Abstract
The seeds of Vigna genus are important food resources and there have already been many reports regarding their bioactivities. In our preliminary bioassay, the chloroform layer of methanol extracts of V. vexillata demonstrated significant anti-inflammatory bioactivity. Therefore, the present research is aimed to purify and identify the anti-inflammatory principles of V. vexillata. One new sterol (1) and two new isoflavones (2,3) were reported from the natural sources for the first time and their chemical structures were determined by the spectroscopic and mass spectrometric analyses. In addition, 37 known compounds were identified by comparison of their physical and spectroscopic data with those reported in the literature. Among the isolates, daidzein (23), abscisic acid (25), and quercetin (40) displayed the most significant inhibition of superoxide anion generation and elastase release.
1. Introduction
Phytochemicals from dietary and medicinal plants have been supposed as promising sources of potential anticancer agents with increasing anticancer evidences coupled with considerations of safety and efficacy [1,2]. Dietary and medicinal plants are importrant sources of phytochemicals for the treatment of cancers [3]. Several phytochemcials purified from natural plants, such as curcumin [4,5], epigallocatechin gallate [6], and soy isoflavones [7] had already been studied in various phases of clinical trials. The rapid population growth increase and inadequate supplies of foods had resulted in the nutrition deficiencies among the people living in the developing countries. The rigorous world food problem has presented an urgent condition for the nutritionists to investigate the choices of utilizing some less known crop seeds as additional sources of foods. With increasing interest in new food sources, the seeds of wild relatives of cultivated plants including the tribal pulses are now receiving more and more attention. The history of legumes is tied in closely with that of human civilization, appearing early in Asia, America, and Europe by thousand years ago, where they became essential for supplementing protein. Recently, there was a report regarding the chemical constituents in the wild soybean Glycine soja and their biological activity [8]. It initiated our interests to explore the chemical compositions of the wild relatives of edible legumes. For example, the seeds of Vigna vexillata are boiled and consumed by the tribal people living in the hilly region of Pune district, India [9,10]. The proximate composition, minerals, seed protein fractions, amino acids, fatty acids, and antinutritional factors of the seeds of V. vexillta were analyzed. However, information on the biochemical composition and chemical constituents of the wild relatives of grain legumes is very rare.
V. vexillata (L.) A. Rich (Fabaceae) is a perennial climbing or trailing herb belonging to the Vigna genus. This genus is widely distributed in tropical Africa, India, Indochina, Australia, Japan, Korea, China, and Taiwan. In Taiwan, the Vigna genus usually grows in grassland, margin of bush, at elevation between 1000 and 1800 m high, in the central mountain area [11]. The extracts of Vigna species have been reported to display the hypoglycemic [12,13], antihypertensive [14,15], cholesterol reduction [16], antioxidant [17,18], antibacterial [19,20], and anti-cancer bioactivities [21–23]. In the preliminary bioassay, at the tested concentration (10 μg/mL) the methanol extract, chloroform, and water fractions of V. vexillata displayed the inhibition of superoxide anion generation and elastase release with percentages of 22.06 ± 5.66%, 57.65 ± 3.69%, and 11.08 ± 5.19%; 19.35 ± 1.52%, 67.27 ± 3.53%, and 11.00 ± 3.37%, respectively (Table 1). Therefore, it is aimed to purify and identify the chemical constituents of the methanol extract V. vexillata in the present research. Furthermore, the anti-inflammatory bioactivities of these purified constituents were also examined to explore the new candidates of phytomedicinal lead compounds.

Table 1.
Inhibitory effects of crude extract and partial purified fractions of V. vexillata on superoxide anion generation and elastase release by human neutrophils in response to N-formyl-l-methionyl-phenylalanine/cytochalasin B (FMLP/CB).
2. Results and Discussion
2.1. Purification and Characterization
Air-dried and powdered whole plants of V. vexillata, including flowers and pods, were extracted with methanol under reflux and concentrated to give a dark brown syrup. The methanol extracts were suspended in water, and successively fractionated with chloroform to afford chloroform and water solubles, respectively. With the assistance of a combination of conventional chromatographic techniques, one new sterol (1) and two new isoflavones (2,3) were determined by the 1D and 2D NMR elucidations, and mass spectral analyses. In addition, 37 known compounds, including stigmast-4-en-3-one (4), stigmast-4,22-dien-3-one (5) [24], (27RS)-cycloart-28-en-3β,27-diol (6) [25], β-sitosterol (7), stigmasterol (8) [26], sitosterol cis-p-coumarate (9) [8], sitosterol trans-p-coumarate (10) [8], lupeol (11) [26], 6β-hydroxy-β-sitosterone (12) [27], sitosterol ferulate (13) [28], (20R)-22E-cholest-4-ene-3,6-dione (14) [29], methylparaben (15) [30], p-hydroxybenzoic acid (16) [30], p-hydroxybenzaldehyde (17) [30], vanillic acid (18) [30], genistein (19) [31], dehydrovomifoliol (20) [32], β-sitostenone (21) [33], 5,7,4′-trihydroxy-3′-methoxy isoflavone (22) [34], daidzein (23) [35], indole-3-carboxaldehyde (24) [36], abscisic acid (25) [37], trans-cinnamic acid (26) [38], β-sitosteryl-3-O-β-glucopyranoside (27) [39], trans-methyl p-coumarate (28) [30], salicylic acid (29) [40], tachioside (30) [41], 3-hydroxy-β-damascone (31) [42], p-hydroxyl phenethanol (32) [43], trans-p-coumaric acid (33) [44], 3,6-dihydroxy-5,6-dihydro-β-ionol (34) [45], dihydrophaseic acid (35) [46], blumenol A (36) [47], isovitexin (37) [48], daidzin (38) [49], vitexin (39) [50], and quercetin (40) [51] were characterized by comparison of their physical and spectroscopic data with those reported in the literature.
2.2. Structural Elucidation of New Compounds 1–3
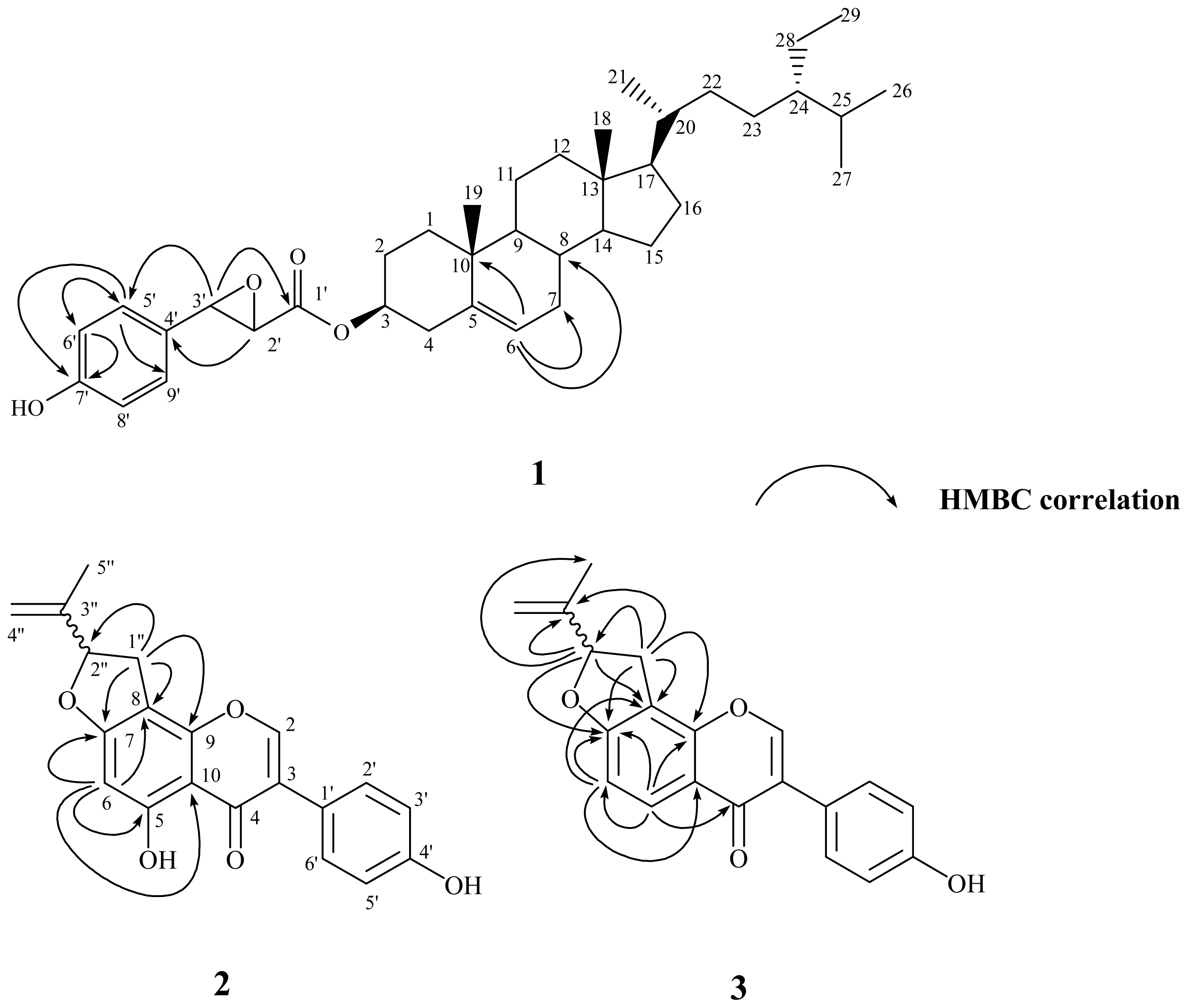
The purified solid 1 was visualized by spraying with 1% (w/v) Ce(SO4)2 in 10% (v/v) aqueous H2SO4 followed by heating at 120 °C and displayed purplish black spots on TLC plate. They also displayed positive responses against the Lieberman–Burchard test. These results suggested that compound 1 possessed steroid basic skeleton [52]. It was isolated as optically active white powder with mp 145–147 °C, and the molecular formula was established as C38H56O4 by the pseudomolecular ion peak at m/z 575.4093 ([M−H]−) in HR-ESI-MS analysis. The UV absorption maxima at 280 and 229 nm were characteristic of a benzene moiety [53]. The IR absorption bands at 3438 and 1721 cm−1 displayed the presence of hydroxyl and carbonyl groups, respectively. In its 1H-NMR spectrum, two singlets at δ 0.68 (3H, s, CH3-18) and 1.01 (3H, s, CH3-19); three doublets at δ 0.82 (3H, d, J = 6.9 Hz, CH3-26), 0.84 (3H, d, J = 6.9 Hz, CH3-27), and 0.92 (3H, d, J = 6.4 Hz, CH3-21); and one triplet at δ 0.85 (3H, t, J = 7.4 Hz, CH3-29), evidenced that this compound was a stigmastane derivative [28]. The resonances located at δ 4.66 (1H, m) and 5.36 (1H, br s, H-6) indicated that an oxygenated substitution and an olefinic functional group were presented in the stigmastane skeleton. In addition, in the aromatic region a typical set of A2B2 signals at δ 6.57 (2H, d, J = 8.3 Hz, H-6′, −8′) and 6.76 (2H, d, J = 8.3 Hz, H-5′, −9′) were attributed to a para-substituted aromatic ring. The 13C NMR spectrum of 1 also exhibited the characteristic signal for one ester carbonyl group at δ 172.0. The above-described spectroscopic characteristics of 1 were very similar to those of sitosterol trans-p-coumarate (10) [8]. The significant 1H and 13C NMR spectral differences between 1 and 10 were that the trans −COCH=CH– fragment in 10 was disappeared. Instead, there were two oxygenated proton signals at δ 3.68 (1H, d, J = 5.9 Hz, H-2′) and 4.23 (1H, d, J = 5.9 Hz, H-3′) indicating the epoxidation of the double bond and this was also confirmed by the corresponding carbon signals at δ 43.9 (C-2′) and 44.3 (C-3′) in the 13C NMR spectrum. In the HMBC spectral analysis (Figure 1), correlation peaks between H-6 (δ 5.36) and C-7 (δ 31.9), C-8 (δ 31.9), C-10 (δ 36.6), inferred that the double bond was located at C-5 and C-6. The presence of a p-hydroxyphenylglycidate moiety was deduced by the 2J, 3J-HMBC correlations through H-5′ (δ 6.76) to C-6′ (δ 115.0), C-9′ (129.1), C-7′ (154.0); H-6′ (δ 6.57) to C-5′ (δ 129.1), C-4′ (δ 131.1), C-7′ (154.0); H-3′ (δ 4.23) to C-5′ (δ 129.1), C-1′ (δ 172.0); and H-2′ (δ 3.68) to C-4′ (δ 131.1). This moiety was attached at C-3 of the stigmastane skeleton to form an ester linkage according to the chemical shift of H-3 (δ 4.66). The stereochemistry of substituent at C-3 was determined as β by the H-3 proton coupling full width at half maximum. The full assignments of 1H and 13C NMR signals were substantiated by extensive 2D NMR experiments. Therefore the chemical structure of 1 was established as shown in Figure 1 and named trivially as vignasterol A.

Figure 1.
Chemical structures and significant heteronuclear multiple bond coherence (HMBC) correlations of 1–3.
Vigvexin A (2), obtained as optically active white powder, was determined to be C20H16O5 from its HR-ESI-MS analytical data. The UV absorption maxima at 340, 296, 265 nm is typical of an isoflavone derivative [53]. The IR absorption bands at 3418 and 1656 cm−1 were in agreement with the presence of a hydroxyl and a conjugated carbonyl groups, respectively. In the 1H NMR spectrum, a downfield singlet at δ 13.26 exchangeable with D2O was assigned to be an intramolecular chelated hydroxyl group at C-5. A characteristic proton singlet δ 8.18 correlated to a carbon signal at δ 154.0 in HMQC spectrum was ascribed to H-2 of the isoflavone basic skeleton. The typical A2B2 doublets located at δ 6.90 (2H, d, J = 8.3 Hz, H-3′, 5′) and 7.43 (2H, d, J = 8.3 Hz, H-2′, 6′) indicated the presence of a para-substituted B-ring. In addition, one more aromatic singlet at δ 6.27 correlated with δ 104.0 (C-8), 106.5 (C-10), 164.7(C-5), and 167.4 (C-7) in HMBC spectrum suggested that only C-6 was not substituted in the A-ring. Another set of proton signals observed at δ 1.78 (3H, s, CH3-5″), δ 3.09 (1H, dd, J = 15.2, 8.6 Hz, H-1″), δ 3.51 (1H, dd, J = 15.2, 8.6 Hz, H-1″), δ 4.95 (1H, s, H-4″), δ 5.12 (1H, s, H-4″), and δ 5.46 (1H, t, J = 8.6 Hz, H-2″) and the 2J, 3J-HMBC correlation between H-1″ (δ 3.09 and 3.51) and C-2″ (δ 88.8), C-8 (δ 104.0), C-9 (δ 153.8), and C-7 (δ 167.4) revealed that one isopropenyl dihydrofuran fragment was attached at C-7 and C-8. Other carbon signals were correlated through the detailed 2D NMR experimental analyses and conclusively the structure of 2 was determined as shown. Due to the limited quantity of this purified compound obtained, the absolute configuration at C-2″ could not be determined and remains unknown.
Compound 3 was also assigned as an isoflavone derivative since it was found to possess characteristic UV and IR spectroscopic data as mentioned for 2. The major differences in the 1H NMR spectrum of 3 were that the downfield chelated hydroxyl singlet at δ 13.26 and the aromatic singlet at δ 6.27 in 2 were replaced by a set of two mutually coupled doublets at δ 6.95 (1H, d, J = 8.6 Hz, H-6) and 8.05 (1H, d, J = 8.6 Hz, H-5), which indicated that C-5 and C-6 were not substituted. It was further confirmed by the HMBC correlations from H-6 (δ 6.95) to C-8 (δ 114.3), C-10 (δ 119.8), and C-7 (δ 165.7); and from H-5 (δ 8.05) to C-6 (δ 109.0), C-9 (δ 154.5), C-7 (δ 165.7), and C-4 (δ 175.8). The location of the fusion of the isopropenyl dihydrofuran ring was also determined to be at C-7 and C-8 since similar HMBC correlations from H-1″ (δ 3.23 and 3.64) to C-2″ (δ 88.7), C-8 (δ 114.3), C-3″ (δ 144.6), C-9 (δ 154.5), and C-7 (δ 165.7); and from H-2″ (δ 5.52) to C-5″ (δ 17.1), C-8 (δ 114.3), C-3″ (δ 144.6), and C-7 (δ 165.7) were observed. Therefore, the chemical structure of 3 was established as displayed in Figure 1 and named as vigvexin B according to the previous convention.
2.3. Anti-Inflammatory Activity
Overexpression of neutrophils had already been regarded to display significant correlations to various human diseases, such as rheumatoid arthritis, ischemia, reperfusion injury, chronic obstructive pulmonary disease, and asthma [54–58]. In response to diverse stimuli, activated neutrophils secreted a series of cytotoxins, such as superoxide anion and elastase [59]. Thus, in infected tissues and organs it was critical to maintain superoxide anion production and elastase release in physiological conditions. Nowadays only a few available agents could directly modulate neutrophil proinflammatory responses in clinical practice. Therefore, those purified compounds isolated in sufficient quantity were evaluated for inhibition of superoxide anion generation and elastase release by human neutrophils in response to FMLP/CB (Table 2) and 1, 3, 19, 22, 23, 25, and 40 at 10 μM concentration exhibited the inhibition percentages higher than 50%. Among those examined constituents, daidzein (23), abscisic acid (25), and quercetin (40) displayed the most significant inhibition of superoxide anion generation and elastase release with IC50 values ranged from 2.66 ± 0.85 to 5.51 ± 1.07 μM, compared with the reference compound LY294002 [60], which displayed IC50 of 1.38 ± 0.22 and 1.95 ± 0.35 μM towards superoxide anion generation and elastase release, respectively. In addition, diphenyleniodonium (DPI), a NADPH oxidase inhibitor, was also used as a positive control for superoxide anion generation with IC50 of 0.93 ± 0.52 μM. Therefore, the extracts and purified principles of V. vexillata have potential to be developed as new anti-inflammatory drugs or health foods.

Table 2.
Inhibitory effects of purified samples from V. vexillata on superoxide anion generation and elastase release by human neutrophils in response to FMLP/CB.
3. Experimental Section
3.1. General
All the chemicals were purchased from Merck KGaA (Darmstadt, Germany) unless specifically indicated. Melting points of purified compounds were determined by a Fisher Scientific melting point measuring apparatus without corrections. Optical rotations were measured with the Atago AP-300 automatic polarimeter. The UV spectra were obtained on a GBC Cintra 101 UV-Vis spectrophotometer. The IR spectra were obtained on a Bruker Tensor 27 FT-IR spectrometer. The mass and high-resolution mass spectra were obtained on a VG platform electrospray mass spectrometer and a Thermo Fisher Scientific LTQ orbitrap XL mass spectrometer (San Jose, CA) operated both in the negative-ion and positive-ion modes. 1H- and 13C-NMR, COSY, NOESY, HMQC, and HMBC spectra were recorded on the Bruker AV-500 and Avance III-400 NMR spectrometers with tetramethylsilane as the internal standard. Standard pulse sequences and parameters were used for the NMR experiments and all chemical shifts were reported in parts per million (ppm, d). Column chromatography was performed on silica gels (Kieselgel 60, 70–230 mesh and 230–400 mesh, Merck KGaA). Thin layer chromatography (TLC) was conducted on precoated Kieselgel 60 F 254 plates (Merck) and the compounds were visualized by UV light or spraying with 10% (v/v) H2SO4 followed by heating at 110 °C for 10 min. High performance liquid chromatography (HPLC) was performed on a Shimadzu LC-10ATVP series pumping system equipped with a Shimadzu SPD-M10AVP diode array detector.
3.2. Plant Materials
The whole plants of V. vexillata L. A. Rich (Fabaceae) were collected in the river shores of Chingshui River in Nantou, Taiwan, in March 2006. The plant materials were authenticated by C. S. Kuoh (Department of Bioscience, National Cheng Kung University, Tainan, Taiwan). A voucher specimen (PCKuo_2006002) was deposited in the herbarium of Department of Biotechnology, National Formosa University, Yunlin, Taiwan.
3.3. Extraction and Isolation
The whole plants of V. vexillata L. (4.9 kg) were powdered and exhaustively extracted with methanol under reflux (10 L × 5 × 8 h), and the combined extracts were concentrated under reduced pressure to give a dark brown syrup (900 g). The crude extract was partitioned between chloroform and water to afford chloroform (210 g) and water extracts (690 g), respectively.
The chloroform extract was subjected to a silica gel column eluted with n-hexane and a step gradient of acetone (100:1 to 1:1) to afford 13 fractions as monitored by TLC. There were no constituents identified from fractions 1–4. Fraction 5 was subjected to silica gel column chromatography with mixture of n-hexane and acetone (50:1) to yield a mixture of 4 and 5 (3.0 mg), and 6 (1.5 mg). Fraction 6 was further resolved on a silica gel column eluted with n-hexane and a step gradient of ethyl acetate (100:1 to 1:1) to give three subfractions (6.1–6.3). Subfraction 6.1 was recrystallized with chloroform and methanol to afford a mixture of 7 and 8 (900.0 mg). Subfraction 6.2 was purified with silica gel column chromatography eluted with the solvent mixture of n-hexane and ethyl acetate (8:1) to yield 9 (8.2 mg), 10 (10.0 mg), and 11 (4.0 mg). Subfraction 6.3 was separated through silica gel column chromatography eluted with chloroform and methanol (100:1) and further purified with preparative TLC on silica gel to afford 12 (1.0 mg). Column chromatography over silica gel of the seventh fraction by the mixture of benzene and ethyl acetate (8:1) and followed by purification with preparative TLC on silica gel to result in 13 (3.0 mg) and 14 (0.5 mg). Fractions 8 and 9 were combined and further separated by repeated column chromatography over silica gel eluted with chloroform and a step gradient with acetone (300:1 to 1:1) followed by purification with preparative TLC on silica gel to yield 1 (2.6 mg), 15 (1.0 mg), 16 (0.5 mg), and 17 (3.0 mg). The tenth fraction was separated by silica gel column chromatography eluted with benzene and a step gradient with ethyl acetate (100:1 to 1:1) to afford six subfractions (10.1–10.6). Subfraction 10.1 was purified with preparative TLC by chloroform and acetone (300:1) to result in 18 (1.0 mg). Subfraction 10.2 was subjected into preparative TLC eluted by benzene and ethyl acetate (10:1) to afford 2 (2.5 mg). With similar procedures eluted with the solvent mixtures of chloroform and acetone (10:1), subfraction 10.3 was purified and resulted in 3 (6.0 mg). The fourth subfraction 10.4 was purified with preparative TLC by n-hexane and acetone (2:1) to result in 19 (3.0 mg). Subfractions 10.5 and 10.6 were resolved with preparative TLC by n-hexane and ethyl acetate (2:1) to afford 20 (3.0 mg) and 21 (1.5 mg), respectively. Fraction 11 was subjected into silica gel column chromatography with the mixing solvent of chloroform and a step gradient with methanol (100:1 to 1:1), and the resulted subfractions were purified with preparative silica gel TLC by chloroform and acetone (100:1) to yield 22 (2.0 mg), 23 (2.0 mg), and 24 (1.0 mg), respectively. Fraction 12 was separated by silica gel column chromatography eluted with chloroform and a step gradient with methanol (30:1 to 1:1) to afford four subfractions (12.1–12.4). There were no significant spots in subfractions 12.1 and 12.2 as monitored by TLC and thus no principles were identified from these fractions. Subfraction 12.3 was purified with the aid of preparative TLC by n-hexane and ethyl acetate (2:1) to yield 25 (3.0 mg) and 26 (4.0 mg). With the similar procedures, 27 (20.0 mg) was characterized from the subfraction 12.4. The last fraction of chloroform extract was separated by silica gel column chromatography eluted with chloroform and methanol (20:1) to yield 16 (0.5 mg).
The water extract was applied to a reversed-phase Diaion HP-20 column eluted with water and methanol gradients to afford 14 fractions as monitored by C-18 TLC, however, no constituents were identified from fractions 1–3. Fractions 4 and 5 were combined and subjected into C-18 column chromatography eluted with water and methanol gradients and further recrystallization with chloroform-methanol to yield 28 (6.0 mg), 29 (2.5 mg), and 30 (5.0 mg), respectively. Fractions 6–8 were merged and purified by C-18 column chromatography eluted with water and methanol gradients followed by preparative TLC eluted with chloroform and methanol (20:1) on the resulted subfractions to afford 31 (20.0 mg), 32 (1.5 mg), 33 (2.0 mg), and 34 (15.0 mg), respectively. Fractions 7–9 were merged and purified by silica gel column chromatography eluted with chloroform and methanol (50:1) and further recrystallization of methanol of the subfractions to result in 35 (3.0 mg) and 36 (2.0 mg). Fractions 12 and 13 were combined and separated by silica gel column chromatography eluted with chloroform and methanol (50:1) to afford four subfractions (12.1–12.4). Subfraction 12.1 was recrystallized with chloroform and methanol to yield 35 (2.0 mg). Subfraction 12.2 was purified by reversed-phase HPLC with a Supelco Discovery® HS C-18 (250 × 4.6 mm, 5μm) column eluted with 0.5 mL/min of MeOH-H2O (40:60) to give 37 (5.0 mg) and 38 (4.0 mg). Subfraction 12.3 was recrystallized with chloroform and methanol to yield 39 (6.0 mg). Fraction 14 was resolved by silica gel column chromatography eluted with chloroform and methanol (50:1) and further recrystallization of methanol of the subfraction to result in 40 (30.0 mg).
3.3.1. Vignasterol A (1)
White powder, mp 145–147 °C (CHCl3); [α]D25 −300.0 (c 0.1, CHCl3). UV (MeOH) λmax (log ɛ): 280 (2.29), 229 (2.72, sh) nm. IR (Neat) νmax: 3438, 2956, 2868, 1721, 1642, 1516, 1461, 1377, 1261, 1207, 1172, 1103, 1063, 1024 cm−1. ESI-MS (rel. int. %): m/z 575 ([M−H]−, 47), 559 (100). HR-ESI-MS: m/z 575.4093 [M−H]− (calcd for C38H55O4, 575.4095). 1H-NMR (CDCl3, 500 MHz): δ 6.76 (2H, d, J = 8.3 Hz, H-5′, −9′), 6.57 (2H, d, J = 8.3 Hz, H-6′, −8′), 5.36 (1H, br s, H-6), 4.66 (1H, m, H-3), 4.23 (1H, d, J = 5.9 Hz, H-3′), 3.68 (1H, d, J = 5.9 Hz, H-2′), 2.34 (2H, m, H-4), 2.00 (2H, m, H-7, −12), 1.85–1.87 (2H, m, H-1, −16), 1.70 (1H, m, H-25), 1.57 (3H, m, H-2, −7, −15), 1.46 (2H, m, H-8, −11), 1.36 (2H, m, H-20, −22), 1.26 (2H, m, H-16, −28), 1.18 (2H, m, H-12, −23), 1.07 (1H, m, H-1), 1.01 (1H, m, H-22), 1.01 (3H, s, CH3-19), 0.92 (3H, d, J = 6.4 Hz, CH3-21), 0.85 (3H, t, J = 7.4 Hz, CH3-29), 0.84 (3H, d, J = 6.9 Hz, CH3-27), 0.83 (1H, m, H-28), 0.82 (3H, d, J = 6.9 Hz, CH3-26), 0.68 (3H, s, CH3-18). 13C-NMR (CDCl3, 100 MHz): δ 172.0 (C-1′), 154.0 (C-7′), 139.6 (C-5), 131.1 (C-4′), 129.1 (C-5′, −9′), 122.8 (C-6), 115.0 (C-6′, −8′), 74.7 (C-3), 56.7 (C-14), 56.1 (C-17), 50.0 (C-9), 45.9 (C-24), 44.3 (C-3′), 43.9 (C-2′), 42.3 (C-13), 39.7 (C-12), 38.1 (C-4), 37.0 (C-1), 36.6 (C-10), 36.2 (C-20), 34.0 (C-22), 31.9 (C-7), 31.9 (C-8), 29.2 (C-25), 28.2 (C-16), 27.8 (C-2), 26.2 (C-23), 24.3 (C-15), 23.1 (C-28), 21.0 (C-11), 19.8 (C-26), 19.3 (C-19), 19.0 (C-27), 18.8 (C-21), 12.0 (C-29), 11.9 (C-18).
3.3.2. Vigvexin A (2)
White powder, mp > 300 °C (MeOH); [α]D25 −100.0 (c 0.2, MeOH). UV (MeOH) λmax (log ɛ): 340 (1.86), 296 (sh, 2.28), 265 (2.84), 222 (sh, 2.52), 213 (2.53) nm. IR (Neat) νmax: 3418, 2928, 1656, 1614, 1515, 1480, 1433, 1402, 1320, 1286, 1251, 1205, 1169, 1131, 1065, 1027 cm−1. ESI-MS (rel. int. %): m/z 335 ([M−H]−, 100), 321 (76), 305 (88). HR-ESI-MS: m/z 335.0905 [M−H]− (calcd for C20H15O5, 335.0914). 1H-NMR (Acetone-d6, 400 MHz): δ 13.26 (1H, s, OH), 8.77 (1H, s, OH), 8.18 (1H, s, H-2), 7.43 (2H, d, J = 8.3 Hz, H-2′, −6′), 6.90 (2H, d, J = 8.3 Hz, H-3′, −5′), 6.27 (1H, s, H-6), 5.46 (1H, t, J = 8.6 Hz, H-2″), 5.12 (1H, s, H-4″), 4.95 (1H, s, H-4″), 3.51 (1H, dd, J = 15.2, 8.6 Hz, H-1″), 3.09 (1H, dd, J = 15.2, 8.6 Hz, H-1″), 1.78 (3H, s, CH3-5″). 13C-NMR (Acetone-d6, 100 MHz): δ 181.8 (C-4), 167.4 (C-7), 164.7 (C-5), 158.6 (C-4′), 154.0 (C-2), 153.8 (C-9), 144.6 (C-3″), 131.3 (C-2′, −6′), 124.2 (C-3), 122.9 (C-1′), 116.0 (C-3′, −5′), 112.9 (C-4″), 106.5 (C-10), 104.0 (C-8), 94.5 (C-6), 88.8 (C-2″), 31.3 (C-1″), 17.1 (C-5″).
3.3.3. Vigvexin B (3)
White plates, mp 227–229 °C (MeOH); [α]D25 −80.0 (c 0.2, MeOH). UV (MeOH) λmax (log ɛ): 307 (2.25), 250 (2.71), 243 (sh, 2.67), 216 (2.38) nm. IR (Neat) νmax: 3292, 2928, 2858, 1627, 1594, 1515, 1449, 1389, 1318, 1270, 1208, 1175, 1105, 1066, 1021 cm-1. ESI-MS (rel. int. %): m/z 343 ([M+Na]+, 28), 321 ([M+H]+, 100). HR-ESI-MS: m/z 343.0944 [M+Na]+ (calcd for C20H16O4Na, 343.0946), 321.1128 [M+H]+ (calcd for C20H17O4, 321.1127). 1H-NMR (Acetone-d6, 400 MHz): δ 8.17 (1H, s, H-2), 8.05 (1H, d, J = 8.6 Hz, H-5), 7.46 (2H, d, J = 8.6 Hz, H-2′, −6′), 6.95 (1H, d, J = 8.6 Hz, H-6), 6.88 (2H, d, J = 8.6 Hz, H-3′, −5′), 5.52 (1H, t, J = 7.8 Hz, H-2″), 5.15 (1H, s, H-4″), 4.97 (1H, s, H-4″), 3.64 (1H, dd, J = 16.0, 7.8 Hz, H-1″), 3.23 (1H, dd, J = 16.0, 7.8 Hz, H-1″), 1.81 (3H, s, CH3-5″). 13C-NMR (Acetone-d6, 100 MHz): δ 175.8 (C-4), 165.7 (C-7), 158.3 (C-4′), 154.5 (C-9), 153.0 (C-2), 144.6 (C-3″), 131.2 (C-2′, −6′), 128.4 (C-5), 125.3 (C-3), 124.3 (C-1′), 119.8 (C-10), 115.9 (C-3′, −5′), 114.3 (C-8), 112.8 (C-4″), 109.0 (C-6), 88.7 (C-2″), 32.0 (C-1″), 17.1 (C-5″).
3.4. Anti-Inflammatory Activity
3.4.1. Preparation of Human Neutrophils
Neutrophils were isolated with a standard method of dextran sedimentation prior to centrifugation in a Ficoll Hypaque gradient and hypotonic lysis of erythrocytes. Blood was drawn from healthy human donors (20–30 years old) by venipuncture into heparin-coated vacutainer tubes, using a protocol approved by the institutional review board at Chang Gung Memorial Hospital. Blood samples were mixed gently with an equal volume of 3% dextran solution. The leukocyte-rich plasma was collected after sedimentation of the red cells for 30 min at room temperature. The leukocyte-rich plasma was transferred on top of 20 mL Ficoll solution (1.077 g/mL) and spun down at 400 g for 40 min at 20 °C. The granulocyte/erythrocyte pellets were resuspended in ice-cold 0.2% NaCl to lyse erythrocytes. After 30 sec, the same volume of 1.6% NaCl solution was added to reconstitute the isotonic condition. Purified neutrophils were pelleted and then resuspended in a calcium (Ca2+)-free Hank’s balanced salt solution (HBSS) buffer at pH 7.4, and were maintained at 4 °C before use.
3.4.2. Measurement of Superoxide Anion Generation
The assay of the generation of superoxide anion was based on the SOD-inhibitable reduction of ferricytochrome c [59]. In brief, after supplementation with 0.5 mg/mL ferricytochrome c and 1 mM Ca2+, neutrophils (6 × 105 cells/mL) were equilibrated at 37 °C for 2 min and incubated with drugs or an equal volume of vehicle (0.1% DMSO, negative control) for 5 min. Cells were activated with 100 nM FMLP during the preincubation of 1 μg/mL cytochalasin B (FMLP/CB) for 3 min. Changes in the absorbance with a reduction in ferricytochrome c at 550 nm were continuously monitored in a double-beam, six-cell positioner spectrophotometer with constant stirring (Hitachi U-3010, Tokyo, Japan). Calculations were based on differences in the reactions with and without SOD (100 U/mL) divided by the extinction coefficient for the reduction of ferricytochrome c (ɛ = 21.1/mM/10 mm).
3.4.3. Measurement of Elastase Release
Degranulation of azurophilic granules was determined by elastase release as described previously [59]. Experiments were performed using MeO-Suc-Ala-Ala-Pro-Val-p-nitroanilide as the elastase substrate. Briefly, after supplementation with MeO-Suc-Ala-Ala-Pro-Val-p-nitroanilide (100 μM), neutrophils (6 × 105/mL) were equilibrated at 37 °C for 2 min and incubated with drugs or an equal volume of vehicle (0.1% DMSO, negative control) for 5 min. Cells were activated by 100 nM FMLP and 0.5 μg/mL cytochalasin B, and changes in absorbance at 405 nm were continuously monitored to assay elastase release. The results were expressed as the percent of elastase release in the FMLP/CB-activated, drug-free control system.
3.5. Statistical Analysis
Results were expressed as mean ± S.D. Computation of 50% inhibitory concentration (IC50) was computer-assisted (PHARM/PCS v.4.2). Statistical comparisons were made between groups using Student’s t test. Values of P less than 0.05 were considered to be statistically significant.
4. Conclusions
The present investigation on the methanol extracts of V. vexillata resulted in the isolation and characterization of totally three new principles (1–3) along with 37 known constituents (4–40). In the screening of their bioactivity, three compounds, daidzein (23), abscisic acid (25), and quercetin (40) from the active chloroform fraction demonstrated significant anti-inflammatory potentials compared with the PI3K inhibitor LY294002 and a NADPH oxidase inhibitor DPI. It is well known that the phosphatidylinositol-3-kinase (PI3K)/protein kinase B (AKT) pathway plays an important role in neutrophil activation. The NADPH oxidase is also important in the inflammatory mechanism. Therefore, the purified principles of V. vexillata were potential to be developed as new anti-inflammatory drugs through the inhibition of PI3K or NADPH oxidase. This is the first report of complete chemical compositions of V. vexillata and it would provide the comprehensive knowledge related to the further discovery of phytochemical lead compounds from natural food sources.
Acknowledgments
The authors are thankful to the National Science Council, Taiwan, ROC, for financial support of the present research. We also thank J. R. Wu, National Cheng Kung University, for recording the NMR spectra.
- Conflict of InterestThe authors have no conflict of interest to report.
References
- Shu, L.; Cheung, K.L.; Khor, T.O.; Chen, C.; Kong, A.N. Phytochemicals: Cancer chemoprevention and suppression of tumor onset and metastasis. Cancer Metastasis Rev 2010, 29, 483–502. [Google Scholar]
- Johnson, S.M.; Wang, X.; Evers, B.M. Triptolide inhibits proliferation and migration of colon cancer cells by inhibition of cell cycle regulators and cytokine receptors. J. Surg. Res 2011, 168, 197–205. [Google Scholar]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The influence of natural products upon drug discovery. Nat. Prod. Rep 2000, 17, 215–234. [Google Scholar]
- Aggarwal, B.B.; Kumar, A.; Bharti, A.C. Anticancer potential of curcumin: Preclinical and clinical studies. Anticancer Res 2003, 23, 363–398. [Google Scholar]
- Bar-Sela, G.; Epelbaum, R.; Schaffer, M. Curcumin as an anti-cancer agent: Review of the gap between basic and clinical applications. Curr. Med. Chem 2010, 17, 190–197. [Google Scholar]
- Na, H.K.; Kim, E.H.; Jung, J.H.; Lee, H.H.; Hyun, J.W.; Surh, Y.J. (−)-Epigallocatechin gallate induces Nrf 2-mediated antioxidant enzyme expression via activation of PI3K and ERK in human mammary epithelial cells. Arch. Biochem. Biophys. 2008, 476, 171–177. [Google Scholar]
- Swami, S.; Krishnan, A.V.; Moreno, J.; Bhattacharyya, R.S.; Gardner, C.; Brooks, J.D.; Peehl, D.M.; Feldman, D. Inhibition of prostaglandin synthesis and actions by genistein in human prostate cancer cells and by soy isoflavones in prostate cancer patients. Int. J. Cancer 2009, 124, 2050–2059. [Google Scholar]
- Zhou, Y.Y.; Luo, S.H.; Yi, T.S.; Li, C.H.; Luo, Q.; Hua, J.; Liu, Y.; Li, S.H. Secondary metabolites from Glycine soja and their growth inhibitory effect against Spodoptera litura. J. Agric. Food Chem 2011, 59, 6004–6010. [Google Scholar]
- Gunjatkar, N.; Vartak, V.D. Enumeration of wild legumes from Pune district, Maharashtra State. J. Econ. Tax. Bot 1982, 3, 1–9. [Google Scholar]
- Siddhuraju, P.; Vijayakumari, K.; Janardhanan, K. Chemical analysis and nutritional assessment of the less known pulses, Vigna aconitifolia (Jacq.) Marechal and Vigna vexillata (L.) A. Rich. Plant Food Hum. Nutr 1994, 45, 103–111. [Google Scholar]
- Huang, T.C.; Ohashi, H. Flora of Taiwan, 2nd ed; Editorial Committee of Flora of Taiwan: Taipei, Taiwan, 1993; Volume 3, p. 393. [Google Scholar]
- Itoh, T.; Kita, N.; Kurokawa, Y.; Kobayashi, M.; Horio, F.; Furuichi, Y. Suppressive effect of a hot water extract of adzuki beans (Vigna angularis) on hyperglycemia after sucrose loading in mice and diabetic rats. Biosci. Biotechnol. Biochem 2004, 68, 2421–2426. [Google Scholar]
- Itoh, T.; Kobayashi, M.; Horio, F.; Furuichi, Y. Hypoglycemic effect of hot-water extract of adzuki beans (Vigna angularis) in spontaneously diabetic KK-Ay mice. Nutrition 2009, 25, 134–141. [Google Scholar]
- Mukai, Y.; Sato, S. Polyphenol-Containing adzuki bean (Vigna angularis) extract attenuates blood pressure elevation and modulates nitric oxide synthase and caveolin-1 expressions in rats with hypertension. Nutr. Metab. Cardiovasc. Dis 2009, 19, 491–497. [Google Scholar]
- Mukai, Y.; Sato, S. Polyphenol-containing azuki bean (Vigna angularis) seed coats attenuate vascular oxidative stress and inflammation in spontaneously hypertensive rats. J. Nutr. Biochem 2011, 22, 16–21. [Google Scholar]
- Itoh, T.; Furuichi, Y. Lowering serum cholesterol level by feeding a 40% ethanol-eluted fraction from HP-20 resin treated with hot water extract of adzuki beans (Vigna angularis) to rats fed a high-fat cholesterol diet. Nutrition 2009, 25, 318–321. [Google Scholar]
- Ariga, T.; Koshiyama, I.; Fukushima, D. Antioxidative properties of procyanidins B-1 and B-3 from adzuki beans in aqueous systems. Agric. Biol. Chem 1988, 52, 2717–2722. [Google Scholar]
- Doblado, R.; Zielinski, H.; Piskula, M.; Kozlowska, H.; Muñoz, R.; Frías, J.; Vidal-Valverde, C. Effect of processing on the antioxidant vitamins and antioxidant capacity of Vigna sinensis var. carilla. J. Agric. Food Chem 2005, 53, 1215–1222. [Google Scholar]
- Hori, Y.; Sato, S.; Hatai, A. Antibacterial activity of plant extracts from adzuki beans (Vigna angularis) in vitro. Phytother. Res 2006, 20, 162–164. [Google Scholar]
- Franco, O.L.; Murad, A.M.; Leite, J.R.; Mendes, P.A.M.; Prates, M.V.; Bloch, C., Jr. Identification of a cowpea γ-thionin with bactericidal activity. FEBS J. 2006, 273, 3489–3497. [Google Scholar]
- Itoh, T.; Itoh, Y.; Mizutani, M.; Fujishiro, K.; Furuichi, Y.; Komiya, T.; Hibasami, H. Hot-water extracts from adzuki beans (Vigna angularis) suppress not only proliferation of KATO III cells in culture but also benzo(a)pyrene-induced tumorigenesis in mouse forestomatch. J. Nutr. Sci. Vitaminol 2004, 50, 295–299. [Google Scholar]
- Itoh, T.; Furuichi, Y. Hot-Water extracts from adzuki beans (Vigna angularis) stimulated not only melanogenesis in cultured mouse B16 melanoma cells but also pigmentation of hair color in C3H mice. Biosci. Biotechnol. Biochem 2005, 69, 873–882. [Google Scholar]
- Joanitii, G.A.; Azevedo, R.B.; Freitas, S.M. Apoptosis and lysosome membrane permeabilization induction on breast cancer cells by an anticarcinogenic Bowman-Birk protease inhibitor from Vigna unguiculata seeds. Cancer Lett 2010, 29, 73–81. [Google Scholar]
- Ambrus, G.; Ilköy, E.; Jekkel, A.; Horváth, G.; Böcskei, Z. Microbial transformation of β-sitosterol and stigmasterol into 26-oxygenated derivatives. Steroids 1995, 60, 621–625. [Google Scholar]
- Kitajima, J.; Kimizuka, K.; Tanaka, Y. New sterols and triterpenoids of Ficus pumila fruit. Chem. Pharm. Bull 1998, 46, 1408–1411. [Google Scholar]
- Kuo, Y.H.; Li, Y.C. Constituents of the bark of Ficus microcarpa L.f. J. Chin. Chem. Soc 1997, 44, 321–325. [Google Scholar]
- Lin, W.Y.; Yen, M.H.; Teng, C.M.; Tsai, I.L.; Chen, I.S. Cerebrosides from the rhizomes of Gynura japonica. J. Chin. Chem. Soc 2004, 51, 1429–1434. [Google Scholar]
- Yasukawa, K.; Akihisa, T.; Kimura, Y.; Tamura, T.; Takido, M. Inhibitory effect of cycloartenol ferulate, a component of rice bran, on tumor promotion in two-stage carcinogenesis in mouse skin. Biol. Pharm. Bull 1998, 21, 1072–1076. [Google Scholar]
- Kontiza, I.; Abatis, D.; Malakate, K.; Vagias, C.; Roussis, V. 3-Keto steroids from the marine organisms Dendrophyllia cornigera and Cymodocea nodosa. Steroids 2006, 71, 177–181. [Google Scholar]
- Chen, C.Y.; Chang, F.R.; Teng, C.M.; Wu, Y.C. Cheritamine, a new N-fatty acyl tryptamine and other constituents from the stem of Annona cherimola. J. Chin. Chem. Soc 1999, 46, 77–86. [Google Scholar]
- Garcez, W.S.; Martins, D.; Garcez, F.R.; Marques, M.R.; Pereira, A.A.; Oliveira, L.A.; Rondon, J.N.; Peruca, A.D. Effect of spores of saprophytic fungi on phytoalexin accumulation in seeds of frog-eye leaf spot and stem canker-resistant and -susceptible soybean (Glycine max L.) cultivars. J. Agric. Food Chem 2000, 48, 3662–3665. [Google Scholar]
- Serra, S.; Barakat, A.; Fuganti, C. Chemoenzymatic resolution of cis- and trans-3,6-dihydroxy-α-ionone. Synthesis of the enantiomeric forms of dehydrovomifoliol and 8,9-dehydrotheaspirone. Tetrahedron Asymmetry 2007, 18, 2573–2580. [Google Scholar]
- Jones, J.B.; Baskevitch, N. Steroids and steroidases XX (1). Aggregation in aqueous solution of steroids with stigmastane type C-17 side chains and its influence on their enzymic transformations. Steroids 1973, 22, 525–538. [Google Scholar]
- Hosny, M.; Rosazza, J.P.N. Microbial hydroxylation and methylation of genistein by Streptomycetes. J. Nat. Prod 1999, 62, 1609–1612. [Google Scholar]
- Lin, Y.L.; Tsai, W.J.; Chen, I.S.; Kuo, Y.H. Chemical constituents from Mucuna membranacea. J. Chin. Chem. Soc 1998, 45, 213–217. [Google Scholar]
- Yang, S.W.; Cordell, G.A. Metabolism studies of indole derivatives using a staurosporine producer, Streptomyces staurosporeus. J. Nat. Prod 1997, 60, 44–48. [Google Scholar]
- Smith, T.R.; Clark, A.J.; Clarkson, G.J.; Taylor, P.C.; Marsh, A. Concise enantioselective synthesis of abscisic acid and a new analogue. Org. Biomol. Chem 2006, 4, 4186–4192. [Google Scholar]
- Hsieh, T.J.; Chang, F.R.; Wu, Y.C. The constituents of Cananga odorata. J. Chin. Chem. Soc 1999, 46, 607–611. [Google Scholar]
- Faizi, S.; Ali, M.; Saleem, R.; Irfanullah; Bibi, S. Complete 1H and 13C NMR assignments of stigma-5-en-3-O-β-glucoside and its acetyl derivative. Magn. Reson. Chem. 2001, 39, 399–405. [Google Scholar]
- Kundo, M. The nuclear magnetic resonance study of several O-disubstituted benzenes. Bull. Chem. Soc. Jpn 1972, 45, 2790–2793. [Google Scholar]
- Zhong, X.N.; Otsuka, H.; Ide, T.; Hirata, E.; Takeda, Y. Hydroquinone diglycoside acyl esters from the leaves of Myrsine seguinii. Phytochemistry 1999, 52, 923–927. [Google Scholar]
- Skouroumounis, G.K.; Sefton, M.A. Acid-catalyzed hydrolysis of alcohols and their β-d-glucopyranosides. J. Agric. Food Chem 2000, 48, 2033–2039. [Google Scholar]
- Xu, Q.M.; Liu, Y.L.; Li, X.R.; Feng, Y.L.; Yang, S.L. Two new phenylglycol derivatives isolated from Syringa reticulata var. mandshurica and their antifungal activities. Chem. Pharm. Bull 2009, 57, 863–866. [Google Scholar]
- Chiang, Y.M.; Liu, H.K.; Lo, J.M.; Chien, S.C.; Chan, Y.F.; Lee, T.H.; Su, J.K.; Kuo, Y.H. Cytotoxic constituents of leaves of Calocedrus formosana. J. Chin. Chem. Soc 2003, 50, 161–166. [Google Scholar]
- Yu, Q.; Otsuka, H.; Hirata, E.; Shinzato, T.; Takeda, Y. Turpinionosides A–E: Megastigmane glucosides from leaves of Turpinia ternata Nakai. Chem. Pharm. Bull 2002, 50, 640–644. [Google Scholar]
- Masamune, T.; Anetai, M.; Fukuzawa, A.; Takasugi, M.; Matsue, H.; Kobayashi, K.; Ueno, S.; Katsui, N. Glycinoeclepins, natural hatching stimuli for the soybean cyst nematode, Heterodera glycines. I. Isolation. Bull. Chem. Soc. Jpn 1987, 60, 981–999. [Google Scholar]
- Ito, N.; Etoh, T.; Hagiwara, H.; Kato, M. Novel synthesis of degradation products of carotenoids, megastigmatrienone analogues and blumenol-A. J. Chem. Soc. Perkin Trans 1997, 1571–1579. [Google Scholar]
- Ling, T.J.; Ling, W.W.; Chen, Y.J.; Wan, X.C.; Xia, T.; Du, X.F.; Zhang, Z.Z. Antiseptic activity and phenolic constituents of the aerial parts of Vitex negundo var. cannabifolia. Molecules 2010, 15, 8469–8477. [Google Scholar]
- Weis, M.; Lim, E.K.; Bruce, N.; Bowles, D. Regioselective glucosylation of aromatic compounds: Screening of a recombinant glycosyltransferase library to identify biocatalysts. Angew. Chem. Int. Ed 2006, 45, 3534–3538. [Google Scholar]
- Wang, P.H.; Lee, S.S. Polar chemical constituents from Phoebe formosana. J. Chin. Chem. Soc 1999, 46, 215–219. [Google Scholar]
- Hua, Y.; Wang, H.Q. Chemical components of Anaphalis sinica Hance. J. Chin. Chem. Soc 2004, 51, 409–415. [Google Scholar]
- Srivastava, A.; Shukla, Y.N. Aryl esters and a coumarin from Aygyreia speciosa. Indian J. Chem. Sect 1998, 37B, 192–194. [Google Scholar]
- Scott, A.I. Interpretation Ultraviolet Spectra of Natural Products, 2nd ed; Pergamon Press: New York, NY, USA; p. 1964.
- Malech, H.L.; Gallin, J.I. Current concepts: Immunology neutrophils in human diseases. N. Engl. J. Med 1987, 317, 687–694. [Google Scholar]
- Witko-Sarsat, V.; Rieu, P.; Descamps-Latscha, B.; Lesavre, P.; Halbwachs-Mecarelli, L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Invest 2000, 80, 617–653. [Google Scholar]
- Okajima, K.; Harada, N.; Uchiba, M. Ranitidine reduces ischemia/reperfusion-induced liver injury in rats by inhibiting neutrophil activation. J. Pharmacol. Exp. Ther 2002, 301, 1157–1165. [Google Scholar]
- Ennis, M. Neutrophils in asthma pathophysiology. Curr. Allergy Asthma Rep 2003, 3, 159–165. [Google Scholar]
- Vinten-Johansen, J. Involvement of neutrophils in the pathogenesis of lethal myocardial reperfusion Injury. Cardiovasc. Res 2004, 61, 481–497. [Google Scholar]
- Hwang, T.L.; Li, G.L.; Lan, Y.H.; Chia, Y.C.; Shieh, P.W.; Wu, Y.H.; Wu, Y.C. Potent inhibition of superoxide anion production in activated human neutrophils by isopedicin, a bioactive component of the Chinese medicinal herb Fissistigma oldhamii. Free Radic. Biol. Med 2009, 46, 520–528. [Google Scholar]
- Yang, M.L.; Kuo, P.C.; Hwang, T.L.; Chiou, W.F.; Qian, K.; Lai, C.Y.; Lee, K.H.; Wu, T.S. Synthesis, in vitro anti-inflammatory and cytotoxic evaluation, and mechanism of action studies of 1-benzoyl-β-carboline and 1-benzoyl-3-carboxy-β-carboline derivatives. Bioorg. Med. Chem 2011, 19, 1674–1682. [Google Scholar]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).