Abstract
This review examines the historical development, ethnopharmacology, traditional applications, phytochemistry, and pharmacological attributes of Atractylodis Rhizoma (AR). Data were collected from a range of electronic databases, academic libraries, and classical literature. In China, AR is highly valued for its medicinal properties. Research has identified 327 compounds, including sesquiterpenes, triterpenes, flavonoids, and phenolics, which contribute to its diverse pharmacological activities, such as antimicrobial, anti-inflammatory, antioxidant, hepatoprotective, and neuroprotective effects. AR is particularly effective in treating modern gastrointestinal disorders and influenza. As a traditional herb with a rich historical background, AR exhibits significant therapeutic potential. This review aims to correlate its active components with its primary therapeutic effects and highlight existing research gaps. Current studies primarily focus on extraction methods and pharmacodynamics. Future research should employ multi-omics and molecular biology techniques to further elucidate active components and their targets, while also addressing the challenge of low bioavailability.
1. Introduction
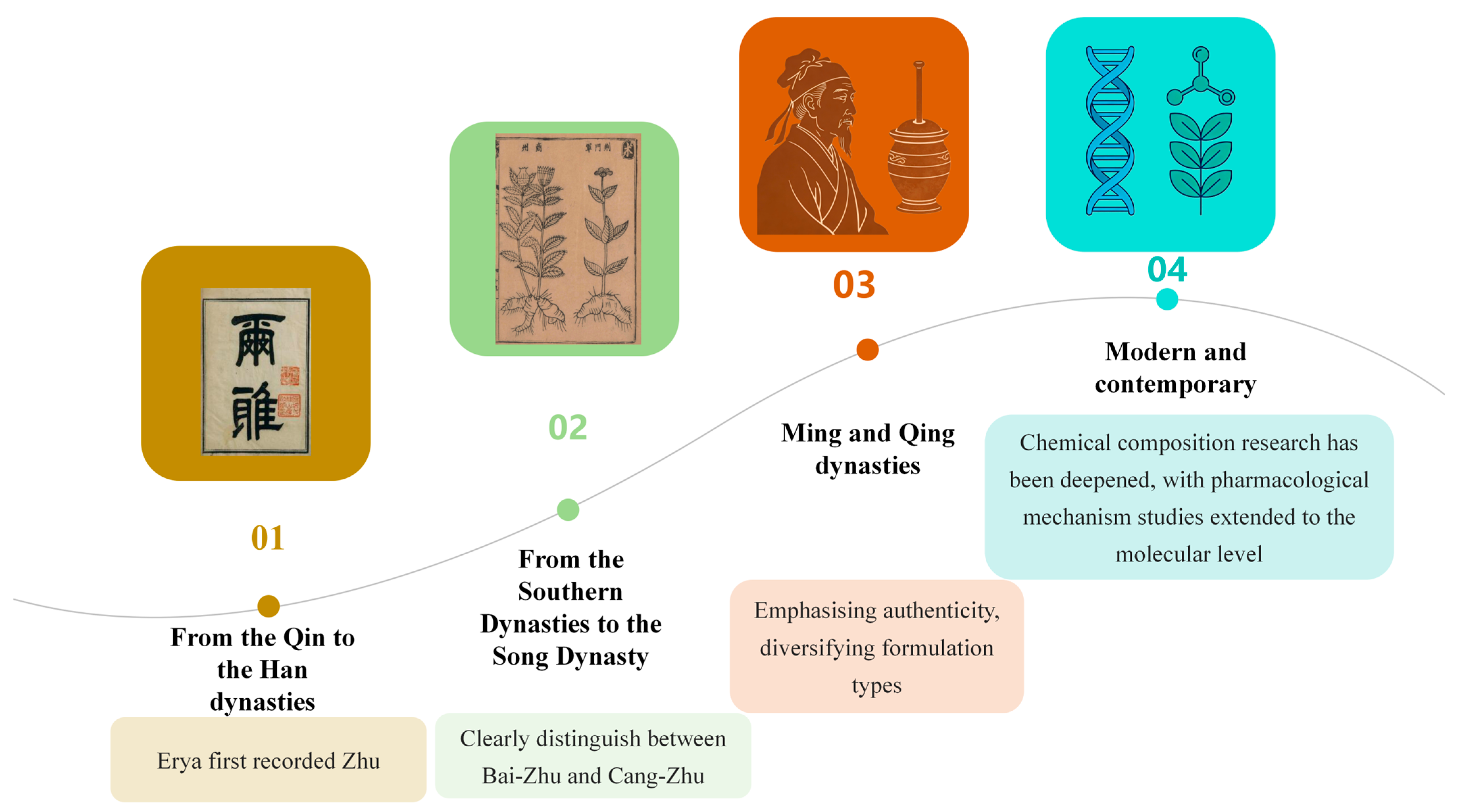
Atractylodis Rhizoma (AR) is a pivotal herb in Traditional Chinese Medicine [1], esteemed for its effectiveness in alleviating dampness, fortifying the spleen, dispelling wind and cold, and enhancing vision (Figure 1). Its historical utilization can be traced back to the Erya (1st–2nd century AD), which documented its properties and nomenclature [2]. Shennong’s Herbal Classic classified it as a “superior” herb for the treatment of wind-cold-damp arthralgia, convulsions, and jaundice [3]. However, herbal texts from the Han and Wei dynasties and earlier periods did not differentiate between AR (Cang-Zhu) and Atractylodis Macrocephalae Rhizoma (Bai-Zhu) [4], referring to both simply as Zhu. It was only after the Ben Cao Jing Ji Zhu that these terms began to be used distinctively. Throughout history, numerous classic formulas have employed Cang-Zhu and Bai-Zhu, such as Wan Dai Decoction [5], Ling Gui Zhu Gan Decoction [6], Ping Wei Powder [7], and Wei Ling Decoction [8]. To prevent confusion and misuse, conducting thorough herbal verification is particularly crucial. The Atractylodis genus primarily includes A. carlinoides (Hand. -Mazz.) Kitam., A. coreana (Nakai) Kitam., A. japonica Koidz. ex Kitam., A. lancea (Thunb.) DC., A. chinensis (DC.) Koidz., and A. macrocephala Koidz., primarily distributed throughout eastern Asia. A. lancea (Thunb.) DC. (Mao-Cang-Zhu) or A. chinensis (DC.) Koidz (Bei-Cang-Zhu) are the two sources of AR, distributed in Shandong, Jiangsu, Zhejiang, Hubei, Sichuan, and other regions of China [9].
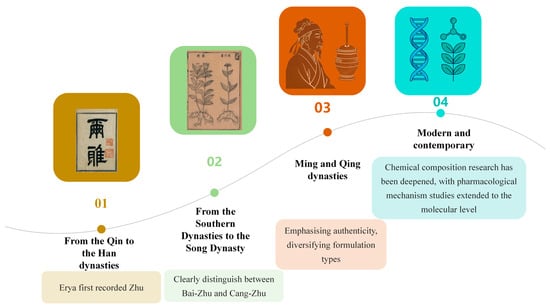
Figure 1.
The Historical Application of Atractylodis Rhizoma. Note: Different colors represent different periods: yellow represents the period when the term first originated; green represents the period when species began to be distinguished; orange represents the period of widespread use; and blue represents the period of modern research.
Significant progress has been made in the isolation, identification, and investigation of potential pharmacological activities of AR compounds [10,11]. While extensive research exists on A. lancea, reports on A. chinensis remain scarce. Given the expanding demand for AR, it is gradually gaining greater attention. A review article published in 2021 systematically summarized the botanical characteristics, traditional uses, phytochemical composition, pharmacological effects, and quality control standards of AR [12]. However, comprehensive research on the structural composition of compounds in AR and their potential mechanisms of action remains insufficient to date.
Against this backdrop, a comprehensive review and analysis of AR is urgently needed. This study begins with the traditional usage of AR, integrates findings from modern technological discoveries, and comprehensively examines the connections and distinctions between traditional applications and contemporary uses. It aims to provide a reference and basis for AR research, offering insights for innovative studies and comprehensive collation of similar Chinese herbal medicines.
2. Materials and Methods
2.1. Search Strategy
A comprehensive online literature search was conducted across multiple databases, including ScienceDirect, Google Scholar, PubMed, Web of Science, CNKI, WFO, MPNS, the Changchun University of Chinese Medicine Library collections, and SciFinder, covering the period from 1996 to 2026. For the methodology outlined in reference [13], the search employed the keywords “Atractylodis Rhizoma” in conjunction with “phytochemistry”, “pharmacology”, or “toxicity”. The references of all retrieved articles were meticulously reviewed to ensure the inclusion of pertinent literature. Uniform selection criteria were consistently applied across all databases, and duplicate studies were systematically eliminated through a two-step process involving automated detection via Zotero 7, followed by manual cross-verification. Additionally, traditional and historical applications were corroborated through a systematic analysis of classical Chinese medical texts. The search strategy comprehensively covered medicinal and dietary records using the nomenclature “Cang Zhu,” “Zhu,” “Ji,” “Shan Ji,” “Tian Su,” and “Yang Bao.” Relevant prescriptions were compiled after the removal of duplicates through cross-database verification.
2.2. Selection Criteria
The inclusion criteria for this study are delineated as follows: the primary focus is on AR, including its extracts and constituent compounds; the investigation encompasses diseases and physiological processes influenced by AR; the study design must be clearly articulated, with results that explore pertinent mechanisms; the research incorporates the most recent findings on the clinical applications and formulations of AR; only literature published within the last 30 years is considered, except in cases of significant historical relevance; and the research must explicitly detail molecular mechanisms or signaling pathways, along with their impact on bioavailability or efficacy.
2.3. Analytical Methods and Software
The graphical illustrations in this paper were created using multiple software tools, including the Home for Researchers (https://professional.home-for-researchers.com/) and Microsoft PowerPoint 2024 (https://www.microsoft.com/). Visualizations presenting the investigation of classical herbal formulas and their modern pharmaceutical applications were generated with Origin 2024 (https://www.originlab.com/). All chemical structures depicted in this study were drawn using ChemDraw 22.0.0.
3. Herbal Textual Research
3.1. Origin
To elucidate the origin of AR, the morphological records from ancient Chinese herbal texts have been systematically compiled and analyzed, as presented in Table 1. This compilation reveals a progressive refinement in the understanding of this herb, transitioning from an undifferentiated archetype to a pharmacognostically distinct entity. The earliest references, such as the Er Ya from the Warring States period, establish the foundational concept of “Zhu”, associating it with thistle-like plants characterized by elliptical leaves, spiny-toothed margins, and capitate inflorescences—morphological features that precisely align with the Asteraceae family to which Atractylodes genus. This initial ambiguity between Cangzhu and Baizhu reflects a holistic view of the genus before species-level differentiation. The critical shift occurred during the Northern and Southern Dynasties, as documented in the Ben Cao Jing Ji Zhu, where the name “Cangzhu” first appeared alongside explicit descriptions of “slender, branchless leaves” and roots that are “small, bitter, and rich in sap”. The emphasis on “rich in sap” directly corresponds to the high volatile oil content characteristic of A. lancea, distinguishing it organoleptically from the milder Baizhu. This text also anchors the herb geographically to Jiangsu Province, particularly the Maoshan region, establishing a provenance that would persist for centuries as the benchmark for superior quality. Subsequent Ming and Qing dynasties texts refined this knowledge with remarkable precision. The Ben Cao Yuan Shi from the Ming dynasty provides an exemplary pharmacognostic profile, extolling “Maoshan Cangzhu” for its “black bark and yellow flesh dotted with red spots”—a direct observation of oil cavities that remains a key identification marker today. It further distinguishes inferior variants by their larger size and excessively pungent taste, demonstrating a sophisticated understanding of intra-specific variation and quality gradation. The Qing dynasty Ben Cao Chong Yuan adds complementary morphological details, such as leaves near the root dividing into three or five forks and purple stems, traits that align closely with A. chinensis, indicating that while A. lancea from Maoshan was considered supreme, related species were also recognized and utilized under the same nomenclature. Throughout this historical trajectory, the consistent recording of production areas—Jiangsu, Henan, Shaanxi, Zhejiang, Anhui, and Hubei—underscores an early awareness of geo-herbalism, with the persistent emphasis on Jiangsu corroborating modern botanical knowledge that A. lancea thrives in the specific pedological conditions of this region. Collectively, the ancient texts demonstrate a cumulative empirical process: the core thistle-like morphology was established in the Er Ya, the pungent and oily nature characteristic of A. lancea was delineated by the Northern and Southern Dynasties, and the final refinement of quality standards based on organoleptic and physical traits—such as the presence of “oil spots” and “frosting”—was achieved by the Ming and Qing scholars. This 2000-year continuum of observation and documentation provides robust philological and botanical evidence that the primary origin of AR is A. lancea, with A. chinensis serving as a geographically and morphologically distinct variant, thereby affirming the foundational accuracy of Chinese Pharmacognosy.

Table 1.
Name, Characteristic traits and Origin of Atractylodis Rhizoma in ancient books.
All prescriptions documented in the Han Dynasty medical text Wu Shi Er Bing Fang employ Zhu. Shennong’s Herbal Classic states: “Zhu, bitter and warm in nature, treats wind-cold-dampness paralysis with deadened muscles, convulsions, jaundice, stops sweating, clears heat, and aids digestion.” Its effectiveness in dispelling wind and eliminating dampness parallels that of modern Cangzhu. Ming Yi Bie Lu notes: “It treats severe wind affecting the body and face, and wind-induced dizziness.” This description of efficacy also corresponds with modern Cangzhu. The aforementioned accounts indicate that medical texts from the Han and Wei dynasties, as well as earlier sources, exclusively documented Zhu. During the Northern and Southern Dynasties, Ben Cao Jing Ji Zhu recorded: “Baizhu has large, hairy leaves that branch out. Its root is sweet with little resin and can be used in pills and powders. Cangzhu has fine leaves without branching, a small root that is bitter with abundant resin, and is suitable for decoctions”. From this point onward, the two varieties of Zhu, Cangzhu and Baizhu, began to be differentiated. During the Song Dynasty, Ben Cao Tu Jing categorized them under the entry Zhu as Cangzhu and Baizhu. Zhu is characterized as follows: “Sprouting in spring, green in color without branches. Also known as Shanji, its leaves resemble thistles. The stem, which is greenish-red, resembles mugwort stalks and can grow to two or three feet in length. It blooms in summer, producing purple-green flowers similar to thistle blossoms, although some may have yellow-white flowers. The plant bears fruit after the summer solstice, and the shoots wither by autumn. Its root resembles ginger, featuring lateral fine roots, black skin, a yellowish-white core, and purple sap”. Based on these morphological descriptions—“yellow-white flowers, root resembling ginger, black skin, yellowish-white core, purple oily sap”—it is clear that Zhu corresponds precisely to the modern plants A. lancea (Mao-Cang-Zhu) or A. chinensis (Bei-Cang-Zhu). Tang Ye Ben Cao of the Yuan Dynasty classifies Baizhu and Cangzhu separately, outlining their distinct functions. According to the Ben Cao Pin Hui Jing Yao from the Ming Dynasty, Cangzhu produces shoots and leaves during spring. The leaves are slim and smooth, growing in pairs opposite each other. The stem resembles wormwood stalks, displaying a greenish-red hue and reaching a length of two to three feet. In the summer, it blossoms with thistle-like flowers in shades of purple and blue. Following the summer solstice, it yields fruit, and as autumn approaches, the shoots wilt. The root resembles ginger but lacks branches, featuring delicate lateral roots. Its skin is black, the flesh is yellow, and the core contains abundant sap. It offers a taste that is bitter, sweet, and pungent. The most esteemed roots are those harvested in spring, autumn, or winter, especially those easily frosted white. The Qing Dynasty’s Ben Cao Chong Yuan provides a detailed account of the stems, leaves, and rhizomes of Cangzhu, stating that near the root of A. chinensis, the leaves divide into three to five forks, with the upper leaves being narrow, elongated, and possessing a green, glossy sheen. The classification standards for Cangzhu and Baizhu during the Qing Dynasty correspond to contemporary norms, where Cangzhu represents a wild variety, while ancient Baizhu encompasses Zhezhu from the Ben Cao Meng Quan and Wuzhu from the Compendium of Materia Medica, both being cultivated types with their cultivation attributed to Tao Hongjing during the Northern and Southern Dynasties. Consequently, the Zhu utilized in the Han Dynasty’s Shang Han Lun should be Cangzhu rather than the subsequently cultivated Baizhu.
Herbal texts prior to the Song Dynasty only documented the regions where Zhu was produced. For instance, the Compendium of Famous Physicians’ Supplementary Records states: “Zhu grows in the valleys of Mount Na, Hanzhong”. The Collected Notes on Ben Cao Jing Ji Zhu states: “It is now found everywhere, but those from Jiangshan, Zishan, and Mount Mao are considered superior.” This indicates that Zhu was produced in Shaanxi and Jiangsu provinces, which remain the primary production areas for Cangzhu today. After the Song Dynasty, ancient herbal texts often distinguished the origins of Cangzhu and Baizhu. For instance, Ben Cao Tu Jing from the Song Dynasty records: “Zhu grows in the valleys of Mountain Zheng, Hanzhong, now known as Shaanxi Province. Now it is found everywhere, with those from Mountain Song and Mountain Mao being the finest”. The Compendium of Materia Medica records: “Cangzhu from Mount Mao is now considered the finest. Its root bark is black with white flesh and yellow spots. Cangzhu from other mountains has larger roots with yellow flesh and a fiercely pungent aroma”. The Republican-era Yao Wu Chan Chu Bian notes: “Tianshengzhu is originally produced in Xiushui County, Jiangxi Province”. As described above, Cangzhu has a broad distribution range. Its earliest recorded origin is Hanzhong, Shaanxi, later gradually expanding to Jiangsu, Henan, and Hubei. The highest quality Cangzhu comes from Mountain Mao, Jiangsu. Currently, it is primarily distributed in Shandong, Jiangsu and Zhejiang, Hubei, Sichuan, and other provinces. With increasing recognition, Bei-Cang-Zhu (A. chinensis) has gradually become mainstream, and its functions and effects are comparable to those of Mao-Cang-Zhu (A. lancea).
3.2. Textual Research of Traditional Ethnopharmacology, Uses and Prescriptions
Ethnopharmacology and ethnic pharmacology are integral components of the theoretical framework of Traditional Chinese Medicine and serve as foundational principles for clinical prescriptions. AR is characterized by its pungent and warm properties, accompanied by a bitter taste, and is associated with the spleen, stomach, and liver meridians. It functions to eliminate dampness, strengthen the spleen, dispel wind, disperse cold, and enhance vision. This herb is predominantly utilized in the treatment of conditions such as dampness obstructing the middle jiao, epigastric and abdominal distension, diarrhea, edema, beriberi paralysis, rheumatic arthralgia, wind-cold common cold, night blindness, and blurred vision.
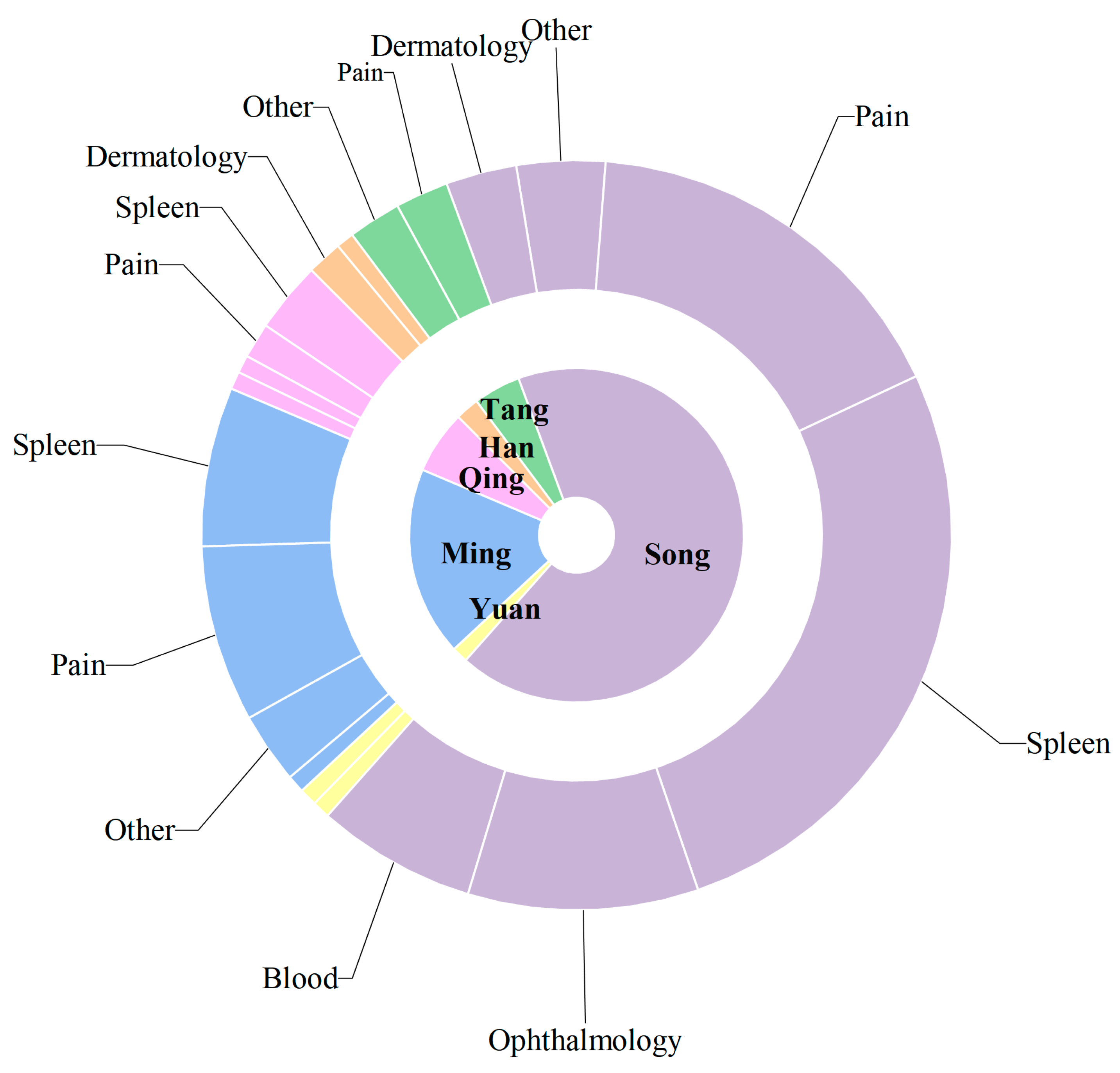
Upon verification, a total of 197 internal formulas and 3 external formulas were identified, of which 54 formulas incorporated rice water-processed AR to address spleen-stomach disharmony and related conditions (refer to Table S1). An analysis of the historical application of AR (Figure 2) spans six dynasties and six disease categories. The Song Dynasty represents the peak period for AR utilization, with the highest number of formulas (88) and the broadest therapeutic scope, particularly emphasizing gastrointestinal disorders and external pathogen/pain conditions, while ophthalmic formulas also occupied a significant proportion. The Ming Dynasty follows with 24 formulas, where external pathogen/pain applications slightly outnumber gastrointestinal ones, reflecting the continued use of AR in treating rheumatic arthralgia. Although fewer formulas are recorded in the Qing Dynasty, gastrointestinal applications remain dominant. Notably, ophthalmic uses are predominantly concentrated in the Song Dynasty and rarely appear in other periods, possibly due to the extensive inclusion of ophthalmic formulas in the comprehensive text Sheng Ji Zong Lu. These findings demonstrate that AR has been consistently applied to gastrointestinal disorders throughout history, further validating the scientific basis and continuity of its traditionally recognized functions of “drying dampness and strengthening the spleen” that persist in modern clinical practice.
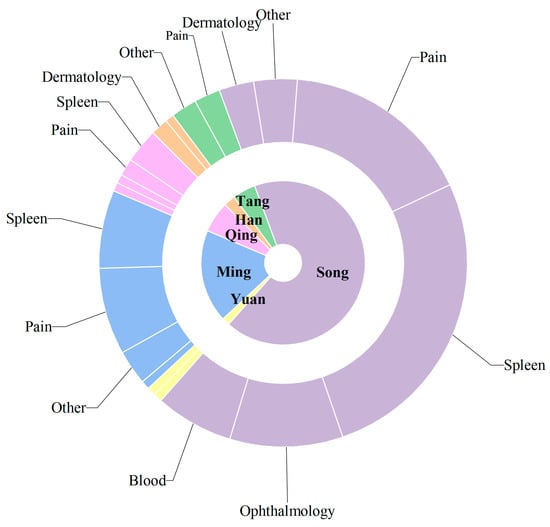
Figure 2.
Historical Application of Atractylodis Rhizoma: A Sunburst Diagram of Therapeutic Categories by Dynasty. Note: (1) Inner Ring: Dynasties, arranged in historical sequence including Han, Tang, Song, Yuan, Ming, Qing. (2) Outer Ring: Therapeutic Categories, derived from the “Traditional uses/Efficacy” field in each formula description. Primarily divided into six categories: Gastrointestinal: Addresses spleen-stomach deficiency, diarrhea, vomiting, abdominal distension, etc. External/Pain: Includes typhoid fever, headache, body pain, rheumatism, joint pain, etc. Ophthalmology: Treats redness and swelling of the eyes, corneal opacity, night blindness, etc. Gynecology/Blood: Covers pregnancy, postpartum conditions, blood stasis, etc. Dermatology: Treats sores, hives, scabies, and tinea. Other: Includes tonifying, qi regulation, phlegm-fluid retention, and other non-categorizable uses. (3) Values: Represents the number of formulas in each category per dynasty. The sum equals the total number of formulas for that dynasty. (4) Colors: Different colors represent different dynasties.
3.3. Research on Modern Preparations
AR is renowned for its distinctive therapeutic properties, including the ability to dry dampness, fortify the spleen, dispel wind, and alleviate cold. These properties have been validated through extensive historical use, resulting in the preservation of numerous traditional formulations that persist to the present day. With the progression of modern pharmacology and advancements in Traditional Chinese Medicine, these compound formulations have been further refined and applied (see Table S2). Consequently, a variety of formulations, such as granules, capsules, tablets, and pills, have been developed. These are extensively employed to treat conditions like dampness obstructing the middle burner and spleen-stomach disharmony, effectively drying dampness and enhancing spleen function.
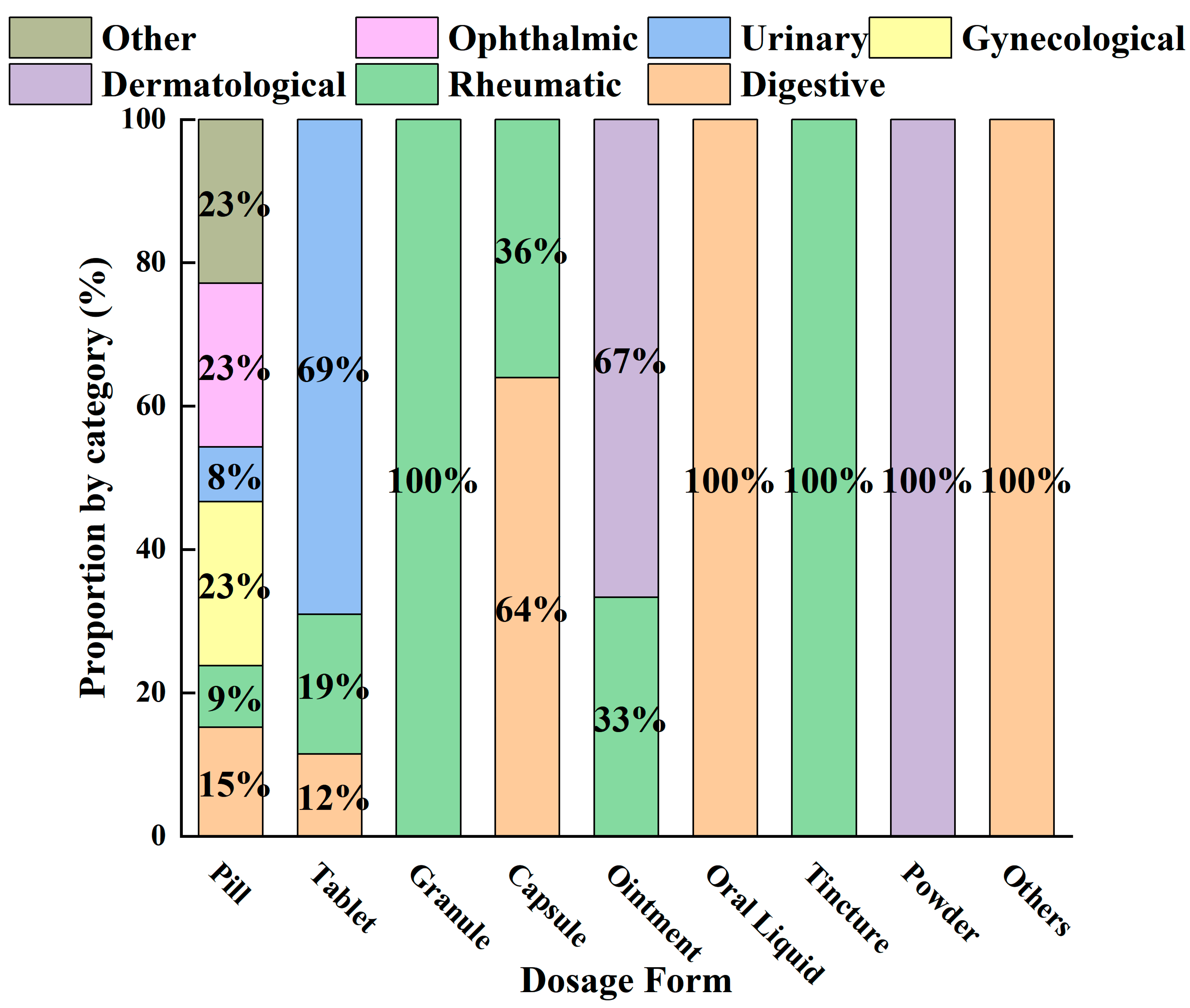
Currently, a statistical analysis of the 54 AR-containing formulations recorded in the Chinese Pharmacopoeia reveals the overall distribution characteristics of this herb in modern patent medicines (Figure 3). The findings demonstrate a pattern characterized by diverse dosage forms, concentrated therapeutic indications, and the integration of traditional and modern pharmaceutical approaches. Pills serve as the core carrier, supporting the extensive application of AR in the digestive, urinary, and rheumatic systems. Meanwhile, modern dosage forms—such as granules, capsules, and tablets—expand the routes of administration and scope of indications while preserving its traditional efficacy. This distribution not only validates the millennium-old medicinal use of AR in a contemporary context but also provides data support and directional guidance for future drug development.
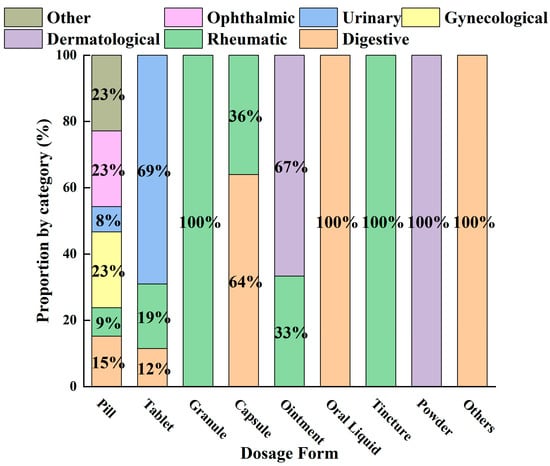
Figure 3.
Distribution of Dosage Forms and Therapeutic Categories in Modern Atractylodis Rhizoma-Containing Patent Medicines.
4. Phytochemistry
To date, a total of 327 small molecules have been identified from AR, encompassing a diverse range of structural classes including sesquiterpenoids, mono-terpenoids, polyacetylenes, triterpenoids, phenolic acids, flavonoids, and steroidal compounds [3]. While this extensive inventory provides a valuable foundation, a critical analytical evaluation is necessary to differentiate compounds based on their abundance, biosynthetic origin, and distribution between the two pharmacopoeial species, A. lancea (AL) and A. chinensis (AC). Such stratification enhances our understanding of the chemotaxonomic markers and the true pharmacologically active principles.
From the perspective of relative abundance, the 327 compounds can be stratified into major bioactives (Table 2), minor components, and trace constituents. The primary and most studied components of AR are lipophilic and accumulate in the essential oil and resin, predominantly comprising sesquiterpenoids and polyacetylenes. Among sesquiterpenoids, atractylon, hinesol, β-eudesmol, and the atractylenolides (I, II, III) are consistently reported as the dominant compounds and form the core bioactive foundation for pharmacological effects such as gastroprotection and anti-inflammation [14]. Quantitative analyses have demonstrated that hinesol and β-eudesmol are characteristically high in AL, while atractylon and γ-eudesmol are notably abundant in AC. Polyacetylenes, particularly atractylodin and its derivatives such as atractylodinol, represent another key group of major bioactives with hepatoprotective properties; these compounds are significantly more concentrated in AL compared to AC. In contrast, a substantial portion of the identified compounds—including numerous saturated and unsaturated hydrocarbons (e.g., various alkanes) and many mono- and sesquiterpene hydrocarbons—are present only in trace amounts. While these trace constituents contribute to the complex aromatic profile of the herb, they are unlikely to be primary drivers of its classical therapeutic actions and are more relevant for establishing complete volatile fingerprints for species differentiation or authenticity testing.
A critical assessment of the phytochemical dataset must also consider the distinction between genuine plant metabolites and potential analytical artifacts. The biosynthetic relationship between atractylon and the atractylenolides warrants particular attention. Atractylon is chemically unstable and readily undergoes autoxidation upon exposure to air, light, or heat [15]. Mechanistic studies have elucidated that atractylon undergoes oxidative conversion through a series of reactions involving water addition, ring cleavage, and carboxylation to generate atractylenolide I, which can further transform into atractylenolides II and III. Consequently, while these lactones are certainly present in processed crude drugs, their quantified levels may not fully represent the native metabolic profile of the fresh rhizome, and a proportion of the measured atractylenolides—particularly in dried or aged samples—may be artifacts derived from atractylon oxidation. This phenomenon is further supported by processing studies demonstrating that stir-frying with bran, a common traditional processing method, leads to decreased levels of both sesquiterpenoids and polyacetylenes, likely due to thermal degradation and oxidative transformations [16]. Additionally, the presence of phthalate esters such as diethyl phthalate, diisobutyl phthalate, and bis(2-ethylhexyl) phthalate is a strong indicator of contamination rather than genuine phytochemical production, as these compounds are ubiquitous plasticizers that readily leach from plastic labware and storage containers during analytical workflows [17].
Comparative analysis between the two official species reveals distinct chemical profiles that underpin their differentiation in traditional medicine and necessitate species specification in modern pharmacological research. The most prominent chemotaxonomic difference lies in the dominant sesquiterpenoid patterns: AL is characterized by high concentrations of hinesol, β-eudesmol, and atractylol, whereas AC is defined by its high content of atractylon and γ-eudesmol, with compounds like β-selinene also showing higher relative abundance in AC. Furthermore, AL exhibits significantly higher accumulation of polyacetylenes, particularly atractylodin and acetylatractylodinol, compared to AC, making polyacetylene content a key differentiating factor that may contribute to variations in reported bioactivities such as hepatoprotection. While most of the remaining compounds, especially trace hydrocarbons and ubiquitous fatty acids, are present in both species, certain compounds such as atrachinenins D, E, F, and G have been reported exclusively in AC based on current literature, representing potential unique biomarkers. These clear qualitative and quantitative differences underscore the absolute necessity of specifying the species used in any experimental study to ensure reproducibility and the correct attribution of observed pharmacological effects.

Table 2.
Some of the chemical constituents from Atractylodis Rhizoma.
Table 2.
Some of the chemical constituents from Atractylodis Rhizoma.
| NO. | Compounds | Structure | AL | AC | Class | References |
|---|---|---|---|---|---|---|
| 1 | α-Pinene | 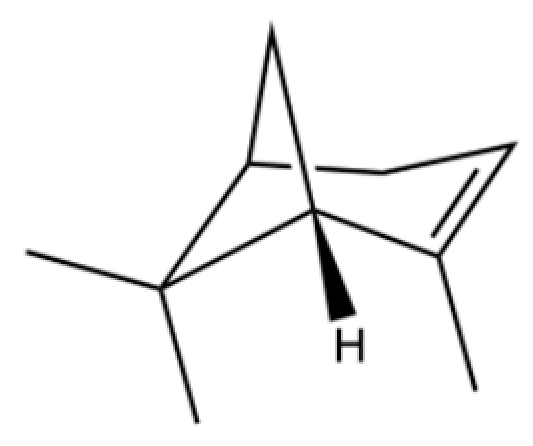 | * | * | Monoterpenoid | [18,19] |
| 2 | Nonane, 2,6-dimethyl- | 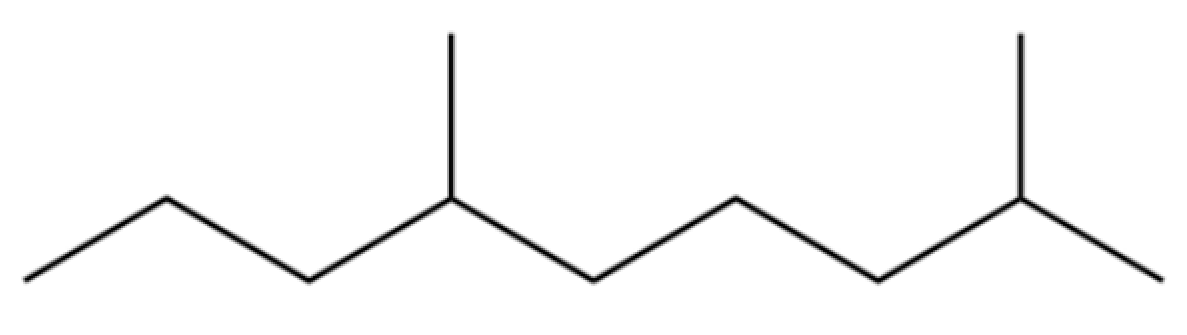 | * | * | Alkane | [20] |
| 3 | Decane, 4-methyl- | 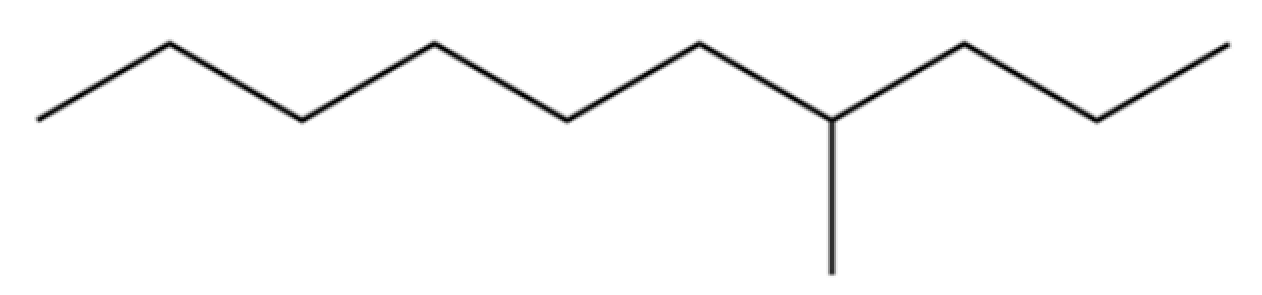 | * | * | Alkane | [20] |
| 4 | 2-Hexanone | 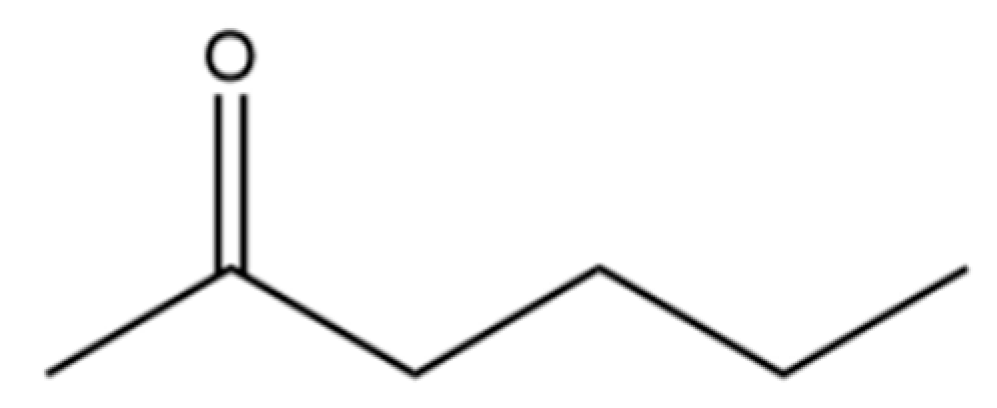 | * | * | Ketone | [20] |
| 5 | α-Phellandrene | 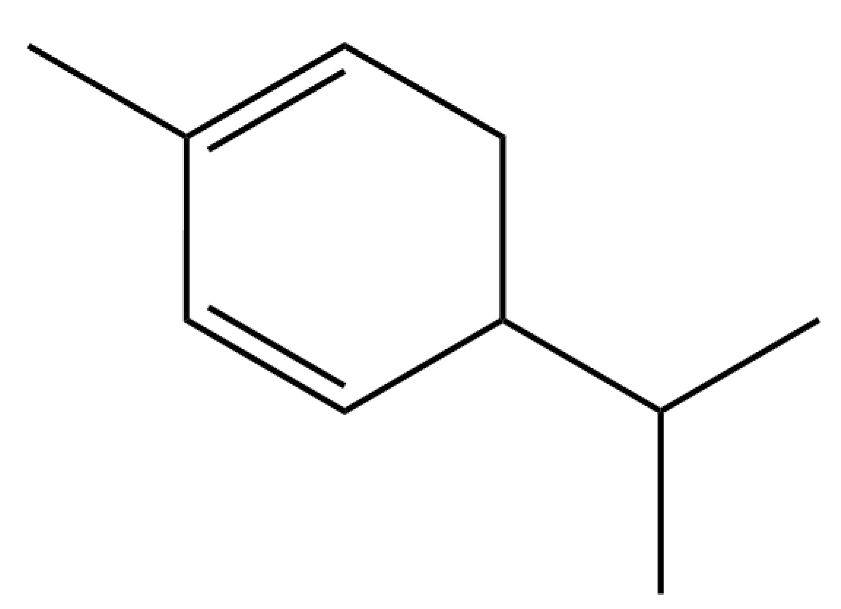 | * | * | Monoterpenoid | [21] |
| 6 | Undecane | 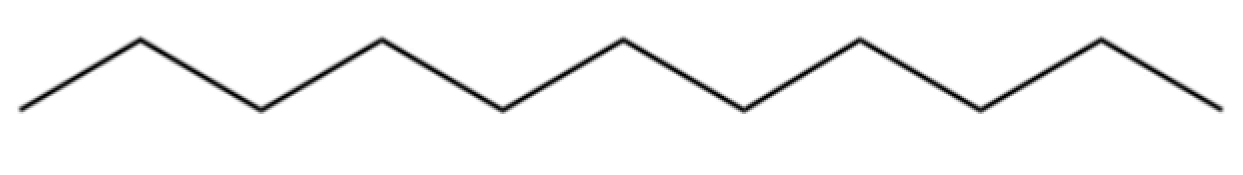 | * | * | Alkane | [20] |
| 7 | 3-Hexanol | 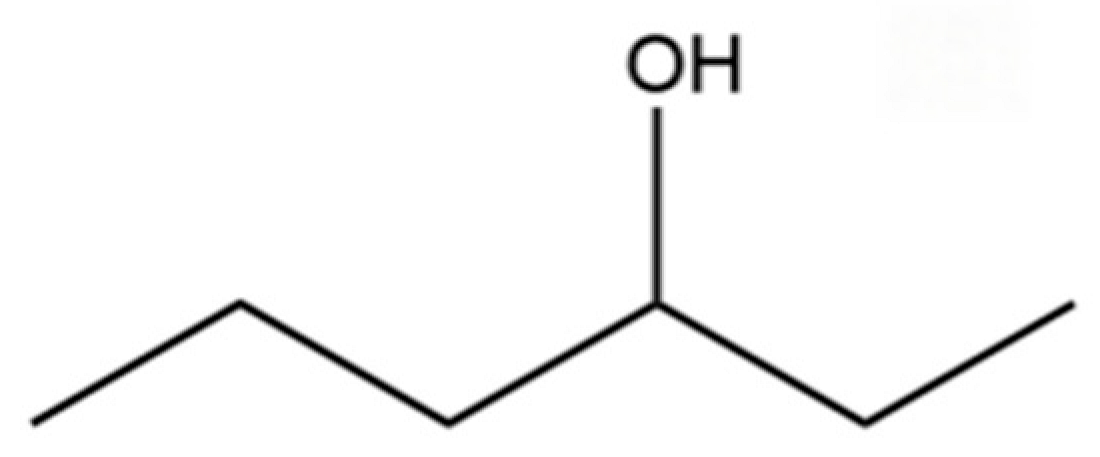 | * | * | Alcohol | [20] |
| 8 | Dodecane |  | * | * | Alkane | [20] |
| 9 | 2,6-Dimethylundecane | 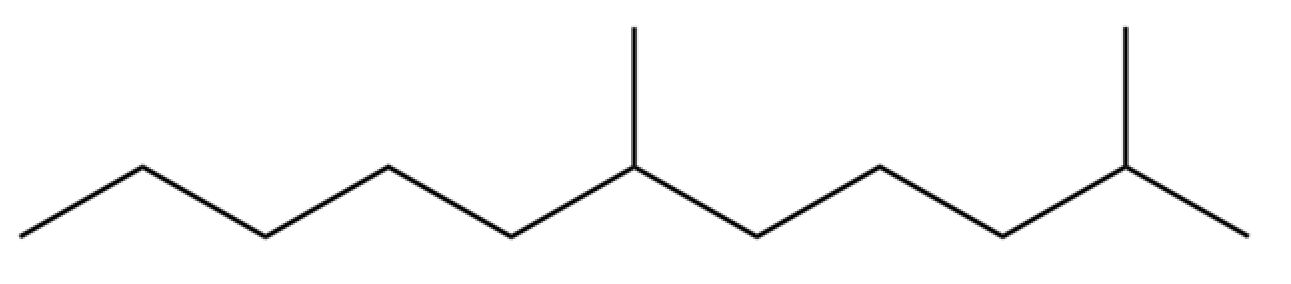 | * | * | Alkane | [20] |
| 10 | 2-Hexanol |  | * | * | Alcohol | [20] |
| 11 | Tridecane |  | * | * | Alkane | [20] |
| 12 | 2,7,10-trimethyl-Dodecane |  | * | * | Alkane | [20] |
| 13 | Silphiperfol-5-ene |  | * | * | Sesquiterpenoid | [22] |
| 14 | 2-Methyltridecane |  | * | * | Alkane | [20] |
| 15 | Atrachinenins G |  | / | ** | Sesquiterpenoid | [23] |
| 16 | 2,6,10-Trimethyltridecane |  | * | * | Alkane | [20] |
| 17 | Tetradecane |  | * | * | Alkane | [20] |
| 18 | α-Guaiene |  | * | * | Sesquiterpenoid | [3] |
| 19 | Pentadecane |  | * | * | Alkane | [20] |
| 20 | Modephene |  | * | * | Sesquiterpenoid | [24] |
| 21 | Cyperene |  | * | * | Sesquiterpenoid | [25] |
| 22 | β-Elemene |  | * | * | Sesquiterpenoid | [21] |
| 23 | Isocomene |  | * | * | Sesquiterpenoid | [20] |
| 24 | β-Isocomene |  | * | * | Sesquiterpenoid | [20] |
| 25 | Caryophyllene | 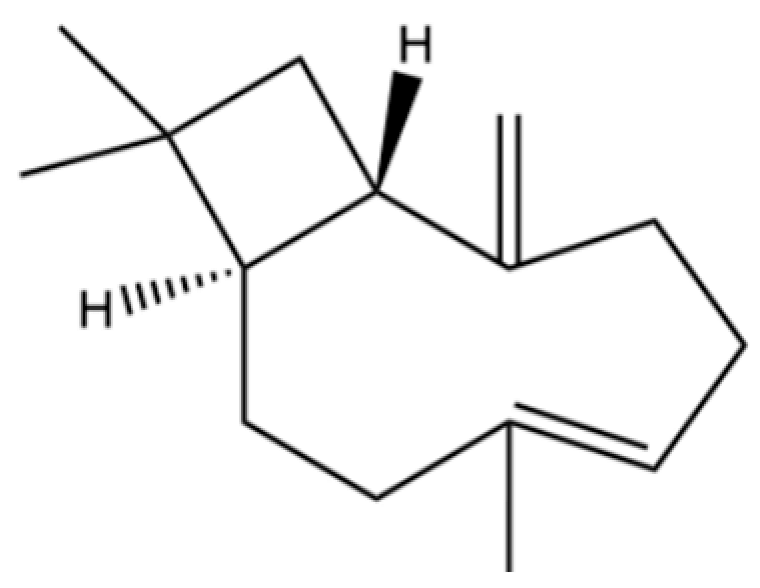 | * | * | Sesquiterpenoid | [25] |
| 26 | Aciphyllene | 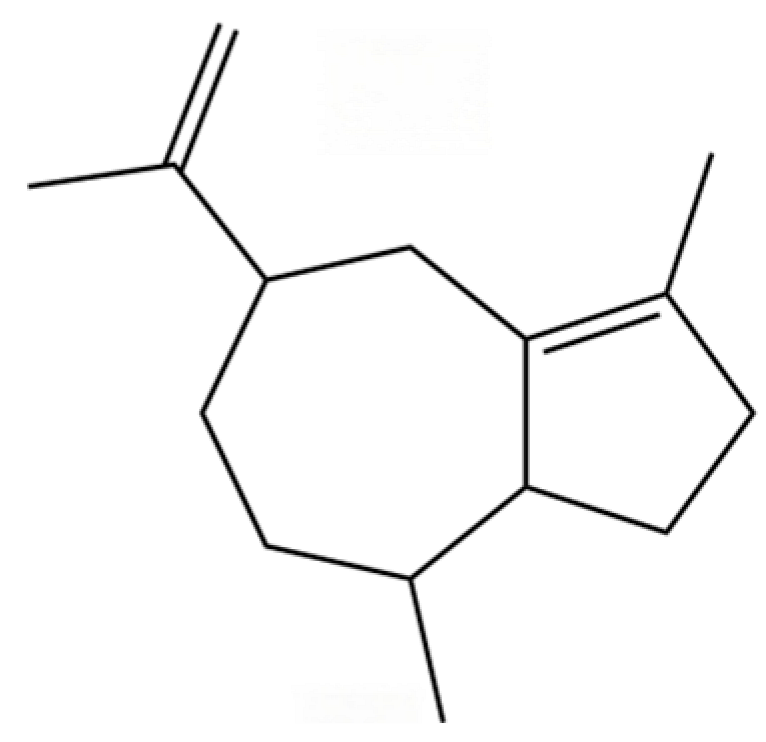 | * | * | Sesquiterpenoid | [20] |
| 27 | β-Famesene | 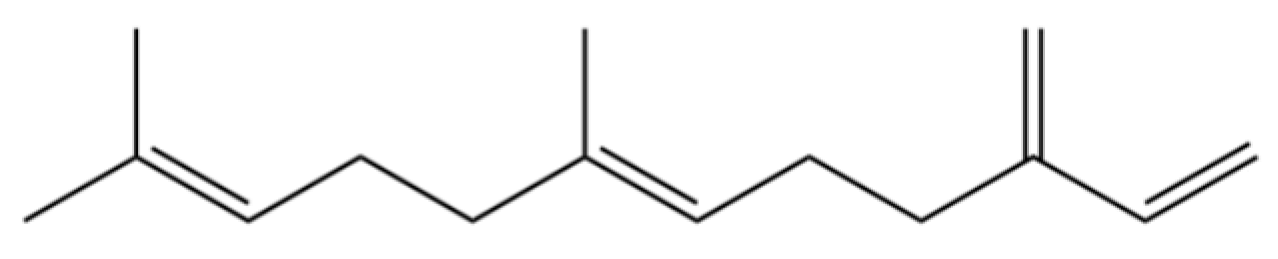 | * | * | Sesquiterpenoid | [20] |
| 28 | Atrachinenins F | 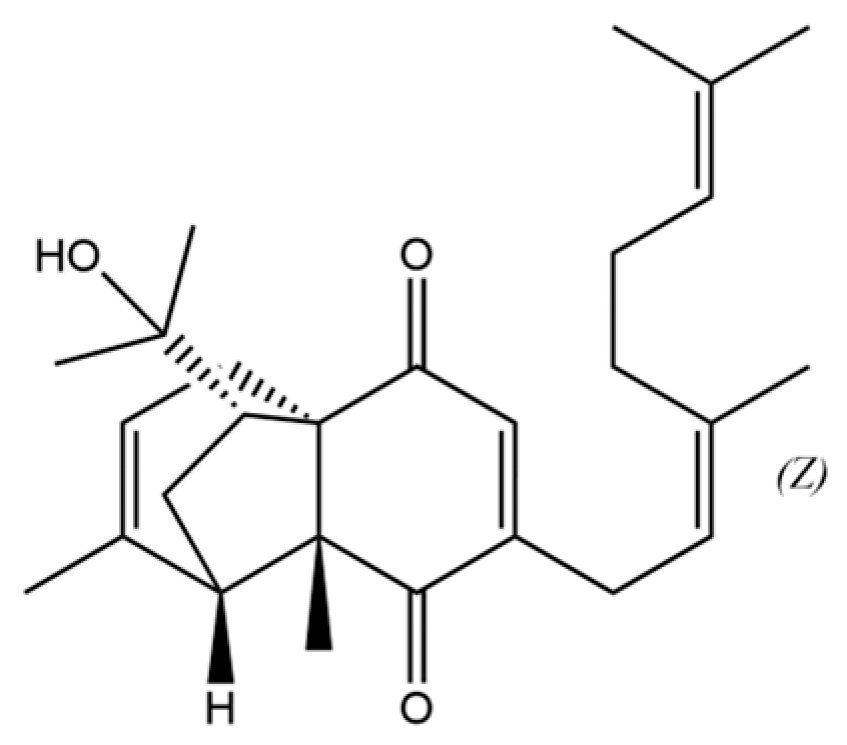 | / | ** | Sesquiterpenoid | [23] |
| 29 | Octadecane |  | * | * | Alkane | [26] |
| 30 | Humulene |  | * | * | Sesquiterpenoid | [21] |
| 31 | 2-Isopropenyl-4a, 8-dimethyl-1,2,3,4,4a, 5,6,7-octahydronaphthalene |  | * | * | Sesquiterpenoid | [20] |
| 32 | (+)-Eudesma-4(14),7(11)-dien- 8-one |  | * | * | Sesquiterpenoid | [25] |
| 33 | Isoborneol | 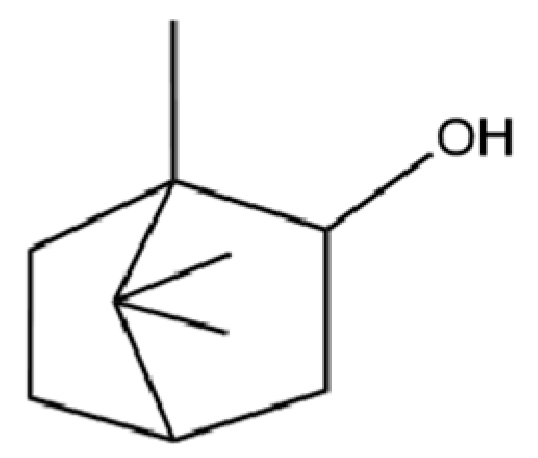 | * | * | Monoterpenoid | [20] |
| 34 | 1-Methyl-4-(6-methylhept-5-en-2-yl) cyclohexa-1,3-diene | 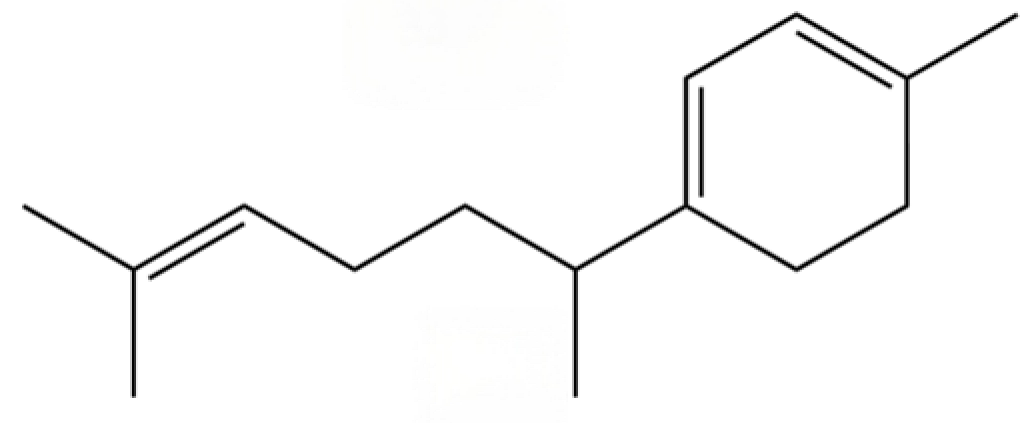 | * | * | Sesquiterpenoid | [20] |
| 35 | Germacrene D | 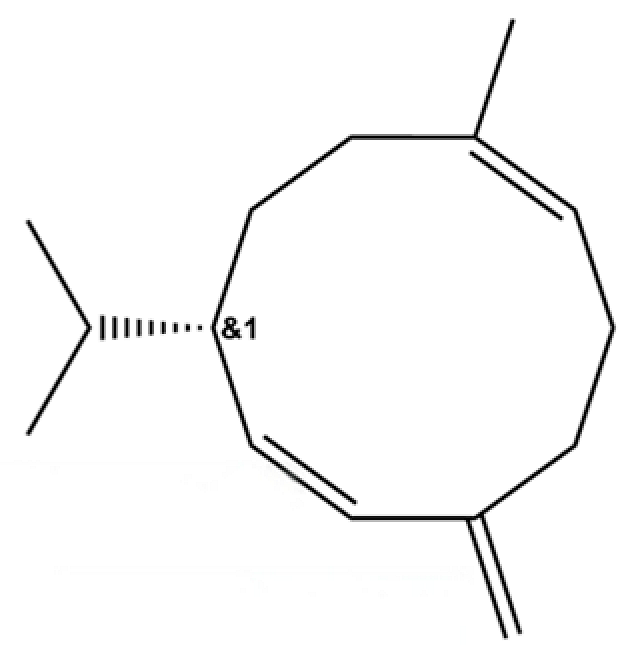 | * | * | Sesquiterpenoid | [27] |
| 36 | β-selinene | 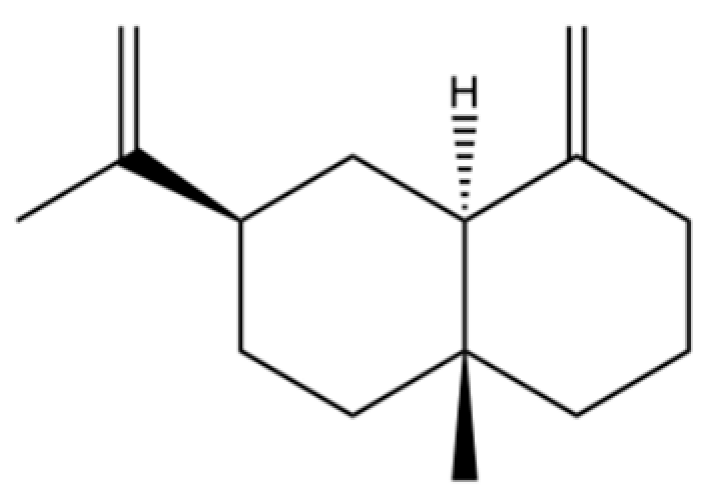 | * | ** | Sesquiterpenoid | [21] |
| 37 | 2-Cyclohexen-1-ol, 3-methyl-6-(1-methylethyl)-, cis- | 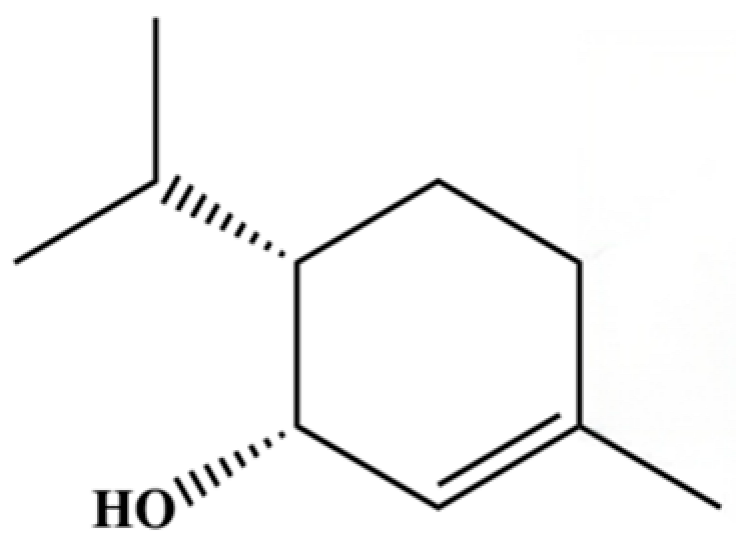 | * | * | Monoterpenoid | [20] |
| 38 | β-Curcumen | 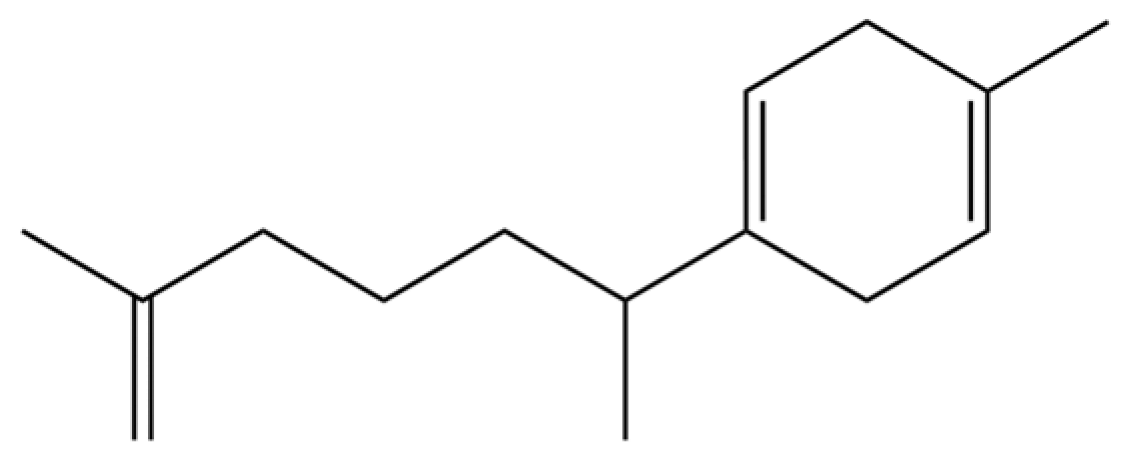 | * | * | Sesquiterpenoid | [20] |
| 39 | Guaia-1 (10),11-diene | 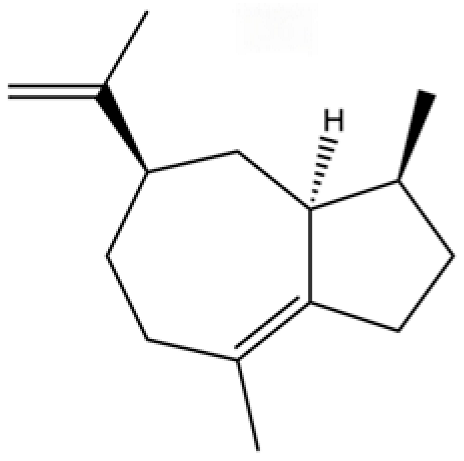 | * | * | Sesquiterpenoid | [20] |
| 40 | 2,6-Octadien-1-ol, 3,7-dimethyl-, acetate | 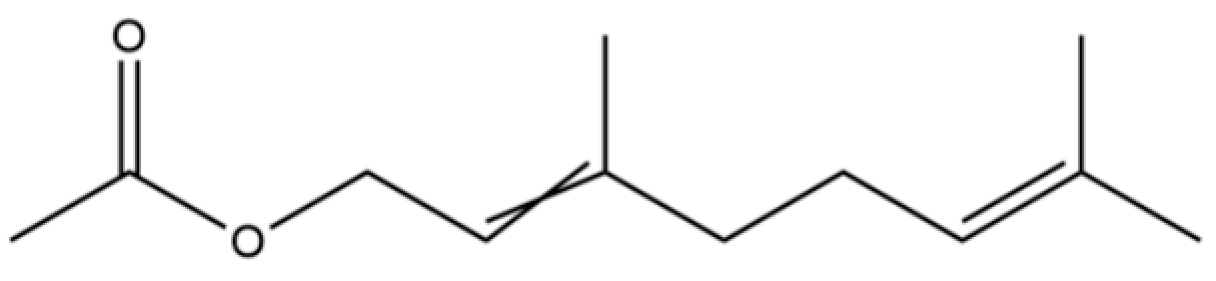 | * | * | Monoterpenoid | [25] |
| 41 | Citronellol | 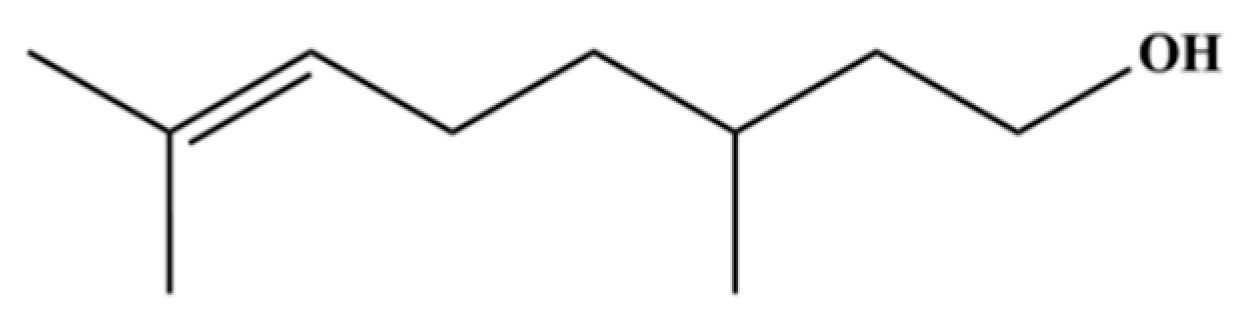 | * | * | Monoterpenoid | [20] |
| 42 | γ-Cadinene | 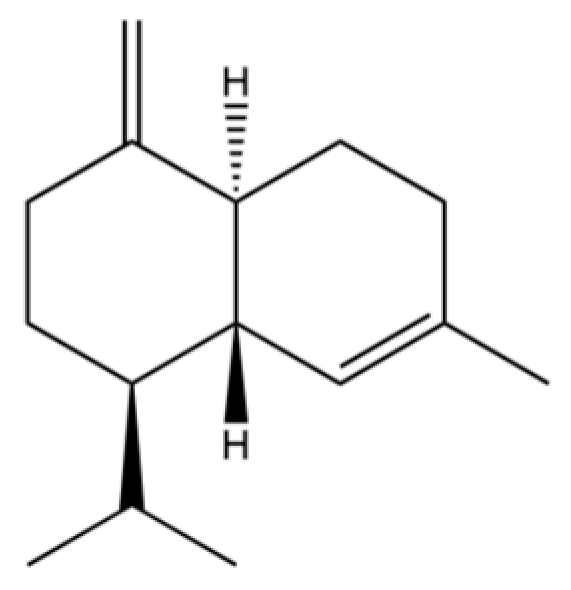 | * | * | Sesquiterpenoid | [21] |
| 43 | Methyl salicylate | 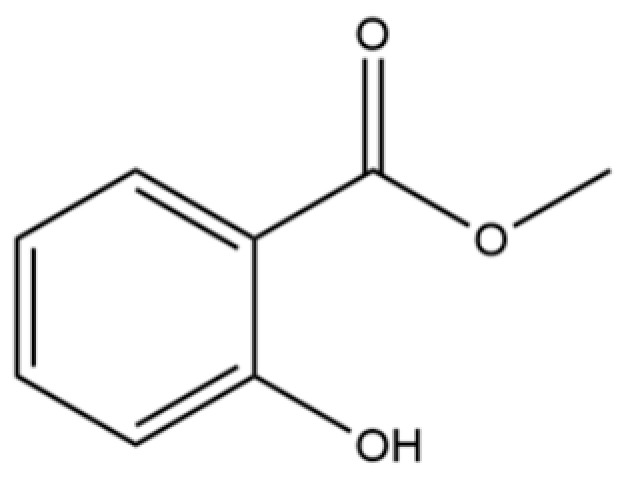 | * | * | Phenolic ester | [20] |
| 44 | β-Sesquiphellandrene | 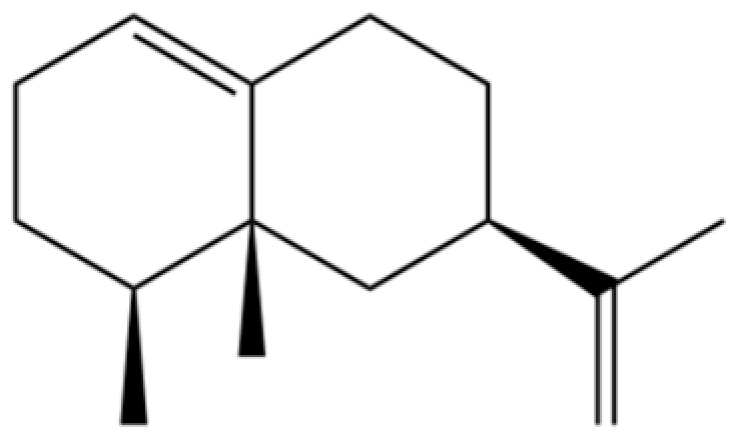 | * | * | Sesquiterpenoid | [25] |
| 45 | β-Vatirenene | 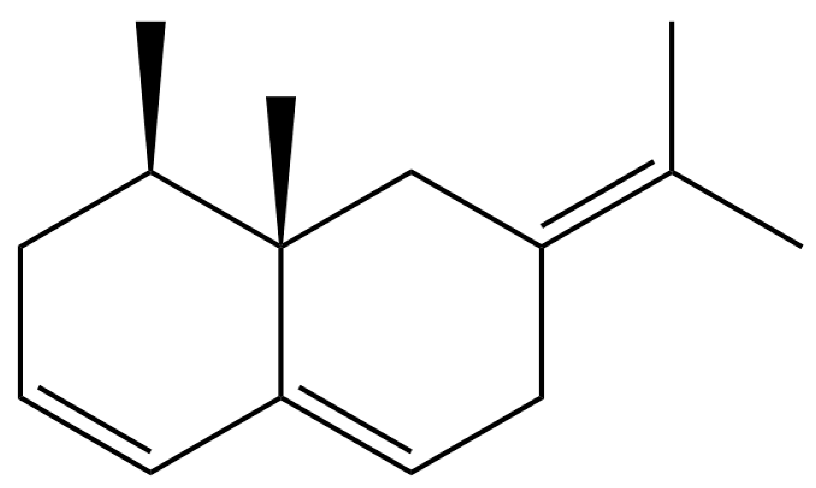 | * | * | Sesquiterpenoid | [25] |
| 46 | α-Curcumene | 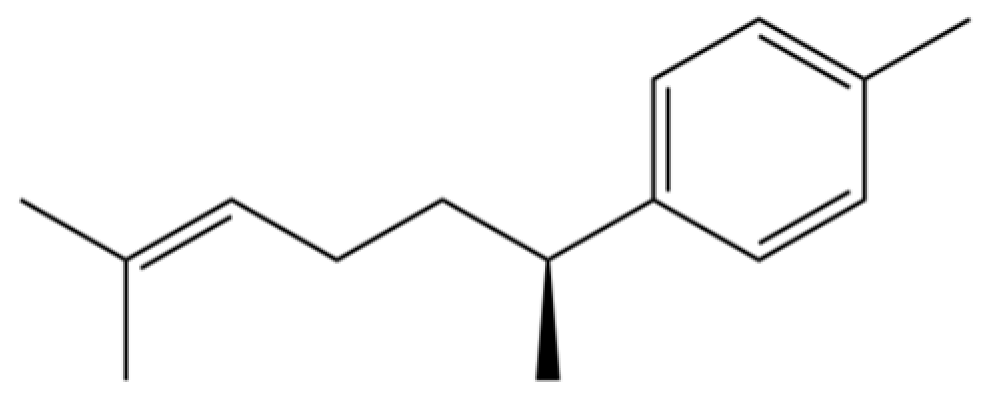 | * | * | Sesquiterpenoid | [21] |
| 47 | Eremophilene | 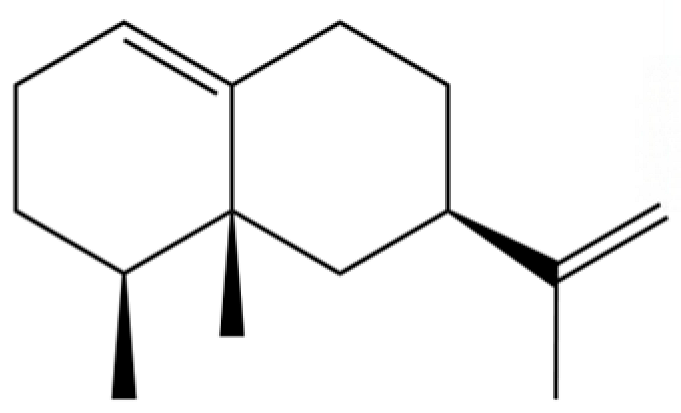 | * | * | Sesquiterpenoid | [20] |
| 48 | cis-Sabinol | 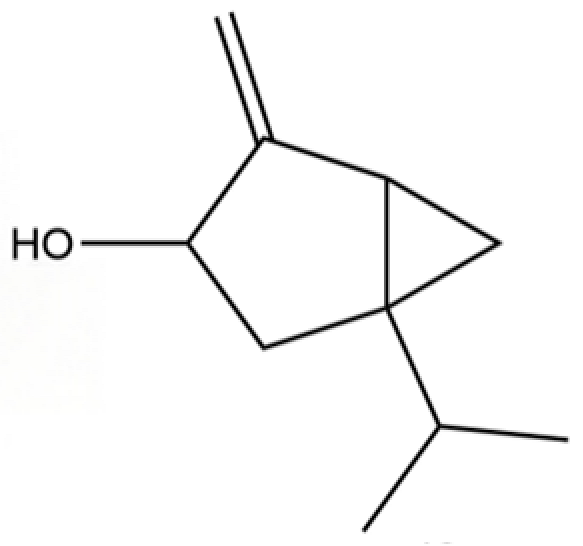 | * | * | Monoterpenoid | [20] |
| 49 | γ-Elemene |  | * | * | Sesquiterpenoid | [25] |
| 50 | Nerol |  | * | * | Monoterpenoid | [20] |
| 51 | Heptadecane, 9-hexyl- |  | * | * | Alkane | [20] |
| 52 | Butylated hydroxytoluene |  | * | * | Phenolic antioxidant | [20] |
| 53 | Cedrene epoxide | 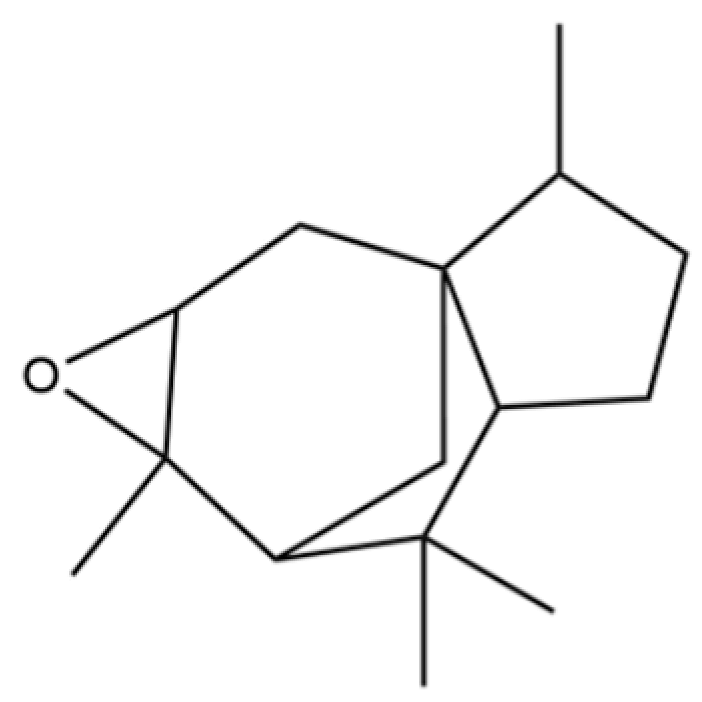 | * | * | Sesquiterpenoid | [20] |
| 54 | (3S,3aR,3bR,4S,7R,7aR)-4-Isopropyl-3,7-dimethyloctahydro -1H-cyclopenta[1,3]cyclopropa[1,2]benzen-3-ol | 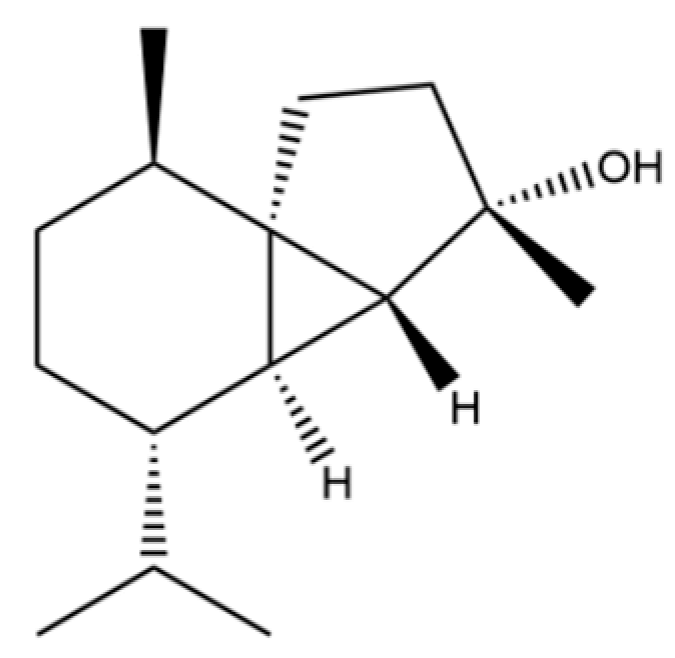 | * | * | Sesquiterpenoid | [20] |
| 55 | trans-Longipinocarveol | 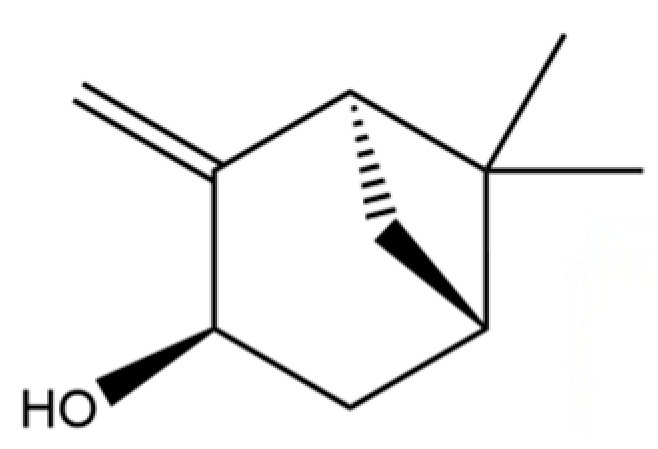 | * | * | Sesquiterpenoid | [20] |
| 56 | Nerolidol | 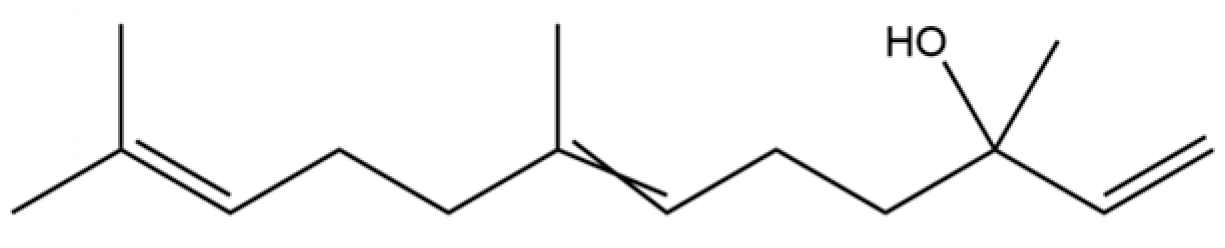 | * | * | Sesquiterpenoid | [28] |
| 57 | Humulene epoxide II | 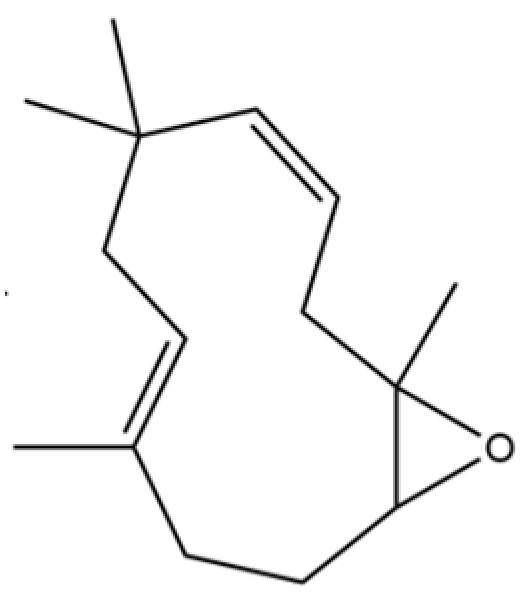 | * | * | Sesquiterpenoid | [20] |
| 58 | Epicubenol | 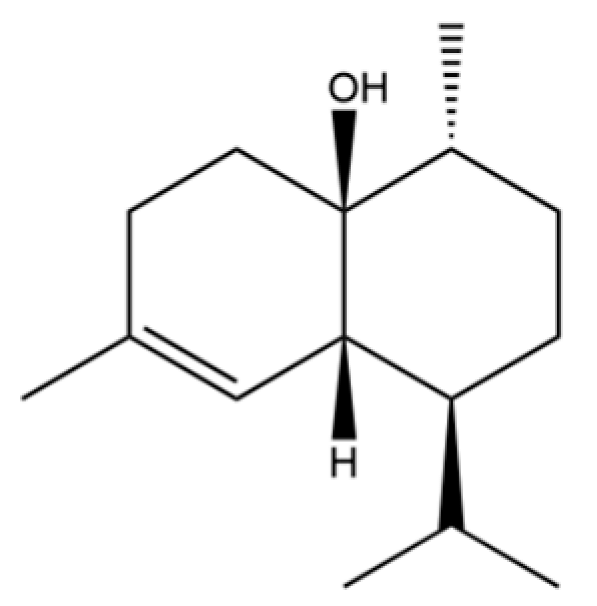 | * | * | Sesquiterpenoid | [20] |
| 59 | 5-Azulenemethanol, 1,2,3,4,5,6,7,8-octahydro-α,α,3,8-tetramethyl- | 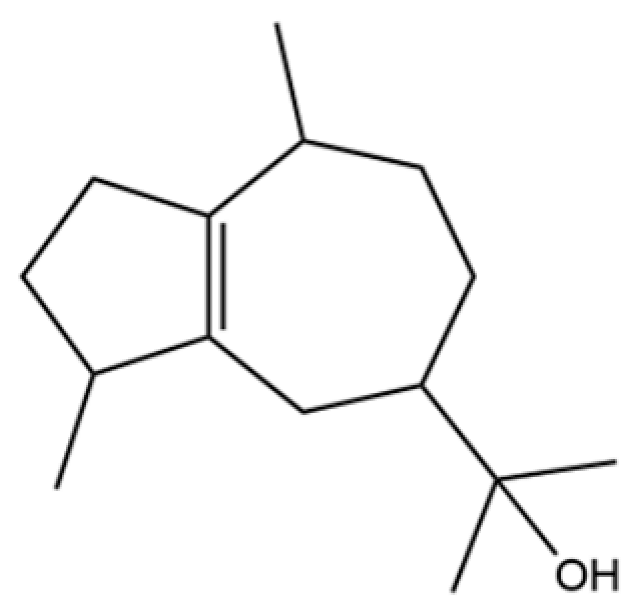 | * | * | Sesquiterpenoid | [20] |
| 60 | 1 (2H)-Naphthalenone, octahydro-4a, 8a-dimethyl-7-(1-methylethyl)-,[4aR-(4aα,7ß,8aα)]- | 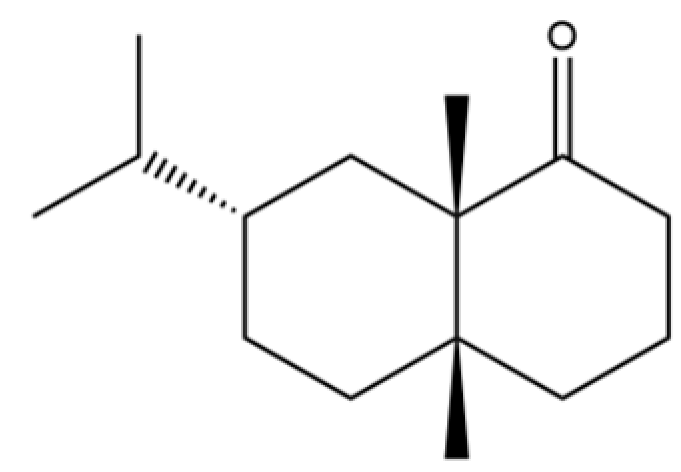 | * | * | Sesquiterpenoid | [20] |
| 61 | Atractylon | 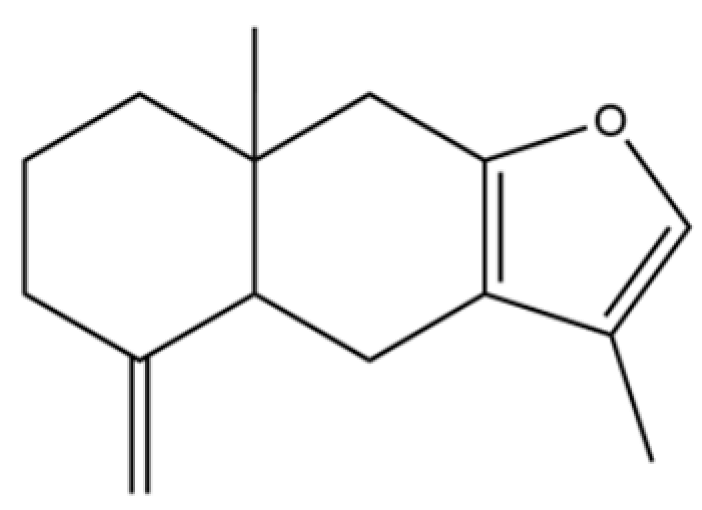 | * | ** | Sesquiterpenoid | [21] |
| 62 | γ-eudesmol | 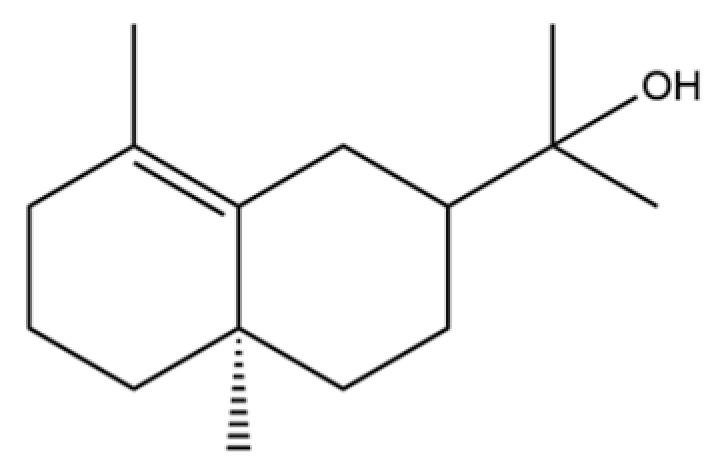 | * | ** | Sesquiterpenoid | [25] |
| 63 | 8,14-Cedranoxide | 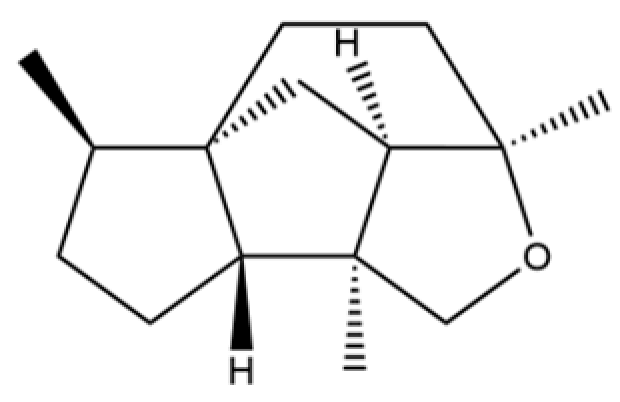 | * | * | Sesquiterpenoid | [20] |
| 64 | Thymol | 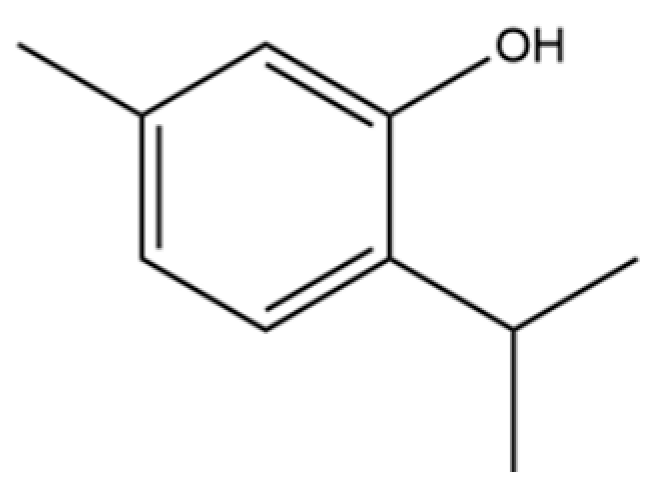 | * | * | Monoterpenoid phenol | [25] |
| 65 | Agarospirol | 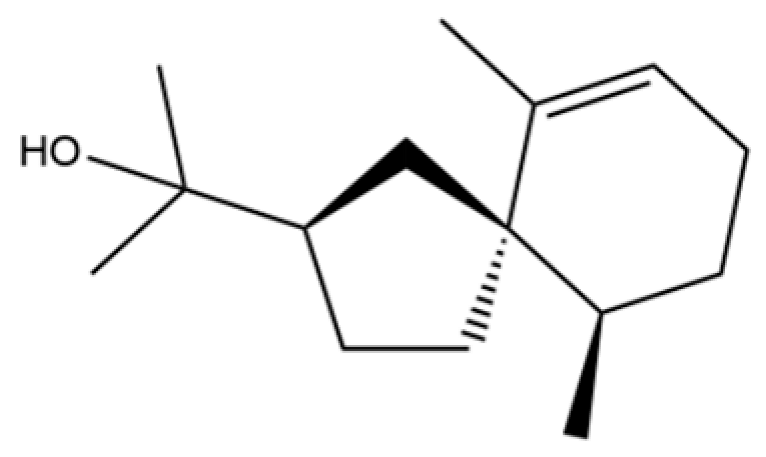 | * | * | Sesquiterpenoid | [29] |
| 66 | Hinesol | 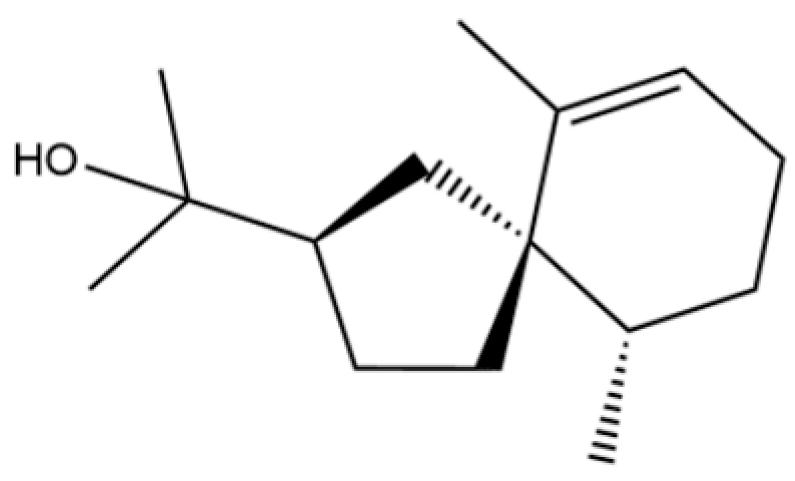 | ** | * | Sesquiterpenoid | [21] |
| 67 | trans-Valerenyl acetate | 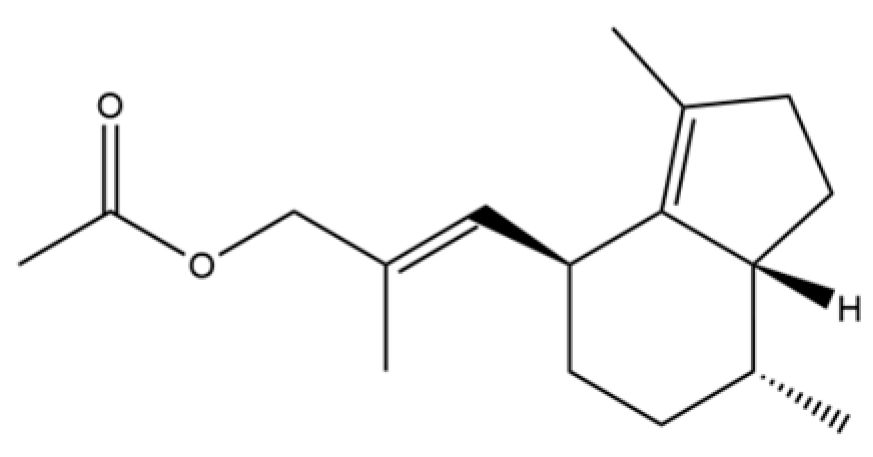 | * | * | Sesquiterpenoid | [20] |
| 68 | 2-methyl-5-(1-methylethyl)-Phenol | 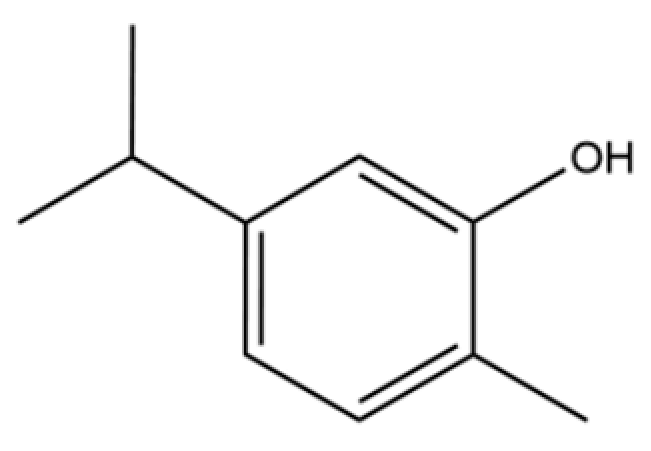 | * | * | Phenolic | [20] |
| 69 | α-Bisabolol | 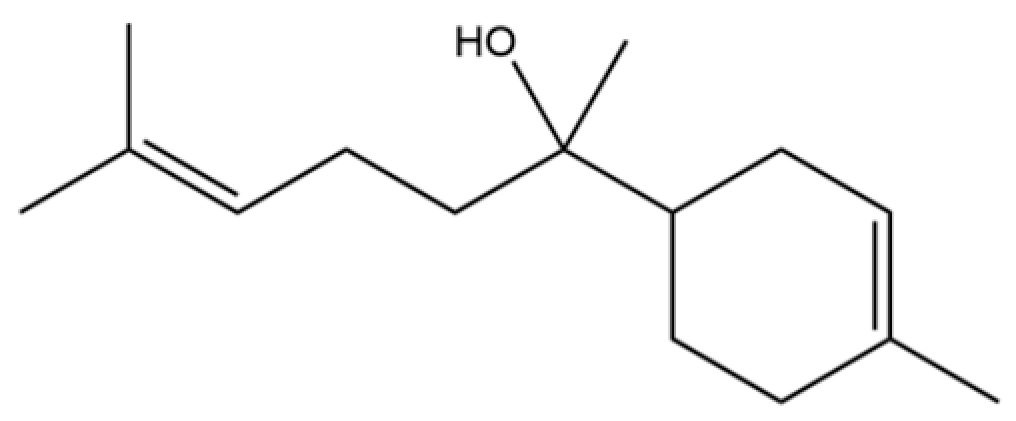 | * | * | Sesquiterpenoid | [25] |
| 70 | Atractylol | 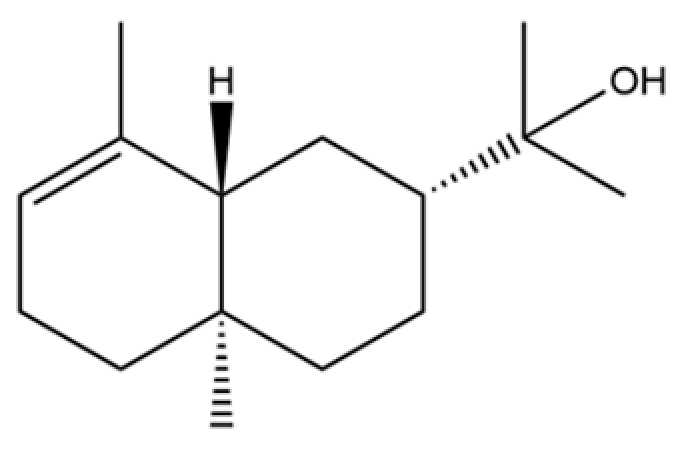 | ** | * | Sesquiterpenoid | [30] |
| 71 | β-Eudesmol | 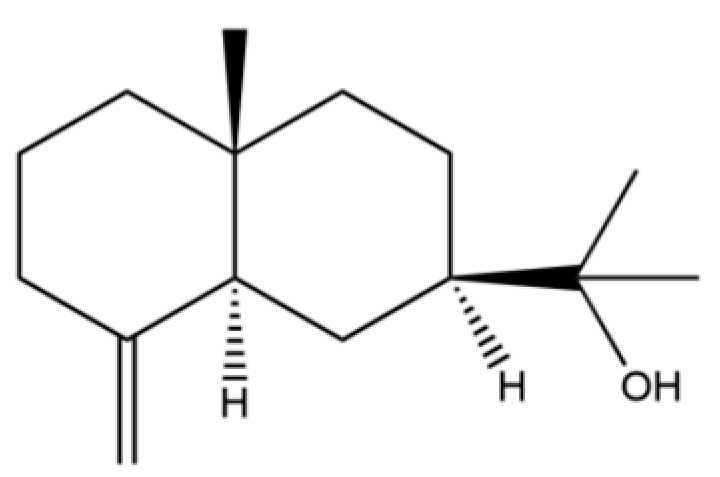 | ** | * | Sesquiterpenoid | [21] |
| 72 | 3,7-dimethyl-6-Octenoic acid | 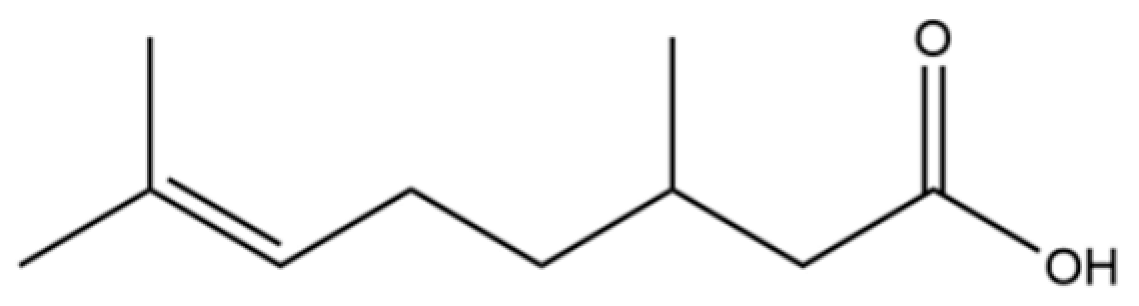 | * | * | Monoterpenoid acid | [20] |
| 73 | α-Elemol | 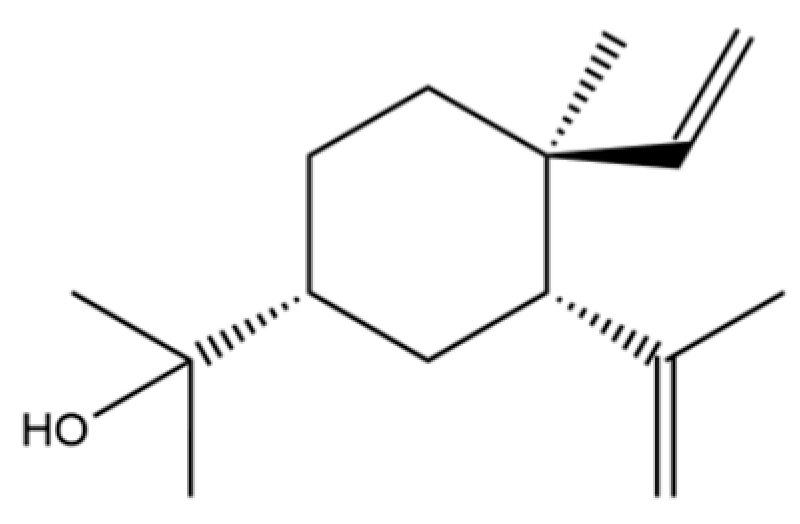 | * | * | Sesquiterpenoid | [31] |
| 74 | Neointermedeol | 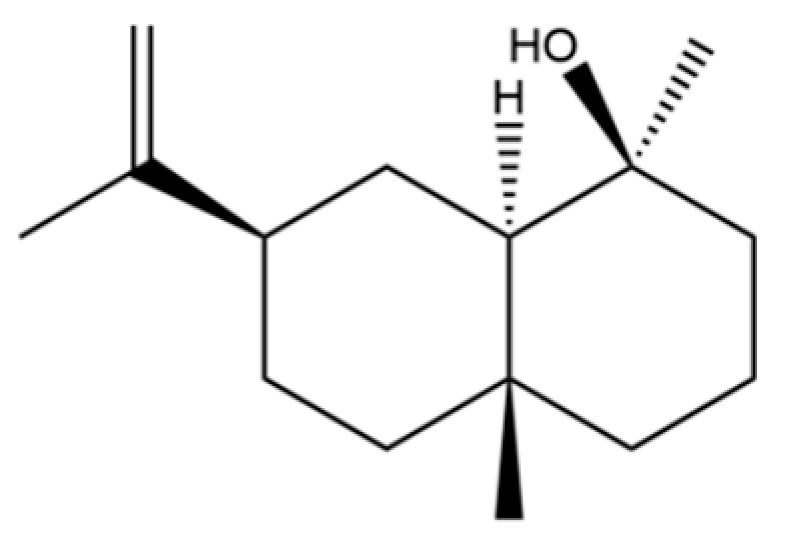 | * | * | Sesquiterpenoid | [20] |
| 75 | Dehydrofukinone | 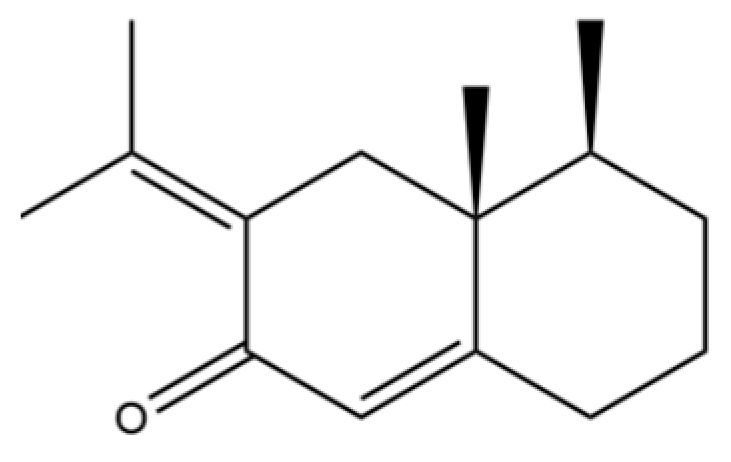 | * | * | Sesquiterpenoid | [20] |
| 76 | Isoaromadendrene epoxide | 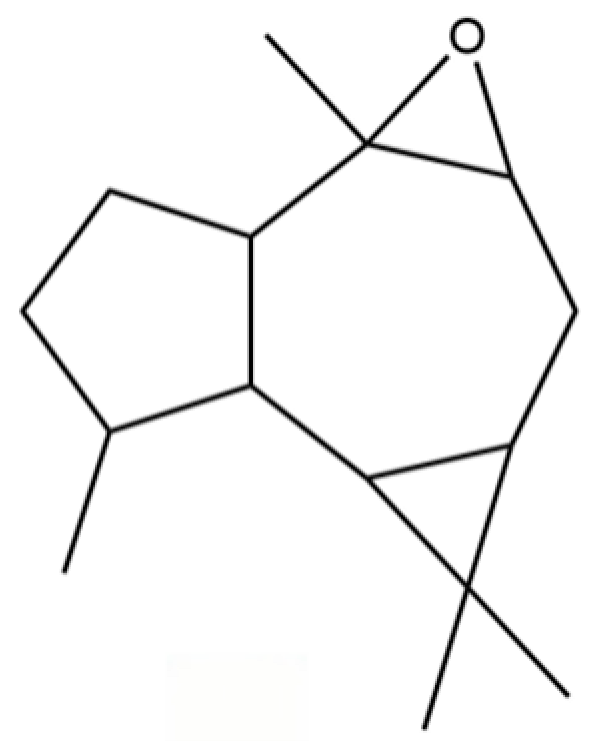 | * | * | Sesquiterpenoid | [20] |
| 77 | Juniper camphor | 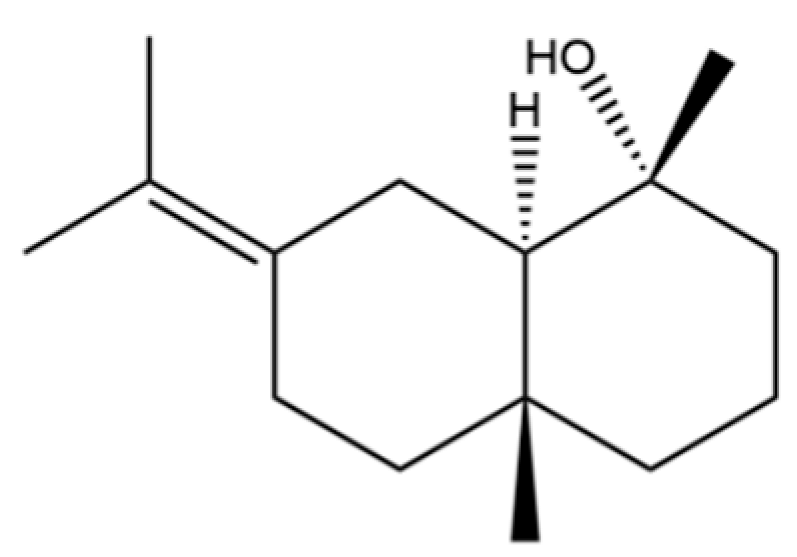 | * | * | Sesquiterpenoid | [3] |
| 78 | 2,4-Di-tert-butylphenol | 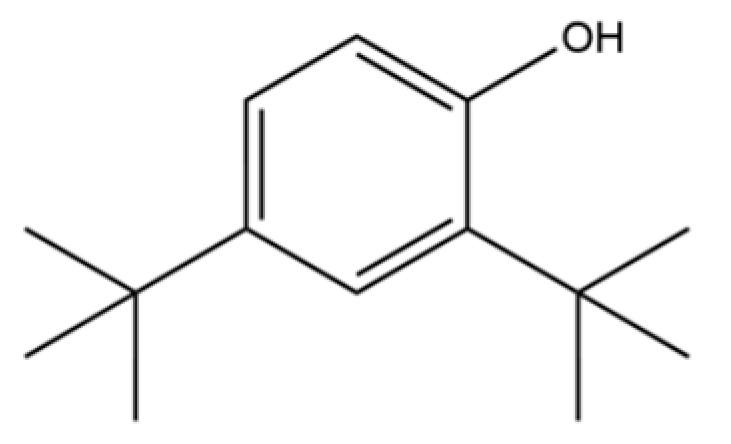 | * | * | Phenolic | [32] |
| 79 | 2 (1H) Naphthalenone, 3,5,6,7,8,8a-hexahydro-4,8a- | 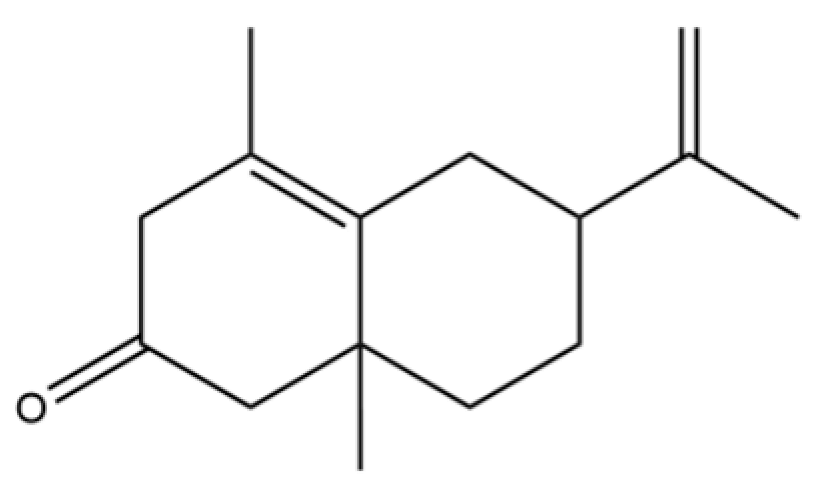 | * | * | Sesquiterpenoid | [20] |
| 80 | Aromadendrene oxide-(1) | 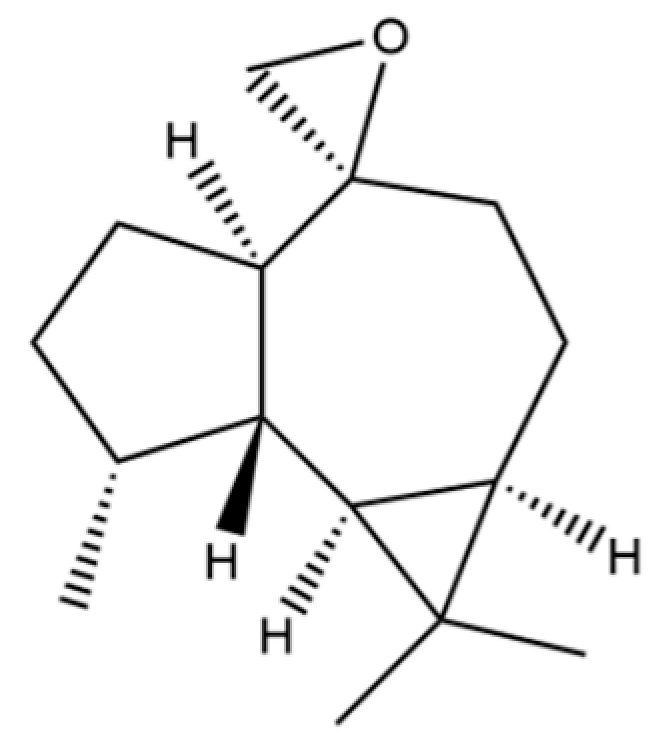 | * | * | Sesquiterpenoid | [20] |
| 81 | Valerenol | 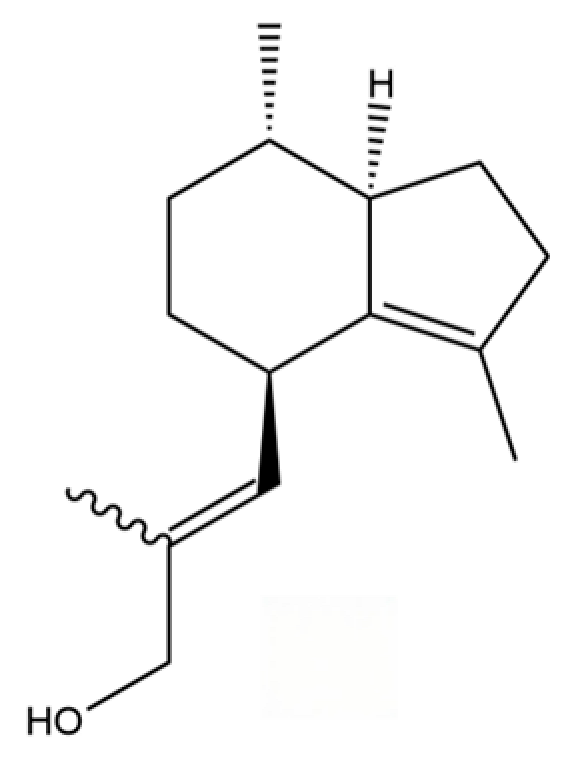 | * | * | Sesquiterpenoid | [20] |
| 82 | 3-Decenoic acid | 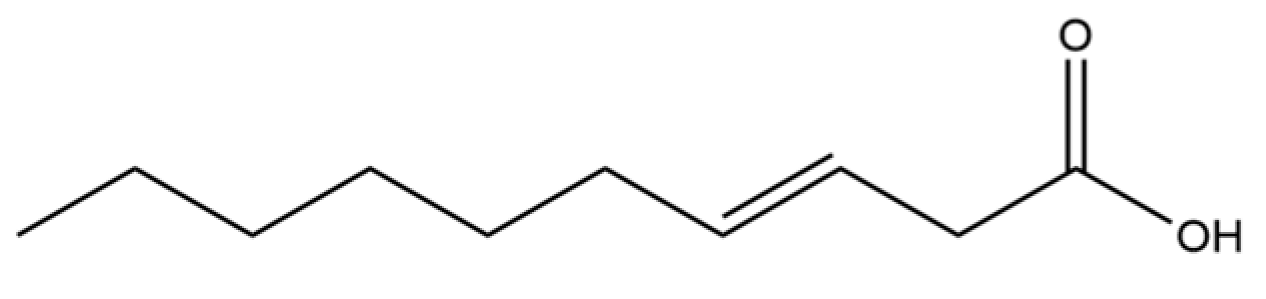 | * | * | Fatty acid | [20] |
| 83 | Heneicosane | 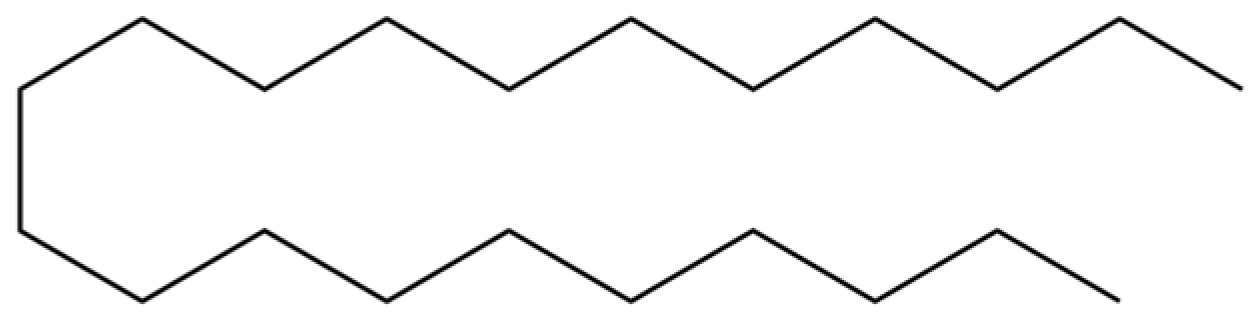 | * | * | Alkane | [26] |
| 84 | Spathulel | 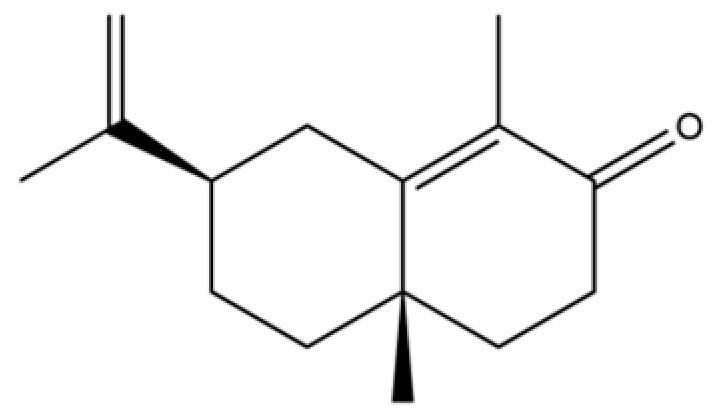 | * | * | Sesquiterpenoid | [20] |
| 85 | Diethyl Phthalate | 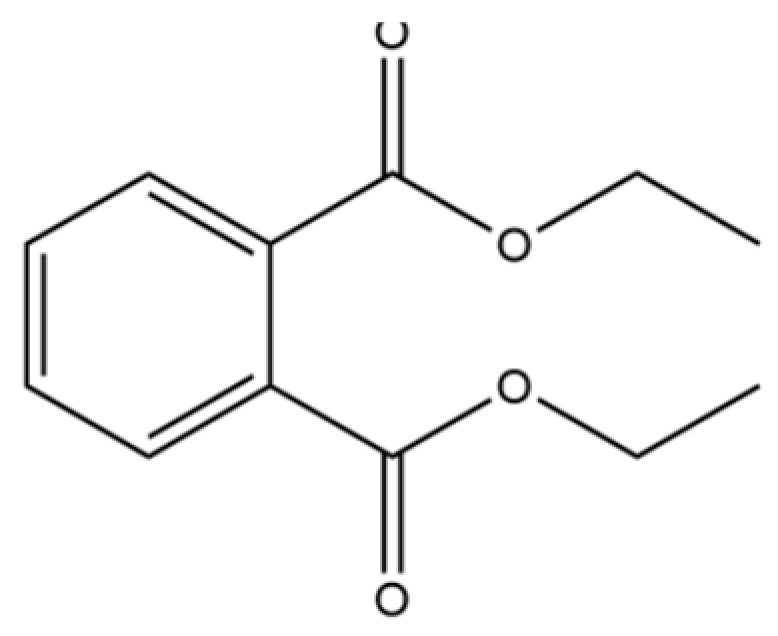 | * | * | Phthalate ester (artifact) | [33] |
| 86 | 1,1,4,7-Tetramethyldecahydro-1H-cyclopropa[e] azulene-4,7-diol | 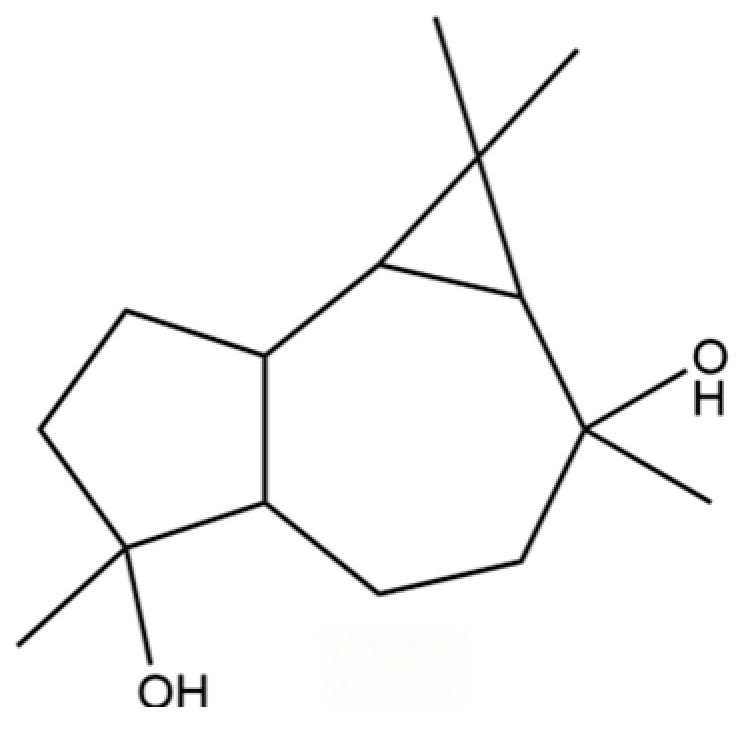 | * | * | Sesquiterpenoid | [20] |
| 87 | α-Serinene | 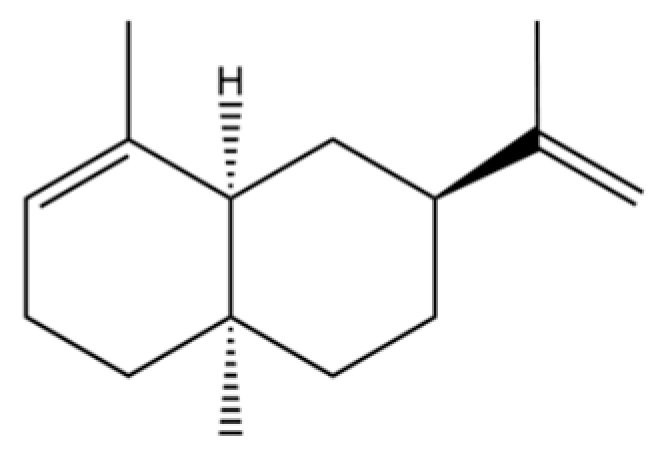 | * | * | Sesquiterpenoid | [20] |
| 88 | Caryophyllene oxide | 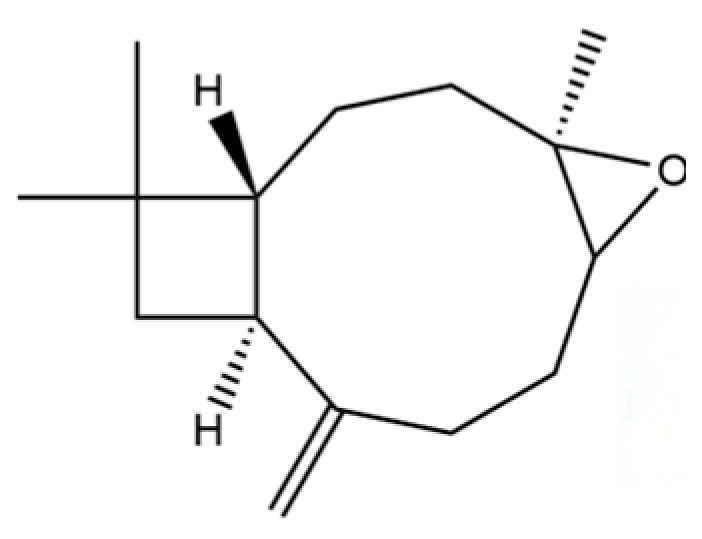 | * | * | Sesquiterpenoid | [21] |
| 89 | trans-9-Hexadecen-1-ol |  | * | * | Fatty alcohol | [20] |
| 90 | Kaur-16-ene |  | * | * | Diterpenoid | [20] |
| 91 | 3-Phenyltoluene |  | * | * | Aromatic hydrocarbon | [20] |
| 92 | 4a, 7-Methano-4aH-naphth[1,8a-b] oxirene, octahydro-4,4,8,8-tetramethyl- |  | * | * | Sesquiterpenoid | [20] |
| 93 | (1R,7S, E)-7-Isopropyl-4,10-dimethylenecyclodec-5-enol | 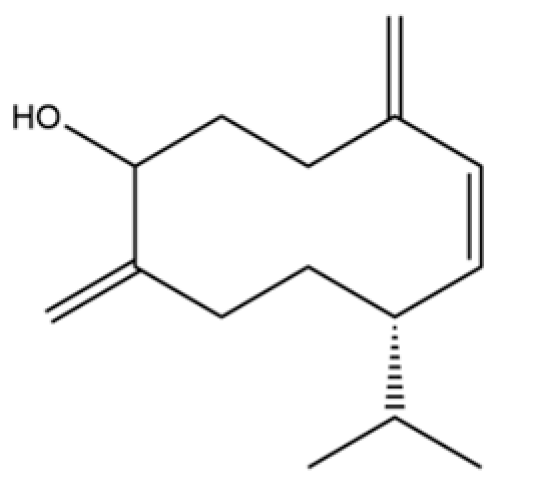 | * | * | Sesquiterpenoid | [20] |
| 94 | Acetic acid n-octadecyl ester | 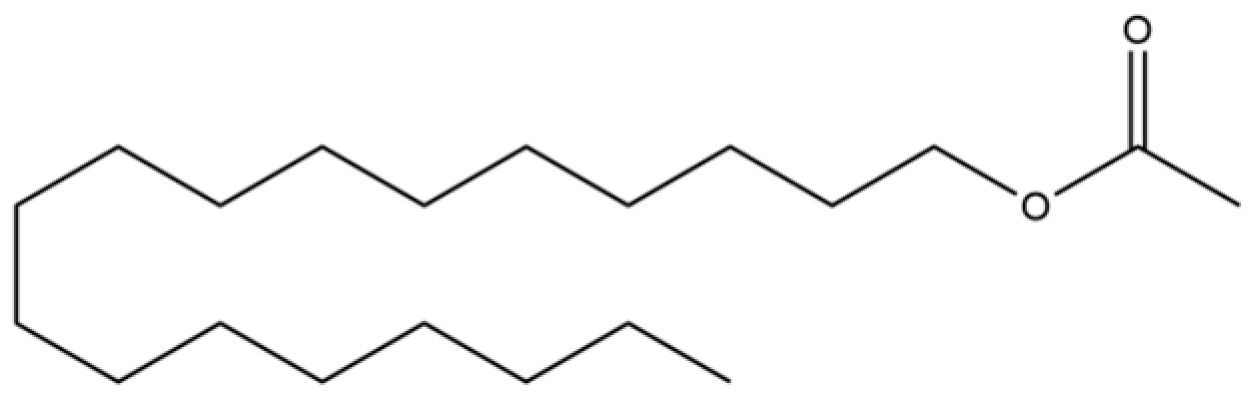 | * | * | Fatty ester | [20] |
| 95 | Methyl 10-trans, 12-cis- octadecadienoate | 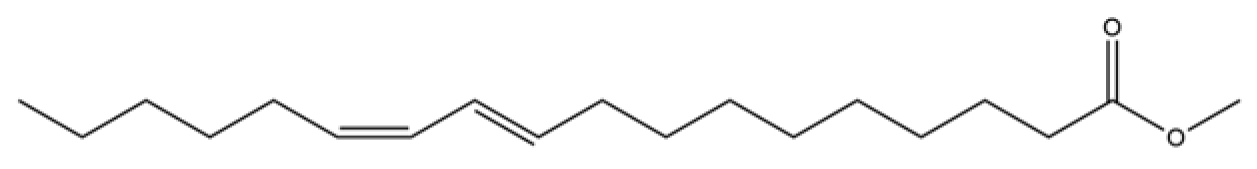 | * | * | Fatty acid methyl ester | [20] |
| 96 | Oplopane | 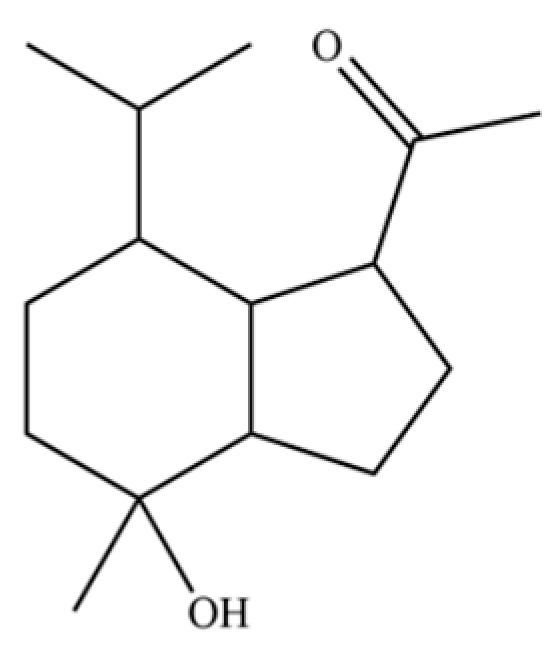 | * | * | Sesquiterpenoid | [20] |
| 97 | Diisobutyl phthalate | 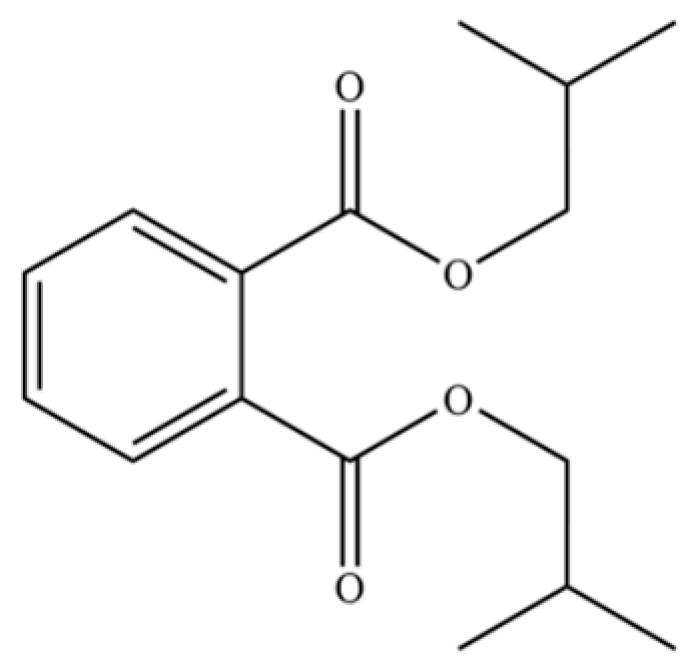 | * | * | Phthalate ester (artifact) | [20] |
| 98 | 2aS,3aR,5aS,9bR)-2a, 5a, 9-Trimethyl-2a, 4,5,5a, | 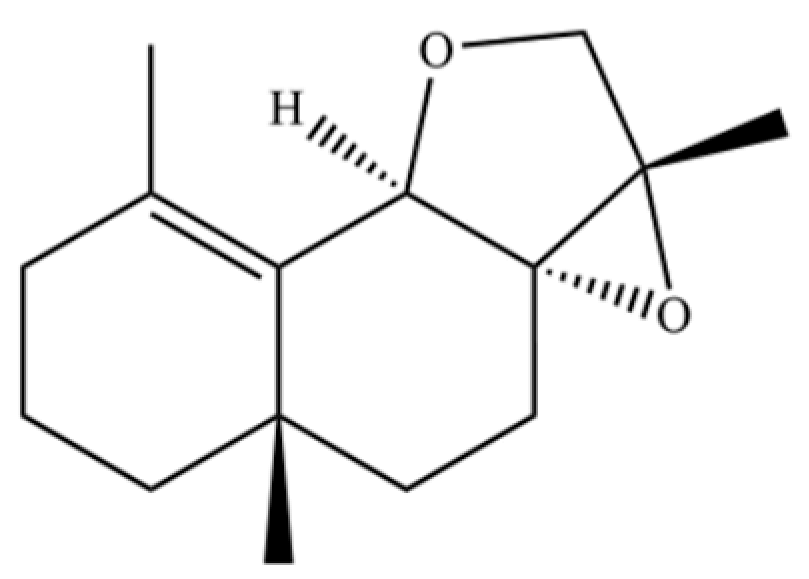 | * | * | Sesquiterpenoid | [20] |
| 99 | Costol | 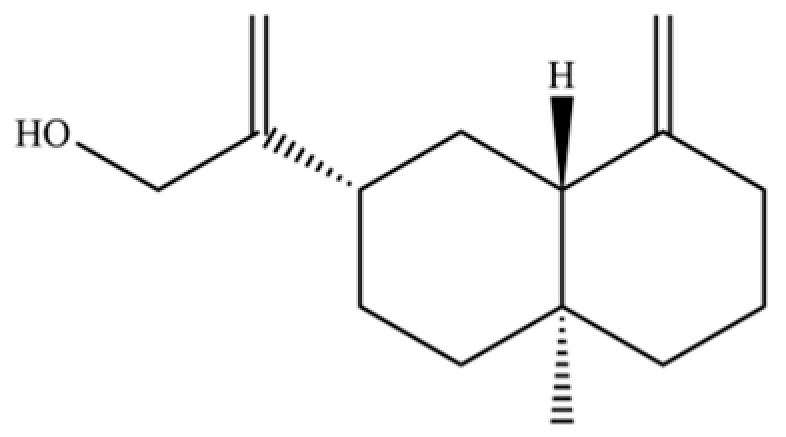 | * | * | Sesquiterpenoid | [20] |
| 100 | Octacosane | 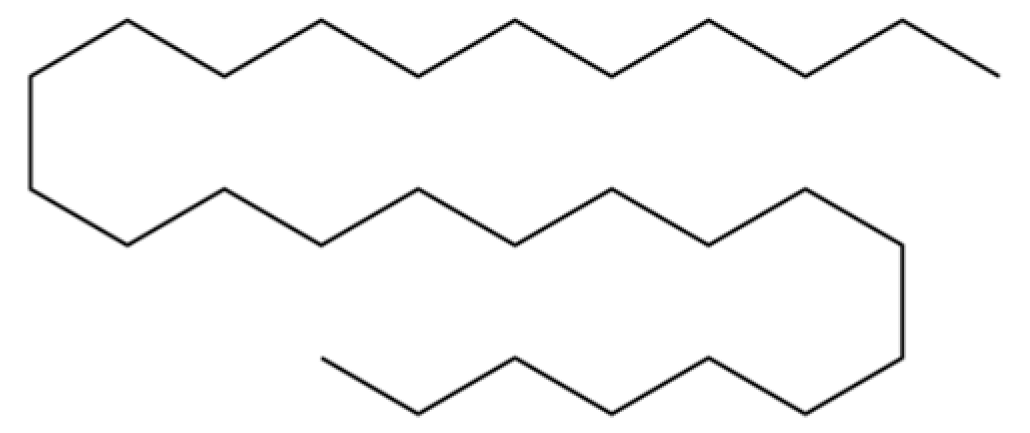 | * | * | Alkane | [20] |
| 101 | 1-Eicosanol |  | * | * | Fatty alcohol | [20] |
| 102 | Valerenyl isovalerate | 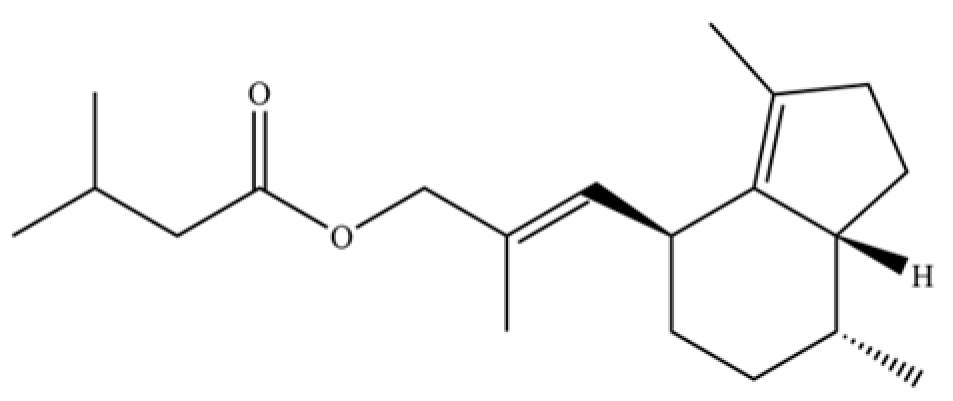 | * | * | Sesquiterpenoid ester | [20] |
| 103 | longifolene-(V4) | 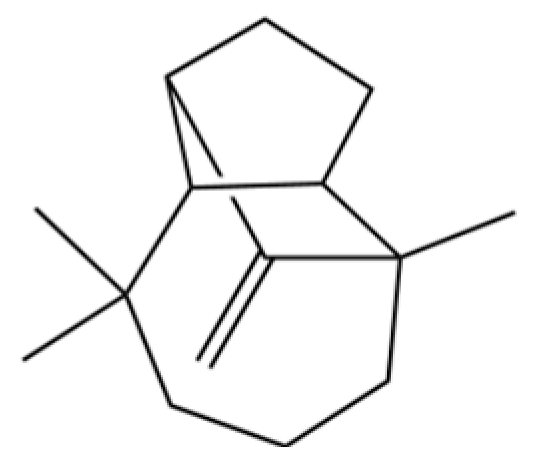 | * | * | Sesquiterpenoid | [25] |
| 104 | Cryptomeridiol | 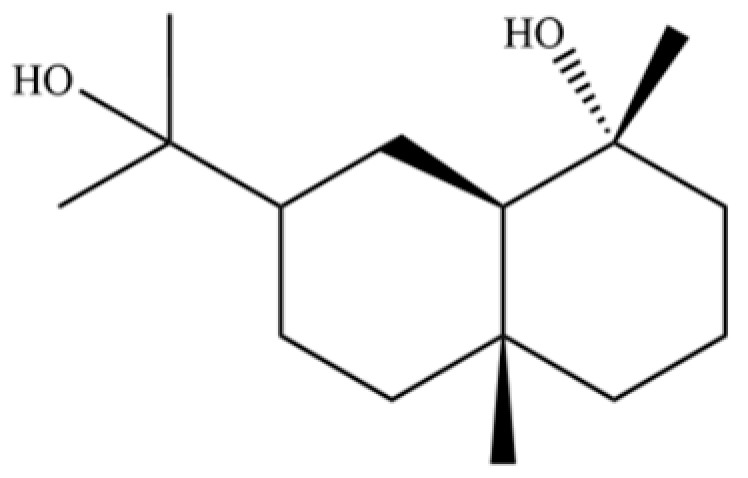 | * | * | Sesquiterpenoid | [20] |
| 105 | Spathulenol | 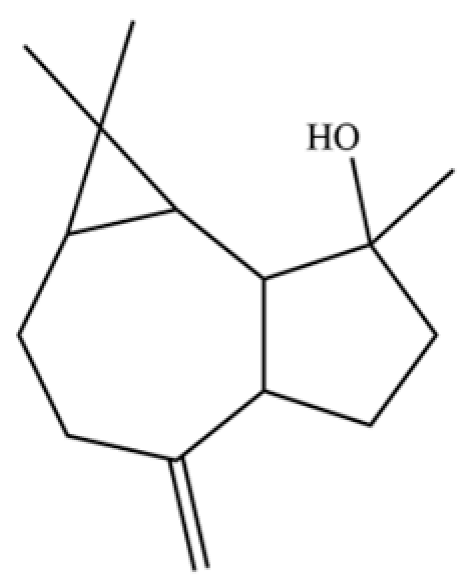 | * | * | Sesquiterpenoid | [25] |
| 106 | Bicyclo[4.4.0]dec-5-ene, 1,5-dimethyl-3-hy-droxy-8-(1-methylene-2-hydroxyethyl-1)- | 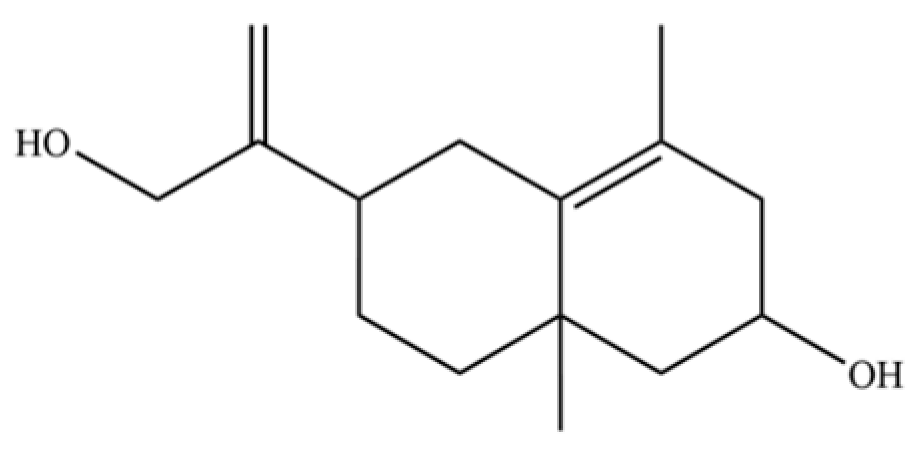 | * | * | Sesquiterpenoid | [20] |
| 107 | 6-Isopropenyl-4,8a-dimethyl-1,2,3,5,6,7,8,8a-octahydronaphthalene-2,3-diol | 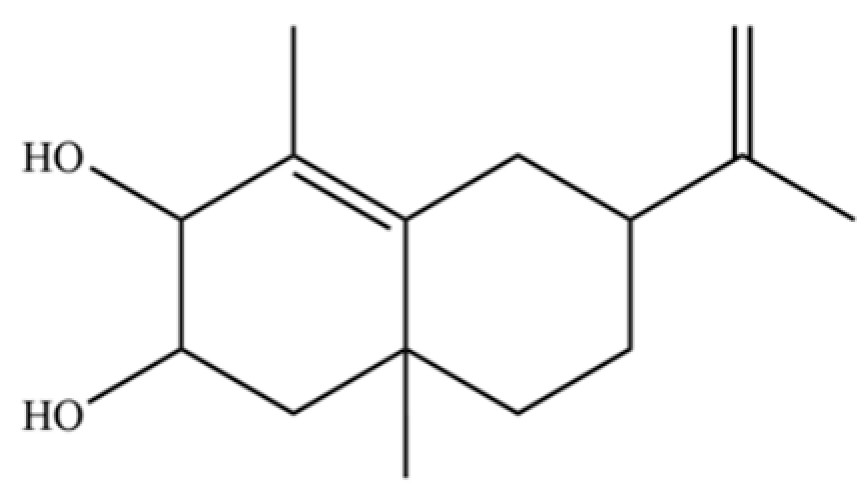 | * | * | Sesquiterpenoid | [20] |
| 108 | n-Hexadecanoic acid | 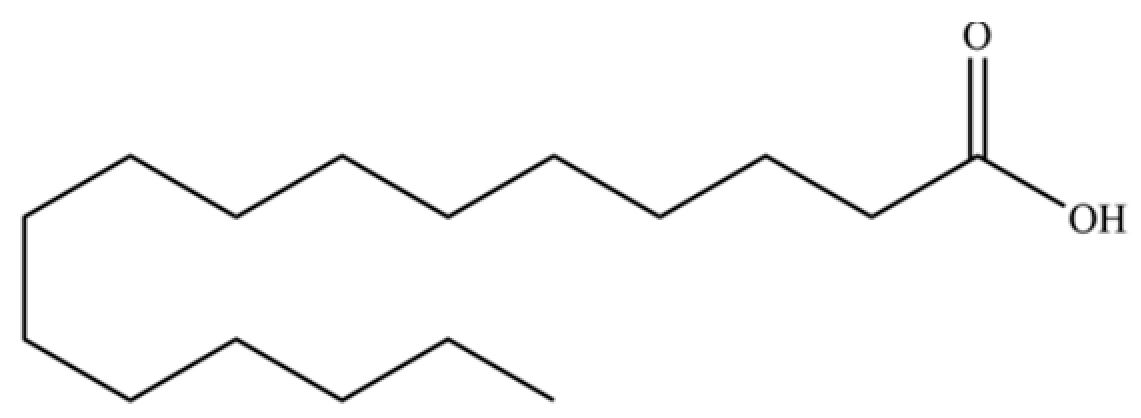 | * | * | Fatty acid | [20] |
| 109 | Atractylolide | 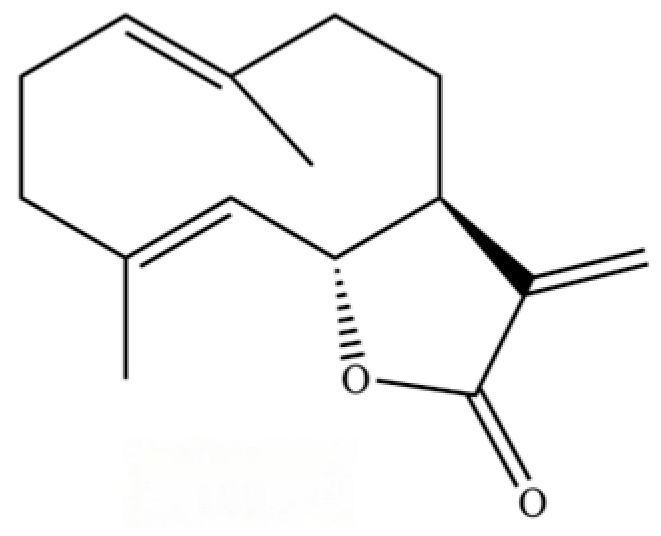 | * | * | Sesquiterpenoid lactone | [34] |
| 110 | Tetratetracontane |  | * | * | Alkane | [20] |
| 111 | α-Cyperone |  | * | * | Sesquiterpenoid | [20] |
| 112 | Squalene |  | * | * | Triterpenoid | [35] |
| 113 | 6-Octadecenoic acid |  | * | * | Fatty acid | [20] |
| 114 | Bicyclo[3.1.0]hexane, 4-methylene-1-(1-methylethyl)- | 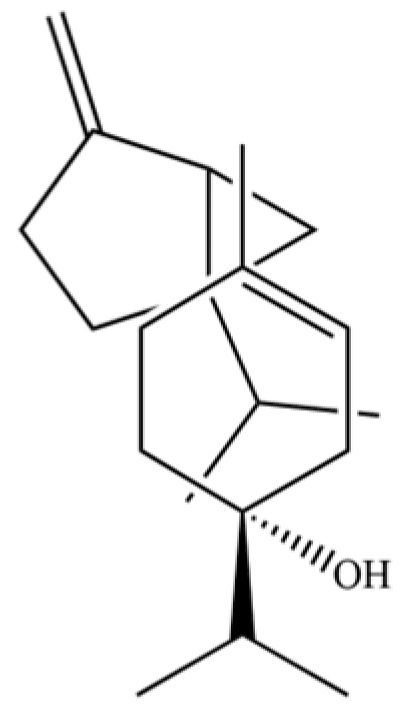 | * | * | Monoterpenoid | [36] |
| 115 | Eucalyptol | 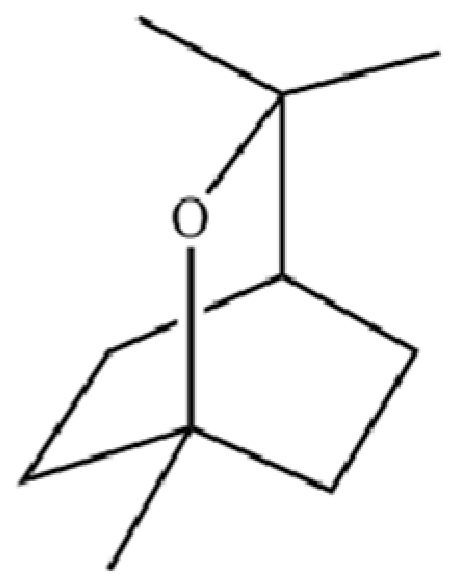 | * | * | Monoterpenoid | [37] |
| 116 | γ-Terpinene | 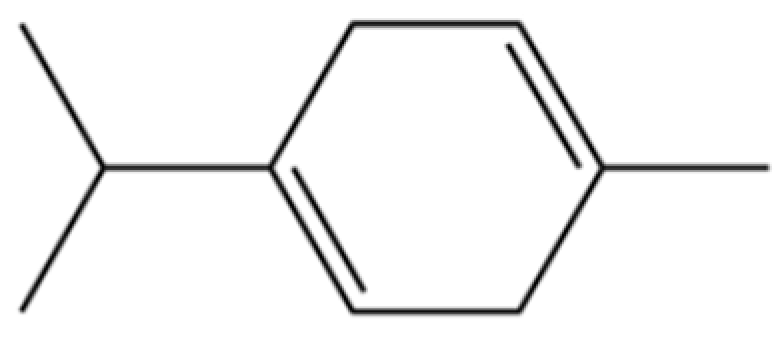 | * | * | Monoterpenoid | [26] |
| 117 | Linalool | 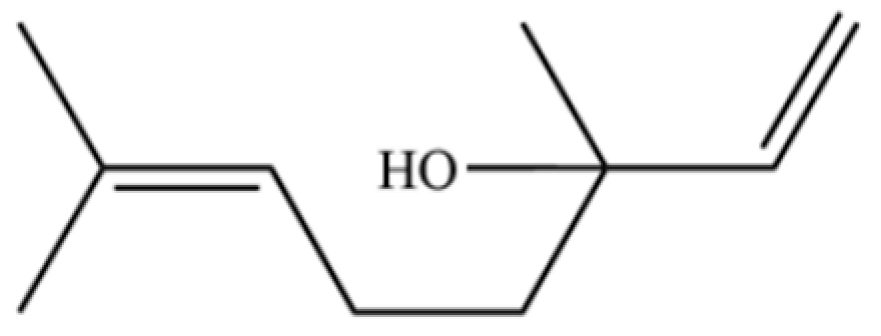 | * | * | Monoterpenoid | [38] |
| 118 | 1,5-Cyclodecadiene, 1,5-dimethyl-8-(1-methylethylidene)-, (E,E)- | 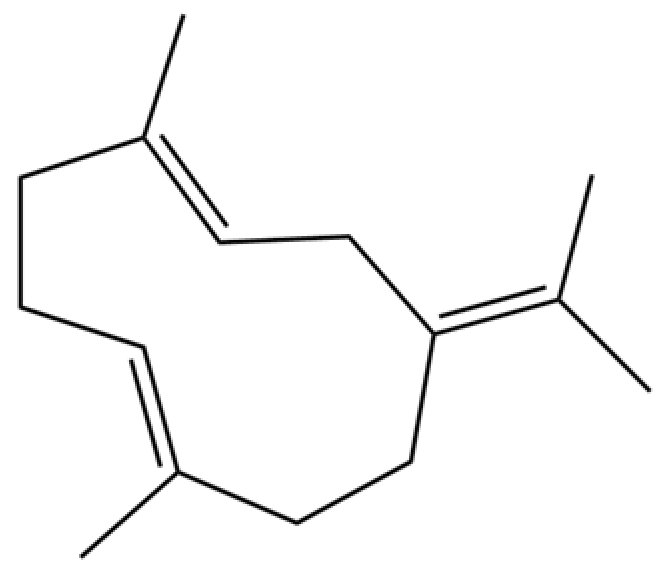 | * | * | Sesquiterpenoid | [36] |
| 119 | β-Pinene | 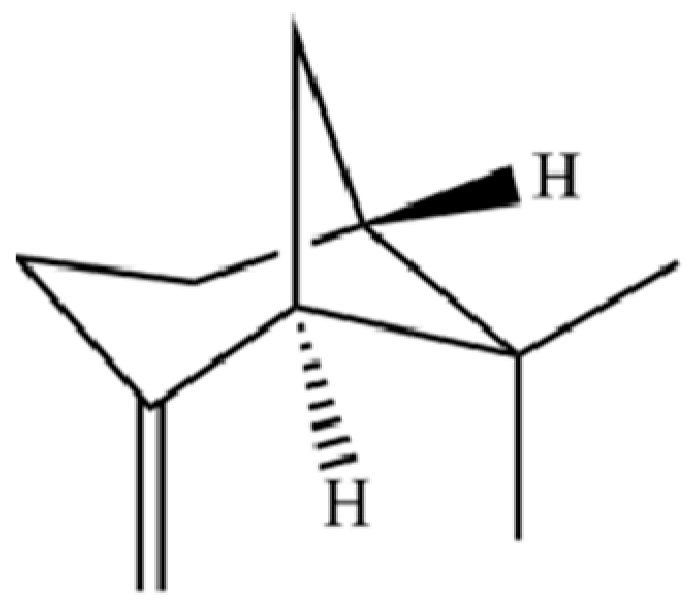 | * | * | Monoterpenoid | [21] |
| 120 | β-Phellandrene | 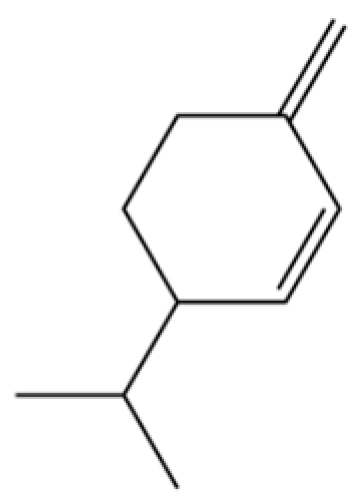 | * | * | Monoterpenoid | [21] |
| 121 | Camphene | 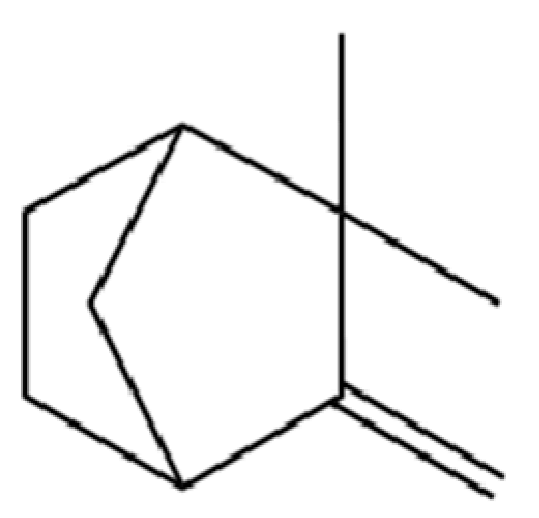 | * | * | Monoterpenoid | [36] |
| 122 | β-Bisabolene |  | * | * | Sesquiterpenoid | [18] |
| 123 | Benzaldehyde |  | * | * | Aromatic aldehyde | [39] |
| 124 | (S)-(+)-alpha-Phellandrene |  | * | * | Monoterpenoid | [36] |
| 125 | Citronellyl acetate |  | * | * | Monoterpenoid ester | [40] |
| 126 | (-)-Bornyl acetate |  | * | * | Monoterpenoid ester | [21] |
| 127 | Bicyclo[3.1.1]heptane, 6,6-dimethyl-2-methylene-, (1S)- |  | * | * | Monoterpenoid | [25] |
| 128 | Tricyclo[2.2.1.0(2,6)]heptane, 1,7,7-trimethyl- |  | * | * | Monoterpenoid | [36] |
| 129 | Acetophenone, 4′-hydroxy- |  | * | * | Phenolic ketone | [36] |
| 130 | (+)-2-Bornanone | 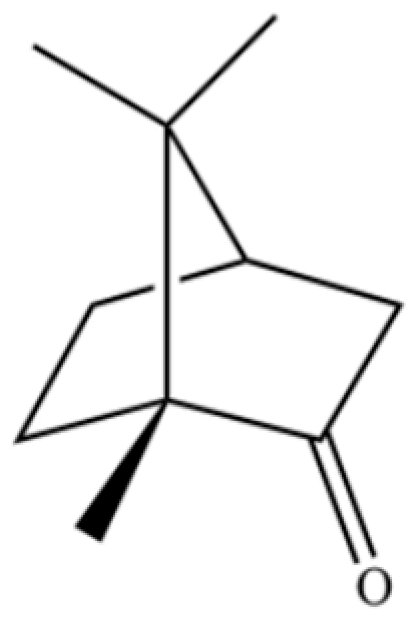 | * | * | Monoterpenoid ketone | [36] |
| 131 | 3-Cyclohexen-1-ol, 4-methyl-1-(1-methylethyl)-, (R)- | 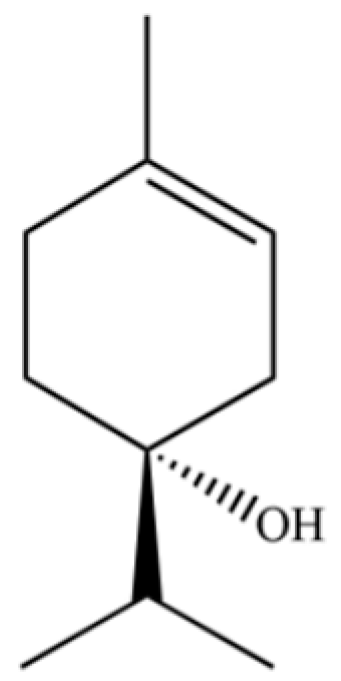 | * | * | Monoterpenoid | [36] |
| 132 | 2(1H)-Pyridinone | 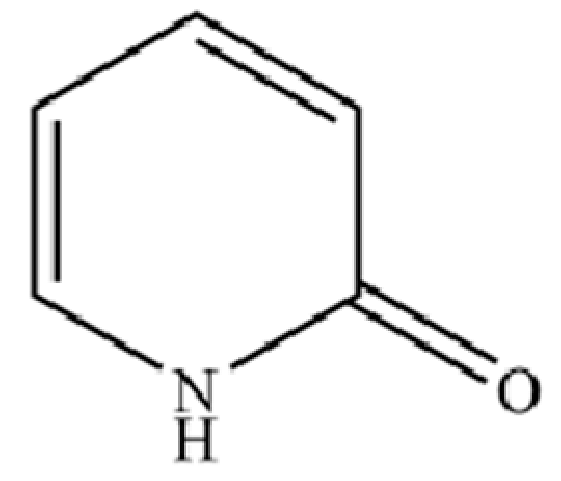 | * | * | PHeterocyclic compound | [36] |
| 133 | Acetic acid, 4-methylphenyl ester | 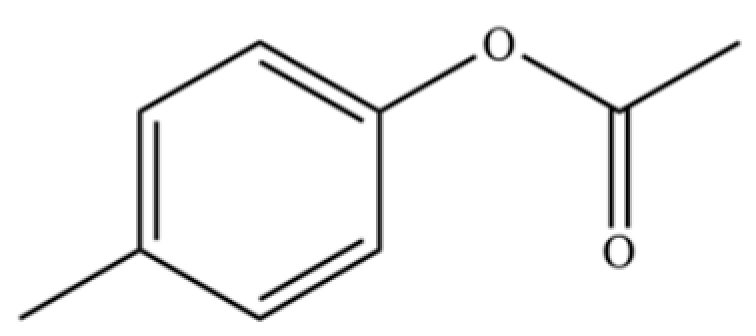 | * | * | Phenolic ester | [36] |
| 134 | L-α-Terpineol | 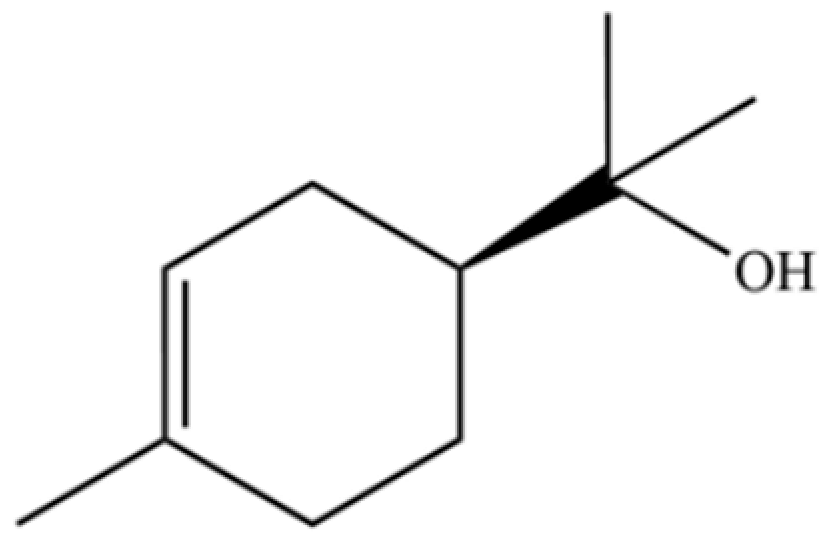 | * | * | Monoterpenoid | [36] |
| 135 | 3-Methyl-4-isopropylphenol | 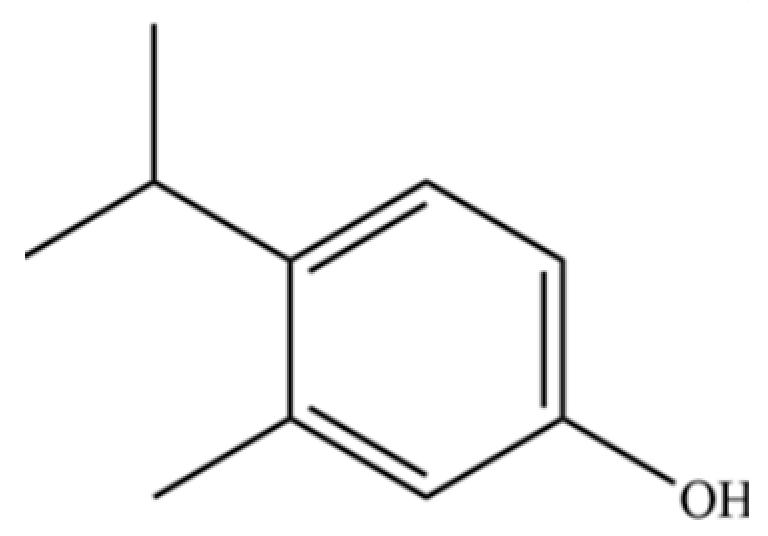 | * | * | Phenolic | [41] |
| 136 | Isoelemicin | 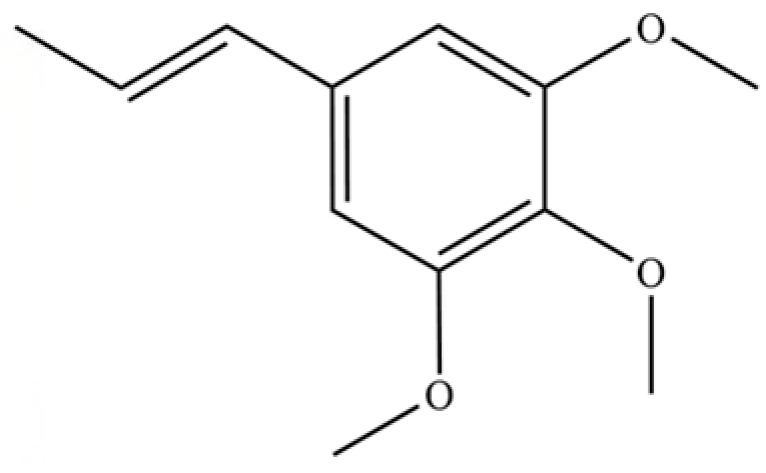 | * | * | Phenylpropanoid | [36] |
| 137 | Furanodienone | 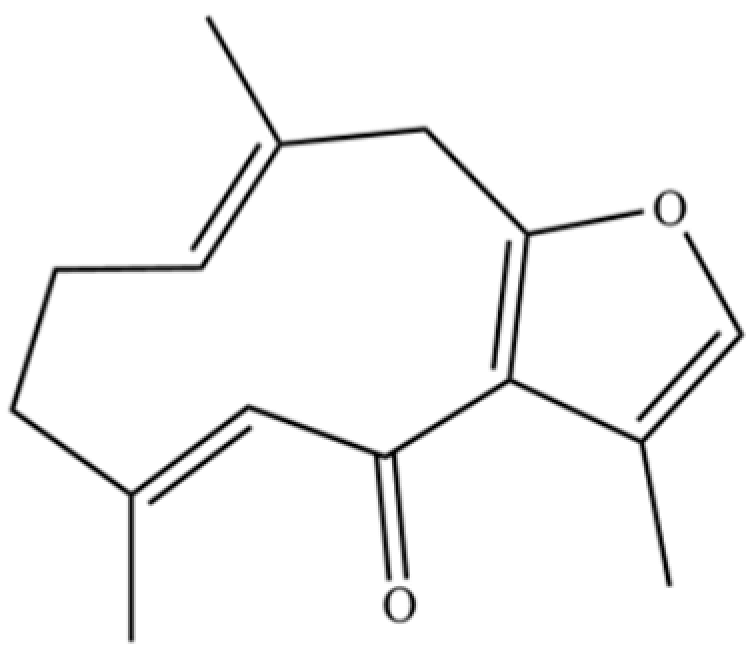 | * | * | Sesquiterpenoid | [36] |
| 138 | (1S)-2,6,6-Trimethylbicyclo[3.1.1]hept-2-ene | 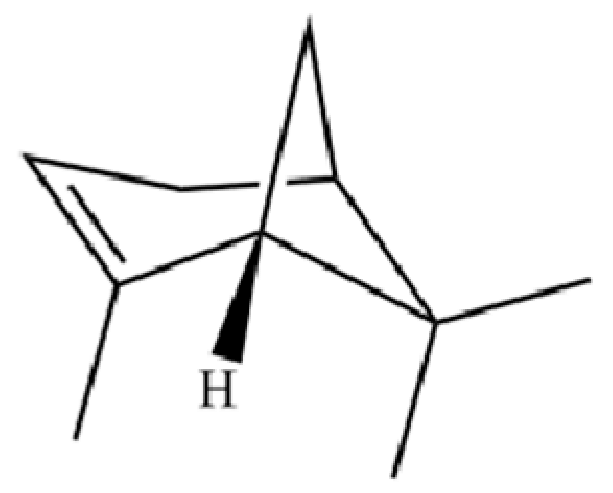 | * | * | Monoterpenoid | [36] |
| 139 | Ethanone, 1-(2,4,6-trihydroxyphenyl)- | 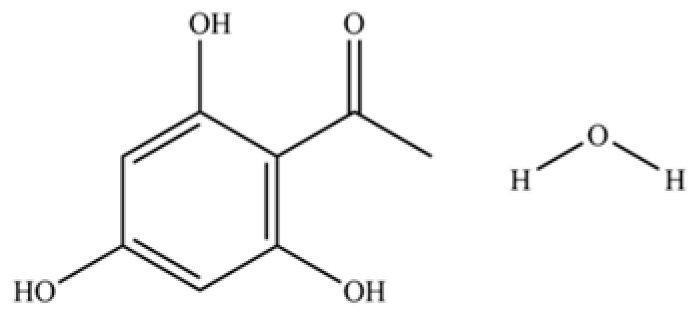 | * | * | Phenolic ketone | [36] |
| 140 | Phenol, 2,3,6-trimethyl- | 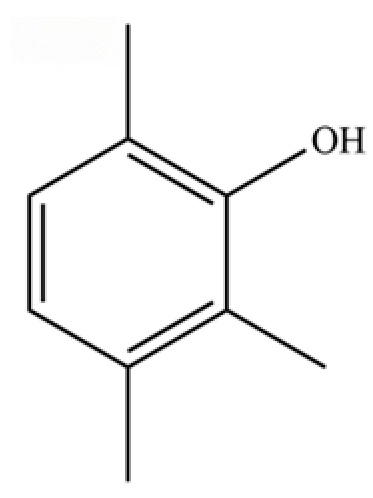 | * | * | Phenolic | [36] |
| 141 | Atractylodin | 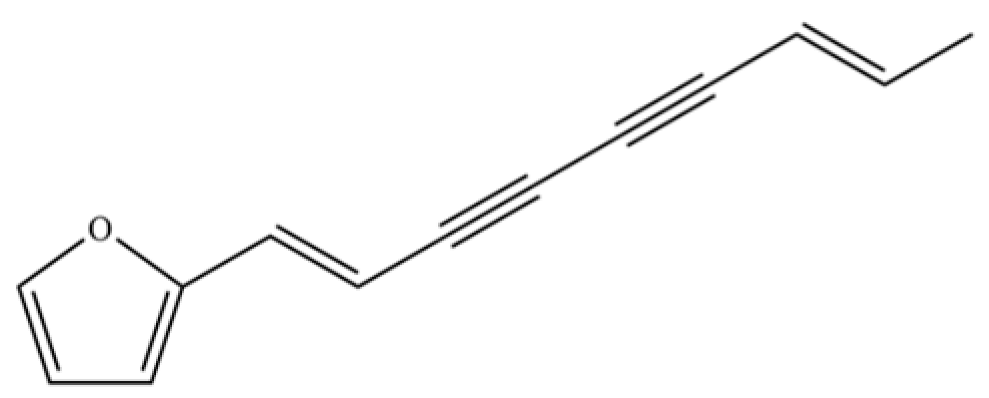 | ** | * | Polyacetylene | [21] |
| 142 | 3-Methyl-3-buten-1-ol, acetate | 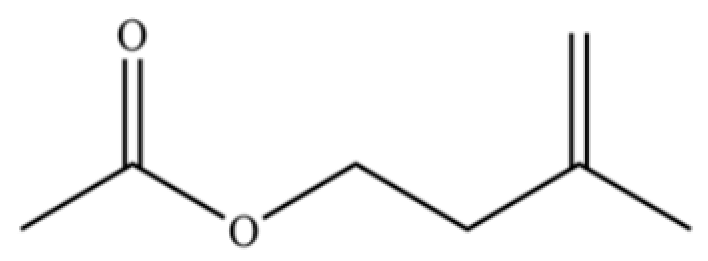 | * | * | Terpene alcohol ester | [36] |
| 143 | α-Thujene | 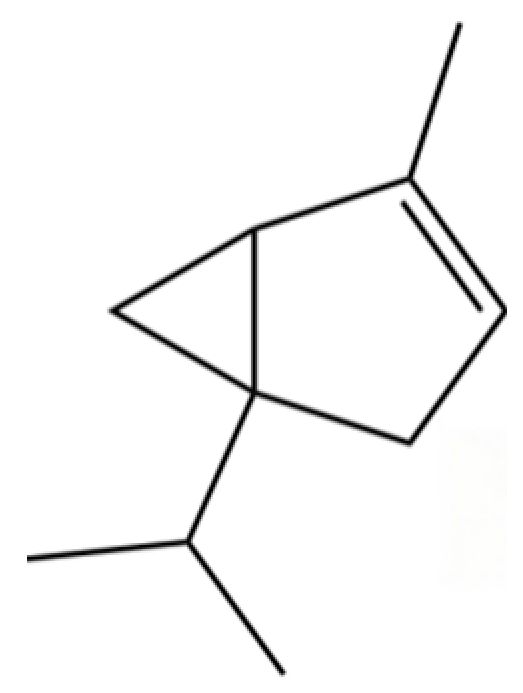 | * | * | Monoterpenoid | [42] |
| 144 | 3-Carene | 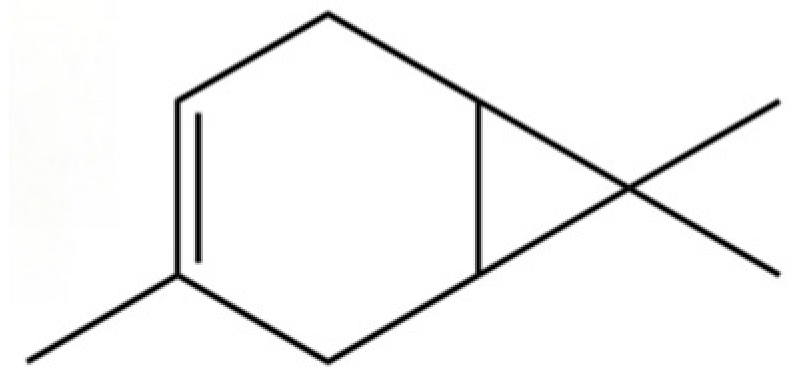 | * | * | Monoterpenoid | [21] |
| 145 | 1,3,8-p-Menthatriene | 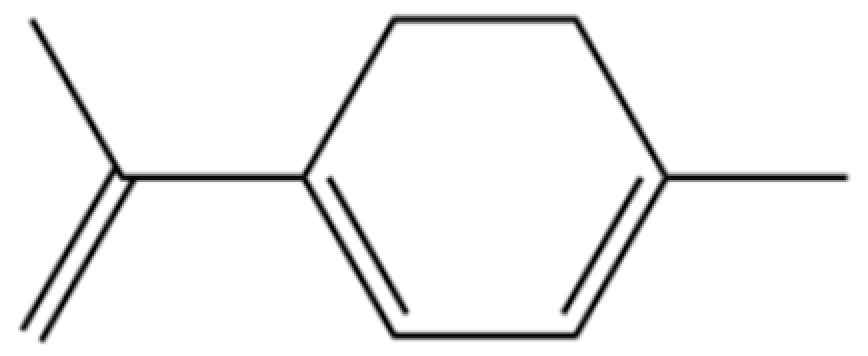 | * | * | Monoterpenoid | [36] |
| 146 | Carveol | 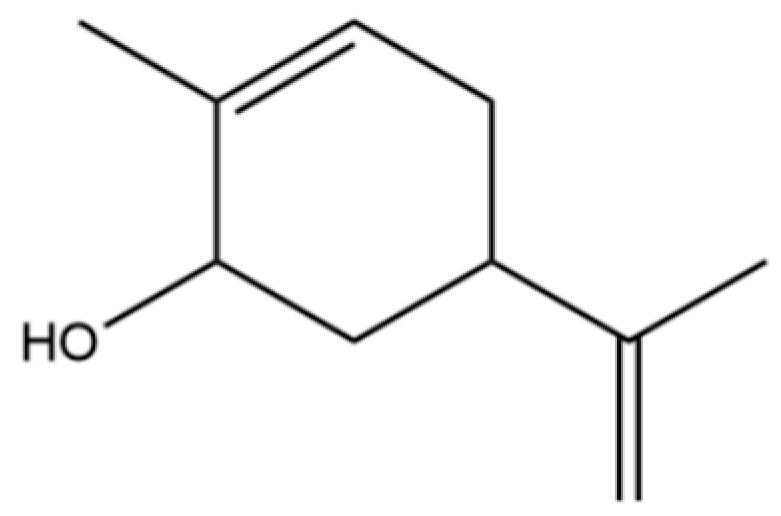 | * | * | Monoterpenoid | [36] |
| 147 | Dill ether | 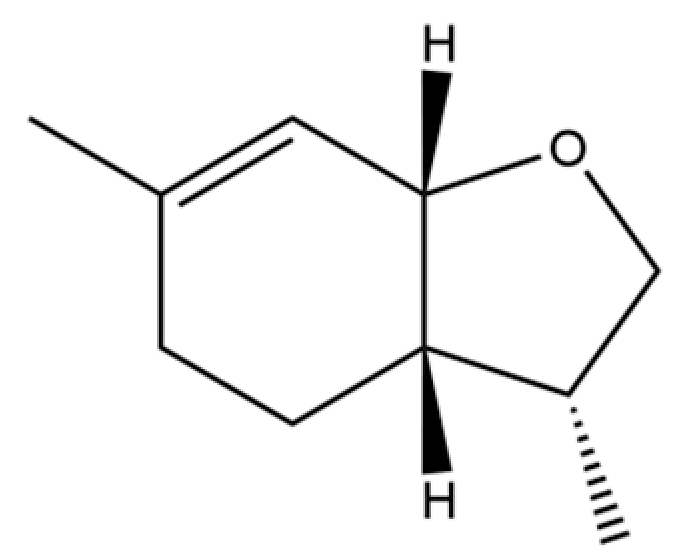 | * | * | Monoterpenoid ether | [36] |
| 148 | trans-Geranic acid methyl ester | 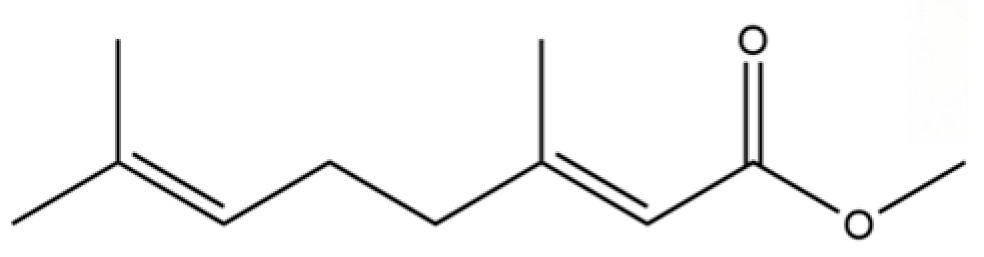 | * | * | Monoterpenoid ester | [36] |
| 149 | 3a,7-Methano-3aH-cyclopentacyclooctene, 1,4,5,6,7,8,9,9a-octahydro-1,1,7-trimethyl-, [3aR-(3a.α,7.α,9a.β)]- | 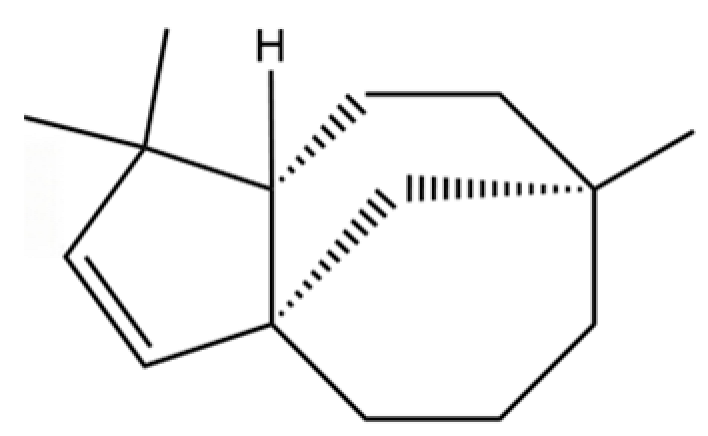 | * | * | Sesquiterpenoid | [36] |
| 150 | 2,6-Octadien-1-ol, 3,7-dimethyl-, acetate, (Z)- | 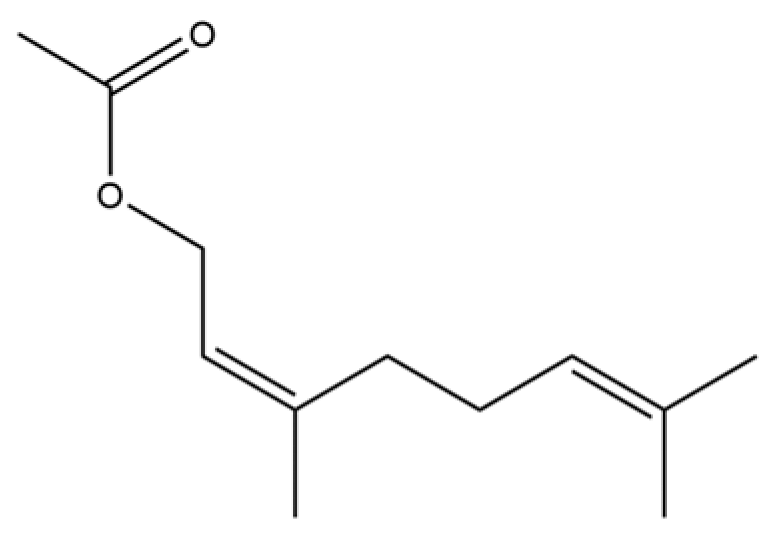 | * | * | Monoterpenoid ester | [36] |
| 151 | Acetic acid, decyl ester | 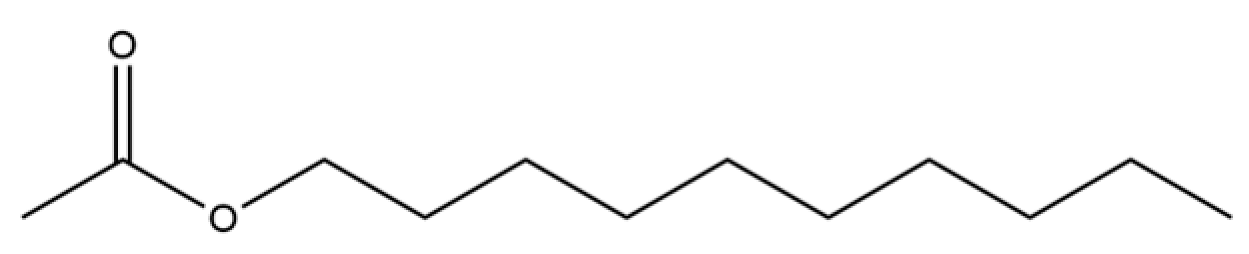 | * | * | Fatty ester | [36] |
| 152 | (+)-4-Carene | 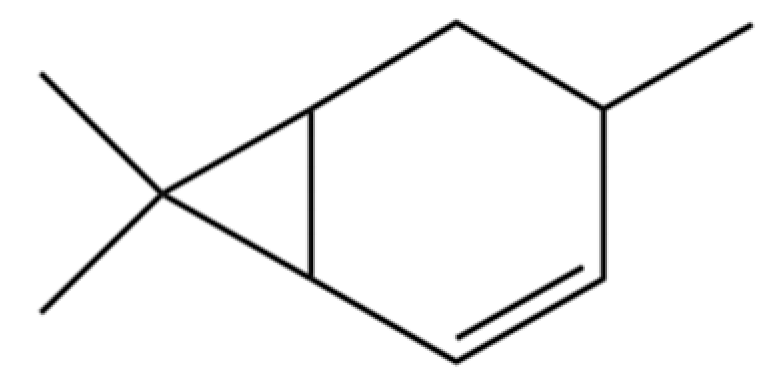 | * | * | Monoterpenoid | [25] |
| 153 | γ-Muurolene | 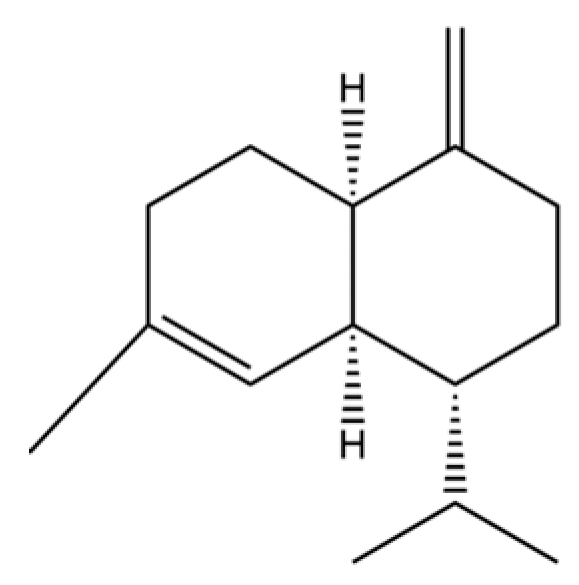 | * | * | Sesquiterpenoid | [43] |
| 154 | (3aR,4R,7R)-1,4,9,9-Tetramethyl-3,4,5,6,7,8-hexahydro-2H-3a,7-methanoazulen-2-one | 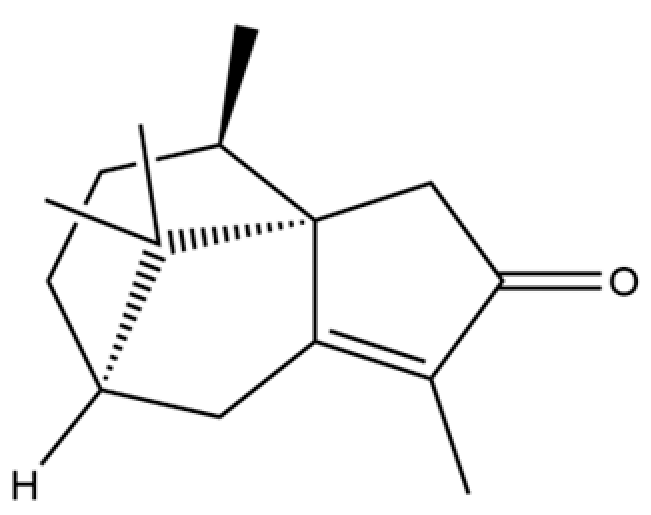 | * | * | Sesquiterpenoid | [36] |
| 155 | Benzimidazo[2,1-a]isoquinoline | 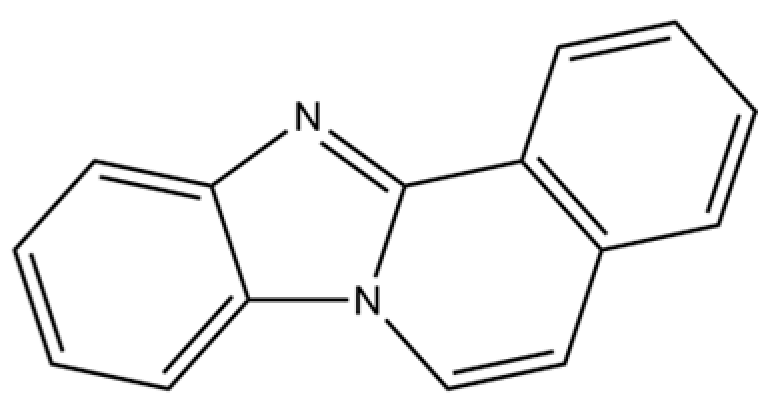 | * | * | Heterocyclic compound | [36] |
| 156 | (4aR,8aR)-5,8a-Dimethyl-3-propan-2-ylidene-1,2,4,4a,7,8-hexahydronaphthalene | 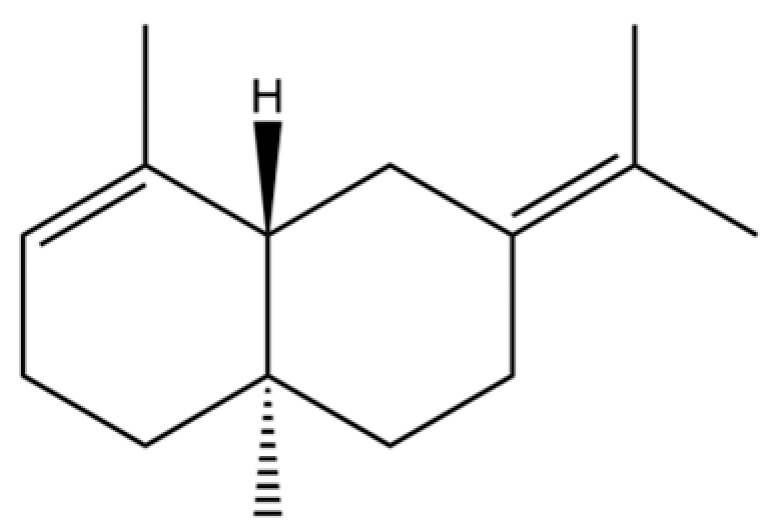 | * | * | Sesquiterpenoid | [36] |
| 157 | (1R,3aS,8aS)-7-Isopropyl-1,4-dimethyl-1,2,3,3a,6,8a-hexahydroazulene | 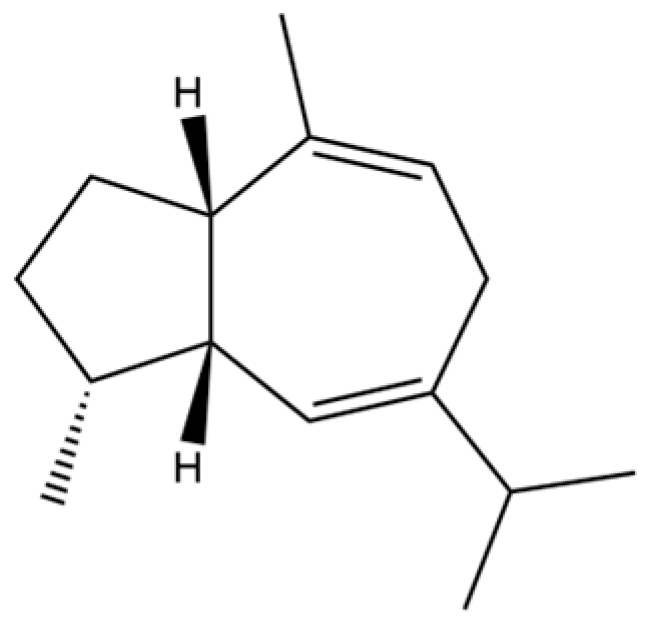 | * | * | Sesquiterpenoid | [36] |
| 158 | Thujopsene |  | * | * | Sesquiterpenoid | [25] |
| 159 | p-Mentha-1,5,8-triene |  | * | * | Monoterpenoid | [36] |
| 160 | 1-Hexen, 2-(p-anisyl)-5-methyl- |  | * | * | Aromatic hydrocarbon | [36] |
| 161 | 6,7-Dimethyl-1,2,3,5,8,8a-hexahydronaphthalene |  | * | * | Sesquiterpenoid | [36] |
| 162 | 2,3-Dimethoxy-5-aminocinnamonitrile | 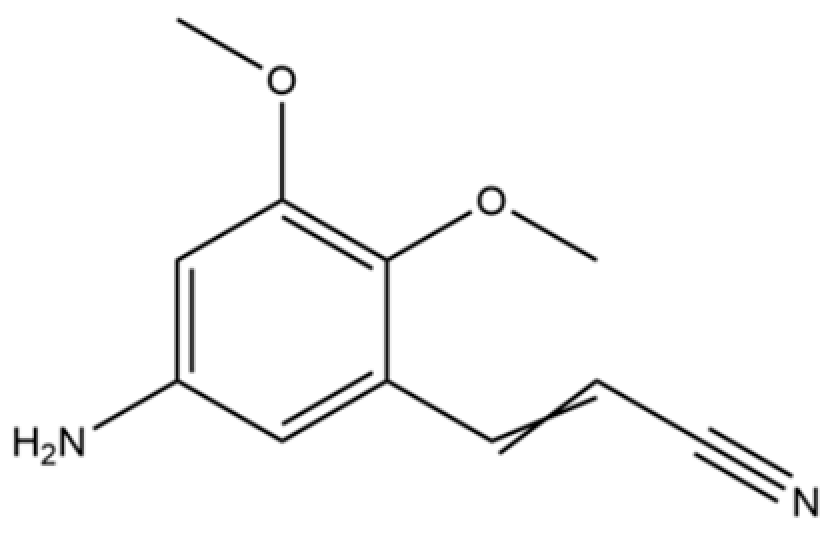 | * | * | Aromatic nitrile | [36] |
| 163 | 1-Penten-3-one, 1-(4-methoxyphenyl)-4-methyl- | 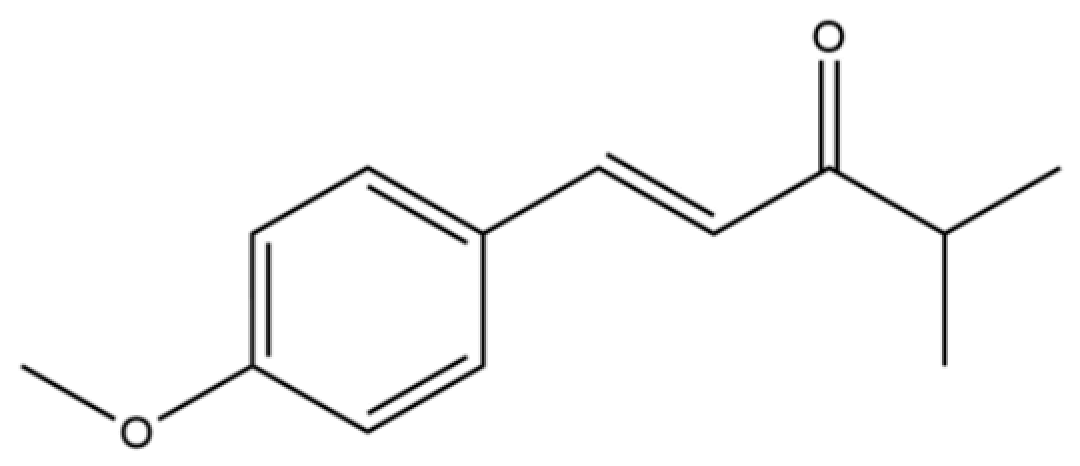 | * | * | Aromatic ketone | [36] |
| 164 | 4a(2H)-Naphthalenol, 1,3,4,5,6,8a-Hexahydro-4,7-dimethyl-1-(1-methylethyl)-, (1S,4S,4aS,8aR)- | 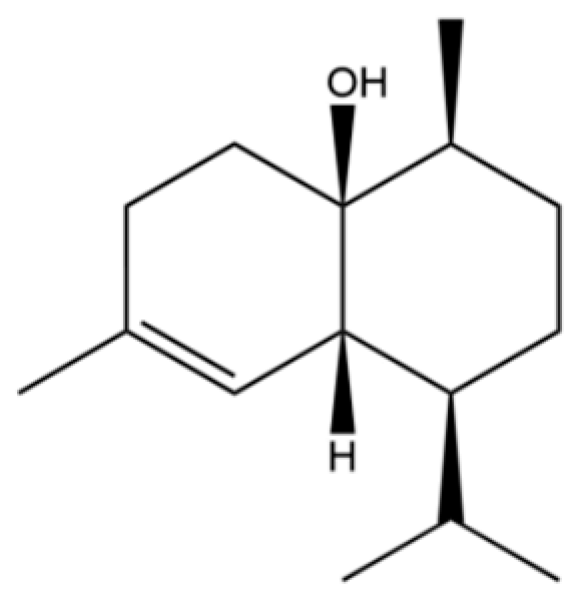 | * | * | Sesquiterpenoid | [36] |
| 165 | Neoisolongifolene, 8,9-dehydro- | 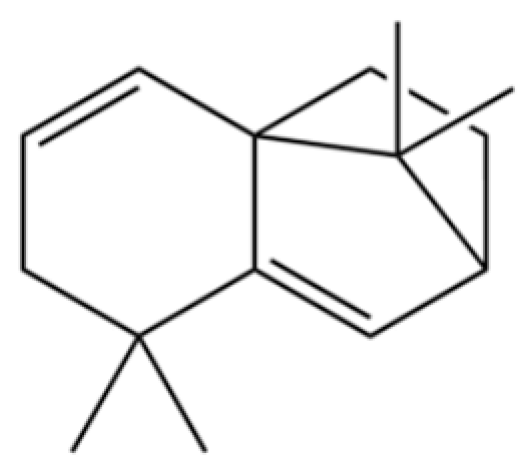 | * | * | Sesquiterpenoid | [36] |
| 166 | 6-Isopropenyl-4,8a-dimethyl-1,2,3,5,6,7,8,8a-octahydro-2-naphthalenyl acetate |  | * | * | Sesquiterpenoid ester | [36] |
| 167 | Bicyclo[3.1.0]hex-2-ene, 4-methylene-1-(1-methylethyl)- |  | * | * | Monoterpenoid | [36] |
| 168 | Benzene, tert-butyl- |  | * | * | Aromatic hydrocarbon | [36] |
| 169 | 1-Decen-3-one |  | * | * | Ketone | [36] |
| 170 | Silphiperfol-5-ene | 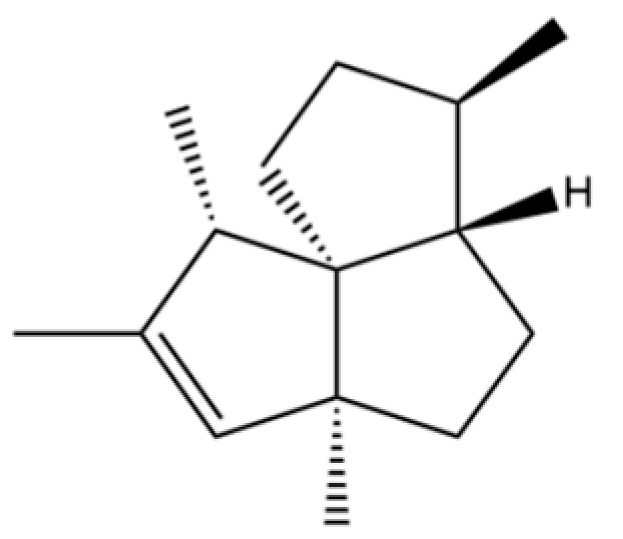 | * | * | Sesquiterpenoid | [22] |
| 171 | 1,1′-Biphenyl, 2,4,6-trimethyl- | 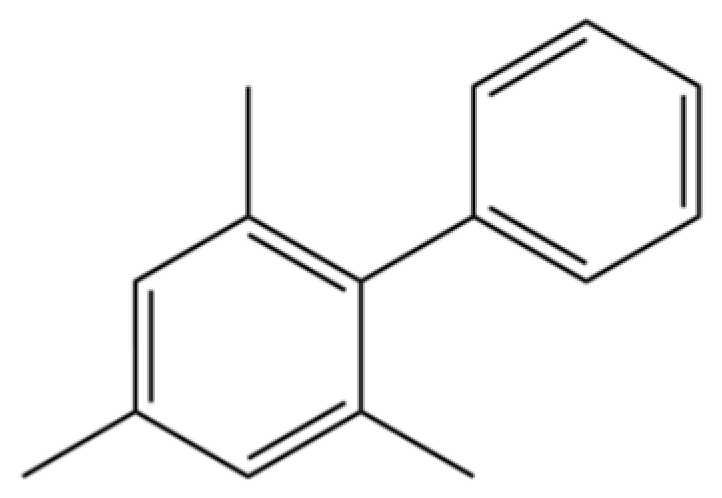 | * | * | Aromatic hydrocarbon | [36] |
| 172 | 4-Methylurazole | 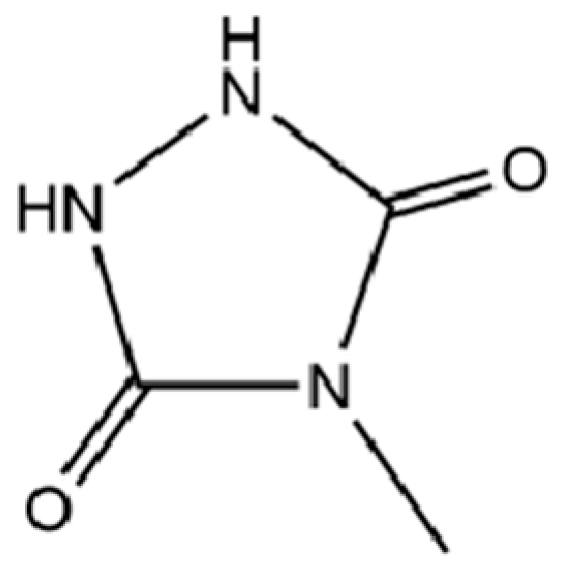 | * | * | Heterocycle | [36] |
| 173 | (1R,3aS,8aS)-1,4,4,6-Tetramethyl-1,2,3,3a,4,5,7,8-octahydrocyclopenta[c]pentalene | 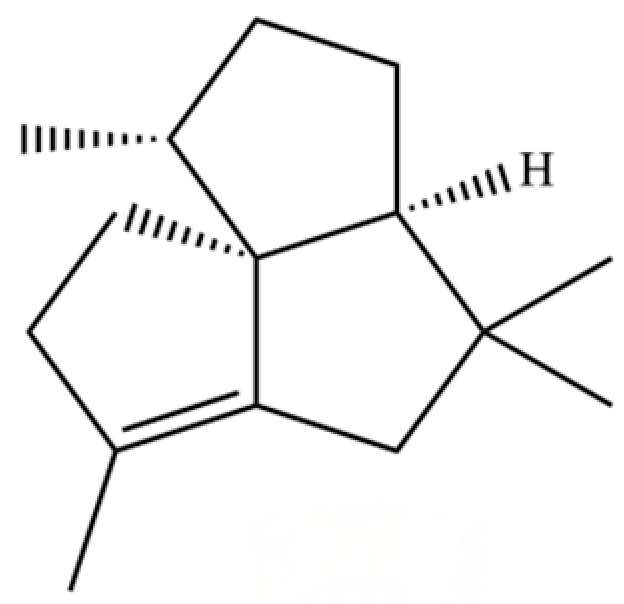 | * | * | Sesquiterpenoid | [36] |
| 174 | 1,3a,4,5a-Tetramethyl-1,2,3,3a,5a,6,7,8-octahydrocyclopenta[c]pentalene | 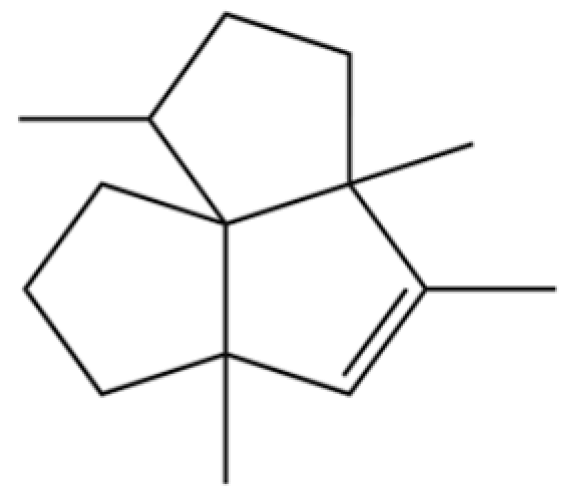 | * | * | Sesquiterpenoid | [36] |
| 175 | 9H-Fluorene, 1,9-dimethyl- | 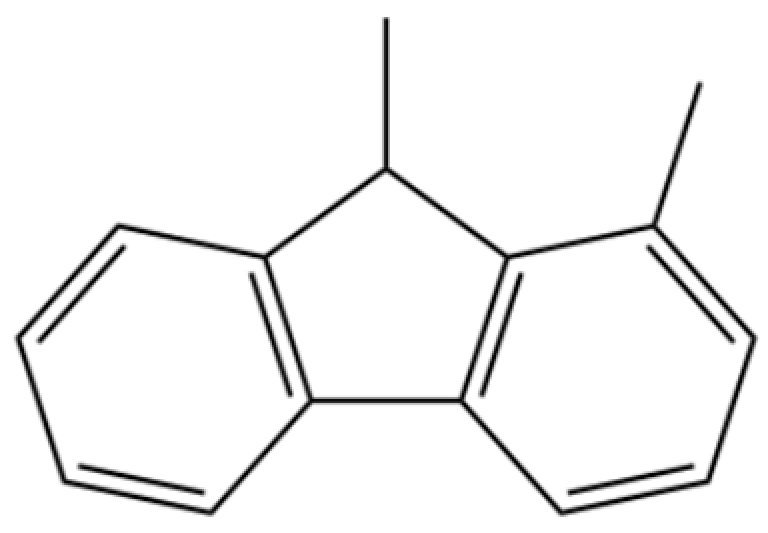 | * | * | Aromatic hydrocarbon | [36] |
| 176 | N-Benzyloxy-2-carbomethoxyaziridine | 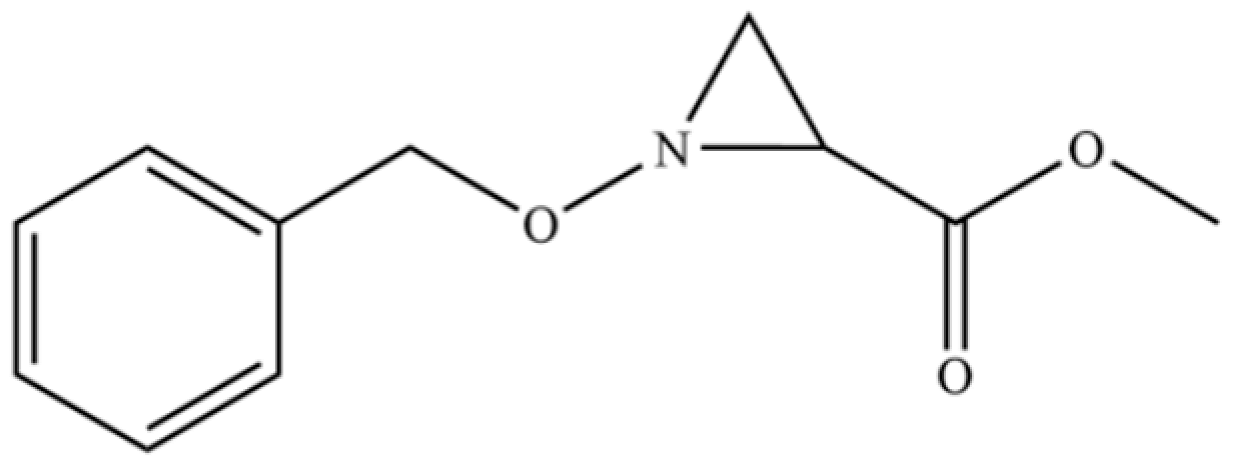 | * | * | Heterocycle | [36] |
| 177 | Naphthalene, 2,3,6-trimethyl- | 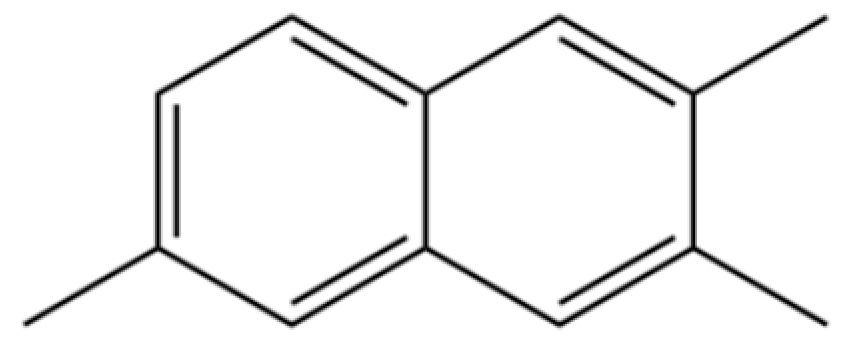 | * | * | Aromatic hydrocarbon | [36] |
| 178 | 2′-Ethoxyacetophenone | 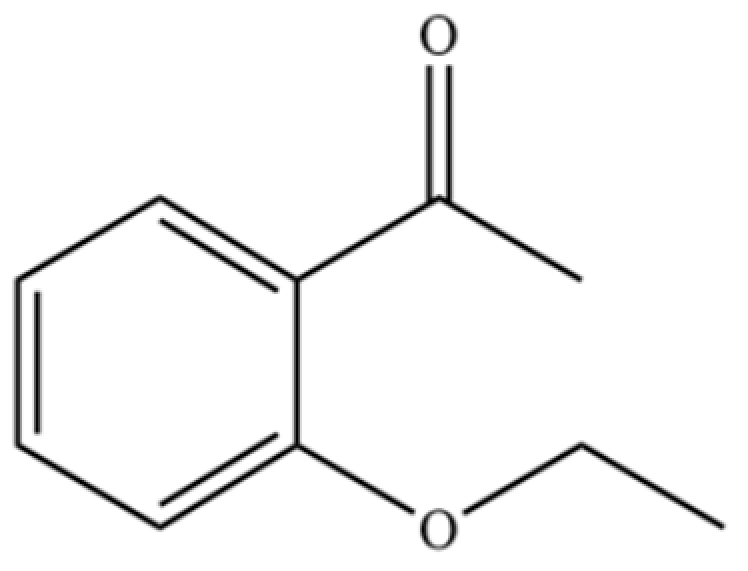 | * | * | Aromatic ketone | [36] |
| 179 | Isobutyric acid, 2-pinen-10-yl ester | 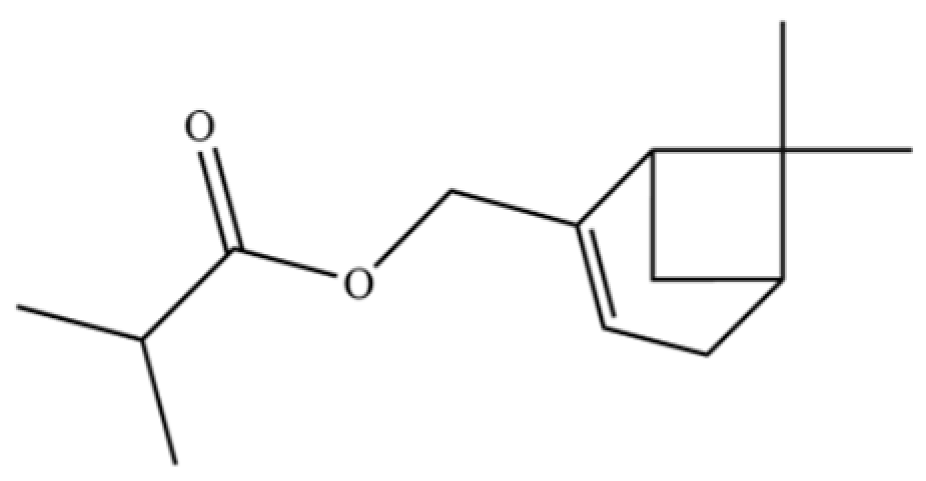 | * | * | Monoterpenoid ester | [36] |
| 180 | 4-Methylphenol, isopropyl ether | 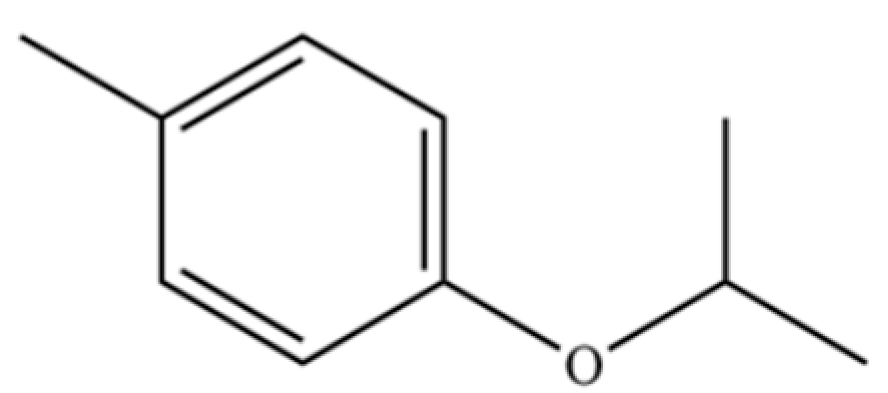 | * | * | Phenolic ether | [36] |
| 181 | Benzene, (butoxymethyl)- | 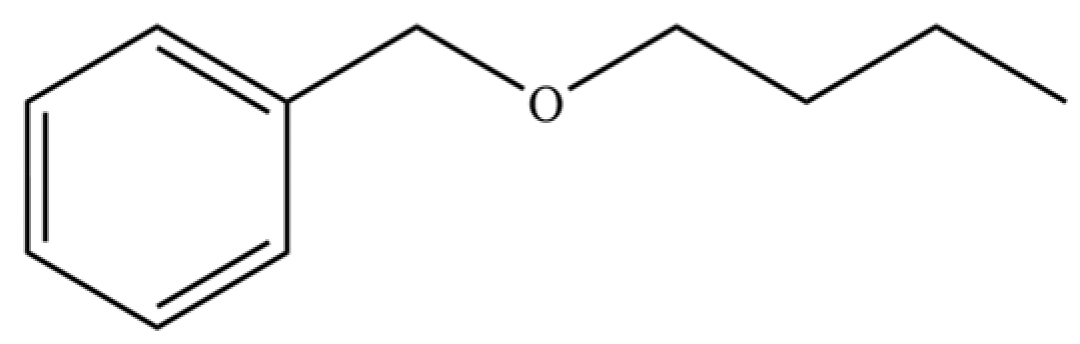 | * | * | Aromatic ether | [36] |
| 182 | 9-Dodecen-1-ol, acetate, (E)- | 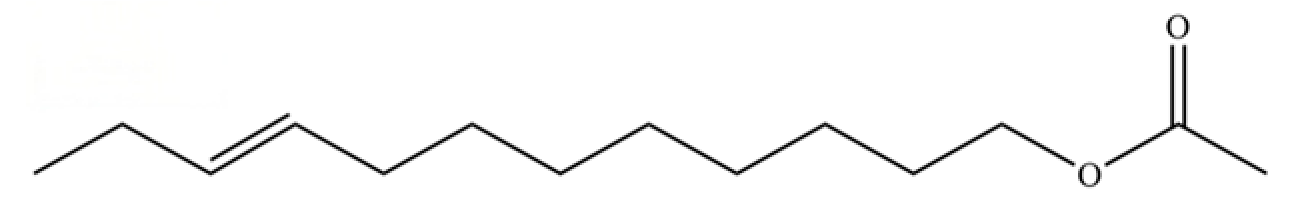 | * | * | Fatty ester | [36] |
| 183 | 5-Decen-1-ol, acetate, (E)- | 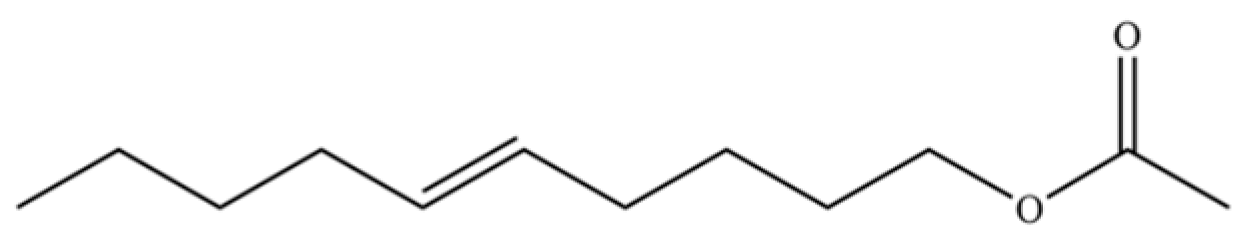 | * | * | Fatty ester | [36] |
| 184 | Hexanal | 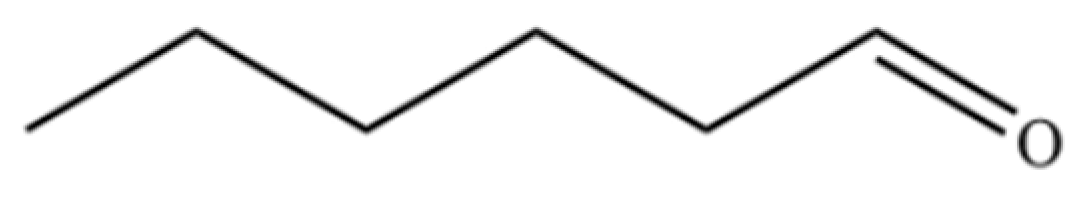 | * | * | Aldehyde | [44] |
| 185 | p-Cymene | 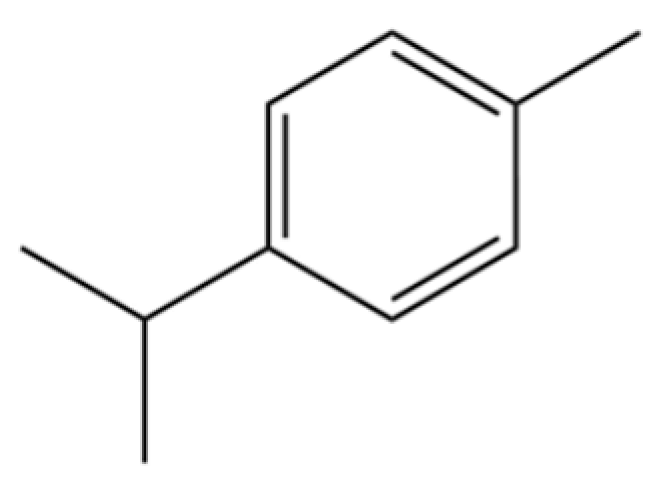 | * | * | Monoterpenoid aromatic | [21] |
| 186 | Furan, 3-(4-methyl-3-pentenyl)- | 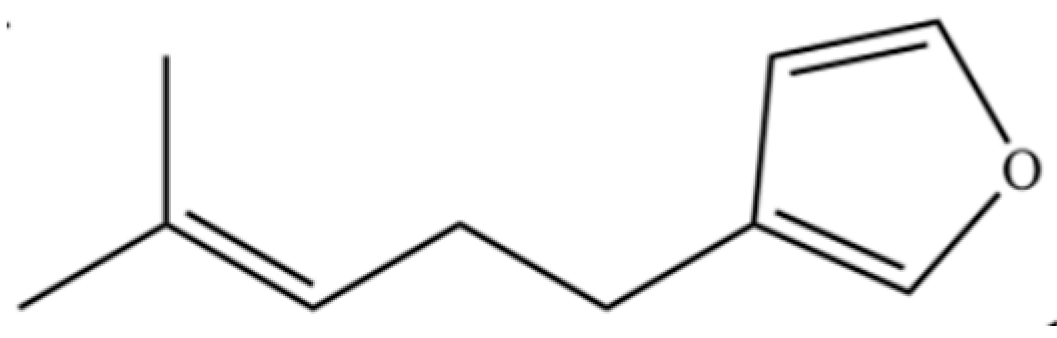 | * | * | Furan derivative | [36] |
| 187 | Geraniol | 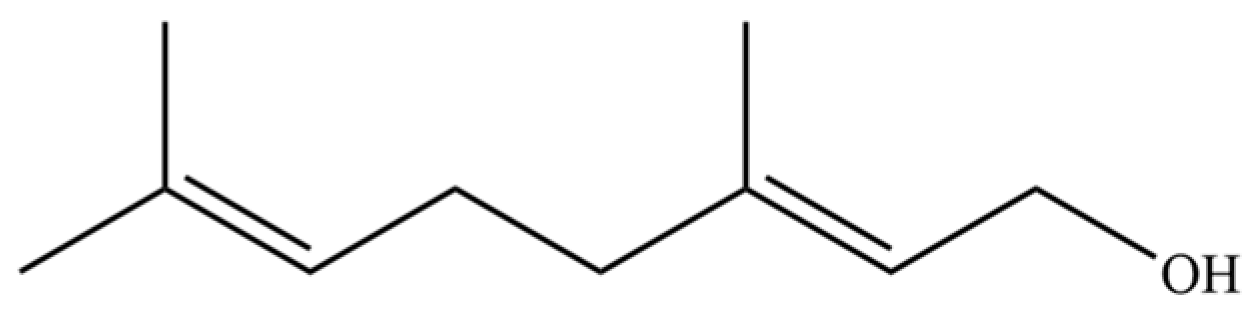 | * | * | Monoterpenoid | [45] |
| 188 | Octanoic acid, ethyl ester | 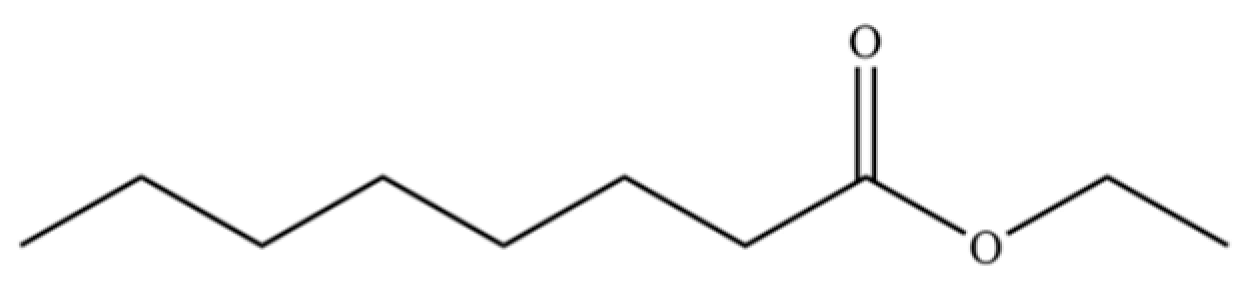 | * | * | Fatty ester | [36] |
| 189 | 2,6-Octadienal, 3,7-dimethyl-, (E)- | 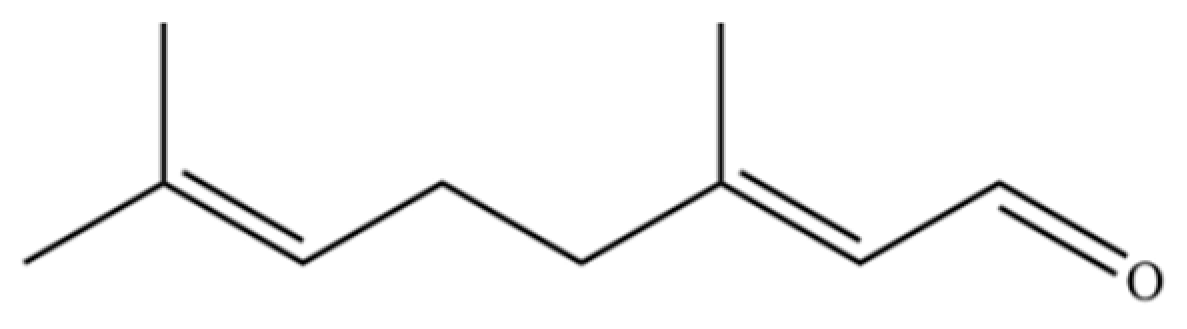 | * | * | Monoterpenoid aldehyde | [36] |
| 190 | Naphthalene, 1,2,3,4-tetrahydro-1,6-dimethyl-4-(1-methylethyl)-, (1S-cis)- | 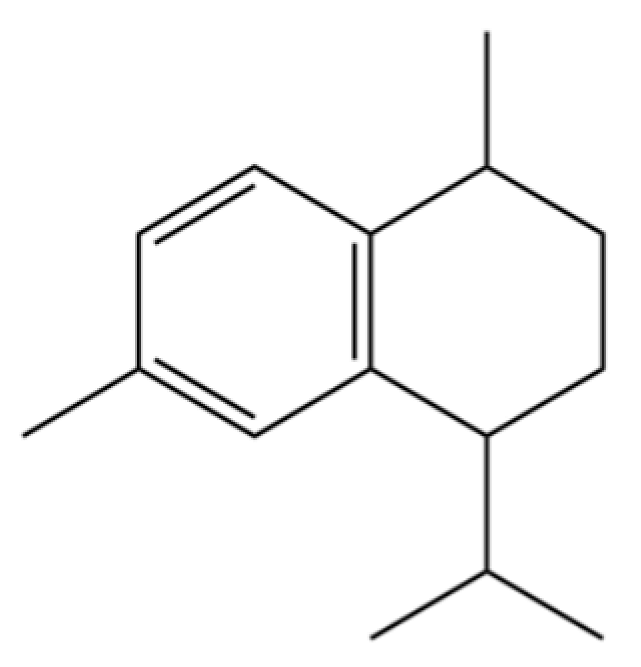 | * | * | Sesquiterpenoid | [36] |
| 191 | Guaiol | 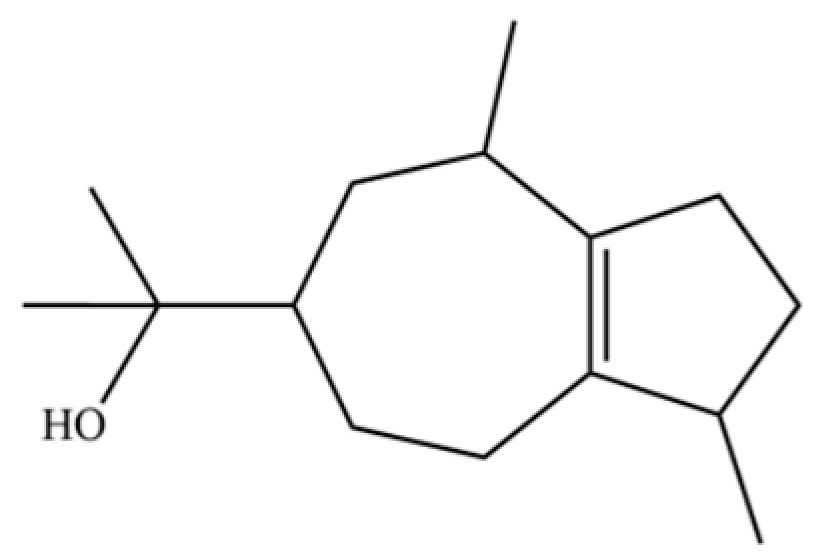 | * | * | Sesquiterpenoid | [45] |
| 192 | 5-Azulenemethanol, 1,2,3,3a,4,5,6,7-octahydro-α,α,3,8-tetramethyl-, [3S-(3.α,3a.β,5.α)]- | 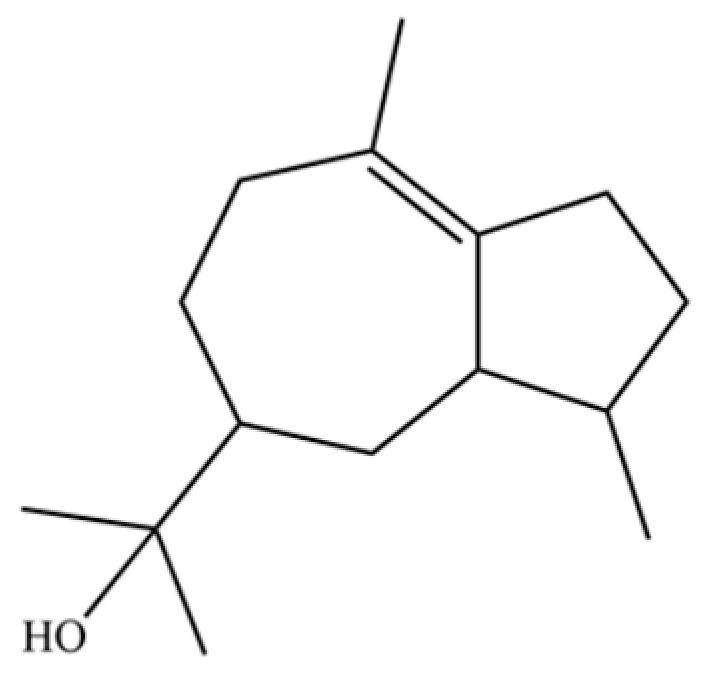 | * | * | Sesquiterpenoid | [36] |
| 193 | α-Calacorene | 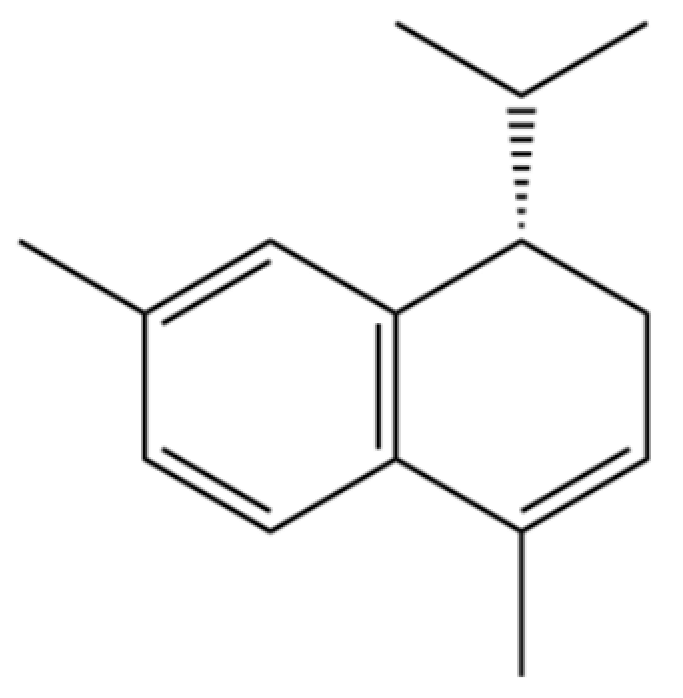 | * | * | Sesquiterpenoid | [36] |
| 194 | 1H-3a,7-Methanoazulene, octahydro-3,8,8-trimethyl-6-methylene-, [3R-(3.α,3a.β,7.β,8a.α)]- | 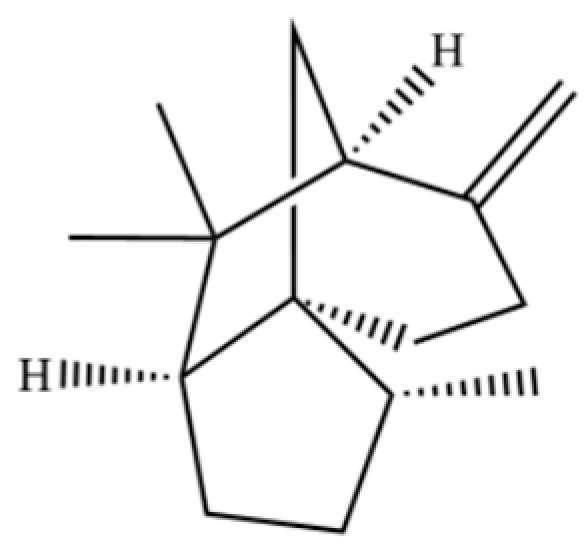 | * | * | Sesquiterpenoid | [36] |
| 195 | Bicyclosesquiphellandrene | 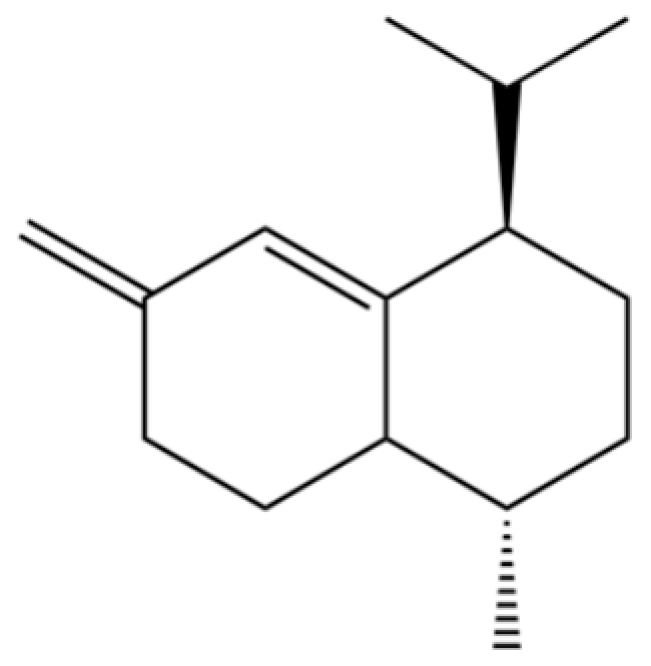 | * | * | Sesquiterpenoid | [36] |
| 196 | Podocarpa-6,13-diene, 13-isopropyl- | 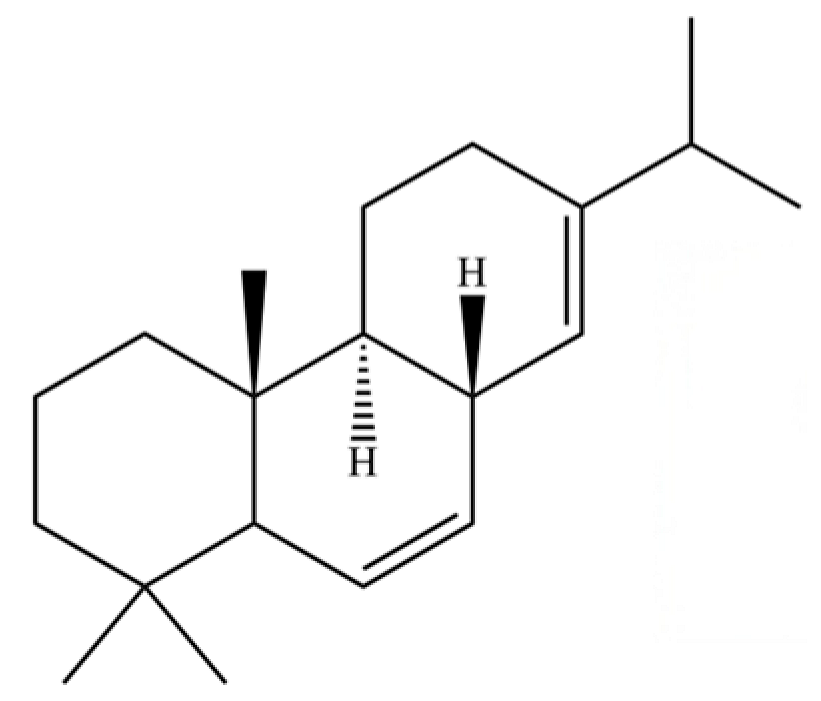 | * | * | Diterpenoid | [36] |
| 197 | Bicyclo[3.1.0]hexan-3-ol, 4-methylene-1-(1-methylethyl)-, acetate | 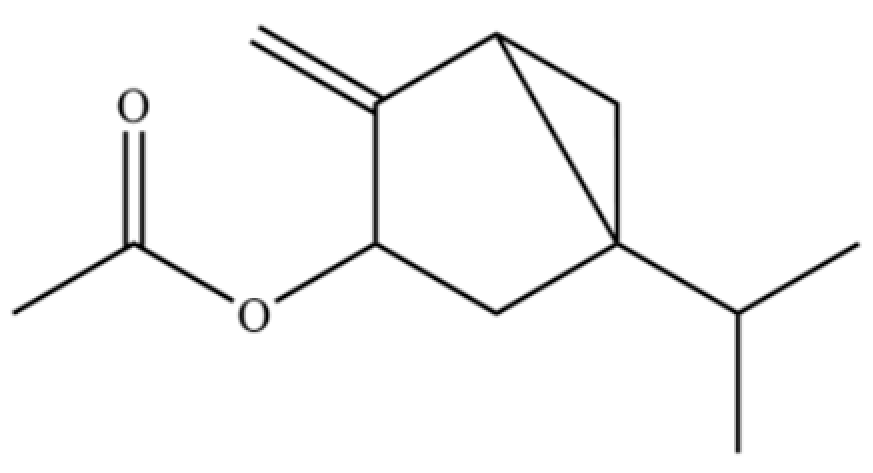 | * | * | Monoterpenoid ester | [36] |
| 198 | Ethanone, 1,1′,1″-(1,3,5-benzenetriyl)tris- |  | * | * | Aromatic ketone | [36] |
| 199 | Ethanone, 1-(5-methyl-1-phenyl-1H-pyrazol-4-yl)- |  | * | * | Heterocyclic ketone | [36] |
| 200 | Naphthalene, 1,2,4a,5,8,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-, (1.α,4a.β,8a.α)-(.+/-.)- |  | * | * | Sesquiterpenoid | [36] |
| 201 | Benzene, 1-methyl-3-(1-methylethyl)- |  | * | * | Aromatic hydrocarbon | [36] |
| 202 | 6-Isopropyl-1,4-dimethylnaphthalene |  | * | * | Aromatic hydrocarbon | [36] |
| 203 | Furan, 2-hexyl- |  | * | * | Furan derivative | [36] |
| 204 | (1S,4S,4aS)-1-Isopropyl-4,7-dimethyl-1,2,3,4,4a,5-hexahydronaphthalene |  | * | * | Sesquiterpenoid | [36] |
| 205 | Benzene, 1-methyl-4-(1,2,2-trimethylcyclopentyl)-, (R)- |  | * | * | Aromatic hydrocarbon | [36] |
| 206 | Cadina-1(10),6,8-triene |  | * | * | Sesquiterpenoid | [36] |
| 207 | Bicyclo[5.2.0]nonane, 2-methylene-4,8,8-trimethyl-4-vinyl- |  | * | * | Sesquiterpenoid | [36] |
| 208 | Isoledene |  | * | * | Sesquiterpenoid | [25] |
| 209 | 1,4,7,-Cycloundecatriene, 1,5,9,9-tetramethyl-, Z,Z,Z- |  | * | * | Sesquiterpenoid | [36] |
| 210 | α-Corocalene |  | * | * | Sesquiterpenoid | [36] |
| 211 | Benzene, 1-ethenyl-3,5-dimethyl- |  | * | * | Aromatic hydrocarbon | [36] |
| 212 | Benzene, 1-methoxy-4-methyl-2-(1-methylethyl)- |  | * | * | Aromatic ether | [36] |
| 213 | 3-Cyclohexen-1-one, 2-isopropyl-5-methyl- |  | * | * | Monoterpenoid ketone | [36] |
| 214 | 2,2′-Isopropylidenebis(5-methylfuran) | 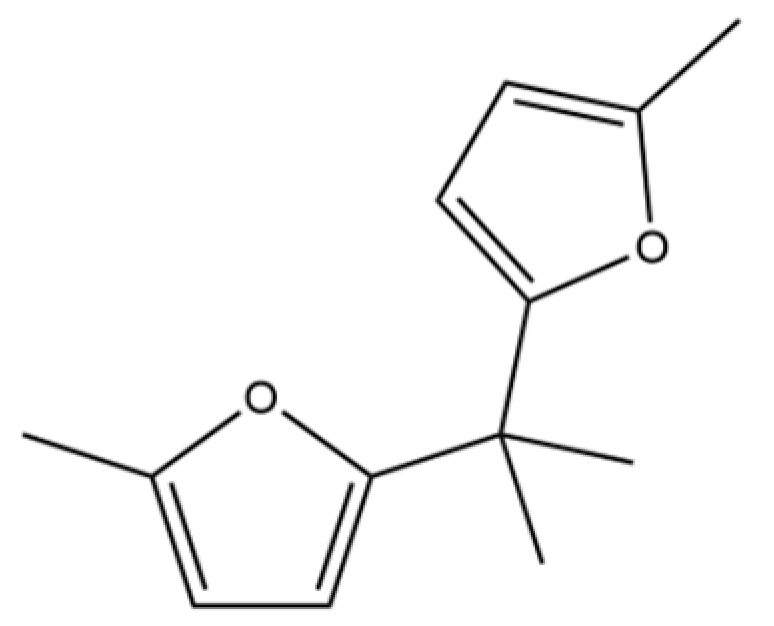 | * | * | Furan derivative | [36] |
| 215 | 3-Acetyl-2,5-dimethylbenzo(b)thiophene | 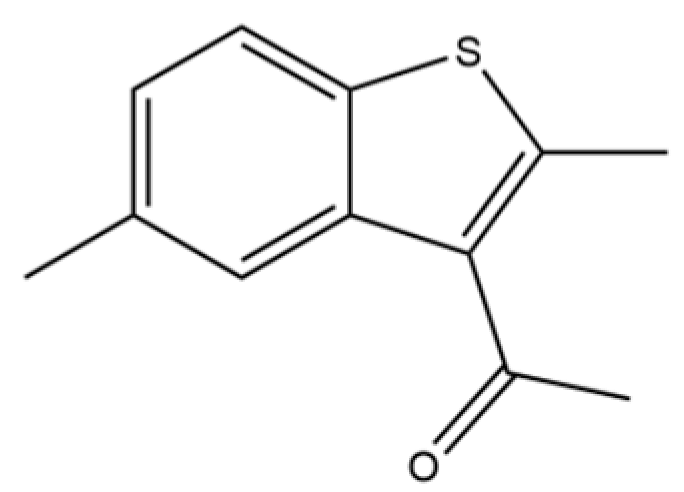 | * | * | Heterocycle | [36] |
| 216 | Benzaldehyde, 2,4-dihydroxy-3,6-dimethyl- | 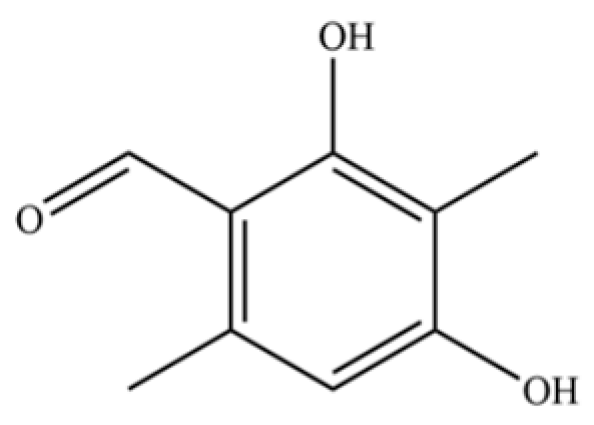 | * | * | Phenolic aldehyde | [36] |
| 217 | β-Panasinsene | 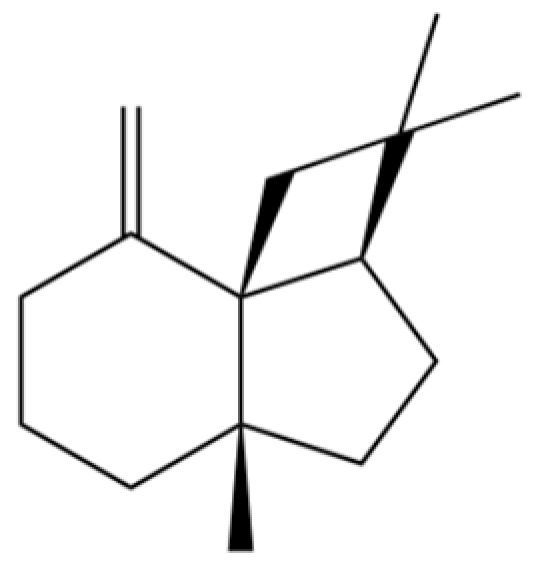 | * | * | Sesquiterpenoid | [46] |
| 218 | 2,3-Dimethyl-5-[(methylthio)propyl]pyrazine | 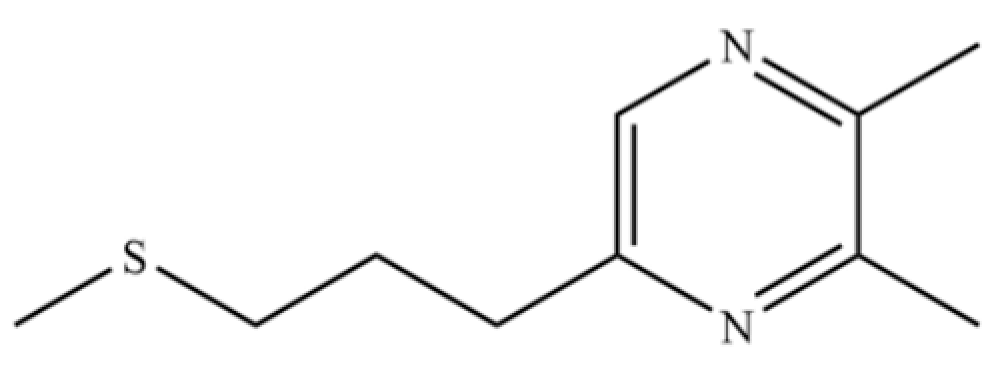 | * | * | Pyrazine derivative | [36] |
| 219 | Tricyclo[4.4.0.02,7]decane, 1-methyl-3-methylene-8-(1-methylethyl)-, stereoisomer | 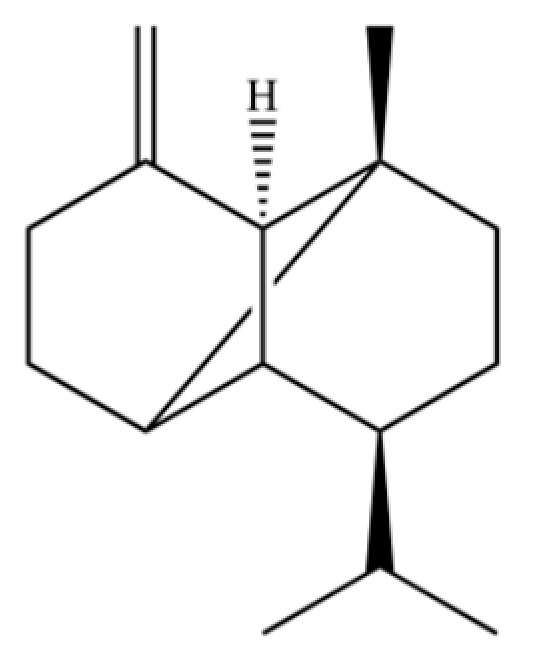 | * | * | Sesquiterpenoid | [36] |
| 220 | Coumarin, 3,4-dihydro-4,4,6,8-tetramethyl- | 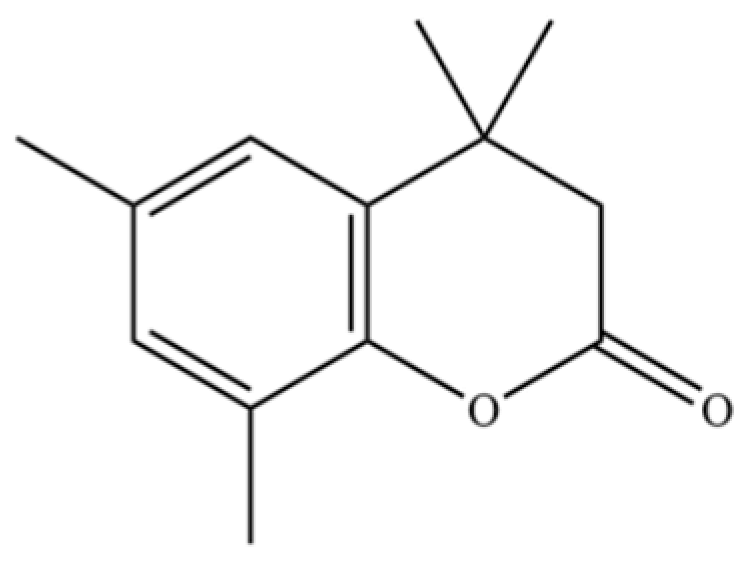 | * | * | Coumarin | [36] |
| 221 | 1,4-Methanocycloocta[d]pyridazine, 1,4,4a,5,6,9,10,10a-octahydro-11,11-dimethyl-, (1.α,4.α,4a.α,10a.α)- | 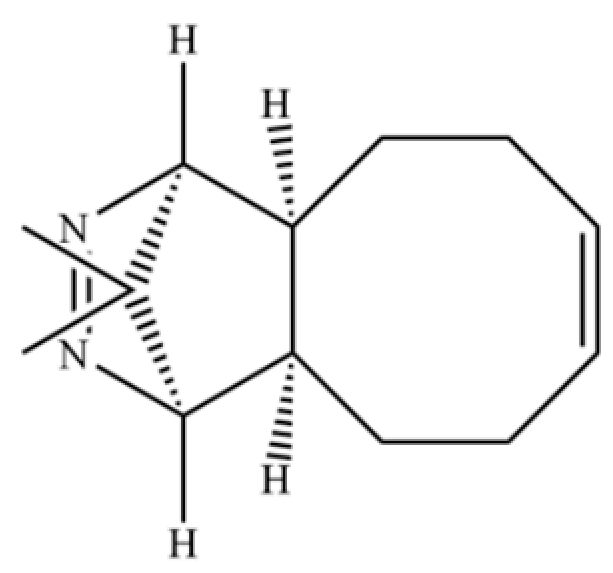 | * | * | Heterocycle | [36] |
| 222 | Dibenzofuran, 2-methoxy- | 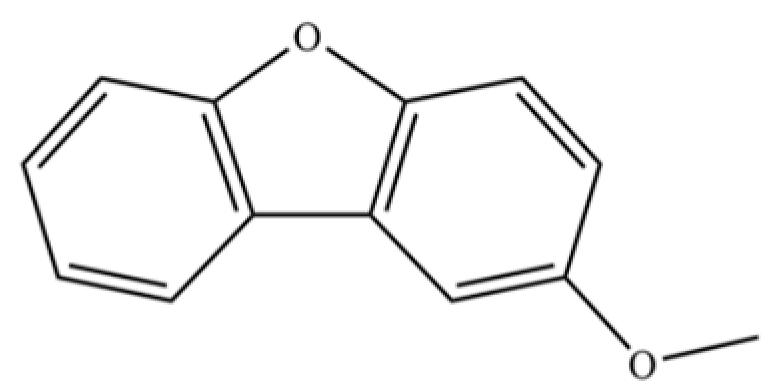 | * | * | Furan derivative | [36] |
| 223 | 1H-Imidazole, 1-acetyl- | 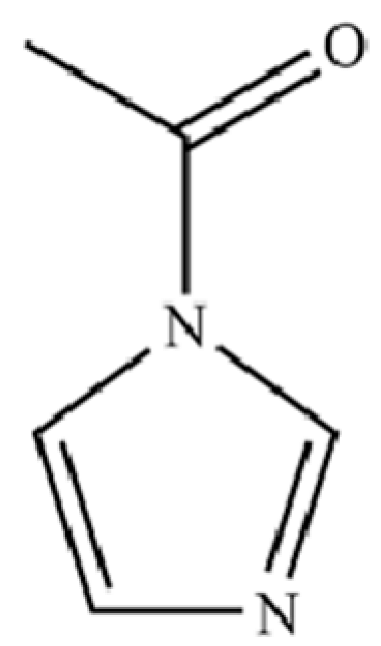 | * | * | Heterocycle | [36] |
| 224 | 4-Formyl-3,5-dimethyl-1H-pyrrole-2-carbonitrile | 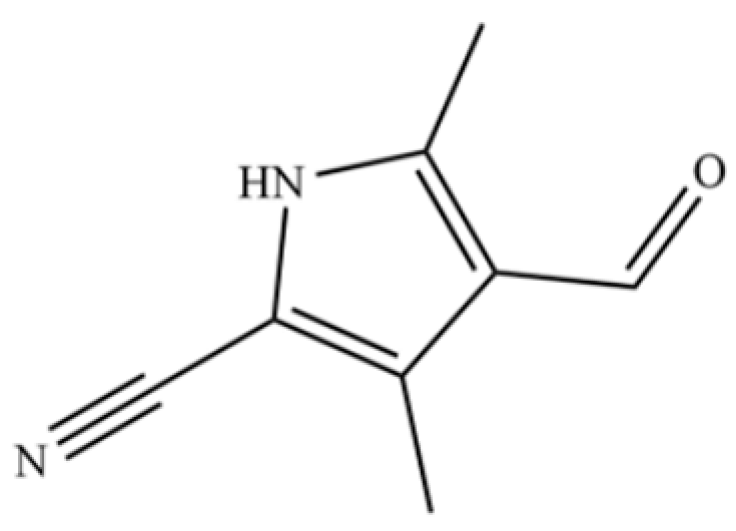 | * | * | Heterocycle | [36] |
| 225 | (4R,4aS,6S)-4,4a-Dimethyl-6-(prop-1-en-2-yl)-1,2,3,4,4a,5,6,7-octahydronaphthalene | 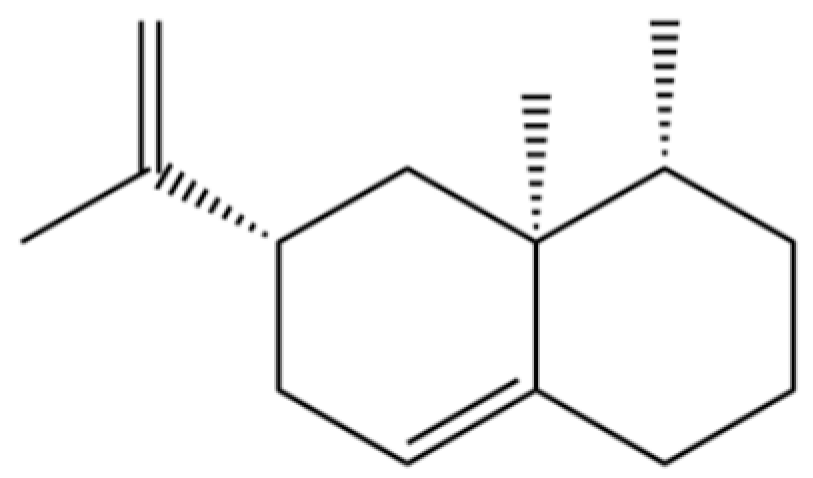 | * | * | Sesquiterpenoid | [36] |
| 226 | 3-Isobutyl-4,5-dimethyl-3H-isobenzofuran-1-one | 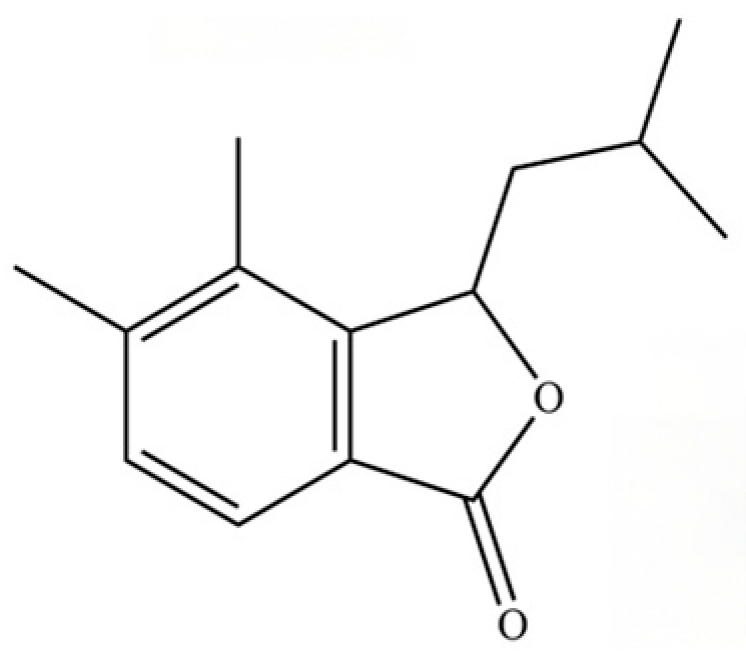 | * | * | Phthalide derivative | [36] |
| 227 | 4-Acetoxy-3-methoxystyrene | 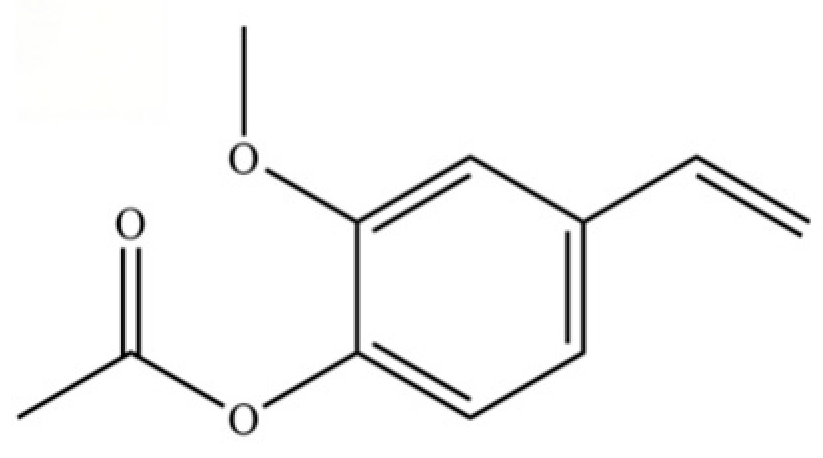 | * | * | Phenylpropanoid | [36] |
| 228 | 2-Acetyl-3,5-dimethylbenzo(b)thiophene | 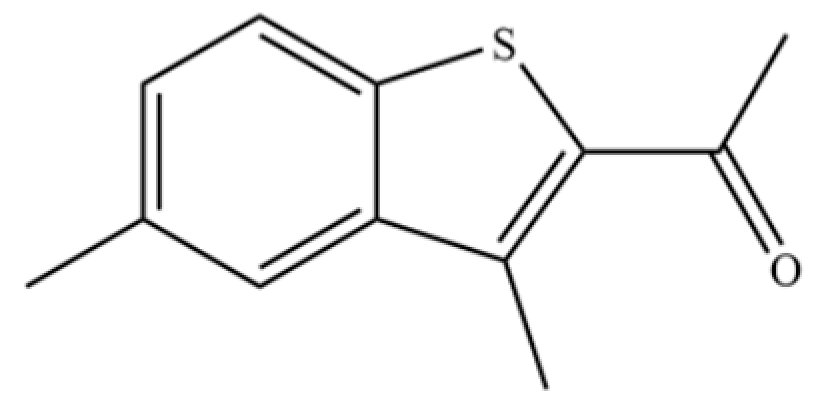 | * | * | Heterocycle | [36] |
| 229 | 1,3,5-Cycloheptatriene, 2,3,4,5,7,7-hexamethyl- | 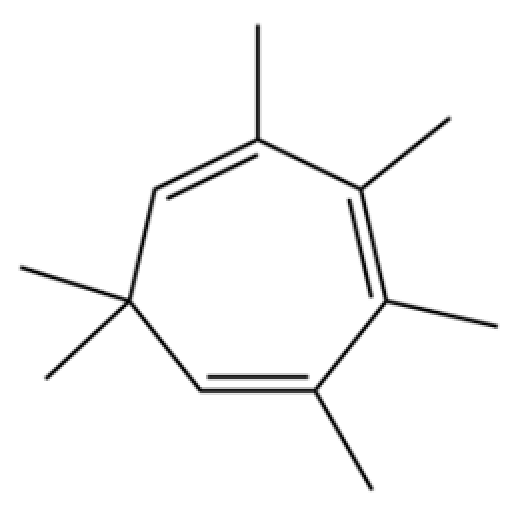 | * | * | Troponoid | [36] |
| 230 | 1H-1,2,3-Triazole | 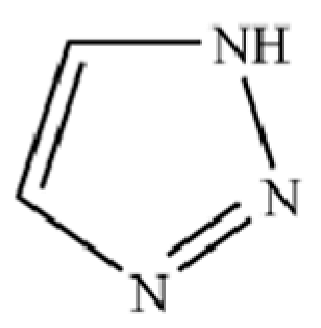 | * | * | Heterocycle | [36] |
| 231 | Carvenone | 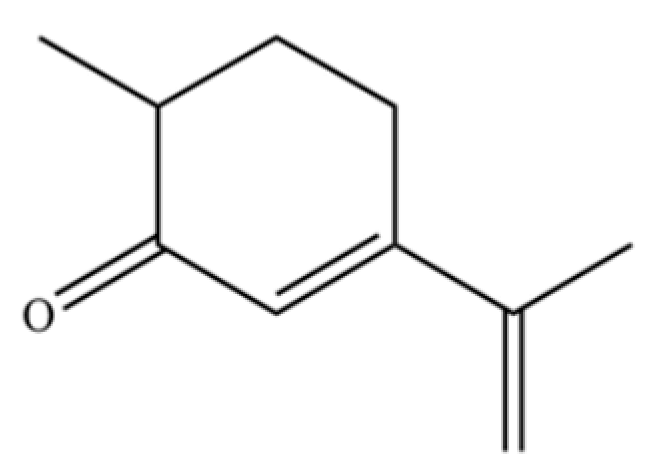 | * | * | Monoterpenoid ketone | [47] |
| 232 | 5-Azulenemethanol, 1,2,3,4,5,6,7,8-octahydro-α,α,3,8-tetramethyl-, acetate, [3S-(3.α,5.α,8.α)]- | 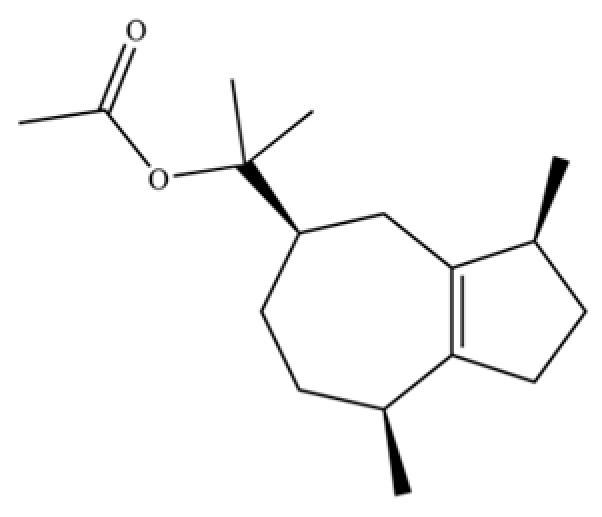 | * | * | Sesquiterpenoid ester | [36] |
| 233 | vanillic acid | 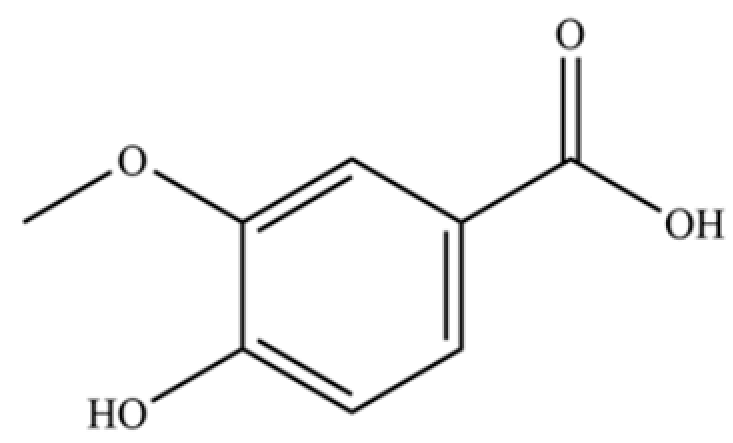 | * | * | Phenolic acid | [48] |
| 234 | L-phenylalanine | 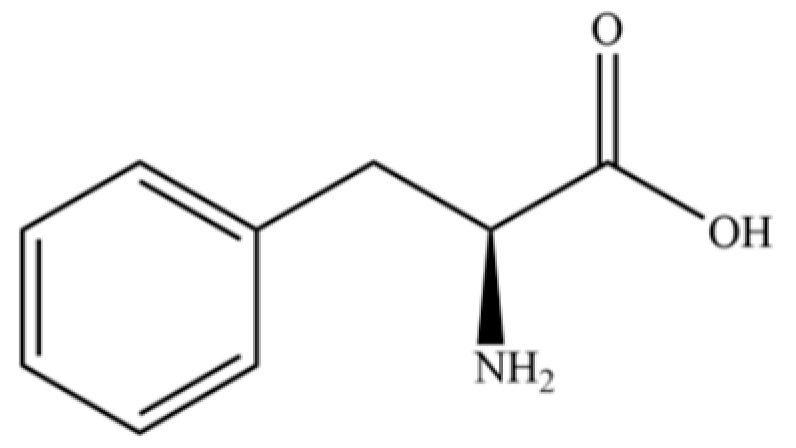 | * | * | Amino acid | [49] |
| 235 | quinic acid | 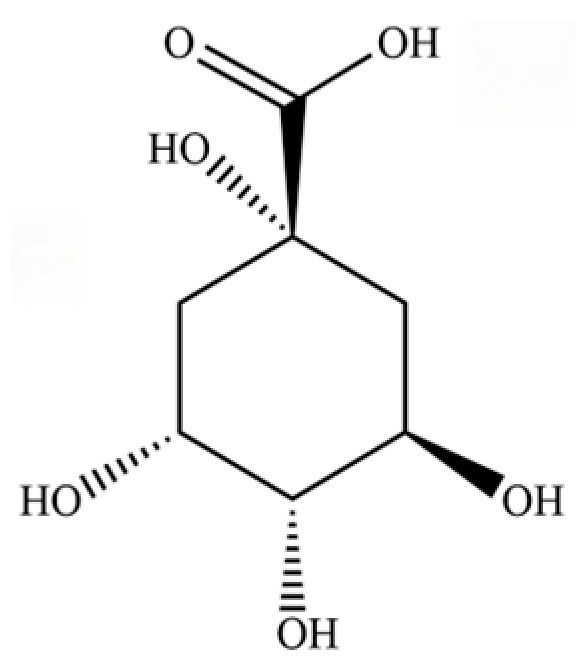 | * | * | Cyclitol carboxylic acid | [3] |
| 236 | D-tryptophan | 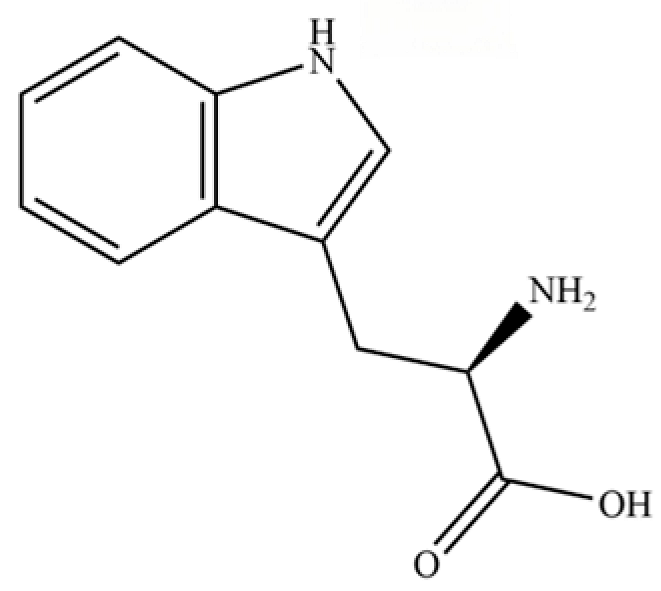 | * | * | Amino acid | [50] |
| 237 | (1R, 4S, 6R)-1, 3, 3-trimethyl-2- oxabicyclo[2.2.2]oct-6-yl-6-O-β-D-glucopyranosyl-β-D-glucopyranoside | 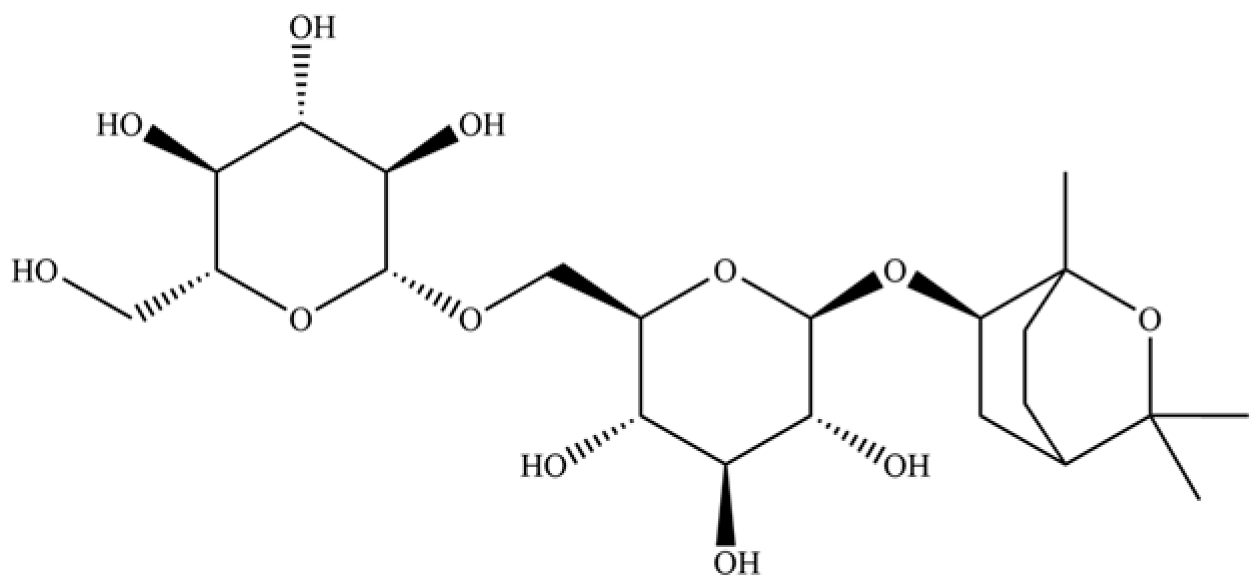 | * | * | Monoterpenoid glycoside | [51] |
| 238 | 2-(3-isopropyl-4-methyl-pent-3- en-1-ynyl)-2-methyl-cyclobutanone | 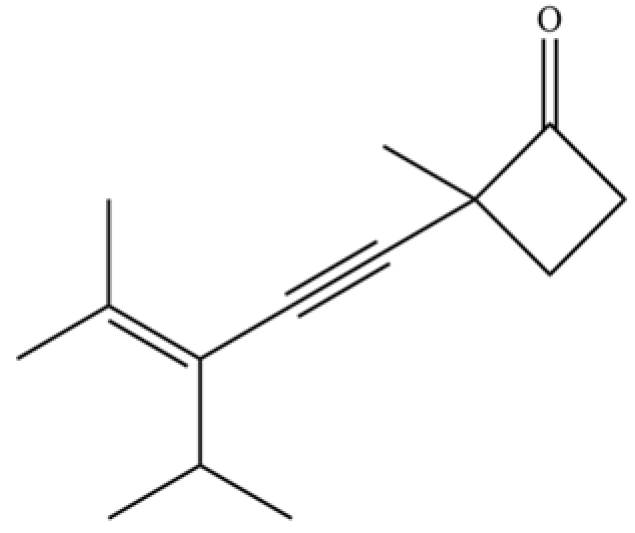 | * | * | Polyacetylene | [51] |
| 239 | citric acid | 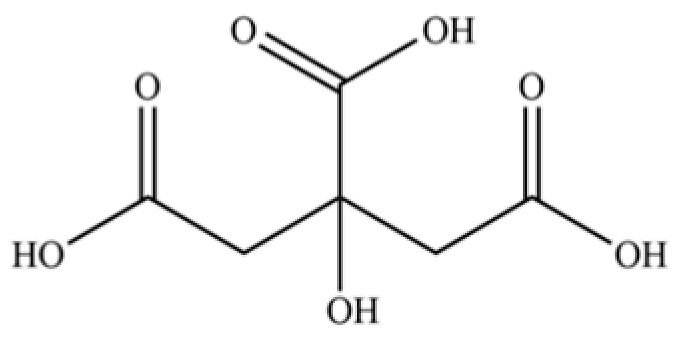 | * | * | Organic acid | [52] |
| 240 | neochlorogenic acid | 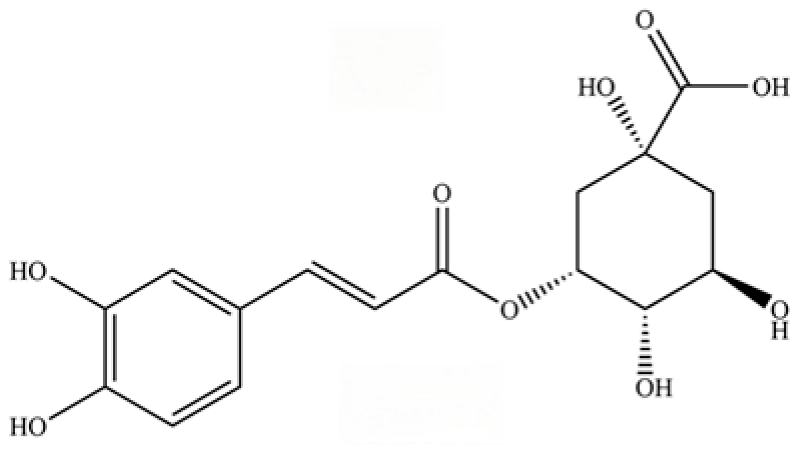 | * | * | Phenolic acid (chlorogenic acid derivative) | [53] |
| 241 | scopoletin | 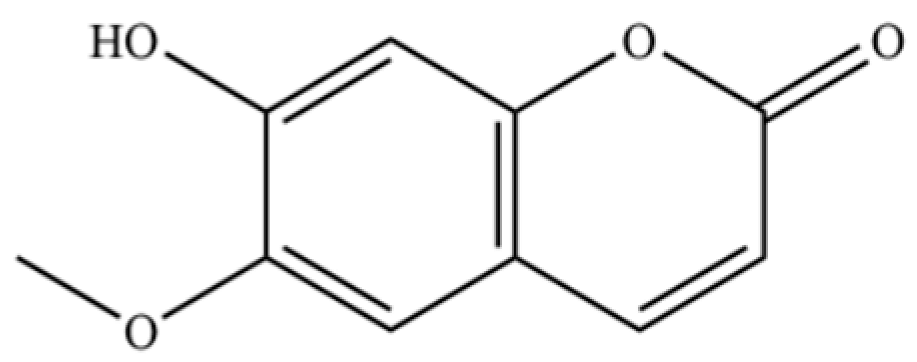 | * | * | Coumarin | [54] |
| 242 | chlorogenic acid | 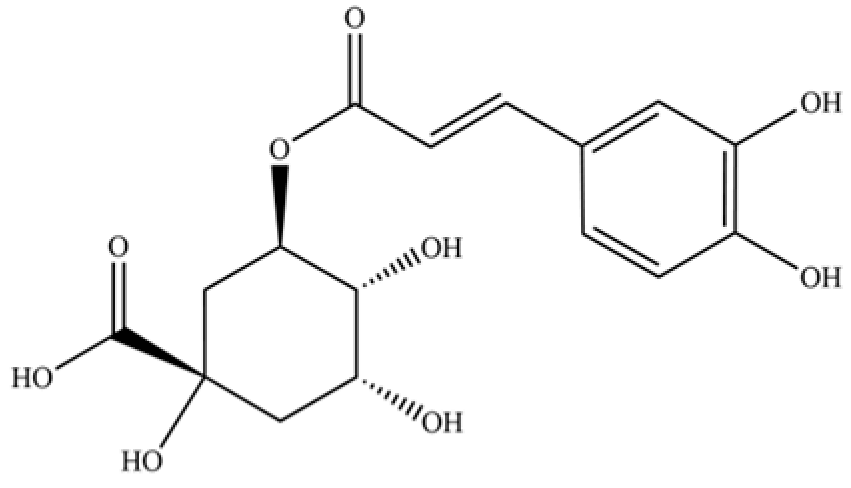 | * | * | Phenolic acid | [55] |
| 243 | atractyloside A | 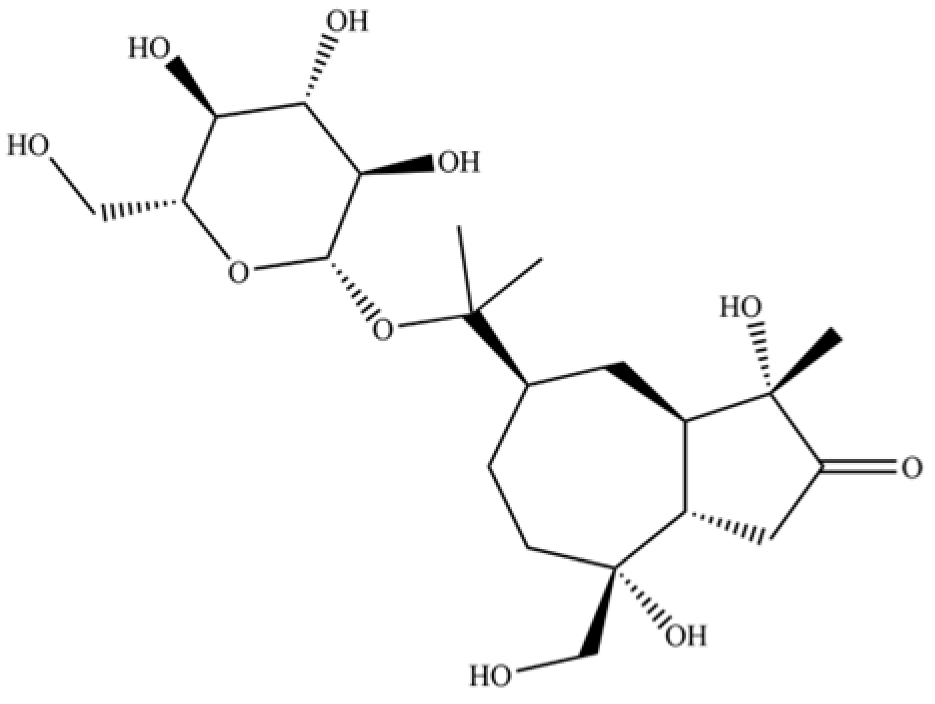 | * | * | Sesquiterpenoid glycoside | [56] |
| 244 | hymecromone | 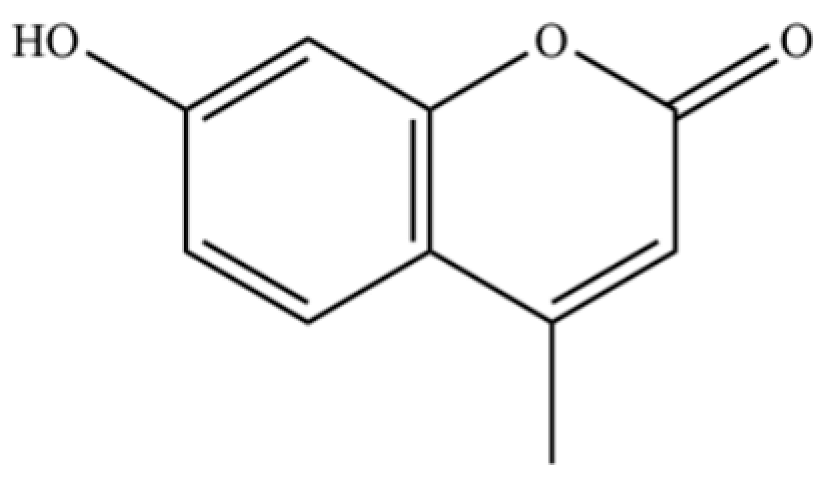 | * | * | Coumarin | [57] |
| 245 | cryptochlorogenin acid | 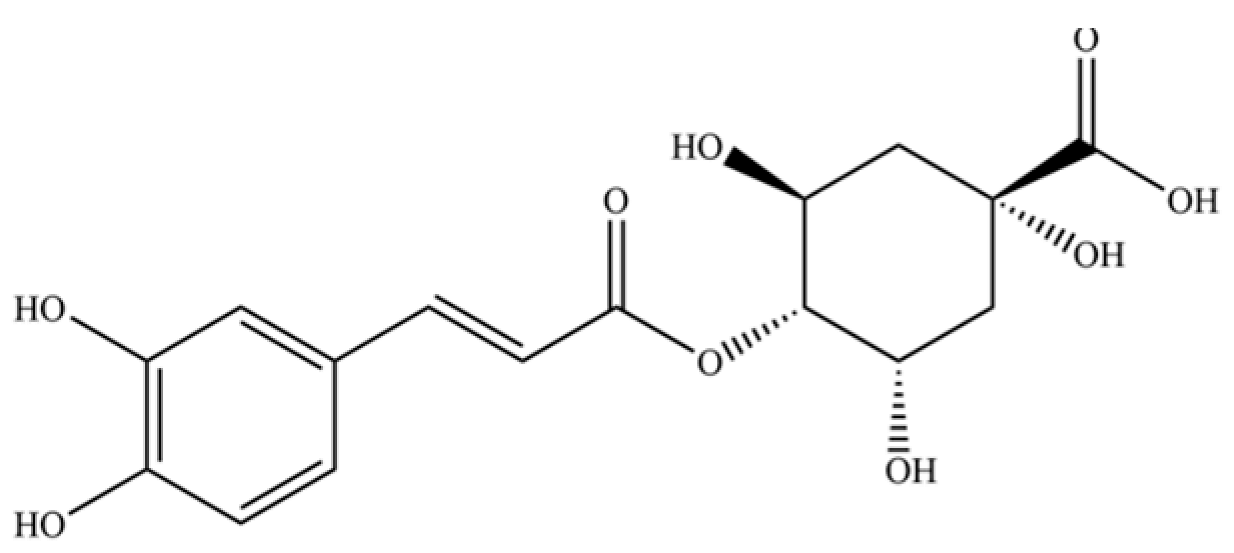 | * | * | Phenolic acid | [58] |
| 246 | coumaroylquinic acid | 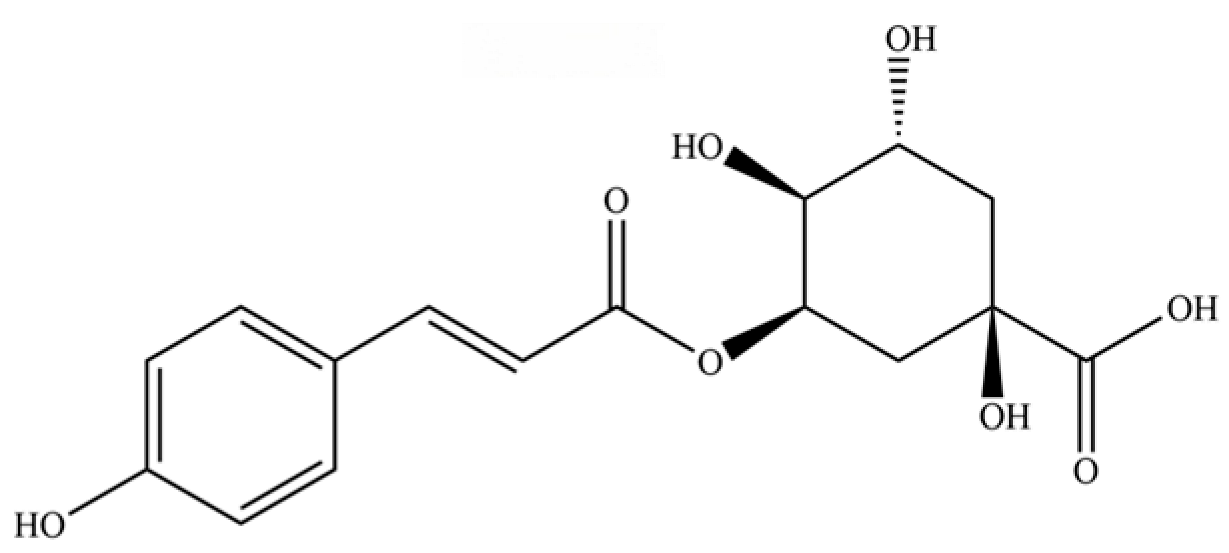 | * | * | Phenolic acid | [58] |
| 247 | 5-O-feruloylquinic acid | 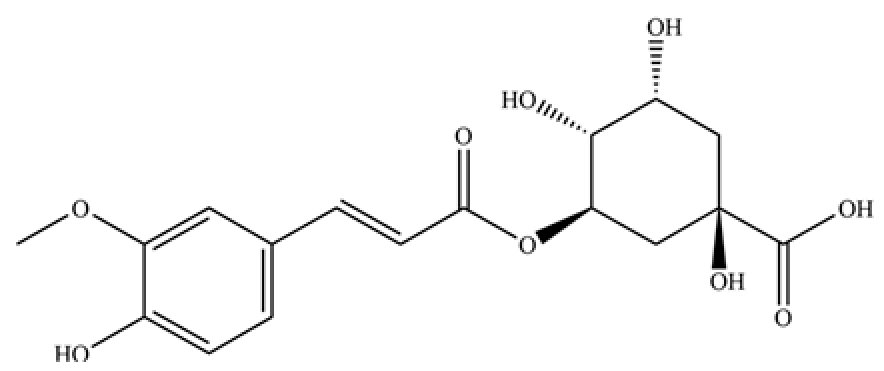 | * | * | Phenolic acid | [58] |
| 248 | dihydrosyrindine | 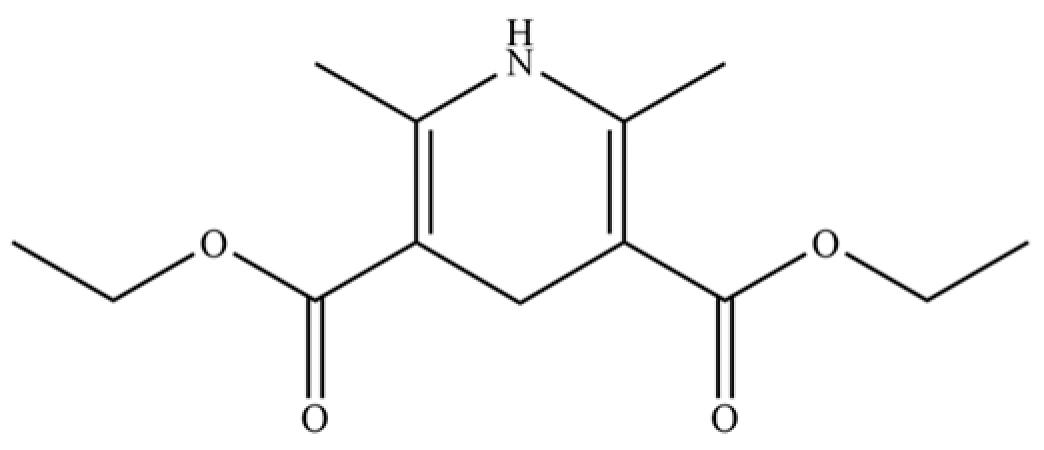 | * | * | Glycosides | [58] |
| 249 | naphthol(1,2)furan-2-one | 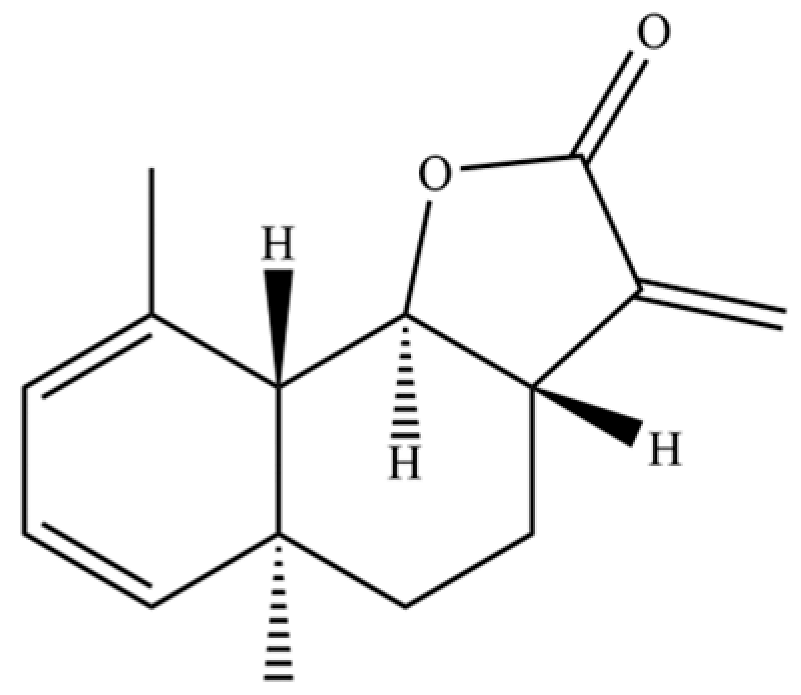 | * | * | Naphthofuran derivative | [51] |
| 250 | rutin | 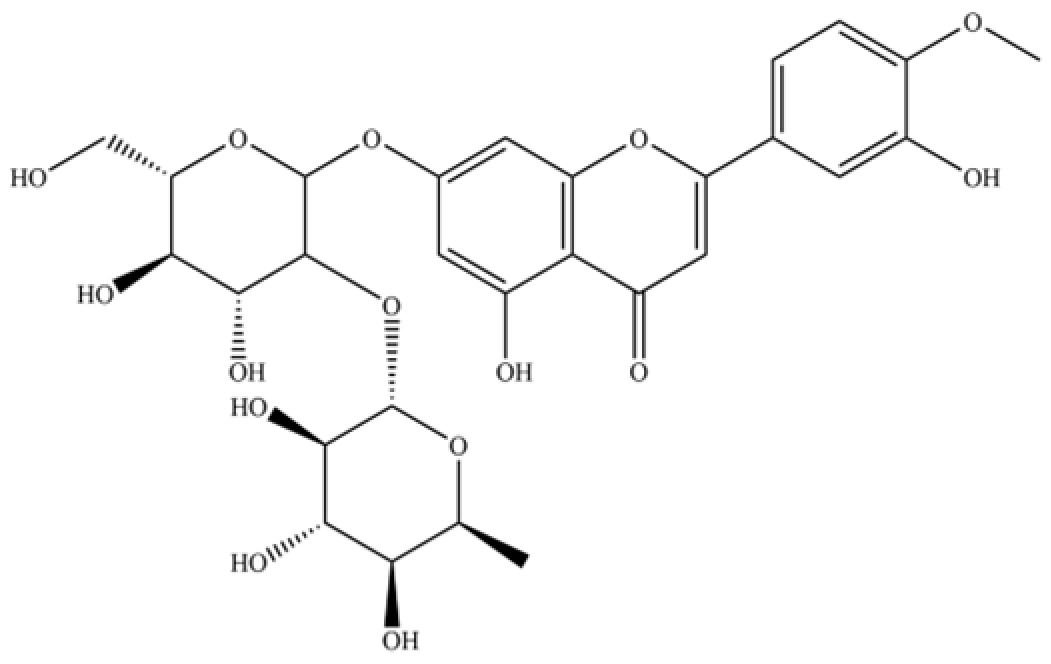 | * | * | Flavonoid glycoside | [59] |
| 251 | atractylenolide III | 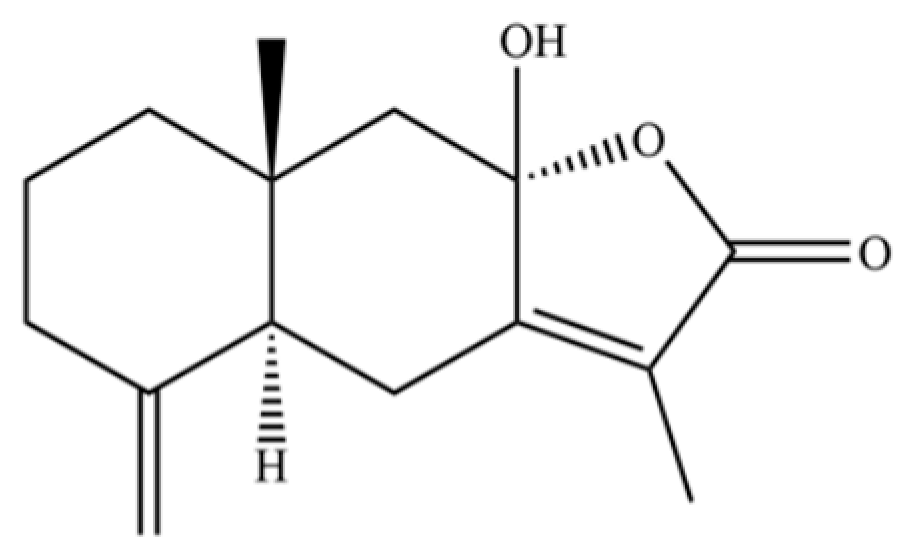 | * | * | Sesquiterpenoid lactone | [60] |
| 252 | vitexin | 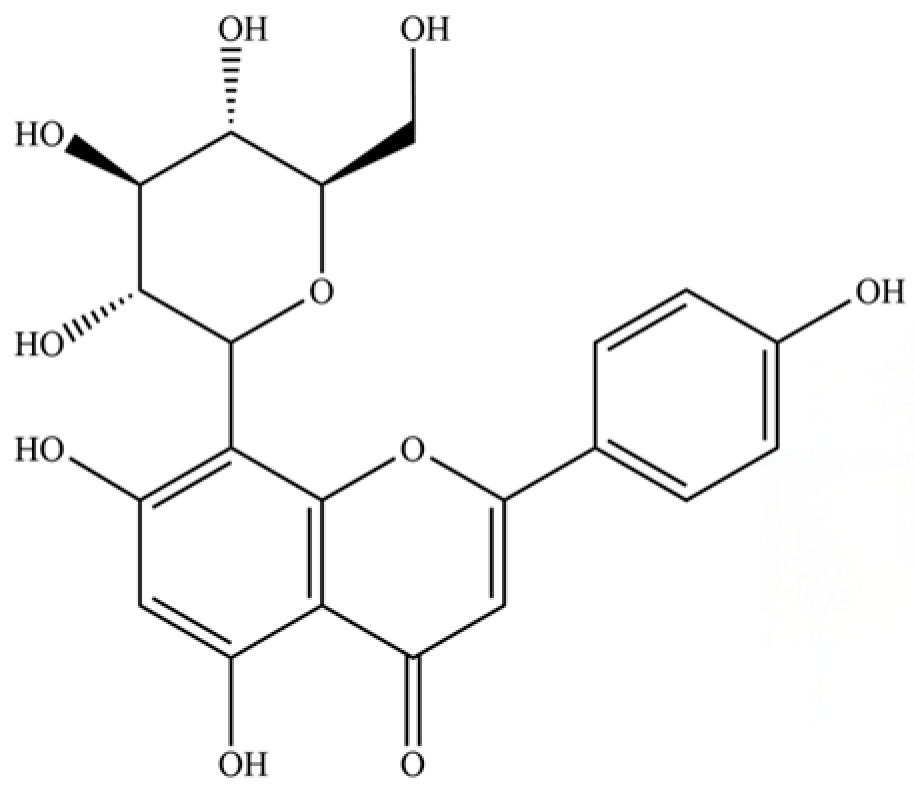 | * | * | Flavonoid glycoside | [61] |
| 253 | naringenin chalcone | 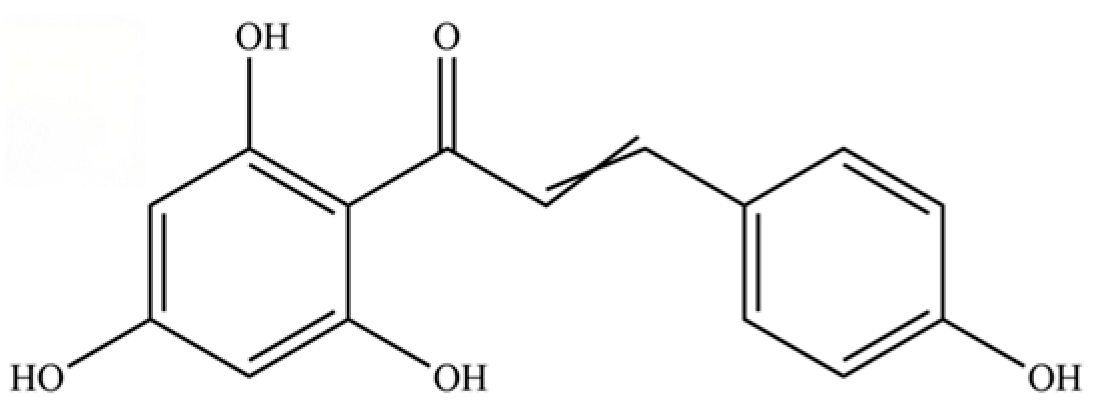 | * | * | Flavonoid (chalcone) | [62] |
| 254 | icariside D1 | 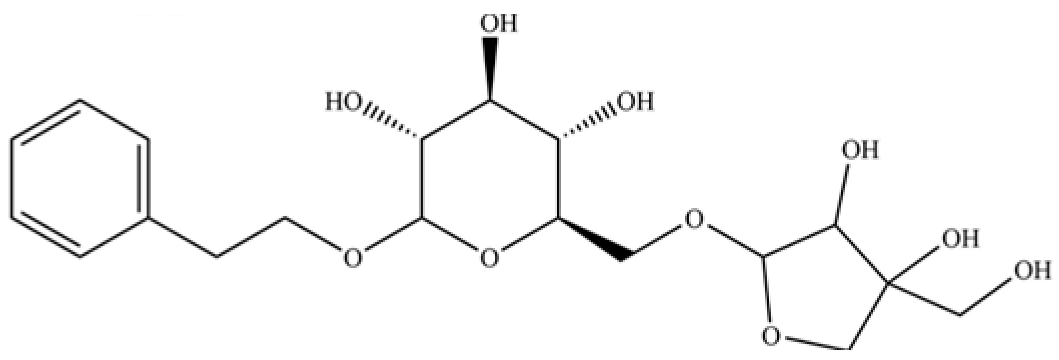 | * | * | Phenolic glycoside | [58] |
| 255 | atractyloside I | 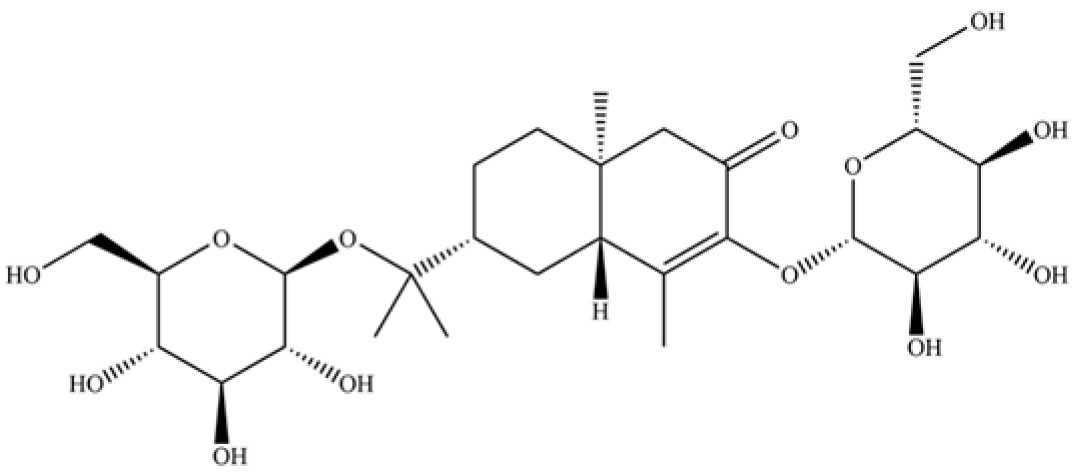 | * | * | Sesquiterpenoid glycoside | [63] |
| 256 | dehydrocostus lactone | 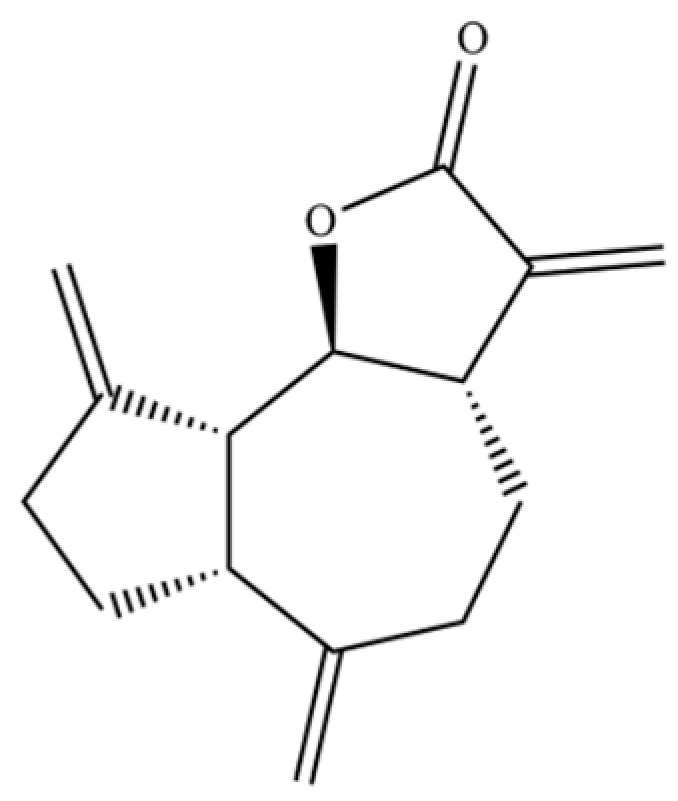 | * | * | Sesquiterpenoid lactone | [51] |
| 257 | wogonoside | 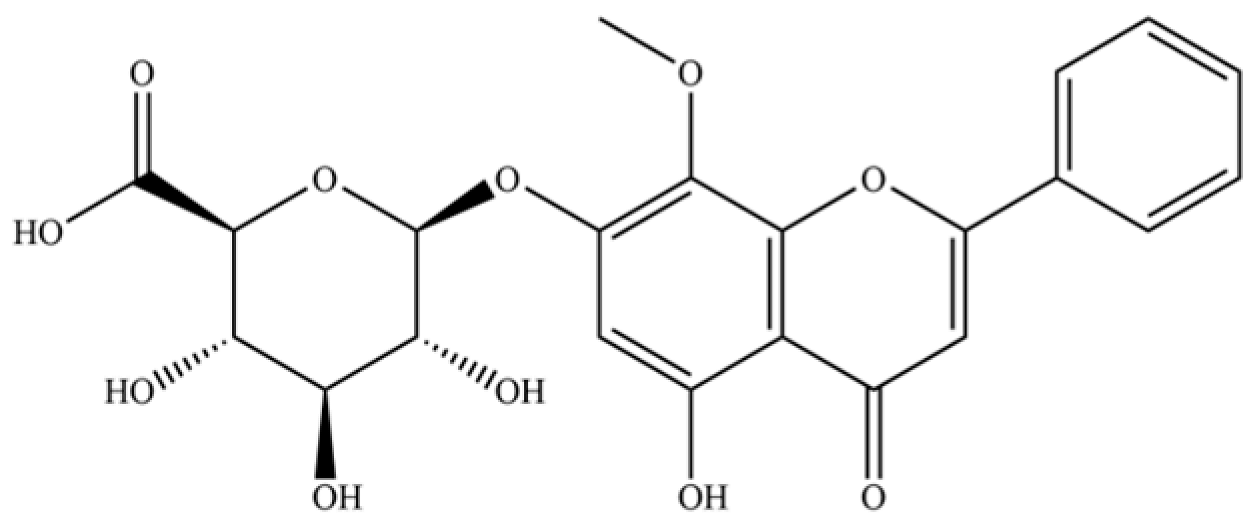 | * | * | Flavonoid glycoside | [64] |
| 258 | atractylenolactam | 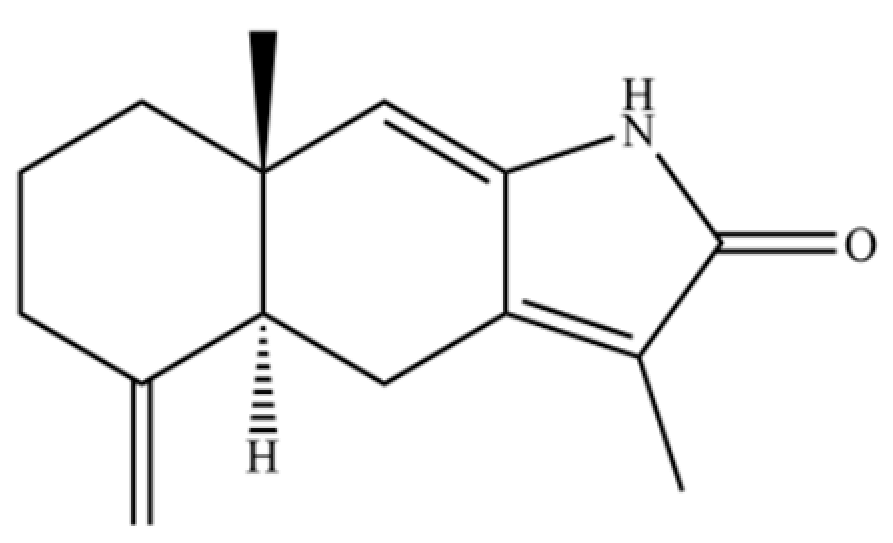 | * | * | Sesquiterpenoid lactam | [65] |
| 259 | atractylodinol | 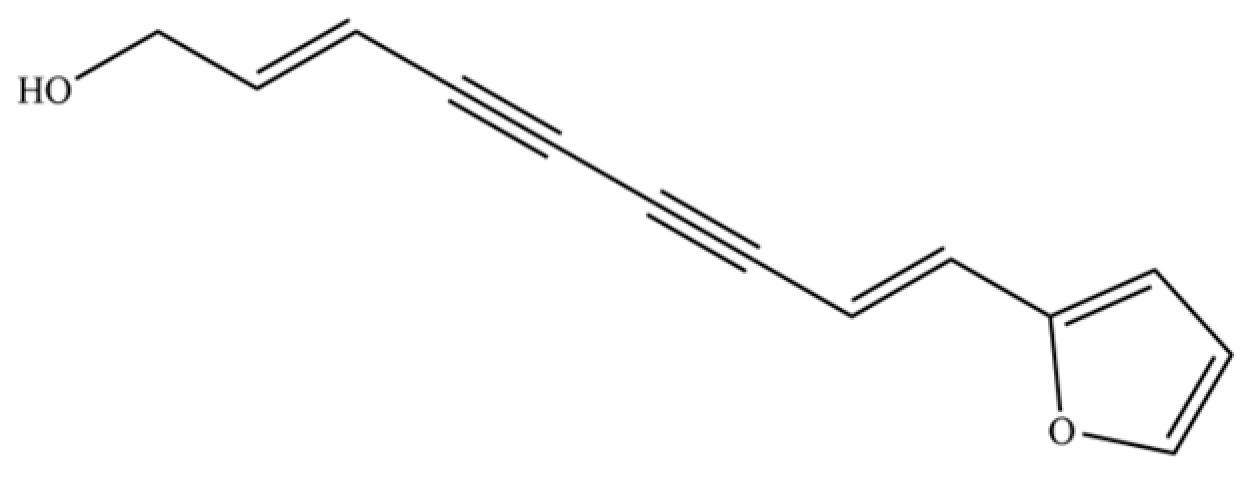 | ** | * | Polyacetylene | [66] |
| 260 | Atrachinenins E | 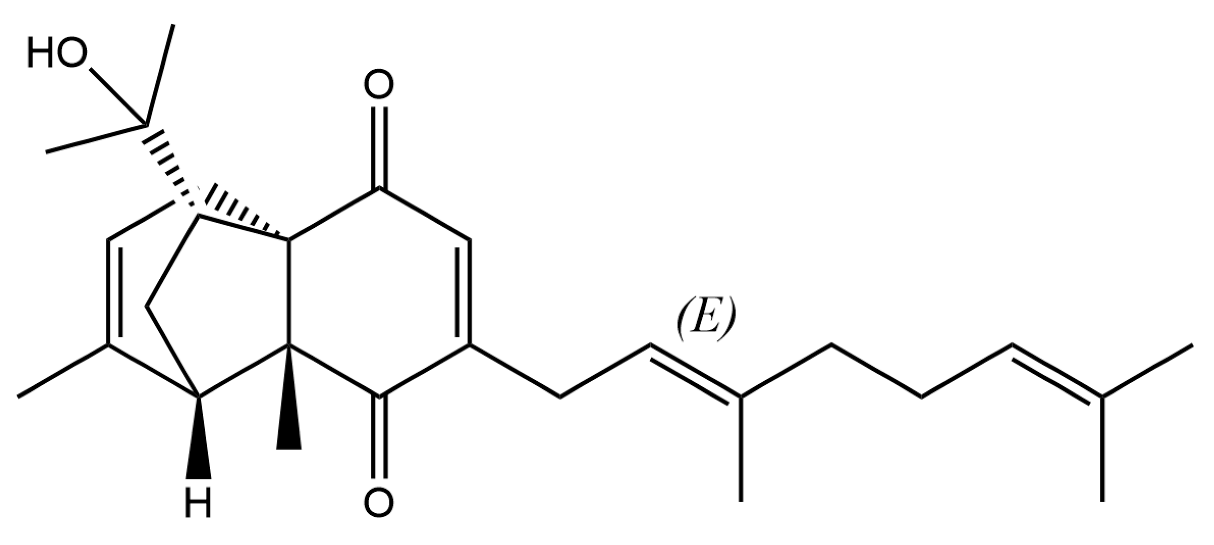 | / | ** | Sesquiterpenoid | [23] |
| 261 | (4β,5β,6β,7β)-Aristol-9-en-8-one | 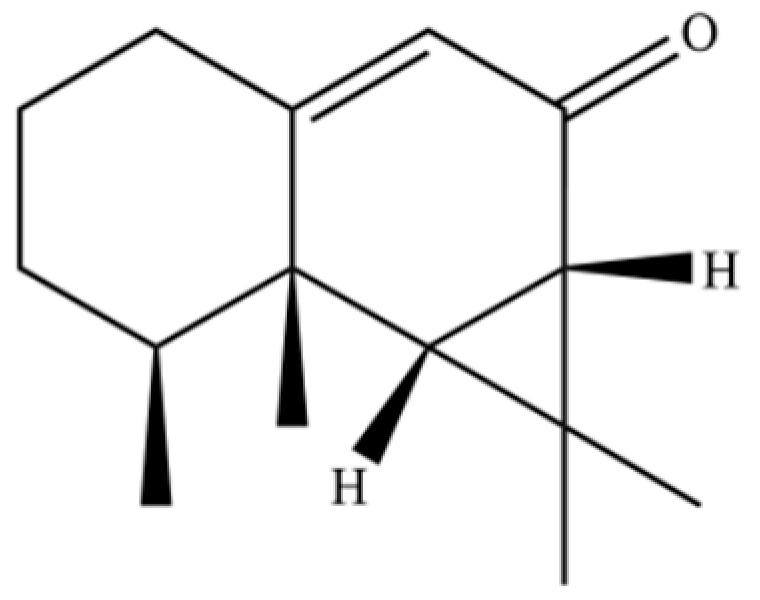 | * | * | Sesquiterpenoid | [51] |
| 262 | diacetyl-atractylodiol | 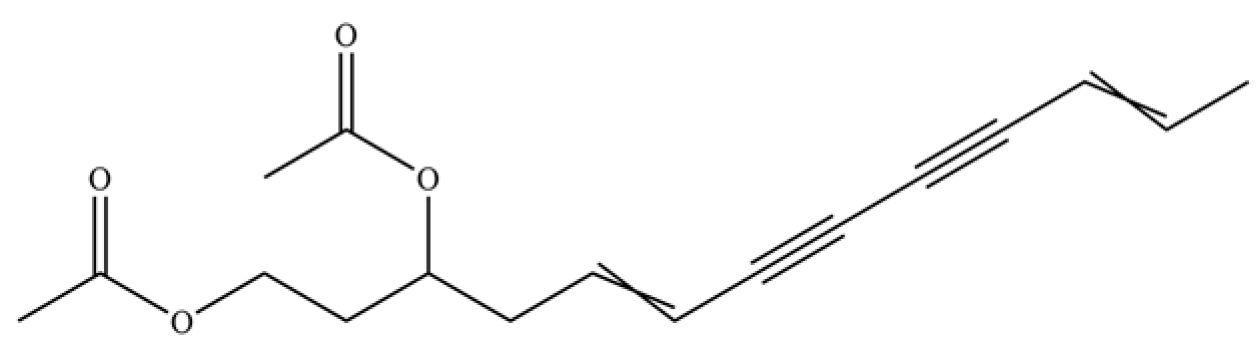 | * | * | Polyacetylene derivative | [67] |
| 263 | (4E,6E,12E)-tetradeca-4,6,12- trien-8,10-diyne-1,3-diyl diacetate | 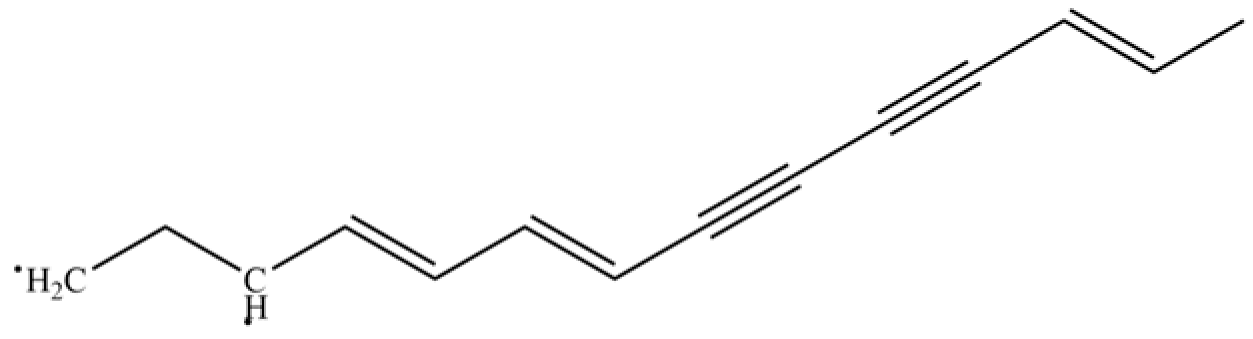 | * | * | Polyacetylene ester | [51] |
| 264 | Acetyltetralone | 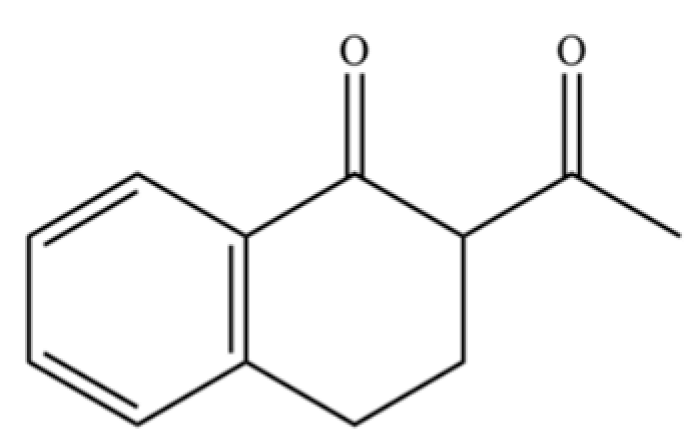 | * | * | Aromatic ketone | [51] |
| 265 | 9a-hydroxy-3,8a-dimethyl-5-methylene-2-oxo-2, 4, 4a, 5, 6, 7, 8, 8a, 9,9a-decahydronaphtho [2,3-b]furan-6-yl acetate | 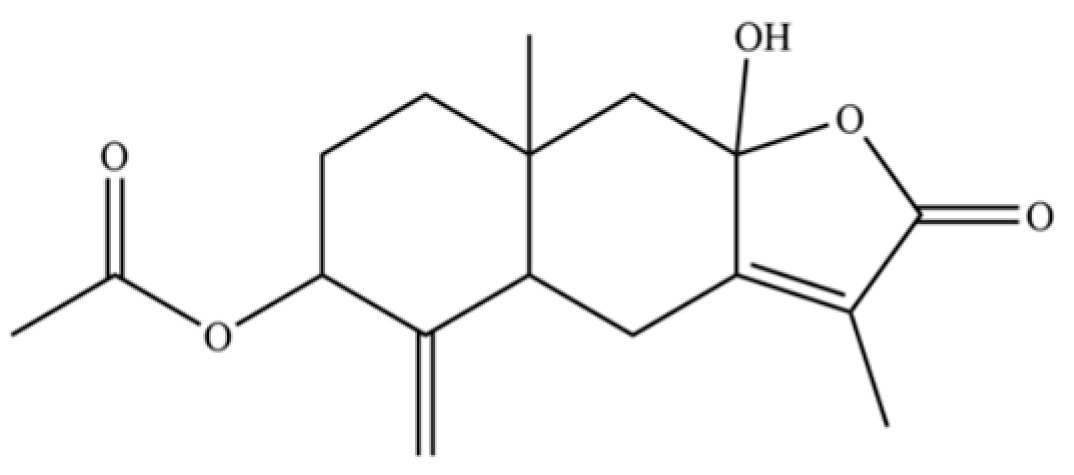 | * | * | Sesquiterpenoid lactone | [51] |
| 266 | 2-(biphenyl-4-yl)acetaldehyde | 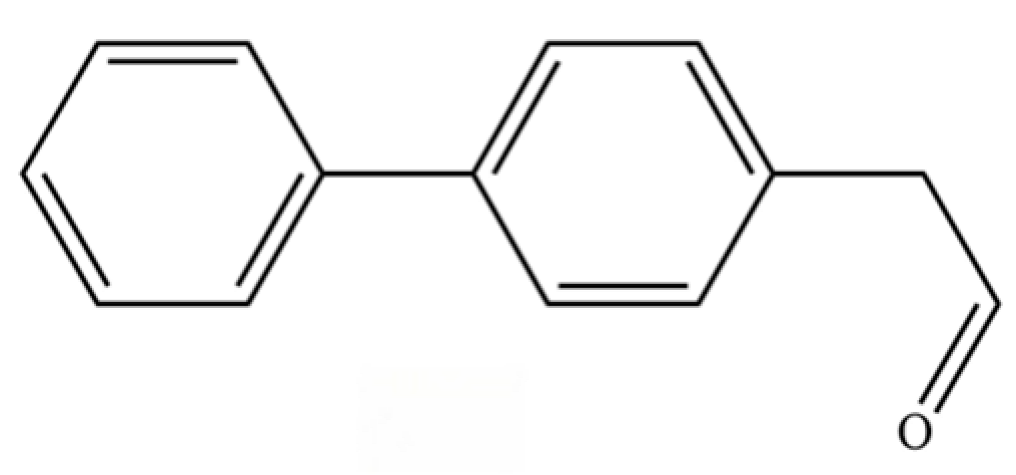 | * | * | Aromatic aldehyde | [42] |
| 267 | 3β-acetoxyatractylon | 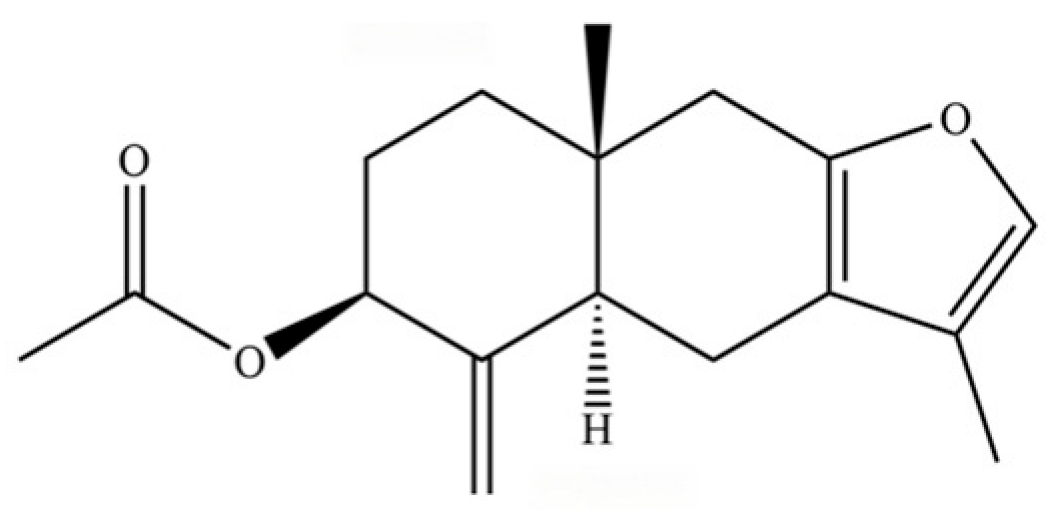 | * | * | Sesquiterpenoid | [51] |
| 268 | atractylenolide II | 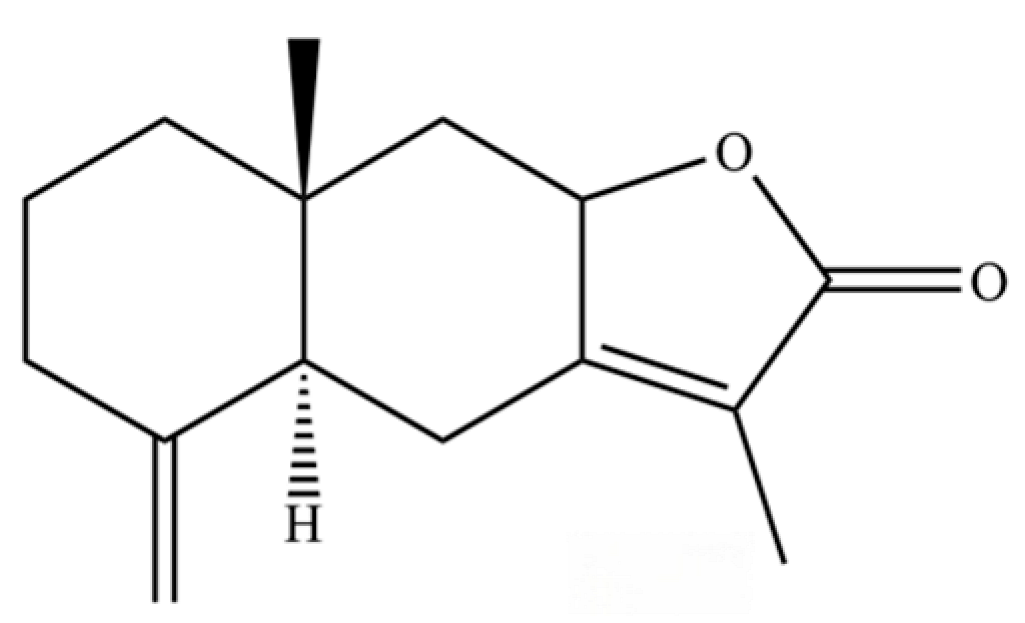 | * | * | Sesquiterpenoid lactone | [60] |
| 269 | atractyloside H | 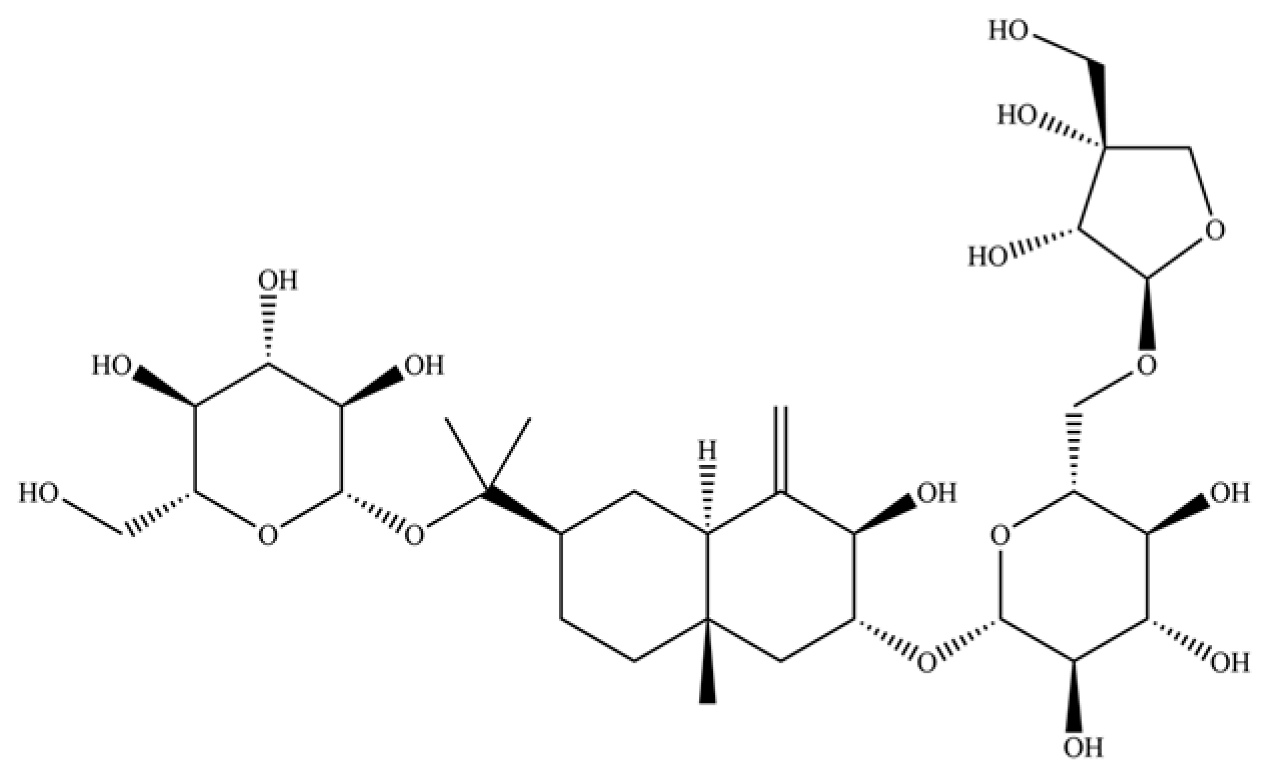 | * | * | Sesquiterpenoid glycoside | [58] |
| 270 | acetylatractylodinol | 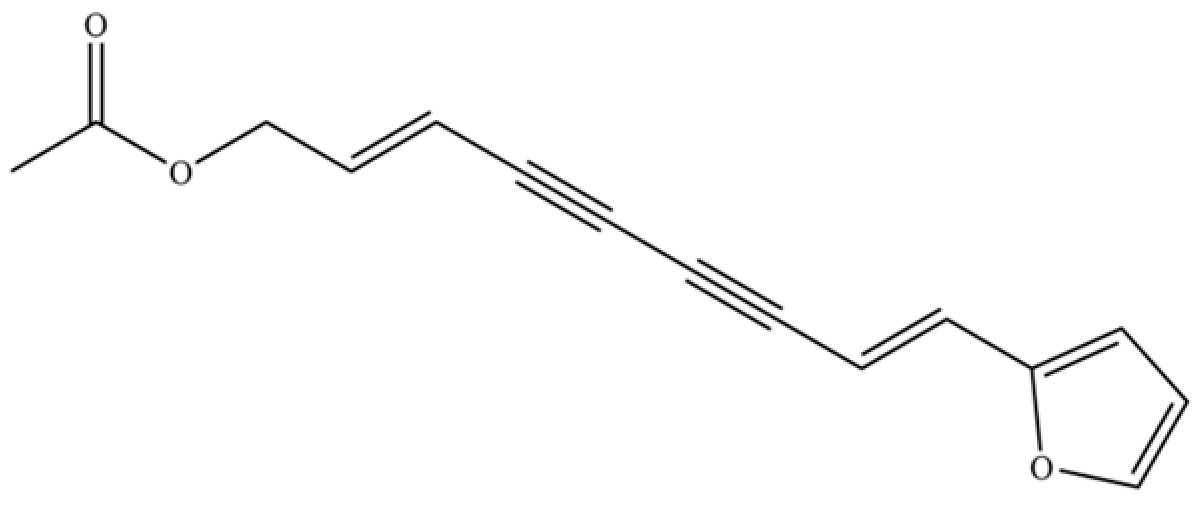 | ** | * | Polyacetylene ester | [68] |
| 271 | atractylenolide I | 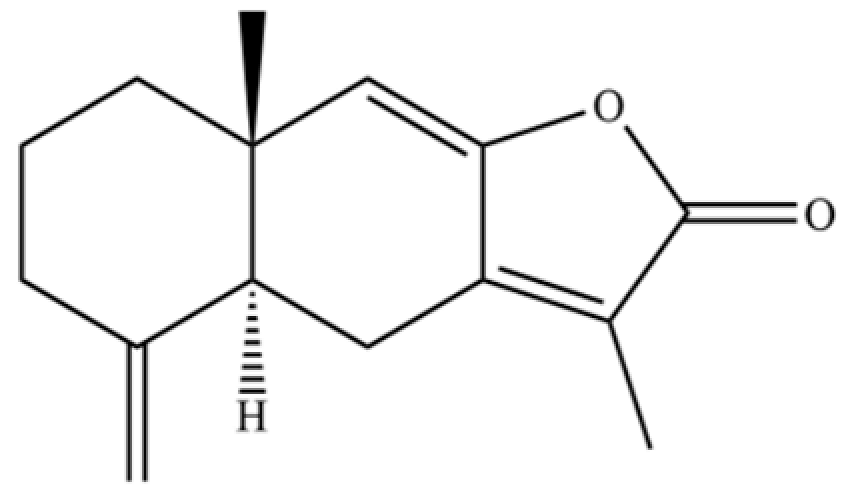 | * | * | Sesquiterpenoid lactone | [21] |
| 272 | 3β-hydroxyatractylon | 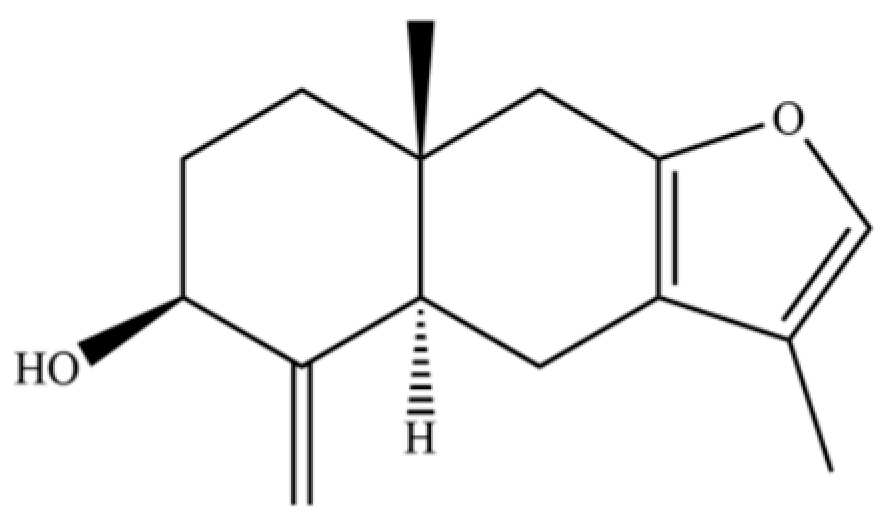 | * | * | Sesquiterpenoid | [58] |
| 273 | α-cyperone | 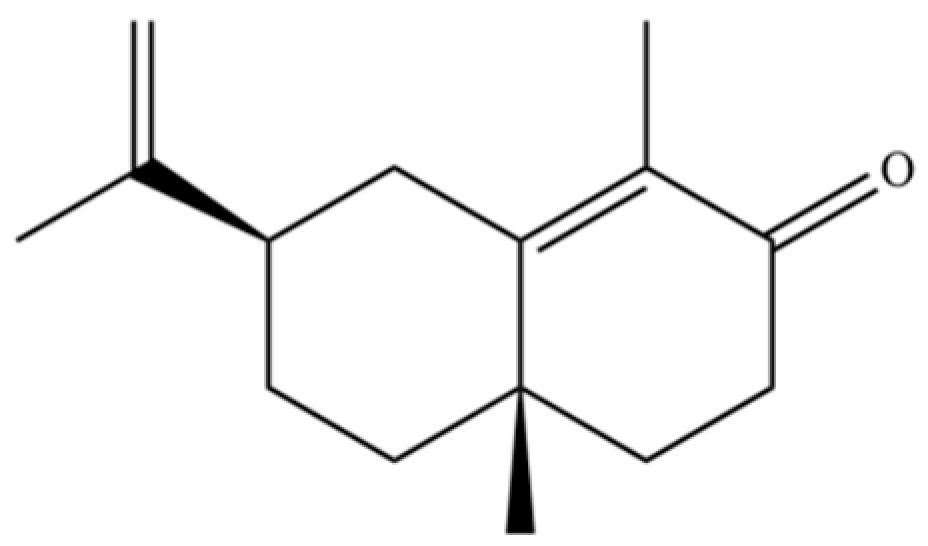 | * | * | Sesquiterpenoid | [69] |
| 274 | 9,10-epoxy-12(Z)-octadecenoic acid | 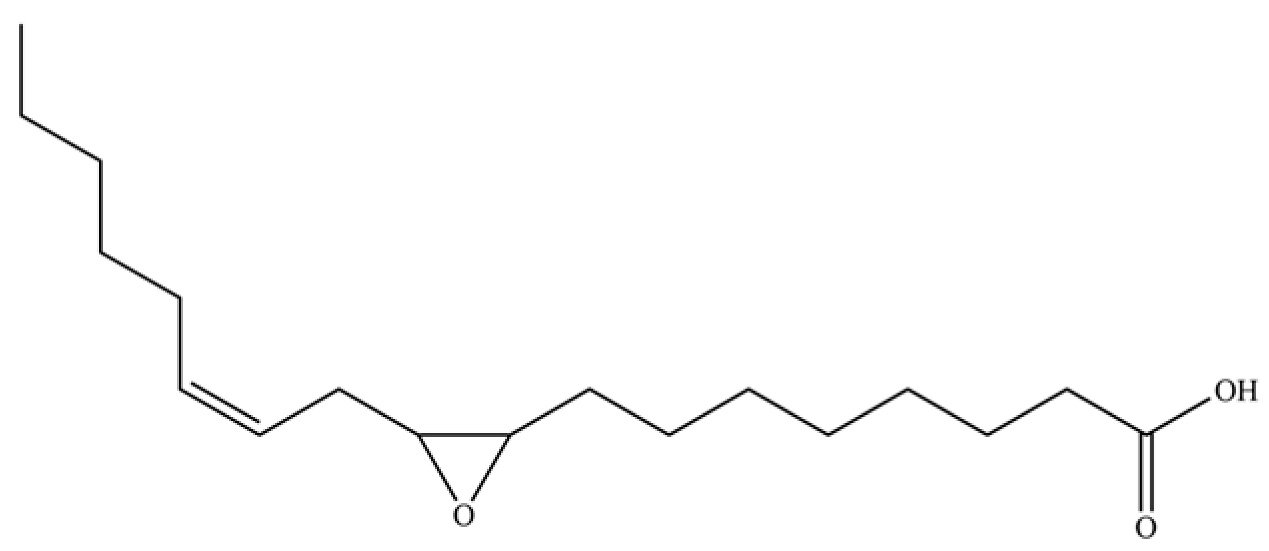 | * | * | Fatty acid epoxide | [51] |
| 275 | (4E,6E,12E)-tetradecadiene-8,10-diyne-1, 3-diol-diacetate | 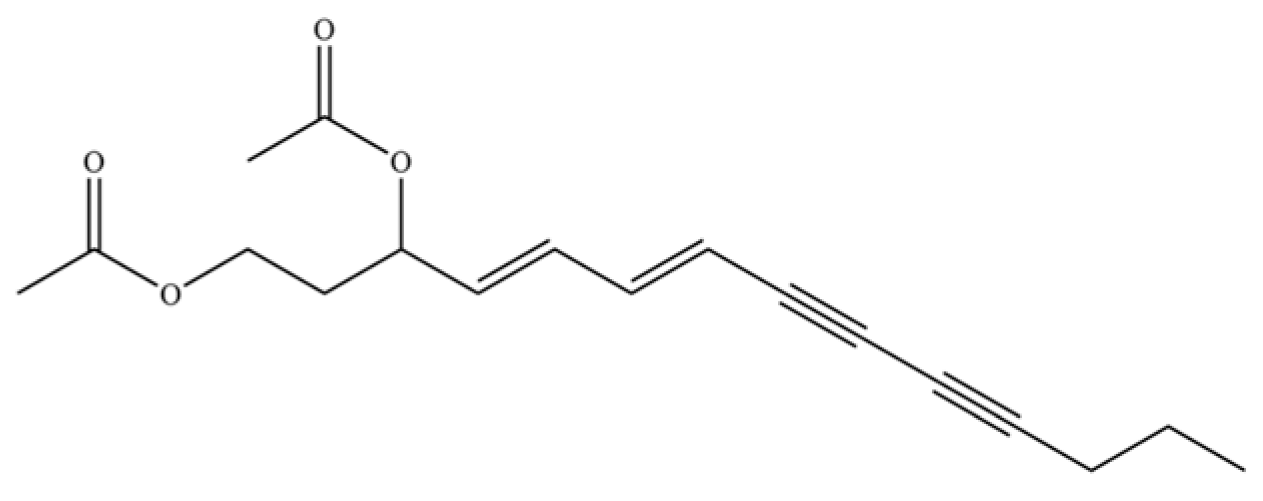 | * | * | Polyacetylene ester | [51] |
| 276 | methyllinolenate | 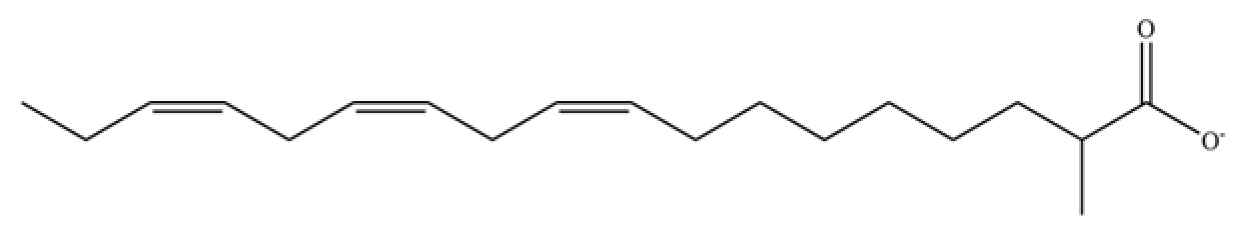 | * | * | Fatty acid methyl ester | [51] |
| 277 | muscone | 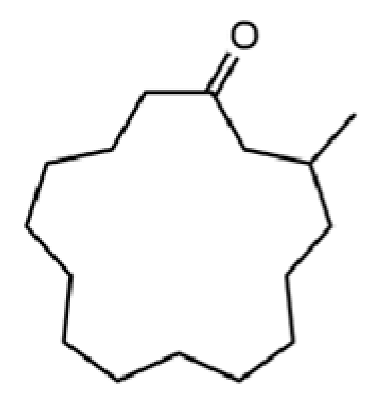 | * | * | Macrocyclic ketone | [51] |
| 278 | 7-methoxy-2-methyl-2-(4- methylpent-3-enyl)-2H- chromene | 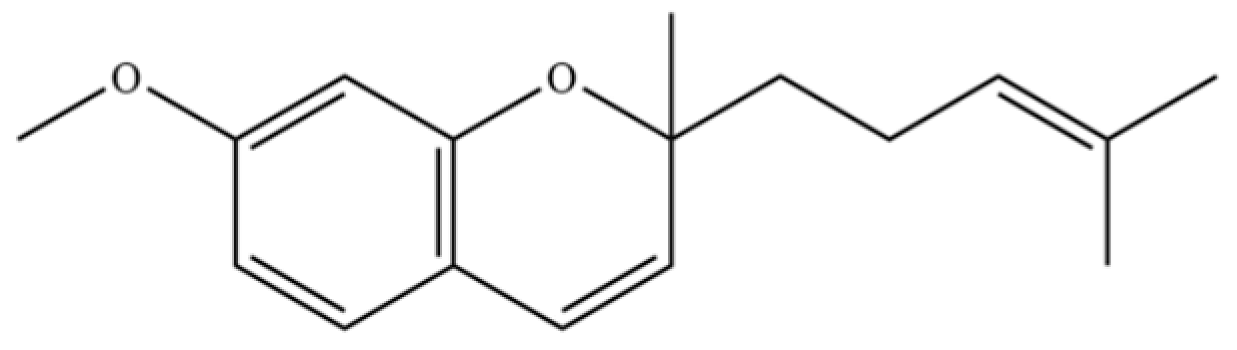 | * | * | Chromene derivative | [51] |
| 279 | 9, 12-octadecadienoic acid | 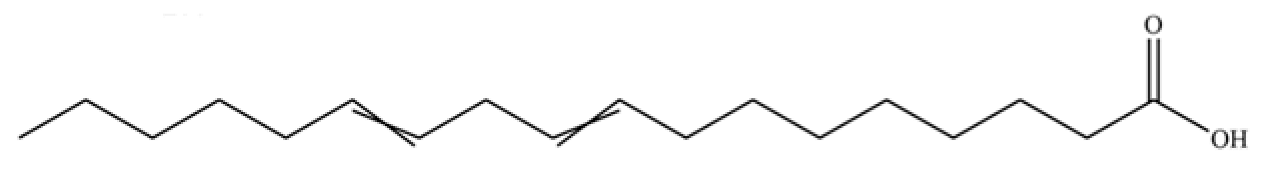 | * | * | Fatty acid | [70] |
| 280 | palmitic acid | 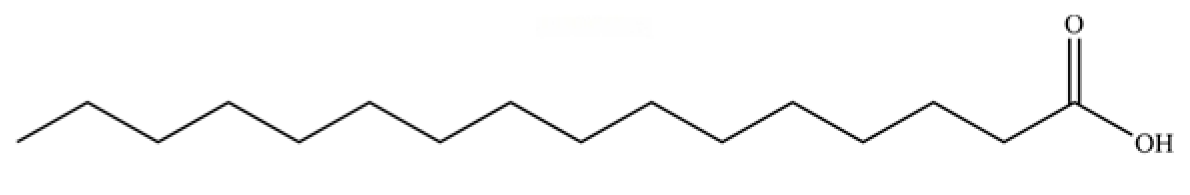 | * | * | Fatty acid | [71] |
| 281 | α-Muurolene | 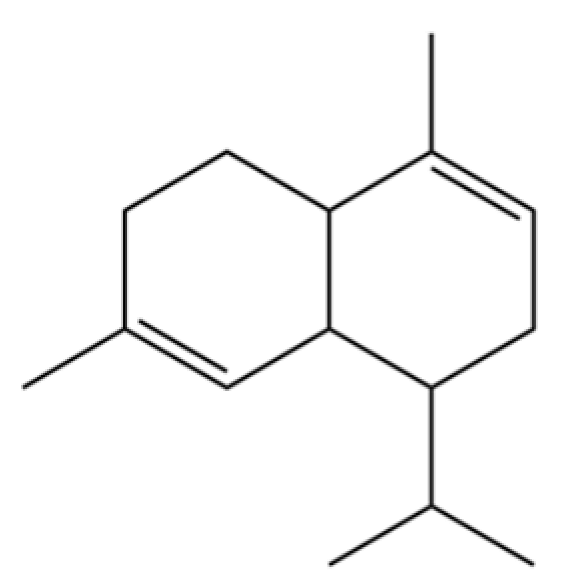 | * | * | Sesquiterpenoid | [72] |
| 282 | δ-Cadinene | 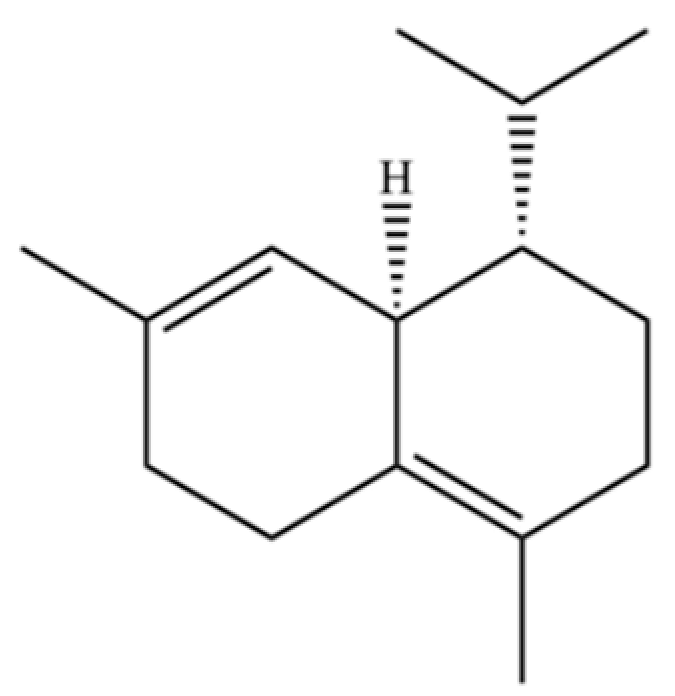 | * | * | Sesquiterpenoid | [21] |
| 283 | Naphthalene | 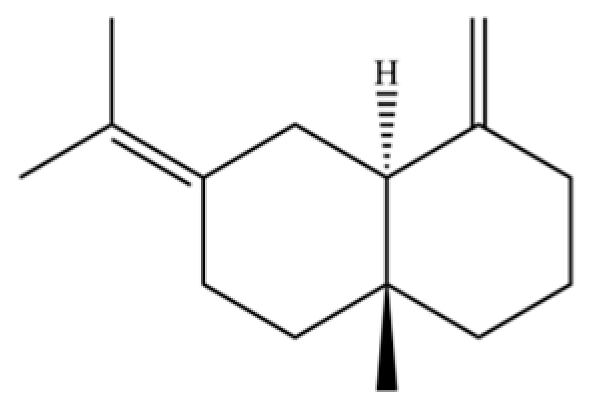 | * | * | Aromatic hydrocarbon | [72] |
| 284 | Aristolone | 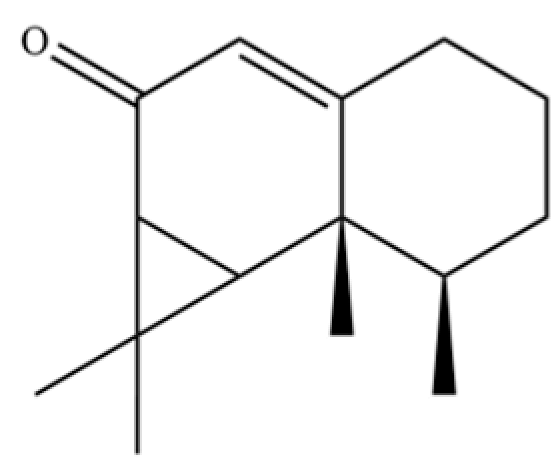 | * | * | Sesquiterpenoid | [72] |
| 285 | Syringin | 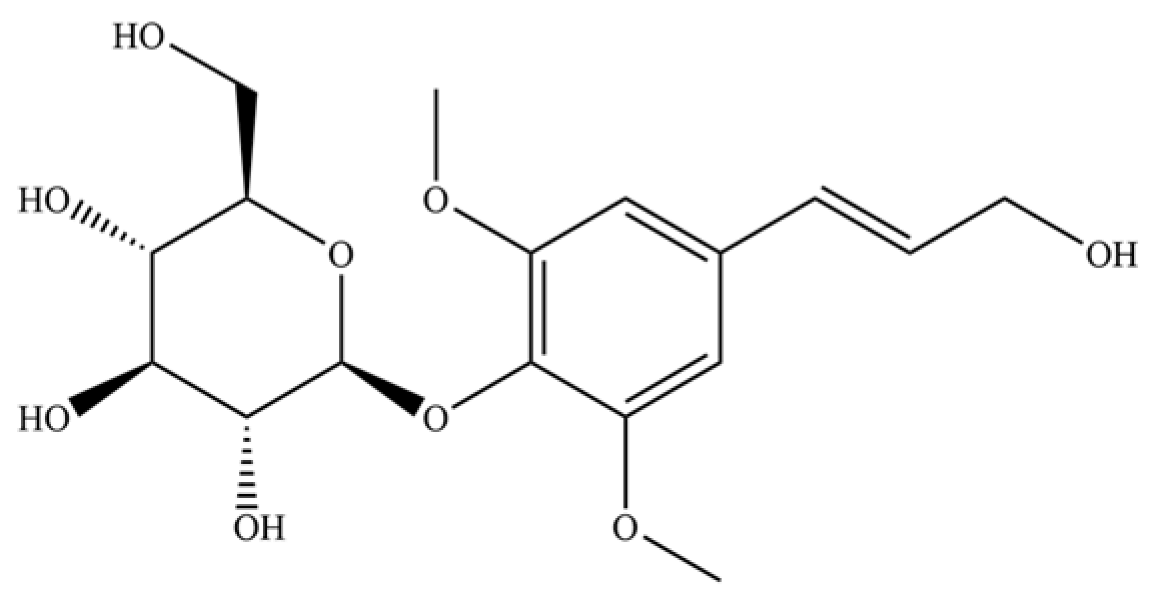 | * | * | Phenolic glycoside | [73] |
| 286 | Undecanedioic acid | 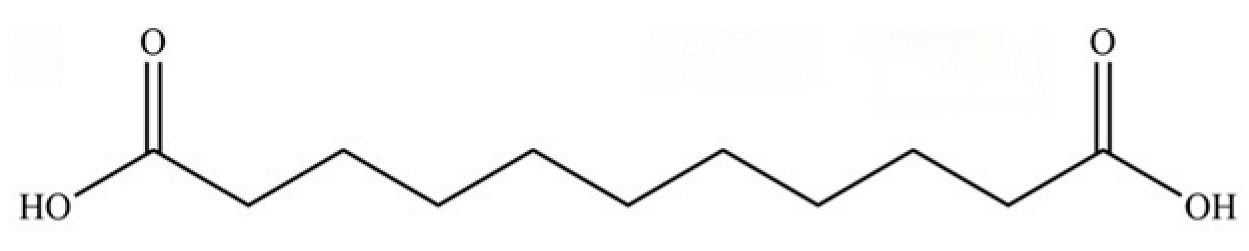 | * | * | Dicarboxylic acid | [74] |
| 287 | Parthenolide | 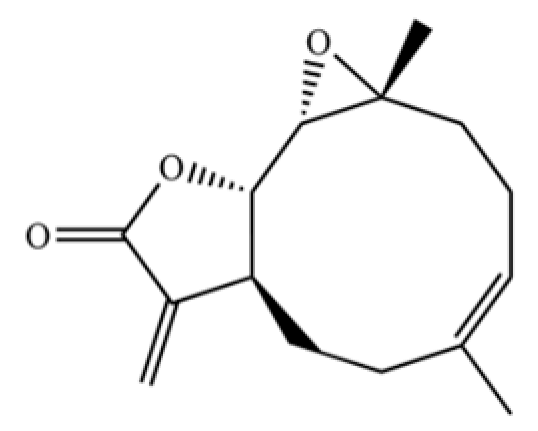 | * | * | Sesquiterpenoid lactone | [74] |
| 288 | Valerenic acid | 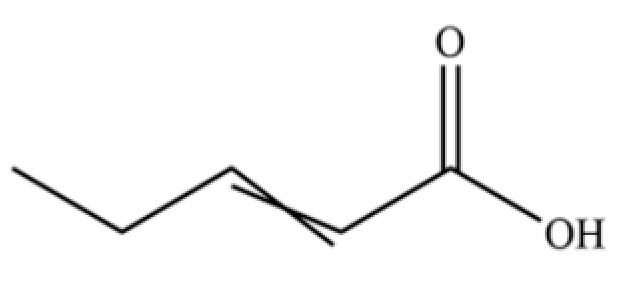 | * | * | Sesquiterpenoid acid | [75] |
| 289 | 3-methyl-1-phenylpent-1-yn-3-ol | 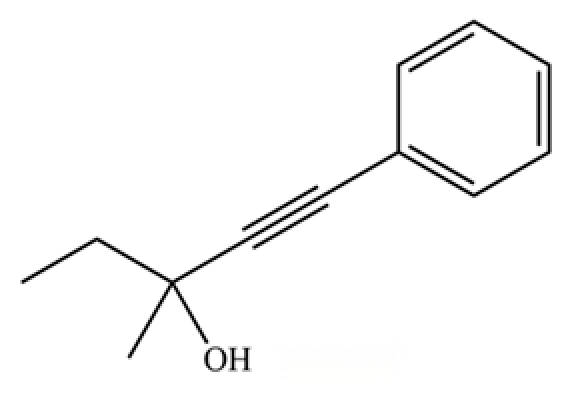 | * | * | Aromatic alcohol | [74] |
| 290 | α-Cyclohexylmandelic acid | 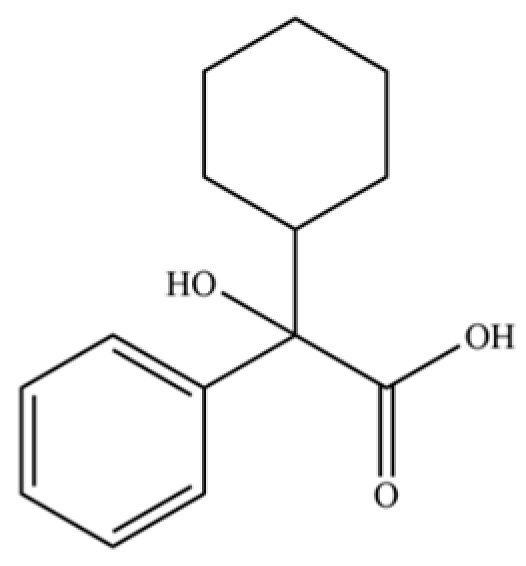 | * | * | Aromatic acid | [74] |
| 291 | 11-propan-2-ylidenetricyclo[4.3.1.12,5]undec-3-en-10-one | 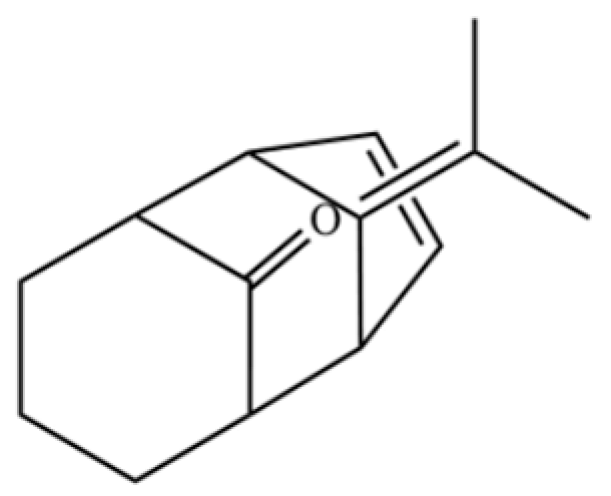 | * | * | Sesquiterpenoid | [74] |
| 292 | Hydroxyvalerenic acid | 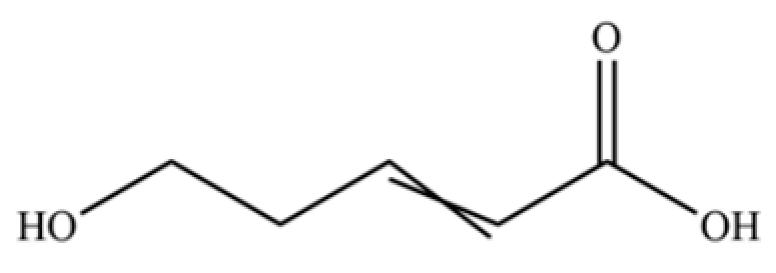 | * | * | Sesquiterpenoid acid | [74] |
| 293 | 3,5-ditert-butylbenzene-1,2-diol | 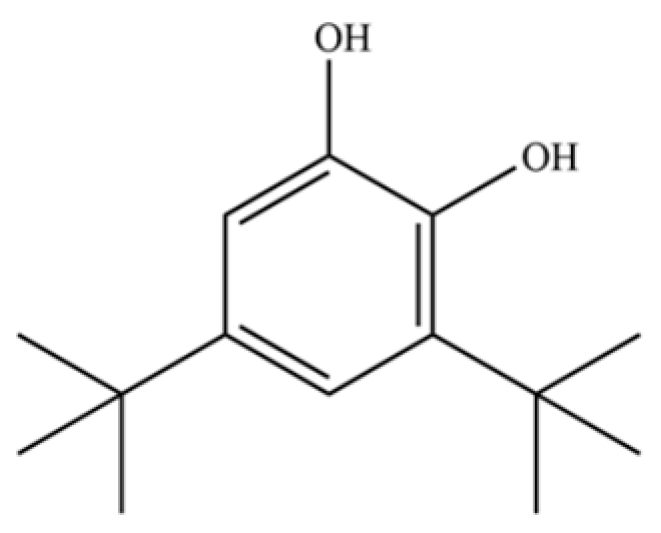 | * | * | Phenolic | [74] |
| 294 | Senkyunolide A | 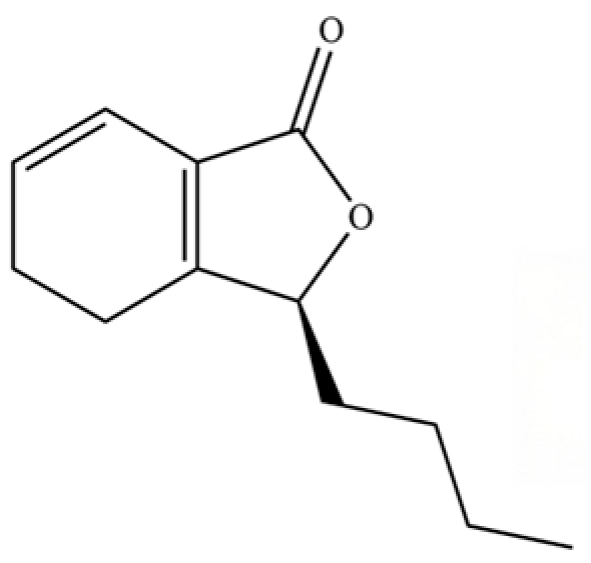 | * | * | Phthalide | [74] |
| 295 | Nootkatone | 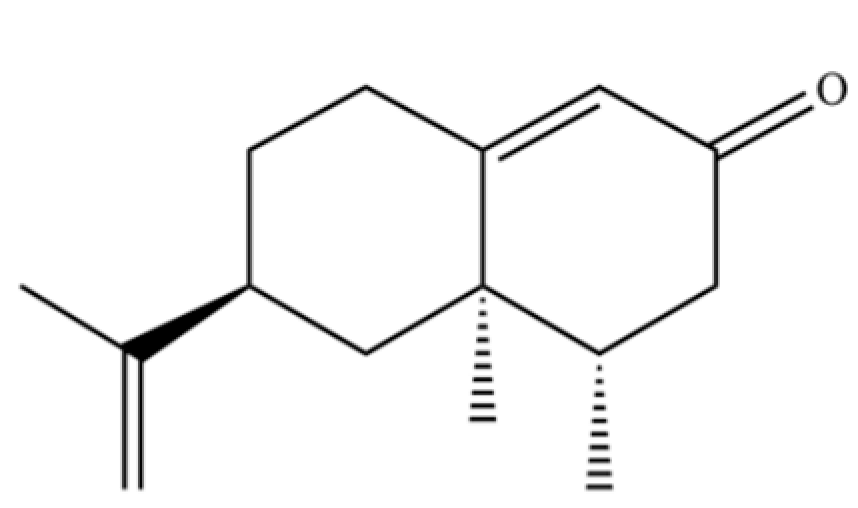 | * | * | Sesquiterpenoid ketone | [74] |
| 296 | 12,13-dihydroxy-9Z-octadecenoic acid | 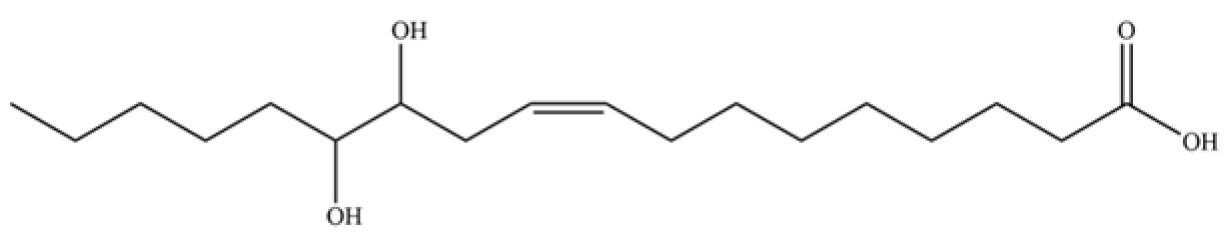 | * | * | Fatty acid | [74] |
| 297 | 2-hydroxyfluorene | 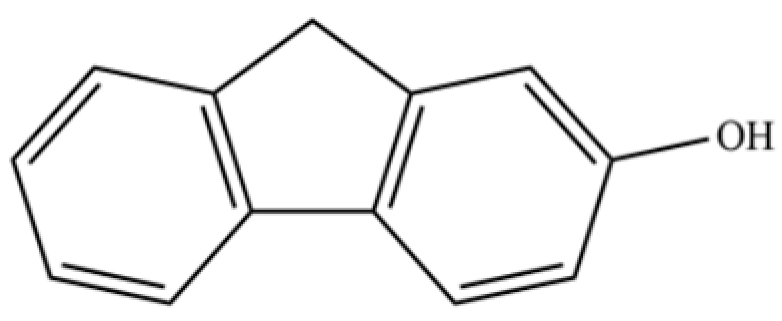 | * | * | Phenolic | [26] |
| 298 | Isoalantolactone | 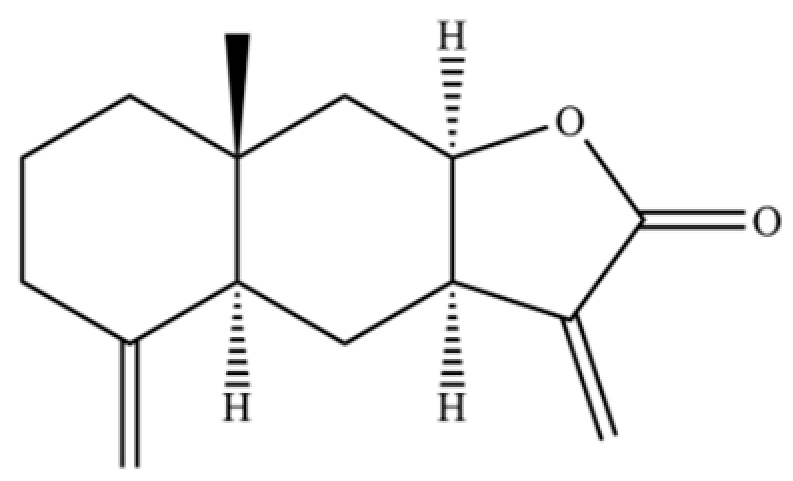 | * | * | Sesquiterpenoid lactone | [74] |
| 299 | Xanthydrol | 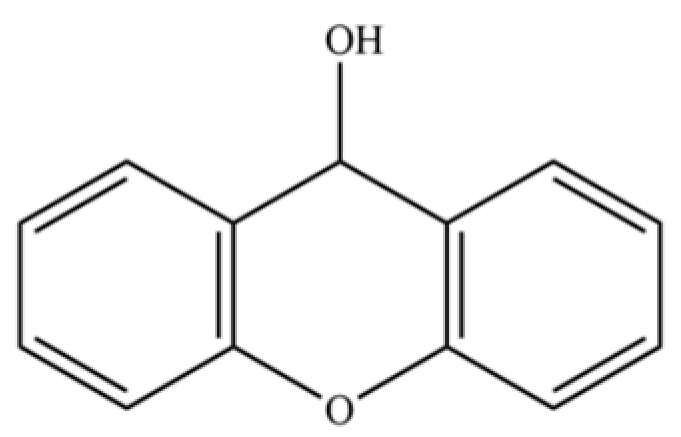 | * | * | Xanthene derivative | [74] |
| 300 | Bis (4-methoxyphenyl) methanol | 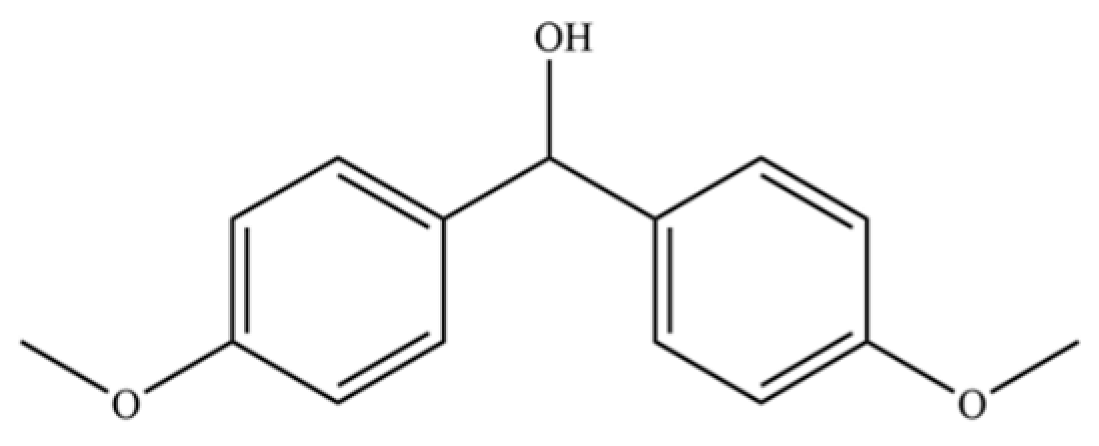 | * | * | Aromatic alcohol | [74] |
| 301 | 1-naphthalenemethanol | 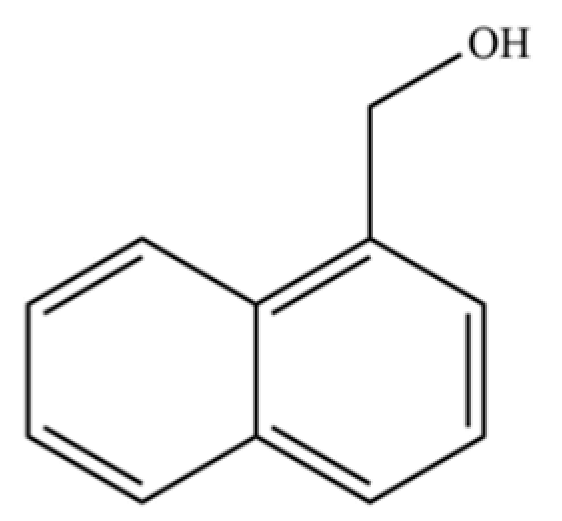 | * | * | Aromatic alcohol | [74] |
| 302 | 2-methylbenzhydrol | 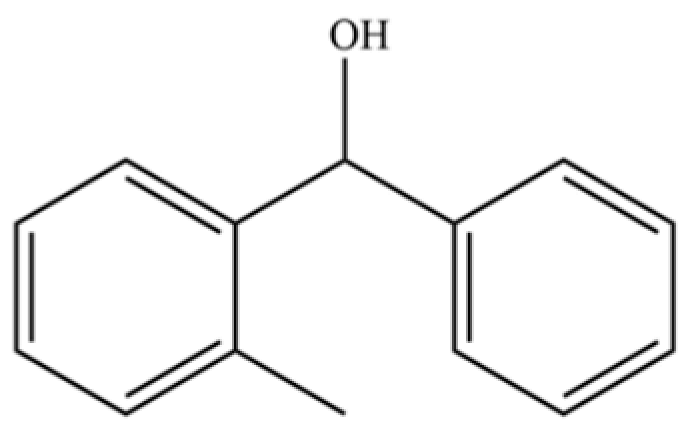 | * | * | Aromatic alcohol | [74] |
| 303 | 3,3′,5,5′-tetramethyldiphenoquinone | 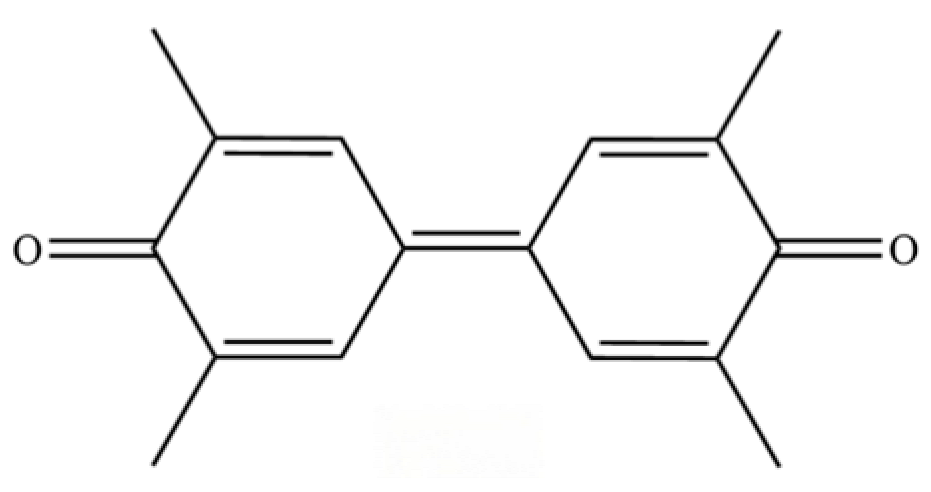 | * | * | Quinone | [74] |
| 304 | (cis+trans)-nerodilol | 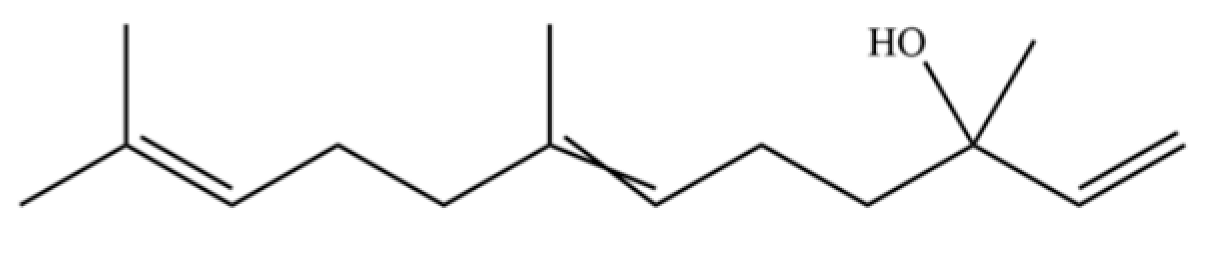 | * | * | Sesquiterpenoid | [74] |
| 305 | Germacrone | 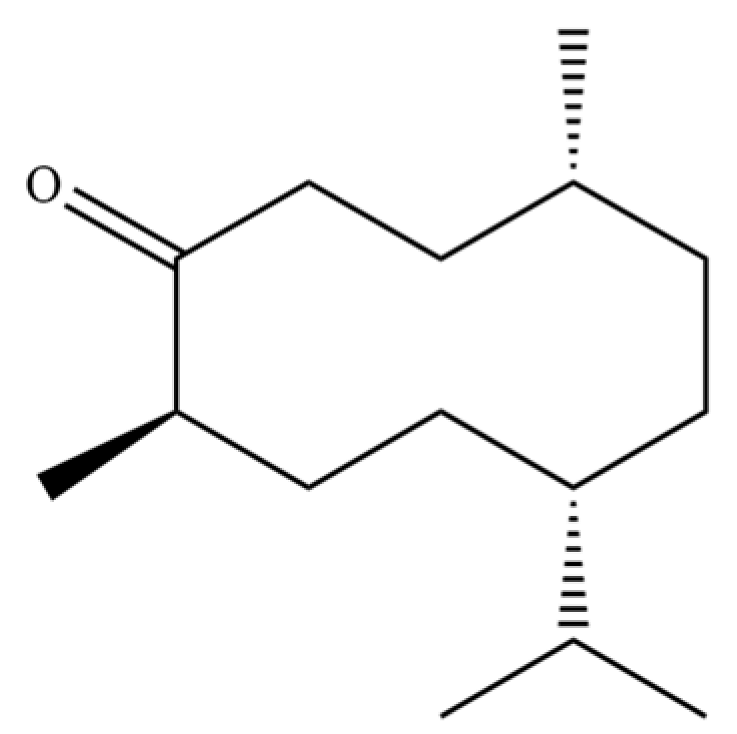 | * | * | Sesquiterpenoid | [74] |
| 306 | 1-palmitoyl-2-hydroxy-sn-glycero-3-phosphoethanolamine | 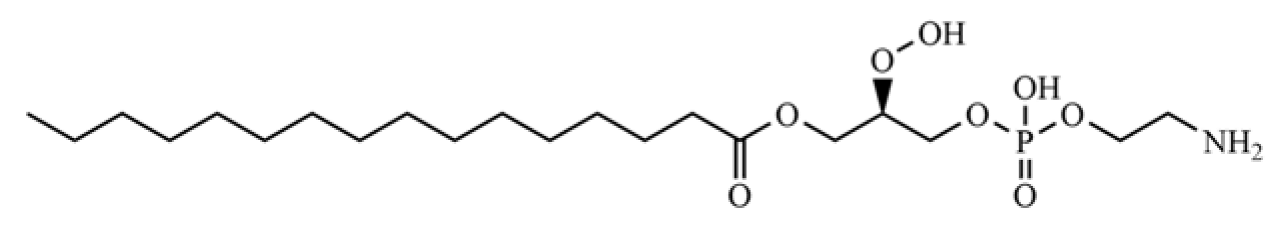 | * | * | Phospholipid | [74] |
| 307 | 1,3-benzenediol, 5-methyl-2-[(1R,6R)-3-methyl-6- (1-methylethenyl)-2-cyclohexen-1-yl] | 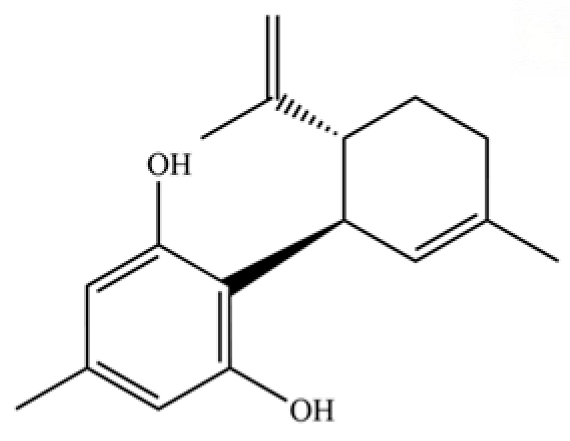 | * | * | Phenolic sesquiterpenoid | [74] |
| 308 | 1-oleoyl-L-α-lysophosphatidic acid | 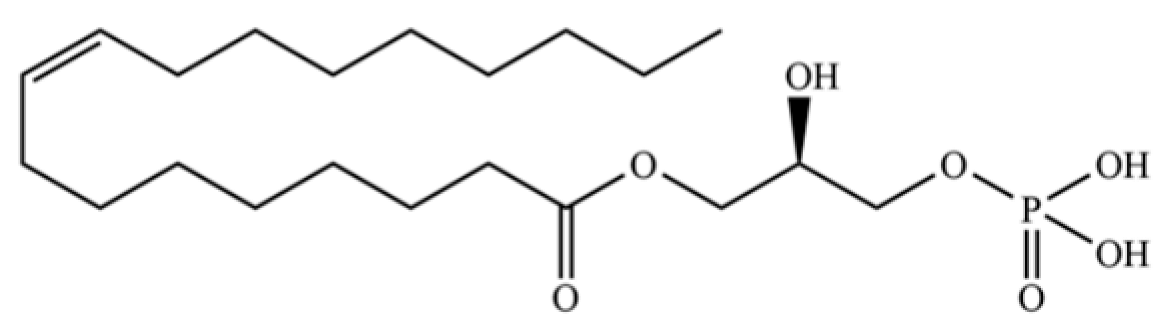 | * | * | Phospholipid | [74] |
| 309 | 2-hydroxypalmitic acid | 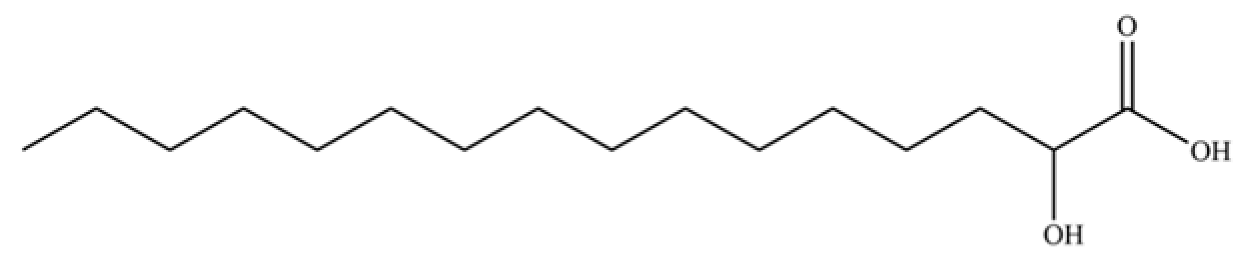 | * | * | Fatty acid | [74] |
| 310 | Oleamide | 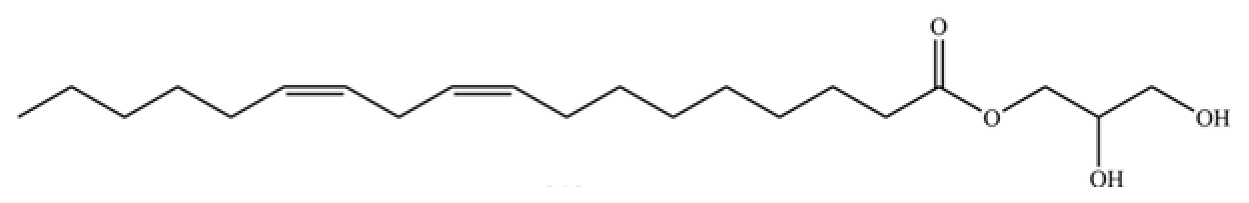 | * | * | Fatty amide | [74] |
| 311 | 9E,11E-octadecadienoic acid | 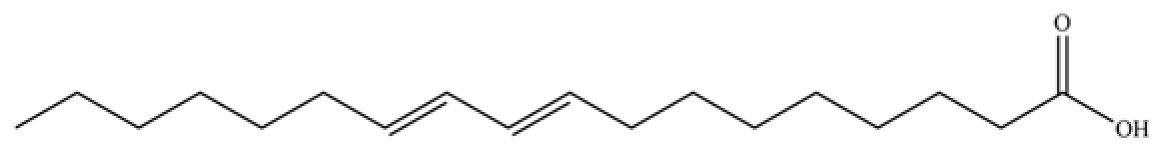 | * | * | Fatty acid | [74] |
| 312 | 1,2-benzenedicarboxylic acid | 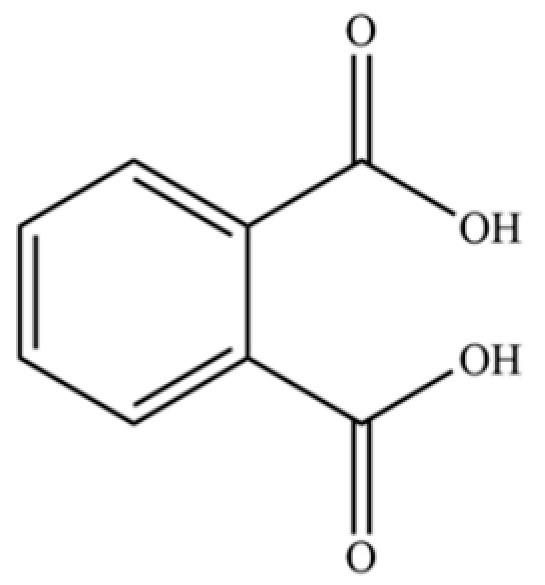 | * | * | Phthalic acid (artifact) | [74] |
| 313 | Bis (2-ethylhexyl) phthalate | 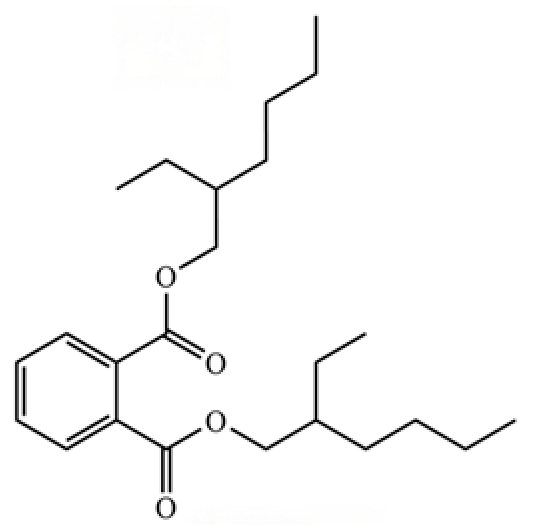 | * | * | Phthalate ester (artifact) | [74] |
| 314 | Terpinolene | 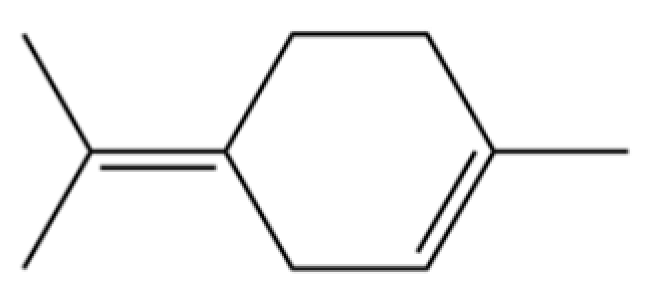 | * | * | Monoterpenoid | [21] |
| 315 | 1-methyl-4-(1-methylethyl)-2-cyclohexen-1-ol | 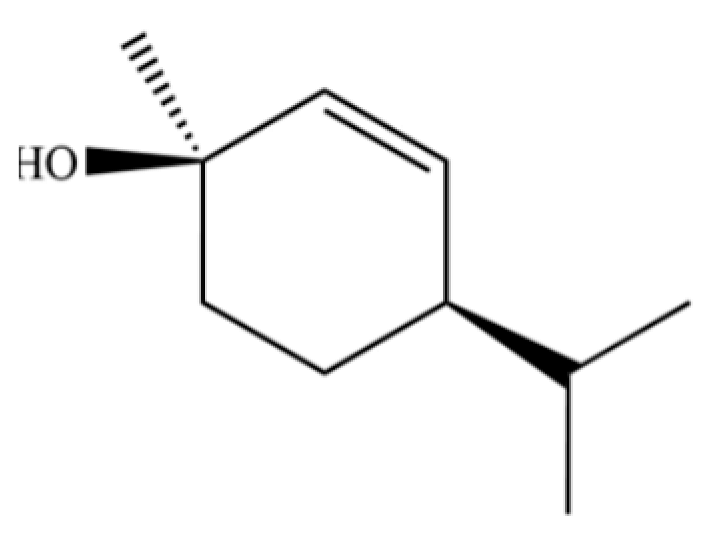 | * | * | Monoterpenoid | [76] |
| 316 | (1R,3R,5R)-1-Isopropyl-4-methylenebicyclo[3.1.0]hexan-3-ol | 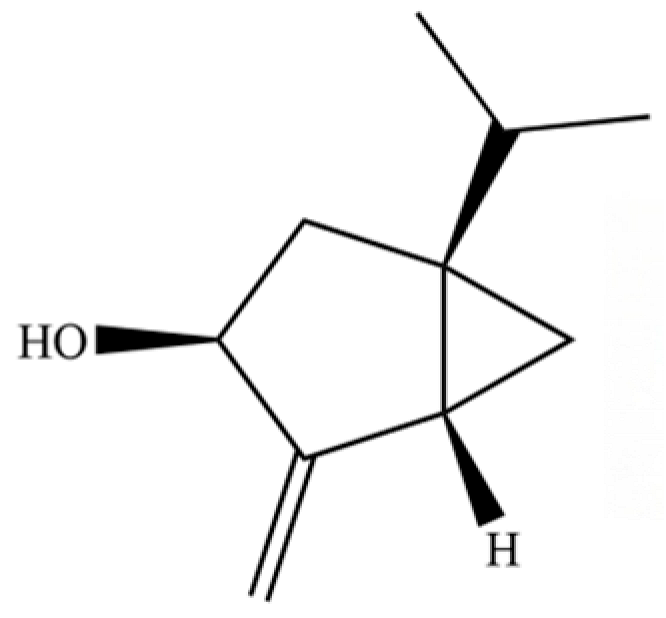 | * | * | Monoterpenoid | [76] |
| 317 | Methyl geraniate | 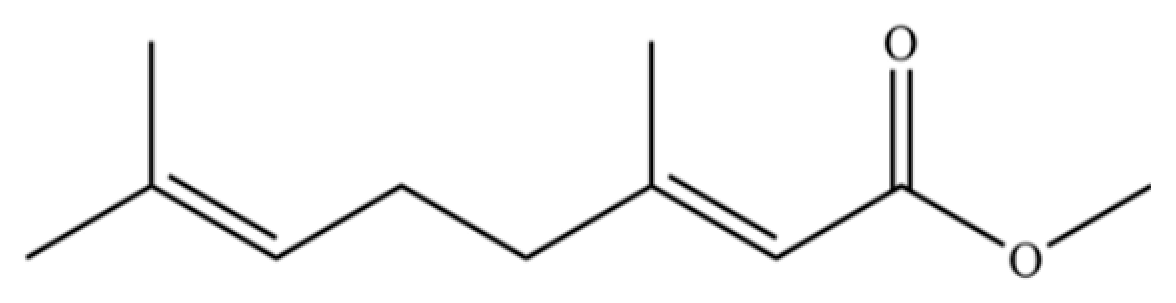 | * | * | Monoterpenoid ester | [76] |
| 318 | Silphiperfol-5-ene | 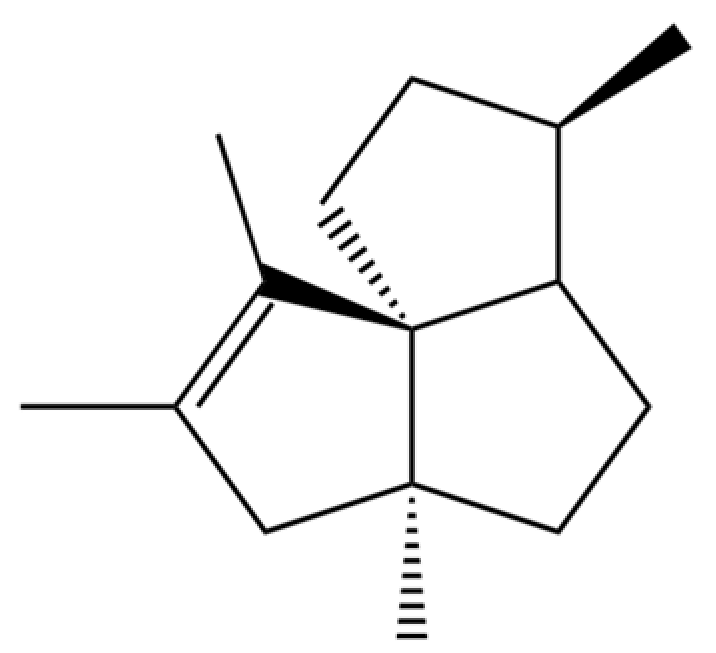 | * | * | Sesquiterpenoid | [76] |
| 319 | δ-Elemene | 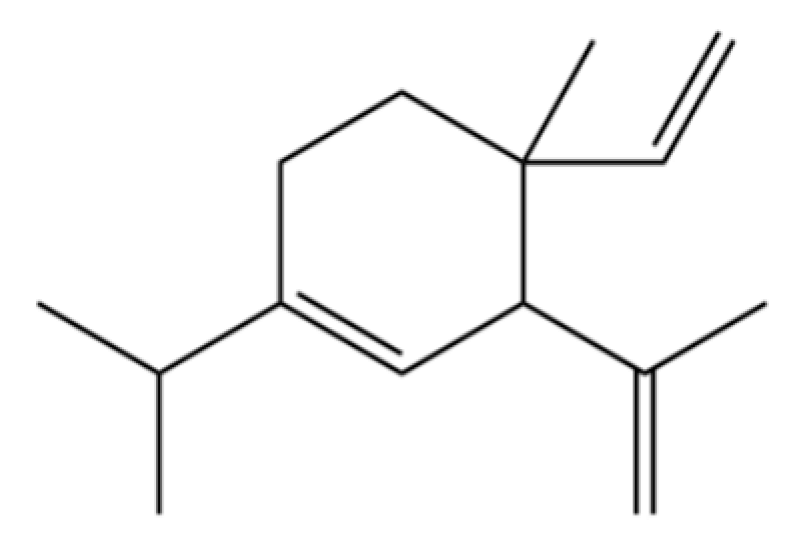 | * | * | Sesquiterpenoid | [21] |
| 320 | 7-epi-Silphiperfol-5-ene | 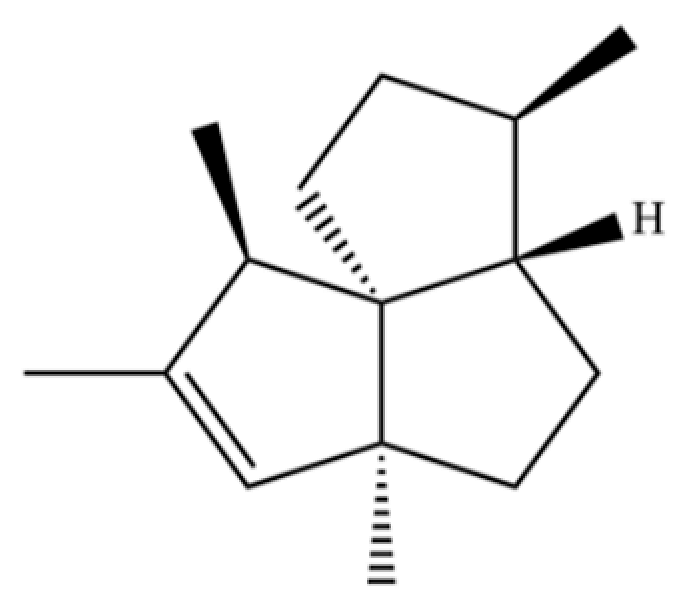 | * | * | Sesquiterpenoid | [76] |
| 321 | Silphinene | 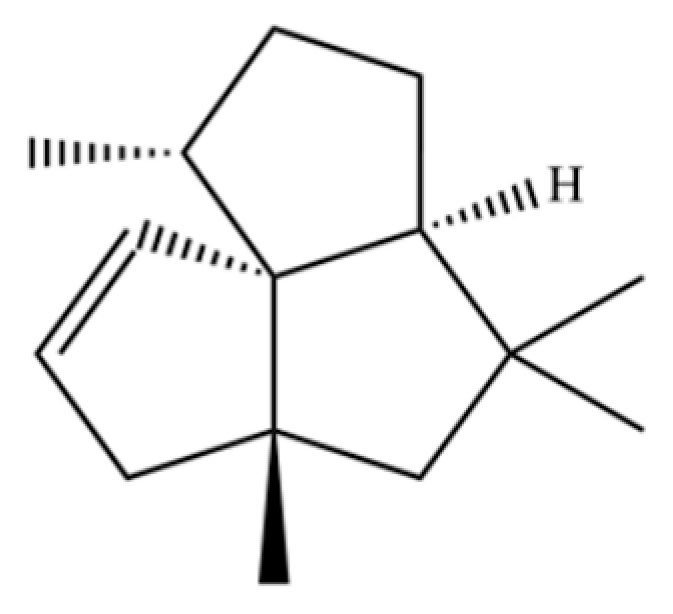 | * | * | Sesquiterpenoid | [76] |
| 322 | γ-selinene |  | * | * | Sesquiterpenoid | [25] |
| 323 | 1-Naphthalenol, 1,2,3,4,4a,5,6,7-octahydro-4a,5-dimethyl-3-(1-methylethenyl)- |  | * | * | Sesquiterpenoid | [76] |
| 324 | 7-epi-alpha-eudesmol |  | * | * | Sesquiterpenoid | [76] |
| 325 | β-guaiene |  | * | * | Sesquiterpenoid | [3] |
| 326 | valencene |  | * | * | Sesquiterpenoid | [77] |
| 327 | Atrachinenins D |  | / | ** | Sesquiterpenoid | [23] |
Note: (1) **: High relative content in the species. (2) *: Present but with lower or unspecified relative content. (3) /: Not reported or considered absent in the species. (4) AL: Atractylodes lancea; AC: Atractylodes chinensis.
5. Pharmacology
5.1. Antimicrobial Effect
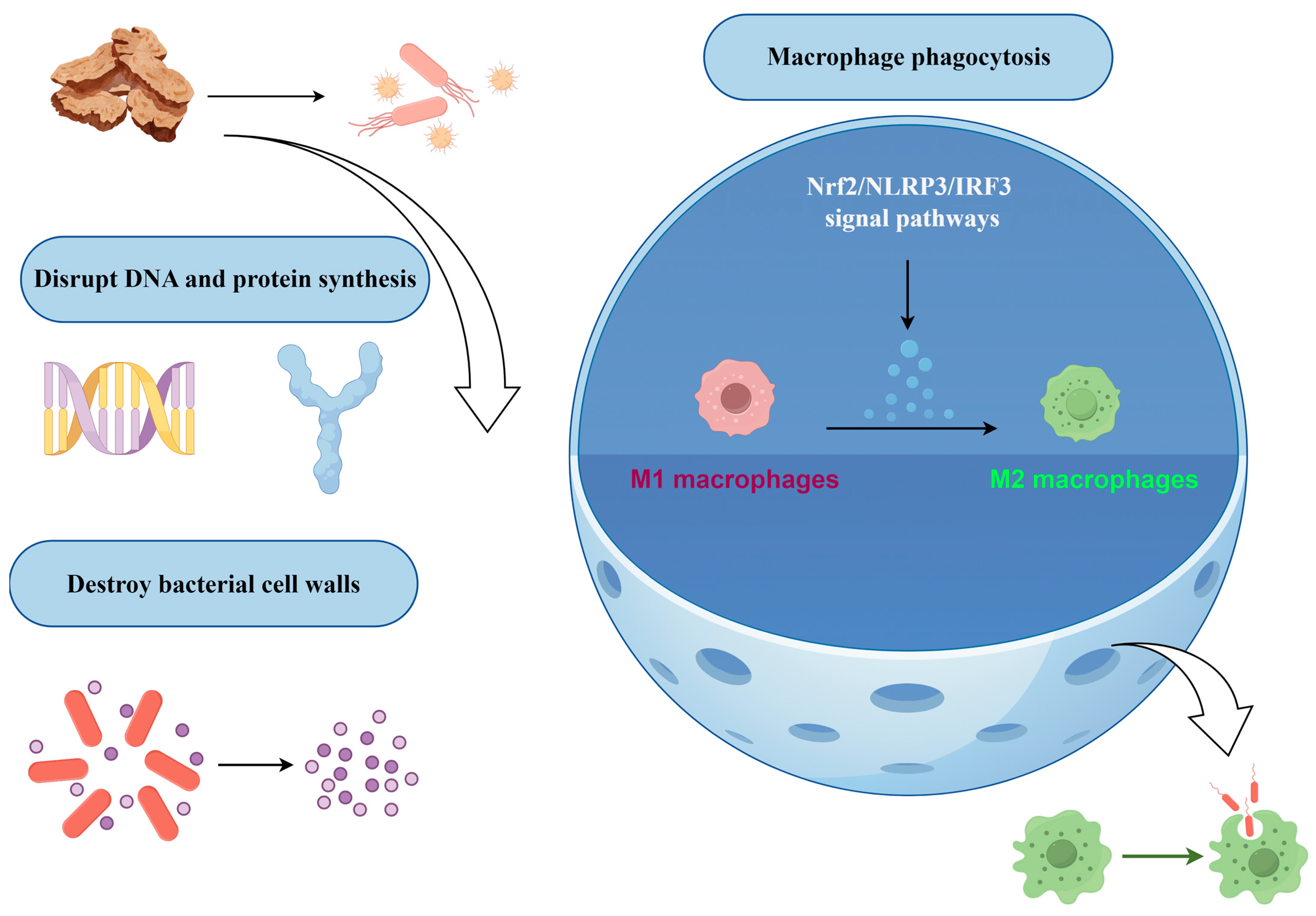
AR exhibits notable antimicrobial properties, particularly against bacteria, parasites, and viruses, as illustrated in Table S3 and Figure 4. Studies have demonstrated that the chloroform extract of AR significantly impedes the adhesion and invasion capabilities of Salmonella typhimurium in vitro [78]. A primary factor contributing to the pronounced antibacterial activity of AR is its high concentration of sesquiterpene compounds, such as cyperene, caryophyllene, aciphyllene, and humulene, among others. The antibacterial efficacy of AR is characterized by a multi-mechanism synergistic pattern at both molecular and cellular levels, with its material basis primarily attributed to a diverse array of sesquiterpene compounds. These components interfere with the survival and pathogenic processes of pathogens through distinct molecular pathways, thereby establishing a broad-spectrum antimicrobial system with potential synergistic effects. The core mechanism of its action involves two principal aspects. Firstly, it directly disrupts microbial cell membranes by increasing membrane permeability through the insertion of hydrophobic compounds [79,80]. This disruption results in intracellular electrolyte leakage, the collapse of the transmembrane proton gradient, the efflux of cellular contents, and ultimately, cell death. Additionally, it targets critical enzymes and the synthesis of biomacromolecules. Research has demonstrated that germacrene D (35) effectively inhibits Staphylococcus aureus by strongly binding to tyrosyl-tRNA synthetase and topoisomerase II, thereby obstructing protein synthesis and DNA replication [81]. Similarly, molecular docking studies predict that humulene (30) acts on DNA gyrase, disrupting bacterial DNA supercoiling [82]. Furthermore, it interferes with quorum sensing and the expression of virulence factors. For example, α-thujene (143) exhibits high affinity for the virulence regulatory proteins LasR and PqsR in Pseudomonas aeruginosa, suggesting its potential to impede bacterial quorum sensing, weaken biofilm formation, and reduce virulence as an anti-virulence agent [83]. Notably, certain components of AR, such as cyperene (21), have been identified for their potential to enhance overall antibacterial efficacy by promoting synergistic interactions among various constituents [84]. Although the aforementioned approach yielded favourable results, the mechanisms described were predominantly derived through virtual screening and computer-aided simulations, necessitating further validation. This finding presents a fresh perspective on tackling the issue of antibiotic resistance frequently linked with single-target antibiotics.
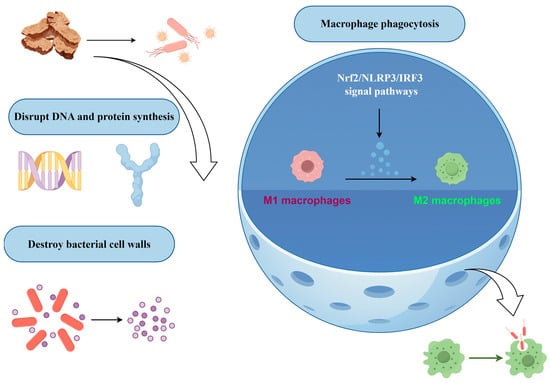
Figure 4.
Schematic Diagram of the Antimicrobial Mechanism of Atractylodis Rhizoma. Note: The arrows in the figure indicate interactions; the red areas represent M1 macrophages, and the green areas represent M2 macrophages. In addition, the first row in the upper left corner represents the effects of AR on microorganisms; the second row shows bacterial DNA and proteins; and the third row depicts the disruption of bacterial internal structures. In the right-hand diagram, the small blue spheres in the center represent chemokines, and the bottom row illustrates phagocytosis by macrophages.
The structure-activity relationships of the active ingredients and their in vivo transformations remain inadequately understood. The majority of research has focused on crude extracts or component mixtures, which complicates the identification of individual contributions. Furthermore, the extrapolation of in vitro antimicrobial activity to in vivo efficacy is constrained by the pharmacokinetic properties and chemical stability of the components, resulting in a substantial deficiency of pertinent data. Consequently, future research should progress beyond mere characterization screening to include experimental validation at functional targets, efficacy evaluation in drug-resistant bacterial models, and rational design based on pharmacophores. This comprehensive approach will facilitate the conversion of the antibacterial potential of AR into novel anti-infective drugs with well-defined mechanisms and clinical translational value.
Beyond its antibacterial properties, AR exhibits diverse biological activities in both antiviral and antiparasitic contexts. Its mechanisms of action encompass various stages of the viral life cycle and critical physiological processes in parasites. Regarding antiviral activity, the active components demonstrate stage-specific intervention capabilities: β-pinene (119) directly influences the adsorption and replication processes of adenovirus type 3 [85]; furanodienone (137) may provide protective effects during the initial phases of viral infection by modulating cell membrane integrity or preserving intracellular reducing conditions [86]; quinic acid (235) derivatives selectively inhibit the intracellular replication of dengue virus without interfering with viral entry [87]; and atractyloside A (243) induces an antiviral immune response by activating the host’s type I interferon signaling pathway (Table S3) [88]. These findings indicate that the antiviral properties of AR exhibit dual characteristics: direct viral suppression and modulation of the host immune response. In terms of antiparasitic activity, component 1-eicosanol (101) demonstrates acaricidal properties that may be associated with its interference in the mite nervous system or its regulation of body wall osmotic pressure [89]. This discovery presents a promising candidate molecule for the development of plant-derived acaricides. However, research in this area is still in the active screening phase, with mechanisms of action primarily based on speculation. Systematic evaluations concerning target confirmation, in vivo efficacy, and environmental safety are currently insufficient. Future investigations should prioritize cross-species and multi-model studies to clarify the specific molecular targets that underlie its antiviral and antiparasitic effects. Furthermore, the potential synergistic effects of its antibacterial components in addressing complex infections or vector-borne diseases warrant exploration. This comprehensive approach will elucidate the scientific value of AR as a multi-target anti-infective medicinal resource.
5.2. Anti-Inflammatory Effect
Inflammation is a multifaceted biological response of the body to external stimuli or injury, involving the orchestrated activity of immune cells [90], blood vessels [91], and molecular mediators [92]. It is intricately linked to cell death and significantly influences the development and advancement of prevalent diseases such as cancer [93,94]. Consequently, the development of safe and potent anti-inflammatory medications is crucial. Presently, extensive research is concentrating on the anti-inflammatory properties of individual natural compounds. These compounds exhibit anti-inflammatory actions by inhibiting LOX, suppressing NF-κB or MAPK signaling pathways, and decreasing cytokines like TNF-α and IL-6. Nonetheless, their efficacy is frequently confined to specific targets or models, lacking a systematic, comprehensive regulation of the inflammatory network. To overcome this limitation, Traditional Chinese Medicine, known for its multi-component synergistic effects, presents a distinctive opportunity. As a case in point, AR emerges as a focal point of research, where its diverse array of compounds collectively establish a highly efficient anti-inflammatory network.
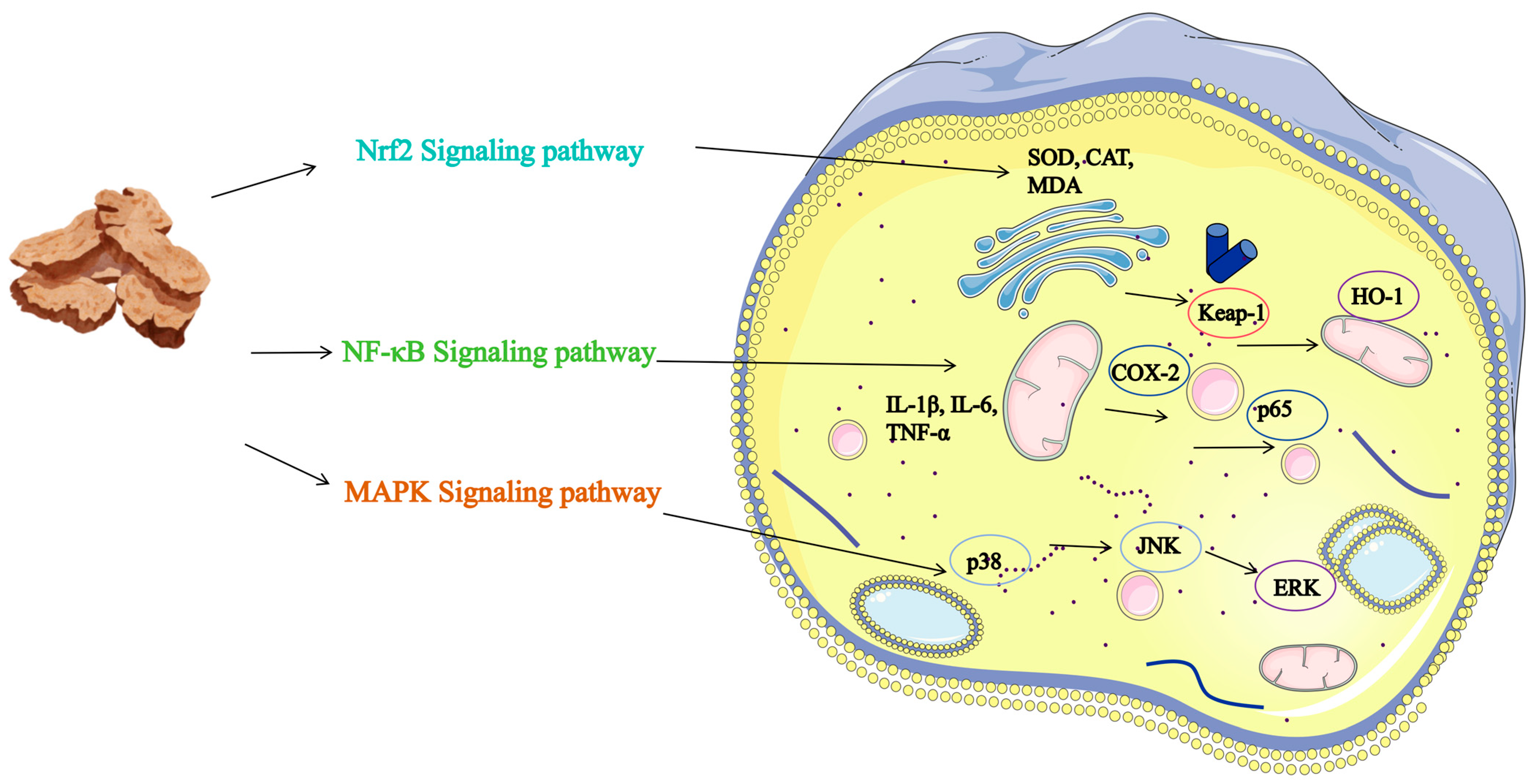
The anti-inflammatory effects of AR arise from the synergistic integration of its diverse active constituents (refer to Table S4 and Figure 5). For example, the organic acid component, cryptochlorogenic acid (compound 245), has been shown to effectively inhibit the expression of COX-2 and iNOS, while also blocking the activation of the MAPK signaling pathway [95]. Additionally, the terpenoid component, Terpinen-4-ol (compound 131), significantly suppresses the production of pro-inflammatory cytokines, such as TNF-α and IL-6, in macrophages, through the regulation of the MAPK pathway [96]. These principal compounds, in conjunction with polysaccharides and other constituents, concurrently target multiple critical nodes within the inflammatory response. They achieve this by reducing the release of pro-inflammatory factors via inhibition of the TLR4/NF-κB pathway, mitigating oxidative stress through activation of the Nrf2/HO-1 pathway, and modulating interactions between gut microbiota and immune cells. This results in multidimensional regulation of both mucosal immunity and systemic inflammation. This multi-targeted, multi-pathway action model, supported by multiple specific compounds, provides greater regulatory flexibility and systemic stability compared to single-component approaches when addressing complex, chronic inflammatory states.
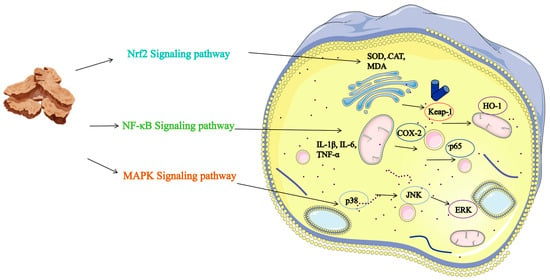
Figure 5.
Schematic Diagram of the Anti-inflammatory Mechanism of Atractylodis Rhizoma. Note: The figure illustrates the specific mechanisms of the Nrf2, NF-κB, and MAPK signaling pathways, distinguished by color. The various targets are labeled in the figure, and arrows indicate direct interactions with those targets or pathways.
5.3. Antioxidant Effect
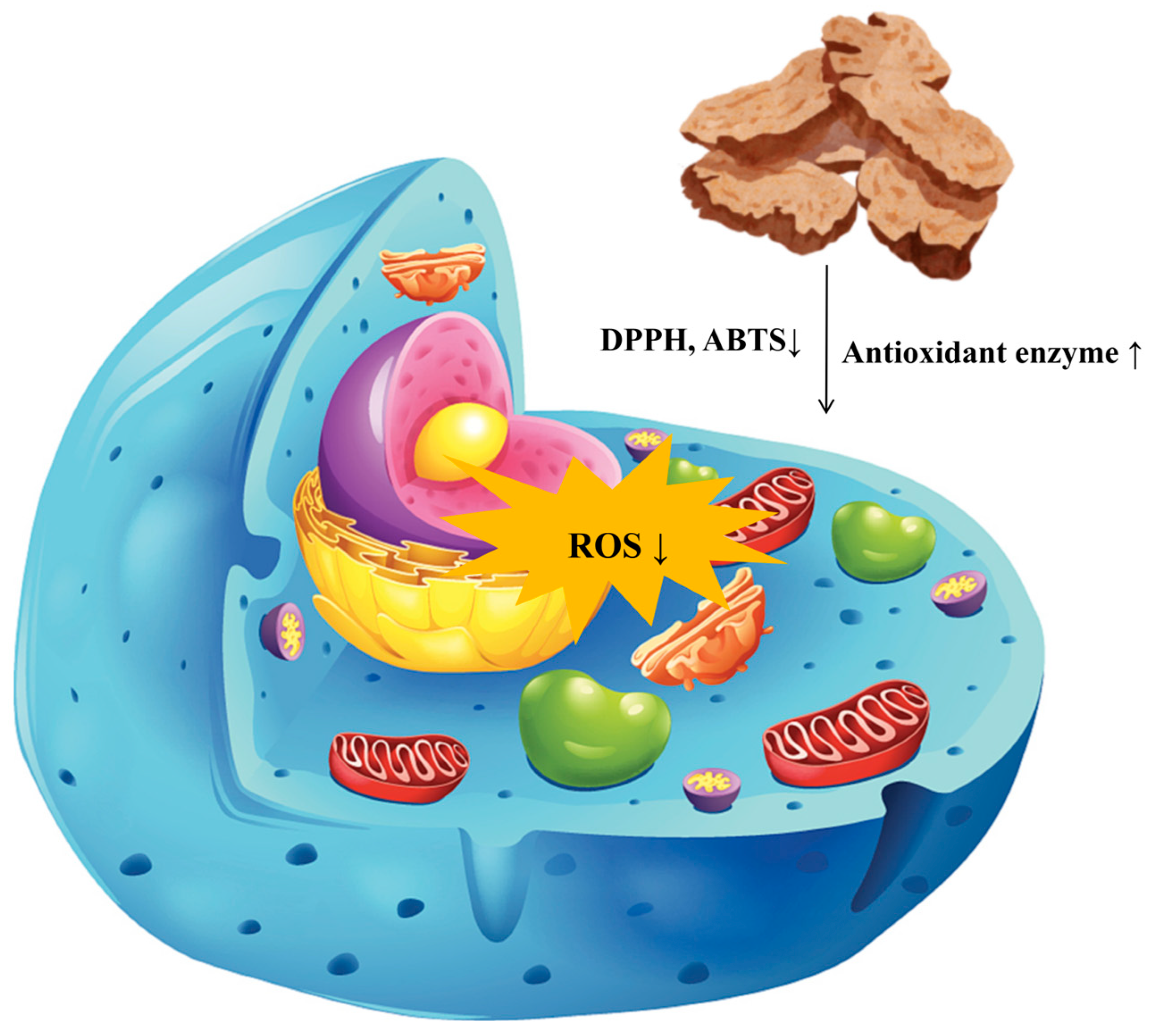
Oxidative stress refers to a condition in living organisms characterized by an imbalance between the production and elimination of reactive oxygen species (ROS), leading to cellular damage [97]. This phenomenon is closely associated with the onset and progression of various diseases, including cardiovascular diseases [98], neurodegenerative disorders [99], and cancer. Therefore, the identification of effective antioxidants is of critical importance. Current research on natural product antioxidants primarily focuses on their ability to directly scavenge free radicals, such as DPPH and ABTS, or to provide reducing equivalents, as evaluated by FRAP and CUPRAC assays [100]. As demonstrated in Table S5 and Figure 6, certain terpenoid and phenolic derivatives exhibit significant activity in chemical models, with effects comparable to those of ascorbic acid. However, their mechanisms of action are predominantly confined to the downstream stage of direct neutralization. Furthermore, their efficacy and stability within complex biological systems are often compromised by low concentrations and rapid metabolism. In contrast, the sesquiterpene lactone components characteristic of AR, which constitute the primary focus of this study, demonstrate a more profound and physiologically relevant mechanism of action. Experimental evidence substantiates that atractylenolactam (258) significantly activates the intracellular Nrf2 antioxidant signaling pathway. Nrf2 serves as a pivotal transcription factor governing cellular defenses against oxidative stress; its activation systematically induces the expression of various phase II detoxification enzymes, such as heme oxygenase-1 (HO-1) and nicotinamide adenine dinucleotide phosphate quinone dehydrogenase 1 (NQO1), alongside endogenous antioxidant enzymes [101]. This finding suggests that the antioxidant effects of AR transcend mere passive radical scavenging, actively augmenting the cells’ intrinsic defense mechanisms. Consequently, this provides a more robust molecular foundation for achieving sustained, broad-spectrum protection against oxidative damage, thereby demonstrating significant advantages, particularly in addressing persistent oxidative stress associated with chronic diseases.
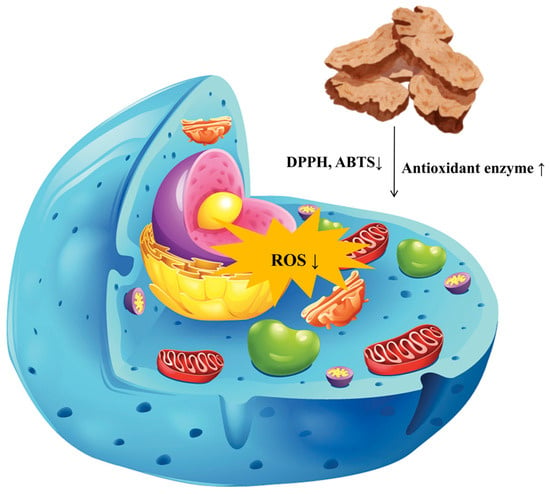
Figure 6.
Schematic Diagram of the Antioxidant Mechanism of Atractylodis Rhizoma. Note: This figure illustrates the antioxidant effects of AR. The large arrows indicate where AR acts on cells; ↑ represents an increase, and ↓ represents a decrease. Additionally, the differently colored elements represent organelles affected by oxidative stress, such as mitochondria and the endoplasmic reticulum.
Furthermore, the antioxidant properties of AR are significantly augmented by the synergistic interactions among its diverse constituents. The data presented in the Table S5 suggest that while the activity of individual compounds may be limited, natural essential oils or extracts frequently demonstrate enhanced overall activity due to the synergistic interactions among their components. AR is abundant in various sesquiterpene lactones, such as atractylenolide I (271), II (268), and III (251), in addition to polyacetylene compounds, such as atractylodin (141). These constituents may engage in network pharmacologic effects through distinct molecular targets and mechanisms of action, resulting in synergistic or additive antioxidant effects. This complex natural defense system, comprising multiple components, poses a greater challenge for the body to compensate for or circumvent compared to a single compound, thereby potentially providing more stable and comprehensive protection. Consequently, AR functions not only as a source of highly active lead compounds but also as a standardized extract. As a natural preparation capable of multi-targeted, systemic regulation of endogenous antioxidant pathways, it exhibits unique potential and broad application prospects in the development of herbal medicines and functional foods aimed at the prevention or adjunctive treatment of oxidative stress-related diseases.
5.4. Hepatoprotective Effect
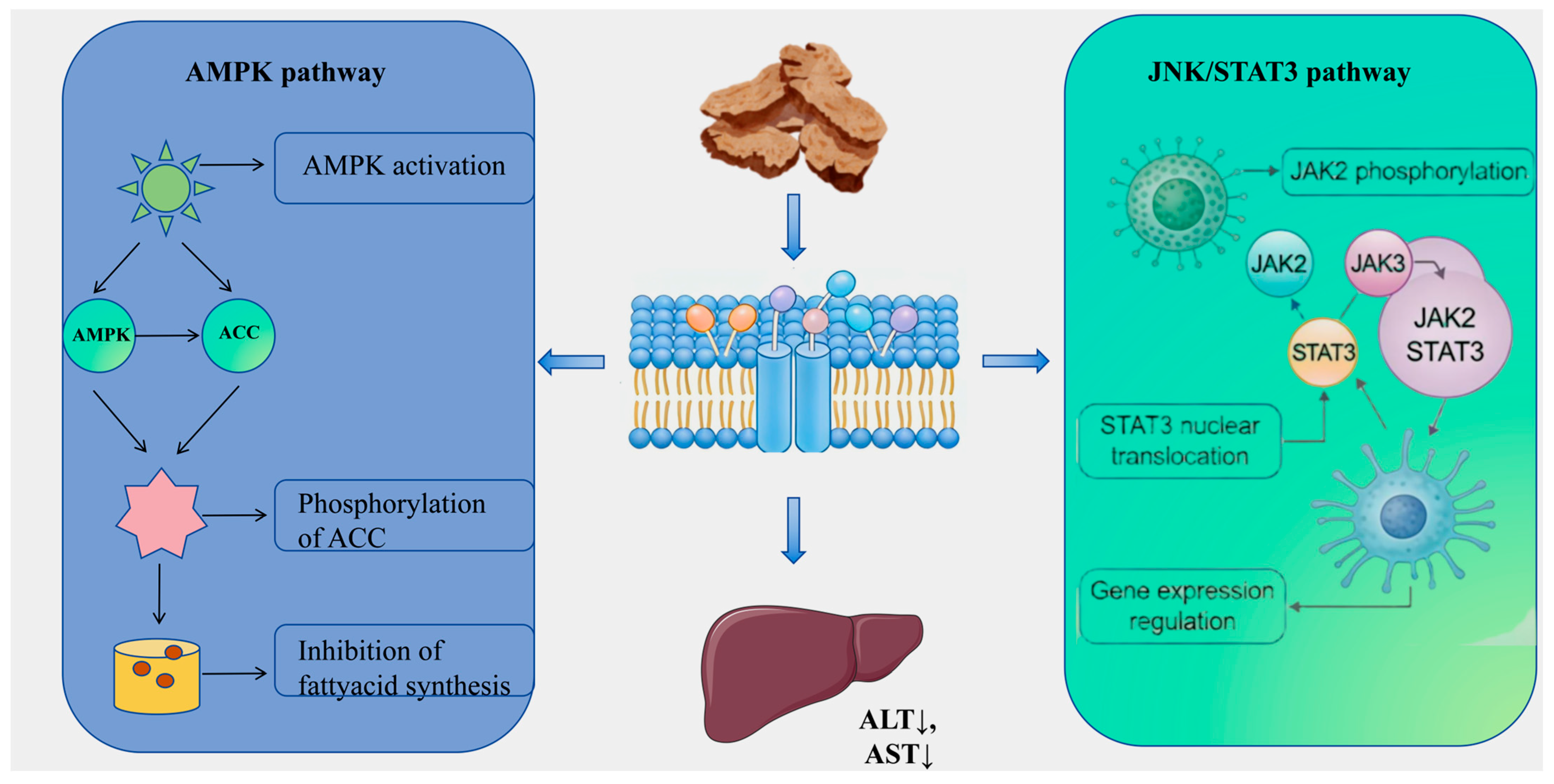
Liver injury, characterized as a complex pathological process arising from various etiological factors, has historically posed significant challenges in pharmacological research, particularly in the formulation of effective prevention and treatment strategies. Conventional single-target interventions frequently prove inadequate in addressing the extensive array of pathways implicated in the initiation and progression of liver injury, which include oxidative stress, lipid metabolism disorders, inflammatory responses, and fibrosis [102]. Within this framework, AR and its diverse bioactive constituents demonstrate considerable hepatoprotective potential and systemic intervention benefits, attributable to their multi-component and multi-target action properties. Emerging research [103] suggests that the hepatoprotective effects of AR are achieved through synergistic regulation across various stages and pathological dimensions of liver injury (refer to Table S6 and Figure 7). In the initial phase of injury, bioactive compounds such as atractylodin (141) and neochlorogenic acid (240) effectively inhibit fatty acid synthesis and promote its oxidation through activation of the AMPK pathway and upregulation of PPARα/CPT-1 expression [104,105]. This modulation of lipid metabolism ameliorates metabolic disorders and addresses the underlying pathology of non-alcoholic fatty liver disease. Simultaneously, compounds such as eucalyptol (115) attenuate drug- or toxin-induced oxidative stress-related liver damage by significantly enhancing the activity of endogenous antioxidant enzymes, including glutathione (GSH) and superoxide dismutase (SOD), while reducing oxidative damage markers such as malondialdehyde (MDA) and 8-hydroxy-2′-deoxyguanosine (8-OHDG) [106]. Moreover, derivatives of AR demonstrate significant potential in mitigating the malignant progression associated with liver damage. Notably, Senkyunolide A (294) alleviates cholestatic liver fibrosis through the modulation of endoplasmic reticulum autophagy, while Germacrone impedes the JAK2/STAT3 signaling pathway, thereby inhibiting hepatic stellate cell activation and collagen deposition [107]. Furthermore, the biomolecules such as Atractylodin (141) and Atractylodes polysaccharide present in AR have been shown to exhibit substantial hepatoprotective properties [108]. This broad spectrum of actions, which includes metabolic regulation, antioxidant defense, and anti-fibrotic effects, provides comprehensive protection against the progressive pathological trajectory of liver injury.
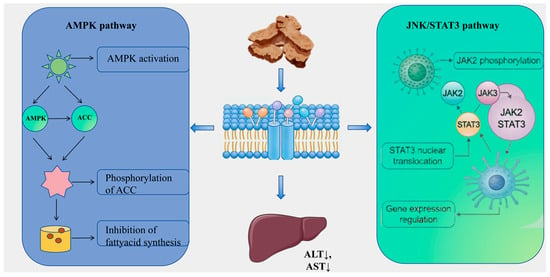
Figure 7.
Schematic Diagram of the Hepatoprotective Mechanism of Atractylodis Rhizoma. Note: ↑ represents an increase, and ↓ represents a decrease.
5.5. Anti-Cancer Effect
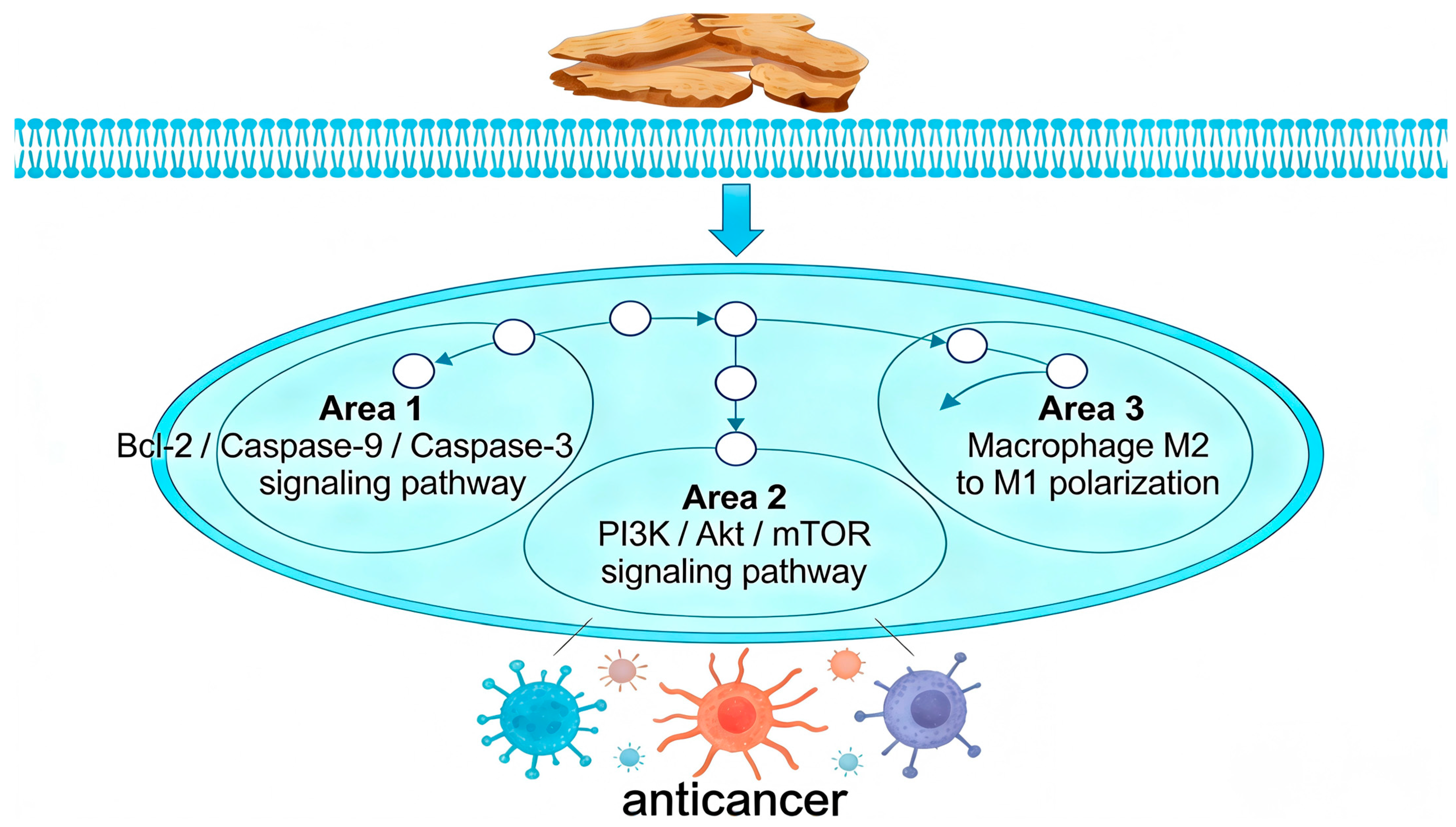
This study provides a comprehensive review of the diverse bioactive compounds found in AR, with a specific focus on their anticancer properties as detailed in Table S7 and Figure 8. Among these compounds, sesquiterpene lactones, including atractylenolide II (268), atractylenolide I (271), and guaiol (191), are identified as the primary active constituents. These compounds demonstrate significant efficacy in modulating key signaling pathways, such as PI3K/Akt/mTOR, STAT3, and ERK. Furthermore, volatile oils and terpenoid components, such as β-eudesmol (71) and δ-elemene (319), play a crucial role in directly inducing tumor cell apoptosis and cell cycle arrest. Phenolic acid derivatives such as Chlorogenic acid (242) further augment these effects by modulating oxidative stress and immune responses. The diversity of these components does not operate in isolation; rather, they collectively contribute to the comprehensive capacity of AR to intervene in tumor biology through multiple targets and at various levels.
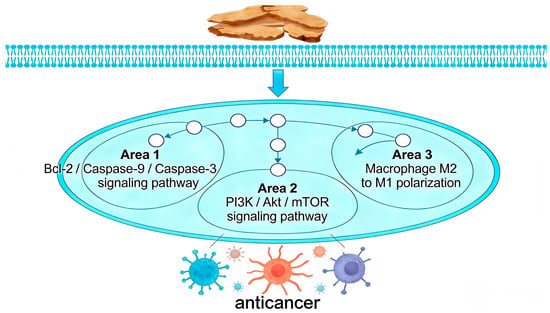
Figure 8.
Schematic Diagram of the Anti-cancer Mechanism of Atractylodis Rhizoma. Note: The arrows indicate how AR exerts its anticancer effects through apoptosis, cell proliferation, and macrophage polarization, respectively. The tumor cells of different colors represent the outcomes resulting from these three mechanisms, including apoptosis, growth inhibition, and phagocytosis.
The notable anti-cancer properties of AR are attributed to its extensive regulation of both the tumor microenvironment and systemic physiology, extending beyond mere direct cytotoxic effects. The primary component, atractylenolide II, exemplifies this characteristic by modulating the immune microenvironment across various cancer types, specifically through the inhibition of M2 macrophage polarization and the downregulation of PD-L1. Additionally, it participates in emerging biological processes such as glycolytic metabolism and the induction of ferroptosis [109,110]. This complex mechanism of a single molecule suggests that its target may be located at an upstream node in the bidirectional regulation between tumor cells and the microenvironment. Furthermore, the therapeutic potential of AR also addresses the systemic manifestations of tumor diseases. For instance, atractylenolide I alleviates cancer cachexia-related muscle wasting by inhibiting the STAT3 pathway [111]. This aligns with the principles of Traditional Chinese Medicine, which focus on fortifying the spleen, enhancing qi, and bolstering the body’s vital energy. It embodies a unique holistic therapeutic approach that concurrently eliminates pathogens and strengthens the body’s defenses.
5.6. Anti-Diabetic Effect
The anti-diabetic potential of AR is notably significant due to its direct hypoglycemic effects, which influence essential pathways of glucose metabolism through synergistic multi-component mechanisms (Table 3). Recent studies indicate that its active constituents can modulate blood glucose regulation through multiple targets. For example, molecular docking analyses have confirmed that γ-cadinene (42), a sesquiterpene component abundant in its essential oil, exhibits a strong binding affinity with the insulin receptor (INSR). This observation suggests that γ-cadinene (42) may mimic or enhance insulin signaling, thereby directly mitigating insulin resistance, which is a critical mechanism for lowering blood glucose levels [112]. Simultaneously, the sesquiterpene constituents of AR essential oil, such as eucalyptol (115) and compounds structurally similar to valeranone (60), may enhance endogenous insulin secretion and utilization by protecting pancreatic beta cells and inhibiting dipeptidyl peptidase-IV (DPP-IV) enzyme activity, respectively [113,114]. Collectively, these mechanisms fortify the insulin axis. Additionally, the extract’s potential α-glucosidase inhibitory activity may directly impede carbohydrate absorption in the intestine, thereby enabling rapid management of postprandial blood glucose surges. This simultaneous intervention across three pivotal pathways—insulin sensitivity, insulin secretion, and postprandial blood glucose regulation—constitutes the primary advantage of AR’s direct hypoglycemic effect.

Table 3.
Anti-diabetic effect of Atractylodis Rhizoma.
In a more comprehensive context, the hypoglycemic properties of AR are intricately linked to both the prevention and treatment of diabetic complications, thereby demonstrating dual benefits in glucose metabolism regulation and tissue protection. Specific constituents, such as eucalyptol (115), play a crucial role in mitigating retinal pigment epithelial barrier dysfunction in diabetic models by downregulating matrix metalloproteinases (MMPs) and reducing apoptosis and oxidative stress, as evidenced by decreased reactive oxygen species (ROS). This underscores its unique value in preventing diabetic retinopathy [114]. Additionally, phenolic acid components, such as cryptochlorogenic acid (245), may inhibit ferroptosis in pancreatic β-cells by activating the Nrf2/GPX4 pathway, thereby providing sustained protection for pancreatic function [119]. This synergistic effect, which includes the concurrent reduction in blood glucose levels along with antioxidant and anti-inflammatory benefits, as well as the protection of microvasculature and vital organs, elevates AR beyond the role of a mere hypoglycemic agent.
5.7. Intestinal Regulatory Function
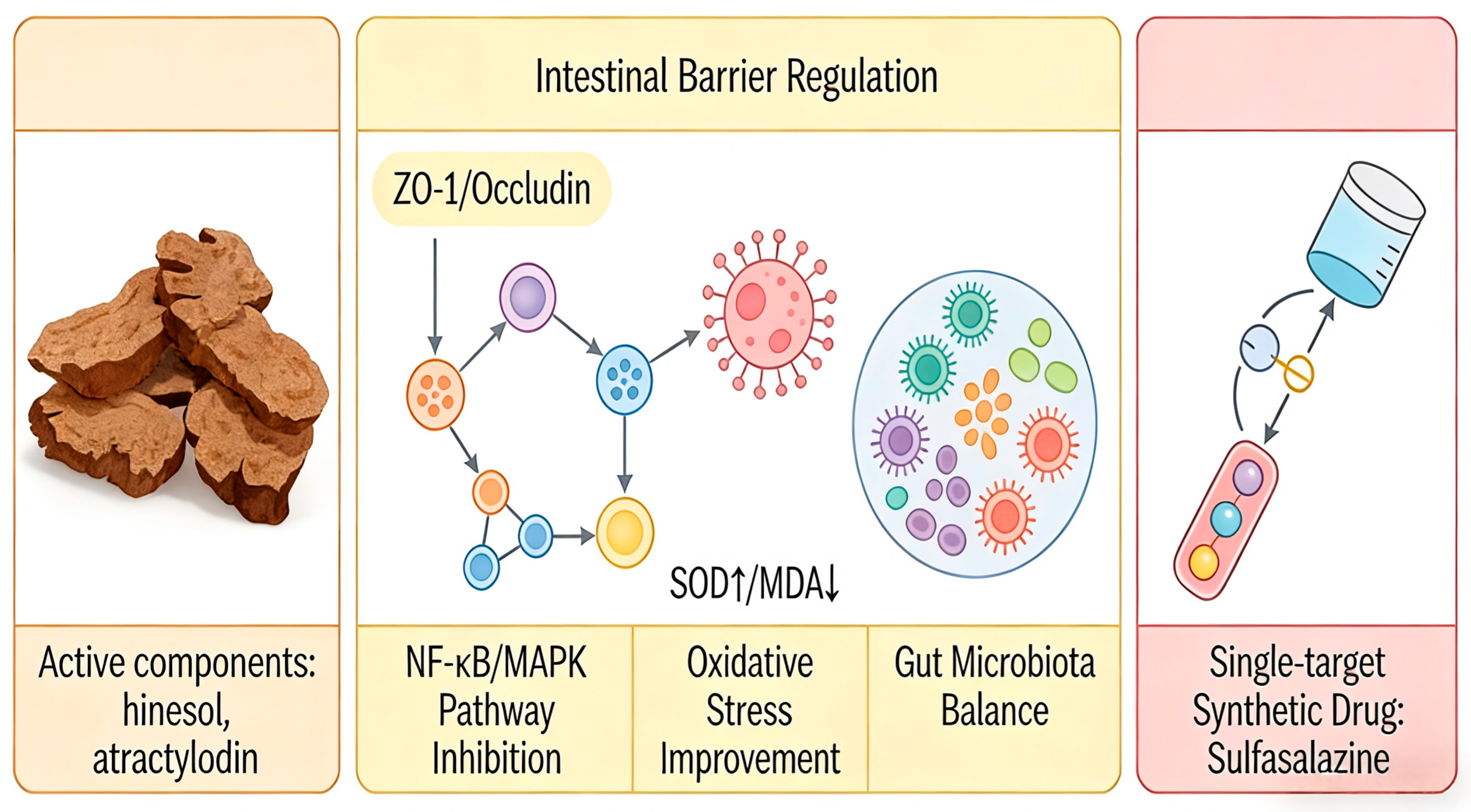
Recent research on ulcerative colitis (UC) and associated intestinal inflammation has increasingly concentrated on natural products, owing to their multi-target effects and favorable safety profiles [121]. Empirical evidence indicates that AR contains bioactive compounds such as sesquiterpenes, polyacetylenes, and phenolic acids, which have demonstrated efficacy in preclinical colitis models (refer to Table S8 and Figure 9). These compounds collectively reduce the disease activity index (DAI) and enhance the integrity of the intestinal barrier, as evidenced by the upregulation of tight junction proteins, including ZO-1, occludin, claudin-1, and MUC2 [122]. Furthermore, they inhibit key pro-inflammatory signaling pathways, notably NF-κB and MAPK. In addition, these compounds modulate oxidative stress markers by elevating levels of SOD, GSH-Px, and CAT, while decreasing MDA levels, and they contribute to the restoration of gut microbiota balance [123]. This multifaceted mechanism of action stands in contrast to the single-target approach of synthetic drugs such as sulfasalazine or mesalazine.
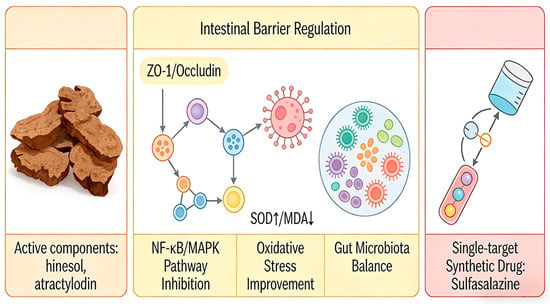
Figure 9.
Schematic Diagram of the Intestinal regulatory Mechanism of Atractylodis Rhizoma. Note: Light orange modules represent AR; light yellow modules indicate that AR acts through four mechanisms simultaneously, with different arrows denoting multi-pathway action; light red modules indicate that synthetic drugs have only one arrow, representing a single target. Additionally, the different colors in the central region represent the interactions among the intestinal barrier, the gut microbiota, and antioxidant enzymes, illustrating AR’s multidimensional regulatory capabilities.
The primary benefit of AR lies in its synergistic and multi-dimensional strategy for addressing intestinal inflammation. Unlike monotherapies that typically target singular pathways, the phytochemical constituents of AR concurrently tackle multiple pathological aspects of ulcerative colitis, such as barrier dysfunction, immune dysregulation, oxidative damage, and microbial dysbiosis. For instance, atractylodin (141) not only inhibits the activation of NF-κB and MAPK pathways but also fosters the growth of beneficial gut microbiota and influences metabolic regulation through GAPDH malonylation [122]. Similarly, hinesol (66) and atractylenolide III (251) collaboratively enhance tight junction integrity while downregulating various cytokines and chemokines [123,124]. This multi-target approach may enhance therapeutic efficacy and reduce the risk of compensatory resistance, offering a treatment strategy that aligns with the principles of “network pharmacology” inherent in herbal medicine.
5.8. Neuroprotective Effect
Research on AR within the realm of neuroprotection is transitioning its role from a traditional medicinal herb to a comprehensive natural compound library with well-defined multi-target mechanisms. Unlike the numerous single compounds documented in the literature, the significance of AR lies in its diverse array of bioactive constituents, including polyacetylenes and sesquiterpene lactones, which collectively form a more extensive neuroprotective network (see Table 4). For instance, its primary component, atractylodin (141), demonstrates anti-inflammatory and antioxidant properties [103]; atractylenolide III (251) modulates astrocyte function [125]; and atractylenolactam (258) suppresses microglial activation [126]. Together, these elements collectively form a functionally synergistic composite system, whose combined effects exceed those of the individual compounds listed in the table. For instance, Linalool primarily addresses oxidative stress, β-Phellandrene specifically inhibits acetylcholinesterase (AChE), and Parthenolide selectively modulates certain inflammatory pathways. This inherent multi-component, multi-pathway synergistic capability confers upon AR an augmented potential for systematic intervention in complex pathologies characterized by multiple interrelated factors, such as neurodegenerative diseases.
To fully capitalize on this advantage, it is essential for research to make substantial advancements across several domains. Mechanistic investigations should transition from a focus on individual pathways to employing computational pharmacology and multi-omics approaches. This will facilitate a comprehensive understanding of how atractylodin (141), atractylenolides, and other critical components of AR interact to modulate the “neuroinflammation-oxidative stress-pyroptosis-autophagy” network. In the realm of formulation development, insights should be drawn from the successful implementation of citronellyl acetate nanoparticles and rutin-AuNPs. Given the characteristics of the active constituents, it is crucial to design innovative nanoscale systems capable of co-loading multiple components to improve brain delivery, thereby overcoming challenges related to bioavailability and targeted delivery [127,128]. Furthermore, validation models for therapeutic efficacy need to evolve beyond single-injury paradigms to establish animal models that more accurately represent the complexity of clinical diseases and the pathomechanism of spleen deficiency with dampness obstruction, as conceptualized in Traditional Chinese Medicine. These models should integrate variables such as aging, metabolic abnormalities, and neuroinflammation.

Table 4.
Neuroprotective effect of Atractylodis Rhizoma.
Table 4.
Neuroprotective effect of Atractylodis Rhizoma.
| Bioactivity | Compounds/Extracts | Testing Subjects (Animal/Model) | Dose | Positive Control | Results/Mechanism | References |
|---|---|---|---|---|---|---|
| Anti-anxiety | Dehydrofukinone (75) | Swiss mice | 10, 22, 30, 100 mg/kg | Diazepam | GABAₐ receptor activity ↑, Chloride ion influx ↑, Voltage-dependent calcium channel activity ↓, Free calcium concentration ↓ | [129] |
| Neuroprotective effect | Isoaromadendrene epoxide (76) from essential oil | In vitro: PC12 and BV2 cells; In vivo: C57BL/6 mice | In vitro: 0.1–10 µg/mL; In vivo: 10–20 mg/kg | Memantine | IL-1β ↓, IL-6 ↓, TNF-α ↓, GABA ↑, 5-HT ↑, NE ↑ | [130] |
| Neuroprotective effect | Atractylodin (141) | C57BL/6J mice | 20 mg/kg and 40 mg/kg | Not mentioned | TNF-α ↓, IL-6 ↓, IL-1β ↓, NLRP3 ↓, NF-κB ↓, BDNF ↑, Akt ↑, MDA ↓, SOD ↑, GSH-Px ↑, GSH ↑ | [103] |
| Neuroprotective effect | Eucalyptol (115) | Wistar rats | 100 mg/kg/d | Not mentioned | MDA ↓, GSH ↑, SOD ↑, GPx ↑, TNF-α ↓, IL-1β ↓, IL-6 ↓, IL-10 ↑, SIRT1 ↑, NF-κB ↓, BDNF ↑, | [131] |
| Neuroprotective effect | Linalool (117) | PC12 Cell | 1–100 µmol/L | Not mentioned | ROS ↓, MDA ↓, DNA ↓, Bax/Bcl-2 ↓, caspase-3 ↓, caspase-9 ↓ | [132] |
| Neuroprotective effect | β-Phellandrene (120) | Enzyme inhibition assay in vitro | 300 µg/mL | Not mentioned | AChE ↓ | [133] |
| Anti-aggregation and disaggregation effects of Aβ amyloid protein | Citronellyl acetate (125) loaded onto CaCO3 nanoparticles | In vitro simulation | 100 µg/mL | BHT, Donepezil, Galantamine | AChE ↓, Aβ aggregation ↓ | [134] |
| Anti-aggregation and disaggregation effects of Aβ amyloid protein | Citronellyl acetate (125) loaded onto hydroxyapatite nanocarriers | In vitro simulation | 100 µg/mL | BHT, Donepezil, Galantamine | AChE ↓, Aβ aggregation ↓ | [127] |
| Neuroprotective effect | (-)-Bornyl acetate (126) | C57BL/6 mice | 100, 200, 400 mg/kg | Not mentioned | CD11b+/CD45+ Cell ↓, IL-1β ↓, IL-6 ↓, TNF-α ↓, iNOS ↓, COX-2 ↓, MCP-1 ↓, MIP-1α ↓, RANTES ↓, p38 ↓, ERK ↓, NF-κB ↓, ZO-1 ↑, Occludin ↑, PECAM-1 ↓, MMP-9 ↓, ICAM-1 ↓ | [135] |
| Anti-neuroinflammatory effect | Carveol (146) | SD rats | 5, 10, 20 mL/kg | Not mentioned | Alleviate thermal pain sensitivity and improve motor coordination, GSH ↑, GST ↑, iNOS ↓, LPO ↓, NF-κB ↓, COX-2 ↓, TNF-α ↓ | [136] |
| Alleviating neuroinflammation and pain in diabetes | Carveol (146) | SD rats | 5, 10, 20 mL/kg | Pregabalin | GSH ↑, GST ↑, LPO ↓, NO ↓, COX-2 ↓, TNF-α ↓, NF-κB ↓ | [137] |
| Improving memory impairment and neuroinflammation | Carveol (146) | SD rats | 50, 100, 200 mg/kg | Donepezil | GSH ↑, GST ↑, CAT ↑, LPO ↓, Aβ ↓, NF-κB ↓, TNF-α ↓, IL-18 ↓, PGE2 ↓ | [138] |
| Improving Parkinson’s disease | Carveol (146) | Albino mice | 50 mg/kg | Not mentioned | ROS ↓, LPO ↓, Nrf2 ↑,HO-1 ↑, NLRP3 ↓ | [139] |
| Improving Alzheimer’s disease | Quinic acid (235) | SH-SY5Y cell | 50–200 µM | Donepezil | ROS ↓, Aβ ↓, phosphorylated tau ↓, MAPK ↓ | [140] |
| Promoting the proliferation and differentiation of neural stem cells | Quinic acid (235) | Hippocampal cell | 50, 100, 200 µM | Not mentioned | Mash1 ↑, Ngn2 ↑, Notch1 ↑, Hes1 ↑ | [141] |
| Improving Alzheimer’s disease | Quinic acid (235) | C57BL/6 mice | 30 mg/kg/d | Not mentioned | IAA ↑, KYNA ↑, DR3 ↓, IKK ↓, NF-κB ↓, Aβ42 ↓, p-Tau ↓ | [142] |
| Improving Alzheimer’s disease | Corogenic acid (242) | C57 mice | 2 mg/kg | Not mentioned | Aβ ↓, BACE1 ↓, TNF-α ↓, IL-1β ↓, IL-6 ↓ | [143] |
| Neuroprotective effect | Rutin (250)-AuNPs | In vitro: SH-SY5Y cells, In vivo: ICR mice | 20–100 µg/mL | Not mentioned | Nrf2 ↑, ARE ↑, ROS ↓ | [128] |
| Improving Alzheimer’s disease | Atractylenolide III (251) | In vitro: C8-D1A astrocytes, In vivo: C57BL/6 mice | In vitro: 100 µM; In vivo: 2.4 mg/kg | Not mentioned | AQP4 ↑ | [125] |
| Neuroprotective effect | Atractylenolactam (258) | In vitro: Bv2 microglia, HT22 neuron cells | 50 µM | Not mentioned | TNF-α ↓, IL-6 ↓, IL-1β ↓ | [126] |
| Antidepressant effect | α-cyperone (273) | In vivo: Wistar rats | 5, 10 mg/kg | Not mentioned | IL-1β ↓, TNF-α ↓, p65 phosphorylation ↓ | [144] |
| Improving Parkinson’s disease | α-cyperone (273) | Wistar rats | 10 mg/kg | Not mentioned | Nrf2 ↑, HO-1 ↑, NF-κB ↓ | [145] |
| Alleviating brain ischaemic damage | Parthenolide (287) | In vivo: Sprague Dawley rats; In vitro: The human microglial clone 3 cell | In vivo: 0.5 mg/kg, 1 mg/kg; In vitro: 2 µM | Gination | RhoA ↓, ROCK ↓, NF-κB ↓ | [146] |
| Improving neurological function following traumatic brain injury | Parthenolide (287) | In vivo: C57BL/6 mice; In vitro: Bv2 microglia, HT22 neuron cells | In vivo: 1 mg/kg; In vitro: 0.5–5 µM | Not mentioned | STAT3 ↓, NF-κB ↓, NLRP1 ↑, NLRP3 ↑, NLRC4 ↑ | [147] |
| Improve cognitive impairment | leamide (310) from Ethanolic Extract of Rosa rugosa Roots | ICR mice | 10, 20, 40 mg/kg | Not mentioned | MDA ↓, ADAM10 ↑ | [148] |
Note: ↑ represents an increase, and ↓ represents a decrease.
5.9. Cardiac-Protective Effect
AR, a traditional medicinal plant, has been shown in recent studies to contain a diverse array of bioactive compounds with cardioprotective potential. These compounds exhibit synergistic effects through multiple targets and pathways, highlighting their significant value in the prevention and treatment of cardiovascular diseases (see Table 5). The compounds listed in this table, including camphene (121), cryptochlorogenic acid (245), atractylenolide II (268), valerenic acid (288), and valencene (327), can primarily be isolated directly from this plant. These constituents enhance antioxidant capacity in animal and cellular models by increasing the ratios of superoxide dismutase (SOD), catalase (CAT), and glutathione/glutathione disulfide (GSH/GSSG). They also suppress oxidative stress and inflammatory pathways by downregulating nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6). Furthermore, they regulate myocardial energy metabolism by activating peroxisome proliferator-activated receptor alpha (PPARα) and promoting fatty acid oxidation [149]. Additionally, these compounds decrease levels of TGF-β1 and collagen I, effectively inhibiting myocardial fibrosis. This action results in anti-hypertrophic effects, enhanced cardiac function, and reduced myocardial damage.
In contrast to existing studies that predominantly focus on individual components or pathways, the primary advantage of AR lies in its comprehensive regulation through multiple components, targets, and pathways. While previous research often aims at specific pathological pathways using synthetic drugs or isolated plant components, the diverse constituents of AR—such as terpenoids, phenylpropanoids, and volatile oils—exert synergistic or cumulative effects on various pathological processes, including myocardial hypertrophy, fibrosis, oxidative damage, and energy metabolism disorders. For example, atractylenolide II and valerenic acid work in concert to alleviate myocardial remodeling and metabolic dysfunction by inhibiting the TGF-β1/Smad pathway and activating the PPARα pathway, respectively [150,151]. Simultaneously, camphene (121) and valencene (327) synergistically enhance the endogenous antioxidant system, thereby mitigating myocardial oxidative damage resulting from ischemia–reperfusion injury or hypertension [149,152]. This multi-tiered, networked mechanism of action aligns with the principles of combination therapy and systemic regulation emphasized in modern cardiovascular disease treatment. Moreover, it endows AR with a unique potential for the prevention and management of complex cardiac conditions, such as metabolic cardiomyopathy.

Table 5.
Cardiac-protective effect of Atractylodis Rhizoma.
Table 5.
Cardiac-protective effect of Atractylodis Rhizoma.
| Bioactivity | Compounds/Extracts | Testing Subjects (Animal/Model) | Dose | Positive Control | Results/Mechanism | References |
|---|---|---|---|---|---|---|
| Cardio-protective effect | Camphene (121) | Wistar rats | 30 µg/g | Not mentioned | LDH ↓, GSH/GSSG ↑; CS ↑; CAT ↑, Mn-SOD ↑, GR ↑; GPx4 ↓ | [152] |
| Improvement of myocardial hypertrophy | Cryptochlorogenin acid (245) | In vivo: SD rats, In vitro: H9c2 cell | In vivo: 200 mg/kg; In vitro: 10, 25, 50 µM | Propranolol | Akt ↓, mTOR ↓, HIF-1α ↓; ANP ↓, BNP ↓ | [151] |
| Improvement of cardiac function | Atractylenolide II (268) | Spontaneous hypertension rats and Wistar Kyoto rats | 10, 30, 60 mg/kg/day | Not mentioned | LVEF ↑, LVMI ↓; Collagen I ↓, α-SMA ↓, Fibronectin ↓; TGF-β1 ↓; SOD ↑, GSH-Px ↑; H2O2 ↓, MDA ↓, NOX ↓ | [150] |
| Anti-hypertrophic effect on the myocardium | Valerenic acid (288) | In vivo: ICR mice; In vitro: H9C2 cardiac myocytes | In vivo: 0.5 mg/kg, 2 mg/kg; In vitro: 1, 5, 10 µM | Fenofibrate | PPARα ↑; IL-6 ↓, IL-1β ↓, TNF-α ↓ | [153] |
| Cardiac protection | Valencene (327) | Wistar rats | 12 mg/kg/day | Not mentioned | cTn-I ↓, Myoglobin ↓; SOD ↑, CAT ↑, GPx ↑; hs-CRP ↓; Homocysteine ↓, NF-κB ↓, TNF-α ↓, IL-1β ↓, IL-6 ↓, IL-10 ↑ | [149] |
Note: ↑ represents an increase, and ↓ represents a decrease.
5.10. Joint Repair Function
Atractylodes, a genus of medicinal plants, is extensively employed in traditional medicine to alleviate dampness obstruction in the middle jiao and rheumatic arthralgia [12]. Notably, AR stands out among its species, as contemporary pharmacological research has underscored its substantial potential in the treatment of arthritis [154]. In contrast to chemically synthesized drugs, which generally target a singular pathway, the anti-arthritic effects of AR are derived from its naturally occurring synergistic pharmacodynamic complex, comprising multiple components. As demonstrated in Table 6, its primary active constituents—such as atractylenolide, eucalyptol, borneol acetate, carvone, geraniol, and ligustilide—exhibit multidimensional intervention in the intricate pathological processes of arthritis through an interconnected network mechanism, as evidenced in both animal and cellular models.
The principal benefit of this approach lies in its systematic regulation of the pathological network encompassing “inflammation-immunity-oxidation-degradation” in arthritis. This method effectively synergizes the inhibition of central inflammatory signaling pathways, such as NF-κB and MAPK, resulting in a comprehensive downregulation of key pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β [155]. Simultaneously, it modulates macrophage polarization, thereby exerting substantial control over the immune-inflammatory network. Additionally, it provides concurrent protection to joint tissue structures. For instance, geraniol (187) mitigates oxidative damage by activating antioxidant pathways, such as Keap1/Nrf2/HO-1 [156]. It also enhances the expression of type II collagen and aggregated proteoglycans while inhibiting MMP13 and ADAMTS-5.

Table 6.
Joint repair function of Atractylodis Rhizoma.
Table 6.
Joint repair function of Atractylodis Rhizoma.
| Bioactivity | Compounds/Extracts | Testing Subjects (Animal/Model) | Dose | Positive Control | Results/Mechanism | References |
|---|---|---|---|---|---|---|
| Joint repair function | Atractylodin (141) | In vitro: BM-DCs cell; In vivo: DBA/1 mice | In vitro: 12.5, 25, 50, 100 µM; In vivo: 40 mg/kg | Quercetin | CD40 ↓, CD80 ↓, CD86 ↓; TNF-α ↓, IL-6 ↓, IL-1β ↓, IL-23 ↓, IL-12 ↓, IFN-γ ↓, NO ↓; p38 ↓, ERK ↓, NF-κB ↓ | [155] |
| Anti-gouty arthritis effect | Eucalyptol (115) | BALB/c mice | 30, 100, 300, 600 mg/kg | Indomethacin | NLRP3 ↓, IL-1β ↓, IL-6 ↓, TNF-α ↓, MPO ↓; ROS ↓, SOD ↑, GSH-Px ↑, MDA ↓; TRPV1 ↑, Nrf2 ↑, HO-1 ↑ | [157] |
| Joint repair function | (-)-Bornyl acetate (126) | DBA/1J mice | 20, 40 mg/kg | Methotrexate | CYP17A1 ↓, HSD17B3 ↓; TLR4 ↓, MAPK ↓, NF-κB ↓ | [158] |
| Joint protection function | Carveol (146) | In vitro: RAW264.7 Macrophages, In vivo: C57BL/6J mice | In vitro: 10, 25, 50 µg/mL; In vivo: 12.5 mg/kg, 25 mg/kg | Not mentioned | M1-type macrophages ↓, IL-1β ↓, IL-6 ↓, TNF-α ↓, iNOS ↓; M2-type macrophages ↑, ARG-1 ↑, CD206 ↑, MGL-1 ↑, MGL-2 ↑; HO-1 ↑, ROS ↓; COX-2 ↓, MMP13 ↓, ADAMTS-5 ↓ | [159] |
| Joint protection function | Geraniol (187) | In vitro: Mouse chondrocytes; In vivo: C57BL/6 mice | In vitro: 1 µM; In vivo: 1 mg/mL | Not mentioned | Col2a1 ↑, Aggrecan ↑; MMP13 ↓, ADAMTS-5 ↓; Keap1 ↓, Nrf2 ↑, HO-1 ↑; p-p65 ↓ | [156] |
| Joint repair function | Senkyunolide A (294) | In vitro: Mouse chondrocytes; In vivo: C57BL/6 mice | In vitro: 20, 40, 80 µg/mL; In vivo: 20, 40 mg/kg | Not mentioned | MMP13 ↓, ADAMTS4/5 ↓; IGF-1 ↑, Aggrecan ↑, Col2a1 ↑; TNF-α ↓, IL-6 ↓, IL-18 ↓; NLRP3 ↓, Caspase-1 ↓ | [160] |
Note: ↑ represents an increase, and ↓ represents a decrease.
5.11. Protective Effect Against Lung Injury
The protective effects of AR against pulmonary injury are attributed to its multi-component synergistic actions, which establish a complex, multi-level, and multi-target regulatory network (Table 7). The active ingredient data compiled in this study elucidate these properties: chlorogenic acid (242) primarily exerts antioxidant effects and regulates macrophage polarization; dehydrocostus lactone (256) inhibits the key glycolytic enzyme PFKFB3, thereby modulating inflammatory metabolism. Acetylatractylodinol (270), isolated from its ethanol extract, concurrently inhibits inflammation mediated by the PI3K/AKT pathway and uniquely alters the host metabolic profile [161]. This indicates that it not only mitigates pulmonary edema and inflammatory responses but also potentially aids lung tissue in fulfilling energy and repair requirements under injury-induced stress through metabolic reprogramming. This comprehensive intervention, targeting the three core pathological pathways—inflammation, oxidative stress, and metabolic disorders—enables its mechanism of action to surpass traditional strategies primarily centered on immunosuppression, such as those exemplified by dexamethasone.

Table 7.
Protective effect against lung injury of Atractylodis Rhizoma.
The metabolic regulatory function of acetylatractylodinol (270) is particularly noteworthy, offering substantial scientific value and potential for the development of AR in the treatment of lung injury. Contemporary therapeutic strategies for acute lung injury (ALI) are transitioning from exclusively anti-inflammatory approaches to those that prioritize the maintenance of tissue homeostasis, as metabolic dysregulation has been recognized as a pivotal factor in the progression of injury [165]. The bioactive constituents of AR such as dehydrocostus lactone (256) include compounds that both regulate inflammation and modulate metabolism, suggesting that the herb or its active fractions may influence more nuanced pathological processes, such as the repair of the alveolar epithelial barrier and the preservation of mitochondrial function. This discovery introduces a promising new paradigm for the development of lung protectants derived from natural products.
5.12. Other Effects
In addition to its previously discussed pharmacological effects, AR exhibits several other notable properties. Extracts of AR demonstrate significant anti-inflammatory effects in murine models of asthma and particulate matter-induced lung inflammation, potentially through the inhibition of the NF-κB pathway [166]. This effect may be attributed to components such as Senkyunolide A (294), which mitigates airway injury and remodeling in asthmatic mice and modulates the progression of asthma, consistent with the findings of [167]. Furthermore, the polysaccharides present in AR exhibit anti-aging properties and may serve as potential antioxidants [168]. These findings support the potential development of AR as a novel pharmaceutical and natural health product. However, factors such as poor water solubility and low bioavailability need to be overcome.
6. Toxicity
An analysis of ancient texts and contemporary research indicates that AR is characterized by minimal toxicity, a finding corroborated by extensive acute and chronic toxicity evaluations. In vivo studies using animal models such as mice, rats, and zebrafish embryos have systematically assessed its safety margin. Specifically, oral acute toxicity tests of A. lancea crude ethanolic extracts demonstrated a median lethal dose (LD50) greater than 2000 mg/kg, with no significant liver or kidney damage or pathological changes observed in other organs [169]. Furthermore, Even after processing, AR does not exhibit any significant toxicity [170]. This further demonstrates that AR is safe within certain dosage ranges. The safety profile is closely linked to its therapeutic mechanisms; studies confirm that AR mitigates inflammation and oxidative stress by inhibiting the NF-κB signaling pathway and activating the Nrf2 pathway without obvious toxic side effects at therapeutic doses [171].
Although the crude herb exhibits a wide safety window, investigations into major active constituents reveal dose-dependent toxicological effects that require careful consideration. AR samples with concentrations ranging from 0 to 25 mg/mL have demonstrated no toxic effects on A549 cells [172]. However, evaluations using in vitro and zebrafish embryo models for constituents such as atractylenolides (I, II, and III), atractylones, and β-eudesmol have identified specific risks. Notably, atractylenolides I (271) and II (268) have been shown to induce significant developmental abnormalities and hepatotoxicity in zebrafish embryos, evidenced by liver atrophy, decreased liver-specific fluorescence intensity, elevated liver enzyme levels, apoptosis, and the inhibition of drug-metabolizing enzymes [173]. At specific concentrations, β-eudesmol (71) and atractylones are linked to decreased embryo survival rates, developmental abnormalities, and alterations in the expression of genes related to oxidative stress, suggesting potential risks of embryotoxicity and oxidative damage [174]. Collectively, while AR possesses a substantial safety margin supporting its traditional use, certain active compounds may pose risks of developmental and hepatotoxicity at elevated doses or during embryonic development. Therefore, it is recommended that clinical applications carefully regulate dosages, avoid prolonged administration of high doses, and advance toxicological research and safety monitoring of these active constituents to ensure safe therapeutic application.
7. Limitations
Despite the comprehensive and systematic nature of this review, which integrates historical herbal textual research with modern phytochemical and pharmacological findings to provide a holistic understanding of AR, several inherent limitations must be acknowledged. These limitations primarily pertain to methodological constraints within the reviewed literature, the predominant reliance on preclinical evidence, and the corresponding lack of robust clinical validation, which collectively warrant a critical interpretation of the findings presented. First, from a methodological perspective, significant heterogeneity exists across the included studies regarding extraction protocols, compound identification, and experimental models. Many pharmacological investigations have utilized crude extracts or essential oils without standardized quantification of their major bioactive constituents, complicating the attribution of observed effects to specific compounds and hindering cross-study comparability. Furthermore, although computational approaches such as molecular docking have been extensively employed to predict mechanisms of action, these in silico findings frequently lack experimental validation through target engagement assays or genetic knockdown models, leaving proposed molecular interactions largely speculative.
Second, the current evidence base is overwhelmingly dominated by preclinical studies, with in vitro experiments on cell lines and in vivo evaluations in rodent models constituting the vast majority of pharmacological data. While these studies provide valuable mechanistic insights and establish biological plausibility, they possess inherent limitations in recapitulating human disease pathophysiology, particularly regarding drug metabolism, immune system interactions, and chronic disease progression. The translational relevance of these findings remains uncertain without confirmation in higher animal models or human systems. Third, and perhaps most critically, clinical evidence validating the therapeutic efficacy and safety of AR in human subjects remains exceptionally limited. The few available clinical studies are characterized by small sample sizes, lack of randomization or blinding, and absence of standardized AR preparations with verified phytochemical profiles. Consequently, definitive conclusions regarding optimal dosing, long-term safety, and comparative effectiveness against standard therapies cannot be drawn. This scarcity of rigorous clinical trials represents the most significant barrier to evidence-based clinical application. Additionally, the two pharmacopoeial species, A. lancea and A. chinensis, are often conflated in both experimental and clinical literature, yet emerging evidence demonstrates substantial chemotaxonomic differences that may translate into differential pharmacological activities. This conflation introduces confounding variables that undermine the specificity and reproducibility of research findings. It must not be overlooked that one reason for AR’s favourable biological activity lies in its nature as a complex mixture of multiple compounds, presenting an unavoidable challenge when considering it for development and therapeutic applications.
By explicitly acknowledging these limitations, this review aims to enhance critical depth and transparency, emphasizing that while the foundational science of AR is robust and promising, its clinical translation remains nascent. Future research must prioritize methodological rigor, standardized phytochemical characterization, and well-designed clinical trials to transform the extensive preclinical promise of AR into evidence-based therapeutic applications.
8. Conclusions
Despite significant advancements in understanding the phytochemistry and pharmacology of AR, several critical challenges and knowledge gaps persist, necessitating a paradigm shift in future research endeavors. The current body of evidence is constrained by three fundamental contradictions: the discordance between complex phytochemical profiles and poorly defined molecular mechanisms, the disparity between potent in vitro bioactivities and unfavorable in vivo pharmacokinetics characterized by low bioavailability, and the clinical interchangeability of two chemically distinct pharmacopoeial species, specifically A. lancea and A. chinensis.
To bridge these gaps and facilitate the evidence-based development of AR from a traditional remedy to a modern therapeutic agent, future investigations should prioritize five interconnected research directions. First, the in vivo fate and direct protein targets of its bioactive constituents demand rigorous elucidation through integrated pharmacokinetic profiling and chemical proteomics approaches such as activity-based protein profiling, thereby establishing a definitive molecular basis for its traditional efficacy in invigorating the spleen and resolving dampness. Second, the synergistic mechanisms underlying its multi-component nature should be systematically deconvoluted by employing network pharmacology integrated with multi-omics readouts including transcriptomics, proteomics, and metabolomics to map the compound–target–pathway–phenotype network, with particular emphasis on the combinatorial effects and potential synergism among major constituents like atractylodin, β-eudesmol, and the atractylenolides. Third, overcoming the intrinsic challenge of poor bioavailability, particularly for hydrophobic sesquiterpenes, is paramount and necessitates the rational design of advanced drug delivery systems including nanocarriers such as liposomes and polymeric nanoparticles, alongside investigations into traditional combination strategies that may enhance gastrointestinal absorption. Fourth, given the pronounced chemotaxonomic variation between A. lancea and A. chinensis, rigorous comparative studies are urgently required to correlate their distinct chemical signatures with differential pharmacological potencies in core therapeutic areas such as gastroprotection and anti-inflammation, thereby providing a scientific rationale for species-specific clinical application and quality control. Finally, a comprehensive safety evaluation framework extending beyond conventional acute toxicity models is essential; this should leverage advanced platforms including zebrafish embryos and organoid technologies to systematically assess the chronic, developmental, and organ-specific toxicity profiles of major constituents, thereby establishing definitive safety margins for clinical translation. By addressing these critical areas through the application of contemporary life sciences methodologies, future research can transform the therapeutic potential of AR into clinically validated, mechanism-based interventions for gastrointestinal, metabolic, and inflammatory disorders.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules31061015/s1, Table S1. Traditional preparations of Atractylodis Rhizoma; Table S2. Modern preparations of Atractylodis Rhizoma; Table S3. Antimicrobial effect of Atractylodis Rhizoma; Table S4. Anti-inflammatory effect of Atractylodis Rhizoma; Table S5. Antioxidant effect of Atractylodis Rhizoma; Table S6. Hepatoprotective effect of Atractylodis Rhizoma; Table S7. Anti-cancer effect of Atractylodis Rhizoma; Table S8. Intestinal regulatory function of Atractylodis Rhizoma.
Author Contributions
J.S.: Writing—review and editing, Writing—original draft, Visualization, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. J.Q.: Writing—review and editing, Visualization. J.T.: Data curation. N.C.: Supervision. M.G.: Writing—review and editing. J.Y.: Writing—review and editing, Supervision, Methodology, Funding acquisition, Conceptualization. B.K.: Writing—review and editing, Supervision, Resources, Funding acquisition, Conceptualization. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Natural Science Foundation of China (82504995), the Anhui Provincial College Student Innovation and Entrepreneurship Training Program (S202512926013), and the Bozhou University Research Start-up Project (BYKQ202536).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript: AR, Atractylodis Rhizoma; MIC, Minimum Inhibitory Concentration; MBC, Minimum Bactericidal Concentration; AA, arachidonic acid; TPA, tetradecanoylphorbol-13-acetate-; DPPH, 1,1-diphenyl-2-picryl-hydrazyl radical; ABTS, 2, 2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); FRAP, ferric reducing antioxidant power; TRP, total reducing power; CUPRAC, cupric reducing antioxidant capacity; NO, Nitric Oxide; 8-OHdG, 8-Hydroxy-2′-deoxyguanosine; Aβ, Amyloid-beta; ACC, Acetyl-CoA Carboxylase; AChE, Acetylcholinesterase; ACLY, ATP-Citrate Lyase; ADAM10, A Disintegrin And Metalloproteinase 10; ADAMTS-4, A Disintegrin And Metalloproteinase with Thrombospondin Motifs 4; ADAMTS-5, A Disintegrin And Metalloproteinase with Thrombospondin Motifs 5; Aggrecan, Aggrecan; Akt, Protein Kinase B; ALP, Alkaline Phosphatase; ALT, Alanine Aminotransferase; AMPK, AMP-activated Protein Kinase; ANP, Atrial Natriuretic Peptide; AQP4, Aquaporin-4; ARE, Antioxidant Response Element; ARG-1, Arginase-1; AST, Aspartate Aminotransferase; ATP6V0D2, V-type Proton ATPase Subunit D2; BACE1, Beta-site APP Cleaving Enzyme 1; Bax, BCL2-Associated X Protein; BCL2, B-cell Lymphoma 2; BDNF, Brain-Derived Neurotrophic Factor; Bcl-2, B-cell Lymphoma 2; Bcl-xl, B-cell Lymphoma-extra Large; BNP, Brain Natriuretic Peptide; Carbamide, Urea; CAT, Catalase; CCL2, C-C Motif Chemokine Ligand 2; CD206, Cluster of Differentiation 206; CD40, Cluster of Differentiation 40; CD80, Cluster of Differentiation 80; CD86, Cluster of Differentiation 86; CDK2, Cyclin-dependent Kinase 2; CHO, Total Cholesterol; CLCC1, Chloride Channel CLIC-like 1; Claudin-1, Claudin-1; COX-2, Cyclooxygenase-2; CPT-1, Carnitine Palmitoyltransferase I; Creatinine, Creatinine; CRP, C-reactive Protein; CRT, Calreticulin; CS, Citrate Synthase; cTn-I, Cardiac Troponin I; CXCL16, C-X-C Motif Chemokine Ligand 16; CYP17A1, Cytochrome P450 Family 17 Subfamily A Member 1; CYP2b10, Cytochrome P450 Family 2 Subfamily B Member 10; CYP3a11, Cytochrome P450 Family 3 Subfamily A Member 11; DAI Score, Disease Activity Index Score; Dgat2, Diacylglycerol O-Acyltransferase 2; DR3, Death Receptor 3; ECAR, Extracellular Acidification Rate; EGFR, Epidermal Growth Factor Receptor; EMT, Epithelial–Mesenchymal Transition; ER, Endoplasmic Reticulum; ERK, Extracellular Signal-regulated Kinase; Fasn, Fatty Acid Synthase; FAK, Focal Adhesion Kinase; FGF1, Fibroblast Growth Factor 1; FGFR, Fibroblast Growth Factor Receptor; G6PD, Glucose-6-Phosphate Dehydrogenase; GABA, Gamma-Aminobutyric Acid; GAPDH, Glyceraldehyde-3-Phosphate Dehydrogenase; GDH, Glutamate Dehydrogenase; GLS, Glutaminase; GPX4, Glutathione Peroxidase 4; GPx, Glutathione Peroxidase; GR, Glutathione Reductase; GSH, Glutathione; GSDMD, Gasdermin D; GSSG, Oxidized Glutathione; GST, Glutathione S-Transferase; H2O2, Hydrogen Peroxide; HAS2, Hyaluronan Synthase 2; HAS3, Hyaluronan Synthase 3; HDL-C, High-Density Lipoprotein Cholesterol; HER2, Human Epidermal Growth Factor Receptor 2; Hes1, Hes Family BHLH Transcription Factor 1; HIF-1α, Hypoxia-Inducible Factor 1-alpha; HMGB1, High Mobility Group Box 1; HO-1, Heme Oxygenase-1; hs-CRP, High-sensitivity C-reactive Protein; HSD17B3, Hydroxysteroid 17-Beta Dehydrogenase 3; IAA, Indole-3-acetic Acid; ICAM-1, Intercellular Adhesion Molecule-1; IFN-γ, Interferon-gamma; IGF-1, Insulin-like Growth Factor 1; IKK, Inhibitor of Nuclear Factor Kappa-B Kinase; IL-1β, Interleukin-1 beta; IL-6, Interleukin-6; IL-8, Interleukin-8; IL-10, Interleukin-10; IL-12, Interleukin-12; IL-17A, Interleukin-17A; IL-18, Interleukin-18; IL-23, Interleukin-23; iNOS, Inducible Nitric Oxide Synthase; IRF1, Interferon Regulatory Factor 1; JAK2, Janus Kinase 2; JNK, c-Jun N-terminal Kinase; Keap1, Kelch-like ECH-associated Protein 1; KYNA, Kynurenic Acid; LAMP1, Lysosomal-associated Membrane Protein 1; LAMP2, Lysosomal-associated Membrane Protein 2; LDH, Lactate Dehydrogenase; LDHA, Lactate Dehydrogenase A; LDL-C, Low-Density Lipoprotein Cholesterol; LPO, Lipid Peroxidation; LPS, Lipopolysaccharide; LVEF, Left Ventricular Ejection Fraction; LVMI, Left Ventricular Mass Index; MCP-1, Monocyte Chemoattractant Protein-1; MDA, Malondialdehyde; Mdr1a, Multidrug Resistance Protein 1a; MGL-1, Macrophage Galactose-type Lectin 1; MGL-2, Macrophage Galactose-type Lectin 2; MIP-1α, Macrophage Inflammatory Protein-1 alpha; MLCK, Myosin Light Chain Kinase; MMP, Matrix Metalloproteinase; MMP-2, Matrix Metalloproteinase 2; MMP-9, Matrix Metalloproteinase 9; MMP13, Matrix Metalloproteinase 13; MPO, Myeloperoxidase; mTOR, Mammalian Target of Rapamycin; MUC2, Mucin 2; MyD88, Myeloid Differentiation Primary Response 88; NE, Norepinephrine; NF-κB, Nuclear Factor Kappa-light-chain-enhancer of Activated B cells; Ngn2, Neurogenin 2; NLRC4, NLR Family CARD Domain Containing 4; NLRP1, NLR Family Pyrin Domain Containing 1; NLRP3, NLR Family Pyrin Domain Containing 3; NCOA4, Nuclear Receptor Coactivator 4; Nrf2, Nuclear Factor Erythroid 2-Related Factor 2; OCR, Oxygen Consumption Rate; Occludin, Occludin; p21, Cyclin-dependent Kinase Inhibitor 1A; p38, p38 Mitogen-Activated Protein Kinase; p53, Tumor Protein p53; p65, Nuclear Factor NF-kappa-B p65 subunit; p70S6K, Ribosomal Protein S6 Kinase; PARP, Poly ADP-ribose Polymerase; PBLD, Phenazine Biosynthesis-Like Protein Domain-containing Protein; PD-L1, Programmed Death-Ligand 1; PECAM-1, Platelet Endothelial Cell Adhesion Molecule-1; PFKFB3, 6-Phosphofructo-2-Kinase/Fructose-2,6-Biphosphatase 3; PI3K, Phosphatidylinositol 3-Kinase; PKM2, Pyruvate Kinase M2; p-MLC, Phosphorylated Myosin Light Chain; PMN, Polymorphonuclear Neutrophil; PPARα, Peroxisome Proliferator-Activated Receptor Alpha; PGE2, Prostaglandin E2; RAGE, Receptor for Advanced Glycation Endproducts; RANTES, Regulated on Activation, Normal T Expressed and Secreted; RhoA, Ras Homolog Family Member A; ROCK, Rho-associated Coiled-coil Containing Protein Kinase; ROS, Reactive Oxygen Species; RXRa, Retinoid X Receptor Alpha; Scd2, Stearoyl-CoA Desaturase 2; Serine, Serine; SIRT1, Sirtuin 1; SNAP23, Synaptosome Associated Protein 23; SOD, Superoxide Dismutase; Srebf1, Sterol Regulatory Element Binding Transcription Factor 1; SREBP-1, Sterol Regulatory Element-Binding Protein 1; Src, Proto-oncogene Tyrosine-Protein Kinase Src; STAT1, Signal Transducer and Activator of Transcription 1; STAT3, Signal Transducer and Activator of Transcription 3; STAT6, Signal Transducer and Activator of Transcription 6; TAC, Total Antioxidant Capacity; TC, Total Cholesterol; TEER, Transepithelial Electrical Resistance; TG, Triglycerides; TGF-β1, Transforming Growth Factor Beta 1; TLR4, Toll-like Receptor 4; TNF-α, Tumor Necrosis Factor-alpha; TRAF6, TNF Receptor Associated Factor 6; TRPM8, Transient Receptor Potential Melastatin 8; TRPV1, Transient Receptor Potential Vanilloid 1; TWIST1, Twist Family BHLH Transcription Factor 1; Uric acid, Uric Acid; VDR, Vitamin D Receptor; VEGFA, Vascular Endothelial Growth Factor A; Vimentin, Vimentin; XCL1, X-C Motif Chemokine Ligand 1; xCT, Cystine/Glutamate Antiporter; XIAP, X-linked Inhibitor of Apoptosis Protein; ZEB1, Zinc Finger E-box Binding Homeobox 1; ZO-1, Zonula Occludens-1; α-SMA, Alpha-Smooth Muscle Actin.
References
- Jun, X.; Fu, P.; Lei, Y.; Cheng, P. Pharmacological effects of medicinal components of Atractylodes lancea (Thunb.) DC. Chin. Med. 2018, 13, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L. Textual Research of Historical and Current Application of Atractylodis Macrocephalae Rhizoma in Classic Famous Prescriptions. Chin. J. Mod. Appl. Pharm. 2024, 41, 2002–2006. [Google Scholar]
- Ke, C.; Qu, L.; Liu, Y.; Xia, Y.; Wang, C.; Xu, K. Rhizoma Atractylodis: A review on processing, chemical composition, pharmacological effects, and product development. Acupunct. Herb. Med. 2025, 5, 115–130. [Google Scholar] [CrossRef]
- Yang, L.; Yu, H.; Hou, A.; Man, W.; Wang, S.; Zhang, J.; Wang, X.; Zheng, S.; Jiang, H.; Kuang, H. A Review of the Ethnopharmacology, Phytochemistry, Pharmacology, Application, Quality Control, Processing, Toxicology, and Pharmacokinetics of the Dried Rhizome of Atractylodes macrocephala. Front. Pharmacol. 2021, 12, 727154. [Google Scholar] [CrossRef]
- Wu, Y.J.; Tang, L. Efficacy Analysis of Wandai Decoction Combined with Traditional Chinese Medicine Fumigation and Washing in Patients with Chronic Vaginitis After Sintilimab Treatment for Small Cell Lung Cancer. Altern. Ther. Health Med. 2023, 29, 268–273. [Google Scholar]
- Yang, Y.L.; Zhao, C.Z.; Zhao, C.C.; Wen, Z.Y.; Ma, Y.Y.; Zhao, X.N.; Wang, L.; Huang, J.L.; Zhou, P. Ling-Gui-Zhu-Gan decoction protects against doxorubicin-induced myocardial injury by downregulating ferroptosis. J. Pharm. Pharmacol. 2024, 76, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhu, T.Y.; Dong, Y.X.; Zhao, C.Y.; Chen, J.H.; Tian, Y.X.; Liu, Y.L.; Hong, X.; Xiong, H. UPLC-Q-TOF-MS-Based Serum Metabolomics Explores the Mechanism of Pingwei Powder in Treating the Damp Retention in the Middle-Jiao Syndrome. Biomed. Chromatogr. 2025, 39, e70143. [Google Scholar] [CrossRef]
- Zhang, Y.N.; Zhang, S.; Fan, Y.M.; Huang, S.J.; Wang, S.M.; Hao, Z.H.; Shen, J.Z. Exploring the Underlying Mechanism of Weiling Decoction Alleviates Cold-Dampness Diarrhea Based on Network Pharmacology, Transcriptomics, Molecular Docking and Experimental Validation. Pharmaceuticals 2025, 18, 109. [Google Scholar] [CrossRef]
- Jiang, Z.; Jin, K.; Zhong, L.; Zheng, Y.; Shao, Q.; Zhang, A. Near-infrared spectroscopy combined with machine learning for rapid identification of Atractylodis rhizoma decoction pieces. Ind. Crops Prod. 2023, 197, 116579. [Google Scholar] [CrossRef]
- Wang, M.; Chen, P.; Yin, M.; Xu, X.; Chen, Y.; Feng, X.; Guan, F.; Liao, P.; Wang, Q. Phytochemical and chemotaxonomic study on Atractylodes lancea. Biochem. Syst. Ecol. 2023, 111, 104734. [Google Scholar] [CrossRef]
- Ma, Z.; Zhao, X.; Xie, Z.; Lv, M.; Gao, J.; Sun, L.; Li, J.; Ren, X. Fourier transform infrared spectroscopy, high-performance liquid chromatography with diode array detection, and gas chromatography-mass spectrometry fingerprints combined with chemometrics for comprehensive evaluation and identification of raw and bran-fried Atractylodis Rhizoma. J. Pharm. Biomed. Anal. 2025, 262, 116872. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.J.; Zhao, Z.Y.; Chang, L.K.; Cao, Y.; Wang, S.; Kang, C.Z.; Wang, H.Y.; Zhou, L.; Huang, L.Q.; Guo, L.P. Atractylodis Rhizoma: A review of its traditional uses, phytochemistry, pharmacology, toxicology and quality control. J. Ethnopharmacol. 2021, 266, 113415. [Google Scholar] [CrossRef]
- Kou, B.; Meng, L.; Zhao, M.; Wang, H.; Lu, C.; Yan, M.; Li, G. Unveiling the power of Pueraria lobata: A comprehensive exploration of its medicinal and edible potentials. Front. Pharmacol. 2025, 16, 1578472. [Google Scholar] [CrossRef]
- Feng, J.X.; Wu, Y.Z.; Li, S.Y.; Chang, A.; Yu, Q.X.; Zhang, H. Correlation between active component content and color of Atractylodes lancea and A. chinensis based on color difference principle. China J. Chin. Mater. Med. 2023, 48, 2086–2091. [Google Scholar] [CrossRef]
- Xiao, W. Kinetics and Mechanism Studies on Oxidizing Reaction of Atractylon in Essential Oil from Aatractylodes mmacrocephala Koidz. Chin. J. Appl. Chem. 2007, 24, 301–305. [Google Scholar]
- Yan, M.; Wei, T.; Zhao, D.; Wei, X.; Chen, F.; Wang, C.; Xiao, C. Comprehensive Quality Evaluation of Bran-processed Cangzhu (Atractylodis Rhizoma) Based on Chemometrics and Entropy Weight TOPSIS Analysis Combined with Multi-Component Quantification. Guid. J. Tradit. Chin. Med. Pharmacol. 2023, 29, 55–62. [Google Scholar]
- Manayi, A.; Kurepaz-mahmoodabadi, M.; Gohari, A.R.; Ajani, Y.; Saeidnia, S. Presence of phthalate derivatives in the essential oils of a medicinal plant Achillea tenuifolia. DARU J. Pharm. Sci. 2014, 22, 78. [Google Scholar] [CrossRef]
- Gan, Y.F.; Yang, T.; Gu, W.; Guo, L.P.; Qiu, R.L.; Wang, S.; Zhang, Y.; Tang, M.; Yang, Z.C. Using HS-GC-MS and flash GC e-nose in combination with chemometric analysis and machine learning algorithms to identify the varieties, geographical origins and production modes of Atractylodes lancea. Ind. Crops Prod. 2024, 209, 117955. [Google Scholar] [CrossRef]
- Gan, Y.; Ju, R.; Peng, Y.; Xiao, S.; Qiu, R.; Wang, S.; Zhang, Y.; Guo, L.; Gu, W. A multi-platform analytical strategy for Atractylodes lancea authentication: Fusion of stable isotope, elemental, chromatographic, and spectroscopic profiles. Talanta 2026, 305, 129603. [Google Scholar] [CrossRef]
- Lu, J.; Chen, W.T.; Zhou, B.W.; Chen, Y.; Wang, X.H.; An, R.; Yang, M. Distinguishing the Rhizomes of Atractylodes japonica, Atractylodes chinensis, and Atractylodes lancea by Comprehensive Two-Dimensional Gas Chromatography Coupled with Mass Spectrometry Combined with Multivariate Data Analysis. Pharmacogn. Mag. 2020, 16, 654–661. [Google Scholar] [CrossRef]
- Ji, L.; Ao, P.; Pan, J.G.; Yang, J.Y.; Yang, J.; Hu, S.L. GC-MS analysis of essential oils from rhizomes of Atractylodes lancea (Thunb.) DC. and A. chinensis (DC.) Koidz. China J. Chin. Mater. Med. 2001, 26, 182–185. [Google Scholar]
- Xu, C.; Meng, L.B.; Lu, M.Q.; Huang, X.Y.; Wang, X.; Gong, F.P.; Gong, Q.F.; Yu, H. Influence of different processing methods on volatile components of Atractylodis Rhizoma based on HS-GC-MS technology. China J. Chin. Mater. Med. 2024, 49, 4427–4436. [Google Scholar] [CrossRef]
- Zhuang, L.X.; Liu, Y.; Wang, S.Y.; Sun, Y.; Pan, J.; Guan, W.; Hao, Z.C.; Kuang, H.X.; Yang, B.Y. Cytotoxic Sesquiterpenoids from Atractylodes chinensis (DC.) Koidz. Chem. Biodivers. 2022, 19, e202200812. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Wang, W.; Liu, R.; Cai, J.; Jiang, Y.; Tang, X.; Wu, H.; Ao, H.; Chen, L. Geographic Differentiation of Essential Oil from Rhizome of Cultivated Atractylodes lancea by Using GC-MS and Chemical Pattern Recognition Analysis. Molecules 2023, 28, 2216. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.-Q.; Huang, L.-F.; Zhou, S.-Y.; Zhang, T.-M.; Liang, Y.-Z. Comparison of the volatile compounds of Atractylodes medicinal plants by headspace solid-phase microextraction-gas chromatography–mass spectrometry. Anal. Chim. Acta 2006, 570, 73–78. [Google Scholar] [CrossRef]
- Jia, C.; Mao, D.; Zhang, W.; Sun, X. Studies on chemical constituents in essential oil from wild Atractylodes lancea in dabie mountains. J. Chin. Med. Mater. 2004, 27, 571–574. [Google Scholar]
- Ahmed, S.; Zhan, C.S.; Yang, Y.Y.; Wang, X.K.; Yang, T.W.; Zhao, Z.Y.; Zhang, Q.Y.; Li, X.H.; Hu, X.B. The Transcript Profile of a Traditional Chinese Medicine, Atractylodes lancea, Revealing Its Sesquiterpenoid Biosynthesis of the Major Active Components. PLoS ONE 2016, 11, e0151975. [Google Scholar] [CrossRef]
- Zhang, Z.A.; Xue, X.Y.; Wu, J.Y.; Zhao, X.; Zhang, H.; Sun, K.; Dai, C.C.; Chen, F. CYP71P2 regulated by MYB44 modulates oxygenated sesquiterpenoids biosynthesis in Atractylodes lancea. Plant Physiol. Biochem. 2025, 229, 110700. [Google Scholar] [CrossRef]
- Xu, X.J.; Xiong, X.H.; He, Z.Q.; Lu, Q.; Wang, L. Optimization of ultrasound pretreatment combined with solvent-free microwave for extracting volatile oil from Atractylodes lancea and its chemical composition and antimicrobial activity. Talanta 2026, 297, 128724. [Google Scholar] [CrossRef]
- Kamauchi, H.; Kinoshita, K.; Takatori, K.; Sugita, T.; Takahashi, K.; Koyama, K. New sesquiterpenoids isolated from Atractylodes lancea fermented by marine fungus. Tetrahedron 2015, 71, 1909–1914. [Google Scholar] [CrossRef]
- Chu, S.S.; Jiang, G.H.; Liu, Z.L. Insecticidal compounds from the essential oil of Chinese medicinal herb Atractylodes chinensis. Pest Manag. Sci. 2011, 67, 1253–1257. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Deng, J.; Duan, G.H.; Chen, L.; Huang, X.; Wang, W.J.; Gong, L.; Zhang, Y.; Yu, K.; Guo, L.P. Insights into the impacts of autotoxic allelochemicals from rhizosphere of Atractylodes lancea on soil microenvironments. Front. Plant Sci. 2023, 14, 1136833. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Yun, Y.K.; Ahn, Y.J. Toxicity of atractylon and atractylenolide III identified in Atractylodes ovata rhizome to Dermatophagoides farinae and Dermatophagoides pteronyssinus. J. Agric. Food Chem. 2007, 55, 6027–6031. [Google Scholar] [CrossRef] [PubMed]
- Bagal, S.K.; Adlington, R.M.; Marquez, R.; Cowley, A.R.; Baldwin, J.E. Studies towards the biomimetic synthesis of bisesquiterpene lactones. Tetrahedron Lett. 2003, 44, 4993–4996. [Google Scholar] [CrossRef]
- Zhao, J.H.; Sun, C.Z.; Shi, F.Y.; Ma, S.S.; Zheng, J.S.; Du, X.; Zhang, L.P. Comparative transcriptome analysis reveals sesquiterpenoid biosynthesis among 1-, 2-and 3-year old Atractylodes chinensis. BMC Plant Biol. 2021, 21, 354. [Google Scholar] [CrossRef]
- Lei, H.; Yue, J.; Yin, X.Y.; Fan, W.; Tan, S.H.; Qin, L.; Zhao, Y.N.; Bai, J.H. HS-SPME coupled with GC-MS for elucidating differences between the volatile components in wild and cultivated Atractylodes chinensis. Phytochem. Anal. PCA 2023, 34, 317–328. [Google Scholar] [CrossRef]
- Li, Y. Impact of Using Natural Herbal Materials for Acupoint Application Combined with an Evidence-Based Supervision Method on Improving Renal Function and Lifestyle Treatment in Hemodialysis Patients. J. Biobased Mater. Bioenergy 2023, 17, 624–630. [Google Scholar] [CrossRef]
- Chen, L.N.; Li, Y.H.; Huang, X.; Deng, J.; Qu, C.L.; Zhang, X.Q.; Huang, B.S.; Zhang, Y.; Gong, L.; Yu, K. Cloning and functional characterization of a terpene synthase gene AlTPS1 from Atractylodes lancea. Biol. Plant. 2021, 65, 351–358. [Google Scholar] [CrossRef]
- Zhou, J.Y.; Li, X.; Zheng, J.Y.; Dai, C.C. Volatiles released by endophytic Pseudomonas fluorescens promoting the growth and volatile oil accumulation in Atractylodes lancea. Plant Physiol. Biochem. 2016, 101, 132–140. [Google Scholar] [CrossRef]
- Zhi, Z.; Xuening, Y.; Shimin, P.; Tao, Z.; Miaoting, S. Comparison of the Volatile Oil Components from Atractylodes chinensis (DC.) Koidz. and Atractylodes lancea (Thunb.) DC. Chin. J. Appl. Chem. 2012, 29, 470–476. [Google Scholar] [CrossRef]
- Fang, J.; Weng, L.; Wang, M.; Xiao, C.; Yang, X.; Sun, J.; Feng, Y. Analysis of chemical compositions in Atractylodes chinensis with rice water before and after processing and its effects on intestinal fungal flora of spleen deficiency diarrhea rats. Chin. Tradit. Herb. Drugs 2023, 53, 7372–7383. [Google Scholar]
- Wang, C.; Xiang, Q.; Zhao, W.; Gong, Q.; Yu, H. Analysis of Chemical Compositions in Atractylodes lancea Rhizoma Before and After Processing with Rice-washed Water by UPLC-Q-TOF-MS. Chin. J. Exp. Tradit. Med. Formulae 2022, 28, 164–173. [Google Scholar] [CrossRef]
- Lin, X.; Chen, Z.; Zou, J.; Shi, Y.; Zhang, X.; Guo, D.; Zhai, B.; Luan, F. Light stability examination and GC-MS analysis of volatile oils of Acori Tatarinowii Rhizoma and Atractylodis Rhizoma under treatment of β-cyclodextrin inclusion and Pickering emulsion technology. Chin. Tradit. Herb. Drugs 2025, 56, 441–457. [Google Scholar]
- Ding, S.; Qiu, M.; Cao, Y.; Pan, L. Identification of Different Volatile Components in Fresh and Processed Atractylodis Macrocephala Rhizoma Based on GC-IMS. J. Instrum. Anal. 2025, 44, 2486–2496. [Google Scholar]
- Liu, X.; Yan, X.; Wei, Y. Analysis of the Essential Oil Compositionsin Rhizome of Atractylodes lancea (Thunb) DC. J. Instrum. Anal. 1998, 17, 56–57. [Google Scholar]
- Wang, F.; Ouyang, Z.; Guo, L.P.; Zhao, M.; Peng, H.S.; Liao, J.L.; Liang, Z.P. Comprehensive chemical pattern recognition of atractylodis rhizoma. China J. Chin. Mater. Med. 2014, 39, 2536–2541. [Google Scholar]
- Li, X.; Bai, Y.; Qu, Y.; Cai, Q. Study on the difference of chemical component in three processed products of Atractylodes chinensis (DC.) Koidz. China J. Tradit. Chin. Med. Pharm. 2024, 39, 6658–6665. [Google Scholar]
- Xin, Y.; Yu, K.C.; Yu, Y.; Wang, H.J. Filtration and qualification for target biomarkers of traditional Chinese medicine formula “fuzi lizhong decoction” acting on stomach ulcer by UPLC/Q-TOF MS. Pak. J. Pharm. Sci. 2022, 35, 519–528. [Google Scholar] [CrossRef]
- Lu, M.X.; Yin, J.Y.; Xu, T.S.; Dai, X.; Liu, T.Y.; Zhang, Y.Y.; Wang, S.; Liu, Y.G.; Shi, H.F.; Zhang, Y.F.; et al. Fuling-Zexie formula attenuates hyperuricemia-induced nephropathy and inhibits JAK2/STAT3 signaling and NLRP3 inflammasome activation in mice. J. Ethnopharmacol. 2024, 319, 117262. [Google Scholar] [CrossRef]
- Deng, A.P.; Li, Y.; Wu, Z.T.; Liu, T.; Kang, L.P.; Nan, T.G.; Zhan, Z.L.; Guo, L.P. Advances in studies on chemical compositions of Atractylodes lancea and their biological activities. China J. Chin. Mater. Med. 2016, 41, 3904–3913. [Google Scholar] [CrossRef]
- Liu, X.; Liang, L.; Cai, G.; Guo, Y.; Gong, J. Multivariate approach to assess the bioactive compounds of Atractylodes chinensis (DC.) Koidz in different harvest periods. J. Chromatogr. B 2024, 1246, 124298. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Y.; Wang, B.; Guan, D.X.; Zhou, J.J.; Men, Z.; Sun, Y.T. A research method of compatibility mechanism of traditional Chinese medicine prescription-taking Yupingfeng San as an example. J. Ethnopharmacol. 2025, 348, 119815. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.Y.; Liu, J.N.; Pang, Y.L.; Wang, H.M. Ultrasonic-assisted deep eutectic solvent extraction and identification of phenolic compounds from Atractylodes chinensis adventitious root culture. J. Chromatogr. A 2025, 1757, 466166. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.Z.; Zeng, L.; Wang, X.P.; Su, L.L.; Sun, M.M.; Wu, B.S.; Yan, S. Loading of AgNPs onto the surface of boron nitride nanosheets for determination of scopoletin in Atractylodes macrocephala. Sci. Rep. 2019, 9, 3864. [Google Scholar] [CrossRef]
- Xia, Z.X.; Li, Q.; Tang, Z.Y. Network pharmacology, molecular docking, and experimental pharmacology explored Ermiao wan protected against periodontitis via the PI3K/AKT and NF-ΚB/MAPK signal pathways. J. Ethnopharmacol. 2023, 303, 115900. [Google Scholar] [CrossRef]
- Feng, Z.M.; Xu, K.; Wang, W.; Du, N.; Zhang, J.H.; Yang, Y.N.; Jiang, J.S.; Zhang, P.C. Two new thiophene polyacetylene glycosides from Atractylodes lancea. J. Asian Nat. Prod. Res. 2018, 20, 531–537. [Google Scholar] [CrossRef]
- Cao, H.; Liu, C.; Chen, G.; Yao, D.; Xiao, Y.; Liu, Y. Metabolic regularity of intestinal flora on alcohol extracts of Atractylodis Rhizoma before and after stir-frying and incremental component atractyloside A by in vitro co-incubation method. Chin. Tradit. Herb. Drugs 2024, 55, 2641–2650. [Google Scholar]
- Zhou, J.; Tang, W.; Chen, J. A UPLC-QTOF-MS/MS Method for the Characterization of Chemical Constituents of Atractylodes lancea (Thunb.) DC. and Atractylodes chinensis (DC.) Koidz. Pharm. Clin. Res. 2020, 28, 321–328. [Google Scholar]
- Zhang, A.; Zhu, J.; Gu, W.; Huang, M.; Wang, S. The optimization of total flavonoid extraction process from the aboveground parts of Atractylodes lancea by orthogonal test and response surface methodology. China Feed 2025, 19, 75–80. [Google Scholar] [CrossRef]
- Cho, H.D.; Kim, U.; Suh, J.H.; Eom, H.Y.; Kim, J.; Lee, S.G.; Choi, Y.S.; Han, S.B. Classification of the medicinal plants of the genus Atractylodes using high-performance liquid chromatography with diode array and tandem mass spectrometry detection combined with multivariate statistical analysis. J. Sep. Sci. 2016, 39, 1286–1294. [Google Scholar] [CrossRef]
- Kim, Y.C.; Jun, M.; Jeong, W.S.; Chung, S.K. Antioxidant properties of flavone C-glycosides from Atradylodes japonica leaves in human low-density lipoprotein oxidation. J. Food Sci. 2005, 70, S575–S580. [Google Scholar] [CrossRef]
- Wang, X.; Han, Y.; Wang, X.; Zhang, Y. Studies on Chemical Composition, Pharmacological Effect, Clinical Application of Pingwei Powder and Its Quality Marker Prediction Analysis. J. Liaoning Univ. Tradit. Chin. Med. 2026, 28, 95–102. [Google Scholar]
- Xu, S.Z.; Qi, X.J.; Liu, Y.Q.; Liu, Y.H.; Lv, X.; Sun, J.Z.; Cai, Q. UPLC-MS/MS of Atractylenolide I, Atractylenolide II, Atractylenolide III, and Atractyloside A in Rat Plasma after Oral Administration of Raw and Wheat Bran-Processed Atractylodis Rhizoma. Molecules 2018, 23, 3234. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, D.; Qin, J.; Wang, H.; Miao, W.; Lv, G. Medicinal compositions of Atractylodis rhizoma: A review. Chin. J. Bioprocess Eng. 2021, 19, 306–313. [Google Scholar]
- Ye, Y.; Chou, G.X.; Wang, H.; Chu, J.H.; Fong, W.F.; Yu, Z.L. Effects of Sesquiterpenes Isolated from Largehead Atractylodes Rhizome on Growth, Migration, and Differentiation of B16 Melanoma Cells. Integr. Cancer Ther. 2011, 10, 92–100. [Google Scholar] [CrossRef]
- Chen, H.P.; Yang, K.; You, C.X.; Zheng, L.S.; Cai, Q.; Wang, C.F.; Du, S.S. Repellency and Toxicity of Essential Oil from Atractylodes chinensis Rhizomes against Liposcelis bostrychophila. J. Food Process. Preserv. 2015, 39, 1913–1918. [Google Scholar] [CrossRef]
- Choi, S.W.; Lee, K.S.; Lee, J.H.; Kang, H.J.; Lee, M.J.; Kim, H.Y.; Park, K.I.; Kim, S.L.; Shin, H.K.; Seo, W.D. Suppression of Akt-HIF-1α signaling axis by diacetyl atractylodiol inhibits hypoxia-induced angiogenesis. Biomol. Biomed. Rep. 2016, 49, 508–513. [Google Scholar] [CrossRef]
- Nakai, Y.; Kido, T.; Hashimoto, K.; Kase, Y.; Sakakibara, I.; Higuchi, M.; Sasaki, H. Effect of the rhizomes of Atractylodes lancea and its constituents on the delay of gastric emptying. J. Ethnopharmacol. 2003, 84, 51–55. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, S.; Li, Q.; Zhang, Y.; He, Y.Q.; Yan, B.; Zhou, L.; Guo, L. Metabolomic profiling and chemical marker identification in medicinal plants of Atractylodes. Sci. Tradit. Chin. Med. 2025, 3, 87–95. [Google Scholar] [CrossRef]
- Yao, D.; Ma, C.Y.; Ke, C.; Wang, D.P.; Xu, K.; Liu, Y.J.; Qu, L.H. Integrating transcriptomics, metabolomics, and microbiomics to explore the mechanism of action of bran-fried Atractylodes lancea rhizome polysaccharide in ameliorating the enhanced pharmacological effects of dextran sodium sulfate-induced colitis. J. Ethnopharmacol. 2025, 349, 119805. [Google Scholar] [CrossRef]
- Ye, Q.Y.; Jiang, Y.; Wu, D.; Cai, J.W.; Jiang, Z.T.; Zhou, Z.; Liu, L.Y.; Ling, Q.H.; Wang, Q.; Zhao, G. Atractylodin alleviates nonalcoholic fatty liver disease by regulating Nrf2-mediated ferroptosis. Heliyon 2023, 9, e18321. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Tian, Y.; Qiao, X.; Li, H.; Ouyang, L.; Li, X.; Geng, X.; Xiao, L.; Ma, Y.; Li, Y. Integrated Analysis of Terpenoid Profiles and Full-Length Transcriptome Reveals the Central Pathways of Sesquiterpene Biosynthesis in Atractylodes chinensis (DC.) Koidz. Int. J. Mol. Sci. 2025, 26, 1074. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, J.; Kamoshita, A.; Ishikawa, T.; Takano, A.; Fukuda, T.; Isoda, S.; Ida, Y. Glycosides of Atractylodes lancea. Chem. Pharm. Bull. 2003, 51, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.L.; Wang, Y.M.; Wang, D.M.; Sun, J.K.; Zhang, Z.G.; Wang, J.H.; Liu, J.; Liu, Q. Phytochemical Profiling of Different Processed Products from Atractyloidis Rhizome using UHPLC/Q-TOF-MS. Pharmacogn. Mag. 2022, 18, 1075–1081. [Google Scholar] [CrossRef]
- Singhuber, J.; Baburin, I.; Kählig, H.; Urban, E.; Kopp, B.; Hering, S. GABAA receptor modulators from Chinese herbal medicines traditionally applied against insomnia and anxiety. Phytomedicine 2012, 19, 334–340. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, G.; Yang, Z.; Zhang, G.; Sun, L.; Wang, M.; Ren, X. Species Differentiation and Quality Evaluation for Atractylodes Medicinal Plants by GC/MS Coupled with Chemometric Analysis. Chem. Biodivers. 2023, 20, e202300793. [Google Scholar] [CrossRef]
- Wang, W.; Jiang, Y.; Song, B.; Tang, X.; Wu, H.; Jin, Z.; Chen, L. Discovery of quality markers in the rhizome of Atractylodes chinensis using GC–MS fingerprint and network pharmacology. Arab. J. Chem. 2023, 16, 105114. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, H.; Li, W.; Zhang, Y.; Luo, J.; Zhao, L.; Shi, F.; Ye, G.; He, X.; Xu, Z.; et al. Chloroform extracts of Atractylodes chinensis inhibit the adhesion and invasion of Salmonella typhimurium. Biomed. Pharmacother. 2022, 154, 113633. [Google Scholar] [CrossRef]
- Zhang, X.C.; Zhu, L.; Li, X.Y.; Liu, L.C.; Lai, P.X. Chemical Composition, and Evaluation of Antibacterial, Antibiofilm and Synergistic Effects with Conventional Antibiotics of Essential Oil from Mallotus repandus. Rec. Nat. Prod. 2021, 15, 324–329. [Google Scholar] [CrossRef]
- Ghavam, M.; Bacchetta, G.; Castangia, I.; Manca, M.L. Phlomoides molucelloides (Bunge) Salmaki essential oil: A traditional remedy revitalized for modern antimicrobial challenges. Inflammopharmacology 2025, 33, 7033–7043. [Google Scholar] [CrossRef]
- Ladjel-Mendil, A.; Amarni, M.; Chelghoum, H.; Lacheheb, S.; Moussa, H.; Aboumustapha, M.; Chebrouk, F.; Boudjelal, A.; Kebir, M.; Benguerba, Y.; et al. Essential oil from aerial parts of Salvia sclarea: A comprehensive study of antibacterial activity through chemical profiling, molecular interactions, and predictive modeling using QSAR_KNN_PCA. J. Essent. Oil Bear. Plants 2025, 28, 902–919. [Google Scholar] [CrossRef]
- Prabaharan, J.; Prabakaran, M.; Prabhakaran, M.; Abinaya, K.; Krishnan, N.; Karen, D.S.; Veena, J.; Dhanbalan, A.K.; Devadasan, V.; Gopinath, S.C.B.; et al. Comparison on extracted metabolites from different regions grown Murraya koenigii and validation by antibacterial, antioxidant, and molecular docking studies. Biomass Convers. Biorefinery 2025, 15, 23835–23863. [Google Scholar] [CrossRef]
- Hassan, W.H.B.; Ghani, A.E.A.; Taema, E.A.; Yahya, G.; El-Sadek, M.E.; Mansour, B.; Abdel-Halim, M.S.; Arafa, A.M. Chemical profile, virtual screening, and virulence-inhibiting properties of Sphagneticola trilobata L. essential oils against Pseudomonas aeruginosa. Sci. Rep. 2025, 15, 11964. [Google Scholar] [CrossRef] [PubMed]
- Ben Hassine, D.; El Euch, S.K.; Rahmani, R.; Ghazouani, N.; Kane, R.; Abderrabba, M.; Bouajila, J. Clove Buds Essential Oil: The Impact of Grinding on the Chemical Composition and Its Biological Activities Involved in Consumer’s Health Security. BioMed Res. Int. 2021, 2021, 9940591. [Google Scholar] [CrossRef]
- Qiu, B.; Wei, F.; Su, J.; Hao, W.; Zhou, J.; Zhao, J.; Wang, Y.; Qu, Z. The Effects of β-Pinene, a Pine Needle Oil Monoterpene, on Adenovirus Type 3. Bull. Exp. Biol. Med. 2022, 172, 345–351. [Google Scholar] [CrossRef]
- Madia, V.N.; De Angelis, M.; De Vita, D.; Messore, A.; De Leo, A.; Ialongo, D.; Tudino, V.; Saccoliti, F.; De Chiara, G.; Garzoli, S.; et al. Investigation of Commiphora myrrha (Nees) Engl. Oil and Its Main Components for Antiviral Activity. Pharmaceuticals 2021, 14, 243. [Google Scholar] [CrossRef]
- Zanello, P.R.; Koishi, A.C.; Rezende, C.D.; Oliveira, L.A.; Pereira, A.A.; de Almeida, M.V.; dos Santos, C.N.D.; Bordignon, J. Quinic acid derivatives inhibit dengue virus replication in vitro. Virol. J. 2015, 12, 223. [Google Scholar] [CrossRef]
- Han, J.C.; Zhu, X.Y.; Gao, Z.H.; Xiao, Y.; Zhang, J.X.; Wang, P.; Fang, J.B.; Li, Y.Q.; Zhu, Y.L.; Li, Y.; et al. Antiviral effects of Atractyloside A on the influenza B virus (Victoria strain) infection. Front. Microbiol. 2023, 13, 1067725. [Google Scholar] [CrossRef]
- Nemoto, K.; Takikawa, H.; Ogura, Y. Syntheses of (+)-costic acid and structurally related eudesmane sesquiterpenoids and their biological evaluations as acaricidal agents against Varroa destructor. J. Pestic. Sci. 2023, 48, 111–115. [Google Scholar] [CrossRef]
- Rodríguez-Morales, P.; Franklin, R.A. Macrophage phenotypes and functions: Resolving inflammation and restoring homeostasis. Trends Immunol. 2023, 44, 986–998. [Google Scholar] [CrossRef]
- Citrin, K.M.; Chaube, B.; Fernández-Hernando, C.; Suárez, Y. Intracellular endothelial cell metabolism in vascular function and dysfunction. Trends Endocrinol. Metab. TEM 2025, 36, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Silveira Rossi, J.L.; Barbalho, S.M.; Reverete de Araujo, R.; Bechara, M.D.; Sloan, K.P.; Sloan, L.A. Metabolic syndrome and cardiovascular diseases: Going beyond traditional risk factors. Diabetes/Metab. Res. Rev. 2022, 38, e3502. [Google Scholar] [CrossRef] [PubMed]
- Oda, H.; Annibaldi, A.; Kastner, D.L.; Aksentijevich, I. Genetic Regulation of Cell Death: Insights from Autoinflammatory Diseases. Annu. Rev. Immunol. 2025, 43, 313–342. [Google Scholar] [CrossRef] [PubMed]
- Mayerhofer, C.; Freedman, R.A.; Parsons, H.A.; Partridge, A.H.; Miller, P.G. Clonal Hematopoiesis in Women with Breast Cancer. J. Clin. Oncol. 2025, 43, 861–867. [Google Scholar] [CrossRef]
- Zhao, X.L.; Yu, L.; Zhang, S.D.; Ping, K.; Ni, H.Y.; Qin, X.Y.; Zhao, C.J.; Wang, W.; Efferth, T.; Fu, Y.J. Cryptochlorogenic acid attenuates LPS-induced inflammatory response and oxidative stress via upregulation of the Nrf2/HO-1 signaling pathway in RAW 264.7 macrophages. Int. Immunopharmacol. 2020, 83, 106436. [Google Scholar] [CrossRef]
- Liu, Y.H.; Tang, X.; Zhang, H.Z.; Zheng, L.Y.; Lai, P.; Guo, C.; Ma, J.F.; Chen, H.B.; Qiu, L.X. Terpinen-4-ol Improves Lipopolysaccharide-Induced Macrophage Inflammation by Regulating Glutamine Metabolism. Foods 2024, 13, 1842. [Google Scholar] [CrossRef]
- An, X.; Yu, W.; Liu, J.; Tang, D.; Yang, L.; Chen, X. Oxidative cell death in cancer: Mechanisms and therapeutic opportunities. Cell Death Dis. 2024, 15, 556. [Google Scholar] [CrossRef]
- Huang, X.; Xie, M.; Wang, Y.; Lu, X.; Mei, F.; Zhang, K.; Yang, X.; Chen, G.; Yin, Y.; Feng, G.; et al. Porphyromonas gingivalis aggravates atherosclerotic plaque instability by promoting lipid-laden macrophage necroptosis. Signal Transduct. Target. Ther. 2025, 10, 171. [Google Scholar] [CrossRef]
- Dhapola, R.; Beura, S.K.; Sharma, P.; Singh, S.K.; HariKrishnaReddy, D. Oxidative stress in Alzheimer’s disease: Current knowledge of signaling pathways and therapeutics. Mol. Biol. Rep. 2024, 51, 48. [Google Scholar] [CrossRef]
- Takatsuka, M.; Goto, S.; Kobayashi, K.; Otsuka, Y.; Shimada, Y. Evaluation of pure antioxidative capacity of antioxidants: ESR spectroscopy of stable radicals by DPPH and ABTS assays with singular value decomposition. Food Biosci. 2022, 48, 101714. [Google Scholar] [CrossRef]
- Yang, X.; Liu, Y.; Cao, J.; Wu, C.; Tang, L.; Bian, W.; Chen, Y.; Yu, L.; Wu, Y.; Li, S.; et al. Targeting epigenetic and post-translational modifications of NRF2: Key regulatory factors in disease treatment. Cell Death Discov. 2025, 11, 189. [Google Scholar] [CrossRef] [PubMed]
- Tacke, F.; Puengel, T.; Loomba, R.; Friedman, S.L. An integrated view of anti-inflammatory and antifibrotic targets for the treatment of NASH. J. Hepatol. 2023, 79, 552–566. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Y.; Li, D.; Yang, T. Atractylodin ameliorates lipopolysaccharide-induced depressive-like behaviors in mice through reducing neuroinflammation and neuronal damage. J. Neuroimmunol. 2024, 390, 578349. [Google Scholar] [CrossRef] [PubMed]
- Song, G.Y.; Kim, S.M.; Back, S.; Yang, S.B.; Yang, Y.M. Atractylodes Lancea and Its Constituent, Atractylodin, Ameliorates Metabolic Dysfunction-Associated Steatotic Liver Disease via AMPK Activation. Biomol. Ther. 2024, 32, 778–792. [Google Scholar] [CrossRef]
- Tsai, M.C.; Wang, C.C.; Tsai, I.N.; Yu, M.H.; Yang, M.Y.; Lee, Y.J.; Chan, K.C.; Wang, C.J. Improving the Effects of Mulberry Leaves and Neochlorogenic Acid on Glucotoxicity-Induced Hepatic Steatosis in High Fat Diet Treated db/db Mice. J. Agric. Food Chem. 2024, 72, 6339–6346. [Google Scholar] [CrossRef] [PubMed]
- Akcakavak, G.; Kazak, F.; Deveci, M.Z.Y. Eucalyptol Protects against Cisplatin-Induced Liver Injury in Rats. Biol. Bull. 2023, 50, 987–994. [Google Scholar] [CrossRef]
- Li, Y.J.; Guo, M.Y.; Qin, W.Q.; Li, J.N.; Li, Y.F.; Zhang, F.K.; Xue, X.Y.; Li, S.; Qu, J.R.; Liu, R.P.; et al. Senkyunolide A ameliorates cholestatic liver fibrosis by controlling CLCC1-mediated endoplasmic reticulum Ca2+ release. Acta Pharmacol. Sin. 2025, 46, 3257–3272. [Google Scholar] [CrossRef]
- Sun, J.; Jiang, Y.; Wang, B.; Yang, J.; Chen, Y.; Luo, H.; Chen, T.; Xiao, C.; Weng, L. Structural characterization of the polysaccharides from Atractylodes chinensis (DC.) Koidz. and the protective effection against alcohol-induced intestinal injury in rats. Int. J. Biol. Macromol. 2024, 282, 136641. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Wang, J.; Jiang, Z.; Zhang, L.; Cui, Y.; Zhao, D.; Wang, Y. Atractylenolide II inhibits tumor-associated macrophages (TAMs)-induced lung cancer cell metastasis. Immunopharmacol. Immunotoxicol. 2022, 44, 227–237. [Google Scholar] [CrossRef]
- Wu, Y.; Dai, S.; Zhang, Y.; Li, Z.; Zhu, B.; Liu, Q.; Wo, L.; Yu, Z.; Yuan, X.; Dou, X. Atractylenolide II combined with Interferon-γ synergistically ameliorates colorectal cancer progression in vivo and in vitro by blocking the NF-kB p65/PD-L1 pathway. J. Cancer 2024, 15, 4328–4344. [Google Scholar] [CrossRef]
- Fan, M.; Gu, X.; Zhang, W.; Shen, Q.; Zhang, R.; Fang, Q.; Wang, Y.; Guo, X.; Zhang, X.; Liu, X. Atractylenolide I ameliorates cancer cachexia through inhibiting biogenesis of IL-6 and tumour-derived extracellular vesicles. J. Cachexia Sarcopenia Muscle 2022, 13, 2724–2739. [Google Scholar] [CrossRef] [PubMed]
- Bourebaba, N.; Kornicka-Garbowska, K.; Marycz, K.; Bourebaba, L.; Kowalczuk, A. Laurus nobilis ethanolic extract attenuates hyperglycemia and hyperinsulinemia-induced insulin resistance in HepG2 cell line through the reduction of oxidative stress and improvement of mitochondrial biogenesis—Possible implication in pharmacotherapy. Mitochondrion 2021, 59, 190–213. [Google Scholar] [CrossRef] [PubMed]
- Paul, R.K.; Ahmad, I.; Patel, H.; Raza, K. Antidiabetic activity of the extracted oil from an Indian indigenous plant, Amberboa ramosa: Evidences from in silico, in vitro and enzyme inhibition kinetic studies. Chem. Pap. 2025, 79, 171–180. [Google Scholar] [CrossRef]
- Kim, D.Y.; Kang, M.K.; Lee, E.J.; Kim, Y.H.; Oh, H.; Kim, S.I.; Oh, S.Y.; Na, W.; Kang, Y.H. Eucalyptol Inhibits Amyloid-β-Induced Barrier Dysfunction in Glucose-Exposed Retinal Pigment Epithelial Cells and Diabetic Eyes. Antioxidants 2020, 9, 1000. [Google Scholar] [CrossRef]
- Xu, Z.; Zhu, J.; Gao, P.; Zhu, X.; Zhang, Y.; Liu, X. Essential Oil from Tetrapanax papyrifer (Hook.) K. Koch: Chemical Composition, Antioxidant Activity, α-Glucosidase Inhibitory Effect Integrating Molecular Docking Analysis. Chem. Biodivers. 2025, 22, e202402533. [Google Scholar] [CrossRef]
- Santos, E.S.; Abrantes Coelho, G.L.; Saraiva Fontes Loula, Y.K.; Saraiva Landim, B.L.; Fernandes Lima, C.N.; Tavares de Sousa Machado, S.; Pereira Lopes, M.J.; Soares Gomes, A.D.; Martins da Costa, J.G.; Alencar de Menezes, I.R.; et al. Hypoglycemic, Hypolipidemic, and Anti-Inflammatory Effects of Beta-Pinene in Diabetic Rats. Evid.-Based Complement. Altern. Med. eCAM 2022, 2022, 8173307. [Google Scholar] [CrossRef]
- Wu, W.; Liao, Y.; Wei, L.; Feng, X.; Dai, Y.; Liu, Q.; Feng, S. Ultrasound-Optimized Extraction and Multi-Target Mechanistic Analysis of Antioxidant and Hypoglycemic Effects of Amomum villosum Essential Oil. Foods 2025, 14, 2772. [Google Scholar] [CrossRef]
- El Azab, E.F.; Mostafa, H.S. Geraniol ameliorates the progression of high fat-diet/streptozotocin-induced type 2 diabetes mellitus in rats via regulation of caspase-3, Bcl-2, and Bax expression. J. Food Biochem. 2022, 46, e14142. [Google Scholar] [CrossRef]
- Zhou, Y. The Protective Effects of Cryptochlorogenic Acid on β-Cells Function in Diabetes in vivo and vitro via Inhibition of Ferroptosis. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 1921–1931. [Google Scholar] [CrossRef]
- Shaedi, N.; Naharudin, I.; Choo, C.Y.; Wong, T.W. Design of oral intestinal-specific alginate-vitexin nanoparticulate system to modulate blood glucose level of diabetic rats. Carbohydr. Polym. 2021, 254, 117312. [Google Scholar] [CrossRef]
- Xue, J.C.; Yuan, S.; Meng, H.; Hou, X.T.; Li, J.; Zhang, H.M.; Chen, L.L.; Zhang, C.H.; Zhang, Q.G. The role and mechanism of flavonoid herbal natural products in ulcerative colitis. Biomed. Pharmacother. 2023, 158, 114086. [Google Scholar] [CrossRef]
- Qu, L.; Lin, X.; Liu, C.; Ke, C.; Zhou, Z.; Xu, K.; Cao, G.; Liu, Y. Atractylodin Attenuates Dextran Sulfate Sodium-Induced Colitis by Alleviating Gut Microbiota Dysbiosis and Inhibiting Inflammatory Response Through the MAPK Pathway. Front. Pharmacol. 2021, 12, 665376. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.X.; Liu, J.; Li, F. Hinesol attenuates DSS-induced ulcerative colitis through the suppression of Src-mediated NF-κB and chemokine signaling pathway. Cell Biochem. Biophys. 2024, 82, 2747–2757. [Google Scholar] [CrossRef] [PubMed]
- Shentu, C.; Mao, M.; Zhu, J.; Meng, Q.; Qian, H.; Li, X.; Zhang, S.; Ding, B.; Dai, S.; Yuan, X. Atractylenolide III Ameliorates Ulcerative Colitis By Targeting IL-17RA to Suppress Macrophage M1 Polarization. J. Agric. Food Chem. 2025, 73, 30761–30776. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Wan, Q.; Chen, C. Atractylenolide III Promotes Astrocyte Aβ Clearance by Up-regulating AQP4 to Improve Alzheimer’s Disease. Folia Biol. 2025, 71, 140–148. [Google Scholar] [CrossRef]
- Sun, T.; Li, Z.; Xiao, B.; Yang, J.; Han, M.; Zhang, J.; Liu, S.; Ma, H.; Song, J.; Su, Y.; et al. Multi-target neuroprotection of Atractylodes macrocephala ethyl acetate extract against Alzheimer’s disease: From bioactivity-guided screening to mechanistic validation. J. Ethnopharmacol. 2026, 355, 120735. [Google Scholar] [CrossRef]
- Nallasamy, P.; Srinivasan, G.; Thatchanamoorthy, T.; Jeyaraj, W.; Natarajan, S. Dual functionality of hydroxyapatite nanocarriers in neurodegenerative biomarker management as immunosensor and neuroprotective therapeutic system. Microchem. J. 2025, 208, 112358. [Google Scholar] [CrossRef]
- Meyer, Z.A.; Rambharose, S. Gold nanoparticles incorporating rutin hydrate for targeting oxidative stress-driven neurodegeneration. BioMetals 2026, 39, 203–229. [Google Scholar] [CrossRef]
- Garlet, Q.I.; Rodrigues, P.; Barbosa, L.B.; Londero, A.L.; Mello, C.F.; Heinzmann, B.M. Nectandra grandiflora essential oil and its isolated sesquiterpenoids minimize anxiety-related behaviors in mice through GABAergic mechanisms. Toxicol. Appl. Pharmacol. 2019, 375, 64–80. [Google Scholar] [CrossRef]
- Nguyen, L.T.H.; Nguyen, N.P.K.; Tran, K.N.; Choi, H.J.; Moon, I.S.; Shin, H.M.; Yang, I.J. Essential oil of Pterocarpus santalinus L. alleviates behavioral impairments in social defeat stress-exposed mice by regulating neurotransmission and neuroinflammation. Biomed. Pharmacother. 2024, 171, 116164. [Google Scholar] [CrossRef]
- Asle-Rousta, M.; Abdollahi, M.; Aghajari, H.M.; Peirovy, Y. Eucalyptol Attenuates Lead-Induced Anxiety-like Behaviors by Suppressing Oxidative Stress and Neuroinflammation, Modulating SIRT1/NF-κB Signaling, and Upregulating BDNF Expression. Biol. Trace Elem. Res. 2025, 203, 5942–5953. [Google Scholar] [CrossRef]
- Hosseini, A.; Pourheidar, E.; Rajabian, A.; Asadpour, E.; Hosseinzadeh, H.; Sadeghnia, H.R. Linalool attenuated ischemic injury in PC12 cells through inhibition of caspase-3 and caspase-9 during apoptosis. Food Sci. Nutr. 2023, 11, 249–260. [Google Scholar] [CrossRef]
- Wee, A.S.; Thew, H.Y.; Liew, S.Y.; Tan, W.N.; Khaw, K.Y. Anti-cholinesterase profile of Murraya koenigii (L.) Spreng essential oil and its chemical constituents. J. Essent. Oil Bear. Plants 2024, 27, 1449–1459. [Google Scholar] [CrossRef]
- Nallasamy, P.; Rajamohamed, B.S.; Jeyaraman, J.; Kathirvel, B.; Natarajan, S. Regenerative marine waste towards CaCO3 nanoformulation for Alzheimer’s therapy. Environ. Res. 2023, 225, 115631. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.I.; Choi, J.H.; Kwon, T.W.; Jo, H.S.; Kim, D.G.; Ko, S.G.; Song, G.J.; Cho, I.H. Neuroprotective effects of bornyl acetate on experimental autoimmune encephalomyelitis via anti-inflammatory effects and maintaining blood-brain-barrier integrity. Phytomedicine 2023, 112, 154569. [Google Scholar] [CrossRef]
- Faheem, M.; Khan, A.U.; Saleem, M.W.; Shah, F.A.; Ali, F.; Khan, A.W.; Li, S. Neuroprotective Effect of Natural Compounds in Paclitaxel-Induced Chronic Inflammatory Pain. Molecules 2022, 27, 4926. [Google Scholar] [CrossRef] [PubMed]
- Faheem, M.; Khan, A.U.; Shah, F.A.; Li, S. Investigation of Natural Compounds for Therapeutic Potential in Streptozotocin-induced Diabetic Neuroinflammation and Neuropathic Pain. Front. Pharmacol. 2022, 13, 1019033. [Google Scholar] [CrossRef] [PubMed]
- Latif, K.; Saneela, S.; Khan, A.U. Ameliorative effect of carveol on scopolamine-induced memory impairment in rats. Iran. J. Basic Med. Sci. 2022, 25, 1504–1512. [Google Scholar] [CrossRef]
- Muhammad, A.J.; Al-Baqami, F.F.; Alanazi, F.E.; Alattar, A.; Alshaman, R.; Rehman, N.U.; Riadi, Y.; Shah, F.A. The Interplay of Carveol and All-Trans Retinoic Acid (ATRA) in Experimental Parkinson’s Disease: Role of Inflammasome-Mediated Pyroptosis and Nrf2. Neurochem. Res. 2024, 49, 3118–3130. [Google Scholar] [CrossRef]
- Rafi, K.; Faizi, S.; Hussain, S.S.; Shamshad, S.; Versiani, M.A.; Simjee, S.U. Quinic acid and its derivatives protect against phytohaemagglutinin-induced Alzheimer’s-like neurotoxicity in SH-SY5Y cells by down-regulating p38 MAPK signaling pathway. Toxicol. Appl. Pharmacol. 2025, 504, 117539. [Google Scholar] [CrossRef]
- Niaz, M.; Iftikhar, K.; Shahid, M.; Faizi, S.; Usman Simjee, S. Quinic acid contributes to neurogenesis: Targeting Notch pathway a key player in hippocampus. Brain Res. 2025, 1846, 149291. [Google Scholar] [CrossRef]
- Li, S.; Cai, Y.; Guan, T.; Zhang, Y.; Huang, K.; Zhang, Z.; Cao, W.; Guan, X. Quinic acid alleviates high-fat diet-induced neuroinflammation by inhibiting DR3/IKK/NF-κB signaling via gut microbial tryptophan metabolites. Gut Microbes 2024, 16, 2374608. [Google Scholar] [CrossRef]
- Wang, C.; Song, X.; Zhang, X.; Li, P.; Wei, W.; Sun, S.; Chen, Y. Multifunctional natural chlorogenic acid based nanocarrier for Alzheimer’s disease treatment. Mater. Today Bio 2025, 32, 101841. [Google Scholar] [CrossRef] [PubMed]
- Qiao, N.; Wang, Q.; Tao, Y.; Wu, J.; Fang, Y.; Ni, Y.; Ding, X. α-Cyperone ameliorates depression in mammary gland hyperplasia and chronic unpredictable mild stress rat by regulating hormone, inflammation, and oxidative stress. Immunopharmacol. Immunotoxicol. 2023, 45, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Hu, G.; Zong, X.; Yang, S.; He, D.; Gao, X.; Liu, D. α-Cyperone protects dopaminergic neurons and inhibits neuroinflammation in LPS-induced Parkinson’s disease rat model via activating Nrf2/HO-1 and suppressing NF-κB signaling pathway. Int. Immunopharmacol. 2023, 115, 109698. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Miao, L.; Peng, Q.; Fan, X.; Song, W.; Yang, B.; Zhang, P.; Liu, G.; Liu, J. Parthenolide modulates cerebral ischemia-induced microglial polarization and alleviates neuroinflammatory injury via the RhoA/ROCK pathway. Phytomedicine 2022, 105, 154373. [Google Scholar] [CrossRef]
- Ding, W.; Cai, C.; Zhu, X.; Wang, J.; Jiang, Q. Parthenolide ameliorates neurological deficits and neuroinflammation in mice with traumatic brain injury by suppressing STAT3/NF-κB and inflammasome activation. Int. Immunopharmacol. 2022, 108, 108913. [Google Scholar] [CrossRef]
- Park, C.K.; Choi, S.J.; Kim, C.R.; Shin, H.R.; Shin, E.C.; Kim, Y.J.; Cho, T.J.; Shin, D.H.; Kim, J.K. Ethanolic Extract of Rosa rugosa Roots and Its Bioactive Compound, Oleamide, Prevented Amyloid β-Induced Oxidative Stress and Improved Behavioral Tests in Mice. Int. J. Mol. Sci. 2025, 26, 4214. [Google Scholar] [CrossRef]
- Shervin Prince, S.; Stanely Mainzen Prince, P.; Berlin Grace, V.M. Valencene post-treatment exhibits cardioprotection via inhibiting cardiac hypertrophy, oxidative stress, nuclear factor- κB inflammatory pathway, and myocardial infarct size in isoproterenol-induced myocardial infarcted rats; A molecular study. Eur. J. Pharmacol. 2022, 927, 174975. [Google Scholar] [CrossRef]
- Song, X.; Wang, L.; Liu, M.; Pan, R.; Song, J.; Kong, J. Atractylenolide II ameliorates myocardial fibrosis and oxidative stress in spontaneous hypertension rats. Technol. Health Care 2024, 32, 131–142. [Google Scholar] [CrossRef]
- Li, J.; Chen, X.; Li, X.; Tang, J.; Li, Y.; Liu, B.; Guo, S. Cryptochlorogenic acid and its metabolites ameliorate myocardial hypertrophy through a HIF1α-related pathway. Food Funct. 2022, 13, 2269–2282. [Google Scholar] [CrossRef] [PubMed]
- Stamatiou, R.; Anagnostopoulou, M.; Ioannidou-Kabouri, K.; Rapti, C.; Lazou, A. Camphene as a Protective Agent in Myocardial Ischemia/Reperfusion Injury. Antioxidants 2024, 13, 405. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Chen, X.; Sun, Q.; Li, J.; Wang, Q.; Wei, P.; Wang, W.; Li, C.; Wang, Y. Valerenic acid attenuates pathological myocardial hypertrophy by promoting the utilization of multiple substrates in the mitochondrial energy metabolism. J. Adv. Res. 2025, 68, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, B.; Cai, Q. Study on the pharmacodynamics and metabolomics of five medicinal species in Atractylodes DC. on rats with rheumatoid arthritis. Biomed. Pharmacother. 2020, 131, 110554. [Google Scholar] [CrossRef]
- Chuang, C.H.; Cheng, Y.C.; Lin, S.C.; Lehman, C.W.; Wang, S.P.; Chen, D.Y.; Tsai, S.W.; Lin, C.C. Atractylodin Suppresses Dendritic Cell Maturation and Ameliorates Collagen-Induced Arthritis in a Mouse Model. J. Agric. Food Chem. 2019, 67, 6773–6784. [Google Scholar] [CrossRef]
- Pan, J.; Cai, Y.; Zhang, C.; Xu, S. Intra-articular delivery of geraniol encapsulated by pH/redox-responsive nanogel ameliorates osteoarthritis by regulating oxidative stress and inflammation. J. Mol. Histol. 2023, 54, 579–591. [Google Scholar] [CrossRef]
- Yin, C.; Liu, B.; Wang, P.; Li, X.; Li, Y.; Zheng, X.; Tai, Y.; Wang, C.; Liu, B. Eucalyptol alleviates inflammation and pain responses in a mouse model of gout arthritis. Br. J. Pharmacol. 2020, 177, 2042–2057. [Google Scholar] [CrossRef]
- Liu, D.; Fu, Q.; Liu, L.G.; Li, W.; Qi, F.; Liu, J.; Shang, L.; Wang, X.; Yang, F.; Li, J.; et al. Screening of potentially active compounds against rheumatoid arthritis in the Juan-Bi decoction using systems pharmacology and animal experiments. Front. Cell Dev. Biol. 2024, 12, 1396890. [Google Scholar] [CrossRef]
- Chen, S.; Xu, H.; He, Y.; Meng, C.; Fan, Y.; Qu, Y.; Wang, Y.; Zhou, W.; Huang, X.; You, H. Carveol alleviates osteoarthritis progression by acting on synovial macrophage polarization transformation: An in vitro and in vivo study. Chem.-Biol. Interact. 2024, 387, 110781. [Google Scholar] [CrossRef]
- Shao, M.; Lv, D.; Zhou, K.; Sun, H.; Wang, Z. Senkyunolide A inhibits the progression of osteoarthritis by inhibiting the NLRP3 signalling pathway. Pharm. Biol. 2022, 60, 535–542. [Google Scholar] [CrossRef]
- Shi, K.; Wang, Y.; Xiao, Y.; Tu, J.; Zhou, Z.; Cao, G.; Liu, Y. Therapeutic effects and mechanism of Atractylodis rhizoma in acute lung injury: Investigation based on an Integrated approach. Front. Pharmacol. 2023, 14, 1181951. [Google Scholar] [CrossRef]
- Xing, H.; Bai, X.; Pei, X.; Zhang, Y.; Zhang, X.; Chen, S.; Li, D.; Lv, B.; Wang, X.; Wu, X. Synergistic anti-oxidative/anti-inflammatory treatment for acute lung injury with selenium based chlorogenic acid nanoparticles through modulating Mapk8ip1/MAPK and Itga2b/PI3k-AKT axis. J. Nanobiotechnol. 2025, 23, 37. [Google Scholar] [CrossRef]
- Yang, S.; Ling, Y.; Zhao, F.; Li, W.; Song, Z.; Wang, L.; Li, Q.; Liu, M.; Tong, Y.; Chen, L.; et al. Hymecromone: A clinical prescription hyaluronan inhibitor for efficiently blocking COVID-19 progression. Signal Transduct. Target. Ther. 2022, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, X.; Zhao, L.; Pan, B.; Xu, X.; Zhu, D. Dehydrocostus Lactone Ameliorates LPS-Induced Acute Lung Injury by Inhibiting PFKFB3-Mediated Glycolysis. J. Cell. Biochem. 2024, 125, e30639. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Dong, J.; Xu, C.; Ni, Y.; Ye, Z.; Sun, Z.; Fan, H.; Chen, Y. Acute lung injury: Pathogenesis and treatment. J. Transl. Med. 2025, 23, 926. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Choi, J.; Seo, J.; Lim, H.; Kang, S.K. CKD-497 inhibits NF-kB signaling and ameliorates inflammation and pulmonary fibrosis in ovalbumin-induced asthma and particulate matter-induced airway inflammatory diseases. Front. Pharmacol. 2024, 15, 1428567. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, C.; Zhao, R.; Yuan, J. Senkyunolide A alleviates asthma by inhibiting TGF-β/Smad2/3 signaling pathway and NLRP3 inflammasome activation. Biochem. Biophys. Res. Commun. 2025, 788, 152809. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, S.; Li, Y.; Zhou, Z.; Zhang, X.; Chen, Y.; Zhao, Y.; Ye, C.; Li, J.; Zhang, N.; et al. Structural characterization and effect of activating autophagy and regulating oxidative stress of polysaccharide from fibrous roots of Atractylodes chinensis. Bioorganic Chem. 2025, 162, 108609. [Google Scholar] [CrossRef]
- Plirat, W.; Chaniad, P.; Phuwajaroanpong, A.; Septama, A.W.; Punsawad, C. Phytochemical, Antimalarial, and Acute Oral Toxicity Properties of Selected Crude Extracts of Prabchompoothaweep Remedy in Plasmodium berghei-Infected Mice. Trop. Med. Infect. Dis. 2022, 7, 395. [Google Scholar] [CrossRef]
- Wang, K.-T.; Chen, L.-G.; Yang, L.-L.; Ke, W.-M.; Chang, H.-C.; Wang, C.-C. Analysis of the sesquiterpenoids in processed atractylodis rhizoma. Chem. Pharm. Bull. 2007, 55, 50–56. [Google Scholar] [CrossRef]
- Shi, K.; Xiao, Y.; Dong, Y.; Wang, D.; Xie, Y.; Tu, J.; Xu, K.; Zhou, Z.; Cao, G.; Liu, Y. Protective Effects of Atractylodis lancea Rhizoma on Lipopolysaccharide-Induced Acute Lung Injury via TLR4/NF-κB and Keap1/Nrf2 Signaling Pathways In Vitro and In Vivo. Int. J. Mol. Sci. 2022, 23, 16134. [Google Scholar] [CrossRef]
- Gao, X.; Ma, D.; Li, K.; Xing, T.; Liu, X.; Peng, L.; Chen, D.; Hao, Z. Non-Targeted Metabolomics Combined with Chemometrics by UHPLC-Orbitrap-HRMS and Antioxidant Activity of Atractylodes chinensis (DC.) Koidez. from Eight Origins. Metabolites 2023, 13, 888. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, Z.; Zhou, Z.; Zhang, N.; Cui, X.; Yu, X.; Zhao, Y.; Wang, Z.; Li, J.; Wu, D. Differential toxic phenotypes and liver injury induced by Atractylenolides (I, II, and III): Insights from zebrafish (Danio rerio) models and network toxicology. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2026, 299, 110365. [Google Scholar] [CrossRef]
- Tshering, G.; Plengsuriyakarn, T.; Na-Bangchang, K.; Pimtong, W. Embryotoxicity evaluation of atractylodin and β-eudesmol using the zebrafish model. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021, 239, 108869. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.