozzani, E.; Parodi, A. Interleukin-4 and Interleukin-13 as Possible Therapeutic Targets in Systemic Sclerosis. Cytokine 2020, 125, 154799. [Google Scholar] [CrossRef] [PubMed]
- Fang, D.; Chen, B.; Lescoat, A.; Khanna, D.; Mu, R. Immune Cell Dysregulation as a Mediator of Fibrosis in Systemic Sclerosis. Nat. Rev. Rheumatol. 2022, 18, 683–693. [Google Scholar] [CrossRef]
- Nakashima, C.; Ishida, Y.; Kaku, Y.; Epstein, E.H.; Otsuka, A.; Kabashima, K. Dupilumab Improved Atypical Fibrotic Skin Plaques in Atopic Dermatitis. Br. J. Dermatol. 2020, 182, 487–488. [Google Scholar] [CrossRef]
- Thomson, N.C.; Patel, M.; Smith, A.D. Lebrikizumab in the Personalized Management of Asthma. Biologics 2012, 6, 329–335. [Google Scholar] [CrossRef]
- Gyawali, B.; Georas, S.N.; Khurana, S. Biologics in Severe Asthma: A State-of-the-Art Review. Eur. Respir. Rev. 2025, 34, 240088. [Google Scholar] [CrossRef]
- Văruț, R.M.; Dalia, D.; Radivojevic, K.; Trasca, D.M.; Stoica, G.-A.; Adrian, N.S.; Carmen, N.E.; Singer, C.E. Targeted Biologic Therapies in Severe Asthma: Mechanisms, Biomarkers, and Clinical Applications. Pharmaceuticals 2025, 18, 1021. [Google Scholar] [CrossRef]
- Gatsounia, A.; Schinas, G.; Danielides, G.; Grafanaki, K.; Mastronikolis, N.; Stathopoulos, C.; Lygeros, S. Epigenetic Mechanisms in CRSwNP: The Role of MicroRNAs as Potential Biomarkers and Therapeutic Targets. Curr. Issues Mol. Biol. 2025, 47, 114. [Google Scholar] [CrossRef]
- Danielides, G.; Lygeros, S.; Kyriakopoulos, G.; Tsapardoni, F.; Grafanaki, K.; Stathopoulos, C.; Naxakis, S.; Danielides, V. Serum and Tissue Periostin Expression in Rhinosinusitis with Nasal Polyps. Am. J. Rhinol. Allergy 2025, 39, 259–265. [Google Scholar] [CrossRef]
- Muñoz-Bellido, F.; Moreno, E.; Dávila, I. Dupilumab: A Review of Present Indications and Off-Label Uses. J. Investig. Allergy Clin. Immunol. 2022, 32, 97–115. [Google Scholar] [CrossRef]
- Guttman-Yassky, E.; Bissonnette, R.; Ungar, B.; Suárez-Fariñas, M.; Ardeleanu, M.; Esaki, H.; Suprun, M.; Estrada, Y.; Xu, H.; Peng, X.; et al. Dupilumab Progressively Improves Systemic and Cutaneous Abnormalities in Patients with Atopic Dermatitis. J. Allergy Clin. Immunol. 2019, 143, 155–172. [Google Scholar] [CrossRef]
- Olbrich, H.; Sadik, C.D.; Ludwig, R.J.; Thaçi, D.; Boch, K. Dupilumab in Inflammatory Skin Diseases: A Systematic Review. Biomolecules 2023, 13, 634. [Google Scholar] [CrossRef] [PubMed]
- Guttman-Yassky, E.; Katoh, N.; J Cork, M.; Jagdeo, J.; F Alexis, A.; Chen, Z.; A Levit, N.; B Rossi, A. Dupilumab Treatment Improves Lichenification in Atopic Dermatitis in Different Age and Racial Groups. J. Drugs Dermatol. 2025, 24, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Diaz, A.; Tan, K.; He, H.; Xu, H.; Cueto, I.; Pavel, A.B.; Krueger, J.G.; Guttman-Yassky, E. Keloid Lesions Show Increased IL-4/IL-13 Signaling and Respond to Th2-Targeting Dupilumab Therapy. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e161–e164. [Google Scholar] [CrossRef]
- Bitterman, D.; Patel, P.; Wang, J.Y.; Kabakova, M.; Zafar, K.; Lee, A.; Gollogly, J.M.; Cohen, M.; Austin, E.; Jagdeo, J. Systematic Review of Dupilumab Safety and Efficacy for Treatment of Keloid Scars. Arch. Dermatol. Res. 2024, 316, 560. [Google Scholar] [CrossRef]
- M Walsh, G. Dupilumab Utility in Difficult-to-Treat Asthma. Immunotherapy 2019, 11, 261–264. [Google Scholar] [CrossRef]
- Young, J.; Spisany, T.; Guidry, C.M.; Hong, J.; Le, J.; El Rassi, E.; Boylan, P.M. Dupilumab for Chronic Obstructive Pulmonary Disease: A Systematic Review. Biologics 2025, 5, 5. [Google Scholar] [CrossRef]
- Wilken, B.F.; Moran-Mendoza, O.; Pourafkari, M.; Asai, Y. Drug-Induced Interstitial Lung Disease Associated with Dupilumab for the Treatment of Atopic Dermatitis. JAAD Case Rep. 2024, 50, 12–15. [Google Scholar] [CrossRef]
- Simpson, E.L.; Bieber, T.; Guttman-Yassky, E.; Beck, L.A.; Blauvelt, A.; Cork, M.J.; Silverberg, J.I.; Deleuran, M.; Kataoka, Y.; Lacour, J.-P.; et al. Two Phase 3 Trials of Dupilumab versus Placebo in Atopic Dermatitis. N. Engl. J. Med. 2016, 375, 2335–2348. [Google Scholar] [CrossRef]
- Bhatt, S.P.; Rabe, K.F.; Hanania, N.A.; Vogelmeier, C.F.; Bafadhel, M.; Christenson, S.A.; Papi, A.; Singh, D.; Laws, E.; Dakin, P.; et al. Dupilumab for Chronic Obstructive Pulmonary Disease with Type 2 Inflammation: A Pooled Analysis of Two Phase 3, Randomised, Double-Blind, Placebo-Controlled Trials. Lancet Respir. Med. 2025, 13, 234–243. [Google Scholar] [CrossRef]
- Sener, S.; Sener, Y.Z.; Batu, E.D.; Sari, A.; Akdogan, A. A Systematic Literature Review of Janus Kinase Inhibitors for the Treatment of Systemic Sclerosis. J. Scleroderma Relat. Disord. 2025; epub ahead of print. [Google Scholar] [CrossRef]
- Talotta, R. The Rationale for Targeting the JAK/STAT Pathway in Scleroderma-Associated Interstitial Lung Disease. Immunotherapy 2021, 13, 241–256. [Google Scholar] [CrossRef]
- Yang, Z.; Li, Z.; Liu, Z.; Li, W.; Jiao, R.; Liu, Y.; Chen, R.; Shi, Y.; Zhang, T.; Liu, J.; et al. Ruxolitinib Attenuates Bleomycin-Induced Pulmonary Fibrosis in Mice by Modulating Macrophage Polarization through the JAK/STAT Signaling Pathway. Int. Immunopharmacol. 2025, 161, 114962. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Wang, W.; Yin, P.; Gao, J.; Na, L.; Sun, Y.; Wang, Z.; Zhang, Z.; Zhao, C. Ruxolitinib Alleviates Renal Interstitial Fibrosis in UUO Mice. Int. J. Biol. Sci. 2020, 16, 194–203. [Google Scholar] [CrossRef]
- Lescoat, A.; Lelong, M.; Jeljeli, M.; Piquet-Pellorce, C.; Morzadec, C.; Ballerie, A.; Jouneau, S.; Jego, P.; Vernhet, L.; Batteux, F.; et al. Combined Anti-Fibrotic and Anti-Inflammatory Properties of JAK-Inhibitors on Macrophages in Vitro and in Vivo: Perspectives for Scleroderma-Associated Interstitial Lung Disease. Biochem. Pharmacol. 2020, 178, 114103. [Google Scholar] [CrossRef]
- Bieber, T.; Thyssen, J.P.; Reich, K.; Simpson, E.L.; Katoh, N.; Torrelo, A.; De Bruin-Weller, M.; Thaci, D.; Bissonnette, R.; Gooderham, M.; et al. Pooled Safety Analysis of Baricitinib in Adult Patients with Atopic Dermatitis from 8 Randomized Clinical Trials. J. Eur. Acad. Dermatol. Venereol. 2021, 35, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Zhang, H.; Chan, L.S. The Involvement of the JAK-STAT Signaling Pathway in Chronic Inflammatory Skin Disease Atopic Dermatitis. JAK-STAT 2013, 2, e24137. [Google Scholar] [CrossRef] [PubMed]
- Guttman-Yassky, E.; Silverberg, J.I.; Nemoto, O.; Forman, S.B.; Wilke, A.; Prescilla, R.; de la Peña, A.; Nunes, F.P.; Janes, J.; Gamalo, M.; et al. Baricitinib in Adult Patients with Moderate-to-Severe Atopic Dermatitis: A Phase 2 Parallel, Double-Blinded, Randomized Placebo-Controlled Multiple-Dose Study. J. Am. Acad. Dermatol. 2019, 80, 913–921.e9. [Google Scholar] [CrossRef]
- Pan, J.; Li, D.; Xu, Y.; Zhang, J.; Wang, Y.; Chen, M.; Lin, S.; Huang, L.; Chung, E.J.; Citrin, D.E.; et al. Inhibition of Bcl-2/Xl With ABT-263 Selectively Kills Senescent Type II Pneumocytes and Reverses Persistent Pulmonary Fibrosis Induced by Ionizing Radiation in Mice. Int. J. Radiat. Oncol. Biol. Phys. 2017, 99, 353–361. [Google Scholar] [CrossRef]
- Kuehl, T.; Lagares, D. BH3 Mimetics as Anti-Fibrotic Therapy: Unleashing the Mitochondrial Pathway of Apoptosis in Myofibroblasts. Matrix Biol. 2018, 68–69, 94–105. [Google Scholar] [CrossRef]
- Mohamad Anuar, N.N.; Nor Hisam, N.S.; Liew, S.L.; Ugusman, A. Clinical Review: Navitoclax as a Pro-Apoptotic and Anti-Fibrotic Agent. Front. Pharmacol. 2020, 11, 564108. [Google Scholar] [CrossRef]
- Ebata, S.; Yoshizaki, A.; Oba, K.; Kashiwabara, K.; Ueda, K.; Uemura, Y.; Watadani, T.; Fukasawa, T.; Miura, S.; Yoshizaki-Ogawa, A.; et al. Safety and Efficacy of Rituximab in Systemic Sclerosis (DESIRES): Open-Label Extension of a Double-Blind, Investigators-Initiated, Randomised, Placebo-Controlled Trial. Lancet Rheumatol. 2022, 4, e546–e555. [Google Scholar] [CrossRef] [PubMed]
- Kuzumi, A.; Ebata, S.; Fukasawa, T.; Matsuda, K.M.; Kotani, H.; Yoshizaki-Ogawa, A.; Sato, S.; Yoshizaki, A. Long-Term Outcomes After Rituximab Treatment for Patients with Systemic Sclerosis: Follow-up of the DESIRES Trial with a Focus on Serum Immunoglobulin Levels. JAMA Dermatol. 2023, 159, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Spino, C.; Johnson, S.; Chung, L.; Whitfield, M.L.; Denton, C.P.; Berrocal, V.; Franks, J.; Mehta, B.; Molitor, J.; et al. Abatacept in Early Diffuse Cutaneous Systemic Sclerosis: Results of a Phase II Investigator-Initiated, Multicenter, Double-Blind, Randomized, Placebo-Controlled Trial. Arthritis Rheumatol. 2020, 72, 125–136. [Google Scholar] [CrossRef]
- Chen, L.; Huang, R.; Huang, C.; Nong, G.; Mo, Y.; Ye, L.; Lin, K.; Chen, A. Cell Therapy for Scleroderma: Progress in Mesenchymal Stem Cells and CAR-T Treatment. Front. Med. 2024, 11, 1530887. [Google Scholar] [CrossRef] [PubMed]
- Clatworthy, M.R. Targeting B Cells and Antibody in Transplantation. Am. J. Transplant. 2011, 11, 1359–1367. [Google Scholar] [CrossRef]
- Singer, N.G.; Caplan, A.I. Mesenchymal Stem Cells: Mechanisms of Inflammation. Annu. Rev. Pathol. 2011, 6, 457–478. [Google Scholar] [CrossRef]
- Zhao, K.; Kong, C.; Shi, N.; Jiang, J.; Li, P. Potential Angiogenic, Immunomodulatory, and Antifibrotic Effects of Mesenchymal Stem Cell-Derived Extracellular Vesicles in Systemic Sclerosis. Front. Immunol. 2023, 14, 1125257. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, S.; Tsuchiya, A.; Iwasawa, T.; Nojiri, S.; Watanabe, T.; Ogawa, M.; Yoshida, T.; Fujiki, K.; Koui, Y.; Kido, T.; et al. Small Extracellular Vesicles Derived from Interferon-γ Pre-Conditioned Mesenchymal Stromal Cells Effectively Treat Liver Fibrosis. npj Regen. Med. 2021, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Wu, Y.; Zhang, B.; Xiong, M.; Yi, Y.; Zhang, Q.; Wu, M. Exosomes Derived from E2F1−/− Adipose-Derived Stem Cells Promote Skin Wound Healing via miR-130b-5p/TGFBR3 Axis. Int. J. Nanomed. 2023, 18, 6275–6292. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Chen, S.; Meng, T.; Liu, L.; Li, L.; Xiang, R.; Zhang, H.; Zhu, Y.; Zhang, X.; Lin, S.; et al. Extracellular Vesicles as Precision Therapeutic Vectors: Charting the Future of Cell-Targeted Therapies. Precis. Med. Eng. 2025, 2, 100031. [Google Scholar] [CrossRef]
- Yu, H.; Liu, S.; Wang, S.; Gu, X. A Narrative Review of the Role of HDAC6 in Idiopathic Pulmonary Fibrosis. J. Thorac. Dis. 2024, 16, 688–695. [Google Scholar] [CrossRef]
- Zheng, L.; Zhu, X.; Du, J.; Liu, Y. The Role of HDAC6 in Fibrosis: A Novel and Effective Therapy Strategy. Eur. J. Med. Res. 2025, 30, 571. [Google Scholar] [CrossRef]
- Borrello, M.T.; Ruzic, D.; Paish, H.; Graham, E.; Collins, A.L.; Scott, R.; Higginbotham, S.; Radovic, B.; Nelson, G.; Bulmer, D.; et al. Pharmacological Manipulation of Liver Fibrosis Progression Using Novel HDAC6 Inhibitors. FEBS J. 2025, 292, 3397–3411. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wu, Y.-X.; Yu, X.-Q.; Zhang, B.-Y.; Ma, L.-Y. EZH2 Serves as a Promising Therapeutic Target for Fibrosis. Bioorg. Chem. 2023, 137, 106578. [Google Scholar] [CrossRef]
- Tsou, P.-S.; Campbell, P.; Amin, M.A.; Coit, P.; Miller, S.; Fox, D.A.; Khanna, D.; Sawalha, A.H. Inhibition of EZH2 Prevents Fibrosis and Restores Normal Angiogenesis in Scleroderma. Proc. Natl. Acad. Sci. USA 2019, 116, 3695–3702. [Google Scholar] [CrossRef]
- Malhotra, S.; Villar, L.M.; Costa, C.; Midaglia, L.; Cubedo, M.; Medina, S.; Fissolo, N.; Río, J.; Castilló, J.; Álvarez-Cermeño, J.C.; et al. Circulating EZH2-Positive T Cells Are Decreased in Multiple Sclerosis Patients. J. Neuroinflamm. 2018, 15, 296. [Google Scholar] [CrossRef]
- Lhuissier, E.; Aury-Landas, J.; Bouet, V.; Bazille, C.; Repesse, Y.; Freret, T.; Boumédiene, K.; Baugé, C. Evaluation of the Impact of S-Adenosylmethionine-Dependent Methyltransferase Inhibitor, 3-Deazaneplanocin A, on Tissue Injury and Cognitive Function in Mice. Oncotarget 2018, 9, 20698–20708. [Google Scholar] [CrossRef] [PubMed]
- Moraitis, S.; Piperi, C. Multi-Faceted Role of Histone Methyltransferase Enhancer of Zeste 2 (EZH2) in Neuroinflammation and Emerging Targeting Options. Biology 2025, 14, 749. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, X.; Hu, X.; Duan, X.; Wan, G.; Li, L.; Feng, Q.; Zhang, Y.; Wang, N.; Yu, L. Covalent Inhibitors of EZH2: Design, Synthesis and Evaluation. Biomed. Pharmacother. 2022, 147, 112617. [Google Scholar] [CrossRef]
- Vecin, N.; Balukoff, N.C.; Yaghi, M.; Gonzalez, T.; Sawaya, A.P.; Strbo, N.; Tomic-Canic, M.; Lev-Tov, H.; Pastar, I. Hidradenitis Suppurativa Tunnels: Unveiling a Unique Disease Entity. JID Innov. 2025, 5, 100350. [Google Scholar] [CrossRef]
- Tzellos, T.; Zouboulis, C.C. Presence of Draining Tunnels Denotes a Distinct Hidradenitis Suppurativa Phenotype. J. Eur. Acad. Dermatol. Venereol. 2025, 39, 1372–1373. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Gao, X.; Jia, R.; Sun, Y.; Ding, Y.; Wang, F.; Wang, Y. Astragaloside IV Inhibits Idiopathic Pulmonary Fibrosis through Activation of Autophagy by miR-21-Mediated PTEN/PI3K/AKT/mTOR Pathway. Cell. Mol. Biol. 2024, 70, 128–136. [Google Scholar] [CrossRef] [PubMed]
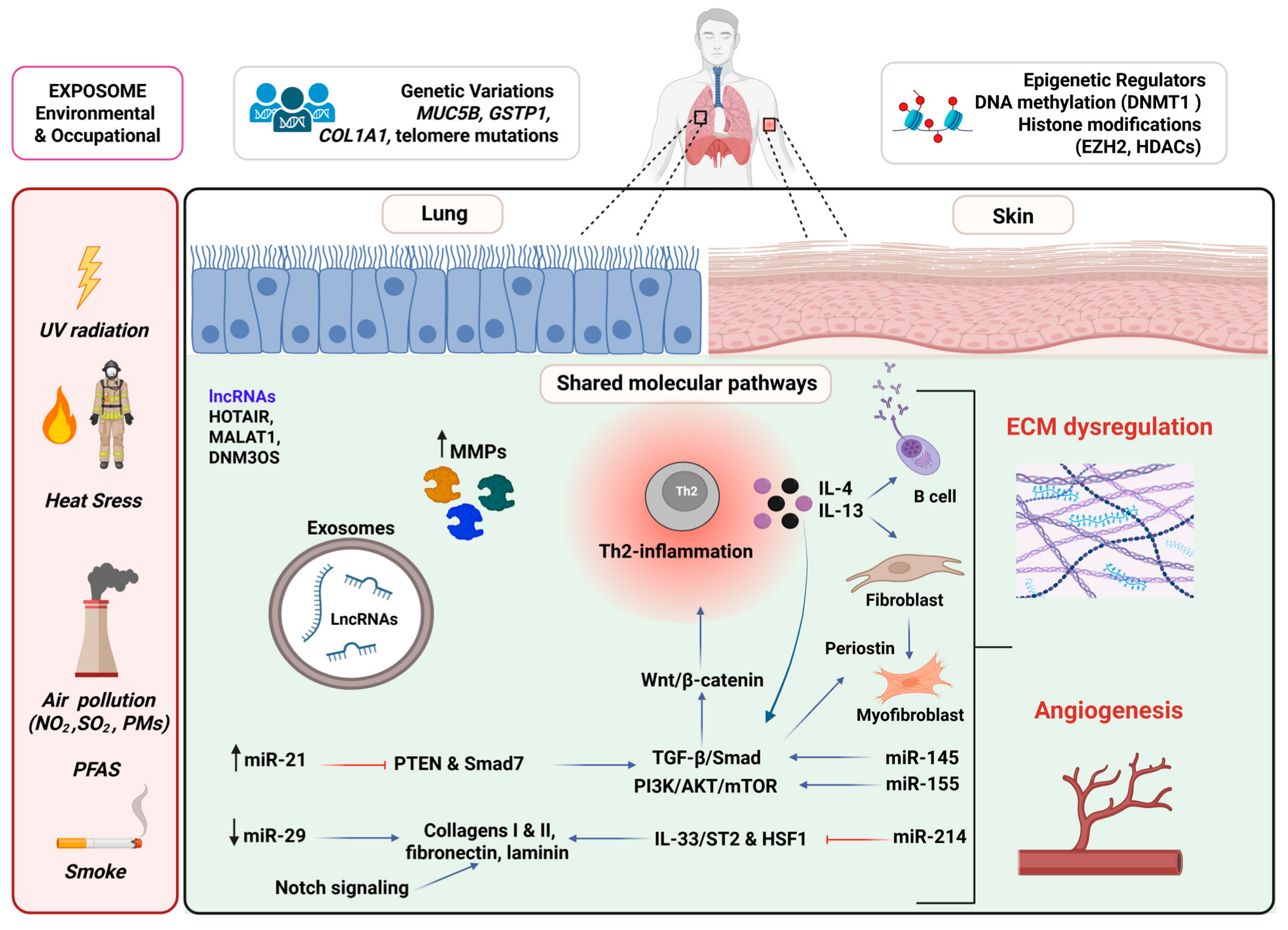
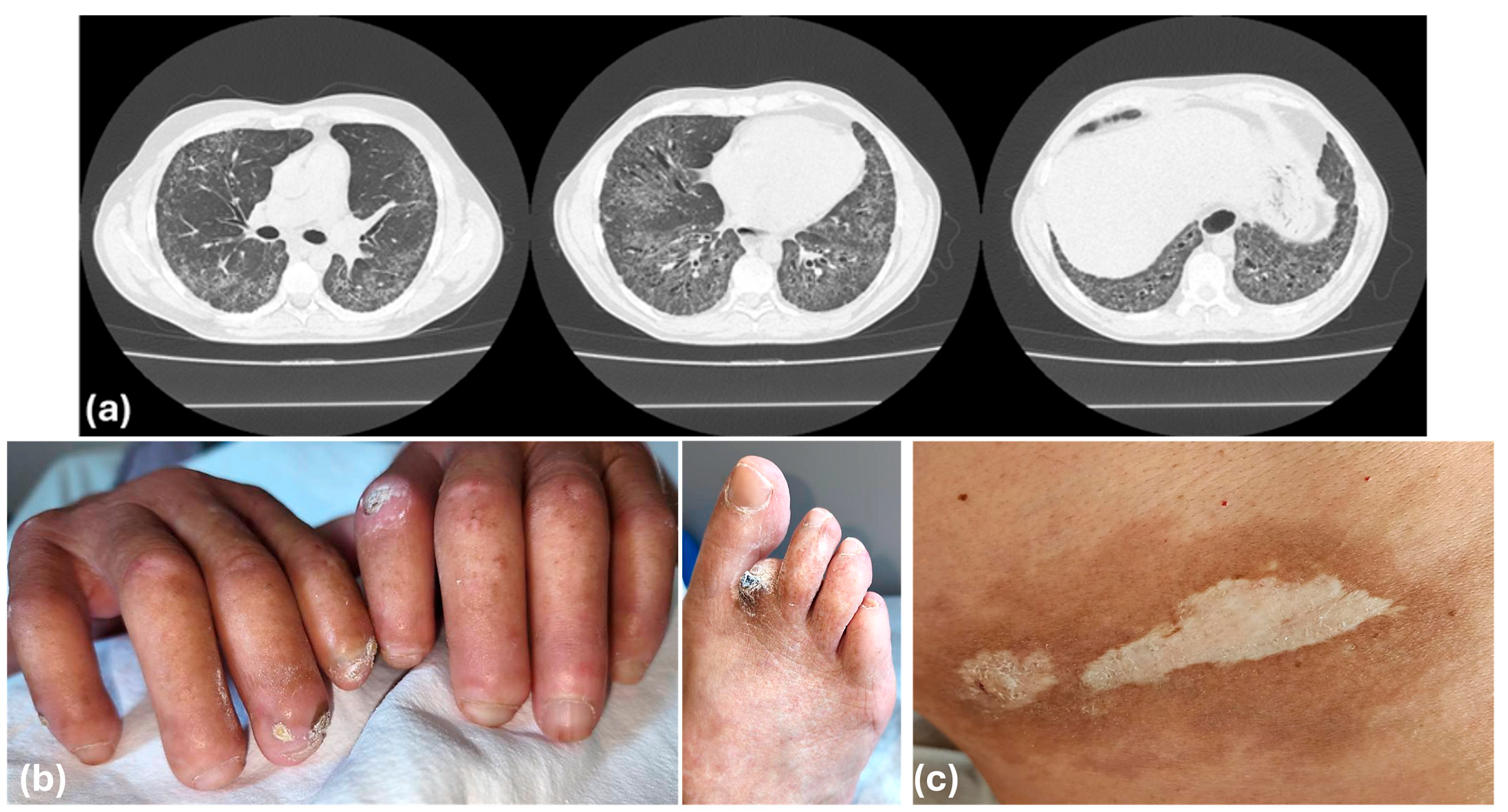
| Strategy/Agent | Mechanistic Target | Fibrotic Pathway Modulation | Skin Relevance | Lung Relevance | Translational Notes |
|---|---|---|---|---|---|
| Pirfenidone | TGF-β, oxidative stress, fibroblast proliferation | Inhibits TGF-β signaling and ROS-mediated ECM accumulation | Investigated in SSc, keloids, hypertrophic scars | Approved for IPF, reduces FVC decline | SSc-ILD trials ongoing; dermal efficacy remains modest |
| Nintedanib | PDGFRα/β | Attenuates fibroblast activation and ECM synthesis | Effective in patient-derived fibroblasts from SSc | Targeting lung fibroblast heterogeneity in preclinical models | Cross-organ translational potential; periostin/PDGF crosstalk |
| Anti-IL-4/IL-13 Biologics (e.g., Dupilumab, Lebrikizumab) | IL-4Rα/JAK–STAT6 axis | Reduces type 2 inflammation, fibroblast activation, and periostin expression | Shown to improve lichenified AD and chronic HS remodeling | Approved for asthma/CRSwNP; reduces airway fibrosis | Trials in systemic fibrosis and lung-AD spectrum diseases |
| PDGFRiInhibitors (e.g., Crenolanib | PDGFRα/β | Attenuates fibroblast activation and ECM synthesis | Effective in patient-derived fibroblasts from SSc | Targeting lung fibroblast heterogeneity in preclinical models | Cross-organ translational potential; periostin/PDGF crosstalk |
| Periostin-targeted therapies | Periostin–integrin axis | Blocks fibroblast migration, ECM stiffening, and TGF-β feedback | HS, keloids, SSc plaques with high periostin expression | Elevated serum periostin in IPF; biomarker of progression | Validated biomarker and emerging therapeutic target |
| RNA-based Therapies (e.g., anti–miR-21, miR-29 mimics) | miRNAs (miR-21 ↑, miR-29 ↓) | Restores antifibrotic miRNA balance; targets TGF-β and ECM genes | Remlarsen (miR-29) for keloids/SSc; anti-miR-21 reduces dermal scarring | Anti-miR-21 attenuates IPF in vivo | Organ-shared regulatory RNAs enable dual indications |
| Epigenetic modulators (EZH2, HDAC inhibitors) | Chromatin modifiers, lncRNA–miRNA axis | Silences profibrotic transcription; reprograms fibroblast phenotype | HDAC inhibitors reduce collagen in keloid/SSc fibroblasts | EZH2 promotes EMT, ECM accumulation in IPF | Precision-targeted and cell-specific epigenetic therapies emerging |
| Senolytic Agents (e.g., Navitoclax, Fisetin) | Senescent fibroblasts, SASP | Induces apoptosis of fibrotic fibroblasts; reduces SASP cytokines | Reverses fibroblast persistence in keloid models; aging skin fibrosis | Preclinical efficacy in IPF and radiation-induced lung injury | Targets inflammaging and chronic remodeling loops |
| Extracellular Vesicle (EV) RNA Delivery | Organ-specific miRNA/lncRNA payloads | Delivers antifibrotic RNA cargo (e.g., miR-29 or miR-148a-3p) to fibroblasts | Experimental in dermal fibrosis, AD, burn injury | MSC-EVs mitigate lung fibrosis via Wnt, TGF-β suppression | Platform for precision, low-toxicity, cross-organ applications |
| JAK Inhibitors (e.g., Ruxolitinib) | JAK1/JAK2; downstream of IL-4/IL-13 | Interrupts immune–fibrotic signaling (STAT6, IL-6, or TGF-β) | Efficacy in SSc, AD with fibrotic plaques | Investigational in fibrosing ILDs, SSc-ILD | Suitable for combined inflammatory–fibrotic phenotypes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grafanaki, K.; Maniatis, A.; Sotiropoulou, V.; Pasmatzi, E.; Tzouvelekis, A. Fibrotic Disease of the Skin and Lung: Shared Pathways, Environmental Drivers, and Therapeutic Opportunities in a Changing Climate. Int. J. Mol. Sci. 2025, 26, 8394. https://doi.org/10.3390/ijms26178394
Grafanaki K, Maniatis A, Sotiropoulou V, Pasmatzi E, Tzouvelekis A. Fibrotic Disease of the Skin and Lung: Shared Pathways, Environmental Drivers, and Therapeutic Opportunities in a Changing Climate. International Journal of Molecular Sciences. 2025; 26(17):8394. https://doi.org/10.3390/ijms26178394
Chicago/Turabian StyleGrafanaki, Katerina, Alexandros Maniatis, Vasilina Sotiropoulou, Efstathia Pasmatzi, and Argyris Tzouvelekis. 2025. "Fibrotic Disease of the Skin and Lung: Shared Pathways, Environmental Drivers, and Therapeutic Opportunities in a Changing Climate" International Journal of Molecular Sciences 26, no. 17: 8394. https://doi.org/10.3390/ijms26178394
APA StyleGrafanaki, K., Maniatis, A., Sotiropoulou, V., Pasmatzi, E., & Tzouvelekis, A. (2025). Fibrotic Disease of the Skin and Lung: Shared Pathways, Environmental Drivers, and Therapeutic Opportunities in a Changing Climate. International Journal of Molecular Sciences, 26(17), 8394. https://doi.org/10.3390/ijms26178394







