In Vitro Evaluation of Natural Sesquiterpene Lactones and Naphthoquinones Against Pancreatic Ductal Adenocarcinoma Cells
Abstract
1. Introduction
2. Results
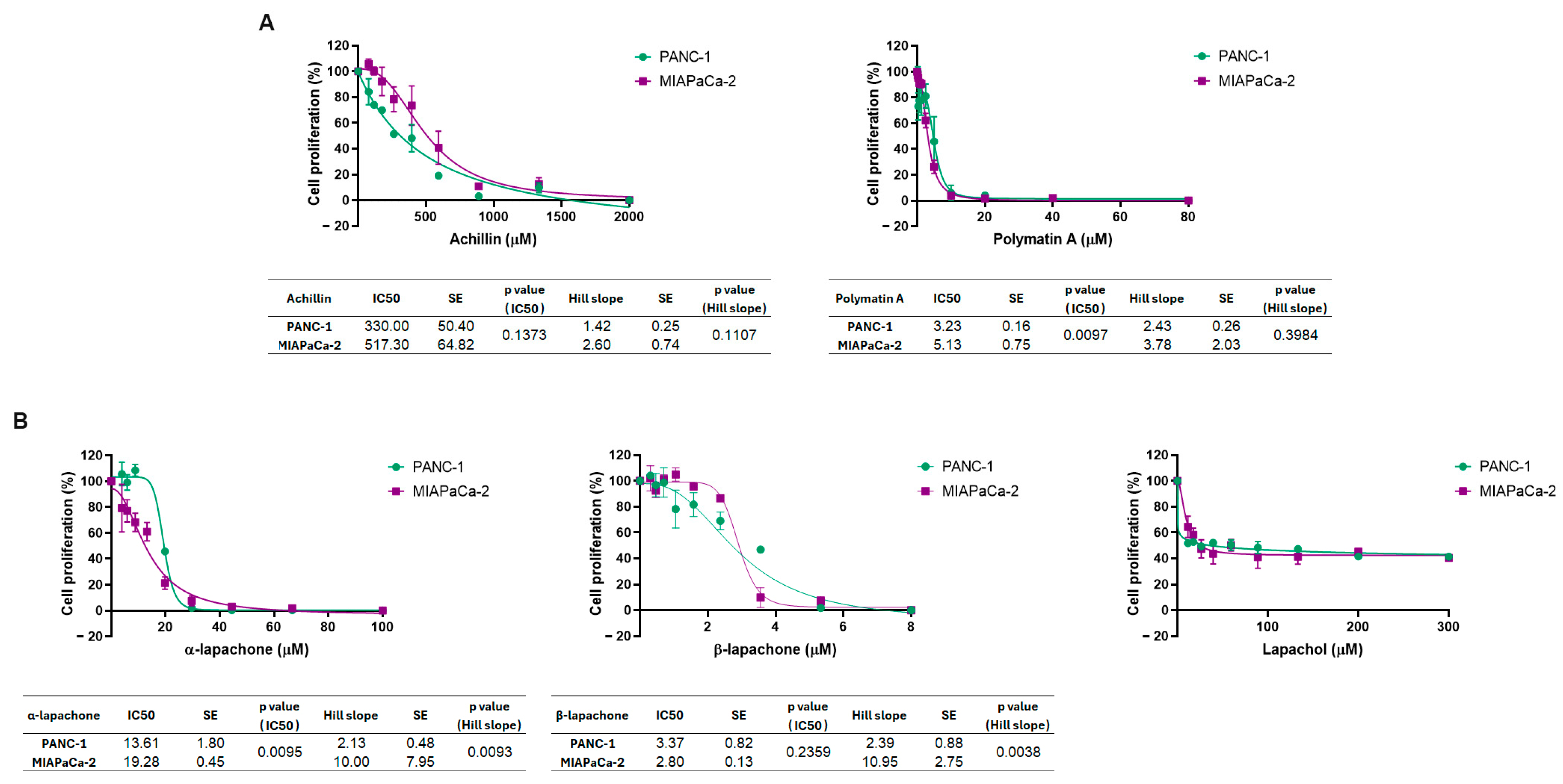
2.1. Assessment of Cell Proliferation Inhibition
2.2. Effects of Natural Compounds on Cellular Redox Metabolic Activity
2.3. Early Mitochondrial Alterations Induced by Natural Compounds
2.4. Cell Death Induction
3. Discussion
Final Considerations and Future Directions
4. Materials and Methods
4.1. Reagents
4.2. Cell Cultures
4.3. Cell Treatments
4.4. Cell Proliferation Assay
4.5. Metabolic Activity Assay
4.6. Assessment of Mitochondrial Membrane Potential and Superoxide Production
4.7. Cell Death Assay
4.8. Statistical Analysis
4.9. Chemical Structure Generation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATCC | American Type Culture Collection |
| BrdU | 5-bromo-2′-deoxyuridine |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DMSO | Dimethyl sulfoxide |
| EC50 | Half-maximal effective concentration |
| FBS | Fetal bovine serum |
| HRP | Horseradish peroxidase |
| IC50 | Half-maximal inhibitory concentration |
| LC50 | Half-maximal lethal concentration |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NF-κB | Nuclear factor kappa B |
| NQO1 | NAD(P)H:quinone oxidoreductase 1 |
| PARP1 | Poly(ADP-ribose) polymerase 1 |
| PBMCs | Peripheral blood mononuclear cells |
| PBS | Phosphate-buffered saline |
| PDAC | Pancreatic ductal adenocarcinoma |
| PI | Propidium iodide |
| ROS | Reactive oxygen species |
| STLs | Sesquiterpene lactones |
| XTT | Sodium 3′-[1-(phenylaminocarbonyl)-3,4-tetrazolium]-bis(4-methoxy-6-nitro)benzene sulfonic acid hydrate |
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Ghaneh, P.; Costello, E.; Neoptolemos, J.P. Biology and management of pancreatic cancer. Postgrad. Med. J. 2007, 84, 478–497. [Google Scholar] [CrossRef] [PubMed]
- Uccello, M.; Moschetta, M.; Mak, G.; Alam, T.; Henriquez, C.M.; Arkenau, H.-T. Towards an optimal treatment algorithm for metastatic pancreatic ductal adenocarcinoma (PDA). Curr. Oncol. 2018, 25, 90–94. [Google Scholar] [CrossRef]
- Desam, N.R.; Al-Rajab, A.J. Herbal biomolecules: Anticancer agents. In Herbal Biomolecules in Healthcare; Academic Press: Cambridge, MA, USA, 2022; pp. 435–474. [Google Scholar] [CrossRef]
- Anesini, C.; Alonso, M.; Martino, R. Antiproliferative and cytotoxic activities. In Sesquiterpene Lactones. Advances in Their Chemistry and Biological Aspects; Sülsen, V., Martino, V., Eds.; Springer: Cham, Switzerland, 2018; pp. 303–323. [Google Scholar]
- Bailly, C. Anticancer Targets and Signaling Pathways Activated by Britannin and Related Pseudoguaianolide Sesquiterpene Lactones. Biomedicines 2021, 9, 1325. [Google Scholar] [CrossRef]
- Cutter, L.R.; Ren, A.R.; Banerjee, I.A. Advances in Naturally and Synthetically Derived Bioactive Sesquiterpenes and Their Derivatives: Applications in Targeting Cancer and Neurodegenerative Diseases. Molecules 2025, 30, 4302. [Google Scholar] [CrossRef]
- Laurella, L.C.; Mirakian, N.T.; García, M.N.; Grasso, D.H.; Sülsen, V.P.; Papademetrio, D.L. Sesquiterpene Lactones as Promising Candidates for Cancer Therapy: Focus on Pancreatic Cancer. Molecules 2022, 27, 3492. [Google Scholar] [CrossRef]
- Beer, M.F.; Bivona, A.E.; Sánchez Alberti, A.; Cerny, N.; Reta, G.F.; Martín, V.S.; Padrón, J.M.; Malchiodi, E.L.; Sülsen, V.P.; Donadel, O.J. Preparation of Sesquiterpene Lactone Derivatives: Cytotoxic Activity and Selectivity of Action. Molecules 2019, 24, 1113. [Google Scholar] [CrossRef]
- Martino, R.; Beer, M.F.; Elso, O.; Donadel, O.; Sülsen, V.; Anesini, C. Sesquiterpene lactones from Ambrosia spp. are active against a murine lymphoma cell line by inducing apoptosis and cell cycle arrest. Toxicol. Vitr. 2015, 29, 1529–1536. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, E.S.; Tajbakhsh, A.; Iranshahy, M.; Asili, J.; Kretschmer, N.; Shakeri, A.; Sahebkar, A. Naphthoquinone Derivatives Isolated from Plants: Recent Advances in Biological Activity. Mini Rev. Med. Chem. 2020, 20, 2019–2035. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Green, I.R. Lapachol and lapachone analogs: A journey of two decades of patent research(1997–2016). Expert Opin. Ther. Pat. 2017, 27, 1111–1121. [Google Scholar] [CrossRef]
- Castellanos, J.R.G.; Prieto, J.M.; Heinrich, M. Red Lapacho (Tabebuia impetiginosa)—A global ethnopharmacological commodity? J. Ethnopharmacol. 2009, 121, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.; Chen, J.W.; Shen, L.S.; Chen, S.; Chen, G.Q. Research advances in natural sesquiterpene lactones: Overcoming cancer drug resistance through modulation of key signaling pathways. Cancer drug Resist. 2025, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Quintana, J.; Estévez, F. Recent Advances on Cytotoxic Sesquiterpene Lactones. Curr. Pharm. Des. 2018, 24, 4355–4361. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Won, Y.K.; Ong, C.N.; Shen, H.M. Anti-cancer potential of sesquiterpene lactones: Bioactivity and molecular mechanisms. Curr. Med. Chem. Anticancer. Agents 2005, 5, 239–249. [Google Scholar] [CrossRef]
- Angulo-Elizari, E.; Henriquez-Figuereo, A.; Morán-Serradilla, C.; Plano, D.; Sanmartín, C. Unlocking the potential of 1,4-naphthoquinones: A comprehensive review of their anticancer properties. Eur. J. Med. Chem. 2024, 268, 116249. [Google Scholar] [CrossRef]
- Njus, D.; Asmaro, K.; Li, G.; Palomino, E. Redox cycling of quinones reduced by ascorbic acid. Chem. Biol. Interact. 2023, 373, 110397. [Google Scholar] [CrossRef]
- Silvers, M.A.; Deja, S.; Singh, N.; Egnatchik, R.A.; Sudderth, J.; Luo, X.; Beg, M.S.; Burgess, S.C.; DeBerardinis, R.J.; Boothman, D.A.; et al. The NQO1 bioactivatable drug, β-lapachone, alters the redox state of NQO1+ pancreatic cancer cells, causing perturbation in central carbon metabolism. J. Biol. Chem. 2017, 292, 18203–18216. [Google Scholar] [CrossRef] [PubMed]
- Pinho, B.R.; Sousa, C.; Valentão, P.; Andrade, P.B. Is nitric oxide decrease observed with naphthoquinones in LPS stimulated RAW 264.7 macrophages a beneficial property? PLoS ONE 2011, 6, e24098. [Google Scholar] [CrossRef]
- Sanchez-Carranza, J.N.; González-Maya, L.; Razo-Hernández, R.S.; Salas-Vidal, E.; Nolasco-Quintana, N.Y.; Clemente-Soto, A.F.; García-Arizmendi, L.; Sánchez-Ramos, M.; Marquina, S.; Alvarez, L. Achillin Increases Chemosensitivity to Paclitaxel, Overcoming Resistance and Enhancing Apoptosis in Human Hepatocellular Carcinoma Cell Line Resistant to Paclitaxel (Hep3B/PTX). Pharmaceutics 2019, 11, 512. [Google Scholar] [CrossRef]
- Gara, R.K.; Srivastava, V.K.; Duggal, S.; Bagga, J.K.; Bhatt, M.; Sanyal, S.; Mishra, D.P. Shikonin selectively induces apoptosis in human prostate cancer cells through the endoplasmic reticulum stress and mitochondrial apoptotic pathway. J. Biomed. Sci. 2015, 22, 26. [Google Scholar] [CrossRef]
- Li, Q.; Wang, Z.; Xie, Y.; Hu, H. Antitumor activity and mechanism of costunolide and dehydrocostus lactone: Two natural sesquiterpene lactones from the Asteraceae family. Biomed. Pharmacother. 2020, 125, 109955. [Google Scholar] [CrossRef]
- Williams, W.; Hall, I.; Grippo, A.; Oswald, C.; Lee, K.; Holbrook, D.; Chaney, S. Inhibition of nucleic acid synthesis in p-388 lymphocytic leukemia tumor cells by helenalin and bis(helenalinyl)malonate in vivo. J. Pharm. Sci. 1988, 77, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Bosco, A.; Golsteyn, R.M. Emerging Anti-Mitotic Activities and Other Bioactivities of Sesquiterpene Compounds upon Human Cells. Molecules 2017, 22, 459. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, G.; Silvers, M.A.; Ilcheva, M.; Liu, Y.; Moore, Z.R.; Luo, X.; Gao, J.; Anderson, G.; Liu, L.; Sarode, V.; et al. Tumor-selective use of DNA base excision repair inhibition in pancreatic cancer using the NQO1 bioactivatable drug, β-lapachone. Sci. Rep. 2015, 5, 17066. [Google Scholar] [CrossRef] [PubMed]
- De Ford, C.; Ulloa, J.L.; Catalán, C.A.; Grau, A.; Martino, V.S.; Muschietti, L.V.; Merfort, I. The sesquiterpene lactone polymatin B from Smallanthus sonchifolius induces different cell death mechanisms in three cancer cell lines. Phytochemistry 2015, 117, 332–339. [Google Scholar] [CrossRef]
- Viera, T.; Patidar, P.L. DNA damage induced by KP372-1 hyperactivates PARP1 and enhances lethality of pancreatic cancer cells with PARP inhibition. Sci. Rep. 2020, 10, 20210. [Google Scholar] [CrossRef]
- Krishnan, P.; Bastow, K.F. Novel mechanism of cellular DNA topoisomerase II inhibition by the pyranonaphthoquinone derivatives alpha-lapachone and beta-lapachone. Cancer Chemother. Pharmacol. 2001, 47, 187–198. [Google Scholar] [CrossRef]
- Yang, Y.; Zhou, X.; Xu, M.; Piao, J.; Zhang, Y.; Lin, Z.; Chen, L. β-lapachone suppresses tumour progression by inhibiting epithelial-to-mesenchymal transition in NQO1-positive breast cancers. Sci. Rep. 2017, 7, 2681. [Google Scholar] [CrossRef]
- Babu, M.S.; Mahanta, S.; Lakhter, A.J.; Hato, T.; Paul, S.; Naidu, S.R. Lapachol inhibits glycolysis in cancer cells by targeting pyruvate kinase M2. PLoS ONE 2018, 13, e0191419. [Google Scholar] [CrossRef]
- Mancini, I.; Vigna, J.; Sighel, D.; Defant, A. Hybrid Molecules Containing Naphthoquinone and Quinolinedione Scaffolds as Antineoplastic Agents. Molecules 2022, 27, 4948. [Google Scholar] [CrossRef]
- Gong, Q.; Hu, J.; Wang, P.; Li, X.; Zhang, X. A comprehensive review on β-lapachone: Mechanisms, structural modifications, and therapeutic potentials. Eur. J. Med. Chem. 2021, 210, 112962. [Google Scholar] [CrossRef]
- Tumbath, S.; Jiang, L.; Li, X.; Zhang, T.; Zahid, K.R.; Zhao, Y.; Zhou, H.; Yin, Z.; Lu, T.; Jiang, S.; et al. β-Lapachone promotes the recruitment and polarization of tumor-associated neutrophils (TANs) toward an antitumor (N1) phenotype in NQO1-positive cancers. Oncoimmunology 2024, 13, 2363000. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, G.; Moore, Z.R.; Luo, X.; Ilcheva, M.; Ali, A.; Padanad, M.; Zhou, Y.; Xie, Y.; Burma, S.; Scaglioni, P.P.; et al. Targeting glutamine metabolism sensitizes pancreatic cancer to PARP-driven metabolic catastrophe induced by ß-lapachone. Cancer Metab. 2015, 3, 12. [Google Scholar] [CrossRef]
- Sülsen, V.P.; Martino, V.S. Overview. In Sesquiterpene Lactones: Advances in Their Chemistry and Biological Aspects; Springer International Publishing: Cham, Switzerland, 2018; pp. 3–17. [Google Scholar] [CrossRef]
- Matos, M.S.; Anastácio, J.D.; Santos, C.N.D. Sesquiterpene Lactones: Promising Natural Compounds to Fight Inflammation. Pharmaceutics 2021, 13, 991. [Google Scholar] [CrossRef] [PubMed]
- Alfhili, M.A.; Alsughayyir, J.; Basudan, A.M. Reprogramming of erythrocyte lifespan by NFκB-TNFα naphthoquinone antagonist β-lapachone is regulated by calcium overload and CK1α. J. Food Biochem. 2021, 45, e13710. [Google Scholar] [CrossRef]
- Miranda, S.E.M.; Lemos, J.d.A.; Fernandes, R.S.; Silva, J.d.O.; Ottoni, F.M.; Townsend, D.M.; Rubello, D.; Alves, R.J.; Cassali, G.D.; Ferreira, L.A.M.; et al. Enhanced antitumor efficacy of lapachol-loaded nanoemulsion in breast cancer tumor model. Biomed. Pharmacother. 2021, 133, 110936. [Google Scholar] [CrossRef]
- Mercado, M.I.; Marcial, G.; Catalán, J.V.; Grau, A.; Catalán, C.A.N.; Ponessa, G.I. Morphoanatomy, histochemistry, essential oil, and other secondary metabolites of Artemisia copa (Asteraceae). Bot. Lett. 2021, 168, 577–593. [Google Scholar] [CrossRef]
- Catalán, J.; Marcial, G.; Schuff, C.; Perotti, M.; Catalán, C. Chemical composition and antioxidant activity of essential oil and extracts from Artemisia copa. Boletín Latinoam. Caribe Plantas Med. Aromat. 2007, 6, 238–239. [Google Scholar]
- Aráoz, M.V.C.; Mercado, M.I.; Grau, A.; Catalán, C.A.N. Intraspecific variation of sesquiterpene lactones associated to a latitudinal gradient in Smallanthus macroscyphus (Heliantheae: Asteraceae). Chemoecology 2016, 26, 143–151. [Google Scholar] [CrossRef]
- De Pedro, A.; Cuenca, M.D.R.; Grau, A.; Catalán, C.A.N.; Gedris, T.E.; Herz, W. Melampolides from Smallanthus macroscyphus. Biochem. Syst. Ecol. 2003, 31, 1067–1071. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mirakian, N.T.; Iácono, R.F.; Pulido, V.B.; Pibuel, M.A.; Lompardía, S.L.; Laurella, L.C.; Pérez-Mauad, N.; Catalán, C.A.N.; Lombardo, T.; Ledesma, M.M.; et al. In Vitro Evaluation of Natural Sesquiterpene Lactones and Naphthoquinones Against Pancreatic Ductal Adenocarcinoma Cells. Molecules 2026, 31, 1014. https://doi.org/10.3390/molecules31061014
Mirakian NT, Iácono RF, Pulido VB, Pibuel MA, Lompardía SL, Laurella LC, Pérez-Mauad N, Catalán CAN, Lombardo T, Ledesma MM, et al. In Vitro Evaluation of Natural Sesquiterpene Lactones and Naphthoquinones Against Pancreatic Ductal Adenocarcinoma Cells. Molecules. 2026; 31(6):1014. https://doi.org/10.3390/molecules31061014
Chicago/Turabian StyleMirakian, Nadia T., Rubén F. Iácono, Viviana B. Pulido, Matías A. Pibuel, Silvina L. Lompardía, Laura C. Laurella, Nicolás Pérez-Mauad, Cesar A. N. Catalán, Tomás Lombardo, Martín M. Ledesma, and et al. 2026. "In Vitro Evaluation of Natural Sesquiterpene Lactones and Naphthoquinones Against Pancreatic Ductal Adenocarcinoma Cells" Molecules 31, no. 6: 1014. https://doi.org/10.3390/molecules31061014
APA StyleMirakian, N. T., Iácono, R. F., Pulido, V. B., Pibuel, M. A., Lompardía, S. L., Laurella, L. C., Pérez-Mauad, N., Catalán, C. A. N., Lombardo, T., Ledesma, M. M., Carlucci, A., Sülsen, V. P., & Papademetrio, D. L. (2026). In Vitro Evaluation of Natural Sesquiterpene Lactones and Naphthoquinones Against Pancreatic Ductal Adenocarcinoma Cells. Molecules, 31(6), 1014. https://doi.org/10.3390/molecules31061014











