Abstract
Frailty is a clinical condition closely related to aging which is characterized by a multidimensional decline in biological reserves, a failure of physiological mechanisms and vulnerability to minor stressors. Chronic inflammation, the impairment of endothelial function, age-related endocrine system modifications and immunosenescence are important mechanisms in the pathophysiology of frailty. Endothelial progenitor cells (EPCs) are considered important contributors of the endothelium homeostasis and turn-over. In the elderly, EPCs are impaired in terms of function, number and survival. In addition, the modification of EPCs’ level and function has been widely demonstrated in atherosclerosis, hypertension and diabetes mellitus, which are the most common age-related diseases. The purpose of this review is to illustrate the role of EPCs in frailty. Initially, we describe the endothelial dysfunction in frailty, the response of EPCs to the endothelial dysfunction associated with frailty and, finally, interventions which may restore the EPCs expression and function in frail people.
1. Introduction
Population aging is a global phenomenon, and the number of persons aged 65 years or over reached 727 million in 2020 [1]. By 2050, the number of older persons is expected to double, and the proportion of the elderly is projected to increase by up to 16% such that one in six persons will be over the age of 65 [2].
Frailty is a clinical condition closely related to ageing which is characterized by a multidimensional decline in biological reserves, a failure of physiological mechanisms and vulnerability to minor stressors [3,4]. Frailty is associated with an increased risk of adverse outcomes including disability, falls, delirium and mortality [5,6,7,8]. The clinical presentation of frailty is characterized by fatigue, unexplained weight loss, frequent infections, balance and gait impairment, impaired awareness and fluctuating disability. Demographic and social factors, physical performance, impaired cognition, multiple chronic diseases and malnutrition are considered crucial risk factors for the onset and progression of frailty [9].
Chronic inflammation, the impairment of endothelial function, the age-related modification of the endocrine system and immunosenescence cross-linked to genetic and environmental factors are important mechanisms in the pathophysiology of frailty. Several inflammatory cytokines including IL-6, tumor necrosis factor-α (TNFα) and the acute phase protein C-reactive protein (CRP) have been independently associated with frailty [10]. Inflammation is associated with the catabolism of skeletal muscles and a reduction in muscle strength, which characterize frailty [11]. The alteration of insulin growth factor (IGF-1) signaling, the reduction in testosterone and estradiol levels and the modification of cortisol release influence the development of vulnerability and frailty [12]. Furthermore, the blunting of the B-cell-controlled antibody response, changes in T-lymphocyte production, the impaired activity of macrophages and a reduction in the number of stem cells respond inappropriately to inflammation, injury and endothelial damage [13].
Endothelium represents a functional barrier between tissues and circulating blood, prevents platelet and leukocyte aggregation and adhesion and produces a variety of vasoregulation factors such as nitric oxide (NO) and endothelins [14]. Alteration in the endothelial cells (ECs) damage/repair balance leads to vascular remodeling and the impairment of NO availability and is a key element in endothelial dysfunction and the atherosclerosis process [14,15]. Atherosclerosis is considered an age-dependent disease and leads to the development of cardiovascular comorbidities [15]. Furthermore, endothelial dysfunction has been suggested as an early predictor of frailty phenotype onset [16].
Endothelial progenitor cells (EPCs) are considered important contributors in the endothelium homeostasis and turn-over. They replace dysfunctional ECs, facilitate re-endothelization and influence the process of age-related vascular remodeling [17]. In the elderly, EPCs are impaired in terms of function, number and survival, and the modification of the circulating EPCs level in relation to physical frailty has been reported [18]. In addition, the modification of the EPCs level and function has been widely demonstrated in atherosclerosis, hypertension, diabetes mellitus, rheumatic disorders and dementia, which are the most common age-related diseases [19,20,21,22,23].
Considering the age-related modification of EPCs, the effect of multiple comorbidities and chronic conditions on EPCs and endothelial dysfunction, EPCs may have an important role in the underlying mechanisms of frailty. In addition, the negative impact of frailty is also characterized by a low quality of life for older people, the failure of the therapeutic management of chronic conditions and increased healthcare costs. Interventions guided by comprehensive geriatric assessments (CGA), which include nutritional support, physical activity and the revision of drug therapy, are beneficial for frail people, but no specific therapy for frailty exists. The purpose of this review is to illustrate the role of EPCs in frailty. Initially, we describe the endothelial dysfunction in frailty, the response of EPCs to the endothelial dysfunction associated with frailty and, finally, interventions that may restore the EPCs expression and function in frail people.
1.1. Main Characteristics of EPCs
EPCs are a heterogeneous group of cells of different origins and in different stages of maturation which contribute to endothelial regeneration and vascular repair [24,25]. The mobilization of EPCs from bone marrow to circulation is controlled by a variety of angiogenic factors, including endothelial NO synthetase (eNOS) and vascular endothelial growth factor (VEGF), which play an important role by enhancing the growth of EPCs, tube formation and the angiogenesis process [26,27].
Based on the phenotype and biological functions of EPCs, two distinct subtypes have been proposed: endothelial colony-forming cells (ECFCs) and myeloid angiogenic cells (MACs) [28].
ECFCs derive from umbilical cord blood or peripheral blood mononuclear cells and are characterized by CD31+, CD45, CD14, CD146+, VE-Cadherin+, von Willebrand factor+ and VEGFR2+ phenotype. Their vasculogenic properties are linked to platelet-derived growth factor BB (PDGF-BB)/platelet-derived growth factor receptor (PDGFR) signaling [29,30].
MACs derive from peripheral blood mononuclear cells grown under endothelial cell culture conditions and are characterized by the following surface cell markers: CD45+, CD14+, CD31+, CD146, CD133 and Tie2. MACs promote angiogenesis through the activation of IL-8/VEGFR2/ERK signaling pathways, which results in endothelial proliferation, migration and tube formation. MACs do not differentiate into ECs but enhance the migration of circulating or vascular wall ECFCs to the injury area, where ECFCs proliferate, differentiate into mature ECs and restore the endothelial integrity of the vascular wall [25].
EPCs homing in the site of injury is characterized by recruitment, mobilization, adhesion and CXCR2, CXCR4 and CCR2 signaling. EPCs’ functional capacity has been reported to ameliorate after CXCR4 increased the expression via AKT/endothelial NO synthase pathways [31], and the augmentation of integrin receptor subunits present in EPCs promotes adhesion properties [32].
Endothelial injury is characterized by the up-regulation of intracellular and vascular adhesion molecules (ICAM-1) (VCAM-1) and hypoxia-inducible factor-1a (HIF-1a), which regulates the release of stromal-derived factor-I alfa (SDF-1a) and VEGF. These factors mediate the trafficking and recruitment of MACs to the target tissue [33,34]. MACs do not directly supply new ECs but activate the resident ECs through the release of growth factors, cytokines and transcription factors [35,36]. The paracrine release of VEGF, hepatic growth factor (HGF), Ang-1, SDF-1a, insulin-like growth factor (IGF)-1 and eNOS by MACs recruits and incorporates ECFCs to the network of new capillary vessels [37,38]. In addition, ECFCs release pro-angiogenic factors [39] and regulate the regenerative potential of mesenchymal stem cells [29]. Despite the exact origin of ECFCs not being clear, the identification of the endothelial progenitor/stem like population at the inner surface of the pre-existing blood vessels with colony-forming abilities has been reported [40]. ECFCs are crucial for the vascular repair and endothelial regeneration in different organs. Indeed, ECFCs have been shown to promote neovascularization and increase microvascular density in myocardial ischemia [41], ischemic stroke [42] and hindlimb ischemia [43].
Chronic inflammation and the release of inflammatory cytokines lead to the excessive proliferation of smooth muscle cells (SMCs), extracellular-matrix (ECM) deposition and the trans-differentiation of SMCs in myofibroblasts (MFs) [44]. MFs promote the activation of pro-inflammatory angiogenic factors and the modification of ECM. The excessive production of IL-6 and VEGF may negatively affect EPCs’ mobilization and recruitment, leading to an overactivation and defective vascular remodeling [17].
Increasing age was associated with lower circulating EPCs and lower VEGF levels in patients undergoing coronary artery bypass grafting. Notably, by-pass surgery could not provide a significant mobilization in EPCs in patients aged 69 years and older [45].
Another study including heart failure with preserved ejection fraction reported that the EPCs of older patients with heart failure with mild reduced ejection fraction presented lower migratory, proliferative and adhesion properties [46]. Compared to younger patients with NSTEMI myocardial infarction, the EPCs presented impaired function, and this was associated with the severity of the disease [47].
A significant reduction in CXCR4 expression in EPCs with aging has been described, and the modification of calcium-regulated functions of the CXCR4/ SDF-1a axis has been suggested as a possible mechanism [48,49]. Notably, the upregulation of CXCR4/JAK-2 signaling has been associated with the amelioration of EPCs properties and the restoration of endothelial dysfunction related to age [50].
Isolation, Culturing and Measurement of Human EPCs
A variety of methods are used for the isolation and quantification of EPCs from peripheral blood. However, cell surface phenotyping and cell cultures have been proposed as the main approaches [28,51,52].
Cell surface phenotyping is based on flow cytometry and fluorescent labeled antibodies. Flow cytometry requires a small amount of blood, and circulating EPCs are quantified as the percentage of the mononuclear cells that express VEGFR2 and CD34 [53]. In addition, CD133 surface protein has been associated with properties of EPCs by different studies [54,55]. Based on these studies, EPCs were identified as circulating CD34 cells that co-express VEGFR2 and CD133 as well. Furthermore, the quantification of EPCs by this approach could provide information regarding the relationship between the number of EPCs and the different diseases state. It should be mentioned that CD34, CD133 and VEGFR2 are not unique for EPCs, and their expression is also present in hematopoietic cells [56]. Other studies have reported that CD34 and CD45 cells enriched for hematopoietic cells co-expressed CD133 without VEGFR2. In addition, the CD34CD45 population of cells, which gave rise to colonies of ECs with high proliferative properties, expressed VEGFR2 without CD133 [51,57].
The isolation of EPCs by the cell culture approach was initially described by Asahara and colleagues [58]. Peripheral blood mononuclear cells were plated on fibronectin-coated dishes within five days, and the adherend cells expressed similar cell surface proteins to human umbilical vein ECs. To deplete the population of macrophages and monocytes that could contaminate the isolation of EPCs, Ito and co-workers [59] after a twenty four-hour period of adhesion, removed the non-adherent cells and re-plated them onto fibronectin-coated dishes for seven days. The cells that emerged after one week of the culture were considered as EPCs colonies. Another study cultured mononuclear cells from peripheral blood for forty-eight hours, and the non-adherent fraction was re-plated in a specific medium onto fibronectin-treated petri dishes. After five to seven days, colony-forming units (ECs) were obtained and named as CFU-Hill [60]. However, other studies demonstrated that CFU-Hill colonies include monocytes, myeloid progenitor cells and T lymphocytes [61,62].
The adherent cells obtained from circulating mononuclear cells plated in culture dishes in an endothelial growth medium present a morphology resembling the spindle-shape cells similar to CFU- Hill after three to four days of the culture and express markers typical of the ECs, such as von Willebrand factor VE-cadherin, CD31 and Tie2. Furthermore, these cells were demonstrated to promote vascularization [63] and are identified as early EPCs or circulating angiogenic cells (CACs). Considering that these cells are generated in in vitro conditions, it has been suggested to refer to them as MACs [28].
Late-outgrowth EPCs are isolated from the mononuclear cells of peripheral blood or umbilical cord blood adherent to collagen-I-coated culture dishes after two weeks of the culture. These cells are defined as ECFCs, differentiate into mature ECs and generate new vessels in vivo [64].
1.2. Frailty Models and Epidemiology
Several instruments have been developed to detect frailty, and, currently, the two main frailty models are: (a) the Fried Frailty Phenotype and (b) the Frailty Index (FI) based on the cumulative deficit model. The frailty phenotype model was established by the presence of the following criteria: (a) unintentional weight loss, (b) self-reported exhaustion, (c) low energy expenditure, (d) slow gait speed and (e) weak grip strength. The presence of one to two of these criteria identifies a pre-frail condition, while people with three or more criteria are considered frail. Despite multiple comorbidities and the presence of cognitive decline not being included in the development of this model, the application of the Frailty Phenotype has been validated, and the identification of frailty with this model has an independent role in the prediction of long-term mortality [7,65].
The cumulative deficit model is computed by the number of health deficits identified in different symptoms, signs, laboratory findings, comorbidities and disabilities [66]. The frailty index is derived from the number of health deficits divided by the total number of variables screened. Importantly, a value of 0.67 seems to identify an amount of frailty that is highly associated with mortality [66]. Several studies have reported that the Frailty Index was strongly related to institutionalization and poor survival [67,68].
Based on these models, many other screening tools have been developed and validated in different populations and clinical settings [69]. For instance, evaluations of the level of deficits based on CGA have been widely applied in different clinical settings [70,71,72]. The Study of Osteoporotic Fractures (SOF) frailty scale is a parsimonious frailty index including: weight loss, the inability to rise from a chair and poor energy, and it is able to predict disability, fractures and falls [73]. The Tilburg Frailty Indicator and the Groningen frailty indicator are questionnaires which evaluate physical, cognitive, social and psychological domains [74]. It should be mentioned that, in terms of the ability to predict negative outcomes, frailty models and screening tools demonstrate a degree of overlap, but different patient cohorts are identified as frail.
The prevalence of frailty varies by classification, sex and geographic region; however, an overall pooled prevalence of 12% has been estimated considering the Frailty Phenotype model or other tools focused on physical frailty [75]. The cumulative deficit model of frailty produces higher estimates of population-level prevalence, accounting for about 24% [75]. For pre-frailty, the overall estimates are 46% for physical frailty and 49% for cumulative deficit models, respectively. The prevalence increases with age, and higher proportions of frailty and pre-frailty are registered among the female population [76]. Regarding prevalence by geographic areas, in Europe, it is 8%, and the highest prevalence is in Africa (about 22%), considering the physical frailty model. Frailty evaluation by the cumulative deficit model produces the highest prevalence in Oceania (31%), followed by Asia (25%). In Europe, this model estimates an overall prevalence of frailty of 22%.
2. Impairment of Endothelial Function in Frailty
Endothelium function includes the exchange of molecules and fluids between blood and surrounding tissues, the maintenance of blood in a fluid state, the facilitation of the immune response, the control of vascular resistance, the regulation of the vascular tone and the creation of a new vascular network [14,17]. The endothelium acts through the paracrine and endocrine pathways and constantly maintains an equilibrium between vasodilatation and vasoconstriction, pro-inflammatory and anti-inflammatory mediators and pro-thrombotic and antithrombotic factors. The impairment of endothelial function is characterized by pro-inflammatory, prothrombotic and vasoconstrictor properties. A blunted response to agonist-induced vasodilation, the impairment of synthesis and the release of NO are the main characteristics of the dysfunctional endothelium.
Data from the Cardiovascular Health Study have reported that frail people are characterized by increased intima-medial thickness of the carotid arterial wall and a reduction in blood flow in the lower extremities despite the absence of clinical cardiovascular diseases [77]. For the first time, the Toledo Study for Healthy Aging revealed an association between endothelial dysfunction and frailty [78]. In this study, pre-frail and frail subjects were characterized by a higher asymmetric dimethylarginine (ADMA) level, which is an endogenous inhibitor of NO synthase. In contrast, the ADMA level was not significantly different in frail and/or pre-frail people compared to that in non-frail people, where atherosclerosis was present. In people without cardiovascular diseases, a higher ADMA level significantly increases the risk of frailty independently by well-established cardiovascular risk factors such as: hypertension, diabetes and dyslipidemia. Chronic inflammation and oxidative stress were suggested as complementary sources of endothelial dysfunction in frailty [79]. Another study also found that serum ADMA levels were correlated with physical domains of frailty. Higher ADMA levels are significantly associated with a lower muscle mass and muscle strength and a slower gait speed [80].
A cross-sectional study investigating the impact of different inflammatory mediators on the frailty status in elderly outpatients found that plasma nitrite levels, which are mainly derived from constitutive NO synthetase activity, were significantly reduced in frail people. This finding was linked to the presence of chronic low-grade inflammation, resulting in progressively increased CRP in frail people. However, after the adjustment for confounders, CRP was not significantly associated with frailty, whereas nitrite levels showed an independent role [81].
Brachial artery flow-mediated dilatation (FMD) and brachial angle pulse wave velocity (baPWV) are indirect measures of endothelial function, and their modifications indicate endothelial impairment, which is associated with atherosclerosis risk. FMD was significantly associated with a lower limb muscle power [82] and muscle mass index in the elderly population [83]. Furthermore, baPWV was negatively associated with handgrip strength dominantly among the non-hypertensive population [84]. Another study including thirty hospitalized elderly patients, where the frailty status was evaluated by the SOF frailty scale, revealed a lower FDD in frail compared to non-frail patients [85]. Frailty was also associated with an abnormal FDD in elderly patients with chronic kidney disease [86]. A lower ankle-brachial index was associated with frailty in two studies, but the analysis adjusted for confounders failed to show a significant correlation [87,88]. A recent cross-sectional analysis of 1096 men revealed that the femoral angle PWV was significantly associated with frailty among men without cardiovascular diseases but not in men with cardiovascular diseases [89].
Interestingly, a recent experimental study observed that the ex vivo measurement of endothelial-dependent dilatation (EDD) and the superoxide-mediated suppression of EDD were impaired in frail mice, independently of age [90]. Mouse frailty was determined by a 31-item frailty index based on clinical signs of deterioration in C57BL/6J mice [91]. The evaluation of the vestibulocochlear, ocular, nasal, respiratory, musculoskeletal, digestive and urogenital signs was included. Notably, endothelial dysfunction was significantly associated with frailty among old female and male mice, but the localization was cerebral arteries for female mice and the mesenteric artery for male mice.
3. EPCs Response to Endothelial Dysfunction in Frailty
The plasma concentration of ADMA has been negatively correlated with the number of MACs and ECFCs. The in vitro differentiation of EPCs was repressed in a concentration-dependent manner by ADMA, which also significantly reduced the incorporation of EPCs into endothelial tube-like structures [92]. Furthermore, the formation of colony-forming units from cultured peripheral blood mononuclear cells was inhibited. In this study, the detrimental effects of ADMA on EPCs were abolished by the co-incubation with the hydroxymethyl glutaryl coenzyme A reductase inhibitor [92]. Other studies have confirmed the suppression of EPCs by ADMA [93,94,95]. The dimethylarginine dimethylaminohydrolase (DDAH2)/ADMA pathway, through the activation of silent inhibitor 1, accelerated the senescence of EPCs [96]. ADMA’s negative role in EPCs function was also explained by the endoplasmatic reticulum (ER) stress pathway through the activation of phosphorylated protein kinase RNA-activated-like ER kinase (PERK), a stress sensor protein. In addition, the inhibition of the ER stress pathway by salubrinal attenuated the ADMA-induced apoptosis of EPCs [97]. Another in vitro study found that ADMA promoted EPCs apoptosis through the phosphorylation of JNK, targeted the inhibition of the JNK by SP600125, alleviated ADMA-induced apoptosis and promoted angiogenesis viability [98].
It has been reported that NOS inhibition attenuated the migration properties of EPCs, while NO donors enhanced VEGF-dependent chemotaxis. Importantly, eNOS levels were also significantly reduced in older patients as compared to healthy volunteers [99]. It has also been demonstrated that eNOS is present in EPCs and is dynamically expressed during the differentiation of EPCs to ECs [100].
The reduction in VEGF expression with aging has been described [101], and VEGF promoted EPCs incorporation into the damaged vessels, enhanced the differentiation of EPCs, as indicated by the increased expression of the EC markers CD31 and vWF, and promoted re-endothelization. The possible mechanism related to the VEGF-induced modification of EPCs has been mediated by connexin 43(Cx43), which is a gap junction protein and an important contributor in the intracellular communication. Indeed, VEGF increases the expression of Cx43 in EPCs and the inhibition of Cx43 expression using short interfering RNA (siRNA) attenuated EPCs gap junction intercellular communication and consequent EPCs differentiation [102].
Interestingly, a recent study revealed that VEGF signaling was greatly reduced in multiple key organs in an experimental model of mouse aging [103]. This was associated with the increased production of soluble VEGFR1, produced through an age-related shift in the alternative splicing of VEGFR1 mRNA, and its activity as a VEGF trap. VEGF supplementation resulted in increased longevity in mice. Considering the strict correlation between VEGF, NOS and EPCs, a reduction in VEGF and NOS expression or an impairment in their signaling is paralleled by a reduction in the impairment of the circulating EPCs number and by the impairment of their differentiation. The role of VEGF and NO in vascular aging has been studied widely; however, more research is necessary to clarify their role in frailty models and, consequently, the modification of EPCs. It has been suggested that the exchange of cAMP/PKA in gap junction proteins could interfere with EPCs functioning, and the blockade of PKA attenuates VEGF-induced EPCs differentiation [102,104]. It has been reported that VEGF activates phospholipase C and phosphatidylinositol-4,5-bisphosphate, which results in a cellular calcium increase and the regulation of endothelial differentiation-related genes.
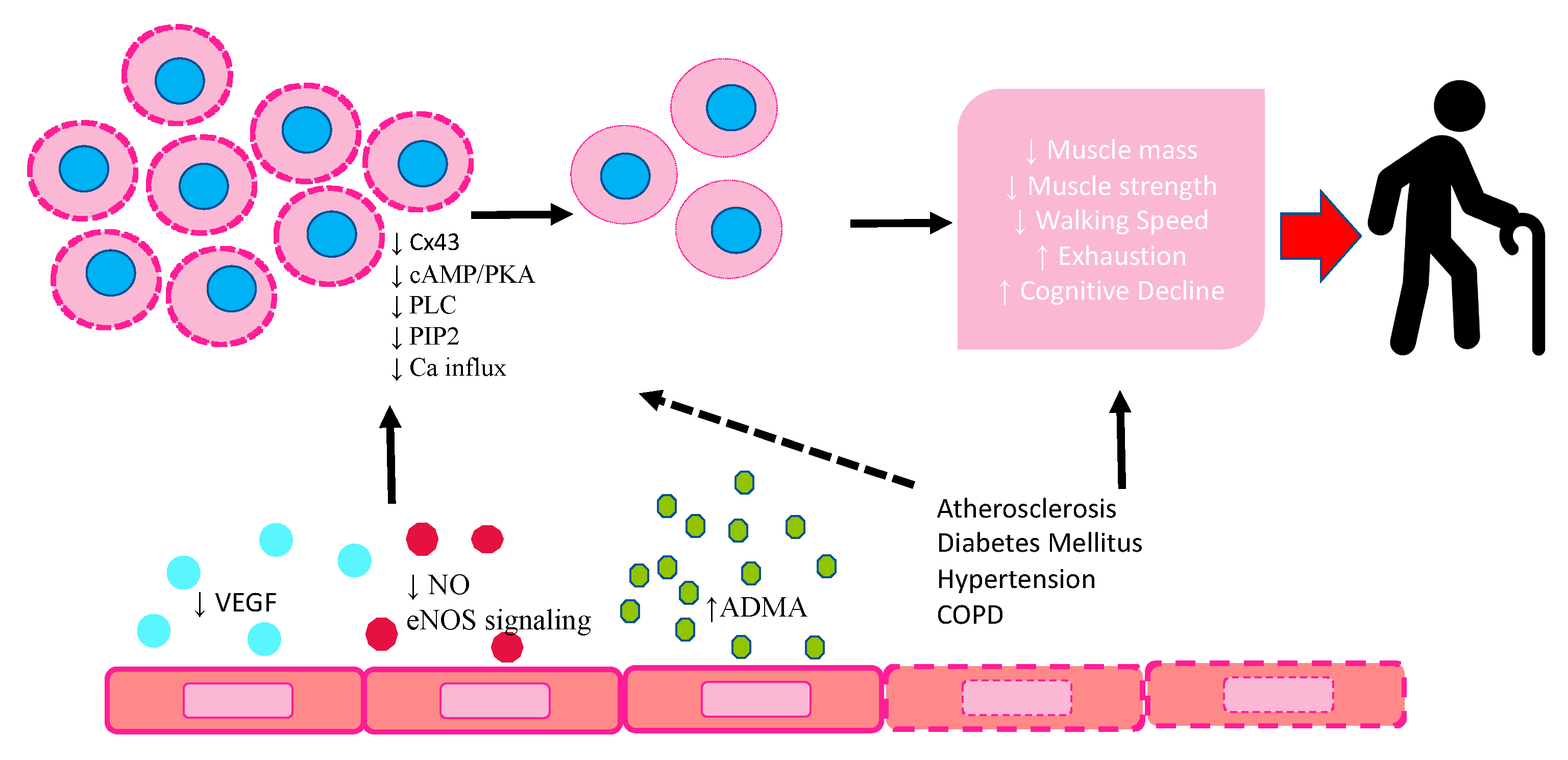
The main mechanisms responsible for EPCs modification in frailty are represented in Figure 1.
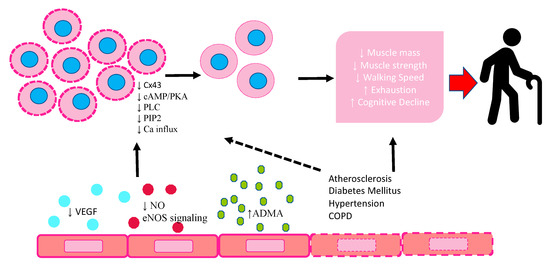
Figure 1.
Schematic diagram highlighting the response of EPCs to endothelium dysfunction in frailty and their modification. VEGF: vascular endothelial growth factor; NO: nitric oxide; eNOS: endothelial nitric oxide synthetase; ADMA: asymmetric dimethylarginine; CX43: connexin 43; cAMP/PKA: cyclic adenosine monophosphate/ protein kinase A; PLC: phospholipase C; PIP2: phosphatidylinositol-4,5-bisphosphate.
4. EPCs Dysfunction and Frailty Domains
It has been demonstrated that lower circulating EPCs are associated with a lower gait speed, a lower six-minute walk distance, a longer time for the chair stand test and a reduced SF-36 physical function score. In this study, EPCs were analyzed by flow cytometry and the analysis of cell surface expression markers: CD34+, CD133+, CD14+ and CD146+. It should be mentioned that the association of EPCs and physical function remained significant across several similar immature EPCs and multiple measures of physical function adjusted for age, BMI, comorbidities and markers of inflammation [105].
The same authors revealed that circulating EPCs levels are not only associated with baseline physical performance but are also able to predict the physical function decline. Lower baseline levels of EPCs were highly predictive of a lower gait speed and a shorter distance walked in 6 min at 3 and 12 months of follow-up. These associations were significant after the adjustment for age, body mass index and inflammation and were independent of interventions provided to improve physical function [106].
In addition, a cross-sectional study including community-dwelling elderly people reported a positive association between circulating EPCs and handgrip strength in hypertensive men. This association remained robust after the adjustment for cardiovascular risk factors [18].
Regarding cognitive function, which is another crucial domain of frailty, it has been reported that a lower level of EPCs was associated with the presence of mild cognitive impairment in the elderly. In addition, a lower circulating level of EPCs was present in patients with worse verbal and visual memory [107]. In contrast, a later study found no association between the EPCs level and cognitive decline in elderly subjects, suggesting that the multimorbidity observed in our patients may lead to opposite and confounding effects on endothelial biomarkers levels [108]. However, another study performed on 509 patients showed that lower counts of EPCs were associated with a worse memory performance and cognitive impairment in patients with coronary artery disease, and the association of EPCs with visual and verbal memory remained significant even after adjusting for confounders [109].
It has been postulated that coronary artery disease, hypertension, diabetes and aortic stenosis are characterized by the impairment of EPCs, and the association between cardiovascular comorbidities and frailty is well established. In addition, the EPCs number and migration were significantly decreased in patients with COPD, another important comorbidity related to frailty [110,111]. Notably, in community-dwelling elderly people affected by osteoporosis, a significant association between the circulating osteogenic cells population, disability and frailty based on the Fried criteria and the deficit model was identified [112]. Recent studies have underlined the potential relationship between reduced levels of circulating CD34+ hemopoietic progenitor stem cells (HSPCs) and frailty [113,114]. Low HSPCs are associated with pre-frailty and frailty and identify individuals with poor cardiovascular outcomes [114].
It should be mentioned that few studies have investigated the association of EPCs with frailty domains, and, moreover, frailty models/EPCs relationships should be explored.
5. Interventions That May Restore EPCs Functioning in Frailty
The effects of physical activity on frailty trajectories, mobility and disability are well documented in different studies [115,116,117,118,119]. The severity of frailty was reduced through one year of supervised home-based physical training [120]. A recent interventional study including patients with chronic heart failure reported that an exercise rehabilitation program enhanced the proliferation, migration and activity of EPCs. At the same time, the apoptosis rate was lower compared to that of the control group. Furthermore, the mRNA expression of PI3K, AKT, eNOS and VEGF was significantly higher in the intervention group compared to that in the control group [121].
Angiotensin II may promote the impairment of endothelial function and has adverse effects on skeletal muscle function and structure in experimental models [122], and elderly women who had taken ACE inhibitors continuously presented a lower decline in muscle strength compared with those who had taken other antihypertensive drugs or those who had never used antihypertensives during a long-term follow-up [123]. It has been reported that ACE inhibitors increased the EPCs level and restored the function of EPCs in hypertensive patients [124]. However, the existing evidence does not support the use of ACE inhibitors or angiotensin receptor blockers as a single intervention for improving physical performance in the elderly [125], and a recent study did not find an improvement in muscle mass related to perindopril and leucin supplementation [126].
A recent study demonstrated that, in patients with heart failure (HF) and diabetes, dapagliflozin, a sodium-glucose cotransporter (SGLT2) inhibitor, provided beneficial effects related to the worsening of HF, hospitalizations and cardiovascular death, regardless of frailty class. Furthermore, in patients with a greater degree of frailty, improvements in symptoms, physical function and quality of life were larger [127]. Even if previous research suggested that SGLT2 inhibitors did not influence the number of EPCs [128], a recent study revealed that canagliflozin increased the CXCR4 receptor expression and migratory profile of EPCs in patients with diabetes [129]. Furthermore, recent evidence suggests that empagliflozin ameliorates the frailty status in elderly people with diabetes by improving endothelial function via the reduction of mitochondrial oxidative stress [130].
6. Conclusions
The current evidence suggests that a reduction in the EPCs number characterizes frailty. The impairment of EPCs function may be a possible independent mechanism involved in the development of frailty. Increased ADMA expression, a reduction in VEGF and an impairment of NOS signaling appear to be the main pathways related to EPCs dysfunction in frailty. EPCs may represent a potential biomarker in the early detection of pre-frailty and frailty, the progression of frailty and the monitoring of interventions guided by frailty deficits. However, the role of EPCs as biomarkers capable of identifying frailty, the impact of EPCs in the clinical outcome of frail people and the modification of EPCs secondary to strategies which aim to ameliorate frailty should be further investigated.
Author Contributions
Conceptualization, K.K. and G.G.; methodology, K.K. and A.P.; investigation, K.K. and G.G.; resources, G.G.; data curation, K.K.; writing—original draft preparation, K.K.; writing—review and editing, K.K.; visualization, A.P.; supervision, G.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- United Nations Department of Economic and Social Affairs Population Division. World Population Ageing 2020 Highlights: Living Arrangements of Older Persons; (ST/ESA/SER.A/451); United Nations: New York, NY, USA, 2020. [Google Scholar]
- United Nations Department of Economic and Social Affairs Population Division. World Population Ageing 2019: Highlights; (ST/ESA/SER.A/430); United Nations: New York, NY, USA, 2019. [Google Scholar]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Cicutto, L.C. Frailty: Is This a New Vital Sign? Chest 2018, 154, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Chong, E.; Ho, E.; Baldevarona-Llego, J.; Chan, M.; Wu, L.; Tay, L. Frailty and Risk of Adverse Outcomes in Hospitalized Older Adults: A Comparison of Different Frailty Measures. J. Am. Med. Dir. Assoc. 2017, 18, 638e7–638e11. [Google Scholar] [CrossRef] [PubMed]
- Balestroni, G.; Panzeri, A.; Omarini, P.; Cerutti, P.; Sacco, D.; Giordano, A.; Pistono, M.; Komici, K.; Rossi Ferrario, S. Psychophysical health of elderly inpatients in cardiac rehabilitation: A retrospective cohort study. Eur. J. Phys. Rehabil. Med. 2020, 56, 197–205. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Komici, K.; Bencivenga, L.; Navani, N.; D’Agnano, V.; Guerra, G.; Bianco, A.; Rengo, G.; Perrotta, F. Frailty in Patients With Lung Cancer: A Systematic Review and Meta-Analysis. Chest 2022, 162, 485–497. [Google Scholar] [CrossRef]
- Hoogendijk, E.O.; Afilalo, J.; Ensrud, K.E.; Kowal, P.; Onder, G.; Fried, L.P. Frailty: Implications for clinical practice and public health. Lancet 2019, 394, 1365–1375. [Google Scholar] [CrossRef]
- Leng, S.X.; Xue, Q.L.; Tian, J.; Walston, J.D.; Fried, L.P. Inflammation and frailty in older women. J. Am. Geriatr. Soc. 2007, 55, 864–871. [Google Scholar] [CrossRef]
- Hubbard, R.E.; O’Mahony, M.S.; Savva, G.M.; Calver, B.L.; Woodhouse, K.W. Inflammation and frailty measures in older people. J. Cell Mol. Med. 2009, 13, 3103–3109. [Google Scholar] [CrossRef]
- Barzilay, J.I.; Blaum, C.; Moore, T.; Xue, Q.L.; Hirsch, C.H.; Walston, J.D.; Fried, L.P. Insulin resistance and inflammation as precursors of frailty: The Cardiovascular Health Study. Arch. Intern. Med. 2007, 167, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.A. The aging immune system: Primer and prospectus. Science 1996, 273, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Gimbrone, M.A., Jr.; Garcia-Cardena, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef]
- Madonna, R.; Novo, G.; Balistreri, C.R. Cellular and molecular basis of the imbalance between vascular damage and repair in ageing and age-related diseases: As biomarkers and targets for new treatments. Mech. Ageing Dev. 2016, 159, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Amarasekera, A.T.; Chang, D.; Schwarz, P.; Tan, T.C. Does vascular endothelial dysfunction play a role in physical frailty and sarcopenia? A systematic review. Age Ageing 2021, 50, 725–732. [Google Scholar] [CrossRef]
- Yang, J.X.; Pan, Y.Y.; Wang, X.X.; Qiu, Y.G.; Mao, W. Endothelial progenitor cells in age-related vascular remodeling. Cell Transplant. 2018, 27, 786–795. [Google Scholar] [CrossRef] [PubMed]
- Yamanashi, H.; Shimizu, Y.; Koyamatsu, J.; Nagayoshi, M.; Kadota, K.; Tamai, M.; Maeda, T. Circulating CD34-Positive Cells Are Associated with Handgrip Strength in Japanese Older Men: The Nagasaki Islands Study. J. Frailty Aging 2017, 6, 6–11. [Google Scholar]
- Qiu, Y.; Zhang, C.; Zhang, G.; Tao, J. Endothelial progenitor cells in cardiovascular diseases. Aging Med. 2018, 1, 204–208. [Google Scholar] [CrossRef]
- Perrotta, F.; Perna, A.; Komici, K.; Nigro, E.; Mollica, M.; D’Agnano, V.; De Luca, A.; Guerra, G. The State of Art of Regenerative Therapy in Cardiovascular Ischemic Disease: Biology, Signaling Pathways, and Epigenetics of Endothelial Progenitor Cells. Cells 2020, 9, 1886. [Google Scholar] [CrossRef]
- Hu, L.; Dai, S.C.; Luan, X.; Chen, J.; Cannavicci, A. Dysfunction and Therapeutic Potential of Endothelial Progenitor Cells in Diabetes Mellitus. J. Clin. Med. Res. 2018, 10, 752–757. [Google Scholar] [CrossRef]
- Komici, K.; Perna, A.; Rocca, A.; Bencivenga, L.; Rengo, G.; Guerra, G. Endothelial Progenitor Cells and Rheumatoid Arthritis: Response to Endothelial Dysfunction and Clinical Evidences. Int. J. Mol. Sci. 2021, 22, 13675. [Google Scholar] [CrossRef]
- Custodia, A.; Ouro, A.; Romaus-Sanjurjo, D.; Pias-Peleteiro, J.M.; de Vries, H.E.; Castillo, J.; Sobrino, T. Endothelial Progenitor Cells and Vascular Alterations in Alzheimer’s Disease. Front Aging Neurosci. 2021, 13, 811210. [Google Scholar] [CrossRef]
- Hristov, M.; Weber, C. Endothelial progenitor cells in vascular repair and remodeling. Pharmacol. Res. 2008, 58, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Kawamoto, A.; Masuda, H. Concise review: Circulating endothelial progenitor cells for vascular medicine. Stem Cells 2011, 29, 1650–1655. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Takahashi, T.; Masuda, H.; Kalka, C.; Chen, D.; Iwaguro, H.; Inai, Y.; Silver, M.; Isner, J.M. VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J. 1999, 18, 3964–3972. [Google Scholar] [CrossRef] [PubMed]
- Aicher, A.; Heeschen, C.; Mildner-Rihm, C.; Urbich, C.; Ihling, C.; Technau-Ihling, K.; Zeiher, A.M.; Dimmeler, S. Essential role of endothelial nitric oxide synthase for mobilization of stem and progenitor cells. Nat. Med. 2003, 9, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Medina, R.J.; Barber, C.L.; Sabatier, F.; Dignat-George, F.; Melero-Martin, J.M.; Khosrotehrani, K.; Ohneda, O.; Randi, A.M.; Chan, J.K.Y.; Yamaguchi, T.; et al. Endothelial Progenitors: A Consensus Statement on Nomenclature. Stem Cells Transl. Med. 2017, 6, 1316–1320. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.Z.; Moreno-Luna, R.; Li, D.; Jaminet, S.C.; Greene, A.K.; Melero-Martin, J.M. Human endothelial colony-forming cells serve as trophic mediators for mesenchymal stem cell engraftment via paracrine signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 10137–10142. [Google Scholar] [CrossRef] [PubMed]
- Moccia, F.; Negri, S.; Shekha, M.; Faris, P.; Guerra, G. Endothelial Ca(2+) Signaling, Angiogenesis and Vasculogenesis: Just What It Takes to Make a Blood Vessel. Int. J. Mol. Sci. 2019, 20, 3962. [Google Scholar] [CrossRef]
- de Nigris, F.; Balestrieri, M.L.; Williams-Ignarro, S.; D’Armiento, F.P.; Lerman, L.O.; Byrns, R.; Crimi, E.; Palagiano, A.; Fatigati, G.; Ignarro, L.J.; et al. Therapeutic effects of autologous bone marrow cells and metabolic intervention in the ischemic hindlimb of spontaneously hypertensive rats involve reduced cell senescence and CXCR4/Akt/eNOS pathways. J. Cardiovasc. Pharmacol. 2007, 50, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Walter, D.H.; Rittig, K.; Bahlmann, F.H.; Kirchmair, R.; Silver, M.; Murayama, T.; Nishimura, H.; Losordo, D.W.; Asahara, T.; Isner, J.M. Statin therapy accelerates reendothelialization: A novel effect involving mobilization and incorporation of bone marrow-derived endothelial progenitor cells. Circulation 2002, 105, 3017–3024. [Google Scholar] [CrossRef]
- Yoon, C.H.; Hur, J.; Oh, I.Y.; Park, K.W.; Kim, T.Y.; Shin, J.H.; Kim, J.H.; Lee, C.S.; Chung, J.K.; Park, Y.B.; et al. Intercellular adhesion molecule-1 is upregulated in ischemic muscle, which mediates trafficking of endothelial progenitor cells. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1066–1072. [Google Scholar] [CrossRef] [PubMed]
- Hookham, M.B.; Ali, I.H.; O’Neill, C.L.; Hackett, E.; Lambe, M.H.; Schmidt, T.; Medina, R.J.; Chamney, S.; Rao, B.; McLoone, E.; et al. Hypoxia-induced responses by endothelial colony-forming cells are modulated by placental growth factor. Stem Cell Res. Ther. 2016, 7, 173. [Google Scholar] [CrossRef] [PubMed]
- Rehman, J.; Li, J.; Orschell, C.M.; March, K.L. Peripheral blood „endothelial progenitor cells” are derived from monocyte/macrophages and secrete angiogenic growth factors. Circulation 2003, 107, 1164–1169. [Google Scholar] [CrossRef]
- Wang, L.; Wang, X.; Xie, G.; Wang, L.; Hill, C.K.; DeLeve, L.D. Liver sinusoidal endothelial cell progenitor cells promote liver regeneration in rats. J. Clin. Investig. 2012, 122, 1567–1573. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Suyama, T.; Yashita, T.; Akimaru, H.; Kurata, H. Bone marrow subpopulations contain distinct types of endothelial progenitor cells and angiogenic cytokine-producing cells. J. Mol. Cell Cardiol. 2007, 43, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Ii, M.; Nishimura, H.; Iwakura, A.; Wecker, A.; Eaton, E.; Asahara, T.; Losordo, D.W. Endothelial progenitor cells are rapidly recruited to myocardium and mediate protective effect of ischemic preconditioning via “imported” nitric oxide synthase activity. Circulation 2005, 111, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
- Collett, J.A.; Mehrotra, P.; Crone, A.; Shelley, W.C.; Yoder, M.C.; Basile, D.P. Endothelial colony-forming cells ameliorate endothelial dysfunction via secreted factors following ischemia-reperfusion injury. Am. J. Physiol. Renal. Physiol. 2017, 312, F897–F907. [Google Scholar] [CrossRef] [PubMed]
- Naito, H.; Kidoya, H.; Sakimoto, S.; Wakabayashi, T.; Takakura, N. Identification and characterization of a resident vascular stem/progenitor cell population in preexisting blood vessels. EMBO J. 2012, 31, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.T.; Coggins, M.; Xiao, C.; Rosenzweig, A.; Bischoff, J. Human vasculogenic cells form functional blood vessels and mitigate adverse remodeling after ischemia reperfusion injury in rats. Angiogenesis 2013, 16, 773–784. [Google Scholar] [CrossRef]
- Ding, J.; Zhao, Z.; Wang, C.; Wang, C.X.; Li, P.C.; Qian, C.; Teng, G.J. Bioluminescence imaging of transplanted human endothelial colony-forming cells in an ischemic mouse model. Brain Res. 2016, 1642, 209–218. [Google Scholar] [CrossRef]
- Kang, K.T.; Lin, R.Z.; Kuppermann, D.; Melero-Martin, J.M.; Bischoff, J. Endothelial colony forming cells and mesenchymal progenitor cells form blood vessels and increase blood flow in ischemic muscle. Sci. Rep. 2017, 7, 770. [Google Scholar] [CrossRef] [PubMed]
- Forte, A.; Della Corte, A.; De Feo, M.; Cerasuolo, F.; Cipollaro, M. Role of myofibroblasts in vascular remodelling: Focus on restenosis and aneurysm. Cardiovasc. Res. 2010, 88, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Scheubel, R.J.; Zorn, H.; Silber, R.E.; Kuss, O.; Morawietz, H.; Holtz, J.; Simm, A. Age-dependent depression in circulating endothelial progenitor cells in patients undergoing coronary artery bypass grafting. J. Am. Coll. Cardiol. 2003, 42, 2073–2080. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Zhang, C.; Cai, G.; Zhang, B.; Huang, Z.; Wu, M.; Zhu, Y.; Luo, L.; He, H.; Yang, Z. Aging-Related Endothelial Progenitor Cell Dysfunction and Its Association with IL-17 and IL-23 in HFmrEF Patients. Oxid. Med. Cell Longev. 2022, 2022, 2281870. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Zhang, C.; Zhu, Y.; Liu, Z.; Liu, G.; Zhang, B.; Tu, C.; Yang, Z. Hypofunction of Circulating Endothelial Progenitor Cells and Aggravated Severity in Elderly Male Patients With Non-ST Segment Elevation Myocardial Infarction: Its Association With Systemic Inflammation. Front. Cardiovasc. Med. 2021, 8, 687590. [Google Scholar] [CrossRef]
- Shao, H.; Xu, Q.; Wu, Q.; Ma, Q.; Salgueiro, L.; Wang, J.; Eton, D.; Webster, K.A.; Yu, H. Defective CXCR4 expression in aged bone marrow cells impairs vascular regeneration. J. Cell Mol. Med. 2011, 15, 2046–2056. [Google Scholar] [CrossRef]
- Xu, Q.; Wang, J.; He, J.; Zhou, M.; Adi, J.; Webster, K.A.; Yu, H. Impaired CXCR4 expression and cell engraftment of bone marrow-derived cells from aged atherogenic mice. Atherosclerosis 2011, 219, 92–99. [Google Scholar] [CrossRef]
- Xia, W.H.; Li, J.; Su, C.; Yang, Z.; Chen, L.; Wu, F.; Zhang, Y.Y.; Yu, B.B.; Qiu, Y.X.; Wang, S.M.; et al. Physical exercise attenuates age-associated reduction in endothelium-reparative capacity of endothelial progenitor cells by increasing CXCR4/JAK-2 signaling in healthy men. Aging Cell 2012, 11, 111–119. [Google Scholar] [CrossRef]
- Hirschi, K.K.; Ingram, D.A.; Yoder, M.C. Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1584–1595. [Google Scholar] [CrossRef]
- Moccia, F.; Zuccolo, E.; Poletto, V.; Cinelli, M.; Bonetti, E.; Guerra, G.; Rosti, V. Endothelial progenitor cells support tumour growth and metastatisation: Implications for the resistance to anti-angiogenic therapy. Tumour. Biol. 2015, 36, 6603–6614. [Google Scholar] [CrossRef]
- Khan, S.S.; Solomon, M.A.; McCoy, J.P., Jr. Detection of circulating endothelial cells and endothelial progenitor cells by flow cytometry. Cytometry B Clin. Cytom. 2005, 64, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Peichev, M.; Naiyer, A.J.; Pereira, D.; Zhu, Z.; Lane, W.J.; Williams, M.; Oz, M.C.; Hicklin, D.J.; Witte, L.; Moore, M.A.; et al. Expression of VEGFR-2 and AC133 by circulating human CD34(+) cells identifies a population of functional endothelial precursors. Blood 2000, 95, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Gehling, U.M.; Ergun, S.; Schumacher, U.; Wagener, C.; Pantel, K.; Otte, M.; Schuch, G.; Schafhausen, P.; Mende, T.; Kilic, N.; et al. In vitro differentiation of endothelial cells from AC133-positive progenitor cells. Blood 2000, 95, 3106–3112. [Google Scholar] [CrossRef]
- Case, J.; Mead, L.E.; Bessler, W.K.; Prater, D.; White, H.A.; Saadatzadeh, M.R.; Bhavsar, J.R.; Yoder, M.C.; Haneline, L.S.; Ingram, D.A. Human CD34+AC133+VEGFR-2+ cells are not endothelial progenitor cells but distinct, primitive hematopoietic progenitors. Exp. Hematol. 2007, 35, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, F.; Van Hauwermeiren, F.; De Smedt, M.; Raedt, R.; Plasschaert, F.; De Buyzere, M.L.; Gillebert, T.C.; Plum, J.; Vandekerckhove, B. Endothelial outgrowth cells are not derived from CD133+ cells or CD45+ hematopoietic precursors. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1572–1579. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Rovira, I.I.; Bloom, M.L.; Takeda, K.; Ferrans, V.J.; Quyyumi, A.A.; Finkel, T. Endothelial progenitor cells as putative targets for angiostatin. Cancer Res. 1999, 59, 5875–5877. [Google Scholar]
- Hill, J.M.; Zalos, G.; Halcox, J.P.; Schenke, W.H.; Waclawiw, M.A.; Quyyumi, A.A.; Finkel, T. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N. Engl. J. Med. 2003, 348, 593–600. [Google Scholar] [CrossRef]
- Yoder, M.C.; Mead, L.E.; Prater, D.; Krier, T.R.; Mroueh, K.N.; Li, F.; Krasich, R.; Temm, C.J.; Prchal, J.T.; Ingram, D.A. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 2007, 109, 1801–1809. [Google Scholar] [CrossRef]
- Rohde, E.; Malischnik, C.; Thaler, D.; Maierhofer, T.; Linkesch, W.; Lanzer, G.; Guelly, C.; Strunk, D. Blood monocytes mimic endothelial progenitor cells. Stem Cells 2006, 24, 357–367. [Google Scholar] [CrossRef]
- Kalka, C.; Masuda, H.; Takahashi, T.; Kalka-Moll, W.M.; Silver, M.; Kearney, M.; Li, T.; Isner, J.M.; Asahara, T. Transplantation of ex vivo expanded endothelial progenitor cells for therapeutic neovascularization. Proc. Natl. Acad. Sci. USA 2000, 97, 3422–3427. [Google Scholar] [CrossRef]
- Lin, Y.; Weisdorf, D.J.; Solovey, A.; Hebbel, R.P. Origins of circulating endothelial cells and endothelial outgrowth from blood. J. Clin. Investig. 2000, 105, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Rothman, M.D.; Leo-Summers, L.; Gill, T.M. Prognostic significance of potential frailty criteria. J. Am. Geriatr. Soc. 2008, 56, 2211–2216. [Google Scholar] [CrossRef]
- Mitnitski, A.B.; Mogilner, A.J.; Rockwood, K. Accumulation of deficits as a proxy measure of aging. Sci. World J. 2001, 1, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Rockwood, K.; Mitnitski, A.; Song, X.; Steen, B.; Skoog, I. Long-term risks of death and institutionalization of elderly people in relation to deficit accumulation at age 70. J. Am. Geriatr. Soc. 2006, 54, 975–979. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Mitnitski, A.; Rockwood, K. Prevalence and 10-year outcomes of frailty in older adults in relation to deficit accumulation. J. Am. Geriatr. Soc. 2010, 58, 681–687. [Google Scholar] [CrossRef]
- Sutton, J.L.; Gould, R.L.; Daley, S.; Coulson, M.C.; Ward, E.V.; Butler, A.M.; Nunn, S.P.; Howard, R.J. Psychometric properties of multicomponent tools designed to assess frailty in older adults: A systematic review. BMC Geriatr. 2016, 16, 55. [Google Scholar] [CrossRef]
- Jones, D.; Song, X.; Mitnitski, A.; Rockwood, K. Evaluation of a frailty index based on a comprehensive geriatric assessment in a population based study of elderly Canadians. Aging Clin. Exp. Res. 2005, 17, 465–471. [Google Scholar] [CrossRef]
- Nishijima, T.F.; Shimokawa, M.; Esaki, T.; Morita, M.; Toh, Y.; Muss, H.B. A 10-Item Frailty Index Based on a Comprehensive Geriatric Assessment (FI-CGA-10) in Older Adults with Cancer: Development and Construct Validation. Oncologist 2021, 26, e1751–e1760. [Google Scholar] [CrossRef]
- Komici, K.; Cappuccio, M.; Scacchi, A.; Vaschetti, R.; Delli Carpini, G.; Picerno, V.; Avella, P.; Brunese, M.C.; Rengo, G.; Guerra, G.; et al. The Prevalence and the Impact of Frailty in Hepato-Biliary Pancreatic Cancers: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 1116. [Google Scholar] [CrossRef]
- Ensrud, K.E.; Ewing, S.K.; Cawthon, P.M.; Fink, H.A.; Taylor, B.C.; Cauley, J.A.; Dam, T.T.; Marshall, L.M.; Orwoll, E.S.; Cummings, S.R.; et al. A comparison of frailty indexes for the prediction of falls, disability, fractures, and mortality in older men. J. Am. Geriatr. Soc. 2009, 57, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Metzelthin, S.F.; Daniels, R.; van Rossum, E.; de Witte, L.; van den Heuvel, W.J.; Kempen, G.I. The psychometric properties of three self-report screening instruments for identifying frail older people in the community. BMC Public Health 2010, 10, 176. [Google Scholar] [CrossRef] [PubMed]
- O’Caoimh, R.; Sezgin, D.; O’Donovan, M.R.; Molloy, D.W.; Clegg, A.; Rockwood, K.; Liew, A. Prevalence of frailty in 62 countries across the world: A systematic review and meta-analysis of population-level studies. Age Ageing 2021, 50, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Lugtenberg, M.; Franse, C.; Fang, X.; Hu, S.; Jin, C.; Raat, H. Risk factors and protective factors associated with incident or increase of frailty among community-dwelling older adults: A systematic review of longitudinal studies. PLoS ONE 2017, 12, e0178383. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.B.; Gottdiener, J.S.; McBurnie, M.A.; Hirsch, C.H.; Kop, W.J.; Tracy, R.; Walston, J.D.; Fried, L.P.; Cardiovascular Health Study Research Group. Associations of subclinical cardiovascular disease with frailty. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M158–M166. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Bouzon, C.; Carcaillon, L.; Garcia-Garcia, F.J.; Amor-Andres, M.S.; El Assar, M.; Rodriguez-Manas, L. Association between endothelial dysfunction and frailty: The Toledo Study for Healthy Aging. Age 2014, 36, 495–505. [Google Scholar] [CrossRef]
- Rodriguez-Manas, L.; El-Assar, M.; Vallejo, S.; Lopez-Doriga, P.; Solis, J.; Petidier, R.; Montes, M.; Nevado, J.; Castro, M.; Gomez-Guerrero, C.; et al. Endothelial dysfunction in aged humans is related with oxidative stress and vascular inflammation. Aging Cell 2009, 8, 226–238. [Google Scholar] [CrossRef]
- Obayashi, K.; Saeki, K.; Maegawa, T.; Sakai, T.; Kitagawa, M.; Otaki, N.; Kataoka, H.; Kurumatani, N. Association of Serum Asymmetric Dimethylarginine With Muscle Strength and Gait Speed: A Cross-Sectional Study of the HEIJO-KYO Cohort. J. Bone Miner Res. 2016, 31, 1107–1113. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Marcos-Perez, D.; Lorenzi, M.; Onder, G.; Gostner, J.M.; Strasser, B.; Fuchs, D.; Bonassi, S. Immunological alterations in frail older adults: A cross sectional study. Exp. Gerontol. 2018, 112, 119–126. [Google Scholar] [CrossRef]
- Heffernan, K.S.; Chale, A.; Hau, C.; Cloutier, G.J.; Phillips, E.M.; Warner, P.; Nickerson, H.; Reid, K.F.; Kuvin, J.T.; Fielding, R.A. Systemic vascular function is associated with muscular power in older adults. J. Aging Res. 2012, 2012, 386387. [Google Scholar] [CrossRef]
- Campos, A.M.; Moura, F.A.; Santos, S.N.; Freitas, W.M.; Sposito, A.C.; Brasilia Study on Healthy, A.; Brasilia Heart, S. Sarcopenia, but not excess weight or increased caloric intake, is associated with coronary subclinical atherosclerosis in the very elderly. Atherosclerosis 2017, 258, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Yamanashi, H.; Kulkarni, B.; Edwards, T.; Kinra, S.; Koyamatsu, J.; Nagayoshi, M.; Shimizu, Y.; Maeda, T.; Cox, S.E. Association between atherosclerosis and handgrip strength in non-hypertensive populations in India and Japan. Geriatr. Gerontol. Int. 2018, 18, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Santillo, E.M.M.; Balestrini, F. Frailty and flow-mediated dilation: A pilot study in hospitalized elderly. J. Curr. Res. Sci. Med. 2016, 2, 92. [Google Scholar] [CrossRef]
- Mansur, H.N.; Lovisi, J.C.; Colugnati, F.A.; Raposo, N.R.; Fernandes, N.M.; Bastos, M.G. Association of frailty with endothelial dysfunction and its possible impact on negative outcomes in Brazilian predialysis patients with chronic kidney disease. BMC Nephrol. 2015, 16, 157. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Auyeung, T.W.; Leung, J.; Kwok, T.; Leung, P.C.; Woo, J. Physical frailty in older adults is associated with metabolic and atherosclerotic risk factors and cognitive impairment independent of muscle mass. J. Nutr. Health Aging 2011, 15, 857–862. [Google Scholar] [CrossRef]
- Del Brutto, O.H.; Mera, R.M.; Brown, D.L.; Nieves, J.L.; Milla-Martinez, M.F.; Fanning, K.D.; Cagino, K.; Zambrano, M.; Sedler, M.J. The association of frailty with abnormal ankle-brachial index determinations is related to age: Results from the Atahualpa Project. Int. J. Cardiol. 2016, 202, 366–367. [Google Scholar] [CrossRef]
- McKechnie, D.G.J.; Patel, M.; Papacosta, A.O.; Lennon, L.T.; Ellins, E.A.; Halcox, J.P.J.; Ramsay, S.E.; Whincup, P.H.; Wannamethee, S.G. Associations between inflammation, coagulation, cardiac strain and injury, and subclinical vascular disease with frailty in older men: A cross-sectional study. BMC Geriatr. 2022, 22, 405. [Google Scholar] [CrossRef]
- Cole, J.A.; Kehmeier, M.N.; Bedell, B.R.; Krishna Kumaran, S.; Henson, G.D.; Walker, A.E. Sex Differences in the Relation between Frailty and Endothelial Dysfunction in Old Mice. J. Gerontol. A Biol. Sci. Med. Sci. 2022, 77, 416–423. [Google Scholar] [CrossRef]
- Whitehead, J.C.; Hildebrand, B.A.; Sun, M.; Rockwood, M.R.; Rose, R.A.; Rockwood, K.; Howlett, S.E. A clinical frailty index in aging mice: Comparisons with frailty index data in humans. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 621–632. [Google Scholar] [CrossRef]
- Thum, T.; Tsikas, D.; Stein, S.; Schultheiss, M.; Eigenthaler, M.; Anker, S.D.; Poole-Wilson, P.A.; Ertl, G.; Bauersachs, J. Suppression of endothelial progenitor cells in human coronary artery disease by the endogenous nitric oxide synthase inhibitor asymmetric dimethylarginine. J. Am. Coll. Cardiol. 2005, 46, 1693–1701. [Google Scholar] [CrossRef]
- Goette, A.; Wolfram, O.; Jentsch-Ullrich, K.; Martens-Lobenhoffer, J.; Scalera, F.; Lendeckel, U.; Bode-Boger, S.M. The effects of G-CSF-induced mobilization of progenitor cells are limited by ADMA. Int. J. Cardiol. 2010, 143, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Surdacki, A.; Marewicz, E.; Wieczorek-Surdacka, E.; Rakowski, T.; Szastak, G.; Pryjma, J.; Dudek, D.; Dubiel, J.S. Synergistic effects of asymmetrical dimethyl-L-arginine accumulation and endothelial progenitor cell deficiency on renal function decline during a 2-year follow-up in stable angina. Nephrol. Dial. Transplant. 2010, 25, 2576–2583. [Google Scholar] [CrossRef] [PubMed]
- Ozkok, A.; Aktas, E.; Yilmaz, A.; Telci, A.; Oflaz, H.; Deniz, G.; Yildiz, A. Decrease in endothelial progenitor cells associated with inflammation, but not with endothelial dysfunction in chronic hemodialysis patients. Clin. Nephrol. 2013, 79, 21–30. [Google Scholar] [CrossRef]
- Yuan, Q.; Hu, C.P.; Gong, Z.C.; Bai, Y.P.; Liu, S.Y.; Li, Y.J.; Jiang, J.L. Accelerated onset of senescence of endothelial progenitor cells in patients with type 2 diabetes mellitus: Role of dimethylarginine dimethylaminohydrolase 2 and asymmetric dimethylarginine. Biochem. Biophys. Res. Commun. 2015, 458, 869–876. [Google Scholar] [CrossRef]
- Ye, S.; Zhou, X.; Lin, J.; Chen, P. Asymmetric Dimethylarginine Induced Apoptosis and Dysfunction of Endothelial Progenitor Cells: Role of Endoplasmic Reticulum Stress Pathway. Biomed. Res. Int. 2017, 2017, 6395601. [Google Scholar] [CrossRef]
- Zhang, F.Q.; Lu, W.; Yuan, W.X.; Li, X. Regulation of c-Jun N-Terminal Protein Kinase (JNK) Pathway in Apoptosis of Endothelial Outgrowth Cells Induced by Asymmetric Dimethylarginine. Med. Sci. Monit. 2017, 23, 2535–2542. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Schanz, A.; Amabile, N.; Jahn, S.; Chen, Q.; Wong, M.L.; Rassaf, T.; Heinen, Y.; Cortese-Krott, M.; Grossman, W.; et al. Nitric oxide synthase expression and functional response to nitric oxide are both important modulators of circulating angiogenic cell response to angiogenic stimuli. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2212–2218. [Google Scholar] [CrossRef]
- Liu, Z.; Jiang, Y.; Hao, H.; Gupta, K.; Xu, J.; Chu, L.; McFalls, E.; Zweier, J.; Verfaillie, C.; Bache, R.J. Endothelial nitric oxide synthase is dynamically expressed during bone marrow stem cell differentiation into endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H1760–H1765. [Google Scholar] [CrossRef]
- Sadoun, E.; Reed, M.J. Impaired angiogenesis in aging is associated with alterations in vessel density, matrix composition, inflammatory response, and growth factor expression. J. Histochem. Cytochem. 2003, 51, 1119–1130. [Google Scholar] [CrossRef]
- Li, L.; Liu, H.; Xu, C.; Deng, M.; Song, M.; Yu, X.; Xu, S.; Zhao, X. VEGF promotes endothelial progenitor cell differentiation and vascular repair through connexin 43. Stem Cell Res. Ther. 2017, 8, 237. [Google Scholar] [CrossRef]
- Grunewald, M.; Kumar, S.; Sharife, H.; Volinsky, E.; Gileles-Hillel, A.; Licht, T.; Permyakova, A.; Hinden, L.; Azar, S.; Friedmann, Y.; et al. Counteracting age-related VEGF signaling insufficiency promotes healthy aging and extends life span. Science 2021, 373, 8479. [Google Scholar] [CrossRef] [PubMed]
- Yamamizu, K.; Kawasaki, K.; Katayama, S.; Watabe, T.; Yamashita, J.K. Enhancement of vascular progenitor potential by protein kinase A through dual induction of Flk-1 and Neuropilin-1. Blood 2009, 114, 3707–3716. [Google Scholar] [CrossRef] [PubMed]
- Povsic, T.J.; Sloane, R.; Zhou, J.; Pieper, C.F.; Pearson, M.P.; Peterson, E.D.; Green, J.B.; Cohen, H.J.; Morey, M.C. Lower levels of circulating progenitor cells are associated with low physical function and performance in elderly men with impaired glucose tolerance: A pilot substudy from the VA Enhanced Fitness trial. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 1559–1566. [Google Scholar] [CrossRef]
- Povsic, T.J.; Sloane, R.; Pieper, C.F.; Pearson, M.P.; Peterson, E.D.; Cohen, H.J.; Morey, M.C. Endothelial Progenitor Cell Levels Predict Future Physical Function: An Exploratory Analysis From the VA Enhanced Fitness Study. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 362–369. [Google Scholar] [CrossRef]
- Nation, D.A.; Tan, A.; Dutt, S.; McIntosh, E.C.; Yew, B.; Ho, J.K.; Blanken, A.E.; Jang, J.Y.; Rodgers, K.E.; Gaubert, A. Circulating Progenitor Cells Correlate with Memory, Posterior Cortical Thickness, and Hippocampal Perfusion. J. Alzheimers Dis. 2018, 61, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Breining, A.; Silvestre, J.S.; Dieudonne, B.; Vilar, J.; Faucounau, V.; Verny, M.; Neri, C.; Boulanger, C.M.; Boddaert, J. Biomarkers of vascular dysfunction and cognitive decline in patients with Alzheimer’s disease: No evidence for association in elderly subjects. Aging Clin. Exp. Res. 2016, 28, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Moazzami, K.; Wittbrodt, M.T.; Lima, B.B.; Kim, J.H.; Hammadah, M.; Ko, Y.A.; Obideen, M.; Abdelhadi, N.; Kaseer, B.; Gafeer, M.M.; et al. Circulating Progenitor Cells and Cognitive Impairment in Men and Women with Coronary Artery Disease. J. Alzheimers Dis. 2020, 74, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xie, C. Human endothelial progenitor cells isolated from COPD patients are dysfunctional. Mol. Cell Biochem. 2012, 363, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Palange, P.; Testa, U.; Huertas, A.; Calabro, L.; Antonucci, R.; Petrucci, E.; Pelosi, E.; Pasquini, L.; Satta, A.; Morici, G.; et al. Circulating haemopoietic and endothelial progenitor cells are decreased in COPD. Eur. Respir. J. 2006, 27, 529–541. [Google Scholar] [CrossRef]
- Gunawardene, P.; Al Saedi, A.; Singh, L.; Bermeo, S.; Vogrin, S.; Phu, S.; Suriyaarachchi, P.; Pignolo, R.J.; Duque, G. Age, gender, and percentage of circulating osteoprogenitor (COP) cells: The COP Study. Exp. Gerontol. 2017, 96, 68–72. [Google Scholar] [CrossRef]
- Grasselli, C.; Bombelli, S.; Eriani, S.; Domenici, G.; Galluccio, R.; Tropeano, C.; De Marco, S.; Bolognesi, M.M.; Torsello, B.; Bianchi, C.; et al. DNA Damage in Circulating Hematopoietic Progenitor Stem Cells as Promising Biological Sensor of Frailty. J. Gerontol. A Biol. Sci. Med. Sci. 2022, 77, 1279–1286. [Google Scholar] [CrossRef] [PubMed]
- Bonora, B.M.; Palano, M.T.; Testa, G.; Fadini, G.P.; Sangalli, E.; Madotto, F.; Persico, G.; Casciaro, F.; Vono, R.; Colpani, O.; et al. Hematopoietic progenitor cell liabilities and alarmins S100A8/A9-related inflammaging associate with frailty and predict poor cardiovascular outcomes in older adults. Aging Cell 2022, 21, e13545. [Google Scholar] [CrossRef] [PubMed]
- Quach, J.; Theou, O.; Perez-Zepeda, M.U.; Godin, J.; Rockwood, K.; Kehler, D.S. Effect of a physical activity intervention and frailty on frailty trajectory and major mobility disability. J. Am. Geriatr. Soc. 2022, 70, 2915–2924. [Google Scholar] [CrossRef] [PubMed]
- Suikkanen, S.; Soukkio, P.; Aartolahti, E.; Kaaria, S.; Kautiainen, H.; Hupli, M.T.; Pitkala, K.; Sipila, S.; Kukkonen-Harjula, K. Effect of 12-Month Supervised, Home-Based Physical Exercise on Functioning among Persons with Signs of Frailty: A Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2021, 102, 2283–2290. [Google Scholar] [CrossRef]
- Kao, C.H.; Chiang, S.L.; Chou, L.W.; Lin, C.H.; Lu, Y.H.; Lu, L.H.; Wang, X.M.; Lin, C.H. Validation of Vibration Exercises on Enhancing Muscle Strength and Upper Limb Functionality among Pre-Frail Community-Dwelling Older Adults. Int. J. Environ. Res. Public Health 2022, 19, 4509. [Google Scholar] [CrossRef]
- Liu, T.; Wang, C.; Sun, J.; Chen, W.; Meng, L.; Li, J.; Cao, M.; Liu, Q.; Chen, C. The Effects of an Integrated Exercise Intervention on the Attenuation of Frailty in Elderly Nursing Homes: A Cluster Randomized Controlled Trial. J. Nutr. Health Aging 2022, 26, 222–229. [Google Scholar] [CrossRef]
- Beigiene, A.; Petruseviciene, D.; Barasaite, V.; Kubilius, R.; Macijauskiene, J. Frailty and Different Exercise Interventions to Improve Gait Speed in Older Adults after Acute Coronary Syndrome. Medicina 2021, 57, 1344. [Google Scholar] [CrossRef]
- Suikkanen, S.; Soukkio, P.; Kautiainen, H.; Kaaria, S.; Hupli, M.T.; Sipila, S.; Pitkala, K.; Aartolahti, E.; Kukkonen-Harjula, K. Changes in the Severity of Frailty among Older Adults after 12 Months of Supervised Home-Based Physical Exercise: A Randomized Clinical Trial. J. Am. Med. Dir. Assoc. 2022, 23, 1717e9–1717e15. [Google Scholar] [CrossRef]
- Chen, J.; Gu, S.; Song, Y.; Ji, X.; Zeng, W.; Wang, X.; Wang, Y.; Feng, Q. The impact of cardiomotor rehabilitation on endothelial function in elderly patients with chronic heart failure. BMC Cardiovasc. Disord. 2021, 21, 524. [Google Scholar] [CrossRef]
- Yoshida, T.; Tabony, A.M.; Galvez, S.; Mitch, W.E.; Higashi, Y.; Sukhanov, S.; Delafontaine, P. Molecular mechanisms and signaling pathways of angiotensin II-induced muscle wasting: Potential therapeutic targets for cardiac cachexia. Int. J. Biochem. Cell Biol. 2013, 45, 2322–2332. [Google Scholar] [CrossRef]
- Onder, G.; Penninx, B.W.; Balkrishnan, R.; Fried, L.P.; Chaves, P.H.; Williamson, J.; Carter, C.; Di Bari, M.; Guralnik, J.M.; Pahor, M. Relation between use of angiotensin-converting enzyme inhibitors and muscle strength and physical function in older women: An observational study. Lancet 2002, 359, 926–930. [Google Scholar] [CrossRef] [PubMed]
- Cacciatore, F.; Bruzzese, G.; Vitale, D.F.; Liguori, A.; de Nigris, F.; Fiorito, C.; Infante, T.; Donatelli, F.; Minucci, P.B.; Ignarro, L.J.; et al. Effects of ACE inhibition on circulating endothelial progenitor cells, vascular damage, and oxidative stress in hypertensive patients. Eur. J. Clin. Pharmacol. 2011, 67, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Caulfield, L.; Heslop, P.; Walesby, K.E.; Sumukadas, D.; Sayer, A.A.; Witham, M.D. Effect of Angiotensin System Inhibitors on Physical Performance in Older People—A Systematic Review and Meta-Analysis. J. Am. Med. Dir. Assoc. 2021, 22, 1215.e2–1221.e2. [Google Scholar] [CrossRef] [PubMed]
- LACE Study Group; Achison, M.; Adamson, S.; Akpan, A.; Aspray, T.; Avenell, A.; Band, M.M.; Bashir, T.; Burton, L.A.; Cvoro, V.; et al. Effect of perindopril or leucine on physical performance in older people with sarcopenia: The LACE randomized controlled trial. J. Cachexia Sarcopenia Muscle 2022, 13, 858–871. [Google Scholar] [PubMed]
- Butt, J.H.; Jhund, P.S.; Belohlavek, J.; de Boer, R.A.; Chiang, C.E.; Desai, A.S.; Drozdz, J.; Hernandez, A.F.; Inzucchi, S.E.; Katova, T.; et al. Efficacy and Safety of Dapagliflozin According to Frailty in Patients with Heart Failure: A Prespecified Analysis of the DELIVER Trial. Circulation 2022, 146, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Bonora, B.M.; Cappellari, R.; Albiero, M.; Avogaro, A.; Fadini, G.P. Effects of SGLT2 Inhibitors on Circulating Stem and Progenitor Cells in Patients With Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2018, 103, 3773–3782. [Google Scholar] [CrossRef]
- Nandula, S.R.; Kundu, N.; Awal, H.B.; Brichacek, B.; Fakhri, M.; Aimalla, N.; Elzarki, A.; Amdur, R.L.; Sen, S. Role of Canagliflozin on function of CD34+ve endothelial progenitor cells (EPC) in patients with type 2 diabetes. Cardiovasc. Diabetol. 2021, 20, 44. [Google Scholar] [CrossRef]
- Mone, P.; Varzideh, F.; Jankauskas, S.S.; Pansini, A.; Lombardi, A.; Frullone, S.; Santulli, G. SGLT2 Inhibition via Empagliflozin Improves Endothelial Function and Reduces Mitochondrial Oxidative Stress: Insights from Frail Hypertensive and Diabetic Patients. Hypertension 2022, 79, 1633–1643. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).